User login
Expanded UCB product can stand alone
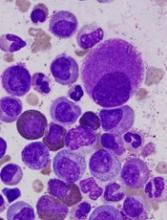
SALT LAKE CITY—The expanded umbilical cord blood (UCB) product NiCord can be used as a stand-alone graft, according to research presented at the 2018 BMT Tandem Meetings.
Researchers found that a single NiCord unit provided “robust” engraftment in a phase 1/2 study of patients with high-risk hematologic malignancies.
NiCord recipients had quicker neutrophil and platelet engraftment than matched control subjects who received standard myeloablative UCB transplant (single or double).
Mitchell Horwitz, MD, of the Duke University Medical Center in Durham, North Carolina, presented these results at the meeting as abstract 49.* The research was sponsored by Gamida Cell, the company developing NiCord.
“[NiCord] is an ex vivo expanded cell product that’s derived from an entire unit of umbilical cord blood,” Dr Horwitz explained. “It’s manufactured starting with a CD133-positive selection, which is the progenitor cell population that’s cultured, and a T-cell containing CD133-negative fraction that is provided also at the time of transplant.”
“The culture system contains nicotinamide—that’s the active ingredient in the culture. And that’s supplemented with cytokines—thrombopoietin, IL-6, FLT-3 ligand, and stem cell factor. The culture is 21 days.”
Previous research showed that double UCB transplant including a NiCord unit could provide benefits over standard double UCB transplant. This led Dr Horwitz and his colleagues to wonder if NiCord could be used as a stand-alone graft.
So the team evaluated the safety and efficacy of NiCord alone in 36 adolescents/adults with high-risk hematologic malignancies.
Patients had acute myelogenous leukemia (n=17), acute lymphoblastic leukemia (n=9), myelodysplastic syndrome (n=7), chronic myelogenous leukemia (n=2), and Hodgkin lymphoma (n=1).
Most patients had intermediate (n=15) or high-risk (n=13) disease. They had a median age of 44 (range, 13-63) and a median weight of 75 kg (range, 41-125).
Treatment
For conditioning, 19 patients received thiotepa, busulfan, and fludarabine. Fifteen patients received total body irradiation and fludarabine with or without cyclophosphamide or thiotepa. And 2 patients received clofarabine, fludarabine, and busulfan.
Most patients had a 4/6 human leukocyte antigen (HLA) match (n=26), 8 had a 5/6 HLA match, and 2 had a 6/6 HLA match.
The median total nucleated cell dose was 2.4 x 107/kg prior to expansion of the UCB unit and 3.7 x 107/kg after expansion. The median CD34+ cell dose was 0.2 x 106/kg and 6.3 x 106/kg, respectively.
“CD34 cells were expanded 33-fold in the 3-week culture system,” Dr Horwitz noted. “That translated to a median CD34 dose of 6.3 x 106/kg, a dose comparable to what would be obtained from an adult donor graft.”
Engraftment
There was 1 case of primary graft failure and 2 cases of secondary graft failure. One case of secondary graft failure was associated with an HHV-6 infection, and the other was due to a lethal adenovirus infection.
Of those patients who engrafted, 97% achieved full donor chimerism, and 3% had mixed chimerism.
Dr Horwitz and his colleagues compared engraftment results in the NiCord recipients to results in a cohort of patients from the CIBMTR registry who underwent UCB transplants from 2010 to 2013. They had similar characteristics as the NiCord patients—age, conditioning regimen, disease status, etc.
In total, there were 148 CIBMTR registry patients, 20% of whom received a single UCB unit.
The median time to neutrophil engraftment was 11.5 days (range, 6-26) with NiCord and 21 days in the CIBMTR matched control cohort (P<0.001). The cumulative incidence of neutrophil engraftment was 94.4% and 89.7%, respectively.
The median time to platelet engraftment was 34 days (range, 25-96) with NiCord and 46 days in the CIBMTR controls (P<0.001). The cumulative incidence of platelet engraftment was 80.6% and 67.1%, respectively.
“There’s a median 10-day reduction in neutrophil recovery [and] 12-day reduction in time to platelet recovery [with NiCord],” Dr Horwitz noted. “There is evidence of robust and durable engraftment with a NiCord unit, with one patient now over 7 years from his first transplant on the pilot trial.”
Relapse, survival, and GVHD
Dr Horwitz reported other outcomes in the NiCord recipients without making comparisons to the CIBMTR matched controls.
The estimated 2-year rate of non-relapse mortality in NiCord recipients was 23.8%, and the estimated 2-year incidence of relapse was 33.2%.
The estimated disease-free survival was 49.1% at 1 year and 43.0% at 2 years. The estimated overall survival was 51.2% at 1 year and 2 years.
At 100 days, the rate of grade 2-4 acute GVHD was 44.0%, and the rate of grade 3-4 acute GVHD was 11.1%.
The estimated 1-year rate of mild to severe chronic GVHD was 40.5%, and the estimated 2-year rate of moderate to severe chronic GVHD was 9.8%.
Dr Horwitz said these “promising results” have led to the launch of a phase 3 registration trial in which researchers are comparing NiCord to standard single or double UCB transplant. The trial is open for accrual.
*Information in the abstract differs from the presentation.
SALT LAKE CITY—The expanded umbilical cord blood (UCB) product NiCord can be used as a stand-alone graft, according to research presented at the 2018 BMT Tandem Meetings.
Researchers found that a single NiCord unit provided “robust” engraftment in a phase 1/2 study of patients with high-risk hematologic malignancies.
NiCord recipients had quicker neutrophil and platelet engraftment than matched control subjects who received standard myeloablative UCB transplant (single or double).
Mitchell Horwitz, MD, of the Duke University Medical Center in Durham, North Carolina, presented these results at the meeting as abstract 49.* The research was sponsored by Gamida Cell, the company developing NiCord.
“[NiCord] is an ex vivo expanded cell product that’s derived from an entire unit of umbilical cord blood,” Dr Horwitz explained. “It’s manufactured starting with a CD133-positive selection, which is the progenitor cell population that’s cultured, and a T-cell containing CD133-negative fraction that is provided also at the time of transplant.”
“The culture system contains nicotinamide—that’s the active ingredient in the culture. And that’s supplemented with cytokines—thrombopoietin, IL-6, FLT-3 ligand, and stem cell factor. The culture is 21 days.”
Previous research showed that double UCB transplant including a NiCord unit could provide benefits over standard double UCB transplant. This led Dr Horwitz and his colleagues to wonder if NiCord could be used as a stand-alone graft.
So the team evaluated the safety and efficacy of NiCord alone in 36 adolescents/adults with high-risk hematologic malignancies.
Patients had acute myelogenous leukemia (n=17), acute lymphoblastic leukemia (n=9), myelodysplastic syndrome (n=7), chronic myelogenous leukemia (n=2), and Hodgkin lymphoma (n=1).
Most patients had intermediate (n=15) or high-risk (n=13) disease. They had a median age of 44 (range, 13-63) and a median weight of 75 kg (range, 41-125).
Treatment
For conditioning, 19 patients received thiotepa, busulfan, and fludarabine. Fifteen patients received total body irradiation and fludarabine with or without cyclophosphamide or thiotepa. And 2 patients received clofarabine, fludarabine, and busulfan.
Most patients had a 4/6 human leukocyte antigen (HLA) match (n=26), 8 had a 5/6 HLA match, and 2 had a 6/6 HLA match.
The median total nucleated cell dose was 2.4 x 107/kg prior to expansion of the UCB unit and 3.7 x 107/kg after expansion. The median CD34+ cell dose was 0.2 x 106/kg and 6.3 x 106/kg, respectively.
“CD34 cells were expanded 33-fold in the 3-week culture system,” Dr Horwitz noted. “That translated to a median CD34 dose of 6.3 x 106/kg, a dose comparable to what would be obtained from an adult donor graft.”
Engraftment
There was 1 case of primary graft failure and 2 cases of secondary graft failure. One case of secondary graft failure was associated with an HHV-6 infection, and the other was due to a lethal adenovirus infection.
Of those patients who engrafted, 97% achieved full donor chimerism, and 3% had mixed chimerism.
Dr Horwitz and his colleagues compared engraftment results in the NiCord recipients to results in a cohort of patients from the CIBMTR registry who underwent UCB transplants from 2010 to 2013. They had similar characteristics as the NiCord patients—age, conditioning regimen, disease status, etc.
In total, there were 148 CIBMTR registry patients, 20% of whom received a single UCB unit.
The median time to neutrophil engraftment was 11.5 days (range, 6-26) with NiCord and 21 days in the CIBMTR matched control cohort (P<0.001). The cumulative incidence of neutrophil engraftment was 94.4% and 89.7%, respectively.
The median time to platelet engraftment was 34 days (range, 25-96) with NiCord and 46 days in the CIBMTR controls (P<0.001). The cumulative incidence of platelet engraftment was 80.6% and 67.1%, respectively.
“There’s a median 10-day reduction in neutrophil recovery [and] 12-day reduction in time to platelet recovery [with NiCord],” Dr Horwitz noted. “There is evidence of robust and durable engraftment with a NiCord unit, with one patient now over 7 years from his first transplant on the pilot trial.”
Relapse, survival, and GVHD
Dr Horwitz reported other outcomes in the NiCord recipients without making comparisons to the CIBMTR matched controls.
The estimated 2-year rate of non-relapse mortality in NiCord recipients was 23.8%, and the estimated 2-year incidence of relapse was 33.2%.
The estimated disease-free survival was 49.1% at 1 year and 43.0% at 2 years. The estimated overall survival was 51.2% at 1 year and 2 years.
At 100 days, the rate of grade 2-4 acute GVHD was 44.0%, and the rate of grade 3-4 acute GVHD was 11.1%.
The estimated 1-year rate of mild to severe chronic GVHD was 40.5%, and the estimated 2-year rate of moderate to severe chronic GVHD was 9.8%.
Dr Horwitz said these “promising results” have led to the launch of a phase 3 registration trial in which researchers are comparing NiCord to standard single or double UCB transplant. The trial is open for accrual.
*Information in the abstract differs from the presentation.
SALT LAKE CITY—The expanded umbilical cord blood (UCB) product NiCord can be used as a stand-alone graft, according to research presented at the 2018 BMT Tandem Meetings.
Researchers found that a single NiCord unit provided “robust” engraftment in a phase 1/2 study of patients with high-risk hematologic malignancies.
NiCord recipients had quicker neutrophil and platelet engraftment than matched control subjects who received standard myeloablative UCB transplant (single or double).
Mitchell Horwitz, MD, of the Duke University Medical Center in Durham, North Carolina, presented these results at the meeting as abstract 49.* The research was sponsored by Gamida Cell, the company developing NiCord.
“[NiCord] is an ex vivo expanded cell product that’s derived from an entire unit of umbilical cord blood,” Dr Horwitz explained. “It’s manufactured starting with a CD133-positive selection, which is the progenitor cell population that’s cultured, and a T-cell containing CD133-negative fraction that is provided also at the time of transplant.”
“The culture system contains nicotinamide—that’s the active ingredient in the culture. And that’s supplemented with cytokines—thrombopoietin, IL-6, FLT-3 ligand, and stem cell factor. The culture is 21 days.”
Previous research showed that double UCB transplant including a NiCord unit could provide benefits over standard double UCB transplant. This led Dr Horwitz and his colleagues to wonder if NiCord could be used as a stand-alone graft.
So the team evaluated the safety and efficacy of NiCord alone in 36 adolescents/adults with high-risk hematologic malignancies.
Patients had acute myelogenous leukemia (n=17), acute lymphoblastic leukemia (n=9), myelodysplastic syndrome (n=7), chronic myelogenous leukemia (n=2), and Hodgkin lymphoma (n=1).
Most patients had intermediate (n=15) or high-risk (n=13) disease. They had a median age of 44 (range, 13-63) and a median weight of 75 kg (range, 41-125).
Treatment
For conditioning, 19 patients received thiotepa, busulfan, and fludarabine. Fifteen patients received total body irradiation and fludarabine with or without cyclophosphamide or thiotepa. And 2 patients received clofarabine, fludarabine, and busulfan.
Most patients had a 4/6 human leukocyte antigen (HLA) match (n=26), 8 had a 5/6 HLA match, and 2 had a 6/6 HLA match.
The median total nucleated cell dose was 2.4 x 107/kg prior to expansion of the UCB unit and 3.7 x 107/kg after expansion. The median CD34+ cell dose was 0.2 x 106/kg and 6.3 x 106/kg, respectively.
“CD34 cells were expanded 33-fold in the 3-week culture system,” Dr Horwitz noted. “That translated to a median CD34 dose of 6.3 x 106/kg, a dose comparable to what would be obtained from an adult donor graft.”
Engraftment
There was 1 case of primary graft failure and 2 cases of secondary graft failure. One case of secondary graft failure was associated with an HHV-6 infection, and the other was due to a lethal adenovirus infection.
Of those patients who engrafted, 97% achieved full donor chimerism, and 3% had mixed chimerism.
Dr Horwitz and his colleagues compared engraftment results in the NiCord recipients to results in a cohort of patients from the CIBMTR registry who underwent UCB transplants from 2010 to 2013. They had similar characteristics as the NiCord patients—age, conditioning regimen, disease status, etc.
In total, there were 148 CIBMTR registry patients, 20% of whom received a single UCB unit.
The median time to neutrophil engraftment was 11.5 days (range, 6-26) with NiCord and 21 days in the CIBMTR matched control cohort (P<0.001). The cumulative incidence of neutrophil engraftment was 94.4% and 89.7%, respectively.
The median time to platelet engraftment was 34 days (range, 25-96) with NiCord and 46 days in the CIBMTR controls (P<0.001). The cumulative incidence of platelet engraftment was 80.6% and 67.1%, respectively.
“There’s a median 10-day reduction in neutrophil recovery [and] 12-day reduction in time to platelet recovery [with NiCord],” Dr Horwitz noted. “There is evidence of robust and durable engraftment with a NiCord unit, with one patient now over 7 years from his first transplant on the pilot trial.”
Relapse, survival, and GVHD
Dr Horwitz reported other outcomes in the NiCord recipients without making comparisons to the CIBMTR matched controls.
The estimated 2-year rate of non-relapse mortality in NiCord recipients was 23.8%, and the estimated 2-year incidence of relapse was 33.2%.
The estimated disease-free survival was 49.1% at 1 year and 43.0% at 2 years. The estimated overall survival was 51.2% at 1 year and 2 years.
At 100 days, the rate of grade 2-4 acute GVHD was 44.0%, and the rate of grade 3-4 acute GVHD was 11.1%.
The estimated 1-year rate of mild to severe chronic GVHD was 40.5%, and the estimated 2-year rate of moderate to severe chronic GVHD was 9.8%.
Dr Horwitz said these “promising results” have led to the launch of a phase 3 registration trial in which researchers are comparing NiCord to standard single or double UCB transplant. The trial is open for accrual.
*Information in the abstract differs from the presentation.
Survival differences among AYAs with blood cancers
A new report has revealed differences in survival among adolescents and young adults (AYAs) with hematologic malignancies.
The report includes information on AYAs—ages 15 to 39—living in Los Angeles County who were diagnosed with common cancers between 1988 and 2014.
The data showed differences in 5-year survival rates according to sex, race, age, and socioeconomic status (SES).
For example, lymphoma survival rates were lower for males, African Americans (AAs), older AYAs, and patients with low socioeconomic status (SES).
For AYAs with leukemias, there was no survival difference according to sex, but AAs had worse survival than patients of other races. And the impact of age and SES varied according to leukemia type.
“Cancer survival data are poorly understood for 15- to 39-year-olds,” noted Amie Hwang, PhD, of the University of Southern California Keck School of Medicine in Los Angeles.
That is why she and her colleagues created the report, “Cancer in Los Angeles County: Survival Among Adolescents and Young Adults 1988-2014.”
According to the authors, this is the first report to break down cancer survival rates for AYAs into segments on race/ethnicity, sex, age group, SES, and cancer stage.
Survival data for patients with hematologic malignancies were as follows.
Acute lymphoblastic leukemia
There were 1137 cases of acute lymphoblastic leukemia in the AYA population in Los Angeles County during the period studied. This included 752 males and 385 females.
Five-year survival was similar between males (43%) and females (41%).
Younger AYAs had better survival than older AYAs (48% for ages 15-24, 35% for ages 25-34, and 32% for ages 35-39).
Survival was highest among non-Latino whites (NLWs, 56%), followed by Asian/Pacific Islanders (APIs, 52%), patients of other/unknown races (51%), Latino whites (LWs, 38%), and AAs (29%).
Survival declined with SES (55% for high, 42% for middle, and 36% for low SES).
Acute myeloid leukemia
There were 1195 cases of acute myeloid leukemia—641 males and 554 females.
Five-year survival was similar for males (40%) and females (43%) as well as for the different age groups (45% for ages 15-24 vs 40% for the older age groups).
Survival was highest among NLWs (44%), followed by LWs (43%), APIs (40%), other/unknown (33%), and AAs (25%).
Survival declined somewhat with SES (49% for high, 39% for middle, and 41% for low SES).
Chronic myeloid leukemia
There were 655 cases of chronic myeloid leukemia—408 males and 247 females.
Five-year survival was similar for males (70%) and females (71%), but it was slightly higher for older AYAs (69% for ages 15-24, 68% for ages 25-34, and 76% for ages 35-39).
Survival was highest among patients in the “other/unknown” race category (76%), followed by LWs (73%), NLWs/APIs (both 72%), and AAs (57%).
Survival declined somewhat with SES (76% for high, 67% for middle, and 68% for low SES).
Hodgkin lymphoma
There were 2993 AYAs diagnosed with Hodgkin lymphoma—1553 males and 1440 females.
The 5-year survival rate was higher in females (93%) than males (86%) and in younger AYAs (93% for ages 15-24, 89% for ages 25-34, and 85% for ages 35-39).
Survival was highest among patients in the “other/unknown” race category (96%), followed by APIs/NLWs (both 91%), LWs (88%), and AAs (83%).
Survival declined with SES (95% for high, 89% for middle, and 83% for low SES).
And survival was lower for patients with advanced-stage disease (93% localized, 94% regional, and 83% distant).
Non-Hodkgin lymphoma
There were 4485 AYAs diagnosed with non-Hodgkin lymphoma during the study period—3064 males and 1421 females.
The 5-year survival rate was higher in females (75%) than males (46%) and in younger AYAs (69% for ages 15-24, 51% for ages 25-34, and 52% for ages 35-39).
Survival was highest among patients in the “other/unknown” race category (88%), followed by APIs (68%), LWs/NLWs (both 53%), and AAs (50%).
Survival declined with SES (68% for high, 54% for middle, and 45% for low SES).
And survival was lower for patients with advanced-stage disease (61% localized, 66% regional, and 46% distant).
“Adolescents and young adults go to the doctor less often because they have this superhero mentality, like they’re invincible,” said author Dennis Deapen, DrPH, of the University of Southern California Keck School of Medicine.
“Once they do go to a health professional, their cancer diagnosis can be delayed because cancer isn’t the first concern doctors have for this age group. It comes as no surprise that patients diagnosed with late-stage cancer have reduced survival rates.” ![]()
A new report has revealed differences in survival among adolescents and young adults (AYAs) with hematologic malignancies.
The report includes information on AYAs—ages 15 to 39—living in Los Angeles County who were diagnosed with common cancers between 1988 and 2014.
The data showed differences in 5-year survival rates according to sex, race, age, and socioeconomic status (SES).
For example, lymphoma survival rates were lower for males, African Americans (AAs), older AYAs, and patients with low socioeconomic status (SES).
For AYAs with leukemias, there was no survival difference according to sex, but AAs had worse survival than patients of other races. And the impact of age and SES varied according to leukemia type.
“Cancer survival data are poorly understood for 15- to 39-year-olds,” noted Amie Hwang, PhD, of the University of Southern California Keck School of Medicine in Los Angeles.
That is why she and her colleagues created the report, “Cancer in Los Angeles County: Survival Among Adolescents and Young Adults 1988-2014.”
According to the authors, this is the first report to break down cancer survival rates for AYAs into segments on race/ethnicity, sex, age group, SES, and cancer stage.
Survival data for patients with hematologic malignancies were as follows.
Acute lymphoblastic leukemia
There were 1137 cases of acute lymphoblastic leukemia in the AYA population in Los Angeles County during the period studied. This included 752 males and 385 females.
Five-year survival was similar between males (43%) and females (41%).
Younger AYAs had better survival than older AYAs (48% for ages 15-24, 35% for ages 25-34, and 32% for ages 35-39).
Survival was highest among non-Latino whites (NLWs, 56%), followed by Asian/Pacific Islanders (APIs, 52%), patients of other/unknown races (51%), Latino whites (LWs, 38%), and AAs (29%).
Survival declined with SES (55% for high, 42% for middle, and 36% for low SES).
Acute myeloid leukemia
There were 1195 cases of acute myeloid leukemia—641 males and 554 females.
Five-year survival was similar for males (40%) and females (43%) as well as for the different age groups (45% for ages 15-24 vs 40% for the older age groups).
Survival was highest among NLWs (44%), followed by LWs (43%), APIs (40%), other/unknown (33%), and AAs (25%).
Survival declined somewhat with SES (49% for high, 39% for middle, and 41% for low SES).
Chronic myeloid leukemia
There were 655 cases of chronic myeloid leukemia—408 males and 247 females.
Five-year survival was similar for males (70%) and females (71%), but it was slightly higher for older AYAs (69% for ages 15-24, 68% for ages 25-34, and 76% for ages 35-39).
Survival was highest among patients in the “other/unknown” race category (76%), followed by LWs (73%), NLWs/APIs (both 72%), and AAs (57%).
Survival declined somewhat with SES (76% for high, 67% for middle, and 68% for low SES).
Hodgkin lymphoma
There were 2993 AYAs diagnosed with Hodgkin lymphoma—1553 males and 1440 females.
The 5-year survival rate was higher in females (93%) than males (86%) and in younger AYAs (93% for ages 15-24, 89% for ages 25-34, and 85% for ages 35-39).
Survival was highest among patients in the “other/unknown” race category (96%), followed by APIs/NLWs (both 91%), LWs (88%), and AAs (83%).
Survival declined with SES (95% for high, 89% for middle, and 83% for low SES).
And survival was lower for patients with advanced-stage disease (93% localized, 94% regional, and 83% distant).
Non-Hodkgin lymphoma
There were 4485 AYAs diagnosed with non-Hodgkin lymphoma during the study period—3064 males and 1421 females.
The 5-year survival rate was higher in females (75%) than males (46%) and in younger AYAs (69% for ages 15-24, 51% for ages 25-34, and 52% for ages 35-39).
Survival was highest among patients in the “other/unknown” race category (88%), followed by APIs (68%), LWs/NLWs (both 53%), and AAs (50%).
Survival declined with SES (68% for high, 54% for middle, and 45% for low SES).
And survival was lower for patients with advanced-stage disease (61% localized, 66% regional, and 46% distant).
“Adolescents and young adults go to the doctor less often because they have this superhero mentality, like they’re invincible,” said author Dennis Deapen, DrPH, of the University of Southern California Keck School of Medicine.
“Once they do go to a health professional, their cancer diagnosis can be delayed because cancer isn’t the first concern doctors have for this age group. It comes as no surprise that patients diagnosed with late-stage cancer have reduced survival rates.” ![]()
A new report has revealed differences in survival among adolescents and young adults (AYAs) with hematologic malignancies.
The report includes information on AYAs—ages 15 to 39—living in Los Angeles County who were diagnosed with common cancers between 1988 and 2014.
The data showed differences in 5-year survival rates according to sex, race, age, and socioeconomic status (SES).
For example, lymphoma survival rates were lower for males, African Americans (AAs), older AYAs, and patients with low socioeconomic status (SES).
For AYAs with leukemias, there was no survival difference according to sex, but AAs had worse survival than patients of other races. And the impact of age and SES varied according to leukemia type.
“Cancer survival data are poorly understood for 15- to 39-year-olds,” noted Amie Hwang, PhD, of the University of Southern California Keck School of Medicine in Los Angeles.
That is why she and her colleagues created the report, “Cancer in Los Angeles County: Survival Among Adolescents and Young Adults 1988-2014.”
According to the authors, this is the first report to break down cancer survival rates for AYAs into segments on race/ethnicity, sex, age group, SES, and cancer stage.
Survival data for patients with hematologic malignancies were as follows.
Acute lymphoblastic leukemia
There were 1137 cases of acute lymphoblastic leukemia in the AYA population in Los Angeles County during the period studied. This included 752 males and 385 females.
Five-year survival was similar between males (43%) and females (41%).
Younger AYAs had better survival than older AYAs (48% for ages 15-24, 35% for ages 25-34, and 32% for ages 35-39).
Survival was highest among non-Latino whites (NLWs, 56%), followed by Asian/Pacific Islanders (APIs, 52%), patients of other/unknown races (51%), Latino whites (LWs, 38%), and AAs (29%).
Survival declined with SES (55% for high, 42% for middle, and 36% for low SES).
Acute myeloid leukemia
There were 1195 cases of acute myeloid leukemia—641 males and 554 females.
Five-year survival was similar for males (40%) and females (43%) as well as for the different age groups (45% for ages 15-24 vs 40% for the older age groups).
Survival was highest among NLWs (44%), followed by LWs (43%), APIs (40%), other/unknown (33%), and AAs (25%).
Survival declined somewhat with SES (49% for high, 39% for middle, and 41% for low SES).
Chronic myeloid leukemia
There were 655 cases of chronic myeloid leukemia—408 males and 247 females.
Five-year survival was similar for males (70%) and females (71%), but it was slightly higher for older AYAs (69% for ages 15-24, 68% for ages 25-34, and 76% for ages 35-39).
Survival was highest among patients in the “other/unknown” race category (76%), followed by LWs (73%), NLWs/APIs (both 72%), and AAs (57%).
Survival declined somewhat with SES (76% for high, 67% for middle, and 68% for low SES).
Hodgkin lymphoma
There were 2993 AYAs diagnosed with Hodgkin lymphoma—1553 males and 1440 females.
The 5-year survival rate was higher in females (93%) than males (86%) and in younger AYAs (93% for ages 15-24, 89% for ages 25-34, and 85% for ages 35-39).
Survival was highest among patients in the “other/unknown” race category (96%), followed by APIs/NLWs (both 91%), LWs (88%), and AAs (83%).
Survival declined with SES (95% for high, 89% for middle, and 83% for low SES).
And survival was lower for patients with advanced-stage disease (93% localized, 94% regional, and 83% distant).
Non-Hodkgin lymphoma
There were 4485 AYAs diagnosed with non-Hodgkin lymphoma during the study period—3064 males and 1421 females.
The 5-year survival rate was higher in females (75%) than males (46%) and in younger AYAs (69% for ages 15-24, 51% for ages 25-34, and 52% for ages 35-39).
Survival was highest among patients in the “other/unknown” race category (88%), followed by APIs (68%), LWs/NLWs (both 53%), and AAs (50%).
Survival declined with SES (68% for high, 54% for middle, and 45% for low SES).
And survival was lower for patients with advanced-stage disease (61% localized, 66% regional, and 46% distant).
“Adolescents and young adults go to the doctor less often because they have this superhero mentality, like they’re invincible,” said author Dennis Deapen, DrPH, of the University of Southern California Keck School of Medicine.
“Once they do go to a health professional, their cancer diagnosis can be delayed because cancer isn’t the first concern doctors have for this age group. It comes as no surprise that patients diagnosed with late-stage cancer have reduced survival rates.” ![]()
Marine animals aid development of cytotoxicity assay
Researchers have looked to deep-sea creatures with the goal of creating a better cytotoxicity assay.
The team harnessed the power of enzymes responsible for marine animal bioluminescence to create the “Matador assay,” which can be used to determine whether cellular and immune-therapeutic agents are actually killing target cells.
The researchers said the Matador assay is quick and simple as well as “highly sensitive,” with the ability to detect cytotoxicity induced by several types of therapies.
Preet M. Chaudhary, MD, PhD, of the University of Southern California Keck School of Medicine in Los Angeles, and his colleagues described the assay in Scientific Reports.
“One of the most promising areas in cancer research is immunotherapy. . .,” Dr Chaudhary said. “It is also one of the most difficult because the methods for testing immunotherapies are not ideal.”
“Radioactive chromium release assay is the gold standard for testing whether an immunotherapy kills cancer cells. This method is expensive, complicated, and requires special disposal practices. Other available methods also suffer from limitations and don’t allow scientists to rapidly screen immunotherapeutic agents to find the best candidates.”
Dr Chaudhary and his colleagues set out to develop a simple, precise, and inexpensive cytotoxicity assay based on marine animal luciferases, the enzymes responsible for bioluminescence.
The team used a group of small crustaceans and deep-sea shrimp, which were selected for their bright bioluminescence. Their luciferases became the basis of the Matador assay.
Engineered to get trapped inside cells, the luciferases leak out of cells when they die, causing a visible glow. The level of luminescence can then be measured with a luminometer.
To test the Matador assay’s effectiveness at measuring cell death, the researchers used several types of cancer cells, including chronic myelogenous leukemia, acute myelogenous leukemia, Burkitt lymphoma, and solid tumor cells.
The team treated these cells with a variety of therapies, including chimeric antigen receptor (CAR) T cells, bispecific T-cell engagers, monoclonal antibodies, and natural killer cells.
Results showed the Matador assay could detect the death of a single cell, a level of sensitivity superior to that of existing cytotoxicity assays.
The researchers also pointed out that the Matador assay is fast, inexpensive, and can be performed in a 384-well plate format, saving time and reagents.
“In our hands, the Matador assay can detect cell death in as little as 30 minutes, which can ultimately translate to more expedient treatments for patients getting cellular immunotherapies such as CAR T cells,” Dr Chaudhary said.
In fact, Dr Chaudhary’s lab has developed more than 75 cancer cell lines expressing the marine luciferases and used them with the Matador assay to develop next-generation CAR T cells.
Dr Chaudhary believes the Matador assay has many potential applications in biomedical research and cellular therapy manufacturing.
“It could potentially play a role in screening other types of anticancer agents or even measuring environmental toxins,” he said. ![]()
Researchers have looked to deep-sea creatures with the goal of creating a better cytotoxicity assay.
The team harnessed the power of enzymes responsible for marine animal bioluminescence to create the “Matador assay,” which can be used to determine whether cellular and immune-therapeutic agents are actually killing target cells.
The researchers said the Matador assay is quick and simple as well as “highly sensitive,” with the ability to detect cytotoxicity induced by several types of therapies.
Preet M. Chaudhary, MD, PhD, of the University of Southern California Keck School of Medicine in Los Angeles, and his colleagues described the assay in Scientific Reports.
“One of the most promising areas in cancer research is immunotherapy. . .,” Dr Chaudhary said. “It is also one of the most difficult because the methods for testing immunotherapies are not ideal.”
“Radioactive chromium release assay is the gold standard for testing whether an immunotherapy kills cancer cells. This method is expensive, complicated, and requires special disposal practices. Other available methods also suffer from limitations and don’t allow scientists to rapidly screen immunotherapeutic agents to find the best candidates.”
Dr Chaudhary and his colleagues set out to develop a simple, precise, and inexpensive cytotoxicity assay based on marine animal luciferases, the enzymes responsible for bioluminescence.
The team used a group of small crustaceans and deep-sea shrimp, which were selected for their bright bioluminescence. Their luciferases became the basis of the Matador assay.
Engineered to get trapped inside cells, the luciferases leak out of cells when they die, causing a visible glow. The level of luminescence can then be measured with a luminometer.
To test the Matador assay’s effectiveness at measuring cell death, the researchers used several types of cancer cells, including chronic myelogenous leukemia, acute myelogenous leukemia, Burkitt lymphoma, and solid tumor cells.
The team treated these cells with a variety of therapies, including chimeric antigen receptor (CAR) T cells, bispecific T-cell engagers, monoclonal antibodies, and natural killer cells.
Results showed the Matador assay could detect the death of a single cell, a level of sensitivity superior to that of existing cytotoxicity assays.
The researchers also pointed out that the Matador assay is fast, inexpensive, and can be performed in a 384-well plate format, saving time and reagents.
“In our hands, the Matador assay can detect cell death in as little as 30 minutes, which can ultimately translate to more expedient treatments for patients getting cellular immunotherapies such as CAR T cells,” Dr Chaudhary said.
In fact, Dr Chaudhary’s lab has developed more than 75 cancer cell lines expressing the marine luciferases and used them with the Matador assay to develop next-generation CAR T cells.
Dr Chaudhary believes the Matador assay has many potential applications in biomedical research and cellular therapy manufacturing.
“It could potentially play a role in screening other types of anticancer agents or even measuring environmental toxins,” he said. ![]()
Researchers have looked to deep-sea creatures with the goal of creating a better cytotoxicity assay.
The team harnessed the power of enzymes responsible for marine animal bioluminescence to create the “Matador assay,” which can be used to determine whether cellular and immune-therapeutic agents are actually killing target cells.
The researchers said the Matador assay is quick and simple as well as “highly sensitive,” with the ability to detect cytotoxicity induced by several types of therapies.
Preet M. Chaudhary, MD, PhD, of the University of Southern California Keck School of Medicine in Los Angeles, and his colleagues described the assay in Scientific Reports.
“One of the most promising areas in cancer research is immunotherapy. . .,” Dr Chaudhary said. “It is also one of the most difficult because the methods for testing immunotherapies are not ideal.”
“Radioactive chromium release assay is the gold standard for testing whether an immunotherapy kills cancer cells. This method is expensive, complicated, and requires special disposal practices. Other available methods also suffer from limitations and don’t allow scientists to rapidly screen immunotherapeutic agents to find the best candidates.”
Dr Chaudhary and his colleagues set out to develop a simple, precise, and inexpensive cytotoxicity assay based on marine animal luciferases, the enzymes responsible for bioluminescence.
The team used a group of small crustaceans and deep-sea shrimp, which were selected for their bright bioluminescence. Their luciferases became the basis of the Matador assay.
Engineered to get trapped inside cells, the luciferases leak out of cells when they die, causing a visible glow. The level of luminescence can then be measured with a luminometer.
To test the Matador assay’s effectiveness at measuring cell death, the researchers used several types of cancer cells, including chronic myelogenous leukemia, acute myelogenous leukemia, Burkitt lymphoma, and solid tumor cells.
The team treated these cells with a variety of therapies, including chimeric antigen receptor (CAR) T cells, bispecific T-cell engagers, monoclonal antibodies, and natural killer cells.
Results showed the Matador assay could detect the death of a single cell, a level of sensitivity superior to that of existing cytotoxicity assays.
The researchers also pointed out that the Matador assay is fast, inexpensive, and can be performed in a 384-well plate format, saving time and reagents.
“In our hands, the Matador assay can detect cell death in as little as 30 minutes, which can ultimately translate to more expedient treatments for patients getting cellular immunotherapies such as CAR T cells,” Dr Chaudhary said.
In fact, Dr Chaudhary’s lab has developed more than 75 cancer cell lines expressing the marine luciferases and used them with the Matador assay to develop next-generation CAR T cells.
Dr Chaudhary believes the Matador assay has many potential applications in biomedical research and cellular therapy manufacturing.
“It could potentially play a role in screening other types of anticancer agents or even measuring environmental toxins,” he said. ![]()
FDA expands indication for bosutinib in newly diagnosed CML
Bosutinib is now approved for the treatment of adults with newly diagnosed chronic phase Philadelphia chromosome–positive (Ph+) chronic myelogenous leukemia (CML).
The Food and Drug Administration granted accelerated approval for bosutinib (Bosulif), which is marketed by Pfizer. The approval is based on data from the randomized, multicenter phase 3 BFORE trial of 487 patients with Ph+ newly diagnosed chronic phase CML who received either bosutinib or imatinib 400 mg once daily. Major molecular response at 12 months was 47.2% (95% confidence interval, 40.9-53.4) in the bosutinib arm and 36.9% (95% CI, 30.8-43.0) in the imatinib arm (two-sided P = .0200).
Bosutinib, a kinase inhibitor, was first approved in September 2012 for the treatment of adult patients with chronic, accelerated, or blast phase Ph+ CML with resistance or intolerance to prior therapy.
The recommended dose of bosutinib for newly diagnosed chronic phase Ph+ CML is 400 mg orally once daily with food.
The most common adverse reactions to the drug in newly diagnosed CML patients are diarrhea, nausea, thrombocytopenia, rash, increased alanine aminotransferase, abdominal pain, and increased aspartate aminotransferase.
Bosutinib is now approved for the treatment of adults with newly diagnosed chronic phase Philadelphia chromosome–positive (Ph+) chronic myelogenous leukemia (CML).
The Food and Drug Administration granted accelerated approval for bosutinib (Bosulif), which is marketed by Pfizer. The approval is based on data from the randomized, multicenter phase 3 BFORE trial of 487 patients with Ph+ newly diagnosed chronic phase CML who received either bosutinib or imatinib 400 mg once daily. Major molecular response at 12 months was 47.2% (95% confidence interval, 40.9-53.4) in the bosutinib arm and 36.9% (95% CI, 30.8-43.0) in the imatinib arm (two-sided P = .0200).
Bosutinib, a kinase inhibitor, was first approved in September 2012 for the treatment of adult patients with chronic, accelerated, or blast phase Ph+ CML with resistance or intolerance to prior therapy.
The recommended dose of bosutinib for newly diagnosed chronic phase Ph+ CML is 400 mg orally once daily with food.
The most common adverse reactions to the drug in newly diagnosed CML patients are diarrhea, nausea, thrombocytopenia, rash, increased alanine aminotransferase, abdominal pain, and increased aspartate aminotransferase.
Bosutinib is now approved for the treatment of adults with newly diagnosed chronic phase Philadelphia chromosome–positive (Ph+) chronic myelogenous leukemia (CML).
The Food and Drug Administration granted accelerated approval for bosutinib (Bosulif), which is marketed by Pfizer. The approval is based on data from the randomized, multicenter phase 3 BFORE trial of 487 patients with Ph+ newly diagnosed chronic phase CML who received either bosutinib or imatinib 400 mg once daily. Major molecular response at 12 months was 47.2% (95% confidence interval, 40.9-53.4) in the bosutinib arm and 36.9% (95% CI, 30.8-43.0) in the imatinib arm (two-sided P = .0200).
Bosutinib, a kinase inhibitor, was first approved in September 2012 for the treatment of adult patients with chronic, accelerated, or blast phase Ph+ CML with resistance or intolerance to prior therapy.
The recommended dose of bosutinib for newly diagnosed chronic phase Ph+ CML is 400 mg orally once daily with food.
The most common adverse reactions to the drug in newly diagnosed CML patients are diarrhea, nausea, thrombocytopenia, rash, increased alanine aminotransferase, abdominal pain, and increased aspartate aminotransferase.
FDA updates nilotinib product label outlining criteria for discontinuation
for patients who meet certain criteria. Nilotinib, a kinase inhibitor that blocks the BCR-ABL protein that promotes abnormal cell growth, was originally approved in 2007 and was indicated for use in patients with Philadelphia chromosome positive (Ph+) chronic myeloid leukemia (CML). In accordance with the new label update, patients who have early phase CML, have been using nilotinib for 3 years or more, and whose leukemia has responded to treatment according to a test that has received FDA marketing authorization, may be eligible to discontinue use of nilotinib.
The information that led to the FDA approved label update was based on two single-arm trials of patients with chronic phase Ph+ CML. The trial measured the length of time patients were able to discontinue use of nilotinib without leukemia returning, and who had entered treatment-free remission (TFR). In the first trial, among 190 newly diagnosed CML patients who discontinued taking nilotinib after using the drug for 3 or more years, 51.6% were still in TFR after about 1 year (48 weeks) and 48.9% were still in TFR after nearly 2 years (96 weeks). Similar results were seen in the second trial, among 126 patients, with 57.9% in TFR after about a year (48 weeks) and 53.2% in TFR after approximately 2 years (96 weeks).
An important element of these trials was regular monitoring of specific RNA information that specifies the level of BCR-ABL protein in the blood using a diagnostic test that has received FDA marketing authorization. Monitoring with a test that accurately detects the reductions of RNA information in the blood with accuracy and precision is critical in discontinuing the use of nilotinib. This monitoring will allow physicians to detect the first signs of relapse.
“Patients diagnosed with CML generally face a lifetime of treatment to keep their leukemia from growing or recurring,” said Richard Pazdur, MD, acting director of the Office of Hematology and Oncology Products in the FDA’s Center for Drug Evaluation and Research. “Today’s approval shows that some patients may be able to stop treatment with Tasigna altogether if they are showing a strong response to therapy. While we welcome this progress in patient care, it’s important to note that any discontinuation of treatment still means patients must be regularly monitored for disease recurrence,” Dr. Pazdur said in the FDA statement.
Common side effects after discontinuing use of nilotinib include body aches and pain in the bones and extremities. Severe side effects of taking nilotinib can include myelosuppression, blockages in the heart and arteries, and inflammation of the pancreas. Severe liver damage can also occur.
Severe side effects typically associated with nilotinib administration occurred less frequently in patients who discontinued the drug. However, the long-term outcomes of patients discontinuing versus continuing treatment are unknown at this time, the FDA noted.
for patients who meet certain criteria. Nilotinib, a kinase inhibitor that blocks the BCR-ABL protein that promotes abnormal cell growth, was originally approved in 2007 and was indicated for use in patients with Philadelphia chromosome positive (Ph+) chronic myeloid leukemia (CML). In accordance with the new label update, patients who have early phase CML, have been using nilotinib for 3 years or more, and whose leukemia has responded to treatment according to a test that has received FDA marketing authorization, may be eligible to discontinue use of nilotinib.
The information that led to the FDA approved label update was based on two single-arm trials of patients with chronic phase Ph+ CML. The trial measured the length of time patients were able to discontinue use of nilotinib without leukemia returning, and who had entered treatment-free remission (TFR). In the first trial, among 190 newly diagnosed CML patients who discontinued taking nilotinib after using the drug for 3 or more years, 51.6% were still in TFR after about 1 year (48 weeks) and 48.9% were still in TFR after nearly 2 years (96 weeks). Similar results were seen in the second trial, among 126 patients, with 57.9% in TFR after about a year (48 weeks) and 53.2% in TFR after approximately 2 years (96 weeks).
An important element of these trials was regular monitoring of specific RNA information that specifies the level of BCR-ABL protein in the blood using a diagnostic test that has received FDA marketing authorization. Monitoring with a test that accurately detects the reductions of RNA information in the blood with accuracy and precision is critical in discontinuing the use of nilotinib. This monitoring will allow physicians to detect the first signs of relapse.
“Patients diagnosed with CML generally face a lifetime of treatment to keep their leukemia from growing or recurring,” said Richard Pazdur, MD, acting director of the Office of Hematology and Oncology Products in the FDA’s Center for Drug Evaluation and Research. “Today’s approval shows that some patients may be able to stop treatment with Tasigna altogether if they are showing a strong response to therapy. While we welcome this progress in patient care, it’s important to note that any discontinuation of treatment still means patients must be regularly monitored for disease recurrence,” Dr. Pazdur said in the FDA statement.
Common side effects after discontinuing use of nilotinib include body aches and pain in the bones and extremities. Severe side effects of taking nilotinib can include myelosuppression, blockages in the heart and arteries, and inflammation of the pancreas. Severe liver damage can also occur.
Severe side effects typically associated with nilotinib administration occurred less frequently in patients who discontinued the drug. However, the long-term outcomes of patients discontinuing versus continuing treatment are unknown at this time, the FDA noted.
for patients who meet certain criteria. Nilotinib, a kinase inhibitor that blocks the BCR-ABL protein that promotes abnormal cell growth, was originally approved in 2007 and was indicated for use in patients with Philadelphia chromosome positive (Ph+) chronic myeloid leukemia (CML). In accordance with the new label update, patients who have early phase CML, have been using nilotinib for 3 years or more, and whose leukemia has responded to treatment according to a test that has received FDA marketing authorization, may be eligible to discontinue use of nilotinib.
The information that led to the FDA approved label update was based on two single-arm trials of patients with chronic phase Ph+ CML. The trial measured the length of time patients were able to discontinue use of nilotinib without leukemia returning, and who had entered treatment-free remission (TFR). In the first trial, among 190 newly diagnosed CML patients who discontinued taking nilotinib after using the drug for 3 or more years, 51.6% were still in TFR after about 1 year (48 weeks) and 48.9% were still in TFR after nearly 2 years (96 weeks). Similar results were seen in the second trial, among 126 patients, with 57.9% in TFR after about a year (48 weeks) and 53.2% in TFR after approximately 2 years (96 weeks).
An important element of these trials was regular monitoring of specific RNA information that specifies the level of BCR-ABL protein in the blood using a diagnostic test that has received FDA marketing authorization. Monitoring with a test that accurately detects the reductions of RNA information in the blood with accuracy and precision is critical in discontinuing the use of nilotinib. This monitoring will allow physicians to detect the first signs of relapse.
“Patients diagnosed with CML generally face a lifetime of treatment to keep their leukemia from growing or recurring,” said Richard Pazdur, MD, acting director of the Office of Hematology and Oncology Products in the FDA’s Center for Drug Evaluation and Research. “Today’s approval shows that some patients may be able to stop treatment with Tasigna altogether if they are showing a strong response to therapy. While we welcome this progress in patient care, it’s important to note that any discontinuation of treatment still means patients must be regularly monitored for disease recurrence,” Dr. Pazdur said in the FDA statement.
Common side effects after discontinuing use of nilotinib include body aches and pain in the bones and extremities. Severe side effects of taking nilotinib can include myelosuppression, blockages in the heart and arteries, and inflammation of the pancreas. Severe liver damage can also occur.
Severe side effects typically associated with nilotinib administration occurred less frequently in patients who discontinued the drug. However, the long-term outcomes of patients discontinuing versus continuing treatment are unknown at this time, the FDA noted.
Nilotinib label updated with info on discontinuation
The US Food and Drug Administration (FDA) has approved an update to the product label for nilotinib (Tasigna) that includes information about how to discontinue the drug in certain patients.
Nilotinib, which was first approved by the FDA in 2007, is indicated for the treatment of patients with Philadelphia chromosome positive (Ph+) chronic myeloid leukemia (CML).
The updated prescribing information for the drug now outlines which of these patients may be eligible to stop receiving nilotinib.
Patients with chronic phase, Ph+ CML who have been taking nilotinib for 3 years or more (as first-line treatment or after failure with imatinib) and have achieved a sustained deep molecular response may be eligible to stop treatment.
Patients must have maintained a molecular response of at least MR4.0 for 1 year prior to discontinuation and achieved an MR4.5 at their last assessment.
Patients must have typical BCR-ABL transcripts (e13a2/b2a2 or e14a2/b3a2), no history of accelerated phase or blast crisis, and no past attempts at treatment discontinuation that resulted in relapse.
After discontinuation, patients must be monitored for possible loss of major molecular response (MMR). BCR-ABL transcript levels must be assessed and a complete blood count with differential performed monthly for 1 year, then every 6 weeks for the second year, and every 12 weeks thereafter.
BCR-ABL transcript levels should be measured using the MolecularMD MRDxTM BCR-ABL test, an FDA-authorized companion diagnostic validated to measure down to MR4.5.
If patients lose MMR, they must restart nilotinib within 4 weeks and receive the dose level they were receiving prior to discontinuation.
BCR-ABL transcript levels should be monitored every 2 weeks until they are lower than MMR for 4 consecutive measurements. Patients can then return to the original monitoring schedule.
The update of nilotinib’s label to include this information was based on results from 2 single-arm trials—ENESTfreedom and ENESTop.
ENESTfreedom
This phase 2 trial included 215 patients with Ph+, chronic phase CML. Researchers evaluated stopping treatment in 190 patients who had achieved a response of MR4.5 with nilotinib as first-line treatment. The patients had sustained deep molecular response for 1 year prior to treatment discontinuation.
Ninety-six weeks after stopping treatment, 48.9% of patients were still in MMR, also known as treatment-free remission (TFR).
Of the 88 patients who restarted nilotinib due to loss of MMR by the cut-off date, 98.9% were able to regain MMR (n=87). One patient discontinued the study at 7.1 weeks without regaining MMR after reinitiating treatment with nilotinib. Eighty-one patients (92.0%) regained MR4.5 by the cut-off date.
Common adverse events (AEs) in patients who discontinued nilotinib included musculoskeletal symptoms such as body aches, bone pain, and pain in extremities. However, these AEs decreased over time.
The incidence of musculoskeletal pain-related AEs decreased from 34.0% to 9.0% during the first and second 48 weeks of the TFR phase, respectively. In comparison, the incidence of such AEs was 17.0% during the treatment consolidation phase.
ENESTop
This phase 2 trial included 163 patients with Ph+, chronic phase CML. Researchers evaluated stopping treatment in 126 patients who had previously received imatinib, then switched to nilotinib and sustained a molecular response for 1 year prior to stopping nilotinib.
At 96 weeks, 53.2% of patients were still in TFR.
Fifty-six patients with confirmed loss of MR4.0 or loss of MMR restarted nilotinib by the cut-off date. Of these patients, 92.9% (n=52) regained both MR4.0 and MR4.5.
As in ENESTfreedom, patients who discontinued nilotinib had musculoskeletal symptoms that decreased over time.
The incidence of musculoskeletal pain-related AEs decreased from 47.9% to 15.1% during the first and second 48 weeks of the TFR phase, respectively. In comparison, the incidence of such AEs was 13.7% during the treatment consolidation phase.
Additional data from ENESTop and ENESTfreedom, as well as the recommendations for stopping nilotinib, are included in the updated prescribing information, which is available at https://www.us.tasigna.com/. ![]()
The US Food and Drug Administration (FDA) has approved an update to the product label for nilotinib (Tasigna) that includes information about how to discontinue the drug in certain patients.
Nilotinib, which was first approved by the FDA in 2007, is indicated for the treatment of patients with Philadelphia chromosome positive (Ph+) chronic myeloid leukemia (CML).
The updated prescribing information for the drug now outlines which of these patients may be eligible to stop receiving nilotinib.
Patients with chronic phase, Ph+ CML who have been taking nilotinib for 3 years or more (as first-line treatment or after failure with imatinib) and have achieved a sustained deep molecular response may be eligible to stop treatment.
Patients must have maintained a molecular response of at least MR4.0 for 1 year prior to discontinuation and achieved an MR4.5 at their last assessment.
Patients must have typical BCR-ABL transcripts (e13a2/b2a2 or e14a2/b3a2), no history of accelerated phase or blast crisis, and no past attempts at treatment discontinuation that resulted in relapse.
After discontinuation, patients must be monitored for possible loss of major molecular response (MMR). BCR-ABL transcript levels must be assessed and a complete blood count with differential performed monthly for 1 year, then every 6 weeks for the second year, and every 12 weeks thereafter.
BCR-ABL transcript levels should be measured using the MolecularMD MRDxTM BCR-ABL test, an FDA-authorized companion diagnostic validated to measure down to MR4.5.
If patients lose MMR, they must restart nilotinib within 4 weeks and receive the dose level they were receiving prior to discontinuation.
BCR-ABL transcript levels should be monitored every 2 weeks until they are lower than MMR for 4 consecutive measurements. Patients can then return to the original monitoring schedule.
The update of nilotinib’s label to include this information was based on results from 2 single-arm trials—ENESTfreedom and ENESTop.
ENESTfreedom
This phase 2 trial included 215 patients with Ph+, chronic phase CML. Researchers evaluated stopping treatment in 190 patients who had achieved a response of MR4.5 with nilotinib as first-line treatment. The patients had sustained deep molecular response for 1 year prior to treatment discontinuation.
Ninety-six weeks after stopping treatment, 48.9% of patients were still in MMR, also known as treatment-free remission (TFR).
Of the 88 patients who restarted nilotinib due to loss of MMR by the cut-off date, 98.9% were able to regain MMR (n=87). One patient discontinued the study at 7.1 weeks without regaining MMR after reinitiating treatment with nilotinib. Eighty-one patients (92.0%) regained MR4.5 by the cut-off date.
Common adverse events (AEs) in patients who discontinued nilotinib included musculoskeletal symptoms such as body aches, bone pain, and pain in extremities. However, these AEs decreased over time.
The incidence of musculoskeletal pain-related AEs decreased from 34.0% to 9.0% during the first and second 48 weeks of the TFR phase, respectively. In comparison, the incidence of such AEs was 17.0% during the treatment consolidation phase.
ENESTop
This phase 2 trial included 163 patients with Ph+, chronic phase CML. Researchers evaluated stopping treatment in 126 patients who had previously received imatinib, then switched to nilotinib and sustained a molecular response for 1 year prior to stopping nilotinib.
At 96 weeks, 53.2% of patients were still in TFR.
Fifty-six patients with confirmed loss of MR4.0 or loss of MMR restarted nilotinib by the cut-off date. Of these patients, 92.9% (n=52) regained both MR4.0 and MR4.5.
As in ENESTfreedom, patients who discontinued nilotinib had musculoskeletal symptoms that decreased over time.
The incidence of musculoskeletal pain-related AEs decreased from 47.9% to 15.1% during the first and second 48 weeks of the TFR phase, respectively. In comparison, the incidence of such AEs was 13.7% during the treatment consolidation phase.
Additional data from ENESTop and ENESTfreedom, as well as the recommendations for stopping nilotinib, are included in the updated prescribing information, which is available at https://www.us.tasigna.com/. ![]()
The US Food and Drug Administration (FDA) has approved an update to the product label for nilotinib (Tasigna) that includes information about how to discontinue the drug in certain patients.
Nilotinib, which was first approved by the FDA in 2007, is indicated for the treatment of patients with Philadelphia chromosome positive (Ph+) chronic myeloid leukemia (CML).
The updated prescribing information for the drug now outlines which of these patients may be eligible to stop receiving nilotinib.
Patients with chronic phase, Ph+ CML who have been taking nilotinib for 3 years or more (as first-line treatment or after failure with imatinib) and have achieved a sustained deep molecular response may be eligible to stop treatment.
Patients must have maintained a molecular response of at least MR4.0 for 1 year prior to discontinuation and achieved an MR4.5 at their last assessment.
Patients must have typical BCR-ABL transcripts (e13a2/b2a2 or e14a2/b3a2), no history of accelerated phase or blast crisis, and no past attempts at treatment discontinuation that resulted in relapse.
After discontinuation, patients must be monitored for possible loss of major molecular response (MMR). BCR-ABL transcript levels must be assessed and a complete blood count with differential performed monthly for 1 year, then every 6 weeks for the second year, and every 12 weeks thereafter.
BCR-ABL transcript levels should be measured using the MolecularMD MRDxTM BCR-ABL test, an FDA-authorized companion diagnostic validated to measure down to MR4.5.
If patients lose MMR, they must restart nilotinib within 4 weeks and receive the dose level they were receiving prior to discontinuation.
BCR-ABL transcript levels should be monitored every 2 weeks until they are lower than MMR for 4 consecutive measurements. Patients can then return to the original monitoring schedule.
The update of nilotinib’s label to include this information was based on results from 2 single-arm trials—ENESTfreedom and ENESTop.
ENESTfreedom
This phase 2 trial included 215 patients with Ph+, chronic phase CML. Researchers evaluated stopping treatment in 190 patients who had achieved a response of MR4.5 with nilotinib as first-line treatment. The patients had sustained deep molecular response for 1 year prior to treatment discontinuation.
Ninety-six weeks after stopping treatment, 48.9% of patients were still in MMR, also known as treatment-free remission (TFR).
Of the 88 patients who restarted nilotinib due to loss of MMR by the cut-off date, 98.9% were able to regain MMR (n=87). One patient discontinued the study at 7.1 weeks without regaining MMR after reinitiating treatment with nilotinib. Eighty-one patients (92.0%) regained MR4.5 by the cut-off date.
Common adverse events (AEs) in patients who discontinued nilotinib included musculoskeletal symptoms such as body aches, bone pain, and pain in extremities. However, these AEs decreased over time.
The incidence of musculoskeletal pain-related AEs decreased from 34.0% to 9.0% during the first and second 48 weeks of the TFR phase, respectively. In comparison, the incidence of such AEs was 17.0% during the treatment consolidation phase.
ENESTop
This phase 2 trial included 163 patients with Ph+, chronic phase CML. Researchers evaluated stopping treatment in 126 patients who had previously received imatinib, then switched to nilotinib and sustained a molecular response for 1 year prior to stopping nilotinib.
At 96 weeks, 53.2% of patients were still in TFR.
Fifty-six patients with confirmed loss of MR4.0 or loss of MMR restarted nilotinib by the cut-off date. Of these patients, 92.9% (n=52) regained both MR4.0 and MR4.5.
As in ENESTfreedom, patients who discontinued nilotinib had musculoskeletal symptoms that decreased over time.
The incidence of musculoskeletal pain-related AEs decreased from 47.9% to 15.1% during the first and second 48 weeks of the TFR phase, respectively. In comparison, the incidence of such AEs was 13.7% during the treatment consolidation phase.
Additional data from ENESTop and ENESTfreedom, as well as the recommendations for stopping nilotinib, are included in the updated prescribing information, which is available at https://www.us.tasigna.com/. ![]()
FDA expands approved use of bosutinib in CML
The US Food and Drug Administration (FDA) has expanded the approved indication for bosutinib (BOSULIF®).
The tyrosine kinase inhibitor (TKI) is now approved to treat adults with newly diagnosed, chronic phase, Philadelphia chromosome-positive (Ph+) chronic myelogenous leukemia (CML).
Bosutinib has accelerated approval for this indication. The approval was based on molecular and cytogenetic response rates.
Continued approval may be contingent upon verification and confirmation of clinical benefit in an ongoing, long-term follow-up trial.
Bosutinib was first approved by the FDA in September 2012. At that time, the TKI was approved to treat adults with chronic, accelerated, or blast phase Ph+ CML with resistance or intolerance to prior therapy.
A 400 mg tablet of bosutinib was recently approved by the FDA, adding to the previously approved 100 mg and 500 mg strengths.
The recommended dose of bosutinib for newly diagnosed patients is 400 mg orally once daily with food.
For patients who are resistant or intolerant to prior TKI therapy, the recommended dose is 500 mg orally once daily with food.
BFORE trial
The approval of bosutinib in adults with newly diagnosed, chronic phase, Ph+ CML was based on the phase 3 BFORE trial. Results from the trial were presented at the 2017 ASCO Annual Meeting.
In this ongoing study, researchers are comparing bosutinib and imatinib as first-line treatment of chronic phase CML.
As of the ASCO presentation, the trial had enrolled 536 patients who were randomized 1:1 to receive bosutinib (n=268) or imatinib (n=268).
The presentation included results in a modified intent-to-treat population of Ph+ patients with e13a2/e14a2 transcripts who had at least 12 months of follow-up. In this group, there were 246 patients in the bosutinib arm and 241 in the imatinib arm.
Most of the patients were still on therapy at the 12-month mark or beyond—78% in the bosutinib arm and 73.2% in the imatinib arm. The median treatment duration was 14.1 months and 13.8 months, respectively.
At 12 months, the rate of major molecular response was 47.2% in the bosutinib arm and 36.9% in the imatinib arm (P=0.02). The rate of complete cytogenetic response was 77.2% and 66.4%, respectively (P<0.008).
One patient in the bosutinib arm and 4 in the imatinib arm discontinued treatment due to disease progression, while 12.7% and 8.7%, respectively, discontinued treatment due to drug-related toxicity.
Adverse events that were more common in the bosutinib arm than the imatinib arm included grade 3 or higher diarrhea (7.8% vs 0.8%), increased alanine levels (19% vs 1.5%), increased aspartate levels (9.7% vs 1.9%), cardiovascular events (3% vs 0.4%), and peripheral vascular events (1.5% vs 1.1%).
Cerebrovascular events were more common with imatinib than bosutinib (0.4% and 0%, respectively).
Pfizer and Avillion entered into an exclusive collaborative development agreement in 2014 to conduct the BFORE trial.
Under the terms of the agreement, Avillion provided funding and conducted the trial to generate the clinical data used to support regulatory filings for marketing authorization for bosutinib as first-line treatment for patients with chronic phase, Ph+ CML.
With this approval, Avillion is eligible to receive milestone payments from Pfizer. Pfizer retains all rights to commercialize bosutinib globally. ![]()
The US Food and Drug Administration (FDA) has expanded the approved indication for bosutinib (BOSULIF®).
The tyrosine kinase inhibitor (TKI) is now approved to treat adults with newly diagnosed, chronic phase, Philadelphia chromosome-positive (Ph+) chronic myelogenous leukemia (CML).
Bosutinib has accelerated approval for this indication. The approval was based on molecular and cytogenetic response rates.
Continued approval may be contingent upon verification and confirmation of clinical benefit in an ongoing, long-term follow-up trial.
Bosutinib was first approved by the FDA in September 2012. At that time, the TKI was approved to treat adults with chronic, accelerated, or blast phase Ph+ CML with resistance or intolerance to prior therapy.
A 400 mg tablet of bosutinib was recently approved by the FDA, adding to the previously approved 100 mg and 500 mg strengths.
The recommended dose of bosutinib for newly diagnosed patients is 400 mg orally once daily with food.
For patients who are resistant or intolerant to prior TKI therapy, the recommended dose is 500 mg orally once daily with food.
BFORE trial
The approval of bosutinib in adults with newly diagnosed, chronic phase, Ph+ CML was based on the phase 3 BFORE trial. Results from the trial were presented at the 2017 ASCO Annual Meeting.
In this ongoing study, researchers are comparing bosutinib and imatinib as first-line treatment of chronic phase CML.
As of the ASCO presentation, the trial had enrolled 536 patients who were randomized 1:1 to receive bosutinib (n=268) or imatinib (n=268).
The presentation included results in a modified intent-to-treat population of Ph+ patients with e13a2/e14a2 transcripts who had at least 12 months of follow-up. In this group, there were 246 patients in the bosutinib arm and 241 in the imatinib arm.
Most of the patients were still on therapy at the 12-month mark or beyond—78% in the bosutinib arm and 73.2% in the imatinib arm. The median treatment duration was 14.1 months and 13.8 months, respectively.
At 12 months, the rate of major molecular response was 47.2% in the bosutinib arm and 36.9% in the imatinib arm (P=0.02). The rate of complete cytogenetic response was 77.2% and 66.4%, respectively (P<0.008).
One patient in the bosutinib arm and 4 in the imatinib arm discontinued treatment due to disease progression, while 12.7% and 8.7%, respectively, discontinued treatment due to drug-related toxicity.
Adverse events that were more common in the bosutinib arm than the imatinib arm included grade 3 or higher diarrhea (7.8% vs 0.8%), increased alanine levels (19% vs 1.5%), increased aspartate levels (9.7% vs 1.9%), cardiovascular events (3% vs 0.4%), and peripheral vascular events (1.5% vs 1.1%).
Cerebrovascular events were more common with imatinib than bosutinib (0.4% and 0%, respectively).
Pfizer and Avillion entered into an exclusive collaborative development agreement in 2014 to conduct the BFORE trial.
Under the terms of the agreement, Avillion provided funding and conducted the trial to generate the clinical data used to support regulatory filings for marketing authorization for bosutinib as first-line treatment for patients with chronic phase, Ph+ CML.
With this approval, Avillion is eligible to receive milestone payments from Pfizer. Pfizer retains all rights to commercialize bosutinib globally. ![]()
The US Food and Drug Administration (FDA) has expanded the approved indication for bosutinib (BOSULIF®).
The tyrosine kinase inhibitor (TKI) is now approved to treat adults with newly diagnosed, chronic phase, Philadelphia chromosome-positive (Ph+) chronic myelogenous leukemia (CML).
Bosutinib has accelerated approval for this indication. The approval was based on molecular and cytogenetic response rates.
Continued approval may be contingent upon verification and confirmation of clinical benefit in an ongoing, long-term follow-up trial.
Bosutinib was first approved by the FDA in September 2012. At that time, the TKI was approved to treat adults with chronic, accelerated, or blast phase Ph+ CML with resistance or intolerance to prior therapy.
A 400 mg tablet of bosutinib was recently approved by the FDA, adding to the previously approved 100 mg and 500 mg strengths.
The recommended dose of bosutinib for newly diagnosed patients is 400 mg orally once daily with food.
For patients who are resistant or intolerant to prior TKI therapy, the recommended dose is 500 mg orally once daily with food.
BFORE trial
The approval of bosutinib in adults with newly diagnosed, chronic phase, Ph+ CML was based on the phase 3 BFORE trial. Results from the trial were presented at the 2017 ASCO Annual Meeting.
In this ongoing study, researchers are comparing bosutinib and imatinib as first-line treatment of chronic phase CML.
As of the ASCO presentation, the trial had enrolled 536 patients who were randomized 1:1 to receive bosutinib (n=268) or imatinib (n=268).
The presentation included results in a modified intent-to-treat population of Ph+ patients with e13a2/e14a2 transcripts who had at least 12 months of follow-up. In this group, there were 246 patients in the bosutinib arm and 241 in the imatinib arm.
Most of the patients were still on therapy at the 12-month mark or beyond—78% in the bosutinib arm and 73.2% in the imatinib arm. The median treatment duration was 14.1 months and 13.8 months, respectively.
At 12 months, the rate of major molecular response was 47.2% in the bosutinib arm and 36.9% in the imatinib arm (P=0.02). The rate of complete cytogenetic response was 77.2% and 66.4%, respectively (P<0.008).
One patient in the bosutinib arm and 4 in the imatinib arm discontinued treatment due to disease progression, while 12.7% and 8.7%, respectively, discontinued treatment due to drug-related toxicity.
Adverse events that were more common in the bosutinib arm than the imatinib arm included grade 3 or higher diarrhea (7.8% vs 0.8%), increased alanine levels (19% vs 1.5%), increased aspartate levels (9.7% vs 1.9%), cardiovascular events (3% vs 0.4%), and peripheral vascular events (1.5% vs 1.1%).
Cerebrovascular events were more common with imatinib than bosutinib (0.4% and 0%, respectively).
Pfizer and Avillion entered into an exclusive collaborative development agreement in 2014 to conduct the BFORE trial.
Under the terms of the agreement, Avillion provided funding and conducted the trial to generate the clinical data used to support regulatory filings for marketing authorization for bosutinib as first-line treatment for patients with chronic phase, Ph+ CML.
With this approval, Avillion is eligible to receive milestone payments from Pfizer. Pfizer retains all rights to commercialize bosutinib globally. ![]()
Company launches digital PCR test for monitoring CML
Bio-Rad Laboratories, Inc., has launched the QXDx BCR-ABL %IS Kit, a digital polymerase chain reaction (PCR) test that can monitor molecular response to therapy in patients with chronic myeloid leukemia (CML).
The kit has a CE-IVD mark and is available for in vitro diagnostic use in Europe, Hong Kong, and New Zealand.
The QXDx BCR-ABL %IS Kit uses Bio-Rad’s Droplet Digital PCR (ddPCR) technology to provide an absolute measure of BCR-ABL transcripts.
The kit measures BCR-ABL1 and ABL1 chromosomal transcripts in total RNA from whole blood of t(9;22)-positive CML patients expressing BCR-ABL1 fusion transcripts type e13a2 and/or e14a2.
The QXDx BCR-ABL %IS Kit measures the e13a2 and/or e14a2 transcripts of BCR-ABL1, normalized to the ABL1 endogenous control. The kit does not differentiate between e13a2 and e14a2 transcripts and does not monitor other rare fusion transcripts resulting from t(9;22).
The kit’s results are reported as percent reduction from a baseline of 100% on the International Scale (%IS) and on a log molecular reduction (MR) scale.
The kit is able to detect deep molecular response values of MR 4.7 (%IS 0.002) or MR 5.0 (%IS 0.001) in 2- or 4-well formats. This exceeds the typical limitations of reverse transcription quantitative PCR-based tests that are reliable down to MR 4.5 (%IS 0.0032).
Bio-Rad says the QXDx BCR-ABL %IS Kit delivers absolute quantitation, which eliminates the need for standard curves and minimizes variation between samples. The kit also provides scalable throughput, allowing for testing of 8 to 48 samples per run.
The QXDx BCR-ABL %IS Kit can be used with Bio-Rad’s QX200 AutoDG ddPCR Dx System or with the QX200 ddPCR Dx System. The QXDx BCR-ABL %IS Kit uses QuantaSoft Software v1.7 for data acquisition and output. ![]()
Bio-Rad Laboratories, Inc., has launched the QXDx BCR-ABL %IS Kit, a digital polymerase chain reaction (PCR) test that can monitor molecular response to therapy in patients with chronic myeloid leukemia (CML).
The kit has a CE-IVD mark and is available for in vitro diagnostic use in Europe, Hong Kong, and New Zealand.
The QXDx BCR-ABL %IS Kit uses Bio-Rad’s Droplet Digital PCR (ddPCR) technology to provide an absolute measure of BCR-ABL transcripts.
The kit measures BCR-ABL1 and ABL1 chromosomal transcripts in total RNA from whole blood of t(9;22)-positive CML patients expressing BCR-ABL1 fusion transcripts type e13a2 and/or e14a2.
The QXDx BCR-ABL %IS Kit measures the e13a2 and/or e14a2 transcripts of BCR-ABL1, normalized to the ABL1 endogenous control. The kit does not differentiate between e13a2 and e14a2 transcripts and does not monitor other rare fusion transcripts resulting from t(9;22).
The kit’s results are reported as percent reduction from a baseline of 100% on the International Scale (%IS) and on a log molecular reduction (MR) scale.
The kit is able to detect deep molecular response values of MR 4.7 (%IS 0.002) or MR 5.0 (%IS 0.001) in 2- or 4-well formats. This exceeds the typical limitations of reverse transcription quantitative PCR-based tests that are reliable down to MR 4.5 (%IS 0.0032).
Bio-Rad says the QXDx BCR-ABL %IS Kit delivers absolute quantitation, which eliminates the need for standard curves and minimizes variation between samples. The kit also provides scalable throughput, allowing for testing of 8 to 48 samples per run.
The QXDx BCR-ABL %IS Kit can be used with Bio-Rad’s QX200 AutoDG ddPCR Dx System or with the QX200 ddPCR Dx System. The QXDx BCR-ABL %IS Kit uses QuantaSoft Software v1.7 for data acquisition and output. ![]()
Bio-Rad Laboratories, Inc., has launched the QXDx BCR-ABL %IS Kit, a digital polymerase chain reaction (PCR) test that can monitor molecular response to therapy in patients with chronic myeloid leukemia (CML).
The kit has a CE-IVD mark and is available for in vitro diagnostic use in Europe, Hong Kong, and New Zealand.
The QXDx BCR-ABL %IS Kit uses Bio-Rad’s Droplet Digital PCR (ddPCR) technology to provide an absolute measure of BCR-ABL transcripts.
The kit measures BCR-ABL1 and ABL1 chromosomal transcripts in total RNA from whole blood of t(9;22)-positive CML patients expressing BCR-ABL1 fusion transcripts type e13a2 and/or e14a2.
The QXDx BCR-ABL %IS Kit measures the e13a2 and/or e14a2 transcripts of BCR-ABL1, normalized to the ABL1 endogenous control. The kit does not differentiate between e13a2 and e14a2 transcripts and does not monitor other rare fusion transcripts resulting from t(9;22).
The kit’s results are reported as percent reduction from a baseline of 100% on the International Scale (%IS) and on a log molecular reduction (MR) scale.
The kit is able to detect deep molecular response values of MR 4.7 (%IS 0.002) or MR 5.0 (%IS 0.001) in 2- or 4-well formats. This exceeds the typical limitations of reverse transcription quantitative PCR-based tests that are reliable down to MR 4.5 (%IS 0.0032).
Bio-Rad says the QXDx BCR-ABL %IS Kit delivers absolute quantitation, which eliminates the need for standard curves and minimizes variation between samples. The kit also provides scalable throughput, allowing for testing of 8 to 48 samples per run.
The QXDx BCR-ABL %IS Kit can be used with Bio-Rad’s QX200 AutoDG ddPCR Dx System or with the QX200 ddPCR Dx System. The QXDx BCR-ABL %IS Kit uses QuantaSoft Software v1.7 for data acquisition and output. ![]()
Companies launch generic busulfan in US
Two companies have announced the US launch of a generic busulfan product, Myleran Injection.
Mylan NV and Aspen have partnered to develop Myleran (busulfan) Injection, 60 mg/10 mL (6 mg/mL) Single-dose Vial, a generic version of Otsuka Pharmaceutical’s Busulfex® Injection.
The US Food and Drug Administration approved Myleran Injection for use in combination with cyclophosphamide as a conditioning regimen prior to allogeneic hematopoietic stem cell transplant in patients with chronic myeloid leukemia. ![]()
Two companies have announced the US launch of a generic busulfan product, Myleran Injection.
Mylan NV and Aspen have partnered to develop Myleran (busulfan) Injection, 60 mg/10 mL (6 mg/mL) Single-dose Vial, a generic version of Otsuka Pharmaceutical’s Busulfex® Injection.
The US Food and Drug Administration approved Myleran Injection for use in combination with cyclophosphamide as a conditioning regimen prior to allogeneic hematopoietic stem cell transplant in patients with chronic myeloid leukemia. ![]()
Two companies have announced the US launch of a generic busulfan product, Myleran Injection.
Mylan NV and Aspen have partnered to develop Myleran (busulfan) Injection, 60 mg/10 mL (6 mg/mL) Single-dose Vial, a generic version of Otsuka Pharmaceutical’s Busulfex® Injection.
The US Food and Drug Administration approved Myleran Injection for use in combination with cyclophosphamide as a conditioning regimen prior to allogeneic hematopoietic stem cell transplant in patients with chronic myeloid leukemia. ![]()
Nilotinib approved to treat kids with CML in EU
The European Commission has approved nilotinib (Tasigna®) for the treatment of pediatric patients.
The drug is now approved to treat children age 2 and older with newly diagnosed, Philadelphia chromosome-positive (Ph+), chronic phase (CP) chronic myeloid leukemia (CML) or Ph+ CP-CML with resistance or intolerance to prior therapy, including imatinib.
Nilotinib is the only second-generation tyrosine kinase inhibitor currently approved in the European Union (EU) for the treatment of Ph+ CP-CML in children. The approval applies to all EU member states.
According to Novartis, the expanded indication for nilotinib is based on 2 prospective studies of the drug in children with Ph+ CP-CML, which were part of a formal “pediatric investigation plan” agreed upon with the European Medicines Agency.
The company said 69 patients received nilotinib in these studies. The patients ranged in age from 2 to 18. They had either newly diagnosed Ph+ CP-CML or Ph+ CP-CML with resistance or intolerance to prior therapy, including imatinib.
In the newly diagnosed patients, the major molecular response (MMR) rate was 60.0% (95% CI: 38.7, 78.9) at 12 cycles, with 15 patients achieving MMR.
In patients with resistance or intolerance to prior therapy, the MMR rate was 40.9% (95% CI: 26.3, 56.8) at 12 cycles, with 18 patients being in MMR.
In newly diagnosed patients, the cumulative MMR rate was 64.0% by cycle 12. In patients with resistance or intolerance to prior therapy, the cumulative MMR rate was 47.7% by cycle 12.
Adverse events were generally consistent with those observed in adults, with the exception of hyperbilirubinemia and transaminase elevation, which were reported at a higher frequency than in adults.
The rate of grade 3/4 hyperbilirubinemia was 13.0%, the rate of grade 3/4 AST elevation was 1.4%, and the rate of grade 3/4 ALT elevation was 8.7%.
There were no deaths on treatment or after treatment discontinuation. ![]()
The European Commission has approved nilotinib (Tasigna®) for the treatment of pediatric patients.
The drug is now approved to treat children age 2 and older with newly diagnosed, Philadelphia chromosome-positive (Ph+), chronic phase (CP) chronic myeloid leukemia (CML) or Ph+ CP-CML with resistance or intolerance to prior therapy, including imatinib.
Nilotinib is the only second-generation tyrosine kinase inhibitor currently approved in the European Union (EU) for the treatment of Ph+ CP-CML in children. The approval applies to all EU member states.
According to Novartis, the expanded indication for nilotinib is based on 2 prospective studies of the drug in children with Ph+ CP-CML, which were part of a formal “pediatric investigation plan” agreed upon with the European Medicines Agency.
The company said 69 patients received nilotinib in these studies. The patients ranged in age from 2 to 18. They had either newly diagnosed Ph+ CP-CML or Ph+ CP-CML with resistance or intolerance to prior therapy, including imatinib.
In the newly diagnosed patients, the major molecular response (MMR) rate was 60.0% (95% CI: 38.7, 78.9) at 12 cycles, with 15 patients achieving MMR.
In patients with resistance or intolerance to prior therapy, the MMR rate was 40.9% (95% CI: 26.3, 56.8) at 12 cycles, with 18 patients being in MMR.
In newly diagnosed patients, the cumulative MMR rate was 64.0% by cycle 12. In patients with resistance or intolerance to prior therapy, the cumulative MMR rate was 47.7% by cycle 12.
Adverse events were generally consistent with those observed in adults, with the exception of hyperbilirubinemia and transaminase elevation, which were reported at a higher frequency than in adults.
The rate of grade 3/4 hyperbilirubinemia was 13.0%, the rate of grade 3/4 AST elevation was 1.4%, and the rate of grade 3/4 ALT elevation was 8.7%.
There were no deaths on treatment or after treatment discontinuation. ![]()
The European Commission has approved nilotinib (Tasigna®) for the treatment of pediatric patients.
The drug is now approved to treat children age 2 and older with newly diagnosed, Philadelphia chromosome-positive (Ph+), chronic phase (CP) chronic myeloid leukemia (CML) or Ph+ CP-CML with resistance or intolerance to prior therapy, including imatinib.
Nilotinib is the only second-generation tyrosine kinase inhibitor currently approved in the European Union (EU) for the treatment of Ph+ CP-CML in children. The approval applies to all EU member states.
According to Novartis, the expanded indication for nilotinib is based on 2 prospective studies of the drug in children with Ph+ CP-CML, which were part of a formal “pediatric investigation plan” agreed upon with the European Medicines Agency.
The company said 69 patients received nilotinib in these studies. The patients ranged in age from 2 to 18. They had either newly diagnosed Ph+ CP-CML or Ph+ CP-CML with resistance or intolerance to prior therapy, including imatinib.
In the newly diagnosed patients, the major molecular response (MMR) rate was 60.0% (95% CI: 38.7, 78.9) at 12 cycles, with 15 patients achieving MMR.
In patients with resistance or intolerance to prior therapy, the MMR rate was 40.9% (95% CI: 26.3, 56.8) at 12 cycles, with 18 patients being in MMR.
In newly diagnosed patients, the cumulative MMR rate was 64.0% by cycle 12. In patients with resistance or intolerance to prior therapy, the cumulative MMR rate was 47.7% by cycle 12.
Adverse events were generally consistent with those observed in adults, with the exception of hyperbilirubinemia and transaminase elevation, which were reported at a higher frequency than in adults.
The rate of grade 3/4 hyperbilirubinemia was 13.0%, the rate of grade 3/4 AST elevation was 1.4%, and the rate of grade 3/4 ALT elevation was 8.7%.
There were no deaths on treatment or after treatment discontinuation.