User login
Adjuvant immunotherapy results ‘encouraging’ in early NSCLC
CHICAGO – Neoadjuvant monotherapy with the immune checkpoint inhibitor atezolizumab is associated with “encouraging” responses with no new safety signals for patients with non–small cell lung cancer (NSCLC), an interim analysis of a multicenter phase 2 trial suggests.
Among 77 of a planned 180 patients with resectable NSCLC enrolled in the LCMC3 (Lung Cancer Mutation Consortium 3) trial, the pathological complete response (pCR) rate following two cycles of neoadjuvant atezolizumab (Tecentriq) and surgery was 5%, and the major pathological response (MPR) rate was 19%, reported David J. Kwiatkowski, MD, PhD, of the Dana-Farber Cancer Institute in Boston.
“Pathological regression moderately correlated with target lesions’ measurements by RECIST [Response Evaluation Criteria in Solid Tumors] and MPR was observed irrespective of PD-L1 expression, although there was some correlation,” he said at the annual meeting of the American Society of Clinical Oncology.
The study was designed to test whether preoperative immunotherapy with an immune checkpoint inhibitor could have additional clinical benefits for patients with early-stage NSCLC.
Investigators are enrolling patients with stage IB, II, IIIA, or selected IIIB resectable, previously untreated NSCLC. Patients receive 1,200 mg atezolizumab on days 1 and 22 (two cycles), followed by surgery on or about day 40.
The primary endpoint, MPR, “means that at the time of surgical resection, all of the samples of the tumor that are cut into sections are reviewed by a pathologist, and an aggregate score of a percent of viable tumor cells is determined based on a comparison of viable tumor cells and necrotic tumor cells and stroma,” Dr. Kwiatkowski said.
The threshold for MPR was 10% or fewer viable tumor cells at the time of resection.
Following surgery, patients received standard-of-care adjuvant chemotherapy and could receive optional continued atezolizumab for an additional 12 months.
At the time of this interim analysis, with a data cutoff of Sept. 5, 2018, 101 patients had been enrolled and were included in the interim safety analysis. Of this group, 11 did not undergo surgery, because of progressive disease, withdrawal of consent, failed echocardiogram (1 patient), or pulmonary artery involvement (1) patient.
Of the 10 patients with either progressive disease and no surgery or unresectable disease at surgery, 8 had stage IIIA tumors and 2 had stage IIIB tumors. All patients with stage I or II disease underwent resection.
Dr. Kwiatkowski presented interim data on 90 patients intended for surgery, of whom 84 had assessment of the primary endpoint, including 7 positive for EGFR and/or ALK, and 77 whose tumors were either EGFR/ALK negative or had unknown status. These 77 patients were the primary efficacy population.
As noted before, among the 77 in the primary efficacy population, 15 (19%) had a MPR, and 4 patients (5%) had a pCR. In addition, 38 patients (49%) had pathological regression of tumor of 50% or greater. Pathological regression correlated significantly with change in tumor lesion size (P less than .001).
Tumor mutational burden, however, was not significantly correlated with MPR or pathological regression.
Among the 101 patients in the safety population, there were two deaths deemed not related to study treatment: one cardiac death post surgical resection, and one from disease progression. Treatment-related adverse events occurred in 57% of patients, including 6% that were grade 3 or greater. Adverse events leading to treatment withdrawal occurred in 5% of patients.
The efficacy interim analysis passed the prespecified futility boundary, and investigators are continuing to enroll patients.
Invited discussant Maximilian Diehn, MD, PhD, of Stanford (Calif.) University commented that neoadjuvant immunotherapy for NSCLC is promising, but added that the MPR endpoint still needs validation.
“Currently, it is not considered a validated surrogate endpoint for survival and therefore is not currently used for drug approvals. Secondly, the optimal cut point may differ by histology, such as being different for adenocarcinoma and squamous cell carcinoma. And this has potential implications for using this in trials that enroll patients of both histologies. And, third, there are some emerging data that MPR may need to measured somewhat differently after immunotherapy than after chemotherapy,” he said.
The study is supported by Genentech. Dr. Kwiatkowski disclosed research funding and a consulting or advisory role for the company. Dr. Diehn reported stock ownership, consulting, research funding, and travel expenses from various companies.
SOURCE: Kwiatkowski DJ et al. ASCO 2019, Abstract 8503.
CHICAGO – Neoadjuvant monotherapy with the immune checkpoint inhibitor atezolizumab is associated with “encouraging” responses with no new safety signals for patients with non–small cell lung cancer (NSCLC), an interim analysis of a multicenter phase 2 trial suggests.
Among 77 of a planned 180 patients with resectable NSCLC enrolled in the LCMC3 (Lung Cancer Mutation Consortium 3) trial, the pathological complete response (pCR) rate following two cycles of neoadjuvant atezolizumab (Tecentriq) and surgery was 5%, and the major pathological response (MPR) rate was 19%, reported David J. Kwiatkowski, MD, PhD, of the Dana-Farber Cancer Institute in Boston.
“Pathological regression moderately correlated with target lesions’ measurements by RECIST [Response Evaluation Criteria in Solid Tumors] and MPR was observed irrespective of PD-L1 expression, although there was some correlation,” he said at the annual meeting of the American Society of Clinical Oncology.
The study was designed to test whether preoperative immunotherapy with an immune checkpoint inhibitor could have additional clinical benefits for patients with early-stage NSCLC.
Investigators are enrolling patients with stage IB, II, IIIA, or selected IIIB resectable, previously untreated NSCLC. Patients receive 1,200 mg atezolizumab on days 1 and 22 (two cycles), followed by surgery on or about day 40.
The primary endpoint, MPR, “means that at the time of surgical resection, all of the samples of the tumor that are cut into sections are reviewed by a pathologist, and an aggregate score of a percent of viable tumor cells is determined based on a comparison of viable tumor cells and necrotic tumor cells and stroma,” Dr. Kwiatkowski said.
The threshold for MPR was 10% or fewer viable tumor cells at the time of resection.
Following surgery, patients received standard-of-care adjuvant chemotherapy and could receive optional continued atezolizumab for an additional 12 months.
At the time of this interim analysis, with a data cutoff of Sept. 5, 2018, 101 patients had been enrolled and were included in the interim safety analysis. Of this group, 11 did not undergo surgery, because of progressive disease, withdrawal of consent, failed echocardiogram (1 patient), or pulmonary artery involvement (1) patient.
Of the 10 patients with either progressive disease and no surgery or unresectable disease at surgery, 8 had stage IIIA tumors and 2 had stage IIIB tumors. All patients with stage I or II disease underwent resection.
Dr. Kwiatkowski presented interim data on 90 patients intended for surgery, of whom 84 had assessment of the primary endpoint, including 7 positive for EGFR and/or ALK, and 77 whose tumors were either EGFR/ALK negative or had unknown status. These 77 patients were the primary efficacy population.
As noted before, among the 77 in the primary efficacy population, 15 (19%) had a MPR, and 4 patients (5%) had a pCR. In addition, 38 patients (49%) had pathological regression of tumor of 50% or greater. Pathological regression correlated significantly with change in tumor lesion size (P less than .001).
Tumor mutational burden, however, was not significantly correlated with MPR or pathological regression.
Among the 101 patients in the safety population, there were two deaths deemed not related to study treatment: one cardiac death post surgical resection, and one from disease progression. Treatment-related adverse events occurred in 57% of patients, including 6% that were grade 3 or greater. Adverse events leading to treatment withdrawal occurred in 5% of patients.
The efficacy interim analysis passed the prespecified futility boundary, and investigators are continuing to enroll patients.
Invited discussant Maximilian Diehn, MD, PhD, of Stanford (Calif.) University commented that neoadjuvant immunotherapy for NSCLC is promising, but added that the MPR endpoint still needs validation.
“Currently, it is not considered a validated surrogate endpoint for survival and therefore is not currently used for drug approvals. Secondly, the optimal cut point may differ by histology, such as being different for adenocarcinoma and squamous cell carcinoma. And this has potential implications for using this in trials that enroll patients of both histologies. And, third, there are some emerging data that MPR may need to measured somewhat differently after immunotherapy than after chemotherapy,” he said.
The study is supported by Genentech. Dr. Kwiatkowski disclosed research funding and a consulting or advisory role for the company. Dr. Diehn reported stock ownership, consulting, research funding, and travel expenses from various companies.
SOURCE: Kwiatkowski DJ et al. ASCO 2019, Abstract 8503.
CHICAGO – Neoadjuvant monotherapy with the immune checkpoint inhibitor atezolizumab is associated with “encouraging” responses with no new safety signals for patients with non–small cell lung cancer (NSCLC), an interim analysis of a multicenter phase 2 trial suggests.
Among 77 of a planned 180 patients with resectable NSCLC enrolled in the LCMC3 (Lung Cancer Mutation Consortium 3) trial, the pathological complete response (pCR) rate following two cycles of neoadjuvant atezolizumab (Tecentriq) and surgery was 5%, and the major pathological response (MPR) rate was 19%, reported David J. Kwiatkowski, MD, PhD, of the Dana-Farber Cancer Institute in Boston.
“Pathological regression moderately correlated with target lesions’ measurements by RECIST [Response Evaluation Criteria in Solid Tumors] and MPR was observed irrespective of PD-L1 expression, although there was some correlation,” he said at the annual meeting of the American Society of Clinical Oncology.
The study was designed to test whether preoperative immunotherapy with an immune checkpoint inhibitor could have additional clinical benefits for patients with early-stage NSCLC.
Investigators are enrolling patients with stage IB, II, IIIA, or selected IIIB resectable, previously untreated NSCLC. Patients receive 1,200 mg atezolizumab on days 1 and 22 (two cycles), followed by surgery on or about day 40.
The primary endpoint, MPR, “means that at the time of surgical resection, all of the samples of the tumor that are cut into sections are reviewed by a pathologist, and an aggregate score of a percent of viable tumor cells is determined based on a comparison of viable tumor cells and necrotic tumor cells and stroma,” Dr. Kwiatkowski said.
The threshold for MPR was 10% or fewer viable tumor cells at the time of resection.
Following surgery, patients received standard-of-care adjuvant chemotherapy and could receive optional continued atezolizumab for an additional 12 months.
At the time of this interim analysis, with a data cutoff of Sept. 5, 2018, 101 patients had been enrolled and were included in the interim safety analysis. Of this group, 11 did not undergo surgery, because of progressive disease, withdrawal of consent, failed echocardiogram (1 patient), or pulmonary artery involvement (1) patient.
Of the 10 patients with either progressive disease and no surgery or unresectable disease at surgery, 8 had stage IIIA tumors and 2 had stage IIIB tumors. All patients with stage I or II disease underwent resection.
Dr. Kwiatkowski presented interim data on 90 patients intended for surgery, of whom 84 had assessment of the primary endpoint, including 7 positive for EGFR and/or ALK, and 77 whose tumors were either EGFR/ALK negative or had unknown status. These 77 patients were the primary efficacy population.
As noted before, among the 77 in the primary efficacy population, 15 (19%) had a MPR, and 4 patients (5%) had a pCR. In addition, 38 patients (49%) had pathological regression of tumor of 50% or greater. Pathological regression correlated significantly with change in tumor lesion size (P less than .001).
Tumor mutational burden, however, was not significantly correlated with MPR or pathological regression.
Among the 101 patients in the safety population, there were two deaths deemed not related to study treatment: one cardiac death post surgical resection, and one from disease progression. Treatment-related adverse events occurred in 57% of patients, including 6% that were grade 3 or greater. Adverse events leading to treatment withdrawal occurred in 5% of patients.
The efficacy interim analysis passed the prespecified futility boundary, and investigators are continuing to enroll patients.
Invited discussant Maximilian Diehn, MD, PhD, of Stanford (Calif.) University commented that neoadjuvant immunotherapy for NSCLC is promising, but added that the MPR endpoint still needs validation.
“Currently, it is not considered a validated surrogate endpoint for survival and therefore is not currently used for drug approvals. Secondly, the optimal cut point may differ by histology, such as being different for adenocarcinoma and squamous cell carcinoma. And this has potential implications for using this in trials that enroll patients of both histologies. And, third, there are some emerging data that MPR may need to measured somewhat differently after immunotherapy than after chemotherapy,” he said.
The study is supported by Genentech. Dr. Kwiatkowski disclosed research funding and a consulting or advisory role for the company. Dr. Diehn reported stock ownership, consulting, research funding, and travel expenses from various companies.
SOURCE: Kwiatkowski DJ et al. ASCO 2019, Abstract 8503.
REPORTING FROM ASCO 2019
Nivo/ipi shrinks early NSCLC before surgery
CHICAGO – Two immune checkpoint inhibitors were better than one as neoadjuvant therapy for patients with resectable early-stage non–small cell lung cancer (NSCLC) in the phase 2 NEOSTAR trial.
Among 44 patients with stage I-IIIA NSCLC who were randomized to either a combination of nivolumab (Opdivo) and ipilimumab (Yervoy) or to nivolumab alone, the combination was associated with higher rates of the primary endpoint of major pathological response (MPR), defined as a reduction in viable tumors cells to 10% or less, reported Tina Cascone, MD, PhD, from the University of Texas MD Anderson Cancer Center in Houston.
“Nivolumab/ipilimumab induced a 44% MPR rate in resected patients, met the trial prespecified boundary with seven MPRs in the intention-to-treat population, and induced pathologic complete responses in 38% of resected patients,” she said at the annual meeting of the American Society of Clinical Oncology.
To test whether neoadjuvant monotherapy or combination therapy could improve outcomes of standard induction chemotherapy, NEOSTAR investigators enrolled patients with NSCLC stage I-IIIA, including patients with a single involved mediastinal node (N2 single station) who were eligible for surgical resection.
The patients were randomized on a 1:1 basis to receive nivolumab 3 mg/kg on days 1, 15 and 29 alone or in combination with ipilimumab delivered 1 mg/kg on day 1, followed by surgery 3-6 weeks after the last study dose and then postoperative standard-of-care chemotherapy.
Of 53 patients screened, 44 were eligible, with 23 randomized to nivolumab monotherapy and 21 randomized to nivolumab/ipilimumab. Of this group, five did not proceed to surgery (one in the monotherapy arm, four in the combination arm) because of either high surgical risk, lack of respectability, or refusal of surgery. The mean age at randomization was 65.6 years. In all, 18% were never smokers, and the remaining 82% were former or current smokers.
The MPR rate in the intention-to-treat population – the primary endpoint – was reached in four patients (17%) in the monotherapy arm and in seven patients (33%) in the combination arm. As noted, the combination arm reached the prespecified boundary of six or more patients with an MPR. All patients in each arm who had an MPR also had a pathologic complete responses.
Of the 39 patients who went on to resection, 37 were evaluable, and in these patients the respective MPR rates were 19% and 44%. Two patients on nivolumab alone and six on nivolumab plus ipilimumab had 0% viable tumor detectable at the time of surgery. Radiographic responses included one complete response in the combination arm and eight total partial responses, in five and three patients, respectively. The objective response rated was 20%. The responses, assessed by Response Evaluation Criteria in Solid Tumors (RECIST) were positively associated with MPR, Dr. Cascone said.
In 11% of patients, the investigators observed apparent radiographic progression after neoadjuvant immune checkpoint inhibitors in mediastinal and or in nonregional nodes. However, pathological assessment and evaluation of the flaring nodes did not reveal evidence of disease, but instead showed noncaseating granulomas that were not present at baseline.
“Awareness of this phenomenon, which we named the ‘nodal immune flare,’ is of critical importance, as if the clinician fails to distinguish the nodal immune flare from disease progression, potential curative surgery for these patients could be avoided,” she said.
Grade 1 or 2 treatment-related adverse events included rash, itching, fatigue, anemia, cough, and diarrhea. Grade 3 or greater treatment-related adverse events included hypoxia, pneumonia, and pneumonitis in the nivolumab monotherapy arm and diarrhea and hyponatremia in the combination group. One patient treated with nivolumab monotherapy, who had achieved 0% viable tumor, had grade 3 pneumonia and pneumonitis, which was treated with steroids that impeded the healing of a bronchopleural fistula and subsequent empyema. Other surgical complications included air leaks, which occurred in five patients in the nivolumab group and three in the nivolumab plus ipilimumab arm.
Two patients died, one in the monotherapy arm from steroid-treated pneumonitis 4.1 months after randomization and one in the combination arm who had progressive disease 2 months after randomization, and died from the disease 15 months later.
Invited discussant Maximilian Diehn, MD, PhD, from Stanford (California) University School of Medicine, commented that the choice of neoadjuvant immunotherapy was not based on molecular markers, “and I think we have a major unmet need for developing biomarkers for personalized treatment in this area.
“Ideally, the biomarkers that we would have in this setting would, A, allow us to identify which patients have micrometastatic disease and therefore are likely to benefit from the upfront systemic therapy and, secondly, also could tell us which neoadjuvant therapy they would respond to, be it immunotherapy, chemotherapy, or the combination,” he added.
The study was supported by Bristol-Myers Squibb. Dr. Cascone disclosed honoraria from the company. Dr. Diehn reported stock ownership, consulting, research funding and travel expenses from various companies.
SOURCE: Cascone T. et al. ASCO 2019, Abstract 8504.
CHICAGO – Two immune checkpoint inhibitors were better than one as neoadjuvant therapy for patients with resectable early-stage non–small cell lung cancer (NSCLC) in the phase 2 NEOSTAR trial.
Among 44 patients with stage I-IIIA NSCLC who were randomized to either a combination of nivolumab (Opdivo) and ipilimumab (Yervoy) or to nivolumab alone, the combination was associated with higher rates of the primary endpoint of major pathological response (MPR), defined as a reduction in viable tumors cells to 10% or less, reported Tina Cascone, MD, PhD, from the University of Texas MD Anderson Cancer Center in Houston.
“Nivolumab/ipilimumab induced a 44% MPR rate in resected patients, met the trial prespecified boundary with seven MPRs in the intention-to-treat population, and induced pathologic complete responses in 38% of resected patients,” she said at the annual meeting of the American Society of Clinical Oncology.
To test whether neoadjuvant monotherapy or combination therapy could improve outcomes of standard induction chemotherapy, NEOSTAR investigators enrolled patients with NSCLC stage I-IIIA, including patients with a single involved mediastinal node (N2 single station) who were eligible for surgical resection.
The patients were randomized on a 1:1 basis to receive nivolumab 3 mg/kg on days 1, 15 and 29 alone or in combination with ipilimumab delivered 1 mg/kg on day 1, followed by surgery 3-6 weeks after the last study dose and then postoperative standard-of-care chemotherapy.
Of 53 patients screened, 44 were eligible, with 23 randomized to nivolumab monotherapy and 21 randomized to nivolumab/ipilimumab. Of this group, five did not proceed to surgery (one in the monotherapy arm, four in the combination arm) because of either high surgical risk, lack of respectability, or refusal of surgery. The mean age at randomization was 65.6 years. In all, 18% were never smokers, and the remaining 82% were former or current smokers.
The MPR rate in the intention-to-treat population – the primary endpoint – was reached in four patients (17%) in the monotherapy arm and in seven patients (33%) in the combination arm. As noted, the combination arm reached the prespecified boundary of six or more patients with an MPR. All patients in each arm who had an MPR also had a pathologic complete responses.
Of the 39 patients who went on to resection, 37 were evaluable, and in these patients the respective MPR rates were 19% and 44%. Two patients on nivolumab alone and six on nivolumab plus ipilimumab had 0% viable tumor detectable at the time of surgery. Radiographic responses included one complete response in the combination arm and eight total partial responses, in five and three patients, respectively. The objective response rated was 20%. The responses, assessed by Response Evaluation Criteria in Solid Tumors (RECIST) were positively associated with MPR, Dr. Cascone said.
In 11% of patients, the investigators observed apparent radiographic progression after neoadjuvant immune checkpoint inhibitors in mediastinal and or in nonregional nodes. However, pathological assessment and evaluation of the flaring nodes did not reveal evidence of disease, but instead showed noncaseating granulomas that were not present at baseline.
“Awareness of this phenomenon, which we named the ‘nodal immune flare,’ is of critical importance, as if the clinician fails to distinguish the nodal immune flare from disease progression, potential curative surgery for these patients could be avoided,” she said.
Grade 1 or 2 treatment-related adverse events included rash, itching, fatigue, anemia, cough, and diarrhea. Grade 3 or greater treatment-related adverse events included hypoxia, pneumonia, and pneumonitis in the nivolumab monotherapy arm and diarrhea and hyponatremia in the combination group. One patient treated with nivolumab monotherapy, who had achieved 0% viable tumor, had grade 3 pneumonia and pneumonitis, which was treated with steroids that impeded the healing of a bronchopleural fistula and subsequent empyema. Other surgical complications included air leaks, which occurred in five patients in the nivolumab group and three in the nivolumab plus ipilimumab arm.
Two patients died, one in the monotherapy arm from steroid-treated pneumonitis 4.1 months after randomization and one in the combination arm who had progressive disease 2 months after randomization, and died from the disease 15 months later.
Invited discussant Maximilian Diehn, MD, PhD, from Stanford (California) University School of Medicine, commented that the choice of neoadjuvant immunotherapy was not based on molecular markers, “and I think we have a major unmet need for developing biomarkers for personalized treatment in this area.
“Ideally, the biomarkers that we would have in this setting would, A, allow us to identify which patients have micrometastatic disease and therefore are likely to benefit from the upfront systemic therapy and, secondly, also could tell us which neoadjuvant therapy they would respond to, be it immunotherapy, chemotherapy, or the combination,” he added.
The study was supported by Bristol-Myers Squibb. Dr. Cascone disclosed honoraria from the company. Dr. Diehn reported stock ownership, consulting, research funding and travel expenses from various companies.
SOURCE: Cascone T. et al. ASCO 2019, Abstract 8504.
CHICAGO – Two immune checkpoint inhibitors were better than one as neoadjuvant therapy for patients with resectable early-stage non–small cell lung cancer (NSCLC) in the phase 2 NEOSTAR trial.
Among 44 patients with stage I-IIIA NSCLC who were randomized to either a combination of nivolumab (Opdivo) and ipilimumab (Yervoy) or to nivolumab alone, the combination was associated with higher rates of the primary endpoint of major pathological response (MPR), defined as a reduction in viable tumors cells to 10% or less, reported Tina Cascone, MD, PhD, from the University of Texas MD Anderson Cancer Center in Houston.
“Nivolumab/ipilimumab induced a 44% MPR rate in resected patients, met the trial prespecified boundary with seven MPRs in the intention-to-treat population, and induced pathologic complete responses in 38% of resected patients,” she said at the annual meeting of the American Society of Clinical Oncology.
To test whether neoadjuvant monotherapy or combination therapy could improve outcomes of standard induction chemotherapy, NEOSTAR investigators enrolled patients with NSCLC stage I-IIIA, including patients with a single involved mediastinal node (N2 single station) who were eligible for surgical resection.
The patients were randomized on a 1:1 basis to receive nivolumab 3 mg/kg on days 1, 15 and 29 alone or in combination with ipilimumab delivered 1 mg/kg on day 1, followed by surgery 3-6 weeks after the last study dose and then postoperative standard-of-care chemotherapy.
Of 53 patients screened, 44 were eligible, with 23 randomized to nivolumab monotherapy and 21 randomized to nivolumab/ipilimumab. Of this group, five did not proceed to surgery (one in the monotherapy arm, four in the combination arm) because of either high surgical risk, lack of respectability, or refusal of surgery. The mean age at randomization was 65.6 years. In all, 18% were never smokers, and the remaining 82% were former or current smokers.
The MPR rate in the intention-to-treat population – the primary endpoint – was reached in four patients (17%) in the monotherapy arm and in seven patients (33%) in the combination arm. As noted, the combination arm reached the prespecified boundary of six or more patients with an MPR. All patients in each arm who had an MPR also had a pathologic complete responses.
Of the 39 patients who went on to resection, 37 were evaluable, and in these patients the respective MPR rates were 19% and 44%. Two patients on nivolumab alone and six on nivolumab plus ipilimumab had 0% viable tumor detectable at the time of surgery. Radiographic responses included one complete response in the combination arm and eight total partial responses, in five and three patients, respectively. The objective response rated was 20%. The responses, assessed by Response Evaluation Criteria in Solid Tumors (RECIST) were positively associated with MPR, Dr. Cascone said.
In 11% of patients, the investigators observed apparent radiographic progression after neoadjuvant immune checkpoint inhibitors in mediastinal and or in nonregional nodes. However, pathological assessment and evaluation of the flaring nodes did not reveal evidence of disease, but instead showed noncaseating granulomas that were not present at baseline.
“Awareness of this phenomenon, which we named the ‘nodal immune flare,’ is of critical importance, as if the clinician fails to distinguish the nodal immune flare from disease progression, potential curative surgery for these patients could be avoided,” she said.
Grade 1 or 2 treatment-related adverse events included rash, itching, fatigue, anemia, cough, and diarrhea. Grade 3 or greater treatment-related adverse events included hypoxia, pneumonia, and pneumonitis in the nivolumab monotherapy arm and diarrhea and hyponatremia in the combination group. One patient treated with nivolumab monotherapy, who had achieved 0% viable tumor, had grade 3 pneumonia and pneumonitis, which was treated with steroids that impeded the healing of a bronchopleural fistula and subsequent empyema. Other surgical complications included air leaks, which occurred in five patients in the nivolumab group and three in the nivolumab plus ipilimumab arm.
Two patients died, one in the monotherapy arm from steroid-treated pneumonitis 4.1 months after randomization and one in the combination arm who had progressive disease 2 months after randomization, and died from the disease 15 months later.
Invited discussant Maximilian Diehn, MD, PhD, from Stanford (California) University School of Medicine, commented that the choice of neoadjuvant immunotherapy was not based on molecular markers, “and I think we have a major unmet need for developing biomarkers for personalized treatment in this area.
“Ideally, the biomarkers that we would have in this setting would, A, allow us to identify which patients have micrometastatic disease and therefore are likely to benefit from the upfront systemic therapy and, secondly, also could tell us which neoadjuvant therapy they would respond to, be it immunotherapy, chemotherapy, or the combination,” he added.
The study was supported by Bristol-Myers Squibb. Dr. Cascone disclosed honoraria from the company. Dr. Diehn reported stock ownership, consulting, research funding and travel expenses from various companies.
SOURCE: Cascone T. et al. ASCO 2019, Abstract 8504.
REPORTING FROM ASCO 2019
Checkpoint inhibitor rechallenge is possible for select patients
Rechallenge resulted in the recurrence of a grade 2 or higher immune-related adverse event (irAE) in 55% of rechallenged patients, but no deaths occurred, according to Audrey Simonaggio, MD, of the department of drug development at Gustave Roussy, Villejuif, France, and colleagues.
In those rechallenged patients who had a second irAE, the second event was not more severe than the first. “The rechallenge should first be assessed in a multidisciplinary team meeting with regard to each patient’s individual risk-reward ratio. ... We recommend close monitoring,” the researchers wrote in a study published in JAMA Oncology.
As there are no specific recommendations to guide the decision to rechallenge, the usefulness of the rechallenge was considered. The readministration could be delayed if the patient was in complete or excellent partial response. The existence of other therapeutic alternatives was also important as was the patient’s clinical state. Rechallenge was considered possible only after the grade of the initial irAE returned to 0 or 1.
“Because of life-threatening risk, we did not support rechallenge for cardiac (myocarditis) and neurologic irAEs [such] as Guillain-Barré syndrome, encephalitis, and severe myositis,” they said. CT scans were used to guide the decision to rechallenge in those with initial lung adverse events.
The cohort study included 93 consecutive adult patients who were referred over an 18-month period to the ImmunoTOX assessment board at the Gustave Roussy cancer center and followed for at least 1 year. The cohort was balanced for gender and ranged in age from 33 to 85 years, with a median age of 62.5 years. Melanoma was the predominant tumor (33%), followed by lung (16%), colorectal (9%), and lymphoma (9%).
The initial immune-related adverse event was a grade 2 event in 46% of patients, grade 3 in 39%, and grade 4 in 15%. Events included hepatitis (18%), skin toxicity (15%), pneumonitis (14%), colitis (12%), and arthralgia (7.5%). A rechallenge with the same anti–PD-1 or anti–PD-L1 was conducted in 43% of patients.
When compared with patients who were not rechallenged, there was no difference in median patient age, time to initial immune-related adverse event (five vs. three treatment cycles), event severity, or steroid use. With a median follow-up period of 14 months, the same or a different immune-related adverse event occurred in 22 patients (55%). A shorter time to the initial event was linked to the occurrence of a second event (9 vs. 15 weeks; P = .04).
“However, we did observe a trend toward a higher recurrence rate after a more severe initial irAE and a trend toward more frequent recurrence in patients treated with corticosteroids after the initial irAE,” the researchers wrote. “An anti–PD-1or anti–PD-L1 rechallenge after a grade 4 irAE should always be considered with caution.” Three of the five patients with these events were being treated for lymphoma, they said.
“As long as patients are closely monitored, anti–PD-1 or anti–PD-L1 rechallenge appears to have an acceptable toxic effect profile. Myocarditis and neurologic toxic effect should remain a contraindication. Rechallenge conditions require further investigation in a prospective clinical trial. ... Well-powered, prospective studies with a larger number of patients would be required to generate information on putative risk factors for the recurrence of irAEs. Our results highlighted the value of a review board, like ImmunoTOX, with intention to build a large irAE database and then establish evidence-based guidelines on the safety of a rechallenge,” the researchers concluded.
The study was supported by the Gustave Roussy cancer center and the Gustave Roussy immunotherapy program. Dr. Simonaggio had no relevant disclosures; several coauthors reported consultancy fees and research support from multiple drug companies.
SOURCE: Simonaggio A et al. JAMA Oncol. 2019 Jun 6. doi:10.1001/jamaoncol.2019.1022.
Rechallenge resulted in the recurrence of a grade 2 or higher immune-related adverse event (irAE) in 55% of rechallenged patients, but no deaths occurred, according to Audrey Simonaggio, MD, of the department of drug development at Gustave Roussy, Villejuif, France, and colleagues.
In those rechallenged patients who had a second irAE, the second event was not more severe than the first. “The rechallenge should first be assessed in a multidisciplinary team meeting with regard to each patient’s individual risk-reward ratio. ... We recommend close monitoring,” the researchers wrote in a study published in JAMA Oncology.
As there are no specific recommendations to guide the decision to rechallenge, the usefulness of the rechallenge was considered. The readministration could be delayed if the patient was in complete or excellent partial response. The existence of other therapeutic alternatives was also important as was the patient’s clinical state. Rechallenge was considered possible only after the grade of the initial irAE returned to 0 or 1.
“Because of life-threatening risk, we did not support rechallenge for cardiac (myocarditis) and neurologic irAEs [such] as Guillain-Barré syndrome, encephalitis, and severe myositis,” they said. CT scans were used to guide the decision to rechallenge in those with initial lung adverse events.
The cohort study included 93 consecutive adult patients who were referred over an 18-month period to the ImmunoTOX assessment board at the Gustave Roussy cancer center and followed for at least 1 year. The cohort was balanced for gender and ranged in age from 33 to 85 years, with a median age of 62.5 years. Melanoma was the predominant tumor (33%), followed by lung (16%), colorectal (9%), and lymphoma (9%).
The initial immune-related adverse event was a grade 2 event in 46% of patients, grade 3 in 39%, and grade 4 in 15%. Events included hepatitis (18%), skin toxicity (15%), pneumonitis (14%), colitis (12%), and arthralgia (7.5%). A rechallenge with the same anti–PD-1 or anti–PD-L1 was conducted in 43% of patients.
When compared with patients who were not rechallenged, there was no difference in median patient age, time to initial immune-related adverse event (five vs. three treatment cycles), event severity, or steroid use. With a median follow-up period of 14 months, the same or a different immune-related adverse event occurred in 22 patients (55%). A shorter time to the initial event was linked to the occurrence of a second event (9 vs. 15 weeks; P = .04).
“However, we did observe a trend toward a higher recurrence rate after a more severe initial irAE and a trend toward more frequent recurrence in patients treated with corticosteroids after the initial irAE,” the researchers wrote. “An anti–PD-1or anti–PD-L1 rechallenge after a grade 4 irAE should always be considered with caution.” Three of the five patients with these events were being treated for lymphoma, they said.
“As long as patients are closely monitored, anti–PD-1 or anti–PD-L1 rechallenge appears to have an acceptable toxic effect profile. Myocarditis and neurologic toxic effect should remain a contraindication. Rechallenge conditions require further investigation in a prospective clinical trial. ... Well-powered, prospective studies with a larger number of patients would be required to generate information on putative risk factors for the recurrence of irAEs. Our results highlighted the value of a review board, like ImmunoTOX, with intention to build a large irAE database and then establish evidence-based guidelines on the safety of a rechallenge,” the researchers concluded.
The study was supported by the Gustave Roussy cancer center and the Gustave Roussy immunotherapy program. Dr. Simonaggio had no relevant disclosures; several coauthors reported consultancy fees and research support from multiple drug companies.
SOURCE: Simonaggio A et al. JAMA Oncol. 2019 Jun 6. doi:10.1001/jamaoncol.2019.1022.
Rechallenge resulted in the recurrence of a grade 2 or higher immune-related adverse event (irAE) in 55% of rechallenged patients, but no deaths occurred, according to Audrey Simonaggio, MD, of the department of drug development at Gustave Roussy, Villejuif, France, and colleagues.
In those rechallenged patients who had a second irAE, the second event was not more severe than the first. “The rechallenge should first be assessed in a multidisciplinary team meeting with regard to each patient’s individual risk-reward ratio. ... We recommend close monitoring,” the researchers wrote in a study published in JAMA Oncology.
As there are no specific recommendations to guide the decision to rechallenge, the usefulness of the rechallenge was considered. The readministration could be delayed if the patient was in complete or excellent partial response. The existence of other therapeutic alternatives was also important as was the patient’s clinical state. Rechallenge was considered possible only after the grade of the initial irAE returned to 0 or 1.
“Because of life-threatening risk, we did not support rechallenge for cardiac (myocarditis) and neurologic irAEs [such] as Guillain-Barré syndrome, encephalitis, and severe myositis,” they said. CT scans were used to guide the decision to rechallenge in those with initial lung adverse events.
The cohort study included 93 consecutive adult patients who were referred over an 18-month period to the ImmunoTOX assessment board at the Gustave Roussy cancer center and followed for at least 1 year. The cohort was balanced for gender and ranged in age from 33 to 85 years, with a median age of 62.5 years. Melanoma was the predominant tumor (33%), followed by lung (16%), colorectal (9%), and lymphoma (9%).
The initial immune-related adverse event was a grade 2 event in 46% of patients, grade 3 in 39%, and grade 4 in 15%. Events included hepatitis (18%), skin toxicity (15%), pneumonitis (14%), colitis (12%), and arthralgia (7.5%). A rechallenge with the same anti–PD-1 or anti–PD-L1 was conducted in 43% of patients.
When compared with patients who were not rechallenged, there was no difference in median patient age, time to initial immune-related adverse event (five vs. three treatment cycles), event severity, or steroid use. With a median follow-up period of 14 months, the same or a different immune-related adverse event occurred in 22 patients (55%). A shorter time to the initial event was linked to the occurrence of a second event (9 vs. 15 weeks; P = .04).
“However, we did observe a trend toward a higher recurrence rate after a more severe initial irAE and a trend toward more frequent recurrence in patients treated with corticosteroids after the initial irAE,” the researchers wrote. “An anti–PD-1or anti–PD-L1 rechallenge after a grade 4 irAE should always be considered with caution.” Three of the five patients with these events were being treated for lymphoma, they said.
“As long as patients are closely monitored, anti–PD-1 or anti–PD-L1 rechallenge appears to have an acceptable toxic effect profile. Myocarditis and neurologic toxic effect should remain a contraindication. Rechallenge conditions require further investigation in a prospective clinical trial. ... Well-powered, prospective studies with a larger number of patients would be required to generate information on putative risk factors for the recurrence of irAEs. Our results highlighted the value of a review board, like ImmunoTOX, with intention to build a large irAE database and then establish evidence-based guidelines on the safety of a rechallenge,” the researchers concluded.
The study was supported by the Gustave Roussy cancer center and the Gustave Roussy immunotherapy program. Dr. Simonaggio had no relevant disclosures; several coauthors reported consultancy fees and research support from multiple drug companies.
SOURCE: Simonaggio A et al. JAMA Oncol. 2019 Jun 6. doi:10.1001/jamaoncol.2019.1022.
FROM JAMA ONCOLOGY
Pembrolizumab improves 5-year OS in advanced NSCLC
CHICAGO – New data suggest pembrolizumab can increase 5-year overall survival (OS) for patients with advanced non–small cell lung cancer (NSCLC).
In the phase 1b KEYNOTE-001 trial, the 5-year OS rate was 23.2% in treatment-naive patients and 15.5% in previously treated patients. This is in comparison to the 5.5% average 5-year OS rate observed in NSCLC patients who receive standard chemotherapy (Noone AM et al. SEER Cancer Statistics Review, 1975-2015).
“In total, the data confirm that pembrolizumab has the potential to improve long-term outcomes for both treatment-naive and previously treated patients with advanced non–small cell lung cancer,” said Edward B. Garon, MD, of the University of California, Los Angeles.
Dr. Garon and colleagues presented these results in a poster at the annual meeting of the American Society for Clinical Oncology, and the data were simultaneously published in the Journal of Clinical Oncology.
KEYNOTE-001 (NCT01295827) enrolled 550 patients with advanced NSCLC who had received no prior therapy (n = 101) or at least one prior line of therapy (n = 449). Initially, patients received pembrolizumab at varying doses depending on body weight, but the protocol was changed to a single dose of pembrolizumab at 200 mg every 3 weeks.
At a median follow-up of 60.6 months, 100 patients were still alive. Sixty patients had received at least 2 years of pembrolizumab, 14 of whom were treatment-naive at baseline, and 46 of whom were previously treated at baseline.
Five-year OS rates were best among patients who had high PD-L1 expression, which was defined as 50% or greater.
Among treatment-naive patients, the 5-year OS rate was 29.6% in PD-L1–high patients and 15.7% in PD-L1–low patients (expression of 1% to 49%). The median OS was 35.4 months and 19.5 months, respectively.
Among previously treated patients, the 5-year OS rate was 25.0% in PD-L1–high patients, 12.6% in patients with PD-L1 expression of 1%-49%, and 3.5% in patients with PD-L1 expression less than 1%. The median OS was 15.4 months, 8.5 months, and 8.6 months, respectively.
Among patients who received at least 2 years of pembrolizumab, the 5-year OS rate was 78.6% in the treatment-naive group and 75.8% in the previously treated group. The objective response rate was 86% and 91%, respectively. The rate of ongoing response at the data cutoff was 58% and 71%, respectively.
“The safety data did not show any unanticipated late toxicity, which I consider encouraging,” Dr. Garon noted.
He said rates of immune-mediated adverse events were similar at 3 years and 5 years of follow-up. At 5 years, 17% of patients (n = 92) had experienced an immune-related adverse event, the most common of which were hypothyroidism (9%), pneumonitis (5%), and hyperthyroidism (2%).
Dr. Garon disclosed relationships with AstraZeneca, Bristol-Myers Squibb, Dracen, Dynavax, Genentech, Iovance Biotherapeutics, Lilly, Merck, Mirati Therapeutics, Neon Therapeutics, and Novartis. KEYNOTE-001 was sponsored by Merck Sharp & Dohme Corp.
SOURCES: Garon E. et al. ASCO 2019, Abstract LBA9015; J Clin Oncol. 2019 June 2. doi: 10.1200/JCO.19.00934
CHICAGO – New data suggest pembrolizumab can increase 5-year overall survival (OS) for patients with advanced non–small cell lung cancer (NSCLC).
In the phase 1b KEYNOTE-001 trial, the 5-year OS rate was 23.2% in treatment-naive patients and 15.5% in previously treated patients. This is in comparison to the 5.5% average 5-year OS rate observed in NSCLC patients who receive standard chemotherapy (Noone AM et al. SEER Cancer Statistics Review, 1975-2015).
“In total, the data confirm that pembrolizumab has the potential to improve long-term outcomes for both treatment-naive and previously treated patients with advanced non–small cell lung cancer,” said Edward B. Garon, MD, of the University of California, Los Angeles.
Dr. Garon and colleagues presented these results in a poster at the annual meeting of the American Society for Clinical Oncology, and the data were simultaneously published in the Journal of Clinical Oncology.
KEYNOTE-001 (NCT01295827) enrolled 550 patients with advanced NSCLC who had received no prior therapy (n = 101) or at least one prior line of therapy (n = 449). Initially, patients received pembrolizumab at varying doses depending on body weight, but the protocol was changed to a single dose of pembrolizumab at 200 mg every 3 weeks.
At a median follow-up of 60.6 months, 100 patients were still alive. Sixty patients had received at least 2 years of pembrolizumab, 14 of whom were treatment-naive at baseline, and 46 of whom were previously treated at baseline.
Five-year OS rates were best among patients who had high PD-L1 expression, which was defined as 50% or greater.
Among treatment-naive patients, the 5-year OS rate was 29.6% in PD-L1–high patients and 15.7% in PD-L1–low patients (expression of 1% to 49%). The median OS was 35.4 months and 19.5 months, respectively.
Among previously treated patients, the 5-year OS rate was 25.0% in PD-L1–high patients, 12.6% in patients with PD-L1 expression of 1%-49%, and 3.5% in patients with PD-L1 expression less than 1%. The median OS was 15.4 months, 8.5 months, and 8.6 months, respectively.
Among patients who received at least 2 years of pembrolizumab, the 5-year OS rate was 78.6% in the treatment-naive group and 75.8% in the previously treated group. The objective response rate was 86% and 91%, respectively. The rate of ongoing response at the data cutoff was 58% and 71%, respectively.
“The safety data did not show any unanticipated late toxicity, which I consider encouraging,” Dr. Garon noted.
He said rates of immune-mediated adverse events were similar at 3 years and 5 years of follow-up. At 5 years, 17% of patients (n = 92) had experienced an immune-related adverse event, the most common of which were hypothyroidism (9%), pneumonitis (5%), and hyperthyroidism (2%).
Dr. Garon disclosed relationships with AstraZeneca, Bristol-Myers Squibb, Dracen, Dynavax, Genentech, Iovance Biotherapeutics, Lilly, Merck, Mirati Therapeutics, Neon Therapeutics, and Novartis. KEYNOTE-001 was sponsored by Merck Sharp & Dohme Corp.
SOURCES: Garon E. et al. ASCO 2019, Abstract LBA9015; J Clin Oncol. 2019 June 2. doi: 10.1200/JCO.19.00934
CHICAGO – New data suggest pembrolizumab can increase 5-year overall survival (OS) for patients with advanced non–small cell lung cancer (NSCLC).
In the phase 1b KEYNOTE-001 trial, the 5-year OS rate was 23.2% in treatment-naive patients and 15.5% in previously treated patients. This is in comparison to the 5.5% average 5-year OS rate observed in NSCLC patients who receive standard chemotherapy (Noone AM et al. SEER Cancer Statistics Review, 1975-2015).
“In total, the data confirm that pembrolizumab has the potential to improve long-term outcomes for both treatment-naive and previously treated patients with advanced non–small cell lung cancer,” said Edward B. Garon, MD, of the University of California, Los Angeles.
Dr. Garon and colleagues presented these results in a poster at the annual meeting of the American Society for Clinical Oncology, and the data were simultaneously published in the Journal of Clinical Oncology.
KEYNOTE-001 (NCT01295827) enrolled 550 patients with advanced NSCLC who had received no prior therapy (n = 101) or at least one prior line of therapy (n = 449). Initially, patients received pembrolizumab at varying doses depending on body weight, but the protocol was changed to a single dose of pembrolizumab at 200 mg every 3 weeks.
At a median follow-up of 60.6 months, 100 patients were still alive. Sixty patients had received at least 2 years of pembrolizumab, 14 of whom were treatment-naive at baseline, and 46 of whom were previously treated at baseline.
Five-year OS rates were best among patients who had high PD-L1 expression, which was defined as 50% or greater.
Among treatment-naive patients, the 5-year OS rate was 29.6% in PD-L1–high patients and 15.7% in PD-L1–low patients (expression of 1% to 49%). The median OS was 35.4 months and 19.5 months, respectively.
Among previously treated patients, the 5-year OS rate was 25.0% in PD-L1–high patients, 12.6% in patients with PD-L1 expression of 1%-49%, and 3.5% in patients with PD-L1 expression less than 1%. The median OS was 15.4 months, 8.5 months, and 8.6 months, respectively.
Among patients who received at least 2 years of pembrolizumab, the 5-year OS rate was 78.6% in the treatment-naive group and 75.8% in the previously treated group. The objective response rate was 86% and 91%, respectively. The rate of ongoing response at the data cutoff was 58% and 71%, respectively.
“The safety data did not show any unanticipated late toxicity, which I consider encouraging,” Dr. Garon noted.
He said rates of immune-mediated adverse events were similar at 3 years and 5 years of follow-up. At 5 years, 17% of patients (n = 92) had experienced an immune-related adverse event, the most common of which were hypothyroidism (9%), pneumonitis (5%), and hyperthyroidism (2%).
Dr. Garon disclosed relationships with AstraZeneca, Bristol-Myers Squibb, Dracen, Dynavax, Genentech, Iovance Biotherapeutics, Lilly, Merck, Mirati Therapeutics, Neon Therapeutics, and Novartis. KEYNOTE-001 was sponsored by Merck Sharp & Dohme Corp.
SOURCES: Garon E. et al. ASCO 2019, Abstract LBA9015; J Clin Oncol. 2019 June 2. doi: 10.1200/JCO.19.00934
REPORTING FROM ASCO 2019
Pembro as good as chemo for gastric cancers with less toxicity
CHICAGO – In gastric and gastroesophageal junction (GEJ) cancers, positive for PD-L1, treatment with the PD-1 inhibitor pembrolizumab offered comparable survival with fewer side effects, according to results of a phase 3 randomized clinical trial.
The checkpoint inhibitor also demonstrated a clinically meaningful improvement in patients who had high levels of PD-L1 expression, with a 2-year survival rate of 39% versus 22% for patients receiving the standard chemotherapy, which consisted of a platinum and a fluoropyrimidine, according to Josep Tabernero, MD, PhD, lead author of the KEYNOTE-062 study.
By contrast, the study failed to demonstrate that pembrolizumab immunotherapy combined with that chemotherapy backbone was superior to chemotherapy alone on survival endpoints, said Dr. Tabernero, Head of the Medical Oncology Department at the Vall d’Hebron Barcelona University Hospital and Institute of Oncology, Spain.
“There are several factors we are evaluating,” Dr. Tabernero said here in a press conference at the annual meeting of the American Society of Clinical Oncology (ASCO). “We still have to do more studies to understand why, with this chemotherapy backbone, we don’t see a clear synergistic effect for superiority in overall survival.”
Nevertheless, these findings make pembrolizumab a “preferred treatment” for many patients, particularly in light of its “substantially improved safety profile” versus chemotherapy, said ASCO Senior Vice President and Chief Medical Officer Richard L. Schilsky, MD.
“What I take away from this study is that for patients with advanced gastric and gastroesophageal cancer, pembrolizumab should really in many cases replace chemotherapy as a first-line treatment for this population,” Dr. Schilsky said in a press conference. “It’s certainly not worse, and it may well be better.”
The KEYNOTE-062 study included 763 patients with HER2-negative, PD-L1-positive advanced gastric or GEJ cancers randomized to one of three arms: pembrolizumab alone for up to 35 cycles, pembrolizumab for up to 35 cycles plus chemotherapy, or placebo plus chemotherapy.
Pembrolizumab alone was not inferior compared to chemotherapy, with median overall survival rates of 10.5 and 11.1 months, respectively (HR, 0.91; 99.2% CI, 0.69-1.18), Dr. Tabernero reported.
Overall survival appeared to be prolonged in patients with high levels of PD-L1 expression, defined as a combined positive score (CPS) of 10 or greater. The median survival in that subgroup was 17.4 months for those receiving pembrolizumab, and just 10.8 months for chemotherapy. However, the design of the study precluded an analysis of statistical significance for this finding, according to Dr. Tabernero.
Looking at the overall study population, pembrolizumab plus chemotherapy did not improve survival versus chemotherapy alone, he added, reporting median overall survivals of 12.5 and 11.1 months, respectively (P = .046).
Subgroup analysis suggested that Asian patients derived particular benefit from pembrolizumab as compared to chemotherapy, though Dr. Tabernero cautioned against overinterpretation of the finding, saying that it could be due to biology, or could be a statistical anomaly.
Funding for the study came from Merck & Co., Inc. Dr. Tabernero reported disclosures related to Bayer, Boehringer Ingelheim, Lilly, MSD, Merck Serono, Novartis, Sanofi, and others.
SOURCE: Tabernero J, et al. ASCO 2019. Abstract LBA4007.
CHICAGO – In gastric and gastroesophageal junction (GEJ) cancers, positive for PD-L1, treatment with the PD-1 inhibitor pembrolizumab offered comparable survival with fewer side effects, according to results of a phase 3 randomized clinical trial.
The checkpoint inhibitor also demonstrated a clinically meaningful improvement in patients who had high levels of PD-L1 expression, with a 2-year survival rate of 39% versus 22% for patients receiving the standard chemotherapy, which consisted of a platinum and a fluoropyrimidine, according to Josep Tabernero, MD, PhD, lead author of the KEYNOTE-062 study.
By contrast, the study failed to demonstrate that pembrolizumab immunotherapy combined with that chemotherapy backbone was superior to chemotherapy alone on survival endpoints, said Dr. Tabernero, Head of the Medical Oncology Department at the Vall d’Hebron Barcelona University Hospital and Institute of Oncology, Spain.
“There are several factors we are evaluating,” Dr. Tabernero said here in a press conference at the annual meeting of the American Society of Clinical Oncology (ASCO). “We still have to do more studies to understand why, with this chemotherapy backbone, we don’t see a clear synergistic effect for superiority in overall survival.”
Nevertheless, these findings make pembrolizumab a “preferred treatment” for many patients, particularly in light of its “substantially improved safety profile” versus chemotherapy, said ASCO Senior Vice President and Chief Medical Officer Richard L. Schilsky, MD.
“What I take away from this study is that for patients with advanced gastric and gastroesophageal cancer, pembrolizumab should really in many cases replace chemotherapy as a first-line treatment for this population,” Dr. Schilsky said in a press conference. “It’s certainly not worse, and it may well be better.”
The KEYNOTE-062 study included 763 patients with HER2-negative, PD-L1-positive advanced gastric or GEJ cancers randomized to one of three arms: pembrolizumab alone for up to 35 cycles, pembrolizumab for up to 35 cycles plus chemotherapy, or placebo plus chemotherapy.
Pembrolizumab alone was not inferior compared to chemotherapy, with median overall survival rates of 10.5 and 11.1 months, respectively (HR, 0.91; 99.2% CI, 0.69-1.18), Dr. Tabernero reported.
Overall survival appeared to be prolonged in patients with high levels of PD-L1 expression, defined as a combined positive score (CPS) of 10 or greater. The median survival in that subgroup was 17.4 months for those receiving pembrolizumab, and just 10.8 months for chemotherapy. However, the design of the study precluded an analysis of statistical significance for this finding, according to Dr. Tabernero.
Looking at the overall study population, pembrolizumab plus chemotherapy did not improve survival versus chemotherapy alone, he added, reporting median overall survivals of 12.5 and 11.1 months, respectively (P = .046).
Subgroup analysis suggested that Asian patients derived particular benefit from pembrolizumab as compared to chemotherapy, though Dr. Tabernero cautioned against overinterpretation of the finding, saying that it could be due to biology, or could be a statistical anomaly.
Funding for the study came from Merck & Co., Inc. Dr. Tabernero reported disclosures related to Bayer, Boehringer Ingelheim, Lilly, MSD, Merck Serono, Novartis, Sanofi, and others.
SOURCE: Tabernero J, et al. ASCO 2019. Abstract LBA4007.
CHICAGO – In gastric and gastroesophageal junction (GEJ) cancers, positive for PD-L1, treatment with the PD-1 inhibitor pembrolizumab offered comparable survival with fewer side effects, according to results of a phase 3 randomized clinical trial.
The checkpoint inhibitor also demonstrated a clinically meaningful improvement in patients who had high levels of PD-L1 expression, with a 2-year survival rate of 39% versus 22% for patients receiving the standard chemotherapy, which consisted of a platinum and a fluoropyrimidine, according to Josep Tabernero, MD, PhD, lead author of the KEYNOTE-062 study.
By contrast, the study failed to demonstrate that pembrolizumab immunotherapy combined with that chemotherapy backbone was superior to chemotherapy alone on survival endpoints, said Dr. Tabernero, Head of the Medical Oncology Department at the Vall d’Hebron Barcelona University Hospital and Institute of Oncology, Spain.
“There are several factors we are evaluating,” Dr. Tabernero said here in a press conference at the annual meeting of the American Society of Clinical Oncology (ASCO). “We still have to do more studies to understand why, with this chemotherapy backbone, we don’t see a clear synergistic effect for superiority in overall survival.”
Nevertheless, these findings make pembrolizumab a “preferred treatment” for many patients, particularly in light of its “substantially improved safety profile” versus chemotherapy, said ASCO Senior Vice President and Chief Medical Officer Richard L. Schilsky, MD.
“What I take away from this study is that for patients with advanced gastric and gastroesophageal cancer, pembrolizumab should really in many cases replace chemotherapy as a first-line treatment for this population,” Dr. Schilsky said in a press conference. “It’s certainly not worse, and it may well be better.”
The KEYNOTE-062 study included 763 patients with HER2-negative, PD-L1-positive advanced gastric or GEJ cancers randomized to one of three arms: pembrolizumab alone for up to 35 cycles, pembrolizumab for up to 35 cycles plus chemotherapy, or placebo plus chemotherapy.
Pembrolizumab alone was not inferior compared to chemotherapy, with median overall survival rates of 10.5 and 11.1 months, respectively (HR, 0.91; 99.2% CI, 0.69-1.18), Dr. Tabernero reported.
Overall survival appeared to be prolonged in patients with high levels of PD-L1 expression, defined as a combined positive score (CPS) of 10 or greater. The median survival in that subgroup was 17.4 months for those receiving pembrolizumab, and just 10.8 months for chemotherapy. However, the design of the study precluded an analysis of statistical significance for this finding, according to Dr. Tabernero.
Looking at the overall study population, pembrolizumab plus chemotherapy did not improve survival versus chemotherapy alone, he added, reporting median overall survivals of 12.5 and 11.1 months, respectively (P = .046).
Subgroup analysis suggested that Asian patients derived particular benefit from pembrolizumab as compared to chemotherapy, though Dr. Tabernero cautioned against overinterpretation of the finding, saying that it could be due to biology, or could be a statistical anomaly.
Funding for the study came from Merck & Co., Inc. Dr. Tabernero reported disclosures related to Bayer, Boehringer Ingelheim, Lilly, MSD, Merck Serono, Novartis, Sanofi, and others.
SOURCE: Tabernero J, et al. ASCO 2019. Abstract LBA4007.
REPORTING FROM ASCO 2019
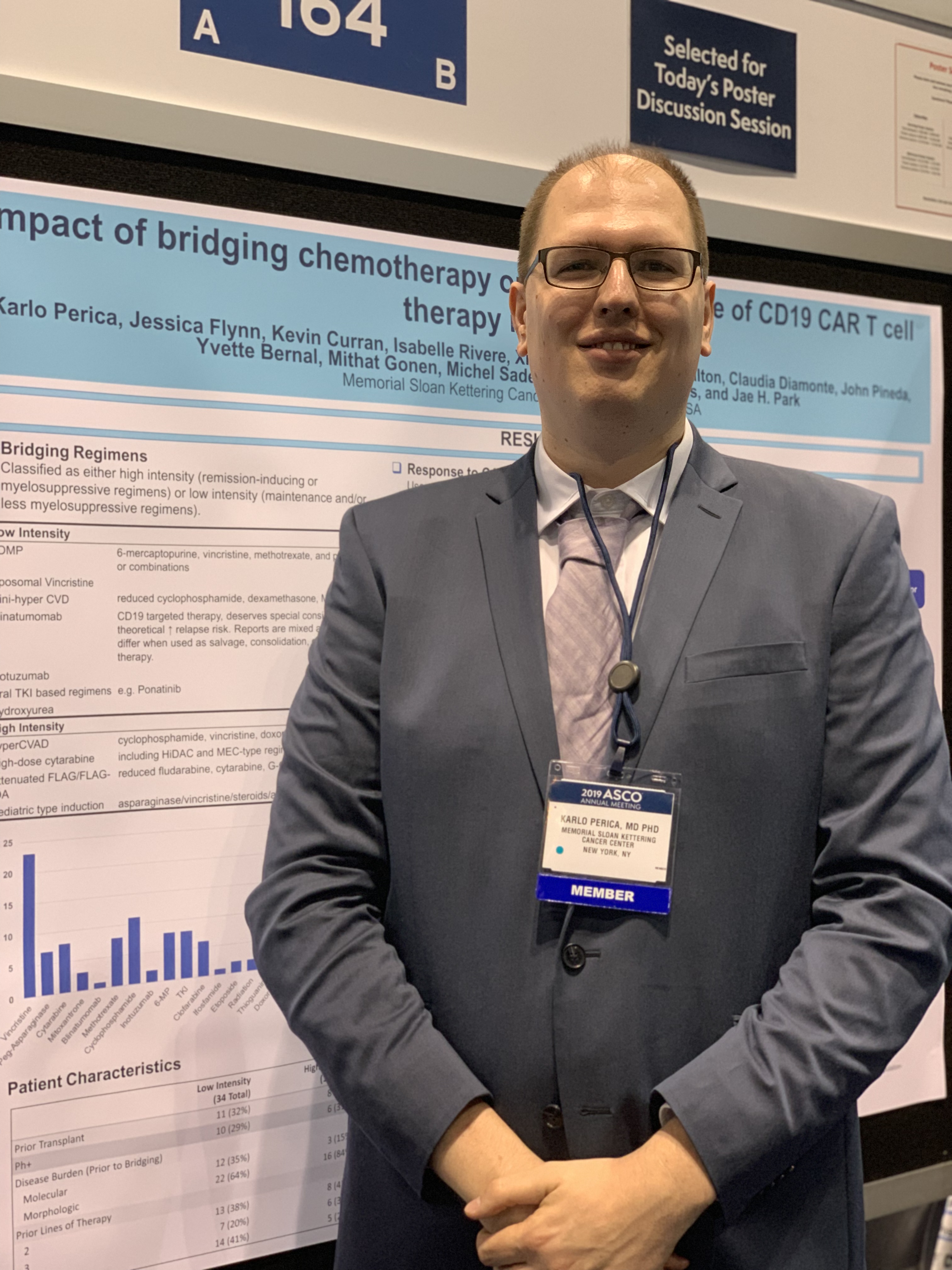
Low intensity bridging may be best path to CAR T in adult ALL
CHICAGO – A low intensity chemotherapy regimen may be the best approach to bridge patients waiting for chimeric antigen receptor (CAR) T-cell therapy, according to a retrospective analysis of adults with acute lymphoblastic leukemia (ALL).
Investigators found that high intensity bridging regimens provided no clear outcome benefit, but did produce a greater number of infections.
But the decision on the type of regimen is very much dependent on the individual patient, Karlo Perica, MD, PhD, of Memorial Sloan Kettering Cancer Center in New York, said at the annual meeting of the American Society of Clinical Oncology.
Dr. Perica and his colleagues at Memorial Sloan Kettering examined the effectiveness and toxicity of bridging therapies provided to relapsed or refractory ALL patients waiting to receive CD19 CAR T-cell therapy as part of a phase 1 trial (N Engl J Med. 2018 Feb 1;378[5]:449-59).
Bridging therapy was defined as any therapy given from leukapheresis to cell infusion.
The low-intensity regimens included POMP (6-mercaptopurine, vincristine, methotrexate, and prednisone, or combinations), liposomal vincristine, mini-hyper CVD (reduced cyclophosphamide, dexamethasone, methotrexate, Ara-C), blinatumomab, inotuzumab, oral tyrosine kinase inhibitor-based regimens, or hydroxyurea.
The high-intensity regimens included hyper-CVAD (cyclophosphamide, vincristine, doxorubicin, dexamethasone), high-dose cytarabine, attenuated FLAG/FLAG-IDA (reduced fludarabine, cytarabine, G-CSF plus or minus idarubicin), and pediatric-type induction.
Of the 53 patients who were ultimately infused with CAR T cells, 19 received some type of high intensity regimen, 29 received low intensity regimens, and 5 received no bridging treatment. The group overall was heavily pretreated. Nearly a third of the low intensity and no bridging patients and 42% of the high intensity patients had previously undergone transplant. More than 40% of the low intensity and no bridging patients and about a quarter of the high intensity bridging group had four or more prior lines of therapy.
The use of high intensity bridging therapy was not associated with improved overall response or relapse-free survival to CAR T-cell therapy, the investigators reported. In a subgroup with 23 high disease burden patients with greater than 20% blasts, there was no difference in MRD-negative complete response by intensity (75% versus 60%, Fisher’s P = .65).
High intensity bridging was also not associated with successful CAR T-cell infusion, versus low intensity regimens (63% versus 79%, P greater than .05) or a combined endpoint of CAR T-cell infusion plus transplant or alternative treatment (80% versus 86%, P greater than .05).
In terms of toxicity, the high intensity bridging regimens were associated with a higher rate of grade 3 or 4 infections – 15 versus 11 infections (Fisher’s P = .002). But there was no association with post-infusion grade 3 or 4 cytokine release syndrome or neurotoxicity.
Dr. Perica said the results reflect that the real goal of bridging is not to reduce disease burden but instead to successfully bring patients to the next phase of their treatment. “The goal of the bridging therapy is to get the patient to the CAR infusion,” he said.
Due to the retrospective nature of the study, Dr. Perica said he can’t recommend any single bridging regimen and he emphasized that the decisions are patient-specific.
The original study was funded by several foundations and Juno Therapeutics. Dr. Perica reported royalties from technology licensed to Neximmune.
SOURCE: Perica K et al. ASCO 2019, Abstract 2520.
CHICAGO – A low intensity chemotherapy regimen may be the best approach to bridge patients waiting for chimeric antigen receptor (CAR) T-cell therapy, according to a retrospective analysis of adults with acute lymphoblastic leukemia (ALL).
Investigators found that high intensity bridging regimens provided no clear outcome benefit, but did produce a greater number of infections.
But the decision on the type of regimen is very much dependent on the individual patient, Karlo Perica, MD, PhD, of Memorial Sloan Kettering Cancer Center in New York, said at the annual meeting of the American Society of Clinical Oncology.
Dr. Perica and his colleagues at Memorial Sloan Kettering examined the effectiveness and toxicity of bridging therapies provided to relapsed or refractory ALL patients waiting to receive CD19 CAR T-cell therapy as part of a phase 1 trial (N Engl J Med. 2018 Feb 1;378[5]:449-59).
Bridging therapy was defined as any therapy given from leukapheresis to cell infusion.
The low-intensity regimens included POMP (6-mercaptopurine, vincristine, methotrexate, and prednisone, or combinations), liposomal vincristine, mini-hyper CVD (reduced cyclophosphamide, dexamethasone, methotrexate, Ara-C), blinatumomab, inotuzumab, oral tyrosine kinase inhibitor-based regimens, or hydroxyurea.
The high-intensity regimens included hyper-CVAD (cyclophosphamide, vincristine, doxorubicin, dexamethasone), high-dose cytarabine, attenuated FLAG/FLAG-IDA (reduced fludarabine, cytarabine, G-CSF plus or minus idarubicin), and pediatric-type induction.
Of the 53 patients who were ultimately infused with CAR T cells, 19 received some type of high intensity regimen, 29 received low intensity regimens, and 5 received no bridging treatment. The group overall was heavily pretreated. Nearly a third of the low intensity and no bridging patients and 42% of the high intensity patients had previously undergone transplant. More than 40% of the low intensity and no bridging patients and about a quarter of the high intensity bridging group had four or more prior lines of therapy.
The use of high intensity bridging therapy was not associated with improved overall response or relapse-free survival to CAR T-cell therapy, the investigators reported. In a subgroup with 23 high disease burden patients with greater than 20% blasts, there was no difference in MRD-negative complete response by intensity (75% versus 60%, Fisher’s P = .65).
High intensity bridging was also not associated with successful CAR T-cell infusion, versus low intensity regimens (63% versus 79%, P greater than .05) or a combined endpoint of CAR T-cell infusion plus transplant or alternative treatment (80% versus 86%, P greater than .05).
In terms of toxicity, the high intensity bridging regimens were associated with a higher rate of grade 3 or 4 infections – 15 versus 11 infections (Fisher’s P = .002). But there was no association with post-infusion grade 3 or 4 cytokine release syndrome or neurotoxicity.
Dr. Perica said the results reflect that the real goal of bridging is not to reduce disease burden but instead to successfully bring patients to the next phase of their treatment. “The goal of the bridging therapy is to get the patient to the CAR infusion,” he said.
Due to the retrospective nature of the study, Dr. Perica said he can’t recommend any single bridging regimen and he emphasized that the decisions are patient-specific.
The original study was funded by several foundations and Juno Therapeutics. Dr. Perica reported royalties from technology licensed to Neximmune.
SOURCE: Perica K et al. ASCO 2019, Abstract 2520.
CHICAGO – A low intensity chemotherapy regimen may be the best approach to bridge patients waiting for chimeric antigen receptor (CAR) T-cell therapy, according to a retrospective analysis of adults with acute lymphoblastic leukemia (ALL).
Investigators found that high intensity bridging regimens provided no clear outcome benefit, but did produce a greater number of infections.
But the decision on the type of regimen is very much dependent on the individual patient, Karlo Perica, MD, PhD, of Memorial Sloan Kettering Cancer Center in New York, said at the annual meeting of the American Society of Clinical Oncology.
Dr. Perica and his colleagues at Memorial Sloan Kettering examined the effectiveness and toxicity of bridging therapies provided to relapsed or refractory ALL patients waiting to receive CD19 CAR T-cell therapy as part of a phase 1 trial (N Engl J Med. 2018 Feb 1;378[5]:449-59).
Bridging therapy was defined as any therapy given from leukapheresis to cell infusion.
The low-intensity regimens included POMP (6-mercaptopurine, vincristine, methotrexate, and prednisone, or combinations), liposomal vincristine, mini-hyper CVD (reduced cyclophosphamide, dexamethasone, methotrexate, Ara-C), blinatumomab, inotuzumab, oral tyrosine kinase inhibitor-based regimens, or hydroxyurea.
The high-intensity regimens included hyper-CVAD (cyclophosphamide, vincristine, doxorubicin, dexamethasone), high-dose cytarabine, attenuated FLAG/FLAG-IDA (reduced fludarabine, cytarabine, G-CSF plus or minus idarubicin), and pediatric-type induction.
Of the 53 patients who were ultimately infused with CAR T cells, 19 received some type of high intensity regimen, 29 received low intensity regimens, and 5 received no bridging treatment. The group overall was heavily pretreated. Nearly a third of the low intensity and no bridging patients and 42% of the high intensity patients had previously undergone transplant. More than 40% of the low intensity and no bridging patients and about a quarter of the high intensity bridging group had four or more prior lines of therapy.
The use of high intensity bridging therapy was not associated with improved overall response or relapse-free survival to CAR T-cell therapy, the investigators reported. In a subgroup with 23 high disease burden patients with greater than 20% blasts, there was no difference in MRD-negative complete response by intensity (75% versus 60%, Fisher’s P = .65).
High intensity bridging was also not associated with successful CAR T-cell infusion, versus low intensity regimens (63% versus 79%, P greater than .05) or a combined endpoint of CAR T-cell infusion plus transplant or alternative treatment (80% versus 86%, P greater than .05).
In terms of toxicity, the high intensity bridging regimens were associated with a higher rate of grade 3 or 4 infections – 15 versus 11 infections (Fisher’s P = .002). But there was no association with post-infusion grade 3 or 4 cytokine release syndrome or neurotoxicity.
Dr. Perica said the results reflect that the real goal of bridging is not to reduce disease burden but instead to successfully bring patients to the next phase of their treatment. “The goal of the bridging therapy is to get the patient to the CAR infusion,” he said.
Due to the retrospective nature of the study, Dr. Perica said he can’t recommend any single bridging regimen and he emphasized that the decisions are patient-specific.
The original study was funded by several foundations and Juno Therapeutics. Dr. Perica reported royalties from technology licensed to Neximmune.
SOURCE: Perica K et al. ASCO 2019, Abstract 2520.
FROM ASCO 2019
Pembro with or without chemo superior to EXTREME for advanced HNSCC
CHICAGO – Pembrolizumab with and without chemotherapy proved superior for overall survival compared with the EXTREME regimen when used first line in certain subgroups of patients with recurrent/metastatic head and neck squamous cell carcinoma (R/M HNSCC), according to “practice-changing” final results from the randomized phase 3 KEYNOTE-048 study.
Compared with 300 patients randomized to receive the EXTREME regimen (a certuximab loading dose followed by carboplatin or cisplatin and 5-fluorouracil), 281 who received pembrolizumab plus chemotherapy (P+C) had superior overall survival (OS) with comparable safety–including both those with programmed death-Ligand 1 (PD-L1) combined positive score (CPS) of 20 or greater (median 14.7 vs 11.0 months; hazard ratio, 0.60) and with CPS of 1 or greater (median, 13.6 vs. 10.4 months; HR, 0.65), Danny Rischin, MD, reported at the annual meeting of the American Society of Clinical Oncology.
The differences were highly statistically significant, said Dr. Rischin, a professor and director of the Division of Cancer Medicine and head of the Department of Medical Oncology at Peter MacCallum Cancer Centre, Melbourne, Australia.
“And this benefit in overall survival in CPS greater than or equal to 20 and greater than or equal to 1 appeared to be present across all the subgroups that we looked at,” he added.
The response rates did not differ between P+C and EXTREME groups, but the median duration of response was significantly greater with P+C vs. EXTREME in both the CPS of 20 or greater and 1 or greater (7.1 vs. 4.2 months and 6.7 vs. 4.3 months, respectively).
Additionally, the final results of the study showed an OS benefit with P+C vs. EXTREME in the total population (13.0 vs. 10.7 months; HR, 0.72), Dr. Rischin said.
The difference between the groups with respect to progression-free survival (PFS), however, was not statistically significant and did not reach the superiority threshold, he noted.
In the 301 patients who received pembrolizumab alone, OS was superior in the CPS 20 or greater and 1 or greater populations (median, 14.8 vs. 10.7 months; HR, 0.58 and 12.4 vs. 10.3 months; HR, 0.74, respectively), compared with EXTREME, but was noninferior in the total population (median 11.5 vs. 10.7 months; HR, 0.83), and safety was favorable .
Again, PFS did not differ between the groups (median, 2.3 vs. 5.2 months; HR, 1.34), and while the overall response rates did not differ significantly, the median duration of response was substantially longer with pembrolizumab at 22.6 vs. 4.5 months with EXTREME, he said.
Study participants had locally incurable R/M HNSCC and no prior systemic therapy in the R/M setting. Those in he P+C arm received pembrolizumab at 200 mg plus 6 cycles of cisplatin at 100 mg/m2 or carboplatin AUC 5, and 5-fluorouracil at a dose of 1000 mg/m2/day for 4 days every 3 weeks; those in the pembrolizumab alone arm received 200 mg every 3 weeks for up to 35 cycles, and those in the EXTREME arm received certuximab at a 400 mg/m2 loading dose followed by 250 mg/m2 weekly with carboplatin AUC 5 or cisplatin at 100 mg/m2, and 5-FU at 1000 mg/m2/day for 4 days for 6 cycles.
“The data from KEYNOTE-048 support pembrolizumab plus platinum-based chemotherapy and pembrolizumab monotherapy as new standard of care monotherapies for recurrent/metastatic head and neck squamous cell carcinoma,” he concluded.
Discussant Vanita Noronha, MD, a professor in the Department of Medical Oncology at Tata Memorial Hospital in Mumbai, India, said that while the findings are practice changing, they also raise a number of questions, such as which patients should get pembrolizumab and which should get P+C, why there is a differential effect of pembrolizumab based on PD-L1 by CPS–and what about those with CPS of 0 or 1-20, and why the response rates and PFS rates were not improved in the pembrolizumab groups.
Other important questions include whether there are predictive biomarkers for response, and whether sequential therapy would be of benefit, she added.
While these and other questions remain to be addressed, the KEYNOTE-048 findings have implications for practice going forward; based on the current data, her approach to treating patients with R/M HNSCC not amenable to radical therapy is to treat with pembrolizumab alone in those with disease-free interval of 6 months or less, she said.
For those with disease-free interval greater than 6 months and good performance status who have controlled comorbidities, are platinum eligible, and for whom the treatment is reimbursable/affordable, treatment depends on symptom severity; she would treat those with mild/moderate symptoms and CPS of 20 or greater with pembrolizumab alone, those with CPS of 1 or greater with P+C or pembrolizumab alone, and those with CPS of 0 or unknown CPS with EXTREME or a similar regimen or with P+C, and she would treat those with severe symptoms with P+C.
“If the patient were a bit borderline, had multiple comorbidities, could not receive platinum, or had financial constraints, I would treat the patient with singe-agent intravenous chemotherapy or with oral metronomic chemotherapy, single-agent targeted therapy or with best supportive care,” she said.
Dr. Rischin has received research funding from Amgen, Bristol-Myers Squibb, Genentech/Roche, GSK, Merck, and Regeneron. Dr. Noronha has received research funding (to her institution) from Amgen,and Sanofi Aventis.
SOURCE: D Rischin et al., ASCO 2019: Abstract 6000.
CHICAGO – Pembrolizumab with and without chemotherapy proved superior for overall survival compared with the EXTREME regimen when used first line in certain subgroups of patients with recurrent/metastatic head and neck squamous cell carcinoma (R/M HNSCC), according to “practice-changing” final results from the randomized phase 3 KEYNOTE-048 study.
Compared with 300 patients randomized to receive the EXTREME regimen (a certuximab loading dose followed by carboplatin or cisplatin and 5-fluorouracil), 281 who received pembrolizumab plus chemotherapy (P+C) had superior overall survival (OS) with comparable safety–including both those with programmed death-Ligand 1 (PD-L1) combined positive score (CPS) of 20 or greater (median 14.7 vs 11.0 months; hazard ratio, 0.60) and with CPS of 1 or greater (median, 13.6 vs. 10.4 months; HR, 0.65), Danny Rischin, MD, reported at the annual meeting of the American Society of Clinical Oncology.
The differences were highly statistically significant, said Dr. Rischin, a professor and director of the Division of Cancer Medicine and head of the Department of Medical Oncology at Peter MacCallum Cancer Centre, Melbourne, Australia.
“And this benefit in overall survival in CPS greater than or equal to 20 and greater than or equal to 1 appeared to be present across all the subgroups that we looked at,” he added.
The response rates did not differ between P+C and EXTREME groups, but the median duration of response was significantly greater with P+C vs. EXTREME in both the CPS of 20 or greater and 1 or greater (7.1 vs. 4.2 months and 6.7 vs. 4.3 months, respectively).
Additionally, the final results of the study showed an OS benefit with P+C vs. EXTREME in the total population (13.0 vs. 10.7 months; HR, 0.72), Dr. Rischin said.
The difference between the groups with respect to progression-free survival (PFS), however, was not statistically significant and did not reach the superiority threshold, he noted.
In the 301 patients who received pembrolizumab alone, OS was superior in the CPS 20 or greater and 1 or greater populations (median, 14.8 vs. 10.7 months; HR, 0.58 and 12.4 vs. 10.3 months; HR, 0.74, respectively), compared with EXTREME, but was noninferior in the total population (median 11.5 vs. 10.7 months; HR, 0.83), and safety was favorable .
Again, PFS did not differ between the groups (median, 2.3 vs. 5.2 months; HR, 1.34), and while the overall response rates did not differ significantly, the median duration of response was substantially longer with pembrolizumab at 22.6 vs. 4.5 months with EXTREME, he said.
Study participants had locally incurable R/M HNSCC and no prior systemic therapy in the R/M setting. Those in he P+C arm received pembrolizumab at 200 mg plus 6 cycles of cisplatin at 100 mg/m2 or carboplatin AUC 5, and 5-fluorouracil at a dose of 1000 mg/m2/day for 4 days every 3 weeks; those in the pembrolizumab alone arm received 200 mg every 3 weeks for up to 35 cycles, and those in the EXTREME arm received certuximab at a 400 mg/m2 loading dose followed by 250 mg/m2 weekly with carboplatin AUC 5 or cisplatin at 100 mg/m2, and 5-FU at 1000 mg/m2/day for 4 days for 6 cycles.
“The data from KEYNOTE-048 support pembrolizumab plus platinum-based chemotherapy and pembrolizumab monotherapy as new standard of care monotherapies for recurrent/metastatic head and neck squamous cell carcinoma,” he concluded.
Discussant Vanita Noronha, MD, a professor in the Department of Medical Oncology at Tata Memorial Hospital in Mumbai, India, said that while the findings are practice changing, they also raise a number of questions, such as which patients should get pembrolizumab and which should get P+C, why there is a differential effect of pembrolizumab based on PD-L1 by CPS–and what about those with CPS of 0 or 1-20, and why the response rates and PFS rates were not improved in the pembrolizumab groups.
Other important questions include whether there are predictive biomarkers for response, and whether sequential therapy would be of benefit, she added.
While these and other questions remain to be addressed, the KEYNOTE-048 findings have implications for practice going forward; based on the current data, her approach to treating patients with R/M HNSCC not amenable to radical therapy is to treat with pembrolizumab alone in those with disease-free interval of 6 months or less, she said.
For those with disease-free interval greater than 6 months and good performance status who have controlled comorbidities, are platinum eligible, and for whom the treatment is reimbursable/affordable, treatment depends on symptom severity; she would treat those with mild/moderate symptoms and CPS of 20 or greater with pembrolizumab alone, those with CPS of 1 or greater with P+C or pembrolizumab alone, and those with CPS of 0 or unknown CPS with EXTREME or a similar regimen or with P+C, and she would treat those with severe symptoms with P+C.
“If the patient were a bit borderline, had multiple comorbidities, could not receive platinum, or had financial constraints, I would treat the patient with singe-agent intravenous chemotherapy or with oral metronomic chemotherapy, single-agent targeted therapy or with best supportive care,” she said.
Dr. Rischin has received research funding from Amgen, Bristol-Myers Squibb, Genentech/Roche, GSK, Merck, and Regeneron. Dr. Noronha has received research funding (to her institution) from Amgen,and Sanofi Aventis.
SOURCE: D Rischin et al., ASCO 2019: Abstract 6000.
CHICAGO – Pembrolizumab with and without chemotherapy proved superior for overall survival compared with the EXTREME regimen when used first line in certain subgroups of patients with recurrent/metastatic head and neck squamous cell carcinoma (R/M HNSCC), according to “practice-changing” final results from the randomized phase 3 KEYNOTE-048 study.
Compared with 300 patients randomized to receive the EXTREME regimen (a certuximab loading dose followed by carboplatin or cisplatin and 5-fluorouracil), 281 who received pembrolizumab plus chemotherapy (P+C) had superior overall survival (OS) with comparable safety–including both those with programmed death-Ligand 1 (PD-L1) combined positive score (CPS) of 20 or greater (median 14.7 vs 11.0 months; hazard ratio, 0.60) and with CPS of 1 or greater (median, 13.6 vs. 10.4 months; HR, 0.65), Danny Rischin, MD, reported at the annual meeting of the American Society of Clinical Oncology.
The differences were highly statistically significant, said Dr. Rischin, a professor and director of the Division of Cancer Medicine and head of the Department of Medical Oncology at Peter MacCallum Cancer Centre, Melbourne, Australia.
“And this benefit in overall survival in CPS greater than or equal to 20 and greater than or equal to 1 appeared to be present across all the subgroups that we looked at,” he added.
The response rates did not differ between P+C and EXTREME groups, but the median duration of response was significantly greater with P+C vs. EXTREME in both the CPS of 20 or greater and 1 or greater (7.1 vs. 4.2 months and 6.7 vs. 4.3 months, respectively).
Additionally, the final results of the study showed an OS benefit with P+C vs. EXTREME in the total population (13.0 vs. 10.7 months; HR, 0.72), Dr. Rischin said.
The difference between the groups with respect to progression-free survival (PFS), however, was not statistically significant and did not reach the superiority threshold, he noted.
In the 301 patients who received pembrolizumab alone, OS was superior in the CPS 20 or greater and 1 or greater populations (median, 14.8 vs. 10.7 months; HR, 0.58 and 12.4 vs. 10.3 months; HR, 0.74, respectively), compared with EXTREME, but was noninferior in the total population (median 11.5 vs. 10.7 months; HR, 0.83), and safety was favorable .
Again, PFS did not differ between the groups (median, 2.3 vs. 5.2 months; HR, 1.34), and while the overall response rates did not differ significantly, the median duration of response was substantially longer with pembrolizumab at 22.6 vs. 4.5 months with EXTREME, he said.
Study participants had locally incurable R/M HNSCC and no prior systemic therapy in the R/M setting. Those in he P+C arm received pembrolizumab at 200 mg plus 6 cycles of cisplatin at 100 mg/m2 or carboplatin AUC 5, and 5-fluorouracil at a dose of 1000 mg/m2/day for 4 days every 3 weeks; those in the pembrolizumab alone arm received 200 mg every 3 weeks for up to 35 cycles, and those in the EXTREME arm received certuximab at a 400 mg/m2 loading dose followed by 250 mg/m2 weekly with carboplatin AUC 5 or cisplatin at 100 mg/m2, and 5-FU at 1000 mg/m2/day for 4 days for 6 cycles.
“The data from KEYNOTE-048 support pembrolizumab plus platinum-based chemotherapy and pembrolizumab monotherapy as new standard of care monotherapies for recurrent/metastatic head and neck squamous cell carcinoma,” he concluded.
Discussant Vanita Noronha, MD, a professor in the Department of Medical Oncology at Tata Memorial Hospital in Mumbai, India, said that while the findings are practice changing, they also raise a number of questions, such as which patients should get pembrolizumab and which should get P+C, why there is a differential effect of pembrolizumab based on PD-L1 by CPS–and what about those with CPS of 0 or 1-20, and why the response rates and PFS rates were not improved in the pembrolizumab groups.
Other important questions include whether there are predictive biomarkers for response, and whether sequential therapy would be of benefit, she added.
While these and other questions remain to be addressed, the KEYNOTE-048 findings have implications for practice going forward; based on the current data, her approach to treating patients with R/M HNSCC not amenable to radical therapy is to treat with pembrolizumab alone in those with disease-free interval of 6 months or less, she said.
For those with disease-free interval greater than 6 months and good performance status who have controlled comorbidities, are platinum eligible, and for whom the treatment is reimbursable/affordable, treatment depends on symptom severity; she would treat those with mild/moderate symptoms and CPS of 20 or greater with pembrolizumab alone, those with CPS of 1 or greater with P+C or pembrolizumab alone, and those with CPS of 0 or unknown CPS with EXTREME or a similar regimen or with P+C, and she would treat those with severe symptoms with P+C.
“If the patient were a bit borderline, had multiple comorbidities, could not receive platinum, or had financial constraints, I would treat the patient with singe-agent intravenous chemotherapy or with oral metronomic chemotherapy, single-agent targeted therapy or with best supportive care,” she said.
Dr. Rischin has received research funding from Amgen, Bristol-Myers Squibb, Genentech/Roche, GSK, Merck, and Regeneron. Dr. Noronha has received research funding (to her institution) from Amgen,and Sanofi Aventis.
SOURCE: D Rischin et al., ASCO 2019: Abstract 6000.
REPORTING FROM ASCO 2019
Novel anti-PD-1 antibody can be given subcutaneously
based on results from an ongoing phase 1 trial.
“We assessed feasibility of monthly subcutaneous administration of PF-06801591, a humanized immunoglobulin G4 monoclonal antibody that binds to the programmed cell death [PD-1] receptor,” Melissa L. Johnson, MD, of the Sarah Cannon Research Institute, Nashville, Tenn., and colleagues wrote in JAMA Oncology. “Subcutaneous administration of an anti-PD-1 antibody in patients with advanced solid tumors appears to be feasible.”
The researchers evaluated the efficacy, safety, and pharmacokinetics of the novel anti-PD-1 therapy in a group of 40 patients with locally advanced or metastatic solid tumors. The antibody was administered in both subcutaneous and intravenous forms.
Study participants were administered the subcutaneous form of the antibody at 300 mg every 4 weeks or the intravenous form at 0.5, 1, 3, or 10 mg/kg every 3 weeks.
“Dose escalation occurred after two to four patients were enrolled per dose level, with additional patients enrolled in each cohort for further assessment,” the researchers wrote.
The primary endpoints were safety and dose-limiting adverse effects. Efficacy, immunogenicity, pharmacokinetics, and PD-1 receptor occupancy were secondary endpoints.
After analysis, Dr. Johnson and colleagues reported that the overall response rate was 18.4%. In addition, no dose-limiting toxicities were detected in both intravenous and subcutaneous groups, but grade 3 or greater adverse events were reported in 6.7% and 16% of patients treated with subcutaneous and intravenous dosage forms, respectively.
“No dose–adverse event associations were observed during intravenous dose escalation, and no serious skin toxic effects occurred with subcutaneous delivery,” they reported.
Based on the findings, the team selected the subcutaneous form (300 mg) of the therapy for additional assessment in the second half of this ongoing trial.
The study was funded by Pfizer. The authors reported financial affiliations with BerGenBio, EMD Serono, Janssen, Mirati Therapeutics, Pfizer, and several others.
SOURCE: Johnson ML et al. JAMA Oncol. 2019 May 30. doi: 10.1001/jamaoncol.2019.0836.
based on results from an ongoing phase 1 trial.
“We assessed feasibility of monthly subcutaneous administration of PF-06801591, a humanized immunoglobulin G4 monoclonal antibody that binds to the programmed cell death [PD-1] receptor,” Melissa L. Johnson, MD, of the Sarah Cannon Research Institute, Nashville, Tenn., and colleagues wrote in JAMA Oncology. “Subcutaneous administration of an anti-PD-1 antibody in patients with advanced solid tumors appears to be feasible.”
The researchers evaluated the efficacy, safety, and pharmacokinetics of the novel anti-PD-1 therapy in a group of 40 patients with locally advanced or metastatic solid tumors. The antibody was administered in both subcutaneous and intravenous forms.
Study participants were administered the subcutaneous form of the antibody at 300 mg every 4 weeks or the intravenous form at 0.5, 1, 3, or 10 mg/kg every 3 weeks.
“Dose escalation occurred after two to four patients were enrolled per dose level, with additional patients enrolled in each cohort for further assessment,” the researchers wrote.
The primary endpoints were safety and dose-limiting adverse effects. Efficacy, immunogenicity, pharmacokinetics, and PD-1 receptor occupancy were secondary endpoints.
After analysis, Dr. Johnson and colleagues reported that the overall response rate was 18.4%. In addition, no dose-limiting toxicities were detected in both intravenous and subcutaneous groups, but grade 3 or greater adverse events were reported in 6.7% and 16% of patients treated with subcutaneous and intravenous dosage forms, respectively.
“No dose–adverse event associations were observed during intravenous dose escalation, and no serious skin toxic effects occurred with subcutaneous delivery,” they reported.
Based on the findings, the team selected the subcutaneous form (300 mg) of the therapy for additional assessment in the second half of this ongoing trial.
The study was funded by Pfizer. The authors reported financial affiliations with BerGenBio, EMD Serono, Janssen, Mirati Therapeutics, Pfizer, and several others.
SOURCE: Johnson ML et al. JAMA Oncol. 2019 May 30. doi: 10.1001/jamaoncol.2019.0836.
based on results from an ongoing phase 1 trial.
“We assessed feasibility of monthly subcutaneous administration of PF-06801591, a humanized immunoglobulin G4 monoclonal antibody that binds to the programmed cell death [PD-1] receptor,” Melissa L. Johnson, MD, of the Sarah Cannon Research Institute, Nashville, Tenn., and colleagues wrote in JAMA Oncology. “Subcutaneous administration of an anti-PD-1 antibody in patients with advanced solid tumors appears to be feasible.”
The researchers evaluated the efficacy, safety, and pharmacokinetics of the novel anti-PD-1 therapy in a group of 40 patients with locally advanced or metastatic solid tumors. The antibody was administered in both subcutaneous and intravenous forms.
Study participants were administered the subcutaneous form of the antibody at 300 mg every 4 weeks or the intravenous form at 0.5, 1, 3, or 10 mg/kg every 3 weeks.
“Dose escalation occurred after two to four patients were enrolled per dose level, with additional patients enrolled in each cohort for further assessment,” the researchers wrote.
The primary endpoints were safety and dose-limiting adverse effects. Efficacy, immunogenicity, pharmacokinetics, and PD-1 receptor occupancy were secondary endpoints.
After analysis, Dr. Johnson and colleagues reported that the overall response rate was 18.4%. In addition, no dose-limiting toxicities were detected in both intravenous and subcutaneous groups, but grade 3 or greater adverse events were reported in 6.7% and 16% of patients treated with subcutaneous and intravenous dosage forms, respectively.
“No dose–adverse event associations were observed during intravenous dose escalation, and no serious skin toxic effects occurred with subcutaneous delivery,” they reported.
Based on the findings, the team selected the subcutaneous form (300 mg) of the therapy for additional assessment in the second half of this ongoing trial.
The study was funded by Pfizer. The authors reported financial affiliations with BerGenBio, EMD Serono, Janssen, Mirati Therapeutics, Pfizer, and several others.
SOURCE: Johnson ML et al. JAMA Oncol. 2019 May 30. doi: 10.1001/jamaoncol.2019.0836.
FROM JAMA ONCOLOGY
Do some EGFR mutation subtypes benefit from immune checkpoint blockade?
Although epidermal growth factor receptor–mutant lung cancers generally don’t benefit from checkpoint inhibitors, there may be some subtypes that do respond, results of a large, multi-institution analysis suggest.
Compared with wild-type lung cancer cases, tumors with epidermal growth factor receptor (EGFR) exon 19 deletion did indeed have worse outcomes after progressive death-1/progressive death–ligand 1 (PD-1/PD-L1) blockade; however, tumors harboring EGFR L858R mutations had comparable response rates and overall survival.
These findings don’t change clinical practice, but do serve as a “foundation” for more research into which patients with EGFR-mutant disease might benefit from immunotherapy, according to Katherine Hastings, PhD, of Yale University, New Haven, Conn., and associates.
“We unequivocally support the guidance that EGFR TKIs should be the preferred first-line treatment option for patients with EGFR-mutant lung cancer,” Dr. Hastings and coauthors wrote in Annals of Oncology.
To date, clinical investigations of immune checkpoint inhibitors in patients with EGFR-mutant lung cancers have largely been discouraging, the authors wrote. However, there have been some exceptions, including a phase 2 study where third-line durvalumab showed activity in some patients with EGFR-positive, PD-L1-expressing advanced non–small cell lung cancer (NSCLC).
Accordingly, the investigators sought to determine whether specific molecular features made a difference. They looked at a total of 171 EGFR-mutant lung cancers that had been treated with PD-1/PD-L1 inhibitors, alone or in combination with a cytotoxic T-lymphocyte antigen 4 inhibitor.
In their analysis, they drilled down on the most common EGFR tyrosine kinase inhibitor–sensitizing alleles: EGFR exon 19 deletions, which represented 76 cases, and EGFR L858R, which represented 44 cases. For comparison, they looked at a cohort of 212 patients with EGFR wild-type NSCLC who had also been treated with immune checkpoint inhibitors.
EGFR exon 19 deletion cases had a significantly lower overall response rate versus EGFR wild-type tumors, at 7% versus 22%, respectively (P = .002), the investigators found. Likewise, overall survival was significantly reduced, with a hazard ratio of 0.69 (95% confidence interval, 0.493-0.965; P = .03).
By contrast, EGFR L858R tumors had similar response rates versus EGFR wild type, at 16% and 22%, respectively (P = .42); overall survival also was similar, with a hazard ratio of 0.917 (95% CI, 0.597-1.409; P = .69).
Of note, progression-free survival was reduced in both EGFR L858R and EGFR exon 19 deletion cases as compared with EGFR wild-type cases, though the investigators wrote that discrepancy might be related to variations in scanning intervals between different institutions that contributed cases to the study.
“Overall, these data suggest that patients with EGFR exon 19–mutant tumors, in particular, have a significantly reduced benefit upon treatment with immune checkpoint inhibitors,” the authors noted.
In a separate but related analysis of 383 patients with EGFR-mutant lung cancer, tumor mutation burden was lower in EGFR exon 19 deletion cases as compared with the EGFR L858R cases. That finding lined up with the immunotherapy response data, the investigators wrote, though it’s unclear what might be driving the differences in tumor burden between these two groups.
There were no differences in response to immune checkpoint blockade based on whether or not tumors harbored the EGFR T790M mutation, the investigators added. Similarly, PD-L1 expression did not impact response.
The study authors reported disclosures related to Astellas Pharma, AstraZeneca, Bristol-Myers Squibb, Calithera Biosciences, Daiichi, Eli Lilly, Merck, Mirati Therapeutics, Novartis, Pfizer, Roche, and Takeda, among others.
SOURCE: Hastings K et al. Ann Oncol. 2019 May 14. doi: 10.1093/annonc/mdz141.
Although epidermal growth factor receptor–mutant lung cancers generally don’t benefit from checkpoint inhibitors, there may be some subtypes that do respond, results of a large, multi-institution analysis suggest.
Compared with wild-type lung cancer cases, tumors with epidermal growth factor receptor (EGFR) exon 19 deletion did indeed have worse outcomes after progressive death-1/progressive death–ligand 1 (PD-1/PD-L1) blockade; however, tumors harboring EGFR L858R mutations had comparable response rates and overall survival.
These findings don’t change clinical practice, but do serve as a “foundation” for more research into which patients with EGFR-mutant disease might benefit from immunotherapy, according to Katherine Hastings, PhD, of Yale University, New Haven, Conn., and associates.
“We unequivocally support the guidance that EGFR TKIs should be the preferred first-line treatment option for patients with EGFR-mutant lung cancer,” Dr. Hastings and coauthors wrote in Annals of Oncology.
To date, clinical investigations of immune checkpoint inhibitors in patients with EGFR-mutant lung cancers have largely been discouraging, the authors wrote. However, there have been some exceptions, including a phase 2 study where third-line durvalumab showed activity in some patients with EGFR-positive, PD-L1-expressing advanced non–small cell lung cancer (NSCLC).
Accordingly, the investigators sought to determine whether specific molecular features made a difference. They looked at a total of 171 EGFR-mutant lung cancers that had been treated with PD-1/PD-L1 inhibitors, alone or in combination with a cytotoxic T-lymphocyte antigen 4 inhibitor.
In their analysis, they drilled down on the most common EGFR tyrosine kinase inhibitor–sensitizing alleles: EGFR exon 19 deletions, which represented 76 cases, and EGFR L858R, which represented 44 cases. For comparison, they looked at a cohort of 212 patients with EGFR wild-type NSCLC who had also been treated with immune checkpoint inhibitors.
EGFR exon 19 deletion cases had a significantly lower overall response rate versus EGFR wild-type tumors, at 7% versus 22%, respectively (P = .002), the investigators found. Likewise, overall survival was significantly reduced, with a hazard ratio of 0.69 (95% confidence interval, 0.493-0.965; P = .03).
By contrast, EGFR L858R tumors had similar response rates versus EGFR wild type, at 16% and 22%, respectively (P = .42); overall survival also was similar, with a hazard ratio of 0.917 (95% CI, 0.597-1.409; P = .69).
Of note, progression-free survival was reduced in both EGFR L858R and EGFR exon 19 deletion cases as compared with EGFR wild-type cases, though the investigators wrote that discrepancy might be related to variations in scanning intervals between different institutions that contributed cases to the study.
“Overall, these data suggest that patients with EGFR exon 19–mutant tumors, in particular, have a significantly reduced benefit upon treatment with immune checkpoint inhibitors,” the authors noted.
In a separate but related analysis of 383 patients with EGFR-mutant lung cancer, tumor mutation burden was lower in EGFR exon 19 deletion cases as compared with the EGFR L858R cases. That finding lined up with the immunotherapy response data, the investigators wrote, though it’s unclear what might be driving the differences in tumor burden between these two groups.
There were no differences in response to immune checkpoint blockade based on whether or not tumors harbored the EGFR T790M mutation, the investigators added. Similarly, PD-L1 expression did not impact response.
The study authors reported disclosures related to Astellas Pharma, AstraZeneca, Bristol-Myers Squibb, Calithera Biosciences, Daiichi, Eli Lilly, Merck, Mirati Therapeutics, Novartis, Pfizer, Roche, and Takeda, among others.
SOURCE: Hastings K et al. Ann Oncol. 2019 May 14. doi: 10.1093/annonc/mdz141.
Although epidermal growth factor receptor–mutant lung cancers generally don’t benefit from checkpoint inhibitors, there may be some subtypes that do respond, results of a large, multi-institution analysis suggest.
Compared with wild-type lung cancer cases, tumors with epidermal growth factor receptor (EGFR) exon 19 deletion did indeed have worse outcomes after progressive death-1/progressive death–ligand 1 (PD-1/PD-L1) blockade; however, tumors harboring EGFR L858R mutations had comparable response rates and overall survival.
These findings don’t change clinical practice, but do serve as a “foundation” for more research into which patients with EGFR-mutant disease might benefit from immunotherapy, according to Katherine Hastings, PhD, of Yale University, New Haven, Conn., and associates.
“We unequivocally support the guidance that EGFR TKIs should be the preferred first-line treatment option for patients with EGFR-mutant lung cancer,” Dr. Hastings and coauthors wrote in Annals of Oncology.
To date, clinical investigations of immune checkpoint inhibitors in patients with EGFR-mutant lung cancers have largely been discouraging, the authors wrote. However, there have been some exceptions, including a phase 2 study where third-line durvalumab showed activity in some patients with EGFR-positive, PD-L1-expressing advanced non–small cell lung cancer (NSCLC).
Accordingly, the investigators sought to determine whether specific molecular features made a difference. They looked at a total of 171 EGFR-mutant lung cancers that had been treated with PD-1/PD-L1 inhibitors, alone or in combination with a cytotoxic T-lymphocyte antigen 4 inhibitor.
In their analysis, they drilled down on the most common EGFR tyrosine kinase inhibitor–sensitizing alleles: EGFR exon 19 deletions, which represented 76 cases, and EGFR L858R, which represented 44 cases. For comparison, they looked at a cohort of 212 patients with EGFR wild-type NSCLC who had also been treated with immune checkpoint inhibitors.
EGFR exon 19 deletion cases had a significantly lower overall response rate versus EGFR wild-type tumors, at 7% versus 22%, respectively (P = .002), the investigators found. Likewise, overall survival was significantly reduced, with a hazard ratio of 0.69 (95% confidence interval, 0.493-0.965; P = .03).
By contrast, EGFR L858R tumors had similar response rates versus EGFR wild type, at 16% and 22%, respectively (P = .42); overall survival also was similar, with a hazard ratio of 0.917 (95% CI, 0.597-1.409; P = .69).
Of note, progression-free survival was reduced in both EGFR L858R and EGFR exon 19 deletion cases as compared with EGFR wild-type cases, though the investigators wrote that discrepancy might be related to variations in scanning intervals between different institutions that contributed cases to the study.
“Overall, these data suggest that patients with EGFR exon 19–mutant tumors, in particular, have a significantly reduced benefit upon treatment with immune checkpoint inhibitors,” the authors noted.
In a separate but related analysis of 383 patients with EGFR-mutant lung cancer, tumor mutation burden was lower in EGFR exon 19 deletion cases as compared with the EGFR L858R cases. That finding lined up with the immunotherapy response data, the investigators wrote, though it’s unclear what might be driving the differences in tumor burden between these two groups.
There were no differences in response to immune checkpoint blockade based on whether or not tumors harbored the EGFR T790M mutation, the investigators added. Similarly, PD-L1 expression did not impact response.
The study authors reported disclosures related to Astellas Pharma, AstraZeneca, Bristol-Myers Squibb, Calithera Biosciences, Daiichi, Eli Lilly, Merck, Mirati Therapeutics, Novartis, Pfizer, Roche, and Takeda, among others.
SOURCE: Hastings K et al. Ann Oncol. 2019 May 14. doi: 10.1093/annonc/mdz141.
FROM ANNALS OF ONCOLOGY
CMS proposes payment increase for administering CAR T in the hospital
Hospitals could get a payment bump for administering chimeric antigen receptor (CAR) T-cell therapies under a proposed rule issued by the Centers for Medicare & Medicaid Services.
The proposal calls for raising the new technology add-on payment (NTAP) associated with the therapies from 50% of the technology to 65%, an increase from $186,500 to $242,450.
Beginning with discharges on Oct. 1, 2019, if discharge costs involving a new medical service or technology exceed the full Medicare Severity Diagnosis-Related Group (DRG) payment, Medicare would make an add-on payment of either 65% of the cost of the new medical service or technology, or 65% of the amount by which the costs of the case exceed the standard DRG payment, whichever is less.
Roy Silverstein, MD, president of the American Society of Hematology, said the group was pleased that the CMS is examining its existing payment policies to identify more realistic ways to account for the costs of administering CAR T-cell therapies.
“While ASH had originally suggested a higher [new technology] payment, any increase is an improvement,” Dr. Silverstein said in a statement. “While the proposal from CMS is promising, it is not a one-stop solution for making CAR T more accessible to patients. Just as these therapies are innovative, it is going to take some innovation on the part of CMS to develop a plan that equitably compensates providers and institutions so that offering the therapy is sustainable.”
The agency’s proposal follows an August 2018 final rule by the CMS that set a new payment scheme for inpatient administration of two CAR T-cell therapies. The rule categorized CAR T-cell therapies under the umbrella of the renamed Medicare Severity–Diagnosis Related Groups (MS-DRG) 016 – Autologous Bone Marrow Transplant with CC/MCC or T-cell Immunotherapy – and assigned ICD-10 PCS procedure codes XW033C3 and XW043C3 to the use of axicabtagene ciloleucel (Yescarta) and tisagenlecleucel (Kymriah) in the inpatient setting for fiscal year 2019, which began in October 2018.
In April 2018, the CMS announced payment rates for outpatient administration of the two drugs, settling on $395,380 for axicabtagene ciloleucel and $500,839 for tisagenlecleucel. The two medications have list prices of $373,000 and $475,000, respectively.
In February 2019, the CMS also proposed to cover CAR T-cell therapy for cancer patients participating in clinical trials that study the treatment’s effectiveness. A final decision on the proposal is expected in May 2019.
In the current proposal, the CMS acknowledged requests calling for the agency to create a new MS-DRG for procedures involving CAR T-cell therapies to improve payment in the inpatient setting. However, the agency declined to create a new MS-DRG for CAR T-cell cases, writing that the move is premature given the relative newness of CAR T-cell therapy and the agency’s proposal to continue new technology add-on payments for fiscal 2020 for Kymriah and Yescarta.
However, the agency is requesting public comments on whether, in light of additional experience with billing and payment for cases involving CAR T-cell therapies to Medicare patients, the CMS should consider using a specific cost-to-charge ratio for ICD-10-PCS procedure codes used to report the performance of procedures involving CAR T-cell therapies.
Comments on the proposed rule will be accepted until June 24.
Hospitals could get a payment bump for administering chimeric antigen receptor (CAR) T-cell therapies under a proposed rule issued by the Centers for Medicare & Medicaid Services.
The proposal calls for raising the new technology add-on payment (NTAP) associated with the therapies from 50% of the technology to 65%, an increase from $186,500 to $242,450.
Beginning with discharges on Oct. 1, 2019, if discharge costs involving a new medical service or technology exceed the full Medicare Severity Diagnosis-Related Group (DRG) payment, Medicare would make an add-on payment of either 65% of the cost of the new medical service or technology, or 65% of the amount by which the costs of the case exceed the standard DRG payment, whichever is less.
Roy Silverstein, MD, president of the American Society of Hematology, said the group was pleased that the CMS is examining its existing payment policies to identify more realistic ways to account for the costs of administering CAR T-cell therapies.
“While ASH had originally suggested a higher [new technology] payment, any increase is an improvement,” Dr. Silverstein said in a statement. “While the proposal from CMS is promising, it is not a one-stop solution for making CAR T more accessible to patients. Just as these therapies are innovative, it is going to take some innovation on the part of CMS to develop a plan that equitably compensates providers and institutions so that offering the therapy is sustainable.”
The agency’s proposal follows an August 2018 final rule by the CMS that set a new payment scheme for inpatient administration of two CAR T-cell therapies. The rule categorized CAR T-cell therapies under the umbrella of the renamed Medicare Severity–Diagnosis Related Groups (MS-DRG) 016 – Autologous Bone Marrow Transplant with CC/MCC or T-cell Immunotherapy – and assigned ICD-10 PCS procedure codes XW033C3 and XW043C3 to the use of axicabtagene ciloleucel (Yescarta) and tisagenlecleucel (Kymriah) in the inpatient setting for fiscal year 2019, which began in October 2018.
In April 2018, the CMS announced payment rates for outpatient administration of the two drugs, settling on $395,380 for axicabtagene ciloleucel and $500,839 for tisagenlecleucel. The two medications have list prices of $373,000 and $475,000, respectively.
In February 2019, the CMS also proposed to cover CAR T-cell therapy for cancer patients participating in clinical trials that study the treatment’s effectiveness. A final decision on the proposal is expected in May 2019.
In the current proposal, the CMS acknowledged requests calling for the agency to create a new MS-DRG for procedures involving CAR T-cell therapies to improve payment in the inpatient setting. However, the agency declined to create a new MS-DRG for CAR T-cell cases, writing that the move is premature given the relative newness of CAR T-cell therapy and the agency’s proposal to continue new technology add-on payments for fiscal 2020 for Kymriah and Yescarta.
However, the agency is requesting public comments on whether, in light of additional experience with billing and payment for cases involving CAR T-cell therapies to Medicare patients, the CMS should consider using a specific cost-to-charge ratio for ICD-10-PCS procedure codes used to report the performance of procedures involving CAR T-cell therapies.
Comments on the proposed rule will be accepted until June 24.
Hospitals could get a payment bump for administering chimeric antigen receptor (CAR) T-cell therapies under a proposed rule issued by the Centers for Medicare & Medicaid Services.
The proposal calls for raising the new technology add-on payment (NTAP) associated with the therapies from 50% of the technology to 65%, an increase from $186,500 to $242,450.
Beginning with discharges on Oct. 1, 2019, if discharge costs involving a new medical service or technology exceed the full Medicare Severity Diagnosis-Related Group (DRG) payment, Medicare would make an add-on payment of either 65% of the cost of the new medical service or technology, or 65% of the amount by which the costs of the case exceed the standard DRG payment, whichever is less.
Roy Silverstein, MD, president of the American Society of Hematology, said the group was pleased that the CMS is examining its existing payment policies to identify more realistic ways to account for the costs of administering CAR T-cell therapies.
“While ASH had originally suggested a higher [new technology] payment, any increase is an improvement,” Dr. Silverstein said in a statement. “While the proposal from CMS is promising, it is not a one-stop solution for making CAR T more accessible to patients. Just as these therapies are innovative, it is going to take some innovation on the part of CMS to develop a plan that equitably compensates providers and institutions so that offering the therapy is sustainable.”
The agency’s proposal follows an August 2018 final rule by the CMS that set a new payment scheme for inpatient administration of two CAR T-cell therapies. The rule categorized CAR T-cell therapies under the umbrella of the renamed Medicare Severity–Diagnosis Related Groups (MS-DRG) 016 – Autologous Bone Marrow Transplant with CC/MCC or T-cell Immunotherapy – and assigned ICD-10 PCS procedure codes XW033C3 and XW043C3 to the use of axicabtagene ciloleucel (Yescarta) and tisagenlecleucel (Kymriah) in the inpatient setting for fiscal year 2019, which began in October 2018.
In April 2018, the CMS announced payment rates for outpatient administration of the two drugs, settling on $395,380 for axicabtagene ciloleucel and $500,839 for tisagenlecleucel. The two medications have list prices of $373,000 and $475,000, respectively.
In February 2019, the CMS also proposed to cover CAR T-cell therapy for cancer patients participating in clinical trials that study the treatment’s effectiveness. A final decision on the proposal is expected in May 2019.
In the current proposal, the CMS acknowledged requests calling for the agency to create a new MS-DRG for procedures involving CAR T-cell therapies to improve payment in the inpatient setting. However, the agency declined to create a new MS-DRG for CAR T-cell cases, writing that the move is premature given the relative newness of CAR T-cell therapy and the agency’s proposal to continue new technology add-on payments for fiscal 2020 for Kymriah and Yescarta.
However, the agency is requesting public comments on whether, in light of additional experience with billing and payment for cases involving CAR T-cell therapies to Medicare patients, the CMS should consider using a specific cost-to-charge ratio for ICD-10-PCS procedure codes used to report the performance of procedures involving CAR T-cell therapies.
Comments on the proposed rule will be accepted until June 24.