User login
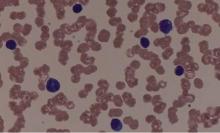
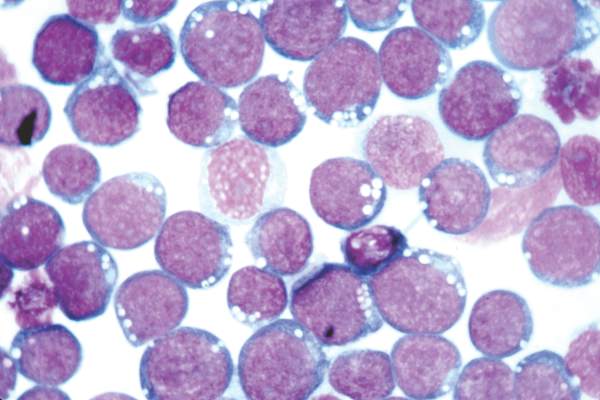
Expert panel offers treatment recommendations in Waldenström macroglobulinemia
Treatment recommendations for Waldenström macroglobulinemia have been updated based on the advice of a task force convened at the Eighth International Workshop on Waldenström Macroglobulinemia; the guidelines have been published in Blood.
The task force was impaneled to review recently published and ongoing clinical trial data as well as the impact of the newly recognized mutations MYD88 and CXCR4 on treatment decisions, indications for B-cell receptor and proteasome inhibitors, and future clinical trial initiatives.
The panel reiterated that the criteria for initiating therapy include immunoglobulin M (IgM)-related complications and/or symptoms that are related to direct involvement of the bone marrow by tumor cells, constitutional symptoms, and bulky extramedullary disease. Patients presenting with symptoms that include symptomatic hyperviscosity, moderate to severe hemolytic anemia, and symptomatic cryoglobulinemia need immediate treatment.
Close observation is recommended for the subgroup of patients who do not really fulfill the criteria for a diagnosis of Waldenström macroglobulinemia (WM), and whose laboratory findings may be the only indicator of the presence of a progressive disease.
Treatment recommendations
For symptomatic patients in the first-line setting, “anti-CD20 monoclonal antibody therapy alone or in combination with chemotherapy is an important standard of care for most patients with WM,” the authors, led by Veronique Leblond, MD, of Pitié-Salpêtrière Hôpital, Paris, wrote.
Rituximab is frequently used in WM, either as monotherapy or in combination with chemotherapeutic agents. The panel cautions that rituximab as monotherapy should be avoided in patients with high IgM levels, because of a lower chance of response and the risk of an IgM flare.
In patients with high IgM levels (typically around 4,000 mg/dL), plasmapheresis can be initiated before rituximab therapy, and plasmapheresis should always and immediately be used when symptomatic hyperviscosity is present. However, plasmapheresis alone is not an effective treatment for WM and must be followed by a rapidly acting cytoreductive regimen.
Several rituximab combinations are recommended by the panel. These include:
• Dexamethasone-rituximab-cyclophosphamide, which is an active and safe option, has a manageable toxicity, and can be considered for frail patients who need combination therapy.
• Bendamustine-rituximab is effective for front-line treatment and is well tolerated even in elderly patients who experience limited episodes of myelosuppression and infections.
Other therapeutic regimens include bortezomib-based therapy, which is recommended for patients with high IgM levels, symptomatic hyperviscosity, cryoglobulinemia or cold agglutinemia, amyloidosis, and renal impairment or in young patients who prefer to avoid alkylator or nucleoside analogue therapy.
Another option is carfilzomib-based therapy, which is an emerging “neuropathy-sparing” regimen for proteasome-inhibitor–based therapy, although it may not be the best choice for elderly patients with preexisting cardiac conditions due to potential cardiac toxicity.
Ibrutinib has been approved as a primary therapy for patients who are not candidates for chemoimmunotherapy, but the authors point out that the optimal use of this agent is still being investigated.
“The aim of the first-line treatments is to reach a high response rate with a prolonged progression-free survival,” write the authors. “The panel agrees that there is need to perform clinical trials with chemotherapy-free combinations with new compounds alone or in combination with anti-CD20 antibodies.”
For symptomatic previously treated patients
The panel also offered recommendations for previously treated symptomatic patients who have relapsed or are refractory to treatment.
Any of the interventions recommended for symptomatic, untreated patients can be considered for those who have already gone through first line therapy. Retreatment can be considered with a specific intervention if a response was achieved for 2 or more years with that therapy, although they caution that patients who have progressed on first-line ibrutinib should not use it again.
Ofatumumab is a potential option for patients who are unable to tolerate rituximab, and nucleoside analogues can be considered in fit patients who have not responded to less-toxic treatments.
Another option in this setting is everolimus, although since it is associated with considerable toxicities, the best candidates for this drug are those who have not responded to or have progressed after multiple lines of other better-tolerated regimens.
Immunomodulatory agents can also be considered, but in the context of a clinical trial only, because of their potential adverse events.
Finally, the panel also agreed that stem cell transplantation should be discussed with select patients, and while it is a feasible and effective treatment option for high-risk WM patients, it should be ideally offered at early relapse.
Investigating B-cell receptor (BCR) pathway inhibitors along with existing and novel compounds in patients in the relapsed/refractory setting should be a priority, according to the panel.
“BCR inhibitors, combined with proteasome inhibitors, would be of interest for overcoming resistance by interfering with the two key pathways that are affected by MYD88,” wrote Dr. Leblond and coauthors.
Treatment recommendations for Waldenström macroglobulinemia have been updated based on the advice of a task force convened at the Eighth International Workshop on Waldenström Macroglobulinemia; the guidelines have been published in Blood.
The task force was impaneled to review recently published and ongoing clinical trial data as well as the impact of the newly recognized mutations MYD88 and CXCR4 on treatment decisions, indications for B-cell receptor and proteasome inhibitors, and future clinical trial initiatives.
The panel reiterated that the criteria for initiating therapy include immunoglobulin M (IgM)-related complications and/or symptoms that are related to direct involvement of the bone marrow by tumor cells, constitutional symptoms, and bulky extramedullary disease. Patients presenting with symptoms that include symptomatic hyperviscosity, moderate to severe hemolytic anemia, and symptomatic cryoglobulinemia need immediate treatment.
Close observation is recommended for the subgroup of patients who do not really fulfill the criteria for a diagnosis of Waldenström macroglobulinemia (WM), and whose laboratory findings may be the only indicator of the presence of a progressive disease.
Treatment recommendations
For symptomatic patients in the first-line setting, “anti-CD20 monoclonal antibody therapy alone or in combination with chemotherapy is an important standard of care for most patients with WM,” the authors, led by Veronique Leblond, MD, of Pitié-Salpêtrière Hôpital, Paris, wrote.
Rituximab is frequently used in WM, either as monotherapy or in combination with chemotherapeutic agents. The panel cautions that rituximab as monotherapy should be avoided in patients with high IgM levels, because of a lower chance of response and the risk of an IgM flare.
In patients with high IgM levels (typically around 4,000 mg/dL), plasmapheresis can be initiated before rituximab therapy, and plasmapheresis should always and immediately be used when symptomatic hyperviscosity is present. However, plasmapheresis alone is not an effective treatment for WM and must be followed by a rapidly acting cytoreductive regimen.
Several rituximab combinations are recommended by the panel. These include:
• Dexamethasone-rituximab-cyclophosphamide, which is an active and safe option, has a manageable toxicity, and can be considered for frail patients who need combination therapy.
• Bendamustine-rituximab is effective for front-line treatment and is well tolerated even in elderly patients who experience limited episodes of myelosuppression and infections.
Other therapeutic regimens include bortezomib-based therapy, which is recommended for patients with high IgM levels, symptomatic hyperviscosity, cryoglobulinemia or cold agglutinemia, amyloidosis, and renal impairment or in young patients who prefer to avoid alkylator or nucleoside analogue therapy.
Another option is carfilzomib-based therapy, which is an emerging “neuropathy-sparing” regimen for proteasome-inhibitor–based therapy, although it may not be the best choice for elderly patients with preexisting cardiac conditions due to potential cardiac toxicity.
Ibrutinib has been approved as a primary therapy for patients who are not candidates for chemoimmunotherapy, but the authors point out that the optimal use of this agent is still being investigated.
“The aim of the first-line treatments is to reach a high response rate with a prolonged progression-free survival,” write the authors. “The panel agrees that there is need to perform clinical trials with chemotherapy-free combinations with new compounds alone or in combination with anti-CD20 antibodies.”
For symptomatic previously treated patients
The panel also offered recommendations for previously treated symptomatic patients who have relapsed or are refractory to treatment.
Any of the interventions recommended for symptomatic, untreated patients can be considered for those who have already gone through first line therapy. Retreatment can be considered with a specific intervention if a response was achieved for 2 or more years with that therapy, although they caution that patients who have progressed on first-line ibrutinib should not use it again.
Ofatumumab is a potential option for patients who are unable to tolerate rituximab, and nucleoside analogues can be considered in fit patients who have not responded to less-toxic treatments.
Another option in this setting is everolimus, although since it is associated with considerable toxicities, the best candidates for this drug are those who have not responded to or have progressed after multiple lines of other better-tolerated regimens.
Immunomodulatory agents can also be considered, but in the context of a clinical trial only, because of their potential adverse events.
Finally, the panel also agreed that stem cell transplantation should be discussed with select patients, and while it is a feasible and effective treatment option for high-risk WM patients, it should be ideally offered at early relapse.
Investigating B-cell receptor (BCR) pathway inhibitors along with existing and novel compounds in patients in the relapsed/refractory setting should be a priority, according to the panel.
“BCR inhibitors, combined with proteasome inhibitors, would be of interest for overcoming resistance by interfering with the two key pathways that are affected by MYD88,” wrote Dr. Leblond and coauthors.
Treatment recommendations for Waldenström macroglobulinemia have been updated based on the advice of a task force convened at the Eighth International Workshop on Waldenström Macroglobulinemia; the guidelines have been published in Blood.
The task force was impaneled to review recently published and ongoing clinical trial data as well as the impact of the newly recognized mutations MYD88 and CXCR4 on treatment decisions, indications for B-cell receptor and proteasome inhibitors, and future clinical trial initiatives.
The panel reiterated that the criteria for initiating therapy include immunoglobulin M (IgM)-related complications and/or symptoms that are related to direct involvement of the bone marrow by tumor cells, constitutional symptoms, and bulky extramedullary disease. Patients presenting with symptoms that include symptomatic hyperviscosity, moderate to severe hemolytic anemia, and symptomatic cryoglobulinemia need immediate treatment.
Close observation is recommended for the subgroup of patients who do not really fulfill the criteria for a diagnosis of Waldenström macroglobulinemia (WM), and whose laboratory findings may be the only indicator of the presence of a progressive disease.
Treatment recommendations
For symptomatic patients in the first-line setting, “anti-CD20 monoclonal antibody therapy alone or in combination with chemotherapy is an important standard of care for most patients with WM,” the authors, led by Veronique Leblond, MD, of Pitié-Salpêtrière Hôpital, Paris, wrote.
Rituximab is frequently used in WM, either as monotherapy or in combination with chemotherapeutic agents. The panel cautions that rituximab as monotherapy should be avoided in patients with high IgM levels, because of a lower chance of response and the risk of an IgM flare.
In patients with high IgM levels (typically around 4,000 mg/dL), plasmapheresis can be initiated before rituximab therapy, and plasmapheresis should always and immediately be used when symptomatic hyperviscosity is present. However, plasmapheresis alone is not an effective treatment for WM and must be followed by a rapidly acting cytoreductive regimen.
Several rituximab combinations are recommended by the panel. These include:
• Dexamethasone-rituximab-cyclophosphamide, which is an active and safe option, has a manageable toxicity, and can be considered for frail patients who need combination therapy.
• Bendamustine-rituximab is effective for front-line treatment and is well tolerated even in elderly patients who experience limited episodes of myelosuppression and infections.
Other therapeutic regimens include bortezomib-based therapy, which is recommended for patients with high IgM levels, symptomatic hyperviscosity, cryoglobulinemia or cold agglutinemia, amyloidosis, and renal impairment or in young patients who prefer to avoid alkylator or nucleoside analogue therapy.
Another option is carfilzomib-based therapy, which is an emerging “neuropathy-sparing” regimen for proteasome-inhibitor–based therapy, although it may not be the best choice for elderly patients with preexisting cardiac conditions due to potential cardiac toxicity.
Ibrutinib has been approved as a primary therapy for patients who are not candidates for chemoimmunotherapy, but the authors point out that the optimal use of this agent is still being investigated.
“The aim of the first-line treatments is to reach a high response rate with a prolonged progression-free survival,” write the authors. “The panel agrees that there is need to perform clinical trials with chemotherapy-free combinations with new compounds alone or in combination with anti-CD20 antibodies.”
For symptomatic previously treated patients
The panel also offered recommendations for previously treated symptomatic patients who have relapsed or are refractory to treatment.
Any of the interventions recommended for symptomatic, untreated patients can be considered for those who have already gone through first line therapy. Retreatment can be considered with a specific intervention if a response was achieved for 2 or more years with that therapy, although they caution that patients who have progressed on first-line ibrutinib should not use it again.
Ofatumumab is a potential option for patients who are unable to tolerate rituximab, and nucleoside analogues can be considered in fit patients who have not responded to less-toxic treatments.
Another option in this setting is everolimus, although since it is associated with considerable toxicities, the best candidates for this drug are those who have not responded to or have progressed after multiple lines of other better-tolerated regimens.
Immunomodulatory agents can also be considered, but in the context of a clinical trial only, because of their potential adverse events.
Finally, the panel also agreed that stem cell transplantation should be discussed with select patients, and while it is a feasible and effective treatment option for high-risk WM patients, it should be ideally offered at early relapse.
Investigating B-cell receptor (BCR) pathway inhibitors along with existing and novel compounds in patients in the relapsed/refractory setting should be a priority, according to the panel.
“BCR inhibitors, combined with proteasome inhibitors, would be of interest for overcoming resistance by interfering with the two key pathways that are affected by MYD88,” wrote Dr. Leblond and coauthors.
FROM BLOOD
Triptorelin doesn’t prevent ovarian failure in young women treated for lymphoma
Protective treatment with the GnRH agonist triptorelin failed to prevent chemotherapy-induced premature ovarian failure in young women with lymphoma, according to a report published in the Journal of Clinical Oncology.
This type of protective therapy has been used for at least 20 years but is still controversial. Some clinical practice guidelines endorse the practice, but others do not. The evidence is both sparse and ambiguous, said Isabelle Demeestere, MD, of the research laboratory on human reproduction, Université Libre de Bruxelles (Belgium), and her associates.
In what they described as “the first randomized clinical trial providing accurate information on ovarian function and fertility after a median of 5 years of follow-up,” the investigators assessed 67 young women (median age, 26 years) treated for Hodgkin’s or non-Hodgkin’s lymphoma at 15 cancer centers in France, Belgium, and Italy. These patients had been randomly assigned to receive chemotherapy plus triptorelin (32 women) or chemotherapy plus placebo injections (35 women) at baseline.
Premature ovarian failure, as measured by elevated FSH levels, occurred in six of the GnRH group and eight of the control group, a nonsignificant difference. Moreover, the rate of recovery of ovarian function, as measured by at least one FSH level of 15 IU/L or less, was the same between the two study groups. And 17 women (53%) in the GnRH group eventually achieved pregnancy, which is not significantly different from the 43% pregnancy rate in the control group (15 of 35 women).
Five women – two in the GnRH group and three in the control group – achieved pregnancy during long-term follow-up, even though they had been classified as having premature ovarian failure. This “confirms the possibility of incidental ovarian cycle recovery, leading to fertility restoration several years after treatment in this young population,” Dr. Demeestere and her associates wrote (J Clin Oncol. 2016. [doi:10.1200/JCO.2015.65.884]).
The study findings “suggest caution” regarding guidelines that recommend using GnRH agonists to preserve fertility in young women undergoing chemotherapy, they added.
Protective treatment with the GnRH agonist triptorelin failed to prevent chemotherapy-induced premature ovarian failure in young women with lymphoma, according to a report published in the Journal of Clinical Oncology.
This type of protective therapy has been used for at least 20 years but is still controversial. Some clinical practice guidelines endorse the practice, but others do not. The evidence is both sparse and ambiguous, said Isabelle Demeestere, MD, of the research laboratory on human reproduction, Université Libre de Bruxelles (Belgium), and her associates.
In what they described as “the first randomized clinical trial providing accurate information on ovarian function and fertility after a median of 5 years of follow-up,” the investigators assessed 67 young women (median age, 26 years) treated for Hodgkin’s or non-Hodgkin’s lymphoma at 15 cancer centers in France, Belgium, and Italy. These patients had been randomly assigned to receive chemotherapy plus triptorelin (32 women) or chemotherapy plus placebo injections (35 women) at baseline.
Premature ovarian failure, as measured by elevated FSH levels, occurred in six of the GnRH group and eight of the control group, a nonsignificant difference. Moreover, the rate of recovery of ovarian function, as measured by at least one FSH level of 15 IU/L or less, was the same between the two study groups. And 17 women (53%) in the GnRH group eventually achieved pregnancy, which is not significantly different from the 43% pregnancy rate in the control group (15 of 35 women).
Five women – two in the GnRH group and three in the control group – achieved pregnancy during long-term follow-up, even though they had been classified as having premature ovarian failure. This “confirms the possibility of incidental ovarian cycle recovery, leading to fertility restoration several years after treatment in this young population,” Dr. Demeestere and her associates wrote (J Clin Oncol. 2016. [doi:10.1200/JCO.2015.65.884]).
The study findings “suggest caution” regarding guidelines that recommend using GnRH agonists to preserve fertility in young women undergoing chemotherapy, they added.
Protective treatment with the GnRH agonist triptorelin failed to prevent chemotherapy-induced premature ovarian failure in young women with lymphoma, according to a report published in the Journal of Clinical Oncology.
This type of protective therapy has been used for at least 20 years but is still controversial. Some clinical practice guidelines endorse the practice, but others do not. The evidence is both sparse and ambiguous, said Isabelle Demeestere, MD, of the research laboratory on human reproduction, Université Libre de Bruxelles (Belgium), and her associates.
In what they described as “the first randomized clinical trial providing accurate information on ovarian function and fertility after a median of 5 years of follow-up,” the investigators assessed 67 young women (median age, 26 years) treated for Hodgkin’s or non-Hodgkin’s lymphoma at 15 cancer centers in France, Belgium, and Italy. These patients had been randomly assigned to receive chemotherapy plus triptorelin (32 women) or chemotherapy plus placebo injections (35 women) at baseline.
Premature ovarian failure, as measured by elevated FSH levels, occurred in six of the GnRH group and eight of the control group, a nonsignificant difference. Moreover, the rate of recovery of ovarian function, as measured by at least one FSH level of 15 IU/L or less, was the same between the two study groups. And 17 women (53%) in the GnRH group eventually achieved pregnancy, which is not significantly different from the 43% pregnancy rate in the control group (15 of 35 women).
Five women – two in the GnRH group and three in the control group – achieved pregnancy during long-term follow-up, even though they had been classified as having premature ovarian failure. This “confirms the possibility of incidental ovarian cycle recovery, leading to fertility restoration several years after treatment in this young population,” Dr. Demeestere and her associates wrote (J Clin Oncol. 2016. [doi:10.1200/JCO.2015.65.884]).
The study findings “suggest caution” regarding guidelines that recommend using GnRH agonists to preserve fertility in young women undergoing chemotherapy, they added.
FROM JOURNAL OF CLINICAL ONCOLOGY
Key clinical point: The GnRH agonist triptorelin failed to prevent chemotherapy-induced premature ovarian failure in young women with lymphoma.
Major finding: Premature ovarian failure occurred in six of the GnRH group and eight of the control group, a nonsignificant difference.
Data source: A multicenter, randomized clinical trial involving 67 women followed for 5 years.
Disclosures: This study was supported by Fonds National de la Recherche Scientifique and Ipsen Pharmaceutical Group. Dr. Demeestere reported having no relevant financial disclosures; her associates reported ties to numerous industry sources.
Long-term analysis shows dwindling RT benefit for DLBCL
Long-term follow-up from two randomized trials shows that the treatment advantage initially seen by adding radiotherapy to the CHOP regimen in patients with limited-stage diffuse large B-cell lymphoma disappeared over time.
After a median follow-up of nearly 18 years, there were no significant differences in either progression-free survival (PFS) or overall survival between patients who had received standard CHOP chemotherapy (cyclophosphamide, doxorubicin, vincristine, and prednisone) for 8 cycles (CHOP8), or 3 cycles of CHOP with involved-field radiotherapy (CHOP3RT), reported Deborah M. Stephens, DO, of the University of Utah in Salt Lake City, and her colleagues.
The addition of rituximab to CHOP plus involved-field radiotherapy (IFRT) in a separate cohort did not appear to reduce the continued relapse risk, the investigators noted (J Clin Oncol. 2016 Jul. doi: 10.1200/JCO.2015.65.4582).
The findings suggest that long-term follow-up of patients enrolled in clinical trials may provide clinically important additional information, and that limited-stage diffuse large B-cell lymphoma (DLBCL) may have a biology that is distinctly different from that of advanced stage DLBCL, the investigators wrote.
Initial results from the SWOG (Southwest Oncology Group) Study 8736, published after a median follow-up of 4.4 years, showed that CHOP3RT was associated with significantly better 5-year PFS and OS rates, compared with patients treated with CHOP8.
A second trial, SWOG Study 0014, looked at the addition of rituximab (Rituxan) to CHOP plus IFRT in patients with high-risk DLBCL with at least one adverse feature of the stage-modified International Prognostic Index. In this trial, after a median follow-up of 5.3 years, 4-year PFS was 88%, and 4-year OS was 92%.
5 years not enough
Results of both trials were published around the 5-year follow-up mark. However, an analysis of 10-year follow-up data from the S8736 trial, published only as an abstract (Blood. 98:724a-725a, 2001; abstr 3024), showed that “the survival curves for CHOP8 and CHOP3RT, surprisingly, began to overlap,” Dr. Stephens and her colleagues wrote.
“Additionally, for both S8736 and S0014, no plateau had been reached in the PFS curves. These data contrasted the expected cure rates for advanced-stage DLBCL, leading to the hypothesis that limited-stage DLBCL may have a unique biology from its advanced-stage counterpart,” they added.
Although more than half of patients with advanced-stage diffuse large B-cell lymphoma (DLBCL) can be cured with anthracycline-based chemotherapy, cure rates are markedly lower among patients with limited-stage disease, defined as Ann Arbor stage I or II, confined to a single irradiation field. Limited-stage disease accounts for approximately one-third DLBCL cases.
To see whether the trends observed at 10 years continued, the investigators conducted a survival analysis from S8736 and compared the data from similar patients in the S0014 study.
They found that after a median follow-up of 17.7 years in S8736, median PFS was 12.0 years for patients treated with CHOP8, and 11.1 years for patients treated with CHOP3RT, a difference that was not statistically significant.
Median OS was 13.0 and 13.7 years, respectively, and was also not significant.
In S0014, after a median follow-up of 12 years, the 5-year OS rate was 82%, and 10-year rate was 67%. In this trial, the investigators observed “a persistent pattern of relapse despite the addition of rituximab.”
“The populations were not entirely identical; however, even the addition of rituximab as per S0014 to combined-modality therapy did not seem to mitigate the continued relapse risk, underscoring the value of prolonged observation of clinical trial patients and possible unique biology of limited-stage DLBCL,” Dr. Stephens and her associates wrote.
The study was sponsored by grants from the National Institutes of Health. Dr. Stephens reported having no financial disclosures. Several coauthors disclosed research funding, consulting, speakers’ bureau participation or travel expenses from various oncology drug makers.
Long-term follow-up from two randomized trials shows that the treatment advantage initially seen by adding radiotherapy to the CHOP regimen in patients with limited-stage diffuse large B-cell lymphoma disappeared over time.
After a median follow-up of nearly 18 years, there were no significant differences in either progression-free survival (PFS) or overall survival between patients who had received standard CHOP chemotherapy (cyclophosphamide, doxorubicin, vincristine, and prednisone) for 8 cycles (CHOP8), or 3 cycles of CHOP with involved-field radiotherapy (CHOP3RT), reported Deborah M. Stephens, DO, of the University of Utah in Salt Lake City, and her colleagues.
The addition of rituximab to CHOP plus involved-field radiotherapy (IFRT) in a separate cohort did not appear to reduce the continued relapse risk, the investigators noted (J Clin Oncol. 2016 Jul. doi: 10.1200/JCO.2015.65.4582).
The findings suggest that long-term follow-up of patients enrolled in clinical trials may provide clinically important additional information, and that limited-stage diffuse large B-cell lymphoma (DLBCL) may have a biology that is distinctly different from that of advanced stage DLBCL, the investigators wrote.
Initial results from the SWOG (Southwest Oncology Group) Study 8736, published after a median follow-up of 4.4 years, showed that CHOP3RT was associated with significantly better 5-year PFS and OS rates, compared with patients treated with CHOP8.
A second trial, SWOG Study 0014, looked at the addition of rituximab (Rituxan) to CHOP plus IFRT in patients with high-risk DLBCL with at least one adverse feature of the stage-modified International Prognostic Index. In this trial, after a median follow-up of 5.3 years, 4-year PFS was 88%, and 4-year OS was 92%.
5 years not enough
Results of both trials were published around the 5-year follow-up mark. However, an analysis of 10-year follow-up data from the S8736 trial, published only as an abstract (Blood. 98:724a-725a, 2001; abstr 3024), showed that “the survival curves for CHOP8 and CHOP3RT, surprisingly, began to overlap,” Dr. Stephens and her colleagues wrote.
“Additionally, for both S8736 and S0014, no plateau had been reached in the PFS curves. These data contrasted the expected cure rates for advanced-stage DLBCL, leading to the hypothesis that limited-stage DLBCL may have a unique biology from its advanced-stage counterpart,” they added.
Although more than half of patients with advanced-stage diffuse large B-cell lymphoma (DLBCL) can be cured with anthracycline-based chemotherapy, cure rates are markedly lower among patients with limited-stage disease, defined as Ann Arbor stage I or II, confined to a single irradiation field. Limited-stage disease accounts for approximately one-third DLBCL cases.
To see whether the trends observed at 10 years continued, the investigators conducted a survival analysis from S8736 and compared the data from similar patients in the S0014 study.
They found that after a median follow-up of 17.7 years in S8736, median PFS was 12.0 years for patients treated with CHOP8, and 11.1 years for patients treated with CHOP3RT, a difference that was not statistically significant.
Median OS was 13.0 and 13.7 years, respectively, and was also not significant.
In S0014, after a median follow-up of 12 years, the 5-year OS rate was 82%, and 10-year rate was 67%. In this trial, the investigators observed “a persistent pattern of relapse despite the addition of rituximab.”
“The populations were not entirely identical; however, even the addition of rituximab as per S0014 to combined-modality therapy did not seem to mitigate the continued relapse risk, underscoring the value of prolonged observation of clinical trial patients and possible unique biology of limited-stage DLBCL,” Dr. Stephens and her associates wrote.
The study was sponsored by grants from the National Institutes of Health. Dr. Stephens reported having no financial disclosures. Several coauthors disclosed research funding, consulting, speakers’ bureau participation or travel expenses from various oncology drug makers.
Long-term follow-up from two randomized trials shows that the treatment advantage initially seen by adding radiotherapy to the CHOP regimen in patients with limited-stage diffuse large B-cell lymphoma disappeared over time.
After a median follow-up of nearly 18 years, there were no significant differences in either progression-free survival (PFS) or overall survival between patients who had received standard CHOP chemotherapy (cyclophosphamide, doxorubicin, vincristine, and prednisone) for 8 cycles (CHOP8), or 3 cycles of CHOP with involved-field radiotherapy (CHOP3RT), reported Deborah M. Stephens, DO, of the University of Utah in Salt Lake City, and her colleagues.
The addition of rituximab to CHOP plus involved-field radiotherapy (IFRT) in a separate cohort did not appear to reduce the continued relapse risk, the investigators noted (J Clin Oncol. 2016 Jul. doi: 10.1200/JCO.2015.65.4582).
The findings suggest that long-term follow-up of patients enrolled in clinical trials may provide clinically important additional information, and that limited-stage diffuse large B-cell lymphoma (DLBCL) may have a biology that is distinctly different from that of advanced stage DLBCL, the investigators wrote.
Initial results from the SWOG (Southwest Oncology Group) Study 8736, published after a median follow-up of 4.4 years, showed that CHOP3RT was associated with significantly better 5-year PFS and OS rates, compared with patients treated with CHOP8.
A second trial, SWOG Study 0014, looked at the addition of rituximab (Rituxan) to CHOP plus IFRT in patients with high-risk DLBCL with at least one adverse feature of the stage-modified International Prognostic Index. In this trial, after a median follow-up of 5.3 years, 4-year PFS was 88%, and 4-year OS was 92%.
5 years not enough
Results of both trials were published around the 5-year follow-up mark. However, an analysis of 10-year follow-up data from the S8736 trial, published only as an abstract (Blood. 98:724a-725a, 2001; abstr 3024), showed that “the survival curves for CHOP8 and CHOP3RT, surprisingly, began to overlap,” Dr. Stephens and her colleagues wrote.
“Additionally, for both S8736 and S0014, no plateau had been reached in the PFS curves. These data contrasted the expected cure rates for advanced-stage DLBCL, leading to the hypothesis that limited-stage DLBCL may have a unique biology from its advanced-stage counterpart,” they added.
Although more than half of patients with advanced-stage diffuse large B-cell lymphoma (DLBCL) can be cured with anthracycline-based chemotherapy, cure rates are markedly lower among patients with limited-stage disease, defined as Ann Arbor stage I or II, confined to a single irradiation field. Limited-stage disease accounts for approximately one-third DLBCL cases.
To see whether the trends observed at 10 years continued, the investigators conducted a survival analysis from S8736 and compared the data from similar patients in the S0014 study.
They found that after a median follow-up of 17.7 years in S8736, median PFS was 12.0 years for patients treated with CHOP8, and 11.1 years for patients treated with CHOP3RT, a difference that was not statistically significant.
Median OS was 13.0 and 13.7 years, respectively, and was also not significant.
In S0014, after a median follow-up of 12 years, the 5-year OS rate was 82%, and 10-year rate was 67%. In this trial, the investigators observed “a persistent pattern of relapse despite the addition of rituximab.”
“The populations were not entirely identical; however, even the addition of rituximab as per S0014 to combined-modality therapy did not seem to mitigate the continued relapse risk, underscoring the value of prolonged observation of clinical trial patients and possible unique biology of limited-stage DLBCL,” Dr. Stephens and her associates wrote.
The study was sponsored by grants from the National Institutes of Health. Dr. Stephens reported having no financial disclosures. Several coauthors disclosed research funding, consulting, speakers’ bureau participation or travel expenses from various oncology drug makers.
FROM THE JOURNAL OF CLINICAL ONCOLOGY
Key clinical point: Long-term observation of clinical trial patients revealed a dwindling of initial survival benefit from radiation added to chemotherapy in limited-stage DLBCL.
Major finding: At a median 17.7 years of follow-up, there were no significant differences in progression-free or overall survival with eight cycles of CHOP or three cycles with IFRT.
Data source: Final analysis of randomized controlled trial in 401 patients with limited-stage diffuse large B-cell lymphoma.
Disclosures: The study was sponsored by grants from the National Institutes of Health. Dr. Stephens reported having no financial disclosures. Several coauthors disclosed research funding, consulting, speakers’ bureau participation or travel expenses from various oncology drug makers.
Bendamustine-based chemotherapy induces high CR rate in relapsed HL
Combination chemotherapy with bendamustine, gemcitabine, and vinorelbine can induce high complete response rates among patients with relapsed or refractory Hodgkin lymphoma (HL) who are candidates for autologous stem cell transplantation, final results of a phase II study show.
Among 59 patients enrolled in the multicenter study, 49 (83%) had responses, including 43 (73%) who achieved a complete response (CR), and 6 (10%) who had a partial response after 4 cycles of chemotherapy with bendamustine (Treanda), gemcitabine (Gemzar), and vinorelbine (BeGEV).
Of the 49 patients with responses, 43 went on to autologous stem cell transplant (ASCT), reported Armando Santoro, MD, from the Humanitas Cancer Center in Rozzano, Italy.
“These findings provide a strong rationale for further development of the BeGEV regimen,” the investigators wrote in a study published online in the Journal of Clinical Oncology (2016 Jul 5. doi: 10.1200/JCO.2016.66.4466).
The investigators had previously shown that a regimen of ifosfamide, gemcitabine, and vinorelbine (IEGV) as salvage chemotherapy prior to ASCT was associated with an 81% overall response rate (ORR) 54% CR rate.
Because bendamustine has shown good activity as monotherapy against relapsed/refractory HL, they conducted a phase II study with an IEGV-like regimen in which bendamustine would be substituted for ifosfamide, to determine whether the substitution could improve response rates.
They enrolled 59 consecutive patients aged 18 years and older with HL that had relapsed or was refractory to one previous line of chemotherapy. Patients were treated with gemcitabine 800 mg/m2 on days 1 and 4, vinorelbine 20 mg/m2 on day 1, and bendamustine 90 mg/m2 on days 2 and 3. Patients also received prednisolone 100 mg/day for days 1 to 4. The patients were treated for four 21-day cycles.
Patients who achieved either a complete or partial response after completion of the four planned cycles then underwent myeloablative therapy with carmustine or fotemustine plus etoposide, cytarabine, and melphalan, followed by reinfusion of mobilized CD34-positive circulating stem cells.
Grade 3 or 4 hematologic toxicities included thrombocytopenia and neutropenia, which occurred in eight patients each. Fifty-five of 57 total evaluable patients had successful mobilization and harvesting of CD34-positive cells, and, as noted before, 43 went on to ASCT.
After a median follow-up of 29 months, the 2-year progression-free survival (PFS) rate for the total population was 62%, and the overall survival rates was nearly 78%. For patients who went on to ASCT, the 2-year PFS rate was 81%, and the 2-year survival rate was 89%.
The authors noted that BeGV was associated with higher CR rates than IEGV, and with “excellent” stem cell mobilization activity and engraftment. Additionally, BeGV has a favorable toxicity profile, because of the absence of ifosfamide which is known to significantly increase risk for hemorrhagic cystitis.
“Because the number of novel agents that may be added in the pretransplantation therapy setting is growing, direct comparisons of combinations incorporating novel agents with BeGEV and other regimens will be necessary to identify the best salvage strategy for relapsed and refractory HL,” the authors wrote.
The study was supported in part by a grant from Mundipharma Pharmaceuticals. Two coauthors disclosed consulting/advisory roles and travel accommodations and expenses from the company.
Combination chemotherapy with bendamustine, gemcitabine, and vinorelbine can induce high complete response rates among patients with relapsed or refractory Hodgkin lymphoma (HL) who are candidates for autologous stem cell transplantation, final results of a phase II study show.
Among 59 patients enrolled in the multicenter study, 49 (83%) had responses, including 43 (73%) who achieved a complete response (CR), and 6 (10%) who had a partial response after 4 cycles of chemotherapy with bendamustine (Treanda), gemcitabine (Gemzar), and vinorelbine (BeGEV).
Of the 49 patients with responses, 43 went on to autologous stem cell transplant (ASCT), reported Armando Santoro, MD, from the Humanitas Cancer Center in Rozzano, Italy.
“These findings provide a strong rationale for further development of the BeGEV regimen,” the investigators wrote in a study published online in the Journal of Clinical Oncology (2016 Jul 5. doi: 10.1200/JCO.2016.66.4466).
The investigators had previously shown that a regimen of ifosfamide, gemcitabine, and vinorelbine (IEGV) as salvage chemotherapy prior to ASCT was associated with an 81% overall response rate (ORR) 54% CR rate.
Because bendamustine has shown good activity as monotherapy against relapsed/refractory HL, they conducted a phase II study with an IEGV-like regimen in which bendamustine would be substituted for ifosfamide, to determine whether the substitution could improve response rates.
They enrolled 59 consecutive patients aged 18 years and older with HL that had relapsed or was refractory to one previous line of chemotherapy. Patients were treated with gemcitabine 800 mg/m2 on days 1 and 4, vinorelbine 20 mg/m2 on day 1, and bendamustine 90 mg/m2 on days 2 and 3. Patients also received prednisolone 100 mg/day for days 1 to 4. The patients were treated for four 21-day cycles.
Patients who achieved either a complete or partial response after completion of the four planned cycles then underwent myeloablative therapy with carmustine or fotemustine plus etoposide, cytarabine, and melphalan, followed by reinfusion of mobilized CD34-positive circulating stem cells.
Grade 3 or 4 hematologic toxicities included thrombocytopenia and neutropenia, which occurred in eight patients each. Fifty-five of 57 total evaluable patients had successful mobilization and harvesting of CD34-positive cells, and, as noted before, 43 went on to ASCT.
After a median follow-up of 29 months, the 2-year progression-free survival (PFS) rate for the total population was 62%, and the overall survival rates was nearly 78%. For patients who went on to ASCT, the 2-year PFS rate was 81%, and the 2-year survival rate was 89%.
The authors noted that BeGV was associated with higher CR rates than IEGV, and with “excellent” stem cell mobilization activity and engraftment. Additionally, BeGV has a favorable toxicity profile, because of the absence of ifosfamide which is known to significantly increase risk for hemorrhagic cystitis.
“Because the number of novel agents that may be added in the pretransplantation therapy setting is growing, direct comparisons of combinations incorporating novel agents with BeGEV and other regimens will be necessary to identify the best salvage strategy for relapsed and refractory HL,” the authors wrote.
The study was supported in part by a grant from Mundipharma Pharmaceuticals. Two coauthors disclosed consulting/advisory roles and travel accommodations and expenses from the company.
Combination chemotherapy with bendamustine, gemcitabine, and vinorelbine can induce high complete response rates among patients with relapsed or refractory Hodgkin lymphoma (HL) who are candidates for autologous stem cell transplantation, final results of a phase II study show.
Among 59 patients enrolled in the multicenter study, 49 (83%) had responses, including 43 (73%) who achieved a complete response (CR), and 6 (10%) who had a partial response after 4 cycles of chemotherapy with bendamustine (Treanda), gemcitabine (Gemzar), and vinorelbine (BeGEV).
Of the 49 patients with responses, 43 went on to autologous stem cell transplant (ASCT), reported Armando Santoro, MD, from the Humanitas Cancer Center in Rozzano, Italy.
“These findings provide a strong rationale for further development of the BeGEV regimen,” the investigators wrote in a study published online in the Journal of Clinical Oncology (2016 Jul 5. doi: 10.1200/JCO.2016.66.4466).
The investigators had previously shown that a regimen of ifosfamide, gemcitabine, and vinorelbine (IEGV) as salvage chemotherapy prior to ASCT was associated with an 81% overall response rate (ORR) 54% CR rate.
Because bendamustine has shown good activity as monotherapy against relapsed/refractory HL, they conducted a phase II study with an IEGV-like regimen in which bendamustine would be substituted for ifosfamide, to determine whether the substitution could improve response rates.
They enrolled 59 consecutive patients aged 18 years and older with HL that had relapsed or was refractory to one previous line of chemotherapy. Patients were treated with gemcitabine 800 mg/m2 on days 1 and 4, vinorelbine 20 mg/m2 on day 1, and bendamustine 90 mg/m2 on days 2 and 3. Patients also received prednisolone 100 mg/day for days 1 to 4. The patients were treated for four 21-day cycles.
Patients who achieved either a complete or partial response after completion of the four planned cycles then underwent myeloablative therapy with carmustine or fotemustine plus etoposide, cytarabine, and melphalan, followed by reinfusion of mobilized CD34-positive circulating stem cells.
Grade 3 or 4 hematologic toxicities included thrombocytopenia and neutropenia, which occurred in eight patients each. Fifty-five of 57 total evaluable patients had successful mobilization and harvesting of CD34-positive cells, and, as noted before, 43 went on to ASCT.
After a median follow-up of 29 months, the 2-year progression-free survival (PFS) rate for the total population was 62%, and the overall survival rates was nearly 78%. For patients who went on to ASCT, the 2-year PFS rate was 81%, and the 2-year survival rate was 89%.
The authors noted that BeGV was associated with higher CR rates than IEGV, and with “excellent” stem cell mobilization activity and engraftment. Additionally, BeGV has a favorable toxicity profile, because of the absence of ifosfamide which is known to significantly increase risk for hemorrhagic cystitis.
“Because the number of novel agents that may be added in the pretransplantation therapy setting is growing, direct comparisons of combinations incorporating novel agents with BeGEV and other regimens will be necessary to identify the best salvage strategy for relapsed and refractory HL,” the authors wrote.
The study was supported in part by a grant from Mundipharma Pharmaceuticals. Two coauthors disclosed consulting/advisory roles and travel accommodations and expenses from the company.
FROM JCO
Key clinical point: Bendamustine in combination chemotherapy produces high response rates in relapsed refractory Hodgkin lymphoma (HL).
Major finding: The complete response rate with bendamustine, gemcitabine, and vinorelbine was 83%.
Data source: Multicenter phase II study of the BeGV salvage chemotherapy regimen in 59 patients with relapsed/refractory HL.
Disclosures: The study was supported in part by a grant from Mundipharma Pharmaceuticals. Two coauthors disclosed consulting/advisory roles and travel accommodations and expenses from the company.
Adding obinutuzumab to bendamustine boosts progression-free survival in rituximab-refractory indolent non-Hodgkin lymphoma
Obinutuzumab and bendamustine followed by obinutuzumab maintenance therapy was superior to bendamustine monotherapy based on progression-free survival in rituximab-refractory patients with indolent non-Hodgkin lymphoma, based on a study published online in the Lancet Oncology.
After a median follow-up of 22 months in the obinutuzumab plus bendamustine group and 20 months in the bendamustine monotherapy group, progression-free survival was significantly longer with obinutuzumab plus bendamustine (median not reached; 95% confidence interval, 22.5 months – not estimable) than with bendamustine monotherapy (14.9 months, range, 12.8-16.6; hazard ratio, 0.55; 95% CI 0.40-0.74; P = .0001). About two-thirds of the nearly 400 patients in both study arms had grade 3-5 adverse events.
The anti-CD20 monoclonal antibody obinutuzumab is an option when patients with indolent non-Hodgkin lymphoma relapse or don’t achieve adequate disease control with rituximab-based treatment, wrote Laurie H. Sehn, MD, of the British Columbia Cancer Agency and the University of British Columbia, Vancouver, and her colleagues.
In an open-label, randomized, phase III study called GADOLIN, patients with CD20-positive indolent non-Hodgkin lymphoma were stratified by indolent non-Hodgkin lymphoma subtype, rituximab-refractory type, number of previous therapies, and geographic region.
For the study, 194 patients were assigned to obinutuzumab plus bendamustine and 202 to bendamustine monotherapy. Trial participants received six 28-day cycles with either obinutuzumab plus bendamustine (obinutuzumab 1,000 mg on days 1, 8, and 15, cycle 1; and on day 1, cycles 2-6) plus bendamustine (90 mg/m2 per day on days 1 and 2, cycles 1-6) or bendamustine monotherapy (120 mg/m2 per day on days 1 and 2 of all cycles). Patients in the obinutuzumab plus bendamustine group whose disease did not progress received obinutuzumab maintenance therapy of 1,000 mg once every 2 months for up to 2 years.
Grade 3-5 adverse events occurred in 68% of 194 patients in the obinutuzumab plus bendamustine group and in 62% of 198 patients in the bendamustine monotherapy group. Grade 3 or worse neutropenia affected 33% of the obinutuzumab plus bendamustine group and 26% of the bendamustine monotherapy group. Other grade 3 or worse events included thrombocytopenia in 11% and 16%, anemia in 8% and 10%, and infusion-related reactions in 11% and 6%. Serious adverse events occurred in 38% in the obinutuzumab plus bendamustine group and in 33% in the bendamustine monotherapy group. Adverse events resulted in death in 6% of patients in each group.
The study was funded by Hoffmann-La Roche. Genentech, the maker of obinutuzumab (Gazyva) in the United States, is a wholly owned member of the Roche Group. Dr. Sehn receives honoraria and is a consultant or advisor to Genentech as well as several other drug companies.
On Twitter @maryjodales
Obinutuzumab and bendamustine followed by obinutuzumab maintenance therapy was superior to bendamustine monotherapy based on progression-free survival in rituximab-refractory patients with indolent non-Hodgkin lymphoma, based on a study published online in the Lancet Oncology.
After a median follow-up of 22 months in the obinutuzumab plus bendamustine group and 20 months in the bendamustine monotherapy group, progression-free survival was significantly longer with obinutuzumab plus bendamustine (median not reached; 95% confidence interval, 22.5 months – not estimable) than with bendamustine monotherapy (14.9 months, range, 12.8-16.6; hazard ratio, 0.55; 95% CI 0.40-0.74; P = .0001). About two-thirds of the nearly 400 patients in both study arms had grade 3-5 adverse events.
The anti-CD20 monoclonal antibody obinutuzumab is an option when patients with indolent non-Hodgkin lymphoma relapse or don’t achieve adequate disease control with rituximab-based treatment, wrote Laurie H. Sehn, MD, of the British Columbia Cancer Agency and the University of British Columbia, Vancouver, and her colleagues.
In an open-label, randomized, phase III study called GADOLIN, patients with CD20-positive indolent non-Hodgkin lymphoma were stratified by indolent non-Hodgkin lymphoma subtype, rituximab-refractory type, number of previous therapies, and geographic region.
For the study, 194 patients were assigned to obinutuzumab plus bendamustine and 202 to bendamustine monotherapy. Trial participants received six 28-day cycles with either obinutuzumab plus bendamustine (obinutuzumab 1,000 mg on days 1, 8, and 15, cycle 1; and on day 1, cycles 2-6) plus bendamustine (90 mg/m2 per day on days 1 and 2, cycles 1-6) or bendamustine monotherapy (120 mg/m2 per day on days 1 and 2 of all cycles). Patients in the obinutuzumab plus bendamustine group whose disease did not progress received obinutuzumab maintenance therapy of 1,000 mg once every 2 months for up to 2 years.
Grade 3-5 adverse events occurred in 68% of 194 patients in the obinutuzumab plus bendamustine group and in 62% of 198 patients in the bendamustine monotherapy group. Grade 3 or worse neutropenia affected 33% of the obinutuzumab plus bendamustine group and 26% of the bendamustine monotherapy group. Other grade 3 or worse events included thrombocytopenia in 11% and 16%, anemia in 8% and 10%, and infusion-related reactions in 11% and 6%. Serious adverse events occurred in 38% in the obinutuzumab plus bendamustine group and in 33% in the bendamustine monotherapy group. Adverse events resulted in death in 6% of patients in each group.
The study was funded by Hoffmann-La Roche. Genentech, the maker of obinutuzumab (Gazyva) in the United States, is a wholly owned member of the Roche Group. Dr. Sehn receives honoraria and is a consultant or advisor to Genentech as well as several other drug companies.
On Twitter @maryjodales
Obinutuzumab and bendamustine followed by obinutuzumab maintenance therapy was superior to bendamustine monotherapy based on progression-free survival in rituximab-refractory patients with indolent non-Hodgkin lymphoma, based on a study published online in the Lancet Oncology.
After a median follow-up of 22 months in the obinutuzumab plus bendamustine group and 20 months in the bendamustine monotherapy group, progression-free survival was significantly longer with obinutuzumab plus bendamustine (median not reached; 95% confidence interval, 22.5 months – not estimable) than with bendamustine monotherapy (14.9 months, range, 12.8-16.6; hazard ratio, 0.55; 95% CI 0.40-0.74; P = .0001). About two-thirds of the nearly 400 patients in both study arms had grade 3-5 adverse events.
The anti-CD20 monoclonal antibody obinutuzumab is an option when patients with indolent non-Hodgkin lymphoma relapse or don’t achieve adequate disease control with rituximab-based treatment, wrote Laurie H. Sehn, MD, of the British Columbia Cancer Agency and the University of British Columbia, Vancouver, and her colleagues.
In an open-label, randomized, phase III study called GADOLIN, patients with CD20-positive indolent non-Hodgkin lymphoma were stratified by indolent non-Hodgkin lymphoma subtype, rituximab-refractory type, number of previous therapies, and geographic region.
For the study, 194 patients were assigned to obinutuzumab plus bendamustine and 202 to bendamustine monotherapy. Trial participants received six 28-day cycles with either obinutuzumab plus bendamustine (obinutuzumab 1,000 mg on days 1, 8, and 15, cycle 1; and on day 1, cycles 2-6) plus bendamustine (90 mg/m2 per day on days 1 and 2, cycles 1-6) or bendamustine monotherapy (120 mg/m2 per day on days 1 and 2 of all cycles). Patients in the obinutuzumab plus bendamustine group whose disease did not progress received obinutuzumab maintenance therapy of 1,000 mg once every 2 months for up to 2 years.
Grade 3-5 adverse events occurred in 68% of 194 patients in the obinutuzumab plus bendamustine group and in 62% of 198 patients in the bendamustine monotherapy group. Grade 3 or worse neutropenia affected 33% of the obinutuzumab plus bendamustine group and 26% of the bendamustine monotherapy group. Other grade 3 or worse events included thrombocytopenia in 11% and 16%, anemia in 8% and 10%, and infusion-related reactions in 11% and 6%. Serious adverse events occurred in 38% in the obinutuzumab plus bendamustine group and in 33% in the bendamustine monotherapy group. Adverse events resulted in death in 6% of patients in each group.
The study was funded by Hoffmann-La Roche. Genentech, the maker of obinutuzumab (Gazyva) in the United States, is a wholly owned member of the Roche Group. Dr. Sehn receives honoraria and is a consultant or advisor to Genentech as well as several other drug companies.
On Twitter @maryjodales
FROM THE LANCET ONCOLOGY
Key clinical point: Obinutuzumab is an option when patients with indolent non-Hodgkin lymphoma relapse or don’t achieve adequate disease control with rituximab-based treatment.
Major finding: Progression-free survival was significantly longer with obinutuzumab plus bendamustine (median not reached; 95% CI, 22.5 months – not estimable) than with bendamustine monotherapy (14.9 months, range,12.8-16.6 months; hazard ratio, 0.55; 95% CI, 0.40-0.74; P = ·0001).
Data source: An open-label, randomized, phase III study of 396 patients.
Disclosures: The study was funded by Hoffmann-La Roche. Genentech, the maker of obinutuzumab (Gazyva) in the United States, is a wholly owned member of the Roche Group. Dr. Sehn receives honoraria and is a consultant or adviser to Genentech as well as several other drug companies.
Follicular lymphoma with histologic transformation may merit ASCT
Among patients with high tumor burden follicular lymphoma (FL) that responded to rituximab chemotherapy but then underwent histologic transformation, median overall survival was not reached when patients received autologous stem cell transplantation (ASCT), but was only 1.7 years otherwise, based on results of an ancillary study of a clinical trial.
In contrast, ASCT did not affect overall survival when patients progressed to untransformed FL, said Dr. Clémentine Sarkozy of Centre Hospitalier Lyon-Sud in Pierre Bénite, France, and her associates. Fully 58% of histologic transformations occurred in the first year of follow-up, highlighting “the necessity for biopsy at the first recurrence of FL,” they wrote online June 13 in the Journal of Clinical Oncology.
Histologic transformation in FL signifies progression to aggressive lymphoma. Studies of histologic transformation and subsequent overall survival in the rituximab era have been retrospective, with variable patient populations and initial management regimens, according to the investigators. Therefore, they followed 1,018 patients from the multicenter, randomized, phase III PRIMA (Primary Rituximab and Maintenance) trial, which evaluated maintenance rituximab therapy among patients with symptomatic FL who had responded to induction chemotherapy plus rituximab (J Clin Oncol. 2016 Jun. doi: 10.1200/JCO.2015.65.7163).
A total of 463 patients (45.5%) experienced disease recurrence or progression, and 194 (42%) were biopsied over a median follow-up time of 6 years. A total of 40 (20.6%) biopsies showed histologic transformation, while 154 (79.4%) had untransformed FL. Median time to recurrence was 9.6 months for patients with histologic transformation and 22.8 months for patients with untransformed FL (P = .02). Median overall survival with histologic transformation was worse than with untransformed FL (3.8 years vs. 6.4 years; hazard ratio, 3.9; 95% confidence interval, 2.2-6.9; P = .001). Furthermore, among patients who progressed within 12 months, median overall survival with histologic transformation was 2 years, compared with 6.4 years for patients with untransformed FL (P = .007).
After salvage therapy, 17 (42%) patients with histologic transformation underwent consolidation with high-dose chemotherapy and ASCT. Median overall survival for these patients was not reached, versus 1.7 years when they did not undergo ASCT. In contrast, ASCT did not improve overall survival among patients with untransformed FL. Results were similar after excluding patients with early progression and patients who were older than 65 years, the investigators reported.
Risk factors for histologic transformation in the univariate analysis included performance status, anemia, high lactate dehydrogenase level, “B” symptoms, histologic grade 3a, and high Follicular Lymphoma International Prognostic Index scores at diagnosis. However, only Eastern Cooperative Oncology Group performance status of 2 to 4 (HR, 5.6; 95% CI, 1.7-17.7), and anemia (HR, 3.7; 95% CI, 1.4-9.7) remained significant in the multivariate analysis. Neither the choice of induction regimen nor the quality of response seemed to affect the likelihood of histologic transformation, and rituximab maintenance therapy did not seem to alter response to salvage treatment or survival after histologic transformation. By necessity, the study excluded patients who did not respond to initial immunochemotherapy, which could have limited the generalizability of the findings, the investigators noted.
The study was funded by Sandoz and Takeda Pharmaceuticals. Dr. Sarkozy disclosed research funding from Sandoz and Takeda Pharmaceuticals and honoraria from Gilead Sciences. Twelve coinvestigators also disclosed ties to Takeda and a number of other pharmaceutical companies. The other seven coinvestigators had no disclosures.
In just 3 years, prospective observational studies and [this] clever ancillary analysis of a prospective clinical trial have better informed the lymphoma community about the expected incidence and timing of transformation in patients with follicular lymphoma after being treated with modern management strategies. But we are still limited by clumsy predictive tools for identifying patients at highest risk. Deeper understanding of biologic and genetic factors of FL subclonal populations as well as the tumor microenvironment will allow for more precise identification of patients truly at risk and potentially will provide actionable targets for abrogating that risk. Future [studies of] transformed lymphoma will hopefully replace variables such as anthracyclines, the Follicular Lymphoma International Prognostic Index, lactate dehydrogenase, and ASCT with promising new variables such as IRF-4, miR-31, bcl-2, pleuripotency, and nuclear factor kappa B pathway genes or new therapies that target these variables. Future analyses should not simply prognosticate who is at risk for transformation, but should predict a specific intervention to either prevent or treat such an event.
Dr. Brian K. Link is at the University of Iowa, Iowa City. He reported ties to AbbVie, Gilead Sciences, Genentech, Sandoz, Pharmacyclics, Millennium Pharmaceuticals, Genentech, Kite Pharma, Seattle Genetics, and Dynavax Technologies. These comments are from his editorial accompanying the report (J Clin Oncol. 2016 Jun. doi: 10.1200/JCO.2016.67.4234).
In just 3 years, prospective observational studies and [this] clever ancillary analysis of a prospective clinical trial have better informed the lymphoma community about the expected incidence and timing of transformation in patients with follicular lymphoma after being treated with modern management strategies. But we are still limited by clumsy predictive tools for identifying patients at highest risk. Deeper understanding of biologic and genetic factors of FL subclonal populations as well as the tumor microenvironment will allow for more precise identification of patients truly at risk and potentially will provide actionable targets for abrogating that risk. Future [studies of] transformed lymphoma will hopefully replace variables such as anthracyclines, the Follicular Lymphoma International Prognostic Index, lactate dehydrogenase, and ASCT with promising new variables such as IRF-4, miR-31, bcl-2, pleuripotency, and nuclear factor kappa B pathway genes or new therapies that target these variables. Future analyses should not simply prognosticate who is at risk for transformation, but should predict a specific intervention to either prevent or treat such an event.
Dr. Brian K. Link is at the University of Iowa, Iowa City. He reported ties to AbbVie, Gilead Sciences, Genentech, Sandoz, Pharmacyclics, Millennium Pharmaceuticals, Genentech, Kite Pharma, Seattle Genetics, and Dynavax Technologies. These comments are from his editorial accompanying the report (J Clin Oncol. 2016 Jun. doi: 10.1200/JCO.2016.67.4234).
In just 3 years, prospective observational studies and [this] clever ancillary analysis of a prospective clinical trial have better informed the lymphoma community about the expected incidence and timing of transformation in patients with follicular lymphoma after being treated with modern management strategies. But we are still limited by clumsy predictive tools for identifying patients at highest risk. Deeper understanding of biologic and genetic factors of FL subclonal populations as well as the tumor microenvironment will allow for more precise identification of patients truly at risk and potentially will provide actionable targets for abrogating that risk. Future [studies of] transformed lymphoma will hopefully replace variables such as anthracyclines, the Follicular Lymphoma International Prognostic Index, lactate dehydrogenase, and ASCT with promising new variables such as IRF-4, miR-31, bcl-2, pleuripotency, and nuclear factor kappa B pathway genes or new therapies that target these variables. Future analyses should not simply prognosticate who is at risk for transformation, but should predict a specific intervention to either prevent or treat such an event.
Dr. Brian K. Link is at the University of Iowa, Iowa City. He reported ties to AbbVie, Gilead Sciences, Genentech, Sandoz, Pharmacyclics, Millennium Pharmaceuticals, Genentech, Kite Pharma, Seattle Genetics, and Dynavax Technologies. These comments are from his editorial accompanying the report (J Clin Oncol. 2016 Jun. doi: 10.1200/JCO.2016.67.4234).
Among patients with high tumor burden follicular lymphoma (FL) that responded to rituximab chemotherapy but then underwent histologic transformation, median overall survival was not reached when patients received autologous stem cell transplantation (ASCT), but was only 1.7 years otherwise, based on results of an ancillary study of a clinical trial.
In contrast, ASCT did not affect overall survival when patients progressed to untransformed FL, said Dr. Clémentine Sarkozy of Centre Hospitalier Lyon-Sud in Pierre Bénite, France, and her associates. Fully 58% of histologic transformations occurred in the first year of follow-up, highlighting “the necessity for biopsy at the first recurrence of FL,” they wrote online June 13 in the Journal of Clinical Oncology.
Histologic transformation in FL signifies progression to aggressive lymphoma. Studies of histologic transformation and subsequent overall survival in the rituximab era have been retrospective, with variable patient populations and initial management regimens, according to the investigators. Therefore, they followed 1,018 patients from the multicenter, randomized, phase III PRIMA (Primary Rituximab and Maintenance) trial, which evaluated maintenance rituximab therapy among patients with symptomatic FL who had responded to induction chemotherapy plus rituximab (J Clin Oncol. 2016 Jun. doi: 10.1200/JCO.2015.65.7163).
A total of 463 patients (45.5%) experienced disease recurrence or progression, and 194 (42%) were biopsied over a median follow-up time of 6 years. A total of 40 (20.6%) biopsies showed histologic transformation, while 154 (79.4%) had untransformed FL. Median time to recurrence was 9.6 months for patients with histologic transformation and 22.8 months for patients with untransformed FL (P = .02). Median overall survival with histologic transformation was worse than with untransformed FL (3.8 years vs. 6.4 years; hazard ratio, 3.9; 95% confidence interval, 2.2-6.9; P = .001). Furthermore, among patients who progressed within 12 months, median overall survival with histologic transformation was 2 years, compared with 6.4 years for patients with untransformed FL (P = .007).
After salvage therapy, 17 (42%) patients with histologic transformation underwent consolidation with high-dose chemotherapy and ASCT. Median overall survival for these patients was not reached, versus 1.7 years when they did not undergo ASCT. In contrast, ASCT did not improve overall survival among patients with untransformed FL. Results were similar after excluding patients with early progression and patients who were older than 65 years, the investigators reported.
Risk factors for histologic transformation in the univariate analysis included performance status, anemia, high lactate dehydrogenase level, “B” symptoms, histologic grade 3a, and high Follicular Lymphoma International Prognostic Index scores at diagnosis. However, only Eastern Cooperative Oncology Group performance status of 2 to 4 (HR, 5.6; 95% CI, 1.7-17.7), and anemia (HR, 3.7; 95% CI, 1.4-9.7) remained significant in the multivariate analysis. Neither the choice of induction regimen nor the quality of response seemed to affect the likelihood of histologic transformation, and rituximab maintenance therapy did not seem to alter response to salvage treatment or survival after histologic transformation. By necessity, the study excluded patients who did not respond to initial immunochemotherapy, which could have limited the generalizability of the findings, the investigators noted.
The study was funded by Sandoz and Takeda Pharmaceuticals. Dr. Sarkozy disclosed research funding from Sandoz and Takeda Pharmaceuticals and honoraria from Gilead Sciences. Twelve coinvestigators also disclosed ties to Takeda and a number of other pharmaceutical companies. The other seven coinvestigators had no disclosures.
Among patients with high tumor burden follicular lymphoma (FL) that responded to rituximab chemotherapy but then underwent histologic transformation, median overall survival was not reached when patients received autologous stem cell transplantation (ASCT), but was only 1.7 years otherwise, based on results of an ancillary study of a clinical trial.
In contrast, ASCT did not affect overall survival when patients progressed to untransformed FL, said Dr. Clémentine Sarkozy of Centre Hospitalier Lyon-Sud in Pierre Bénite, France, and her associates. Fully 58% of histologic transformations occurred in the first year of follow-up, highlighting “the necessity for biopsy at the first recurrence of FL,” they wrote online June 13 in the Journal of Clinical Oncology.
Histologic transformation in FL signifies progression to aggressive lymphoma. Studies of histologic transformation and subsequent overall survival in the rituximab era have been retrospective, with variable patient populations and initial management regimens, according to the investigators. Therefore, they followed 1,018 patients from the multicenter, randomized, phase III PRIMA (Primary Rituximab and Maintenance) trial, which evaluated maintenance rituximab therapy among patients with symptomatic FL who had responded to induction chemotherapy plus rituximab (J Clin Oncol. 2016 Jun. doi: 10.1200/JCO.2015.65.7163).
A total of 463 patients (45.5%) experienced disease recurrence or progression, and 194 (42%) were biopsied over a median follow-up time of 6 years. A total of 40 (20.6%) biopsies showed histologic transformation, while 154 (79.4%) had untransformed FL. Median time to recurrence was 9.6 months for patients with histologic transformation and 22.8 months for patients with untransformed FL (P = .02). Median overall survival with histologic transformation was worse than with untransformed FL (3.8 years vs. 6.4 years; hazard ratio, 3.9; 95% confidence interval, 2.2-6.9; P = .001). Furthermore, among patients who progressed within 12 months, median overall survival with histologic transformation was 2 years, compared with 6.4 years for patients with untransformed FL (P = .007).
After salvage therapy, 17 (42%) patients with histologic transformation underwent consolidation with high-dose chemotherapy and ASCT. Median overall survival for these patients was not reached, versus 1.7 years when they did not undergo ASCT. In contrast, ASCT did not improve overall survival among patients with untransformed FL. Results were similar after excluding patients with early progression and patients who were older than 65 years, the investigators reported.
Risk factors for histologic transformation in the univariate analysis included performance status, anemia, high lactate dehydrogenase level, “B” symptoms, histologic grade 3a, and high Follicular Lymphoma International Prognostic Index scores at diagnosis. However, only Eastern Cooperative Oncology Group performance status of 2 to 4 (HR, 5.6; 95% CI, 1.7-17.7), and anemia (HR, 3.7; 95% CI, 1.4-9.7) remained significant in the multivariate analysis. Neither the choice of induction regimen nor the quality of response seemed to affect the likelihood of histologic transformation, and rituximab maintenance therapy did not seem to alter response to salvage treatment or survival after histologic transformation. By necessity, the study excluded patients who did not respond to initial immunochemotherapy, which could have limited the generalizability of the findings, the investigators noted.
The study was funded by Sandoz and Takeda Pharmaceuticals. Dr. Sarkozy disclosed research funding from Sandoz and Takeda Pharmaceuticals and honoraria from Gilead Sciences. Twelve coinvestigators also disclosed ties to Takeda and a number of other pharmaceutical companies. The other seven coinvestigators had no disclosures.
FROM THE JOURNAL OF CLINICAL ONCOLOGY
Key clinical point: Histologic transformation of follicular lymphoma tends to occur early and may merit intensive salvage with autologous stem cell transplantation.
Major finding: Median overall survival was not reached among patients who received ASCT and was 1.7 years in those who didn’t have ASCT.
Data source: A study of 1,018 patients from the multicenter, randomized, phase III PRIMA (Primary Rituximab and Maintenance) trial.
Disclosures: The study was funded by Sandoz and Takeda Pharmaceuticals. Dr. Sarkozy disclosed research funding from Sandoz and Takeda Pharmaceuticals and honoraria from Gilead Sciences. Twelve coinvestigators also disclosed ties to Takeda and a number of other pharmaceutical companies. The other seven coinvestigators had no disclosures.
Brentuximab vedotin boosted PET-negative rate in Hodgkin
CHICAGO – Brentuximab vedotin appears to be safe and effective in eradicating residual disease after induction chemotherapy and may replace radiation for consolidation in patients with limited stage non-bulky Hodgkin lymphoma, Dr. Steven I. Park reported at the annual meeting of the American Society of Clinical Oncology.
After two cycles of ABVD [doxorubicin (Adriamycin), bleomycin, vinblastine, and dacarbazine], 72% of 40 evaluable patients achieved PET-negative disease. After completing brentuximab vedotin therapy, 90% of patients had PET-negative disease. With a median follow-up of 12 months, the estimated 1-year progression-free survival rate is 91%, and the overall survival rate is 96%.
The current standard therapy for limited stage Hodgkin lymphoma is about 4-6 cycles of chemotherapy with or without consolidative radiation therapy. The goal of the study was to reduce the number of cycles of chemotherapy and avoid radiation therapy, which has an unclear overall survival advantage and risks long-term side effects, noted Dr. Park of the University of North Carolina at Chapel Hill, Lineberger Comprehensive Cancer Center.
In this phase II multicenter study, 41 patients with previously untreated limited stage non-bulky Hodgkin lymphoma received ABVD followed by brentuximab vedotin (NCT01578967). Study patients’ median age was 29 years (range 19-67), and 46% presented with unfavorable disease. Over 90% of patients received four or fewer cycles of ABVD, and one patient received radiation due to disease progression.
Grade 3 or higher toxicities associated with brentuximab vedotin included neutropenia in three patients and peripheral neuropathy and rash in one patient each. One patient developed pancreatitis and died due to sepsis and hepatic failure, a rare complication of brentuximab vedotin that cautions regarding its use in patients with hepatic function limitations, Dr. Park said.
According to Seattle Genetics, the maker of brentuximab vedotin, the drug is an anti-CD30 monoclonal antibody attached by a protease-cleavable linker to the cytotoxic agent monomethyl auristatin E, which leads to target cell death when internalized into CD30-expressing tumor cells.
Dr. Park disclosed research funding from Seattle Genetics, the maker of brentuximab vedotin, as well as Teva.
On Twitter @maryjodales
CHICAGO – Brentuximab vedotin appears to be safe and effective in eradicating residual disease after induction chemotherapy and may replace radiation for consolidation in patients with limited stage non-bulky Hodgkin lymphoma, Dr. Steven I. Park reported at the annual meeting of the American Society of Clinical Oncology.
After two cycles of ABVD [doxorubicin (Adriamycin), bleomycin, vinblastine, and dacarbazine], 72% of 40 evaluable patients achieved PET-negative disease. After completing brentuximab vedotin therapy, 90% of patients had PET-negative disease. With a median follow-up of 12 months, the estimated 1-year progression-free survival rate is 91%, and the overall survival rate is 96%.
The current standard therapy for limited stage Hodgkin lymphoma is about 4-6 cycles of chemotherapy with or without consolidative radiation therapy. The goal of the study was to reduce the number of cycles of chemotherapy and avoid radiation therapy, which has an unclear overall survival advantage and risks long-term side effects, noted Dr. Park of the University of North Carolina at Chapel Hill, Lineberger Comprehensive Cancer Center.
In this phase II multicenter study, 41 patients with previously untreated limited stage non-bulky Hodgkin lymphoma received ABVD followed by brentuximab vedotin (NCT01578967). Study patients’ median age was 29 years (range 19-67), and 46% presented with unfavorable disease. Over 90% of patients received four or fewer cycles of ABVD, and one patient received radiation due to disease progression.
Grade 3 or higher toxicities associated with brentuximab vedotin included neutropenia in three patients and peripheral neuropathy and rash in one patient each. One patient developed pancreatitis and died due to sepsis and hepatic failure, a rare complication of brentuximab vedotin that cautions regarding its use in patients with hepatic function limitations, Dr. Park said.
According to Seattle Genetics, the maker of brentuximab vedotin, the drug is an anti-CD30 monoclonal antibody attached by a protease-cleavable linker to the cytotoxic agent monomethyl auristatin E, which leads to target cell death when internalized into CD30-expressing tumor cells.
Dr. Park disclosed research funding from Seattle Genetics, the maker of brentuximab vedotin, as well as Teva.
On Twitter @maryjodales
CHICAGO – Brentuximab vedotin appears to be safe and effective in eradicating residual disease after induction chemotherapy and may replace radiation for consolidation in patients with limited stage non-bulky Hodgkin lymphoma, Dr. Steven I. Park reported at the annual meeting of the American Society of Clinical Oncology.
After two cycles of ABVD [doxorubicin (Adriamycin), bleomycin, vinblastine, and dacarbazine], 72% of 40 evaluable patients achieved PET-negative disease. After completing brentuximab vedotin therapy, 90% of patients had PET-negative disease. With a median follow-up of 12 months, the estimated 1-year progression-free survival rate is 91%, and the overall survival rate is 96%.
The current standard therapy for limited stage Hodgkin lymphoma is about 4-6 cycles of chemotherapy with or without consolidative radiation therapy. The goal of the study was to reduce the number of cycles of chemotherapy and avoid radiation therapy, which has an unclear overall survival advantage and risks long-term side effects, noted Dr. Park of the University of North Carolina at Chapel Hill, Lineberger Comprehensive Cancer Center.
In this phase II multicenter study, 41 patients with previously untreated limited stage non-bulky Hodgkin lymphoma received ABVD followed by brentuximab vedotin (NCT01578967). Study patients’ median age was 29 years (range 19-67), and 46% presented with unfavorable disease. Over 90% of patients received four or fewer cycles of ABVD, and one patient received radiation due to disease progression.
Grade 3 or higher toxicities associated with brentuximab vedotin included neutropenia in three patients and peripheral neuropathy and rash in one patient each. One patient developed pancreatitis and died due to sepsis and hepatic failure, a rare complication of brentuximab vedotin that cautions regarding its use in patients with hepatic function limitations, Dr. Park said.
According to Seattle Genetics, the maker of brentuximab vedotin, the drug is an anti-CD30 monoclonal antibody attached by a protease-cleavable linker to the cytotoxic agent monomethyl auristatin E, which leads to target cell death when internalized into CD30-expressing tumor cells.
Dr. Park disclosed research funding from Seattle Genetics, the maker of brentuximab vedotin, as well as Teva.
On Twitter @maryjodales
AT THE 2016 ASCO ANNUAL MEETING
Key clinical point: Brentuximab vedotin appears to eradicate residual disease after induction chemotherapy in a small study of patients with limited stage non-bulky Hodgkin lymphoma.
Major finding: In 40 evaluable patients, 72% were PET-negative after two cycles of ABVD; brentuximab vedotin consolidation boosted PET-negative status to 90% of patients.
Data source: A phase II multicenter study of 41 patients with previously untreated limited stage non-bulky Hodgkin lymphoma.
Disclosures: Dr. Park disclosed research funding from Seattle Genetics, the maker of brentuximab vedotin, as well as Teva.
Enzastaurin flops as maintenance in treated DLBCL
Maintenance therapy with enzastaurin, an experimental agent directed against B-cell malignancies, did not improve disease-free survival among patients with high-risk diffuse large B-cell lymphomas following complete responses to chemotherapy with rituximab.
In a randomized, double-blind trial, after a median follow-up of 48 months, the hazard ratio for disease-free survival with enzastaurin vs. placebo, the primary endpoint, was 0.92 (P = .541), reported Dr. Michael Crump of Princess Margaret Cancer Centre in Toronto, and colleagues.
“The risk of treatment failure, however defined, is likely to be different in the subpopulation of patients who achieve remission after that treatment. Furthermore, identifying the value of specific biomarkers in predicting therapeutic response to novel targeted agents may be necessary in guiding future trials within defined subgroups of patients with DLBCL,” they wrote in the study, published online May 23 in Journal of Clinical Oncology.
Enzastaurin is a selective inhibitor of the protein kinase C-beta isoform (PKC-beta) expressed in both normal and malignant B cells. It has been shown to decrease tumor proliferation and induced apoptosis in cancer cells, and has been shown to have activity against relapsed or refractory DLBCL, mantle cell lymphoma, and follicular lymphoma, the authors explained.
Dr. Crump and colleagues conducted a phase III study to determine whether enzastaurin could be effective as maintenance therapy in patients with DLBCL at high risk for relapse after having had complete responses to first-line therapy with rituximab, cyclophosphamide, doxorubicin, vincristine, and prednisone (R-CHOP).
They enrolled 758 patients with stage II bulky DLBCL or stage III-IV disease who had three or more International Prognostic Index risk factors at diagnosis, and who had achieved either a confirmed or unconfirmed complete response after six to eight cycles of R-CHOP.
The patients were randomly assigned on a 2:1 basis to receive either oral enzastaurin 500 mg daily or placebo for 3 years, or until disease progression or unacceptable toxicity,
As noted, there was no significant difference in DFS with the active drug vs. placebo. In addition, in correlative analyses looking for biomarkers of response by cell of origin (i.e., germinal-center or non–germinal-center B cell) or by PKC-beta protein expression, the authors found no significant associations with either DFS or overall survival.
Enzastaurin was generally safe, with minor and manageable adverse events. More patients in the enzastaurin arm had episodes of QTc prolongation, but these did not require discontinuation of the drug.
Dr. Crump and coauthors disclosed consulting, advising, research funding and other relationships with various companies, including Eli Lilly. Five coauthors are Lilly employees.
Maintenance therapy with enzastaurin, an experimental agent directed against B-cell malignancies, did not improve disease-free survival among patients with high-risk diffuse large B-cell lymphomas following complete responses to chemotherapy with rituximab.
In a randomized, double-blind trial, after a median follow-up of 48 months, the hazard ratio for disease-free survival with enzastaurin vs. placebo, the primary endpoint, was 0.92 (P = .541), reported Dr. Michael Crump of Princess Margaret Cancer Centre in Toronto, and colleagues.
“The risk of treatment failure, however defined, is likely to be different in the subpopulation of patients who achieve remission after that treatment. Furthermore, identifying the value of specific biomarkers in predicting therapeutic response to novel targeted agents may be necessary in guiding future trials within defined subgroups of patients with DLBCL,” they wrote in the study, published online May 23 in Journal of Clinical Oncology.
Enzastaurin is a selective inhibitor of the protein kinase C-beta isoform (PKC-beta) expressed in both normal and malignant B cells. It has been shown to decrease tumor proliferation and induced apoptosis in cancer cells, and has been shown to have activity against relapsed or refractory DLBCL, mantle cell lymphoma, and follicular lymphoma, the authors explained.
Dr. Crump and colleagues conducted a phase III study to determine whether enzastaurin could be effective as maintenance therapy in patients with DLBCL at high risk for relapse after having had complete responses to first-line therapy with rituximab, cyclophosphamide, doxorubicin, vincristine, and prednisone (R-CHOP).
They enrolled 758 patients with stage II bulky DLBCL or stage III-IV disease who had three or more International Prognostic Index risk factors at diagnosis, and who had achieved either a confirmed or unconfirmed complete response after six to eight cycles of R-CHOP.
The patients were randomly assigned on a 2:1 basis to receive either oral enzastaurin 500 mg daily or placebo for 3 years, or until disease progression or unacceptable toxicity,
As noted, there was no significant difference in DFS with the active drug vs. placebo. In addition, in correlative analyses looking for biomarkers of response by cell of origin (i.e., germinal-center or non–germinal-center B cell) or by PKC-beta protein expression, the authors found no significant associations with either DFS or overall survival.
Enzastaurin was generally safe, with minor and manageable adverse events. More patients in the enzastaurin arm had episodes of QTc prolongation, but these did not require discontinuation of the drug.
Dr. Crump and coauthors disclosed consulting, advising, research funding and other relationships with various companies, including Eli Lilly. Five coauthors are Lilly employees.
Maintenance therapy with enzastaurin, an experimental agent directed against B-cell malignancies, did not improve disease-free survival among patients with high-risk diffuse large B-cell lymphomas following complete responses to chemotherapy with rituximab.
In a randomized, double-blind trial, after a median follow-up of 48 months, the hazard ratio for disease-free survival with enzastaurin vs. placebo, the primary endpoint, was 0.92 (P = .541), reported Dr. Michael Crump of Princess Margaret Cancer Centre in Toronto, and colleagues.
“The risk of treatment failure, however defined, is likely to be different in the subpopulation of patients who achieve remission after that treatment. Furthermore, identifying the value of specific biomarkers in predicting therapeutic response to novel targeted agents may be necessary in guiding future trials within defined subgroups of patients with DLBCL,” they wrote in the study, published online May 23 in Journal of Clinical Oncology.
Enzastaurin is a selective inhibitor of the protein kinase C-beta isoform (PKC-beta) expressed in both normal and malignant B cells. It has been shown to decrease tumor proliferation and induced apoptosis in cancer cells, and has been shown to have activity against relapsed or refractory DLBCL, mantle cell lymphoma, and follicular lymphoma, the authors explained.
Dr. Crump and colleagues conducted a phase III study to determine whether enzastaurin could be effective as maintenance therapy in patients with DLBCL at high risk for relapse after having had complete responses to first-line therapy with rituximab, cyclophosphamide, doxorubicin, vincristine, and prednisone (R-CHOP).
They enrolled 758 patients with stage II bulky DLBCL or stage III-IV disease who had three or more International Prognostic Index risk factors at diagnosis, and who had achieved either a confirmed or unconfirmed complete response after six to eight cycles of R-CHOP.
The patients were randomly assigned on a 2:1 basis to receive either oral enzastaurin 500 mg daily or placebo for 3 years, or until disease progression or unacceptable toxicity,
As noted, there was no significant difference in DFS with the active drug vs. placebo. In addition, in correlative analyses looking for biomarkers of response by cell of origin (i.e., germinal-center or non–germinal-center B cell) or by PKC-beta protein expression, the authors found no significant associations with either DFS or overall survival.
Enzastaurin was generally safe, with minor and manageable adverse events. More patients in the enzastaurin arm had episodes of QTc prolongation, but these did not require discontinuation of the drug.
Dr. Crump and coauthors disclosed consulting, advising, research funding and other relationships with various companies, including Eli Lilly. Five coauthors are Lilly employees.
FROM JOURNAL OF CLINICAL ONCOLOGY
Key clinical point: The experimental agent enzastaurin was no better than placebo as maintenance therapy in patients who had achieved a complete response to R-CHOP.
Major finding: At 48 months median follow-up, the hazard ratio for enzastaurin was 0.92 (P = ns).
Data source: Randomized, double-blind, placebo controlled trial of 758 patients with diffuse large B-cell lymphoma in complete remission after first-line therapy with R-CHOP.
Disclosures: Dr. Crump and coauthors disclosed consulting, advising, research funding and other relationships with various companies, including Eli Lilly. Five coauthors are Lilly employees.
Epstein-Barr virus DNA in plasma reliably detects EBV-positive lymphoproliferative disorders
Detection of Epstein-Barr virus DNA in plasma reliably signaled “a broad range” of EBV+ diseases, according to investigators.
In contrast, the presence of EBV DNA in peripheral blood mononuclear cells did not reliably predict EBV diseases, said Dr. Jennifer A. Kanakry and her associates at Johns Hopkins University, Baltimore. Patients without EBV diseases can have EBV DNA in their PBMCs, particularly if they are immunocompromised, the researchers observed.
Latent EBV infection is associated with lymphomas, lymphoproliferative disorders, hemophagocytic lymphohistiocytosis, solid tumors, and other diseases. To characterize the relationship between these diseases and EBV DNA, the researchers studied viral quantitative real-time polymerase chain reaction assays of plasma and PBMCs from 2,146 patients tested at Johns Hopkins over 5 years. Patients were usually immunocompromised and hospitalized, the investigators noted (Blood 2016;127:2007-17).
A total of 535 patients (25%) had EBV detected in plasma or PBMCs. Notably, 69% of patients who did not have EBV diseases had EBV in PBMCs, but not in plasma. Among 105 patients with active systemic EBV+ diseases, 99% had EBV DNA in plasma, but only 54% had EBV in PBMCs. Furthermore, the number of copies of EBV DNA distinguished untreated EBV+ lymphoma, remitted EBV+ lymphoma, and EBV- lymphoma, and also distinguished untreated, EBV+ post-transplantation lymphoproliferative disorder (PTLD), EBV+ PTLD in remission, and EBV– PTLD.
“Cell-free (plasma) EBV DNA performs better than cellular EBV DNA as a marker of a broad range of EBV+ diseases,” the investigators concluded. “Within a largely immunocompromised and hospitalized cohort, detection of EBV DNA in plasma is uncommon in the absence of EBV+ disease.”
The National Cancer Institute, National Institutes of Health, and Center for AIDS Research funded the study. The researchers had no disclosures.
Detection of Epstein-Barr virus DNA in plasma reliably signaled “a broad range” of EBV+ diseases, according to investigators.
In contrast, the presence of EBV DNA in peripheral blood mononuclear cells did not reliably predict EBV diseases, said Dr. Jennifer A. Kanakry and her associates at Johns Hopkins University, Baltimore. Patients without EBV diseases can have EBV DNA in their PBMCs, particularly if they are immunocompromised, the researchers observed.
Latent EBV infection is associated with lymphomas, lymphoproliferative disorders, hemophagocytic lymphohistiocytosis, solid tumors, and other diseases. To characterize the relationship between these diseases and EBV DNA, the researchers studied viral quantitative real-time polymerase chain reaction assays of plasma and PBMCs from 2,146 patients tested at Johns Hopkins over 5 years. Patients were usually immunocompromised and hospitalized, the investigators noted (Blood 2016;127:2007-17).
A total of 535 patients (25%) had EBV detected in plasma or PBMCs. Notably, 69% of patients who did not have EBV diseases had EBV in PBMCs, but not in plasma. Among 105 patients with active systemic EBV+ diseases, 99% had EBV DNA in plasma, but only 54% had EBV in PBMCs. Furthermore, the number of copies of EBV DNA distinguished untreated EBV+ lymphoma, remitted EBV+ lymphoma, and EBV- lymphoma, and also distinguished untreated, EBV+ post-transplantation lymphoproliferative disorder (PTLD), EBV+ PTLD in remission, and EBV– PTLD.
“Cell-free (plasma) EBV DNA performs better than cellular EBV DNA as a marker of a broad range of EBV+ diseases,” the investigators concluded. “Within a largely immunocompromised and hospitalized cohort, detection of EBV DNA in plasma is uncommon in the absence of EBV+ disease.”
The National Cancer Institute, National Institutes of Health, and Center for AIDS Research funded the study. The researchers had no disclosures.
Detection of Epstein-Barr virus DNA in plasma reliably signaled “a broad range” of EBV+ diseases, according to investigators.
In contrast, the presence of EBV DNA in peripheral blood mononuclear cells did not reliably predict EBV diseases, said Dr. Jennifer A. Kanakry and her associates at Johns Hopkins University, Baltimore. Patients without EBV diseases can have EBV DNA in their PBMCs, particularly if they are immunocompromised, the researchers observed.
Latent EBV infection is associated with lymphomas, lymphoproliferative disorders, hemophagocytic lymphohistiocytosis, solid tumors, and other diseases. To characterize the relationship between these diseases and EBV DNA, the researchers studied viral quantitative real-time polymerase chain reaction assays of plasma and PBMCs from 2,146 patients tested at Johns Hopkins over 5 years. Patients were usually immunocompromised and hospitalized, the investigators noted (Blood 2016;127:2007-17).
A total of 535 patients (25%) had EBV detected in plasma or PBMCs. Notably, 69% of patients who did not have EBV diseases had EBV in PBMCs, but not in plasma. Among 105 patients with active systemic EBV+ diseases, 99% had EBV DNA in plasma, but only 54% had EBV in PBMCs. Furthermore, the number of copies of EBV DNA distinguished untreated EBV+ lymphoma, remitted EBV+ lymphoma, and EBV- lymphoma, and also distinguished untreated, EBV+ post-transplantation lymphoproliferative disorder (PTLD), EBV+ PTLD in remission, and EBV– PTLD.
“Cell-free (plasma) EBV DNA performs better than cellular EBV DNA as a marker of a broad range of EBV+ diseases,” the investigators concluded. “Within a largely immunocompromised and hospitalized cohort, detection of EBV DNA in plasma is uncommon in the absence of EBV+ disease.”
The National Cancer Institute, National Institutes of Health, and Center for AIDS Research funded the study. The researchers had no disclosures.
FROM BLOOD
Key clinical point: Epstein-Barr virus DNA was a more specific indicator of EBV+ diseases when detected in plasma, as opposed to peripheral blood mononuclear cells.
Major finding: Among 105 patients with active systemic EBV+ diseases, 99% had EBV DNA in plasma, but 54% had EBV in PBMCs.
Data source: Viral quantitative real-time polymerase chain reaction assays of plasma and PBMCs from 2,146 patients.
Disclosures: The National Cancer Institute, National Institutes of Health, and Center for AIDS Research funded the study. The researchers had no disclosures.
Low transformation rate in nodular lymphocyte–predominant Hodgkin lymphoma
Fewer than 8% of cases of nodular lymphocyte–predominant Hodgkin lymphoma (NLPHL) transformed to diffuse large B-cell lymphoma (DLBCL), based on a large prospective single-center study with long-term follow-up.
This rate was lower than the risk of transformation reported for transformed follicular lymphoma or chronic lymphocytic leukemia, according to Dr. Saad Kenderian and his associates at the Mayo Clinic, Rochester, Minn. Transformation was significantly associated with splenic involvement at presentation and with prior chemotherapy exposure, but did not worsen overall survival, they added.
“To our knowledge, this cohort represents the largest analysis to date of consecutive patients with NLPHL,” they said.
The study comprised 222 patients with newly diagnosed NLPHL who were treated at Mayo Clinic between 1970 and 2011. Median age at diagnosis was 40 years, and two-thirds of patients were men. The median follow-up period was 16 years (Blood 2016;12:1960-6. doi: 10.1182/blood-2015-08-665505).
During follow up, 17 cases (7.6%) transformed to DLBCL, for a transformation rate of 0.74 cases for every 100 patient-years, the investigators said. Median time to transformation was 35 months (range, 6-268 months). Predictors of transformation included any prior chemotherapy exposure (P = .04) and splenic involvement (P = .03). The rates of 40-year freedom from transformation were 87% when there was no splenic involvement and 21% when the spleen was involved, and were 87% if radiation therapy was used as a single modality compared with 77% in patients treated with prior chemotherapy or chemoradiation.
Five-year overall survival was 76% in patients with transformed disease, which was similar to overall survival among patients whose disease did not transform to DLBCL, the researchers noted.
Other studies of NLPHL have reported anywhere from a 2% to a 17% transformation rate, but those studies had smaller sample sizes, shorter follow-up periods, and less rigorous enrollment criteria and methods to confirm transformation, the investigators noted. “The finding of splenic involvement as a risk factor for transformation was reported by previous investigators. Interestingly, the association between exposure to prior chemotherapy and reduced freedom from transformation has not been reported in the past, but it has been observed in other low-grade lymphoma studies,” they added. “In contrast to follicular lymphoma, transformed NLPHL is not associated with an adverse impact on OS, suggesting a possibly different biology of transformation.”
The research was partially supported by Lymphoma SPORE and the Predolin Foundation. The investigators had no disclosures.
Kenderian et al. report a lower rate of transformation (7.6%) to diffuse large B-cell lymphoma for patients with nodular lymphocyte–predominant Hodgkin lymphoma compared with other series and found that transformation did not have a negative impact on overall survival. Reassuringly, even if transformation occurs, it is generally at a low rate. Also, these patients do well with additional treatment and do not have worse overall survival. At the MD Anderson Cancer Center, we have used a regimen based on R-CHOP and have not seen transformations. But only through large cooperative clinical trials can we determine whether R-CHOP or other more novel regimens are actually superior to ABVD (doxorubicin, bleomycin, vinblastine, dacarbazine) or rituximab (R)-ABVD for patients at high risk of transformation.
Dr. Michelle Fanale is at the University of Texas MD Anderson Cancer Center, Houston. She had no disclosures. These comments are from her editorial (Blood 2016;1927:1946-7 doi: 10.1182/blood-2016-03-699108).
Kenderian et al. report a lower rate of transformation (7.6%) to diffuse large B-cell lymphoma for patients with nodular lymphocyte–predominant Hodgkin lymphoma compared with other series and found that transformation did not have a negative impact on overall survival. Reassuringly, even if transformation occurs, it is generally at a low rate. Also, these patients do well with additional treatment and do not have worse overall survival. At the MD Anderson Cancer Center, we have used a regimen based on R-CHOP and have not seen transformations. But only through large cooperative clinical trials can we determine whether R-CHOP or other more novel regimens are actually superior to ABVD (doxorubicin, bleomycin, vinblastine, dacarbazine) or rituximab (R)-ABVD for patients at high risk of transformation.
Dr. Michelle Fanale is at the University of Texas MD Anderson Cancer Center, Houston. She had no disclosures. These comments are from her editorial (Blood 2016;1927:1946-7 doi: 10.1182/blood-2016-03-699108).
Kenderian et al. report a lower rate of transformation (7.6%) to diffuse large B-cell lymphoma for patients with nodular lymphocyte–predominant Hodgkin lymphoma compared with other series and found that transformation did not have a negative impact on overall survival. Reassuringly, even if transformation occurs, it is generally at a low rate. Also, these patients do well with additional treatment and do not have worse overall survival. At the MD Anderson Cancer Center, we have used a regimen based on R-CHOP and have not seen transformations. But only through large cooperative clinical trials can we determine whether R-CHOP or other more novel regimens are actually superior to ABVD (doxorubicin, bleomycin, vinblastine, dacarbazine) or rituximab (R)-ABVD for patients at high risk of transformation.
Dr. Michelle Fanale is at the University of Texas MD Anderson Cancer Center, Houston. She had no disclosures. These comments are from her editorial (Blood 2016;1927:1946-7 doi: 10.1182/blood-2016-03-699108).
Fewer than 8% of cases of nodular lymphocyte–predominant Hodgkin lymphoma (NLPHL) transformed to diffuse large B-cell lymphoma (DLBCL), based on a large prospective single-center study with long-term follow-up.
This rate was lower than the risk of transformation reported for transformed follicular lymphoma or chronic lymphocytic leukemia, according to Dr. Saad Kenderian and his associates at the Mayo Clinic, Rochester, Minn. Transformation was significantly associated with splenic involvement at presentation and with prior chemotherapy exposure, but did not worsen overall survival, they added.
“To our knowledge, this cohort represents the largest analysis to date of consecutive patients with NLPHL,” they said.
The study comprised 222 patients with newly diagnosed NLPHL who were treated at Mayo Clinic between 1970 and 2011. Median age at diagnosis was 40 years, and two-thirds of patients were men. The median follow-up period was 16 years (Blood 2016;12:1960-6. doi: 10.1182/blood-2015-08-665505).
During follow up, 17 cases (7.6%) transformed to DLBCL, for a transformation rate of 0.74 cases for every 100 patient-years, the investigators said. Median time to transformation was 35 months (range, 6-268 months). Predictors of transformation included any prior chemotherapy exposure (P = .04) and splenic involvement (P = .03). The rates of 40-year freedom from transformation were 87% when there was no splenic involvement and 21% when the spleen was involved, and were 87% if radiation therapy was used as a single modality compared with 77% in patients treated with prior chemotherapy or chemoradiation.
Five-year overall survival was 76% in patients with transformed disease, which was similar to overall survival among patients whose disease did not transform to DLBCL, the researchers noted.
Other studies of NLPHL have reported anywhere from a 2% to a 17% transformation rate, but those studies had smaller sample sizes, shorter follow-up periods, and less rigorous enrollment criteria and methods to confirm transformation, the investigators noted. “The finding of splenic involvement as a risk factor for transformation was reported by previous investigators. Interestingly, the association between exposure to prior chemotherapy and reduced freedom from transformation has not been reported in the past, but it has been observed in other low-grade lymphoma studies,” they added. “In contrast to follicular lymphoma, transformed NLPHL is not associated with an adverse impact on OS, suggesting a possibly different biology of transformation.”
The research was partially supported by Lymphoma SPORE and the Predolin Foundation. The investigators had no disclosures.
Fewer than 8% of cases of nodular lymphocyte–predominant Hodgkin lymphoma (NLPHL) transformed to diffuse large B-cell lymphoma (DLBCL), based on a large prospective single-center study with long-term follow-up.
This rate was lower than the risk of transformation reported for transformed follicular lymphoma or chronic lymphocytic leukemia, according to Dr. Saad Kenderian and his associates at the Mayo Clinic, Rochester, Minn. Transformation was significantly associated with splenic involvement at presentation and with prior chemotherapy exposure, but did not worsen overall survival, they added.
“To our knowledge, this cohort represents the largest analysis to date of consecutive patients with NLPHL,” they said.
The study comprised 222 patients with newly diagnosed NLPHL who were treated at Mayo Clinic between 1970 and 2011. Median age at diagnosis was 40 years, and two-thirds of patients were men. The median follow-up period was 16 years (Blood 2016;12:1960-6. doi: 10.1182/blood-2015-08-665505).
During follow up, 17 cases (7.6%) transformed to DLBCL, for a transformation rate of 0.74 cases for every 100 patient-years, the investigators said. Median time to transformation was 35 months (range, 6-268 months). Predictors of transformation included any prior chemotherapy exposure (P = .04) and splenic involvement (P = .03). The rates of 40-year freedom from transformation were 87% when there was no splenic involvement and 21% when the spleen was involved, and were 87% if radiation therapy was used as a single modality compared with 77% in patients treated with prior chemotherapy or chemoradiation.
Five-year overall survival was 76% in patients with transformed disease, which was similar to overall survival among patients whose disease did not transform to DLBCL, the researchers noted.
Other studies of NLPHL have reported anywhere from a 2% to a 17% transformation rate, but those studies had smaller sample sizes, shorter follow-up periods, and less rigorous enrollment criteria and methods to confirm transformation, the investigators noted. “The finding of splenic involvement as a risk factor for transformation was reported by previous investigators. Interestingly, the association between exposure to prior chemotherapy and reduced freedom from transformation has not been reported in the past, but it has been observed in other low-grade lymphoma studies,” they added. “In contrast to follicular lymphoma, transformed NLPHL is not associated with an adverse impact on OS, suggesting a possibly different biology of transformation.”
The research was partially supported by Lymphoma SPORE and the Predolin Foundation. The investigators had no disclosures.
FROM BLOOD
Key clinical point: The risk of transformation to diffuse large B-cell lymphoma is low in patients with nodular lymphocyte–predominant Hodgkin lymphoma.
Major finding: Only 7.6% of cases transformed over a median of 16 years of follow-up, and transformation did not worsen overall survival.
Data source: A prospective single-center study of 222 consecutive adults with NLPHL.
Disclosures: The research was partially supported by Lymphoma SPORE and the Predolin Foundation. The investigators had no disclosures.