User login
Gilteritinib prolonged survival in FLT3-mutated AML
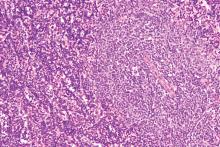
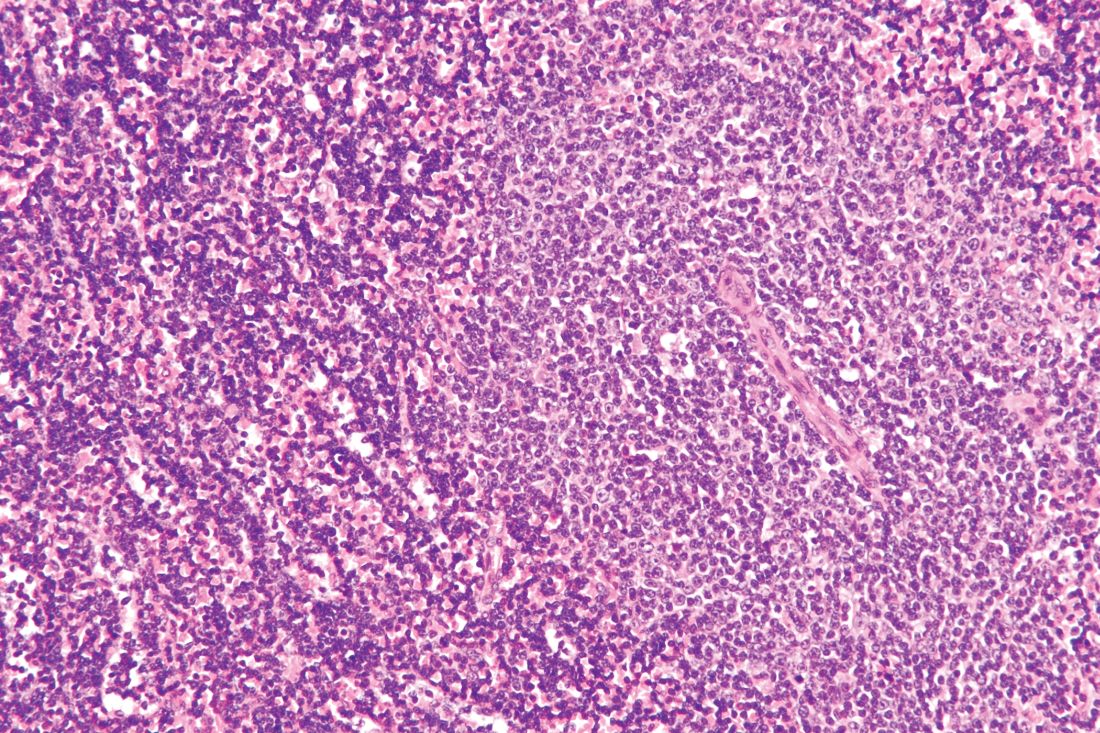
ATLANTA – The FLT3 inhibitor gilteritinib (Xospata) significantly prolonged overall survival, compared with salvage chemotherapy, in patients with FLT3-mutated relapsed/refractory acute myeloid leukemia, Alexander E. Perl, MD, from the Abramson Cancer Center at the University of Pennsylvania in Philadelphia reported at the 2019 annual meeting of the American Association for Cancer Research (AACR).
In a video interview, Dr. Perl discussed the results of the ADMIRAL global phase 3 randomized trial and described the current state of therapy for patients with relapsed/refractory AML bearing FLT3 mutations. Up to 70% of patients with acute myeloid leukemia will experience a relapse, and up to 40% may have disease that is resistant to induction chemotherapy. Survival for these patients is generally poor.
In particular, patients with acute myeloid leukemia and FLT3-activating mutations are at increased risk for early relapse and poor overall survival.
The ADMIRAL trial is funded by Astellas Pharma. Dr. Perl disclosed advisory board participation, consulting fees, and institutional support from Astellas and others.
ATLANTA – The FLT3 inhibitor gilteritinib (Xospata) significantly prolonged overall survival, compared with salvage chemotherapy, in patients with FLT3-mutated relapsed/refractory acute myeloid leukemia, Alexander E. Perl, MD, from the Abramson Cancer Center at the University of Pennsylvania in Philadelphia reported at the 2019 annual meeting of the American Association for Cancer Research (AACR).
In a video interview, Dr. Perl discussed the results of the ADMIRAL global phase 3 randomized trial and described the current state of therapy for patients with relapsed/refractory AML bearing FLT3 mutations. Up to 70% of patients with acute myeloid leukemia will experience a relapse, and up to 40% may have disease that is resistant to induction chemotherapy. Survival for these patients is generally poor.
In particular, patients with acute myeloid leukemia and FLT3-activating mutations are at increased risk for early relapse and poor overall survival.
The ADMIRAL trial is funded by Astellas Pharma. Dr. Perl disclosed advisory board participation, consulting fees, and institutional support from Astellas and others.
ATLANTA – The FLT3 inhibitor gilteritinib (Xospata) significantly prolonged overall survival, compared with salvage chemotherapy, in patients with FLT3-mutated relapsed/refractory acute myeloid leukemia, Alexander E. Perl, MD, from the Abramson Cancer Center at the University of Pennsylvania in Philadelphia reported at the 2019 annual meeting of the American Association for Cancer Research (AACR).
In a video interview, Dr. Perl discussed the results of the ADMIRAL global phase 3 randomized trial and described the current state of therapy for patients with relapsed/refractory AML bearing FLT3 mutations. Up to 70% of patients with acute myeloid leukemia will experience a relapse, and up to 40% may have disease that is resistant to induction chemotherapy. Survival for these patients is generally poor.
In particular, patients with acute myeloid leukemia and FLT3-activating mutations are at increased risk for early relapse and poor overall survival.
The ADMIRAL trial is funded by Astellas Pharma. Dr. Perl disclosed advisory board participation, consulting fees, and institutional support from Astellas and others.
REPORTING FROM AACR 2019
Bendamustine/rituximab combo proves viable for comorbid CLL
A combination of bendamustine and rituximab generated an 88% overall response rate and 96% overall survival rate at 2 years among patients with chronic lymphocytic leukemia (CLL) in a study of 83 patients aged 53-83 years.
Although combined fludarabine, cyclophosphamide, and rituximab has demonstrated success in younger patients with CLL, this therapy is often considered too aggressive for the majority of CLL patients, who tend to be older and have multiple comorbidities, wrote Martin Špacek, MD, of Charles University and General University Hospital in Prague and his colleagues.
The alternative treatment combination of bendamustine and rituximab (BR) has not been well studied in patients with comorbidities, they said.
In a study published in Leukemia Research, the researchers enrolled 83 previously untreated adults with progressive CLL. The average age of the participants was 71 years, and 61% were men. The median creatinine clearance for the study population was 65 mL/min, and all patients had comorbidities, defined as scores greater than 6 on the Cumulative Illness Rating Scale (CIRS).
All patients were prescribed 90 mg/m2 bendamustine on days 1 and 2 combined with 375 mg/m2 rituximab on day 0 of the first course, and 500 mg/m2 rituximab on day 1 during subsequent courses every 28 days for a maximum of six cycles.
The overall response rate to BR was 88.0%, with a complete response rate of 20.5%. At 2 years, progression-free survival and overall survival rates were 69.9% and 96.2%, respectively.
A total of 51 patients (61.4%) experienced at least one grade 3 or 4 adverse event. The most common hematologic effects were neutropenia (40 patients), thrombocytopenia (14 patients), and anemia (8 patients). The most common nonhematologic effects were grade 3– or grade 4–level infections in 12 patients. Six patients developed severe skin rash.
Additionally, one patient developed sepsis during treatment and died after the first course of therapy.
“Age and CIRS failed to predict any severe toxicities or BR dose reduction,” the researchers noted.
The findings support data from previous studies and represent the largest study of CLL patients with significant comorbidities to be treated with BR, the researchers said.
More prospective research is needed, but the results demonstrate that “chemoimmunotherapy with BR is an effective therapeutic option with manageable toxicity for the initial treatment of CLL patients with significant comorbidities,” the investigators wrote.
The study was supported by the Ministry of Health, Czech Republic, the Charles University Progres program, and the Czech CLL Study Group. Researchers reported honoraria and travel grants from Mundipharma and Roche.
SOURCE: Spacek M et al. Leuk Res. 2019;79:17-21.
A combination of bendamustine and rituximab generated an 88% overall response rate and 96% overall survival rate at 2 years among patients with chronic lymphocytic leukemia (CLL) in a study of 83 patients aged 53-83 years.
Although combined fludarabine, cyclophosphamide, and rituximab has demonstrated success in younger patients with CLL, this therapy is often considered too aggressive for the majority of CLL patients, who tend to be older and have multiple comorbidities, wrote Martin Špacek, MD, of Charles University and General University Hospital in Prague and his colleagues.
The alternative treatment combination of bendamustine and rituximab (BR) has not been well studied in patients with comorbidities, they said.
In a study published in Leukemia Research, the researchers enrolled 83 previously untreated adults with progressive CLL. The average age of the participants was 71 years, and 61% were men. The median creatinine clearance for the study population was 65 mL/min, and all patients had comorbidities, defined as scores greater than 6 on the Cumulative Illness Rating Scale (CIRS).
All patients were prescribed 90 mg/m2 bendamustine on days 1 and 2 combined with 375 mg/m2 rituximab on day 0 of the first course, and 500 mg/m2 rituximab on day 1 during subsequent courses every 28 days for a maximum of six cycles.
The overall response rate to BR was 88.0%, with a complete response rate of 20.5%. At 2 years, progression-free survival and overall survival rates were 69.9% and 96.2%, respectively.
A total of 51 patients (61.4%) experienced at least one grade 3 or 4 adverse event. The most common hematologic effects were neutropenia (40 patients), thrombocytopenia (14 patients), and anemia (8 patients). The most common nonhematologic effects were grade 3– or grade 4–level infections in 12 patients. Six patients developed severe skin rash.
Additionally, one patient developed sepsis during treatment and died after the first course of therapy.
“Age and CIRS failed to predict any severe toxicities or BR dose reduction,” the researchers noted.
The findings support data from previous studies and represent the largest study of CLL patients with significant comorbidities to be treated with BR, the researchers said.
More prospective research is needed, but the results demonstrate that “chemoimmunotherapy with BR is an effective therapeutic option with manageable toxicity for the initial treatment of CLL patients with significant comorbidities,” the investigators wrote.
The study was supported by the Ministry of Health, Czech Republic, the Charles University Progres program, and the Czech CLL Study Group. Researchers reported honoraria and travel grants from Mundipharma and Roche.
SOURCE: Spacek M et al. Leuk Res. 2019;79:17-21.
A combination of bendamustine and rituximab generated an 88% overall response rate and 96% overall survival rate at 2 years among patients with chronic lymphocytic leukemia (CLL) in a study of 83 patients aged 53-83 years.
Although combined fludarabine, cyclophosphamide, and rituximab has demonstrated success in younger patients with CLL, this therapy is often considered too aggressive for the majority of CLL patients, who tend to be older and have multiple comorbidities, wrote Martin Špacek, MD, of Charles University and General University Hospital in Prague and his colleagues.
The alternative treatment combination of bendamustine and rituximab (BR) has not been well studied in patients with comorbidities, they said.
In a study published in Leukemia Research, the researchers enrolled 83 previously untreated adults with progressive CLL. The average age of the participants was 71 years, and 61% were men. The median creatinine clearance for the study population was 65 mL/min, and all patients had comorbidities, defined as scores greater than 6 on the Cumulative Illness Rating Scale (CIRS).
All patients were prescribed 90 mg/m2 bendamustine on days 1 and 2 combined with 375 mg/m2 rituximab on day 0 of the first course, and 500 mg/m2 rituximab on day 1 during subsequent courses every 28 days for a maximum of six cycles.
The overall response rate to BR was 88.0%, with a complete response rate of 20.5%. At 2 years, progression-free survival and overall survival rates were 69.9% and 96.2%, respectively.
A total of 51 patients (61.4%) experienced at least one grade 3 or 4 adverse event. The most common hematologic effects were neutropenia (40 patients), thrombocytopenia (14 patients), and anemia (8 patients). The most common nonhematologic effects were grade 3– or grade 4–level infections in 12 patients. Six patients developed severe skin rash.
Additionally, one patient developed sepsis during treatment and died after the first course of therapy.
“Age and CIRS failed to predict any severe toxicities or BR dose reduction,” the researchers noted.
The findings support data from previous studies and represent the largest study of CLL patients with significant comorbidities to be treated with BR, the researchers said.
More prospective research is needed, but the results demonstrate that “chemoimmunotherapy with BR is an effective therapeutic option with manageable toxicity for the initial treatment of CLL patients with significant comorbidities,” the investigators wrote.
The study was supported by the Ministry of Health, Czech Republic, the Charles University Progres program, and the Czech CLL Study Group. Researchers reported honoraria and travel grants from Mundipharma and Roche.
SOURCE: Spacek M et al. Leuk Res. 2019;79:17-21.
FROM LEUKEMIA RESEARCH
Key clinical point:
Major finding: The overall response rate for the combination therapy was 88.0%; complete response was 20.5%.
Study details: A prospective, observational study of 83 patients with chronic lymphocytic leukemia.
Disclosures: The study was supported by the Ministry of Health, Czech Republic, the Charles University Progres program, and the Czech CLL Study Group. Researchers reported honoraria and travel grants from Mundipharma and Roche.
Source: Spacek M et al. Leuk Res. 2019;79:17-21.
Whole-genome sequencing demonstrates clinical relevance
GLASGOW – Whole genome sequencing (WGS) appears capable of replacing cytogenetic testing and next generation sequencing (NGS) for the detection of clinically relevant molecular abnormalities in hematological malignancies, according to investigators.
A comparison of WGS with fluorescence in situ hybridization (FISH) showed that WGS caught all the same significant structural variants, plus some abnormalities that FISH had not detected, reported lead author Shirley Henderson, PhD, lead for cancer molecular diagnostics at Genomics England in Oxford.
Although further validation is needed, these findings, reported at the annual meeting of the British Society for Haematology, support an ongoing effort to validate the clinical reliability of WGS, which is currently reserved for research purposes.
“It’s vitally important that the clinical community engage with this and understand both the power and the limitations of this technique and how this work is going to be interpreted for the benefit of patients,” said Adele Fielding, PhD, session chair from University College London’s Cancer Institute.
The investigators compared WGS with FISH for detection of clinically significant structural variants (SVs) and copy number variants (CNVs) in tumor samples from 34 patients with acute myeloid leukemia (AML) and acute lymphoblastic leukemia (ALL).
The 252 standard of care FISH tests – conducted at three separate clinical diagnostic centers in the United Kingdom – included 138 SVs and 114 CNVs. WGS relied on a combination of bioinformatics and visual inspection of Circos plots. WGS confirmed all of the SVs detected by FISH with high confidence; WGS detected four additional SVs, also with high confidence, including an ETV6-RUNX1 fusion not detected by FISH because of probe limitations.
Results for CNVs were similar, with WGS detecting 78 out of 85 positive CNVs. Six of the missed positives were associated with low quality samples or low level mutations in the FISH test, suggesting that at least some positives may have been detected with better samples. Only one negative CNV from FISH was missed by WGS.
Overall, WGS had a false positive rate of less than 5% and a positive percentage agreement with FISH that exceeded 90%.
“Further work is required to fully validate all aspects of the WGS analysis pipeline,” Dr. Henderson said. “But these results indicate that WGS has the potential to reliably detect SVs and CNVs in these conditions while offering the advantage of detecting all SVs and CNVs present without the need for additional interrogation of the sample by multiple tests or probes.”
Dr. Henderson noted that there is really no “perfect method” for identifying structural and copy number variants at the present time.
Small variants are relatively easy to detect with techniques such as karyotyping and gene banding, but these tests have shortcomings, namely, that they require live cells and have “fairly high failure rates for various reasons,” Dr. Henderson said.
“FISH is an incredibly useful test and it has higher resolution than gene banding, but the problem with FISH is that you only find what you’re looking at,” Dr. Henderson said. “It’s not genome wide; it’s very targeted.”
Similarly, polymerase chain reaction (PCR), including next generation sequencing (NGS), can detect molecular abnormalities, but only those that are targeted, which may necessitate multiple tests, she said.
“If you start looking for all of the structural variants [with existing techniques], then you’re going to be doing an awful lot of tests,” Dr. Henderson said.
Another potential benefit of WGS is that it is “future resistant,” Dr. Henderson said. “As new biomarkers are discovered, you don’t have to redesign a new targeted test. It will also detect emerging biomarkers, such as mutational signatures and burden.”
The study was sponsored by NHS England. The investigators reported having no conflicts of interest.
SOURCE: Henderson S et al. BSH 2019, Abstract OR-002.
GLASGOW – Whole genome sequencing (WGS) appears capable of replacing cytogenetic testing and next generation sequencing (NGS) for the detection of clinically relevant molecular abnormalities in hematological malignancies, according to investigators.
A comparison of WGS with fluorescence in situ hybridization (FISH) showed that WGS caught all the same significant structural variants, plus some abnormalities that FISH had not detected, reported lead author Shirley Henderson, PhD, lead for cancer molecular diagnostics at Genomics England in Oxford.
Although further validation is needed, these findings, reported at the annual meeting of the British Society for Haematology, support an ongoing effort to validate the clinical reliability of WGS, which is currently reserved for research purposes.
“It’s vitally important that the clinical community engage with this and understand both the power and the limitations of this technique and how this work is going to be interpreted for the benefit of patients,” said Adele Fielding, PhD, session chair from University College London’s Cancer Institute.
The investigators compared WGS with FISH for detection of clinically significant structural variants (SVs) and copy number variants (CNVs) in tumor samples from 34 patients with acute myeloid leukemia (AML) and acute lymphoblastic leukemia (ALL).
The 252 standard of care FISH tests – conducted at three separate clinical diagnostic centers in the United Kingdom – included 138 SVs and 114 CNVs. WGS relied on a combination of bioinformatics and visual inspection of Circos plots. WGS confirmed all of the SVs detected by FISH with high confidence; WGS detected four additional SVs, also with high confidence, including an ETV6-RUNX1 fusion not detected by FISH because of probe limitations.
Results for CNVs were similar, with WGS detecting 78 out of 85 positive CNVs. Six of the missed positives were associated with low quality samples or low level mutations in the FISH test, suggesting that at least some positives may have been detected with better samples. Only one negative CNV from FISH was missed by WGS.
Overall, WGS had a false positive rate of less than 5% and a positive percentage agreement with FISH that exceeded 90%.
“Further work is required to fully validate all aspects of the WGS analysis pipeline,” Dr. Henderson said. “But these results indicate that WGS has the potential to reliably detect SVs and CNVs in these conditions while offering the advantage of detecting all SVs and CNVs present without the need for additional interrogation of the sample by multiple tests or probes.”
Dr. Henderson noted that there is really no “perfect method” for identifying structural and copy number variants at the present time.
Small variants are relatively easy to detect with techniques such as karyotyping and gene banding, but these tests have shortcomings, namely, that they require live cells and have “fairly high failure rates for various reasons,” Dr. Henderson said.
“FISH is an incredibly useful test and it has higher resolution than gene banding, but the problem with FISH is that you only find what you’re looking at,” Dr. Henderson said. “It’s not genome wide; it’s very targeted.”
Similarly, polymerase chain reaction (PCR), including next generation sequencing (NGS), can detect molecular abnormalities, but only those that are targeted, which may necessitate multiple tests, she said.
“If you start looking for all of the structural variants [with existing techniques], then you’re going to be doing an awful lot of tests,” Dr. Henderson said.
Another potential benefit of WGS is that it is “future resistant,” Dr. Henderson said. “As new biomarkers are discovered, you don’t have to redesign a new targeted test. It will also detect emerging biomarkers, such as mutational signatures and burden.”
The study was sponsored by NHS England. The investigators reported having no conflicts of interest.
SOURCE: Henderson S et al. BSH 2019, Abstract OR-002.
GLASGOW – Whole genome sequencing (WGS) appears capable of replacing cytogenetic testing and next generation sequencing (NGS) for the detection of clinically relevant molecular abnormalities in hematological malignancies, according to investigators.
A comparison of WGS with fluorescence in situ hybridization (FISH) showed that WGS caught all the same significant structural variants, plus some abnormalities that FISH had not detected, reported lead author Shirley Henderson, PhD, lead for cancer molecular diagnostics at Genomics England in Oxford.
Although further validation is needed, these findings, reported at the annual meeting of the British Society for Haematology, support an ongoing effort to validate the clinical reliability of WGS, which is currently reserved for research purposes.
“It’s vitally important that the clinical community engage with this and understand both the power and the limitations of this technique and how this work is going to be interpreted for the benefit of patients,” said Adele Fielding, PhD, session chair from University College London’s Cancer Institute.
The investigators compared WGS with FISH for detection of clinically significant structural variants (SVs) and copy number variants (CNVs) in tumor samples from 34 patients with acute myeloid leukemia (AML) and acute lymphoblastic leukemia (ALL).
The 252 standard of care FISH tests – conducted at three separate clinical diagnostic centers in the United Kingdom – included 138 SVs and 114 CNVs. WGS relied on a combination of bioinformatics and visual inspection of Circos plots. WGS confirmed all of the SVs detected by FISH with high confidence; WGS detected four additional SVs, also with high confidence, including an ETV6-RUNX1 fusion not detected by FISH because of probe limitations.
Results for CNVs were similar, with WGS detecting 78 out of 85 positive CNVs. Six of the missed positives were associated with low quality samples or low level mutations in the FISH test, suggesting that at least some positives may have been detected with better samples. Only one negative CNV from FISH was missed by WGS.
Overall, WGS had a false positive rate of less than 5% and a positive percentage agreement with FISH that exceeded 90%.
“Further work is required to fully validate all aspects of the WGS analysis pipeline,” Dr. Henderson said. “But these results indicate that WGS has the potential to reliably detect SVs and CNVs in these conditions while offering the advantage of detecting all SVs and CNVs present without the need for additional interrogation of the sample by multiple tests or probes.”
Dr. Henderson noted that there is really no “perfect method” for identifying structural and copy number variants at the present time.
Small variants are relatively easy to detect with techniques such as karyotyping and gene banding, but these tests have shortcomings, namely, that they require live cells and have “fairly high failure rates for various reasons,” Dr. Henderson said.
“FISH is an incredibly useful test and it has higher resolution than gene banding, but the problem with FISH is that you only find what you’re looking at,” Dr. Henderson said. “It’s not genome wide; it’s very targeted.”
Similarly, polymerase chain reaction (PCR), including next generation sequencing (NGS), can detect molecular abnormalities, but only those that are targeted, which may necessitate multiple tests, she said.
“If you start looking for all of the structural variants [with existing techniques], then you’re going to be doing an awful lot of tests,” Dr. Henderson said.
Another potential benefit of WGS is that it is “future resistant,” Dr. Henderson said. “As new biomarkers are discovered, you don’t have to redesign a new targeted test. It will also detect emerging biomarkers, such as mutational signatures and burden.”
The study was sponsored by NHS England. The investigators reported having no conflicts of interest.
SOURCE: Henderson S et al. BSH 2019, Abstract OR-002.
REPORTING FROM BSH 2019
Ibrutinib sustained responses in refractory CLL in long-term follow-up
Prolonged exposure to ibrutinib showed sustained progression-free and overall survival and had tolerable safety outcomes in patients with relapsed or refractory chronic lymphocytic leukemia, according to a post hoc analysis of the phase 3 RESONATE trial.
“This study ... provides further evidence for efficacy and safety with prolonged treatment across multiple high-risk genomic and clinical disease features and with increasing depth of response,” John C. Byrd, MD, of the Ohio State University, Columbus, and his colleagues wrote in Blood.
RESONATE included 391 high-risk patients with relapsed or refractory chronic lymphocytic leukemia (CLL). Study participants were randomized in a 1:1 fashion to receive ibrutinib 420 mg daily or ofatumumab (initial infusion of 300 mg followed by seven weekly infusions and four monthly infusions of 2,000 mg) for a maximum of 24 weeks. Drug therapy was continued until cancer progression or intolerable toxicity of either agent was seen.
“Primary analysis at median follow-up of 9.7 months demonstrated superiority of ibrutinib over ofatumumab in PFS [progression-free survival], OS [overall survival], and overall response,” the researchers wrote. “With extended follow-up of median 44 months, these same results persist; a plateau of PFS has not yet been reached in this long-term follow-up. We also observe very durable remissions among patients of all genomic groups, including those with del(17)(p13.1), del(11)(q22.3), or unmutated IgHV [immunoglobulin heavy chain gene], who are traditionally considered high-risk populations.”
After an extended follow-up (median, 44 months), the team found that the PFS benefit with ibrutinib was sustained, compared with ofatumumab (hazard ratio, 0.133; 95% confidence interval, 0.099-0.178; P less than .0001). The 3-year PFS rate was 59% for ibrutinib, compared with 3% for ofatumumab. Similar PFS benefits were seen among subgroups of high- and very high–risk patients, based on their scores on the International Prognostic Index for CLL.
The OS benefit was also sustained in those randomized to ibrutinib (HR, 0.591; 95% CI, 0.378-0.926; P = .0208). The continued OS benefit with ibrutinib versus ofatumumab continued even after a sensitivity analysis adjusted for crossover of patients to ibrutinib.
With respect to safety, adverse events of any grade were similar to previous reports of ibrutinib. In fact, the prevalence of adverse events (grade 3 or higher) decreased over time for participants that continued on ibrutinib.
“Multiple studies are ongoing to investigate ibrutinib earlier in the course of CLL therapy, including phase 3 studies of first-line ibrutinib [or ibrutinib combined with anti-CD20 therapy], compared with standard chemoimmunotherapy regimens,” they wrote.
The study was sponsored by Pharmacyclics and Janssen. The authors reported financial disclosures related to the sponsors and several other companies.
SOURCE: Byrd JC et al. Blood. 2019 Mar 6. doi: 10.1182/blood-2018-08-870238.
Prolonged exposure to ibrutinib showed sustained progression-free and overall survival and had tolerable safety outcomes in patients with relapsed or refractory chronic lymphocytic leukemia, according to a post hoc analysis of the phase 3 RESONATE trial.
“This study ... provides further evidence for efficacy and safety with prolonged treatment across multiple high-risk genomic and clinical disease features and with increasing depth of response,” John C. Byrd, MD, of the Ohio State University, Columbus, and his colleagues wrote in Blood.
RESONATE included 391 high-risk patients with relapsed or refractory chronic lymphocytic leukemia (CLL). Study participants were randomized in a 1:1 fashion to receive ibrutinib 420 mg daily or ofatumumab (initial infusion of 300 mg followed by seven weekly infusions and four monthly infusions of 2,000 mg) for a maximum of 24 weeks. Drug therapy was continued until cancer progression or intolerable toxicity of either agent was seen.
“Primary analysis at median follow-up of 9.7 months demonstrated superiority of ibrutinib over ofatumumab in PFS [progression-free survival], OS [overall survival], and overall response,” the researchers wrote. “With extended follow-up of median 44 months, these same results persist; a plateau of PFS has not yet been reached in this long-term follow-up. We also observe very durable remissions among patients of all genomic groups, including those with del(17)(p13.1), del(11)(q22.3), or unmutated IgHV [immunoglobulin heavy chain gene], who are traditionally considered high-risk populations.”
After an extended follow-up (median, 44 months), the team found that the PFS benefit with ibrutinib was sustained, compared with ofatumumab (hazard ratio, 0.133; 95% confidence interval, 0.099-0.178; P less than .0001). The 3-year PFS rate was 59% for ibrutinib, compared with 3% for ofatumumab. Similar PFS benefits were seen among subgroups of high- and very high–risk patients, based on their scores on the International Prognostic Index for CLL.
The OS benefit was also sustained in those randomized to ibrutinib (HR, 0.591; 95% CI, 0.378-0.926; P = .0208). The continued OS benefit with ibrutinib versus ofatumumab continued even after a sensitivity analysis adjusted for crossover of patients to ibrutinib.
With respect to safety, adverse events of any grade were similar to previous reports of ibrutinib. In fact, the prevalence of adverse events (grade 3 or higher) decreased over time for participants that continued on ibrutinib.
“Multiple studies are ongoing to investigate ibrutinib earlier in the course of CLL therapy, including phase 3 studies of first-line ibrutinib [or ibrutinib combined with anti-CD20 therapy], compared with standard chemoimmunotherapy regimens,” they wrote.
The study was sponsored by Pharmacyclics and Janssen. The authors reported financial disclosures related to the sponsors and several other companies.
SOURCE: Byrd JC et al. Blood. 2019 Mar 6. doi: 10.1182/blood-2018-08-870238.
Prolonged exposure to ibrutinib showed sustained progression-free and overall survival and had tolerable safety outcomes in patients with relapsed or refractory chronic lymphocytic leukemia, according to a post hoc analysis of the phase 3 RESONATE trial.
“This study ... provides further evidence for efficacy and safety with prolonged treatment across multiple high-risk genomic and clinical disease features and with increasing depth of response,” John C. Byrd, MD, of the Ohio State University, Columbus, and his colleagues wrote in Blood.
RESONATE included 391 high-risk patients with relapsed or refractory chronic lymphocytic leukemia (CLL). Study participants were randomized in a 1:1 fashion to receive ibrutinib 420 mg daily or ofatumumab (initial infusion of 300 mg followed by seven weekly infusions and four monthly infusions of 2,000 mg) for a maximum of 24 weeks. Drug therapy was continued until cancer progression or intolerable toxicity of either agent was seen.
“Primary analysis at median follow-up of 9.7 months demonstrated superiority of ibrutinib over ofatumumab in PFS [progression-free survival], OS [overall survival], and overall response,” the researchers wrote. “With extended follow-up of median 44 months, these same results persist; a plateau of PFS has not yet been reached in this long-term follow-up. We also observe very durable remissions among patients of all genomic groups, including those with del(17)(p13.1), del(11)(q22.3), or unmutated IgHV [immunoglobulin heavy chain gene], who are traditionally considered high-risk populations.”
After an extended follow-up (median, 44 months), the team found that the PFS benefit with ibrutinib was sustained, compared with ofatumumab (hazard ratio, 0.133; 95% confidence interval, 0.099-0.178; P less than .0001). The 3-year PFS rate was 59% for ibrutinib, compared with 3% for ofatumumab. Similar PFS benefits were seen among subgroups of high- and very high–risk patients, based on their scores on the International Prognostic Index for CLL.
The OS benefit was also sustained in those randomized to ibrutinib (HR, 0.591; 95% CI, 0.378-0.926; P = .0208). The continued OS benefit with ibrutinib versus ofatumumab continued even after a sensitivity analysis adjusted for crossover of patients to ibrutinib.
With respect to safety, adverse events of any grade were similar to previous reports of ibrutinib. In fact, the prevalence of adverse events (grade 3 or higher) decreased over time for participants that continued on ibrutinib.
“Multiple studies are ongoing to investigate ibrutinib earlier in the course of CLL therapy, including phase 3 studies of first-line ibrutinib [or ibrutinib combined with anti-CD20 therapy], compared with standard chemoimmunotherapy regimens,” they wrote.
The study was sponsored by Pharmacyclics and Janssen. The authors reported financial disclosures related to the sponsors and several other companies.
SOURCE: Byrd JC et al. Blood. 2019 Mar 6. doi: 10.1182/blood-2018-08-870238.
FROM BLOOD
Venetoclax and obinutuzumab induces deep responses in CLL
The combination of venetoclax and obinutuzumab provided high response rates and deep remissions regardless of cytogenetic risk factors in patients with chronic lymphocytic leukemia, according to recently reported results of a phase 1b study.
The regimen elicited high rates of undetectable minimal residual disease in peripheral blood and had an acceptable safety profile with manageable toxicities in the study reported in Blood, which included patients with previously untreated or relapsed/refractory chronic lymphocytic leukemia (CLL).
“The deep remission rates we observed with venetoclax-obinutuzumab have not been reported with previously available CLL treatments, including FCR [fludarabine, cyclophosphamide, and rituximab], which is currently considered the most efficacious regimen with limited-duration therapy,” wrote the investigators, led by Ian W. Flinn, MD, PhD, of Sarah Cannon Research Institute/Tennessee Oncology, Nashville.
Venetoclax-obinutuzumab combinations are meanwhile being tested in other studies – including the phase 3 CLL13 and CLL14 studies – which have enrolled previously untreated fit or unfit CLL patients, respectively.
“If the primary endpoints of these large-scale trials are met, venetoclax-obinutuzumab may become a new standard treatment option in [first-line] CLL, irrespective of clinical fitness,” Dr. Flinn and his colleagues wrote in their report.
The present phase 1b, dose-escalation study enrolled 32 patients who were previously untreated (median age, 63 years) and 46 patients who were relapsed or refractory to previous treatments (median age, 61 years).
Doses of venetoclax were escalated from 100 mg to 400 mg to determine its maximum tolerated dose when combined with obinutuzumab, the investigators wrote. Some patients received venetoclax first, while others received obinutuzumab first, for a total of 1 year of treatment.
The study confirmed favorable risk-benefit treatment used a dose of 400 mg venetoclax plus the standard dose of obinutuzumab, according to the researchers.
The overall best response rate was 95% for relapsed/refractory patients, including a 37% rate of complete response or complete response with incomplete marrow recovery. In previously untreated patients, the overall best response rate was 100%, including a 78% rate of complete responses by those criteria.
Undetectable minimal residual disease was observed in 64% of relapsed/refractory patients and 91% of previously untreated patients at 3 months after the last obinutuzumab dose, the investigators reported.
There were no dose-limiting toxicities in the study, no clinical tumor lysis syndrome, and no differences between the two schedules (venetoclax first or obinutuzumab first) in terms of adverse events, the investigators wrote.
Neutropenia was the most common serious (grade 3-4) adverse event, occurring in 58% of relapsed/refractory patients and 53% of patients treated in the first line. Grade 3-4 infections were seen in 29% and 13% of the relapsed/refractory and previously untreated patients, respectively.
There were no fatal infections among previously untreated patients, while three relapsed/refractory patients (7%) had fatal adverse events, including one case of acute respiratory failure in a patient with suspected Richter’s transformation, pneumonia in a patient with metastatic squamous cell lung carcinoma, and another case of pneumonia occurring about 3 months after the last dose of venetoclax.
Genentech and AbbVie provided financial support for the study. Dr. Flinn reported receiving research funding for his institution from Genentech, AbbVie, and several other companies.
SOURCE: Flinn IW et al. Blood. 2019 Mar 12. doi: 10.1182/blood-2019-01-896290.
The combination of venetoclax and obinutuzumab provided high response rates and deep remissions regardless of cytogenetic risk factors in patients with chronic lymphocytic leukemia, according to recently reported results of a phase 1b study.
The regimen elicited high rates of undetectable minimal residual disease in peripheral blood and had an acceptable safety profile with manageable toxicities in the study reported in Blood, which included patients with previously untreated or relapsed/refractory chronic lymphocytic leukemia (CLL).
“The deep remission rates we observed with venetoclax-obinutuzumab have not been reported with previously available CLL treatments, including FCR [fludarabine, cyclophosphamide, and rituximab], which is currently considered the most efficacious regimen with limited-duration therapy,” wrote the investigators, led by Ian W. Flinn, MD, PhD, of Sarah Cannon Research Institute/Tennessee Oncology, Nashville.
Venetoclax-obinutuzumab combinations are meanwhile being tested in other studies – including the phase 3 CLL13 and CLL14 studies – which have enrolled previously untreated fit or unfit CLL patients, respectively.
“If the primary endpoints of these large-scale trials are met, venetoclax-obinutuzumab may become a new standard treatment option in [first-line] CLL, irrespective of clinical fitness,” Dr. Flinn and his colleagues wrote in their report.
The present phase 1b, dose-escalation study enrolled 32 patients who were previously untreated (median age, 63 years) and 46 patients who were relapsed or refractory to previous treatments (median age, 61 years).
Doses of venetoclax were escalated from 100 mg to 400 mg to determine its maximum tolerated dose when combined with obinutuzumab, the investigators wrote. Some patients received venetoclax first, while others received obinutuzumab first, for a total of 1 year of treatment.
The study confirmed favorable risk-benefit treatment used a dose of 400 mg venetoclax plus the standard dose of obinutuzumab, according to the researchers.
The overall best response rate was 95% for relapsed/refractory patients, including a 37% rate of complete response or complete response with incomplete marrow recovery. In previously untreated patients, the overall best response rate was 100%, including a 78% rate of complete responses by those criteria.
Undetectable minimal residual disease was observed in 64% of relapsed/refractory patients and 91% of previously untreated patients at 3 months after the last obinutuzumab dose, the investigators reported.
There were no dose-limiting toxicities in the study, no clinical tumor lysis syndrome, and no differences between the two schedules (venetoclax first or obinutuzumab first) in terms of adverse events, the investigators wrote.
Neutropenia was the most common serious (grade 3-4) adverse event, occurring in 58% of relapsed/refractory patients and 53% of patients treated in the first line. Grade 3-4 infections were seen in 29% and 13% of the relapsed/refractory and previously untreated patients, respectively.
There were no fatal infections among previously untreated patients, while three relapsed/refractory patients (7%) had fatal adverse events, including one case of acute respiratory failure in a patient with suspected Richter’s transformation, pneumonia in a patient with metastatic squamous cell lung carcinoma, and another case of pneumonia occurring about 3 months after the last dose of venetoclax.
Genentech and AbbVie provided financial support for the study. Dr. Flinn reported receiving research funding for his institution from Genentech, AbbVie, and several other companies.
SOURCE: Flinn IW et al. Blood. 2019 Mar 12. doi: 10.1182/blood-2019-01-896290.
The combination of venetoclax and obinutuzumab provided high response rates and deep remissions regardless of cytogenetic risk factors in patients with chronic lymphocytic leukemia, according to recently reported results of a phase 1b study.
The regimen elicited high rates of undetectable minimal residual disease in peripheral blood and had an acceptable safety profile with manageable toxicities in the study reported in Blood, which included patients with previously untreated or relapsed/refractory chronic lymphocytic leukemia (CLL).
“The deep remission rates we observed with venetoclax-obinutuzumab have not been reported with previously available CLL treatments, including FCR [fludarabine, cyclophosphamide, and rituximab], which is currently considered the most efficacious regimen with limited-duration therapy,” wrote the investigators, led by Ian W. Flinn, MD, PhD, of Sarah Cannon Research Institute/Tennessee Oncology, Nashville.
Venetoclax-obinutuzumab combinations are meanwhile being tested in other studies – including the phase 3 CLL13 and CLL14 studies – which have enrolled previously untreated fit or unfit CLL patients, respectively.
“If the primary endpoints of these large-scale trials are met, venetoclax-obinutuzumab may become a new standard treatment option in [first-line] CLL, irrespective of clinical fitness,” Dr. Flinn and his colleagues wrote in their report.
The present phase 1b, dose-escalation study enrolled 32 patients who were previously untreated (median age, 63 years) and 46 patients who were relapsed or refractory to previous treatments (median age, 61 years).
Doses of venetoclax were escalated from 100 mg to 400 mg to determine its maximum tolerated dose when combined with obinutuzumab, the investigators wrote. Some patients received venetoclax first, while others received obinutuzumab first, for a total of 1 year of treatment.
The study confirmed favorable risk-benefit treatment used a dose of 400 mg venetoclax plus the standard dose of obinutuzumab, according to the researchers.
The overall best response rate was 95% for relapsed/refractory patients, including a 37% rate of complete response or complete response with incomplete marrow recovery. In previously untreated patients, the overall best response rate was 100%, including a 78% rate of complete responses by those criteria.
Undetectable minimal residual disease was observed in 64% of relapsed/refractory patients and 91% of previously untreated patients at 3 months after the last obinutuzumab dose, the investigators reported.
There were no dose-limiting toxicities in the study, no clinical tumor lysis syndrome, and no differences between the two schedules (venetoclax first or obinutuzumab first) in terms of adverse events, the investigators wrote.
Neutropenia was the most common serious (grade 3-4) adverse event, occurring in 58% of relapsed/refractory patients and 53% of patients treated in the first line. Grade 3-4 infections were seen in 29% and 13% of the relapsed/refractory and previously untreated patients, respectively.
There were no fatal infections among previously untreated patients, while three relapsed/refractory patients (7%) had fatal adverse events, including one case of acute respiratory failure in a patient with suspected Richter’s transformation, pneumonia in a patient with metastatic squamous cell lung carcinoma, and another case of pneumonia occurring about 3 months after the last dose of venetoclax.
Genentech and AbbVie provided financial support for the study. Dr. Flinn reported receiving research funding for his institution from Genentech, AbbVie, and several other companies.
SOURCE: Flinn IW et al. Blood. 2019 Mar 12. doi: 10.1182/blood-2019-01-896290.
FROM BLOOD
MRD status at transplant predicts outcomes in ALL patients
HOUSTON – Acute lymphoblastic leukemia patients with measurable residual disease (MRD) negativity prior to hematopoietic cell transplantation achieve better outcomes than do those who are MRD positive, particularly when total body irradiation (TBI)–based conditioning is used, a large retrospective study suggests.
Of 2,780 ALL patients who underwent hematopoietic cell transplantation (HCT) in first or second complete remission (CR), and who were included in the study, 1,816 were MRD negative before transplantation and 964 were MRD positive.
Overall, with follow-up of 40-44 months, MRD positivity was a significant independent predictor of lower overall survival (OS; hazard ratio, 1.19), leukemia-free survival (LFS; HR, 1.26), and higher relapse incidence (RI; 1.51), Arnon Nagler, MD, reported at the Transplantation & Cellular Therapy Meetings.
Conditioning was TBI-based in 76% of the patients; when these patients were compared with those who received chemotherapy-based conditioning, they were found to have better OS, LFS, and RI (HRs, 0.75, 0.70, and 0.60, respectively), said Dr. Nagler, director of both the division of hematology and the bone marrow transplantation and cord blood bank at the Chaim Sheba Medical Center, Tel-Hashomer, and professor of medicine at Tel Aviv University, both in Israel.
“There was no significant interaction between the MRD status and the conditioning,” he said.
On multivariate analysis, MRD positivity was found to be associated with lower OS and LFS (HRs, 1.26 and 1.3), and higher RI (HR, 1.53) in the TBI group, and with higher RI (HR 1.58) in the chemotherapy group, he said. There was no significant association between MRD and other outcomes in this last cohort, he added, noting that TBI-based conditioning was associated with improved OS, LFS, and RI in both MRD-negative and MRD-positive patients.
“MRD is an extremely important prognostic factor for ALL,” he said, noting that its prognostic value in this setting has been established in multiple studies, and that MRD measured at the end of induction is increasingly used to guide further therapy.
However, although MRD detectable immediately before HCT is known to be associated with poor outcomes, it has been unclear if – or to what extent – this differs with different types of conditioning, he added.
“So the aim of this study was to explore if MRD detectable before allogeneic HCT for ALL is associated with different outcomes in adult patients receiving myeloablative conditioning, either TBI or chemotherapy based,” he said at the meeting held by the American Society for Blood and Marrow Transplantation and the Center for International Blood and Marrow Transplant Research.
At its meeting, the American Society for Blood and Marrow Transplantation announced a new name for the society: American Society for Transplantation and Cellular Therapy (ASTCT).
Patients included in the analysis had a median age of 38 years and underwent HCT between 2000 and 2017 using sibling or unrelated 9/10 or 10/10 matched donors. None received blinatumomab or inotuzumab, Dr. Nagler said, adding that more patients are likely to achieve MRD negativity with these agents.
It will be interesting to see if the prognostic value of MRD will remain as strong with the new agents, and if TBI will be “a strong factor in overall survival and disease-free survival” with modern immunotherapy, he concluded.
The study was conducted on behalf of the Acute Leukemia Working Party of the European Society for Blood and Marrow Transplantation (EBMT).
Dr. Nagler reported having no relevant financial disclosures.
SOURCE: Nagler A et al. TCT 2019, Abstract 7.
HOUSTON – Acute lymphoblastic leukemia patients with measurable residual disease (MRD) negativity prior to hematopoietic cell transplantation achieve better outcomes than do those who are MRD positive, particularly when total body irradiation (TBI)–based conditioning is used, a large retrospective study suggests.
Of 2,780 ALL patients who underwent hematopoietic cell transplantation (HCT) in first or second complete remission (CR), and who were included in the study, 1,816 were MRD negative before transplantation and 964 were MRD positive.
Overall, with follow-up of 40-44 months, MRD positivity was a significant independent predictor of lower overall survival (OS; hazard ratio, 1.19), leukemia-free survival (LFS; HR, 1.26), and higher relapse incidence (RI; 1.51), Arnon Nagler, MD, reported at the Transplantation & Cellular Therapy Meetings.
Conditioning was TBI-based in 76% of the patients; when these patients were compared with those who received chemotherapy-based conditioning, they were found to have better OS, LFS, and RI (HRs, 0.75, 0.70, and 0.60, respectively), said Dr. Nagler, director of both the division of hematology and the bone marrow transplantation and cord blood bank at the Chaim Sheba Medical Center, Tel-Hashomer, and professor of medicine at Tel Aviv University, both in Israel.
“There was no significant interaction between the MRD status and the conditioning,” he said.
On multivariate analysis, MRD positivity was found to be associated with lower OS and LFS (HRs, 1.26 and 1.3), and higher RI (HR, 1.53) in the TBI group, and with higher RI (HR 1.58) in the chemotherapy group, he said. There was no significant association between MRD and other outcomes in this last cohort, he added, noting that TBI-based conditioning was associated with improved OS, LFS, and RI in both MRD-negative and MRD-positive patients.
“MRD is an extremely important prognostic factor for ALL,” he said, noting that its prognostic value in this setting has been established in multiple studies, and that MRD measured at the end of induction is increasingly used to guide further therapy.
However, although MRD detectable immediately before HCT is known to be associated with poor outcomes, it has been unclear if – or to what extent – this differs with different types of conditioning, he added.
“So the aim of this study was to explore if MRD detectable before allogeneic HCT for ALL is associated with different outcomes in adult patients receiving myeloablative conditioning, either TBI or chemotherapy based,” he said at the meeting held by the American Society for Blood and Marrow Transplantation and the Center for International Blood and Marrow Transplant Research.
At its meeting, the American Society for Blood and Marrow Transplantation announced a new name for the society: American Society for Transplantation and Cellular Therapy (ASTCT).
Patients included in the analysis had a median age of 38 years and underwent HCT between 2000 and 2017 using sibling or unrelated 9/10 or 10/10 matched donors. None received blinatumomab or inotuzumab, Dr. Nagler said, adding that more patients are likely to achieve MRD negativity with these agents.
It will be interesting to see if the prognostic value of MRD will remain as strong with the new agents, and if TBI will be “a strong factor in overall survival and disease-free survival” with modern immunotherapy, he concluded.
The study was conducted on behalf of the Acute Leukemia Working Party of the European Society for Blood and Marrow Transplantation (EBMT).
Dr. Nagler reported having no relevant financial disclosures.
SOURCE: Nagler A et al. TCT 2019, Abstract 7.
HOUSTON – Acute lymphoblastic leukemia patients with measurable residual disease (MRD) negativity prior to hematopoietic cell transplantation achieve better outcomes than do those who are MRD positive, particularly when total body irradiation (TBI)–based conditioning is used, a large retrospective study suggests.
Of 2,780 ALL patients who underwent hematopoietic cell transplantation (HCT) in first or second complete remission (CR), and who were included in the study, 1,816 were MRD negative before transplantation and 964 were MRD positive.
Overall, with follow-up of 40-44 months, MRD positivity was a significant independent predictor of lower overall survival (OS; hazard ratio, 1.19), leukemia-free survival (LFS; HR, 1.26), and higher relapse incidence (RI; 1.51), Arnon Nagler, MD, reported at the Transplantation & Cellular Therapy Meetings.
Conditioning was TBI-based in 76% of the patients; when these patients were compared with those who received chemotherapy-based conditioning, they were found to have better OS, LFS, and RI (HRs, 0.75, 0.70, and 0.60, respectively), said Dr. Nagler, director of both the division of hematology and the bone marrow transplantation and cord blood bank at the Chaim Sheba Medical Center, Tel-Hashomer, and professor of medicine at Tel Aviv University, both in Israel.
“There was no significant interaction between the MRD status and the conditioning,” he said.
On multivariate analysis, MRD positivity was found to be associated with lower OS and LFS (HRs, 1.26 and 1.3), and higher RI (HR, 1.53) in the TBI group, and with higher RI (HR 1.58) in the chemotherapy group, he said. There was no significant association between MRD and other outcomes in this last cohort, he added, noting that TBI-based conditioning was associated with improved OS, LFS, and RI in both MRD-negative and MRD-positive patients.
“MRD is an extremely important prognostic factor for ALL,” he said, noting that its prognostic value in this setting has been established in multiple studies, and that MRD measured at the end of induction is increasingly used to guide further therapy.
However, although MRD detectable immediately before HCT is known to be associated with poor outcomes, it has been unclear if – or to what extent – this differs with different types of conditioning, he added.
“So the aim of this study was to explore if MRD detectable before allogeneic HCT for ALL is associated with different outcomes in adult patients receiving myeloablative conditioning, either TBI or chemotherapy based,” he said at the meeting held by the American Society for Blood and Marrow Transplantation and the Center for International Blood and Marrow Transplant Research.
At its meeting, the American Society for Blood and Marrow Transplantation announced a new name for the society: American Society for Transplantation and Cellular Therapy (ASTCT).
Patients included in the analysis had a median age of 38 years and underwent HCT between 2000 and 2017 using sibling or unrelated 9/10 or 10/10 matched donors. None received blinatumomab or inotuzumab, Dr. Nagler said, adding that more patients are likely to achieve MRD negativity with these agents.
It will be interesting to see if the prognostic value of MRD will remain as strong with the new agents, and if TBI will be “a strong factor in overall survival and disease-free survival” with modern immunotherapy, he concluded.
The study was conducted on behalf of the Acute Leukemia Working Party of the European Society for Blood and Marrow Transplantation (EBMT).
Dr. Nagler reported having no relevant financial disclosures.
SOURCE: Nagler A et al. TCT 2019, Abstract 7.
REPORTING FROM TCT 2019
Secondary AML in first remission predicts outcomes
HOUSTON – Secondary acute myeloid leukemia (sAML) predicts outcomes after stem cell transplantation in first complete remission, whereas factors such as age, cytogenetics, and performance status are more relevant predictors of outcomes in patients with de novo AML, according to a large, registry-based analysis.
Of 11,439 patients with de novo AML and 1,325 with sAML identified in the registry, 7,691 and 909, respectively, underwent a stem cell transplant (SCT) in first complete remission (CR1), Bipin Savani, MD, said at the Transplantation & Cellular Therapies Meetings.
The 3-year cumulative incidence of relapse (CIR) and nonrelapse mortality (NRM) rates in those who underwent SCT in CR1 were higher in the sAML versus de novo AML groups (35% vs. 28.5% for CIR and 23.4% vs. 16.4% for NRM, respectively), said Dr. Savani, professor of medicine, director of the Long-Term Transplant Clinic, and medical director of the Stem Cell Transplant Processing Laboratory at Vanderbilt University Medical Center & Veterans Affairs Medical Center, Nashville, Tenn.
The 3-year overall survival (OS), leukemia-free survival (LFS), and graft-versus-host disease/relapse-free survival (GRFS) were significantly lower in the sAML group versus the de novo AML group (46.7% vs. 60.8% for OS; 41.6% vs. 55.1% for LFS; and 28.4% vs. 28.6% for GRFS).
Multivariate analysis controlling for risk factors and stratified by disease stage at SCT showed that sAML in CR1 was significantly associated with higher NRM (hazard ratio, 1.32) and CIR (HR, 1.28), and with lower LFS (HR, 1.30), OS (HR, 1.32) and GRFS (HR, 1.20).
Other significant predictors of OS in the model were age, cytogenetics, patient/donor sex combination, Karnofsky performance status (KPS), and donor, he said at the meeting held by the American Society for Blood and Marrow Transplantation and the Center for International Blood and Marrow Transplant Research. At its meeting, the American Society for Blood and Marrow Transplantation announced a new name for the society: American Society for Transplantation and Cellular Therapy (ASTCT).
In the patients who underwent SCT for primary refractory AML (607 with de novo AML and 199 with sAML) or relapsed AML (1,009 with de novo AML and 124 with sAML), the outcomes were generally inferior to those seen with SCT in CR1. However, sAML in those patients did not predict outcomes, Dr. Savani said, noting that outcome in those cases were predicted by age, cytogenetics, and KPS.
In an analysis of 877 pairs matched for age, disease stage at SCT, KPS, conditioning, in vivo/ex vivo T-cell depletion, donor, donor/recipient sex and cytomegalovirus-status combination, cytogenetics, and graft source, the finding that sAML was associated with significantly higher NRM, and lower LFS, OS, and GRFS overall was confirmed.
However, stratification by stage at the time of SCT again showed that the differences between groups were only seen among those transplanted in CR1, and not in those with advanced disease at the time of transplant.
Patients included in the study were adults aged 18 years and older who underwent SCT for de novo or sAML from a matched related, unrelated, or T-cell replete haploidentical donor between 2000 and 2016.
The findings confirm the general belief that the prognosis in AML secondary to another hematologic neoplasia or malignant disease is poorer than that for de novo AML, and clarify the role of this difference for SCT, Dr. Savani said.
“These data may help to improve risk stratification and prognostic estimates after allogeneic hematopoietic cell transplantation for acute myeloid leukemia,” he concluded.
Dr. Savani reported having no financial disclosures.
SOURCE: Savani B et al. TCT 2019, Abstract 12.
HOUSTON – Secondary acute myeloid leukemia (sAML) predicts outcomes after stem cell transplantation in first complete remission, whereas factors such as age, cytogenetics, and performance status are more relevant predictors of outcomes in patients with de novo AML, according to a large, registry-based analysis.
Of 11,439 patients with de novo AML and 1,325 with sAML identified in the registry, 7,691 and 909, respectively, underwent a stem cell transplant (SCT) in first complete remission (CR1), Bipin Savani, MD, said at the Transplantation & Cellular Therapies Meetings.
The 3-year cumulative incidence of relapse (CIR) and nonrelapse mortality (NRM) rates in those who underwent SCT in CR1 were higher in the sAML versus de novo AML groups (35% vs. 28.5% for CIR and 23.4% vs. 16.4% for NRM, respectively), said Dr. Savani, professor of medicine, director of the Long-Term Transplant Clinic, and medical director of the Stem Cell Transplant Processing Laboratory at Vanderbilt University Medical Center & Veterans Affairs Medical Center, Nashville, Tenn.
The 3-year overall survival (OS), leukemia-free survival (LFS), and graft-versus-host disease/relapse-free survival (GRFS) were significantly lower in the sAML group versus the de novo AML group (46.7% vs. 60.8% for OS; 41.6% vs. 55.1% for LFS; and 28.4% vs. 28.6% for GRFS).
Multivariate analysis controlling for risk factors and stratified by disease stage at SCT showed that sAML in CR1 was significantly associated with higher NRM (hazard ratio, 1.32) and CIR (HR, 1.28), and with lower LFS (HR, 1.30), OS (HR, 1.32) and GRFS (HR, 1.20).
Other significant predictors of OS in the model were age, cytogenetics, patient/donor sex combination, Karnofsky performance status (KPS), and donor, he said at the meeting held by the American Society for Blood and Marrow Transplantation and the Center for International Blood and Marrow Transplant Research. At its meeting, the American Society for Blood and Marrow Transplantation announced a new name for the society: American Society for Transplantation and Cellular Therapy (ASTCT).
In the patients who underwent SCT for primary refractory AML (607 with de novo AML and 199 with sAML) or relapsed AML (1,009 with de novo AML and 124 with sAML), the outcomes were generally inferior to those seen with SCT in CR1. However, sAML in those patients did not predict outcomes, Dr. Savani said, noting that outcome in those cases were predicted by age, cytogenetics, and KPS.
In an analysis of 877 pairs matched for age, disease stage at SCT, KPS, conditioning, in vivo/ex vivo T-cell depletion, donor, donor/recipient sex and cytomegalovirus-status combination, cytogenetics, and graft source, the finding that sAML was associated with significantly higher NRM, and lower LFS, OS, and GRFS overall was confirmed.
However, stratification by stage at the time of SCT again showed that the differences between groups were only seen among those transplanted in CR1, and not in those with advanced disease at the time of transplant.
Patients included in the study were adults aged 18 years and older who underwent SCT for de novo or sAML from a matched related, unrelated, or T-cell replete haploidentical donor between 2000 and 2016.
The findings confirm the general belief that the prognosis in AML secondary to another hematologic neoplasia or malignant disease is poorer than that for de novo AML, and clarify the role of this difference for SCT, Dr. Savani said.
“These data may help to improve risk stratification and prognostic estimates after allogeneic hematopoietic cell transplantation for acute myeloid leukemia,” he concluded.
Dr. Savani reported having no financial disclosures.
SOURCE: Savani B et al. TCT 2019, Abstract 12.
HOUSTON – Secondary acute myeloid leukemia (sAML) predicts outcomes after stem cell transplantation in first complete remission, whereas factors such as age, cytogenetics, and performance status are more relevant predictors of outcomes in patients with de novo AML, according to a large, registry-based analysis.
Of 11,439 patients with de novo AML and 1,325 with sAML identified in the registry, 7,691 and 909, respectively, underwent a stem cell transplant (SCT) in first complete remission (CR1), Bipin Savani, MD, said at the Transplantation & Cellular Therapies Meetings.
The 3-year cumulative incidence of relapse (CIR) and nonrelapse mortality (NRM) rates in those who underwent SCT in CR1 were higher in the sAML versus de novo AML groups (35% vs. 28.5% for CIR and 23.4% vs. 16.4% for NRM, respectively), said Dr. Savani, professor of medicine, director of the Long-Term Transplant Clinic, and medical director of the Stem Cell Transplant Processing Laboratory at Vanderbilt University Medical Center & Veterans Affairs Medical Center, Nashville, Tenn.
The 3-year overall survival (OS), leukemia-free survival (LFS), and graft-versus-host disease/relapse-free survival (GRFS) were significantly lower in the sAML group versus the de novo AML group (46.7% vs. 60.8% for OS; 41.6% vs. 55.1% for LFS; and 28.4% vs. 28.6% for GRFS).
Multivariate analysis controlling for risk factors and stratified by disease stage at SCT showed that sAML in CR1 was significantly associated with higher NRM (hazard ratio, 1.32) and CIR (HR, 1.28), and with lower LFS (HR, 1.30), OS (HR, 1.32) and GRFS (HR, 1.20).
Other significant predictors of OS in the model were age, cytogenetics, patient/donor sex combination, Karnofsky performance status (KPS), and donor, he said at the meeting held by the American Society for Blood and Marrow Transplantation and the Center for International Blood and Marrow Transplant Research. At its meeting, the American Society for Blood and Marrow Transplantation announced a new name for the society: American Society for Transplantation and Cellular Therapy (ASTCT).
In the patients who underwent SCT for primary refractory AML (607 with de novo AML and 199 with sAML) or relapsed AML (1,009 with de novo AML and 124 with sAML), the outcomes were generally inferior to those seen with SCT in CR1. However, sAML in those patients did not predict outcomes, Dr. Savani said, noting that outcome in those cases were predicted by age, cytogenetics, and KPS.
In an analysis of 877 pairs matched for age, disease stage at SCT, KPS, conditioning, in vivo/ex vivo T-cell depletion, donor, donor/recipient sex and cytomegalovirus-status combination, cytogenetics, and graft source, the finding that sAML was associated with significantly higher NRM, and lower LFS, OS, and GRFS overall was confirmed.
However, stratification by stage at the time of SCT again showed that the differences between groups were only seen among those transplanted in CR1, and not in those with advanced disease at the time of transplant.
Patients included in the study were adults aged 18 years and older who underwent SCT for de novo or sAML from a matched related, unrelated, or T-cell replete haploidentical donor between 2000 and 2016.
The findings confirm the general belief that the prognosis in AML secondary to another hematologic neoplasia or malignant disease is poorer than that for de novo AML, and clarify the role of this difference for SCT, Dr. Savani said.
“These data may help to improve risk stratification and prognostic estimates after allogeneic hematopoietic cell transplantation for acute myeloid leukemia,” he concluded.
Dr. Savani reported having no financial disclosures.
SOURCE: Savani B et al. TCT 2019, Abstract 12.
REPORTING FROM TCT 2019
Haplo-HSCT bests chemotherapy for MRD-positive adult ALL
HOUSTON – Haploidentical stem cell transplantation (Haplo-HSCT) outperforms chemotherapy for the treatment of adults with acute lymphoblastic leukemia (ALL) in first complete remission, findings from a prospective multicenter trial suggest.
The 2-year leukemia-free survival (LFS) was about 70% in 49 patients in first remission who received haplo-HSCT vs. 40% in 40 patients who received chemotherapy, and 2-year overall survival (OS) was about 80% vs. 50% in the groups, respectively, Meng Lv, MD, PhD, of Peking University People’s Hospital in Beijing reported at the Transplantation & Cellular Therapy Meetings.
“This result is comparable to results of our previous reports,” he said at the meeting held by the American Society for Blood and Marrow Transplantation and the Center for International Blood and Marrow Transplant Research.
He noted that the findings also support those from other institutions.
Study subjects initially included 112 newly diagnosed standard-risk ALL patients aged 18-39 years without high-risk features who achieved complete remission (CR) after one or two cycles of induction. They were consecutively enrolled at five centers in China, including high-volume centers, between July 2014 and June 2017 and were followed for a median of 24.6 months.
Subjects without a suitable HLA-matched sibling donor (MSD) or HLA-matched unrelated donor after two cycles of consolidation with hyper-CVAD chemotherapy were eligible for haplo-HSCT or further hyper-CVAD chemotherapy.
The final analysis included 89 patients after 23 were excluded because of early relapse (6 patients) or a decision to undergo MSD HSCT (16 patients), or unrelated donor-HSCT (1 patient), Dr. Lv said, noting that landmark analysis was used when comparing the outcomes of patients receiving haplo-HSCT with those receiving chemotherapy.
Multivariate analysis with adjustment for a propensity score calculated for each patient showed that treatment (haplo-HSCT vs. chemotherapy) independently predicted LFS (hazard ratio, 0.388), OS (HR, 0.346), and cumulative incidence of relapse (CIR; HR, 0.247). Minimal residual disease (MRD) positivity after the first consolidation was an independent risk factor for LFS (HR, 2.162) and CIR (HR, 3.667). Additionally, diagnosis (T- vs. B-cell) was an independent risk factor for OS (HR, 2.267), Dr. Lv said, adding that nonrelapse mortality was similar in the groups in the propensity score–adjusted analysis.
The findings overall show that haplo-HSCT has variable impact on survival in standard-risk ALL, when compared with traditional chemotherapy, with subgroup analyses showing MRD-positive patients deriving the greatest benefit, he said. Future studies are planned to look more closely at MRD-positive disease and the possible benefits of postponing transplant until the second CR.
At its meeting, the American Society for Blood and Marrow Transplantation announced a new name for the society: American Society for Transplantation and Cellular Therapy (ASTCT).
Dr. Lv reported having no financial disclosures.
SOURCE: Lv M et al. TCT 2019, Abstract 8.
HOUSTON – Haploidentical stem cell transplantation (Haplo-HSCT) outperforms chemotherapy for the treatment of adults with acute lymphoblastic leukemia (ALL) in first complete remission, findings from a prospective multicenter trial suggest.
The 2-year leukemia-free survival (LFS) was about 70% in 49 patients in first remission who received haplo-HSCT vs. 40% in 40 patients who received chemotherapy, and 2-year overall survival (OS) was about 80% vs. 50% in the groups, respectively, Meng Lv, MD, PhD, of Peking University People’s Hospital in Beijing reported at the Transplantation & Cellular Therapy Meetings.
“This result is comparable to results of our previous reports,” he said at the meeting held by the American Society for Blood and Marrow Transplantation and the Center for International Blood and Marrow Transplant Research.
He noted that the findings also support those from other institutions.
Study subjects initially included 112 newly diagnosed standard-risk ALL patients aged 18-39 years without high-risk features who achieved complete remission (CR) after one or two cycles of induction. They were consecutively enrolled at five centers in China, including high-volume centers, between July 2014 and June 2017 and were followed for a median of 24.6 months.
Subjects without a suitable HLA-matched sibling donor (MSD) or HLA-matched unrelated donor after two cycles of consolidation with hyper-CVAD chemotherapy were eligible for haplo-HSCT or further hyper-CVAD chemotherapy.
The final analysis included 89 patients after 23 were excluded because of early relapse (6 patients) or a decision to undergo MSD HSCT (16 patients), or unrelated donor-HSCT (1 patient), Dr. Lv said, noting that landmark analysis was used when comparing the outcomes of patients receiving haplo-HSCT with those receiving chemotherapy.
Multivariate analysis with adjustment for a propensity score calculated for each patient showed that treatment (haplo-HSCT vs. chemotherapy) independently predicted LFS (hazard ratio, 0.388), OS (HR, 0.346), and cumulative incidence of relapse (CIR; HR, 0.247). Minimal residual disease (MRD) positivity after the first consolidation was an independent risk factor for LFS (HR, 2.162) and CIR (HR, 3.667). Additionally, diagnosis (T- vs. B-cell) was an independent risk factor for OS (HR, 2.267), Dr. Lv said, adding that nonrelapse mortality was similar in the groups in the propensity score–adjusted analysis.
The findings overall show that haplo-HSCT has variable impact on survival in standard-risk ALL, when compared with traditional chemotherapy, with subgroup analyses showing MRD-positive patients deriving the greatest benefit, he said. Future studies are planned to look more closely at MRD-positive disease and the possible benefits of postponing transplant until the second CR.
At its meeting, the American Society for Blood and Marrow Transplantation announced a new name for the society: American Society for Transplantation and Cellular Therapy (ASTCT).
Dr. Lv reported having no financial disclosures.
SOURCE: Lv M et al. TCT 2019, Abstract 8.
HOUSTON – Haploidentical stem cell transplantation (Haplo-HSCT) outperforms chemotherapy for the treatment of adults with acute lymphoblastic leukemia (ALL) in first complete remission, findings from a prospective multicenter trial suggest.
The 2-year leukemia-free survival (LFS) was about 70% in 49 patients in first remission who received haplo-HSCT vs. 40% in 40 patients who received chemotherapy, and 2-year overall survival (OS) was about 80% vs. 50% in the groups, respectively, Meng Lv, MD, PhD, of Peking University People’s Hospital in Beijing reported at the Transplantation & Cellular Therapy Meetings.
“This result is comparable to results of our previous reports,” he said at the meeting held by the American Society for Blood and Marrow Transplantation and the Center for International Blood and Marrow Transplant Research.
He noted that the findings also support those from other institutions.
Study subjects initially included 112 newly diagnosed standard-risk ALL patients aged 18-39 years without high-risk features who achieved complete remission (CR) after one or two cycles of induction. They were consecutively enrolled at five centers in China, including high-volume centers, between July 2014 and June 2017 and were followed for a median of 24.6 months.
Subjects without a suitable HLA-matched sibling donor (MSD) or HLA-matched unrelated donor after two cycles of consolidation with hyper-CVAD chemotherapy were eligible for haplo-HSCT or further hyper-CVAD chemotherapy.
The final analysis included 89 patients after 23 were excluded because of early relapse (6 patients) or a decision to undergo MSD HSCT (16 patients), or unrelated donor-HSCT (1 patient), Dr. Lv said, noting that landmark analysis was used when comparing the outcomes of patients receiving haplo-HSCT with those receiving chemotherapy.
Multivariate analysis with adjustment for a propensity score calculated for each patient showed that treatment (haplo-HSCT vs. chemotherapy) independently predicted LFS (hazard ratio, 0.388), OS (HR, 0.346), and cumulative incidence of relapse (CIR; HR, 0.247). Minimal residual disease (MRD) positivity after the first consolidation was an independent risk factor for LFS (HR, 2.162) and CIR (HR, 3.667). Additionally, diagnosis (T- vs. B-cell) was an independent risk factor for OS (HR, 2.267), Dr. Lv said, adding that nonrelapse mortality was similar in the groups in the propensity score–adjusted analysis.
The findings overall show that haplo-HSCT has variable impact on survival in standard-risk ALL, when compared with traditional chemotherapy, with subgroup analyses showing MRD-positive patients deriving the greatest benefit, he said. Future studies are planned to look more closely at MRD-positive disease and the possible benefits of postponing transplant until the second CR.
At its meeting, the American Society for Blood and Marrow Transplantation announced a new name for the society: American Society for Transplantation and Cellular Therapy (ASTCT).
Dr. Lv reported having no financial disclosures.
SOURCE: Lv M et al. TCT 2019, Abstract 8.
REPORTING FROM TCT 2019
Anti-CD45 conditioning looks safe, feasible in relapsed AML
HOUSTON – A novel anti-CD45 targeted conditioning regimen is feasible for use in older patients with relapsed or refractory acute myeloid leukemia undergoing transplant, according to preliminary results of a randomized, phase 3 trial.
Treatment with iodine-131 apamistamab (Iomab-B) has thus far has resulted in successful engraftment for all patients who have received it and gone on to transplant, despite active disease and high bone marrow blast burden prior to transplantation, according to Sergio A. Giralt, MD, of Memorial Sloan Kettering Cancer Center, New York.
There has been no nonrelapse mortality related to the novel regimen in the ongoing trial, which compares Iomab-B as targeted conditioning prior to allogeneic hematopoietic stem cell transplant (HCT) with standard of care regimens, Dr. Giralt said in an update on the trial, known as SIERRA.
SIERRA is the only ongoing, randomized, phase 3 clinical trial to offer a transplant option in patients aged 55 years or older with active relapsed or refractory acute myeloid leukemia (AML), Dr. Giralt said at the Transplantation & Cellular Therapy Meetings.
“This is an underserved population in which traditional transplant techniques have very poor results, and there are limited options for patients with active disease,” Dr. Giralt said. “Of note, many transplant centers today do not consider these patients eligible for transplant.”
A total of 150 patients are to be enrolled in SIERRA and randomized either to investigator’s choice of salvage induction chemotherapy including approved targeted agents or to the experimental arm, which consists of an individualized dose of Iomab-B 12 days prior to HCT with fludarabine and total body irradiation as transplant conditioning.
Dr. Giralt presented an update on the first 38 patients in SIERRA, representing 25% of the total enrollment target.
Of 18 patients randomized to Iomab-B and transplanted, the median number of days to absolute neutrophil count engraftment was 13, Dr. Giralt said.
In the control arm, 15 of 19 (79%) failed to achieve complete remission, and of those 10 crossed over to receive Iomab-B and transplant. Days to engraftment, full donor chimerism, and dose delivered to the bone marrow were all similar in the crossover group, compared with those initially randomized to the novel therapy.
Nonhematologic grade 3 or 4 toxicities were similar between the Iomab-B arm and the conventional care arm, and included febrile neutropenia, stomatitis, and other side effects typical for these patients.
There were no grade 3 or 4 Iomab-B infusion-related reactions, and four mild cases of chronic graft-versus-host disease occurred in Iomab-B treated patients.
Nonrelapse mortality was “extremely low” with no cases in the 100 days post transplant in those initially randomized to Iomab-B, and only one case in a crossover patient, Dr. Giralt said.
“These results are encouraging, and can broaden transplant eligibility and improve outcomes,” he added.
The meeting is held by the American Society for Blood and Marrow Transplantation and the Center for International Blood and Marrow Transplant Research. At its meeting, the American Society for Blood and Marrow Transplantation announced a new name for the society: American Society for Transplantation and Cellular Therapy (ASTCT).
The SIERRA study is supported by Actinium Pharmaceuticals. Dr. Giralt reported disclosures related to Actinium and several other companies.
SOURCE: Giralt SA et al. TCT 2019, Abstract LBA3.
HOUSTON – A novel anti-CD45 targeted conditioning regimen is feasible for use in older patients with relapsed or refractory acute myeloid leukemia undergoing transplant, according to preliminary results of a randomized, phase 3 trial.
Treatment with iodine-131 apamistamab (Iomab-B) has thus far has resulted in successful engraftment for all patients who have received it and gone on to transplant, despite active disease and high bone marrow blast burden prior to transplantation, according to Sergio A. Giralt, MD, of Memorial Sloan Kettering Cancer Center, New York.
There has been no nonrelapse mortality related to the novel regimen in the ongoing trial, which compares Iomab-B as targeted conditioning prior to allogeneic hematopoietic stem cell transplant (HCT) with standard of care regimens, Dr. Giralt said in an update on the trial, known as SIERRA.
SIERRA is the only ongoing, randomized, phase 3 clinical trial to offer a transplant option in patients aged 55 years or older with active relapsed or refractory acute myeloid leukemia (AML), Dr. Giralt said at the Transplantation & Cellular Therapy Meetings.
“This is an underserved population in which traditional transplant techniques have very poor results, and there are limited options for patients with active disease,” Dr. Giralt said. “Of note, many transplant centers today do not consider these patients eligible for transplant.”
A total of 150 patients are to be enrolled in SIERRA and randomized either to investigator’s choice of salvage induction chemotherapy including approved targeted agents or to the experimental arm, which consists of an individualized dose of Iomab-B 12 days prior to HCT with fludarabine and total body irradiation as transplant conditioning.
Dr. Giralt presented an update on the first 38 patients in SIERRA, representing 25% of the total enrollment target.
Of 18 patients randomized to Iomab-B and transplanted, the median number of days to absolute neutrophil count engraftment was 13, Dr. Giralt said.
In the control arm, 15 of 19 (79%) failed to achieve complete remission, and of those 10 crossed over to receive Iomab-B and transplant. Days to engraftment, full donor chimerism, and dose delivered to the bone marrow were all similar in the crossover group, compared with those initially randomized to the novel therapy.
Nonhematologic grade 3 or 4 toxicities were similar between the Iomab-B arm and the conventional care arm, and included febrile neutropenia, stomatitis, and other side effects typical for these patients.
There were no grade 3 or 4 Iomab-B infusion-related reactions, and four mild cases of chronic graft-versus-host disease occurred in Iomab-B treated patients.
Nonrelapse mortality was “extremely low” with no cases in the 100 days post transplant in those initially randomized to Iomab-B, and only one case in a crossover patient, Dr. Giralt said.
“These results are encouraging, and can broaden transplant eligibility and improve outcomes,” he added.
The meeting is held by the American Society for Blood and Marrow Transplantation and the Center for International Blood and Marrow Transplant Research. At its meeting, the American Society for Blood and Marrow Transplantation announced a new name for the society: American Society for Transplantation and Cellular Therapy (ASTCT).
The SIERRA study is supported by Actinium Pharmaceuticals. Dr. Giralt reported disclosures related to Actinium and several other companies.
SOURCE: Giralt SA et al. TCT 2019, Abstract LBA3.
HOUSTON – A novel anti-CD45 targeted conditioning regimen is feasible for use in older patients with relapsed or refractory acute myeloid leukemia undergoing transplant, according to preliminary results of a randomized, phase 3 trial.
Treatment with iodine-131 apamistamab (Iomab-B) has thus far has resulted in successful engraftment for all patients who have received it and gone on to transplant, despite active disease and high bone marrow blast burden prior to transplantation, according to Sergio A. Giralt, MD, of Memorial Sloan Kettering Cancer Center, New York.
There has been no nonrelapse mortality related to the novel regimen in the ongoing trial, which compares Iomab-B as targeted conditioning prior to allogeneic hematopoietic stem cell transplant (HCT) with standard of care regimens, Dr. Giralt said in an update on the trial, known as SIERRA.
SIERRA is the only ongoing, randomized, phase 3 clinical trial to offer a transplant option in patients aged 55 years or older with active relapsed or refractory acute myeloid leukemia (AML), Dr. Giralt said at the Transplantation & Cellular Therapy Meetings.
“This is an underserved population in which traditional transplant techniques have very poor results, and there are limited options for patients with active disease,” Dr. Giralt said. “Of note, many transplant centers today do not consider these patients eligible for transplant.”
A total of 150 patients are to be enrolled in SIERRA and randomized either to investigator’s choice of salvage induction chemotherapy including approved targeted agents or to the experimental arm, which consists of an individualized dose of Iomab-B 12 days prior to HCT with fludarabine and total body irradiation as transplant conditioning.
Dr. Giralt presented an update on the first 38 patients in SIERRA, representing 25% of the total enrollment target.
Of 18 patients randomized to Iomab-B and transplanted, the median number of days to absolute neutrophil count engraftment was 13, Dr. Giralt said.
In the control arm, 15 of 19 (79%) failed to achieve complete remission, and of those 10 crossed over to receive Iomab-B and transplant. Days to engraftment, full donor chimerism, and dose delivered to the bone marrow were all similar in the crossover group, compared with those initially randomized to the novel therapy.
Nonhematologic grade 3 or 4 toxicities were similar between the Iomab-B arm and the conventional care arm, and included febrile neutropenia, stomatitis, and other side effects typical for these patients.
There were no grade 3 or 4 Iomab-B infusion-related reactions, and four mild cases of chronic graft-versus-host disease occurred in Iomab-B treated patients.
Nonrelapse mortality was “extremely low” with no cases in the 100 days post transplant in those initially randomized to Iomab-B, and only one case in a crossover patient, Dr. Giralt said.
“These results are encouraging, and can broaden transplant eligibility and improve outcomes,” he added.
The meeting is held by the American Society for Blood and Marrow Transplantation and the Center for International Blood and Marrow Transplant Research. At its meeting, the American Society for Blood and Marrow Transplantation announced a new name for the society: American Society for Transplantation and Cellular Therapy (ASTCT).
The SIERRA study is supported by Actinium Pharmaceuticals. Dr. Giralt reported disclosures related to Actinium and several other companies.
SOURCE: Giralt SA et al. TCT 2019, Abstract LBA3.
REPORTING FROM TCT 2019
HDACi, HMA combo improves survival for older AML patients
For patients older than 65 years with newly diagnosed acute myeloid leukemia (AML) who were ineligible for standard induction therapy, adding the investigational pan-histone deacetylase (pan-HDAC) inhibitor pracinostat to azacitidine resulted in better complete remission and overall survival rates than azacitidine alone, results of a multicenter phase 2 trial showed.
Among 50 patients treated with the combination, 26 (52%) achieved the primary endpoint of either a complete remission (CR), CR with incomplete recovery of blood counts (CRi), or morphologic leukemia-free state (MLFS).
The median overall survival (OS) was 19.1 months, which compares favorably with historical data on similar patients treated with single-agent azacitidine, reported Guillermo Garcia-Manero, MD, from the University of Texas MD Anderson Cancer Center in Houston and his colleagues.
“[T]his study shows that pracinostat in combination with azacitidine has the potential to be a safe and effective regimen in the frontline treatment of older patients with AML unfit for [induction chemotherapy],” they wrote in Blood Advances.
Pracinostat is an oral pan-HDAC inhibitor that has been shown to have modest activity against AML as a single agent, but synergistic activity when combined with hypomethylating agent azacitidine, a standard of care for older patients with AML in the trial.
The investigators enrolled 50 patients with a median age of 75 years (range, 66-84 years). The cohort included 33 patients with de novo AML, 12 with AML secondary to myelodysplasia syndrome or myleoproliferative neoplasia, and five with therapy-related AML.
The patients were treated with pracinostat 60 mg daily for 3 days each week for 3 consecutive weeks in addition to azacitidine 75 mg/m2 daily for 7 days in a 28-day cycle.
As noted, 26 patients reached the clinical endpoint, including 21 with a CR, 2 with a CRi, and 3 with MLFS. Additionally, two patients had a partial response (PR) and four had a PR with incomplete recovery of blood counts.
The median OS was 19.1 months, and the median progression-free survival (PFS) was 12.6 months. The 1-year OS rate was 62%. The 60-day mortality rate was 10%.
The authors noted that the survival data were superior to those seen in the phase 3 AZA-AML-001 study, which compared azacitidine therapy with conventional regimens in patients older than 65 years with newly diagnosed AML who were not eligible for stem cell transplants. In that trial, median OS was 10.4 months, the CR rate was 19.5% (vs. 49% in the present study), the 1-year OS rate was 46.5%, and the 60-day mortality rate was 16.2%.
They acknowledged, however, that the validity of the comparison is limited by their study’s small sample size, potential differences between the study populations, and lack of a control group in the present study. The investigators also found that clearance rates of baseline somatic mutations correlated with response to treatment.
Grade 3 or greater treatment-emergent adverse events occurred in 43 of the 50 patients, including infections, thrombocytopenias, and febrile neutropenias.
“On the basis of these encouraging results, a phase 3, multicenter, double-blind, randomized study of pracinostat vs. placebo with azacitidine (NCT03151408) is currently ongoing to confirm superiority of the combination in this difficult-to-treat AML population,” the investigators wrote.
The study was supported by research funding from MEI Pharma, which helped develop pracinostat. Dr. Garcia-Manero reported having no disclosures. Multiple coauthors reported financial relationships with MEI and others. One coauthor is an MEI employee.
SOURCE: Garcia-Manero G et al. Blood Adv. 2019 Feb 26;3(4):508-18.
For patients older than 65 years with newly diagnosed acute myeloid leukemia (AML) who were ineligible for standard induction therapy, adding the investigational pan-histone deacetylase (pan-HDAC) inhibitor pracinostat to azacitidine resulted in better complete remission and overall survival rates than azacitidine alone, results of a multicenter phase 2 trial showed.
Among 50 patients treated with the combination, 26 (52%) achieved the primary endpoint of either a complete remission (CR), CR with incomplete recovery of blood counts (CRi), or morphologic leukemia-free state (MLFS).
The median overall survival (OS) was 19.1 months, which compares favorably with historical data on similar patients treated with single-agent azacitidine, reported Guillermo Garcia-Manero, MD, from the University of Texas MD Anderson Cancer Center in Houston and his colleagues.
“[T]his study shows that pracinostat in combination with azacitidine has the potential to be a safe and effective regimen in the frontline treatment of older patients with AML unfit for [induction chemotherapy],” they wrote in Blood Advances.
Pracinostat is an oral pan-HDAC inhibitor that has been shown to have modest activity against AML as a single agent, but synergistic activity when combined with hypomethylating agent azacitidine, a standard of care for older patients with AML in the trial.
The investigators enrolled 50 patients with a median age of 75 years (range, 66-84 years). The cohort included 33 patients with de novo AML, 12 with AML secondary to myelodysplasia syndrome or myleoproliferative neoplasia, and five with therapy-related AML.
The patients were treated with pracinostat 60 mg daily for 3 days each week for 3 consecutive weeks in addition to azacitidine 75 mg/m2 daily for 7 days in a 28-day cycle.
As noted, 26 patients reached the clinical endpoint, including 21 with a CR, 2 with a CRi, and 3 with MLFS. Additionally, two patients had a partial response (PR) and four had a PR with incomplete recovery of blood counts.
The median OS was 19.1 months, and the median progression-free survival (PFS) was 12.6 months. The 1-year OS rate was 62%. The 60-day mortality rate was 10%.
The authors noted that the survival data were superior to those seen in the phase 3 AZA-AML-001 study, which compared azacitidine therapy with conventional regimens in patients older than 65 years with newly diagnosed AML who were not eligible for stem cell transplants. In that trial, median OS was 10.4 months, the CR rate was 19.5% (vs. 49% in the present study), the 1-year OS rate was 46.5%, and the 60-day mortality rate was 16.2%.
They acknowledged, however, that the validity of the comparison is limited by their study’s small sample size, potential differences between the study populations, and lack of a control group in the present study. The investigators also found that clearance rates of baseline somatic mutations correlated with response to treatment.
Grade 3 or greater treatment-emergent adverse events occurred in 43 of the 50 patients, including infections, thrombocytopenias, and febrile neutropenias.
“On the basis of these encouraging results, a phase 3, multicenter, double-blind, randomized study of pracinostat vs. placebo with azacitidine (NCT03151408) is currently ongoing to confirm superiority of the combination in this difficult-to-treat AML population,” the investigators wrote.
The study was supported by research funding from MEI Pharma, which helped develop pracinostat. Dr. Garcia-Manero reported having no disclosures. Multiple coauthors reported financial relationships with MEI and others. One coauthor is an MEI employee.
SOURCE: Garcia-Manero G et al. Blood Adv. 2019 Feb 26;3(4):508-18.
For patients older than 65 years with newly diagnosed acute myeloid leukemia (AML) who were ineligible for standard induction therapy, adding the investigational pan-histone deacetylase (pan-HDAC) inhibitor pracinostat to azacitidine resulted in better complete remission and overall survival rates than azacitidine alone, results of a multicenter phase 2 trial showed.
Among 50 patients treated with the combination, 26 (52%) achieved the primary endpoint of either a complete remission (CR), CR with incomplete recovery of blood counts (CRi), or morphologic leukemia-free state (MLFS).
The median overall survival (OS) was 19.1 months, which compares favorably with historical data on similar patients treated with single-agent azacitidine, reported Guillermo Garcia-Manero, MD, from the University of Texas MD Anderson Cancer Center in Houston and his colleagues.
“[T]his study shows that pracinostat in combination with azacitidine has the potential to be a safe and effective regimen in the frontline treatment of older patients with AML unfit for [induction chemotherapy],” they wrote in Blood Advances.
Pracinostat is an oral pan-HDAC inhibitor that has been shown to have modest activity against AML as a single agent, but synergistic activity when combined with hypomethylating agent azacitidine, a standard of care for older patients with AML in the trial.
The investigators enrolled 50 patients with a median age of 75 years (range, 66-84 years). The cohort included 33 patients with de novo AML, 12 with AML secondary to myelodysplasia syndrome or myleoproliferative neoplasia, and five with therapy-related AML.
The patients were treated with pracinostat 60 mg daily for 3 days each week for 3 consecutive weeks in addition to azacitidine 75 mg/m2 daily for 7 days in a 28-day cycle.
As noted, 26 patients reached the clinical endpoint, including 21 with a CR, 2 with a CRi, and 3 with MLFS. Additionally, two patients had a partial response (PR) and four had a PR with incomplete recovery of blood counts.
The median OS was 19.1 months, and the median progression-free survival (PFS) was 12.6 months. The 1-year OS rate was 62%. The 60-day mortality rate was 10%.
The authors noted that the survival data were superior to those seen in the phase 3 AZA-AML-001 study, which compared azacitidine therapy with conventional regimens in patients older than 65 years with newly diagnosed AML who were not eligible for stem cell transplants. In that trial, median OS was 10.4 months, the CR rate was 19.5% (vs. 49% in the present study), the 1-year OS rate was 46.5%, and the 60-day mortality rate was 16.2%.
They acknowledged, however, that the validity of the comparison is limited by their study’s small sample size, potential differences between the study populations, and lack of a control group in the present study. The investigators also found that clearance rates of baseline somatic mutations correlated with response to treatment.
Grade 3 or greater treatment-emergent adverse events occurred in 43 of the 50 patients, including infections, thrombocytopenias, and febrile neutropenias.
“On the basis of these encouraging results, a phase 3, multicenter, double-blind, randomized study of pracinostat vs. placebo with azacitidine (NCT03151408) is currently ongoing to confirm superiority of the combination in this difficult-to-treat AML population,” the investigators wrote.
The study was supported by research funding from MEI Pharma, which helped develop pracinostat. Dr. Garcia-Manero reported having no disclosures. Multiple coauthors reported financial relationships with MEI and others. One coauthor is an MEI employee.
SOURCE: Garcia-Manero G et al. Blood Adv. 2019 Feb 26;3(4):508-18.
FROM BLOOD ADVANCES