User login
CCSs have higher burden of chronic conditions
Adult survivors of childhood cancer have a greater cumulative burden of chronic health conditions than the general public, according to research published in The Lancet.
The study showed that, by age 50, childhood cancer survivors (CCSs) had experienced, on average, 17.1 chronic health conditions, and matched control subjects had experienced 9.2.
“The cumulative burden of chronic disease revealed in this analysis, along with the complexity and severity of chronic conditions some survivors experience, found childhood cancer survivors to be a vulnerable, medically complex population,” said study author Nickhill Bhakta, MD, of St. Jude Children’s Research Hospital in Memphis, Tennessee.
For this study, Dr Bhakta and his colleagues assessed the lifelong impact of 168 chronic health conditions—such as hepatic, thyroid, ocular, and reproductive disorders—on CCSs and control subjects.
The 3010 evaluable CCSs had survived 10 years or longer from their initial cancer diagnosis and were 18 years or older as of June 30, 2015. The 272 controls had no history of pediatric cancer and were matched to CCSs by age and sex.
At age 50, the cumulative incidence of chronic health conditions (of any grade) was 99.9% in CCSs and 96.0% in controls (P<0.0001). The cumulative incidence of grade 3 to 5 chronic health conditions was 96.0% and 84.9%, respectively (P<0.0001).
The cumulative burden for CCSs was 17.1 chronic health conditions, including 4.7 that were grade 3 to 5. For controls, the cumulative burden was 9.2 chronic health conditions, including 2.3 that were grade 3 to 5 (P<0.0001 for both comparisons).
The researchers said second neoplasms, spinal disorders, and pulmonary disease were major contributors to the excess total cumulative burden observed in CCSs. However, there was “notable heterogeneity” in burden according to the patients’ primary cancer diagnosis.
For instance, growth hormone deficiency was in the top 10th percentile of chronic health conditions for survivors of acute lymphoblastic leukemia but not for controls.
And pulmonary function deficits were in the top 10th percentile for survivors of acute myeloid leukemia and Hodgkin lymphoma but not for controls or survivors of acute lymphoblastic leukemia or non-Hodgkin lymphoma.
“This study found that the average childhood cancer survivor has a cumulative burden of chronic disease that requires a significant time investment by healthcare providers to disentangle and manage—time that community providers are unlikely to have,” Dr Bhakta said.
“The results suggest that childhood cancer survivors may benefit from the integrated, specialized healthcare delivery that is being tried for individuals infected with HIV or those with other complex, chronic health problems.” ![]()
Adult survivors of childhood cancer have a greater cumulative burden of chronic health conditions than the general public, according to research published in The Lancet.
The study showed that, by age 50, childhood cancer survivors (CCSs) had experienced, on average, 17.1 chronic health conditions, and matched control subjects had experienced 9.2.
“The cumulative burden of chronic disease revealed in this analysis, along with the complexity and severity of chronic conditions some survivors experience, found childhood cancer survivors to be a vulnerable, medically complex population,” said study author Nickhill Bhakta, MD, of St. Jude Children’s Research Hospital in Memphis, Tennessee.
For this study, Dr Bhakta and his colleagues assessed the lifelong impact of 168 chronic health conditions—such as hepatic, thyroid, ocular, and reproductive disorders—on CCSs and control subjects.
The 3010 evaluable CCSs had survived 10 years or longer from their initial cancer diagnosis and were 18 years or older as of June 30, 2015. The 272 controls had no history of pediatric cancer and were matched to CCSs by age and sex.
At age 50, the cumulative incidence of chronic health conditions (of any grade) was 99.9% in CCSs and 96.0% in controls (P<0.0001). The cumulative incidence of grade 3 to 5 chronic health conditions was 96.0% and 84.9%, respectively (P<0.0001).
The cumulative burden for CCSs was 17.1 chronic health conditions, including 4.7 that were grade 3 to 5. For controls, the cumulative burden was 9.2 chronic health conditions, including 2.3 that were grade 3 to 5 (P<0.0001 for both comparisons).
The researchers said second neoplasms, spinal disorders, and pulmonary disease were major contributors to the excess total cumulative burden observed in CCSs. However, there was “notable heterogeneity” in burden according to the patients’ primary cancer diagnosis.
For instance, growth hormone deficiency was in the top 10th percentile of chronic health conditions for survivors of acute lymphoblastic leukemia but not for controls.
And pulmonary function deficits were in the top 10th percentile for survivors of acute myeloid leukemia and Hodgkin lymphoma but not for controls or survivors of acute lymphoblastic leukemia or non-Hodgkin lymphoma.
“This study found that the average childhood cancer survivor has a cumulative burden of chronic disease that requires a significant time investment by healthcare providers to disentangle and manage—time that community providers are unlikely to have,” Dr Bhakta said.
“The results suggest that childhood cancer survivors may benefit from the integrated, specialized healthcare delivery that is being tried for individuals infected with HIV or those with other complex, chronic health problems.” ![]()
Adult survivors of childhood cancer have a greater cumulative burden of chronic health conditions than the general public, according to research published in The Lancet.
The study showed that, by age 50, childhood cancer survivors (CCSs) had experienced, on average, 17.1 chronic health conditions, and matched control subjects had experienced 9.2.
“The cumulative burden of chronic disease revealed in this analysis, along with the complexity and severity of chronic conditions some survivors experience, found childhood cancer survivors to be a vulnerable, medically complex population,” said study author Nickhill Bhakta, MD, of St. Jude Children’s Research Hospital in Memphis, Tennessee.
For this study, Dr Bhakta and his colleagues assessed the lifelong impact of 168 chronic health conditions—such as hepatic, thyroid, ocular, and reproductive disorders—on CCSs and control subjects.
The 3010 evaluable CCSs had survived 10 years or longer from their initial cancer diagnosis and were 18 years or older as of June 30, 2015. The 272 controls had no history of pediatric cancer and were matched to CCSs by age and sex.
At age 50, the cumulative incidence of chronic health conditions (of any grade) was 99.9% in CCSs and 96.0% in controls (P<0.0001). The cumulative incidence of grade 3 to 5 chronic health conditions was 96.0% and 84.9%, respectively (P<0.0001).
The cumulative burden for CCSs was 17.1 chronic health conditions, including 4.7 that were grade 3 to 5. For controls, the cumulative burden was 9.2 chronic health conditions, including 2.3 that were grade 3 to 5 (P<0.0001 for both comparisons).
The researchers said second neoplasms, spinal disorders, and pulmonary disease were major contributors to the excess total cumulative burden observed in CCSs. However, there was “notable heterogeneity” in burden according to the patients’ primary cancer diagnosis.
For instance, growth hormone deficiency was in the top 10th percentile of chronic health conditions for survivors of acute lymphoblastic leukemia but not for controls.
And pulmonary function deficits were in the top 10th percentile for survivors of acute myeloid leukemia and Hodgkin lymphoma but not for controls or survivors of acute lymphoblastic leukemia or non-Hodgkin lymphoma.
“This study found that the average childhood cancer survivor has a cumulative burden of chronic disease that requires a significant time investment by healthcare providers to disentangle and manage—time that community providers are unlikely to have,” Dr Bhakta said.
“The results suggest that childhood cancer survivors may benefit from the integrated, specialized healthcare delivery that is being tried for individuals infected with HIV or those with other complex, chronic health problems.” ![]()
New AML approvals changing the treatment landscape
With a recent flurry of new drug approvals, the treatment landscape for acute myeloid leukemia has expanded, raising new questions about how to incorporate those drugs into patient care.
Until about a decade ago, advances in AML therapy centered mainly around iterations of daunorubicin and cytarabine. Now, novel and targeted agents, many specifically going after mutational byproducts, are yielding some great results and raising hopes for better survival outcomes, Jeffrey Lancet, MD, said in an interview.
“When I go to sleep at night, I often dream about ... 10-year survival rates in the 80% range. And then I wake up ... and I realize this is actually [the survival curve for chronic myeloid leukemia]. This is where we’d like to be [with AML].” Those outcomes are a long way off, but appreciable incremental gains may lie ahead with the recent advances in AML therapy, said Dr. Lancet, chair of the department of malignant hematology at Moffitt Cancer Center in Tampa.
In addition to the new approvals, 16 drugs are in late stage clinical development and will likely contribute to an AML market that is expected to surpass $1.5 billion by 2026, according to projections by the market intelligence company GlobalData.
Vyxeos
The liposome-encapsulated combination of daunorubicin and cytarabine (Vyxeos) was approved in August by the Food and Drug Administration for the treatment of therapy-related AML and AML with myelodysplasia-related changes.
In a phase 3 randomized trial, the fixed-dose combination product was associated with median overall survival of 9.6 months, compared with 5.9 months with a standard combination of cytarabine and daunorubicin (7+3).
“I would envision that Vyxeos will hold and become the primary standard of care for fit chemotherapy-suitable older patients, or any patients for that matter, who are dealing with secondary-like AML or high-risk AML, based on the phase 3 results that we demonstrated,” Dr. Lancet, the principal investigator for the trial, said in an interview.
Asked whether the improved survival with Vyxeos is primarily related to more patients becoming transplant eligible or to significant reductions in disease burden, Dr. Lancet remarked that it’s likely a mixture of both.
The high remission rate with Vyxeos vs. standard 7+3 therapy means Vyxeos has the ability to stand on its own, and “the potential to send more patients to transplant and to get better results.”
“Transplant is part of the continuum of care of AML, including in older patients, and Vyxeos is going to become a standard part of that care,” he remarked. But transplant outcomes were not a predesignated component of the phase 3 trial, and further study will be needed to determine Vyxeos’ role as a bridge to transplant. “At this stage I can reasonably state that it has a role in the upfront therapy of secondary and high-risk AML, regardless of whether the patient is being considered for transplant.”
The early stages of working Vyxeos into the therapeutic mix come with some challenges, however, according to Donna Capozzi, PharmD.
Vyxeos is a fixed-dose combination that comes in vials containing 44 mg daunorubicin and 100 mg cytarabine encapsulated in liposomes. Patient dosing is based on the daunorubicin component and calculated based on body surface area (mg/m2), meaning the cytarabine dose does not need to be calculated. There are both pros and cons to this approach, she explained.
Benefits include a longer half-life with Vyxeos vs. standard 7+3, and the fact that during induction the drug is delivered on days 1, 3, and 5 for 90 minutes rather than continuously for 7 days as with 7+3, Dr. Capozzi said.
The main concern relates to ensuring that the dosing is calculated based on the proper component, she said.
“We had our first patient last week. It was very time consuming, with double and triple checking to make sure everything was correct,” she said. Preparing the drug is also time-consuming, as it involves multiple steps, such as warming, which is not required with standard 7+3; the additional labor factors will have to be built into workflow, she noted.
“The other piece not fully in place right now is building [the use of Vyxeos] into electronic health records,” she said, adding that safeguards put into place through EHRs will also help to streamline the administration process.
For example, cardiac toxicity is a known effect of daunorubicin; the EHR will help track lifetime cumulative dosing of that component, which is otherwise challenging, especially when using a combination product, she said.
The process will get easier over time, as use of Vyxeos becomes more prevalent in practice, she added. “None of these are insurmountable issues.”
Cost is another matter. Based on average wholesale prices, the cost per cycle is approximately $40,000 with Vyxeos vs. about $4,300 for conventional 7+3 therapy, Dr. Capozzi said. Given the differential, there will be a great deal of debate as to which patients will derive the most benefit from Vyxeos, she said.
Also, it will take time to figure out the extent of adverse events. “For liposomal products in general, rash-type side effects can be really significant. Hand-foot syndrome was not reported in the initial trials, but we’ll keep our eyes open to see how that plays out,” she said noting that the one patient treated so far at the University of Pennsylvania is doing very well. “We will learn more with real world experience.”
Oral targeted therapies
Enasidenib (Idhifa) was approved under priority review in August in conjunction with a companion diagnostic IDH2 assay for patients with relapsed or refractory disease and specific mutations in the IDH2 gene. Midostaurin (Rydapt) was approved in April for use in conjunction with standard daunorubicin and cytarabine induction and cytarabine consolidation in adults with FLT3 mutation-positive AML.
In a phase 1 dose escalation study reported at the annual meeting of the European Hematology Association, enasidenib was associated with an overall response rate of 37% in patients with relapsed/refractory AML, including 20.1% complete responses and 7.9% complete responses with incomplete recovery of platelets or incomplete hematologic recovery, 3.7% with partial responses, and 5.1% with a morphologic leukemia-free state. Patients who had a CR had a median overall survival of 22.9 months. For patients with responses other than CR, the median OS was 15.1 months. For patients with no response to the drug, the median OS was 5.6 months, Dr. Eytan M. Stein, of Memorial Sloan Kettering Cancer Center in New York, reported.
Additionally, need for transfusions was reduced in 34% of 157 patients who required transfusions at study entry.
“In a relapsed or refractory group of patients where there’s no true standard of care, this drug definitely represents a major breakthrough and has a lot of utility as a single agent, as a potential bridge to a transplant, and in combination with new or even old drugs – including regular old induction chemotherapy as a way to improve responses and outcomes in the future,” Dr. Lancet said, adding that as an oral agent it has potential for development as a maintenance strategy.
This agent could have a large impact, he said, adding: “I think this sets the paradigm for novel targeted therapies.”
Midostaurin has also emerged as a new standard of care, particularly for younger patients, Dr. Lancet said.
The approval of the multitargeted kinase inhibitor was based on the results of the randomized, placebo-controlled phase 3 RATIFY trial, which demonstrated significantly longer overall and event-free survival vs. placebo and standard chemotherapy in newly diagnosed AML patients with FLT3 gene mutations.
“I think this will be the new comparator for future studies, whatever they may be, for this patient population,” he said.
Dr. Capozzi noted that she has had some difficulty obtaining prior authorization for enasidenib due to its high cost (about $1,000/day).
The drug is taken orally on days 8-21 of a 28-day treatment cycle. In RATIFY, patients who achieved complete remission after induction therapy received four 28-day cycles of consolidation therapy.
Dr. Capozzi noted that the dosing regimen can be confusing, as it changes depending on whether it is used for induction or consolidation. It remains to be seen how these agents will fit into the treatment setting, she said.
Targeted therapies in development
Other targeted therapies in development for AML include an IDH1 inhibitor, the BCL2 inhibitor venetoclax, and several second-generation FLT3 inhibitors such as gilteritinib, Dr. Lancet said.
Venetoclax, which is currently approved for chronic lymphocytic leukemia, has shown single agent activity, but is even more promising in combination with low-dose cytarabine or aza-nucleosides, he noted.
For example, in one recent study reported at the annual congress of the European Hematology Association, response rates in older, newly diagnosed AML patients were as high as 72% for azacitidine plus venetoclax, and 76% for decitabine plus venetoclax.
“So there’s a lot of interest and promise,” Dr. Lancet said, adding that venetoclax may have broad application in AML. “We’ll be seeing a lot more data in the next year or two.”
An unusual aspect of venetoclax, which is used often for CLL, is the need for observation during dose escalation, Dr. Capozzi noted. Patients tend to question the need for admission for observation with the use of an oral agent, thus efforts are underway to develop criteria for outpatient observation.
Otherwise, venetoclax is fairly easy to access and use, and is well tolerated, she said.
“I expect as we learn more about where (venetoclax) fits in, it will be a much more commonplace agent” as part of AML therapy, she said.
Gilteritinib, as well as the second generation FLT3 inhibitors quizartinib and crenolanib, are also of interest in AML. With midostaurin already on the market, however, different strategies are being pursued, Dr. Lancet said.
“I believe gilteritinib is entering the fray in relapsed/refractory disease, and crenolanib is being looked at in the upfront FLT3 AML-positive setting and ultimately will be compared to midostaurin in combination with chemotherapy in that setting,” he added, noting that these drugs have the advantage of being more potent and selective inhibitors of FLT3, and some appear to have the ability to target resistance-conferring mutations.
“It still remains to be determined what the ultimate role will be, especially now that midostaurin is approved as frontline therapy and, in my opinion, will likely be entrenched there for awhile,” he said. “It’s a fairly competitive field right now, but certainly one where there’s a lot of excitement. The encouraging part is the second generation inhibitors, especially crenolanib and gilteritinib, are able to rescue some patients who may have failed primary therapy with an FLT3 inhibitor.”
Future direction and outcomes
So how should one go about selecting therapies, in the absence of data on combining therapies, for patients with multiple mutations?
Ideally, that means teasing out which of the AML patient’s mutations is clonal and the driver of their disease, and which one is subclonal. There are no guarantees, but that seems like a rational way to begin and move the field forward to studies of combination therapies, Dr. Lancet said.
“I think with the right combinations that target leukemias that are mutationally driven, there is potential to treat subsets of patient with very targeted therapies that will lead to prolonged survival. Right now, for the most part, we don’t have drugs for many of the targets that are very important in AML, and we don’t always know which target is driving the disease ... these are considerations that remain to be discovered,” he said. “But I do think that in 10 years we will have the ability with novel drugs and increased understanding of the clinical relevance of these targets to really personalize the approach more so than we are today, and to increase response rates significantly and improve survival as a result.”
Dr. Lancet is a consultant for Jazz Pharmaceuticals, Daiichi Sankyo, and Celgene. Dr. Capozzi reported having no disclosures.
With a recent flurry of new drug approvals, the treatment landscape for acute myeloid leukemia has expanded, raising new questions about how to incorporate those drugs into patient care.
Until about a decade ago, advances in AML therapy centered mainly around iterations of daunorubicin and cytarabine. Now, novel and targeted agents, many specifically going after mutational byproducts, are yielding some great results and raising hopes for better survival outcomes, Jeffrey Lancet, MD, said in an interview.
“When I go to sleep at night, I often dream about ... 10-year survival rates in the 80% range. And then I wake up ... and I realize this is actually [the survival curve for chronic myeloid leukemia]. This is where we’d like to be [with AML].” Those outcomes are a long way off, but appreciable incremental gains may lie ahead with the recent advances in AML therapy, said Dr. Lancet, chair of the department of malignant hematology at Moffitt Cancer Center in Tampa.
In addition to the new approvals, 16 drugs are in late stage clinical development and will likely contribute to an AML market that is expected to surpass $1.5 billion by 2026, according to projections by the market intelligence company GlobalData.
Vyxeos
The liposome-encapsulated combination of daunorubicin and cytarabine (Vyxeos) was approved in August by the Food and Drug Administration for the treatment of therapy-related AML and AML with myelodysplasia-related changes.
In a phase 3 randomized trial, the fixed-dose combination product was associated with median overall survival of 9.6 months, compared with 5.9 months with a standard combination of cytarabine and daunorubicin (7+3).
“I would envision that Vyxeos will hold and become the primary standard of care for fit chemotherapy-suitable older patients, or any patients for that matter, who are dealing with secondary-like AML or high-risk AML, based on the phase 3 results that we demonstrated,” Dr. Lancet, the principal investigator for the trial, said in an interview.
Asked whether the improved survival with Vyxeos is primarily related to more patients becoming transplant eligible or to significant reductions in disease burden, Dr. Lancet remarked that it’s likely a mixture of both.
The high remission rate with Vyxeos vs. standard 7+3 therapy means Vyxeos has the ability to stand on its own, and “the potential to send more patients to transplant and to get better results.”
“Transplant is part of the continuum of care of AML, including in older patients, and Vyxeos is going to become a standard part of that care,” he remarked. But transplant outcomes were not a predesignated component of the phase 3 trial, and further study will be needed to determine Vyxeos’ role as a bridge to transplant. “At this stage I can reasonably state that it has a role in the upfront therapy of secondary and high-risk AML, regardless of whether the patient is being considered for transplant.”
The early stages of working Vyxeos into the therapeutic mix come with some challenges, however, according to Donna Capozzi, PharmD.
Vyxeos is a fixed-dose combination that comes in vials containing 44 mg daunorubicin and 100 mg cytarabine encapsulated in liposomes. Patient dosing is based on the daunorubicin component and calculated based on body surface area (mg/m2), meaning the cytarabine dose does not need to be calculated. There are both pros and cons to this approach, she explained.
Benefits include a longer half-life with Vyxeos vs. standard 7+3, and the fact that during induction the drug is delivered on days 1, 3, and 5 for 90 minutes rather than continuously for 7 days as with 7+3, Dr. Capozzi said.
The main concern relates to ensuring that the dosing is calculated based on the proper component, she said.
“We had our first patient last week. It was very time consuming, with double and triple checking to make sure everything was correct,” she said. Preparing the drug is also time-consuming, as it involves multiple steps, such as warming, which is not required with standard 7+3; the additional labor factors will have to be built into workflow, she noted.
“The other piece not fully in place right now is building [the use of Vyxeos] into electronic health records,” she said, adding that safeguards put into place through EHRs will also help to streamline the administration process.
For example, cardiac toxicity is a known effect of daunorubicin; the EHR will help track lifetime cumulative dosing of that component, which is otherwise challenging, especially when using a combination product, she said.
The process will get easier over time, as use of Vyxeos becomes more prevalent in practice, she added. “None of these are insurmountable issues.”
Cost is another matter. Based on average wholesale prices, the cost per cycle is approximately $40,000 with Vyxeos vs. about $4,300 for conventional 7+3 therapy, Dr. Capozzi said. Given the differential, there will be a great deal of debate as to which patients will derive the most benefit from Vyxeos, she said.
Also, it will take time to figure out the extent of adverse events. “For liposomal products in general, rash-type side effects can be really significant. Hand-foot syndrome was not reported in the initial trials, but we’ll keep our eyes open to see how that plays out,” she said noting that the one patient treated so far at the University of Pennsylvania is doing very well. “We will learn more with real world experience.”
Oral targeted therapies
Enasidenib (Idhifa) was approved under priority review in August in conjunction with a companion diagnostic IDH2 assay for patients with relapsed or refractory disease and specific mutations in the IDH2 gene. Midostaurin (Rydapt) was approved in April for use in conjunction with standard daunorubicin and cytarabine induction and cytarabine consolidation in adults with FLT3 mutation-positive AML.
In a phase 1 dose escalation study reported at the annual meeting of the European Hematology Association, enasidenib was associated with an overall response rate of 37% in patients with relapsed/refractory AML, including 20.1% complete responses and 7.9% complete responses with incomplete recovery of platelets or incomplete hematologic recovery, 3.7% with partial responses, and 5.1% with a morphologic leukemia-free state. Patients who had a CR had a median overall survival of 22.9 months. For patients with responses other than CR, the median OS was 15.1 months. For patients with no response to the drug, the median OS was 5.6 months, Dr. Eytan M. Stein, of Memorial Sloan Kettering Cancer Center in New York, reported.
Additionally, need for transfusions was reduced in 34% of 157 patients who required transfusions at study entry.
“In a relapsed or refractory group of patients where there’s no true standard of care, this drug definitely represents a major breakthrough and has a lot of utility as a single agent, as a potential bridge to a transplant, and in combination with new or even old drugs – including regular old induction chemotherapy as a way to improve responses and outcomes in the future,” Dr. Lancet said, adding that as an oral agent it has potential for development as a maintenance strategy.
This agent could have a large impact, he said, adding: “I think this sets the paradigm for novel targeted therapies.”
Midostaurin has also emerged as a new standard of care, particularly for younger patients, Dr. Lancet said.
The approval of the multitargeted kinase inhibitor was based on the results of the randomized, placebo-controlled phase 3 RATIFY trial, which demonstrated significantly longer overall and event-free survival vs. placebo and standard chemotherapy in newly diagnosed AML patients with FLT3 gene mutations.
“I think this will be the new comparator for future studies, whatever they may be, for this patient population,” he said.
Dr. Capozzi noted that she has had some difficulty obtaining prior authorization for enasidenib due to its high cost (about $1,000/day).
The drug is taken orally on days 8-21 of a 28-day treatment cycle. In RATIFY, patients who achieved complete remission after induction therapy received four 28-day cycles of consolidation therapy.
Dr. Capozzi noted that the dosing regimen can be confusing, as it changes depending on whether it is used for induction or consolidation. It remains to be seen how these agents will fit into the treatment setting, she said.
Targeted therapies in development
Other targeted therapies in development for AML include an IDH1 inhibitor, the BCL2 inhibitor venetoclax, and several second-generation FLT3 inhibitors such as gilteritinib, Dr. Lancet said.
Venetoclax, which is currently approved for chronic lymphocytic leukemia, has shown single agent activity, but is even more promising in combination with low-dose cytarabine or aza-nucleosides, he noted.
For example, in one recent study reported at the annual congress of the European Hematology Association, response rates in older, newly diagnosed AML patients were as high as 72% for azacitidine plus venetoclax, and 76% for decitabine plus venetoclax.
“So there’s a lot of interest and promise,” Dr. Lancet said, adding that venetoclax may have broad application in AML. “We’ll be seeing a lot more data in the next year or two.”
An unusual aspect of venetoclax, which is used often for CLL, is the need for observation during dose escalation, Dr. Capozzi noted. Patients tend to question the need for admission for observation with the use of an oral agent, thus efforts are underway to develop criteria for outpatient observation.
Otherwise, venetoclax is fairly easy to access and use, and is well tolerated, she said.
“I expect as we learn more about where (venetoclax) fits in, it will be a much more commonplace agent” as part of AML therapy, she said.
Gilteritinib, as well as the second generation FLT3 inhibitors quizartinib and crenolanib, are also of interest in AML. With midostaurin already on the market, however, different strategies are being pursued, Dr. Lancet said.
“I believe gilteritinib is entering the fray in relapsed/refractory disease, and crenolanib is being looked at in the upfront FLT3 AML-positive setting and ultimately will be compared to midostaurin in combination with chemotherapy in that setting,” he added, noting that these drugs have the advantage of being more potent and selective inhibitors of FLT3, and some appear to have the ability to target resistance-conferring mutations.
“It still remains to be determined what the ultimate role will be, especially now that midostaurin is approved as frontline therapy and, in my opinion, will likely be entrenched there for awhile,” he said. “It’s a fairly competitive field right now, but certainly one where there’s a lot of excitement. The encouraging part is the second generation inhibitors, especially crenolanib and gilteritinib, are able to rescue some patients who may have failed primary therapy with an FLT3 inhibitor.”
Future direction and outcomes
So how should one go about selecting therapies, in the absence of data on combining therapies, for patients with multiple mutations?
Ideally, that means teasing out which of the AML patient’s mutations is clonal and the driver of their disease, and which one is subclonal. There are no guarantees, but that seems like a rational way to begin and move the field forward to studies of combination therapies, Dr. Lancet said.
“I think with the right combinations that target leukemias that are mutationally driven, there is potential to treat subsets of patient with very targeted therapies that will lead to prolonged survival. Right now, for the most part, we don’t have drugs for many of the targets that are very important in AML, and we don’t always know which target is driving the disease ... these are considerations that remain to be discovered,” he said. “But I do think that in 10 years we will have the ability with novel drugs and increased understanding of the clinical relevance of these targets to really personalize the approach more so than we are today, and to increase response rates significantly and improve survival as a result.”
Dr. Lancet is a consultant for Jazz Pharmaceuticals, Daiichi Sankyo, and Celgene. Dr. Capozzi reported having no disclosures.
With a recent flurry of new drug approvals, the treatment landscape for acute myeloid leukemia has expanded, raising new questions about how to incorporate those drugs into patient care.
Until about a decade ago, advances in AML therapy centered mainly around iterations of daunorubicin and cytarabine. Now, novel and targeted agents, many specifically going after mutational byproducts, are yielding some great results and raising hopes for better survival outcomes, Jeffrey Lancet, MD, said in an interview.
“When I go to sleep at night, I often dream about ... 10-year survival rates in the 80% range. And then I wake up ... and I realize this is actually [the survival curve for chronic myeloid leukemia]. This is where we’d like to be [with AML].” Those outcomes are a long way off, but appreciable incremental gains may lie ahead with the recent advances in AML therapy, said Dr. Lancet, chair of the department of malignant hematology at Moffitt Cancer Center in Tampa.
In addition to the new approvals, 16 drugs are in late stage clinical development and will likely contribute to an AML market that is expected to surpass $1.5 billion by 2026, according to projections by the market intelligence company GlobalData.
Vyxeos
The liposome-encapsulated combination of daunorubicin and cytarabine (Vyxeos) was approved in August by the Food and Drug Administration for the treatment of therapy-related AML and AML with myelodysplasia-related changes.
In a phase 3 randomized trial, the fixed-dose combination product was associated with median overall survival of 9.6 months, compared with 5.9 months with a standard combination of cytarabine and daunorubicin (7+3).
“I would envision that Vyxeos will hold and become the primary standard of care for fit chemotherapy-suitable older patients, or any patients for that matter, who are dealing with secondary-like AML or high-risk AML, based on the phase 3 results that we demonstrated,” Dr. Lancet, the principal investigator for the trial, said in an interview.
Asked whether the improved survival with Vyxeos is primarily related to more patients becoming transplant eligible or to significant reductions in disease burden, Dr. Lancet remarked that it’s likely a mixture of both.
The high remission rate with Vyxeos vs. standard 7+3 therapy means Vyxeos has the ability to stand on its own, and “the potential to send more patients to transplant and to get better results.”
“Transplant is part of the continuum of care of AML, including in older patients, and Vyxeos is going to become a standard part of that care,” he remarked. But transplant outcomes were not a predesignated component of the phase 3 trial, and further study will be needed to determine Vyxeos’ role as a bridge to transplant. “At this stage I can reasonably state that it has a role in the upfront therapy of secondary and high-risk AML, regardless of whether the patient is being considered for transplant.”
The early stages of working Vyxeos into the therapeutic mix come with some challenges, however, according to Donna Capozzi, PharmD.
Vyxeos is a fixed-dose combination that comes in vials containing 44 mg daunorubicin and 100 mg cytarabine encapsulated in liposomes. Patient dosing is based on the daunorubicin component and calculated based on body surface area (mg/m2), meaning the cytarabine dose does not need to be calculated. There are both pros and cons to this approach, she explained.
Benefits include a longer half-life with Vyxeos vs. standard 7+3, and the fact that during induction the drug is delivered on days 1, 3, and 5 for 90 minutes rather than continuously for 7 days as with 7+3, Dr. Capozzi said.
The main concern relates to ensuring that the dosing is calculated based on the proper component, she said.
“We had our first patient last week. It was very time consuming, with double and triple checking to make sure everything was correct,” she said. Preparing the drug is also time-consuming, as it involves multiple steps, such as warming, which is not required with standard 7+3; the additional labor factors will have to be built into workflow, she noted.
“The other piece not fully in place right now is building [the use of Vyxeos] into electronic health records,” she said, adding that safeguards put into place through EHRs will also help to streamline the administration process.
For example, cardiac toxicity is a known effect of daunorubicin; the EHR will help track lifetime cumulative dosing of that component, which is otherwise challenging, especially when using a combination product, she said.
The process will get easier over time, as use of Vyxeos becomes more prevalent in practice, she added. “None of these are insurmountable issues.”
Cost is another matter. Based on average wholesale prices, the cost per cycle is approximately $40,000 with Vyxeos vs. about $4,300 for conventional 7+3 therapy, Dr. Capozzi said. Given the differential, there will be a great deal of debate as to which patients will derive the most benefit from Vyxeos, she said.
Also, it will take time to figure out the extent of adverse events. “For liposomal products in general, rash-type side effects can be really significant. Hand-foot syndrome was not reported in the initial trials, but we’ll keep our eyes open to see how that plays out,” she said noting that the one patient treated so far at the University of Pennsylvania is doing very well. “We will learn more with real world experience.”
Oral targeted therapies
Enasidenib (Idhifa) was approved under priority review in August in conjunction with a companion diagnostic IDH2 assay for patients with relapsed or refractory disease and specific mutations in the IDH2 gene. Midostaurin (Rydapt) was approved in April for use in conjunction with standard daunorubicin and cytarabine induction and cytarabine consolidation in adults with FLT3 mutation-positive AML.
In a phase 1 dose escalation study reported at the annual meeting of the European Hematology Association, enasidenib was associated with an overall response rate of 37% in patients with relapsed/refractory AML, including 20.1% complete responses and 7.9% complete responses with incomplete recovery of platelets or incomplete hematologic recovery, 3.7% with partial responses, and 5.1% with a morphologic leukemia-free state. Patients who had a CR had a median overall survival of 22.9 months. For patients with responses other than CR, the median OS was 15.1 months. For patients with no response to the drug, the median OS was 5.6 months, Dr. Eytan M. Stein, of Memorial Sloan Kettering Cancer Center in New York, reported.
Additionally, need for transfusions was reduced in 34% of 157 patients who required transfusions at study entry.
“In a relapsed or refractory group of patients where there’s no true standard of care, this drug definitely represents a major breakthrough and has a lot of utility as a single agent, as a potential bridge to a transplant, and in combination with new or even old drugs – including regular old induction chemotherapy as a way to improve responses and outcomes in the future,” Dr. Lancet said, adding that as an oral agent it has potential for development as a maintenance strategy.
This agent could have a large impact, he said, adding: “I think this sets the paradigm for novel targeted therapies.”
Midostaurin has also emerged as a new standard of care, particularly for younger patients, Dr. Lancet said.
The approval of the multitargeted kinase inhibitor was based on the results of the randomized, placebo-controlled phase 3 RATIFY trial, which demonstrated significantly longer overall and event-free survival vs. placebo and standard chemotherapy in newly diagnosed AML patients with FLT3 gene mutations.
“I think this will be the new comparator for future studies, whatever they may be, for this patient population,” he said.
Dr. Capozzi noted that she has had some difficulty obtaining prior authorization for enasidenib due to its high cost (about $1,000/day).
The drug is taken orally on days 8-21 of a 28-day treatment cycle. In RATIFY, patients who achieved complete remission after induction therapy received four 28-day cycles of consolidation therapy.
Dr. Capozzi noted that the dosing regimen can be confusing, as it changes depending on whether it is used for induction or consolidation. It remains to be seen how these agents will fit into the treatment setting, she said.
Targeted therapies in development
Other targeted therapies in development for AML include an IDH1 inhibitor, the BCL2 inhibitor venetoclax, and several second-generation FLT3 inhibitors such as gilteritinib, Dr. Lancet said.
Venetoclax, which is currently approved for chronic lymphocytic leukemia, has shown single agent activity, but is even more promising in combination with low-dose cytarabine or aza-nucleosides, he noted.
For example, in one recent study reported at the annual congress of the European Hematology Association, response rates in older, newly diagnosed AML patients were as high as 72% for azacitidine plus venetoclax, and 76% for decitabine plus venetoclax.
“So there’s a lot of interest and promise,” Dr. Lancet said, adding that venetoclax may have broad application in AML. “We’ll be seeing a lot more data in the next year or two.”
An unusual aspect of venetoclax, which is used often for CLL, is the need for observation during dose escalation, Dr. Capozzi noted. Patients tend to question the need for admission for observation with the use of an oral agent, thus efforts are underway to develop criteria for outpatient observation.
Otherwise, venetoclax is fairly easy to access and use, and is well tolerated, she said.
“I expect as we learn more about where (venetoclax) fits in, it will be a much more commonplace agent” as part of AML therapy, she said.
Gilteritinib, as well as the second generation FLT3 inhibitors quizartinib and crenolanib, are also of interest in AML. With midostaurin already on the market, however, different strategies are being pursued, Dr. Lancet said.
“I believe gilteritinib is entering the fray in relapsed/refractory disease, and crenolanib is being looked at in the upfront FLT3 AML-positive setting and ultimately will be compared to midostaurin in combination with chemotherapy in that setting,” he added, noting that these drugs have the advantage of being more potent and selective inhibitors of FLT3, and some appear to have the ability to target resistance-conferring mutations.
“It still remains to be determined what the ultimate role will be, especially now that midostaurin is approved as frontline therapy and, in my opinion, will likely be entrenched there for awhile,” he said. “It’s a fairly competitive field right now, but certainly one where there’s a lot of excitement. The encouraging part is the second generation inhibitors, especially crenolanib and gilteritinib, are able to rescue some patients who may have failed primary therapy with an FLT3 inhibitor.”
Future direction and outcomes
So how should one go about selecting therapies, in the absence of data on combining therapies, for patients with multiple mutations?
Ideally, that means teasing out which of the AML patient’s mutations is clonal and the driver of their disease, and which one is subclonal. There are no guarantees, but that seems like a rational way to begin and move the field forward to studies of combination therapies, Dr. Lancet said.
“I think with the right combinations that target leukemias that are mutationally driven, there is potential to treat subsets of patient with very targeted therapies that will lead to prolonged survival. Right now, for the most part, we don’t have drugs for many of the targets that are very important in AML, and we don’t always know which target is driving the disease ... these are considerations that remain to be discovered,” he said. “But I do think that in 10 years we will have the ability with novel drugs and increased understanding of the clinical relevance of these targets to really personalize the approach more so than we are today, and to increase response rates significantly and improve survival as a result.”
Dr. Lancet is a consultant for Jazz Pharmaceuticals, Daiichi Sankyo, and Celgene. Dr. Capozzi reported having no disclosures.
Studies of donor CAR T cells placed on hold
The US Food and Drug Administration (FDA) has placed a clinical hold on both phase 1 studies of UCART123, a universal (allogeneic) chimeric antigen receptor (CAR) T-cell therapy targeting CD123.
One study was designed for patients with acute myeloid leukemia (AML), and the other was designed for patients with blastic plasmacytoid dendritic cell neoplasm (BPDCN).
The clinical hold is due to the death of the first patient treated in the BPDCN trial.
The hold means no new subjects can be enrolled in either trial, and there can be no further dosing of subjects who are already enrolled.
BPDCN patient
The first patient treated in the BPDCN trial was a 78-year-old male who had received 1 prior therapy. He presented with relapsed/refractory BPDCN with 30% blasts in his bone marrow and cutaneous lesions (biopsy-proven BPDCN) at baseline.
The patient first received pre-conditioning with fludarabine (30 mg/m²/day for 4 days) and cyclophosphamide (1 g/m²/day for 3 days).
On August 16, 2017 (Day 0), the patient received UCART123 at 6.25 x 105 cells/kg, the first dose level explored in the protocol, without complication.
By Day 5, the patient had developed grade 2 cytokine release syndrome (CRS) and a grade 3 lung infection, which improved after a first dose of tocilizumab and the administration of broad-spectrum, intravenous antibiotics.
On Day 8, the patient was found to have more severe CRS (ultimately grade 5) and grade 4 capillary leak syndrome. Despite receiving treatment in keeping with CRS management, including the administration of corticosteroids and tociluzumab as well as intensive care unit support, the patient died on Day 9.
AML patient
The first patient treated in the AML study experienced similar adverse effects as the BPDCN patient but is still alive.
The AML patient was a 58-year-old woman with 84% blasts in her bone marrow at baseline.
On June 27, 2017 (Day 0), the patient received the same pre-conditioning regimen and the same dose of UCART123 as the BPDCN patient, without complication.
By Day 8, the AML patient had developed grade 2 CRS. This worsened to grade 3 on Day 9 and resolved on Day 11 with treatment in the intensive care unit.
The patient also experienced grade 4 capillary leak syndrome on Day 9 that resolved on Day 12.
Next steps
The data safety monitoring board for these trials met on August 28 and recommended lowering the dose of UCART123 to 6.25 x 104 cells/kg in both studies and capping cyclophosphamide to a total dose of 4 g over 3 days.
Cellectis, the company developing UCART123, said it is working with study investigators and the FDA to resume both trials with an amended protocol that includes these dose adjustments. ![]()
The US Food and Drug Administration (FDA) has placed a clinical hold on both phase 1 studies of UCART123, a universal (allogeneic) chimeric antigen receptor (CAR) T-cell therapy targeting CD123.
One study was designed for patients with acute myeloid leukemia (AML), and the other was designed for patients with blastic plasmacytoid dendritic cell neoplasm (BPDCN).
The clinical hold is due to the death of the first patient treated in the BPDCN trial.
The hold means no new subjects can be enrolled in either trial, and there can be no further dosing of subjects who are already enrolled.
BPDCN patient
The first patient treated in the BPDCN trial was a 78-year-old male who had received 1 prior therapy. He presented with relapsed/refractory BPDCN with 30% blasts in his bone marrow and cutaneous lesions (biopsy-proven BPDCN) at baseline.
The patient first received pre-conditioning with fludarabine (30 mg/m²/day for 4 days) and cyclophosphamide (1 g/m²/day for 3 days).
On August 16, 2017 (Day 0), the patient received UCART123 at 6.25 x 105 cells/kg, the first dose level explored in the protocol, without complication.
By Day 5, the patient had developed grade 2 cytokine release syndrome (CRS) and a grade 3 lung infection, which improved after a first dose of tocilizumab and the administration of broad-spectrum, intravenous antibiotics.
On Day 8, the patient was found to have more severe CRS (ultimately grade 5) and grade 4 capillary leak syndrome. Despite receiving treatment in keeping with CRS management, including the administration of corticosteroids and tociluzumab as well as intensive care unit support, the patient died on Day 9.
AML patient
The first patient treated in the AML study experienced similar adverse effects as the BPDCN patient but is still alive.
The AML patient was a 58-year-old woman with 84% blasts in her bone marrow at baseline.
On June 27, 2017 (Day 0), the patient received the same pre-conditioning regimen and the same dose of UCART123 as the BPDCN patient, without complication.
By Day 8, the AML patient had developed grade 2 CRS. This worsened to grade 3 on Day 9 and resolved on Day 11 with treatment in the intensive care unit.
The patient also experienced grade 4 capillary leak syndrome on Day 9 that resolved on Day 12.
Next steps
The data safety monitoring board for these trials met on August 28 and recommended lowering the dose of UCART123 to 6.25 x 104 cells/kg in both studies and capping cyclophosphamide to a total dose of 4 g over 3 days.
Cellectis, the company developing UCART123, said it is working with study investigators and the FDA to resume both trials with an amended protocol that includes these dose adjustments. ![]()
The US Food and Drug Administration (FDA) has placed a clinical hold on both phase 1 studies of UCART123, a universal (allogeneic) chimeric antigen receptor (CAR) T-cell therapy targeting CD123.
One study was designed for patients with acute myeloid leukemia (AML), and the other was designed for patients with blastic plasmacytoid dendritic cell neoplasm (BPDCN).
The clinical hold is due to the death of the first patient treated in the BPDCN trial.
The hold means no new subjects can be enrolled in either trial, and there can be no further dosing of subjects who are already enrolled.
BPDCN patient
The first patient treated in the BPDCN trial was a 78-year-old male who had received 1 prior therapy. He presented with relapsed/refractory BPDCN with 30% blasts in his bone marrow and cutaneous lesions (biopsy-proven BPDCN) at baseline.
The patient first received pre-conditioning with fludarabine (30 mg/m²/day for 4 days) and cyclophosphamide (1 g/m²/day for 3 days).
On August 16, 2017 (Day 0), the patient received UCART123 at 6.25 x 105 cells/kg, the first dose level explored in the protocol, without complication.
By Day 5, the patient had developed grade 2 cytokine release syndrome (CRS) and a grade 3 lung infection, which improved after a first dose of tocilizumab and the administration of broad-spectrum, intravenous antibiotics.
On Day 8, the patient was found to have more severe CRS (ultimately grade 5) and grade 4 capillary leak syndrome. Despite receiving treatment in keeping with CRS management, including the administration of corticosteroids and tociluzumab as well as intensive care unit support, the patient died on Day 9.
AML patient
The first patient treated in the AML study experienced similar adverse effects as the BPDCN patient but is still alive.
The AML patient was a 58-year-old woman with 84% blasts in her bone marrow at baseline.
On June 27, 2017 (Day 0), the patient received the same pre-conditioning regimen and the same dose of UCART123 as the BPDCN patient, without complication.
By Day 8, the AML patient had developed grade 2 CRS. This worsened to grade 3 on Day 9 and resolved on Day 11 with treatment in the intensive care unit.
The patient also experienced grade 4 capillary leak syndrome on Day 9 that resolved on Day 12.
Next steps
The data safety monitoring board for these trials met on August 28 and recommended lowering the dose of UCART123 to 6.25 x 104 cells/kg in both studies and capping cyclophosphamide to a total dose of 4 g over 3 days.
Cellectis, the company developing UCART123, said it is working with study investigators and the FDA to resume both trials with an amended protocol that includes these dose adjustments. ![]()
FDA grants orphan designation to product for CMV
The US Food and Drug Administration (FDA) has granted orphan drug designation to ATA230 for the treatment of cytomegalovirus (CMV) viremia and disease in immunocompromised patients.
ATA230 is an allogeneic, cytotoxic T-lymphocyte (CTL) product targeting antigens expressed by CMV.
The product is under investigation in phase 2 trials of patients with CMV viremia and disease who are refractory or resistant to antiviral treatment.
Atara Biotherapeutics, Inc., the company developing ATA230, said it will evaluate development plans for this therapy with the FDA and other global health authorities after beginning phase 3 studies of another product, ATA129.
The company said it decided to prioritize ATA129, which is being developed to treat patients with Epstein-Barr-virus-associated post-transplant lymphoproliferative disorder.
Phase 2 trial of ATA230
Researchers reported phase 2 results with ATA230 at the 2016 ASH Annual Meeting.
The data encompassed 15 patients with documented CMV mutations conferring resistance to antiviral therapies. The patients had received a median of 3 prior therapies.
Eleven of the 15 patients (73.3%) responded to ATA230, 6 with complete responses and 5 with partial responses.
At 6 months, the overall survival was 72.7% in responders and 25% in non-responders.
Within the 6 months of follow-up, 1 of the 11 responders died of CMV, and 3 of the 4 non-responders died of CMV.
Adverse events occurred in 6 patients. One grade 3 event and 1 grade 4 event were considered possibly related to ATA230.
About orphan designation
The FDA grants orphan designation to products intended to treat, diagnose, or prevent diseases/disorders that affect fewer than 200,000 people in the US.
The designation provides incentives for sponsors to develop products for rare diseases. This may include tax credits toward the cost of clinical trials, prescription drug user fee waivers, and 7 years of market exclusivity if the product is approved. ![]()
The US Food and Drug Administration (FDA) has granted orphan drug designation to ATA230 for the treatment of cytomegalovirus (CMV) viremia and disease in immunocompromised patients.
ATA230 is an allogeneic, cytotoxic T-lymphocyte (CTL) product targeting antigens expressed by CMV.
The product is under investigation in phase 2 trials of patients with CMV viremia and disease who are refractory or resistant to antiviral treatment.
Atara Biotherapeutics, Inc., the company developing ATA230, said it will evaluate development plans for this therapy with the FDA and other global health authorities after beginning phase 3 studies of another product, ATA129.
The company said it decided to prioritize ATA129, which is being developed to treat patients with Epstein-Barr-virus-associated post-transplant lymphoproliferative disorder.
Phase 2 trial of ATA230
Researchers reported phase 2 results with ATA230 at the 2016 ASH Annual Meeting.
The data encompassed 15 patients with documented CMV mutations conferring resistance to antiviral therapies. The patients had received a median of 3 prior therapies.
Eleven of the 15 patients (73.3%) responded to ATA230, 6 with complete responses and 5 with partial responses.
At 6 months, the overall survival was 72.7% in responders and 25% in non-responders.
Within the 6 months of follow-up, 1 of the 11 responders died of CMV, and 3 of the 4 non-responders died of CMV.
Adverse events occurred in 6 patients. One grade 3 event and 1 grade 4 event were considered possibly related to ATA230.
About orphan designation
The FDA grants orphan designation to products intended to treat, diagnose, or prevent diseases/disorders that affect fewer than 200,000 people in the US.
The designation provides incentives for sponsors to develop products for rare diseases. This may include tax credits toward the cost of clinical trials, prescription drug user fee waivers, and 7 years of market exclusivity if the product is approved. ![]()
The US Food and Drug Administration (FDA) has granted orphan drug designation to ATA230 for the treatment of cytomegalovirus (CMV) viremia and disease in immunocompromised patients.
ATA230 is an allogeneic, cytotoxic T-lymphocyte (CTL) product targeting antigens expressed by CMV.
The product is under investigation in phase 2 trials of patients with CMV viremia and disease who are refractory or resistant to antiviral treatment.
Atara Biotherapeutics, Inc., the company developing ATA230, said it will evaluate development plans for this therapy with the FDA and other global health authorities after beginning phase 3 studies of another product, ATA129.
The company said it decided to prioritize ATA129, which is being developed to treat patients with Epstein-Barr-virus-associated post-transplant lymphoproliferative disorder.
Phase 2 trial of ATA230
Researchers reported phase 2 results with ATA230 at the 2016 ASH Annual Meeting.
The data encompassed 15 patients with documented CMV mutations conferring resistance to antiviral therapies. The patients had received a median of 3 prior therapies.
Eleven of the 15 patients (73.3%) responded to ATA230, 6 with complete responses and 5 with partial responses.
At 6 months, the overall survival was 72.7% in responders and 25% in non-responders.
Within the 6 months of follow-up, 1 of the 11 responders died of CMV, and 3 of the 4 non-responders died of CMV.
Adverse events occurred in 6 patients. One grade 3 event and 1 grade 4 event were considered possibly related to ATA230.
About orphan designation
The FDA grants orphan designation to products intended to treat, diagnose, or prevent diseases/disorders that affect fewer than 200,000 people in the US.
The designation provides incentives for sponsors to develop products for rare diseases. This may include tax credits toward the cost of clinical trials, prescription drug user fee waivers, and 7 years of market exclusivity if the product is approved. ![]()
FDA grants orphan designation to product for GVHD
The US Food and Drug Administration (FDA) has granted orphan drug designation to ApoGraft™ as prophylaxis for acute and chronic graft-versus-host disease (GVHD) in transplant recipients.
ApoGraft is a mobilized peripheral blood cell product collected via apheresis from a matched, related donor. The product is exposed to the apoptotic mediator Fas ligand prior to transplantation.
ApoGraft was designed to eliminate immune responses after transplantation of foreign cells and tissues.
ApoGraft is being developed by Cellect Biotechnology Ltd.
The company is testing ApoGraft as acute GVHD prophylaxis in a phase 1/2 trial.
The trial is currently enrolling patients with hemato-oncology disorders who are eligible for allogeneic, HLA-matched hematopoietic stem cell transplant (HSCT).
The study is expected to have 4 cohorts, each consisting of 3 patients.
The difference between the cohorts is the amount of apoptotic mediator Fas ligand (APO010) to which the graft is exposed during incubation prior to ApoGraft transplantation and HSCT:
- 10 ng/mL APO010 in Cohort 1
- 25 ng/mL APO010 in Cohort 2
- 50 ng/mL APO010 in Cohort 3
- 100 ng/mL APO010 in Cohort 4.
The study is expected to progress from one cohort to the next based on an independent data safety monitoring board review and analysis of safety data.
About orphan designation
The FDA grants orphan designation to products intended to treat, diagnose, or prevent diseases/disorders that affect fewer than 200,000 people in the US.
The designation provides incentives for sponsors to develop products for rare diseases. This may include tax credits toward the cost of clinical trials, prescription drug user fee waivers, and 7 years of market exclusivity if the product is approved. ![]()
The US Food and Drug Administration (FDA) has granted orphan drug designation to ApoGraft™ as prophylaxis for acute and chronic graft-versus-host disease (GVHD) in transplant recipients.
ApoGraft is a mobilized peripheral blood cell product collected via apheresis from a matched, related donor. The product is exposed to the apoptotic mediator Fas ligand prior to transplantation.
ApoGraft was designed to eliminate immune responses after transplantation of foreign cells and tissues.
ApoGraft is being developed by Cellect Biotechnology Ltd.
The company is testing ApoGraft as acute GVHD prophylaxis in a phase 1/2 trial.
The trial is currently enrolling patients with hemato-oncology disorders who are eligible for allogeneic, HLA-matched hematopoietic stem cell transplant (HSCT).
The study is expected to have 4 cohorts, each consisting of 3 patients.
The difference between the cohorts is the amount of apoptotic mediator Fas ligand (APO010) to which the graft is exposed during incubation prior to ApoGraft transplantation and HSCT:
- 10 ng/mL APO010 in Cohort 1
- 25 ng/mL APO010 in Cohort 2
- 50 ng/mL APO010 in Cohort 3
- 100 ng/mL APO010 in Cohort 4.
The study is expected to progress from one cohort to the next based on an independent data safety monitoring board review and analysis of safety data.
About orphan designation
The FDA grants orphan designation to products intended to treat, diagnose, or prevent diseases/disorders that affect fewer than 200,000 people in the US.
The designation provides incentives for sponsors to develop products for rare diseases. This may include tax credits toward the cost of clinical trials, prescription drug user fee waivers, and 7 years of market exclusivity if the product is approved. ![]()
The US Food and Drug Administration (FDA) has granted orphan drug designation to ApoGraft™ as prophylaxis for acute and chronic graft-versus-host disease (GVHD) in transplant recipients.
ApoGraft is a mobilized peripheral blood cell product collected via apheresis from a matched, related donor. The product is exposed to the apoptotic mediator Fas ligand prior to transplantation.
ApoGraft was designed to eliminate immune responses after transplantation of foreign cells and tissues.
ApoGraft is being developed by Cellect Biotechnology Ltd.
The company is testing ApoGraft as acute GVHD prophylaxis in a phase 1/2 trial.
The trial is currently enrolling patients with hemato-oncology disorders who are eligible for allogeneic, HLA-matched hematopoietic stem cell transplant (HSCT).
The study is expected to have 4 cohorts, each consisting of 3 patients.
The difference between the cohorts is the amount of apoptotic mediator Fas ligand (APO010) to which the graft is exposed during incubation prior to ApoGraft transplantation and HSCT:
- 10 ng/mL APO010 in Cohort 1
- 25 ng/mL APO010 in Cohort 2
- 50 ng/mL APO010 in Cohort 3
- 100 ng/mL APO010 in Cohort 4.
The study is expected to progress from one cohort to the next based on an independent data safety monitoring board review and analysis of safety data.
About orphan designation
The FDA grants orphan designation to products intended to treat, diagnose, or prevent diseases/disorders that affect fewer than 200,000 people in the US.
The designation provides incentives for sponsors to develop products for rare diseases. This may include tax credits toward the cost of clinical trials, prescription drug user fee waivers, and 7 years of market exclusivity if the product is approved. ![]()
Withdrawn AML drug back on market in US
The US Food and Drug Administration (FDA) has approved use of gemtuzumab ozogamicin (GO, Mylotarg), a treatment that was initially approved by the agency in 2000 but later pulled from the US market.
GO is an antibody-drug conjugate that consists of the cytotoxic agent calicheamicin attached to a monoclonal antibody targeting CD33.
GO is now approved to treat adults with newly diagnosed, CD33-positive acute myeloid leukemia (AML) and patients age 2 and older with CD33-positive, relapsed or refractory AML.
GO can be given alone or in combination with daunorubicin and cytarabine.
The prescribing information for GO includes a boxed warning detailing the risk of hepatotoxicity, including veno-occlusive disease or sinusoidal obstruction syndrome, associated with GO.
GO originates from a collaboration between Pfizer and Celltech, now UCB. Pfizer has sole responsibility for all manufacturing, clinical development, and commercialization activities for this molecule.
Market withdrawal and subsequent trials
GO was originally approved under the FDA’s accelerated approval program in 2000 for use as a single agent in patients with CD33-positive AML who had experienced their first relapse and were 60 years of age or older.
In 2010, Pfizer voluntarily withdrew GO from the US market due to the results of a confirmatory phase 3 trial, SWOG S0106.
This trial showed there was no clinical benefit for patients who received GO plus daunorubicin and cytarabine over patients who received only daunorubicin and cytarabine.
In addition, the rate of fatal, treatment-related toxicity was significantly higher in the GO arm of the study.
Because of the unmet need for effective treatments in AML, investigators expressed an interest in evaluating different doses and schedules of GO.
These independent investigators, with Pfizer’s support, conducted clinical trials that yielded more information on the efficacy and safety of GO.
The trials—ALFA-0701, AML-19, and MyloFrance-1—supported the new approval of GO. Updated data from these trials are included in the prescribing information, which is available for download at www.mylotarg.com. ![]()
The US Food and Drug Administration (FDA) has approved use of gemtuzumab ozogamicin (GO, Mylotarg), a treatment that was initially approved by the agency in 2000 but later pulled from the US market.
GO is an antibody-drug conjugate that consists of the cytotoxic agent calicheamicin attached to a monoclonal antibody targeting CD33.
GO is now approved to treat adults with newly diagnosed, CD33-positive acute myeloid leukemia (AML) and patients age 2 and older with CD33-positive, relapsed or refractory AML.
GO can be given alone or in combination with daunorubicin and cytarabine.
The prescribing information for GO includes a boxed warning detailing the risk of hepatotoxicity, including veno-occlusive disease or sinusoidal obstruction syndrome, associated with GO.
GO originates from a collaboration between Pfizer and Celltech, now UCB. Pfizer has sole responsibility for all manufacturing, clinical development, and commercialization activities for this molecule.
Market withdrawal and subsequent trials
GO was originally approved under the FDA’s accelerated approval program in 2000 for use as a single agent in patients with CD33-positive AML who had experienced their first relapse and were 60 years of age or older.
In 2010, Pfizer voluntarily withdrew GO from the US market due to the results of a confirmatory phase 3 trial, SWOG S0106.
This trial showed there was no clinical benefit for patients who received GO plus daunorubicin and cytarabine over patients who received only daunorubicin and cytarabine.
In addition, the rate of fatal, treatment-related toxicity was significantly higher in the GO arm of the study.
Because of the unmet need for effective treatments in AML, investigators expressed an interest in evaluating different doses and schedules of GO.
These independent investigators, with Pfizer’s support, conducted clinical trials that yielded more information on the efficacy and safety of GO.
The trials—ALFA-0701, AML-19, and MyloFrance-1—supported the new approval of GO. Updated data from these trials are included in the prescribing information, which is available for download at www.mylotarg.com. ![]()
The US Food and Drug Administration (FDA) has approved use of gemtuzumab ozogamicin (GO, Mylotarg), a treatment that was initially approved by the agency in 2000 but later pulled from the US market.
GO is an antibody-drug conjugate that consists of the cytotoxic agent calicheamicin attached to a monoclonal antibody targeting CD33.
GO is now approved to treat adults with newly diagnosed, CD33-positive acute myeloid leukemia (AML) and patients age 2 and older with CD33-positive, relapsed or refractory AML.
GO can be given alone or in combination with daunorubicin and cytarabine.
The prescribing information for GO includes a boxed warning detailing the risk of hepatotoxicity, including veno-occlusive disease or sinusoidal obstruction syndrome, associated with GO.
GO originates from a collaboration between Pfizer and Celltech, now UCB. Pfizer has sole responsibility for all manufacturing, clinical development, and commercialization activities for this molecule.
Market withdrawal and subsequent trials
GO was originally approved under the FDA’s accelerated approval program in 2000 for use as a single agent in patients with CD33-positive AML who had experienced their first relapse and were 60 years of age or older.
In 2010, Pfizer voluntarily withdrew GO from the US market due to the results of a confirmatory phase 3 trial, SWOG S0106.
This trial showed there was no clinical benefit for patients who received GO plus daunorubicin and cytarabine over patients who received only daunorubicin and cytarabine.
In addition, the rate of fatal, treatment-related toxicity was significantly higher in the GO arm of the study.
Because of the unmet need for effective treatments in AML, investigators expressed an interest in evaluating different doses and schedules of GO.
These independent investigators, with Pfizer’s support, conducted clinical trials that yielded more information on the efficacy and safety of GO.
The trials—ALFA-0701, AML-19, and MyloFrance-1—supported the new approval of GO. Updated data from these trials are included in the prescribing information, which is available for download at www.mylotarg.com. ![]()
FDA reapproves gemtuzumab ozogamicin for CD33-positive AML treatment
The Food and Drug Administration has approved gemtuzumab ozogamicin (Mylotarg) for the treatment of newly diagnosed CD33-positive acute myeloid leukemia in adults, according to a press release.
Approval was based on results from three clinical trials. In the first, newly diagnosed AML patients who received gemtuzumab ozogamicin plus chemotherapy had significantly longer event-free survival than did patients who received chemotherapy alone. In a second trial, patients who received gemtuzumab ozogamicin alone had better overall survival compared to those who received only best supportive care. In the third clinical trial, 26% of patients who had experienced a relapse and received gemtuzumab ozogamicin experienced a remission.
Common side effects of gemtuzumab ozogamicin include fever, nausea, infection, vomiting, bleeding, thrombocytopenia, stomatitis, constipation, rash, headache, elevated liver function tests, and neutropenia; it is not recommended for women who are pregnant or breastfeeding.
Gemtuzumab ozogamicin was also approved to treat patients older than 2 years old who have experienced a relapse or have not responded to initial treatment.
“Mylotarg’s history underscores the importance of examining alternative dosing, scheduling, and administration of therapies for patients with cancer, especially in those who may be most vulnerable to the side effects of treatment,” Richard Pazdur, MD, director of the FDA’s Oncology Center of Excellence, said in the press release.
The Food and Drug Administration has approved gemtuzumab ozogamicin (Mylotarg) for the treatment of newly diagnosed CD33-positive acute myeloid leukemia in adults, according to a press release.
Approval was based on results from three clinical trials. In the first, newly diagnosed AML patients who received gemtuzumab ozogamicin plus chemotherapy had significantly longer event-free survival than did patients who received chemotherapy alone. In a second trial, patients who received gemtuzumab ozogamicin alone had better overall survival compared to those who received only best supportive care. In the third clinical trial, 26% of patients who had experienced a relapse and received gemtuzumab ozogamicin experienced a remission.
Common side effects of gemtuzumab ozogamicin include fever, nausea, infection, vomiting, bleeding, thrombocytopenia, stomatitis, constipation, rash, headache, elevated liver function tests, and neutropenia; it is not recommended for women who are pregnant or breastfeeding.
Gemtuzumab ozogamicin was also approved to treat patients older than 2 years old who have experienced a relapse or have not responded to initial treatment.
“Mylotarg’s history underscores the importance of examining alternative dosing, scheduling, and administration of therapies for patients with cancer, especially in those who may be most vulnerable to the side effects of treatment,” Richard Pazdur, MD, director of the FDA’s Oncology Center of Excellence, said in the press release.
The Food and Drug Administration has approved gemtuzumab ozogamicin (Mylotarg) for the treatment of newly diagnosed CD33-positive acute myeloid leukemia in adults, according to a press release.
Approval was based on results from three clinical trials. In the first, newly diagnosed AML patients who received gemtuzumab ozogamicin plus chemotherapy had significantly longer event-free survival than did patients who received chemotherapy alone. In a second trial, patients who received gemtuzumab ozogamicin alone had better overall survival compared to those who received only best supportive care. In the third clinical trial, 26% of patients who had experienced a relapse and received gemtuzumab ozogamicin experienced a remission.
Common side effects of gemtuzumab ozogamicin include fever, nausea, infection, vomiting, bleeding, thrombocytopenia, stomatitis, constipation, rash, headache, elevated liver function tests, and neutropenia; it is not recommended for women who are pregnant or breastfeeding.
Gemtuzumab ozogamicin was also approved to treat patients older than 2 years old who have experienced a relapse or have not responded to initial treatment.
“Mylotarg’s history underscores the importance of examining alternative dosing, scheduling, and administration of therapies for patients with cancer, especially in those who may be most vulnerable to the side effects of treatment,” Richard Pazdur, MD, director of the FDA’s Oncology Center of Excellence, said in the press release.
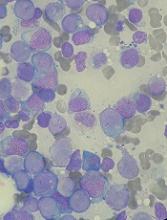
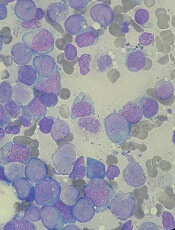
Nanocarriers could treat leukemia, lymphoma and improve HSCT
Researchers say they have created nanoparticles loaded with messenger RNA (mRNA) that can give cells the ability to fight cancers and other diseases.
To use these freeze-dried nanocarriers, the team added water and introduced the resulting mixture to cells.
The nanocarriers were able to target T cells and hematopoietic stem cells (HSCs), delivering mRNA directly to the cells and triggering short-term gene expression.
The T cells were then able to fight leukemia and lymphoma in vitro and in vivo. And the HSCs demonstrated improvements in growth and regenerative potential.
Matthias Stephan, MD, PhD, of Fred Hutchinson Cancer Research Center in Seattle, Washington, and his colleagues described this research in Nature Communications.
“We developed a nanocarrier that binds and condenses synthetic mRNA and protects it from degradation,” Dr Stephan said.
The researchers surrounded the nanocarrier with a negatively charged envelope with a targeting ligand attached to the surface so the carrier homes and binds to a particular cell type. When this happens, the cell engulfs the carrier, which can be loaded with different types of manmade mRNA.
The researchers mixed the freeze-dried nanocarriers with water and samples of cells. Within 4 hours, cells started showing signs that editing had taken effect.
The team noted that boosters can be given if needed. And the nanocarriers are made from a dissolving biomaterial, so they are removed from the body like other cell waste.
Testing the carriers
Dr Stephan and his colleagues tested their nanocarriers in 3 ways.
First, the researchers tested nanoparticles carrying a gene-editing tool to T cells that snipped out their natural T-cell receptors and was paired with genes encoding a chimeric antigen receptor (CAR).
The resulting CAR T cells maintained their ability to proliferate and successfully eliminated leukemia cells.
Next, the researchers tested nanocarriers targeted to CAR T cells and containing foxo1 mRNA. This prompted the T cells to develop into a type of memory cell with enhanced antitumor activity.
The team found these CAR T cells induced “substantial disease regression” and prolonged survival in a mouse model of B-cell lymphoma.
Finally, the researchers tested nanocarriers targeted to HSCs. The carriers were equipped with mRNA that “induced key regulators of self-renewal,” accelerating the growth and regenerative potential of the HSCs in vitro.
Future possibilities
Dr Stephan and his colleagues noted that these nanocarriers are built on existing technology and can be used by individuals without knowledge of nanotechnology. Therefore, the team hopes the nanocarriers will be an off-the-shelf way for cell-therapy engineers to develop new approaches to treat diseases.
The researchers believe the nanocarriers could replace electroporation, a multistep cell-manufacturing technique that requires specialized equipment and clean rooms. The team noted that up to 60 times more cells survive the introduction of the nanocarriers than survive electroporation.
“You can imagine taking the nanoparticles, injecting them into a patient, and then you don’t have to culture cells at all anymore,” Dr Stephan said.
He is now looking for commercial partners to move the technology toward additional applications and into clinical trials. ![]()
Researchers say they have created nanoparticles loaded with messenger RNA (mRNA) that can give cells the ability to fight cancers and other diseases.
To use these freeze-dried nanocarriers, the team added water and introduced the resulting mixture to cells.
The nanocarriers were able to target T cells and hematopoietic stem cells (HSCs), delivering mRNA directly to the cells and triggering short-term gene expression.
The T cells were then able to fight leukemia and lymphoma in vitro and in vivo. And the HSCs demonstrated improvements in growth and regenerative potential.
Matthias Stephan, MD, PhD, of Fred Hutchinson Cancer Research Center in Seattle, Washington, and his colleagues described this research in Nature Communications.
“We developed a nanocarrier that binds and condenses synthetic mRNA and protects it from degradation,” Dr Stephan said.
The researchers surrounded the nanocarrier with a negatively charged envelope with a targeting ligand attached to the surface so the carrier homes and binds to a particular cell type. When this happens, the cell engulfs the carrier, which can be loaded with different types of manmade mRNA.
The researchers mixed the freeze-dried nanocarriers with water and samples of cells. Within 4 hours, cells started showing signs that editing had taken effect.
The team noted that boosters can be given if needed. And the nanocarriers are made from a dissolving biomaterial, so they are removed from the body like other cell waste.
Testing the carriers
Dr Stephan and his colleagues tested their nanocarriers in 3 ways.
First, the researchers tested nanoparticles carrying a gene-editing tool to T cells that snipped out their natural T-cell receptors and was paired with genes encoding a chimeric antigen receptor (CAR).
The resulting CAR T cells maintained their ability to proliferate and successfully eliminated leukemia cells.
Next, the researchers tested nanocarriers targeted to CAR T cells and containing foxo1 mRNA. This prompted the T cells to develop into a type of memory cell with enhanced antitumor activity.
The team found these CAR T cells induced “substantial disease regression” and prolonged survival in a mouse model of B-cell lymphoma.
Finally, the researchers tested nanocarriers targeted to HSCs. The carriers were equipped with mRNA that “induced key regulators of self-renewal,” accelerating the growth and regenerative potential of the HSCs in vitro.
Future possibilities
Dr Stephan and his colleagues noted that these nanocarriers are built on existing technology and can be used by individuals without knowledge of nanotechnology. Therefore, the team hopes the nanocarriers will be an off-the-shelf way for cell-therapy engineers to develop new approaches to treat diseases.
The researchers believe the nanocarriers could replace electroporation, a multistep cell-manufacturing technique that requires specialized equipment and clean rooms. The team noted that up to 60 times more cells survive the introduction of the nanocarriers than survive electroporation.
“You can imagine taking the nanoparticles, injecting them into a patient, and then you don’t have to culture cells at all anymore,” Dr Stephan said.
He is now looking for commercial partners to move the technology toward additional applications and into clinical trials. ![]()
Researchers say they have created nanoparticles loaded with messenger RNA (mRNA) that can give cells the ability to fight cancers and other diseases.
To use these freeze-dried nanocarriers, the team added water and introduced the resulting mixture to cells.
The nanocarriers were able to target T cells and hematopoietic stem cells (HSCs), delivering mRNA directly to the cells and triggering short-term gene expression.
The T cells were then able to fight leukemia and lymphoma in vitro and in vivo. And the HSCs demonstrated improvements in growth and regenerative potential.
Matthias Stephan, MD, PhD, of Fred Hutchinson Cancer Research Center in Seattle, Washington, and his colleagues described this research in Nature Communications.
“We developed a nanocarrier that binds and condenses synthetic mRNA and protects it from degradation,” Dr Stephan said.
The researchers surrounded the nanocarrier with a negatively charged envelope with a targeting ligand attached to the surface so the carrier homes and binds to a particular cell type. When this happens, the cell engulfs the carrier, which can be loaded with different types of manmade mRNA.
The researchers mixed the freeze-dried nanocarriers with water and samples of cells. Within 4 hours, cells started showing signs that editing had taken effect.
The team noted that boosters can be given if needed. And the nanocarriers are made from a dissolving biomaterial, so they are removed from the body like other cell waste.
Testing the carriers
Dr Stephan and his colleagues tested their nanocarriers in 3 ways.
First, the researchers tested nanoparticles carrying a gene-editing tool to T cells that snipped out their natural T-cell receptors and was paired with genes encoding a chimeric antigen receptor (CAR).
The resulting CAR T cells maintained their ability to proliferate and successfully eliminated leukemia cells.
Next, the researchers tested nanocarriers targeted to CAR T cells and containing foxo1 mRNA. This prompted the T cells to develop into a type of memory cell with enhanced antitumor activity.
The team found these CAR T cells induced “substantial disease regression” and prolonged survival in a mouse model of B-cell lymphoma.
Finally, the researchers tested nanocarriers targeted to HSCs. The carriers were equipped with mRNA that “induced key regulators of self-renewal,” accelerating the growth and regenerative potential of the HSCs in vitro.
Future possibilities
Dr Stephan and his colleagues noted that these nanocarriers are built on existing technology and can be used by individuals without knowledge of nanotechnology. Therefore, the team hopes the nanocarriers will be an off-the-shelf way for cell-therapy engineers to develop new approaches to treat diseases.
The researchers believe the nanocarriers could replace electroporation, a multistep cell-manufacturing technique that requires specialized equipment and clean rooms. The team noted that up to 60 times more cells survive the introduction of the nanocarriers than survive electroporation.
“You can imagine taking the nanoparticles, injecting them into a patient, and then you don’t have to culture cells at all anymore,” Dr Stephan said.
He is now looking for commercial partners to move the technology toward additional applications and into clinical trials. ![]()
Young cancer patients want more digital support, survey says
A recent survey of young cancer patients in the UK revealed their desire for additional digital oncology resources.
The survey showed that many of the patients already used digital resources to access information about their cancer.
However, many also expressed a need for additional resources such as online counseling or psychological support.
Esha Abrol, of Camden and Islington NHS Foundation Trust in London, UK, and her colleagues reported these findings in the Journal of Cancer Survivorship.
The researchers surveyed 102 cancer patients, ages 13 to 24, about digital media use.
The patients reported having active digital lives, with 41.6% of them rating digital resources as “essential” to their lives.
The patients reported using a variety of healthcare-related digital resources to access information about their disease, including independent sources and ones recommended to them by the professional team treating them.
Half (51%) of respondents said they kept in contact through digital means with other patients they had met during treatment. Twelve percent contacted others or started new digital relationships with people they had never met in person.
However, most of the patients were still most likely to get information about their treatment from professionals in a face-to-face environment, such as when visiting their doctor.
“This is a reassuring and appropriate finding, as this is the conventional means by which teenage and young adult oncology care is delivered by the multidisciplinary team, whether the young people choose to engage or not,” Abrol said.
At the same time, many survey respondents expressed a desire for virtual online groups (54.3%), online counseling or psychological support (43.5%), and the ability to receive (66.3%) and share (48.9%) clinical information online.
Young adults (ages 19 to 24) were more interested than adolescents (ages 13 to 18) in online counseling options and preferred receiving clinical information online.
The researchers said this may reflect young adults’ greater independence, resilience, breadth of experience in the digital world, and confidence in discussing clinical matters online.
Abrol believes these preliminary results can help inform the development of local, national, and global services to teenagers and young adults with cancer to address their unmet needs.
“These digital support resources have the potential to improve patient experience and engagement for an important subsection of teenagers and young people treated for cancer,” Abrol said. ![]()
A recent survey of young cancer patients in the UK revealed their desire for additional digital oncology resources.
The survey showed that many of the patients already used digital resources to access information about their cancer.
However, many also expressed a need for additional resources such as online counseling or psychological support.
Esha Abrol, of Camden and Islington NHS Foundation Trust in London, UK, and her colleagues reported these findings in the Journal of Cancer Survivorship.
The researchers surveyed 102 cancer patients, ages 13 to 24, about digital media use.
The patients reported having active digital lives, with 41.6% of them rating digital resources as “essential” to their lives.
The patients reported using a variety of healthcare-related digital resources to access information about their disease, including independent sources and ones recommended to them by the professional team treating them.
Half (51%) of respondents said they kept in contact through digital means with other patients they had met during treatment. Twelve percent contacted others or started new digital relationships with people they had never met in person.
However, most of the patients were still most likely to get information about their treatment from professionals in a face-to-face environment, such as when visiting their doctor.
“This is a reassuring and appropriate finding, as this is the conventional means by which teenage and young adult oncology care is delivered by the multidisciplinary team, whether the young people choose to engage or not,” Abrol said.
At the same time, many survey respondents expressed a desire for virtual online groups (54.3%), online counseling or psychological support (43.5%), and the ability to receive (66.3%) and share (48.9%) clinical information online.
Young adults (ages 19 to 24) were more interested than adolescents (ages 13 to 18) in online counseling options and preferred receiving clinical information online.
The researchers said this may reflect young adults’ greater independence, resilience, breadth of experience in the digital world, and confidence in discussing clinical matters online.
Abrol believes these preliminary results can help inform the development of local, national, and global services to teenagers and young adults with cancer to address their unmet needs.
“These digital support resources have the potential to improve patient experience and engagement for an important subsection of teenagers and young people treated for cancer,” Abrol said. ![]()
A recent survey of young cancer patients in the UK revealed their desire for additional digital oncology resources.
The survey showed that many of the patients already used digital resources to access information about their cancer.
However, many also expressed a need for additional resources such as online counseling or psychological support.
Esha Abrol, of Camden and Islington NHS Foundation Trust in London, UK, and her colleagues reported these findings in the Journal of Cancer Survivorship.
The researchers surveyed 102 cancer patients, ages 13 to 24, about digital media use.
The patients reported having active digital lives, with 41.6% of them rating digital resources as “essential” to their lives.
The patients reported using a variety of healthcare-related digital resources to access information about their disease, including independent sources and ones recommended to them by the professional team treating them.
Half (51%) of respondents said they kept in contact through digital means with other patients they had met during treatment. Twelve percent contacted others or started new digital relationships with people they had never met in person.
However, most of the patients were still most likely to get information about their treatment from professionals in a face-to-face environment, such as when visiting their doctor.
“This is a reassuring and appropriate finding, as this is the conventional means by which teenage and young adult oncology care is delivered by the multidisciplinary team, whether the young people choose to engage or not,” Abrol said.
At the same time, many survey respondents expressed a desire for virtual online groups (54.3%), online counseling or psychological support (43.5%), and the ability to receive (66.3%) and share (48.9%) clinical information online.
Young adults (ages 19 to 24) were more interested than adolescents (ages 13 to 18) in online counseling options and preferred receiving clinical information online.
The researchers said this may reflect young adults’ greater independence, resilience, breadth of experience in the digital world, and confidence in discussing clinical matters online.
Abrol believes these preliminary results can help inform the development of local, national, and global services to teenagers and young adults with cancer to address their unmet needs.
“These digital support resources have the potential to improve patient experience and engagement for an important subsection of teenagers and young people treated for cancer,” Abrol said.
FDA approves first CAR T-cell therapy to treat ALL
The US Food and Drug Administration (FDA) has approved the first chimeric antigen receptor (CAR) T-cell therapy, tisagenlecleucel (KymriahTM, formerly CTL019).
The therapy is approved for use in children and young adults up to 25 years of age who have B-cell precursor acute lymphoblastic leukemia (ALL) that is refractory or in second or later relapse.
Tisagenlecleucel consists of autologous T cells expressing a CD19-specific CAR.
The therapy was first developed by the University of Pennsylvania. In 2012, the university and Novartis entered into a global collaboration to further research, develop, and commercialize CAR T-cell therapies. Novartis holds the worldwide rights to tisagenlecleucel and other therapies developed through the collaboration.
The application for tisagenlecleucel was supported by results from 3 clinical trials:
- A pilot study presented at the 2015 ASH Annual Meeting
- The phase 2 ENSIGN trial, which was presented at the 2016 ASH Annual Meeting
- The phase 2 ELIANA study, which was recently presented at the 22nd Congress of the European Hematology Association (EHA).
Safety concerns
The prescribing information for tisagenlecleucel includes a boxed warning noting that the treatment poses a risk of cytokine release syndrome (CRS) and neurological toxicity, both of which can be life-threatening.
Because of the risk of CRS, the FDA has expanded the approved use of tocilizumab (Actemra) to include treatment of CAR T-cell-induced severe or life-threatening CRS in patients age 2 and older.
The risk of CRS and neurological toxicity also prompted the FDA to approve tisagenlecleucel with a risk evaluation and mitigation strategy (REMS), which includes elements to assure safe use.
The FDA is requiring that hospitals and their associated clinics that dispense tisagenlecleucel be specially certified. As part of that certification, staff involved in the prescribing, dispensing, or administration of tisagenlecleucel are required to be trained to recognize and manage CRS and neurological events.
Additionally, the certified healthcare settings are required to have protocols in place to ensure that tisagenlecleucel is only given to patients after verifying that tocilizumab is available for immediate administration.
The REMS program specifies that patients be informed of the signs and symptoms of CRS and neurological toxicities following infusion and of the importance of promptly returning to the treatment site if they develop fever or other adverse reactions after receiving tisagenlecleucel.
To further evaluate the long-term safety of tisagenlecleucel, Novartis is required to conduct a post-marketing observational study involving patients who received the treatment.
Access and cost
Tisagenlecleucel will be manufactured for each individual patient at Novartis’s facility in Morris Plains, New Jersey.
Novartis said it has designed a manufacturing and supply chain platform that allows for an individualized treatment approach on a global scale. This process includes cryopreservation of a patient’s harvested cells, providing the flexibility to initiate treatment with tisagenlecleucel based on the individual patient’s condition.
Tisagenlecleucel will reportedly cost $475,000 for a single course of treatment. However, Novartis said it will help patients navigate insurance coverage and provide financial assistance for those who are uninsured or underinsured.
In addition, patients will only be required to pay for tisagenlecleucel if they respond within a month of receiving the treatment. This is a result of a collaboration between Novartis and the US Centers for Medicare and Medicaid Services that is focused on delivering value-based care. The approach is intended to include indication-based pricing for medicines and supports payments for a medicine based on the clinical outcomes achieved.
The US Food and Drug Administration (FDA) has approved the first chimeric antigen receptor (CAR) T-cell therapy, tisagenlecleucel (KymriahTM, formerly CTL019).
The therapy is approved for use in children and young adults up to 25 years of age who have B-cell precursor acute lymphoblastic leukemia (ALL) that is refractory or in second or later relapse.
Tisagenlecleucel consists of autologous T cells expressing a CD19-specific CAR.
The therapy was first developed by the University of Pennsylvania. In 2012, the university and Novartis entered into a global collaboration to further research, develop, and commercialize CAR T-cell therapies. Novartis holds the worldwide rights to tisagenlecleucel and other therapies developed through the collaboration.
The application for tisagenlecleucel was supported by results from 3 clinical trials:
- A pilot study presented at the 2015 ASH Annual Meeting
- The phase 2 ENSIGN trial, which was presented at the 2016 ASH Annual Meeting
- The phase 2 ELIANA study, which was recently presented at the 22nd Congress of the European Hematology Association (EHA).
Safety concerns
The prescribing information for tisagenlecleucel includes a boxed warning noting that the treatment poses a risk of cytokine release syndrome (CRS) and neurological toxicity, both of which can be life-threatening.
Because of the risk of CRS, the FDA has expanded the approved use of tocilizumab (Actemra) to include treatment of CAR T-cell-induced severe or life-threatening CRS in patients age 2 and older.
The risk of CRS and neurological toxicity also prompted the FDA to approve tisagenlecleucel with a risk evaluation and mitigation strategy (REMS), which includes elements to assure safe use.
The FDA is requiring that hospitals and their associated clinics that dispense tisagenlecleucel be specially certified. As part of that certification, staff involved in the prescribing, dispensing, or administration of tisagenlecleucel are required to be trained to recognize and manage CRS and neurological events.
Additionally, the certified healthcare settings are required to have protocols in place to ensure that tisagenlecleucel is only given to patients after verifying that tocilizumab is available for immediate administration.
The REMS program specifies that patients be informed of the signs and symptoms of CRS and neurological toxicities following infusion and of the importance of promptly returning to the treatment site if they develop fever or other adverse reactions after receiving tisagenlecleucel.
To further evaluate the long-term safety of tisagenlecleucel, Novartis is required to conduct a post-marketing observational study involving patients who received the treatment.
Access and cost
Tisagenlecleucel will be manufactured for each individual patient at Novartis’s facility in Morris Plains, New Jersey.
Novartis said it has designed a manufacturing and supply chain platform that allows for an individualized treatment approach on a global scale. This process includes cryopreservation of a patient’s harvested cells, providing the flexibility to initiate treatment with tisagenlecleucel based on the individual patient’s condition.
Tisagenlecleucel will reportedly cost $475,000 for a single course of treatment. However, Novartis said it will help patients navigate insurance coverage and provide financial assistance for those who are uninsured or underinsured.
In addition, patients will only be required to pay for tisagenlecleucel if they respond within a month of receiving the treatment. This is a result of a collaboration between Novartis and the US Centers for Medicare and Medicaid Services that is focused on delivering value-based care. The approach is intended to include indication-based pricing for medicines and supports payments for a medicine based on the clinical outcomes achieved.
The US Food and Drug Administration (FDA) has approved the first chimeric antigen receptor (CAR) T-cell therapy, tisagenlecleucel (KymriahTM, formerly CTL019).
The therapy is approved for use in children and young adults up to 25 years of age who have B-cell precursor acute lymphoblastic leukemia (ALL) that is refractory or in second or later relapse.
Tisagenlecleucel consists of autologous T cells expressing a CD19-specific CAR.
The therapy was first developed by the University of Pennsylvania. In 2012, the university and Novartis entered into a global collaboration to further research, develop, and commercialize CAR T-cell therapies. Novartis holds the worldwide rights to tisagenlecleucel and other therapies developed through the collaboration.
The application for tisagenlecleucel was supported by results from 3 clinical trials:
- A pilot study presented at the 2015 ASH Annual Meeting
- The phase 2 ENSIGN trial, which was presented at the 2016 ASH Annual Meeting
- The phase 2 ELIANA study, which was recently presented at the 22nd Congress of the European Hematology Association (EHA).
Safety concerns
The prescribing information for tisagenlecleucel includes a boxed warning noting that the treatment poses a risk of cytokine release syndrome (CRS) and neurological toxicity, both of which can be life-threatening.
Because of the risk of CRS, the FDA has expanded the approved use of tocilizumab (Actemra) to include treatment of CAR T-cell-induced severe or life-threatening CRS in patients age 2 and older.
The risk of CRS and neurological toxicity also prompted the FDA to approve tisagenlecleucel with a risk evaluation and mitigation strategy (REMS), which includes elements to assure safe use.
The FDA is requiring that hospitals and their associated clinics that dispense tisagenlecleucel be specially certified. As part of that certification, staff involved in the prescribing, dispensing, or administration of tisagenlecleucel are required to be trained to recognize and manage CRS and neurological events.
Additionally, the certified healthcare settings are required to have protocols in place to ensure that tisagenlecleucel is only given to patients after verifying that tocilizumab is available for immediate administration.
The REMS program specifies that patients be informed of the signs and symptoms of CRS and neurological toxicities following infusion and of the importance of promptly returning to the treatment site if they develop fever or other adverse reactions after receiving tisagenlecleucel.
To further evaluate the long-term safety of tisagenlecleucel, Novartis is required to conduct a post-marketing observational study involving patients who received the treatment.
Access and cost
Tisagenlecleucel will be manufactured for each individual patient at Novartis’s facility in Morris Plains, New Jersey.
Novartis said it has designed a manufacturing and supply chain platform that allows for an individualized treatment approach on a global scale. This process includes cryopreservation of a patient’s harvested cells, providing the flexibility to initiate treatment with tisagenlecleucel based on the individual patient’s condition.
Tisagenlecleucel will reportedly cost $475,000 for a single course of treatment. However, Novartis said it will help patients navigate insurance coverage and provide financial assistance for those who are uninsured or underinsured.
In addition, patients will only be required to pay for tisagenlecleucel if they respond within a month of receiving the treatment. This is a result of a collaboration between Novartis and the US Centers for Medicare and Medicaid Services that is focused on delivering value-based care. The approach is intended to include indication-based pricing for medicines and supports payments for a medicine based on the clinical outcomes achieved.