User login
Health Canada approves product for adult ALL
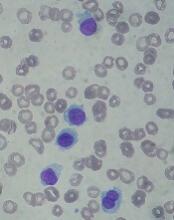
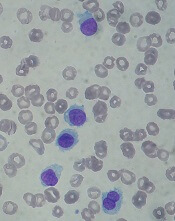
Health Canada has approved inotuzumab ozogamicin (Besponsa™) as monotherapy for adults with relapsed or refractory, CD22-positive, B-cell precursor acute lymphoblastic leukemia (ALL).
Inotuzumab ozogamicin is the first and only CD22-directed antibody-drug conjugate approved for this indication.
The product consists of a monoclonal antibody targeting CD22 and a cytotoxic agent known as calicheamicin.
Health Canada’s approval of inotuzumab ozogamicin is based on results from the phase 3 INO-VATE trial, which were published in NEJM in June 2016.
The trial enrolled 326 adults with relapsed or refractory B-cell precursor ALL.
Patients received inotuzumab ozogamicin or 1 of 3 chemotherapy regimens—high-dose cytarabine; cytarabine plus mitoxantrone; or fludarabine, cytarabine, and granulocyte colony-stimulating factor.
The rate of complete remission, including incomplete hematologic recovery, was 80.7% in the inotuzumab arm and 29.4% in the chemotherapy arm (P<0.001). The median duration of remission was 4.6 months and 3.1 months, respectively (P=0.03).
Forty-one percent of patients treated with inotuzumab and 11% of those who received chemotherapy proceeded to stem cell transplant directly after treatment (P<0.001).
The median progression-free survival was 5.0 months in the inotuzumab arm and 1.8 months in the chemotherapy arm (P<0.001).
The median overall survival was 7.7 months and 6.7 months, respectively (P=0.04). This did not meet the prespecified boundary of significance (P=0.0208).
Liver-related adverse events were more common in the inotuzumab arm than the chemotherapy arm. The most frequent of these were increased aspartate aminotransferase level (20% vs 10%), hyperbilirubinemia (15% vs 10%), and increased alanine aminotransferase level (14% vs 11%).
Veno-occlusive liver disease occurred in 11% of patients in the inotuzumab arm and 1% in the chemotherapy arm.
There were 17 deaths during treatment in the inotuzumab arm and 11 in the chemotherapy arm. Four deaths were considered related to inotuzumab, and 2 were deemed related to chemotherapy.
Health Canada has approved inotuzumab ozogamicin (Besponsa™) as monotherapy for adults with relapsed or refractory, CD22-positive, B-cell precursor acute lymphoblastic leukemia (ALL).
Inotuzumab ozogamicin is the first and only CD22-directed antibody-drug conjugate approved for this indication.
The product consists of a monoclonal antibody targeting CD22 and a cytotoxic agent known as calicheamicin.
Health Canada’s approval of inotuzumab ozogamicin is based on results from the phase 3 INO-VATE trial, which were published in NEJM in June 2016.
The trial enrolled 326 adults with relapsed or refractory B-cell precursor ALL.
Patients received inotuzumab ozogamicin or 1 of 3 chemotherapy regimens—high-dose cytarabine; cytarabine plus mitoxantrone; or fludarabine, cytarabine, and granulocyte colony-stimulating factor.
The rate of complete remission, including incomplete hematologic recovery, was 80.7% in the inotuzumab arm and 29.4% in the chemotherapy arm (P<0.001). The median duration of remission was 4.6 months and 3.1 months, respectively (P=0.03).
Forty-one percent of patients treated with inotuzumab and 11% of those who received chemotherapy proceeded to stem cell transplant directly after treatment (P<0.001).
The median progression-free survival was 5.0 months in the inotuzumab arm and 1.8 months in the chemotherapy arm (P<0.001).
The median overall survival was 7.7 months and 6.7 months, respectively (P=0.04). This did not meet the prespecified boundary of significance (P=0.0208).
Liver-related adverse events were more common in the inotuzumab arm than the chemotherapy arm. The most frequent of these were increased aspartate aminotransferase level (20% vs 10%), hyperbilirubinemia (15% vs 10%), and increased alanine aminotransferase level (14% vs 11%).
Veno-occlusive liver disease occurred in 11% of patients in the inotuzumab arm and 1% in the chemotherapy arm.
There were 17 deaths during treatment in the inotuzumab arm and 11 in the chemotherapy arm. Four deaths were considered related to inotuzumab, and 2 were deemed related to chemotherapy.
Health Canada has approved inotuzumab ozogamicin (Besponsa™) as monotherapy for adults with relapsed or refractory, CD22-positive, B-cell precursor acute lymphoblastic leukemia (ALL).
Inotuzumab ozogamicin is the first and only CD22-directed antibody-drug conjugate approved for this indication.
The product consists of a monoclonal antibody targeting CD22 and a cytotoxic agent known as calicheamicin.
Health Canada’s approval of inotuzumab ozogamicin is based on results from the phase 3 INO-VATE trial, which were published in NEJM in June 2016.
The trial enrolled 326 adults with relapsed or refractory B-cell precursor ALL.
Patients received inotuzumab ozogamicin or 1 of 3 chemotherapy regimens—high-dose cytarabine; cytarabine plus mitoxantrone; or fludarabine, cytarabine, and granulocyte colony-stimulating factor.
The rate of complete remission, including incomplete hematologic recovery, was 80.7% in the inotuzumab arm and 29.4% in the chemotherapy arm (P<0.001). The median duration of remission was 4.6 months and 3.1 months, respectively (P=0.03).
Forty-one percent of patients treated with inotuzumab and 11% of those who received chemotherapy proceeded to stem cell transplant directly after treatment (P<0.001).
The median progression-free survival was 5.0 months in the inotuzumab arm and 1.8 months in the chemotherapy arm (P<0.001).
The median overall survival was 7.7 months and 6.7 months, respectively (P=0.04). This did not meet the prespecified boundary of significance (P=0.0208).
Liver-related adverse events were more common in the inotuzumab arm than the chemotherapy arm. The most frequent of these were increased aspartate aminotransferase level (20% vs 10%), hyperbilirubinemia (15% vs 10%), and increased alanine aminotransferase level (14% vs 11%).
Veno-occlusive liver disease occurred in 11% of patients in the inotuzumab arm and 1% in the chemotherapy arm.
There were 17 deaths during treatment in the inotuzumab arm and 11 in the chemotherapy arm. Four deaths were considered related to inotuzumab, and 2 were deemed related to chemotherapy.
Drug receives priority review for HCL
The US Food and Drug Administration (FDA) has accepted for priority review the biologics license application (BLA) for moxetumomab pasudotox, an investigational anti-CD22 recombinant immunotoxin.
With this BLA, AstraZeneca is seeking approval for moxetumomab pasudotox for the treatment of adults with hairy cell leukemia (HCL) who have received at least 2 prior lines of therapy.
The FDA expects to make a decision on the BLA in the third quarter of this year.
The FDA aims to take action on a priority review application within 6 months of receiving it, rather than the standard 10 months.
The agency grants priority review to applications for products that may provide significant improvements in the treatment, diagnosis, or prevention of serious conditions.
About moxetumomab pasudotox
Moxetumomab pasudotox (formerly CAT-8015 or HA22) is composed of a binding portion of an anti-CD22 antibody fused to a toxin. After binding to CD22, the molecule is internalized, processed, and releases its modified protein toxin, which inhibits protein translation and leads to apoptosis.
In addition to priority review, moxetumomab pasudotox has received orphan drug designation from the FDA.
Moxetumomab pasudotox has been tested in a phase 1 trial. Initial results from this trial were published in the Journal of Clinical Oncology in 2012. Long-term follow-up was presented at the 2017 ASH Annual Meeting.
The ASH data included 49 patients with relapsed/refractory HCL. Their median age was 57 (range, 40-77), most (n=41) were male, and they had a median of 35 (range, 1-60,444) circulating HCL cells/mm3 at baseline (in 48 evaluable patients).
Twenty-eight patients received moxetumomab pasudotox in the dose-escalation portion of the study—at 5, 10, 20, 30, 40, or 50 µg/kg—and 21 received the drug at 50 µg/kg for the extension portion of the study.
Among the 33 patients who received moxetumomab pasudotox at 50 µg/kg, the overall response rate was 88%, and the complete response (CR) rate was 64% (n=21). The median time to CR was 3.6 months, and the median duration of CR was 70.3 months.
The median follow-up was 75 months for the entire study population. At 72 months, the progression-free survival (PFS) rate was 77%.
The researchers found that minimal residual disease (MRD) negativity (via immunohistochemistry) was associated with extended response duration and prolonged PFS.
The MRD evaluation included 19 MRD+ patients and 18 MRD- patients. Forty-seven percent of the MRD+ patients (n=9) and 94% of the MRD- patients (n=17) had a CR as their best response.
The median duration of CR was 13.1 months among the MRD+ patients and was not reached among the MRD- patients (P=0.0002). The median PFS was 82.1 months among the MRD+ patients and not reached among the MRD- patients (P=0.0031).
Moxetumomab pasudotox did not undergo phase 2 testing but proceeded to a phase 3 trial. In this single-arm study, researchers evaluated the drug in HCL patients who had received at least 2 prior therapies.
According to AstraZeneca, the study’s primary endpoint—durable CR—was met. The company said the phase 3 results will be presented at an upcoming medical meeting.
The US Food and Drug Administration (FDA) has accepted for priority review the biologics license application (BLA) for moxetumomab pasudotox, an investigational anti-CD22 recombinant immunotoxin.
With this BLA, AstraZeneca is seeking approval for moxetumomab pasudotox for the treatment of adults with hairy cell leukemia (HCL) who have received at least 2 prior lines of therapy.
The FDA expects to make a decision on the BLA in the third quarter of this year.
The FDA aims to take action on a priority review application within 6 months of receiving it, rather than the standard 10 months.
The agency grants priority review to applications for products that may provide significant improvements in the treatment, diagnosis, or prevention of serious conditions.
About moxetumomab pasudotox
Moxetumomab pasudotox (formerly CAT-8015 or HA22) is composed of a binding portion of an anti-CD22 antibody fused to a toxin. After binding to CD22, the molecule is internalized, processed, and releases its modified protein toxin, which inhibits protein translation and leads to apoptosis.
In addition to priority review, moxetumomab pasudotox has received orphan drug designation from the FDA.
Moxetumomab pasudotox has been tested in a phase 1 trial. Initial results from this trial were published in the Journal of Clinical Oncology in 2012. Long-term follow-up was presented at the 2017 ASH Annual Meeting.
The ASH data included 49 patients with relapsed/refractory HCL. Their median age was 57 (range, 40-77), most (n=41) were male, and they had a median of 35 (range, 1-60,444) circulating HCL cells/mm3 at baseline (in 48 evaluable patients).
Twenty-eight patients received moxetumomab pasudotox in the dose-escalation portion of the study—at 5, 10, 20, 30, 40, or 50 µg/kg—and 21 received the drug at 50 µg/kg for the extension portion of the study.
Among the 33 patients who received moxetumomab pasudotox at 50 µg/kg, the overall response rate was 88%, and the complete response (CR) rate was 64% (n=21). The median time to CR was 3.6 months, and the median duration of CR was 70.3 months.
The median follow-up was 75 months for the entire study population. At 72 months, the progression-free survival (PFS) rate was 77%.
The researchers found that minimal residual disease (MRD) negativity (via immunohistochemistry) was associated with extended response duration and prolonged PFS.
The MRD evaluation included 19 MRD+ patients and 18 MRD- patients. Forty-seven percent of the MRD+ patients (n=9) and 94% of the MRD- patients (n=17) had a CR as their best response.
The median duration of CR was 13.1 months among the MRD+ patients and was not reached among the MRD- patients (P=0.0002). The median PFS was 82.1 months among the MRD+ patients and not reached among the MRD- patients (P=0.0031).
Moxetumomab pasudotox did not undergo phase 2 testing but proceeded to a phase 3 trial. In this single-arm study, researchers evaluated the drug in HCL patients who had received at least 2 prior therapies.
According to AstraZeneca, the study’s primary endpoint—durable CR—was met. The company said the phase 3 results will be presented at an upcoming medical meeting.
The US Food and Drug Administration (FDA) has accepted for priority review the biologics license application (BLA) for moxetumomab pasudotox, an investigational anti-CD22 recombinant immunotoxin.
With this BLA, AstraZeneca is seeking approval for moxetumomab pasudotox for the treatment of adults with hairy cell leukemia (HCL) who have received at least 2 prior lines of therapy.
The FDA expects to make a decision on the BLA in the third quarter of this year.
The FDA aims to take action on a priority review application within 6 months of receiving it, rather than the standard 10 months.
The agency grants priority review to applications for products that may provide significant improvements in the treatment, diagnosis, or prevention of serious conditions.
About moxetumomab pasudotox
Moxetumomab pasudotox (formerly CAT-8015 or HA22) is composed of a binding portion of an anti-CD22 antibody fused to a toxin. After binding to CD22, the molecule is internalized, processed, and releases its modified protein toxin, which inhibits protein translation and leads to apoptosis.
In addition to priority review, moxetumomab pasudotox has received orphan drug designation from the FDA.
Moxetumomab pasudotox has been tested in a phase 1 trial. Initial results from this trial were published in the Journal of Clinical Oncology in 2012. Long-term follow-up was presented at the 2017 ASH Annual Meeting.
The ASH data included 49 patients with relapsed/refractory HCL. Their median age was 57 (range, 40-77), most (n=41) were male, and they had a median of 35 (range, 1-60,444) circulating HCL cells/mm3 at baseline (in 48 evaluable patients).
Twenty-eight patients received moxetumomab pasudotox in the dose-escalation portion of the study—at 5, 10, 20, 30, 40, or 50 µg/kg—and 21 received the drug at 50 µg/kg for the extension portion of the study.
Among the 33 patients who received moxetumomab pasudotox at 50 µg/kg, the overall response rate was 88%, and the complete response (CR) rate was 64% (n=21). The median time to CR was 3.6 months, and the median duration of CR was 70.3 months.
The median follow-up was 75 months for the entire study population. At 72 months, the progression-free survival (PFS) rate was 77%.
The researchers found that minimal residual disease (MRD) negativity (via immunohistochemistry) was associated with extended response duration and prolonged PFS.
The MRD evaluation included 19 MRD+ patients and 18 MRD- patients. Forty-seven percent of the MRD+ patients (n=9) and 94% of the MRD- patients (n=17) had a CR as their best response.
The median duration of CR was 13.1 months among the MRD+ patients and was not reached among the MRD- patients (P=0.0002). The median PFS was 82.1 months among the MRD+ patients and not reached among the MRD- patients (P=0.0031).
Moxetumomab pasudotox did not undergo phase 2 testing but proceeded to a phase 3 trial. In this single-arm study, researchers evaluated the drug in HCL patients who had received at least 2 prior therapies.
According to AstraZeneca, the study’s primary endpoint—durable CR—was met. The company said the phase 3 results will be presented at an upcoming medical meeting.
EC expands indication for denosumab to MM
The European Commission (EC) has approved an expanded indication for denosumab (Xgeva).
The drug is now approved for the prevention of skeletal-related events (SREs) in adults with advanced malignancies involving bone, which includes patients with multiple myeloma (MM).
Approval from the EC provides a centralized marketing authorization with unified labeling in all member countries of the European Union.
Norway, Iceland, and Liechtenstein will make corresponding decisions on the basis of the EC’s decision.
Denosumab was previously approved by the EC to prevent SREs—defined as radiation to bone, pathologic fracture, surgery to bone, and spinal cord compression—in adults with bone metastases from solid tumors.
Denosumab also received EC approval for the treatment of adults and skeletally mature adolescents with giant cell tumor of bone that is unresectable or where surgical resection is likely to result in severe morbidity.
The EC’s approval for denosumab in MM patients is based on data from the ’482 study, which were recently published in The Lancet Oncology.
In this phase 3 trial, denosumab proved non-inferior to zoledronic acid for delaying SREs in patients with newly diagnosed MM and bone disease.
Researchers randomized 1718 patients to receive subcutaneous denosumab at 120 mg and intravenous placebo every 4 weeks (n=859) or intravenous zoledronic acid at 4 mg (adjusted for renal function at baseline) and subcutaneous placebo every 4 weeks (n=859).
All patients also received investigators’ choice of first-line MM therapy.
The median time to first on-study SRE was 22.8 months for patients in the denosumab arm and 24 months for those in the zoledronic acid arm (hazard ratio=0.98; 95% confidence interval: 0.85-1.14; P for non-inferiority=0.010).
There were fewer renal treatment-emergent adverse events in the denosumab arm than the zoledronic acid arm—10% and 17%, respectively.
But there were more hypocalcemia adverse events in the denosumab arm than the zoledronic acid arm—17% and 12%, respectively.
The European Commission (EC) has approved an expanded indication for denosumab (Xgeva).
The drug is now approved for the prevention of skeletal-related events (SREs) in adults with advanced malignancies involving bone, which includes patients with multiple myeloma (MM).
Approval from the EC provides a centralized marketing authorization with unified labeling in all member countries of the European Union.
Norway, Iceland, and Liechtenstein will make corresponding decisions on the basis of the EC’s decision.
Denosumab was previously approved by the EC to prevent SREs—defined as radiation to bone, pathologic fracture, surgery to bone, and spinal cord compression—in adults with bone metastases from solid tumors.
Denosumab also received EC approval for the treatment of adults and skeletally mature adolescents with giant cell tumor of bone that is unresectable or where surgical resection is likely to result in severe morbidity.
The EC’s approval for denosumab in MM patients is based on data from the ’482 study, which were recently published in The Lancet Oncology.
In this phase 3 trial, denosumab proved non-inferior to zoledronic acid for delaying SREs in patients with newly diagnosed MM and bone disease.
Researchers randomized 1718 patients to receive subcutaneous denosumab at 120 mg and intravenous placebo every 4 weeks (n=859) or intravenous zoledronic acid at 4 mg (adjusted for renal function at baseline) and subcutaneous placebo every 4 weeks (n=859).
All patients also received investigators’ choice of first-line MM therapy.
The median time to first on-study SRE was 22.8 months for patients in the denosumab arm and 24 months for those in the zoledronic acid arm (hazard ratio=0.98; 95% confidence interval: 0.85-1.14; P for non-inferiority=0.010).
There were fewer renal treatment-emergent adverse events in the denosumab arm than the zoledronic acid arm—10% and 17%, respectively.
But there were more hypocalcemia adverse events in the denosumab arm than the zoledronic acid arm—17% and 12%, respectively.
The European Commission (EC) has approved an expanded indication for denosumab (Xgeva).
The drug is now approved for the prevention of skeletal-related events (SREs) in adults with advanced malignancies involving bone, which includes patients with multiple myeloma (MM).
Approval from the EC provides a centralized marketing authorization with unified labeling in all member countries of the European Union.
Norway, Iceland, and Liechtenstein will make corresponding decisions on the basis of the EC’s decision.
Denosumab was previously approved by the EC to prevent SREs—defined as radiation to bone, pathologic fracture, surgery to bone, and spinal cord compression—in adults with bone metastases from solid tumors.
Denosumab also received EC approval for the treatment of adults and skeletally mature adolescents with giant cell tumor of bone that is unresectable or where surgical resection is likely to result in severe morbidity.
The EC’s approval for denosumab in MM patients is based on data from the ’482 study, which were recently published in The Lancet Oncology.
In this phase 3 trial, denosumab proved non-inferior to zoledronic acid for delaying SREs in patients with newly diagnosed MM and bone disease.
Researchers randomized 1718 patients to receive subcutaneous denosumab at 120 mg and intravenous placebo every 4 weeks (n=859) or intravenous zoledronic acid at 4 mg (adjusted for renal function at baseline) and subcutaneous placebo every 4 weeks (n=859).
All patients also received investigators’ choice of first-line MM therapy.
The median time to first on-study SRE was 22.8 months for patients in the denosumab arm and 24 months for those in the zoledronic acid arm (hazard ratio=0.98; 95% confidence interval: 0.85-1.14; P for non-inferiority=0.010).
There were fewer renal treatment-emergent adverse events in the denosumab arm than the zoledronic acid arm—10% and 17%, respectively.
But there were more hypocalcemia adverse events in the denosumab arm than the zoledronic acid arm—17% and 12%, respectively.
Drug nets orphan designation for SCD
The European Commission (EC) has granted orphan designation to Altemia (formerly SC411) for the treatment of pediatric patients with sickle cell disease (SCD).
Altemia gelatin capsules are designed to replenish the lipids destroyed by sickle hemoglobin.
Altemia is intended to be taken once daily to reduce vaso-occlusive crises, anemia, organ damage, and other complications of SCD.
Altemia consists of a mixture of fatty acids, primarily in the form of Ethyl Cervonate (a proprietary blend of docosahexaenoic acid and other omega-3 fatty acids), and surface active agents formulated using Advanced Lipid Technologies.
Advanced Lipid Technologies are proprietary formulation and manufacturing techniques used by Sancilio Pharmaceuticals Company, Inc. (SPCI) to create lipophilic drug products.
Last November, SPCI reported topline data from its phase 2 study of Altemia, the SCOT trial (NCT02973360). The trial included pediatric patients, ages 5 to 17, with SCD.
The study’s primary endpoint was the change from baseline in blood cell membranes’ fatty acids concentration. SPCI said Altemia showed a significant improvement in this endpoint, compared to placebo, within 4 weeks of treatment initiation.
Patients who received Altemia also had significant improvements in markers of coagulation (D-dimer), inflammation (C-reactive protein), and adhesion (E-selectin) after 8 weeks of treatment.
And patients treated with Altemia had a “clinically meaningful” reduction in vaso-occlusive events, according to SPCI.
There were no treatment-related serious adverse events reported.
Ninety-four percent of patients completed the study, and most have chosen to participate in the open-label extension phase, in which researchers will continue monitoring the safety and effectiveness of Altemia.
SPCI said additional analyses of SCOT data are ongoing, and the company plans to present detailed data from the study in journals and at upcoming scientific conferences.
About orphan designation
Orphan designation provides regulatory and financial incentives for companies to develop and market therapies that treat life-threatening or chronically debilitating conditions affecting no more than 5 in 10,000 people in the European Union, and where no satisfactory treatment is available.
Orphan designation provides a 10-year period of marketing exclusivity if the drug receives regulatory approval.
The designation also provides incentives for companies seeking protocol assistance from the European Medicines Agency during the product development phase and direct access to the centralized authorization procedure.
The European Commission (EC) has granted orphan designation to Altemia (formerly SC411) for the treatment of pediatric patients with sickle cell disease (SCD).
Altemia gelatin capsules are designed to replenish the lipids destroyed by sickle hemoglobin.
Altemia is intended to be taken once daily to reduce vaso-occlusive crises, anemia, organ damage, and other complications of SCD.
Altemia consists of a mixture of fatty acids, primarily in the form of Ethyl Cervonate (a proprietary blend of docosahexaenoic acid and other omega-3 fatty acids), and surface active agents formulated using Advanced Lipid Technologies.
Advanced Lipid Technologies are proprietary formulation and manufacturing techniques used by Sancilio Pharmaceuticals Company, Inc. (SPCI) to create lipophilic drug products.
Last November, SPCI reported topline data from its phase 2 study of Altemia, the SCOT trial (NCT02973360). The trial included pediatric patients, ages 5 to 17, with SCD.
The study’s primary endpoint was the change from baseline in blood cell membranes’ fatty acids concentration. SPCI said Altemia showed a significant improvement in this endpoint, compared to placebo, within 4 weeks of treatment initiation.
Patients who received Altemia also had significant improvements in markers of coagulation (D-dimer), inflammation (C-reactive protein), and adhesion (E-selectin) after 8 weeks of treatment.
And patients treated with Altemia had a “clinically meaningful” reduction in vaso-occlusive events, according to SPCI.
There were no treatment-related serious adverse events reported.
Ninety-four percent of patients completed the study, and most have chosen to participate in the open-label extension phase, in which researchers will continue monitoring the safety and effectiveness of Altemia.
SPCI said additional analyses of SCOT data are ongoing, and the company plans to present detailed data from the study in journals and at upcoming scientific conferences.
About orphan designation
Orphan designation provides regulatory and financial incentives for companies to develop and market therapies that treat life-threatening or chronically debilitating conditions affecting no more than 5 in 10,000 people in the European Union, and where no satisfactory treatment is available.
Orphan designation provides a 10-year period of marketing exclusivity if the drug receives regulatory approval.
The designation also provides incentives for companies seeking protocol assistance from the European Medicines Agency during the product development phase and direct access to the centralized authorization procedure.
The European Commission (EC) has granted orphan designation to Altemia (formerly SC411) for the treatment of pediatric patients with sickle cell disease (SCD).
Altemia gelatin capsules are designed to replenish the lipids destroyed by sickle hemoglobin.
Altemia is intended to be taken once daily to reduce vaso-occlusive crises, anemia, organ damage, and other complications of SCD.
Altemia consists of a mixture of fatty acids, primarily in the form of Ethyl Cervonate (a proprietary blend of docosahexaenoic acid and other omega-3 fatty acids), and surface active agents formulated using Advanced Lipid Technologies.
Advanced Lipid Technologies are proprietary formulation and manufacturing techniques used by Sancilio Pharmaceuticals Company, Inc. (SPCI) to create lipophilic drug products.
Last November, SPCI reported topline data from its phase 2 study of Altemia, the SCOT trial (NCT02973360). The trial included pediatric patients, ages 5 to 17, with SCD.
The study’s primary endpoint was the change from baseline in blood cell membranes’ fatty acids concentration. SPCI said Altemia showed a significant improvement in this endpoint, compared to placebo, within 4 weeks of treatment initiation.
Patients who received Altemia also had significant improvements in markers of coagulation (D-dimer), inflammation (C-reactive protein), and adhesion (E-selectin) after 8 weeks of treatment.
And patients treated with Altemia had a “clinically meaningful” reduction in vaso-occlusive events, according to SPCI.
There were no treatment-related serious adverse events reported.
Ninety-four percent of patients completed the study, and most have chosen to participate in the open-label extension phase, in which researchers will continue monitoring the safety and effectiveness of Altemia.
SPCI said additional analyses of SCOT data are ongoing, and the company plans to present detailed data from the study in journals and at upcoming scientific conferences.
About orphan designation
Orphan designation provides regulatory and financial incentives for companies to develop and market therapies that treat life-threatening or chronically debilitating conditions affecting no more than 5 in 10,000 people in the European Union, and where no satisfactory treatment is available.
Orphan designation provides a 10-year period of marketing exclusivity if the drug receives regulatory approval.
The designation also provides incentives for companies seeking protocol assistance from the European Medicines Agency during the product development phase and direct access to the centralized authorization procedure.
FDA approves blinatumomab to treat MRD+ BCP-ALL
The US Food and Drug Administration (FDA) has expanded the approved indication for blinatumomab (Blincyto®).
The drug is now approved to treat adults and children with B-cell precursor acute lymphoblastic leukemia (BCP-ALL) in first or second complete remission (CR) with minimal residual disease (MRD) greater than or equal to 0.1%.
Blinatumomab received accelerated approval for this indication because the drug has not yet shown a clinical benefit in these patients.
The FDA’s accelerated approval program allows conditional approval of a drug that fills an unmet medical need for a serious condition.
Accelerated approval is based on surrogate or intermediate endpoints—in this case, MRD response rate and hematologic relapse-free survival (RFS)—that are reasonably likely to predict clinical benefit.
Continued approval of blinatumomab for the aforementioned indication may be contingent upon verification of clinical benefit in confirmatory trials.
“This is the first FDA-approved treatment for patients with MRD-positive ALL,” said Richard Pazdur, MD, director of the FDA’s Oncology Center of Excellence.
“Because patients who have MRD are more likely to relapse, having a treatment option that eliminates even very low amounts of residual leukemia cells may help keep the cancer in remission longer. We look forward to furthering our understanding about the reduction in MRD after treatment with Blincyto. Studies are being conducted to assess how Blincyto affects long-term survival outcomes in patients with MRD.”
About blinatumomab
Blinatumomab is a bispecific, CD19-directed, CD3 T-cell engager (BiTE®) antibody construct that binds to CD19 expressed on the surface of cells of B-lineage origin and CD3 expressed on the surface of T cells.
In 2014, the FDA granted blinatumomab accelerated approval to treat adults with Philadelphia chromosome-negative (Ph-) relapsed/refractory BCP-ALL.
In 2016, the FDA granted the therapy accelerated approval for pediatric patients with Ph- relapsed/refractory BCP-ALL.
Last year, the FDA granted blinatumomab full approval for pediatric and adult patients with Ph- or Ph+ relapsed/refractory BCP-ALL.
The FDA-approved prescribing information for blinatumomab includes a boxed warning for cytokine release syndrome and neurologic toxicities. Blinatumomab is also under a risk evaluation and mitigation strategy program in the US.
BLAST study
The new accelerated approval for blinatumomab was supported by results from the phase 2 BLAST study, which were published in Blood in January.
The study enrolled adults with MRD-positive BCP-ALL in complete hematologic remission after 3 or more cycles of intensive chemotherapy.
Patients received continuous intravenous infusions of blinatumomab at 15 μg/m2/day for 4 weeks, followed by 2 weeks off. They received up to 4 cycles of treatment and could undergo hematopoietic stem cell transplant (HSCT) at any time after the first cycle.
In all, there were 116 patients who received at least 1 infusion of blinatumomab. Seventy-six patients went on to HSCT while in continuous CR after cycle 1 (n=27), 2 (n=36), or 3/4 (n=13).
The study’s primary endpoint was the rate of complete MRD response within the first treatment cycle, and 78% of evaluable patients (88/113) achieved this endpoint.
A key secondary endpoint was RFS at 18 months. There were 110 patients evaluable for this endpoint. They all had Ph- BCP-ALL and <5% blasts at baseline.
The estimated RFS at 18 months was 54%, and the median RFS was 18.9 months. The median RFS was 24.6 months for patients treated in first CR and 11.0 months for patients treated in a later CR (P=0.004).
Another key endpoint was overall survival (OS). The median OS was 36.5 months, both for the 110 patients in the RFS analysis and for the entire study population.
In a landmark analysis, complete MRD responders had longer OS than MRD nonresponders—38.9 months and 12.5 months, respectively (P=0.002). And complete MRD responders had longer RFS than nonresponders—23.6 months and 5.7 months, respectively (P=0.002).
All 116 patients who started cycle 1 had at least 1 adverse event (AE). The rate of grade 3 AEs was 33%, and the rate of grade 4 AEs was 27%. These AEs were considered treatment-related in 29% (grade 3) and 22% (grade 4) of patients.
Four (3%) patients developed cytokine release syndrome—2 with grade 1 and 2 with grade 3. All of these events occurred during cycle 1.
Fifty-three percent of patients (n=61) had neurologic events. In most cases (97%, n=59), these events resolved.
There were 2 fatal AEs during the treatment period, both in cycle 1. One of these events—atypical pneumonitis with H1N1 influenza—was considered treatment-related. The other event—subdural hemorrhage—was considered unrelated to treatment.
There were 4 fatal AEs reported after blinatumomab treatment. Two of these deaths—due to multifocal CNS lesions and graft-versus-host disease—occurred in HSCT recipients. The other 2 deaths—due to disease progression and multi-organ failure—occurred in nontransplanted patients after relapse.
The US Food and Drug Administration (FDA) has expanded the approved indication for blinatumomab (Blincyto®).
The drug is now approved to treat adults and children with B-cell precursor acute lymphoblastic leukemia (BCP-ALL) in first or second complete remission (CR) with minimal residual disease (MRD) greater than or equal to 0.1%.
Blinatumomab received accelerated approval for this indication because the drug has not yet shown a clinical benefit in these patients.
The FDA’s accelerated approval program allows conditional approval of a drug that fills an unmet medical need for a serious condition.
Accelerated approval is based on surrogate or intermediate endpoints—in this case, MRD response rate and hematologic relapse-free survival (RFS)—that are reasonably likely to predict clinical benefit.
Continued approval of blinatumomab for the aforementioned indication may be contingent upon verification of clinical benefit in confirmatory trials.
“This is the first FDA-approved treatment for patients with MRD-positive ALL,” said Richard Pazdur, MD, director of the FDA’s Oncology Center of Excellence.
“Because patients who have MRD are more likely to relapse, having a treatment option that eliminates even very low amounts of residual leukemia cells may help keep the cancer in remission longer. We look forward to furthering our understanding about the reduction in MRD after treatment with Blincyto. Studies are being conducted to assess how Blincyto affects long-term survival outcomes in patients with MRD.”
About blinatumomab
Blinatumomab is a bispecific, CD19-directed, CD3 T-cell engager (BiTE®) antibody construct that binds to CD19 expressed on the surface of cells of B-lineage origin and CD3 expressed on the surface of T cells.
In 2014, the FDA granted blinatumomab accelerated approval to treat adults with Philadelphia chromosome-negative (Ph-) relapsed/refractory BCP-ALL.
In 2016, the FDA granted the therapy accelerated approval for pediatric patients with Ph- relapsed/refractory BCP-ALL.
Last year, the FDA granted blinatumomab full approval for pediatric and adult patients with Ph- or Ph+ relapsed/refractory BCP-ALL.
The FDA-approved prescribing information for blinatumomab includes a boxed warning for cytokine release syndrome and neurologic toxicities. Blinatumomab is also under a risk evaluation and mitigation strategy program in the US.
BLAST study
The new accelerated approval for blinatumomab was supported by results from the phase 2 BLAST study, which were published in Blood in January.
The study enrolled adults with MRD-positive BCP-ALL in complete hematologic remission after 3 or more cycles of intensive chemotherapy.
Patients received continuous intravenous infusions of blinatumomab at 15 μg/m2/day for 4 weeks, followed by 2 weeks off. They received up to 4 cycles of treatment and could undergo hematopoietic stem cell transplant (HSCT) at any time after the first cycle.
In all, there were 116 patients who received at least 1 infusion of blinatumomab. Seventy-six patients went on to HSCT while in continuous CR after cycle 1 (n=27), 2 (n=36), or 3/4 (n=13).
The study’s primary endpoint was the rate of complete MRD response within the first treatment cycle, and 78% of evaluable patients (88/113) achieved this endpoint.
A key secondary endpoint was RFS at 18 months. There were 110 patients evaluable for this endpoint. They all had Ph- BCP-ALL and <5% blasts at baseline.
The estimated RFS at 18 months was 54%, and the median RFS was 18.9 months. The median RFS was 24.6 months for patients treated in first CR and 11.0 months for patients treated in a later CR (P=0.004).
Another key endpoint was overall survival (OS). The median OS was 36.5 months, both for the 110 patients in the RFS analysis and for the entire study population.
In a landmark analysis, complete MRD responders had longer OS than MRD nonresponders—38.9 months and 12.5 months, respectively (P=0.002). And complete MRD responders had longer RFS than nonresponders—23.6 months and 5.7 months, respectively (P=0.002).
All 116 patients who started cycle 1 had at least 1 adverse event (AE). The rate of grade 3 AEs was 33%, and the rate of grade 4 AEs was 27%. These AEs were considered treatment-related in 29% (grade 3) and 22% (grade 4) of patients.
Four (3%) patients developed cytokine release syndrome—2 with grade 1 and 2 with grade 3. All of these events occurred during cycle 1.
Fifty-three percent of patients (n=61) had neurologic events. In most cases (97%, n=59), these events resolved.
There were 2 fatal AEs during the treatment period, both in cycle 1. One of these events—atypical pneumonitis with H1N1 influenza—was considered treatment-related. The other event—subdural hemorrhage—was considered unrelated to treatment.
There were 4 fatal AEs reported after blinatumomab treatment. Two of these deaths—due to multifocal CNS lesions and graft-versus-host disease—occurred in HSCT recipients. The other 2 deaths—due to disease progression and multi-organ failure—occurred in nontransplanted patients after relapse.
The US Food and Drug Administration (FDA) has expanded the approved indication for blinatumomab (Blincyto®).
The drug is now approved to treat adults and children with B-cell precursor acute lymphoblastic leukemia (BCP-ALL) in first or second complete remission (CR) with minimal residual disease (MRD) greater than or equal to 0.1%.
Blinatumomab received accelerated approval for this indication because the drug has not yet shown a clinical benefit in these patients.
The FDA’s accelerated approval program allows conditional approval of a drug that fills an unmet medical need for a serious condition.
Accelerated approval is based on surrogate or intermediate endpoints—in this case, MRD response rate and hematologic relapse-free survival (RFS)—that are reasonably likely to predict clinical benefit.
Continued approval of blinatumomab for the aforementioned indication may be contingent upon verification of clinical benefit in confirmatory trials.
“This is the first FDA-approved treatment for patients with MRD-positive ALL,” said Richard Pazdur, MD, director of the FDA’s Oncology Center of Excellence.
“Because patients who have MRD are more likely to relapse, having a treatment option that eliminates even very low amounts of residual leukemia cells may help keep the cancer in remission longer. We look forward to furthering our understanding about the reduction in MRD after treatment with Blincyto. Studies are being conducted to assess how Blincyto affects long-term survival outcomes in patients with MRD.”
About blinatumomab
Blinatumomab is a bispecific, CD19-directed, CD3 T-cell engager (BiTE®) antibody construct that binds to CD19 expressed on the surface of cells of B-lineage origin and CD3 expressed on the surface of T cells.
In 2014, the FDA granted blinatumomab accelerated approval to treat adults with Philadelphia chromosome-negative (Ph-) relapsed/refractory BCP-ALL.
In 2016, the FDA granted the therapy accelerated approval for pediatric patients with Ph- relapsed/refractory BCP-ALL.
Last year, the FDA granted blinatumomab full approval for pediatric and adult patients with Ph- or Ph+ relapsed/refractory BCP-ALL.
The FDA-approved prescribing information for blinatumomab includes a boxed warning for cytokine release syndrome and neurologic toxicities. Blinatumomab is also under a risk evaluation and mitigation strategy program in the US.
BLAST study
The new accelerated approval for blinatumomab was supported by results from the phase 2 BLAST study, which were published in Blood in January.
The study enrolled adults with MRD-positive BCP-ALL in complete hematologic remission after 3 or more cycles of intensive chemotherapy.
Patients received continuous intravenous infusions of blinatumomab at 15 μg/m2/day for 4 weeks, followed by 2 weeks off. They received up to 4 cycles of treatment and could undergo hematopoietic stem cell transplant (HSCT) at any time after the first cycle.
In all, there were 116 patients who received at least 1 infusion of blinatumomab. Seventy-six patients went on to HSCT while in continuous CR after cycle 1 (n=27), 2 (n=36), or 3/4 (n=13).
The study’s primary endpoint was the rate of complete MRD response within the first treatment cycle, and 78% of evaluable patients (88/113) achieved this endpoint.
A key secondary endpoint was RFS at 18 months. There were 110 patients evaluable for this endpoint. They all had Ph- BCP-ALL and <5% blasts at baseline.
The estimated RFS at 18 months was 54%, and the median RFS was 18.9 months. The median RFS was 24.6 months for patients treated in first CR and 11.0 months for patients treated in a later CR (P=0.004).
Another key endpoint was overall survival (OS). The median OS was 36.5 months, both for the 110 patients in the RFS analysis and for the entire study population.
In a landmark analysis, complete MRD responders had longer OS than MRD nonresponders—38.9 months and 12.5 months, respectively (P=0.002). And complete MRD responders had longer RFS than nonresponders—23.6 months and 5.7 months, respectively (P=0.002).
All 116 patients who started cycle 1 had at least 1 adverse event (AE). The rate of grade 3 AEs was 33%, and the rate of grade 4 AEs was 27%. These AEs were considered treatment-related in 29% (grade 3) and 22% (grade 4) of patients.
Four (3%) patients developed cytokine release syndrome—2 with grade 1 and 2 with grade 3. All of these events occurred during cycle 1.
Fifty-three percent of patients (n=61) had neurologic events. In most cases (97%, n=59), these events resolved.
There were 2 fatal AEs during the treatment period, both in cycle 1. One of these events—atypical pneumonitis with H1N1 influenza—was considered treatment-related. The other event—subdural hemorrhage—was considered unrelated to treatment.
There were 4 fatal AEs reported after blinatumomab treatment. Two of these deaths—due to multifocal CNS lesions and graft-versus-host disease—occurred in HSCT recipients. The other 2 deaths—due to disease progression and multi-organ failure—occurred in nontransplanted patients after relapse.
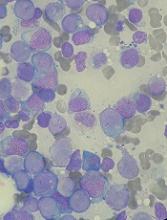
Drug receives orphan designation for AML
The US Food and Drug Administration (FDA) has granted orphan designation to MAX-40279 for the treatment of acute myeloid leukemia (AML).
MAX-40279 is a multi-target kinase inhibitor being developed by MaxiNovel Pharmaceuticals, Inc.
The drug mainly targets FMS-related tyrosine kinase 3 (FLT3) and fibroblast growth factor receptor (FGFR).
MAX-40279 demonstrated “potent” inhibition of FLT3 and FGFR in preclinical testing, according to MaxiNovel Pharmaceuticals, Inc.
The company is currently testing MAX-40279 in a phase 1 trial of patients with AML (NCT03412292).
About orphan designation
The FDA grants orphan designation to products intended to treat, diagnose, or prevent diseases/disorders that affect fewer than 200,000 people in the US.
The designation provides incentives for sponsors to develop products for rare diseases. This may include tax credits toward the cost of clinical trials, prescription drug user fee waivers, and 7 years of market exclusivity if the product is approved.
The US Food and Drug Administration (FDA) has granted orphan designation to MAX-40279 for the treatment of acute myeloid leukemia (AML).
MAX-40279 is a multi-target kinase inhibitor being developed by MaxiNovel Pharmaceuticals, Inc.
The drug mainly targets FMS-related tyrosine kinase 3 (FLT3) and fibroblast growth factor receptor (FGFR).
MAX-40279 demonstrated “potent” inhibition of FLT3 and FGFR in preclinical testing, according to MaxiNovel Pharmaceuticals, Inc.
The company is currently testing MAX-40279 in a phase 1 trial of patients with AML (NCT03412292).
About orphan designation
The FDA grants orphan designation to products intended to treat, diagnose, or prevent diseases/disorders that affect fewer than 200,000 people in the US.
The designation provides incentives for sponsors to develop products for rare diseases. This may include tax credits toward the cost of clinical trials, prescription drug user fee waivers, and 7 years of market exclusivity if the product is approved.
The US Food and Drug Administration (FDA) has granted orphan designation to MAX-40279 for the treatment of acute myeloid leukemia (AML).
MAX-40279 is a multi-target kinase inhibitor being developed by MaxiNovel Pharmaceuticals, Inc.
The drug mainly targets FMS-related tyrosine kinase 3 (FLT3) and fibroblast growth factor receptor (FGFR).
MAX-40279 demonstrated “potent” inhibition of FLT3 and FGFR in preclinical testing, according to MaxiNovel Pharmaceuticals, Inc.
The company is currently testing MAX-40279 in a phase 1 trial of patients with AML (NCT03412292).
About orphan designation
The FDA grants orphan designation to products intended to treat, diagnose, or prevent diseases/disorders that affect fewer than 200,000 people in the US.
The designation provides incentives for sponsors to develop products for rare diseases. This may include tax credits toward the cost of clinical trials, prescription drug user fee waivers, and 7 years of market exclusivity if the product is approved.
Deaths in patients on emicizumab
Two hemophilia organizations have notified the public of 5 deaths in adult patients receiving emicizumab (Hemlibra).
All 5 deaths—occurring in 2016 (n=1), 2017 (n=2), and this year (n=2)—were deemed unrelated to emicizumab by the investigator or treating physician.
The National Hemophilia Foundation and Hemophilia Federation of America reported these deaths after receiving notifications from Genentech.
The company said it has limited information about the circumstances of the deaths.
However, Genentech did say the 2016 death, 1 of the 2017 deaths, and 1 of the 2018 deaths occurred in patients receiving emicizumab via a compassionate use program.
Compassionate use of emicizumab has been available on a case-by-case basis, following a request to Roche from a patient’s treating physician, if the patient has a serious or life-threatening condition, has exhausted all other treatment options, and is unable to participate in a clinical trial.
The other death in 2017 occurred in a patient on the phase 3 HAVEN 1 trial and was reported along with the other results from that trial.
The remaining death in 2018 was in a patient receiving emicizumab via an expanded access protocol.
This protocol, which was reviewed by the US Food and Drug Administration, allowed US patients who were not participating in a clinical trial of emicizumab but who met eligibility criteria similar to key studies to have access to emicizumab prior to approval, which occurred in November 2017.
In response to the deaths, Genentech has pledged to share information on any adverse events that impact the overall benefit/risk profile of emicizumab.
“We are committed to providing timely and transparent updates on the safety profile of Hemlibra to health authorities, healthcare professionals, and the hemophilia community,” the company said.
For more information, patients and healthcare providers can call Genentech’s medical communications line at 1-800-821-8590.
Two hemophilia organizations have notified the public of 5 deaths in adult patients receiving emicizumab (Hemlibra).
All 5 deaths—occurring in 2016 (n=1), 2017 (n=2), and this year (n=2)—were deemed unrelated to emicizumab by the investigator or treating physician.
The National Hemophilia Foundation and Hemophilia Federation of America reported these deaths after receiving notifications from Genentech.
The company said it has limited information about the circumstances of the deaths.
However, Genentech did say the 2016 death, 1 of the 2017 deaths, and 1 of the 2018 deaths occurred in patients receiving emicizumab via a compassionate use program.
Compassionate use of emicizumab has been available on a case-by-case basis, following a request to Roche from a patient’s treating physician, if the patient has a serious or life-threatening condition, has exhausted all other treatment options, and is unable to participate in a clinical trial.
The other death in 2017 occurred in a patient on the phase 3 HAVEN 1 trial and was reported along with the other results from that trial.
The remaining death in 2018 was in a patient receiving emicizumab via an expanded access protocol.
This protocol, which was reviewed by the US Food and Drug Administration, allowed US patients who were not participating in a clinical trial of emicizumab but who met eligibility criteria similar to key studies to have access to emicizumab prior to approval, which occurred in November 2017.
In response to the deaths, Genentech has pledged to share information on any adverse events that impact the overall benefit/risk profile of emicizumab.
“We are committed to providing timely and transparent updates on the safety profile of Hemlibra to health authorities, healthcare professionals, and the hemophilia community,” the company said.
For more information, patients and healthcare providers can call Genentech’s medical communications line at 1-800-821-8590.
Two hemophilia organizations have notified the public of 5 deaths in adult patients receiving emicizumab (Hemlibra).
All 5 deaths—occurring in 2016 (n=1), 2017 (n=2), and this year (n=2)—were deemed unrelated to emicizumab by the investigator or treating physician.
The National Hemophilia Foundation and Hemophilia Federation of America reported these deaths after receiving notifications from Genentech.
The company said it has limited information about the circumstances of the deaths.
However, Genentech did say the 2016 death, 1 of the 2017 deaths, and 1 of the 2018 deaths occurred in patients receiving emicizumab via a compassionate use program.
Compassionate use of emicizumab has been available on a case-by-case basis, following a request to Roche from a patient’s treating physician, if the patient has a serious or life-threatening condition, has exhausted all other treatment options, and is unable to participate in a clinical trial.
The other death in 2017 occurred in a patient on the phase 3 HAVEN 1 trial and was reported along with the other results from that trial.
The remaining death in 2018 was in a patient receiving emicizumab via an expanded access protocol.
This protocol, which was reviewed by the US Food and Drug Administration, allowed US patients who were not participating in a clinical trial of emicizumab but who met eligibility criteria similar to key studies to have access to emicizumab prior to approval, which occurred in November 2017.
In response to the deaths, Genentech has pledged to share information on any adverse events that impact the overall benefit/risk profile of emicizumab.
“We are committed to providing timely and transparent updates on the safety profile of Hemlibra to health authorities, healthcare professionals, and the hemophilia community,” the company said.
For more information, patients and healthcare providers can call Genentech’s medical communications line at 1-800-821-8590.
CHMP recommends approval for generic prasugrel
The European Medicines Agency’s Committee for Medicinal Products for Human Use (CHMP) has recommended granting marketing authorization to a generic version of prasugrel called Prasugrel Mylan.
Mylan S.A.S. is seeking approval for this product to be co-administered with acetylsalicylic acid for the prevention of atherothrombotic events in adults with acute coronary syndrome (ie, unstable angina, non-ST segment elevation myocardial infarction, or ST segment elevation myocardial infarction) undergoing primary or delayed percutaneous coronary intervention.
The CHMP’s opinion on Prasugrel Mylan will be reviewed by the European Commission (EC).
If the EC agrees with the CHMP, the commission will grant a centralized marketing authorization that will be valid in the European Union. Norway, Iceland, and Liechtenstein will make corresponding decisions on the basis of the EC’s decision.
The EC typically makes a decision within 67 days of the CHMP’s recommendation.
If approved, Prasugrel Mylan will be available as 5 mg and 10 mg film-coated tablets.
Prasugrel is an inhibitor of platelet activation and aggregation that acts through the irreversible binding of its active metabolite to the P2Y12 class of ADP receptors on platelets.
Since platelets participate in the initiation and/or evolution of thrombotic complications of atherosclerotic disease, inhibition of platelet function can reduce the risk of cardiovascular events such as death, myocardial infarction, or stroke.
Prasugrel Mylan is a generic of Efient, which has been authorized in the European Union since 2009.
According to the CHMP, studies have demonstrated that Prasugrel Mylan is of “satisfactory quality” and bioequivalent to Efient.
In the TRITON–TIMI 38 study, treatment with prasugrel (Efient) was associated with significantly reduced rates of ischemic events, including stent thrombosis, when compared to treatment with clopidogrel in patients with moderate- to high-risk acute coronary syndromes with scheduled percutaneous coronary intervention.
However, prasugrel was also associated with an increased risk of major bleeding, including fatal bleeding. Still, there was no significant difference in mortality between the treatment groups.
These results were published in NEJM in 2007.
The European Medicines Agency’s Committee for Medicinal Products for Human Use (CHMP) has recommended granting marketing authorization to a generic version of prasugrel called Prasugrel Mylan.
Mylan S.A.S. is seeking approval for this product to be co-administered with acetylsalicylic acid for the prevention of atherothrombotic events in adults with acute coronary syndrome (ie, unstable angina, non-ST segment elevation myocardial infarction, or ST segment elevation myocardial infarction) undergoing primary or delayed percutaneous coronary intervention.
The CHMP’s opinion on Prasugrel Mylan will be reviewed by the European Commission (EC).
If the EC agrees with the CHMP, the commission will grant a centralized marketing authorization that will be valid in the European Union. Norway, Iceland, and Liechtenstein will make corresponding decisions on the basis of the EC’s decision.
The EC typically makes a decision within 67 days of the CHMP’s recommendation.
If approved, Prasugrel Mylan will be available as 5 mg and 10 mg film-coated tablets.
Prasugrel is an inhibitor of platelet activation and aggregation that acts through the irreversible binding of its active metabolite to the P2Y12 class of ADP receptors on platelets.
Since platelets participate in the initiation and/or evolution of thrombotic complications of atherosclerotic disease, inhibition of platelet function can reduce the risk of cardiovascular events such as death, myocardial infarction, or stroke.
Prasugrel Mylan is a generic of Efient, which has been authorized in the European Union since 2009.
According to the CHMP, studies have demonstrated that Prasugrel Mylan is of “satisfactory quality” and bioequivalent to Efient.
In the TRITON–TIMI 38 study, treatment with prasugrel (Efient) was associated with significantly reduced rates of ischemic events, including stent thrombosis, when compared to treatment with clopidogrel in patients with moderate- to high-risk acute coronary syndromes with scheduled percutaneous coronary intervention.
However, prasugrel was also associated with an increased risk of major bleeding, including fatal bleeding. Still, there was no significant difference in mortality between the treatment groups.
These results were published in NEJM in 2007.
The European Medicines Agency’s Committee for Medicinal Products for Human Use (CHMP) has recommended granting marketing authorization to a generic version of prasugrel called Prasugrel Mylan.
Mylan S.A.S. is seeking approval for this product to be co-administered with acetylsalicylic acid for the prevention of atherothrombotic events in adults with acute coronary syndrome (ie, unstable angina, non-ST segment elevation myocardial infarction, or ST segment elevation myocardial infarction) undergoing primary or delayed percutaneous coronary intervention.
The CHMP’s opinion on Prasugrel Mylan will be reviewed by the European Commission (EC).
If the EC agrees with the CHMP, the commission will grant a centralized marketing authorization that will be valid in the European Union. Norway, Iceland, and Liechtenstein will make corresponding decisions on the basis of the EC’s decision.
The EC typically makes a decision within 67 days of the CHMP’s recommendation.
If approved, Prasugrel Mylan will be available as 5 mg and 10 mg film-coated tablets.
Prasugrel is an inhibitor of platelet activation and aggregation that acts through the irreversible binding of its active metabolite to the P2Y12 class of ADP receptors on platelets.
Since platelets participate in the initiation and/or evolution of thrombotic complications of atherosclerotic disease, inhibition of platelet function can reduce the risk of cardiovascular events such as death, myocardial infarction, or stroke.
Prasugrel Mylan is a generic of Efient, which has been authorized in the European Union since 2009.
According to the CHMP, studies have demonstrated that Prasugrel Mylan is of “satisfactory quality” and bioequivalent to Efient.
In the TRITON–TIMI 38 study, treatment with prasugrel (Efient) was associated with significantly reduced rates of ischemic events, including stent thrombosis, when compared to treatment with clopidogrel in patients with moderate- to high-risk acute coronary syndromes with scheduled percutaneous coronary intervention.
However, prasugrel was also associated with an increased risk of major bleeding, including fatal bleeding. Still, there was no significant difference in mortality between the treatment groups.
These results were published in NEJM in 2007.
Product approved for hemophilia patients in Japan
Japan’s Ministry of Health, Labour and Welfare (MHLW) has approved the bispecific factor IXa- and factor X-directed antibody emicizumab (Hemlibra), according to Chugai Pharmaceutical Co., Ltd.
Emicizumab is now approved for use in Japan as routine prophylaxis to prevent or reduce the frequency of bleeding episodes in patients with hemophilia A and factor VIII inhibitors.
There are a few conditions for this approval, including requirements for a risk management plan, early phase post-marketing vigilance, and post-marketing drug use surveillance.
A risk management plan is intended to assess measures for appropriate management of the risks associated with a drug, either at regular intervals or in response to the progress of post-marketing surveillance.
According to Japan’s Pharmaceuticals and Medical Devices Agency, a risk management plan must consist of 3 elements:
- Safety specification—Important adverse drug reactions and missing information
- Pharmacovigilance activities—Information collection activities performed in the post-marketing period
- Risk-minimization activities—Safety measures taken to minimize risks, which consists of providing information to healthcare professionals and setting the terms of use for a drug.
For the early phase post-marketing vigilance requirement, Chugai must provide safety information to healthcare professionals and collect information on adverse reactions to emicizumab in the early post-marketing phase.
Post-marketing drug use surveillance for emicizumab will include all patients receiving the product and is scheduled to continue until data is collected from approximately 100 people. The goal is to understand background information on patients receiving emicizumab, as well as to collect safety and efficacy data on the product and take necessary measures for the appropriate use of emicizumab.
The data collected via this surveillance effort will be reviewed to determine whether new surveillance or further safety measures are needed. Results of the surveillance will be reported to the regulatory authorities, and the data will be presented at scientific meetings, according to Chugai.
Phase 3 studies
The MHLW’s approval of emicizumab is based on data from a pair of phase 3 studies—HAVEN 1 and HAVEN 2.
Results from HAVEN 1 were published in NEJM and presented at the 26th ISTH Congress in July 2017. Updated results from HAVEN 2 were presented at the 2017 ASH Annual Meeting in December.
HAVEN 1
This study enrolled 109 patients (age 12 and older) with hemophilia A and factor VIII inhibitors who were previously treated with bypassing agents (BPAs) on-demand or as prophylaxis.
The patients were randomized to receive emicizumab prophylaxis or no prophylaxis. On-demand treatment of breakthrough bleeds with BPAs was allowed.
There was a significant reduction in treated bleeds of 87% with emicizumab prophylaxis compared to no prophylaxis (95% CI: 72.3; 94.3, P<0.0001). And there was an 80% reduction in all bleeds with emicizumab (95% CI: 62.5; 89.8, P<0.0001).
Adverse events (AEs) occurring in at least 5% of patients treated with emicizumab were local injection site reactions, headache, fatigue, upper respiratory tract infection, and arthralgia.
Two patients experienced thromboembolic events (TEs). Three had thrombotic microangiopathy (TMA) while receiving emicizumab prophylaxis and more than 100 u/kg/day of activated prothrombin complex concentrate, on average, for 24 hours or more before the event. Two of these patients had also received recombinant factor VIIa.
Neither TE required anticoagulation therapy, and 1 patient restarted emicizumab. The cases of TMA observed were transient, and 1 patient restarted emicizumab.
HAVEN 2
In this single-arm trial, researchers evaluated emicizumab prophylaxis in 60 patients, ages 1 to 17, who had hemophilia A with factor VIII inhibitors.
The efficacy analysis included 57 patients who were younger than 12. The 3 older patients were only included in the safety analysis.
Of the 57 patients, 64.9% had 0 bleeds, 94.7% had 0 treated bleeds, and 98.2% had 0 treated spontaneous bleeds and 0 treated joint bleeds. None of the patients had treated target joint bleeds.
Forty patients had a total of 201 AEs. The most common of these were viral upper respiratory tract infections (16.7%) and injection site reactions (16.7%).
There were no TEs or TMA events, and none of the patients tested positive for anti-drug antibodies. None of the 7 serious AEs in this trial were considered treatment-related.
Japan’s Ministry of Health, Labour and Welfare (MHLW) has approved the bispecific factor IXa- and factor X-directed antibody emicizumab (Hemlibra), according to Chugai Pharmaceutical Co., Ltd.
Emicizumab is now approved for use in Japan as routine prophylaxis to prevent or reduce the frequency of bleeding episodes in patients with hemophilia A and factor VIII inhibitors.
There are a few conditions for this approval, including requirements for a risk management plan, early phase post-marketing vigilance, and post-marketing drug use surveillance.
A risk management plan is intended to assess measures for appropriate management of the risks associated with a drug, either at regular intervals or in response to the progress of post-marketing surveillance.
According to Japan’s Pharmaceuticals and Medical Devices Agency, a risk management plan must consist of 3 elements:
- Safety specification—Important adverse drug reactions and missing information
- Pharmacovigilance activities—Information collection activities performed in the post-marketing period
- Risk-minimization activities—Safety measures taken to minimize risks, which consists of providing information to healthcare professionals and setting the terms of use for a drug.
For the early phase post-marketing vigilance requirement, Chugai must provide safety information to healthcare professionals and collect information on adverse reactions to emicizumab in the early post-marketing phase.
Post-marketing drug use surveillance for emicizumab will include all patients receiving the product and is scheduled to continue until data is collected from approximately 100 people. The goal is to understand background information on patients receiving emicizumab, as well as to collect safety and efficacy data on the product and take necessary measures for the appropriate use of emicizumab.
The data collected via this surveillance effort will be reviewed to determine whether new surveillance or further safety measures are needed. Results of the surveillance will be reported to the regulatory authorities, and the data will be presented at scientific meetings, according to Chugai.
Phase 3 studies
The MHLW’s approval of emicizumab is based on data from a pair of phase 3 studies—HAVEN 1 and HAVEN 2.
Results from HAVEN 1 were published in NEJM and presented at the 26th ISTH Congress in July 2017. Updated results from HAVEN 2 were presented at the 2017 ASH Annual Meeting in December.
HAVEN 1
This study enrolled 109 patients (age 12 and older) with hemophilia A and factor VIII inhibitors who were previously treated with bypassing agents (BPAs) on-demand or as prophylaxis.
The patients were randomized to receive emicizumab prophylaxis or no prophylaxis. On-demand treatment of breakthrough bleeds with BPAs was allowed.
There was a significant reduction in treated bleeds of 87% with emicizumab prophylaxis compared to no prophylaxis (95% CI: 72.3; 94.3, P<0.0001). And there was an 80% reduction in all bleeds with emicizumab (95% CI: 62.5; 89.8, P<0.0001).
Adverse events (AEs) occurring in at least 5% of patients treated with emicizumab were local injection site reactions, headache, fatigue, upper respiratory tract infection, and arthralgia.
Two patients experienced thromboembolic events (TEs). Three had thrombotic microangiopathy (TMA) while receiving emicizumab prophylaxis and more than 100 u/kg/day of activated prothrombin complex concentrate, on average, for 24 hours or more before the event. Two of these patients had also received recombinant factor VIIa.
Neither TE required anticoagulation therapy, and 1 patient restarted emicizumab. The cases of TMA observed were transient, and 1 patient restarted emicizumab.
HAVEN 2
In this single-arm trial, researchers evaluated emicizumab prophylaxis in 60 patients, ages 1 to 17, who had hemophilia A with factor VIII inhibitors.
The efficacy analysis included 57 patients who were younger than 12. The 3 older patients were only included in the safety analysis.
Of the 57 patients, 64.9% had 0 bleeds, 94.7% had 0 treated bleeds, and 98.2% had 0 treated spontaneous bleeds and 0 treated joint bleeds. None of the patients had treated target joint bleeds.
Forty patients had a total of 201 AEs. The most common of these were viral upper respiratory tract infections (16.7%) and injection site reactions (16.7%).
There were no TEs or TMA events, and none of the patients tested positive for anti-drug antibodies. None of the 7 serious AEs in this trial were considered treatment-related.
Japan’s Ministry of Health, Labour and Welfare (MHLW) has approved the bispecific factor IXa- and factor X-directed antibody emicizumab (Hemlibra), according to Chugai Pharmaceutical Co., Ltd.
Emicizumab is now approved for use in Japan as routine prophylaxis to prevent or reduce the frequency of bleeding episodes in patients with hemophilia A and factor VIII inhibitors.
There are a few conditions for this approval, including requirements for a risk management plan, early phase post-marketing vigilance, and post-marketing drug use surveillance.
A risk management plan is intended to assess measures for appropriate management of the risks associated with a drug, either at regular intervals or in response to the progress of post-marketing surveillance.
According to Japan’s Pharmaceuticals and Medical Devices Agency, a risk management plan must consist of 3 elements:
- Safety specification—Important adverse drug reactions and missing information
- Pharmacovigilance activities—Information collection activities performed in the post-marketing period
- Risk-minimization activities—Safety measures taken to minimize risks, which consists of providing information to healthcare professionals and setting the terms of use for a drug.
For the early phase post-marketing vigilance requirement, Chugai must provide safety information to healthcare professionals and collect information on adverse reactions to emicizumab in the early post-marketing phase.
Post-marketing drug use surveillance for emicizumab will include all patients receiving the product and is scheduled to continue until data is collected from approximately 100 people. The goal is to understand background information on patients receiving emicizumab, as well as to collect safety and efficacy data on the product and take necessary measures for the appropriate use of emicizumab.
The data collected via this surveillance effort will be reviewed to determine whether new surveillance or further safety measures are needed. Results of the surveillance will be reported to the regulatory authorities, and the data will be presented at scientific meetings, according to Chugai.
Phase 3 studies
The MHLW’s approval of emicizumab is based on data from a pair of phase 3 studies—HAVEN 1 and HAVEN 2.
Results from HAVEN 1 were published in NEJM and presented at the 26th ISTH Congress in July 2017. Updated results from HAVEN 2 were presented at the 2017 ASH Annual Meeting in December.
HAVEN 1
This study enrolled 109 patients (age 12 and older) with hemophilia A and factor VIII inhibitors who were previously treated with bypassing agents (BPAs) on-demand or as prophylaxis.
The patients were randomized to receive emicizumab prophylaxis or no prophylaxis. On-demand treatment of breakthrough bleeds with BPAs was allowed.
There was a significant reduction in treated bleeds of 87% with emicizumab prophylaxis compared to no prophylaxis (95% CI: 72.3; 94.3, P<0.0001). And there was an 80% reduction in all bleeds with emicizumab (95% CI: 62.5; 89.8, P<0.0001).
Adverse events (AEs) occurring in at least 5% of patients treated with emicizumab were local injection site reactions, headache, fatigue, upper respiratory tract infection, and arthralgia.
Two patients experienced thromboembolic events (TEs). Three had thrombotic microangiopathy (TMA) while receiving emicizumab prophylaxis and more than 100 u/kg/day of activated prothrombin complex concentrate, on average, for 24 hours or more before the event. Two of these patients had also received recombinant factor VIIa.
Neither TE required anticoagulation therapy, and 1 patient restarted emicizumab. The cases of TMA observed were transient, and 1 patient restarted emicizumab.
HAVEN 2
In this single-arm trial, researchers evaluated emicizumab prophylaxis in 60 patients, ages 1 to 17, who had hemophilia A with factor VIII inhibitors.
The efficacy analysis included 57 patients who were younger than 12. The 3 older patients were only included in the safety analysis.
Of the 57 patients, 64.9% had 0 bleeds, 94.7% had 0 treated bleeds, and 98.2% had 0 treated spontaneous bleeds and 0 treated joint bleeds. None of the patients had treated target joint bleeds.
Forty patients had a total of 201 AEs. The most common of these were viral upper respiratory tract infections (16.7%) and injection site reactions (16.7%).
There were no TEs or TMA events, and none of the patients tested positive for anti-drug antibodies. None of the 7 serious AEs in this trial were considered treatment-related.
FDA approves nilotinib for kids with CML
The US Food and Drug Administration (FDA) has expanded the approved indication for nilotinib (Tasigna®) to include the treatment of children.
The drug is now approved to treat patients age 1 and older who have newly diagnosed Philadelphia chromosome-positive (Ph+) chronic myeloid leukemia (CML) in the chronic phase.
Nilotinib is also approved to treat pediatric patients age 1 and older who have chronic phase, Ph+ CML that is resistant or intolerant to prior tyrosine kinase inhibitor (TKI) therapy, as well as adults with Ph+ CML in chronic phase and accelerated phase that is resistant or intolerant to prior therapy including imatinib.
The new pediatric indications for nilotinib, granted under the FDA’s priority review designation, are based on results from 2 studies of the drug—a phase 1 and phase 2.
According to Novartis, the studies included 69 CML patients who ranged in age from 2 to 17. They had either newly diagnosed, chronic phase, Ph+ CML or chronic phase, Ph+ CML with resistance or intolerance to prior TKI therapy.
In the newly diagnosed patients, the major molecular response (MMR) rate was 60.0% at 12 cycles, with 15 patients achieving MMR.
In patients with resistance or intolerance to prior therapy, the MMR rate was 40.9% at 12 cycles, with 18 patients being in MMR.
In newly diagnosed patients, the cumulative MMR rate was 64.0% by cycle 12. In patients with resistance or intolerance to prior therapy, the cumulative MMR rate was 47.7% by cycle 12.
Adverse events were generally consistent with those observed in adults, with the exception of hyperbilirubinemia and transaminase elevation, which were reported at a higher frequency than in adults.
The rate of grade 3/4 hyperbilirubinemia was 13.0%, the rate of grade 3/4 AST elevation was 1.4%, and the rate of grade 3/4 ALT elevation was 8.7%.
There were no deaths on treatment or after treatment discontinuation.
There was 1 patient with resistant/intolerant CML who progressed to advance phase/blast crisis after about 10 months on nilotinib.
The US Food and Drug Administration (FDA) has expanded the approved indication for nilotinib (Tasigna®) to include the treatment of children.
The drug is now approved to treat patients age 1 and older who have newly diagnosed Philadelphia chromosome-positive (Ph+) chronic myeloid leukemia (CML) in the chronic phase.
Nilotinib is also approved to treat pediatric patients age 1 and older who have chronic phase, Ph+ CML that is resistant or intolerant to prior tyrosine kinase inhibitor (TKI) therapy, as well as adults with Ph+ CML in chronic phase and accelerated phase that is resistant or intolerant to prior therapy including imatinib.
The new pediatric indications for nilotinib, granted under the FDA’s priority review designation, are based on results from 2 studies of the drug—a phase 1 and phase 2.
According to Novartis, the studies included 69 CML patients who ranged in age from 2 to 17. They had either newly diagnosed, chronic phase, Ph+ CML or chronic phase, Ph+ CML with resistance or intolerance to prior TKI therapy.
In the newly diagnosed patients, the major molecular response (MMR) rate was 60.0% at 12 cycles, with 15 patients achieving MMR.
In patients with resistance or intolerance to prior therapy, the MMR rate was 40.9% at 12 cycles, with 18 patients being in MMR.
In newly diagnosed patients, the cumulative MMR rate was 64.0% by cycle 12. In patients with resistance or intolerance to prior therapy, the cumulative MMR rate was 47.7% by cycle 12.
Adverse events were generally consistent with those observed in adults, with the exception of hyperbilirubinemia and transaminase elevation, which were reported at a higher frequency than in adults.
The rate of grade 3/4 hyperbilirubinemia was 13.0%, the rate of grade 3/4 AST elevation was 1.4%, and the rate of grade 3/4 ALT elevation was 8.7%.
There were no deaths on treatment or after treatment discontinuation.
There was 1 patient with resistant/intolerant CML who progressed to advance phase/blast crisis after about 10 months on nilotinib.
The US Food and Drug Administration (FDA) has expanded the approved indication for nilotinib (Tasigna®) to include the treatment of children.
The drug is now approved to treat patients age 1 and older who have newly diagnosed Philadelphia chromosome-positive (Ph+) chronic myeloid leukemia (CML) in the chronic phase.
Nilotinib is also approved to treat pediatric patients age 1 and older who have chronic phase, Ph+ CML that is resistant or intolerant to prior tyrosine kinase inhibitor (TKI) therapy, as well as adults with Ph+ CML in chronic phase and accelerated phase that is resistant or intolerant to prior therapy including imatinib.
The new pediatric indications for nilotinib, granted under the FDA’s priority review designation, are based on results from 2 studies of the drug—a phase 1 and phase 2.
According to Novartis, the studies included 69 CML patients who ranged in age from 2 to 17. They had either newly diagnosed, chronic phase, Ph+ CML or chronic phase, Ph+ CML with resistance or intolerance to prior TKI therapy.
In the newly diagnosed patients, the major molecular response (MMR) rate was 60.0% at 12 cycles, with 15 patients achieving MMR.
In patients with resistance or intolerance to prior therapy, the MMR rate was 40.9% at 12 cycles, with 18 patients being in MMR.
In newly diagnosed patients, the cumulative MMR rate was 64.0% by cycle 12. In patients with resistance or intolerance to prior therapy, the cumulative MMR rate was 47.7% by cycle 12.
Adverse events were generally consistent with those observed in adults, with the exception of hyperbilirubinemia and transaminase elevation, which were reported at a higher frequency than in adults.
The rate of grade 3/4 hyperbilirubinemia was 13.0%, the rate of grade 3/4 AST elevation was 1.4%, and the rate of grade 3/4 ALT elevation was 8.7%.
There were no deaths on treatment or after treatment discontinuation.
There was 1 patient with resistant/intolerant CML who progressed to advance phase/blast crisis after about 10 months on nilotinib.