User login
Drug receives priority review for FL
The US Food and Drug Administration (FDA) has granted priority review to a supplemental biologics license application (sBLA) for obinutuzumab (Gazyva®).
With this sBLA, Genentech is seeking approval for obinutuzumab to be used, first in combination with chemotherapy and then alone as maintenance, in patients with previously untreated follicular lymphoma (FL).
The FDA grants priority review to applications for products that may provide significant improvements in the treatment, diagnosis, or prevention of serious conditions.
The agency’s goal is to take action on a priority review application within 6 months of receiving it, rather than the standard 10 months.
The FDA plans to make a decision on the sBLA for obinutuzumab by December 23, 2017.
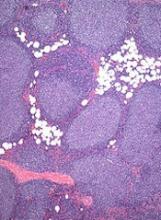
The sBLA is supported by results of the GALLIUM study, which were presented at the 2016 ASH Annual Meeting.
About obinutuzumab
Obinutuzumab is a glycoengineered, humanized, monoclonal antibody that selectively binds to the extracellular domain of the CD20 antigen on B cells.
The drug is FDA-approved for use in combination with chlorambucil to treat patients with previously untreated chronic lymphocytic leukemia.
Obinutuzumab is also approved to treat FL patients who relapse after, or are refractory to, a rituximab-containing regimen. In these patients, obinutuzumab is given first in combination with bendamustine and then alone as maintenance.
The full prescribing information for obinutuzumab is available at http://www.Gazyva.com.
About the GALLIUM study
GALLIUM enrolled 1401 patients with previously untreated, indolent non-Hodgkin lymphoma, including 1202 with FL.
Half of the FL patients (n=601) were randomized to receive obinutuzumab plus chemotherapy (followed by obinutuzumab maintenance for up to 2 years), and half were randomized to rituximab plus chemotherapy (followed by rituximab maintenance for up to 2 years).
The different chemotherapies used were CHOP (cyclophosphamide, doxorubicin, vincristine, and prednisolone), CVP (cyclophosphamide, vincristine, and prednisolone), and bendamustine.
Patients who received obinutuzumab had significantly better progression-free survival than patients who received rituximab. The 3-year progression-free survival rate was 73.3% in the rituximab arm and 80% in the obinutuzumab arm (hazard ratio [HR]=0.66, P=0.0012).
There was no significant difference between the treatment arms with regard to overall survival. The 3-year overall survival was 92.1% in the rituximab arm and 94% in the obinutuzumab arm (HR=0.75, P=0.21).
The overall incidence of adverse events (AEs) was 98.3% in the rituximab arm and 99.5% in the obinutuzumab arm. The incidence of serious AEs was 39.9% and 46.1%, respectively.
The incidence of grade 3 or higher AEs was higher among patients who received obinutuzumab.
Grade 3 or higher AEs occurring in at least 5% of patients in either arm (rituximab and obinutuzumab, respectively) included neutropenia (67.8% and 74.6%), leukopenia (37.9% and 43.9%), febrile neutropenia (4.9% and 6.9%), infections and infestations (3.7% and 6.7%), and thrombocytopenia (2.7% and 6.1%). ![]()
The US Food and Drug Administration (FDA) has granted priority review to a supplemental biologics license application (sBLA) for obinutuzumab (Gazyva®).
With this sBLA, Genentech is seeking approval for obinutuzumab to be used, first in combination with chemotherapy and then alone as maintenance, in patients with previously untreated follicular lymphoma (FL).
The FDA grants priority review to applications for products that may provide significant improvements in the treatment, diagnosis, or prevention of serious conditions.
The agency’s goal is to take action on a priority review application within 6 months of receiving it, rather than the standard 10 months.
The FDA plans to make a decision on the sBLA for obinutuzumab by December 23, 2017.
The sBLA is supported by results of the GALLIUM study, which were presented at the 2016 ASH Annual Meeting.
About obinutuzumab
Obinutuzumab is a glycoengineered, humanized, monoclonal antibody that selectively binds to the extracellular domain of the CD20 antigen on B cells.
The drug is FDA-approved for use in combination with chlorambucil to treat patients with previously untreated chronic lymphocytic leukemia.
Obinutuzumab is also approved to treat FL patients who relapse after, or are refractory to, a rituximab-containing regimen. In these patients, obinutuzumab is given first in combination with bendamustine and then alone as maintenance.
The full prescribing information for obinutuzumab is available at http://www.Gazyva.com.
About the GALLIUM study
GALLIUM enrolled 1401 patients with previously untreated, indolent non-Hodgkin lymphoma, including 1202 with FL.
Half of the FL patients (n=601) were randomized to receive obinutuzumab plus chemotherapy (followed by obinutuzumab maintenance for up to 2 years), and half were randomized to rituximab plus chemotherapy (followed by rituximab maintenance for up to 2 years).
The different chemotherapies used were CHOP (cyclophosphamide, doxorubicin, vincristine, and prednisolone), CVP (cyclophosphamide, vincristine, and prednisolone), and bendamustine.
Patients who received obinutuzumab had significantly better progression-free survival than patients who received rituximab. The 3-year progression-free survival rate was 73.3% in the rituximab arm and 80% in the obinutuzumab arm (hazard ratio [HR]=0.66, P=0.0012).
There was no significant difference between the treatment arms with regard to overall survival. The 3-year overall survival was 92.1% in the rituximab arm and 94% in the obinutuzumab arm (HR=0.75, P=0.21).
The overall incidence of adverse events (AEs) was 98.3% in the rituximab arm and 99.5% in the obinutuzumab arm. The incidence of serious AEs was 39.9% and 46.1%, respectively.
The incidence of grade 3 or higher AEs was higher among patients who received obinutuzumab.
Grade 3 or higher AEs occurring in at least 5% of patients in either arm (rituximab and obinutuzumab, respectively) included neutropenia (67.8% and 74.6%), leukopenia (37.9% and 43.9%), febrile neutropenia (4.9% and 6.9%), infections and infestations (3.7% and 6.7%), and thrombocytopenia (2.7% and 6.1%). ![]()
The US Food and Drug Administration (FDA) has granted priority review to a supplemental biologics license application (sBLA) for obinutuzumab (Gazyva®).
With this sBLA, Genentech is seeking approval for obinutuzumab to be used, first in combination with chemotherapy and then alone as maintenance, in patients with previously untreated follicular lymphoma (FL).
The FDA grants priority review to applications for products that may provide significant improvements in the treatment, diagnosis, or prevention of serious conditions.
The agency’s goal is to take action on a priority review application within 6 months of receiving it, rather than the standard 10 months.
The FDA plans to make a decision on the sBLA for obinutuzumab by December 23, 2017.
The sBLA is supported by results of the GALLIUM study, which were presented at the 2016 ASH Annual Meeting.
About obinutuzumab
Obinutuzumab is a glycoengineered, humanized, monoclonal antibody that selectively binds to the extracellular domain of the CD20 antigen on B cells.
The drug is FDA-approved for use in combination with chlorambucil to treat patients with previously untreated chronic lymphocytic leukemia.
Obinutuzumab is also approved to treat FL patients who relapse after, or are refractory to, a rituximab-containing regimen. In these patients, obinutuzumab is given first in combination with bendamustine and then alone as maintenance.
The full prescribing information for obinutuzumab is available at http://www.Gazyva.com.
About the GALLIUM study
GALLIUM enrolled 1401 patients with previously untreated, indolent non-Hodgkin lymphoma, including 1202 with FL.
Half of the FL patients (n=601) were randomized to receive obinutuzumab plus chemotherapy (followed by obinutuzumab maintenance for up to 2 years), and half were randomized to rituximab plus chemotherapy (followed by rituximab maintenance for up to 2 years).
The different chemotherapies used were CHOP (cyclophosphamide, doxorubicin, vincristine, and prednisolone), CVP (cyclophosphamide, vincristine, and prednisolone), and bendamustine.
Patients who received obinutuzumab had significantly better progression-free survival than patients who received rituximab. The 3-year progression-free survival rate was 73.3% in the rituximab arm and 80% in the obinutuzumab arm (hazard ratio [HR]=0.66, P=0.0012).
There was no significant difference between the treatment arms with regard to overall survival. The 3-year overall survival was 92.1% in the rituximab arm and 94% in the obinutuzumab arm (HR=0.75, P=0.21).
The overall incidence of adverse events (AEs) was 98.3% in the rituximab arm and 99.5% in the obinutuzumab arm. The incidence of serious AEs was 39.9% and 46.1%, respectively.
The incidence of grade 3 or higher AEs was higher among patients who received obinutuzumab.
Grade 3 or higher AEs occurring in at least 5% of patients in either arm (rituximab and obinutuzumab, respectively) included neutropenia (67.8% and 74.6%), leukopenia (37.9% and 43.9%), febrile neutropenia (4.9% and 6.9%), infections and infestations (3.7% and 6.7%), and thrombocytopenia (2.7% and 6.1%). ![]()
Drug granted breakthrough designation for CTCL
The US Food and Drug Administration (FDA) has granted breakthrough therapy designation to mogamulizumab for the treatment of adults with cutaneous T-cell lymphoma (CTCL) who have received at least 1 prior systemic therapy.
Mogamulizumab is a humanized monoclonal antibody directed against CCR4. It is being developed by Kyowa Hakko Kirin Co., Ltd.
The breakthrough designation for mogamulizumab in CTCL is based on data from the phase 3 MAVORIC study, the largest randomized trial in CTCL.
In MAVORIC, researchers are comparing mogamulizumab and vorinostat in patients with CTCL (both mycosis fungoides and Sézary syndrome) who have failed at least 1 prior systemic treatment.
In April, Kyowa Hakko Kirin announced results from this trial, which showed that patients treated with mogamulizumab have significantly better progression-free survival than patients treated with vorinostat. The company also said mogamulizumab has a tolerable safety profile.
Kyowa Hakko Kirin has not provided any data from MAVORIC but is working with investigators on the future presentation and publication of results from this trial.
Results of a phase 1/2 study of mogamulizumab in previously treated CTCL patients were published in Blood in March 2015.
About breakthrough designation
The FDA’s breakthrough designation is intended to expedite the development and review of new treatments for serious or life-threatening conditions.
The designation entitles the company developing a therapy to more intensive FDA guidance on an efficient and accelerated development program, as well as eligibility for other actions to expedite FDA review, such as rolling submission and priority review.
To earn breakthrough designation, a treatment must show encouraging early clinical results demonstrating substantial improvement over available therapies with regard to a clinically significant endpoint, or it must fulfill an unmet need. ![]()
The US Food and Drug Administration (FDA) has granted breakthrough therapy designation to mogamulizumab for the treatment of adults with cutaneous T-cell lymphoma (CTCL) who have received at least 1 prior systemic therapy.
Mogamulizumab is a humanized monoclonal antibody directed against CCR4. It is being developed by Kyowa Hakko Kirin Co., Ltd.
The breakthrough designation for mogamulizumab in CTCL is based on data from the phase 3 MAVORIC study, the largest randomized trial in CTCL.
In MAVORIC, researchers are comparing mogamulizumab and vorinostat in patients with CTCL (both mycosis fungoides and Sézary syndrome) who have failed at least 1 prior systemic treatment.
In April, Kyowa Hakko Kirin announced results from this trial, which showed that patients treated with mogamulizumab have significantly better progression-free survival than patients treated with vorinostat. The company also said mogamulizumab has a tolerable safety profile.
Kyowa Hakko Kirin has not provided any data from MAVORIC but is working with investigators on the future presentation and publication of results from this trial.
Results of a phase 1/2 study of mogamulizumab in previously treated CTCL patients were published in Blood in March 2015.
About breakthrough designation
The FDA’s breakthrough designation is intended to expedite the development and review of new treatments for serious or life-threatening conditions.
The designation entitles the company developing a therapy to more intensive FDA guidance on an efficient and accelerated development program, as well as eligibility for other actions to expedite FDA review, such as rolling submission and priority review.
To earn breakthrough designation, a treatment must show encouraging early clinical results demonstrating substantial improvement over available therapies with regard to a clinically significant endpoint, or it must fulfill an unmet need. ![]()
The US Food and Drug Administration (FDA) has granted breakthrough therapy designation to mogamulizumab for the treatment of adults with cutaneous T-cell lymphoma (CTCL) who have received at least 1 prior systemic therapy.
Mogamulizumab is a humanized monoclonal antibody directed against CCR4. It is being developed by Kyowa Hakko Kirin Co., Ltd.
The breakthrough designation for mogamulizumab in CTCL is based on data from the phase 3 MAVORIC study, the largest randomized trial in CTCL.
In MAVORIC, researchers are comparing mogamulizumab and vorinostat in patients with CTCL (both mycosis fungoides and Sézary syndrome) who have failed at least 1 prior systemic treatment.
In April, Kyowa Hakko Kirin announced results from this trial, which showed that patients treated with mogamulizumab have significantly better progression-free survival than patients treated with vorinostat. The company also said mogamulizumab has a tolerable safety profile.
Kyowa Hakko Kirin has not provided any data from MAVORIC but is working with investigators on the future presentation and publication of results from this trial.
Results of a phase 1/2 study of mogamulizumab in previously treated CTCL patients were published in Blood in March 2015.
About breakthrough designation
The FDA’s breakthrough designation is intended to expedite the development and review of new treatments for serious or life-threatening conditions.
The designation entitles the company developing a therapy to more intensive FDA guidance on an efficient and accelerated development program, as well as eligibility for other actions to expedite FDA review, such as rolling submission and priority review.
To earn breakthrough designation, a treatment must show encouraging early clinical results demonstrating substantial improvement over available therapies with regard to a clinically significant endpoint, or it must fulfill an unmet need. ![]()
FDA grants priority review to BLA for emicizumab
The US Food and Drug Administration (FDA) has accepted for priority review the biologics license application (BLA) for emicizumab.
The BLA is for emicizumab as once-weekly prophylaxis for adults, adolescents, and children with hemophilia A and factor VIII inhibitors.
The FDA grants priority review to applications for products that may provide significant improvements in the treatment, diagnosis, or prevention of serious conditions.
The agency’s goal is to take action on a priority review application within 6 months of receiving it, rather than the standard 10 months.
The FDA plans to make a decision on the BLA for emicizumab by February 23, 2018.
About emicizumab
Emicizumab (formerly ACE910) is an investigational, bispecific monoclonal antibody designed to bring together factors IXa and X, proteins required to activate the natural coagulation cascade and restore the blood clotting process.
The drug is administered by subcutaneous injection of a ready-to-use solution. It was created by Chugai Pharmaceutical Co., Ltd. and is being co-developed by Chugai, Roche, and Genentech.
The BLA for emicizumab is based on results from a pair of phase 3 studies—HAVEN 1 and HAVEN 2.
Results from HAVEN 1 were published in NEJM and presented at the 26th ISTH Congress in July. Interim results from HAVEN 2 were presented at ISTH as well.
HAVEN 1
HAVEN 1 is a randomized, phase 3 study in which researchers evaluated the efficacy, safety, and pharmacokinetics of emicizumab prophylaxis compared to on-demand bypassing agents (BPAs; no prophylaxis) in adults and adolescents (12 years of age and older) with hemophilia A and inhibitors to factor VIII.
The study included 109 patients who were previously treated with BPAs on-demand or as prophylaxis.
There was a significant reduction in treated bleeds of 87% (risk rate=0.13, P<0.0001) with emicizumab compared with no prophylaxis.
Adverse events occurring in at least 5% of patients treated with emicizumab were local injection site reactions, headache, fatigue, upper respiratory tract infection, and arthralgia.
Two patients experienced thromboembolic events (TEs), and 3 had thrombotic microangiopathy (TMA) while receiving emicizumab prophylaxis and more than 100 u/kg/day of the BPA activated prothrombin complex concentrate, on average, for 24 hours or more before the event. Two of these patients had also received recombinant factor VIIa.
Neither TE required anticoagulation therapy, and 1 patient restarted emicizumab. The cases of TMA observed were transient, and 1 patient restarted emicizumab.
HAVEN 2
HAVEN 2 is a single-arm, phase 3 study in which researchers are evaluating the efficacy, safety, and pharmacokinetics of once-weekly emicizumab in children (younger than 12 years of age) with hemophilia A and inhibitors to factor VIII who require treatment with BPAs.
The interim analysis included 19 children. After a median observation time of 12 weeks, 1 of the 19 children had a treated bleed. There were no reported joint or muscle bleeds.
The most common adverse events were mild injection site reactions and nasopharyngitis. No TEs or TMA events were observed. ![]()
The US Food and Drug Administration (FDA) has accepted for priority review the biologics license application (BLA) for emicizumab.
The BLA is for emicizumab as once-weekly prophylaxis for adults, adolescents, and children with hemophilia A and factor VIII inhibitors.
The FDA grants priority review to applications for products that may provide significant improvements in the treatment, diagnosis, or prevention of serious conditions.
The agency’s goal is to take action on a priority review application within 6 months of receiving it, rather than the standard 10 months.
The FDA plans to make a decision on the BLA for emicizumab by February 23, 2018.
About emicizumab
Emicizumab (formerly ACE910) is an investigational, bispecific monoclonal antibody designed to bring together factors IXa and X, proteins required to activate the natural coagulation cascade and restore the blood clotting process.
The drug is administered by subcutaneous injection of a ready-to-use solution. It was created by Chugai Pharmaceutical Co., Ltd. and is being co-developed by Chugai, Roche, and Genentech.
The BLA for emicizumab is based on results from a pair of phase 3 studies—HAVEN 1 and HAVEN 2.
Results from HAVEN 1 were published in NEJM and presented at the 26th ISTH Congress in July. Interim results from HAVEN 2 were presented at ISTH as well.
HAVEN 1
HAVEN 1 is a randomized, phase 3 study in which researchers evaluated the efficacy, safety, and pharmacokinetics of emicizumab prophylaxis compared to on-demand bypassing agents (BPAs; no prophylaxis) in adults and adolescents (12 years of age and older) with hemophilia A and inhibitors to factor VIII.
The study included 109 patients who were previously treated with BPAs on-demand or as prophylaxis.
There was a significant reduction in treated bleeds of 87% (risk rate=0.13, P<0.0001) with emicizumab compared with no prophylaxis.
Adverse events occurring in at least 5% of patients treated with emicizumab were local injection site reactions, headache, fatigue, upper respiratory tract infection, and arthralgia.
Two patients experienced thromboembolic events (TEs), and 3 had thrombotic microangiopathy (TMA) while receiving emicizumab prophylaxis and more than 100 u/kg/day of the BPA activated prothrombin complex concentrate, on average, for 24 hours or more before the event. Two of these patients had also received recombinant factor VIIa.
Neither TE required anticoagulation therapy, and 1 patient restarted emicizumab. The cases of TMA observed were transient, and 1 patient restarted emicizumab.
HAVEN 2
HAVEN 2 is a single-arm, phase 3 study in which researchers are evaluating the efficacy, safety, and pharmacokinetics of once-weekly emicizumab in children (younger than 12 years of age) with hemophilia A and inhibitors to factor VIII who require treatment with BPAs.
The interim analysis included 19 children. After a median observation time of 12 weeks, 1 of the 19 children had a treated bleed. There were no reported joint or muscle bleeds.
The most common adverse events were mild injection site reactions and nasopharyngitis. No TEs or TMA events were observed. ![]()
The US Food and Drug Administration (FDA) has accepted for priority review the biologics license application (BLA) for emicizumab.
The BLA is for emicizumab as once-weekly prophylaxis for adults, adolescents, and children with hemophilia A and factor VIII inhibitors.
The FDA grants priority review to applications for products that may provide significant improvements in the treatment, diagnosis, or prevention of serious conditions.
The agency’s goal is to take action on a priority review application within 6 months of receiving it, rather than the standard 10 months.
The FDA plans to make a decision on the BLA for emicizumab by February 23, 2018.
About emicizumab
Emicizumab (formerly ACE910) is an investigational, bispecific monoclonal antibody designed to bring together factors IXa and X, proteins required to activate the natural coagulation cascade and restore the blood clotting process.
The drug is administered by subcutaneous injection of a ready-to-use solution. It was created by Chugai Pharmaceutical Co., Ltd. and is being co-developed by Chugai, Roche, and Genentech.
The BLA for emicizumab is based on results from a pair of phase 3 studies—HAVEN 1 and HAVEN 2.
Results from HAVEN 1 were published in NEJM and presented at the 26th ISTH Congress in July. Interim results from HAVEN 2 were presented at ISTH as well.
HAVEN 1
HAVEN 1 is a randomized, phase 3 study in which researchers evaluated the efficacy, safety, and pharmacokinetics of emicizumab prophylaxis compared to on-demand bypassing agents (BPAs; no prophylaxis) in adults and adolescents (12 years of age and older) with hemophilia A and inhibitors to factor VIII.
The study included 109 patients who were previously treated with BPAs on-demand or as prophylaxis.
There was a significant reduction in treated bleeds of 87% (risk rate=0.13, P<0.0001) with emicizumab compared with no prophylaxis.
Adverse events occurring in at least 5% of patients treated with emicizumab were local injection site reactions, headache, fatigue, upper respiratory tract infection, and arthralgia.
Two patients experienced thromboembolic events (TEs), and 3 had thrombotic microangiopathy (TMA) while receiving emicizumab prophylaxis and more than 100 u/kg/day of the BPA activated prothrombin complex concentrate, on average, for 24 hours or more before the event. Two of these patients had also received recombinant factor VIIa.
Neither TE required anticoagulation therapy, and 1 patient restarted emicizumab. The cases of TMA observed were transient, and 1 patient restarted emicizumab.
HAVEN 2
HAVEN 2 is a single-arm, phase 3 study in which researchers are evaluating the efficacy, safety, and pharmacokinetics of once-weekly emicizumab in children (younger than 12 years of age) with hemophilia A and inhibitors to factor VIII who require treatment with BPAs.
The interim analysis included 19 children. After a median observation time of 12 weeks, 1 of the 19 children had a treated bleed. There were no reported joint or muscle bleeds.
The most common adverse events were mild injection site reactions and nasopharyngitis. No TEs or TMA events were observed. ![]()
FDA approves new strengths of hemophilia therapy
The US Food and Drug Administration (FDA) has approved new product strengths for the factor VIII therapy simoctocog alfa (NUWIQ®).
Simoctocog alfa is the first B-domain-deleted recombinant factor VIII product derived from a human cell line—not chemically modified or fused with another protein—designed to treat hemophilia A.
Simoctocog alfa is FDA-approved to treat adults and children with hemophilia A. This includes on-demand treatment and control of bleeding episodes, routine prophylaxis to reduce the frequency of bleeding episodes, and perioperative management of bleeding.
Now, the FDA has approved single-dose simoctocog alfa vial strengths of 2500, 3000, and 4000 International Units (IU), which will be available for order in the US starting September 2017.
These new vial strengths will be provided in addition to the already available strengths of 250, 500, 1000, and 2000 IU.
“The new vial options will benefit patients, physicians, and healthcare professionals by providing greater treatment flexibility and convenience,” said Flemming Nielsen, president of Octapharma USA, makers of simoctocog alfa.
According to Octapharma, the additional vial strengths offer benefits beyond potentially reducing the number of vials used per patient. The new vial sizes may benefit heavier patients who could use fewer product vials and, in some cases, just one vial.
More vial options will increase dosing flexibility by allowing physicians to select various vial combinations to align closer to the prescribed dose. The new vial sizes could be particularly beneficial to patients using a pharmacokinetic (PK)-guided, personalized prophylaxis approach.
Results of Octapharma’s clinical trial on the PK-guided dosing with simoctocog alfa were published in Haemophilia in April.
This study, known as GENA-21 or NuPreviq, enrolled 66 previously treated adults with severe hemophilia A. Patients were originally started on infusions 3 times a week or every other day. Subsequent dosing intervals were then determined based on individual PK data.
The median dosing interval with PK-guided prophylaxis was 3.5 days, and 57% of patients were able to decrease infusions to twice-weekly or less.
The median weekly prophylaxis dose was reduced by 7.2%, from 100.0 IU kg−1 with standard prophylaxis to 92.8 IU kg−1 during the last 2 months of personalized prophylaxis.
For all bleeds, the mean annualized bleeding rate (ABR) during personalized prophylaxis was 1.45, and the median was 0 (interquartile range, [IQR]: 0, 1.9).
For spontaneous bleeds, the mean ABR was 0.79, and the median was 0 (IQR: 0, 0). For joint bleeds, the mean ABR was 0.91, and the median was 0 (IQR: 0, 0).
None of the patients developed FVIII inhibitors. There were no treatment-related serious or severe adverse events, clinically significant abnormalities in laboratory parameters, or cases of thromboembolism.
The ongoing trial GENA-21b is designed to confirm the results of GENA-21. ![]()
The US Food and Drug Administration (FDA) has approved new product strengths for the factor VIII therapy simoctocog alfa (NUWIQ®).
Simoctocog alfa is the first B-domain-deleted recombinant factor VIII product derived from a human cell line—not chemically modified or fused with another protein—designed to treat hemophilia A.
Simoctocog alfa is FDA-approved to treat adults and children with hemophilia A. This includes on-demand treatment and control of bleeding episodes, routine prophylaxis to reduce the frequency of bleeding episodes, and perioperative management of bleeding.
Now, the FDA has approved single-dose simoctocog alfa vial strengths of 2500, 3000, and 4000 International Units (IU), which will be available for order in the US starting September 2017.
These new vial strengths will be provided in addition to the already available strengths of 250, 500, 1000, and 2000 IU.
“The new vial options will benefit patients, physicians, and healthcare professionals by providing greater treatment flexibility and convenience,” said Flemming Nielsen, president of Octapharma USA, makers of simoctocog alfa.
According to Octapharma, the additional vial strengths offer benefits beyond potentially reducing the number of vials used per patient. The new vial sizes may benefit heavier patients who could use fewer product vials and, in some cases, just one vial.
More vial options will increase dosing flexibility by allowing physicians to select various vial combinations to align closer to the prescribed dose. The new vial sizes could be particularly beneficial to patients using a pharmacokinetic (PK)-guided, personalized prophylaxis approach.
Results of Octapharma’s clinical trial on the PK-guided dosing with simoctocog alfa were published in Haemophilia in April.
This study, known as GENA-21 or NuPreviq, enrolled 66 previously treated adults with severe hemophilia A. Patients were originally started on infusions 3 times a week or every other day. Subsequent dosing intervals were then determined based on individual PK data.
The median dosing interval with PK-guided prophylaxis was 3.5 days, and 57% of patients were able to decrease infusions to twice-weekly or less.
The median weekly prophylaxis dose was reduced by 7.2%, from 100.0 IU kg−1 with standard prophylaxis to 92.8 IU kg−1 during the last 2 months of personalized prophylaxis.
For all bleeds, the mean annualized bleeding rate (ABR) during personalized prophylaxis was 1.45, and the median was 0 (interquartile range, [IQR]: 0, 1.9).
For spontaneous bleeds, the mean ABR was 0.79, and the median was 0 (IQR: 0, 0). For joint bleeds, the mean ABR was 0.91, and the median was 0 (IQR: 0, 0).
None of the patients developed FVIII inhibitors. There were no treatment-related serious or severe adverse events, clinically significant abnormalities in laboratory parameters, or cases of thromboembolism.
The ongoing trial GENA-21b is designed to confirm the results of GENA-21. ![]()
The US Food and Drug Administration (FDA) has approved new product strengths for the factor VIII therapy simoctocog alfa (NUWIQ®).
Simoctocog alfa is the first B-domain-deleted recombinant factor VIII product derived from a human cell line—not chemically modified or fused with another protein—designed to treat hemophilia A.
Simoctocog alfa is FDA-approved to treat adults and children with hemophilia A. This includes on-demand treatment and control of bleeding episodes, routine prophylaxis to reduce the frequency of bleeding episodes, and perioperative management of bleeding.
Now, the FDA has approved single-dose simoctocog alfa vial strengths of 2500, 3000, and 4000 International Units (IU), which will be available for order in the US starting September 2017.
These new vial strengths will be provided in addition to the already available strengths of 250, 500, 1000, and 2000 IU.
“The new vial options will benefit patients, physicians, and healthcare professionals by providing greater treatment flexibility and convenience,” said Flemming Nielsen, president of Octapharma USA, makers of simoctocog alfa.
According to Octapharma, the additional vial strengths offer benefits beyond potentially reducing the number of vials used per patient. The new vial sizes may benefit heavier patients who could use fewer product vials and, in some cases, just one vial.
More vial options will increase dosing flexibility by allowing physicians to select various vial combinations to align closer to the prescribed dose. The new vial sizes could be particularly beneficial to patients using a pharmacokinetic (PK)-guided, personalized prophylaxis approach.
Results of Octapharma’s clinical trial on the PK-guided dosing with simoctocog alfa were published in Haemophilia in April.
This study, known as GENA-21 or NuPreviq, enrolled 66 previously treated adults with severe hemophilia A. Patients were originally started on infusions 3 times a week or every other day. Subsequent dosing intervals were then determined based on individual PK data.
The median dosing interval with PK-guided prophylaxis was 3.5 days, and 57% of patients were able to decrease infusions to twice-weekly or less.
The median weekly prophylaxis dose was reduced by 7.2%, from 100.0 IU kg−1 with standard prophylaxis to 92.8 IU kg−1 during the last 2 months of personalized prophylaxis.
For all bleeds, the mean annualized bleeding rate (ABR) during personalized prophylaxis was 1.45, and the median was 0 (interquartile range, [IQR]: 0, 1.9).
For spontaneous bleeds, the mean ABR was 0.79, and the median was 0 (IQR: 0, 0). For joint bleeds, the mean ABR was 0.91, and the median was 0 (IQR: 0, 0).
None of the patients developed FVIII inhibitors. There were no treatment-related serious or severe adverse events, clinically significant abnormalities in laboratory parameters, or cases of thromboembolism.
The ongoing trial GENA-21b is designed to confirm the results of GENA-21. ![]()
Study: Reference pricing does reduce prescription costs
Reference pricing effectively encourages patients to spend significantly less on prescription drugs, according to research published in NEJM.
Under the reference pricing strategy, the insurer or employer establishes its maximum contribution toward the price of therapeutically similar drugs, and the patient must pay the remainder out of pocket.
The insurer/employer contribution is based on the price of the lowest-priced drug in the therapeutic class, called the reference drug.
“If the patient chooses a cheap or moderately priced option, the employer’s contribution will cover most of the cost,” said study author James C. Robinson, PhD, of the University of California at Berkeley.
“However, if the patient insists on a particularly high-priced option, he or she will need to make a meaningful payment from personal resources.”
It has been theorized that this policy would encourage patients to save money by selecting cheaper drugs. However, little is known about how the policy has actually influenced patient spending.
The new study showed that reference pricing was associated with significant changes in drug selection and spending for patients covered by employment-based insurance in the US.
Researchers analyzed changes in prescriptions and pricing for 1302 drugs in 78 therapeutic classes in the US, before and after an alliance of private employers began using reference pricing.
The trends were compared to a cohort without reference pricing. The study’s dataset included 1.1 million prescriptions reimbursed from 2010 to 2014.
Implementation of reference pricing was associated with a 7% increase in prescriptions filled for the low-price reference drug within its therapeutic class, a 14% decrease in average price paid, and a 5% increase in consumer cost-sharing.
In the first 18 months after implementation, employers’ spending dropped $1.34 million, and employees’ cost-sharing increased $120,000.
Based on these findings, Dr Robinson and his colleagues concluded that reference pricing may be one instrument for influencing patients’ drug choices and drug prices paid by employers and insurers. The team believes that, in the future, pharmaceutical companies charging “premium prices” may need to demonstrate that their drugs provide “premium performance.”
“There is huge and unjustified variation within and across geographic areas in the prices charged for almost every test and treatment, drug and device, office visit and hospitalization,” Dr Robinson said.
“It’s not a surprise when one considers that most patients are covered by health insurance and, hence, do not shop among competing providers on the basis of price. Some providers look at price-unconscious consumer demand and ask themselves, ‘Why don’t we raise our prices?’”
This research was funded by the US Agency for Healthcare Research and Quality and the Genentech Foundation. ![]()
Reference pricing effectively encourages patients to spend significantly less on prescription drugs, according to research published in NEJM.
Under the reference pricing strategy, the insurer or employer establishes its maximum contribution toward the price of therapeutically similar drugs, and the patient must pay the remainder out of pocket.
The insurer/employer contribution is based on the price of the lowest-priced drug in the therapeutic class, called the reference drug.
“If the patient chooses a cheap or moderately priced option, the employer’s contribution will cover most of the cost,” said study author James C. Robinson, PhD, of the University of California at Berkeley.
“However, if the patient insists on a particularly high-priced option, he or she will need to make a meaningful payment from personal resources.”
It has been theorized that this policy would encourage patients to save money by selecting cheaper drugs. However, little is known about how the policy has actually influenced patient spending.
The new study showed that reference pricing was associated with significant changes in drug selection and spending for patients covered by employment-based insurance in the US.
Researchers analyzed changes in prescriptions and pricing for 1302 drugs in 78 therapeutic classes in the US, before and after an alliance of private employers began using reference pricing.
The trends were compared to a cohort without reference pricing. The study’s dataset included 1.1 million prescriptions reimbursed from 2010 to 2014.
Implementation of reference pricing was associated with a 7% increase in prescriptions filled for the low-price reference drug within its therapeutic class, a 14% decrease in average price paid, and a 5% increase in consumer cost-sharing.
In the first 18 months after implementation, employers’ spending dropped $1.34 million, and employees’ cost-sharing increased $120,000.
Based on these findings, Dr Robinson and his colleagues concluded that reference pricing may be one instrument for influencing patients’ drug choices and drug prices paid by employers and insurers. The team believes that, in the future, pharmaceutical companies charging “premium prices” may need to demonstrate that their drugs provide “premium performance.”
“There is huge and unjustified variation within and across geographic areas in the prices charged for almost every test and treatment, drug and device, office visit and hospitalization,” Dr Robinson said.
“It’s not a surprise when one considers that most patients are covered by health insurance and, hence, do not shop among competing providers on the basis of price. Some providers look at price-unconscious consumer demand and ask themselves, ‘Why don’t we raise our prices?’”
This research was funded by the US Agency for Healthcare Research and Quality and the Genentech Foundation. ![]()
Reference pricing effectively encourages patients to spend significantly less on prescription drugs, according to research published in NEJM.
Under the reference pricing strategy, the insurer or employer establishes its maximum contribution toward the price of therapeutically similar drugs, and the patient must pay the remainder out of pocket.
The insurer/employer contribution is based on the price of the lowest-priced drug in the therapeutic class, called the reference drug.
“If the patient chooses a cheap or moderately priced option, the employer’s contribution will cover most of the cost,” said study author James C. Robinson, PhD, of the University of California at Berkeley.
“However, if the patient insists on a particularly high-priced option, he or she will need to make a meaningful payment from personal resources.”
It has been theorized that this policy would encourage patients to save money by selecting cheaper drugs. However, little is known about how the policy has actually influenced patient spending.
The new study showed that reference pricing was associated with significant changes in drug selection and spending for patients covered by employment-based insurance in the US.
Researchers analyzed changes in prescriptions and pricing for 1302 drugs in 78 therapeutic classes in the US, before and after an alliance of private employers began using reference pricing.
The trends were compared to a cohort without reference pricing. The study’s dataset included 1.1 million prescriptions reimbursed from 2010 to 2014.
Implementation of reference pricing was associated with a 7% increase in prescriptions filled for the low-price reference drug within its therapeutic class, a 14% decrease in average price paid, and a 5% increase in consumer cost-sharing.
In the first 18 months after implementation, employers’ spending dropped $1.34 million, and employees’ cost-sharing increased $120,000.
Based on these findings, Dr Robinson and his colleagues concluded that reference pricing may be one instrument for influencing patients’ drug choices and drug prices paid by employers and insurers. The team believes that, in the future, pharmaceutical companies charging “premium prices” may need to demonstrate that their drugs provide “premium performance.”
“There is huge and unjustified variation within and across geographic areas in the prices charged for almost every test and treatment, drug and device, office visit and hospitalization,” Dr Robinson said.
“It’s not a surprise when one considers that most patients are covered by health insurance and, hence, do not shop among competing providers on the basis of price. Some providers look at price-unconscious consumer demand and ask themselves, ‘Why don’t we raise our prices?’”
This research was funded by the US Agency for Healthcare Research and Quality and the Genentech Foundation. ![]()
Drug granted orphan designation for chemo-induced ototoxicity
The US Food and Drug Administration (FDA) has granted orphan drug designation to SENS-401 to be used for the prevention of platinum-induced ototoxicity in pediatric patients.
Platinum-based chemotherapies, particularly cisplatin, can induce severe hearing loss in cancer patients, but there is no pharmaceutical agent approved to treat this side effect.
“Hearing loss in pediatric oncology patients is one of the most frequent side effects of cisplatin treatment and may disable them for the rest of their lives,” said Nawal Ouzren, CEO of Sensorion, the company developing SENS-401.
“Based on its unique profile and the data generated to date, we believe SENS-401 has the potential to be a safe and effective treatment for this serious medical condition where a significant unmet need exists. As such, we look forward to working with the FDA and EMA [European Medicines Agency] to set up an IND [investigational new drug application] and design a phase 2 clinical trial in order to evaluate SENS-401 in this indication.”
About SENS-401
SENS-401 (R-azasetron besylate) is a small molecule intended to protect and preserve inner ear tissue when lesions cause progressive or sequelar hearing impairments. The drug can be taken orally or via an injection.
SENS-401 is one of the two enantiomer forms of SENS-218 (azasetron), a racemic molecule belonging to the family of setrons marketed in Asia under the name Serotone. Pharmacological and pharmacokinetic tests have shown a superior profile for SENS-401 compared with the other enantiomer or the racemic form.
Healthy subjects demonstrated a “very good clinical tolerance” to SENS-401 in a phase 1 study, according to Sensorion. The company is planning to launch a phase 2 trial of the drug for platinum-induced ototoxicity in 2018.
About orphan designation
The FDA grants orphan designation to products intended to treat, diagnose, or prevent diseases/disorders that affect fewer than 200,000 people in the US.
The designation provides incentives for sponsors to develop products for rare diseases. This may include tax credits toward the cost of clinical trials, prescription drug user fee waivers, and 7 years of market exclusivity if the product is approved. ![]()
The US Food and Drug Administration (FDA) has granted orphan drug designation to SENS-401 to be used for the prevention of platinum-induced ototoxicity in pediatric patients.
Platinum-based chemotherapies, particularly cisplatin, can induce severe hearing loss in cancer patients, but there is no pharmaceutical agent approved to treat this side effect.
“Hearing loss in pediatric oncology patients is one of the most frequent side effects of cisplatin treatment and may disable them for the rest of their lives,” said Nawal Ouzren, CEO of Sensorion, the company developing SENS-401.
“Based on its unique profile and the data generated to date, we believe SENS-401 has the potential to be a safe and effective treatment for this serious medical condition where a significant unmet need exists. As such, we look forward to working with the FDA and EMA [European Medicines Agency] to set up an IND [investigational new drug application] and design a phase 2 clinical trial in order to evaluate SENS-401 in this indication.”
About SENS-401
SENS-401 (R-azasetron besylate) is a small molecule intended to protect and preserve inner ear tissue when lesions cause progressive or sequelar hearing impairments. The drug can be taken orally or via an injection.
SENS-401 is one of the two enantiomer forms of SENS-218 (azasetron), a racemic molecule belonging to the family of setrons marketed in Asia under the name Serotone. Pharmacological and pharmacokinetic tests have shown a superior profile for SENS-401 compared with the other enantiomer or the racemic form.
Healthy subjects demonstrated a “very good clinical tolerance” to SENS-401 in a phase 1 study, according to Sensorion. The company is planning to launch a phase 2 trial of the drug for platinum-induced ototoxicity in 2018.
About orphan designation
The FDA grants orphan designation to products intended to treat, diagnose, or prevent diseases/disorders that affect fewer than 200,000 people in the US.
The designation provides incentives for sponsors to develop products for rare diseases. This may include tax credits toward the cost of clinical trials, prescription drug user fee waivers, and 7 years of market exclusivity if the product is approved. ![]()
The US Food and Drug Administration (FDA) has granted orphan drug designation to SENS-401 to be used for the prevention of platinum-induced ototoxicity in pediatric patients.
Platinum-based chemotherapies, particularly cisplatin, can induce severe hearing loss in cancer patients, but there is no pharmaceutical agent approved to treat this side effect.
“Hearing loss in pediatric oncology patients is one of the most frequent side effects of cisplatin treatment and may disable them for the rest of their lives,” said Nawal Ouzren, CEO of Sensorion, the company developing SENS-401.
“Based on its unique profile and the data generated to date, we believe SENS-401 has the potential to be a safe and effective treatment for this serious medical condition where a significant unmet need exists. As such, we look forward to working with the FDA and EMA [European Medicines Agency] to set up an IND [investigational new drug application] and design a phase 2 clinical trial in order to evaluate SENS-401 in this indication.”
About SENS-401
SENS-401 (R-azasetron besylate) is a small molecule intended to protect and preserve inner ear tissue when lesions cause progressive or sequelar hearing impairments. The drug can be taken orally or via an injection.
SENS-401 is one of the two enantiomer forms of SENS-218 (azasetron), a racemic molecule belonging to the family of setrons marketed in Asia under the name Serotone. Pharmacological and pharmacokinetic tests have shown a superior profile for SENS-401 compared with the other enantiomer or the racemic form.
Healthy subjects demonstrated a “very good clinical tolerance” to SENS-401 in a phase 1 study, according to Sensorion. The company is planning to launch a phase 2 trial of the drug for platinum-induced ototoxicity in 2018.
About orphan designation
The FDA grants orphan designation to products intended to treat, diagnose, or prevent diseases/disorders that affect fewer than 200,000 people in the US.
The designation provides incentives for sponsors to develop products for rare diseases. This may include tax credits toward the cost of clinical trials, prescription drug user fee waivers, and 7 years of market exclusivity if the product is approved. ![]()
FDA grants drug orphan designation for AML
The US Food and Drug Administration (FDA) has granted orphan drug designation to SY-1425 for the treatment of acute myeloid leukemia (AML).
SY-1425 is an oral, first-in-class, selective retinoic acid receptor alpha (RARα) agonist. It is currently under investigation in a phase 2 trial of patients with AML and myelodysplastic syndromes (MDS).
“We believe that SY-1425 may provide a meaningful benefit for subsets of AML patients whose disease is driven by abnormally high expression of the RARA or IRF8 genes,” said David A. Roth, MD, chief medical officer at Syros Pharmaceuticals, the company developing SY-1425.
“Receiving orphan drug designation is an important regulatory milestone in the development of SY-1425. We’re pleased with the continued progress of the ongoing phase 2 clinical trial, and we look forward to presenting initial clinical data in the fourth quarter of this year.”
Using its gene control platform, Syros discovered subsets of AML and MDS patients with super-enhancers associated with RARA or IRF8. These super-enhancers are believed to drive overexpression of the RARA or IRF8 genes, locking cells in an immature, undifferentiated, and proliferative state and leading to disease.
In preclinical studies, SY-1425 promoted differentiation of AML cells with high RARA or IRF8 expression and inhibited tumor growth and prolonged survival in patient-derived xenograft models of AML with high RARA expression.
In the ongoing phase 2 trial, researchers are assessing the safety and efficacy of SY-1425 as a single agent in 4 AML and MDS patient populations, as well as SY-1425 in combination with azacitidine in newly diagnosed AML patients who are not suitable candidates for standard chemotherapy.
All patients are prospectively selected using biomarkers for high expression of RARA or IRF8. Additional details about the trial can be found at https://clinicaltrials.gov/ct2/show/NCT02807558.
About orphan designation
The FDA grants orphan designation to products intended to treat, diagnose, or prevent diseases/disorders that affect fewer than 200,000 people in the US.
The designation provides incentives for sponsors to develop products for rare diseases. This may include tax credits toward the cost of clinical trials, prescription drug user fee waivers, and 7 years of market exclusivity if the product is approved. ![]()
The US Food and Drug Administration (FDA) has granted orphan drug designation to SY-1425 for the treatment of acute myeloid leukemia (AML).
SY-1425 is an oral, first-in-class, selective retinoic acid receptor alpha (RARα) agonist. It is currently under investigation in a phase 2 trial of patients with AML and myelodysplastic syndromes (MDS).
“We believe that SY-1425 may provide a meaningful benefit for subsets of AML patients whose disease is driven by abnormally high expression of the RARA or IRF8 genes,” said David A. Roth, MD, chief medical officer at Syros Pharmaceuticals, the company developing SY-1425.
“Receiving orphan drug designation is an important regulatory milestone in the development of SY-1425. We’re pleased with the continued progress of the ongoing phase 2 clinical trial, and we look forward to presenting initial clinical data in the fourth quarter of this year.”
Using its gene control platform, Syros discovered subsets of AML and MDS patients with super-enhancers associated with RARA or IRF8. These super-enhancers are believed to drive overexpression of the RARA or IRF8 genes, locking cells in an immature, undifferentiated, and proliferative state and leading to disease.
In preclinical studies, SY-1425 promoted differentiation of AML cells with high RARA or IRF8 expression and inhibited tumor growth and prolonged survival in patient-derived xenograft models of AML with high RARA expression.
In the ongoing phase 2 trial, researchers are assessing the safety and efficacy of SY-1425 as a single agent in 4 AML and MDS patient populations, as well as SY-1425 in combination with azacitidine in newly diagnosed AML patients who are not suitable candidates for standard chemotherapy.
All patients are prospectively selected using biomarkers for high expression of RARA or IRF8. Additional details about the trial can be found at https://clinicaltrials.gov/ct2/show/NCT02807558.
About orphan designation
The FDA grants orphan designation to products intended to treat, diagnose, or prevent diseases/disorders that affect fewer than 200,000 people in the US.
The designation provides incentives for sponsors to develop products for rare diseases. This may include tax credits toward the cost of clinical trials, prescription drug user fee waivers, and 7 years of market exclusivity if the product is approved. ![]()
The US Food and Drug Administration (FDA) has granted orphan drug designation to SY-1425 for the treatment of acute myeloid leukemia (AML).
SY-1425 is an oral, first-in-class, selective retinoic acid receptor alpha (RARα) agonist. It is currently under investigation in a phase 2 trial of patients with AML and myelodysplastic syndromes (MDS).
“We believe that SY-1425 may provide a meaningful benefit for subsets of AML patients whose disease is driven by abnormally high expression of the RARA or IRF8 genes,” said David A. Roth, MD, chief medical officer at Syros Pharmaceuticals, the company developing SY-1425.
“Receiving orphan drug designation is an important regulatory milestone in the development of SY-1425. We’re pleased with the continued progress of the ongoing phase 2 clinical trial, and we look forward to presenting initial clinical data in the fourth quarter of this year.”
Using its gene control platform, Syros discovered subsets of AML and MDS patients with super-enhancers associated with RARA or IRF8. These super-enhancers are believed to drive overexpression of the RARA or IRF8 genes, locking cells in an immature, undifferentiated, and proliferative state and leading to disease.
In preclinical studies, SY-1425 promoted differentiation of AML cells with high RARA or IRF8 expression and inhibited tumor growth and prolonged survival in patient-derived xenograft models of AML with high RARA expression.
In the ongoing phase 2 trial, researchers are assessing the safety and efficacy of SY-1425 as a single agent in 4 AML and MDS patient populations, as well as SY-1425 in combination with azacitidine in newly diagnosed AML patients who are not suitable candidates for standard chemotherapy.
All patients are prospectively selected using biomarkers for high expression of RARA or IRF8. Additional details about the trial can be found at https://clinicaltrials.gov/ct2/show/NCT02807558.
About orphan designation
The FDA grants orphan designation to products intended to treat, diagnose, or prevent diseases/disorders that affect fewer than 200,000 people in the US.
The designation provides incentives for sponsors to develop products for rare diseases. This may include tax credits toward the cost of clinical trials, prescription drug user fee waivers, and 7 years of market exclusivity if the product is approved.
Researchers compare world health authorities
A new study has revealed substantial differences between health authorities in different regions of the world.
A pair of researchers compared 12 different regulatory authorities responsible for approving drugs and medical products.
The researchers collected data* on annual budgets, new drug approvals per year, numbers of reviewers, standard and median review times, fees for new drug applications (NDAs), and other measurements.
The results were published in Nature Reviews Drug Discovery.
For the 2015 fiscal year, the US Food and Drug Administration (FDA) had the highest budget—$1.19 billion—and India’s Central Drugs Standard Control Organization (CDSCO) had the lowest—$26 million.
In 2016, the FDA again had the highest budget—$1.23 billion—while Health Canada and Switzerland’s SwissMedic had the lowest—$108 million.
In 2016, the European Medicines Agency (EMA) had the highest number of reviewers—4500—and SwissMedic had the lowest—60. (Data from 2015 were not included.)
In 2015, Japan’s Pharmaceuticals and Medical Devices Agency had the highest number of NDA submissions—127—and Health Canada had the lowest—27. Meanwhile, the Chinese FDA had the highest number of new drug approvals—72—and India’s CDSCO had the lowest—17.
The UK’s Medicines and Healthcare products Regulatory Agency (MHRA) technically had the most new drug approvals in 2015, at 146, but not all of these were unique, as the number included all decentralized applications, both with the UK as the reference member state and approvals from concerned member states.
In 2016, the EMA had the highest number of NDA submissions—68—and Health Canada had the lowest—25. Singapore’s Health Sciences Authority had the highest number of new drug approvals—72—while the US FDA and India’s CDSCO had the lowest—22.
The shortest standard review period was 210 days. This is the standard for the EMA, the UK’s MHRA, and Russia’s Roszdravnadzor. The regulatory agency with the longest standard review time—900 days—is the Chinese FDA.
The shortest median time to new drug approval in 2015 was 230 days, for the UK’s MHRA. The longest was 834 days, for the Brazilian Health Surveillance Agency.
The highest NDA review fees were those charged by the US FDA—$2.3 million. The lowest were those charged by India’s CDSCO—50,000 Indian rupees or about USD$1000.
The researchers noted that these data suggest products are being evaluated via different processes and according to different standards, which makes it challenging for pharmaceutical companies to develop drugs for simultaneous submission to all regulatory authorities.
Therefore, a harmonization of approval requirements and processes could significantly improve efficiency.
“Patients would profit especially since new drugs would be available faster and at lower prices,” said study author Thomas D. Szucs, MD, PhD, of the University of Basel in Switzerland.
“This suggests that companies and authorities should strengthen their international collaboration and communicate better with each other.”
*Some data were missing for most of the 12 agencies studied.
A new study has revealed substantial differences between health authorities in different regions of the world.
A pair of researchers compared 12 different regulatory authorities responsible for approving drugs and medical products.
The researchers collected data* on annual budgets, new drug approvals per year, numbers of reviewers, standard and median review times, fees for new drug applications (NDAs), and other measurements.
The results were published in Nature Reviews Drug Discovery.
For the 2015 fiscal year, the US Food and Drug Administration (FDA) had the highest budget—$1.19 billion—and India’s Central Drugs Standard Control Organization (CDSCO) had the lowest—$26 million.
In 2016, the FDA again had the highest budget—$1.23 billion—while Health Canada and Switzerland’s SwissMedic had the lowest—$108 million.
In 2016, the European Medicines Agency (EMA) had the highest number of reviewers—4500—and SwissMedic had the lowest—60. (Data from 2015 were not included.)
In 2015, Japan’s Pharmaceuticals and Medical Devices Agency had the highest number of NDA submissions—127—and Health Canada had the lowest—27. Meanwhile, the Chinese FDA had the highest number of new drug approvals—72—and India’s CDSCO had the lowest—17.
The UK’s Medicines and Healthcare products Regulatory Agency (MHRA) technically had the most new drug approvals in 2015, at 146, but not all of these were unique, as the number included all decentralized applications, both with the UK as the reference member state and approvals from concerned member states.
In 2016, the EMA had the highest number of NDA submissions—68—and Health Canada had the lowest—25. Singapore’s Health Sciences Authority had the highest number of new drug approvals—72—while the US FDA and India’s CDSCO had the lowest—22.
The shortest standard review period was 210 days. This is the standard for the EMA, the UK’s MHRA, and Russia’s Roszdravnadzor. The regulatory agency with the longest standard review time—900 days—is the Chinese FDA.
The shortest median time to new drug approval in 2015 was 230 days, for the UK’s MHRA. The longest was 834 days, for the Brazilian Health Surveillance Agency.
The highest NDA review fees were those charged by the US FDA—$2.3 million. The lowest were those charged by India’s CDSCO—50,000 Indian rupees or about USD$1000.
The researchers noted that these data suggest products are being evaluated via different processes and according to different standards, which makes it challenging for pharmaceutical companies to develop drugs for simultaneous submission to all regulatory authorities.
Therefore, a harmonization of approval requirements and processes could significantly improve efficiency.
“Patients would profit especially since new drugs would be available faster and at lower prices,” said study author Thomas D. Szucs, MD, PhD, of the University of Basel in Switzerland.
“This suggests that companies and authorities should strengthen their international collaboration and communicate better with each other.”
*Some data were missing for most of the 12 agencies studied.
A new study has revealed substantial differences between health authorities in different regions of the world.
A pair of researchers compared 12 different regulatory authorities responsible for approving drugs and medical products.
The researchers collected data* on annual budgets, new drug approvals per year, numbers of reviewers, standard and median review times, fees for new drug applications (NDAs), and other measurements.
The results were published in Nature Reviews Drug Discovery.
For the 2015 fiscal year, the US Food and Drug Administration (FDA) had the highest budget—$1.19 billion—and India’s Central Drugs Standard Control Organization (CDSCO) had the lowest—$26 million.
In 2016, the FDA again had the highest budget—$1.23 billion—while Health Canada and Switzerland’s SwissMedic had the lowest—$108 million.
In 2016, the European Medicines Agency (EMA) had the highest number of reviewers—4500—and SwissMedic had the lowest—60. (Data from 2015 were not included.)
In 2015, Japan’s Pharmaceuticals and Medical Devices Agency had the highest number of NDA submissions—127—and Health Canada had the lowest—27. Meanwhile, the Chinese FDA had the highest number of new drug approvals—72—and India’s CDSCO had the lowest—17.
The UK’s Medicines and Healthcare products Regulatory Agency (MHRA) technically had the most new drug approvals in 2015, at 146, but not all of these were unique, as the number included all decentralized applications, both with the UK as the reference member state and approvals from concerned member states.
In 2016, the EMA had the highest number of NDA submissions—68—and Health Canada had the lowest—25. Singapore’s Health Sciences Authority had the highest number of new drug approvals—72—while the US FDA and India’s CDSCO had the lowest—22.
The shortest standard review period was 210 days. This is the standard for the EMA, the UK’s MHRA, and Russia’s Roszdravnadzor. The regulatory agency with the longest standard review time—900 days—is the Chinese FDA.
The shortest median time to new drug approval in 2015 was 230 days, for the UK’s MHRA. The longest was 834 days, for the Brazilian Health Surveillance Agency.
The highest NDA review fees were those charged by the US FDA—$2.3 million. The lowest were those charged by India’s CDSCO—50,000 Indian rupees or about USD$1000.
The researchers noted that these data suggest products are being evaluated via different processes and according to different standards, which makes it challenging for pharmaceutical companies to develop drugs for simultaneous submission to all regulatory authorities.
Therefore, a harmonization of approval requirements and processes could significantly improve efficiency.
“Patients would profit especially since new drugs would be available faster and at lower prices,” said study author Thomas D. Szucs, MD, PhD, of the University of Basel in Switzerland.
“This suggests that companies and authorities should strengthen their international collaboration and communicate better with each other.”
*Some data were missing for most of the 12 agencies studied.
FDA approves inotuzumab ozogamicin for rel/ref ALL
The US Food and Drug Administration (FDA) has approved inotuzumab ozogamicin (Besponsa®) for the treatment of adults with relapsed or refractory B-cell precursor acute lymphoblastic leukemia (ALL).
The labeling for inotuzumab ozogamicin includes a boxed warning stating that the drug poses a risk of hepatotoxicity, including hepatic veno-occlusive disease (or sinusoidal obstruction syndrome), as well as an increased risk of post-transplant non-relapse mortality.
The full prescribing information for inotuzumab ozogamicin is available at https://www.accessdata.fda.gov/drugsatfda_docs/label/2017/761040s000lbl.pdf.
Inotuzumab ozogamicin is an antibody-drug conjugate that consists of a monoclonal antibody targeting CD22 and a cytotoxic agent known as calicheamicin.
The product originates from a collaboration between Pfizer and Celltech (now UCB), but Pfizer has sole responsibility for all manufacturing and clinical development activities.
Inotuzumab ozogamicin was reviewed and approved under the FDA’s breakthrough therapy designation and priority review programs.
The application for inotuzumab ozogamicin was supported by results from the phase 3 INO-VATE trial, which were published in NEJM in June 2016.
The trial enrolled 326 adults with relapsed or refractory B-cell ALL. Patients received inotuzumab ozogamicin or 1 of 3 chemotherapy regimens (high-dose cytarabine, cytarabine plus mitoxantrone, or fludarabine, cytarabine, and granulocyte colony-stimulating factor).
The rate of complete remission, including incomplete hematologic recovery, was 80.7% in the inotuzumab arm and 29.4% in the chemotherapy arm (P<0.001). The median duration of remission was 4.6 months and 3.1 months, respectively (P=0.03).
Forty-one percent of patients treated with inotuzumab and 11% of those who received chemotherapy proceeded to stem cell transplant directly after treatment (P<0.001).
The median progression-free survival was 5.0 months in the inotuzumab arm and 1.8 months in the chemotherapy arm (P<0.001).
The median overall survival was 7.7 months and 6.7 months, respectively (P=0.04). This did not meet the prespecified boundary of significance (P=0.0208).
Liver-related adverse events were more common in the inotuzumab arm than the chemotherapy arm. The most frequent of these were increased aspartate aminotransferase level (20% vs 10%), hyperbilirubinemia (15% vs 10%), and increased alanine aminotransferase level (14% vs 11%).
Veno-occlusive liver disease occurred in 11% of patients in the inotuzumab arm and 1% in the chemotherapy arm.
There were 17 deaths during treatment in the inotuzumab arm and 11 in the chemotherapy arm. Four deaths were considered related to inotuzumab, and 2 were thought to be related to chemotherapy.
The US Food and Drug Administration (FDA) has approved inotuzumab ozogamicin (Besponsa®) for the treatment of adults with relapsed or refractory B-cell precursor acute lymphoblastic leukemia (ALL).
The labeling for inotuzumab ozogamicin includes a boxed warning stating that the drug poses a risk of hepatotoxicity, including hepatic veno-occlusive disease (or sinusoidal obstruction syndrome), as well as an increased risk of post-transplant non-relapse mortality.
The full prescribing information for inotuzumab ozogamicin is available at https://www.accessdata.fda.gov/drugsatfda_docs/label/2017/761040s000lbl.pdf.
Inotuzumab ozogamicin is an antibody-drug conjugate that consists of a monoclonal antibody targeting CD22 and a cytotoxic agent known as calicheamicin.
The product originates from a collaboration between Pfizer and Celltech (now UCB), but Pfizer has sole responsibility for all manufacturing and clinical development activities.
Inotuzumab ozogamicin was reviewed and approved under the FDA’s breakthrough therapy designation and priority review programs.
The application for inotuzumab ozogamicin was supported by results from the phase 3 INO-VATE trial, which were published in NEJM in June 2016.
The trial enrolled 326 adults with relapsed or refractory B-cell ALL. Patients received inotuzumab ozogamicin or 1 of 3 chemotherapy regimens (high-dose cytarabine, cytarabine plus mitoxantrone, or fludarabine, cytarabine, and granulocyte colony-stimulating factor).
The rate of complete remission, including incomplete hematologic recovery, was 80.7% in the inotuzumab arm and 29.4% in the chemotherapy arm (P<0.001). The median duration of remission was 4.6 months and 3.1 months, respectively (P=0.03).
Forty-one percent of patients treated with inotuzumab and 11% of those who received chemotherapy proceeded to stem cell transplant directly after treatment (P<0.001).
The median progression-free survival was 5.0 months in the inotuzumab arm and 1.8 months in the chemotherapy arm (P<0.001).
The median overall survival was 7.7 months and 6.7 months, respectively (P=0.04). This did not meet the prespecified boundary of significance (P=0.0208).
Liver-related adverse events were more common in the inotuzumab arm than the chemotherapy arm. The most frequent of these were increased aspartate aminotransferase level (20% vs 10%), hyperbilirubinemia (15% vs 10%), and increased alanine aminotransferase level (14% vs 11%).
Veno-occlusive liver disease occurred in 11% of patients in the inotuzumab arm and 1% in the chemotherapy arm.
There were 17 deaths during treatment in the inotuzumab arm and 11 in the chemotherapy arm. Four deaths were considered related to inotuzumab, and 2 were thought to be related to chemotherapy.
The US Food and Drug Administration (FDA) has approved inotuzumab ozogamicin (Besponsa®) for the treatment of adults with relapsed or refractory B-cell precursor acute lymphoblastic leukemia (ALL).
The labeling for inotuzumab ozogamicin includes a boxed warning stating that the drug poses a risk of hepatotoxicity, including hepatic veno-occlusive disease (or sinusoidal obstruction syndrome), as well as an increased risk of post-transplant non-relapse mortality.
The full prescribing information for inotuzumab ozogamicin is available at https://www.accessdata.fda.gov/drugsatfda_docs/label/2017/761040s000lbl.pdf.
Inotuzumab ozogamicin is an antibody-drug conjugate that consists of a monoclonal antibody targeting CD22 and a cytotoxic agent known as calicheamicin.
The product originates from a collaboration between Pfizer and Celltech (now UCB), but Pfizer has sole responsibility for all manufacturing and clinical development activities.
Inotuzumab ozogamicin was reviewed and approved under the FDA’s breakthrough therapy designation and priority review programs.
The application for inotuzumab ozogamicin was supported by results from the phase 3 INO-VATE trial, which were published in NEJM in June 2016.
The trial enrolled 326 adults with relapsed or refractory B-cell ALL. Patients received inotuzumab ozogamicin or 1 of 3 chemotherapy regimens (high-dose cytarabine, cytarabine plus mitoxantrone, or fludarabine, cytarabine, and granulocyte colony-stimulating factor).
The rate of complete remission, including incomplete hematologic recovery, was 80.7% in the inotuzumab arm and 29.4% in the chemotherapy arm (P<0.001). The median duration of remission was 4.6 months and 3.1 months, respectively (P=0.03).
Forty-one percent of patients treated with inotuzumab and 11% of those who received chemotherapy proceeded to stem cell transplant directly after treatment (P<0.001).
The median progression-free survival was 5.0 months in the inotuzumab arm and 1.8 months in the chemotherapy arm (P<0.001).
The median overall survival was 7.7 months and 6.7 months, respectively (P=0.04). This did not meet the prespecified boundary of significance (P=0.0208).
Liver-related adverse events were more common in the inotuzumab arm than the chemotherapy arm. The most frequent of these were increased aspartate aminotransferase level (20% vs 10%), hyperbilirubinemia (15% vs 10%), and increased alanine aminotransferase level (14% vs 11%).
Veno-occlusive liver disease occurred in 11% of patients in the inotuzumab arm and 1% in the chemotherapy arm.
There were 17 deaths during treatment in the inotuzumab arm and 11 in the chemotherapy arm. Four deaths were considered related to inotuzumab, and 2 were thought to be related to chemotherapy.
Post-approval trials for accelerated drugs fall short
New research has revealed shortcomings of post-approval studies for drugs granted accelerated approval in the US.
Researchers found that, for drugs granted accelerated approval from 2009 to 2013, both pre-approval and post-approval trials had limitations in their design and the endpoints used.
“One might expect accelerated approval confirmatory trials to be much more rigorous than the pre-approval trials,” said study author Aaron S. Kesselheim, MD, of Brigham and Women’s Hospital in Boston, Massachusetts.
“But we found that there were few differences in these key design features of the trials conducted before or after approval.”
Dr Kesselheim and his colleagues reported these findings in JAMA.
The researchers examined pre- and post-approval clinical trials of drugs granted accelerated approval by the US Food and Drug Administration (FDA) between 2009 and 2013.
During that time, the FDA granted 22 drugs accelerated approval for 24 indications (15 of them for hematologic disorders).
Fourteen of the indications were approved on the basis of single-intervention-group studies that enrolled a median of 132 patients.
The FDA ordered 38 post-approval studies to confirm the safety and efficacy of the drugs.
Three years after the last drug’s approval, half of those studies (n=19) were not complete. Eight (42%) of the incomplete studies were either terminated or delayed by more than 1 year.
For 14 of the 24 indications (58%), results from the post-approval studies were not available after a median of 5 years of follow-up.
Study comparison
Published reports were available for 18 of the 19 completed post-approval studies. The characteristics of these studies did not differ much from the 30 pre-approval studies.
There were no statistically significant differences with regard to median patient enrollment (P=0.17), the use of randomized (P=0.31) or double-blind trials (P=0.17), the use of placebo as a comparator (P=0.17), or the lack of a comparator (P=0.21).
However, there was a significant difference in the use of an active comparator (P=0.02), with more post-approval studies using an active comparator.
The researchers also found that 17 of the 18 post-approval trials still used surrogate measures of effect as primary endpoints.
There was no significant difference between pre- and post-approval trials when it came to the use of disease response (P=0.17) or most other surrogate measures (P=0.21) as the trials’ primary endpoint.
The same was true for overall survival (P=0.20), although significantly more post-approval studies used progression-free survival (P=0.001) as a primary endpoint.
“It is important to use clinical endpoints in testing investigational drugs whenever possible because there are numerous cases of drugs approved on the basis of a surrogate measure that turn out to later not effect actual clinical outcomes—or even make them worse,” Dr Kesselheim said.
To address these issues and improve the quality of confirmatory studies, Dr Kesselheim suggested the FDA clearly describe the limitations in the pre-approval data that will need to be addressed in post-approval studies.
He also suggested the agency work with manufacturers to ensure that post-approval studies are conducted using design features that will be optimally useful for confirming the efficacy of the drug.
New research has revealed shortcomings of post-approval studies for drugs granted accelerated approval in the US.
Researchers found that, for drugs granted accelerated approval from 2009 to 2013, both pre-approval and post-approval trials had limitations in their design and the endpoints used.
“One might expect accelerated approval confirmatory trials to be much more rigorous than the pre-approval trials,” said study author Aaron S. Kesselheim, MD, of Brigham and Women’s Hospital in Boston, Massachusetts.
“But we found that there were few differences in these key design features of the trials conducted before or after approval.”
Dr Kesselheim and his colleagues reported these findings in JAMA.
The researchers examined pre- and post-approval clinical trials of drugs granted accelerated approval by the US Food and Drug Administration (FDA) between 2009 and 2013.
During that time, the FDA granted 22 drugs accelerated approval for 24 indications (15 of them for hematologic disorders).
Fourteen of the indications were approved on the basis of single-intervention-group studies that enrolled a median of 132 patients.
The FDA ordered 38 post-approval studies to confirm the safety and efficacy of the drugs.
Three years after the last drug’s approval, half of those studies (n=19) were not complete. Eight (42%) of the incomplete studies were either terminated or delayed by more than 1 year.
For 14 of the 24 indications (58%), results from the post-approval studies were not available after a median of 5 years of follow-up.
Study comparison
Published reports were available for 18 of the 19 completed post-approval studies. The characteristics of these studies did not differ much from the 30 pre-approval studies.
There were no statistically significant differences with regard to median patient enrollment (P=0.17), the use of randomized (P=0.31) or double-blind trials (P=0.17), the use of placebo as a comparator (P=0.17), or the lack of a comparator (P=0.21).
However, there was a significant difference in the use of an active comparator (P=0.02), with more post-approval studies using an active comparator.
The researchers also found that 17 of the 18 post-approval trials still used surrogate measures of effect as primary endpoints.
There was no significant difference between pre- and post-approval trials when it came to the use of disease response (P=0.17) or most other surrogate measures (P=0.21) as the trials’ primary endpoint.
The same was true for overall survival (P=0.20), although significantly more post-approval studies used progression-free survival (P=0.001) as a primary endpoint.
“It is important to use clinical endpoints in testing investigational drugs whenever possible because there are numerous cases of drugs approved on the basis of a surrogate measure that turn out to later not effect actual clinical outcomes—or even make them worse,” Dr Kesselheim said.
To address these issues and improve the quality of confirmatory studies, Dr Kesselheim suggested the FDA clearly describe the limitations in the pre-approval data that will need to be addressed in post-approval studies.
He also suggested the agency work with manufacturers to ensure that post-approval studies are conducted using design features that will be optimally useful for confirming the efficacy of the drug.
New research has revealed shortcomings of post-approval studies for drugs granted accelerated approval in the US.
Researchers found that, for drugs granted accelerated approval from 2009 to 2013, both pre-approval and post-approval trials had limitations in their design and the endpoints used.
“One might expect accelerated approval confirmatory trials to be much more rigorous than the pre-approval trials,” said study author Aaron S. Kesselheim, MD, of Brigham and Women’s Hospital in Boston, Massachusetts.
“But we found that there were few differences in these key design features of the trials conducted before or after approval.”
Dr Kesselheim and his colleagues reported these findings in JAMA.
The researchers examined pre- and post-approval clinical trials of drugs granted accelerated approval by the US Food and Drug Administration (FDA) between 2009 and 2013.
During that time, the FDA granted 22 drugs accelerated approval for 24 indications (15 of them for hematologic disorders).
Fourteen of the indications were approved on the basis of single-intervention-group studies that enrolled a median of 132 patients.
The FDA ordered 38 post-approval studies to confirm the safety and efficacy of the drugs.
Three years after the last drug’s approval, half of those studies (n=19) were not complete. Eight (42%) of the incomplete studies were either terminated or delayed by more than 1 year.
For 14 of the 24 indications (58%), results from the post-approval studies were not available after a median of 5 years of follow-up.
Study comparison
Published reports were available for 18 of the 19 completed post-approval studies. The characteristics of these studies did not differ much from the 30 pre-approval studies.
There were no statistically significant differences with regard to median patient enrollment (P=0.17), the use of randomized (P=0.31) or double-blind trials (P=0.17), the use of placebo as a comparator (P=0.17), or the lack of a comparator (P=0.21).
However, there was a significant difference in the use of an active comparator (P=0.02), with more post-approval studies using an active comparator.
The researchers also found that 17 of the 18 post-approval trials still used surrogate measures of effect as primary endpoints.
There was no significant difference between pre- and post-approval trials when it came to the use of disease response (P=0.17) or most other surrogate measures (P=0.21) as the trials’ primary endpoint.
The same was true for overall survival (P=0.20), although significantly more post-approval studies used progression-free survival (P=0.001) as a primary endpoint.
“It is important to use clinical endpoints in testing investigational drugs whenever possible because there are numerous cases of drugs approved on the basis of a surrogate measure that turn out to later not effect actual clinical outcomes—or even make them worse,” Dr Kesselheim said.
To address these issues and improve the quality of confirmatory studies, Dr Kesselheim suggested the FDA clearly describe the limitations in the pre-approval data that will need to be addressed in post-approval studies.
He also suggested the agency work with manufacturers to ensure that post-approval studies are conducted using design features that will be optimally useful for confirming the efficacy of the drug.