User login
Discovery reveals potential for viral cancer treatment

Image by Eric Smith
Researchers say they have discovered critical details that explain how a cellular response system tells the difference between damage to the body’s own DNA and the foreign DNA of an invading virus.
The team believes this discovery could aid the development of new cancer-selective viral therapies, and it may help explain why aging, cancers, and other diseases
seem to open the door to viral infections.
“Our study reveals fundamental mechanisms that distinguish DNA breaks at cellular and viral genomes to trigger different responses that protect the host,” said Clodagh O’Shea, of the Salk Institute for Biological Studies in La Jolla, California.
“The findings may also explain why certain conditions like aging, cancer chemotherapy, and inflammation make us more susceptible to viral infection.”
Dr O’Shea and Govind Shah, PhD, also of the Salk Institute, reported these findings in Cell.
The pair described how a cluster of proteins known as the MRN complex detects DNA breaks and amplifies its response through histones.
MRN starts a domino effect, activating histones on surrounding chromosomes, which summons a cascade of additional proteins and results in a cell-wide, all-hands-on-deck alarm to help mend the DNA.
If the cell can’t fix the DNA break, it will induce apoptosis—a self-destruct mechanism that helps to prevent mutated cells from replicating and therefore prevents tumor growth.
“What’s interesting is that even a single break transmits a global signal through the cell, halting cell division and growth,” Dr O’Shea said. “This response prevents replication so the cell doesn’t pass on a break.”
Drs O’Shea and Shah also found that, when it comes to defending against DNA viruses, the cell’s response system begins the same way—with MRN detecting breaks. But it never progresses to the global alarm signal in the case of the virus.
Typically, a common DNA virus enters the cell’s nucleus and turns on genes to replicate its own DNA. The cell detects the unauthorized replication, and the MRN complex grabs and selectively neutralizes viral DNA without triggering a global response that would arrest or kill the cell.
So the MRN response to the virus stays localized and only selectively prevents viral, but not cellular, replication.
When both threats to the genome are present, MRN will activate the massive response at the DNA break, and no MRN is left to respond to the virus. This means the virus is effectively ignored while the cell responds to the more massive alarm.
“The requirement of MRN for sensing both cellular and viral genome breaks has profound consequences,” Dr O’Shea said.
“When MRN is recruited to cellular DNA breaks, it can no longer sense and respond to incoming viral genomes. Thus, the act of responding to cellular genome breaks inactivates the host’s defenses to viral replication.”
Dr O’Shea said this may explain why people who have high levels of cellular DNA damage—such as cancer patients—are more susceptible to viral infections.
“Having damaged DNA compromises our cells’ ability to fight viral infection, while having healthy DNA boosts our cells’ ability to catch viral DNA,” Dr Shah said. “Our work implies that we may be able to engineer viruses that selectively kill cancer cells.”
The researchers aim to use this new knowledge to create viruses that are destroyed in normal cells but replicate specifically in cancer cells.
Unlike normal cells, cancer cells almost always have very high levels of DNA damage. In cancer cells, MRN is already so preoccupied with responding to DNA breaks that an engineered virus could sneak in undetected.
“Cancer cells, by definition, have high mutation rates and genomic instability even at the very earliest stages,” Dr O’Shea said. “So you could imagine building a virus that could destroy even the earliest lesions and be used as a prophylactic.” ![]()

Image by Eric Smith
Researchers say they have discovered critical details that explain how a cellular response system tells the difference between damage to the body’s own DNA and the foreign DNA of an invading virus.
The team believes this discovery could aid the development of new cancer-selective viral therapies, and it may help explain why aging, cancers, and other diseases
seem to open the door to viral infections.
“Our study reveals fundamental mechanisms that distinguish DNA breaks at cellular and viral genomes to trigger different responses that protect the host,” said Clodagh O’Shea, of the Salk Institute for Biological Studies in La Jolla, California.
“The findings may also explain why certain conditions like aging, cancer chemotherapy, and inflammation make us more susceptible to viral infection.”
Dr O’Shea and Govind Shah, PhD, also of the Salk Institute, reported these findings in Cell.
The pair described how a cluster of proteins known as the MRN complex detects DNA breaks and amplifies its response through histones.
MRN starts a domino effect, activating histones on surrounding chromosomes, which summons a cascade of additional proteins and results in a cell-wide, all-hands-on-deck alarm to help mend the DNA.
If the cell can’t fix the DNA break, it will induce apoptosis—a self-destruct mechanism that helps to prevent mutated cells from replicating and therefore prevents tumor growth.
“What’s interesting is that even a single break transmits a global signal through the cell, halting cell division and growth,” Dr O’Shea said. “This response prevents replication so the cell doesn’t pass on a break.”
Drs O’Shea and Shah also found that, when it comes to defending against DNA viruses, the cell’s response system begins the same way—with MRN detecting breaks. But it never progresses to the global alarm signal in the case of the virus.
Typically, a common DNA virus enters the cell’s nucleus and turns on genes to replicate its own DNA. The cell detects the unauthorized replication, and the MRN complex grabs and selectively neutralizes viral DNA without triggering a global response that would arrest or kill the cell.
So the MRN response to the virus stays localized and only selectively prevents viral, but not cellular, replication.
When both threats to the genome are present, MRN will activate the massive response at the DNA break, and no MRN is left to respond to the virus. This means the virus is effectively ignored while the cell responds to the more massive alarm.
“The requirement of MRN for sensing both cellular and viral genome breaks has profound consequences,” Dr O’Shea said.
“When MRN is recruited to cellular DNA breaks, it can no longer sense and respond to incoming viral genomes. Thus, the act of responding to cellular genome breaks inactivates the host’s defenses to viral replication.”
Dr O’Shea said this may explain why people who have high levels of cellular DNA damage—such as cancer patients—are more susceptible to viral infections.
“Having damaged DNA compromises our cells’ ability to fight viral infection, while having healthy DNA boosts our cells’ ability to catch viral DNA,” Dr Shah said. “Our work implies that we may be able to engineer viruses that selectively kill cancer cells.”
The researchers aim to use this new knowledge to create viruses that are destroyed in normal cells but replicate specifically in cancer cells.
Unlike normal cells, cancer cells almost always have very high levels of DNA damage. In cancer cells, MRN is already so preoccupied with responding to DNA breaks that an engineered virus could sneak in undetected.
“Cancer cells, by definition, have high mutation rates and genomic instability even at the very earliest stages,” Dr O’Shea said. “So you could imagine building a virus that could destroy even the earliest lesions and be used as a prophylactic.” ![]()

Image by Eric Smith
Researchers say they have discovered critical details that explain how a cellular response system tells the difference between damage to the body’s own DNA and the foreign DNA of an invading virus.
The team believes this discovery could aid the development of new cancer-selective viral therapies, and it may help explain why aging, cancers, and other diseases
seem to open the door to viral infections.
“Our study reveals fundamental mechanisms that distinguish DNA breaks at cellular and viral genomes to trigger different responses that protect the host,” said Clodagh O’Shea, of the Salk Institute for Biological Studies in La Jolla, California.
“The findings may also explain why certain conditions like aging, cancer chemotherapy, and inflammation make us more susceptible to viral infection.”
Dr O’Shea and Govind Shah, PhD, also of the Salk Institute, reported these findings in Cell.
The pair described how a cluster of proteins known as the MRN complex detects DNA breaks and amplifies its response through histones.
MRN starts a domino effect, activating histones on surrounding chromosomes, which summons a cascade of additional proteins and results in a cell-wide, all-hands-on-deck alarm to help mend the DNA.
If the cell can’t fix the DNA break, it will induce apoptosis—a self-destruct mechanism that helps to prevent mutated cells from replicating and therefore prevents tumor growth.
“What’s interesting is that even a single break transmits a global signal through the cell, halting cell division and growth,” Dr O’Shea said. “This response prevents replication so the cell doesn’t pass on a break.”
Drs O’Shea and Shah also found that, when it comes to defending against DNA viruses, the cell’s response system begins the same way—with MRN detecting breaks. But it never progresses to the global alarm signal in the case of the virus.
Typically, a common DNA virus enters the cell’s nucleus and turns on genes to replicate its own DNA. The cell detects the unauthorized replication, and the MRN complex grabs and selectively neutralizes viral DNA without triggering a global response that would arrest or kill the cell.
So the MRN response to the virus stays localized and only selectively prevents viral, but not cellular, replication.
When both threats to the genome are present, MRN will activate the massive response at the DNA break, and no MRN is left to respond to the virus. This means the virus is effectively ignored while the cell responds to the more massive alarm.
“The requirement of MRN for sensing both cellular and viral genome breaks has profound consequences,” Dr O’Shea said.
“When MRN is recruited to cellular DNA breaks, it can no longer sense and respond to incoming viral genomes. Thus, the act of responding to cellular genome breaks inactivates the host’s defenses to viral replication.”
Dr O’Shea said this may explain why people who have high levels of cellular DNA damage—such as cancer patients—are more susceptible to viral infections.
“Having damaged DNA compromises our cells’ ability to fight viral infection, while having healthy DNA boosts our cells’ ability to catch viral DNA,” Dr Shah said. “Our work implies that we may be able to engineer viruses that selectively kill cancer cells.”
The researchers aim to use this new knowledge to create viruses that are destroyed in normal cells but replicate specifically in cancer cells.
Unlike normal cells, cancer cells almost always have very high levels of DNA damage. In cancer cells, MRN is already so preoccupied with responding to DNA breaks that an engineered virus could sneak in undetected.
“Cancer cells, by definition, have high mutation rates and genomic instability even at the very earliest stages,” Dr O’Shea said. “So you could imagine building a virus that could destroy even the earliest lesions and be used as a prophylactic.” ![]()
Database details driver mutations

Photo by Darren Baker
Scientists have created an online database of mutations that have been shown to drive cancers in preclinical or clinical research.
The database, called the Cancer Driver Log (CanDL), currently includes mutations in 62 genes, with hundreds of distinct variants across multiple cancers.
Sameek Roychowdhury, MD, PhD, of The Ohio State University in Columbus, and his colleauges described CanDL in the Journal of Molecular Diagnostics.
“CanDL is a database of gene mutations that have been functionally characterized or have been targeted clinically or preclinically with approved or investigational agents,” Dr Roychowdhury explained.
“Currently, pathology laboratories that sequence tumor tissue must manually research the scientific literature for individual mutations to determine whether they are considered a driver or a passenger to facilitate clinical interpretation.”
“CanDL expedites this time-consuming process by placing key information about known and possible driver mutations that might be effective targets for drug development at their fingertips.”
CanDL entries can be searched by gene or amino acid variants, and they can be downloaded for custom analyses.
The database also includes a mechanism for users to contribute novel driver mutations in open collaboration with the Roychowdhury lab. The team plans to update the database quarterly. ![]()

Photo by Darren Baker
Scientists have created an online database of mutations that have been shown to drive cancers in preclinical or clinical research.
The database, called the Cancer Driver Log (CanDL), currently includes mutations in 62 genes, with hundreds of distinct variants across multiple cancers.
Sameek Roychowdhury, MD, PhD, of The Ohio State University in Columbus, and his colleauges described CanDL in the Journal of Molecular Diagnostics.
“CanDL is a database of gene mutations that have been functionally characterized or have been targeted clinically or preclinically with approved or investigational agents,” Dr Roychowdhury explained.
“Currently, pathology laboratories that sequence tumor tissue must manually research the scientific literature for individual mutations to determine whether they are considered a driver or a passenger to facilitate clinical interpretation.”
“CanDL expedites this time-consuming process by placing key information about known and possible driver mutations that might be effective targets for drug development at their fingertips.”
CanDL entries can be searched by gene or amino acid variants, and they can be downloaded for custom analyses.
The database also includes a mechanism for users to contribute novel driver mutations in open collaboration with the Roychowdhury lab. The team plans to update the database quarterly. ![]()

Photo by Darren Baker
Scientists have created an online database of mutations that have been shown to drive cancers in preclinical or clinical research.
The database, called the Cancer Driver Log (CanDL), currently includes mutations in 62 genes, with hundreds of distinct variants across multiple cancers.
Sameek Roychowdhury, MD, PhD, of The Ohio State University in Columbus, and his colleauges described CanDL in the Journal of Molecular Diagnostics.
“CanDL is a database of gene mutations that have been functionally characterized or have been targeted clinically or preclinically with approved or investigational agents,” Dr Roychowdhury explained.
“Currently, pathology laboratories that sequence tumor tissue must manually research the scientific literature for individual mutations to determine whether they are considered a driver or a passenger to facilitate clinical interpretation.”
“CanDL expedites this time-consuming process by placing key information about known and possible driver mutations that might be effective targets for drug development at their fingertips.”
CanDL entries can be searched by gene or amino acid variants, and they can be downloaded for custom analyses.
The database also includes a mechanism for users to contribute novel driver mutations in open collaboration with the Roychowdhury lab. The team plans to update the database quarterly. ![]()
Team quantifies CAM use among seniors with cancer

Photo by Rhoda Baer
A new study suggests that seniors with cancer may be taking complementary or alternative medicines (CAMs) without their oncologists’ knowledge.
In this single-center study, 27% of senior cancer patients took CAMs at some point during their cancer care.
CAM usage was highest among patients ages 80 to 89, women, Caucasians, and patients with solid tumor malignancies.
Polypharmacy and certain comorbidities were linked to CAM use as well.
Researchers reported these findings in the Journal of Geriatric Oncology.
“Currently, few oncologists are aware of the alternative medicines their patients take,” said study author Ginah Nightingale, PharmD, of Thomas Jefferson University in Philadelphia, Pennsylvania.
“Patients often fail to disclose the CAMs they take because they think they are safe, natural, nontoxic, and not relevant to their cancer care; because they think their doctor will disapprove; or because the doctor doesn’t specifically ask.”
To quantify CAM use in older cancer patients treated at their institution, Dr Nightingale and her colleagues surveyed patients who came to the Senior Adult Oncology Center at Thomas Jefferson University.
In a single visit, patients were seen by a medical oncologist, geriatrician, clinical pharmacist, social worker, and dietician. As part of this assessment, the patients brought in the contents of their medicine cabinets, and the medications they actively used were reviewed and recorded.
A total of 234 patients were included in the final analysis. Their mean age was 79.9 (range, 61–98). Most (87%) had solid tumor malignancies, were Caucasian (74%), and were female (64%).
In all, 26.5% of patients (n=62) had taken at least 1 CAM during their cancer care, with 19.2% taking 1 CAM, 6.4% taking 2, 0.4% taking 3, and 0.4% taking 4 or more CAMs. The highest number of CAMs taken was 10.
CAM usage was highest among patients ages 80 to 89, women, Caucasians, and patients with solid tumor malignancies.
Comorbidities significantly associated with CAM use were vision impairment (P=0.048) and urologic comorbidities (P=0.021). Polypharmacy (concurrent use of 5 or more medications) was significantly associated with CAM use as well (P=0.045).
Some of the commonly used CAMs were mega-dose vitamins or minerals, as well as treatments for macular degeneration, stomach probiotics, and joint health.
The researchers did not examine the potential adverse effects of these medications, but Dr Nightingale said some are known to have a biochemical effect on the body and other drugs.
“It is very important to do a comprehensive screen of all of the medications that older cancer patients take, including CAMs,” she added. “Clear and transparent documentation of CAM use should be recorded in the patient’s medical record. This documentation should indicate that patient-specific communication and/or education was provided so that shared and informed decisions by the patient can be made regarding the continued use of these medications.” ![]()

Photo by Rhoda Baer
A new study suggests that seniors with cancer may be taking complementary or alternative medicines (CAMs) without their oncologists’ knowledge.
In this single-center study, 27% of senior cancer patients took CAMs at some point during their cancer care.
CAM usage was highest among patients ages 80 to 89, women, Caucasians, and patients with solid tumor malignancies.
Polypharmacy and certain comorbidities were linked to CAM use as well.
Researchers reported these findings in the Journal of Geriatric Oncology.
“Currently, few oncologists are aware of the alternative medicines their patients take,” said study author Ginah Nightingale, PharmD, of Thomas Jefferson University in Philadelphia, Pennsylvania.
“Patients often fail to disclose the CAMs they take because they think they are safe, natural, nontoxic, and not relevant to their cancer care; because they think their doctor will disapprove; or because the doctor doesn’t specifically ask.”
To quantify CAM use in older cancer patients treated at their institution, Dr Nightingale and her colleagues surveyed patients who came to the Senior Adult Oncology Center at Thomas Jefferson University.
In a single visit, patients were seen by a medical oncologist, geriatrician, clinical pharmacist, social worker, and dietician. As part of this assessment, the patients brought in the contents of their medicine cabinets, and the medications they actively used were reviewed and recorded.
A total of 234 patients were included in the final analysis. Their mean age was 79.9 (range, 61–98). Most (87%) had solid tumor malignancies, were Caucasian (74%), and were female (64%).
In all, 26.5% of patients (n=62) had taken at least 1 CAM during their cancer care, with 19.2% taking 1 CAM, 6.4% taking 2, 0.4% taking 3, and 0.4% taking 4 or more CAMs. The highest number of CAMs taken was 10.
CAM usage was highest among patients ages 80 to 89, women, Caucasians, and patients with solid tumor malignancies.
Comorbidities significantly associated with CAM use were vision impairment (P=0.048) and urologic comorbidities (P=0.021). Polypharmacy (concurrent use of 5 or more medications) was significantly associated with CAM use as well (P=0.045).
Some of the commonly used CAMs were mega-dose vitamins or minerals, as well as treatments for macular degeneration, stomach probiotics, and joint health.
The researchers did not examine the potential adverse effects of these medications, but Dr Nightingale said some are known to have a biochemical effect on the body and other drugs.
“It is very important to do a comprehensive screen of all of the medications that older cancer patients take, including CAMs,” she added. “Clear and transparent documentation of CAM use should be recorded in the patient’s medical record. This documentation should indicate that patient-specific communication and/or education was provided so that shared and informed decisions by the patient can be made regarding the continued use of these medications.” ![]()

Photo by Rhoda Baer
A new study suggests that seniors with cancer may be taking complementary or alternative medicines (CAMs) without their oncologists’ knowledge.
In this single-center study, 27% of senior cancer patients took CAMs at some point during their cancer care.
CAM usage was highest among patients ages 80 to 89, women, Caucasians, and patients with solid tumor malignancies.
Polypharmacy and certain comorbidities were linked to CAM use as well.
Researchers reported these findings in the Journal of Geriatric Oncology.
“Currently, few oncologists are aware of the alternative medicines their patients take,” said study author Ginah Nightingale, PharmD, of Thomas Jefferson University in Philadelphia, Pennsylvania.
“Patients often fail to disclose the CAMs they take because they think they are safe, natural, nontoxic, and not relevant to their cancer care; because they think their doctor will disapprove; or because the doctor doesn’t specifically ask.”
To quantify CAM use in older cancer patients treated at their institution, Dr Nightingale and her colleagues surveyed patients who came to the Senior Adult Oncology Center at Thomas Jefferson University.
In a single visit, patients were seen by a medical oncologist, geriatrician, clinical pharmacist, social worker, and dietician. As part of this assessment, the patients brought in the contents of their medicine cabinets, and the medications they actively used were reviewed and recorded.
A total of 234 patients were included in the final analysis. Their mean age was 79.9 (range, 61–98). Most (87%) had solid tumor malignancies, were Caucasian (74%), and were female (64%).
In all, 26.5% of patients (n=62) had taken at least 1 CAM during their cancer care, with 19.2% taking 1 CAM, 6.4% taking 2, 0.4% taking 3, and 0.4% taking 4 or more CAMs. The highest number of CAMs taken was 10.
CAM usage was highest among patients ages 80 to 89, women, Caucasians, and patients with solid tumor malignancies.
Comorbidities significantly associated with CAM use were vision impairment (P=0.048) and urologic comorbidities (P=0.021). Polypharmacy (concurrent use of 5 or more medications) was significantly associated with CAM use as well (P=0.045).
Some of the commonly used CAMs were mega-dose vitamins or minerals, as well as treatments for macular degeneration, stomach probiotics, and joint health.
The researchers did not examine the potential adverse effects of these medications, but Dr Nightingale said some are known to have a biochemical effect on the body and other drugs.
“It is very important to do a comprehensive screen of all of the medications that older cancer patients take, including CAMs,” she added. “Clear and transparent documentation of CAM use should be recorded in the patient’s medical record. This documentation should indicate that patient-specific communication and/or education was provided so that shared and informed decisions by the patient can be made regarding the continued use of these medications.” ![]()
Study reveals approaches to aid, prevent apoptosis

apoptosis in cancer cells
Scientists say they have gained new insight into the role Bax plays in apoptosis.
The Bax protein is known to be a key regulator of apoptosis, mediating the release of cytochrome c to the cytosol via oligomerization in the outer mitochondrial membrane before pore formation.
But the exact mechanism of Bax assembly was previously unclear.
Now, research published in Nature Communications has provided some clarity.
Katia Cosentino, PhD, of the Max Planck Institute for Intelligent Systems in Stuttgart, Germany, and her colleagues conducted this research, examining how the mitochondrial membrane becomes permeable.
The team’s experiments on artificial membrane systems showed that Bax is initially inserted into the membrane as a single molecule.
Once inserted, one Bax molecule will join up with a second Bax molecule to form a stable complex, the Bax dimers. From these dimers, larger complexes are formed.
“Surprisingly, Bax complexes have no standard size, but we observed a mixture of different-sized Bax species, and these species are mostly based on dimer units,” Dr Cosentino said.
She and her colleagues noted that these Bax complexes form the pores through which cytochrome c exits the mitochondrial membrane.
But the process of pore formation is finely controlled by other proteins. Some (such as cBid) enable the assembly of Bax elements, while others (such as Bcl-xL) induce their dismantling.
“The differing size of the Bax complexes in the pore formation is likely part of the reason why earlier investigations on pore formation conveyed contradictory results,” Dr Cosentino said.
She and her colleagues believe that, based on these findings, they can make some initial recommendations for medical intervention in the apoptotic process.
They think that, to promote apoptosis, it should be enough to initiate the first step of activating Bax proteins because the subsequent steps of self-organization will then happen automatically.
Conversely, the team’s findings suggest apoptosis can be prevented when drugs force the dismantling of the Bax dimers into their individual elements. ![]()

apoptosis in cancer cells
Scientists say they have gained new insight into the role Bax plays in apoptosis.
The Bax protein is known to be a key regulator of apoptosis, mediating the release of cytochrome c to the cytosol via oligomerization in the outer mitochondrial membrane before pore formation.
But the exact mechanism of Bax assembly was previously unclear.
Now, research published in Nature Communications has provided some clarity.
Katia Cosentino, PhD, of the Max Planck Institute for Intelligent Systems in Stuttgart, Germany, and her colleagues conducted this research, examining how the mitochondrial membrane becomes permeable.
The team’s experiments on artificial membrane systems showed that Bax is initially inserted into the membrane as a single molecule.
Once inserted, one Bax molecule will join up with a second Bax molecule to form a stable complex, the Bax dimers. From these dimers, larger complexes are formed.
“Surprisingly, Bax complexes have no standard size, but we observed a mixture of different-sized Bax species, and these species are mostly based on dimer units,” Dr Cosentino said.
She and her colleagues noted that these Bax complexes form the pores through which cytochrome c exits the mitochondrial membrane.
But the process of pore formation is finely controlled by other proteins. Some (such as cBid) enable the assembly of Bax elements, while others (such as Bcl-xL) induce their dismantling.
“The differing size of the Bax complexes in the pore formation is likely part of the reason why earlier investigations on pore formation conveyed contradictory results,” Dr Cosentino said.
She and her colleagues believe that, based on these findings, they can make some initial recommendations for medical intervention in the apoptotic process.
They think that, to promote apoptosis, it should be enough to initiate the first step of activating Bax proteins because the subsequent steps of self-organization will then happen automatically.
Conversely, the team’s findings suggest apoptosis can be prevented when drugs force the dismantling of the Bax dimers into their individual elements. ![]()

apoptosis in cancer cells
Scientists say they have gained new insight into the role Bax plays in apoptosis.
The Bax protein is known to be a key regulator of apoptosis, mediating the release of cytochrome c to the cytosol via oligomerization in the outer mitochondrial membrane before pore formation.
But the exact mechanism of Bax assembly was previously unclear.
Now, research published in Nature Communications has provided some clarity.
Katia Cosentino, PhD, of the Max Planck Institute for Intelligent Systems in Stuttgart, Germany, and her colleagues conducted this research, examining how the mitochondrial membrane becomes permeable.
The team’s experiments on artificial membrane systems showed that Bax is initially inserted into the membrane as a single molecule.
Once inserted, one Bax molecule will join up with a second Bax molecule to form a stable complex, the Bax dimers. From these dimers, larger complexes are formed.
“Surprisingly, Bax complexes have no standard size, but we observed a mixture of different-sized Bax species, and these species are mostly based on dimer units,” Dr Cosentino said.
She and her colleagues noted that these Bax complexes form the pores through which cytochrome c exits the mitochondrial membrane.
But the process of pore formation is finely controlled by other proteins. Some (such as cBid) enable the assembly of Bax elements, while others (such as Bcl-xL) induce their dismantling.
“The differing size of the Bax complexes in the pore formation is likely part of the reason why earlier investigations on pore formation conveyed contradictory results,” Dr Cosentino said.
She and her colleagues believe that, based on these findings, they can make some initial recommendations for medical intervention in the apoptotic process.
They think that, to promote apoptosis, it should be enough to initiate the first step of activating Bax proteins because the subsequent steps of self-organization will then happen automatically.
Conversely, the team’s findings suggest apoptosis can be prevented when drugs force the dismantling of the Bax dimers into their individual elements. ![]()
Enzyme may be target for malaria, toxoplasmosis
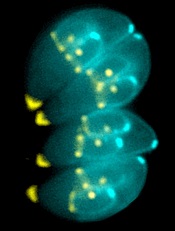
Image by Ke Hu & John Murray
Researchers say they have determined the structure of an enzyme that is vital to the infectious behavior of the parasites that cause toxoplasmosis and malaria.
And this has revealed a potentially druggable target that could prevent the parasites from entering and exiting host cells.
Sebastian Lourido, PhD, of the Whitehead Institute for Biomedical Research in Cambridge, Massachusetts, and his colleagues described this work in PNAS.
The researchers noted that the toxoplasmosis-causing parasite Toxoplasma gondii is closely related to the malaria-causing Plasmodium parasites. So research on T gondii can provide insights into Plasmodium’s inner workings.
For this study, Dr Lourido and his colleagues wanted to learn more about calcium-dependent protein kinases (CDPKs), enzymes that are needed for T gondii and related parasites to invade and exit host cells, move, and reproduce.
To investigate CDPKs, the team used single-domain antibody fragments derived from alpacas. Unlike humans, whose antibodies have a heavy chain and a light chain, alpacas create heavy-chain-only antibodies, which can be engineered into even smaller antibody fragments known as nanobodies.
Alpaca nanobodies have a unique shape that allows them to reach into a protein’s nooks and crannies, which are inaccessible to conventional antibodies.
The researchers identified a nanobody against the T gondii enzyme CDPK1 that binds the kinase’s regulatory domain and revealed a previously unappreciated feature of its activation.
The nanobody, called 1B7, stabilizes CDPK1 in a conformation that allowed the researchers to determine the kinase’s structure and describe the nanobody’s interaction with the molecule.
With the structure in hand, the team created long-timescale molecular dynamics simulations of the enzyme, to model the events leading to kinase inactivation.
Structural homology between CDPKs and the calmodulin-dependent kinases (CaMKs) found in humans led to earlier assumptions that both types of enzymes are activated in a similar fashion. But this new work shows otherwise.
A CaMK is activated when a wedge holding it in an inactive state is knocked away. In contrast, Dr Lourido likened a CDPK’s active conformation to a broken arm that must be splinted in two places to maintain its integrity.
When the rigid splint is removed, the kinase loses its structural ability to function. By blocking CDPK1’s regulatory domain, the 1B7 nanobody inhibits the kinase by preventing the enzyme’s “splint” from attaching.
“This work reveals something interesting about this class of enzymes,” Dr Lourido said. “It’s the first time a calcium-regulated kinase has been shown to be activated in this manner. The principle that we identify is really important. We’ve found a new vulnerability within an enzyme that we know is extremely important to this class of parasites, including Plasmodium . . . , and is absent from humans.”
Because humans lack similar kinases, drugs that target CDPKs would not affect host cells.
“The location where 1B7 binds to CDPK1 is a new drug target that people had not considered before,” said study author Jessica Ingram, PhD, also of the Whitehead Institute for Biomedical Research.
“We’d like to do some drug screens in the presence of the nanobody to see if we can find small molecules that bind in the same way. We could also look at other nanobodies against other kinases to see if this is applicable to other parasites and systems.” ![]()

Image by Ke Hu & John Murray
Researchers say they have determined the structure of an enzyme that is vital to the infectious behavior of the parasites that cause toxoplasmosis and malaria.
And this has revealed a potentially druggable target that could prevent the parasites from entering and exiting host cells.
Sebastian Lourido, PhD, of the Whitehead Institute for Biomedical Research in Cambridge, Massachusetts, and his colleagues described this work in PNAS.
The researchers noted that the toxoplasmosis-causing parasite Toxoplasma gondii is closely related to the malaria-causing Plasmodium parasites. So research on T gondii can provide insights into Plasmodium’s inner workings.
For this study, Dr Lourido and his colleagues wanted to learn more about calcium-dependent protein kinases (CDPKs), enzymes that are needed for T gondii and related parasites to invade and exit host cells, move, and reproduce.
To investigate CDPKs, the team used single-domain antibody fragments derived from alpacas. Unlike humans, whose antibodies have a heavy chain and a light chain, alpacas create heavy-chain-only antibodies, which can be engineered into even smaller antibody fragments known as nanobodies.
Alpaca nanobodies have a unique shape that allows them to reach into a protein’s nooks and crannies, which are inaccessible to conventional antibodies.
The researchers identified a nanobody against the T gondii enzyme CDPK1 that binds the kinase’s regulatory domain and revealed a previously unappreciated feature of its activation.
The nanobody, called 1B7, stabilizes CDPK1 in a conformation that allowed the researchers to determine the kinase’s structure and describe the nanobody’s interaction with the molecule.
With the structure in hand, the team created long-timescale molecular dynamics simulations of the enzyme, to model the events leading to kinase inactivation.
Structural homology between CDPKs and the calmodulin-dependent kinases (CaMKs) found in humans led to earlier assumptions that both types of enzymes are activated in a similar fashion. But this new work shows otherwise.
A CaMK is activated when a wedge holding it in an inactive state is knocked away. In contrast, Dr Lourido likened a CDPK’s active conformation to a broken arm that must be splinted in two places to maintain its integrity.
When the rigid splint is removed, the kinase loses its structural ability to function. By blocking CDPK1’s regulatory domain, the 1B7 nanobody inhibits the kinase by preventing the enzyme’s “splint” from attaching.
“This work reveals something interesting about this class of enzymes,” Dr Lourido said. “It’s the first time a calcium-regulated kinase has been shown to be activated in this manner. The principle that we identify is really important. We’ve found a new vulnerability within an enzyme that we know is extremely important to this class of parasites, including Plasmodium . . . , and is absent from humans.”
Because humans lack similar kinases, drugs that target CDPKs would not affect host cells.
“The location where 1B7 binds to CDPK1 is a new drug target that people had not considered before,” said study author Jessica Ingram, PhD, also of the Whitehead Institute for Biomedical Research.
“We’d like to do some drug screens in the presence of the nanobody to see if we can find small molecules that bind in the same way. We could also look at other nanobodies against other kinases to see if this is applicable to other parasites and systems.” ![]()

Image by Ke Hu & John Murray
Researchers say they have determined the structure of an enzyme that is vital to the infectious behavior of the parasites that cause toxoplasmosis and malaria.
And this has revealed a potentially druggable target that could prevent the parasites from entering and exiting host cells.
Sebastian Lourido, PhD, of the Whitehead Institute for Biomedical Research in Cambridge, Massachusetts, and his colleagues described this work in PNAS.
The researchers noted that the toxoplasmosis-causing parasite Toxoplasma gondii is closely related to the malaria-causing Plasmodium parasites. So research on T gondii can provide insights into Plasmodium’s inner workings.
For this study, Dr Lourido and his colleagues wanted to learn more about calcium-dependent protein kinases (CDPKs), enzymes that are needed for T gondii and related parasites to invade and exit host cells, move, and reproduce.
To investigate CDPKs, the team used single-domain antibody fragments derived from alpacas. Unlike humans, whose antibodies have a heavy chain and a light chain, alpacas create heavy-chain-only antibodies, which can be engineered into even smaller antibody fragments known as nanobodies.
Alpaca nanobodies have a unique shape that allows them to reach into a protein’s nooks and crannies, which are inaccessible to conventional antibodies.
The researchers identified a nanobody against the T gondii enzyme CDPK1 that binds the kinase’s regulatory domain and revealed a previously unappreciated feature of its activation.
The nanobody, called 1B7, stabilizes CDPK1 in a conformation that allowed the researchers to determine the kinase’s structure and describe the nanobody’s interaction with the molecule.
With the structure in hand, the team created long-timescale molecular dynamics simulations of the enzyme, to model the events leading to kinase inactivation.
Structural homology between CDPKs and the calmodulin-dependent kinases (CaMKs) found in humans led to earlier assumptions that both types of enzymes are activated in a similar fashion. But this new work shows otherwise.
A CaMK is activated when a wedge holding it in an inactive state is knocked away. In contrast, Dr Lourido likened a CDPK’s active conformation to a broken arm that must be splinted in two places to maintain its integrity.
When the rigid splint is removed, the kinase loses its structural ability to function. By blocking CDPK1’s regulatory domain, the 1B7 nanobody inhibits the kinase by preventing the enzyme’s “splint” from attaching.
“This work reveals something interesting about this class of enzymes,” Dr Lourido said. “It’s the first time a calcium-regulated kinase has been shown to be activated in this manner. The principle that we identify is really important. We’ve found a new vulnerability within an enzyme that we know is extremely important to this class of parasites, including Plasmodium . . . , and is absent from humans.”
Because humans lack similar kinases, drugs that target CDPKs would not affect host cells.
“The location where 1B7 binds to CDPK1 is a new drug target that people had not considered before,” said study author Jessica Ingram, PhD, also of the Whitehead Institute for Biomedical Research.
“We’d like to do some drug screens in the presence of the nanobody to see if we can find small molecules that bind in the same way. We could also look at other nanobodies against other kinases to see if this is applicable to other parasites and systems.” ![]()
Drug gets orphan designation for CDI
The US Food and Drug Administration (FDA) has granted orphan designation to SER-109 for the prevention of recurrent Clostridium difficile infection (CDI) in adults.
SER-109 is a microbiome therapeutic designed to treat recurrent CDI by correcting dysbiosis of the human microbiome.
In a single dose of 4 capsules, SER-109 re-introduces an ecology of purified bacterial spores that should restore the microbiome to a healthy state, allowing it to carry out key biological functions, including resisting Clostridium difficile.
“SER-109 is intended to re-introduce essential bacteria that restore the body’s natural resistance to CDI by re-establishing the ecology of the colonic microbiome,” explained Roger Pomerantz, MD, of Seres Therapeutics, Inc., the company developing SER-109.
“Because we’re focused on treating the underlying cause of the disease, we believe we have the potential to break the cycle of recurrent CDI and have a significant impact for patients.”
SER-109 is currently being investigated in a phase 2 trial. In addition to orphan designation, SER-109 has breakthrough designation from the FDA.
Trials of SER-109
Researchers reported phase 1/2 results with SER-109 at the 2014 Interscience Conference on Antimicrobial Agents and Chemotherapy.
The study had 2 cohorts containing 15 patients each. Patients were between 18 and 90 years old, had 3 or more laboratory-confirmed CDI episodes over 1 year, had a life expectancy greater than 3 months, and were able to give informed consent.
Patients in cohort 1 received a mean SER-109 dose of 1.5 x 109 spores, and those in cohort 2 received a mean dose of 1 x 108 spores. SER-109 was deemed effective if patients did not have a CDI recurrence in the 8-week period after they received SER-109.
In cohort 1, 87% of patients (13/15) achieved the efficacy endpoint. Two patients had transient, self-limited diarrhea with a positive C difficile test, but both reached the week 8 endpoint without needing antibiotic therapy for CDI. Thus, in cohort 1, the clinical cure rate was 100%.
In cohort 2, 93% of patients (14/15) reached the 8-week endpoint CDI-free. One patient failed per protocol.
The researchers said there were no drug-related serious adverse events in this trial.
Seres Therapeutics is currently conducting a multicenter, randomized, placebo-controlled, phase 2 study (ECOSPOR) to assess the efficacy and safety of SER-109 in preventing recurrent CDI. The company expects results from this study to be available mid-2016.
About orphan and breakthrough designation
The FDA grants orphan designation to drugs that are intended to treat diseases or conditions affecting fewer than 200,000 patients in the US.
Orphan designation provides the sponsor of a drug with various development incentives, including opportunities to apply for research-related tax credits and grant funding, assistance in designing clinical trials, and 7 years of US marketing exclusivity if the drug is approved.
The FDA’s breakthrough therapy designation is intended to expedite the development and review of a drug candidate intended to treat a serious or life-threatening condition.
The benefits of breakthrough designation include the same benefits as fast track designation—priority review of a new drug application, rolling review, etc.—plus an organizational commitment involving the FDA’s senior managers with more intensive guidance from the FDA. ![]()

The US Food and Drug Administration (FDA) has granted orphan designation to SER-109 for the prevention of recurrent Clostridium difficile infection (CDI) in adults.
SER-109 is a microbiome therapeutic designed to treat recurrent CDI by correcting dysbiosis of the human microbiome.
In a single dose of 4 capsules, SER-109 re-introduces an ecology of purified bacterial spores that should restore the microbiome to a healthy state, allowing it to carry out key biological functions, including resisting Clostridium difficile.
“SER-109 is intended to re-introduce essential bacteria that restore the body’s natural resistance to CDI by re-establishing the ecology of the colonic microbiome,” explained Roger Pomerantz, MD, of Seres Therapeutics, Inc., the company developing SER-109.
“Because we’re focused on treating the underlying cause of the disease, we believe we have the potential to break the cycle of recurrent CDI and have a significant impact for patients.”
SER-109 is currently being investigated in a phase 2 trial. In addition to orphan designation, SER-109 has breakthrough designation from the FDA.
Trials of SER-109
Researchers reported phase 1/2 results with SER-109 at the 2014 Interscience Conference on Antimicrobial Agents and Chemotherapy.
The study had 2 cohorts containing 15 patients each. Patients were between 18 and 90 years old, had 3 or more laboratory-confirmed CDI episodes over 1 year, had a life expectancy greater than 3 months, and were able to give informed consent.
Patients in cohort 1 received a mean SER-109 dose of 1.5 x 109 spores, and those in cohort 2 received a mean dose of 1 x 108 spores. SER-109 was deemed effective if patients did not have a CDI recurrence in the 8-week period after they received SER-109.
In cohort 1, 87% of patients (13/15) achieved the efficacy endpoint. Two patients had transient, self-limited diarrhea with a positive C difficile test, but both reached the week 8 endpoint without needing antibiotic therapy for CDI. Thus, in cohort 1, the clinical cure rate was 100%.
In cohort 2, 93% of patients (14/15) reached the 8-week endpoint CDI-free. One patient failed per protocol.
The researchers said there were no drug-related serious adverse events in this trial.
Seres Therapeutics is currently conducting a multicenter, randomized, placebo-controlled, phase 2 study (ECOSPOR) to assess the efficacy and safety of SER-109 in preventing recurrent CDI. The company expects results from this study to be available mid-2016.
About orphan and breakthrough designation
The FDA grants orphan designation to drugs that are intended to treat diseases or conditions affecting fewer than 200,000 patients in the US.
Orphan designation provides the sponsor of a drug with various development incentives, including opportunities to apply for research-related tax credits and grant funding, assistance in designing clinical trials, and 7 years of US marketing exclusivity if the drug is approved.
The FDA’s breakthrough therapy designation is intended to expedite the development and review of a drug candidate intended to treat a serious or life-threatening condition.
The benefits of breakthrough designation include the same benefits as fast track designation—priority review of a new drug application, rolling review, etc.—plus an organizational commitment involving the FDA’s senior managers with more intensive guidance from the FDA. ![]()

The US Food and Drug Administration (FDA) has granted orphan designation to SER-109 for the prevention of recurrent Clostridium difficile infection (CDI) in adults.
SER-109 is a microbiome therapeutic designed to treat recurrent CDI by correcting dysbiosis of the human microbiome.
In a single dose of 4 capsules, SER-109 re-introduces an ecology of purified bacterial spores that should restore the microbiome to a healthy state, allowing it to carry out key biological functions, including resisting Clostridium difficile.
“SER-109 is intended to re-introduce essential bacteria that restore the body’s natural resistance to CDI by re-establishing the ecology of the colonic microbiome,” explained Roger Pomerantz, MD, of Seres Therapeutics, Inc., the company developing SER-109.
“Because we’re focused on treating the underlying cause of the disease, we believe we have the potential to break the cycle of recurrent CDI and have a significant impact for patients.”
SER-109 is currently being investigated in a phase 2 trial. In addition to orphan designation, SER-109 has breakthrough designation from the FDA.
Trials of SER-109
Researchers reported phase 1/2 results with SER-109 at the 2014 Interscience Conference on Antimicrobial Agents and Chemotherapy.
The study had 2 cohorts containing 15 patients each. Patients were between 18 and 90 years old, had 3 or more laboratory-confirmed CDI episodes over 1 year, had a life expectancy greater than 3 months, and were able to give informed consent.
Patients in cohort 1 received a mean SER-109 dose of 1.5 x 109 spores, and those in cohort 2 received a mean dose of 1 x 108 spores. SER-109 was deemed effective if patients did not have a CDI recurrence in the 8-week period after they received SER-109.
In cohort 1, 87% of patients (13/15) achieved the efficacy endpoint. Two patients had transient, self-limited diarrhea with a positive C difficile test, but both reached the week 8 endpoint without needing antibiotic therapy for CDI. Thus, in cohort 1, the clinical cure rate was 100%.
In cohort 2, 93% of patients (14/15) reached the 8-week endpoint CDI-free. One patient failed per protocol.
The researchers said there were no drug-related serious adverse events in this trial.
Seres Therapeutics is currently conducting a multicenter, randomized, placebo-controlled, phase 2 study (ECOSPOR) to assess the efficacy and safety of SER-109 in preventing recurrent CDI. The company expects results from this study to be available mid-2016.
About orphan and breakthrough designation
The FDA grants orphan designation to drugs that are intended to treat diseases or conditions affecting fewer than 200,000 patients in the US.
Orphan designation provides the sponsor of a drug with various development incentives, including opportunities to apply for research-related tax credits and grant funding, assistance in designing clinical trials, and 7 years of US marketing exclusivity if the drug is approved.
The FDA’s breakthrough therapy designation is intended to expedite the development and review of a drug candidate intended to treat a serious or life-threatening condition.
The benefits of breakthrough designation include the same benefits as fast track designation—priority review of a new drug application, rolling review, etc.—plus an organizational commitment involving the FDA’s senior managers with more intensive guidance from the FDA. ![]()
Pathway appears key to fighting adenovirus

Using an animal model they developed, researchers have identified a pathway that inhibits replication of the adenovirus.
The team generated a new strain of Syrian hamster, a model in which human adenovirus replicates and causes illness similar to that observed in humans.
Experiments with this model suggested the Type I interferon pathway plays a key role in inhibiting adenovirus replication.
“[L]ike many other viruses, adenovirus can replicate at will when a patient’s immune system is suppressed,” said William Wold, PhD, of Saint Louis University in Missouri.
“Adenovirus can become very dangerous, such as for a child who is undergoing a bone marrow transplant to treat leukemia.”
Previously, Dr Wold led a research team that identified the Syrian hamster as an appropriate animal model to study adenovirus because species C human adenoviruses replicate in these animals.
For the current study, which was published in PLOS Pathogens, Dr Wold and his colleagues conducted experiments with a new Syrian hamster strain. In these animals, the STAT2 gene was functionally knocked out by site-specific gene targeting.
The researchers found that STAT2-knockout hamsters were extremely sensitive to infection with type 5 human adenovirus (Ad5).
The team infected both STAT2-knockout hamsters and wild-type controls with Ad5. Knockout hamsters had 100 to 1000 times the viral load of controls.
The knockout hamsters also had pathology characteristic of advanced adenovirus infection—yellow, mottled livers and enlarged gall bladders—whereas controls did not.
The adaptive immune response to Ad5 remained intact in the STAT2-knockout hamsters, as surviving animals were able to clear the virus.
However, the Type 1 interferon response was hampered in these animals. Knocking out STAT2 disrupted the Type 1 interferon pathway by interrupting the cascade of cell signaling.
The researchers said their findings suggest the disrupted Type I interferon pathway contributed to the increased Ad5 replication in the STAT2-knockout hamsters.
“Besides providing an insight into adenovirus infection in humans, our results are also interesting from the perspective of the animal model,” Dr Wold said. “The STAT2-knockout Syrian hamster may also be an important animal model for studying other viral infections, including Ebola, hanta, and dengue viruses.”
The model was created by Zhongde Wang, PhD, and his colleagues at Utah State University in Logan, Utah. Dr Wang’s lab is the first to develop gene-targeting technologies in the Syrian hamster.
“The success we achieved in conducting gene-targeting in the Syrian hamster has provided the opportunity to create models for many of the human diseases for which there are either no existent animal models or severe limitations in the available animal models,” Dr Wang said. ![]()

Using an animal model they developed, researchers have identified a pathway that inhibits replication of the adenovirus.
The team generated a new strain of Syrian hamster, a model in which human adenovirus replicates and causes illness similar to that observed in humans.
Experiments with this model suggested the Type I interferon pathway plays a key role in inhibiting adenovirus replication.
“[L]ike many other viruses, adenovirus can replicate at will when a patient’s immune system is suppressed,” said William Wold, PhD, of Saint Louis University in Missouri.
“Adenovirus can become very dangerous, such as for a child who is undergoing a bone marrow transplant to treat leukemia.”
Previously, Dr Wold led a research team that identified the Syrian hamster as an appropriate animal model to study adenovirus because species C human adenoviruses replicate in these animals.
For the current study, which was published in PLOS Pathogens, Dr Wold and his colleagues conducted experiments with a new Syrian hamster strain. In these animals, the STAT2 gene was functionally knocked out by site-specific gene targeting.
The researchers found that STAT2-knockout hamsters were extremely sensitive to infection with type 5 human adenovirus (Ad5).
The team infected both STAT2-knockout hamsters and wild-type controls with Ad5. Knockout hamsters had 100 to 1000 times the viral load of controls.
The knockout hamsters also had pathology characteristic of advanced adenovirus infection—yellow, mottled livers and enlarged gall bladders—whereas controls did not.
The adaptive immune response to Ad5 remained intact in the STAT2-knockout hamsters, as surviving animals were able to clear the virus.
However, the Type 1 interferon response was hampered in these animals. Knocking out STAT2 disrupted the Type 1 interferon pathway by interrupting the cascade of cell signaling.
The researchers said their findings suggest the disrupted Type I interferon pathway contributed to the increased Ad5 replication in the STAT2-knockout hamsters.
“Besides providing an insight into adenovirus infection in humans, our results are also interesting from the perspective of the animal model,” Dr Wold said. “The STAT2-knockout Syrian hamster may also be an important animal model for studying other viral infections, including Ebola, hanta, and dengue viruses.”
The model was created by Zhongde Wang, PhD, and his colleagues at Utah State University in Logan, Utah. Dr Wang’s lab is the first to develop gene-targeting technologies in the Syrian hamster.
“The success we achieved in conducting gene-targeting in the Syrian hamster has provided the opportunity to create models for many of the human diseases for which there are either no existent animal models or severe limitations in the available animal models,” Dr Wang said. ![]()

Using an animal model they developed, researchers have identified a pathway that inhibits replication of the adenovirus.
The team generated a new strain of Syrian hamster, a model in which human adenovirus replicates and causes illness similar to that observed in humans.
Experiments with this model suggested the Type I interferon pathway plays a key role in inhibiting adenovirus replication.
“[L]ike many other viruses, adenovirus can replicate at will when a patient’s immune system is suppressed,” said William Wold, PhD, of Saint Louis University in Missouri.
“Adenovirus can become very dangerous, such as for a child who is undergoing a bone marrow transplant to treat leukemia.”
Previously, Dr Wold led a research team that identified the Syrian hamster as an appropriate animal model to study adenovirus because species C human adenoviruses replicate in these animals.
For the current study, which was published in PLOS Pathogens, Dr Wold and his colleagues conducted experiments with a new Syrian hamster strain. In these animals, the STAT2 gene was functionally knocked out by site-specific gene targeting.
The researchers found that STAT2-knockout hamsters were extremely sensitive to infection with type 5 human adenovirus (Ad5).
The team infected both STAT2-knockout hamsters and wild-type controls with Ad5. Knockout hamsters had 100 to 1000 times the viral load of controls.
The knockout hamsters also had pathology characteristic of advanced adenovirus infection—yellow, mottled livers and enlarged gall bladders—whereas controls did not.
The adaptive immune response to Ad5 remained intact in the STAT2-knockout hamsters, as surviving animals were able to clear the virus.
However, the Type 1 interferon response was hampered in these animals. Knocking out STAT2 disrupted the Type 1 interferon pathway by interrupting the cascade of cell signaling.
The researchers said their findings suggest the disrupted Type I interferon pathway contributed to the increased Ad5 replication in the STAT2-knockout hamsters.
“Besides providing an insight into adenovirus infection in humans, our results are also interesting from the perspective of the animal model,” Dr Wold said. “The STAT2-knockout Syrian hamster may also be an important animal model for studying other viral infections, including Ebola, hanta, and dengue viruses.”
The model was created by Zhongde Wang, PhD, and his colleagues at Utah State University in Logan, Utah. Dr Wang’s lab is the first to develop gene-targeting technologies in the Syrian hamster.
“The success we achieved in conducting gene-targeting in the Syrian hamster has provided the opportunity to create models for many of the human diseases for which there are either no existent animal models or severe limitations in the available animal models,” Dr Wang said.
Survey reveals inconsistency in infection prevention practices
in the intensive care unit
Results of an anonymous survey suggest healthcare professionals in the US may not consistently follow recommendations for preventing bloodstream infections in patients with arterial catheters.
Of the roughly 1000 critical care clinicians surveyed, fewer than half said they comply with the current Centers for Disease Control and Prevention (CDC) guidelines, which recommend the use of limited barrier precautions during arterial catheter insertion.
This includes sterile gloves, a surgical cap, a surgical mask, and a small sterile drape.
“Barrier precautions are employed inconsistently by critical care clinicians across the nation, and such individuals underestimate the infection risks posed by arterial catheters,” said Leonard A. Mermel, DO, of Rhode Island Hospital in Providence.
He and his colleagues reported these findings in Critical Care Medicine.
The researchers sent an anonymous, web-based survey to 11,361 physicians, nurse practitioners, physician assistants, respiratory therapists, and registered nurses who receive emails from the Society of Critical Care Medicine.
There were 1265 responses (an 11% response rate) and 1029 eligible participants after exclusions.
Of the eligible respondents, 44% said they used CDC-recommended barrier precautions during arterial catheter insertion, and 15% said they use full barrier precautions.
However, 39% of respondents said they would support mandatory use of full barrier precautions during arterial catheter insertion.
“There appears to be a significant deviation from clinical guidelines regarding a very commonly performed procedure in critically ill patients,” said study author Andrew Levinson, MD, also of Rhode Island Hospital.
“Bloodstream infections are largely preventable, and if the survey results mirror the clinical practice in the US, there’s work to be done in reducing risk of such infections.”
The survey also indicated that respondents underestimate the risk of bloodstream infections associated with arterial catheters. The respondents’ mean estimate of infection incidence was 0.3 per 1000 catheter-days, and the median estimate was 0.1 per 1000 catheter-days.
However, Dr Mermel and his colleagues said recent studies have suggested the incidence of bloodstream infections associated with arterial catheters in the US is 0.9 to 3.4 per 1000 catheter-days.

in the intensive care unit
Results of an anonymous survey suggest healthcare professionals in the US may not consistently follow recommendations for preventing bloodstream infections in patients with arterial catheters.
Of the roughly 1000 critical care clinicians surveyed, fewer than half said they comply with the current Centers for Disease Control and Prevention (CDC) guidelines, which recommend the use of limited barrier precautions during arterial catheter insertion.
This includes sterile gloves, a surgical cap, a surgical mask, and a small sterile drape.
“Barrier precautions are employed inconsistently by critical care clinicians across the nation, and such individuals underestimate the infection risks posed by arterial catheters,” said Leonard A. Mermel, DO, of Rhode Island Hospital in Providence.
He and his colleagues reported these findings in Critical Care Medicine.
The researchers sent an anonymous, web-based survey to 11,361 physicians, nurse practitioners, physician assistants, respiratory therapists, and registered nurses who receive emails from the Society of Critical Care Medicine.
There were 1265 responses (an 11% response rate) and 1029 eligible participants after exclusions.
Of the eligible respondents, 44% said they used CDC-recommended barrier precautions during arterial catheter insertion, and 15% said they use full barrier precautions.
However, 39% of respondents said they would support mandatory use of full barrier precautions during arterial catheter insertion.
“There appears to be a significant deviation from clinical guidelines regarding a very commonly performed procedure in critically ill patients,” said study author Andrew Levinson, MD, also of Rhode Island Hospital.
“Bloodstream infections are largely preventable, and if the survey results mirror the clinical practice in the US, there’s work to be done in reducing risk of such infections.”
The survey also indicated that respondents underestimate the risk of bloodstream infections associated with arterial catheters. The respondents’ mean estimate of infection incidence was 0.3 per 1000 catheter-days, and the median estimate was 0.1 per 1000 catheter-days.
However, Dr Mermel and his colleagues said recent studies have suggested the incidence of bloodstream infections associated with arterial catheters in the US is 0.9 to 3.4 per 1000 catheter-days.

in the intensive care unit
Results of an anonymous survey suggest healthcare professionals in the US may not consistently follow recommendations for preventing bloodstream infections in patients with arterial catheters.
Of the roughly 1000 critical care clinicians surveyed, fewer than half said they comply with the current Centers for Disease Control and Prevention (CDC) guidelines, which recommend the use of limited barrier precautions during arterial catheter insertion.
This includes sterile gloves, a surgical cap, a surgical mask, and a small sterile drape.
“Barrier precautions are employed inconsistently by critical care clinicians across the nation, and such individuals underestimate the infection risks posed by arterial catheters,” said Leonard A. Mermel, DO, of Rhode Island Hospital in Providence.
He and his colleagues reported these findings in Critical Care Medicine.
The researchers sent an anonymous, web-based survey to 11,361 physicians, nurse practitioners, physician assistants, respiratory therapists, and registered nurses who receive emails from the Society of Critical Care Medicine.
There were 1265 responses (an 11% response rate) and 1029 eligible participants after exclusions.
Of the eligible respondents, 44% said they used CDC-recommended barrier precautions during arterial catheter insertion, and 15% said they use full barrier precautions.
However, 39% of respondents said they would support mandatory use of full barrier precautions during arterial catheter insertion.
“There appears to be a significant deviation from clinical guidelines regarding a very commonly performed procedure in critically ill patients,” said study author Andrew Levinson, MD, also of Rhode Island Hospital.
“Bloodstream infections are largely preventable, and if the survey results mirror the clinical practice in the US, there’s work to be done in reducing risk of such infections.”
The survey also indicated that respondents underestimate the risk of bloodstream infections associated with arterial catheters. The respondents’ mean estimate of infection incidence was 0.3 per 1000 catheter-days, and the median estimate was 0.1 per 1000 catheter-days.
However, Dr Mermel and his colleagues said recent studies have suggested the incidence of bloodstream infections associated with arterial catheters in the US is 0.9 to 3.4 per 1000 catheter-days.
Blood-cleansing device on the way to the clinic, team says

for Staphylococcus infection
Photo by Bill Branson
Last year, researchers reported promising preclinical results with a device that can treat sepsis by mimicking the human spleen. The device filtered pathogens and toxins from the blood by passing it through a dialysis-like circuit.
Now, the researchers have developed a new, more streamlined device that, they believe, is more likely to translate to the clinic. The new device also synergizes with conventional antibiotic therapies.
The team described this device in Biomaterials.
“The inflammatory cascade that leads to sepsis is triggered by pathogens and, specifically, by the toxins they release,” said study author Donald Ingber, MD, PhD, of the Wyss Institute for Biologically Inspired Engineering at Harvard University in Cambridge, Massachusetts.
“Thus, the most effective strategy is to treat with the best antibiotics you can muster, while also removing the toxins and remaining pathogens from the patient’s blood as quickly as possible.”
How the device works
The researchers say their new blood-cleansing approach can be completed quickly, without even identifying the infectious agent. This is because the device uses a proprietary pathogen-capturing agent, known as FcMBL, that binds all types of live and dead infectious microbes, including bacteria, fungi, viruses, and the toxins they release.
FcMBL is a genetically engineered blood protein inspired by a naturally occurring human molecule called mannose binding lectin (MBL). MBL is found in the innate immune system and binds to toxic invaders, marking them for capture by immune cells in the spleen.
The researchers’ original device concept was similar to how a dialysis machine works. Infected blood in an animal, or potentially one day in a patient, is flowed from one vein through catheters to the device.
There, FcMBL-coated magnetic beads are added to the blood, and the bead-bound pathogens are extracted from the circulating blood by magnets within the device before the cleansed blood is returned to the body through another vein.
The new device removes the complexity, regulatory challenges, and cost associated with the magnetic beads and microfluidic architecture of its predecessor. But it still uses the FcMBL protein to bind to pathogens and toxins.
The new system uses hollow fiber filters similar to those currently used in hemodialysis, but the inner walls of these filters are coated with FcMBL protein to remove pathogens from circulating blood.
Test results
In in vitro tests with human blood, the device reduced the number of Gram-negative bacteria (Escherichia coli), Gram-positive bacteria (Staphylococcus aureus), fungi (Candida albicans), and lipopolysaccharide-endotoxins by 90% to 99%.
In tests with rats, the device reduced levels of circulating pathogens and endotoxins by more than 99%, and it prevented pathogen engraftment and inflammatory cell recruitment in the spleen, lung, liver, and kidney.
The researchers also tested the device in combination with bacteriocidal antibiotics in rats. The antibiotics prompted a “major increase” in the release of microbial fragments, or pathogen-associated molecular patterns (PAMPs).
The device could remove PAMPs from the blood within 2 hours, but high levels of PAMPs remained in rats treated with antibiotics alone.
The researchers said PAMP removal reduced organ pathogen and endotoxin loads, suppressed inflammatory responses, and resulted in more stable vital signs.
“Using the device, alone or alongside antibiotics, we can quickly bring blood back to normal conditions, curtailing an inflammatory response rather than exacerbating it,” said Tohid Fatanat Didar, PhD, of the Wyss Institute.
“If all goes well, physicians will someday be able to use the device in tandem with standard antibiotic treatments to deliver a one-two punch to pathogens, synergistically killing and cleansing all live and dead invaders from the bloodstream.”
As the device has proven effective in small animal experiments, the researchers are planning to move to large animal studies. They must demonstrate proof-of-concept in these models before advancing to clinical trials.
“Our goal is to see this move out of the lab and into hospitals, as well as onto the battlefield,” Dr Ingber said, “where it can save lives within years rather than decades.”

for Staphylococcus infection
Photo by Bill Branson
Last year, researchers reported promising preclinical results with a device that can treat sepsis by mimicking the human spleen. The device filtered pathogens and toxins from the blood by passing it through a dialysis-like circuit.
Now, the researchers have developed a new, more streamlined device that, they believe, is more likely to translate to the clinic. The new device also synergizes with conventional antibiotic therapies.
The team described this device in Biomaterials.
“The inflammatory cascade that leads to sepsis is triggered by pathogens and, specifically, by the toxins they release,” said study author Donald Ingber, MD, PhD, of the Wyss Institute for Biologically Inspired Engineering at Harvard University in Cambridge, Massachusetts.
“Thus, the most effective strategy is to treat with the best antibiotics you can muster, while also removing the toxins and remaining pathogens from the patient’s blood as quickly as possible.”
How the device works
The researchers say their new blood-cleansing approach can be completed quickly, without even identifying the infectious agent. This is because the device uses a proprietary pathogen-capturing agent, known as FcMBL, that binds all types of live and dead infectious microbes, including bacteria, fungi, viruses, and the toxins they release.
FcMBL is a genetically engineered blood protein inspired by a naturally occurring human molecule called mannose binding lectin (MBL). MBL is found in the innate immune system and binds to toxic invaders, marking them for capture by immune cells in the spleen.
The researchers’ original device concept was similar to how a dialysis machine works. Infected blood in an animal, or potentially one day in a patient, is flowed from one vein through catheters to the device.
There, FcMBL-coated magnetic beads are added to the blood, and the bead-bound pathogens are extracted from the circulating blood by magnets within the device before the cleansed blood is returned to the body through another vein.
The new device removes the complexity, regulatory challenges, and cost associated with the magnetic beads and microfluidic architecture of its predecessor. But it still uses the FcMBL protein to bind to pathogens and toxins.
The new system uses hollow fiber filters similar to those currently used in hemodialysis, but the inner walls of these filters are coated with FcMBL protein to remove pathogens from circulating blood.
Test results
In in vitro tests with human blood, the device reduced the number of Gram-negative bacteria (Escherichia coli), Gram-positive bacteria (Staphylococcus aureus), fungi (Candida albicans), and lipopolysaccharide-endotoxins by 90% to 99%.
In tests with rats, the device reduced levels of circulating pathogens and endotoxins by more than 99%, and it prevented pathogen engraftment and inflammatory cell recruitment in the spleen, lung, liver, and kidney.
The researchers also tested the device in combination with bacteriocidal antibiotics in rats. The antibiotics prompted a “major increase” in the release of microbial fragments, or pathogen-associated molecular patterns (PAMPs).
The device could remove PAMPs from the blood within 2 hours, but high levels of PAMPs remained in rats treated with antibiotics alone.
The researchers said PAMP removal reduced organ pathogen and endotoxin loads, suppressed inflammatory responses, and resulted in more stable vital signs.
“Using the device, alone or alongside antibiotics, we can quickly bring blood back to normal conditions, curtailing an inflammatory response rather than exacerbating it,” said Tohid Fatanat Didar, PhD, of the Wyss Institute.
“If all goes well, physicians will someday be able to use the device in tandem with standard antibiotic treatments to deliver a one-two punch to pathogens, synergistically killing and cleansing all live and dead invaders from the bloodstream.”
As the device has proven effective in small animal experiments, the researchers are planning to move to large animal studies. They must demonstrate proof-of-concept in these models before advancing to clinical trials.
“Our goal is to see this move out of the lab and into hospitals, as well as onto the battlefield,” Dr Ingber said, “where it can save lives within years rather than decades.”

for Staphylococcus infection
Photo by Bill Branson
Last year, researchers reported promising preclinical results with a device that can treat sepsis by mimicking the human spleen. The device filtered pathogens and toxins from the blood by passing it through a dialysis-like circuit.
Now, the researchers have developed a new, more streamlined device that, they believe, is more likely to translate to the clinic. The new device also synergizes with conventional antibiotic therapies.
The team described this device in Biomaterials.
“The inflammatory cascade that leads to sepsis is triggered by pathogens and, specifically, by the toxins they release,” said study author Donald Ingber, MD, PhD, of the Wyss Institute for Biologically Inspired Engineering at Harvard University in Cambridge, Massachusetts.
“Thus, the most effective strategy is to treat with the best antibiotics you can muster, while also removing the toxins and remaining pathogens from the patient’s blood as quickly as possible.”
How the device works
The researchers say their new blood-cleansing approach can be completed quickly, without even identifying the infectious agent. This is because the device uses a proprietary pathogen-capturing agent, known as FcMBL, that binds all types of live and dead infectious microbes, including bacteria, fungi, viruses, and the toxins they release.
FcMBL is a genetically engineered blood protein inspired by a naturally occurring human molecule called mannose binding lectin (MBL). MBL is found in the innate immune system and binds to toxic invaders, marking them for capture by immune cells in the spleen.
The researchers’ original device concept was similar to how a dialysis machine works. Infected blood in an animal, or potentially one day in a patient, is flowed from one vein through catheters to the device.
There, FcMBL-coated magnetic beads are added to the blood, and the bead-bound pathogens are extracted from the circulating blood by magnets within the device before the cleansed blood is returned to the body through another vein.
The new device removes the complexity, regulatory challenges, and cost associated with the magnetic beads and microfluidic architecture of its predecessor. But it still uses the FcMBL protein to bind to pathogens and toxins.
The new system uses hollow fiber filters similar to those currently used in hemodialysis, but the inner walls of these filters are coated with FcMBL protein to remove pathogens from circulating blood.
Test results
In in vitro tests with human blood, the device reduced the number of Gram-negative bacteria (Escherichia coli), Gram-positive bacteria (Staphylococcus aureus), fungi (Candida albicans), and lipopolysaccharide-endotoxins by 90% to 99%.
In tests with rats, the device reduced levels of circulating pathogens and endotoxins by more than 99%, and it prevented pathogen engraftment and inflammatory cell recruitment in the spleen, lung, liver, and kidney.
The researchers also tested the device in combination with bacteriocidal antibiotics in rats. The antibiotics prompted a “major increase” in the release of microbial fragments, or pathogen-associated molecular patterns (PAMPs).
The device could remove PAMPs from the blood within 2 hours, but high levels of PAMPs remained in rats treated with antibiotics alone.
The researchers said PAMP removal reduced organ pathogen and endotoxin loads, suppressed inflammatory responses, and resulted in more stable vital signs.
“Using the device, alone or alongside antibiotics, we can quickly bring blood back to normal conditions, curtailing an inflammatory response rather than exacerbating it,” said Tohid Fatanat Didar, PhD, of the Wyss Institute.
“If all goes well, physicians will someday be able to use the device in tandem with standard antibiotic treatments to deliver a one-two punch to pathogens, synergistically killing and cleansing all live and dead invaders from the bloodstream.”
As the device has proven effective in small animal experiments, the researchers are planning to move to large animal studies. They must demonstrate proof-of-concept in these models before advancing to clinical trials.
“Our goal is to see this move out of the lab and into hospitals, as well as onto the battlefield,” Dr Ingber said, “where it can save lives within years rather than decades.”
T-cell finding may have broad implications

resolution microscope
Image courtesy of UNSW
Early exposure to inflammatory cytokines can “paralyze” CD4 T cells, according to research published in Immunity.
The study suggests this mechanism may act as a firewall, shutting down the immune response before it gets out of hand.
According to researchers, this discovery could lead to more effective immunotherapies for cancer and reduce the need for immunosuppressants in transplant patients, among other applications.
“There’s a 3-signal process to activate T cells, of which each component is essential for proper activation,” said study author Gail Sckisel, PhD, of the University of California, Davis in Sacramento.
“But no one had really looked at what happens if they are delivered out of sequence. If the third signal—cytokines—is given prematurely, it basically paralyzes CD4 T cells.”
To be activated, T cells must first recognize an antigen, receive appropriate costimulatory signals, and then encounter inflammatory cytokines to expand the immune response. Until now, no one realized that sending the third signal early—as is done with some immunotherapies—could actually hamper overall immunity.
“These stimulatory immunotherapies are designed to activate the immune system,” Dr Sckisel explained. “But considering how T cells respond, that approach could damage a patient’s ability to fight off pathogens. While immunotherapies might fight cancer, they may also open the door to opportunistic infections.”
She and her colleagues demonstrated this principle in mice. After they received systemic immunotherapy, the animals had trouble mounting a primary T-cell response.
The researchers confirmed this finding in samples from patients receiving high-dose interleukin 2 to treat metastatic melanoma.
“We need to be very careful because immunotherapy could be generating both short-term gain and long-term loss,” said study author William Murphy, PhD, also of the University of California, Davis.
“The patients who were receiving immunotherapy were totally shut down, which shows how profoundly we were suppressing the immune system.”
In addition to illuminating how T cells respond to cancer immunotherapy, the study also provides insights into autoimmune disorders. The researchers believe this CD4 paralysis mechanism could play a role in preventing autoimmunity, a hypothesis they supported by testing immunotherapy in a multiple sclerosis model.
By shutting down CD4 T cells, immune stimulation prevented an autoimmune response. This provides the opportunity to paralyze the immune system to prevent autoimmunity or modulate it to accept transplanted cells or entire organs.
“Transplant patients go on immunosuppressants for the rest of their lives, but if we could safely induce paralysis just prior to surgery, it’s possible that patients could develop tolerance,” Dr Sckisel said.
CD4 paralysis may also be co-opted by pathogens, such as HIV, which could use this chronic inflammation response to disable the immune system.
“This really highlights the importance of CD4 T cells,” Dr Murphy said. “The fact that they’re regulated and suppressed means they are definitely the orchestrators we need to take into account. It also shows how smart HIV is. The virus has been telling us CD4 T cells are critical because that’s what it attacks.”
The team’s next step is to continue this research in older mice. Age can bring a measurable loss in immune function, and inflammation may play a role in that process.
“For elderly people who have flu or pneumonia, their immune systems are activated, but maybe they can’t fight anything else,” Dr Murphy said. “This could change how we treat people who are very sick. If we can block pathways that suppress the immune response, we may be able to better fight infection.”

resolution microscope
Image courtesy of UNSW
Early exposure to inflammatory cytokines can “paralyze” CD4 T cells, according to research published in Immunity.
The study suggests this mechanism may act as a firewall, shutting down the immune response before it gets out of hand.
According to researchers, this discovery could lead to more effective immunotherapies for cancer and reduce the need for immunosuppressants in transplant patients, among other applications.
“There’s a 3-signal process to activate T cells, of which each component is essential for proper activation,” said study author Gail Sckisel, PhD, of the University of California, Davis in Sacramento.
“But no one had really looked at what happens if they are delivered out of sequence. If the third signal—cytokines—is given prematurely, it basically paralyzes CD4 T cells.”
To be activated, T cells must first recognize an antigen, receive appropriate costimulatory signals, and then encounter inflammatory cytokines to expand the immune response. Until now, no one realized that sending the third signal early—as is done with some immunotherapies—could actually hamper overall immunity.
“These stimulatory immunotherapies are designed to activate the immune system,” Dr Sckisel explained. “But considering how T cells respond, that approach could damage a patient’s ability to fight off pathogens. While immunotherapies might fight cancer, they may also open the door to opportunistic infections.”
She and her colleagues demonstrated this principle in mice. After they received systemic immunotherapy, the animals had trouble mounting a primary T-cell response.
The researchers confirmed this finding in samples from patients receiving high-dose interleukin 2 to treat metastatic melanoma.
“We need to be very careful because immunotherapy could be generating both short-term gain and long-term loss,” said study author William Murphy, PhD, also of the University of California, Davis.
“The patients who were receiving immunotherapy were totally shut down, which shows how profoundly we were suppressing the immune system.”
In addition to illuminating how T cells respond to cancer immunotherapy, the study also provides insights into autoimmune disorders. The researchers believe this CD4 paralysis mechanism could play a role in preventing autoimmunity, a hypothesis they supported by testing immunotherapy in a multiple sclerosis model.
By shutting down CD4 T cells, immune stimulation prevented an autoimmune response. This provides the opportunity to paralyze the immune system to prevent autoimmunity or modulate it to accept transplanted cells or entire organs.
“Transplant patients go on immunosuppressants for the rest of their lives, but if we could safely induce paralysis just prior to surgery, it’s possible that patients could develop tolerance,” Dr Sckisel said.
CD4 paralysis may also be co-opted by pathogens, such as HIV, which could use this chronic inflammation response to disable the immune system.
“This really highlights the importance of CD4 T cells,” Dr Murphy said. “The fact that they’re regulated and suppressed means they are definitely the orchestrators we need to take into account. It also shows how smart HIV is. The virus has been telling us CD4 T cells are critical because that’s what it attacks.”
The team’s next step is to continue this research in older mice. Age can bring a measurable loss in immune function, and inflammation may play a role in that process.
“For elderly people who have flu or pneumonia, their immune systems are activated, but maybe they can’t fight anything else,” Dr Murphy said. “This could change how we treat people who are very sick. If we can block pathways that suppress the immune response, we may be able to better fight infection.”

resolution microscope
Image courtesy of UNSW
Early exposure to inflammatory cytokines can “paralyze” CD4 T cells, according to research published in Immunity.
The study suggests this mechanism may act as a firewall, shutting down the immune response before it gets out of hand.
According to researchers, this discovery could lead to more effective immunotherapies for cancer and reduce the need for immunosuppressants in transplant patients, among other applications.
“There’s a 3-signal process to activate T cells, of which each component is essential for proper activation,” said study author Gail Sckisel, PhD, of the University of California, Davis in Sacramento.
“But no one had really looked at what happens if they are delivered out of sequence. If the third signal—cytokines—is given prematurely, it basically paralyzes CD4 T cells.”
To be activated, T cells must first recognize an antigen, receive appropriate costimulatory signals, and then encounter inflammatory cytokines to expand the immune response. Until now, no one realized that sending the third signal early—as is done with some immunotherapies—could actually hamper overall immunity.
“These stimulatory immunotherapies are designed to activate the immune system,” Dr Sckisel explained. “But considering how T cells respond, that approach could damage a patient’s ability to fight off pathogens. While immunotherapies might fight cancer, they may also open the door to opportunistic infections.”
She and her colleagues demonstrated this principle in mice. After they received systemic immunotherapy, the animals had trouble mounting a primary T-cell response.
The researchers confirmed this finding in samples from patients receiving high-dose interleukin 2 to treat metastatic melanoma.
“We need to be very careful because immunotherapy could be generating both short-term gain and long-term loss,” said study author William Murphy, PhD, also of the University of California, Davis.
“The patients who were receiving immunotherapy were totally shut down, which shows how profoundly we were suppressing the immune system.”
In addition to illuminating how T cells respond to cancer immunotherapy, the study also provides insights into autoimmune disorders. The researchers believe this CD4 paralysis mechanism could play a role in preventing autoimmunity, a hypothesis they supported by testing immunotherapy in a multiple sclerosis model.
By shutting down CD4 T cells, immune stimulation prevented an autoimmune response. This provides the opportunity to paralyze the immune system to prevent autoimmunity or modulate it to accept transplanted cells or entire organs.
“Transplant patients go on immunosuppressants for the rest of their lives, but if we could safely induce paralysis just prior to surgery, it’s possible that patients could develop tolerance,” Dr Sckisel said.
CD4 paralysis may also be co-opted by pathogens, such as HIV, which could use this chronic inflammation response to disable the immune system.
“This really highlights the importance of CD4 T cells,” Dr Murphy said. “The fact that they’re regulated and suppressed means they are definitely the orchestrators we need to take into account. It also shows how smart HIV is. The virus has been telling us CD4 T cells are critical because that’s what it attacks.”
The team’s next step is to continue this research in older mice. Age can bring a measurable loss in immune function, and inflammation may play a role in that process.
“For elderly people who have flu or pneumonia, their immune systems are activated, but maybe they can’t fight anything else,” Dr Murphy said. “This could change how we treat people who are very sick. If we can block pathways that suppress the immune response, we may be able to better fight infection.”