User login
Cancer survivors mirror spouses’ moods

chemotherapy
Photo by Rhoda Baer
Cancer survivors’ moods are impacted—both positively and negatively—by their spouses’ moods, according to research published in Cancer Epidemiology, Biomarkers & Prevention.
In the study, cancer survivors whose spouses reported depressed moods were more likely to be depressed after about a year of follow-up, and survivors whose spouses reported better mental and physical health-related quality of life (HRQOL) were less likely to be depressed.
However, survivors’ moods did not have the same impact on their spouses.
“We were surprised that the effects of the spouses on the survivors were so much larger in this study than the effect of the survivors on their spouses,” said study author Kristin Litzelman, PhD, of the National Cancer Institute in Bethesda, Maryland. “We expected to see a more reciprocal relationship.”
Dr Litzelman and her colleagues conducted this research in an attempt to understand how cancer survivors and their families influence one another. The team hoped to identify ways to improve the healthcare both parties receive and thereby improve their health and well-being.
The researchers analyzed data from 910 cancer patients and their spouses, comparing them to 910 couples without any kind of cancer-related health problem.
The team used statistical models to assess how each spouse’s quality of life or depression at one time point was associated with his or her partner’s risk of depression around 11 months later. The researchers took into account a person’s previously reported mood, demographic characteristics, and other factors.
The results showed that, when spouses reported feeling depressed, cancer survivors were about 4 times more likely to report being depressed 11 months later (odds ratio [OR]=4.27). This association was stronger among female cancer survivors (OR=9.49) than male survivors (OR=3.98).
Cancer survivors whose spouses reported better HRQOL had a 30% decrease in depressed mood per 10-point improvement in HRQOL score. The ORs were 0.72 for mental health and 0.68 for physical health. The associations between spousal HRQOL and survivor depressed mood were similar for male and female survivors.
The researchers noted that cancer survivors’ moods did not have a significant impact on their spouses’ risk of depressed mood 11 months later.
And the team did not see mood associations in couples without any cancer-related health problems.
“This finding certainly needs to be backed up by other studies, but it highlights the importance of family well-being in cancer survivor outcomes,” Dr Litzelman said. “Our research highlights that spouses need to take care of themselves, not just for their own sake, but also for the sake of the cancer survivor.”
“Our findings also suggest that, when caring for cancer survivors, clinicians may want to assess the well-being of spousal caregivers. Future research could test whether including caregivers in the survivorship care plan might help to improve outcomes for both caregivers and for cancer survivors.” ![]()

chemotherapy
Photo by Rhoda Baer
Cancer survivors’ moods are impacted—both positively and negatively—by their spouses’ moods, according to research published in Cancer Epidemiology, Biomarkers & Prevention.
In the study, cancer survivors whose spouses reported depressed moods were more likely to be depressed after about a year of follow-up, and survivors whose spouses reported better mental and physical health-related quality of life (HRQOL) were less likely to be depressed.
However, survivors’ moods did not have the same impact on their spouses.
“We were surprised that the effects of the spouses on the survivors were so much larger in this study than the effect of the survivors on their spouses,” said study author Kristin Litzelman, PhD, of the National Cancer Institute in Bethesda, Maryland. “We expected to see a more reciprocal relationship.”
Dr Litzelman and her colleagues conducted this research in an attempt to understand how cancer survivors and their families influence one another. The team hoped to identify ways to improve the healthcare both parties receive and thereby improve their health and well-being.
The researchers analyzed data from 910 cancer patients and their spouses, comparing them to 910 couples without any kind of cancer-related health problem.
The team used statistical models to assess how each spouse’s quality of life or depression at one time point was associated with his or her partner’s risk of depression around 11 months later. The researchers took into account a person’s previously reported mood, demographic characteristics, and other factors.
The results showed that, when spouses reported feeling depressed, cancer survivors were about 4 times more likely to report being depressed 11 months later (odds ratio [OR]=4.27). This association was stronger among female cancer survivors (OR=9.49) than male survivors (OR=3.98).
Cancer survivors whose spouses reported better HRQOL had a 30% decrease in depressed mood per 10-point improvement in HRQOL score. The ORs were 0.72 for mental health and 0.68 for physical health. The associations between spousal HRQOL and survivor depressed mood were similar for male and female survivors.
The researchers noted that cancer survivors’ moods did not have a significant impact on their spouses’ risk of depressed mood 11 months later.
And the team did not see mood associations in couples without any cancer-related health problems.
“This finding certainly needs to be backed up by other studies, but it highlights the importance of family well-being in cancer survivor outcomes,” Dr Litzelman said. “Our research highlights that spouses need to take care of themselves, not just for their own sake, but also for the sake of the cancer survivor.”
“Our findings also suggest that, when caring for cancer survivors, clinicians may want to assess the well-being of spousal caregivers. Future research could test whether including caregivers in the survivorship care plan might help to improve outcomes for both caregivers and for cancer survivors.” ![]()

chemotherapy
Photo by Rhoda Baer
Cancer survivors’ moods are impacted—both positively and negatively—by their spouses’ moods, according to research published in Cancer Epidemiology, Biomarkers & Prevention.
In the study, cancer survivors whose spouses reported depressed moods were more likely to be depressed after about a year of follow-up, and survivors whose spouses reported better mental and physical health-related quality of life (HRQOL) were less likely to be depressed.
However, survivors’ moods did not have the same impact on their spouses.
“We were surprised that the effects of the spouses on the survivors were so much larger in this study than the effect of the survivors on their spouses,” said study author Kristin Litzelman, PhD, of the National Cancer Institute in Bethesda, Maryland. “We expected to see a more reciprocal relationship.”
Dr Litzelman and her colleagues conducted this research in an attempt to understand how cancer survivors and their families influence one another. The team hoped to identify ways to improve the healthcare both parties receive and thereby improve their health and well-being.
The researchers analyzed data from 910 cancer patients and their spouses, comparing them to 910 couples without any kind of cancer-related health problem.
The team used statistical models to assess how each spouse’s quality of life or depression at one time point was associated with his or her partner’s risk of depression around 11 months later. The researchers took into account a person’s previously reported mood, demographic characteristics, and other factors.
The results showed that, when spouses reported feeling depressed, cancer survivors were about 4 times more likely to report being depressed 11 months later (odds ratio [OR]=4.27). This association was stronger among female cancer survivors (OR=9.49) than male survivors (OR=3.98).
Cancer survivors whose spouses reported better HRQOL had a 30% decrease in depressed mood per 10-point improvement in HRQOL score. The ORs were 0.72 for mental health and 0.68 for physical health. The associations between spousal HRQOL and survivor depressed mood were similar for male and female survivors.
The researchers noted that cancer survivors’ moods did not have a significant impact on their spouses’ risk of depressed mood 11 months later.
And the team did not see mood associations in couples without any cancer-related health problems.
“This finding certainly needs to be backed up by other studies, but it highlights the importance of family well-being in cancer survivor outcomes,” Dr Litzelman said. “Our research highlights that spouses need to take care of themselves, not just for their own sake, but also for the sake of the cancer survivor.”
“Our findings also suggest that, when caring for cancer survivors, clinicians may want to assess the well-being of spousal caregivers. Future research could test whether including caregivers in the survivorship care plan might help to improve outcomes for both caregivers and for cancer survivors.” ![]()
CDS systems often can’t tell if imaging is appropriate

Photo courtesy of NIH
Tools that help physicians decide whether to use diagnostic imaging can help reduce the use of unnecessary tests.
But new research suggests these tools may not be able to determine which tests are necessary most of the time.
The tools in question are computerized clinical decision support (CDS) systems, which match a patient’s characteristics against appropriateness
criteria to produce algorithmic treatment recommendations.
In a study published in JAMA, CDS systems did increase orders of imaging tests rated as “appropriate.”
However, the systems were not able to assign appropriateness ratings for a majority of tests because no appropriateness criteria were available for a particular test, or because the systems themselves were not able to find matching criteria.
“The increase in orders rated as appropriate is promising, but the number of tests that were not rated indicates there is room for further improvement of these tools,” said study author Peter S. Hussey, PhD, of the RAND Corporation in Boston, Massachusetts.
Study details
Dr Hussey and his colleagues used data from the Medicare Imaging Demonstration to evaluate the relationship of CDS system use with the proportion of imaging orders matched to appropriateness criteria, the appropriateness of ordered images, and the proportion of orders that changed after feedback.
The team compared 2 time periods during which clinicians used computerized radiology order entry systems and CDS systems for MRI, CT, and nuclear medicine procedures.
During a 6-month baseline period, the CDS systems tracked whether orders were linked with appropriateness criteria but did not provide clinicians with feedback on the appropriateness of orders.
During the 18-month intervention period, the CDS systems provided feedback indicating whether the order was linked to appropriateness criteria and, if so, the appropriateness rating, any recommendations for alternative orders, and a link to documentation supporting each rating.
National medical specialty societies developed the appropriateness criteria using expert panels that reviewed evidence and completed a structured rating process. The same appropriateness criteria were loaded into the CDS systems tools for all participating clinicians.
In all, 3340 clinicians placed 117,348 orders for advanced diagnostic imaging procedures.
Results
The CDS systems could not match most orders to appropriateness criteria. The systems did not identify relevant criteria for 63.3% of orders made during the baseline period and 66.5% of orders made during the intervention period.
Of the orders CDS systems could rate, 73.7% ordered during the baseline period and 81% ordered during the intervention period were rated as appropriate, and 11.1% and 6.4%, respectively, were rated inappropriate.
Of the orders that were initially rated as inappropriate, 4.8% were changed, and 1.9% were canceled.
When the CDS systems suggested an alternative for inappropriate orders, 9.9% of the orders were changed, and 0.4% were canceled. When the systems did not provide an alternative, 1.4% of inappropriate orders were changed, and 2.8% were canceled.
“In response to these findings, we recommend that clinical decision support efforts should focus on tools that help clinicians perform their work more efficiently and effectively,” said study author Katherine Kahn, MD, of the University of California, Los Angeles.
“We need a more comprehensive set of evidence-based guidelines that cover a greater proportion of advanced imaging orders for Medicare patients, and provide better methods for communicating feedback to clinicians.” ![]()

Photo courtesy of NIH
Tools that help physicians decide whether to use diagnostic imaging can help reduce the use of unnecessary tests.
But new research suggests these tools may not be able to determine which tests are necessary most of the time.
The tools in question are computerized clinical decision support (CDS) systems, which match a patient’s characteristics against appropriateness
criteria to produce algorithmic treatment recommendations.
In a study published in JAMA, CDS systems did increase orders of imaging tests rated as “appropriate.”
However, the systems were not able to assign appropriateness ratings for a majority of tests because no appropriateness criteria were available for a particular test, or because the systems themselves were not able to find matching criteria.
“The increase in orders rated as appropriate is promising, but the number of tests that were not rated indicates there is room for further improvement of these tools,” said study author Peter S. Hussey, PhD, of the RAND Corporation in Boston, Massachusetts.
Study details
Dr Hussey and his colleagues used data from the Medicare Imaging Demonstration to evaluate the relationship of CDS system use with the proportion of imaging orders matched to appropriateness criteria, the appropriateness of ordered images, and the proportion of orders that changed after feedback.
The team compared 2 time periods during which clinicians used computerized radiology order entry systems and CDS systems for MRI, CT, and nuclear medicine procedures.
During a 6-month baseline period, the CDS systems tracked whether orders were linked with appropriateness criteria but did not provide clinicians with feedback on the appropriateness of orders.
During the 18-month intervention period, the CDS systems provided feedback indicating whether the order was linked to appropriateness criteria and, if so, the appropriateness rating, any recommendations for alternative orders, and a link to documentation supporting each rating.
National medical specialty societies developed the appropriateness criteria using expert panels that reviewed evidence and completed a structured rating process. The same appropriateness criteria were loaded into the CDS systems tools for all participating clinicians.
In all, 3340 clinicians placed 117,348 orders for advanced diagnostic imaging procedures.
Results
The CDS systems could not match most orders to appropriateness criteria. The systems did not identify relevant criteria for 63.3% of orders made during the baseline period and 66.5% of orders made during the intervention period.
Of the orders CDS systems could rate, 73.7% ordered during the baseline period and 81% ordered during the intervention period were rated as appropriate, and 11.1% and 6.4%, respectively, were rated inappropriate.
Of the orders that were initially rated as inappropriate, 4.8% were changed, and 1.9% were canceled.
When the CDS systems suggested an alternative for inappropriate orders, 9.9% of the orders were changed, and 0.4% were canceled. When the systems did not provide an alternative, 1.4% of inappropriate orders were changed, and 2.8% were canceled.
“In response to these findings, we recommend that clinical decision support efforts should focus on tools that help clinicians perform their work more efficiently and effectively,” said study author Katherine Kahn, MD, of the University of California, Los Angeles.
“We need a more comprehensive set of evidence-based guidelines that cover a greater proportion of advanced imaging orders for Medicare patients, and provide better methods for communicating feedback to clinicians.” ![]()

Photo courtesy of NIH
Tools that help physicians decide whether to use diagnostic imaging can help reduce the use of unnecessary tests.
But new research suggests these tools may not be able to determine which tests are necessary most of the time.
The tools in question are computerized clinical decision support (CDS) systems, which match a patient’s characteristics against appropriateness
criteria to produce algorithmic treatment recommendations.
In a study published in JAMA, CDS systems did increase orders of imaging tests rated as “appropriate.”
However, the systems were not able to assign appropriateness ratings for a majority of tests because no appropriateness criteria were available for a particular test, or because the systems themselves were not able to find matching criteria.
“The increase in orders rated as appropriate is promising, but the number of tests that were not rated indicates there is room for further improvement of these tools,” said study author Peter S. Hussey, PhD, of the RAND Corporation in Boston, Massachusetts.
Study details
Dr Hussey and his colleagues used data from the Medicare Imaging Demonstration to evaluate the relationship of CDS system use with the proportion of imaging orders matched to appropriateness criteria, the appropriateness of ordered images, and the proportion of orders that changed after feedback.
The team compared 2 time periods during which clinicians used computerized radiology order entry systems and CDS systems for MRI, CT, and nuclear medicine procedures.
During a 6-month baseline period, the CDS systems tracked whether orders were linked with appropriateness criteria but did not provide clinicians with feedback on the appropriateness of orders.
During the 18-month intervention period, the CDS systems provided feedback indicating whether the order was linked to appropriateness criteria and, if so, the appropriateness rating, any recommendations for alternative orders, and a link to documentation supporting each rating.
National medical specialty societies developed the appropriateness criteria using expert panels that reviewed evidence and completed a structured rating process. The same appropriateness criteria were loaded into the CDS systems tools for all participating clinicians.
In all, 3340 clinicians placed 117,348 orders for advanced diagnostic imaging procedures.
Results
The CDS systems could not match most orders to appropriateness criteria. The systems did not identify relevant criteria for 63.3% of orders made during the baseline period and 66.5% of orders made during the intervention period.
Of the orders CDS systems could rate, 73.7% ordered during the baseline period and 81% ordered during the intervention period were rated as appropriate, and 11.1% and 6.4%, respectively, were rated inappropriate.
Of the orders that were initially rated as inappropriate, 4.8% were changed, and 1.9% were canceled.
When the CDS systems suggested an alternative for inappropriate orders, 9.9% of the orders were changed, and 0.4% were canceled. When the systems did not provide an alternative, 1.4% of inappropriate orders were changed, and 2.8% were canceled.
“In response to these findings, we recommend that clinical decision support efforts should focus on tools that help clinicians perform their work more efficiently and effectively,” said study author Katherine Kahn, MD, of the University of California, Los Angeles.
“We need a more comprehensive set of evidence-based guidelines that cover a greater proportion of advanced imaging orders for Medicare patients, and provide better methods for communicating feedback to clinicians.” ![]()
Scientists uncover structure of TOR complex 2

Photo courtesy of
University of Geneva
A group of researchers has developed a new tool to study the structure and function of target of rapamycin complex 2 (TORC2), which helps explain why rapamycin cannot access the TOR protein in this complex.
TOR is essential for the growth of normal cells but is hyperactive in tumor cells. Rapamycin is an immunosuppressant and anticancer agent that inactivates TOR in TORC1 but not in TORC2.
“In order to more easily study TORC2, we wanted to learn how to make this complex sensitive to rapamycin,” said Robbie Loewith, PhD, of the University of Geneva in Switzerland.
So Dr Loewith and a team of scientists from Switzerland, France, and the UK set out to elucidate how TORC2 works. The team reported their findings in Molecular Cell.
Using crosslinking-mass spectrometry and electron microscopy, they discovered that TORC2 has 3 features in common with TORC1: a rhomboid shape, 2-fold symmetry, and a central cavity delimited by the interface of its protein chains.
The 2 complexes differ markedly, however, in overall size, surface area of the interface, and the volume and shape of the central cavity.
By determining the structure of TORC2, the team could observe which subunit within TORC2 was obstructing the rapamycin binding site on TOR.
“By deleting part of this subunit, we generated a variant of TORC2 sensitive to rapamycin,” said Manoel Prouteau, PhD, also of the University of Geneva.
This allowed the researchers to study how TORC2 acts to stimulate cell growth.
Now, they hope to identify a specific inhibitor of endogenous TORC2 that could also be an effective anticancer agent.
“Our discovery that TORC2 inhibition alone is sufficient to block the cell cycle suggests that mTORC2-specific inhibitors may provide new and potentially better therapeutic alternatives,” the team concluded. ![]()

Photo courtesy of
University of Geneva
A group of researchers has developed a new tool to study the structure and function of target of rapamycin complex 2 (TORC2), which helps explain why rapamycin cannot access the TOR protein in this complex.
TOR is essential for the growth of normal cells but is hyperactive in tumor cells. Rapamycin is an immunosuppressant and anticancer agent that inactivates TOR in TORC1 but not in TORC2.
“In order to more easily study TORC2, we wanted to learn how to make this complex sensitive to rapamycin,” said Robbie Loewith, PhD, of the University of Geneva in Switzerland.
So Dr Loewith and a team of scientists from Switzerland, France, and the UK set out to elucidate how TORC2 works. The team reported their findings in Molecular Cell.
Using crosslinking-mass spectrometry and electron microscopy, they discovered that TORC2 has 3 features in common with TORC1: a rhomboid shape, 2-fold symmetry, and a central cavity delimited by the interface of its protein chains.
The 2 complexes differ markedly, however, in overall size, surface area of the interface, and the volume and shape of the central cavity.
By determining the structure of TORC2, the team could observe which subunit within TORC2 was obstructing the rapamycin binding site on TOR.
“By deleting part of this subunit, we generated a variant of TORC2 sensitive to rapamycin,” said Manoel Prouteau, PhD, also of the University of Geneva.
This allowed the researchers to study how TORC2 acts to stimulate cell growth.
Now, they hope to identify a specific inhibitor of endogenous TORC2 that could also be an effective anticancer agent.
“Our discovery that TORC2 inhibition alone is sufficient to block the cell cycle suggests that mTORC2-specific inhibitors may provide new and potentially better therapeutic alternatives,” the team concluded. ![]()

Photo courtesy of
University of Geneva
A group of researchers has developed a new tool to study the structure and function of target of rapamycin complex 2 (TORC2), which helps explain why rapamycin cannot access the TOR protein in this complex.
TOR is essential for the growth of normal cells but is hyperactive in tumor cells. Rapamycin is an immunosuppressant and anticancer agent that inactivates TOR in TORC1 but not in TORC2.
“In order to more easily study TORC2, we wanted to learn how to make this complex sensitive to rapamycin,” said Robbie Loewith, PhD, of the University of Geneva in Switzerland.
So Dr Loewith and a team of scientists from Switzerland, France, and the UK set out to elucidate how TORC2 works. The team reported their findings in Molecular Cell.
Using crosslinking-mass spectrometry and electron microscopy, they discovered that TORC2 has 3 features in common with TORC1: a rhomboid shape, 2-fold symmetry, and a central cavity delimited by the interface of its protein chains.
The 2 complexes differ markedly, however, in overall size, surface area of the interface, and the volume and shape of the central cavity.
By determining the structure of TORC2, the team could observe which subunit within TORC2 was obstructing the rapamycin binding site on TOR.
“By deleting part of this subunit, we generated a variant of TORC2 sensitive to rapamycin,” said Manoel Prouteau, PhD, also of the University of Geneva.
This allowed the researchers to study how TORC2 acts to stimulate cell growth.
Now, they hope to identify a specific inhibitor of endogenous TORC2 that could also be an effective anticancer agent.
“Our discovery that TORC2 inhibition alone is sufficient to block the cell cycle suggests that mTORC2-specific inhibitors may provide new and potentially better therapeutic alternatives,” the team concluded. ![]()
Urine test could reduce need for blood samples

Photo by Juan D. Alfonso
A new approach to urine testing could make the tests more versatile and therefore decrease the need for blood tests, according to researchers.
They believe the method could also reduce costs, produce results faster than current tests, and lower the volume of urine needed for a sample.
R. Kenneth Marcus, PhD, of Clemson University in South Carolina, and his colleagues described this method in Proteomics-Clinical Applications.
Dr Marcus noted that the trouble with testing urine is that it’s awash in salt, so it can be tricky to isolate the proteins that act as biomarkers.
To overcome this problem, he and his colleagues used a string made of capillary-channeled polymer fibers. The team packed the fibers into plastic tubes and then passed urine samples through the tubes by spinning them in a centrifuge for 30 seconds.
Then, the researchers ran de-ionized water through the tubes for a minute to wash off salt and other contaminants.
As proteins are hydrophobic, they remained stuck to the fibers. The team extracted the proteins by running a solvent through the tubes in the centrifuge for 30 seconds.
When this process was complete, the researchers were left with purified proteins that could be stored in a plastic vial and refrigerated until testing time.
The team was able to extract 12 samples in about 5 minutes, limited only by centrifuge capacity.
In addition to being faster and cheaper than current urine tests, the new testing method should also make it easier to test urine samples from infants, Dr Marcus said.
One of the challenges now is getting a large enough sample, but the new method requires only a few microliters of urine. ![]()

Photo by Juan D. Alfonso
A new approach to urine testing could make the tests more versatile and therefore decrease the need for blood tests, according to researchers.
They believe the method could also reduce costs, produce results faster than current tests, and lower the volume of urine needed for a sample.
R. Kenneth Marcus, PhD, of Clemson University in South Carolina, and his colleagues described this method in Proteomics-Clinical Applications.
Dr Marcus noted that the trouble with testing urine is that it’s awash in salt, so it can be tricky to isolate the proteins that act as biomarkers.
To overcome this problem, he and his colleagues used a string made of capillary-channeled polymer fibers. The team packed the fibers into plastic tubes and then passed urine samples through the tubes by spinning them in a centrifuge for 30 seconds.
Then, the researchers ran de-ionized water through the tubes for a minute to wash off salt and other contaminants.
As proteins are hydrophobic, they remained stuck to the fibers. The team extracted the proteins by running a solvent through the tubes in the centrifuge for 30 seconds.
When this process was complete, the researchers were left with purified proteins that could be stored in a plastic vial and refrigerated until testing time.
The team was able to extract 12 samples in about 5 minutes, limited only by centrifuge capacity.
In addition to being faster and cheaper than current urine tests, the new testing method should also make it easier to test urine samples from infants, Dr Marcus said.
One of the challenges now is getting a large enough sample, but the new method requires only a few microliters of urine. ![]()

Photo by Juan D. Alfonso
A new approach to urine testing could make the tests more versatile and therefore decrease the need for blood tests, according to researchers.
They believe the method could also reduce costs, produce results faster than current tests, and lower the volume of urine needed for a sample.
R. Kenneth Marcus, PhD, of Clemson University in South Carolina, and his colleagues described this method in Proteomics-Clinical Applications.
Dr Marcus noted that the trouble with testing urine is that it’s awash in salt, so it can be tricky to isolate the proteins that act as biomarkers.
To overcome this problem, he and his colleagues used a string made of capillary-channeled polymer fibers. The team packed the fibers into plastic tubes and then passed urine samples through the tubes by spinning them in a centrifuge for 30 seconds.
Then, the researchers ran de-ionized water through the tubes for a minute to wash off salt and other contaminants.
As proteins are hydrophobic, they remained stuck to the fibers. The team extracted the proteins by running a solvent through the tubes in the centrifuge for 30 seconds.
When this process was complete, the researchers were left with purified proteins that could be stored in a plastic vial and refrigerated until testing time.
The team was able to extract 12 samples in about 5 minutes, limited only by centrifuge capacity.
In addition to being faster and cheaper than current urine tests, the new testing method should also make it easier to test urine samples from infants, Dr Marcus said.
One of the challenges now is getting a large enough sample, but the new method requires only a few microliters of urine. ![]()
Understanding taste dysfunction in cancer
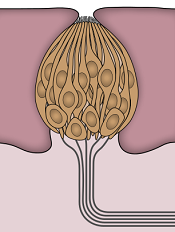
Image by Jonas Töle
Investigators have identified a molecular pathway that aids the renewal of taste buds, and they believe this discovery may have implications for cancer patients who suffer from an altered sense of taste during treatment.
“Taste dysfunction can . . . result from an alteration of the renewal capacities of taste buds and is often associated with psychological distress and malnutrition,” said Dany Gaillard, PhD, of the University of Colorado Anschutz Medical Campus in Aurora.
He and his colleagues decided to investigate this dysfunction using mouse models, and the group reported their findings in PLOS Genetics.
The investigators discovered that a protein in the Wnt pathway, ß-catenin, controls the renewal of taste cells by regulating separate stages of taste-cell turnover.
Previous research showed that Wnt/β-catenin signaling is crucial in developing taste buds in embryos and regulating the renewal of epithelial tissue in adults, including skin, hair follicles, the intestine, and the mouth.
“We show that activating this pathway directs the newly born cells to become, primarily, a specific taste- cell type whose role is to support the other taste cells and help them work efficiently,” said Linda Barlow, PhD, also of the University of Colorado Anschutz Medical Campus.
As chemotherapy destroys dividing precursor cells, including those that produce taste cells, the investigators believe that activating Wnt signaling may be a way to renew taste buds after chemotherapy.
New small-molecule drugs that specifically block the Wnt pathway are under development, and Drs Gaillard and Barlow predict these drugs could also cause taste dysfunction.
Dr Barlow said more research is needed to understand how taste is altered at the cellular level, but this research holds promise for developing new ways to improve cancer patients’ quality of life. ![]()

Image by Jonas Töle
Investigators have identified a molecular pathway that aids the renewal of taste buds, and they believe this discovery may have implications for cancer patients who suffer from an altered sense of taste during treatment.
“Taste dysfunction can . . . result from an alteration of the renewal capacities of taste buds and is often associated with psychological distress and malnutrition,” said Dany Gaillard, PhD, of the University of Colorado Anschutz Medical Campus in Aurora.
He and his colleagues decided to investigate this dysfunction using mouse models, and the group reported their findings in PLOS Genetics.
The investigators discovered that a protein in the Wnt pathway, ß-catenin, controls the renewal of taste cells by regulating separate stages of taste-cell turnover.
Previous research showed that Wnt/β-catenin signaling is crucial in developing taste buds in embryos and regulating the renewal of epithelial tissue in adults, including skin, hair follicles, the intestine, and the mouth.
“We show that activating this pathway directs the newly born cells to become, primarily, a specific taste- cell type whose role is to support the other taste cells and help them work efficiently,” said Linda Barlow, PhD, also of the University of Colorado Anschutz Medical Campus.
As chemotherapy destroys dividing precursor cells, including those that produce taste cells, the investigators believe that activating Wnt signaling may be a way to renew taste buds after chemotherapy.
New small-molecule drugs that specifically block the Wnt pathway are under development, and Drs Gaillard and Barlow predict these drugs could also cause taste dysfunction.
Dr Barlow said more research is needed to understand how taste is altered at the cellular level, but this research holds promise for developing new ways to improve cancer patients’ quality of life. ![]()

Image by Jonas Töle
Investigators have identified a molecular pathway that aids the renewal of taste buds, and they believe this discovery may have implications for cancer patients who suffer from an altered sense of taste during treatment.
“Taste dysfunction can . . . result from an alteration of the renewal capacities of taste buds and is often associated with psychological distress and malnutrition,” said Dany Gaillard, PhD, of the University of Colorado Anschutz Medical Campus in Aurora.
He and his colleagues decided to investigate this dysfunction using mouse models, and the group reported their findings in PLOS Genetics.
The investigators discovered that a protein in the Wnt pathway, ß-catenin, controls the renewal of taste cells by regulating separate stages of taste-cell turnover.
Previous research showed that Wnt/β-catenin signaling is crucial in developing taste buds in embryos and regulating the renewal of epithelial tissue in adults, including skin, hair follicles, the intestine, and the mouth.
“We show that activating this pathway directs the newly born cells to become, primarily, a specific taste- cell type whose role is to support the other taste cells and help them work efficiently,” said Linda Barlow, PhD, also of the University of Colorado Anschutz Medical Campus.
As chemotherapy destroys dividing precursor cells, including those that produce taste cells, the investigators believe that activating Wnt signaling may be a way to renew taste buds after chemotherapy.
New small-molecule drugs that specifically block the Wnt pathway are under development, and Drs Gaillard and Barlow predict these drugs could also cause taste dysfunction.
Dr Barlow said more research is needed to understand how taste is altered at the cellular level, but this research holds promise for developing new ways to improve cancer patients’ quality of life. ![]()
Herbs reduce fatigue in cancer patients

Photo by Alexander Baxevanis
An herbal mixture used in traditional Chinese medicine can reduce fatigue in cancer patients, results of a phase 1/2 study suggest.
The mixture, Ren Shen Yangrong Tang (RSYRT), is a soup containing 12 herbs.
In the study, cancer patients suffering from moderate to severe fatigue reported significantly less fatigue after taking RSYRT for 2 to 3 weeks.
Researchers reported these results in the Journal of Alternative and Complementary Medicine.
Yichen Xu, MD, of Beijing Cancer Hospital & Institute in China, and colleagues evaluated RSYRT in 33 patients who had completed cancer treatment. The patients had stable disease and no anemia.
Eleven patients had moderate fatigue (a score of 4-6 on a 0-10 scale), and 22 had severe fatigue (a score of 7-10). All patients had experienced fatigue for at least 4 months.
Patients took RSYRT twice a day for 6 weeks and experienced a significant decrease in fatigue severity. The mean fatigue score decreased from 7.06 at baseline to 3.30 at the 6-week mark (P<0.001).
The fatigue category also changed significantly (P=0.024). Among the 22 patients who had severe fatigue before RSYRT, half had mild fatigue after therapy, and half had moderate fatigue.
Among the 11 patients who had moderate fatigue at baseline, only 1 still had moderate fatigue after receiving RSYRT. The rest had mild fatigue.
All of the patients said they felt better after taking RSYRT for 4 weeks.
There were no “uncomfortable events” related to RSYRT, such as gastrointestinal upset, insomnia, headache, or rash. None of the patients required a dose reduction or dose interruption.
None of the patients had blood chemistry abnormalities or abnormal liver/kidney function. Two patients who had a change in ST segment before RSYRT had normal electrocardiogram results after treatment. ![]()

Photo by Alexander Baxevanis
An herbal mixture used in traditional Chinese medicine can reduce fatigue in cancer patients, results of a phase 1/2 study suggest.
The mixture, Ren Shen Yangrong Tang (RSYRT), is a soup containing 12 herbs.
In the study, cancer patients suffering from moderate to severe fatigue reported significantly less fatigue after taking RSYRT for 2 to 3 weeks.
Researchers reported these results in the Journal of Alternative and Complementary Medicine.
Yichen Xu, MD, of Beijing Cancer Hospital & Institute in China, and colleagues evaluated RSYRT in 33 patients who had completed cancer treatment. The patients had stable disease and no anemia.
Eleven patients had moderate fatigue (a score of 4-6 on a 0-10 scale), and 22 had severe fatigue (a score of 7-10). All patients had experienced fatigue for at least 4 months.
Patients took RSYRT twice a day for 6 weeks and experienced a significant decrease in fatigue severity. The mean fatigue score decreased from 7.06 at baseline to 3.30 at the 6-week mark (P<0.001).
The fatigue category also changed significantly (P=0.024). Among the 22 patients who had severe fatigue before RSYRT, half had mild fatigue after therapy, and half had moderate fatigue.
Among the 11 patients who had moderate fatigue at baseline, only 1 still had moderate fatigue after receiving RSYRT. The rest had mild fatigue.
All of the patients said they felt better after taking RSYRT for 4 weeks.
There were no “uncomfortable events” related to RSYRT, such as gastrointestinal upset, insomnia, headache, or rash. None of the patients required a dose reduction or dose interruption.
None of the patients had blood chemistry abnormalities or abnormal liver/kidney function. Two patients who had a change in ST segment before RSYRT had normal electrocardiogram results after treatment. ![]()

Photo by Alexander Baxevanis
An herbal mixture used in traditional Chinese medicine can reduce fatigue in cancer patients, results of a phase 1/2 study suggest.
The mixture, Ren Shen Yangrong Tang (RSYRT), is a soup containing 12 herbs.
In the study, cancer patients suffering from moderate to severe fatigue reported significantly less fatigue after taking RSYRT for 2 to 3 weeks.
Researchers reported these results in the Journal of Alternative and Complementary Medicine.
Yichen Xu, MD, of Beijing Cancer Hospital & Institute in China, and colleagues evaluated RSYRT in 33 patients who had completed cancer treatment. The patients had stable disease and no anemia.
Eleven patients had moderate fatigue (a score of 4-6 on a 0-10 scale), and 22 had severe fatigue (a score of 7-10). All patients had experienced fatigue for at least 4 months.
Patients took RSYRT twice a day for 6 weeks and experienced a significant decrease in fatigue severity. The mean fatigue score decreased from 7.06 at baseline to 3.30 at the 6-week mark (P<0.001).
The fatigue category also changed significantly (P=0.024). Among the 22 patients who had severe fatigue before RSYRT, half had mild fatigue after therapy, and half had moderate fatigue.
Among the 11 patients who had moderate fatigue at baseline, only 1 still had moderate fatigue after receiving RSYRT. The rest had mild fatigue.
All of the patients said they felt better after taking RSYRT for 4 weeks.
There were no “uncomfortable events” related to RSYRT, such as gastrointestinal upset, insomnia, headache, or rash. None of the patients required a dose reduction or dose interruption.
None of the patients had blood chemistry abnormalities or abnormal liver/kidney function. Two patients who had a change in ST segment before RSYRT had normal electrocardiogram results after treatment. ![]()
Co-infection may boost malaria mortality

Co-infection with malaria and a virus closely related to the Epstein-Barr virus (EBV) may make the malaria lethal, according to preclinical research published in PLOS Pathogens.
Children in sub-Saharan Africa become infected with EBV in infancy.
Within the same time period, they become susceptible to malaria parasite infection because protective antibodies from their mothers fade away.
“Where we think kids get into trouble is when both infections are happening at the same time, because case reports show EBV can produce a weeks-long suppression of the immune system,” said Tracey Lamb, PhD, of Emory University School of Medicine in Atlanta, Georgia.
Dr Lamb and her colleagues studied mice infected by the malaria parasite Plasmodium yoelii, which is usually non-lethal because the mice develop antibodies that control the parasites.
The researchers found that co-infection with murine gammaherpesvirus 68 (MHV68), a close relative of EBV that infects mice, made P yoelii lethal.
However, mice that had entered the chronic phase of MHV68 infection (several weeks to months after primary infection) were not affected.
The experiments indicated that MHV68 infection hinders the immune system in developing antibodies against P yoelii.
“These results are part of a pattern of evidence suggesting that clinicians treating severe malaria should check for acute EBV co-infection, and that ongoing malaria studies should include EBV as a potential risk factor for more severe forms of the disease,” said Caline Matar, a graduate student at Emory University School of Medicine.
“This phenomenon may not be unique to EBV,” added Sam Speck, PhD, also of Emory University School of Medicine.
“[I]nfections with other pathogens may also exacerbate malarial disease, since many pathogens have the capacity to suppress various components of the host immune response.” ![]()

Co-infection with malaria and a virus closely related to the Epstein-Barr virus (EBV) may make the malaria lethal, according to preclinical research published in PLOS Pathogens.
Children in sub-Saharan Africa become infected with EBV in infancy.
Within the same time period, they become susceptible to malaria parasite infection because protective antibodies from their mothers fade away.
“Where we think kids get into trouble is when both infections are happening at the same time, because case reports show EBV can produce a weeks-long suppression of the immune system,” said Tracey Lamb, PhD, of Emory University School of Medicine in Atlanta, Georgia.
Dr Lamb and her colleagues studied mice infected by the malaria parasite Plasmodium yoelii, which is usually non-lethal because the mice develop antibodies that control the parasites.
The researchers found that co-infection with murine gammaherpesvirus 68 (MHV68), a close relative of EBV that infects mice, made P yoelii lethal.
However, mice that had entered the chronic phase of MHV68 infection (several weeks to months after primary infection) were not affected.
The experiments indicated that MHV68 infection hinders the immune system in developing antibodies against P yoelii.
“These results are part of a pattern of evidence suggesting that clinicians treating severe malaria should check for acute EBV co-infection, and that ongoing malaria studies should include EBV as a potential risk factor for more severe forms of the disease,” said Caline Matar, a graduate student at Emory University School of Medicine.
“This phenomenon may not be unique to EBV,” added Sam Speck, PhD, also of Emory University School of Medicine.
“[I]nfections with other pathogens may also exacerbate malarial disease, since many pathogens have the capacity to suppress various components of the host immune response.” ![]()

Co-infection with malaria and a virus closely related to the Epstein-Barr virus (EBV) may make the malaria lethal, according to preclinical research published in PLOS Pathogens.
Children in sub-Saharan Africa become infected with EBV in infancy.
Within the same time period, they become susceptible to malaria parasite infection because protective antibodies from their mothers fade away.
“Where we think kids get into trouble is when both infections are happening at the same time, because case reports show EBV can produce a weeks-long suppression of the immune system,” said Tracey Lamb, PhD, of Emory University School of Medicine in Atlanta, Georgia.
Dr Lamb and her colleagues studied mice infected by the malaria parasite Plasmodium yoelii, which is usually non-lethal because the mice develop antibodies that control the parasites.
The researchers found that co-infection with murine gammaherpesvirus 68 (MHV68), a close relative of EBV that infects mice, made P yoelii lethal.
However, mice that had entered the chronic phase of MHV68 infection (several weeks to months after primary infection) were not affected.
The experiments indicated that MHV68 infection hinders the immune system in developing antibodies against P yoelii.
“These results are part of a pattern of evidence suggesting that clinicians treating severe malaria should check for acute EBV co-infection, and that ongoing malaria studies should include EBV as a potential risk factor for more severe forms of the disease,” said Caline Matar, a graduate student at Emory University School of Medicine.
“This phenomenon may not be unique to EBV,” added Sam Speck, PhD, also of Emory University School of Medicine.
“[I]nfections with other pathogens may also exacerbate malarial disease, since many pathogens have the capacity to suppress various components of the host immune response.”
New insecticide targets malaria-carrying mosquito

Photo courtesy of the CDC
Recent progress in halting the spread of malaria has hinged, in part, on the use of insecticide-treated bed nets and spraying programs that target Anopheles gambiae mosquitoes.
Unfortunately, the mosquitoes are developing resistance to insecticides such as pyrethroid.
Wondering if they could defeat the mosquitoes by developing a new insecticide,a group of researchers set out to make blood meals toxic for Anopheles gambiae.
The team described their method in The Journal of Experimental Biology.
The researchers decided to target the mosquito glutamate gated chloride channel (AgGluCl), which is an essential component of the insect’s nervous system.
They generated antibodies that specifically targeted a portion of the protein exposed on the surface of nerves, a strategy they acknowledged was somewhat risky.
“Antibodies against a single mosquito antigen have never been shown to have mosquitocidal properties before, and the majority of previous research had focused on midgut antigens, while we were targeting a neuronal antigen expressed only in tissues found outside of the midgut,” said study author Jacob Meyers, a graduate student at Colorado State University in Fort Collins.
After injecting rabbits with a tiny portion of the surface of the AgGluCl protein channel, the researchers waited for the rabbits’ immune systems to begin producing antibodies tailored to the channel.
Then, the team collected the antibodies, mixed them with fresh blood, and fed the mixture to malaria-carrying mosquitoes (Anopheles gambiae), yellow fever-carrying mosquitoes (Aedes aegypti), and mosquitoes that carry the West Nile virus (Culex tarsalis).
Neither the yellow fever nor West Nile virus mosquitoes responded to the spiked blood. However, significant numbers of Anopheles gambiae mosquitoes died after consuming the blood/antibody cocktail. The highest antibody doses killed more than 90% of the insects within a day.
The researchers looked into why the yellow fever and West Nile virus mosquitoes had been immune to the antibodies and found the antibodies could not pass across the mosquitoes’ guts into the hemolymph. But the antibodies passed into the hemolymph of Anopheles gambiae mosquitoes with ease.
Intrigued by the antibodies’ mode of action, the researchers fed the insects a blood meal laced with the antibodies and a lethal dose of ivermectin, an insecticide that also targets the AgGluCl protein channel.
Then, the team monitored the mosquitoes’ survival to find out more about how the antibodies may destroy the insects. Mosquitoes that received ivermectin with the antibodies were more likely to survive than insects that received ivermectin alone.
“We believe that ivermectin is able to bind to AgGluCl,” Meyers said, “but the antibody keeps the channel from opening and becoming active.”
Having shown that antibodies targeted to the glutamate gated chloride channel in blood meals can be effective insecticides, the researchers are interested to find out if antibody-laced blood meals are equally deadly outside the lab.
First, though, the team plans to immunize cattle against the AgGluCl antigen and feed Anopheles gambiae on the immunized cattle in the lab. If the strategy proves successful, Meyers envisages a large-scale cattle immunization program as part of a combined attack on the malaria parasite.

Photo courtesy of the CDC
Recent progress in halting the spread of malaria has hinged, in part, on the use of insecticide-treated bed nets and spraying programs that target Anopheles gambiae mosquitoes.
Unfortunately, the mosquitoes are developing resistance to insecticides such as pyrethroid.
Wondering if they could defeat the mosquitoes by developing a new insecticide,a group of researchers set out to make blood meals toxic for Anopheles gambiae.
The team described their method in The Journal of Experimental Biology.
The researchers decided to target the mosquito glutamate gated chloride channel (AgGluCl), which is an essential component of the insect’s nervous system.
They generated antibodies that specifically targeted a portion of the protein exposed on the surface of nerves, a strategy they acknowledged was somewhat risky.
“Antibodies against a single mosquito antigen have never been shown to have mosquitocidal properties before, and the majority of previous research had focused on midgut antigens, while we were targeting a neuronal antigen expressed only in tissues found outside of the midgut,” said study author Jacob Meyers, a graduate student at Colorado State University in Fort Collins.
After injecting rabbits with a tiny portion of the surface of the AgGluCl protein channel, the researchers waited for the rabbits’ immune systems to begin producing antibodies tailored to the channel.
Then, the team collected the antibodies, mixed them with fresh blood, and fed the mixture to malaria-carrying mosquitoes (Anopheles gambiae), yellow fever-carrying mosquitoes (Aedes aegypti), and mosquitoes that carry the West Nile virus (Culex tarsalis).
Neither the yellow fever nor West Nile virus mosquitoes responded to the spiked blood. However, significant numbers of Anopheles gambiae mosquitoes died after consuming the blood/antibody cocktail. The highest antibody doses killed more than 90% of the insects within a day.
The researchers looked into why the yellow fever and West Nile virus mosquitoes had been immune to the antibodies and found the antibodies could not pass across the mosquitoes’ guts into the hemolymph. But the antibodies passed into the hemolymph of Anopheles gambiae mosquitoes with ease.
Intrigued by the antibodies’ mode of action, the researchers fed the insects a blood meal laced with the antibodies and a lethal dose of ivermectin, an insecticide that also targets the AgGluCl protein channel.
Then, the team monitored the mosquitoes’ survival to find out more about how the antibodies may destroy the insects. Mosquitoes that received ivermectin with the antibodies were more likely to survive than insects that received ivermectin alone.
“We believe that ivermectin is able to bind to AgGluCl,” Meyers said, “but the antibody keeps the channel from opening and becoming active.”
Having shown that antibodies targeted to the glutamate gated chloride channel in blood meals can be effective insecticides, the researchers are interested to find out if antibody-laced blood meals are equally deadly outside the lab.
First, though, the team plans to immunize cattle against the AgGluCl antigen and feed Anopheles gambiae on the immunized cattle in the lab. If the strategy proves successful, Meyers envisages a large-scale cattle immunization program as part of a combined attack on the malaria parasite.

Photo courtesy of the CDC
Recent progress in halting the spread of malaria has hinged, in part, on the use of insecticide-treated bed nets and spraying programs that target Anopheles gambiae mosquitoes.
Unfortunately, the mosquitoes are developing resistance to insecticides such as pyrethroid.
Wondering if they could defeat the mosquitoes by developing a new insecticide,a group of researchers set out to make blood meals toxic for Anopheles gambiae.
The team described their method in The Journal of Experimental Biology.
The researchers decided to target the mosquito glutamate gated chloride channel (AgGluCl), which is an essential component of the insect’s nervous system.
They generated antibodies that specifically targeted a portion of the protein exposed on the surface of nerves, a strategy they acknowledged was somewhat risky.
“Antibodies against a single mosquito antigen have never been shown to have mosquitocidal properties before, and the majority of previous research had focused on midgut antigens, while we were targeting a neuronal antigen expressed only in tissues found outside of the midgut,” said study author Jacob Meyers, a graduate student at Colorado State University in Fort Collins.
After injecting rabbits with a tiny portion of the surface of the AgGluCl protein channel, the researchers waited for the rabbits’ immune systems to begin producing antibodies tailored to the channel.
Then, the team collected the antibodies, mixed them with fresh blood, and fed the mixture to malaria-carrying mosquitoes (Anopheles gambiae), yellow fever-carrying mosquitoes (Aedes aegypti), and mosquitoes that carry the West Nile virus (Culex tarsalis).
Neither the yellow fever nor West Nile virus mosquitoes responded to the spiked blood. However, significant numbers of Anopheles gambiae mosquitoes died after consuming the blood/antibody cocktail. The highest antibody doses killed more than 90% of the insects within a day.
The researchers looked into why the yellow fever and West Nile virus mosquitoes had been immune to the antibodies and found the antibodies could not pass across the mosquitoes’ guts into the hemolymph. But the antibodies passed into the hemolymph of Anopheles gambiae mosquitoes with ease.
Intrigued by the antibodies’ mode of action, the researchers fed the insects a blood meal laced with the antibodies and a lethal dose of ivermectin, an insecticide that also targets the AgGluCl protein channel.
Then, the team monitored the mosquitoes’ survival to find out more about how the antibodies may destroy the insects. Mosquitoes that received ivermectin with the antibodies were more likely to survive than insects that received ivermectin alone.
“We believe that ivermectin is able to bind to AgGluCl,” Meyers said, “but the antibody keeps the channel from opening and becoming active.”
Having shown that antibodies targeted to the glutamate gated chloride channel in blood meals can be effective insecticides, the researchers are interested to find out if antibody-laced blood meals are equally deadly outside the lab.
First, though, the team plans to immunize cattle against the AgGluCl antigen and feed Anopheles gambiae on the immunized cattle in the lab. If the strategy proves successful, Meyers envisages a large-scale cattle immunization program as part of a combined attack on the malaria parasite.
System can identify life-threatening bleeding

Photo courtesy of UAB Hospital
A computerized system that analyzes vital signs can help healthcare professionals more accurately diagnose trauma patients with life-threatening bleeding, according to research published in Shock.
The system, known as APPRAISE, simultaneously analyzes blood pressure, heart rate, and breathing patterns during emergency transport.
And investigators found that APPRAISE accurately detected most cases of life-threatening bleeding.
“Providing faster care to patients who are bleeding to death saves lives,” said study author Andrew Reisner, MD, of Massachusetts General Hospital in Boston.
“While the clinical information that ambulance crews call in to trauma centers was sufficient to determine the presence of a life-threatening hemorrhage in about half the patients we studied, many other patients were in a ‘gray area’ and may or may not have been at risk of bleeding to death.”
“Our study demonstrated that automated analysis of patients’ vital signs during prehospital transport was significantly better at discriminating between patients who did and did not have life-threatening hemorrhage.”
The APPRAISE system incorporates software based on statistical techniques currently used in stock market trading and manufacturing to determine whether particular data points represent real problems and not random fluctuations.
The system uses an ultracompact personal computer to analyze data gathered by a standard patient monitor used in emergency transport vehicles.
For this study, the system was installed in 2 MedFlight helicopters and collected data on more than 200 trauma patients transported to participating Boston hospitals from February 2010 to December 2012. So that patients’ care was not affected by a still-unproven system, the APPRAISE system’s analysis was not provided to MedFlight crews.
The researchers also analyzed information from a 2005 study of vital sign data gathered manually by a Houston-based air ambulance system.
The team found the APPRAISE system could identify, with 76% sensitivity, patients who needed 9 or more units of packed red blood cells within 24 hours.
This was significantly more sensitive (P<0.05) than any prehospital Shock Index of 1.4 or higher (59%), initial systolic blood pressure less than 110 mmHg (50%), and any prehospital systolic blood pressure less than 90 mmHg (50%).
However, there was no signficant difference between the different measures with regard to specificity for identifying patients who did not need a blood transfusion within 24 hours.
Specificity was 87% for APPRAISE, 88% for any Shock Index of 1.4 or higher, 88% for initial systolic blood pressure less than 110 mmHg, and 90% for any prehospital systolic blood pressure less than 90 mmHg.
Notifications provided by APPRAISE would have been available within 10 minutes of initial monitoring and a median of 20 minutes before patients arrived at the trauma centers.
“The fact that decisions to proceed with surgery or to replenish lost blood often occur only after patients’ arrival means there are delays—sometimes brief but sometimes prolonged—in initiating such life-saving interventions,” Dr Reisner said.
“We are now working on a follow-up study to use this system in actual trauma care and will be measuring whether it truly leads to faster treatment of life-threatening hemorrhage and better patient outcomes. This approach could also be helpful for patients transported by ground ambulance and for hospitalized patients at risk of unexpected hemorrhage, such as during recovery from major surgery.”

Photo courtesy of UAB Hospital
A computerized system that analyzes vital signs can help healthcare professionals more accurately diagnose trauma patients with life-threatening bleeding, according to research published in Shock.
The system, known as APPRAISE, simultaneously analyzes blood pressure, heart rate, and breathing patterns during emergency transport.
And investigators found that APPRAISE accurately detected most cases of life-threatening bleeding.
“Providing faster care to patients who are bleeding to death saves lives,” said study author Andrew Reisner, MD, of Massachusetts General Hospital in Boston.
“While the clinical information that ambulance crews call in to trauma centers was sufficient to determine the presence of a life-threatening hemorrhage in about half the patients we studied, many other patients were in a ‘gray area’ and may or may not have been at risk of bleeding to death.”
“Our study demonstrated that automated analysis of patients’ vital signs during prehospital transport was significantly better at discriminating between patients who did and did not have life-threatening hemorrhage.”
The APPRAISE system incorporates software based on statistical techniques currently used in stock market trading and manufacturing to determine whether particular data points represent real problems and not random fluctuations.
The system uses an ultracompact personal computer to analyze data gathered by a standard patient monitor used in emergency transport vehicles.
For this study, the system was installed in 2 MedFlight helicopters and collected data on more than 200 trauma patients transported to participating Boston hospitals from February 2010 to December 2012. So that patients’ care was not affected by a still-unproven system, the APPRAISE system’s analysis was not provided to MedFlight crews.
The researchers also analyzed information from a 2005 study of vital sign data gathered manually by a Houston-based air ambulance system.
The team found the APPRAISE system could identify, with 76% sensitivity, patients who needed 9 or more units of packed red blood cells within 24 hours.
This was significantly more sensitive (P<0.05) than any prehospital Shock Index of 1.4 or higher (59%), initial systolic blood pressure less than 110 mmHg (50%), and any prehospital systolic blood pressure less than 90 mmHg (50%).
However, there was no signficant difference between the different measures with regard to specificity for identifying patients who did not need a blood transfusion within 24 hours.
Specificity was 87% for APPRAISE, 88% for any Shock Index of 1.4 or higher, 88% for initial systolic blood pressure less than 110 mmHg, and 90% for any prehospital systolic blood pressure less than 90 mmHg.
Notifications provided by APPRAISE would have been available within 10 minutes of initial monitoring and a median of 20 minutes before patients arrived at the trauma centers.
“The fact that decisions to proceed with surgery or to replenish lost blood often occur only after patients’ arrival means there are delays—sometimes brief but sometimes prolonged—in initiating such life-saving interventions,” Dr Reisner said.
“We are now working on a follow-up study to use this system in actual trauma care and will be measuring whether it truly leads to faster treatment of life-threatening hemorrhage and better patient outcomes. This approach could also be helpful for patients transported by ground ambulance and for hospitalized patients at risk of unexpected hemorrhage, such as during recovery from major surgery.”

Photo courtesy of UAB Hospital
A computerized system that analyzes vital signs can help healthcare professionals more accurately diagnose trauma patients with life-threatening bleeding, according to research published in Shock.
The system, known as APPRAISE, simultaneously analyzes blood pressure, heart rate, and breathing patterns during emergency transport.
And investigators found that APPRAISE accurately detected most cases of life-threatening bleeding.
“Providing faster care to patients who are bleeding to death saves lives,” said study author Andrew Reisner, MD, of Massachusetts General Hospital in Boston.
“While the clinical information that ambulance crews call in to trauma centers was sufficient to determine the presence of a life-threatening hemorrhage in about half the patients we studied, many other patients were in a ‘gray area’ and may or may not have been at risk of bleeding to death.”
“Our study demonstrated that automated analysis of patients’ vital signs during prehospital transport was significantly better at discriminating between patients who did and did not have life-threatening hemorrhage.”
The APPRAISE system incorporates software based on statistical techniques currently used in stock market trading and manufacturing to determine whether particular data points represent real problems and not random fluctuations.
The system uses an ultracompact personal computer to analyze data gathered by a standard patient monitor used in emergency transport vehicles.
For this study, the system was installed in 2 MedFlight helicopters and collected data on more than 200 trauma patients transported to participating Boston hospitals from February 2010 to December 2012. So that patients’ care was not affected by a still-unproven system, the APPRAISE system’s analysis was not provided to MedFlight crews.
The researchers also analyzed information from a 2005 study of vital sign data gathered manually by a Houston-based air ambulance system.
The team found the APPRAISE system could identify, with 76% sensitivity, patients who needed 9 or more units of packed red blood cells within 24 hours.
This was significantly more sensitive (P<0.05) than any prehospital Shock Index of 1.4 or higher (59%), initial systolic blood pressure less than 110 mmHg (50%), and any prehospital systolic blood pressure less than 90 mmHg (50%).
However, there was no signficant difference between the different measures with regard to specificity for identifying patients who did not need a blood transfusion within 24 hours.
Specificity was 87% for APPRAISE, 88% for any Shock Index of 1.4 or higher, 88% for initial systolic blood pressure less than 110 mmHg, and 90% for any prehospital systolic blood pressure less than 90 mmHg.
Notifications provided by APPRAISE would have been available within 10 minutes of initial monitoring and a median of 20 minutes before patients arrived at the trauma centers.
“The fact that decisions to proceed with surgery or to replenish lost blood often occur only after patients’ arrival means there are delays—sometimes brief but sometimes prolonged—in initiating such life-saving interventions,” Dr Reisner said.
“We are now working on a follow-up study to use this system in actual trauma care and will be measuring whether it truly leads to faster treatment of life-threatening hemorrhage and better patient outcomes. This approach could also be helpful for patients transported by ground ambulance and for hospitalized patients at risk of unexpected hemorrhage, such as during recovery from major surgery.”
CTLs captured on video destroying cancer cells
cancer cells (blue)
Image courtesy of Gillian
Griffiths and Jonny Settle
New research has illuminated the behavior of cytotoxic T cells (CTLs) as they hunt down and eliminate cancer cells.
Investigators used novel imaging techniques to capture the process on film and described their findings in an article published in Immunity.
The team captured the footage through high-resolution, 3D, time-lapse, multi-color imaging, making use of both spinning disk confocal microscopy and lattice light sheet microscopy.
These techniques involve capturing “slices” of an object and “stitching” them together to provide the final 3D images across the whole cell.
These approaches allowed the investigators to determine the order of events leading to the lethal “hit” CTLs deliver to cancer cells.
“Inside all of us lurks an army of ‘serial killers’ whose primary function is to kill again and again,” said study author Gillian Griffiths, PhD, of the Cambridge Institute for Medical Research in the UK.
“These [CTLs] patrol our bodies, identifying and destroying virally infected and cancer cells, and they do so with remarkable precision and efficiency.”
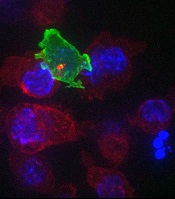
The CTLs, seen in the video as orange or green amorphous “blobs,” move around rapidly, investigating their environment as they travel.
When a CTL finds a cancer cell (blue), membrane protrusions rapidly explore the surface of the cell, checking for tell-tale signs that this is an uninvited guest.
The CTL binds to the cancer cell and injects cytotoxins (red) down microtubules to the interface between the T cell and the cancer cell, before puncturing the surface of the cancer cell and delivering its deadly cargo.
“In our bodies, where cells are packed together, it’s essential that the T cell focuses the lethal hit on its target,” Dr Griffiths explained. “Otherwise, it will cause collateral damage to neighboring, healthy cells.”
“Once the cytotoxins are injected into the cancer cell, its fate is sealed, and we can watch as it withers and dies. The T cell then moves on, hungry to find another victim.”
The investigators’ video is available on YouTube.

cancer cells (blue)
Image courtesy of Gillian
Griffiths and Jonny Settle
New research has illuminated the behavior of cytotoxic T cells (CTLs) as they hunt down and eliminate cancer cells.
Investigators used novel imaging techniques to capture the process on film and described their findings in an article published in Immunity.
The team captured the footage through high-resolution, 3D, time-lapse, multi-color imaging, making use of both spinning disk confocal microscopy and lattice light sheet microscopy.
These techniques involve capturing “slices” of an object and “stitching” them together to provide the final 3D images across the whole cell.
These approaches allowed the investigators to determine the order of events leading to the lethal “hit” CTLs deliver to cancer cells.
“Inside all of us lurks an army of ‘serial killers’ whose primary function is to kill again and again,” said study author Gillian Griffiths, PhD, of the Cambridge Institute for Medical Research in the UK.
“These [CTLs] patrol our bodies, identifying and destroying virally infected and cancer cells, and they do so with remarkable precision and efficiency.”
The CTLs, seen in the video as orange or green amorphous “blobs,” move around rapidly, investigating their environment as they travel.
When a CTL finds a cancer cell (blue), membrane protrusions rapidly explore the surface of the cell, checking for tell-tale signs that this is an uninvited guest.
The CTL binds to the cancer cell and injects cytotoxins (red) down microtubules to the interface between the T cell and the cancer cell, before puncturing the surface of the cancer cell and delivering its deadly cargo.
“In our bodies, where cells are packed together, it’s essential that the T cell focuses the lethal hit on its target,” Dr Griffiths explained. “Otherwise, it will cause collateral damage to neighboring, healthy cells.”
“Once the cytotoxins are injected into the cancer cell, its fate is sealed, and we can watch as it withers and dies. The T cell then moves on, hungry to find another victim.”
The investigators’ video is available on YouTube.

cancer cells (blue)
Image courtesy of Gillian
Griffiths and Jonny Settle
New research has illuminated the behavior of cytotoxic T cells (CTLs) as they hunt down and eliminate cancer cells.
Investigators used novel imaging techniques to capture the process on film and described their findings in an article published in Immunity.
The team captured the footage through high-resolution, 3D, time-lapse, multi-color imaging, making use of both spinning disk confocal microscopy and lattice light sheet microscopy.
These techniques involve capturing “slices” of an object and “stitching” them together to provide the final 3D images across the whole cell.
These approaches allowed the investigators to determine the order of events leading to the lethal “hit” CTLs deliver to cancer cells.
“Inside all of us lurks an army of ‘serial killers’ whose primary function is to kill again and again,” said study author Gillian Griffiths, PhD, of the Cambridge Institute for Medical Research in the UK.
“These [CTLs] patrol our bodies, identifying and destroying virally infected and cancer cells, and they do so with remarkable precision and efficiency.”
The CTLs, seen in the video as orange or green amorphous “blobs,” move around rapidly, investigating their environment as they travel.
When a CTL finds a cancer cell (blue), membrane protrusions rapidly explore the surface of the cell, checking for tell-tale signs that this is an uninvited guest.
The CTL binds to the cancer cell and injects cytotoxins (red) down microtubules to the interface between the T cell and the cancer cell, before puncturing the surface of the cancer cell and delivering its deadly cargo.
“In our bodies, where cells are packed together, it’s essential that the T cell focuses the lethal hit on its target,” Dr Griffiths explained. “Otherwise, it will cause collateral damage to neighboring, healthy cells.”
“Once the cytotoxins are injected into the cancer cell, its fate is sealed, and we can watch as it withers and dies. The T cell then moves on, hungry to find another victim.”
The investigators’ video is available on YouTube.