User login
Retrospective Review of Dual CGRP-Targeted Regimens for Acute and Preventive Treatment of Migraines in a Veteran Population
Retrospective Review of Dual CGRP-Targeted Regimens for Acute and Preventive Treatment of Migraines in a Veteran Population
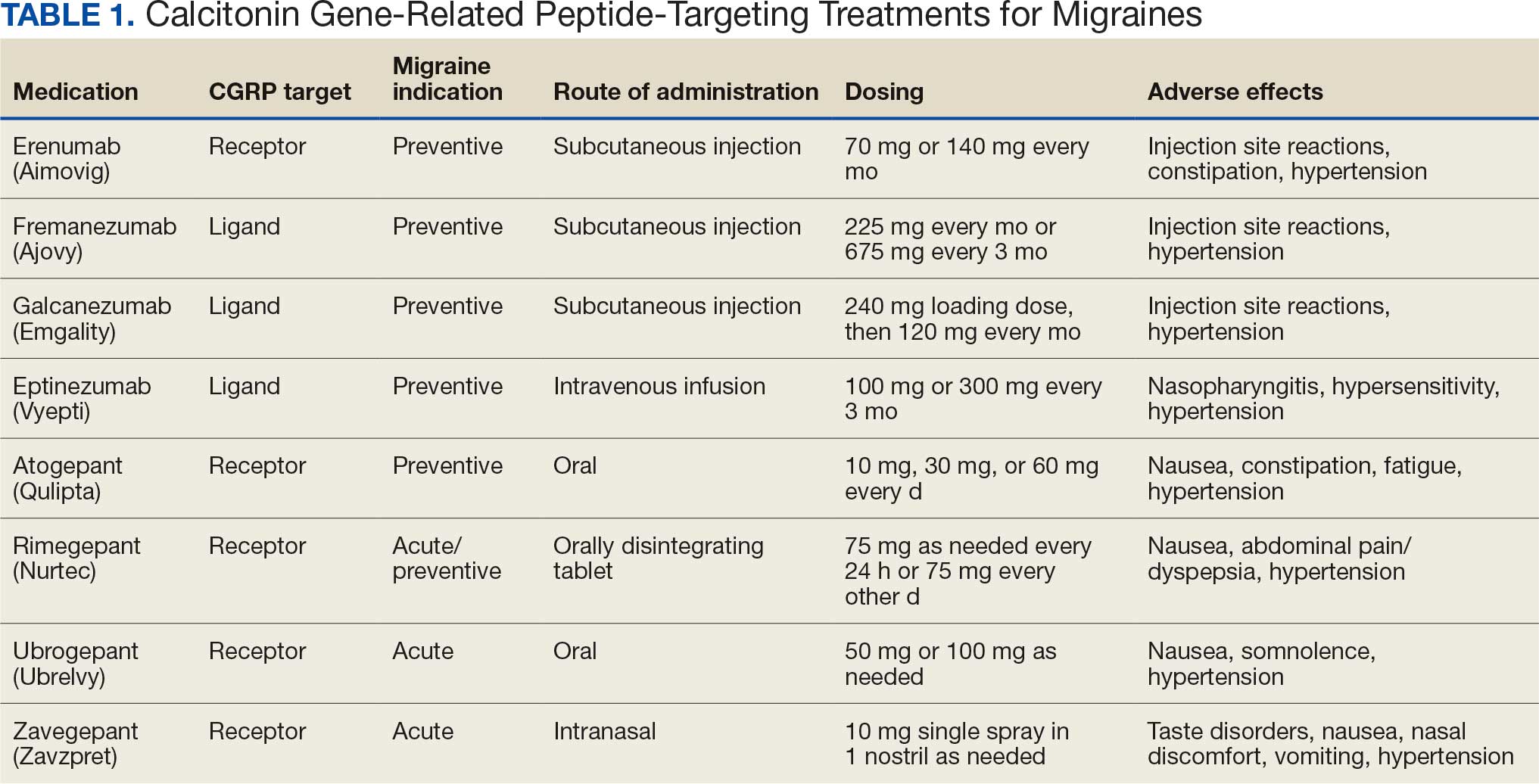
Calcitonin gene-related peptide (CGRP) is a neuropeptide that plays a key role in migraine pathophysiology by promoting the dilation of cerebral blood vessels and transmitting pain signals.1 CGRP has generated interest for the prevention and acute treatment of migraine. Since 2018, 8 novel CGRP-targeting therapies have been approved by the US Food and Drug Administration (FDA) for the management of migraines.2,3 For migraine prevention, there are 4 injectable monoclonal antibodies (mAbs) directed against the CGRP receptor (erenumab) or the CGRP ligand (fremanezumab, galcanezumab, and eptinezumab). There are also 2 oral small-molecule CGRP receptor antagonists, termed gepants, that also are approved for migraine prevention (atogepant and rimegepant). Three gepants are approved for acute migraine treatment and are administered orally (rimegepant and ubrogepant) or intranasally (zavegepant) (Table 1).
CGRP-targeting therapies have received attention for their role in vasodilation within the cerebral, coronary, and renal vasculature.4 CGRP-mediated vasodilatory effects cause systemic regulation of blood pressure (BP) and play a protective role in hypertension.2 Some studies, particularly with erenumab, have shown that the inhibitory role of the agent leads to an increase in BP, as well as gastrointestinal issues such as constipation.2,5 The FDA recently updated monitoring recommendations for all CGRP-targeting therapies to include the potential for BP elevations and hypertension. Outside of this, there is no definitive evidence linking dual CGRP-targeted therapy to higher cardiovascular or gastrointestinal risks and prescribing information does not carry contraindications.6
In a 2021 consensus statement, the American Headache Society (AHS) recommended CGRP-targeting therapies for migraine prevention after inability to tolerate or inadequate response to an 8-week trial of ≥ 2 drug classes including antihypertensives, antiseizure medications, antidepressants, and onabotulinumtoxinA.7 For acute treatment, AHS recommended gepant use after contraindication to or inadequate response to ≥ 2 triptans. Guidance on combination CGRP-targeting therapies for both prevention and acute treatment was not provided.7 More recently, the AHS published a position statement noting substantial efficacy and safety data for CGRP-targeting therapies and suggested its consideration as a first-line option for migraine prevention, though use for acute treatment or combination CGRP-targeting therapies for both prevention and acute treatment were not addressed.8
The International Headache Society guidelines for the acute treatment of migraines recommend nonopioid analgesics as first-line therapy for mild migraine attacks. For moderate to severe attacks, triptans with or without a nonopioid analgesic were recommended as first-line therapy, prior to consideration of CGRP-targeted therapy.9 The increased use of this new drug class has also led to combination use of CGRP-targeting therapies for migraine prevention and acute treatment as seen in clinical practice and reflected by some case reports, case series, and small studies describing such use.10-14 In light of the similar mechanism of action of these therapies and the physiologic role of CGRP, there have been calls for safety evaluation.15
To our knowledge, no studies have evaluated dual CGRP-targeting regimens for migraine in the veteran population. In 2023, the US Department of Veterans Affairs (VA) and US Department of Defense (DoD) updated their clinical practice guidelines for the management of headache.3 For migraine prevention, the VA/DoD guidelines include a strong recommendation for the use of erenumab, fremanezumab, and galcanezumab; a weak recommendation for the use of atogepant; and a recommendation neither for nor against the use of rimegepant. For acute treatment, the guidelines assign a weak recommendation for the use of rimegepant and ubrogepant. Combination use was not addressed.3
Prior to the VA/DoD guidelines, the Veterans Health Administration restricted the dual use of CGRP-targeting therapies for both preventive and acute migraine treatment. However, the VA Pharmacy Benefit Management Service removed the restriction in the Criteria for Use documents, allowing broader access to these medications for veterans.16-22 This change permits the use of CGRP-targeting drugs for both acute and preventive migraine treatment after initial data reflecting real-world case reports and open-label studies suggested possible efficacy without a clear safety concern.11,12 This study aims to fill the gap in the literature by evaluating the safety, efficacy, and overall outcomes of combination CGRP-targeting treatment for migraine prevention and acute treatment in a veteran population.
Methods
This single-center, retrospective, medication use evaluation at the Ralph H. Johnson VA Medical Center (RHJVAMC) was reviewed by the RHJVAMC Research and Development Committee and Quality Improvement Program Evaluation Self Certification Tool, which both determined that institutional review board approval was not required because it was considered part of routine care and quality improvement. Computerized Patient Record System (CPRS) data were reviewed between April 1, 2023 (after the Criteria for Use for CGRP-targeting therapies was updated), through January 31, 2025. Patients were included if they had a confirmed diagnosis of migraine using the International Classification of Headache Disorders, 3rd edition criteria and had concomitant active prescriptions for both a preventive and acute treatment CGRP-targeting agent during the project period.23 Only patients receiving care from the RHJVAMC neurology department were included.
The primary objective was to assess the safety of dual CGRP-targeting therapies for migraine treatment. Key safety endpoints included effects on liver function, kidney function, and BP. Safety outcomes were graded using Common Terminology Criteria for Adverse Events.24 Changes in liver function were categorized as grade 1, 2, or 3 elevations: grade 1 (aspartate aminotransferase [AST]/alanine aminotransferase [ALT] up to 3x the upper limit of normal [ULN] or bilirubin > 1.5 x ULN); grade 2 (AST/ALT 3-6 x ULN or bilirubin 1.5-3 x ULN); and grade 3 (AST/ALT 5-10 x ULN or bilirubin 3-10 x ULN). Kidney function changes were assessed by serum creatinine levels using a similar grading system: Grade 1 (≤ 1.5 x ULN); grade 2 (1.5-3 x baseline of normal); and grade 3 (3-6 x ULN or baseline). Changes in BP were monitored from baseline to the time of the first neurology follow-up. Elevations were grouped into 2 categories, defined as BP ≥ 140 mm Hg systolic and/or 90 mm Hg diastolic (category 1) and ≥ 160 mm Hg systolic and/or 100 mm Hg diastolic (category 2). Neurology documentation was also reviewed in CPRS for individual patient-reported adverse effects (AEs). Safety endpoints were tracked for any occurrence during the project period.
The secondary objective was to describe the patient-reported efficacy of adding a gepant for acute migraine treatment to existing CGRP-targeting therapies for migraine prevention, in those patients who were stable for ≥ 12 weeks on the preventive therapy. Neurology documentation of headache characteristics, including headache severity as rated on a numerical pain score from 0 (no pain) to 10 (worst pain), and duration of headaches (in hours) were recorded during the project period. Changes in headache characteristics were tracked from baseline (ie, the neurology visit when the gepant was first requested) to the first neurology follow-up within 6 months of initiating gepant for acute treatment. If ranges were provided within documentation, a mean was calculated and used for data collection. Neurology documentation was also reviewed for any patient report of overall effectiveness with the added gepant, and categorized as symptoms improved, worsened, or did not change based on subjective report. Descriptive statistics were used for data analysis. A 1-sample Wilcoxon signed rank test was performed as an exploratory analysis for change in headache characteristics from baseline to first neurology follow-up within 6 months. Each individual CGRP regimen was counted as a unique data point to adequately describe changes associated with each new medication and/or dose adjustment. Therefore, patients could be included more than once to account for each distinct treatment regimen.
Results
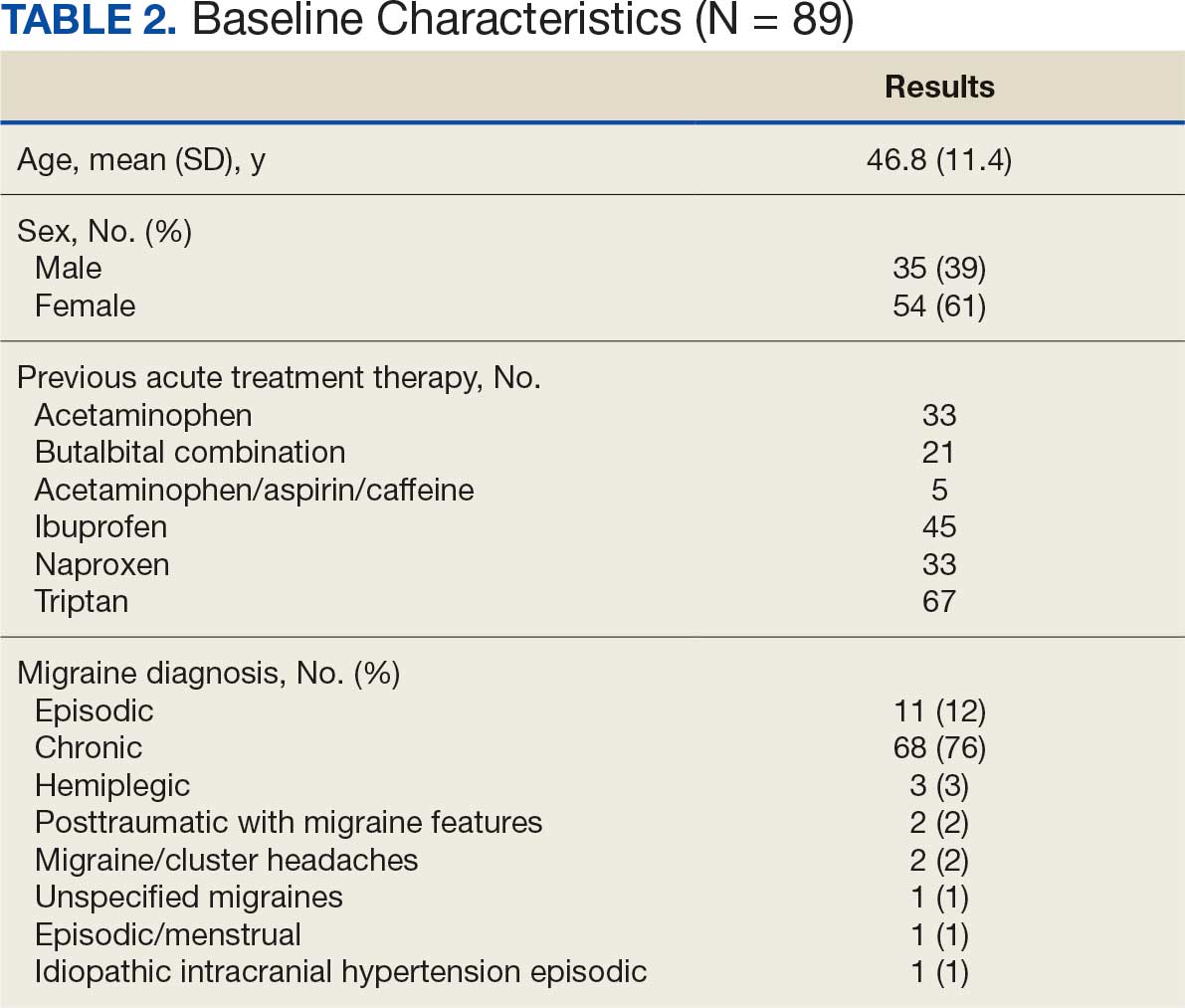
From April 1, 2023, to January 31, 2025, 96 patients were identified with active prescriptions for dual CGRP-targeting therapies. Of the 96 patients, 89 were included in the final analysis; 1 patient lacked a migraine diagnosis and 6 did not have a concomitant dual CGRP-targeted regimen and were excluded. The mean age of patients was 46.8 years and 54 (61%) were female. The most common migraine diagnosis was chronic migraine in 68 patients (76%). Triptans, ibuprofen, and acetaminophen were the most commonly used acute treatment medications (Table 2).
Safety Assessment
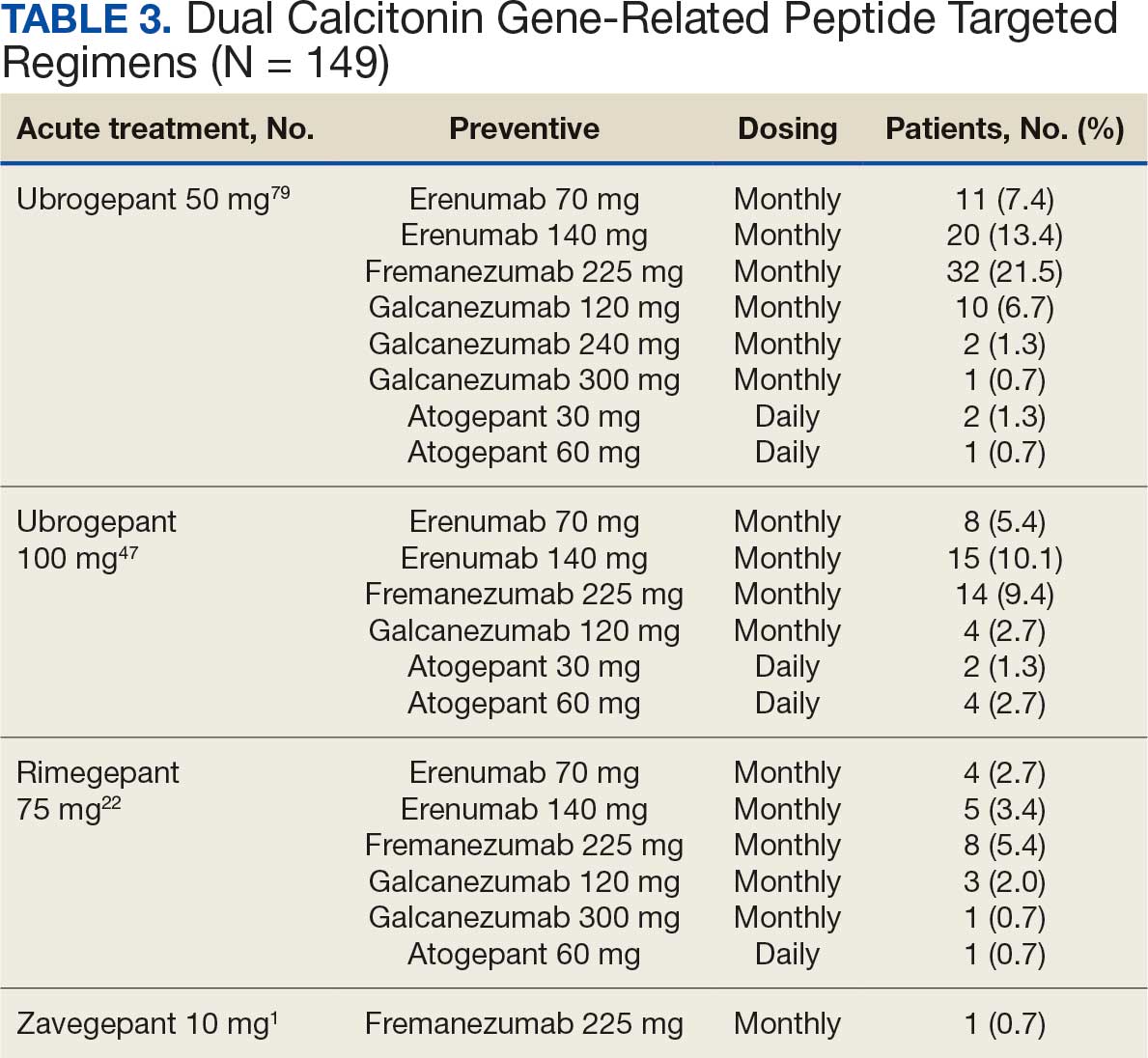
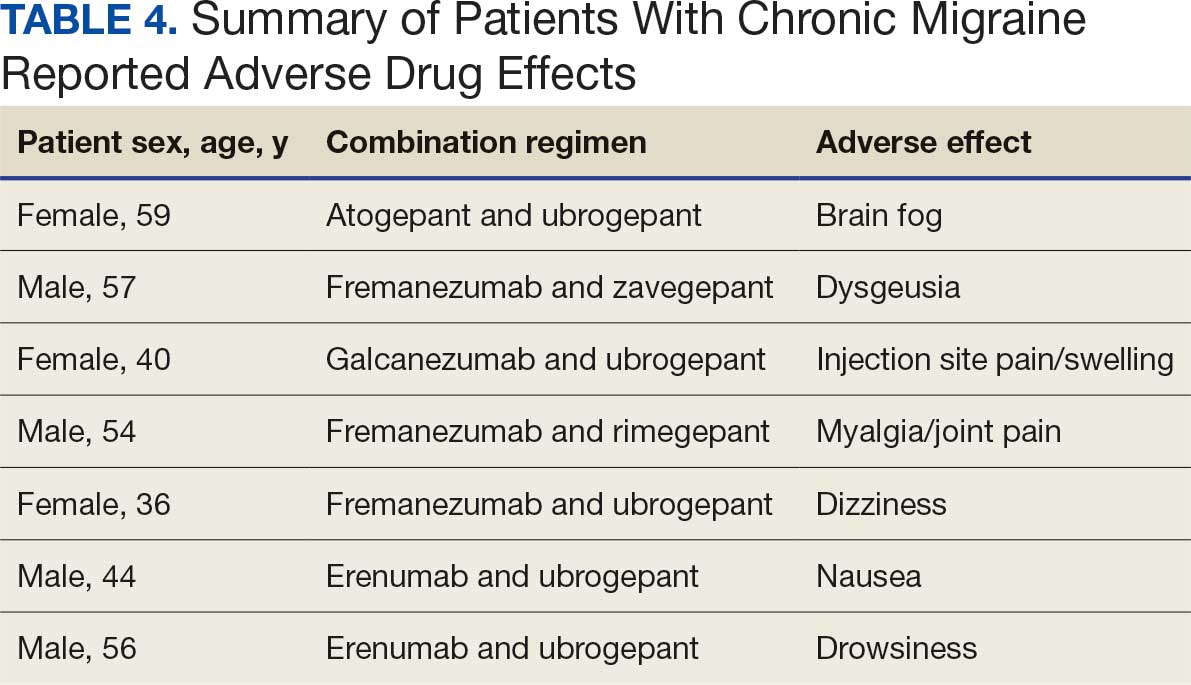
Many of the 89 unique patients trialed > 1 regimen. Thus, for the safety analysis, we analyzed 149 patients on unique dual CGRP-targeting regimens (Table 3). Ubrogepant was used by 126 patients (84.6%) for acute treatment. For preventive therapy, 63 patients (42.3%) used erenumab injections and 55 (36.9%) used fremanezumab injections. Seven patients (4.7%) reported AEs (Table 4). Five of the 7 AEs were noted in the package inserts.25-32 One patient taking both atogepant and ubrogepant reported brain fog that resolved after a dose reduction of atogepant to every other day dosing. A patient taking fremanezumab and rimegepant reported myalgia/joint pain after the first fremanezumab injection, which resolved after a few days and did not recur during the study period.
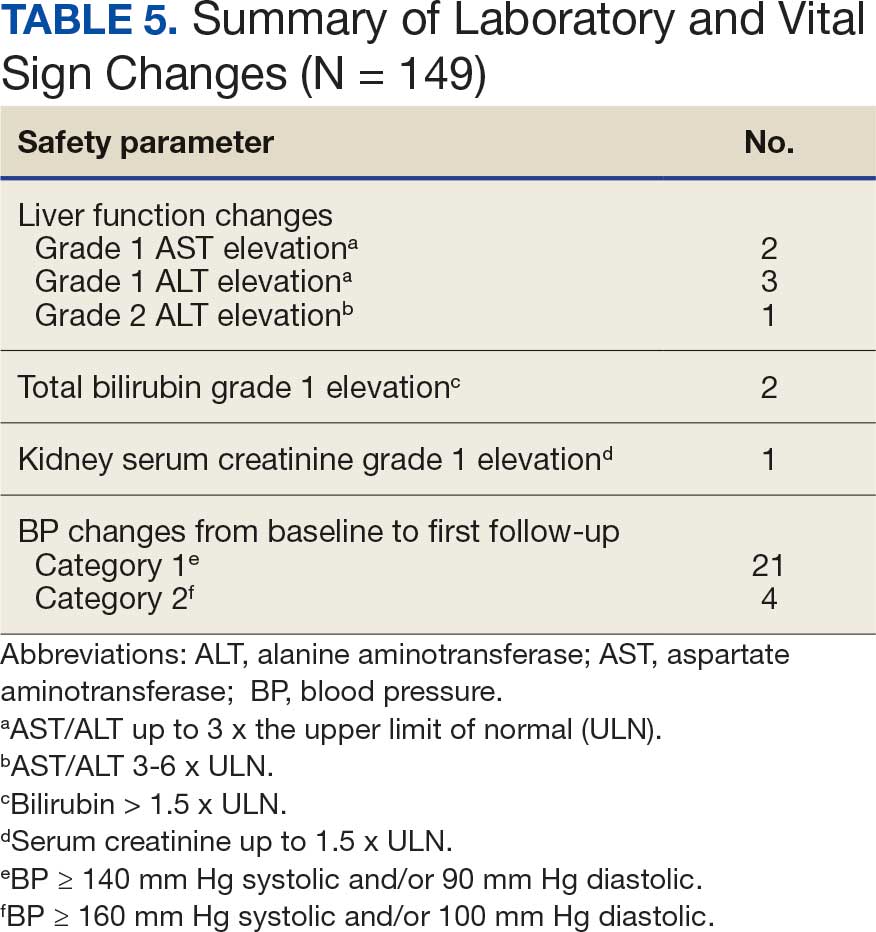
Nine of 149 patient regimens (6.0%) were associated with changes in liver function tests or serum creatinine, though all but 1 were grade 1 (1 patient had a grade 2 ALT elevation). Twenty-five patients (16.8%) experienced changes in BP, most of which were category 1 elevations. Four patients had systolic or diastolic BP ≥ 160 mm Hg or 100 mm Hg, respectively (Table 5).
Efficacy Assessment
Of the 149 unique dual CGRP regimens, 59 were eligible for the exploratory efficacy analysis. Data were excluded from the efficacy analysis if patients had not been on a stable CGRP preventive migraine regimen for ≥ 12 weeks prior to the addition of a gepant. Fourteen regimens were excluded due to a lack of clear documentation on efficacy, leaving 45 analyzed regimens. Of the 45 regimens, 34 were from unique patients. There was no median change in migraine intensity or duration found in the efficacy analysis (0.0, P = .18, and 0.0, P = .92, respectively). Ten patients on dual CGRP therapy reported that the addition of a gepant for acute treatment improved their symptoms, 20 reported that their symptoms were unchanged and/or worsened, and 29 lacked documentation.
Discussion
This study aimed to describe the safety and efficacy of concomitant CGRP regimens for migraine prevention and acute treatment. To our knowledge, this was the first descriptive study of these agents in a veteran population. The potential for increased AEs with concomitant use of CGRP antagonists is due to the similarities in the mechanism of action between the agents, which both target the same receptor/ligand pathway. Given CGRP activity in both the gastrointestinal and cardiovascular systems, the potential for related AEs is speculative. Patient-reported AEs occurred in 7 of 149 unique treatment regimens reviewed for an incidence rate < 5%. All AEs were nonserious and self-limiting.
Our findings are consistent with available research. A 2024 retrospective, exploratory real-world study evaluating the safety and tolerability of combining CGRP-targeting mAbs with gepants reported findings consistent with our results. This analysis included adult patients treated with ≥ 1 previous anti-CGRP mAb and found that 234 of 516 patients included received a combination of a gepant in addition to a CGRP-targeting mAb. Of these 234 patients, 1.3% reported nonserious AEs.33 Similarly, in a multicenter, open-label, long-term safety study in adults experiencing multiple monthly migraine attacks, a subgroup of 13 participants taking a stable dose of an anti-CGRP mAb also took rimegepant 75 mg as needed for acute treatment for 12 weeks. These patients experienced no serious AEs or any AEs leading to discontinuation.14 A study evaluating the drug-drug interaction, safety, and tolerability of dual therapy (atogepant 60 mg daily and ubrogepant 100 mg every 3 days) in 26 patients found no serious AEs, including no significant changes from baseline in laboratory results, vital signs, or safety-related 12-lead electrocardiogram parameters.15The TANDEM real-world, open-label, prospective study demonstrated similar results. It evaluated the safety and tolerability of concomitant use of ubrogepant and atogepant in patients with episodic migraines and found no increase in AEs when comparing atogepant alone with combination therapy. Twenty-six patients (9.9%) discontinued treatment due to AEs. The most common treatment-related AEs were constipation, nausea, decreased appetite, and fatigue. Efficacy data were also noted to be an exploratory endpoint in the TANDEM study; however, results have not been published.12
Within this safety analysis, new onset gastrointestinal AEs, specifically nausea, only occurred in 1 patient. Hypertension occurred in 25 treatment regimens (16.8%) for 21 unique patients (4 BP elevations occurred in 1 patient on 4 different regimens). However, the retrospective nature of reporting may limit accurate assessment. A closer analysis determined that elevated BP readings correlated with elevated pain scores at the time of the readings, which could have factored into the BP elevations. However, ongoing monitoring is needed due to an increased risk of hypertension, particularly given recent FDA labeling updates for CGRP-targeting therapies including gepants. In light of this, and the overall low incidence of hypertension reported, no new safety concerns were identified.
Limitations
Efficacy data in this project were exploratory. This evaluation did not show a significant difference in migraine intensity or duration after adding a gepant for acute treatment. The study was not powered to detect a significant difference. Limited data exist assessing efficacy outcomes with dual CGRP-targeting treatment regimens. The COURAGE study assessed the real-world effectiveness of ubrogepant and CGRP mAbs with or without the addition of onabotulinumtoxinA. The final analysis of the ubrogepant and CGRP mAb arm included 245 total patients and assessed meaningful migraine pain relief, restoration of normal function after a migraine, and treatment satisfaction. By hour 2, 61.6% of patients reported achieving migraine pain relief, rising to 80.4% by hour 4. Return to normal function occurred in 34.7% at hour 2 and 55.5% by hour 4.13 The long-term safety and efficacy of combining erenumab and rimegepant were described in a case series involving 2 patients. Both patients reported that the concomitant CGRP-targeted therapies were effective and reported no AEs.14
The retrospective design of this study meant that there was potential for limited documentation and introduction of bias into the results. Data were collected at a single VA health care system, and thus, results may not be generalizable to a broader population. However, the study population was consistent with the higher incidence of migraine expected in females in the general population. The sample size was limited, particularly in the exploratory efficacy endpoint assessment.
Limitations were observed due to inconsistent documentation regarding headache characteristics, making it challenging to draw meaningful conclusions from this data set. Additional confounding factors, including polypharmacy, nonadherence to medications, and comorbidities, may have skewed results. For example, while our study design required that the preventive CGRP-targeting medication be stable for 12 weeks for inclusion in further efficacy analysis, other medications commonly used for migraine prevention may have been adjusted (which was not accounted for in this analysis). Given this, more large-scale, placebo-controlled, randomized studies are needed to continue to assess the safety and efficacy of these combination treatment regimens.
Conclusions
Few AEs or safety events were reported with combination CGRP-targeting treatment for acute and preventive treatment of migraine. Those that were identified were considered mild. Efficacy data were limited, and further studies are needed to fully assess outcomes.
- Wattiez AS, Sowers LP, Russo AF. Calcitonin gene-related peptide (CGRP): role in migraine pathophysiology and therapeutic targeting. Expert Opin Ther Targets. 2020;24:91-100. doi:10.1080/14728222.2020.1724285
- Shah T, Bedrin K, Tinsley A. Calcitonin gene relating peptide inhibitors in combination for migraine treatment: a mini-review. Front Pain Res (Lausanne). 2023;4:1130239. doi:10.3389/fpain.2023.1130239
- Department of Veterans Affairs/Department of Defense. VA/DoD clinical practice guideline for management of headache. September 2023. Accessed February 4, 2026. https://www.healthquality.va.gov/guidelines/pain/headache/VA-DoD-CPG-Headache-Full-CPG.pdf
- Russell FA, King R, Smillie SJ, et al. Calcitonin gene-related peptide: physiology and pathophysiology. Physiol Rev. 2014;94:1099-1142. doi:10.1152/physrev.00034.2013
- de Vries Lentsch S, van der Arend BWH, VanDenBrink AM, et al. Blood pressure in patients with migraine treated with monoclonal anti-CGRP (receptor) antibodies: a prospective follow-up study. Neurology. 2022;99:e1897-e1904. doi:10.1212/WNL.0000000000201008
- Favoni V, Giani L, Al-Hassany L, et al. CGRP and migraine from a cardiovascular point of view: what do we expect from blocking CGRP?. J Headache Pain. 2019;20:27. doi:10.1186/s10194-019-0979-y
- Ailani J, Burch RC, Robbins MS, et al. The American Headache Society Consensus Statement: update on integrating new migraine treatments into clinical practice. Headache. 2021;61:1021-1039. doi:10.1111/head.14153
- Charles AC, Digre KB, Goadsby PJ, et al. Calcitonin gene-related peptide-targeting therapies are a first-line option for the prevention of migraine: an American Headache Society position statement update. Headache. 2024;64:333-341. doi:10.1111/head.14692
- Puledda F, Sacco S, Diener HC, et al. International Headache Society global practice recommendations for the acute pharmacological treatment of migraine. Cephalalgia. 2024;44:3331024241252666. doi:10.1177/03331024241252666
- Berman G, Croop R, Kudrow D, et al. Safety of rimegepant, an oral CGRP receptor antagonist, plus CGRP monoclonal antibodies for migraine. Headache. 2020;60:1734-1742. doi:10.1111/head.13930
- Blumenfeld AM, Boinpally R, De Abreu Ferreira R, et al. Phase Ib, open-label, fixed-sequence, drug-drug interaction, safety, and tolerability study between atogepant and ubrogepant in participants with a history of migraine. Headache. 2023;63:322-332. doi:10.1111/head.14433
- Ailani J, Lipton RB, Blumenfeld AM, et al. Safety and tolerability of ubrogepant for the acute treatment of migraine in participants taking atogepant for the preventive treatment of episodic migraine: results from the TANDEM study. Headache. 2025;65:1005-1014. doi:10.1111/head.14871
- Lipton RB, Contreras-De Lama J, Serrano D, et al. Real-world use of ubrogepant as acute treatment for migraine with an anti-calcitonin gene-related peptide monoclonal antibody: results from COURAGE. Neurol Ther. 2024;13:69-83. doi:10.1007/s40120-023-00556-8
- Mullin K, Kudrow D, Croop R, et al. Potential for treatment benefit of small molecule CGRP receptor antagonist plus monoclonal antibody in migraine therapy. Neurology. 2020;94:e2121-e2125. doi:10.1212/WNL.0000000000008944
- Ihara K, Takizawa T, Watanabe N, et al. Potential benefits and possible risks of CGRP-targeted multitherapy in migraine. Expert Opin Drug Metab Toxicol. 2024;20:1-4. doi:10.1080/17425255.2024.2316131
- US Department of Veterans Affairs, Pharmacy Benefits Management Services. Ubrogepant (Ubrelvy) criteria for use. July 2025. Accessed March 4, 2026. https://www.va.gov/formularyadvisor/DOC_PDF/CFU_Ubrogepant_UBRELVY_CFU_Rev_Jul_2025.pdf
- US Department of Veterans Affairs, Pharmacy Benefits Management Services. Rimegepant (Nurtec) for abortive migraine treatment criteria for use. July 2025. Accessed March 4, 2026. https://www.va.gov/formularyadvisor/DOC_PDF/CFU_Rimegepant_NURTEC_for_abortive_migraine_CFU_rev_Jul_2025.pdf
- US Department of Veterans Affairs, Pharmacy Benefits Management Services. Rimegepant (Nurtec) for episodic migraine prevention criteria for use. July 2025. Accessed March 4, 2026. https://www.va.gov/formularyadvisor/DOC_PDF/CFU_Rimegepant_NURTEC_for_episodic_migraine_prevention_CFU_rev_Jul_2025.pdf
- US Department of Veterans Affairs, Pharmacy Benefits Management Services. Erenumab-aooe (Aimovig) for chronic migraine prevention criteria for use. July 2025. Accessed March 4, 2026. https://www.va.gov/formularyadvisor/DOC_PDF/CFU_Erenumab_AIMOVIG_for_chronic_migraine_CFU_rev_Jul_2025.pdf
- US Department of Veterans Affairs, Pharmacy Benefits Management Services. Erenumab-aooe (Aimovig) for episodic migraine prevention criteria for use. July 2025. Accessed March 4, 2026. https://www.va.gov/formularyadvisor/DOC_PDF/CFU_Erenumab_AIMOVIG_for_episodic_migraine_CFU_rev_Jul_2025.pdf
- US Department of Veterans Affairs, Pharmacy Benefits Management Services. Galcanezumab-gnlm (Emgality) for cluster headache criteria for use. July 2025. Accessed March 4, 2026. https://www.va.gov/formularyadvisor/DOC_PDF/CFU_Galcanezumab_EMGALITY_for_cluster_headache_CFU_rev_Jul_2025.pdf
- US Department of Veterans Affairs, Pharmacy Benefits Management Services. Atogepant (Qulipta) for chronic migraine prevention criteria for use. July 2025. Accessed March 4, 2026. https://www.va.gov/formularyadvisor/DOC_PDF/CFU_Atogepant_QULIPTA_for_chronic_migraine_prevention_CFU_rev_Jul_2025.pdf
- Headache Classification Committee of the International Headache Society (IHS) The International Classification of Headache Disorders, 3rd edition. Cephalalgia. 2018;38:1-211. doi:10.1177/0333102417738202
- US Dept of Health and Human Services. Common Terminology Criteria for Adverse Events (CTCAE) version 5.0. November 27, 2017. Accessed March 4, 2026. https://dctd.cancer.gov/research/ctep-trials/for-sites/adverse-events/ctcae-v5-5x7.pdf
- Aimovig (erenumab-aooe) injection prescribing information. Amegen Inc. Updated March 2025. Accessed March 4, 2026. https://www.accessdata.fda.gov/drugsatfda_docs/label/2025/761077s026lbl.pdf
- Ajovy (fremanezumab-vfrm) injection prescribing information. Teva Pharmaceuticals. Updated August 2025. Accessed March 4, 2026. https://www.accessdata.fda.gov/drugsatfda_docs/label/2025/761089s031lbl.pdf
- Vyepti (eptinezumab-jjmr) injection prescribing information. Lundbeck Seattle Biopharmaceuticals. Updated October 2025. Accessed March 4, 2026. https://www.accessdata.fda.gov/drugsatfda_docs/label/2025/761119s011lbl.pdf
- Emgality (galcanezumab-gnlm) injection prescribing information. Eli Lilly and Company. Updated March 2025. Accessed March 4, 2026. https://www.accessdata.fda.gov/drugsatfda_docs/label/2025/761063s010lbl.pdf
- Qulipta (atogepant) tablets prescribing information. AbbVie Inc. Updated September 2025. Accessed March 4, 2026. https://www.accessdata.fda.gov/drugsatfda_docs/label/2025/215206s013lbl.pdf
- Nurtec ODT (rimegepant) orally disintegrating tablets prescribing information. Pfzier Labs. Updated August 2025. Accessed March 4, 2026. https://www.accessdata.fda.gov/drugsatfda_docs/label/2025/212728s028lbl.pdf
- Ubrelvy (Ubrogepant) tablets prescribing information. AbbVie Inc. Updated June 2025. Accessed March 4, 2026. https://www.accessdata.fda.gov/drugsatfda_docs/label/2025/211765s012lbl.pdf
- Zavzpret (zavegepant) intranasal spray prescribing information. Pfzier Labs. Updated August 2025. Accessed March 4, 2026. https://www.accessdata.fda.gov/drugsatfda_docs/label/2025/216386s007lbl.pdf
- Alsaadi T, Suliman R, Santos V, et al. Safety and tolerability of combining CGRP monoclonal antibodies with gepants in patients with migraine: a retrospective study. Neurol Ther. 2024;13:465-473. doi:10.1007/s40120-024-00586-w
Calcitonin gene-related peptide (CGRP) is a neuropeptide that plays a key role in migraine pathophysiology by promoting the dilation of cerebral blood vessels and transmitting pain signals.1 CGRP has generated interest for the prevention and acute treatment of migraine. Since 2018, 8 novel CGRP-targeting therapies have been approved by the US Food and Drug Administration (FDA) for the management of migraines.2,3 For migraine prevention, there are 4 injectable monoclonal antibodies (mAbs) directed against the CGRP receptor (erenumab) or the CGRP ligand (fremanezumab, galcanezumab, and eptinezumab). There are also 2 oral small-molecule CGRP receptor antagonists, termed gepants, that also are approved for migraine prevention (atogepant and rimegepant). Three gepants are approved for acute migraine treatment and are administered orally (rimegepant and ubrogepant) or intranasally (zavegepant) (Table 1).

CGRP-targeting therapies have received attention for their role in vasodilation within the cerebral, coronary, and renal vasculature.4 CGRP-mediated vasodilatory effects cause systemic regulation of blood pressure (BP) and play a protective role in hypertension.2 Some studies, particularly with erenumab, have shown that the inhibitory role of the agent leads to an increase in BP, as well as gastrointestinal issues such as constipation.2,5 The FDA recently updated monitoring recommendations for all CGRP-targeting therapies to include the potential for BP elevations and hypertension. Outside of this, there is no definitive evidence linking dual CGRP-targeted therapy to higher cardiovascular or gastrointestinal risks and prescribing information does not carry contraindications.6
In a 2021 consensus statement, the American Headache Society (AHS) recommended CGRP-targeting therapies for migraine prevention after inability to tolerate or inadequate response to an 8-week trial of ≥ 2 drug classes including antihypertensives, antiseizure medications, antidepressants, and onabotulinumtoxinA.7 For acute treatment, AHS recommended gepant use after contraindication to or inadequate response to ≥ 2 triptans. Guidance on combination CGRP-targeting therapies for both prevention and acute treatment was not provided.7 More recently, the AHS published a position statement noting substantial efficacy and safety data for CGRP-targeting therapies and suggested its consideration as a first-line option for migraine prevention, though use for acute treatment or combination CGRP-targeting therapies for both prevention and acute treatment were not addressed.8
The International Headache Society guidelines for the acute treatment of migraines recommend nonopioid analgesics as first-line therapy for mild migraine attacks. For moderate to severe attacks, triptans with or without a nonopioid analgesic were recommended as first-line therapy, prior to consideration of CGRP-targeted therapy.9 The increased use of this new drug class has also led to combination use of CGRP-targeting therapies for migraine prevention and acute treatment as seen in clinical practice and reflected by some case reports, case series, and small studies describing such use.10-14 In light of the similar mechanism of action of these therapies and the physiologic role of CGRP, there have been calls for safety evaluation.15
To our knowledge, no studies have evaluated dual CGRP-targeting regimens for migraine in the veteran population. In 2023, the US Department of Veterans Affairs (VA) and US Department of Defense (DoD) updated their clinical practice guidelines for the management of headache.3 For migraine prevention, the VA/DoD guidelines include a strong recommendation for the use of erenumab, fremanezumab, and galcanezumab; a weak recommendation for the use of atogepant; and a recommendation neither for nor against the use of rimegepant. For acute treatment, the guidelines assign a weak recommendation for the use of rimegepant and ubrogepant. Combination use was not addressed.3
Prior to the VA/DoD guidelines, the Veterans Health Administration restricted the dual use of CGRP-targeting therapies for both preventive and acute migraine treatment. However, the VA Pharmacy Benefit Management Service removed the restriction in the Criteria for Use documents, allowing broader access to these medications for veterans.16-22 This change permits the use of CGRP-targeting drugs for both acute and preventive migraine treatment after initial data reflecting real-world case reports and open-label studies suggested possible efficacy without a clear safety concern.11,12 This study aims to fill the gap in the literature by evaluating the safety, efficacy, and overall outcomes of combination CGRP-targeting treatment for migraine prevention and acute treatment in a veteran population.
Methods
This single-center, retrospective, medication use evaluation at the Ralph H. Johnson VA Medical Center (RHJVAMC) was reviewed by the RHJVAMC Research and Development Committee and Quality Improvement Program Evaluation Self Certification Tool, which both determined that institutional review board approval was not required because it was considered part of routine care and quality improvement. Computerized Patient Record System (CPRS) data were reviewed between April 1, 2023 (after the Criteria for Use for CGRP-targeting therapies was updated), through January 31, 2025. Patients were included if they had a confirmed diagnosis of migraine using the International Classification of Headache Disorders, 3rd edition criteria and had concomitant active prescriptions for both a preventive and acute treatment CGRP-targeting agent during the project period.23 Only patients receiving care from the RHJVAMC neurology department were included.
The primary objective was to assess the safety of dual CGRP-targeting therapies for migraine treatment. Key safety endpoints included effects on liver function, kidney function, and BP. Safety outcomes were graded using Common Terminology Criteria for Adverse Events.24 Changes in liver function were categorized as grade 1, 2, or 3 elevations: grade 1 (aspartate aminotransferase [AST]/alanine aminotransferase [ALT] up to 3x the upper limit of normal [ULN] or bilirubin > 1.5 x ULN); grade 2 (AST/ALT 3-6 x ULN or bilirubin 1.5-3 x ULN); and grade 3 (AST/ALT 5-10 x ULN or bilirubin 3-10 x ULN). Kidney function changes were assessed by serum creatinine levels using a similar grading system: Grade 1 (≤ 1.5 x ULN); grade 2 (1.5-3 x baseline of normal); and grade 3 (3-6 x ULN or baseline). Changes in BP were monitored from baseline to the time of the first neurology follow-up. Elevations were grouped into 2 categories, defined as BP ≥ 140 mm Hg systolic and/or 90 mm Hg diastolic (category 1) and ≥ 160 mm Hg systolic and/or 100 mm Hg diastolic (category 2). Neurology documentation was also reviewed in CPRS for individual patient-reported adverse effects (AEs). Safety endpoints were tracked for any occurrence during the project period.
The secondary objective was to describe the patient-reported efficacy of adding a gepant for acute migraine treatment to existing CGRP-targeting therapies for migraine prevention, in those patients who were stable for ≥ 12 weeks on the preventive therapy. Neurology documentation of headache characteristics, including headache severity as rated on a numerical pain score from 0 (no pain) to 10 (worst pain), and duration of headaches (in hours) were recorded during the project period. Changes in headache characteristics were tracked from baseline (ie, the neurology visit when the gepant was first requested) to the first neurology follow-up within 6 months of initiating gepant for acute treatment. If ranges were provided within documentation, a mean was calculated and used for data collection. Neurology documentation was also reviewed for any patient report of overall effectiveness with the added gepant, and categorized as symptoms improved, worsened, or did not change based on subjective report. Descriptive statistics were used for data analysis. A 1-sample Wilcoxon signed rank test was performed as an exploratory analysis for change in headache characteristics from baseline to first neurology follow-up within 6 months. Each individual CGRP regimen was counted as a unique data point to adequately describe changes associated with each new medication and/or dose adjustment. Therefore, patients could be included more than once to account for each distinct treatment regimen.
Results
From April 1, 2023, to January 31, 2025, 96 patients were identified with active prescriptions for dual CGRP-targeting therapies. Of the 96 patients, 89 were included in the final analysis; 1 patient lacked a migraine diagnosis and 6 did not have a concomitant dual CGRP-targeted regimen and were excluded. The mean age of patients was 46.8 years and 54 (61%) were female. The most common migraine diagnosis was chronic migraine in 68 patients (76%). Triptans, ibuprofen, and acetaminophen were the most commonly used acute treatment medications (Table 2).

Safety Assessment
Many of the 89 unique patients trialed > 1 regimen. Thus, for the safety analysis, we analyzed 149 patients on unique dual CGRP-targeting regimens (Table 3). Ubrogepant was used by 126 patients (84.6%) for acute treatment. For preventive therapy, 63 patients (42.3%) used erenumab injections and 55 (36.9%) used fremanezumab injections. Seven patients (4.7%) reported AEs (Table 4). Five of the 7 AEs were noted in the package inserts.25-32 One patient taking both atogepant and ubrogepant reported brain fog that resolved after a dose reduction of atogepant to every other day dosing. A patient taking fremanezumab and rimegepant reported myalgia/joint pain after the first fremanezumab injection, which resolved after a few days and did not recur during the study period.


Nine of 149 patient regimens (6.0%) were associated with changes in liver function tests or serum creatinine, though all but 1 were grade 1 (1 patient had a grade 2 ALT elevation). Twenty-five patients (16.8%) experienced changes in BP, most of which were category 1 elevations. Four patients had systolic or diastolic BP ≥ 160 mm Hg or 100 mm Hg, respectively (Table 5).

Efficacy Assessment
Of the 149 unique dual CGRP regimens, 59 were eligible for the exploratory efficacy analysis. Data were excluded from the efficacy analysis if patients had not been on a stable CGRP preventive migraine regimen for ≥ 12 weeks prior to the addition of a gepant. Fourteen regimens were excluded due to a lack of clear documentation on efficacy, leaving 45 analyzed regimens. Of the 45 regimens, 34 were from unique patients. There was no median change in migraine intensity or duration found in the efficacy analysis (0.0, P = .18, and 0.0, P = .92, respectively). Ten patients on dual CGRP therapy reported that the addition of a gepant for acute treatment improved their symptoms, 20 reported that their symptoms were unchanged and/or worsened, and 29 lacked documentation.
Discussion
This study aimed to describe the safety and efficacy of concomitant CGRP regimens for migraine prevention and acute treatment. To our knowledge, this was the first descriptive study of these agents in a veteran population. The potential for increased AEs with concomitant use of CGRP antagonists is due to the similarities in the mechanism of action between the agents, which both target the same receptor/ligand pathway. Given CGRP activity in both the gastrointestinal and cardiovascular systems, the potential for related AEs is speculative. Patient-reported AEs occurred in 7 of 149 unique treatment regimens reviewed for an incidence rate < 5%. All AEs were nonserious and self-limiting.
Our findings are consistent with available research. A 2024 retrospective, exploratory real-world study evaluating the safety and tolerability of combining CGRP-targeting mAbs with gepants reported findings consistent with our results. This analysis included adult patients treated with ≥ 1 previous anti-CGRP mAb and found that 234 of 516 patients included received a combination of a gepant in addition to a CGRP-targeting mAb. Of these 234 patients, 1.3% reported nonserious AEs.33 Similarly, in a multicenter, open-label, long-term safety study in adults experiencing multiple monthly migraine attacks, a subgroup of 13 participants taking a stable dose of an anti-CGRP mAb also took rimegepant 75 mg as needed for acute treatment for 12 weeks. These patients experienced no serious AEs or any AEs leading to discontinuation.14 A study evaluating the drug-drug interaction, safety, and tolerability of dual therapy (atogepant 60 mg daily and ubrogepant 100 mg every 3 days) in 26 patients found no serious AEs, including no significant changes from baseline in laboratory results, vital signs, or safety-related 12-lead electrocardiogram parameters.15The TANDEM real-world, open-label, prospective study demonstrated similar results. It evaluated the safety and tolerability of concomitant use of ubrogepant and atogepant in patients with episodic migraines and found no increase in AEs when comparing atogepant alone with combination therapy. Twenty-six patients (9.9%) discontinued treatment due to AEs. The most common treatment-related AEs were constipation, nausea, decreased appetite, and fatigue. Efficacy data were also noted to be an exploratory endpoint in the TANDEM study; however, results have not been published.12
Within this safety analysis, new onset gastrointestinal AEs, specifically nausea, only occurred in 1 patient. Hypertension occurred in 25 treatment regimens (16.8%) for 21 unique patients (4 BP elevations occurred in 1 patient on 4 different regimens). However, the retrospective nature of reporting may limit accurate assessment. A closer analysis determined that elevated BP readings correlated with elevated pain scores at the time of the readings, which could have factored into the BP elevations. However, ongoing monitoring is needed due to an increased risk of hypertension, particularly given recent FDA labeling updates for CGRP-targeting therapies including gepants. In light of this, and the overall low incidence of hypertension reported, no new safety concerns were identified.
Limitations
Efficacy data in this project were exploratory. This evaluation did not show a significant difference in migraine intensity or duration after adding a gepant for acute treatment. The study was not powered to detect a significant difference. Limited data exist assessing efficacy outcomes with dual CGRP-targeting treatment regimens. The COURAGE study assessed the real-world effectiveness of ubrogepant and CGRP mAbs with or without the addition of onabotulinumtoxinA. The final analysis of the ubrogepant and CGRP mAb arm included 245 total patients and assessed meaningful migraine pain relief, restoration of normal function after a migraine, and treatment satisfaction. By hour 2, 61.6% of patients reported achieving migraine pain relief, rising to 80.4% by hour 4. Return to normal function occurred in 34.7% at hour 2 and 55.5% by hour 4.13 The long-term safety and efficacy of combining erenumab and rimegepant were described in a case series involving 2 patients. Both patients reported that the concomitant CGRP-targeted therapies were effective and reported no AEs.14
The retrospective design of this study meant that there was potential for limited documentation and introduction of bias into the results. Data were collected at a single VA health care system, and thus, results may not be generalizable to a broader population. However, the study population was consistent with the higher incidence of migraine expected in females in the general population. The sample size was limited, particularly in the exploratory efficacy endpoint assessment.
Limitations were observed due to inconsistent documentation regarding headache characteristics, making it challenging to draw meaningful conclusions from this data set. Additional confounding factors, including polypharmacy, nonadherence to medications, and comorbidities, may have skewed results. For example, while our study design required that the preventive CGRP-targeting medication be stable for 12 weeks for inclusion in further efficacy analysis, other medications commonly used for migraine prevention may have been adjusted (which was not accounted for in this analysis). Given this, more large-scale, placebo-controlled, randomized studies are needed to continue to assess the safety and efficacy of these combination treatment regimens.
Conclusions
Few AEs or safety events were reported with combination CGRP-targeting treatment for acute and preventive treatment of migraine. Those that were identified were considered mild. Efficacy data were limited, and further studies are needed to fully assess outcomes.
Calcitonin gene-related peptide (CGRP) is a neuropeptide that plays a key role in migraine pathophysiology by promoting the dilation of cerebral blood vessels and transmitting pain signals.1 CGRP has generated interest for the prevention and acute treatment of migraine. Since 2018, 8 novel CGRP-targeting therapies have been approved by the US Food and Drug Administration (FDA) for the management of migraines.2,3 For migraine prevention, there are 4 injectable monoclonal antibodies (mAbs) directed against the CGRP receptor (erenumab) or the CGRP ligand (fremanezumab, galcanezumab, and eptinezumab). There are also 2 oral small-molecule CGRP receptor antagonists, termed gepants, that also are approved for migraine prevention (atogepant and rimegepant). Three gepants are approved for acute migraine treatment and are administered orally (rimegepant and ubrogepant) or intranasally (zavegepant) (Table 1).

CGRP-targeting therapies have received attention for their role in vasodilation within the cerebral, coronary, and renal vasculature.4 CGRP-mediated vasodilatory effects cause systemic regulation of blood pressure (BP) and play a protective role in hypertension.2 Some studies, particularly with erenumab, have shown that the inhibitory role of the agent leads to an increase in BP, as well as gastrointestinal issues such as constipation.2,5 The FDA recently updated monitoring recommendations for all CGRP-targeting therapies to include the potential for BP elevations and hypertension. Outside of this, there is no definitive evidence linking dual CGRP-targeted therapy to higher cardiovascular or gastrointestinal risks and prescribing information does not carry contraindications.6
In a 2021 consensus statement, the American Headache Society (AHS) recommended CGRP-targeting therapies for migraine prevention after inability to tolerate or inadequate response to an 8-week trial of ≥ 2 drug classes including antihypertensives, antiseizure medications, antidepressants, and onabotulinumtoxinA.7 For acute treatment, AHS recommended gepant use after contraindication to or inadequate response to ≥ 2 triptans. Guidance on combination CGRP-targeting therapies for both prevention and acute treatment was not provided.7 More recently, the AHS published a position statement noting substantial efficacy and safety data for CGRP-targeting therapies and suggested its consideration as a first-line option for migraine prevention, though use for acute treatment or combination CGRP-targeting therapies for both prevention and acute treatment were not addressed.8
The International Headache Society guidelines for the acute treatment of migraines recommend nonopioid analgesics as first-line therapy for mild migraine attacks. For moderate to severe attacks, triptans with or without a nonopioid analgesic were recommended as first-line therapy, prior to consideration of CGRP-targeted therapy.9 The increased use of this new drug class has also led to combination use of CGRP-targeting therapies for migraine prevention and acute treatment as seen in clinical practice and reflected by some case reports, case series, and small studies describing such use.10-14 In light of the similar mechanism of action of these therapies and the physiologic role of CGRP, there have been calls for safety evaluation.15
To our knowledge, no studies have evaluated dual CGRP-targeting regimens for migraine in the veteran population. In 2023, the US Department of Veterans Affairs (VA) and US Department of Defense (DoD) updated their clinical practice guidelines for the management of headache.3 For migraine prevention, the VA/DoD guidelines include a strong recommendation for the use of erenumab, fremanezumab, and galcanezumab; a weak recommendation for the use of atogepant; and a recommendation neither for nor against the use of rimegepant. For acute treatment, the guidelines assign a weak recommendation for the use of rimegepant and ubrogepant. Combination use was not addressed.3
Prior to the VA/DoD guidelines, the Veterans Health Administration restricted the dual use of CGRP-targeting therapies for both preventive and acute migraine treatment. However, the VA Pharmacy Benefit Management Service removed the restriction in the Criteria for Use documents, allowing broader access to these medications for veterans.16-22 This change permits the use of CGRP-targeting drugs for both acute and preventive migraine treatment after initial data reflecting real-world case reports and open-label studies suggested possible efficacy without a clear safety concern.11,12 This study aims to fill the gap in the literature by evaluating the safety, efficacy, and overall outcomes of combination CGRP-targeting treatment for migraine prevention and acute treatment in a veteran population.
Methods
This single-center, retrospective, medication use evaluation at the Ralph H. Johnson VA Medical Center (RHJVAMC) was reviewed by the RHJVAMC Research and Development Committee and Quality Improvement Program Evaluation Self Certification Tool, which both determined that institutional review board approval was not required because it was considered part of routine care and quality improvement. Computerized Patient Record System (CPRS) data were reviewed between April 1, 2023 (after the Criteria for Use for CGRP-targeting therapies was updated), through January 31, 2025. Patients were included if they had a confirmed diagnosis of migraine using the International Classification of Headache Disorders, 3rd edition criteria and had concomitant active prescriptions for both a preventive and acute treatment CGRP-targeting agent during the project period.23 Only patients receiving care from the RHJVAMC neurology department were included.
The primary objective was to assess the safety of dual CGRP-targeting therapies for migraine treatment. Key safety endpoints included effects on liver function, kidney function, and BP. Safety outcomes were graded using Common Terminology Criteria for Adverse Events.24 Changes in liver function were categorized as grade 1, 2, or 3 elevations: grade 1 (aspartate aminotransferase [AST]/alanine aminotransferase [ALT] up to 3x the upper limit of normal [ULN] or bilirubin > 1.5 x ULN); grade 2 (AST/ALT 3-6 x ULN or bilirubin 1.5-3 x ULN); and grade 3 (AST/ALT 5-10 x ULN or bilirubin 3-10 x ULN). Kidney function changes were assessed by serum creatinine levels using a similar grading system: Grade 1 (≤ 1.5 x ULN); grade 2 (1.5-3 x baseline of normal); and grade 3 (3-6 x ULN or baseline). Changes in BP were monitored from baseline to the time of the first neurology follow-up. Elevations were grouped into 2 categories, defined as BP ≥ 140 mm Hg systolic and/or 90 mm Hg diastolic (category 1) and ≥ 160 mm Hg systolic and/or 100 mm Hg diastolic (category 2). Neurology documentation was also reviewed in CPRS for individual patient-reported adverse effects (AEs). Safety endpoints were tracked for any occurrence during the project period.
The secondary objective was to describe the patient-reported efficacy of adding a gepant for acute migraine treatment to existing CGRP-targeting therapies for migraine prevention, in those patients who were stable for ≥ 12 weeks on the preventive therapy. Neurology documentation of headache characteristics, including headache severity as rated on a numerical pain score from 0 (no pain) to 10 (worst pain), and duration of headaches (in hours) were recorded during the project period. Changes in headache characteristics were tracked from baseline (ie, the neurology visit when the gepant was first requested) to the first neurology follow-up within 6 months of initiating gepant for acute treatment. If ranges were provided within documentation, a mean was calculated and used for data collection. Neurology documentation was also reviewed for any patient report of overall effectiveness with the added gepant, and categorized as symptoms improved, worsened, or did not change based on subjective report. Descriptive statistics were used for data analysis. A 1-sample Wilcoxon signed rank test was performed as an exploratory analysis for change in headache characteristics from baseline to first neurology follow-up within 6 months. Each individual CGRP regimen was counted as a unique data point to adequately describe changes associated with each new medication and/or dose adjustment. Therefore, patients could be included more than once to account for each distinct treatment regimen.
Results
From April 1, 2023, to January 31, 2025, 96 patients were identified with active prescriptions for dual CGRP-targeting therapies. Of the 96 patients, 89 were included in the final analysis; 1 patient lacked a migraine diagnosis and 6 did not have a concomitant dual CGRP-targeted regimen and were excluded. The mean age of patients was 46.8 years and 54 (61%) were female. The most common migraine diagnosis was chronic migraine in 68 patients (76%). Triptans, ibuprofen, and acetaminophen were the most commonly used acute treatment medications (Table 2).

Safety Assessment
Many of the 89 unique patients trialed > 1 regimen. Thus, for the safety analysis, we analyzed 149 patients on unique dual CGRP-targeting regimens (Table 3). Ubrogepant was used by 126 patients (84.6%) for acute treatment. For preventive therapy, 63 patients (42.3%) used erenumab injections and 55 (36.9%) used fremanezumab injections. Seven patients (4.7%) reported AEs (Table 4). Five of the 7 AEs were noted in the package inserts.25-32 One patient taking both atogepant and ubrogepant reported brain fog that resolved after a dose reduction of atogepant to every other day dosing. A patient taking fremanezumab and rimegepant reported myalgia/joint pain after the first fremanezumab injection, which resolved after a few days and did not recur during the study period.


Nine of 149 patient regimens (6.0%) were associated with changes in liver function tests or serum creatinine, though all but 1 were grade 1 (1 patient had a grade 2 ALT elevation). Twenty-five patients (16.8%) experienced changes in BP, most of which were category 1 elevations. Four patients had systolic or diastolic BP ≥ 160 mm Hg or 100 mm Hg, respectively (Table 5).

Efficacy Assessment
Of the 149 unique dual CGRP regimens, 59 were eligible for the exploratory efficacy analysis. Data were excluded from the efficacy analysis if patients had not been on a stable CGRP preventive migraine regimen for ≥ 12 weeks prior to the addition of a gepant. Fourteen regimens were excluded due to a lack of clear documentation on efficacy, leaving 45 analyzed regimens. Of the 45 regimens, 34 were from unique patients. There was no median change in migraine intensity or duration found in the efficacy analysis (0.0, P = .18, and 0.0, P = .92, respectively). Ten patients on dual CGRP therapy reported that the addition of a gepant for acute treatment improved their symptoms, 20 reported that their symptoms were unchanged and/or worsened, and 29 lacked documentation.
Discussion
This study aimed to describe the safety and efficacy of concomitant CGRP regimens for migraine prevention and acute treatment. To our knowledge, this was the first descriptive study of these agents in a veteran population. The potential for increased AEs with concomitant use of CGRP antagonists is due to the similarities in the mechanism of action between the agents, which both target the same receptor/ligand pathway. Given CGRP activity in both the gastrointestinal and cardiovascular systems, the potential for related AEs is speculative. Patient-reported AEs occurred in 7 of 149 unique treatment regimens reviewed for an incidence rate < 5%. All AEs were nonserious and self-limiting.
Our findings are consistent with available research. A 2024 retrospective, exploratory real-world study evaluating the safety and tolerability of combining CGRP-targeting mAbs with gepants reported findings consistent with our results. This analysis included adult patients treated with ≥ 1 previous anti-CGRP mAb and found that 234 of 516 patients included received a combination of a gepant in addition to a CGRP-targeting mAb. Of these 234 patients, 1.3% reported nonserious AEs.33 Similarly, in a multicenter, open-label, long-term safety study in adults experiencing multiple monthly migraine attacks, a subgroup of 13 participants taking a stable dose of an anti-CGRP mAb also took rimegepant 75 mg as needed for acute treatment for 12 weeks. These patients experienced no serious AEs or any AEs leading to discontinuation.14 A study evaluating the drug-drug interaction, safety, and tolerability of dual therapy (atogepant 60 mg daily and ubrogepant 100 mg every 3 days) in 26 patients found no serious AEs, including no significant changes from baseline in laboratory results, vital signs, or safety-related 12-lead electrocardiogram parameters.15The TANDEM real-world, open-label, prospective study demonstrated similar results. It evaluated the safety and tolerability of concomitant use of ubrogepant and atogepant in patients with episodic migraines and found no increase in AEs when comparing atogepant alone with combination therapy. Twenty-six patients (9.9%) discontinued treatment due to AEs. The most common treatment-related AEs were constipation, nausea, decreased appetite, and fatigue. Efficacy data were also noted to be an exploratory endpoint in the TANDEM study; however, results have not been published.12
Within this safety analysis, new onset gastrointestinal AEs, specifically nausea, only occurred in 1 patient. Hypertension occurred in 25 treatment regimens (16.8%) for 21 unique patients (4 BP elevations occurred in 1 patient on 4 different regimens). However, the retrospective nature of reporting may limit accurate assessment. A closer analysis determined that elevated BP readings correlated with elevated pain scores at the time of the readings, which could have factored into the BP elevations. However, ongoing monitoring is needed due to an increased risk of hypertension, particularly given recent FDA labeling updates for CGRP-targeting therapies including gepants. In light of this, and the overall low incidence of hypertension reported, no new safety concerns were identified.
Limitations
Efficacy data in this project were exploratory. This evaluation did not show a significant difference in migraine intensity or duration after adding a gepant for acute treatment. The study was not powered to detect a significant difference. Limited data exist assessing efficacy outcomes with dual CGRP-targeting treatment regimens. The COURAGE study assessed the real-world effectiveness of ubrogepant and CGRP mAbs with or without the addition of onabotulinumtoxinA. The final analysis of the ubrogepant and CGRP mAb arm included 245 total patients and assessed meaningful migraine pain relief, restoration of normal function after a migraine, and treatment satisfaction. By hour 2, 61.6% of patients reported achieving migraine pain relief, rising to 80.4% by hour 4. Return to normal function occurred in 34.7% at hour 2 and 55.5% by hour 4.13 The long-term safety and efficacy of combining erenumab and rimegepant were described in a case series involving 2 patients. Both patients reported that the concomitant CGRP-targeted therapies were effective and reported no AEs.14
The retrospective design of this study meant that there was potential for limited documentation and introduction of bias into the results. Data were collected at a single VA health care system, and thus, results may not be generalizable to a broader population. However, the study population was consistent with the higher incidence of migraine expected in females in the general population. The sample size was limited, particularly in the exploratory efficacy endpoint assessment.
Limitations were observed due to inconsistent documentation regarding headache characteristics, making it challenging to draw meaningful conclusions from this data set. Additional confounding factors, including polypharmacy, nonadherence to medications, and comorbidities, may have skewed results. For example, while our study design required that the preventive CGRP-targeting medication be stable for 12 weeks for inclusion in further efficacy analysis, other medications commonly used for migraine prevention may have been adjusted (which was not accounted for in this analysis). Given this, more large-scale, placebo-controlled, randomized studies are needed to continue to assess the safety and efficacy of these combination treatment regimens.
Conclusions
Few AEs or safety events were reported with combination CGRP-targeting treatment for acute and preventive treatment of migraine. Those that were identified were considered mild. Efficacy data were limited, and further studies are needed to fully assess outcomes.
- Wattiez AS, Sowers LP, Russo AF. Calcitonin gene-related peptide (CGRP): role in migraine pathophysiology and therapeutic targeting. Expert Opin Ther Targets. 2020;24:91-100. doi:10.1080/14728222.2020.1724285
- Shah T, Bedrin K, Tinsley A. Calcitonin gene relating peptide inhibitors in combination for migraine treatment: a mini-review. Front Pain Res (Lausanne). 2023;4:1130239. doi:10.3389/fpain.2023.1130239
- Department of Veterans Affairs/Department of Defense. VA/DoD clinical practice guideline for management of headache. September 2023. Accessed February 4, 2026. https://www.healthquality.va.gov/guidelines/pain/headache/VA-DoD-CPG-Headache-Full-CPG.pdf
- Russell FA, King R, Smillie SJ, et al. Calcitonin gene-related peptide: physiology and pathophysiology. Physiol Rev. 2014;94:1099-1142. doi:10.1152/physrev.00034.2013
- de Vries Lentsch S, van der Arend BWH, VanDenBrink AM, et al. Blood pressure in patients with migraine treated with monoclonal anti-CGRP (receptor) antibodies: a prospective follow-up study. Neurology. 2022;99:e1897-e1904. doi:10.1212/WNL.0000000000201008
- Favoni V, Giani L, Al-Hassany L, et al. CGRP and migraine from a cardiovascular point of view: what do we expect from blocking CGRP?. J Headache Pain. 2019;20:27. doi:10.1186/s10194-019-0979-y
- Ailani J, Burch RC, Robbins MS, et al. The American Headache Society Consensus Statement: update on integrating new migraine treatments into clinical practice. Headache. 2021;61:1021-1039. doi:10.1111/head.14153
- Charles AC, Digre KB, Goadsby PJ, et al. Calcitonin gene-related peptide-targeting therapies are a first-line option for the prevention of migraine: an American Headache Society position statement update. Headache. 2024;64:333-341. doi:10.1111/head.14692
- Puledda F, Sacco S, Diener HC, et al. International Headache Society global practice recommendations for the acute pharmacological treatment of migraine. Cephalalgia. 2024;44:3331024241252666. doi:10.1177/03331024241252666
- Berman G, Croop R, Kudrow D, et al. Safety of rimegepant, an oral CGRP receptor antagonist, plus CGRP monoclonal antibodies for migraine. Headache. 2020;60:1734-1742. doi:10.1111/head.13930
- Blumenfeld AM, Boinpally R, De Abreu Ferreira R, et al. Phase Ib, open-label, fixed-sequence, drug-drug interaction, safety, and tolerability study between atogepant and ubrogepant in participants with a history of migraine. Headache. 2023;63:322-332. doi:10.1111/head.14433
- Ailani J, Lipton RB, Blumenfeld AM, et al. Safety and tolerability of ubrogepant for the acute treatment of migraine in participants taking atogepant for the preventive treatment of episodic migraine: results from the TANDEM study. Headache. 2025;65:1005-1014. doi:10.1111/head.14871
- Lipton RB, Contreras-De Lama J, Serrano D, et al. Real-world use of ubrogepant as acute treatment for migraine with an anti-calcitonin gene-related peptide monoclonal antibody: results from COURAGE. Neurol Ther. 2024;13:69-83. doi:10.1007/s40120-023-00556-8
- Mullin K, Kudrow D, Croop R, et al. Potential for treatment benefit of small molecule CGRP receptor antagonist plus monoclonal antibody in migraine therapy. Neurology. 2020;94:e2121-e2125. doi:10.1212/WNL.0000000000008944
- Ihara K, Takizawa T, Watanabe N, et al. Potential benefits and possible risks of CGRP-targeted multitherapy in migraine. Expert Opin Drug Metab Toxicol. 2024;20:1-4. doi:10.1080/17425255.2024.2316131
- US Department of Veterans Affairs, Pharmacy Benefits Management Services. Ubrogepant (Ubrelvy) criteria for use. July 2025. Accessed March 4, 2026. https://www.va.gov/formularyadvisor/DOC_PDF/CFU_Ubrogepant_UBRELVY_CFU_Rev_Jul_2025.pdf
- US Department of Veterans Affairs, Pharmacy Benefits Management Services. Rimegepant (Nurtec) for abortive migraine treatment criteria for use. July 2025. Accessed March 4, 2026. https://www.va.gov/formularyadvisor/DOC_PDF/CFU_Rimegepant_NURTEC_for_abortive_migraine_CFU_rev_Jul_2025.pdf
- US Department of Veterans Affairs, Pharmacy Benefits Management Services. Rimegepant (Nurtec) for episodic migraine prevention criteria for use. July 2025. Accessed March 4, 2026. https://www.va.gov/formularyadvisor/DOC_PDF/CFU_Rimegepant_NURTEC_for_episodic_migraine_prevention_CFU_rev_Jul_2025.pdf
- US Department of Veterans Affairs, Pharmacy Benefits Management Services. Erenumab-aooe (Aimovig) for chronic migraine prevention criteria for use. July 2025. Accessed March 4, 2026. https://www.va.gov/formularyadvisor/DOC_PDF/CFU_Erenumab_AIMOVIG_for_chronic_migraine_CFU_rev_Jul_2025.pdf
- US Department of Veterans Affairs, Pharmacy Benefits Management Services. Erenumab-aooe (Aimovig) for episodic migraine prevention criteria for use. July 2025. Accessed March 4, 2026. https://www.va.gov/formularyadvisor/DOC_PDF/CFU_Erenumab_AIMOVIG_for_episodic_migraine_CFU_rev_Jul_2025.pdf
- US Department of Veterans Affairs, Pharmacy Benefits Management Services. Galcanezumab-gnlm (Emgality) for cluster headache criteria for use. July 2025. Accessed March 4, 2026. https://www.va.gov/formularyadvisor/DOC_PDF/CFU_Galcanezumab_EMGALITY_for_cluster_headache_CFU_rev_Jul_2025.pdf
- US Department of Veterans Affairs, Pharmacy Benefits Management Services. Atogepant (Qulipta) for chronic migraine prevention criteria for use. July 2025. Accessed March 4, 2026. https://www.va.gov/formularyadvisor/DOC_PDF/CFU_Atogepant_QULIPTA_for_chronic_migraine_prevention_CFU_rev_Jul_2025.pdf
- Headache Classification Committee of the International Headache Society (IHS) The International Classification of Headache Disorders, 3rd edition. Cephalalgia. 2018;38:1-211. doi:10.1177/0333102417738202
- US Dept of Health and Human Services. Common Terminology Criteria for Adverse Events (CTCAE) version 5.0. November 27, 2017. Accessed March 4, 2026. https://dctd.cancer.gov/research/ctep-trials/for-sites/adverse-events/ctcae-v5-5x7.pdf
- Aimovig (erenumab-aooe) injection prescribing information. Amegen Inc. Updated March 2025. Accessed March 4, 2026. https://www.accessdata.fda.gov/drugsatfda_docs/label/2025/761077s026lbl.pdf
- Ajovy (fremanezumab-vfrm) injection prescribing information. Teva Pharmaceuticals. Updated August 2025. Accessed March 4, 2026. https://www.accessdata.fda.gov/drugsatfda_docs/label/2025/761089s031lbl.pdf
- Vyepti (eptinezumab-jjmr) injection prescribing information. Lundbeck Seattle Biopharmaceuticals. Updated October 2025. Accessed March 4, 2026. https://www.accessdata.fda.gov/drugsatfda_docs/label/2025/761119s011lbl.pdf
- Emgality (galcanezumab-gnlm) injection prescribing information. Eli Lilly and Company. Updated March 2025. Accessed March 4, 2026. https://www.accessdata.fda.gov/drugsatfda_docs/label/2025/761063s010lbl.pdf
- Qulipta (atogepant) tablets prescribing information. AbbVie Inc. Updated September 2025. Accessed March 4, 2026. https://www.accessdata.fda.gov/drugsatfda_docs/label/2025/215206s013lbl.pdf
- Nurtec ODT (rimegepant) orally disintegrating tablets prescribing information. Pfzier Labs. Updated August 2025. Accessed March 4, 2026. https://www.accessdata.fda.gov/drugsatfda_docs/label/2025/212728s028lbl.pdf
- Ubrelvy (Ubrogepant) tablets prescribing information. AbbVie Inc. Updated June 2025. Accessed March 4, 2026. https://www.accessdata.fda.gov/drugsatfda_docs/label/2025/211765s012lbl.pdf
- Zavzpret (zavegepant) intranasal spray prescribing information. Pfzier Labs. Updated August 2025. Accessed March 4, 2026. https://www.accessdata.fda.gov/drugsatfda_docs/label/2025/216386s007lbl.pdf
- Alsaadi T, Suliman R, Santos V, et al. Safety and tolerability of combining CGRP monoclonal antibodies with gepants in patients with migraine: a retrospective study. Neurol Ther. 2024;13:465-473. doi:10.1007/s40120-024-00586-w
- Wattiez AS, Sowers LP, Russo AF. Calcitonin gene-related peptide (CGRP): role in migraine pathophysiology and therapeutic targeting. Expert Opin Ther Targets. 2020;24:91-100. doi:10.1080/14728222.2020.1724285
- Shah T, Bedrin K, Tinsley A. Calcitonin gene relating peptide inhibitors in combination for migraine treatment: a mini-review. Front Pain Res (Lausanne). 2023;4:1130239. doi:10.3389/fpain.2023.1130239
- Department of Veterans Affairs/Department of Defense. VA/DoD clinical practice guideline for management of headache. September 2023. Accessed February 4, 2026. https://www.healthquality.va.gov/guidelines/pain/headache/VA-DoD-CPG-Headache-Full-CPG.pdf
- Russell FA, King R, Smillie SJ, et al. Calcitonin gene-related peptide: physiology and pathophysiology. Physiol Rev. 2014;94:1099-1142. doi:10.1152/physrev.00034.2013
- de Vries Lentsch S, van der Arend BWH, VanDenBrink AM, et al. Blood pressure in patients with migraine treated with monoclonal anti-CGRP (receptor) antibodies: a prospective follow-up study. Neurology. 2022;99:e1897-e1904. doi:10.1212/WNL.0000000000201008
- Favoni V, Giani L, Al-Hassany L, et al. CGRP and migraine from a cardiovascular point of view: what do we expect from blocking CGRP?. J Headache Pain. 2019;20:27. doi:10.1186/s10194-019-0979-y
- Ailani J, Burch RC, Robbins MS, et al. The American Headache Society Consensus Statement: update on integrating new migraine treatments into clinical practice. Headache. 2021;61:1021-1039. doi:10.1111/head.14153
- Charles AC, Digre KB, Goadsby PJ, et al. Calcitonin gene-related peptide-targeting therapies are a first-line option for the prevention of migraine: an American Headache Society position statement update. Headache. 2024;64:333-341. doi:10.1111/head.14692
- Puledda F, Sacco S, Diener HC, et al. International Headache Society global practice recommendations for the acute pharmacological treatment of migraine. Cephalalgia. 2024;44:3331024241252666. doi:10.1177/03331024241252666
- Berman G, Croop R, Kudrow D, et al. Safety of rimegepant, an oral CGRP receptor antagonist, plus CGRP monoclonal antibodies for migraine. Headache. 2020;60:1734-1742. doi:10.1111/head.13930
- Blumenfeld AM, Boinpally R, De Abreu Ferreira R, et al. Phase Ib, open-label, fixed-sequence, drug-drug interaction, safety, and tolerability study between atogepant and ubrogepant in participants with a history of migraine. Headache. 2023;63:322-332. doi:10.1111/head.14433
- Ailani J, Lipton RB, Blumenfeld AM, et al. Safety and tolerability of ubrogepant for the acute treatment of migraine in participants taking atogepant for the preventive treatment of episodic migraine: results from the TANDEM study. Headache. 2025;65:1005-1014. doi:10.1111/head.14871
- Lipton RB, Contreras-De Lama J, Serrano D, et al. Real-world use of ubrogepant as acute treatment for migraine with an anti-calcitonin gene-related peptide monoclonal antibody: results from COURAGE. Neurol Ther. 2024;13:69-83. doi:10.1007/s40120-023-00556-8
- Mullin K, Kudrow D, Croop R, et al. Potential for treatment benefit of small molecule CGRP receptor antagonist plus monoclonal antibody in migraine therapy. Neurology. 2020;94:e2121-e2125. doi:10.1212/WNL.0000000000008944
- Ihara K, Takizawa T, Watanabe N, et al. Potential benefits and possible risks of CGRP-targeted multitherapy in migraine. Expert Opin Drug Metab Toxicol. 2024;20:1-4. doi:10.1080/17425255.2024.2316131
- US Department of Veterans Affairs, Pharmacy Benefits Management Services. Ubrogepant (Ubrelvy) criteria for use. July 2025. Accessed March 4, 2026. https://www.va.gov/formularyadvisor/DOC_PDF/CFU_Ubrogepant_UBRELVY_CFU_Rev_Jul_2025.pdf
- US Department of Veterans Affairs, Pharmacy Benefits Management Services. Rimegepant (Nurtec) for abortive migraine treatment criteria for use. July 2025. Accessed March 4, 2026. https://www.va.gov/formularyadvisor/DOC_PDF/CFU_Rimegepant_NURTEC_for_abortive_migraine_CFU_rev_Jul_2025.pdf
- US Department of Veterans Affairs, Pharmacy Benefits Management Services. Rimegepant (Nurtec) for episodic migraine prevention criteria for use. July 2025. Accessed March 4, 2026. https://www.va.gov/formularyadvisor/DOC_PDF/CFU_Rimegepant_NURTEC_for_episodic_migraine_prevention_CFU_rev_Jul_2025.pdf
- US Department of Veterans Affairs, Pharmacy Benefits Management Services. Erenumab-aooe (Aimovig) for chronic migraine prevention criteria for use. July 2025. Accessed March 4, 2026. https://www.va.gov/formularyadvisor/DOC_PDF/CFU_Erenumab_AIMOVIG_for_chronic_migraine_CFU_rev_Jul_2025.pdf
- US Department of Veterans Affairs, Pharmacy Benefits Management Services. Erenumab-aooe (Aimovig) for episodic migraine prevention criteria for use. July 2025. Accessed March 4, 2026. https://www.va.gov/formularyadvisor/DOC_PDF/CFU_Erenumab_AIMOVIG_for_episodic_migraine_CFU_rev_Jul_2025.pdf
- US Department of Veterans Affairs, Pharmacy Benefits Management Services. Galcanezumab-gnlm (Emgality) for cluster headache criteria for use. July 2025. Accessed March 4, 2026. https://www.va.gov/formularyadvisor/DOC_PDF/CFU_Galcanezumab_EMGALITY_for_cluster_headache_CFU_rev_Jul_2025.pdf
- US Department of Veterans Affairs, Pharmacy Benefits Management Services. Atogepant (Qulipta) for chronic migraine prevention criteria for use. July 2025. Accessed March 4, 2026. https://www.va.gov/formularyadvisor/DOC_PDF/CFU_Atogepant_QULIPTA_for_chronic_migraine_prevention_CFU_rev_Jul_2025.pdf
- Headache Classification Committee of the International Headache Society (IHS) The International Classification of Headache Disorders, 3rd edition. Cephalalgia. 2018;38:1-211. doi:10.1177/0333102417738202
- US Dept of Health and Human Services. Common Terminology Criteria for Adverse Events (CTCAE) version 5.0. November 27, 2017. Accessed March 4, 2026. https://dctd.cancer.gov/research/ctep-trials/for-sites/adverse-events/ctcae-v5-5x7.pdf
- Aimovig (erenumab-aooe) injection prescribing information. Amegen Inc. Updated March 2025. Accessed March 4, 2026. https://www.accessdata.fda.gov/drugsatfda_docs/label/2025/761077s026lbl.pdf
- Ajovy (fremanezumab-vfrm) injection prescribing information. Teva Pharmaceuticals. Updated August 2025. Accessed March 4, 2026. https://www.accessdata.fda.gov/drugsatfda_docs/label/2025/761089s031lbl.pdf
- Vyepti (eptinezumab-jjmr) injection prescribing information. Lundbeck Seattle Biopharmaceuticals. Updated October 2025. Accessed March 4, 2026. https://www.accessdata.fda.gov/drugsatfda_docs/label/2025/761119s011lbl.pdf
- Emgality (galcanezumab-gnlm) injection prescribing information. Eli Lilly and Company. Updated March 2025. Accessed March 4, 2026. https://www.accessdata.fda.gov/drugsatfda_docs/label/2025/761063s010lbl.pdf
- Qulipta (atogepant) tablets prescribing information. AbbVie Inc. Updated September 2025. Accessed March 4, 2026. https://www.accessdata.fda.gov/drugsatfda_docs/label/2025/215206s013lbl.pdf
- Nurtec ODT (rimegepant) orally disintegrating tablets prescribing information. Pfzier Labs. Updated August 2025. Accessed March 4, 2026. https://www.accessdata.fda.gov/drugsatfda_docs/label/2025/212728s028lbl.pdf
- Ubrelvy (Ubrogepant) tablets prescribing information. AbbVie Inc. Updated June 2025. Accessed March 4, 2026. https://www.accessdata.fda.gov/drugsatfda_docs/label/2025/211765s012lbl.pdf
- Zavzpret (zavegepant) intranasal spray prescribing information. Pfzier Labs. Updated August 2025. Accessed March 4, 2026. https://www.accessdata.fda.gov/drugsatfda_docs/label/2025/216386s007lbl.pdf
- Alsaadi T, Suliman R, Santos V, et al. Safety and tolerability of combining CGRP monoclonal antibodies with gepants in patients with migraine: a retrospective study. Neurol Ther. 2024;13:465-473. doi:10.1007/s40120-024-00586-w
Retrospective Review of Dual CGRP-Targeted Regimens for Acute and Preventive Treatment of Migraines in a Veteran Population
Retrospective Review of Dual CGRP-Targeted Regimens for Acute and Preventive Treatment of Migraines in a Veteran Population