User login
Retrospective Review of Dual CGRP-Targeted Regimens for Acute and Preventive Treatment of Migraines in a Veteran Population
Retrospective Review of Dual CGRP-Targeted Regimens for Acute and Preventive Treatment of Migraines in a Veteran Population
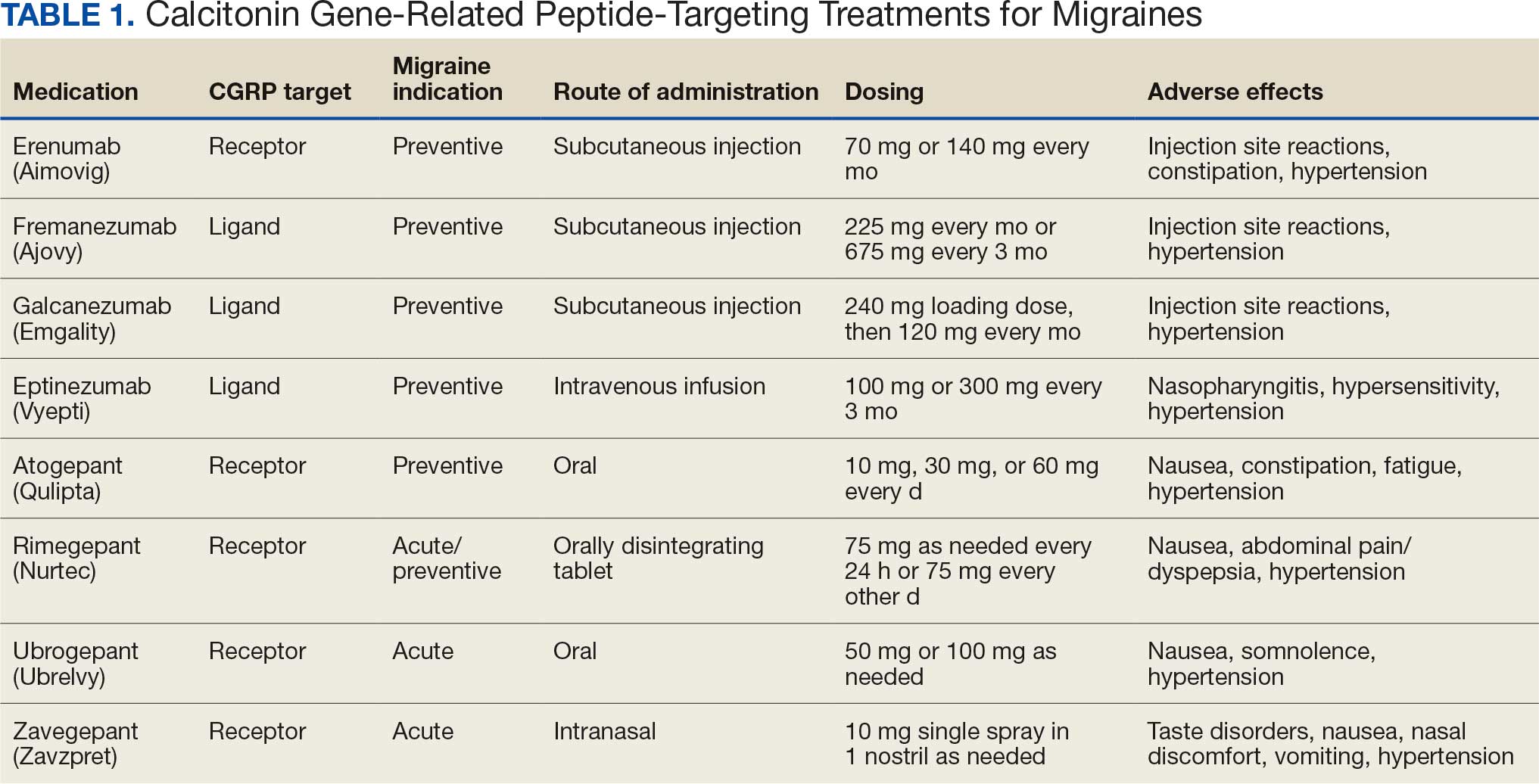
Calcitonin gene-related peptide (CGRP) is a neuropeptide that plays a key role in migraine pathophysiology by promoting the dilation of cerebral blood vessels and transmitting pain signals.1 CGRP has generated interest for the prevention and acute treatment of migraine. Since 2018, 8 novel CGRP-targeting therapies have been approved by the US Food and Drug Administration (FDA) for the management of migraines.2,3 For migraine prevention, there are 4 injectable monoclonal antibodies (mAbs) directed against the CGRP receptor (erenumab) or the CGRP ligand (fremanezumab, galcanezumab, and eptinezumab). There are also 2 oral small-molecule CGRP receptor antagonists, termed gepants, that also are approved for migraine prevention (atogepant and rimegepant). Three gepants are approved for acute migraine treatment and are administered orally (rimegepant and ubrogepant) or intranasally (zavegepant) (Table 1).
CGRP-targeting therapies have received attention for their role in vasodilation within the cerebral, coronary, and renal vasculature.4 CGRP-mediated vasodilatory effects cause systemic regulation of blood pressure (BP) and play a protective role in hypertension.2 Some studies, particularly with erenumab, have shown that the inhibitory role of the agent leads to an increase in BP, as well as gastrointestinal issues such as constipation.2,5 The FDA recently updated monitoring recommendations for all CGRP-targeting therapies to include the potential for BP elevations and hypertension. Outside of this, there is no definitive evidence linking dual CGRP-targeted therapy to higher cardiovascular or gastrointestinal risks and prescribing information does not carry contraindications.6
In a 2021 consensus statement, the American Headache Society (AHS) recommended CGRP-targeting therapies for migraine prevention after inability to tolerate or inadequate response to an 8-week trial of ≥ 2 drug classes including antihypertensives, antiseizure medications, antidepressants, and onabotulinumtoxinA.7 For acute treatment, AHS recommended gepant use after contraindication to or inadequate response to ≥ 2 triptans. Guidance on combination CGRP-targeting therapies for both prevention and acute treatment was not provided.7 More recently, the AHS published a position statement noting substantial efficacy and safety data for CGRP-targeting therapies and suggested its consideration as a first-line option for migraine prevention, though use for acute treatment or combination CGRP-targeting therapies for both prevention and acute treatment were not addressed.8
The International Headache Society guidelines for the acute treatment of migraines recommend nonopioid analgesics as first-line therapy for mild migraine attacks. For moderate to severe attacks, triptans with or without a nonopioid analgesic were recommended as first-line therapy, prior to consideration of CGRP-targeted therapy.9 The increased use of this new drug class has also led to combination use of CGRP-targeting therapies for migraine prevention and acute treatment as seen in clinical practice and reflected by some case reports, case series, and small studies describing such use.10-14 In light of the similar mechanism of action of these therapies and the physiologic role of CGRP, there have been calls for safety evaluation.15
To our knowledge, no studies have evaluated dual CGRP-targeting regimens for migraine in the veteran population. In 2023, the US Department of Veterans Affairs (VA) and US Department of Defense (DoD) updated their clinical practice guidelines for the management of headache.3 For migraine prevention, the VA/DoD guidelines include a strong recommendation for the use of erenumab, fremanezumab, and galcanezumab; a weak recommendation for the use of atogepant; and a recommendation neither for nor against the use of rimegepant. For acute treatment, the guidelines assign a weak recommendation for the use of rimegepant and ubrogepant. Combination use was not addressed.3
Prior to the VA/DoD guidelines, the Veterans Health Administration restricted the dual use of CGRP-targeting therapies for both preventive and acute migraine treatment. However, the VA Pharmacy Benefit Management Service removed the restriction in the Criteria for Use documents, allowing broader access to these medications for veterans.16-22 This change permits the use of CGRP-targeting drugs for both acute and preventive migraine treatment after initial data reflecting real-world case reports and open-label studies suggested possible efficacy without a clear safety concern.11,12 This study aims to fill the gap in the literature by evaluating the safety, efficacy, and overall outcomes of combination CGRP-targeting treatment for migraine prevention and acute treatment in a veteran population.
Methods
This single-center, retrospective, medication use evaluation at the Ralph H. Johnson VA Medical Center (RHJVAMC) was reviewed by the RHJVAMC Research and Development Committee and Quality Improvement Program Evaluation Self Certification Tool, which both determined that institutional review board approval was not required because it was considered part of routine care and quality improvement. Computerized Patient Record System (CPRS) data were reviewed between April 1, 2023 (after the Criteria for Use for CGRP-targeting therapies was updated), through January 31, 2025. Patients were included if they had a confirmed diagnosis of migraine using the International Classification of Headache Disorders, 3rd edition criteria and had concomitant active prescriptions for both a preventive and acute treatment CGRP-targeting agent during the project period.23 Only patients receiving care from the RHJVAMC neurology department were included.
The primary objective was to assess the safety of dual CGRP-targeting therapies for migraine treatment. Key safety endpoints included effects on liver function, kidney function, and BP. Safety outcomes were graded using Common Terminology Criteria for Adverse Events.24 Changes in liver function were categorized as grade 1, 2, or 3 elevations: grade 1 (aspartate aminotransferase [AST]/alanine aminotransferase [ALT] up to 3x the upper limit of normal [ULN] or bilirubin > 1.5 x ULN); grade 2 (AST/ALT 3-6 x ULN or bilirubin 1.5-3 x ULN); and grade 3 (AST/ALT 5-10 x ULN or bilirubin 3-10 x ULN). Kidney function changes were assessed by serum creatinine levels using a similar grading system: Grade 1 (≤ 1.5 x ULN); grade 2 (1.5-3 x baseline of normal); and grade 3 (3-6 x ULN or baseline). Changes in BP were monitored from baseline to the time of the first neurology follow-up. Elevations were grouped into 2 categories, defined as BP ≥ 140 mm Hg systolic and/or 90 mm Hg diastolic (category 1) and ≥ 160 mm Hg systolic and/or 100 mm Hg diastolic (category 2). Neurology documentation was also reviewed in CPRS for individual patient-reported adverse effects (AEs). Safety endpoints were tracked for any occurrence during the project period.
The secondary objective was to describe the patient-reported efficacy of adding a gepant for acute migraine treatment to existing CGRP-targeting therapies for migraine prevention, in those patients who were stable for ≥ 12 weeks on the preventive therapy. Neurology documentation of headache characteristics, including headache severity as rated on a numerical pain score from 0 (no pain) to 10 (worst pain), and duration of headaches (in hours) were recorded during the project period. Changes in headache characteristics were tracked from baseline (ie, the neurology visit when the gepant was first requested) to the first neurology follow-up within 6 months of initiating gepant for acute treatment. If ranges were provided within documentation, a mean was calculated and used for data collection. Neurology documentation was also reviewed for any patient report of overall effectiveness with the added gepant, and categorized as symptoms improved, worsened, or did not change based on subjective report. Descriptive statistics were used for data analysis. A 1-sample Wilcoxon signed rank test was performed as an exploratory analysis for change in headache characteristics from baseline to first neurology follow-up within 6 months. Each individual CGRP regimen was counted as a unique data point to adequately describe changes associated with each new medication and/or dose adjustment. Therefore, patients could be included more than once to account for each distinct treatment regimen.
Results
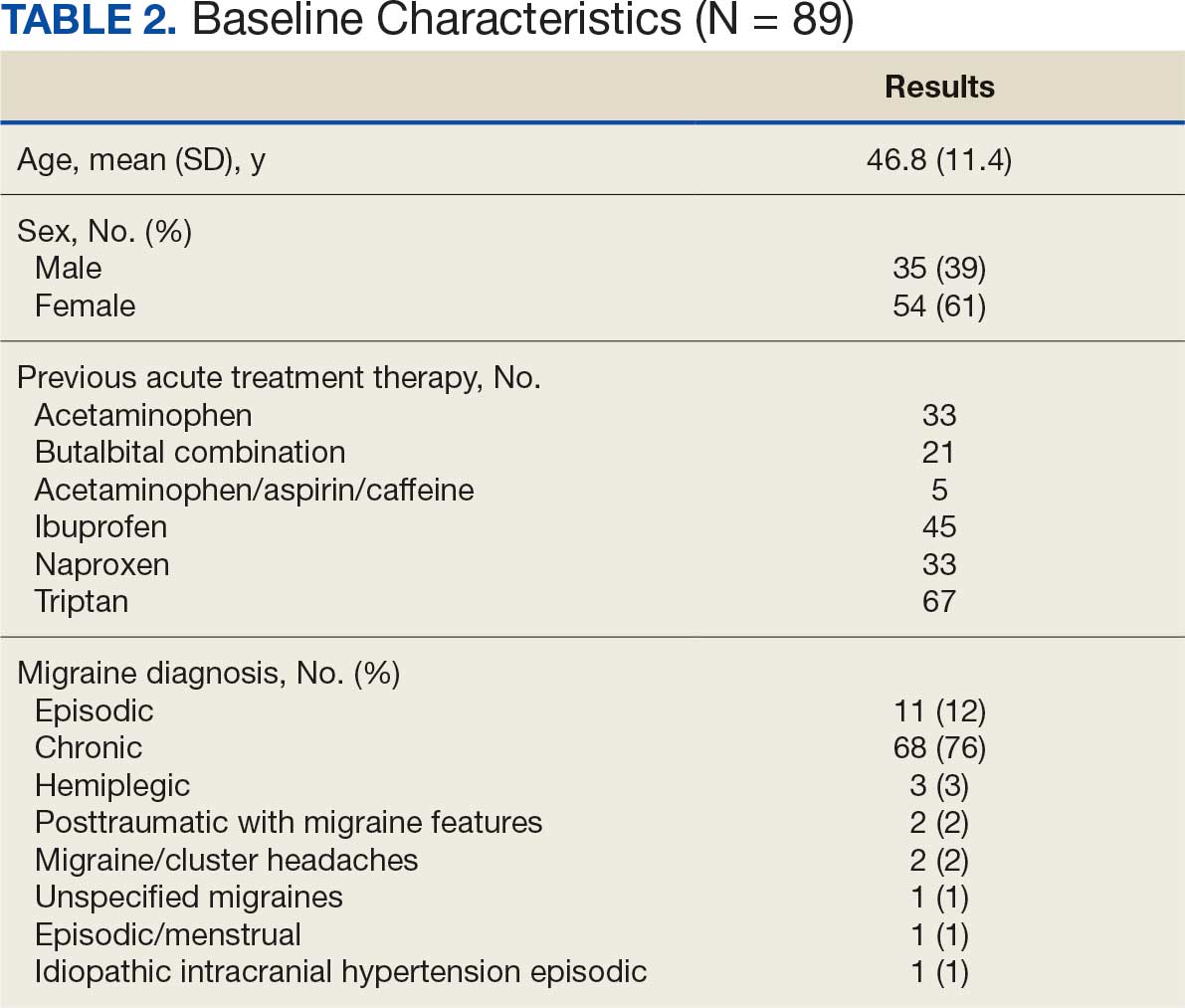
From April 1, 2023, to January 31, 2025, 96 patients were identified with active prescriptions for dual CGRP-targeting therapies. Of the 96 patients, 89 were included in the final analysis; 1 patient lacked a migraine diagnosis and 6 did not have a concomitant dual CGRP-targeted regimen and were excluded. The mean age of patients was 46.8 years and 54 (61%) were female. The most common migraine diagnosis was chronic migraine in 68 patients (76%). Triptans, ibuprofen, and acetaminophen were the most commonly used acute treatment medications (Table 2).
Safety Assessment
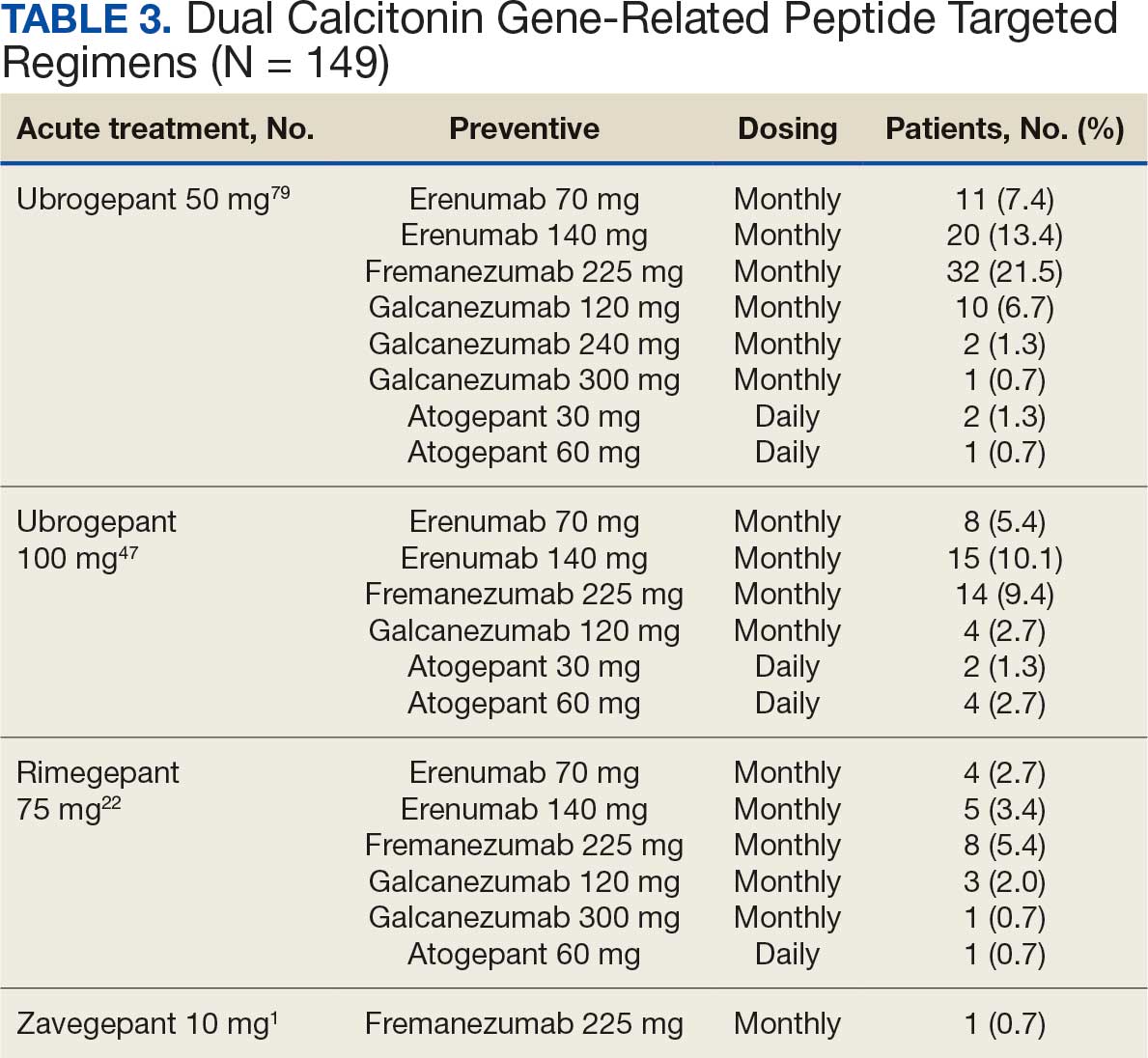
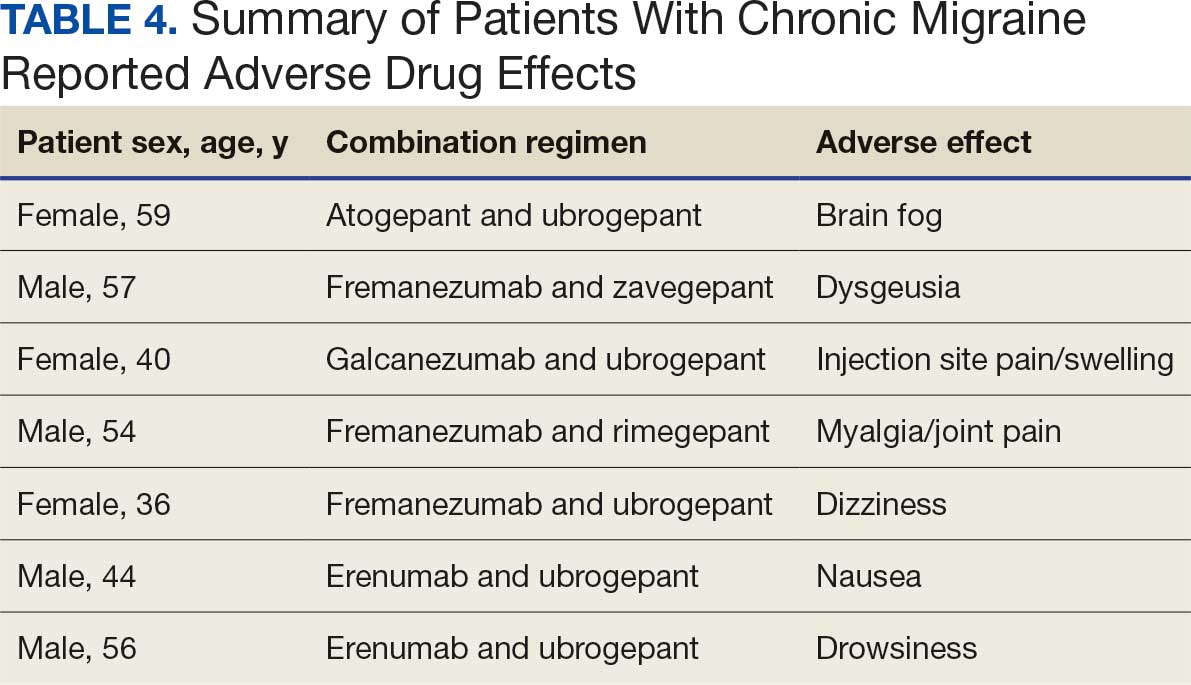
Many of the 89 unique patients trialed > 1 regimen. Thus, for the safety analysis, we analyzed 149 patients on unique dual CGRP-targeting regimens (Table 3). Ubrogepant was used by 126 patients (84.6%) for acute treatment. For preventive therapy, 63 patients (42.3%) used erenumab injections and 55 (36.9%) used fremanezumab injections. Seven patients (4.7%) reported AEs (Table 4). Five of the 7 AEs were noted in the package inserts.25-32 One patient taking both atogepant and ubrogepant reported brain fog that resolved after a dose reduction of atogepant to every other day dosing. A patient taking fremanezumab and rimegepant reported myalgia/joint pain after the first fremanezumab injection, which resolved after a few days and did not recur during the study period.
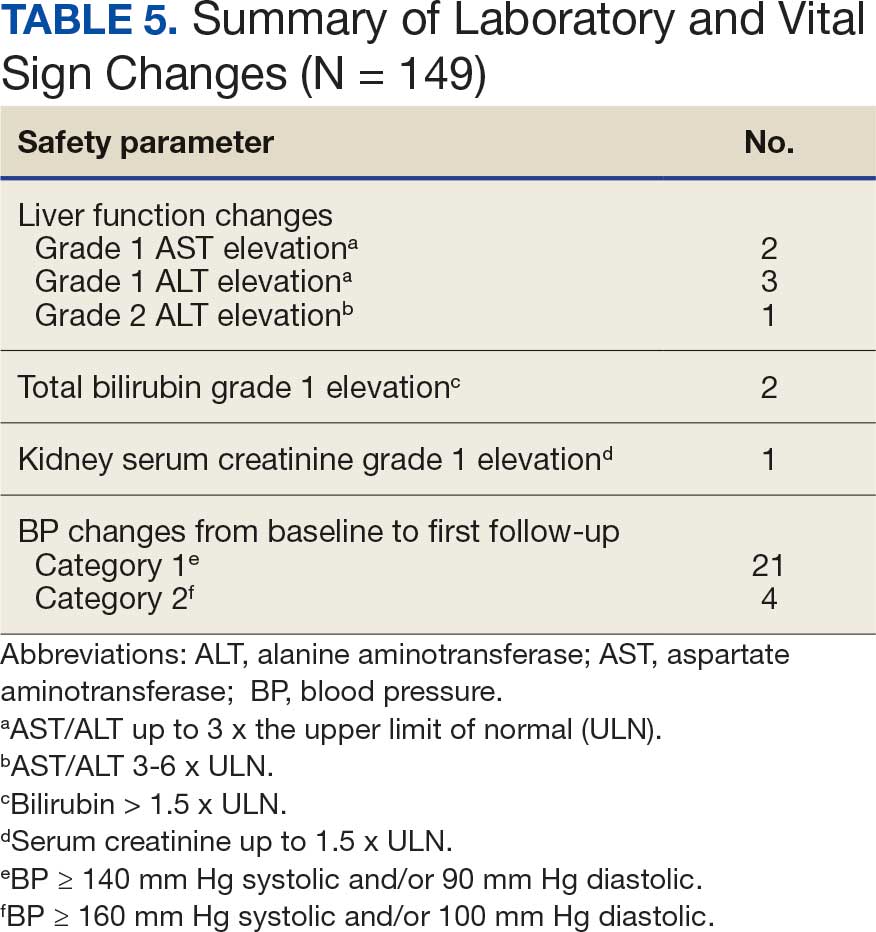
Nine of 149 patient regimens (6.0%) were associated with changes in liver function tests or serum creatinine, though all but 1 were grade 1 (1 patient had a grade 2 ALT elevation). Twenty-five patients (16.8%) experienced changes in BP, most of which were category 1 elevations. Four patients had systolic or diastolic BP ≥ 160 mm Hg or 100 mm Hg, respectively (Table 5).
Efficacy Assessment
Of the 149 unique dual CGRP regimens, 59 were eligible for the exploratory efficacy analysis. Data were excluded from the efficacy analysis if patients had not been on a stable CGRP preventive migraine regimen for ≥ 12 weeks prior to the addition of a gepant. Fourteen regimens were excluded due to a lack of clear documentation on efficacy, leaving 45 analyzed regimens. Of the 45 regimens, 34 were from unique patients. There was no median change in migraine intensity or duration found in the efficacy analysis (0.0, P = .18, and 0.0, P = .92, respectively). Ten patients on dual CGRP therapy reported that the addition of a gepant for acute treatment improved their symptoms, 20 reported that their symptoms were unchanged and/or worsened, and 29 lacked documentation.
Discussion
This study aimed to describe the safety and efficacy of concomitant CGRP regimens for migraine prevention and acute treatment. To our knowledge, this was the first descriptive study of these agents in a veteran population. The potential for increased AEs with concomitant use of CGRP antagonists is due to the similarities in the mechanism of action between the agents, which both target the same receptor/ligand pathway. Given CGRP activity in both the gastrointestinal and cardiovascular systems, the potential for related AEs is speculative. Patient-reported AEs occurred in 7 of 149 unique treatment regimens reviewed for an incidence rate < 5%. All AEs were nonserious and self-limiting.
Our findings are consistent with available research. A 2024 retrospective, exploratory real-world study evaluating the safety and tolerability of combining CGRP-targeting mAbs with gepants reported findings consistent with our results. This analysis included adult patients treated with ≥ 1 previous anti-CGRP mAb and found that 234 of 516 patients included received a combination of a gepant in addition to a CGRP-targeting mAb. Of these 234 patients, 1.3% reported nonserious AEs.33 Similarly, in a multicenter, open-label, long-term safety study in adults experiencing multiple monthly migraine attacks, a subgroup of 13 participants taking a stable dose of an anti-CGRP mAb also took rimegepant 75 mg as needed for acute treatment for 12 weeks. These patients experienced no serious AEs or any AEs leading to discontinuation.14 A study evaluating the drug-drug interaction, safety, and tolerability of dual therapy (atogepant 60 mg daily and ubrogepant 100 mg every 3 days) in 26 patients found no serious AEs, including no significant changes from baseline in laboratory results, vital signs, or safety-related 12-lead electrocardiogram parameters.15The TANDEM real-world, open-label, prospective study demonstrated similar results. It evaluated the safety and tolerability of concomitant use of ubrogepant and atogepant in patients with episodic migraines and found no increase in AEs when comparing atogepant alone with combination therapy. Twenty-six patients (9.9%) discontinued treatment due to AEs. The most common treatment-related AEs were constipation, nausea, decreased appetite, and fatigue. Efficacy data were also noted to be an exploratory endpoint in the TANDEM study; however, results have not been published.12
Within this safety analysis, new onset gastrointestinal AEs, specifically nausea, only occurred in 1 patient. Hypertension occurred in 25 treatment regimens (16.8%) for 21 unique patients (4 BP elevations occurred in 1 patient on 4 different regimens). However, the retrospective nature of reporting may limit accurate assessment. A closer analysis determined that elevated BP readings correlated with elevated pain scores at the time of the readings, which could have factored into the BP elevations. However, ongoing monitoring is needed due to an increased risk of hypertension, particularly given recent FDA labeling updates for CGRP-targeting therapies including gepants. In light of this, and the overall low incidence of hypertension reported, no new safety concerns were identified.
Limitations
Efficacy data in this project were exploratory. This evaluation did not show a significant difference in migraine intensity or duration after adding a gepant for acute treatment. The study was not powered to detect a significant difference. Limited data exist assessing efficacy outcomes with dual CGRP-targeting treatment regimens. The COURAGE study assessed the real-world effectiveness of ubrogepant and CGRP mAbs with or without the addition of onabotulinumtoxinA. The final analysis of the ubrogepant and CGRP mAb arm included 245 total patients and assessed meaningful migraine pain relief, restoration of normal function after a migraine, and treatment satisfaction. By hour 2, 61.6% of patients reported achieving migraine pain relief, rising to 80.4% by hour 4. Return to normal function occurred in 34.7% at hour 2 and 55.5% by hour 4.13 The long-term safety and efficacy of combining erenumab and rimegepant were described in a case series involving 2 patients. Both patients reported that the concomitant CGRP-targeted therapies were effective and reported no AEs.14
The retrospective design of this study meant that there was potential for limited documentation and introduction of bias into the results. Data were collected at a single VA health care system, and thus, results may not be generalizable to a broader population. However, the study population was consistent with the higher incidence of migraine expected in females in the general population. The sample size was limited, particularly in the exploratory efficacy endpoint assessment.
Limitations were observed due to inconsistent documentation regarding headache characteristics, making it challenging to draw meaningful conclusions from this data set. Additional confounding factors, including polypharmacy, nonadherence to medications, and comorbidities, may have skewed results. For example, while our study design required that the preventive CGRP-targeting medication be stable for 12 weeks for inclusion in further efficacy analysis, other medications commonly used for migraine prevention may have been adjusted (which was not accounted for in this analysis). Given this, more large-scale, placebo-controlled, randomized studies are needed to continue to assess the safety and efficacy of these combination treatment regimens.
Conclusions
Few AEs or safety events were reported with combination CGRP-targeting treatment for acute and preventive treatment of migraine. Those that were identified were considered mild. Efficacy data were limited, and further studies are needed to fully assess outcomes.
- Wattiez AS, Sowers LP, Russo AF. Calcitonin gene-related peptide (CGRP): role in migraine pathophysiology and therapeutic targeting. Expert Opin Ther Targets. 2020;24:91-100. doi:10.1080/14728222.2020.1724285
- Shah T, Bedrin K, Tinsley A. Calcitonin gene relating peptide inhibitors in combination for migraine treatment: a mini-review. Front Pain Res (Lausanne). 2023;4:1130239. doi:10.3389/fpain.2023.1130239
- Department of Veterans Affairs/Department of Defense. VA/DoD clinical practice guideline for management of headache. September 2023. Accessed February 4, 2026. https://www.healthquality.va.gov/guidelines/pain/headache/VA-DoD-CPG-Headache-Full-CPG.pdf
- Russell FA, King R, Smillie SJ, et al. Calcitonin gene-related peptide: physiology and pathophysiology. Physiol Rev. 2014;94:1099-1142. doi:10.1152/physrev.00034.2013
- de Vries Lentsch S, van der Arend BWH, VanDenBrink AM, et al. Blood pressure in patients with migraine treated with monoclonal anti-CGRP (receptor) antibodies: a prospective follow-up study. Neurology. 2022;99:e1897-e1904. doi:10.1212/WNL.0000000000201008
- Favoni V, Giani L, Al-Hassany L, et al. CGRP and migraine from a cardiovascular point of view: what do we expect from blocking CGRP?. J Headache Pain. 2019;20:27. doi:10.1186/s10194-019-0979-y
- Ailani J, Burch RC, Robbins MS, et al. The American Headache Society Consensus Statement: update on integrating new migraine treatments into clinical practice. Headache. 2021;61:1021-1039. doi:10.1111/head.14153
- Charles AC, Digre KB, Goadsby PJ, et al. Calcitonin gene-related peptide-targeting therapies are a first-line option for the prevention of migraine: an American Headache Society position statement update. Headache. 2024;64:333-341. doi:10.1111/head.14692
- Puledda F, Sacco S, Diener HC, et al. International Headache Society global practice recommendations for the acute pharmacological treatment of migraine. Cephalalgia. 2024;44:3331024241252666. doi:10.1177/03331024241252666
- Berman G, Croop R, Kudrow D, et al. Safety of rimegepant, an oral CGRP receptor antagonist, plus CGRP monoclonal antibodies for migraine. Headache. 2020;60:1734-1742. doi:10.1111/head.13930
- Blumenfeld AM, Boinpally R, De Abreu Ferreira R, et al. Phase Ib, open-label, fixed-sequence, drug-drug interaction, safety, and tolerability study between atogepant and ubrogepant in participants with a history of migraine. Headache. 2023;63:322-332. doi:10.1111/head.14433
- Ailani J, Lipton RB, Blumenfeld AM, et al. Safety and tolerability of ubrogepant for the acute treatment of migraine in participants taking atogepant for the preventive treatment of episodic migraine: results from the TANDEM study. Headache. 2025;65:1005-1014. doi:10.1111/head.14871
- Lipton RB, Contreras-De Lama J, Serrano D, et al. Real-world use of ubrogepant as acute treatment for migraine with an anti-calcitonin gene-related peptide monoclonal antibody: results from COURAGE. Neurol Ther. 2024;13:69-83. doi:10.1007/s40120-023-00556-8
- Mullin K, Kudrow D, Croop R, et al. Potential for treatment benefit of small molecule CGRP receptor antagonist plus monoclonal antibody in migraine therapy. Neurology. 2020;94:e2121-e2125. doi:10.1212/WNL.0000000000008944
- Ihara K, Takizawa T, Watanabe N, et al. Potential benefits and possible risks of CGRP-targeted multitherapy in migraine. Expert Opin Drug Metab Toxicol. 2024;20:1-4. doi:10.1080/17425255.2024.2316131
- US Department of Veterans Affairs, Pharmacy Benefits Management Services. Ubrogepant (Ubrelvy) criteria for use. July 2025. Accessed March 4, 2026. https://www.va.gov/formularyadvisor/DOC_PDF/CFU_Ubrogepant_UBRELVY_CFU_Rev_Jul_2025.pdf
- US Department of Veterans Affairs, Pharmacy Benefits Management Services. Rimegepant (Nurtec) for abortive migraine treatment criteria for use. July 2025. Accessed March 4, 2026. https://www.va.gov/formularyadvisor/DOC_PDF/CFU_Rimegepant_NURTEC_for_abortive_migraine_CFU_rev_Jul_2025.pdf
- US Department of Veterans Affairs, Pharmacy Benefits Management Services. Rimegepant (Nurtec) for episodic migraine prevention criteria for use. July 2025. Accessed March 4, 2026. https://www.va.gov/formularyadvisor/DOC_PDF/CFU_Rimegepant_NURTEC_for_episodic_migraine_prevention_CFU_rev_Jul_2025.pdf
- US Department of Veterans Affairs, Pharmacy Benefits Management Services. Erenumab-aooe (Aimovig) for chronic migraine prevention criteria for use. July 2025. Accessed March 4, 2026. https://www.va.gov/formularyadvisor/DOC_PDF/CFU_Erenumab_AIMOVIG_for_chronic_migraine_CFU_rev_Jul_2025.pdf
- US Department of Veterans Affairs, Pharmacy Benefits Management Services. Erenumab-aooe (Aimovig) for episodic migraine prevention criteria for use. July 2025. Accessed March 4, 2026. https://www.va.gov/formularyadvisor/DOC_PDF/CFU_Erenumab_AIMOVIG_for_episodic_migraine_CFU_rev_Jul_2025.pdf
- US Department of Veterans Affairs, Pharmacy Benefits Management Services. Galcanezumab-gnlm (Emgality) for cluster headache criteria for use. July 2025. Accessed March 4, 2026. https://www.va.gov/formularyadvisor/DOC_PDF/CFU_Galcanezumab_EMGALITY_for_cluster_headache_CFU_rev_Jul_2025.pdf
- US Department of Veterans Affairs, Pharmacy Benefits Management Services. Atogepant (Qulipta) for chronic migraine prevention criteria for use. July 2025. Accessed March 4, 2026. https://www.va.gov/formularyadvisor/DOC_PDF/CFU_Atogepant_QULIPTA_for_chronic_migraine_prevention_CFU_rev_Jul_2025.pdf
- Headache Classification Committee of the International Headache Society (IHS) The International Classification of Headache Disorders, 3rd edition. Cephalalgia. 2018;38:1-211. doi:10.1177/0333102417738202
- US Dept of Health and Human Services. Common Terminology Criteria for Adverse Events (CTCAE) version 5.0. November 27, 2017. Accessed March 4, 2026. https://dctd.cancer.gov/research/ctep-trials/for-sites/adverse-events/ctcae-v5-5x7.pdf
- Aimovig (erenumab-aooe) injection prescribing information. Amegen Inc. Updated March 2025. Accessed March 4, 2026. https://www.accessdata.fda.gov/drugsatfda_docs/label/2025/761077s026lbl.pdf
- Ajovy (fremanezumab-vfrm) injection prescribing information. Teva Pharmaceuticals. Updated August 2025. Accessed March 4, 2026. https://www.accessdata.fda.gov/drugsatfda_docs/label/2025/761089s031lbl.pdf
- Vyepti (eptinezumab-jjmr) injection prescribing information. Lundbeck Seattle Biopharmaceuticals. Updated October 2025. Accessed March 4, 2026. https://www.accessdata.fda.gov/drugsatfda_docs/label/2025/761119s011lbl.pdf
- Emgality (galcanezumab-gnlm) injection prescribing information. Eli Lilly and Company. Updated March 2025. Accessed March 4, 2026. https://www.accessdata.fda.gov/drugsatfda_docs/label/2025/761063s010lbl.pdf
- Qulipta (atogepant) tablets prescribing information. AbbVie Inc. Updated September 2025. Accessed March 4, 2026. https://www.accessdata.fda.gov/drugsatfda_docs/label/2025/215206s013lbl.pdf
- Nurtec ODT (rimegepant) orally disintegrating tablets prescribing information. Pfzier Labs. Updated August 2025. Accessed March 4, 2026. https://www.accessdata.fda.gov/drugsatfda_docs/label/2025/212728s028lbl.pdf
- Ubrelvy (Ubrogepant) tablets prescribing information. AbbVie Inc. Updated June 2025. Accessed March 4, 2026. https://www.accessdata.fda.gov/drugsatfda_docs/label/2025/211765s012lbl.pdf
- Zavzpret (zavegepant) intranasal spray prescribing information. Pfzier Labs. Updated August 2025. Accessed March 4, 2026. https://www.accessdata.fda.gov/drugsatfda_docs/label/2025/216386s007lbl.pdf
- Alsaadi T, Suliman R, Santos V, et al. Safety and tolerability of combining CGRP monoclonal antibodies with gepants in patients with migraine: a retrospective study. Neurol Ther. 2024;13:465-473. doi:10.1007/s40120-024-00586-w
Calcitonin gene-related peptide (CGRP) is a neuropeptide that plays a key role in migraine pathophysiology by promoting the dilation of cerebral blood vessels and transmitting pain signals.1 CGRP has generated interest for the prevention and acute treatment of migraine. Since 2018, 8 novel CGRP-targeting therapies have been approved by the US Food and Drug Administration (FDA) for the management of migraines.2,3 For migraine prevention, there are 4 injectable monoclonal antibodies (mAbs) directed against the CGRP receptor (erenumab) or the CGRP ligand (fremanezumab, galcanezumab, and eptinezumab). There are also 2 oral small-molecule CGRP receptor antagonists, termed gepants, that also are approved for migraine prevention (atogepant and rimegepant). Three gepants are approved for acute migraine treatment and are administered orally (rimegepant and ubrogepant) or intranasally (zavegepant) (Table 1).

CGRP-targeting therapies have received attention for their role in vasodilation within the cerebral, coronary, and renal vasculature.4 CGRP-mediated vasodilatory effects cause systemic regulation of blood pressure (BP) and play a protective role in hypertension.2 Some studies, particularly with erenumab, have shown that the inhibitory role of the agent leads to an increase in BP, as well as gastrointestinal issues such as constipation.2,5 The FDA recently updated monitoring recommendations for all CGRP-targeting therapies to include the potential for BP elevations and hypertension. Outside of this, there is no definitive evidence linking dual CGRP-targeted therapy to higher cardiovascular or gastrointestinal risks and prescribing information does not carry contraindications.6
In a 2021 consensus statement, the American Headache Society (AHS) recommended CGRP-targeting therapies for migraine prevention after inability to tolerate or inadequate response to an 8-week trial of ≥ 2 drug classes including antihypertensives, antiseizure medications, antidepressants, and onabotulinumtoxinA.7 For acute treatment, AHS recommended gepant use after contraindication to or inadequate response to ≥ 2 triptans. Guidance on combination CGRP-targeting therapies for both prevention and acute treatment was not provided.7 More recently, the AHS published a position statement noting substantial efficacy and safety data for CGRP-targeting therapies and suggested its consideration as a first-line option for migraine prevention, though use for acute treatment or combination CGRP-targeting therapies for both prevention and acute treatment were not addressed.8
The International Headache Society guidelines for the acute treatment of migraines recommend nonopioid analgesics as first-line therapy for mild migraine attacks. For moderate to severe attacks, triptans with or without a nonopioid analgesic were recommended as first-line therapy, prior to consideration of CGRP-targeted therapy.9 The increased use of this new drug class has also led to combination use of CGRP-targeting therapies for migraine prevention and acute treatment as seen in clinical practice and reflected by some case reports, case series, and small studies describing such use.10-14 In light of the similar mechanism of action of these therapies and the physiologic role of CGRP, there have been calls for safety evaluation.15
To our knowledge, no studies have evaluated dual CGRP-targeting regimens for migraine in the veteran population. In 2023, the US Department of Veterans Affairs (VA) and US Department of Defense (DoD) updated their clinical practice guidelines for the management of headache.3 For migraine prevention, the VA/DoD guidelines include a strong recommendation for the use of erenumab, fremanezumab, and galcanezumab; a weak recommendation for the use of atogepant; and a recommendation neither for nor against the use of rimegepant. For acute treatment, the guidelines assign a weak recommendation for the use of rimegepant and ubrogepant. Combination use was not addressed.3
Prior to the VA/DoD guidelines, the Veterans Health Administration restricted the dual use of CGRP-targeting therapies for both preventive and acute migraine treatment. However, the VA Pharmacy Benefit Management Service removed the restriction in the Criteria for Use documents, allowing broader access to these medications for veterans.16-22 This change permits the use of CGRP-targeting drugs for both acute and preventive migraine treatment after initial data reflecting real-world case reports and open-label studies suggested possible efficacy without a clear safety concern.11,12 This study aims to fill the gap in the literature by evaluating the safety, efficacy, and overall outcomes of combination CGRP-targeting treatment for migraine prevention and acute treatment in a veteran population.
Methods
This single-center, retrospective, medication use evaluation at the Ralph H. Johnson VA Medical Center (RHJVAMC) was reviewed by the RHJVAMC Research and Development Committee and Quality Improvement Program Evaluation Self Certification Tool, which both determined that institutional review board approval was not required because it was considered part of routine care and quality improvement. Computerized Patient Record System (CPRS) data were reviewed between April 1, 2023 (after the Criteria for Use for CGRP-targeting therapies was updated), through January 31, 2025. Patients were included if they had a confirmed diagnosis of migraine using the International Classification of Headache Disorders, 3rd edition criteria and had concomitant active prescriptions for both a preventive and acute treatment CGRP-targeting agent during the project period.23 Only patients receiving care from the RHJVAMC neurology department were included.
The primary objective was to assess the safety of dual CGRP-targeting therapies for migraine treatment. Key safety endpoints included effects on liver function, kidney function, and BP. Safety outcomes were graded using Common Terminology Criteria for Adverse Events.24 Changes in liver function were categorized as grade 1, 2, or 3 elevations: grade 1 (aspartate aminotransferase [AST]/alanine aminotransferase [ALT] up to 3x the upper limit of normal [ULN] or bilirubin > 1.5 x ULN); grade 2 (AST/ALT 3-6 x ULN or bilirubin 1.5-3 x ULN); and grade 3 (AST/ALT 5-10 x ULN or bilirubin 3-10 x ULN). Kidney function changes were assessed by serum creatinine levels using a similar grading system: Grade 1 (≤ 1.5 x ULN); grade 2 (1.5-3 x baseline of normal); and grade 3 (3-6 x ULN or baseline). Changes in BP were monitored from baseline to the time of the first neurology follow-up. Elevations were grouped into 2 categories, defined as BP ≥ 140 mm Hg systolic and/or 90 mm Hg diastolic (category 1) and ≥ 160 mm Hg systolic and/or 100 mm Hg diastolic (category 2). Neurology documentation was also reviewed in CPRS for individual patient-reported adverse effects (AEs). Safety endpoints were tracked for any occurrence during the project period.
The secondary objective was to describe the patient-reported efficacy of adding a gepant for acute migraine treatment to existing CGRP-targeting therapies for migraine prevention, in those patients who were stable for ≥ 12 weeks on the preventive therapy. Neurology documentation of headache characteristics, including headache severity as rated on a numerical pain score from 0 (no pain) to 10 (worst pain), and duration of headaches (in hours) were recorded during the project period. Changes in headache characteristics were tracked from baseline (ie, the neurology visit when the gepant was first requested) to the first neurology follow-up within 6 months of initiating gepant for acute treatment. If ranges were provided within documentation, a mean was calculated and used for data collection. Neurology documentation was also reviewed for any patient report of overall effectiveness with the added gepant, and categorized as symptoms improved, worsened, or did not change based on subjective report. Descriptive statistics were used for data analysis. A 1-sample Wilcoxon signed rank test was performed as an exploratory analysis for change in headache characteristics from baseline to first neurology follow-up within 6 months. Each individual CGRP regimen was counted as a unique data point to adequately describe changes associated with each new medication and/or dose adjustment. Therefore, patients could be included more than once to account for each distinct treatment regimen.
Results
From April 1, 2023, to January 31, 2025, 96 patients were identified with active prescriptions for dual CGRP-targeting therapies. Of the 96 patients, 89 were included in the final analysis; 1 patient lacked a migraine diagnosis and 6 did not have a concomitant dual CGRP-targeted regimen and were excluded. The mean age of patients was 46.8 years and 54 (61%) were female. The most common migraine diagnosis was chronic migraine in 68 patients (76%). Triptans, ibuprofen, and acetaminophen were the most commonly used acute treatment medications (Table 2).

Safety Assessment
Many of the 89 unique patients trialed > 1 regimen. Thus, for the safety analysis, we analyzed 149 patients on unique dual CGRP-targeting regimens (Table 3). Ubrogepant was used by 126 patients (84.6%) for acute treatment. For preventive therapy, 63 patients (42.3%) used erenumab injections and 55 (36.9%) used fremanezumab injections. Seven patients (4.7%) reported AEs (Table 4). Five of the 7 AEs were noted in the package inserts.25-32 One patient taking both atogepant and ubrogepant reported brain fog that resolved after a dose reduction of atogepant to every other day dosing. A patient taking fremanezumab and rimegepant reported myalgia/joint pain after the first fremanezumab injection, which resolved after a few days and did not recur during the study period.


Nine of 149 patient regimens (6.0%) were associated with changes in liver function tests or serum creatinine, though all but 1 were grade 1 (1 patient had a grade 2 ALT elevation). Twenty-five patients (16.8%) experienced changes in BP, most of which were category 1 elevations. Four patients had systolic or diastolic BP ≥ 160 mm Hg or 100 mm Hg, respectively (Table 5).

Efficacy Assessment
Of the 149 unique dual CGRP regimens, 59 were eligible for the exploratory efficacy analysis. Data were excluded from the efficacy analysis if patients had not been on a stable CGRP preventive migraine regimen for ≥ 12 weeks prior to the addition of a gepant. Fourteen regimens were excluded due to a lack of clear documentation on efficacy, leaving 45 analyzed regimens. Of the 45 regimens, 34 were from unique patients. There was no median change in migraine intensity or duration found in the efficacy analysis (0.0, P = .18, and 0.0, P = .92, respectively). Ten patients on dual CGRP therapy reported that the addition of a gepant for acute treatment improved their symptoms, 20 reported that their symptoms were unchanged and/or worsened, and 29 lacked documentation.
Discussion
This study aimed to describe the safety and efficacy of concomitant CGRP regimens for migraine prevention and acute treatment. To our knowledge, this was the first descriptive study of these agents in a veteran population. The potential for increased AEs with concomitant use of CGRP antagonists is due to the similarities in the mechanism of action between the agents, which both target the same receptor/ligand pathway. Given CGRP activity in both the gastrointestinal and cardiovascular systems, the potential for related AEs is speculative. Patient-reported AEs occurred in 7 of 149 unique treatment regimens reviewed for an incidence rate < 5%. All AEs were nonserious and self-limiting.
Our findings are consistent with available research. A 2024 retrospective, exploratory real-world study evaluating the safety and tolerability of combining CGRP-targeting mAbs with gepants reported findings consistent with our results. This analysis included adult patients treated with ≥ 1 previous anti-CGRP mAb and found that 234 of 516 patients included received a combination of a gepant in addition to a CGRP-targeting mAb. Of these 234 patients, 1.3% reported nonserious AEs.33 Similarly, in a multicenter, open-label, long-term safety study in adults experiencing multiple monthly migraine attacks, a subgroup of 13 participants taking a stable dose of an anti-CGRP mAb also took rimegepant 75 mg as needed for acute treatment for 12 weeks. These patients experienced no serious AEs or any AEs leading to discontinuation.14 A study evaluating the drug-drug interaction, safety, and tolerability of dual therapy (atogepant 60 mg daily and ubrogepant 100 mg every 3 days) in 26 patients found no serious AEs, including no significant changes from baseline in laboratory results, vital signs, or safety-related 12-lead electrocardiogram parameters.15The TANDEM real-world, open-label, prospective study demonstrated similar results. It evaluated the safety and tolerability of concomitant use of ubrogepant and atogepant in patients with episodic migraines and found no increase in AEs when comparing atogepant alone with combination therapy. Twenty-six patients (9.9%) discontinued treatment due to AEs. The most common treatment-related AEs were constipation, nausea, decreased appetite, and fatigue. Efficacy data were also noted to be an exploratory endpoint in the TANDEM study; however, results have not been published.12
Within this safety analysis, new onset gastrointestinal AEs, specifically nausea, only occurred in 1 patient. Hypertension occurred in 25 treatment regimens (16.8%) for 21 unique patients (4 BP elevations occurred in 1 patient on 4 different regimens). However, the retrospective nature of reporting may limit accurate assessment. A closer analysis determined that elevated BP readings correlated with elevated pain scores at the time of the readings, which could have factored into the BP elevations. However, ongoing monitoring is needed due to an increased risk of hypertension, particularly given recent FDA labeling updates for CGRP-targeting therapies including gepants. In light of this, and the overall low incidence of hypertension reported, no new safety concerns were identified.
Limitations
Efficacy data in this project were exploratory. This evaluation did not show a significant difference in migraine intensity or duration after adding a gepant for acute treatment. The study was not powered to detect a significant difference. Limited data exist assessing efficacy outcomes with dual CGRP-targeting treatment regimens. The COURAGE study assessed the real-world effectiveness of ubrogepant and CGRP mAbs with or without the addition of onabotulinumtoxinA. The final analysis of the ubrogepant and CGRP mAb arm included 245 total patients and assessed meaningful migraine pain relief, restoration of normal function after a migraine, and treatment satisfaction. By hour 2, 61.6% of patients reported achieving migraine pain relief, rising to 80.4% by hour 4. Return to normal function occurred in 34.7% at hour 2 and 55.5% by hour 4.13 The long-term safety and efficacy of combining erenumab and rimegepant were described in a case series involving 2 patients. Both patients reported that the concomitant CGRP-targeted therapies were effective and reported no AEs.14
The retrospective design of this study meant that there was potential for limited documentation and introduction of bias into the results. Data were collected at a single VA health care system, and thus, results may not be generalizable to a broader population. However, the study population was consistent with the higher incidence of migraine expected in females in the general population. The sample size was limited, particularly in the exploratory efficacy endpoint assessment.
Limitations were observed due to inconsistent documentation regarding headache characteristics, making it challenging to draw meaningful conclusions from this data set. Additional confounding factors, including polypharmacy, nonadherence to medications, and comorbidities, may have skewed results. For example, while our study design required that the preventive CGRP-targeting medication be stable for 12 weeks for inclusion in further efficacy analysis, other medications commonly used for migraine prevention may have been adjusted (which was not accounted for in this analysis). Given this, more large-scale, placebo-controlled, randomized studies are needed to continue to assess the safety and efficacy of these combination treatment regimens.
Conclusions
Few AEs or safety events were reported with combination CGRP-targeting treatment for acute and preventive treatment of migraine. Those that were identified were considered mild. Efficacy data were limited, and further studies are needed to fully assess outcomes.
Calcitonin gene-related peptide (CGRP) is a neuropeptide that plays a key role in migraine pathophysiology by promoting the dilation of cerebral blood vessels and transmitting pain signals.1 CGRP has generated interest for the prevention and acute treatment of migraine. Since 2018, 8 novel CGRP-targeting therapies have been approved by the US Food and Drug Administration (FDA) for the management of migraines.2,3 For migraine prevention, there are 4 injectable monoclonal antibodies (mAbs) directed against the CGRP receptor (erenumab) or the CGRP ligand (fremanezumab, galcanezumab, and eptinezumab). There are also 2 oral small-molecule CGRP receptor antagonists, termed gepants, that also are approved for migraine prevention (atogepant and rimegepant). Three gepants are approved for acute migraine treatment and are administered orally (rimegepant and ubrogepant) or intranasally (zavegepant) (Table 1).

CGRP-targeting therapies have received attention for their role in vasodilation within the cerebral, coronary, and renal vasculature.4 CGRP-mediated vasodilatory effects cause systemic regulation of blood pressure (BP) and play a protective role in hypertension.2 Some studies, particularly with erenumab, have shown that the inhibitory role of the agent leads to an increase in BP, as well as gastrointestinal issues such as constipation.2,5 The FDA recently updated monitoring recommendations for all CGRP-targeting therapies to include the potential for BP elevations and hypertension. Outside of this, there is no definitive evidence linking dual CGRP-targeted therapy to higher cardiovascular or gastrointestinal risks and prescribing information does not carry contraindications.6
In a 2021 consensus statement, the American Headache Society (AHS) recommended CGRP-targeting therapies for migraine prevention after inability to tolerate or inadequate response to an 8-week trial of ≥ 2 drug classes including antihypertensives, antiseizure medications, antidepressants, and onabotulinumtoxinA.7 For acute treatment, AHS recommended gepant use after contraindication to or inadequate response to ≥ 2 triptans. Guidance on combination CGRP-targeting therapies for both prevention and acute treatment was not provided.7 More recently, the AHS published a position statement noting substantial efficacy and safety data for CGRP-targeting therapies and suggested its consideration as a first-line option for migraine prevention, though use for acute treatment or combination CGRP-targeting therapies for both prevention and acute treatment were not addressed.8
The International Headache Society guidelines for the acute treatment of migraines recommend nonopioid analgesics as first-line therapy for mild migraine attacks. For moderate to severe attacks, triptans with or without a nonopioid analgesic were recommended as first-line therapy, prior to consideration of CGRP-targeted therapy.9 The increased use of this new drug class has also led to combination use of CGRP-targeting therapies for migraine prevention and acute treatment as seen in clinical practice and reflected by some case reports, case series, and small studies describing such use.10-14 In light of the similar mechanism of action of these therapies and the physiologic role of CGRP, there have been calls for safety evaluation.15
To our knowledge, no studies have evaluated dual CGRP-targeting regimens for migraine in the veteran population. In 2023, the US Department of Veterans Affairs (VA) and US Department of Defense (DoD) updated their clinical practice guidelines for the management of headache.3 For migraine prevention, the VA/DoD guidelines include a strong recommendation for the use of erenumab, fremanezumab, and galcanezumab; a weak recommendation for the use of atogepant; and a recommendation neither for nor against the use of rimegepant. For acute treatment, the guidelines assign a weak recommendation for the use of rimegepant and ubrogepant. Combination use was not addressed.3
Prior to the VA/DoD guidelines, the Veterans Health Administration restricted the dual use of CGRP-targeting therapies for both preventive and acute migraine treatment. However, the VA Pharmacy Benefit Management Service removed the restriction in the Criteria for Use documents, allowing broader access to these medications for veterans.16-22 This change permits the use of CGRP-targeting drugs for both acute and preventive migraine treatment after initial data reflecting real-world case reports and open-label studies suggested possible efficacy without a clear safety concern.11,12 This study aims to fill the gap in the literature by evaluating the safety, efficacy, and overall outcomes of combination CGRP-targeting treatment for migraine prevention and acute treatment in a veteran population.
Methods
This single-center, retrospective, medication use evaluation at the Ralph H. Johnson VA Medical Center (RHJVAMC) was reviewed by the RHJVAMC Research and Development Committee and Quality Improvement Program Evaluation Self Certification Tool, which both determined that institutional review board approval was not required because it was considered part of routine care and quality improvement. Computerized Patient Record System (CPRS) data were reviewed between April 1, 2023 (after the Criteria for Use for CGRP-targeting therapies was updated), through January 31, 2025. Patients were included if they had a confirmed diagnosis of migraine using the International Classification of Headache Disorders, 3rd edition criteria and had concomitant active prescriptions for both a preventive and acute treatment CGRP-targeting agent during the project period.23 Only patients receiving care from the RHJVAMC neurology department were included.
The primary objective was to assess the safety of dual CGRP-targeting therapies for migraine treatment. Key safety endpoints included effects on liver function, kidney function, and BP. Safety outcomes were graded using Common Terminology Criteria for Adverse Events.24 Changes in liver function were categorized as grade 1, 2, or 3 elevations: grade 1 (aspartate aminotransferase [AST]/alanine aminotransferase [ALT] up to 3x the upper limit of normal [ULN] or bilirubin > 1.5 x ULN); grade 2 (AST/ALT 3-6 x ULN or bilirubin 1.5-3 x ULN); and grade 3 (AST/ALT 5-10 x ULN or bilirubin 3-10 x ULN). Kidney function changes were assessed by serum creatinine levels using a similar grading system: Grade 1 (≤ 1.5 x ULN); grade 2 (1.5-3 x baseline of normal); and grade 3 (3-6 x ULN or baseline). Changes in BP were monitored from baseline to the time of the first neurology follow-up. Elevations were grouped into 2 categories, defined as BP ≥ 140 mm Hg systolic and/or 90 mm Hg diastolic (category 1) and ≥ 160 mm Hg systolic and/or 100 mm Hg diastolic (category 2). Neurology documentation was also reviewed in CPRS for individual patient-reported adverse effects (AEs). Safety endpoints were tracked for any occurrence during the project period.
The secondary objective was to describe the patient-reported efficacy of adding a gepant for acute migraine treatment to existing CGRP-targeting therapies for migraine prevention, in those patients who were stable for ≥ 12 weeks on the preventive therapy. Neurology documentation of headache characteristics, including headache severity as rated on a numerical pain score from 0 (no pain) to 10 (worst pain), and duration of headaches (in hours) were recorded during the project period. Changes in headache characteristics were tracked from baseline (ie, the neurology visit when the gepant was first requested) to the first neurology follow-up within 6 months of initiating gepant for acute treatment. If ranges were provided within documentation, a mean was calculated and used for data collection. Neurology documentation was also reviewed for any patient report of overall effectiveness with the added gepant, and categorized as symptoms improved, worsened, or did not change based on subjective report. Descriptive statistics were used for data analysis. A 1-sample Wilcoxon signed rank test was performed as an exploratory analysis for change in headache characteristics from baseline to first neurology follow-up within 6 months. Each individual CGRP regimen was counted as a unique data point to adequately describe changes associated with each new medication and/or dose adjustment. Therefore, patients could be included more than once to account for each distinct treatment regimen.
Results
From April 1, 2023, to January 31, 2025, 96 patients were identified with active prescriptions for dual CGRP-targeting therapies. Of the 96 patients, 89 were included in the final analysis; 1 patient lacked a migraine diagnosis and 6 did not have a concomitant dual CGRP-targeted regimen and were excluded. The mean age of patients was 46.8 years and 54 (61%) were female. The most common migraine diagnosis was chronic migraine in 68 patients (76%). Triptans, ibuprofen, and acetaminophen were the most commonly used acute treatment medications (Table 2).

Safety Assessment
Many of the 89 unique patients trialed > 1 regimen. Thus, for the safety analysis, we analyzed 149 patients on unique dual CGRP-targeting regimens (Table 3). Ubrogepant was used by 126 patients (84.6%) for acute treatment. For preventive therapy, 63 patients (42.3%) used erenumab injections and 55 (36.9%) used fremanezumab injections. Seven patients (4.7%) reported AEs (Table 4). Five of the 7 AEs were noted in the package inserts.25-32 One patient taking both atogepant and ubrogepant reported brain fog that resolved after a dose reduction of atogepant to every other day dosing. A patient taking fremanezumab and rimegepant reported myalgia/joint pain after the first fremanezumab injection, which resolved after a few days and did not recur during the study period.


Nine of 149 patient regimens (6.0%) were associated with changes in liver function tests or serum creatinine, though all but 1 were grade 1 (1 patient had a grade 2 ALT elevation). Twenty-five patients (16.8%) experienced changes in BP, most of which were category 1 elevations. Four patients had systolic or diastolic BP ≥ 160 mm Hg or 100 mm Hg, respectively (Table 5).

Efficacy Assessment
Of the 149 unique dual CGRP regimens, 59 were eligible for the exploratory efficacy analysis. Data were excluded from the efficacy analysis if patients had not been on a stable CGRP preventive migraine regimen for ≥ 12 weeks prior to the addition of a gepant. Fourteen regimens were excluded due to a lack of clear documentation on efficacy, leaving 45 analyzed regimens. Of the 45 regimens, 34 were from unique patients. There was no median change in migraine intensity or duration found in the efficacy analysis (0.0, P = .18, and 0.0, P = .92, respectively). Ten patients on dual CGRP therapy reported that the addition of a gepant for acute treatment improved their symptoms, 20 reported that their symptoms were unchanged and/or worsened, and 29 lacked documentation.
Discussion
This study aimed to describe the safety and efficacy of concomitant CGRP regimens for migraine prevention and acute treatment. To our knowledge, this was the first descriptive study of these agents in a veteran population. The potential for increased AEs with concomitant use of CGRP antagonists is due to the similarities in the mechanism of action between the agents, which both target the same receptor/ligand pathway. Given CGRP activity in both the gastrointestinal and cardiovascular systems, the potential for related AEs is speculative. Patient-reported AEs occurred in 7 of 149 unique treatment regimens reviewed for an incidence rate < 5%. All AEs were nonserious and self-limiting.
Our findings are consistent with available research. A 2024 retrospective, exploratory real-world study evaluating the safety and tolerability of combining CGRP-targeting mAbs with gepants reported findings consistent with our results. This analysis included adult patients treated with ≥ 1 previous anti-CGRP mAb and found that 234 of 516 patients included received a combination of a gepant in addition to a CGRP-targeting mAb. Of these 234 patients, 1.3% reported nonserious AEs.33 Similarly, in a multicenter, open-label, long-term safety study in adults experiencing multiple monthly migraine attacks, a subgroup of 13 participants taking a stable dose of an anti-CGRP mAb also took rimegepant 75 mg as needed for acute treatment for 12 weeks. These patients experienced no serious AEs or any AEs leading to discontinuation.14 A study evaluating the drug-drug interaction, safety, and tolerability of dual therapy (atogepant 60 mg daily and ubrogepant 100 mg every 3 days) in 26 patients found no serious AEs, including no significant changes from baseline in laboratory results, vital signs, or safety-related 12-lead electrocardiogram parameters.15The TANDEM real-world, open-label, prospective study demonstrated similar results. It evaluated the safety and tolerability of concomitant use of ubrogepant and atogepant in patients with episodic migraines and found no increase in AEs when comparing atogepant alone with combination therapy. Twenty-six patients (9.9%) discontinued treatment due to AEs. The most common treatment-related AEs were constipation, nausea, decreased appetite, and fatigue. Efficacy data were also noted to be an exploratory endpoint in the TANDEM study; however, results have not been published.12
Within this safety analysis, new onset gastrointestinal AEs, specifically nausea, only occurred in 1 patient. Hypertension occurred in 25 treatment regimens (16.8%) for 21 unique patients (4 BP elevations occurred in 1 patient on 4 different regimens). However, the retrospective nature of reporting may limit accurate assessment. A closer analysis determined that elevated BP readings correlated with elevated pain scores at the time of the readings, which could have factored into the BP elevations. However, ongoing monitoring is needed due to an increased risk of hypertension, particularly given recent FDA labeling updates for CGRP-targeting therapies including gepants. In light of this, and the overall low incidence of hypertension reported, no new safety concerns were identified.
Limitations
Efficacy data in this project were exploratory. This evaluation did not show a significant difference in migraine intensity or duration after adding a gepant for acute treatment. The study was not powered to detect a significant difference. Limited data exist assessing efficacy outcomes with dual CGRP-targeting treatment regimens. The COURAGE study assessed the real-world effectiveness of ubrogepant and CGRP mAbs with or without the addition of onabotulinumtoxinA. The final analysis of the ubrogepant and CGRP mAb arm included 245 total patients and assessed meaningful migraine pain relief, restoration of normal function after a migraine, and treatment satisfaction. By hour 2, 61.6% of patients reported achieving migraine pain relief, rising to 80.4% by hour 4. Return to normal function occurred in 34.7% at hour 2 and 55.5% by hour 4.13 The long-term safety and efficacy of combining erenumab and rimegepant were described in a case series involving 2 patients. Both patients reported that the concomitant CGRP-targeted therapies were effective and reported no AEs.14
The retrospective design of this study meant that there was potential for limited documentation and introduction of bias into the results. Data were collected at a single VA health care system, and thus, results may not be generalizable to a broader population. However, the study population was consistent with the higher incidence of migraine expected in females in the general population. The sample size was limited, particularly in the exploratory efficacy endpoint assessment.
Limitations were observed due to inconsistent documentation regarding headache characteristics, making it challenging to draw meaningful conclusions from this data set. Additional confounding factors, including polypharmacy, nonadherence to medications, and comorbidities, may have skewed results. For example, while our study design required that the preventive CGRP-targeting medication be stable for 12 weeks for inclusion in further efficacy analysis, other medications commonly used for migraine prevention may have been adjusted (which was not accounted for in this analysis). Given this, more large-scale, placebo-controlled, randomized studies are needed to continue to assess the safety and efficacy of these combination treatment regimens.
Conclusions
Few AEs or safety events were reported with combination CGRP-targeting treatment for acute and preventive treatment of migraine. Those that were identified were considered mild. Efficacy data were limited, and further studies are needed to fully assess outcomes.
- Wattiez AS, Sowers LP, Russo AF. Calcitonin gene-related peptide (CGRP): role in migraine pathophysiology and therapeutic targeting. Expert Opin Ther Targets. 2020;24:91-100. doi:10.1080/14728222.2020.1724285
- Shah T, Bedrin K, Tinsley A. Calcitonin gene relating peptide inhibitors in combination for migraine treatment: a mini-review. Front Pain Res (Lausanne). 2023;4:1130239. doi:10.3389/fpain.2023.1130239
- Department of Veterans Affairs/Department of Defense. VA/DoD clinical practice guideline for management of headache. September 2023. Accessed February 4, 2026. https://www.healthquality.va.gov/guidelines/pain/headache/VA-DoD-CPG-Headache-Full-CPG.pdf
- Russell FA, King R, Smillie SJ, et al. Calcitonin gene-related peptide: physiology and pathophysiology. Physiol Rev. 2014;94:1099-1142. doi:10.1152/physrev.00034.2013
- de Vries Lentsch S, van der Arend BWH, VanDenBrink AM, et al. Blood pressure in patients with migraine treated with monoclonal anti-CGRP (receptor) antibodies: a prospective follow-up study. Neurology. 2022;99:e1897-e1904. doi:10.1212/WNL.0000000000201008
- Favoni V, Giani L, Al-Hassany L, et al. CGRP and migraine from a cardiovascular point of view: what do we expect from blocking CGRP?. J Headache Pain. 2019;20:27. doi:10.1186/s10194-019-0979-y
- Ailani J, Burch RC, Robbins MS, et al. The American Headache Society Consensus Statement: update on integrating new migraine treatments into clinical practice. Headache. 2021;61:1021-1039. doi:10.1111/head.14153
- Charles AC, Digre KB, Goadsby PJ, et al. Calcitonin gene-related peptide-targeting therapies are a first-line option for the prevention of migraine: an American Headache Society position statement update. Headache. 2024;64:333-341. doi:10.1111/head.14692
- Puledda F, Sacco S, Diener HC, et al. International Headache Society global practice recommendations for the acute pharmacological treatment of migraine. Cephalalgia. 2024;44:3331024241252666. doi:10.1177/03331024241252666
- Berman G, Croop R, Kudrow D, et al. Safety of rimegepant, an oral CGRP receptor antagonist, plus CGRP monoclonal antibodies for migraine. Headache. 2020;60:1734-1742. doi:10.1111/head.13930
- Blumenfeld AM, Boinpally R, De Abreu Ferreira R, et al. Phase Ib, open-label, fixed-sequence, drug-drug interaction, safety, and tolerability study between atogepant and ubrogepant in participants with a history of migraine. Headache. 2023;63:322-332. doi:10.1111/head.14433
- Ailani J, Lipton RB, Blumenfeld AM, et al. Safety and tolerability of ubrogepant for the acute treatment of migraine in participants taking atogepant for the preventive treatment of episodic migraine: results from the TANDEM study. Headache. 2025;65:1005-1014. doi:10.1111/head.14871
- Lipton RB, Contreras-De Lama J, Serrano D, et al. Real-world use of ubrogepant as acute treatment for migraine with an anti-calcitonin gene-related peptide monoclonal antibody: results from COURAGE. Neurol Ther. 2024;13:69-83. doi:10.1007/s40120-023-00556-8
- Mullin K, Kudrow D, Croop R, et al. Potential for treatment benefit of small molecule CGRP receptor antagonist plus monoclonal antibody in migraine therapy. Neurology. 2020;94:e2121-e2125. doi:10.1212/WNL.0000000000008944
- Ihara K, Takizawa T, Watanabe N, et al. Potential benefits and possible risks of CGRP-targeted multitherapy in migraine. Expert Opin Drug Metab Toxicol. 2024;20:1-4. doi:10.1080/17425255.2024.2316131
- US Department of Veterans Affairs, Pharmacy Benefits Management Services. Ubrogepant (Ubrelvy) criteria for use. July 2025. Accessed March 4, 2026. https://www.va.gov/formularyadvisor/DOC_PDF/CFU_Ubrogepant_UBRELVY_CFU_Rev_Jul_2025.pdf
- US Department of Veterans Affairs, Pharmacy Benefits Management Services. Rimegepant (Nurtec) for abortive migraine treatment criteria for use. July 2025. Accessed March 4, 2026. https://www.va.gov/formularyadvisor/DOC_PDF/CFU_Rimegepant_NURTEC_for_abortive_migraine_CFU_rev_Jul_2025.pdf
- US Department of Veterans Affairs, Pharmacy Benefits Management Services. Rimegepant (Nurtec) for episodic migraine prevention criteria for use. July 2025. Accessed March 4, 2026. https://www.va.gov/formularyadvisor/DOC_PDF/CFU_Rimegepant_NURTEC_for_episodic_migraine_prevention_CFU_rev_Jul_2025.pdf
- US Department of Veterans Affairs, Pharmacy Benefits Management Services. Erenumab-aooe (Aimovig) for chronic migraine prevention criteria for use. July 2025. Accessed March 4, 2026. https://www.va.gov/formularyadvisor/DOC_PDF/CFU_Erenumab_AIMOVIG_for_chronic_migraine_CFU_rev_Jul_2025.pdf
- US Department of Veterans Affairs, Pharmacy Benefits Management Services. Erenumab-aooe (Aimovig) for episodic migraine prevention criteria for use. July 2025. Accessed March 4, 2026. https://www.va.gov/formularyadvisor/DOC_PDF/CFU_Erenumab_AIMOVIG_for_episodic_migraine_CFU_rev_Jul_2025.pdf
- US Department of Veterans Affairs, Pharmacy Benefits Management Services. Galcanezumab-gnlm (Emgality) for cluster headache criteria for use. July 2025. Accessed March 4, 2026. https://www.va.gov/formularyadvisor/DOC_PDF/CFU_Galcanezumab_EMGALITY_for_cluster_headache_CFU_rev_Jul_2025.pdf
- US Department of Veterans Affairs, Pharmacy Benefits Management Services. Atogepant (Qulipta) for chronic migraine prevention criteria for use. July 2025. Accessed March 4, 2026. https://www.va.gov/formularyadvisor/DOC_PDF/CFU_Atogepant_QULIPTA_for_chronic_migraine_prevention_CFU_rev_Jul_2025.pdf
- Headache Classification Committee of the International Headache Society (IHS) The International Classification of Headache Disorders, 3rd edition. Cephalalgia. 2018;38:1-211. doi:10.1177/0333102417738202
- US Dept of Health and Human Services. Common Terminology Criteria for Adverse Events (CTCAE) version 5.0. November 27, 2017. Accessed March 4, 2026. https://dctd.cancer.gov/research/ctep-trials/for-sites/adverse-events/ctcae-v5-5x7.pdf
- Aimovig (erenumab-aooe) injection prescribing information. Amegen Inc. Updated March 2025. Accessed March 4, 2026. https://www.accessdata.fda.gov/drugsatfda_docs/label/2025/761077s026lbl.pdf
- Ajovy (fremanezumab-vfrm) injection prescribing information. Teva Pharmaceuticals. Updated August 2025. Accessed March 4, 2026. https://www.accessdata.fda.gov/drugsatfda_docs/label/2025/761089s031lbl.pdf
- Vyepti (eptinezumab-jjmr) injection prescribing information. Lundbeck Seattle Biopharmaceuticals. Updated October 2025. Accessed March 4, 2026. https://www.accessdata.fda.gov/drugsatfda_docs/label/2025/761119s011lbl.pdf
- Emgality (galcanezumab-gnlm) injection prescribing information. Eli Lilly and Company. Updated March 2025. Accessed March 4, 2026. https://www.accessdata.fda.gov/drugsatfda_docs/label/2025/761063s010lbl.pdf
- Qulipta (atogepant) tablets prescribing information. AbbVie Inc. Updated September 2025. Accessed March 4, 2026. https://www.accessdata.fda.gov/drugsatfda_docs/label/2025/215206s013lbl.pdf
- Nurtec ODT (rimegepant) orally disintegrating tablets prescribing information. Pfzier Labs. Updated August 2025. Accessed March 4, 2026. https://www.accessdata.fda.gov/drugsatfda_docs/label/2025/212728s028lbl.pdf
- Ubrelvy (Ubrogepant) tablets prescribing information. AbbVie Inc. Updated June 2025. Accessed March 4, 2026. https://www.accessdata.fda.gov/drugsatfda_docs/label/2025/211765s012lbl.pdf
- Zavzpret (zavegepant) intranasal spray prescribing information. Pfzier Labs. Updated August 2025. Accessed March 4, 2026. https://www.accessdata.fda.gov/drugsatfda_docs/label/2025/216386s007lbl.pdf
- Alsaadi T, Suliman R, Santos V, et al. Safety and tolerability of combining CGRP monoclonal antibodies with gepants in patients with migraine: a retrospective study. Neurol Ther. 2024;13:465-473. doi:10.1007/s40120-024-00586-w
- Wattiez AS, Sowers LP, Russo AF. Calcitonin gene-related peptide (CGRP): role in migraine pathophysiology and therapeutic targeting. Expert Opin Ther Targets. 2020;24:91-100. doi:10.1080/14728222.2020.1724285
- Shah T, Bedrin K, Tinsley A. Calcitonin gene relating peptide inhibitors in combination for migraine treatment: a mini-review. Front Pain Res (Lausanne). 2023;4:1130239. doi:10.3389/fpain.2023.1130239
- Department of Veterans Affairs/Department of Defense. VA/DoD clinical practice guideline for management of headache. September 2023. Accessed February 4, 2026. https://www.healthquality.va.gov/guidelines/pain/headache/VA-DoD-CPG-Headache-Full-CPG.pdf
- Russell FA, King R, Smillie SJ, et al. Calcitonin gene-related peptide: physiology and pathophysiology. Physiol Rev. 2014;94:1099-1142. doi:10.1152/physrev.00034.2013
- de Vries Lentsch S, van der Arend BWH, VanDenBrink AM, et al. Blood pressure in patients with migraine treated with monoclonal anti-CGRP (receptor) antibodies: a prospective follow-up study. Neurology. 2022;99:e1897-e1904. doi:10.1212/WNL.0000000000201008
- Favoni V, Giani L, Al-Hassany L, et al. CGRP and migraine from a cardiovascular point of view: what do we expect from blocking CGRP?. J Headache Pain. 2019;20:27. doi:10.1186/s10194-019-0979-y
- Ailani J, Burch RC, Robbins MS, et al. The American Headache Society Consensus Statement: update on integrating new migraine treatments into clinical practice. Headache. 2021;61:1021-1039. doi:10.1111/head.14153
- Charles AC, Digre KB, Goadsby PJ, et al. Calcitonin gene-related peptide-targeting therapies are a first-line option for the prevention of migraine: an American Headache Society position statement update. Headache. 2024;64:333-341. doi:10.1111/head.14692
- Puledda F, Sacco S, Diener HC, et al. International Headache Society global practice recommendations for the acute pharmacological treatment of migraine. Cephalalgia. 2024;44:3331024241252666. doi:10.1177/03331024241252666
- Berman G, Croop R, Kudrow D, et al. Safety of rimegepant, an oral CGRP receptor antagonist, plus CGRP monoclonal antibodies for migraine. Headache. 2020;60:1734-1742. doi:10.1111/head.13930
- Blumenfeld AM, Boinpally R, De Abreu Ferreira R, et al. Phase Ib, open-label, fixed-sequence, drug-drug interaction, safety, and tolerability study between atogepant and ubrogepant in participants with a history of migraine. Headache. 2023;63:322-332. doi:10.1111/head.14433
- Ailani J, Lipton RB, Blumenfeld AM, et al. Safety and tolerability of ubrogepant for the acute treatment of migraine in participants taking atogepant for the preventive treatment of episodic migraine: results from the TANDEM study. Headache. 2025;65:1005-1014. doi:10.1111/head.14871
- Lipton RB, Contreras-De Lama J, Serrano D, et al. Real-world use of ubrogepant as acute treatment for migraine with an anti-calcitonin gene-related peptide monoclonal antibody: results from COURAGE. Neurol Ther. 2024;13:69-83. doi:10.1007/s40120-023-00556-8
- Mullin K, Kudrow D, Croop R, et al. Potential for treatment benefit of small molecule CGRP receptor antagonist plus monoclonal antibody in migraine therapy. Neurology. 2020;94:e2121-e2125. doi:10.1212/WNL.0000000000008944
- Ihara K, Takizawa T, Watanabe N, et al. Potential benefits and possible risks of CGRP-targeted multitherapy in migraine. Expert Opin Drug Metab Toxicol. 2024;20:1-4. doi:10.1080/17425255.2024.2316131
- US Department of Veterans Affairs, Pharmacy Benefits Management Services. Ubrogepant (Ubrelvy) criteria for use. July 2025. Accessed March 4, 2026. https://www.va.gov/formularyadvisor/DOC_PDF/CFU_Ubrogepant_UBRELVY_CFU_Rev_Jul_2025.pdf
- US Department of Veterans Affairs, Pharmacy Benefits Management Services. Rimegepant (Nurtec) for abortive migraine treatment criteria for use. July 2025. Accessed March 4, 2026. https://www.va.gov/formularyadvisor/DOC_PDF/CFU_Rimegepant_NURTEC_for_abortive_migraine_CFU_rev_Jul_2025.pdf
- US Department of Veterans Affairs, Pharmacy Benefits Management Services. Rimegepant (Nurtec) for episodic migraine prevention criteria for use. July 2025. Accessed March 4, 2026. https://www.va.gov/formularyadvisor/DOC_PDF/CFU_Rimegepant_NURTEC_for_episodic_migraine_prevention_CFU_rev_Jul_2025.pdf
- US Department of Veterans Affairs, Pharmacy Benefits Management Services. Erenumab-aooe (Aimovig) for chronic migraine prevention criteria for use. July 2025. Accessed March 4, 2026. https://www.va.gov/formularyadvisor/DOC_PDF/CFU_Erenumab_AIMOVIG_for_chronic_migraine_CFU_rev_Jul_2025.pdf
- US Department of Veterans Affairs, Pharmacy Benefits Management Services. Erenumab-aooe (Aimovig) for episodic migraine prevention criteria for use. July 2025. Accessed March 4, 2026. https://www.va.gov/formularyadvisor/DOC_PDF/CFU_Erenumab_AIMOVIG_for_episodic_migraine_CFU_rev_Jul_2025.pdf
- US Department of Veterans Affairs, Pharmacy Benefits Management Services. Galcanezumab-gnlm (Emgality) for cluster headache criteria for use. July 2025. Accessed March 4, 2026. https://www.va.gov/formularyadvisor/DOC_PDF/CFU_Galcanezumab_EMGALITY_for_cluster_headache_CFU_rev_Jul_2025.pdf
- US Department of Veterans Affairs, Pharmacy Benefits Management Services. Atogepant (Qulipta) for chronic migraine prevention criteria for use. July 2025. Accessed March 4, 2026. https://www.va.gov/formularyadvisor/DOC_PDF/CFU_Atogepant_QULIPTA_for_chronic_migraine_prevention_CFU_rev_Jul_2025.pdf
- Headache Classification Committee of the International Headache Society (IHS) The International Classification of Headache Disorders, 3rd edition. Cephalalgia. 2018;38:1-211. doi:10.1177/0333102417738202
- US Dept of Health and Human Services. Common Terminology Criteria for Adverse Events (CTCAE) version 5.0. November 27, 2017. Accessed March 4, 2026. https://dctd.cancer.gov/research/ctep-trials/for-sites/adverse-events/ctcae-v5-5x7.pdf
- Aimovig (erenumab-aooe) injection prescribing information. Amegen Inc. Updated March 2025. Accessed March 4, 2026. https://www.accessdata.fda.gov/drugsatfda_docs/label/2025/761077s026lbl.pdf
- Ajovy (fremanezumab-vfrm) injection prescribing information. Teva Pharmaceuticals. Updated August 2025. Accessed March 4, 2026. https://www.accessdata.fda.gov/drugsatfda_docs/label/2025/761089s031lbl.pdf
- Vyepti (eptinezumab-jjmr) injection prescribing information. Lundbeck Seattle Biopharmaceuticals. Updated October 2025. Accessed March 4, 2026. https://www.accessdata.fda.gov/drugsatfda_docs/label/2025/761119s011lbl.pdf
- Emgality (galcanezumab-gnlm) injection prescribing information. Eli Lilly and Company. Updated March 2025. Accessed March 4, 2026. https://www.accessdata.fda.gov/drugsatfda_docs/label/2025/761063s010lbl.pdf
- Qulipta (atogepant) tablets prescribing information. AbbVie Inc. Updated September 2025. Accessed March 4, 2026. https://www.accessdata.fda.gov/drugsatfda_docs/label/2025/215206s013lbl.pdf
- Nurtec ODT (rimegepant) orally disintegrating tablets prescribing information. Pfzier Labs. Updated August 2025. Accessed March 4, 2026. https://www.accessdata.fda.gov/drugsatfda_docs/label/2025/212728s028lbl.pdf
- Ubrelvy (Ubrogepant) tablets prescribing information. AbbVie Inc. Updated June 2025. Accessed March 4, 2026. https://www.accessdata.fda.gov/drugsatfda_docs/label/2025/211765s012lbl.pdf
- Zavzpret (zavegepant) intranasal spray prescribing information. Pfzier Labs. Updated August 2025. Accessed March 4, 2026. https://www.accessdata.fda.gov/drugsatfda_docs/label/2025/216386s007lbl.pdf
- Alsaadi T, Suliman R, Santos V, et al. Safety and tolerability of combining CGRP monoclonal antibodies with gepants in patients with migraine: a retrospective study. Neurol Ther. 2024;13:465-473. doi:10.1007/s40120-024-00586-w
Retrospective Review of Dual CGRP-Targeted Regimens for Acute and Preventive Treatment of Migraines in a Veteran Population
Retrospective Review of Dual CGRP-Targeted Regimens for Acute and Preventive Treatment of Migraines in a Veteran Population
Total Brain Diagnostics: Advancing Precision Brain and Mental Health at the Department of Veterans Affairs
Total Brain Diagnostics: Advancing Precision Brain and Mental Health at the Department of Veterans Affairs
In leveraging existing, readily available evidence-based health care information (eg, systematic reviews, clinical practice guidelines), clinicians have historically made recommendations based on treatment responses of the average patient.1 Recently, this approach has been expanded into data-driven, evidence-based precision medical care for individuals across a wide range of disciplines and care settings. These precision medicine approaches use information related to an individual’s genes, environment, and lifestyle to tailor recommendations regarding prevention, diagnosis, and treatment.
Applying precision medicine approaches to the unique exposures and experiences of service members and veterans—particularly those who served in combat environments—through the incorporation of biopsychosocial factors into medical decision-making may be even more pertinent. This sentiment is reflected in Section 305 of the Commander John Scott Hannon Veterans Mental Health Care Improvement Act of 2019, which outlines the Precision Medicine Initiative of the US Department of Veterans Affairs (VA) to identify and validate brain and mental health biomarkers.2 Despite widespread consensus regarding the promise of precision medicine, large, rich datasets with elements pertaining to common military exposures such as traumatic brain injury (TBI) and posttraumatic stress disorder (PTSD) are limited.
Existing datasets, most of which are relatively small or focus on specific cohorts (eg, older veterans, transitioning veterans), continue to create barriers to advancing precision medicine. For example, in classically designed clinical trials, analyses are generally conducted in a manner that may obfuscate efficacy among subcohorts of individuals, thereby underscoring the need to explore alternative strategies to unify existing datasets capable of revealing such heterogeneity.3 The evidence base for precision medical care is limited, drawing from published trials with relatively small sample sizes and even larger cohort studies have limited biomarker data. Additionally, these models are often exploratory during development, and to avoid statistical overfitting of an exploratory model, validation in similar datasets is needed—an added burden when data sources are small or underpowered to begin with.
A promising approach is to combine and harmonize the largest, most deeply characterized data sources from similar samples. Although combining such datasets may appear to require minimal time and effort, harmonizing similar variables in an evidence-based and replicable manner requires time and expertise, even when participant characteristics and outcomes are similar.4-7
Challenges related to harmonization are related to the wide range of strategies (eg, self-report questionnaires, clinical interviews, electronic health record review) used to measure common brain and mental health constructs, such as depression. Even when similar methods (eg, self-report measures) are implemented, challenges persist. For example, if a study used a depression measure that focused primarily on cognitive symptoms (eg, pessimism, self-dislike, suicidal ideation) and another study used a depression measure composed of items more heavily weighted towards somatic symptoms (eg, insomnia, loss of appetite, weight loss, decreased libido), combining their data could be challenging, particularly if researchers, clinicians, or administrators are interested in more than dichotomous outcomes (eg, depression vs no depression).8,9
To address this knowledge gap and harmonize multimodal data from varied sources, well-planned and reproducible curation is needed. Longitudinal cohort studies of service members and veterans with military combat and training exposure histories provide researchers and other stakeholders access to extant biopsychosocial data shown to affect risk for adverse health outcomes; however, efforts to facilitate individually tailored treatment or other precision medicine approaches would benefit from the synthesis of such datasets.10
Members of the VA Total Brain Diagnostics (TBD) team are engaged in harmonizing variables from the Long-Term Impact of Military-Relevant Brain Injury Consortium–Chronic Effects of Neurotrauma Consortium (LIMBIC-CENC)11 and the Translational Research Center for TBI and Stress Disorders (TRACTS).12-21 While there is overlap across LIMBIC-CENC and TRACTS with respect to data domains, considerable data harmonization is needed to allow for future valid and meaningful analyses, particularly those involving multivariable predictors.
Data Sources
Both data sources for the TBD harmonization project, LIMBIC-CENC and TRACTS, include extensive, longitudinal data collected from relatively large cohorts of veterans and service members with combat exposure. Both studies collect detailed data related to potential brain injury history and include participants with and without a history of TBI. Similarly, both include extensive collection of fluid biomarkers and imaging data, as well as measures of biopsychosocial functioning.
Data collection sites for LIMBIC-CENC include 16 recruitment sites, 9 at VA medical centers (Richmond, Houston, Tampa, San Antonio, Portland, Minneapolis, Boston, Salisbury, San Diego) and 7 at military treatment sites (Alexandria, San Diego, Tampa, Tacoma, Columbia, Coronado, Hinesville), in addition to 11 assessment sites (Richmond, Houston, Tampa, San Antonio, Portland, Minneapolis, Boston, Salisbury, San Diego, Alexandria, Augusta). Data for TRACTS are collected at sites in Boston and Houston.
LIMBIC-CENC is a 12-year, 17-site cohort of service members and veteran participants with combat exposure who are well characterized at baseline and undergo annual reassessments. As of December 2025, > 3100 participants have been recruited, and nearly 90% remain in follow-up. Data collection includes > 6200 annual follow-up evaluations and > 1550 5-year re-evaluations, with 400 enrolled participants followed up annually.
TRACTS is a 16-year, 2-site cohort of veterans with combat exposure who complete comprehensive assessments at enrollment, undergo annual reassessments, and complete comprehensive reassessment every 5 years thereafter. As of December 2025, > 1075 participants have completed baseline (Time 1) assessments, > 600 have completed the 2-year re-evaluation (Time 2), > 175 have completed the 5-year re-evaluation (Time 3), and > 35 have completed 10-year evaluations (Time 4), with about 50 new participants added and 100 enrolled participants followed up annually. More data on participant characteristics are available for both LIMBIC-CENC and TRACTS in previous publications.11,22These 2 ongoing, prospective, longitudinal cohorts of service members and veterans offer access to a wide range of potential risk factors that can affect response to care and outcomes, including demographics (eg, age, sex), injury characteristics (eg, pre-exposure factors, exposure factors), biomarkers (eg, serum, saliva, brain imaging, evoked potentials), and functional measures (eg, computerized posturography, computerized eye tracking, sensory testing, clinical examination, neuropsychological assessments, symptom questionnaires).
Harmonization Strategy
Pooling and harmonizing data from large studies evaluating similar participant cohorts and conditions involves numerous steps to appropriately handle a variety of measurements and disparate variable names. The TBD team adapted a model data harmonization system developed by O’Neil et al through initial work harmonizing the Federal Interagency Traumatic Brain Injury Research Informatics System (FITBIR).4-7 This process was expanded and generalized by the research team to combine data from LIMBIC-CENC and TRACTS to create a single pooled dataset for analysis (Figure).
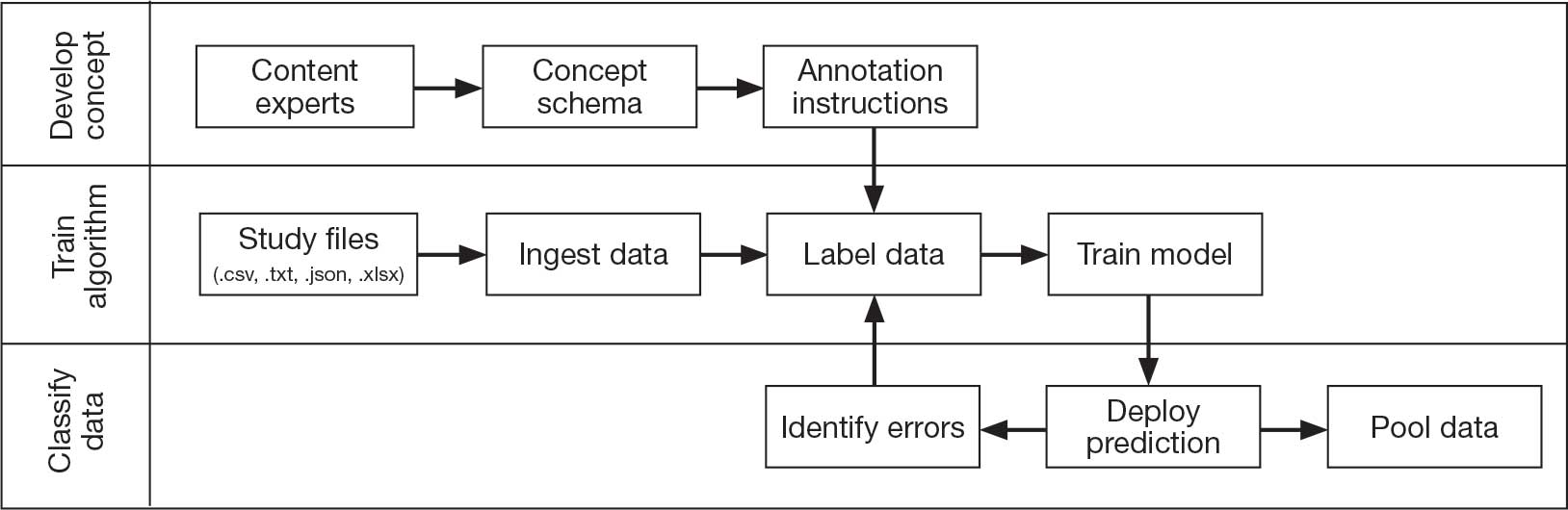
Injury Research database.
This approach was selected because it accommodates heterogeneous study designs (eg, cross-sectional, longitudinal, case-control), data collection methods (eg, clinical assessment, self-reported, objective blood, and imaging biomarkers), and various assessments of the same construct (ie, different measures of brain injury). While exact matches for data collection methods and measures may be easily harmonized, the timing of assessment, number of assessments, assessment tool version, and other factors must be considered. The goal was to harmonize data from LIMBIC-CENC and TRACTS to allow additional data sources to be harmonized and incorporated in the future.
Original data files from each study were reshaped to represent participant-level observations with 1 unique measurement per row. The measurement represents what information was collected and the value recorded represents the unique observation. These data are linked to metadata from the original study, which includes the study’s definition of each measurement, how it was collected, and any available information regarding when it was collected in reference to study enrollment or injury. Additional information on the file source, row, and column position of each data point was added to enable recreation of the original data as needed.
The resulting dataset was used to harmonize measurements from LIMBIC-CENC and TRACTS into a priori-defined schemas for brain- and mental health-relevant concepts, including TBI severity, PTSD, substance use, depression, suicidal ideation, and functioning (including cognitive, physical, and social functioning). This process was facilitated using natural language processing (NLP). Each study uniquely defines all measurements and provides written definitions with the data. Measurement definitions serve as records describing what was collected, how it was collected, and how the study may have uniquely defined information for its purposes. For example, definitions of exposure to brain injury and severity of brain injury may differ between studies, and the study-provided definition defines these differences.
Definitions were converted into numeric vectors through sentence embedding, a process that preserves the semantic meaning of the definition.23 Cosine similarity was used as the primary metric to compare the semantic textual similarity between pairs of measurement definitions. Cosine similarity ranges from 0 to 1, where 0 indicates no meaningful similarity and 1 indicates they have identical meanings.24 This approach leverages the relationship between the definitions of each measurement provided by a study and enables quick comparison of all pairwise combinations of measurement definitions between studies.
Subsets of similar measurements across studies were organized into a priori-defined schema. Clinical experts then reviewed each schema and further refined them into domains, (eg, mechanism of injury, clinical signs, acute symptoms) and subdomains (children), such as loss of consciousness, amnesia, and alteration of consciousness. This approach allows efficient handling of 2 specific cases that commonly occur when pooling and harmonizing datasets: (1) identifying the same measurement with differing names; and (2) identifying different measurements with definitions that each relate to the same domain.
The Table provides a general example of the schema for TBI severity. This was an iterative process in which clinical experts reviewed study-defined measurement definitions to develop general harmonized domains, and NLP techniques facilitated and accelerated identification and organization of measurements within these domains.
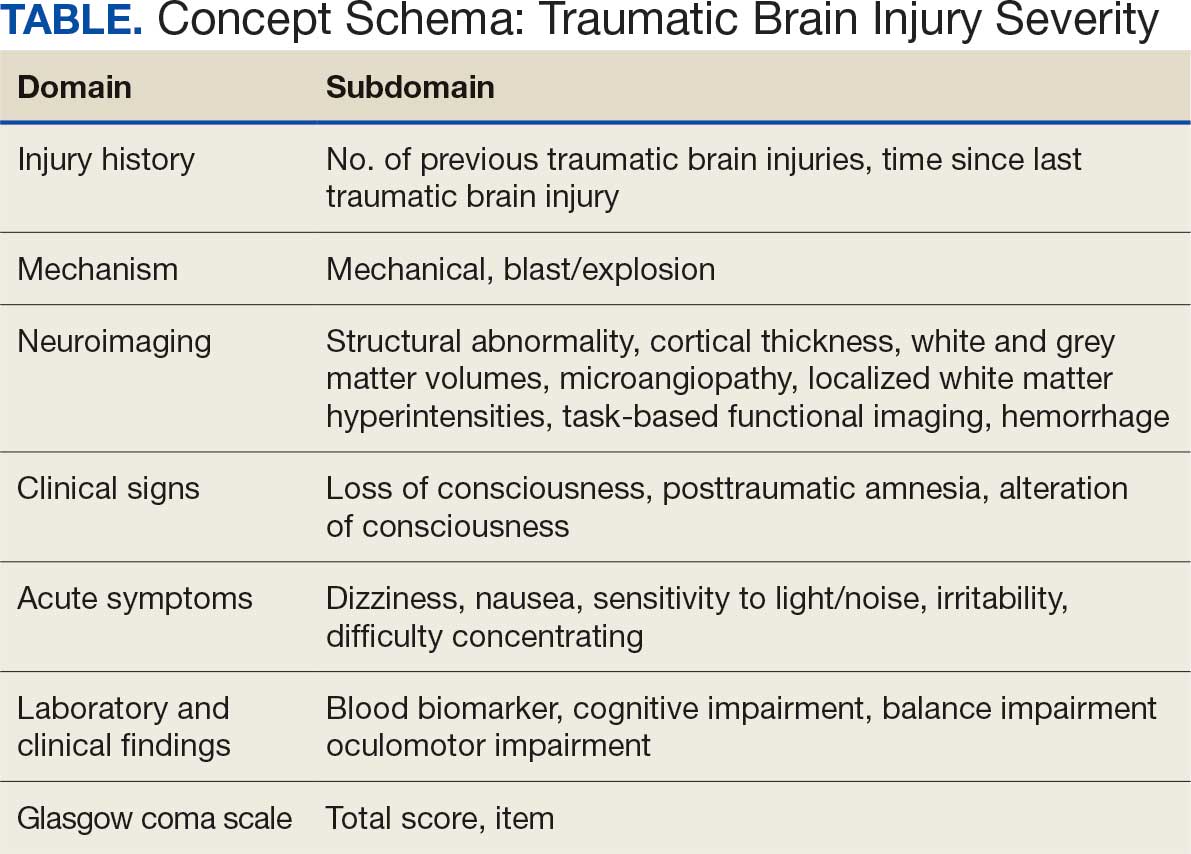
Expected Impact
Harmonization combining LIMBIC-CENC and TRACTS datasets is ongoing. Preliminary descriptive analyses of baseline cohort data indicate that harmonization across data sources is appropriate, given the lack of significant heterogeneity across sites and studies for most domains. Work by members of the TBD team is expected to lay the foundation for the use of existing and ongoing prospective, longitudinal datasets (eg, LIMBIC-CENC, TRACTS) and linked large datasets (eg, VA Informatics and Computing Infrastructure including electronic health records, VA Million Veteran Program, DaVINCI [US Department of Defense and VA Infrastructure for Clinical Intelligence]) to generate generalizable, clinically relevant information to advance precision brain and mental health care among service members and veterans.
By enhancing existing practice, this synthesized dataset has the potential to inform tailored and personalized medicine approaches designed to meet the needs of veterans and service members. These data will serve as the starting point for multivariable models examining the intersection of physiologic, behavioral, and environmental factors. The goal of this data harmonization effort is to better elucidate how clinicians and researchers can select optimal approaches for veterans and service members with TBI histories by accounting for a comprehensive set of physiologic, behavioral, and environmental factors in an individually tailored manner. These data may further extend existing clinical practice guideline approaches, inform shared decision-making, and enhance functional outcomes beyond those currently available.
Conclusions
Individuals who have served in the military have unique biopsychosocial exposures that are associated with brain and mental health disorders. To address these needs, the nationwide TBD team has initiated the creation of a unified, longitudinal dataset that includes harmonized measures from existing LIMBIC-CENC and TRACTS protocols. Initial data harmonization efforts are required to facilitate precision prognostics, diagnostics, and tailored interventions, with the goal of improving veterans’ brain and mental health and psychosocial functioning and enabling tailored and evidence-informed, individualized clinical care.
- The Promise of Precision Medicine. National Institutes of Health (NIH). Updated January 21, 2025. Accessed January 5, 2026. https://www.nih.gov/about-nih/nih-turning-discovery-into-health/promise-precision-medicine.
- Commander John Scott Hannon Veterans Mental Health Care Improvement Act of 2019, S 785, 116th Cong (2019-2020) Accessed January 5, 2026. https://www.congress.gov/bill/116th-congress/senate-bill/785
- Cheng C, Messerschmidt L, Bravo I, et al. A general primer for data harmonization. Sci Data. 2024;11:152. doi:10.1038/s41597-024-02956-3
- Neil M, Cameron D, Clauss K, et al. A proof-of-concept study demonstrating how FITBIR datasets can be harmonized to examine posttraumatic stress disorder-traumatic brain injury associations. J Behav Data Sci. 2024;4:45-62. doi:10.35566/jbds/oneil
- O’Neil ME, Cameron D, Krushnic D, et al. Using harmonized FITBIR datasets to examine associations between TBI history and cognitive functioning. Appl Neuropsychol Adult. doi:10.1080/23279095.2024.2401974
- O’Neil ME, Krushnic D, Clauss K, et al. Harmonizing federal interagency traumatic brain injury research data to examine depression and suicide-related outcomes. Rehabil Psychol. 2024;69:159-170. doi:10.1037/rep0000547
- O’Neil ME, Krushnic D, Walker WC, et al. Increased risk for clinically significant sleep disturbances in mild traumatic brain injury: an approach to leveraging the federal interagency traumatic brain injury research database. Brain Sci. 2024;14:921. doi:10.3390/brainsci14090921
- Uher R, Perlis RH, Placentino A, et al. Self-report and clinician-rated measures of depression severity: can one replace the other? Depress Anxiety. 2012;29:1043-1049. doi:10.1002/da.21993
- Hung CI, Weng LJ, Su YJ, et al. Depression and somatic symptoms scale: a new scale with both depression and somatic symptoms emphasized. Psychiatry Clin Neurosci. 2006;60:700-708. doi:10.1111/j.1440-1819.2006.01585.x
- Stewart IJ, Howard JT, Amuan ME, et al. Traumatic brain injury is associated with the subsequent risk of atrial fibrillation or atrial flutter. Heart Rhythm. 2025;22:661-667. doi:10.1016/j.hrthm.2024.09.019
- Cifu DX. Clinical research findings from the long-term impact of military-relevant brain injury consortium-chronic effects of neurotrauma consortium (LIMBIC-CENC) 2013-2021. Brain Inj. 2022;36:587-597.doi:10.1080/02699052.2022.2033843
- Fonda JR, Fredman L, Brogly SB, et al. Traumatic brain injury and attempted suicide among veterans of the wars in Iraq and Afghanistan. Am J Epidemiol. 2017;186:220-226. doi:10.1093/aje/kwx044
- Fortier CB, Amick MM, Kenna A, et al. Correspondence of the Boston Assessment of Traumatic Brain Injury-Lifetime (BAT-L) clinical interview and the VA TBI screen. J Head Trauma Rehabil. 2015;30:E1-7. doi:10.1097/htr.0000000000000008
- Grande LJ, Robinson ME, Radigan LJ, et al. Verbal memory deficits in OEF/OIF/OND veterans exposed to blasts at close range. J Int Neuropsychol Soc. 2018;24:466-475. doi:10.1017/S1355617717001242
- Hayes JP, Logue MW, Sadeh N, et al. Mild traumatic brain injury is associated with reduced cortical thickness in those at risk for Alzheimer’s disease. Brain. 2017;140:813-825. doi:10.1093/brain/aww344
- Lippa SM, Fonda JR, Fortier CB, et al. Deployment-related psychiatric and behavioral conditions and their association with functional disability in OEF/OIF/OND veterans. J Trauma Stress. 2015;28:25-33. doi:10.1002/jts.21979
- McGlinchey RE, Milberg WP, Fonda JR, et al. A methodology for assessing deployment trauma and its consequences in OEF/OIF/OND veterans: the TRACTS longitudinal prospective cohort study. Int J Methods Psychiatr Res. 2017;26:e1556. doi:10.1002/mpr.1556
- Radigan LJ, McGlinchey RE, Milberg WP, et al. Correspondence of the Boston Assessment of Traumatic Brain Injury-Lifetime and the VA Comprehensive TBI Evaluation. J Head Trauma Rehabil. 2018;33:E51-E55. doi:10.1097/htr.0000000000000361
- Sydnor VJ, Bouix S, Pasternak O, et al. Mild traumatic brain injury impacts associations between limbic system microstructure and post-traumatic stress disorder symptomatology. Neuroimage Clin. 2020;26:102190. doi:10.1016/j.nicl.2020.102190
- Van Etten EJ, Knight AR, Colaizzi TA, et al. Peritraumatic context and long-term outcomes of concussion. JAMA Netw Open. 2025;8:e2455622. doi:10.1001/jamanetworkopen.2024.55622
- Andrews RJ, Fonda JR, Levin LK, et al. Comprehensive analysis of the predictors of neurobehavioral symptom reporting in veterans. Neurology. 2018;91:e732-e745. doi:10.1212/wnl.0000000000006034
- McGlinchey RE, Milberg WP, Fonda JR, Fortier CB. A methodology for assessing deployment trauma and its consequences in OEF/OIF/OND veterans: the TRACTS longitudional prospective cohort study. Int J Methods Psychiatr Res. 2017;26:e1556. doi:10.1002/mpr.1556
- Reimers N, Gurevych I. Sentence-BERT: Sentence embeddings using Siamese BERT-Networks. 2019. Conference on Empirical Methods in Natural Language Processing.
- Singhal A. Modern information retrieval: a brief overview. IEEE Data Eng Bull. 2001;24:34-43.
In leveraging existing, readily available evidence-based health care information (eg, systematic reviews, clinical practice guidelines), clinicians have historically made recommendations based on treatment responses of the average patient.1 Recently, this approach has been expanded into data-driven, evidence-based precision medical care for individuals across a wide range of disciplines and care settings. These precision medicine approaches use information related to an individual’s genes, environment, and lifestyle to tailor recommendations regarding prevention, diagnosis, and treatment.
Applying precision medicine approaches to the unique exposures and experiences of service members and veterans—particularly those who served in combat environments—through the incorporation of biopsychosocial factors into medical decision-making may be even more pertinent. This sentiment is reflected in Section 305 of the Commander John Scott Hannon Veterans Mental Health Care Improvement Act of 2019, which outlines the Precision Medicine Initiative of the US Department of Veterans Affairs (VA) to identify and validate brain and mental health biomarkers.2 Despite widespread consensus regarding the promise of precision medicine, large, rich datasets with elements pertaining to common military exposures such as traumatic brain injury (TBI) and posttraumatic stress disorder (PTSD) are limited.
Existing datasets, most of which are relatively small or focus on specific cohorts (eg, older veterans, transitioning veterans), continue to create barriers to advancing precision medicine. For example, in classically designed clinical trials, analyses are generally conducted in a manner that may obfuscate efficacy among subcohorts of individuals, thereby underscoring the need to explore alternative strategies to unify existing datasets capable of revealing such heterogeneity.3 The evidence base for precision medical care is limited, drawing from published trials with relatively small sample sizes and even larger cohort studies have limited biomarker data. Additionally, these models are often exploratory during development, and to avoid statistical overfitting of an exploratory model, validation in similar datasets is needed—an added burden when data sources are small or underpowered to begin with.
A promising approach is to combine and harmonize the largest, most deeply characterized data sources from similar samples. Although combining such datasets may appear to require minimal time and effort, harmonizing similar variables in an evidence-based and replicable manner requires time and expertise, even when participant characteristics and outcomes are similar.4-7
Challenges related to harmonization are related to the wide range of strategies (eg, self-report questionnaires, clinical interviews, electronic health record review) used to measure common brain and mental health constructs, such as depression. Even when similar methods (eg, self-report measures) are implemented, challenges persist. For example, if a study used a depression measure that focused primarily on cognitive symptoms (eg, pessimism, self-dislike, suicidal ideation) and another study used a depression measure composed of items more heavily weighted towards somatic symptoms (eg, insomnia, loss of appetite, weight loss, decreased libido), combining their data could be challenging, particularly if researchers, clinicians, or administrators are interested in more than dichotomous outcomes (eg, depression vs no depression).8,9
To address this knowledge gap and harmonize multimodal data from varied sources, well-planned and reproducible curation is needed. Longitudinal cohort studies of service members and veterans with military combat and training exposure histories provide researchers and other stakeholders access to extant biopsychosocial data shown to affect risk for adverse health outcomes; however, efforts to facilitate individually tailored treatment or other precision medicine approaches would benefit from the synthesis of such datasets.10
Members of the VA Total Brain Diagnostics (TBD) team are engaged in harmonizing variables from the Long-Term Impact of Military-Relevant Brain Injury Consortium–Chronic Effects of Neurotrauma Consortium (LIMBIC-CENC)11 and the Translational Research Center for TBI and Stress Disorders (TRACTS).12-21 While there is overlap across LIMBIC-CENC and TRACTS with respect to data domains, considerable data harmonization is needed to allow for future valid and meaningful analyses, particularly those involving multivariable predictors.
Data Sources
Both data sources for the TBD harmonization project, LIMBIC-CENC and TRACTS, include extensive, longitudinal data collected from relatively large cohorts of veterans and service members with combat exposure. Both studies collect detailed data related to potential brain injury history and include participants with and without a history of TBI. Similarly, both include extensive collection of fluid biomarkers and imaging data, as well as measures of biopsychosocial functioning.
Data collection sites for LIMBIC-CENC include 16 recruitment sites, 9 at VA medical centers (Richmond, Houston, Tampa, San Antonio, Portland, Minneapolis, Boston, Salisbury, San Diego) and 7 at military treatment sites (Alexandria, San Diego, Tampa, Tacoma, Columbia, Coronado, Hinesville), in addition to 11 assessment sites (Richmond, Houston, Tampa, San Antonio, Portland, Minneapolis, Boston, Salisbury, San Diego, Alexandria, Augusta). Data for TRACTS are collected at sites in Boston and Houston.
LIMBIC-CENC is a 12-year, 17-site cohort of service members and veteran participants with combat exposure who are well characterized at baseline and undergo annual reassessments. As of December 2025, > 3100 participants have been recruited, and nearly 90% remain in follow-up. Data collection includes > 6200 annual follow-up evaluations and > 1550 5-year re-evaluations, with 400 enrolled participants followed up annually.
TRACTS is a 16-year, 2-site cohort of veterans with combat exposure who complete comprehensive assessments at enrollment, undergo annual reassessments, and complete comprehensive reassessment every 5 years thereafter. As of December 2025, > 1075 participants have completed baseline (Time 1) assessments, > 600 have completed the 2-year re-evaluation (Time 2), > 175 have completed the 5-year re-evaluation (Time 3), and > 35 have completed 10-year evaluations (Time 4), with about 50 new participants added and 100 enrolled participants followed up annually. More data on participant characteristics are available for both LIMBIC-CENC and TRACTS in previous publications.11,22These 2 ongoing, prospective, longitudinal cohorts of service members and veterans offer access to a wide range of potential risk factors that can affect response to care and outcomes, including demographics (eg, age, sex), injury characteristics (eg, pre-exposure factors, exposure factors), biomarkers (eg, serum, saliva, brain imaging, evoked potentials), and functional measures (eg, computerized posturography, computerized eye tracking, sensory testing, clinical examination, neuropsychological assessments, symptom questionnaires).
Harmonization Strategy
Pooling and harmonizing data from large studies evaluating similar participant cohorts and conditions involves numerous steps to appropriately handle a variety of measurements and disparate variable names. The TBD team adapted a model data harmonization system developed by O’Neil et al through initial work harmonizing the Federal Interagency Traumatic Brain Injury Research Informatics System (FITBIR).4-7 This process was expanded and generalized by the research team to combine data from LIMBIC-CENC and TRACTS to create a single pooled dataset for analysis (Figure).

Injury Research database.
This approach was selected because it accommodates heterogeneous study designs (eg, cross-sectional, longitudinal, case-control), data collection methods (eg, clinical assessment, self-reported, objective blood, and imaging biomarkers), and various assessments of the same construct (ie, different measures of brain injury). While exact matches for data collection methods and measures may be easily harmonized, the timing of assessment, number of assessments, assessment tool version, and other factors must be considered. The goal was to harmonize data from LIMBIC-CENC and TRACTS to allow additional data sources to be harmonized and incorporated in the future.
Original data files from each study were reshaped to represent participant-level observations with 1 unique measurement per row. The measurement represents what information was collected and the value recorded represents the unique observation. These data are linked to metadata from the original study, which includes the study’s definition of each measurement, how it was collected, and any available information regarding when it was collected in reference to study enrollment or injury. Additional information on the file source, row, and column position of each data point was added to enable recreation of the original data as needed.
The resulting dataset was used to harmonize measurements from LIMBIC-CENC and TRACTS into a priori-defined schemas for brain- and mental health-relevant concepts, including TBI severity, PTSD, substance use, depression, suicidal ideation, and functioning (including cognitive, physical, and social functioning). This process was facilitated using natural language processing (NLP). Each study uniquely defines all measurements and provides written definitions with the data. Measurement definitions serve as records describing what was collected, how it was collected, and how the study may have uniquely defined information for its purposes. For example, definitions of exposure to brain injury and severity of brain injury may differ between studies, and the study-provided definition defines these differences.
Definitions were converted into numeric vectors through sentence embedding, a process that preserves the semantic meaning of the definition.23 Cosine similarity was used as the primary metric to compare the semantic textual similarity between pairs of measurement definitions. Cosine similarity ranges from 0 to 1, where 0 indicates no meaningful similarity and 1 indicates they have identical meanings.24 This approach leverages the relationship between the definitions of each measurement provided by a study and enables quick comparison of all pairwise combinations of measurement definitions between studies.
Subsets of similar measurements across studies were organized into a priori-defined schema. Clinical experts then reviewed each schema and further refined them into domains, (eg, mechanism of injury, clinical signs, acute symptoms) and subdomains (children), such as loss of consciousness, amnesia, and alteration of consciousness. This approach allows efficient handling of 2 specific cases that commonly occur when pooling and harmonizing datasets: (1) identifying the same measurement with differing names; and (2) identifying different measurements with definitions that each relate to the same domain.
The Table provides a general example of the schema for TBI severity. This was an iterative process in which clinical experts reviewed study-defined measurement definitions to develop general harmonized domains, and NLP techniques facilitated and accelerated identification and organization of measurements within these domains.

Expected Impact
Harmonization combining LIMBIC-CENC and TRACTS datasets is ongoing. Preliminary descriptive analyses of baseline cohort data indicate that harmonization across data sources is appropriate, given the lack of significant heterogeneity across sites and studies for most domains. Work by members of the TBD team is expected to lay the foundation for the use of existing and ongoing prospective, longitudinal datasets (eg, LIMBIC-CENC, TRACTS) and linked large datasets (eg, VA Informatics and Computing Infrastructure including electronic health records, VA Million Veteran Program, DaVINCI [US Department of Defense and VA Infrastructure for Clinical Intelligence]) to generate generalizable, clinically relevant information to advance precision brain and mental health care among service members and veterans.
By enhancing existing practice, this synthesized dataset has the potential to inform tailored and personalized medicine approaches designed to meet the needs of veterans and service members. These data will serve as the starting point for multivariable models examining the intersection of physiologic, behavioral, and environmental factors. The goal of this data harmonization effort is to better elucidate how clinicians and researchers can select optimal approaches for veterans and service members with TBI histories by accounting for a comprehensive set of physiologic, behavioral, and environmental factors in an individually tailored manner. These data may further extend existing clinical practice guideline approaches, inform shared decision-making, and enhance functional outcomes beyond those currently available.
Conclusions
Individuals who have served in the military have unique biopsychosocial exposures that are associated with brain and mental health disorders. To address these needs, the nationwide TBD team has initiated the creation of a unified, longitudinal dataset that includes harmonized measures from existing LIMBIC-CENC and TRACTS protocols. Initial data harmonization efforts are required to facilitate precision prognostics, diagnostics, and tailored interventions, with the goal of improving veterans’ brain and mental health and psychosocial functioning and enabling tailored and evidence-informed, individualized clinical care.
In leveraging existing, readily available evidence-based health care information (eg, systematic reviews, clinical practice guidelines), clinicians have historically made recommendations based on treatment responses of the average patient.1 Recently, this approach has been expanded into data-driven, evidence-based precision medical care for individuals across a wide range of disciplines and care settings. These precision medicine approaches use information related to an individual’s genes, environment, and lifestyle to tailor recommendations regarding prevention, diagnosis, and treatment.
Applying precision medicine approaches to the unique exposures and experiences of service members and veterans—particularly those who served in combat environments—through the incorporation of biopsychosocial factors into medical decision-making may be even more pertinent. This sentiment is reflected in Section 305 of the Commander John Scott Hannon Veterans Mental Health Care Improvement Act of 2019, which outlines the Precision Medicine Initiative of the US Department of Veterans Affairs (VA) to identify and validate brain and mental health biomarkers.2 Despite widespread consensus regarding the promise of precision medicine, large, rich datasets with elements pertaining to common military exposures such as traumatic brain injury (TBI) and posttraumatic stress disorder (PTSD) are limited.
Existing datasets, most of which are relatively small or focus on specific cohorts (eg, older veterans, transitioning veterans), continue to create barriers to advancing precision medicine. For example, in classically designed clinical trials, analyses are generally conducted in a manner that may obfuscate efficacy among subcohorts of individuals, thereby underscoring the need to explore alternative strategies to unify existing datasets capable of revealing such heterogeneity.3 The evidence base for precision medical care is limited, drawing from published trials with relatively small sample sizes and even larger cohort studies have limited biomarker data. Additionally, these models are often exploratory during development, and to avoid statistical overfitting of an exploratory model, validation in similar datasets is needed—an added burden when data sources are small or underpowered to begin with.
A promising approach is to combine and harmonize the largest, most deeply characterized data sources from similar samples. Although combining such datasets may appear to require minimal time and effort, harmonizing similar variables in an evidence-based and replicable manner requires time and expertise, even when participant characteristics and outcomes are similar.4-7
Challenges related to harmonization are related to the wide range of strategies (eg, self-report questionnaires, clinical interviews, electronic health record review) used to measure common brain and mental health constructs, such as depression. Even when similar methods (eg, self-report measures) are implemented, challenges persist. For example, if a study used a depression measure that focused primarily on cognitive symptoms (eg, pessimism, self-dislike, suicidal ideation) and another study used a depression measure composed of items more heavily weighted towards somatic symptoms (eg, insomnia, loss of appetite, weight loss, decreased libido), combining their data could be challenging, particularly if researchers, clinicians, or administrators are interested in more than dichotomous outcomes (eg, depression vs no depression).8,9
To address this knowledge gap and harmonize multimodal data from varied sources, well-planned and reproducible curation is needed. Longitudinal cohort studies of service members and veterans with military combat and training exposure histories provide researchers and other stakeholders access to extant biopsychosocial data shown to affect risk for adverse health outcomes; however, efforts to facilitate individually tailored treatment or other precision medicine approaches would benefit from the synthesis of such datasets.10
Members of the VA Total Brain Diagnostics (TBD) team are engaged in harmonizing variables from the Long-Term Impact of Military-Relevant Brain Injury Consortium–Chronic Effects of Neurotrauma Consortium (LIMBIC-CENC)11 and the Translational Research Center for TBI and Stress Disorders (TRACTS).12-21 While there is overlap across LIMBIC-CENC and TRACTS with respect to data domains, considerable data harmonization is needed to allow for future valid and meaningful analyses, particularly those involving multivariable predictors.
Data Sources
Both data sources for the TBD harmonization project, LIMBIC-CENC and TRACTS, include extensive, longitudinal data collected from relatively large cohorts of veterans and service members with combat exposure. Both studies collect detailed data related to potential brain injury history and include participants with and without a history of TBI. Similarly, both include extensive collection of fluid biomarkers and imaging data, as well as measures of biopsychosocial functioning.
Data collection sites for LIMBIC-CENC include 16 recruitment sites, 9 at VA medical centers (Richmond, Houston, Tampa, San Antonio, Portland, Minneapolis, Boston, Salisbury, San Diego) and 7 at military treatment sites (Alexandria, San Diego, Tampa, Tacoma, Columbia, Coronado, Hinesville), in addition to 11 assessment sites (Richmond, Houston, Tampa, San Antonio, Portland, Minneapolis, Boston, Salisbury, San Diego, Alexandria, Augusta). Data for TRACTS are collected at sites in Boston and Houston.
LIMBIC-CENC is a 12-year, 17-site cohort of service members and veteran participants with combat exposure who are well characterized at baseline and undergo annual reassessments. As of December 2025, > 3100 participants have been recruited, and nearly 90% remain in follow-up. Data collection includes > 6200 annual follow-up evaluations and > 1550 5-year re-evaluations, with 400 enrolled participants followed up annually.
TRACTS is a 16-year, 2-site cohort of veterans with combat exposure who complete comprehensive assessments at enrollment, undergo annual reassessments, and complete comprehensive reassessment every 5 years thereafter. As of December 2025, > 1075 participants have completed baseline (Time 1) assessments, > 600 have completed the 2-year re-evaluation (Time 2), > 175 have completed the 5-year re-evaluation (Time 3), and > 35 have completed 10-year evaluations (Time 4), with about 50 new participants added and 100 enrolled participants followed up annually. More data on participant characteristics are available for both LIMBIC-CENC and TRACTS in previous publications.11,22These 2 ongoing, prospective, longitudinal cohorts of service members and veterans offer access to a wide range of potential risk factors that can affect response to care and outcomes, including demographics (eg, age, sex), injury characteristics (eg, pre-exposure factors, exposure factors), biomarkers (eg, serum, saliva, brain imaging, evoked potentials), and functional measures (eg, computerized posturography, computerized eye tracking, sensory testing, clinical examination, neuropsychological assessments, symptom questionnaires).
Harmonization Strategy
Pooling and harmonizing data from large studies evaluating similar participant cohorts and conditions involves numerous steps to appropriately handle a variety of measurements and disparate variable names. The TBD team adapted a model data harmonization system developed by O’Neil et al through initial work harmonizing the Federal Interagency Traumatic Brain Injury Research Informatics System (FITBIR).4-7 This process was expanded and generalized by the research team to combine data from LIMBIC-CENC and TRACTS to create a single pooled dataset for analysis (Figure).

Injury Research database.
This approach was selected because it accommodates heterogeneous study designs (eg, cross-sectional, longitudinal, case-control), data collection methods (eg, clinical assessment, self-reported, objective blood, and imaging biomarkers), and various assessments of the same construct (ie, different measures of brain injury). While exact matches for data collection methods and measures may be easily harmonized, the timing of assessment, number of assessments, assessment tool version, and other factors must be considered. The goal was to harmonize data from LIMBIC-CENC and TRACTS to allow additional data sources to be harmonized and incorporated in the future.
Original data files from each study were reshaped to represent participant-level observations with 1 unique measurement per row. The measurement represents what information was collected and the value recorded represents the unique observation. These data are linked to metadata from the original study, which includes the study’s definition of each measurement, how it was collected, and any available information regarding when it was collected in reference to study enrollment or injury. Additional information on the file source, row, and column position of each data point was added to enable recreation of the original data as needed.
The resulting dataset was used to harmonize measurements from LIMBIC-CENC and TRACTS into a priori-defined schemas for brain- and mental health-relevant concepts, including TBI severity, PTSD, substance use, depression, suicidal ideation, and functioning (including cognitive, physical, and social functioning). This process was facilitated using natural language processing (NLP). Each study uniquely defines all measurements and provides written definitions with the data. Measurement definitions serve as records describing what was collected, how it was collected, and how the study may have uniquely defined information for its purposes. For example, definitions of exposure to brain injury and severity of brain injury may differ between studies, and the study-provided definition defines these differences.
Definitions were converted into numeric vectors through sentence embedding, a process that preserves the semantic meaning of the definition.23 Cosine similarity was used as the primary metric to compare the semantic textual similarity between pairs of measurement definitions. Cosine similarity ranges from 0 to 1, where 0 indicates no meaningful similarity and 1 indicates they have identical meanings.24 This approach leverages the relationship between the definitions of each measurement provided by a study and enables quick comparison of all pairwise combinations of measurement definitions between studies.
Subsets of similar measurements across studies were organized into a priori-defined schema. Clinical experts then reviewed each schema and further refined them into domains, (eg, mechanism of injury, clinical signs, acute symptoms) and subdomains (children), such as loss of consciousness, amnesia, and alteration of consciousness. This approach allows efficient handling of 2 specific cases that commonly occur when pooling and harmonizing datasets: (1) identifying the same measurement with differing names; and (2) identifying different measurements with definitions that each relate to the same domain.
The Table provides a general example of the schema for TBI severity. This was an iterative process in which clinical experts reviewed study-defined measurement definitions to develop general harmonized domains, and NLP techniques facilitated and accelerated identification and organization of measurements within these domains.

Expected Impact
Harmonization combining LIMBIC-CENC and TRACTS datasets is ongoing. Preliminary descriptive analyses of baseline cohort data indicate that harmonization across data sources is appropriate, given the lack of significant heterogeneity across sites and studies for most domains. Work by members of the TBD team is expected to lay the foundation for the use of existing and ongoing prospective, longitudinal datasets (eg, LIMBIC-CENC, TRACTS) and linked large datasets (eg, VA Informatics and Computing Infrastructure including electronic health records, VA Million Veteran Program, DaVINCI [US Department of Defense and VA Infrastructure for Clinical Intelligence]) to generate generalizable, clinically relevant information to advance precision brain and mental health care among service members and veterans.
By enhancing existing practice, this synthesized dataset has the potential to inform tailored and personalized medicine approaches designed to meet the needs of veterans and service members. These data will serve as the starting point for multivariable models examining the intersection of physiologic, behavioral, and environmental factors. The goal of this data harmonization effort is to better elucidate how clinicians and researchers can select optimal approaches for veterans and service members with TBI histories by accounting for a comprehensive set of physiologic, behavioral, and environmental factors in an individually tailored manner. These data may further extend existing clinical practice guideline approaches, inform shared decision-making, and enhance functional outcomes beyond those currently available.
Conclusions
Individuals who have served in the military have unique biopsychosocial exposures that are associated with brain and mental health disorders. To address these needs, the nationwide TBD team has initiated the creation of a unified, longitudinal dataset that includes harmonized measures from existing LIMBIC-CENC and TRACTS protocols. Initial data harmonization efforts are required to facilitate precision prognostics, diagnostics, and tailored interventions, with the goal of improving veterans’ brain and mental health and psychosocial functioning and enabling tailored and evidence-informed, individualized clinical care.
- The Promise of Precision Medicine. National Institutes of Health (NIH). Updated January 21, 2025. Accessed January 5, 2026. https://www.nih.gov/about-nih/nih-turning-discovery-into-health/promise-precision-medicine.
- Commander John Scott Hannon Veterans Mental Health Care Improvement Act of 2019, S 785, 116th Cong (2019-2020) Accessed January 5, 2026. https://www.congress.gov/bill/116th-congress/senate-bill/785
- Cheng C, Messerschmidt L, Bravo I, et al. A general primer for data harmonization. Sci Data. 2024;11:152. doi:10.1038/s41597-024-02956-3
- Neil M, Cameron D, Clauss K, et al. A proof-of-concept study demonstrating how FITBIR datasets can be harmonized to examine posttraumatic stress disorder-traumatic brain injury associations. J Behav Data Sci. 2024;4:45-62. doi:10.35566/jbds/oneil
- O’Neil ME, Cameron D, Krushnic D, et al. Using harmonized FITBIR datasets to examine associations between TBI history and cognitive functioning. Appl Neuropsychol Adult. doi:10.1080/23279095.2024.2401974
- O’Neil ME, Krushnic D, Clauss K, et al. Harmonizing federal interagency traumatic brain injury research data to examine depression and suicide-related outcomes. Rehabil Psychol. 2024;69:159-170. doi:10.1037/rep0000547
- O’Neil ME, Krushnic D, Walker WC, et al. Increased risk for clinically significant sleep disturbances in mild traumatic brain injury: an approach to leveraging the federal interagency traumatic brain injury research database. Brain Sci. 2024;14:921. doi:10.3390/brainsci14090921
- Uher R, Perlis RH, Placentino A, et al. Self-report and clinician-rated measures of depression severity: can one replace the other? Depress Anxiety. 2012;29:1043-1049. doi:10.1002/da.21993
- Hung CI, Weng LJ, Su YJ, et al. Depression and somatic symptoms scale: a new scale with both depression and somatic symptoms emphasized. Psychiatry Clin Neurosci. 2006;60:700-708. doi:10.1111/j.1440-1819.2006.01585.x
- Stewart IJ, Howard JT, Amuan ME, et al. Traumatic brain injury is associated with the subsequent risk of atrial fibrillation or atrial flutter. Heart Rhythm. 2025;22:661-667. doi:10.1016/j.hrthm.2024.09.019
- Cifu DX. Clinical research findings from the long-term impact of military-relevant brain injury consortium-chronic effects of neurotrauma consortium (LIMBIC-CENC) 2013-2021. Brain Inj. 2022;36:587-597.doi:10.1080/02699052.2022.2033843
- Fonda JR, Fredman L, Brogly SB, et al. Traumatic brain injury and attempted suicide among veterans of the wars in Iraq and Afghanistan. Am J Epidemiol. 2017;186:220-226. doi:10.1093/aje/kwx044
- Fortier CB, Amick MM, Kenna A, et al. Correspondence of the Boston Assessment of Traumatic Brain Injury-Lifetime (BAT-L) clinical interview and the VA TBI screen. J Head Trauma Rehabil. 2015;30:E1-7. doi:10.1097/htr.0000000000000008
- Grande LJ, Robinson ME, Radigan LJ, et al. Verbal memory deficits in OEF/OIF/OND veterans exposed to blasts at close range. J Int Neuropsychol Soc. 2018;24:466-475. doi:10.1017/S1355617717001242
- Hayes JP, Logue MW, Sadeh N, et al. Mild traumatic brain injury is associated with reduced cortical thickness in those at risk for Alzheimer’s disease. Brain. 2017;140:813-825. doi:10.1093/brain/aww344
- Lippa SM, Fonda JR, Fortier CB, et al. Deployment-related psychiatric and behavioral conditions and their association with functional disability in OEF/OIF/OND veterans. J Trauma Stress. 2015;28:25-33. doi:10.1002/jts.21979
- McGlinchey RE, Milberg WP, Fonda JR, et al. A methodology for assessing deployment trauma and its consequences in OEF/OIF/OND veterans: the TRACTS longitudinal prospective cohort study. Int J Methods Psychiatr Res. 2017;26:e1556. doi:10.1002/mpr.1556
- Radigan LJ, McGlinchey RE, Milberg WP, et al. Correspondence of the Boston Assessment of Traumatic Brain Injury-Lifetime and the VA Comprehensive TBI Evaluation. J Head Trauma Rehabil. 2018;33:E51-E55. doi:10.1097/htr.0000000000000361
- Sydnor VJ, Bouix S, Pasternak O, et al. Mild traumatic brain injury impacts associations between limbic system microstructure and post-traumatic stress disorder symptomatology. Neuroimage Clin. 2020;26:102190. doi:10.1016/j.nicl.2020.102190
- Van Etten EJ, Knight AR, Colaizzi TA, et al. Peritraumatic context and long-term outcomes of concussion. JAMA Netw Open. 2025;8:e2455622. doi:10.1001/jamanetworkopen.2024.55622
- Andrews RJ, Fonda JR, Levin LK, et al. Comprehensive analysis of the predictors of neurobehavioral symptom reporting in veterans. Neurology. 2018;91:e732-e745. doi:10.1212/wnl.0000000000006034
- McGlinchey RE, Milberg WP, Fonda JR, Fortier CB. A methodology for assessing deployment trauma and its consequences in OEF/OIF/OND veterans: the TRACTS longitudional prospective cohort study. Int J Methods Psychiatr Res. 2017;26:e1556. doi:10.1002/mpr.1556
- Reimers N, Gurevych I. Sentence-BERT: Sentence embeddings using Siamese BERT-Networks. 2019. Conference on Empirical Methods in Natural Language Processing.
- Singhal A. Modern information retrieval: a brief overview. IEEE Data Eng Bull. 2001;24:34-43.
- The Promise of Precision Medicine. National Institutes of Health (NIH). Updated January 21, 2025. Accessed January 5, 2026. https://www.nih.gov/about-nih/nih-turning-discovery-into-health/promise-precision-medicine.
- Commander John Scott Hannon Veterans Mental Health Care Improvement Act of 2019, S 785, 116th Cong (2019-2020) Accessed January 5, 2026. https://www.congress.gov/bill/116th-congress/senate-bill/785
- Cheng C, Messerschmidt L, Bravo I, et al. A general primer for data harmonization. Sci Data. 2024;11:152. doi:10.1038/s41597-024-02956-3
- Neil M, Cameron D, Clauss K, et al. A proof-of-concept study demonstrating how FITBIR datasets can be harmonized to examine posttraumatic stress disorder-traumatic brain injury associations. J Behav Data Sci. 2024;4:45-62. doi:10.35566/jbds/oneil
- O’Neil ME, Cameron D, Krushnic D, et al. Using harmonized FITBIR datasets to examine associations between TBI history and cognitive functioning. Appl Neuropsychol Adult. doi:10.1080/23279095.2024.2401974
- O’Neil ME, Krushnic D, Clauss K, et al. Harmonizing federal interagency traumatic brain injury research data to examine depression and suicide-related outcomes. Rehabil Psychol. 2024;69:159-170. doi:10.1037/rep0000547
- O’Neil ME, Krushnic D, Walker WC, et al. Increased risk for clinically significant sleep disturbances in mild traumatic brain injury: an approach to leveraging the federal interagency traumatic brain injury research database. Brain Sci. 2024;14:921. doi:10.3390/brainsci14090921
- Uher R, Perlis RH, Placentino A, et al. Self-report and clinician-rated measures of depression severity: can one replace the other? Depress Anxiety. 2012;29:1043-1049. doi:10.1002/da.21993
- Hung CI, Weng LJ, Su YJ, et al. Depression and somatic symptoms scale: a new scale with both depression and somatic symptoms emphasized. Psychiatry Clin Neurosci. 2006;60:700-708. doi:10.1111/j.1440-1819.2006.01585.x
- Stewart IJ, Howard JT, Amuan ME, et al. Traumatic brain injury is associated with the subsequent risk of atrial fibrillation or atrial flutter. Heart Rhythm. 2025;22:661-667. doi:10.1016/j.hrthm.2024.09.019
- Cifu DX. Clinical research findings from the long-term impact of military-relevant brain injury consortium-chronic effects of neurotrauma consortium (LIMBIC-CENC) 2013-2021. Brain Inj. 2022;36:587-597.doi:10.1080/02699052.2022.2033843
- Fonda JR, Fredman L, Brogly SB, et al. Traumatic brain injury and attempted suicide among veterans of the wars in Iraq and Afghanistan. Am J Epidemiol. 2017;186:220-226. doi:10.1093/aje/kwx044
- Fortier CB, Amick MM, Kenna A, et al. Correspondence of the Boston Assessment of Traumatic Brain Injury-Lifetime (BAT-L) clinical interview and the VA TBI screen. J Head Trauma Rehabil. 2015;30:E1-7. doi:10.1097/htr.0000000000000008
- Grande LJ, Robinson ME, Radigan LJ, et al. Verbal memory deficits in OEF/OIF/OND veterans exposed to blasts at close range. J Int Neuropsychol Soc. 2018;24:466-475. doi:10.1017/S1355617717001242
- Hayes JP, Logue MW, Sadeh N, et al. Mild traumatic brain injury is associated with reduced cortical thickness in those at risk for Alzheimer’s disease. Brain. 2017;140:813-825. doi:10.1093/brain/aww344
- Lippa SM, Fonda JR, Fortier CB, et al. Deployment-related psychiatric and behavioral conditions and their association with functional disability in OEF/OIF/OND veterans. J Trauma Stress. 2015;28:25-33. doi:10.1002/jts.21979
- McGlinchey RE, Milberg WP, Fonda JR, et al. A methodology for assessing deployment trauma and its consequences in OEF/OIF/OND veterans: the TRACTS longitudinal prospective cohort study. Int J Methods Psychiatr Res. 2017;26:e1556. doi:10.1002/mpr.1556
- Radigan LJ, McGlinchey RE, Milberg WP, et al. Correspondence of the Boston Assessment of Traumatic Brain Injury-Lifetime and the VA Comprehensive TBI Evaluation. J Head Trauma Rehabil. 2018;33:E51-E55. doi:10.1097/htr.0000000000000361
- Sydnor VJ, Bouix S, Pasternak O, et al. Mild traumatic brain injury impacts associations between limbic system microstructure and post-traumatic stress disorder symptomatology. Neuroimage Clin. 2020;26:102190. doi:10.1016/j.nicl.2020.102190
- Van Etten EJ, Knight AR, Colaizzi TA, et al. Peritraumatic context and long-term outcomes of concussion. JAMA Netw Open. 2025;8:e2455622. doi:10.1001/jamanetworkopen.2024.55622
- Andrews RJ, Fonda JR, Levin LK, et al. Comprehensive analysis of the predictors of neurobehavioral symptom reporting in veterans. Neurology. 2018;91:e732-e745. doi:10.1212/wnl.0000000000006034
- McGlinchey RE, Milberg WP, Fonda JR, Fortier CB. A methodology for assessing deployment trauma and its consequences in OEF/OIF/OND veterans: the TRACTS longitudional prospective cohort study. Int J Methods Psychiatr Res. 2017;26:e1556. doi:10.1002/mpr.1556
- Reimers N, Gurevych I. Sentence-BERT: Sentence embeddings using Siamese BERT-Networks. 2019. Conference on Empirical Methods in Natural Language Processing.
- Singhal A. Modern information retrieval: a brief overview. IEEE Data Eng Bull. 2001;24:34-43.
Total Brain Diagnostics: Advancing Precision Brain and Mental Health at the Department of Veterans Affairs
Total Brain Diagnostics: Advancing Precision Brain and Mental Health at the Department of Veterans Affairs
Can Telehealth Improve Access to Amyloid-Targeting Therapies for Veterans Living With Alzheimer Disease?
Can Telehealth Improve Access to Amyloid-Targeting Therapies for Veterans Living With Alzheimer Disease?
The Veterans Health Administration (VHA) is the largest US integrated health care system, providing health care to > 9 million veterans annually. Dementia affects > 7.2 million Americans, and an estimated 450,000 veterans live with Alzheimer disease (AD).1,2 Compared with the general population, veterans have a higher burden of chronic medical conditions and are disproportionately affected by AD due to exposure to military-related risk factors (eg, traumatic brain injury and posttraumatic stress disorder) and the high prevalence of nonmilitary risk factors, such as cardiovascular disease. The VHA is a pioneer in dementia care, having established a Dementia System of Care to provide primary and specialty care to veterans with dementia. The VHA also is leading the way in implementing the Institute for Healthcare Improvement Age-Friendly Health Systems (AFHS) framework for providing goal-concordant care in > 100 VHA medical centers. The VHA aims to be the largest AFHS in the country.
AD profoundly affects individuals and their families. The progressive nature of the most common form of dementia diminishes the quality of life for patients as well as their care partners in an ongoing fashion, often leading to emotional, physical, and financial strain. Costs for health and long-term care for people living with AD and other dementias were projected at $360 billion in 2024, largely due to the need for nursing home care.1 Although several oral medications are available, their capacity to effectively mitigate the negative effects of AD is limited. Cholinesterase inhibitors and memantine may offer temporary symptomatic relief, but they do not alter disease progression.3 The use of these agents is relatively low, with about one-third of patients diagnosed with AD receiving these medications.4
Amyloid-Targeting Therapies
Recent advancements in biologics, particularly amyloid-targeting therapies, such as lecanemab and donanemab, offer new hope for managing AD. Older adults treated with these medications show less decline on measures of cognition and function than those receiving a placebo at 18 months.5,6 However, accessing and using these medications is challenging.
Use of amyloid-targeting therapies poses challenges. The medications are expensive, potentially placing a financial burden on patients, families, and health care systems.7 Determining initial eligibility for treatment requires a battery of cognitive assessments, laboratory tests, advanced radiologic studies (eg, magnetic resonance imaging [MRI] of the brain and amyloid positron emission tomography [PET] scans), and possible cerebrospinal fluid (CSF) testing. Frequent ongoing assessments are necessary to monitor safety and efficacy. These treatments carry substantial risks, particularly amyloid-related imaging abnormalities (ARIA) such as cerebral edema, microhemorrhages, and superficial siderosis. Therefore, follow-up assessments typically occur around months 2, 3, 4, and 7, depending on which medication is selected. Finally, at present, both agents must be intravenous (IV)-administered in a monitored clinical setting, which requires additional coordination, transportation, and cost.
Ongoing evaluations and in-person administration particularly affect patients and care partners with limitations regarding transportation, time off work, and navigating complex health care systems.8 VHA clinicians at sites that have implemented or are interested in implementing amyloid-targeting therapy programs endorse similar challenges when implementing these therapies in their US Department of Veterans Affairs (VA) medical centers (VAMCs).9
The VHA was one of the first health care systems to use amyloid-targeting therapies, covering the cost of lecanemab and donanemab, in addition to costs associated with concomitant evaluation and testing. However, given the safety concerns with this novel class of medications, the VHA National Formulary Committee developed criteria for use and recommended the VA Center for Medication Safety (VAMedSAFE) conduct a mandatory real-time medication use evaluation (MUE). VAMedSAFE developed the MUE to monitor the safe and appropriate use of amyloid-targeting therapy for AD. Two authors (AJO, SMH) partnered with VAMedSAFE through the VA Pittsburgh Healthcare System Technology Enhancing Cognition and Health–Geriatric Research, Education, and Clinical Center (TECH-GRECC) to provide clinical expertise, substantive feedback for the development of the MUE, and guidance for VHA sites starting amyloid targeting-therapy programs. We started a VHA Amyloid-Targeting Therapy for AD SharePoint collaborative platform and VHA AD Therapeutics Community of Practice (CoP) for shared learning (Figure). The private SharePoint platform houses an array of implementation materials for VAMCs starting programs: key documents and links; educational materials; sample guidelines; note templates; and electronic health record screenshots. The CoP allows VHAs to share best practices and discuss challenges.
Even with these advantages, we found that ensuring the safe and appropriate use of amyloid-targeting therapies did not overcome the barriers associated with their complexity. This was especially true for veterans living in rural areas. Only 4 VAMCs had administered amyloid-targeting therapies in the first year they were available. Preliminary data demonstrated that 27 (84%) of 32 veterans who initiated lecanemab in the VHA between October 2023 and September 2024 resided in urban areas.10 To address the underutilization of amyloid-targeting therapy, we propose leveraging the strengths of VHA telehealth to facilitate expansion of access to these medications for veterans with early AD. Telehealth may substantially increase access to evaluation for veterans with early dementia and, when medically appropriate, to receive amyloid-targeting therapies by reducing transportation needs and mitigating costs while ensuring appropriate monitoring through ongoing clinical assessments.
Using Telehealth
The VHA is a pioneer in telehealth, with programs dating back to 2003.11 Between October 1, 2018, and September 30, 2019, the VHA served > 900,000 veterans through the provision of > 2.6 million episodes of care via telehealth.12 The COVID-19 pandemic further cemented the role of telemedicine as an essential component of health care. Telehealth has demonstrated success in the assessment and management of individuals living with dementia. At the VHA, the GRECC-Connect Project is a partnership between 9 urban GRECC sites that seek to provide consultative geriatric and dementia care to rural veterans through telehealth.13 Additional evidence supports the potential to leverage telehealth to effectively communicate results of amyloid PET scans.14
This approach is not without limitations such as the digital divide, or the gap that separates technology-enabled individuals and those unprepared to adopt technology due to limited digital literacy levels or access to needed hardware, software, and connectivity. The VHA has taken steps to address these digital divide barriers by broadly providing tools—such as tablets and broadband connectivity—to veterans. Specifically, the VHA has instituted digital divide consults to determine whether telehealth could be a potential solution for appropriate veterans and to provide an iPad (if eligible) to connect with VA clinicians. Complementary to the digital divide consult, a VHA-specific telehealth preparedness assessment tool is under development and being tested by 2 authors (JF, SMH). This telehealth preparedness assessment tool is designed to aid in the seamless integration of telehealth services with the support of tailored education materials specific to gaps in digital literacy that a veteran might experience.
Building on these initiatives, there is an opportunity to expand access to amyloid-targeting therapies, regardless of distance to large VAMCs, by leveraging telehealth as an alternative method of connecting patients with specialty care. Specifically, a hybrid approach could be used to accomplish the myriad initial and follow-up tasks involved in the provision of amyloid-targeting therapies (Table). Not all VHA facilities possess the specialty expertise to prescribe these medications, and local clinicians may not have sufficient knowledge and clinical support to prescribe and monitor these therapies.
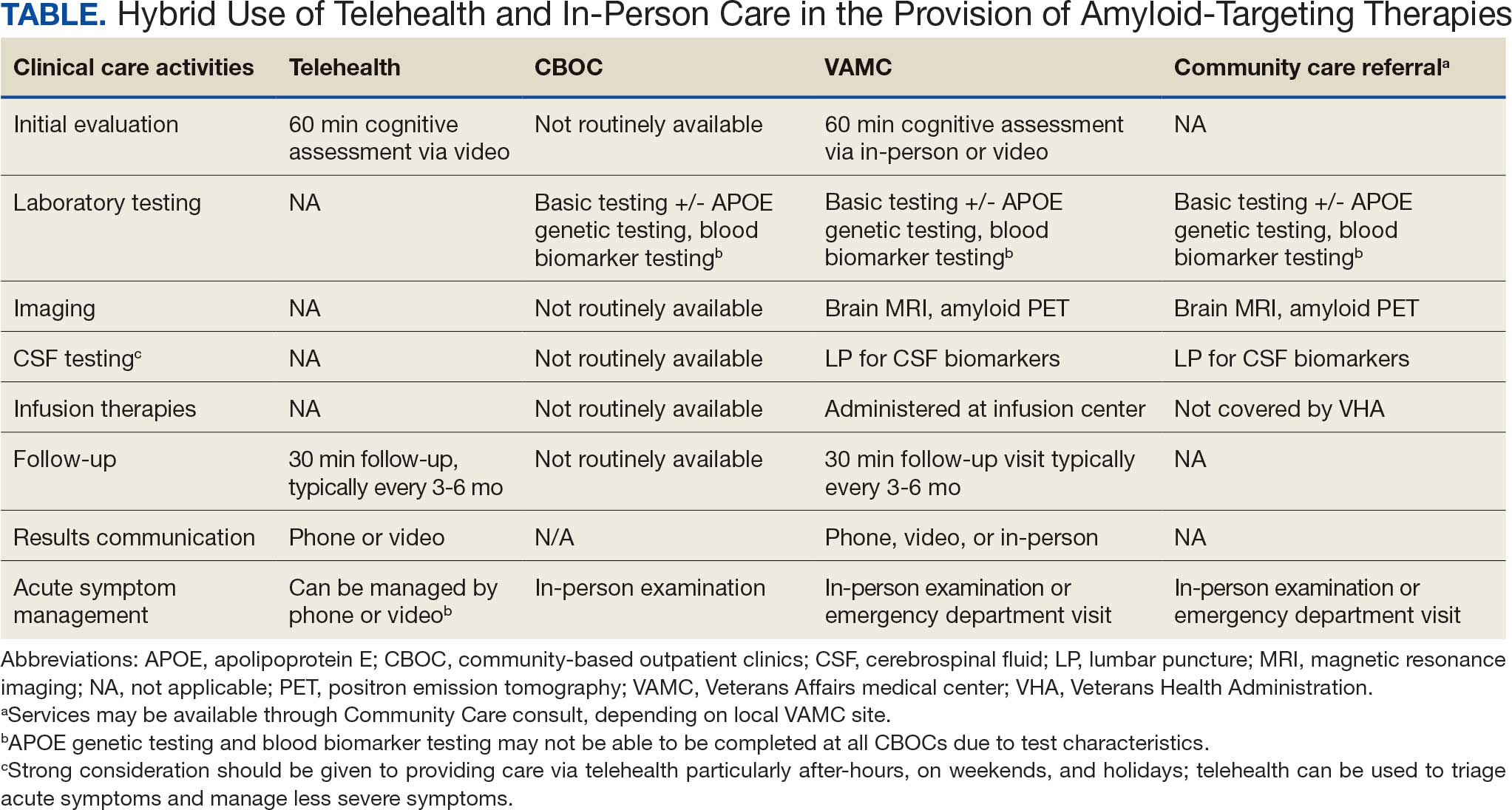
The first step is identifying local and regional subject matter experts, followed by the development and expansion of these networks. The National TeleNeurology Program is a good example of a national telehealth program that leverages technology to bring specialty services to rural areas with limited access to care. Although amyloid-targeting therapies often require more complex logistics, such as laboratory tests and imaging, these initial hurdles can be overcome through localized services and collaboration between VAMCs.
While treatment and imaging will most likely need to occur at a VAMC, most basic laboratory studies can be performed at community-based outpatient clinics (CBOCs). Some CBOCs may not be able to process more specialized laboratory tests such as apolipoprotein E genetic testing. Samples for these tests can be collected and processed at VAMCs, which usually have contracts with outside laboratories capable of performing these studies. Most, although not all, VAMCs offer advanced imaging, including MRI of the brain and amyloid PETs. VAMCs without those modalities may need to coordinate with other regional VAMCs. Additionally, a pilot program is already underway whereby VAMCs without the ability to quantify the amount of amyloid on PETs are able to leverage technology and collaborations with other VAMCs to obtain these data.
Once the initial phases of evaluation and care are completed, telemedicine can be leveraged for follow-up and ongoing management. Interdisciplinary teams can help facilitate care related to amyloid-targeting therapies, including the close monitoring of veterans for development of ARIA.15 To achieve this monitoring, specialty clinic teams prescribing amyloid-targeting therapies, which may be geographically distant, need to coordinate with local primary care clinical teams and emergency clinicians. All of these health care team members, along with neurologists and neurosurgeons, should be involved in the development and implementation of protocols in the event that patients present to their local primary or specialty care clinics or emergency department with ARIA symptoms.
If amyloid-targeting therapies are to be provided along with other emerging treatments for rural veterans, telehealth must be part of the solution. There is a pressing need to explore innovative evaluation and delivery models for these therapies, particularly as we expect additional diagnostics and therapeutics to be available in the future. With the advent of commercially available blood tests (ie, blood biomarkers) for AD, there is hope for a transition away from PETs and CSF testing given their cost, limited access, and invasiveness for diagnosis and monitoring of AD. These advances will increase the utility of telehealth to help rural veterans access amyloid-targeting therapies.
Additionally, administering the drug at home or at local clinics, supported by a dedicated health care team or home health agency, could further improve accessibility. Telehealth can be leveraged in this scenario, allowing specialty clinics and specialists to connect with patients and clinicians based out of local clinics or even home health agencies. In this scenario, specialists can provide hands-on care guidance and oversight even though they may be geographically distant from care recipients. Transitioning from IV administration to subcutaneous formulations would further enhance convenience and reduce barriers; these formulations may be available soon.16 Addressing logistical challenges to care and access through technology-based solutions will require coordinated efforts and continued VHA investment.
Conclusions
The VHA has a large population of veterans with dementia, and the costs to care for these veterans will only increase. While the current benefits of amyloid-targeting therapies are modest, now is the time to establish care processes that will support future innovations in amyloid-targeting therapies and other treatments and diagnostics. We are developing better ways to detect AD using clinical decision support tools, improving care pathways and the management of AD, and leveraging telehealth to improve access. The VA is conducting research to investigate whether a cognitive screening and laboratory evaluation that includes a telehealth preparedness assessment will be feasible and effective for improving the detection of AD and access to treatment, and we plan to publish the results.
The lessons learned can be extended to non-VHA care settings to help achieve potential benefits for other patients with early AD. Emerging therapies have the potential to improve the quality of life for both patients and care partners, adding life to years and not just years to life. Policymakers and payors must prioritize research funding to evaluate the safety and efficacy of these approaches to the delivery of health services, ensuring that emerging therapies are accessible for all individuals affected by AD.
- Alzheimer’s Association. 2025 Alzheimer’s disease facts and figures. Alzheimers Dement. 2025;21(4):e70235. doi:10.1002/alz.70235
- US Department of Veterans Affairs. Statistical Projections of Alzheimer’s Dementia for VA Patients, VA Enrollees, and US Veterans. December 18, 2020. Accessed November 2, 2025. https://www.va.gov/GERIATRICS/docs/VHA_ALZHEIMERS_DEMENTIA_Statistical_Projections_FY21_and_FY33_sgc121820.pdf
- Casey DA, Antimisiaris D, O’Brien J. Drugs for Alzheimer’s disease: are they effective? P T. 2010;35(4):208-211.
- Barthold D, Joyce G, Ferido P, et al. Pharmaceutical treatment for Alzheimer’s disease and related dementias: utilization and disparities. J Alzheimers Dis. 2020;76(2):579-589. doi:10.3233/JAD-200133
- Sims JR, Zimmer JA, Evans CD, et al. Donanemab in early symptomatic Alzheimer disease: the TRAILBLAZER-ALZ 2 randomized clinical trial. JAMA. 2023;330(6):512-527. doi:10.1001/jama.2023.13239
- van Dyck CH, Swanson CJ, Aisen P, et al. Lecanemab in early Alzheimer’s disease. N Engl J Med. 2023;388(1):9-21. doi:10.1056/NEJMoa2212948
- Tanne JH. Lecanemab: US Veterans Health Administration will cover cost of new Alzheimer’s drug. BMJ. 2023;380:p628. doi:10.1136/bmj.p628
- Nadeau SE. Lecanemab questions. Neurology. 2024;102(7):e209320. doi:10.1212/WNL.0000000000209320 9. O’Donnell AJ, Fortunato AT, Spitznogle BL, et al. Implementation of lecanemab for Alzheimer’s disease: facilitators and barriers. Presented at: American Geriatrics Society 2025 Annual Scientific Meeting, Chicago. May 2025.
- O’Donnell AJ, Zhao X, Parr A, et al. Use of lecanemab for Alzheimer’s disease within the Veteran’s Health Foundation: early findings. Abstract presented at: Alzheimer’s Association International Conference 2025; July 27, 2025; Toronto, Canada.
- O’Donnell AJ, Zhao X, Parr A, et al. Use of lecanemab for Alzheimer’s disease within the Veteran’s Health Foundation: early findings. Abstract presented at: Alzheimer’s Association International Conference 2025; July 27, 2025; Toronto, Canada.
- Hopp F, Whitten P, Subramanian U, et al. Perspectives from the Veterans Health Administration about opportunities and barriers in telemedicine. J Telemed Telecare. 2006;12(8):404-409. doi:10.1258/135763306779378717
- VA reports significant increase in veteran use of telehealth services. News release. US Department of Veterans Affairs. November 22, 2019. Accessed November 19, 2025. https://news.va.gov/press-room/va-reports-significant-increase-in-veteran-use-of-telehealth-services/
- Powers BB, Homer MC, Morone N, et al. Creation of an interprofessional teledementia clinic for rural veterans: preliminary data. J Am Geriatr Soc. 2017;65(5):1092-1099. doi:10.1111/jgs.14839
- Erickson CM, Chin NA, Rosario HL, et al. Feasibility of virtual Alzheimer’s biomarker disclosure: findings from an observational cohort. Alzheimers Dement (N Y). 2023;9(3):e12413. doi:10.1002/trc2.12413
- Turk KW, Knobel MD, Nothern A, et al. An interprofessional team for disease-modifying therapy in Alzheimer disease implementation. Neurol Clin Pract. 2024;14(6):e200346. doi:10.1212/CPJ.0000000000200346
- FDA accepts LEQEMBI® (lecanemab-irmb) biologics license application for subcutaneous maintenance dosing for the treatment of early Alzheimer’s disease. News release. Elsai US. January 13, 2025. Accessed November 2, 2025. https://media-us.eisai.com/2025-01-13-FDA-Accepts-LEQEMBI-R-lecanemab-irmb-Biologics-License-Application-for-Subcutaneous-Maintenance-Dosing-for-the-Treatment-of-Early-Alzheimers-Disease
The Veterans Health Administration (VHA) is the largest US integrated health care system, providing health care to > 9 million veterans annually. Dementia affects > 7.2 million Americans, and an estimated 450,000 veterans live with Alzheimer disease (AD).1,2 Compared with the general population, veterans have a higher burden of chronic medical conditions and are disproportionately affected by AD due to exposure to military-related risk factors (eg, traumatic brain injury and posttraumatic stress disorder) and the high prevalence of nonmilitary risk factors, such as cardiovascular disease. The VHA is a pioneer in dementia care, having established a Dementia System of Care to provide primary and specialty care to veterans with dementia. The VHA also is leading the way in implementing the Institute for Healthcare Improvement Age-Friendly Health Systems (AFHS) framework for providing goal-concordant care in > 100 VHA medical centers. The VHA aims to be the largest AFHS in the country.
AD profoundly affects individuals and their families. The progressive nature of the most common form of dementia diminishes the quality of life for patients as well as their care partners in an ongoing fashion, often leading to emotional, physical, and financial strain. Costs for health and long-term care for people living with AD and other dementias were projected at $360 billion in 2024, largely due to the need for nursing home care.1 Although several oral medications are available, their capacity to effectively mitigate the negative effects of AD is limited. Cholinesterase inhibitors and memantine may offer temporary symptomatic relief, but they do not alter disease progression.3 The use of these agents is relatively low, with about one-third of patients diagnosed with AD receiving these medications.4
Amyloid-Targeting Therapies
Recent advancements in biologics, particularly amyloid-targeting therapies, such as lecanemab and donanemab, offer new hope for managing AD. Older adults treated with these medications show less decline on measures of cognition and function than those receiving a placebo at 18 months.5,6 However, accessing and using these medications is challenging.
Use of amyloid-targeting therapies poses challenges. The medications are expensive, potentially placing a financial burden on patients, families, and health care systems.7 Determining initial eligibility for treatment requires a battery of cognitive assessments, laboratory tests, advanced radiologic studies (eg, magnetic resonance imaging [MRI] of the brain and amyloid positron emission tomography [PET] scans), and possible cerebrospinal fluid (CSF) testing. Frequent ongoing assessments are necessary to monitor safety and efficacy. These treatments carry substantial risks, particularly amyloid-related imaging abnormalities (ARIA) such as cerebral edema, microhemorrhages, and superficial siderosis. Therefore, follow-up assessments typically occur around months 2, 3, 4, and 7, depending on which medication is selected. Finally, at present, both agents must be intravenous (IV)-administered in a monitored clinical setting, which requires additional coordination, transportation, and cost.
Ongoing evaluations and in-person administration particularly affect patients and care partners with limitations regarding transportation, time off work, and navigating complex health care systems.8 VHA clinicians at sites that have implemented or are interested in implementing amyloid-targeting therapy programs endorse similar challenges when implementing these therapies in their US Department of Veterans Affairs (VA) medical centers (VAMCs).9
The VHA was one of the first health care systems to use amyloid-targeting therapies, covering the cost of lecanemab and donanemab, in addition to costs associated with concomitant evaluation and testing. However, given the safety concerns with this novel class of medications, the VHA National Formulary Committee developed criteria for use and recommended the VA Center for Medication Safety (VAMedSAFE) conduct a mandatory real-time medication use evaluation (MUE). VAMedSAFE developed the MUE to monitor the safe and appropriate use of amyloid-targeting therapy for AD. Two authors (AJO, SMH) partnered with VAMedSAFE through the VA Pittsburgh Healthcare System Technology Enhancing Cognition and Health–Geriatric Research, Education, and Clinical Center (TECH-GRECC) to provide clinical expertise, substantive feedback for the development of the MUE, and guidance for VHA sites starting amyloid targeting-therapy programs. We started a VHA Amyloid-Targeting Therapy for AD SharePoint collaborative platform and VHA AD Therapeutics Community of Practice (CoP) for shared learning (Figure). The private SharePoint platform houses an array of implementation materials for VAMCs starting programs: key documents and links; educational materials; sample guidelines; note templates; and electronic health record screenshots. The CoP allows VHAs to share best practices and discuss challenges.

Even with these advantages, we found that ensuring the safe and appropriate use of amyloid-targeting therapies did not overcome the barriers associated with their complexity. This was especially true for veterans living in rural areas. Only 4 VAMCs had administered amyloid-targeting therapies in the first year they were available. Preliminary data demonstrated that 27 (84%) of 32 veterans who initiated lecanemab in the VHA between October 2023 and September 2024 resided in urban areas.10 To address the underutilization of amyloid-targeting therapy, we propose leveraging the strengths of VHA telehealth to facilitate expansion of access to these medications for veterans with early AD. Telehealth may substantially increase access to evaluation for veterans with early dementia and, when medically appropriate, to receive amyloid-targeting therapies by reducing transportation needs and mitigating costs while ensuring appropriate monitoring through ongoing clinical assessments.
Using Telehealth
The VHA is a pioneer in telehealth, with programs dating back to 2003.11 Between October 1, 2018, and September 30, 2019, the VHA served > 900,000 veterans through the provision of > 2.6 million episodes of care via telehealth.12 The COVID-19 pandemic further cemented the role of telemedicine as an essential component of health care. Telehealth has demonstrated success in the assessment and management of individuals living with dementia. At the VHA, the GRECC-Connect Project is a partnership between 9 urban GRECC sites that seek to provide consultative geriatric and dementia care to rural veterans through telehealth.13 Additional evidence supports the potential to leverage telehealth to effectively communicate results of amyloid PET scans.14
This approach is not without limitations such as the digital divide, or the gap that separates technology-enabled individuals and those unprepared to adopt technology due to limited digital literacy levels or access to needed hardware, software, and connectivity. The VHA has taken steps to address these digital divide barriers by broadly providing tools—such as tablets and broadband connectivity—to veterans. Specifically, the VHA has instituted digital divide consults to determine whether telehealth could be a potential solution for appropriate veterans and to provide an iPad (if eligible) to connect with VA clinicians. Complementary to the digital divide consult, a VHA-specific telehealth preparedness assessment tool is under development and being tested by 2 authors (JF, SMH). This telehealth preparedness assessment tool is designed to aid in the seamless integration of telehealth services with the support of tailored education materials specific to gaps in digital literacy that a veteran might experience.
Building on these initiatives, there is an opportunity to expand access to amyloid-targeting therapies, regardless of distance to large VAMCs, by leveraging telehealth as an alternative method of connecting patients with specialty care. Specifically, a hybrid approach could be used to accomplish the myriad initial and follow-up tasks involved in the provision of amyloid-targeting therapies (Table). Not all VHA facilities possess the specialty expertise to prescribe these medications, and local clinicians may not have sufficient knowledge and clinical support to prescribe and monitor these therapies.

The first step is identifying local and regional subject matter experts, followed by the development and expansion of these networks. The National TeleNeurology Program is a good example of a national telehealth program that leverages technology to bring specialty services to rural areas with limited access to care. Although amyloid-targeting therapies often require more complex logistics, such as laboratory tests and imaging, these initial hurdles can be overcome through localized services and collaboration between VAMCs.
While treatment and imaging will most likely need to occur at a VAMC, most basic laboratory studies can be performed at community-based outpatient clinics (CBOCs). Some CBOCs may not be able to process more specialized laboratory tests such as apolipoprotein E genetic testing. Samples for these tests can be collected and processed at VAMCs, which usually have contracts with outside laboratories capable of performing these studies. Most, although not all, VAMCs offer advanced imaging, including MRI of the brain and amyloid PETs. VAMCs without those modalities may need to coordinate with other regional VAMCs. Additionally, a pilot program is already underway whereby VAMCs without the ability to quantify the amount of amyloid on PETs are able to leverage technology and collaborations with other VAMCs to obtain these data.
Once the initial phases of evaluation and care are completed, telemedicine can be leveraged for follow-up and ongoing management. Interdisciplinary teams can help facilitate care related to amyloid-targeting therapies, including the close monitoring of veterans for development of ARIA.15 To achieve this monitoring, specialty clinic teams prescribing amyloid-targeting therapies, which may be geographically distant, need to coordinate with local primary care clinical teams and emergency clinicians. All of these health care team members, along with neurologists and neurosurgeons, should be involved in the development and implementation of protocols in the event that patients present to their local primary or specialty care clinics or emergency department with ARIA symptoms.
If amyloid-targeting therapies are to be provided along with other emerging treatments for rural veterans, telehealth must be part of the solution. There is a pressing need to explore innovative evaluation and delivery models for these therapies, particularly as we expect additional diagnostics and therapeutics to be available in the future. With the advent of commercially available blood tests (ie, blood biomarkers) for AD, there is hope for a transition away from PETs and CSF testing given their cost, limited access, and invasiveness for diagnosis and monitoring of AD. These advances will increase the utility of telehealth to help rural veterans access amyloid-targeting therapies.
Additionally, administering the drug at home or at local clinics, supported by a dedicated health care team or home health agency, could further improve accessibility. Telehealth can be leveraged in this scenario, allowing specialty clinics and specialists to connect with patients and clinicians based out of local clinics or even home health agencies. In this scenario, specialists can provide hands-on care guidance and oversight even though they may be geographically distant from care recipients. Transitioning from IV administration to subcutaneous formulations would further enhance convenience and reduce barriers; these formulations may be available soon.16 Addressing logistical challenges to care and access through technology-based solutions will require coordinated efforts and continued VHA investment.
Conclusions
The VHA has a large population of veterans with dementia, and the costs to care for these veterans will only increase. While the current benefits of amyloid-targeting therapies are modest, now is the time to establish care processes that will support future innovations in amyloid-targeting therapies and other treatments and diagnostics. We are developing better ways to detect AD using clinical decision support tools, improving care pathways and the management of AD, and leveraging telehealth to improve access. The VA is conducting research to investigate whether a cognitive screening and laboratory evaluation that includes a telehealth preparedness assessment will be feasible and effective for improving the detection of AD and access to treatment, and we plan to publish the results.
The lessons learned can be extended to non-VHA care settings to help achieve potential benefits for other patients with early AD. Emerging therapies have the potential to improve the quality of life for both patients and care partners, adding life to years and not just years to life. Policymakers and payors must prioritize research funding to evaluate the safety and efficacy of these approaches to the delivery of health services, ensuring that emerging therapies are accessible for all individuals affected by AD.
The Veterans Health Administration (VHA) is the largest US integrated health care system, providing health care to > 9 million veterans annually. Dementia affects > 7.2 million Americans, and an estimated 450,000 veterans live with Alzheimer disease (AD).1,2 Compared with the general population, veterans have a higher burden of chronic medical conditions and are disproportionately affected by AD due to exposure to military-related risk factors (eg, traumatic brain injury and posttraumatic stress disorder) and the high prevalence of nonmilitary risk factors, such as cardiovascular disease. The VHA is a pioneer in dementia care, having established a Dementia System of Care to provide primary and specialty care to veterans with dementia. The VHA also is leading the way in implementing the Institute for Healthcare Improvement Age-Friendly Health Systems (AFHS) framework for providing goal-concordant care in > 100 VHA medical centers. The VHA aims to be the largest AFHS in the country.
AD profoundly affects individuals and their families. The progressive nature of the most common form of dementia diminishes the quality of life for patients as well as their care partners in an ongoing fashion, often leading to emotional, physical, and financial strain. Costs for health and long-term care for people living with AD and other dementias were projected at $360 billion in 2024, largely due to the need for nursing home care.1 Although several oral medications are available, their capacity to effectively mitigate the negative effects of AD is limited. Cholinesterase inhibitors and memantine may offer temporary symptomatic relief, but they do not alter disease progression.3 The use of these agents is relatively low, with about one-third of patients diagnosed with AD receiving these medications.4
Amyloid-Targeting Therapies
Recent advancements in biologics, particularly amyloid-targeting therapies, such as lecanemab and donanemab, offer new hope for managing AD. Older adults treated with these medications show less decline on measures of cognition and function than those receiving a placebo at 18 months.5,6 However, accessing and using these medications is challenging.
Use of amyloid-targeting therapies poses challenges. The medications are expensive, potentially placing a financial burden on patients, families, and health care systems.7 Determining initial eligibility for treatment requires a battery of cognitive assessments, laboratory tests, advanced radiologic studies (eg, magnetic resonance imaging [MRI] of the brain and amyloid positron emission tomography [PET] scans), and possible cerebrospinal fluid (CSF) testing. Frequent ongoing assessments are necessary to monitor safety and efficacy. These treatments carry substantial risks, particularly amyloid-related imaging abnormalities (ARIA) such as cerebral edema, microhemorrhages, and superficial siderosis. Therefore, follow-up assessments typically occur around months 2, 3, 4, and 7, depending on which medication is selected. Finally, at present, both agents must be intravenous (IV)-administered in a monitored clinical setting, which requires additional coordination, transportation, and cost.
Ongoing evaluations and in-person administration particularly affect patients and care partners with limitations regarding transportation, time off work, and navigating complex health care systems.8 VHA clinicians at sites that have implemented or are interested in implementing amyloid-targeting therapy programs endorse similar challenges when implementing these therapies in their US Department of Veterans Affairs (VA) medical centers (VAMCs).9
The VHA was one of the first health care systems to use amyloid-targeting therapies, covering the cost of lecanemab and donanemab, in addition to costs associated with concomitant evaluation and testing. However, given the safety concerns with this novel class of medications, the VHA National Formulary Committee developed criteria for use and recommended the VA Center for Medication Safety (VAMedSAFE) conduct a mandatory real-time medication use evaluation (MUE). VAMedSAFE developed the MUE to monitor the safe and appropriate use of amyloid-targeting therapy for AD. Two authors (AJO, SMH) partnered with VAMedSAFE through the VA Pittsburgh Healthcare System Technology Enhancing Cognition and Health–Geriatric Research, Education, and Clinical Center (TECH-GRECC) to provide clinical expertise, substantive feedback for the development of the MUE, and guidance for VHA sites starting amyloid targeting-therapy programs. We started a VHA Amyloid-Targeting Therapy for AD SharePoint collaborative platform and VHA AD Therapeutics Community of Practice (CoP) for shared learning (Figure). The private SharePoint platform houses an array of implementation materials for VAMCs starting programs: key documents and links; educational materials; sample guidelines; note templates; and electronic health record screenshots. The CoP allows VHAs to share best practices and discuss challenges.

Even with these advantages, we found that ensuring the safe and appropriate use of amyloid-targeting therapies did not overcome the barriers associated with their complexity. This was especially true for veterans living in rural areas. Only 4 VAMCs had administered amyloid-targeting therapies in the first year they were available. Preliminary data demonstrated that 27 (84%) of 32 veterans who initiated lecanemab in the VHA between October 2023 and September 2024 resided in urban areas.10 To address the underutilization of amyloid-targeting therapy, we propose leveraging the strengths of VHA telehealth to facilitate expansion of access to these medications for veterans with early AD. Telehealth may substantially increase access to evaluation for veterans with early dementia and, when medically appropriate, to receive amyloid-targeting therapies by reducing transportation needs and mitigating costs while ensuring appropriate monitoring through ongoing clinical assessments.
Using Telehealth
The VHA is a pioneer in telehealth, with programs dating back to 2003.11 Between October 1, 2018, and September 30, 2019, the VHA served > 900,000 veterans through the provision of > 2.6 million episodes of care via telehealth.12 The COVID-19 pandemic further cemented the role of telemedicine as an essential component of health care. Telehealth has demonstrated success in the assessment and management of individuals living with dementia. At the VHA, the GRECC-Connect Project is a partnership between 9 urban GRECC sites that seek to provide consultative geriatric and dementia care to rural veterans through telehealth.13 Additional evidence supports the potential to leverage telehealth to effectively communicate results of amyloid PET scans.14
This approach is not without limitations such as the digital divide, or the gap that separates technology-enabled individuals and those unprepared to adopt technology due to limited digital literacy levels or access to needed hardware, software, and connectivity. The VHA has taken steps to address these digital divide barriers by broadly providing tools—such as tablets and broadband connectivity—to veterans. Specifically, the VHA has instituted digital divide consults to determine whether telehealth could be a potential solution for appropriate veterans and to provide an iPad (if eligible) to connect with VA clinicians. Complementary to the digital divide consult, a VHA-specific telehealth preparedness assessment tool is under development and being tested by 2 authors (JF, SMH). This telehealth preparedness assessment tool is designed to aid in the seamless integration of telehealth services with the support of tailored education materials specific to gaps in digital literacy that a veteran might experience.
Building on these initiatives, there is an opportunity to expand access to amyloid-targeting therapies, regardless of distance to large VAMCs, by leveraging telehealth as an alternative method of connecting patients with specialty care. Specifically, a hybrid approach could be used to accomplish the myriad initial and follow-up tasks involved in the provision of amyloid-targeting therapies (Table). Not all VHA facilities possess the specialty expertise to prescribe these medications, and local clinicians may not have sufficient knowledge and clinical support to prescribe and monitor these therapies.

The first step is identifying local and regional subject matter experts, followed by the development and expansion of these networks. The National TeleNeurology Program is a good example of a national telehealth program that leverages technology to bring specialty services to rural areas with limited access to care. Although amyloid-targeting therapies often require more complex logistics, such as laboratory tests and imaging, these initial hurdles can be overcome through localized services and collaboration between VAMCs.
While treatment and imaging will most likely need to occur at a VAMC, most basic laboratory studies can be performed at community-based outpatient clinics (CBOCs). Some CBOCs may not be able to process more specialized laboratory tests such as apolipoprotein E genetic testing. Samples for these tests can be collected and processed at VAMCs, which usually have contracts with outside laboratories capable of performing these studies. Most, although not all, VAMCs offer advanced imaging, including MRI of the brain and amyloid PETs. VAMCs without those modalities may need to coordinate with other regional VAMCs. Additionally, a pilot program is already underway whereby VAMCs without the ability to quantify the amount of amyloid on PETs are able to leverage technology and collaborations with other VAMCs to obtain these data.
Once the initial phases of evaluation and care are completed, telemedicine can be leveraged for follow-up and ongoing management. Interdisciplinary teams can help facilitate care related to amyloid-targeting therapies, including the close monitoring of veterans for development of ARIA.15 To achieve this monitoring, specialty clinic teams prescribing amyloid-targeting therapies, which may be geographically distant, need to coordinate with local primary care clinical teams and emergency clinicians. All of these health care team members, along with neurologists and neurosurgeons, should be involved in the development and implementation of protocols in the event that patients present to their local primary or specialty care clinics or emergency department with ARIA symptoms.
If amyloid-targeting therapies are to be provided along with other emerging treatments for rural veterans, telehealth must be part of the solution. There is a pressing need to explore innovative evaluation and delivery models for these therapies, particularly as we expect additional diagnostics and therapeutics to be available in the future. With the advent of commercially available blood tests (ie, blood biomarkers) for AD, there is hope for a transition away from PETs and CSF testing given their cost, limited access, and invasiveness for diagnosis and monitoring of AD. These advances will increase the utility of telehealth to help rural veterans access amyloid-targeting therapies.
Additionally, administering the drug at home or at local clinics, supported by a dedicated health care team or home health agency, could further improve accessibility. Telehealth can be leveraged in this scenario, allowing specialty clinics and specialists to connect with patients and clinicians based out of local clinics or even home health agencies. In this scenario, specialists can provide hands-on care guidance and oversight even though they may be geographically distant from care recipients. Transitioning from IV administration to subcutaneous formulations would further enhance convenience and reduce barriers; these formulations may be available soon.16 Addressing logistical challenges to care and access through technology-based solutions will require coordinated efforts and continued VHA investment.
Conclusions
The VHA has a large population of veterans with dementia, and the costs to care for these veterans will only increase. While the current benefits of amyloid-targeting therapies are modest, now is the time to establish care processes that will support future innovations in amyloid-targeting therapies and other treatments and diagnostics. We are developing better ways to detect AD using clinical decision support tools, improving care pathways and the management of AD, and leveraging telehealth to improve access. The VA is conducting research to investigate whether a cognitive screening and laboratory evaluation that includes a telehealth preparedness assessment will be feasible and effective for improving the detection of AD and access to treatment, and we plan to publish the results.
The lessons learned can be extended to non-VHA care settings to help achieve potential benefits for other patients with early AD. Emerging therapies have the potential to improve the quality of life for both patients and care partners, adding life to years and not just years to life. Policymakers and payors must prioritize research funding to evaluate the safety and efficacy of these approaches to the delivery of health services, ensuring that emerging therapies are accessible for all individuals affected by AD.
- Alzheimer’s Association. 2025 Alzheimer’s disease facts and figures. Alzheimers Dement. 2025;21(4):e70235. doi:10.1002/alz.70235
- US Department of Veterans Affairs. Statistical Projections of Alzheimer’s Dementia for VA Patients, VA Enrollees, and US Veterans. December 18, 2020. Accessed November 2, 2025. https://www.va.gov/GERIATRICS/docs/VHA_ALZHEIMERS_DEMENTIA_Statistical_Projections_FY21_and_FY33_sgc121820.pdf
- Casey DA, Antimisiaris D, O’Brien J. Drugs for Alzheimer’s disease: are they effective? P T. 2010;35(4):208-211.
- Barthold D, Joyce G, Ferido P, et al. Pharmaceutical treatment for Alzheimer’s disease and related dementias: utilization and disparities. J Alzheimers Dis. 2020;76(2):579-589. doi:10.3233/JAD-200133
- Sims JR, Zimmer JA, Evans CD, et al. Donanemab in early symptomatic Alzheimer disease: the TRAILBLAZER-ALZ 2 randomized clinical trial. JAMA. 2023;330(6):512-527. doi:10.1001/jama.2023.13239
- van Dyck CH, Swanson CJ, Aisen P, et al. Lecanemab in early Alzheimer’s disease. N Engl J Med. 2023;388(1):9-21. doi:10.1056/NEJMoa2212948
- Tanne JH. Lecanemab: US Veterans Health Administration will cover cost of new Alzheimer’s drug. BMJ. 2023;380:p628. doi:10.1136/bmj.p628
- Nadeau SE. Lecanemab questions. Neurology. 2024;102(7):e209320. doi:10.1212/WNL.0000000000209320 9. O’Donnell AJ, Fortunato AT, Spitznogle BL, et al. Implementation of lecanemab for Alzheimer’s disease: facilitators and barriers. Presented at: American Geriatrics Society 2025 Annual Scientific Meeting, Chicago. May 2025.
- O’Donnell AJ, Zhao X, Parr A, et al. Use of lecanemab for Alzheimer’s disease within the Veteran’s Health Foundation: early findings. Abstract presented at: Alzheimer’s Association International Conference 2025; July 27, 2025; Toronto, Canada.
- O’Donnell AJ, Zhao X, Parr A, et al. Use of lecanemab for Alzheimer’s disease within the Veteran’s Health Foundation: early findings. Abstract presented at: Alzheimer’s Association International Conference 2025; July 27, 2025; Toronto, Canada.
- Hopp F, Whitten P, Subramanian U, et al. Perspectives from the Veterans Health Administration about opportunities and barriers in telemedicine. J Telemed Telecare. 2006;12(8):404-409. doi:10.1258/135763306779378717
- VA reports significant increase in veteran use of telehealth services. News release. US Department of Veterans Affairs. November 22, 2019. Accessed November 19, 2025. https://news.va.gov/press-room/va-reports-significant-increase-in-veteran-use-of-telehealth-services/
- Powers BB, Homer MC, Morone N, et al. Creation of an interprofessional teledementia clinic for rural veterans: preliminary data. J Am Geriatr Soc. 2017;65(5):1092-1099. doi:10.1111/jgs.14839
- Erickson CM, Chin NA, Rosario HL, et al. Feasibility of virtual Alzheimer’s biomarker disclosure: findings from an observational cohort. Alzheimers Dement (N Y). 2023;9(3):e12413. doi:10.1002/trc2.12413
- Turk KW, Knobel MD, Nothern A, et al. An interprofessional team for disease-modifying therapy in Alzheimer disease implementation. Neurol Clin Pract. 2024;14(6):e200346. doi:10.1212/CPJ.0000000000200346
- FDA accepts LEQEMBI® (lecanemab-irmb) biologics license application for subcutaneous maintenance dosing for the treatment of early Alzheimer’s disease. News release. Elsai US. January 13, 2025. Accessed November 2, 2025. https://media-us.eisai.com/2025-01-13-FDA-Accepts-LEQEMBI-R-lecanemab-irmb-Biologics-License-Application-for-Subcutaneous-Maintenance-Dosing-for-the-Treatment-of-Early-Alzheimers-Disease
- Alzheimer’s Association. 2025 Alzheimer’s disease facts and figures. Alzheimers Dement. 2025;21(4):e70235. doi:10.1002/alz.70235
- US Department of Veterans Affairs. Statistical Projections of Alzheimer’s Dementia for VA Patients, VA Enrollees, and US Veterans. December 18, 2020. Accessed November 2, 2025. https://www.va.gov/GERIATRICS/docs/VHA_ALZHEIMERS_DEMENTIA_Statistical_Projections_FY21_and_FY33_sgc121820.pdf
- Casey DA, Antimisiaris D, O’Brien J. Drugs for Alzheimer’s disease: are they effective? P T. 2010;35(4):208-211.
- Barthold D, Joyce G, Ferido P, et al. Pharmaceutical treatment for Alzheimer’s disease and related dementias: utilization and disparities. J Alzheimers Dis. 2020;76(2):579-589. doi:10.3233/JAD-200133
- Sims JR, Zimmer JA, Evans CD, et al. Donanemab in early symptomatic Alzheimer disease: the TRAILBLAZER-ALZ 2 randomized clinical trial. JAMA. 2023;330(6):512-527. doi:10.1001/jama.2023.13239
- van Dyck CH, Swanson CJ, Aisen P, et al. Lecanemab in early Alzheimer’s disease. N Engl J Med. 2023;388(1):9-21. doi:10.1056/NEJMoa2212948
- Tanne JH. Lecanemab: US Veterans Health Administration will cover cost of new Alzheimer’s drug. BMJ. 2023;380:p628. doi:10.1136/bmj.p628
- Nadeau SE. Lecanemab questions. Neurology. 2024;102(7):e209320. doi:10.1212/WNL.0000000000209320 9. O’Donnell AJ, Fortunato AT, Spitznogle BL, et al. Implementation of lecanemab for Alzheimer’s disease: facilitators and barriers. Presented at: American Geriatrics Society 2025 Annual Scientific Meeting, Chicago. May 2025.
- O’Donnell AJ, Zhao X, Parr A, et al. Use of lecanemab for Alzheimer’s disease within the Veteran’s Health Foundation: early findings. Abstract presented at: Alzheimer’s Association International Conference 2025; July 27, 2025; Toronto, Canada.
- O’Donnell AJ, Zhao X, Parr A, et al. Use of lecanemab for Alzheimer’s disease within the Veteran’s Health Foundation: early findings. Abstract presented at: Alzheimer’s Association International Conference 2025; July 27, 2025; Toronto, Canada.
- Hopp F, Whitten P, Subramanian U, et al. Perspectives from the Veterans Health Administration about opportunities and barriers in telemedicine. J Telemed Telecare. 2006;12(8):404-409. doi:10.1258/135763306779378717
- VA reports significant increase in veteran use of telehealth services. News release. US Department of Veterans Affairs. November 22, 2019. Accessed November 19, 2025. https://news.va.gov/press-room/va-reports-significant-increase-in-veteran-use-of-telehealth-services/
- Powers BB, Homer MC, Morone N, et al. Creation of an interprofessional teledementia clinic for rural veterans: preliminary data. J Am Geriatr Soc. 2017;65(5):1092-1099. doi:10.1111/jgs.14839
- Erickson CM, Chin NA, Rosario HL, et al. Feasibility of virtual Alzheimer’s biomarker disclosure: findings from an observational cohort. Alzheimers Dement (N Y). 2023;9(3):e12413. doi:10.1002/trc2.12413
- Turk KW, Knobel MD, Nothern A, et al. An interprofessional team for disease-modifying therapy in Alzheimer disease implementation. Neurol Clin Pract. 2024;14(6):e200346. doi:10.1212/CPJ.0000000000200346
- FDA accepts LEQEMBI® (lecanemab-irmb) biologics license application for subcutaneous maintenance dosing for the treatment of early Alzheimer’s disease. News release. Elsai US. January 13, 2025. Accessed November 2, 2025. https://media-us.eisai.com/2025-01-13-FDA-Accepts-LEQEMBI-R-lecanemab-irmb-Biologics-License-Application-for-Subcutaneous-Maintenance-Dosing-for-the-Treatment-of-Early-Alzheimers-Disease
Can Telehealth Improve Access to Amyloid-Targeting Therapies for Veterans Living With Alzheimer Disease?
Can Telehealth Improve Access to Amyloid-Targeting Therapies for Veterans Living With Alzheimer Disease?
Impact of Blast Exposures
Blast exposure has been associated with a wide range of negative outcomes, including alterations in brain structure and function, poorer cognitive functioning, and increased severity of psychiatric and health symptoms. Long-term effects also include chronic secondary downstream effects, such as neuroinflammation, neurotoxicity, cellular senescence, and neurodegeneration.
Now, a recent US Department of Veterans Affairs (VA) study of 114 post-9/11 combat veterans suggests that lifetime blast exposure severity is independently associated with accelerated epigenetic aging, even after accounting for PTSD and TBI. The field of epigenetics refers to how environment influences genes by changing the chemicals attached to them.
This cross-sectional study analyzed participants enrolled in 2 coordinated VA research protocols: the Chronic Effects of Neurotrauma Consortium Study 34 and the Post-Deployment Mental Health Study. Researchers measured biological aging using DunedinPACE, an epigenetic biomarker derived from whole-blood DNA methylation data.
Greater blast exposure severity was significantly associated with faster DunedinPACE. Mild TBI history was also independently associated with faster aging, whereas PTSD diagnosis was not. No significant interaction effects were observed. Exploratory analyses suggested that higher-intensity and more frequent blast exposures contributed to more accelerated aging.
The researchers said their findings suggest that accelerated biological aging may represent a pathway linking blast exposure to increased vulnerability for age-related disease and could inform early identification of at-risk veterans.
Preclinical work has “undeniably demonstrated that primary blast forces can directly induce neurotrauma with associated, ongoing symptoms,” according to the authors of a 2024 study. “[H]owever, these findings have not translated into clinical work.” Most human studies of blast exposure use data obtained from assessments of TBI. That approach is limited, they said, because blast exposure does not always result in symptoms of concussion or TBI, and clinical symptoms of TBI are not necessary for blast-induced neurotrauma to occur.
Moreover, understanding how and why blast exposure often results in negative consequences is still lagging, and interventions and treatments have lagged comparatively, the researchers noted. In large part, they added, this is because there is no broadly endorsed definition of blast exposure. They illustrated their point with examples of terms used in earlier research: blast TBI, primary blast TBI, pressure severity, distance from the blast, and frequency of exposure. The lack of standardized language, they suggested, “prevents synthesis of existing literature into a cohesive understanding of the field.”
Those researchers called for concerted and collaborative efforts to advance the study of blast exposure, including developing a standardized definition of blast exposure and curating an empirical literature base allowing clear comparisons of results across studies. They also urged raising awareness about blast-related negative outcomes with education at all levels: continuing education opportunities, round tables at annual conference meetings, grand rounds in hospital or academic medical center settings, and journal clubs.
Blast exposure has been associated with a wide range of negative outcomes, including alterations in brain structure and function, poorer cognitive functioning, and increased severity of psychiatric and health symptoms. Long-term effects also include chronic secondary downstream effects, such as neuroinflammation, neurotoxicity, cellular senescence, and neurodegeneration.
Now, a recent US Department of Veterans Affairs (VA) study of 114 post-9/11 combat veterans suggests that lifetime blast exposure severity is independently associated with accelerated epigenetic aging, even after accounting for PTSD and TBI. The field of epigenetics refers to how environment influences genes by changing the chemicals attached to them.
This cross-sectional study analyzed participants enrolled in 2 coordinated VA research protocols: the Chronic Effects of Neurotrauma Consortium Study 34 and the Post-Deployment Mental Health Study. Researchers measured biological aging using DunedinPACE, an epigenetic biomarker derived from whole-blood DNA methylation data.
Greater blast exposure severity was significantly associated with faster DunedinPACE. Mild TBI history was also independently associated with faster aging, whereas PTSD diagnosis was not. No significant interaction effects were observed. Exploratory analyses suggested that higher-intensity and more frequent blast exposures contributed to more accelerated aging.
The researchers said their findings suggest that accelerated biological aging may represent a pathway linking blast exposure to increased vulnerability for age-related disease and could inform early identification of at-risk veterans.
Preclinical work has “undeniably demonstrated that primary blast forces can directly induce neurotrauma with associated, ongoing symptoms,” according to the authors of a 2024 study. “[H]owever, these findings have not translated into clinical work.” Most human studies of blast exposure use data obtained from assessments of TBI. That approach is limited, they said, because blast exposure does not always result in symptoms of concussion or TBI, and clinical symptoms of TBI are not necessary for blast-induced neurotrauma to occur.
Moreover, understanding how and why blast exposure often results in negative consequences is still lagging, and interventions and treatments have lagged comparatively, the researchers noted. In large part, they added, this is because there is no broadly endorsed definition of blast exposure. They illustrated their point with examples of terms used in earlier research: blast TBI, primary blast TBI, pressure severity, distance from the blast, and frequency of exposure. The lack of standardized language, they suggested, “prevents synthesis of existing literature into a cohesive understanding of the field.”
Those researchers called for concerted and collaborative efforts to advance the study of blast exposure, including developing a standardized definition of blast exposure and curating an empirical literature base allowing clear comparisons of results across studies. They also urged raising awareness about blast-related negative outcomes with education at all levels: continuing education opportunities, round tables at annual conference meetings, grand rounds in hospital or academic medical center settings, and journal clubs.
Blast exposure has been associated with a wide range of negative outcomes, including alterations in brain structure and function, poorer cognitive functioning, and increased severity of psychiatric and health symptoms. Long-term effects also include chronic secondary downstream effects, such as neuroinflammation, neurotoxicity, cellular senescence, and neurodegeneration.
Now, a recent US Department of Veterans Affairs (VA) study of 114 post-9/11 combat veterans suggests that lifetime blast exposure severity is independently associated with accelerated epigenetic aging, even after accounting for PTSD and TBI. The field of epigenetics refers to how environment influences genes by changing the chemicals attached to them.
This cross-sectional study analyzed participants enrolled in 2 coordinated VA research protocols: the Chronic Effects of Neurotrauma Consortium Study 34 and the Post-Deployment Mental Health Study. Researchers measured biological aging using DunedinPACE, an epigenetic biomarker derived from whole-blood DNA methylation data.
Greater blast exposure severity was significantly associated with faster DunedinPACE. Mild TBI history was also independently associated with faster aging, whereas PTSD diagnosis was not. No significant interaction effects were observed. Exploratory analyses suggested that higher-intensity and more frequent blast exposures contributed to more accelerated aging.
The researchers said their findings suggest that accelerated biological aging may represent a pathway linking blast exposure to increased vulnerability for age-related disease and could inform early identification of at-risk veterans.
Preclinical work has “undeniably demonstrated that primary blast forces can directly induce neurotrauma with associated, ongoing symptoms,” according to the authors of a 2024 study. “[H]owever, these findings have not translated into clinical work.” Most human studies of blast exposure use data obtained from assessments of TBI. That approach is limited, they said, because blast exposure does not always result in symptoms of concussion or TBI, and clinical symptoms of TBI are not necessary for blast-induced neurotrauma to occur.
Moreover, understanding how and why blast exposure often results in negative consequences is still lagging, and interventions and treatments have lagged comparatively, the researchers noted. In large part, they added, this is because there is no broadly endorsed definition of blast exposure. They illustrated their point with examples of terms used in earlier research: blast TBI, primary blast TBI, pressure severity, distance from the blast, and frequency of exposure. The lack of standardized language, they suggested, “prevents synthesis of existing literature into a cohesive understanding of the field.”
Those researchers called for concerted and collaborative efforts to advance the study of blast exposure, including developing a standardized definition of blast exposure and curating an empirical literature base allowing clear comparisons of results across studies. They also urged raising awareness about blast-related negative outcomes with education at all levels: continuing education opportunities, round tables at annual conference meetings, grand rounds in hospital or academic medical center settings, and journal clubs.
Burn Pit Exposure Linked to Higher Neurologic Risk in Veterans
CHICAGO — Exposure to toxic smoke from military burn pits may be putting millions of US veterans at risk for neurologic disorders, new research suggested, raising fresh questions about the long-term health consequences of a widely used wartime waste-disposal practice.
Investigators found that veterans who lived or worked near the open-air pits — used to burn everything from plastics to medical waste — were significantly more likely to develop conditions including headache and vertigo.
“It’s been estimated that over time, approximately 4 million veterans may have been exposed to the combustion of plastics, metals, medical waste, human waste, and other chemicals,” said study investigator Sarah Anthony, research assistant with the VA Headache Centers of Excellence and the Department of Neurology at Yale School of Medicine, New Haven, Connecticut.
The findings were presented on April 21 at the American Academy of Neurology (AAN) 2026 Annual Meeting.
Toxic Legacy
Open burn pits — large, open-air sites used to burn military waste — were widely used during US deployments in Iraq and Afghanistan after 9/11. The fires produced thick plumes of smoke containing a complex mix of hazardous pollutants, including fine particulate matter, volatile organic compounds, and carcinogenic chemicals, exposing service members who lived and worked nearby.
Amid growing concerns, the Department of Veterans Affairs (VA) established the Airborne Hazards and Open Burn Pit Registry in 2014 to study the long-term health effects of deployment-related airborne exposures, including burn pits, Anthony said.
Since its inception, multiple studies have linked burn pit exposure to respiratory conditions such as chronic bronchitis and chronic obstructive pulmonary disease, as well as cardiovascular issues such as hypertension. More recently, Anthony et al reported an association between burn pit exposure and incident headache.
To better understand whether those risks extend beyond previously reported conditions, the researchers set out to examine the relationship between burn pit exposure and a broader range of neurologic disorders.
Analyzing data from > 245,000 registry participants, researchers found that 66% had ≥ 1 neurologic diagnosis, largely driven by common conditions such as headache.
Those with the highest exposure — living near burn pits and performing related duties — had 36% higher odds of developing a neurologic disorder than less-exposed veterans (odds ratio [OR], 1.36).
The risk for any headache disorder was 57% higher (OR, 1.57) and the risk for vertigo was 25% higher (OR, 1.25) in those with the highest exposure levels than in their peers with lower burn pit exposure.
For every additional 6 months of exposure, the odds of developing any neurologic disorder, headache, or vertigo continued to rise, reinforcing concerns about long-term harm to the brain.
There were no statistically significant associations between burn pit exposure and several other neurologic diseases, including epilepsy and amyotrophic lateral sclerosis.
The researchers also found an inverse association between burn pit exposure and Parkinson’s disease, meaning exposed veterans appeared less likely to be diagnosed with the disorder. However, Anthony cautioned that this may reflect the relatively young age of the study population rather than a true protective effect.
Anthony emphasized that the findings are preliminary and may underestimate long-term risks as many neurologic diseases develop over decades. She also noted that burn pit exposure was based on self-reported registry data, which has the potential to introduce bias.
Additional work is needed to understand the neurologic sequela of deployment-related airborne hazards, including exposure to open burn pits “as this remains important for veteran health policy, long term surveillance, and clinical care,” she said.
Long-term monitoring is critical, particularly as exposed veterans age and further studies should consider leveraging data from the VA toxic exposure screening initiatives, which are now part of routine care, as mandated by the Promise to Address Comprehensive Toxics Act.
Predictable Associations, More Study Needed
These findings, said David D. Lo, MD, PhD, distinguished professor of biomedical sciences, University of California Riverside, School of Medicine, said this is “another study that aims to highlight an association between possible burn pit exposure and a variety of health effects.”
Given the well-established risks of inhaling smoke from burning mixed waste, the findings are not surprising, said Lo, who was not involved in the research.
He noted that the study has important limitations, particularly because exposure was based on self-reports, making it difficult to accurately measure how much smoke individual participants were actually exposed to.
“If these findings are suggestive, it is hoped that they spur very real direct funding for more detailed clinical studies on the mechanisms of how the burn pit exposure would actually be responsible for one or more of the clinical outcomes listed in this study, instead of just adding up more statistical correlations,” said Lo.
This study had no commercial funding. Disclosure information for study authors is available in the original study publication. Lo reported having no relevant disclosures.
A version of this article appeared on Medscape.com.
CHICAGO — Exposure to toxic smoke from military burn pits may be putting millions of US veterans at risk for neurologic disorders, new research suggested, raising fresh questions about the long-term health consequences of a widely used wartime waste-disposal practice.
Investigators found that veterans who lived or worked near the open-air pits — used to burn everything from plastics to medical waste — were significantly more likely to develop conditions including headache and vertigo.
“It’s been estimated that over time, approximately 4 million veterans may have been exposed to the combustion of plastics, metals, medical waste, human waste, and other chemicals,” said study investigator Sarah Anthony, research assistant with the VA Headache Centers of Excellence and the Department of Neurology at Yale School of Medicine, New Haven, Connecticut.
The findings were presented on April 21 at the American Academy of Neurology (AAN) 2026 Annual Meeting.
Toxic Legacy
Open burn pits — large, open-air sites used to burn military waste — were widely used during US deployments in Iraq and Afghanistan after 9/11. The fires produced thick plumes of smoke containing a complex mix of hazardous pollutants, including fine particulate matter, volatile organic compounds, and carcinogenic chemicals, exposing service members who lived and worked nearby.
Amid growing concerns, the Department of Veterans Affairs (VA) established the Airborne Hazards and Open Burn Pit Registry in 2014 to study the long-term health effects of deployment-related airborne exposures, including burn pits, Anthony said.
Since its inception, multiple studies have linked burn pit exposure to respiratory conditions such as chronic bronchitis and chronic obstructive pulmonary disease, as well as cardiovascular issues such as hypertension. More recently, Anthony et al reported an association between burn pit exposure and incident headache.
To better understand whether those risks extend beyond previously reported conditions, the researchers set out to examine the relationship between burn pit exposure and a broader range of neurologic disorders.
Analyzing data from > 245,000 registry participants, researchers found that 66% had ≥ 1 neurologic diagnosis, largely driven by common conditions such as headache.
Those with the highest exposure — living near burn pits and performing related duties — had 36% higher odds of developing a neurologic disorder than less-exposed veterans (odds ratio [OR], 1.36).
The risk for any headache disorder was 57% higher (OR, 1.57) and the risk for vertigo was 25% higher (OR, 1.25) in those with the highest exposure levels than in their peers with lower burn pit exposure.
For every additional 6 months of exposure, the odds of developing any neurologic disorder, headache, or vertigo continued to rise, reinforcing concerns about long-term harm to the brain.
There were no statistically significant associations between burn pit exposure and several other neurologic diseases, including epilepsy and amyotrophic lateral sclerosis.
The researchers also found an inverse association between burn pit exposure and Parkinson’s disease, meaning exposed veterans appeared less likely to be diagnosed with the disorder. However, Anthony cautioned that this may reflect the relatively young age of the study population rather than a true protective effect.
Anthony emphasized that the findings are preliminary and may underestimate long-term risks as many neurologic diseases develop over decades. She also noted that burn pit exposure was based on self-reported registry data, which has the potential to introduce bias.
Additional work is needed to understand the neurologic sequela of deployment-related airborne hazards, including exposure to open burn pits “as this remains important for veteran health policy, long term surveillance, and clinical care,” she said.
Long-term monitoring is critical, particularly as exposed veterans age and further studies should consider leveraging data from the VA toxic exposure screening initiatives, which are now part of routine care, as mandated by the Promise to Address Comprehensive Toxics Act.
Predictable Associations, More Study Needed
These findings, said David D. Lo, MD, PhD, distinguished professor of biomedical sciences, University of California Riverside, School of Medicine, said this is “another study that aims to highlight an association between possible burn pit exposure and a variety of health effects.”
Given the well-established risks of inhaling smoke from burning mixed waste, the findings are not surprising, said Lo, who was not involved in the research.
He noted that the study has important limitations, particularly because exposure was based on self-reports, making it difficult to accurately measure how much smoke individual participants were actually exposed to.
“If these findings are suggestive, it is hoped that they spur very real direct funding for more detailed clinical studies on the mechanisms of how the burn pit exposure would actually be responsible for one or more of the clinical outcomes listed in this study, instead of just adding up more statistical correlations,” said Lo.
This study had no commercial funding. Disclosure information for study authors is available in the original study publication. Lo reported having no relevant disclosures.
A version of this article appeared on Medscape.com.
CHICAGO — Exposure to toxic smoke from military burn pits may be putting millions of US veterans at risk for neurologic disorders, new research suggested, raising fresh questions about the long-term health consequences of a widely used wartime waste-disposal practice.
Investigators found that veterans who lived or worked near the open-air pits — used to burn everything from plastics to medical waste — were significantly more likely to develop conditions including headache and vertigo.
“It’s been estimated that over time, approximately 4 million veterans may have been exposed to the combustion of plastics, metals, medical waste, human waste, and other chemicals,” said study investigator Sarah Anthony, research assistant with the VA Headache Centers of Excellence and the Department of Neurology at Yale School of Medicine, New Haven, Connecticut.
The findings were presented on April 21 at the American Academy of Neurology (AAN) 2026 Annual Meeting.
Toxic Legacy
Open burn pits — large, open-air sites used to burn military waste — were widely used during US deployments in Iraq and Afghanistan after 9/11. The fires produced thick plumes of smoke containing a complex mix of hazardous pollutants, including fine particulate matter, volatile organic compounds, and carcinogenic chemicals, exposing service members who lived and worked nearby.
Amid growing concerns, the Department of Veterans Affairs (VA) established the Airborne Hazards and Open Burn Pit Registry in 2014 to study the long-term health effects of deployment-related airborne exposures, including burn pits, Anthony said.
Since its inception, multiple studies have linked burn pit exposure to respiratory conditions such as chronic bronchitis and chronic obstructive pulmonary disease, as well as cardiovascular issues such as hypertension. More recently, Anthony et al reported an association between burn pit exposure and incident headache.
To better understand whether those risks extend beyond previously reported conditions, the researchers set out to examine the relationship between burn pit exposure and a broader range of neurologic disorders.
Analyzing data from > 245,000 registry participants, researchers found that 66% had ≥ 1 neurologic diagnosis, largely driven by common conditions such as headache.
Those with the highest exposure — living near burn pits and performing related duties — had 36% higher odds of developing a neurologic disorder than less-exposed veterans (odds ratio [OR], 1.36).
The risk for any headache disorder was 57% higher (OR, 1.57) and the risk for vertigo was 25% higher (OR, 1.25) in those with the highest exposure levels than in their peers with lower burn pit exposure.
For every additional 6 months of exposure, the odds of developing any neurologic disorder, headache, or vertigo continued to rise, reinforcing concerns about long-term harm to the brain.
There were no statistically significant associations between burn pit exposure and several other neurologic diseases, including epilepsy and amyotrophic lateral sclerosis.
The researchers also found an inverse association between burn pit exposure and Parkinson’s disease, meaning exposed veterans appeared less likely to be diagnosed with the disorder. However, Anthony cautioned that this may reflect the relatively young age of the study population rather than a true protective effect.
Anthony emphasized that the findings are preliminary and may underestimate long-term risks as many neurologic diseases develop over decades. She also noted that burn pit exposure was based on self-reported registry data, which has the potential to introduce bias.
Additional work is needed to understand the neurologic sequela of deployment-related airborne hazards, including exposure to open burn pits “as this remains important for veteran health policy, long term surveillance, and clinical care,” she said.
Long-term monitoring is critical, particularly as exposed veterans age and further studies should consider leveraging data from the VA toxic exposure screening initiatives, which are now part of routine care, as mandated by the Promise to Address Comprehensive Toxics Act.
Predictable Associations, More Study Needed
These findings, said David D. Lo, MD, PhD, distinguished professor of biomedical sciences, University of California Riverside, School of Medicine, said this is “another study that aims to highlight an association between possible burn pit exposure and a variety of health effects.”
Given the well-established risks of inhaling smoke from burning mixed waste, the findings are not surprising, said Lo, who was not involved in the research.
He noted that the study has important limitations, particularly because exposure was based on self-reports, making it difficult to accurately measure how much smoke individual participants were actually exposed to.
“If these findings are suggestive, it is hoped that they spur very real direct funding for more detailed clinical studies on the mechanisms of how the burn pit exposure would actually be responsible for one or more of the clinical outcomes listed in this study, instead of just adding up more statistical correlations,” said Lo.
This study had no commercial funding. Disclosure information for study authors is available in the original study publication. Lo reported having no relevant disclosures.
A version of this article appeared on Medscape.com.
FROM AAN 2026
The BEACON Act: Partnership, Privatization, or Both?
Seeking to modernize treatment for traumatic brain injury (TBI), Reps. Jack Bergman (R-MI) and Sarah Elfreth (D-MD) introduced the bipartisan BEACON Act to Congress on January 9. The legislation aims to expand access to innovative, evidence-based, nonpharmacological therapies to treat TBI beyond medication-centered approaches that do not always address the long-term and individualized needs of these veterans. These current methods leave “gaps in recovery, wellness, and post-service outcomes,” Bergman and Elfreth argued.
During a March 5 House Committee on Veterans’ Affairs Subcommittee on Health hearing, discussion centered on the proposed BEACON Act, as well as the additional challenges Neurology Centers of Excellence (CoEs) face to address TBI in veterans.
The act proposes awarding $60 million in grants over 3 years to private entities for TBI treatment and research and establishing 2 US Department of Veterans Affairs (VA) grant programs. The TBI Innovation Grant Program would support clinical studies and partnerships between community health care institutions, academic institutions, and the VA. The Independent Research Grant Program would advance third-party research and “implementation of proven alternative treatments,” with oversight by an independent entity modeled after the VA National Center for PTSD.
The proposed legislation has drawn criticism. “I do not disagree that veterans may need support from several different avenues to support their recovery journeys and I don't discount the role that nonprofits and academic affiliates play in facilitating and supporting that care,” said Ranking Member Rep. Julia Brownley (D-CA) said. “However, I need to draw the line at legislation that will take money from existing VA programs and redirect it to outside organizations and providers to do essentially the very same thing VA is already doing.”
Russell Gore, MD, a neurologist and chief medical officer of Avalon Action Alliance, called VA TBI care fragmented and said the BEACON Act offers an opportunity to enhance it.
“This legislation is designed to evaluate effective treatments and leverage civilian and academic TBI expertise that is aligned with the VA’s mission,” he said. “This is not an attempt to privatize care, but to complement VA research and clinical capacity… With smart, coordinated partnerships and targeted investment, we can reach more veterans earlier, treat them more effectively.”
The VA has 5 polytrauma rehabilitation centers, 23 polytrauma network sites, and numerous clinics supporting > 110 TBI teams. It also has 42 CoEs related to neurology.
In a prepared statement, Glenn Graham, MD, PhD, retired Executive Director of the VA’s Neurology Clinical Programs representing the Association of VA Neurology Services cited the CoEs’ contribution to standardization of care. “Without systemwide coordination, practice patterns can vary. A veteran in a rural facility should receive the same standard of neurological assessment and management as a veteran treated in one of our flagship medical centers,” he said, before highlighting the capabilities of tele-neurology, electronic consultation, and remote interpretation of diagnostic studies to reduce travel burdens and promote equity in access.
Graham cautioned, though, that the CoEs face challenges with budgeting and recent VA reductions in force. The proposed legislation, Graham said, would use VA appropriations to fund extramural research and “could drain vital resources from ongoing research, training and clinical programs, diverting funds to institutions with uncertain track records and limited experience working with the veteran population.”
Several people highlighted the world-renowned research coming out of the VA, efforts that both veterans and the general public endorse.
Russell Lemle, former chief psychologist for the San Francisco VA Healthcare System and a senior policy analyst at the Veterans Healthcare Policy Institute, wrote with Jasper Craven: “The private sector has nothing commensurate with this level of care. And yet this bill would push TBI treatment out to private grantees, part of the accelerating movement to privatize the entire VA—even its signature, best-in-class programs.
“The act aims to divert resources from the VA’s world-class TBI and PTSD programs by creating a parallel treatment framework.”
Gore, however, said the Avalon Action Alliance supports a “fill-the-void” approach of “capacity augmentation, not privatization.”
“The intent is to complement VA by partnering with high-performing programs capable of delivering comprehensive assessment, interdisciplinary treatment, and structured follow-up for veterans who are not effectively reached (or not successfully retained) within traditional pathways,” he said.
Seeking to modernize treatment for traumatic brain injury (TBI), Reps. Jack Bergman (R-MI) and Sarah Elfreth (D-MD) introduced the bipartisan BEACON Act to Congress on January 9. The legislation aims to expand access to innovative, evidence-based, nonpharmacological therapies to treat TBI beyond medication-centered approaches that do not always address the long-term and individualized needs of these veterans. These current methods leave “gaps in recovery, wellness, and post-service outcomes,” Bergman and Elfreth argued.
During a March 5 House Committee on Veterans’ Affairs Subcommittee on Health hearing, discussion centered on the proposed BEACON Act, as well as the additional challenges Neurology Centers of Excellence (CoEs) face to address TBI in veterans.
The act proposes awarding $60 million in grants over 3 years to private entities for TBI treatment and research and establishing 2 US Department of Veterans Affairs (VA) grant programs. The TBI Innovation Grant Program would support clinical studies and partnerships between community health care institutions, academic institutions, and the VA. The Independent Research Grant Program would advance third-party research and “implementation of proven alternative treatments,” with oversight by an independent entity modeled after the VA National Center for PTSD.
The proposed legislation has drawn criticism. “I do not disagree that veterans may need support from several different avenues to support their recovery journeys and I don't discount the role that nonprofits and academic affiliates play in facilitating and supporting that care,” said Ranking Member Rep. Julia Brownley (D-CA) said. “However, I need to draw the line at legislation that will take money from existing VA programs and redirect it to outside organizations and providers to do essentially the very same thing VA is already doing.”
Russell Gore, MD, a neurologist and chief medical officer of Avalon Action Alliance, called VA TBI care fragmented and said the BEACON Act offers an opportunity to enhance it.
“This legislation is designed to evaluate effective treatments and leverage civilian and academic TBI expertise that is aligned with the VA’s mission,” he said. “This is not an attempt to privatize care, but to complement VA research and clinical capacity… With smart, coordinated partnerships and targeted investment, we can reach more veterans earlier, treat them more effectively.”
The VA has 5 polytrauma rehabilitation centers, 23 polytrauma network sites, and numerous clinics supporting > 110 TBI teams. It also has 42 CoEs related to neurology.
In a prepared statement, Glenn Graham, MD, PhD, retired Executive Director of the VA’s Neurology Clinical Programs representing the Association of VA Neurology Services cited the CoEs’ contribution to standardization of care. “Without systemwide coordination, practice patterns can vary. A veteran in a rural facility should receive the same standard of neurological assessment and management as a veteran treated in one of our flagship medical centers,” he said, before highlighting the capabilities of tele-neurology, electronic consultation, and remote interpretation of diagnostic studies to reduce travel burdens and promote equity in access.
Graham cautioned, though, that the CoEs face challenges with budgeting and recent VA reductions in force. The proposed legislation, Graham said, would use VA appropriations to fund extramural research and “could drain vital resources from ongoing research, training and clinical programs, diverting funds to institutions with uncertain track records and limited experience working with the veteran population.”
Several people highlighted the world-renowned research coming out of the VA, efforts that both veterans and the general public endorse.
Russell Lemle, former chief psychologist for the San Francisco VA Healthcare System and a senior policy analyst at the Veterans Healthcare Policy Institute, wrote with Jasper Craven: “The private sector has nothing commensurate with this level of care. And yet this bill would push TBI treatment out to private grantees, part of the accelerating movement to privatize the entire VA—even its signature, best-in-class programs.
“The act aims to divert resources from the VA’s world-class TBI and PTSD programs by creating a parallel treatment framework.”
Gore, however, said the Avalon Action Alliance supports a “fill-the-void” approach of “capacity augmentation, not privatization.”
“The intent is to complement VA by partnering with high-performing programs capable of delivering comprehensive assessment, interdisciplinary treatment, and structured follow-up for veterans who are not effectively reached (or not successfully retained) within traditional pathways,” he said.
Seeking to modernize treatment for traumatic brain injury (TBI), Reps. Jack Bergman (R-MI) and Sarah Elfreth (D-MD) introduced the bipartisan BEACON Act to Congress on January 9. The legislation aims to expand access to innovative, evidence-based, nonpharmacological therapies to treat TBI beyond medication-centered approaches that do not always address the long-term and individualized needs of these veterans. These current methods leave “gaps in recovery, wellness, and post-service outcomes,” Bergman and Elfreth argued.
During a March 5 House Committee on Veterans’ Affairs Subcommittee on Health hearing, discussion centered on the proposed BEACON Act, as well as the additional challenges Neurology Centers of Excellence (CoEs) face to address TBI in veterans.
The act proposes awarding $60 million in grants over 3 years to private entities for TBI treatment and research and establishing 2 US Department of Veterans Affairs (VA) grant programs. The TBI Innovation Grant Program would support clinical studies and partnerships between community health care institutions, academic institutions, and the VA. The Independent Research Grant Program would advance third-party research and “implementation of proven alternative treatments,” with oversight by an independent entity modeled after the VA National Center for PTSD.
The proposed legislation has drawn criticism. “I do not disagree that veterans may need support from several different avenues to support their recovery journeys and I don't discount the role that nonprofits and academic affiliates play in facilitating and supporting that care,” said Ranking Member Rep. Julia Brownley (D-CA) said. “However, I need to draw the line at legislation that will take money from existing VA programs and redirect it to outside organizations and providers to do essentially the very same thing VA is already doing.”
Russell Gore, MD, a neurologist and chief medical officer of Avalon Action Alliance, called VA TBI care fragmented and said the BEACON Act offers an opportunity to enhance it.
“This legislation is designed to evaluate effective treatments and leverage civilian and academic TBI expertise that is aligned with the VA’s mission,” he said. “This is not an attempt to privatize care, but to complement VA research and clinical capacity… With smart, coordinated partnerships and targeted investment, we can reach more veterans earlier, treat them more effectively.”
The VA has 5 polytrauma rehabilitation centers, 23 polytrauma network sites, and numerous clinics supporting > 110 TBI teams. It also has 42 CoEs related to neurology.
In a prepared statement, Glenn Graham, MD, PhD, retired Executive Director of the VA’s Neurology Clinical Programs representing the Association of VA Neurology Services cited the CoEs’ contribution to standardization of care. “Without systemwide coordination, practice patterns can vary. A veteran in a rural facility should receive the same standard of neurological assessment and management as a veteran treated in one of our flagship medical centers,” he said, before highlighting the capabilities of tele-neurology, electronic consultation, and remote interpretation of diagnostic studies to reduce travel burdens and promote equity in access.
Graham cautioned, though, that the CoEs face challenges with budgeting and recent VA reductions in force. The proposed legislation, Graham said, would use VA appropriations to fund extramural research and “could drain vital resources from ongoing research, training and clinical programs, diverting funds to institutions with uncertain track records and limited experience working with the veteran population.”
Several people highlighted the world-renowned research coming out of the VA, efforts that both veterans and the general public endorse.
Russell Lemle, former chief psychologist for the San Francisco VA Healthcare System and a senior policy analyst at the Veterans Healthcare Policy Institute, wrote with Jasper Craven: “The private sector has nothing commensurate with this level of care. And yet this bill would push TBI treatment out to private grantees, part of the accelerating movement to privatize the entire VA—even its signature, best-in-class programs.
“The act aims to divert resources from the VA’s world-class TBI and PTSD programs by creating a parallel treatment framework.”
Gore, however, said the Avalon Action Alliance supports a “fill-the-void” approach of “capacity augmentation, not privatization.”
“The intent is to complement VA by partnering with high-performing programs capable of delivering comprehensive assessment, interdisciplinary treatment, and structured follow-up for veterans who are not effectively reached (or not successfully retained) within traditional pathways,” he said.
Hearing Addresses Neurology CoE Challenges
Hearing Addresses Neurology CoE Challenges
On January 8, 2020, Iran fired 15 ballistic missiles at the Al-Asad Airbase, where Alan Johnson, an Army Lieutenant Colonel and Aeromedical Physician Assistant, was deployed.
“I have no memory of the first 3 missile impacts because the third missile impact knocked me unconscious,” Johnson said in a statement to a House Committee on Veterans’ Affairs subcommittee on Health in a March 5 hearing. “I woke up just in time to experience missiles 4, 5, and 6.”
March is Brain Injury Awareness month, highlighting how nearly 1 in 4 veterans has screened positive for probable traumatic brain injury (TBI). Veterans with TBI also have a higher risk of suicide: in 2023, the suicide rate for veterans with a recent TBI diagnosis was > 94% higher than for veterans without a TBI diagnosis.
“For many veterans, TBI is not a single episode of care; it is a chronic neurological condition requiring coordinated, longitudinal management,” Glenn D. Graham, MD, PhD, president of the Association of VA Neurology Service (AVANS) and former executive director of the US Department of Veterans Affairs (VA) Neurology Clinical Programs said in a statement. “TBI is neurologically complex and often intertwined with other conditions … Accurate diagnosis and effective treatment require subspecialty expertise in areas such as epilepsy, headache medicine, and neurodegenerative disease. The Centers of Excellence (CoE) ensure that this expertise is available across our national system.”
An estimated 25% of service members who have been hospitalized with TBI will develop long-term disability. Studies show direct links between TBI and the development of neurological disorders. Lt. Col. Johnson, for instance, has been diagnosed with posttraumatic stress disorder, cranial nerve damage, double vision, chronic insomnia, ringing in the ears, neck pain, balance problems, difficulty in word finding, and depression. After 37 years in emergency medicine, Johnson said, he had to “bench” himself due to the sequelae: “I can’t do what I love to do anymore.”
However, many service members may not be diagnosed correctly. Blast-related brain injuries may be delayed, subtle, and easily missed in combat environments. In research Johnson coauthored, > 20% of troops were diagnosed with mild TBIs 4 weeks after the attack. Moreover, he said, soldiers being screened may underreport their symptoms in order to return to duty.
Timely diagnosis is key, but so is consistent follow-up. Ranking Member Rep. Julia Brownley (D-CA) said, “TBI is not an illness that goes away with medicine … It is a long-term chronic condition for which many veterans need ongoing integrated and well-coordinated care.”
The Veterans Health Administration (VHA) has 5 polytrauma rehabilitation centers, 23 polytrauma network sites, numerous polytrauma support clinics, and > 110 TBI teams. Rachel McArdle, deputy executive director of rehabilitation and prosthetic services at VHA, told the subcommittee that since 2007, VHA has screened 1.8 million veterans for TBI. Every veteran, she said, receives an individualized plan addressing physical, cognitive, and emotional needs, often integrated with mental health services and patient-centered care approaches.
Graham and others expressed concern that despite their importance, the CoEs faced daunting challenges.
“Budgets have generally increased in recent years, but often unpredictably,” Graham noted. “Due to the recent focus on downsizing VHA staffing, a number of key positions are currently vacant due to clinical and administrative staff reassignment, resignation to accept positions outside VHA, or opting for early or standard retirement.”
In a statement, Natalia S. Rost, MD, MPH, President of the American Academy of Neurology, urged Congress to continue to provide funds for Neurology CoEs: “We look forward to continuing to work with Congress to secure robust, sustained funding to ensure our nation’s veterans receive the highest quality of neurologic care for years to come.”
Joel Scholten, MD, VA Executive Director of Physical Medicine and Rehabilitation, told the panel that the VA Office of Research and Development allocated $50 million for fiscal year 2025 research projects on TBI. Some are aimed at developing better biomarkers not only for TBI but also co-occurring mental health diagnoses. “As we work to better understand and better identify biomarkers not only for TBI but also looking at those associated or affiliated risk factors that can enhance suicide risk, we'll better be able to care for veterans.”
“I’m confident that the VA has all the data, legal authority, and funding it needs to effectively treat TBI,” Rep. Mariannette Miller-Meeks (R-IA), subcommittee chair, added. “Here's where I’ve seen the VA needs improvement: Consistent quality in patient care and data.”
Still, Graham argued that staffing reductions may be straining VHA’s ability to continue its mission. Anxiety about job security, increased vacancies, inadequate space in overcrowded VA medical centers due to the return to office mandate, and the loss of psychological safety and a positive workplace culture threatened the quality of neurology care at VHA.
“The VHA has long promoted the path to becoming a high reliability organization, with an obsessive attention to accuracy and avoidance of clinical errors, in a climate of psychological safety that encourages reporting of mistakes and ‘near misses’ in a concerted effort to prevent patient harm,” he argued. “Unfortunately, these principles appear to be in abeyance at present.”
On January 8, 2020, Iran fired 15 ballistic missiles at the Al-Asad Airbase, where Alan Johnson, an Army Lieutenant Colonel and Aeromedical Physician Assistant, was deployed.
“I have no memory of the first 3 missile impacts because the third missile impact knocked me unconscious,” Johnson said in a statement to a House Committee on Veterans’ Affairs subcommittee on Health in a March 5 hearing. “I woke up just in time to experience missiles 4, 5, and 6.”
March is Brain Injury Awareness month, highlighting how nearly 1 in 4 veterans has screened positive for probable traumatic brain injury (TBI). Veterans with TBI also have a higher risk of suicide: in 2023, the suicide rate for veterans with a recent TBI diagnosis was > 94% higher than for veterans without a TBI diagnosis.
“For many veterans, TBI is not a single episode of care; it is a chronic neurological condition requiring coordinated, longitudinal management,” Glenn D. Graham, MD, PhD, president of the Association of VA Neurology Service (AVANS) and former executive director of the US Department of Veterans Affairs (VA) Neurology Clinical Programs said in a statement. “TBI is neurologically complex and often intertwined with other conditions … Accurate diagnosis and effective treatment require subspecialty expertise in areas such as epilepsy, headache medicine, and neurodegenerative disease. The Centers of Excellence (CoE) ensure that this expertise is available across our national system.”
An estimated 25% of service members who have been hospitalized with TBI will develop long-term disability. Studies show direct links between TBI and the development of neurological disorders. Lt. Col. Johnson, for instance, has been diagnosed with posttraumatic stress disorder, cranial nerve damage, double vision, chronic insomnia, ringing in the ears, neck pain, balance problems, difficulty in word finding, and depression. After 37 years in emergency medicine, Johnson said, he had to “bench” himself due to the sequelae: “I can’t do what I love to do anymore.”
However, many service members may not be diagnosed correctly. Blast-related brain injuries may be delayed, subtle, and easily missed in combat environments. In research Johnson coauthored, > 20% of troops were diagnosed with mild TBIs 4 weeks after the attack. Moreover, he said, soldiers being screened may underreport their symptoms in order to return to duty.
Timely diagnosis is key, but so is consistent follow-up. Ranking Member Rep. Julia Brownley (D-CA) said, “TBI is not an illness that goes away with medicine … It is a long-term chronic condition for which many veterans need ongoing integrated and well-coordinated care.”
The Veterans Health Administration (VHA) has 5 polytrauma rehabilitation centers, 23 polytrauma network sites, numerous polytrauma support clinics, and > 110 TBI teams. Rachel McArdle, deputy executive director of rehabilitation and prosthetic services at VHA, told the subcommittee that since 2007, VHA has screened 1.8 million veterans for TBI. Every veteran, she said, receives an individualized plan addressing physical, cognitive, and emotional needs, often integrated with mental health services and patient-centered care approaches.
Graham and others expressed concern that despite their importance, the CoEs faced daunting challenges.
“Budgets have generally increased in recent years, but often unpredictably,” Graham noted. “Due to the recent focus on downsizing VHA staffing, a number of key positions are currently vacant due to clinical and administrative staff reassignment, resignation to accept positions outside VHA, or opting for early or standard retirement.”
In a statement, Natalia S. Rost, MD, MPH, President of the American Academy of Neurology, urged Congress to continue to provide funds for Neurology CoEs: “We look forward to continuing to work with Congress to secure robust, sustained funding to ensure our nation’s veterans receive the highest quality of neurologic care for years to come.”
Joel Scholten, MD, VA Executive Director of Physical Medicine and Rehabilitation, told the panel that the VA Office of Research and Development allocated $50 million for fiscal year 2025 research projects on TBI. Some are aimed at developing better biomarkers not only for TBI but also co-occurring mental health diagnoses. “As we work to better understand and better identify biomarkers not only for TBI but also looking at those associated or affiliated risk factors that can enhance suicide risk, we'll better be able to care for veterans.”
“I’m confident that the VA has all the data, legal authority, and funding it needs to effectively treat TBI,” Rep. Mariannette Miller-Meeks (R-IA), subcommittee chair, added. “Here's where I’ve seen the VA needs improvement: Consistent quality in patient care and data.”
Still, Graham argued that staffing reductions may be straining VHA’s ability to continue its mission. Anxiety about job security, increased vacancies, inadequate space in overcrowded VA medical centers due to the return to office mandate, and the loss of psychological safety and a positive workplace culture threatened the quality of neurology care at VHA.
“The VHA has long promoted the path to becoming a high reliability organization, with an obsessive attention to accuracy and avoidance of clinical errors, in a climate of psychological safety that encourages reporting of mistakes and ‘near misses’ in a concerted effort to prevent patient harm,” he argued. “Unfortunately, these principles appear to be in abeyance at present.”
On January 8, 2020, Iran fired 15 ballistic missiles at the Al-Asad Airbase, where Alan Johnson, an Army Lieutenant Colonel and Aeromedical Physician Assistant, was deployed.
“I have no memory of the first 3 missile impacts because the third missile impact knocked me unconscious,” Johnson said in a statement to a House Committee on Veterans’ Affairs subcommittee on Health in a March 5 hearing. “I woke up just in time to experience missiles 4, 5, and 6.”
March is Brain Injury Awareness month, highlighting how nearly 1 in 4 veterans has screened positive for probable traumatic brain injury (TBI). Veterans with TBI also have a higher risk of suicide: in 2023, the suicide rate for veterans with a recent TBI diagnosis was > 94% higher than for veterans without a TBI diagnosis.
“For many veterans, TBI is not a single episode of care; it is a chronic neurological condition requiring coordinated, longitudinal management,” Glenn D. Graham, MD, PhD, president of the Association of VA Neurology Service (AVANS) and former executive director of the US Department of Veterans Affairs (VA) Neurology Clinical Programs said in a statement. “TBI is neurologically complex and often intertwined with other conditions … Accurate diagnosis and effective treatment require subspecialty expertise in areas such as epilepsy, headache medicine, and neurodegenerative disease. The Centers of Excellence (CoE) ensure that this expertise is available across our national system.”
An estimated 25% of service members who have been hospitalized with TBI will develop long-term disability. Studies show direct links between TBI and the development of neurological disorders. Lt. Col. Johnson, for instance, has been diagnosed with posttraumatic stress disorder, cranial nerve damage, double vision, chronic insomnia, ringing in the ears, neck pain, balance problems, difficulty in word finding, and depression. After 37 years in emergency medicine, Johnson said, he had to “bench” himself due to the sequelae: “I can’t do what I love to do anymore.”
However, many service members may not be diagnosed correctly. Blast-related brain injuries may be delayed, subtle, and easily missed in combat environments. In research Johnson coauthored, > 20% of troops were diagnosed with mild TBIs 4 weeks after the attack. Moreover, he said, soldiers being screened may underreport their symptoms in order to return to duty.
Timely diagnosis is key, but so is consistent follow-up. Ranking Member Rep. Julia Brownley (D-CA) said, “TBI is not an illness that goes away with medicine … It is a long-term chronic condition for which many veterans need ongoing integrated and well-coordinated care.”
The Veterans Health Administration (VHA) has 5 polytrauma rehabilitation centers, 23 polytrauma network sites, numerous polytrauma support clinics, and > 110 TBI teams. Rachel McArdle, deputy executive director of rehabilitation and prosthetic services at VHA, told the subcommittee that since 2007, VHA has screened 1.8 million veterans for TBI. Every veteran, she said, receives an individualized plan addressing physical, cognitive, and emotional needs, often integrated with mental health services and patient-centered care approaches.
Graham and others expressed concern that despite their importance, the CoEs faced daunting challenges.
“Budgets have generally increased in recent years, but often unpredictably,” Graham noted. “Due to the recent focus on downsizing VHA staffing, a number of key positions are currently vacant due to clinical and administrative staff reassignment, resignation to accept positions outside VHA, or opting for early or standard retirement.”
In a statement, Natalia S. Rost, MD, MPH, President of the American Academy of Neurology, urged Congress to continue to provide funds for Neurology CoEs: “We look forward to continuing to work with Congress to secure robust, sustained funding to ensure our nation’s veterans receive the highest quality of neurologic care for years to come.”
Joel Scholten, MD, VA Executive Director of Physical Medicine and Rehabilitation, told the panel that the VA Office of Research and Development allocated $50 million for fiscal year 2025 research projects on TBI. Some are aimed at developing better biomarkers not only for TBI but also co-occurring mental health diagnoses. “As we work to better understand and better identify biomarkers not only for TBI but also looking at those associated or affiliated risk factors that can enhance suicide risk, we'll better be able to care for veterans.”
“I’m confident that the VA has all the data, legal authority, and funding it needs to effectively treat TBI,” Rep. Mariannette Miller-Meeks (R-IA), subcommittee chair, added. “Here's where I’ve seen the VA needs improvement: Consistent quality in patient care and data.”
Still, Graham argued that staffing reductions may be straining VHA’s ability to continue its mission. Anxiety about job security, increased vacancies, inadequate space in overcrowded VA medical centers due to the return to office mandate, and the loss of psychological safety and a positive workplace culture threatened the quality of neurology care at VHA.
“The VHA has long promoted the path to becoming a high reliability organization, with an obsessive attention to accuracy and avoidance of clinical errors, in a climate of psychological safety that encourages reporting of mistakes and ‘near misses’ in a concerted effort to prevent patient harm,” he argued. “Unfortunately, these principles appear to be in abeyance at present.”
Hearing Addresses Neurology CoE Challenges
Hearing Addresses Neurology CoE Challenges
Advanced CTE Associated With Dementia in Veterans Study
A study in veterans has found a link between dementia and severe chronic traumatic encephalopathy (CTE)—a degenerative brain disorder diagnosed after death that typically affects contact sports athletes and military personnel. Brain donors with advanced CTE (stage 4) were nearly 4.5 times more likely to have developed dementia than those without CTE. Individuals with stage 3 CTE had more than double the risk of dementia. The study was published in January in Alzheimer's and Dementia.
CTE stages 1 and 2 were not associated with dementia, cognitive impairment, or functional decline. Researchers also did not observe mood or behavioral symptoms at any stage of the disease. Researchers from the Boston University CTE Center and Veterans Affairs Boston Healthcare System (VABHS) led the study, which was funded by grants from the National Institutes of Health (NIH).
“This study proves that CTE is not a benign brain disease and that it has a significant impact on people’s lives,” coauthor Ann C. McKee, MD, chief of neuropathology at VABHS and director of the Boston University CTE Center, told Federal Practitioner.
McKee added that this research “provides evidence of a robust association between CTE and dementia, as well as cognitive symptoms, supporting our suspicions of CTE being a possible cause of dementia.”
Because CTE can only be diagnosed after death, researchers analyzed 614 donated brains from individuals with known exposure to repetitive head impacts. Among these donors, 366 (59.6%) had CTE and 248 (40.4%) did not. Most donors were male (97%), and most played American football (80.3%). Of the 614 donated brains, 20 (3.3%) were female. The average age of death from these 614 was 52 years, ranging from 13 to 98 years.
None of the donors had any of the 3 most common neurodegenerative causes of dementia: Alzheimer disease, dementia with Lewy bodies, or frontotemporal lobar degeneration.
Researchers also collected clinical information from individuals close to the donors. Typically, these are family members or close contacts through retrospective evaluations that combined online surveys, telephone interviews, and medical records.
Data collected included demographics; educational attainment; athletic history (including sport, level of play, position, age at first exposure, and duration); military history; traumatic brain injury history; substance use; and medical, social, and family histories.
CTE is often misdiagnosed as Alzheimer disease. In this study, among those diagnosed with dementia, 40% were informed they had Alzheimer, yet autopsy findings later showed no evidence of the disease. Another 38% were told the cause of dementia was unknown or could not be specified.
“In cases of dementia, when there is a history of repetitive head impacts from contact sports, military activities, or other exposures, CTE should be considered in the differential diagnosis,” McKee said. “Efforts should be made to distinguish CTE from Alzheimer disease and other causes of dementia during life.”
CTE shares features with Alzheimer, specifically the accumulation of abnormal tau protein. In healthy brains, tau helps maintain the stability and proper function of nerve cells. In CTE, however, tau accumulates in small clumps inside nerve cells that eventually form larger tangles.
Normally, the body clears excess tau protein, but in neurodegenerative diseases this process fails. The ensuing buildup damages brain cells, leading to cell death and the progressive symptoms of dementia.
Understanding how brain changes, including those related to CTE, relate to symptoms is of “paramount importance,” said Heather M. Snyder, PhD, senior vice president of medical and scientific relations at the Alzheimer’s Association in Chicago, who was not involved in the study.
Snyder described the research as “the first study to definitely demonstrate that brain changes caused by CTE are associated with the presence of dementia symptoms.” She also noted that the findings suggest a dose-response relationship, with more severe brain changes linked to worse cognitive symptoms.
The findings “open up new paths of research,” Snyder told Federal Practitioner, but also emphasized that improved tools are needed to detect these CTE-related brain changes in living individuals.
“While we have made significant progress in understanding the diseases that cause dementia, we have much to learn,” Snyder said. “Continued and steadfast investment in research remains a priority to improve early detection during life and develop personalized approaches.”
Ann McKee reported that she is a member of the Mackey-White Committee of the National Football League Players Association and received funding from the National Institutes of Health, US Department of Veteran Affairs, the Buoniconti Foundation and the MacParkman Foundation during the conduct of the study. She reports honorarium for speaking engagements.
Heather Snyder is a full-time employee of the Alzheimer’s Association, Chicago, IL and has a spouse who is employed by Abbott in an unrelated area. She has no financial conflicts to disclose.
A study in veterans has found a link between dementia and severe chronic traumatic encephalopathy (CTE)—a degenerative brain disorder diagnosed after death that typically affects contact sports athletes and military personnel. Brain donors with advanced CTE (stage 4) were nearly 4.5 times more likely to have developed dementia than those without CTE. Individuals with stage 3 CTE had more than double the risk of dementia. The study was published in January in Alzheimer's and Dementia.
CTE stages 1 and 2 were not associated with dementia, cognitive impairment, or functional decline. Researchers also did not observe mood or behavioral symptoms at any stage of the disease. Researchers from the Boston University CTE Center and Veterans Affairs Boston Healthcare System (VABHS) led the study, which was funded by grants from the National Institutes of Health (NIH).
“This study proves that CTE is not a benign brain disease and that it has a significant impact on people’s lives,” coauthor Ann C. McKee, MD, chief of neuropathology at VABHS and director of the Boston University CTE Center, told Federal Practitioner.
McKee added that this research “provides evidence of a robust association between CTE and dementia, as well as cognitive symptoms, supporting our suspicions of CTE being a possible cause of dementia.”
Because CTE can only be diagnosed after death, researchers analyzed 614 donated brains from individuals with known exposure to repetitive head impacts. Among these donors, 366 (59.6%) had CTE and 248 (40.4%) did not. Most donors were male (97%), and most played American football (80.3%). Of the 614 donated brains, 20 (3.3%) were female. The average age of death from these 614 was 52 years, ranging from 13 to 98 years.
None of the donors had any of the 3 most common neurodegenerative causes of dementia: Alzheimer disease, dementia with Lewy bodies, or frontotemporal lobar degeneration.
Researchers also collected clinical information from individuals close to the donors. Typically, these are family members or close contacts through retrospective evaluations that combined online surveys, telephone interviews, and medical records.
Data collected included demographics; educational attainment; athletic history (including sport, level of play, position, age at first exposure, and duration); military history; traumatic brain injury history; substance use; and medical, social, and family histories.
CTE is often misdiagnosed as Alzheimer disease. In this study, among those diagnosed with dementia, 40% were informed they had Alzheimer, yet autopsy findings later showed no evidence of the disease. Another 38% were told the cause of dementia was unknown or could not be specified.
“In cases of dementia, when there is a history of repetitive head impacts from contact sports, military activities, or other exposures, CTE should be considered in the differential diagnosis,” McKee said. “Efforts should be made to distinguish CTE from Alzheimer disease and other causes of dementia during life.”
CTE shares features with Alzheimer, specifically the accumulation of abnormal tau protein. In healthy brains, tau helps maintain the stability and proper function of nerve cells. In CTE, however, tau accumulates in small clumps inside nerve cells that eventually form larger tangles.
Normally, the body clears excess tau protein, but in neurodegenerative diseases this process fails. The ensuing buildup damages brain cells, leading to cell death and the progressive symptoms of dementia.
Understanding how brain changes, including those related to CTE, relate to symptoms is of “paramount importance,” said Heather M. Snyder, PhD, senior vice president of medical and scientific relations at the Alzheimer’s Association in Chicago, who was not involved in the study.
Snyder described the research as “the first study to definitely demonstrate that brain changes caused by CTE are associated with the presence of dementia symptoms.” She also noted that the findings suggest a dose-response relationship, with more severe brain changes linked to worse cognitive symptoms.
The findings “open up new paths of research,” Snyder told Federal Practitioner, but also emphasized that improved tools are needed to detect these CTE-related brain changes in living individuals.
“While we have made significant progress in understanding the diseases that cause dementia, we have much to learn,” Snyder said. “Continued and steadfast investment in research remains a priority to improve early detection during life and develop personalized approaches.”
Ann McKee reported that she is a member of the Mackey-White Committee of the National Football League Players Association and received funding from the National Institutes of Health, US Department of Veteran Affairs, the Buoniconti Foundation and the MacParkman Foundation during the conduct of the study. She reports honorarium for speaking engagements.
Heather Snyder is a full-time employee of the Alzheimer’s Association, Chicago, IL and has a spouse who is employed by Abbott in an unrelated area. She has no financial conflicts to disclose.
A study in veterans has found a link between dementia and severe chronic traumatic encephalopathy (CTE)—a degenerative brain disorder diagnosed after death that typically affects contact sports athletes and military personnel. Brain donors with advanced CTE (stage 4) were nearly 4.5 times more likely to have developed dementia than those without CTE. Individuals with stage 3 CTE had more than double the risk of dementia. The study was published in January in Alzheimer's and Dementia.
CTE stages 1 and 2 were not associated with dementia, cognitive impairment, or functional decline. Researchers also did not observe mood or behavioral symptoms at any stage of the disease. Researchers from the Boston University CTE Center and Veterans Affairs Boston Healthcare System (VABHS) led the study, which was funded by grants from the National Institutes of Health (NIH).
“This study proves that CTE is not a benign brain disease and that it has a significant impact on people’s lives,” coauthor Ann C. McKee, MD, chief of neuropathology at VABHS and director of the Boston University CTE Center, told Federal Practitioner.
McKee added that this research “provides evidence of a robust association between CTE and dementia, as well as cognitive symptoms, supporting our suspicions of CTE being a possible cause of dementia.”
Because CTE can only be diagnosed after death, researchers analyzed 614 donated brains from individuals with known exposure to repetitive head impacts. Among these donors, 366 (59.6%) had CTE and 248 (40.4%) did not. Most donors were male (97%), and most played American football (80.3%). Of the 614 donated brains, 20 (3.3%) were female. The average age of death from these 614 was 52 years, ranging from 13 to 98 years.
None of the donors had any of the 3 most common neurodegenerative causes of dementia: Alzheimer disease, dementia with Lewy bodies, or frontotemporal lobar degeneration.
Researchers also collected clinical information from individuals close to the donors. Typically, these are family members or close contacts through retrospective evaluations that combined online surveys, telephone interviews, and medical records.
Data collected included demographics; educational attainment; athletic history (including sport, level of play, position, age at first exposure, and duration); military history; traumatic brain injury history; substance use; and medical, social, and family histories.
CTE is often misdiagnosed as Alzheimer disease. In this study, among those diagnosed with dementia, 40% were informed they had Alzheimer, yet autopsy findings later showed no evidence of the disease. Another 38% were told the cause of dementia was unknown or could not be specified.
“In cases of dementia, when there is a history of repetitive head impacts from contact sports, military activities, or other exposures, CTE should be considered in the differential diagnosis,” McKee said. “Efforts should be made to distinguish CTE from Alzheimer disease and other causes of dementia during life.”
CTE shares features with Alzheimer, specifically the accumulation of abnormal tau protein. In healthy brains, tau helps maintain the stability and proper function of nerve cells. In CTE, however, tau accumulates in small clumps inside nerve cells that eventually form larger tangles.
Normally, the body clears excess tau protein, but in neurodegenerative diseases this process fails. The ensuing buildup damages brain cells, leading to cell death and the progressive symptoms of dementia.
Understanding how brain changes, including those related to CTE, relate to symptoms is of “paramount importance,” said Heather M. Snyder, PhD, senior vice president of medical and scientific relations at the Alzheimer’s Association in Chicago, who was not involved in the study.
Snyder described the research as “the first study to definitely demonstrate that brain changes caused by CTE are associated with the presence of dementia symptoms.” She also noted that the findings suggest a dose-response relationship, with more severe brain changes linked to worse cognitive symptoms.
The findings “open up new paths of research,” Snyder told Federal Practitioner, but also emphasized that improved tools are needed to detect these CTE-related brain changes in living individuals.
“While we have made significant progress in understanding the diseases that cause dementia, we have much to learn,” Snyder said. “Continued and steadfast investment in research remains a priority to improve early detection during life and develop personalized approaches.”
Ann McKee reported that she is a member of the Mackey-White Committee of the National Football League Players Association and received funding from the National Institutes of Health, US Department of Veteran Affairs, the Buoniconti Foundation and the MacParkman Foundation during the conduct of the study. She reports honorarium for speaking engagements.
Heather Snyder is a full-time employee of the Alzheimer’s Association, Chicago, IL and has a spouse who is employed by Abbott in an unrelated area. She has no financial conflicts to disclose.
Stereotactic Radiation Linked to Better Brain Mets Outcomes
Stereotactic Radiation Linked to Better Brain Mets Outcomes
TOPLINE:
In patients with 5-20 brain metastases, stereotactic radiation improved symptoms and reduced interference with daily functioning compared to hippocampal-avoidance whole brain radiation. The weighted composite MD Anderson Symptom Inventory-Brain Tumor score changed from 2.69 to 2.37 with stereotactic radiation compared with 2.29 to 3.03 with hippocampal-avoidance whole brain radiation.
METHODOLOGY:
- Randomized trials have shown stereotactic radiation preserves neurocognitive function and patient-reported outcomes compared with whole brain radiation in patients with four or less brain metastases. For patients with more than four brain metastases, published randomized comparisons of stereotactic radiation vs whole brain radiation were lacking prior to this study.
- Researchers conducted a phase 3, open-label, randomized clinical trial at four US-based centers, enrolling 196 patients between April 2017 and May 2024, with final follow-up in March 2025.
- Participants included patients with 5-20 brain metastases and no prior brain-directed radiation, with a median of 14 brain metastases per patient and 25% having undergone prior neurosurgical resection.
- The primary outcome was the mean weighted patient-reported symptom severity and interference score change over 6 months. The researchers used the MD Anderson Symptom Inventory-Brain Tumor instrument, with scores ranging from 0-10 and change range of -10 to 10, to measure outcomes.
- Stereotactic radiation was delivered in either 1 day (20 Gy) or five daily fractions (30 Gy, or 25 Gy for surgically removed tumors), while hippocampal-avoidance whole brain radiation was administered as 30 Gy in 10 daily fractions with memantine.
TAKEAWAY:
- Primary outcome analysis showed that stereotactic radiation was linked to a change in the weighted composite MD Anderson Symptom Inventory-Brain Tumor score of 2.69 to 2.37 (mean change, -0.32) compared with 2.29 to 3.03 (mean change, 0.74) with hippocampal-avoidance whole brain radiation (mean difference, -1.06; 95% CI, -1.54 to -0.58; P < .001).
- Functional independence via the Barthel Index was better in the stereotactic radiation group at 4 months (mean difference, 6.79; 95% CI, 1.19-12.38; P = .02) and 12 months (mean difference, 7.92; 95% CI, 1.34-14.49; P = .02).
- New brain metastases were more frequent with stereotactic radiation (1-year cumulative incidence, 45.4% vs 24.2%; P = .003), while local recurrence was lower (3.2% vs 39.5%; P < .001).
- Grade 3-5 adverse events occurred in 12% of stereotactic radiation patients vs 13% in the hippocampal-avoidance whole brain radiation group, with fatigue being most common (28% vs 44%).
IN PRACTICE:
“While [the trial] clearly demonstrates that patients with 5-20 brain metastases have improved symptom burden and lowered interference with daily functioning, there are questions that remain for stereotactic radiosurgery in this population. Patients receiving stereotactic radiosurgery for brain metastases have a higher need for future salvage procedures, and this rate of salvage procedures is higher for patients with an increased number of brain metastases at diagnosis… Moreover, it has been shown that the upfront decision between stereotactic radiosurgery and whole brain radiotherapy is the single decision that contributes most to the cost of care of a patient with brain metastases,” said Michael Chan, MD, in an accompanying editorial published in JAMA.
SOURCE:
The study was led by Ayal A. Aizer, MD, MHS, Brigham and Women’s Hospital/Dana-Farber Cancer Institute, Boston. It was published online on February 19 in JAMA.
LIMITATIONS:
According to the authors, the study was not blinded, and the primary outcome was subjective. High mortality limited long-term data collection, reducing precision and biasing outcomes toward survivors. Additionally, randomization was not stratified by treating center, allowing possible unmeasured imbalances. The minimal clinically important difference had not been defined for many study outcome measures.
DISCLOSURES:
The trial was supported by Varian, a Siemens Healthineers Company. Aizer disclosed receiving grants from NH TherAguix Research outside the submitted work. Additional disclosures are noted in the original article.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication.
A version of this article first appeared on Medscape.com.
TOPLINE:
In patients with 5-20 brain metastases, stereotactic radiation improved symptoms and reduced interference with daily functioning compared to hippocampal-avoidance whole brain radiation. The weighted composite MD Anderson Symptom Inventory-Brain Tumor score changed from 2.69 to 2.37 with stereotactic radiation compared with 2.29 to 3.03 with hippocampal-avoidance whole brain radiation.
METHODOLOGY:
- Randomized trials have shown stereotactic radiation preserves neurocognitive function and patient-reported outcomes compared with whole brain radiation in patients with four or less brain metastases. For patients with more than four brain metastases, published randomized comparisons of stereotactic radiation vs whole brain radiation were lacking prior to this study.
- Researchers conducted a phase 3, open-label, randomized clinical trial at four US-based centers, enrolling 196 patients between April 2017 and May 2024, with final follow-up in March 2025.
- Participants included patients with 5-20 brain metastases and no prior brain-directed radiation, with a median of 14 brain metastases per patient and 25% having undergone prior neurosurgical resection.
- The primary outcome was the mean weighted patient-reported symptom severity and interference score change over 6 months. The researchers used the MD Anderson Symptom Inventory-Brain Tumor instrument, with scores ranging from 0-10 and change range of -10 to 10, to measure outcomes.
- Stereotactic radiation was delivered in either 1 day (20 Gy) or five daily fractions (30 Gy, or 25 Gy for surgically removed tumors), while hippocampal-avoidance whole brain radiation was administered as 30 Gy in 10 daily fractions with memantine.
TAKEAWAY:
- Primary outcome analysis showed that stereotactic radiation was linked to a change in the weighted composite MD Anderson Symptom Inventory-Brain Tumor score of 2.69 to 2.37 (mean change, -0.32) compared with 2.29 to 3.03 (mean change, 0.74) with hippocampal-avoidance whole brain radiation (mean difference, -1.06; 95% CI, -1.54 to -0.58; P < .001).
- Functional independence via the Barthel Index was better in the stereotactic radiation group at 4 months (mean difference, 6.79; 95% CI, 1.19-12.38; P = .02) and 12 months (mean difference, 7.92; 95% CI, 1.34-14.49; P = .02).
- New brain metastases were more frequent with stereotactic radiation (1-year cumulative incidence, 45.4% vs 24.2%; P = .003), while local recurrence was lower (3.2% vs 39.5%; P < .001).
- Grade 3-5 adverse events occurred in 12% of stereotactic radiation patients vs 13% in the hippocampal-avoidance whole brain radiation group, with fatigue being most common (28% vs 44%).
IN PRACTICE:
“While [the trial] clearly demonstrates that patients with 5-20 brain metastases have improved symptom burden and lowered interference with daily functioning, there are questions that remain for stereotactic radiosurgery in this population. Patients receiving stereotactic radiosurgery for brain metastases have a higher need for future salvage procedures, and this rate of salvage procedures is higher for patients with an increased number of brain metastases at diagnosis… Moreover, it has been shown that the upfront decision between stereotactic radiosurgery and whole brain radiotherapy is the single decision that contributes most to the cost of care of a patient with brain metastases,” said Michael Chan, MD, in an accompanying editorial published in JAMA.
SOURCE:
The study was led by Ayal A. Aizer, MD, MHS, Brigham and Women’s Hospital/Dana-Farber Cancer Institute, Boston. It was published online on February 19 in JAMA.
LIMITATIONS:
According to the authors, the study was not blinded, and the primary outcome was subjective. High mortality limited long-term data collection, reducing precision and biasing outcomes toward survivors. Additionally, randomization was not stratified by treating center, allowing possible unmeasured imbalances. The minimal clinically important difference had not been defined for many study outcome measures.
DISCLOSURES:
The trial was supported by Varian, a Siemens Healthineers Company. Aizer disclosed receiving grants from NH TherAguix Research outside the submitted work. Additional disclosures are noted in the original article.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication.
A version of this article first appeared on Medscape.com.
TOPLINE:
In patients with 5-20 brain metastases, stereotactic radiation improved symptoms and reduced interference with daily functioning compared to hippocampal-avoidance whole brain radiation. The weighted composite MD Anderson Symptom Inventory-Brain Tumor score changed from 2.69 to 2.37 with stereotactic radiation compared with 2.29 to 3.03 with hippocampal-avoidance whole brain radiation.
METHODOLOGY:
- Randomized trials have shown stereotactic radiation preserves neurocognitive function and patient-reported outcomes compared with whole brain radiation in patients with four or less brain metastases. For patients with more than four brain metastases, published randomized comparisons of stereotactic radiation vs whole brain radiation were lacking prior to this study.
- Researchers conducted a phase 3, open-label, randomized clinical trial at four US-based centers, enrolling 196 patients between April 2017 and May 2024, with final follow-up in March 2025.
- Participants included patients with 5-20 brain metastases and no prior brain-directed radiation, with a median of 14 brain metastases per patient and 25% having undergone prior neurosurgical resection.
- The primary outcome was the mean weighted patient-reported symptom severity and interference score change over 6 months. The researchers used the MD Anderson Symptom Inventory-Brain Tumor instrument, with scores ranging from 0-10 and change range of -10 to 10, to measure outcomes.
- Stereotactic radiation was delivered in either 1 day (20 Gy) or five daily fractions (30 Gy, or 25 Gy for surgically removed tumors), while hippocampal-avoidance whole brain radiation was administered as 30 Gy in 10 daily fractions with memantine.
TAKEAWAY:
- Primary outcome analysis showed that stereotactic radiation was linked to a change in the weighted composite MD Anderson Symptom Inventory-Brain Tumor score of 2.69 to 2.37 (mean change, -0.32) compared with 2.29 to 3.03 (mean change, 0.74) with hippocampal-avoidance whole brain radiation (mean difference, -1.06; 95% CI, -1.54 to -0.58; P < .001).
- Functional independence via the Barthel Index was better in the stereotactic radiation group at 4 months (mean difference, 6.79; 95% CI, 1.19-12.38; P = .02) and 12 months (mean difference, 7.92; 95% CI, 1.34-14.49; P = .02).
- New brain metastases were more frequent with stereotactic radiation (1-year cumulative incidence, 45.4% vs 24.2%; P = .003), while local recurrence was lower (3.2% vs 39.5%; P < .001).
- Grade 3-5 adverse events occurred in 12% of stereotactic radiation patients vs 13% in the hippocampal-avoidance whole brain radiation group, with fatigue being most common (28% vs 44%).
IN PRACTICE:
“While [the trial] clearly demonstrates that patients with 5-20 brain metastases have improved symptom burden and lowered interference with daily functioning, there are questions that remain for stereotactic radiosurgery in this population. Patients receiving stereotactic radiosurgery for brain metastases have a higher need for future salvage procedures, and this rate of salvage procedures is higher for patients with an increased number of brain metastases at diagnosis… Moreover, it has been shown that the upfront decision between stereotactic radiosurgery and whole brain radiotherapy is the single decision that contributes most to the cost of care of a patient with brain metastases,” said Michael Chan, MD, in an accompanying editorial published in JAMA.
SOURCE:
The study was led by Ayal A. Aizer, MD, MHS, Brigham and Women’s Hospital/Dana-Farber Cancer Institute, Boston. It was published online on February 19 in JAMA.
LIMITATIONS:
According to the authors, the study was not blinded, and the primary outcome was subjective. High mortality limited long-term data collection, reducing precision and biasing outcomes toward survivors. Additionally, randomization was not stratified by treating center, allowing possible unmeasured imbalances. The minimal clinically important difference had not been defined for many study outcome measures.
DISCLOSURES:
The trial was supported by Varian, a Siemens Healthineers Company. Aizer disclosed receiving grants from NH TherAguix Research outside the submitted work. Additional disclosures are noted in the original article.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication.
A version of this article first appeared on Medscape.com.
Stereotactic Radiation Linked to Better Brain Mets Outcomes
Stereotactic Radiation Linked to Better Brain Mets Outcomes
Development and Validation of an Administrative Algorithm to Identify Veterans With Epilepsy
Development and Validation of an Administrative Algorithm to Identify Veterans With Epilepsy
Epilepsy affects about 4.5 million people in the United States and 150,000 new individuals are diagnosed each year.1,2 In 2019, epilepsy-attributable health care spending for noninstitutionalized people was around $5.4 billion and total epilepsy-attributable and epilepsy or seizure health care-related costs totaled $54 billion.3
Accurate surveillance of epilepsy in large health care systems can potentially improve health care delivery and resource allocation. A 2012 Institute of Medicine (IOM) report identified 13 recommendations to guide public health action on epilepsy, including validation of standard definitions for case ascertainment, identification of epilepsy through screening programs or protocols, and expansion of surveillance to better understand disease burden.4
A systematic review of validation studies concluded that it is reasonable to use administrative data to identify people with epilepsy in epidemiologic research. Combining The International Classification of Diseases (ICD) codes for epilepsy (ICD-10, G40-41; ICD-9, 345) with antiseizure medications (ASMs) could provide high positive predictive values (PPVs) and combining symptoms codes for convulsions (ICD-10, R56; ICD-9, 780.3, 780.39) with ASMs could lead to high sensitivity.5 However, identifying individuals with epilepsy from administrative data in large managed health care organizations is challenging.6 The IOM report noted that large managed health care organizations presented varying incidence and prevalence estimates due to differing methodology, geographic area, demographics, and definitions of epilepsy.
The Veterans Health Administration (VHA) is the largest integrated US health care system, providing care to > 9.1 million veterans.7 To improve the health and well-being of veterans with epilepsy (VWEs), a network of sites was established in 2008 called the US Department of Veterans Affairs (VA) Epilepsy Centers of Excellence (ECoE). Subsequent to the creation of the ECoE, efforts were made to identify VWEs within VHA databases.8,9 Prior to fiscal year (FY) 2016, the ECoE adopted a modified version of a well-established epilepsy diagnostic algorithm developed by Holden et al for large managed care organizations.10 The original algorithm identified patients by cross-matching ASMs with ICD-9 codes for an index year. But it failed to capture a considerable number of stable patients with epilepsy in the VHA due to incomplete documentation, and had false positives due to inclusion of patients identified from diagnostic clinics. The modified algorithm the ECoE used prior to FY 2016 considered additional prior years and excluded encounters from diagnostic clinics. The result was an improvement in the sensitivity and specificity of the algorithm. Researchers evaluating 500 patients with epilepsy estimated that the modified algorithm had a PPV of 82.0% (95% CI, 78.6%-85.4%).11
After implementation of ICD-10 codes in the VHA in FY 2016, the task of reliably and efficiently identifying VWE led to a 3-tier algorithm. This article presents a validation of the different tiers of this algorithm after the implementation of ICD-10 diagnosis codes and summarizes the surveillance data collected over the years within the VHA showing the trends of epilepsy.
Methods
The VHA National Neurology office commissioned a Neurology Cube dashboard in FY 2021 in collaboration with VHA Support Service Center (VSSC) for reporting and surveillance of VWEs as a quality improvement initiative. The Neurology Cube uses a 3-tier system for identifying VWE in the VHA databases. VSSC programmers extract data from the VHA Corporate Data Warehouse (CDW) and utilize Microsoft SQL Server and Microsoft Power BI for Neurology Cube reports. The 3-tier system identifies VWE and divides them into distinct groups. The first tier identifies VWE with the highest degree of confidence; Tiers 2 and 3 represent identification with successively lesser degrees of confidence (Figure 1).
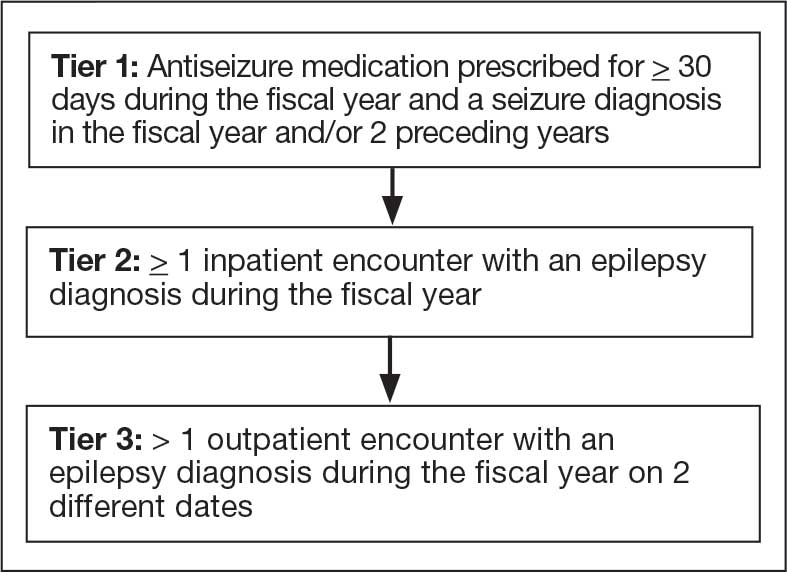
Tier 1
Definition. For a given index year and the preceding 2 years, any of following diagnosis codes on ≥ 1 clinical encounter are considered: 345.xx (epilepsy in ICD-9), 780.3x (other convulsions in ICD-9), G40.xxx (epilepsy in ICD-10), R40.4 (transient alteration of awareness), R56.1 (posttraumatic seizures), or R56.9 (unspecified convulsions). To reduce false positive rates, EEG clinic visits, which may include long-term monitoring, are excluded. Patients identified with ICD codes are then evaluated for an ASM prescription for ≥ 30 days during the index year. ASMs are listed in Appendix 1.
Validation. The development and validation of ICD-9 diagnosis codes crossmatched with an ASM prescription in the VHA has been published elsewhere.11 In FY 2017, after implementation of ICD-10 diagnostic codes, Tier 1 development and validation was performed in 2 phases. Even though Tier 1 study phases were conducted and completed during FY 2017, the patients for Tier 1 were identified from evaluation of FY 2016 data (October 1, 2015, to September 30, 2016). After the pilot analysis, the Tier 1 definition was implemented, and a chart review of 625 randomized patients was conducted at 5 sites for validation. Adequate preliminary data was not available to perform a sample size estimation for this study. Therefore, a practical target of 125 patients was set for Tier 1 from each site to obtain a final sample size of 625 patients. This second phase validated that the crossmatch of ICD-10 diagnosis codes with ASMs had a high PPV for identifying VWE.
Tiers 2 and 3
Definitions. For an index year, Tier 2 includes patients with ≥ 1 inpatient encounter documentation of either ICD-9 345.xx or ICD-10 G40.xxx, excluding EEG clinics. Tier 3 Includes patients who have had ≥ 2 outpatient encounters with diagnosis codes 345.xx or G40.xxx on 2 separate days, excluding EEG clinics. Tiers 2 and 3 do not require ASM prescriptions; this helps to identify VWEs who may be getting their medications outside of VHA or those who have received a new diagnosis.
Validations. Tiers 2 and 3 were included in the epilepsy identification algorithm in FY 2021 after validation was performed on a sample of 8 patients in each tier. Five patients were subsequently identified as having epilepsy in Tier 2 and 6 patients were identified in Tier 3. A more comprehensive validation of Tiers 2 and 3 was performed during FY 2022 that included patients at 5 sites seen during FY 2019 to FY 2022. Since yearly trends showed only about 8% of total patients were identified as having epilepsy through Tiers 2 and 3 we sought ≥ 20 patients per tier for the 5 sites for a total of 200 patients to ensure representation across the VHA. The final count was 126 patients for Tier 2 and 174 patients for Tier 3 (n = 300).
Gold Standard Criteria for Epilepsy Diagnosis
We used the International League Against Epilepsy (ILAE) definition of epilepsy for the validation of the 3 algorithm tiers. ILAE defines epilepsy as ≥ 2 unprovoked (or reflex) seizures occurring > 24 hours apart or 1 unprovoked (or reflex) seizure and a probability of further seizures similar to the general recurrence risk (≥ 60%) after 2 unprovoked seizures, occurring over the next 10 years.12
A standard protocol was provided to evaluators to identify patients using the VHA Computerized Patient Record System (Appendix 1). After review, evaluators categorized each patient in 1 of 4 ways: (1) Yes, definite: The patient’s health care practitioner (HCP) believes the patient has epilepsy and is treating with medication; (2) Yes, uncertain: The HCP has enough suspicion of epilepsy that a medication is prescribed, but uncertainty is expressed of the diagnosis; (3) No, definite: The HCP does not believe the patient has epilepsy and is therefore not treating with medication for seizure; (4) No, uncertain: The HCP is not treating with medication for epilepsy, because the diagnostic suspicion is not high enough, but there is suspicion for epilepsy.
As a quality improvement operational project, the Epilepsy National Program Office approved this validation project and determined that institutional review board approval was not required.
Statistical Analysis
Counts and percentages were computed for categories of epilepsy status. PPV of each tier was estimated with asymptotic 95% CIs.
Results
ICD-10 codes for 480 patients were evaluated in Tier 1 phase 1; 13.8% were documented with G40.xxx, 27.9% with R56.1, 34.4% with R56.9, and 24.0% with R40.4 (Appendix 2). In total, 68.1% fulfilled the criteria of epilepsy, 19.2% did not, and 12.7% were uncertain). From the validation of Tier 1 phase 2 (n = 625), the PPV of the algorithm for patients presumed to have epilepsy (definite and uncertain) was 85.1% (95% CI, 82.1%-87.8%) (Table).
Of 300 patients evaluated, 126 (42.0%) were evaluated for Tier 2 with a PPV of 61.9% (95% CI, 53.4%-70.4%), and 174 (58.0%) patients were evaluated for Tier 3 with a PPV of 59.8% (95% CI, 52.5%-67.1%. The PPV of the algorithm for patients presumed to have epilepsy (definite and uncertain) were combined to calculate the PPV. Estimates of VHA VWE counts were computed for each tier from FY 2014 to FY 2023 using the VSSC Neurology Cube (Figure 2). For all years, > 92% patients were classified using the Tier 1 definition.
Discussion
The development and validation of the 3-tier diagnostic algorithm represents an important advancement in the surveillance and management of epilepsy among veterans within the VHA. The validation of this algorithm also demonstrates its practical utility in a large, integrated health care system.
Specific challenges were encountered when attempting to use pre-existing algorithms; these challenges included differences in the usage patterns of diagnostic codes and the patterns of ASM use within the VHA. These challenges prompted the need for a tailored approach, which led to the development of this algorithm. The inclusion of additional ICD-10 codes led to further revisions and subsequent validation. While many of the basic concepts of the algorithm, including ICD codes and ASMs, could work in other institutions, it would be wise for health care organizations to develop their own algorithms because of certain variables, including organizational size, patient demographics, common comorbidities, and the specific configurations of electronic health records and administrative data systems.
Studies have shown that ICD-10 codes for epilepsy (G40.* and/or R56.9) perform well in identifying epilepsy whether they are assigned by neurologists (sensitivity, 97.7%; specificity, 44.1%; PPV, 96.2%; negative predictive value, 57.7%), or in emergency department or hospital discharges (PPV, 75.5%).13,14 The pilot study of the algorithm’s Tier 1 development (phase 1) evaluated whether the selected ICD-10 diagnostic codes accurately included the VWE population within the VHA and revealed that while most codes (eg, epilepsy [G40.xxx]; posttraumatic seizures [R56.1]; and unspecified convulsions [R56.9]), had a low false positive rate (< 16%), the R40.4 code (transient alteration of awareness) had a higher false positivity of 42%. While this is not surprising given the broad spectrum of conditions that can manifest as transient alteration of awareness, it underscores the inherent challenges in diagnosing epilepsy using diagnosis codes.
In phase 2, the Tier 1 algorithm was validated as effective for identifying VWE in the VHA system, as its PPV was determined to be high (85%). In comparison, Tiers 2 and 3, whose criteria did not require data on VHA prescribed ASM use, had lower tiers of epilepsy predictability (PPV about 60% for both). This was thought to be acceptable because Tiers 2 and 3 represent a smaller population of the identified VWEs (about 8%). These VWEs may otherwise have been missed, partly because veterans are not required to get ASMs from the VHA.
Upon VHA implementation in FY 2021, this diagnostic algorithm exhibited significant clinical utility when integrated within the VSSC Neurology Cube. It facilitated an efficient approach to identifying VWEs using readily available databases. This led to better tracking of real-time epilepsy cases, which facilitated improving current resource allocation and targeted intervention strategies such as identification of drug-resistant epilepsy patients, optimizing strategies for telehealth and patient outreach for awareness of epilepsy care resources within VHA. Meanwhile, data acquired by the algorithm over the decade since its development (FY 2014 to FY 2023) contributed to more accurate epidemiologic information and identification of historic trends. Development of the algorithm represents one of the ways ECoEs have led to improved care for VWEs. ECoEs have been shown to improve health care for veterans in several metrics.15
A strength of this study is the rigorous multitiered validation process to confirm the diagnostic accuracy of ICD-10 codes against the gold standard ILAE definition of epilepsy to identify “definite” epilepsy cases within the VHA. The use of specific ICD codes further enhances the precision of epilepsy diagnoses. The inclusion of ASMs, which are sometimes prescribed for conditions other than epilepsy, could potentially inflate false positive rates.16
This study focused exclusively on the identification and validation of definite epilepsy cases within the VHA VSSC database, employing more stringent diagnostic criteria to ensure the highest level of certainty in ascertaining epilepsy. It is important to note there is a separate category of probable epilepsy, which involves a broader set of diagnostic criteria. While not covered in this study, probable epilepsy would be subject to future research and validation, which could provide insights into a wider spectrum of epilepsy diagnoses. Such future research could help refine the algorithm’s applicability and accuracy and potentially lead to more comprehensive surveillance and management strategies in clinical practice.
This study highlights the inherent challenges in leveraging administrative data for disease identification, particularly for conditions such as epilepsy, where diagnostic clarity can be complex. However, other conditions such as multiple sclerosis have noted similar success with the use of VHA administrative data for categorizing disease.17
Limitations
The algorithm discussed in this article is, in and of itself, generalizable. However, the validation process was unique to the VHA patient population, limiting the generalizability of the findings. Documentation practices and HCP attitudes within the VHA may differ from those in other health care settings. Identifying people with epilepsy can be challenging because of changing definitions of epilepsy over time. In addition to clinical evaluation, EEG and magnetic resonance imaging results, response to ASM treatment, and video-EEG monitoring of habitual events all can help establish the diagnosis. Therefore, studies may vary in how inclusive or exclusive the criteria are. ASMs such as gabapentin, pregabalin, carbamazepine, lamotrigine, topiramate, and valproate are used to treat other conditions, including headaches, generalized pain, and mood disorders. Consequently, including these ASMs in the Tier 1 definition may have increased the false positive rate. Additional research is needed to evaluate whether excluding these ASMs from the algorithm based on specific criteria (eg, dose of ASM used) can further refine the algorithm to identify patients with epilepsy.
Further refinement of this algorithm may also occur as technology changes. Future electronic health records may allow better tracking of different epilepsy factors, the integration of additional diagnostic criteria, and the use of natural language processing or other forms of artificial intelligence.
Conclusions
This study presents a significant step forward in epilepsy surveillance within the VHA. The algorithm offers a robust tool for identifying VWEs with good PPVs, facilitating better resource allocation and targeted care. Despite its limitations, this research lays a foundation for future advancements in the management and understanding of epilepsy within large health care systems. Since this VHA algorithm is based on ASMs and ICD diagnosis codes from patient records, other large managed health care systems also may be able to adapt this algorithm to their data specifications.

- Kobau R, Luncheon C, Greenlund K. Active epilepsy prevalence among U.S. adults is 1.1% and differs by educational level-National Health Interview Survey, United States, 2021. Epilepsy Behav. 2023;142:109180. doi:10.1016/j.yebeh.2023.109180
- GBD 2017 US Neurological Disorders Collaborators, Feigin VL, Vos T, et al. Burden of neurological disorders across the US from 1990-2017: a global burden of disease study. JAMA Neurol. 2021;78:165-176. doi:10.1001/jamaneurol.2020.4152
- Moura LMVR, Karakis I, Zack MM, et al. Drivers of US health care spending for persons with seizures and/or epilepsies, 2010-2018. Epilepsia. 2022;63:2144-2154. doi:10.1111/epi.17305
- Institute of Medicine. Epilepsy Across the Spectrum: Promoting Health and Understanding. The National Academies Press; 2012. Accessed November 11, 2025. www.nap.edu/catalog/13379
- Mbizvo GK, Bennett KH, Schnier C, Simpson CR, Duncan SE, Chin RFM. The accuracy of using administrative healthcare data to identify epilepsy cases: A systematic review of validation studies. Epilepsia. 2020;61:1319-1335. doi:10.1111/epi.16547
- Montouris GD. How will primary care physicians, specialists, and managed care treat epilepsy in the new millennium? Neurology. 2000;55:S42-S44.
- US Department of Veterans Affairs. Veterans Health Administration: About VHA. Accessed November 11, 2025. https://www.va.gov/health/aboutvha.asp
- Veterans’ Mental Health and Other Care Improvements Act of 2008, S 2162, 110th Cong (2008). Accessed November 11, 2025. https://www.congress.gov/bill/110th-congress/senate-bill/2162
- Rehman R, Kelly PR, Husain AM, Tran TT. Characteristics of Veterans diagnosed with seizures within Veterans Health Administration. J Rehabil Res Dev. 2015;52(7):751-762. doi:10.1682/JRRD.2014.10.0241
- Holden EW, Grossman E, Nguyen HT, et al. Developing a computer algorithm to identify epilepsy cases in managed care organizations. Dis Manag. 2005;8:1-14. doi:10.1089/dis.2005.8.1
- Rehman R, Everhart A, Frontera AT, et al. Implementation of an established algorithm and modifications for the identification of epilepsy patients in the Veterans Health Administration. Epilepsy Res. 2016;127:284-290. doi:10.1016/j.eplepsyres.2016.09.012
- Fisher RS, Acevedo C, Arzimanoglou A, et al. ILAE official report: a practical clinical definition of epilepsy. Epilepsia. 2014;55:475-482. doi:10.1111/epi.12550
- Smith JR, Jones FJS, Fureman BE, et al. Accuracy of ICD-10-CM claims-based definitions for epilepsy and seizure type. Epilepsy Res. 2020;166:106414. doi:10.1016/j.eplepsyres.2020.106414
- Jetté N, Reid AY, Quan H, et al. How accurate is ICD coding for epilepsy? Epilepsia. 2010;51:62-69. doi:10.1111/j.1528-1167.2009.02201.x
- Kelly P, Chinta R, Privitera G. Do centers of excellence reduce health care costs? Evidence from the US Veterans Health Administration Centers for Epilepsy. Glob Bus Organ Excell. 2015;34:18-29.
- Haneef Z, Rehman R, Husain AM. Association between standardized mortality ratio and utilization of care in US veterans with drug-resistant epilepsy compared with all US veterans and the US general population. JAMA Neurol. 2022;79:879-887. doi:10.1001/jamaneurol.2022.2290
- Culpepper WJ, Marrie RA, Langer-Gould A, et al. Validation of an algorithm for identifying MS cases in administrative health claims datasets. Neurology. 2019;92:e1016-e1028 doi:10.1212/WNL.0000000000007043
Epilepsy affects about 4.5 million people in the United States and 150,000 new individuals are diagnosed each year.1,2 In 2019, epilepsy-attributable health care spending for noninstitutionalized people was around $5.4 billion and total epilepsy-attributable and epilepsy or seizure health care-related costs totaled $54 billion.3
Accurate surveillance of epilepsy in large health care systems can potentially improve health care delivery and resource allocation. A 2012 Institute of Medicine (IOM) report identified 13 recommendations to guide public health action on epilepsy, including validation of standard definitions for case ascertainment, identification of epilepsy through screening programs or protocols, and expansion of surveillance to better understand disease burden.4
A systematic review of validation studies concluded that it is reasonable to use administrative data to identify people with epilepsy in epidemiologic research. Combining The International Classification of Diseases (ICD) codes for epilepsy (ICD-10, G40-41; ICD-9, 345) with antiseizure medications (ASMs) could provide high positive predictive values (PPVs) and combining symptoms codes for convulsions (ICD-10, R56; ICD-9, 780.3, 780.39) with ASMs could lead to high sensitivity.5 However, identifying individuals with epilepsy from administrative data in large managed health care organizations is challenging.6 The IOM report noted that large managed health care organizations presented varying incidence and prevalence estimates due to differing methodology, geographic area, demographics, and definitions of epilepsy.
The Veterans Health Administration (VHA) is the largest integrated US health care system, providing care to > 9.1 million veterans.7 To improve the health and well-being of veterans with epilepsy (VWEs), a network of sites was established in 2008 called the US Department of Veterans Affairs (VA) Epilepsy Centers of Excellence (ECoE). Subsequent to the creation of the ECoE, efforts were made to identify VWEs within VHA databases.8,9 Prior to fiscal year (FY) 2016, the ECoE adopted a modified version of a well-established epilepsy diagnostic algorithm developed by Holden et al for large managed care organizations.10 The original algorithm identified patients by cross-matching ASMs with ICD-9 codes for an index year. But it failed to capture a considerable number of stable patients with epilepsy in the VHA due to incomplete documentation, and had false positives due to inclusion of patients identified from diagnostic clinics. The modified algorithm the ECoE used prior to FY 2016 considered additional prior years and excluded encounters from diagnostic clinics. The result was an improvement in the sensitivity and specificity of the algorithm. Researchers evaluating 500 patients with epilepsy estimated that the modified algorithm had a PPV of 82.0% (95% CI, 78.6%-85.4%).11
After implementation of ICD-10 codes in the VHA in FY 2016, the task of reliably and efficiently identifying VWE led to a 3-tier algorithm. This article presents a validation of the different tiers of this algorithm after the implementation of ICD-10 diagnosis codes and summarizes the surveillance data collected over the years within the VHA showing the trends of epilepsy.
Methods
The VHA National Neurology office commissioned a Neurology Cube dashboard in FY 2021 in collaboration with VHA Support Service Center (VSSC) for reporting and surveillance of VWEs as a quality improvement initiative. The Neurology Cube uses a 3-tier system for identifying VWE in the VHA databases. VSSC programmers extract data from the VHA Corporate Data Warehouse (CDW) and utilize Microsoft SQL Server and Microsoft Power BI for Neurology Cube reports. The 3-tier system identifies VWE and divides them into distinct groups. The first tier identifies VWE with the highest degree of confidence; Tiers 2 and 3 represent identification with successively lesser degrees of confidence (Figure 1).

Tier 1
Definition. For a given index year and the preceding 2 years, any of following diagnosis codes on ≥ 1 clinical encounter are considered: 345.xx (epilepsy in ICD-9), 780.3x (other convulsions in ICD-9), G40.xxx (epilepsy in ICD-10), R40.4 (transient alteration of awareness), R56.1 (posttraumatic seizures), or R56.9 (unspecified convulsions). To reduce false positive rates, EEG clinic visits, which may include long-term monitoring, are excluded. Patients identified with ICD codes are then evaluated for an ASM prescription for ≥ 30 days during the index year. ASMs are listed in Appendix 1.
Validation. The development and validation of ICD-9 diagnosis codes crossmatched with an ASM prescription in the VHA has been published elsewhere.11 In FY 2017, after implementation of ICD-10 diagnostic codes, Tier 1 development and validation was performed in 2 phases. Even though Tier 1 study phases were conducted and completed during FY 2017, the patients for Tier 1 were identified from evaluation of FY 2016 data (October 1, 2015, to September 30, 2016). After the pilot analysis, the Tier 1 definition was implemented, and a chart review of 625 randomized patients was conducted at 5 sites for validation. Adequate preliminary data was not available to perform a sample size estimation for this study. Therefore, a practical target of 125 patients was set for Tier 1 from each site to obtain a final sample size of 625 patients. This second phase validated that the crossmatch of ICD-10 diagnosis codes with ASMs had a high PPV for identifying VWE.
Tiers 2 and 3
Definitions. For an index year, Tier 2 includes patients with ≥ 1 inpatient encounter documentation of either ICD-9 345.xx or ICD-10 G40.xxx, excluding EEG clinics. Tier 3 Includes patients who have had ≥ 2 outpatient encounters with diagnosis codes 345.xx or G40.xxx on 2 separate days, excluding EEG clinics. Tiers 2 and 3 do not require ASM prescriptions; this helps to identify VWEs who may be getting their medications outside of VHA or those who have received a new diagnosis.
Validations. Tiers 2 and 3 were included in the epilepsy identification algorithm in FY 2021 after validation was performed on a sample of 8 patients in each tier. Five patients were subsequently identified as having epilepsy in Tier 2 and 6 patients were identified in Tier 3. A more comprehensive validation of Tiers 2 and 3 was performed during FY 2022 that included patients at 5 sites seen during FY 2019 to FY 2022. Since yearly trends showed only about 8% of total patients were identified as having epilepsy through Tiers 2 and 3 we sought ≥ 20 patients per tier for the 5 sites for a total of 200 patients to ensure representation across the VHA. The final count was 126 patients for Tier 2 and 174 patients for Tier 3 (n = 300).
Gold Standard Criteria for Epilepsy Diagnosis
We used the International League Against Epilepsy (ILAE) definition of epilepsy for the validation of the 3 algorithm tiers. ILAE defines epilepsy as ≥ 2 unprovoked (or reflex) seizures occurring > 24 hours apart or 1 unprovoked (or reflex) seizure and a probability of further seizures similar to the general recurrence risk (≥ 60%) after 2 unprovoked seizures, occurring over the next 10 years.12
A standard protocol was provided to evaluators to identify patients using the VHA Computerized Patient Record System (Appendix 1). After review, evaluators categorized each patient in 1 of 4 ways: (1) Yes, definite: The patient’s health care practitioner (HCP) believes the patient has epilepsy and is treating with medication; (2) Yes, uncertain: The HCP has enough suspicion of epilepsy that a medication is prescribed, but uncertainty is expressed of the diagnosis; (3) No, definite: The HCP does not believe the patient has epilepsy and is therefore not treating with medication for seizure; (4) No, uncertain: The HCP is not treating with medication for epilepsy, because the diagnostic suspicion is not high enough, but there is suspicion for epilepsy.
As a quality improvement operational project, the Epilepsy National Program Office approved this validation project and determined that institutional review board approval was not required.
Statistical Analysis
Counts and percentages were computed for categories of epilepsy status. PPV of each tier was estimated with asymptotic 95% CIs.
Results
ICD-10 codes for 480 patients were evaluated in Tier 1 phase 1; 13.8% were documented with G40.xxx, 27.9% with R56.1, 34.4% with R56.9, and 24.0% with R40.4 (Appendix 2). In total, 68.1% fulfilled the criteria of epilepsy, 19.2% did not, and 12.7% were uncertain). From the validation of Tier 1 phase 2 (n = 625), the PPV of the algorithm for patients presumed to have epilepsy (definite and uncertain) was 85.1% (95% CI, 82.1%-87.8%) (Table).

Of 300 patients evaluated, 126 (42.0%) were evaluated for Tier 2 with a PPV of 61.9% (95% CI, 53.4%-70.4%), and 174 (58.0%) patients were evaluated for Tier 3 with a PPV of 59.8% (95% CI, 52.5%-67.1%. The PPV of the algorithm for patients presumed to have epilepsy (definite and uncertain) were combined to calculate the PPV. Estimates of VHA VWE counts were computed for each tier from FY 2014 to FY 2023 using the VSSC Neurology Cube (Figure 2). For all years, > 92% patients were classified using the Tier 1 definition.

Discussion
The development and validation of the 3-tier diagnostic algorithm represents an important advancement in the surveillance and management of epilepsy among veterans within the VHA. The validation of this algorithm also demonstrates its practical utility in a large, integrated health care system.
Specific challenges were encountered when attempting to use pre-existing algorithms; these challenges included differences in the usage patterns of diagnostic codes and the patterns of ASM use within the VHA. These challenges prompted the need for a tailored approach, which led to the development of this algorithm. The inclusion of additional ICD-10 codes led to further revisions and subsequent validation. While many of the basic concepts of the algorithm, including ICD codes and ASMs, could work in other institutions, it would be wise for health care organizations to develop their own algorithms because of certain variables, including organizational size, patient demographics, common comorbidities, and the specific configurations of electronic health records and administrative data systems.
Studies have shown that ICD-10 codes for epilepsy (G40.* and/or R56.9) perform well in identifying epilepsy whether they are assigned by neurologists (sensitivity, 97.7%; specificity, 44.1%; PPV, 96.2%; negative predictive value, 57.7%), or in emergency department or hospital discharges (PPV, 75.5%).13,14 The pilot study of the algorithm’s Tier 1 development (phase 1) evaluated whether the selected ICD-10 diagnostic codes accurately included the VWE population within the VHA and revealed that while most codes (eg, epilepsy [G40.xxx]; posttraumatic seizures [R56.1]; and unspecified convulsions [R56.9]), had a low false positive rate (< 16%), the R40.4 code (transient alteration of awareness) had a higher false positivity of 42%. While this is not surprising given the broad spectrum of conditions that can manifest as transient alteration of awareness, it underscores the inherent challenges in diagnosing epilepsy using diagnosis codes.
In phase 2, the Tier 1 algorithm was validated as effective for identifying VWE in the VHA system, as its PPV was determined to be high (85%). In comparison, Tiers 2 and 3, whose criteria did not require data on VHA prescribed ASM use, had lower tiers of epilepsy predictability (PPV about 60% for both). This was thought to be acceptable because Tiers 2 and 3 represent a smaller population of the identified VWEs (about 8%). These VWEs may otherwise have been missed, partly because veterans are not required to get ASMs from the VHA.
Upon VHA implementation in FY 2021, this diagnostic algorithm exhibited significant clinical utility when integrated within the VSSC Neurology Cube. It facilitated an efficient approach to identifying VWEs using readily available databases. This led to better tracking of real-time epilepsy cases, which facilitated improving current resource allocation and targeted intervention strategies such as identification of drug-resistant epilepsy patients, optimizing strategies for telehealth and patient outreach for awareness of epilepsy care resources within VHA. Meanwhile, data acquired by the algorithm over the decade since its development (FY 2014 to FY 2023) contributed to more accurate epidemiologic information and identification of historic trends. Development of the algorithm represents one of the ways ECoEs have led to improved care for VWEs. ECoEs have been shown to improve health care for veterans in several metrics.15
A strength of this study is the rigorous multitiered validation process to confirm the diagnostic accuracy of ICD-10 codes against the gold standard ILAE definition of epilepsy to identify “definite” epilepsy cases within the VHA. The use of specific ICD codes further enhances the precision of epilepsy diagnoses. The inclusion of ASMs, which are sometimes prescribed for conditions other than epilepsy, could potentially inflate false positive rates.16
This study focused exclusively on the identification and validation of definite epilepsy cases within the VHA VSSC database, employing more stringent diagnostic criteria to ensure the highest level of certainty in ascertaining epilepsy. It is important to note there is a separate category of probable epilepsy, which involves a broader set of diagnostic criteria. While not covered in this study, probable epilepsy would be subject to future research and validation, which could provide insights into a wider spectrum of epilepsy diagnoses. Such future research could help refine the algorithm’s applicability and accuracy and potentially lead to more comprehensive surveillance and management strategies in clinical practice.
This study highlights the inherent challenges in leveraging administrative data for disease identification, particularly for conditions such as epilepsy, where diagnostic clarity can be complex. However, other conditions such as multiple sclerosis have noted similar success with the use of VHA administrative data for categorizing disease.17
Limitations
The algorithm discussed in this article is, in and of itself, generalizable. However, the validation process was unique to the VHA patient population, limiting the generalizability of the findings. Documentation practices and HCP attitudes within the VHA may differ from those in other health care settings. Identifying people with epilepsy can be challenging because of changing definitions of epilepsy over time. In addition to clinical evaluation, EEG and magnetic resonance imaging results, response to ASM treatment, and video-EEG monitoring of habitual events all can help establish the diagnosis. Therefore, studies may vary in how inclusive or exclusive the criteria are. ASMs such as gabapentin, pregabalin, carbamazepine, lamotrigine, topiramate, and valproate are used to treat other conditions, including headaches, generalized pain, and mood disorders. Consequently, including these ASMs in the Tier 1 definition may have increased the false positive rate. Additional research is needed to evaluate whether excluding these ASMs from the algorithm based on specific criteria (eg, dose of ASM used) can further refine the algorithm to identify patients with epilepsy.
Further refinement of this algorithm may also occur as technology changes. Future electronic health records may allow better tracking of different epilepsy factors, the integration of additional diagnostic criteria, and the use of natural language processing or other forms of artificial intelligence.
Conclusions
This study presents a significant step forward in epilepsy surveillance within the VHA. The algorithm offers a robust tool for identifying VWEs with good PPVs, facilitating better resource allocation and targeted care. Despite its limitations, this research lays a foundation for future advancements in the management and understanding of epilepsy within large health care systems. Since this VHA algorithm is based on ASMs and ICD diagnosis codes from patient records, other large managed health care systems also may be able to adapt this algorithm to their data specifications.


Epilepsy affects about 4.5 million people in the United States and 150,000 new individuals are diagnosed each year.1,2 In 2019, epilepsy-attributable health care spending for noninstitutionalized people was around $5.4 billion and total epilepsy-attributable and epilepsy or seizure health care-related costs totaled $54 billion.3
Accurate surveillance of epilepsy in large health care systems can potentially improve health care delivery and resource allocation. A 2012 Institute of Medicine (IOM) report identified 13 recommendations to guide public health action on epilepsy, including validation of standard definitions for case ascertainment, identification of epilepsy through screening programs or protocols, and expansion of surveillance to better understand disease burden.4
A systematic review of validation studies concluded that it is reasonable to use administrative data to identify people with epilepsy in epidemiologic research. Combining The International Classification of Diseases (ICD) codes for epilepsy (ICD-10, G40-41; ICD-9, 345) with antiseizure medications (ASMs) could provide high positive predictive values (PPVs) and combining symptoms codes for convulsions (ICD-10, R56; ICD-9, 780.3, 780.39) with ASMs could lead to high sensitivity.5 However, identifying individuals with epilepsy from administrative data in large managed health care organizations is challenging.6 The IOM report noted that large managed health care organizations presented varying incidence and prevalence estimates due to differing methodology, geographic area, demographics, and definitions of epilepsy.
The Veterans Health Administration (VHA) is the largest integrated US health care system, providing care to > 9.1 million veterans.7 To improve the health and well-being of veterans with epilepsy (VWEs), a network of sites was established in 2008 called the US Department of Veterans Affairs (VA) Epilepsy Centers of Excellence (ECoE). Subsequent to the creation of the ECoE, efforts were made to identify VWEs within VHA databases.8,9 Prior to fiscal year (FY) 2016, the ECoE adopted a modified version of a well-established epilepsy diagnostic algorithm developed by Holden et al for large managed care organizations.10 The original algorithm identified patients by cross-matching ASMs with ICD-9 codes for an index year. But it failed to capture a considerable number of stable patients with epilepsy in the VHA due to incomplete documentation, and had false positives due to inclusion of patients identified from diagnostic clinics. The modified algorithm the ECoE used prior to FY 2016 considered additional prior years and excluded encounters from diagnostic clinics. The result was an improvement in the sensitivity and specificity of the algorithm. Researchers evaluating 500 patients with epilepsy estimated that the modified algorithm had a PPV of 82.0% (95% CI, 78.6%-85.4%).11
After implementation of ICD-10 codes in the VHA in FY 2016, the task of reliably and efficiently identifying VWE led to a 3-tier algorithm. This article presents a validation of the different tiers of this algorithm after the implementation of ICD-10 diagnosis codes and summarizes the surveillance data collected over the years within the VHA showing the trends of epilepsy.
Methods
The VHA National Neurology office commissioned a Neurology Cube dashboard in FY 2021 in collaboration with VHA Support Service Center (VSSC) for reporting and surveillance of VWEs as a quality improvement initiative. The Neurology Cube uses a 3-tier system for identifying VWE in the VHA databases. VSSC programmers extract data from the VHA Corporate Data Warehouse (CDW) and utilize Microsoft SQL Server and Microsoft Power BI for Neurology Cube reports. The 3-tier system identifies VWE and divides them into distinct groups. The first tier identifies VWE with the highest degree of confidence; Tiers 2 and 3 represent identification with successively lesser degrees of confidence (Figure 1).

Tier 1
Definition. For a given index year and the preceding 2 years, any of following diagnosis codes on ≥ 1 clinical encounter are considered: 345.xx (epilepsy in ICD-9), 780.3x (other convulsions in ICD-9), G40.xxx (epilepsy in ICD-10), R40.4 (transient alteration of awareness), R56.1 (posttraumatic seizures), or R56.9 (unspecified convulsions). To reduce false positive rates, EEG clinic visits, which may include long-term monitoring, are excluded. Patients identified with ICD codes are then evaluated for an ASM prescription for ≥ 30 days during the index year. ASMs are listed in Appendix 1.
Validation. The development and validation of ICD-9 diagnosis codes crossmatched with an ASM prescription in the VHA has been published elsewhere.11 In FY 2017, after implementation of ICD-10 diagnostic codes, Tier 1 development and validation was performed in 2 phases. Even though Tier 1 study phases were conducted and completed during FY 2017, the patients for Tier 1 were identified from evaluation of FY 2016 data (October 1, 2015, to September 30, 2016). After the pilot analysis, the Tier 1 definition was implemented, and a chart review of 625 randomized patients was conducted at 5 sites for validation. Adequate preliminary data was not available to perform a sample size estimation for this study. Therefore, a practical target of 125 patients was set for Tier 1 from each site to obtain a final sample size of 625 patients. This second phase validated that the crossmatch of ICD-10 diagnosis codes with ASMs had a high PPV for identifying VWE.
Tiers 2 and 3
Definitions. For an index year, Tier 2 includes patients with ≥ 1 inpatient encounter documentation of either ICD-9 345.xx or ICD-10 G40.xxx, excluding EEG clinics. Tier 3 Includes patients who have had ≥ 2 outpatient encounters with diagnosis codes 345.xx or G40.xxx on 2 separate days, excluding EEG clinics. Tiers 2 and 3 do not require ASM prescriptions; this helps to identify VWEs who may be getting their medications outside of VHA or those who have received a new diagnosis.
Validations. Tiers 2 and 3 were included in the epilepsy identification algorithm in FY 2021 after validation was performed on a sample of 8 patients in each tier. Five patients were subsequently identified as having epilepsy in Tier 2 and 6 patients were identified in Tier 3. A more comprehensive validation of Tiers 2 and 3 was performed during FY 2022 that included patients at 5 sites seen during FY 2019 to FY 2022. Since yearly trends showed only about 8% of total patients were identified as having epilepsy through Tiers 2 and 3 we sought ≥ 20 patients per tier for the 5 sites for a total of 200 patients to ensure representation across the VHA. The final count was 126 patients for Tier 2 and 174 patients for Tier 3 (n = 300).
Gold Standard Criteria for Epilepsy Diagnosis
We used the International League Against Epilepsy (ILAE) definition of epilepsy for the validation of the 3 algorithm tiers. ILAE defines epilepsy as ≥ 2 unprovoked (or reflex) seizures occurring > 24 hours apart or 1 unprovoked (or reflex) seizure and a probability of further seizures similar to the general recurrence risk (≥ 60%) after 2 unprovoked seizures, occurring over the next 10 years.12
A standard protocol was provided to evaluators to identify patients using the VHA Computerized Patient Record System (Appendix 1). After review, evaluators categorized each patient in 1 of 4 ways: (1) Yes, definite: The patient’s health care practitioner (HCP) believes the patient has epilepsy and is treating with medication; (2) Yes, uncertain: The HCP has enough suspicion of epilepsy that a medication is prescribed, but uncertainty is expressed of the diagnosis; (3) No, definite: The HCP does not believe the patient has epilepsy and is therefore not treating with medication for seizure; (4) No, uncertain: The HCP is not treating with medication for epilepsy, because the diagnostic suspicion is not high enough, but there is suspicion for epilepsy.
As a quality improvement operational project, the Epilepsy National Program Office approved this validation project and determined that institutional review board approval was not required.
Statistical Analysis
Counts and percentages were computed for categories of epilepsy status. PPV of each tier was estimated with asymptotic 95% CIs.
Results
ICD-10 codes for 480 patients were evaluated in Tier 1 phase 1; 13.8% were documented with G40.xxx, 27.9% with R56.1, 34.4% with R56.9, and 24.0% with R40.4 (Appendix 2). In total, 68.1% fulfilled the criteria of epilepsy, 19.2% did not, and 12.7% were uncertain). From the validation of Tier 1 phase 2 (n = 625), the PPV of the algorithm for patients presumed to have epilepsy (definite and uncertain) was 85.1% (95% CI, 82.1%-87.8%) (Table).

Of 300 patients evaluated, 126 (42.0%) were evaluated for Tier 2 with a PPV of 61.9% (95% CI, 53.4%-70.4%), and 174 (58.0%) patients were evaluated for Tier 3 with a PPV of 59.8% (95% CI, 52.5%-67.1%. The PPV of the algorithm for patients presumed to have epilepsy (definite and uncertain) were combined to calculate the PPV. Estimates of VHA VWE counts were computed for each tier from FY 2014 to FY 2023 using the VSSC Neurology Cube (Figure 2). For all years, > 92% patients were classified using the Tier 1 definition.

Discussion
The development and validation of the 3-tier diagnostic algorithm represents an important advancement in the surveillance and management of epilepsy among veterans within the VHA. The validation of this algorithm also demonstrates its practical utility in a large, integrated health care system.
Specific challenges were encountered when attempting to use pre-existing algorithms; these challenges included differences in the usage patterns of diagnostic codes and the patterns of ASM use within the VHA. These challenges prompted the need for a tailored approach, which led to the development of this algorithm. The inclusion of additional ICD-10 codes led to further revisions and subsequent validation. While many of the basic concepts of the algorithm, including ICD codes and ASMs, could work in other institutions, it would be wise for health care organizations to develop their own algorithms because of certain variables, including organizational size, patient demographics, common comorbidities, and the specific configurations of electronic health records and administrative data systems.
Studies have shown that ICD-10 codes for epilepsy (G40.* and/or R56.9) perform well in identifying epilepsy whether they are assigned by neurologists (sensitivity, 97.7%; specificity, 44.1%; PPV, 96.2%; negative predictive value, 57.7%), or in emergency department or hospital discharges (PPV, 75.5%).13,14 The pilot study of the algorithm’s Tier 1 development (phase 1) evaluated whether the selected ICD-10 diagnostic codes accurately included the VWE population within the VHA and revealed that while most codes (eg, epilepsy [G40.xxx]; posttraumatic seizures [R56.1]; and unspecified convulsions [R56.9]), had a low false positive rate (< 16%), the R40.4 code (transient alteration of awareness) had a higher false positivity of 42%. While this is not surprising given the broad spectrum of conditions that can manifest as transient alteration of awareness, it underscores the inherent challenges in diagnosing epilepsy using diagnosis codes.
In phase 2, the Tier 1 algorithm was validated as effective for identifying VWE in the VHA system, as its PPV was determined to be high (85%). In comparison, Tiers 2 and 3, whose criteria did not require data on VHA prescribed ASM use, had lower tiers of epilepsy predictability (PPV about 60% for both). This was thought to be acceptable because Tiers 2 and 3 represent a smaller population of the identified VWEs (about 8%). These VWEs may otherwise have been missed, partly because veterans are not required to get ASMs from the VHA.
Upon VHA implementation in FY 2021, this diagnostic algorithm exhibited significant clinical utility when integrated within the VSSC Neurology Cube. It facilitated an efficient approach to identifying VWEs using readily available databases. This led to better tracking of real-time epilepsy cases, which facilitated improving current resource allocation and targeted intervention strategies such as identification of drug-resistant epilepsy patients, optimizing strategies for telehealth and patient outreach for awareness of epilepsy care resources within VHA. Meanwhile, data acquired by the algorithm over the decade since its development (FY 2014 to FY 2023) contributed to more accurate epidemiologic information and identification of historic trends. Development of the algorithm represents one of the ways ECoEs have led to improved care for VWEs. ECoEs have been shown to improve health care for veterans in several metrics.15
A strength of this study is the rigorous multitiered validation process to confirm the diagnostic accuracy of ICD-10 codes against the gold standard ILAE definition of epilepsy to identify “definite” epilepsy cases within the VHA. The use of specific ICD codes further enhances the precision of epilepsy diagnoses. The inclusion of ASMs, which are sometimes prescribed for conditions other than epilepsy, could potentially inflate false positive rates.16
This study focused exclusively on the identification and validation of definite epilepsy cases within the VHA VSSC database, employing more stringent diagnostic criteria to ensure the highest level of certainty in ascertaining epilepsy. It is important to note there is a separate category of probable epilepsy, which involves a broader set of diagnostic criteria. While not covered in this study, probable epilepsy would be subject to future research and validation, which could provide insights into a wider spectrum of epilepsy diagnoses. Such future research could help refine the algorithm’s applicability and accuracy and potentially lead to more comprehensive surveillance and management strategies in clinical practice.
This study highlights the inherent challenges in leveraging administrative data for disease identification, particularly for conditions such as epilepsy, where diagnostic clarity can be complex. However, other conditions such as multiple sclerosis have noted similar success with the use of VHA administrative data for categorizing disease.17
Limitations
The algorithm discussed in this article is, in and of itself, generalizable. However, the validation process was unique to the VHA patient population, limiting the generalizability of the findings. Documentation practices and HCP attitudes within the VHA may differ from those in other health care settings. Identifying people with epilepsy can be challenging because of changing definitions of epilepsy over time. In addition to clinical evaluation, EEG and magnetic resonance imaging results, response to ASM treatment, and video-EEG monitoring of habitual events all can help establish the diagnosis. Therefore, studies may vary in how inclusive or exclusive the criteria are. ASMs such as gabapentin, pregabalin, carbamazepine, lamotrigine, topiramate, and valproate are used to treat other conditions, including headaches, generalized pain, and mood disorders. Consequently, including these ASMs in the Tier 1 definition may have increased the false positive rate. Additional research is needed to evaluate whether excluding these ASMs from the algorithm based on specific criteria (eg, dose of ASM used) can further refine the algorithm to identify patients with epilepsy.
Further refinement of this algorithm may also occur as technology changes. Future electronic health records may allow better tracking of different epilepsy factors, the integration of additional diagnostic criteria, and the use of natural language processing or other forms of artificial intelligence.
Conclusions
This study presents a significant step forward in epilepsy surveillance within the VHA. The algorithm offers a robust tool for identifying VWEs with good PPVs, facilitating better resource allocation and targeted care. Despite its limitations, this research lays a foundation for future advancements in the management and understanding of epilepsy within large health care systems. Since this VHA algorithm is based on ASMs and ICD diagnosis codes from patient records, other large managed health care systems also may be able to adapt this algorithm to their data specifications.


- Kobau R, Luncheon C, Greenlund K. Active epilepsy prevalence among U.S. adults is 1.1% and differs by educational level-National Health Interview Survey, United States, 2021. Epilepsy Behav. 2023;142:109180. doi:10.1016/j.yebeh.2023.109180
- GBD 2017 US Neurological Disorders Collaborators, Feigin VL, Vos T, et al. Burden of neurological disorders across the US from 1990-2017: a global burden of disease study. JAMA Neurol. 2021;78:165-176. doi:10.1001/jamaneurol.2020.4152
- Moura LMVR, Karakis I, Zack MM, et al. Drivers of US health care spending for persons with seizures and/or epilepsies, 2010-2018. Epilepsia. 2022;63:2144-2154. doi:10.1111/epi.17305
- Institute of Medicine. Epilepsy Across the Spectrum: Promoting Health and Understanding. The National Academies Press; 2012. Accessed November 11, 2025. www.nap.edu/catalog/13379
- Mbizvo GK, Bennett KH, Schnier C, Simpson CR, Duncan SE, Chin RFM. The accuracy of using administrative healthcare data to identify epilepsy cases: A systematic review of validation studies. Epilepsia. 2020;61:1319-1335. doi:10.1111/epi.16547
- Montouris GD. How will primary care physicians, specialists, and managed care treat epilepsy in the new millennium? Neurology. 2000;55:S42-S44.
- US Department of Veterans Affairs. Veterans Health Administration: About VHA. Accessed November 11, 2025. https://www.va.gov/health/aboutvha.asp
- Veterans’ Mental Health and Other Care Improvements Act of 2008, S 2162, 110th Cong (2008). Accessed November 11, 2025. https://www.congress.gov/bill/110th-congress/senate-bill/2162
- Rehman R, Kelly PR, Husain AM, Tran TT. Characteristics of Veterans diagnosed with seizures within Veterans Health Administration. J Rehabil Res Dev. 2015;52(7):751-762. doi:10.1682/JRRD.2014.10.0241
- Holden EW, Grossman E, Nguyen HT, et al. Developing a computer algorithm to identify epilepsy cases in managed care organizations. Dis Manag. 2005;8:1-14. doi:10.1089/dis.2005.8.1
- Rehman R, Everhart A, Frontera AT, et al. Implementation of an established algorithm and modifications for the identification of epilepsy patients in the Veterans Health Administration. Epilepsy Res. 2016;127:284-290. doi:10.1016/j.eplepsyres.2016.09.012
- Fisher RS, Acevedo C, Arzimanoglou A, et al. ILAE official report: a practical clinical definition of epilepsy. Epilepsia. 2014;55:475-482. doi:10.1111/epi.12550
- Smith JR, Jones FJS, Fureman BE, et al. Accuracy of ICD-10-CM claims-based definitions for epilepsy and seizure type. Epilepsy Res. 2020;166:106414. doi:10.1016/j.eplepsyres.2020.106414
- Jetté N, Reid AY, Quan H, et al. How accurate is ICD coding for epilepsy? Epilepsia. 2010;51:62-69. doi:10.1111/j.1528-1167.2009.02201.x
- Kelly P, Chinta R, Privitera G. Do centers of excellence reduce health care costs? Evidence from the US Veterans Health Administration Centers for Epilepsy. Glob Bus Organ Excell. 2015;34:18-29.
- Haneef Z, Rehman R, Husain AM. Association between standardized mortality ratio and utilization of care in US veterans with drug-resistant epilepsy compared with all US veterans and the US general population. JAMA Neurol. 2022;79:879-887. doi:10.1001/jamaneurol.2022.2290
- Culpepper WJ, Marrie RA, Langer-Gould A, et al. Validation of an algorithm for identifying MS cases in administrative health claims datasets. Neurology. 2019;92:e1016-e1028 doi:10.1212/WNL.0000000000007043
- Kobau R, Luncheon C, Greenlund K. Active epilepsy prevalence among U.S. adults is 1.1% and differs by educational level-National Health Interview Survey, United States, 2021. Epilepsy Behav. 2023;142:109180. doi:10.1016/j.yebeh.2023.109180
- GBD 2017 US Neurological Disorders Collaborators, Feigin VL, Vos T, et al. Burden of neurological disorders across the US from 1990-2017: a global burden of disease study. JAMA Neurol. 2021;78:165-176. doi:10.1001/jamaneurol.2020.4152
- Moura LMVR, Karakis I, Zack MM, et al. Drivers of US health care spending for persons with seizures and/or epilepsies, 2010-2018. Epilepsia. 2022;63:2144-2154. doi:10.1111/epi.17305
- Institute of Medicine. Epilepsy Across the Spectrum: Promoting Health and Understanding. The National Academies Press; 2012. Accessed November 11, 2025. www.nap.edu/catalog/13379
- Mbizvo GK, Bennett KH, Schnier C, Simpson CR, Duncan SE, Chin RFM. The accuracy of using administrative healthcare data to identify epilepsy cases: A systematic review of validation studies. Epilepsia. 2020;61:1319-1335. doi:10.1111/epi.16547
- Montouris GD. How will primary care physicians, specialists, and managed care treat epilepsy in the new millennium? Neurology. 2000;55:S42-S44.
- US Department of Veterans Affairs. Veterans Health Administration: About VHA. Accessed November 11, 2025. https://www.va.gov/health/aboutvha.asp
- Veterans’ Mental Health and Other Care Improvements Act of 2008, S 2162, 110th Cong (2008). Accessed November 11, 2025. https://www.congress.gov/bill/110th-congress/senate-bill/2162
- Rehman R, Kelly PR, Husain AM, Tran TT. Characteristics of Veterans diagnosed with seizures within Veterans Health Administration. J Rehabil Res Dev. 2015;52(7):751-762. doi:10.1682/JRRD.2014.10.0241
- Holden EW, Grossman E, Nguyen HT, et al. Developing a computer algorithm to identify epilepsy cases in managed care organizations. Dis Manag. 2005;8:1-14. doi:10.1089/dis.2005.8.1
- Rehman R, Everhart A, Frontera AT, et al. Implementation of an established algorithm and modifications for the identification of epilepsy patients in the Veterans Health Administration. Epilepsy Res. 2016;127:284-290. doi:10.1016/j.eplepsyres.2016.09.012
- Fisher RS, Acevedo C, Arzimanoglou A, et al. ILAE official report: a practical clinical definition of epilepsy. Epilepsia. 2014;55:475-482. doi:10.1111/epi.12550
- Smith JR, Jones FJS, Fureman BE, et al. Accuracy of ICD-10-CM claims-based definitions for epilepsy and seizure type. Epilepsy Res. 2020;166:106414. doi:10.1016/j.eplepsyres.2020.106414
- Jetté N, Reid AY, Quan H, et al. How accurate is ICD coding for epilepsy? Epilepsia. 2010;51:62-69. doi:10.1111/j.1528-1167.2009.02201.x
- Kelly P, Chinta R, Privitera G. Do centers of excellence reduce health care costs? Evidence from the US Veterans Health Administration Centers for Epilepsy. Glob Bus Organ Excell. 2015;34:18-29.
- Haneef Z, Rehman R, Husain AM. Association between standardized mortality ratio and utilization of care in US veterans with drug-resistant epilepsy compared with all US veterans and the US general population. JAMA Neurol. 2022;79:879-887. doi:10.1001/jamaneurol.2022.2290
- Culpepper WJ, Marrie RA, Langer-Gould A, et al. Validation of an algorithm for identifying MS cases in administrative health claims datasets. Neurology. 2019;92:e1016-e1028 doi:10.1212/WNL.0000000000007043
Development and Validation of an Administrative Algorithm to Identify Veterans With Epilepsy
Development and Validation of an Administrative Algorithm to Identify Veterans With Epilepsy