User login
New recommendations for managing adult AML

Photo courtesy of CDC
The European LeukemiaNet (ELN) has released updated recommendations for the diagnosis and treatment of acute myeloid leukemia (AML) in adults.
The recommendations include revised ELN genetic categories, a proposed response category based on minimal residual disease status, and a proposed category for progressive disease for clinical trials.
They also include the updated World Health Organization classification of myeloid neoplasms and acute leukemia.
The recommendations are published in Blood.
“These guidelines are an important update of the current and widely used recommendations for managing AML, for constructing clinical trials, and for predicting outcomes of AML patients,” said Clara D. Bloomfield, MD, of The Ohio State University Comprehensive Cancer Center in Columbus.
“They will be the new standard of care and will replace the 2010 ELN recommendations for managing AML patients and designing clinical trials.”
Dr Bloomfield said updating the ELN recommendations was prompted by new insights into the molecular and genomic causes of AML, by the development of new genetic tests and tests for detecting minimal residual disease, and by the development of novel anti-leukemic agents.
Changes of note, according to Dr Bloomfield, are that there are now 3 genetic risk categories rather than 4, and the FLT3-ITD mutation has been added as a marker of risk.
In addition, “complete remission with no evidence of minimal residual disease” is a new proposed response category. This criterion requires that genetic markers present at diagnosis are no longer detectable.
“It is no longer good enough to examine bone marrow samples and say the leukemia is gone,” Dr Bloomfield said. “We must also see the loss of genetic markers.”
Another change is that “progressive disease” is a new provisional response category to be used in clinical trials only. The purpose of the category is to harmonize the various definitions of progressive disease that are used in different clinical trials. ![]()

Photo courtesy of CDC
The European LeukemiaNet (ELN) has released updated recommendations for the diagnosis and treatment of acute myeloid leukemia (AML) in adults.
The recommendations include revised ELN genetic categories, a proposed response category based on minimal residual disease status, and a proposed category for progressive disease for clinical trials.
They also include the updated World Health Organization classification of myeloid neoplasms and acute leukemia.
The recommendations are published in Blood.
“These guidelines are an important update of the current and widely used recommendations for managing AML, for constructing clinical trials, and for predicting outcomes of AML patients,” said Clara D. Bloomfield, MD, of The Ohio State University Comprehensive Cancer Center in Columbus.
“They will be the new standard of care and will replace the 2010 ELN recommendations for managing AML patients and designing clinical trials.”
Dr Bloomfield said updating the ELN recommendations was prompted by new insights into the molecular and genomic causes of AML, by the development of new genetic tests and tests for detecting minimal residual disease, and by the development of novel anti-leukemic agents.
Changes of note, according to Dr Bloomfield, are that there are now 3 genetic risk categories rather than 4, and the FLT3-ITD mutation has been added as a marker of risk.
In addition, “complete remission with no evidence of minimal residual disease” is a new proposed response category. This criterion requires that genetic markers present at diagnosis are no longer detectable.
“It is no longer good enough to examine bone marrow samples and say the leukemia is gone,” Dr Bloomfield said. “We must also see the loss of genetic markers.”
Another change is that “progressive disease” is a new provisional response category to be used in clinical trials only. The purpose of the category is to harmonize the various definitions of progressive disease that are used in different clinical trials. ![]()

Photo courtesy of CDC
The European LeukemiaNet (ELN) has released updated recommendations for the diagnosis and treatment of acute myeloid leukemia (AML) in adults.
The recommendations include revised ELN genetic categories, a proposed response category based on minimal residual disease status, and a proposed category for progressive disease for clinical trials.
They also include the updated World Health Organization classification of myeloid neoplasms and acute leukemia.
The recommendations are published in Blood.
“These guidelines are an important update of the current and widely used recommendations for managing AML, for constructing clinical trials, and for predicting outcomes of AML patients,” said Clara D. Bloomfield, MD, of The Ohio State University Comprehensive Cancer Center in Columbus.
“They will be the new standard of care and will replace the 2010 ELN recommendations for managing AML patients and designing clinical trials.”
Dr Bloomfield said updating the ELN recommendations was prompted by new insights into the molecular and genomic causes of AML, by the development of new genetic tests and tests for detecting minimal residual disease, and by the development of novel anti-leukemic agents.
Changes of note, according to Dr Bloomfield, are that there are now 3 genetic risk categories rather than 4, and the FLT3-ITD mutation has been added as a marker of risk.
In addition, “complete remission with no evidence of minimal residual disease” is a new proposed response category. This criterion requires that genetic markers present at diagnosis are no longer detectable.
“It is no longer good enough to examine bone marrow samples and say the leukemia is gone,” Dr Bloomfield said. “We must also see the loss of genetic markers.”
Another change is that “progressive disease” is a new provisional response category to be used in clinical trials only. The purpose of the category is to harmonize the various definitions of progressive disease that are used in different clinical trials. ![]()
Study provides new insight into B-cell metabolism
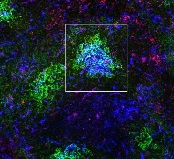
the spleen of a mouse, showing
inactivated GSK3 (magenta)
in B cells (blue) near follicular
dendritic cells (green).
Image from the lab of
Robert Rickert, PhD
Research published in Nature Immunology helps explain how B-cell metabolism adapts to different environments.
The study suggests the protein GSK3 acts as a metabolic checkpoint regulator in B cells, promoting the survival of circulating B cells while limiting the growth and proliferation of B cells in germinal centers.
“Our research shows that the protein GSK3 plays a crucial role in helping B cells meet the energy needs of their distinct states,” said study author Robert Rickert, PhD, of Sanford Burnham Prebys Medical Discovery Institute in La Jolla, California.
“The findings are particularly relevant for certain B-cell pathologies, including lymphoma subtypes, where there is an increased demand for energy to support the hyperproliferation of cells in a microenvironment that may be limited in nutrients.”
Dr Rickert and his colleagues noted that B cells predominate in a quiescent state until they encounter an antigen, which prompts the cells to grow, proliferate, and differentiate.
The team’s new study showed that GSK3 adjusts B-cell metabolism to match the needs of these different cell states.
In circulating B cells, GSK3 limits overall metabolic activity. In proliferating B cells in germinal centers, GSK3 slows glycolysis and the production of mitochondria.
In fact, GSK3 function is essential for B-cell survival in germinal centers. To understand why, the researchers looked at how B cells in these regions generate energy.
The team found that because these B cells are so metabolically active, they consume nearly all available glucose. That switches on glycolysis.
High glycolytic activity leads to an accumulation of toxic reactive oxygen species, as does rapid manufacture of mitochondria, which tend to leak the same chemicals.
Thus, by restraining the metabolism in specific ways, GSK3 prevents cell death induced by reactive oxygen species.
“Our results were really surprising,” Dr Rickert said. “Until now, we would have thought that slowing metabolism would only be important for preventing B cells from becoming cancerous, which it indeed may be. These studies provide insight into the dynamic nature of B-cell metabolism that literally ‘fuels’ differentiation in the germinal center to produce an effective antibody response.”
“It’s not yet clear whether or how GSK3 might be a target for future therapies for B cell-related diseases, but this research opens a lot of doors for further studies. To start with, we plan to investigate how GSK3 is regulated in lymphoma and how that relates to changes in metabolism. That research could lead to new approaches to treating lymphoma.”
This research was performed in collaboration with scientists at Eli Lilly and the Lunenfeld-Tanenbaum Research Institute at the University of Toronto. Funding was provided by the National Institutes of Health, the Lilly Research Award Program, the Arthritis National Research Foundation, and the Canadian Institutes of Health Research. ![]()

the spleen of a mouse, showing
inactivated GSK3 (magenta)
in B cells (blue) near follicular
dendritic cells (green).
Image from the lab of
Robert Rickert, PhD
Research published in Nature Immunology helps explain how B-cell metabolism adapts to different environments.
The study suggests the protein GSK3 acts as a metabolic checkpoint regulator in B cells, promoting the survival of circulating B cells while limiting the growth and proliferation of B cells in germinal centers.
“Our research shows that the protein GSK3 plays a crucial role in helping B cells meet the energy needs of their distinct states,” said study author Robert Rickert, PhD, of Sanford Burnham Prebys Medical Discovery Institute in La Jolla, California.
“The findings are particularly relevant for certain B-cell pathologies, including lymphoma subtypes, where there is an increased demand for energy to support the hyperproliferation of cells in a microenvironment that may be limited in nutrients.”
Dr Rickert and his colleagues noted that B cells predominate in a quiescent state until they encounter an antigen, which prompts the cells to grow, proliferate, and differentiate.
The team’s new study showed that GSK3 adjusts B-cell metabolism to match the needs of these different cell states.
In circulating B cells, GSK3 limits overall metabolic activity. In proliferating B cells in germinal centers, GSK3 slows glycolysis and the production of mitochondria.
In fact, GSK3 function is essential for B-cell survival in germinal centers. To understand why, the researchers looked at how B cells in these regions generate energy.
The team found that because these B cells are so metabolically active, they consume nearly all available glucose. That switches on glycolysis.
High glycolytic activity leads to an accumulation of toxic reactive oxygen species, as does rapid manufacture of mitochondria, which tend to leak the same chemicals.
Thus, by restraining the metabolism in specific ways, GSK3 prevents cell death induced by reactive oxygen species.
“Our results were really surprising,” Dr Rickert said. “Until now, we would have thought that slowing metabolism would only be important for preventing B cells from becoming cancerous, which it indeed may be. These studies provide insight into the dynamic nature of B-cell metabolism that literally ‘fuels’ differentiation in the germinal center to produce an effective antibody response.”
“It’s not yet clear whether or how GSK3 might be a target for future therapies for B cell-related diseases, but this research opens a lot of doors for further studies. To start with, we plan to investigate how GSK3 is regulated in lymphoma and how that relates to changes in metabolism. That research could lead to new approaches to treating lymphoma.”
This research was performed in collaboration with scientists at Eli Lilly and the Lunenfeld-Tanenbaum Research Institute at the University of Toronto. Funding was provided by the National Institutes of Health, the Lilly Research Award Program, the Arthritis National Research Foundation, and the Canadian Institutes of Health Research. ![]()

the spleen of a mouse, showing
inactivated GSK3 (magenta)
in B cells (blue) near follicular
dendritic cells (green).
Image from the lab of
Robert Rickert, PhD
Research published in Nature Immunology helps explain how B-cell metabolism adapts to different environments.
The study suggests the protein GSK3 acts as a metabolic checkpoint regulator in B cells, promoting the survival of circulating B cells while limiting the growth and proliferation of B cells in germinal centers.
“Our research shows that the protein GSK3 plays a crucial role in helping B cells meet the energy needs of their distinct states,” said study author Robert Rickert, PhD, of Sanford Burnham Prebys Medical Discovery Institute in La Jolla, California.
“The findings are particularly relevant for certain B-cell pathologies, including lymphoma subtypes, where there is an increased demand for energy to support the hyperproliferation of cells in a microenvironment that may be limited in nutrients.”
Dr Rickert and his colleagues noted that B cells predominate in a quiescent state until they encounter an antigen, which prompts the cells to grow, proliferate, and differentiate.
The team’s new study showed that GSK3 adjusts B-cell metabolism to match the needs of these different cell states.
In circulating B cells, GSK3 limits overall metabolic activity. In proliferating B cells in germinal centers, GSK3 slows glycolysis and the production of mitochondria.
In fact, GSK3 function is essential for B-cell survival in germinal centers. To understand why, the researchers looked at how B cells in these regions generate energy.
The team found that because these B cells are so metabolically active, they consume nearly all available glucose. That switches on glycolysis.
High glycolytic activity leads to an accumulation of toxic reactive oxygen species, as does rapid manufacture of mitochondria, which tend to leak the same chemicals.
Thus, by restraining the metabolism in specific ways, GSK3 prevents cell death induced by reactive oxygen species.
“Our results were really surprising,” Dr Rickert said. “Until now, we would have thought that slowing metabolism would only be important for preventing B cells from becoming cancerous, which it indeed may be. These studies provide insight into the dynamic nature of B-cell metabolism that literally ‘fuels’ differentiation in the germinal center to produce an effective antibody response.”
“It’s not yet clear whether or how GSK3 might be a target for future therapies for B cell-related diseases, but this research opens a lot of doors for further studies. To start with, we plan to investigate how GSK3 is regulated in lymphoma and how that relates to changes in metabolism. That research could lead to new approaches to treating lymphoma.”
This research was performed in collaboration with scientists at Eli Lilly and the Lunenfeld-Tanenbaum Research Institute at the University of Toronto. Funding was provided by the National Institutes of Health, the Lilly Research Award Program, the Arthritis National Research Foundation, and the Canadian Institutes of Health Research. ![]()
CHMP recommends lenalidomide maintenance

Photo courtesy of Celgene
The European Medicines Agency’s (EMA) Committee for Medicinal Products for Human Use (CHMP) has recommended a new indication for lenalidomide (Revlimid®).
The CHMP advised the European Commission (EC) to approve the use of lenalidomide as maintenance therapy in adults who had newly diagnosed multiple myeloma (MM) prior to receiving an autologous stem cell transplant (ASCT).
If approved by the EC, lenalidomide will be the first licensed maintenance treatment available to this patient population in the European Union.
The EC, which generally follows the CHMP’s recommendations, is expected to make its final decision on this use of lenalidomide in approximately 2 months.
If approval is granted, detailed conditions for the use of lenalidomide will be described in the Summary of Product Characteristics, which will be published in the revised European Public Assessment Report.
Lenalidomide is a product of Celgene.
The CHMP’s recommendation to approve lenalidomide as maintenance in MM was based on the results of 2 cooperative group-led studies, CALGB 10010410 and IFM 2005-0211. Results from both studies were published in NEJM in May 2012.
CALGB 100104 was a phase 3, double-blind study of 460 patients with newly diagnosed MM undergoing ASCT. The patients received continuous daily treatment with lenalidomide or placebo until relapse.
IFM 2005-02 was a phase 3, double-blind study of 614 patients newly diagnosed with MM. The patients were randomized to receive a 2-month consolidation regimen post-ASCT of lenalidomide monotherapy, followed by continuous daily treatment with lenalidomide or placebo until relapse.
“Studies show that maintenance treatment after ASCT with Revlimid may help control residual malignant cells and delay tumor growth by enhancing immune function,” said Michel Attal, MD, of the Institut Universitaire du Cancer Toulouse Oncopole and Institut Claudius Regaud in France.
“Our primary goal is to delay disease progression for as long as possible, and we have seen in several independent studies that Revlimid maintenance after ASCT can halve the risk of disease progression by sustaining the response.” ![]()

Photo courtesy of Celgene
The European Medicines Agency’s (EMA) Committee for Medicinal Products for Human Use (CHMP) has recommended a new indication for lenalidomide (Revlimid®).
The CHMP advised the European Commission (EC) to approve the use of lenalidomide as maintenance therapy in adults who had newly diagnosed multiple myeloma (MM) prior to receiving an autologous stem cell transplant (ASCT).
If approved by the EC, lenalidomide will be the first licensed maintenance treatment available to this patient population in the European Union.
The EC, which generally follows the CHMP’s recommendations, is expected to make its final decision on this use of lenalidomide in approximately 2 months.
If approval is granted, detailed conditions for the use of lenalidomide will be described in the Summary of Product Characteristics, which will be published in the revised European Public Assessment Report.
Lenalidomide is a product of Celgene.
The CHMP’s recommendation to approve lenalidomide as maintenance in MM was based on the results of 2 cooperative group-led studies, CALGB 10010410 and IFM 2005-0211. Results from both studies were published in NEJM in May 2012.
CALGB 100104 was a phase 3, double-blind study of 460 patients with newly diagnosed MM undergoing ASCT. The patients received continuous daily treatment with lenalidomide or placebo until relapse.
IFM 2005-02 was a phase 3, double-blind study of 614 patients newly diagnosed with MM. The patients were randomized to receive a 2-month consolidation regimen post-ASCT of lenalidomide monotherapy, followed by continuous daily treatment with lenalidomide or placebo until relapse.
“Studies show that maintenance treatment after ASCT with Revlimid may help control residual malignant cells and delay tumor growth by enhancing immune function,” said Michel Attal, MD, of the Institut Universitaire du Cancer Toulouse Oncopole and Institut Claudius Regaud in France.
“Our primary goal is to delay disease progression for as long as possible, and we have seen in several independent studies that Revlimid maintenance after ASCT can halve the risk of disease progression by sustaining the response.” ![]()

Photo courtesy of Celgene
The European Medicines Agency’s (EMA) Committee for Medicinal Products for Human Use (CHMP) has recommended a new indication for lenalidomide (Revlimid®).
The CHMP advised the European Commission (EC) to approve the use of lenalidomide as maintenance therapy in adults who had newly diagnosed multiple myeloma (MM) prior to receiving an autologous stem cell transplant (ASCT).
If approved by the EC, lenalidomide will be the first licensed maintenance treatment available to this patient population in the European Union.
The EC, which generally follows the CHMP’s recommendations, is expected to make its final decision on this use of lenalidomide in approximately 2 months.
If approval is granted, detailed conditions for the use of lenalidomide will be described in the Summary of Product Characteristics, which will be published in the revised European Public Assessment Report.
Lenalidomide is a product of Celgene.
The CHMP’s recommendation to approve lenalidomide as maintenance in MM was based on the results of 2 cooperative group-led studies, CALGB 10010410 and IFM 2005-0211. Results from both studies were published in NEJM in May 2012.
CALGB 100104 was a phase 3, double-blind study of 460 patients with newly diagnosed MM undergoing ASCT. The patients received continuous daily treatment with lenalidomide or placebo until relapse.
IFM 2005-02 was a phase 3, double-blind study of 614 patients newly diagnosed with MM. The patients were randomized to receive a 2-month consolidation regimen post-ASCT of lenalidomide monotherapy, followed by continuous daily treatment with lenalidomide or placebo until relapse.
“Studies show that maintenance treatment after ASCT with Revlimid may help control residual malignant cells and delay tumor growth by enhancing immune function,” said Michel Attal, MD, of the Institut Universitaire du Cancer Toulouse Oncopole and Institut Claudius Regaud in France.
“Our primary goal is to delay disease progression for as long as possible, and we have seen in several independent studies that Revlimid maintenance after ASCT can halve the risk of disease progression by sustaining the response.” ![]()
Software could improve image analysis, team says
Researchers say they have developed new software that will analyze medical and scientific images faster and more accurately than ever before.
The team says this software, Tracking Equilibrium and Nonequilibrium shifts in Data (TREND), can analyze any series of images, including nuclear magnetic resonance images, computerized tomography scans, ultrasound images, video images, and imaging from scientific equipment of all kinds.
The researchers described the TREND software in Biophysical Journal.
The team said TREND can study sets of images to resolve and track the changes among the images.
And the software can analyze videos to plot and resolve changes as well as reconstruct videos to focus only on the individual processes and changes of interest.
“TREND allows accurate, rapid analysis of incredibly complex and nuanced images, which can potentially save doctors, patients, and scientists countless hours and money,” said Steve Van Doren, PhD, of the University of Missouri in Columbia, Missouri.
“TREND has allowed us to advance our own research into enzyme interactions considerably. Previously, it would take us weeks to analyze a single group of images. With TREND, that analysis now takes only a few minutes and is more accurate and consistent than if a human performed the work.” ![]()

Researchers say they have developed new software that will analyze medical and scientific images faster and more accurately than ever before.
The team says this software, Tracking Equilibrium and Nonequilibrium shifts in Data (TREND), can analyze any series of images, including nuclear magnetic resonance images, computerized tomography scans, ultrasound images, video images, and imaging from scientific equipment of all kinds.
The researchers described the TREND software in Biophysical Journal.
The team said TREND can study sets of images to resolve and track the changes among the images.
And the software can analyze videos to plot and resolve changes as well as reconstruct videos to focus only on the individual processes and changes of interest.
“TREND allows accurate, rapid analysis of incredibly complex and nuanced images, which can potentially save doctors, patients, and scientists countless hours and money,” said Steve Van Doren, PhD, of the University of Missouri in Columbia, Missouri.
“TREND has allowed us to advance our own research into enzyme interactions considerably. Previously, it would take us weeks to analyze a single group of images. With TREND, that analysis now takes only a few minutes and is more accurate and consistent than if a human performed the work.” ![]()

Researchers say they have developed new software that will analyze medical and scientific images faster and more accurately than ever before.
The team says this software, Tracking Equilibrium and Nonequilibrium shifts in Data (TREND), can analyze any series of images, including nuclear magnetic resonance images, computerized tomography scans, ultrasound images, video images, and imaging from scientific equipment of all kinds.
The researchers described the TREND software in Biophysical Journal.
The team said TREND can study sets of images to resolve and track the changes among the images.
And the software can analyze videos to plot and resolve changes as well as reconstruct videos to focus only on the individual processes and changes of interest.
“TREND allows accurate, rapid analysis of incredibly complex and nuanced images, which can potentially save doctors, patients, and scientists countless hours and money,” said Steve Van Doren, PhD, of the University of Missouri in Columbia, Missouri.
“TREND has allowed us to advance our own research into enzyme interactions considerably. Previously, it would take us weeks to analyze a single group of images. With TREND, that analysis now takes only a few minutes and is more accurate and consistent than if a human performed the work.” ![]()
Combo granted orphan designation for DLBCL

The US Food and Drug Administration (FDA) has granted orphan drug designation for the combination of TG-1101 (ublituximab), an anti-CD20 monoclonal antibody, and TGR-1202, a PI3K delta inhibitor, in the treatment of diffuse large B-cell lymphoma (DLBCL).
The combination is currently being evaluated in patients with relapsed or refractory DLBCL in the phase 2b UNITY-DLBCL trial.
Ublituximab and TGR-1202 are both products of TG Therapeutics, Inc.
Updated results from a phase 1 study of ublituximab and TGR-1202 in patients with DLBCL and other malignancies were presented at the 21st Congress of the European Hematology Association.
The data included 165 patients treated with varying doses of TGR-1202 alone (n=90) or in combination with ublituximab (n=75). The patients were heavily pretreated, with the majority having 3 or more prior lines of therapy.
There were 7 evaluable patients with DLBCL who received the combination at the phase 3 doses— ublituximab at 900 mg and TGR-1202 at 800 mg micronized.
The overall response rate for this group was 57%. Of the 4 responders, 1 patient had a complete response, and 3 had a partial response. Two patients had stable disease, and 1 progressed.
In the overall study population, the most common adverse events were diarrhea (47%), nausea (45%), fatigue (37%), vomiting (27%), and neutropenia (21%). The most common grade 3/4 adverse events were neutropenia (18%) and anemia (5%).
About orphan designation
The FDA grants orphan designation to drugs and biologics intended to
treat, diagnose, or prevent rare diseases/disorders affecting fewer than
200,000 people in the US.
Orphan designation provides companies
with certain incentives to develop products for rare diseases. This
includes a 50% tax break on research and development, a fee waiver,
access to federal grants, and 7 years of market exclusivity if the
product is approved. ![]()

The US Food and Drug Administration (FDA) has granted orphan drug designation for the combination of TG-1101 (ublituximab), an anti-CD20 monoclonal antibody, and TGR-1202, a PI3K delta inhibitor, in the treatment of diffuse large B-cell lymphoma (DLBCL).
The combination is currently being evaluated in patients with relapsed or refractory DLBCL in the phase 2b UNITY-DLBCL trial.
Ublituximab and TGR-1202 are both products of TG Therapeutics, Inc.
Updated results from a phase 1 study of ublituximab and TGR-1202 in patients with DLBCL and other malignancies were presented at the 21st Congress of the European Hematology Association.
The data included 165 patients treated with varying doses of TGR-1202 alone (n=90) or in combination with ublituximab (n=75). The patients were heavily pretreated, with the majority having 3 or more prior lines of therapy.
There were 7 evaluable patients with DLBCL who received the combination at the phase 3 doses— ublituximab at 900 mg and TGR-1202 at 800 mg micronized.
The overall response rate for this group was 57%. Of the 4 responders, 1 patient had a complete response, and 3 had a partial response. Two patients had stable disease, and 1 progressed.
In the overall study population, the most common adverse events were diarrhea (47%), nausea (45%), fatigue (37%), vomiting (27%), and neutropenia (21%). The most common grade 3/4 adverse events were neutropenia (18%) and anemia (5%).
About orphan designation
The FDA grants orphan designation to drugs and biologics intended to
treat, diagnose, or prevent rare diseases/disorders affecting fewer than
200,000 people in the US.
Orphan designation provides companies
with certain incentives to develop products for rare diseases. This
includes a 50% tax break on research and development, a fee waiver,
access to federal grants, and 7 years of market exclusivity if the
product is approved. ![]()

The US Food and Drug Administration (FDA) has granted orphan drug designation for the combination of TG-1101 (ublituximab), an anti-CD20 monoclonal antibody, and TGR-1202, a PI3K delta inhibitor, in the treatment of diffuse large B-cell lymphoma (DLBCL).
The combination is currently being evaluated in patients with relapsed or refractory DLBCL in the phase 2b UNITY-DLBCL trial.
Ublituximab and TGR-1202 are both products of TG Therapeutics, Inc.
Updated results from a phase 1 study of ublituximab and TGR-1202 in patients with DLBCL and other malignancies were presented at the 21st Congress of the European Hematology Association.
The data included 165 patients treated with varying doses of TGR-1202 alone (n=90) or in combination with ublituximab (n=75). The patients were heavily pretreated, with the majority having 3 or more prior lines of therapy.
There were 7 evaluable patients with DLBCL who received the combination at the phase 3 doses— ublituximab at 900 mg and TGR-1202 at 800 mg micronized.
The overall response rate for this group was 57%. Of the 4 responders, 1 patient had a complete response, and 3 had a partial response. Two patients had stable disease, and 1 progressed.
In the overall study population, the most common adverse events were diarrhea (47%), nausea (45%), fatigue (37%), vomiting (27%), and neutropenia (21%). The most common grade 3/4 adverse events were neutropenia (18%) and anemia (5%).
About orphan designation
The FDA grants orphan designation to drugs and biologics intended to
treat, diagnose, or prevent rare diseases/disorders affecting fewer than
200,000 people in the US.
Orphan designation provides companies
with certain incentives to develop products for rare diseases. This
includes a 50% tax break on research and development, a fee waiver,
access to federal grants, and 7 years of market exclusivity if the
product is approved. ![]()
Distress linked to higher risk of death from leukemia, other cancers

patient and her father
Photo by Rhoda Baer
A study published in The BMJ suggests that higher levels of psychological distress (anxiety and depression) may be associated with an increased risk of death from leukemia and other cancers.
The findings are observational, so no firm conclusions about cause and effect can be drawn.
However, the researchers said the findings add to the growing evidence that psychological distress could have some predictive capacity for certain physical conditions.
There is some evidence that psychological distress (anxiety and depression) is related to increased rates of cardiovascular disease, but links with different types of cancer are either unclear or untested.
So David Batty, PhD, of University College London in the UK, and his colleagues set out to examine if psychological distress is a potential predictor of site-specific cancer mortality.
The researchers analyzed data from 16 studies (13 from England and 3 from Scotland), which started between 1994 and 2008. The data included 163,363 men and women age 16 or over who were free from cancer at the start of the study.
Psychological distress scores were measured using the general health questionnaire (GHQ-12), and participants were monitored for an average of 9.5 years. During this time, there were 4353 deaths from cancer.
Several factors that could have influenced the results were taken into account, including age, sex, education, socioeconomic status, body mass index, smoking, and alcohol intake.
“After statistical control for these factors, the results show that, compared with people in the least distressed group, death rates in the most distressed group were consistently higher for cancer of the bowel, prostate, pancreas, and esophagus and for leukemia,” Dr Batty said.
He and his colleagues pointed out that this association may also be affected by reverse causality, where undiagnosed (early) cancer might have had an underlying impact on mood.
In a bid to correct for this, the team conducted a further analysis excluding study participants who died in the first 5 years of follow-up, but this made no difference to the findings. The links between distress and cancer remained.
Specifically, compared to people in the least distressed group (GHQ-12 score 0-6), death rates in the most distressed group (score 7-12) were consistently increased for cancer of all sites combined, with a multivariable adjusted hazard ratio (HR) of 1.32.
Death rates were also increased for colorectal (HR=1.84), prostate (HR=2.42), pancreatic (HR=2.76), and esophageal cancers (HR=2.59), as well as for leukemia (HR=3.86).
“Our findings contribute to the evidence that poor mental health might have some predictive capacity for certain physical diseases,” Dr Batty said, “but we are a long way off from knowing if these relationships are truly causal.” ![]()

patient and her father
Photo by Rhoda Baer
A study published in The BMJ suggests that higher levels of psychological distress (anxiety and depression) may be associated with an increased risk of death from leukemia and other cancers.
The findings are observational, so no firm conclusions about cause and effect can be drawn.
However, the researchers said the findings add to the growing evidence that psychological distress could have some predictive capacity for certain physical conditions.
There is some evidence that psychological distress (anxiety and depression) is related to increased rates of cardiovascular disease, but links with different types of cancer are either unclear or untested.
So David Batty, PhD, of University College London in the UK, and his colleagues set out to examine if psychological distress is a potential predictor of site-specific cancer mortality.
The researchers analyzed data from 16 studies (13 from England and 3 from Scotland), which started between 1994 and 2008. The data included 163,363 men and women age 16 or over who were free from cancer at the start of the study.
Psychological distress scores were measured using the general health questionnaire (GHQ-12), and participants were monitored for an average of 9.5 years. During this time, there were 4353 deaths from cancer.
Several factors that could have influenced the results were taken into account, including age, sex, education, socioeconomic status, body mass index, smoking, and alcohol intake.
“After statistical control for these factors, the results show that, compared with people in the least distressed group, death rates in the most distressed group were consistently higher for cancer of the bowel, prostate, pancreas, and esophagus and for leukemia,” Dr Batty said.
He and his colleagues pointed out that this association may also be affected by reverse causality, where undiagnosed (early) cancer might have had an underlying impact on mood.
In a bid to correct for this, the team conducted a further analysis excluding study participants who died in the first 5 years of follow-up, but this made no difference to the findings. The links between distress and cancer remained.
Specifically, compared to people in the least distressed group (GHQ-12 score 0-6), death rates in the most distressed group (score 7-12) were consistently increased for cancer of all sites combined, with a multivariable adjusted hazard ratio (HR) of 1.32.
Death rates were also increased for colorectal (HR=1.84), prostate (HR=2.42), pancreatic (HR=2.76), and esophageal cancers (HR=2.59), as well as for leukemia (HR=3.86).
“Our findings contribute to the evidence that poor mental health might have some predictive capacity for certain physical diseases,” Dr Batty said, “but we are a long way off from knowing if these relationships are truly causal.” ![]()

patient and her father
Photo by Rhoda Baer
A study published in The BMJ suggests that higher levels of psychological distress (anxiety and depression) may be associated with an increased risk of death from leukemia and other cancers.
The findings are observational, so no firm conclusions about cause and effect can be drawn.
However, the researchers said the findings add to the growing evidence that psychological distress could have some predictive capacity for certain physical conditions.
There is some evidence that psychological distress (anxiety and depression) is related to increased rates of cardiovascular disease, but links with different types of cancer are either unclear or untested.
So David Batty, PhD, of University College London in the UK, and his colleagues set out to examine if psychological distress is a potential predictor of site-specific cancer mortality.
The researchers analyzed data from 16 studies (13 from England and 3 from Scotland), which started between 1994 and 2008. The data included 163,363 men and women age 16 or over who were free from cancer at the start of the study.
Psychological distress scores were measured using the general health questionnaire (GHQ-12), and participants were monitored for an average of 9.5 years. During this time, there were 4353 deaths from cancer.
Several factors that could have influenced the results were taken into account, including age, sex, education, socioeconomic status, body mass index, smoking, and alcohol intake.
“After statistical control for these factors, the results show that, compared with people in the least distressed group, death rates in the most distressed group were consistently higher for cancer of the bowel, prostate, pancreas, and esophagus and for leukemia,” Dr Batty said.
He and his colleagues pointed out that this association may also be affected by reverse causality, where undiagnosed (early) cancer might have had an underlying impact on mood.
In a bid to correct for this, the team conducted a further analysis excluding study participants who died in the first 5 years of follow-up, but this made no difference to the findings. The links between distress and cancer remained.
Specifically, compared to people in the least distressed group (GHQ-12 score 0-6), death rates in the most distressed group (score 7-12) were consistently increased for cancer of all sites combined, with a multivariable adjusted hazard ratio (HR) of 1.32.
Death rates were also increased for colorectal (HR=1.84), prostate (HR=2.42), pancreatic (HR=2.76), and esophageal cancers (HR=2.59), as well as for leukemia (HR=3.86).
“Our findings contribute to the evidence that poor mental health might have some predictive capacity for certain physical diseases,” Dr Batty said, “but we are a long way off from knowing if these relationships are truly causal.” ![]()
Why young hemophiliacs skip prophylaxis

Some young hemophilia patients may not be sticking to their prophylaxis regimens for psychosocial reasons, according to research published in PLOS ONE.
The study included 78 hemophilia patients, ages 12 to 25, treated at 13 centers in England and Wales.
“This research has a specific focus on young people rather than including patients of all age groups,” said study author Sandra Van Os, a PhD student at the University of Hertfordshire in Hatfield, UK.
“This is important since young people are likely to have had a very different experience than the older generations due to the revolutionary improvements in treatment achieved during the past 2 decades. Another key issue is rather than asking parents or healthcare professionals to estimate adherence as previous studies have done, this study asked young people directly.”
The results showed that 18% of the patients did not adhere to their prophylaxis regimens. Most non-adherence was due to forgetting treatment rather than intentionally skipping it.
But there were a few psychosocial reasons for non-adherence. Patients reported non-adherence due to a lack of social support and the perceived impact of prophylaxis on their everyday lives.
Patients also lacked understanding about the effectiveness of prophylaxis and its importance in treating hemophilia.
The patients were more likely to adhere to treatment when they perceived the need for prophylaxis to be greater than their concern over taking it.
And the study suggested a strong emotional reaction to a bleed may encourage young people to adhere to their treatment regimen.
“Interestingly, the findings suggest that, in addition to social support and treatment beliefs, emotional responses in relation to hemophilia, such as fear, anger, or distress, may also contribute to better adherence,” van Os said.
“It is important that patients receive sufficient and appropriate social support in order to stay on track with their treatment. It will also be beneficial to reduce potential concerns about prophylaxis and to assess whether patients understand their treatment sufficiently well and the role they themselves have to play in its effectiveness.”
This research was supported by external, peer-reviewed funding from the Bayer Haemophilia Awards Programme (Caregiver Award awarded to van Os). ![]()

Some young hemophilia patients may not be sticking to their prophylaxis regimens for psychosocial reasons, according to research published in PLOS ONE.
The study included 78 hemophilia patients, ages 12 to 25, treated at 13 centers in England and Wales.
“This research has a specific focus on young people rather than including patients of all age groups,” said study author Sandra Van Os, a PhD student at the University of Hertfordshire in Hatfield, UK.
“This is important since young people are likely to have had a very different experience than the older generations due to the revolutionary improvements in treatment achieved during the past 2 decades. Another key issue is rather than asking parents or healthcare professionals to estimate adherence as previous studies have done, this study asked young people directly.”
The results showed that 18% of the patients did not adhere to their prophylaxis regimens. Most non-adherence was due to forgetting treatment rather than intentionally skipping it.
But there were a few psychosocial reasons for non-adherence. Patients reported non-adherence due to a lack of social support and the perceived impact of prophylaxis on their everyday lives.
Patients also lacked understanding about the effectiveness of prophylaxis and its importance in treating hemophilia.
The patients were more likely to adhere to treatment when they perceived the need for prophylaxis to be greater than their concern over taking it.
And the study suggested a strong emotional reaction to a bleed may encourage young people to adhere to their treatment regimen.
“Interestingly, the findings suggest that, in addition to social support and treatment beliefs, emotional responses in relation to hemophilia, such as fear, anger, or distress, may also contribute to better adherence,” van Os said.
“It is important that patients receive sufficient and appropriate social support in order to stay on track with their treatment. It will also be beneficial to reduce potential concerns about prophylaxis and to assess whether patients understand their treatment sufficiently well and the role they themselves have to play in its effectiveness.”
This research was supported by external, peer-reviewed funding from the Bayer Haemophilia Awards Programme (Caregiver Award awarded to van Os). ![]()

Some young hemophilia patients may not be sticking to their prophylaxis regimens for psychosocial reasons, according to research published in PLOS ONE.
The study included 78 hemophilia patients, ages 12 to 25, treated at 13 centers in England and Wales.
“This research has a specific focus on young people rather than including patients of all age groups,” said study author Sandra Van Os, a PhD student at the University of Hertfordshire in Hatfield, UK.
“This is important since young people are likely to have had a very different experience than the older generations due to the revolutionary improvements in treatment achieved during the past 2 decades. Another key issue is rather than asking parents or healthcare professionals to estimate adherence as previous studies have done, this study asked young people directly.”
The results showed that 18% of the patients did not adhere to their prophylaxis regimens. Most non-adherence was due to forgetting treatment rather than intentionally skipping it.
But there were a few psychosocial reasons for non-adherence. Patients reported non-adherence due to a lack of social support and the perceived impact of prophylaxis on their everyday lives.
Patients also lacked understanding about the effectiveness of prophylaxis and its importance in treating hemophilia.
The patients were more likely to adhere to treatment when they perceived the need for prophylaxis to be greater than their concern over taking it.
And the study suggested a strong emotional reaction to a bleed may encourage young people to adhere to their treatment regimen.
“Interestingly, the findings suggest that, in addition to social support and treatment beliefs, emotional responses in relation to hemophilia, such as fear, anger, or distress, may also contribute to better adherence,” van Os said.
“It is important that patients receive sufficient and appropriate social support in order to stay on track with their treatment. It will also be beneficial to reduce potential concerns about prophylaxis and to assess whether patients understand their treatment sufficiently well and the role they themselves have to play in its effectiveness.”
This research was supported by external, peer-reviewed funding from the Bayer Haemophilia Awards Programme (Caregiver Award awarded to van Os).
Donor CAR T cells bridge to HSCT in infants with ALL
treated with UCART19
Photo courtesy of GOSH
New research suggests that “universal,” donor-derived, chimeric antigen receptor (CAR) T cells may be a viable treatment option for very young children who do not have sufficient healthy T cells for autologous CAR T-cell therapy.
The universal CAR T-cell therapy, known as UCART19, was given to 2 infants with relapsed/refractory B-cell acute lymphoblastic leukemia (ALL) who had previously
exhausted all other treatment options.
Both infants achieved remission after UCART19 and were able to proceed to transplant.
Both were still alive and leukemia-free at last follow-up—12 months and 18 months after UCART19, respectively.
Waseem Qasim, MBBS, PhD, of University College London’s Institute of Child Health and Great Ormond Street Hospital (GOSH) in London, UK, and his colleagues reported these results in Science Translational Medicine.
About the therapy
UCART19 consists of donor T cells modified using transcription activator-like effector nucleases. The cells are programmed to target CD19 and be insensitive to alemtuzumab. That way, a patient can receive alemtuzumab to prevent rejection of HLA-mismatched cells.
UCART19 was under development by Cellectis but is now being developed by Servier and Pfizer Inc. Pfizer has exclusive rights to develop and commercialize UCART19 in the US, while Servier retains exclusive rights for all other countries.
In Science Translational Medicine, Dr Qasim and his colleagues reported results in the first patients ever treated with UCART19. That research was funded, in part, by Cellectis.
Subject 1
The first patient was an 11-month-old, mixed-race infant with high-risk, t(11;19), CD19+ B-ALL. She had already failed chemotherapy, an allogeneic hematopoietic stem cell transplant (HSCT), and blinatumomab.
Prior to UCART19, she received a dose of vincristine and asparaginase and 7 days of dexamethasone, followed by cytoreduction with fludarabine, cyclophosphamide, and alemtuzumab. She then received a single dose of UCART19 (4.6 × 106/kg).
The patient had neutrophil recovery by day 30, although this was dependent on granulocyte colony-stimulating factor. After that, she developed multilineage cytopenia that persisted until she underwent a second allogeneic HSCT.
Prior to the second HSCT, the patient achieved cytogenetic and molecular remission but also developed grade 2 skin graft-vs-host disease. This was managed with systemic steroids.
The child received rituximab and conditioning with antithymocyte globulin, fludarabine, cyclophosphamide, thiotepa, and total body irradiation, followed by an HSCT from the original mismatched, unrelated donor.
The child achieved a complete remission and has remained minimal residual disease-negative, with full donor chimerism and normalized lymphocyte profiles, at 18 months after UCART19.
Results with this patient were previously reported by GOSH in November 2015 and at the 2015 ASH Annual Meeting.
Subject 2
The second patient was a 16-month-old Caucasian infant who had been diagnosed at 4 weeks of age with high-risk, congenital, mixed lineage leukemia–rearranged B-ALL.
She had already undergone HSCT from a matched, unrelated donor but relapsed. She did not respond to subsequent blinatumomab.
After these failed treatments, the patient received fludarabine, cyclophosphamide, and alemtuzumab, followed by a single infusion of UCART19 (4.0 × 106/kg).
She developed an erythematous rash after treatment, but this was immediately responsive to topical steroids.
The child achieved donor-derived neutrophil recovery and went on to receive a transplant from the same matched, unrelated donor as her previous HSCT.
The child received the transplant within 10 weeks of UCART19 therapy, after receiving rituximab and conditioning with antithymocyte globulin, fludarabine, cyclophosphamide, and total body irradiation.
At 12 months after UCART19, the child remains minimal residual disease-negative and “clinically well.”
“Both infants who have had this treatment have been at home for some time and are doing well,” Dr Qasim said. “We continue to monitor them closely, and, while we have reduced the frequency of their hospital visits and checks, we will still need to keep an eye on them for some time.”
Dr Qasim added that there are phase 1 trials of UCART19 underway for children and adults with chronic lymphocytic leukemia and acute lymphoblastic leukemia.

treated with UCART19
Photo courtesy of GOSH
New research suggests that “universal,” donor-derived, chimeric antigen receptor (CAR) T cells may be a viable treatment option for very young children who do not have sufficient healthy T cells for autologous CAR T-cell therapy.
The universal CAR T-cell therapy, known as UCART19, was given to 2 infants with relapsed/refractory B-cell acute lymphoblastic leukemia (ALL) who had previously
exhausted all other treatment options.
Both infants achieved remission after UCART19 and were able to proceed to transplant.
Both were still alive and leukemia-free at last follow-up—12 months and 18 months after UCART19, respectively.
Waseem Qasim, MBBS, PhD, of University College London’s Institute of Child Health and Great Ormond Street Hospital (GOSH) in London, UK, and his colleagues reported these results in Science Translational Medicine.
About the therapy
UCART19 consists of donor T cells modified using transcription activator-like effector nucleases. The cells are programmed to target CD19 and be insensitive to alemtuzumab. That way, a patient can receive alemtuzumab to prevent rejection of HLA-mismatched cells.
UCART19 was under development by Cellectis but is now being developed by Servier and Pfizer Inc. Pfizer has exclusive rights to develop and commercialize UCART19 in the US, while Servier retains exclusive rights for all other countries.
In Science Translational Medicine, Dr Qasim and his colleagues reported results in the first patients ever treated with UCART19. That research was funded, in part, by Cellectis.
Subject 1
The first patient was an 11-month-old, mixed-race infant with high-risk, t(11;19), CD19+ B-ALL. She had already failed chemotherapy, an allogeneic hematopoietic stem cell transplant (HSCT), and blinatumomab.
Prior to UCART19, she received a dose of vincristine and asparaginase and 7 days of dexamethasone, followed by cytoreduction with fludarabine, cyclophosphamide, and alemtuzumab. She then received a single dose of UCART19 (4.6 × 106/kg).
The patient had neutrophil recovery by day 30, although this was dependent on granulocyte colony-stimulating factor. After that, she developed multilineage cytopenia that persisted until she underwent a second allogeneic HSCT.
Prior to the second HSCT, the patient achieved cytogenetic and molecular remission but also developed grade 2 skin graft-vs-host disease. This was managed with systemic steroids.
The child received rituximab and conditioning with antithymocyte globulin, fludarabine, cyclophosphamide, thiotepa, and total body irradiation, followed by an HSCT from the original mismatched, unrelated donor.
The child achieved a complete remission and has remained minimal residual disease-negative, with full donor chimerism and normalized lymphocyte profiles, at 18 months after UCART19.
Results with this patient were previously reported by GOSH in November 2015 and at the 2015 ASH Annual Meeting.
Subject 2
The second patient was a 16-month-old Caucasian infant who had been diagnosed at 4 weeks of age with high-risk, congenital, mixed lineage leukemia–rearranged B-ALL.
She had already undergone HSCT from a matched, unrelated donor but relapsed. She did not respond to subsequent blinatumomab.
After these failed treatments, the patient received fludarabine, cyclophosphamide, and alemtuzumab, followed by a single infusion of UCART19 (4.0 × 106/kg).
She developed an erythematous rash after treatment, but this was immediately responsive to topical steroids.
The child achieved donor-derived neutrophil recovery and went on to receive a transplant from the same matched, unrelated donor as her previous HSCT.
The child received the transplant within 10 weeks of UCART19 therapy, after receiving rituximab and conditioning with antithymocyte globulin, fludarabine, cyclophosphamide, and total body irradiation.
At 12 months after UCART19, the child remains minimal residual disease-negative and “clinically well.”
“Both infants who have had this treatment have been at home for some time and are doing well,” Dr Qasim said. “We continue to monitor them closely, and, while we have reduced the frequency of their hospital visits and checks, we will still need to keep an eye on them for some time.”
Dr Qasim added that there are phase 1 trials of UCART19 underway for children and adults with chronic lymphocytic leukemia and acute lymphoblastic leukemia.

treated with UCART19
Photo courtesy of GOSH
New research suggests that “universal,” donor-derived, chimeric antigen receptor (CAR) T cells may be a viable treatment option for very young children who do not have sufficient healthy T cells for autologous CAR T-cell therapy.
The universal CAR T-cell therapy, known as UCART19, was given to 2 infants with relapsed/refractory B-cell acute lymphoblastic leukemia (ALL) who had previously
exhausted all other treatment options.
Both infants achieved remission after UCART19 and were able to proceed to transplant.
Both were still alive and leukemia-free at last follow-up—12 months and 18 months after UCART19, respectively.
Waseem Qasim, MBBS, PhD, of University College London’s Institute of Child Health and Great Ormond Street Hospital (GOSH) in London, UK, and his colleagues reported these results in Science Translational Medicine.
About the therapy
UCART19 consists of donor T cells modified using transcription activator-like effector nucleases. The cells are programmed to target CD19 and be insensitive to alemtuzumab. That way, a patient can receive alemtuzumab to prevent rejection of HLA-mismatched cells.
UCART19 was under development by Cellectis but is now being developed by Servier and Pfizer Inc. Pfizer has exclusive rights to develop and commercialize UCART19 in the US, while Servier retains exclusive rights for all other countries.
In Science Translational Medicine, Dr Qasim and his colleagues reported results in the first patients ever treated with UCART19. That research was funded, in part, by Cellectis.
Subject 1
The first patient was an 11-month-old, mixed-race infant with high-risk, t(11;19), CD19+ B-ALL. She had already failed chemotherapy, an allogeneic hematopoietic stem cell transplant (HSCT), and blinatumomab.
Prior to UCART19, she received a dose of vincristine and asparaginase and 7 days of dexamethasone, followed by cytoreduction with fludarabine, cyclophosphamide, and alemtuzumab. She then received a single dose of UCART19 (4.6 × 106/kg).
The patient had neutrophil recovery by day 30, although this was dependent on granulocyte colony-stimulating factor. After that, she developed multilineage cytopenia that persisted until she underwent a second allogeneic HSCT.
Prior to the second HSCT, the patient achieved cytogenetic and molecular remission but also developed grade 2 skin graft-vs-host disease. This was managed with systemic steroids.
The child received rituximab and conditioning with antithymocyte globulin, fludarabine, cyclophosphamide, thiotepa, and total body irradiation, followed by an HSCT from the original mismatched, unrelated donor.
The child achieved a complete remission and has remained minimal residual disease-negative, with full donor chimerism and normalized lymphocyte profiles, at 18 months after UCART19.
Results with this patient were previously reported by GOSH in November 2015 and at the 2015 ASH Annual Meeting.
Subject 2
The second patient was a 16-month-old Caucasian infant who had been diagnosed at 4 weeks of age with high-risk, congenital, mixed lineage leukemia–rearranged B-ALL.
She had already undergone HSCT from a matched, unrelated donor but relapsed. She did not respond to subsequent blinatumomab.
After these failed treatments, the patient received fludarabine, cyclophosphamide, and alemtuzumab, followed by a single infusion of UCART19 (4.0 × 106/kg).
She developed an erythematous rash after treatment, but this was immediately responsive to topical steroids.
The child achieved donor-derived neutrophil recovery and went on to receive a transplant from the same matched, unrelated donor as her previous HSCT.
The child received the transplant within 10 weeks of UCART19 therapy, after receiving rituximab and conditioning with antithymocyte globulin, fludarabine, cyclophosphamide, and total body irradiation.
At 12 months after UCART19, the child remains minimal residual disease-negative and “clinically well.”
“Both infants who have had this treatment have been at home for some time and are doing well,” Dr Qasim said. “We continue to monitor them closely, and, while we have reduced the frequency of their hospital visits and checks, we will still need to keep an eye on them for some time.”
Dr Qasim added that there are phase 1 trials of UCART19 underway for children and adults with chronic lymphocytic leukemia and acute lymphoblastic leukemia.
Roche launches new blood analyzer

Photo by Graham Colm
Roche has announced the launch of its cobas m 511 integrated hematology analyzer in countries that recognize the CE mark.*
The cobas m 511 combines 3 components of the hematology testing process—a digital morphology analyzer, cell counter, and classifier—into a single system that prepares, stains, and analyzes microscopy blood slides.
Roche said cobas m 511 provides greater accuracy and consistency than current technologies by identifying, counting, isolating, and categorizing blood cells, then presenting the digital images of all these cell types.
The company said this automation and digitalization reduces the need for resource-intensive manual microscope reviews, supports clinicians to share challenging cases around the world, and enables the delivery of quicker results, which ultimately aid patient diagnoses.
The cobas m 511 uses Bloodhound® technology for printing, staining, and imaging. This technology uses 30 µL of blood to print a monolayer onto the slide, stains for further analysis of the morphology, and enables classification of cells displayed on a viewing station.
Unlike the indirect methods commonly used in blood analysis today, the cobas m 511 images individual cells directly.
Based on these direct images, the Bloodhound® technology counts, analyzes morphology, and then classifies every cell in the viewing area to provide a standard complete blood count and 5-part differential and reticulocyte count.
While hematologists will continue to have the option of looking at slides under their microscopes, the cobas m 511 provides cell-by-cell images that, in many cases, may eliminate the need for microscopic review.
“With this launch, patients will benefit from a faster and more accurate diagnosis of blood diseases as diverse as anemia and leukemia,” said Roland Diggelmann, CEO of Roche Diagnostics.
“We are entering a new area of innovation with Roche in hematology testing, supporting customers with integrated and efficient laboratory solutions, which deliver increased medical value.”
*Local product availability may vary independently from CE mark approval. The cobas m 511 integrated hematology analyzer is not available in countries with previously agreed third-party vendor agreements.

Photo by Graham Colm
Roche has announced the launch of its cobas m 511 integrated hematology analyzer in countries that recognize the CE mark.*
The cobas m 511 combines 3 components of the hematology testing process—a digital morphology analyzer, cell counter, and classifier—into a single system that prepares, stains, and analyzes microscopy blood slides.
Roche said cobas m 511 provides greater accuracy and consistency than current technologies by identifying, counting, isolating, and categorizing blood cells, then presenting the digital images of all these cell types.
The company said this automation and digitalization reduces the need for resource-intensive manual microscope reviews, supports clinicians to share challenging cases around the world, and enables the delivery of quicker results, which ultimately aid patient diagnoses.
The cobas m 511 uses Bloodhound® technology for printing, staining, and imaging. This technology uses 30 µL of blood to print a monolayer onto the slide, stains for further analysis of the morphology, and enables classification of cells displayed on a viewing station.
Unlike the indirect methods commonly used in blood analysis today, the cobas m 511 images individual cells directly.
Based on these direct images, the Bloodhound® technology counts, analyzes morphology, and then classifies every cell in the viewing area to provide a standard complete blood count and 5-part differential and reticulocyte count.
While hematologists will continue to have the option of looking at slides under their microscopes, the cobas m 511 provides cell-by-cell images that, in many cases, may eliminate the need for microscopic review.
“With this launch, patients will benefit from a faster and more accurate diagnosis of blood diseases as diverse as anemia and leukemia,” said Roland Diggelmann, CEO of Roche Diagnostics.
“We are entering a new area of innovation with Roche in hematology testing, supporting customers with integrated and efficient laboratory solutions, which deliver increased medical value.”
*Local product availability may vary independently from CE mark approval. The cobas m 511 integrated hematology analyzer is not available in countries with previously agreed third-party vendor agreements.

Photo by Graham Colm
Roche has announced the launch of its cobas m 511 integrated hematology analyzer in countries that recognize the CE mark.*
The cobas m 511 combines 3 components of the hematology testing process—a digital morphology analyzer, cell counter, and classifier—into a single system that prepares, stains, and analyzes microscopy blood slides.
Roche said cobas m 511 provides greater accuracy and consistency than current technologies by identifying, counting, isolating, and categorizing blood cells, then presenting the digital images of all these cell types.
The company said this automation and digitalization reduces the need for resource-intensive manual microscope reviews, supports clinicians to share challenging cases around the world, and enables the delivery of quicker results, which ultimately aid patient diagnoses.
The cobas m 511 uses Bloodhound® technology for printing, staining, and imaging. This technology uses 30 µL of blood to print a monolayer onto the slide, stains for further analysis of the morphology, and enables classification of cells displayed on a viewing station.
Unlike the indirect methods commonly used in blood analysis today, the cobas m 511 images individual cells directly.
Based on these direct images, the Bloodhound® technology counts, analyzes morphology, and then classifies every cell in the viewing area to provide a standard complete blood count and 5-part differential and reticulocyte count.
While hematologists will continue to have the option of looking at slides under their microscopes, the cobas m 511 provides cell-by-cell images that, in many cases, may eliminate the need for microscopic review.
“With this launch, patients will benefit from a faster and more accurate diagnosis of blood diseases as diverse as anemia and leukemia,” said Roland Diggelmann, CEO of Roche Diagnostics.
“We are entering a new area of innovation with Roche in hematology testing, supporting customers with integrated and efficient laboratory solutions, which deliver increased medical value.”
*Local product availability may vary independently from CE mark approval. The cobas m 511 integrated hematology analyzer is not available in countries with previously agreed third-party vendor agreements.
Anticoagulants often unnecessary after surgery, analysis suggests
Many surgical patients may be receiving anticoagulants they don’t need, according to research published in Annals of Surgery.
The study challenges standard of care guidelines, which recommend that all general surgery patients receive treatment to prevent venous thromboembolism (VTE).
The new findings suggest that anticoagulants may be unnecessary for most surgical patients and could even be harmful to some.
“A ‘one-size-fits-all approach’ doesn’t always make sense,” said study author Christopher Pannucci, MD, of the University of Utah in Salt Lake City.
“A healthy 35-year-old is very different from someone who is 85 and has a history of clots. Our research indicates that there could be a substantial number of people who are being over-treated.”
Dr Pannucci and his colleagues reviewed data from 13 studies to determine which surgical patients were most likely, and least likely, to benefit from anticoagulants. There was data on VTE events in 11 studies (n=14,776) and data on clinically relevant bleeding in 8 studies (n=7590).
In most of the studies, patients received mechanical VTE prophylaxis, which meant elastic compression and/or sequential compression devices.
Some studies compared mechanical prophylaxis to anticoagulants, including heparin, low-molecular-weight heparin, direct factor Xa inhibitors, direct thrombin inhibitors, warfarin, dextran, and aspirin.
The studies included a broad range of surgical patients, from individuals with few VTE risk factors to those with multiple risk factors, such as obesity, advanced age, and personal or family history of VTE.
The patients were divided into 1 of 5 categories indicating overall VTE risk. Assessment was based on the Caprini score.
VTE risk without anticoagulant treatment
There were 11 studies in which some patients did not receive anticoagulants (n=6085).
Among these patients, those who were classified as having the highest risk of VTE were 14 times more likely to develop VTE than patients in the low-risk category—10.7% vs 0.7%.
These findings were independent of surgery type.
“It was eye-opening to see that there is this huge variability in risk among the overall group of patients that walk into your office,” Dr Pannucci said. “Unless you consider a patient’s risk based on their individual factors, you would never know.”
VTE outcomes by risk score
When given, anticoagulants did significantly reduce the risk of VTE for the overall study population and for high-risk patients.
The odds ratios (ORs) were 0.66 (P=0.001) for the overall population, 0.60 (P=0.04) for patients with Caprini scores of 7 to 8, and 0.41 (P=0.0002) for patients with scores higher than 8.
Unfortunately, anticoagulants did not make a significant difference in VTE rates for mid- or low-risk patients.
The ORs were 0.45 (P=0.31) for patients with Caprini scores of 0 to 2, 1.31 (P=0.57) for patients with scores of 3 to 4, and 0.96 (P=0.85) for patients with scores of 5 to 6.
Risk of bleeding
Anticoagulants significantly increased clinically relevant bleeding for the overall population. The OR was 1.69 (P=0.006).
Patients who received anticoagulants were not significantly more likely to have clinically relevant bleeding if they had risk scores of 0 to 2 (OR=2.47, P=0.61), 3 to 4 (OR=1.05, P=0.87), 5 to 6 (OR=2.10, P=0.06), 7 to 8 (OR=3.15, P=0.16), or >8 (OR=2.31, P=0.16).
“For the first time, we have data that prophylaxis for the highest-risk groups is beneficial, and data that suggests that lower-risk patients may need no prophylaxis,” said study author Peter Henke, MD, of the University of Michigan in Ann Arbor.
He and his colleagues noted, however, that prospective studies are needed to confirm these findings.

Many surgical patients may be receiving anticoagulants they don’t need, according to research published in Annals of Surgery.
The study challenges standard of care guidelines, which recommend that all general surgery patients receive treatment to prevent venous thromboembolism (VTE).
The new findings suggest that anticoagulants may be unnecessary for most surgical patients and could even be harmful to some.
“A ‘one-size-fits-all approach’ doesn’t always make sense,” said study author Christopher Pannucci, MD, of the University of Utah in Salt Lake City.
“A healthy 35-year-old is very different from someone who is 85 and has a history of clots. Our research indicates that there could be a substantial number of people who are being over-treated.”
Dr Pannucci and his colleagues reviewed data from 13 studies to determine which surgical patients were most likely, and least likely, to benefit from anticoagulants. There was data on VTE events in 11 studies (n=14,776) and data on clinically relevant bleeding in 8 studies (n=7590).
In most of the studies, patients received mechanical VTE prophylaxis, which meant elastic compression and/or sequential compression devices.
Some studies compared mechanical prophylaxis to anticoagulants, including heparin, low-molecular-weight heparin, direct factor Xa inhibitors, direct thrombin inhibitors, warfarin, dextran, and aspirin.
The studies included a broad range of surgical patients, from individuals with few VTE risk factors to those with multiple risk factors, such as obesity, advanced age, and personal or family history of VTE.
The patients were divided into 1 of 5 categories indicating overall VTE risk. Assessment was based on the Caprini score.
VTE risk without anticoagulant treatment
There were 11 studies in which some patients did not receive anticoagulants (n=6085).
Among these patients, those who were classified as having the highest risk of VTE were 14 times more likely to develop VTE than patients in the low-risk category—10.7% vs 0.7%.
These findings were independent of surgery type.
“It was eye-opening to see that there is this huge variability in risk among the overall group of patients that walk into your office,” Dr Pannucci said. “Unless you consider a patient’s risk based on their individual factors, you would never know.”
VTE outcomes by risk score
When given, anticoagulants did significantly reduce the risk of VTE for the overall study population and for high-risk patients.
The odds ratios (ORs) were 0.66 (P=0.001) for the overall population, 0.60 (P=0.04) for patients with Caprini scores of 7 to 8, and 0.41 (P=0.0002) for patients with scores higher than 8.
Unfortunately, anticoagulants did not make a significant difference in VTE rates for mid- or low-risk patients.
The ORs were 0.45 (P=0.31) for patients with Caprini scores of 0 to 2, 1.31 (P=0.57) for patients with scores of 3 to 4, and 0.96 (P=0.85) for patients with scores of 5 to 6.
Risk of bleeding
Anticoagulants significantly increased clinically relevant bleeding for the overall population. The OR was 1.69 (P=0.006).
Patients who received anticoagulants were not significantly more likely to have clinically relevant bleeding if they had risk scores of 0 to 2 (OR=2.47, P=0.61), 3 to 4 (OR=1.05, P=0.87), 5 to 6 (OR=2.10, P=0.06), 7 to 8 (OR=3.15, P=0.16), or >8 (OR=2.31, P=0.16).
“For the first time, we have data that prophylaxis for the highest-risk groups is beneficial, and data that suggests that lower-risk patients may need no prophylaxis,” said study author Peter Henke, MD, of the University of Michigan in Ann Arbor.
He and his colleagues noted, however, that prospective studies are needed to confirm these findings.

Many surgical patients may be receiving anticoagulants they don’t need, according to research published in Annals of Surgery.
The study challenges standard of care guidelines, which recommend that all general surgery patients receive treatment to prevent venous thromboembolism (VTE).
The new findings suggest that anticoagulants may be unnecessary for most surgical patients and could even be harmful to some.
“A ‘one-size-fits-all approach’ doesn’t always make sense,” said study author Christopher Pannucci, MD, of the University of Utah in Salt Lake City.
“A healthy 35-year-old is very different from someone who is 85 and has a history of clots. Our research indicates that there could be a substantial number of people who are being over-treated.”
Dr Pannucci and his colleagues reviewed data from 13 studies to determine which surgical patients were most likely, and least likely, to benefit from anticoagulants. There was data on VTE events in 11 studies (n=14,776) and data on clinically relevant bleeding in 8 studies (n=7590).
In most of the studies, patients received mechanical VTE prophylaxis, which meant elastic compression and/or sequential compression devices.
Some studies compared mechanical prophylaxis to anticoagulants, including heparin, low-molecular-weight heparin, direct factor Xa inhibitors, direct thrombin inhibitors, warfarin, dextran, and aspirin.
The studies included a broad range of surgical patients, from individuals with few VTE risk factors to those with multiple risk factors, such as obesity, advanced age, and personal or family history of VTE.
The patients were divided into 1 of 5 categories indicating overall VTE risk. Assessment was based on the Caprini score.
VTE risk without anticoagulant treatment
There were 11 studies in which some patients did not receive anticoagulants (n=6085).
Among these patients, those who were classified as having the highest risk of VTE were 14 times more likely to develop VTE than patients in the low-risk category—10.7% vs 0.7%.
These findings were independent of surgery type.
“It was eye-opening to see that there is this huge variability in risk among the overall group of patients that walk into your office,” Dr Pannucci said. “Unless you consider a patient’s risk based on their individual factors, you would never know.”
VTE outcomes by risk score
When given, anticoagulants did significantly reduce the risk of VTE for the overall study population and for high-risk patients.
The odds ratios (ORs) were 0.66 (P=0.001) for the overall population, 0.60 (P=0.04) for patients with Caprini scores of 7 to 8, and 0.41 (P=0.0002) for patients with scores higher than 8.
Unfortunately, anticoagulants did not make a significant difference in VTE rates for mid- or low-risk patients.
The ORs were 0.45 (P=0.31) for patients with Caprini scores of 0 to 2, 1.31 (P=0.57) for patients with scores of 3 to 4, and 0.96 (P=0.85) for patients with scores of 5 to 6.
Risk of bleeding
Anticoagulants significantly increased clinically relevant bleeding for the overall population. The OR was 1.69 (P=0.006).
Patients who received anticoagulants were not significantly more likely to have clinically relevant bleeding if they had risk scores of 0 to 2 (OR=2.47, P=0.61), 3 to 4 (OR=1.05, P=0.87), 5 to 6 (OR=2.10, P=0.06), 7 to 8 (OR=3.15, P=0.16), or >8 (OR=2.31, P=0.16).
“For the first time, we have data that prophylaxis for the highest-risk groups is beneficial, and data that suggests that lower-risk patients may need no prophylaxis,” said study author Peter Henke, MD, of the University of Michigan in Ann Arbor.
He and his colleagues noted, however, that prospective studies are needed to confirm these findings.