User login
Combo appears effective against B-ALL

Combining a MEK inhibitor and a BCL-2/BCL-XL inhibitor may be a feasible treatment option for B-cell acute lymphoblastic leukemia (B-ALL), according to preclinical research published in Cell Death and Disease.
Researchers found that, when given alone, the MEK inhibitor trametinib did not block B-ALL cell growth.
And the BCL-2/BCL-XL inhibitors navitoclax (ABT-263) and venetoclax (ABT-199) did not prove particularly effective either.
However, combining trametinib with navitoclax or venetoclax successfully induced apoptosis in B-ALL cells.
“Cancer cells often outwit us by rewiring themselves, but this early research offers a promising idea to get ahead of them,” said study author Richard Marais, PhD, of the Cancer Research UK Manchester Institute.
“We’ll still need to do further research to prove that this is the case beyond cancer cells in the laboratory, and it may take many years before we see it in the clinic, but it’s the first step to finding a new, effective drug combination for B-cell acute lymphoblastic leukemia.”
Dr Marais and his colleagues found that, although the MEK/ERK pathway is activated in B-ALL cells driven by different oncogenes, MEK inhibition alone did not suppress B-ALL cell growth.
And although B-ALL cells were sensitive to treatment with navitoclax or venetoclax alone, the researchers did not see complete loss of cell viability at clinically achievable doses.
However, trametinib did synergize with either navitoclax or venetoclax to suppress proliferation and induce apoptosis in B-ALL cells.
Further investigation revealed that the resistance of B-ALL cells to BCL-2/BCL-XL inhibition is mediated by MCL-1. And the synergism between trametinib and navitoclax/venetoclax is mediated by the pro-apoptotic factor BIM.
BIM is dephosphorylated as a result of MEK inhibition, which allows it to bind to and neutralize MCL-1, thereby enhancing BCL-2/BCL-XL inhibitor-induced cell death.
The researchers said they observed this effect in B-ALL cells driven by a range of genetic abnormalities, so the combination of a MEK inhibitor and a BCL-2/BCL-XL inhibitor could have therapeutic potential in a range of B-ALL subtypes. ![]()

Combining a MEK inhibitor and a BCL-2/BCL-XL inhibitor may be a feasible treatment option for B-cell acute lymphoblastic leukemia (B-ALL), according to preclinical research published in Cell Death and Disease.
Researchers found that, when given alone, the MEK inhibitor trametinib did not block B-ALL cell growth.
And the BCL-2/BCL-XL inhibitors navitoclax (ABT-263) and venetoclax (ABT-199) did not prove particularly effective either.
However, combining trametinib with navitoclax or venetoclax successfully induced apoptosis in B-ALL cells.
“Cancer cells often outwit us by rewiring themselves, but this early research offers a promising idea to get ahead of them,” said study author Richard Marais, PhD, of the Cancer Research UK Manchester Institute.
“We’ll still need to do further research to prove that this is the case beyond cancer cells in the laboratory, and it may take many years before we see it in the clinic, but it’s the first step to finding a new, effective drug combination for B-cell acute lymphoblastic leukemia.”
Dr Marais and his colleagues found that, although the MEK/ERK pathway is activated in B-ALL cells driven by different oncogenes, MEK inhibition alone did not suppress B-ALL cell growth.
And although B-ALL cells were sensitive to treatment with navitoclax or venetoclax alone, the researchers did not see complete loss of cell viability at clinically achievable doses.
However, trametinib did synergize with either navitoclax or venetoclax to suppress proliferation and induce apoptosis in B-ALL cells.
Further investigation revealed that the resistance of B-ALL cells to BCL-2/BCL-XL inhibition is mediated by MCL-1. And the synergism between trametinib and navitoclax/venetoclax is mediated by the pro-apoptotic factor BIM.
BIM is dephosphorylated as a result of MEK inhibition, which allows it to bind to and neutralize MCL-1, thereby enhancing BCL-2/BCL-XL inhibitor-induced cell death.
The researchers said they observed this effect in B-ALL cells driven by a range of genetic abnormalities, so the combination of a MEK inhibitor and a BCL-2/BCL-XL inhibitor could have therapeutic potential in a range of B-ALL subtypes. ![]()

Combining a MEK inhibitor and a BCL-2/BCL-XL inhibitor may be a feasible treatment option for B-cell acute lymphoblastic leukemia (B-ALL), according to preclinical research published in Cell Death and Disease.
Researchers found that, when given alone, the MEK inhibitor trametinib did not block B-ALL cell growth.
And the BCL-2/BCL-XL inhibitors navitoclax (ABT-263) and venetoclax (ABT-199) did not prove particularly effective either.
However, combining trametinib with navitoclax or venetoclax successfully induced apoptosis in B-ALL cells.
“Cancer cells often outwit us by rewiring themselves, but this early research offers a promising idea to get ahead of them,” said study author Richard Marais, PhD, of the Cancer Research UK Manchester Institute.
“We’ll still need to do further research to prove that this is the case beyond cancer cells in the laboratory, and it may take many years before we see it in the clinic, but it’s the first step to finding a new, effective drug combination for B-cell acute lymphoblastic leukemia.”
Dr Marais and his colleagues found that, although the MEK/ERK pathway is activated in B-ALL cells driven by different oncogenes, MEK inhibition alone did not suppress B-ALL cell growth.
And although B-ALL cells were sensitive to treatment with navitoclax or venetoclax alone, the researchers did not see complete loss of cell viability at clinically achievable doses.
However, trametinib did synergize with either navitoclax or venetoclax to suppress proliferation and induce apoptosis in B-ALL cells.
Further investigation revealed that the resistance of B-ALL cells to BCL-2/BCL-XL inhibition is mediated by MCL-1. And the synergism between trametinib and navitoclax/venetoclax is mediated by the pro-apoptotic factor BIM.
BIM is dephosphorylated as a result of MEK inhibition, which allows it to bind to and neutralize MCL-1, thereby enhancing BCL-2/BCL-XL inhibitor-induced cell death.
The researchers said they observed this effect in B-ALL cells driven by a range of genetic abnormalities, so the combination of a MEK inhibitor and a BCL-2/BCL-XL inhibitor could have therapeutic potential in a range of B-ALL subtypes. ![]()
Why married cancer patients fare better

Photo by Alena Kratochvilova
Results from two new studies provide a possible explanation for the link between marital status and survival in cancer patients.
Previous studies have shown that married cancer patients are more likely to survive and tend to have longer survival times than unmarried cancer patients.
Now, a pair of studies published in Cancer suggest it is the social support a patient receives from a spouse that may improve the patient’s outcome.
In the first study, Scarlett Lin Gomez, PhD, of the Cancer Prevention Institute of California, and her colleagues assessed the impact of socioeconomic factors and marital status on survival in cancer patients.
The team found evidence to suggest that economic resources play a minimal role in explaining the inferior survival observed in unmarried cancer patients.
In the second study, María Elena Martínez, PhD, of UC San Diego Moores Cancer Center, and her colleagues assessed the roles that race/ethnicity, sex, and nativity play in the survival of married and unmarried cancer patients.
The group found that not being married was associated with higher mortality, but the association varied by race/ethnicity and sex. The researchers believe these differences can be explained by the differences in social support networks between racial/ethnic groups and between men and women.
Patient cohort
Both studies were conducted on the same cohort of patients from the California Cancer Registry.
The researchers studied 783,167 cancer patients—393,470 males and 389,697 females. They were diagnosed from 2000 through 2009 with a first primary, invasive cancer of the 10 most common sites of cancer-related death for each sex, which included leukemias and lymphomas.
The patients were followed through December 31, 2012. A total of 386,607 patients died from any cause—204,007 males and 182,600 females.
Economic factors
Dr Gomez and her colleagues evaluated health insurance status and neighborhood socioeconomic status for the nearly 800,000 patients.
The researchers found that unmarried cancer patients had a greater risk of death than married patients, and this risk was higher among males than females. The hazard ratio (HR) for males was 1.27 (95% CI, 1.26-1.29), and the HR for females was 1.19 (95% CI, 1.18-1.20, P-interaction <0.001).
When the researchers adjusted for insurance status and neighborhood socioeconomic status, the marital status HRs decreased to 1.22 (95% CI, 1.21–1.24) for males and 1.15 (95% CI, 1.14–1.16) for females.
Based on these results, the researchers concluded that the survival benefit of marriage operates independently of the economic resources evaluated in this study.
“While other studies have found similar protective effects associated with being married, ours is the first in a large, population-based setting to assess the extent to which economic resources explain these protective effects,” Dr Gomez said. “Our study provides evidence for social support as a key driver.”
Race/ethnicity, nativity, and sex
Dr Martínez and her colleagues found that all-cause mortality was higher in the unmarried patients than in the married patients, but this varied significantly according to race/ethnicity and sex.
Marriage conferred less of a survival benefit for women than for men. However, for both sexes, non-Hispanic whites benefitted the most from being married, and Hispanics and Asian Pacific Islanders benefitted less.
Among males, the adjusted HRs were 1.24 (95% CI, 1.23-1.26) in non-Hispanic whites, 1.20 (95% CI, 1.16-1.24) in blacks, 1.20 (95% CI, 1.17-1.23) in Hispanics, and 1.11 (95% CI, 1.07-1.15) in Asian Pacific Islanders.
In females, the adjusted HRs were 1.17 (95% CI, 1.15-1.18) in non-Hispanic whites, 1.09 (95% CI, 1.05-1.13) in blacks, 1.11 (95% CI, 1.08-1.14) in Hispanics, and 1.07 (95% CI, 1.04-1.11) in Asian Pacific Islanders.
The researchers also found that all-cause mortality associated with unmarried status was higher in US-born Asian Pacific Islander and Hispanic men and women relative to their foreign-born counterparts.
“The results suggest that the more acculturated you become to US culture, the more it impacts cancer survivorship,” Dr Martínez said. “Our hypothesis is that non-Hispanic whites don’t have the same social network as other cultures that have stronger bonds with family and friends outside of marriage.”
“As individuals acculturate, they tend to lose those bonds. It’s also been shown that women seek out help for health concerns more frequently than men, and women tend to remind spouses to see their physicians and live a healthy lifestyle.” ![]()

Photo by Alena Kratochvilova
Results from two new studies provide a possible explanation for the link between marital status and survival in cancer patients.
Previous studies have shown that married cancer patients are more likely to survive and tend to have longer survival times than unmarried cancer patients.
Now, a pair of studies published in Cancer suggest it is the social support a patient receives from a spouse that may improve the patient’s outcome.
In the first study, Scarlett Lin Gomez, PhD, of the Cancer Prevention Institute of California, and her colleagues assessed the impact of socioeconomic factors and marital status on survival in cancer patients.
The team found evidence to suggest that economic resources play a minimal role in explaining the inferior survival observed in unmarried cancer patients.
In the second study, María Elena Martínez, PhD, of UC San Diego Moores Cancer Center, and her colleagues assessed the roles that race/ethnicity, sex, and nativity play in the survival of married and unmarried cancer patients.
The group found that not being married was associated with higher mortality, but the association varied by race/ethnicity and sex. The researchers believe these differences can be explained by the differences in social support networks between racial/ethnic groups and between men and women.
Patient cohort
Both studies were conducted on the same cohort of patients from the California Cancer Registry.
The researchers studied 783,167 cancer patients—393,470 males and 389,697 females. They were diagnosed from 2000 through 2009 with a first primary, invasive cancer of the 10 most common sites of cancer-related death for each sex, which included leukemias and lymphomas.
The patients were followed through December 31, 2012. A total of 386,607 patients died from any cause—204,007 males and 182,600 females.
Economic factors
Dr Gomez and her colleagues evaluated health insurance status and neighborhood socioeconomic status for the nearly 800,000 patients.
The researchers found that unmarried cancer patients had a greater risk of death than married patients, and this risk was higher among males than females. The hazard ratio (HR) for males was 1.27 (95% CI, 1.26-1.29), and the HR for females was 1.19 (95% CI, 1.18-1.20, P-interaction <0.001).
When the researchers adjusted for insurance status and neighborhood socioeconomic status, the marital status HRs decreased to 1.22 (95% CI, 1.21–1.24) for males and 1.15 (95% CI, 1.14–1.16) for females.
Based on these results, the researchers concluded that the survival benefit of marriage operates independently of the economic resources evaluated in this study.
“While other studies have found similar protective effects associated with being married, ours is the first in a large, population-based setting to assess the extent to which economic resources explain these protective effects,” Dr Gomez said. “Our study provides evidence for social support as a key driver.”
Race/ethnicity, nativity, and sex
Dr Martínez and her colleagues found that all-cause mortality was higher in the unmarried patients than in the married patients, but this varied significantly according to race/ethnicity and sex.
Marriage conferred less of a survival benefit for women than for men. However, for both sexes, non-Hispanic whites benefitted the most from being married, and Hispanics and Asian Pacific Islanders benefitted less.
Among males, the adjusted HRs were 1.24 (95% CI, 1.23-1.26) in non-Hispanic whites, 1.20 (95% CI, 1.16-1.24) in blacks, 1.20 (95% CI, 1.17-1.23) in Hispanics, and 1.11 (95% CI, 1.07-1.15) in Asian Pacific Islanders.
In females, the adjusted HRs were 1.17 (95% CI, 1.15-1.18) in non-Hispanic whites, 1.09 (95% CI, 1.05-1.13) in blacks, 1.11 (95% CI, 1.08-1.14) in Hispanics, and 1.07 (95% CI, 1.04-1.11) in Asian Pacific Islanders.
The researchers also found that all-cause mortality associated with unmarried status was higher in US-born Asian Pacific Islander and Hispanic men and women relative to their foreign-born counterparts.
“The results suggest that the more acculturated you become to US culture, the more it impacts cancer survivorship,” Dr Martínez said. “Our hypothesis is that non-Hispanic whites don’t have the same social network as other cultures that have stronger bonds with family and friends outside of marriage.”
“As individuals acculturate, they tend to lose those bonds. It’s also been shown that women seek out help for health concerns more frequently than men, and women tend to remind spouses to see their physicians and live a healthy lifestyle.” ![]()

Photo by Alena Kratochvilova
Results from two new studies provide a possible explanation for the link between marital status and survival in cancer patients.
Previous studies have shown that married cancer patients are more likely to survive and tend to have longer survival times than unmarried cancer patients.
Now, a pair of studies published in Cancer suggest it is the social support a patient receives from a spouse that may improve the patient’s outcome.
In the first study, Scarlett Lin Gomez, PhD, of the Cancer Prevention Institute of California, and her colleagues assessed the impact of socioeconomic factors and marital status on survival in cancer patients.
The team found evidence to suggest that economic resources play a minimal role in explaining the inferior survival observed in unmarried cancer patients.
In the second study, María Elena Martínez, PhD, of UC San Diego Moores Cancer Center, and her colleagues assessed the roles that race/ethnicity, sex, and nativity play in the survival of married and unmarried cancer patients.
The group found that not being married was associated with higher mortality, but the association varied by race/ethnicity and sex. The researchers believe these differences can be explained by the differences in social support networks between racial/ethnic groups and between men and women.
Patient cohort
Both studies were conducted on the same cohort of patients from the California Cancer Registry.
The researchers studied 783,167 cancer patients—393,470 males and 389,697 females. They were diagnosed from 2000 through 2009 with a first primary, invasive cancer of the 10 most common sites of cancer-related death for each sex, which included leukemias and lymphomas.
The patients were followed through December 31, 2012. A total of 386,607 patients died from any cause—204,007 males and 182,600 females.
Economic factors
Dr Gomez and her colleagues evaluated health insurance status and neighborhood socioeconomic status for the nearly 800,000 patients.
The researchers found that unmarried cancer patients had a greater risk of death than married patients, and this risk was higher among males than females. The hazard ratio (HR) for males was 1.27 (95% CI, 1.26-1.29), and the HR for females was 1.19 (95% CI, 1.18-1.20, P-interaction <0.001).
When the researchers adjusted for insurance status and neighborhood socioeconomic status, the marital status HRs decreased to 1.22 (95% CI, 1.21–1.24) for males and 1.15 (95% CI, 1.14–1.16) for females.
Based on these results, the researchers concluded that the survival benefit of marriage operates independently of the economic resources evaluated in this study.
“While other studies have found similar protective effects associated with being married, ours is the first in a large, population-based setting to assess the extent to which economic resources explain these protective effects,” Dr Gomez said. “Our study provides evidence for social support as a key driver.”
Race/ethnicity, nativity, and sex
Dr Martínez and her colleagues found that all-cause mortality was higher in the unmarried patients than in the married patients, but this varied significantly according to race/ethnicity and sex.
Marriage conferred less of a survival benefit for women than for men. However, for both sexes, non-Hispanic whites benefitted the most from being married, and Hispanics and Asian Pacific Islanders benefitted less.
Among males, the adjusted HRs were 1.24 (95% CI, 1.23-1.26) in non-Hispanic whites, 1.20 (95% CI, 1.16-1.24) in blacks, 1.20 (95% CI, 1.17-1.23) in Hispanics, and 1.11 (95% CI, 1.07-1.15) in Asian Pacific Islanders.
In females, the adjusted HRs were 1.17 (95% CI, 1.15-1.18) in non-Hispanic whites, 1.09 (95% CI, 1.05-1.13) in blacks, 1.11 (95% CI, 1.08-1.14) in Hispanics, and 1.07 (95% CI, 1.04-1.11) in Asian Pacific Islanders.
The researchers also found that all-cause mortality associated with unmarried status was higher in US-born Asian Pacific Islander and Hispanic men and women relative to their foreign-born counterparts.
“The results suggest that the more acculturated you become to US culture, the more it impacts cancer survivorship,” Dr Martínez said. “Our hypothesis is that non-Hispanic whites don’t have the same social network as other cultures that have stronger bonds with family and friends outside of marriage.”
“As individuals acculturate, they tend to lose those bonds. It’s also been shown that women seek out help for health concerns more frequently than men, and women tend to remind spouses to see their physicians and live a healthy lifestyle.” ![]()
Work ‘paves the way’ for platelet manufacture

in the bone marrow
Researchers in the UK have developed a new method of producing megakaryocytes from human pluripotent stem cells (hPSCs).
The team said their method generated large quantities of functional megakaryocytes that produced functional platelets.
They believe this work brings us one step closer to manufacturing platelets for transfusion.
The research was published in Nature Communications.
“Making megakaryocytes and platelets from stem cells for transfusion has been a long-standing challenge because of the sheer numbers we need to produce to make a single unit for transfusion,” said study author Cedric Ghevaert, MD, PhD, of the University of Cambridge and NHS Blood and Transplant in Cambridge, UK.
“We have found a way to ‘rewire’ the stem cells to make them become megakaryocytes a lot faster and more efficiently. It is a major step forward towards our goal to one day make blood cells in the laboratory to transfuse to patients.”
Dr Ghevaert and his colleagues employed a “forward programming” strategy in which 3 transcription factors—GATA1, FLI1, and TAL1—were used drive the differentiation of hPSCs into megakaryocytes.
The team said the forward-programmed megakaryocytes matured into platelet-producing cells that could be cryopreserved, maintained, and amplified in vitro for more than 90 days. The average yield was 200,000 megakaryocytes per hPSC.
The researchers generated platelets from the forward-programmed megakaryocytes via culture. And tests suggested these platelets were functionally similar to donor-derived platelets.
“The success of this research team in producing megakaryocytes in the laboratory has paved the way for the ultimate goal—manufacturing platelets for transfusion,” said Edwin Massey, MBChB, associate medical director for diagnostic and therapeutic services at NHS Blood and Transplant, who was not involved in this research.
“It will, however, be many years before a process for the large-scale production of platelets is developed. Donated platelets will still be needed by patients for the foreseeable future, either as part of a blood donation or by dedicated platelet donation using a machine collection process.” ![]()

in the bone marrow
Researchers in the UK have developed a new method of producing megakaryocytes from human pluripotent stem cells (hPSCs).
The team said their method generated large quantities of functional megakaryocytes that produced functional platelets.
They believe this work brings us one step closer to manufacturing platelets for transfusion.
The research was published in Nature Communications.
“Making megakaryocytes and platelets from stem cells for transfusion has been a long-standing challenge because of the sheer numbers we need to produce to make a single unit for transfusion,” said study author Cedric Ghevaert, MD, PhD, of the University of Cambridge and NHS Blood and Transplant in Cambridge, UK.
“We have found a way to ‘rewire’ the stem cells to make them become megakaryocytes a lot faster and more efficiently. It is a major step forward towards our goal to one day make blood cells in the laboratory to transfuse to patients.”
Dr Ghevaert and his colleagues employed a “forward programming” strategy in which 3 transcription factors—GATA1, FLI1, and TAL1—were used drive the differentiation of hPSCs into megakaryocytes.
The team said the forward-programmed megakaryocytes matured into platelet-producing cells that could be cryopreserved, maintained, and amplified in vitro for more than 90 days. The average yield was 200,000 megakaryocytes per hPSC.
The researchers generated platelets from the forward-programmed megakaryocytes via culture. And tests suggested these platelets were functionally similar to donor-derived platelets.
“The success of this research team in producing megakaryocytes in the laboratory has paved the way for the ultimate goal—manufacturing platelets for transfusion,” said Edwin Massey, MBChB, associate medical director for diagnostic and therapeutic services at NHS Blood and Transplant, who was not involved in this research.
“It will, however, be many years before a process for the large-scale production of platelets is developed. Donated platelets will still be needed by patients for the foreseeable future, either as part of a blood donation or by dedicated platelet donation using a machine collection process.” ![]()

in the bone marrow
Researchers in the UK have developed a new method of producing megakaryocytes from human pluripotent stem cells (hPSCs).
The team said their method generated large quantities of functional megakaryocytes that produced functional platelets.
They believe this work brings us one step closer to manufacturing platelets for transfusion.
The research was published in Nature Communications.
“Making megakaryocytes and platelets from stem cells for transfusion has been a long-standing challenge because of the sheer numbers we need to produce to make a single unit for transfusion,” said study author Cedric Ghevaert, MD, PhD, of the University of Cambridge and NHS Blood and Transplant in Cambridge, UK.
“We have found a way to ‘rewire’ the stem cells to make them become megakaryocytes a lot faster and more efficiently. It is a major step forward towards our goal to one day make blood cells in the laboratory to transfuse to patients.”
Dr Ghevaert and his colleagues employed a “forward programming” strategy in which 3 transcription factors—GATA1, FLI1, and TAL1—were used drive the differentiation of hPSCs into megakaryocytes.
The team said the forward-programmed megakaryocytes matured into platelet-producing cells that could be cryopreserved, maintained, and amplified in vitro for more than 90 days. The average yield was 200,000 megakaryocytes per hPSC.
The researchers generated platelets from the forward-programmed megakaryocytes via culture. And tests suggested these platelets were functionally similar to donor-derived platelets.
“The success of this research team in producing megakaryocytes in the laboratory has paved the way for the ultimate goal—manufacturing platelets for transfusion,” said Edwin Massey, MBChB, associate medical director for diagnostic and therapeutic services at NHS Blood and Transplant, who was not involved in this research.
“It will, however, be many years before a process for the large-scale production of platelets is developed. Donated platelets will still be needed by patients for the foreseeable future, either as part of a blood donation or by dedicated platelet donation using a machine collection process.” ![]()
Discovery could aid development of new sepsis therapies
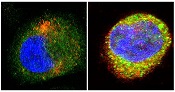
antibodies toward SHARPIN
(red) and caspase 1 (green).
Cell on right showing increase
in caspase 1 upon stimulation
with LPS and ATP and co-
localization with SHARPIN as
visualized by the merged
fluorescence (yellow). Nuclei
are stained blue with DAPI.
Image courtesy of The
American Journal of Pathology
A new study suggests that SHARPIN, a protein involved in regulating inflammation, has anti-septic effects.
Researchers believe this discovery, reported in The American Journal of Pathology, could spur the development of novel treatments for sepsis.
“Sepsis has been linked to enhanced activity of the enzyme caspase 1 and aberrant expression of pro-inflammatory interleukins 1β and 18,” said Liliana Schaefer, MD, of Goethe-Universität in Frankfurt, Germany.
“SHARPIN binds to caspase 1 and inhibits its activation. Our study proposes that the caspase 1/SHARPIN interaction may be a key pharmacological target in sepsis and perhaps in other inflammatory conditions where SHARPIN is involved.”
The researchers found that sepsis in mice bred to be deficient in SHARPIN resulted in enhanced levels of interleukins 1β and 18 and active caspase 1, as well as shortened survival.
Treatment with a caspase 1 inhibitor reversed these effects—reducing levels of interleukins 1β and 18, decreasing cell death in the spleen, and prolonging survival.
The researchers also found evidence to suggest this mechanism may be relevant to human sepsis.
“We found a decline in SHARPIN levels in septic patients correlating with enhanced activation of caspase 1 in circulating mononuclear cells and an increase of interleukin 1β/18 in the plasma,” Dr Schaefer said.
“Our findings suggest that using pharmacological caspase 1 inhibitors could be beneficial in septic patients with low SHARPIN levels, and these therapies may be more efficient than other anti-inflammatory therapies.” ![]()

antibodies toward SHARPIN
(red) and caspase 1 (green).
Cell on right showing increase
in caspase 1 upon stimulation
with LPS and ATP and co-
localization with SHARPIN as
visualized by the merged
fluorescence (yellow). Nuclei
are stained blue with DAPI.
Image courtesy of The
American Journal of Pathology
A new study suggests that SHARPIN, a protein involved in regulating inflammation, has anti-septic effects.
Researchers believe this discovery, reported in The American Journal of Pathology, could spur the development of novel treatments for sepsis.
“Sepsis has been linked to enhanced activity of the enzyme caspase 1 and aberrant expression of pro-inflammatory interleukins 1β and 18,” said Liliana Schaefer, MD, of Goethe-Universität in Frankfurt, Germany.
“SHARPIN binds to caspase 1 and inhibits its activation. Our study proposes that the caspase 1/SHARPIN interaction may be a key pharmacological target in sepsis and perhaps in other inflammatory conditions where SHARPIN is involved.”
The researchers found that sepsis in mice bred to be deficient in SHARPIN resulted in enhanced levels of interleukins 1β and 18 and active caspase 1, as well as shortened survival.
Treatment with a caspase 1 inhibitor reversed these effects—reducing levels of interleukins 1β and 18, decreasing cell death in the spleen, and prolonging survival.
The researchers also found evidence to suggest this mechanism may be relevant to human sepsis.
“We found a decline in SHARPIN levels in septic patients correlating with enhanced activation of caspase 1 in circulating mononuclear cells and an increase of interleukin 1β/18 in the plasma,” Dr Schaefer said.
“Our findings suggest that using pharmacological caspase 1 inhibitors could be beneficial in septic patients with low SHARPIN levels, and these therapies may be more efficient than other anti-inflammatory therapies.” ![]()

antibodies toward SHARPIN
(red) and caspase 1 (green).
Cell on right showing increase
in caspase 1 upon stimulation
with LPS and ATP and co-
localization with SHARPIN as
visualized by the merged
fluorescence (yellow). Nuclei
are stained blue with DAPI.
Image courtesy of The
American Journal of Pathology
A new study suggests that SHARPIN, a protein involved in regulating inflammation, has anti-septic effects.
Researchers believe this discovery, reported in The American Journal of Pathology, could spur the development of novel treatments for sepsis.
“Sepsis has been linked to enhanced activity of the enzyme caspase 1 and aberrant expression of pro-inflammatory interleukins 1β and 18,” said Liliana Schaefer, MD, of Goethe-Universität in Frankfurt, Germany.
“SHARPIN binds to caspase 1 and inhibits its activation. Our study proposes that the caspase 1/SHARPIN interaction may be a key pharmacological target in sepsis and perhaps in other inflammatory conditions where SHARPIN is involved.”
The researchers found that sepsis in mice bred to be deficient in SHARPIN resulted in enhanced levels of interleukins 1β and 18 and active caspase 1, as well as shortened survival.
Treatment with a caspase 1 inhibitor reversed these effects—reducing levels of interleukins 1β and 18, decreasing cell death in the spleen, and prolonging survival.
The researchers also found evidence to suggest this mechanism may be relevant to human sepsis.
“We found a decline in SHARPIN levels in septic patients correlating with enhanced activation of caspase 1 in circulating mononuclear cells and an increase of interleukin 1β/18 in the plasma,” Dr Schaefer said.
“Our findings suggest that using pharmacological caspase 1 inhibitors could be beneficial in septic patients with low SHARPIN levels, and these therapies may be more efficient than other anti-inflammatory therapies.” ![]()
Inhibitor could overcome TKI resistance in Ph+ B-ALL

Results of preclinical research indicate that combining 2 kinase inhibitors may be a promising treatment strategy for Philadelphia chromosome-positive B-cell acute lymphoblastic leukemia (Ph+ B-ALL).
Researchers found that combining a tyrosine kinase inhibitor (TKI) and an inhibitor of focal adhesion kinase (FAK) was “remarkably effective” against Ph+ B-ALL in vitro and in vivo.
The TKI dasatinib and the FAK inhibitor VS-4718 decreased leukemic cell survival and adhesion, inhibited tumor growth, and prolonged survival in mouse models of Ph+ B-ALL.
Charles Mullighan, MBBS, MD, of St. Jude Children’s Research Hospital in Memphis, Tennessee, and his colleagues conducted this research and reported their results in JCI Insight.
The researchers noted that patients with Ph+ ALL have shown resistance to TKI therapy, and this resistance has been tied to alterations in IKZF1.
As FAK expression is elevated in IKZF1-mutated leukemias, the team speculated that adding a FAK inhibitor to TKI therapy might lead to better results.
First, the researchers set out to confirm that FAK is overexpressed in Ph+ B-ALL. Their experiments revealed upregulation of the FAK pathway in Ph+ B-ALL cells, with further overexpression of FAK in IKZF1-mutated Ph+ B-ALL cells.
When they inhibited FAK with VS-4718, the team observed decreases in the survival, clonogenicity, and adhesion of IKZF1-mutated Ph+ B-ALL cells from both mice and humans.
Next, the researchers found that VS-4718 synergizes with the TKI dasatinib in vitro and in vivo.
In in vitro experiments with both mouse and human Ph+ B-ALL cells, the combination decreased cell survival and adhesion and inhibited downstream targets of FAK.
In mouse models of Ph+ B-ALL, VS-4718 proved ineffective when given alone.
However, the researchers said the combination of VS-4718 and dasatinib “dramatically” decreased leukemic burden and extended the lives of mice.
In fact, 1 long-term survivor achieved a complete remission that endured after treatment was stopped.
The researchers said these results suggest that targeting FAK with VS-4718 can overcome the deleterious effects of FAK overexpression in Ph+ B-ALL, potentiating responsiveness to TKIs. ![]()

Results of preclinical research indicate that combining 2 kinase inhibitors may be a promising treatment strategy for Philadelphia chromosome-positive B-cell acute lymphoblastic leukemia (Ph+ B-ALL).
Researchers found that combining a tyrosine kinase inhibitor (TKI) and an inhibitor of focal adhesion kinase (FAK) was “remarkably effective” against Ph+ B-ALL in vitro and in vivo.
The TKI dasatinib and the FAK inhibitor VS-4718 decreased leukemic cell survival and adhesion, inhibited tumor growth, and prolonged survival in mouse models of Ph+ B-ALL.
Charles Mullighan, MBBS, MD, of St. Jude Children’s Research Hospital in Memphis, Tennessee, and his colleagues conducted this research and reported their results in JCI Insight.
The researchers noted that patients with Ph+ ALL have shown resistance to TKI therapy, and this resistance has been tied to alterations in IKZF1.
As FAK expression is elevated in IKZF1-mutated leukemias, the team speculated that adding a FAK inhibitor to TKI therapy might lead to better results.
First, the researchers set out to confirm that FAK is overexpressed in Ph+ B-ALL. Their experiments revealed upregulation of the FAK pathway in Ph+ B-ALL cells, with further overexpression of FAK in IKZF1-mutated Ph+ B-ALL cells.
When they inhibited FAK with VS-4718, the team observed decreases in the survival, clonogenicity, and adhesion of IKZF1-mutated Ph+ B-ALL cells from both mice and humans.
Next, the researchers found that VS-4718 synergizes with the TKI dasatinib in vitro and in vivo.
In in vitro experiments with both mouse and human Ph+ B-ALL cells, the combination decreased cell survival and adhesion and inhibited downstream targets of FAK.
In mouse models of Ph+ B-ALL, VS-4718 proved ineffective when given alone.
However, the researchers said the combination of VS-4718 and dasatinib “dramatically” decreased leukemic burden and extended the lives of mice.
In fact, 1 long-term survivor achieved a complete remission that endured after treatment was stopped.
The researchers said these results suggest that targeting FAK with VS-4718 can overcome the deleterious effects of FAK overexpression in Ph+ B-ALL, potentiating responsiveness to TKIs. ![]()

Results of preclinical research indicate that combining 2 kinase inhibitors may be a promising treatment strategy for Philadelphia chromosome-positive B-cell acute lymphoblastic leukemia (Ph+ B-ALL).
Researchers found that combining a tyrosine kinase inhibitor (TKI) and an inhibitor of focal adhesion kinase (FAK) was “remarkably effective” against Ph+ B-ALL in vitro and in vivo.
The TKI dasatinib and the FAK inhibitor VS-4718 decreased leukemic cell survival and adhesion, inhibited tumor growth, and prolonged survival in mouse models of Ph+ B-ALL.
Charles Mullighan, MBBS, MD, of St. Jude Children’s Research Hospital in Memphis, Tennessee, and his colleagues conducted this research and reported their results in JCI Insight.
The researchers noted that patients with Ph+ ALL have shown resistance to TKI therapy, and this resistance has been tied to alterations in IKZF1.
As FAK expression is elevated in IKZF1-mutated leukemias, the team speculated that adding a FAK inhibitor to TKI therapy might lead to better results.
First, the researchers set out to confirm that FAK is overexpressed in Ph+ B-ALL. Their experiments revealed upregulation of the FAK pathway in Ph+ B-ALL cells, with further overexpression of FAK in IKZF1-mutated Ph+ B-ALL cells.
When they inhibited FAK with VS-4718, the team observed decreases in the survival, clonogenicity, and adhesion of IKZF1-mutated Ph+ B-ALL cells from both mice and humans.
Next, the researchers found that VS-4718 synergizes with the TKI dasatinib in vitro and in vivo.
In in vitro experiments with both mouse and human Ph+ B-ALL cells, the combination decreased cell survival and adhesion and inhibited downstream targets of FAK.
In mouse models of Ph+ B-ALL, VS-4718 proved ineffective when given alone.
However, the researchers said the combination of VS-4718 and dasatinib “dramatically” decreased leukemic burden and extended the lives of mice.
In fact, 1 long-term survivor achieved a complete remission that endured after treatment was stopped.
The researchers said these results suggest that targeting FAK with VS-4718 can overcome the deleterious effects of FAK overexpression in Ph+ B-ALL, potentiating responsiveness to TKIs. ![]()
Slowing the progression of sickle cell disease
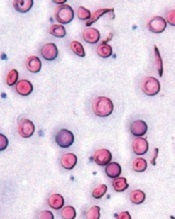
Image courtesy of the
University of Michigan
Activating the antioxidant regulator Nrf2 may slow the progression of sickle cell disease (SCD), according to preclinical research published in JCI Insight.
Investigators found the severity of hemolytic anemia, vascular inflammation, and lung injury increased with age in mice with SCD.
However, activating Nrf2 in young animals had a prophylactic effect, reducing the severity of these adverse effects and improving survival.
To uncover these findings, Solomon Ofori-Acquah, PhD, of the University of Pittsburgh in Pennsylvania, and his colleagues conducted a 10-month longitudinal observational study of mice with SCD.
The team found that, in mice with homozygous SCD (SS), there was a link between intravascular hemolysis, vascular inflammation, lung injury, and early death.
Mice as young as 2 months showed exacerbation of intravascular hemolysis. And additional investigation linked worsening intravascular hemolysis and oxidative stress to the release of VE-cadherin and progressive lung damage in aging SS mice.
The investigators knew that Nrf2 regulates the expression of genes that protect against the effects of intravascular hemolysis. So they decided to see if activating Nrf2 in young mice with SCD would slow the disease progression that occurs with age.
The team took SS mice that were about a month old and randomized them to receive 3H-1, 2-dithiole-3-thione (D3T) or a DMSO vehicle for 3 months or longer.
Treatment with D3T stabilized the concentration of hemoglobin, increased white blood cell counts, increased reticulocyte counts (though not significantly), kept HO-1 levels stable, increased levels of NQO1 and ferritin, and impeded the progression of endothelial dysfunction.
The investigators also looked at the role of Nrf2 in nonhematopoietic tissues and were surprised to find that Nrf2 deficiency in nonhematopoietic tissues exacerbated anemia and caused premature pulmonary edema in mice with SCD.
The team said this suggests a dominant protective role for nonhematopoietic Nrf2 against tissue damage in both erythroid and nonerythroid tissues in SCD.
And, when taken together, the results of this research indicate that activating Nrf2 can impede the onset of the severe adult phenotype of SCD in mice. ![]()

Image courtesy of the
University of Michigan
Activating the antioxidant regulator Nrf2 may slow the progression of sickle cell disease (SCD), according to preclinical research published in JCI Insight.
Investigators found the severity of hemolytic anemia, vascular inflammation, and lung injury increased with age in mice with SCD.
However, activating Nrf2 in young animals had a prophylactic effect, reducing the severity of these adverse effects and improving survival.
To uncover these findings, Solomon Ofori-Acquah, PhD, of the University of Pittsburgh in Pennsylvania, and his colleagues conducted a 10-month longitudinal observational study of mice with SCD.
The team found that, in mice with homozygous SCD (SS), there was a link between intravascular hemolysis, vascular inflammation, lung injury, and early death.
Mice as young as 2 months showed exacerbation of intravascular hemolysis. And additional investigation linked worsening intravascular hemolysis and oxidative stress to the release of VE-cadherin and progressive lung damage in aging SS mice.
The investigators knew that Nrf2 regulates the expression of genes that protect against the effects of intravascular hemolysis. So they decided to see if activating Nrf2 in young mice with SCD would slow the disease progression that occurs with age.
The team took SS mice that were about a month old and randomized them to receive 3H-1, 2-dithiole-3-thione (D3T) or a DMSO vehicle for 3 months or longer.
Treatment with D3T stabilized the concentration of hemoglobin, increased white blood cell counts, increased reticulocyte counts (though not significantly), kept HO-1 levels stable, increased levels of NQO1 and ferritin, and impeded the progression of endothelial dysfunction.
The investigators also looked at the role of Nrf2 in nonhematopoietic tissues and were surprised to find that Nrf2 deficiency in nonhematopoietic tissues exacerbated anemia and caused premature pulmonary edema in mice with SCD.
The team said this suggests a dominant protective role for nonhematopoietic Nrf2 against tissue damage in both erythroid and nonerythroid tissues in SCD.
And, when taken together, the results of this research indicate that activating Nrf2 can impede the onset of the severe adult phenotype of SCD in mice. ![]()

Image courtesy of the
University of Michigan
Activating the antioxidant regulator Nrf2 may slow the progression of sickle cell disease (SCD), according to preclinical research published in JCI Insight.
Investigators found the severity of hemolytic anemia, vascular inflammation, and lung injury increased with age in mice with SCD.
However, activating Nrf2 in young animals had a prophylactic effect, reducing the severity of these adverse effects and improving survival.
To uncover these findings, Solomon Ofori-Acquah, PhD, of the University of Pittsburgh in Pennsylvania, and his colleagues conducted a 10-month longitudinal observational study of mice with SCD.
The team found that, in mice with homozygous SCD (SS), there was a link between intravascular hemolysis, vascular inflammation, lung injury, and early death.
Mice as young as 2 months showed exacerbation of intravascular hemolysis. And additional investigation linked worsening intravascular hemolysis and oxidative stress to the release of VE-cadherin and progressive lung damage in aging SS mice.
The investigators knew that Nrf2 regulates the expression of genes that protect against the effects of intravascular hemolysis. So they decided to see if activating Nrf2 in young mice with SCD would slow the disease progression that occurs with age.
The team took SS mice that were about a month old and randomized them to receive 3H-1, 2-dithiole-3-thione (D3T) or a DMSO vehicle for 3 months or longer.
Treatment with D3T stabilized the concentration of hemoglobin, increased white blood cell counts, increased reticulocyte counts (though not significantly), kept HO-1 levels stable, increased levels of NQO1 and ferritin, and impeded the progression of endothelial dysfunction.
The investigators also looked at the role of Nrf2 in nonhematopoietic tissues and were surprised to find that Nrf2 deficiency in nonhematopoietic tissues exacerbated anemia and caused premature pulmonary edema in mice with SCD.
The team said this suggests a dominant protective role for nonhematopoietic Nrf2 against tissue damage in both erythroid and nonerythroid tissues in SCD.
And, when taken together, the results of this research indicate that activating Nrf2 can impede the onset of the severe adult phenotype of SCD in mice. ![]()
EC approves drug for pediatric ITP

Photo by Logan Tuttle
The European Commission (EC) has approved eltrombopag (Revolade), a once-daily oral thrombopoietin receptor agonist, to treat pediatric patients (age 1 and older) with chronic immune thrombocytopenia (ITP) that is refractory to other therapies.
This approval includes the use of tablets and a new oral suspension formulation of eltrombopag, which is designed for younger children who may not be able to swallow tablets.
The approval applies to all 28 member states of the European Union plus Iceland, Norway, and Liechtenstein.
Eltrombopag was previously approved by the EC for use in adults with refractory chronic ITP. The drug is also approved in the EC to treat adults with severe aplastic anemia and adults with chronic hepatitis C virus infection who have thrombocytopenia.
Eltrombopag is made by Novartis. For more details on the drug, see the full Summary of Product Characteristics, available on the European Medicines Agency’s website.
The EC’s latest approval of eltrombopag was based on data from 2 double-blind, placebo-controlled trials—the phase 2 PETIT trial and the phase 3 PETIT2 trial.
PETIT trials: Efficacy
The PETIT trial included 67 ITP patients stratified by age cohort (12-17 years, 6-11 years, and 1-5 years). They were randomized (2:1) to receive eltrombopag or placebo for 7 weeks. The eltrombopag dose was titrated to a target platelet count of 50-200 x 109/L.
The primary efficacy endpoint was the proportion of subjects achieving platelet counts of 50 x 109/L or higher at least once between days 8 and 43 of the randomized period of the study.
Significantly more patients in the eltrombopag arm met this endpoint—62.2%—compared to 31.8% in the placebo arm (P=0.011).
The PETIT2 trial enrolled 92 patients with chronic ITP who were randomized (2:1) to receive eltrombopag or placebo for 13 weeks. The eltrombopag dose was titrated to a target platelet count of 50-200 x 109/L.
The primary efficacy endpoint was the proportion of subjects who achieved platelet counts of 50 x 109/L or higher for at least 6 out of 8 weeks, between weeks 5 and 12 of the randomized period.
Significantly more patients in the eltrombopag arm met this endpoint—41.3%—compared to 3.4% of patients in the placebo arm (P<0.001).
PETIT trials: Safety
For both trials, there were 107 eltrombopag-treated patients evaluable for safety.
The most common adverse events that occurred more frequently in the eltrombopag arms than the placebo arms were upper respiratory tract infection, nasopharyngitis, cough, diarrhea, pyrexia, rhinitis, abdominal pain, oropharyngeal pain, toothache, increased ALT/AST, rash, and rhinorrhea.
Serious adverse events were reported in 8% of patients during the randomized part of both trials, although no serious adverse event occurred in more than 1 patient.
An ALT elevation of at least 3 times the upper limit of normal occurred in 5% of eltrombopag-treated patients. Of those patients, 2% had ALT increases of at least 5 times the upper limit of normal.
There were no deaths or thromboembolic events during either study. ![]()

Photo by Logan Tuttle
The European Commission (EC) has approved eltrombopag (Revolade), a once-daily oral thrombopoietin receptor agonist, to treat pediatric patients (age 1 and older) with chronic immune thrombocytopenia (ITP) that is refractory to other therapies.
This approval includes the use of tablets and a new oral suspension formulation of eltrombopag, which is designed for younger children who may not be able to swallow tablets.
The approval applies to all 28 member states of the European Union plus Iceland, Norway, and Liechtenstein.
Eltrombopag was previously approved by the EC for use in adults with refractory chronic ITP. The drug is also approved in the EC to treat adults with severe aplastic anemia and adults with chronic hepatitis C virus infection who have thrombocytopenia.
Eltrombopag is made by Novartis. For more details on the drug, see the full Summary of Product Characteristics, available on the European Medicines Agency’s website.
The EC’s latest approval of eltrombopag was based on data from 2 double-blind, placebo-controlled trials—the phase 2 PETIT trial and the phase 3 PETIT2 trial.
PETIT trials: Efficacy
The PETIT trial included 67 ITP patients stratified by age cohort (12-17 years, 6-11 years, and 1-5 years). They were randomized (2:1) to receive eltrombopag or placebo for 7 weeks. The eltrombopag dose was titrated to a target platelet count of 50-200 x 109/L.
The primary efficacy endpoint was the proportion of subjects achieving platelet counts of 50 x 109/L or higher at least once between days 8 and 43 of the randomized period of the study.
Significantly more patients in the eltrombopag arm met this endpoint—62.2%—compared to 31.8% in the placebo arm (P=0.011).
The PETIT2 trial enrolled 92 patients with chronic ITP who were randomized (2:1) to receive eltrombopag or placebo for 13 weeks. The eltrombopag dose was titrated to a target platelet count of 50-200 x 109/L.
The primary efficacy endpoint was the proportion of subjects who achieved platelet counts of 50 x 109/L or higher for at least 6 out of 8 weeks, between weeks 5 and 12 of the randomized period.
Significantly more patients in the eltrombopag arm met this endpoint—41.3%—compared to 3.4% of patients in the placebo arm (P<0.001).
PETIT trials: Safety
For both trials, there were 107 eltrombopag-treated patients evaluable for safety.
The most common adverse events that occurred more frequently in the eltrombopag arms than the placebo arms were upper respiratory tract infection, nasopharyngitis, cough, diarrhea, pyrexia, rhinitis, abdominal pain, oropharyngeal pain, toothache, increased ALT/AST, rash, and rhinorrhea.
Serious adverse events were reported in 8% of patients during the randomized part of both trials, although no serious adverse event occurred in more than 1 patient.
An ALT elevation of at least 3 times the upper limit of normal occurred in 5% of eltrombopag-treated patients. Of those patients, 2% had ALT increases of at least 5 times the upper limit of normal.
There were no deaths or thromboembolic events during either study. ![]()

Photo by Logan Tuttle
The European Commission (EC) has approved eltrombopag (Revolade), a once-daily oral thrombopoietin receptor agonist, to treat pediatric patients (age 1 and older) with chronic immune thrombocytopenia (ITP) that is refractory to other therapies.
This approval includes the use of tablets and a new oral suspension formulation of eltrombopag, which is designed for younger children who may not be able to swallow tablets.
The approval applies to all 28 member states of the European Union plus Iceland, Norway, and Liechtenstein.
Eltrombopag was previously approved by the EC for use in adults with refractory chronic ITP. The drug is also approved in the EC to treat adults with severe aplastic anemia and adults with chronic hepatitis C virus infection who have thrombocytopenia.
Eltrombopag is made by Novartis. For more details on the drug, see the full Summary of Product Characteristics, available on the European Medicines Agency’s website.
The EC’s latest approval of eltrombopag was based on data from 2 double-blind, placebo-controlled trials—the phase 2 PETIT trial and the phase 3 PETIT2 trial.
PETIT trials: Efficacy
The PETIT trial included 67 ITP patients stratified by age cohort (12-17 years, 6-11 years, and 1-5 years). They were randomized (2:1) to receive eltrombopag or placebo for 7 weeks. The eltrombopag dose was titrated to a target platelet count of 50-200 x 109/L.
The primary efficacy endpoint was the proportion of subjects achieving platelet counts of 50 x 109/L or higher at least once between days 8 and 43 of the randomized period of the study.
Significantly more patients in the eltrombopag arm met this endpoint—62.2%—compared to 31.8% in the placebo arm (P=0.011).
The PETIT2 trial enrolled 92 patients with chronic ITP who were randomized (2:1) to receive eltrombopag or placebo for 13 weeks. The eltrombopag dose was titrated to a target platelet count of 50-200 x 109/L.
The primary efficacy endpoint was the proportion of subjects who achieved platelet counts of 50 x 109/L or higher for at least 6 out of 8 weeks, between weeks 5 and 12 of the randomized period.
Significantly more patients in the eltrombopag arm met this endpoint—41.3%—compared to 3.4% of patients in the placebo arm (P<0.001).
PETIT trials: Safety
For both trials, there were 107 eltrombopag-treated patients evaluable for safety.
The most common adverse events that occurred more frequently in the eltrombopag arms than the placebo arms were upper respiratory tract infection, nasopharyngitis, cough, diarrhea, pyrexia, rhinitis, abdominal pain, oropharyngeal pain, toothache, increased ALT/AST, rash, and rhinorrhea.
Serious adverse events were reported in 8% of patients during the randomized part of both trials, although no serious adverse event occurred in more than 1 patient.
An ALT elevation of at least 3 times the upper limit of normal occurred in 5% of eltrombopag-treated patients. Of those patients, 2% had ALT increases of at least 5 times the upper limit of normal.
There were no deaths or thromboembolic events during either study.
Study suggests iPSCs pose no cancer risk

Image from the Salk Institute
In tracking the mutational history of somatic cells and induced pluripotent stem cells (iPSCs), researchers found that somatic cells accumulate mutations more frequently than iPSCs.
And none of the mutations found in iPSCs were associated with cancers.
“None of the mutations we found in induced pluripotent stem cells were cancer-driver mutations or mutations in cancer-causing genes,” said Foad Rouhani, of the Wellcome Trust Sanger Institute in the UK.
“We didn’t find anything that would preclude the use of [iPSCs] in therapeutic medicine.”
Rouhani and his colleagues reported these findings in PLOS Genetics.
The researchers generated iPSCs using cells from healthy individuals, then sequenced the genomes of the somatic cells and the derived iPSCs.
They found that somatic cells had a mutation rate of 14 single nucleotide variants per cell per generation, and the mutation rate for iPSCs was 10-fold lower.
The researchers said this is the first time that mutation rates of both types of cells, the donor cell and iPSC, have been calculated and compared.
“Until now, the question of whether generating [iPSCs] and growing them in cell culture creates mutations has not been addressed in detail,” said study author Allan Bradley, PhD, of the Wellcome Trust Sanger Institute.
“If human cells are really to be reprogrammed on a large scale for use in regenerative medicine, then understanding the mutations the donor cells carry will be a crucial step. We now have the tools to do this.”
The researchers also used the iPSCs to trace the history of every mutation that one endothelial progenitor cell had developed from the time it was a fertilized egg to the moment it was taken out of the body.
They said the ability to track the genetic changes in cells over a lifetime could improve scientists’ understanding of how, when, and why mutations lead to cancer.

Image from the Salk Institute
In tracking the mutational history of somatic cells and induced pluripotent stem cells (iPSCs), researchers found that somatic cells accumulate mutations more frequently than iPSCs.
And none of the mutations found in iPSCs were associated with cancers.
“None of the mutations we found in induced pluripotent stem cells were cancer-driver mutations or mutations in cancer-causing genes,” said Foad Rouhani, of the Wellcome Trust Sanger Institute in the UK.
“We didn’t find anything that would preclude the use of [iPSCs] in therapeutic medicine.”
Rouhani and his colleagues reported these findings in PLOS Genetics.
The researchers generated iPSCs using cells from healthy individuals, then sequenced the genomes of the somatic cells and the derived iPSCs.
They found that somatic cells had a mutation rate of 14 single nucleotide variants per cell per generation, and the mutation rate for iPSCs was 10-fold lower.
The researchers said this is the first time that mutation rates of both types of cells, the donor cell and iPSC, have been calculated and compared.
“Until now, the question of whether generating [iPSCs] and growing them in cell culture creates mutations has not been addressed in detail,” said study author Allan Bradley, PhD, of the Wellcome Trust Sanger Institute.
“If human cells are really to be reprogrammed on a large scale for use in regenerative medicine, then understanding the mutations the donor cells carry will be a crucial step. We now have the tools to do this.”
The researchers also used the iPSCs to trace the history of every mutation that one endothelial progenitor cell had developed from the time it was a fertilized egg to the moment it was taken out of the body.
They said the ability to track the genetic changes in cells over a lifetime could improve scientists’ understanding of how, when, and why mutations lead to cancer.

Image from the Salk Institute
In tracking the mutational history of somatic cells and induced pluripotent stem cells (iPSCs), researchers found that somatic cells accumulate mutations more frequently than iPSCs.
And none of the mutations found in iPSCs were associated with cancers.
“None of the mutations we found in induced pluripotent stem cells were cancer-driver mutations or mutations in cancer-causing genes,” said Foad Rouhani, of the Wellcome Trust Sanger Institute in the UK.
“We didn’t find anything that would preclude the use of [iPSCs] in therapeutic medicine.”
Rouhani and his colleagues reported these findings in PLOS Genetics.
The researchers generated iPSCs using cells from healthy individuals, then sequenced the genomes of the somatic cells and the derived iPSCs.
They found that somatic cells had a mutation rate of 14 single nucleotide variants per cell per generation, and the mutation rate for iPSCs was 10-fold lower.
The researchers said this is the first time that mutation rates of both types of cells, the donor cell and iPSC, have been calculated and compared.
“Until now, the question of whether generating [iPSCs] and growing them in cell culture creates mutations has not been addressed in detail,” said study author Allan Bradley, PhD, of the Wellcome Trust Sanger Institute.
“If human cells are really to be reprogrammed on a large scale for use in regenerative medicine, then understanding the mutations the donor cells carry will be a crucial step. We now have the tools to do this.”
The researchers also used the iPSCs to trace the history of every mutation that one endothelial progenitor cell had developed from the time it was a fertilized egg to the moment it was taken out of the body.
They said the ability to track the genetic changes in cells over a lifetime could improve scientists’ understanding of how, when, and why mutations lead to cancer.
Expanded UCB product provides clinical benefit

Photo courtesy of NHS
VALENCIA, SPAIN—The expanded umbilical cord blood (UCB) product NiCord can provide clinical benefits in patients with high-risk hematologic malignancies, according to data presented at the 42nd Annual Meeting of the European Society for Blood and Marrow Transplantation.
NiCord consists of cells from a single UCB unit cultured in nicotinamide—a vitamin B derivative—and cytokines that are typically used for expansion—thrombopoietin, interleukin 6, FLT3 ligand, and stem cell factor.
The data showed that patients transplanted with NiCord had fewer moderate to severe bacterial infections and shorter hospital stays than patients who received standard UCB transplants.
“We saw a significant reduction in serious bacterial infections during the first 100 days in the NiCord group,” said Mitchell Horwitz, MD, of the Duke University School of Medicine in Durham, North Carolina.
“This is encouraging because this type of infection is a major cause of early death following UCB transplantation. We also saw a significant reduction in hospitalization time in the NiCord group, indicating a faster recovery of these patients in comparison to those transplanted with standard umbilical cord blood.”
These results were presented at the meeting as abstract O090. The research was funded by Gamida Cell, the company developing NiCord.
Dr Horwitz and his colleagues analyzed 18 patients with high-risk hematologic malignancies—most with acute leukemia or myelodysplastic syndromes (90%)—who were transplanted with NiCord.
Ten of the patients received NiCord with a second, unmanipulated UCB unit, and 8 patients received NiCord as a single UCB graft.
The researchers compared these patients to 101 patients who received standard single or double UCB transplants at Duke University from January 2005 to March 2015.
Patients in both groups received a total body irradiation-based myeloablative preparative regimen.
The median time to neutrophil engraftment was significantly shorter in the NiCord group than the control group—12.5 days and 27 days, respectively (P<0.001).
All 18 patients in the Nicord group and 100 patients in the control group had at least 1 infection.
Patients in the NiCord group had a significantly lower incidence of grade 2-3 bacterial infections than patients in the control group—22% and 54%, respectively (P=0.015).
However, there was no significant difference between the groups with regard to grade 2-3 viral infections (39% and 35%, respectively, P=0.729), fungal infections (0% and 5%, respectively, P=1.0), or non-microbiologically defined infections (0% and 17%, respectively, P=0.072).
In the first 100 days after transplant, patients in the NiCord group spent significantly more days out of the hospital than patients in the control group. The median number of days for each group was 74 and 53, respectively (P=0.002).
“These results demonstrate that the rapid hematopoietic recovery from NiCord transplantation results in clinical benefit, in comparison to similar site controls,” Dr Horwitz concluded.

Photo courtesy of NHS
VALENCIA, SPAIN—The expanded umbilical cord blood (UCB) product NiCord can provide clinical benefits in patients with high-risk hematologic malignancies, according to data presented at the 42nd Annual Meeting of the European Society for Blood and Marrow Transplantation.
NiCord consists of cells from a single UCB unit cultured in nicotinamide—a vitamin B derivative—and cytokines that are typically used for expansion—thrombopoietin, interleukin 6, FLT3 ligand, and stem cell factor.
The data showed that patients transplanted with NiCord had fewer moderate to severe bacterial infections and shorter hospital stays than patients who received standard UCB transplants.
“We saw a significant reduction in serious bacterial infections during the first 100 days in the NiCord group,” said Mitchell Horwitz, MD, of the Duke University School of Medicine in Durham, North Carolina.
“This is encouraging because this type of infection is a major cause of early death following UCB transplantation. We also saw a significant reduction in hospitalization time in the NiCord group, indicating a faster recovery of these patients in comparison to those transplanted with standard umbilical cord blood.”
These results were presented at the meeting as abstract O090. The research was funded by Gamida Cell, the company developing NiCord.
Dr Horwitz and his colleagues analyzed 18 patients with high-risk hematologic malignancies—most with acute leukemia or myelodysplastic syndromes (90%)—who were transplanted with NiCord.
Ten of the patients received NiCord with a second, unmanipulated UCB unit, and 8 patients received NiCord as a single UCB graft.
The researchers compared these patients to 101 patients who received standard single or double UCB transplants at Duke University from January 2005 to March 2015.
Patients in both groups received a total body irradiation-based myeloablative preparative regimen.
The median time to neutrophil engraftment was significantly shorter in the NiCord group than the control group—12.5 days and 27 days, respectively (P<0.001).
All 18 patients in the Nicord group and 100 patients in the control group had at least 1 infection.
Patients in the NiCord group had a significantly lower incidence of grade 2-3 bacterial infections than patients in the control group—22% and 54%, respectively (P=0.015).
However, there was no significant difference between the groups with regard to grade 2-3 viral infections (39% and 35%, respectively, P=0.729), fungal infections (0% and 5%, respectively, P=1.0), or non-microbiologically defined infections (0% and 17%, respectively, P=0.072).
In the first 100 days after transplant, patients in the NiCord group spent significantly more days out of the hospital than patients in the control group. The median number of days for each group was 74 and 53, respectively (P=0.002).
“These results demonstrate that the rapid hematopoietic recovery from NiCord transplantation results in clinical benefit, in comparison to similar site controls,” Dr Horwitz concluded.

Photo courtesy of NHS
VALENCIA, SPAIN—The expanded umbilical cord blood (UCB) product NiCord can provide clinical benefits in patients with high-risk hematologic malignancies, according to data presented at the 42nd Annual Meeting of the European Society for Blood and Marrow Transplantation.
NiCord consists of cells from a single UCB unit cultured in nicotinamide—a vitamin B derivative—and cytokines that are typically used for expansion—thrombopoietin, interleukin 6, FLT3 ligand, and stem cell factor.
The data showed that patients transplanted with NiCord had fewer moderate to severe bacterial infections and shorter hospital stays than patients who received standard UCB transplants.
“We saw a significant reduction in serious bacterial infections during the first 100 days in the NiCord group,” said Mitchell Horwitz, MD, of the Duke University School of Medicine in Durham, North Carolina.
“This is encouraging because this type of infection is a major cause of early death following UCB transplantation. We also saw a significant reduction in hospitalization time in the NiCord group, indicating a faster recovery of these patients in comparison to those transplanted with standard umbilical cord blood.”
These results were presented at the meeting as abstract O090. The research was funded by Gamida Cell, the company developing NiCord.
Dr Horwitz and his colleagues analyzed 18 patients with high-risk hematologic malignancies—most with acute leukemia or myelodysplastic syndromes (90%)—who were transplanted with NiCord.
Ten of the patients received NiCord with a second, unmanipulated UCB unit, and 8 patients received NiCord as a single UCB graft.
The researchers compared these patients to 101 patients who received standard single or double UCB transplants at Duke University from January 2005 to March 2015.
Patients in both groups received a total body irradiation-based myeloablative preparative regimen.
The median time to neutrophil engraftment was significantly shorter in the NiCord group than the control group—12.5 days and 27 days, respectively (P<0.001).
All 18 patients in the Nicord group and 100 patients in the control group had at least 1 infection.
Patients in the NiCord group had a significantly lower incidence of grade 2-3 bacterial infections than patients in the control group—22% and 54%, respectively (P=0.015).
However, there was no significant difference between the groups with regard to grade 2-3 viral infections (39% and 35%, respectively, P=0.729), fungal infections (0% and 5%, respectively, P=1.0), or non-microbiologically defined infections (0% and 17%, respectively, P=0.072).
In the first 100 days after transplant, patients in the NiCord group spent significantly more days out of the hospital than patients in the control group. The median number of days for each group was 74 and 53, respectively (P=0.002).
“These results demonstrate that the rapid hematopoietic recovery from NiCord transplantation results in clinical benefit, in comparison to similar site controls,” Dr Horwitz concluded.
Subgroup benefits from long-term DAPT

Photo courtesy of AstraZeneca
CHICAGO—Long-term use of dual antiplatelet therapy (DAPT) can benefit patients with a history of myocardial infarction (MI) and peripheral artery disease (PAD), according to data presented at the American College of Cardiology’s 65th Annual Scientific Session.
The data were from a subanalysis of the PEGASUS-TIMI 54 trial, in which researchers evaluated long-term use of aspirin, with or without the antiplatelet agent ticagrelor, in patients with a history of MI and at least 1 additional risk factor for thrombotic cardiovascular (CV) events.
The analysis suggested that, in stable patients with a history of MI, concomitant PAD is associated with a higher risk of major adverse cardiac events (MACE).
However, long-term DAPT with ticagrelor and aspirin can reduce the incidence of MACE in these patients, when compared to aspirin plus a placebo.
These results were presented as abstract 907-04 and simultaneously published in the Journal of American College of Cardiology. The trial was sponsored by AstraZeneca, the company developing ticagrelor.
Patients in the PEGASUS-TIMI 54 trial were randomized to receive aspirin plus twice-daily doses of ticagrelor at 90 mg, ticagrelor at 60 mg, or placebo.
The study’s primary efficacy endpoint was the incidence of MACE, which was defined as a composite of CV death, MI, or stroke.
The subanalysis showed that the 1143 patients with a prior MI and PAD had a higher incidence of MACE at 3 years than patients without PAD—19.3% and 8.4%, respectively (P<0.001).
The increased risk of MACE in patients with PAD persisted after the researchers adjusted for differences in patient characteristics at baseline. The hazard ratio (HR) was 1.60 (95% CI 1.20-2.13, P=0.0013).
Patients with PAD had a higher risk of CV death (HR 1.84, 95% CI 1.16-2.94, P=0.0102), stroke (HR 2.31, 95% CI 1.26–4.25, P=0.0071), and mortality (HR 2.05, 95% CI 1.43-2.94, P<0.001) than patients without PAD.
Patients who received DAPT (ticagrelor at either dose plus aspirin) had a lower risk of MACE at 3 years than patients who received placebo plus aspirin. This was true for patients with PAD (HR 0.75, 95% CI 0.55-1.01) and without it (HR 0.86, 95% CI 0.77-0.96, P-interaction=0.41).
However, because of their higher absolute risk of MACE, patients with PAD had a greater absolute risk reduction (4.1%) than patients without PAD.
The risk of TIMI major bleeding was not significantly higher in patients with PAD than in those without it (HR 1.57, 95% CI 0.47-5.22, P=0.46).
For patients with PAD, TIMI major bleeding occurred more frequently with ticagrelor at 90 mg plus aspirin than with placebo plus aspirin (HR 1.46, 95% CI 0.39-5.43, P=0.57) and with ticagrelor at 60 mg plus aspirin than with placebo plus aspirin (HR 1.18, 95% CI 0.29-4.70, P=0.82), though the differences were not significant.
“Patients with prior MI and PAD are at further heightened risk of ischemic events relative to patients with prior MI and no PAD, even when accounting for other risk factors,” said study investigator Marc Bonaca, MD, of Brigham and Women’s Hospital in Boston, Massachusetts.
“Because of their heightened ischemic risk, patients in the subgroup analysis with a prior MI and PAD appear to have a higher absolute risk reduction with ticagrelor than those without. These findings may be helpful to clinicians in identifying patients with prior MI who they feel could benefit from prolonged therapy with ticagrelor.”

Photo courtesy of AstraZeneca
CHICAGO—Long-term use of dual antiplatelet therapy (DAPT) can benefit patients with a history of myocardial infarction (MI) and peripheral artery disease (PAD), according to data presented at the American College of Cardiology’s 65th Annual Scientific Session.
The data were from a subanalysis of the PEGASUS-TIMI 54 trial, in which researchers evaluated long-term use of aspirin, with or without the antiplatelet agent ticagrelor, in patients with a history of MI and at least 1 additional risk factor for thrombotic cardiovascular (CV) events.
The analysis suggested that, in stable patients with a history of MI, concomitant PAD is associated with a higher risk of major adverse cardiac events (MACE).
However, long-term DAPT with ticagrelor and aspirin can reduce the incidence of MACE in these patients, when compared to aspirin plus a placebo.
These results were presented as abstract 907-04 and simultaneously published in the Journal of American College of Cardiology. The trial was sponsored by AstraZeneca, the company developing ticagrelor.
Patients in the PEGASUS-TIMI 54 trial were randomized to receive aspirin plus twice-daily doses of ticagrelor at 90 mg, ticagrelor at 60 mg, or placebo.
The study’s primary efficacy endpoint was the incidence of MACE, which was defined as a composite of CV death, MI, or stroke.
The subanalysis showed that the 1143 patients with a prior MI and PAD had a higher incidence of MACE at 3 years than patients without PAD—19.3% and 8.4%, respectively (P<0.001).
The increased risk of MACE in patients with PAD persisted after the researchers adjusted for differences in patient characteristics at baseline. The hazard ratio (HR) was 1.60 (95% CI 1.20-2.13, P=0.0013).
Patients with PAD had a higher risk of CV death (HR 1.84, 95% CI 1.16-2.94, P=0.0102), stroke (HR 2.31, 95% CI 1.26–4.25, P=0.0071), and mortality (HR 2.05, 95% CI 1.43-2.94, P<0.001) than patients without PAD.
Patients who received DAPT (ticagrelor at either dose plus aspirin) had a lower risk of MACE at 3 years than patients who received placebo plus aspirin. This was true for patients with PAD (HR 0.75, 95% CI 0.55-1.01) and without it (HR 0.86, 95% CI 0.77-0.96, P-interaction=0.41).
However, because of their higher absolute risk of MACE, patients with PAD had a greater absolute risk reduction (4.1%) than patients without PAD.
The risk of TIMI major bleeding was not significantly higher in patients with PAD than in those without it (HR 1.57, 95% CI 0.47-5.22, P=0.46).
For patients with PAD, TIMI major bleeding occurred more frequently with ticagrelor at 90 mg plus aspirin than with placebo plus aspirin (HR 1.46, 95% CI 0.39-5.43, P=0.57) and with ticagrelor at 60 mg plus aspirin than with placebo plus aspirin (HR 1.18, 95% CI 0.29-4.70, P=0.82), though the differences were not significant.
“Patients with prior MI and PAD are at further heightened risk of ischemic events relative to patients with prior MI and no PAD, even when accounting for other risk factors,” said study investigator Marc Bonaca, MD, of Brigham and Women’s Hospital in Boston, Massachusetts.
“Because of their heightened ischemic risk, patients in the subgroup analysis with a prior MI and PAD appear to have a higher absolute risk reduction with ticagrelor than those without. These findings may be helpful to clinicians in identifying patients with prior MI who they feel could benefit from prolonged therapy with ticagrelor.”

Photo courtesy of AstraZeneca
CHICAGO—Long-term use of dual antiplatelet therapy (DAPT) can benefit patients with a history of myocardial infarction (MI) and peripheral artery disease (PAD), according to data presented at the American College of Cardiology’s 65th Annual Scientific Session.
The data were from a subanalysis of the PEGASUS-TIMI 54 trial, in which researchers evaluated long-term use of aspirin, with or without the antiplatelet agent ticagrelor, in patients with a history of MI and at least 1 additional risk factor for thrombotic cardiovascular (CV) events.
The analysis suggested that, in stable patients with a history of MI, concomitant PAD is associated with a higher risk of major adverse cardiac events (MACE).
However, long-term DAPT with ticagrelor and aspirin can reduce the incidence of MACE in these patients, when compared to aspirin plus a placebo.
These results were presented as abstract 907-04 and simultaneously published in the Journal of American College of Cardiology. The trial was sponsored by AstraZeneca, the company developing ticagrelor.
Patients in the PEGASUS-TIMI 54 trial were randomized to receive aspirin plus twice-daily doses of ticagrelor at 90 mg, ticagrelor at 60 mg, or placebo.
The study’s primary efficacy endpoint was the incidence of MACE, which was defined as a composite of CV death, MI, or stroke.
The subanalysis showed that the 1143 patients with a prior MI and PAD had a higher incidence of MACE at 3 years than patients without PAD—19.3% and 8.4%, respectively (P<0.001).
The increased risk of MACE in patients with PAD persisted after the researchers adjusted for differences in patient characteristics at baseline. The hazard ratio (HR) was 1.60 (95% CI 1.20-2.13, P=0.0013).
Patients with PAD had a higher risk of CV death (HR 1.84, 95% CI 1.16-2.94, P=0.0102), stroke (HR 2.31, 95% CI 1.26–4.25, P=0.0071), and mortality (HR 2.05, 95% CI 1.43-2.94, P<0.001) than patients without PAD.
Patients who received DAPT (ticagrelor at either dose plus aspirin) had a lower risk of MACE at 3 years than patients who received placebo plus aspirin. This was true for patients with PAD (HR 0.75, 95% CI 0.55-1.01) and without it (HR 0.86, 95% CI 0.77-0.96, P-interaction=0.41).
However, because of their higher absolute risk of MACE, patients with PAD had a greater absolute risk reduction (4.1%) than patients without PAD.
The risk of TIMI major bleeding was not significantly higher in patients with PAD than in those without it (HR 1.57, 95% CI 0.47-5.22, P=0.46).
For patients with PAD, TIMI major bleeding occurred more frequently with ticagrelor at 90 mg plus aspirin than with placebo plus aspirin (HR 1.46, 95% CI 0.39-5.43, P=0.57) and with ticagrelor at 60 mg plus aspirin than with placebo plus aspirin (HR 1.18, 95% CI 0.29-4.70, P=0.82), though the differences were not significant.
“Patients with prior MI and PAD are at further heightened risk of ischemic events relative to patients with prior MI and no PAD, even when accounting for other risk factors,” said study investigator Marc Bonaca, MD, of Brigham and Women’s Hospital in Boston, Massachusetts.
“Because of their heightened ischemic risk, patients in the subgroup analysis with a prior MI and PAD appear to have a higher absolute risk reduction with ticagrelor than those without. These findings may be helpful to clinicians in identifying patients with prior MI who they feel could benefit from prolonged therapy with ticagrelor.”