User login
Should immediate cord clamping be performed for preterm infants?
WHAT DOES THIS MEAN FOR PRACTICE?
- Evaluate carefully between which babies require immediate resuscitation and which babies can have cord clamping delayed
- Evaluation requires patience on the part of the OB, neonatologist, and nurse resuscitating team
- Close collaboration among all team members is required
Does maternal sleep position affect risk of stillbirth?
WHAT DOES THIS MEAN FOR PRACTICE?
Encourage pregnant patients to not go to sleep in the supine position, especially those who:
- are obese
- have medical complications of pregnancy
- have a history of prior stillbirth
- smoke
- are of advanced maternal age
What is the optimal opioid prescription length after women’s health surgical procedures?
WHAT DOES THIS MEAN FOR PRACTICE?
- 7-day opioid prescriptions should be sufficient after common gyn procedures
- Monitor patients closely
- Transfer patients as soon as possible to non-opioid pain medication
What is the ideal treatment timing for bisphosphonate therapy?
WHAT DOES THIS MEAN FOR PRACTICE?
Oral bisphosphonates remain in our armamentarium for reducing fracture in postmenopausal patients
Ideal duration of therapy remains unclear
Counsel patients: up to 2 y of therapy reduces fracture risk; >10 y of therapy may increase fracture risk
2017 Update on obstetrics
In this Update we discuss several exciting new recommendations for preventive treatments in pregnancy and prenatal diagnostic tests. Our A-to-Z coverage includes:
- antenatal steroids in late preterm pregnancy
- expanded list of high-risk conditions warranting low-dose aspirin for preeclampsia prevention
- chromosomal microarray analysis versus karyotype for specific clinical situations
- Zika virus infection evolving information.
Next: New recommendation for timing of late preterm antenatal steroids
New recommendation offered for timing of late preterm antenatal steroids
Gyamfi-Bannerman C, Thom EA, Blackwell SC, et al; for the NICHD Maternal-Fetal Medicine Units Network. Antenatal betamethasone for women at risk for late preterm delivery. N Engl J Med. 2016;374(14):1311-1320.
American College of Obstetricians and Gynecologists. Committee Opinion No. 677. Antenatal corticosteroidtherapy for fetal maturation. Obstet Gynecol. 2016;128(4):e187-e194.
Kamath-Rayne BD, Rozance PJ, Goldenberg RL, Jobe AH. Antenatal corticosteroids beyond 34 weeks gestation: what do we do now? Am J Obstet Gynecol. 2016;215(4):423-430.
A dramatic recommendation for obstetric practice change occurred in 2016: the option of administering antenatal steroids for fetal lung maturity after 34 weeks. In the Antenatal Late Preterm Steroids (ALPS) trial of betamethasone in the late preterm period in patients at "high risk" of imminent delivery, Gyamfi-Bannerman and colleagues demonstrated that the treated group had a significant decrease in the rate of neonatal respiratory complications.
The primary outcome, a composite of respiratory morbidities (including transient tachypnea of the newborn, surfactant use, and need for resuscitation at birth) within the first 72 hours of life, had significant differences between groups, occurring in 165 of 1,427 infants (11.6%) in the betamethasone-treated group and 202 of 1,400 (14.4%) in the placebo group (relative risk in the betamethasone group, 0.80; 95% confidence interval, 0.66-0.97; P = .02). However, there was no statistically significant difference in respiratory distress syndrome, apnea, or pneumonia between groups, and the significant difference noted in bronchopulmonary dysplasia was based on a total number of 11 cases.
In response to these findings, both the American College of Obstetricians and Gynecologists (ACOG) and the Society for Maternal-Fetal Medicine (SMFM) released practice advisories and interim updates, culminating in a final recommendation for a single course of betamethasone in patients at high risk of preterm delivery between 34 and 36 6/7 weeks who have not received a previous course.
Related article:
When could use of antenatal corticosteroids in the late preterm birth period be beneficial?
In a thorough review of the literature on antenatal steroid use, Kamath-Rayne and colleagues highlighted several factors that should be considered before adopting universal use of steroids at >34 weeks. These include:
- The definition of "high risk of imminent delivery" as preterm labor with at least 3-cm dilation or 75% effacement, or spontaneous rupture of membranes. The effect of less stringent inclusion criteria in real-world clinical practice is not known, and many patients who will go on to deliver at term will receive steroids unnecessarily.
- Multiple gestation, patients with pre-existing diabetes, women who had previously received a course of steroids, and fetuses with anomalies were excluded from the ALPS study. Use of antenatal steroids in these groups at >34 weeks should be evaluated before universal adoption.
Related article:
What is the ideal gestational age for twin delivery to minimize perinatal deaths?
- The incidence of neonatal hypoglycemia in the treated group was significantly increased. This affects our colleagues in pediatrics considerably from a systems standpoint (need for changes to newborn protocols and communication between services).
- The long-term outcomes of patients exposed to steroids in the late preterm period are yet to be delineated, specifically, the potential neurodevelopmental effects of a medication known to alter preterm brain development as well as cardiovascular and metabolic consequences.
Next: Low-dose aspirin for reducing preeclampsia risk
Low-dose aspirin clearly is effective for reducing the risk of preeclampsia
American College of Obstetricians and Gynecologists. Hypertension in pregnancy. Report of the American College of Obstetricians and Gynecologists' Task Force on Hypertension in Pregnancy. Obstet Gynecol. 2013;122(5):1122-1131.
Henderson JT, Whitlock EP, O'Connor E, Senger CA, Thompson JH, Rowland MG. Low-dose aspirin for prevention of morbidity and mortality from preeclampsia: a systematic evidence review for the US Preventive Services Task Force. Ann Intern Med. 2014;160(10):695-703.
LeFevre ML; US Preventive Services Task Force. Low-dose aspirin use for the prevention of morbidity and mortality from preeclampsia: US Preventive Services Task Force recommendation statement. Ann Intern Med. 2014;161(11):819-826.
American College of Obstetricians and Gynecologists. Practice advisory on low-dose aspirin and prevention of preeclampsia: updated recommendations. http://www.acog.org/About-ACOG/News-Room/Practice-Advisories/Practice-Advisory-Low-Dose-Aspirin-and-Prevention-of-Preeclampsia-Updated-Recommendations. Published July 11, 2016. Accessed December 6, 2016.
In the 2013 ACOG Task Force on Hypertension in Pregnancy report, low-dose aspirin (60-80 mg) was recommended to be initiated in the late first trimester to reduce preeclampsia risk for women with:
- prior early onset preeclampsia with preterm delivery at <34 weeks' gestation, or
- preeclampsia in more than one prior pregnancy.
This recommendation was based on several meta-analyses that demonstrated a 10% to 17% reduction in risk with no increase in bleeding, placental abruption, or other adverse events.
In 2014, the US Preventive Services Task Force (USPSTF) conducted a systematic evidence review of low-dose aspirin use for prevention of morbidity and mortality from preeclampsia. That report revealed a 24% risk reduction of preeclampsia in high-risk women treated with low-dose aspirin, as well as a 14% reduction in preterm birth and a 20% reduction in fetal growth restriction. A final statement from the USPSTF in 2014 recommended low-dose aspirin (60-150 mg) starting between 12 and 28 weeks' gestation for women at "high" risk who have:
- a history of preeclampsia, especially if accompanied by an adverse outcome
- multifetal gestation
- chronic hypertension
- diabetes (type 1 or type 2)
- renal disease
- autoimmune disease (such as systematic lupus erythematosus, antiphospholipid syndrome).
Related article:
Start offering aspirin to pregnant women at high risk for preeclampsia
As of July 11, 2016, ACOG supports this expanded list of high-risk conditions. Additionally, the USPSTF identified a "moderate" risk group in which low-dose aspirin may be considered if a patient has several risk factors, such as obesity, nulliparity, family history of preeclampsia, age 35 years or older, or another poor pregnancy outcome. ACOG notes, however, that the evidence supporting this practice is uncertain and does not make a recommendation regarding aspirin use in this population. Further study should be conducted to determine the benefit of low-dose aspirin in these patients as well as the long-term effects of treatment on maternal and child outcomes.
Next: CMA for prenatal genetic diagnosis
Chromosomal microarray analysis is preferable to karyotype in certain situations
Pauli JM, Repke JT. Update on obstetrics. OBG Manag. 2013;25(1):28-32.
Society for Maternal-Fetal Medicine (SMFM), Dugoff L, Norton ME, Kuller JA. The use of chromosomal microarray for prenatal diagnosis. Am J Obstet Gynecol. 2016;215(4):B2-B9.
American College of Obstetricians and Gynecologists. Committee Opinion No. 682. Microarrays and next- generation sequencing technology: the use of advanced genetic diagnostic tools in obstetrics and gynecology.Obstet Gynecol. 2016;128(6):e262-e268.
We previously addressed the use of chromosomal microarray analysis (CMA) for prenatal diagnosis in our 2013 "Update on obstetrics," specifically, the question of whether CMA could replace karyotype. The main differences between karyotype and CMA are that 1) only karyotype can detect balanced translocations/inversions and 2) only CMA can detect copy number variants (CNV). There are some differences in the technology and capabilities of the 2 types of CMA currently available as well.
In our 2013 article we concluded that "The total costs of such an approach--test, interpretation, counseling, and long-term follow-up of uncertain results--are unknown at this time and may prove to be unaffordable on a population-wide basis." Today, the cost of CMA is still higher than karyotype, but it is expected to decrease and insurance coverage for this test is expected to increase.
Related article:
Cell-free DNA screening for women at low risk for fetal aneuploidy
Both SMFM and ACOG released recommendations in 2016 regarding the use of CMA in prenatal genetic diagnosis, summarized as follows:
- CMA is recommended over karyotype for fetuses with structural abnormalities on ultrasound
- The detection rate for clinically relevant abnormal CNVs in this population is about 6%
- CMA is recommended for diagnosis for stillbirth specimens
- CMA does not require dividing cells and may be a quicker and more reliable test in this population
- Karotype or fluorescence in situ hybridization (FISH) is recommended for fetuses with ultrasound findings suggestive of aneuploidy
- If it is negative, then CMA is recommended
- Karyotype or CMA is recommended for patients desiring prenatal diagnostic testing with a normal fetal ultrasound
- The detection rate for clinically relevant CNVs in this population (advanced maternal age, abnormal serum screening, prior aneuploidy, parental anxiety) is about 1%
- Pretest and posttest counseling about the limitations of CMA and a 2% risk of detection of variants of unknown significance (VUS) should be performed by a provider who has expertise in CMA and who has access to databases with genotype/phenotype information for VUS
- This counseling should also include the possibility of diagnosis of nonpaternity, consanguinity, and adult-onset disease
- Karyotype is recommended for couples with recurrent pregnancy loss
- The identification of balanced translocations in this population is most relevant in this patient population
- Prenatal diagnosis with routine use of whole-genome or whole-exome sequencing is not recommended.
Next: Zika virus: Check for updates
Zika virus infection: Check often for the latest updates
American College of Obstetricians and Gynecologists, Society for Maternal-Fetal Medicine. Practice advisory on Zika virus. http://www.acog.org/About-ACOG/News-Room/Practice-Advisories/Practice-Advisory-Interim-Guidance-for-Care-of-Obstetric-Patients- During-a-Zika-Virus-Outbreak. Published December 5, 2016. Accessed December 6, 2016.
Centers for Disease Control and Prevention. Zika virus. http://www.cdc.gov/zika/pregnancy/index.html. Updated August 22, 2016. Accessed December 6, 2016.
Petersen EE, Meaney-Delman D, Neblett-Fanfair R, et al. Update: interim guidance for preconception counseling and prevention of sexual transmission of Zika virus for persons with possible Zika virus exposure--United States, September 2016. MMWR Morbid Mortal Wkly Rep. 2016;65(39):1077-1081.
A yearly update on obstetrics would be remiss without mention of the Zika virus and its impact on pregnancy and reproduction. That being said, any recommendations we offer may be out of date by the time this article is published given the rapidly changing picture of Zika virus since it first dominated the headlines in 2016. Here are the basics as summarized from ACOG and the Centers for Disease Control and Prevention (CDC):
Viral spread. Zika virus may be spread in several ways: by an infected Aedes species mosquito, mother to fetus, sexual contact, blood transfusion, or laboratory exposure.
Symptoms of infection include conjunctivitis, fever, rash, and arthralgia, but most patients (4/5) are asymptomatic.
Sequelae. Zika virus infection during pregnancy is believed to cause fetal and neonatal microcephaly, intracranial calcifications, and brain and eye abnormalities. The rate of these findings in infected individuals, as well as the rate of vertical transmission, is not known.
Travel advisory. Pregnant women should not travel to areas with active Zika infection (the CDC website regularly updates these restricted areas).
Preventive measures. If traveling to an area of active Zika infection, pregnant women should take preventative measures day and night against mosquito bites, such as use of insect repellents approved by the Environmental Protection Agency, clothing that covers exposed skin, and staying indoors.
Safe sex. Abstinence or consistent condom use is recommended for pregnant women with partners who travel to or live in areas of active Zika infection.
Delay conception. Conception should be postponed for at least 6 months in men with Zika infection and at least 8 weeks in women with Zika infection.
Testing recommendations. Pregnant women with Zika virus exposure should be tested, regardless of symptoms. Symptomatic exposed nonpregnant women and all men should be tested.
Prenatal surveillance. High-risk consultation and serial ultrasounds for fetal anatomy and growth should be considered in patients with Zika virus infection during pregnancy. Amniocentesis can be considered on a case-by-case basis.
Related article:
Zika virus update: A rapidly moving target
Share your thoughts! Send your Letter to the Editor to [email protected]. Please include your name and the city and state in which you practice.
In this Update we discuss several exciting new recommendations for preventive treatments in pregnancy and prenatal diagnostic tests. Our A-to-Z coverage includes:
- antenatal steroids in late preterm pregnancy
- expanded list of high-risk conditions warranting low-dose aspirin for preeclampsia prevention
- chromosomal microarray analysis versus karyotype for specific clinical situations
- Zika virus infection evolving information.
Next: New recommendation for timing of late preterm antenatal steroids
New recommendation offered for timing of late preterm antenatal steroids
Gyamfi-Bannerman C, Thom EA, Blackwell SC, et al; for the NICHD Maternal-Fetal Medicine Units Network. Antenatal betamethasone for women at risk for late preterm delivery. N Engl J Med. 2016;374(14):1311-1320.
American College of Obstetricians and Gynecologists. Committee Opinion No. 677. Antenatal corticosteroidtherapy for fetal maturation. Obstet Gynecol. 2016;128(4):e187-e194.
Kamath-Rayne BD, Rozance PJ, Goldenberg RL, Jobe AH. Antenatal corticosteroids beyond 34 weeks gestation: what do we do now? Am J Obstet Gynecol. 2016;215(4):423-430.
A dramatic recommendation for obstetric practice change occurred in 2016: the option of administering antenatal steroids for fetal lung maturity after 34 weeks. In the Antenatal Late Preterm Steroids (ALPS) trial of betamethasone in the late preterm period in patients at "high risk" of imminent delivery, Gyamfi-Bannerman and colleagues demonstrated that the treated group had a significant decrease in the rate of neonatal respiratory complications.
The primary outcome, a composite of respiratory morbidities (including transient tachypnea of the newborn, surfactant use, and need for resuscitation at birth) within the first 72 hours of life, had significant differences between groups, occurring in 165 of 1,427 infants (11.6%) in the betamethasone-treated group and 202 of 1,400 (14.4%) in the placebo group (relative risk in the betamethasone group, 0.80; 95% confidence interval, 0.66-0.97; P = .02). However, there was no statistically significant difference in respiratory distress syndrome, apnea, or pneumonia between groups, and the significant difference noted in bronchopulmonary dysplasia was based on a total number of 11 cases.
In response to these findings, both the American College of Obstetricians and Gynecologists (ACOG) and the Society for Maternal-Fetal Medicine (SMFM) released practice advisories and interim updates, culminating in a final recommendation for a single course of betamethasone in patients at high risk of preterm delivery between 34 and 36 6/7 weeks who have not received a previous course.
Related article:
When could use of antenatal corticosteroids in the late preterm birth period be beneficial?
In a thorough review of the literature on antenatal steroid use, Kamath-Rayne and colleagues highlighted several factors that should be considered before adopting universal use of steroids at >34 weeks. These include:
- The definition of "high risk of imminent delivery" as preterm labor with at least 3-cm dilation or 75% effacement, or spontaneous rupture of membranes. The effect of less stringent inclusion criteria in real-world clinical practice is not known, and many patients who will go on to deliver at term will receive steroids unnecessarily.
- Multiple gestation, patients with pre-existing diabetes, women who had previously received a course of steroids, and fetuses with anomalies were excluded from the ALPS study. Use of antenatal steroids in these groups at >34 weeks should be evaluated before universal adoption.
Related article:
What is the ideal gestational age for twin delivery to minimize perinatal deaths?
- The incidence of neonatal hypoglycemia in the treated group was significantly increased. This affects our colleagues in pediatrics considerably from a systems standpoint (need for changes to newborn protocols and communication between services).
- The long-term outcomes of patients exposed to steroids in the late preterm period are yet to be delineated, specifically, the potential neurodevelopmental effects of a medication known to alter preterm brain development as well as cardiovascular and metabolic consequences.
Next: Low-dose aspirin for reducing preeclampsia risk
Low-dose aspirin clearly is effective for reducing the risk of preeclampsia
American College of Obstetricians and Gynecologists. Hypertension in pregnancy. Report of the American College of Obstetricians and Gynecologists' Task Force on Hypertension in Pregnancy. Obstet Gynecol. 2013;122(5):1122-1131.
Henderson JT, Whitlock EP, O'Connor E, Senger CA, Thompson JH, Rowland MG. Low-dose aspirin for prevention of morbidity and mortality from preeclampsia: a systematic evidence review for the US Preventive Services Task Force. Ann Intern Med. 2014;160(10):695-703.
LeFevre ML; US Preventive Services Task Force. Low-dose aspirin use for the prevention of morbidity and mortality from preeclampsia: US Preventive Services Task Force recommendation statement. Ann Intern Med. 2014;161(11):819-826.
American College of Obstetricians and Gynecologists. Practice advisory on low-dose aspirin and prevention of preeclampsia: updated recommendations. http://www.acog.org/About-ACOG/News-Room/Practice-Advisories/Practice-Advisory-Low-Dose-Aspirin-and-Prevention-of-Preeclampsia-Updated-Recommendations. Published July 11, 2016. Accessed December 6, 2016.
In the 2013 ACOG Task Force on Hypertension in Pregnancy report, low-dose aspirin (60-80 mg) was recommended to be initiated in the late first trimester to reduce preeclampsia risk for women with:
- prior early onset preeclampsia with preterm delivery at <34 weeks' gestation, or
- preeclampsia in more than one prior pregnancy.
This recommendation was based on several meta-analyses that demonstrated a 10% to 17% reduction in risk with no increase in bleeding, placental abruption, or other adverse events.
In 2014, the US Preventive Services Task Force (USPSTF) conducted a systematic evidence review of low-dose aspirin use for prevention of morbidity and mortality from preeclampsia. That report revealed a 24% risk reduction of preeclampsia in high-risk women treated with low-dose aspirin, as well as a 14% reduction in preterm birth and a 20% reduction in fetal growth restriction. A final statement from the USPSTF in 2014 recommended low-dose aspirin (60-150 mg) starting between 12 and 28 weeks' gestation for women at "high" risk who have:
- a history of preeclampsia, especially if accompanied by an adverse outcome
- multifetal gestation
- chronic hypertension
- diabetes (type 1 or type 2)
- renal disease
- autoimmune disease (such as systematic lupus erythematosus, antiphospholipid syndrome).
Related article:
Start offering aspirin to pregnant women at high risk for preeclampsia
As of July 11, 2016, ACOG supports this expanded list of high-risk conditions. Additionally, the USPSTF identified a "moderate" risk group in which low-dose aspirin may be considered if a patient has several risk factors, such as obesity, nulliparity, family history of preeclampsia, age 35 years or older, or another poor pregnancy outcome. ACOG notes, however, that the evidence supporting this practice is uncertain and does not make a recommendation regarding aspirin use in this population. Further study should be conducted to determine the benefit of low-dose aspirin in these patients as well as the long-term effects of treatment on maternal and child outcomes.
Next: CMA for prenatal genetic diagnosis
Chromosomal microarray analysis is preferable to karyotype in certain situations
Pauli JM, Repke JT. Update on obstetrics. OBG Manag. 2013;25(1):28-32.
Society for Maternal-Fetal Medicine (SMFM), Dugoff L, Norton ME, Kuller JA. The use of chromosomal microarray for prenatal diagnosis. Am J Obstet Gynecol. 2016;215(4):B2-B9.
American College of Obstetricians and Gynecologists. Committee Opinion No. 682. Microarrays and next- generation sequencing technology: the use of advanced genetic diagnostic tools in obstetrics and gynecology.Obstet Gynecol. 2016;128(6):e262-e268.
We previously addressed the use of chromosomal microarray analysis (CMA) for prenatal diagnosis in our 2013 "Update on obstetrics," specifically, the question of whether CMA could replace karyotype. The main differences between karyotype and CMA are that 1) only karyotype can detect balanced translocations/inversions and 2) only CMA can detect copy number variants (CNV). There are some differences in the technology and capabilities of the 2 types of CMA currently available as well.
In our 2013 article we concluded that "The total costs of such an approach--test, interpretation, counseling, and long-term follow-up of uncertain results--are unknown at this time and may prove to be unaffordable on a population-wide basis." Today, the cost of CMA is still higher than karyotype, but it is expected to decrease and insurance coverage for this test is expected to increase.
Related article:
Cell-free DNA screening for women at low risk for fetal aneuploidy
Both SMFM and ACOG released recommendations in 2016 regarding the use of CMA in prenatal genetic diagnosis, summarized as follows:
- CMA is recommended over karyotype for fetuses with structural abnormalities on ultrasound
- The detection rate for clinically relevant abnormal CNVs in this population is about 6%
- CMA is recommended for diagnosis for stillbirth specimens
- CMA does not require dividing cells and may be a quicker and more reliable test in this population
- Karotype or fluorescence in situ hybridization (FISH) is recommended for fetuses with ultrasound findings suggestive of aneuploidy
- If it is negative, then CMA is recommended
- Karyotype or CMA is recommended for patients desiring prenatal diagnostic testing with a normal fetal ultrasound
- The detection rate for clinically relevant CNVs in this population (advanced maternal age, abnormal serum screening, prior aneuploidy, parental anxiety) is about 1%
- Pretest and posttest counseling about the limitations of CMA and a 2% risk of detection of variants of unknown significance (VUS) should be performed by a provider who has expertise in CMA and who has access to databases with genotype/phenotype information for VUS
- This counseling should also include the possibility of diagnosis of nonpaternity, consanguinity, and adult-onset disease
- Karyotype is recommended for couples with recurrent pregnancy loss
- The identification of balanced translocations in this population is most relevant in this patient population
- Prenatal diagnosis with routine use of whole-genome or whole-exome sequencing is not recommended.
Next: Zika virus: Check for updates
Zika virus infection: Check often for the latest updates
American College of Obstetricians and Gynecologists, Society for Maternal-Fetal Medicine. Practice advisory on Zika virus. http://www.acog.org/About-ACOG/News-Room/Practice-Advisories/Practice-Advisory-Interim-Guidance-for-Care-of-Obstetric-Patients- During-a-Zika-Virus-Outbreak. Published December 5, 2016. Accessed December 6, 2016.
Centers for Disease Control and Prevention. Zika virus. http://www.cdc.gov/zika/pregnancy/index.html. Updated August 22, 2016. Accessed December 6, 2016.
Petersen EE, Meaney-Delman D, Neblett-Fanfair R, et al. Update: interim guidance for preconception counseling and prevention of sexual transmission of Zika virus for persons with possible Zika virus exposure--United States, September 2016. MMWR Morbid Mortal Wkly Rep. 2016;65(39):1077-1081.
A yearly update on obstetrics would be remiss without mention of the Zika virus and its impact on pregnancy and reproduction. That being said, any recommendations we offer may be out of date by the time this article is published given the rapidly changing picture of Zika virus since it first dominated the headlines in 2016. Here are the basics as summarized from ACOG and the Centers for Disease Control and Prevention (CDC):
Viral spread. Zika virus may be spread in several ways: by an infected Aedes species mosquito, mother to fetus, sexual contact, blood transfusion, or laboratory exposure.
Symptoms of infection include conjunctivitis, fever, rash, and arthralgia, but most patients (4/5) are asymptomatic.
Sequelae. Zika virus infection during pregnancy is believed to cause fetal and neonatal microcephaly, intracranial calcifications, and brain and eye abnormalities. The rate of these findings in infected individuals, as well as the rate of vertical transmission, is not known.
Travel advisory. Pregnant women should not travel to areas with active Zika infection (the CDC website regularly updates these restricted areas).
Preventive measures. If traveling to an area of active Zika infection, pregnant women should take preventative measures day and night against mosquito bites, such as use of insect repellents approved by the Environmental Protection Agency, clothing that covers exposed skin, and staying indoors.
Safe sex. Abstinence or consistent condom use is recommended for pregnant women with partners who travel to or live in areas of active Zika infection.
Delay conception. Conception should be postponed for at least 6 months in men with Zika infection and at least 8 weeks in women with Zika infection.
Testing recommendations. Pregnant women with Zika virus exposure should be tested, regardless of symptoms. Symptomatic exposed nonpregnant women and all men should be tested.
Prenatal surveillance. High-risk consultation and serial ultrasounds for fetal anatomy and growth should be considered in patients with Zika virus infection during pregnancy. Amniocentesis can be considered on a case-by-case basis.
Related article:
Zika virus update: A rapidly moving target
Share your thoughts! Send your Letter to the Editor to [email protected]. Please include your name and the city and state in which you practice.
In this Update we discuss several exciting new recommendations for preventive treatments in pregnancy and prenatal diagnostic tests. Our A-to-Z coverage includes:
- antenatal steroids in late preterm pregnancy
- expanded list of high-risk conditions warranting low-dose aspirin for preeclampsia prevention
- chromosomal microarray analysis versus karyotype for specific clinical situations
- Zika virus infection evolving information.
Next: New recommendation for timing of late preterm antenatal steroids
New recommendation offered for timing of late preterm antenatal steroids
Gyamfi-Bannerman C, Thom EA, Blackwell SC, et al; for the NICHD Maternal-Fetal Medicine Units Network. Antenatal betamethasone for women at risk for late preterm delivery. N Engl J Med. 2016;374(14):1311-1320.
American College of Obstetricians and Gynecologists. Committee Opinion No. 677. Antenatal corticosteroidtherapy for fetal maturation. Obstet Gynecol. 2016;128(4):e187-e194.
Kamath-Rayne BD, Rozance PJ, Goldenberg RL, Jobe AH. Antenatal corticosteroids beyond 34 weeks gestation: what do we do now? Am J Obstet Gynecol. 2016;215(4):423-430.
A dramatic recommendation for obstetric practice change occurred in 2016: the option of administering antenatal steroids for fetal lung maturity after 34 weeks. In the Antenatal Late Preterm Steroids (ALPS) trial of betamethasone in the late preterm period in patients at "high risk" of imminent delivery, Gyamfi-Bannerman and colleagues demonstrated that the treated group had a significant decrease in the rate of neonatal respiratory complications.
The primary outcome, a composite of respiratory morbidities (including transient tachypnea of the newborn, surfactant use, and need for resuscitation at birth) within the first 72 hours of life, had significant differences between groups, occurring in 165 of 1,427 infants (11.6%) in the betamethasone-treated group and 202 of 1,400 (14.4%) in the placebo group (relative risk in the betamethasone group, 0.80; 95% confidence interval, 0.66-0.97; P = .02). However, there was no statistically significant difference in respiratory distress syndrome, apnea, or pneumonia between groups, and the significant difference noted in bronchopulmonary dysplasia was based on a total number of 11 cases.
In response to these findings, both the American College of Obstetricians and Gynecologists (ACOG) and the Society for Maternal-Fetal Medicine (SMFM) released practice advisories and interim updates, culminating in a final recommendation for a single course of betamethasone in patients at high risk of preterm delivery between 34 and 36 6/7 weeks who have not received a previous course.
Related article:
When could use of antenatal corticosteroids in the late preterm birth period be beneficial?
In a thorough review of the literature on antenatal steroid use, Kamath-Rayne and colleagues highlighted several factors that should be considered before adopting universal use of steroids at >34 weeks. These include:
- The definition of "high risk of imminent delivery" as preterm labor with at least 3-cm dilation or 75% effacement, or spontaneous rupture of membranes. The effect of less stringent inclusion criteria in real-world clinical practice is not known, and many patients who will go on to deliver at term will receive steroids unnecessarily.
- Multiple gestation, patients with pre-existing diabetes, women who had previously received a course of steroids, and fetuses with anomalies were excluded from the ALPS study. Use of antenatal steroids in these groups at >34 weeks should be evaluated before universal adoption.
Related article:
What is the ideal gestational age for twin delivery to minimize perinatal deaths?
- The incidence of neonatal hypoglycemia in the treated group was significantly increased. This affects our colleagues in pediatrics considerably from a systems standpoint (need for changes to newborn protocols and communication between services).
- The long-term outcomes of patients exposed to steroids in the late preterm period are yet to be delineated, specifically, the potential neurodevelopmental effects of a medication known to alter preterm brain development as well as cardiovascular and metabolic consequences.
Next: Low-dose aspirin for reducing preeclampsia risk
Low-dose aspirin clearly is effective for reducing the risk of preeclampsia
American College of Obstetricians and Gynecologists. Hypertension in pregnancy. Report of the American College of Obstetricians and Gynecologists' Task Force on Hypertension in Pregnancy. Obstet Gynecol. 2013;122(5):1122-1131.
Henderson JT, Whitlock EP, O'Connor E, Senger CA, Thompson JH, Rowland MG. Low-dose aspirin for prevention of morbidity and mortality from preeclampsia: a systematic evidence review for the US Preventive Services Task Force. Ann Intern Med. 2014;160(10):695-703.
LeFevre ML; US Preventive Services Task Force. Low-dose aspirin use for the prevention of morbidity and mortality from preeclampsia: US Preventive Services Task Force recommendation statement. Ann Intern Med. 2014;161(11):819-826.
American College of Obstetricians and Gynecologists. Practice advisory on low-dose aspirin and prevention of preeclampsia: updated recommendations. http://www.acog.org/About-ACOG/News-Room/Practice-Advisories/Practice-Advisory-Low-Dose-Aspirin-and-Prevention-of-Preeclampsia-Updated-Recommendations. Published July 11, 2016. Accessed December 6, 2016.
In the 2013 ACOG Task Force on Hypertension in Pregnancy report, low-dose aspirin (60-80 mg) was recommended to be initiated in the late first trimester to reduce preeclampsia risk for women with:
- prior early onset preeclampsia with preterm delivery at <34 weeks' gestation, or
- preeclampsia in more than one prior pregnancy.
This recommendation was based on several meta-analyses that demonstrated a 10% to 17% reduction in risk with no increase in bleeding, placental abruption, or other adverse events.
In 2014, the US Preventive Services Task Force (USPSTF) conducted a systematic evidence review of low-dose aspirin use for prevention of morbidity and mortality from preeclampsia. That report revealed a 24% risk reduction of preeclampsia in high-risk women treated with low-dose aspirin, as well as a 14% reduction in preterm birth and a 20% reduction in fetal growth restriction. A final statement from the USPSTF in 2014 recommended low-dose aspirin (60-150 mg) starting between 12 and 28 weeks' gestation for women at "high" risk who have:
- a history of preeclampsia, especially if accompanied by an adverse outcome
- multifetal gestation
- chronic hypertension
- diabetes (type 1 or type 2)
- renal disease
- autoimmune disease (such as systematic lupus erythematosus, antiphospholipid syndrome).
Related article:
Start offering aspirin to pregnant women at high risk for preeclampsia
As of July 11, 2016, ACOG supports this expanded list of high-risk conditions. Additionally, the USPSTF identified a "moderate" risk group in which low-dose aspirin may be considered if a patient has several risk factors, such as obesity, nulliparity, family history of preeclampsia, age 35 years or older, or another poor pregnancy outcome. ACOG notes, however, that the evidence supporting this practice is uncertain and does not make a recommendation regarding aspirin use in this population. Further study should be conducted to determine the benefit of low-dose aspirin in these patients as well as the long-term effects of treatment on maternal and child outcomes.
Next: CMA for prenatal genetic diagnosis
Chromosomal microarray analysis is preferable to karyotype in certain situations
Pauli JM, Repke JT. Update on obstetrics. OBG Manag. 2013;25(1):28-32.
Society for Maternal-Fetal Medicine (SMFM), Dugoff L, Norton ME, Kuller JA. The use of chromosomal microarray for prenatal diagnosis. Am J Obstet Gynecol. 2016;215(4):B2-B9.
American College of Obstetricians and Gynecologists. Committee Opinion No. 682. Microarrays and next- generation sequencing technology: the use of advanced genetic diagnostic tools in obstetrics and gynecology.Obstet Gynecol. 2016;128(6):e262-e268.
We previously addressed the use of chromosomal microarray analysis (CMA) for prenatal diagnosis in our 2013 "Update on obstetrics," specifically, the question of whether CMA could replace karyotype. The main differences between karyotype and CMA are that 1) only karyotype can detect balanced translocations/inversions and 2) only CMA can detect copy number variants (CNV). There are some differences in the technology and capabilities of the 2 types of CMA currently available as well.
In our 2013 article we concluded that "The total costs of such an approach--test, interpretation, counseling, and long-term follow-up of uncertain results--are unknown at this time and may prove to be unaffordable on a population-wide basis." Today, the cost of CMA is still higher than karyotype, but it is expected to decrease and insurance coverage for this test is expected to increase.
Related article:
Cell-free DNA screening for women at low risk for fetal aneuploidy
Both SMFM and ACOG released recommendations in 2016 regarding the use of CMA in prenatal genetic diagnosis, summarized as follows:
- CMA is recommended over karyotype for fetuses with structural abnormalities on ultrasound
- The detection rate for clinically relevant abnormal CNVs in this population is about 6%
- CMA is recommended for diagnosis for stillbirth specimens
- CMA does not require dividing cells and may be a quicker and more reliable test in this population
- Karotype or fluorescence in situ hybridization (FISH) is recommended for fetuses with ultrasound findings suggestive of aneuploidy
- If it is negative, then CMA is recommended
- Karyotype or CMA is recommended for patients desiring prenatal diagnostic testing with a normal fetal ultrasound
- The detection rate for clinically relevant CNVs in this population (advanced maternal age, abnormal serum screening, prior aneuploidy, parental anxiety) is about 1%
- Pretest and posttest counseling about the limitations of CMA and a 2% risk of detection of variants of unknown significance (VUS) should be performed by a provider who has expertise in CMA and who has access to databases with genotype/phenotype information for VUS
- This counseling should also include the possibility of diagnosis of nonpaternity, consanguinity, and adult-onset disease
- Karyotype is recommended for couples with recurrent pregnancy loss
- The identification of balanced translocations in this population is most relevant in this patient population
- Prenatal diagnosis with routine use of whole-genome or whole-exome sequencing is not recommended.
Next: Zika virus: Check for updates
Zika virus infection: Check often for the latest updates
American College of Obstetricians and Gynecologists, Society for Maternal-Fetal Medicine. Practice advisory on Zika virus. http://www.acog.org/About-ACOG/News-Room/Practice-Advisories/Practice-Advisory-Interim-Guidance-for-Care-of-Obstetric-Patients- During-a-Zika-Virus-Outbreak. Published December 5, 2016. Accessed December 6, 2016.
Centers for Disease Control and Prevention. Zika virus. http://www.cdc.gov/zika/pregnancy/index.html. Updated August 22, 2016. Accessed December 6, 2016.
Petersen EE, Meaney-Delman D, Neblett-Fanfair R, et al. Update: interim guidance for preconception counseling and prevention of sexual transmission of Zika virus for persons with possible Zika virus exposure--United States, September 2016. MMWR Morbid Mortal Wkly Rep. 2016;65(39):1077-1081.
A yearly update on obstetrics would be remiss without mention of the Zika virus and its impact on pregnancy and reproduction. That being said, any recommendations we offer may be out of date by the time this article is published given the rapidly changing picture of Zika virus since it first dominated the headlines in 2016. Here are the basics as summarized from ACOG and the Centers for Disease Control and Prevention (CDC):
Viral spread. Zika virus may be spread in several ways: by an infected Aedes species mosquito, mother to fetus, sexual contact, blood transfusion, or laboratory exposure.
Symptoms of infection include conjunctivitis, fever, rash, and arthralgia, but most patients (4/5) are asymptomatic.
Sequelae. Zika virus infection during pregnancy is believed to cause fetal and neonatal microcephaly, intracranial calcifications, and brain and eye abnormalities. The rate of these findings in infected individuals, as well as the rate of vertical transmission, is not known.
Travel advisory. Pregnant women should not travel to areas with active Zika infection (the CDC website regularly updates these restricted areas).
Preventive measures. If traveling to an area of active Zika infection, pregnant women should take preventative measures day and night against mosquito bites, such as use of insect repellents approved by the Environmental Protection Agency, clothing that covers exposed skin, and staying indoors.
Safe sex. Abstinence or consistent condom use is recommended for pregnant women with partners who travel to or live in areas of active Zika infection.
Delay conception. Conception should be postponed for at least 6 months in men with Zika infection and at least 8 weeks in women with Zika infection.
Testing recommendations. Pregnant women with Zika virus exposure should be tested, regardless of symptoms. Symptomatic exposed nonpregnant women and all men should be tested.
Prenatal surveillance. High-risk consultation and serial ultrasounds for fetal anatomy and growth should be considered in patients with Zika virus infection during pregnancy. Amniocentesis can be considered on a case-by-case basis.
Related article:
Zika virus update: A rapidly moving target
Share your thoughts! Send your Letter to the Editor to [email protected]. Please include your name and the city and state in which you practice.
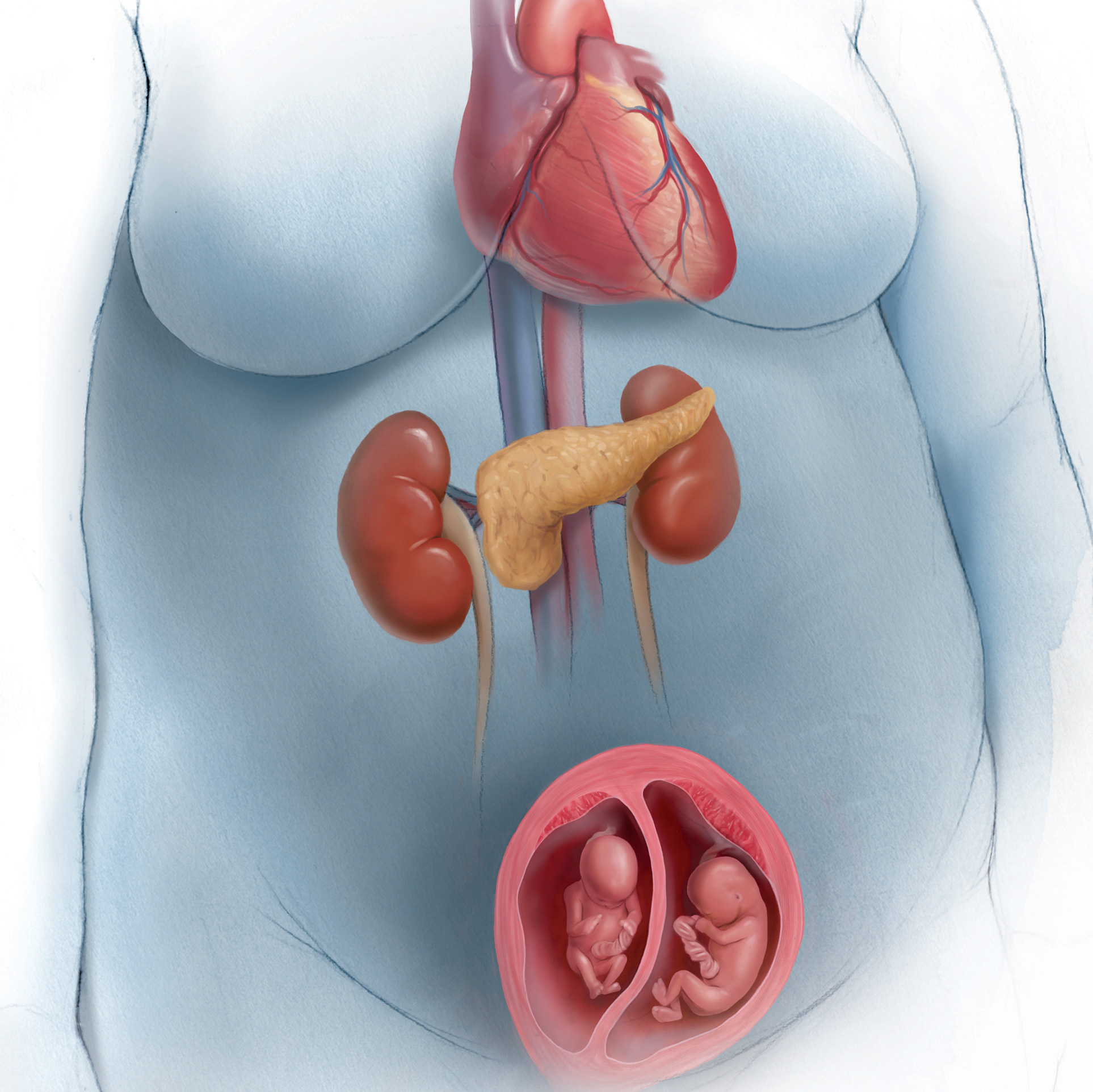
What is the ideal gestational age for twin delivery to minimize perinatal deaths?
EXPERT COMMENTARY
Cheong-See and colleagues conducted a comprehensive review and analysis of 32 studies of uncomplicated dichorionic and monochorionic twin pregnancies to determine the risks of stillbirth and neonatal complications by gestational age.
Details of the study
The authors searched major databases for studies on twin pregnancies that reported rates of stillbirth as well as neonatal outcomes (neonatal mortality was defined as death up to 28 days after delivery). A total of 32 studies were included in the analysis, with 29,685 dichorionic and 5,486 monochorionic pregnancies in 35,171 women. The authors estimated the gestational-age specific differences in risk for stillbirths and neonatal deaths after 34 weeks’ gestation.
In dichorionic pregnancies, the prospective weekly pooled risk of stillbirths from expectant management and the risk of neonatal mortality from delivery were balanced at 37 weeks of gestation (risk difference, 1.2/1,000, 95% CI, −1.3 to 3.6; I2 = 0%). In monochorionic pregnancies, after 36 weeks there was a trend toward an increase in stillbirths compared with neonatal deaths, with a pooled risk difference of 2.5/1,000 (95% CI, −12.4 to 17.4; I2 = 0%). Neonatal morbidity rates were consistently reduced with increasing gestational age in both monochorionic and dichorionic pregnancies.
The researchers’ recommendations
The authors recommended that dichorionic pregnancies be delivered at 37 weeks and that the evidence for delivery of monochorionic twins prior to 36 weeks is lacking. While the analysis is comprehensive and well done, it cannot escape the limitations that afflict all systematic reviews and meta-analyses, and these limitations are well addressed by the authors. Several factors, however, warrant caution regarding the adoption of the authors’ recommendations.
Cautions. First, determination of chorionicity may not have been accurate in all of the studies reviewed. Additionally, we have no data on how these pregnancies were managed with respect to antepartum fetal surveillance, ultrasound surveillance for growth and discordancy, and management of labor and delivery. There are no data on the quality of the ultrasound examinations being performed at each of the centers.
Also, the factors that may increase the risk of stillbirth are not necessarily the same factors that may influence the neonatal death rate, and this review moves between the use of these terms (stillbirth rate, neonatal mortality rate, and perinatal mortality rate) fairly frequently. For example, an improperly managed labor, an unanticipated difficult breech extraction, or the need for emergent cesarean delivery of the second twin might contribute to the neonatal death rate irrespective of gestational age at delivery. The authors acknowledge that outcomes may have been influenced by differences in obstetric and neonatal management of twin pregnancies that were observed between centers. Another concern is the authors’ use of unpublished aggregate and individual patient data.
While this comprehensive and very well conducted review and meta-analysis highlights the complexity of balancing stillbirth risk against neonatal mortality risk, the limitations of the study are too substantial to allow for any change in current practice. My recommendation for the timing of twin delivery is to adhere to the guidelines that are currently supported by both the Society for Maternal-Fetal Medicine and the American College of Obstetricians and Gynecologists.1 These guidelines recommend that, for dichorionic-diamniotic twin pregnancy, the general timing of delivery be at early term, with suggested specific timing at 38 0/7 to 38 6/7 weeks of gestation. For monochorionic-diamniotic twin pregnancy, the general timing of delivery may be at late preterm/early term, with suggested specific timing at 34 0/7 to 37 6/7 weeks of gestation. Delivery decisions made within these date ranges depend on numerous factors discussed in the guidelines, and timing of delivery should be individualized.1
-- JOHN T. REPKE, MD
Share your thoughts! Send your Letter to the Editor to [email protected]. Please include your name and the city and state in which you practice.
- American College of Obstetricians and Gynecologists Committee on Obstetric Practice, Society for Maternal-Fetal Medicine. ACOG Committee Opinion No. 560. Medically indicated late-preterm and early-term deliveries. Obstet Gynecol. 2013;121(4):908–910. Reaffirmed 2015.
EXPERT COMMENTARY
Cheong-See and colleagues conducted a comprehensive review and analysis of 32 studies of uncomplicated dichorionic and monochorionic twin pregnancies to determine the risks of stillbirth and neonatal complications by gestational age.
Details of the study
The authors searched major databases for studies on twin pregnancies that reported rates of stillbirth as well as neonatal outcomes (neonatal mortality was defined as death up to 28 days after delivery). A total of 32 studies were included in the analysis, with 29,685 dichorionic and 5,486 monochorionic pregnancies in 35,171 women. The authors estimated the gestational-age specific differences in risk for stillbirths and neonatal deaths after 34 weeks’ gestation.
In dichorionic pregnancies, the prospective weekly pooled risk of stillbirths from expectant management and the risk of neonatal mortality from delivery were balanced at 37 weeks of gestation (risk difference, 1.2/1,000, 95% CI, −1.3 to 3.6; I2 = 0%). In monochorionic pregnancies, after 36 weeks there was a trend toward an increase in stillbirths compared with neonatal deaths, with a pooled risk difference of 2.5/1,000 (95% CI, −12.4 to 17.4; I2 = 0%). Neonatal morbidity rates were consistently reduced with increasing gestational age in both monochorionic and dichorionic pregnancies.
The researchers’ recommendations
The authors recommended that dichorionic pregnancies be delivered at 37 weeks and that the evidence for delivery of monochorionic twins prior to 36 weeks is lacking. While the analysis is comprehensive and well done, it cannot escape the limitations that afflict all systematic reviews and meta-analyses, and these limitations are well addressed by the authors. Several factors, however, warrant caution regarding the adoption of the authors’ recommendations.
Cautions. First, determination of chorionicity may not have been accurate in all of the studies reviewed. Additionally, we have no data on how these pregnancies were managed with respect to antepartum fetal surveillance, ultrasound surveillance for growth and discordancy, and management of labor and delivery. There are no data on the quality of the ultrasound examinations being performed at each of the centers.
Also, the factors that may increase the risk of stillbirth are not necessarily the same factors that may influence the neonatal death rate, and this review moves between the use of these terms (stillbirth rate, neonatal mortality rate, and perinatal mortality rate) fairly frequently. For example, an improperly managed labor, an unanticipated difficult breech extraction, or the need for emergent cesarean delivery of the second twin might contribute to the neonatal death rate irrespective of gestational age at delivery. The authors acknowledge that outcomes may have been influenced by differences in obstetric and neonatal management of twin pregnancies that were observed between centers. Another concern is the authors’ use of unpublished aggregate and individual patient data.
While this comprehensive and very well conducted review and meta-analysis highlights the complexity of balancing stillbirth risk against neonatal mortality risk, the limitations of the study are too substantial to allow for any change in current practice. My recommendation for the timing of twin delivery is to adhere to the guidelines that are currently supported by both the Society for Maternal-Fetal Medicine and the American College of Obstetricians and Gynecologists.1 These guidelines recommend that, for dichorionic-diamniotic twin pregnancy, the general timing of delivery be at early term, with suggested specific timing at 38 0/7 to 38 6/7 weeks of gestation. For monochorionic-diamniotic twin pregnancy, the general timing of delivery may be at late preterm/early term, with suggested specific timing at 34 0/7 to 37 6/7 weeks of gestation. Delivery decisions made within these date ranges depend on numerous factors discussed in the guidelines, and timing of delivery should be individualized.1
-- JOHN T. REPKE, MD
Share your thoughts! Send your Letter to the Editor to [email protected]. Please include your name and the city and state in which you practice.
EXPERT COMMENTARY
Cheong-See and colleagues conducted a comprehensive review and analysis of 32 studies of uncomplicated dichorionic and monochorionic twin pregnancies to determine the risks of stillbirth and neonatal complications by gestational age.
Details of the study
The authors searched major databases for studies on twin pregnancies that reported rates of stillbirth as well as neonatal outcomes (neonatal mortality was defined as death up to 28 days after delivery). A total of 32 studies were included in the analysis, with 29,685 dichorionic and 5,486 monochorionic pregnancies in 35,171 women. The authors estimated the gestational-age specific differences in risk for stillbirths and neonatal deaths after 34 weeks’ gestation.
In dichorionic pregnancies, the prospective weekly pooled risk of stillbirths from expectant management and the risk of neonatal mortality from delivery were balanced at 37 weeks of gestation (risk difference, 1.2/1,000, 95% CI, −1.3 to 3.6; I2 = 0%). In monochorionic pregnancies, after 36 weeks there was a trend toward an increase in stillbirths compared with neonatal deaths, with a pooled risk difference of 2.5/1,000 (95% CI, −12.4 to 17.4; I2 = 0%). Neonatal morbidity rates were consistently reduced with increasing gestational age in both monochorionic and dichorionic pregnancies.
The researchers’ recommendations
The authors recommended that dichorionic pregnancies be delivered at 37 weeks and that the evidence for delivery of monochorionic twins prior to 36 weeks is lacking. While the analysis is comprehensive and well done, it cannot escape the limitations that afflict all systematic reviews and meta-analyses, and these limitations are well addressed by the authors. Several factors, however, warrant caution regarding the adoption of the authors’ recommendations.
Cautions. First, determination of chorionicity may not have been accurate in all of the studies reviewed. Additionally, we have no data on how these pregnancies were managed with respect to antepartum fetal surveillance, ultrasound surveillance for growth and discordancy, and management of labor and delivery. There are no data on the quality of the ultrasound examinations being performed at each of the centers.
Also, the factors that may increase the risk of stillbirth are not necessarily the same factors that may influence the neonatal death rate, and this review moves between the use of these terms (stillbirth rate, neonatal mortality rate, and perinatal mortality rate) fairly frequently. For example, an improperly managed labor, an unanticipated difficult breech extraction, or the need for emergent cesarean delivery of the second twin might contribute to the neonatal death rate irrespective of gestational age at delivery. The authors acknowledge that outcomes may have been influenced by differences in obstetric and neonatal management of twin pregnancies that were observed between centers. Another concern is the authors’ use of unpublished aggregate and individual patient data.
While this comprehensive and very well conducted review and meta-analysis highlights the complexity of balancing stillbirth risk against neonatal mortality risk, the limitations of the study are too substantial to allow for any change in current practice. My recommendation for the timing of twin delivery is to adhere to the guidelines that are currently supported by both the Society for Maternal-Fetal Medicine and the American College of Obstetricians and Gynecologists.1 These guidelines recommend that, for dichorionic-diamniotic twin pregnancy, the general timing of delivery be at early term, with suggested specific timing at 38 0/7 to 38 6/7 weeks of gestation. For monochorionic-diamniotic twin pregnancy, the general timing of delivery may be at late preterm/early term, with suggested specific timing at 34 0/7 to 37 6/7 weeks of gestation. Delivery decisions made within these date ranges depend on numerous factors discussed in the guidelines, and timing of delivery should be individualized.1
-- JOHN T. REPKE, MD
Share your thoughts! Send your Letter to the Editor to [email protected]. Please include your name and the city and state in which you practice.
- American College of Obstetricians and Gynecologists Committee on Obstetric Practice, Society for Maternal-Fetal Medicine. ACOG Committee Opinion No. 560. Medically indicated late-preterm and early-term deliveries. Obstet Gynecol. 2013;121(4):908–910. Reaffirmed 2015.
- American College of Obstetricians and Gynecologists Committee on Obstetric Practice, Society for Maternal-Fetal Medicine. ACOG Committee Opinion No. 560. Medically indicated late-preterm and early-term deliveries. Obstet Gynecol. 2013;121(4):908–910. Reaffirmed 2015.
Does influenza immunization during pregnancy confer flu protection to newborns?
A recent report on data obtained from the Intermountain Healthcare facilities in Utah and Idaho suggests that maternal influenza vaccination, besides its maternal protective effect, also may benefit the newborn by protecting against influenza. I think that ObGyns generally accept the notion that influenza vaccination has defined maternal benefit. Notably, acceptance of influenza vaccine became much more widespread during and following the H1N1 flu pandemic of 2009. The fact that there may be neonatal and infant benefit as well is certainly not objectionable, especially since flu vaccine is not recommended for infants less than 6 months of age.
Details of the study
Shakib and colleagues’ goal was to compare influenza outcomes in infants younger than 6 months born to women who self-reported receiving or not receiving influenza vaccine during pregnancy. The study cohort included 245,386 women and 249,387 infants who were born between December 2005 and March 2014. The outcomes studied were influenza-like illness (ILI), laboratory-confirmed influenza, and influenza hospitalizations.
Of 866 infants younger than 6 months born to women who had received a flu shot, 32 had 1 or more ILI encounters, compared with 834 born to women who were not immunized (relative risk [RR], 0.36; 95% confidence interval [CI], 0.26–0.52; P<.001). Laboratory-confirmed influenza was identified in 658 infants; of these, 20 were born to women reporting immunization and 638 were born to unimmunized women (RR, 0.30; 95% CI, 0.19–0.46; P<.001). Finally, 151 infants with laboratory-confirmed influenza were hospitalized; 3 were born to immunized women and 148 to women who had not received a flu shot (RR, 0.19; 95% CI, 0.06–0.60; P = .005).
The authors calculated that infants born to women who reported influenza immunization during pregnancy had risk reductions of 64% for ILI, 70% for laboratory-confirmed influenza, and 81% for influenza hospitalizations.
Consider immunization benefits realistically
The authors state in their published report that “Protecting young infants from influenza through maternal immunization during pregnancy is a public health priority.” This may be overstating their case, for the following reasons. First, influenza vaccine has varying degrees of efficacy depending on the particular year and influenza strains that predominate, and it never has been shown to be entirely protective. Second, when looking only at this study’s laboratory-proven cases of influenza in newborns, infants whose mothers were vaccinated had a case rate of 0.84/1,000, while the case rate in newborns born to unvaccinated women was 2.83/1,000. While this shows a difference, the report provides no additional outcome data regarding morbidity or mortality.
In fact, although infant hospitalization rates differed (0.13/1,000 born to vaccinated women versus 0.66/1,000 born to unvaccinated women), there were no influenza-related mortalities in this cohort. The effect seems better when including “influenza-like illness,” but it is unclear why we should think that the influenza vaccine protects against infection that is not caused by influenza.
What this EVIDENCE means for practice
ObGyns should continue to promote influenza vaccination during pregnancy for the maternal protection it imparts. These new data on the potential for newborn protection, although not conclusive, may improve maternal acceptance of vaccination; from that perspective, these data are valuable.
— John T. Repke, MD
Share your thoughts on this article! Send your Letter to the Editor to [email protected]. Please include your name and the city and state in which you practice.
A recent report on data obtained from the Intermountain Healthcare facilities in Utah and Idaho suggests that maternal influenza vaccination, besides its maternal protective effect, also may benefit the newborn by protecting against influenza. I think that ObGyns generally accept the notion that influenza vaccination has defined maternal benefit. Notably, acceptance of influenza vaccine became much more widespread during and following the H1N1 flu pandemic of 2009. The fact that there may be neonatal and infant benefit as well is certainly not objectionable, especially since flu vaccine is not recommended for infants less than 6 months of age.
Details of the study
Shakib and colleagues’ goal was to compare influenza outcomes in infants younger than 6 months born to women who self-reported receiving or not receiving influenza vaccine during pregnancy. The study cohort included 245,386 women and 249,387 infants who were born between December 2005 and March 2014. The outcomes studied were influenza-like illness (ILI), laboratory-confirmed influenza, and influenza hospitalizations.
Of 866 infants younger than 6 months born to women who had received a flu shot, 32 had 1 or more ILI encounters, compared with 834 born to women who were not immunized (relative risk [RR], 0.36; 95% confidence interval [CI], 0.26–0.52; P<.001). Laboratory-confirmed influenza was identified in 658 infants; of these, 20 were born to women reporting immunization and 638 were born to unimmunized women (RR, 0.30; 95% CI, 0.19–0.46; P<.001). Finally, 151 infants with laboratory-confirmed influenza were hospitalized; 3 were born to immunized women and 148 to women who had not received a flu shot (RR, 0.19; 95% CI, 0.06–0.60; P = .005).
The authors calculated that infants born to women who reported influenza immunization during pregnancy had risk reductions of 64% for ILI, 70% for laboratory-confirmed influenza, and 81% for influenza hospitalizations.
Consider immunization benefits realistically
The authors state in their published report that “Protecting young infants from influenza through maternal immunization during pregnancy is a public health priority.” This may be overstating their case, for the following reasons. First, influenza vaccine has varying degrees of efficacy depending on the particular year and influenza strains that predominate, and it never has been shown to be entirely protective. Second, when looking only at this study’s laboratory-proven cases of influenza in newborns, infants whose mothers were vaccinated had a case rate of 0.84/1,000, while the case rate in newborns born to unvaccinated women was 2.83/1,000. While this shows a difference, the report provides no additional outcome data regarding morbidity or mortality.
In fact, although infant hospitalization rates differed (0.13/1,000 born to vaccinated women versus 0.66/1,000 born to unvaccinated women), there were no influenza-related mortalities in this cohort. The effect seems better when including “influenza-like illness,” but it is unclear why we should think that the influenza vaccine protects against infection that is not caused by influenza.
What this EVIDENCE means for practice
ObGyns should continue to promote influenza vaccination during pregnancy for the maternal protection it imparts. These new data on the potential for newborn protection, although not conclusive, may improve maternal acceptance of vaccination; from that perspective, these data are valuable.
— John T. Repke, MD
Share your thoughts on this article! Send your Letter to the Editor to [email protected]. Please include your name and the city and state in which you practice.
A recent report on data obtained from the Intermountain Healthcare facilities in Utah and Idaho suggests that maternal influenza vaccination, besides its maternal protective effect, also may benefit the newborn by protecting against influenza. I think that ObGyns generally accept the notion that influenza vaccination has defined maternal benefit. Notably, acceptance of influenza vaccine became much more widespread during and following the H1N1 flu pandemic of 2009. The fact that there may be neonatal and infant benefit as well is certainly not objectionable, especially since flu vaccine is not recommended for infants less than 6 months of age.
Details of the study
Shakib and colleagues’ goal was to compare influenza outcomes in infants younger than 6 months born to women who self-reported receiving or not receiving influenza vaccine during pregnancy. The study cohort included 245,386 women and 249,387 infants who were born between December 2005 and March 2014. The outcomes studied were influenza-like illness (ILI), laboratory-confirmed influenza, and influenza hospitalizations.
Of 866 infants younger than 6 months born to women who had received a flu shot, 32 had 1 or more ILI encounters, compared with 834 born to women who were not immunized (relative risk [RR], 0.36; 95% confidence interval [CI], 0.26–0.52; P<.001). Laboratory-confirmed influenza was identified in 658 infants; of these, 20 were born to women reporting immunization and 638 were born to unimmunized women (RR, 0.30; 95% CI, 0.19–0.46; P<.001). Finally, 151 infants with laboratory-confirmed influenza were hospitalized; 3 were born to immunized women and 148 to women who had not received a flu shot (RR, 0.19; 95% CI, 0.06–0.60; P = .005).
The authors calculated that infants born to women who reported influenza immunization during pregnancy had risk reductions of 64% for ILI, 70% for laboratory-confirmed influenza, and 81% for influenza hospitalizations.
Consider immunization benefits realistically
The authors state in their published report that “Protecting young infants from influenza through maternal immunization during pregnancy is a public health priority.” This may be overstating their case, for the following reasons. First, influenza vaccine has varying degrees of efficacy depending on the particular year and influenza strains that predominate, and it never has been shown to be entirely protective. Second, when looking only at this study’s laboratory-proven cases of influenza in newborns, infants whose mothers were vaccinated had a case rate of 0.84/1,000, while the case rate in newborns born to unvaccinated women was 2.83/1,000. While this shows a difference, the report provides no additional outcome data regarding morbidity or mortality.
In fact, although infant hospitalization rates differed (0.13/1,000 born to vaccinated women versus 0.66/1,000 born to unvaccinated women), there were no influenza-related mortalities in this cohort. The effect seems better when including “influenza-like illness,” but it is unclear why we should think that the influenza vaccine protects against infection that is not caused by influenza.
What this EVIDENCE means for practice
ObGyns should continue to promote influenza vaccination during pregnancy for the maternal protection it imparts. These new data on the potential for newborn protection, although not conclusive, may improve maternal acceptance of vaccination; from that perspective, these data are valuable.
— John T. Repke, MD
Share your thoughts on this article! Send your Letter to the Editor to [email protected]. Please include your name and the city and state in which you practice.
Shoulder dystocia: Taking the fear out of management
2016 Update on obstetrics
Some areas of obstetric care are not as clearcut as others in this time of rapid medical evolution. In this Update, we discuss 3 of them:
- management of twin gestations
- management of chronic hypertension in pregnancy
- cell-free DNA screening for fetal aneuploidy.
To our benefit, both the American College of Obstetricians and Gynecologists (ACOG) and the Society for Maternal-Fetal Medicine (SMFM) have weighed in on important aspects of these areas of obstetric care.
Society for Maternal-Fetal Medicine, Simpson LL. Twin-twin transfusion syndrome. Am J Obstet Gynecol. 2013;208(1):3–18.
Society for Maternal-Fetal Medicine. Checklists and Safety Bundles. https://www.smfm.org/mfm-practice/checklists-and-safety-bundles. Published March 2015. Accessed December 7, 2015.
American College of Obstetricians and Gynecologists. Practice bulletin No. 144. Multifetal gestations: twin, triplet, and higher-order multifetal pregnancies. Obstet Gynecol. 2014;123(5):1118–1132.
From the maternal perspective, twin pregnancies are known to have higher risks than their singleton counterparts for such complications as hypertension, preeclampsia, diabetes, hemorrhage, cesarean delivery, postpartum depression, and anemia. These complications are managed essentially the same way regardless of the number of fetuses.
From the fetal/neonatal perspective, twin gestations may carry increased risks of congenital anomalies, preterm birth, and aneuploidy, which are managed similarly to singleton gestations overall, with certain adjustments as necessary.
Twin pregnancies do have unique risks, however, that are managed differently from the time chorionicity is established until delivery. The level of risk increases as the number of chorions and amnions decreases.
A basic management plan for twin gestations consists of a number of components, elucidated below.
1. Determine chorionicity and amnionicityThis determination is most reliably performed late in the first trimester and must be done using ultrasound. The inter-twin membrane should be identified. At 11 to 14 weeks, the presence of the “lambda sign,” a triangular projection of tissue that extends from the chorionic surface, is indicative of a dichorionic pregnancy, while a “T sign” suggests a monochorionic pregnancy (sensitivities 97%–100%; specificities 98%–100%). Alternatively, demonstration of discordant genders or separate placentas may be used later in pregnancy.
2. Monitor growth every 4 weeks in dichorionic twinsDichorionic twins are, by default, diamniotic. After the anatomic survey, growth surveillance should be conducted approximately every 4 weeks.
Discordant growth usually is defined as a difference of 20% or more in weight between the twins, based on the weight of the larger twin. As an isolated finding with both fetuses of normal weight, this discordance has not been demonstrated to increase adverse outcomes. Routine antenatal surveillance is not necessarily indicated.
Fetal growth restriction of one twin or a coexisting abnormality should prompt antenatal testing and/or earlier delivery. Any maternal comorbidities such as hypertension or diabetes also would be indications for testing. Otherwise, delivery is recommended at 38 weeks’ to 38-6/7 weeks’ gestation.
After 32 weeks, the mode of delivery may be vaginal if the presenting twin is vertex and the delivery provider can perform breech extraction or internal podalic version, if necessary.
3. Monochorionic/diamniotic twins also warrant regular surveillanceThe shared placenta places these pregnancies at increased risk for twin-to-twin transfusion syndrome (TTTS), a fetal-placental imbalance in which one twin “transfuses” the other. Ten percent to 15% of monochorionic pregnancies develop TTTS, which is associated with high rates of morbidity and mortality, even when treated.
Antenatal surveillance of these pregnancies involves ultrasonography assessment every 2 weeks, starting at 16 weeks. At each examination, the deepest vertical pocket (DVP) of fluid and presence of each fetal bladder are documented. This limited assessment alternates with a growth assessment every 2 weeks. SMFM recommends this biweekly assessment until 28 weeks, then every 2 to 3 weeks until delivery.
Stage 1 TTTS is defined by the polyhydramnios/oligohydramnios sequence (DVP of one fetus <2 cm, with DVP of the other >8 cm).
Evaluation for treatment of TTTS with laser coagulation (preferred) or amnio‑reduction should take place after the diagnosis is made, along with increased fetal surveillance.
SMFM also recommends fetal echocardiography due to the 9-fold increased risk of cardiac anomalies in monochorionic pregnancies.
Other complications of monochorionic/diamniotic twins include selective fetal growth restriction (due to unequal sharing of the placenta), twin reversed arterial perfusion (TRAP) sequence, and twin anemia-polycythemia sequence (TAPS).
Antenatal surveillance of all monochorionic twins is recommended, given the increased risk of stillbirth; many centers start testing at 32 weeks’ gestation. According to ACOG, uncomplicated monochorionic/diamniotic twins should be delivered at 34 weeks’ to 37-6/7 weeks’ gestation. Fetal growth restriction or other comorbidities may prompt delivery as early as 32 weeks.
4. Know the risks of monoamniotic twin gestationsThese twins are at increased risk for intrauterine fetal death due to cord entanglement, as well as TTTS, TAPS, and fetal growth restriction. Routine growth assessment and evaluation for TTTS are similar to those for monochorionic/diamniotic twins (without the option of polyhydramnios/oligohydramnios measurement), but the overall management of these pregnancies is unknown.
Protocols may range from outpatient antenatal testing to hospitalization to 24 to 28 weeks’ gestation with daily antenatal testing or attempted continuous monitoring. Delivery by cesarean delivery is recommended at 32 to 34 weeks’ gestation.
What this EVIDENCE means for practice Monochorionic twins need specific and frequent monitoring due to significantly increased risk for both fetal and placental complications. They justify late preterm or early term delivery.
Management of chronic hypertension in pregnancy: Reserve therapy for severe hypertension
Cell-free DNA screening for fetal aneuploidy: Strengths and limitations
Some areas of obstetric care are not as clearcut as others in this time of rapid medical evolution. In this Update, we discuss 3 of them:
- management of twin gestations
- management of chronic hypertension in pregnancy
- cell-free DNA screening for fetal aneuploidy.
To our benefit, both the American College of Obstetricians and Gynecologists (ACOG) and the Society for Maternal-Fetal Medicine (SMFM) have weighed in on important aspects of these areas of obstetric care.
Society for Maternal-Fetal Medicine, Simpson LL. Twin-twin transfusion syndrome. Am J Obstet Gynecol. 2013;208(1):3–18.
Society for Maternal-Fetal Medicine. Checklists and Safety Bundles. https://www.smfm.org/mfm-practice/checklists-and-safety-bundles. Published March 2015. Accessed December 7, 2015.
American College of Obstetricians and Gynecologists. Practice bulletin No. 144. Multifetal gestations: twin, triplet, and higher-order multifetal pregnancies. Obstet Gynecol. 2014;123(5):1118–1132.
From the maternal perspective, twin pregnancies are known to have higher risks than their singleton counterparts for such complications as hypertension, preeclampsia, diabetes, hemorrhage, cesarean delivery, postpartum depression, and anemia. These complications are managed essentially the same way regardless of the number of fetuses.
From the fetal/neonatal perspective, twin gestations may carry increased risks of congenital anomalies, preterm birth, and aneuploidy, which are managed similarly to singleton gestations overall, with certain adjustments as necessary.
Twin pregnancies do have unique risks, however, that are managed differently from the time chorionicity is established until delivery. The level of risk increases as the number of chorions and amnions decreases.
A basic management plan for twin gestations consists of a number of components, elucidated below.
1. Determine chorionicity and amnionicityThis determination is most reliably performed late in the first trimester and must be done using ultrasound. The inter-twin membrane should be identified. At 11 to 14 weeks, the presence of the “lambda sign,” a triangular projection of tissue that extends from the chorionic surface, is indicative of a dichorionic pregnancy, while a “T sign” suggests a monochorionic pregnancy (sensitivities 97%–100%; specificities 98%–100%). Alternatively, demonstration of discordant genders or separate placentas may be used later in pregnancy.
2. Monitor growth every 4 weeks in dichorionic twinsDichorionic twins are, by default, diamniotic. After the anatomic survey, growth surveillance should be conducted approximately every 4 weeks.
Discordant growth usually is defined as a difference of 20% or more in weight between the twins, based on the weight of the larger twin. As an isolated finding with both fetuses of normal weight, this discordance has not been demonstrated to increase adverse outcomes. Routine antenatal surveillance is not necessarily indicated.
Fetal growth restriction of one twin or a coexisting abnormality should prompt antenatal testing and/or earlier delivery. Any maternal comorbidities such as hypertension or diabetes also would be indications for testing. Otherwise, delivery is recommended at 38 weeks’ to 38-6/7 weeks’ gestation.
After 32 weeks, the mode of delivery may be vaginal if the presenting twin is vertex and the delivery provider can perform breech extraction or internal podalic version, if necessary.
3. Monochorionic/diamniotic twins also warrant regular surveillanceThe shared placenta places these pregnancies at increased risk for twin-to-twin transfusion syndrome (TTTS), a fetal-placental imbalance in which one twin “transfuses” the other. Ten percent to 15% of monochorionic pregnancies develop TTTS, which is associated with high rates of morbidity and mortality, even when treated.
Antenatal surveillance of these pregnancies involves ultrasonography assessment every 2 weeks, starting at 16 weeks. At each examination, the deepest vertical pocket (DVP) of fluid and presence of each fetal bladder are documented. This limited assessment alternates with a growth assessment every 2 weeks. SMFM recommends this biweekly assessment until 28 weeks, then every 2 to 3 weeks until delivery.
Stage 1 TTTS is defined by the polyhydramnios/oligohydramnios sequence (DVP of one fetus <2 cm, with DVP of the other >8 cm).
Evaluation for treatment of TTTS with laser coagulation (preferred) or amnio‑reduction should take place after the diagnosis is made, along with increased fetal surveillance.
SMFM also recommends fetal echocardiography due to the 9-fold increased risk of cardiac anomalies in monochorionic pregnancies.
Other complications of monochorionic/diamniotic twins include selective fetal growth restriction (due to unequal sharing of the placenta), twin reversed arterial perfusion (TRAP) sequence, and twin anemia-polycythemia sequence (TAPS).
Antenatal surveillance of all monochorionic twins is recommended, given the increased risk of stillbirth; many centers start testing at 32 weeks’ gestation. According to ACOG, uncomplicated monochorionic/diamniotic twins should be delivered at 34 weeks’ to 37-6/7 weeks’ gestation. Fetal growth restriction or other comorbidities may prompt delivery as early as 32 weeks.
4. Know the risks of monoamniotic twin gestationsThese twins are at increased risk for intrauterine fetal death due to cord entanglement, as well as TTTS, TAPS, and fetal growth restriction. Routine growth assessment and evaluation for TTTS are similar to those for monochorionic/diamniotic twins (without the option of polyhydramnios/oligohydramnios measurement), but the overall management of these pregnancies is unknown.
Protocols may range from outpatient antenatal testing to hospitalization to 24 to 28 weeks’ gestation with daily antenatal testing or attempted continuous monitoring. Delivery by cesarean delivery is recommended at 32 to 34 weeks’ gestation.
What this EVIDENCE means for practice Monochorionic twins need specific and frequent monitoring due to significantly increased risk for both fetal and placental complications. They justify late preterm or early term delivery.
Management of chronic hypertension in pregnancy: Reserve therapy for severe hypertension
Cell-free DNA screening for fetal aneuploidy: Strengths and limitations
Some areas of obstetric care are not as clearcut as others in this time of rapid medical evolution. In this Update, we discuss 3 of them:
- management of twin gestations
- management of chronic hypertension in pregnancy
- cell-free DNA screening for fetal aneuploidy.
To our benefit, both the American College of Obstetricians and Gynecologists (ACOG) and the Society for Maternal-Fetal Medicine (SMFM) have weighed in on important aspects of these areas of obstetric care.
Society for Maternal-Fetal Medicine, Simpson LL. Twin-twin transfusion syndrome. Am J Obstet Gynecol. 2013;208(1):3–18.
Society for Maternal-Fetal Medicine. Checklists and Safety Bundles. https://www.smfm.org/mfm-practice/checklists-and-safety-bundles. Published March 2015. Accessed December 7, 2015.
American College of Obstetricians and Gynecologists. Practice bulletin No. 144. Multifetal gestations: twin, triplet, and higher-order multifetal pregnancies. Obstet Gynecol. 2014;123(5):1118–1132.
From the maternal perspective, twin pregnancies are known to have higher risks than their singleton counterparts for such complications as hypertension, preeclampsia, diabetes, hemorrhage, cesarean delivery, postpartum depression, and anemia. These complications are managed essentially the same way regardless of the number of fetuses.
From the fetal/neonatal perspective, twin gestations may carry increased risks of congenital anomalies, preterm birth, and aneuploidy, which are managed similarly to singleton gestations overall, with certain adjustments as necessary.
Twin pregnancies do have unique risks, however, that are managed differently from the time chorionicity is established until delivery. The level of risk increases as the number of chorions and amnions decreases.
A basic management plan for twin gestations consists of a number of components, elucidated below.
1. Determine chorionicity and amnionicityThis determination is most reliably performed late in the first trimester and must be done using ultrasound. The inter-twin membrane should be identified. At 11 to 14 weeks, the presence of the “lambda sign,” a triangular projection of tissue that extends from the chorionic surface, is indicative of a dichorionic pregnancy, while a “T sign” suggests a monochorionic pregnancy (sensitivities 97%–100%; specificities 98%–100%). Alternatively, demonstration of discordant genders or separate placentas may be used later in pregnancy.
2. Monitor growth every 4 weeks in dichorionic twinsDichorionic twins are, by default, diamniotic. After the anatomic survey, growth surveillance should be conducted approximately every 4 weeks.
Discordant growth usually is defined as a difference of 20% or more in weight between the twins, based on the weight of the larger twin. As an isolated finding with both fetuses of normal weight, this discordance has not been demonstrated to increase adverse outcomes. Routine antenatal surveillance is not necessarily indicated.
Fetal growth restriction of one twin or a coexisting abnormality should prompt antenatal testing and/or earlier delivery. Any maternal comorbidities such as hypertension or diabetes also would be indications for testing. Otherwise, delivery is recommended at 38 weeks’ to 38-6/7 weeks’ gestation.
After 32 weeks, the mode of delivery may be vaginal if the presenting twin is vertex and the delivery provider can perform breech extraction or internal podalic version, if necessary.
3. Monochorionic/diamniotic twins also warrant regular surveillanceThe shared placenta places these pregnancies at increased risk for twin-to-twin transfusion syndrome (TTTS), a fetal-placental imbalance in which one twin “transfuses” the other. Ten percent to 15% of monochorionic pregnancies develop TTTS, which is associated with high rates of morbidity and mortality, even when treated.
Antenatal surveillance of these pregnancies involves ultrasonography assessment every 2 weeks, starting at 16 weeks. At each examination, the deepest vertical pocket (DVP) of fluid and presence of each fetal bladder are documented. This limited assessment alternates with a growth assessment every 2 weeks. SMFM recommends this biweekly assessment until 28 weeks, then every 2 to 3 weeks until delivery.
Stage 1 TTTS is defined by the polyhydramnios/oligohydramnios sequence (DVP of one fetus <2 cm, with DVP of the other >8 cm).
Evaluation for treatment of TTTS with laser coagulation (preferred) or amnio‑reduction should take place after the diagnosis is made, along with increased fetal surveillance.
SMFM also recommends fetal echocardiography due to the 9-fold increased risk of cardiac anomalies in monochorionic pregnancies.
Other complications of monochorionic/diamniotic twins include selective fetal growth restriction (due to unequal sharing of the placenta), twin reversed arterial perfusion (TRAP) sequence, and twin anemia-polycythemia sequence (TAPS).
Antenatal surveillance of all monochorionic twins is recommended, given the increased risk of stillbirth; many centers start testing at 32 weeks’ gestation. According to ACOG, uncomplicated monochorionic/diamniotic twins should be delivered at 34 weeks’ to 37-6/7 weeks’ gestation. Fetal growth restriction or other comorbidities may prompt delivery as early as 32 weeks.
4. Know the risks of monoamniotic twin gestationsThese twins are at increased risk for intrauterine fetal death due to cord entanglement, as well as TTTS, TAPS, and fetal growth restriction. Routine growth assessment and evaluation for TTTS are similar to those for monochorionic/diamniotic twins (without the option of polyhydramnios/oligohydramnios measurement), but the overall management of these pregnancies is unknown.
Protocols may range from outpatient antenatal testing to hospitalization to 24 to 28 weeks’ gestation with daily antenatal testing or attempted continuous monitoring. Delivery by cesarean delivery is recommended at 32 to 34 weeks’ gestation.
What this EVIDENCE means for practice Monochorionic twins need specific and frequent monitoring due to significantly increased risk for both fetal and placental complications. They justify late preterm or early term delivery.
Management of chronic hypertension in pregnancy: Reserve therapy for severe hypertension
Cell-free DNA screening for fetal aneuploidy: Strengths and limitations
In this article
- Know the risks of monoamniotic twin gestations
- “Less tight” versus “tight” BP control
- Cell-free DNA screening for aneuploidy
When is cell-free DNA best used as a primary screen?
Cell-free DNA screening, or so-called noninvasive prenatal testing (NIPT), has had greatly increased utilization recently as advances in technology have elevated it almost to the level of a diagnostic test for detection of certain aneuploidies. Although it is still considered a screening test, recent interest has arisen regarding population screening and whether or not this test should be universally used as first-line or whether it should still be restricted to specific high-risk populations.
In the current study, Kaimal and colleagues attempted to determine the best strategy for utilization of NIPT using a decision-analytic model. For their study many assumptions had to be made in order to allow for calculation of detection rates and for determination of cost and quality-adjusted life years. (The model followed a theoretical cohort of women desiring prenatal testing [screening or diagnostic or both] from the time of their initial test through the end of their pregnancy, the birth of their neonate, and the remainder of their own life expectancy.)
The conclusion of the authors is that traditional multiple marker screening remains the optimal choice for most women (those aged 20 to 38 years) but that NIPT becomes the optimal strategy at age 38. The goal of this study was not just to determine cost-effectiveness but also to attempt to devise a strategy that would optimize detection of aneuploidy and minimize the need for the performance of diagnostic procedures.
Data not available in this study included such things as population differences with respect to the acceptance of pregnancy termination as an option, and the potential utility of first-trimester ultrasound screening for structural defects that might not be accounted for with NIPT (such as thickened nuchal translucency, altered cardiac axis, cranial defects, and abdominal wall defects).
What this evidence means for practice
For now, the best approach would be to adhere to current recommendations as outlined in the 2015 American College of Obstetricians and Gynecologists (ACOG) Committee Opinion.1 Summarized, these are:
- Do not utilize NIPT in low-risk populations (although this opinion also suggests that patients may opt to have this test performed regardless of risk status with the understanding that detection rates are lower in low-risk populations and insurance coverage may be different).
- Offer NIPT to high-risk women as a first-line screen, as is suggested in the current study with respect to maternal age criteria (ACOG uses an age cut-off of 35 years, not 38).
- Utilize NIPT as a follow-up test after conventional testing suggests increased risk status in those patients wishing to avoid invasive diagnostic testing.
- The ACOG position remains that all women, regardless of age, who desire the most comprehensive information available regarding fetal chromosomal abnormalities should be offered diagnostic testing (chorionic villus sampling and amniocentesis).
Also, as mentioned in the committee opinion, this technology is evolving rapidly and all practitioners should closely follow this evolution with respect to changing efficacy and changing cost.
—John T. Repke, MD
Share your thoughts! Send your Letter to the Editor to [email protected]. Please include your name and the city and state in which you practice.
Reference
- American College of Obstetricians and Gynecologists. Committee Opinion No. 640. Cell-free DNA screening for fetal aneuploidy. Obstet Gynecol. 2015;126(3):e31–e37.
Cell-free DNA screening, or so-called noninvasive prenatal testing (NIPT), has had greatly increased utilization recently as advances in technology have elevated it almost to the level of a diagnostic test for detection of certain aneuploidies. Although it is still considered a screening test, recent interest has arisen regarding population screening and whether or not this test should be universally used as first-line or whether it should still be restricted to specific high-risk populations.
In the current study, Kaimal and colleagues attempted to determine the best strategy for utilization of NIPT using a decision-analytic model. For their study many assumptions had to be made in order to allow for calculation of detection rates and for determination of cost and quality-adjusted life years. (The model followed a theoretical cohort of women desiring prenatal testing [screening or diagnostic or both] from the time of their initial test through the end of their pregnancy, the birth of their neonate, and the remainder of their own life expectancy.)
The conclusion of the authors is that traditional multiple marker screening remains the optimal choice for most women (those aged 20 to 38 years) but that NIPT becomes the optimal strategy at age 38. The goal of this study was not just to determine cost-effectiveness but also to attempt to devise a strategy that would optimize detection of aneuploidy and minimize the need for the performance of diagnostic procedures.
Data not available in this study included such things as population differences with respect to the acceptance of pregnancy termination as an option, and the potential utility of first-trimester ultrasound screening for structural defects that might not be accounted for with NIPT (such as thickened nuchal translucency, altered cardiac axis, cranial defects, and abdominal wall defects).
What this evidence means for practice
For now, the best approach would be to adhere to current recommendations as outlined in the 2015 American College of Obstetricians and Gynecologists (ACOG) Committee Opinion.1 Summarized, these are:
- Do not utilize NIPT in low-risk populations (although this opinion also suggests that patients may opt to have this test performed regardless of risk status with the understanding that detection rates are lower in low-risk populations and insurance coverage may be different).
- Offer NIPT to high-risk women as a first-line screen, as is suggested in the current study with respect to maternal age criteria (ACOG uses an age cut-off of 35 years, not 38).
- Utilize NIPT as a follow-up test after conventional testing suggests increased risk status in those patients wishing to avoid invasive diagnostic testing.
- The ACOG position remains that all women, regardless of age, who desire the most comprehensive information available regarding fetal chromosomal abnormalities should be offered diagnostic testing (chorionic villus sampling and amniocentesis).
Also, as mentioned in the committee opinion, this technology is evolving rapidly and all practitioners should closely follow this evolution with respect to changing efficacy and changing cost.
—John T. Repke, MD
Share your thoughts! Send your Letter to the Editor to [email protected]. Please include your name and the city and state in which you practice.
Cell-free DNA screening, or so-called noninvasive prenatal testing (NIPT), has had greatly increased utilization recently as advances in technology have elevated it almost to the level of a diagnostic test for detection of certain aneuploidies. Although it is still considered a screening test, recent interest has arisen regarding population screening and whether or not this test should be universally used as first-line or whether it should still be restricted to specific high-risk populations.
In the current study, Kaimal and colleagues attempted to determine the best strategy for utilization of NIPT using a decision-analytic model. For their study many assumptions had to be made in order to allow for calculation of detection rates and for determination of cost and quality-adjusted life years. (The model followed a theoretical cohort of women desiring prenatal testing [screening or diagnostic or both] from the time of their initial test through the end of their pregnancy, the birth of their neonate, and the remainder of their own life expectancy.)
The conclusion of the authors is that traditional multiple marker screening remains the optimal choice for most women (those aged 20 to 38 years) but that NIPT becomes the optimal strategy at age 38. The goal of this study was not just to determine cost-effectiveness but also to attempt to devise a strategy that would optimize detection of aneuploidy and minimize the need for the performance of diagnostic procedures.
Data not available in this study included such things as population differences with respect to the acceptance of pregnancy termination as an option, and the potential utility of first-trimester ultrasound screening for structural defects that might not be accounted for with NIPT (such as thickened nuchal translucency, altered cardiac axis, cranial defects, and abdominal wall defects).
What this evidence means for practice
For now, the best approach would be to adhere to current recommendations as outlined in the 2015 American College of Obstetricians and Gynecologists (ACOG) Committee Opinion.1 Summarized, these are:
- Do not utilize NIPT in low-risk populations (although this opinion also suggests that patients may opt to have this test performed regardless of risk status with the understanding that detection rates are lower in low-risk populations and insurance coverage may be different).
- Offer NIPT to high-risk women as a first-line screen, as is suggested in the current study with respect to maternal age criteria (ACOG uses an age cut-off of 35 years, not 38).
- Utilize NIPT as a follow-up test after conventional testing suggests increased risk status in those patients wishing to avoid invasive diagnostic testing.
- The ACOG position remains that all women, regardless of age, who desire the most comprehensive information available regarding fetal chromosomal abnormalities should be offered diagnostic testing (chorionic villus sampling and amniocentesis).
Also, as mentioned in the committee opinion, this technology is evolving rapidly and all practitioners should closely follow this evolution with respect to changing efficacy and changing cost.
—John T. Repke, MD
Share your thoughts! Send your Letter to the Editor to [email protected]. Please include your name and the city and state in which you practice.
Reference
- American College of Obstetricians and Gynecologists. Committee Opinion No. 640. Cell-free DNA screening for fetal aneuploidy. Obstet Gynecol. 2015;126(3):e31–e37.
Reference
- American College of Obstetricians and Gynecologists. Committee Opinion No. 640. Cell-free DNA screening for fetal aneuploidy. Obstet Gynecol. 2015;126(3):e31–e37.