User login
N-acetylcysteine: A potential treatment for substance use disorders
Pharmacologic treatment options for many substance use disorders (SUDs) are limited. This is especially true for cocaine use disorder and cannabis use disorder, for which there are no FDA-approved medications. FDA-approved medications for other SUDs often take the form of replacement or agonist therapies (eg, nicotine replacement therapy) that substitute the effects of the substance to aid in cessation. Other pharmacotherapies treat symptoms of withdrawal, reduce craving, or provide aversive counter-conditioning if the patient consumes the substance while on the medication (eg, disulfiram).
The over-the-counter (OTC) antioxidant N-acetylcysteine (NAC) may be a potential treatment for SUDs. Although NAC is not approved by the FDA for treating SUDs, its proposed mechanism of action differs from that of current FDA-approved medications for SUDs. NAC’s potential for broad applicability, favorable adverse-effect profile, accessibility, and low cost make it an intriguing option for patients with multiple comorbidities, and potentially for individuals with polysubstance use. This article reviews the current evidence supporting NAC for treating SUDs, to provide insight about which patients may benefit from NAC and under which circumstances they are most likely to benefit.
NAC may correct glutamate dysregulation
Approximately 85% of individuals with an SUD do not seek treatment for it, and those who do are older, have a longer history of use, have more severe dependence, and have sought treatment numerous times before.1 By the time most people seek treatment, years of chronic substance use have likely led to significant brain-related adaptations. Individuals with SUDs often indicate that their substance use began as a pleasurable activity—the effects of the drug were enjoyable and they were motivated to use it again. With repeated substance use, they may begin to develop a stronger urge to use the drug, driven not necessarily by a desire for pleasure, but by compulsion.2
Numerous neural adaptations underlie the transition from “liking” a substance to engaging in the compulsive use that is characteristic of an SUD.2 For example, repeated use of an addictive substance may result in excess glutamate in the nucleus accumbens,3,4 an area of the brain that plays a critical role in motivation and learning. As a result, it has been proposed that pharmacotherapies that help correct glutamate dysregulation may be effective in promoting abstinence or preventing relapse to a substance.5,6
NAC may reverse the neural dysfunction seen in SUDs. As an OTC antioxidant that impacts glutamatergic functioning in the brain, NAC has long been used to treat acetaminophen overdose; however, in recent years, researchers have begun to tap its potential for treating substance use and psychiatric disorders. NAC is thought to upregulate the glutamate transporter (GLT-1) that removes excess glutamate from the nucleus accumbens.6 Several published reviews provide more in-depth information about the neurobiology of NAC.6-10
The adverse-effect profile of NAC is relatively benign. Nausea, vomiting, diarrhea, and sleepiness are relatively infrequent and mild.11,12 The bioavailability of NAC is about 4% to 9%, with an approximate half-life of 6.25 hours when orally administered.13 Because NAC is classified as an OTC supplement, the potency and preparation may vary by supplier. To maximize consistency, NAC should be obtained from a supplier that meets United States Pharmacopeia (USP) standards.
NAC for SUDs: Emerging evidence
Several recent reviews have described the efficacy of NAC for SUDs and other psychiatric disorders. Here we summarize the current research examining the efficacy of NAC for stimulant (ie, cocaine and methamphetamine), cannabis, tobacco, and alcohol use disorders.
Continue to: Stimulant use disorders
Stimulant use disorders. The United Nations Office for Drugs and Crime estimates that worldwide, more than 18 million people use cocaine and more than 35 million use amphetamines.14 There are currently no FDA-approved treatments for stimulant use disorders, and clinicians treating patients with cocaine or amphetamine dependence often are at a loss for how best to promote abstinence. Recent studies suggest that NAC may decrease drug-seeking behavior and cravings in adults who seek treatment. The results of studies examining NAC for treating cocaine use and methamphetamine use are summarized in Table 115-17 and Table 2,18,19 respectively.
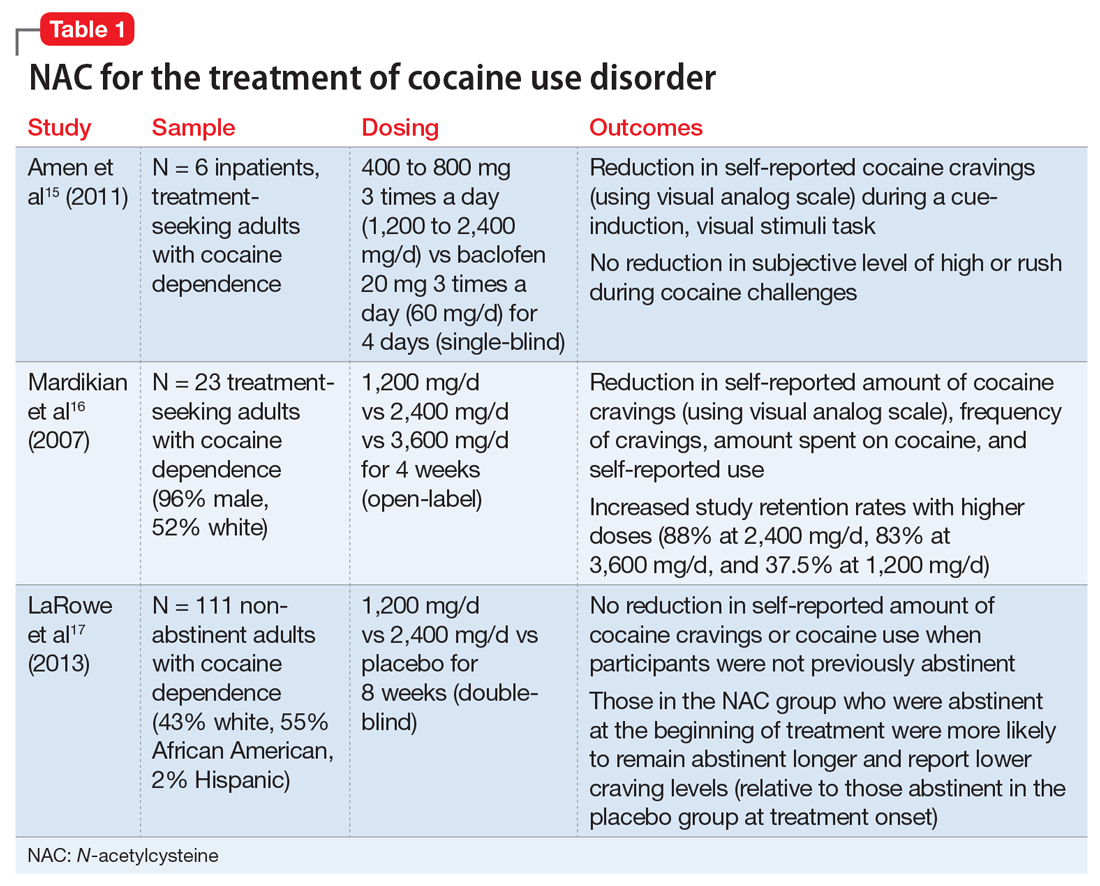
Cocaine cessation and relapse prevention. Several small pilot projects15,16 found that compared with placebo, various doses of NAC reduced craving (as measured with a visual analog scale). However, in a double-blind, placebo-controlled study, NAC did not decrease cravings or use after 8 weeks of treatment in individuals with cocaine use disorder who were still using cocaine (ie, they had not yet become abstinent). Interestingly, those who were abstinent when treatment began reported lower craving and remained abstinent longer if they received NAC (vs placebo), which suggests that NAC may be useful for preventing relapse.17

Methamphetamine cessation and relapse prevention. One study (N = 32) that evaluated the use of NAC, 1,200 mg/d for 4 weeks, vs placebo found reduced cravings among methamphetamine users who were seeking treatment.18 In contrast, a study of 31 methamphetamine users who were not seeking treatment evaluated the use of NAC, 2,400 mg/d, plus naltrexone, 200 mg/d, vs placebo for 8 weeks.19 It found no significant differences in craving or use patterns. Further research is needed to optimize the use of NAC for stimulant use disorders, and to better understand the role that abstinence plays.
Appropriate populations. The most support for use of NAC has been as an anti-relapse agent in treatment-seeking adults.
Continue to: Safety and dosing
Safety and dosing. Suggested dosages for the treatment of cocaine use disorder range from 1,200 to 3,600 mg/d (typically 600 to 1,800 mg twice daily, due to NAC’s short half-life), with higher retention rates noted in individuals who received 2,400 mg/d and 3,600 mg/d.16
Clinical implications. NAC is thought to act as an anti-relapse agent, rather than an agent that can help someone who is actively using stimulants to stop. Consequently, NAC will likely be most helpful for patients who are motivated to quit and are abstinent when they start taking NAC; however, this hypothesis needs further testing.
Cannabis use disorder
There are no FDA-approved treatments for cannabis use disorder. Individuals who use marijuana or other forms of cannabis may be less likely to report negative consequences or seek treatment compared with those who use other substances. Approximately 9% of individuals who use marijuana develop cannabis use disorder20; those who begin using marijuana earlier in adolescence are at increased risk.21 Commonly reported reasons for wanting to stop using marijuana include being concerned about health consequences, regaining or demonstrating self-control, saving money, avoiding legal consequences, obtaining or keeping employment, and reducing interpersonal conflict.22,23 Table 324-27 summarizes initial evidence that suggests NAC may be particularly useful in reducing marijuana use among adolescents (age 15 to 21).24,25

Cessation. An open-label, pilot clinical trial found significant reductions in self-reported marijuana use and craving—but not in biomarkers of use—among 24 adolescents after 4 weeks of NAC, 1,200 mg twice daily.24 In an 8-week, double-blind, randomized controlled trial of 116 adolescents, NAC, 1,200 mg twice daily, plus contingency management doubled the odds of abstinence, but had no effect on self-reported craving or use.25,26 In a sample of 302 adults, a 12-week trial of NAC, 1,200 mg twice daily, plus contingency management was no more effective than contingency management alone in promoting abstinence.27
Continue to: Appropriate populations
Appropriate populations. Evidence is stronger for use of NAC among adolescents (age 15 to 21) than for individuals older than age 21.25,27 Further research is needed to explore potential reasons for age-specific effects.
Safety and dosing. A safe and potentially efficacious dosage for the treatment of cannabis use disorder is 2,400 mg/d (1,200 mg twice daily).24,25,27
Clinical implications. Combined with contingency management, NAC might be efficacious for adolescents with cannabis use disorder, with treatment gains evident by the fourth week of treatment.24,25 To date, no clinical trials have examined the efficacy of NAC for treating cannabis use disorder without adjunctive contingency management, and research is needed to isolate the clinical effect of NAC among adolescents.
Tobacco use disorder
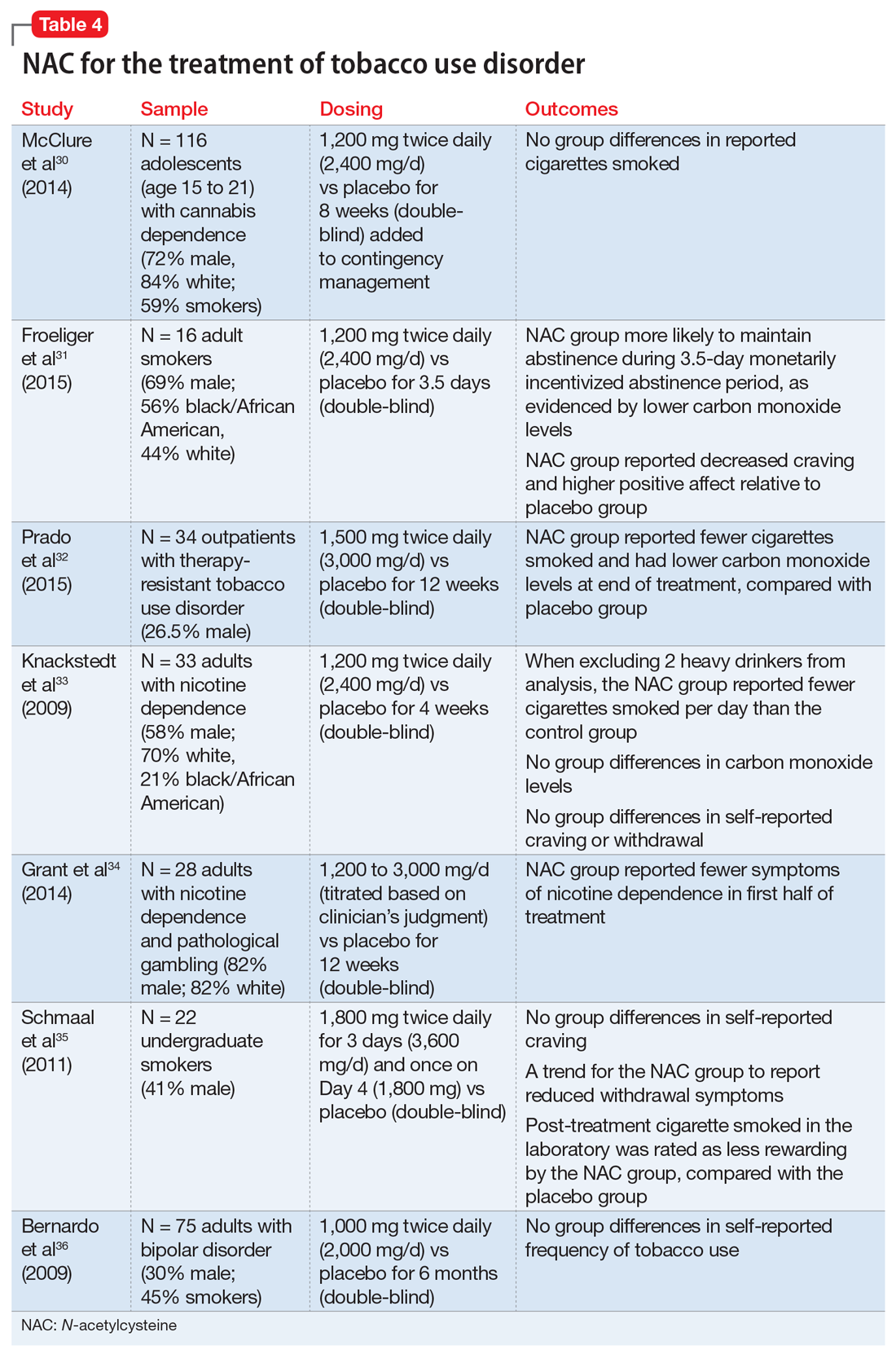
Cigarette smoking remains a leading cause of preventable death in the United States,28 and nearly 70% of people who start using tobacco become dependent.20 Existing FDA-approved treatments include nicotine replacement products, varenicline, and bupropion. Even though efficacious treatments exist, successful and sustained quit attempts are infrequent.29 NAC may exert a complementary effect to existing tobacco cessation interventions, such as varenicline.30 While these medications promote abstinence, NAC may be particularly beneficial in preventing relapse after abstinence has been achieved (Table 430-36).
Continue to: Cessation and relapse prevention
Cessation and relapse prevention. Several pilot studies found that adult smokers who received NAC (alone or in combination with another treatment) had lower carbon monoxide levels,31,32 smoked fewer cigarettes,32,33 and had fewer self-reported symptoms of nicotine dependence34 and/or less craving for cigarettes.31 However, one study of 33 smokers did not find a reduction in craving or carbon monoxide for NAC compared with placebo.33 Another pilot study of 22 young adult smokers found that those who received NAC rated their first cigarette after treatment (smoked in the laboratory) as less rewarding, relative to smokers who received a placebo.35
Secondary analyses of adults with bipolar disorder36 and adolescents with cannabis use disorder37 found no decreases in tobacco use among those who received NAC compared with placebo. However, the studies in these analyses did not specifically recruit tobacco users, and participants who were tobacco users were not necessarily interested in quitting. This may partially explain discrepant findings.
Appropriate populations. NAC has been studied mostly in adult cigarette smokers.
Safety and dosing. Suggested dosages for treating tobacco use disorder range from 1,200 to 3,600 mg/d (600 to 1,800 mg twice daily).
Continue to: Clinical implications
Clinical implications. Data on NAC’s efficacy for tobacco use disorder come from small, pilot trials. Although initial evidence is promising, it is premature to suggest NAC for smoking cessation until a fully powered, randomized clinical trial provides evidence of efficacy.
Alcohol use disorder
Alcohol use disorders are widely prevalent; 13.9% of U.S. adults met criteria in the past year, and 29.1% of U.S. adults meet criteria in their lifetime.38 Alcohol use disorders can result in significant negative consequences, including relationship problems, violent behavior, medical problems, and death. Existing FDA-approved medications for alcohol use disorder include naltrexone, acamprosate, and disulfiram.
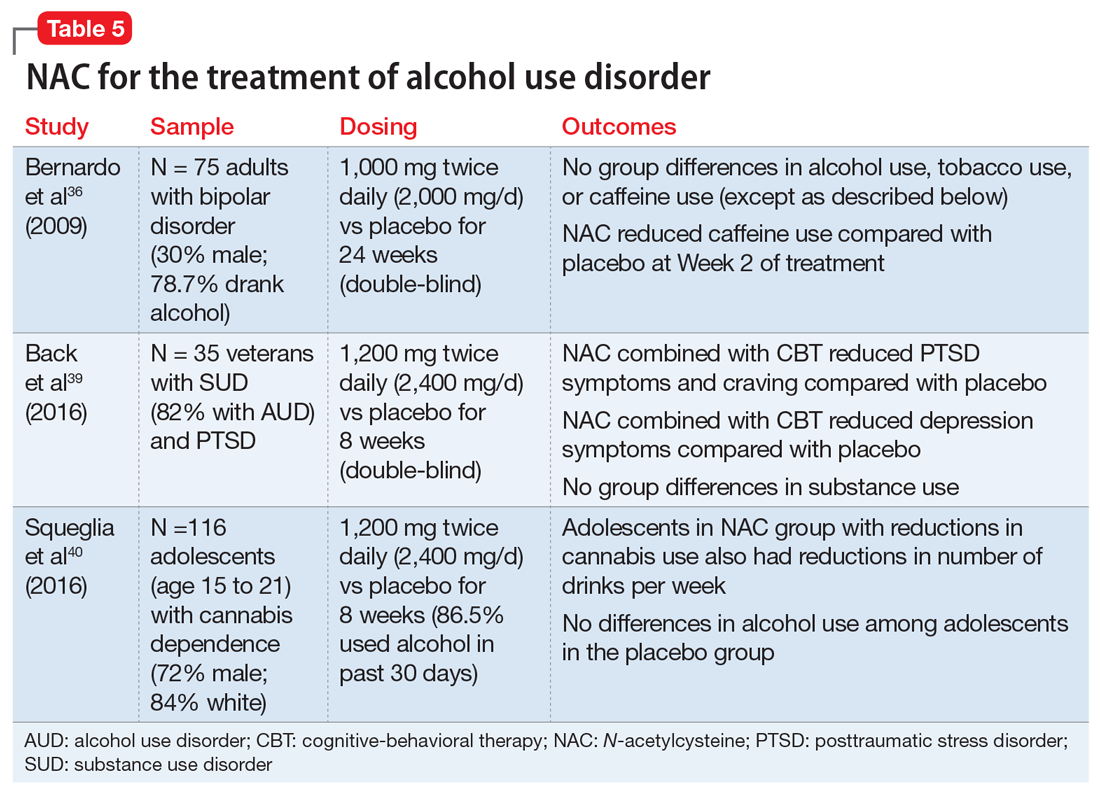
Due to the severe potential health consequences of alcohol, NAC has been examined as a possible aid in preventing relapse. However, most studies have been conducted using animals. Three studies have examined alcohol use in humans (Table 536,39,40). One was a pilot study,39 and the other 2 were secondary data analyses.36,40 None of them specifically focused on alcohol use disorders. A pilot study of 35 veterans with co-occurring posttraumautic stress disorder (PTSD) and SUDs (82% of whom had an alcohol use disorder) found that compared with placebo, NAC significantly decreased PTSD symptoms, craving, and depression.39 In a study of 75 adults with bipolar disorder, secondary alcohol use was not significantly reduced.36 However, one study suggested that NAC may decrease adolescent alcohol and marijuana co-use.40 Future work is needed to examine the potential clinical utility of NAC in individuals with alcohol use disorders.
Findings from animal studies indicate that NAC may:
- reduce alcohol-seeking41
- reduce withdrawal symptoms42
- reduce the teratogenic effects of alcohol43
- prevent alcohol toxicity44
- reduce health-related consequences of alcohol (eg, myocardial oxidative stress45 and alcohol-related steatohepatitis46).
Continue to: Appropriate populations
Appropriate populations. Pilot studies have suggested that appropriate populations may include veterans with SUD and PTSD39 and adolescents with marijuana dependence who use alcohol.40
Safety and dosing. Suggested dosages for the treatment of alcohol use disorder based on these studies range from 1,000 to 2,400 mg/d (500 to 1,200 mg twice daily).
Clinical implications. Future work is needed to determine if NAC is effective for treating alcohol use disorders. Ongoing randomized clinical trials are examining the efficacy of NAC in reducing alcohol use among individuals with alcohol use disorder. It is premature to recommend NAC for treatment of alcohol use disorders.
Other psychiatric uses
Although we have highlighted NAC’s effect on glutamatergic transmission, evidence suggests that NAC may have multiple mechanisms of action that could impact psychiatric functioning. For example, NAC may also reverse oxidative stress, which is frequently observed in psychiatric disorders such as schizophrenia and bipolar disorder.10,12 NAC also has anti-inflammatory properties. When inflammatory pathways of the CNS are dysregulated, production of neurotransmitters may be impaired, resulting in depression-like symptoms.10,12,47 Preliminary evidence suggests that NAC may be effective in treating mood-related symptoms (eg, irritability, depression) in individuals with psychiatric disorders (eg, bipolar and depressive disorders, PTSD, and SUDs) and general symptoms of schizophrenia, obsessive-compulsive disorder, and trichotillomania, although mixed findings in controlled studies suggest a need for further research.12,39
Continue to: NAC: A promising candidate
NAC: A promising candidate
Initial evidence suggests NAC may be helpful for treating patients with SUDs. A patient seeking SUD treatment who is treated with NAC may experience a decreased drive, craving, or compulsion to use. Notably, NAC may be particularly useful in preventing relapse after an individual has achieved abstinence. Evidence suggests that NAC may be useful in the treatment of adults with cocaine use disorders who have achieved abstinence, and adolescents with cannabis use disorders. Preliminary results for adult tobacco use disorder are also promising. Human data examining the efficacy of NAC for alcohol use disorder is limited. Researchers’ ongoing challenge is to identify which patients with which SUDs are most likely to benefit from NAC, and to create clear clinical guidelines for the provider.
Bottom Line
N-acetylcysteine is likely to have modest effects for some patients who have a substance use disorder, particularly adults who use cocaine and adolescents who use marijuana. It may be useful in preventing relapse to substance use after an individual has achieved abstinence.
Related Resources
- Deepmala, Slattery J, Kumar N, et al. Clinical trials of N-acetylcysteine in psychiatry and neurology: a systematic review. Neurosci Biobehav Rev. 2015;55:294-321.
- Roberts-Wolfe D, Kalivas P. Glutamate transporter GLT-1 as a therapeutic target for substance use disorders. CNS Neurol Disord Drug Targets. 2015;14(6):745-756.
- National Institute on Drug Abuse. Treatment approaches for drug addiction. https://www.drugabuse.gov/publications/drugfacts/treatment-approaches-drug-addiction.
Drug Brand Names
Acamprosate • Campral
Acetaminophen • Tylenol
Baclofen • Lioresal
Bupropion • Zyban
Disulfiram • Antabuse
Naltrexone • Revia,Vivitrol
Varenicline • Chantix
1. Grella CE, Karno MP, Warda US, et al. Perceptions of need and help received for substance dependence in a national probability survey. Psychiatr Serv. 2009;60(8):1068-1074.
2. Everitt BJ, Robbins TW. Drug addiction: updating actions to habits to compulsions ten years on. Annu Rev Psychol. 2016;67:23-50.
3. McFarland K, Lapish CC, Kalivas PW. Prefrontal glutamate release into the core of the nucleus accumbens mediates cocaine-induced reinstatement of drug-seeking behavior. J Neurosci. 2003;23(8):3531-3537.
4. LaLumiere RT, Kalivas PW. Glutamate release in the nucleus accumbens core is necessary for heroin seeking. J Neurosci. 2008;28(12):3170-3177.
5. Kalivas PW, Volkow ND. New medications for drug addiction hiding in glutamatergic neuroplasticity. Mol Psychiatry. 2011;16(10):974-986.
6. Roberts-Wolfe D, Kalivas PW. Glutamate transporter GLT-1 as a therapeutic target for substance use disorders. CNS Neurol Disord Drug Targets. 2015;14(6):745-756.
7. Berk M, Malhi GS, Gray LJ, et al. The promise of N-acetylcysteine in neuropsychiatry. Trends Pharmacol Sci. 2013;34(3):167-177.
8. McClure EA, Gipson CD, Malcolm RJ, et al. Potential role of N-acetylcysteine in the management of substance use disorders. CNS drugs. 2014;28(2):95-106.
9. Deepmala, Slattery J, Kumar N, et al. Clinical trials of N-acetylcysteine in psychiatry and neurology: a systematic review. Neurosci Biobehav Rev. 2015;55:294-321.
10. Minarini A, Ferrari S, Galletti M, et al. N-acetylcysteine in the treatment of psychiatric disorders: current status and future prospects. Expert Opin Drug Metab Toxicol. 2017;13(3):279-292.
11. Grandjean EM, Berthet P, Ruffman R, et al. Efficacy of oral long-term N‑acetylcysteine in chronic bronchopulmonary disease: a meta-analysis of published double-blind, placebo-controlled clinical trials. Clin Ther. 2000;22(2):209‑221.
12. Rhodes K, Braakhuis A. Performance and side effects of supplementation with N-acetylcysteine: a systematic review and meta-analysis. Sports Med. 2017;47(8):1619-1636.
13. Olsson B, Johansson M, Gabrielsson J, et al. Pharmacokinetics and bioavailability of reduced and oxidized N-acetylcysteine. Eur J Clin Pharmacol. 1988;34(1):77-82.
14. United Nations Office on Drugs and Crime. World Drug Report 2016 (United Nations publication, Sales No. E.16.XI.7). https://www.unodc.org/doc/wdr2016/WORLD_DRUG_REPORT_2016_web.pdf. Published May 2016. Accessed April 26, 2018.
15. Amen SL, Piacentine LB, Ahmad ME, et al. Repeated N-acetyl cysteine reduces cocaine seeking in rodents and craving in cocaine-dependent humans. Neuropsychopharmacology. 2011;36(4):871-878.
16. Mardikian PN, LaRowe SD, Hedden S, et al. An open-label trial of N-acetylcysteine for the treatment of cocaine dependence: a pilot study. Prog Neuropsychopharmacol Biol Psychiatry. 2007;31(2):389-394.
17. LaRowe SD, Kalivas PW, Nicholas JS, et al. A double‐blind placebo‐controlled trial of N‐acetylcysteine in the treatment of cocaine dependence. Am J Addict. 2013;22(5):443-452.
18. Mousavi SG, Sharbafchi MR, Salehi M, et al. The efficacy of N-acetylcysteine in the treatment of methamphetamine dependence: a double-blind controlled, crossover study. Arch Iran Med. 2015;18(1):28-33.
19. Grant JE, Odlaug BL, Kim SW. A double-blind, placebo-controlled study of N-acetyl cysteine plus naltrexone for methamphetamine dependence. Eur Neuropsychopharmacol. 2010;20(11):823-828.
20. Lopez-Quintero C, Pérez de los Cobos J, Hasin DS, et al. Probability and predictors of transition from first use to dependence on nicotine, alcohol, cannabis, and cocaine: results of the National Epidemiologic Survey on Alcohol and Related Conditions (NESARC). Drug Alcohol Depend. 2011;115(1-2):120-130.
21. Chen CY, O’Brien MS, Anthony JC. Who becomes cannabis dependent soon after onset of use? Epidemiological evidence from the United States: 2000-2001. Drug Alcohol Depend. 2005;79(1):11-22.
22. Copersino ML, Boyd SJ, Tashkin DP, et al. Quitting among non-treatment-seeking marijuana users: reasons and changes in other substance use. Am J Addict. 2006;15(4):297-302.
23. Weiner MD, Sussman S, McCuller WJ, et al. Factors in marijuana cessation among high-risk youth. J Drug Educ. 1999;29(4):337-357.
24. Gray KM, Watson NL, Carpenter MJ, et al. N-acetylcysteine (NAC) in young marijuana users: an open-label pilot study. Am J Addict. 2010;19(2):187-189.
25. Gray KM, Carpenter MJ, Baker NL, et al. A double-blind randomized controlled trial of N-acetylcysteine in cannabis-dependent adolescents. Am J Psychiatry. 2012;169(8):805-812.
26. Roten AT, Baker NL, Gray KM. Marijuana craving trajectories in an adolescent marijuana cessation pharmacotherapy trial. Addict Behav. 2013;38(3):1788-1791.
27. Gray KM, Sonne SC, McClure EA, et al. A randomized placebo-controlled trial of N-acetylcysteine for cannabis use disorder in adults. Drug Alcohol Depend. 2017;177:249-257.
28. Rostron B. Mortality risks associated with environmental tobacco smoke exposure in the United States. Nicotine Tob Res. 2013;15(10):1722-1728.
29. Centers for Disease Control and Prevention. Quitting smoking among adults – United States, 2001–2010. MMWR. 2011;60(44):1513-1519.
30. McClure EA, Baker NL, Gipson CD, et al. An open-label pilot trial of N-acetylcysteine and varenicline in adult cigarette smokers. Am J Drug Alcohol Abuse. 2015;41(1):52-56.
31. Froeliger B, McConnell P, Stankeviciute N, et al. The effects of N-acetylcysteine on frontostriatal resting-state functional connectivity, withdrawal symptoms and smoking abstinence: a double-blind, placebo-controlled fMRI pilot study. Drug Alcohol Depend. 2015;156:234-242.
32. Prado E, Maes M, Piccoli LG, et al. N-acetylcysteine for therapy-resistant tobacco use disorder: a pilot study. Redox Rep. 2015;20(5):215-222.
33. Knackstedt LA, LaRowe S, Mardikian P, et al. The role of cystine-glutamate exchange in nicotine dependence in rats and humans. Biol Psychiatry. 2009;65(10):841-845.
34. Grant JE, Odlaug BL, Chamberlain SR, et al. A randomized, placebo-controlled trial of N-acetylcysteine plus imaginal desensitization for nicotine-dependent pathological gamblers. J Clin Psychiatry. 2014;75(1):39-45.
35. Schmaal L, Berk L, Hulstijn KP, et al. Efficacy of N-acetylcysteine in the treatment of nicotine dependence: a double-blind placebo-controlled pilot study. Eur Addiction Res. 2011;17(4):211-216.
36. Bernardo M, Dodd S, Gama CS, et al. Effects of N‐acetylcysteine on substance use in bipolar disorder: a randomised placebo‐controlled clinical trial. Acta Neuropsychiatr. 2009;21(5):239-245.
37. McClure EA, Baker NL, Gray KM. Cigarette smoking during an N-acetylcysteine-assisted cannabis cessation trial in adolescents. Am J Drug Alcohol Abuse. 2014;40(4):285-291.
38. Grant BF, Goldstein RB, Saha TD, et al. Epidemiology of DSM-5 alcohol use disorder: Results from the National Epidemiologic Survey on Alcohol and Related Conditions III. JAMA Psychiatry. 2015;72(8):757-766.
39. Back SE, McCauley JL, Korte KJ, et al. A double-blind randomized controlled pilot trial of N-acetylcysteine in veterans with PTSD and substance use disorders. J Clin Psychiatry. 2016;77(11):e1439-e1446.
40. Squeglia LM, Baker NL, McClure EA, et al. Alcohol use during a trial of N-acetylcysteine for adolescent marijuana cessation. Addict Behav. 2016;63:172-177.
41. Lebourgeois S, González-Marín MC, Jeanblanc J, et al. Effect of N-acetylcysteine on motivation, seeking and relapse to ethanol self-administration. Addict Biol. 2018;23(2):643-652.
42. Schneider R Jr, Santos CF, Clarimundo V, et al. N-acetylcysteine prevents behavioral and biochemical changes induced by alcohol cessation in rats. Alcohol. 2015;49(3):259-263.
43. Parnell SE, Sulik KK, Dehart DB, et al. Reduction of ethanol-induced ocular abnormalities in mice via dietary administration of N-acetylcysteine. Alcohol. 2010;44(7-8):699-705.
44. Ozkol H, Bulut G, Balahoroglu R, et al. Protective effects of Selenium, N-acetylcysteine and Vitamin E against acute ethanol intoxication in rats. Biol Trace Elem Res. 2017;175(1):177-185.
45. Seiva FR, Amauchi JF, Rocha KK, et al. Alcoholism and alcohol abstinence: N-acetylcysteine to improve energy expenditure, myocardial oxidative stress, and energy metabolism in alcoholic heart disease. Alcohol. 2009;43(8):649-656.
46. Setshedi M, Longato L, Petersen DR, et al. Limited therapeutic effect of N‐acetylcysteine on hepatic insulin resistance in an experimental model of alcohol‐induced steatohepatitis. Alcohol Clin Exp Res. 2011;35(12):2139-2151.
47. Miller AH, Maletic V, Raison CL. Inflammation and its discontents: the role of cytokines in the pathophysiology of major depression. Biol Psychiatry. 2009;65(9):732-741.
Pharmacologic treatment options for many substance use disorders (SUDs) are limited. This is especially true for cocaine use disorder and cannabis use disorder, for which there are no FDA-approved medications. FDA-approved medications for other SUDs often take the form of replacement or agonist therapies (eg, nicotine replacement therapy) that substitute the effects of the substance to aid in cessation. Other pharmacotherapies treat symptoms of withdrawal, reduce craving, or provide aversive counter-conditioning if the patient consumes the substance while on the medication (eg, disulfiram).
The over-the-counter (OTC) antioxidant N-acetylcysteine (NAC) may be a potential treatment for SUDs. Although NAC is not approved by the FDA for treating SUDs, its proposed mechanism of action differs from that of current FDA-approved medications for SUDs. NAC’s potential for broad applicability, favorable adverse-effect profile, accessibility, and low cost make it an intriguing option for patients with multiple comorbidities, and potentially for individuals with polysubstance use. This article reviews the current evidence supporting NAC for treating SUDs, to provide insight about which patients may benefit from NAC and under which circumstances they are most likely to benefit.
NAC may correct glutamate dysregulation
Approximately 85% of individuals with an SUD do not seek treatment for it, and those who do are older, have a longer history of use, have more severe dependence, and have sought treatment numerous times before.1 By the time most people seek treatment, years of chronic substance use have likely led to significant brain-related adaptations. Individuals with SUDs often indicate that their substance use began as a pleasurable activity—the effects of the drug were enjoyable and they were motivated to use it again. With repeated substance use, they may begin to develop a stronger urge to use the drug, driven not necessarily by a desire for pleasure, but by compulsion.2
Numerous neural adaptations underlie the transition from “liking” a substance to engaging in the compulsive use that is characteristic of an SUD.2 For example, repeated use of an addictive substance may result in excess glutamate in the nucleus accumbens,3,4 an area of the brain that plays a critical role in motivation and learning. As a result, it has been proposed that pharmacotherapies that help correct glutamate dysregulation may be effective in promoting abstinence or preventing relapse to a substance.5,6
NAC may reverse the neural dysfunction seen in SUDs. As an OTC antioxidant that impacts glutamatergic functioning in the brain, NAC has long been used to treat acetaminophen overdose; however, in recent years, researchers have begun to tap its potential for treating substance use and psychiatric disorders. NAC is thought to upregulate the glutamate transporter (GLT-1) that removes excess glutamate from the nucleus accumbens.6 Several published reviews provide more in-depth information about the neurobiology of NAC.6-10
The adverse-effect profile of NAC is relatively benign. Nausea, vomiting, diarrhea, and sleepiness are relatively infrequent and mild.11,12 The bioavailability of NAC is about 4% to 9%, with an approximate half-life of 6.25 hours when orally administered.13 Because NAC is classified as an OTC supplement, the potency and preparation may vary by supplier. To maximize consistency, NAC should be obtained from a supplier that meets United States Pharmacopeia (USP) standards.
NAC for SUDs: Emerging evidence
Several recent reviews have described the efficacy of NAC for SUDs and other psychiatric disorders. Here we summarize the current research examining the efficacy of NAC for stimulant (ie, cocaine and methamphetamine), cannabis, tobacco, and alcohol use disorders.
Continue to: Stimulant use disorders
Stimulant use disorders. The United Nations Office for Drugs and Crime estimates that worldwide, more than 18 million people use cocaine and more than 35 million use amphetamines.14 There are currently no FDA-approved treatments for stimulant use disorders, and clinicians treating patients with cocaine or amphetamine dependence often are at a loss for how best to promote abstinence. Recent studies suggest that NAC may decrease drug-seeking behavior and cravings in adults who seek treatment. The results of studies examining NAC for treating cocaine use and methamphetamine use are summarized in Table 115-17 and Table 2,18,19 respectively.

Cocaine cessation and relapse prevention. Several small pilot projects15,16 found that compared with placebo, various doses of NAC reduced craving (as measured with a visual analog scale). However, in a double-blind, placebo-controlled study, NAC did not decrease cravings or use after 8 weeks of treatment in individuals with cocaine use disorder who were still using cocaine (ie, they had not yet become abstinent). Interestingly, those who were abstinent when treatment began reported lower craving and remained abstinent longer if they received NAC (vs placebo), which suggests that NAC may be useful for preventing relapse.17

Methamphetamine cessation and relapse prevention. One study (N = 32) that evaluated the use of NAC, 1,200 mg/d for 4 weeks, vs placebo found reduced cravings among methamphetamine users who were seeking treatment.18 In contrast, a study of 31 methamphetamine users who were not seeking treatment evaluated the use of NAC, 2,400 mg/d, plus naltrexone, 200 mg/d, vs placebo for 8 weeks.19 It found no significant differences in craving or use patterns. Further research is needed to optimize the use of NAC for stimulant use disorders, and to better understand the role that abstinence plays.
Appropriate populations. The most support for use of NAC has been as an anti-relapse agent in treatment-seeking adults.
Continue to: Safety and dosing
Safety and dosing. Suggested dosages for the treatment of cocaine use disorder range from 1,200 to 3,600 mg/d (typically 600 to 1,800 mg twice daily, due to NAC’s short half-life), with higher retention rates noted in individuals who received 2,400 mg/d and 3,600 mg/d.16
Clinical implications. NAC is thought to act as an anti-relapse agent, rather than an agent that can help someone who is actively using stimulants to stop. Consequently, NAC will likely be most helpful for patients who are motivated to quit and are abstinent when they start taking NAC; however, this hypothesis needs further testing.
Cannabis use disorder
There are no FDA-approved treatments for cannabis use disorder. Individuals who use marijuana or other forms of cannabis may be less likely to report negative consequences or seek treatment compared with those who use other substances. Approximately 9% of individuals who use marijuana develop cannabis use disorder20; those who begin using marijuana earlier in adolescence are at increased risk.21 Commonly reported reasons for wanting to stop using marijuana include being concerned about health consequences, regaining or demonstrating self-control, saving money, avoiding legal consequences, obtaining or keeping employment, and reducing interpersonal conflict.22,23 Table 324-27 summarizes initial evidence that suggests NAC may be particularly useful in reducing marijuana use among adolescents (age 15 to 21).24,25

Cessation. An open-label, pilot clinical trial found significant reductions in self-reported marijuana use and craving—but not in biomarkers of use—among 24 adolescents after 4 weeks of NAC, 1,200 mg twice daily.24 In an 8-week, double-blind, randomized controlled trial of 116 adolescents, NAC, 1,200 mg twice daily, plus contingency management doubled the odds of abstinence, but had no effect on self-reported craving or use.25,26 In a sample of 302 adults, a 12-week trial of NAC, 1,200 mg twice daily, plus contingency management was no more effective than contingency management alone in promoting abstinence.27
Continue to: Appropriate populations
Appropriate populations. Evidence is stronger for use of NAC among adolescents (age 15 to 21) than for individuals older than age 21.25,27 Further research is needed to explore potential reasons for age-specific effects.
Safety and dosing. A safe and potentially efficacious dosage for the treatment of cannabis use disorder is 2,400 mg/d (1,200 mg twice daily).24,25,27
Clinical implications. Combined with contingency management, NAC might be efficacious for adolescents with cannabis use disorder, with treatment gains evident by the fourth week of treatment.24,25 To date, no clinical trials have examined the efficacy of NAC for treating cannabis use disorder without adjunctive contingency management, and research is needed to isolate the clinical effect of NAC among adolescents.
Tobacco use disorder
Cigarette smoking remains a leading cause of preventable death in the United States,28 and nearly 70% of people who start using tobacco become dependent.20 Existing FDA-approved treatments include nicotine replacement products, varenicline, and bupropion. Even though efficacious treatments exist, successful and sustained quit attempts are infrequent.29 NAC may exert a complementary effect to existing tobacco cessation interventions, such as varenicline.30 While these medications promote abstinence, NAC may be particularly beneficial in preventing relapse after abstinence has been achieved (Table 430-36).

Continue to: Cessation and relapse prevention
Cessation and relapse prevention. Several pilot studies found that adult smokers who received NAC (alone or in combination with another treatment) had lower carbon monoxide levels,31,32 smoked fewer cigarettes,32,33 and had fewer self-reported symptoms of nicotine dependence34 and/or less craving for cigarettes.31 However, one study of 33 smokers did not find a reduction in craving or carbon monoxide for NAC compared with placebo.33 Another pilot study of 22 young adult smokers found that those who received NAC rated their first cigarette after treatment (smoked in the laboratory) as less rewarding, relative to smokers who received a placebo.35
Secondary analyses of adults with bipolar disorder36 and adolescents with cannabis use disorder37 found no decreases in tobacco use among those who received NAC compared with placebo. However, the studies in these analyses did not specifically recruit tobacco users, and participants who were tobacco users were not necessarily interested in quitting. This may partially explain discrepant findings.
Appropriate populations. NAC has been studied mostly in adult cigarette smokers.
Safety and dosing. Suggested dosages for treating tobacco use disorder range from 1,200 to 3,600 mg/d (600 to 1,800 mg twice daily).
Continue to: Clinical implications
Clinical implications. Data on NAC’s efficacy for tobacco use disorder come from small, pilot trials. Although initial evidence is promising, it is premature to suggest NAC for smoking cessation until a fully powered, randomized clinical trial provides evidence of efficacy.
Alcohol use disorder
Alcohol use disorders are widely prevalent; 13.9% of U.S. adults met criteria in the past year, and 29.1% of U.S. adults meet criteria in their lifetime.38 Alcohol use disorders can result in significant negative consequences, including relationship problems, violent behavior, medical problems, and death. Existing FDA-approved medications for alcohol use disorder include naltrexone, acamprosate, and disulfiram.
Due to the severe potential health consequences of alcohol, NAC has been examined as a possible aid in preventing relapse. However, most studies have been conducted using animals. Three studies have examined alcohol use in humans (Table 536,39,40). One was a pilot study,39 and the other 2 were secondary data analyses.36,40 None of them specifically focused on alcohol use disorders. A pilot study of 35 veterans with co-occurring posttraumautic stress disorder (PTSD) and SUDs (82% of whom had an alcohol use disorder) found that compared with placebo, NAC significantly decreased PTSD symptoms, craving, and depression.39 In a study of 75 adults with bipolar disorder, secondary alcohol use was not significantly reduced.36 However, one study suggested that NAC may decrease adolescent alcohol and marijuana co-use.40 Future work is needed to examine the potential clinical utility of NAC in individuals with alcohol use disorders.

Findings from animal studies indicate that NAC may:
- reduce alcohol-seeking41
- reduce withdrawal symptoms42
- reduce the teratogenic effects of alcohol43
- prevent alcohol toxicity44
- reduce health-related consequences of alcohol (eg, myocardial oxidative stress45 and alcohol-related steatohepatitis46).
Continue to: Appropriate populations
Appropriate populations. Pilot studies have suggested that appropriate populations may include veterans with SUD and PTSD39 and adolescents with marijuana dependence who use alcohol.40
Safety and dosing. Suggested dosages for the treatment of alcohol use disorder based on these studies range from 1,000 to 2,400 mg/d (500 to 1,200 mg twice daily).
Clinical implications. Future work is needed to determine if NAC is effective for treating alcohol use disorders. Ongoing randomized clinical trials are examining the efficacy of NAC in reducing alcohol use among individuals with alcohol use disorder. It is premature to recommend NAC for treatment of alcohol use disorders.
Other psychiatric uses
Although we have highlighted NAC’s effect on glutamatergic transmission, evidence suggests that NAC may have multiple mechanisms of action that could impact psychiatric functioning. For example, NAC may also reverse oxidative stress, which is frequently observed in psychiatric disorders such as schizophrenia and bipolar disorder.10,12 NAC also has anti-inflammatory properties. When inflammatory pathways of the CNS are dysregulated, production of neurotransmitters may be impaired, resulting in depression-like symptoms.10,12,47 Preliminary evidence suggests that NAC may be effective in treating mood-related symptoms (eg, irritability, depression) in individuals with psychiatric disorders (eg, bipolar and depressive disorders, PTSD, and SUDs) and general symptoms of schizophrenia, obsessive-compulsive disorder, and trichotillomania, although mixed findings in controlled studies suggest a need for further research.12,39
Continue to: NAC: A promising candidate
NAC: A promising candidate
Initial evidence suggests NAC may be helpful for treating patients with SUDs. A patient seeking SUD treatment who is treated with NAC may experience a decreased drive, craving, or compulsion to use. Notably, NAC may be particularly useful in preventing relapse after an individual has achieved abstinence. Evidence suggests that NAC may be useful in the treatment of adults with cocaine use disorders who have achieved abstinence, and adolescents with cannabis use disorders. Preliminary results for adult tobacco use disorder are also promising. Human data examining the efficacy of NAC for alcohol use disorder is limited. Researchers’ ongoing challenge is to identify which patients with which SUDs are most likely to benefit from NAC, and to create clear clinical guidelines for the provider.
Bottom Line
N-acetylcysteine is likely to have modest effects for some patients who have a substance use disorder, particularly adults who use cocaine and adolescents who use marijuana. It may be useful in preventing relapse to substance use after an individual has achieved abstinence.
Related Resources
- Deepmala, Slattery J, Kumar N, et al. Clinical trials of N-acetylcysteine in psychiatry and neurology: a systematic review. Neurosci Biobehav Rev. 2015;55:294-321.
- Roberts-Wolfe D, Kalivas P. Glutamate transporter GLT-1 as a therapeutic target for substance use disorders. CNS Neurol Disord Drug Targets. 2015;14(6):745-756.
- National Institute on Drug Abuse. Treatment approaches for drug addiction. https://www.drugabuse.gov/publications/drugfacts/treatment-approaches-drug-addiction.
Drug Brand Names
Acamprosate • Campral
Acetaminophen • Tylenol
Baclofen • Lioresal
Bupropion • Zyban
Disulfiram • Antabuse
Naltrexone • Revia,Vivitrol
Varenicline • Chantix
Pharmacologic treatment options for many substance use disorders (SUDs) are limited. This is especially true for cocaine use disorder and cannabis use disorder, for which there are no FDA-approved medications. FDA-approved medications for other SUDs often take the form of replacement or agonist therapies (eg, nicotine replacement therapy) that substitute the effects of the substance to aid in cessation. Other pharmacotherapies treat symptoms of withdrawal, reduce craving, or provide aversive counter-conditioning if the patient consumes the substance while on the medication (eg, disulfiram).
The over-the-counter (OTC) antioxidant N-acetylcysteine (NAC) may be a potential treatment for SUDs. Although NAC is not approved by the FDA for treating SUDs, its proposed mechanism of action differs from that of current FDA-approved medications for SUDs. NAC’s potential for broad applicability, favorable adverse-effect profile, accessibility, and low cost make it an intriguing option for patients with multiple comorbidities, and potentially for individuals with polysubstance use. This article reviews the current evidence supporting NAC for treating SUDs, to provide insight about which patients may benefit from NAC and under which circumstances they are most likely to benefit.
NAC may correct glutamate dysregulation
Approximately 85% of individuals with an SUD do not seek treatment for it, and those who do are older, have a longer history of use, have more severe dependence, and have sought treatment numerous times before.1 By the time most people seek treatment, years of chronic substance use have likely led to significant brain-related adaptations. Individuals with SUDs often indicate that their substance use began as a pleasurable activity—the effects of the drug were enjoyable and they were motivated to use it again. With repeated substance use, they may begin to develop a stronger urge to use the drug, driven not necessarily by a desire for pleasure, but by compulsion.2
Numerous neural adaptations underlie the transition from “liking” a substance to engaging in the compulsive use that is characteristic of an SUD.2 For example, repeated use of an addictive substance may result in excess glutamate in the nucleus accumbens,3,4 an area of the brain that plays a critical role in motivation and learning. As a result, it has been proposed that pharmacotherapies that help correct glutamate dysregulation may be effective in promoting abstinence or preventing relapse to a substance.5,6
NAC may reverse the neural dysfunction seen in SUDs. As an OTC antioxidant that impacts glutamatergic functioning in the brain, NAC has long been used to treat acetaminophen overdose; however, in recent years, researchers have begun to tap its potential for treating substance use and psychiatric disorders. NAC is thought to upregulate the glutamate transporter (GLT-1) that removes excess glutamate from the nucleus accumbens.6 Several published reviews provide more in-depth information about the neurobiology of NAC.6-10
The adverse-effect profile of NAC is relatively benign. Nausea, vomiting, diarrhea, and sleepiness are relatively infrequent and mild.11,12 The bioavailability of NAC is about 4% to 9%, with an approximate half-life of 6.25 hours when orally administered.13 Because NAC is classified as an OTC supplement, the potency and preparation may vary by supplier. To maximize consistency, NAC should be obtained from a supplier that meets United States Pharmacopeia (USP) standards.
NAC for SUDs: Emerging evidence
Several recent reviews have described the efficacy of NAC for SUDs and other psychiatric disorders. Here we summarize the current research examining the efficacy of NAC for stimulant (ie, cocaine and methamphetamine), cannabis, tobacco, and alcohol use disorders.
Continue to: Stimulant use disorders
Stimulant use disorders. The United Nations Office for Drugs and Crime estimates that worldwide, more than 18 million people use cocaine and more than 35 million use amphetamines.14 There are currently no FDA-approved treatments for stimulant use disorders, and clinicians treating patients with cocaine or amphetamine dependence often are at a loss for how best to promote abstinence. Recent studies suggest that NAC may decrease drug-seeking behavior and cravings in adults who seek treatment. The results of studies examining NAC for treating cocaine use and methamphetamine use are summarized in Table 115-17 and Table 2,18,19 respectively.

Cocaine cessation and relapse prevention. Several small pilot projects15,16 found that compared with placebo, various doses of NAC reduced craving (as measured with a visual analog scale). However, in a double-blind, placebo-controlled study, NAC did not decrease cravings or use after 8 weeks of treatment in individuals with cocaine use disorder who were still using cocaine (ie, they had not yet become abstinent). Interestingly, those who were abstinent when treatment began reported lower craving and remained abstinent longer if they received NAC (vs placebo), which suggests that NAC may be useful for preventing relapse.17

Methamphetamine cessation and relapse prevention. One study (N = 32) that evaluated the use of NAC, 1,200 mg/d for 4 weeks, vs placebo found reduced cravings among methamphetamine users who were seeking treatment.18 In contrast, a study of 31 methamphetamine users who were not seeking treatment evaluated the use of NAC, 2,400 mg/d, plus naltrexone, 200 mg/d, vs placebo for 8 weeks.19 It found no significant differences in craving or use patterns. Further research is needed to optimize the use of NAC for stimulant use disorders, and to better understand the role that abstinence plays.
Appropriate populations. The most support for use of NAC has been as an anti-relapse agent in treatment-seeking adults.
Continue to: Safety and dosing
Safety and dosing. Suggested dosages for the treatment of cocaine use disorder range from 1,200 to 3,600 mg/d (typically 600 to 1,800 mg twice daily, due to NAC’s short half-life), with higher retention rates noted in individuals who received 2,400 mg/d and 3,600 mg/d.16
Clinical implications. NAC is thought to act as an anti-relapse agent, rather than an agent that can help someone who is actively using stimulants to stop. Consequently, NAC will likely be most helpful for patients who are motivated to quit and are abstinent when they start taking NAC; however, this hypothesis needs further testing.
Cannabis use disorder
There are no FDA-approved treatments for cannabis use disorder. Individuals who use marijuana or other forms of cannabis may be less likely to report negative consequences or seek treatment compared with those who use other substances. Approximately 9% of individuals who use marijuana develop cannabis use disorder20; those who begin using marijuana earlier in adolescence are at increased risk.21 Commonly reported reasons for wanting to stop using marijuana include being concerned about health consequences, regaining or demonstrating self-control, saving money, avoiding legal consequences, obtaining or keeping employment, and reducing interpersonal conflict.22,23 Table 324-27 summarizes initial evidence that suggests NAC may be particularly useful in reducing marijuana use among adolescents (age 15 to 21).24,25

Cessation. An open-label, pilot clinical trial found significant reductions in self-reported marijuana use and craving—but not in biomarkers of use—among 24 adolescents after 4 weeks of NAC, 1,200 mg twice daily.24 In an 8-week, double-blind, randomized controlled trial of 116 adolescents, NAC, 1,200 mg twice daily, plus contingency management doubled the odds of abstinence, but had no effect on self-reported craving or use.25,26 In a sample of 302 adults, a 12-week trial of NAC, 1,200 mg twice daily, plus contingency management was no more effective than contingency management alone in promoting abstinence.27
Continue to: Appropriate populations
Appropriate populations. Evidence is stronger for use of NAC among adolescents (age 15 to 21) than for individuals older than age 21.25,27 Further research is needed to explore potential reasons for age-specific effects.
Safety and dosing. A safe and potentially efficacious dosage for the treatment of cannabis use disorder is 2,400 mg/d (1,200 mg twice daily).24,25,27
Clinical implications. Combined with contingency management, NAC might be efficacious for adolescents with cannabis use disorder, with treatment gains evident by the fourth week of treatment.24,25 To date, no clinical trials have examined the efficacy of NAC for treating cannabis use disorder without adjunctive contingency management, and research is needed to isolate the clinical effect of NAC among adolescents.
Tobacco use disorder
Cigarette smoking remains a leading cause of preventable death in the United States,28 and nearly 70% of people who start using tobacco become dependent.20 Existing FDA-approved treatments include nicotine replacement products, varenicline, and bupropion. Even though efficacious treatments exist, successful and sustained quit attempts are infrequent.29 NAC may exert a complementary effect to existing tobacco cessation interventions, such as varenicline.30 While these medications promote abstinence, NAC may be particularly beneficial in preventing relapse after abstinence has been achieved (Table 430-36).

Continue to: Cessation and relapse prevention
Cessation and relapse prevention. Several pilot studies found that adult smokers who received NAC (alone or in combination with another treatment) had lower carbon monoxide levels,31,32 smoked fewer cigarettes,32,33 and had fewer self-reported symptoms of nicotine dependence34 and/or less craving for cigarettes.31 However, one study of 33 smokers did not find a reduction in craving or carbon monoxide for NAC compared with placebo.33 Another pilot study of 22 young adult smokers found that those who received NAC rated their first cigarette after treatment (smoked in the laboratory) as less rewarding, relative to smokers who received a placebo.35
Secondary analyses of adults with bipolar disorder36 and adolescents with cannabis use disorder37 found no decreases in tobacco use among those who received NAC compared with placebo. However, the studies in these analyses did not specifically recruit tobacco users, and participants who were tobacco users were not necessarily interested in quitting. This may partially explain discrepant findings.
Appropriate populations. NAC has been studied mostly in adult cigarette smokers.
Safety and dosing. Suggested dosages for treating tobacco use disorder range from 1,200 to 3,600 mg/d (600 to 1,800 mg twice daily).
Continue to: Clinical implications
Clinical implications. Data on NAC’s efficacy for tobacco use disorder come from small, pilot trials. Although initial evidence is promising, it is premature to suggest NAC for smoking cessation until a fully powered, randomized clinical trial provides evidence of efficacy.
Alcohol use disorder
Alcohol use disorders are widely prevalent; 13.9% of U.S. adults met criteria in the past year, and 29.1% of U.S. adults meet criteria in their lifetime.38 Alcohol use disorders can result in significant negative consequences, including relationship problems, violent behavior, medical problems, and death. Existing FDA-approved medications for alcohol use disorder include naltrexone, acamprosate, and disulfiram.
Due to the severe potential health consequences of alcohol, NAC has been examined as a possible aid in preventing relapse. However, most studies have been conducted using animals. Three studies have examined alcohol use in humans (Table 536,39,40). One was a pilot study,39 and the other 2 were secondary data analyses.36,40 None of them specifically focused on alcohol use disorders. A pilot study of 35 veterans with co-occurring posttraumautic stress disorder (PTSD) and SUDs (82% of whom had an alcohol use disorder) found that compared with placebo, NAC significantly decreased PTSD symptoms, craving, and depression.39 In a study of 75 adults with bipolar disorder, secondary alcohol use was not significantly reduced.36 However, one study suggested that NAC may decrease adolescent alcohol and marijuana co-use.40 Future work is needed to examine the potential clinical utility of NAC in individuals with alcohol use disorders.

Findings from animal studies indicate that NAC may:
- reduce alcohol-seeking41
- reduce withdrawal symptoms42
- reduce the teratogenic effects of alcohol43
- prevent alcohol toxicity44
- reduce health-related consequences of alcohol (eg, myocardial oxidative stress45 and alcohol-related steatohepatitis46).
Continue to: Appropriate populations
Appropriate populations. Pilot studies have suggested that appropriate populations may include veterans with SUD and PTSD39 and adolescents with marijuana dependence who use alcohol.40
Safety and dosing. Suggested dosages for the treatment of alcohol use disorder based on these studies range from 1,000 to 2,400 mg/d (500 to 1,200 mg twice daily).
Clinical implications. Future work is needed to determine if NAC is effective for treating alcohol use disorders. Ongoing randomized clinical trials are examining the efficacy of NAC in reducing alcohol use among individuals with alcohol use disorder. It is premature to recommend NAC for treatment of alcohol use disorders.
Other psychiatric uses
Although we have highlighted NAC’s effect on glutamatergic transmission, evidence suggests that NAC may have multiple mechanisms of action that could impact psychiatric functioning. For example, NAC may also reverse oxidative stress, which is frequently observed in psychiatric disorders such as schizophrenia and bipolar disorder.10,12 NAC also has anti-inflammatory properties. When inflammatory pathways of the CNS are dysregulated, production of neurotransmitters may be impaired, resulting in depression-like symptoms.10,12,47 Preliminary evidence suggests that NAC may be effective in treating mood-related symptoms (eg, irritability, depression) in individuals with psychiatric disorders (eg, bipolar and depressive disorders, PTSD, and SUDs) and general symptoms of schizophrenia, obsessive-compulsive disorder, and trichotillomania, although mixed findings in controlled studies suggest a need for further research.12,39
Continue to: NAC: A promising candidate
NAC: A promising candidate
Initial evidence suggests NAC may be helpful for treating patients with SUDs. A patient seeking SUD treatment who is treated with NAC may experience a decreased drive, craving, or compulsion to use. Notably, NAC may be particularly useful in preventing relapse after an individual has achieved abstinence. Evidence suggests that NAC may be useful in the treatment of adults with cocaine use disorders who have achieved abstinence, and adolescents with cannabis use disorders. Preliminary results for adult tobacco use disorder are also promising. Human data examining the efficacy of NAC for alcohol use disorder is limited. Researchers’ ongoing challenge is to identify which patients with which SUDs are most likely to benefit from NAC, and to create clear clinical guidelines for the provider.
Bottom Line
N-acetylcysteine is likely to have modest effects for some patients who have a substance use disorder, particularly adults who use cocaine and adolescents who use marijuana. It may be useful in preventing relapse to substance use after an individual has achieved abstinence.
Related Resources
- Deepmala, Slattery J, Kumar N, et al. Clinical trials of N-acetylcysteine in psychiatry and neurology: a systematic review. Neurosci Biobehav Rev. 2015;55:294-321.
- Roberts-Wolfe D, Kalivas P. Glutamate transporter GLT-1 as a therapeutic target for substance use disorders. CNS Neurol Disord Drug Targets. 2015;14(6):745-756.
- National Institute on Drug Abuse. Treatment approaches for drug addiction. https://www.drugabuse.gov/publications/drugfacts/treatment-approaches-drug-addiction.
Drug Brand Names
Acamprosate • Campral
Acetaminophen • Tylenol
Baclofen • Lioresal
Bupropion • Zyban
Disulfiram • Antabuse
Naltrexone • Revia,Vivitrol
Varenicline • Chantix
1. Grella CE, Karno MP, Warda US, et al. Perceptions of need and help received for substance dependence in a national probability survey. Psychiatr Serv. 2009;60(8):1068-1074.
2. Everitt BJ, Robbins TW. Drug addiction: updating actions to habits to compulsions ten years on. Annu Rev Psychol. 2016;67:23-50.
3. McFarland K, Lapish CC, Kalivas PW. Prefrontal glutamate release into the core of the nucleus accumbens mediates cocaine-induced reinstatement of drug-seeking behavior. J Neurosci. 2003;23(8):3531-3537.
4. LaLumiere RT, Kalivas PW. Glutamate release in the nucleus accumbens core is necessary for heroin seeking. J Neurosci. 2008;28(12):3170-3177.
5. Kalivas PW, Volkow ND. New medications for drug addiction hiding in glutamatergic neuroplasticity. Mol Psychiatry. 2011;16(10):974-986.
6. Roberts-Wolfe D, Kalivas PW. Glutamate transporter GLT-1 as a therapeutic target for substance use disorders. CNS Neurol Disord Drug Targets. 2015;14(6):745-756.
7. Berk M, Malhi GS, Gray LJ, et al. The promise of N-acetylcysteine in neuropsychiatry. Trends Pharmacol Sci. 2013;34(3):167-177.
8. McClure EA, Gipson CD, Malcolm RJ, et al. Potential role of N-acetylcysteine in the management of substance use disorders. CNS drugs. 2014;28(2):95-106.
9. Deepmala, Slattery J, Kumar N, et al. Clinical trials of N-acetylcysteine in psychiatry and neurology: a systematic review. Neurosci Biobehav Rev. 2015;55:294-321.
10. Minarini A, Ferrari S, Galletti M, et al. N-acetylcysteine in the treatment of psychiatric disorders: current status and future prospects. Expert Opin Drug Metab Toxicol. 2017;13(3):279-292.
11. Grandjean EM, Berthet P, Ruffman R, et al. Efficacy of oral long-term N‑acetylcysteine in chronic bronchopulmonary disease: a meta-analysis of published double-blind, placebo-controlled clinical trials. Clin Ther. 2000;22(2):209‑221.
12. Rhodes K, Braakhuis A. Performance and side effects of supplementation with N-acetylcysteine: a systematic review and meta-analysis. Sports Med. 2017;47(8):1619-1636.
13. Olsson B, Johansson M, Gabrielsson J, et al. Pharmacokinetics and bioavailability of reduced and oxidized N-acetylcysteine. Eur J Clin Pharmacol. 1988;34(1):77-82.
14. United Nations Office on Drugs and Crime. World Drug Report 2016 (United Nations publication, Sales No. E.16.XI.7). https://www.unodc.org/doc/wdr2016/WORLD_DRUG_REPORT_2016_web.pdf. Published May 2016. Accessed April 26, 2018.
15. Amen SL, Piacentine LB, Ahmad ME, et al. Repeated N-acetyl cysteine reduces cocaine seeking in rodents and craving in cocaine-dependent humans. Neuropsychopharmacology. 2011;36(4):871-878.
16. Mardikian PN, LaRowe SD, Hedden S, et al. An open-label trial of N-acetylcysteine for the treatment of cocaine dependence: a pilot study. Prog Neuropsychopharmacol Biol Psychiatry. 2007;31(2):389-394.
17. LaRowe SD, Kalivas PW, Nicholas JS, et al. A double‐blind placebo‐controlled trial of N‐acetylcysteine in the treatment of cocaine dependence. Am J Addict. 2013;22(5):443-452.
18. Mousavi SG, Sharbafchi MR, Salehi M, et al. The efficacy of N-acetylcysteine in the treatment of methamphetamine dependence: a double-blind controlled, crossover study. Arch Iran Med. 2015;18(1):28-33.
19. Grant JE, Odlaug BL, Kim SW. A double-blind, placebo-controlled study of N-acetyl cysteine plus naltrexone for methamphetamine dependence. Eur Neuropsychopharmacol. 2010;20(11):823-828.
20. Lopez-Quintero C, Pérez de los Cobos J, Hasin DS, et al. Probability and predictors of transition from first use to dependence on nicotine, alcohol, cannabis, and cocaine: results of the National Epidemiologic Survey on Alcohol and Related Conditions (NESARC). Drug Alcohol Depend. 2011;115(1-2):120-130.
21. Chen CY, O’Brien MS, Anthony JC. Who becomes cannabis dependent soon after onset of use? Epidemiological evidence from the United States: 2000-2001. Drug Alcohol Depend. 2005;79(1):11-22.
22. Copersino ML, Boyd SJ, Tashkin DP, et al. Quitting among non-treatment-seeking marijuana users: reasons and changes in other substance use. Am J Addict. 2006;15(4):297-302.
23. Weiner MD, Sussman S, McCuller WJ, et al. Factors in marijuana cessation among high-risk youth. J Drug Educ. 1999;29(4):337-357.
24. Gray KM, Watson NL, Carpenter MJ, et al. N-acetylcysteine (NAC) in young marijuana users: an open-label pilot study. Am J Addict. 2010;19(2):187-189.
25. Gray KM, Carpenter MJ, Baker NL, et al. A double-blind randomized controlled trial of N-acetylcysteine in cannabis-dependent adolescents. Am J Psychiatry. 2012;169(8):805-812.
26. Roten AT, Baker NL, Gray KM. Marijuana craving trajectories in an adolescent marijuana cessation pharmacotherapy trial. Addict Behav. 2013;38(3):1788-1791.
27. Gray KM, Sonne SC, McClure EA, et al. A randomized placebo-controlled trial of N-acetylcysteine for cannabis use disorder in adults. Drug Alcohol Depend. 2017;177:249-257.
28. Rostron B. Mortality risks associated with environmental tobacco smoke exposure in the United States. Nicotine Tob Res. 2013;15(10):1722-1728.
29. Centers for Disease Control and Prevention. Quitting smoking among adults – United States, 2001–2010. MMWR. 2011;60(44):1513-1519.
30. McClure EA, Baker NL, Gipson CD, et al. An open-label pilot trial of N-acetylcysteine and varenicline in adult cigarette smokers. Am J Drug Alcohol Abuse. 2015;41(1):52-56.
31. Froeliger B, McConnell P, Stankeviciute N, et al. The effects of N-acetylcysteine on frontostriatal resting-state functional connectivity, withdrawal symptoms and smoking abstinence: a double-blind, placebo-controlled fMRI pilot study. Drug Alcohol Depend. 2015;156:234-242.
32. Prado E, Maes M, Piccoli LG, et al. N-acetylcysteine for therapy-resistant tobacco use disorder: a pilot study. Redox Rep. 2015;20(5):215-222.
33. Knackstedt LA, LaRowe S, Mardikian P, et al. The role of cystine-glutamate exchange in nicotine dependence in rats and humans. Biol Psychiatry. 2009;65(10):841-845.
34. Grant JE, Odlaug BL, Chamberlain SR, et al. A randomized, placebo-controlled trial of N-acetylcysteine plus imaginal desensitization for nicotine-dependent pathological gamblers. J Clin Psychiatry. 2014;75(1):39-45.
35. Schmaal L, Berk L, Hulstijn KP, et al. Efficacy of N-acetylcysteine in the treatment of nicotine dependence: a double-blind placebo-controlled pilot study. Eur Addiction Res. 2011;17(4):211-216.
36. Bernardo M, Dodd S, Gama CS, et al. Effects of N‐acetylcysteine on substance use in bipolar disorder: a randomised placebo‐controlled clinical trial. Acta Neuropsychiatr. 2009;21(5):239-245.
37. McClure EA, Baker NL, Gray KM. Cigarette smoking during an N-acetylcysteine-assisted cannabis cessation trial in adolescents. Am J Drug Alcohol Abuse. 2014;40(4):285-291.
38. Grant BF, Goldstein RB, Saha TD, et al. Epidemiology of DSM-5 alcohol use disorder: Results from the National Epidemiologic Survey on Alcohol and Related Conditions III. JAMA Psychiatry. 2015;72(8):757-766.
39. Back SE, McCauley JL, Korte KJ, et al. A double-blind randomized controlled pilot trial of N-acetylcysteine in veterans with PTSD and substance use disorders. J Clin Psychiatry. 2016;77(11):e1439-e1446.
40. Squeglia LM, Baker NL, McClure EA, et al. Alcohol use during a trial of N-acetylcysteine for adolescent marijuana cessation. Addict Behav. 2016;63:172-177.
41. Lebourgeois S, González-Marín MC, Jeanblanc J, et al. Effect of N-acetylcysteine on motivation, seeking and relapse to ethanol self-administration. Addict Biol. 2018;23(2):643-652.
42. Schneider R Jr, Santos CF, Clarimundo V, et al. N-acetylcysteine prevents behavioral and biochemical changes induced by alcohol cessation in rats. Alcohol. 2015;49(3):259-263.
43. Parnell SE, Sulik KK, Dehart DB, et al. Reduction of ethanol-induced ocular abnormalities in mice via dietary administration of N-acetylcysteine. Alcohol. 2010;44(7-8):699-705.
44. Ozkol H, Bulut G, Balahoroglu R, et al. Protective effects of Selenium, N-acetylcysteine and Vitamin E against acute ethanol intoxication in rats. Biol Trace Elem Res. 2017;175(1):177-185.
45. Seiva FR, Amauchi JF, Rocha KK, et al. Alcoholism and alcohol abstinence: N-acetylcysteine to improve energy expenditure, myocardial oxidative stress, and energy metabolism in alcoholic heart disease. Alcohol. 2009;43(8):649-656.
46. Setshedi M, Longato L, Petersen DR, et al. Limited therapeutic effect of N‐acetylcysteine on hepatic insulin resistance in an experimental model of alcohol‐induced steatohepatitis. Alcohol Clin Exp Res. 2011;35(12):2139-2151.
47. Miller AH, Maletic V, Raison CL. Inflammation and its discontents: the role of cytokines in the pathophysiology of major depression. Biol Psychiatry. 2009;65(9):732-741.
1. Grella CE, Karno MP, Warda US, et al. Perceptions of need and help received for substance dependence in a national probability survey. Psychiatr Serv. 2009;60(8):1068-1074.
2. Everitt BJ, Robbins TW. Drug addiction: updating actions to habits to compulsions ten years on. Annu Rev Psychol. 2016;67:23-50.
3. McFarland K, Lapish CC, Kalivas PW. Prefrontal glutamate release into the core of the nucleus accumbens mediates cocaine-induced reinstatement of drug-seeking behavior. J Neurosci. 2003;23(8):3531-3537.
4. LaLumiere RT, Kalivas PW. Glutamate release in the nucleus accumbens core is necessary for heroin seeking. J Neurosci. 2008;28(12):3170-3177.
5. Kalivas PW, Volkow ND. New medications for drug addiction hiding in glutamatergic neuroplasticity. Mol Psychiatry. 2011;16(10):974-986.
6. Roberts-Wolfe D, Kalivas PW. Glutamate transporter GLT-1 as a therapeutic target for substance use disorders. CNS Neurol Disord Drug Targets. 2015;14(6):745-756.
7. Berk M, Malhi GS, Gray LJ, et al. The promise of N-acetylcysteine in neuropsychiatry. Trends Pharmacol Sci. 2013;34(3):167-177.
8. McClure EA, Gipson CD, Malcolm RJ, et al. Potential role of N-acetylcysteine in the management of substance use disorders. CNS drugs. 2014;28(2):95-106.
9. Deepmala, Slattery J, Kumar N, et al. Clinical trials of N-acetylcysteine in psychiatry and neurology: a systematic review. Neurosci Biobehav Rev. 2015;55:294-321.
10. Minarini A, Ferrari S, Galletti M, et al. N-acetylcysteine in the treatment of psychiatric disorders: current status and future prospects. Expert Opin Drug Metab Toxicol. 2017;13(3):279-292.
11. Grandjean EM, Berthet P, Ruffman R, et al. Efficacy of oral long-term N‑acetylcysteine in chronic bronchopulmonary disease: a meta-analysis of published double-blind, placebo-controlled clinical trials. Clin Ther. 2000;22(2):209‑221.
12. Rhodes K, Braakhuis A. Performance and side effects of supplementation with N-acetylcysteine: a systematic review and meta-analysis. Sports Med. 2017;47(8):1619-1636.
13. Olsson B, Johansson M, Gabrielsson J, et al. Pharmacokinetics and bioavailability of reduced and oxidized N-acetylcysteine. Eur J Clin Pharmacol. 1988;34(1):77-82.
14. United Nations Office on Drugs and Crime. World Drug Report 2016 (United Nations publication, Sales No. E.16.XI.7). https://www.unodc.org/doc/wdr2016/WORLD_DRUG_REPORT_2016_web.pdf. Published May 2016. Accessed April 26, 2018.
15. Amen SL, Piacentine LB, Ahmad ME, et al. Repeated N-acetyl cysteine reduces cocaine seeking in rodents and craving in cocaine-dependent humans. Neuropsychopharmacology. 2011;36(4):871-878.
16. Mardikian PN, LaRowe SD, Hedden S, et al. An open-label trial of N-acetylcysteine for the treatment of cocaine dependence: a pilot study. Prog Neuropsychopharmacol Biol Psychiatry. 2007;31(2):389-394.
17. LaRowe SD, Kalivas PW, Nicholas JS, et al. A double‐blind placebo‐controlled trial of N‐acetylcysteine in the treatment of cocaine dependence. Am J Addict. 2013;22(5):443-452.
18. Mousavi SG, Sharbafchi MR, Salehi M, et al. The efficacy of N-acetylcysteine in the treatment of methamphetamine dependence: a double-blind controlled, crossover study. Arch Iran Med. 2015;18(1):28-33.
19. Grant JE, Odlaug BL, Kim SW. A double-blind, placebo-controlled study of N-acetyl cysteine plus naltrexone for methamphetamine dependence. Eur Neuropsychopharmacol. 2010;20(11):823-828.
20. Lopez-Quintero C, Pérez de los Cobos J, Hasin DS, et al. Probability and predictors of transition from first use to dependence on nicotine, alcohol, cannabis, and cocaine: results of the National Epidemiologic Survey on Alcohol and Related Conditions (NESARC). Drug Alcohol Depend. 2011;115(1-2):120-130.
21. Chen CY, O’Brien MS, Anthony JC. Who becomes cannabis dependent soon after onset of use? Epidemiological evidence from the United States: 2000-2001. Drug Alcohol Depend. 2005;79(1):11-22.
22. Copersino ML, Boyd SJ, Tashkin DP, et al. Quitting among non-treatment-seeking marijuana users: reasons and changes in other substance use. Am J Addict. 2006;15(4):297-302.
23. Weiner MD, Sussman S, McCuller WJ, et al. Factors in marijuana cessation among high-risk youth. J Drug Educ. 1999;29(4):337-357.
24. Gray KM, Watson NL, Carpenter MJ, et al. N-acetylcysteine (NAC) in young marijuana users: an open-label pilot study. Am J Addict. 2010;19(2):187-189.
25. Gray KM, Carpenter MJ, Baker NL, et al. A double-blind randomized controlled trial of N-acetylcysteine in cannabis-dependent adolescents. Am J Psychiatry. 2012;169(8):805-812.
26. Roten AT, Baker NL, Gray KM. Marijuana craving trajectories in an adolescent marijuana cessation pharmacotherapy trial. Addict Behav. 2013;38(3):1788-1791.
27. Gray KM, Sonne SC, McClure EA, et al. A randomized placebo-controlled trial of N-acetylcysteine for cannabis use disorder in adults. Drug Alcohol Depend. 2017;177:249-257.
28. Rostron B. Mortality risks associated with environmental tobacco smoke exposure in the United States. Nicotine Tob Res. 2013;15(10):1722-1728.
29. Centers for Disease Control and Prevention. Quitting smoking among adults – United States, 2001–2010. MMWR. 2011;60(44):1513-1519.
30. McClure EA, Baker NL, Gipson CD, et al. An open-label pilot trial of N-acetylcysteine and varenicline in adult cigarette smokers. Am J Drug Alcohol Abuse. 2015;41(1):52-56.
31. Froeliger B, McConnell P, Stankeviciute N, et al. The effects of N-acetylcysteine on frontostriatal resting-state functional connectivity, withdrawal symptoms and smoking abstinence: a double-blind, placebo-controlled fMRI pilot study. Drug Alcohol Depend. 2015;156:234-242.
32. Prado E, Maes M, Piccoli LG, et al. N-acetylcysteine for therapy-resistant tobacco use disorder: a pilot study. Redox Rep. 2015;20(5):215-222.
33. Knackstedt LA, LaRowe S, Mardikian P, et al. The role of cystine-glutamate exchange in nicotine dependence in rats and humans. Biol Psychiatry. 2009;65(10):841-845.
34. Grant JE, Odlaug BL, Chamberlain SR, et al. A randomized, placebo-controlled trial of N-acetylcysteine plus imaginal desensitization for nicotine-dependent pathological gamblers. J Clin Psychiatry. 2014;75(1):39-45.
35. Schmaal L, Berk L, Hulstijn KP, et al. Efficacy of N-acetylcysteine in the treatment of nicotine dependence: a double-blind placebo-controlled pilot study. Eur Addiction Res. 2011;17(4):211-216.
36. Bernardo M, Dodd S, Gama CS, et al. Effects of N‐acetylcysteine on substance use in bipolar disorder: a randomised placebo‐controlled clinical trial. Acta Neuropsychiatr. 2009;21(5):239-245.
37. McClure EA, Baker NL, Gray KM. Cigarette smoking during an N-acetylcysteine-assisted cannabis cessation trial in adolescents. Am J Drug Alcohol Abuse. 2014;40(4):285-291.
38. Grant BF, Goldstein RB, Saha TD, et al. Epidemiology of DSM-5 alcohol use disorder: Results from the National Epidemiologic Survey on Alcohol and Related Conditions III. JAMA Psychiatry. 2015;72(8):757-766.
39. Back SE, McCauley JL, Korte KJ, et al. A double-blind randomized controlled pilot trial of N-acetylcysteine in veterans with PTSD and substance use disorders. J Clin Psychiatry. 2016;77(11):e1439-e1446.
40. Squeglia LM, Baker NL, McClure EA, et al. Alcohol use during a trial of N-acetylcysteine for adolescent marijuana cessation. Addict Behav. 2016;63:172-177.
41. Lebourgeois S, González-Marín MC, Jeanblanc J, et al. Effect of N-acetylcysteine on motivation, seeking and relapse to ethanol self-administration. Addict Biol. 2018;23(2):643-652.
42. Schneider R Jr, Santos CF, Clarimundo V, et al. N-acetylcysteine prevents behavioral and biochemical changes induced by alcohol cessation in rats. Alcohol. 2015;49(3):259-263.
43. Parnell SE, Sulik KK, Dehart DB, et al. Reduction of ethanol-induced ocular abnormalities in mice via dietary administration of N-acetylcysteine. Alcohol. 2010;44(7-8):699-705.
44. Ozkol H, Bulut G, Balahoroglu R, et al. Protective effects of Selenium, N-acetylcysteine and Vitamin E against acute ethanol intoxication in rats. Biol Trace Elem Res. 2017;175(1):177-185.
45. Seiva FR, Amauchi JF, Rocha KK, et al. Alcoholism and alcohol abstinence: N-acetylcysteine to improve energy expenditure, myocardial oxidative stress, and energy metabolism in alcoholic heart disease. Alcohol. 2009;43(8):649-656.
46. Setshedi M, Longato L, Petersen DR, et al. Limited therapeutic effect of N‐acetylcysteine on hepatic insulin resistance in an experimental model of alcohol‐induced steatohepatitis. Alcohol Clin Exp Res. 2011;35(12):2139-2151.
47. Miller AH, Maletic V, Raison CL. Inflammation and its discontents: the role of cytokines in the pathophysiology of major depression. Biol Psychiatry. 2009;65(9):732-741.
Alcohol dependence in women: Comorbidities can complicate treatment
Ms. F, a 53-year-old high school English teacher, is referred to you by her family physician after she was suspended from work for suspected intoxication. She was divorced 2 years ago from her husband of 20 years, and she says her drinking has escalated to 2 bottles of wine every night. She wants to reduce her alcohol use but experiences shakiness, nausea, and diaphoresis when she tries to cut back.
Ms. F began drinking at age 16 “to feel more comfortable in social situations” and has experienced binge drinking with intermittent blackouts. She denies illicit drug use and legal consequences from drinking. Her father died from cirrhosis at age 58. Her mother suffers from depression but is in remission with medication.
Ms. F is hesitant to date or establish intimate relationships. She has stopped attending church, a book club, and her 15-year-old daughter’s booster club activities.
For years, little was known about alcohol use and alcohol-related problems in women such as Ms. F.1 Alcohol dependence studies rarely included women, so findings and treatment outcomes observed in men were assumed to apply to both genders.
Awareness of gender differences in addiction has grown (Box 1).2-5 Biological and psychosocial differences between alcohol-dependent women and men now are understood to influence etiology, epidemiology, psychiatric and medical comorbidity, course of illness, and treatment outcomes. This article discusses recent insights into planning treatment to address specific needs of alcohol-dependent women.
Epidemiologic surveys consistently show higher rates of alcohol use disorders in men than women, but recent data suggest a narrowing of the gender gap. Studies from the National Institute on Alcohol Abuse and Alcoholism (NIAAA) from 1991 to 1992 and 2001 to 2002, found:
- a significant increase in the 12-month prevalence of alcohol abuse among men (4.7% to 6.9%) and women (1.5% to 2.6%).
- a significant decrease in alcohol dependence among men (6.3% to 5.4%) but not women (from 2.6% to 2.3%).
- a significant increase in rate of alcohol dependence among African-American women age 18 to 29 (from 2.1% to 3.8%).
- the gender differential in alcohol dependence narrowed from 3.8% to 3.1%.2,3
Similarly, studies conducted 60 years ago showed that boys were more likely than girls to report first alcohol use between ages 10 and 14 (4:1 ratio). Now the age of first alcohol use is relatively equivalent in boys and girls.3 This convergence is disconcerting and suggests that screening and prevention initiatives for adolescents need to increase their focus on girls.
Overall, these observations support the ‘convergence hypothesis,’ which holds that the gap between men’s and women’s alcohol consumption has narrowed as women’s use of alcohol has increased.4,5
Accelerated consequences
As a group, women may consume less alcohol than men but progress more rapidly to alcohol-related illnesses and negative consequences. Specifically, alcohol-dependent women develop liver disease, hypertension, and gastrointestinal hemorrhage more rapidly than alcohol-dependent men.3 Cognitive deficits and brain atrophy also develop sooner in alcohol-dependent women than men.6 Causes of this accelerated progression—”telescoping”—include gender-specific biological differences:
- Women have lower levels of gastric alcohol dehydrogenase—the enzyme that initiates alcohol metabolism—and therefore experience a higher blood alcohol concentration than men drinking the same amount of alcohol.
- Women have less total body water and less capacity to dilute alcohol than men.
‘At-risk’ drinking. Gender-related physiologic differences have led to different thresholds for defining “at-risk drinking” and binge drinking for men and women (Table 1).7 Alcohol use also has negative effects on women’s reproductive system and menstrual cycle. Women with alcohol dependence have higher rates of sexual dysfunction, irregular menstrual cycles, early menopause, and amenorrhea as compared with nonalcohol-dependent women.8
Table 1
Different thresholds: Number of drinks* that increases risk for alcohol-related problems
| Drinks/week | Drinks/day | |
|---|---|---|
| Men | >14 | >4 |
| Women | >7 | >3 |
| *A standard drink is 12 ounces of beer, 5 ounces of wine, or 1.5 ounces of spirits | ||
| Source: Reference 7 | ||
Motives for drinking
Research suggests different motives for alcohol use in girls and women vs boys and men. Women appear more likely to use alcohol to “self-medicate” negative affect or emotional pain (Box 2).9-12 Men are more likely to use alcohol to enhance pleasurable emotional states, “feel high” or because of social pressures to conform.5
Drinking partners. Compared with men’s motives for alcohol use, women’s drinking motives are more strongly influenced by their spouse’s or partner’s drinking patterns. In a large twin study (N = 5,974), 13% of alcohol-dependent women vs 3% of alcohol-dependent men reported that their spouse had a history of alcohol problems.9 Thus, consider the addiction status and drinking patterns of a woman’s partner when developing a treatment plan and relapse prevention strategies.
Trauma and alcohol use. Converging lines of evidence suggest that a relationship among trauma, posttraumatic stress disorder, and substance use disorders is particularly important for women. Early life stress—particularly sexual abuse—is more common in girls than in boys and is associated with the risk of developing substance use disorders.10
Women in alcohol treatment programs report more moderate (87%) and severe (40%) intimate partner violence than women in community samples (28% and 8%, respectively), as well as higher rates of childhood physical abuse (37% vs 11%, respectively).11 Moreover, alcohol abuse places women at risk for repeated victimization and serves to perpetuate a cycle of victimization and addiction.12 These are critical issues to assess and address when treating women with alcohol dependence.
In a study of >1,200 adolescents, girls age 13 With age, drinking for social and enhancement motives increases more among male than female adolescents. Drinking to reduce or manage negative affect is associated with an increased risk of heavy drinking (particularly for adolescent girls), more frequent intoxication, and alcohol dependence.14 These findings suggest:
- Young women with alcohol use problems may benefit from learning strategies to regulate negative emotions and alleviate depression or anxiety symptoms that may be contributing to their drinking.
- Young men with alcohol use problems may benefit from learning to manage peer pressure and from finding alternate sources of pleasure enhancement.5
CASE CONTINUED: Depressed and anxious
Ms. F admits to depressed mood, crying episodes, isolation from others, anhedonia, feelings of guilt, low motivation, difficulty concentrating at work, restless sleep, and weight loss. These symptoms recurred soon after she and her husband separated 4 years ago. She denies suicidal thoughts or suicide attempts.
She suffered similar episodes in the past, including when she was breastfeeding and abstinent from alcohol after the birth of her daughter. Her obstetrician treated her depression with fluoxetine. Problems with anxiety began in high school, especially associated with parties and dating. In college, she often would drink beer before class presentations.
You diagnose alcohol dependence, major depressive disorder, and social phobia. You also ask Ms. F about a history of trauma. She reports that her father was “real harsh” when he was drinking and often hit her and her sister. She also relates being struck by her ex-husband during arguments. Screening with the Clinician-Administered PTSD Scale is negative for posttraumatic stress disorder (PTSD), however.
Comorbid psychiatric disorders
Gender differences in reasons for alcohol use may be related to women’s higher rate of medical and psychiatric comorbidity (Table 2).6,8,15,16 Vital signs, physical examination, and lab work are helpful for diagnosis and monitoring of medical comorbidities and complications. The gamma-glutamyl transferase (GGT) test for liver disease and carbohydrate deficient transferrin (CDT) test for chronic heavy alcohol consumption are less sensitive and specific in women than in men.17 Even so, monitoring GGT and CDT results over time may serve as a valuable marker of continued drinking by women.
Mood and anxiety disorders are significantly more common in women than in men among individuals with alcohol use disorders.15,16 This pattern is not unique to alcohol-dependent persons, however. In the general population:
- women are more likely than men to meet diagnostic criteria for anxiety, depression, bulimia nervosa, and borderline personality disorder
- men are more likely than women to meet diagnostic criteria for antisocial personality disorder.18
Comorbid disorders in women with alcohol dependence
| Psychiatric Posttraumatic stress disorder Other anxiety disorders (such as panic disorder without agoraphobia, simple phobia, or social phobia) Major depressive disorder Cognitive impairment |
| Medical Hypertension Fatty liver disease Gastrointestinal hemorrhage Brain atrophy Reproductive system irregularities |
| Source: References 6,8,15,16 |
Suicide risk. A recent study by Connor et al20 examined suicidal ideation, planning, and attempts in 3,729 alcohol dependent subjects (35% female) and found an association between female gender and both planned (odds ratio [OR] = 3.4) and unplanned suicide attempts (OR = 3.8). Further research is needed to clarify the relationship between female gender, alcohol dependence, and suicide risk, behaviors, and attempts.
Anxiety disorders are the most common psychiatric disorders in women,21 and social anxiety and social phobia may play a predisposing role in alcohol dependence.22 Individuals with social anxiety may self-medicate with alcohol as a social lubricant. Some research suggests that anxiety disorders are more severe in alcohol-dependent women than in men with similarly severe alcohol dependence.22
23
Therefore, when assessing and treating alcohol-dependent women, screen for trauma history as well as mood and anxiety disorders. To optimize outcomes, treat these disorders simultaneously with the alcohol use disorder.24
Treatment planning
Underuse of treatment programs. Women with alcohol dependence are more likely to seek treatment in primary care or mental health settings, rather than in alcohol treatment settings.25,26 Women’s underuse of alcohol treatment programs is likely related to:
- greater stigma associated with alcohol use for women as compared with men
- socioeconomic factors, including pregnancy, child care, and concerns about child custody issues.25
Gender-specific treatment? Women-only treatment programs have been studied because of observed differences in men’s and women’s interaction styles and the hypothesis that men’s traditional societal dominance could negatively affect women in mixed-gender groups.25 Better treatment outcomes have been hypothesized if treatment is tailored to address women’s unique issues: risk factors for alcohol dependence, course of disease progression, medical problems associated with alcohol dependence, and reasons for relapse.
Gender-specific treatment may provide an environment where women—particularly those with a history of trauma from a male perpetrator—feel safe discussing issues related to their alcohol problems. For practical purposes, these programs also may be more likely to address women’s needs for on-site child care, prenatal care, and mental health programming.
A recent study compared a manual-based 12-session women’s recovery group with mixed-gender, manualized group drug counseling (GDC). The women’s recovery group focused on gender-specific topics such as relationships, the caregiver role, trauma, comorbid psychiatric conditions (including eating, mood, and anxiety disorders), and the effects of drug and alcohol use on women’s health. The women’s recovery group was:
- as effective as mixed-gender GDC in reducing substance use during the 12-week treatment
- significantly more effective during the 6-month post-treatment phase.28
CASE CONTINUED: Developing a treatment plan
Ms. F identifies 3 triggers for her alcohol use: a stressful day at school, arguments with her ex-husband, and feeling lonely. Because these are high-risk situations for relapse, you incorporate strategies to deal with them into her treatment plan. Other factors to consider:
- whether she requires detoxification
- an FDA-approved medication for alcohol dependence (acamprosate, oral or injectable naltrexone, or disulfiram)
- cognitive-behavioral therapy and medication for major depression and social phobia
- referral to psychosocial support groups (such as Alcoholics Anonymous).
Three factors determine the need for detoxification: the course of previous alcohol withdrawals (alcoholic hallucinosis, seizures, or delirium tremens), elevated vital signs or other evidence of autonomic hyperactivity such as diaphoresis or tremors, and the patient’s general medical condition. During early recovery, monitor patients closely to assess mood and anxiety symptoms. Blood alcohol tests or GGT and CDT are useful to monitor self-reported abstinence.
Related resources
- National Clearing House for Alcohol and Drug Information. Publications and materials on women and substance abuse. Substance Abuse and Mental Health Services Administration. http://ncadistore.samhsa.gov/catalogresults.aspx?h=drugs&topic=11.
- National Institute on Drug Abuse (www.nida.nih.gov). Articles that address women’s health and gender differences. www.drugabuse.gov/NIDA_Notes/NN0013.html.
- Acamprosate • Campral
- Disulfiram • Antabuse
- Fluoxetine • Prozac
- Naltrexone • Vivitrol, ReVia
Drs. Payne, Back, Wright, and Hartwell report no financial relationship with any company whose products are mentioned in this article or with manufacturers of competing products.
Dr. Brady receives research support from Abbott Laboratories, GlaxoSmithKline, Forest Pharmaceuticals, and Wyeth. She is a consultant and speaker for Pfizer Inc., Eli Lilly and Company, Abbott Laboratories, GlaxoSmithKline, and Forest Pharmaceuticals.
1. Brady KT, Back SE, Greenfield S, eds. Women and addiction: a comprehensive handbook. New York, NY: Guilford Press; 2009.
2. Grant BF, Dawson DA, Stinson FS, et al. The 12-month prevalence and trends in DSM-IV alcohol abuse and dependence: United States, 1991-1992 and 2001-2002. Drug Alcohol Depend. 2004;74(3):223-234.
3. Greenfield SH. Women and alcohol use disorders. Harv Rev Psychiatry. 2002;10(2):76-85.
4. Greenfield TK, Room R. Situational norms for drinking and drunkenness: trends in the US adult population, 1979-1990. Addiction. 1997;92:33-47.
5. Stewart SH, Gavric D, Collins P. Women, girls, and alcohol. In: Brady KT, Back SE, Greenfield S, eds. Women and addiction: a comprehensive handbook. New York, NY: Guilford Press; 2009.
6. Mann K, Ackermann K, Croissant B, et al. Neuroimaging of gender differences in alcohol dependence: are women more vulnerable? Alcohol Clin Exp Res. 2005;29(5):896-901.
7. National Institute of Alcohol Abuse and Alcoholism. Helping patients who drink too much: a clinician’s guide. Bethesda, MD: National Institute of Alcohol Abuse and Alcoholism; 2005. NIH Publication No. 07-3769.
8. Blume SB, Zilberman ML. Addiction in women. In: Galanter M, Kleber H, eds. Textbook of substance abuse treatment. 3rd ed. Washington, DC: American Psychiatric Publishing, Inc; 2004:539-546.
9. Grant JD, Heath AC, Bucholz KK, et al. Spousal concordance for alcohol dependence: evidence for assortative mating or spousal interaction effect? Alcohol Clin Exp Res. 2007;31(5):717-728.
10. Kendler KS, Bulik CM, Silberg J, et al. Childhood sexual abuse and adult psychiatric and substance use disorders in women: an epidemiological and co-twin control analysis. Arch Gen Psychiatry. 2000;57:953-959.
11. Miller BA, Wilsnack SC, Cunradi CB. Family violence and victimization: treatment issues for women with alcohol problems. Alcohol Clin Exp Res. 2000;24(8):1287-1297.
12. Kilpatrick DG, Resnick HS, Saunders BE, et al. Victimization, posttraumatic stress disorder, and substance use and abuse among women. In: Wetherington CL, Roman AB, eds. Drug addiction research and the health of women. Rockville, MD: U.S. Department of Health and Human Services; 1998. NIH 98-4290:285-307.
13. Cooper ML. Motivations for alcohol use among adolescents: development and validation of a four-factor model. Psychol Assess. 1994;6(2):117-128.
14. Prescott CA, Cross RJ, Kuhn JW, et al. Is risk for alcoholism mediated by individual differences in drinking motivations? Alcohol Clin Exp Res. 2004;28(1):29-39.
15. Brady KT, Grice DE, Dustan L, et al. Gender differences in substance use disorders. Am J Psychiatry. 1993;150:1707-1711.
16. Helzer JE, Pryzbeck TR. The co-occurrence of alcoholism with other psychiatric disorders in the general population and its impact on treatment. J Stud Alcohol. 1988;49(3):219-224.
17. Center for Substance Abuse Treatment. Detoxification and substance abuse treatment. Treatment Improvement Protocol (TIP) Series 45. Rockville, MD: Substance Abuse and Mental Health Services Administration, 2006. DHHS (SMA) 06-4131.
18. Sinha R, Rounsaville BJ. Sex differences in depressed substance abusers. J Clin Psychiatry. 2002;63(7):616-627.
19. Gilman SE, Abraham HD. A longitudinal study of the order of onset of alcohol dependence and major depression. Drug Alcohol Depend. 2001;63(3):277-286.
20. Connor KR, Hesselbrock VM, Meldrum SC, et al. Transitions to, and correlates of, suicidal ideations, plans, and unplanned and planned suicide attempts among 3,728 men and women with alcohol dependence. J Stud Alcohol Drugs. 2007;68:654-662.
21. Pigott TA. Anxiety disorders in women. Psychiatr Clin North Am. 2003;26(3):621-672.
22. Randall CL, Thomas SE, Thevos AK. Gender comparison in alcoholics with concurrent social phobia: implications for alcoholism treatment. Am J Addict. 2000;9(3):202-215.
23. Logan TK, Walker R, Cole J, et al. Victimization and substance abuse among women: contributing factors, interventions, and implications. Review of General Psychology. 2002;6:325-397.
24. Volkow N. Comorbidity: addiction and other mental illnesses. Rockville, MD: U.S. Department of Health and Human Services; 2009. NIH 08-5771.
25. Greenfield SF, Brooks AJ, Gordon SM, et al. Substance abuse treatment entry, retention, and outcome in women: a review of the literature. Drug Alcohol Depend. 2007;86(1):1-21.
26. Green CA. Gender and use of substance abuse treatment services. Alcohol Res Health. 2006;29(1):55-62.
27. Brady TM, Ashley OS. Women in substance abuse treatment: results from the alcohol and drug services study (ADSS). Rockville, MD: Substance Abuse and Mental Health Services Administration, Office of Applied Studies; 2005. DHHS Publication No. SMA 04-3968, Analytic Series A-26. Available at: http://www.oas.samhsa.gov/womenTX/womenTX.pdf. Accessed July 22, 2007.
28. Greenfield SF, Trucco EM, McHugh RK, et al. The Women’s Recovery Group Study: a stage I trial of women-focused group therapy for substance use disorders versus mixedgender group drug counseling. Drug Alcohol Depend. 2007;90:39-47.
29. Garbutt JC, Kranzler HR, O’Malley SS, et al. Efficacy and tolerability of long-acting injectable naltrexone for alcohol dependence: a randomized controlled trial. JAMA. 2005;293(13):1617-1625.
30. O’Malley SS, Sinha R, Grilo CM, et al. Naltrexone and cognitive behavioral coping skills therapy for the treatment of alcohol drinking and eating disorder features in alcoholdependent women: a randomized controlled trial. Alcohol Clin Exp Res. 2007;31(4):625-634.
Ms. F, a 53-year-old high school English teacher, is referred to you by her family physician after she was suspended from work for suspected intoxication. She was divorced 2 years ago from her husband of 20 years, and she says her drinking has escalated to 2 bottles of wine every night. She wants to reduce her alcohol use but experiences shakiness, nausea, and diaphoresis when she tries to cut back.
Ms. F began drinking at age 16 “to feel more comfortable in social situations” and has experienced binge drinking with intermittent blackouts. She denies illicit drug use and legal consequences from drinking. Her father died from cirrhosis at age 58. Her mother suffers from depression but is in remission with medication.
Ms. F is hesitant to date or establish intimate relationships. She has stopped attending church, a book club, and her 15-year-old daughter’s booster club activities.
For years, little was known about alcohol use and alcohol-related problems in women such as Ms. F.1 Alcohol dependence studies rarely included women, so findings and treatment outcomes observed in men were assumed to apply to both genders.
Awareness of gender differences in addiction has grown (Box 1).2-5 Biological and psychosocial differences between alcohol-dependent women and men now are understood to influence etiology, epidemiology, psychiatric and medical comorbidity, course of illness, and treatment outcomes. This article discusses recent insights into planning treatment to address specific needs of alcohol-dependent women.
Epidemiologic surveys consistently show higher rates of alcohol use disorders in men than women, but recent data suggest a narrowing of the gender gap. Studies from the National Institute on Alcohol Abuse and Alcoholism (NIAAA) from 1991 to 1992 and 2001 to 2002, found:
- a significant increase in the 12-month prevalence of alcohol abuse among men (4.7% to 6.9%) and women (1.5% to 2.6%).
- a significant decrease in alcohol dependence among men (6.3% to 5.4%) but not women (from 2.6% to 2.3%).
- a significant increase in rate of alcohol dependence among African-American women age 18 to 29 (from 2.1% to 3.8%).
- the gender differential in alcohol dependence narrowed from 3.8% to 3.1%.2,3
Similarly, studies conducted 60 years ago showed that boys were more likely than girls to report first alcohol use between ages 10 and 14 (4:1 ratio). Now the age of first alcohol use is relatively equivalent in boys and girls.3 This convergence is disconcerting and suggests that screening and prevention initiatives for adolescents need to increase their focus on girls.
Overall, these observations support the ‘convergence hypothesis,’ which holds that the gap between men’s and women’s alcohol consumption has narrowed as women’s use of alcohol has increased.4,5
Accelerated consequences
As a group, women may consume less alcohol than men but progress more rapidly to alcohol-related illnesses and negative consequences. Specifically, alcohol-dependent women develop liver disease, hypertension, and gastrointestinal hemorrhage more rapidly than alcohol-dependent men.3 Cognitive deficits and brain atrophy also develop sooner in alcohol-dependent women than men.6 Causes of this accelerated progression—”telescoping”—include gender-specific biological differences:
- Women have lower levels of gastric alcohol dehydrogenase—the enzyme that initiates alcohol metabolism—and therefore experience a higher blood alcohol concentration than men drinking the same amount of alcohol.
- Women have less total body water and less capacity to dilute alcohol than men.
‘At-risk’ drinking. Gender-related physiologic differences have led to different thresholds for defining “at-risk drinking” and binge drinking for men and women (Table 1).7 Alcohol use also has negative effects on women’s reproductive system and menstrual cycle. Women with alcohol dependence have higher rates of sexual dysfunction, irregular menstrual cycles, early menopause, and amenorrhea as compared with nonalcohol-dependent women.8
Table 1
Different thresholds: Number of drinks* that increases risk for alcohol-related problems
| Drinks/week | Drinks/day | |
|---|---|---|
| Men | >14 | >4 |
| Women | >7 | >3 |
| *A standard drink is 12 ounces of beer, 5 ounces of wine, or 1.5 ounces of spirits | ||
| Source: Reference 7 | ||
Motives for drinking
Research suggests different motives for alcohol use in girls and women vs boys and men. Women appear more likely to use alcohol to “self-medicate” negative affect or emotional pain (Box 2).9-12 Men are more likely to use alcohol to enhance pleasurable emotional states, “feel high” or because of social pressures to conform.5
Drinking partners. Compared with men’s motives for alcohol use, women’s drinking motives are more strongly influenced by their spouse’s or partner’s drinking patterns. In a large twin study (N = 5,974), 13% of alcohol-dependent women vs 3% of alcohol-dependent men reported that their spouse had a history of alcohol problems.9 Thus, consider the addiction status and drinking patterns of a woman’s partner when developing a treatment plan and relapse prevention strategies.
Trauma and alcohol use. Converging lines of evidence suggest that a relationship among trauma, posttraumatic stress disorder, and substance use disorders is particularly important for women. Early life stress—particularly sexual abuse—is more common in girls than in boys and is associated with the risk of developing substance use disorders.10
Women in alcohol treatment programs report more moderate (87%) and severe (40%) intimate partner violence than women in community samples (28% and 8%, respectively), as well as higher rates of childhood physical abuse (37% vs 11%, respectively).11 Moreover, alcohol abuse places women at risk for repeated victimization and serves to perpetuate a cycle of victimization and addiction.12 These are critical issues to assess and address when treating women with alcohol dependence.
In a study of >1,200 adolescents, girls age 13 With age, drinking for social and enhancement motives increases more among male than female adolescents. Drinking to reduce or manage negative affect is associated with an increased risk of heavy drinking (particularly for adolescent girls), more frequent intoxication, and alcohol dependence.14 These findings suggest:
- Young women with alcohol use problems may benefit from learning strategies to regulate negative emotions and alleviate depression or anxiety symptoms that may be contributing to their drinking.
- Young men with alcohol use problems may benefit from learning to manage peer pressure and from finding alternate sources of pleasure enhancement.5
CASE CONTINUED: Depressed and anxious
Ms. F admits to depressed mood, crying episodes, isolation from others, anhedonia, feelings of guilt, low motivation, difficulty concentrating at work, restless sleep, and weight loss. These symptoms recurred soon after she and her husband separated 4 years ago. She denies suicidal thoughts or suicide attempts.
She suffered similar episodes in the past, including when she was breastfeeding and abstinent from alcohol after the birth of her daughter. Her obstetrician treated her depression with fluoxetine. Problems with anxiety began in high school, especially associated with parties and dating. In college, she often would drink beer before class presentations.
You diagnose alcohol dependence, major depressive disorder, and social phobia. You also ask Ms. F about a history of trauma. She reports that her father was “real harsh” when he was drinking and often hit her and her sister. She also relates being struck by her ex-husband during arguments. Screening with the Clinician-Administered PTSD Scale is negative for posttraumatic stress disorder (PTSD), however.
Comorbid psychiatric disorders
Gender differences in reasons for alcohol use may be related to women’s higher rate of medical and psychiatric comorbidity (Table 2).6,8,15,16 Vital signs, physical examination, and lab work are helpful for diagnosis and monitoring of medical comorbidities and complications. The gamma-glutamyl transferase (GGT) test for liver disease and carbohydrate deficient transferrin (CDT) test for chronic heavy alcohol consumption are less sensitive and specific in women than in men.17 Even so, monitoring GGT and CDT results over time may serve as a valuable marker of continued drinking by women.
Mood and anxiety disorders are significantly more common in women than in men among individuals with alcohol use disorders.15,16 This pattern is not unique to alcohol-dependent persons, however. In the general population:
- women are more likely than men to meet diagnostic criteria for anxiety, depression, bulimia nervosa, and borderline personality disorder
- men are more likely than women to meet diagnostic criteria for antisocial personality disorder.18
Comorbid disorders in women with alcohol dependence
| Psychiatric Posttraumatic stress disorder Other anxiety disorders (such as panic disorder without agoraphobia, simple phobia, or social phobia) Major depressive disorder Cognitive impairment |
| Medical Hypertension Fatty liver disease Gastrointestinal hemorrhage Brain atrophy Reproductive system irregularities |
| Source: References 6,8,15,16 |
Suicide risk. A recent study by Connor et al20 examined suicidal ideation, planning, and attempts in 3,729 alcohol dependent subjects (35% female) and found an association between female gender and both planned (odds ratio [OR] = 3.4) and unplanned suicide attempts (OR = 3.8). Further research is needed to clarify the relationship between female gender, alcohol dependence, and suicide risk, behaviors, and attempts.
Anxiety disorders are the most common psychiatric disorders in women,21 and social anxiety and social phobia may play a predisposing role in alcohol dependence.22 Individuals with social anxiety may self-medicate with alcohol as a social lubricant. Some research suggests that anxiety disorders are more severe in alcohol-dependent women than in men with similarly severe alcohol dependence.22
23
Therefore, when assessing and treating alcohol-dependent women, screen for trauma history as well as mood and anxiety disorders. To optimize outcomes, treat these disorders simultaneously with the alcohol use disorder.24
Treatment planning
Underuse of treatment programs. Women with alcohol dependence are more likely to seek treatment in primary care or mental health settings, rather than in alcohol treatment settings.25,26 Women’s underuse of alcohol treatment programs is likely related to:
- greater stigma associated with alcohol use for women as compared with men
- socioeconomic factors, including pregnancy, child care, and concerns about child custody issues.25
Gender-specific treatment? Women-only treatment programs have been studied because of observed differences in men’s and women’s interaction styles and the hypothesis that men’s traditional societal dominance could negatively affect women in mixed-gender groups.25 Better treatment outcomes have been hypothesized if treatment is tailored to address women’s unique issues: risk factors for alcohol dependence, course of disease progression, medical problems associated with alcohol dependence, and reasons for relapse.
Gender-specific treatment may provide an environment where women—particularly those with a history of trauma from a male perpetrator—feel safe discussing issues related to their alcohol problems. For practical purposes, these programs also may be more likely to address women’s needs for on-site child care, prenatal care, and mental health programming.
A recent study compared a manual-based 12-session women’s recovery group with mixed-gender, manualized group drug counseling (GDC). The women’s recovery group focused on gender-specific topics such as relationships, the caregiver role, trauma, comorbid psychiatric conditions (including eating, mood, and anxiety disorders), and the effects of drug and alcohol use on women’s health. The women’s recovery group was:
- as effective as mixed-gender GDC in reducing substance use during the 12-week treatment
- significantly more effective during the 6-month post-treatment phase.28
CASE CONTINUED: Developing a treatment plan
Ms. F identifies 3 triggers for her alcohol use: a stressful day at school, arguments with her ex-husband, and feeling lonely. Because these are high-risk situations for relapse, you incorporate strategies to deal with them into her treatment plan. Other factors to consider:
- whether she requires detoxification
- an FDA-approved medication for alcohol dependence (acamprosate, oral or injectable naltrexone, or disulfiram)
- cognitive-behavioral therapy and medication for major depression and social phobia
- referral to psychosocial support groups (such as Alcoholics Anonymous).
Three factors determine the need for detoxification: the course of previous alcohol withdrawals (alcoholic hallucinosis, seizures, or delirium tremens), elevated vital signs or other evidence of autonomic hyperactivity such as diaphoresis or tremors, and the patient’s general medical condition. During early recovery, monitor patients closely to assess mood and anxiety symptoms. Blood alcohol tests or GGT and CDT are useful to monitor self-reported abstinence.
Related resources
- National Clearing House for Alcohol and Drug Information. Publications and materials on women and substance abuse. Substance Abuse and Mental Health Services Administration. http://ncadistore.samhsa.gov/catalogresults.aspx?h=drugs&topic=11.
- National Institute on Drug Abuse (www.nida.nih.gov). Articles that address women’s health and gender differences. www.drugabuse.gov/NIDA_Notes/NN0013.html.
- Acamprosate • Campral
- Disulfiram • Antabuse
- Fluoxetine • Prozac
- Naltrexone • Vivitrol, ReVia
Drs. Payne, Back, Wright, and Hartwell report no financial relationship with any company whose products are mentioned in this article or with manufacturers of competing products.
Dr. Brady receives research support from Abbott Laboratories, GlaxoSmithKline, Forest Pharmaceuticals, and Wyeth. She is a consultant and speaker for Pfizer Inc., Eli Lilly and Company, Abbott Laboratories, GlaxoSmithKline, and Forest Pharmaceuticals.
Ms. F, a 53-year-old high school English teacher, is referred to you by her family physician after she was suspended from work for suspected intoxication. She was divorced 2 years ago from her husband of 20 years, and she says her drinking has escalated to 2 bottles of wine every night. She wants to reduce her alcohol use but experiences shakiness, nausea, and diaphoresis when she tries to cut back.
Ms. F began drinking at age 16 “to feel more comfortable in social situations” and has experienced binge drinking with intermittent blackouts. She denies illicit drug use and legal consequences from drinking. Her father died from cirrhosis at age 58. Her mother suffers from depression but is in remission with medication.
Ms. F is hesitant to date or establish intimate relationships. She has stopped attending church, a book club, and her 15-year-old daughter’s booster club activities.
For years, little was known about alcohol use and alcohol-related problems in women such as Ms. F.1 Alcohol dependence studies rarely included women, so findings and treatment outcomes observed in men were assumed to apply to both genders.
Awareness of gender differences in addiction has grown (Box 1).2-5 Biological and psychosocial differences between alcohol-dependent women and men now are understood to influence etiology, epidemiology, psychiatric and medical comorbidity, course of illness, and treatment outcomes. This article discusses recent insights into planning treatment to address specific needs of alcohol-dependent women.
Epidemiologic surveys consistently show higher rates of alcohol use disorders in men than women, but recent data suggest a narrowing of the gender gap. Studies from the National Institute on Alcohol Abuse and Alcoholism (NIAAA) from 1991 to 1992 and 2001 to 2002, found:
- a significant increase in the 12-month prevalence of alcohol abuse among men (4.7% to 6.9%) and women (1.5% to 2.6%).
- a significant decrease in alcohol dependence among men (6.3% to 5.4%) but not women (from 2.6% to 2.3%).
- a significant increase in rate of alcohol dependence among African-American women age 18 to 29 (from 2.1% to 3.8%).
- the gender differential in alcohol dependence narrowed from 3.8% to 3.1%.2,3
Similarly, studies conducted 60 years ago showed that boys were more likely than girls to report first alcohol use between ages 10 and 14 (4:1 ratio). Now the age of first alcohol use is relatively equivalent in boys and girls.3 This convergence is disconcerting and suggests that screening and prevention initiatives for adolescents need to increase their focus on girls.
Overall, these observations support the ‘convergence hypothesis,’ which holds that the gap between men’s and women’s alcohol consumption has narrowed as women’s use of alcohol has increased.4,5
Accelerated consequences
As a group, women may consume less alcohol than men but progress more rapidly to alcohol-related illnesses and negative consequences. Specifically, alcohol-dependent women develop liver disease, hypertension, and gastrointestinal hemorrhage more rapidly than alcohol-dependent men.3 Cognitive deficits and brain atrophy also develop sooner in alcohol-dependent women than men.6 Causes of this accelerated progression—”telescoping”—include gender-specific biological differences:
- Women have lower levels of gastric alcohol dehydrogenase—the enzyme that initiates alcohol metabolism—and therefore experience a higher blood alcohol concentration than men drinking the same amount of alcohol.
- Women have less total body water and less capacity to dilute alcohol than men.
‘At-risk’ drinking. Gender-related physiologic differences have led to different thresholds for defining “at-risk drinking” and binge drinking for men and women (Table 1).7 Alcohol use also has negative effects on women’s reproductive system and menstrual cycle. Women with alcohol dependence have higher rates of sexual dysfunction, irregular menstrual cycles, early menopause, and amenorrhea as compared with nonalcohol-dependent women.8
Table 1
Different thresholds: Number of drinks* that increases risk for alcohol-related problems
| Drinks/week | Drinks/day | |
|---|---|---|
| Men | >14 | >4 |
| Women | >7 | >3 |
| *A standard drink is 12 ounces of beer, 5 ounces of wine, or 1.5 ounces of spirits | ||
| Source: Reference 7 | ||
Motives for drinking
Research suggests different motives for alcohol use in girls and women vs boys and men. Women appear more likely to use alcohol to “self-medicate” negative affect or emotional pain (Box 2).9-12 Men are more likely to use alcohol to enhance pleasurable emotional states, “feel high” or because of social pressures to conform.5
Drinking partners. Compared with men’s motives for alcohol use, women’s drinking motives are more strongly influenced by their spouse’s or partner’s drinking patterns. In a large twin study (N = 5,974), 13% of alcohol-dependent women vs 3% of alcohol-dependent men reported that their spouse had a history of alcohol problems.9 Thus, consider the addiction status and drinking patterns of a woman’s partner when developing a treatment plan and relapse prevention strategies.
Trauma and alcohol use. Converging lines of evidence suggest that a relationship among trauma, posttraumatic stress disorder, and substance use disorders is particularly important for women. Early life stress—particularly sexual abuse—is more common in girls than in boys and is associated with the risk of developing substance use disorders.10
Women in alcohol treatment programs report more moderate (87%) and severe (40%) intimate partner violence than women in community samples (28% and 8%, respectively), as well as higher rates of childhood physical abuse (37% vs 11%, respectively).11 Moreover, alcohol abuse places women at risk for repeated victimization and serves to perpetuate a cycle of victimization and addiction.12 These are critical issues to assess and address when treating women with alcohol dependence.
In a study of >1,200 adolescents, girls age 13 With age, drinking for social and enhancement motives increases more among male than female adolescents. Drinking to reduce or manage negative affect is associated with an increased risk of heavy drinking (particularly for adolescent girls), more frequent intoxication, and alcohol dependence.14 These findings suggest:
- Young women with alcohol use problems may benefit from learning strategies to regulate negative emotions and alleviate depression or anxiety symptoms that may be contributing to their drinking.
- Young men with alcohol use problems may benefit from learning to manage peer pressure and from finding alternate sources of pleasure enhancement.5
CASE CONTINUED: Depressed and anxious
Ms. F admits to depressed mood, crying episodes, isolation from others, anhedonia, feelings of guilt, low motivation, difficulty concentrating at work, restless sleep, and weight loss. These symptoms recurred soon after she and her husband separated 4 years ago. She denies suicidal thoughts or suicide attempts.
She suffered similar episodes in the past, including when she was breastfeeding and abstinent from alcohol after the birth of her daughter. Her obstetrician treated her depression with fluoxetine. Problems with anxiety began in high school, especially associated with parties and dating. In college, she often would drink beer before class presentations.
You diagnose alcohol dependence, major depressive disorder, and social phobia. You also ask Ms. F about a history of trauma. She reports that her father was “real harsh” when he was drinking and often hit her and her sister. She also relates being struck by her ex-husband during arguments. Screening with the Clinician-Administered PTSD Scale is negative for posttraumatic stress disorder (PTSD), however.
Comorbid psychiatric disorders
Gender differences in reasons for alcohol use may be related to women’s higher rate of medical and psychiatric comorbidity (Table 2).6,8,15,16 Vital signs, physical examination, and lab work are helpful for diagnosis and monitoring of medical comorbidities and complications. The gamma-glutamyl transferase (GGT) test for liver disease and carbohydrate deficient transferrin (CDT) test for chronic heavy alcohol consumption are less sensitive and specific in women than in men.17 Even so, monitoring GGT and CDT results over time may serve as a valuable marker of continued drinking by women.
Mood and anxiety disorders are significantly more common in women than in men among individuals with alcohol use disorders.15,16 This pattern is not unique to alcohol-dependent persons, however. In the general population:
- women are more likely than men to meet diagnostic criteria for anxiety, depression, bulimia nervosa, and borderline personality disorder
- men are more likely than women to meet diagnostic criteria for antisocial personality disorder.18
Comorbid disorders in women with alcohol dependence
| Psychiatric Posttraumatic stress disorder Other anxiety disorders (such as panic disorder without agoraphobia, simple phobia, or social phobia) Major depressive disorder Cognitive impairment |
| Medical Hypertension Fatty liver disease Gastrointestinal hemorrhage Brain atrophy Reproductive system irregularities |
| Source: References 6,8,15,16 |
Suicide risk. A recent study by Connor et al20 examined suicidal ideation, planning, and attempts in 3,729 alcohol dependent subjects (35% female) and found an association between female gender and both planned (odds ratio [OR] = 3.4) and unplanned suicide attempts (OR = 3.8). Further research is needed to clarify the relationship between female gender, alcohol dependence, and suicide risk, behaviors, and attempts.
Anxiety disorders are the most common psychiatric disorders in women,21 and social anxiety and social phobia may play a predisposing role in alcohol dependence.22 Individuals with social anxiety may self-medicate with alcohol as a social lubricant. Some research suggests that anxiety disorders are more severe in alcohol-dependent women than in men with similarly severe alcohol dependence.22
23
Therefore, when assessing and treating alcohol-dependent women, screen for trauma history as well as mood and anxiety disorders. To optimize outcomes, treat these disorders simultaneously with the alcohol use disorder.24
Treatment planning
Underuse of treatment programs. Women with alcohol dependence are more likely to seek treatment in primary care or mental health settings, rather than in alcohol treatment settings.25,26 Women’s underuse of alcohol treatment programs is likely related to:
- greater stigma associated with alcohol use for women as compared with men
- socioeconomic factors, including pregnancy, child care, and concerns about child custody issues.25
Gender-specific treatment? Women-only treatment programs have been studied because of observed differences in men’s and women’s interaction styles and the hypothesis that men’s traditional societal dominance could negatively affect women in mixed-gender groups.25 Better treatment outcomes have been hypothesized if treatment is tailored to address women’s unique issues: risk factors for alcohol dependence, course of disease progression, medical problems associated with alcohol dependence, and reasons for relapse.
Gender-specific treatment may provide an environment where women—particularly those with a history of trauma from a male perpetrator—feel safe discussing issues related to their alcohol problems. For practical purposes, these programs also may be more likely to address women’s needs for on-site child care, prenatal care, and mental health programming.
A recent study compared a manual-based 12-session women’s recovery group with mixed-gender, manualized group drug counseling (GDC). The women’s recovery group focused on gender-specific topics such as relationships, the caregiver role, trauma, comorbid psychiatric conditions (including eating, mood, and anxiety disorders), and the effects of drug and alcohol use on women’s health. The women’s recovery group was:
- as effective as mixed-gender GDC in reducing substance use during the 12-week treatment
- significantly more effective during the 6-month post-treatment phase.28
CASE CONTINUED: Developing a treatment plan
Ms. F identifies 3 triggers for her alcohol use: a stressful day at school, arguments with her ex-husband, and feeling lonely. Because these are high-risk situations for relapse, you incorporate strategies to deal with them into her treatment plan. Other factors to consider:
- whether she requires detoxification
- an FDA-approved medication for alcohol dependence (acamprosate, oral or injectable naltrexone, or disulfiram)
- cognitive-behavioral therapy and medication for major depression and social phobia
- referral to psychosocial support groups (such as Alcoholics Anonymous).
Three factors determine the need for detoxification: the course of previous alcohol withdrawals (alcoholic hallucinosis, seizures, or delirium tremens), elevated vital signs or other evidence of autonomic hyperactivity such as diaphoresis or tremors, and the patient’s general medical condition. During early recovery, monitor patients closely to assess mood and anxiety symptoms. Blood alcohol tests or GGT and CDT are useful to monitor self-reported abstinence.
Related resources
- National Clearing House for Alcohol and Drug Information. Publications and materials on women and substance abuse. Substance Abuse and Mental Health Services Administration. http://ncadistore.samhsa.gov/catalogresults.aspx?h=drugs&topic=11.
- National Institute on Drug Abuse (www.nida.nih.gov). Articles that address women’s health and gender differences. www.drugabuse.gov/NIDA_Notes/NN0013.html.
- Acamprosate • Campral
- Disulfiram • Antabuse
- Fluoxetine • Prozac
- Naltrexone • Vivitrol, ReVia
Drs. Payne, Back, Wright, and Hartwell report no financial relationship with any company whose products are mentioned in this article or with manufacturers of competing products.
Dr. Brady receives research support from Abbott Laboratories, GlaxoSmithKline, Forest Pharmaceuticals, and Wyeth. She is a consultant and speaker for Pfizer Inc., Eli Lilly and Company, Abbott Laboratories, GlaxoSmithKline, and Forest Pharmaceuticals.
1. Brady KT, Back SE, Greenfield S, eds. Women and addiction: a comprehensive handbook. New York, NY: Guilford Press; 2009.
2. Grant BF, Dawson DA, Stinson FS, et al. The 12-month prevalence and trends in DSM-IV alcohol abuse and dependence: United States, 1991-1992 and 2001-2002. Drug Alcohol Depend. 2004;74(3):223-234.
3. Greenfield SH. Women and alcohol use disorders. Harv Rev Psychiatry. 2002;10(2):76-85.
4. Greenfield TK, Room R. Situational norms for drinking and drunkenness: trends in the US adult population, 1979-1990. Addiction. 1997;92:33-47.
5. Stewart SH, Gavric D, Collins P. Women, girls, and alcohol. In: Brady KT, Back SE, Greenfield S, eds. Women and addiction: a comprehensive handbook. New York, NY: Guilford Press; 2009.
6. Mann K, Ackermann K, Croissant B, et al. Neuroimaging of gender differences in alcohol dependence: are women more vulnerable? Alcohol Clin Exp Res. 2005;29(5):896-901.
7. National Institute of Alcohol Abuse and Alcoholism. Helping patients who drink too much: a clinician’s guide. Bethesda, MD: National Institute of Alcohol Abuse and Alcoholism; 2005. NIH Publication No. 07-3769.
8. Blume SB, Zilberman ML. Addiction in women. In: Galanter M, Kleber H, eds. Textbook of substance abuse treatment. 3rd ed. Washington, DC: American Psychiatric Publishing, Inc; 2004:539-546.
9. Grant JD, Heath AC, Bucholz KK, et al. Spousal concordance for alcohol dependence: evidence for assortative mating or spousal interaction effect? Alcohol Clin Exp Res. 2007;31(5):717-728.
10. Kendler KS, Bulik CM, Silberg J, et al. Childhood sexual abuse and adult psychiatric and substance use disorders in women: an epidemiological and co-twin control analysis. Arch Gen Psychiatry. 2000;57:953-959.
11. Miller BA, Wilsnack SC, Cunradi CB. Family violence and victimization: treatment issues for women with alcohol problems. Alcohol Clin Exp Res. 2000;24(8):1287-1297.
12. Kilpatrick DG, Resnick HS, Saunders BE, et al. Victimization, posttraumatic stress disorder, and substance use and abuse among women. In: Wetherington CL, Roman AB, eds. Drug addiction research and the health of women. Rockville, MD: U.S. Department of Health and Human Services; 1998. NIH 98-4290:285-307.
13. Cooper ML. Motivations for alcohol use among adolescents: development and validation of a four-factor model. Psychol Assess. 1994;6(2):117-128.
14. Prescott CA, Cross RJ, Kuhn JW, et al. Is risk for alcoholism mediated by individual differences in drinking motivations? Alcohol Clin Exp Res. 2004;28(1):29-39.
15. Brady KT, Grice DE, Dustan L, et al. Gender differences in substance use disorders. Am J Psychiatry. 1993;150:1707-1711.
16. Helzer JE, Pryzbeck TR. The co-occurrence of alcoholism with other psychiatric disorders in the general population and its impact on treatment. J Stud Alcohol. 1988;49(3):219-224.
17. Center for Substance Abuse Treatment. Detoxification and substance abuse treatment. Treatment Improvement Protocol (TIP) Series 45. Rockville, MD: Substance Abuse and Mental Health Services Administration, 2006. DHHS (SMA) 06-4131.
18. Sinha R, Rounsaville BJ. Sex differences in depressed substance abusers. J Clin Psychiatry. 2002;63(7):616-627.
19. Gilman SE, Abraham HD. A longitudinal study of the order of onset of alcohol dependence and major depression. Drug Alcohol Depend. 2001;63(3):277-286.
20. Connor KR, Hesselbrock VM, Meldrum SC, et al. Transitions to, and correlates of, suicidal ideations, plans, and unplanned and planned suicide attempts among 3,728 men and women with alcohol dependence. J Stud Alcohol Drugs. 2007;68:654-662.
21. Pigott TA. Anxiety disorders in women. Psychiatr Clin North Am. 2003;26(3):621-672.
22. Randall CL, Thomas SE, Thevos AK. Gender comparison in alcoholics with concurrent social phobia: implications for alcoholism treatment. Am J Addict. 2000;9(3):202-215.
23. Logan TK, Walker R, Cole J, et al. Victimization and substance abuse among women: contributing factors, interventions, and implications. Review of General Psychology. 2002;6:325-397.
24. Volkow N. Comorbidity: addiction and other mental illnesses. Rockville, MD: U.S. Department of Health and Human Services; 2009. NIH 08-5771.
25. Greenfield SF, Brooks AJ, Gordon SM, et al. Substance abuse treatment entry, retention, and outcome in women: a review of the literature. Drug Alcohol Depend. 2007;86(1):1-21.
26. Green CA. Gender and use of substance abuse treatment services. Alcohol Res Health. 2006;29(1):55-62.
27. Brady TM, Ashley OS. Women in substance abuse treatment: results from the alcohol and drug services study (ADSS). Rockville, MD: Substance Abuse and Mental Health Services Administration, Office of Applied Studies; 2005. DHHS Publication No. SMA 04-3968, Analytic Series A-26. Available at: http://www.oas.samhsa.gov/womenTX/womenTX.pdf. Accessed July 22, 2007.
28. Greenfield SF, Trucco EM, McHugh RK, et al. The Women’s Recovery Group Study: a stage I trial of women-focused group therapy for substance use disorders versus mixedgender group drug counseling. Drug Alcohol Depend. 2007;90:39-47.
29. Garbutt JC, Kranzler HR, O’Malley SS, et al. Efficacy and tolerability of long-acting injectable naltrexone for alcohol dependence: a randomized controlled trial. JAMA. 2005;293(13):1617-1625.
30. O’Malley SS, Sinha R, Grilo CM, et al. Naltrexone and cognitive behavioral coping skills therapy for the treatment of alcohol drinking and eating disorder features in alcoholdependent women: a randomized controlled trial. Alcohol Clin Exp Res. 2007;31(4):625-634.
1. Brady KT, Back SE, Greenfield S, eds. Women and addiction: a comprehensive handbook. New York, NY: Guilford Press; 2009.
2. Grant BF, Dawson DA, Stinson FS, et al. The 12-month prevalence and trends in DSM-IV alcohol abuse and dependence: United States, 1991-1992 and 2001-2002. Drug Alcohol Depend. 2004;74(3):223-234.
3. Greenfield SH. Women and alcohol use disorders. Harv Rev Psychiatry. 2002;10(2):76-85.
4. Greenfield TK, Room R. Situational norms for drinking and drunkenness: trends in the US adult population, 1979-1990. Addiction. 1997;92:33-47.
5. Stewart SH, Gavric D, Collins P. Women, girls, and alcohol. In: Brady KT, Back SE, Greenfield S, eds. Women and addiction: a comprehensive handbook. New York, NY: Guilford Press; 2009.
6. Mann K, Ackermann K, Croissant B, et al. Neuroimaging of gender differences in alcohol dependence: are women more vulnerable? Alcohol Clin Exp Res. 2005;29(5):896-901.
7. National Institute of Alcohol Abuse and Alcoholism. Helping patients who drink too much: a clinician’s guide. Bethesda, MD: National Institute of Alcohol Abuse and Alcoholism; 2005. NIH Publication No. 07-3769.
8. Blume SB, Zilberman ML. Addiction in women. In: Galanter M, Kleber H, eds. Textbook of substance abuse treatment. 3rd ed. Washington, DC: American Psychiatric Publishing, Inc; 2004:539-546.
9. Grant JD, Heath AC, Bucholz KK, et al. Spousal concordance for alcohol dependence: evidence for assortative mating or spousal interaction effect? Alcohol Clin Exp Res. 2007;31(5):717-728.
10. Kendler KS, Bulik CM, Silberg J, et al. Childhood sexual abuse and adult psychiatric and substance use disorders in women: an epidemiological and co-twin control analysis. Arch Gen Psychiatry. 2000;57:953-959.
11. Miller BA, Wilsnack SC, Cunradi CB. Family violence and victimization: treatment issues for women with alcohol problems. Alcohol Clin Exp Res. 2000;24(8):1287-1297.
12. Kilpatrick DG, Resnick HS, Saunders BE, et al. Victimization, posttraumatic stress disorder, and substance use and abuse among women. In: Wetherington CL, Roman AB, eds. Drug addiction research and the health of women. Rockville, MD: U.S. Department of Health and Human Services; 1998. NIH 98-4290:285-307.
13. Cooper ML. Motivations for alcohol use among adolescents: development and validation of a four-factor model. Psychol Assess. 1994;6(2):117-128.
14. Prescott CA, Cross RJ, Kuhn JW, et al. Is risk for alcoholism mediated by individual differences in drinking motivations? Alcohol Clin Exp Res. 2004;28(1):29-39.
15. Brady KT, Grice DE, Dustan L, et al. Gender differences in substance use disorders. Am J Psychiatry. 1993;150:1707-1711.
16. Helzer JE, Pryzbeck TR. The co-occurrence of alcoholism with other psychiatric disorders in the general population and its impact on treatment. J Stud Alcohol. 1988;49(3):219-224.
17. Center for Substance Abuse Treatment. Detoxification and substance abuse treatment. Treatment Improvement Protocol (TIP) Series 45. Rockville, MD: Substance Abuse and Mental Health Services Administration, 2006. DHHS (SMA) 06-4131.
18. Sinha R, Rounsaville BJ. Sex differences in depressed substance abusers. J Clin Psychiatry. 2002;63(7):616-627.
19. Gilman SE, Abraham HD. A longitudinal study of the order of onset of alcohol dependence and major depression. Drug Alcohol Depend. 2001;63(3):277-286.
20. Connor KR, Hesselbrock VM, Meldrum SC, et al. Transitions to, and correlates of, suicidal ideations, plans, and unplanned and planned suicide attempts among 3,728 men and women with alcohol dependence. J Stud Alcohol Drugs. 2007;68:654-662.
21. Pigott TA. Anxiety disorders in women. Psychiatr Clin North Am. 2003;26(3):621-672.
22. Randall CL, Thomas SE, Thevos AK. Gender comparison in alcoholics with concurrent social phobia: implications for alcoholism treatment. Am J Addict. 2000;9(3):202-215.
23. Logan TK, Walker R, Cole J, et al. Victimization and substance abuse among women: contributing factors, interventions, and implications. Review of General Psychology. 2002;6:325-397.
24. Volkow N. Comorbidity: addiction and other mental illnesses. Rockville, MD: U.S. Department of Health and Human Services; 2009. NIH 08-5771.
25. Greenfield SF, Brooks AJ, Gordon SM, et al. Substance abuse treatment entry, retention, and outcome in women: a review of the literature. Drug Alcohol Depend. 2007;86(1):1-21.
26. Green CA. Gender and use of substance abuse treatment services. Alcohol Res Health. 2006;29(1):55-62.
27. Brady TM, Ashley OS. Women in substance abuse treatment: results from the alcohol and drug services study (ADSS). Rockville, MD: Substance Abuse and Mental Health Services Administration, Office of Applied Studies; 2005. DHHS Publication No. SMA 04-3968, Analytic Series A-26. Available at: http://www.oas.samhsa.gov/womenTX/womenTX.pdf. Accessed July 22, 2007.
28. Greenfield SF, Trucco EM, McHugh RK, et al. The Women’s Recovery Group Study: a stage I trial of women-focused group therapy for substance use disorders versus mixedgender group drug counseling. Drug Alcohol Depend. 2007;90:39-47.
29. Garbutt JC, Kranzler HR, O’Malley SS, et al. Efficacy and tolerability of long-acting injectable naltrexone for alcohol dependence: a randomized controlled trial. JAMA. 2005;293(13):1617-1625.
30. O’Malley SS, Sinha R, Grilo CM, et al. Naltrexone and cognitive behavioral coping skills therapy for the treatment of alcohol drinking and eating disorder features in alcoholdependent women: a randomized controlled trial. Alcohol Clin Exp Res. 2007;31(4):625-634.
