User login
M. Alexander Otto began his reporting career early in 1999 covering the pharmaceutical industry for a national pharmacists' magazine and freelancing for the Washington Post and other newspapers. He then joined BNA, now part of Bloomberg News, covering health law and the protection of people and animals in medical research. Alex next worked for the McClatchy Company. Based on his work, Alex won a year-long Knight Science Journalism Fellowship to MIT in 2008-2009. He joined the company shortly thereafter. Alex has a newspaper journalism degree from Syracuse (N.Y.) University and a master's degree in medical science -- a physician assistant degree -- from George Washington University. Alex is based in Seattle.
Maternal DTaP Vaccination Protected Newborns From Pertussis
VANCOUVER, B.C. – Infants born to women who receive diphtheria-tetanus-acellular pertussis vaccine during pregnancy have higher pertussis antibody levels during their first few months of life than infants born to unvaccinated women, Dr. Abbey Hardy-Fairbanks reported.
The levels are sufficient to protect infants against pertussis prior to their first diphtheria-tetanus-acellular pertussis (DTaP) shot at around 2 months, a period of "significant pertussis morbidity and mortality," said Dr. Hardy-Fairbanks, an ob.gyn. at the University of Iowa, Iowa City. "This is the first evidence to document that pertussis immunization during pregnancy is likely to be beneficial to infants when they are most vulnerable to pertussis disease. [Physicians] should consider vaccination of women during pregnancy with DTaP," she said at the annual meeting of the Infectious Diseases Society of America.
In the prospective cohort study, 16 (23%) of 70 pregnant women received DTaP vaccine; 54 (77%) pregnant women selected as controls did not and had not been vaccinated for at least 2 years.
Four of the women (25%) in the DTaP group were vaccinated in the first trimester, eight (50%) in the second, and four (25%) in the third. Vaccination did not cause any adverse pregnancy outcomes.
Maternal blood and cord blood were collected at delivery. Blood was also collected from children before and after their primary DTaP series and toddler booster doses at 12-18 months.
Blood samples were measured for pertussis antigens, including pertussis toxoid, filamentous hemagglutinin, pertactin, and fimbriae, by enzyme-linked immunosorbent assay.
Newborns in the DTaP group had higher pertussis antibody concentrations than their mothers, "showing efficient placental transfer of antibodies to the infant," Dr. Hardy-Fairbanks said.
They also had substantially higher concentrations than infants in the control group prior to the start of the primary DTaP series, and the differences were statistically significant.
However, at month 7, following completion of the DTaP series, infants born to vaccinated mothers had slightly lower antibody levels than infants in the control group. The differences were not statistically significant, but "may represent some blunting of the infant immune response to the [vaccine]," Dr. Hardy-Fairbanks said.
By the time they got their toddler booster doses, however, antibody levels "were essentially equivalent" in the two groups, she said.
Dr. Hardy-Fairbanks said the infants’ responses to polio, Haemophilus influenzae
type b, and hepatitis B vaccines are being analyzed.
Dr. Hardy-Fairbanks said she had no conflicts of interest. The study was funded by Sanofi-Pasteur, maker of Daptacel DTaP vaccine.
Dr. Sarah Long thanked the study authors for their work. "Your findings are so very helpful. We don’t have this kind of information."
She was concerned, however, that infants born to vaccinated mothers mounted only a blunted immune response to their primary DTaP vaccine series, and wondered if responses would be blunted to other vaccines. The study’s presenter said the question is currently being investigated, but so far that does not appear to be the case.
Dr. Sarah Long is the chief of the section of infectious diseases at St. Christopher’s Hospital for Children in Philadelphia. She said she had no conflicts of interest.
Dr. Sarah Long thanked the study authors for their work. "Your findings are so very helpful. We don’t have this kind of information."
She was concerned, however, that infants born to vaccinated mothers mounted only a blunted immune response to their primary DTaP vaccine series, and wondered if responses would be blunted to other vaccines. The study’s presenter said the question is currently being investigated, but so far that does not appear to be the case.
Dr. Sarah Long is the chief of the section of infectious diseases at St. Christopher’s Hospital for Children in Philadelphia. She said she had no conflicts of interest.
Dr. Sarah Long thanked the study authors for their work. "Your findings are so very helpful. We don’t have this kind of information."
She was concerned, however, that infants born to vaccinated mothers mounted only a blunted immune response to their primary DTaP vaccine series, and wondered if responses would be blunted to other vaccines. The study’s presenter said the question is currently being investigated, but so far that does not appear to be the case.
Dr. Sarah Long is the chief of the section of infectious diseases at St. Christopher’s Hospital for Children in Philadelphia. She said she had no conflicts of interest.
VANCOUVER, B.C. – Infants born to women who receive diphtheria-tetanus-acellular pertussis vaccine during pregnancy have higher pertussis antibody levels during their first few months of life than infants born to unvaccinated women, Dr. Abbey Hardy-Fairbanks reported.
The levels are sufficient to protect infants against pertussis prior to their first diphtheria-tetanus-acellular pertussis (DTaP) shot at around 2 months, a period of "significant pertussis morbidity and mortality," said Dr. Hardy-Fairbanks, an ob.gyn. at the University of Iowa, Iowa City. "This is the first evidence to document that pertussis immunization during pregnancy is likely to be beneficial to infants when they are most vulnerable to pertussis disease. [Physicians] should consider vaccination of women during pregnancy with DTaP," she said at the annual meeting of the Infectious Diseases Society of America.
In the prospective cohort study, 16 (23%) of 70 pregnant women received DTaP vaccine; 54 (77%) pregnant women selected as controls did not and had not been vaccinated for at least 2 years.
Four of the women (25%) in the DTaP group were vaccinated in the first trimester, eight (50%) in the second, and four (25%) in the third. Vaccination did not cause any adverse pregnancy outcomes.
Maternal blood and cord blood were collected at delivery. Blood was also collected from children before and after their primary DTaP series and toddler booster doses at 12-18 months.
Blood samples were measured for pertussis antigens, including pertussis toxoid, filamentous hemagglutinin, pertactin, and fimbriae, by enzyme-linked immunosorbent assay.
Newborns in the DTaP group had higher pertussis antibody concentrations than their mothers, "showing efficient placental transfer of antibodies to the infant," Dr. Hardy-Fairbanks said.
They also had substantially higher concentrations than infants in the control group prior to the start of the primary DTaP series, and the differences were statistically significant.
However, at month 7, following completion of the DTaP series, infants born to vaccinated mothers had slightly lower antibody levels than infants in the control group. The differences were not statistically significant, but "may represent some blunting of the infant immune response to the [vaccine]," Dr. Hardy-Fairbanks said.
By the time they got their toddler booster doses, however, antibody levels "were essentially equivalent" in the two groups, she said.
Dr. Hardy-Fairbanks said the infants’ responses to polio, Haemophilus influenzae
type b, and hepatitis B vaccines are being analyzed.
Dr. Hardy-Fairbanks said she had no conflicts of interest. The study was funded by Sanofi-Pasteur, maker of Daptacel DTaP vaccine.
VANCOUVER, B.C. – Infants born to women who receive diphtheria-tetanus-acellular pertussis vaccine during pregnancy have higher pertussis antibody levels during their first few months of life than infants born to unvaccinated women, Dr. Abbey Hardy-Fairbanks reported.
The levels are sufficient to protect infants against pertussis prior to their first diphtheria-tetanus-acellular pertussis (DTaP) shot at around 2 months, a period of "significant pertussis morbidity and mortality," said Dr. Hardy-Fairbanks, an ob.gyn. at the University of Iowa, Iowa City. "This is the first evidence to document that pertussis immunization during pregnancy is likely to be beneficial to infants when they are most vulnerable to pertussis disease. [Physicians] should consider vaccination of women during pregnancy with DTaP," she said at the annual meeting of the Infectious Diseases Society of America.
In the prospective cohort study, 16 (23%) of 70 pregnant women received DTaP vaccine; 54 (77%) pregnant women selected as controls did not and had not been vaccinated for at least 2 years.
Four of the women (25%) in the DTaP group were vaccinated in the first trimester, eight (50%) in the second, and four (25%) in the third. Vaccination did not cause any adverse pregnancy outcomes.
Maternal blood and cord blood were collected at delivery. Blood was also collected from children before and after their primary DTaP series and toddler booster doses at 12-18 months.
Blood samples were measured for pertussis antigens, including pertussis toxoid, filamentous hemagglutinin, pertactin, and fimbriae, by enzyme-linked immunosorbent assay.
Newborns in the DTaP group had higher pertussis antibody concentrations than their mothers, "showing efficient placental transfer of antibodies to the infant," Dr. Hardy-Fairbanks said.
They also had substantially higher concentrations than infants in the control group prior to the start of the primary DTaP series, and the differences were statistically significant.
However, at month 7, following completion of the DTaP series, infants born to vaccinated mothers had slightly lower antibody levels than infants in the control group. The differences were not statistically significant, but "may represent some blunting of the infant immune response to the [vaccine]," Dr. Hardy-Fairbanks said.
By the time they got their toddler booster doses, however, antibody levels "were essentially equivalent" in the two groups, she said.
Dr. Hardy-Fairbanks said the infants’ responses to polio, Haemophilus influenzae
type b, and hepatitis B vaccines are being analyzed.
Dr. Hardy-Fairbanks said she had no conflicts of interest. The study was funded by Sanofi-Pasteur, maker of Daptacel DTaP vaccine.
FROM THE ANNUAL MEETING OF THE INFECTIOUS DISEASES SOCIETY OF AMERICA
Major Finding: Newborns in the DTaP group had substantially higher concentrations than infants in the control group prior to the start of their primary DTaP series, and the differences were statistically significant.
Data Source: Prospective cohort study involving 70 women and their infants; some of the mothers received DTaP vaccine during pregnancy.
Disclosures: Dr. Hardy-Fairbanks said she had no conflicts of interest. The study was funded by Sanofi-Pasteur, maker of Daptacel DTaP vaccine.
ATLAS Score Correlates With Outcomes in C. Difficile
VANCOUVER, B.C. - A simple scoring of five bedside assessments when Clostridium difficile infection is first diagnosed correlates significantly with cure rate.
"The higher the score, the lower the cure rate," said Dr. Mark Miller, head of the division of infectious diseases and chief of the department of microbiology at SMBD–Jewish General Hospital, McGill University, Montreal, who presented the findings.
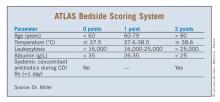
The five parameters are age, temperature, leukocytosis, albumin, and systemic concomitant antibiotic use, ATLAS for short. The first four are rated on a 0-2 scale; 2 is added to the score if the patient is on systemic antibiotics, 0 if not. ATLAS scores range from 0 to 10 (see box).
The score also correlates with recurrence, but the correlation is not statistically significant.
Dr. Miller said there is a need to be able to categorize patients by C. difficile infection (CDI) severity to determine who should be treated aggressively, assign and assess outcomes in clinical studies, and communicate with other medical workers.
"If someone calls up and says ‘I have a case of moderate CDI,’ it’s pretty much left up to the imagination about what they are talking about," at present, he said.
Although much work has been done previously to create a prognostic system for CDI, proposed systems have not been adequately validated, Dr. Miller said.
However, "if you look at all these publications, it’s all the same risk factors," he added.
So Dr. Miller and his colleagues combined them. "What we came up with was a simple combination of the bedside risk factors that are easy to collect and, we feel, should be most associated with cure and recurrence."
C. difficile strain type was omitted because it’s not usually known at the time of diagnosis; baseline serum creatinine isn’t either, so its elevation above baseline also was excluded.
ATLAS was tested using patient data from a large North American trial comparing fidaxomicin to vancomycin for CDI.
The ATLAS scores of 516 patients were calculated at their time of diagnosis and matched against their cure rates following 10 days of study treatment.
There was "an excellent correlation with cure rate," Dr. Miller said. (R2 0.88, P value less than .001).
Patients with an ATLAS score of 0 had a 98% cure rate; the rate dropped incrementally with higher scores. ATLAS scores of 7 corresponded to a 55% cure rate.
Dr. Miller and his colleagues then checked the 450 subjects cured after treatment to see who had gotten another C. difficile infection.
"With recurrence, the ATLAS score didn’t fair quite so well," he said.
Recurrence rates climbed with higher scores; 11% of patients with a 0 score had a recurrence, 43% with a score of 6.
But the correlation was weak (R2, 0.32) and insignificant (P, .14).
A subgroup analysis found that 229 patients assigned to the vancomycin arm threw the recurrence results off (R2, 0.02, P, .762). ATLAS scores predicted recurrence better in 221 fidaxomicin subjects (R2, 0.70, P, .009).
Recurrence rates in the vancomycin arm were much higher, not neatly distributed along a curve, which might have thrown off the results, Dr. Miller said.
Perhaps, there may also "be some additional refinement of the systemic antibiotics score that would improve" ATLAS’s correlation with recurrence, he said.
A second study presented in Vancouver showed significant correlation between ATLAS scores and 30-day CDI mortality in 308 adults aged 60 years or older.
"ATLAS score appears to ... predict severity in CDI in our patient population," according to the abstract, of which Dr. Miller was a coauthor.
Dr. Miller disclosed that he is a scientific adviser and grant investigator to several pharmaceutical companies, including Merck & Co., Novartis Pharmaceutical, and Optimer Pharmaceuticals, makers of fidaxomicin.
VANCOUVER, B.C. - A simple scoring of five bedside assessments when Clostridium difficile infection is first diagnosed correlates significantly with cure rate.
"The higher the score, the lower the cure rate," said Dr. Mark Miller, head of the division of infectious diseases and chief of the department of microbiology at SMBD–Jewish General Hospital, McGill University, Montreal, who presented the findings.
The five parameters are age, temperature, leukocytosis, albumin, and systemic concomitant antibiotic use, ATLAS for short. The first four are rated on a 0-2 scale; 2 is added to the score if the patient is on systemic antibiotics, 0 if not. ATLAS scores range from 0 to 10 (see box).
The score also correlates with recurrence, but the correlation is not statistically significant.
Dr. Miller said there is a need to be able to categorize patients by C. difficile infection (CDI) severity to determine who should be treated aggressively, assign and assess outcomes in clinical studies, and communicate with other medical workers.
"If someone calls up and says ‘I have a case of moderate CDI,’ it’s pretty much left up to the imagination about what they are talking about," at present, he said.
Although much work has been done previously to create a prognostic system for CDI, proposed systems have not been adequately validated, Dr. Miller said.
However, "if you look at all these publications, it’s all the same risk factors," he added.
So Dr. Miller and his colleagues combined them. "What we came up with was a simple combination of the bedside risk factors that are easy to collect and, we feel, should be most associated with cure and recurrence."
C. difficile strain type was omitted because it’s not usually known at the time of diagnosis; baseline serum creatinine isn’t either, so its elevation above baseline also was excluded.
ATLAS was tested using patient data from a large North American trial comparing fidaxomicin to vancomycin for CDI.
The ATLAS scores of 516 patients were calculated at their time of diagnosis and matched against their cure rates following 10 days of study treatment.
There was "an excellent correlation with cure rate," Dr. Miller said. (R2 0.88, P value less than .001).
Patients with an ATLAS score of 0 had a 98% cure rate; the rate dropped incrementally with higher scores. ATLAS scores of 7 corresponded to a 55% cure rate.
Dr. Miller and his colleagues then checked the 450 subjects cured after treatment to see who had gotten another C. difficile infection.
"With recurrence, the ATLAS score didn’t fair quite so well," he said.
Recurrence rates climbed with higher scores; 11% of patients with a 0 score had a recurrence, 43% with a score of 6.
But the correlation was weak (R2, 0.32) and insignificant (P, .14).
A subgroup analysis found that 229 patients assigned to the vancomycin arm threw the recurrence results off (R2, 0.02, P, .762). ATLAS scores predicted recurrence better in 221 fidaxomicin subjects (R2, 0.70, P, .009).
Recurrence rates in the vancomycin arm were much higher, not neatly distributed along a curve, which might have thrown off the results, Dr. Miller said.
Perhaps, there may also "be some additional refinement of the systemic antibiotics score that would improve" ATLAS’s correlation with recurrence, he said.
A second study presented in Vancouver showed significant correlation between ATLAS scores and 30-day CDI mortality in 308 adults aged 60 years or older.
"ATLAS score appears to ... predict severity in CDI in our patient population," according to the abstract, of which Dr. Miller was a coauthor.
Dr. Miller disclosed that he is a scientific adviser and grant investigator to several pharmaceutical companies, including Merck & Co., Novartis Pharmaceutical, and Optimer Pharmaceuticals, makers of fidaxomicin.
VANCOUVER, B.C. - A simple scoring of five bedside assessments when Clostridium difficile infection is first diagnosed correlates significantly with cure rate.
"The higher the score, the lower the cure rate," said Dr. Mark Miller, head of the division of infectious diseases and chief of the department of microbiology at SMBD–Jewish General Hospital, McGill University, Montreal, who presented the findings.
The five parameters are age, temperature, leukocytosis, albumin, and systemic concomitant antibiotic use, ATLAS for short. The first four are rated on a 0-2 scale; 2 is added to the score if the patient is on systemic antibiotics, 0 if not. ATLAS scores range from 0 to 10 (see box).
The score also correlates with recurrence, but the correlation is not statistically significant.
Dr. Miller said there is a need to be able to categorize patients by C. difficile infection (CDI) severity to determine who should be treated aggressively, assign and assess outcomes in clinical studies, and communicate with other medical workers.
"If someone calls up and says ‘I have a case of moderate CDI,’ it’s pretty much left up to the imagination about what they are talking about," at present, he said.
Although much work has been done previously to create a prognostic system for CDI, proposed systems have not been adequately validated, Dr. Miller said.
However, "if you look at all these publications, it’s all the same risk factors," he added.
So Dr. Miller and his colleagues combined them. "What we came up with was a simple combination of the bedside risk factors that are easy to collect and, we feel, should be most associated with cure and recurrence."
C. difficile strain type was omitted because it’s not usually known at the time of diagnosis; baseline serum creatinine isn’t either, so its elevation above baseline also was excluded.
ATLAS was tested using patient data from a large North American trial comparing fidaxomicin to vancomycin for CDI.
The ATLAS scores of 516 patients were calculated at their time of diagnosis and matched against their cure rates following 10 days of study treatment.
There was "an excellent correlation with cure rate," Dr. Miller said. (R2 0.88, P value less than .001).
Patients with an ATLAS score of 0 had a 98% cure rate; the rate dropped incrementally with higher scores. ATLAS scores of 7 corresponded to a 55% cure rate.
Dr. Miller and his colleagues then checked the 450 subjects cured after treatment to see who had gotten another C. difficile infection.
"With recurrence, the ATLAS score didn’t fair quite so well," he said.
Recurrence rates climbed with higher scores; 11% of patients with a 0 score had a recurrence, 43% with a score of 6.
But the correlation was weak (R2, 0.32) and insignificant (P, .14).
A subgroup analysis found that 229 patients assigned to the vancomycin arm threw the recurrence results off (R2, 0.02, P, .762). ATLAS scores predicted recurrence better in 221 fidaxomicin subjects (R2, 0.70, P, .009).
Recurrence rates in the vancomycin arm were much higher, not neatly distributed along a curve, which might have thrown off the results, Dr. Miller said.
Perhaps, there may also "be some additional refinement of the systemic antibiotics score that would improve" ATLAS’s correlation with recurrence, he said.
A second study presented in Vancouver showed significant correlation between ATLAS scores and 30-day CDI mortality in 308 adults aged 60 years or older.
"ATLAS score appears to ... predict severity in CDI in our patient population," according to the abstract, of which Dr. Miller was a coauthor.
Dr. Miller disclosed that he is a scientific adviser and grant investigator to several pharmaceutical companies, including Merck & Co., Novartis Pharmaceutical, and Optimer Pharmaceuticals, makers of fidaxomicin.
FROM THE ANNUAL MEETING OF THE INFECTIOUS DISEASES SOCIETY OF AMERICA
ATLAS Score Correlates With Outcomes in C. Difficile
VANCOUVER, B.C. - A simple scoring of five bedside assessments when Clostridium difficile infection is first diagnosed correlates significantly with cure rate.
"The higher the score, the lower the cure rate," said Dr. Mark Miller, head of the division of infectious diseases and chief of the department of microbiology at SMBD–Jewish General Hospital, McGill University, Montreal, who presented the findings.
The five parameters are age, temperature, leukocytosis, albumin, and systemic concomitant antibiotic use, ATLAS for short. The first four are rated on a 0-2 scale; 2 is added to the score if the patient is on systemic antibiotics, 0 if not. ATLAS scores range from 0 to 10 (see box).
The score also correlates with recurrence, but the correlation is not statistically significant.
Dr. Miller said there is a need to be able to categorize patients by C. difficile infection (CDI) severity to determine who should be treated aggressively, assign and assess outcomes in clinical studies, and communicate with other medical workers.
"If someone calls up and says ‘I have a case of moderate CDI,’ it’s pretty much left up to the imagination about what they are talking about," at present, he said.
Although much work has been done previously to create a prognostic system for CDI, proposed systems have not been adequately validated, Dr. Miller said.
However, "if you look at all these publications, it’s all the same risk factors," he added.
So Dr. Miller and his colleagues combined them. "What we came up with was a simple combination of the bedside risk factors that are easy to collect and, we feel, should be most associated with cure and recurrence."
C. difficile strain type was omitted because it’s not usually known at the time of diagnosis; baseline serum creatinine isn’t either, so its elevation above baseline also was excluded.
ATLAS was tested using patient data from a large North American trial comparing fidaxomicin to vancomycin for CDI.
The ATLAS scores of 516 patients were calculated at their time of diagnosis and matched against their cure rates following 10 days of study treatment.
There was "an excellent correlation with cure rate," Dr. Miller said. (R2 0.88, P value less than .001).
Patients with an ATLAS score of 0 had a 98% cure rate; the rate dropped incrementally with higher scores. ATLAS scores of 7 corresponded to a 55% cure rate.
Dr. Miller and his colleagues then checked the 450 subjects cured after treatment to see who had gotten another C. difficile infection.
"With recurrence, the ATLAS score didn’t fair quite so well," he said.
Recurrence rates climbed with higher scores; 11% of patients with a 0 score had a recurrence, 43% with a score of 6.
But the correlation was weak (R2, 0.32) and insignificant (P, .14).
A subgroup analysis found that 229 patients assigned to the vancomycin arm threw the recurrence results off (R2, 0.02, P, .762). ATLAS scores predicted recurrence better in 221 fidaxomicin subjects (R2, 0.70, P, .009).
Recurrence rates in the vancomycin arm were much higher, not neatly distributed along a curve, which might have thrown off the results, Dr. Miller said.
Perhaps, there may also "be some additional refinement of the systemic antibiotics score that would improve" ATLAS’s correlation with recurrence, he said.
A second study presented in Vancouver showed significant correlation between ATLAS scores and 30-day CDI mortality in 308 adults aged 60 years or older.
"ATLAS score appears to ... predict severity in CDI in our patient population," according to the abstract, of which Dr. Miller was a coauthor.
Dr. Miller disclosed that he is a scientific adviser and grant investigator to several pharmaceutical companies, including Merck & Co., Novartis Pharmaceutical, and Optimer Pharmaceuticals, makers of fidaxomicin.
VANCOUVER, B.C. - A simple scoring of five bedside assessments when Clostridium difficile infection is first diagnosed correlates significantly with cure rate.
"The higher the score, the lower the cure rate," said Dr. Mark Miller, head of the division of infectious diseases and chief of the department of microbiology at SMBD–Jewish General Hospital, McGill University, Montreal, who presented the findings.
The five parameters are age, temperature, leukocytosis, albumin, and systemic concomitant antibiotic use, ATLAS for short. The first four are rated on a 0-2 scale; 2 is added to the score if the patient is on systemic antibiotics, 0 if not. ATLAS scores range from 0 to 10 (see box).
The score also correlates with recurrence, but the correlation is not statistically significant.
Dr. Miller said there is a need to be able to categorize patients by C. difficile infection (CDI) severity to determine who should be treated aggressively, assign and assess outcomes in clinical studies, and communicate with other medical workers.
"If someone calls up and says ‘I have a case of moderate CDI,’ it’s pretty much left up to the imagination about what they are talking about," at present, he said.
Although much work has been done previously to create a prognostic system for CDI, proposed systems have not been adequately validated, Dr. Miller said.
However, "if you look at all these publications, it’s all the same risk factors," he added.
So Dr. Miller and his colleagues combined them. "What we came up with was a simple combination of the bedside risk factors that are easy to collect and, we feel, should be most associated with cure and recurrence."
C. difficile strain type was omitted because it’s not usually known at the time of diagnosis; baseline serum creatinine isn’t either, so its elevation above baseline also was excluded.
ATLAS was tested using patient data from a large North American trial comparing fidaxomicin to vancomycin for CDI.
The ATLAS scores of 516 patients were calculated at their time of diagnosis and matched against their cure rates following 10 days of study treatment.
There was "an excellent correlation with cure rate," Dr. Miller said. (R2 0.88, P value less than .001).
Patients with an ATLAS score of 0 had a 98% cure rate; the rate dropped incrementally with higher scores. ATLAS scores of 7 corresponded to a 55% cure rate.
Dr. Miller and his colleagues then checked the 450 subjects cured after treatment to see who had gotten another C. difficile infection.
"With recurrence, the ATLAS score didn’t fair quite so well," he said.
Recurrence rates climbed with higher scores; 11% of patients with a 0 score had a recurrence, 43% with a score of 6.
But the correlation was weak (R2, 0.32) and insignificant (P, .14).
A subgroup analysis found that 229 patients assigned to the vancomycin arm threw the recurrence results off (R2, 0.02, P, .762). ATLAS scores predicted recurrence better in 221 fidaxomicin subjects (R2, 0.70, P, .009).
Recurrence rates in the vancomycin arm were much higher, not neatly distributed along a curve, which might have thrown off the results, Dr. Miller said.
Perhaps, there may also "be some additional refinement of the systemic antibiotics score that would improve" ATLAS’s correlation with recurrence, he said.
A second study presented in Vancouver showed significant correlation between ATLAS scores and 30-day CDI mortality in 308 adults aged 60 years or older.
"ATLAS score appears to ... predict severity in CDI in our patient population," according to the abstract, of which Dr. Miller was a coauthor.
Dr. Miller disclosed that he is a scientific adviser and grant investigator to several pharmaceutical companies, including Merck & Co., Novartis Pharmaceutical, and Optimer Pharmaceuticals, makers of fidaxomicin.
VANCOUVER, B.C. - A simple scoring of five bedside assessments when Clostridium difficile infection is first diagnosed correlates significantly with cure rate.
"The higher the score, the lower the cure rate," said Dr. Mark Miller, head of the division of infectious diseases and chief of the department of microbiology at SMBD–Jewish General Hospital, McGill University, Montreal, who presented the findings.
The five parameters are age, temperature, leukocytosis, albumin, and systemic concomitant antibiotic use, ATLAS for short. The first four are rated on a 0-2 scale; 2 is added to the score if the patient is on systemic antibiotics, 0 if not. ATLAS scores range from 0 to 10 (see box).
The score also correlates with recurrence, but the correlation is not statistically significant.
Dr. Miller said there is a need to be able to categorize patients by C. difficile infection (CDI) severity to determine who should be treated aggressively, assign and assess outcomes in clinical studies, and communicate with other medical workers.
"If someone calls up and says ‘I have a case of moderate CDI,’ it’s pretty much left up to the imagination about what they are talking about," at present, he said.
Although much work has been done previously to create a prognostic system for CDI, proposed systems have not been adequately validated, Dr. Miller said.
However, "if you look at all these publications, it’s all the same risk factors," he added.
So Dr. Miller and his colleagues combined them. "What we came up with was a simple combination of the bedside risk factors that are easy to collect and, we feel, should be most associated with cure and recurrence."
C. difficile strain type was omitted because it’s not usually known at the time of diagnosis; baseline serum creatinine isn’t either, so its elevation above baseline also was excluded.
ATLAS was tested using patient data from a large North American trial comparing fidaxomicin to vancomycin for CDI.
The ATLAS scores of 516 patients were calculated at their time of diagnosis and matched against their cure rates following 10 days of study treatment.
There was "an excellent correlation with cure rate," Dr. Miller said. (R2 0.88, P value less than .001).
Patients with an ATLAS score of 0 had a 98% cure rate; the rate dropped incrementally with higher scores. ATLAS scores of 7 corresponded to a 55% cure rate.
Dr. Miller and his colleagues then checked the 450 subjects cured after treatment to see who had gotten another C. difficile infection.
"With recurrence, the ATLAS score didn’t fair quite so well," he said.
Recurrence rates climbed with higher scores; 11% of patients with a 0 score had a recurrence, 43% with a score of 6.
But the correlation was weak (R2, 0.32) and insignificant (P, .14).
A subgroup analysis found that 229 patients assigned to the vancomycin arm threw the recurrence results off (R2, 0.02, P, .762). ATLAS scores predicted recurrence better in 221 fidaxomicin subjects (R2, 0.70, P, .009).
Recurrence rates in the vancomycin arm were much higher, not neatly distributed along a curve, which might have thrown off the results, Dr. Miller said.
Perhaps, there may also "be some additional refinement of the systemic antibiotics score that would improve" ATLAS’s correlation with recurrence, he said.
A second study presented in Vancouver showed significant correlation between ATLAS scores and 30-day CDI mortality in 308 adults aged 60 years or older.
"ATLAS score appears to ... predict severity in CDI in our patient population," according to the abstract, of which Dr. Miller was a coauthor.
Dr. Miller disclosed that he is a scientific adviser and grant investigator to several pharmaceutical companies, including Merck & Co., Novartis Pharmaceutical, and Optimer Pharmaceuticals, makers of fidaxomicin.
FROM THE ANNUAL MEETING OF THE INFECTIOUS DISEASES SOCIETY OF AMERICA
Major Finding: A score based on age, temperature, leukocytosis, albumin, and systemic antibiotic use correlates with cure rates in C. difficile infection with a P value of less than.001.
Data Source: ATLAS was tested using patient data from a large North American trial comparing fidaxomicin to vancomycin for CDI. ATLAS scores for 516 patients with CDI were calculated at their time of diagnosis and matched against their cure rates following 10 days of study treatment.
Disclosures: Dr. Miller is a scientific adviser and grant investigator to several pharmaceutical companies, including Merck & Co., Novartis Pharmaceuticals, and Optimer Pharmaceuticals.
More Convenient Shots Increased Immunization Rates in Health Care Workers
VANCOUVER, B.C. – At a time when only about half of health care workers are immunized against influenza, a concerted effort by a hospital in Omaha, Neb., led to a remarkable upswing in the number of employees there who got a flu shot, without being mandated to do so, according to Dr. Archana Chatterjee.
Through common sense approaches like offering employees shots after hours and on weekends, and requiring those who refused a shot to wear a surgical mask, Children’s Hospital and Medical Center saw its health care worker vaccination rates climb steadily from about 52% in 2002 to 86% in 2008.
To get the remaining holdouts, the hospital did enact a mandate in 2009, but "I think the important thing is that we did not have protests; we did not have people refusing vaccinations when it came to the mandate in 2009," Dr. Chatterjee said at the annual meeting of the Infectious Diseases Society of America.
There were no legal challenges to the mandate or widespread employee dissatisfaction, unlike at other institutions. Today, the vaccination rate at the hospital is about 97% among the 1,860 employees. Health care workers at the hospital are not unionized, said Dr. Chatterjee, a hospital epidemiologist and pediatrics professor at Creighton University, Omaha.
"I think the reason [there were no protests] is that in previous years, we had worked with the employees to respond to their concerns [and to] educate them. That’s what resulted in relatively high rates of immunization against influenza, even before the mandate was put in place," she said.
Dr. Chatterjee gave an example of the education efforts. "We had a surgeon who thought he never had influenza. Those are the kinds of myths that we have to dispel to help people recognize that influenza can be mild or even asymptomatic in some people," but they are still able to spread it, she said.
Other steps included not charging for the shots, taking the shot cart to where people were working on the patient floors and having coworker colleagues administer the vaccine, offering vaccinations during annual physicals, and putting reminders about flu shots on the hospital’s computer system.
Also, "last year there was a request to provide vaccine to family members, and we actually did do that, as well, at various venues at various times." Dr. Chatterjee said that the key to the program’s success was that "we were responsive to the issues that were being raised. There was something that they saw changed as a result of bringing that issue to us," she said.
The hospital also updated the form employees must sign if they refused the vaccine.
"The original form just said ‘I refuse to have the vaccine’," Dr. Chatterjee said.
The updated one "made it very clear that if they refuse the vaccine, they are putting their patients at risk and putting their coworkers at risk," she said.
The form also requires employees to wear a surgical mask throughout the influenza season if they refuse the vaccine, even if they produce a medical exemption.
The updated refusal form states that "those who do not comply with the mask requirement will be subject to corrective action up to and including termination."
When employees ignored the order, "we would get reports from people saying, ‘You know xyz has not been vaccinated and is not wearing a mask’?"; everyone on the unit knew who was not vaccinated.
In those cases, infection control nurses would visit the workers and remind them that they had signed the document.
"A couple of people actually started [wearing the masks] and then realized what an onerous job wearing a mask for the entire shift was. Those medical exemptions quickly disappeared," Dr. Chatterjee said.
"It was pretty clear to us that there were some key changes that made big differences, and the mask issue was one of those," she said.
Dr. Andrew Pavia, chief of the division of infectious disease in the department of pediatrics at the University of Utah, Salt Lake City, noted that such voluntary programs go only so far in raising vaccination rates. "The best possible voluntary programs only get health care worker vaccination rates into the mid-80% range. This is the reason that so many health care organizations have endorsed mandatory immunization."
There isn’t an adequate percentage below 100% at which you stop transmission, and "so we need to target something that approaches 100%. It appears, right now, that mandatory immunization is the policy that works."
Dr. Chatterjee and Dr. Pavia reported having no conflicts of interest.
VANCOUVER, B.C. – At a time when only about half of health care workers are immunized against influenza, a concerted effort by a hospital in Omaha, Neb., led to a remarkable upswing in the number of employees there who got a flu shot, without being mandated to do so, according to Dr. Archana Chatterjee.
Through common sense approaches like offering employees shots after hours and on weekends, and requiring those who refused a shot to wear a surgical mask, Children’s Hospital and Medical Center saw its health care worker vaccination rates climb steadily from about 52% in 2002 to 86% in 2008.
To get the remaining holdouts, the hospital did enact a mandate in 2009, but "I think the important thing is that we did not have protests; we did not have people refusing vaccinations when it came to the mandate in 2009," Dr. Chatterjee said at the annual meeting of the Infectious Diseases Society of America.
There were no legal challenges to the mandate or widespread employee dissatisfaction, unlike at other institutions. Today, the vaccination rate at the hospital is about 97% among the 1,860 employees. Health care workers at the hospital are not unionized, said Dr. Chatterjee, a hospital epidemiologist and pediatrics professor at Creighton University, Omaha.
"I think the reason [there were no protests] is that in previous years, we had worked with the employees to respond to their concerns [and to] educate them. That’s what resulted in relatively high rates of immunization against influenza, even before the mandate was put in place," she said.
Dr. Chatterjee gave an example of the education efforts. "We had a surgeon who thought he never had influenza. Those are the kinds of myths that we have to dispel to help people recognize that influenza can be mild or even asymptomatic in some people," but they are still able to spread it, she said.
Other steps included not charging for the shots, taking the shot cart to where people were working on the patient floors and having coworker colleagues administer the vaccine, offering vaccinations during annual physicals, and putting reminders about flu shots on the hospital’s computer system.
Also, "last year there was a request to provide vaccine to family members, and we actually did do that, as well, at various venues at various times." Dr. Chatterjee said that the key to the program’s success was that "we were responsive to the issues that were being raised. There was something that they saw changed as a result of bringing that issue to us," she said.
The hospital also updated the form employees must sign if they refused the vaccine.
"The original form just said ‘I refuse to have the vaccine’," Dr. Chatterjee said.
The updated one "made it very clear that if they refuse the vaccine, they are putting their patients at risk and putting their coworkers at risk," she said.
The form also requires employees to wear a surgical mask throughout the influenza season if they refuse the vaccine, even if they produce a medical exemption.
The updated refusal form states that "those who do not comply with the mask requirement will be subject to corrective action up to and including termination."
When employees ignored the order, "we would get reports from people saying, ‘You know xyz has not been vaccinated and is not wearing a mask’?"; everyone on the unit knew who was not vaccinated.
In those cases, infection control nurses would visit the workers and remind them that they had signed the document.
"A couple of people actually started [wearing the masks] and then realized what an onerous job wearing a mask for the entire shift was. Those medical exemptions quickly disappeared," Dr. Chatterjee said.
"It was pretty clear to us that there were some key changes that made big differences, and the mask issue was one of those," she said.
Dr. Andrew Pavia, chief of the division of infectious disease in the department of pediatrics at the University of Utah, Salt Lake City, noted that such voluntary programs go only so far in raising vaccination rates. "The best possible voluntary programs only get health care worker vaccination rates into the mid-80% range. This is the reason that so many health care organizations have endorsed mandatory immunization."
There isn’t an adequate percentage below 100% at which you stop transmission, and "so we need to target something that approaches 100%. It appears, right now, that mandatory immunization is the policy that works."
Dr. Chatterjee and Dr. Pavia reported having no conflicts of interest.
VANCOUVER, B.C. – At a time when only about half of health care workers are immunized against influenza, a concerted effort by a hospital in Omaha, Neb., led to a remarkable upswing in the number of employees there who got a flu shot, without being mandated to do so, according to Dr. Archana Chatterjee.
Through common sense approaches like offering employees shots after hours and on weekends, and requiring those who refused a shot to wear a surgical mask, Children’s Hospital and Medical Center saw its health care worker vaccination rates climb steadily from about 52% in 2002 to 86% in 2008.
To get the remaining holdouts, the hospital did enact a mandate in 2009, but "I think the important thing is that we did not have protests; we did not have people refusing vaccinations when it came to the mandate in 2009," Dr. Chatterjee said at the annual meeting of the Infectious Diseases Society of America.
There were no legal challenges to the mandate or widespread employee dissatisfaction, unlike at other institutions. Today, the vaccination rate at the hospital is about 97% among the 1,860 employees. Health care workers at the hospital are not unionized, said Dr. Chatterjee, a hospital epidemiologist and pediatrics professor at Creighton University, Omaha.
"I think the reason [there were no protests] is that in previous years, we had worked with the employees to respond to their concerns [and to] educate them. That’s what resulted in relatively high rates of immunization against influenza, even before the mandate was put in place," she said.
Dr. Chatterjee gave an example of the education efforts. "We had a surgeon who thought he never had influenza. Those are the kinds of myths that we have to dispel to help people recognize that influenza can be mild or even asymptomatic in some people," but they are still able to spread it, she said.
Other steps included not charging for the shots, taking the shot cart to where people were working on the patient floors and having coworker colleagues administer the vaccine, offering vaccinations during annual physicals, and putting reminders about flu shots on the hospital’s computer system.
Also, "last year there was a request to provide vaccine to family members, and we actually did do that, as well, at various venues at various times." Dr. Chatterjee said that the key to the program’s success was that "we were responsive to the issues that were being raised. There was something that they saw changed as a result of bringing that issue to us," she said.
The hospital also updated the form employees must sign if they refused the vaccine.
"The original form just said ‘I refuse to have the vaccine’," Dr. Chatterjee said.
The updated one "made it very clear that if they refuse the vaccine, they are putting their patients at risk and putting their coworkers at risk," she said.
The form also requires employees to wear a surgical mask throughout the influenza season if they refuse the vaccine, even if they produce a medical exemption.
The updated refusal form states that "those who do not comply with the mask requirement will be subject to corrective action up to and including termination."
When employees ignored the order, "we would get reports from people saying, ‘You know xyz has not been vaccinated and is not wearing a mask’?"; everyone on the unit knew who was not vaccinated.
In those cases, infection control nurses would visit the workers and remind them that they had signed the document.
"A couple of people actually started [wearing the masks] and then realized what an onerous job wearing a mask for the entire shift was. Those medical exemptions quickly disappeared," Dr. Chatterjee said.
"It was pretty clear to us that there were some key changes that made big differences, and the mask issue was one of those," she said.
Dr. Andrew Pavia, chief of the division of infectious disease in the department of pediatrics at the University of Utah, Salt Lake City, noted that such voluntary programs go only so far in raising vaccination rates. "The best possible voluntary programs only get health care worker vaccination rates into the mid-80% range. This is the reason that so many health care organizations have endorsed mandatory immunization."
There isn’t an adequate percentage below 100% at which you stop transmission, and "so we need to target something that approaches 100%. It appears, right now, that mandatory immunization is the policy that works."
Dr. Chatterjee and Dr. Pavia reported having no conflicts of interest.
FROM THE ANNUAL MEETING OF THE INFECTIOUS DISEASES SOCIETY OF AMERICA
Common Sense Policy Boosts Vaccination of Health Care Workers
VANCOUVER, B.C. – At a time when only about half of health care workers are immunized against influenza, a concerted effort by a hospital in Omaha, Neb., led to a remarkable upswing in the number of employees there who got a flu shot, without being mandated to do so, according to Dr. Archana Chatterjee.
Through common sense approaches like offering employees shots after hours and on weekends, and requiring those who refused a shot to wear a surgical mask, Children’s Hospital and Medical Center saw its health care worker vaccination rates climb steadily from about 52% in 2002 to 86% in 2008.
To get the remaining holdouts, the hospital did enact a mandate in 2009, but "I think the important thing is that we did not have protests; we did not have people refusing vaccinations when it came to the mandate in 2009," Dr. Chatterjee said at the annual meeting of the Infectious Diseases Society of America.
There were no legal challenges to the mandate or widespread employee dissatisfaction, unlike at other institutions. Today, the vaccination rate at the hospital is about 97% among the 1,860 employees. Health care workers at the hospital are not unionized, said Dr. Chatterjee, a hospital epidemiologist and pediatrics professor at Creighton University, Omaha.
"I think the reason [there were no protests] is that in previous years, we had worked with the employees to respond to their concerns [and to] educate them. That’s what resulted in relatively high rates of immunization against influenza, even before the mandate was put in place," she said.
Dr. Chatterjee gave an example of the education efforts. "We had a surgeon who thought he never had influenza. Those are the kinds of myths that we have to dispel to help people recognize that influenza can be mild or even asymptomatic in some people," but they are still able to spread it, she said.
Other steps included not charging for the shots, taking the shot cart to where people were working on the patient floors and having coworker colleagues administer the vaccine, offering vaccinations during annual physicals, and putting reminders about flu shots on the hospital’s computer system.
Also, "last year there was a request to provide vaccine to family members, and we actually did do that, as well, at various venues at various times." Dr. Chatterjee said that the key to the program’s success was that "we were responsive to the issues that were being raised. There was something that they saw changed as a result of bringing that issue to us," she said.
The hospital also updated the form employees must sign if they refused the vaccine.
"The original form just said 'I refuse to have the vaccine'," Dr. Chatterjee said.
The updated one "made it very clear that if they refuse the vaccine, they are putting their patients at risk and putting their coworkers at risk," she said.
The form also requires employees to wear a surgical mask throughout the influenza season if they refuse the vaccine, even if they produce a medical exemption.
The updated refusal form states that "those who do not comply with the mask requirement will be subject to corrective action up to and including termination."
When employees ignored the order, "we would get reports from people saying, 'You know xyz has not been vaccinated and is not wearing a mask'?"; everyone on the unit knew who was not vaccinated.
In those cases, infection control nurses would visit the workers and remind them that they had signed the document.
"A couple of people actually started [wearing the masks] and then realized what an onerous job wearing a mask for the entire shift was. Those medical exemptions quickly disappeared," Dr. Chatterjee said.
"It was pretty clear to us that there were some key changes that made big differences, and the mask issue was one of those," she said.
Dr. Andrew Pavia, chief of the division of infectious disease in the department of pediatrics at the University of Utah, Salt Lake City, noted that such voluntary programs go only so far in raising vaccination rates. "The best possible voluntary programs only get health care worker vaccination rates into the mid-80% range. This is the reason that so many health care organizations have endorsed mandatory immunization."
There isn't an adequate percentage below 100% at which you stop transmission, and "so we need to target something that approaches 100%. It appears, right now, that mandatory immunization is the policy that works."
Dr. Chatterjee and Dr. Pavia reported having no conflicts of interest.
VANCOUVER, B.C. – At a time when only about half of health care workers are immunized against influenza, a concerted effort by a hospital in Omaha, Neb., led to a remarkable upswing in the number of employees there who got a flu shot, without being mandated to do so, according to Dr. Archana Chatterjee.
Through common sense approaches like offering employees shots after hours and on weekends, and requiring those who refused a shot to wear a surgical mask, Children’s Hospital and Medical Center saw its health care worker vaccination rates climb steadily from about 52% in 2002 to 86% in 2008.
To get the remaining holdouts, the hospital did enact a mandate in 2009, but "I think the important thing is that we did not have protests; we did not have people refusing vaccinations when it came to the mandate in 2009," Dr. Chatterjee said at the annual meeting of the Infectious Diseases Society of America.
There were no legal challenges to the mandate or widespread employee dissatisfaction, unlike at other institutions. Today, the vaccination rate at the hospital is about 97% among the 1,860 employees. Health care workers at the hospital are not unionized, said Dr. Chatterjee, a hospital epidemiologist and pediatrics professor at Creighton University, Omaha.
"I think the reason [there were no protests] is that in previous years, we had worked with the employees to respond to their concerns [and to] educate them. That’s what resulted in relatively high rates of immunization against influenza, even before the mandate was put in place," she said.
Dr. Chatterjee gave an example of the education efforts. "We had a surgeon who thought he never had influenza. Those are the kinds of myths that we have to dispel to help people recognize that influenza can be mild or even asymptomatic in some people," but they are still able to spread it, she said.
Other steps included not charging for the shots, taking the shot cart to where people were working on the patient floors and having coworker colleagues administer the vaccine, offering vaccinations during annual physicals, and putting reminders about flu shots on the hospital’s computer system.
Also, "last year there was a request to provide vaccine to family members, and we actually did do that, as well, at various venues at various times." Dr. Chatterjee said that the key to the program’s success was that "we were responsive to the issues that were being raised. There was something that they saw changed as a result of bringing that issue to us," she said.
The hospital also updated the form employees must sign if they refused the vaccine.
"The original form just said 'I refuse to have the vaccine'," Dr. Chatterjee said.
The updated one "made it very clear that if they refuse the vaccine, they are putting their patients at risk and putting their coworkers at risk," she said.
The form also requires employees to wear a surgical mask throughout the influenza season if they refuse the vaccine, even if they produce a medical exemption.
The updated refusal form states that "those who do not comply with the mask requirement will be subject to corrective action up to and including termination."
When employees ignored the order, "we would get reports from people saying, 'You know xyz has not been vaccinated and is not wearing a mask'?"; everyone on the unit knew who was not vaccinated.
In those cases, infection control nurses would visit the workers and remind them that they had signed the document.
"A couple of people actually started [wearing the masks] and then realized what an onerous job wearing a mask for the entire shift was. Those medical exemptions quickly disappeared," Dr. Chatterjee said.
"It was pretty clear to us that there were some key changes that made big differences, and the mask issue was one of those," she said.
Dr. Andrew Pavia, chief of the division of infectious disease in the department of pediatrics at the University of Utah, Salt Lake City, noted that such voluntary programs go only so far in raising vaccination rates. "The best possible voluntary programs only get health care worker vaccination rates into the mid-80% range. This is the reason that so many health care organizations have endorsed mandatory immunization."
There isn't an adequate percentage below 100% at which you stop transmission, and "so we need to target something that approaches 100%. It appears, right now, that mandatory immunization is the policy that works."
Dr. Chatterjee and Dr. Pavia reported having no conflicts of interest.
VANCOUVER, B.C. – At a time when only about half of health care workers are immunized against influenza, a concerted effort by a hospital in Omaha, Neb., led to a remarkable upswing in the number of employees there who got a flu shot, without being mandated to do so, according to Dr. Archana Chatterjee.
Through common sense approaches like offering employees shots after hours and on weekends, and requiring those who refused a shot to wear a surgical mask, Children’s Hospital and Medical Center saw its health care worker vaccination rates climb steadily from about 52% in 2002 to 86% in 2008.
To get the remaining holdouts, the hospital did enact a mandate in 2009, but "I think the important thing is that we did not have protests; we did not have people refusing vaccinations when it came to the mandate in 2009," Dr. Chatterjee said at the annual meeting of the Infectious Diseases Society of America.
There were no legal challenges to the mandate or widespread employee dissatisfaction, unlike at other institutions. Today, the vaccination rate at the hospital is about 97% among the 1,860 employees. Health care workers at the hospital are not unionized, said Dr. Chatterjee, a hospital epidemiologist and pediatrics professor at Creighton University, Omaha.
"I think the reason [there were no protests] is that in previous years, we had worked with the employees to respond to their concerns [and to] educate them. That’s what resulted in relatively high rates of immunization against influenza, even before the mandate was put in place," she said.
Dr. Chatterjee gave an example of the education efforts. "We had a surgeon who thought he never had influenza. Those are the kinds of myths that we have to dispel to help people recognize that influenza can be mild or even asymptomatic in some people," but they are still able to spread it, she said.
Other steps included not charging for the shots, taking the shot cart to where people were working on the patient floors and having coworker colleagues administer the vaccine, offering vaccinations during annual physicals, and putting reminders about flu shots on the hospital’s computer system.
Also, "last year there was a request to provide vaccine to family members, and we actually did do that, as well, at various venues at various times." Dr. Chatterjee said that the key to the program’s success was that "we were responsive to the issues that were being raised. There was something that they saw changed as a result of bringing that issue to us," she said.
The hospital also updated the form employees must sign if they refused the vaccine.
"The original form just said 'I refuse to have the vaccine'," Dr. Chatterjee said.
The updated one "made it very clear that if they refuse the vaccine, they are putting their patients at risk and putting their coworkers at risk," she said.
The form also requires employees to wear a surgical mask throughout the influenza season if they refuse the vaccine, even if they produce a medical exemption.
The updated refusal form states that "those who do not comply with the mask requirement will be subject to corrective action up to and including termination."
When employees ignored the order, "we would get reports from people saying, 'You know xyz has not been vaccinated and is not wearing a mask'?"; everyone on the unit knew who was not vaccinated.
In those cases, infection control nurses would visit the workers and remind them that they had signed the document.
"A couple of people actually started [wearing the masks] and then realized what an onerous job wearing a mask for the entire shift was. Those medical exemptions quickly disappeared," Dr. Chatterjee said.
"It was pretty clear to us that there were some key changes that made big differences, and the mask issue was one of those," she said.
Dr. Andrew Pavia, chief of the division of infectious disease in the department of pediatrics at the University of Utah, Salt Lake City, noted that such voluntary programs go only so far in raising vaccination rates. "The best possible voluntary programs only get health care worker vaccination rates into the mid-80% range. This is the reason that so many health care organizations have endorsed mandatory immunization."
There isn't an adequate percentage below 100% at which you stop transmission, and "so we need to target something that approaches 100%. It appears, right now, that mandatory immunization is the policy that works."
Dr. Chatterjee and Dr. Pavia reported having no conflicts of interest.
FROM THE ANNUAL MEETING OF THE INFECTIOUS DISEASES SOCIETY OF AMERICA
Major Finding: About 86% of health care workers at a hospital in Omaha, Neb., agreed to get a flu shot, without being mandated to do so. Commonsense approaches, like making shots available after hours for night workers, increased vaccination rates.
Data Source: An observational study looking at vaccination rates before and after policy changes.
Disclosures: Dr. Chatterjee and Dr. Pavia said they had no conflicts of interest.
More Convenient Shots Increased Immunization Rates in Health Care Workers
VANCOUVER, B.C. – At a time when only about half of health care workers are immunized against influenza, a concerted effort by a hospital in Omaha, Neb., led to a remarkable upswing in the number of employees there who got a flu shot, without being mandated to do so, according to Dr. Archana Chatterjee.
Through common sense approaches like offering employees shots after hours and on weekends, and requiring those who refused a shot to wear a surgical mask, Children’s Hospital and Medical Center saw its health care worker vaccination rates climb steadily from about 52% in 2002 to 86% in 2008.
To get the remaining holdouts, the hospital did enact a mandate in 2009, but "I think the important thing is that we did not have protests; we did not have people refusing vaccinations when it came to the mandate in 2009," Dr. Chatterjee said at the annual meeting of the Infectious Diseases Society of America.
There were no legal challenges to the mandate or widespread employee dissatisfaction, unlike at other institutions. Today, the vaccination rate at the hospital is about 97% among the 1,860 employees. Health care workers at the hospital are not unionized, said Dr. Chatterjee, a hospital epidemiologist and pediatrics professor at Creighton University, Omaha.
"I think the reason [there were no protests] is that in previous years, we had worked with the employees to respond to their concerns [and to] educate them. That’s what resulted in relatively high rates of immunization against influenza, even before the mandate was put in place," she said.
Dr. Chatterjee gave an example of the education efforts. "We had a surgeon who thought he never had influenza. Those are the kinds of myths that we have to dispel to help people recognize that influenza can be mild or even asymptomatic in some people," but they are still able to spread it, she said.
Other steps included not charging for the shots, taking the shot cart to where people were working on the patient floors and having coworker colleagues administer the vaccine, offering vaccinations during annual physicals, and putting reminders about flu shots on the hospital’s computer system.
Also, "last year there was a request to provide vaccine to family members, and we actually did do that, as well, at various venues at various times." Dr. Chatterjee said that the key to the program’s success was that "we were responsive to the issues that were being raised. There was something that they saw changed as a result of bringing that issue to us," she said.
The hospital also updated the form employees must sign if they refused the vaccine.
"The original form just said ‘I refuse to have the vaccine’," Dr. Chatterjee said.
The updated one "made it very clear that if they refuse the vaccine, they are putting their patients at risk and putting their coworkers at risk," she said.
The form also requires employees to wear a surgical mask throughout the influenza season if they refuse the vaccine, even if they produce a medical exemption.
The updated refusal form states that "those who do not comply with the mask requirement will be subject to corrective action up to and including termination."
When employees ignored the order, "we would get reports from people saying, ‘You know xyz has not been vaccinated and is not wearing a mask’?"; everyone on the unit knew who was not vaccinated.
In those cases, infection control nurses would visit the workers and remind them that they had signed the document.
"A couple of people actually started [wearing the masks] and then realized what an onerous job wearing a mask for the entire shift was. Those medical exemptions quickly disappeared," Dr. Chatterjee said.
"It was pretty clear to us that there were some key changes that made big differences, and the mask issue was one of those," she said.
Dr. Andrew Pavia, chief of the division of infectious disease in the department of pediatrics at the University of Utah, Salt Lake City, noted that such voluntary programs go only so far in raising vaccination rates. "The best possible voluntary programs only get health care worker vaccination rates into the mid-80% range. This is the reason that so many health care organizations have endorsed mandatory immunization."
There isn’t an adequate percentage below 100% at which you stop transmission, and "so we need to target something that approaches 100%. It appears, right now, that mandatory immunization is the policy that works."
Dr. Chatterjee and Dr. Pavia reported having no conflicts of interest.
VANCOUVER, B.C. – At a time when only about half of health care workers are immunized against influenza, a concerted effort by a hospital in Omaha, Neb., led to a remarkable upswing in the number of employees there who got a flu shot, without being mandated to do so, according to Dr. Archana Chatterjee.
Through common sense approaches like offering employees shots after hours and on weekends, and requiring those who refused a shot to wear a surgical mask, Children’s Hospital and Medical Center saw its health care worker vaccination rates climb steadily from about 52% in 2002 to 86% in 2008.
To get the remaining holdouts, the hospital did enact a mandate in 2009, but "I think the important thing is that we did not have protests; we did not have people refusing vaccinations when it came to the mandate in 2009," Dr. Chatterjee said at the annual meeting of the Infectious Diseases Society of America.
There were no legal challenges to the mandate or widespread employee dissatisfaction, unlike at other institutions. Today, the vaccination rate at the hospital is about 97% among the 1,860 employees. Health care workers at the hospital are not unionized, said Dr. Chatterjee, a hospital epidemiologist and pediatrics professor at Creighton University, Omaha.
"I think the reason [there were no protests] is that in previous years, we had worked with the employees to respond to their concerns [and to] educate them. That’s what resulted in relatively high rates of immunization against influenza, even before the mandate was put in place," she said.
Dr. Chatterjee gave an example of the education efforts. "We had a surgeon who thought he never had influenza. Those are the kinds of myths that we have to dispel to help people recognize that influenza can be mild or even asymptomatic in some people," but they are still able to spread it, she said.
Other steps included not charging for the shots, taking the shot cart to where people were working on the patient floors and having coworker colleagues administer the vaccine, offering vaccinations during annual physicals, and putting reminders about flu shots on the hospital’s computer system.
Also, "last year there was a request to provide vaccine to family members, and we actually did do that, as well, at various venues at various times." Dr. Chatterjee said that the key to the program’s success was that "we were responsive to the issues that were being raised. There was something that they saw changed as a result of bringing that issue to us," she said.
The hospital also updated the form employees must sign if they refused the vaccine.
"The original form just said ‘I refuse to have the vaccine’," Dr. Chatterjee said.
The updated one "made it very clear that if they refuse the vaccine, they are putting their patients at risk and putting their coworkers at risk," she said.
The form also requires employees to wear a surgical mask throughout the influenza season if they refuse the vaccine, even if they produce a medical exemption.
The updated refusal form states that "those who do not comply with the mask requirement will be subject to corrective action up to and including termination."
When employees ignored the order, "we would get reports from people saying, ‘You know xyz has not been vaccinated and is not wearing a mask’?"; everyone on the unit knew who was not vaccinated.
In those cases, infection control nurses would visit the workers and remind them that they had signed the document.
"A couple of people actually started [wearing the masks] and then realized what an onerous job wearing a mask for the entire shift was. Those medical exemptions quickly disappeared," Dr. Chatterjee said.
"It was pretty clear to us that there were some key changes that made big differences, and the mask issue was one of those," she said.
Dr. Andrew Pavia, chief of the division of infectious disease in the department of pediatrics at the University of Utah, Salt Lake City, noted that such voluntary programs go only so far in raising vaccination rates. "The best possible voluntary programs only get health care worker vaccination rates into the mid-80% range. This is the reason that so many health care organizations have endorsed mandatory immunization."
There isn’t an adequate percentage below 100% at which you stop transmission, and "so we need to target something that approaches 100%. It appears, right now, that mandatory immunization is the policy that works."
Dr. Chatterjee and Dr. Pavia reported having no conflicts of interest.
VANCOUVER, B.C. – At a time when only about half of health care workers are immunized against influenza, a concerted effort by a hospital in Omaha, Neb., led to a remarkable upswing in the number of employees there who got a flu shot, without being mandated to do so, according to Dr. Archana Chatterjee.
Through common sense approaches like offering employees shots after hours and on weekends, and requiring those who refused a shot to wear a surgical mask, Children’s Hospital and Medical Center saw its health care worker vaccination rates climb steadily from about 52% in 2002 to 86% in 2008.
To get the remaining holdouts, the hospital did enact a mandate in 2009, but "I think the important thing is that we did not have protests; we did not have people refusing vaccinations when it came to the mandate in 2009," Dr. Chatterjee said at the annual meeting of the Infectious Diseases Society of America.
There were no legal challenges to the mandate or widespread employee dissatisfaction, unlike at other institutions. Today, the vaccination rate at the hospital is about 97% among the 1,860 employees. Health care workers at the hospital are not unionized, said Dr. Chatterjee, a hospital epidemiologist and pediatrics professor at Creighton University, Omaha.
"I think the reason [there were no protests] is that in previous years, we had worked with the employees to respond to their concerns [and to] educate them. That’s what resulted in relatively high rates of immunization against influenza, even before the mandate was put in place," she said.
Dr. Chatterjee gave an example of the education efforts. "We had a surgeon who thought he never had influenza. Those are the kinds of myths that we have to dispel to help people recognize that influenza can be mild or even asymptomatic in some people," but they are still able to spread it, she said.
Other steps included not charging for the shots, taking the shot cart to where people were working on the patient floors and having coworker colleagues administer the vaccine, offering vaccinations during annual physicals, and putting reminders about flu shots on the hospital’s computer system.
Also, "last year there was a request to provide vaccine to family members, and we actually did do that, as well, at various venues at various times." Dr. Chatterjee said that the key to the program’s success was that "we were responsive to the issues that were being raised. There was something that they saw changed as a result of bringing that issue to us," she said.
The hospital also updated the form employees must sign if they refused the vaccine.
"The original form just said ‘I refuse to have the vaccine’," Dr. Chatterjee said.
The updated one "made it very clear that if they refuse the vaccine, they are putting their patients at risk and putting their coworkers at risk," she said.
The form also requires employees to wear a surgical mask throughout the influenza season if they refuse the vaccine, even if they produce a medical exemption.
The updated refusal form states that "those who do not comply with the mask requirement will be subject to corrective action up to and including termination."
When employees ignored the order, "we would get reports from people saying, ‘You know xyz has not been vaccinated and is not wearing a mask’?"; everyone on the unit knew who was not vaccinated.
In those cases, infection control nurses would visit the workers and remind them that they had signed the document.
"A couple of people actually started [wearing the masks] and then realized what an onerous job wearing a mask for the entire shift was. Those medical exemptions quickly disappeared," Dr. Chatterjee said.
"It was pretty clear to us that there were some key changes that made big differences, and the mask issue was one of those," she said.
Dr. Andrew Pavia, chief of the division of infectious disease in the department of pediatrics at the University of Utah, Salt Lake City, noted that such voluntary programs go only so far in raising vaccination rates. "The best possible voluntary programs only get health care worker vaccination rates into the mid-80% range. This is the reason that so many health care organizations have endorsed mandatory immunization."
There isn’t an adequate percentage below 100% at which you stop transmission, and "so we need to target something that approaches 100%. It appears, right now, that mandatory immunization is the policy that works."
Dr. Chatterjee and Dr. Pavia reported having no conflicts of interest.
FROM THE ANNUAL MEETING OF THE INFECTIOUS DISEASES SOCIETY OF AMERICA
Major Finding: About 86% of health care workers at a hospital in Omaha, Neb., agreed to get a flu shot, without being mandated to do so. Commonsense approaches, like making shots available after hours for night workers, increased vaccination rates.
Data Source: An observational study looking at vaccination rates before and after policy changes.
Disclosures: Dr. Chatterjee and Dr. Pavia said they had no conflicts of interest.
Reports Claim FDA Blocking Importation of Cantharidin
Cantharidin, used for decades by pediatric dermatologists to treat molluscum contagiosum, has become hard to obtain, according to several dermatologists and a supplier.
Physicians suspect, and a Food and Drug Administration document suggests, the FDA is blocking importation of the agent.
Cantharidin has been a concern at the FDA since at least the late 1990s, when it was proposed for but not added to the list of bulk substances allowed by the agency to be compounded. The substance has never gone through formal agency review and approval.
According to Deborah Grafelman, president of Delasco Dermatologic Lab and Supply in Council Bluffs, Iowa, it was only recently that cantharidin imports from China have been blocked.
Her company once supplied cantharidin to about 700 clients, including physician offices, clinics, and hospitals, at a volume of about 2,500 vial sales per year, but this activity has stopped.
The FDA has "apparently been cracking down on it recently. We could get cantharidin from our supplier in 2008," but not since, she said.
Dr. Robert Sidbury, chief of the division of dermatology at Seattle Children's Hospital and the University of Washington, said he had been told by FDA staff that they encourage interested physicians to request an Investigational New Drug (IND) application, which would allow use of cantharidin as an investigational drug.
There was talk of doing just that at the Society for Pediatric Dermatology’s annual meeting in Portland, Ore., this summer.
Following a presentation by Dr. Sidbury that touched upon the situation, an audience member said "we need to take collective action and work together, not play victim. As subspecialists, we can prove it's in the best interests of patients ... even if it takes years to get an IND and do a study to show it's safe."
After repeated inquiries from Skin & Allergy News, the FDA did not provide any information on the situation, including how many shipments may have been blocked and if the agency has new safety concerns about cantharidin. Agency officials declined to comment.
However, a Jan. 7, 1999, Federal Register notice that proposed cantharidin for the bulk-substance list gave insight into the agency's concerns. It refers to cantharidin as an "an extremely toxic substance."
An Aug. 31, 2009, order blocking Delasco's importation of 0.1 kg noted, "The article appears to be a new drug without an approved new drug application."
The same issue was raised in an FDA Jan. 14, 2008, warning letter to Bellevue Pharmacy Solutions of St. Louis, Mo., which ordered the pharmacy to stop compounding cantharidin.
Because cantharidin is not an active ingredient in any FDA-approved drug, the "FDA does not sanction" its use in pharmacy compounding, the letter noted.
The agency "will not exercise enforcement discretion toward your firm's continued compounding of" cantharidin, however, failure to comply could result in "legal action without further notice, including, without limitation, seizure and injunction," according to the letter.
Officials from Bellevue Pharmacy Solutions declined to comment for this story.
Dermatologists are frustrated that a treatment they have relied on for years is no longer available.
"Used appropriately, it's effective and well tolerated," said Dr. Anthony J. Mancini, head of the division of pediatric dermatology at Northwestern University and Children's Memorial Hospital in Chicago, who used to purchase cantharidin from Delasco Dermatologic Lab and Supply.
"It's the best treatment for molluscum, successful in over 95% of our patients. We are just trying to offer patients what’s best for them, and now we can't even apply a little beetle juice to these common and often irritating lesions," Dr. Mancini said.
Last winter, Dr. Mancini and his colleagues discovered they could no longer get cantharidin, and became aware of the FDA's apparent ban on the product.
In a March 15 notice to "all faculty, trainees, [and] nurses in dermatology," he wrote, "as of today, we are no longer allowed to utilize cantharidin from any compounding pharmacy to treat molluscum."
Also on March 15, he asked the agency by e-mail "if it is possible to convene an FDA group to re-evaluate this agent."
The message included five references to studies over the past decade that show cantharidin to be a safe and effective molluscum treatment. Dr. Mancini said he has received no response to his message.
Cantharidin is a vesicant extracted from crushed Chinese blister beetles. Applied topically, it is absorbed into epidermal cell membranes, leading to acantholysis, intraepidermal blistering, and resolution of the lesion. Redness and transient burning sensations are among possible complications, though these effects are exceedingly rare. Use of the agent generally does not lead to scarring. However, severe blistering can occur with improper use; and ingestion, especially by children, can be fatal (Arch. Dermatol. 2001;137:1357-60).
"The raw material is very caustic and requires extreme safety precautions when handling, but once compounded with collodion, it becomes a very effective agent for use in dermatology," Ms. Grafelman of Delasco Dermatologic Lab and Supply noted.
Cantharidin is also being researched for a variety of other uses. Clinicaltrials.gov lists recently completed randomized, placebo-controlled trials for treating molluscum contagiosum in children with cantharidin and for treating cutaneous verucca vulgaris with cantharidin along with liquid nitrogen in adults.
Cantharidin has also recently been studied for its effects on cancer cells and parasites (Int. J. Oncol. 2010;37:1243-50; Exp. Parasitol. 2010;126:126-9 ).
Dr. Mancini and Dr. Sidbury said they have no financial interests in cantharidin.
Cantharidin, used for decades by pediatric dermatologists to treat molluscum contagiosum, has become hard to obtain, according to several dermatologists and a supplier.
Physicians suspect, and a Food and Drug Administration document suggests, the FDA is blocking importation of the agent.
Cantharidin has been a concern at the FDA since at least the late 1990s, when it was proposed for but not added to the list of bulk substances allowed by the agency to be compounded. The substance has never gone through formal agency review and approval.
According to Deborah Grafelman, president of Delasco Dermatologic Lab and Supply in Council Bluffs, Iowa, it was only recently that cantharidin imports from China have been blocked.
Her company once supplied cantharidin to about 700 clients, including physician offices, clinics, and hospitals, at a volume of about 2,500 vial sales per year, but this activity has stopped.
The FDA has "apparently been cracking down on it recently. We could get cantharidin from our supplier in 2008," but not since, she said.
Dr. Robert Sidbury, chief of the division of dermatology at Seattle Children's Hospital and the University of Washington, said he had been told by FDA staff that they encourage interested physicians to request an Investigational New Drug (IND) application, which would allow use of cantharidin as an investigational drug.
There was talk of doing just that at the Society for Pediatric Dermatology’s annual meeting in Portland, Ore., this summer.
Following a presentation by Dr. Sidbury that touched upon the situation, an audience member said "we need to take collective action and work together, not play victim. As subspecialists, we can prove it's in the best interests of patients ... even if it takes years to get an IND and do a study to show it's safe."
After repeated inquiries from Skin & Allergy News, the FDA did not provide any information on the situation, including how many shipments may have been blocked and if the agency has new safety concerns about cantharidin. Agency officials declined to comment.
However, a Jan. 7, 1999, Federal Register notice that proposed cantharidin for the bulk-substance list gave insight into the agency's concerns. It refers to cantharidin as an "an extremely toxic substance."
An Aug. 31, 2009, order blocking Delasco's importation of 0.1 kg noted, "The article appears to be a new drug without an approved new drug application."
The same issue was raised in an FDA Jan. 14, 2008, warning letter to Bellevue Pharmacy Solutions of St. Louis, Mo., which ordered the pharmacy to stop compounding cantharidin.
Because cantharidin is not an active ingredient in any FDA-approved drug, the "FDA does not sanction" its use in pharmacy compounding, the letter noted.
The agency "will not exercise enforcement discretion toward your firm's continued compounding of" cantharidin, however, failure to comply could result in "legal action without further notice, including, without limitation, seizure and injunction," according to the letter.
Officials from Bellevue Pharmacy Solutions declined to comment for this story.
Dermatologists are frustrated that a treatment they have relied on for years is no longer available.
"Used appropriately, it's effective and well tolerated," said Dr. Anthony J. Mancini, head of the division of pediatric dermatology at Northwestern University and Children's Memorial Hospital in Chicago, who used to purchase cantharidin from Delasco Dermatologic Lab and Supply.
"It's the best treatment for molluscum, successful in over 95% of our patients. We are just trying to offer patients what’s best for them, and now we can't even apply a little beetle juice to these common and often irritating lesions," Dr. Mancini said.
Last winter, Dr. Mancini and his colleagues discovered they could no longer get cantharidin, and became aware of the FDA's apparent ban on the product.
In a March 15 notice to "all faculty, trainees, [and] nurses in dermatology," he wrote, "as of today, we are no longer allowed to utilize cantharidin from any compounding pharmacy to treat molluscum."
Also on March 15, he asked the agency by e-mail "if it is possible to convene an FDA group to re-evaluate this agent."
The message included five references to studies over the past decade that show cantharidin to be a safe and effective molluscum treatment. Dr. Mancini said he has received no response to his message.
Cantharidin is a vesicant extracted from crushed Chinese blister beetles. Applied topically, it is absorbed into epidermal cell membranes, leading to acantholysis, intraepidermal blistering, and resolution of the lesion. Redness and transient burning sensations are among possible complications, though these effects are exceedingly rare. Use of the agent generally does not lead to scarring. However, severe blistering can occur with improper use; and ingestion, especially by children, can be fatal (Arch. Dermatol. 2001;137:1357-60).
"The raw material is very caustic and requires extreme safety precautions when handling, but once compounded with collodion, it becomes a very effective agent for use in dermatology," Ms. Grafelman of Delasco Dermatologic Lab and Supply noted.
Cantharidin is also being researched for a variety of other uses. Clinicaltrials.gov lists recently completed randomized, placebo-controlled trials for treating molluscum contagiosum in children with cantharidin and for treating cutaneous verucca vulgaris with cantharidin along with liquid nitrogen in adults.
Cantharidin has also recently been studied for its effects on cancer cells and parasites (Int. J. Oncol. 2010;37:1243-50; Exp. Parasitol. 2010;126:126-9 ).
Dr. Mancini and Dr. Sidbury said they have no financial interests in cantharidin.
Cantharidin, used for decades by pediatric dermatologists to treat molluscum contagiosum, has become hard to obtain, according to several dermatologists and a supplier.
Physicians suspect, and a Food and Drug Administration document suggests, the FDA is blocking importation of the agent.
Cantharidin has been a concern at the FDA since at least the late 1990s, when it was proposed for but not added to the list of bulk substances allowed by the agency to be compounded. The substance has never gone through formal agency review and approval.
According to Deborah Grafelman, president of Delasco Dermatologic Lab and Supply in Council Bluffs, Iowa, it was only recently that cantharidin imports from China have been blocked.
Her company once supplied cantharidin to about 700 clients, including physician offices, clinics, and hospitals, at a volume of about 2,500 vial sales per year, but this activity has stopped.
The FDA has "apparently been cracking down on it recently. We could get cantharidin from our supplier in 2008," but not since, she said.
Dr. Robert Sidbury, chief of the division of dermatology at Seattle Children's Hospital and the University of Washington, said he had been told by FDA staff that they encourage interested physicians to request an Investigational New Drug (IND) application, which would allow use of cantharidin as an investigational drug.
There was talk of doing just that at the Society for Pediatric Dermatology’s annual meeting in Portland, Ore., this summer.
Following a presentation by Dr. Sidbury that touched upon the situation, an audience member said "we need to take collective action and work together, not play victim. As subspecialists, we can prove it's in the best interests of patients ... even if it takes years to get an IND and do a study to show it's safe."
After repeated inquiries from Skin & Allergy News, the FDA did not provide any information on the situation, including how many shipments may have been blocked and if the agency has new safety concerns about cantharidin. Agency officials declined to comment.
However, a Jan. 7, 1999, Federal Register notice that proposed cantharidin for the bulk-substance list gave insight into the agency's concerns. It refers to cantharidin as an "an extremely toxic substance."
An Aug. 31, 2009, order blocking Delasco's importation of 0.1 kg noted, "The article appears to be a new drug without an approved new drug application."
The same issue was raised in an FDA Jan. 14, 2008, warning letter to Bellevue Pharmacy Solutions of St. Louis, Mo., which ordered the pharmacy to stop compounding cantharidin.
Because cantharidin is not an active ingredient in any FDA-approved drug, the "FDA does not sanction" its use in pharmacy compounding, the letter noted.
The agency "will not exercise enforcement discretion toward your firm's continued compounding of" cantharidin, however, failure to comply could result in "legal action without further notice, including, without limitation, seizure and injunction," according to the letter.
Officials from Bellevue Pharmacy Solutions declined to comment for this story.
Dermatologists are frustrated that a treatment they have relied on for years is no longer available.
"Used appropriately, it's effective and well tolerated," said Dr. Anthony J. Mancini, head of the division of pediatric dermatology at Northwestern University and Children's Memorial Hospital in Chicago, who used to purchase cantharidin from Delasco Dermatologic Lab and Supply.
"It's the best treatment for molluscum, successful in over 95% of our patients. We are just trying to offer patients what’s best for them, and now we can't even apply a little beetle juice to these common and often irritating lesions," Dr. Mancini said.
Last winter, Dr. Mancini and his colleagues discovered they could no longer get cantharidin, and became aware of the FDA's apparent ban on the product.
In a March 15 notice to "all faculty, trainees, [and] nurses in dermatology," he wrote, "as of today, we are no longer allowed to utilize cantharidin from any compounding pharmacy to treat molluscum."
Also on March 15, he asked the agency by e-mail "if it is possible to convene an FDA group to re-evaluate this agent."
The message included five references to studies over the past decade that show cantharidin to be a safe and effective molluscum treatment. Dr. Mancini said he has received no response to his message.
Cantharidin is a vesicant extracted from crushed Chinese blister beetles. Applied topically, it is absorbed into epidermal cell membranes, leading to acantholysis, intraepidermal blistering, and resolution of the lesion. Redness and transient burning sensations are among possible complications, though these effects are exceedingly rare. Use of the agent generally does not lead to scarring. However, severe blistering can occur with improper use; and ingestion, especially by children, can be fatal (Arch. Dermatol. 2001;137:1357-60).
"The raw material is very caustic and requires extreme safety precautions when handling, but once compounded with collodion, it becomes a very effective agent for use in dermatology," Ms. Grafelman of Delasco Dermatologic Lab and Supply noted.
Cantharidin is also being researched for a variety of other uses. Clinicaltrials.gov lists recently completed randomized, placebo-controlled trials for treating molluscum contagiosum in children with cantharidin and for treating cutaneous verucca vulgaris with cantharidin along with liquid nitrogen in adults.
Cantharidin has also recently been studied for its effects on cancer cells and parasites (Int. J. Oncol. 2010;37:1243-50; Exp. Parasitol. 2010;126:126-9 ).
Dr. Mancini and Dr. Sidbury said they have no financial interests in cantharidin.
FDA Apparently Blocking Importation of Cantharidin
Cantharidin, used for decades by pediatric dermatologists to treat molluscum contagiosum, has become hard to obtain, according to several dermatologists and a supplier.
Physicians suspect, and a Food and Drug Administration document suggests, the FDA is blocking importation of the agent.
Cantharidin has been a concern at the FDA since at least the late 1990s, when it was proposed for but not added to the list of bulk substances allowed by the agency to be compounded. The substance has never gone through formal agency review and approval.
According to Deborah Grafelman, president of Delasco Dermatologic Lab and Supply in Council Bluffs, Iowa, it was only recently that cantharidin imports from China have been blocked.
Her company once supplied cantharidin to about 700 clients, including physician offices, clinics, and hospitals, at a volume of about 2,500 vial sales per year, but this activity has stopped.
The FDA has "apparently been cracking down on it recently. We could get cantharidin from our supplier in 2008," but not since, she said.
Dr. Robert Sidbury, chief of the division of dermatology at Seattle Children's Hospital and the University of Washington, said he had been told by FDA staff that they encourage interested physicians to request an Investigational New Drug (IND) application, which would allow use of cantharidin as an investigational drug.
There was talk of doing just that at the Society for Pediatric Dermatology’s annual meeting in Portland, Ore., this summer.
Following a presentation by Dr. Sidbury that touched upon the situation, an audience member said "we need to take collective action and work together, not play victim. As subspecialists, we can prove it's in the best interests of patients ... even if it takes years to get an IND and do a study to show it's safe."
After repeated inquiries from Internal Medicine News, the FDA did not provide any information on the situation, including how many shipments may have been blocked and if the agency has new safety concerns about cantharidin. Agency officials declined to comment.
However, a Jan. 7, 1999, Federal Register notice that proposed cantharidin for the bulk-substance list gave insight into the agency's concerns. It refers to cantharidin as an "an extremely toxic substance."
An Aug. 31, 2009, order blocking Delasco's importation of 0.1 kg noted, "The article appears to be a new drug without an approved new drug application."
The same issue was raised in an FDA Jan. 14, 2008, warning letter to Bellevue Pharmacy Solutions of St. Louis, Mo., which ordered the pharmacy to stop compounding cantharidin.
Because cantharidin is not an active ingredient in any FDA-approved drug, the "FDA does not sanction" its use in pharmacy compounding, the letter noted.
The agency "will not exercise enforcement discretion toward your firm's continued compounding of" cantharidin, however, failure to comply could result in "legal action without further notice, including, without limitation, seizure and injunction," according to the letter.
Officials from Bellevue Pharmacy Solutions declined to comment for this story.
Dermatologists are frustrated that a treatment they have relied on for years is no longer available.
"Used appropriately, it's effective and well tolerated," said Dr. Anthony J. Mancini, head of the division of pediatric dermatology at Northwestern University and Children's Memorial Hospital in Chicago, who used to purchase cantharidin from Delasco Dermatologic Lab and Supply.
"It's the best treatment for molluscum, successful in over 95% of our patients. We are just trying to offer patients what’s best for them, and now we can't even apply a little beetle juice to these common and often irritating lesions," Dr. Mancini said.
Last winter, Dr. Mancini and his colleagues discovered they could no longer get cantharidin, and became aware of the FDA's apparent ban on the product.
In a March 15 notice to "all faculty, trainees, [and] nurses in dermatology," he wrote, "as of today, we are no longer allowed to utilize cantharidin from any compounding pharmacy to treat molluscum."
Also on March 15, he asked the agency by e-mail "if it is possible to convene an FDA group to re-evaluate this agent."
The message included five references to studies over the past decade that show cantharidin to be a safe and effective molluscum treatment. Dr. Mancini said he has received no response to his message.
Cantharidin is a vesicant extracted from crushed Chinese blister beetles. Applied topically, it is absorbed into epidermal cell membranes, leading to acantholysis, intraepidermal blistering, and resolution of the lesion. Redness and transient burning sensations are among possible complications, though these effects are exceedingly rare. Use of the agent generally does not lead to scarring. However, severe blistering can occur with improper use; and ingestion, especially by children, can be fatal (Arch. Dermatol. 2001;137:1357-60).
"The raw material is very caustic and requires extreme safety precautions when handling, but once compounded with collodion, it becomes a very effective agent for use in dermatology," Ms. Grafelman of Delasco Dermatologic Lab and Supply noted.
Cantharidin is also being researched for a variety of other uses. Clinicaltrials.gov lists recently completed randomized, placebo-controlled trials for treating molluscum contagiosum in children with cantharidin and for treating cutaneous verucca vulgaris with cantharidin along with liquid nitrogen in adults.
Cantharidin has also recently been studied for its effects on cancer cells and parasites (Int. J. Oncol. 2010;37:1243-50; Exp. Parasitol. 2010;126:126-9 ).
Dr. Mancini and Dr. Sidbury said they have no financial interests in cantharidin.
Cantharidin, used for decades by pediatric dermatologists to treat molluscum contagiosum, has become hard to obtain, according to several dermatologists and a supplier.
Physicians suspect, and a Food and Drug Administration document suggests, the FDA is blocking importation of the agent.
Cantharidin has been a concern at the FDA since at least the late 1990s, when it was proposed for but not added to the list of bulk substances allowed by the agency to be compounded. The substance has never gone through formal agency review and approval.
According to Deborah Grafelman, president of Delasco Dermatologic Lab and Supply in Council Bluffs, Iowa, it was only recently that cantharidin imports from China have been blocked.
Her company once supplied cantharidin to about 700 clients, including physician offices, clinics, and hospitals, at a volume of about 2,500 vial sales per year, but this activity has stopped.
The FDA has "apparently been cracking down on it recently. We could get cantharidin from our supplier in 2008," but not since, she said.
Dr. Robert Sidbury, chief of the division of dermatology at Seattle Children's Hospital and the University of Washington, said he had been told by FDA staff that they encourage interested physicians to request an Investigational New Drug (IND) application, which would allow use of cantharidin as an investigational drug.
There was talk of doing just that at the Society for Pediatric Dermatology’s annual meeting in Portland, Ore., this summer.
Following a presentation by Dr. Sidbury that touched upon the situation, an audience member said "we need to take collective action and work together, not play victim. As subspecialists, we can prove it's in the best interests of patients ... even if it takes years to get an IND and do a study to show it's safe."
After repeated inquiries from Internal Medicine News, the FDA did not provide any information on the situation, including how many shipments may have been blocked and if the agency has new safety concerns about cantharidin. Agency officials declined to comment.
However, a Jan. 7, 1999, Federal Register notice that proposed cantharidin for the bulk-substance list gave insight into the agency's concerns. It refers to cantharidin as an "an extremely toxic substance."
An Aug. 31, 2009, order blocking Delasco's importation of 0.1 kg noted, "The article appears to be a new drug without an approved new drug application."
The same issue was raised in an FDA Jan. 14, 2008, warning letter to Bellevue Pharmacy Solutions of St. Louis, Mo., which ordered the pharmacy to stop compounding cantharidin.
Because cantharidin is not an active ingredient in any FDA-approved drug, the "FDA does not sanction" its use in pharmacy compounding, the letter noted.
The agency "will not exercise enforcement discretion toward your firm's continued compounding of" cantharidin, however, failure to comply could result in "legal action without further notice, including, without limitation, seizure and injunction," according to the letter.
Officials from Bellevue Pharmacy Solutions declined to comment for this story.
Dermatologists are frustrated that a treatment they have relied on for years is no longer available.
"Used appropriately, it's effective and well tolerated," said Dr. Anthony J. Mancini, head of the division of pediatric dermatology at Northwestern University and Children's Memorial Hospital in Chicago, who used to purchase cantharidin from Delasco Dermatologic Lab and Supply.
"It's the best treatment for molluscum, successful in over 95% of our patients. We are just trying to offer patients what’s best for them, and now we can't even apply a little beetle juice to these common and often irritating lesions," Dr. Mancini said.
Last winter, Dr. Mancini and his colleagues discovered they could no longer get cantharidin, and became aware of the FDA's apparent ban on the product.
In a March 15 notice to "all faculty, trainees, [and] nurses in dermatology," he wrote, "as of today, we are no longer allowed to utilize cantharidin from any compounding pharmacy to treat molluscum."
Also on March 15, he asked the agency by e-mail "if it is possible to convene an FDA group to re-evaluate this agent."
The message included five references to studies over the past decade that show cantharidin to be a safe and effective molluscum treatment. Dr. Mancini said he has received no response to his message.
Cantharidin is a vesicant extracted from crushed Chinese blister beetles. Applied topically, it is absorbed into epidermal cell membranes, leading to acantholysis, intraepidermal blistering, and resolution of the lesion. Redness and transient burning sensations are among possible complications, though these effects are exceedingly rare. Use of the agent generally does not lead to scarring. However, severe blistering can occur with improper use; and ingestion, especially by children, can be fatal (Arch. Dermatol. 2001;137:1357-60).
"The raw material is very caustic and requires extreme safety precautions when handling, but once compounded with collodion, it becomes a very effective agent for use in dermatology," Ms. Grafelman of Delasco Dermatologic Lab and Supply noted.
Cantharidin is also being researched for a variety of other uses. Clinicaltrials.gov lists recently completed randomized, placebo-controlled trials for treating molluscum contagiosum in children with cantharidin and for treating cutaneous verucca vulgaris with cantharidin along with liquid nitrogen in adults.
Cantharidin has also recently been studied for its effects on cancer cells and parasites (Int. J. Oncol. 2010;37:1243-50; Exp. Parasitol. 2010;126:126-9 ).
Dr. Mancini and Dr. Sidbury said they have no financial interests in cantharidin.
Cantharidin, used for decades by pediatric dermatologists to treat molluscum contagiosum, has become hard to obtain, according to several dermatologists and a supplier.
Physicians suspect, and a Food and Drug Administration document suggests, the FDA is blocking importation of the agent.
Cantharidin has been a concern at the FDA since at least the late 1990s, when it was proposed for but not added to the list of bulk substances allowed by the agency to be compounded. The substance has never gone through formal agency review and approval.
According to Deborah Grafelman, president of Delasco Dermatologic Lab and Supply in Council Bluffs, Iowa, it was only recently that cantharidin imports from China have been blocked.
Her company once supplied cantharidin to about 700 clients, including physician offices, clinics, and hospitals, at a volume of about 2,500 vial sales per year, but this activity has stopped.
The FDA has "apparently been cracking down on it recently. We could get cantharidin from our supplier in 2008," but not since, she said.
Dr. Robert Sidbury, chief of the division of dermatology at Seattle Children's Hospital and the University of Washington, said he had been told by FDA staff that they encourage interested physicians to request an Investigational New Drug (IND) application, which would allow use of cantharidin as an investigational drug.
There was talk of doing just that at the Society for Pediatric Dermatology’s annual meeting in Portland, Ore., this summer.
Following a presentation by Dr. Sidbury that touched upon the situation, an audience member said "we need to take collective action and work together, not play victim. As subspecialists, we can prove it's in the best interests of patients ... even if it takes years to get an IND and do a study to show it's safe."
After repeated inquiries from Internal Medicine News, the FDA did not provide any information on the situation, including how many shipments may have been blocked and if the agency has new safety concerns about cantharidin. Agency officials declined to comment.
However, a Jan. 7, 1999, Federal Register notice that proposed cantharidin for the bulk-substance list gave insight into the agency's concerns. It refers to cantharidin as an "an extremely toxic substance."
An Aug. 31, 2009, order blocking Delasco's importation of 0.1 kg noted, "The article appears to be a new drug without an approved new drug application."
The same issue was raised in an FDA Jan. 14, 2008, warning letter to Bellevue Pharmacy Solutions of St. Louis, Mo., which ordered the pharmacy to stop compounding cantharidin.
Because cantharidin is not an active ingredient in any FDA-approved drug, the "FDA does not sanction" its use in pharmacy compounding, the letter noted.
The agency "will not exercise enforcement discretion toward your firm's continued compounding of" cantharidin, however, failure to comply could result in "legal action without further notice, including, without limitation, seizure and injunction," according to the letter.
Officials from Bellevue Pharmacy Solutions declined to comment for this story.
Dermatologists are frustrated that a treatment they have relied on for years is no longer available.
"Used appropriately, it's effective and well tolerated," said Dr. Anthony J. Mancini, head of the division of pediatric dermatology at Northwestern University and Children's Memorial Hospital in Chicago, who used to purchase cantharidin from Delasco Dermatologic Lab and Supply.
"It's the best treatment for molluscum, successful in over 95% of our patients. We are just trying to offer patients what’s best for them, and now we can't even apply a little beetle juice to these common and often irritating lesions," Dr. Mancini said.
Last winter, Dr. Mancini and his colleagues discovered they could no longer get cantharidin, and became aware of the FDA's apparent ban on the product.
In a March 15 notice to "all faculty, trainees, [and] nurses in dermatology," he wrote, "as of today, we are no longer allowed to utilize cantharidin from any compounding pharmacy to treat molluscum."
Also on March 15, he asked the agency by e-mail "if it is possible to convene an FDA group to re-evaluate this agent."
The message included five references to studies over the past decade that show cantharidin to be a safe and effective molluscum treatment. Dr. Mancini said he has received no response to his message.
Cantharidin is a vesicant extracted from crushed Chinese blister beetles. Applied topically, it is absorbed into epidermal cell membranes, leading to acantholysis, intraepidermal blistering, and resolution of the lesion. Redness and transient burning sensations are among possible complications, though these effects are exceedingly rare. Use of the agent generally does not lead to scarring. However, severe blistering can occur with improper use; and ingestion, especially by children, can be fatal (Arch. Dermatol. 2001;137:1357-60).
"The raw material is very caustic and requires extreme safety precautions when handling, but once compounded with collodion, it becomes a very effective agent for use in dermatology," Ms. Grafelman of Delasco Dermatologic Lab and Supply noted.
Cantharidin is also being researched for a variety of other uses. Clinicaltrials.gov lists recently completed randomized, placebo-controlled trials for treating molluscum contagiosum in children with cantharidin and for treating cutaneous verucca vulgaris with cantharidin along with liquid nitrogen in adults.
Cantharidin has also recently been studied for its effects on cancer cells and parasites (Int. J. Oncol. 2010;37:1243-50; Exp. Parasitol. 2010;126:126-9 ).
Dr. Mancini and Dr. Sidbury said they have no financial interests in cantharidin.
Select Criteria Denote High-Risk SLE Pregnancies
VANCOUVER, B.C. — Monthly monitoring by rheumatologists of every pregnancy in every woman with systemic lupus erythematosus may be unnecessary, according to Dr. Michelle Petri.
A relatively small list of criteria can distinguish high-risk pregnancies in women with systemic lupus erythematosus (SLE) – ones that carry a higher likelihood of miscarriage, extreme prematurity, and SLE flare – from others, and signal the need for intensive monitoring by obstetricians and rheumatologists, Dr. Petri said at the meeting.
At present, however, there is little effort to make such distinctions, so most SLE pregnancies are subjected to monthly visits to rheumatologists and obstetricians, and, starting at week 26, weekly monitoring by obstetricians.
That's not always necessary; women are subjected to needless anxiety and hospital resources are wasted, Dr. Petri said.
Based on the Hopkins Lupus Cohort, a database that has been tracking several thousand patients with SLE over the past 25 years, Dr. Petri and her colleague, Duke University rheumatologist Dr. Megan Clowse, have identified those factors that truly put women and fetuses at risk during SLE pregnancies.
Pregnancy and the postpartum period are hard on the kidneys of women with SLE, though organ involvement elsewhere in the body tends to lessen, said Dr. Petri, professor of rheumatology at Johns Hopkins University, Baltimore.
“Proteinuria from active lupus significantly increases, and this continues even after delivery,” she added.
Therefore, pregnant women with lupus nephritis truly do need close monitoring. Dr. Petri recommended monthly urine protein-creatinine ratios to detect a worsening of the condition and the need for treatment.
She noted that the ranges on urine dipsticks are too broad; the dipstick is not adequate as a monitoring tool for nephritis.
In terms of fetal health, the risk of miscarriage doubles if, at the first pregnancy visit, a woman is proteinuric, thrombocytopenic, or hypertensive, or has a history of antiphospholipid syndrome.
The risk triples if two or more of these conditions are present, Dr. Petri said. The presence of antithyroid antibodies also increases the risk of miscarriage.
In addition, active SLE, especially if accompanied by anti–double-stranded DNA antibody or low complement levels, predicts extreme prematurity. Autoimmune thyroid disease also appears to be associated with preterm birth.
Screening for the various factors, “we can predict at the first pregnancy visit if there's going to be a poor outcome,” Dr. Petri said.
If the risk factors are present, monthly monitoring by a high-risk obstetrician, followed by weekly monitoring at week 26, are appropriate to gauge if, and when, a rescue delivery is needed.
Otherwise, and absent renal involvement in the pregnant patient, SLE pregnancies may not need to be classified as high risk, Dr. Petri said.
“Since we can stratify women at risk for miscarriage and extreme prematurity, and know the only organ we have to worry about is the kidney, we can come closer to using our resources appropriately,” Dr. Petri said.
To reassure women, rheumatologists should “get the word out to patients that high-risk interventions are not necessary for every [SLE pregnancy],” she said.
Dr. Petri said she had no disclosures to report.
VANCOUVER, B.C. — Monthly monitoring by rheumatologists of every pregnancy in every woman with systemic lupus erythematosus may be unnecessary, according to Dr. Michelle Petri.
A relatively small list of criteria can distinguish high-risk pregnancies in women with systemic lupus erythematosus (SLE) – ones that carry a higher likelihood of miscarriage, extreme prematurity, and SLE flare – from others, and signal the need for intensive monitoring by obstetricians and rheumatologists, Dr. Petri said at the meeting.
At present, however, there is little effort to make such distinctions, so most SLE pregnancies are subjected to monthly visits to rheumatologists and obstetricians, and, starting at week 26, weekly monitoring by obstetricians.
That's not always necessary; women are subjected to needless anxiety and hospital resources are wasted, Dr. Petri said.
Based on the Hopkins Lupus Cohort, a database that has been tracking several thousand patients with SLE over the past 25 years, Dr. Petri and her colleague, Duke University rheumatologist Dr. Megan Clowse, have identified those factors that truly put women and fetuses at risk during SLE pregnancies.
Pregnancy and the postpartum period are hard on the kidneys of women with SLE, though organ involvement elsewhere in the body tends to lessen, said Dr. Petri, professor of rheumatology at Johns Hopkins University, Baltimore.
“Proteinuria from active lupus significantly increases, and this continues even after delivery,” she added.
Therefore, pregnant women with lupus nephritis truly do need close monitoring. Dr. Petri recommended monthly urine protein-creatinine ratios to detect a worsening of the condition and the need for treatment.
She noted that the ranges on urine dipsticks are too broad; the dipstick is not adequate as a monitoring tool for nephritis.
In terms of fetal health, the risk of miscarriage doubles if, at the first pregnancy visit, a woman is proteinuric, thrombocytopenic, or hypertensive, or has a history of antiphospholipid syndrome.
The risk triples if two or more of these conditions are present, Dr. Petri said. The presence of antithyroid antibodies also increases the risk of miscarriage.
In addition, active SLE, especially if accompanied by anti–double-stranded DNA antibody or low complement levels, predicts extreme prematurity. Autoimmune thyroid disease also appears to be associated with preterm birth.
Screening for the various factors, “we can predict at the first pregnancy visit if there's going to be a poor outcome,” Dr. Petri said.
If the risk factors are present, monthly monitoring by a high-risk obstetrician, followed by weekly monitoring at week 26, are appropriate to gauge if, and when, a rescue delivery is needed.
Otherwise, and absent renal involvement in the pregnant patient, SLE pregnancies may not need to be classified as high risk, Dr. Petri said.
“Since we can stratify women at risk for miscarriage and extreme prematurity, and know the only organ we have to worry about is the kidney, we can come closer to using our resources appropriately,” Dr. Petri said.
To reassure women, rheumatologists should “get the word out to patients that high-risk interventions are not necessary for every [SLE pregnancy],” she said.
Dr. Petri said she had no disclosures to report.
VANCOUVER, B.C. — Monthly monitoring by rheumatologists of every pregnancy in every woman with systemic lupus erythematosus may be unnecessary, according to Dr. Michelle Petri.
A relatively small list of criteria can distinguish high-risk pregnancies in women with systemic lupus erythematosus (SLE) – ones that carry a higher likelihood of miscarriage, extreme prematurity, and SLE flare – from others, and signal the need for intensive monitoring by obstetricians and rheumatologists, Dr. Petri said at the meeting.
At present, however, there is little effort to make such distinctions, so most SLE pregnancies are subjected to monthly visits to rheumatologists and obstetricians, and, starting at week 26, weekly monitoring by obstetricians.
That's not always necessary; women are subjected to needless anxiety and hospital resources are wasted, Dr. Petri said.
Based on the Hopkins Lupus Cohort, a database that has been tracking several thousand patients with SLE over the past 25 years, Dr. Petri and her colleague, Duke University rheumatologist Dr. Megan Clowse, have identified those factors that truly put women and fetuses at risk during SLE pregnancies.
Pregnancy and the postpartum period are hard on the kidneys of women with SLE, though organ involvement elsewhere in the body tends to lessen, said Dr. Petri, professor of rheumatology at Johns Hopkins University, Baltimore.
“Proteinuria from active lupus significantly increases, and this continues even after delivery,” she added.
Therefore, pregnant women with lupus nephritis truly do need close monitoring. Dr. Petri recommended monthly urine protein-creatinine ratios to detect a worsening of the condition and the need for treatment.
She noted that the ranges on urine dipsticks are too broad; the dipstick is not adequate as a monitoring tool for nephritis.
In terms of fetal health, the risk of miscarriage doubles if, at the first pregnancy visit, a woman is proteinuric, thrombocytopenic, or hypertensive, or has a history of antiphospholipid syndrome.
The risk triples if two or more of these conditions are present, Dr. Petri said. The presence of antithyroid antibodies also increases the risk of miscarriage.
In addition, active SLE, especially if accompanied by anti–double-stranded DNA antibody or low complement levels, predicts extreme prematurity. Autoimmune thyroid disease also appears to be associated with preterm birth.
Screening for the various factors, “we can predict at the first pregnancy visit if there's going to be a poor outcome,” Dr. Petri said.
If the risk factors are present, monthly monitoring by a high-risk obstetrician, followed by weekly monitoring at week 26, are appropriate to gauge if, and when, a rescue delivery is needed.
Otherwise, and absent renal involvement in the pregnant patient, SLE pregnancies may not need to be classified as high risk, Dr. Petri said.
“Since we can stratify women at risk for miscarriage and extreme prematurity, and know the only organ we have to worry about is the kidney, we can come closer to using our resources appropriately,” Dr. Petri said.
To reassure women, rheumatologists should “get the word out to patients that high-risk interventions are not necessary for every [SLE pregnancy],” she said.
Dr. Petri said she had no disclosures to report.
From the International Congress on Systemic Lupus Erythematosus
Risks for Flu Admissions Examined
VANCOUVER, B.C. – The risk that a child will be hospitalized with influenza more than doubles if more than half of the household members smoke, maternal age is below 22 years, or household income is below the poverty line.
However, the risk is halved if children are breastfed for 6 months or more, or if caregivers are immunized against the influenza if their children are not, according to Dr. Nila Dharan, who is with New York University's division of infectious diseases.
Dr. Dharan and her colleagues compared the medical and demographic histories of 290 children aged 6-59 months hospitalized with influenza with 1,089 age- and zip code–matched children who were not hospitalized with influenza.
Although most of the risk and protective factors are known, “This study is the largest to date examining the risk of childhood exposure to cigarette smoke and hospitalization for influenza and highlights the importance of reducing children's environmental exposure to smoke, and the need for vaccination of both the child and their household contacts,” she said.
Among other findings, children not current with their vaccinations had a greater risk of influenza hospitalization (odds ratio 1.7), highlighting “the benefits of routine childhood vaccinations which may reduce the risk of secondary bacterial infections,” Dr. Dharan said at the meeting.
Of all the risk factors, however, preexisting medical conditions were the most significant, including pulmonary and neurologic conditions, but especially hematologic or oncologic ones (OR 12.1).
The study found a statistically significant trend toward greater protection the longer children are breastfed. Breastfeeding for their first year or longer was protective for children aged 2-5 years (OR 0.5); not being breastfed increased the risk of influenza-hospitalization (OR 1.7).
Day care did not increase influenza hospitalization risk (OR 1.1), but being a boy did (OR 1.3).
The study culled information during the 2005-2008 influenza seasons from 10 sites in the Center for Disease Control and Prevention's Emerging Infections Program.
Dr. Dharan said that she has no disclosures.
VANCOUVER, B.C. – The risk that a child will be hospitalized with influenza more than doubles if more than half of the household members smoke, maternal age is below 22 years, or household income is below the poverty line.
However, the risk is halved if children are breastfed for 6 months or more, or if caregivers are immunized against the influenza if their children are not, according to Dr. Nila Dharan, who is with New York University's division of infectious diseases.
Dr. Dharan and her colleagues compared the medical and demographic histories of 290 children aged 6-59 months hospitalized with influenza with 1,089 age- and zip code–matched children who were not hospitalized with influenza.
Although most of the risk and protective factors are known, “This study is the largest to date examining the risk of childhood exposure to cigarette smoke and hospitalization for influenza and highlights the importance of reducing children's environmental exposure to smoke, and the need for vaccination of both the child and their household contacts,” she said.
Among other findings, children not current with their vaccinations had a greater risk of influenza hospitalization (odds ratio 1.7), highlighting “the benefits of routine childhood vaccinations which may reduce the risk of secondary bacterial infections,” Dr. Dharan said at the meeting.
Of all the risk factors, however, preexisting medical conditions were the most significant, including pulmonary and neurologic conditions, but especially hematologic or oncologic ones (OR 12.1).
The study found a statistically significant trend toward greater protection the longer children are breastfed. Breastfeeding for their first year or longer was protective for children aged 2-5 years (OR 0.5); not being breastfed increased the risk of influenza-hospitalization (OR 1.7).
Day care did not increase influenza hospitalization risk (OR 1.1), but being a boy did (OR 1.3).
The study culled information during the 2005-2008 influenza seasons from 10 sites in the Center for Disease Control and Prevention's Emerging Infections Program.
Dr. Dharan said that she has no disclosures.
VANCOUVER, B.C. – The risk that a child will be hospitalized with influenza more than doubles if more than half of the household members smoke, maternal age is below 22 years, or household income is below the poverty line.
However, the risk is halved if children are breastfed for 6 months or more, or if caregivers are immunized against the influenza if their children are not, according to Dr. Nila Dharan, who is with New York University's division of infectious diseases.
Dr. Dharan and her colleagues compared the medical and demographic histories of 290 children aged 6-59 months hospitalized with influenza with 1,089 age- and zip code–matched children who were not hospitalized with influenza.
Although most of the risk and protective factors are known, “This study is the largest to date examining the risk of childhood exposure to cigarette smoke and hospitalization for influenza and highlights the importance of reducing children's environmental exposure to smoke, and the need for vaccination of both the child and their household contacts,” she said.
Among other findings, children not current with their vaccinations had a greater risk of influenza hospitalization (odds ratio 1.7), highlighting “the benefits of routine childhood vaccinations which may reduce the risk of secondary bacterial infections,” Dr. Dharan said at the meeting.
Of all the risk factors, however, preexisting medical conditions were the most significant, including pulmonary and neurologic conditions, but especially hematologic or oncologic ones (OR 12.1).
The study found a statistically significant trend toward greater protection the longer children are breastfed. Breastfeeding for their first year or longer was protective for children aged 2-5 years (OR 0.5); not being breastfed increased the risk of influenza-hospitalization (OR 1.7).
Day care did not increase influenza hospitalization risk (OR 1.1), but being a boy did (OR 1.3).
The study culled information during the 2005-2008 influenza seasons from 10 sites in the Center for Disease Control and Prevention's Emerging Infections Program.
Dr. Dharan said that she has no disclosures.