User login
Study: COVID-19 can kill months after infection
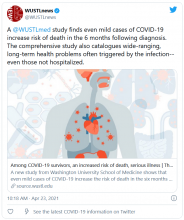
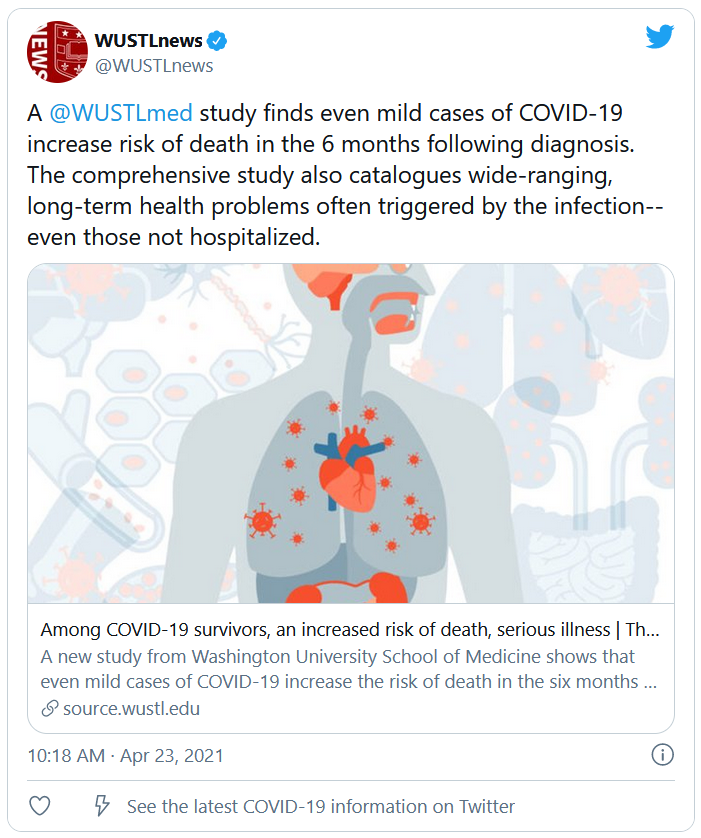
Long-haul COVID-19 patients face many health threats – including a higher chance of dying – up to 6 months after they catch the virus, according to a massive study published in the journal Nature.
Researchers examined more than 87,000 COVID-19 patients and nearly 5 million control patients in a federal database. They found COVID-19 patients had a 59% higher risk of death up to 6 months after infection, compared with noninfected people.
Those findings translate into about 8 extra deaths per 1,000 patients over 6 months, because many deaths caused by long-term COVID complications are not recorded as COVID-19 deaths, the researchers said. Among patients who were hospitalized and died after more than 30 days, there were 29 excess deaths per 1,000 patients over 6 months.
“As far as total pandemic death toll, these numbers suggest that the deaths we’re counting due to the immediate viral infection are only the tip of the iceberg,” Ziyad Al-Aly, MD, the senior author of the study and a director of the Clinical Epidemiology Center at the Veterans Affairs St. Louis Health Care System, said in a news release from the Washington University, St. Louis.
Johns Hopkins University in Baltimore says more than 3 million people worldwide and about 570,000 people in the United States have died of coronavirus-related reasons.
Long-haul COVID patients also had a much higher chance of getting sick, and not just in the respiratory system, according to the study.
The patients had a high rate of stroke and other nervous system ailments, mental health problems such as depression, the onset of diabetes, heart disease and other coronary problems, diarrhea and digestive disorders, kidney disease, blood clots, joint pain, hair loss, and general fatigue.
Patients often had clusters of these ailments. And the more severe the case of COVID-19, the higher the chance of long-term health problems, the study said.
Researchers based their study on health care databases of the U.S. Department of Veterans Affairs. Besides the 87,000 COVID patients, the database included about 5 million patients who didn’t catch COVID. The veterans in the study were about 88% men, but the large sample size included 8,880 women with confirmed cases, the news release said.
Dr. Al-Aly, an assistant professor at Washington University, said the study shows that long-haul COVID-19 could be “America’s next big health crisis.”
“Our study demonstrates that, up to 6 months after diagnosis, the risk of death following even a mild case of COVID-19 is not trivial and increases with disease severity,” he said. “Given that more than 30 million Americans have been infected with this virus, and given that the burden of long COVID-19 is substantial, the lingering effects of this disease will reverberate for many years and even decades.”
A version of this article first appeared on WebMD.com.
Long-haul COVID-19 patients face many health threats – including a higher chance of dying – up to 6 months after they catch the virus, according to a massive study published in the journal Nature.
Researchers examined more than 87,000 COVID-19 patients and nearly 5 million control patients in a federal database. They found COVID-19 patients had a 59% higher risk of death up to 6 months after infection, compared with noninfected people.
Those findings translate into about 8 extra deaths per 1,000 patients over 6 months, because many deaths caused by long-term COVID complications are not recorded as COVID-19 deaths, the researchers said. Among patients who were hospitalized and died after more than 30 days, there were 29 excess deaths per 1,000 patients over 6 months.
“As far as total pandemic death toll, these numbers suggest that the deaths we’re counting due to the immediate viral infection are only the tip of the iceberg,” Ziyad Al-Aly, MD, the senior author of the study and a director of the Clinical Epidemiology Center at the Veterans Affairs St. Louis Health Care System, said in a news release from the Washington University, St. Louis.
Johns Hopkins University in Baltimore says more than 3 million people worldwide and about 570,000 people in the United States have died of coronavirus-related reasons.
Long-haul COVID patients also had a much higher chance of getting sick, and not just in the respiratory system, according to the study.
The patients had a high rate of stroke and other nervous system ailments, mental health problems such as depression, the onset of diabetes, heart disease and other coronary problems, diarrhea and digestive disorders, kidney disease, blood clots, joint pain, hair loss, and general fatigue.
Patients often had clusters of these ailments. And the more severe the case of COVID-19, the higher the chance of long-term health problems, the study said.
Researchers based their study on health care databases of the U.S. Department of Veterans Affairs. Besides the 87,000 COVID patients, the database included about 5 million patients who didn’t catch COVID. The veterans in the study were about 88% men, but the large sample size included 8,880 women with confirmed cases, the news release said.
Dr. Al-Aly, an assistant professor at Washington University, said the study shows that long-haul COVID-19 could be “America’s next big health crisis.”
“Our study demonstrates that, up to 6 months after diagnosis, the risk of death following even a mild case of COVID-19 is not trivial and increases with disease severity,” he said. “Given that more than 30 million Americans have been infected with this virus, and given that the burden of long COVID-19 is substantial, the lingering effects of this disease will reverberate for many years and even decades.”
A version of this article first appeared on WebMD.com.
Long-haul COVID-19 patients face many health threats – including a higher chance of dying – up to 6 months after they catch the virus, according to a massive study published in the journal Nature.
Researchers examined more than 87,000 COVID-19 patients and nearly 5 million control patients in a federal database. They found COVID-19 patients had a 59% higher risk of death up to 6 months after infection, compared with noninfected people.
Those findings translate into about 8 extra deaths per 1,000 patients over 6 months, because many deaths caused by long-term COVID complications are not recorded as COVID-19 deaths, the researchers said. Among patients who were hospitalized and died after more than 30 days, there were 29 excess deaths per 1,000 patients over 6 months.
“As far as total pandemic death toll, these numbers suggest that the deaths we’re counting due to the immediate viral infection are only the tip of the iceberg,” Ziyad Al-Aly, MD, the senior author of the study and a director of the Clinical Epidemiology Center at the Veterans Affairs St. Louis Health Care System, said in a news release from the Washington University, St. Louis.
Johns Hopkins University in Baltimore says more than 3 million people worldwide and about 570,000 people in the United States have died of coronavirus-related reasons.
Long-haul COVID patients also had a much higher chance of getting sick, and not just in the respiratory system, according to the study.
The patients had a high rate of stroke and other nervous system ailments, mental health problems such as depression, the onset of diabetes, heart disease and other coronary problems, diarrhea and digestive disorders, kidney disease, blood clots, joint pain, hair loss, and general fatigue.
Patients often had clusters of these ailments. And the more severe the case of COVID-19, the higher the chance of long-term health problems, the study said.
Researchers based their study on health care databases of the U.S. Department of Veterans Affairs. Besides the 87,000 COVID patients, the database included about 5 million patients who didn’t catch COVID. The veterans in the study were about 88% men, but the large sample size included 8,880 women with confirmed cases, the news release said.
Dr. Al-Aly, an assistant professor at Washington University, said the study shows that long-haul COVID-19 could be “America’s next big health crisis.”
“Our study demonstrates that, up to 6 months after diagnosis, the risk of death following even a mild case of COVID-19 is not trivial and increases with disease severity,” he said. “Given that more than 30 million Americans have been infected with this virus, and given that the burden of long COVID-19 is substantial, the lingering effects of this disease will reverberate for many years and even decades.”
A version of this article first appeared on WebMD.com.
FDA to recommend limits on heavy metals in baby foods
The Food and Drug Administration has responded to congressional criticism and launched a multiyear plan to reduce the amount of heavy metals such as mercury and arsenic found in baby food.
Called “Closer to Zero,” the FDA plan calls for continued scientific investigation, establishes acceptable levels of heavy metals, sets up a way to monitor manufacturers’ compliance, and sets “action levels.”
“Although the FDA’s testing shows that children are not at an immediate health risk from exposure to toxic elements at the levels found in foods, we are starting the plan’s work immediately, with both short- and long-term goals for achieving continued improvements in reducing levels of toxic elements in these foods over time,” the FDA said.
However, Closer to Zero will only make recommendations on heavy metal levels.
“Although action levels are not binding, we have seen that, over the years, our guidance on action levels and other actions have contributed to significant reductions of toxic elements in food,” an FDA spokeswoman wrote in a statement, according to the Washington Post.
A congressional panel said in February 2021 that major brands of commercial baby food routinely have high levels of toxic heavy metals. The House Oversight Committee said this leaves babies at risk for serious developmental and neurologic problems.
The committee sharply criticized the FDA for not taking action.
“Despite the well-known risks of harm to babies from toxic heavy metals, FDA has not taken adequate steps to decrease their presence in baby foods,” the committee said. “FDA has not issued thresholds for the vast majority of toxic heavy metals in baby foods and does not require warning labels on any baby food products.”
A version of this article first appeared on WebMD.com.
The Food and Drug Administration has responded to congressional criticism and launched a multiyear plan to reduce the amount of heavy metals such as mercury and arsenic found in baby food.
Called “Closer to Zero,” the FDA plan calls for continued scientific investigation, establishes acceptable levels of heavy metals, sets up a way to monitor manufacturers’ compliance, and sets “action levels.”
“Although the FDA’s testing shows that children are not at an immediate health risk from exposure to toxic elements at the levels found in foods, we are starting the plan’s work immediately, with both short- and long-term goals for achieving continued improvements in reducing levels of toxic elements in these foods over time,” the FDA said.
However, Closer to Zero will only make recommendations on heavy metal levels.
“Although action levels are not binding, we have seen that, over the years, our guidance on action levels and other actions have contributed to significant reductions of toxic elements in food,” an FDA spokeswoman wrote in a statement, according to the Washington Post.
A congressional panel said in February 2021 that major brands of commercial baby food routinely have high levels of toxic heavy metals. The House Oversight Committee said this leaves babies at risk for serious developmental and neurologic problems.
The committee sharply criticized the FDA for not taking action.
“Despite the well-known risks of harm to babies from toxic heavy metals, FDA has not taken adequate steps to decrease their presence in baby foods,” the committee said. “FDA has not issued thresholds for the vast majority of toxic heavy metals in baby foods and does not require warning labels on any baby food products.”
A version of this article first appeared on WebMD.com.
The Food and Drug Administration has responded to congressional criticism and launched a multiyear plan to reduce the amount of heavy metals such as mercury and arsenic found in baby food.
Called “Closer to Zero,” the FDA plan calls for continued scientific investigation, establishes acceptable levels of heavy metals, sets up a way to monitor manufacturers’ compliance, and sets “action levels.”
“Although the FDA’s testing shows that children are not at an immediate health risk from exposure to toxic elements at the levels found in foods, we are starting the plan’s work immediately, with both short- and long-term goals for achieving continued improvements in reducing levels of toxic elements in these foods over time,” the FDA said.
However, Closer to Zero will only make recommendations on heavy metal levels.
“Although action levels are not binding, we have seen that, over the years, our guidance on action levels and other actions have contributed to significant reductions of toxic elements in food,” an FDA spokeswoman wrote in a statement, according to the Washington Post.
A congressional panel said in February 2021 that major brands of commercial baby food routinely have high levels of toxic heavy metals. The House Oversight Committee said this leaves babies at risk for serious developmental and neurologic problems.
The committee sharply criticized the FDA for not taking action.
“Despite the well-known risks of harm to babies from toxic heavy metals, FDA has not taken adequate steps to decrease their presence in baby foods,” the committee said. “FDA has not issued thresholds for the vast majority of toxic heavy metals in baby foods and does not require warning labels on any baby food products.”
A version of this article first appeared on WebMD.com.
Arkansas first state to ban transgender medical treatments for youths
Arkansas has become the first state to pass a law prohibiting doctors from giving gender-affirming medical treatments to transgender youths, CNN reported.
Gov. Asa Hutchinson had vetoed the bill on April 5, calling it a “product of the cultural war in America.” But on April 6, the state House and Senate voted to override the veto, making it state law, CNN reported.
At least 17 other states are considering similar legislation, but the Arkansas bill was the first to reach the governor’s desk, the Washington Post reported.
The bill bans doctors from prescribing puberty blockers, hormone therapies, or genital-altering surgeries for anybody under 18. Ever referring a youth for such treatment from another doctor is prohibited.
“It is of grave concern to the General Assembly that the medical community is allowing individuals who experience distress at identifying with their biological sex to be subjects of irreversible and drastic nongenital gender reassignment surgery and irreversible, permanently sterilizing genital gender reassignment surgery, despite the lack of studies showing that the benefits of such extreme interventions outweigh the risks,” the text of the bill said.
Gov. Hutchinson, a Republican, had called the measure a “vast government overreach” in announcing his veto.
“The bill is overbroad, extreme, and does not grandfather those young people who are currently under hormone treatment,” Gov. Hutchinson said. “The young people who are currently under a doctor’s care will be without treatment when this law goes into effect. That means they will be looking to the black market or go out of state ... to find the treatment that they want and need. This is not the right path to put them on.”
Many medical groups have come out against this kind of legislation. The American Academy of Child and Adolescent Psychiatry says it “strongly opposes any efforts – legal, legislative, and otherwise – to block access to these recognized interventions.”
Chase Strangio, the deputy director for transgender justice with the American Civil Liberty Union’s LGBTQ & HIV Project, complimented Gov. Hutchinson for his veto. On April 6, he said the ACLU is preparing to challenge the bill in court, CNN said.
A version of this article first appeared on Medscape.com.
Arkansas has become the first state to pass a law prohibiting doctors from giving gender-affirming medical treatments to transgender youths, CNN reported.
Gov. Asa Hutchinson had vetoed the bill on April 5, calling it a “product of the cultural war in America.” But on April 6, the state House and Senate voted to override the veto, making it state law, CNN reported.
At least 17 other states are considering similar legislation, but the Arkansas bill was the first to reach the governor’s desk, the Washington Post reported.
The bill bans doctors from prescribing puberty blockers, hormone therapies, or genital-altering surgeries for anybody under 18. Ever referring a youth for such treatment from another doctor is prohibited.
“It is of grave concern to the General Assembly that the medical community is allowing individuals who experience distress at identifying with their biological sex to be subjects of irreversible and drastic nongenital gender reassignment surgery and irreversible, permanently sterilizing genital gender reassignment surgery, despite the lack of studies showing that the benefits of such extreme interventions outweigh the risks,” the text of the bill said.
Gov. Hutchinson, a Republican, had called the measure a “vast government overreach” in announcing his veto.
“The bill is overbroad, extreme, and does not grandfather those young people who are currently under hormone treatment,” Gov. Hutchinson said. “The young people who are currently under a doctor’s care will be without treatment when this law goes into effect. That means they will be looking to the black market or go out of state ... to find the treatment that they want and need. This is not the right path to put them on.”
Many medical groups have come out against this kind of legislation. The American Academy of Child and Adolescent Psychiatry says it “strongly opposes any efforts – legal, legislative, and otherwise – to block access to these recognized interventions.”
Chase Strangio, the deputy director for transgender justice with the American Civil Liberty Union’s LGBTQ & HIV Project, complimented Gov. Hutchinson for his veto. On April 6, he said the ACLU is preparing to challenge the bill in court, CNN said.
A version of this article first appeared on Medscape.com.
Arkansas has become the first state to pass a law prohibiting doctors from giving gender-affirming medical treatments to transgender youths, CNN reported.
Gov. Asa Hutchinson had vetoed the bill on April 5, calling it a “product of the cultural war in America.” But on April 6, the state House and Senate voted to override the veto, making it state law, CNN reported.
At least 17 other states are considering similar legislation, but the Arkansas bill was the first to reach the governor’s desk, the Washington Post reported.
The bill bans doctors from prescribing puberty blockers, hormone therapies, or genital-altering surgeries for anybody under 18. Ever referring a youth for such treatment from another doctor is prohibited.
“It is of grave concern to the General Assembly that the medical community is allowing individuals who experience distress at identifying with their biological sex to be subjects of irreversible and drastic nongenital gender reassignment surgery and irreversible, permanently sterilizing genital gender reassignment surgery, despite the lack of studies showing that the benefits of such extreme interventions outweigh the risks,” the text of the bill said.
Gov. Hutchinson, a Republican, had called the measure a “vast government overreach” in announcing his veto.
“The bill is overbroad, extreme, and does not grandfather those young people who are currently under hormone treatment,” Gov. Hutchinson said. “The young people who are currently under a doctor’s care will be without treatment when this law goes into effect. That means they will be looking to the black market or go out of state ... to find the treatment that they want and need. This is not the right path to put them on.”
Many medical groups have come out against this kind of legislation. The American Academy of Child and Adolescent Psychiatry says it “strongly opposes any efforts – legal, legislative, and otherwise – to block access to these recognized interventions.”
Chase Strangio, the deputy director for transgender justice with the American Civil Liberty Union’s LGBTQ & HIV Project, complimented Gov. Hutchinson for his veto. On April 6, he said the ACLU is preparing to challenge the bill in court, CNN said.
A version of this article first appeared on Medscape.com.
Pfizer: Vaccine shown 100% effective in children aged 12-15
The study enrolled 2,260 adolescents aged 12-15. No infections were reported in the group given the vaccine produced by Pfizer and its European partner, BioNTech, the release said. The placebo group reported 18 cases of COVID-19.
The vaccinated children showed a strong antibody response with no serious side effects.
Albert Bourla, PhD, chairman and CEO of Pfizer, said the company plans to seek Food and Drug Administration emergency use authorization, which could allow this age group to be vaccinated before the start of the next school year. Pfizer will also seek authorization from the European Medicines Agency.
“We share the urgency to expand the authorization of our vaccine to use in younger populations and are encouraged by the clinical trial data from adolescents between the ages of 12 and 15,” Dr. Bourla said in the release.
The clinical trials showed a stronger response in children aged 12-15 than the 95% effectiveness reported in clinical trials in adults. The Pfizer vaccine is now authorized to be given to people aged 16 and up in the United States.
Health experts said the clinical trials – while not peer-reviewed – amounted to very good news.
“The sooner that we can get vaccines into as many people as possible, regardless of their age, the sooner we will be able to really feel like we’re ending this pandemic for good,” Angela Rasmussen, PhD, a virologist affiliated with Georgetown University in Washington, told The New York Times.
Anthony Fauci, MD, director of the National Institute of Allergy and Infectious Diseases, recently said that getting children vaccinated is an important step toward achieving herd immunity.
“We don’t really know what that magical point of herd immunity is, but we do know that if we get the overwhelming population vaccinated, we’re going to be in good shape,” he said earlier in March during a hearing of the Senate Health, Education, Labor, and Pensions Committee. “We ultimately would like to get and have to get children into that mix.”
Pfizer said it started clinical trials during the week of March 23 with children aged 5-11 and will next start trials with children aged 2-5, followed by children aged 6 months to 2 years. Vaccine makers Moderna and AstraZeneca also have started clinical trials in younger children.
A version of this article first appeared on WebMD.com.
The study enrolled 2,260 adolescents aged 12-15. No infections were reported in the group given the vaccine produced by Pfizer and its European partner, BioNTech, the release said. The placebo group reported 18 cases of COVID-19.
The vaccinated children showed a strong antibody response with no serious side effects.
Albert Bourla, PhD, chairman and CEO of Pfizer, said the company plans to seek Food and Drug Administration emergency use authorization, which could allow this age group to be vaccinated before the start of the next school year. Pfizer will also seek authorization from the European Medicines Agency.
“We share the urgency to expand the authorization of our vaccine to use in younger populations and are encouraged by the clinical trial data from adolescents between the ages of 12 and 15,” Dr. Bourla said in the release.
The clinical trials showed a stronger response in children aged 12-15 than the 95% effectiveness reported in clinical trials in adults. The Pfizer vaccine is now authorized to be given to people aged 16 and up in the United States.
Health experts said the clinical trials – while not peer-reviewed – amounted to very good news.
“The sooner that we can get vaccines into as many people as possible, regardless of their age, the sooner we will be able to really feel like we’re ending this pandemic for good,” Angela Rasmussen, PhD, a virologist affiliated with Georgetown University in Washington, told The New York Times.
Anthony Fauci, MD, director of the National Institute of Allergy and Infectious Diseases, recently said that getting children vaccinated is an important step toward achieving herd immunity.
“We don’t really know what that magical point of herd immunity is, but we do know that if we get the overwhelming population vaccinated, we’re going to be in good shape,” he said earlier in March during a hearing of the Senate Health, Education, Labor, and Pensions Committee. “We ultimately would like to get and have to get children into that mix.”
Pfizer said it started clinical trials during the week of March 23 with children aged 5-11 and will next start trials with children aged 2-5, followed by children aged 6 months to 2 years. Vaccine makers Moderna and AstraZeneca also have started clinical trials in younger children.
A version of this article first appeared on WebMD.com.
The study enrolled 2,260 adolescents aged 12-15. No infections were reported in the group given the vaccine produced by Pfizer and its European partner, BioNTech, the release said. The placebo group reported 18 cases of COVID-19.
The vaccinated children showed a strong antibody response with no serious side effects.
Albert Bourla, PhD, chairman and CEO of Pfizer, said the company plans to seek Food and Drug Administration emergency use authorization, which could allow this age group to be vaccinated before the start of the next school year. Pfizer will also seek authorization from the European Medicines Agency.
“We share the urgency to expand the authorization of our vaccine to use in younger populations and are encouraged by the clinical trial data from adolescents between the ages of 12 and 15,” Dr. Bourla said in the release.
The clinical trials showed a stronger response in children aged 12-15 than the 95% effectiveness reported in clinical trials in adults. The Pfizer vaccine is now authorized to be given to people aged 16 and up in the United States.
Health experts said the clinical trials – while not peer-reviewed – amounted to very good news.
“The sooner that we can get vaccines into as many people as possible, regardless of their age, the sooner we will be able to really feel like we’re ending this pandemic for good,” Angela Rasmussen, PhD, a virologist affiliated with Georgetown University in Washington, told The New York Times.
Anthony Fauci, MD, director of the National Institute of Allergy and Infectious Diseases, recently said that getting children vaccinated is an important step toward achieving herd immunity.
“We don’t really know what that magical point of herd immunity is, but we do know that if we get the overwhelming population vaccinated, we’re going to be in good shape,” he said earlier in March during a hearing of the Senate Health, Education, Labor, and Pensions Committee. “We ultimately would like to get and have to get children into that mix.”
Pfizer said it started clinical trials during the week of March 23 with children aged 5-11 and will next start trials with children aged 2-5, followed by children aged 6 months to 2 years. Vaccine makers Moderna and AstraZeneca also have started clinical trials in younger children.
A version of this article first appeared on WebMD.com.
Fauci worries about possible post–COVID-19 ‘mental health pandemic’
Anthony Fauci, MD, says he’s concerned about how Americans will react once the coronavirus pandemic is brought under control, CBS News reports.
Noting that an American Psychological Association survey showed people reporting high stress levels because of the pandemic, CBS’s Norah O’Donnell asked if Dr. Fauci was concerned about a possible “mental health pandemic.”
“Very much so,” Dr. Fauci, director of the National Institute of Allergy and Infectious Diseases and a top White House coronavirus adviser, replied.
“That’s the reason why I want to get the virological aspect of this pandemic behind us as quickly as we possibly can because the long-term ravages of this are so multifaceted,” Dr. Fauci said.
, he said.
“And then the other things: Not only the mental health effects, but many people have put off routine types of medical examinations that they normally would have done,” Dr. Fauci said.
“I hope we don’t see an increase in some preventable situations that would not have happened if people had the normal access to medical care, which clearly was interrupted by the shutdown associated with COVID-19,” he added.
The American Psychological Association released the survey results March 11 in what many people consider the 1-year anniversary of the start of the coronavirus pandemic.
“The prolonged stress experienced by adults, especially the high levels of stress reported by Americans directly linked to the pandemic, is seriously affecting mental and physical health, including changes to weight, sleep and alcohol use,” the APA said in a news release.
Some of the key findings of the survey include:
- 61% of respondents reported experiencing undesired weight changes since the start of the pandemic.
- 67% said their sleep habits changed, with 35% saying they slept more and 31% less.
- 23% reported drinking more alcohol to cope with stress.
- 47% said they delayed or canceled health care services because of the pandemic.
- 48% said their stress levels had increased.
A version of this article first appeared on Medscape.com.
Anthony Fauci, MD, says he’s concerned about how Americans will react once the coronavirus pandemic is brought under control, CBS News reports.
Noting that an American Psychological Association survey showed people reporting high stress levels because of the pandemic, CBS’s Norah O’Donnell asked if Dr. Fauci was concerned about a possible “mental health pandemic.”
“Very much so,” Dr. Fauci, director of the National Institute of Allergy and Infectious Diseases and a top White House coronavirus adviser, replied.
“That’s the reason why I want to get the virological aspect of this pandemic behind us as quickly as we possibly can because the long-term ravages of this are so multifaceted,” Dr. Fauci said.
, he said.
“And then the other things: Not only the mental health effects, but many people have put off routine types of medical examinations that they normally would have done,” Dr. Fauci said.
“I hope we don’t see an increase in some preventable situations that would not have happened if people had the normal access to medical care, which clearly was interrupted by the shutdown associated with COVID-19,” he added.
The American Psychological Association released the survey results March 11 in what many people consider the 1-year anniversary of the start of the coronavirus pandemic.
“The prolonged stress experienced by adults, especially the high levels of stress reported by Americans directly linked to the pandemic, is seriously affecting mental and physical health, including changes to weight, sleep and alcohol use,” the APA said in a news release.
Some of the key findings of the survey include:
- 61% of respondents reported experiencing undesired weight changes since the start of the pandemic.
- 67% said their sleep habits changed, with 35% saying they slept more and 31% less.
- 23% reported drinking more alcohol to cope with stress.
- 47% said they delayed or canceled health care services because of the pandemic.
- 48% said their stress levels had increased.
A version of this article first appeared on Medscape.com.
Anthony Fauci, MD, says he’s concerned about how Americans will react once the coronavirus pandemic is brought under control, CBS News reports.
Noting that an American Psychological Association survey showed people reporting high stress levels because of the pandemic, CBS’s Norah O’Donnell asked if Dr. Fauci was concerned about a possible “mental health pandemic.”
“Very much so,” Dr. Fauci, director of the National Institute of Allergy and Infectious Diseases and a top White House coronavirus adviser, replied.
“That’s the reason why I want to get the virological aspect of this pandemic behind us as quickly as we possibly can because the long-term ravages of this are so multifaceted,” Dr. Fauci said.
, he said.
“And then the other things: Not only the mental health effects, but many people have put off routine types of medical examinations that they normally would have done,” Dr. Fauci said.
“I hope we don’t see an increase in some preventable situations that would not have happened if people had the normal access to medical care, which clearly was interrupted by the shutdown associated with COVID-19,” he added.
The American Psychological Association released the survey results March 11 in what many people consider the 1-year anniversary of the start of the coronavirus pandemic.
“The prolonged stress experienced by adults, especially the high levels of stress reported by Americans directly linked to the pandemic, is seriously affecting mental and physical health, including changes to weight, sleep and alcohol use,” the APA said in a news release.
Some of the key findings of the survey include:
- 61% of respondents reported experiencing undesired weight changes since the start of the pandemic.
- 67% said their sleep habits changed, with 35% saying they slept more and 31% less.
- 23% reported drinking more alcohol to cope with stress.
- 47% said they delayed or canceled health care services because of the pandemic.
- 48% said their stress levels had increased.
A version of this article first appeared on Medscape.com.
Antidepressant may help COVID-19 patients avoid serious illness
The antidepressant fluvoxamine shows promise in preventing people infected with coronavirus from developing serious symptoms and having to be hospitalized, according to a nonrandomized study of California racetrack workers.
“What we observed was that of all the patients who received fluvoxamine, none of them had a severe COVID infection that affected their lungs or their respiratory status,” Caline Mattar, MD, told KNBC in Los Angeles. Dr. Mattar is an infectious disease researcher at Washington University in St. Louis who helped conduct the study that was published in Open Forum Infectious Diseases.
Fluvoxamine, which is sold under the brand name Luvox, is a selective serotonin reuptake inhibitor (SSRI) often prescribed for people diagnosed with obsessive-compulsive disorder. It’s been on the market for over a decade.
Two-hundred employees at Golden Gate Fields Racetrack in Berkeley, Calif., tested positive for COVID-19 last November. Track physician David Seftel, MD, offered fluvoxamine to 113 of them, having learned of a previous randomized study of COVID-19 patients that indicated fluvoxamine helped ward off serious illness, Science News said.
The 65 workers who took a 2-week course of the drug didn’t have to be hospitalized, didn’t have serious symptoms, and felt better after 2 weeks, the study said. Six of the 48 workers who turned down fluvoxamine had to be hospitalized, two required intensive care, and one died, the study said.
“Overall, fluvoxamine appears promising as early treatment for COVID-19 to prevent clinical deterioration requiring hospitalization and to prevent possible long haul symptoms persisting beyond 2 weeks,” the study said.
They said their research needed verification from a randomized, controlled trial. Such a study is now being conducted by Washington University and other schools, KNBC said.
The track workers who were infected were predominantly male and Latino, and 30% had chronic medical problems such as diabetes or high blood pressure, Science News said.
A version of this article first appeared on WebMD.com.
The antidepressant fluvoxamine shows promise in preventing people infected with coronavirus from developing serious symptoms and having to be hospitalized, according to a nonrandomized study of California racetrack workers.
“What we observed was that of all the patients who received fluvoxamine, none of them had a severe COVID infection that affected their lungs or their respiratory status,” Caline Mattar, MD, told KNBC in Los Angeles. Dr. Mattar is an infectious disease researcher at Washington University in St. Louis who helped conduct the study that was published in Open Forum Infectious Diseases.
Fluvoxamine, which is sold under the brand name Luvox, is a selective serotonin reuptake inhibitor (SSRI) often prescribed for people diagnosed with obsessive-compulsive disorder. It’s been on the market for over a decade.
Two-hundred employees at Golden Gate Fields Racetrack in Berkeley, Calif., tested positive for COVID-19 last November. Track physician David Seftel, MD, offered fluvoxamine to 113 of them, having learned of a previous randomized study of COVID-19 patients that indicated fluvoxamine helped ward off serious illness, Science News said.
The 65 workers who took a 2-week course of the drug didn’t have to be hospitalized, didn’t have serious symptoms, and felt better after 2 weeks, the study said. Six of the 48 workers who turned down fluvoxamine had to be hospitalized, two required intensive care, and one died, the study said.
“Overall, fluvoxamine appears promising as early treatment for COVID-19 to prevent clinical deterioration requiring hospitalization and to prevent possible long haul symptoms persisting beyond 2 weeks,” the study said.
They said their research needed verification from a randomized, controlled trial. Such a study is now being conducted by Washington University and other schools, KNBC said.
The track workers who were infected were predominantly male and Latino, and 30% had chronic medical problems such as diabetes or high blood pressure, Science News said.
A version of this article first appeared on WebMD.com.
The antidepressant fluvoxamine shows promise in preventing people infected with coronavirus from developing serious symptoms and having to be hospitalized, according to a nonrandomized study of California racetrack workers.
“What we observed was that of all the patients who received fluvoxamine, none of them had a severe COVID infection that affected their lungs or their respiratory status,” Caline Mattar, MD, told KNBC in Los Angeles. Dr. Mattar is an infectious disease researcher at Washington University in St. Louis who helped conduct the study that was published in Open Forum Infectious Diseases.
Fluvoxamine, which is sold under the brand name Luvox, is a selective serotonin reuptake inhibitor (SSRI) often prescribed for people diagnosed with obsessive-compulsive disorder. It’s been on the market for over a decade.
Two-hundred employees at Golden Gate Fields Racetrack in Berkeley, Calif., tested positive for COVID-19 last November. Track physician David Seftel, MD, offered fluvoxamine to 113 of them, having learned of a previous randomized study of COVID-19 patients that indicated fluvoxamine helped ward off serious illness, Science News said.
The 65 workers who took a 2-week course of the drug didn’t have to be hospitalized, didn’t have serious symptoms, and felt better after 2 weeks, the study said. Six of the 48 workers who turned down fluvoxamine had to be hospitalized, two required intensive care, and one died, the study said.
“Overall, fluvoxamine appears promising as early treatment for COVID-19 to prevent clinical deterioration requiring hospitalization and to prevent possible long haul symptoms persisting beyond 2 weeks,” the study said.
They said their research needed verification from a randomized, controlled trial. Such a study is now being conducted by Washington University and other schools, KNBC said.
The track workers who were infected were predominantly male and Latino, and 30% had chronic medical problems such as diabetes or high blood pressure, Science News said.
A version of this article first appeared on WebMD.com.
Moderna needs more kids for COVID vaccine trials
according to the company CEO and a federal official.
The Moderna vaccine was authorized for use in December and is now being given to people 18 and over. But children would receive lower doses, so new clinical trials must be done, Moderna CEO Stephane Bancel said at the JPMorgan virtual Health Care Conference on Monday.
Clinical trials on children 11 and younger “will take much longer, because we have to age deescalate and start at a lower dose. So we should not anticipate clinical data in 2021, but more in 2022,” Ms. Bancel said, according to Business Insider.
Moderna’s clinical trials for 12- to 17-year-olds started 4 weeks ago, but the company is having trouble getting enough participants, said Moncef Slaoui, PhD, the scientific head of Operation Warp Speed, the U.S. government’s vaccine effort. That could delay Food and Drug Administration approval, he said.
“It’s really very important for all of us, for all the population in America, to realize that we can’t have that indication unless adolescents aged 12-18 decide to participate,” Dr. Slaoui said, according to USA Today.
He said the adolescent trials are getting only about 800 volunteers a month, but need at least 3,000 volunteers to complete the study, USA Today reported. Parents interested in having their child participate can check eligibility and sign at this website.
The Pfizer/BioNTech vaccine won authorization for use in 16- to 17-year-olds as well as adults.
The coronavirus doesn’t appear to have as serious complications for children as for adults.
“At this time, it appears that severe illness due to COVID-19 is rare among children,” the American Association of Pediatrics says. “However, there is an urgent need to collect more data on longer-term impacts of the pandemic on children, including ways the virus may harm the long-term physical health of infected children, as well as its emotional and mental health effects.”
The association says 179 children had died of COVID-related reasons in 43 states and New York City as of Dec. 31, 2020. That’s about 0.06% of total COVID deaths, it says.
But children do get sick. As of Jan. 7, 2021, nearly 2.3 million children had tested positive for COVID-19 since the start of the pandemic, the association says.
A version of this article first appeared on WebMD.com.
according to the company CEO and a federal official.
The Moderna vaccine was authorized for use in December and is now being given to people 18 and over. But children would receive lower doses, so new clinical trials must be done, Moderna CEO Stephane Bancel said at the JPMorgan virtual Health Care Conference on Monday.
Clinical trials on children 11 and younger “will take much longer, because we have to age deescalate and start at a lower dose. So we should not anticipate clinical data in 2021, but more in 2022,” Ms. Bancel said, according to Business Insider.
Moderna’s clinical trials for 12- to 17-year-olds started 4 weeks ago, but the company is having trouble getting enough participants, said Moncef Slaoui, PhD, the scientific head of Operation Warp Speed, the U.S. government’s vaccine effort. That could delay Food and Drug Administration approval, he said.
“It’s really very important for all of us, for all the population in America, to realize that we can’t have that indication unless adolescents aged 12-18 decide to participate,” Dr. Slaoui said, according to USA Today.
He said the adolescent trials are getting only about 800 volunteers a month, but need at least 3,000 volunteers to complete the study, USA Today reported. Parents interested in having their child participate can check eligibility and sign at this website.
The Pfizer/BioNTech vaccine won authorization for use in 16- to 17-year-olds as well as adults.
The coronavirus doesn’t appear to have as serious complications for children as for adults.
“At this time, it appears that severe illness due to COVID-19 is rare among children,” the American Association of Pediatrics says. “However, there is an urgent need to collect more data on longer-term impacts of the pandemic on children, including ways the virus may harm the long-term physical health of infected children, as well as its emotional and mental health effects.”
The association says 179 children had died of COVID-related reasons in 43 states and New York City as of Dec. 31, 2020. That’s about 0.06% of total COVID deaths, it says.
But children do get sick. As of Jan. 7, 2021, nearly 2.3 million children had tested positive for COVID-19 since the start of the pandemic, the association says.
A version of this article first appeared on WebMD.com.
according to the company CEO and a federal official.
The Moderna vaccine was authorized for use in December and is now being given to people 18 and over. But children would receive lower doses, so new clinical trials must be done, Moderna CEO Stephane Bancel said at the JPMorgan virtual Health Care Conference on Monday.
Clinical trials on children 11 and younger “will take much longer, because we have to age deescalate and start at a lower dose. So we should not anticipate clinical data in 2021, but more in 2022,” Ms. Bancel said, according to Business Insider.
Moderna’s clinical trials for 12- to 17-year-olds started 4 weeks ago, but the company is having trouble getting enough participants, said Moncef Slaoui, PhD, the scientific head of Operation Warp Speed, the U.S. government’s vaccine effort. That could delay Food and Drug Administration approval, he said.
“It’s really very important for all of us, for all the population in America, to realize that we can’t have that indication unless adolescents aged 12-18 decide to participate,” Dr. Slaoui said, according to USA Today.
He said the adolescent trials are getting only about 800 volunteers a month, but need at least 3,000 volunteers to complete the study, USA Today reported. Parents interested in having their child participate can check eligibility and sign at this website.
The Pfizer/BioNTech vaccine won authorization for use in 16- to 17-year-olds as well as adults.
The coronavirus doesn’t appear to have as serious complications for children as for adults.
“At this time, it appears that severe illness due to COVID-19 is rare among children,” the American Association of Pediatrics says. “However, there is an urgent need to collect more data on longer-term impacts of the pandemic on children, including ways the virus may harm the long-term physical health of infected children, as well as its emotional and mental health effects.”
The association says 179 children had died of COVID-related reasons in 43 states and New York City as of Dec. 31, 2020. That’s about 0.06% of total COVID deaths, it says.
But children do get sick. As of Jan. 7, 2021, nearly 2.3 million children had tested positive for COVID-19 since the start of the pandemic, the association says.
A version of this article first appeared on WebMD.com.
COVID protections suppressed flu season in U.S.
Last fall, health experts said it was possible the United States could experience an easy 2020-21 flu season because health measures to fight COVID-19 would also thwart the spread of influenza.
It looks like that happened – and then some. Numbers are strikingly low for cases of the flu and other common respiratory and gastrointestinal viruses, health experts told the Washington Post.
“It’s crazy,” Lynnette Brammer, MPH, who leads the domestic influenza surveillance team at the Centers for Disease Control and Prevention, told the Washington Post. “This is my 30th flu season. I never would have expected to see flu activity this low.”
Influenza A, influenza B, parainfluenza, norovirus, respiratory syncytial virus, human metapneumovirus, and the bacteria that cause whooping cough and pneumonia are circulating at near-record-low levels.
As an example, the Washington Post said in the third week of December 2019, the CDC’s network of clinical labs reported 16.2% of almost 30,000 samples tested positive for influenza A. During the same period in 2020, only 0.3% tested positive.
But there’s a possible downside to this suppression of viruses, because flu and other viruses may rebound once the coronavirus is brought under control.
“The best analogy is to a forest fire,” Bryan Grenfell, PhD, an epidemiologist and population biologist at Princeton (N.J.) University, told the Washington Post. “For the fire to spread, it needs to have unburned wood. For epidemics to spread, they require people who haven’t previously been infected. So if people don’t get infected this year by these viruses, they likely will at some point later on.”
American health experts like Anthony Fauci, MD, director of the National Institute of Allergy and Infectious Disease, said last fall that they noticed Australia and other nations in the southern hemisphere had easy flu seasons, apparently because of COVID protection measures. The flu season there runs March through August.
COVID-19 now has a very low presence in Australia, but in recent months the flu has been making a comeback. Flu cases among children aged 5 and younger rose sixfold by December, when such cases are usually at their lowest, the Washington Post said.
“That’s an important cautionary tale for us,” said Kevin Messacar, MD, an infectious disease doctor at Children’s Hospital Colorado, Aurora. “Just because we get through the winter and don’t see much RSV or influenza doesn’t mean we’ll be out of the woods.”
A version of this article first appeared on WebMD.com.
Last fall, health experts said it was possible the United States could experience an easy 2020-21 flu season because health measures to fight COVID-19 would also thwart the spread of influenza.
It looks like that happened – and then some. Numbers are strikingly low for cases of the flu and other common respiratory and gastrointestinal viruses, health experts told the Washington Post.
“It’s crazy,” Lynnette Brammer, MPH, who leads the domestic influenza surveillance team at the Centers for Disease Control and Prevention, told the Washington Post. “This is my 30th flu season. I never would have expected to see flu activity this low.”
Influenza A, influenza B, parainfluenza, norovirus, respiratory syncytial virus, human metapneumovirus, and the bacteria that cause whooping cough and pneumonia are circulating at near-record-low levels.
As an example, the Washington Post said in the third week of December 2019, the CDC’s network of clinical labs reported 16.2% of almost 30,000 samples tested positive for influenza A. During the same period in 2020, only 0.3% tested positive.
But there’s a possible downside to this suppression of viruses, because flu and other viruses may rebound once the coronavirus is brought under control.
“The best analogy is to a forest fire,” Bryan Grenfell, PhD, an epidemiologist and population biologist at Princeton (N.J.) University, told the Washington Post. “For the fire to spread, it needs to have unburned wood. For epidemics to spread, they require people who haven’t previously been infected. So if people don’t get infected this year by these viruses, they likely will at some point later on.”
American health experts like Anthony Fauci, MD, director of the National Institute of Allergy and Infectious Disease, said last fall that they noticed Australia and other nations in the southern hemisphere had easy flu seasons, apparently because of COVID protection measures. The flu season there runs March through August.
COVID-19 now has a very low presence in Australia, but in recent months the flu has been making a comeback. Flu cases among children aged 5 and younger rose sixfold by December, when such cases are usually at their lowest, the Washington Post said.
“That’s an important cautionary tale for us,” said Kevin Messacar, MD, an infectious disease doctor at Children’s Hospital Colorado, Aurora. “Just because we get through the winter and don’t see much RSV or influenza doesn’t mean we’ll be out of the woods.”
A version of this article first appeared on WebMD.com.
Last fall, health experts said it was possible the United States could experience an easy 2020-21 flu season because health measures to fight COVID-19 would also thwart the spread of influenza.
It looks like that happened – and then some. Numbers are strikingly low for cases of the flu and other common respiratory and gastrointestinal viruses, health experts told the Washington Post.
“It’s crazy,” Lynnette Brammer, MPH, who leads the domestic influenza surveillance team at the Centers for Disease Control and Prevention, told the Washington Post. “This is my 30th flu season. I never would have expected to see flu activity this low.”
Influenza A, influenza B, parainfluenza, norovirus, respiratory syncytial virus, human metapneumovirus, and the bacteria that cause whooping cough and pneumonia are circulating at near-record-low levels.
As an example, the Washington Post said in the third week of December 2019, the CDC’s network of clinical labs reported 16.2% of almost 30,000 samples tested positive for influenza A. During the same period in 2020, only 0.3% tested positive.
But there’s a possible downside to this suppression of viruses, because flu and other viruses may rebound once the coronavirus is brought under control.
“The best analogy is to a forest fire,” Bryan Grenfell, PhD, an epidemiologist and population biologist at Princeton (N.J.) University, told the Washington Post. “For the fire to spread, it needs to have unburned wood. For epidemics to spread, they require people who haven’t previously been infected. So if people don’t get infected this year by these viruses, they likely will at some point later on.”
American health experts like Anthony Fauci, MD, director of the National Institute of Allergy and Infectious Disease, said last fall that they noticed Australia and other nations in the southern hemisphere had easy flu seasons, apparently because of COVID protection measures. The flu season there runs March through August.
COVID-19 now has a very low presence in Australia, but in recent months the flu has been making a comeback. Flu cases among children aged 5 and younger rose sixfold by December, when such cases are usually at their lowest, the Washington Post said.
“That’s an important cautionary tale for us,” said Kevin Messacar, MD, an infectious disease doctor at Children’s Hospital Colorado, Aurora. “Just because we get through the winter and don’t see much RSV or influenza doesn’t mean we’ll be out of the woods.”
A version of this article first appeared on WebMD.com.
CDC issues COVID-19 vaccine guidance for underlying conditions
The Centers for Disease Control and Prevention has issued updated guidance for people with underlying medical conditions who are considering getting the coronavirus vaccine.
“Adults of any age with certain underlying medical conditions are at increased risk for severe illness from the virus that causes COVID-19,” the CDC said in the guidance, posted on Dec. 26. “mRNA COVID-19 vaccines may be administered to people with underlying medical conditions provided they have not had a severe allergic reaction to any of the ingredients in the vaccine.”
Both the Pfizer and Moderna vaccines use mRNA, or messenger RNA.
The CDC guidance had specific information for people with HIV, weakened immune systems, and autoimmune conditions such as Guillain-Barré syndrome (GBS) and Bell’s palsy who are thinking of getting the vaccine.
People with HIV and weakened immune systems “may receive a COVID-19 vaccine. However, they should be aware of the limited safety data,” the CDC said.
There’s no information available yet about the safety of the vaccines for people with weakened immune systems. People with HIV were included in clinical trials, but “safety data specific to this group are not yet available at this time,” the CDC said.
Cases of Bell’s palsy, a temporary facial paralysis, were reported in people receiving the Pfizer and Moderna vaccines in clinical trials, the Food and Drug Administration said Dec. 17.
But the new CDC guidance said that the FDA “does not consider these to be above the rate expected in the general population. They have not concluded these cases were caused by vaccination. Therefore, persons who have previously had Bell’s palsy may receive an mRNA COVID-19 vaccine.”
Researchers have determined the vaccines are safe for people with GBS, a rare autoimmune disorder in which the body’s immune system attacks nerves just as they leave the spinal cord, the CDC said.
“To date, no cases of GBS have been reported following vaccination among participants in the mRNA COVID-19 vaccine clinical trials,” the CDC guidance said. “With few exceptions, the independent Advisory Committee on Immunization Practices general best practice guidelines for immunization do not include a history of GBS as a precaution to vaccination with other vaccines.”
For months, the CDC and other health authorities have said that people with certain medical conditions are at an increased risk of developing severe cases of COVID-19.
A version of this article first appeared on Medscape.com.
The Centers for Disease Control and Prevention has issued updated guidance for people with underlying medical conditions who are considering getting the coronavirus vaccine.
“Adults of any age with certain underlying medical conditions are at increased risk for severe illness from the virus that causes COVID-19,” the CDC said in the guidance, posted on Dec. 26. “mRNA COVID-19 vaccines may be administered to people with underlying medical conditions provided they have not had a severe allergic reaction to any of the ingredients in the vaccine.”
Both the Pfizer and Moderna vaccines use mRNA, or messenger RNA.
The CDC guidance had specific information for people with HIV, weakened immune systems, and autoimmune conditions such as Guillain-Barré syndrome (GBS) and Bell’s palsy who are thinking of getting the vaccine.
People with HIV and weakened immune systems “may receive a COVID-19 vaccine. However, they should be aware of the limited safety data,” the CDC said.
There’s no information available yet about the safety of the vaccines for people with weakened immune systems. People with HIV were included in clinical trials, but “safety data specific to this group are not yet available at this time,” the CDC said.
Cases of Bell’s palsy, a temporary facial paralysis, were reported in people receiving the Pfizer and Moderna vaccines in clinical trials, the Food and Drug Administration said Dec. 17.
But the new CDC guidance said that the FDA “does not consider these to be above the rate expected in the general population. They have not concluded these cases were caused by vaccination. Therefore, persons who have previously had Bell’s palsy may receive an mRNA COVID-19 vaccine.”
Researchers have determined the vaccines are safe for people with GBS, a rare autoimmune disorder in which the body’s immune system attacks nerves just as they leave the spinal cord, the CDC said.
“To date, no cases of GBS have been reported following vaccination among participants in the mRNA COVID-19 vaccine clinical trials,” the CDC guidance said. “With few exceptions, the independent Advisory Committee on Immunization Practices general best practice guidelines for immunization do not include a history of GBS as a precaution to vaccination with other vaccines.”
For months, the CDC and other health authorities have said that people with certain medical conditions are at an increased risk of developing severe cases of COVID-19.
A version of this article first appeared on Medscape.com.
The Centers for Disease Control and Prevention has issued updated guidance for people with underlying medical conditions who are considering getting the coronavirus vaccine.
“Adults of any age with certain underlying medical conditions are at increased risk for severe illness from the virus that causes COVID-19,” the CDC said in the guidance, posted on Dec. 26. “mRNA COVID-19 vaccines may be administered to people with underlying medical conditions provided they have not had a severe allergic reaction to any of the ingredients in the vaccine.”
Both the Pfizer and Moderna vaccines use mRNA, or messenger RNA.
The CDC guidance had specific information for people with HIV, weakened immune systems, and autoimmune conditions such as Guillain-Barré syndrome (GBS) and Bell’s palsy who are thinking of getting the vaccine.
People with HIV and weakened immune systems “may receive a COVID-19 vaccine. However, they should be aware of the limited safety data,” the CDC said.
There’s no information available yet about the safety of the vaccines for people with weakened immune systems. People with HIV were included in clinical trials, but “safety data specific to this group are not yet available at this time,” the CDC said.
Cases of Bell’s palsy, a temporary facial paralysis, were reported in people receiving the Pfizer and Moderna vaccines in clinical trials, the Food and Drug Administration said Dec. 17.
But the new CDC guidance said that the FDA “does not consider these to be above the rate expected in the general population. They have not concluded these cases were caused by vaccination. Therefore, persons who have previously had Bell’s palsy may receive an mRNA COVID-19 vaccine.”
Researchers have determined the vaccines are safe for people with GBS, a rare autoimmune disorder in which the body’s immune system attacks nerves just as they leave the spinal cord, the CDC said.
“To date, no cases of GBS have been reported following vaccination among participants in the mRNA COVID-19 vaccine clinical trials,” the CDC guidance said. “With few exceptions, the independent Advisory Committee on Immunization Practices general best practice guidelines for immunization do not include a history of GBS as a precaution to vaccination with other vaccines.”
For months, the CDC and other health authorities have said that people with certain medical conditions are at an increased risk of developing severe cases of COVID-19.
A version of this article first appeared on Medscape.com.
COVID-19 variant sparks U.K. travel restrictions
Researchers have detected a highly contagious coronavirus variant in the United Kingdom, leading Prime Minister Boris Johnson to shut down parts of the country and triggering other nations to impose travel and shipping restrictions on England.
Mr. Johnson held a crisis meeting with ministers Monday after Saturday’s shutdown announcement. The prime minister said in a nationally televised address that this coronavirus variant may be “up to 70% more transmissible than the old variant” and was probably responsible for an increase in cases in southeastern England.
“There is still much we don’t know. While we are fairly certain the variant is transmitted more quickly, there is no evidence to suggest that it is more lethal or causes more severe illness. Equally there is no evidence to suggest the vaccine will be any less effective against the new variant,” he said.
Public Health England says it is working to learn as much about the variant as possible. “We know that mortality is a lagging indicator, and we will need to continually monitor this over the coming weeks,” the agency says.
That scientific uncertainty about the variant’s threat shook European nations that were rushing to ship goods to England in advance of a Dec. 31 Brexit deadline. Under Brexit, which is short for “British exit,” the United Kingdom will leave the European Union on Jan. 31, 2020. Until then, the two sides will come up with new trade and security relationships.
European Union members Austria, Belgium, Bulgaria, France, Germany, Ireland, Italy, and the Netherlands announced travel restrictions hours after Johnson’s speech.
Those restrictions created food uncertainty across the U.K., which imports about a quarter of its food from the EU, according to The New York Times. Long lines of trucks heading to ports in the U.K. came to a standstill on major roads such as the M20 near Kent and the Port of Dover.
Outside Europe, Canada, India, Iran, Israel, Hong Kong, Saudi Arabia, and Turkey banned all incoming flights from the U.K. And more bans could come.
The U.S. reaction
The United States has not imposed any new limits on travel with the United Kingdom, although New York Gov. Andrew Cuomo (D) has requested all passengers bound for John F. Kennedy International Airport from the U.K. be tested before boarding and a new travel ban be placed for Europe. He says the federal government must take action now to avoid a crisis situation like the one New York experienced in March and April.
“The United States has a number of flights coming in from the U.K. each day, and we have done absolutely nothing,” Mr. Cuomo said in a statement on the governor’s webpage. “To me, this is reprehensible because this is what happened in the spring. How many times in life do you have to make the same mistake before you learn?”
Leading U.S. health officials have downplayed the dangers of the virus.
“We don’t know that it’s more dangerous, and very importantly, we have not seen a single mutation yet that would make it evade the vaccine,” U.S. Assistant Secretary of Health and Human Services Adm. Brett Giroir, MD, said Sunday on ABC’s This Week with George Stephanopoulos. “I can’t say that won’t happen in the future, but right now it looks like the vaccine will cover everything that we see.”
Dr. Giroir said the HHS and other U.S. government agencies will monitor the variant.
“Viruses mutate,” he said. “We’ve seen almost 4,000 different mutations among this virus. There is no indication that the mutation right now that they’re talking about is overcoming England.”
Where did the variant come from?
Public Health England says the coronavirus variant had existed in the U.K. since September and circulated at very low levels until mid-November.
“The increase in cases linked to the new variant first came to light in late November when PHE was investigating why infection rates in Kent were not falling despite national restrictions. We then discovered a cluster linked to this variant spreading rapidly into London and Essex,” the agency said.
Public Health England says there’s no evidence the new variant is resistant to the Pfizer-BioNTech vaccine, which is now being given across the country to high-priority groups such as health care workers.
An article in The BMJ, a British medical journal, says the variant was first detected by Covid-19 Genomics UK, a consortium that tests the random genetic sequencing of positive COVID-19 samples around the U.K. The variant cases were mostly in the southeast of England.
A University of Birmingham professor said in a Dec. 15 briefing that the variant accounts for 20% of viruses sequenced in Norfolk, 10% in Essex, and 3% in Suffolk. “There are no data to suggest it had been imported from abroad, so it is likely to have evolved in the U.K.,” he said.
The variant is named VUI-202012/01, for the first “variant under investigation” in December 2020, BMJ says. It’s defined by a set of 17 mutations, with the most significant mutation in the spike protein the virus uses to bind to the human ACE2 receptor.
“Changes in this part of spike protein may, in theory, result in the virus becoming more infectious and spreading more easily between people,” the article says.
The European Centre for Disease Prevention and Control says the variant emerged during the time of year when people usually socialize more.
“There is no indication at this point of increased infection severity associated with the new variant,” the agency said. “A few cases with the new variant have to date been reported by Denmark and the Netherlands and, according to media reports, in Belgium.”
Mr. Johnson announced tighter restrictions on England’s hardest-hit areas, such as the southeast and east of England, where new coronavirus cases have continued to rise. And he said people must cut back on their Christmas socializing.
“In England, those living in tier 4 areas should not mix with anyone outside their own household at Christmas, though support bubbles will remain in place for those at particular risk of loneliness or isolation,” he said.
A version of this article first appeared on WebMD.com.
Researchers have detected a highly contagious coronavirus variant in the United Kingdom, leading Prime Minister Boris Johnson to shut down parts of the country and triggering other nations to impose travel and shipping restrictions on England.
Mr. Johnson held a crisis meeting with ministers Monday after Saturday’s shutdown announcement. The prime minister said in a nationally televised address that this coronavirus variant may be “up to 70% more transmissible than the old variant” and was probably responsible for an increase in cases in southeastern England.
“There is still much we don’t know. While we are fairly certain the variant is transmitted more quickly, there is no evidence to suggest that it is more lethal or causes more severe illness. Equally there is no evidence to suggest the vaccine will be any less effective against the new variant,” he said.
Public Health England says it is working to learn as much about the variant as possible. “We know that mortality is a lagging indicator, and we will need to continually monitor this over the coming weeks,” the agency says.
That scientific uncertainty about the variant’s threat shook European nations that were rushing to ship goods to England in advance of a Dec. 31 Brexit deadline. Under Brexit, which is short for “British exit,” the United Kingdom will leave the European Union on Jan. 31, 2020. Until then, the two sides will come up with new trade and security relationships.
European Union members Austria, Belgium, Bulgaria, France, Germany, Ireland, Italy, and the Netherlands announced travel restrictions hours after Johnson’s speech.
Those restrictions created food uncertainty across the U.K., which imports about a quarter of its food from the EU, according to The New York Times. Long lines of trucks heading to ports in the U.K. came to a standstill on major roads such as the M20 near Kent and the Port of Dover.
Outside Europe, Canada, India, Iran, Israel, Hong Kong, Saudi Arabia, and Turkey banned all incoming flights from the U.K. And more bans could come.
The U.S. reaction
The United States has not imposed any new limits on travel with the United Kingdom, although New York Gov. Andrew Cuomo (D) has requested all passengers bound for John F. Kennedy International Airport from the U.K. be tested before boarding and a new travel ban be placed for Europe. He says the federal government must take action now to avoid a crisis situation like the one New York experienced in March and April.
“The United States has a number of flights coming in from the U.K. each day, and we have done absolutely nothing,” Mr. Cuomo said in a statement on the governor’s webpage. “To me, this is reprehensible because this is what happened in the spring. How many times in life do you have to make the same mistake before you learn?”
Leading U.S. health officials have downplayed the dangers of the virus.
“We don’t know that it’s more dangerous, and very importantly, we have not seen a single mutation yet that would make it evade the vaccine,” U.S. Assistant Secretary of Health and Human Services Adm. Brett Giroir, MD, said Sunday on ABC’s This Week with George Stephanopoulos. “I can’t say that won’t happen in the future, but right now it looks like the vaccine will cover everything that we see.”
Dr. Giroir said the HHS and other U.S. government agencies will monitor the variant.
“Viruses mutate,” he said. “We’ve seen almost 4,000 different mutations among this virus. There is no indication that the mutation right now that they’re talking about is overcoming England.”
Where did the variant come from?
Public Health England says the coronavirus variant had existed in the U.K. since September and circulated at very low levels until mid-November.
“The increase in cases linked to the new variant first came to light in late November when PHE was investigating why infection rates in Kent were not falling despite national restrictions. We then discovered a cluster linked to this variant spreading rapidly into London and Essex,” the agency said.
Public Health England says there’s no evidence the new variant is resistant to the Pfizer-BioNTech vaccine, which is now being given across the country to high-priority groups such as health care workers.
An article in The BMJ, a British medical journal, says the variant was first detected by Covid-19 Genomics UK, a consortium that tests the random genetic sequencing of positive COVID-19 samples around the U.K. The variant cases were mostly in the southeast of England.
A University of Birmingham professor said in a Dec. 15 briefing that the variant accounts for 20% of viruses sequenced in Norfolk, 10% in Essex, and 3% in Suffolk. “There are no data to suggest it had been imported from abroad, so it is likely to have evolved in the U.K.,” he said.
The variant is named VUI-202012/01, for the first “variant under investigation” in December 2020, BMJ says. It’s defined by a set of 17 mutations, with the most significant mutation in the spike protein the virus uses to bind to the human ACE2 receptor.
“Changes in this part of spike protein may, in theory, result in the virus becoming more infectious and spreading more easily between people,” the article says.
The European Centre for Disease Prevention and Control says the variant emerged during the time of year when people usually socialize more.
“There is no indication at this point of increased infection severity associated with the new variant,” the agency said. “A few cases with the new variant have to date been reported by Denmark and the Netherlands and, according to media reports, in Belgium.”
Mr. Johnson announced tighter restrictions on England’s hardest-hit areas, such as the southeast and east of England, where new coronavirus cases have continued to rise. And he said people must cut back on their Christmas socializing.
“In England, those living in tier 4 areas should not mix with anyone outside their own household at Christmas, though support bubbles will remain in place for those at particular risk of loneliness or isolation,” he said.
A version of this article first appeared on WebMD.com.
Researchers have detected a highly contagious coronavirus variant in the United Kingdom, leading Prime Minister Boris Johnson to shut down parts of the country and triggering other nations to impose travel and shipping restrictions on England.
Mr. Johnson held a crisis meeting with ministers Monday after Saturday’s shutdown announcement. The prime minister said in a nationally televised address that this coronavirus variant may be “up to 70% more transmissible than the old variant” and was probably responsible for an increase in cases in southeastern England.
“There is still much we don’t know. While we are fairly certain the variant is transmitted more quickly, there is no evidence to suggest that it is more lethal or causes more severe illness. Equally there is no evidence to suggest the vaccine will be any less effective against the new variant,” he said.
Public Health England says it is working to learn as much about the variant as possible. “We know that mortality is a lagging indicator, and we will need to continually monitor this over the coming weeks,” the agency says.
That scientific uncertainty about the variant’s threat shook European nations that were rushing to ship goods to England in advance of a Dec. 31 Brexit deadline. Under Brexit, which is short for “British exit,” the United Kingdom will leave the European Union on Jan. 31, 2020. Until then, the two sides will come up with new trade and security relationships.
European Union members Austria, Belgium, Bulgaria, France, Germany, Ireland, Italy, and the Netherlands announced travel restrictions hours after Johnson’s speech.
Those restrictions created food uncertainty across the U.K., which imports about a quarter of its food from the EU, according to The New York Times. Long lines of trucks heading to ports in the U.K. came to a standstill on major roads such as the M20 near Kent and the Port of Dover.
Outside Europe, Canada, India, Iran, Israel, Hong Kong, Saudi Arabia, and Turkey banned all incoming flights from the U.K. And more bans could come.
The U.S. reaction
The United States has not imposed any new limits on travel with the United Kingdom, although New York Gov. Andrew Cuomo (D) has requested all passengers bound for John F. Kennedy International Airport from the U.K. be tested before boarding and a new travel ban be placed for Europe. He says the federal government must take action now to avoid a crisis situation like the one New York experienced in March and April.
“The United States has a number of flights coming in from the U.K. each day, and we have done absolutely nothing,” Mr. Cuomo said in a statement on the governor’s webpage. “To me, this is reprehensible because this is what happened in the spring. How many times in life do you have to make the same mistake before you learn?”
Leading U.S. health officials have downplayed the dangers of the virus.
“We don’t know that it’s more dangerous, and very importantly, we have not seen a single mutation yet that would make it evade the vaccine,” U.S. Assistant Secretary of Health and Human Services Adm. Brett Giroir, MD, said Sunday on ABC’s This Week with George Stephanopoulos. “I can’t say that won’t happen in the future, but right now it looks like the vaccine will cover everything that we see.”
Dr. Giroir said the HHS and other U.S. government agencies will monitor the variant.
“Viruses mutate,” he said. “We’ve seen almost 4,000 different mutations among this virus. There is no indication that the mutation right now that they’re talking about is overcoming England.”
Where did the variant come from?
Public Health England says the coronavirus variant had existed in the U.K. since September and circulated at very low levels until mid-November.
“The increase in cases linked to the new variant first came to light in late November when PHE was investigating why infection rates in Kent were not falling despite national restrictions. We then discovered a cluster linked to this variant spreading rapidly into London and Essex,” the agency said.
Public Health England says there’s no evidence the new variant is resistant to the Pfizer-BioNTech vaccine, which is now being given across the country to high-priority groups such as health care workers.
An article in The BMJ, a British medical journal, says the variant was first detected by Covid-19 Genomics UK, a consortium that tests the random genetic sequencing of positive COVID-19 samples around the U.K. The variant cases were mostly in the southeast of England.
A University of Birmingham professor said in a Dec. 15 briefing that the variant accounts for 20% of viruses sequenced in Norfolk, 10% in Essex, and 3% in Suffolk. “There are no data to suggest it had been imported from abroad, so it is likely to have evolved in the U.K.,” he said.
The variant is named VUI-202012/01, for the first “variant under investigation” in December 2020, BMJ says. It’s defined by a set of 17 mutations, with the most significant mutation in the spike protein the virus uses to bind to the human ACE2 receptor.
“Changes in this part of spike protein may, in theory, result in the virus becoming more infectious and spreading more easily between people,” the article says.
The European Centre for Disease Prevention and Control says the variant emerged during the time of year when people usually socialize more.
“There is no indication at this point of increased infection severity associated with the new variant,” the agency said. “A few cases with the new variant have to date been reported by Denmark and the Netherlands and, according to media reports, in Belgium.”
Mr. Johnson announced tighter restrictions on England’s hardest-hit areas, such as the southeast and east of England, where new coronavirus cases have continued to rise. And he said people must cut back on their Christmas socializing.
“In England, those living in tier 4 areas should not mix with anyone outside their own household at Christmas, though support bubbles will remain in place for those at particular risk of loneliness or isolation,” he said.
A version of this article first appeared on WebMD.com.