User login
Prevalence of Cancer in Thyroid Nodules In the Veteran Population (FULL)
Thyroid nodules are identified incidentally in 4% to 10% of the general population in the US.1,2 Clinicians and patients often are concerned about potential malignancy when thyroid nodules are identified because 5% to 15% of nodules will be cancerous.1 The most common form of cancer is papillary carcinoma followed by follicular carcinoma.2 Initially, serum thyroid-stimulating hormone (TSH) levels and thyroid ultrasound are used to evaluate a thyroid nodule because both tests can reveal vital information about malignancy potential.3 Ultrasound characteristics, such as macrocalcifications, hypoechogenicity, absence of halo, increased vascularity, and irregular nodular margins, increase suspicion for malignancy and warrant further investigation.3
Ultrasound-guided fine-needle aspiration (FNA) is the modality of choice for evaluation of thyroid nodules with sensitivity and specificity > 90%.2,4 Most patients receive a definitive diagnosis with this test; however, about 25% of cases are indeterminate based on the Bethesda System and require surgical investigation.3
Currently, it is well accepted clinical practice to refer all nodules > 4 cm for surgical intervention regardless of malignancy risk factors or the mass effect of the nodule.3-6 The preference for surgery—rather than FNA—is because of the notable false negative rate with FNA in larger nodules; studies have described false negative rates for FNA close to 10%.7,8 In contrast, Megwalu recently reported a FNA false negative rate of 0%.9
The risk of malignancy associated with nodule size has been researched for many years, but studies have produced conflicting results. In this retrospective cohort study, the authors compared malignancy rates between patients with nodules ≥ 3 cm and those with nodules < 3 cm.
Methods
The authors performed a retrospective chart review of the medical records of 329 patients presenting for thyroid nodule evaluation found on physical exam or incidentally identified with imaging at the Dayton Veteran Affairs Medical Center from January 2000 to May 2016. Data collection included sex, age, race, personal history of neck radiation treatment, family history of thyroid cancer, personal history of thyroid cancer, hot nodules/Graves disease, abnormal neck lymph nodes, and serum TSH levels. The authors looked for an association between TSH level and cancer. Hot thyroid nodules are known to have low risk of malignancy.
All patients aged 18 to 99 years with a thyroid nodule evaluated with FNA were included in the study. Patients were divided into 2 groups, those with nodules ≥ 3 cm and those with nodules < 3 cm. For nodules requiring subsequent biopsies, only the initial nodule biopsy was included in our study. The 3-cm cutoff was selected based on previous studies.1,5,10 Patients who did not undergo a FNA study were excluded. Indications for surgery were positive FNA results, suspicious imaging, size of nodule, or patient preference.
Means and standard deviations are reported for continuous variables and counts and percentages for categorical variables. We used the Mann-Whitney test for comparisons involving continuous variables with 2 groups and the Kruskal-Wallis test for 4 groups. The chi-square test—corrected for continuity if necessary—was used to compare 2 categorical variables. We used multiple logistic regression to adjust for demographic and clinical variables other than nodule size that were related to malignancy. Inferences were made at the 0.05 level of significance.
Results
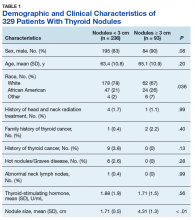
A total of 329 patients with thyroid nodules were identified: 236 were < 3 cm and 93 were ≥ 3 cm. The 2 groups differed on race, with more white patients in the < 3-cm nodule group (78% vs 67%, P = .036) (Table 1).
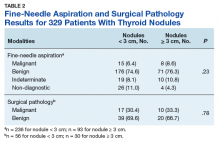
Prevalence of cancer based on FNA in nodules < 3 cm was 6.4% (95% CI, 3.6%–10.3%) and nodules ≥ 3 cm was 8.6% (95% CI, 3.8%–16.2%; P = .23) (Table 2).
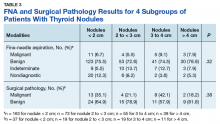
When divided into 4 subgroups, cancer using FNA was found in 35.1% of nodules < 2 cm, 21.1% of nodules 2 cm to < 3 cm, 42.1% of nodules 3 cm to 4 cm, and 18.2% of nodules > 4 cm (P = .32) (Table 3).
Surgical pathology results showed 17 cases of papillary carcinoma in nodules < 3 cm, whereas there were 9 cases of papillary carcinoma and 1 case of follicular carcinoma in nodules > 3 cm. When correlated with the cytology results, 10 cases were reported as benign, 11 were malignant, and 6 samples were non-diagnostic.
There were 30 nondiagnostic FNA samples: 7 patients had surgery, 19 were monitored with serial imaging, 2 were lost to follow-up, and 2 expired for other reasons. Of the 19 patients who were monitored with serial imaging, the nodules were stable and did not require repeat sampling.
Discussion
The authors found no relationship between thyroid nodule size and malignancy over a 16-year period in a veteran population, either with FNA or surgical pathology. The lack of relationship persists when adjusted for the only nonthyroid variable on which the 2 groups differed (race).
The finding of no relationship between larger thyroid nodule size and cancer is consistent with other studies. In a 10-year chart review of 695 patients at Walter Reed Army Medical Center, Burch and colleagues found a malignancy rate of 18.6% but no association between thyroid nodule size and malignancy.11 They concluded that nodules ≥ 4 cm did not increase malignancy risk. In a 3-year retrospective study of 326 patients, Mangister and colleagues reported that the malignancy rate was higher in nodules < 3 cm (48.4%) compared with nodules ≥ 3 cm (33.3%).10 This study concluded that the malignancy potential of thyroid nodules peaked at 2 cm and decreased at > 3 cm. Kamran and colleagues reported a nonlinear relationship between nodule size and malignancy with a threshold of 2 cm, beyond which there was no increased risk of malignancy.1
Conversely, in a prospective study Kuru and colleagues followed 571 patients who had undergone thyroidectomy and found that nodules ≥ 4 cm were associated with increased malignancy risk compared with nodules < 4 cm. However, with a cutoff of 3 cm there was no relationship.5 Discrepancies among studies might be because of variability in patient demographics and the prevalence of thyroid cancer in a specific institution. Although the majority of thyroid nodules are seen in females, the current study’s population was predominantly male and entirely veteran. Consequently, interpretation of these studies highlight the need to individualize clinical decision-making for each patient.
Limitations
This study has several limitations. It was conducted at a single institution with a group of veterans, which limits the ability to generalize its results to the general population. Second, data omissions are likely in retrospective chart reviews, and ensuring accuracy of data collection could be challenging. Third, all thyroid nodules found to be benign with cytology did not undergo surgical intervention to confirm the diagnosis; therefore, only 93 of 329 nodules were evaluated with the definitive diagnostic test. Therefore, selection bias was introduced into the nodule size comparisons when surgical intervention was used to measure the outcome. However, because false negative rates for FNA is low, likely few malignant nodules were missed. In addition, all patients with thyroid nodules are not referred for surgery because of potential complications.
Conclusion
This study strongly suggests there is no increased or decreased cancer risk for thyroid nodules ≥ 3 cm compared with those < 3 cm. Current clinical practice is to refer patients with larger nodules for surgical evaluation. In a large systemic review, Shin and colleagues reported higher pretest probability of malignancy in larger nodules and recommended consideration of surgical intervention for nodules > 3 cm because of false negatives and concerns for diagnostic inaccuracy with FNA.8 Although data were mixed, Shin and colleagues reported higher incidence of false negative FNA results in larger nodules.8 Given the authors’ findings and earlier conflicting results, the decision for surgical intervention cannot be made solely on nodule size and requires consideration of additional factors including FNA results, nodule characteristics, patient risk factors, and patient preference.
1. Kamran SC, Marqusee E, Kim MI, et al. Thyroid nodule size and prediction of cancer. J Clin Endocrinol Metab. 2013;98(2):564-570.
2. Haugen BR, Alexander EK, Bible KC, et al. 2015 American Thyroid Association Management guidelines for adult patients with thyroid nodules and differentiated thyroid cancer: The American Thyroid Association Guidelines Task Force on Thyroid Nodules and Differentiated Thyroid Cancer. Thyroid. 2016;26(1):1-33.
3. Popoveniuc G, Jonklaas J. Thyroid nodules. Med Clin North Am. 2012;96(2):329-349.
4. Amrikachi M, Ramzy I, Rubenfeld S, Wheeler TM. Accuracy of fine needle aspiration of thyroid. Arch Pathol Lab Med. 2001;125(4):484-488.
5. Kuru B, Gulcelik NE, Gulcelik MA, Dincer H. Predictive index for carcinoma of thyroid nodules and its integration with fine-needle aspiration cytology. Head Neck. 2009;31(7):856-866.
6. Kim JH, Kim NK, Oh YL, et al. The validity of ultrasonography-guided fine needle aspiration biopsy in thyroid nodules 4 cm or larger depends on ultrasound characteristics. Endocrinol Metab (Seoul). 2014;29(4):545-552.
7. Wharry LI, McCoy KL, Stang MT, et al. Thyroid nodules (≥4 cm): can ultrasound and cytology reliably exclude cancer? World J Surg. 2014;38(3):614-621.
8. Pinchot SN, Al-Wagih H, Schaefer S, Sippel R, Chen H. Accuracy of fine needle aspiration biopsy for predicting neoplasm or carcinoma in thyroid nodules 4 cm or larger. Arch Surg. 2009;144(7):649-655.
9. Megwalu UC. Risk of malignancy in thyroid nodules 4 cm or larger. Endocrinol Metab (Seoul). 2017;32(1):77-82.
10. Magister MJ, Chaikhoutdinov I, Schaefer E, et al. Association of thyroid nodule size and Bethesda class with rate of malignant disease. JAMA Otolaryngol Head Neck Surg. 2015;141(12):1089-1095.
11. Shrestha M, Crothers BA, Burch HB. The impact of thyroid nodule size on the risk of malignancy and accuracy of fine needle aspiration: a 10-year study from a single institution. Thyroid. 2012;22(12):1251-1256.
Thyroid nodules are identified incidentally in 4% to 10% of the general population in the US.1,2 Clinicians and patients often are concerned about potential malignancy when thyroid nodules are identified because 5% to 15% of nodules will be cancerous.1 The most common form of cancer is papillary carcinoma followed by follicular carcinoma.2 Initially, serum thyroid-stimulating hormone (TSH) levels and thyroid ultrasound are used to evaluate a thyroid nodule because both tests can reveal vital information about malignancy potential.3 Ultrasound characteristics, such as macrocalcifications, hypoechogenicity, absence of halo, increased vascularity, and irregular nodular margins, increase suspicion for malignancy and warrant further investigation.3
Ultrasound-guided fine-needle aspiration (FNA) is the modality of choice for evaluation of thyroid nodules with sensitivity and specificity > 90%.2,4 Most patients receive a definitive diagnosis with this test; however, about 25% of cases are indeterminate based on the Bethesda System and require surgical investigation.3
Currently, it is well accepted clinical practice to refer all nodules > 4 cm for surgical intervention regardless of malignancy risk factors or the mass effect of the nodule.3-6 The preference for surgery—rather than FNA—is because of the notable false negative rate with FNA in larger nodules; studies have described false negative rates for FNA close to 10%.7,8 In contrast, Megwalu recently reported a FNA false negative rate of 0%.9
The risk of malignancy associated with nodule size has been researched for many years, but studies have produced conflicting results. In this retrospective cohort study, the authors compared malignancy rates between patients with nodules ≥ 3 cm and those with nodules < 3 cm.
Methods
The authors performed a retrospective chart review of the medical records of 329 patients presenting for thyroid nodule evaluation found on physical exam or incidentally identified with imaging at the Dayton Veteran Affairs Medical Center from January 2000 to May 2016. Data collection included sex, age, race, personal history of neck radiation treatment, family history of thyroid cancer, personal history of thyroid cancer, hot nodules/Graves disease, abnormal neck lymph nodes, and serum TSH levels. The authors looked for an association between TSH level and cancer. Hot thyroid nodules are known to have low risk of malignancy.
All patients aged 18 to 99 years with a thyroid nodule evaluated with FNA were included in the study. Patients were divided into 2 groups, those with nodules ≥ 3 cm and those with nodules < 3 cm. For nodules requiring subsequent biopsies, only the initial nodule biopsy was included in our study. The 3-cm cutoff was selected based on previous studies.1,5,10 Patients who did not undergo a FNA study were excluded. Indications for surgery were positive FNA results, suspicious imaging, size of nodule, or patient preference.
Means and standard deviations are reported for continuous variables and counts and percentages for categorical variables. We used the Mann-Whitney test for comparisons involving continuous variables with 2 groups and the Kruskal-Wallis test for 4 groups. The chi-square test—corrected for continuity if necessary—was used to compare 2 categorical variables. We used multiple logistic regression to adjust for demographic and clinical variables other than nodule size that were related to malignancy. Inferences were made at the 0.05 level of significance.
Results
A total of 329 patients with thyroid nodules were identified: 236 were < 3 cm and 93 were ≥ 3 cm. The 2 groups differed on race, with more white patients in the < 3-cm nodule group (78% vs 67%, P = .036) (Table 1).
Prevalence of cancer based on FNA in nodules < 3 cm was 6.4% (95% CI, 3.6%–10.3%) and nodules ≥ 3 cm was 8.6% (95% CI, 3.8%–16.2%; P = .23) (Table 2).
When divided into 4 subgroups, cancer using FNA was found in 35.1% of nodules < 2 cm, 21.1% of nodules 2 cm to < 3 cm, 42.1% of nodules 3 cm to 4 cm, and 18.2% of nodules > 4 cm (P = .32) (Table 3).
Surgical pathology results showed 17 cases of papillary carcinoma in nodules < 3 cm, whereas there were 9 cases of papillary carcinoma and 1 case of follicular carcinoma in nodules > 3 cm. When correlated with the cytology results, 10 cases were reported as benign, 11 were malignant, and 6 samples were non-diagnostic.
There were 30 nondiagnostic FNA samples: 7 patients had surgery, 19 were monitored with serial imaging, 2 were lost to follow-up, and 2 expired for other reasons. Of the 19 patients who were monitored with serial imaging, the nodules were stable and did not require repeat sampling.
Discussion
The authors found no relationship between thyroid nodule size and malignancy over a 16-year period in a veteran population, either with FNA or surgical pathology. The lack of relationship persists when adjusted for the only nonthyroid variable on which the 2 groups differed (race).
The finding of no relationship between larger thyroid nodule size and cancer is consistent with other studies. In a 10-year chart review of 695 patients at Walter Reed Army Medical Center, Burch and colleagues found a malignancy rate of 18.6% but no association between thyroid nodule size and malignancy.11 They concluded that nodules ≥ 4 cm did not increase malignancy risk. In a 3-year retrospective study of 326 patients, Mangister and colleagues reported that the malignancy rate was higher in nodules < 3 cm (48.4%) compared with nodules ≥ 3 cm (33.3%).10 This study concluded that the malignancy potential of thyroid nodules peaked at 2 cm and decreased at > 3 cm. Kamran and colleagues reported a nonlinear relationship between nodule size and malignancy with a threshold of 2 cm, beyond which there was no increased risk of malignancy.1
Conversely, in a prospective study Kuru and colleagues followed 571 patients who had undergone thyroidectomy and found that nodules ≥ 4 cm were associated with increased malignancy risk compared with nodules < 4 cm. However, with a cutoff of 3 cm there was no relationship.5 Discrepancies among studies might be because of variability in patient demographics and the prevalence of thyroid cancer in a specific institution. Although the majority of thyroid nodules are seen in females, the current study’s population was predominantly male and entirely veteran. Consequently, interpretation of these studies highlight the need to individualize clinical decision-making for each patient.
Limitations
This study has several limitations. It was conducted at a single institution with a group of veterans, which limits the ability to generalize its results to the general population. Second, data omissions are likely in retrospective chart reviews, and ensuring accuracy of data collection could be challenging. Third, all thyroid nodules found to be benign with cytology did not undergo surgical intervention to confirm the diagnosis; therefore, only 93 of 329 nodules were evaluated with the definitive diagnostic test. Therefore, selection bias was introduced into the nodule size comparisons when surgical intervention was used to measure the outcome. However, because false negative rates for FNA is low, likely few malignant nodules were missed. In addition, all patients with thyroid nodules are not referred for surgery because of potential complications.
Conclusion
This study strongly suggests there is no increased or decreased cancer risk for thyroid nodules ≥ 3 cm compared with those < 3 cm. Current clinical practice is to refer patients with larger nodules for surgical evaluation. In a large systemic review, Shin and colleagues reported higher pretest probability of malignancy in larger nodules and recommended consideration of surgical intervention for nodules > 3 cm because of false negatives and concerns for diagnostic inaccuracy with FNA.8 Although data were mixed, Shin and colleagues reported higher incidence of false negative FNA results in larger nodules.8 Given the authors’ findings and earlier conflicting results, the decision for surgical intervention cannot be made solely on nodule size and requires consideration of additional factors including FNA results, nodule characteristics, patient risk factors, and patient preference.
Thyroid nodules are identified incidentally in 4% to 10% of the general population in the US.1,2 Clinicians and patients often are concerned about potential malignancy when thyroid nodules are identified because 5% to 15% of nodules will be cancerous.1 The most common form of cancer is papillary carcinoma followed by follicular carcinoma.2 Initially, serum thyroid-stimulating hormone (TSH) levels and thyroid ultrasound are used to evaluate a thyroid nodule because both tests can reveal vital information about malignancy potential.3 Ultrasound characteristics, such as macrocalcifications, hypoechogenicity, absence of halo, increased vascularity, and irregular nodular margins, increase suspicion for malignancy and warrant further investigation.3
Ultrasound-guided fine-needle aspiration (FNA) is the modality of choice for evaluation of thyroid nodules with sensitivity and specificity > 90%.2,4 Most patients receive a definitive diagnosis with this test; however, about 25% of cases are indeterminate based on the Bethesda System and require surgical investigation.3
Currently, it is well accepted clinical practice to refer all nodules > 4 cm for surgical intervention regardless of malignancy risk factors or the mass effect of the nodule.3-6 The preference for surgery—rather than FNA—is because of the notable false negative rate with FNA in larger nodules; studies have described false negative rates for FNA close to 10%.7,8 In contrast, Megwalu recently reported a FNA false negative rate of 0%.9
The risk of malignancy associated with nodule size has been researched for many years, but studies have produced conflicting results. In this retrospective cohort study, the authors compared malignancy rates between patients with nodules ≥ 3 cm and those with nodules < 3 cm.
Methods
The authors performed a retrospective chart review of the medical records of 329 patients presenting for thyroid nodule evaluation found on physical exam or incidentally identified with imaging at the Dayton Veteran Affairs Medical Center from January 2000 to May 2016. Data collection included sex, age, race, personal history of neck radiation treatment, family history of thyroid cancer, personal history of thyroid cancer, hot nodules/Graves disease, abnormal neck lymph nodes, and serum TSH levels. The authors looked for an association between TSH level and cancer. Hot thyroid nodules are known to have low risk of malignancy.
All patients aged 18 to 99 years with a thyroid nodule evaluated with FNA were included in the study. Patients were divided into 2 groups, those with nodules ≥ 3 cm and those with nodules < 3 cm. For nodules requiring subsequent biopsies, only the initial nodule biopsy was included in our study. The 3-cm cutoff was selected based on previous studies.1,5,10 Patients who did not undergo a FNA study were excluded. Indications for surgery were positive FNA results, suspicious imaging, size of nodule, or patient preference.
Means and standard deviations are reported for continuous variables and counts and percentages for categorical variables. We used the Mann-Whitney test for comparisons involving continuous variables with 2 groups and the Kruskal-Wallis test for 4 groups. The chi-square test—corrected for continuity if necessary—was used to compare 2 categorical variables. We used multiple logistic regression to adjust for demographic and clinical variables other than nodule size that were related to malignancy. Inferences were made at the 0.05 level of significance.
Results
A total of 329 patients with thyroid nodules were identified: 236 were < 3 cm and 93 were ≥ 3 cm. The 2 groups differed on race, with more white patients in the < 3-cm nodule group (78% vs 67%, P = .036) (Table 1).
Prevalence of cancer based on FNA in nodules < 3 cm was 6.4% (95% CI, 3.6%–10.3%) and nodules ≥ 3 cm was 8.6% (95% CI, 3.8%–16.2%; P = .23) (Table 2).
When divided into 4 subgroups, cancer using FNA was found in 35.1% of nodules < 2 cm, 21.1% of nodules 2 cm to < 3 cm, 42.1% of nodules 3 cm to 4 cm, and 18.2% of nodules > 4 cm (P = .32) (Table 3).
Surgical pathology results showed 17 cases of papillary carcinoma in nodules < 3 cm, whereas there were 9 cases of papillary carcinoma and 1 case of follicular carcinoma in nodules > 3 cm. When correlated with the cytology results, 10 cases were reported as benign, 11 were malignant, and 6 samples were non-diagnostic.
There were 30 nondiagnostic FNA samples: 7 patients had surgery, 19 were monitored with serial imaging, 2 were lost to follow-up, and 2 expired for other reasons. Of the 19 patients who were monitored with serial imaging, the nodules were stable and did not require repeat sampling.
Discussion
The authors found no relationship between thyroid nodule size and malignancy over a 16-year period in a veteran population, either with FNA or surgical pathology. The lack of relationship persists when adjusted for the only nonthyroid variable on which the 2 groups differed (race).
The finding of no relationship between larger thyroid nodule size and cancer is consistent with other studies. In a 10-year chart review of 695 patients at Walter Reed Army Medical Center, Burch and colleagues found a malignancy rate of 18.6% but no association between thyroid nodule size and malignancy.11 They concluded that nodules ≥ 4 cm did not increase malignancy risk. In a 3-year retrospective study of 326 patients, Mangister and colleagues reported that the malignancy rate was higher in nodules < 3 cm (48.4%) compared with nodules ≥ 3 cm (33.3%).10 This study concluded that the malignancy potential of thyroid nodules peaked at 2 cm and decreased at > 3 cm. Kamran and colleagues reported a nonlinear relationship between nodule size and malignancy with a threshold of 2 cm, beyond which there was no increased risk of malignancy.1
Conversely, in a prospective study Kuru and colleagues followed 571 patients who had undergone thyroidectomy and found that nodules ≥ 4 cm were associated with increased malignancy risk compared with nodules < 4 cm. However, with a cutoff of 3 cm there was no relationship.5 Discrepancies among studies might be because of variability in patient demographics and the prevalence of thyroid cancer in a specific institution. Although the majority of thyroid nodules are seen in females, the current study’s population was predominantly male and entirely veteran. Consequently, interpretation of these studies highlight the need to individualize clinical decision-making for each patient.
Limitations
This study has several limitations. It was conducted at a single institution with a group of veterans, which limits the ability to generalize its results to the general population. Second, data omissions are likely in retrospective chart reviews, and ensuring accuracy of data collection could be challenging. Third, all thyroid nodules found to be benign with cytology did not undergo surgical intervention to confirm the diagnosis; therefore, only 93 of 329 nodules were evaluated with the definitive diagnostic test. Therefore, selection bias was introduced into the nodule size comparisons when surgical intervention was used to measure the outcome. However, because false negative rates for FNA is low, likely few malignant nodules were missed. In addition, all patients with thyroid nodules are not referred for surgery because of potential complications.
Conclusion
This study strongly suggests there is no increased or decreased cancer risk for thyroid nodules ≥ 3 cm compared with those < 3 cm. Current clinical practice is to refer patients with larger nodules for surgical evaluation. In a large systemic review, Shin and colleagues reported higher pretest probability of malignancy in larger nodules and recommended consideration of surgical intervention for nodules > 3 cm because of false negatives and concerns for diagnostic inaccuracy with FNA.8 Although data were mixed, Shin and colleagues reported higher incidence of false negative FNA results in larger nodules.8 Given the authors’ findings and earlier conflicting results, the decision for surgical intervention cannot be made solely on nodule size and requires consideration of additional factors including FNA results, nodule characteristics, patient risk factors, and patient preference.
1. Kamran SC, Marqusee E, Kim MI, et al. Thyroid nodule size and prediction of cancer. J Clin Endocrinol Metab. 2013;98(2):564-570.
2. Haugen BR, Alexander EK, Bible KC, et al. 2015 American Thyroid Association Management guidelines for adult patients with thyroid nodules and differentiated thyroid cancer: The American Thyroid Association Guidelines Task Force on Thyroid Nodules and Differentiated Thyroid Cancer. Thyroid. 2016;26(1):1-33.
3. Popoveniuc G, Jonklaas J. Thyroid nodules. Med Clin North Am. 2012;96(2):329-349.
4. Amrikachi M, Ramzy I, Rubenfeld S, Wheeler TM. Accuracy of fine needle aspiration of thyroid. Arch Pathol Lab Med. 2001;125(4):484-488.
5. Kuru B, Gulcelik NE, Gulcelik MA, Dincer H. Predictive index for carcinoma of thyroid nodules and its integration with fine-needle aspiration cytology. Head Neck. 2009;31(7):856-866.
6. Kim JH, Kim NK, Oh YL, et al. The validity of ultrasonography-guided fine needle aspiration biopsy in thyroid nodules 4 cm or larger depends on ultrasound characteristics. Endocrinol Metab (Seoul). 2014;29(4):545-552.
7. Wharry LI, McCoy KL, Stang MT, et al. Thyroid nodules (≥4 cm): can ultrasound and cytology reliably exclude cancer? World J Surg. 2014;38(3):614-621.
8. Pinchot SN, Al-Wagih H, Schaefer S, Sippel R, Chen H. Accuracy of fine needle aspiration biopsy for predicting neoplasm or carcinoma in thyroid nodules 4 cm or larger. Arch Surg. 2009;144(7):649-655.
9. Megwalu UC. Risk of malignancy in thyroid nodules 4 cm or larger. Endocrinol Metab (Seoul). 2017;32(1):77-82.
10. Magister MJ, Chaikhoutdinov I, Schaefer E, et al. Association of thyroid nodule size and Bethesda class with rate of malignant disease. JAMA Otolaryngol Head Neck Surg. 2015;141(12):1089-1095.
11. Shrestha M, Crothers BA, Burch HB. The impact of thyroid nodule size on the risk of malignancy and accuracy of fine needle aspiration: a 10-year study from a single institution. Thyroid. 2012;22(12):1251-1256.
1. Kamran SC, Marqusee E, Kim MI, et al. Thyroid nodule size and prediction of cancer. J Clin Endocrinol Metab. 2013;98(2):564-570.
2. Haugen BR, Alexander EK, Bible KC, et al. 2015 American Thyroid Association Management guidelines for adult patients with thyroid nodules and differentiated thyroid cancer: The American Thyroid Association Guidelines Task Force on Thyroid Nodules and Differentiated Thyroid Cancer. Thyroid. 2016;26(1):1-33.
3. Popoveniuc G, Jonklaas J. Thyroid nodules. Med Clin North Am. 2012;96(2):329-349.
4. Amrikachi M, Ramzy I, Rubenfeld S, Wheeler TM. Accuracy of fine needle aspiration of thyroid. Arch Pathol Lab Med. 2001;125(4):484-488.
5. Kuru B, Gulcelik NE, Gulcelik MA, Dincer H. Predictive index for carcinoma of thyroid nodules and its integration with fine-needle aspiration cytology. Head Neck. 2009;31(7):856-866.
6. Kim JH, Kim NK, Oh YL, et al. The validity of ultrasonography-guided fine needle aspiration biopsy in thyroid nodules 4 cm or larger depends on ultrasound characteristics. Endocrinol Metab (Seoul). 2014;29(4):545-552.
7. Wharry LI, McCoy KL, Stang MT, et al. Thyroid nodules (≥4 cm): can ultrasound and cytology reliably exclude cancer? World J Surg. 2014;38(3):614-621.
8. Pinchot SN, Al-Wagih H, Schaefer S, Sippel R, Chen H. Accuracy of fine needle aspiration biopsy for predicting neoplasm or carcinoma in thyroid nodules 4 cm or larger. Arch Surg. 2009;144(7):649-655.
9. Megwalu UC. Risk of malignancy in thyroid nodules 4 cm or larger. Endocrinol Metab (Seoul). 2017;32(1):77-82.
10. Magister MJ, Chaikhoutdinov I, Schaefer E, et al. Association of thyroid nodule size and Bethesda class with rate of malignant disease. JAMA Otolaryngol Head Neck Surg. 2015;141(12):1089-1095.
11. Shrestha M, Crothers BA, Burch HB. The impact of thyroid nodule size on the risk of malignancy and accuracy of fine needle aspiration: a 10-year study from a single institution. Thyroid. 2012;22(12):1251-1256.
Significance of Bacteriuria
Staphylococcus aureus (SA) infection can cause a wide range of clinical syndromes, from folliculitis to life‐threatening endocarditis. Further, SA is second only to S. epidermidis as a cause of bacteremia in hospitalized patients.1, 2 Recent single‐institution studies suggests that SA could be the most frequent cause of nosocomial bacteremia,3, 4 but this needs to be validated in multicenter studies. SA bacteremia (SAB) is often complicated by hematogenous seeding into deep tissues or prosthetic material. The association of future hardware infection following SAB is well documented.5, 6 One study showed that SAB can precede and be associated with prosthetic joint infections in up to 34% of cases.6 Intravascular cardiac devices can also be infected by SAB, with rates from 28% to 75% depending on how early the bacteremia occurred in relation to the implantation of the device.5 Risk stratification for these complications is a clinical challenge. Fowler et al.7 postulated some clinical identifiers of complicated SAB; however, predicting which patients will develop a complication from SAB remains very difficult. Muder et al.8 demonstrated that the presence of SA bacteriuria (SABU) correlates with subsequent SAB, but a possible association of SABU with complicated bacteremia was not examined. A more recent study from Huggan et al.9 has suggested a possible association between SABU and poor clinical outcomes in adults with SAB.
We hypothesized that the presence of SABU would identify those patients at increased risk of complications from SAB. SABU may be a practical, economical, and readily available predictor of complicated SAB. Those patients at higher risk for complications may require a more aggressive diagnostic and therapeutic approach.
Methods
We conducted a retrospective cohort study of SAB patients with and without SA in the urine to investigate the association between SABU and the outcomes of the complications and mortality.
The study was conducted at Miami Valley Hospital (MVH, Dayton, OH), an 848‐bed, level 1 trauma center with 69 intensive care unit (ICU) beds. MVH is a community teaching hospital affiliated with Wright State University Boonshoft School of Medicine and averages 35,000 admissions per year. The same microbiology laboratory (Compunet Clinical Laboratories) processed all the blood and urine culture specimens of the patients in this study.
The inclusion criteria were as follows: 1) admission to MVH between January 1, 2004 and December 31, 2007 with a documented episode of SAB (at least 1 positive blood culture); and 2) a documented urine culture within 7 days of the episode of SAB. Patients without a documented urine culture or with inadequate/emncomplete treatment for SAB were excluded. A total of 118 patients were included based on the presence of a positive blood culture for SA and the presence of a documented urine culture. Patient electronic and paper records were reviewed by 3 of the investigators (E.V.P.‐J., S.D.B., and W.B.B.). Patients subsequently admitted to MVH and to MVH's companion medical center in Dayton, Good Samaritan Hospital, were followed through the electronic medical record common to both institutions.
Study patients were divided into 2 cohorts. One cohort included the patients with a urine culture that grew SA, either methicillin‐resistant SA (MRSA) or methicillin‐susceptible SA (MSSA). The other cohort included patients who had either a negative urine culture or a positive urine culture with organisms other than SA. The age, sex, date of admission, length of stay, and duration of follow‐up were recorded for each patient. Clinical variables included blood culture and urine culture results, presence of intravenous catheters, antibiotic therapy and duration, presence of comorbidities, and clinical outcomes (complications and death).
The primary outcome was complications during hospital admission. The 8 complications investigated were as follows: endocarditis, osteomyelitis, septic arthritis, thrombophlebitis, septic shock, septic embolism/abscess, persistent SAB (lasting more than 5 days after starting adequate SA treatment), and recurrent SAB. In addition, the 2 groups were compared on: 1) any complication, 2) average complications, 3) early complications (ie, within the current hospital admission), and 4) delayed complications (ie, complications diagnosed on subsequent admissions).
Statistical Methods
Means standard deviations (SDs) are reported for continuous variables while frequencies and percents are reported for categorical variables. The independent samples t test for continuous variables and the chi square test or Fisher's exact test for categorical variables were used to compare the two cohorts. Inferences were made at the 0.05 level of significance with no correction for multiple comparisons. SPSS 11.0 software (SPSS, Inc., Chicago, IL) was used for all analyses.
Results
Of the 118 patients, 58 were female (49.2%) and 60 male (50.8%). The age of the patients was 63.3 16.7 years (mean SD). The length of hospital stay was 19.3 17.0 days, and the duration of follow up was 8.3 5.7 months. MRSA was isolated in 75 patients (63.6%) and MSSA in 43 patients (36.4%). In the 28 patients with SA in urine cultures, MRSA was found more frequently than MSSA (20 vs. 8 patients). The acquisition of SAB was equally divided among outpatient (35.6%), healthcare‐associated (30.5%), and hospital‐acquired (33.9%) settings.
Table 1 shows that the group with SABU did not differ from the group without SABU in age (66 years vs. 62 years; P = 0.29), sex (43% male vs. 53% male; P = 0.33), length of hospital stay (18 days vs. 20 days; P = 0.59), and duration of follow‐up (6.6 months vs. 8.8 months; P = 0.064). The 2 cohorts also did not differ on the proportion with MRSA bacteremia (71% vs. 61%; P = 0.32), origin of SAB (P = 0.12), and the presence of comorbidities (diabetes mellitus, cardiomyopathy/congestive heart failure, malignancy, renal disease, and immunosuppression) (all P values > 0.30).
| Characteristic | S. aureus Bacteriuria (n = 28) | No S. aureus Bacteriuria (n = 90) | P Value* |
|---|---|---|---|
| |||
| Age (years) (mean SD) | 66.3 16.3 | 62.4 16.8 | 0.29 |
| Male sex (n [%[) | 12 (42.9) | 48 (53.3) | 0.33 |
| Length of stay (days) (mean SD) | 17.8 16.1 | 19.7 17.3 | 0.59 |
| Follow‐up (months) (mean SD) | 6.6 5.3 | 8.8 5.7 | 0.064 |
| Blood culture (n [%]) | |||
| MRSA | 20 (71.4) | 55 (61.1) | 0.32 |
| MSSA | 8 (28.6) | 35 (38.9) | |
| Origin of the bacteremia [n (%)] | 0.12 | ||
| Community‐acquired | 13 (46.4) | 29 (32.2) | |
| Healthcare‐acquired | 10 (35.7) | 26 (28.9) | |
| Hospital‐acquired | 5 (17.9) | 35 (38.9) | |
| Comorbidities (n [%]) | |||
| DM | 11 (39.3) | 38 (42.2) | 0.78 |
| CHF | 5 (17.9) | 20 (22.2) | 0.62 |
| Cancer | 7 (25.0) | 15 (16.7) | 0.32 |
| ESRD | 4 (14.3) | 12 (13.3) | 1.00 |
| Immunosuppression | 6 (21.4) | 15 (16.7) | 0.58 |
| Patients lost to follow‐up (n [%]) | 5 (17.8) | 8 (8.8) | 0.19 |
Table 2 shows that patients in the SABU group were nearly twice as likely to have a complication as the group without SABU (64% vs. 33%; P = 0.004) and had a higher mean number of complications (0.89 vs. 0.48; P = 0.016). Patients in the SABU group also were more likely to have early complications (64% vs. 23%; P < 0.001) but no more likely to have a delayed complication (14% vs. 12%; P = 0.75). Of the 8 specific complications evaluated, the 2 groups differed only on the presence of septic shock, with the SABU group having 3 times more patients with this complication (21% vs. 7%; P = 0.035). Also, a higher proportion of patients died in the SABU group (32.1% vs. 14.4%; P = 0.036).
| Outcome | S. aureus Bacteriuria (n = 28) | No S. aureus Bacteriuria (n = 90) | P Value* |
|---|---|---|---|
| |||
| Any complication (n [%]) | 18 (64.3) | 30 (33.3) | 0.004 |
| Average complications (mean SD) | 0.89 0.83 | 0.48 0.77 | 0.016 |
| Timing of complication (n [%]) | |||
| Early | 18 (64.3) | 21 (23.3) | <0.001 |
| Delayed | 4 (14.3) | 11 (12.2) | 0.75 |
| By specific complication, n (%) | |||
| Endocarditis | 1 (3.6) | 5 (5.6) | 1.00 |
| Osteomyelitis | 3 (10.7) | 5 (5.6) | 0.39 |
| Septic arthritis | 2 (7.1) | 3 (3.3) | 0.59 |
| Thrombophlebitis | 1 (3.6) | 3 (3.3) | 1.00 |
| Septic shock | 6 (21.4) | 6 (6.7) | 0.035 |
| Septic embolism/abscess | 6 (21.4) | 10 (11.1) | 0.21 |
| Persistent SAB | 3 (10.7) | 3 (3.3) | 0.14 |
| Recurrent SAB | 3 (10.7) | 8 (8.9) | 0.72 |
| Death (n [%]) | 9 (32.1) | 13 (14.4) | 0.036 |
Patients with MRSA (n = 75) and those with MSSA (n = 43) did not differ on any complication, average complications, early or late complications, or 7 of the specific complications (data not shown). Only with thrombophlebitis did the 2 groups differ; the MSSA group had 4 (9.3%) patients with this complication while none in the MRSA group were affected (P = 0.016).
Discussion
In our retrospective analysis, SAB with concomitant SABU was associated with more severe disease, complications, and death. Compared to SAB patients without SA in the urine, those with SAB and SA in the urine had more total complications and more early complications, especially septic shock. Further, the proportion of deaths in the SABU cohort was more than twice as high (32% vs. 14%). Therefore, the presence of SABU in patients with SAB could potentially be a useful predictor of complicated SAB and death.
The relationship between SABU and early complications and death remained after excluding the complication of septic shock/need for vasopressors from the analysis (data not shown). The lack of relationship between SABU and delayed complications might have been due to the adequacy of treatment for SAB. Appropriateness of therapy, a criterion for patient inclusion, may have lessened the likelihood of an insufficient treatment plan causing complications. Those patients with MRSA did not differ from those with MSSA on the mean number of complications or early and delayed complications. A greater proportion of MSSA patients had thrombophlebitis than MRSA patients.
Other investigations have identified predictors of mortality or complications from SAB,7, 912 but SABU was not included as a variable in most of these studies. Fowler et al.7 proposed a prognostic model of complicated SAB using the predictors from their study; community acquisition of organisms, persistent bacteremia, persistent fever over 72 hours, and skin examination suggestive of an acute systemic infection. Muder et al.8 reported a relationship between SABU and subsequent SAB, but they did not examine the association between SABU and the risk of complicated SAB. Huggan et al.9 found that concomitant SABU is associated with ICU admission and increased in‐hospital mortality in patients with SAB.
SAB patients with SABU may be at risk for early complications. Consequently, such patients may warrant more aggressive evaluation and treatment. Further, SABU in patients with SAB may be indicative of an endocarditis‐like condition. SA is rarely isolated from the urinary tract as a uropathogen, although it may colonize indwelling catheters and may cause catheter‐related urinary tract infections.13, 14 Thus, when present in urine, SA could be a marker of deep tissue dissemination with the potential to cause complications. Guidelines for the management of intravascular device‐associated bacteremia have been published by the Infectious Diseases Society of America (IDSA) and other organizations,15, 16 and recent studies have demonstrated the effectiveness of newer agents for the management of SAB.17 Nevertheless, there is still controversy regarding some aspects of the management of SAB (eg, duration of therapy, criteria for echocardiographic evaluation, role of combination therapy). The presence of SABU, the marker evaluated in our study, may be an additional factor to consider when deciding upon duration of therapy and whether to obtain echocardiography or other imaging.
Our study was limited by its retrospective nature. Patient records were not always complete. For example, not all patients had echocardiography to evaluate for endocarditis or venous ultrasound to evaluate for septic thrombophlebitis. Also, the presence (or proper removal) of intravascular or urinary catheters could not be documented reliably in all patients. In addition, the 7‐day cutoff for obtaining urine cultures may have been too lenient, leading to underdiagnosis of bacteriuria. Finally, while 13 patients were lost to follow‐up, the 2 groups (SABU and No SABU) did not differ in the proportion lost.
In conclusion, our study found that SABU may be a useful predictor of complicated SAB and death. SAB patients with SABU may be at risk for more and earlier complications. These patients may need closer monitoring due to the higher risk of septic shock and death. Additional therapeutic and management recommendations might include: 1) longer duration of therapy even if a removable source of the bacteremia is identified; 2) more frequent and better supervised follow‐up; and 3) imaging studies including either computed tomography (CT) scans or ultrasound for thorough evaluation of complications. Prospective studies including randomized controlled trials are required before implementing these suggested diagnostic and therapeutic recommendations.
Acknowledgements
The authors thank and acknowledge Logan McCool and Adam Woiwood for their administrative contributions to the study. E.V.P.‐J., as the principal investigator, had full access to all of the data in the study and takes responsibility for the integrity of the data and the accuracy of the data analysis.
- ,,, et al.Prevalence and drug susceptibility of pathogens causing bloodstream infections in northern Italy: a two‐year study in 16 hospitals.Eur J Clin Microbiol Infect Dis.2002;21(12):849–855.
- ,,, et al.Nosocomial bloodstream infections in ICU and non‐ICU patients.Am J Infec Control.2005;33(6):333–340.
- ,,, et al.Age‐ and sex‐associated trends in bloodstream infection: a population‐based study in Olmsted County, Minnesota.Arch Intern Med.2007;167(8):834–839.
- ,,.Bloodstream infections in a geriatric cohort: a population‐based study.Am J Med.2007;120(12):1078–1883.
- ,,, et al.Staphylococcus aureus bacteremia in patients with permanent pacemakers or implantable cardioverter‐defibrillators.Circulation.2001;104(9):1029–1033.
- ,,, et al.Infection of orthopedic prostheses after Staphylococcus aureus bacteremia.Clin Infect Dis.2001;32(4):647–649.
- ,,, et al.Clinical identifiers of complicated Staphylococcus aureus bacteremia.Arch Intern Med.2003;163(17):2066–2072.
- ,,, et al.Isolation of Staphylococcus aureus from the urinary tract: association of isolation with symptomatic UTI and subsequent staphylococcal bacteremia.Clin Infect Dis.2006;42(1):46–50.
- ,,, et al.Concomitant Staphylococcus aureus bacteriuria is associated with poor clinical outcome in adults with S. aureus bacteremia.J Hosp Infect.2008;69:345–349.
- ,,, et al.Persistent Staphylococcus aureus bacteremia. An analysis of risk factors and outcomes.Arch Int Med.2007;167(17):1861–1867.
- .Staphylococcus aureus bacteremia in older adults: predictors of 7‐day mortality and infection with a methicillin‐resistant strain.Infect Control Hosp Epidemiol.2006;27(11):1219–1225.
- ,,, et al.Infective endocarditis. Diagnosis, antimicrobial therapy, and management of complications: a statement for healthcare professionals from the Committee on Rheumatic Fever, Endocarditis, and Kawasaki Disease, Council on Cardiovascular Disease in the Young, and the Councils on Clinical Cardiology, Stroke, and Cardiovascular Surgery and Anesthesia, American Heart Association: endorsed by the Infectious Diseases Society of America.Circulation2005;111(23):e394–e434.
- ,,, et al.Antibiotic sensitivity of bacteria associated with community‐acquired urinary tract infection in Britain.J Antimicrob Chemother.1999;44(3):359–365.
- .Antibiotic susceptibility of bacterial strains isolated from patients with community‐acquired urinary tract infections in France.Eur J Clin Microbiol Infect Dis.2000;19(2):112–117.
- ,.Management of methicillin‐resistant Staphylococcus aureus bacteremia.Clin Infect Dis.2008;46(suppl 5):S386–S393.
- ,,, et al.Guidelines for the management of intravascular catheter‐related infections.Clin Infect Dis.2001;32(9):1249–1272.
- ,,.Daptomycin versus standard therapy for bacteremia and endocarditis caused by Staphylococcus aureus.N Engl J Med.2006;355(7):653–665.
Staphylococcus aureus (SA) infection can cause a wide range of clinical syndromes, from folliculitis to life‐threatening endocarditis. Further, SA is second only to S. epidermidis as a cause of bacteremia in hospitalized patients.1, 2 Recent single‐institution studies suggests that SA could be the most frequent cause of nosocomial bacteremia,3, 4 but this needs to be validated in multicenter studies. SA bacteremia (SAB) is often complicated by hematogenous seeding into deep tissues or prosthetic material. The association of future hardware infection following SAB is well documented.5, 6 One study showed that SAB can precede and be associated with prosthetic joint infections in up to 34% of cases.6 Intravascular cardiac devices can also be infected by SAB, with rates from 28% to 75% depending on how early the bacteremia occurred in relation to the implantation of the device.5 Risk stratification for these complications is a clinical challenge. Fowler et al.7 postulated some clinical identifiers of complicated SAB; however, predicting which patients will develop a complication from SAB remains very difficult. Muder et al.8 demonstrated that the presence of SA bacteriuria (SABU) correlates with subsequent SAB, but a possible association of SABU with complicated bacteremia was not examined. A more recent study from Huggan et al.9 has suggested a possible association between SABU and poor clinical outcomes in adults with SAB.
We hypothesized that the presence of SABU would identify those patients at increased risk of complications from SAB. SABU may be a practical, economical, and readily available predictor of complicated SAB. Those patients at higher risk for complications may require a more aggressive diagnostic and therapeutic approach.
Methods
We conducted a retrospective cohort study of SAB patients with and without SA in the urine to investigate the association between SABU and the outcomes of the complications and mortality.
The study was conducted at Miami Valley Hospital (MVH, Dayton, OH), an 848‐bed, level 1 trauma center with 69 intensive care unit (ICU) beds. MVH is a community teaching hospital affiliated with Wright State University Boonshoft School of Medicine and averages 35,000 admissions per year. The same microbiology laboratory (Compunet Clinical Laboratories) processed all the blood and urine culture specimens of the patients in this study.
The inclusion criteria were as follows: 1) admission to MVH between January 1, 2004 and December 31, 2007 with a documented episode of SAB (at least 1 positive blood culture); and 2) a documented urine culture within 7 days of the episode of SAB. Patients without a documented urine culture or with inadequate/emncomplete treatment for SAB were excluded. A total of 118 patients were included based on the presence of a positive blood culture for SA and the presence of a documented urine culture. Patient electronic and paper records were reviewed by 3 of the investigators (E.V.P.‐J., S.D.B., and W.B.B.). Patients subsequently admitted to MVH and to MVH's companion medical center in Dayton, Good Samaritan Hospital, were followed through the electronic medical record common to both institutions.
Study patients were divided into 2 cohorts. One cohort included the patients with a urine culture that grew SA, either methicillin‐resistant SA (MRSA) or methicillin‐susceptible SA (MSSA). The other cohort included patients who had either a negative urine culture or a positive urine culture with organisms other than SA. The age, sex, date of admission, length of stay, and duration of follow‐up were recorded for each patient. Clinical variables included blood culture and urine culture results, presence of intravenous catheters, antibiotic therapy and duration, presence of comorbidities, and clinical outcomes (complications and death).
The primary outcome was complications during hospital admission. The 8 complications investigated were as follows: endocarditis, osteomyelitis, septic arthritis, thrombophlebitis, septic shock, septic embolism/abscess, persistent SAB (lasting more than 5 days after starting adequate SA treatment), and recurrent SAB. In addition, the 2 groups were compared on: 1) any complication, 2) average complications, 3) early complications (ie, within the current hospital admission), and 4) delayed complications (ie, complications diagnosed on subsequent admissions).
Statistical Methods
Means standard deviations (SDs) are reported for continuous variables while frequencies and percents are reported for categorical variables. The independent samples t test for continuous variables and the chi square test or Fisher's exact test for categorical variables were used to compare the two cohorts. Inferences were made at the 0.05 level of significance with no correction for multiple comparisons. SPSS 11.0 software (SPSS, Inc., Chicago, IL) was used for all analyses.
Results
Of the 118 patients, 58 were female (49.2%) and 60 male (50.8%). The age of the patients was 63.3 16.7 years (mean SD). The length of hospital stay was 19.3 17.0 days, and the duration of follow up was 8.3 5.7 months. MRSA was isolated in 75 patients (63.6%) and MSSA in 43 patients (36.4%). In the 28 patients with SA in urine cultures, MRSA was found more frequently than MSSA (20 vs. 8 patients). The acquisition of SAB was equally divided among outpatient (35.6%), healthcare‐associated (30.5%), and hospital‐acquired (33.9%) settings.
Table 1 shows that the group with SABU did not differ from the group without SABU in age (66 years vs. 62 years; P = 0.29), sex (43% male vs. 53% male; P = 0.33), length of hospital stay (18 days vs. 20 days; P = 0.59), and duration of follow‐up (6.6 months vs. 8.8 months; P = 0.064). The 2 cohorts also did not differ on the proportion with MRSA bacteremia (71% vs. 61%; P = 0.32), origin of SAB (P = 0.12), and the presence of comorbidities (diabetes mellitus, cardiomyopathy/congestive heart failure, malignancy, renal disease, and immunosuppression) (all P values > 0.30).
| Characteristic | S. aureus Bacteriuria (n = 28) | No S. aureus Bacteriuria (n = 90) | P Value* |
|---|---|---|---|
| |||
| Age (years) (mean SD) | 66.3 16.3 | 62.4 16.8 | 0.29 |
| Male sex (n [%[) | 12 (42.9) | 48 (53.3) | 0.33 |
| Length of stay (days) (mean SD) | 17.8 16.1 | 19.7 17.3 | 0.59 |
| Follow‐up (months) (mean SD) | 6.6 5.3 | 8.8 5.7 | 0.064 |
| Blood culture (n [%]) | |||
| MRSA | 20 (71.4) | 55 (61.1) | 0.32 |
| MSSA | 8 (28.6) | 35 (38.9) | |
| Origin of the bacteremia [n (%)] | 0.12 | ||
| Community‐acquired | 13 (46.4) | 29 (32.2) | |
| Healthcare‐acquired | 10 (35.7) | 26 (28.9) | |
| Hospital‐acquired | 5 (17.9) | 35 (38.9) | |
| Comorbidities (n [%]) | |||
| DM | 11 (39.3) | 38 (42.2) | 0.78 |
| CHF | 5 (17.9) | 20 (22.2) | 0.62 |
| Cancer | 7 (25.0) | 15 (16.7) | 0.32 |
| ESRD | 4 (14.3) | 12 (13.3) | 1.00 |
| Immunosuppression | 6 (21.4) | 15 (16.7) | 0.58 |
| Patients lost to follow‐up (n [%]) | 5 (17.8) | 8 (8.8) | 0.19 |
Table 2 shows that patients in the SABU group were nearly twice as likely to have a complication as the group without SABU (64% vs. 33%; P = 0.004) and had a higher mean number of complications (0.89 vs. 0.48; P = 0.016). Patients in the SABU group also were more likely to have early complications (64% vs. 23%; P < 0.001) but no more likely to have a delayed complication (14% vs. 12%; P = 0.75). Of the 8 specific complications evaluated, the 2 groups differed only on the presence of septic shock, with the SABU group having 3 times more patients with this complication (21% vs. 7%; P = 0.035). Also, a higher proportion of patients died in the SABU group (32.1% vs. 14.4%; P = 0.036).
| Outcome | S. aureus Bacteriuria (n = 28) | No S. aureus Bacteriuria (n = 90) | P Value* |
|---|---|---|---|
| |||
| Any complication (n [%]) | 18 (64.3) | 30 (33.3) | 0.004 |
| Average complications (mean SD) | 0.89 0.83 | 0.48 0.77 | 0.016 |
| Timing of complication (n [%]) | |||
| Early | 18 (64.3) | 21 (23.3) | <0.001 |
| Delayed | 4 (14.3) | 11 (12.2) | 0.75 |
| By specific complication, n (%) | |||
| Endocarditis | 1 (3.6) | 5 (5.6) | 1.00 |
| Osteomyelitis | 3 (10.7) | 5 (5.6) | 0.39 |
| Septic arthritis | 2 (7.1) | 3 (3.3) | 0.59 |
| Thrombophlebitis | 1 (3.6) | 3 (3.3) | 1.00 |
| Septic shock | 6 (21.4) | 6 (6.7) | 0.035 |
| Septic embolism/abscess | 6 (21.4) | 10 (11.1) | 0.21 |
| Persistent SAB | 3 (10.7) | 3 (3.3) | 0.14 |
| Recurrent SAB | 3 (10.7) | 8 (8.9) | 0.72 |
| Death (n [%]) | 9 (32.1) | 13 (14.4) | 0.036 |
Patients with MRSA (n = 75) and those with MSSA (n = 43) did not differ on any complication, average complications, early or late complications, or 7 of the specific complications (data not shown). Only with thrombophlebitis did the 2 groups differ; the MSSA group had 4 (9.3%) patients with this complication while none in the MRSA group were affected (P = 0.016).
Discussion
In our retrospective analysis, SAB with concomitant SABU was associated with more severe disease, complications, and death. Compared to SAB patients without SA in the urine, those with SAB and SA in the urine had more total complications and more early complications, especially septic shock. Further, the proportion of deaths in the SABU cohort was more than twice as high (32% vs. 14%). Therefore, the presence of SABU in patients with SAB could potentially be a useful predictor of complicated SAB and death.
The relationship between SABU and early complications and death remained after excluding the complication of septic shock/need for vasopressors from the analysis (data not shown). The lack of relationship between SABU and delayed complications might have been due to the adequacy of treatment for SAB. Appropriateness of therapy, a criterion for patient inclusion, may have lessened the likelihood of an insufficient treatment plan causing complications. Those patients with MRSA did not differ from those with MSSA on the mean number of complications or early and delayed complications. A greater proportion of MSSA patients had thrombophlebitis than MRSA patients.
Other investigations have identified predictors of mortality or complications from SAB,7, 912 but SABU was not included as a variable in most of these studies. Fowler et al.7 proposed a prognostic model of complicated SAB using the predictors from their study; community acquisition of organisms, persistent bacteremia, persistent fever over 72 hours, and skin examination suggestive of an acute systemic infection. Muder et al.8 reported a relationship between SABU and subsequent SAB, but they did not examine the association between SABU and the risk of complicated SAB. Huggan et al.9 found that concomitant SABU is associated with ICU admission and increased in‐hospital mortality in patients with SAB.
SAB patients with SABU may be at risk for early complications. Consequently, such patients may warrant more aggressive evaluation and treatment. Further, SABU in patients with SAB may be indicative of an endocarditis‐like condition. SA is rarely isolated from the urinary tract as a uropathogen, although it may colonize indwelling catheters and may cause catheter‐related urinary tract infections.13, 14 Thus, when present in urine, SA could be a marker of deep tissue dissemination with the potential to cause complications. Guidelines for the management of intravascular device‐associated bacteremia have been published by the Infectious Diseases Society of America (IDSA) and other organizations,15, 16 and recent studies have demonstrated the effectiveness of newer agents for the management of SAB.17 Nevertheless, there is still controversy regarding some aspects of the management of SAB (eg, duration of therapy, criteria for echocardiographic evaluation, role of combination therapy). The presence of SABU, the marker evaluated in our study, may be an additional factor to consider when deciding upon duration of therapy and whether to obtain echocardiography or other imaging.
Our study was limited by its retrospective nature. Patient records were not always complete. For example, not all patients had echocardiography to evaluate for endocarditis or venous ultrasound to evaluate for septic thrombophlebitis. Also, the presence (or proper removal) of intravascular or urinary catheters could not be documented reliably in all patients. In addition, the 7‐day cutoff for obtaining urine cultures may have been too lenient, leading to underdiagnosis of bacteriuria. Finally, while 13 patients were lost to follow‐up, the 2 groups (SABU and No SABU) did not differ in the proportion lost.
In conclusion, our study found that SABU may be a useful predictor of complicated SAB and death. SAB patients with SABU may be at risk for more and earlier complications. These patients may need closer monitoring due to the higher risk of septic shock and death. Additional therapeutic and management recommendations might include: 1) longer duration of therapy even if a removable source of the bacteremia is identified; 2) more frequent and better supervised follow‐up; and 3) imaging studies including either computed tomography (CT) scans or ultrasound for thorough evaluation of complications. Prospective studies including randomized controlled trials are required before implementing these suggested diagnostic and therapeutic recommendations.
Acknowledgements
The authors thank and acknowledge Logan McCool and Adam Woiwood for their administrative contributions to the study. E.V.P.‐J., as the principal investigator, had full access to all of the data in the study and takes responsibility for the integrity of the data and the accuracy of the data analysis.
Staphylococcus aureus (SA) infection can cause a wide range of clinical syndromes, from folliculitis to life‐threatening endocarditis. Further, SA is second only to S. epidermidis as a cause of bacteremia in hospitalized patients.1, 2 Recent single‐institution studies suggests that SA could be the most frequent cause of nosocomial bacteremia,3, 4 but this needs to be validated in multicenter studies. SA bacteremia (SAB) is often complicated by hematogenous seeding into deep tissues or prosthetic material. The association of future hardware infection following SAB is well documented.5, 6 One study showed that SAB can precede and be associated with prosthetic joint infections in up to 34% of cases.6 Intravascular cardiac devices can also be infected by SAB, with rates from 28% to 75% depending on how early the bacteremia occurred in relation to the implantation of the device.5 Risk stratification for these complications is a clinical challenge. Fowler et al.7 postulated some clinical identifiers of complicated SAB; however, predicting which patients will develop a complication from SAB remains very difficult. Muder et al.8 demonstrated that the presence of SA bacteriuria (SABU) correlates with subsequent SAB, but a possible association of SABU with complicated bacteremia was not examined. A more recent study from Huggan et al.9 has suggested a possible association between SABU and poor clinical outcomes in adults with SAB.
We hypothesized that the presence of SABU would identify those patients at increased risk of complications from SAB. SABU may be a practical, economical, and readily available predictor of complicated SAB. Those patients at higher risk for complications may require a more aggressive diagnostic and therapeutic approach.
Methods
We conducted a retrospective cohort study of SAB patients with and without SA in the urine to investigate the association between SABU and the outcomes of the complications and mortality.
The study was conducted at Miami Valley Hospital (MVH, Dayton, OH), an 848‐bed, level 1 trauma center with 69 intensive care unit (ICU) beds. MVH is a community teaching hospital affiliated with Wright State University Boonshoft School of Medicine and averages 35,000 admissions per year. The same microbiology laboratory (Compunet Clinical Laboratories) processed all the blood and urine culture specimens of the patients in this study.
The inclusion criteria were as follows: 1) admission to MVH between January 1, 2004 and December 31, 2007 with a documented episode of SAB (at least 1 positive blood culture); and 2) a documented urine culture within 7 days of the episode of SAB. Patients without a documented urine culture or with inadequate/emncomplete treatment for SAB were excluded. A total of 118 patients were included based on the presence of a positive blood culture for SA and the presence of a documented urine culture. Patient electronic and paper records were reviewed by 3 of the investigators (E.V.P.‐J., S.D.B., and W.B.B.). Patients subsequently admitted to MVH and to MVH's companion medical center in Dayton, Good Samaritan Hospital, were followed through the electronic medical record common to both institutions.
Study patients were divided into 2 cohorts. One cohort included the patients with a urine culture that grew SA, either methicillin‐resistant SA (MRSA) or methicillin‐susceptible SA (MSSA). The other cohort included patients who had either a negative urine culture or a positive urine culture with organisms other than SA. The age, sex, date of admission, length of stay, and duration of follow‐up were recorded for each patient. Clinical variables included blood culture and urine culture results, presence of intravenous catheters, antibiotic therapy and duration, presence of comorbidities, and clinical outcomes (complications and death).
The primary outcome was complications during hospital admission. The 8 complications investigated were as follows: endocarditis, osteomyelitis, septic arthritis, thrombophlebitis, septic shock, septic embolism/abscess, persistent SAB (lasting more than 5 days after starting adequate SA treatment), and recurrent SAB. In addition, the 2 groups were compared on: 1) any complication, 2) average complications, 3) early complications (ie, within the current hospital admission), and 4) delayed complications (ie, complications diagnosed on subsequent admissions).
Statistical Methods
Means standard deviations (SDs) are reported for continuous variables while frequencies and percents are reported for categorical variables. The independent samples t test for continuous variables and the chi square test or Fisher's exact test for categorical variables were used to compare the two cohorts. Inferences were made at the 0.05 level of significance with no correction for multiple comparisons. SPSS 11.0 software (SPSS, Inc., Chicago, IL) was used for all analyses.
Results
Of the 118 patients, 58 were female (49.2%) and 60 male (50.8%). The age of the patients was 63.3 16.7 years (mean SD). The length of hospital stay was 19.3 17.0 days, and the duration of follow up was 8.3 5.7 months. MRSA was isolated in 75 patients (63.6%) and MSSA in 43 patients (36.4%). In the 28 patients with SA in urine cultures, MRSA was found more frequently than MSSA (20 vs. 8 patients). The acquisition of SAB was equally divided among outpatient (35.6%), healthcare‐associated (30.5%), and hospital‐acquired (33.9%) settings.
Table 1 shows that the group with SABU did not differ from the group without SABU in age (66 years vs. 62 years; P = 0.29), sex (43% male vs. 53% male; P = 0.33), length of hospital stay (18 days vs. 20 days; P = 0.59), and duration of follow‐up (6.6 months vs. 8.8 months; P = 0.064). The 2 cohorts also did not differ on the proportion with MRSA bacteremia (71% vs. 61%; P = 0.32), origin of SAB (P = 0.12), and the presence of comorbidities (diabetes mellitus, cardiomyopathy/congestive heart failure, malignancy, renal disease, and immunosuppression) (all P values > 0.30).
| Characteristic | S. aureus Bacteriuria (n = 28) | No S. aureus Bacteriuria (n = 90) | P Value* |
|---|---|---|---|
| |||
| Age (years) (mean SD) | 66.3 16.3 | 62.4 16.8 | 0.29 |
| Male sex (n [%[) | 12 (42.9) | 48 (53.3) | 0.33 |
| Length of stay (days) (mean SD) | 17.8 16.1 | 19.7 17.3 | 0.59 |
| Follow‐up (months) (mean SD) | 6.6 5.3 | 8.8 5.7 | 0.064 |
| Blood culture (n [%]) | |||
| MRSA | 20 (71.4) | 55 (61.1) | 0.32 |
| MSSA | 8 (28.6) | 35 (38.9) | |
| Origin of the bacteremia [n (%)] | 0.12 | ||
| Community‐acquired | 13 (46.4) | 29 (32.2) | |
| Healthcare‐acquired | 10 (35.7) | 26 (28.9) | |
| Hospital‐acquired | 5 (17.9) | 35 (38.9) | |
| Comorbidities (n [%]) | |||
| DM | 11 (39.3) | 38 (42.2) | 0.78 |
| CHF | 5 (17.9) | 20 (22.2) | 0.62 |
| Cancer | 7 (25.0) | 15 (16.7) | 0.32 |
| ESRD | 4 (14.3) | 12 (13.3) | 1.00 |
| Immunosuppression | 6 (21.4) | 15 (16.7) | 0.58 |
| Patients lost to follow‐up (n [%]) | 5 (17.8) | 8 (8.8) | 0.19 |
Table 2 shows that patients in the SABU group were nearly twice as likely to have a complication as the group without SABU (64% vs. 33%; P = 0.004) and had a higher mean number of complications (0.89 vs. 0.48; P = 0.016). Patients in the SABU group also were more likely to have early complications (64% vs. 23%; P < 0.001) but no more likely to have a delayed complication (14% vs. 12%; P = 0.75). Of the 8 specific complications evaluated, the 2 groups differed only on the presence of septic shock, with the SABU group having 3 times more patients with this complication (21% vs. 7%; P = 0.035). Also, a higher proportion of patients died in the SABU group (32.1% vs. 14.4%; P = 0.036).
| Outcome | S. aureus Bacteriuria (n = 28) | No S. aureus Bacteriuria (n = 90) | P Value* |
|---|---|---|---|
| |||
| Any complication (n [%]) | 18 (64.3) | 30 (33.3) | 0.004 |
| Average complications (mean SD) | 0.89 0.83 | 0.48 0.77 | 0.016 |
| Timing of complication (n [%]) | |||
| Early | 18 (64.3) | 21 (23.3) | <0.001 |
| Delayed | 4 (14.3) | 11 (12.2) | 0.75 |
| By specific complication, n (%) | |||
| Endocarditis | 1 (3.6) | 5 (5.6) | 1.00 |
| Osteomyelitis | 3 (10.7) | 5 (5.6) | 0.39 |
| Septic arthritis | 2 (7.1) | 3 (3.3) | 0.59 |
| Thrombophlebitis | 1 (3.6) | 3 (3.3) | 1.00 |
| Septic shock | 6 (21.4) | 6 (6.7) | 0.035 |
| Septic embolism/abscess | 6 (21.4) | 10 (11.1) | 0.21 |
| Persistent SAB | 3 (10.7) | 3 (3.3) | 0.14 |
| Recurrent SAB | 3 (10.7) | 8 (8.9) | 0.72 |
| Death (n [%]) | 9 (32.1) | 13 (14.4) | 0.036 |
Patients with MRSA (n = 75) and those with MSSA (n = 43) did not differ on any complication, average complications, early or late complications, or 7 of the specific complications (data not shown). Only with thrombophlebitis did the 2 groups differ; the MSSA group had 4 (9.3%) patients with this complication while none in the MRSA group were affected (P = 0.016).
Discussion
In our retrospective analysis, SAB with concomitant SABU was associated with more severe disease, complications, and death. Compared to SAB patients without SA in the urine, those with SAB and SA in the urine had more total complications and more early complications, especially septic shock. Further, the proportion of deaths in the SABU cohort was more than twice as high (32% vs. 14%). Therefore, the presence of SABU in patients with SAB could potentially be a useful predictor of complicated SAB and death.
The relationship between SABU and early complications and death remained after excluding the complication of septic shock/need for vasopressors from the analysis (data not shown). The lack of relationship between SABU and delayed complications might have been due to the adequacy of treatment for SAB. Appropriateness of therapy, a criterion for patient inclusion, may have lessened the likelihood of an insufficient treatment plan causing complications. Those patients with MRSA did not differ from those with MSSA on the mean number of complications or early and delayed complications. A greater proportion of MSSA patients had thrombophlebitis than MRSA patients.
Other investigations have identified predictors of mortality or complications from SAB,7, 912 but SABU was not included as a variable in most of these studies. Fowler et al.7 proposed a prognostic model of complicated SAB using the predictors from their study; community acquisition of organisms, persistent bacteremia, persistent fever over 72 hours, and skin examination suggestive of an acute systemic infection. Muder et al.8 reported a relationship between SABU and subsequent SAB, but they did not examine the association between SABU and the risk of complicated SAB. Huggan et al.9 found that concomitant SABU is associated with ICU admission and increased in‐hospital mortality in patients with SAB.
SAB patients with SABU may be at risk for early complications. Consequently, such patients may warrant more aggressive evaluation and treatment. Further, SABU in patients with SAB may be indicative of an endocarditis‐like condition. SA is rarely isolated from the urinary tract as a uropathogen, although it may colonize indwelling catheters and may cause catheter‐related urinary tract infections.13, 14 Thus, when present in urine, SA could be a marker of deep tissue dissemination with the potential to cause complications. Guidelines for the management of intravascular device‐associated bacteremia have been published by the Infectious Diseases Society of America (IDSA) and other organizations,15, 16 and recent studies have demonstrated the effectiveness of newer agents for the management of SAB.17 Nevertheless, there is still controversy regarding some aspects of the management of SAB (eg, duration of therapy, criteria for echocardiographic evaluation, role of combination therapy). The presence of SABU, the marker evaluated in our study, may be an additional factor to consider when deciding upon duration of therapy and whether to obtain echocardiography or other imaging.
Our study was limited by its retrospective nature. Patient records were not always complete. For example, not all patients had echocardiography to evaluate for endocarditis or venous ultrasound to evaluate for septic thrombophlebitis. Also, the presence (or proper removal) of intravascular or urinary catheters could not be documented reliably in all patients. In addition, the 7‐day cutoff for obtaining urine cultures may have been too lenient, leading to underdiagnosis of bacteriuria. Finally, while 13 patients were lost to follow‐up, the 2 groups (SABU and No SABU) did not differ in the proportion lost.
In conclusion, our study found that SABU may be a useful predictor of complicated SAB and death. SAB patients with SABU may be at risk for more and earlier complications. These patients may need closer monitoring due to the higher risk of septic shock and death. Additional therapeutic and management recommendations might include: 1) longer duration of therapy even if a removable source of the bacteremia is identified; 2) more frequent and better supervised follow‐up; and 3) imaging studies including either computed tomography (CT) scans or ultrasound for thorough evaluation of complications. Prospective studies including randomized controlled trials are required before implementing these suggested diagnostic and therapeutic recommendations.
Acknowledgements
The authors thank and acknowledge Logan McCool and Adam Woiwood for their administrative contributions to the study. E.V.P.‐J., as the principal investigator, had full access to all of the data in the study and takes responsibility for the integrity of the data and the accuracy of the data analysis.
- ,,, et al.Prevalence and drug susceptibility of pathogens causing bloodstream infections in northern Italy: a two‐year study in 16 hospitals.Eur J Clin Microbiol Infect Dis.2002;21(12):849–855.
- ,,, et al.Nosocomial bloodstream infections in ICU and non‐ICU patients.Am J Infec Control.2005;33(6):333–340.
- ,,, et al.Age‐ and sex‐associated trends in bloodstream infection: a population‐based study in Olmsted County, Minnesota.Arch Intern Med.2007;167(8):834–839.
- ,,.Bloodstream infections in a geriatric cohort: a population‐based study.Am J Med.2007;120(12):1078–1883.
- ,,, et al.Staphylococcus aureus bacteremia in patients with permanent pacemakers or implantable cardioverter‐defibrillators.Circulation.2001;104(9):1029–1033.
- ,,, et al.Infection of orthopedic prostheses after Staphylococcus aureus bacteremia.Clin Infect Dis.2001;32(4):647–649.
- ,,, et al.Clinical identifiers of complicated Staphylococcus aureus bacteremia.Arch Intern Med.2003;163(17):2066–2072.
- ,,, et al.Isolation of Staphylococcus aureus from the urinary tract: association of isolation with symptomatic UTI and subsequent staphylococcal bacteremia.Clin Infect Dis.2006;42(1):46–50.
- ,,, et al.Concomitant Staphylococcus aureus bacteriuria is associated with poor clinical outcome in adults with S. aureus bacteremia.J Hosp Infect.2008;69:345–349.
- ,,, et al.Persistent Staphylococcus aureus bacteremia. An analysis of risk factors and outcomes.Arch Int Med.2007;167(17):1861–1867.
- .Staphylococcus aureus bacteremia in older adults: predictors of 7‐day mortality and infection with a methicillin‐resistant strain.Infect Control Hosp Epidemiol.2006;27(11):1219–1225.
- ,,, et al.Infective endocarditis. Diagnosis, antimicrobial therapy, and management of complications: a statement for healthcare professionals from the Committee on Rheumatic Fever, Endocarditis, and Kawasaki Disease, Council on Cardiovascular Disease in the Young, and the Councils on Clinical Cardiology, Stroke, and Cardiovascular Surgery and Anesthesia, American Heart Association: endorsed by the Infectious Diseases Society of America.Circulation2005;111(23):e394–e434.
- ,,, et al.Antibiotic sensitivity of bacteria associated with community‐acquired urinary tract infection in Britain.J Antimicrob Chemother.1999;44(3):359–365.
- .Antibiotic susceptibility of bacterial strains isolated from patients with community‐acquired urinary tract infections in France.Eur J Clin Microbiol Infect Dis.2000;19(2):112–117.
- ,.Management of methicillin‐resistant Staphylococcus aureus bacteremia.Clin Infect Dis.2008;46(suppl 5):S386–S393.
- ,,, et al.Guidelines for the management of intravascular catheter‐related infections.Clin Infect Dis.2001;32(9):1249–1272.
- ,,.Daptomycin versus standard therapy for bacteremia and endocarditis caused by Staphylococcus aureus.N Engl J Med.2006;355(7):653–665.
- ,,, et al.Prevalence and drug susceptibility of pathogens causing bloodstream infections in northern Italy: a two‐year study in 16 hospitals.Eur J Clin Microbiol Infect Dis.2002;21(12):849–855.
- ,,, et al.Nosocomial bloodstream infections in ICU and non‐ICU patients.Am J Infec Control.2005;33(6):333–340.
- ,,, et al.Age‐ and sex‐associated trends in bloodstream infection: a population‐based study in Olmsted County, Minnesota.Arch Intern Med.2007;167(8):834–839.
- ,,.Bloodstream infections in a geriatric cohort: a population‐based study.Am J Med.2007;120(12):1078–1883.
- ,,, et al.Staphylococcus aureus bacteremia in patients with permanent pacemakers or implantable cardioverter‐defibrillators.Circulation.2001;104(9):1029–1033.
- ,,, et al.Infection of orthopedic prostheses after Staphylococcus aureus bacteremia.Clin Infect Dis.2001;32(4):647–649.
- ,,, et al.Clinical identifiers of complicated Staphylococcus aureus bacteremia.Arch Intern Med.2003;163(17):2066–2072.
- ,,, et al.Isolation of Staphylococcus aureus from the urinary tract: association of isolation with symptomatic UTI and subsequent staphylococcal bacteremia.Clin Infect Dis.2006;42(1):46–50.
- ,,, et al.Concomitant Staphylococcus aureus bacteriuria is associated with poor clinical outcome in adults with S. aureus bacteremia.J Hosp Infect.2008;69:345–349.
- ,,, et al.Persistent Staphylococcus aureus bacteremia. An analysis of risk factors and outcomes.Arch Int Med.2007;167(17):1861–1867.
- .Staphylococcus aureus bacteremia in older adults: predictors of 7‐day mortality and infection with a methicillin‐resistant strain.Infect Control Hosp Epidemiol.2006;27(11):1219–1225.
- ,,, et al.Infective endocarditis. Diagnosis, antimicrobial therapy, and management of complications: a statement for healthcare professionals from the Committee on Rheumatic Fever, Endocarditis, and Kawasaki Disease, Council on Cardiovascular Disease in the Young, and the Councils on Clinical Cardiology, Stroke, and Cardiovascular Surgery and Anesthesia, American Heart Association: endorsed by the Infectious Diseases Society of America.Circulation2005;111(23):e394–e434.
- ,,, et al.Antibiotic sensitivity of bacteria associated with community‐acquired urinary tract infection in Britain.J Antimicrob Chemother.1999;44(3):359–365.
- .Antibiotic susceptibility of bacterial strains isolated from patients with community‐acquired urinary tract infections in France.Eur J Clin Microbiol Infect Dis.2000;19(2):112–117.
- ,.Management of methicillin‐resistant Staphylococcus aureus bacteremia.Clin Infect Dis.2008;46(suppl 5):S386–S393.
- ,,, et al.Guidelines for the management of intravascular catheter‐related infections.Clin Infect Dis.2001;32(9):1249–1272.
- ,,.Daptomycin versus standard therapy for bacteremia and endocarditis caused by Staphylococcus aureus.N Engl J Med.2006;355(7):653–665.
Copyright © 2010 Society of Hospital Medicine