User login
Identifying an Idle Line for Its Removal
Infections acquired in the hospital are termed healthcare‐associated infections (HAIs) and include central lineassociated blood stream infections (CLABSIs). Among HAIs, CLABSIs cause the highest number of preventable deaths.[1] Central venous catheters (CVCs) or central lines are commonly used in the hospital.[2] Each year their use is linked to 250,000 cases of CLABSIs in the United States.[3] Some CLABSIs may be prevented by the prompt removal of the line.[4] However, CVCs are often retained after their clinical indication has lapsed and are then referred to as idle lines.[5, 6] In this work, we propose and theoretically test a guideline to facilitate the safe removal of an idle line by observing the agreement and disagreement between actual practice and the proposed guideline.
METHODS
Setting
This work was conducted at a large, urban, tertiary care, academic health center in the United States as a collaborative effort to improve quality at our institution.[7]
Design and Patients
The reports linked with the electronic medical records at our institution include a daily, ward‐by‐ward listing of patients who have access other than a peripheral line in place. This central line dashboard accesses the information on intravenous access charted by bedside nurses to create a list of patients on every ward who have any kind of central access. Temporary central venous lines (CVLs), peripherally inserted central catheters (PICCs), ports, and dialysis catheters are all included. The unit charge nurses and managers use this dashboard to facilitate compliance with line care bundles. We used this source to identify patients with either type of CVC (CVLs or PICCs) on 8 days in August 2014, September 2014, and October 2014. Patients were included if they had a CVC and were on a general medical or surgical ward bed on audit day. CVLs at all sites were included (femoral, subclavian, and internal jugular). Patients in an intensive care unit (ICU) or progressive care unit on the day of the audit were excluded. Patients whose catheters were for chemotherapy and those admitted for a transplant or receiving palliative or hospice care were also excluded.
Data Collection
A protocol for data collection was written out, and a training session was held to review definitions, data sources, and methods to ensure consistency. Two authors (M.M. and J.D.) assisted by an experienced clinical nurse specialist collected data on the patients captured on audit days. Each chart was reviewed on the day of the audit, the 2 days preceding the audit day, and then followed until the patient was either discharged from the hospital or transferred to a higher level of care, died, or transitioned to palliative or hospice care. Demographics, details about the line, and the criteria for justified use were extracted from the electronic medical record.
Definitions
Justified and Idle Days
To justify the presence of a CVC on any given day, we used criteria that fell under 3 categories: intravenous (IV) access needs, unstable vitals, or meeting sepsis/systemic inflammatory response syndrome (SIRS) criteria (Table 1). For vital signs, a single abnormal reading was counted as fulfilling criteria for that day. If no criterion for justified use was met, the line was considered idle for that day.
|
| IV access needs |
| Expected duration of IV antibiotics >6 days |
| Administration of TPN |
| Anticipated requirement of home IV medications |
| Requirement of IV medications with documented difficult access |
| Hemorrhage requiring blood transfusions |
| Requiring more than 3 infusions |
| Requiring more than 2 infusions and blood transfusions |
| Abnormal vitals |
| Diastolic blood pressure >120 mm Hg |
| Systolic blood pressure 90 mm Hg |
| Systolic blood pressure >200 mm Hg |
| Heart rate >120 beats per minute |
| Heart rate 50 beats per minute |
| Respiratory rate >30 breaths per minute |
| Respiratory rate 10 breaths per minute |
| Oxygen saturation 90% as measured by pulse oximetry |
| Meeting SIRS criteria (2 or more of the following present) |
| Temp >38C, Temp 36C, heart rate >90 beats per minute, respiratory rate >20 breaths per minute, WBC >12,000/mm3, WBC 1,000/mm3, bandemia >10% |
Qualifying IV access needs were defined similarly to those previously used,[5, 6] whereas those for SIRS followed the current consensus.[8] To determine the number of IV medications or infusions, the medication administration record was reviewed. If 3 or more infusions were found, their compatibility was checked using the same database that nurses use at our institution. Difficult IV access was inferred from the indication for line placement, coupled with the absence of documentation of a peripheral IV. Clinical progress notes were reviewed to extract information on the length of proposed IV antibiotic courses, and discharge instructions were reviewed to verify whether the line was removed prior to discharge or not. The cutoffs for diastolic blood pressure, respiratory rate, and oxygen saturation used to label patients hemodynamically labile are the same as those used by previous authors and also constitute the definition of hypertensive urgency.[5, 9] However, we diverged from the values previously used for tachycardia, bradycardia, and systolic hypotension using heart rates >120 and 50 beats per minute (compared to >130 and 40 beats per minute) and systolics 90 mm Hg (compared to 80 mm Hg) to justify the line.[5] Early warning scores have been used to identify hospitalized ward patients who are at risk for clinical deterioration. Although each score utilizes different thresholds, the risk for clinical deterioration increases as the vitals worsen.[10] Bearing this in mind, the thresholds we elected to use are more clinically conservative and also parallel the nursing call orders currently used at our institution.
Proposed Guideline
We propose the guideline that a CVC may be safely removed the day after the first idle day.
RESULTS
A total of 126 lines were observed in 126 patients. Eighty‐three (65.9%) of the lines were PICCs. The remaining 43 (34.1%) were CVLs. The indications for line placement were distributed between the need for central access, total parenteral nutrition, or antibiotics (Table 2).
| Description | Value |
|---|---|
| |
| Age in yrs mean (SD) | 55.7 (18) |
| Gender, n (%) | |
| Female | 66 (52.4) |
| Male | 60 (47.6) |
| Type of line, n (%) | |
| PICC | 83 (65.9) |
| CVL | 43 (34.1) |
| Indication for line placement, n (%) | |
| Meds requiring central access or TPN | 36 (28.6) |
| Antibiotics | 34 (27.0) |
| Hemodynamic instability | 30 (23.8) |
| Poor access with multiple IV medications | 18 (14.3) |
| Unknown | 8 (6.3) |
| Line removed prior to discharge, n (%) | |
| Yes | 76 (60.3) |
| No | 50 (39.7) |
Out of the 126 patients, 50 (39.7%) were discharged from the hospital, died, were transferred to a higher level of care, or transitioned to palliative or hospice care with the line in place. In the remaining 76 patients, the audit captured 635 days, out of which a line was in place for 522 (82.2%) days. Of these 522 days, the line's presence was justified by our criteria for 351 (67.2%) days. The most common reason for a line to be justified on any given day was the need for antibiotics followed by the presence of SIRS criteria (Table 3). The remaining 171 (32.7%) days were idle.
| Criteria | N | % |
|---|---|---|
| ||
| No. of factors justifying use | ||
| 1 | 184 | 52.4% |
| 2 | 127 | 36.2% |
| >2 | 40 | 11.4 |
| Reason for justifying line* | ||
| Anticipate home or >6 days of antibiotic use | 181 | 51.6 |
| SIRS criteria | 124 | 35.3 |
| TPN | 96 | 27.4 |
| Hemodynamic instability based on hr and bp | 78 | 22.2 |
| Poor access with need for IV medications | 57 | 16.2 |
| Respiratory rate (10 or >30/minute) | 25 | 7.1 |
| Active hemorrhage requiring transfusions | 12 | 3.4 |
| >3 infusions | 6 | 1.7 |
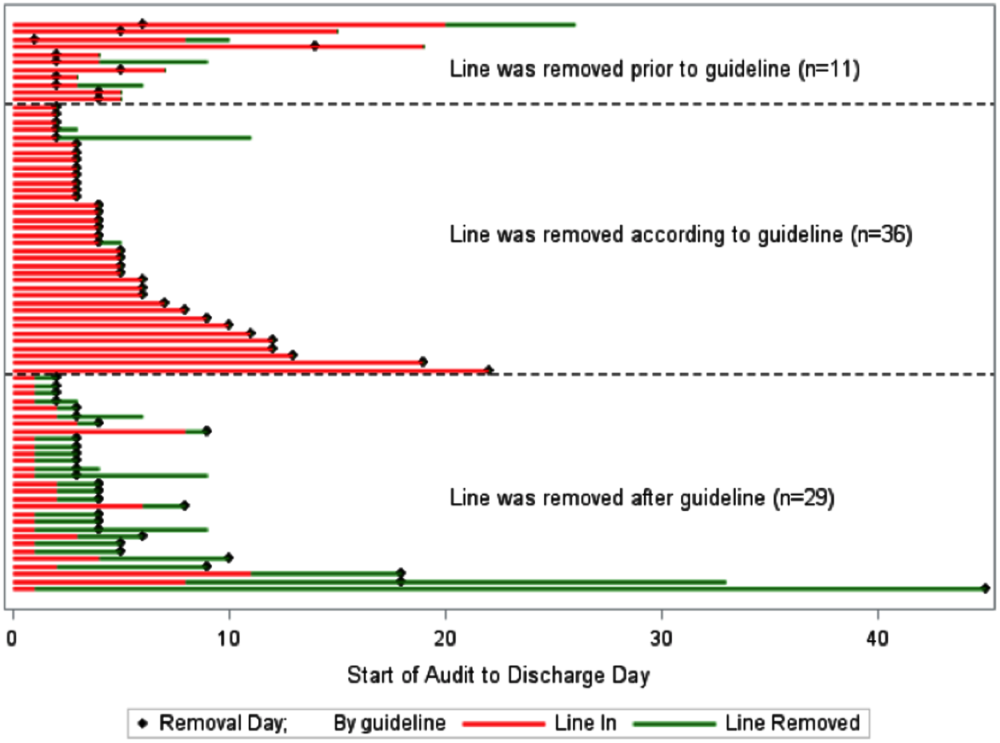
A comparison of the actual removal of the 76 central lines in practice relative to the proposed guideline of removing it the day following the first idle day is displayed in Figure 1. The central line was removed prior to our proposed guideline in 11 (14.5%) patients, and waiting for an idle day in these patients would have added 46 line days. In almost half the patients (n = 36, 47.4%), the line was removed in agreement with the proposed guideline. None of the patients in whom the line was removed prior to or in accordance with our proposed guideline required a line reinsertion. Line removal was delayed in 29 (38.2%) patients when compared to our proposed guideline. In these patients, following the guideline would have created 122 line‐free days. Most (n = 102, 83.6%) of these potential line‐free days were idle. Twenty (16.4%) were justified, of which half (n = 10) were justified by meeting SIRS criteria.

DISCUSSION
Approximately 1 in every 25 inpatients in the United States has at least 1 HAI on any given day.[11] The case fatality rate from a CLABSI may be as high as 12%, and up to 70% of these infections may be preventable.[1, 12] Interventions successful in decreasing CLABSIs have focused on patients in ICUs.[13] However, CVCs are increasingly prevalent outside the ICU, with over 4.5 million line days in non‐ICU beds reported to the National Healthcare Safety Network in 2012 compared to 2.5 million in 2010.[2, 14] However, adherence rates to infection control practices may be lower on the wards than in the ICUs.[6, 15] Consequently, although the number of CLABSIs has declined over the last decade, most are now occurring outside the ICU.[16] These trends underscore the need to develop strategies aimed at CLABSI prevention on the floors.
Analogous to the life cycle of a urinary catheter described by Meddings et al.,[17] strategies to prevent CLABSIs and other CVC‐related complications may be designed around the life cycle of a CVC. The life cycle starts with insertion and moves on to the maintenance, removal, and possible reinsertion of the line. The process thus starts with the decision to place the line. Over the last decade, this decision making has changed in part due to PICCs. This shift is reflected in PICC prevalence rates: in 2001, 11% of audited central lines were PICCs compared to 56% in 2007.[5, 6] In our audit, 66% of the CVCs were PICCs. This increase in the use of PICCs may be attributable to the ease and safety of their placement coupled with the increased availability of vascular access placement teams.[18] The risk of overuse that may result from such expediency may be countered by adhering to guidelines such as the Michigan Appropriateness Guide for Intravenous Catheters, which provides both clinically detailed guidance and an impetus for reflective decision making around intravenous access.[19]
The placement of CVCs for prolonged parenteral antibiotics may be a particular subset that bears further exploration. Similar to previous reports, we found that a large number of the CVCs were both inserted for and justified by the need for IV antibiotics.[5] Guidelines delineated by the Infectious Diseases Society of America regarding outpatient parenteral antibiotics weigh both the duration of therapy and the antimicrobial's potential for causing phlebitis when recommending the type of intravascular access.[20] Many courses may therefore be completed through peripheral or midline catheters. Developing strong partnerships between infectious disease specialists, hospitalists, and the facilities or home‐care services treating these patients may curtail the use of CVCs for antimicrobial administration.
The main focus of our work is on facilitating the safe removal of CVCs. The risk of CLABSIs increases each day a CVC is in place, and guidelines to prevent CLABSIs include recommendations to promptly remove nonessential catheters.[4, 21] There is also an emerging understanding that the risk of a PICC‐related CLABSI approaches that from a traditional central line in hospitalized patients, and PICCs confer an increased risk of venous thromboembolism.[18, 22] Although nearly half of surveyed hospitalists recently reported leaving PICCs in place until discharge day, our data suggest that this practice may be driven by the trajectory of a patient's recovery as much as by knowledge gaps related to the use of PICCs.[23] In nearly half the instances, clinical practice already mirrors our proposed guideline, with line removal coinciding with both the timing proposed by our guideline and discharge day. However, there is room for improvement, as line removal may have been expedited in the 29 patients in whom the line was retained after the first idle day. Maintaining an awareness of its presence and weighing its risks and benefits daily may facilitate the removal of a CVC. Based on the recent findings that up to a quarter of clinicians are unaware that their patients have a central line, the mere reminder of the presence of a line using such criteria may expedite its removal by triggering a purposeful reassessment of its ongoing need.[24] Premature CVC removal requiring line reinsertion is an unintended consequence that may emerge from the earlier removal of lines. In our sample, none of the patients who had lines removed either prior to or in accordance with our proposed guideline required a line reinsertion. In addition to line reinsertion, delays in laboratory testing and reporting due to the unavailability of access, increased patient discomfort, or increased workload on the bedside nurse or vascular access team must also be considered when implementing strategies aimed at decreasing line days.
We envisage using these criteria to both empower practitioners with knowledge and foster shared accountability between all team members by using a uniform tool. This can occur through partnerships between infection control, clinical nurse specialists, bedside nursing, and physicians. The electronic medical record could be leveraged to scan the record for the criteria and create a notification when the line becomes idle. In alignment with the Michigan Appropriateness Guide for Intravenous Catheters guidelines, we do not support the removal of lines by nursing staff without physician notification.[19] Such principles have been successfully harnessed in strategies to prevent both catheter‐associated urinary tract infections and CLABSIs in ICUs.[13, 25] In light of the complexity surrounding the decision making for CVCs, our criteria were focused on the wards and erred on the side of clinical caution. This clinical conservatism is apparent in the patients in whom lines were removed prior to what our guideline would propose, yet none of the patients required a line reinsertion. As concerns about recrudescent clinical instability may drive decision making around line removal, such conservatism may be warranted initially. However, the fidelity of these criteria in the clinical setting will need prospective validation. In particular, the inclusion of SIRS criteria may have led to an overestimation of justified days. Further studies may be needed to refine the criteria and find a clinical hierarchy that balances the risks and benefits of retaining a central line.
Our work has certain limitations. It is a single center's experience, and our findings may not therefore be generalizable. Except for when the indication for the line was for difficult access, we did not attempt to verify the presence of a peripheral IV. This, in combination with the inclusion of SIRS criteria, likely leads to an underestimation of idle days. In the interest of focusing on patients in whom the decision making around a line would be the least controversial, we did not continue to follow patients who were transferred to a higher level of care. It is possible, however, that these transfers were precipitated by line‐associated complications such as sepsis and would be important to track. We did not measure the agreement between data collectors, although definitions and methodologies were standardized and reviewed prior to data collection. As this was an observational assessment of a proposed guideline, we cannot predict how the recommendations generated by it will be received by clinicians. Although this may prove to be a barrier in adoption, we hope that the conversation it initiates leads to change.
Hospitalists are positioned to potentially influence the entire life cycle of a central line on the floor. Strategies can be enacted at each stage to help decrease the potential of harm from these devices to our patients. Creating and testing criteria and guidelines such as we propose represents just 1 such strategy in a multidisciplinary effort to provide the best possible care we can.
Acknowledgements
The authors thank Jennifer Dunscomb, Kristen Kelly, and their teams, and Deanna Sidwell, Todd Biggerstaff, Joan Miller, Rob Clark, and the tireless providers at Indiana University Health Methodist Hospital for their support.
Disclosures: This work was supported by the Indiana University Health Values Grant for research. The authors have no conflicts of interests to report.
- , , , , , . Estimating the proportion of healthcare‐associated infections that are reasonably preventable and the related mortality and costs. Infect Control Hosp Epidemiol. 2011;32(2):101–114.
- , , , et al. National Healthcare Safety Network (NHSN) report, data summary for 2012, device‐associated module. Am J Infect Control. 2013;41(12):1148–1166.
- , , . The risk of bloodstream infection in adults with different intravascular devices: a systematic review of 200 published prospective studies. Mayo Clin Proc. 2006;81(9):1159–1171.
- , , , et al. Guidelines for the prevention of intravascular catheter‐related infections. Clin Infect Dis. 2011;52(9):e162–e193.
- , , , et al. Temporary central venous catheter utilization patterns in a large tertiary care center: tracking the “idle central venous catheter.” Infect Control Hosp Epidemiol. 2012;33(1):50–57.
- , , , , , . Unnecessary use of central venous catheters: the need to look outside the intensive care unit. Infect Control Hosp Epidemiol. 2004;25(3):266–268.
- IU Health Methodist Hospital website. Available at: http://iuhealth.org/methodist/aboIut. Accessed October 20, 2014.
- , , , et al. Definitions for Sepsis and Organ Failure and Guidelines for the Use of Innovative Therapies in Sepsis. The ACCP/SCCM Consensus Conference Committee. American College of Chest Physicians/Society of Critical Care Medicine. Chest. 2009;136(5 suppl):e28.
- , , , , , . Acute hypertension: a systematic review and appraisal of guidelines. Ochsner J. 2014;14(4):655–663.
- , , . Predicting clinical deterioration in the hospital: the impact of outcome selection. Resuscitation. 2013;84(5):564–568.
- , , , et al. Multistate point‐prevalence survey of health care–associated infections. N Engl J Med. 2014;370(13):1198–1208.
- , , , et al. Estimating health care‐associated infections and deaths in U.S. hospitals, 2002. Public Health Rep. 2007;122(2):160–166.
- , , , et al. An intervention to decrease catheter‐related bloodstream infections in the ICU. N Engl J Med. 2006;355(26):2725–2732.
- , , , et al. Data summary for 2011, device‐associated module. Centers for Disease Control and Prevention. National Healthcare Safety Network (NHSN) Report. Available at: http://www.cdc.gov/nhsn/PDFs/dataStat/NHSN‐Report‐2011‐Data‐Summary.pdf. Published April 1, 2013. Last accessed January 2015.
- , , . Idle central venous catheter‐days pose infection risk for patients after discharge from intensive care. Am J Infect Control. 2014;42(4):453–455.
- , . Update on emerging infections: news from the Centers for Disease Control and Prevention. Vital signs: central line‐associated blood stream infections—United States, 2001, 2008, and 2009. Ann Emerg Med. 2011;58(5):447–451.
- , , , , , . Reducing unnecessary urinary catheter use and other strategies to prevent catheter‐associated urinary tract infection: an integrative review. BMJ Qual Saf. 2014;23(4):277–289.
- , , , , . The risk of bloodstream infection associated with peripherally inserted central catheters compared with central venous catheters in adults: a systematic review and meta‐analysis. Infect Control Hosp Epidemiol. 2013;34(9):908–918.
- , , , et al. The Michigan Appropriateness Guide for Intravenous Catheters (MAGIC): results from a multispecialty panel using the RAND/UCLA Appropriateness Method. Ann Intern Med. 2015;163(6 suppl):S1–S40.
- , , , et al. Practice guidelines for outpatient parenteral antimicrobial therapy. IDSA guidelines. Clin Infect Dis. 2004;38(12):1651–1672.
- , . Nonuniform risk of bloodstream infection with increasing central venous catheter‐days. Infect Control Hosp Epidemiol. 2005;26(8):715–719.
- , , , et al. Risk of venous thromboembolism associated with peripherally inserted central catheters: a systematic review and meta‐analysis. Lancet. 2013;382(9889):311–325.
- , , , , . Hospitalist experiences, practice, opinions, and knowledge regarding peripherally inserted central catheters: results of a national survey. J Hosp Med. 2013;8(11):635–638.
- , , , et al. Do clinicians know which of their patients have central venous catheters? Ann Intern Med. 2014;161(8):562.
- , , , , , . Reducing foley catheter device days in an intensive care unit: using the evidence to change practice. AACN Adv Crit Care. 2006;17(3):272–283.
Infections acquired in the hospital are termed healthcare‐associated infections (HAIs) and include central lineassociated blood stream infections (CLABSIs). Among HAIs, CLABSIs cause the highest number of preventable deaths.[1] Central venous catheters (CVCs) or central lines are commonly used in the hospital.[2] Each year their use is linked to 250,000 cases of CLABSIs in the United States.[3] Some CLABSIs may be prevented by the prompt removal of the line.[4] However, CVCs are often retained after their clinical indication has lapsed and are then referred to as idle lines.[5, 6] In this work, we propose and theoretically test a guideline to facilitate the safe removal of an idle line by observing the agreement and disagreement between actual practice and the proposed guideline.
METHODS
Setting
This work was conducted at a large, urban, tertiary care, academic health center in the United States as a collaborative effort to improve quality at our institution.[7]
Design and Patients
The reports linked with the electronic medical records at our institution include a daily, ward‐by‐ward listing of patients who have access other than a peripheral line in place. This central line dashboard accesses the information on intravenous access charted by bedside nurses to create a list of patients on every ward who have any kind of central access. Temporary central venous lines (CVLs), peripherally inserted central catheters (PICCs), ports, and dialysis catheters are all included. The unit charge nurses and managers use this dashboard to facilitate compliance with line care bundles. We used this source to identify patients with either type of CVC (CVLs or PICCs) on 8 days in August 2014, September 2014, and October 2014. Patients were included if they had a CVC and were on a general medical or surgical ward bed on audit day. CVLs at all sites were included (femoral, subclavian, and internal jugular). Patients in an intensive care unit (ICU) or progressive care unit on the day of the audit were excluded. Patients whose catheters were for chemotherapy and those admitted for a transplant or receiving palliative or hospice care were also excluded.
Data Collection
A protocol for data collection was written out, and a training session was held to review definitions, data sources, and methods to ensure consistency. Two authors (M.M. and J.D.) assisted by an experienced clinical nurse specialist collected data on the patients captured on audit days. Each chart was reviewed on the day of the audit, the 2 days preceding the audit day, and then followed until the patient was either discharged from the hospital or transferred to a higher level of care, died, or transitioned to palliative or hospice care. Demographics, details about the line, and the criteria for justified use were extracted from the electronic medical record.
Definitions
Justified and Idle Days
To justify the presence of a CVC on any given day, we used criteria that fell under 3 categories: intravenous (IV) access needs, unstable vitals, or meeting sepsis/systemic inflammatory response syndrome (SIRS) criteria (Table 1). For vital signs, a single abnormal reading was counted as fulfilling criteria for that day. If no criterion for justified use was met, the line was considered idle for that day.
|
| IV access needs |
| Expected duration of IV antibiotics >6 days |
| Administration of TPN |
| Anticipated requirement of home IV medications |
| Requirement of IV medications with documented difficult access |
| Hemorrhage requiring blood transfusions |
| Requiring more than 3 infusions |
| Requiring more than 2 infusions and blood transfusions |
| Abnormal vitals |
| Diastolic blood pressure >120 mm Hg |
| Systolic blood pressure 90 mm Hg |
| Systolic blood pressure >200 mm Hg |
| Heart rate >120 beats per minute |
| Heart rate 50 beats per minute |
| Respiratory rate >30 breaths per minute |
| Respiratory rate 10 breaths per minute |
| Oxygen saturation 90% as measured by pulse oximetry |
| Meeting SIRS criteria (2 or more of the following present) |
| Temp >38C, Temp 36C, heart rate >90 beats per minute, respiratory rate >20 breaths per minute, WBC >12,000/mm3, WBC 1,000/mm3, bandemia >10% |
Qualifying IV access needs were defined similarly to those previously used,[5, 6] whereas those for SIRS followed the current consensus.[8] To determine the number of IV medications or infusions, the medication administration record was reviewed. If 3 or more infusions were found, their compatibility was checked using the same database that nurses use at our institution. Difficult IV access was inferred from the indication for line placement, coupled with the absence of documentation of a peripheral IV. Clinical progress notes were reviewed to extract information on the length of proposed IV antibiotic courses, and discharge instructions were reviewed to verify whether the line was removed prior to discharge or not. The cutoffs for diastolic blood pressure, respiratory rate, and oxygen saturation used to label patients hemodynamically labile are the same as those used by previous authors and also constitute the definition of hypertensive urgency.[5, 9] However, we diverged from the values previously used for tachycardia, bradycardia, and systolic hypotension using heart rates >120 and 50 beats per minute (compared to >130 and 40 beats per minute) and systolics 90 mm Hg (compared to 80 mm Hg) to justify the line.[5] Early warning scores have been used to identify hospitalized ward patients who are at risk for clinical deterioration. Although each score utilizes different thresholds, the risk for clinical deterioration increases as the vitals worsen.[10] Bearing this in mind, the thresholds we elected to use are more clinically conservative and also parallel the nursing call orders currently used at our institution.
Proposed Guideline
We propose the guideline that a CVC may be safely removed the day after the first idle day.
RESULTS
A total of 126 lines were observed in 126 patients. Eighty‐three (65.9%) of the lines were PICCs. The remaining 43 (34.1%) were CVLs. The indications for line placement were distributed between the need for central access, total parenteral nutrition, or antibiotics (Table 2).
| Description | Value |
|---|---|
| |
| Age in yrs mean (SD) | 55.7 (18) |
| Gender, n (%) | |
| Female | 66 (52.4) |
| Male | 60 (47.6) |
| Type of line, n (%) | |
| PICC | 83 (65.9) |
| CVL | 43 (34.1) |
| Indication for line placement, n (%) | |
| Meds requiring central access or TPN | 36 (28.6) |
| Antibiotics | 34 (27.0) |
| Hemodynamic instability | 30 (23.8) |
| Poor access with multiple IV medications | 18 (14.3) |
| Unknown | 8 (6.3) |
| Line removed prior to discharge, n (%) | |
| Yes | 76 (60.3) |
| No | 50 (39.7) |
Out of the 126 patients, 50 (39.7%) were discharged from the hospital, died, were transferred to a higher level of care, or transitioned to palliative or hospice care with the line in place. In the remaining 76 patients, the audit captured 635 days, out of which a line was in place for 522 (82.2%) days. Of these 522 days, the line's presence was justified by our criteria for 351 (67.2%) days. The most common reason for a line to be justified on any given day was the need for antibiotics followed by the presence of SIRS criteria (Table 3). The remaining 171 (32.7%) days were idle.
| Criteria | N | % |
|---|---|---|
| ||
| No. of factors justifying use | ||
| 1 | 184 | 52.4% |
| 2 | 127 | 36.2% |
| >2 | 40 | 11.4 |
| Reason for justifying line* | ||
| Anticipate home or >6 days of antibiotic use | 181 | 51.6 |
| SIRS criteria | 124 | 35.3 |
| TPN | 96 | 27.4 |
| Hemodynamic instability based on hr and bp | 78 | 22.2 |
| Poor access with need for IV medications | 57 | 16.2 |
| Respiratory rate (10 or >30/minute) | 25 | 7.1 |
| Active hemorrhage requiring transfusions | 12 | 3.4 |
| >3 infusions | 6 | 1.7 |
A comparison of the actual removal of the 76 central lines in practice relative to the proposed guideline of removing it the day following the first idle day is displayed in Figure 1. The central line was removed prior to our proposed guideline in 11 (14.5%) patients, and waiting for an idle day in these patients would have added 46 line days. In almost half the patients (n = 36, 47.4%), the line was removed in agreement with the proposed guideline. None of the patients in whom the line was removed prior to or in accordance with our proposed guideline required a line reinsertion. Line removal was delayed in 29 (38.2%) patients when compared to our proposed guideline. In these patients, following the guideline would have created 122 line‐free days. Most (n = 102, 83.6%) of these potential line‐free days were idle. Twenty (16.4%) were justified, of which half (n = 10) were justified by meeting SIRS criteria.

DISCUSSION
Approximately 1 in every 25 inpatients in the United States has at least 1 HAI on any given day.[11] The case fatality rate from a CLABSI may be as high as 12%, and up to 70% of these infections may be preventable.[1, 12] Interventions successful in decreasing CLABSIs have focused on patients in ICUs.[13] However, CVCs are increasingly prevalent outside the ICU, with over 4.5 million line days in non‐ICU beds reported to the National Healthcare Safety Network in 2012 compared to 2.5 million in 2010.[2, 14] However, adherence rates to infection control practices may be lower on the wards than in the ICUs.[6, 15] Consequently, although the number of CLABSIs has declined over the last decade, most are now occurring outside the ICU.[16] These trends underscore the need to develop strategies aimed at CLABSI prevention on the floors.
Analogous to the life cycle of a urinary catheter described by Meddings et al.,[17] strategies to prevent CLABSIs and other CVC‐related complications may be designed around the life cycle of a CVC. The life cycle starts with insertion and moves on to the maintenance, removal, and possible reinsertion of the line. The process thus starts with the decision to place the line. Over the last decade, this decision making has changed in part due to PICCs. This shift is reflected in PICC prevalence rates: in 2001, 11% of audited central lines were PICCs compared to 56% in 2007.[5, 6] In our audit, 66% of the CVCs were PICCs. This increase in the use of PICCs may be attributable to the ease and safety of their placement coupled with the increased availability of vascular access placement teams.[18] The risk of overuse that may result from such expediency may be countered by adhering to guidelines such as the Michigan Appropriateness Guide for Intravenous Catheters, which provides both clinically detailed guidance and an impetus for reflective decision making around intravenous access.[19]
The placement of CVCs for prolonged parenteral antibiotics may be a particular subset that bears further exploration. Similar to previous reports, we found that a large number of the CVCs were both inserted for and justified by the need for IV antibiotics.[5] Guidelines delineated by the Infectious Diseases Society of America regarding outpatient parenteral antibiotics weigh both the duration of therapy and the antimicrobial's potential for causing phlebitis when recommending the type of intravascular access.[20] Many courses may therefore be completed through peripheral or midline catheters. Developing strong partnerships between infectious disease specialists, hospitalists, and the facilities or home‐care services treating these patients may curtail the use of CVCs for antimicrobial administration.
The main focus of our work is on facilitating the safe removal of CVCs. The risk of CLABSIs increases each day a CVC is in place, and guidelines to prevent CLABSIs include recommendations to promptly remove nonessential catheters.[4, 21] There is also an emerging understanding that the risk of a PICC‐related CLABSI approaches that from a traditional central line in hospitalized patients, and PICCs confer an increased risk of venous thromboembolism.[18, 22] Although nearly half of surveyed hospitalists recently reported leaving PICCs in place until discharge day, our data suggest that this practice may be driven by the trajectory of a patient's recovery as much as by knowledge gaps related to the use of PICCs.[23] In nearly half the instances, clinical practice already mirrors our proposed guideline, with line removal coinciding with both the timing proposed by our guideline and discharge day. However, there is room for improvement, as line removal may have been expedited in the 29 patients in whom the line was retained after the first idle day. Maintaining an awareness of its presence and weighing its risks and benefits daily may facilitate the removal of a CVC. Based on the recent findings that up to a quarter of clinicians are unaware that their patients have a central line, the mere reminder of the presence of a line using such criteria may expedite its removal by triggering a purposeful reassessment of its ongoing need.[24] Premature CVC removal requiring line reinsertion is an unintended consequence that may emerge from the earlier removal of lines. In our sample, none of the patients who had lines removed either prior to or in accordance with our proposed guideline required a line reinsertion. In addition to line reinsertion, delays in laboratory testing and reporting due to the unavailability of access, increased patient discomfort, or increased workload on the bedside nurse or vascular access team must also be considered when implementing strategies aimed at decreasing line days.
We envisage using these criteria to both empower practitioners with knowledge and foster shared accountability between all team members by using a uniform tool. This can occur through partnerships between infection control, clinical nurse specialists, bedside nursing, and physicians. The electronic medical record could be leveraged to scan the record for the criteria and create a notification when the line becomes idle. In alignment with the Michigan Appropriateness Guide for Intravenous Catheters guidelines, we do not support the removal of lines by nursing staff without physician notification.[19] Such principles have been successfully harnessed in strategies to prevent both catheter‐associated urinary tract infections and CLABSIs in ICUs.[13, 25] In light of the complexity surrounding the decision making for CVCs, our criteria were focused on the wards and erred on the side of clinical caution. This clinical conservatism is apparent in the patients in whom lines were removed prior to what our guideline would propose, yet none of the patients required a line reinsertion. As concerns about recrudescent clinical instability may drive decision making around line removal, such conservatism may be warranted initially. However, the fidelity of these criteria in the clinical setting will need prospective validation. In particular, the inclusion of SIRS criteria may have led to an overestimation of justified days. Further studies may be needed to refine the criteria and find a clinical hierarchy that balances the risks and benefits of retaining a central line.
Our work has certain limitations. It is a single center's experience, and our findings may not therefore be generalizable. Except for when the indication for the line was for difficult access, we did not attempt to verify the presence of a peripheral IV. This, in combination with the inclusion of SIRS criteria, likely leads to an underestimation of idle days. In the interest of focusing on patients in whom the decision making around a line would be the least controversial, we did not continue to follow patients who were transferred to a higher level of care. It is possible, however, that these transfers were precipitated by line‐associated complications such as sepsis and would be important to track. We did not measure the agreement between data collectors, although definitions and methodologies were standardized and reviewed prior to data collection. As this was an observational assessment of a proposed guideline, we cannot predict how the recommendations generated by it will be received by clinicians. Although this may prove to be a barrier in adoption, we hope that the conversation it initiates leads to change.
Hospitalists are positioned to potentially influence the entire life cycle of a central line on the floor. Strategies can be enacted at each stage to help decrease the potential of harm from these devices to our patients. Creating and testing criteria and guidelines such as we propose represents just 1 such strategy in a multidisciplinary effort to provide the best possible care we can.
Acknowledgements
The authors thank Jennifer Dunscomb, Kristen Kelly, and their teams, and Deanna Sidwell, Todd Biggerstaff, Joan Miller, Rob Clark, and the tireless providers at Indiana University Health Methodist Hospital for their support.
Disclosures: This work was supported by the Indiana University Health Values Grant for research. The authors have no conflicts of interests to report.
Infections acquired in the hospital are termed healthcare‐associated infections (HAIs) and include central lineassociated blood stream infections (CLABSIs). Among HAIs, CLABSIs cause the highest number of preventable deaths.[1] Central venous catheters (CVCs) or central lines are commonly used in the hospital.[2] Each year their use is linked to 250,000 cases of CLABSIs in the United States.[3] Some CLABSIs may be prevented by the prompt removal of the line.[4] However, CVCs are often retained after their clinical indication has lapsed and are then referred to as idle lines.[5, 6] In this work, we propose and theoretically test a guideline to facilitate the safe removal of an idle line by observing the agreement and disagreement between actual practice and the proposed guideline.
METHODS
Setting
This work was conducted at a large, urban, tertiary care, academic health center in the United States as a collaborative effort to improve quality at our institution.[7]
Design and Patients
The reports linked with the electronic medical records at our institution include a daily, ward‐by‐ward listing of patients who have access other than a peripheral line in place. This central line dashboard accesses the information on intravenous access charted by bedside nurses to create a list of patients on every ward who have any kind of central access. Temporary central venous lines (CVLs), peripherally inserted central catheters (PICCs), ports, and dialysis catheters are all included. The unit charge nurses and managers use this dashboard to facilitate compliance with line care bundles. We used this source to identify patients with either type of CVC (CVLs or PICCs) on 8 days in August 2014, September 2014, and October 2014. Patients were included if they had a CVC and were on a general medical or surgical ward bed on audit day. CVLs at all sites were included (femoral, subclavian, and internal jugular). Patients in an intensive care unit (ICU) or progressive care unit on the day of the audit were excluded. Patients whose catheters were for chemotherapy and those admitted for a transplant or receiving palliative or hospice care were also excluded.
Data Collection
A protocol for data collection was written out, and a training session was held to review definitions, data sources, and methods to ensure consistency. Two authors (M.M. and J.D.) assisted by an experienced clinical nurse specialist collected data on the patients captured on audit days. Each chart was reviewed on the day of the audit, the 2 days preceding the audit day, and then followed until the patient was either discharged from the hospital or transferred to a higher level of care, died, or transitioned to palliative or hospice care. Demographics, details about the line, and the criteria for justified use were extracted from the electronic medical record.
Definitions
Justified and Idle Days
To justify the presence of a CVC on any given day, we used criteria that fell under 3 categories: intravenous (IV) access needs, unstable vitals, or meeting sepsis/systemic inflammatory response syndrome (SIRS) criteria (Table 1). For vital signs, a single abnormal reading was counted as fulfilling criteria for that day. If no criterion for justified use was met, the line was considered idle for that day.
|
| IV access needs |
| Expected duration of IV antibiotics >6 days |
| Administration of TPN |
| Anticipated requirement of home IV medications |
| Requirement of IV medications with documented difficult access |
| Hemorrhage requiring blood transfusions |
| Requiring more than 3 infusions |
| Requiring more than 2 infusions and blood transfusions |
| Abnormal vitals |
| Diastolic blood pressure >120 mm Hg |
| Systolic blood pressure 90 mm Hg |
| Systolic blood pressure >200 mm Hg |
| Heart rate >120 beats per minute |
| Heart rate 50 beats per minute |
| Respiratory rate >30 breaths per minute |
| Respiratory rate 10 breaths per minute |
| Oxygen saturation 90% as measured by pulse oximetry |
| Meeting SIRS criteria (2 or more of the following present) |
| Temp >38C, Temp 36C, heart rate >90 beats per minute, respiratory rate >20 breaths per minute, WBC >12,000/mm3, WBC 1,000/mm3, bandemia >10% |
Qualifying IV access needs were defined similarly to those previously used,[5, 6] whereas those for SIRS followed the current consensus.[8] To determine the number of IV medications or infusions, the medication administration record was reviewed. If 3 or more infusions were found, their compatibility was checked using the same database that nurses use at our institution. Difficult IV access was inferred from the indication for line placement, coupled with the absence of documentation of a peripheral IV. Clinical progress notes were reviewed to extract information on the length of proposed IV antibiotic courses, and discharge instructions were reviewed to verify whether the line was removed prior to discharge or not. The cutoffs for diastolic blood pressure, respiratory rate, and oxygen saturation used to label patients hemodynamically labile are the same as those used by previous authors and also constitute the definition of hypertensive urgency.[5, 9] However, we diverged from the values previously used for tachycardia, bradycardia, and systolic hypotension using heart rates >120 and 50 beats per minute (compared to >130 and 40 beats per minute) and systolics 90 mm Hg (compared to 80 mm Hg) to justify the line.[5] Early warning scores have been used to identify hospitalized ward patients who are at risk for clinical deterioration. Although each score utilizes different thresholds, the risk for clinical deterioration increases as the vitals worsen.[10] Bearing this in mind, the thresholds we elected to use are more clinically conservative and also parallel the nursing call orders currently used at our institution.
Proposed Guideline
We propose the guideline that a CVC may be safely removed the day after the first idle day.
RESULTS
A total of 126 lines were observed in 126 patients. Eighty‐three (65.9%) of the lines were PICCs. The remaining 43 (34.1%) were CVLs. The indications for line placement were distributed between the need for central access, total parenteral nutrition, or antibiotics (Table 2).
| Description | Value |
|---|---|
| |
| Age in yrs mean (SD) | 55.7 (18) |
| Gender, n (%) | |
| Female | 66 (52.4) |
| Male | 60 (47.6) |
| Type of line, n (%) | |
| PICC | 83 (65.9) |
| CVL | 43 (34.1) |
| Indication for line placement, n (%) | |
| Meds requiring central access or TPN | 36 (28.6) |
| Antibiotics | 34 (27.0) |
| Hemodynamic instability | 30 (23.8) |
| Poor access with multiple IV medications | 18 (14.3) |
| Unknown | 8 (6.3) |
| Line removed prior to discharge, n (%) | |
| Yes | 76 (60.3) |
| No | 50 (39.7) |
Out of the 126 patients, 50 (39.7%) were discharged from the hospital, died, were transferred to a higher level of care, or transitioned to palliative or hospice care with the line in place. In the remaining 76 patients, the audit captured 635 days, out of which a line was in place for 522 (82.2%) days. Of these 522 days, the line's presence was justified by our criteria for 351 (67.2%) days. The most common reason for a line to be justified on any given day was the need for antibiotics followed by the presence of SIRS criteria (Table 3). The remaining 171 (32.7%) days were idle.
| Criteria | N | % |
|---|---|---|
| ||
| No. of factors justifying use | ||
| 1 | 184 | 52.4% |
| 2 | 127 | 36.2% |
| >2 | 40 | 11.4 |
| Reason for justifying line* | ||
| Anticipate home or >6 days of antibiotic use | 181 | 51.6 |
| SIRS criteria | 124 | 35.3 |
| TPN | 96 | 27.4 |
| Hemodynamic instability based on hr and bp | 78 | 22.2 |
| Poor access with need for IV medications | 57 | 16.2 |
| Respiratory rate (10 or >30/minute) | 25 | 7.1 |
| Active hemorrhage requiring transfusions | 12 | 3.4 |
| >3 infusions | 6 | 1.7 |
A comparison of the actual removal of the 76 central lines in practice relative to the proposed guideline of removing it the day following the first idle day is displayed in Figure 1. The central line was removed prior to our proposed guideline in 11 (14.5%) patients, and waiting for an idle day in these patients would have added 46 line days. In almost half the patients (n = 36, 47.4%), the line was removed in agreement with the proposed guideline. None of the patients in whom the line was removed prior to or in accordance with our proposed guideline required a line reinsertion. Line removal was delayed in 29 (38.2%) patients when compared to our proposed guideline. In these patients, following the guideline would have created 122 line‐free days. Most (n = 102, 83.6%) of these potential line‐free days were idle. Twenty (16.4%) were justified, of which half (n = 10) were justified by meeting SIRS criteria.

DISCUSSION
Approximately 1 in every 25 inpatients in the United States has at least 1 HAI on any given day.[11] The case fatality rate from a CLABSI may be as high as 12%, and up to 70% of these infections may be preventable.[1, 12] Interventions successful in decreasing CLABSIs have focused on patients in ICUs.[13] However, CVCs are increasingly prevalent outside the ICU, with over 4.5 million line days in non‐ICU beds reported to the National Healthcare Safety Network in 2012 compared to 2.5 million in 2010.[2, 14] However, adherence rates to infection control practices may be lower on the wards than in the ICUs.[6, 15] Consequently, although the number of CLABSIs has declined over the last decade, most are now occurring outside the ICU.[16] These trends underscore the need to develop strategies aimed at CLABSI prevention on the floors.
Analogous to the life cycle of a urinary catheter described by Meddings et al.,[17] strategies to prevent CLABSIs and other CVC‐related complications may be designed around the life cycle of a CVC. The life cycle starts with insertion and moves on to the maintenance, removal, and possible reinsertion of the line. The process thus starts with the decision to place the line. Over the last decade, this decision making has changed in part due to PICCs. This shift is reflected in PICC prevalence rates: in 2001, 11% of audited central lines were PICCs compared to 56% in 2007.[5, 6] In our audit, 66% of the CVCs were PICCs. This increase in the use of PICCs may be attributable to the ease and safety of their placement coupled with the increased availability of vascular access placement teams.[18] The risk of overuse that may result from such expediency may be countered by adhering to guidelines such as the Michigan Appropriateness Guide for Intravenous Catheters, which provides both clinically detailed guidance and an impetus for reflective decision making around intravenous access.[19]
The placement of CVCs for prolonged parenteral antibiotics may be a particular subset that bears further exploration. Similar to previous reports, we found that a large number of the CVCs were both inserted for and justified by the need for IV antibiotics.[5] Guidelines delineated by the Infectious Diseases Society of America regarding outpatient parenteral antibiotics weigh both the duration of therapy and the antimicrobial's potential for causing phlebitis when recommending the type of intravascular access.[20] Many courses may therefore be completed through peripheral or midline catheters. Developing strong partnerships between infectious disease specialists, hospitalists, and the facilities or home‐care services treating these patients may curtail the use of CVCs for antimicrobial administration.
The main focus of our work is on facilitating the safe removal of CVCs. The risk of CLABSIs increases each day a CVC is in place, and guidelines to prevent CLABSIs include recommendations to promptly remove nonessential catheters.[4, 21] There is also an emerging understanding that the risk of a PICC‐related CLABSI approaches that from a traditional central line in hospitalized patients, and PICCs confer an increased risk of venous thromboembolism.[18, 22] Although nearly half of surveyed hospitalists recently reported leaving PICCs in place until discharge day, our data suggest that this practice may be driven by the trajectory of a patient's recovery as much as by knowledge gaps related to the use of PICCs.[23] In nearly half the instances, clinical practice already mirrors our proposed guideline, with line removal coinciding with both the timing proposed by our guideline and discharge day. However, there is room for improvement, as line removal may have been expedited in the 29 patients in whom the line was retained after the first idle day. Maintaining an awareness of its presence and weighing its risks and benefits daily may facilitate the removal of a CVC. Based on the recent findings that up to a quarter of clinicians are unaware that their patients have a central line, the mere reminder of the presence of a line using such criteria may expedite its removal by triggering a purposeful reassessment of its ongoing need.[24] Premature CVC removal requiring line reinsertion is an unintended consequence that may emerge from the earlier removal of lines. In our sample, none of the patients who had lines removed either prior to or in accordance with our proposed guideline required a line reinsertion. In addition to line reinsertion, delays in laboratory testing and reporting due to the unavailability of access, increased patient discomfort, or increased workload on the bedside nurse or vascular access team must also be considered when implementing strategies aimed at decreasing line days.
We envisage using these criteria to both empower practitioners with knowledge and foster shared accountability between all team members by using a uniform tool. This can occur through partnerships between infection control, clinical nurse specialists, bedside nursing, and physicians. The electronic medical record could be leveraged to scan the record for the criteria and create a notification when the line becomes idle. In alignment with the Michigan Appropriateness Guide for Intravenous Catheters guidelines, we do not support the removal of lines by nursing staff without physician notification.[19] Such principles have been successfully harnessed in strategies to prevent both catheter‐associated urinary tract infections and CLABSIs in ICUs.[13, 25] In light of the complexity surrounding the decision making for CVCs, our criteria were focused on the wards and erred on the side of clinical caution. This clinical conservatism is apparent in the patients in whom lines were removed prior to what our guideline would propose, yet none of the patients required a line reinsertion. As concerns about recrudescent clinical instability may drive decision making around line removal, such conservatism may be warranted initially. However, the fidelity of these criteria in the clinical setting will need prospective validation. In particular, the inclusion of SIRS criteria may have led to an overestimation of justified days. Further studies may be needed to refine the criteria and find a clinical hierarchy that balances the risks and benefits of retaining a central line.
Our work has certain limitations. It is a single center's experience, and our findings may not therefore be generalizable. Except for when the indication for the line was for difficult access, we did not attempt to verify the presence of a peripheral IV. This, in combination with the inclusion of SIRS criteria, likely leads to an underestimation of idle days. In the interest of focusing on patients in whom the decision making around a line would be the least controversial, we did not continue to follow patients who were transferred to a higher level of care. It is possible, however, that these transfers were precipitated by line‐associated complications such as sepsis and would be important to track. We did not measure the agreement between data collectors, although definitions and methodologies were standardized and reviewed prior to data collection. As this was an observational assessment of a proposed guideline, we cannot predict how the recommendations generated by it will be received by clinicians. Although this may prove to be a barrier in adoption, we hope that the conversation it initiates leads to change.
Hospitalists are positioned to potentially influence the entire life cycle of a central line on the floor. Strategies can be enacted at each stage to help decrease the potential of harm from these devices to our patients. Creating and testing criteria and guidelines such as we propose represents just 1 such strategy in a multidisciplinary effort to provide the best possible care we can.
Acknowledgements
The authors thank Jennifer Dunscomb, Kristen Kelly, and their teams, and Deanna Sidwell, Todd Biggerstaff, Joan Miller, Rob Clark, and the tireless providers at Indiana University Health Methodist Hospital for their support.
Disclosures: This work was supported by the Indiana University Health Values Grant for research. The authors have no conflicts of interests to report.
- , , , , , . Estimating the proportion of healthcare‐associated infections that are reasonably preventable and the related mortality and costs. Infect Control Hosp Epidemiol. 2011;32(2):101–114.
- , , , et al. National Healthcare Safety Network (NHSN) report, data summary for 2012, device‐associated module. Am J Infect Control. 2013;41(12):1148–1166.
- , , . The risk of bloodstream infection in adults with different intravascular devices: a systematic review of 200 published prospective studies. Mayo Clin Proc. 2006;81(9):1159–1171.
- , , , et al. Guidelines for the prevention of intravascular catheter‐related infections. Clin Infect Dis. 2011;52(9):e162–e193.
- , , , et al. Temporary central venous catheter utilization patterns in a large tertiary care center: tracking the “idle central venous catheter.” Infect Control Hosp Epidemiol. 2012;33(1):50–57.
- , , , , , . Unnecessary use of central venous catheters: the need to look outside the intensive care unit. Infect Control Hosp Epidemiol. 2004;25(3):266–268.
- IU Health Methodist Hospital website. Available at: http://iuhealth.org/methodist/aboIut. Accessed October 20, 2014.
- , , , et al. Definitions for Sepsis and Organ Failure and Guidelines for the Use of Innovative Therapies in Sepsis. The ACCP/SCCM Consensus Conference Committee. American College of Chest Physicians/Society of Critical Care Medicine. Chest. 2009;136(5 suppl):e28.
- , , , , , . Acute hypertension: a systematic review and appraisal of guidelines. Ochsner J. 2014;14(4):655–663.
- , , . Predicting clinical deterioration in the hospital: the impact of outcome selection. Resuscitation. 2013;84(5):564–568.
- , , , et al. Multistate point‐prevalence survey of health care–associated infections. N Engl J Med. 2014;370(13):1198–1208.
- , , , et al. Estimating health care‐associated infections and deaths in U.S. hospitals, 2002. Public Health Rep. 2007;122(2):160–166.
- , , , et al. An intervention to decrease catheter‐related bloodstream infections in the ICU. N Engl J Med. 2006;355(26):2725–2732.
- , , , et al. Data summary for 2011, device‐associated module. Centers for Disease Control and Prevention. National Healthcare Safety Network (NHSN) Report. Available at: http://www.cdc.gov/nhsn/PDFs/dataStat/NHSN‐Report‐2011‐Data‐Summary.pdf. Published April 1, 2013. Last accessed January 2015.
- , , . Idle central venous catheter‐days pose infection risk for patients after discharge from intensive care. Am J Infect Control. 2014;42(4):453–455.
- , . Update on emerging infections: news from the Centers for Disease Control and Prevention. Vital signs: central line‐associated blood stream infections—United States, 2001, 2008, and 2009. Ann Emerg Med. 2011;58(5):447–451.
- , , , , , . Reducing unnecessary urinary catheter use and other strategies to prevent catheter‐associated urinary tract infection: an integrative review. BMJ Qual Saf. 2014;23(4):277–289.
- , , , , . The risk of bloodstream infection associated with peripherally inserted central catheters compared with central venous catheters in adults: a systematic review and meta‐analysis. Infect Control Hosp Epidemiol. 2013;34(9):908–918.
- , , , et al. The Michigan Appropriateness Guide for Intravenous Catheters (MAGIC): results from a multispecialty panel using the RAND/UCLA Appropriateness Method. Ann Intern Med. 2015;163(6 suppl):S1–S40.
- , , , et al. Practice guidelines for outpatient parenteral antimicrobial therapy. IDSA guidelines. Clin Infect Dis. 2004;38(12):1651–1672.
- , . Nonuniform risk of bloodstream infection with increasing central venous catheter‐days. Infect Control Hosp Epidemiol. 2005;26(8):715–719.
- , , , et al. Risk of venous thromboembolism associated with peripherally inserted central catheters: a systematic review and meta‐analysis. Lancet. 2013;382(9889):311–325.
- , , , , . Hospitalist experiences, practice, opinions, and knowledge regarding peripherally inserted central catheters: results of a national survey. J Hosp Med. 2013;8(11):635–638.
- , , , et al. Do clinicians know which of their patients have central venous catheters? Ann Intern Med. 2014;161(8):562.
- , , , , , . Reducing foley catheter device days in an intensive care unit: using the evidence to change practice. AACN Adv Crit Care. 2006;17(3):272–283.
- , , , , , . Estimating the proportion of healthcare‐associated infections that are reasonably preventable and the related mortality and costs. Infect Control Hosp Epidemiol. 2011;32(2):101–114.
- , , , et al. National Healthcare Safety Network (NHSN) report, data summary for 2012, device‐associated module. Am J Infect Control. 2013;41(12):1148–1166.
- , , . The risk of bloodstream infection in adults with different intravascular devices: a systematic review of 200 published prospective studies. Mayo Clin Proc. 2006;81(9):1159–1171.
- , , , et al. Guidelines for the prevention of intravascular catheter‐related infections. Clin Infect Dis. 2011;52(9):e162–e193.
- , , , et al. Temporary central venous catheter utilization patterns in a large tertiary care center: tracking the “idle central venous catheter.” Infect Control Hosp Epidemiol. 2012;33(1):50–57.
- , , , , , . Unnecessary use of central venous catheters: the need to look outside the intensive care unit. Infect Control Hosp Epidemiol. 2004;25(3):266–268.
- IU Health Methodist Hospital website. Available at: http://iuhealth.org/methodist/aboIut. Accessed October 20, 2014.
- , , , et al. Definitions for Sepsis and Organ Failure and Guidelines for the Use of Innovative Therapies in Sepsis. The ACCP/SCCM Consensus Conference Committee. American College of Chest Physicians/Society of Critical Care Medicine. Chest. 2009;136(5 suppl):e28.
- , , , , , . Acute hypertension: a systematic review and appraisal of guidelines. Ochsner J. 2014;14(4):655–663.
- , , . Predicting clinical deterioration in the hospital: the impact of outcome selection. Resuscitation. 2013;84(5):564–568.
- , , , et al. Multistate point‐prevalence survey of health care–associated infections. N Engl J Med. 2014;370(13):1198–1208.
- , , , et al. Estimating health care‐associated infections and deaths in U.S. hospitals, 2002. Public Health Rep. 2007;122(2):160–166.
- , , , et al. An intervention to decrease catheter‐related bloodstream infections in the ICU. N Engl J Med. 2006;355(26):2725–2732.
- , , , et al. Data summary for 2011, device‐associated module. Centers for Disease Control and Prevention. National Healthcare Safety Network (NHSN) Report. Available at: http://www.cdc.gov/nhsn/PDFs/dataStat/NHSN‐Report‐2011‐Data‐Summary.pdf. Published April 1, 2013. Last accessed January 2015.
- , , . Idle central venous catheter‐days pose infection risk for patients after discharge from intensive care. Am J Infect Control. 2014;42(4):453–455.
- , . Update on emerging infections: news from the Centers for Disease Control and Prevention. Vital signs: central line‐associated blood stream infections—United States, 2001, 2008, and 2009. Ann Emerg Med. 2011;58(5):447–451.
- , , , , , . Reducing unnecessary urinary catheter use and other strategies to prevent catheter‐associated urinary tract infection: an integrative review. BMJ Qual Saf. 2014;23(4):277–289.
- , , , , . The risk of bloodstream infection associated with peripherally inserted central catheters compared with central venous catheters in adults: a systematic review and meta‐analysis. Infect Control Hosp Epidemiol. 2013;34(9):908–918.
- , , , et al. The Michigan Appropriateness Guide for Intravenous Catheters (MAGIC): results from a multispecialty panel using the RAND/UCLA Appropriateness Method. Ann Intern Med. 2015;163(6 suppl):S1–S40.
- , , , et al. Practice guidelines for outpatient parenteral antimicrobial therapy. IDSA guidelines. Clin Infect Dis. 2004;38(12):1651–1672.
- , . Nonuniform risk of bloodstream infection with increasing central venous catheter‐days. Infect Control Hosp Epidemiol. 2005;26(8):715–719.
- , , , et al. Risk of venous thromboembolism associated with peripherally inserted central catheters: a systematic review and meta‐analysis. Lancet. 2013;382(9889):311–325.
- , , , , . Hospitalist experiences, practice, opinions, and knowledge regarding peripherally inserted central catheters: results of a national survey. J Hosp Med. 2013;8(11):635–638.
- , , , et al. Do clinicians know which of their patients have central venous catheters? Ann Intern Med. 2014;161(8):562.
- , , , , , . Reducing foley catheter device days in an intensive care unit: using the evidence to change practice. AACN Adv Crit Care. 2006;17(3):272–283.
Redesigning Inpatient Care
Despite an estimated annual $2.6 trillion expenditure on healthcare, the United States performs poorly on indicators of health and harm during care.[1, 2, 3] Hospitals around the nation are working to improve the care they deliver. We describe a model developed at our institution and report the evaluation of the outcomes associated with its implementation on the general medical and surgical units. The Indiana University Institutional Review Board approved this work.
SETTING AND DEFINITIONS
Indiana University Health Methodist Hospital (MH) is an academic center in Indianapolis, Indiana, serving over 30,000 patients annually.[4] In 2012, responding to the coexisting needs to improve quality and contain costs, the MH leadership team redesigned care in the hospital. The new model centers around accountable care teams (ACTs). Each ACT is a geographically defined set of providers accepting ownership for the clinical, service, and financial outcomes of their respective inpatient unit. The units studied are described in Table 1.
| Unit | No. of Beds | Predominant Diagnosis (Maximum Domain Score)* | |
|---|---|---|---|
| |||
| Medical units with progressive‐care beds | 1 | 33 | Pulmonary (3.4, 3.5, 5) |
| 2 | 28 | Cardiology (4.8, 3.5, 4) | |
| 3 | 24 | General medical (4.8, 3.5, 4) | |
| Medical units without progressive‐care beds | 4 | 36 | Renal/diabetic (4, 3.5, 5) |
| 5 | 24 | General medical (3.75, 4, 5) | |
| Surgical units with progressive‐care beds | 6 | 51 | Cardiothoracic surgery/cardiology (4, 4, 5) |
| 7 | 29 | Trauma/general surgery (3.75, 3.5, 5) | |
| 8 | 23 | Neurosurgical/neurological (4.8, 5, 5) | |
| 9 | 24 | Neurosurgical/neurological (4.4, 4.5, 5) | |
| Surgical units without progressive‐care beds | 10 | 29 | General/urologic/gynecologic/plastic surgery (3.4, 3, 2) |
| 11 | 26 | Orthopedic surgery (4.6, 4, 5) | |
THE ACT MODEL
The model comprises 8 interventions rooted in 3 foundational domains: (1) enhancing interprofessional collaboration (IPC), (2) enabling data‐driven decisions, and (3) providing leadership. Each intervention is briefly described under its main focus (see Supporting Information, Appendix A, in the online version of this article for further details).
Enhancing IPC
Geographical Cohorting of Patients and Providers
Hospitalist providers are localized for 4 consecutive months to 1 unit. An interdisciplinary team including a case manager, clinical nurse specialist, pharmacist, nutritionist, and social worker also serve each unit. Learners (residents, pharmacy, and medical students) are embedded in the team when rotating on the hospital medicine service. The presence of unit‐based nurse managers and charge nurses predates the model and is retained.
Bedside Collaborative Rounding
Geographically cohorted providers round on their patients with the bedside nurse guided by a customizable script.
Daily Huddle
The hospitalist, learners, and the interdisciplinary team for the unit meet each weekday to discuss patients' needs for a safe transition out of the hospital. Each unit determined the timing, location, and script for the huddle while retaining the focus on discharge planning (see Supporting Information, Appendix A2, in the online version of this article for a sample script).
Hospitalist and Specialty Comanagement Agreements
Guidelines delineating responsibilities for providers of each specialty were developed. Examples include orders pertaining to the management of a dialysis catheter in a patient with end‐stage renal disease, the removal of drains in postsurgical patients, and wound care.
Unit White Board
Each unit has a white board at the nursing station. Similar to the huddle, it is focused on discharge planning.
Enabling Data‐Driven Decisions
Monthly Review of Unit‐Level Data
Data analytics at our institution developed a data dashboard. Key metrics including length of stay (LOS), patient satisfaction scores, readmission rates, and costs are tracked and attributed to the discharging unit. The data are collated monthly by the ACT program director and distributed to each unit's leadership. Monthly interdisciplinary meetings are held to review trends. Learners are encouraged but not required to attend.
Weekly Patient Satisfaction Rounding
The unit's nurse manager and physician leader conduct weekly satisfaction rounds on patients. The conversation is open‐ended and focused on eliciting positive and negative experiences.
Providing Leadership
Designated hospitalist and, where relevant, specialty leaders are committed to serve each unit for at least 1 year as a resource for both medical and operational problem solving. The leader stays closely connected with the unit's nurse manager. In addition to day‐to‐day troubleshooting, the leader is responsible for monitoring outcome trends. There is currently no stipend, training, or other incentive offered for the role.
Implementation Timelines and ACT Scores
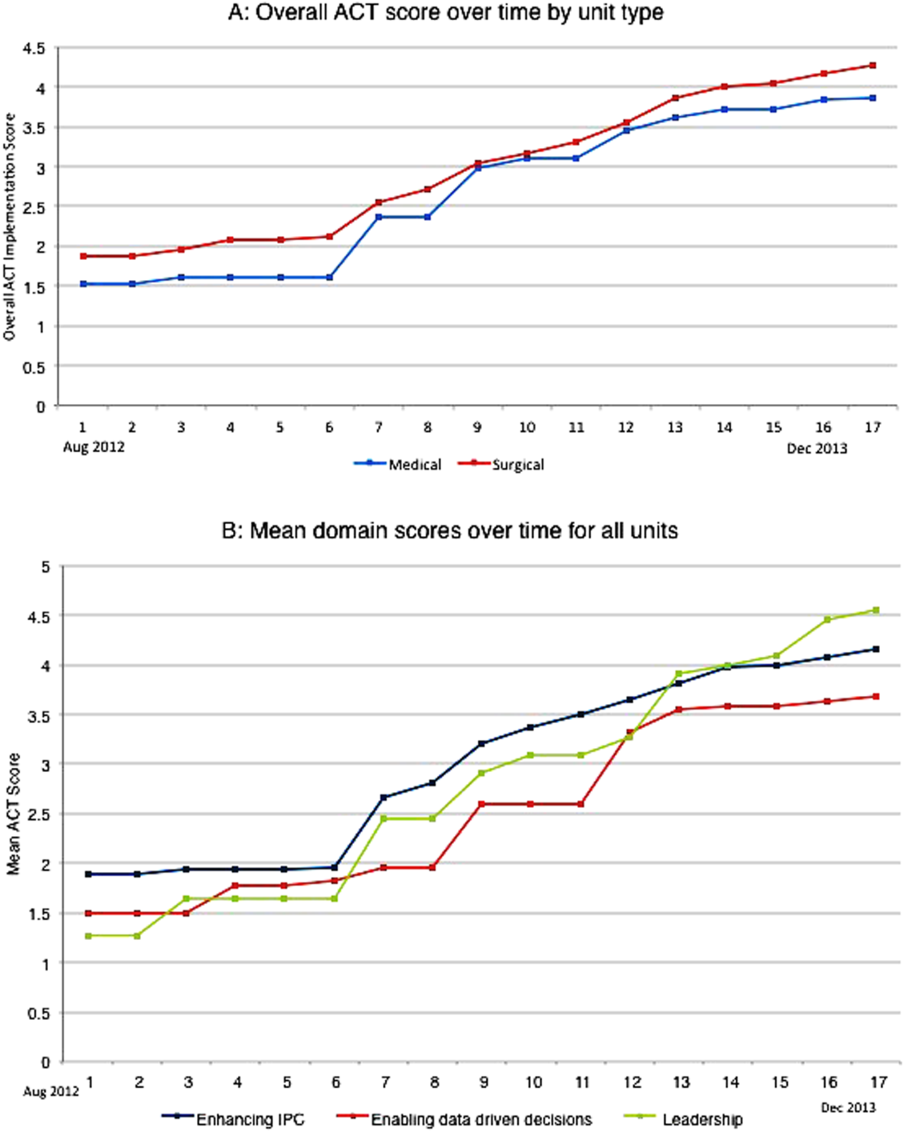
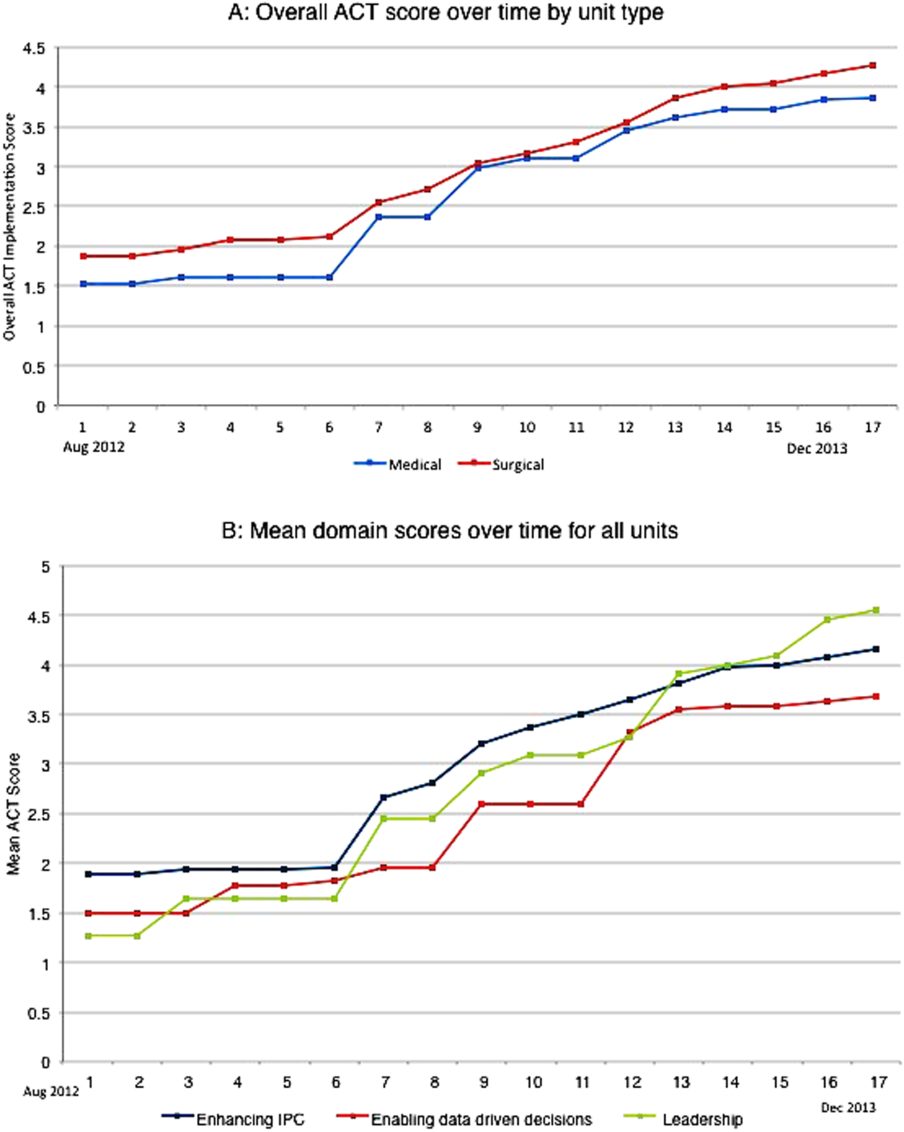
The development of the ACTs started in the spring of 2012. Physician, nursing, and pharmacy support was sought, and a pilot unit was formed in August 2012. The model was cascaded hospital wide by December 2013, with support from the ACT program director (A.N.). The program director observed and scored the uptake of each intervention by each unit monthly. A score of 1 denoted no implementation, whereas 5 denoted complete implementation. The criteria for scoring are presented in Table 2. The monthly scores for all 8 interventions in each of the 11 units were averaged as an overall ACT score, which reflects the implementation dose of the ACT model. Monthly domain scores for enhancing IPC and enabling data‐driven decisions were also calculated as the average score within each domain. This yielded 3 domain scores. Figure 1A plots by month the overall ACT score for the medical and surgical units, and Figure 1B plots the implementation score for the 3 domains between August 2012 and December 2013 for all units. The uptake of the interventions varied between units. This allowed our analysis to explore the dose relationships between the model and outcomes independent of underlying time trends that may be affected by concomitant initiatives.
| 1 | 2 | 3 | 4 | 5 | |
|---|---|---|---|---|---|
| |||||
| Geographical cohorting of patients and the ACT* | None | At least 1 discipline comprising the ACT is unit based | All disciplines comprising the ACT except the hospitalist unit based | All disciplines including the hospitalist unit based | 4 + 80% of hospitalist provider's patients on the unit |
| Bedside collaborative rounding | None | Occurring 1 day a week on at least 25% of the patients on the unit | Occurring 2 to 3 days a week on at least 50% of the patients on the unit | Occurring 3 to 4 days a week on at least 75% of the patients on the unit | Occurring MondayFriday on all patients on the unit |
| Daily huddle | None | Occurring daily, 1 out of 4 ACT disciplines represented, at least 25% of patients on the unit discussed | Occurring daily, 2 out of 4 ACT disciplines represented, at least 50% of patients on the unit discussed | Occurring daily, 3 out of 4 ACT disciplines represented, at least 75% of patients on the unit discussed | Occurring daily, all disciplines of the ACT represented, all patients on the unit discussed |
| Hospitalist and specialty comanagement agreements | None | One out of 3 specialists represented on the unit collaborating with the hospitalists on at least 25% of relevant patients | One out of 3 specialists represented on the unit collaborating with the hospitalists on at least 50% of relevant patients | Two out of 3 specialists on the unit collaborating with the hospitalists on at least 75% of relevant patients | All specialists on the unit collaborating with the hospitalists on all relevant patients on the unit |
| Unit white board | None | Present but only used by nursing | Present and used by all ACT disciplines except physician providers | Present and used by entire ACT; use inconsistent | Present and used MondayFriday by all disciplines of ACT |
| Monthly review of unit level data | None | Nurse manager reviewing data with ACT program director | Nurse manager and unit leader reviewing data with ACT program director | Meeting either not consistently occurring monthly or not consistently attended by entire ACT | Monthly meeting with entire ACT |
| Weekly patient satisfaction rounding | None | Nurse manager performing up to 1 week a month | Nurse manager performing weekly | Nurse and physician leader performing up to 3 times a month | Nurse and physician leader performing weekly |
| Leadership | None | For units with specialties, either hospitalist or specialist leader identified | Both hospitalist and specialist leader Identified | Both hospitalist and specialist leaders (where applicable) identified and partially engaged in leadership role | Both hospitalist and specialist leaders (where applicable) identified and engaged in leadership role |
Outcomes
Monthly data between August 2012 and December 2013 were analyzed.
Measures of Value
MH is a member of the University Health Consortium, which measures outcomes of participants relative to their peers. MH measures LOS index as a ratio of observed LOS to expected LOS that is adjusted for severity of illness.[5]
Variable direct costs (VDCs) are costs that a hospital can save if a service is not provided.[6] A hospital's case‐mix index (CMI) represents the average diagnosis‐related group relative weight for that hospital. We track VDCs adjusted for CMI (CMI‐adjusted VDC).[7]
Thirty‐day readmission rate is the percentage of cases that are readmitted to MH within 30 days of discharge from the index admission.[8]
Measures of Patient Satisfaction
The Hospital Consumer Assessment of Healthcare Providers and Systems (HCAHPS) survey covers topics relevant to a patient's experience in the hospital.[9] Patient satisfaction scores are tracked by responses to the HCAHPS survey.
Measures of Provider Satisfaction
Hospitalist and specialty providers, leadership, and case management teams were surveyed via email through SurveyMonkey in July 2014. The survey included Likert responses that elicited opinions and comments about the ACT model.
Statistical Methods
The primary predictor of interest was the monthly overall ACT score. We also explored the domain scores as well as the individual scores for each intervention. Generalized linear mixed models were fit to investigate the association between each predictor (overall ACT score, ACT domain scores, and individual implementation scores) and each outcome (LOS index, CMI‐adjusted VDC, 30‐day readmission rate, and overall patient satisfaction). The model for testing each ACT score also included covariates of inpatient units as a random effect, as well as date and type of unit as fixed effects. We set the statistical significance level at 0.01 and reported 99% confidence intervals.
Descriptive statistics were used to report the provider satisfaction survey results.
RESULTS
The overall ACT score was associated with LOS index and CMI‐adjusted VDC (both P < 0.001). For every 1‐unit increase in the overall ACT score, LOS index decreased by 0.078 and CMI‐adjusted VDC decreased by $273.99 (Table 3).
| Length of Stay Index | CMI Adjusted VDC | |||
|---|---|---|---|---|
| Estimate (99% CI)* | P Value | Estimate (99% CI)* | P Value | |
| ||||
| Overall ACT Score | 0.078 (0.123 to 0.032) | <0.001 | 274.0 (477.31 to 70.68) | <0.001 |
| Enhancing IPC | 0.071 (0.117 to 0.026) | <0.001 | 284.7 (488.08 to 81.23) | <0.001 |
| Enabling data‐driven decisions | 0.044 (0.080 to 0.009) | 0.002 | 145.4 (304.57 to 13.81) | 0.02 |
| Providing leadership | 0.027 (0.049 to 0.005) | 0.001 | 69.9 (169.00 to 29.26) | 0.07 |
Looking at domains, enhancing IPC resulted in statistically significant decreases in both LOS index and CMI‐adjusted VDC, but providing leadership and enabling data‐driven decisions decreased only the LOS index. Most of the 8 individual interventions were associated with at least 1 of these 2 outcomes. (Even where the associations were not significant, they were all in the direction of decreasing LOS and cost). In these models, the covariate of type of units (medical vs surgical) was not associated with LOS or cost. There was no significant time trend in LOS or cost, except in models where an intervention had no association with either outcome. Inclusion of all individual effective interventions in the same statistical model to assess their relative contributions was not possible because they were highly correlated (correlations 0.450.89).
Thirty‐day readmissions and patient satisfaction were not significantly associated with the overall ACT score, but exploratory analyses showed that patient satisfaction increased with the implementation of geographical cohorting (P = 0.007).
Survey Results
The response rate was 87% (96/110). Between 85% and 96% of respondents either agreed or strongly agreed that the ACT model had improved the quality and safety of the care delivered, improved communication between providers and patients, and improved their own engagement and job satisfaction. Overall, 78% of the respondents either agreed or strongly agreed that the model improved efficiency (Table 4). Suggestions for improvements revolved around increasing the emphasis on patient centeredness and bedside nursing engagement.
| The ACT Model | Strongly Agree, n (%) | Agree, n (%) | Disagree, n (%) | Strongly Disagree, n (%) |
|---|---|---|---|---|
| ||||
| Has improved the quality and safety of patient care | 46 (47.9) | 46 (47.9) | 2 (2.1) | 2 (2.1) |
| Has improved communication with patients and families | 42 (43.7) | 47 (49.0) | 5 (5.2) | 2 (2.1) |
| Has improved your efficiency/productivity | 31 (32.6) | 43 (45.3) | 17 (17.9) | 4 (4.2) |
| Has improved your engagement and job satisfaction | 33 (34.4) | 49 (51.0) | 10 (10.4) | 4 (4.2) |
| Is a better model of delivering patient care | 45 (47.4) | 44 (46.3) | 2 (2.1) | 4 (4.2) |
DISCUSSION
The serious problems in US healthcare constitute an urgent imperative to innovate and reform.[10] Inpatient care reflects 31% of the expenditure on healthcare, and in 2010, 35.1 million patients were discharged from the hospital after spending an average of 4.8 days as an inpatient.[11] These figures represent an immense opportunity to intervene. Measuring the impact of quality improvement efforts is often complicated by concomitant changes that affect outcomes over the interval studied. Our approach allowed us to detect statistically significant changes in LOS index and CMI‐adjusted VDC associated with the ACT implementation dose that could be separated from the underlying time trends.
The ACT model we describe is rooted in improving 3 foundational domains; quantifying each intervention's compartmentalized contribution, however, proved difficult. Each intervention intertwines with the others to create changes in attitudes, knowledge, and culture that are difficult to measure yet may synergistically affect outcomes. For example, although geographical cohorting appears to have the strongest statistical association with outcomes, this may be mediated by how it enables other processes to take place more effectively. Based on this analysis, therefore, the ACT model may best be considered a bundled intervention.
The team caring for a patient during hospitalization is so complex that fewer than a quarter of patients know their physician's or nurse's name.[12] This complexity impairs communication between patients and providers and between the providers themselves. Communication failures are consistently identified as root causes in sentinel events reported to the Joint Commission.[13] IPC is the process by which different professional groups work together to positively impact health care. IPC overlaps with communication, coordination, and teamwork, and improvements in IPC may improve care.[14] Some elements of the model we describe have been tested previously.[15, 16, 17] Localization of teams may increase productivity and the frequency with which physicians and nurses communicate. Localization also decreases the number of pages received and steps walked by providers during a workday.[15, 16, 17] However, these studies reported a trend toward an increase in the LOS and neutral effects on cost and readmission rates. We found statistically significant decreases in both LOS and cost associated with the geographic cohorting of patients and providers. Notably, our model localized not only the physician providers but also the interdisciplinary team of pharmacists, clinical nurse specialists, case managers, and social workers. This proximity may facilitate IPC between all members that culminates in improved efficiency. The possibility of delays in discharges to avoid new admissions in a geographically structured team has previously been raised to explain the associated increases in LOS.[16, 17] The accountability of each unit for its metrics, the communication between nursing and physicians, and the timely availability of the unit's performance data aligns everyone toward a shared goal and provides some protection from an unintended consequence.
Structured interdisciplinary rounds decrease adverse events and improve teamwork ratings.[18, 19] The huddle in our model is a forum to collaborate between disciplines that proved to be effective in decreasing LOS and costs. Our huddle aims to discuss all the patients on the unit. This allows the team to assist each other in problem solving for the entire unit and not just the patients on the geographically cohorted team. This approach, in addition to the improved IPC fostered by the ACT model, may help explain how benefits in LOS and costs permeated across all 11 diverse units despite the presence of patients who are not directly served by the geographically cohorted team.
High‐performing clinical systems maintain an awareness of their overarching mission and unit‐based leaders can influence the frontline by reiterating the organizational mission and aligning efforts with outcomes.[20] Our leadership model is similar to those described by other institutions in the strong partnerships between physicians and nursing.[21] As outlined by Kim et al., investing in the professional development of the unit leaders may help them fulfill their roles and serve the organization better.[21]
The fragmentation and lack of ownership over the continuum of patient care causes duplication and waste. The proposal in the Accountable Care Act to create accountable care organizations is rooted in the understanding that providers and organizations will seek out new ways of improving quality when held accountable for their outcomes.[22] To foster ownership and accountability, reporting of metrics at the unit level is needed. Furthermore, an informational infrastructure is critical, as improvements cannot occur without the availability of data to both monitor performance and measure the effect of interventions.[10, 23] Even without any other interventions, providing feedback alone is an effective way of changing practices.[24] According to Berwick et al., this phenomenon reflects practitioners' intrinsic motivation to simply want to be better.[25] Our monthly review of each unit's data is an effective way to provide timely feedback to the frontline that sparks pride, ownership, and innovative thinking.
Based on our mean ACT score and CMI‐adjusted VDC reductions alone, we estimate savings of $649.36 per hospitalization (mean increase in ACT implementation of 2.37 times reduction in cost index of $273.99 per unit increase in overall ACT score). This figure does not include savings realized through reductions in LOS. This is a small decrease relative to the mean cost of hospitalization, yet when compounded over the annual MH census, it would result in substantial savings. The model relied on the restructuring of the existing workforce and the only direct additional cost was the early salary support for the ACT program director.
Limitations
We recognize several limitations. It is a single center's experience and may not be generalizable. The diffusion of knowledge and culture carried between units and the relatively rapid implementation timeline did not allow for a control unit. A single observer assigned our implementation scores, and therefore we cannot report measures of inter‐rater reliability. However, defined criteria and direct observations were used wherever possible. Although administratively available data have their limitations, where available, we used measurements that are adjusted for severity of illness and CMI. We therefore feel that this dataset is an accurate representation of currently reported national quality indicators.
FURTHER DIRECTIONS
Although there is a need to improve our healthcare system, interventions should be deliberate and evidence based wherever possible.[26] Geographic cohorting may decrease the frequency of paging interruptions for physicians and practitioners while increasing face‐to‐face interruptions.[27] The net effect on safety with this trade‐off should be investigated.
The presence of an intervention does not guarantee its success. Despite geographic cohorting and interdisciplinary meetings, communication that influences physician decision making may not improve.[28] Although instruments to measure ratings of team work and collaboration are available, focusing on clinically relevant outcomes of teamwork, such as prevention of harm, may be more empowering feedback for the frontline. Formal cost‐benefit analyses and outcomes related to physician and nursing retention will be equally important for assessing the sustainability of the model. Involving patients and their caregivers and inviting their perspectives as care is redesigned will also be critical in maintaining patient centeredness. Research addressing interventions to mediate preventable readmission risk and understanding the drivers of patient satisfaction is also needed.
The true value of the model may be in its potential to monitor and drive change within itself. Continuously aligning aims, incentives, performance measures, and feedback will help support this innovation and drive. This affects not only patient care but creates microcosms within which research and education can thrive. We hope that our experience will help guide other institutions as we all strive in our journey to improve the care we deliver.
Acknowledgements
The authors thank the Indiana University Health Physicians hospitalists at MH, Sandy Janitz and Decision Support, the Indiana University Health executive leadership team, Robert Clark, Malaz Boustani, Dennis Watson, Nadia Adams, Todd Biggerstaff, Deanne Kashiwagi, and the tireless providers at MH for their support.
Disclosure: This work was supported by a grant from the Indiana University Health Values Fund. The authors have no conflicts of interest to disclose.
- Committee on Quality of Health Care in America; Institute of Medicine. Crossing the Quality Chasm: A New Health System for the 21st Century. Washington, DC: The National Academies Press; 2001.
- . Is US health really the best in the world? JAMA. 2000;284(4):483–485.
- , , , , , . Temporal trends in rates of patient harm resulting from medical care. N Engl J Med. 2010;363(22):2124–2134.
- Indiana University Health. Available at: http://iuhealth.org/methodist/aboIut/. Accessed October 20, 2014.
- University Health Consortium. Available at: https://www.uhc.edu/docs/45014769_QSS_dashboard_FAQs.pdf. Accessed October 23, 2014.
- , , , et al. Distribution of variable vs fixed costs of hospital care. JAMA. 1999;281(7):644–649.
- Centers for Medicare and Medicaid Services. Case mix index. Available at: http://www.cms.gov/Medicare/Medicare‐Fee‐for‐Service‐Payment/AcuteInpatientPPS/Acute‐Inpatient‐Files‐for‐Download‐Items/CMS022630.html. Accessed May 4, 2015.
- University Health Consortium. Available at: https://www.uhc.edu. Accessed October 23, 2014.
- Centers for Medicare and Medicaid Services. Hospital Consumer Assessment of Healthcare Providers and Systems. HCAHPS survey content and administration. Centers for Medicare 280(11):1000–1005.
- Centers for Disease Control and Prevention. FastStats. Available at: http://www.cdc.gov/nchs/fastats/default.htm. Accessed October 27, 2014.
- , . Does your patient know your name? An approach to enhancing patients' awareness of their caretaker's name. J Healthc Qual. 2005;27(4):53–56.
- The Joint Commission. Sentinel event data: root causes by event type 2004‐third quarter. Available at: http://www.jointcommissionorg. Available at: http://www.jointcommission.org/assets/1/18/Root_Causes_by_Event_Type_2004-2Q2013.pdf. Accessed March 26, 2014.
- , , . Interprofessional collaboration: effects of practice‐based interventions on professional practice and healthcare outcomes. Cochrane Database Syst Rev. 2009;(3):CD000072.
- , , , et al. Impact of localizing physicians to hospital units on nurse–physician communication and agreement on the plan of care. J Gen Intern Med. 2009;24(11):1223–1227.
- , , , et al. Impact of localizing general medical teams to a single nursing unit. J Hosp Med. 2012;7(7):551–556.
- , , , et al. Implementation of a physician assistant/hospitalist service in an academic medical center: impact on efficiency and patient outcomes. J Hosp Med. 2008;3(5):361–368.
- , , , , , . Improving teamwork: impact of structured interdisciplinary rounds on a medical teaching unit. J Gen Intern Med. 2010;25(8):826–832.
- , , , ; High Performance Teams and the Hospital of the Future Project Team. Interdisciplinary teamwork in hospitals: a review and practical recommendations for improvement. J Hosp Med. 2011;7(1):48–54.
- , , , , , . Microsystems in health care: part 8. Developing people and improving work life: what front‐line staff told us. Jt Comm J Qual Saf. 2003;29(10):512–522.
- , , , , , . Unit‐based interprofessional leadership models in six US hospitals. J Hosp Med. 2014;9(8):545–550.
- , , , . Creating accountable care organizations: the extended hospital medical staff. Health Aff (Millwood). 2007;26(1):w44–w57.
- , . Using performance measurement to drive improvement: a road map for change. Med Care. 2003;41(1 suppl):I48–I60.
- , . Changing physicians' practices. N Engl J Med. 1993;329(17):1271–1273.
- , , . Connections between quality measurement and improvement. Med Care. 2003;41(1 suppl):I30–I38.
- , , . The tension between needing to improve care and knowing how to do it. N Engl J Med. 2007;357(6):608–613.
- , . A qualitative evaluation of geographical localization of hospitalists: how unintended consequences may impact quality. J Gen Intern Med. 2014;29(7):1009–1016.
- , , , , . Disengaged: a qualitative study of communication and collaboration between physicians and other professions on general internal medicine wards. BMC Health Serv Res. 2013;13:494.
Despite an estimated annual $2.6 trillion expenditure on healthcare, the United States performs poorly on indicators of health and harm during care.[1, 2, 3] Hospitals around the nation are working to improve the care they deliver. We describe a model developed at our institution and report the evaluation of the outcomes associated with its implementation on the general medical and surgical units. The Indiana University Institutional Review Board approved this work.
SETTING AND DEFINITIONS
Indiana University Health Methodist Hospital (MH) is an academic center in Indianapolis, Indiana, serving over 30,000 patients annually.[4] In 2012, responding to the coexisting needs to improve quality and contain costs, the MH leadership team redesigned care in the hospital. The new model centers around accountable care teams (ACTs). Each ACT is a geographically defined set of providers accepting ownership for the clinical, service, and financial outcomes of their respective inpatient unit. The units studied are described in Table 1.
| Unit | No. of Beds | Predominant Diagnosis (Maximum Domain Score)* | |
|---|---|---|---|
| |||
| Medical units with progressive‐care beds | 1 | 33 | Pulmonary (3.4, 3.5, 5) |
| 2 | 28 | Cardiology (4.8, 3.5, 4) | |
| 3 | 24 | General medical (4.8, 3.5, 4) | |
| Medical units without progressive‐care beds | 4 | 36 | Renal/diabetic (4, 3.5, 5) |
| 5 | 24 | General medical (3.75, 4, 5) | |
| Surgical units with progressive‐care beds | 6 | 51 | Cardiothoracic surgery/cardiology (4, 4, 5) |
| 7 | 29 | Trauma/general surgery (3.75, 3.5, 5) | |
| 8 | 23 | Neurosurgical/neurological (4.8, 5, 5) | |
| 9 | 24 | Neurosurgical/neurological (4.4, 4.5, 5) | |
| Surgical units without progressive‐care beds | 10 | 29 | General/urologic/gynecologic/plastic surgery (3.4, 3, 2) |
| 11 | 26 | Orthopedic surgery (4.6, 4, 5) | |
THE ACT MODEL
The model comprises 8 interventions rooted in 3 foundational domains: (1) enhancing interprofessional collaboration (IPC), (2) enabling data‐driven decisions, and (3) providing leadership. Each intervention is briefly described under its main focus (see Supporting Information, Appendix A, in the online version of this article for further details).
Enhancing IPC
Geographical Cohorting of Patients and Providers
Hospitalist providers are localized for 4 consecutive months to 1 unit. An interdisciplinary team including a case manager, clinical nurse specialist, pharmacist, nutritionist, and social worker also serve each unit. Learners (residents, pharmacy, and medical students) are embedded in the team when rotating on the hospital medicine service. The presence of unit‐based nurse managers and charge nurses predates the model and is retained.
Bedside Collaborative Rounding
Geographically cohorted providers round on their patients with the bedside nurse guided by a customizable script.
Daily Huddle
The hospitalist, learners, and the interdisciplinary team for the unit meet each weekday to discuss patients' needs for a safe transition out of the hospital. Each unit determined the timing, location, and script for the huddle while retaining the focus on discharge planning (see Supporting Information, Appendix A2, in the online version of this article for a sample script).
Hospitalist and Specialty Comanagement Agreements
Guidelines delineating responsibilities for providers of each specialty were developed. Examples include orders pertaining to the management of a dialysis catheter in a patient with end‐stage renal disease, the removal of drains in postsurgical patients, and wound care.
Unit White Board
Each unit has a white board at the nursing station. Similar to the huddle, it is focused on discharge planning.
Enabling Data‐Driven Decisions
Monthly Review of Unit‐Level Data
Data analytics at our institution developed a data dashboard. Key metrics including length of stay (LOS), patient satisfaction scores, readmission rates, and costs are tracked and attributed to the discharging unit. The data are collated monthly by the ACT program director and distributed to each unit's leadership. Monthly interdisciplinary meetings are held to review trends. Learners are encouraged but not required to attend.
Weekly Patient Satisfaction Rounding
The unit's nurse manager and physician leader conduct weekly satisfaction rounds on patients. The conversation is open‐ended and focused on eliciting positive and negative experiences.
Providing Leadership
Designated hospitalist and, where relevant, specialty leaders are committed to serve each unit for at least 1 year as a resource for both medical and operational problem solving. The leader stays closely connected with the unit's nurse manager. In addition to day‐to‐day troubleshooting, the leader is responsible for monitoring outcome trends. There is currently no stipend, training, or other incentive offered for the role.
Implementation Timelines and ACT Scores
The development of the ACTs started in the spring of 2012. Physician, nursing, and pharmacy support was sought, and a pilot unit was formed in August 2012. The model was cascaded hospital wide by December 2013, with support from the ACT program director (A.N.). The program director observed and scored the uptake of each intervention by each unit monthly. A score of 1 denoted no implementation, whereas 5 denoted complete implementation. The criteria for scoring are presented in Table 2. The monthly scores for all 8 interventions in each of the 11 units were averaged as an overall ACT score, which reflects the implementation dose of the ACT model. Monthly domain scores for enhancing IPC and enabling data‐driven decisions were also calculated as the average score within each domain. This yielded 3 domain scores. Figure 1A plots by month the overall ACT score for the medical and surgical units, and Figure 1B plots the implementation score for the 3 domains between August 2012 and December 2013 for all units. The uptake of the interventions varied between units. This allowed our analysis to explore the dose relationships between the model and outcomes independent of underlying time trends that may be affected by concomitant initiatives.
| 1 | 2 | 3 | 4 | 5 | |
|---|---|---|---|---|---|
| |||||
| Geographical cohorting of patients and the ACT* | None | At least 1 discipline comprising the ACT is unit based | All disciplines comprising the ACT except the hospitalist unit based | All disciplines including the hospitalist unit based | 4 + 80% of hospitalist provider's patients on the unit |
| Bedside collaborative rounding | None | Occurring 1 day a week on at least 25% of the patients on the unit | Occurring 2 to 3 days a week on at least 50% of the patients on the unit | Occurring 3 to 4 days a week on at least 75% of the patients on the unit | Occurring MondayFriday on all patients on the unit |
| Daily huddle | None | Occurring daily, 1 out of 4 ACT disciplines represented, at least 25% of patients on the unit discussed | Occurring daily, 2 out of 4 ACT disciplines represented, at least 50% of patients on the unit discussed | Occurring daily, 3 out of 4 ACT disciplines represented, at least 75% of patients on the unit discussed | Occurring daily, all disciplines of the ACT represented, all patients on the unit discussed |
| Hospitalist and specialty comanagement agreements | None | One out of 3 specialists represented on the unit collaborating with the hospitalists on at least 25% of relevant patients | One out of 3 specialists represented on the unit collaborating with the hospitalists on at least 50% of relevant patients | Two out of 3 specialists on the unit collaborating with the hospitalists on at least 75% of relevant patients | All specialists on the unit collaborating with the hospitalists on all relevant patients on the unit |
| Unit white board | None | Present but only used by nursing | Present and used by all ACT disciplines except physician providers | Present and used by entire ACT; use inconsistent | Present and used MondayFriday by all disciplines of ACT |
| Monthly review of unit level data | None | Nurse manager reviewing data with ACT program director | Nurse manager and unit leader reviewing data with ACT program director | Meeting either not consistently occurring monthly or not consistently attended by entire ACT | Monthly meeting with entire ACT |
| Weekly patient satisfaction rounding | None | Nurse manager performing up to 1 week a month | Nurse manager performing weekly | Nurse and physician leader performing up to 3 times a month | Nurse and physician leader performing weekly |
| Leadership | None | For units with specialties, either hospitalist or specialist leader identified | Both hospitalist and specialist leader Identified | Both hospitalist and specialist leaders (where applicable) identified and partially engaged in leadership role | Both hospitalist and specialist leaders (where applicable) identified and engaged in leadership role |

Outcomes
Monthly data between August 2012 and December 2013 were analyzed.
Measures of Value
MH is a member of the University Health Consortium, which measures outcomes of participants relative to their peers. MH measures LOS index as a ratio of observed LOS to expected LOS that is adjusted for severity of illness.[5]
Variable direct costs (VDCs) are costs that a hospital can save if a service is not provided.[6] A hospital's case‐mix index (CMI) represents the average diagnosis‐related group relative weight for that hospital. We track VDCs adjusted for CMI (CMI‐adjusted VDC).[7]
Thirty‐day readmission rate is the percentage of cases that are readmitted to MH within 30 days of discharge from the index admission.[8]
Measures of Patient Satisfaction
The Hospital Consumer Assessment of Healthcare Providers and Systems (HCAHPS) survey covers topics relevant to a patient's experience in the hospital.[9] Patient satisfaction scores are tracked by responses to the HCAHPS survey.
Measures of Provider Satisfaction
Hospitalist and specialty providers, leadership, and case management teams were surveyed via email through SurveyMonkey in July 2014. The survey included Likert responses that elicited opinions and comments about the ACT model.
Statistical Methods
The primary predictor of interest was the monthly overall ACT score. We also explored the domain scores as well as the individual scores for each intervention. Generalized linear mixed models were fit to investigate the association between each predictor (overall ACT score, ACT domain scores, and individual implementation scores) and each outcome (LOS index, CMI‐adjusted VDC, 30‐day readmission rate, and overall patient satisfaction). The model for testing each ACT score also included covariates of inpatient units as a random effect, as well as date and type of unit as fixed effects. We set the statistical significance level at 0.01 and reported 99% confidence intervals.
Descriptive statistics were used to report the provider satisfaction survey results.
RESULTS
The overall ACT score was associated with LOS index and CMI‐adjusted VDC (both P < 0.001). For every 1‐unit increase in the overall ACT score, LOS index decreased by 0.078 and CMI‐adjusted VDC decreased by $273.99 (Table 3).
| Length of Stay Index | CMI Adjusted VDC | |||
|---|---|---|---|---|
| Estimate (99% CI)* | P Value | Estimate (99% CI)* | P Value | |
| ||||
| Overall ACT Score | 0.078 (0.123 to 0.032) | <0.001 | 274.0 (477.31 to 70.68) | <0.001 |
| Enhancing IPC | 0.071 (0.117 to 0.026) | <0.001 | 284.7 (488.08 to 81.23) | <0.001 |
| Enabling data‐driven decisions | 0.044 (0.080 to 0.009) | 0.002 | 145.4 (304.57 to 13.81) | 0.02 |
| Providing leadership | 0.027 (0.049 to 0.005) | 0.001 | 69.9 (169.00 to 29.26) | 0.07 |
Looking at domains, enhancing IPC resulted in statistically significant decreases in both LOS index and CMI‐adjusted VDC, but providing leadership and enabling data‐driven decisions decreased only the LOS index. Most of the 8 individual interventions were associated with at least 1 of these 2 outcomes. (Even where the associations were not significant, they were all in the direction of decreasing LOS and cost). In these models, the covariate of type of units (medical vs surgical) was not associated with LOS or cost. There was no significant time trend in LOS or cost, except in models where an intervention had no association with either outcome. Inclusion of all individual effective interventions in the same statistical model to assess their relative contributions was not possible because they were highly correlated (correlations 0.450.89).
Thirty‐day readmissions and patient satisfaction were not significantly associated with the overall ACT score, but exploratory analyses showed that patient satisfaction increased with the implementation of geographical cohorting (P = 0.007).
Survey Results
The response rate was 87% (96/110). Between 85% and 96% of respondents either agreed or strongly agreed that the ACT model had improved the quality and safety of the care delivered, improved communication between providers and patients, and improved their own engagement and job satisfaction. Overall, 78% of the respondents either agreed or strongly agreed that the model improved efficiency (Table 4). Suggestions for improvements revolved around increasing the emphasis on patient centeredness and bedside nursing engagement.
| The ACT Model | Strongly Agree, n (%) | Agree, n (%) | Disagree, n (%) | Strongly Disagree, n (%) |
|---|---|---|---|---|
| ||||
| Has improved the quality and safety of patient care | 46 (47.9) | 46 (47.9) | 2 (2.1) | 2 (2.1) |
| Has improved communication with patients and families | 42 (43.7) | 47 (49.0) | 5 (5.2) | 2 (2.1) |
| Has improved your efficiency/productivity | 31 (32.6) | 43 (45.3) | 17 (17.9) | 4 (4.2) |
| Has improved your engagement and job satisfaction | 33 (34.4) | 49 (51.0) | 10 (10.4) | 4 (4.2) |
| Is a better model of delivering patient care | 45 (47.4) | 44 (46.3) | 2 (2.1) | 4 (4.2) |
DISCUSSION
The serious problems in US healthcare constitute an urgent imperative to innovate and reform.[10] Inpatient care reflects 31% of the expenditure on healthcare, and in 2010, 35.1 million patients were discharged from the hospital after spending an average of 4.8 days as an inpatient.[11] These figures represent an immense opportunity to intervene. Measuring the impact of quality improvement efforts is often complicated by concomitant changes that affect outcomes over the interval studied. Our approach allowed us to detect statistically significant changes in LOS index and CMI‐adjusted VDC associated with the ACT implementation dose that could be separated from the underlying time trends.
The ACT model we describe is rooted in improving 3 foundational domains; quantifying each intervention's compartmentalized contribution, however, proved difficult. Each intervention intertwines with the others to create changes in attitudes, knowledge, and culture that are difficult to measure yet may synergistically affect outcomes. For example, although geographical cohorting appears to have the strongest statistical association with outcomes, this may be mediated by how it enables other processes to take place more effectively. Based on this analysis, therefore, the ACT model may best be considered a bundled intervention.
The team caring for a patient during hospitalization is so complex that fewer than a quarter of patients know their physician's or nurse's name.[12] This complexity impairs communication between patients and providers and between the providers themselves. Communication failures are consistently identified as root causes in sentinel events reported to the Joint Commission.[13] IPC is the process by which different professional groups work together to positively impact health care. IPC overlaps with communication, coordination, and teamwork, and improvements in IPC may improve care.[14] Some elements of the model we describe have been tested previously.[15, 16, 17] Localization of teams may increase productivity and the frequency with which physicians and nurses communicate. Localization also decreases the number of pages received and steps walked by providers during a workday.[15, 16, 17] However, these studies reported a trend toward an increase in the LOS and neutral effects on cost and readmission rates. We found statistically significant decreases in both LOS and cost associated with the geographic cohorting of patients and providers. Notably, our model localized not only the physician providers but also the interdisciplinary team of pharmacists, clinical nurse specialists, case managers, and social workers. This proximity may facilitate IPC between all members that culminates in improved efficiency. The possibility of delays in discharges to avoid new admissions in a geographically structured team has previously been raised to explain the associated increases in LOS.[16, 17] The accountability of each unit for its metrics, the communication between nursing and physicians, and the timely availability of the unit's performance data aligns everyone toward a shared goal and provides some protection from an unintended consequence.
Structured interdisciplinary rounds decrease adverse events and improve teamwork ratings.[18, 19] The huddle in our model is a forum to collaborate between disciplines that proved to be effective in decreasing LOS and costs. Our huddle aims to discuss all the patients on the unit. This allows the team to assist each other in problem solving for the entire unit and not just the patients on the geographically cohorted team. This approach, in addition to the improved IPC fostered by the ACT model, may help explain how benefits in LOS and costs permeated across all 11 diverse units despite the presence of patients who are not directly served by the geographically cohorted team.
High‐performing clinical systems maintain an awareness of their overarching mission and unit‐based leaders can influence the frontline by reiterating the organizational mission and aligning efforts with outcomes.[20] Our leadership model is similar to those described by other institutions in the strong partnerships between physicians and nursing.[21] As outlined by Kim et al., investing in the professional development of the unit leaders may help them fulfill their roles and serve the organization better.[21]
The fragmentation and lack of ownership over the continuum of patient care causes duplication and waste. The proposal in the Accountable Care Act to create accountable care organizations is rooted in the understanding that providers and organizations will seek out new ways of improving quality when held accountable for their outcomes.[22] To foster ownership and accountability, reporting of metrics at the unit level is needed. Furthermore, an informational infrastructure is critical, as improvements cannot occur without the availability of data to both monitor performance and measure the effect of interventions.[10, 23] Even without any other interventions, providing feedback alone is an effective way of changing practices.[24] According to Berwick et al., this phenomenon reflects practitioners' intrinsic motivation to simply want to be better.[25] Our monthly review of each unit's data is an effective way to provide timely feedback to the frontline that sparks pride, ownership, and innovative thinking.
Based on our mean ACT score and CMI‐adjusted VDC reductions alone, we estimate savings of $649.36 per hospitalization (mean increase in ACT implementation of 2.37 times reduction in cost index of $273.99 per unit increase in overall ACT score). This figure does not include savings realized through reductions in LOS. This is a small decrease relative to the mean cost of hospitalization, yet when compounded over the annual MH census, it would result in substantial savings. The model relied on the restructuring of the existing workforce and the only direct additional cost was the early salary support for the ACT program director.
Limitations
We recognize several limitations. It is a single center's experience and may not be generalizable. The diffusion of knowledge and culture carried between units and the relatively rapid implementation timeline did not allow for a control unit. A single observer assigned our implementation scores, and therefore we cannot report measures of inter‐rater reliability. However, defined criteria and direct observations were used wherever possible. Although administratively available data have their limitations, where available, we used measurements that are adjusted for severity of illness and CMI. We therefore feel that this dataset is an accurate representation of currently reported national quality indicators.
FURTHER DIRECTIONS
Although there is a need to improve our healthcare system, interventions should be deliberate and evidence based wherever possible.[26] Geographic cohorting may decrease the frequency of paging interruptions for physicians and practitioners while increasing face‐to‐face interruptions.[27] The net effect on safety with this trade‐off should be investigated.
The presence of an intervention does not guarantee its success. Despite geographic cohorting and interdisciplinary meetings, communication that influences physician decision making may not improve.[28] Although instruments to measure ratings of team work and collaboration are available, focusing on clinically relevant outcomes of teamwork, such as prevention of harm, may be more empowering feedback for the frontline. Formal cost‐benefit analyses and outcomes related to physician and nursing retention will be equally important for assessing the sustainability of the model. Involving patients and their caregivers and inviting their perspectives as care is redesigned will also be critical in maintaining patient centeredness. Research addressing interventions to mediate preventable readmission risk and understanding the drivers of patient satisfaction is also needed.
The true value of the model may be in its potential to monitor and drive change within itself. Continuously aligning aims, incentives, performance measures, and feedback will help support this innovation and drive. This affects not only patient care but creates microcosms within which research and education can thrive. We hope that our experience will help guide other institutions as we all strive in our journey to improve the care we deliver.
Acknowledgements
The authors thank the Indiana University Health Physicians hospitalists at MH, Sandy Janitz and Decision Support, the Indiana University Health executive leadership team, Robert Clark, Malaz Boustani, Dennis Watson, Nadia Adams, Todd Biggerstaff, Deanne Kashiwagi, and the tireless providers at MH for their support.
Disclosure: This work was supported by a grant from the Indiana University Health Values Fund. The authors have no conflicts of interest to disclose.
Despite an estimated annual $2.6 trillion expenditure on healthcare, the United States performs poorly on indicators of health and harm during care.[1, 2, 3] Hospitals around the nation are working to improve the care they deliver. We describe a model developed at our institution and report the evaluation of the outcomes associated with its implementation on the general medical and surgical units. The Indiana University Institutional Review Board approved this work.
SETTING AND DEFINITIONS
Indiana University Health Methodist Hospital (MH) is an academic center in Indianapolis, Indiana, serving over 30,000 patients annually.[4] In 2012, responding to the coexisting needs to improve quality and contain costs, the MH leadership team redesigned care in the hospital. The new model centers around accountable care teams (ACTs). Each ACT is a geographically defined set of providers accepting ownership for the clinical, service, and financial outcomes of their respective inpatient unit. The units studied are described in Table 1.
| Unit | No. of Beds | Predominant Diagnosis (Maximum Domain Score)* | |
|---|---|---|---|
| |||
| Medical units with progressive‐care beds | 1 | 33 | Pulmonary (3.4, 3.5, 5) |
| 2 | 28 | Cardiology (4.8, 3.5, 4) | |
| 3 | 24 | General medical (4.8, 3.5, 4) | |
| Medical units without progressive‐care beds | 4 | 36 | Renal/diabetic (4, 3.5, 5) |
| 5 | 24 | General medical (3.75, 4, 5) | |
| Surgical units with progressive‐care beds | 6 | 51 | Cardiothoracic surgery/cardiology (4, 4, 5) |
| 7 | 29 | Trauma/general surgery (3.75, 3.5, 5) | |
| 8 | 23 | Neurosurgical/neurological (4.8, 5, 5) | |
| 9 | 24 | Neurosurgical/neurological (4.4, 4.5, 5) | |
| Surgical units without progressive‐care beds | 10 | 29 | General/urologic/gynecologic/plastic surgery (3.4, 3, 2) |
| 11 | 26 | Orthopedic surgery (4.6, 4, 5) | |
THE ACT MODEL
The model comprises 8 interventions rooted in 3 foundational domains: (1) enhancing interprofessional collaboration (IPC), (2) enabling data‐driven decisions, and (3) providing leadership. Each intervention is briefly described under its main focus (see Supporting Information, Appendix A, in the online version of this article for further details).
Enhancing IPC
Geographical Cohorting of Patients and Providers
Hospitalist providers are localized for 4 consecutive months to 1 unit. An interdisciplinary team including a case manager, clinical nurse specialist, pharmacist, nutritionist, and social worker also serve each unit. Learners (residents, pharmacy, and medical students) are embedded in the team when rotating on the hospital medicine service. The presence of unit‐based nurse managers and charge nurses predates the model and is retained.
Bedside Collaborative Rounding
Geographically cohorted providers round on their patients with the bedside nurse guided by a customizable script.
Daily Huddle
The hospitalist, learners, and the interdisciplinary team for the unit meet each weekday to discuss patients' needs for a safe transition out of the hospital. Each unit determined the timing, location, and script for the huddle while retaining the focus on discharge planning (see Supporting Information, Appendix A2, in the online version of this article for a sample script).
Hospitalist and Specialty Comanagement Agreements
Guidelines delineating responsibilities for providers of each specialty were developed. Examples include orders pertaining to the management of a dialysis catheter in a patient with end‐stage renal disease, the removal of drains in postsurgical patients, and wound care.
Unit White Board
Each unit has a white board at the nursing station. Similar to the huddle, it is focused on discharge planning.
Enabling Data‐Driven Decisions
Monthly Review of Unit‐Level Data
Data analytics at our institution developed a data dashboard. Key metrics including length of stay (LOS), patient satisfaction scores, readmission rates, and costs are tracked and attributed to the discharging unit. The data are collated monthly by the ACT program director and distributed to each unit's leadership. Monthly interdisciplinary meetings are held to review trends. Learners are encouraged but not required to attend.
Weekly Patient Satisfaction Rounding
The unit's nurse manager and physician leader conduct weekly satisfaction rounds on patients. The conversation is open‐ended and focused on eliciting positive and negative experiences.
Providing Leadership
Designated hospitalist and, where relevant, specialty leaders are committed to serve each unit for at least 1 year as a resource for both medical and operational problem solving. The leader stays closely connected with the unit's nurse manager. In addition to day‐to‐day troubleshooting, the leader is responsible for monitoring outcome trends. There is currently no stipend, training, or other incentive offered for the role.
Implementation Timelines and ACT Scores
The development of the ACTs started in the spring of 2012. Physician, nursing, and pharmacy support was sought, and a pilot unit was formed in August 2012. The model was cascaded hospital wide by December 2013, with support from the ACT program director (A.N.). The program director observed and scored the uptake of each intervention by each unit monthly. A score of 1 denoted no implementation, whereas 5 denoted complete implementation. The criteria for scoring are presented in Table 2. The monthly scores for all 8 interventions in each of the 11 units were averaged as an overall ACT score, which reflects the implementation dose of the ACT model. Monthly domain scores for enhancing IPC and enabling data‐driven decisions were also calculated as the average score within each domain. This yielded 3 domain scores. Figure 1A plots by month the overall ACT score for the medical and surgical units, and Figure 1B plots the implementation score for the 3 domains between August 2012 and December 2013 for all units. The uptake of the interventions varied between units. This allowed our analysis to explore the dose relationships between the model and outcomes independent of underlying time trends that may be affected by concomitant initiatives.
| 1 | 2 | 3 | 4 | 5 | |
|---|---|---|---|---|---|
| |||||
| Geographical cohorting of patients and the ACT* | None | At least 1 discipline comprising the ACT is unit based | All disciplines comprising the ACT except the hospitalist unit based | All disciplines including the hospitalist unit based | 4 + 80% of hospitalist provider's patients on the unit |
| Bedside collaborative rounding | None | Occurring 1 day a week on at least 25% of the patients on the unit | Occurring 2 to 3 days a week on at least 50% of the patients on the unit | Occurring 3 to 4 days a week on at least 75% of the patients on the unit | Occurring MondayFriday on all patients on the unit |
| Daily huddle | None | Occurring daily, 1 out of 4 ACT disciplines represented, at least 25% of patients on the unit discussed | Occurring daily, 2 out of 4 ACT disciplines represented, at least 50% of patients on the unit discussed | Occurring daily, 3 out of 4 ACT disciplines represented, at least 75% of patients on the unit discussed | Occurring daily, all disciplines of the ACT represented, all patients on the unit discussed |
| Hospitalist and specialty comanagement agreements | None | One out of 3 specialists represented on the unit collaborating with the hospitalists on at least 25% of relevant patients | One out of 3 specialists represented on the unit collaborating with the hospitalists on at least 50% of relevant patients | Two out of 3 specialists on the unit collaborating with the hospitalists on at least 75% of relevant patients | All specialists on the unit collaborating with the hospitalists on all relevant patients on the unit |
| Unit white board | None | Present but only used by nursing | Present and used by all ACT disciplines except physician providers | Present and used by entire ACT; use inconsistent | Present and used MondayFriday by all disciplines of ACT |
| Monthly review of unit level data | None | Nurse manager reviewing data with ACT program director | Nurse manager and unit leader reviewing data with ACT program director | Meeting either not consistently occurring monthly or not consistently attended by entire ACT | Monthly meeting with entire ACT |
| Weekly patient satisfaction rounding | None | Nurse manager performing up to 1 week a month | Nurse manager performing weekly | Nurse and physician leader performing up to 3 times a month | Nurse and physician leader performing weekly |
| Leadership | None | For units with specialties, either hospitalist or specialist leader identified | Both hospitalist and specialist leader Identified | Both hospitalist and specialist leaders (where applicable) identified and partially engaged in leadership role | Both hospitalist and specialist leaders (where applicable) identified and engaged in leadership role |

Outcomes
Monthly data between August 2012 and December 2013 were analyzed.
Measures of Value
MH is a member of the University Health Consortium, which measures outcomes of participants relative to their peers. MH measures LOS index as a ratio of observed LOS to expected LOS that is adjusted for severity of illness.[5]
Variable direct costs (VDCs) are costs that a hospital can save if a service is not provided.[6] A hospital's case‐mix index (CMI) represents the average diagnosis‐related group relative weight for that hospital. We track VDCs adjusted for CMI (CMI‐adjusted VDC).[7]
Thirty‐day readmission rate is the percentage of cases that are readmitted to MH within 30 days of discharge from the index admission.[8]
Measures of Patient Satisfaction
The Hospital Consumer Assessment of Healthcare Providers and Systems (HCAHPS) survey covers topics relevant to a patient's experience in the hospital.[9] Patient satisfaction scores are tracked by responses to the HCAHPS survey.
Measures of Provider Satisfaction
Hospitalist and specialty providers, leadership, and case management teams were surveyed via email through SurveyMonkey in July 2014. The survey included Likert responses that elicited opinions and comments about the ACT model.
Statistical Methods
The primary predictor of interest was the monthly overall ACT score. We also explored the domain scores as well as the individual scores for each intervention. Generalized linear mixed models were fit to investigate the association between each predictor (overall ACT score, ACT domain scores, and individual implementation scores) and each outcome (LOS index, CMI‐adjusted VDC, 30‐day readmission rate, and overall patient satisfaction). The model for testing each ACT score also included covariates of inpatient units as a random effect, as well as date and type of unit as fixed effects. We set the statistical significance level at 0.01 and reported 99% confidence intervals.
Descriptive statistics were used to report the provider satisfaction survey results.
RESULTS
The overall ACT score was associated with LOS index and CMI‐adjusted VDC (both P < 0.001). For every 1‐unit increase in the overall ACT score, LOS index decreased by 0.078 and CMI‐adjusted VDC decreased by $273.99 (Table 3).
| Length of Stay Index | CMI Adjusted VDC | |||
|---|---|---|---|---|
| Estimate (99% CI)* | P Value | Estimate (99% CI)* | P Value | |
| ||||
| Overall ACT Score | 0.078 (0.123 to 0.032) | <0.001 | 274.0 (477.31 to 70.68) | <0.001 |
| Enhancing IPC | 0.071 (0.117 to 0.026) | <0.001 | 284.7 (488.08 to 81.23) | <0.001 |
| Enabling data‐driven decisions | 0.044 (0.080 to 0.009) | 0.002 | 145.4 (304.57 to 13.81) | 0.02 |
| Providing leadership | 0.027 (0.049 to 0.005) | 0.001 | 69.9 (169.00 to 29.26) | 0.07 |
Looking at domains, enhancing IPC resulted in statistically significant decreases in both LOS index and CMI‐adjusted VDC, but providing leadership and enabling data‐driven decisions decreased only the LOS index. Most of the 8 individual interventions were associated with at least 1 of these 2 outcomes. (Even where the associations were not significant, they were all in the direction of decreasing LOS and cost). In these models, the covariate of type of units (medical vs surgical) was not associated with LOS or cost. There was no significant time trend in LOS or cost, except in models where an intervention had no association with either outcome. Inclusion of all individual effective interventions in the same statistical model to assess their relative contributions was not possible because they were highly correlated (correlations 0.450.89).
Thirty‐day readmissions and patient satisfaction were not significantly associated with the overall ACT score, but exploratory analyses showed that patient satisfaction increased with the implementation of geographical cohorting (P = 0.007).
Survey Results
The response rate was 87% (96/110). Between 85% and 96% of respondents either agreed or strongly agreed that the ACT model had improved the quality and safety of the care delivered, improved communication between providers and patients, and improved their own engagement and job satisfaction. Overall, 78% of the respondents either agreed or strongly agreed that the model improved efficiency (Table 4). Suggestions for improvements revolved around increasing the emphasis on patient centeredness and bedside nursing engagement.
| The ACT Model | Strongly Agree, n (%) | Agree, n (%) | Disagree, n (%) | Strongly Disagree, n (%) |
|---|---|---|---|---|
| ||||
| Has improved the quality and safety of patient care | 46 (47.9) | 46 (47.9) | 2 (2.1) | 2 (2.1) |
| Has improved communication with patients and families | 42 (43.7) | 47 (49.0) | 5 (5.2) | 2 (2.1) |
| Has improved your efficiency/productivity | 31 (32.6) | 43 (45.3) | 17 (17.9) | 4 (4.2) |
| Has improved your engagement and job satisfaction | 33 (34.4) | 49 (51.0) | 10 (10.4) | 4 (4.2) |
| Is a better model of delivering patient care | 45 (47.4) | 44 (46.3) | 2 (2.1) | 4 (4.2) |
DISCUSSION
The serious problems in US healthcare constitute an urgent imperative to innovate and reform.[10] Inpatient care reflects 31% of the expenditure on healthcare, and in 2010, 35.1 million patients were discharged from the hospital after spending an average of 4.8 days as an inpatient.[11] These figures represent an immense opportunity to intervene. Measuring the impact of quality improvement efforts is often complicated by concomitant changes that affect outcomes over the interval studied. Our approach allowed us to detect statistically significant changes in LOS index and CMI‐adjusted VDC associated with the ACT implementation dose that could be separated from the underlying time trends.
The ACT model we describe is rooted in improving 3 foundational domains; quantifying each intervention's compartmentalized contribution, however, proved difficult. Each intervention intertwines with the others to create changes in attitudes, knowledge, and culture that are difficult to measure yet may synergistically affect outcomes. For example, although geographical cohorting appears to have the strongest statistical association with outcomes, this may be mediated by how it enables other processes to take place more effectively. Based on this analysis, therefore, the ACT model may best be considered a bundled intervention.
The team caring for a patient during hospitalization is so complex that fewer than a quarter of patients know their physician's or nurse's name.[12] This complexity impairs communication between patients and providers and between the providers themselves. Communication failures are consistently identified as root causes in sentinel events reported to the Joint Commission.[13] IPC is the process by which different professional groups work together to positively impact health care. IPC overlaps with communication, coordination, and teamwork, and improvements in IPC may improve care.[14] Some elements of the model we describe have been tested previously.[15, 16, 17] Localization of teams may increase productivity and the frequency with which physicians and nurses communicate. Localization also decreases the number of pages received and steps walked by providers during a workday.[15, 16, 17] However, these studies reported a trend toward an increase in the LOS and neutral effects on cost and readmission rates. We found statistically significant decreases in both LOS and cost associated with the geographic cohorting of patients and providers. Notably, our model localized not only the physician providers but also the interdisciplinary team of pharmacists, clinical nurse specialists, case managers, and social workers. This proximity may facilitate IPC between all members that culminates in improved efficiency. The possibility of delays in discharges to avoid new admissions in a geographically structured team has previously been raised to explain the associated increases in LOS.[16, 17] The accountability of each unit for its metrics, the communication between nursing and physicians, and the timely availability of the unit's performance data aligns everyone toward a shared goal and provides some protection from an unintended consequence.
Structured interdisciplinary rounds decrease adverse events and improve teamwork ratings.[18, 19] The huddle in our model is a forum to collaborate between disciplines that proved to be effective in decreasing LOS and costs. Our huddle aims to discuss all the patients on the unit. This allows the team to assist each other in problem solving for the entire unit and not just the patients on the geographically cohorted team. This approach, in addition to the improved IPC fostered by the ACT model, may help explain how benefits in LOS and costs permeated across all 11 diverse units despite the presence of patients who are not directly served by the geographically cohorted team.
High‐performing clinical systems maintain an awareness of their overarching mission and unit‐based leaders can influence the frontline by reiterating the organizational mission and aligning efforts with outcomes.[20] Our leadership model is similar to those described by other institutions in the strong partnerships between physicians and nursing.[21] As outlined by Kim et al., investing in the professional development of the unit leaders may help them fulfill their roles and serve the organization better.[21]
The fragmentation and lack of ownership over the continuum of patient care causes duplication and waste. The proposal in the Accountable Care Act to create accountable care organizations is rooted in the understanding that providers and organizations will seek out new ways of improving quality when held accountable for their outcomes.[22] To foster ownership and accountability, reporting of metrics at the unit level is needed. Furthermore, an informational infrastructure is critical, as improvements cannot occur without the availability of data to both monitor performance and measure the effect of interventions.[10, 23] Even without any other interventions, providing feedback alone is an effective way of changing practices.[24] According to Berwick et al., this phenomenon reflects practitioners' intrinsic motivation to simply want to be better.[25] Our monthly review of each unit's data is an effective way to provide timely feedback to the frontline that sparks pride, ownership, and innovative thinking.
Based on our mean ACT score and CMI‐adjusted VDC reductions alone, we estimate savings of $649.36 per hospitalization (mean increase in ACT implementation of 2.37 times reduction in cost index of $273.99 per unit increase in overall ACT score). This figure does not include savings realized through reductions in LOS. This is a small decrease relative to the mean cost of hospitalization, yet when compounded over the annual MH census, it would result in substantial savings. The model relied on the restructuring of the existing workforce and the only direct additional cost was the early salary support for the ACT program director.
Limitations
We recognize several limitations. It is a single center's experience and may not be generalizable. The diffusion of knowledge and culture carried between units and the relatively rapid implementation timeline did not allow for a control unit. A single observer assigned our implementation scores, and therefore we cannot report measures of inter‐rater reliability. However, defined criteria and direct observations were used wherever possible. Although administratively available data have their limitations, where available, we used measurements that are adjusted for severity of illness and CMI. We therefore feel that this dataset is an accurate representation of currently reported national quality indicators.
FURTHER DIRECTIONS
Although there is a need to improve our healthcare system, interventions should be deliberate and evidence based wherever possible.[26] Geographic cohorting may decrease the frequency of paging interruptions for physicians and practitioners while increasing face‐to‐face interruptions.[27] The net effect on safety with this trade‐off should be investigated.
The presence of an intervention does not guarantee its success. Despite geographic cohorting and interdisciplinary meetings, communication that influences physician decision making may not improve.[28] Although instruments to measure ratings of team work and collaboration are available, focusing on clinically relevant outcomes of teamwork, such as prevention of harm, may be more empowering feedback for the frontline. Formal cost‐benefit analyses and outcomes related to physician and nursing retention will be equally important for assessing the sustainability of the model. Involving patients and their caregivers and inviting their perspectives as care is redesigned will also be critical in maintaining patient centeredness. Research addressing interventions to mediate preventable readmission risk and understanding the drivers of patient satisfaction is also needed.
The true value of the model may be in its potential to monitor and drive change within itself. Continuously aligning aims, incentives, performance measures, and feedback will help support this innovation and drive. This affects not only patient care but creates microcosms within which research and education can thrive. We hope that our experience will help guide other institutions as we all strive in our journey to improve the care we deliver.
Acknowledgements
The authors thank the Indiana University Health Physicians hospitalists at MH, Sandy Janitz and Decision Support, the Indiana University Health executive leadership team, Robert Clark, Malaz Boustani, Dennis Watson, Nadia Adams, Todd Biggerstaff, Deanne Kashiwagi, and the tireless providers at MH for their support.
Disclosure: This work was supported by a grant from the Indiana University Health Values Fund. The authors have no conflicts of interest to disclose.
- Committee on Quality of Health Care in America; Institute of Medicine. Crossing the Quality Chasm: A New Health System for the 21st Century. Washington, DC: The National Academies Press; 2001.
- . Is US health really the best in the world? JAMA. 2000;284(4):483–485.
- , , , , , . Temporal trends in rates of patient harm resulting from medical care. N Engl J Med. 2010;363(22):2124–2134.
- Indiana University Health. Available at: http://iuhealth.org/methodist/aboIut/. Accessed October 20, 2014.
- University Health Consortium. Available at: https://www.uhc.edu/docs/45014769_QSS_dashboard_FAQs.pdf. Accessed October 23, 2014.
- , , , et al. Distribution of variable vs fixed costs of hospital care. JAMA. 1999;281(7):644–649.
- Centers for Medicare and Medicaid Services. Case mix index. Available at: http://www.cms.gov/Medicare/Medicare‐Fee‐for‐Service‐Payment/AcuteInpatientPPS/Acute‐Inpatient‐Files‐for‐Download‐Items/CMS022630.html. Accessed May 4, 2015.
- University Health Consortium. Available at: https://www.uhc.edu. Accessed October 23, 2014.
- Centers for Medicare and Medicaid Services. Hospital Consumer Assessment of Healthcare Providers and Systems. HCAHPS survey content and administration. Centers for Medicare 280(11):1000–1005.
- Centers for Disease Control and Prevention. FastStats. Available at: http://www.cdc.gov/nchs/fastats/default.htm. Accessed October 27, 2014.
- , . Does your patient know your name? An approach to enhancing patients' awareness of their caretaker's name. J Healthc Qual. 2005;27(4):53–56.
- The Joint Commission. Sentinel event data: root causes by event type 2004‐third quarter. Available at: http://www.jointcommissionorg. Available at: http://www.jointcommission.org/assets/1/18/Root_Causes_by_Event_Type_2004-2Q2013.pdf. Accessed March 26, 2014.
- , , . Interprofessional collaboration: effects of practice‐based interventions on professional practice and healthcare outcomes. Cochrane Database Syst Rev. 2009;(3):CD000072.
- , , , et al. Impact of localizing physicians to hospital units on nurse–physician communication and agreement on the plan of care. J Gen Intern Med. 2009;24(11):1223–1227.
- , , , et al. Impact of localizing general medical teams to a single nursing unit. J Hosp Med. 2012;7(7):551–556.
- , , , et al. Implementation of a physician assistant/hospitalist service in an academic medical center: impact on efficiency and patient outcomes. J Hosp Med. 2008;3(5):361–368.
- , , , , , . Improving teamwork: impact of structured interdisciplinary rounds on a medical teaching unit. J Gen Intern Med. 2010;25(8):826–832.
- , , , ; High Performance Teams and the Hospital of the Future Project Team. Interdisciplinary teamwork in hospitals: a review and practical recommendations for improvement. J Hosp Med. 2011;7(1):48–54.
- , , , , , . Microsystems in health care: part 8. Developing people and improving work life: what front‐line staff told us. Jt Comm J Qual Saf. 2003;29(10):512–522.
- , , , , , . Unit‐based interprofessional leadership models in six US hospitals. J Hosp Med. 2014;9(8):545–550.
- , , , . Creating accountable care organizations: the extended hospital medical staff. Health Aff (Millwood). 2007;26(1):w44–w57.
- , . Using performance measurement to drive improvement: a road map for change. Med Care. 2003;41(1 suppl):I48–I60.
- , . Changing physicians' practices. N Engl J Med. 1993;329(17):1271–1273.
- , , . Connections between quality measurement and improvement. Med Care. 2003;41(1 suppl):I30–I38.
- , , . The tension between needing to improve care and knowing how to do it. N Engl J Med. 2007;357(6):608–613.
- , . A qualitative evaluation of geographical localization of hospitalists: how unintended consequences may impact quality. J Gen Intern Med. 2014;29(7):1009–1016.
- , , , , . Disengaged: a qualitative study of communication and collaboration between physicians and other professions on general internal medicine wards. BMC Health Serv Res. 2013;13:494.
- Committee on Quality of Health Care in America; Institute of Medicine. Crossing the Quality Chasm: A New Health System for the 21st Century. Washington, DC: The National Academies Press; 2001.
- . Is US health really the best in the world? JAMA. 2000;284(4):483–485.
- , , , , , . Temporal trends in rates of patient harm resulting from medical care. N Engl J Med. 2010;363(22):2124–2134.
- Indiana University Health. Available at: http://iuhealth.org/methodist/aboIut/. Accessed October 20, 2014.
- University Health Consortium. Available at: https://www.uhc.edu/docs/45014769_QSS_dashboard_FAQs.pdf. Accessed October 23, 2014.
- , , , et al. Distribution of variable vs fixed costs of hospital care. JAMA. 1999;281(7):644–649.
- Centers for Medicare and Medicaid Services. Case mix index. Available at: http://www.cms.gov/Medicare/Medicare‐Fee‐for‐Service‐Payment/AcuteInpatientPPS/Acute‐Inpatient‐Files‐for‐Download‐Items/CMS022630.html. Accessed May 4, 2015.
- University Health Consortium. Available at: https://www.uhc.edu. Accessed October 23, 2014.
- Centers for Medicare and Medicaid Services. Hospital Consumer Assessment of Healthcare Providers and Systems. HCAHPS survey content and administration. Centers for Medicare 280(11):1000–1005.
- Centers for Disease Control and Prevention. FastStats. Available at: http://www.cdc.gov/nchs/fastats/default.htm. Accessed October 27, 2014.
- , . Does your patient know your name? An approach to enhancing patients' awareness of their caretaker's name. J Healthc Qual. 2005;27(4):53–56.
- The Joint Commission. Sentinel event data: root causes by event type 2004‐third quarter. Available at: http://www.jointcommissionorg. Available at: http://www.jointcommission.org/assets/1/18/Root_Causes_by_Event_Type_2004-2Q2013.pdf. Accessed March 26, 2014.
- , , . Interprofessional collaboration: effects of practice‐based interventions on professional practice and healthcare outcomes. Cochrane Database Syst Rev. 2009;(3):CD000072.
- , , , et al. Impact of localizing physicians to hospital units on nurse–physician communication and agreement on the plan of care. J Gen Intern Med. 2009;24(11):1223–1227.
- , , , et al. Impact of localizing general medical teams to a single nursing unit. J Hosp Med. 2012;7(7):551–556.
- , , , et al. Implementation of a physician assistant/hospitalist service in an academic medical center: impact on efficiency and patient outcomes. J Hosp Med. 2008;3(5):361–368.
- , , , , , . Improving teamwork: impact of structured interdisciplinary rounds on a medical teaching unit. J Gen Intern Med. 2010;25(8):826–832.
- , , , ; High Performance Teams and the Hospital of the Future Project Team. Interdisciplinary teamwork in hospitals: a review and practical recommendations for improvement. J Hosp Med. 2011;7(1):48–54.
- , , , , , . Microsystems in health care: part 8. Developing people and improving work life: what front‐line staff told us. Jt Comm J Qual Saf. 2003;29(10):512–522.
- , , , , , . Unit‐based interprofessional leadership models in six US hospitals. J Hosp Med. 2014;9(8):545–550.
- , , , . Creating accountable care organizations: the extended hospital medical staff. Health Aff (Millwood). 2007;26(1):w44–w57.
- , . Using performance measurement to drive improvement: a road map for change. Med Care. 2003;41(1 suppl):I48–I60.
- , . Changing physicians' practices. N Engl J Med. 1993;329(17):1271–1273.
- , , . Connections between quality measurement and improvement. Med Care. 2003;41(1 suppl):I30–I38.
- , , . The tension between needing to improve care and knowing how to do it. N Engl J Med. 2007;357(6):608–613.
- , . A qualitative evaluation of geographical localization of hospitalists: how unintended consequences may impact quality. J Gen Intern Med. 2014;29(7):1009–1016.
- , , , , . Disengaged: a qualitative study of communication and collaboration between physicians and other professions on general internal medicine wards. BMC Health Serv Res. 2013;13:494.
© 2015 Society of Hospital Medicine
Impact of CI Among Hospitalized Elders
In 2001, approximately 12.6 million individuals age 65 and older were discharged from American hospitals with an average length of stay of 5.8 days1 and up to 66% of them suffered from cognitive impairment (CI).220 CI in hospitalized older adults includes a variety of disorders ranging from mild cognitive deficit, delirium, to full‐blown dementia. Dementia is a syndrome of decline in memory plus at least 1 other cognitive domain, such as language, visuospatial, or executive function sufficient to interfere with social or occupational functioning in an alert person.21 Delirium is a disturbance of consciousness with reduced ability to focus, sustain, or shift attention that occurs over a short period of time and tends to fluctuate over the course of the day.22 Mild CI without dementia is defined as the presence of a cognitive deficit in the absence of delirium that does not affect functional performance.23
Hospitalized older adults with CI are vulnerable to hospital complications, including delirium, physical restraints, urinary catheters, and tethers.2, 3, 2435 The management of their medical or surgical illnesses requires avoiding certain medications with anticholinergic activities that might worsen cognition.36 Furthermore, CI may delay diagnostic and therapeutic procedures, demand more time for informed consentrelated issues, and result in difficulty in adherence to medical recommendations.37, 38 The special needs of hospitalized older adults with delirium and dementia has been shown to increase demands on nursing staff, risk of postdischarge institutionalization, length of stay, and health care costs.310, 27, 3948 We wanted to look specifically at CI because it often goes undetected4951 and can have a great impact on the hospital course of elders.
Screening for CI among hospitalized older adults has been considered to have potential benefit in hospital care of older adults.52 Screening may lead to early detection by uncovering subtle symptoms not yet apparent to families or other caregivers who know the patient well but do not notice small declines or changes in day‐to‐day functioning. Early recognition of CI may lead to early treatment and subsequently may delay progression of cognitive decline and improve health outcomes. Screening may enhance physician prescribing practices and reduce exposure to harmful medications among these vulnerable patients. Finally, delirium is an important prognostic indicator, and screening patients could provide invaluable information toward the overall clinical picture. Despite all of this, the current literature does not provide sufficient information to support the use of routine screening on admission.220, 41, 5254 Most of the published studies were conducted among elders who stayed in the hospital for more than 48 hours, missing data on the crucial first 48 hours of the hospital course.220, 41, 5254 These studies did not evaluate the impact of unrecognized CI on the hospital course and the majority of these studies were not conducted in the urban and lower socioeconomic status populations of elders that are the most vulnerable to bad health outcomes.220, 41, 5254 Finally, few studies evaluated the impact of delirium superimposed on CI on the hospital course and mortality of elders.220, 41, 5254
With these details in mind, we wanted to explore the impact of CI recognition among patients age 65 years and older admitted to the medical services of an urban, public hospital in Indianapolis to determine the prevalence and the impact of recognized and unrecognized CI on the hospital course of these elders. Furthermore, we examined the role of delirium superimposed on these hospitalized elders with CI.
Patients and Methods
The study was approved by the Indiana University Purdue University at Indianapolis Institutional Review Board (IRB).
Study Setting and Population
The study was conducted on the inpatient general medicine service of Wishard Memorial Hospital (WMH). WMH is a 450‐bed, university‐affiliated, urban, public hospital that is staffed by Indiana University School of Medicine faculty and house staff. It serves a population of approximately 750,000 in Marion County.
Inclusion and Exclusion Criteria
Patients were enrolled in the study based on the following criteria: (1) at least 65 years of age; (2) hospitalized on a medical ward; (3) able to speak English; and (4) have CI at the time of hospital admission (see below). Patients were excluded if they had previously enrolled in the study, were enrolled in another clinical study at the time of admission, or were aphasic or unresponsive at the time of screening.
Cognitive Screening
CI was determined by the Short Portable Mental Status Questionnaire (SPMSQ),55, 56 chosen for its accuracy56 and the fact that it is entirely verbal in administration. In most cases, patients were followed and reassessed daily. Patients having 2 or more errors, indicating a score of 8 or less on the SPMSQ after adjusting for race and education were considered to have cognitive impairment. The SPMSQ is a brief 10‐item screening test with a sensitivity of 86% and specificity 99.0% for dementia among medical inpatients.56 At the time of cognitive screening, delirium was assessed by using the Confusion Assessment Method (CAM).22 This was also done daily in most cases. The CAM22 is a structured instrument that evaluates the 10 symptoms of delirium specified in the Diagnostic and Statistical Manual of Mental Disorders (DSM)‐III‐R: acute onset, fluctuating course, inattention, disorganized thinking, altered level of consciousness, disorientation, memory impairment, perceptual disturbances, psychomotor agitation or retardation, and sleep/wake disturbance. The CAM score is determined by examining the patient, investigating the chart and interviewing the nurse and/or a family member for: (1) acute and fluctuating changes in mental status, (2) inattention, (3) disorganized or incoherent thinking, and (4) altered level of consciousness. A CAM score is considered to be positive if the patient displays both (1) and (2) with at least one of (3) or (4). The CAM diagnosis of delirium was validated against the clinical judgment of a psychiatrist and found to have a sensitivity of 97% and a specificity of 92%.22 A research assistant (RA) was trained for a period of 9 months by a physician as a rater to interview the patient and administer both the SPMSQ and the CAM at the time of admission and then every weekday. When feasible, the RA administered both the SPMSQ and the CAM within the first few hours of hospitalization, and then followed up with our patients each day. More than 70% of our initial cognitive screening occurred in the first 48 hours of hospital admission, and was repeated on a daily basis. In addition to cognitive assessment, the RA reported the presence or absence of Foley catheterization, physical restraints, and tethers during the cognitive assessment. Agreement was obtained from the general internal medicine group practice physicians both to participate in the study and to request screening for CI as part of the recognized admission standard of care among their hospitalized patients aged 65 years and older. The study coordinator was notified of all admissions for patients aged 65 or older by the hospital intranet e‐mail and paging system. Admission notifications were sent by page and e‐mail on an hourly basis from Monday through Friday, 8:00 AM through 5:00 PM. Those admissions occurring between the hours of 5:00 PM and 8:00 AM were sent during the next normal batch notification. Pages and e‐mails for admissions occurring on Saturday and Sunday were sent on Monday morning at 8:00 AM.
Regenstrief Medical Record System at WMH
The computerized Regenstrief Medical Record System (RMRS) is the primary instrument for processing data and monitoring patient and physician activity for Wishard Health System.57, 58 The RMRS is a modular system, composed of Registration and Scheduling, Laboratory, and Pharmacy database modules. The Registration and Scheduling module is used to make all outpatient appointments for the office practices associated with Wishard Health System. The Laboratory module handles all data for all inpatient and outpatient laboratories. This module also produces all laboratory reports and data used for billing. In addition to laboratory data, this module stores coded results and full‐text interpretations of all imaging studies and special procedures. The Pharmacy module contains information on medication orders captured by the computerized physician order enter (CPOE). The Database module stores all the above data by date in a fully‐coded form. Thus, these data are readily retrievable for individual patients by healthcare providers using online terminals. Data for large numbers of patients are retrievable using a locally developed English‐like language called CARE. Patients can be identified either by a certain restriction list (eg, the list of subjects in a study) or by clinical criteria. The RMRS also maintains a number of other databases including diagnoses, vital signs, results of laboratory tests and diagnostic tests, full‐text discharge summaries, preventive health maneuvers, and detailed information on all inpatient and outpatient charges. It contains death certificate information from the Indiana State Board of Health for all registered patients who die in, or outside of, Indiana. Therefore, the RMRS collects and monitors a broad array of physician and patient activity, practice patterns, utilization, diagnostic test finding, and offers a wonderful array of outcome measures.
Other Data Collections
Patient demographics such as age, gender, race, and education level were determined by the RMRS and by information obtained during the time of cognitive screening. Length of hospital stay and 30‐day posthospitalization mortality were obtained from the RMRS. Comorbidity level was measured by reviewing the RMRS and determining each patient's Charlson comorbidity index total score.59, 60 This score was determined using International Statistical Classification of Diseases and Related Health Problems, 9th edition (ICD‐9) codes gathered from 1 year prior to admission until the patient was discharged from the hospital. Anticholinergic medications were determined by using the Anticholinergic Cognitive Burden Scale,61 an expert‐based practical index. The scale was developed based on a review of all published studies from 1996 to 2007 that measured the anticholinergic activities of a drug and its association with cognitive function in older adults. The list of drugs reviewed was presented to an expert interdisciplinary panel that included geriatricians, geriatric pharmacists, geriatric psychiatrists, general physicians, geriatric nurses, and aging brain researchers. The panel categorized each medication into a possible or definite anticholinergic category based on the severity of its cognitive anticholinergic effects.61 A patient who received at least 1 order of a possible or definite anticholinergic during their hospitalization was considered to be an anticholinergic user. Prior recognition of CI was determined by searching the RMRS for any ICD‐9 code (see Appendix) indicative of dementia, Alzheimer disease, or delirium reported at hospital admission, discharge, or during an 1‐year period prior to hospitalization for every patient enrolled in the study. Those patients with documented ICD‐9 codes were felt recognized as having some form of cognitive impairment. Those who had a positive screen but no prior documentation according to ICD‐9 coding, were said to have unrecognized CI.
Analysis
Descriptive statistics were calculated, including percentages for binary categorical variables, and means and standard deviations for continuous variables. Comparisons between groups were based upon Fisher's Exact Tests for binary categorical variables and t tests for continuous variables. When controlling for covariates such as age, gender, race, Charlson comorbidity index, and SPMSQ at screening, group comparisons were made by using logistic regression for binary categorical variables and multiple regression for continuous variables. Since the distributions of length of stay and Charlson comorbidity index were skewed, all statistical tests comparing them across groups were actually performed on their log‐transformed values.
Results
The Prevalence and Recognition of CI
Table 1 describes the demographic characteristic of our study population, which is a reflection of the public and urban nature of our target hospital. Our study assessed the cognitive status of 997 older adults usually (>70% of the time) within 48 hours of their admission to the medical ward of this urban hospital between July of 2006 and March 2008 (see Table 1) and found that 43% of these elders had evidence of CI as determined by a SPMSQ score of 8 points or less. However, 61% of the 424 cognitively impaired elders were not documented or recognized by the electronic medical record system to have cognitive deficit.
| Variable | n | %/Mean (SD) |
|---|---|---|
| ||
| Age (years), mean (SD) | 997 | 74.8 (7.5) |
| Age 85 (%) | 997 | 12.6 |
| Female (%) | 997 | 67.8 |
| African American (%) | 997 | 59.4 |
| Education (years), mean (SD) | 910 | 10.3 (2.8) |
| Education <12 years (%) | 910 | 59.1 |
| Screened within 48 hours of admission (%) | 997 | 73.2 |
| SPMSQ score at screening, mean (SD) | 997 | 7.7 (2.8) |
| Cognitive impairment based on the SPMSQ score 8 (%) | 997 | 42.5 |
The Impact of Unrecognized CI on the Hospital Course
As expected, hospitalized elders with documented CI were older (mean age 79.1 years vs. 76.1 years; P < 0.001) and had worse cognitive function upon screening than those with unrecognized CI (mean SPMSQ 3.4 points vs. 6.3; P < 0.001). Furthermore, CI recognition was influenced by the elders' race and comorbidity (Table 2); a higher percentage of elders with documented CI were African American (69% vs. 54%; P = 0.003) and had less comorbidity (mean Charlson index 1.9 vs. 2.3; P = 0.03). After adjusting for age, gender, race, comorbidity, and cognitive function at screening, our study found no differences between elders with previously recognized CI and those with unrecognized CI in regard to the length of hospital stay (6.7 days vs. 7.5 days; P = 0.59), 30‐day posthospital mortality (4.8% vs. 6.6%; P > 0.2), home discharge (32% vs. 45%; P > 0.7), hospital readmission (19.2% vs.18.8%; P > 0.6), delirium incidence (27% vs. 21%; P > 0.9), and physical restraints (1.8% vs. 1.5%; P > 0.4). We also found that elders with undocumented CI were not more likely to receive definite anticholinergics (33.2% vs. 32.7%; P > 0.9).
| CI Documented | CI Undocumented | P Value | P Value* | |
|---|---|---|---|---|
| ||||
| n (%) | 165 (39) | 259 (61) | n/a | |
| Age, mean (SD) | 79.1 (7.9) | 76.1 (8.0) | <0.001 | |
| Female (%) | 68.5 | 64.5 | 0.40 | |
| African American (%) | 68.5 | 53.7 | <0.01 | |
| SPMSQ at screen, mean (SD) | 3.4 (2.7) | 6.3 (2.1) | <0.001 | |
| Charlson comorbidity index, mean (SD) | 1.9 (1.9) | 2.3 (2.1) | 0.03 | |
| Length of hospital stay, mean (SD) | 6.7 (5.1) | 7.5 (7.1) | 0.49 | 0.59 |
| Survived at 30 days postdischarge (%) | 95.2 | 93.4 | 0.53 | 0.25 |
| Discharged home (%) | 31.5 | 45.2 | 0.01 | 0.74 |
| Readmission within 30 days after discharge home (%) | 19.2 | 18.8 | 0.99 | 0.66 |
| Incidence of delirium (%) | 26.7 | 20.6 | 0.52 | 0.99 |
| Observed with Foley catheter (%) | 43.6 | 27.4 | <0.001 | 0.61 |
| Observed with physical restraint (%) | 1.8 | 1.5 | 0.99 | 0.31 |
| Observed with tethers (%) | 81.8 | 73.8 | 0.06 | 0.58 |
| With at least 1 Ach (%) | 83.6 | 90.7 | 0.03 | 0.22 |
| Possible Ach (%) | 81.2 | 88.4 | 0.05 | 0.31 |
| Definite Ach (%) | 32.7 | 33.2 | 0.99 | 0.64 |
The Impact of Delirium on the Hospital Course of Elders with CI
Among the 424 hospitalized elders with CI, 163 (38%) had delirium at least once during their hospital course and 24% had delirium on the day of hospital discharge. In comparison to elders who had CI but not delirium during their hospitalization (Table 3), those with at least 1 day of delirium had a higher 30‐day posthospitalization mortality risk (8.6% vs. 4.2%; P = 0.09), stayed in the hospital 3.3 additional days (9.2 days vs. 5.9 days; P < 0.001), were less likely to be discharged home (25% vs. 49%; P < 0.001), were more likely to receive a Foley catheterization (52% vs. 23%; P < 0.001), more likely to be physically restrained (4% vs. 0%; P < 0.01), and more likely to receive tethers during their care (89% vs. 69%; P < 0.001). There was no statistically significant difference between the 2 groups in terms of 30‐day hospital readmission rates or in their use of definite anticholinergics (Table 3).
| Delirium+* | Delirium | P value | |
|---|---|---|---|
| |||
| n (%) | 163 (38) | 261 (62) | n/a |
| Age, mean (SD) | 78.4 (8.5) | 76.5 (7.8) | 0.02 |
| Female (%) | 60.1 | 69.7 | 0.05 |
| African American (%) | 64.4 | 56.3 | 0.10 |
| Charlson comorbidity index, mean (SD) | 1.8 (1.9) | 2.3 (2.1) | 0.01 |
| Length of hospital stay, mean (SD) | 9.2 (7.9) | 5.9 (4.9) | <0.001 |
| Survived at 30‐day postdischarge (%) | 91.4 | 95.8 | 0.09 |
| Discharged home (%) | 24.5 | 49.4 | <0.001 |
| Readmission within 30 days after discharge home (%) | 22.5 | 17.8 | 0.50 |
| Observed with Foley catheter (%) | 51.5 | 22.6 | <0.001 |
| Observed with physical restraint (%) | 4.3 | 0.0 | <0.01 |
| Observed with tethers (%) | 89.0 | 69.4 | <0.001 |
| With at least 1 anticholinergic (%) | 83.4 | 90.8 | 0.03 |
| Possible anticholinergic (%) | 80.4 | 88.9 | 0.02 |
| Definite anticholinergic (%) | 36.8 | 30.7 | 0.20 |
Discussion
Our study found that in an urban, public hospital, acute or preexisting CI affects more than one‐third of hospitalized elders admitted to general medical services. Unfortunately, our hospital system does not currently recognize the majority of these vulnerable patients. Our study also found that delirium affects more than one‐third of hospitalized elders with CI during their hospital course. Delirium complicates hospital care by prolonging length of stay and decreasing the probability of surviving and getting discharged home. It leads to high use of Foley catheterization, physical restraints, and tethers.
The high prevalence of CI with and without delirium in our cohort is within the rates reported previously in the literature. It is estimated that the prevalence of CI in hospitalized older adults ranges from 14% to 66%, depending on the method used to measure cognition, the definition of CI, and the type of hospital ward (surgical, medical, and geriatric units).220 One particular study that used a similar cognitive assessment method reported higher prevalence rates for both CI and delirium.11 The study randomly evaluated a sample of 201 patients age 65 and over who were hospitalized for a medical illness and found that 56% of the cohort suffered from CI and among those with CI, 47% had delirium.11 The difference between this finding and our study is most likely due to our sampling technique; more than 70% of our cognitive screening occurred in the first 48 hours of hospital admission whereas the Australian study, in similar enrollment criteria to all of the published studies in this area, excluded patients who were discharged within 48 hours of admission. We believe, however, that by including the first 48 hours of admission in our design, our study provides a more generalizable reflection of the actual acute care experience.
The impact of delirium on the course of hospital care found in our study supports some of the findings from previous studies conducted in the past 2 decades.5, 6, 11 Despite 2 decades of clinical research, delirium continues to increase mortality, hospital stays, and posthospital institutionalization.
We were surprised to find that patients suffering from delirium continue to receive at least 1 definite anticholinergic medication. Such medications are considered inappropriate among patients with any form of cognitive impairment.36, 62 Although the impact of anticholinergic medications on hospitalized outcomes is less well‐described, their use has been suspected to negatively impact long‐term outcomes of cognitive impairment.61, 63 Our study found no difference in the use of anticholinergic medications between those with CI who experienced delirium and those who did not; however, the total burden of anticholinergic medication was not assessed in a quantitative manner. It is still unknown if certain anticholinergic medications or a cumulative effect of anticholinergic medications may impact cognitive or health‐related outcomes in a vulnerable older population with CI.
Although our study reported for the first time in a systematic way the rate of undocumented CI among hospitalized elders found to have CI on admission, we found no impact of such underrecognition on the length of hospital stay, mortality, discharge location, and delirium occurrence. Although the use of anticholinergic medications is not recommended for patients with any form of CI, our results indicate that a significant number of patients with cognitive impairment continue to receive inappropriate medications. CI recognition in the elderly was not shown to have a statistically significant affect on length of stay, cost, or mortality.
Our study has some limitations. First of all, we did not determine the underlying types of CI such as Alzheimer disease, vascular dementia, mild cognitive impairment, or reversible etiology other than delirium. Such a categorization requires posthospital assessment, which was not included in our study design. Second, our delirium incidence rate and delirium impact on hospital outcomes might be very conservative and may underestimate its true prevalence and correlation due to our data collection methods. Despite the fluctuating nature of delirium, our study was not designed to assess the presence of delirium every shift and tried to assess cognitive function on a daily basis throughout the patient's hospitalization. Therefore, the severity and duration of delirium could not be accurately assessed. Our reported rates of use of Foley catheterization, physical restraints, and tethers are also very conservative and we could not determine the appropriateness of these procedures. Our study was conducted in 1 public hospital in an urban city with a higher percentage of African Americans. Thus, our sample is not a true representative sample. However, studies with significant representation of minority groups are not common in the research literature, especially in CI research; we hope to fulfill some of the gaps in the literature regarding the most vulnerable older American population. Finally, we were limited in our use of ICD‐9 coding to determine if patients had previously been recognized by other providers as having CI. ICD‐9 coding, while useful, is not perfect in identifying all if a patient's medical problems. Use of coding to determine whether a patient had been recognized as impaired also does not allow us to determine when the diagnosis was made.
In conclusion, our study evaluated cognitive impairment in hospitalized elders and found that in our cohort of 997 patients, 43% were cognitively impaired on admission. Of those with CI, 61% were not documented or recognized as impaired. We found no statistically significant difference between those with documented CI and those with undocumented CI in terms of length of stay, mortality, home discharge, readmission rates, incidence of delirium, or potential to receive anticholinergics or restraints. Among those with CI, 38% had delirium. Those with delirium experienced increased length of stay, decreased discharge to home, and increased use of Foley catheters and restraints.
- ,.National hospital discharge survey: annual summary, 1994.Vital Health Stat 13.1997;(128):i–v;1–50.
- .The dilemma of delirium: clinical and research controversies regarding diagnosis and evaluation of delirium in hospitalized elderly medical patients.Am J Med.1994;97:278–288.
- ,,.Dementia in medical wards.J Clin Epidemiol.1988;41:123–126.
- ,,,,,.Mental and behavioral disturbances in dementia: findings from the Cache County Study on Memory in Aging.Am J Psychiatry.2000;157:708–714.
- ,,, et al.Randomized, placebo‐controlled, double‐blind clinical trial of sertraline in the treatment of depression complicating Alzheimer's disease: initial results from the Depression in Alzheimer's Disease study.Am J Psychiatry.2000;157:1686–1689.
- ,,.Dementia in elderly persons in a general hospital.Am J Psychiatry.2000;157:704–707.
- ,,,,.A prospective study of the impact of psychiatric comorbidity on length of hospital stays of elderly medical‐surgical inpatients.Psychosomatics.1998;39:273–280.
- ,.Psychiatric comorbidity and length of stay in the general hospital. A critical review of outcome studies.Psychosomatics.1994;35:233–252.
- ,,,.Dementia among medical inpatients. Evaluation of 2000 consecutive admissions.Arch Intern Med.1986;146:1923–1926.
- ,,,.The consequences of non‐cognitive symptoms of dementia in medical hospital departments.Int J Psychiatry Med.2003;33:257–271.
- ,,.Cognitive impairment in medical inpatients. I: Screening for dementia—is history better than mental state?Age Ageing.1997;26:31–35.
- ,,, et al.Acute confusional states in elderly patients treated for femoral neck fracture.J Am Geriatr Soc.1988;36:525–530.
- ,.A prospective study of elderly general surgical patients: II. Post‐operative complications.Age Ageing.1989;18:316–326.
- ,,,.Dementia and depression in elderly medical inpatients.Int Psychogeriatr.2000;12:67–75.
- ,,.[Psychiatric disorders in elderly general hospital patients: incidence and long‐term prognosis].Nervenarzt.1993;64:53–61. [German]
- ,.Delirium and dementia in acute medical admissions of elderly patients in Iceland.Acta Psychiatr Scand.1993;87:123–127.
- ,,,,,.[Psychiatric morbidity in elderly patients admitted to a general hospital. A day‐prevalence study].Med Clin (Barc).1991;97:206–210. [Spanish]
- ,,,,,.Detection of psychiatric disorders in elderly medical inpatients.Age Ageing.1994;23:307–311.
- ,,,.Cognitive impairment, emotional disorder and length of stay of elderly patients in a district general hospital.Br J Med Psychol.1987;60(Pt 2):133–139.
- ,,.An investigation of the components of best nursing practice in the care of acutely ill hospitalized older patients with coincidental dementia: a multi‐method design.J Adv Nurs1999;30:1127–1136.
- American Psychiatric Association.Diagnostic and Statistical Manual of Mental Disorders.4th ed.Washington, DC:American Psychiatric Association;1994.
- ,,,,,.Clarifying confusion: the confusion assessment method. A new method for detection of delirium.Ann Intern Med.1990;113:941–948.
- ,,, et al.Prevalence of cognitive impairment: data from the Indianapolis Study of Health and Aging.Neurology.2001;57:1655–1662.
- ,,.Delirium: a symptom of how hospital care is failing older persons and a window to improve quality of hospital care.Am J Med.1999;106:565–573.
- ,,, et al.A multicomponent intervention to prevent delirium in hospitalized older patients.N Engl J Med.1999;340:669–676.
- ,,,.Iatrogenic causes of falls in hospitalised elderly patients: a case‐control study.Postgrad Med J.2002;78:487–489.
- ,,.A prospective study of delirium in hospitalized elderly.JAMA.1990;263:1097–1101.
- ,.The prognostic significance of delirium in older hospital patients.J Am Geriatr Soc.1997;45:174–178.
- ,.Prognosis of delirium in elderly hospital patients.CMAJ.1993;149:41–46.
- ,,,,.The detection of psychiatric morbidity and its effects on outcome in acute elderly medical admissions.Int J Ger Psych1991;6:861–866.
- ,,.Adverse consequences of hospitalization in the elderly.Soc Sci Med.1982;16:1033–1038.
- ,,, et al.Incidence of adverse events and negligence in hospitalized patients. Results of the Harvard Medical Practice Study I.N Engl J Med.1991;324:370–376.
- ,,,.Delirium in elderly patients: an overview of the state of the science.J Gerontol Nurs.2001;27:12–20.
- ,,,,.A predictive model for delirium in hospitalized elderly medical patients based on admission characteristics.Ann Intern Med.1993;119:474–481.
- ,.Precipitating factors for delirium in hospitalized elderly persons. Predictive model and interrelationship with baseline vulnerability.JAMA.1996;275:852–857.
- ,,,,,.Updating the Beers criteria for potentially inappropriate medication use in older adults: results of a US consensus panel of experts.Arch Intern Med.2003;163:2716–2724.
- ,,, et al.A clinical prediction rule for delirium after elective noncardiac surgery.JAMA.1994;271:134–139.
- ,,, et al.The relationship of postoperative delirium with psychoactive medications.JAMA.1994;272:1518–1522.
- ,,, et al.How do delirium and dementia increase length of stay of elderly general medical inpatients?Psychosomatics.2004;45:235–242.
- ,,,,,.The relationship between a dementia diagnosis, chronic illness, Medicare expenditures, and hospital use.J Am Geriatr Soc.2004;52:187–194.
- ,,, et al.Risk factors for delirium in hospitalized elderly.JAMA.1992;267:827–831.
- ,,,,.Psychological comorbidity and length of stay in the general hospital.Am J Psychiatry.1991;148:324–329.
- ,,, et al.Delirium. The occurrence and persistence of symptoms among elderly hospitalized patients.Arch Intern Med.1992;152:334–340.
- ,,.What happens to medical patients with psychiatric disorder?J Psychosom Res1988;32:541–549.
- ,,,,.[Dementia syndromes and length of stay of elderly patients in internal medicine].Ann Med Interne (Paris).1997;148:424–426. [French]
- ,,, et al.[Hospital discharge planning and length of hospital stay in elderly patients admitted through the emergency department].Rev Epidemiol Sante Publique.1995;43:337–347. [French]
- ,,,,.The effect of dementia on acute care in a geriatric medical unit.Int Psychogeriatr.1992;4:231–239.
- ,,,.Cognitive impairment. Can it predict the course of hospitalized patients?J Am Geriatr Soc.1986;34:579–585.
- ,,,,;US Preventive Services Task Force. Screening for dementia in primary care: a summary of the evidence for the U.S. Preventive Services Task Force.Ann Intern Med.2003;138(11):927–937.
- ,,,.Cognitive impairment in the elderly medically ill: how often is it missed?Int J Geriatr Psychiatry.1993;8:929–937.
- .Recognition of cognitive impairment in elderly medical in‐patients.J R Soc Med.1995;88:183–184.
- ,.Quality indicators for dementia in vulnerable community‐dwelling and hospitalized elders.Ann Intern Med.2001;135:668–676.
- ,,,.Reducing delirium after hip fracture: a randomized trial.J Am Geriatr Soc.2001;49:516–522.
- ,.Prevalence of psychotic symptoms in delirium.Psychosomatics.2000;41:519–522.
- .A short portable mental status questionnaire for the assessment of organic brain deficit in elderly patients.J Am Geriatr Soc.1975;23:433–441.
- ,,,.Short Portable Mental Status Questionnaire as a screening test for dementia and delirium among the elderly.J Am Geriatr Soc.1987;35:412–416.
- ,,, et al.The Regenstrief Medical Record System: a quarter century experience.Int J Med Inform.1999;54:225–253.
- ,,,,,.Factors determining the decision to institutionalize dementing individuals: a prospective study.Gerontologist.1993;33:714–720.
- ,,,,,.Resuscitation: how do we decide? A prospective study of physicians' preferences and the clinical course of hospitalized patients.JAMA.1986;255:1316–1322.
- ,,,,,.Assessing illness severity: does clinical judgment work?J Chronic Dis.1986;39:439–452.
- ,,,,.Impact of anticholinergics on the aging brain: a review and practical application.Aging Health.2008;4(3):311–320.
- ,.Delirium in hospitalized older adults. In: Ham R, Sloane P, Warshaw G, eds.Primary Care Geriatrics: A Case‐Based Approach.5th ed.Philadelphia, PA:Mosby Elsevier;2007:210–218.
- ,,, et al.The association between cognition and histamine‐2 receptor antagonists in African Americans.J Am Geriatr Soc.2007;55(8):1248–1253.
In 2001, approximately 12.6 million individuals age 65 and older were discharged from American hospitals with an average length of stay of 5.8 days1 and up to 66% of them suffered from cognitive impairment (CI).220 CI in hospitalized older adults includes a variety of disorders ranging from mild cognitive deficit, delirium, to full‐blown dementia. Dementia is a syndrome of decline in memory plus at least 1 other cognitive domain, such as language, visuospatial, or executive function sufficient to interfere with social or occupational functioning in an alert person.21 Delirium is a disturbance of consciousness with reduced ability to focus, sustain, or shift attention that occurs over a short period of time and tends to fluctuate over the course of the day.22 Mild CI without dementia is defined as the presence of a cognitive deficit in the absence of delirium that does not affect functional performance.23
Hospitalized older adults with CI are vulnerable to hospital complications, including delirium, physical restraints, urinary catheters, and tethers.2, 3, 2435 The management of their medical or surgical illnesses requires avoiding certain medications with anticholinergic activities that might worsen cognition.36 Furthermore, CI may delay diagnostic and therapeutic procedures, demand more time for informed consentrelated issues, and result in difficulty in adherence to medical recommendations.37, 38 The special needs of hospitalized older adults with delirium and dementia has been shown to increase demands on nursing staff, risk of postdischarge institutionalization, length of stay, and health care costs.310, 27, 3948 We wanted to look specifically at CI because it often goes undetected4951 and can have a great impact on the hospital course of elders.
Screening for CI among hospitalized older adults has been considered to have potential benefit in hospital care of older adults.52 Screening may lead to early detection by uncovering subtle symptoms not yet apparent to families or other caregivers who know the patient well but do not notice small declines or changes in day‐to‐day functioning. Early recognition of CI may lead to early treatment and subsequently may delay progression of cognitive decline and improve health outcomes. Screening may enhance physician prescribing practices and reduce exposure to harmful medications among these vulnerable patients. Finally, delirium is an important prognostic indicator, and screening patients could provide invaluable information toward the overall clinical picture. Despite all of this, the current literature does not provide sufficient information to support the use of routine screening on admission.220, 41, 5254 Most of the published studies were conducted among elders who stayed in the hospital for more than 48 hours, missing data on the crucial first 48 hours of the hospital course.220, 41, 5254 These studies did not evaluate the impact of unrecognized CI on the hospital course and the majority of these studies were not conducted in the urban and lower socioeconomic status populations of elders that are the most vulnerable to bad health outcomes.220, 41, 5254 Finally, few studies evaluated the impact of delirium superimposed on CI on the hospital course and mortality of elders.220, 41, 5254
With these details in mind, we wanted to explore the impact of CI recognition among patients age 65 years and older admitted to the medical services of an urban, public hospital in Indianapolis to determine the prevalence and the impact of recognized and unrecognized CI on the hospital course of these elders. Furthermore, we examined the role of delirium superimposed on these hospitalized elders with CI.
Patients and Methods
The study was approved by the Indiana University Purdue University at Indianapolis Institutional Review Board (IRB).
Study Setting and Population
The study was conducted on the inpatient general medicine service of Wishard Memorial Hospital (WMH). WMH is a 450‐bed, university‐affiliated, urban, public hospital that is staffed by Indiana University School of Medicine faculty and house staff. It serves a population of approximately 750,000 in Marion County.
Inclusion and Exclusion Criteria
Patients were enrolled in the study based on the following criteria: (1) at least 65 years of age; (2) hospitalized on a medical ward; (3) able to speak English; and (4) have CI at the time of hospital admission (see below). Patients were excluded if they had previously enrolled in the study, were enrolled in another clinical study at the time of admission, or were aphasic or unresponsive at the time of screening.
Cognitive Screening
CI was determined by the Short Portable Mental Status Questionnaire (SPMSQ),55, 56 chosen for its accuracy56 and the fact that it is entirely verbal in administration. In most cases, patients were followed and reassessed daily. Patients having 2 or more errors, indicating a score of 8 or less on the SPMSQ after adjusting for race and education were considered to have cognitive impairment. The SPMSQ is a brief 10‐item screening test with a sensitivity of 86% and specificity 99.0% for dementia among medical inpatients.56 At the time of cognitive screening, delirium was assessed by using the Confusion Assessment Method (CAM).22 This was also done daily in most cases. The CAM22 is a structured instrument that evaluates the 10 symptoms of delirium specified in the Diagnostic and Statistical Manual of Mental Disorders (DSM)‐III‐R: acute onset, fluctuating course, inattention, disorganized thinking, altered level of consciousness, disorientation, memory impairment, perceptual disturbances, psychomotor agitation or retardation, and sleep/wake disturbance. The CAM score is determined by examining the patient, investigating the chart and interviewing the nurse and/or a family member for: (1) acute and fluctuating changes in mental status, (2) inattention, (3) disorganized or incoherent thinking, and (4) altered level of consciousness. A CAM score is considered to be positive if the patient displays both (1) and (2) with at least one of (3) or (4). The CAM diagnosis of delirium was validated against the clinical judgment of a psychiatrist and found to have a sensitivity of 97% and a specificity of 92%.22 A research assistant (RA) was trained for a period of 9 months by a physician as a rater to interview the patient and administer both the SPMSQ and the CAM at the time of admission and then every weekday. When feasible, the RA administered both the SPMSQ and the CAM within the first few hours of hospitalization, and then followed up with our patients each day. More than 70% of our initial cognitive screening occurred in the first 48 hours of hospital admission, and was repeated on a daily basis. In addition to cognitive assessment, the RA reported the presence or absence of Foley catheterization, physical restraints, and tethers during the cognitive assessment. Agreement was obtained from the general internal medicine group practice physicians both to participate in the study and to request screening for CI as part of the recognized admission standard of care among their hospitalized patients aged 65 years and older. The study coordinator was notified of all admissions for patients aged 65 or older by the hospital intranet e‐mail and paging system. Admission notifications were sent by page and e‐mail on an hourly basis from Monday through Friday, 8:00 AM through 5:00 PM. Those admissions occurring between the hours of 5:00 PM and 8:00 AM were sent during the next normal batch notification. Pages and e‐mails for admissions occurring on Saturday and Sunday were sent on Monday morning at 8:00 AM.
Regenstrief Medical Record System at WMH
The computerized Regenstrief Medical Record System (RMRS) is the primary instrument for processing data and monitoring patient and physician activity for Wishard Health System.57, 58 The RMRS is a modular system, composed of Registration and Scheduling, Laboratory, and Pharmacy database modules. The Registration and Scheduling module is used to make all outpatient appointments for the office practices associated with Wishard Health System. The Laboratory module handles all data for all inpatient and outpatient laboratories. This module also produces all laboratory reports and data used for billing. In addition to laboratory data, this module stores coded results and full‐text interpretations of all imaging studies and special procedures. The Pharmacy module contains information on medication orders captured by the computerized physician order enter (CPOE). The Database module stores all the above data by date in a fully‐coded form. Thus, these data are readily retrievable for individual patients by healthcare providers using online terminals. Data for large numbers of patients are retrievable using a locally developed English‐like language called CARE. Patients can be identified either by a certain restriction list (eg, the list of subjects in a study) or by clinical criteria. The RMRS also maintains a number of other databases including diagnoses, vital signs, results of laboratory tests and diagnostic tests, full‐text discharge summaries, preventive health maneuvers, and detailed information on all inpatient and outpatient charges. It contains death certificate information from the Indiana State Board of Health for all registered patients who die in, or outside of, Indiana. Therefore, the RMRS collects and monitors a broad array of physician and patient activity, practice patterns, utilization, diagnostic test finding, and offers a wonderful array of outcome measures.
Other Data Collections
Patient demographics such as age, gender, race, and education level were determined by the RMRS and by information obtained during the time of cognitive screening. Length of hospital stay and 30‐day posthospitalization mortality were obtained from the RMRS. Comorbidity level was measured by reviewing the RMRS and determining each patient's Charlson comorbidity index total score.59, 60 This score was determined using International Statistical Classification of Diseases and Related Health Problems, 9th edition (ICD‐9) codes gathered from 1 year prior to admission until the patient was discharged from the hospital. Anticholinergic medications were determined by using the Anticholinergic Cognitive Burden Scale,61 an expert‐based practical index. The scale was developed based on a review of all published studies from 1996 to 2007 that measured the anticholinergic activities of a drug and its association with cognitive function in older adults. The list of drugs reviewed was presented to an expert interdisciplinary panel that included geriatricians, geriatric pharmacists, geriatric psychiatrists, general physicians, geriatric nurses, and aging brain researchers. The panel categorized each medication into a possible or definite anticholinergic category based on the severity of its cognitive anticholinergic effects.61 A patient who received at least 1 order of a possible or definite anticholinergic during their hospitalization was considered to be an anticholinergic user. Prior recognition of CI was determined by searching the RMRS for any ICD‐9 code (see Appendix) indicative of dementia, Alzheimer disease, or delirium reported at hospital admission, discharge, or during an 1‐year period prior to hospitalization for every patient enrolled in the study. Those patients with documented ICD‐9 codes were felt recognized as having some form of cognitive impairment. Those who had a positive screen but no prior documentation according to ICD‐9 coding, were said to have unrecognized CI.
Analysis
Descriptive statistics were calculated, including percentages for binary categorical variables, and means and standard deviations for continuous variables. Comparisons between groups were based upon Fisher's Exact Tests for binary categorical variables and t tests for continuous variables. When controlling for covariates such as age, gender, race, Charlson comorbidity index, and SPMSQ at screening, group comparisons were made by using logistic regression for binary categorical variables and multiple regression for continuous variables. Since the distributions of length of stay and Charlson comorbidity index were skewed, all statistical tests comparing them across groups were actually performed on their log‐transformed values.
Results
The Prevalence and Recognition of CI
Table 1 describes the demographic characteristic of our study population, which is a reflection of the public and urban nature of our target hospital. Our study assessed the cognitive status of 997 older adults usually (>70% of the time) within 48 hours of their admission to the medical ward of this urban hospital between July of 2006 and March 2008 (see Table 1) and found that 43% of these elders had evidence of CI as determined by a SPMSQ score of 8 points or less. However, 61% of the 424 cognitively impaired elders were not documented or recognized by the electronic medical record system to have cognitive deficit.
| Variable | n | %/Mean (SD) |
|---|---|---|
| ||
| Age (years), mean (SD) | 997 | 74.8 (7.5) |
| Age 85 (%) | 997 | 12.6 |
| Female (%) | 997 | 67.8 |
| African American (%) | 997 | 59.4 |
| Education (years), mean (SD) | 910 | 10.3 (2.8) |
| Education <12 years (%) | 910 | 59.1 |
| Screened within 48 hours of admission (%) | 997 | 73.2 |
| SPMSQ score at screening, mean (SD) | 997 | 7.7 (2.8) |
| Cognitive impairment based on the SPMSQ score 8 (%) | 997 | 42.5 |
The Impact of Unrecognized CI on the Hospital Course
As expected, hospitalized elders with documented CI were older (mean age 79.1 years vs. 76.1 years; P < 0.001) and had worse cognitive function upon screening than those with unrecognized CI (mean SPMSQ 3.4 points vs. 6.3; P < 0.001). Furthermore, CI recognition was influenced by the elders' race and comorbidity (Table 2); a higher percentage of elders with documented CI were African American (69% vs. 54%; P = 0.003) and had less comorbidity (mean Charlson index 1.9 vs. 2.3; P = 0.03). After adjusting for age, gender, race, comorbidity, and cognitive function at screening, our study found no differences between elders with previously recognized CI and those with unrecognized CI in regard to the length of hospital stay (6.7 days vs. 7.5 days; P = 0.59), 30‐day posthospital mortality (4.8% vs. 6.6%; P > 0.2), home discharge (32% vs. 45%; P > 0.7), hospital readmission (19.2% vs.18.8%; P > 0.6), delirium incidence (27% vs. 21%; P > 0.9), and physical restraints (1.8% vs. 1.5%; P > 0.4). We also found that elders with undocumented CI were not more likely to receive definite anticholinergics (33.2% vs. 32.7%; P > 0.9).
| CI Documented | CI Undocumented | P Value | P Value* | |
|---|---|---|---|---|
| ||||
| n (%) | 165 (39) | 259 (61) | n/a | |
| Age, mean (SD) | 79.1 (7.9) | 76.1 (8.0) | <0.001 | |
| Female (%) | 68.5 | 64.5 | 0.40 | |
| African American (%) | 68.5 | 53.7 | <0.01 | |
| SPMSQ at screen, mean (SD) | 3.4 (2.7) | 6.3 (2.1) | <0.001 | |
| Charlson comorbidity index, mean (SD) | 1.9 (1.9) | 2.3 (2.1) | 0.03 | |
| Length of hospital stay, mean (SD) | 6.7 (5.1) | 7.5 (7.1) | 0.49 | 0.59 |
| Survived at 30 days postdischarge (%) | 95.2 | 93.4 | 0.53 | 0.25 |
| Discharged home (%) | 31.5 | 45.2 | 0.01 | 0.74 |
| Readmission within 30 days after discharge home (%) | 19.2 | 18.8 | 0.99 | 0.66 |
| Incidence of delirium (%) | 26.7 | 20.6 | 0.52 | 0.99 |
| Observed with Foley catheter (%) | 43.6 | 27.4 | <0.001 | 0.61 |
| Observed with physical restraint (%) | 1.8 | 1.5 | 0.99 | 0.31 |
| Observed with tethers (%) | 81.8 | 73.8 | 0.06 | 0.58 |
| With at least 1 Ach (%) | 83.6 | 90.7 | 0.03 | 0.22 |
| Possible Ach (%) | 81.2 | 88.4 | 0.05 | 0.31 |
| Definite Ach (%) | 32.7 | 33.2 | 0.99 | 0.64 |
The Impact of Delirium on the Hospital Course of Elders with CI
Among the 424 hospitalized elders with CI, 163 (38%) had delirium at least once during their hospital course and 24% had delirium on the day of hospital discharge. In comparison to elders who had CI but not delirium during their hospitalization (Table 3), those with at least 1 day of delirium had a higher 30‐day posthospitalization mortality risk (8.6% vs. 4.2%; P = 0.09), stayed in the hospital 3.3 additional days (9.2 days vs. 5.9 days; P < 0.001), were less likely to be discharged home (25% vs. 49%; P < 0.001), were more likely to receive a Foley catheterization (52% vs. 23%; P < 0.001), more likely to be physically restrained (4% vs. 0%; P < 0.01), and more likely to receive tethers during their care (89% vs. 69%; P < 0.001). There was no statistically significant difference between the 2 groups in terms of 30‐day hospital readmission rates or in their use of definite anticholinergics (Table 3).
| Delirium+* | Delirium | P value | |
|---|---|---|---|
| |||
| n (%) | 163 (38) | 261 (62) | n/a |
| Age, mean (SD) | 78.4 (8.5) | 76.5 (7.8) | 0.02 |
| Female (%) | 60.1 | 69.7 | 0.05 |
| African American (%) | 64.4 | 56.3 | 0.10 |
| Charlson comorbidity index, mean (SD) | 1.8 (1.9) | 2.3 (2.1) | 0.01 |
| Length of hospital stay, mean (SD) | 9.2 (7.9) | 5.9 (4.9) | <0.001 |
| Survived at 30‐day postdischarge (%) | 91.4 | 95.8 | 0.09 |
| Discharged home (%) | 24.5 | 49.4 | <0.001 |
| Readmission within 30 days after discharge home (%) | 22.5 | 17.8 | 0.50 |
| Observed with Foley catheter (%) | 51.5 | 22.6 | <0.001 |
| Observed with physical restraint (%) | 4.3 | 0.0 | <0.01 |
| Observed with tethers (%) | 89.0 | 69.4 | <0.001 |
| With at least 1 anticholinergic (%) | 83.4 | 90.8 | 0.03 |
| Possible anticholinergic (%) | 80.4 | 88.9 | 0.02 |
| Definite anticholinergic (%) | 36.8 | 30.7 | 0.20 |
Discussion
Our study found that in an urban, public hospital, acute or preexisting CI affects more than one‐third of hospitalized elders admitted to general medical services. Unfortunately, our hospital system does not currently recognize the majority of these vulnerable patients. Our study also found that delirium affects more than one‐third of hospitalized elders with CI during their hospital course. Delirium complicates hospital care by prolonging length of stay and decreasing the probability of surviving and getting discharged home. It leads to high use of Foley catheterization, physical restraints, and tethers.
The high prevalence of CI with and without delirium in our cohort is within the rates reported previously in the literature. It is estimated that the prevalence of CI in hospitalized older adults ranges from 14% to 66%, depending on the method used to measure cognition, the definition of CI, and the type of hospital ward (surgical, medical, and geriatric units).220 One particular study that used a similar cognitive assessment method reported higher prevalence rates for both CI and delirium.11 The study randomly evaluated a sample of 201 patients age 65 and over who were hospitalized for a medical illness and found that 56% of the cohort suffered from CI and among those with CI, 47% had delirium.11 The difference between this finding and our study is most likely due to our sampling technique; more than 70% of our cognitive screening occurred in the first 48 hours of hospital admission whereas the Australian study, in similar enrollment criteria to all of the published studies in this area, excluded patients who were discharged within 48 hours of admission. We believe, however, that by including the first 48 hours of admission in our design, our study provides a more generalizable reflection of the actual acute care experience.
The impact of delirium on the course of hospital care found in our study supports some of the findings from previous studies conducted in the past 2 decades.5, 6, 11 Despite 2 decades of clinical research, delirium continues to increase mortality, hospital stays, and posthospital institutionalization.
We were surprised to find that patients suffering from delirium continue to receive at least 1 definite anticholinergic medication. Such medications are considered inappropriate among patients with any form of cognitive impairment.36, 62 Although the impact of anticholinergic medications on hospitalized outcomes is less well‐described, their use has been suspected to negatively impact long‐term outcomes of cognitive impairment.61, 63 Our study found no difference in the use of anticholinergic medications between those with CI who experienced delirium and those who did not; however, the total burden of anticholinergic medication was not assessed in a quantitative manner. It is still unknown if certain anticholinergic medications or a cumulative effect of anticholinergic medications may impact cognitive or health‐related outcomes in a vulnerable older population with CI.
Although our study reported for the first time in a systematic way the rate of undocumented CI among hospitalized elders found to have CI on admission, we found no impact of such underrecognition on the length of hospital stay, mortality, discharge location, and delirium occurrence. Although the use of anticholinergic medications is not recommended for patients with any form of CI, our results indicate that a significant number of patients with cognitive impairment continue to receive inappropriate medications. CI recognition in the elderly was not shown to have a statistically significant affect on length of stay, cost, or mortality.
Our study has some limitations. First of all, we did not determine the underlying types of CI such as Alzheimer disease, vascular dementia, mild cognitive impairment, or reversible etiology other than delirium. Such a categorization requires posthospital assessment, which was not included in our study design. Second, our delirium incidence rate and delirium impact on hospital outcomes might be very conservative and may underestimate its true prevalence and correlation due to our data collection methods. Despite the fluctuating nature of delirium, our study was not designed to assess the presence of delirium every shift and tried to assess cognitive function on a daily basis throughout the patient's hospitalization. Therefore, the severity and duration of delirium could not be accurately assessed. Our reported rates of use of Foley catheterization, physical restraints, and tethers are also very conservative and we could not determine the appropriateness of these procedures. Our study was conducted in 1 public hospital in an urban city with a higher percentage of African Americans. Thus, our sample is not a true representative sample. However, studies with significant representation of minority groups are not common in the research literature, especially in CI research; we hope to fulfill some of the gaps in the literature regarding the most vulnerable older American population. Finally, we were limited in our use of ICD‐9 coding to determine if patients had previously been recognized by other providers as having CI. ICD‐9 coding, while useful, is not perfect in identifying all if a patient's medical problems. Use of coding to determine whether a patient had been recognized as impaired also does not allow us to determine when the diagnosis was made.
In conclusion, our study evaluated cognitive impairment in hospitalized elders and found that in our cohort of 997 patients, 43% were cognitively impaired on admission. Of those with CI, 61% were not documented or recognized as impaired. We found no statistically significant difference between those with documented CI and those with undocumented CI in terms of length of stay, mortality, home discharge, readmission rates, incidence of delirium, or potential to receive anticholinergics or restraints. Among those with CI, 38% had delirium. Those with delirium experienced increased length of stay, decreased discharge to home, and increased use of Foley catheters and restraints.
In 2001, approximately 12.6 million individuals age 65 and older were discharged from American hospitals with an average length of stay of 5.8 days1 and up to 66% of them suffered from cognitive impairment (CI).220 CI in hospitalized older adults includes a variety of disorders ranging from mild cognitive deficit, delirium, to full‐blown dementia. Dementia is a syndrome of decline in memory plus at least 1 other cognitive domain, such as language, visuospatial, or executive function sufficient to interfere with social or occupational functioning in an alert person.21 Delirium is a disturbance of consciousness with reduced ability to focus, sustain, or shift attention that occurs over a short period of time and tends to fluctuate over the course of the day.22 Mild CI without dementia is defined as the presence of a cognitive deficit in the absence of delirium that does not affect functional performance.23
Hospitalized older adults with CI are vulnerable to hospital complications, including delirium, physical restraints, urinary catheters, and tethers.2, 3, 2435 The management of their medical or surgical illnesses requires avoiding certain medications with anticholinergic activities that might worsen cognition.36 Furthermore, CI may delay diagnostic and therapeutic procedures, demand more time for informed consentrelated issues, and result in difficulty in adherence to medical recommendations.37, 38 The special needs of hospitalized older adults with delirium and dementia has been shown to increase demands on nursing staff, risk of postdischarge institutionalization, length of stay, and health care costs.310, 27, 3948 We wanted to look specifically at CI because it often goes undetected4951 and can have a great impact on the hospital course of elders.
Screening for CI among hospitalized older adults has been considered to have potential benefit in hospital care of older adults.52 Screening may lead to early detection by uncovering subtle symptoms not yet apparent to families or other caregivers who know the patient well but do not notice small declines or changes in day‐to‐day functioning. Early recognition of CI may lead to early treatment and subsequently may delay progression of cognitive decline and improve health outcomes. Screening may enhance physician prescribing practices and reduce exposure to harmful medications among these vulnerable patients. Finally, delirium is an important prognostic indicator, and screening patients could provide invaluable information toward the overall clinical picture. Despite all of this, the current literature does not provide sufficient information to support the use of routine screening on admission.220, 41, 5254 Most of the published studies were conducted among elders who stayed in the hospital for more than 48 hours, missing data on the crucial first 48 hours of the hospital course.220, 41, 5254 These studies did not evaluate the impact of unrecognized CI on the hospital course and the majority of these studies were not conducted in the urban and lower socioeconomic status populations of elders that are the most vulnerable to bad health outcomes.220, 41, 5254 Finally, few studies evaluated the impact of delirium superimposed on CI on the hospital course and mortality of elders.220, 41, 5254
With these details in mind, we wanted to explore the impact of CI recognition among patients age 65 years and older admitted to the medical services of an urban, public hospital in Indianapolis to determine the prevalence and the impact of recognized and unrecognized CI on the hospital course of these elders. Furthermore, we examined the role of delirium superimposed on these hospitalized elders with CI.
Patients and Methods
The study was approved by the Indiana University Purdue University at Indianapolis Institutional Review Board (IRB).
Study Setting and Population
The study was conducted on the inpatient general medicine service of Wishard Memorial Hospital (WMH). WMH is a 450‐bed, university‐affiliated, urban, public hospital that is staffed by Indiana University School of Medicine faculty and house staff. It serves a population of approximately 750,000 in Marion County.
Inclusion and Exclusion Criteria
Patients were enrolled in the study based on the following criteria: (1) at least 65 years of age; (2) hospitalized on a medical ward; (3) able to speak English; and (4) have CI at the time of hospital admission (see below). Patients were excluded if they had previously enrolled in the study, were enrolled in another clinical study at the time of admission, or were aphasic or unresponsive at the time of screening.
Cognitive Screening
CI was determined by the Short Portable Mental Status Questionnaire (SPMSQ),55, 56 chosen for its accuracy56 and the fact that it is entirely verbal in administration. In most cases, patients were followed and reassessed daily. Patients having 2 or more errors, indicating a score of 8 or less on the SPMSQ after adjusting for race and education were considered to have cognitive impairment. The SPMSQ is a brief 10‐item screening test with a sensitivity of 86% and specificity 99.0% for dementia among medical inpatients.56 At the time of cognitive screening, delirium was assessed by using the Confusion Assessment Method (CAM).22 This was also done daily in most cases. The CAM22 is a structured instrument that evaluates the 10 symptoms of delirium specified in the Diagnostic and Statistical Manual of Mental Disorders (DSM)‐III‐R: acute onset, fluctuating course, inattention, disorganized thinking, altered level of consciousness, disorientation, memory impairment, perceptual disturbances, psychomotor agitation or retardation, and sleep/wake disturbance. The CAM score is determined by examining the patient, investigating the chart and interviewing the nurse and/or a family member for: (1) acute and fluctuating changes in mental status, (2) inattention, (3) disorganized or incoherent thinking, and (4) altered level of consciousness. A CAM score is considered to be positive if the patient displays both (1) and (2) with at least one of (3) or (4). The CAM diagnosis of delirium was validated against the clinical judgment of a psychiatrist and found to have a sensitivity of 97% and a specificity of 92%.22 A research assistant (RA) was trained for a period of 9 months by a physician as a rater to interview the patient and administer both the SPMSQ and the CAM at the time of admission and then every weekday. When feasible, the RA administered both the SPMSQ and the CAM within the first few hours of hospitalization, and then followed up with our patients each day. More than 70% of our initial cognitive screening occurred in the first 48 hours of hospital admission, and was repeated on a daily basis. In addition to cognitive assessment, the RA reported the presence or absence of Foley catheterization, physical restraints, and tethers during the cognitive assessment. Agreement was obtained from the general internal medicine group practice physicians both to participate in the study and to request screening for CI as part of the recognized admission standard of care among their hospitalized patients aged 65 years and older. The study coordinator was notified of all admissions for patients aged 65 or older by the hospital intranet e‐mail and paging system. Admission notifications were sent by page and e‐mail on an hourly basis from Monday through Friday, 8:00 AM through 5:00 PM. Those admissions occurring between the hours of 5:00 PM and 8:00 AM were sent during the next normal batch notification. Pages and e‐mails for admissions occurring on Saturday and Sunday were sent on Monday morning at 8:00 AM.
Regenstrief Medical Record System at WMH
The computerized Regenstrief Medical Record System (RMRS) is the primary instrument for processing data and monitoring patient and physician activity for Wishard Health System.57, 58 The RMRS is a modular system, composed of Registration and Scheduling, Laboratory, and Pharmacy database modules. The Registration and Scheduling module is used to make all outpatient appointments for the office practices associated with Wishard Health System. The Laboratory module handles all data for all inpatient and outpatient laboratories. This module also produces all laboratory reports and data used for billing. In addition to laboratory data, this module stores coded results and full‐text interpretations of all imaging studies and special procedures. The Pharmacy module contains information on medication orders captured by the computerized physician order enter (CPOE). The Database module stores all the above data by date in a fully‐coded form. Thus, these data are readily retrievable for individual patients by healthcare providers using online terminals. Data for large numbers of patients are retrievable using a locally developed English‐like language called CARE. Patients can be identified either by a certain restriction list (eg, the list of subjects in a study) or by clinical criteria. The RMRS also maintains a number of other databases including diagnoses, vital signs, results of laboratory tests and diagnostic tests, full‐text discharge summaries, preventive health maneuvers, and detailed information on all inpatient and outpatient charges. It contains death certificate information from the Indiana State Board of Health for all registered patients who die in, or outside of, Indiana. Therefore, the RMRS collects and monitors a broad array of physician and patient activity, practice patterns, utilization, diagnostic test finding, and offers a wonderful array of outcome measures.
Other Data Collections
Patient demographics such as age, gender, race, and education level were determined by the RMRS and by information obtained during the time of cognitive screening. Length of hospital stay and 30‐day posthospitalization mortality were obtained from the RMRS. Comorbidity level was measured by reviewing the RMRS and determining each patient's Charlson comorbidity index total score.59, 60 This score was determined using International Statistical Classification of Diseases and Related Health Problems, 9th edition (ICD‐9) codes gathered from 1 year prior to admission until the patient was discharged from the hospital. Anticholinergic medications were determined by using the Anticholinergic Cognitive Burden Scale,61 an expert‐based practical index. The scale was developed based on a review of all published studies from 1996 to 2007 that measured the anticholinergic activities of a drug and its association with cognitive function in older adults. The list of drugs reviewed was presented to an expert interdisciplinary panel that included geriatricians, geriatric pharmacists, geriatric psychiatrists, general physicians, geriatric nurses, and aging brain researchers. The panel categorized each medication into a possible or definite anticholinergic category based on the severity of its cognitive anticholinergic effects.61 A patient who received at least 1 order of a possible or definite anticholinergic during their hospitalization was considered to be an anticholinergic user. Prior recognition of CI was determined by searching the RMRS for any ICD‐9 code (see Appendix) indicative of dementia, Alzheimer disease, or delirium reported at hospital admission, discharge, or during an 1‐year period prior to hospitalization for every patient enrolled in the study. Those patients with documented ICD‐9 codes were felt recognized as having some form of cognitive impairment. Those who had a positive screen but no prior documentation according to ICD‐9 coding, were said to have unrecognized CI.
Analysis
Descriptive statistics were calculated, including percentages for binary categorical variables, and means and standard deviations for continuous variables. Comparisons between groups were based upon Fisher's Exact Tests for binary categorical variables and t tests for continuous variables. When controlling for covariates such as age, gender, race, Charlson comorbidity index, and SPMSQ at screening, group comparisons were made by using logistic regression for binary categorical variables and multiple regression for continuous variables. Since the distributions of length of stay and Charlson comorbidity index were skewed, all statistical tests comparing them across groups were actually performed on their log‐transformed values.
Results
The Prevalence and Recognition of CI
Table 1 describes the demographic characteristic of our study population, which is a reflection of the public and urban nature of our target hospital. Our study assessed the cognitive status of 997 older adults usually (>70% of the time) within 48 hours of their admission to the medical ward of this urban hospital between July of 2006 and March 2008 (see Table 1) and found that 43% of these elders had evidence of CI as determined by a SPMSQ score of 8 points or less. However, 61% of the 424 cognitively impaired elders were not documented or recognized by the electronic medical record system to have cognitive deficit.
| Variable | n | %/Mean (SD) |
|---|---|---|
| ||
| Age (years), mean (SD) | 997 | 74.8 (7.5) |
| Age 85 (%) | 997 | 12.6 |
| Female (%) | 997 | 67.8 |
| African American (%) | 997 | 59.4 |
| Education (years), mean (SD) | 910 | 10.3 (2.8) |
| Education <12 years (%) | 910 | 59.1 |
| Screened within 48 hours of admission (%) | 997 | 73.2 |
| SPMSQ score at screening, mean (SD) | 997 | 7.7 (2.8) |
| Cognitive impairment based on the SPMSQ score 8 (%) | 997 | 42.5 |
The Impact of Unrecognized CI on the Hospital Course
As expected, hospitalized elders with documented CI were older (mean age 79.1 years vs. 76.1 years; P < 0.001) and had worse cognitive function upon screening than those with unrecognized CI (mean SPMSQ 3.4 points vs. 6.3; P < 0.001). Furthermore, CI recognition was influenced by the elders' race and comorbidity (Table 2); a higher percentage of elders with documented CI were African American (69% vs. 54%; P = 0.003) and had less comorbidity (mean Charlson index 1.9 vs. 2.3; P = 0.03). After adjusting for age, gender, race, comorbidity, and cognitive function at screening, our study found no differences between elders with previously recognized CI and those with unrecognized CI in regard to the length of hospital stay (6.7 days vs. 7.5 days; P = 0.59), 30‐day posthospital mortality (4.8% vs. 6.6%; P > 0.2), home discharge (32% vs. 45%; P > 0.7), hospital readmission (19.2% vs.18.8%; P > 0.6), delirium incidence (27% vs. 21%; P > 0.9), and physical restraints (1.8% vs. 1.5%; P > 0.4). We also found that elders with undocumented CI were not more likely to receive definite anticholinergics (33.2% vs. 32.7%; P > 0.9).
| CI Documented | CI Undocumented | P Value | P Value* | |
|---|---|---|---|---|
| ||||
| n (%) | 165 (39) | 259 (61) | n/a | |
| Age, mean (SD) | 79.1 (7.9) | 76.1 (8.0) | <0.001 | |
| Female (%) | 68.5 | 64.5 | 0.40 | |
| African American (%) | 68.5 | 53.7 | <0.01 | |
| SPMSQ at screen, mean (SD) | 3.4 (2.7) | 6.3 (2.1) | <0.001 | |
| Charlson comorbidity index, mean (SD) | 1.9 (1.9) | 2.3 (2.1) | 0.03 | |
| Length of hospital stay, mean (SD) | 6.7 (5.1) | 7.5 (7.1) | 0.49 | 0.59 |
| Survived at 30 days postdischarge (%) | 95.2 | 93.4 | 0.53 | 0.25 |
| Discharged home (%) | 31.5 | 45.2 | 0.01 | 0.74 |
| Readmission within 30 days after discharge home (%) | 19.2 | 18.8 | 0.99 | 0.66 |
| Incidence of delirium (%) | 26.7 | 20.6 | 0.52 | 0.99 |
| Observed with Foley catheter (%) | 43.6 | 27.4 | <0.001 | 0.61 |
| Observed with physical restraint (%) | 1.8 | 1.5 | 0.99 | 0.31 |
| Observed with tethers (%) | 81.8 | 73.8 | 0.06 | 0.58 |
| With at least 1 Ach (%) | 83.6 | 90.7 | 0.03 | 0.22 |
| Possible Ach (%) | 81.2 | 88.4 | 0.05 | 0.31 |
| Definite Ach (%) | 32.7 | 33.2 | 0.99 | 0.64 |
The Impact of Delirium on the Hospital Course of Elders with CI
Among the 424 hospitalized elders with CI, 163 (38%) had delirium at least once during their hospital course and 24% had delirium on the day of hospital discharge. In comparison to elders who had CI but not delirium during their hospitalization (Table 3), those with at least 1 day of delirium had a higher 30‐day posthospitalization mortality risk (8.6% vs. 4.2%; P = 0.09), stayed in the hospital 3.3 additional days (9.2 days vs. 5.9 days; P < 0.001), were less likely to be discharged home (25% vs. 49%; P < 0.001), were more likely to receive a Foley catheterization (52% vs. 23%; P < 0.001), more likely to be physically restrained (4% vs. 0%; P < 0.01), and more likely to receive tethers during their care (89% vs. 69%; P < 0.001). There was no statistically significant difference between the 2 groups in terms of 30‐day hospital readmission rates or in their use of definite anticholinergics (Table 3).
| Delirium+* | Delirium | P value | |
|---|---|---|---|
| |||
| n (%) | 163 (38) | 261 (62) | n/a |
| Age, mean (SD) | 78.4 (8.5) | 76.5 (7.8) | 0.02 |
| Female (%) | 60.1 | 69.7 | 0.05 |
| African American (%) | 64.4 | 56.3 | 0.10 |
| Charlson comorbidity index, mean (SD) | 1.8 (1.9) | 2.3 (2.1) | 0.01 |
| Length of hospital stay, mean (SD) | 9.2 (7.9) | 5.9 (4.9) | <0.001 |
| Survived at 30‐day postdischarge (%) | 91.4 | 95.8 | 0.09 |
| Discharged home (%) | 24.5 | 49.4 | <0.001 |
| Readmission within 30 days after discharge home (%) | 22.5 | 17.8 | 0.50 |
| Observed with Foley catheter (%) | 51.5 | 22.6 | <0.001 |
| Observed with physical restraint (%) | 4.3 | 0.0 | <0.01 |
| Observed with tethers (%) | 89.0 | 69.4 | <0.001 |
| With at least 1 anticholinergic (%) | 83.4 | 90.8 | 0.03 |
| Possible anticholinergic (%) | 80.4 | 88.9 | 0.02 |
| Definite anticholinergic (%) | 36.8 | 30.7 | 0.20 |
Discussion
Our study found that in an urban, public hospital, acute or preexisting CI affects more than one‐third of hospitalized elders admitted to general medical services. Unfortunately, our hospital system does not currently recognize the majority of these vulnerable patients. Our study also found that delirium affects more than one‐third of hospitalized elders with CI during their hospital course. Delirium complicates hospital care by prolonging length of stay and decreasing the probability of surviving and getting discharged home. It leads to high use of Foley catheterization, physical restraints, and tethers.
The high prevalence of CI with and without delirium in our cohort is within the rates reported previously in the literature. It is estimated that the prevalence of CI in hospitalized older adults ranges from 14% to 66%, depending on the method used to measure cognition, the definition of CI, and the type of hospital ward (surgical, medical, and geriatric units).220 One particular study that used a similar cognitive assessment method reported higher prevalence rates for both CI and delirium.11 The study randomly evaluated a sample of 201 patients age 65 and over who were hospitalized for a medical illness and found that 56% of the cohort suffered from CI and among those with CI, 47% had delirium.11 The difference between this finding and our study is most likely due to our sampling technique; more than 70% of our cognitive screening occurred in the first 48 hours of hospital admission whereas the Australian study, in similar enrollment criteria to all of the published studies in this area, excluded patients who were discharged within 48 hours of admission. We believe, however, that by including the first 48 hours of admission in our design, our study provides a more generalizable reflection of the actual acute care experience.
The impact of delirium on the course of hospital care found in our study supports some of the findings from previous studies conducted in the past 2 decades.5, 6, 11 Despite 2 decades of clinical research, delirium continues to increase mortality, hospital stays, and posthospital institutionalization.
We were surprised to find that patients suffering from delirium continue to receive at least 1 definite anticholinergic medication. Such medications are considered inappropriate among patients with any form of cognitive impairment.36, 62 Although the impact of anticholinergic medications on hospitalized outcomes is less well‐described, their use has been suspected to negatively impact long‐term outcomes of cognitive impairment.61, 63 Our study found no difference in the use of anticholinergic medications between those with CI who experienced delirium and those who did not; however, the total burden of anticholinergic medication was not assessed in a quantitative manner. It is still unknown if certain anticholinergic medications or a cumulative effect of anticholinergic medications may impact cognitive or health‐related outcomes in a vulnerable older population with CI.
Although our study reported for the first time in a systematic way the rate of undocumented CI among hospitalized elders found to have CI on admission, we found no impact of such underrecognition on the length of hospital stay, mortality, discharge location, and delirium occurrence. Although the use of anticholinergic medications is not recommended for patients with any form of CI, our results indicate that a significant number of patients with cognitive impairment continue to receive inappropriate medications. CI recognition in the elderly was not shown to have a statistically significant affect on length of stay, cost, or mortality.
Our study has some limitations. First of all, we did not determine the underlying types of CI such as Alzheimer disease, vascular dementia, mild cognitive impairment, or reversible etiology other than delirium. Such a categorization requires posthospital assessment, which was not included in our study design. Second, our delirium incidence rate and delirium impact on hospital outcomes might be very conservative and may underestimate its true prevalence and correlation due to our data collection methods. Despite the fluctuating nature of delirium, our study was not designed to assess the presence of delirium every shift and tried to assess cognitive function on a daily basis throughout the patient's hospitalization. Therefore, the severity and duration of delirium could not be accurately assessed. Our reported rates of use of Foley catheterization, physical restraints, and tethers are also very conservative and we could not determine the appropriateness of these procedures. Our study was conducted in 1 public hospital in an urban city with a higher percentage of African Americans. Thus, our sample is not a true representative sample. However, studies with significant representation of minority groups are not common in the research literature, especially in CI research; we hope to fulfill some of the gaps in the literature regarding the most vulnerable older American population. Finally, we were limited in our use of ICD‐9 coding to determine if patients had previously been recognized by other providers as having CI. ICD‐9 coding, while useful, is not perfect in identifying all if a patient's medical problems. Use of coding to determine whether a patient had been recognized as impaired also does not allow us to determine when the diagnosis was made.
In conclusion, our study evaluated cognitive impairment in hospitalized elders and found that in our cohort of 997 patients, 43% were cognitively impaired on admission. Of those with CI, 61% were not documented or recognized as impaired. We found no statistically significant difference between those with documented CI and those with undocumented CI in terms of length of stay, mortality, home discharge, readmission rates, incidence of delirium, or potential to receive anticholinergics or restraints. Among those with CI, 38% had delirium. Those with delirium experienced increased length of stay, decreased discharge to home, and increased use of Foley catheters and restraints.
- ,.National hospital discharge survey: annual summary, 1994.Vital Health Stat 13.1997;(128):i–v;1–50.
- .The dilemma of delirium: clinical and research controversies regarding diagnosis and evaluation of delirium in hospitalized elderly medical patients.Am J Med.1994;97:278–288.
- ,,.Dementia in medical wards.J Clin Epidemiol.1988;41:123–126.
- ,,,,,.Mental and behavioral disturbances in dementia: findings from the Cache County Study on Memory in Aging.Am J Psychiatry.2000;157:708–714.
- ,,, et al.Randomized, placebo‐controlled, double‐blind clinical trial of sertraline in the treatment of depression complicating Alzheimer's disease: initial results from the Depression in Alzheimer's Disease study.Am J Psychiatry.2000;157:1686–1689.
- ,,.Dementia in elderly persons in a general hospital.Am J Psychiatry.2000;157:704–707.
- ,,,,.A prospective study of the impact of psychiatric comorbidity on length of hospital stays of elderly medical‐surgical inpatients.Psychosomatics.1998;39:273–280.
- ,.Psychiatric comorbidity and length of stay in the general hospital. A critical review of outcome studies.Psychosomatics.1994;35:233–252.
- ,,,.Dementia among medical inpatients. Evaluation of 2000 consecutive admissions.Arch Intern Med.1986;146:1923–1926.
- ,,,.The consequences of non‐cognitive symptoms of dementia in medical hospital departments.Int J Psychiatry Med.2003;33:257–271.
- ,,.Cognitive impairment in medical inpatients. I: Screening for dementia—is history better than mental state?Age Ageing.1997;26:31–35.
- ,,, et al.Acute confusional states in elderly patients treated for femoral neck fracture.J Am Geriatr Soc.1988;36:525–530.
- ,.A prospective study of elderly general surgical patients: II. Post‐operative complications.Age Ageing.1989;18:316–326.
- ,,,.Dementia and depression in elderly medical inpatients.Int Psychogeriatr.2000;12:67–75.
- ,,.[Psychiatric disorders in elderly general hospital patients: incidence and long‐term prognosis].Nervenarzt.1993;64:53–61. [German]
- ,.Delirium and dementia in acute medical admissions of elderly patients in Iceland.Acta Psychiatr Scand.1993;87:123–127.
- ,,,,,.[Psychiatric morbidity in elderly patients admitted to a general hospital. A day‐prevalence study].Med Clin (Barc).1991;97:206–210. [Spanish]
- ,,,,,.Detection of psychiatric disorders in elderly medical inpatients.Age Ageing.1994;23:307–311.
- ,,,.Cognitive impairment, emotional disorder and length of stay of elderly patients in a district general hospital.Br J Med Psychol.1987;60(Pt 2):133–139.
- ,,.An investigation of the components of best nursing practice in the care of acutely ill hospitalized older patients with coincidental dementia: a multi‐method design.J Adv Nurs1999;30:1127–1136.
- American Psychiatric Association.Diagnostic and Statistical Manual of Mental Disorders.4th ed.Washington, DC:American Psychiatric Association;1994.
- ,,,,,.Clarifying confusion: the confusion assessment method. A new method for detection of delirium.Ann Intern Med.1990;113:941–948.
- ,,, et al.Prevalence of cognitive impairment: data from the Indianapolis Study of Health and Aging.Neurology.2001;57:1655–1662.
- ,,.Delirium: a symptom of how hospital care is failing older persons and a window to improve quality of hospital care.Am J Med.1999;106:565–573.
- ,,, et al.A multicomponent intervention to prevent delirium in hospitalized older patients.N Engl J Med.1999;340:669–676.
- ,,,.Iatrogenic causes of falls in hospitalised elderly patients: a case‐control study.Postgrad Med J.2002;78:487–489.
- ,,.A prospective study of delirium in hospitalized elderly.JAMA.1990;263:1097–1101.
- ,.The prognostic significance of delirium in older hospital patients.J Am Geriatr Soc.1997;45:174–178.
- ,.Prognosis of delirium in elderly hospital patients.CMAJ.1993;149:41–46.
- ,,,,.The detection of psychiatric morbidity and its effects on outcome in acute elderly medical admissions.Int J Ger Psych1991;6:861–866.
- ,,.Adverse consequences of hospitalization in the elderly.Soc Sci Med.1982;16:1033–1038.
- ,,, et al.Incidence of adverse events and negligence in hospitalized patients. Results of the Harvard Medical Practice Study I.N Engl J Med.1991;324:370–376.
- ,,,.Delirium in elderly patients: an overview of the state of the science.J Gerontol Nurs.2001;27:12–20.
- ,,,,.A predictive model for delirium in hospitalized elderly medical patients based on admission characteristics.Ann Intern Med.1993;119:474–481.
- ,.Precipitating factors for delirium in hospitalized elderly persons. Predictive model and interrelationship with baseline vulnerability.JAMA.1996;275:852–857.
- ,,,,,.Updating the Beers criteria for potentially inappropriate medication use in older adults: results of a US consensus panel of experts.Arch Intern Med.2003;163:2716–2724.
- ,,, et al.A clinical prediction rule for delirium after elective noncardiac surgery.JAMA.1994;271:134–139.
- ,,, et al.The relationship of postoperative delirium with psychoactive medications.JAMA.1994;272:1518–1522.
- ,,, et al.How do delirium and dementia increase length of stay of elderly general medical inpatients?Psychosomatics.2004;45:235–242.
- ,,,,,.The relationship between a dementia diagnosis, chronic illness, Medicare expenditures, and hospital use.J Am Geriatr Soc.2004;52:187–194.
- ,,, et al.Risk factors for delirium in hospitalized elderly.JAMA.1992;267:827–831.
- ,,,,.Psychological comorbidity and length of stay in the general hospital.Am J Psychiatry.1991;148:324–329.
- ,,, et al.Delirium. The occurrence and persistence of symptoms among elderly hospitalized patients.Arch Intern Med.1992;152:334–340.
- ,,.What happens to medical patients with psychiatric disorder?J Psychosom Res1988;32:541–549.
- ,,,,.[Dementia syndromes and length of stay of elderly patients in internal medicine].Ann Med Interne (Paris).1997;148:424–426. [French]
- ,,, et al.[Hospital discharge planning and length of hospital stay in elderly patients admitted through the emergency department].Rev Epidemiol Sante Publique.1995;43:337–347. [French]
- ,,,,.The effect of dementia on acute care in a geriatric medical unit.Int Psychogeriatr.1992;4:231–239.
- ,,,.Cognitive impairment. Can it predict the course of hospitalized patients?J Am Geriatr Soc.1986;34:579–585.
- ,,,,;US Preventive Services Task Force. Screening for dementia in primary care: a summary of the evidence for the U.S. Preventive Services Task Force.Ann Intern Med.2003;138(11):927–937.
- ,,,.Cognitive impairment in the elderly medically ill: how often is it missed?Int J Geriatr Psychiatry.1993;8:929–937.
- .Recognition of cognitive impairment in elderly medical in‐patients.J R Soc Med.1995;88:183–184.
- ,.Quality indicators for dementia in vulnerable community‐dwelling and hospitalized elders.Ann Intern Med.2001;135:668–676.
- ,,,.Reducing delirium after hip fracture: a randomized trial.J Am Geriatr Soc.2001;49:516–522.
- ,.Prevalence of psychotic symptoms in delirium.Psychosomatics.2000;41:519–522.
- .A short portable mental status questionnaire for the assessment of organic brain deficit in elderly patients.J Am Geriatr Soc.1975;23:433–441.
- ,,,.Short Portable Mental Status Questionnaire as a screening test for dementia and delirium among the elderly.J Am Geriatr Soc.1987;35:412–416.
- ,,, et al.The Regenstrief Medical Record System: a quarter century experience.Int J Med Inform.1999;54:225–253.
- ,,,,,.Factors determining the decision to institutionalize dementing individuals: a prospective study.Gerontologist.1993;33:714–720.
- ,,,,,.Resuscitation: how do we decide? A prospective study of physicians' preferences and the clinical course of hospitalized patients.JAMA.1986;255:1316–1322.
- ,,,,,.Assessing illness severity: does clinical judgment work?J Chronic Dis.1986;39:439–452.
- ,,,,.Impact of anticholinergics on the aging brain: a review and practical application.Aging Health.2008;4(3):311–320.
- ,.Delirium in hospitalized older adults. In: Ham R, Sloane P, Warshaw G, eds.Primary Care Geriatrics: A Case‐Based Approach.5th ed.Philadelphia, PA:Mosby Elsevier;2007:210–218.
- ,,, et al.The association between cognition and histamine‐2 receptor antagonists in African Americans.J Am Geriatr Soc.2007;55(8):1248–1253.
- ,.National hospital discharge survey: annual summary, 1994.Vital Health Stat 13.1997;(128):i–v;1–50.
- .The dilemma of delirium: clinical and research controversies regarding diagnosis and evaluation of delirium in hospitalized elderly medical patients.Am J Med.1994;97:278–288.
- ,,.Dementia in medical wards.J Clin Epidemiol.1988;41:123–126.
- ,,,,,.Mental and behavioral disturbances in dementia: findings from the Cache County Study on Memory in Aging.Am J Psychiatry.2000;157:708–714.
- ,,, et al.Randomized, placebo‐controlled, double‐blind clinical trial of sertraline in the treatment of depression complicating Alzheimer's disease: initial results from the Depression in Alzheimer's Disease study.Am J Psychiatry.2000;157:1686–1689.
- ,,.Dementia in elderly persons in a general hospital.Am J Psychiatry.2000;157:704–707.
- ,,,,.A prospective study of the impact of psychiatric comorbidity on length of hospital stays of elderly medical‐surgical inpatients.Psychosomatics.1998;39:273–280.
- ,.Psychiatric comorbidity and length of stay in the general hospital. A critical review of outcome studies.Psychosomatics.1994;35:233–252.
- ,,,.Dementia among medical inpatients. Evaluation of 2000 consecutive admissions.Arch Intern Med.1986;146:1923–1926.
- ,,,.The consequences of non‐cognitive symptoms of dementia in medical hospital departments.Int J Psychiatry Med.2003;33:257–271.
- ,,.Cognitive impairment in medical inpatients. I: Screening for dementia—is history better than mental state?Age Ageing.1997;26:31–35.
- ,,, et al.Acute confusional states in elderly patients treated for femoral neck fracture.J Am Geriatr Soc.1988;36:525–530.
- ,.A prospective study of elderly general surgical patients: II. Post‐operative complications.Age Ageing.1989;18:316–326.
- ,,,.Dementia and depression in elderly medical inpatients.Int Psychogeriatr.2000;12:67–75.
- ,,.[Psychiatric disorders in elderly general hospital patients: incidence and long‐term prognosis].Nervenarzt.1993;64:53–61. [German]
- ,.Delirium and dementia in acute medical admissions of elderly patients in Iceland.Acta Psychiatr Scand.1993;87:123–127.
- ,,,,,.[Psychiatric morbidity in elderly patients admitted to a general hospital. A day‐prevalence study].Med Clin (Barc).1991;97:206–210. [Spanish]
- ,,,,,.Detection of psychiatric disorders in elderly medical inpatients.Age Ageing.1994;23:307–311.
- ,,,.Cognitive impairment, emotional disorder and length of stay of elderly patients in a district general hospital.Br J Med Psychol.1987;60(Pt 2):133–139.
- ,,.An investigation of the components of best nursing practice in the care of acutely ill hospitalized older patients with coincidental dementia: a multi‐method design.J Adv Nurs1999;30:1127–1136.
- American Psychiatric Association.Diagnostic and Statistical Manual of Mental Disorders.4th ed.Washington, DC:American Psychiatric Association;1994.
- ,,,,,.Clarifying confusion: the confusion assessment method. A new method for detection of delirium.Ann Intern Med.1990;113:941–948.
- ,,, et al.Prevalence of cognitive impairment: data from the Indianapolis Study of Health and Aging.Neurology.2001;57:1655–1662.
- ,,.Delirium: a symptom of how hospital care is failing older persons and a window to improve quality of hospital care.Am J Med.1999;106:565–573.
- ,,, et al.A multicomponent intervention to prevent delirium in hospitalized older patients.N Engl J Med.1999;340:669–676.
- ,,,.Iatrogenic causes of falls in hospitalised elderly patients: a case‐control study.Postgrad Med J.2002;78:487–489.
- ,,.A prospective study of delirium in hospitalized elderly.JAMA.1990;263:1097–1101.
- ,.The prognostic significance of delirium in older hospital patients.J Am Geriatr Soc.1997;45:174–178.
- ,.Prognosis of delirium in elderly hospital patients.CMAJ.1993;149:41–46.
- ,,,,.The detection of psychiatric morbidity and its effects on outcome in acute elderly medical admissions.Int J Ger Psych1991;6:861–866.
- ,,.Adverse consequences of hospitalization in the elderly.Soc Sci Med.1982;16:1033–1038.
- ,,, et al.Incidence of adverse events and negligence in hospitalized patients. Results of the Harvard Medical Practice Study I.N Engl J Med.1991;324:370–376.
- ,,,.Delirium in elderly patients: an overview of the state of the science.J Gerontol Nurs.2001;27:12–20.
- ,,,,.A predictive model for delirium in hospitalized elderly medical patients based on admission characteristics.Ann Intern Med.1993;119:474–481.
- ,.Precipitating factors for delirium in hospitalized elderly persons. Predictive model and interrelationship with baseline vulnerability.JAMA.1996;275:852–857.
- ,,,,,.Updating the Beers criteria for potentially inappropriate medication use in older adults: results of a US consensus panel of experts.Arch Intern Med.2003;163:2716–2724.
- ,,, et al.A clinical prediction rule for delirium after elective noncardiac surgery.JAMA.1994;271:134–139.
- ,,, et al.The relationship of postoperative delirium with psychoactive medications.JAMA.1994;272:1518–1522.
- ,,, et al.How do delirium and dementia increase length of stay of elderly general medical inpatients?Psychosomatics.2004;45:235–242.
- ,,,,,.The relationship between a dementia diagnosis, chronic illness, Medicare expenditures, and hospital use.J Am Geriatr Soc.2004;52:187–194.
- ,,, et al.Risk factors for delirium in hospitalized elderly.JAMA.1992;267:827–831.
- ,,,,.Psychological comorbidity and length of stay in the general hospital.Am J Psychiatry.1991;148:324–329.
- ,,, et al.Delirium. The occurrence and persistence of symptoms among elderly hospitalized patients.Arch Intern Med.1992;152:334–340.
- ,,.What happens to medical patients with psychiatric disorder?J Psychosom Res1988;32:541–549.
- ,,,,.[Dementia syndromes and length of stay of elderly patients in internal medicine].Ann Med Interne (Paris).1997;148:424–426. [French]
- ,,, et al.[Hospital discharge planning and length of hospital stay in elderly patients admitted through the emergency department].Rev Epidemiol Sante Publique.1995;43:337–347. [French]
- ,,,,.The effect of dementia on acute care in a geriatric medical unit.Int Psychogeriatr.1992;4:231–239.
- ,,,.Cognitive impairment. Can it predict the course of hospitalized patients?J Am Geriatr Soc.1986;34:579–585.
- ,,,,;US Preventive Services Task Force. Screening for dementia in primary care: a summary of the evidence for the U.S. Preventive Services Task Force.Ann Intern Med.2003;138(11):927–937.
- ,,,.Cognitive impairment in the elderly medically ill: how often is it missed?Int J Geriatr Psychiatry.1993;8:929–937.
- .Recognition of cognitive impairment in elderly medical in‐patients.J R Soc Med.1995;88:183–184.
- ,.Quality indicators for dementia in vulnerable community‐dwelling and hospitalized elders.Ann Intern Med.2001;135:668–676.
- ,,,.Reducing delirium after hip fracture: a randomized trial.J Am Geriatr Soc.2001;49:516–522.
- ,.Prevalence of psychotic symptoms in delirium.Psychosomatics.2000;41:519–522.
- .A short portable mental status questionnaire for the assessment of organic brain deficit in elderly patients.J Am Geriatr Soc.1975;23:433–441.
- ,,,.Short Portable Mental Status Questionnaire as a screening test for dementia and delirium among the elderly.J Am Geriatr Soc.1987;35:412–416.
- ,,, et al.The Regenstrief Medical Record System: a quarter century experience.Int J Med Inform.1999;54:225–253.
- ,,,,,.Factors determining the decision to institutionalize dementing individuals: a prospective study.Gerontologist.1993;33:714–720.
- ,,,,,.Resuscitation: how do we decide? A prospective study of physicians' preferences and the clinical course of hospitalized patients.JAMA.1986;255:1316–1322.
- ,,,,,.Assessing illness severity: does clinical judgment work?J Chronic Dis.1986;39:439–452.
- ,,,,.Impact of anticholinergics on the aging brain: a review and practical application.Aging Health.2008;4(3):311–320.
- ,.Delirium in hospitalized older adults. In: Ham R, Sloane P, Warshaw G, eds.Primary Care Geriatrics: A Case‐Based Approach.5th ed.Philadelphia, PA:Mosby Elsevier;2007:210–218.
- ,,, et al.The association between cognition and histamine‐2 receptor antagonists in African Americans.J Am Geriatr Soc.2007;55(8):1248–1253.
Copyright © 2010 Society of Hospital Medicine