User login
SILVER SPRING, MD. – Expert advisers expressed little confidence in the Food and Drug Administration estimate that women who undergo a hysterectomy or myomectomy for a presumed fibroid have about a 1 in 350 chance of having an unsuspected sarcoma – the first of several issues the advisers will address at a 2-day meeting on the risks of power morcellation in this setting.
Members of the FDA Obstetrics and Gynecology Devices Advisory Committee said July 10 that they considered the data weak and confusing. Several pointed out that the studies used in the FDA analysis were limited by publication bias, that many patients with fibroids were not included in these estimates, and that better estimates could be obtained with other data sources.
Based on the FDA estimates, clinicians would expect to see about one case of an undiagnosed sarcoma per year, but most do not, one panelist noted.
The estimate is based on an analysis of nine studies of unsuspected uterine sarcomas and leiomyosarcomas (LMSs) treated with a morcellator during a hysterectomy or myomectomy, published between 1990 and 2012. The agency estimated that peritoneal dissemination and/or cancer up-staging to FIGO stage III/IV occurs in about 25%-64% of cases where a uterine sarcoma is morcellated, and that these women have poorer disease-free survival and overall survival than women not treated with morcellation.
The FDA convened the advisory panel meeting to address the benefits, risks, and clinical role of laparoscopic power morcellators (LPMs) in the treatment of women with uterine fibroids, and the use of what the agency described as “potential” mitigation strategies that might reduce or eliminate the risks of disseminating cancerous tissue into the pelvis and abdomen of women with an unsuspected uterine sarcoma or LMS.
In April, the FDA issued a safety communication about the risks of spreading cancerous tissue beyond the uterus when morcellation is used in a patient with an unsuspected uterine sarcoma – and first cited the 1-in-350 risk, notably higher than previously reported rates.The FDA recommended that the use of LPMs during a hysterectomy or myomectomy in women with fibroids should be discouraged and that physicians should discuss alternative treatment options with these patients. But if power morcellation was considered the best option, patients should be informed that their fibroids “may contain unexpected cancerous tissue and that laparoscopic power morcellation may spread the cancer, significantly worsening their prognosis,” the FDA advisory said.
At the meeting, Dr. Ron Yustein, of the FDA Office of Surveillance and Biometrics, said that as of June 22, the voluntary FDA medical device adverse event reporting program had received 21 reports of dissemination of malignancy (mostly LMS) after morcellation of suspected fibroids, including seven reported to be fatal.
Family members of women who had developed disseminated disease after morcellation of an unsuspected sarcoma interrupted the panel discussion to point out they are aware of at least 130 such cases.
LPMs, also cleared for general surgery and urologic indications, are considered class II “moderate risk” devices and were cleared for use based on older devices that were already on the market, with little if any clinical testing in humans. The first LPM for a gynecologic indication was cleared in 1995. LPMs include electromechanical morcellators, which typically have spinning blades to cut up the tissue, and one electrosurgical device that uses radiofrequency energy.
Clinical data are required for class III medical devices to be approved, and the FDA is considering whether to require clinical data for LPMs used in gynecologic indications.
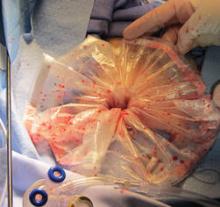
Dr. Craig Sobolewski, who spoke about surgical options for fibroids at the FDA’s invitation, said that the use of specimen collection bags during morcellation dropped because the varied sizes of specimens make it difficult to use bags, and it can be difficult to view a spinning blade through the bag.
The use of collection bags was “minuscule,” but has increased subsequent to the April FDA advisory, said Dr. Sobolewski, chief of the division of minimally invasive gynecologic surgery, Duke University, Durham, N.C.
One “very positive outcome” of this attention will be innovations to address this risk, which will “hopefully enable us to continue to offer this approach ... while simultaneously mitigating the risk,” he said.
The meeting continues July 11, when the panel will address labeling of these devices to reflect the risk of morcellating an unsuspected sarcoma. The panel will not be asked whether these devices should be taken off the market.
Dr. Sobolewski disclosed being a consultant and speaker for Covidien, and is a consultant for and holds stock options in TransEnterix. (Neither company has morcellation products; Covidien manufactures specimen retrieval bags.) None of the panel members has relevant disclosures.
SILVER SPRING, MD. – Expert advisers expressed little confidence in the Food and Drug Administration estimate that women who undergo a hysterectomy or myomectomy for a presumed fibroid have about a 1 in 350 chance of having an unsuspected sarcoma – the first of several issues the advisers will address at a 2-day meeting on the risks of power morcellation in this setting.
Members of the FDA Obstetrics and Gynecology Devices Advisory Committee said July 10 that they considered the data weak and confusing. Several pointed out that the studies used in the FDA analysis were limited by publication bias, that many patients with fibroids were not included in these estimates, and that better estimates could be obtained with other data sources.
Based on the FDA estimates, clinicians would expect to see about one case of an undiagnosed sarcoma per year, but most do not, one panelist noted.
The estimate is based on an analysis of nine studies of unsuspected uterine sarcomas and leiomyosarcomas (LMSs) treated with a morcellator during a hysterectomy or myomectomy, published between 1990 and 2012. The agency estimated that peritoneal dissemination and/or cancer up-staging to FIGO stage III/IV occurs in about 25%-64% of cases where a uterine sarcoma is morcellated, and that these women have poorer disease-free survival and overall survival than women not treated with morcellation.
The FDA convened the advisory panel meeting to address the benefits, risks, and clinical role of laparoscopic power morcellators (LPMs) in the treatment of women with uterine fibroids, and the use of what the agency described as “potential” mitigation strategies that might reduce or eliminate the risks of disseminating cancerous tissue into the pelvis and abdomen of women with an unsuspected uterine sarcoma or LMS.
In April, the FDA issued a safety communication about the risks of spreading cancerous tissue beyond the uterus when morcellation is used in a patient with an unsuspected uterine sarcoma – and first cited the 1-in-350 risk, notably higher than previously reported rates.The FDA recommended that the use of LPMs during a hysterectomy or myomectomy in women with fibroids should be discouraged and that physicians should discuss alternative treatment options with these patients. But if power morcellation was considered the best option, patients should be informed that their fibroids “may contain unexpected cancerous tissue and that laparoscopic power morcellation may spread the cancer, significantly worsening their prognosis,” the FDA advisory said.
At the meeting, Dr. Ron Yustein, of the FDA Office of Surveillance and Biometrics, said that as of June 22, the voluntary FDA medical device adverse event reporting program had received 21 reports of dissemination of malignancy (mostly LMS) after morcellation of suspected fibroids, including seven reported to be fatal.
Family members of women who had developed disseminated disease after morcellation of an unsuspected sarcoma interrupted the panel discussion to point out they are aware of at least 130 such cases.
LPMs, also cleared for general surgery and urologic indications, are considered class II “moderate risk” devices and were cleared for use based on older devices that were already on the market, with little if any clinical testing in humans. The first LPM for a gynecologic indication was cleared in 1995. LPMs include electromechanical morcellators, which typically have spinning blades to cut up the tissue, and one electrosurgical device that uses radiofrequency energy.
Clinical data are required for class III medical devices to be approved, and the FDA is considering whether to require clinical data for LPMs used in gynecologic indications.
Dr. Craig Sobolewski, who spoke about surgical options for fibroids at the FDA’s invitation, said that the use of specimen collection bags during morcellation dropped because the varied sizes of specimens make it difficult to use bags, and it can be difficult to view a spinning blade through the bag.
The use of collection bags was “minuscule,” but has increased subsequent to the April FDA advisory, said Dr. Sobolewski, chief of the division of minimally invasive gynecologic surgery, Duke University, Durham, N.C.
One “very positive outcome” of this attention will be innovations to address this risk, which will “hopefully enable us to continue to offer this approach ... while simultaneously mitigating the risk,” he said.
The meeting continues July 11, when the panel will address labeling of these devices to reflect the risk of morcellating an unsuspected sarcoma. The panel will not be asked whether these devices should be taken off the market.
Dr. Sobolewski disclosed being a consultant and speaker for Covidien, and is a consultant for and holds stock options in TransEnterix. (Neither company has morcellation products; Covidien manufactures specimen retrieval bags.) None of the panel members has relevant disclosures.
SILVER SPRING, MD. – Expert advisers expressed little confidence in the Food and Drug Administration estimate that women who undergo a hysterectomy or myomectomy for a presumed fibroid have about a 1 in 350 chance of having an unsuspected sarcoma – the first of several issues the advisers will address at a 2-day meeting on the risks of power morcellation in this setting.
Members of the FDA Obstetrics and Gynecology Devices Advisory Committee said July 10 that they considered the data weak and confusing. Several pointed out that the studies used in the FDA analysis were limited by publication bias, that many patients with fibroids were not included in these estimates, and that better estimates could be obtained with other data sources.
Based on the FDA estimates, clinicians would expect to see about one case of an undiagnosed sarcoma per year, but most do not, one panelist noted.
The estimate is based on an analysis of nine studies of unsuspected uterine sarcomas and leiomyosarcomas (LMSs) treated with a morcellator during a hysterectomy or myomectomy, published between 1990 and 2012. The agency estimated that peritoneal dissemination and/or cancer up-staging to FIGO stage III/IV occurs in about 25%-64% of cases where a uterine sarcoma is morcellated, and that these women have poorer disease-free survival and overall survival than women not treated with morcellation.
The FDA convened the advisory panel meeting to address the benefits, risks, and clinical role of laparoscopic power morcellators (LPMs) in the treatment of women with uterine fibroids, and the use of what the agency described as “potential” mitigation strategies that might reduce or eliminate the risks of disseminating cancerous tissue into the pelvis and abdomen of women with an unsuspected uterine sarcoma or LMS.
In April, the FDA issued a safety communication about the risks of spreading cancerous tissue beyond the uterus when morcellation is used in a patient with an unsuspected uterine sarcoma – and first cited the 1-in-350 risk, notably higher than previously reported rates.The FDA recommended that the use of LPMs during a hysterectomy or myomectomy in women with fibroids should be discouraged and that physicians should discuss alternative treatment options with these patients. But if power morcellation was considered the best option, patients should be informed that their fibroids “may contain unexpected cancerous tissue and that laparoscopic power morcellation may spread the cancer, significantly worsening their prognosis,” the FDA advisory said.
At the meeting, Dr. Ron Yustein, of the FDA Office of Surveillance and Biometrics, said that as of June 22, the voluntary FDA medical device adverse event reporting program had received 21 reports of dissemination of malignancy (mostly LMS) after morcellation of suspected fibroids, including seven reported to be fatal.
Family members of women who had developed disseminated disease after morcellation of an unsuspected sarcoma interrupted the panel discussion to point out they are aware of at least 130 such cases.
LPMs, also cleared for general surgery and urologic indications, are considered class II “moderate risk” devices and were cleared for use based on older devices that were already on the market, with little if any clinical testing in humans. The first LPM for a gynecologic indication was cleared in 1995. LPMs include electromechanical morcellators, which typically have spinning blades to cut up the tissue, and one electrosurgical device that uses radiofrequency energy.
Clinical data are required for class III medical devices to be approved, and the FDA is considering whether to require clinical data for LPMs used in gynecologic indications.
Dr. Craig Sobolewski, who spoke about surgical options for fibroids at the FDA’s invitation, said that the use of specimen collection bags during morcellation dropped because the varied sizes of specimens make it difficult to use bags, and it can be difficult to view a spinning blade through the bag.
The use of collection bags was “minuscule,” but has increased subsequent to the April FDA advisory, said Dr. Sobolewski, chief of the division of minimally invasive gynecologic surgery, Duke University, Durham, N.C.
One “very positive outcome” of this attention will be innovations to address this risk, which will “hopefully enable us to continue to offer this approach ... while simultaneously mitigating the risk,” he said.
The meeting continues July 11, when the panel will address labeling of these devices to reflect the risk of morcellating an unsuspected sarcoma. The panel will not be asked whether these devices should be taken off the market.
Dr. Sobolewski disclosed being a consultant and speaker for Covidien, and is a consultant for and holds stock options in TransEnterix. (Neither company has morcellation products; Covidien manufactures specimen retrieval bags.) None of the panel members has relevant disclosures.