User login
Acne vulgaris (AV) is a common skin disease that usually presents in adolescence and can persist into adulthood. Some cases may start in adulthood, especially in women. Acne vulgaris remains a challenge to treat successfully, both in teenagers and adults. Unrealistic expectations that therapy will rapidly clear and sustain clearance of AV completely can lead to incomplete adherence or complete cessation of treatment.1-4 Local tolerability reactions also may decrease adherence to topical medications. Suboptimal adherence to medications for AV is one of the major reasons for treatment failure.5 Acne vulgaris can strongly influence psychological well-being and self-esteem.6 In general, severe AV causes more psychological distress, but the adverse emotional impact of AV can be independent of its severity.7
An effective relationship between the patient and his/her physician and staff is believed to be important in setting realistic expectations, optimizing adherence, and achieving a positive therapeutic outcome. One component related to setting reasonable expectations is the discussion about when the patient may begin to visibly perceive that the treatment regimen is working. This article evaluates the time course of a clinically meaningful response using pivotal trial data with clindamycin phosphate 1.2%–benzoyl peroxide 3.75% (clindamycin-BP 3.75%) gel for treatment of AV.
Are data available that evaluate the time course of a clinically relevant response to treatment of AV?
Unfortunately, data on what might be perceived as a clinically meaningful improvement in AV and how long it might take to achieve this treatment effect are limited. A meta-analysis of more than 4000 patients with moderate to severe AV suggested that a 10% to 20% difference in acne lesion counts from baseline as compared to a subsequent designated time point was clinically relevant.8 A review of 24 comparative studies of patients with mild to moderate AV used a primary outcome parameter of a 25% reduction in mean inflammatory lesion count to evaluate time to onset of action (TOA) to achieve a clinically meaningful benefit.9 This outcome was based on a previously identified threshold of clinical relevance and the authors’ clinical experience in a patient population with milder AV. In this same analysis, a difference of greater than 4 days between the active group and the vehicle group was considered to be relevant to the patient.9
A faster onset of visible improvement as perceived by the patient should be more desirable and is likely to improve treatment adherence, as long as it is not counterbalanced by an increase in adverse events.
What is meant by TOA?
Time to onset of action refers to the duration required to achieve a 25% mean lesion count reduction from baseline, which is believed to correlate with the time point at which many patients would be able to perceive visible improvement when viewing their full face. Therefore, TOA represents an attempt to correlate data that is quantitative (based on lesion count reduction) with what is likely to be the average time that a patient may qualitatively observe an initial visible improvement in their AV. This concept may be useful as a tool when communicating with AV patients but should not be used in a way that will overpromise and underdeliver; rather, it is a guide for discussion with the patient and with a parent or guardian when applicable.
Consistent with the comparative AV study analysis that evaluated TOA, a linear course of lesion reductions between the provided time intervals was assumed. In this linear model, the TOA was calculated using the 2 extracted lesion count values between which the 25% lesion reduction was achieved as well as their corresponding given time points.9 Differences between the results in the active and vehicle study arms were calculated for a number of determinants.
How was pivotal trial data with clindamycin-BP 3.75% gel used to assess TOA?
A total of 498 patients with moderate to severe AV were randomized (1:1) to receive clindamycin-BP 3.75% gel or vehicle in a multicenter, double-blind, controlled, 12-week, 2-arm study.10 Before randomization, patients were stratified by acne severity based on a static Evaluator’s Global Severity Score (EGSS) ranging from 0 (clear) to 5 (very severe). Specifically, moderate AV (EGSS of 3) was described as predominantly noninflammatory lesions with evidence of multiple inflammatory lesions; several to many comedones, papules, and pustules; and no more than 1 small nodulocystic lesion. Severe AV (EGSS of 4) was characterized by inflammatory lesions; numerous comedones, papules, and pustules; and possibly a few nodulocystic lesions.10
Male and female patients aged 12 to 40 years with moderate to severe AV—defined as 20 to 40 inflammatory lesions (papules, pustules, nodules), 20 to 100 noninflammatory lesions (comedones), and no more than 2 nodules—were included in the study. Standard washout periods were required for patients using prior prescription and over-the-counter acne treatments.10
Efficacy evaluations included inflammatory and noninflammatory lesion counts and EGSS at screening, baseline, and during treatment (weeks 4, 8, and 12).10 Primary efficacy end points included absolute change in mean inflammatory and noninflammatory lesion counts and the proportion of patients who achieved at least a 2-grade reduction in EGSS from baseline to week 12 (treatment success at end of study). Secondary efficacy end points included mean percentage change from baseline to week 12 in inflammatory and noninflammatory lesion counts and the proportion of patients who considered themselves clear or almost clear at week 12.10
After 12 weeks of daily treatment, inflammatory and noninflammatory lesion counts decreased by a mean of 60.4% and 51.8%, respectively, with clindamycin-BP 3.75% gel compared to 31.3% and 27.6%, respectively, with vehicle (both P<.001). At weeks 4, 8, and 12, the difference in inflammatory and noninflammatory lesion counts for the active treatment was 17.4%, 24.8%, and 29.1%, respectively, and 8.1%, 19.8%, and 24.2%, respectively, for vehicle.10
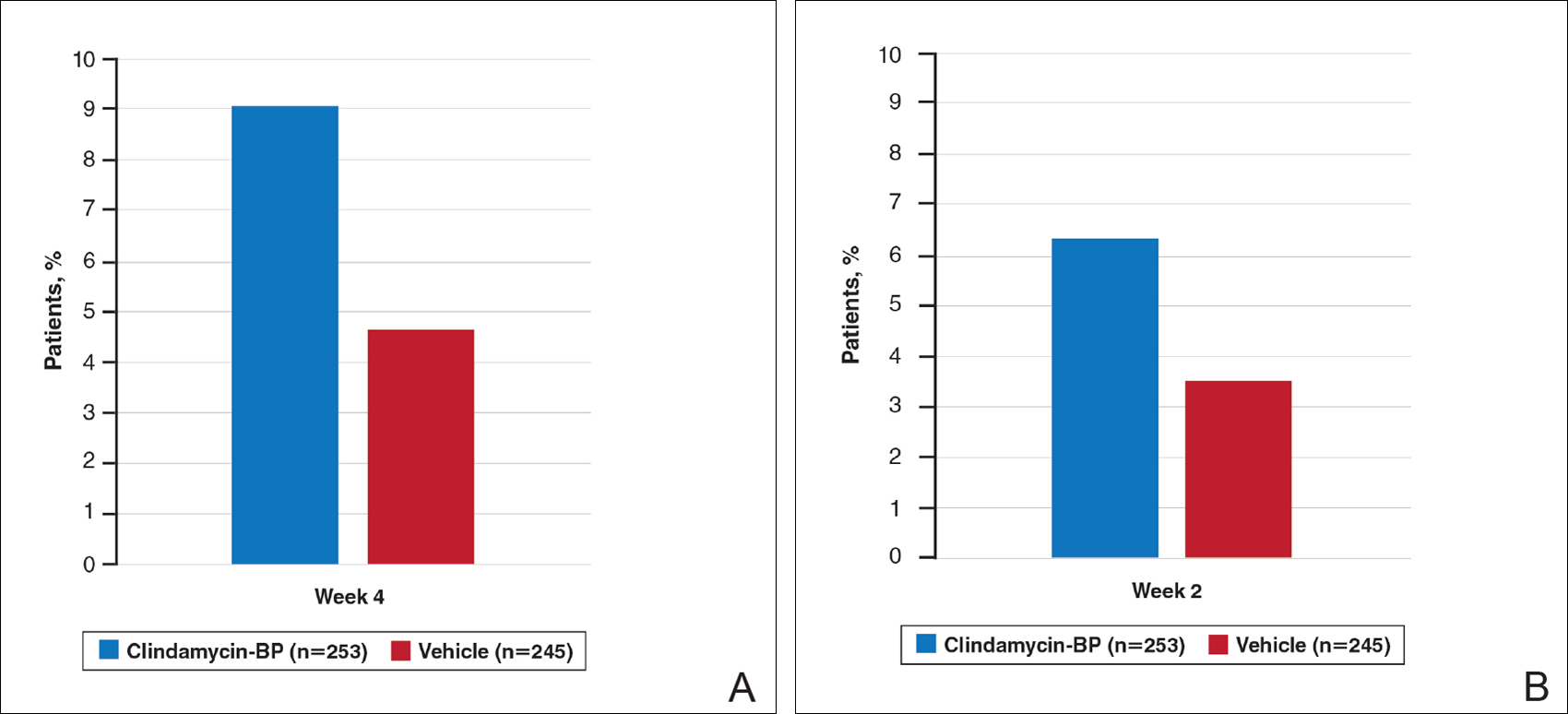
Treatment success (at least a 2-grade improvement in EGSS) was achieved by 9.1% of patients using clindamycin-BP 3.75% gel compared to 4.6% using vehicle by week 4. Additionally, 6.3% of patients considered their AV as clear or almost clear compared to 3.5% with vehicle at week 2 (Figure 1).10
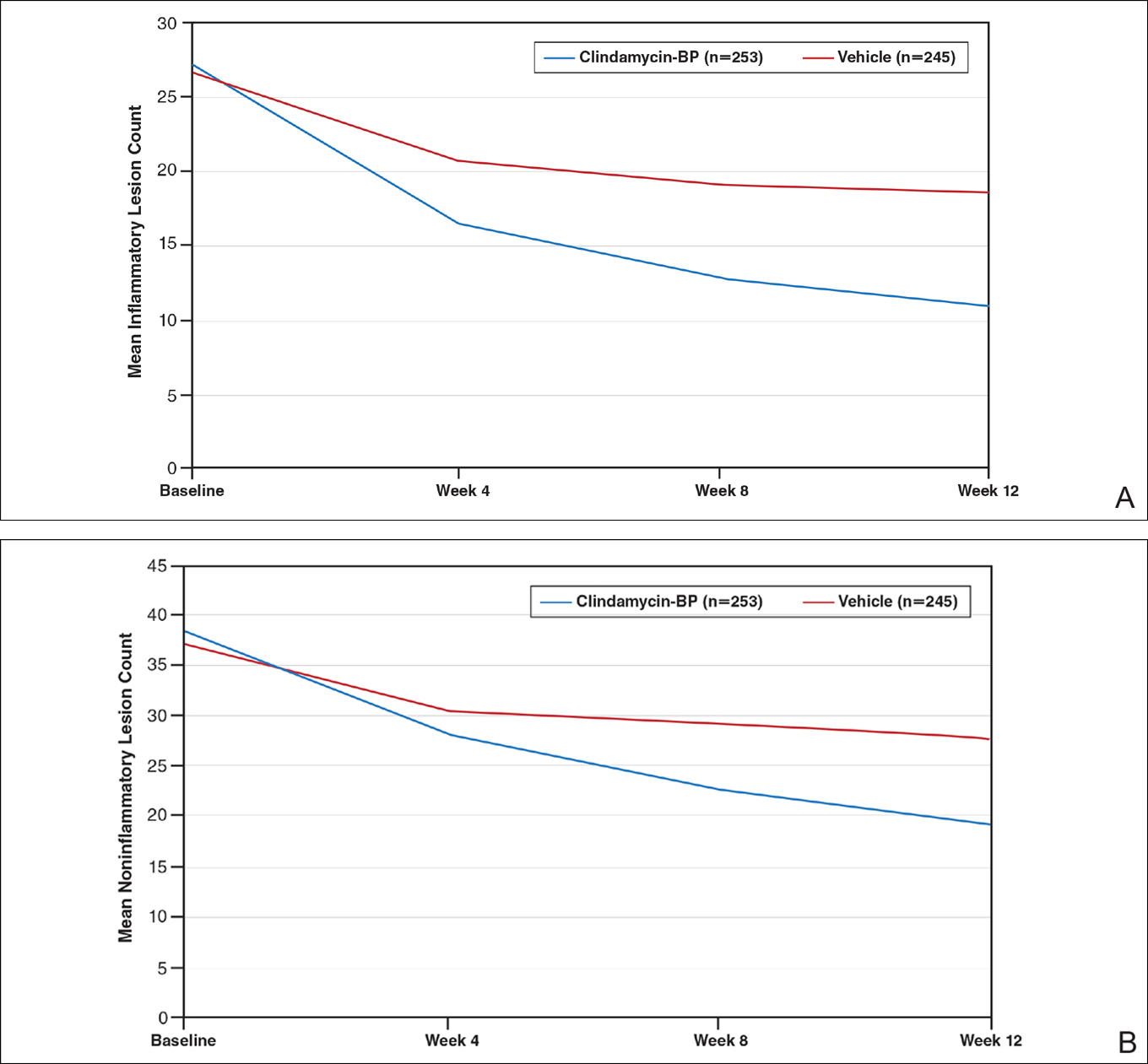
This analysis represents the first attempt to evaluate and report TOA results with clindamycin-BP 3.75% gel. Time to onset of action for inflammatory lesions treated with clindamycin-BP 3.75% gel was calculated as 2.5 weeks versus 6.2 weeks for vehicle (Figure 2A). Time to onset of action for noninflammatory lesions was 3.7 weeks with clindamycin-BP 3.75% gel versus 8.6 weeks with vehicle (Figure 2B). The difference in TOA between the active and vehicle study groups was 3.7 weeks and 4.9 weeks, respectively. In addition, among actively treated patients, TOA was shorter in females (2.1 weeks) than in males (2.6 weeks) and in moderate AV (2.5 weeks) compared to severe AV (3.0 weeks).
Comment
Differences in lesion counts between clindamycin-BP 3.75% gel and vehicle suggest a clinically relevant benefit in favor of active treatment with both inflammatory and noninflammatory lesions. Nearly twice as many patients were rated as treatment successes using EGSS by week 4 or clear or almost clear as early as week 2 compared to the vehicle group.10 However, these data are suggested as an overall guide but do not provide adequate guidance on when visible improvement may start to be evident in a given patient.
The analysis reported here shows a TOA of 2.5 weeks with clindamycin-BP 3.75% gel for inflammatory lesions, approximately 4 weeks faster than with the vehicle. In most cases, a reduction in inflammatory lesions is more likely to have a greater impact on patient perception of TOA. Unless a patient is aware or focused enough to actively distinguish visibly between inflammatory and noninflammatory (comedonal) AV lesions, their eye is more likely to be drawn initially to reduction in inflammatory lesions, which are erythematous and more visible at a greater viewing distance. Although noninflammatory AV lesions usually require closer inspection to visualize them (especially closed comedones), they are often slower to respond to treatment. Analysis of the pivotal trial data reports a longer TOA with clindamycin-BP 3.75% gel for noninflammatory lesions (3.7 weeks) versus inflammatory lesions (2.5 weeks).
As expected, TOA was shorter in patients with moderate AV than severe AV (2.5 weeks vs 3.0 weeks). Time to onset of action also was shorter in females overall. It is unclear why we see gender differences in acne studies. A number of reasons have been suggested, including differences in AV pathophysiology and/or treatment adherence.11,12 Greater efficacy of clindamycin-BP 3.75% gel in females compared with males has already been reported, and better overall efficacy leading to a shorter TOA has been noted by others.13
There are limitations with this analysis. First, it is not possible to assess the contributions from each of the monads to the efficacy of clindamycin-BP 3.75% gel or TOA. Also, the data extraction method used assumes a linear progression model during the provided time points and was used to provide some comparison with calculations for other combination products.9 Although no strong deviations from the linear model are likely, calculations of TOA using other methodologies may give different results. The definition of a clinically meaningful benefit, defined here as a 25% reduction in the mean lesion count, has been used as a guide, but it has not been validated in clinical practice. It also is important to recognize that the initial visible perception of improvement of AV is likely to differ based on interpatient variability; that is, how different individuals perceive improvement. It also may be affected by differences in baseline severity of AV among different patients. Additionally, the TOA reflects an average duration of time, so it should not be described to patients as a suggestion of when they will definitely see visible improvement in their AV.
Conclusion
Unrealistic expectations of acne therapy or poor tolerability can lead to low adherence and poor clinical outcomes.1-4 The data on TOA reported here suggests that a clinically meaningful benefit with clindamycin-BP 3.75% gel may be seen in some patients within 2 to 3 weeks and maybe sooner in females or those with milder disease; however, longer durations may be required in some patients. This information can help clinicians and their staff in providing reasonable expectations and stress the importance of encouraging patients about the need to adhere to treatment.
Acknowledgments
The author thanks Brian Bulley, MSc (Inergy Limited, Lindfield, West Sussex, United Kingdom), for publication support. Valeant Pharmaceuticals North America, LLC, funded Inergy’s activities pertaining to this analysis. The author did not receive funding or any form of compensation for authorship of this publication.
- Krakowski AC, Stendardo S, Eichenfield LF. Practical considerations in acne treatment and the clinical impact of topical combination therapy. Pediatr Dermatol. 2008;25(suppl 1):1-14.
- Yentzer BA, Ade RA, Fountain JM, et al. Simplifying regimens promotes greater adherence and outcomes with topical acne medications: a randomized controlled trial. Cutis. 2010;86:103-108.
- Zaghloul SS, Cunliffe WJ, Goodfield MJ. Objective assessment of compliance with treatments in acne. Br J Dermatol. 2005;152:1015-1021.
- Snyder S, Crandell I, Davis SA, et al. Medical adherence to acne therapy: a systematic review. Am J Clin Dermatol. 2014;15:87-94.
- Miyachi Y, Hayashi N, Furukawa F, et al. Acne management in Japan: study of patient adherence. Dermatology. 2011;223:174-181.
- Zauli S, Caracciolo S, Borghi A, et al. Which factors influence quality of life in acne patients? J Eur Acad Dermatol Venereol. 2014;28:46-50.
- Mulder MM, Sigurdsson V, van Zuuren EJ, et al. Psychosocial impact of acne vulgaris. evaluation of the relation between a change in clinical acne severity and psychosocial state. Dermatology. 2001;203:124-130.
- Gerlinger C, Stadtler G, Gotzelmann R, et al. A noninferiority margin for acne lesion counts. Drug Inf J. 2008;42:607-615.
- Jacobs A, Starke G, Rosumeck S, et al. Systematic review on the rapidity of the onset of action of topical treatments in the therapy of mild-to-moderate acne vulgaris. Br J Dermatol. 2014;170:557-564.
- Pariser DM, Rich P, Cook-Bolden FE, et al. An aqueous gel fixed combination of clindamycin phosphate 1.2% and benzoyl peroxide 3.75% for the once-daily treatment of moderate to severe acne vulgaris. J Drugs Dermatol. 2014;13:611-617.
- Tanghetti E, Harper JC, Oefelein MG. The efficacy and tolerability of dapsone 5% gel in female vs male patients with facial acne vulgaris: gender as a clinically relevant outcome variable. J Drugs Dermatol. 2012;11:1417-1421.
- Lott R, Taylor SL, O’Neill JL, et al. Medication adherence among acne patients: a review. J Cosmet Dermatol. 2010;9:160-166.
- Harper JC. The efficacy and tolerability of a fixed combination clindamycin (1.2%) and benzoyl peroxide (3.75%) aqueous gel in patients with facial acne vulgaris: gender as a clinically relevant outcome variable. J Drugs Dermatol. 2015;14:381-384.
Acne vulgaris (AV) is a common skin disease that usually presents in adolescence and can persist into adulthood. Some cases may start in adulthood, especially in women. Acne vulgaris remains a challenge to treat successfully, both in teenagers and adults. Unrealistic expectations that therapy will rapidly clear and sustain clearance of AV completely can lead to incomplete adherence or complete cessation of treatment.1-4 Local tolerability reactions also may decrease adherence to topical medications. Suboptimal adherence to medications for AV is one of the major reasons for treatment failure.5 Acne vulgaris can strongly influence psychological well-being and self-esteem.6 In general, severe AV causes more psychological distress, but the adverse emotional impact of AV can be independent of its severity.7
An effective relationship between the patient and his/her physician and staff is believed to be important in setting realistic expectations, optimizing adherence, and achieving a positive therapeutic outcome. One component related to setting reasonable expectations is the discussion about when the patient may begin to visibly perceive that the treatment regimen is working. This article evaluates the time course of a clinically meaningful response using pivotal trial data with clindamycin phosphate 1.2%–benzoyl peroxide 3.75% (clindamycin-BP 3.75%) gel for treatment of AV.
Are data available that evaluate the time course of a clinically relevant response to treatment of AV?
Unfortunately, data on what might be perceived as a clinically meaningful improvement in AV and how long it might take to achieve this treatment effect are limited. A meta-analysis of more than 4000 patients with moderate to severe AV suggested that a 10% to 20% difference in acne lesion counts from baseline as compared to a subsequent designated time point was clinically relevant.8 A review of 24 comparative studies of patients with mild to moderate AV used a primary outcome parameter of a 25% reduction in mean inflammatory lesion count to evaluate time to onset of action (TOA) to achieve a clinically meaningful benefit.9 This outcome was based on a previously identified threshold of clinical relevance and the authors’ clinical experience in a patient population with milder AV. In this same analysis, a difference of greater than 4 days between the active group and the vehicle group was considered to be relevant to the patient.9
A faster onset of visible improvement as perceived by the patient should be more desirable and is likely to improve treatment adherence, as long as it is not counterbalanced by an increase in adverse events.
What is meant by TOA?
Time to onset of action refers to the duration required to achieve a 25% mean lesion count reduction from baseline, which is believed to correlate with the time point at which many patients would be able to perceive visible improvement when viewing their full face. Therefore, TOA represents an attempt to correlate data that is quantitative (based on lesion count reduction) with what is likely to be the average time that a patient may qualitatively observe an initial visible improvement in their AV. This concept may be useful as a tool when communicating with AV patients but should not be used in a way that will overpromise and underdeliver; rather, it is a guide for discussion with the patient and with a parent or guardian when applicable.
Consistent with the comparative AV study analysis that evaluated TOA, a linear course of lesion reductions between the provided time intervals was assumed. In this linear model, the TOA was calculated using the 2 extracted lesion count values between which the 25% lesion reduction was achieved as well as their corresponding given time points.9 Differences between the results in the active and vehicle study arms were calculated for a number of determinants.
How was pivotal trial data with clindamycin-BP 3.75% gel used to assess TOA?
A total of 498 patients with moderate to severe AV were randomized (1:1) to receive clindamycin-BP 3.75% gel or vehicle in a multicenter, double-blind, controlled, 12-week, 2-arm study.10 Before randomization, patients were stratified by acne severity based on a static Evaluator’s Global Severity Score (EGSS) ranging from 0 (clear) to 5 (very severe). Specifically, moderate AV (EGSS of 3) was described as predominantly noninflammatory lesions with evidence of multiple inflammatory lesions; several to many comedones, papules, and pustules; and no more than 1 small nodulocystic lesion. Severe AV (EGSS of 4) was characterized by inflammatory lesions; numerous comedones, papules, and pustules; and possibly a few nodulocystic lesions.10
Male and female patients aged 12 to 40 years with moderate to severe AV—defined as 20 to 40 inflammatory lesions (papules, pustules, nodules), 20 to 100 noninflammatory lesions (comedones), and no more than 2 nodules—were included in the study. Standard washout periods were required for patients using prior prescription and over-the-counter acne treatments.10
Efficacy evaluations included inflammatory and noninflammatory lesion counts and EGSS at screening, baseline, and during treatment (weeks 4, 8, and 12).10 Primary efficacy end points included absolute change in mean inflammatory and noninflammatory lesion counts and the proportion of patients who achieved at least a 2-grade reduction in EGSS from baseline to week 12 (treatment success at end of study). Secondary efficacy end points included mean percentage change from baseline to week 12 in inflammatory and noninflammatory lesion counts and the proportion of patients who considered themselves clear or almost clear at week 12.10
After 12 weeks of daily treatment, inflammatory and noninflammatory lesion counts decreased by a mean of 60.4% and 51.8%, respectively, with clindamycin-BP 3.75% gel compared to 31.3% and 27.6%, respectively, with vehicle (both P<.001). At weeks 4, 8, and 12, the difference in inflammatory and noninflammatory lesion counts for the active treatment was 17.4%, 24.8%, and 29.1%, respectively, and 8.1%, 19.8%, and 24.2%, respectively, for vehicle.10
Treatment success (at least a 2-grade improvement in EGSS) was achieved by 9.1% of patients using clindamycin-BP 3.75% gel compared to 4.6% using vehicle by week 4. Additionally, 6.3% of patients considered their AV as clear or almost clear compared to 3.5% with vehicle at week 2 (Figure 1).10

This analysis represents the first attempt to evaluate and report TOA results with clindamycin-BP 3.75% gel. Time to onset of action for inflammatory lesions treated with clindamycin-BP 3.75% gel was calculated as 2.5 weeks versus 6.2 weeks for vehicle (Figure 2A). Time to onset of action for noninflammatory lesions was 3.7 weeks with clindamycin-BP 3.75% gel versus 8.6 weeks with vehicle (Figure 2B). The difference in TOA between the active and vehicle study groups was 3.7 weeks and 4.9 weeks, respectively. In addition, among actively treated patients, TOA was shorter in females (2.1 weeks) than in males (2.6 weeks) and in moderate AV (2.5 weeks) compared to severe AV (3.0 weeks).

Comment
Differences in lesion counts between clindamycin-BP 3.75% gel and vehicle suggest a clinically relevant benefit in favor of active treatment with both inflammatory and noninflammatory lesions. Nearly twice as many patients were rated as treatment successes using EGSS by week 4 or clear or almost clear as early as week 2 compared to the vehicle group.10 However, these data are suggested as an overall guide but do not provide adequate guidance on when visible improvement may start to be evident in a given patient.
The analysis reported here shows a TOA of 2.5 weeks with clindamycin-BP 3.75% gel for inflammatory lesions, approximately 4 weeks faster than with the vehicle. In most cases, a reduction in inflammatory lesions is more likely to have a greater impact on patient perception of TOA. Unless a patient is aware or focused enough to actively distinguish visibly between inflammatory and noninflammatory (comedonal) AV lesions, their eye is more likely to be drawn initially to reduction in inflammatory lesions, which are erythematous and more visible at a greater viewing distance. Although noninflammatory AV lesions usually require closer inspection to visualize them (especially closed comedones), they are often slower to respond to treatment. Analysis of the pivotal trial data reports a longer TOA with clindamycin-BP 3.75% gel for noninflammatory lesions (3.7 weeks) versus inflammatory lesions (2.5 weeks).
As expected, TOA was shorter in patients with moderate AV than severe AV (2.5 weeks vs 3.0 weeks). Time to onset of action also was shorter in females overall. It is unclear why we see gender differences in acne studies. A number of reasons have been suggested, including differences in AV pathophysiology and/or treatment adherence.11,12 Greater efficacy of clindamycin-BP 3.75% gel in females compared with males has already been reported, and better overall efficacy leading to a shorter TOA has been noted by others.13
There are limitations with this analysis. First, it is not possible to assess the contributions from each of the monads to the efficacy of clindamycin-BP 3.75% gel or TOA. Also, the data extraction method used assumes a linear progression model during the provided time points and was used to provide some comparison with calculations for other combination products.9 Although no strong deviations from the linear model are likely, calculations of TOA using other methodologies may give different results. The definition of a clinically meaningful benefit, defined here as a 25% reduction in the mean lesion count, has been used as a guide, but it has not been validated in clinical practice. It also is important to recognize that the initial visible perception of improvement of AV is likely to differ based on interpatient variability; that is, how different individuals perceive improvement. It also may be affected by differences in baseline severity of AV among different patients. Additionally, the TOA reflects an average duration of time, so it should not be described to patients as a suggestion of when they will definitely see visible improvement in their AV.
Conclusion
Unrealistic expectations of acne therapy or poor tolerability can lead to low adherence and poor clinical outcomes.1-4 The data on TOA reported here suggests that a clinically meaningful benefit with clindamycin-BP 3.75% gel may be seen in some patients within 2 to 3 weeks and maybe sooner in females or those with milder disease; however, longer durations may be required in some patients. This information can help clinicians and their staff in providing reasonable expectations and stress the importance of encouraging patients about the need to adhere to treatment.
Acknowledgments
The author thanks Brian Bulley, MSc (Inergy Limited, Lindfield, West Sussex, United Kingdom), for publication support. Valeant Pharmaceuticals North America, LLC, funded Inergy’s activities pertaining to this analysis. The author did not receive funding or any form of compensation for authorship of this publication.
Acne vulgaris (AV) is a common skin disease that usually presents in adolescence and can persist into adulthood. Some cases may start in adulthood, especially in women. Acne vulgaris remains a challenge to treat successfully, both in teenagers and adults. Unrealistic expectations that therapy will rapidly clear and sustain clearance of AV completely can lead to incomplete adherence or complete cessation of treatment.1-4 Local tolerability reactions also may decrease adherence to topical medications. Suboptimal adherence to medications for AV is one of the major reasons for treatment failure.5 Acne vulgaris can strongly influence psychological well-being and self-esteem.6 In general, severe AV causes more psychological distress, but the adverse emotional impact of AV can be independent of its severity.7
An effective relationship between the patient and his/her physician and staff is believed to be important in setting realistic expectations, optimizing adherence, and achieving a positive therapeutic outcome. One component related to setting reasonable expectations is the discussion about when the patient may begin to visibly perceive that the treatment regimen is working. This article evaluates the time course of a clinically meaningful response using pivotal trial data with clindamycin phosphate 1.2%–benzoyl peroxide 3.75% (clindamycin-BP 3.75%) gel for treatment of AV.
Are data available that evaluate the time course of a clinically relevant response to treatment of AV?
Unfortunately, data on what might be perceived as a clinically meaningful improvement in AV and how long it might take to achieve this treatment effect are limited. A meta-analysis of more than 4000 patients with moderate to severe AV suggested that a 10% to 20% difference in acne lesion counts from baseline as compared to a subsequent designated time point was clinically relevant.8 A review of 24 comparative studies of patients with mild to moderate AV used a primary outcome parameter of a 25% reduction in mean inflammatory lesion count to evaluate time to onset of action (TOA) to achieve a clinically meaningful benefit.9 This outcome was based on a previously identified threshold of clinical relevance and the authors’ clinical experience in a patient population with milder AV. In this same analysis, a difference of greater than 4 days between the active group and the vehicle group was considered to be relevant to the patient.9
A faster onset of visible improvement as perceived by the patient should be more desirable and is likely to improve treatment adherence, as long as it is not counterbalanced by an increase in adverse events.
What is meant by TOA?
Time to onset of action refers to the duration required to achieve a 25% mean lesion count reduction from baseline, which is believed to correlate with the time point at which many patients would be able to perceive visible improvement when viewing their full face. Therefore, TOA represents an attempt to correlate data that is quantitative (based on lesion count reduction) with what is likely to be the average time that a patient may qualitatively observe an initial visible improvement in their AV. This concept may be useful as a tool when communicating with AV patients but should not be used in a way that will overpromise and underdeliver; rather, it is a guide for discussion with the patient and with a parent or guardian when applicable.
Consistent with the comparative AV study analysis that evaluated TOA, a linear course of lesion reductions between the provided time intervals was assumed. In this linear model, the TOA was calculated using the 2 extracted lesion count values between which the 25% lesion reduction was achieved as well as their corresponding given time points.9 Differences between the results in the active and vehicle study arms were calculated for a number of determinants.
How was pivotal trial data with clindamycin-BP 3.75% gel used to assess TOA?
A total of 498 patients with moderate to severe AV were randomized (1:1) to receive clindamycin-BP 3.75% gel or vehicle in a multicenter, double-blind, controlled, 12-week, 2-arm study.10 Before randomization, patients were stratified by acne severity based on a static Evaluator’s Global Severity Score (EGSS) ranging from 0 (clear) to 5 (very severe). Specifically, moderate AV (EGSS of 3) was described as predominantly noninflammatory lesions with evidence of multiple inflammatory lesions; several to many comedones, papules, and pustules; and no more than 1 small nodulocystic lesion. Severe AV (EGSS of 4) was characterized by inflammatory lesions; numerous comedones, papules, and pustules; and possibly a few nodulocystic lesions.10
Male and female patients aged 12 to 40 years with moderate to severe AV—defined as 20 to 40 inflammatory lesions (papules, pustules, nodules), 20 to 100 noninflammatory lesions (comedones), and no more than 2 nodules—were included in the study. Standard washout periods were required for patients using prior prescription and over-the-counter acne treatments.10
Efficacy evaluations included inflammatory and noninflammatory lesion counts and EGSS at screening, baseline, and during treatment (weeks 4, 8, and 12).10 Primary efficacy end points included absolute change in mean inflammatory and noninflammatory lesion counts and the proportion of patients who achieved at least a 2-grade reduction in EGSS from baseline to week 12 (treatment success at end of study). Secondary efficacy end points included mean percentage change from baseline to week 12 in inflammatory and noninflammatory lesion counts and the proportion of patients who considered themselves clear or almost clear at week 12.10
After 12 weeks of daily treatment, inflammatory and noninflammatory lesion counts decreased by a mean of 60.4% and 51.8%, respectively, with clindamycin-BP 3.75% gel compared to 31.3% and 27.6%, respectively, with vehicle (both P<.001). At weeks 4, 8, and 12, the difference in inflammatory and noninflammatory lesion counts for the active treatment was 17.4%, 24.8%, and 29.1%, respectively, and 8.1%, 19.8%, and 24.2%, respectively, for vehicle.10
Treatment success (at least a 2-grade improvement in EGSS) was achieved by 9.1% of patients using clindamycin-BP 3.75% gel compared to 4.6% using vehicle by week 4. Additionally, 6.3% of patients considered their AV as clear or almost clear compared to 3.5% with vehicle at week 2 (Figure 1).10

This analysis represents the first attempt to evaluate and report TOA results with clindamycin-BP 3.75% gel. Time to onset of action for inflammatory lesions treated with clindamycin-BP 3.75% gel was calculated as 2.5 weeks versus 6.2 weeks for vehicle (Figure 2A). Time to onset of action for noninflammatory lesions was 3.7 weeks with clindamycin-BP 3.75% gel versus 8.6 weeks with vehicle (Figure 2B). The difference in TOA between the active and vehicle study groups was 3.7 weeks and 4.9 weeks, respectively. In addition, among actively treated patients, TOA was shorter in females (2.1 weeks) than in males (2.6 weeks) and in moderate AV (2.5 weeks) compared to severe AV (3.0 weeks).

Comment
Differences in lesion counts between clindamycin-BP 3.75% gel and vehicle suggest a clinically relevant benefit in favor of active treatment with both inflammatory and noninflammatory lesions. Nearly twice as many patients were rated as treatment successes using EGSS by week 4 or clear or almost clear as early as week 2 compared to the vehicle group.10 However, these data are suggested as an overall guide but do not provide adequate guidance on when visible improvement may start to be evident in a given patient.
The analysis reported here shows a TOA of 2.5 weeks with clindamycin-BP 3.75% gel for inflammatory lesions, approximately 4 weeks faster than with the vehicle. In most cases, a reduction in inflammatory lesions is more likely to have a greater impact on patient perception of TOA. Unless a patient is aware or focused enough to actively distinguish visibly between inflammatory and noninflammatory (comedonal) AV lesions, their eye is more likely to be drawn initially to reduction in inflammatory lesions, which are erythematous and more visible at a greater viewing distance. Although noninflammatory AV lesions usually require closer inspection to visualize them (especially closed comedones), they are often slower to respond to treatment. Analysis of the pivotal trial data reports a longer TOA with clindamycin-BP 3.75% gel for noninflammatory lesions (3.7 weeks) versus inflammatory lesions (2.5 weeks).
As expected, TOA was shorter in patients with moderate AV than severe AV (2.5 weeks vs 3.0 weeks). Time to onset of action also was shorter in females overall. It is unclear why we see gender differences in acne studies. A number of reasons have been suggested, including differences in AV pathophysiology and/or treatment adherence.11,12 Greater efficacy of clindamycin-BP 3.75% gel in females compared with males has already been reported, and better overall efficacy leading to a shorter TOA has been noted by others.13
There are limitations with this analysis. First, it is not possible to assess the contributions from each of the monads to the efficacy of clindamycin-BP 3.75% gel or TOA. Also, the data extraction method used assumes a linear progression model during the provided time points and was used to provide some comparison with calculations for other combination products.9 Although no strong deviations from the linear model are likely, calculations of TOA using other methodologies may give different results. The definition of a clinically meaningful benefit, defined here as a 25% reduction in the mean lesion count, has been used as a guide, but it has not been validated in clinical practice. It also is important to recognize that the initial visible perception of improvement of AV is likely to differ based on interpatient variability; that is, how different individuals perceive improvement. It also may be affected by differences in baseline severity of AV among different patients. Additionally, the TOA reflects an average duration of time, so it should not be described to patients as a suggestion of when they will definitely see visible improvement in their AV.
Conclusion
Unrealistic expectations of acne therapy or poor tolerability can lead to low adherence and poor clinical outcomes.1-4 The data on TOA reported here suggests that a clinically meaningful benefit with clindamycin-BP 3.75% gel may be seen in some patients within 2 to 3 weeks and maybe sooner in females or those with milder disease; however, longer durations may be required in some patients. This information can help clinicians and their staff in providing reasonable expectations and stress the importance of encouraging patients about the need to adhere to treatment.
Acknowledgments
The author thanks Brian Bulley, MSc (Inergy Limited, Lindfield, West Sussex, United Kingdom), for publication support. Valeant Pharmaceuticals North America, LLC, funded Inergy’s activities pertaining to this analysis. The author did not receive funding or any form of compensation for authorship of this publication.
- Krakowski AC, Stendardo S, Eichenfield LF. Practical considerations in acne treatment and the clinical impact of topical combination therapy. Pediatr Dermatol. 2008;25(suppl 1):1-14.
- Yentzer BA, Ade RA, Fountain JM, et al. Simplifying regimens promotes greater adherence and outcomes with topical acne medications: a randomized controlled trial. Cutis. 2010;86:103-108.
- Zaghloul SS, Cunliffe WJ, Goodfield MJ. Objective assessment of compliance with treatments in acne. Br J Dermatol. 2005;152:1015-1021.
- Snyder S, Crandell I, Davis SA, et al. Medical adherence to acne therapy: a systematic review. Am J Clin Dermatol. 2014;15:87-94.
- Miyachi Y, Hayashi N, Furukawa F, et al. Acne management in Japan: study of patient adherence. Dermatology. 2011;223:174-181.
- Zauli S, Caracciolo S, Borghi A, et al. Which factors influence quality of life in acne patients? J Eur Acad Dermatol Venereol. 2014;28:46-50.
- Mulder MM, Sigurdsson V, van Zuuren EJ, et al. Psychosocial impact of acne vulgaris. evaluation of the relation between a change in clinical acne severity and psychosocial state. Dermatology. 2001;203:124-130.
- Gerlinger C, Stadtler G, Gotzelmann R, et al. A noninferiority margin for acne lesion counts. Drug Inf J. 2008;42:607-615.
- Jacobs A, Starke G, Rosumeck S, et al. Systematic review on the rapidity of the onset of action of topical treatments in the therapy of mild-to-moderate acne vulgaris. Br J Dermatol. 2014;170:557-564.
- Pariser DM, Rich P, Cook-Bolden FE, et al. An aqueous gel fixed combination of clindamycin phosphate 1.2% and benzoyl peroxide 3.75% for the once-daily treatment of moderate to severe acne vulgaris. J Drugs Dermatol. 2014;13:611-617.
- Tanghetti E, Harper JC, Oefelein MG. The efficacy and tolerability of dapsone 5% gel in female vs male patients with facial acne vulgaris: gender as a clinically relevant outcome variable. J Drugs Dermatol. 2012;11:1417-1421.
- Lott R, Taylor SL, O’Neill JL, et al. Medication adherence among acne patients: a review. J Cosmet Dermatol. 2010;9:160-166.
- Harper JC. The efficacy and tolerability of a fixed combination clindamycin (1.2%) and benzoyl peroxide (3.75%) aqueous gel in patients with facial acne vulgaris: gender as a clinically relevant outcome variable. J Drugs Dermatol. 2015;14:381-384.
- Krakowski AC, Stendardo S, Eichenfield LF. Practical considerations in acne treatment and the clinical impact of topical combination therapy. Pediatr Dermatol. 2008;25(suppl 1):1-14.
- Yentzer BA, Ade RA, Fountain JM, et al. Simplifying regimens promotes greater adherence and outcomes with topical acne medications: a randomized controlled trial. Cutis. 2010;86:103-108.
- Zaghloul SS, Cunliffe WJ, Goodfield MJ. Objective assessment of compliance with treatments in acne. Br J Dermatol. 2005;152:1015-1021.
- Snyder S, Crandell I, Davis SA, et al. Medical adherence to acne therapy: a systematic review. Am J Clin Dermatol. 2014;15:87-94.
- Miyachi Y, Hayashi N, Furukawa F, et al. Acne management in Japan: study of patient adherence. Dermatology. 2011;223:174-181.
- Zauli S, Caracciolo S, Borghi A, et al. Which factors influence quality of life in acne patients? J Eur Acad Dermatol Venereol. 2014;28:46-50.
- Mulder MM, Sigurdsson V, van Zuuren EJ, et al. Psychosocial impact of acne vulgaris. evaluation of the relation between a change in clinical acne severity and psychosocial state. Dermatology. 2001;203:124-130.
- Gerlinger C, Stadtler G, Gotzelmann R, et al. A noninferiority margin for acne lesion counts. Drug Inf J. 2008;42:607-615.
- Jacobs A, Starke G, Rosumeck S, et al. Systematic review on the rapidity of the onset of action of topical treatments in the therapy of mild-to-moderate acne vulgaris. Br J Dermatol. 2014;170:557-564.
- Pariser DM, Rich P, Cook-Bolden FE, et al. An aqueous gel fixed combination of clindamycin phosphate 1.2% and benzoyl peroxide 3.75% for the once-daily treatment of moderate to severe acne vulgaris. J Drugs Dermatol. 2014;13:611-617.
- Tanghetti E, Harper JC, Oefelein MG. The efficacy and tolerability of dapsone 5% gel in female vs male patients with facial acne vulgaris: gender as a clinically relevant outcome variable. J Drugs Dermatol. 2012;11:1417-1421.
- Lott R, Taylor SL, O’Neill JL, et al. Medication adherence among acne patients: a review. J Cosmet Dermatol. 2010;9:160-166.
- Harper JC. The efficacy and tolerability of a fixed combination clindamycin (1.2%) and benzoyl peroxide (3.75%) aqueous gel in patients with facial acne vulgaris: gender as a clinically relevant outcome variable. J Drugs Dermatol. 2015;14:381-384.
Practice Points
- Time to onset of action (TOA) refers to how long it takes after starting a therapy for a patient to perceive visible improvement.
- Time to onset of action has been determined based on data to date to correlate overall with a 25% lesion reduction.
- The TOA for clindamycin phosphate 1.2%–benzoyl peroxide 3.75% gel applied once daily based on analysis of pivotal trial data is 3 weeks or less depending on the severity of acne vulgaris at baseline.