User login
Welcome to Current Psychiatry, a leading source of information, online and in print, for practitioners of psychiatry and its related subspecialties, including addiction psychiatry, child and adolescent psychiatry, and geriatric psychiatry. This Web site contains evidence-based reviews of the prevention, diagnosis, and treatment of mental illness and psychological disorders; case reports; updates on psychopharmacology; news about the specialty of psychiatry; pearls for practice; and other topics of interest and use to this audience.
Dear Drupal User: You're seeing this because you're logged in to Drupal, and not redirected to MDedge.com/psychiatry.
Depression
adolescent depression
adolescent major depressive disorder
adolescent schizophrenia
adolescent with major depressive disorder
animals
autism
baby
brexpiprazole
child
child bipolar
child depression
child schizophrenia
children with bipolar disorder
children with depression
children with major depressive disorder
compulsive behaviors
cure
elderly bipolar
elderly depression
elderly major depressive disorder
elderly schizophrenia
elderly with dementia
first break
first episode
gambling
gaming
geriatric depression
geriatric major depressive disorder
geriatric schizophrenia
infant
kid
major depressive disorder
major depressive disorder in adolescents
major depressive disorder in children
parenting
pediatric
pediatric bipolar
pediatric depression
pediatric major depressive disorder
pediatric schizophrenia
pregnancy
pregnant
rexulti
skin care
teen
wine
section[contains(@class, 'nav-hidden')]
footer[@id='footer']
div[contains(@class, 'pane-pub-article-current-psychiatry')]
div[contains(@class, 'pane-pub-home-current-psychiatry')]
div[contains(@class, 'pane-pub-topic-current-psychiatry')]
div[contains(@class, 'panel-panel-inner')]
div[contains(@class, 'pane-node-field-article-topics')]
section[contains(@class, 'footer-nav-section-wrapper')]
Is this patient dangerous? 5 steps to assess risk for violence
“Will this patient turn violent?” Psychiatrists face this tough question every day. Although predicting a complex behavior such as violence is nearly impossible, we can prepare for dangerous behavior and improve our safety by:
- knowing the risk factors for patient violence
- assessing individuals for violence potential before clinical encounters
- controlling situations to reduce injury risk.
In one study, more than 50% of psychiatrists and 75% of mental health nurses reported an act or threat of violence from patients within the past year.1 To help you avoid becoming a statistic, this article provides a 5-step procedure (Figure 1). to quickly assess and respond to risk of violence in a psychiatric patient.
Step 1: Seek patient history
A careful review of past events and those immediately preceding the clinical encounter is the best tool for assessing potential for violence. The more you can learn from the patient chart and other sources before you see the patient, the better (Table 1). Valuable clues can be obtained from interviews with family members, outpatient providers, police officers, and others who have had pertinent social contact with the patient.
Figure 5 steps to assess and reduce the risk of patient violence
Past violence is the most powerful predictor of future violence, according to published studies. Higher frequency of aggressive episodes, greater degree of aggressive injury, and lack of apparent provocation in past episodes all increase the violence risk.3
A minority of patients account for most aggressive acts in clinical encounters. One study showed that recidivists committed 53% of all violent behaviors in a health care setting.4 A patient’s history of violence should be flagged in the chart and verbally passed on to staff to alert providers of increased risk.
However, not having a violent history does not guarantee that a patient will not become dangerous during a clinical encounter. All patients with a violent past had an initial violent episode, and that first time can occur in a practice setting.
Psychotic states by themselves appear to increase the risk of violence, although the literature is mixed.5,6 Clearly, however, psychotic states associated with arousal or agitation do predispose patients to violence, especially if the psychosis involves active paranoid delusions or hallucinations associated with negative affect (anger, sadness, anxiety).7
Increased rates of violence have also been reported in psychiatric patients with:
- acute manic states associated with arousal or agitation8
- nonspecific neurologic abnormalities such as abnormal EEGs, localizing neurologic signs, or “soft signs” (impaired face-hand test, graphesthesia, stereognosis).9
Demographic variables associated with higher violence rates include ages 15 to 24, nonwhite race, male gender, poverty, and low educational level. Other variables include history of abuse, victimization, family violence, limited employment skills, and “rootlessness,” such as poor family network and frequent moves or job changes.10
Psychiatric diagnoses associated with increased risk of violence include schizophrenia, bipolar mania, alcohol and other substance abuse, and personality disorders.11-13 In clinical practice, however, I find psychiatric diagnoses less useful in predicting violence than the patient’s arousal state and the other risk factors discussed above.
Step 2: Evaluate the context
In addition to evidence-supported risk factors (Table 2), context—or the broader situation in which a patient is embedded at the time of psychiatric evaluation—plays a prominent role in potentially violent situations. For example, if “divorce” is listed as a presenting factor:
- Is the patient recently divorced, or did it occur years ago?
- Does he hate all women or just his ex-wife?
- Was she having an affair, and did he just learn about this?
In other words, environmental stresses can be acute and destabilizing or part of the patient’s chronic life picture and serve in homeostatic functioning.
Step 3: Identify arousal states
Patients rarely commit violent acts when their anxiety and moods are well controlled. They are more likely to become aggressive in high arousal states.
Fear is probably an element of most situations where patients act out violently. Because the fearful patient may not exhibit easily interpreted danger signals, however, you may unwittingly provoke an assault by violating his or her personal space. A fearful, paranoid patient requires a greater-than-usual “intimate zone,” although this need for increased space may not be obvious.
Minimize provocation by explaining your actions and behaviors in advance (such as, “I would like to enter the room, sit down, and talk with you for about 20 minutes”). Be business-like with paranoid patients. Avoid exuding warmth, as they may view attempts at warmth as having sinister intent.
Clinicians are sometimes injured when trying to prevent a fearful, paranoid patient from fleeing. To avoid injury, don’t stand between the patient and the door. Let the patient escape from the immediate situation, and enlist security or police in further intervention attempts.
Anger is easy to recognize by signs of mounting tension. Loud voice, inappropriate staring, banging objects, clenched fists, agitated pacing, and verbal threats are common in the angry patient before a violent episode. Although this seems self-evident, it is surprising how many violent acts occur when these signs are obvious and noted by staff, yet no de-escalation measures are taken.
A patient’s verbal threats can actually help the clinician. This “red flag” alerts staff to focus on de-escalation techniques and prepare for a violent situation.
Confusion can be an underlying risk factor in patients with delirium or nonspecific organic brain syndrome. These patients may strike out unexpectedly when health care personnel are attempting to do routine procedures, and clinicians are sometimes caught off-guard when operating in a care-giving rather than defensive mode.
Table 1
Will this patient become violent? Questions to consider before a clinical encounter
Long-term behavior
|
Immediate situation
|
Clinicians can often avoid arousing confused patients by using orienting techniques and explaining their actions. For example, a nurse might say, “Hello Mr. X, I am a nurse and you are in this hospital for treatment of your illness. I will need to use this machine to check your blood pressure.”
Humiliation. Men in particular can react aggressively to loss of self-esteem and feelings of powerlessness. Take note if a man has been humiliated in front of family before being brought for evaluation; for example, was he removed by police in an emergency detention situation? This patient may need to act out violently to restore his sense of self.
Staff can lessen a patient’s potential to act on humiliation by using a therapeutic, esteem-building interview technique. For example, address the patient as “Mr.” instead of by first name, and highlight his strengths or accomplishments early in the interview.
Table 2
Risk factors for violence among psychiatric patients*
|
| * As identified in the literature. |
Step 4: Structure the interview for safety
The time you take before an interview to learn about a patient’s violence history, context, and arousal state is time well-spent and more patient-specific than past diagnoses. This information allows you to prepare for a safe intervention.
Interview environment. The physical and social environment where you interview the patient may contribute to violence potential.
- Is the patient being interviewed in a cramped room or an open hallway?
- Is the evaluation unit overcrowded?
- Are security personnel visible?
- Is the examiner of the same race or ethnic background as the patient?
Cramped and overcrowded conditions on a psychiatric ward have been associated with higher rates of patient violence.2 In one case of context-specific violence, a veteran with known institutional transference issues toward the government attacked providers in a VA hospital on several occasions but did not exhibit this behavior in other, non-VA medical settings.
Take control of the interview and treatment situation. Use the physical space and personnel as you would any other intervention tool—to increase safety and decrease potential for violent behavior. For example, some patients do better when interviewed in a small, private setting. Other interviews must be conducted in a triage area while police escorts hold the patient and handcuffs remain on.
Ideally, you and the patient should have equal access to the door if you conduct the psychiatric interview in an enclosed room. With high-risk patients, arrange your seating at a 90-degree angle—rather than face-to-face—to limit sustained, confrontational eye contact. Sit at greater than an arm swing or leg kick away from the patient, and require him or her to remain seated during the interview (or you will promptly leave).
In the outpatient practice, terminate the interview or evaluation session if a patient in a negative affective arousal state does not allow verbal redirection. Before you make any movement to exit, however, announce, “I am leaving the room now.”
Trust your intuition. I do not enter a closed, private space with a patient unless I feel safe. If I feel afraid, I take that as a valuable warning that further safety measures are necessary.
Use restraints as needed. When patients with a history of violence are brought to the hospital in high arousal states, I let them remain in restraint with security present during the initial interview. If the patient cannot have a back-and-forth conversation with me, I keep the security force present until I believe my verbal interactions have a substantial effect.
Patients must be responsive to talking interventions before restraint, security, or other environmental safety measures are removed. Some patients do not reach this point until after tranquilizing medications are given.
Step 5: Tthe clinical encounter
When discussing how to assess the likelihood of patient violence during a clinical encounter, a psychiatric colleague once commented, “Risk factors make you worry more; nothing makes you worry less.”
In other words, keep your guard up. Let clinical judgment take precedence over statistics when you are evaluating any patient. Statistics represent frequencies or averages; they may or may not apply to any one individual.
Techniques for assessing and treating violent patients are beyond the scope of this article, but at the very least:
- obtain training in safety/treatment protocols for violent patients
- ensure that your hospital/clinic has procedures in place to improve safety and to handle violent situations.
Visible, high numbers of confident-appearing—but not confrontational—staff or security may dissuade the patient from acting out. Then, most often, force will not be needed. If force is needed to control a violent patient, make sure the staff’s response is strong and overwhelming.
For every violent act requiring staff intervention, automatically schedule a debriefing session for those involved to assess the incident and allow them to express their feelings.
Related resources
- American Association for Emergency Psychiatry. www.emergencypsychiatry.org
- Volavka J. The neurobiology of violence: an update. J Neuropsychiatry Clin Neurosci 1999;11:307-14.
- McNiel DE, Eisner JP, Binder RL. The relationship between command hallucinations and violence. Psychiatric Services 2000;51:1288-92.
1. Nolan P, Dallender J, Soares J, et al. Violence in mental health care: the experiences of mental health nurses and psychiatrists. J Adv Nurs 1999;30:934-41.
2. Blomhoff S, Seim S, Friis S. Can prediction of violence among psychiatric inpatients be improved? Hosp Community Psychiatry 1990;41:771-5.
3. Convit A, Isay D, Otis D, et al. Characteristics of repeatedly assaultive psychiatric inpatients. Hosp Community Psychiatry 1990;41:1112-5.
4. Taylor P. Motives for offending among violent and psychotic men. Br J Psychiatry 1985;147:491-8.
5. Junginger J, Parks-Levy J, McGuire L. Delusions and symptom-consistent violence. Psychiatr Serv 1998;49:218-20.
6. Cheung P, Schweitzer I, Crowley K, et al. Violence in schizophrenia: role of hallucinations and delusions. Schizophr Res 1997;26:181-90.
7. Binder R, McNiel D. Effects of diagnosis and context on dangerousness. Am J Psychiatry 1988;145:728-32.
8. Convit A, Jaeger J, Pin Lin S, et al. Predicting assaultiveness in psychiatric inpatients: A pilot study. Hosp Community Psychiatry 1988;39:429-34.
9. Hyman S. The violent patient. In: Hyman S (ed). Manual of psychiatric emergencies. Boston: Little, Brown and Co., 1988;23-31.
10. Swartz M, Swanson J, Hiday V, et al. Violence and severe mental illness: the effects of substance abuse and nonadherence to medication. Am J Psychiatry 1998;155:226-31.
11. Owen C, Tarantello C, Jones M, et al. Repetitively violent patients in psychiatric units. Psychiatr Serv 1998;49:1458-61.
12. Citrome L, Volavka J. Clinical management of persistent aggressive behavior in schizophrenia, part I. Definitions, epidemiology, assessment and acute treatment. Essen Psychopharmacol 2002;5:1-16.
13. Abeyasinghe R, Jayasekera R. Violence in a general hospital psychiatry unit for men. Ceylon Med J 2003;48(2):45-7.
“Will this patient turn violent?” Psychiatrists face this tough question every day. Although predicting a complex behavior such as violence is nearly impossible, we can prepare for dangerous behavior and improve our safety by:
- knowing the risk factors for patient violence
- assessing individuals for violence potential before clinical encounters
- controlling situations to reduce injury risk.
In one study, more than 50% of psychiatrists and 75% of mental health nurses reported an act or threat of violence from patients within the past year.1 To help you avoid becoming a statistic, this article provides a 5-step procedure (Figure 1). to quickly assess and respond to risk of violence in a psychiatric patient.
Step 1: Seek patient history
A careful review of past events and those immediately preceding the clinical encounter is the best tool for assessing potential for violence. The more you can learn from the patient chart and other sources before you see the patient, the better (Table 1). Valuable clues can be obtained from interviews with family members, outpatient providers, police officers, and others who have had pertinent social contact with the patient.
Figure 5 steps to assess and reduce the risk of patient violence
Past violence is the most powerful predictor of future violence, according to published studies. Higher frequency of aggressive episodes, greater degree of aggressive injury, and lack of apparent provocation in past episodes all increase the violence risk.3
A minority of patients account for most aggressive acts in clinical encounters. One study showed that recidivists committed 53% of all violent behaviors in a health care setting.4 A patient’s history of violence should be flagged in the chart and verbally passed on to staff to alert providers of increased risk.
However, not having a violent history does not guarantee that a patient will not become dangerous during a clinical encounter. All patients with a violent past had an initial violent episode, and that first time can occur in a practice setting.
Psychotic states by themselves appear to increase the risk of violence, although the literature is mixed.5,6 Clearly, however, psychotic states associated with arousal or agitation do predispose patients to violence, especially if the psychosis involves active paranoid delusions or hallucinations associated with negative affect (anger, sadness, anxiety).7
Increased rates of violence have also been reported in psychiatric patients with:
- acute manic states associated with arousal or agitation8
- nonspecific neurologic abnormalities such as abnormal EEGs, localizing neurologic signs, or “soft signs” (impaired face-hand test, graphesthesia, stereognosis).9
Demographic variables associated with higher violence rates include ages 15 to 24, nonwhite race, male gender, poverty, and low educational level. Other variables include history of abuse, victimization, family violence, limited employment skills, and “rootlessness,” such as poor family network and frequent moves or job changes.10
Psychiatric diagnoses associated with increased risk of violence include schizophrenia, bipolar mania, alcohol and other substance abuse, and personality disorders.11-13 In clinical practice, however, I find psychiatric diagnoses less useful in predicting violence than the patient’s arousal state and the other risk factors discussed above.
Step 2: Evaluate the context
In addition to evidence-supported risk factors (Table 2), context—or the broader situation in which a patient is embedded at the time of psychiatric evaluation—plays a prominent role in potentially violent situations. For example, if “divorce” is listed as a presenting factor:
- Is the patient recently divorced, or did it occur years ago?
- Does he hate all women or just his ex-wife?
- Was she having an affair, and did he just learn about this?
In other words, environmental stresses can be acute and destabilizing or part of the patient’s chronic life picture and serve in homeostatic functioning.
Step 3: Identify arousal states
Patients rarely commit violent acts when their anxiety and moods are well controlled. They are more likely to become aggressive in high arousal states.
Fear is probably an element of most situations where patients act out violently. Because the fearful patient may not exhibit easily interpreted danger signals, however, you may unwittingly provoke an assault by violating his or her personal space. A fearful, paranoid patient requires a greater-than-usual “intimate zone,” although this need for increased space may not be obvious.
Minimize provocation by explaining your actions and behaviors in advance (such as, “I would like to enter the room, sit down, and talk with you for about 20 minutes”). Be business-like with paranoid patients. Avoid exuding warmth, as they may view attempts at warmth as having sinister intent.
Clinicians are sometimes injured when trying to prevent a fearful, paranoid patient from fleeing. To avoid injury, don’t stand between the patient and the door. Let the patient escape from the immediate situation, and enlist security or police in further intervention attempts.
Anger is easy to recognize by signs of mounting tension. Loud voice, inappropriate staring, banging objects, clenched fists, agitated pacing, and verbal threats are common in the angry patient before a violent episode. Although this seems self-evident, it is surprising how many violent acts occur when these signs are obvious and noted by staff, yet no de-escalation measures are taken.
A patient’s verbal threats can actually help the clinician. This “red flag” alerts staff to focus on de-escalation techniques and prepare for a violent situation.
Confusion can be an underlying risk factor in patients with delirium or nonspecific organic brain syndrome. These patients may strike out unexpectedly when health care personnel are attempting to do routine procedures, and clinicians are sometimes caught off-guard when operating in a care-giving rather than defensive mode.
Table 1
Will this patient become violent? Questions to consider before a clinical encounter
Long-term behavior
|
Immediate situation
|
Clinicians can often avoid arousing confused patients by using orienting techniques and explaining their actions. For example, a nurse might say, “Hello Mr. X, I am a nurse and you are in this hospital for treatment of your illness. I will need to use this machine to check your blood pressure.”
Humiliation. Men in particular can react aggressively to loss of self-esteem and feelings of powerlessness. Take note if a man has been humiliated in front of family before being brought for evaluation; for example, was he removed by police in an emergency detention situation? This patient may need to act out violently to restore his sense of self.
Staff can lessen a patient’s potential to act on humiliation by using a therapeutic, esteem-building interview technique. For example, address the patient as “Mr.” instead of by first name, and highlight his strengths or accomplishments early in the interview.
Table 2
Risk factors for violence among psychiatric patients*
|
| * As identified in the literature. |
Step 4: Structure the interview for safety
The time you take before an interview to learn about a patient’s violence history, context, and arousal state is time well-spent and more patient-specific than past diagnoses. This information allows you to prepare for a safe intervention.
Interview environment. The physical and social environment where you interview the patient may contribute to violence potential.
- Is the patient being interviewed in a cramped room or an open hallway?
- Is the evaluation unit overcrowded?
- Are security personnel visible?
- Is the examiner of the same race or ethnic background as the patient?
Cramped and overcrowded conditions on a psychiatric ward have been associated with higher rates of patient violence.2 In one case of context-specific violence, a veteran with known institutional transference issues toward the government attacked providers in a VA hospital on several occasions but did not exhibit this behavior in other, non-VA medical settings.
Take control of the interview and treatment situation. Use the physical space and personnel as you would any other intervention tool—to increase safety and decrease potential for violent behavior. For example, some patients do better when interviewed in a small, private setting. Other interviews must be conducted in a triage area while police escorts hold the patient and handcuffs remain on.
Ideally, you and the patient should have equal access to the door if you conduct the psychiatric interview in an enclosed room. With high-risk patients, arrange your seating at a 90-degree angle—rather than face-to-face—to limit sustained, confrontational eye contact. Sit at greater than an arm swing or leg kick away from the patient, and require him or her to remain seated during the interview (or you will promptly leave).
In the outpatient practice, terminate the interview or evaluation session if a patient in a negative affective arousal state does not allow verbal redirection. Before you make any movement to exit, however, announce, “I am leaving the room now.”
Trust your intuition. I do not enter a closed, private space with a patient unless I feel safe. If I feel afraid, I take that as a valuable warning that further safety measures are necessary.
Use restraints as needed. When patients with a history of violence are brought to the hospital in high arousal states, I let them remain in restraint with security present during the initial interview. If the patient cannot have a back-and-forth conversation with me, I keep the security force present until I believe my verbal interactions have a substantial effect.
Patients must be responsive to talking interventions before restraint, security, or other environmental safety measures are removed. Some patients do not reach this point until after tranquilizing medications are given.
Step 5: Tthe clinical encounter
When discussing how to assess the likelihood of patient violence during a clinical encounter, a psychiatric colleague once commented, “Risk factors make you worry more; nothing makes you worry less.”
In other words, keep your guard up. Let clinical judgment take precedence over statistics when you are evaluating any patient. Statistics represent frequencies or averages; they may or may not apply to any one individual.
Techniques for assessing and treating violent patients are beyond the scope of this article, but at the very least:
- obtain training in safety/treatment protocols for violent patients
- ensure that your hospital/clinic has procedures in place to improve safety and to handle violent situations.
Visible, high numbers of confident-appearing—but not confrontational—staff or security may dissuade the patient from acting out. Then, most often, force will not be needed. If force is needed to control a violent patient, make sure the staff’s response is strong and overwhelming.
For every violent act requiring staff intervention, automatically schedule a debriefing session for those involved to assess the incident and allow them to express their feelings.
Related resources
- American Association for Emergency Psychiatry. www.emergencypsychiatry.org
- Volavka J. The neurobiology of violence: an update. J Neuropsychiatry Clin Neurosci 1999;11:307-14.
- McNiel DE, Eisner JP, Binder RL. The relationship between command hallucinations and violence. Psychiatric Services 2000;51:1288-92.
“Will this patient turn violent?” Psychiatrists face this tough question every day. Although predicting a complex behavior such as violence is nearly impossible, we can prepare for dangerous behavior and improve our safety by:
- knowing the risk factors for patient violence
- assessing individuals for violence potential before clinical encounters
- controlling situations to reduce injury risk.
In one study, more than 50% of psychiatrists and 75% of mental health nurses reported an act or threat of violence from patients within the past year.1 To help you avoid becoming a statistic, this article provides a 5-step procedure (Figure 1). to quickly assess and respond to risk of violence in a psychiatric patient.
Step 1: Seek patient history
A careful review of past events and those immediately preceding the clinical encounter is the best tool for assessing potential for violence. The more you can learn from the patient chart and other sources before you see the patient, the better (Table 1). Valuable clues can be obtained from interviews with family members, outpatient providers, police officers, and others who have had pertinent social contact with the patient.
Figure 5 steps to assess and reduce the risk of patient violence
Past violence is the most powerful predictor of future violence, according to published studies. Higher frequency of aggressive episodes, greater degree of aggressive injury, and lack of apparent provocation in past episodes all increase the violence risk.3
A minority of patients account for most aggressive acts in clinical encounters. One study showed that recidivists committed 53% of all violent behaviors in a health care setting.4 A patient’s history of violence should be flagged in the chart and verbally passed on to staff to alert providers of increased risk.
However, not having a violent history does not guarantee that a patient will not become dangerous during a clinical encounter. All patients with a violent past had an initial violent episode, and that first time can occur in a practice setting.
Psychotic states by themselves appear to increase the risk of violence, although the literature is mixed.5,6 Clearly, however, psychotic states associated with arousal or agitation do predispose patients to violence, especially if the psychosis involves active paranoid delusions or hallucinations associated with negative affect (anger, sadness, anxiety).7
Increased rates of violence have also been reported in psychiatric patients with:
- acute manic states associated with arousal or agitation8
- nonspecific neurologic abnormalities such as abnormal EEGs, localizing neurologic signs, or “soft signs” (impaired face-hand test, graphesthesia, stereognosis).9
Demographic variables associated with higher violence rates include ages 15 to 24, nonwhite race, male gender, poverty, and low educational level. Other variables include history of abuse, victimization, family violence, limited employment skills, and “rootlessness,” such as poor family network and frequent moves or job changes.10
Psychiatric diagnoses associated with increased risk of violence include schizophrenia, bipolar mania, alcohol and other substance abuse, and personality disorders.11-13 In clinical practice, however, I find psychiatric diagnoses less useful in predicting violence than the patient’s arousal state and the other risk factors discussed above.
Step 2: Evaluate the context
In addition to evidence-supported risk factors (Table 2), context—or the broader situation in which a patient is embedded at the time of psychiatric evaluation—plays a prominent role in potentially violent situations. For example, if “divorce” is listed as a presenting factor:
- Is the patient recently divorced, or did it occur years ago?
- Does he hate all women or just his ex-wife?
- Was she having an affair, and did he just learn about this?
In other words, environmental stresses can be acute and destabilizing or part of the patient’s chronic life picture and serve in homeostatic functioning.
Step 3: Identify arousal states
Patients rarely commit violent acts when their anxiety and moods are well controlled. They are more likely to become aggressive in high arousal states.
Fear is probably an element of most situations where patients act out violently. Because the fearful patient may not exhibit easily interpreted danger signals, however, you may unwittingly provoke an assault by violating his or her personal space. A fearful, paranoid patient requires a greater-than-usual “intimate zone,” although this need for increased space may not be obvious.
Minimize provocation by explaining your actions and behaviors in advance (such as, “I would like to enter the room, sit down, and talk with you for about 20 minutes”). Be business-like with paranoid patients. Avoid exuding warmth, as they may view attempts at warmth as having sinister intent.
Clinicians are sometimes injured when trying to prevent a fearful, paranoid patient from fleeing. To avoid injury, don’t stand between the patient and the door. Let the patient escape from the immediate situation, and enlist security or police in further intervention attempts.
Anger is easy to recognize by signs of mounting tension. Loud voice, inappropriate staring, banging objects, clenched fists, agitated pacing, and verbal threats are common in the angry patient before a violent episode. Although this seems self-evident, it is surprising how many violent acts occur when these signs are obvious and noted by staff, yet no de-escalation measures are taken.
A patient’s verbal threats can actually help the clinician. This “red flag” alerts staff to focus on de-escalation techniques and prepare for a violent situation.
Confusion can be an underlying risk factor in patients with delirium or nonspecific organic brain syndrome. These patients may strike out unexpectedly when health care personnel are attempting to do routine procedures, and clinicians are sometimes caught off-guard when operating in a care-giving rather than defensive mode.
Table 1
Will this patient become violent? Questions to consider before a clinical encounter
Long-term behavior
|
Immediate situation
|
Clinicians can often avoid arousing confused patients by using orienting techniques and explaining their actions. For example, a nurse might say, “Hello Mr. X, I am a nurse and you are in this hospital for treatment of your illness. I will need to use this machine to check your blood pressure.”
Humiliation. Men in particular can react aggressively to loss of self-esteem and feelings of powerlessness. Take note if a man has been humiliated in front of family before being brought for evaluation; for example, was he removed by police in an emergency detention situation? This patient may need to act out violently to restore his sense of self.
Staff can lessen a patient’s potential to act on humiliation by using a therapeutic, esteem-building interview technique. For example, address the patient as “Mr.” instead of by first name, and highlight his strengths or accomplishments early in the interview.
Table 2
Risk factors for violence among psychiatric patients*
|
| * As identified in the literature. |
Step 4: Structure the interview for safety
The time you take before an interview to learn about a patient’s violence history, context, and arousal state is time well-spent and more patient-specific than past diagnoses. This information allows you to prepare for a safe intervention.
Interview environment. The physical and social environment where you interview the patient may contribute to violence potential.
- Is the patient being interviewed in a cramped room or an open hallway?
- Is the evaluation unit overcrowded?
- Are security personnel visible?
- Is the examiner of the same race or ethnic background as the patient?
Cramped and overcrowded conditions on a psychiatric ward have been associated with higher rates of patient violence.2 In one case of context-specific violence, a veteran with known institutional transference issues toward the government attacked providers in a VA hospital on several occasions but did not exhibit this behavior in other, non-VA medical settings.
Take control of the interview and treatment situation. Use the physical space and personnel as you would any other intervention tool—to increase safety and decrease potential for violent behavior. For example, some patients do better when interviewed in a small, private setting. Other interviews must be conducted in a triage area while police escorts hold the patient and handcuffs remain on.
Ideally, you and the patient should have equal access to the door if you conduct the psychiatric interview in an enclosed room. With high-risk patients, arrange your seating at a 90-degree angle—rather than face-to-face—to limit sustained, confrontational eye contact. Sit at greater than an arm swing or leg kick away from the patient, and require him or her to remain seated during the interview (or you will promptly leave).
In the outpatient practice, terminate the interview or evaluation session if a patient in a negative affective arousal state does not allow verbal redirection. Before you make any movement to exit, however, announce, “I am leaving the room now.”
Trust your intuition. I do not enter a closed, private space with a patient unless I feel safe. If I feel afraid, I take that as a valuable warning that further safety measures are necessary.
Use restraints as needed. When patients with a history of violence are brought to the hospital in high arousal states, I let them remain in restraint with security present during the initial interview. If the patient cannot have a back-and-forth conversation with me, I keep the security force present until I believe my verbal interactions have a substantial effect.
Patients must be responsive to talking interventions before restraint, security, or other environmental safety measures are removed. Some patients do not reach this point until after tranquilizing medications are given.
Step 5: Tthe clinical encounter
When discussing how to assess the likelihood of patient violence during a clinical encounter, a psychiatric colleague once commented, “Risk factors make you worry more; nothing makes you worry less.”
In other words, keep your guard up. Let clinical judgment take precedence over statistics when you are evaluating any patient. Statistics represent frequencies or averages; they may or may not apply to any one individual.
Techniques for assessing and treating violent patients are beyond the scope of this article, but at the very least:
- obtain training in safety/treatment protocols for violent patients
- ensure that your hospital/clinic has procedures in place to improve safety and to handle violent situations.
Visible, high numbers of confident-appearing—but not confrontational—staff or security may dissuade the patient from acting out. Then, most often, force will not be needed. If force is needed to control a violent patient, make sure the staff’s response is strong and overwhelming.
For every violent act requiring staff intervention, automatically schedule a debriefing session for those involved to assess the incident and allow them to express their feelings.
Related resources
- American Association for Emergency Psychiatry. www.emergencypsychiatry.org
- Volavka J. The neurobiology of violence: an update. J Neuropsychiatry Clin Neurosci 1999;11:307-14.
- McNiel DE, Eisner JP, Binder RL. The relationship between command hallucinations and violence. Psychiatric Services 2000;51:1288-92.
1. Nolan P, Dallender J, Soares J, et al. Violence in mental health care: the experiences of mental health nurses and psychiatrists. J Adv Nurs 1999;30:934-41.
2. Blomhoff S, Seim S, Friis S. Can prediction of violence among psychiatric inpatients be improved? Hosp Community Psychiatry 1990;41:771-5.
3. Convit A, Isay D, Otis D, et al. Characteristics of repeatedly assaultive psychiatric inpatients. Hosp Community Psychiatry 1990;41:1112-5.
4. Taylor P. Motives for offending among violent and psychotic men. Br J Psychiatry 1985;147:491-8.
5. Junginger J, Parks-Levy J, McGuire L. Delusions and symptom-consistent violence. Psychiatr Serv 1998;49:218-20.
6. Cheung P, Schweitzer I, Crowley K, et al. Violence in schizophrenia: role of hallucinations and delusions. Schizophr Res 1997;26:181-90.
7. Binder R, McNiel D. Effects of diagnosis and context on dangerousness. Am J Psychiatry 1988;145:728-32.
8. Convit A, Jaeger J, Pin Lin S, et al. Predicting assaultiveness in psychiatric inpatients: A pilot study. Hosp Community Psychiatry 1988;39:429-34.
9. Hyman S. The violent patient. In: Hyman S (ed). Manual of psychiatric emergencies. Boston: Little, Brown and Co., 1988;23-31.
10. Swartz M, Swanson J, Hiday V, et al. Violence and severe mental illness: the effects of substance abuse and nonadherence to medication. Am J Psychiatry 1998;155:226-31.
11. Owen C, Tarantello C, Jones M, et al. Repetitively violent patients in psychiatric units. Psychiatr Serv 1998;49:1458-61.
12. Citrome L, Volavka J. Clinical management of persistent aggressive behavior in schizophrenia, part I. Definitions, epidemiology, assessment and acute treatment. Essen Psychopharmacol 2002;5:1-16.
13. Abeyasinghe R, Jayasekera R. Violence in a general hospital psychiatry unit for men. Ceylon Med J 2003;48(2):45-7.
1. Nolan P, Dallender J, Soares J, et al. Violence in mental health care: the experiences of mental health nurses and psychiatrists. J Adv Nurs 1999;30:934-41.
2. Blomhoff S, Seim S, Friis S. Can prediction of violence among psychiatric inpatients be improved? Hosp Community Psychiatry 1990;41:771-5.
3. Convit A, Isay D, Otis D, et al. Characteristics of repeatedly assaultive psychiatric inpatients. Hosp Community Psychiatry 1990;41:1112-5.
4. Taylor P. Motives for offending among violent and psychotic men. Br J Psychiatry 1985;147:491-8.
5. Junginger J, Parks-Levy J, McGuire L. Delusions and symptom-consistent violence. Psychiatr Serv 1998;49:218-20.
6. Cheung P, Schweitzer I, Crowley K, et al. Violence in schizophrenia: role of hallucinations and delusions. Schizophr Res 1997;26:181-90.
7. Binder R, McNiel D. Effects of diagnosis and context on dangerousness. Am J Psychiatry 1988;145:728-32.
8. Convit A, Jaeger J, Pin Lin S, et al. Predicting assaultiveness in psychiatric inpatients: A pilot study. Hosp Community Psychiatry 1988;39:429-34.
9. Hyman S. The violent patient. In: Hyman S (ed). Manual of psychiatric emergencies. Boston: Little, Brown and Co., 1988;23-31.
10. Swartz M, Swanson J, Hiday V, et al. Violence and severe mental illness: the effects of substance abuse and nonadherence to medication. Am J Psychiatry 1998;155:226-31.
11. Owen C, Tarantello C, Jones M, et al. Repetitively violent patients in psychiatric units. Psychiatr Serv 1998;49:1458-61.
12. Citrome L, Volavka J. Clinical management of persistent aggressive behavior in schizophrenia, part I. Definitions, epidemiology, assessment and acute treatment. Essen Psychopharmacol 2002;5:1-16.
13. Abeyasinghe R, Jayasekera R. Violence in a general hospital psychiatry unit for men. Ceylon Med J 2003;48(2):45-7.
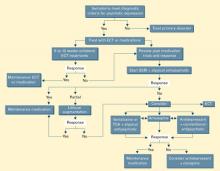
TMP bipolar algorithms: Not ‘cookbook’ medicine
How do you feel about the state, insurance companies, or professional societies telling you how to practice medicine? Most of us would take strong exception to that idea, almost as if we had been asked, “How do you feel about the butcher of Baghdad?” One reason we became doctors—in addition to wanting to heal the sick—was that we do not like anyone telling us what to do.
Let me rephrase the question: “How do you feel about evidence-based guidelines that could help you make increasingly complex decisions about which medications to use under which circumstances?” At worst, most of us would respond, “Well, I don’t really need them, but I refer to them from time to time. And I certainly know a lot of practitioners who could benefit from them.”
Two ways to ask the same question, with a big difference in response. I resist anyone’s attempts to write a “cookbook” for my clinical practice, but I am interested in anything that helps me practice rationally.
This year, Trisha Suppes, MD, PhD, and the other the Texas Medication Algorithm Project (TMAP) collaborators will update their treatment algorithms to include evidence published since 2000. In this issue, Dr. Suppes and Geetha Shivakumar, MD, of the University of Texas/Southwestern Medical Center’s department of psychiatry preview potential updates in the TMAP bipolar mania and bipolar depression algorithms. They make it clear that algorithms can be valuable tools when carefully designed and implemented.
Despite my wariness about practice guidelines, I found this article quite palatable and—I must admit—useful. I hope you do, too.
How do you feel about the state, insurance companies, or professional societies telling you how to practice medicine? Most of us would take strong exception to that idea, almost as if we had been asked, “How do you feel about the butcher of Baghdad?” One reason we became doctors—in addition to wanting to heal the sick—was that we do not like anyone telling us what to do.
Let me rephrase the question: “How do you feel about evidence-based guidelines that could help you make increasingly complex decisions about which medications to use under which circumstances?” At worst, most of us would respond, “Well, I don’t really need them, but I refer to them from time to time. And I certainly know a lot of practitioners who could benefit from them.”
Two ways to ask the same question, with a big difference in response. I resist anyone’s attempts to write a “cookbook” for my clinical practice, but I am interested in anything that helps me practice rationally.
This year, Trisha Suppes, MD, PhD, and the other the Texas Medication Algorithm Project (TMAP) collaborators will update their treatment algorithms to include evidence published since 2000. In this issue, Dr. Suppes and Geetha Shivakumar, MD, of the University of Texas/Southwestern Medical Center’s department of psychiatry preview potential updates in the TMAP bipolar mania and bipolar depression algorithms. They make it clear that algorithms can be valuable tools when carefully designed and implemented.
Despite my wariness about practice guidelines, I found this article quite palatable and—I must admit—useful. I hope you do, too.
How do you feel about the state, insurance companies, or professional societies telling you how to practice medicine? Most of us would take strong exception to that idea, almost as if we had been asked, “How do you feel about the butcher of Baghdad?” One reason we became doctors—in addition to wanting to heal the sick—was that we do not like anyone telling us what to do.
Let me rephrase the question: “How do you feel about evidence-based guidelines that could help you make increasingly complex decisions about which medications to use under which circumstances?” At worst, most of us would respond, “Well, I don’t really need them, but I refer to them from time to time. And I certainly know a lot of practitioners who could benefit from them.”
Two ways to ask the same question, with a big difference in response. I resist anyone’s attempts to write a “cookbook” for my clinical practice, but I am interested in anything that helps me practice rationally.
This year, Trisha Suppes, MD, PhD, and the other the Texas Medication Algorithm Project (TMAP) collaborators will update their treatment algorithms to include evidence published since 2000. In this issue, Dr. Suppes and Geetha Shivakumar, MD, of the University of Texas/Southwestern Medical Center’s department of psychiatry preview potential updates in the TMAP bipolar mania and bipolar depression algorithms. They make it clear that algorithms can be valuable tools when carefully designed and implemented.
Despite my wariness about practice guidelines, I found this article quite palatable and—I must admit—useful. I hope you do, too.
Online social networking: How to make friends fast
Next time you meet someone at a clinical conference, don’t just hand that person a business card.
Instead, invite the colleague to join your online social network. Within days, your new acquaintance will have access to hundreds of potential business contacts-an ever-expanding network that otherwise would have taken years to build.
How online networks work
In the Internet age, people connect by meeting online in chat forums devoted to a favorite subject, exchanging e-mails after reading a mailing list or Web log, or finding relevant Web sites.
Online social networking takes this interaction one step further: Users join social networks and then invite others to join, allowing people to meet friends of friends for business or pleasure.
With popular file-sharing networks such as Kazaa and Napster, strangers can share music and other computer files. Online social networking sites work differently, but the idea is the same: to share resources.
For social purposes, these sites let users see lists of other peoples’ contacts, providing an opportunity to make new friends based on common interests. For business, interaction may be done directly by reviewing a profile or indirectly via a chain of mutual contacts in a network. As others on the network keep inviting new members and opening paths to new contact lists, your social and/or professional network will continuously grow.
How online networking can help you
Developing contacts at clinical conferences is crucial to our livelihood, but too often business cards are lost or the contact’s context is forgotten.
By contrast, with online social networking, contacts are developed and stay online. What’s more, the contact’s profile information enhances the context.
This service not only cements existing connections but may open the door to new, more worthwhile contacts. For example, the network may provide:
- a mechanism to discreetly market your services and seek job openings.
- a source of referrals for your patients who are moving to areas where you know few or no physicians. Each contact can check his or her network for area doctors. This could also lead to more patients for a doctor in that area.
Online social networking also can promote an exchange of ideas and expertise. Many large companies use this technology to solicit strategic planning ideas from their workforces. This saves companies the expense of an outside consultant.1
How to get started
Most social networking sites provide free accounts, using a valid e-mail address as the primary method of contact (Table).
Once you activate your account, you should set up a profile that highlights your interests, specialties, and types of offers you wish to receive. You are now ready to invite friends to join your network. From there, you can find other members with common interests (eg, colleagues in medical practice).
Most sites let you determine which information to make public or private, such as your e-mail address or phone number. Sites such as LinkedIn give you additional control by blocking communications from sources other than your trusted connections; you can also elect to anonymously decline requests for contact.
Some sites offer premium accounts, which for $5 to $10 a month offer services such as resume management, advanced searches, and information on who has reviewed your profile.
Risks
Some networking sites are not secure.2 This may open your social network to spam, or another user might be able to change your information. To prevent this, only use social network providers who implement SSL-level security.
Level of trust from network to network is another issue. For example, if you do not trust one colleague’s opinion, that person’s network may be not worth keeping. You may wish to keep the contact anyway because some knowledge-good or bad-may be better than no information at all.
Table
Online Networking Sites
| Site | URL |
|---|---|
| Business-oriented sites | |
| INWYK | www.itsnotwhatyouknow.com |
| www.linkedin.com | |
| Ryze | www.ryze.com |
| Spoke | www.spoke.com |
| Socially oriented sites | |
| Evite | www.evite.com |
| Friendster | www.friendster.com |
| Huminity | www.huminity.com |
| Myspace | www.myspace.com |
| Ringo | www.ringo.com |
| Tickle | www.emode.com |
Related Resources
www.ringo.com. Click on “take a tour” for a quick tutorial on online social networking.
If you have questions about these products or comments about Psyber Psychiatry, click here to contact Dr. Luo or send an e-mail to: [email protected].
Disclosure
Dr. Luo reports no financial relationship with any company whose products are mentioned in this article. The opinions expressed by Dr. Luo in this column are his own and do not necessarily reflect those of Current Psychiatry.
(accessed Jan. 12, 2004)
1. Kimball L, Rheingold H. How online social networks benefit organizations. Howard Rheingold Associates. Available at: http://www.rheingold.com/Associates/onlinenetworks.html.
2. Newitz A. Defenses lacking at social network sites. SecurityFocus. Available at: http://www.securityfocus.com/news/7739.
Next time you meet someone at a clinical conference, don’t just hand that person a business card.
Instead, invite the colleague to join your online social network. Within days, your new acquaintance will have access to hundreds of potential business contacts-an ever-expanding network that otherwise would have taken years to build.
How online networks work
In the Internet age, people connect by meeting online in chat forums devoted to a favorite subject, exchanging e-mails after reading a mailing list or Web log, or finding relevant Web sites.
Online social networking takes this interaction one step further: Users join social networks and then invite others to join, allowing people to meet friends of friends for business or pleasure.
With popular file-sharing networks such as Kazaa and Napster, strangers can share music and other computer files. Online social networking sites work differently, but the idea is the same: to share resources.
For social purposes, these sites let users see lists of other peoples’ contacts, providing an opportunity to make new friends based on common interests. For business, interaction may be done directly by reviewing a profile or indirectly via a chain of mutual contacts in a network. As others on the network keep inviting new members and opening paths to new contact lists, your social and/or professional network will continuously grow.
How online networking can help you
Developing contacts at clinical conferences is crucial to our livelihood, but too often business cards are lost or the contact’s context is forgotten.
By contrast, with online social networking, contacts are developed and stay online. What’s more, the contact’s profile information enhances the context.
This service not only cements existing connections but may open the door to new, more worthwhile contacts. For example, the network may provide:
- a mechanism to discreetly market your services and seek job openings.
- a source of referrals for your patients who are moving to areas where you know few or no physicians. Each contact can check his or her network for area doctors. This could also lead to more patients for a doctor in that area.
Online social networking also can promote an exchange of ideas and expertise. Many large companies use this technology to solicit strategic planning ideas from their workforces. This saves companies the expense of an outside consultant.1
How to get started
Most social networking sites provide free accounts, using a valid e-mail address as the primary method of contact (Table).
Once you activate your account, you should set up a profile that highlights your interests, specialties, and types of offers you wish to receive. You are now ready to invite friends to join your network. From there, you can find other members with common interests (eg, colleagues in medical practice).
Most sites let you determine which information to make public or private, such as your e-mail address or phone number. Sites such as LinkedIn give you additional control by blocking communications from sources other than your trusted connections; you can also elect to anonymously decline requests for contact.
Some sites offer premium accounts, which for $5 to $10 a month offer services such as resume management, advanced searches, and information on who has reviewed your profile.
Risks
Some networking sites are not secure.2 This may open your social network to spam, or another user might be able to change your information. To prevent this, only use social network providers who implement SSL-level security.
Level of trust from network to network is another issue. For example, if you do not trust one colleague’s opinion, that person’s network may be not worth keeping. You may wish to keep the contact anyway because some knowledge-good or bad-may be better than no information at all.
Table
Online Networking Sites
| Site | URL |
|---|---|
| Business-oriented sites | |
| INWYK | www.itsnotwhatyouknow.com |
| www.linkedin.com | |
| Ryze | www.ryze.com |
| Spoke | www.spoke.com |
| Socially oriented sites | |
| Evite | www.evite.com |
| Friendster | www.friendster.com |
| Huminity | www.huminity.com |
| Myspace | www.myspace.com |
| Ringo | www.ringo.com |
| Tickle | www.emode.com |
Related Resources
www.ringo.com. Click on “take a tour” for a quick tutorial on online social networking.
If you have questions about these products or comments about Psyber Psychiatry, click here to contact Dr. Luo or send an e-mail to: [email protected].
Disclosure
Dr. Luo reports no financial relationship with any company whose products are mentioned in this article. The opinions expressed by Dr. Luo in this column are his own and do not necessarily reflect those of Current Psychiatry.
Next time you meet someone at a clinical conference, don’t just hand that person a business card.
Instead, invite the colleague to join your online social network. Within days, your new acquaintance will have access to hundreds of potential business contacts-an ever-expanding network that otherwise would have taken years to build.
How online networks work
In the Internet age, people connect by meeting online in chat forums devoted to a favorite subject, exchanging e-mails after reading a mailing list or Web log, or finding relevant Web sites.
Online social networking takes this interaction one step further: Users join social networks and then invite others to join, allowing people to meet friends of friends for business or pleasure.
With popular file-sharing networks such as Kazaa and Napster, strangers can share music and other computer files. Online social networking sites work differently, but the idea is the same: to share resources.
For social purposes, these sites let users see lists of other peoples’ contacts, providing an opportunity to make new friends based on common interests. For business, interaction may be done directly by reviewing a profile or indirectly via a chain of mutual contacts in a network. As others on the network keep inviting new members and opening paths to new contact lists, your social and/or professional network will continuously grow.
How online networking can help you
Developing contacts at clinical conferences is crucial to our livelihood, but too often business cards are lost or the contact’s context is forgotten.
By contrast, with online social networking, contacts are developed and stay online. What’s more, the contact’s profile information enhances the context.
This service not only cements existing connections but may open the door to new, more worthwhile contacts. For example, the network may provide:
- a mechanism to discreetly market your services and seek job openings.
- a source of referrals for your patients who are moving to areas where you know few or no physicians. Each contact can check his or her network for area doctors. This could also lead to more patients for a doctor in that area.
Online social networking also can promote an exchange of ideas and expertise. Many large companies use this technology to solicit strategic planning ideas from their workforces. This saves companies the expense of an outside consultant.1
How to get started
Most social networking sites provide free accounts, using a valid e-mail address as the primary method of contact (Table).
Once you activate your account, you should set up a profile that highlights your interests, specialties, and types of offers you wish to receive. You are now ready to invite friends to join your network. From there, you can find other members with common interests (eg, colleagues in medical practice).
Most sites let you determine which information to make public or private, such as your e-mail address or phone number. Sites such as LinkedIn give you additional control by blocking communications from sources other than your trusted connections; you can also elect to anonymously decline requests for contact.
Some sites offer premium accounts, which for $5 to $10 a month offer services such as resume management, advanced searches, and information on who has reviewed your profile.
Risks
Some networking sites are not secure.2 This may open your social network to spam, or another user might be able to change your information. To prevent this, only use social network providers who implement SSL-level security.
Level of trust from network to network is another issue. For example, if you do not trust one colleague’s opinion, that person’s network may be not worth keeping. You may wish to keep the contact anyway because some knowledge-good or bad-may be better than no information at all.
Table
Online Networking Sites
| Site | URL |
|---|---|
| Business-oriented sites | |
| INWYK | www.itsnotwhatyouknow.com |
| www.linkedin.com | |
| Ryze | www.ryze.com |
| Spoke | www.spoke.com |
| Socially oriented sites | |
| Evite | www.evite.com |
| Friendster | www.friendster.com |
| Huminity | www.huminity.com |
| Myspace | www.myspace.com |
| Ringo | www.ringo.com |
| Tickle | www.emode.com |
Related Resources
www.ringo.com. Click on “take a tour” for a quick tutorial on online social networking.
If you have questions about these products or comments about Psyber Psychiatry, click here to contact Dr. Luo or send an e-mail to: [email protected].
Disclosure
Dr. Luo reports no financial relationship with any company whose products are mentioned in this article. The opinions expressed by Dr. Luo in this column are his own and do not necessarily reflect those of Current Psychiatry.
(accessed Jan. 12, 2004)
1. Kimball L, Rheingold H. How online social networks benefit organizations. Howard Rheingold Associates. Available at: http://www.rheingold.com/Associates/onlinenetworks.html.
2. Newitz A. Defenses lacking at social network sites. SecurityFocus. Available at: http://www.securityfocus.com/news/7739.
(accessed Jan. 12, 2004)
1. Kimball L, Rheingold H. How online social networks benefit organizations. Howard Rheingold Associates. Available at: http://www.rheingold.com/Associates/onlinenetworks.html.
2. Newitz A. Defenses lacking at social network sites. SecurityFocus. Available at: http://www.securityfocus.com/news/7739.
When sleep apnea mimics psychopathology
Symptoms of obstructive sleep apnea (OSA) often mimic psychopathology. Because of this, patients with OSA who exhibit these symptoms often are misdiagnosed as having a psychiatric disorder.
Consider OSA in the differential diagnosis of:
- depression. Sleep-disordered breathing is five times more prevalent in adults and children with depression than in nondepressed patients. Psychotic features also positively correlate with OSA.1
- anxiety. Physiologic and hormonal changes associated with OSA can cause panic attacks.
- attention-deficit/hyperactivity disorder (ADHD). Attention, concentration, and vigilance are often impaired in adults and children with OSA. Up to one-third of children with frequent, loud snoring display inattention and hyperactivity.2
- memory impairment. Deficits in working and long-term episodic memory are common in OSA.
- executive dysfunction. Patients with OSA often cannot sustain an organized, goal-directed, flexible approach to problem solving.
- erectile dysfunction. Pathologic processes activated by OSA may predispose men to impaired erectile function.3
- School phobia. Poor academic functioning is common in children with OSA. These children resist going to school because of a resultant loss of self-esteem. Excessive daytime sleepiness also contributes to poor academic performance.2
- Behavioral problems in children. Sleep deprivation often manifests as irritability and oppositional behavior.
Disturbances in intellectual and executive functioning are strongly correlated with hypoxemia. Deficits in vigilance, alertness, and memory correlate with measures of sleep fragmentation.4
When to suspect sleep apnea
Refer patients to a pulmonologist, ENT specialist, or sleep disorders center if the history and physical exam reveal excessive daytime sleepiness, frequent nocturia, morning headaches, nasal quality to the voice, enlarged tonsils and adenoids in children, or loud snoring or gasping sounds during sleep (consider interviewing the patient’s bed partner).
Risk factors such as family history, recessed chin, smoking, neck size >16 inches, male gender, enlarged tonsils and adenoids, and age >40 may also point to OSA. Also watch for:
- ethnicity. OSA is most prevalent among Pacific Islanders, Hispanics, and African-Americans.
- BMI >25 in adults younger than age 65. However, OSA is often missed in young people who are not obese.
1. Obayon M. The effects of breathing-related sleep disorders on mood disturbances in the general population. J Clin Psychiatry 2003;64:1195-1200.
2. O’Brien L, Gozal D. Behavioural and neurocognitive implications of snoring and obstructive sleep apnoea in children: facts and theory. Paediatr Respir Rev 2002;3:3-9.
3. Arruda-Olson AM, Olson LJ, Nehra A, Somers VK. Sleep apnea and cardiovascular disease. Implications for understanding erectile dysfunction. Herz 2003;28:298-303.
4. Salorio C, White D, Piccirillo J, et al. Learning, memory and executive control in individuals with obstructive sleep apnea syndrome. J Clin Exp Neuropsychol 2002;24:93-100.
Dr. Lundt is an affiliate faculty member, Idaho State University, Pocatello. She practices psychiatry in Boise.
Symptoms of obstructive sleep apnea (OSA) often mimic psychopathology. Because of this, patients with OSA who exhibit these symptoms often are misdiagnosed as having a psychiatric disorder.
Consider OSA in the differential diagnosis of:
- depression. Sleep-disordered breathing is five times more prevalent in adults and children with depression than in nondepressed patients. Psychotic features also positively correlate with OSA.1
- anxiety. Physiologic and hormonal changes associated with OSA can cause panic attacks.
- attention-deficit/hyperactivity disorder (ADHD). Attention, concentration, and vigilance are often impaired in adults and children with OSA. Up to one-third of children with frequent, loud snoring display inattention and hyperactivity.2
- memory impairment. Deficits in working and long-term episodic memory are common in OSA.
- executive dysfunction. Patients with OSA often cannot sustain an organized, goal-directed, flexible approach to problem solving.
- erectile dysfunction. Pathologic processes activated by OSA may predispose men to impaired erectile function.3
- School phobia. Poor academic functioning is common in children with OSA. These children resist going to school because of a resultant loss of self-esteem. Excessive daytime sleepiness also contributes to poor academic performance.2
- Behavioral problems in children. Sleep deprivation often manifests as irritability and oppositional behavior.
Disturbances in intellectual and executive functioning are strongly correlated with hypoxemia. Deficits in vigilance, alertness, and memory correlate with measures of sleep fragmentation.4
When to suspect sleep apnea
Refer patients to a pulmonologist, ENT specialist, or sleep disorders center if the history and physical exam reveal excessive daytime sleepiness, frequent nocturia, morning headaches, nasal quality to the voice, enlarged tonsils and adenoids in children, or loud snoring or gasping sounds during sleep (consider interviewing the patient’s bed partner).
Risk factors such as family history, recessed chin, smoking, neck size >16 inches, male gender, enlarged tonsils and adenoids, and age >40 may also point to OSA. Also watch for:
- ethnicity. OSA is most prevalent among Pacific Islanders, Hispanics, and African-Americans.
- BMI >25 in adults younger than age 65. However, OSA is often missed in young people who are not obese.
Symptoms of obstructive sleep apnea (OSA) often mimic psychopathology. Because of this, patients with OSA who exhibit these symptoms often are misdiagnosed as having a psychiatric disorder.
Consider OSA in the differential diagnosis of:
- depression. Sleep-disordered breathing is five times more prevalent in adults and children with depression than in nondepressed patients. Psychotic features also positively correlate with OSA.1
- anxiety. Physiologic and hormonal changes associated with OSA can cause panic attacks.
- attention-deficit/hyperactivity disorder (ADHD). Attention, concentration, and vigilance are often impaired in adults and children with OSA. Up to one-third of children with frequent, loud snoring display inattention and hyperactivity.2
- memory impairment. Deficits in working and long-term episodic memory are common in OSA.
- executive dysfunction. Patients with OSA often cannot sustain an organized, goal-directed, flexible approach to problem solving.
- erectile dysfunction. Pathologic processes activated by OSA may predispose men to impaired erectile function.3
- School phobia. Poor academic functioning is common in children with OSA. These children resist going to school because of a resultant loss of self-esteem. Excessive daytime sleepiness also contributes to poor academic performance.2
- Behavioral problems in children. Sleep deprivation often manifests as irritability and oppositional behavior.
Disturbances in intellectual and executive functioning are strongly correlated with hypoxemia. Deficits in vigilance, alertness, and memory correlate with measures of sleep fragmentation.4
When to suspect sleep apnea
Refer patients to a pulmonologist, ENT specialist, or sleep disorders center if the history and physical exam reveal excessive daytime sleepiness, frequent nocturia, morning headaches, nasal quality to the voice, enlarged tonsils and adenoids in children, or loud snoring or gasping sounds during sleep (consider interviewing the patient’s bed partner).
Risk factors such as family history, recessed chin, smoking, neck size >16 inches, male gender, enlarged tonsils and adenoids, and age >40 may also point to OSA. Also watch for:
- ethnicity. OSA is most prevalent among Pacific Islanders, Hispanics, and African-Americans.
- BMI >25 in adults younger than age 65. However, OSA is often missed in young people who are not obese.
1. Obayon M. The effects of breathing-related sleep disorders on mood disturbances in the general population. J Clin Psychiatry 2003;64:1195-1200.
2. O’Brien L, Gozal D. Behavioural and neurocognitive implications of snoring and obstructive sleep apnoea in children: facts and theory. Paediatr Respir Rev 2002;3:3-9.
3. Arruda-Olson AM, Olson LJ, Nehra A, Somers VK. Sleep apnea and cardiovascular disease. Implications for understanding erectile dysfunction. Herz 2003;28:298-303.
4. Salorio C, White D, Piccirillo J, et al. Learning, memory and executive control in individuals with obstructive sleep apnea syndrome. J Clin Exp Neuropsychol 2002;24:93-100.
Dr. Lundt is an affiliate faculty member, Idaho State University, Pocatello. She practices psychiatry in Boise.
1. Obayon M. The effects of breathing-related sleep disorders on mood disturbances in the general population. J Clin Psychiatry 2003;64:1195-1200.
2. O’Brien L, Gozal D. Behavioural and neurocognitive implications of snoring and obstructive sleep apnoea in children: facts and theory. Paediatr Respir Rev 2002;3:3-9.
3. Arruda-Olson AM, Olson LJ, Nehra A, Somers VK. Sleep apnea and cardiovascular disease. Implications for understanding erectile dysfunction. Herz 2003;28:298-303.
4. Salorio C, White D, Piccirillo J, et al. Learning, memory and executive control in individuals with obstructive sleep apnea syndrome. J Clin Exp Neuropsychol 2002;24:93-100.
Dr. Lundt is an affiliate faculty member, Idaho State University, Pocatello. She practices psychiatry in Boise.
Auto accidents and physician liability
In “Practice, not malpractice” (Current Psychiatry, December 2003), the authors note that a psychiatrist can be held liable for injuries resulting from a patient’s automobile accident.
Holding a treating psychiatrist responsible for a patient’s inability to operate a car, truck, boat, or airplane could expose us to further liability or litigation. It goes beyond our requirement to subscribe to a “standard of care”—now we must also be omniscient.
How far should a psychiatrist go to prevent a patient from driving? The literature offers few answers and many questions remain unanswered:
- Is warning the patient about medication side effects enough to reduce our risk?
- Do we also need to warn the patient’s family, employer, and the division of motor vehicles (DMV)? Or do we hold the patient’s keys until a family member arrives?
- What if the patient has no family? Shall we keep the keys indefinitely? Or do we have the police or DMV suspend the patient’s license?
I have come across several patients who have been driving for months while taking alprazolam, 12 mg/d, diazepam, 100 mg/d, or clonazepam, 10 mg/d. Countless other drivers are taking divalproex, mirtazapine, olanzapine, quetiapine, and other routinely prescribed, sedating medications. Who determines that a certain dosage is or is not affecting the patient’s ability to drive? The doctor or the driving instructors at DMV?
The issue becomes more unclear when I am asked to assess a patient’s ability to drive based on his or her poor judgment, poor attention and concentration, impulsivity, or potential for relapse into substance abuse. Does acute psychosis increase the incidence of car accidents, and if so to what extent? Is the risk sufficient to take away someone’s right to drive?
These are just some of the questions we confront when asked to assess a patient’s ability to drive. Whose job is it to judge competence behind the wheel? Because competence is task specific, I think a driving instructor is better qualified than a psychiatrist to answer this question.
Numan M. Gharaibeh, MD
Attending psychiatrist
Adult Day Treatment Program
The Institute of Living
Hartford, CT
In “Practice, not malpractice” (Current Psychiatry, December 2003), the authors note that a psychiatrist can be held liable for injuries resulting from a patient’s automobile accident.
Holding a treating psychiatrist responsible for a patient’s inability to operate a car, truck, boat, or airplane could expose us to further liability or litigation. It goes beyond our requirement to subscribe to a “standard of care”—now we must also be omniscient.
How far should a psychiatrist go to prevent a patient from driving? The literature offers few answers and many questions remain unanswered:
- Is warning the patient about medication side effects enough to reduce our risk?
- Do we also need to warn the patient’s family, employer, and the division of motor vehicles (DMV)? Or do we hold the patient’s keys until a family member arrives?
- What if the patient has no family? Shall we keep the keys indefinitely? Or do we have the police or DMV suspend the patient’s license?
I have come across several patients who have been driving for months while taking alprazolam, 12 mg/d, diazepam, 100 mg/d, or clonazepam, 10 mg/d. Countless other drivers are taking divalproex, mirtazapine, olanzapine, quetiapine, and other routinely prescribed, sedating medications. Who determines that a certain dosage is or is not affecting the patient’s ability to drive? The doctor or the driving instructors at DMV?
The issue becomes more unclear when I am asked to assess a patient’s ability to drive based on his or her poor judgment, poor attention and concentration, impulsivity, or potential for relapse into substance abuse. Does acute psychosis increase the incidence of car accidents, and if so to what extent? Is the risk sufficient to take away someone’s right to drive?
These are just some of the questions we confront when asked to assess a patient’s ability to drive. Whose job is it to judge competence behind the wheel? Because competence is task specific, I think a driving instructor is better qualified than a psychiatrist to answer this question.
Numan M. Gharaibeh, MD
Attending psychiatrist
Adult Day Treatment Program
The Institute of Living
Hartford, CT
In “Practice, not malpractice” (Current Psychiatry, December 2003), the authors note that a psychiatrist can be held liable for injuries resulting from a patient’s automobile accident.
Holding a treating psychiatrist responsible for a patient’s inability to operate a car, truck, boat, or airplane could expose us to further liability or litigation. It goes beyond our requirement to subscribe to a “standard of care”—now we must also be omniscient.
How far should a psychiatrist go to prevent a patient from driving? The literature offers few answers and many questions remain unanswered:
- Is warning the patient about medication side effects enough to reduce our risk?
- Do we also need to warn the patient’s family, employer, and the division of motor vehicles (DMV)? Or do we hold the patient’s keys until a family member arrives?
- What if the patient has no family? Shall we keep the keys indefinitely? Or do we have the police or DMV suspend the patient’s license?
I have come across several patients who have been driving for months while taking alprazolam, 12 mg/d, diazepam, 100 mg/d, or clonazepam, 10 mg/d. Countless other drivers are taking divalproex, mirtazapine, olanzapine, quetiapine, and other routinely prescribed, sedating medications. Who determines that a certain dosage is or is not affecting the patient’s ability to drive? The doctor or the driving instructors at DMV?
The issue becomes more unclear when I am asked to assess a patient’s ability to drive based on his or her poor judgment, poor attention and concentration, impulsivity, or potential for relapse into substance abuse. Does acute psychosis increase the incidence of car accidents, and if so to what extent? Is the risk sufficient to take away someone’s right to drive?
These are just some of the questions we confront when asked to assess a patient’s ability to drive. Whose job is it to judge competence behind the wheel? Because competence is task specific, I think a driving instructor is better qualified than a psychiatrist to answer this question.
Numan M. Gharaibeh, MD
Attending psychiatrist
Adult Day Treatment Program
The Institute of Living
Hartford, CT
Psychotic depression: State-of-the-art algorithm improves odds for remission
Psychotic depression requires a unique antidepressant approach, but how can you be sure that a patient’s major depression has psychotic features? Delusions or hallucinations—psychotic depression’s hallmarks—may not be obvious.
This article describes how to detect the distinctive diagnostic signs of psychosis in a patient with a major depressive episode. We offer a treatment algorithm for:
- choosing between electroconvulsive therapy (ECT) and medication
- safely combining antidepressant and antipsychotic agents
- addressing partial or nonresponse to ECT or medications.
Psychotic or nonpsychotic?
Similar clinical presentations make it difficult to distinguish psychotic depression from nonpsychotic depression, schizophrenia spectrum disorders, bipolar disorder, posttraumatic stress disorder (PTSD), obsessive-compulsive disorder (OCD), and body dysmorphic disorders. Comorbid substance abuse/dependency disorders can also complicate psychotic depression’s clinical manifestations and outcomes.
Because delusions and hallucinations are often subtle, researchers have sought other symptoms to differentiate psychotic from nonpsychotic depression. For example, patients with psychotic depression are more likely to exhibit paranoia1 (Table 1), which may explain their underreporting of symptoms.
Table 1
Diagnostic characteristics of psychotic depression
| DSM-IV hallmark symptoms |
| Delusions or hallucinations in the context of a depressive episode |
| More subtle symptoms may include: |
|
Using the Hamilton Rating Scale for Depression (HRSD), Frances and colleagues2 compared 64 depressed patients (34 with psychotic features and 30 without). On the scale’s paranoia item, the psychotic group’s mean score was 1.10, compared with 0.15 for those without psychosis (p = 0.01).
Family history and clinical course. Some studies suggest that first-degree relatives of patients with psychotic depression may have elevated rates of depression and the psychotic subtype.3 Patients with psychotic depression typically suffer morefrequent relapses or recurrences and therefore:
- use more psychiatric services
- are more disabled
- have a poorer clinical course.4
Suicide risk. Psychotic depression is associated with increased risk of self-harm and hospitalization compared with nonpsychotic depression. Patients hospitalized for a major depressive episode are five times more likely to commit suicide if they show evidence of delusions.5
Social impairment. Patients with psychotic depression often have “troubled” lives, with difficult marital and parental relationships, residential instability, inadequate support networks, and low economic status. These problems may be related to subtle cognitive deficits caused by hypothalamic-pituitary-adrenal (HPA) axis disturbance and elevated cortisol levels.6
Confronting similar presentations
Using the BPRS. The Brief Psychiatric Rating Scale (BPRS) is a useful tool to differentiate psychotic depression from nonpsychotic depression. It can flag symptoms such as suspiciousness, grandiosity, and somatization that even a seasoned psychiatrist might miss. The BPRS also points out:
- Any sign of psychosis is sufficient to designate major depression as “psychotic.”
- One well-developed diagnostic sign is sufficient to warrant treatment for psychotic depression.
Schizophrenia spectrum disorders. When psychosis is prominent (particularly in young adults), differentiating schizophrenic spectrum disorders from psychotic depression can be extremely challenging. Although few biological differences have been documented, patients with psychotic depression and schizophrenia differ in HPA axis activity and all-night sleep electroencephalogram readings.7
When the diagnosis is unclear, maintain a high index of suspicion for psychotic depression and its subtleties, and schedule frequent follow-up appointments.
Conversion to bipolar disorder. Adolescents diagnosed with unipolar major depression are at risk for converting to bipolar disorder, particularly if their depression includes psychotic features. In 60 hospitalized adolescents diagnosed with unipolar depression, a 20% conversion rate to bipolar disorder was predicted in part by a cluster of depressive symptoms:
- mood-congruent psychotic features (75% of converters vs. 6% of nonconverters, p< 0.001)
- psychomotor retardation
- rapid symptom onset.8
A similar study reported a 20% conversion rate to bipolar disorder in 206 adolescent outpatients diagnosed with unipolar depression.9 Psychotic depression was more common in converters (42%) than in nonconverters (15%).
Anxiety disorders—such as PTSD or OCD—can be difficult to distinguish from psychotic depression when they present with sensory disturbance.
When in doubt, explore:
- obsessions
- intrusive thoughts
- psychomotor behaviors
- fear of certain external events or people without consistent cues from reality.
PTSD and psychotic depression are not mutually exclusive; a patient may have both.10
Body dysmorphic disorder. Body image concerns correlate with poor self-esteem and depression.11 According to DSM-IV criteria, an individual with body dysmorphic disorder displays excessive concern over an imagined or slight defect, and this concern causes substantial distress or functional impairment. The concern also is not better accounted for by another mental disorder, such as psychotic depression or an eating disorder.
The body is often a focus of psychotic depression’s delusions. During depressive episodes, a patient may have a frank belief about a body part that is not consistent with reality. The history may include negative medical workups or preoccupation with having a serious illness.
Hypochondriasis is a characteristic of psychotic depression, and distinguishing body dysmorphic disorder and other somatoform-spectrum disorders from psychotic depression’s delusions may be difficult:
- Delusions in body dysmorphic disorder tend to be fixed over time.
- Delusions in psychotic depression tend to fluctuate in severity and may subside when the acute psychotic-depressive episode resolves.
ECT may be slightly more effective than medications for treating psychotic depression.13 ECT is not readily available in some regions, however, and the public has negative perceptions of “shock treatment.” Unfortunately, this stigma is often more influential than the evidence. According to some studies, less than 8% of U.S. psychiatrists offer ECT.14
Because medications are usually needed for maintenance treatment in psychotic depression, many clinicians choose medications over ECT as a first-line treatment. ECT should be considered as a first-line treatment for psychotic depression:
- in patients with a history of good response to ECT
- in patients with suicidal intent or severe inanition
- in older patients
- as second-line therapy for patients who fail to respond to or experience complications with medications.
Treatment recommendations
When a patient meets diagnostic criteria for psychotic depression, American Psychiatric Association practice guidelines12 recommend ECT or an antidepressant plus an antipsychotic. Although ECT may be slightly more effective than medications for treating psychotic depression, it is not readily available in many areas (Box).13,14
Medication has been shown to be effective in early studies that combined tricyclic antidepressants (TCAs) with conventional antipsychotics and in trials using selective serotonin reuptake inhibitors (SSRIs) and atypical antipsychotics.
Table 2
Medications reported effective for treating psychotic major depression
| Study, year of publication | Antipsychotic | Antidepressant |
|---|---|---|
| Double-blind studies | ||
| Spiker et al, 1985 | Perphenazine, 54 to 64 mg/d | Amitriptyline, 200 mg/d |
| Anton et al, 1990 | None | Amoxapine, 400 mg/d |
| Dube et al, 2002 | Olanzapine, 5 to 20 mg/d | Fluoxetine, 20 to 80 mg/d |
| Case reports and open-label studies | ||
| Quitkin et al, 1978 | Imipramine, 300 mg/d | None |
| Manberg et al, 1984 | Haloperidol, 20 mg/d | Bupropion, 300 mg/d |
| Nelson et al, 1986 | Perphenazine, 45 mg/d | Desipramine, 150 mg/d |
| Aronson et al, 1987 | Chlorpromazine, 1,000 mg/d | None |
| Howarth et al, 1989 | Imipramine, 248 mg/d | None |
| Rothschild et al, 1993 | Perphenazine, 32 mg/d | Fluoxetine, 40 mg/d |
| Banov et al, 1994 | Clozapine, 325 mg/d | None |
| Jacobsen, 1995 | Risperidone, 2.5 mg/d | None |
| Wolfersdorf et al, 1995 | Haloperidol, 2.5 to 10 mg/d | Paroxetine, 20 mg/d |
| Zarate et al, 2000 | Quetiapine (various) | ? (naturalistic chart review) |
Spiker and colleagues15 treated 58 patients with psychotic depression for 35 days, using amitriptyline, 200 mg/d; perphenazine, 64 mg/d; or the same dosages of amitriptyline plus perphenazine.15 Fourteen of 18 patients (78%) taking combination therapy achieved a >50% reduction in HRSD score, compared with 7 of 17 (41%) taking amitriptyline alone and 3 of 16 (19%) taking perphenazine alone.
In a more recent study,16 249 patients with psychotic depression were randomly assigned to:
- olanzapine, 5 to 20 mg/d, plus fluoxetine, 20 to 80 mg/d
- olanzapine, 5 to 20 mg/d, plus placebo
- or placebo.
Patients receiving olanzapine plus fluoxetine showed greater improvement in HRSD scores, compared with olanzapine monotherapy or placebo. Anecdotal reports indicate that quetiapine, risperidone, or olanzapine may be effective for patients with psychotic depression.17,18
We usually start with an SSRI plus an atypical antipsychotic (Algorithm). The atypicals have fewer side effects than conventional antipsychotics and may offer intrinsic antidepressant qualities through their effects on serotonin type-2 receptors. Table 2 lists recommended dosages.
When initial treatment fails
Consider second- and third-line options when patients fail to achieve remission with ECT or an SSRI plus an atypical antipsychotic. Document that first-line trials were of sufficient duration (8 to 12 weeks) and dosage.
We define remission as:
- HRSD score of <10 for at least 2 weeks
- score of 1 (no delusions or hallucinations) on the Schedule for Affective Disorders and Schizophrenia (SADS)
- and no longer meeting full criteria for a major depressive episode on the Structured Clinical Interview for DSM-IV.
We define partial remission as:
- HRSD score between 11 and 17
- HRSD improvement of >30% from baseline
- score of 1 on the SADS
- and no longer meeting full DSM-IV criteria for a major depressive episode.
Algorithm State-of-the-art treatment of psychotic major depression
Lithium. We suggest adding lithium when patients respond partially to an SSRI/atypical antipsychotic combination. Although limited evidence supports lithium augmentation of antidepressants for psychotic depression, this strategy is often used.
Adding lithium to an antidepressant/antipsychotic combination was examined in a retrospective chart review of patients treated for bipolar and unipolar psychotic depression.19 Lithium, 600 to 1,200 mg/d, was added when patients did not respond to desipramine, 150 mg/d, plus either perphenazine, 12 to 64 mg/d, or haloperidol, 4 to 20 mg/d. Eight of nine patients with bipolar psychotic depression achieved remission with the added lithium, compared with 3 of 12 patients with unipolar psychotic depression (p = 0.003).
To our knowledge, no data indicate how long to continue lithium augmentation. We start older adults on 300 mg/d and younger adults on 600 mg/d and increase by 300 mg per week. Target serum levels are 0.5 to 0.8 mEq/L, and maximum dosage is 1,200 mg/d for young adults and 900 mg/d for frail or elderly patients. We follow thyroid, renal, and hydration status and monitor for weight gain, tremors, cognitive slowing, and GI disturbances.
Other second-line options. Sufficient data support using the second-line drugs in our algorithm as first-line agents. However, the second-line agents pose a greater risk of adverse effects and decreased tolerability than SSRIs plus atypical antipsychotics. Second-line options include:
- SSRIs plus conventional antipsychotics
- amoxapine, a derivative of the conventional antipsychotic loxapine
- venlafaxine or TCAs plus atypical antipsychotics.
As with first-line therapy, 8 to 12 weeks is an adequate trial for second-line medications. ECT may be considered for patients who fail to respond to medications or experience complications.
SSRI/conventional antipsychotic. Our group used fluoxetine, 20 to 40 mg/d, plus perphenazine, 32 mg/d, in the first study of combined SSRI/conventional antipsychotic therapy for patients with psychotic depression.20 After 5 weeks, 22 of 30 patients’ HRSD and BPRS scores were reduced by >50%.
Amoxapine monotherapy. Anton and Burch21 compared amoxapine, 400 mg/d, with amitriptyline, 200 mg/d, plus perphenazine, 32 mg/d. Response rates (>50% reduction on the HRSD) were 71% and 81% for the two groups, respectively. Extrapyramidal symptoms (EPS) were more frequent with the combination therapy.
Venlafaxine’s mechanism of action is thought to be similar to that of TCAs, and we know from the Spiker study15 that TCAs are effective in treating psychotic depression. To our knowledge, venlafaxine dosages for psychotic depression have not been studied; 75 to 375 mg/d is recommended for nonpsychotic depression. Potential side effects include insomnia, nervousness, nausea, headache, dry mouth, fatigue, and elevations of supine diastolic blood pressure.
Third-line therapy. Clozapine may be considered when second-line options do not achieve adequate results.22 When making this choice, consider the need for biweekly blood monitoring and the risk of serious side effects such as agranulocytosis and seizures.
Maintenance therapy
Psychotic depression has a higher relapse rate than nonpsychotic depression. Relapse rates are 50 to 92% in patients with psychotic depression, and recurrence often develops within 2 to 14 months after recovery from the index episode.23 With little data on which to base a maintenance regimen, we recommend that you continue antipsychotics for 4 months after the acute episode resolves.
Recently our group reported that after a taper of perphenazine—following 4 months of treatment with fluoxetine and perphenazine—22 of 30 patients (73%) showed no signs of relapse over the next 11 months.24 We usually maintain patients on antidepressants indefinitely.
Related resources
- DeBattista C, Rothschild AJ, Schatzberg AF. A dynamic algorithm for the treatment of psychotic major depression. Psychiatric Ann 2002;32:681-91.
- Rothschild AJ. Challenges in the treatment of depression with psychotic features. Biol Psychiatry 2003;53:680-90.
- National Institutes of Health. http://www.clinicaltrials.gov. Enter “Medication treatment for psychotic depression” in “Search clinical trials” field, then click on appropriate link.
Drug brand names
- Amitriptyline • Elavil
- Amoxapine • Asendin
- Bupropion • Wellbutrin
- Chlorpromazine • Thorazine
- Clozapine • Clozaril
- Desipramine • Norpramin
- Fluoxetine • Prozac
- Imipramine • Tofranil
- Loxapine • Loxitane
- Olanzapine • Zyprexa
- Paroxetine • Paxil
- Perphenazine • Trilafon
- Risperidone • Risperdal
- Quetiapine • Seroquel
- Venlafaxine • Effexor
Disclosure
Dr. Bell reports no financial relationship with any company whose products are mentioned in this article or with manufacturers of competing products.
Dr. Rothschild receives research support from Bristol-Myers Squibb, Eli Lilly and Co., Merck & Co., Wyeth Pharmaceuticals, and the National Institute of Mental Health. He is a consultant to and/or speaker for Forest Pharmaceuticals, Eli Lilly and Co., Abbott Laboratories, Bristol-Myers Squibb, and Pfizer Inc. In the past, he has been a consultant to and received research grants from Corcept Therapeutics.
1. Lykouras E, Malliaras D, Christodoulou GN, et al. Delusional depression: phenomenology and response to treatment, a prospective study. Acta Psychiatry Scand 1986;73:324-9.
2. Frances A, Brown RP, Kocsis JG, Mann JJ. Psychotic depression: a separate entity? Am J Psychiatry 1981;138:831-3.
3. Leckman JF, Weissman MM, Prusoff BA, et al. Subtypes of depression: family study perspective. Arch Gen Psychiatry 1984;41:833-9.
4. Coryell W, Leon A, Winokur G, et al. The importance of psychotic features to long-term course in depressive disorders. Am J Psychiatry 1996;153:483-9.
5. Roose SP, Glassman AH, Walsh BT, et al. Depression, delusions, and suicide. Am J Psychiatry 1983;140:1159-62.
6. Rothschild AJ, Samson JA, Bond TC, et al. Hypothalamic-pituitary-adrenal axis activity and one-year outcome in depression. Biol Psychiatry 1993;34:392-400.
7. Rothschild AJ, Schatzberg AF. Diagnosis and treatment of psychotic (delusional) depression. In: Grunhaus L, Greden JF (eds). Severe depressive disorder. Washington, DC: American Psychiatric Press, 1994;195-207.
8. Strober M, Carlson G. Bipolar illness in adolescents with major depression: Clinical, genetic, and psychopharmacologic predictors in a three to four year prospective follow-up investigation. Arch Gen Psychiatry 1982;39:549-55.
9. Akiskal HS, Walker P, Puzantian VR, et al. Bipolar outcome in the course of depressive illness: Phenomenological, familial, and pharmacologic predictors. J Affect Disord 1983;5:115-28.
10. Zimmerman M, Mattia JI. Psychotic subtyping of major depressive disorder and posttraumatic stress disorder. J Clin Psychiatry 1999;60(5):311-4.
11. Buhne A, Keuthen N. Prevalence of symptoms of body dysmorphic disorder and its correlates: a cross-cultural comparison. Psychosomatics 2002;43:486-90.
12. American Psychiatric Association Practice guidelines for the treatment of major depressive disorder (revision). Am J Psychiatry 200;157:(suppl 4).
13. Parker G, Roy K, Hadzi-Pavlovic D, Pedic F. Psychotic (delusional) depression: A meta-analysis of physical treatments. J Affect Disord 1992;24:17-24.
14. Thompson JW, Weiner RD, Myers CP. Use of ECT in the United States in 1975, 1980 and 1986. Am J Psychiatry 1994;151:1657-61.
15. Spiker DG, Weiss JC, Dealy RS, et al. The pharmacological treatment of delusional depression. Am J Psychiatry 1985;142:430-6.
16. Dube S, Rothschild A, Andersen SE, et al. Olanzapine-fluoxetine combination for psychotic depression (presentation). Barcelona, Spain: European College of Neuropsychopharmacology, 2002.
17. Hillert A, Maier W, Wetzel H, Benkert O. Risperidone in the treatment of disorders with a combined psychotic and depressive syndrome: a functional approach. Pharmacopsychiatry 1992;25:213-17.
18. Zarate CA, Jr, Rothschild AJ, Fletcher KE, et al. Clinical predictors of acute response with quetiapine in psychotic mood disorder. J Clin Psychiatry 2000;61:185-9.
19. Nelson JC, Mazure CM. Lithium augmentation in psychotic depression refractory to combined drug treatment. Am J Psychiatry 1986;143:363-6.
20. Rothschild AJ, Samson JA, Bessette MP, Carter-Campbell JT. Efficacy of combination fluoxetine and perphenazine in the treatment of psychotic depression. J Clin Psychiatry 1993;54:338-42.
21. Anton RF, Jr, Burch EA, Jr. Amoxapine versus amitriptyline combined with perphenazine in the treatment of psychotic depression. Am J Psychiatry 1990;147:1203-8.
22. Banov MD, Zarate CA, Jr, Tohen M, et al. Clozapine therapy in refractory affective disorder: polarity predicts response in long-term follow-up. J Clin Psychiatry 1994;55:295-300.
23. Aronson TA, Shukla S, Gujavarty K, et al. Relapse in delusional depression: a retrospective study of the course of treatment. Comp Psychiatry 1988;29:12-21.
24. Rothschild AJ, Duval SE. How long should patients with psychotic depression stay on the antipsychotic medication. J Clin Psychiatry 2003;64:390-6.
Psychotic depression requires a unique antidepressant approach, but how can you be sure that a patient’s major depression has psychotic features? Delusions or hallucinations—psychotic depression’s hallmarks—may not be obvious.
This article describes how to detect the distinctive diagnostic signs of psychosis in a patient with a major depressive episode. We offer a treatment algorithm for:
- choosing between electroconvulsive therapy (ECT) and medication
- safely combining antidepressant and antipsychotic agents
- addressing partial or nonresponse to ECT or medications.
Psychotic or nonpsychotic?
Similar clinical presentations make it difficult to distinguish psychotic depression from nonpsychotic depression, schizophrenia spectrum disorders, bipolar disorder, posttraumatic stress disorder (PTSD), obsessive-compulsive disorder (OCD), and body dysmorphic disorders. Comorbid substance abuse/dependency disorders can also complicate psychotic depression’s clinical manifestations and outcomes.
Because delusions and hallucinations are often subtle, researchers have sought other symptoms to differentiate psychotic from nonpsychotic depression. For example, patients with psychotic depression are more likely to exhibit paranoia1 (Table 1), which may explain their underreporting of symptoms.
Table 1
Diagnostic characteristics of psychotic depression
| DSM-IV hallmark symptoms |
| Delusions or hallucinations in the context of a depressive episode |
| More subtle symptoms may include: |
|
Using the Hamilton Rating Scale for Depression (HRSD), Frances and colleagues2 compared 64 depressed patients (34 with psychotic features and 30 without). On the scale’s paranoia item, the psychotic group’s mean score was 1.10, compared with 0.15 for those without psychosis (p = 0.01).
Family history and clinical course. Some studies suggest that first-degree relatives of patients with psychotic depression may have elevated rates of depression and the psychotic subtype.3 Patients with psychotic depression typically suffer morefrequent relapses or recurrences and therefore:
- use more psychiatric services
- are more disabled
- have a poorer clinical course.4
Suicide risk. Psychotic depression is associated with increased risk of self-harm and hospitalization compared with nonpsychotic depression. Patients hospitalized for a major depressive episode are five times more likely to commit suicide if they show evidence of delusions.5
Social impairment. Patients with psychotic depression often have “troubled” lives, with difficult marital and parental relationships, residential instability, inadequate support networks, and low economic status. These problems may be related to subtle cognitive deficits caused by hypothalamic-pituitary-adrenal (HPA) axis disturbance and elevated cortisol levels.6
Confronting similar presentations
Using the BPRS. The Brief Psychiatric Rating Scale (BPRS) is a useful tool to differentiate psychotic depression from nonpsychotic depression. It can flag symptoms such as suspiciousness, grandiosity, and somatization that even a seasoned psychiatrist might miss. The BPRS also points out:
- Any sign of psychosis is sufficient to designate major depression as “psychotic.”
- One well-developed diagnostic sign is sufficient to warrant treatment for psychotic depression.
Schizophrenia spectrum disorders. When psychosis is prominent (particularly in young adults), differentiating schizophrenic spectrum disorders from psychotic depression can be extremely challenging. Although few biological differences have been documented, patients with psychotic depression and schizophrenia differ in HPA axis activity and all-night sleep electroencephalogram readings.7
When the diagnosis is unclear, maintain a high index of suspicion for psychotic depression and its subtleties, and schedule frequent follow-up appointments.
Conversion to bipolar disorder. Adolescents diagnosed with unipolar major depression are at risk for converting to bipolar disorder, particularly if their depression includes psychotic features. In 60 hospitalized adolescents diagnosed with unipolar depression, a 20% conversion rate to bipolar disorder was predicted in part by a cluster of depressive symptoms:
- mood-congruent psychotic features (75% of converters vs. 6% of nonconverters, p< 0.001)
- psychomotor retardation
- rapid symptom onset.8
A similar study reported a 20% conversion rate to bipolar disorder in 206 adolescent outpatients diagnosed with unipolar depression.9 Psychotic depression was more common in converters (42%) than in nonconverters (15%).
Anxiety disorders—such as PTSD or OCD—can be difficult to distinguish from psychotic depression when they present with sensory disturbance.
When in doubt, explore:
- obsessions
- intrusive thoughts
- psychomotor behaviors
- fear of certain external events or people without consistent cues from reality.
PTSD and psychotic depression are not mutually exclusive; a patient may have both.10
Body dysmorphic disorder. Body image concerns correlate with poor self-esteem and depression.11 According to DSM-IV criteria, an individual with body dysmorphic disorder displays excessive concern over an imagined or slight defect, and this concern causes substantial distress or functional impairment. The concern also is not better accounted for by another mental disorder, such as psychotic depression or an eating disorder.
The body is often a focus of psychotic depression’s delusions. During depressive episodes, a patient may have a frank belief about a body part that is not consistent with reality. The history may include negative medical workups or preoccupation with having a serious illness.
Hypochondriasis is a characteristic of psychotic depression, and distinguishing body dysmorphic disorder and other somatoform-spectrum disorders from psychotic depression’s delusions may be difficult:
- Delusions in body dysmorphic disorder tend to be fixed over time.
- Delusions in psychotic depression tend to fluctuate in severity and may subside when the acute psychotic-depressive episode resolves.
ECT may be slightly more effective than medications for treating psychotic depression.13 ECT is not readily available in some regions, however, and the public has negative perceptions of “shock treatment.” Unfortunately, this stigma is often more influential than the evidence. According to some studies, less than 8% of U.S. psychiatrists offer ECT.14
Because medications are usually needed for maintenance treatment in psychotic depression, many clinicians choose medications over ECT as a first-line treatment. ECT should be considered as a first-line treatment for psychotic depression:
- in patients with a history of good response to ECT
- in patients with suicidal intent or severe inanition
- in older patients
- as second-line therapy for patients who fail to respond to or experience complications with medications.
Treatment recommendations
When a patient meets diagnostic criteria for psychotic depression, American Psychiatric Association practice guidelines12 recommend ECT or an antidepressant plus an antipsychotic. Although ECT may be slightly more effective than medications for treating psychotic depression, it is not readily available in many areas (Box).13,14
Medication has been shown to be effective in early studies that combined tricyclic antidepressants (TCAs) with conventional antipsychotics and in trials using selective serotonin reuptake inhibitors (SSRIs) and atypical antipsychotics.
Table 2
Medications reported effective for treating psychotic major depression
| Study, year of publication | Antipsychotic | Antidepressant |
|---|---|---|
| Double-blind studies | ||
| Spiker et al, 1985 | Perphenazine, 54 to 64 mg/d | Amitriptyline, 200 mg/d |
| Anton et al, 1990 | None | Amoxapine, 400 mg/d |
| Dube et al, 2002 | Olanzapine, 5 to 20 mg/d | Fluoxetine, 20 to 80 mg/d |
| Case reports and open-label studies | ||
| Quitkin et al, 1978 | Imipramine, 300 mg/d | None |
| Manberg et al, 1984 | Haloperidol, 20 mg/d | Bupropion, 300 mg/d |
| Nelson et al, 1986 | Perphenazine, 45 mg/d | Desipramine, 150 mg/d |
| Aronson et al, 1987 | Chlorpromazine, 1,000 mg/d | None |
| Howarth et al, 1989 | Imipramine, 248 mg/d | None |
| Rothschild et al, 1993 | Perphenazine, 32 mg/d | Fluoxetine, 40 mg/d |
| Banov et al, 1994 | Clozapine, 325 mg/d | None |
| Jacobsen, 1995 | Risperidone, 2.5 mg/d | None |
| Wolfersdorf et al, 1995 | Haloperidol, 2.5 to 10 mg/d | Paroxetine, 20 mg/d |
| Zarate et al, 2000 | Quetiapine (various) | ? (naturalistic chart review) |
Spiker and colleagues15 treated 58 patients with psychotic depression for 35 days, using amitriptyline, 200 mg/d; perphenazine, 64 mg/d; or the same dosages of amitriptyline plus perphenazine.15 Fourteen of 18 patients (78%) taking combination therapy achieved a >50% reduction in HRSD score, compared with 7 of 17 (41%) taking amitriptyline alone and 3 of 16 (19%) taking perphenazine alone.
In a more recent study,16 249 patients with psychotic depression were randomly assigned to:
- olanzapine, 5 to 20 mg/d, plus fluoxetine, 20 to 80 mg/d
- olanzapine, 5 to 20 mg/d, plus placebo
- or placebo.
Patients receiving olanzapine plus fluoxetine showed greater improvement in HRSD scores, compared with olanzapine monotherapy or placebo. Anecdotal reports indicate that quetiapine, risperidone, or olanzapine may be effective for patients with psychotic depression.17,18
We usually start with an SSRI plus an atypical antipsychotic (Algorithm). The atypicals have fewer side effects than conventional antipsychotics and may offer intrinsic antidepressant qualities through their effects on serotonin type-2 receptors. Table 2 lists recommended dosages.
When initial treatment fails
Consider second- and third-line options when patients fail to achieve remission with ECT or an SSRI plus an atypical antipsychotic. Document that first-line trials were of sufficient duration (8 to 12 weeks) and dosage.
We define remission as:
- HRSD score of <10 for at least 2 weeks
- score of 1 (no delusions or hallucinations) on the Schedule for Affective Disorders and Schizophrenia (SADS)
- and no longer meeting full criteria for a major depressive episode on the Structured Clinical Interview for DSM-IV.
We define partial remission as:
- HRSD score between 11 and 17
- HRSD improvement of >30% from baseline
- score of 1 on the SADS
- and no longer meeting full DSM-IV criteria for a major depressive episode.
Algorithm State-of-the-art treatment of psychotic major depression
Lithium. We suggest adding lithium when patients respond partially to an SSRI/atypical antipsychotic combination. Although limited evidence supports lithium augmentation of antidepressants for psychotic depression, this strategy is often used.
Adding lithium to an antidepressant/antipsychotic combination was examined in a retrospective chart review of patients treated for bipolar and unipolar psychotic depression.19 Lithium, 600 to 1,200 mg/d, was added when patients did not respond to desipramine, 150 mg/d, plus either perphenazine, 12 to 64 mg/d, or haloperidol, 4 to 20 mg/d. Eight of nine patients with bipolar psychotic depression achieved remission with the added lithium, compared with 3 of 12 patients with unipolar psychotic depression (p = 0.003).
To our knowledge, no data indicate how long to continue lithium augmentation. We start older adults on 300 mg/d and younger adults on 600 mg/d and increase by 300 mg per week. Target serum levels are 0.5 to 0.8 mEq/L, and maximum dosage is 1,200 mg/d for young adults and 900 mg/d for frail or elderly patients. We follow thyroid, renal, and hydration status and monitor for weight gain, tremors, cognitive slowing, and GI disturbances.
Other second-line options. Sufficient data support using the second-line drugs in our algorithm as first-line agents. However, the second-line agents pose a greater risk of adverse effects and decreased tolerability than SSRIs plus atypical antipsychotics. Second-line options include:
- SSRIs plus conventional antipsychotics
- amoxapine, a derivative of the conventional antipsychotic loxapine
- venlafaxine or TCAs plus atypical antipsychotics.
As with first-line therapy, 8 to 12 weeks is an adequate trial for second-line medications. ECT may be considered for patients who fail to respond to medications or experience complications.
SSRI/conventional antipsychotic. Our group used fluoxetine, 20 to 40 mg/d, plus perphenazine, 32 mg/d, in the first study of combined SSRI/conventional antipsychotic therapy for patients with psychotic depression.20 After 5 weeks, 22 of 30 patients’ HRSD and BPRS scores were reduced by >50%.
Amoxapine monotherapy. Anton and Burch21 compared amoxapine, 400 mg/d, with amitriptyline, 200 mg/d, plus perphenazine, 32 mg/d. Response rates (>50% reduction on the HRSD) were 71% and 81% for the two groups, respectively. Extrapyramidal symptoms (EPS) were more frequent with the combination therapy.
Venlafaxine’s mechanism of action is thought to be similar to that of TCAs, and we know from the Spiker study15 that TCAs are effective in treating psychotic depression. To our knowledge, venlafaxine dosages for psychotic depression have not been studied; 75 to 375 mg/d is recommended for nonpsychotic depression. Potential side effects include insomnia, nervousness, nausea, headache, dry mouth, fatigue, and elevations of supine diastolic blood pressure.
Third-line therapy. Clozapine may be considered when second-line options do not achieve adequate results.22 When making this choice, consider the need for biweekly blood monitoring and the risk of serious side effects such as agranulocytosis and seizures.
Maintenance therapy
Psychotic depression has a higher relapse rate than nonpsychotic depression. Relapse rates are 50 to 92% in patients with psychotic depression, and recurrence often develops within 2 to 14 months after recovery from the index episode.23 With little data on which to base a maintenance regimen, we recommend that you continue antipsychotics for 4 months after the acute episode resolves.
Recently our group reported that after a taper of perphenazine—following 4 months of treatment with fluoxetine and perphenazine—22 of 30 patients (73%) showed no signs of relapse over the next 11 months.24 We usually maintain patients on antidepressants indefinitely.
Related resources
- DeBattista C, Rothschild AJ, Schatzberg AF. A dynamic algorithm for the treatment of psychotic major depression. Psychiatric Ann 2002;32:681-91.
- Rothschild AJ. Challenges in the treatment of depression with psychotic features. Biol Psychiatry 2003;53:680-90.
- National Institutes of Health. http://www.clinicaltrials.gov. Enter “Medication treatment for psychotic depression” in “Search clinical trials” field, then click on appropriate link.
Drug brand names
- Amitriptyline • Elavil
- Amoxapine • Asendin
- Bupropion • Wellbutrin
- Chlorpromazine • Thorazine
- Clozapine • Clozaril
- Desipramine • Norpramin
- Fluoxetine • Prozac
- Imipramine • Tofranil
- Loxapine • Loxitane
- Olanzapine • Zyprexa
- Paroxetine • Paxil
- Perphenazine • Trilafon
- Risperidone • Risperdal
- Quetiapine • Seroquel
- Venlafaxine • Effexor
Disclosure
Dr. Bell reports no financial relationship with any company whose products are mentioned in this article or with manufacturers of competing products.
Dr. Rothschild receives research support from Bristol-Myers Squibb, Eli Lilly and Co., Merck & Co., Wyeth Pharmaceuticals, and the National Institute of Mental Health. He is a consultant to and/or speaker for Forest Pharmaceuticals, Eli Lilly and Co., Abbott Laboratories, Bristol-Myers Squibb, and Pfizer Inc. In the past, he has been a consultant to and received research grants from Corcept Therapeutics.
Psychotic depression requires a unique antidepressant approach, but how can you be sure that a patient’s major depression has psychotic features? Delusions or hallucinations—psychotic depression’s hallmarks—may not be obvious.
This article describes how to detect the distinctive diagnostic signs of psychosis in a patient with a major depressive episode. We offer a treatment algorithm for:
- choosing between electroconvulsive therapy (ECT) and medication
- safely combining antidepressant and antipsychotic agents
- addressing partial or nonresponse to ECT or medications.
Psychotic or nonpsychotic?
Similar clinical presentations make it difficult to distinguish psychotic depression from nonpsychotic depression, schizophrenia spectrum disorders, bipolar disorder, posttraumatic stress disorder (PTSD), obsessive-compulsive disorder (OCD), and body dysmorphic disorders. Comorbid substance abuse/dependency disorders can also complicate psychotic depression’s clinical manifestations and outcomes.
Because delusions and hallucinations are often subtle, researchers have sought other symptoms to differentiate psychotic from nonpsychotic depression. For example, patients with psychotic depression are more likely to exhibit paranoia1 (Table 1), which may explain their underreporting of symptoms.
Table 1
Diagnostic characteristics of psychotic depression
| DSM-IV hallmark symptoms |
| Delusions or hallucinations in the context of a depressive episode |
| More subtle symptoms may include: |
|
Using the Hamilton Rating Scale for Depression (HRSD), Frances and colleagues2 compared 64 depressed patients (34 with psychotic features and 30 without). On the scale’s paranoia item, the psychotic group’s mean score was 1.10, compared with 0.15 for those without psychosis (p = 0.01).
Family history and clinical course. Some studies suggest that first-degree relatives of patients with psychotic depression may have elevated rates of depression and the psychotic subtype.3 Patients with psychotic depression typically suffer morefrequent relapses or recurrences and therefore:
- use more psychiatric services
- are more disabled
- have a poorer clinical course.4
Suicide risk. Psychotic depression is associated with increased risk of self-harm and hospitalization compared with nonpsychotic depression. Patients hospitalized for a major depressive episode are five times more likely to commit suicide if they show evidence of delusions.5
Social impairment. Patients with psychotic depression often have “troubled” lives, with difficult marital and parental relationships, residential instability, inadequate support networks, and low economic status. These problems may be related to subtle cognitive deficits caused by hypothalamic-pituitary-adrenal (HPA) axis disturbance and elevated cortisol levels.6
Confronting similar presentations
Using the BPRS. The Brief Psychiatric Rating Scale (BPRS) is a useful tool to differentiate psychotic depression from nonpsychotic depression. It can flag symptoms such as suspiciousness, grandiosity, and somatization that even a seasoned psychiatrist might miss. The BPRS also points out:
- Any sign of psychosis is sufficient to designate major depression as “psychotic.”
- One well-developed diagnostic sign is sufficient to warrant treatment for psychotic depression.
Schizophrenia spectrum disorders. When psychosis is prominent (particularly in young adults), differentiating schizophrenic spectrum disorders from psychotic depression can be extremely challenging. Although few biological differences have been documented, patients with psychotic depression and schizophrenia differ in HPA axis activity and all-night sleep electroencephalogram readings.7
When the diagnosis is unclear, maintain a high index of suspicion for psychotic depression and its subtleties, and schedule frequent follow-up appointments.
Conversion to bipolar disorder. Adolescents diagnosed with unipolar major depression are at risk for converting to bipolar disorder, particularly if their depression includes psychotic features. In 60 hospitalized adolescents diagnosed with unipolar depression, a 20% conversion rate to bipolar disorder was predicted in part by a cluster of depressive symptoms:
- mood-congruent psychotic features (75% of converters vs. 6% of nonconverters, p< 0.001)
- psychomotor retardation
- rapid symptom onset.8
A similar study reported a 20% conversion rate to bipolar disorder in 206 adolescent outpatients diagnosed with unipolar depression.9 Psychotic depression was more common in converters (42%) than in nonconverters (15%).
Anxiety disorders—such as PTSD or OCD—can be difficult to distinguish from psychotic depression when they present with sensory disturbance.
When in doubt, explore:
- obsessions
- intrusive thoughts
- psychomotor behaviors
- fear of certain external events or people without consistent cues from reality.
PTSD and psychotic depression are not mutually exclusive; a patient may have both.10
Body dysmorphic disorder. Body image concerns correlate with poor self-esteem and depression.11 According to DSM-IV criteria, an individual with body dysmorphic disorder displays excessive concern over an imagined or slight defect, and this concern causes substantial distress or functional impairment. The concern also is not better accounted for by another mental disorder, such as psychotic depression or an eating disorder.
The body is often a focus of psychotic depression’s delusions. During depressive episodes, a patient may have a frank belief about a body part that is not consistent with reality. The history may include negative medical workups or preoccupation with having a serious illness.
Hypochondriasis is a characteristic of psychotic depression, and distinguishing body dysmorphic disorder and other somatoform-spectrum disorders from psychotic depression’s delusions may be difficult:
- Delusions in body dysmorphic disorder tend to be fixed over time.
- Delusions in psychotic depression tend to fluctuate in severity and may subside when the acute psychotic-depressive episode resolves.
ECT may be slightly more effective than medications for treating psychotic depression.13 ECT is not readily available in some regions, however, and the public has negative perceptions of “shock treatment.” Unfortunately, this stigma is often more influential than the evidence. According to some studies, less than 8% of U.S. psychiatrists offer ECT.14
Because medications are usually needed for maintenance treatment in psychotic depression, many clinicians choose medications over ECT as a first-line treatment. ECT should be considered as a first-line treatment for psychotic depression:
- in patients with a history of good response to ECT
- in patients with suicidal intent or severe inanition
- in older patients
- as second-line therapy for patients who fail to respond to or experience complications with medications.
Treatment recommendations
When a patient meets diagnostic criteria for psychotic depression, American Psychiatric Association practice guidelines12 recommend ECT or an antidepressant plus an antipsychotic. Although ECT may be slightly more effective than medications for treating psychotic depression, it is not readily available in many areas (Box).13,14
Medication has been shown to be effective in early studies that combined tricyclic antidepressants (TCAs) with conventional antipsychotics and in trials using selective serotonin reuptake inhibitors (SSRIs) and atypical antipsychotics.
Table 2
Medications reported effective for treating psychotic major depression
| Study, year of publication | Antipsychotic | Antidepressant |
|---|---|---|
| Double-blind studies | ||
| Spiker et al, 1985 | Perphenazine, 54 to 64 mg/d | Amitriptyline, 200 mg/d |
| Anton et al, 1990 | None | Amoxapine, 400 mg/d |
| Dube et al, 2002 | Olanzapine, 5 to 20 mg/d | Fluoxetine, 20 to 80 mg/d |
| Case reports and open-label studies | ||
| Quitkin et al, 1978 | Imipramine, 300 mg/d | None |
| Manberg et al, 1984 | Haloperidol, 20 mg/d | Bupropion, 300 mg/d |
| Nelson et al, 1986 | Perphenazine, 45 mg/d | Desipramine, 150 mg/d |
| Aronson et al, 1987 | Chlorpromazine, 1,000 mg/d | None |
| Howarth et al, 1989 | Imipramine, 248 mg/d | None |
| Rothschild et al, 1993 | Perphenazine, 32 mg/d | Fluoxetine, 40 mg/d |
| Banov et al, 1994 | Clozapine, 325 mg/d | None |
| Jacobsen, 1995 | Risperidone, 2.5 mg/d | None |
| Wolfersdorf et al, 1995 | Haloperidol, 2.5 to 10 mg/d | Paroxetine, 20 mg/d |
| Zarate et al, 2000 | Quetiapine (various) | ? (naturalistic chart review) |
Spiker and colleagues15 treated 58 patients with psychotic depression for 35 days, using amitriptyline, 200 mg/d; perphenazine, 64 mg/d; or the same dosages of amitriptyline plus perphenazine.15 Fourteen of 18 patients (78%) taking combination therapy achieved a >50% reduction in HRSD score, compared with 7 of 17 (41%) taking amitriptyline alone and 3 of 16 (19%) taking perphenazine alone.
In a more recent study,16 249 patients with psychotic depression were randomly assigned to:
- olanzapine, 5 to 20 mg/d, plus fluoxetine, 20 to 80 mg/d
- olanzapine, 5 to 20 mg/d, plus placebo
- or placebo.
Patients receiving olanzapine plus fluoxetine showed greater improvement in HRSD scores, compared with olanzapine monotherapy or placebo. Anecdotal reports indicate that quetiapine, risperidone, or olanzapine may be effective for patients with psychotic depression.17,18
We usually start with an SSRI plus an atypical antipsychotic (Algorithm). The atypicals have fewer side effects than conventional antipsychotics and may offer intrinsic antidepressant qualities through their effects on serotonin type-2 receptors. Table 2 lists recommended dosages.
When initial treatment fails
Consider second- and third-line options when patients fail to achieve remission with ECT or an SSRI plus an atypical antipsychotic. Document that first-line trials were of sufficient duration (8 to 12 weeks) and dosage.
We define remission as:
- HRSD score of <10 for at least 2 weeks
- score of 1 (no delusions or hallucinations) on the Schedule for Affective Disorders and Schizophrenia (SADS)
- and no longer meeting full criteria for a major depressive episode on the Structured Clinical Interview for DSM-IV.
We define partial remission as:
- HRSD score between 11 and 17
- HRSD improvement of >30% from baseline
- score of 1 on the SADS
- and no longer meeting full DSM-IV criteria for a major depressive episode.
Algorithm State-of-the-art treatment of psychotic major depression
Lithium. We suggest adding lithium when patients respond partially to an SSRI/atypical antipsychotic combination. Although limited evidence supports lithium augmentation of antidepressants for psychotic depression, this strategy is often used.
Adding lithium to an antidepressant/antipsychotic combination was examined in a retrospective chart review of patients treated for bipolar and unipolar psychotic depression.19 Lithium, 600 to 1,200 mg/d, was added when patients did not respond to desipramine, 150 mg/d, plus either perphenazine, 12 to 64 mg/d, or haloperidol, 4 to 20 mg/d. Eight of nine patients with bipolar psychotic depression achieved remission with the added lithium, compared with 3 of 12 patients with unipolar psychotic depression (p = 0.003).
To our knowledge, no data indicate how long to continue lithium augmentation. We start older adults on 300 mg/d and younger adults on 600 mg/d and increase by 300 mg per week. Target serum levels are 0.5 to 0.8 mEq/L, and maximum dosage is 1,200 mg/d for young adults and 900 mg/d for frail or elderly patients. We follow thyroid, renal, and hydration status and monitor for weight gain, tremors, cognitive slowing, and GI disturbances.
Other second-line options. Sufficient data support using the second-line drugs in our algorithm as first-line agents. However, the second-line agents pose a greater risk of adverse effects and decreased tolerability than SSRIs plus atypical antipsychotics. Second-line options include:
- SSRIs plus conventional antipsychotics
- amoxapine, a derivative of the conventional antipsychotic loxapine
- venlafaxine or TCAs plus atypical antipsychotics.
As with first-line therapy, 8 to 12 weeks is an adequate trial for second-line medications. ECT may be considered for patients who fail to respond to medications or experience complications.
SSRI/conventional antipsychotic. Our group used fluoxetine, 20 to 40 mg/d, plus perphenazine, 32 mg/d, in the first study of combined SSRI/conventional antipsychotic therapy for patients with psychotic depression.20 After 5 weeks, 22 of 30 patients’ HRSD and BPRS scores were reduced by >50%.
Amoxapine monotherapy. Anton and Burch21 compared amoxapine, 400 mg/d, with amitriptyline, 200 mg/d, plus perphenazine, 32 mg/d. Response rates (>50% reduction on the HRSD) were 71% and 81% for the two groups, respectively. Extrapyramidal symptoms (EPS) were more frequent with the combination therapy.
Venlafaxine’s mechanism of action is thought to be similar to that of TCAs, and we know from the Spiker study15 that TCAs are effective in treating psychotic depression. To our knowledge, venlafaxine dosages for psychotic depression have not been studied; 75 to 375 mg/d is recommended for nonpsychotic depression. Potential side effects include insomnia, nervousness, nausea, headache, dry mouth, fatigue, and elevations of supine diastolic blood pressure.
Third-line therapy. Clozapine may be considered when second-line options do not achieve adequate results.22 When making this choice, consider the need for biweekly blood monitoring and the risk of serious side effects such as agranulocytosis and seizures.
Maintenance therapy
Psychotic depression has a higher relapse rate than nonpsychotic depression. Relapse rates are 50 to 92% in patients with psychotic depression, and recurrence often develops within 2 to 14 months after recovery from the index episode.23 With little data on which to base a maintenance regimen, we recommend that you continue antipsychotics for 4 months after the acute episode resolves.
Recently our group reported that after a taper of perphenazine—following 4 months of treatment with fluoxetine and perphenazine—22 of 30 patients (73%) showed no signs of relapse over the next 11 months.24 We usually maintain patients on antidepressants indefinitely.
Related resources
- DeBattista C, Rothschild AJ, Schatzberg AF. A dynamic algorithm for the treatment of psychotic major depression. Psychiatric Ann 2002;32:681-91.
- Rothschild AJ. Challenges in the treatment of depression with psychotic features. Biol Psychiatry 2003;53:680-90.
- National Institutes of Health. http://www.clinicaltrials.gov. Enter “Medication treatment for psychotic depression” in “Search clinical trials” field, then click on appropriate link.
Drug brand names
- Amitriptyline • Elavil
- Amoxapine • Asendin
- Bupropion • Wellbutrin
- Chlorpromazine • Thorazine
- Clozapine • Clozaril
- Desipramine • Norpramin
- Fluoxetine • Prozac
- Imipramine • Tofranil
- Loxapine • Loxitane
- Olanzapine • Zyprexa
- Paroxetine • Paxil
- Perphenazine • Trilafon
- Risperidone • Risperdal
- Quetiapine • Seroquel
- Venlafaxine • Effexor
Disclosure
Dr. Bell reports no financial relationship with any company whose products are mentioned in this article or with manufacturers of competing products.
Dr. Rothschild receives research support from Bristol-Myers Squibb, Eli Lilly and Co., Merck & Co., Wyeth Pharmaceuticals, and the National Institute of Mental Health. He is a consultant to and/or speaker for Forest Pharmaceuticals, Eli Lilly and Co., Abbott Laboratories, Bristol-Myers Squibb, and Pfizer Inc. In the past, he has been a consultant to and received research grants from Corcept Therapeutics.
1. Lykouras E, Malliaras D, Christodoulou GN, et al. Delusional depression: phenomenology and response to treatment, a prospective study. Acta Psychiatry Scand 1986;73:324-9.
2. Frances A, Brown RP, Kocsis JG, Mann JJ. Psychotic depression: a separate entity? Am J Psychiatry 1981;138:831-3.
3. Leckman JF, Weissman MM, Prusoff BA, et al. Subtypes of depression: family study perspective. Arch Gen Psychiatry 1984;41:833-9.
4. Coryell W, Leon A, Winokur G, et al. The importance of psychotic features to long-term course in depressive disorders. Am J Psychiatry 1996;153:483-9.
5. Roose SP, Glassman AH, Walsh BT, et al. Depression, delusions, and suicide. Am J Psychiatry 1983;140:1159-62.
6. Rothschild AJ, Samson JA, Bond TC, et al. Hypothalamic-pituitary-adrenal axis activity and one-year outcome in depression. Biol Psychiatry 1993;34:392-400.
7. Rothschild AJ, Schatzberg AF. Diagnosis and treatment of psychotic (delusional) depression. In: Grunhaus L, Greden JF (eds). Severe depressive disorder. Washington, DC: American Psychiatric Press, 1994;195-207.
8. Strober M, Carlson G. Bipolar illness in adolescents with major depression: Clinical, genetic, and psychopharmacologic predictors in a three to four year prospective follow-up investigation. Arch Gen Psychiatry 1982;39:549-55.
9. Akiskal HS, Walker P, Puzantian VR, et al. Bipolar outcome in the course of depressive illness: Phenomenological, familial, and pharmacologic predictors. J Affect Disord 1983;5:115-28.
10. Zimmerman M, Mattia JI. Psychotic subtyping of major depressive disorder and posttraumatic stress disorder. J Clin Psychiatry 1999;60(5):311-4.
11. Buhne A, Keuthen N. Prevalence of symptoms of body dysmorphic disorder and its correlates: a cross-cultural comparison. Psychosomatics 2002;43:486-90.
12. American Psychiatric Association Practice guidelines for the treatment of major depressive disorder (revision). Am J Psychiatry 200;157:(suppl 4).
13. Parker G, Roy K, Hadzi-Pavlovic D, Pedic F. Psychotic (delusional) depression: A meta-analysis of physical treatments. J Affect Disord 1992;24:17-24.
14. Thompson JW, Weiner RD, Myers CP. Use of ECT in the United States in 1975, 1980 and 1986. Am J Psychiatry 1994;151:1657-61.
15. Spiker DG, Weiss JC, Dealy RS, et al. The pharmacological treatment of delusional depression. Am J Psychiatry 1985;142:430-6.
16. Dube S, Rothschild A, Andersen SE, et al. Olanzapine-fluoxetine combination for psychotic depression (presentation). Barcelona, Spain: European College of Neuropsychopharmacology, 2002.
17. Hillert A, Maier W, Wetzel H, Benkert O. Risperidone in the treatment of disorders with a combined psychotic and depressive syndrome: a functional approach. Pharmacopsychiatry 1992;25:213-17.
18. Zarate CA, Jr, Rothschild AJ, Fletcher KE, et al. Clinical predictors of acute response with quetiapine in psychotic mood disorder. J Clin Psychiatry 2000;61:185-9.
19. Nelson JC, Mazure CM. Lithium augmentation in psychotic depression refractory to combined drug treatment. Am J Psychiatry 1986;143:363-6.
20. Rothschild AJ, Samson JA, Bessette MP, Carter-Campbell JT. Efficacy of combination fluoxetine and perphenazine in the treatment of psychotic depression. J Clin Psychiatry 1993;54:338-42.
21. Anton RF, Jr, Burch EA, Jr. Amoxapine versus amitriptyline combined with perphenazine in the treatment of psychotic depression. Am J Psychiatry 1990;147:1203-8.
22. Banov MD, Zarate CA, Jr, Tohen M, et al. Clozapine therapy in refractory affective disorder: polarity predicts response in long-term follow-up. J Clin Psychiatry 1994;55:295-300.
23. Aronson TA, Shukla S, Gujavarty K, et al. Relapse in delusional depression: a retrospective study of the course of treatment. Comp Psychiatry 1988;29:12-21.
24. Rothschild AJ, Duval SE. How long should patients with psychotic depression stay on the antipsychotic medication. J Clin Psychiatry 2003;64:390-6.
1. Lykouras E, Malliaras D, Christodoulou GN, et al. Delusional depression: phenomenology and response to treatment, a prospective study. Acta Psychiatry Scand 1986;73:324-9.
2. Frances A, Brown RP, Kocsis JG, Mann JJ. Psychotic depression: a separate entity? Am J Psychiatry 1981;138:831-3.
3. Leckman JF, Weissman MM, Prusoff BA, et al. Subtypes of depression: family study perspective. Arch Gen Psychiatry 1984;41:833-9.
4. Coryell W, Leon A, Winokur G, et al. The importance of psychotic features to long-term course in depressive disorders. Am J Psychiatry 1996;153:483-9.
5. Roose SP, Glassman AH, Walsh BT, et al. Depression, delusions, and suicide. Am J Psychiatry 1983;140:1159-62.
6. Rothschild AJ, Samson JA, Bond TC, et al. Hypothalamic-pituitary-adrenal axis activity and one-year outcome in depression. Biol Psychiatry 1993;34:392-400.
7. Rothschild AJ, Schatzberg AF. Diagnosis and treatment of psychotic (delusional) depression. In: Grunhaus L, Greden JF (eds). Severe depressive disorder. Washington, DC: American Psychiatric Press, 1994;195-207.
8. Strober M, Carlson G. Bipolar illness in adolescents with major depression: Clinical, genetic, and psychopharmacologic predictors in a three to four year prospective follow-up investigation. Arch Gen Psychiatry 1982;39:549-55.
9. Akiskal HS, Walker P, Puzantian VR, et al. Bipolar outcome in the course of depressive illness: Phenomenological, familial, and pharmacologic predictors. J Affect Disord 1983;5:115-28.
10. Zimmerman M, Mattia JI. Psychotic subtyping of major depressive disorder and posttraumatic stress disorder. J Clin Psychiatry 1999;60(5):311-4.
11. Buhne A, Keuthen N. Prevalence of symptoms of body dysmorphic disorder and its correlates: a cross-cultural comparison. Psychosomatics 2002;43:486-90.
12. American Psychiatric Association Practice guidelines for the treatment of major depressive disorder (revision). Am J Psychiatry 200;157:(suppl 4).
13. Parker G, Roy K, Hadzi-Pavlovic D, Pedic F. Psychotic (delusional) depression: A meta-analysis of physical treatments. J Affect Disord 1992;24:17-24.
14. Thompson JW, Weiner RD, Myers CP. Use of ECT in the United States in 1975, 1980 and 1986. Am J Psychiatry 1994;151:1657-61.
15. Spiker DG, Weiss JC, Dealy RS, et al. The pharmacological treatment of delusional depression. Am J Psychiatry 1985;142:430-6.
16. Dube S, Rothschild A, Andersen SE, et al. Olanzapine-fluoxetine combination for psychotic depression (presentation). Barcelona, Spain: European College of Neuropsychopharmacology, 2002.
17. Hillert A, Maier W, Wetzel H, Benkert O. Risperidone in the treatment of disorders with a combined psychotic and depressive syndrome: a functional approach. Pharmacopsychiatry 1992;25:213-17.
18. Zarate CA, Jr, Rothschild AJ, Fletcher KE, et al. Clinical predictors of acute response with quetiapine in psychotic mood disorder. J Clin Psychiatry 2000;61:185-9.
19. Nelson JC, Mazure CM. Lithium augmentation in psychotic depression refractory to combined drug treatment. Am J Psychiatry 1986;143:363-6.
20. Rothschild AJ, Samson JA, Bessette MP, Carter-Campbell JT. Efficacy of combination fluoxetine and perphenazine in the treatment of psychotic depression. J Clin Psychiatry 1993;54:338-42.
21. Anton RF, Jr, Burch EA, Jr. Amoxapine versus amitriptyline combined with perphenazine in the treatment of psychotic depression. Am J Psychiatry 1990;147:1203-8.
22. Banov MD, Zarate CA, Jr, Tohen M, et al. Clozapine therapy in refractory affective disorder: polarity predicts response in long-term follow-up. J Clin Psychiatry 1994;55:295-300.
23. Aronson TA, Shukla S, Gujavarty K, et al. Relapse in delusional depression: a retrospective study of the course of treatment. Comp Psychiatry 1988;29:12-21.
24. Rothschild AJ, Duval SE. How long should patients with psychotic depression stay on the antipsychotic medication. J Clin Psychiatry 2003;64:390-6.
Instant messaging: The right call for your practice?
If you cannot get to a phone and you need to make immediate contact, instant messaging (IM) may be the right call.
IM, the equivalent of ‘instant e-mail,’ allows almost real-time communication and exchange of information over the Internet. IM is most popular among teenagers and college students, but medical providers and other professionals have begun to recognize its potential for speeding communication.
How IM started
Before the World Wide Web existed, electronic bulletin boards (BBS) were a popular mode of communication. Users would log in and browse various message topics, posting comments and questions for others to answer. Users also could check who was logged into the BBS and post messages to one or more of those contacts. This was done in a Telnet session, which is a method for connecting to another computer on the Internet. Telnet sessions hark back to mainframe computing, when computer terminals without processing power provided access to the central computer.
In 1996, Mirabilis released ICQ, which enabled Internet users to locate each other on the Internet without connecting to a BBS-by creating direct peer-to-peer communication channels. Companies such as America On Line, Microsoft, and Yahoo! also developed IM systems, but none could connect with one another because there is no common standard. AOL later purchased Mirabilis and incorporated its technology into the AOL Instant Messenger (AIM).
Doing IM: What you need
To do instant messaging, you need an Internet connection and the appropriate client. Keep in mind that your client should be:
- compatible with several systems
- or the same client that your messaging partner uses. New users typically choose an IM system that a friend or colleague is using.
Choosing an IM client is a matter of preference because all systems offer similar features (E-therapy: Alerting patients to the benefits, risks,“ November 2002).
Because IM is a direct communication channel with visual references to previous dialogue, it creates the environment of an active conversation. One drawback is that a participant’s typing speed typically limits the conversation’s pace. Using the voice feature overcomes this limitation, but this feature will not work properly without a broadband Internet connection, such as over a cable modem or digital subscriber line.
Risks of IM-based therapy
The IM communication channel’s lack of security poses a significant risk, particularly when used for psychotherapy. This active connection broadcasts its availability over the Internet, making it vulnerable to hackers. What’s more, the IM client may change your security settings for your Web browser1 or allow remote access to your computer by a hacker.2
Making your IM system secure
On the corporate level, vendors such as Yahoo!, Microsoft, AOL and others have created products to secure public IM systems, such as Akonix and Endeavors Technology. These products are gateways that maintain corporate data within the company network, encrypt information, specify and limit certain features, and log conversations. This level of security does not exist over major public IM networks for individual users.
Secure Shuttle Transport, DBabble, and PSST provide encrypted communication to individual users.
Table
Instant messaging clients
| Client | URL | Compatability | Operating systems |
|---|---|---|---|
| Yahoo! | http://messenger.yahoo.com/ | Yahoo only | Windows, Mac, Unix, SMS |
| AIM | http://www.aim.com/ | AOL, ICQ | Windows, Mac, Linux, Palm, Windows Mobile, SMS |
| MSN | http://messenger.msn.com/ | MSN only | Windows, Mac, Windows Mobile |
| ICQ | http://web.icq.com | ICQ, AOL | Windows, Mac, Windows Mobile, Palm, SMS |
| Trillian | http://www.ceruleanstudios.com/ | Yahoo, AOL, MSN, ICQ | Windows |
| Gaim | http://gaim.sourceforge.net/ | Yahoo, AOL, MSN, ICQ | Windows, Mac, Linux, BSD |
| Odigo | http://www.odigo.org/ | Yahoo, AOL, MSN, ICQ | Windows, Java |
| Easy Message | http://www.easymessage.net/ | Yahoo, AOL, MSN, ICQ | Windows |
| PalTalk | http://www.paltalk.com | PalTalk only | Windows |
If you have questions about these products or comments about Psyber Psychiatry, click here to contact Dr. Luo or e-mail him at: [email protected].
Disclosure
Dr. Luo reports no financial relationship with any company whose products are mentioned in this article. The opinions expressed by Dr. Luo in this column are his own and do not necessarily reflect those of Current Psychiatry.
(accessed Dec. 16, 2003)
1. Langa F. More instant messaging security holes. Information Week Oct. 1, 2001. Available at: http://www.informationweek.com/story/IWK20010927S0021
2. Saunders C. Yahoo! faces messenger flaw. Instant Messaging Planet.com Dec. 3, 2003. Available at: http://www.instantmessagingplanet.com/security/article.php/3116181
If you cannot get to a phone and you need to make immediate contact, instant messaging (IM) may be the right call.
IM, the equivalent of ‘instant e-mail,’ allows almost real-time communication and exchange of information over the Internet. IM is most popular among teenagers and college students, but medical providers and other professionals have begun to recognize its potential for speeding communication.
How IM started
Before the World Wide Web existed, electronic bulletin boards (BBS) were a popular mode of communication. Users would log in and browse various message topics, posting comments and questions for others to answer. Users also could check who was logged into the BBS and post messages to one or more of those contacts. This was done in a Telnet session, which is a method for connecting to another computer on the Internet. Telnet sessions hark back to mainframe computing, when computer terminals without processing power provided access to the central computer.
In 1996, Mirabilis released ICQ, which enabled Internet users to locate each other on the Internet without connecting to a BBS-by creating direct peer-to-peer communication channels. Companies such as America On Line, Microsoft, and Yahoo! also developed IM systems, but none could connect with one another because there is no common standard. AOL later purchased Mirabilis and incorporated its technology into the AOL Instant Messenger (AIM).
Doing IM: What you need
To do instant messaging, you need an Internet connection and the appropriate client. Keep in mind that your client should be:
- compatible with several systems
- or the same client that your messaging partner uses. New users typically choose an IM system that a friend or colleague is using.
Choosing an IM client is a matter of preference because all systems offer similar features (E-therapy: Alerting patients to the benefits, risks,“ November 2002).
Because IM is a direct communication channel with visual references to previous dialogue, it creates the environment of an active conversation. One drawback is that a participant’s typing speed typically limits the conversation’s pace. Using the voice feature overcomes this limitation, but this feature will not work properly without a broadband Internet connection, such as over a cable modem or digital subscriber line.
Risks of IM-based therapy
The IM communication channel’s lack of security poses a significant risk, particularly when used for psychotherapy. This active connection broadcasts its availability over the Internet, making it vulnerable to hackers. What’s more, the IM client may change your security settings for your Web browser1 or allow remote access to your computer by a hacker.2
Making your IM system secure
On the corporate level, vendors such as Yahoo!, Microsoft, AOL and others have created products to secure public IM systems, such as Akonix and Endeavors Technology. These products are gateways that maintain corporate data within the company network, encrypt information, specify and limit certain features, and log conversations. This level of security does not exist over major public IM networks for individual users.
Secure Shuttle Transport, DBabble, and PSST provide encrypted communication to individual users.
Table
Instant messaging clients
| Client | URL | Compatability | Operating systems |
|---|---|---|---|
| Yahoo! | http://messenger.yahoo.com/ | Yahoo only | Windows, Mac, Unix, SMS |
| AIM | http://www.aim.com/ | AOL, ICQ | Windows, Mac, Linux, Palm, Windows Mobile, SMS |
| MSN | http://messenger.msn.com/ | MSN only | Windows, Mac, Windows Mobile |
| ICQ | http://web.icq.com | ICQ, AOL | Windows, Mac, Windows Mobile, Palm, SMS |
| Trillian | http://www.ceruleanstudios.com/ | Yahoo, AOL, MSN, ICQ | Windows |
| Gaim | http://gaim.sourceforge.net/ | Yahoo, AOL, MSN, ICQ | Windows, Mac, Linux, BSD |
| Odigo | http://www.odigo.org/ | Yahoo, AOL, MSN, ICQ | Windows, Java |
| Easy Message | http://www.easymessage.net/ | Yahoo, AOL, MSN, ICQ | Windows |
| PalTalk | http://www.paltalk.com | PalTalk only | Windows |
If you have questions about these products or comments about Psyber Psychiatry, click here to contact Dr. Luo or e-mail him at: [email protected].
Disclosure
Dr. Luo reports no financial relationship with any company whose products are mentioned in this article. The opinions expressed by Dr. Luo in this column are his own and do not necessarily reflect those of Current Psychiatry.
If you cannot get to a phone and you need to make immediate contact, instant messaging (IM) may be the right call.
IM, the equivalent of ‘instant e-mail,’ allows almost real-time communication and exchange of information over the Internet. IM is most popular among teenagers and college students, but medical providers and other professionals have begun to recognize its potential for speeding communication.
How IM started
Before the World Wide Web existed, electronic bulletin boards (BBS) were a popular mode of communication. Users would log in and browse various message topics, posting comments and questions for others to answer. Users also could check who was logged into the BBS and post messages to one or more of those contacts. This was done in a Telnet session, which is a method for connecting to another computer on the Internet. Telnet sessions hark back to mainframe computing, when computer terminals without processing power provided access to the central computer.
In 1996, Mirabilis released ICQ, which enabled Internet users to locate each other on the Internet without connecting to a BBS-by creating direct peer-to-peer communication channels. Companies such as America On Line, Microsoft, and Yahoo! also developed IM systems, but none could connect with one another because there is no common standard. AOL later purchased Mirabilis and incorporated its technology into the AOL Instant Messenger (AIM).
Doing IM: What you need
To do instant messaging, you need an Internet connection and the appropriate client. Keep in mind that your client should be:
- compatible with several systems
- or the same client that your messaging partner uses. New users typically choose an IM system that a friend or colleague is using.
Choosing an IM client is a matter of preference because all systems offer similar features (E-therapy: Alerting patients to the benefits, risks,“ November 2002).
Because IM is a direct communication channel with visual references to previous dialogue, it creates the environment of an active conversation. One drawback is that a participant’s typing speed typically limits the conversation’s pace. Using the voice feature overcomes this limitation, but this feature will not work properly without a broadband Internet connection, such as over a cable modem or digital subscriber line.
Risks of IM-based therapy
The IM communication channel’s lack of security poses a significant risk, particularly when used for psychotherapy. This active connection broadcasts its availability over the Internet, making it vulnerable to hackers. What’s more, the IM client may change your security settings for your Web browser1 or allow remote access to your computer by a hacker.2
Making your IM system secure
On the corporate level, vendors such as Yahoo!, Microsoft, AOL and others have created products to secure public IM systems, such as Akonix and Endeavors Technology. These products are gateways that maintain corporate data within the company network, encrypt information, specify and limit certain features, and log conversations. This level of security does not exist over major public IM networks for individual users.
Secure Shuttle Transport, DBabble, and PSST provide encrypted communication to individual users.
Table
Instant messaging clients
| Client | URL | Compatability | Operating systems |
|---|---|---|---|
| Yahoo! | http://messenger.yahoo.com/ | Yahoo only | Windows, Mac, Unix, SMS |
| AIM | http://www.aim.com/ | AOL, ICQ | Windows, Mac, Linux, Palm, Windows Mobile, SMS |
| MSN | http://messenger.msn.com/ | MSN only | Windows, Mac, Windows Mobile |
| ICQ | http://web.icq.com | ICQ, AOL | Windows, Mac, Windows Mobile, Palm, SMS |
| Trillian | http://www.ceruleanstudios.com/ | Yahoo, AOL, MSN, ICQ | Windows |
| Gaim | http://gaim.sourceforge.net/ | Yahoo, AOL, MSN, ICQ | Windows, Mac, Linux, BSD |
| Odigo | http://www.odigo.org/ | Yahoo, AOL, MSN, ICQ | Windows, Java |
| Easy Message | http://www.easymessage.net/ | Yahoo, AOL, MSN, ICQ | Windows |
| PalTalk | http://www.paltalk.com | PalTalk only | Windows |
If you have questions about these products or comments about Psyber Psychiatry, click here to contact Dr. Luo or e-mail him at: [email protected].
Disclosure
Dr. Luo reports no financial relationship with any company whose products are mentioned in this article. The opinions expressed by Dr. Luo in this column are his own and do not necessarily reflect those of Current Psychiatry.
(accessed Dec. 16, 2003)
1. Langa F. More instant messaging security holes. Information Week Oct. 1, 2001. Available at: http://www.informationweek.com/story/IWK20010927S0021
2. Saunders C. Yahoo! faces messenger flaw. Instant Messaging Planet.com Dec. 3, 2003. Available at: http://www.instantmessagingplanet.com/security/article.php/3116181
(accessed Dec. 16, 2003)
1. Langa F. More instant messaging security holes. Information Week Oct. 1, 2001. Available at: http://www.informationweek.com/story/IWK20010927S0021
2. Saunders C. Yahoo! faces messenger flaw. Instant Messaging Planet.com Dec. 3, 2003. Available at: http://www.instantmessagingplanet.com/security/article.php/3116181
Use, abuse, or misuse? Knowing when to stop benzodiazepines
Many patients who request a benzodiazepine dose increase need more medication to manage their anxiety or panic disorder. Some patients, however, are misusing or abusing the drug.
How can you tell when to stop a benzodiazepine prescription? Consider these cases:
Case 1
Mr. A says a 1-week trial of diazepam, 5 mg tid, has alleviated his anxiety “a little” but adds that he has to “double-up” to get complete relief.
Is Mr. A being honest? Probably, because he is volunteering the information. Remember that diazepam, 10 mg every 4 hours, is within the agent’s therapeutic range for severe anxiety.
Recommendation: Increase diazepam to 10 mg tid and continue monitoring use. Expect that the patient may need additional doses during periods of greater stress.
Case 2
Three days after his last visit, Mr. B says he lost his clonazepam or it was stolen.
Is Mr. B being honest? Possibly not. Patients who abuse their prescriptions often make this claim.
Recommendation: Admonish Mr. B that clonazepam is a controlled substance. Tell him you will terminate the prescription if he cannot keep it secure.
Schedule a follow-up appointment. At that visit, order a toxicology screen and remind the patient that he must manage his prescription.
Case 3
Mr. C—who has been taking lorazepam, 5 mg tid to qid, for 1 year—mentions that he recently had a drink at a party. During his initial evaluation, he denied alcohol use. Upon further questioning, he admits that he drinks “socially” 4 to 6 times per year but never has more than a second drink on any occasion.
Is Mr. C being honest? Probably. Patients who drink minimally commonly deny alcohol use.
Recommendation: Explain the need to avoid alcohol when taking benzodiazepines but do not give the impression that an occasional drink will lead to serious harm or death.
Also find out how much and how often your patient drinks. Make sure “a drink now and then” is not in fact a few drinks 2 or 3 nights a week.
Case 4
Ms. D says her diazepam use has escalated to 40 to 60 mg/d, depending on her anxiety level. One day she calls to reschedule her appointment; her speech sounds slightly slurred.
Is Ms. D being honest? Probably, but she may be misusing diazepam or abusing another substance.
Recommendation: See the patient ASAP. Order urine and serum toxicology and a breath alcohol test. Consider inpatient detoxification. Also consider switching anxiolytics, as benzodiazepine abusers often crave one agent but not another.
Case 5
A new patient—Ms. E, age 70—says she has been taking diazepam, 5 mg qid, for 35 years.
Recommendation: Continue the prescription. Stopping diazepam for fear of long-term adverse effects could cause an upheaval for this patient.
Dr. Roth is attending psychiatrist, Department of Veterans Affairs Medical Center, North Chicago, IL.
Many patients who request a benzodiazepine dose increase need more medication to manage their anxiety or panic disorder. Some patients, however, are misusing or abusing the drug.
How can you tell when to stop a benzodiazepine prescription? Consider these cases:
Case 1
Mr. A says a 1-week trial of diazepam, 5 mg tid, has alleviated his anxiety “a little” but adds that he has to “double-up” to get complete relief.
Is Mr. A being honest? Probably, because he is volunteering the information. Remember that diazepam, 10 mg every 4 hours, is within the agent’s therapeutic range for severe anxiety.
Recommendation: Increase diazepam to 10 mg tid and continue monitoring use. Expect that the patient may need additional doses during periods of greater stress.
Case 2
Three days after his last visit, Mr. B says he lost his clonazepam or it was stolen.
Is Mr. B being honest? Possibly not. Patients who abuse their prescriptions often make this claim.
Recommendation: Admonish Mr. B that clonazepam is a controlled substance. Tell him you will terminate the prescription if he cannot keep it secure.
Schedule a follow-up appointment. At that visit, order a toxicology screen and remind the patient that he must manage his prescription.
Case 3
Mr. C—who has been taking lorazepam, 5 mg tid to qid, for 1 year—mentions that he recently had a drink at a party. During his initial evaluation, he denied alcohol use. Upon further questioning, he admits that he drinks “socially” 4 to 6 times per year but never has more than a second drink on any occasion.
Is Mr. C being honest? Probably. Patients who drink minimally commonly deny alcohol use.
Recommendation: Explain the need to avoid alcohol when taking benzodiazepines but do not give the impression that an occasional drink will lead to serious harm or death.
Also find out how much and how often your patient drinks. Make sure “a drink now and then” is not in fact a few drinks 2 or 3 nights a week.
Case 4
Ms. D says her diazepam use has escalated to 40 to 60 mg/d, depending on her anxiety level. One day she calls to reschedule her appointment; her speech sounds slightly slurred.
Is Ms. D being honest? Probably, but she may be misusing diazepam or abusing another substance.
Recommendation: See the patient ASAP. Order urine and serum toxicology and a breath alcohol test. Consider inpatient detoxification. Also consider switching anxiolytics, as benzodiazepine abusers often crave one agent but not another.
Case 5
A new patient—Ms. E, age 70—says she has been taking diazepam, 5 mg qid, for 35 years.
Recommendation: Continue the prescription. Stopping diazepam for fear of long-term adverse effects could cause an upheaval for this patient.
Many patients who request a benzodiazepine dose increase need more medication to manage their anxiety or panic disorder. Some patients, however, are misusing or abusing the drug.
How can you tell when to stop a benzodiazepine prescription? Consider these cases:
Case 1
Mr. A says a 1-week trial of diazepam, 5 mg tid, has alleviated his anxiety “a little” but adds that he has to “double-up” to get complete relief.
Is Mr. A being honest? Probably, because he is volunteering the information. Remember that diazepam, 10 mg every 4 hours, is within the agent’s therapeutic range for severe anxiety.
Recommendation: Increase diazepam to 10 mg tid and continue monitoring use. Expect that the patient may need additional doses during periods of greater stress.
Case 2
Three days after his last visit, Mr. B says he lost his clonazepam or it was stolen.
Is Mr. B being honest? Possibly not. Patients who abuse their prescriptions often make this claim.
Recommendation: Admonish Mr. B that clonazepam is a controlled substance. Tell him you will terminate the prescription if he cannot keep it secure.
Schedule a follow-up appointment. At that visit, order a toxicology screen and remind the patient that he must manage his prescription.
Case 3
Mr. C—who has been taking lorazepam, 5 mg tid to qid, for 1 year—mentions that he recently had a drink at a party. During his initial evaluation, he denied alcohol use. Upon further questioning, he admits that he drinks “socially” 4 to 6 times per year but never has more than a second drink on any occasion.
Is Mr. C being honest? Probably. Patients who drink minimally commonly deny alcohol use.
Recommendation: Explain the need to avoid alcohol when taking benzodiazepines but do not give the impression that an occasional drink will lead to serious harm or death.
Also find out how much and how often your patient drinks. Make sure “a drink now and then” is not in fact a few drinks 2 or 3 nights a week.
Case 4
Ms. D says her diazepam use has escalated to 40 to 60 mg/d, depending on her anxiety level. One day she calls to reschedule her appointment; her speech sounds slightly slurred.
Is Ms. D being honest? Probably, but she may be misusing diazepam or abusing another substance.
Recommendation: See the patient ASAP. Order urine and serum toxicology and a breath alcohol test. Consider inpatient detoxification. Also consider switching anxiolytics, as benzodiazepine abusers often crave one agent but not another.
Case 5
A new patient—Ms. E, age 70—says she has been taking diazepam, 5 mg qid, for 35 years.
Recommendation: Continue the prescription. Stopping diazepam for fear of long-term adverse effects could cause an upheaval for this patient.
Dr. Roth is attending psychiatrist, Department of Veterans Affairs Medical Center, North Chicago, IL.
Dr. Roth is attending psychiatrist, Department of Veterans Affairs Medical Center, North Chicago, IL.
Memantine: New option for advanced Alzheimer’s
As America’s population ages, the need to find new treatments for Alzheimer’s disease (AD) is increasingly urgent. Agents that have reached the medical mainstream in recent years target the disease in its mild to moderate stages. Memantine recently gained FDA approval for treating moderate to severe AD.
HOW IT WORKS
Memantine is an uncompetitive N-methyl-D-aspartate (NMDA) receptor antagonist. NMDA receptors mediate the effects of the excitatory amino acid glutamate, promote entry of calcium through ion channel pores, and are essential for normal learning and memory.1 Prolonged excessive glutamate stimulation, however, can lead to excitotoxicity and nerve cell death.
High-affinity NMDA receptor antagonists cause unacceptable side effects in humans and have not been well tolerated in clinical trials. By contrast, memantine—a moderate- to low-affinity NMDA receptor antagonist with rapid blocking/unblocking kinetics—has been well tolerated in clinical trials. The agent is readily displaced by presynaptic stimuli to allow normal channel function, but it reduces calcium influx from chronic low-amplitude glutamate stimulation.2
Table
Memantine: Fast facts
| Drug brand name: Namenda |
| Class: NMDA receptor antagonist |
| FDA-approved indication: Moderate to severe Alzheimer’s disease |
| Approval date: Oct. 17, 2003 |
| Manufacturer: Forest Pharmaceuticals |
| Dosing forms: 5 mg, 10 mg (“titration packets” containing 5-mg and 10-mg tablets are available) |
| Recommended dosage: Begin at 5 mg/d for 1 week; increase to 5 mg bid the second week, then to 10 mg in the morning and 5 mg in the evening for the third week; increase to 10 mg bid for continued dosing |
Memantine’s voltage-dependent characteristics allow it to block low-level tonic pathologic activation of NMDA receptors caused by low glutamate concentrations. This property also allows physiologic activation of receptors after synaptic release of larger glutamate concentrations that produce membrane depolarization.2 Memantine has demonstrated neuroprotection of neurons exposed to glutamate in a variety of in-vitro preparations.3
In experimental models, memantine has been shown to prolong long-term potentiation, a neurophysiologic correlate of learning and memory. Rats treated with memantine show enhanced learning recovery following entorhinal cortex lesions.3
Memantine has been shown to protect cholinergic cells in both acute and chronic animal models. It also prevents pathologic changes in the hippocampus produced by direct injection of betaamyloid protein.3 These findings suggest that memantine may improve learning and memory and may have neuroprotective properties in AD.
PHARMACOKINETICS
Memantine is absorbed completely from the GI tract and reaches maximum serum concentration in 6 to 8 hours. It is widely distributed and passes the blood-brain barrier with CSF concentrations approximately one-half those of serum levels. Dosages between 5 and 30 mg/d result in serum levels of 0.025 to 0.529 mmol. Relatively little biotransformation occurs.
The agent’s half-life ranges between 75 and 100 hours.4 Memantine is 10% to 45% protein bound, and 80% of circulating memantine is present as the parent compound. These kinetics justify once-daily dosing, although memantine usually is given bid.
Three metabolites have been identified, none of which exhibit NMDA receptor antagonist activity. Memantine minimally inhibits cytochrome P-450 enzymes, so interactions with drugs metabolized by these enzymes are unlikely.5
Memantine may potentiate the effects of barbiturates, neuroleptics, anticholinergics, L-dopa, ketamine, amantadine, dextromethorphan, and dopaminergic agonists. Concomitant use of memantine and amantadine should be avoided because the compounds are chemically related and both are NMDA antagonists. Memantine may hinder the effects of dantrolene or baclofen, so doses of these agents may need to be adjusted upward.
Memantine is eliminated almost completely via renal cation transport proteins. Drugs that use the same transport system—such as cimetidine, ranitidine, procainamide, quinine, and nicotine—may interact with memantine, possibly leading to increased plasma levels of these agents.
Hydrochlorothiazide activity is reduced by 20% when memantine is co-administered. Sodium bicarbonate, carbonic anhydrous inhibitors, and other drugs that alkalinize the urine may reduce memantine clearance and increase its serum levels.4
In healthy elderly volunteers with normal and reduced renal function, researchers observed a significant correlation between creatine clearance and total renal clearance of memantine, suggesting that patients with renal disease may require lower dosages.5
EFFICACY
In a preliminary, placebo-controlled study7 of patients with vascular- or Alzheimer’s-type dementia, memantine was associated with improved Clinical Global Impression of Change and Behavioral Rating Scale for Geriatric Patients scores. Mini-Mental State Examination (MMSE) scores for all patients entering the study were <10, indicating severe cognitive impairment. Global measures improved in 61 of 82 (73%) patients taking memantine, 10 mg/d, and in 38 of 84 (45%) patients taking placebo. Care dependence improved 3.1 points in the memantine group and 1.1 points in the placebo group.
Reisberg et al8 gave memantine, 20 mg/d, or placebo to 252 patients with AD across 28 weeks. The memantine group performed at significantly higher functional levels than the placebo group on the Alzheimer’s Disease Cooperative Study ADL Scale and the Severe Impairment Battery (SIB). The differences on the Clinical Interview-Based Impression of Change with caregiver input (CIBIC-plus) scale were nearly significant (p = 0.06). Patients entering the study had MMSE scores between 3 and 14. The magnitude of drug-placebo difference was modest (approximately 6 points on the SIB).
In a third pivotal trial, 403 patients with AD were randomly assigned to memantine, 20 mg/d, or placebo across 24 weeks. All patients were also taking the cholinesterase inhibitor donepezil, 10 mg/d.9 The memantine/donepezil group scored higher than the placebo/donepezil group on several scales. MMSE scores at entry ranged from 5 to 14. Drug-placebo differences were similar in magnitude to those observed in earlier studies.
TOLERABILITY
Controlled trials of memantine in patients with AD demonstrated few adverse effects.
Reisberg et al8 reported that 84% of memantine-group patients and 87% of the placebo group experienced adverse effects. More placebo-group than memantine-group patients (17% vs. 10%) discontinued the study because of adverse events. Agitation was the most commonly cited reason for discontinuation (7% of the placebo group and 5% of those taking memantine). No adverse event was significantly more common in the memantine group.
Tariot et al9 noted that confusion and headache were somewhat more common among those receiving memantine versus placebo. In other studies, symptoms possibly related to memantine included headache, akathisia, insomnia, increased motor activity, and excitement.6,10-12
CO-ADMINISTRATION WITH CHOLINESTERASE INHIBITORS
The range of AD severity targeted by memantine overlaps that addressed by the cholinesterase inhibitors donepezil, galantamine, and rivastigmine, which are indicated for mild to moderate AD. Many patients will receive both memantine and a cholinesterase inhibitor.
Data show this combination therapy to be clinically safe. Tariot et al9 found no increase in adverse events when memantine was co-administered with donepezil. Post-marketing surveillance studies in Germany indicate low rates of adverse events among patients receiving a cholinesterase inhibitor and memantine.13 In-vitro laboratory data indicate that memantine does not affect or interact with cholinesterase inhibition.14
Memantine is not metabolized by liver enzymes. No interaction with antidepressants or antipsychotics commonly used in AD is anticipated.
CLINICAL IMPLICATIONS
Memantine, with a mechanism of action different from that of existing agents, offers a new avenue of therapeutic intervention and expands the spectrum of patients who may benefit from FDA-approved drug therapy.
Research is needed to determine whether memantine is useful in earlier stages of AD and in treating mild cognitive impairment. The role of glutamate excitotoxicity in AD also needs to be defined.
Related resources
- Alzheimer’s Association. www.alz.org
- Mendez M, Cummings JL. Dementia: a clinical approach(3rd ed). Boston: Butterworth Heinemann, 2003.
Drug brand names
- Amantadine • Symmetrel
- Cimetidine • Tagamet
- Dantrolene • Dantrium
- Donepezil • Aricept
- Galantamine • Reminyl
- Memantine • Namenda
- Procainamide • Procanbid
- Procainamide • Exelon
Disclosure
The author has received research/grant support and/or is a consultant to AstraZeneca Pharmaceuticals, Bristol-Myers Squibb, Eisai Pharmaceuticals, Eli Lilly and Co., Forest Pharmaceuticals, GlaxoSmithKline, Janssen Pharmaceutica, Novartis Pharmaceuticals Corp., and Pfizer Inc.
:
1. Holt WF. Glutamate in health and disease: the role of inhibitors. In: Bar PR, Beal MF (eds). Neuroprotection in CNS diseases. New York: Marcel Dekker, 1997;87-119.
2. Parsons CG, Danysz W, Quack G. Memantine and the aminoalkyl-cyclohexane MRZ 2/579 are moderate affinity uncompetitive NMDA receptor antagonists—in vitro characterisation. Amino Acids 2000;19:157-66.
3. Parsons CG, Danysz W, Quack G. Memantine is a clinically well tolerated N-methyl-D-aspartate (NMDA) receptor antagonist—a review of preclinical data. Neuropharmacol 1999;38:735-67.
4. Merz Pharma. Scientific information: Akatinol memantine Frankfurt, Germany: Merz Pharma clinical research department, 1998:44.
5. Axura (memantine) product information. Available at: http://www.pharmaworld.com
6. Kilpatrick GJ, Tilbrook GS. Memantine. Merz. Curr Opin Investig Drugs 2002;3:798-806.
7. Winblad B, Poritis N. Memantine in severe dementia: results of the 9M-Best Study (Benefit and efficacy in severely demented patients during treatment with memantine). Int J Geriatr Psychiatry 1999;14:135-46.
8. Reisberg B, Doody R, Stoffler A, et al. Memantine in moderate-to-severe Alzheimer’s disease. N Engl J Med 2003;348:1333-41.
9. Tariot P, Farlow M, Grossberg G, et al. Memantine/donepezil dualtherapy is superior to placebo/donepezil therapy for treatment of moderate to severe Alzheimer’s disease. San Juan, Puerto Rico: American College of Neuropsychopharmacology annual meeting, 2002.
10. Ambrozi L, Danielczyk W. Treatment of impaired cerebral function in psychogeriatric patients with memantine—results of a phase II double-blind study. Pharmacopsychiatry 1988;21:144-6.
11. Gortelmeyer R, Erbler H. Memantine in the treatment of mild to moderate dementia syndrome. A double-blind placebo-controlled study. Arzneimittelforschung 1992;42:904-13.
12. Fleischhacker WW, Buchgeher A, Schubert H. Memantine in the treatment of senile dementia of the Alzheimer type. Prog Neuropsychopharmacol Biol Psychiatry 1986;10:87-93.
13. Hartmann S, Mobius HJ. Tolerability of memantine in combination with cholinesterase inhibitors in dementia therapy. Int Clin Psychopharmacol 2003;18:81-5.
14. Wenk GL, Quack G, Mobius HJ, Danysz W. No interaction of memantine with acetylcholinesterase inhibitors approved for clinical use. Life Sciences 2000;66:1079-83.
As America’s population ages, the need to find new treatments for Alzheimer’s disease (AD) is increasingly urgent. Agents that have reached the medical mainstream in recent years target the disease in its mild to moderate stages. Memantine recently gained FDA approval for treating moderate to severe AD.
HOW IT WORKS
Memantine is an uncompetitive N-methyl-D-aspartate (NMDA) receptor antagonist. NMDA receptors mediate the effects of the excitatory amino acid glutamate, promote entry of calcium through ion channel pores, and are essential for normal learning and memory.1 Prolonged excessive glutamate stimulation, however, can lead to excitotoxicity and nerve cell death.
High-affinity NMDA receptor antagonists cause unacceptable side effects in humans and have not been well tolerated in clinical trials. By contrast, memantine—a moderate- to low-affinity NMDA receptor antagonist with rapid blocking/unblocking kinetics—has been well tolerated in clinical trials. The agent is readily displaced by presynaptic stimuli to allow normal channel function, but it reduces calcium influx from chronic low-amplitude glutamate stimulation.2
Table
Memantine: Fast facts
| Drug brand name: Namenda |
| Class: NMDA receptor antagonist |
| FDA-approved indication: Moderate to severe Alzheimer’s disease |
| Approval date: Oct. 17, 2003 |
| Manufacturer: Forest Pharmaceuticals |
| Dosing forms: 5 mg, 10 mg (“titration packets” containing 5-mg and 10-mg tablets are available) |
| Recommended dosage: Begin at 5 mg/d for 1 week; increase to 5 mg bid the second week, then to 10 mg in the morning and 5 mg in the evening for the third week; increase to 10 mg bid for continued dosing |
Memantine’s voltage-dependent characteristics allow it to block low-level tonic pathologic activation of NMDA receptors caused by low glutamate concentrations. This property also allows physiologic activation of receptors after synaptic release of larger glutamate concentrations that produce membrane depolarization.2 Memantine has demonstrated neuroprotection of neurons exposed to glutamate in a variety of in-vitro preparations.3
In experimental models, memantine has been shown to prolong long-term potentiation, a neurophysiologic correlate of learning and memory. Rats treated with memantine show enhanced learning recovery following entorhinal cortex lesions.3
Memantine has been shown to protect cholinergic cells in both acute and chronic animal models. It also prevents pathologic changes in the hippocampus produced by direct injection of betaamyloid protein.3 These findings suggest that memantine may improve learning and memory and may have neuroprotective properties in AD.
PHARMACOKINETICS
Memantine is absorbed completely from the GI tract and reaches maximum serum concentration in 6 to 8 hours. It is widely distributed and passes the blood-brain barrier with CSF concentrations approximately one-half those of serum levels. Dosages between 5 and 30 mg/d result in serum levels of 0.025 to 0.529 mmol. Relatively little biotransformation occurs.
The agent’s half-life ranges between 75 and 100 hours.4 Memantine is 10% to 45% protein bound, and 80% of circulating memantine is present as the parent compound. These kinetics justify once-daily dosing, although memantine usually is given bid.
Three metabolites have been identified, none of which exhibit NMDA receptor antagonist activity. Memantine minimally inhibits cytochrome P-450 enzymes, so interactions with drugs metabolized by these enzymes are unlikely.5
Memantine may potentiate the effects of barbiturates, neuroleptics, anticholinergics, L-dopa, ketamine, amantadine, dextromethorphan, and dopaminergic agonists. Concomitant use of memantine and amantadine should be avoided because the compounds are chemically related and both are NMDA antagonists. Memantine may hinder the effects of dantrolene or baclofen, so doses of these agents may need to be adjusted upward.
Memantine is eliminated almost completely via renal cation transport proteins. Drugs that use the same transport system—such as cimetidine, ranitidine, procainamide, quinine, and nicotine—may interact with memantine, possibly leading to increased plasma levels of these agents.
Hydrochlorothiazide activity is reduced by 20% when memantine is co-administered. Sodium bicarbonate, carbonic anhydrous inhibitors, and other drugs that alkalinize the urine may reduce memantine clearance and increase its serum levels.4
In healthy elderly volunteers with normal and reduced renal function, researchers observed a significant correlation between creatine clearance and total renal clearance of memantine, suggesting that patients with renal disease may require lower dosages.5
EFFICACY
In a preliminary, placebo-controlled study7 of patients with vascular- or Alzheimer’s-type dementia, memantine was associated with improved Clinical Global Impression of Change and Behavioral Rating Scale for Geriatric Patients scores. Mini-Mental State Examination (MMSE) scores for all patients entering the study were <10, indicating severe cognitive impairment. Global measures improved in 61 of 82 (73%) patients taking memantine, 10 mg/d, and in 38 of 84 (45%) patients taking placebo. Care dependence improved 3.1 points in the memantine group and 1.1 points in the placebo group.
Reisberg et al8 gave memantine, 20 mg/d, or placebo to 252 patients with AD across 28 weeks. The memantine group performed at significantly higher functional levels than the placebo group on the Alzheimer’s Disease Cooperative Study ADL Scale and the Severe Impairment Battery (SIB). The differences on the Clinical Interview-Based Impression of Change with caregiver input (CIBIC-plus) scale were nearly significant (p = 0.06). Patients entering the study had MMSE scores between 3 and 14. The magnitude of drug-placebo difference was modest (approximately 6 points on the SIB).
In a third pivotal trial, 403 patients with AD were randomly assigned to memantine, 20 mg/d, or placebo across 24 weeks. All patients were also taking the cholinesterase inhibitor donepezil, 10 mg/d.9 The memantine/donepezil group scored higher than the placebo/donepezil group on several scales. MMSE scores at entry ranged from 5 to 14. Drug-placebo differences were similar in magnitude to those observed in earlier studies.
TOLERABILITY
Controlled trials of memantine in patients with AD demonstrated few adverse effects.
Reisberg et al8 reported that 84% of memantine-group patients and 87% of the placebo group experienced adverse effects. More placebo-group than memantine-group patients (17% vs. 10%) discontinued the study because of adverse events. Agitation was the most commonly cited reason for discontinuation (7% of the placebo group and 5% of those taking memantine). No adverse event was significantly more common in the memantine group.
Tariot et al9 noted that confusion and headache were somewhat more common among those receiving memantine versus placebo. In other studies, symptoms possibly related to memantine included headache, akathisia, insomnia, increased motor activity, and excitement.6,10-12
CO-ADMINISTRATION WITH CHOLINESTERASE INHIBITORS
The range of AD severity targeted by memantine overlaps that addressed by the cholinesterase inhibitors donepezil, galantamine, and rivastigmine, which are indicated for mild to moderate AD. Many patients will receive both memantine and a cholinesterase inhibitor.
Data show this combination therapy to be clinically safe. Tariot et al9 found no increase in adverse events when memantine was co-administered with donepezil. Post-marketing surveillance studies in Germany indicate low rates of adverse events among patients receiving a cholinesterase inhibitor and memantine.13 In-vitro laboratory data indicate that memantine does not affect or interact with cholinesterase inhibition.14
Memantine is not metabolized by liver enzymes. No interaction with antidepressants or antipsychotics commonly used in AD is anticipated.
CLINICAL IMPLICATIONS
Memantine, with a mechanism of action different from that of existing agents, offers a new avenue of therapeutic intervention and expands the spectrum of patients who may benefit from FDA-approved drug therapy.
Research is needed to determine whether memantine is useful in earlier stages of AD and in treating mild cognitive impairment. The role of glutamate excitotoxicity in AD also needs to be defined.
Related resources
- Alzheimer’s Association. www.alz.org
- Mendez M, Cummings JL. Dementia: a clinical approach(3rd ed). Boston: Butterworth Heinemann, 2003.
Drug brand names
- Amantadine • Symmetrel
- Cimetidine • Tagamet
- Dantrolene • Dantrium
- Donepezil • Aricept
- Galantamine • Reminyl
- Memantine • Namenda
- Procainamide • Procanbid
- Procainamide • Exelon
Disclosure
The author has received research/grant support and/or is a consultant to AstraZeneca Pharmaceuticals, Bristol-Myers Squibb, Eisai Pharmaceuticals, Eli Lilly and Co., Forest Pharmaceuticals, GlaxoSmithKline, Janssen Pharmaceutica, Novartis Pharmaceuticals Corp., and Pfizer Inc.
As America’s population ages, the need to find new treatments for Alzheimer’s disease (AD) is increasingly urgent. Agents that have reached the medical mainstream in recent years target the disease in its mild to moderate stages. Memantine recently gained FDA approval for treating moderate to severe AD.
HOW IT WORKS
Memantine is an uncompetitive N-methyl-D-aspartate (NMDA) receptor antagonist. NMDA receptors mediate the effects of the excitatory amino acid glutamate, promote entry of calcium through ion channel pores, and are essential for normal learning and memory.1 Prolonged excessive glutamate stimulation, however, can lead to excitotoxicity and nerve cell death.
High-affinity NMDA receptor antagonists cause unacceptable side effects in humans and have not been well tolerated in clinical trials. By contrast, memantine—a moderate- to low-affinity NMDA receptor antagonist with rapid blocking/unblocking kinetics—has been well tolerated in clinical trials. The agent is readily displaced by presynaptic stimuli to allow normal channel function, but it reduces calcium influx from chronic low-amplitude glutamate stimulation.2
Table
Memantine: Fast facts
| Drug brand name: Namenda |
| Class: NMDA receptor antagonist |
| FDA-approved indication: Moderate to severe Alzheimer’s disease |
| Approval date: Oct. 17, 2003 |
| Manufacturer: Forest Pharmaceuticals |
| Dosing forms: 5 mg, 10 mg (“titration packets” containing 5-mg and 10-mg tablets are available) |
| Recommended dosage: Begin at 5 mg/d for 1 week; increase to 5 mg bid the second week, then to 10 mg in the morning and 5 mg in the evening for the third week; increase to 10 mg bid for continued dosing |
Memantine’s voltage-dependent characteristics allow it to block low-level tonic pathologic activation of NMDA receptors caused by low glutamate concentrations. This property also allows physiologic activation of receptors after synaptic release of larger glutamate concentrations that produce membrane depolarization.2 Memantine has demonstrated neuroprotection of neurons exposed to glutamate in a variety of in-vitro preparations.3
In experimental models, memantine has been shown to prolong long-term potentiation, a neurophysiologic correlate of learning and memory. Rats treated with memantine show enhanced learning recovery following entorhinal cortex lesions.3
Memantine has been shown to protect cholinergic cells in both acute and chronic animal models. It also prevents pathologic changes in the hippocampus produced by direct injection of betaamyloid protein.3 These findings suggest that memantine may improve learning and memory and may have neuroprotective properties in AD.
PHARMACOKINETICS
Memantine is absorbed completely from the GI tract and reaches maximum serum concentration in 6 to 8 hours. It is widely distributed and passes the blood-brain barrier with CSF concentrations approximately one-half those of serum levels. Dosages between 5 and 30 mg/d result in serum levels of 0.025 to 0.529 mmol. Relatively little biotransformation occurs.
The agent’s half-life ranges between 75 and 100 hours.4 Memantine is 10% to 45% protein bound, and 80% of circulating memantine is present as the parent compound. These kinetics justify once-daily dosing, although memantine usually is given bid.
Three metabolites have been identified, none of which exhibit NMDA receptor antagonist activity. Memantine minimally inhibits cytochrome P-450 enzymes, so interactions with drugs metabolized by these enzymes are unlikely.5
Memantine may potentiate the effects of barbiturates, neuroleptics, anticholinergics, L-dopa, ketamine, amantadine, dextromethorphan, and dopaminergic agonists. Concomitant use of memantine and amantadine should be avoided because the compounds are chemically related and both are NMDA antagonists. Memantine may hinder the effects of dantrolene or baclofen, so doses of these agents may need to be adjusted upward.
Memantine is eliminated almost completely via renal cation transport proteins. Drugs that use the same transport system—such as cimetidine, ranitidine, procainamide, quinine, and nicotine—may interact with memantine, possibly leading to increased plasma levels of these agents.
Hydrochlorothiazide activity is reduced by 20% when memantine is co-administered. Sodium bicarbonate, carbonic anhydrous inhibitors, and other drugs that alkalinize the urine may reduce memantine clearance and increase its serum levels.4
In healthy elderly volunteers with normal and reduced renal function, researchers observed a significant correlation between creatine clearance and total renal clearance of memantine, suggesting that patients with renal disease may require lower dosages.5
EFFICACY
In a preliminary, placebo-controlled study7 of patients with vascular- or Alzheimer’s-type dementia, memantine was associated with improved Clinical Global Impression of Change and Behavioral Rating Scale for Geriatric Patients scores. Mini-Mental State Examination (MMSE) scores for all patients entering the study were <10, indicating severe cognitive impairment. Global measures improved in 61 of 82 (73%) patients taking memantine, 10 mg/d, and in 38 of 84 (45%) patients taking placebo. Care dependence improved 3.1 points in the memantine group and 1.1 points in the placebo group.
Reisberg et al8 gave memantine, 20 mg/d, or placebo to 252 patients with AD across 28 weeks. The memantine group performed at significantly higher functional levels than the placebo group on the Alzheimer’s Disease Cooperative Study ADL Scale and the Severe Impairment Battery (SIB). The differences on the Clinical Interview-Based Impression of Change with caregiver input (CIBIC-plus) scale were nearly significant (p = 0.06). Patients entering the study had MMSE scores between 3 and 14. The magnitude of drug-placebo difference was modest (approximately 6 points on the SIB).
In a third pivotal trial, 403 patients with AD were randomly assigned to memantine, 20 mg/d, or placebo across 24 weeks. All patients were also taking the cholinesterase inhibitor donepezil, 10 mg/d.9 The memantine/donepezil group scored higher than the placebo/donepezil group on several scales. MMSE scores at entry ranged from 5 to 14. Drug-placebo differences were similar in magnitude to those observed in earlier studies.
TOLERABILITY
Controlled trials of memantine in patients with AD demonstrated few adverse effects.
Reisberg et al8 reported that 84% of memantine-group patients and 87% of the placebo group experienced adverse effects. More placebo-group than memantine-group patients (17% vs. 10%) discontinued the study because of adverse events. Agitation was the most commonly cited reason for discontinuation (7% of the placebo group and 5% of those taking memantine). No adverse event was significantly more common in the memantine group.
Tariot et al9 noted that confusion and headache were somewhat more common among those receiving memantine versus placebo. In other studies, symptoms possibly related to memantine included headache, akathisia, insomnia, increased motor activity, and excitement.6,10-12
CO-ADMINISTRATION WITH CHOLINESTERASE INHIBITORS
The range of AD severity targeted by memantine overlaps that addressed by the cholinesterase inhibitors donepezil, galantamine, and rivastigmine, which are indicated for mild to moderate AD. Many patients will receive both memantine and a cholinesterase inhibitor.
Data show this combination therapy to be clinically safe. Tariot et al9 found no increase in adverse events when memantine was co-administered with donepezil. Post-marketing surveillance studies in Germany indicate low rates of adverse events among patients receiving a cholinesterase inhibitor and memantine.13 In-vitro laboratory data indicate that memantine does not affect or interact with cholinesterase inhibition.14
Memantine is not metabolized by liver enzymes. No interaction with antidepressants or antipsychotics commonly used in AD is anticipated.
CLINICAL IMPLICATIONS
Memantine, with a mechanism of action different from that of existing agents, offers a new avenue of therapeutic intervention and expands the spectrum of patients who may benefit from FDA-approved drug therapy.
Research is needed to determine whether memantine is useful in earlier stages of AD and in treating mild cognitive impairment. The role of glutamate excitotoxicity in AD also needs to be defined.
Related resources
- Alzheimer’s Association. www.alz.org
- Mendez M, Cummings JL. Dementia: a clinical approach(3rd ed). Boston: Butterworth Heinemann, 2003.
Drug brand names
- Amantadine • Symmetrel
- Cimetidine • Tagamet
- Dantrolene • Dantrium
- Donepezil • Aricept
- Galantamine • Reminyl
- Memantine • Namenda
- Procainamide • Procanbid
- Procainamide • Exelon
Disclosure
The author has received research/grant support and/or is a consultant to AstraZeneca Pharmaceuticals, Bristol-Myers Squibb, Eisai Pharmaceuticals, Eli Lilly and Co., Forest Pharmaceuticals, GlaxoSmithKline, Janssen Pharmaceutica, Novartis Pharmaceuticals Corp., and Pfizer Inc.
:
1. Holt WF. Glutamate in health and disease: the role of inhibitors. In: Bar PR, Beal MF (eds). Neuroprotection in CNS diseases. New York: Marcel Dekker, 1997;87-119.
2. Parsons CG, Danysz W, Quack G. Memantine and the aminoalkyl-cyclohexane MRZ 2/579 are moderate affinity uncompetitive NMDA receptor antagonists—in vitro characterisation. Amino Acids 2000;19:157-66.
3. Parsons CG, Danysz W, Quack G. Memantine is a clinically well tolerated N-methyl-D-aspartate (NMDA) receptor antagonist—a review of preclinical data. Neuropharmacol 1999;38:735-67.
4. Merz Pharma. Scientific information: Akatinol memantine Frankfurt, Germany: Merz Pharma clinical research department, 1998:44.
5. Axura (memantine) product information. Available at: http://www.pharmaworld.com
6. Kilpatrick GJ, Tilbrook GS. Memantine. Merz. Curr Opin Investig Drugs 2002;3:798-806.
7. Winblad B, Poritis N. Memantine in severe dementia: results of the 9M-Best Study (Benefit and efficacy in severely demented patients during treatment with memantine). Int J Geriatr Psychiatry 1999;14:135-46.
8. Reisberg B, Doody R, Stoffler A, et al. Memantine in moderate-to-severe Alzheimer’s disease. N Engl J Med 2003;348:1333-41.
9. Tariot P, Farlow M, Grossberg G, et al. Memantine/donepezil dualtherapy is superior to placebo/donepezil therapy for treatment of moderate to severe Alzheimer’s disease. San Juan, Puerto Rico: American College of Neuropsychopharmacology annual meeting, 2002.
10. Ambrozi L, Danielczyk W. Treatment of impaired cerebral function in psychogeriatric patients with memantine—results of a phase II double-blind study. Pharmacopsychiatry 1988;21:144-6.
11. Gortelmeyer R, Erbler H. Memantine in the treatment of mild to moderate dementia syndrome. A double-blind placebo-controlled study. Arzneimittelforschung 1992;42:904-13.
12. Fleischhacker WW, Buchgeher A, Schubert H. Memantine in the treatment of senile dementia of the Alzheimer type. Prog Neuropsychopharmacol Biol Psychiatry 1986;10:87-93.
13. Hartmann S, Mobius HJ. Tolerability of memantine in combination with cholinesterase inhibitors in dementia therapy. Int Clin Psychopharmacol 2003;18:81-5.
14. Wenk GL, Quack G, Mobius HJ, Danysz W. No interaction of memantine with acetylcholinesterase inhibitors approved for clinical use. Life Sciences 2000;66:1079-83.
:
1. Holt WF. Glutamate in health and disease: the role of inhibitors. In: Bar PR, Beal MF (eds). Neuroprotection in CNS diseases. New York: Marcel Dekker, 1997;87-119.
2. Parsons CG, Danysz W, Quack G. Memantine and the aminoalkyl-cyclohexane MRZ 2/579 are moderate affinity uncompetitive NMDA receptor antagonists—in vitro characterisation. Amino Acids 2000;19:157-66.
3. Parsons CG, Danysz W, Quack G. Memantine is a clinically well tolerated N-methyl-D-aspartate (NMDA) receptor antagonist—a review of preclinical data. Neuropharmacol 1999;38:735-67.
4. Merz Pharma. Scientific information: Akatinol memantine Frankfurt, Germany: Merz Pharma clinical research department, 1998:44.
5. Axura (memantine) product information. Available at: http://www.pharmaworld.com
6. Kilpatrick GJ, Tilbrook GS. Memantine. Merz. Curr Opin Investig Drugs 2002;3:798-806.
7. Winblad B, Poritis N. Memantine in severe dementia: results of the 9M-Best Study (Benefit and efficacy in severely demented patients during treatment with memantine). Int J Geriatr Psychiatry 1999;14:135-46.
8. Reisberg B, Doody R, Stoffler A, et al. Memantine in moderate-to-severe Alzheimer’s disease. N Engl J Med 2003;348:1333-41.
9. Tariot P, Farlow M, Grossberg G, et al. Memantine/donepezil dualtherapy is superior to placebo/donepezil therapy for treatment of moderate to severe Alzheimer’s disease. San Juan, Puerto Rico: American College of Neuropsychopharmacology annual meeting, 2002.
10. Ambrozi L, Danielczyk W. Treatment of impaired cerebral function in psychogeriatric patients with memantine—results of a phase II double-blind study. Pharmacopsychiatry 1988;21:144-6.
11. Gortelmeyer R, Erbler H. Memantine in the treatment of mild to moderate dementia syndrome. A double-blind placebo-controlled study. Arzneimittelforschung 1992;42:904-13.
12. Fleischhacker WW, Buchgeher A, Schubert H. Memantine in the treatment of senile dementia of the Alzheimer type. Prog Neuropsychopharmacol Biol Psychiatry 1986;10:87-93.
13. Hartmann S, Mobius HJ. Tolerability of memantine in combination with cholinesterase inhibitors in dementia therapy. Int Clin Psychopharmacol 2003;18:81-5.
14. Wenk GL, Quack G, Mobius HJ, Danysz W. No interaction of memantine with acetylcholinesterase inhibitors approved for clinical use. Life Sciences 2000;66:1079-83.
Omega-3 fatty acids: Do ‘fish oils’ have a therapeutic role in psychiatry?
Fourteen clinical trials in the past 3 years have examined the potential of omega-3 fatty acids in treating psychiatric disorders. Preliminary findings in at least 700 patients suggest that:
- omega-3 fatty acids used as adjuncts or monotherapy appear well-tolerated and safe in psychiatric disorders
- efficacy data vary by disorder
- the two marine omega-3 fatty acids may differ in efficacy.
Although we cannot offer specific guidance for using omega-3 fatty acids at this time, we can update you on recent trials of these “fish oils” in depression, bipolar disorder, schizophrenia, and other psychiatric disorders.
Treating depression
Prevalence rates of major depression1,2 and suicidal ideation3 decrease in populations as fish consumption increases. Some studies4,5 have shown omega-3 fatty acid deficiency in erythrocyte membranes and serum of depressed patients. This putative deficiency has been hypothesized to lead to:
- alterations in membrane fluidity, which affect monoamine (particularly serotonin) neurotransmission6,7
- an imbalance between omega-6 and omega-3 fatty acids, which affects the inflammatory response system (Box).5-12
- Nemets et al. 13 Twenty patients with recurrent major depression taking maintenance antidepressants were randomly assigned to adjunctive ethyl-EPA, 2 grams/d, or placebo for 4 weeks. Patients given ethyl-EPA showed significantly greater improvement than the placebo group in depressive symptoms, as measured by the Hamilton Rating Scale for Depression (HRSD).13
- Peet and Horrobin. 14 Seventy depressed patients taking antidepressants were randomly assigned to adjunctive ethyl-EPA (1, 2, or 4 grams/d) or placebo for 12 weeks. Only the group taking ethyl-EPA, 1 gram/d, showed significantly greater improvement than the placebo group.
- Su et al. 15 Twenty-eight patients taking antidepressants for major depression were randomly assigned to adjunctive omega-3 fatty acids (4.4 grams/d of EPA plus 2.2 grams/d of DHA) or placebo. After 8 weeks, depressive symptoms improved significantly more in the adjunctive treatment group.
- Marangell et al. 16 Thirty-six patients with mild to moderate depression (defined as a score of 17 on the 28-item HRSD) were randomly assigned to monotherapy with DHA, 2 grams/d, or placebo. Response rates after 6 weeks were comparable (27.8% with DHA versus 23.5% with placebo).
Polyunsaturated fatty acids contain a hydrocarbon chain with two or more double bonds. They are divided into families based on the location of their first double bond relative to the methyl end carbon—the “omega” carbon. Polyunsaturated fatty acids of interest in psychiatry include:
- omega-6 fatty acids—arachidonic acid (AA) and linoleic acid (LA)
- omega-3 fatty acids—eicosapentaenoic acid (EPA), docosahexaenoic acid (DHA), and alpha-linolenic acid (ALA).
Omega-3 and omega-6 fatty acids are called “essential” because they must be obtained from dietary sources. EPA and DHA are derived largely from wild—not farm-raised—fish, including sea bass, mackerel, pike, sardines, salmon, trout, herring, and cod liver oil.8 ALA, a precursor to both EPA and DHA, is derived from plant sources such as flaxseed oil, canola oil, walnuts, and soybean oil.
Polyunsaturated fatty acids, particularly AA and DHA, are important components of the phospholipid bilayer of neuronal cell membranes.They increase the ability of phospholipids to move “fluidly” within the membrane and modulate neurotransmission6,7 and signal transduction pathways9,10 thought to be important in psychiatric disorders. They also are precursors for eicosanoid molecules (such as prostaglandins and leukotrienes) and cytokines. Thus, an imbalance favoring omega-6 fatty acids over omega-3 fatty acids may lead to overproduction of pro-inflammatory cytokines.11
Omega-3 fatty acids are thought to be beneficial in numerous inflammatory and cardiovascular diseases. The American Heart Association’s dietary guidelines include dietary sources of omega-3 fatty acids as part of a healthy diet.12 Unfortunately, typical Western culture diets disproportionately favor foods rich in cholesterol and omega-6 fatty acids instead.
Table 1
Controlled trials of omega-3 fatty acids in treating major depression
| Author, year of publication | Duration and dosages | Number of patients | Results |
|---|---|---|---|
| Adjunctive therapy | |||
| Nemets et al, 2002 13 | 4 weeks, 2 grams/d of ethyl-EPA in recurrent depression | 20 | Significantly greater reduction in mean HRSD scores in ethyl-EPA group(-12.4) compared with placebo group (-1.6) 6 of 10 patients in ethyl-EPA group achieved 50% reduction in HRSD scores, compared with 1 in 10 patients in placebo group |
| Peet and Horrobin, 2002 14 | 12 weeks, 1, 2, or 4 grams/d of ethyl-EPA | 70 | Patients receiving 1 gram/d of ethyl-EPA showed significantly greater reduction in:
|
| Su et al, 2003 15 | 8 weeks, 4.4 grams/d of EPA and 2.2 grams/d of DHA | 28 | Treatment group showed significantly greater reduction in HRSD scores from baseline at weeks 4, 6, and 8 than placebo group |
| Monotherapy | |||
| Marangell et al, 2003 16 | 6 weeks, 2 grams/d of DHA | 36 | Little difference between response rates in DHA group (27.8%) and placebo group (23.5%) |
| BDI: Beck Depression Inventory | |||
| DHA: docosahexaenoic acid | |||
| EPA: eicosapentaenoic acid | |||
| HRSD: Hamilton Rating Scale for Depression | |||
| MADRS: Montgomery-Åsberg Depression Rating Scale | |||
Analysis. For patients with unipolar depression who were treated with omega-3 fatty acids:
- the most promisingresults have been seen with adjunctive EPA
- safety and tolerability have been good across studies.
No positive monotherapy studies have been published. Studies are needed to confirm EPA’s efficacy in unipolar depression and to determine the most effective dosage.
Treating bipolar disorder
EPA and DHA have been studied in bipolar disorder (Table 2) because their actions in modulating signal transduction pathways resemble those of lithium and valproate.10,17 Biochemical studies also have shown decreased AA and DHA in erythrocyte membranes of manic patients compared with controls.18
- Stoll et al. 19 Thirty patients receiving usual treatment for bipolar disorder were randomly assigned to adjunctive omega-3 fatty acids (6.2 grams/d of EPA plus 3.4 grams/d of DHA) or placebo for 4 months. Results were promising; patients receiving the omega-3 fatty acids remained in remission significantly longer than the placebo group.
- Keck et al. 20,21 On the other hand, two more-recent studies were disappointing. Both were 4-month, randomized, controlled trials in which patients received adjunctive EPA, 6 grams/d, or placebo. One study enrolled 59 patients with acute bipolar depression;20 the other enrolled 62 patients with rapid-cycling bipolar disorder.21 EPA was well-tolerated, but both studies found little difference in effectiveness between EPA and placebo.
Table 2
Controlled trials of adjunctive omega-3 fatty acids in treating bipolar disorder
| Author, year of publication | Duration and dosages | Number of patients | Results |
|---|---|---|---|
| Stoll et al, 1999 19 | 4 months, maintenance therapy (6.2 grams/d of EPA and 3.4 grams/d of DHA) in patients with bipolar I or II disorder | 30 | Significantly longer remission in omega-3 fatty acid group compared with placebo group |
| Keck et al, 2003 20 | 4 months, 6 grams/d of EPA in patients with acute bipolar depression | 59 | No significant difference in mean change from baseline to endpoint between EPA and placebo groups |
| Keck et al, 2003 21 | 4 months, 6 grams/d of EPA in patients with rapid-cycling bipolar disorder | 62 | Little difference in mean change from baseline to endpoint between EPA and placebo groups |
| DHA: docosahexaenoic acid | |||
| EPA: eicosapentaenoic acid | |||
Analysis. Further studies are needed to determine omega-3 fatty acids’ usefulness in treating bipolar illness.
Treating schizophrenia
Essential fatty acid deficiency and resulting lipid membrane abnormalities have been hypothesized to play a role in schizophrenia onset.22 Moreover, epidemiologic data suggest an association between high fish consumption and positive outcomes in patients with schizophrenia.23
Open-label trials, adjunctive therapy
- Mellor et al. 24 Twenty patients receiving antipsychotics for schizophrenia were treated for 6 weeks with 10 grams/d of a fish oil formulation containing 1.7 grams of EPA and 1.1 grams of DHA (Table 3). Psychotic symptoms improved significantly and were correlated with increased omega-3 fatty acid levels in erythrocyte membranes. Tardive dyskinesia also improved significantly, as measured by Abnormal Involuntary Movement Scale (AIMS) scores.
- Arvindakshan et al. 25 Thirty-three patients receiving antipsychotics for schizophrenia were given omega-3 fatty acids (360 mg/d of EPA and 240 mg/d of DHA) plus antioxidants (800 IU vitamin E and 1,000 IU vitamin C) for 4 months. Symptom and quality-of-life measures improved significantly, and clinical improvement was retained after 4 months of supplement washout.
Table 3
Clinical trials of omega-3 fatty acids in treating schizophrenia
| Authors, year of publication | Duration and dosages | Number of patients | Results |
|---|---|---|---|
| Open-label trials, adjunctive therapy | |||
| Mellor et al, 1995 24 | 6 weeks, 10 grams/d of fish oil (1.7 grams EPA and 1.1 grams DHA) | 20 | Significant improvement on PANSS and AIMS scores from baseline to endpoint |
| Arvindakshan et al, 2003 25 | 4 months, 360 mg/d of EPA and 240 mg/d of DHA, plus antioxidants (1,000 IU of vitamin C and 800 IU of vitamin E) | 33 | Significant improvements on total BPRS, PANSS, and Henrich’s Quality of Life Scale scores; improvements sustained after 4 months of supplementation washout |
| Controlled trials, adjunctive therapy | |||
| Peet et al, 2001 26 | 3 months, 2 grams/d of EPA or DHA | 45 | Greater improvement in total PANSS scores with EPA compared with DHA and placebo; EPA more effective than DHA in treating positive symptoms |
| Fenton et al, 2001 27 | 16 weeks, 3 grams/d of ethyl-EPA in patients with schizophrenia or schizoaffective disorder | 87 | No difference between ethyl-EPA and placebo groups in positive or negative symptoms, cognition, mood, or EPS |
| Peet et al, 2002 28 | 12 weeks, 1, 2, or 4 grams/d of ethyl-EPA with typical and atypical antipsychotics, including clozapine | 115 | Significantly greater improvement in mean total PANSS scores in clozapine-treated patients taking ethyl-EPA, 2 grams/d, compared with placebo; no difference between ethyl-EPAand placebo in patients taking typical or atypical antipsychotics |
| Emsley et al, 2002 29 | 12 weeks, 3 grams/d of ethyl-EPA | 40 | Significantly greater reduction in total PANSS and EPS Rating Scale dyskinesia scores in ethyl-EPA group compared with placebo |
| Controlled trial, monotherapy | |||
| Peet et al, 2001 26 | 3 months, 2 grams/d of EPA | 26 | EPA-treated patients had significantly lower PANSS scores at endpoint, compared with placebo; significantly more patients on placebo required antipsychotics (12 of 12) than did those on EPA (8 of 14) |
| AIMS: Abnormal Involuntary Movement Scale | |||
| BPRS: Brief Psychiatric Rating Scale | |||
| DHA: docosahexaenoic acid | |||
| EPA: eicosapentaenoic acid | |||
| EPS: extrapyramidal symptoms | |||
| PANSS: Positive and Negative Syndrome Scale | |||
Controlled trials, adjunctive therapy
- Peet et al. 26 In a 3-month study, 45 patients with schizophrenia were randomly assigned to adjunctive EPA or DHA (2 grams/d) or placebo. Those receiving EPA showed significantly greater improvement as measured by the Positive and Negative Syndrome Scale (PANSS), compared with DHA or placebo.
- Fenton et al. 27 In a 16-week study, 87 patients with schizophrenia or schizoaffective disorder were randomly assigned to adjunctive ethyl-EPA, 3 grams/d, or placebo. Little difference was noted in outcome measures of psychotic symptoms, mood, cognition, or extrapyramidal symptoms.
- Peet et al. 28 In a 12-week study. 115 patients with schizophrenia receiving typical antipsychotics, clozapine, or other atypical antipsychotics were randomly assigned to adjunctive ethyl-EPA (1, 2, or 4 grams/d) or placebo. Those taking clozapine improved significantly more with 2 grams/d of ethyl-EPA compared with patients receiving placebo. Little difference was noted between ethyl-EPA and placebo among patients taking typical or atypical antipsychotics.
- Emsley et al. 29 Forty patients with schizophrenia were randomly assigned to adjunctive ethyl-EPA, 3 grams/d, or placebo across 12 weeks. The ethyl-EPA group showed greater improvement in total PANSS scores and reduced dyskinesia, compared with placebo. Further analysis suggested, however, that the reduced dyskinesia scores at least partially accounted for the PANSS changes.
Controlled trial, monotherapy
- Peet et al. 26 Twenty-six patients with schizophrenia were randomly assigned to EPA, 2 grams/d, or placebo. After 3 months, those receiving EPA had significantly lower PANSS scores, and fewer (8 of 14) required antipsychotics than did those receiving placebo (12 of 12).
Analysis. Adjunctive ethyl-EPA (and perhaps combinations of EPA and DHA) may help patients with schizophrenia who are taking typical or atypical antipsychotics. EPA monotherapy also may be useful. Data are limited, however, and studies are needed before such use could be recommended.
Treating other disorders
Postpartum depression. The developing fetus and neonate require DHA from maternal stores for neurologic development. Maternal DHA depletion30 has been hypothesized to put mothers at risk for postpartum depression.31 An ecological study with data from 23 countries found that higher concentrations of DHA in maternal breast milk and greater seafood consumption predicted lower postpartum depression rates.32
In a randomized, controlled trial, giving DHA, 200 mg/d, to breastfeeding women during the first 4 months postpartum increased maternal plasma phospholipid content by 8%, compared with a 31% decrease in women given placebo.33
Data from randomized, controlled trials are needed to assess whether omega-3 fatty acid supplementation during pregnancy and the postpartum protects against postpartum depression.
Borderline personality disorder. In an 8-week controlled trial, Zanarini and Frankenburg34 randomly assigned 20 subjects with borderline personality disorder to monotherapy with ethyl-EPA, 1 gram/d, or placebo. Depressive symptoms improved and aggression decreased significantly in the ethyl-EPA group, suggesting the need for further research.
ADHD. Low DHA levels have been found in serum35 and erythrocytes36 of hyperactive children when compared with controls. Limited data in boys ages 6 to 12 also suggest an inverse relationship between plasma omega-3 fatty acids and behavior problems, as measured by the Connors’ Rating Scale.37
More research is needed into omega-3 fatty acids’ potential role in treating attention-deficit/hyperactivity disorder (ADHD), even though results of one controlled trial of adjunctive DHA in ADHD were disappointing.38
Dementia. Some large, prospective, epidemiologic studies39-41—but not others42—found an inverse relationship between dietary intake of omega-3 fatty acids and risk of cognitive decline or dementia.
Related resources
- USDA Nutrient Data Laboratory. http://www.nalusda.gov/fnic/foodcomp (accessed Dec. 1, 2003)
- Stoll AL. The omega-3 connection: the groundbreaking omega-3 antidepression diet and brain program. New York: Simon and Schuster, 2001.
Drug brand names
- Clozapine • Clozaril
Disclosure
The authors report no financial relationship with any company whose products are mentioned in this article or with manufacturers of competing products.
1. Hibbeln JR. Fish consumption and major depression. Lancet 1998;351(9110):1213.-
2. Tanskanen A, Hibbeln JR, Tuomilehto J, et al. Fish consumption and depressive symptoms in the general population in Finland. Psychiatr Serv 2001;52(4):529-31.
3. Tanskanen A, Hibelln JR, Hintikka J, et al. Fish consumption, depression, and suicidality in a general population. Arch Gen Psychiatry 2001;58(5):512-3.
4. Edwards R, Peet M, Shay J, Horrobin D. Omega-3 polyunsaturated fatty acid levels in the diet and in red blood cell membranes of depressed patients. J Affect Disord 1998;48(2-3):149-55.
5. Maes M, Christophe A, Delanghe J, et al. Lowered omega-3 polyunsaturated fatty acids in serum phospholipids and cholesterol esters of depressed patients. Psychiatry Res 1999;85(3):275-91.
6. Lundbaek JA, Andersen OS. Lysophospholipids modulate channel function by altering the mechanical properties of lipid bilayers. J Gen Physiol 1994;104(4):645-73.
7. Delion S, Chalon S, Guilloteau D, et al. Alpha-linolenic acid dietary deficiency alters age-related changes of dopaminergic and serotonergic neurotransmission in the rat frontal cortex. J Neurochem 1996;66(4):1582-91.
8. Passi S, Cataudella S, Di Marco P, et al. Fatty acid composition and antioxidant levels in muscle tissue of different Mediterranean marine species of fish and shellfish. J Agric Food Chem 2002;50(25):7314-22.
9. Hudson CJ, Young LT, Li PP, Warsh JJ. CNS signal transduction in the pathophysiology and pharmacology of affective disorders and schizophrenia. Synapse 1993;13(3):278-93.
10. Sperling RI, Benincaso AI, Knoell CT, et al. Dietary omega-3 polyunsaturated fatty acids inhibit phosphoinositide formation and chemotaxis in neutrophils. J Clin Invest 1993;91(2):651-60.
11. Endres S. Messengers and mediators: interactions among lipids, eicosanoids and cytokines. Am J Clin Nutr 1993;57(5 suppl):798S-800S.
12. Kris-Etherton PM, Harris WS, Appel LJ. Fish consumption, fish oil, omega-3 fatty acids, and cardiovascular disease. Arterioscler Thromb Vasc Biol 2003;23(2):e20-e30.
13. Nemets B, Stahl Z, Belmaker RH. Addition of omega-3 fatty acid to maintenance medication treatment for recurrent unipolar depressive disorder. Am J Psychiatry 2002;159(3):477-9.
14. Peet M, Horrobin DF. A dose-ranging study of the effects of ethyl-eicosapentaenoate in patients with ongoing depression despite apparently adequate treatment with standard drugs. Arch Gen Psychiatry 2002;59(10):913-9.
15. Su K-P, Huang S-Y, Chiu C-C, Shen WW. Omega-3 fatty acids in major depressive disorder: a preliminary double-blind, placebo-con-trolled trial. Eur Neuropsychopharmacol 2003;13(4):267-71.
16. Marangell LB, Martinez JM, Zboyan HA, et al. A double-blind, placebo-controlled study of the omega-3 fatty acid docosahexaenoic acid in the treatment of major depression. Am J Psychiatry 2003;160(5):996-8.
17. Stoll AL, Severus WE. Mood stabilizers: shared mechanisms of action at postsynaptic signal transduction and kindling processes. Harv Rev Psychiatry 1996;4(2):77-89.
18. Chiu CC, Huang SY, Su KP, et al. Polyunsaturated fatty acid deficit in patients with bipolar disorder. Eur Neuropsychopharmacol 2003;13(2):99-103.
19. Stoll AL, Severus WE, Freeman MP, et al. Omega-3 fatty acids in bipolar disorder: a preliminary double-blind, placebo-controlled trial. Arch Gen Psychiatry 1999;56(5):407-12.
20. Keck PE, Jr, McElroy SL, Freeman MP, et al. Randomized, placebo-controlled trial of eicosapentaenoic acid in bipolar depression. Bipolar Disord 2003;5(suppl 1):58.-
21. Keck PE, Jr, McElroy SL, Freeman MP, et al. Randomized, placebo-controlled trial of eicosapentaenoic acid in rapid cycling bipolar disorder. Bipolar Disord 2003;5(suppl 1):58.-
22. Horrobin DF. The membrane phospholipid hypothesis as a biochemical basis for the neurodevelopmental concept of schizophrenia. Schizophr Res 1998;30(3):193-208.
23. Christensen O, Christensen E. Fat consumption and schizophrenia. Acta Psychiatr Scand 1988;78(5):587-591
24. Mellor JE, Laugharne JD, Peet M. Schizophrenic symptoms and dietary intake of n-3 fatty acids. Schizophr Res 1995;18(1):85-6.
25. Arvindakshan M, Ghate M, Ranjekar PK, et al. Supplementation with a combination of omega-3 fatty acids and antioxidants (vitamins E and C) improves the outcome of schizophrenia. Schizophr Res 2003;62(3):195-204.
26. Peet M, Brind J, Ramchand CN, et al. Two double-blind placebo-controlled pilot studies of eicosapentaenoic acid in the treatment of schizophrenia. Schizophr Res 2001;49(3):243-51.
27. Fenton WS, Dickerson F, Boronow J, et al. A placebo-controlled trial of omega-3 fatty acid (ethyl eicosapentaenoic acid) supplementation for residual symptoms and cognitive impairment in schizophrenia. Am J Psychiatry 2001;158(12):2071-4.
28. Peet M, Horrobin DF. E-E Multicentre Study Group. A dose-ranging exploratory study of the effects of ethyl-eicosapentaenoate in patients with persistent schizophrenic symptoms. J Psychiatr Res 2002;36(1):7-18.
29. Emsley R, Myburgh C, Oosthuizen P, Van Rensburg SJ. Randomized, placebo-controlled study of ethyl-eicosapentaenoic acid as supplemental treatment in schizophrenia. Am J Psychiatry 2002;159(9):1596-8.
30. Al MD, van Houwelingen AC, Kester AD, et al. Maternal essential fatty acid patterns during normal pregnancy and their relationship to neonatal essential fatty acid status. Br J Nutr 1995;74(1):55-68.
31. Hibbeln JR, Salem N, Jr. Dietary polyunsaturated fatty acids and depression: when cholesterol does not satisfy. Am J Clin Nutr 1995;62(1):1-9.
32. Hibbeln JR. Seafood consumption, the DHA content of mothers’ milk and prevalence rates of postpartum depression: a cross-national, ecological analysis. J Affect Disord 2002;69(1-3):15-29.
33. Llorente AM, Jensen CL, Voigt RG, et al. Effect of maternal docosahexaenoic acid supplementation on postpartum depression and information processing. Am J Obstet Gynecol 2003;188(5):1348-53.
34. Zanarini MC, Frankenburg FR. Omega-3 fatty acid treatment of women with borderline personality disorder: a double-blind, placebo-controlled pilot study. Am J Psychiatry 2003;160(1):167-9.
35. Mitchell EA, Aman MG, Turbott SH, Manku M. Clinical characteristics and serum essential fatty acid levels in hyperactive children. Clin Pediatr 1987;26(8):406-11.
36. Stevens LJ, Zentall SS, Deck JL, et al. Essential fatty acid metabolism in boys with attention-deficit hyperactivity disorder. Am J Clin Nutr 1995;62(4):761-8.
37. Stevens LJ, Zentall SS, Abate ML, et al. Omega-3 fatty acids in boys with behavior, learning, and health problems. Physiol Behav 1996;59(4-5):915-20.
38. Voigt RG, Llorente AM, Jensen CL, et al. A randomized, double-blind, placebo-controlled trial of docosahexaenoic acid supplementation in children with attention-deficit/hyperactivity disorder. J Pediatr 2001;139(2):189-96.
39. Kalmijn S, Launer LJ, Ott A, et al. Dietary fat intake and the risk of incident dementia in the Rotterdam Study. Ann Neurol 1997;42(5):776-82.
40. Barberger-Gateau P, Letenneur L, Deschamps V, et al. Fish, meat, and risk of dementia: cohort study. BMJ 2002;325(7370):932-3.
41. Morris MC, Evans DA, Bienias JL, et al. Consumption of fish and n-3 fatty acids and risk of incident Alzheimer disease. Arch Neurol 2003;60(7):940-6.
42. Engelhart MJ, Geerlings MI, Ruitenberg A, et al. Diet and risk of dementia: does fat matter? The Rotterdam Study. Neurology 2002;59(12):1915-21.
Fourteen clinical trials in the past 3 years have examined the potential of omega-3 fatty acids in treating psychiatric disorders. Preliminary findings in at least 700 patients suggest that:
- omega-3 fatty acids used as adjuncts or monotherapy appear well-tolerated and safe in psychiatric disorders
- efficacy data vary by disorder
- the two marine omega-3 fatty acids may differ in efficacy.
Although we cannot offer specific guidance for using omega-3 fatty acids at this time, we can update you on recent trials of these “fish oils” in depression, bipolar disorder, schizophrenia, and other psychiatric disorders.
Treating depression
Prevalence rates of major depression1,2 and suicidal ideation3 decrease in populations as fish consumption increases. Some studies4,5 have shown omega-3 fatty acid deficiency in erythrocyte membranes and serum of depressed patients. This putative deficiency has been hypothesized to lead to:
- alterations in membrane fluidity, which affect monoamine (particularly serotonin) neurotransmission6,7
- an imbalance between omega-6 and omega-3 fatty acids, which affects the inflammatory response system (Box).5-12
- Nemets et al. 13 Twenty patients with recurrent major depression taking maintenance antidepressants were randomly assigned to adjunctive ethyl-EPA, 2 grams/d, or placebo for 4 weeks. Patients given ethyl-EPA showed significantly greater improvement than the placebo group in depressive symptoms, as measured by the Hamilton Rating Scale for Depression (HRSD).13
- Peet and Horrobin. 14 Seventy depressed patients taking antidepressants were randomly assigned to adjunctive ethyl-EPA (1, 2, or 4 grams/d) or placebo for 12 weeks. Only the group taking ethyl-EPA, 1 gram/d, showed significantly greater improvement than the placebo group.
- Su et al. 15 Twenty-eight patients taking antidepressants for major depression were randomly assigned to adjunctive omega-3 fatty acids (4.4 grams/d of EPA plus 2.2 grams/d of DHA) or placebo. After 8 weeks, depressive symptoms improved significantly more in the adjunctive treatment group.
- Marangell et al. 16 Thirty-six patients with mild to moderate depression (defined as a score of 17 on the 28-item HRSD) were randomly assigned to monotherapy with DHA, 2 grams/d, or placebo. Response rates after 6 weeks were comparable (27.8% with DHA versus 23.5% with placebo).
Polyunsaturated fatty acids contain a hydrocarbon chain with two or more double bonds. They are divided into families based on the location of their first double bond relative to the methyl end carbon—the “omega” carbon. Polyunsaturated fatty acids of interest in psychiatry include:
- omega-6 fatty acids—arachidonic acid (AA) and linoleic acid (LA)
- omega-3 fatty acids—eicosapentaenoic acid (EPA), docosahexaenoic acid (DHA), and alpha-linolenic acid (ALA).
Omega-3 and omega-6 fatty acids are called “essential” because they must be obtained from dietary sources. EPA and DHA are derived largely from wild—not farm-raised—fish, including sea bass, mackerel, pike, sardines, salmon, trout, herring, and cod liver oil.8 ALA, a precursor to both EPA and DHA, is derived from plant sources such as flaxseed oil, canola oil, walnuts, and soybean oil.
Polyunsaturated fatty acids, particularly AA and DHA, are important components of the phospholipid bilayer of neuronal cell membranes.They increase the ability of phospholipids to move “fluidly” within the membrane and modulate neurotransmission6,7 and signal transduction pathways9,10 thought to be important in psychiatric disorders. They also are precursors for eicosanoid molecules (such as prostaglandins and leukotrienes) and cytokines. Thus, an imbalance favoring omega-6 fatty acids over omega-3 fatty acids may lead to overproduction of pro-inflammatory cytokines.11
Omega-3 fatty acids are thought to be beneficial in numerous inflammatory and cardiovascular diseases. The American Heart Association’s dietary guidelines include dietary sources of omega-3 fatty acids as part of a healthy diet.12 Unfortunately, typical Western culture diets disproportionately favor foods rich in cholesterol and omega-6 fatty acids instead.
Table 1
Controlled trials of omega-3 fatty acids in treating major depression
| Author, year of publication | Duration and dosages | Number of patients | Results |
|---|---|---|---|
| Adjunctive therapy | |||
| Nemets et al, 2002 13 | 4 weeks, 2 grams/d of ethyl-EPA in recurrent depression | 20 | Significantly greater reduction in mean HRSD scores in ethyl-EPA group(-12.4) compared with placebo group (-1.6) 6 of 10 patients in ethyl-EPA group achieved 50% reduction in HRSD scores, compared with 1 in 10 patients in placebo group |
| Peet and Horrobin, 2002 14 | 12 weeks, 1, 2, or 4 grams/d of ethyl-EPA | 70 | Patients receiving 1 gram/d of ethyl-EPA showed significantly greater reduction in:
|
| Su et al, 2003 15 | 8 weeks, 4.4 grams/d of EPA and 2.2 grams/d of DHA | 28 | Treatment group showed significantly greater reduction in HRSD scores from baseline at weeks 4, 6, and 8 than placebo group |
| Monotherapy | |||
| Marangell et al, 2003 16 | 6 weeks, 2 grams/d of DHA | 36 | Little difference between response rates in DHA group (27.8%) and placebo group (23.5%) |
| BDI: Beck Depression Inventory | |||
| DHA: docosahexaenoic acid | |||
| EPA: eicosapentaenoic acid | |||
| HRSD: Hamilton Rating Scale for Depression | |||
| MADRS: Montgomery-Åsberg Depression Rating Scale | |||
Analysis. For patients with unipolar depression who were treated with omega-3 fatty acids:
- the most promisingresults have been seen with adjunctive EPA
- safety and tolerability have been good across studies.
No positive monotherapy studies have been published. Studies are needed to confirm EPA’s efficacy in unipolar depression and to determine the most effective dosage.
Treating bipolar disorder
EPA and DHA have been studied in bipolar disorder (Table 2) because their actions in modulating signal transduction pathways resemble those of lithium and valproate.10,17 Biochemical studies also have shown decreased AA and DHA in erythrocyte membranes of manic patients compared with controls.18
- Stoll et al. 19 Thirty patients receiving usual treatment for bipolar disorder were randomly assigned to adjunctive omega-3 fatty acids (6.2 grams/d of EPA plus 3.4 grams/d of DHA) or placebo for 4 months. Results were promising; patients receiving the omega-3 fatty acids remained in remission significantly longer than the placebo group.
- Keck et al. 20,21 On the other hand, two more-recent studies were disappointing. Both were 4-month, randomized, controlled trials in which patients received adjunctive EPA, 6 grams/d, or placebo. One study enrolled 59 patients with acute bipolar depression;20 the other enrolled 62 patients with rapid-cycling bipolar disorder.21 EPA was well-tolerated, but both studies found little difference in effectiveness between EPA and placebo.
Table 2
Controlled trials of adjunctive omega-3 fatty acids in treating bipolar disorder
| Author, year of publication | Duration and dosages | Number of patients | Results |
|---|---|---|---|
| Stoll et al, 1999 19 | 4 months, maintenance therapy (6.2 grams/d of EPA and 3.4 grams/d of DHA) in patients with bipolar I or II disorder | 30 | Significantly longer remission in omega-3 fatty acid group compared with placebo group |
| Keck et al, 2003 20 | 4 months, 6 grams/d of EPA in patients with acute bipolar depression | 59 | No significant difference in mean change from baseline to endpoint between EPA and placebo groups |
| Keck et al, 2003 21 | 4 months, 6 grams/d of EPA in patients with rapid-cycling bipolar disorder | 62 | Little difference in mean change from baseline to endpoint between EPA and placebo groups |
| DHA: docosahexaenoic acid | |||
| EPA: eicosapentaenoic acid | |||
Analysis. Further studies are needed to determine omega-3 fatty acids’ usefulness in treating bipolar illness.
Treating schizophrenia
Essential fatty acid deficiency and resulting lipid membrane abnormalities have been hypothesized to play a role in schizophrenia onset.22 Moreover, epidemiologic data suggest an association between high fish consumption and positive outcomes in patients with schizophrenia.23
Open-label trials, adjunctive therapy
- Mellor et al. 24 Twenty patients receiving antipsychotics for schizophrenia were treated for 6 weeks with 10 grams/d of a fish oil formulation containing 1.7 grams of EPA and 1.1 grams of DHA (Table 3). Psychotic symptoms improved significantly and were correlated with increased omega-3 fatty acid levels in erythrocyte membranes. Tardive dyskinesia also improved significantly, as measured by Abnormal Involuntary Movement Scale (AIMS) scores.
- Arvindakshan et al. 25 Thirty-three patients receiving antipsychotics for schizophrenia were given omega-3 fatty acids (360 mg/d of EPA and 240 mg/d of DHA) plus antioxidants (800 IU vitamin E and 1,000 IU vitamin C) for 4 months. Symptom and quality-of-life measures improved significantly, and clinical improvement was retained after 4 months of supplement washout.
Table 3
Clinical trials of omega-3 fatty acids in treating schizophrenia
| Authors, year of publication | Duration and dosages | Number of patients | Results |
|---|---|---|---|
| Open-label trials, adjunctive therapy | |||
| Mellor et al, 1995 24 | 6 weeks, 10 grams/d of fish oil (1.7 grams EPA and 1.1 grams DHA) | 20 | Significant improvement on PANSS and AIMS scores from baseline to endpoint |
| Arvindakshan et al, 2003 25 | 4 months, 360 mg/d of EPA and 240 mg/d of DHA, plus antioxidants (1,000 IU of vitamin C and 800 IU of vitamin E) | 33 | Significant improvements on total BPRS, PANSS, and Henrich’s Quality of Life Scale scores; improvements sustained after 4 months of supplementation washout |
| Controlled trials, adjunctive therapy | |||
| Peet et al, 2001 26 | 3 months, 2 grams/d of EPA or DHA | 45 | Greater improvement in total PANSS scores with EPA compared with DHA and placebo; EPA more effective than DHA in treating positive symptoms |
| Fenton et al, 2001 27 | 16 weeks, 3 grams/d of ethyl-EPA in patients with schizophrenia or schizoaffective disorder | 87 | No difference between ethyl-EPA and placebo groups in positive or negative symptoms, cognition, mood, or EPS |
| Peet et al, 2002 28 | 12 weeks, 1, 2, or 4 grams/d of ethyl-EPA with typical and atypical antipsychotics, including clozapine | 115 | Significantly greater improvement in mean total PANSS scores in clozapine-treated patients taking ethyl-EPA, 2 grams/d, compared with placebo; no difference between ethyl-EPAand placebo in patients taking typical or atypical antipsychotics |
| Emsley et al, 2002 29 | 12 weeks, 3 grams/d of ethyl-EPA | 40 | Significantly greater reduction in total PANSS and EPS Rating Scale dyskinesia scores in ethyl-EPA group compared with placebo |
| Controlled trial, monotherapy | |||
| Peet et al, 2001 26 | 3 months, 2 grams/d of EPA | 26 | EPA-treated patients had significantly lower PANSS scores at endpoint, compared with placebo; significantly more patients on placebo required antipsychotics (12 of 12) than did those on EPA (8 of 14) |
| AIMS: Abnormal Involuntary Movement Scale | |||
| BPRS: Brief Psychiatric Rating Scale | |||
| DHA: docosahexaenoic acid | |||
| EPA: eicosapentaenoic acid | |||
| EPS: extrapyramidal symptoms | |||
| PANSS: Positive and Negative Syndrome Scale | |||
Controlled trials, adjunctive therapy
- Peet et al. 26 In a 3-month study, 45 patients with schizophrenia were randomly assigned to adjunctive EPA or DHA (2 grams/d) or placebo. Those receiving EPA showed significantly greater improvement as measured by the Positive and Negative Syndrome Scale (PANSS), compared with DHA or placebo.
- Fenton et al. 27 In a 16-week study, 87 patients with schizophrenia or schizoaffective disorder were randomly assigned to adjunctive ethyl-EPA, 3 grams/d, or placebo. Little difference was noted in outcome measures of psychotic symptoms, mood, cognition, or extrapyramidal symptoms.
- Peet et al. 28 In a 12-week study. 115 patients with schizophrenia receiving typical antipsychotics, clozapine, or other atypical antipsychotics were randomly assigned to adjunctive ethyl-EPA (1, 2, or 4 grams/d) or placebo. Those taking clozapine improved significantly more with 2 grams/d of ethyl-EPA compared with patients receiving placebo. Little difference was noted between ethyl-EPA and placebo among patients taking typical or atypical antipsychotics.
- Emsley et al. 29 Forty patients with schizophrenia were randomly assigned to adjunctive ethyl-EPA, 3 grams/d, or placebo across 12 weeks. The ethyl-EPA group showed greater improvement in total PANSS scores and reduced dyskinesia, compared with placebo. Further analysis suggested, however, that the reduced dyskinesia scores at least partially accounted for the PANSS changes.
Controlled trial, monotherapy
- Peet et al. 26 Twenty-six patients with schizophrenia were randomly assigned to EPA, 2 grams/d, or placebo. After 3 months, those receiving EPA had significantly lower PANSS scores, and fewer (8 of 14) required antipsychotics than did those receiving placebo (12 of 12).
Analysis. Adjunctive ethyl-EPA (and perhaps combinations of EPA and DHA) may help patients with schizophrenia who are taking typical or atypical antipsychotics. EPA monotherapy also may be useful. Data are limited, however, and studies are needed before such use could be recommended.
Treating other disorders
Postpartum depression. The developing fetus and neonate require DHA from maternal stores for neurologic development. Maternal DHA depletion30 has been hypothesized to put mothers at risk for postpartum depression.31 An ecological study with data from 23 countries found that higher concentrations of DHA in maternal breast milk and greater seafood consumption predicted lower postpartum depression rates.32
In a randomized, controlled trial, giving DHA, 200 mg/d, to breastfeeding women during the first 4 months postpartum increased maternal plasma phospholipid content by 8%, compared with a 31% decrease in women given placebo.33
Data from randomized, controlled trials are needed to assess whether omega-3 fatty acid supplementation during pregnancy and the postpartum protects against postpartum depression.
Borderline personality disorder. In an 8-week controlled trial, Zanarini and Frankenburg34 randomly assigned 20 subjects with borderline personality disorder to monotherapy with ethyl-EPA, 1 gram/d, or placebo. Depressive symptoms improved and aggression decreased significantly in the ethyl-EPA group, suggesting the need for further research.
ADHD. Low DHA levels have been found in serum35 and erythrocytes36 of hyperactive children when compared with controls. Limited data in boys ages 6 to 12 also suggest an inverse relationship between plasma omega-3 fatty acids and behavior problems, as measured by the Connors’ Rating Scale.37
More research is needed into omega-3 fatty acids’ potential role in treating attention-deficit/hyperactivity disorder (ADHD), even though results of one controlled trial of adjunctive DHA in ADHD were disappointing.38
Dementia. Some large, prospective, epidemiologic studies39-41—but not others42—found an inverse relationship between dietary intake of omega-3 fatty acids and risk of cognitive decline or dementia.
Related resources
- USDA Nutrient Data Laboratory. http://www.nalusda.gov/fnic/foodcomp (accessed Dec. 1, 2003)
- Stoll AL. The omega-3 connection: the groundbreaking omega-3 antidepression diet and brain program. New York: Simon and Schuster, 2001.
Drug brand names
- Clozapine • Clozaril
Disclosure
The authors report no financial relationship with any company whose products are mentioned in this article or with manufacturers of competing products.
Fourteen clinical trials in the past 3 years have examined the potential of omega-3 fatty acids in treating psychiatric disorders. Preliminary findings in at least 700 patients suggest that:
- omega-3 fatty acids used as adjuncts or monotherapy appear well-tolerated and safe in psychiatric disorders
- efficacy data vary by disorder
- the two marine omega-3 fatty acids may differ in efficacy.
Although we cannot offer specific guidance for using omega-3 fatty acids at this time, we can update you on recent trials of these “fish oils” in depression, bipolar disorder, schizophrenia, and other psychiatric disorders.
Treating depression
Prevalence rates of major depression1,2 and suicidal ideation3 decrease in populations as fish consumption increases. Some studies4,5 have shown omega-3 fatty acid deficiency in erythrocyte membranes and serum of depressed patients. This putative deficiency has been hypothesized to lead to:
- alterations in membrane fluidity, which affect monoamine (particularly serotonin) neurotransmission6,7
- an imbalance between omega-6 and omega-3 fatty acids, which affects the inflammatory response system (Box).5-12
- Nemets et al. 13 Twenty patients with recurrent major depression taking maintenance antidepressants were randomly assigned to adjunctive ethyl-EPA, 2 grams/d, or placebo for 4 weeks. Patients given ethyl-EPA showed significantly greater improvement than the placebo group in depressive symptoms, as measured by the Hamilton Rating Scale for Depression (HRSD).13
- Peet and Horrobin. 14 Seventy depressed patients taking antidepressants were randomly assigned to adjunctive ethyl-EPA (1, 2, or 4 grams/d) or placebo for 12 weeks. Only the group taking ethyl-EPA, 1 gram/d, showed significantly greater improvement than the placebo group.
- Su et al. 15 Twenty-eight patients taking antidepressants for major depression were randomly assigned to adjunctive omega-3 fatty acids (4.4 grams/d of EPA plus 2.2 grams/d of DHA) or placebo. After 8 weeks, depressive symptoms improved significantly more in the adjunctive treatment group.
- Marangell et al. 16 Thirty-six patients with mild to moderate depression (defined as a score of 17 on the 28-item HRSD) were randomly assigned to monotherapy with DHA, 2 grams/d, or placebo. Response rates after 6 weeks were comparable (27.8% with DHA versus 23.5% with placebo).
Polyunsaturated fatty acids contain a hydrocarbon chain with two or more double bonds. They are divided into families based on the location of their first double bond relative to the methyl end carbon—the “omega” carbon. Polyunsaturated fatty acids of interest in psychiatry include:
- omega-6 fatty acids—arachidonic acid (AA) and linoleic acid (LA)
- omega-3 fatty acids—eicosapentaenoic acid (EPA), docosahexaenoic acid (DHA), and alpha-linolenic acid (ALA).
Omega-3 and omega-6 fatty acids are called “essential” because they must be obtained from dietary sources. EPA and DHA are derived largely from wild—not farm-raised—fish, including sea bass, mackerel, pike, sardines, salmon, trout, herring, and cod liver oil.8 ALA, a precursor to both EPA and DHA, is derived from plant sources such as flaxseed oil, canola oil, walnuts, and soybean oil.
Polyunsaturated fatty acids, particularly AA and DHA, are important components of the phospholipid bilayer of neuronal cell membranes.They increase the ability of phospholipids to move “fluidly” within the membrane and modulate neurotransmission6,7 and signal transduction pathways9,10 thought to be important in psychiatric disorders. They also are precursors for eicosanoid molecules (such as prostaglandins and leukotrienes) and cytokines. Thus, an imbalance favoring omega-6 fatty acids over omega-3 fatty acids may lead to overproduction of pro-inflammatory cytokines.11
Omega-3 fatty acids are thought to be beneficial in numerous inflammatory and cardiovascular diseases. The American Heart Association’s dietary guidelines include dietary sources of omega-3 fatty acids as part of a healthy diet.12 Unfortunately, typical Western culture diets disproportionately favor foods rich in cholesterol and omega-6 fatty acids instead.
Table 1
Controlled trials of omega-3 fatty acids in treating major depression
| Author, year of publication | Duration and dosages | Number of patients | Results |
|---|---|---|---|
| Adjunctive therapy | |||
| Nemets et al, 2002 13 | 4 weeks, 2 grams/d of ethyl-EPA in recurrent depression | 20 | Significantly greater reduction in mean HRSD scores in ethyl-EPA group(-12.4) compared with placebo group (-1.6) 6 of 10 patients in ethyl-EPA group achieved 50% reduction in HRSD scores, compared with 1 in 10 patients in placebo group |
| Peet and Horrobin, 2002 14 | 12 weeks, 1, 2, or 4 grams/d of ethyl-EPA | 70 | Patients receiving 1 gram/d of ethyl-EPA showed significantly greater reduction in:
|
| Su et al, 2003 15 | 8 weeks, 4.4 grams/d of EPA and 2.2 grams/d of DHA | 28 | Treatment group showed significantly greater reduction in HRSD scores from baseline at weeks 4, 6, and 8 than placebo group |
| Monotherapy | |||
| Marangell et al, 2003 16 | 6 weeks, 2 grams/d of DHA | 36 | Little difference between response rates in DHA group (27.8%) and placebo group (23.5%) |
| BDI: Beck Depression Inventory | |||
| DHA: docosahexaenoic acid | |||
| EPA: eicosapentaenoic acid | |||
| HRSD: Hamilton Rating Scale for Depression | |||
| MADRS: Montgomery-Åsberg Depression Rating Scale | |||
Analysis. For patients with unipolar depression who were treated with omega-3 fatty acids:
- the most promisingresults have been seen with adjunctive EPA
- safety and tolerability have been good across studies.
No positive monotherapy studies have been published. Studies are needed to confirm EPA’s efficacy in unipolar depression and to determine the most effective dosage.
Treating bipolar disorder
EPA and DHA have been studied in bipolar disorder (Table 2) because their actions in modulating signal transduction pathways resemble those of lithium and valproate.10,17 Biochemical studies also have shown decreased AA and DHA in erythrocyte membranes of manic patients compared with controls.18
- Stoll et al. 19 Thirty patients receiving usual treatment for bipolar disorder were randomly assigned to adjunctive omega-3 fatty acids (6.2 grams/d of EPA plus 3.4 grams/d of DHA) or placebo for 4 months. Results were promising; patients receiving the omega-3 fatty acids remained in remission significantly longer than the placebo group.
- Keck et al. 20,21 On the other hand, two more-recent studies were disappointing. Both were 4-month, randomized, controlled trials in which patients received adjunctive EPA, 6 grams/d, or placebo. One study enrolled 59 patients with acute bipolar depression;20 the other enrolled 62 patients with rapid-cycling bipolar disorder.21 EPA was well-tolerated, but both studies found little difference in effectiveness between EPA and placebo.
Table 2
Controlled trials of adjunctive omega-3 fatty acids in treating bipolar disorder
| Author, year of publication | Duration and dosages | Number of patients | Results |
|---|---|---|---|
| Stoll et al, 1999 19 | 4 months, maintenance therapy (6.2 grams/d of EPA and 3.4 grams/d of DHA) in patients with bipolar I or II disorder | 30 | Significantly longer remission in omega-3 fatty acid group compared with placebo group |
| Keck et al, 2003 20 | 4 months, 6 grams/d of EPA in patients with acute bipolar depression | 59 | No significant difference in mean change from baseline to endpoint between EPA and placebo groups |
| Keck et al, 2003 21 | 4 months, 6 grams/d of EPA in patients with rapid-cycling bipolar disorder | 62 | Little difference in mean change from baseline to endpoint between EPA and placebo groups |
| DHA: docosahexaenoic acid | |||
| EPA: eicosapentaenoic acid | |||
Analysis. Further studies are needed to determine omega-3 fatty acids’ usefulness in treating bipolar illness.
Treating schizophrenia
Essential fatty acid deficiency and resulting lipid membrane abnormalities have been hypothesized to play a role in schizophrenia onset.22 Moreover, epidemiologic data suggest an association between high fish consumption and positive outcomes in patients with schizophrenia.23
Open-label trials, adjunctive therapy
- Mellor et al. 24 Twenty patients receiving antipsychotics for schizophrenia were treated for 6 weeks with 10 grams/d of a fish oil formulation containing 1.7 grams of EPA and 1.1 grams of DHA (Table 3). Psychotic symptoms improved significantly and were correlated with increased omega-3 fatty acid levels in erythrocyte membranes. Tardive dyskinesia also improved significantly, as measured by Abnormal Involuntary Movement Scale (AIMS) scores.
- Arvindakshan et al. 25 Thirty-three patients receiving antipsychotics for schizophrenia were given omega-3 fatty acids (360 mg/d of EPA and 240 mg/d of DHA) plus antioxidants (800 IU vitamin E and 1,000 IU vitamin C) for 4 months. Symptom and quality-of-life measures improved significantly, and clinical improvement was retained after 4 months of supplement washout.
Table 3
Clinical trials of omega-3 fatty acids in treating schizophrenia
| Authors, year of publication | Duration and dosages | Number of patients | Results |
|---|---|---|---|
| Open-label trials, adjunctive therapy | |||
| Mellor et al, 1995 24 | 6 weeks, 10 grams/d of fish oil (1.7 grams EPA and 1.1 grams DHA) | 20 | Significant improvement on PANSS and AIMS scores from baseline to endpoint |
| Arvindakshan et al, 2003 25 | 4 months, 360 mg/d of EPA and 240 mg/d of DHA, plus antioxidants (1,000 IU of vitamin C and 800 IU of vitamin E) | 33 | Significant improvements on total BPRS, PANSS, and Henrich’s Quality of Life Scale scores; improvements sustained after 4 months of supplementation washout |
| Controlled trials, adjunctive therapy | |||
| Peet et al, 2001 26 | 3 months, 2 grams/d of EPA or DHA | 45 | Greater improvement in total PANSS scores with EPA compared with DHA and placebo; EPA more effective than DHA in treating positive symptoms |
| Fenton et al, 2001 27 | 16 weeks, 3 grams/d of ethyl-EPA in patients with schizophrenia or schizoaffective disorder | 87 | No difference between ethyl-EPA and placebo groups in positive or negative symptoms, cognition, mood, or EPS |
| Peet et al, 2002 28 | 12 weeks, 1, 2, or 4 grams/d of ethyl-EPA with typical and atypical antipsychotics, including clozapine | 115 | Significantly greater improvement in mean total PANSS scores in clozapine-treated patients taking ethyl-EPA, 2 grams/d, compared with placebo; no difference between ethyl-EPAand placebo in patients taking typical or atypical antipsychotics |
| Emsley et al, 2002 29 | 12 weeks, 3 grams/d of ethyl-EPA | 40 | Significantly greater reduction in total PANSS and EPS Rating Scale dyskinesia scores in ethyl-EPA group compared with placebo |
| Controlled trial, monotherapy | |||
| Peet et al, 2001 26 | 3 months, 2 grams/d of EPA | 26 | EPA-treated patients had significantly lower PANSS scores at endpoint, compared with placebo; significantly more patients on placebo required antipsychotics (12 of 12) than did those on EPA (8 of 14) |
| AIMS: Abnormal Involuntary Movement Scale | |||
| BPRS: Brief Psychiatric Rating Scale | |||
| DHA: docosahexaenoic acid | |||
| EPA: eicosapentaenoic acid | |||
| EPS: extrapyramidal symptoms | |||
| PANSS: Positive and Negative Syndrome Scale | |||
Controlled trials, adjunctive therapy
- Peet et al. 26 In a 3-month study, 45 patients with schizophrenia were randomly assigned to adjunctive EPA or DHA (2 grams/d) or placebo. Those receiving EPA showed significantly greater improvement as measured by the Positive and Negative Syndrome Scale (PANSS), compared with DHA or placebo.
- Fenton et al. 27 In a 16-week study, 87 patients with schizophrenia or schizoaffective disorder were randomly assigned to adjunctive ethyl-EPA, 3 grams/d, or placebo. Little difference was noted in outcome measures of psychotic symptoms, mood, cognition, or extrapyramidal symptoms.
- Peet et al. 28 In a 12-week study. 115 patients with schizophrenia receiving typical antipsychotics, clozapine, or other atypical antipsychotics were randomly assigned to adjunctive ethyl-EPA (1, 2, or 4 grams/d) or placebo. Those taking clozapine improved significantly more with 2 grams/d of ethyl-EPA compared with patients receiving placebo. Little difference was noted between ethyl-EPA and placebo among patients taking typical or atypical antipsychotics.
- Emsley et al. 29 Forty patients with schizophrenia were randomly assigned to adjunctive ethyl-EPA, 3 grams/d, or placebo across 12 weeks. The ethyl-EPA group showed greater improvement in total PANSS scores and reduced dyskinesia, compared with placebo. Further analysis suggested, however, that the reduced dyskinesia scores at least partially accounted for the PANSS changes.
Controlled trial, monotherapy
- Peet et al. 26 Twenty-six patients with schizophrenia were randomly assigned to EPA, 2 grams/d, or placebo. After 3 months, those receiving EPA had significantly lower PANSS scores, and fewer (8 of 14) required antipsychotics than did those receiving placebo (12 of 12).
Analysis. Adjunctive ethyl-EPA (and perhaps combinations of EPA and DHA) may help patients with schizophrenia who are taking typical or atypical antipsychotics. EPA monotherapy also may be useful. Data are limited, however, and studies are needed before such use could be recommended.
Treating other disorders
Postpartum depression. The developing fetus and neonate require DHA from maternal stores for neurologic development. Maternal DHA depletion30 has been hypothesized to put mothers at risk for postpartum depression.31 An ecological study with data from 23 countries found that higher concentrations of DHA in maternal breast milk and greater seafood consumption predicted lower postpartum depression rates.32
In a randomized, controlled trial, giving DHA, 200 mg/d, to breastfeeding women during the first 4 months postpartum increased maternal plasma phospholipid content by 8%, compared with a 31% decrease in women given placebo.33
Data from randomized, controlled trials are needed to assess whether omega-3 fatty acid supplementation during pregnancy and the postpartum protects against postpartum depression.
Borderline personality disorder. In an 8-week controlled trial, Zanarini and Frankenburg34 randomly assigned 20 subjects with borderline personality disorder to monotherapy with ethyl-EPA, 1 gram/d, or placebo. Depressive symptoms improved and aggression decreased significantly in the ethyl-EPA group, suggesting the need for further research.
ADHD. Low DHA levels have been found in serum35 and erythrocytes36 of hyperactive children when compared with controls. Limited data in boys ages 6 to 12 also suggest an inverse relationship between plasma omega-3 fatty acids and behavior problems, as measured by the Connors’ Rating Scale.37
More research is needed into omega-3 fatty acids’ potential role in treating attention-deficit/hyperactivity disorder (ADHD), even though results of one controlled trial of adjunctive DHA in ADHD were disappointing.38
Dementia. Some large, prospective, epidemiologic studies39-41—but not others42—found an inverse relationship between dietary intake of omega-3 fatty acids and risk of cognitive decline or dementia.
Related resources
- USDA Nutrient Data Laboratory. http://www.nalusda.gov/fnic/foodcomp (accessed Dec. 1, 2003)
- Stoll AL. The omega-3 connection: the groundbreaking omega-3 antidepression diet and brain program. New York: Simon and Schuster, 2001.
Drug brand names
- Clozapine • Clozaril
Disclosure
The authors report no financial relationship with any company whose products are mentioned in this article or with manufacturers of competing products.
1. Hibbeln JR. Fish consumption and major depression. Lancet 1998;351(9110):1213.-
2. Tanskanen A, Hibbeln JR, Tuomilehto J, et al. Fish consumption and depressive symptoms in the general population in Finland. Psychiatr Serv 2001;52(4):529-31.
3. Tanskanen A, Hibelln JR, Hintikka J, et al. Fish consumption, depression, and suicidality in a general population. Arch Gen Psychiatry 2001;58(5):512-3.
4. Edwards R, Peet M, Shay J, Horrobin D. Omega-3 polyunsaturated fatty acid levels in the diet and in red blood cell membranes of depressed patients. J Affect Disord 1998;48(2-3):149-55.
5. Maes M, Christophe A, Delanghe J, et al. Lowered omega-3 polyunsaturated fatty acids in serum phospholipids and cholesterol esters of depressed patients. Psychiatry Res 1999;85(3):275-91.
6. Lundbaek JA, Andersen OS. Lysophospholipids modulate channel function by altering the mechanical properties of lipid bilayers. J Gen Physiol 1994;104(4):645-73.
7. Delion S, Chalon S, Guilloteau D, et al. Alpha-linolenic acid dietary deficiency alters age-related changes of dopaminergic and serotonergic neurotransmission in the rat frontal cortex. J Neurochem 1996;66(4):1582-91.
8. Passi S, Cataudella S, Di Marco P, et al. Fatty acid composition and antioxidant levels in muscle tissue of different Mediterranean marine species of fish and shellfish. J Agric Food Chem 2002;50(25):7314-22.
9. Hudson CJ, Young LT, Li PP, Warsh JJ. CNS signal transduction in the pathophysiology and pharmacology of affective disorders and schizophrenia. Synapse 1993;13(3):278-93.
10. Sperling RI, Benincaso AI, Knoell CT, et al. Dietary omega-3 polyunsaturated fatty acids inhibit phosphoinositide formation and chemotaxis in neutrophils. J Clin Invest 1993;91(2):651-60.
11. Endres S. Messengers and mediators: interactions among lipids, eicosanoids and cytokines. Am J Clin Nutr 1993;57(5 suppl):798S-800S.
12. Kris-Etherton PM, Harris WS, Appel LJ. Fish consumption, fish oil, omega-3 fatty acids, and cardiovascular disease. Arterioscler Thromb Vasc Biol 2003;23(2):e20-e30.
13. Nemets B, Stahl Z, Belmaker RH. Addition of omega-3 fatty acid to maintenance medication treatment for recurrent unipolar depressive disorder. Am J Psychiatry 2002;159(3):477-9.
14. Peet M, Horrobin DF. A dose-ranging study of the effects of ethyl-eicosapentaenoate in patients with ongoing depression despite apparently adequate treatment with standard drugs. Arch Gen Psychiatry 2002;59(10):913-9.
15. Su K-P, Huang S-Y, Chiu C-C, Shen WW. Omega-3 fatty acids in major depressive disorder: a preliminary double-blind, placebo-con-trolled trial. Eur Neuropsychopharmacol 2003;13(4):267-71.
16. Marangell LB, Martinez JM, Zboyan HA, et al. A double-blind, placebo-controlled study of the omega-3 fatty acid docosahexaenoic acid in the treatment of major depression. Am J Psychiatry 2003;160(5):996-8.
17. Stoll AL, Severus WE. Mood stabilizers: shared mechanisms of action at postsynaptic signal transduction and kindling processes. Harv Rev Psychiatry 1996;4(2):77-89.
18. Chiu CC, Huang SY, Su KP, et al. Polyunsaturated fatty acid deficit in patients with bipolar disorder. Eur Neuropsychopharmacol 2003;13(2):99-103.
19. Stoll AL, Severus WE, Freeman MP, et al. Omega-3 fatty acids in bipolar disorder: a preliminary double-blind, placebo-controlled trial. Arch Gen Psychiatry 1999;56(5):407-12.
20. Keck PE, Jr, McElroy SL, Freeman MP, et al. Randomized, placebo-controlled trial of eicosapentaenoic acid in bipolar depression. Bipolar Disord 2003;5(suppl 1):58.-
21. Keck PE, Jr, McElroy SL, Freeman MP, et al. Randomized, placebo-controlled trial of eicosapentaenoic acid in rapid cycling bipolar disorder. Bipolar Disord 2003;5(suppl 1):58.-
22. Horrobin DF. The membrane phospholipid hypothesis as a biochemical basis for the neurodevelopmental concept of schizophrenia. Schizophr Res 1998;30(3):193-208.
23. Christensen O, Christensen E. Fat consumption and schizophrenia. Acta Psychiatr Scand 1988;78(5):587-591
24. Mellor JE, Laugharne JD, Peet M. Schizophrenic symptoms and dietary intake of n-3 fatty acids. Schizophr Res 1995;18(1):85-6.
25. Arvindakshan M, Ghate M, Ranjekar PK, et al. Supplementation with a combination of omega-3 fatty acids and antioxidants (vitamins E and C) improves the outcome of schizophrenia. Schizophr Res 2003;62(3):195-204.
26. Peet M, Brind J, Ramchand CN, et al. Two double-blind placebo-controlled pilot studies of eicosapentaenoic acid in the treatment of schizophrenia. Schizophr Res 2001;49(3):243-51.
27. Fenton WS, Dickerson F, Boronow J, et al. A placebo-controlled trial of omega-3 fatty acid (ethyl eicosapentaenoic acid) supplementation for residual symptoms and cognitive impairment in schizophrenia. Am J Psychiatry 2001;158(12):2071-4.
28. Peet M, Horrobin DF. E-E Multicentre Study Group. A dose-ranging exploratory study of the effects of ethyl-eicosapentaenoate in patients with persistent schizophrenic symptoms. J Psychiatr Res 2002;36(1):7-18.
29. Emsley R, Myburgh C, Oosthuizen P, Van Rensburg SJ. Randomized, placebo-controlled study of ethyl-eicosapentaenoic acid as supplemental treatment in schizophrenia. Am J Psychiatry 2002;159(9):1596-8.
30. Al MD, van Houwelingen AC, Kester AD, et al. Maternal essential fatty acid patterns during normal pregnancy and their relationship to neonatal essential fatty acid status. Br J Nutr 1995;74(1):55-68.
31. Hibbeln JR, Salem N, Jr. Dietary polyunsaturated fatty acids and depression: when cholesterol does not satisfy. Am J Clin Nutr 1995;62(1):1-9.
32. Hibbeln JR. Seafood consumption, the DHA content of mothers’ milk and prevalence rates of postpartum depression: a cross-national, ecological analysis. J Affect Disord 2002;69(1-3):15-29.
33. Llorente AM, Jensen CL, Voigt RG, et al. Effect of maternal docosahexaenoic acid supplementation on postpartum depression and information processing. Am J Obstet Gynecol 2003;188(5):1348-53.
34. Zanarini MC, Frankenburg FR. Omega-3 fatty acid treatment of women with borderline personality disorder: a double-blind, placebo-controlled pilot study. Am J Psychiatry 2003;160(1):167-9.
35. Mitchell EA, Aman MG, Turbott SH, Manku M. Clinical characteristics and serum essential fatty acid levels in hyperactive children. Clin Pediatr 1987;26(8):406-11.
36. Stevens LJ, Zentall SS, Deck JL, et al. Essential fatty acid metabolism in boys with attention-deficit hyperactivity disorder. Am J Clin Nutr 1995;62(4):761-8.
37. Stevens LJ, Zentall SS, Abate ML, et al. Omega-3 fatty acids in boys with behavior, learning, and health problems. Physiol Behav 1996;59(4-5):915-20.
38. Voigt RG, Llorente AM, Jensen CL, et al. A randomized, double-blind, placebo-controlled trial of docosahexaenoic acid supplementation in children with attention-deficit/hyperactivity disorder. J Pediatr 2001;139(2):189-96.
39. Kalmijn S, Launer LJ, Ott A, et al. Dietary fat intake and the risk of incident dementia in the Rotterdam Study. Ann Neurol 1997;42(5):776-82.
40. Barberger-Gateau P, Letenneur L, Deschamps V, et al. Fish, meat, and risk of dementia: cohort study. BMJ 2002;325(7370):932-3.
41. Morris MC, Evans DA, Bienias JL, et al. Consumption of fish and n-3 fatty acids and risk of incident Alzheimer disease. Arch Neurol 2003;60(7):940-6.
42. Engelhart MJ, Geerlings MI, Ruitenberg A, et al. Diet and risk of dementia: does fat matter? The Rotterdam Study. Neurology 2002;59(12):1915-21.
1. Hibbeln JR. Fish consumption and major depression. Lancet 1998;351(9110):1213.-
2. Tanskanen A, Hibbeln JR, Tuomilehto J, et al. Fish consumption and depressive symptoms in the general population in Finland. Psychiatr Serv 2001;52(4):529-31.
3. Tanskanen A, Hibelln JR, Hintikka J, et al. Fish consumption, depression, and suicidality in a general population. Arch Gen Psychiatry 2001;58(5):512-3.
4. Edwards R, Peet M, Shay J, Horrobin D. Omega-3 polyunsaturated fatty acid levels in the diet and in red blood cell membranes of depressed patients. J Affect Disord 1998;48(2-3):149-55.
5. Maes M, Christophe A, Delanghe J, et al. Lowered omega-3 polyunsaturated fatty acids in serum phospholipids and cholesterol esters of depressed patients. Psychiatry Res 1999;85(3):275-91.
6. Lundbaek JA, Andersen OS. Lysophospholipids modulate channel function by altering the mechanical properties of lipid bilayers. J Gen Physiol 1994;104(4):645-73.
7. Delion S, Chalon S, Guilloteau D, et al. Alpha-linolenic acid dietary deficiency alters age-related changes of dopaminergic and serotonergic neurotransmission in the rat frontal cortex. J Neurochem 1996;66(4):1582-91.
8. Passi S, Cataudella S, Di Marco P, et al. Fatty acid composition and antioxidant levels in muscle tissue of different Mediterranean marine species of fish and shellfish. J Agric Food Chem 2002;50(25):7314-22.
9. Hudson CJ, Young LT, Li PP, Warsh JJ. CNS signal transduction in the pathophysiology and pharmacology of affective disorders and schizophrenia. Synapse 1993;13(3):278-93.
10. Sperling RI, Benincaso AI, Knoell CT, et al. Dietary omega-3 polyunsaturated fatty acids inhibit phosphoinositide formation and chemotaxis in neutrophils. J Clin Invest 1993;91(2):651-60.
11. Endres S. Messengers and mediators: interactions among lipids, eicosanoids and cytokines. Am J Clin Nutr 1993;57(5 suppl):798S-800S.
12. Kris-Etherton PM, Harris WS, Appel LJ. Fish consumption, fish oil, omega-3 fatty acids, and cardiovascular disease. Arterioscler Thromb Vasc Biol 2003;23(2):e20-e30.
13. Nemets B, Stahl Z, Belmaker RH. Addition of omega-3 fatty acid to maintenance medication treatment for recurrent unipolar depressive disorder. Am J Psychiatry 2002;159(3):477-9.
14. Peet M, Horrobin DF. A dose-ranging study of the effects of ethyl-eicosapentaenoate in patients with ongoing depression despite apparently adequate treatment with standard drugs. Arch Gen Psychiatry 2002;59(10):913-9.
15. Su K-P, Huang S-Y, Chiu C-C, Shen WW. Omega-3 fatty acids in major depressive disorder: a preliminary double-blind, placebo-con-trolled trial. Eur Neuropsychopharmacol 2003;13(4):267-71.
16. Marangell LB, Martinez JM, Zboyan HA, et al. A double-blind, placebo-controlled study of the omega-3 fatty acid docosahexaenoic acid in the treatment of major depression. Am J Psychiatry 2003;160(5):996-8.
17. Stoll AL, Severus WE. Mood stabilizers: shared mechanisms of action at postsynaptic signal transduction and kindling processes. Harv Rev Psychiatry 1996;4(2):77-89.
18. Chiu CC, Huang SY, Su KP, et al. Polyunsaturated fatty acid deficit in patients with bipolar disorder. Eur Neuropsychopharmacol 2003;13(2):99-103.
19. Stoll AL, Severus WE, Freeman MP, et al. Omega-3 fatty acids in bipolar disorder: a preliminary double-blind, placebo-controlled trial. Arch Gen Psychiatry 1999;56(5):407-12.
20. Keck PE, Jr, McElroy SL, Freeman MP, et al. Randomized, placebo-controlled trial of eicosapentaenoic acid in bipolar depression. Bipolar Disord 2003;5(suppl 1):58.-
21. Keck PE, Jr, McElroy SL, Freeman MP, et al. Randomized, placebo-controlled trial of eicosapentaenoic acid in rapid cycling bipolar disorder. Bipolar Disord 2003;5(suppl 1):58.-
22. Horrobin DF. The membrane phospholipid hypothesis as a biochemical basis for the neurodevelopmental concept of schizophrenia. Schizophr Res 1998;30(3):193-208.
23. Christensen O, Christensen E. Fat consumption and schizophrenia. Acta Psychiatr Scand 1988;78(5):587-591
24. Mellor JE, Laugharne JD, Peet M. Schizophrenic symptoms and dietary intake of n-3 fatty acids. Schizophr Res 1995;18(1):85-6.
25. Arvindakshan M, Ghate M, Ranjekar PK, et al. Supplementation with a combination of omega-3 fatty acids and antioxidants (vitamins E and C) improves the outcome of schizophrenia. Schizophr Res 2003;62(3):195-204.
26. Peet M, Brind J, Ramchand CN, et al. Two double-blind placebo-controlled pilot studies of eicosapentaenoic acid in the treatment of schizophrenia. Schizophr Res 2001;49(3):243-51.
27. Fenton WS, Dickerson F, Boronow J, et al. A placebo-controlled trial of omega-3 fatty acid (ethyl eicosapentaenoic acid) supplementation for residual symptoms and cognitive impairment in schizophrenia. Am J Psychiatry 2001;158(12):2071-4.
28. Peet M, Horrobin DF. E-E Multicentre Study Group. A dose-ranging exploratory study of the effects of ethyl-eicosapentaenoate in patients with persistent schizophrenic symptoms. J Psychiatr Res 2002;36(1):7-18.
29. Emsley R, Myburgh C, Oosthuizen P, Van Rensburg SJ. Randomized, placebo-controlled study of ethyl-eicosapentaenoic acid as supplemental treatment in schizophrenia. Am J Psychiatry 2002;159(9):1596-8.
30. Al MD, van Houwelingen AC, Kester AD, et al. Maternal essential fatty acid patterns during normal pregnancy and their relationship to neonatal essential fatty acid status. Br J Nutr 1995;74(1):55-68.
31. Hibbeln JR, Salem N, Jr. Dietary polyunsaturated fatty acids and depression: when cholesterol does not satisfy. Am J Clin Nutr 1995;62(1):1-9.
32. Hibbeln JR. Seafood consumption, the DHA content of mothers’ milk and prevalence rates of postpartum depression: a cross-national, ecological analysis. J Affect Disord 2002;69(1-3):15-29.
33. Llorente AM, Jensen CL, Voigt RG, et al. Effect of maternal docosahexaenoic acid supplementation on postpartum depression and information processing. Am J Obstet Gynecol 2003;188(5):1348-53.
34. Zanarini MC, Frankenburg FR. Omega-3 fatty acid treatment of women with borderline personality disorder: a double-blind, placebo-controlled pilot study. Am J Psychiatry 2003;160(1):167-9.
35. Mitchell EA, Aman MG, Turbott SH, Manku M. Clinical characteristics and serum essential fatty acid levels in hyperactive children. Clin Pediatr 1987;26(8):406-11.
36. Stevens LJ, Zentall SS, Deck JL, et al. Essential fatty acid metabolism in boys with attention-deficit hyperactivity disorder. Am J Clin Nutr 1995;62(4):761-8.
37. Stevens LJ, Zentall SS, Abate ML, et al. Omega-3 fatty acids in boys with behavior, learning, and health problems. Physiol Behav 1996;59(4-5):915-20.
38. Voigt RG, Llorente AM, Jensen CL, et al. A randomized, double-blind, placebo-controlled trial of docosahexaenoic acid supplementation in children with attention-deficit/hyperactivity disorder. J Pediatr 2001;139(2):189-96.
39. Kalmijn S, Launer LJ, Ott A, et al. Dietary fat intake and the risk of incident dementia in the Rotterdam Study. Ann Neurol 1997;42(5):776-82.
40. Barberger-Gateau P, Letenneur L, Deschamps V, et al. Fish, meat, and risk of dementia: cohort study. BMJ 2002;325(7370):932-3.
41. Morris MC, Evans DA, Bienias JL, et al. Consumption of fish and n-3 fatty acids and risk of incident Alzheimer disease. Arch Neurol 2003;60(7):940-6.
42. Engelhart MJ, Geerlings MI, Ruitenberg A, et al. Diet and risk of dementia: does fat matter? The Rotterdam Study. Neurology 2002;59(12):1915-21.