User login
Codes, Contracts, and Commitments: Who Defines What is a Profession?
Codes, Contracts, and Commitments: Who Defines What is a Profession?
A professional is someone who can do his best work when he doesn’t feel like it.
Alistair Cooke
When I was a young person with no idea about growing up to be something, my father used to tell me there were 4 learned professions: medicine to heal the body, law to protect the body politic, teaching to nurture the mind, and the clergy to care for the soul.1 That adage, or some version of it, is attributed to a variety of sources, likely because it captures something essential and timeless about the learned professions. I write this as a much older person, and it has been my privilege to have worked in some capacity in all 4 of these venerable vocations.
There are many more recognized professions now than in my father’s time with new ones still emerging as the world becomes more complicated and specialized. In November 2025, however, the growth of the professions was dealt a serious blow when the US Department of Education (DOE) redefined what constitutes a profession for the purpose of federal funding of graduate degrees.2 The internet is understandably abuzz with opinions across the political spectrum. What is missing from many of these discussions is an understanding of the criteria for a profession and, even more importantly, who has the authority to decide when an individual or a group has met that standard.
But first, what and why did the DOE make this change? The One Big Beautiful Bill Act charged the DOE with reducing what it claims is massive overspending on graduate education by limiting the programs that meet the definition of a “professional degree” eligible for higher funding. Of my father’s 4, medicine (including dentistry) and law made the cut with students in those professions able to borrow up to $200,000 in direct unsubsidized student loans while those in other programs would be limited to $100,000.2
As one of the oldest and most respected professions in America, nursing has received the most media attention, yet there are also other important and valued professions that are missing from the DOE list.3 The excluded professions also include: physician assistants, physical therapists, audiologists, architects, accountants, educators, and social workers. The proposed regulatory changes are not yet finalized and Congressional representatives, health care experts, and a myriad of professional associations have rightly objected the reclassification will only worsen the critical shortage of nurses, teachers, and other helping professions the country is already facing.4
There are thousands of federal health care professionals who worked long and hard to achieve their goals whom this Act undervalues. Moreover, the regulatory change leaves many students enrolled in education and training programs under federal practice auspices confused and overwhelmed. Perhaps they can take some hope and inspiration from the recognition that historically and philosophically, no agency or administration can unilaterally define what is a profession.
The literature on professionalism is voluminous, in large part because it has been surprisingly difficult to reach a consensus definition. A proposed definition from scholars captures most of the key aspects of a profession. While it is drawn from the medical literature, it applies to most of the caring professions the DOE disqualified. For pedagogic purposes, the definition is parsed into discrete criteria in the Table.5

Even this simple summary makes it obvious that a government agency alone could not possibly have the competence to determine who meets these complex technical and moral criteria. The members of the profession must assume a primary role in that determination. The complicated history of the professions shows that the locus of these decisions has resided in various combinations of educational institutions, such as nursing schools,6 professional societies (eg, National Association of Social Workers),7 and certifying boards (eg, National Commission on Certification of Physician Assistants).8 States, not the federal government, have long played a key part in defining professions in the US, through their authority to grant licenses to practice.9
In response to criticism, the DOE has stated that “the definition of a ‘professional degree’ is an internal definition used by the Department of Education to distinguish among programs that qualify for higher loan limits, not a value judgment about the importance of programs. It has no bearing on whether a program is professional in nature or not.”2 Given the ancient compact between society and the professions in which the government subsidizes the training of professionals dedicated to public service, it is hard to see how these changes can be dismissed as merely semantic and not a promissory breach.10
I recognize that this abstract editorial is little comfort to beleaguered and demoralized professionals and students. Still, it offers a voice of support for each federal practitioner or trainee who fulfills the epigraph’s description of a professional day after day. The nurse who works the extra shift without complaint or resentment so that veterans receive the care they deserve, the social worker who responds on a weekend night to an active duty family without food so they do not spend another night hungry, and the physician assistant who makes it into the isolated public health clinic despite the terrible weather so there is someone ready to take care for patients in need. The proposed policy shift cannot in any meaningful sense rob them of their identity as individuals committed to a code of caring. However, without an intact social compact, it may well remove their practical ability to remain and enter the helping professions to the detriment of us all.
- Wade JW. Public responsibilities of the learned professions. Louisiana Law Rev. 1960;21:130-148
- US Department of Education. Myth vs. fact: the definition of professional degrees. Press Release. November 24, 2025. Accessed December 22, 2025. https://www.ed.gov/about/news/press-release/myth-vs-fact-definition-of-professional-degrees
- Laws J. Full list of degrees not classed as “professional” by Trump admin. Newsweek. Updated November 26, 2025. Accessed December 22, 2025. https://www.newsweek.com/full-list-degrees-professional-trump-administration-11085695
- New York Academy of Medicine. Response to stripping “professional status” as proposed by the Department of Education. New York Academy of Medicine. November 24, 2025. Accessed December 22, 2025. https://nyam.org/article/response-to-stripping-professional-status-as-proposed-by-the-department-of-education
- Cruess SR, Johnston S, Cruess RL. “Profession”: a working definition for medical educators. Teach Learn Med. 2004;16:74-76. doi:10.1207/s15328015tlm1601_15
- American Association of Colleges of Nursing. Nursing is a professional degree. American Association of Colleges of Nursing. Accessed December 20, 2025. https://www.aacnnursing.org/policy-advocacy/take-action/nursing-is-a-professional-degree
- National Association of Social Workers. Social work is a profession. Social Workers. Accessed December 20, 2025. https://www.socialworkers.org
- National Commission on Certification of Physician Assistants. Accessed December 20, 2025. https://www.nccpa.net/about-nccpa/#who-we-are
- The Federation of State Boards of Physical Therapy. Accessed December 20, 2025. https://www.fsbpt.org/About-Us/Staff-Home
- Cruess SR, Cruess RL. Professionalism and medicine’s contract with social contract with society. Virtual Mentor. 2004;6:185-188. doi:10.1001/virtualmentor.2004.6.4.msoc1-040
A professional is someone who can do his best work when he doesn’t feel like it.
Alistair Cooke
When I was a young person with no idea about growing up to be something, my father used to tell me there were 4 learned professions: medicine to heal the body, law to protect the body politic, teaching to nurture the mind, and the clergy to care for the soul.1 That adage, or some version of it, is attributed to a variety of sources, likely because it captures something essential and timeless about the learned professions. I write this as a much older person, and it has been my privilege to have worked in some capacity in all 4 of these venerable vocations.
There are many more recognized professions now than in my father’s time with new ones still emerging as the world becomes more complicated and specialized. In November 2025, however, the growth of the professions was dealt a serious blow when the US Department of Education (DOE) redefined what constitutes a profession for the purpose of federal funding of graduate degrees.2 The internet is understandably abuzz with opinions across the political spectrum. What is missing from many of these discussions is an understanding of the criteria for a profession and, even more importantly, who has the authority to decide when an individual or a group has met that standard.
But first, what and why did the DOE make this change? The One Big Beautiful Bill Act charged the DOE with reducing what it claims is massive overspending on graduate education by limiting the programs that meet the definition of a “professional degree” eligible for higher funding. Of my father’s 4, medicine (including dentistry) and law made the cut with students in those professions able to borrow up to $200,000 in direct unsubsidized student loans while those in other programs would be limited to $100,000.2
As one of the oldest and most respected professions in America, nursing has received the most media attention, yet there are also other important and valued professions that are missing from the DOE list.3 The excluded professions also include: physician assistants, physical therapists, audiologists, architects, accountants, educators, and social workers. The proposed regulatory changes are not yet finalized and Congressional representatives, health care experts, and a myriad of professional associations have rightly objected the reclassification will only worsen the critical shortage of nurses, teachers, and other helping professions the country is already facing.4
There are thousands of federal health care professionals who worked long and hard to achieve their goals whom this Act undervalues. Moreover, the regulatory change leaves many students enrolled in education and training programs under federal practice auspices confused and overwhelmed. Perhaps they can take some hope and inspiration from the recognition that historically and philosophically, no agency or administration can unilaterally define what is a profession.
The literature on professionalism is voluminous, in large part because it has been surprisingly difficult to reach a consensus definition. A proposed definition from scholars captures most of the key aspects of a profession. While it is drawn from the medical literature, it applies to most of the caring professions the DOE disqualified. For pedagogic purposes, the definition is parsed into discrete criteria in the Table.5

Even this simple summary makes it obvious that a government agency alone could not possibly have the competence to determine who meets these complex technical and moral criteria. The members of the profession must assume a primary role in that determination. The complicated history of the professions shows that the locus of these decisions has resided in various combinations of educational institutions, such as nursing schools,6 professional societies (eg, National Association of Social Workers),7 and certifying boards (eg, National Commission on Certification of Physician Assistants).8 States, not the federal government, have long played a key part in defining professions in the US, through their authority to grant licenses to practice.9
In response to criticism, the DOE has stated that “the definition of a ‘professional degree’ is an internal definition used by the Department of Education to distinguish among programs that qualify for higher loan limits, not a value judgment about the importance of programs. It has no bearing on whether a program is professional in nature or not.”2 Given the ancient compact between society and the professions in which the government subsidizes the training of professionals dedicated to public service, it is hard to see how these changes can be dismissed as merely semantic and not a promissory breach.10
I recognize that this abstract editorial is little comfort to beleaguered and demoralized professionals and students. Still, it offers a voice of support for each federal practitioner or trainee who fulfills the epigraph’s description of a professional day after day. The nurse who works the extra shift without complaint or resentment so that veterans receive the care they deserve, the social worker who responds on a weekend night to an active duty family without food so they do not spend another night hungry, and the physician assistant who makes it into the isolated public health clinic despite the terrible weather so there is someone ready to take care for patients in need. The proposed policy shift cannot in any meaningful sense rob them of their identity as individuals committed to a code of caring. However, without an intact social compact, it may well remove their practical ability to remain and enter the helping professions to the detriment of us all.
A professional is someone who can do his best work when he doesn’t feel like it.
Alistair Cooke
When I was a young person with no idea about growing up to be something, my father used to tell me there were 4 learned professions: medicine to heal the body, law to protect the body politic, teaching to nurture the mind, and the clergy to care for the soul.1 That adage, or some version of it, is attributed to a variety of sources, likely because it captures something essential and timeless about the learned professions. I write this as a much older person, and it has been my privilege to have worked in some capacity in all 4 of these venerable vocations.
There are many more recognized professions now than in my father’s time with new ones still emerging as the world becomes more complicated and specialized. In November 2025, however, the growth of the professions was dealt a serious blow when the US Department of Education (DOE) redefined what constitutes a profession for the purpose of federal funding of graduate degrees.2 The internet is understandably abuzz with opinions across the political spectrum. What is missing from many of these discussions is an understanding of the criteria for a profession and, even more importantly, who has the authority to decide when an individual or a group has met that standard.
But first, what and why did the DOE make this change? The One Big Beautiful Bill Act charged the DOE with reducing what it claims is massive overspending on graduate education by limiting the programs that meet the definition of a “professional degree” eligible for higher funding. Of my father’s 4, medicine (including dentistry) and law made the cut with students in those professions able to borrow up to $200,000 in direct unsubsidized student loans while those in other programs would be limited to $100,000.2
As one of the oldest and most respected professions in America, nursing has received the most media attention, yet there are also other important and valued professions that are missing from the DOE list.3 The excluded professions also include: physician assistants, physical therapists, audiologists, architects, accountants, educators, and social workers. The proposed regulatory changes are not yet finalized and Congressional representatives, health care experts, and a myriad of professional associations have rightly objected the reclassification will only worsen the critical shortage of nurses, teachers, and other helping professions the country is already facing.4
There are thousands of federal health care professionals who worked long and hard to achieve their goals whom this Act undervalues. Moreover, the regulatory change leaves many students enrolled in education and training programs under federal practice auspices confused and overwhelmed. Perhaps they can take some hope and inspiration from the recognition that historically and philosophically, no agency or administration can unilaterally define what is a profession.
The literature on professionalism is voluminous, in large part because it has been surprisingly difficult to reach a consensus definition. A proposed definition from scholars captures most of the key aspects of a profession. While it is drawn from the medical literature, it applies to most of the caring professions the DOE disqualified. For pedagogic purposes, the definition is parsed into discrete criteria in the Table.5

Even this simple summary makes it obvious that a government agency alone could not possibly have the competence to determine who meets these complex technical and moral criteria. The members of the profession must assume a primary role in that determination. The complicated history of the professions shows that the locus of these decisions has resided in various combinations of educational institutions, such as nursing schools,6 professional societies (eg, National Association of Social Workers),7 and certifying boards (eg, National Commission on Certification of Physician Assistants).8 States, not the federal government, have long played a key part in defining professions in the US, through their authority to grant licenses to practice.9
In response to criticism, the DOE has stated that “the definition of a ‘professional degree’ is an internal definition used by the Department of Education to distinguish among programs that qualify for higher loan limits, not a value judgment about the importance of programs. It has no bearing on whether a program is professional in nature or not.”2 Given the ancient compact between society and the professions in which the government subsidizes the training of professionals dedicated to public service, it is hard to see how these changes can be dismissed as merely semantic and not a promissory breach.10
I recognize that this abstract editorial is little comfort to beleaguered and demoralized professionals and students. Still, it offers a voice of support for each federal practitioner or trainee who fulfills the epigraph’s description of a professional day after day. The nurse who works the extra shift without complaint or resentment so that veterans receive the care they deserve, the social worker who responds on a weekend night to an active duty family without food so they do not spend another night hungry, and the physician assistant who makes it into the isolated public health clinic despite the terrible weather so there is someone ready to take care for patients in need. The proposed policy shift cannot in any meaningful sense rob them of their identity as individuals committed to a code of caring. However, without an intact social compact, it may well remove their practical ability to remain and enter the helping professions to the detriment of us all.
- Wade JW. Public responsibilities of the learned professions. Louisiana Law Rev. 1960;21:130-148
- US Department of Education. Myth vs. fact: the definition of professional degrees. Press Release. November 24, 2025. Accessed December 22, 2025. https://www.ed.gov/about/news/press-release/myth-vs-fact-definition-of-professional-degrees
- Laws J. Full list of degrees not classed as “professional” by Trump admin. Newsweek. Updated November 26, 2025. Accessed December 22, 2025. https://www.newsweek.com/full-list-degrees-professional-trump-administration-11085695
- New York Academy of Medicine. Response to stripping “professional status” as proposed by the Department of Education. New York Academy of Medicine. November 24, 2025. Accessed December 22, 2025. https://nyam.org/article/response-to-stripping-professional-status-as-proposed-by-the-department-of-education
- Cruess SR, Johnston S, Cruess RL. “Profession”: a working definition for medical educators. Teach Learn Med. 2004;16:74-76. doi:10.1207/s15328015tlm1601_15
- American Association of Colleges of Nursing. Nursing is a professional degree. American Association of Colleges of Nursing. Accessed December 20, 2025. https://www.aacnnursing.org/policy-advocacy/take-action/nursing-is-a-professional-degree
- National Association of Social Workers. Social work is a profession. Social Workers. Accessed December 20, 2025. https://www.socialworkers.org
- National Commission on Certification of Physician Assistants. Accessed December 20, 2025. https://www.nccpa.net/about-nccpa/#who-we-are
- The Federation of State Boards of Physical Therapy. Accessed December 20, 2025. https://www.fsbpt.org/About-Us/Staff-Home
- Cruess SR, Cruess RL. Professionalism and medicine’s contract with social contract with society. Virtual Mentor. 2004;6:185-188. doi:10.1001/virtualmentor.2004.6.4.msoc1-040
- Wade JW. Public responsibilities of the learned professions. Louisiana Law Rev. 1960;21:130-148
- US Department of Education. Myth vs. fact: the definition of professional degrees. Press Release. November 24, 2025. Accessed December 22, 2025. https://www.ed.gov/about/news/press-release/myth-vs-fact-definition-of-professional-degrees
- Laws J. Full list of degrees not classed as “professional” by Trump admin. Newsweek. Updated November 26, 2025. Accessed December 22, 2025. https://www.newsweek.com/full-list-degrees-professional-trump-administration-11085695
- New York Academy of Medicine. Response to stripping “professional status” as proposed by the Department of Education. New York Academy of Medicine. November 24, 2025. Accessed December 22, 2025. https://nyam.org/article/response-to-stripping-professional-status-as-proposed-by-the-department-of-education
- Cruess SR, Johnston S, Cruess RL. “Profession”: a working definition for medical educators. Teach Learn Med. 2004;16:74-76. doi:10.1207/s15328015tlm1601_15
- American Association of Colleges of Nursing. Nursing is a professional degree. American Association of Colleges of Nursing. Accessed December 20, 2025. https://www.aacnnursing.org/policy-advocacy/take-action/nursing-is-a-professional-degree
- National Association of Social Workers. Social work is a profession. Social Workers. Accessed December 20, 2025. https://www.socialworkers.org
- National Commission on Certification of Physician Assistants. Accessed December 20, 2025. https://www.nccpa.net/about-nccpa/#who-we-are
- The Federation of State Boards of Physical Therapy. Accessed December 20, 2025. https://www.fsbpt.org/About-Us/Staff-Home
- Cruess SR, Cruess RL. Professionalism and medicine’s contract with social contract with society. Virtual Mentor. 2004;6:185-188. doi:10.1001/virtualmentor.2004.6.4.msoc1-040
Codes, Contracts, and Commitments: Who Defines What is a Profession?
Codes, Contracts, and Commitments: Who Defines What is a Profession?
Can Telehealth Improve Access to Amyloid-Targeting Therapies for Veterans Living With Alzheimer Disease?
Can Telehealth Improve Access to Amyloid-Targeting Therapies for Veterans Living With Alzheimer Disease?
The Veterans Health Administration (VHA) is the largest US integrated health care system, providing health care to > 9 million veterans annually. Dementia affects > 7.2 million Americans, and an estimated 450,000 veterans live with Alzheimer disease (AD).1,2 Compared with the general population, veterans have a higher burden of chronic medical conditions and are disproportionately affected by AD due to exposure to military-related risk factors (eg, traumatic brain injury and posttraumatic stress disorder) and the high prevalence of nonmilitary risk factors, such as cardiovascular disease. The VHA is a pioneer in dementia care, having established a Dementia System of Care to provide primary and specialty care to veterans with dementia. The VHA also is leading the way in implementing the Institute for Healthcare Improvement Age-Friendly Health Systems (AFHS) framework for providing goal-concordant care in > 100 VHA medical centers. The VHA aims to be the largest AFHS in the country.
AD profoundly affects individuals and their families. The progressive nature of the most common form of dementia diminishes the quality of life for patients as well as their care partners in an ongoing fashion, often leading to emotional, physical, and financial strain. Costs for health and long-term care for people living with AD and other dementias were projected at $360 billion in 2024, largely due to the need for nursing home care.1 Although several oral medications are available, their capacity to effectively mitigate the negative effects of AD is limited. Cholinesterase inhibitors and memantine may offer temporary symptomatic relief, but they do not alter disease progression.3 The use of these agents is relatively low, with about one-third of patients diagnosed with AD receiving these medications.4
Amyloid-Targeting Therapies
Recent advancements in biologics, particularly amyloid-targeting therapies, such as lecanemab and donanemab, offer new hope for managing AD. Older adults treated with these medications show less decline on measures of cognition and function than those receiving a placebo at 18 months.5,6 However, accessing and using these medications is challenging.
Use of amyloid-targeting therapies poses challenges. The medications are expensive, potentially placing a financial burden on patients, families, and health care systems.7 Determining initial eligibility for treatment requires a battery of cognitive assessments, laboratory tests, advanced radiologic studies (eg, magnetic resonance imaging [MRI] of the brain and amyloid positron emission tomography [PET] scans), and possible cerebrospinal fluid (CSF) testing. Frequent ongoing assessments are necessary to monitor safety and efficacy. These treatments carry substantial risks, particularly amyloid-related imaging abnormalities (ARIA) such as cerebral edema, microhemorrhages, and superficial siderosis. Therefore, follow-up assessments typically occur around months 2, 3, 4, and 7, depending on which medication is selected. Finally, at present, both agents must be intravenous (IV)-administered in a monitored clinical setting, which requires additional coordination, transportation, and cost.
Ongoing evaluations and in-person administration particularly affect patients and care partners with limitations regarding transportation, time off work, and navigating complex health care systems.8 VHA clinicians at sites that have implemented or are interested in implementing amyloid-targeting therapy programs endorse similar challenges when implementing these therapies in their US Department of Veterans Affairs (VA) medical centers (VAMCs).9
The VHA was one of the first health care systems to use amyloid-targeting therapies, covering the cost of lecanemab and donanemab, in addition to costs associated with concomitant evaluation and testing. However, given the safety concerns with this novel class of medications, the VHA National Formulary Committee developed criteria for use and recommended the VA Center for Medication Safety (VAMedSAFE) conduct a mandatory real-time medication use evaluation (MUE). VAMedSAFE developed the MUE to monitor the safe and appropriate use of amyloid-targeting therapy for AD. Two authors (AJO, SMH) partnered with VAMedSAFE through the VA Pittsburgh Healthcare System Technology Enhancing Cognition and Health–Geriatric Research, Education, and Clinical Center (TECH-GRECC) to provide clinical expertise, substantive feedback for the development of the MUE, and guidance for VHA sites starting amyloid targeting-therapy programs. We started a VHA Amyloid-Targeting Therapy for AD SharePoint collaborative platform and VHA AD Therapeutics Community of Practice (CoP) for shared learning (Figure). The private SharePoint platform houses an array of implementation materials for VAMCs starting programs: key documents and links; educational materials; sample guidelines; note templates; and electronic health record screenshots. The CoP allows VHAs to share best practices and discuss challenges.
Even with these advantages, we found that ensuring the safe and appropriate use of amyloid-targeting therapies did not overcome the barriers associated with their complexity. This was especially true for veterans living in rural areas. Only 4 VAMCs had administered amyloid-targeting therapies in the first year they were available. Preliminary data demonstrated that 27 (84%) of 32 veterans who initiated lecanemab in the VHA between October 2023 and September 2024 resided in urban areas.10 To address the underutilization of amyloid-targeting therapy, we propose leveraging the strengths of VHA telehealth to facilitate expansion of access to these medications for veterans with early AD. Telehealth may substantially increase access to evaluation for veterans with early dementia and, when medically appropriate, to receive amyloid-targeting therapies by reducing transportation needs and mitigating costs while ensuring appropriate monitoring through ongoing clinical assessments.
Using Telehealth
The VHA is a pioneer in telehealth, with programs dating back to 2003.11 Between October 1, 2018, and September 30, 2019, the VHA served > 900,000 veterans through the provision of > 2.6 million episodes of care via telehealth.12 The COVID-19 pandemic further cemented the role of telemedicine as an essential component of health care. Telehealth has demonstrated success in the assessment and management of individuals living with dementia. At the VHA, the GRECC-Connect Project is a partnership between 9 urban GRECC sites that seek to provide consultative geriatric and dementia care to rural veterans through telehealth.13 Additional evidence supports the potential to leverage telehealth to effectively communicate results of amyloid PET scans.14
This approach is not without limitations such as the digital divide, or the gap that separates technology-enabled individuals and those unprepared to adopt technology due to limited digital literacy levels or access to needed hardware, software, and connectivity. The VHA has taken steps to address these digital divide barriers by broadly providing tools—such as tablets and broadband connectivity—to veterans. Specifically, the VHA has instituted digital divide consults to determine whether telehealth could be a potential solution for appropriate veterans and to provide an iPad (if eligible) to connect with VA clinicians. Complementary to the digital divide consult, a VHA-specific telehealth preparedness assessment tool is under development and being tested by 2 authors (JF, SMH). This telehealth preparedness assessment tool is designed to aid in the seamless integration of telehealth services with the support of tailored education materials specific to gaps in digital literacy that a veteran might experience.
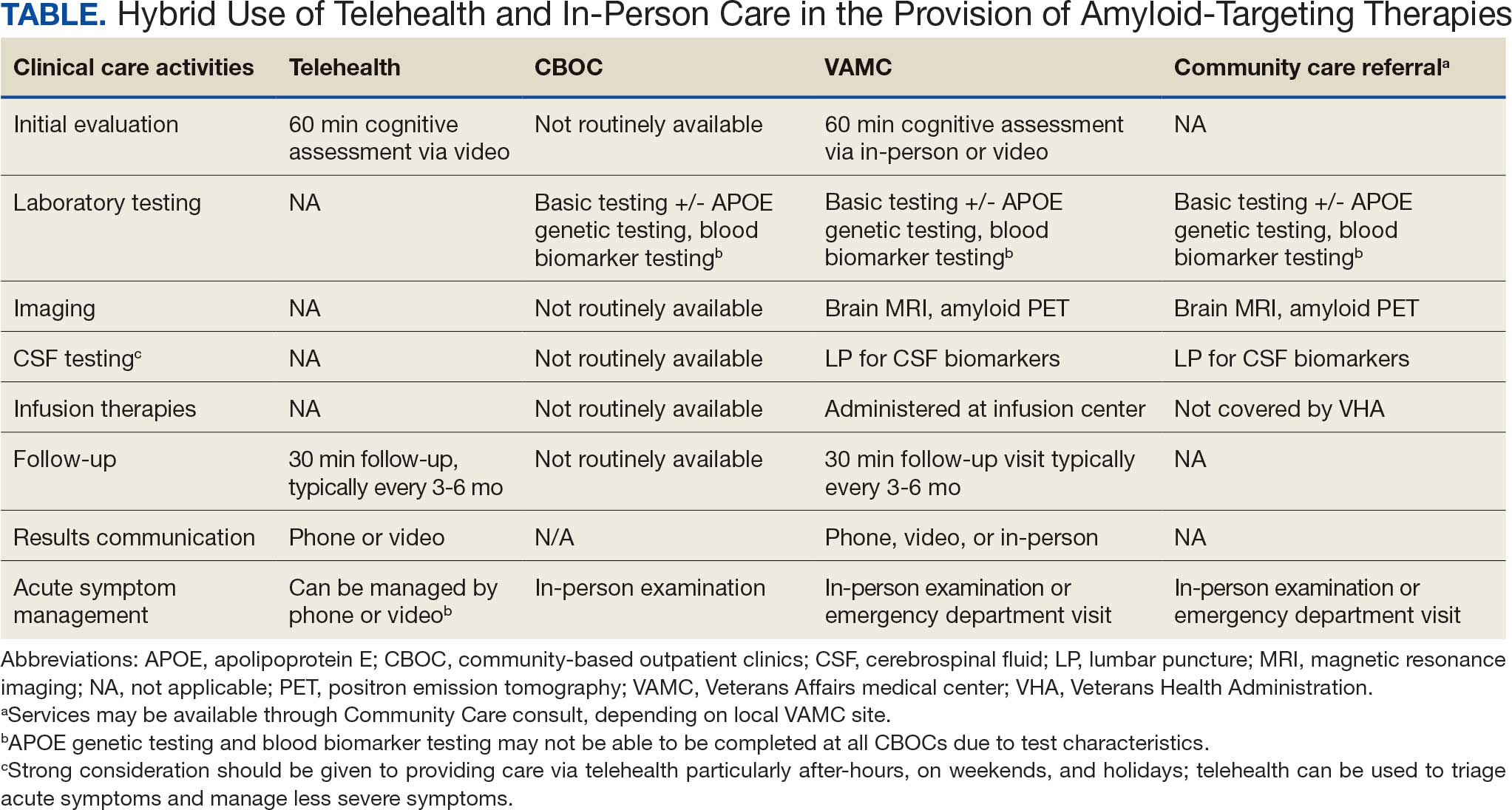
Building on these initiatives, there is an opportunity to expand access to amyloid-targeting therapies, regardless of distance to large VAMCs, by leveraging telehealth as an alternative method of connecting patients with specialty care. Specifically, a hybrid approach could be used to accomplish the myriad initial and follow-up tasks involved in the provision of amyloid-targeting therapies (Table). Not all VHA facilities possess the specialty expertise to prescribe these medications, and local clinicians may not have sufficient knowledge and clinical support to prescribe and monitor these therapies.
The first step is identifying local and regional subject matter experts, followed by the development and expansion of these networks. The National TeleNeurology Program is a good example of a national telehealth program that leverages technology to bring specialty services to rural areas with limited access to care. Although amyloid-targeting therapies often require more complex logistics, such as laboratory tests and imaging, these initial hurdles can be overcome through localized services and collaboration between VAMCs.
While treatment and imaging will most likely need to occur at a VAMC, most basic laboratory studies can be performed at community-based outpatient clinics (CBOCs). Some CBOCs may not be able to process more specialized laboratory tests such as apolipoprotein E genetic testing. Samples for these tests can be collected and processed at VAMCs, which usually have contracts with outside laboratories capable of performing these studies. Most, although not all, VAMCs offer advanced imaging, including MRI of the brain and amyloid PETs. VAMCs without those modalities may need to coordinate with other regional VAMCs. Additionally, a pilot program is already underway whereby VAMCs without the ability to quantify the amount of amyloid on PETs are able to leverage technology and collaborations with other VAMCs to obtain these data.
Once the initial phases of evaluation and care are completed, telemedicine can be leveraged for follow-up and ongoing management. Interdisciplinary teams can help facilitate care related to amyloid-targeting therapies, including the close monitoring of veterans for development of ARIA.15 To achieve this monitoring, specialty clinic teams prescribing amyloid-targeting therapies, which may be geographically distant, need to coordinate with local primary care clinical teams and emergency clinicians. All of these health care team members, along with neurologists and neurosurgeons, should be involved in the development and implementation of protocols in the event that patients present to their local primary or specialty care clinics or emergency department with ARIA symptoms.
If amyloid-targeting therapies are to be provided along with other emerging treatments for rural veterans, telehealth must be part of the solution. There is a pressing need to explore innovative evaluation and delivery models for these therapies, particularly as we expect additional diagnostics and therapeutics to be available in the future. With the advent of commercially available blood tests (ie, blood biomarkers) for AD, there is hope for a transition away from PETs and CSF testing given their cost, limited access, and invasiveness for diagnosis and monitoring of AD. These advances will increase the utility of telehealth to help rural veterans access amyloid-targeting therapies.
Additionally, administering the drug at home or at local clinics, supported by a dedicated health care team or home health agency, could further improve accessibility. Telehealth can be leveraged in this scenario, allowing specialty clinics and specialists to connect with patients and clinicians based out of local clinics or even home health agencies. In this scenario, specialists can provide hands-on care guidance and oversight even though they may be geographically distant from care recipients. Transitioning from IV administration to subcutaneous formulations would further enhance convenience and reduce barriers; these formulations may be available soon.16 Addressing logistical challenges to care and access through technology-based solutions will require coordinated efforts and continued VHA investment.
Conclusions
The VHA has a large population of veterans with dementia, and the costs to care for these veterans will only increase. While the current benefits of amyloid-targeting therapies are modest, now is the time to establish care processes that will support future innovations in amyloid-targeting therapies and other treatments and diagnostics. We are developing better ways to detect AD using clinical decision support tools, improving care pathways and the management of AD, and leveraging telehealth to improve access. The VA is conducting research to investigate whether a cognitive screening and laboratory evaluation that includes a telehealth preparedness assessment will be feasible and effective for improving the detection of AD and access to treatment, and we plan to publish the results.
The lessons learned can be extended to non-VHA care settings to help achieve potential benefits for other patients with early AD. Emerging therapies have the potential to improve the quality of life for both patients and care partners, adding life to years and not just years to life. Policymakers and payors must prioritize research funding to evaluate the safety and efficacy of these approaches to the delivery of health services, ensuring that emerging therapies are accessible for all individuals affected by AD.
- Alzheimer’s Association. 2025 Alzheimer’s disease facts and figures. Alzheimers Dement. 2025;21(4):e70235. doi:10.1002/alz.70235
- US Department of Veterans Affairs. Statistical Projections of Alzheimer’s Dementia for VA Patients, VA Enrollees, and US Veterans. December 18, 2020. Accessed November 2, 2025. https://www.va.gov/GERIATRICS/docs/VHA_ALZHEIMERS_DEMENTIA_Statistical_Projections_FY21_and_FY33_sgc121820.pdf
- Casey DA, Antimisiaris D, O’Brien J. Drugs for Alzheimer’s disease: are they effective? P T. 2010;35(4):208-211.
- Barthold D, Joyce G, Ferido P, et al. Pharmaceutical treatment for Alzheimer’s disease and related dementias: utilization and disparities. J Alzheimers Dis. 2020;76(2):579-589. doi:10.3233/JAD-200133
- Sims JR, Zimmer JA, Evans CD, et al. Donanemab in early symptomatic Alzheimer disease: the TRAILBLAZER-ALZ 2 randomized clinical trial. JAMA. 2023;330(6):512-527. doi:10.1001/jama.2023.13239
- van Dyck CH, Swanson CJ, Aisen P, et al. Lecanemab in early Alzheimer’s disease. N Engl J Med. 2023;388(1):9-21. doi:10.1056/NEJMoa2212948
- Tanne JH. Lecanemab: US Veterans Health Administration will cover cost of new Alzheimer’s drug. BMJ. 2023;380:p628. doi:10.1136/bmj.p628
- Nadeau SE. Lecanemab questions. Neurology. 2024;102(7):e209320. doi:10.1212/WNL.0000000000209320 9. O’Donnell AJ, Fortunato AT, Spitznogle BL, et al. Implementation of lecanemab for Alzheimer’s disease: facilitators and barriers. Presented at: American Geriatrics Society 2025 Annual Scientific Meeting, Chicago. May 2025.
- O’Donnell AJ, Zhao X, Parr A, et al. Use of lecanemab for Alzheimer’s disease within the Veteran’s Health Foundation: early findings. Abstract presented at: Alzheimer’s Association International Conference 2025; July 27, 2025; Toronto, Canada.
- O’Donnell AJ, Zhao X, Parr A, et al. Use of lecanemab for Alzheimer’s disease within the Veteran’s Health Foundation: early findings. Abstract presented at: Alzheimer’s Association International Conference 2025; July 27, 2025; Toronto, Canada.
- Hopp F, Whitten P, Subramanian U, et al. Perspectives from the Veterans Health Administration about opportunities and barriers in telemedicine. J Telemed Telecare. 2006;12(8):404-409. doi:10.1258/135763306779378717
- VA reports significant increase in veteran use of telehealth services. News release. US Department of Veterans Affairs. November 22, 2019. Accessed November 19, 2025. https://news.va.gov/press-room/va-reports-significant-increase-in-veteran-use-of-telehealth-services/
- Powers BB, Homer MC, Morone N, et al. Creation of an interprofessional teledementia clinic for rural veterans: preliminary data. J Am Geriatr Soc. 2017;65(5):1092-1099. doi:10.1111/jgs.14839
- Erickson CM, Chin NA, Rosario HL, et al. Feasibility of virtual Alzheimer’s biomarker disclosure: findings from an observational cohort. Alzheimers Dement (N Y). 2023;9(3):e12413. doi:10.1002/trc2.12413
- Turk KW, Knobel MD, Nothern A, et al. An interprofessional team for disease-modifying therapy in Alzheimer disease implementation. Neurol Clin Pract. 2024;14(6):e200346. doi:10.1212/CPJ.0000000000200346
- FDA accepts LEQEMBI® (lecanemab-irmb) biologics license application for subcutaneous maintenance dosing for the treatment of early Alzheimer’s disease. News release. Elsai US. January 13, 2025. Accessed November 2, 2025. https://media-us.eisai.com/2025-01-13-FDA-Accepts-LEQEMBI-R-lecanemab-irmb-Biologics-License-Application-for-Subcutaneous-Maintenance-Dosing-for-the-Treatment-of-Early-Alzheimers-Disease
The Veterans Health Administration (VHA) is the largest US integrated health care system, providing health care to > 9 million veterans annually. Dementia affects > 7.2 million Americans, and an estimated 450,000 veterans live with Alzheimer disease (AD).1,2 Compared with the general population, veterans have a higher burden of chronic medical conditions and are disproportionately affected by AD due to exposure to military-related risk factors (eg, traumatic brain injury and posttraumatic stress disorder) and the high prevalence of nonmilitary risk factors, such as cardiovascular disease. The VHA is a pioneer in dementia care, having established a Dementia System of Care to provide primary and specialty care to veterans with dementia. The VHA also is leading the way in implementing the Institute for Healthcare Improvement Age-Friendly Health Systems (AFHS) framework for providing goal-concordant care in > 100 VHA medical centers. The VHA aims to be the largest AFHS in the country.
AD profoundly affects individuals and their families. The progressive nature of the most common form of dementia diminishes the quality of life for patients as well as their care partners in an ongoing fashion, often leading to emotional, physical, and financial strain. Costs for health and long-term care for people living with AD and other dementias were projected at $360 billion in 2024, largely due to the need for nursing home care.1 Although several oral medications are available, their capacity to effectively mitigate the negative effects of AD is limited. Cholinesterase inhibitors and memantine may offer temporary symptomatic relief, but they do not alter disease progression.3 The use of these agents is relatively low, with about one-third of patients diagnosed with AD receiving these medications.4
Amyloid-Targeting Therapies
Recent advancements in biologics, particularly amyloid-targeting therapies, such as lecanemab and donanemab, offer new hope for managing AD. Older adults treated with these medications show less decline on measures of cognition and function than those receiving a placebo at 18 months.5,6 However, accessing and using these medications is challenging.
Use of amyloid-targeting therapies poses challenges. The medications are expensive, potentially placing a financial burden on patients, families, and health care systems.7 Determining initial eligibility for treatment requires a battery of cognitive assessments, laboratory tests, advanced radiologic studies (eg, magnetic resonance imaging [MRI] of the brain and amyloid positron emission tomography [PET] scans), and possible cerebrospinal fluid (CSF) testing. Frequent ongoing assessments are necessary to monitor safety and efficacy. These treatments carry substantial risks, particularly amyloid-related imaging abnormalities (ARIA) such as cerebral edema, microhemorrhages, and superficial siderosis. Therefore, follow-up assessments typically occur around months 2, 3, 4, and 7, depending on which medication is selected. Finally, at present, both agents must be intravenous (IV)-administered in a monitored clinical setting, which requires additional coordination, transportation, and cost.
Ongoing evaluations and in-person administration particularly affect patients and care partners with limitations regarding transportation, time off work, and navigating complex health care systems.8 VHA clinicians at sites that have implemented or are interested in implementing amyloid-targeting therapy programs endorse similar challenges when implementing these therapies in their US Department of Veterans Affairs (VA) medical centers (VAMCs).9
The VHA was one of the first health care systems to use amyloid-targeting therapies, covering the cost of lecanemab and donanemab, in addition to costs associated with concomitant evaluation and testing. However, given the safety concerns with this novel class of medications, the VHA National Formulary Committee developed criteria for use and recommended the VA Center for Medication Safety (VAMedSAFE) conduct a mandatory real-time medication use evaluation (MUE). VAMedSAFE developed the MUE to monitor the safe and appropriate use of amyloid-targeting therapy for AD. Two authors (AJO, SMH) partnered with VAMedSAFE through the VA Pittsburgh Healthcare System Technology Enhancing Cognition and Health–Geriatric Research, Education, and Clinical Center (TECH-GRECC) to provide clinical expertise, substantive feedback for the development of the MUE, and guidance for VHA sites starting amyloid targeting-therapy programs. We started a VHA Amyloid-Targeting Therapy for AD SharePoint collaborative platform and VHA AD Therapeutics Community of Practice (CoP) for shared learning (Figure). The private SharePoint platform houses an array of implementation materials for VAMCs starting programs: key documents and links; educational materials; sample guidelines; note templates; and electronic health record screenshots. The CoP allows VHAs to share best practices and discuss challenges.

Even with these advantages, we found that ensuring the safe and appropriate use of amyloid-targeting therapies did not overcome the barriers associated with their complexity. This was especially true for veterans living in rural areas. Only 4 VAMCs had administered amyloid-targeting therapies in the first year they were available. Preliminary data demonstrated that 27 (84%) of 32 veterans who initiated lecanemab in the VHA between October 2023 and September 2024 resided in urban areas.10 To address the underutilization of amyloid-targeting therapy, we propose leveraging the strengths of VHA telehealth to facilitate expansion of access to these medications for veterans with early AD. Telehealth may substantially increase access to evaluation for veterans with early dementia and, when medically appropriate, to receive amyloid-targeting therapies by reducing transportation needs and mitigating costs while ensuring appropriate monitoring through ongoing clinical assessments.
Using Telehealth
The VHA is a pioneer in telehealth, with programs dating back to 2003.11 Between October 1, 2018, and September 30, 2019, the VHA served > 900,000 veterans through the provision of > 2.6 million episodes of care via telehealth.12 The COVID-19 pandemic further cemented the role of telemedicine as an essential component of health care. Telehealth has demonstrated success in the assessment and management of individuals living with dementia. At the VHA, the GRECC-Connect Project is a partnership between 9 urban GRECC sites that seek to provide consultative geriatric and dementia care to rural veterans through telehealth.13 Additional evidence supports the potential to leverage telehealth to effectively communicate results of amyloid PET scans.14
This approach is not without limitations such as the digital divide, or the gap that separates technology-enabled individuals and those unprepared to adopt technology due to limited digital literacy levels or access to needed hardware, software, and connectivity. The VHA has taken steps to address these digital divide barriers by broadly providing tools—such as tablets and broadband connectivity—to veterans. Specifically, the VHA has instituted digital divide consults to determine whether telehealth could be a potential solution for appropriate veterans and to provide an iPad (if eligible) to connect with VA clinicians. Complementary to the digital divide consult, a VHA-specific telehealth preparedness assessment tool is under development and being tested by 2 authors (JF, SMH). This telehealth preparedness assessment tool is designed to aid in the seamless integration of telehealth services with the support of tailored education materials specific to gaps in digital literacy that a veteran might experience.
Building on these initiatives, there is an opportunity to expand access to amyloid-targeting therapies, regardless of distance to large VAMCs, by leveraging telehealth as an alternative method of connecting patients with specialty care. Specifically, a hybrid approach could be used to accomplish the myriad initial and follow-up tasks involved in the provision of amyloid-targeting therapies (Table). Not all VHA facilities possess the specialty expertise to prescribe these medications, and local clinicians may not have sufficient knowledge and clinical support to prescribe and monitor these therapies.

The first step is identifying local and regional subject matter experts, followed by the development and expansion of these networks. The National TeleNeurology Program is a good example of a national telehealth program that leverages technology to bring specialty services to rural areas with limited access to care. Although amyloid-targeting therapies often require more complex logistics, such as laboratory tests and imaging, these initial hurdles can be overcome through localized services and collaboration between VAMCs.
While treatment and imaging will most likely need to occur at a VAMC, most basic laboratory studies can be performed at community-based outpatient clinics (CBOCs). Some CBOCs may not be able to process more specialized laboratory tests such as apolipoprotein E genetic testing. Samples for these tests can be collected and processed at VAMCs, which usually have contracts with outside laboratories capable of performing these studies. Most, although not all, VAMCs offer advanced imaging, including MRI of the brain and amyloid PETs. VAMCs without those modalities may need to coordinate with other regional VAMCs. Additionally, a pilot program is already underway whereby VAMCs without the ability to quantify the amount of amyloid on PETs are able to leverage technology and collaborations with other VAMCs to obtain these data.
Once the initial phases of evaluation and care are completed, telemedicine can be leveraged for follow-up and ongoing management. Interdisciplinary teams can help facilitate care related to amyloid-targeting therapies, including the close monitoring of veterans for development of ARIA.15 To achieve this monitoring, specialty clinic teams prescribing amyloid-targeting therapies, which may be geographically distant, need to coordinate with local primary care clinical teams and emergency clinicians. All of these health care team members, along with neurologists and neurosurgeons, should be involved in the development and implementation of protocols in the event that patients present to their local primary or specialty care clinics or emergency department with ARIA symptoms.
If amyloid-targeting therapies are to be provided along with other emerging treatments for rural veterans, telehealth must be part of the solution. There is a pressing need to explore innovative evaluation and delivery models for these therapies, particularly as we expect additional diagnostics and therapeutics to be available in the future. With the advent of commercially available blood tests (ie, blood biomarkers) for AD, there is hope for a transition away from PETs and CSF testing given their cost, limited access, and invasiveness for diagnosis and monitoring of AD. These advances will increase the utility of telehealth to help rural veterans access amyloid-targeting therapies.
Additionally, administering the drug at home or at local clinics, supported by a dedicated health care team or home health agency, could further improve accessibility. Telehealth can be leveraged in this scenario, allowing specialty clinics and specialists to connect with patients and clinicians based out of local clinics or even home health agencies. In this scenario, specialists can provide hands-on care guidance and oversight even though they may be geographically distant from care recipients. Transitioning from IV administration to subcutaneous formulations would further enhance convenience and reduce barriers; these formulations may be available soon.16 Addressing logistical challenges to care and access through technology-based solutions will require coordinated efforts and continued VHA investment.
Conclusions
The VHA has a large population of veterans with dementia, and the costs to care for these veterans will only increase. While the current benefits of amyloid-targeting therapies are modest, now is the time to establish care processes that will support future innovations in amyloid-targeting therapies and other treatments and diagnostics. We are developing better ways to detect AD using clinical decision support tools, improving care pathways and the management of AD, and leveraging telehealth to improve access. The VA is conducting research to investigate whether a cognitive screening and laboratory evaluation that includes a telehealth preparedness assessment will be feasible and effective for improving the detection of AD and access to treatment, and we plan to publish the results.
The lessons learned can be extended to non-VHA care settings to help achieve potential benefits for other patients with early AD. Emerging therapies have the potential to improve the quality of life for both patients and care partners, adding life to years and not just years to life. Policymakers and payors must prioritize research funding to evaluate the safety and efficacy of these approaches to the delivery of health services, ensuring that emerging therapies are accessible for all individuals affected by AD.
The Veterans Health Administration (VHA) is the largest US integrated health care system, providing health care to > 9 million veterans annually. Dementia affects > 7.2 million Americans, and an estimated 450,000 veterans live with Alzheimer disease (AD).1,2 Compared with the general population, veterans have a higher burden of chronic medical conditions and are disproportionately affected by AD due to exposure to military-related risk factors (eg, traumatic brain injury and posttraumatic stress disorder) and the high prevalence of nonmilitary risk factors, such as cardiovascular disease. The VHA is a pioneer in dementia care, having established a Dementia System of Care to provide primary and specialty care to veterans with dementia. The VHA also is leading the way in implementing the Institute for Healthcare Improvement Age-Friendly Health Systems (AFHS) framework for providing goal-concordant care in > 100 VHA medical centers. The VHA aims to be the largest AFHS in the country.
AD profoundly affects individuals and their families. The progressive nature of the most common form of dementia diminishes the quality of life for patients as well as their care partners in an ongoing fashion, often leading to emotional, physical, and financial strain. Costs for health and long-term care for people living with AD and other dementias were projected at $360 billion in 2024, largely due to the need for nursing home care.1 Although several oral medications are available, their capacity to effectively mitigate the negative effects of AD is limited. Cholinesterase inhibitors and memantine may offer temporary symptomatic relief, but they do not alter disease progression.3 The use of these agents is relatively low, with about one-third of patients diagnosed with AD receiving these medications.4
Amyloid-Targeting Therapies
Recent advancements in biologics, particularly amyloid-targeting therapies, such as lecanemab and donanemab, offer new hope for managing AD. Older adults treated with these medications show less decline on measures of cognition and function than those receiving a placebo at 18 months.5,6 However, accessing and using these medications is challenging.
Use of amyloid-targeting therapies poses challenges. The medications are expensive, potentially placing a financial burden on patients, families, and health care systems.7 Determining initial eligibility for treatment requires a battery of cognitive assessments, laboratory tests, advanced radiologic studies (eg, magnetic resonance imaging [MRI] of the brain and amyloid positron emission tomography [PET] scans), and possible cerebrospinal fluid (CSF) testing. Frequent ongoing assessments are necessary to monitor safety and efficacy. These treatments carry substantial risks, particularly amyloid-related imaging abnormalities (ARIA) such as cerebral edema, microhemorrhages, and superficial siderosis. Therefore, follow-up assessments typically occur around months 2, 3, 4, and 7, depending on which medication is selected. Finally, at present, both agents must be intravenous (IV)-administered in a monitored clinical setting, which requires additional coordination, transportation, and cost.
Ongoing evaluations and in-person administration particularly affect patients and care partners with limitations regarding transportation, time off work, and navigating complex health care systems.8 VHA clinicians at sites that have implemented or are interested in implementing amyloid-targeting therapy programs endorse similar challenges when implementing these therapies in their US Department of Veterans Affairs (VA) medical centers (VAMCs).9
The VHA was one of the first health care systems to use amyloid-targeting therapies, covering the cost of lecanemab and donanemab, in addition to costs associated with concomitant evaluation and testing. However, given the safety concerns with this novel class of medications, the VHA National Formulary Committee developed criteria for use and recommended the VA Center for Medication Safety (VAMedSAFE) conduct a mandatory real-time medication use evaluation (MUE). VAMedSAFE developed the MUE to monitor the safe and appropriate use of amyloid-targeting therapy for AD. Two authors (AJO, SMH) partnered with VAMedSAFE through the VA Pittsburgh Healthcare System Technology Enhancing Cognition and Health–Geriatric Research, Education, and Clinical Center (TECH-GRECC) to provide clinical expertise, substantive feedback for the development of the MUE, and guidance for VHA sites starting amyloid targeting-therapy programs. We started a VHA Amyloid-Targeting Therapy for AD SharePoint collaborative platform and VHA AD Therapeutics Community of Practice (CoP) for shared learning (Figure). The private SharePoint platform houses an array of implementation materials for VAMCs starting programs: key documents and links; educational materials; sample guidelines; note templates; and electronic health record screenshots. The CoP allows VHAs to share best practices and discuss challenges.

Even with these advantages, we found that ensuring the safe and appropriate use of amyloid-targeting therapies did not overcome the barriers associated with their complexity. This was especially true for veterans living in rural areas. Only 4 VAMCs had administered amyloid-targeting therapies in the first year they were available. Preliminary data demonstrated that 27 (84%) of 32 veterans who initiated lecanemab in the VHA between October 2023 and September 2024 resided in urban areas.10 To address the underutilization of amyloid-targeting therapy, we propose leveraging the strengths of VHA telehealth to facilitate expansion of access to these medications for veterans with early AD. Telehealth may substantially increase access to evaluation for veterans with early dementia and, when medically appropriate, to receive amyloid-targeting therapies by reducing transportation needs and mitigating costs while ensuring appropriate monitoring through ongoing clinical assessments.
Using Telehealth
The VHA is a pioneer in telehealth, with programs dating back to 2003.11 Between October 1, 2018, and September 30, 2019, the VHA served > 900,000 veterans through the provision of > 2.6 million episodes of care via telehealth.12 The COVID-19 pandemic further cemented the role of telemedicine as an essential component of health care. Telehealth has demonstrated success in the assessment and management of individuals living with dementia. At the VHA, the GRECC-Connect Project is a partnership between 9 urban GRECC sites that seek to provide consultative geriatric and dementia care to rural veterans through telehealth.13 Additional evidence supports the potential to leverage telehealth to effectively communicate results of amyloid PET scans.14
This approach is not without limitations such as the digital divide, or the gap that separates technology-enabled individuals and those unprepared to adopt technology due to limited digital literacy levels or access to needed hardware, software, and connectivity. The VHA has taken steps to address these digital divide barriers by broadly providing tools—such as tablets and broadband connectivity—to veterans. Specifically, the VHA has instituted digital divide consults to determine whether telehealth could be a potential solution for appropriate veterans and to provide an iPad (if eligible) to connect with VA clinicians. Complementary to the digital divide consult, a VHA-specific telehealth preparedness assessment tool is under development and being tested by 2 authors (JF, SMH). This telehealth preparedness assessment tool is designed to aid in the seamless integration of telehealth services with the support of tailored education materials specific to gaps in digital literacy that a veteran might experience.
Building on these initiatives, there is an opportunity to expand access to amyloid-targeting therapies, regardless of distance to large VAMCs, by leveraging telehealth as an alternative method of connecting patients with specialty care. Specifically, a hybrid approach could be used to accomplish the myriad initial and follow-up tasks involved in the provision of amyloid-targeting therapies (Table). Not all VHA facilities possess the specialty expertise to prescribe these medications, and local clinicians may not have sufficient knowledge and clinical support to prescribe and monitor these therapies.

The first step is identifying local and regional subject matter experts, followed by the development and expansion of these networks. The National TeleNeurology Program is a good example of a national telehealth program that leverages technology to bring specialty services to rural areas with limited access to care. Although amyloid-targeting therapies often require more complex logistics, such as laboratory tests and imaging, these initial hurdles can be overcome through localized services and collaboration between VAMCs.
While treatment and imaging will most likely need to occur at a VAMC, most basic laboratory studies can be performed at community-based outpatient clinics (CBOCs). Some CBOCs may not be able to process more specialized laboratory tests such as apolipoprotein E genetic testing. Samples for these tests can be collected and processed at VAMCs, which usually have contracts with outside laboratories capable of performing these studies. Most, although not all, VAMCs offer advanced imaging, including MRI of the brain and amyloid PETs. VAMCs without those modalities may need to coordinate with other regional VAMCs. Additionally, a pilot program is already underway whereby VAMCs without the ability to quantify the amount of amyloid on PETs are able to leverage technology and collaborations with other VAMCs to obtain these data.
Once the initial phases of evaluation and care are completed, telemedicine can be leveraged for follow-up and ongoing management. Interdisciplinary teams can help facilitate care related to amyloid-targeting therapies, including the close monitoring of veterans for development of ARIA.15 To achieve this monitoring, specialty clinic teams prescribing amyloid-targeting therapies, which may be geographically distant, need to coordinate with local primary care clinical teams and emergency clinicians. All of these health care team members, along with neurologists and neurosurgeons, should be involved in the development and implementation of protocols in the event that patients present to their local primary or specialty care clinics or emergency department with ARIA symptoms.
If amyloid-targeting therapies are to be provided along with other emerging treatments for rural veterans, telehealth must be part of the solution. There is a pressing need to explore innovative evaluation and delivery models for these therapies, particularly as we expect additional diagnostics and therapeutics to be available in the future. With the advent of commercially available blood tests (ie, blood biomarkers) for AD, there is hope for a transition away from PETs and CSF testing given their cost, limited access, and invasiveness for diagnosis and monitoring of AD. These advances will increase the utility of telehealth to help rural veterans access amyloid-targeting therapies.
Additionally, administering the drug at home or at local clinics, supported by a dedicated health care team or home health agency, could further improve accessibility. Telehealth can be leveraged in this scenario, allowing specialty clinics and specialists to connect with patients and clinicians based out of local clinics or even home health agencies. In this scenario, specialists can provide hands-on care guidance and oversight even though they may be geographically distant from care recipients. Transitioning from IV administration to subcutaneous formulations would further enhance convenience and reduce barriers; these formulations may be available soon.16 Addressing logistical challenges to care and access through technology-based solutions will require coordinated efforts and continued VHA investment.
Conclusions
The VHA has a large population of veterans with dementia, and the costs to care for these veterans will only increase. While the current benefits of amyloid-targeting therapies are modest, now is the time to establish care processes that will support future innovations in amyloid-targeting therapies and other treatments and diagnostics. We are developing better ways to detect AD using clinical decision support tools, improving care pathways and the management of AD, and leveraging telehealth to improve access. The VA is conducting research to investigate whether a cognitive screening and laboratory evaluation that includes a telehealth preparedness assessment will be feasible and effective for improving the detection of AD and access to treatment, and we plan to publish the results.
The lessons learned can be extended to non-VHA care settings to help achieve potential benefits for other patients with early AD. Emerging therapies have the potential to improve the quality of life for both patients and care partners, adding life to years and not just years to life. Policymakers and payors must prioritize research funding to evaluate the safety and efficacy of these approaches to the delivery of health services, ensuring that emerging therapies are accessible for all individuals affected by AD.
- Alzheimer’s Association. 2025 Alzheimer’s disease facts and figures. Alzheimers Dement. 2025;21(4):e70235. doi:10.1002/alz.70235
- US Department of Veterans Affairs. Statistical Projections of Alzheimer’s Dementia for VA Patients, VA Enrollees, and US Veterans. December 18, 2020. Accessed November 2, 2025. https://www.va.gov/GERIATRICS/docs/VHA_ALZHEIMERS_DEMENTIA_Statistical_Projections_FY21_and_FY33_sgc121820.pdf
- Casey DA, Antimisiaris D, O’Brien J. Drugs for Alzheimer’s disease: are they effective? P T. 2010;35(4):208-211.
- Barthold D, Joyce G, Ferido P, et al. Pharmaceutical treatment for Alzheimer’s disease and related dementias: utilization and disparities. J Alzheimers Dis. 2020;76(2):579-589. doi:10.3233/JAD-200133
- Sims JR, Zimmer JA, Evans CD, et al. Donanemab in early symptomatic Alzheimer disease: the TRAILBLAZER-ALZ 2 randomized clinical trial. JAMA. 2023;330(6):512-527. doi:10.1001/jama.2023.13239
- van Dyck CH, Swanson CJ, Aisen P, et al. Lecanemab in early Alzheimer’s disease. N Engl J Med. 2023;388(1):9-21. doi:10.1056/NEJMoa2212948
- Tanne JH. Lecanemab: US Veterans Health Administration will cover cost of new Alzheimer’s drug. BMJ. 2023;380:p628. doi:10.1136/bmj.p628
- Nadeau SE. Lecanemab questions. Neurology. 2024;102(7):e209320. doi:10.1212/WNL.0000000000209320 9. O’Donnell AJ, Fortunato AT, Spitznogle BL, et al. Implementation of lecanemab for Alzheimer’s disease: facilitators and barriers. Presented at: American Geriatrics Society 2025 Annual Scientific Meeting, Chicago. May 2025.
- O’Donnell AJ, Zhao X, Parr A, et al. Use of lecanemab for Alzheimer’s disease within the Veteran’s Health Foundation: early findings. Abstract presented at: Alzheimer’s Association International Conference 2025; July 27, 2025; Toronto, Canada.
- O’Donnell AJ, Zhao X, Parr A, et al. Use of lecanemab for Alzheimer’s disease within the Veteran’s Health Foundation: early findings. Abstract presented at: Alzheimer’s Association International Conference 2025; July 27, 2025; Toronto, Canada.
- Hopp F, Whitten P, Subramanian U, et al. Perspectives from the Veterans Health Administration about opportunities and barriers in telemedicine. J Telemed Telecare. 2006;12(8):404-409. doi:10.1258/135763306779378717
- VA reports significant increase in veteran use of telehealth services. News release. US Department of Veterans Affairs. November 22, 2019. Accessed November 19, 2025. https://news.va.gov/press-room/va-reports-significant-increase-in-veteran-use-of-telehealth-services/
- Powers BB, Homer MC, Morone N, et al. Creation of an interprofessional teledementia clinic for rural veterans: preliminary data. J Am Geriatr Soc. 2017;65(5):1092-1099. doi:10.1111/jgs.14839
- Erickson CM, Chin NA, Rosario HL, et al. Feasibility of virtual Alzheimer’s biomarker disclosure: findings from an observational cohort. Alzheimers Dement (N Y). 2023;9(3):e12413. doi:10.1002/trc2.12413
- Turk KW, Knobel MD, Nothern A, et al. An interprofessional team for disease-modifying therapy in Alzheimer disease implementation. Neurol Clin Pract. 2024;14(6):e200346. doi:10.1212/CPJ.0000000000200346
- FDA accepts LEQEMBI® (lecanemab-irmb) biologics license application for subcutaneous maintenance dosing for the treatment of early Alzheimer’s disease. News release. Elsai US. January 13, 2025. Accessed November 2, 2025. https://media-us.eisai.com/2025-01-13-FDA-Accepts-LEQEMBI-R-lecanemab-irmb-Biologics-License-Application-for-Subcutaneous-Maintenance-Dosing-for-the-Treatment-of-Early-Alzheimers-Disease
- Alzheimer’s Association. 2025 Alzheimer’s disease facts and figures. Alzheimers Dement. 2025;21(4):e70235. doi:10.1002/alz.70235
- US Department of Veterans Affairs. Statistical Projections of Alzheimer’s Dementia for VA Patients, VA Enrollees, and US Veterans. December 18, 2020. Accessed November 2, 2025. https://www.va.gov/GERIATRICS/docs/VHA_ALZHEIMERS_DEMENTIA_Statistical_Projections_FY21_and_FY33_sgc121820.pdf
- Casey DA, Antimisiaris D, O’Brien J. Drugs for Alzheimer’s disease: are they effective? P T. 2010;35(4):208-211.
- Barthold D, Joyce G, Ferido P, et al. Pharmaceutical treatment for Alzheimer’s disease and related dementias: utilization and disparities. J Alzheimers Dis. 2020;76(2):579-589. doi:10.3233/JAD-200133
- Sims JR, Zimmer JA, Evans CD, et al. Donanemab in early symptomatic Alzheimer disease: the TRAILBLAZER-ALZ 2 randomized clinical trial. JAMA. 2023;330(6):512-527. doi:10.1001/jama.2023.13239
- van Dyck CH, Swanson CJ, Aisen P, et al. Lecanemab in early Alzheimer’s disease. N Engl J Med. 2023;388(1):9-21. doi:10.1056/NEJMoa2212948
- Tanne JH. Lecanemab: US Veterans Health Administration will cover cost of new Alzheimer’s drug. BMJ. 2023;380:p628. doi:10.1136/bmj.p628
- Nadeau SE. Lecanemab questions. Neurology. 2024;102(7):e209320. doi:10.1212/WNL.0000000000209320 9. O’Donnell AJ, Fortunato AT, Spitznogle BL, et al. Implementation of lecanemab for Alzheimer’s disease: facilitators and barriers. Presented at: American Geriatrics Society 2025 Annual Scientific Meeting, Chicago. May 2025.
- O’Donnell AJ, Zhao X, Parr A, et al. Use of lecanemab for Alzheimer’s disease within the Veteran’s Health Foundation: early findings. Abstract presented at: Alzheimer’s Association International Conference 2025; July 27, 2025; Toronto, Canada.
- O’Donnell AJ, Zhao X, Parr A, et al. Use of lecanemab for Alzheimer’s disease within the Veteran’s Health Foundation: early findings. Abstract presented at: Alzheimer’s Association International Conference 2025; July 27, 2025; Toronto, Canada.
- Hopp F, Whitten P, Subramanian U, et al. Perspectives from the Veterans Health Administration about opportunities and barriers in telemedicine. J Telemed Telecare. 2006;12(8):404-409. doi:10.1258/135763306779378717
- VA reports significant increase in veteran use of telehealth services. News release. US Department of Veterans Affairs. November 22, 2019. Accessed November 19, 2025. https://news.va.gov/press-room/va-reports-significant-increase-in-veteran-use-of-telehealth-services/
- Powers BB, Homer MC, Morone N, et al. Creation of an interprofessional teledementia clinic for rural veterans: preliminary data. J Am Geriatr Soc. 2017;65(5):1092-1099. doi:10.1111/jgs.14839
- Erickson CM, Chin NA, Rosario HL, et al. Feasibility of virtual Alzheimer’s biomarker disclosure: findings from an observational cohort. Alzheimers Dement (N Y). 2023;9(3):e12413. doi:10.1002/trc2.12413
- Turk KW, Knobel MD, Nothern A, et al. An interprofessional team for disease-modifying therapy in Alzheimer disease implementation. Neurol Clin Pract. 2024;14(6):e200346. doi:10.1212/CPJ.0000000000200346
- FDA accepts LEQEMBI® (lecanemab-irmb) biologics license application for subcutaneous maintenance dosing for the treatment of early Alzheimer’s disease. News release. Elsai US. January 13, 2025. Accessed November 2, 2025. https://media-us.eisai.com/2025-01-13-FDA-Accepts-LEQEMBI-R-lecanemab-irmb-Biologics-License-Application-for-Subcutaneous-Maintenance-Dosing-for-the-Treatment-of-Early-Alzheimers-Disease
Can Telehealth Improve Access to Amyloid-Targeting Therapies for Veterans Living With Alzheimer Disease?
Can Telehealth Improve Access to Amyloid-Targeting Therapies for Veterans Living With Alzheimer Disease?
The Once and Future Veterans Health Administration
The Once and Future Veterans Health Administration
He who thus considers things in their first growth and origin ... will obtain the clearest view of them. Politics, Book I, Part II by Aristotle
Many seasoned observers of federal practice have signaled that the future of US Department of Veterans Affairs (VA) health care is threatened as never before. Political forces and economic interests are siphoning Veterans Health Administration (VHA) capital and human resources into the community with an ineluctable push toward privatization.1
This Veterans Day, the vitality, if not the very viability of veteran health care, is in serious jeopardy, so it seems fitting to review the rationale for having institutions dedicated to the specialized medical treatment of veterans. Aristotle advises us on how to undertake this intellectual exercise in the epigraph. This column will revisit the historical origins of VA medicine to better appreciate the justification of an agency committed to this unique purpose and what may be sacrificed if it is decimated.
The provision of medical care focused on the injuries and illnesses of warriors is as old as war. The ancient Romans had among the first veterans’ hospital, named a valetudinarium. Sick and injured members of the Roman legions received state-of-the-art medical and surgical care from military doctors inside these facilities.2
In the United States, federal practice emerged almost simultaneously with the birth of a nation. Wounded troops and families of slain soldiers required rehabilitation and support from the fledgling federal government. This began a pattern of development in which each war generated novel injuries and disorders that required the VA to evolve (Table).3

Many arguments can be marshalled to demonstrate the importance of not just ensuring VA health care survives but also has the resources needed to thrive. I will highlight what I argue are the most important justifications for its existence.
The ethical argument: President Abraham Lincoln and a long line of government officials for more than 2 centuries have called the provision of high-quality health care focused on veterans a sacred trust. Failing to fulfill that promise is a violation of the deepest principles of veracity and fidelity that those who govern owe to the citizens who selflessly sacrificed time, health, and even in some cases life, for the safety and well-being of their country.4
The quality argument: Dozens of studies have found that compared to the community, many areas of veteran medical care are just plain better. Two surveys particularly salient in the aging veteran population illustrate this growing body of positive research. The most recent and largest survey of Medicare patients found that VHA hospitals surpassed community-based hospitals on all 10 metrics.5 A retrospective cohort study of mortality compared veterans transported by ambulance to VHA or community-based hospitals. The researchers found that those taken to VHA facilities had a 30-day all cause adjustment mortality 20 times lower than those taken to civilian hospitals, especially among minoritized populations who generally have higher mortality.6
The cultural argument: Glance at almost any form of communication from veterans or about their health care and you will apprehend common cultural themes. Even when frustrated that the system has not lived up to their expectations, and perhaps because of their sense of belonging, they voice ownership of VHA as their medical home. Surveys of veteran experiences have shown many feel more comfortable receiving care in the company of comrades in arms and from health care professionals with expertise and experience with veterans’ distinctive medical problems and the military values that inform their preferences for care.7
The complexity argument: Anyone who has worked even a short time in a VHA hospital or clinic knows the patients are in general more complicated than similar patients in the community. Multiple medical, geriatric, neuropsychiatric, substance use, and social comorbidities are the expectation, not the exception, as in some civilian systems. Many of the conditions common in the VHA such as traumatic brain injury, service-connected cancers, suicidal ideation, environmental exposures, and posttraumatic stress disorder would be encountered in community health care settings. The differences between VHA and community care led the RAND Corporation to caution that “Community care providers might not be equipped to handle the needs of veterans.”8
Let me bring this 1000-foot view of the crisis facing federal practice down to the literal level of my own home. For many years I have had a wonderful mechanic who has a mobile bike service. I was talking to him as he fixed my trike. I never knew he was a Vietnam era veteran, and he didn’t realize that I was a career VA health care professional at the very VHA hospital where he received care. He spontaneously told me that, “when I first got out, the VA was awful, but now it is wonderful and they are so good to me. I would not go anywhere else.” For the many veterans of that era who would echo his sentiments, we must not allow the VA to lose all it has gained since that painful time
Another philosopher, Søren Kierkegaard, wrote that “life must be understood backwards but lived forwards.”9 Our own brief back to the future journey in this editorial has, I hope, shown that VHA medical institutions and health professionals cannot be replaced with or replicated by civilian systems and clinicians. Continued attempts to do so betray the trust and risks the health and well-being of veterans. It also would deprive the country of research, innovation, and education that make unparalleled contributions to public health. Ultimately, these efforts to diminish VHA compromise the solidarity of service members with each other and with their federal practitioners. If this trend to dismantle an organization that originated with the sole purpose of caring for veterans continues, then the public expressions of respect and gratitude will sound shallower and more tentative with each passing Veterans Day.
- Quil L. Hundreds of VA clinicians warn that cuts threaten vet’s health care. National Public Radio. October 1, 2025. Accessed October 27, 2025. https://www.npr.org/2025/10/01/nx-s1-5554394/hundreds-of-va-clinicians-warn-that-cuts-threaten-vets-health-care
- Nutton V. Ancient Medicine. 2nd ed. Routledge; 2012.
- US Department of Veterans Affairs. VA History Summary. Updated June 13, 2025. Accessed October 27, 2025. https://department.va.gov/history/history-overview/
- Geppert CMA. Learning from history: the ethical foundation of VA health care. Fed Pract. 2016;33:6-7.
- US Department of Veterans Affairs. Nationwide patient survey shows VA hospitals outperform non-VA hospitals. News release. June 14, 2023. Accessed October 27, 2025. https://news.va.gov/press-room/nationwide-patient-survey-shows-va-hospitals-outperform-non-va-hospitals
- Chan DC, Danesh K, Costantini S, Card D, Taylor L, Studdert DM. Mortality among US veterans after emergency visits to Veterans Affairs and other hospitals: retrospective cohort study. BMJ. 2022;376:e068099. doi:10.1136/bmj-2021-068099
- Vigilante K, Batten SV, Shang Q, et al. Camaraderie among US veterans and their preferences for health care systems and practitioners. JAMA Netw Open. 2025;8(4):e255253. doi:10.1001/jamanetworkopen.2025.5253
- Rasmussen P, Farmer CM. The promise and challenges of VA community care: veterans’ issues in focus. Rand Health Q. 2023;10:9.
- Kierkegaard S. Journalen JJ:167 (1843) in: Søren Kierkegaards Skrifter. Vol 18. Copenhagen; 1997:306.
He who thus considers things in their first growth and origin ... will obtain the clearest view of them. Politics, Book I, Part II by Aristotle
Many seasoned observers of federal practice have signaled that the future of US Department of Veterans Affairs (VA) health care is threatened as never before. Political forces and economic interests are siphoning Veterans Health Administration (VHA) capital and human resources into the community with an ineluctable push toward privatization.1
This Veterans Day, the vitality, if not the very viability of veteran health care, is in serious jeopardy, so it seems fitting to review the rationale for having institutions dedicated to the specialized medical treatment of veterans. Aristotle advises us on how to undertake this intellectual exercise in the epigraph. This column will revisit the historical origins of VA medicine to better appreciate the justification of an agency committed to this unique purpose and what may be sacrificed if it is decimated.
The provision of medical care focused on the injuries and illnesses of warriors is as old as war. The ancient Romans had among the first veterans’ hospital, named a valetudinarium. Sick and injured members of the Roman legions received state-of-the-art medical and surgical care from military doctors inside these facilities.2
In the United States, federal practice emerged almost simultaneously with the birth of a nation. Wounded troops and families of slain soldiers required rehabilitation and support from the fledgling federal government. This began a pattern of development in which each war generated novel injuries and disorders that required the VA to evolve (Table).3

Many arguments can be marshalled to demonstrate the importance of not just ensuring VA health care survives but also has the resources needed to thrive. I will highlight what I argue are the most important justifications for its existence.
The ethical argument: President Abraham Lincoln and a long line of government officials for more than 2 centuries have called the provision of high-quality health care focused on veterans a sacred trust. Failing to fulfill that promise is a violation of the deepest principles of veracity and fidelity that those who govern owe to the citizens who selflessly sacrificed time, health, and even in some cases life, for the safety and well-being of their country.4
The quality argument: Dozens of studies have found that compared to the community, many areas of veteran medical care are just plain better. Two surveys particularly salient in the aging veteran population illustrate this growing body of positive research. The most recent and largest survey of Medicare patients found that VHA hospitals surpassed community-based hospitals on all 10 metrics.5 A retrospective cohort study of mortality compared veterans transported by ambulance to VHA or community-based hospitals. The researchers found that those taken to VHA facilities had a 30-day all cause adjustment mortality 20 times lower than those taken to civilian hospitals, especially among minoritized populations who generally have higher mortality.6
The cultural argument: Glance at almost any form of communication from veterans or about their health care and you will apprehend common cultural themes. Even when frustrated that the system has not lived up to their expectations, and perhaps because of their sense of belonging, they voice ownership of VHA as their medical home. Surveys of veteran experiences have shown many feel more comfortable receiving care in the company of comrades in arms and from health care professionals with expertise and experience with veterans’ distinctive medical problems and the military values that inform their preferences for care.7
The complexity argument: Anyone who has worked even a short time in a VHA hospital or clinic knows the patients are in general more complicated than similar patients in the community. Multiple medical, geriatric, neuropsychiatric, substance use, and social comorbidities are the expectation, not the exception, as in some civilian systems. Many of the conditions common in the VHA such as traumatic brain injury, service-connected cancers, suicidal ideation, environmental exposures, and posttraumatic stress disorder would be encountered in community health care settings. The differences between VHA and community care led the RAND Corporation to caution that “Community care providers might not be equipped to handle the needs of veterans.”8
Let me bring this 1000-foot view of the crisis facing federal practice down to the literal level of my own home. For many years I have had a wonderful mechanic who has a mobile bike service. I was talking to him as he fixed my trike. I never knew he was a Vietnam era veteran, and he didn’t realize that I was a career VA health care professional at the very VHA hospital where he received care. He spontaneously told me that, “when I first got out, the VA was awful, but now it is wonderful and they are so good to me. I would not go anywhere else.” For the many veterans of that era who would echo his sentiments, we must not allow the VA to lose all it has gained since that painful time
Another philosopher, Søren Kierkegaard, wrote that “life must be understood backwards but lived forwards.”9 Our own brief back to the future journey in this editorial has, I hope, shown that VHA medical institutions and health professionals cannot be replaced with or replicated by civilian systems and clinicians. Continued attempts to do so betray the trust and risks the health and well-being of veterans. It also would deprive the country of research, innovation, and education that make unparalleled contributions to public health. Ultimately, these efforts to diminish VHA compromise the solidarity of service members with each other and with their federal practitioners. If this trend to dismantle an organization that originated with the sole purpose of caring for veterans continues, then the public expressions of respect and gratitude will sound shallower and more tentative with each passing Veterans Day.
He who thus considers things in their first growth and origin ... will obtain the clearest view of them. Politics, Book I, Part II by Aristotle
Many seasoned observers of federal practice have signaled that the future of US Department of Veterans Affairs (VA) health care is threatened as never before. Political forces and economic interests are siphoning Veterans Health Administration (VHA) capital and human resources into the community with an ineluctable push toward privatization.1
This Veterans Day, the vitality, if not the very viability of veteran health care, is in serious jeopardy, so it seems fitting to review the rationale for having institutions dedicated to the specialized medical treatment of veterans. Aristotle advises us on how to undertake this intellectual exercise in the epigraph. This column will revisit the historical origins of VA medicine to better appreciate the justification of an agency committed to this unique purpose and what may be sacrificed if it is decimated.
The provision of medical care focused on the injuries and illnesses of warriors is as old as war. The ancient Romans had among the first veterans’ hospital, named a valetudinarium. Sick and injured members of the Roman legions received state-of-the-art medical and surgical care from military doctors inside these facilities.2
In the United States, federal practice emerged almost simultaneously with the birth of a nation. Wounded troops and families of slain soldiers required rehabilitation and support from the fledgling federal government. This began a pattern of development in which each war generated novel injuries and disorders that required the VA to evolve (Table).3

Many arguments can be marshalled to demonstrate the importance of not just ensuring VA health care survives but also has the resources needed to thrive. I will highlight what I argue are the most important justifications for its existence.
The ethical argument: President Abraham Lincoln and a long line of government officials for more than 2 centuries have called the provision of high-quality health care focused on veterans a sacred trust. Failing to fulfill that promise is a violation of the deepest principles of veracity and fidelity that those who govern owe to the citizens who selflessly sacrificed time, health, and even in some cases life, for the safety and well-being of their country.4
The quality argument: Dozens of studies have found that compared to the community, many areas of veteran medical care are just plain better. Two surveys particularly salient in the aging veteran population illustrate this growing body of positive research. The most recent and largest survey of Medicare patients found that VHA hospitals surpassed community-based hospitals on all 10 metrics.5 A retrospective cohort study of mortality compared veterans transported by ambulance to VHA or community-based hospitals. The researchers found that those taken to VHA facilities had a 30-day all cause adjustment mortality 20 times lower than those taken to civilian hospitals, especially among minoritized populations who generally have higher mortality.6
The cultural argument: Glance at almost any form of communication from veterans or about their health care and you will apprehend common cultural themes. Even when frustrated that the system has not lived up to their expectations, and perhaps because of their sense of belonging, they voice ownership of VHA as their medical home. Surveys of veteran experiences have shown many feel more comfortable receiving care in the company of comrades in arms and from health care professionals with expertise and experience with veterans’ distinctive medical problems and the military values that inform their preferences for care.7
The complexity argument: Anyone who has worked even a short time in a VHA hospital or clinic knows the patients are in general more complicated than similar patients in the community. Multiple medical, geriatric, neuropsychiatric, substance use, and social comorbidities are the expectation, not the exception, as in some civilian systems. Many of the conditions common in the VHA such as traumatic brain injury, service-connected cancers, suicidal ideation, environmental exposures, and posttraumatic stress disorder would be encountered in community health care settings. The differences between VHA and community care led the RAND Corporation to caution that “Community care providers might not be equipped to handle the needs of veterans.”8
Let me bring this 1000-foot view of the crisis facing federal practice down to the literal level of my own home. For many years I have had a wonderful mechanic who has a mobile bike service. I was talking to him as he fixed my trike. I never knew he was a Vietnam era veteran, and he didn’t realize that I was a career VA health care professional at the very VHA hospital where he received care. He spontaneously told me that, “when I first got out, the VA was awful, but now it is wonderful and they are so good to me. I would not go anywhere else.” For the many veterans of that era who would echo his sentiments, we must not allow the VA to lose all it has gained since that painful time
Another philosopher, Søren Kierkegaard, wrote that “life must be understood backwards but lived forwards.”9 Our own brief back to the future journey in this editorial has, I hope, shown that VHA medical institutions and health professionals cannot be replaced with or replicated by civilian systems and clinicians. Continued attempts to do so betray the trust and risks the health and well-being of veterans. It also would deprive the country of research, innovation, and education that make unparalleled contributions to public health. Ultimately, these efforts to diminish VHA compromise the solidarity of service members with each other and with their federal practitioners. If this trend to dismantle an organization that originated with the sole purpose of caring for veterans continues, then the public expressions of respect and gratitude will sound shallower and more tentative with each passing Veterans Day.
- Quil L. Hundreds of VA clinicians warn that cuts threaten vet’s health care. National Public Radio. October 1, 2025. Accessed October 27, 2025. https://www.npr.org/2025/10/01/nx-s1-5554394/hundreds-of-va-clinicians-warn-that-cuts-threaten-vets-health-care
- Nutton V. Ancient Medicine. 2nd ed. Routledge; 2012.
- US Department of Veterans Affairs. VA History Summary. Updated June 13, 2025. Accessed October 27, 2025. https://department.va.gov/history/history-overview/
- Geppert CMA. Learning from history: the ethical foundation of VA health care. Fed Pract. 2016;33:6-7.
- US Department of Veterans Affairs. Nationwide patient survey shows VA hospitals outperform non-VA hospitals. News release. June 14, 2023. Accessed October 27, 2025. https://news.va.gov/press-room/nationwide-patient-survey-shows-va-hospitals-outperform-non-va-hospitals
- Chan DC, Danesh K, Costantini S, Card D, Taylor L, Studdert DM. Mortality among US veterans after emergency visits to Veterans Affairs and other hospitals: retrospective cohort study. BMJ. 2022;376:e068099. doi:10.1136/bmj-2021-068099
- Vigilante K, Batten SV, Shang Q, et al. Camaraderie among US veterans and their preferences for health care systems and practitioners. JAMA Netw Open. 2025;8(4):e255253. doi:10.1001/jamanetworkopen.2025.5253
- Rasmussen P, Farmer CM. The promise and challenges of VA community care: veterans’ issues in focus. Rand Health Q. 2023;10:9.
- Kierkegaard S. Journalen JJ:167 (1843) in: Søren Kierkegaards Skrifter. Vol 18. Copenhagen; 1997:306.
- Quil L. Hundreds of VA clinicians warn that cuts threaten vet’s health care. National Public Radio. October 1, 2025. Accessed October 27, 2025. https://www.npr.org/2025/10/01/nx-s1-5554394/hundreds-of-va-clinicians-warn-that-cuts-threaten-vets-health-care
- Nutton V. Ancient Medicine. 2nd ed. Routledge; 2012.
- US Department of Veterans Affairs. VA History Summary. Updated June 13, 2025. Accessed October 27, 2025. https://department.va.gov/history/history-overview/
- Geppert CMA. Learning from history: the ethical foundation of VA health care. Fed Pract. 2016;33:6-7.
- US Department of Veterans Affairs. Nationwide patient survey shows VA hospitals outperform non-VA hospitals. News release. June 14, 2023. Accessed October 27, 2025. https://news.va.gov/press-room/nationwide-patient-survey-shows-va-hospitals-outperform-non-va-hospitals
- Chan DC, Danesh K, Costantini S, Card D, Taylor L, Studdert DM. Mortality among US veterans after emergency visits to Veterans Affairs and other hospitals: retrospective cohort study. BMJ. 2022;376:e068099. doi:10.1136/bmj-2021-068099
- Vigilante K, Batten SV, Shang Q, et al. Camaraderie among US veterans and their preferences for health care systems and practitioners. JAMA Netw Open. 2025;8(4):e255253. doi:10.1001/jamanetworkopen.2025.5253
- Rasmussen P, Farmer CM. The promise and challenges of VA community care: veterans’ issues in focus. Rand Health Q. 2023;10:9.
- Kierkegaard S. Journalen JJ:167 (1843) in: Søren Kierkegaards Skrifter. Vol 18. Copenhagen; 1997:306.
The Once and Future Veterans Health Administration
The Once and Future Veterans Health Administration
Special Report II: Tackling Burnout
Last month, we introduced the epidemic of burnout and the adverse consequences for both our vascular surgery patients and ourselves. Today we will outline a framework for addressing these issues. The foundation of this framework is informed by the social and neurosciences.
From the perspective of the social scientist: Christina Maslach, the originator of the widely used Maslach Burnout Inventory, theorized that burnout arises from a chronic mismatch between people and their work setting in some or all of the following domains: Workload (too much, wrong kind); control (lack of autonomy, or insufficient control over resources); reward (insufficient financial or social rewards commensurate with achievements); community (loss of positive connection with others); fairness (lack of perceived fairness, inequity of work, pay, or promotion); and values (conflict of personal and organizational values). The reality of practicing medicine in today’s business milieu – of achieving service efficiencies by meeting performance targets – brings many of these mismatches into sharp focus.
From the perspective of the neuroscientist: Recent advances, including functional MRI, have demonstrated that the human brain is hard wired for compassion. Compassion is the deep feeling that arises when confronted with another’s suffering, coupled with a strong desire to alleviate that suffering. There are at least two neural pathways: one activated during empathy, having us experience another’s pain; and the other activated during compassion, resulting in our sense of reward. Thus, burnout is thought to occur when you know what your patient needs but you are unable to deliver it. Compassionate medical care is purposeful work, which promotes a sense of reward and mitigates burnout.
Because burnout affects all caregivers (anyone who touches the patient), a successful program addressing workforce well-being must be comprehensive and organization wide, similar to successful patient safety, CPI [continuous process improvement] and LEAN [Six Sigma] initiatives.
There are no shortcuts. Creating a culture of compassionate, collaborative care requires an understanding of the interrelationships between the individual provider, the unit or team, and organizational leadership.
1) The individual provider: There is evidence to support the use of programs that build personal resilience. A recently published meta-analysis by West and colleagues concluded that while no specific physician burnout intervention has been shown to be better than other types of interventions, mindfulness, stress management, and small-group discussions can be effective approaches to reducing burnout scores. Strategies to build individual resilience, such as mindfulness and meditation, are easy to teach but place the burden for success on the individual. No amount of resilience can withstand an unsupportive or toxic workplace environment, so both individual and organizational strategies in combination are necessary.
2) The unit or team: Scheduling time for open and honest discussion of social and emotional issues that arise in caring for patients helps nourish caregiver to caregiver compassion. The Schwartz Center for Compassionate Healthcare is a national nonprofit leading the movement to bring compassion to every patient-caregiver interaction. More than 425 health care organization are Schwartz Center members and conduct Schwartz Rounds™ to bring doctors, nurses, and other caregivers together to discuss the human side of health care. (www.theschwartzcenter.org). Team member to team member support is essential for navigating the stressors of practice. With having lunch in front of your computer being the norm, and the disappearance of traditional spaces for colleagues to connect (for example, nurses’ lounge, physician dining rooms), the opportunity for caregivers to have a safe place to escape, a place to have their own humanity reaffirmed, a place to offer support to their peers, has been eliminated.
3) Organizational Leadership: Making compassion a core value, articulating it, and establishing metrics whereby it can be measured, is a good start. The barriers to a culture of compassion are related to our systems of care. There are burgeoning administrative and documentation tasks to be performed, and productivity expectations that turn our clinics and hospitals into assembly lines. No, we cannot expect the EMR [electronic medical records] to be eliminated, but workforce well-being cannot be sustainable in the context of inadequate resources. A culture of compassionate collaborative care requires programs and policies that are implemented by the organization itself. Examples of organization-wide initiatives that support workforce well-being and provider engagement include: screening for caregiver burnout, establishing policies for managing adverse events with an eye toward the second victim, and most importantly, supporting systems that preserve work control autonomy of physicians and nurses in clinical settings. The business sector has long recognized that workplace stress is a function of how demanding a person’s job is and how much control that person has over his or her responsibilities. The business community has also recognized that the experience of the worker (provider) drives the experience of the customer (patient). In a study of hospital compassionate practices and HCAHPS [the Hospital Consumer Assessment of Healthcare Providers and Systems], McClelland and Vogus reported that how well a hospital compassionately supports it employees and rewards compassionate acts is significantly and positively is associated with that hospital’s ratings and likelihood of patients recommending it.
How does the Society of Vascular Surgery, or any professional medical/nursing society for that matter, fit into this model?
We propose that the SVS find ways to empower their members to be agents for culture change within their own health care organizations. How might this be done:
- Teach organizational leadership skills, starting with the SVS Board of Directors, the presidential line, and the chairs of committees. Offer leadership courses at the Annual Meeting.
- Develop a community of caregivers committed to creating a compassionate collaborative culture. The SVS is a founding member of the Schwartz Center Healthcare Society Leadership Council, and you, as members of the SVS benefit from reduced registration at the Annual Compassion in Action Healthcare Conference, June 24-27, 2017 in Boston. (http://compassioninactionconference.org) This conference is designed to be highly experiential, using a hands-on “how to do it” model.
- The SVS should make improving the overall wellness of its members a specific goal and find specific metrics to monitor our progress towards this goal. Members can be provided with the tools to identify, monitor, and measure burnout and compassion. Each committee and council of the SVS can reexamine their objectives through the lens of reducing burnout and improving the wellness of vascular surgeons.
- Provide members with evidence-based programs that build personal resilience. This will not be a successful initiative unless our surgeons recognize and acknowledge the symptoms of burnout, and are willing to admit vulnerability. Without doing so, it is difficult to reach out for help.
- Redesign postgraduate resident and fellowship education. Standardizing clinical care may reduce variation and promote efficiency. However, when processes such as time-limited appointment scheduling, EMR templates, and protocols that drive physician-patient interactions are embedded in Resident and Fellowship education, the result may well be inflexibility in practice, reduced face time with patients, and interactions that lack compassion; all leading to burnout. Graduate Medical Education leaders must develop programs that support the learner’s ability to connect with patients and families, cultivate and role-model skills and behaviors that strengthen compassionate interactions, and strive to develop clinical practice models that increase Resident and Fellow work control autonomy.
The SVS should work proactively to optimize workload, fairness, and reward on a larger scale for its members as it relates to the EMR, reimbursement, and systems coverage. While we may be relatively small in size, as leaders, we are perfectly poised to address these larger, global issues. Perhaps working within the current system (i.e., PAC and APM task force) and considering innovative solutions at a national leadership scale, the SVS can direct real change!
Changing culture is not easy, nor quick, nor does it have an easy-to-follow blueprint. The first step is recognizing the need. The second is taking a leadership role. The third is thinking deeply about implementation.
*The authors extend their thanks and appreciation for the guidance, resources and support of Michael Goldberg, MD, scholar in residence, Schwartz Center for Compassionate Care, Boston and clinical professor of orthopedics at Seattle Children’s Hospital.
REFERENCES
1. J Managerial Psychol. (2007) 22:309-28
2. Annu Rev Neurosci. (2012) 35:1-23
3. Medicine. (2016) 44:583-5
4. J Health Organization Manag. (2015) 29:973-87
5. De Zulueta P Developing compassionate leadership in health care: an integrative review. J Healthcare Leadership. (2016) 8:1-10
6. Dolan ED, Morh D, Lempa M et al. Using a single item to measure burnout in primary care staff: A psychometry evaluation. J Gen Intern Med. (2015) 30:582-7
7. Karasek RA Job demands, job decision latitude, and mental strain: implications for job design. Administrative Sciences Quarterly (1979) 24: 285-308
8. Lee VS, Miller T, Daniels C, et al. Creating the exceptional patient experience in one academic health system. Acad Med. (2016) 91:338-44
9. Linzer M, Levine R, Meltzer D, et al. 10 bold steps to prevent burnout in general internal medicine. J Gen Intern Med. (2013) 29:18-20
10. Lown BA, Manning CF The Schwartz Center Rounds: Evaluation of an interdisciplinary approach to enhancing patient-centered communication, teamwork, and provider support. Acad Med. (2010) 85:1073-81
11. Lown BA, Muncer SJ, Chadwick R Can compassionate healthcare be measured? The Schwartz Center Compassionate Care Scale. Patient Education and Counseling (2015) 98:1005-10
12. Lown BA, McIntosh S, Gaines ME, et. al. Integrating compassionate collaborative care (“the Triple C”) into health professional education to advance the triple aim of health care. Acad Med (2016) 91:1-7
13. Lown BA A social neuroscience-informed model for teaching and practicing compassion in health care. Medical Education (2016) 50: 332-342
14. Maslach C, Schaufeli WG, Leiter MP Job burnout. Annu Rev Psychol (2001) 52:397-422
15. McClelland LE, Vogus TJ Compassion practices and HCAHPS: Does rewarding and supporting workplace compassion influence patient perceptions? HSR: Health Serv Res. (2014) 49:1670-83
16. Shanafelt TD, Noseworthy JH Executive leadership and physician well-being: Nine organizational strategies to promote engagement and reduce burnout. Mayo Clin Proc. (2016) 6:1-18
17. Shanafelt TD, Dyrbye LN, West CP Addressing physician burnout: the way forward. JAMA (2017) 317:901-2
18. Singer T, Klimecki OM Empathy and compassion Curr Biol. (2014) 24: R875-8
19. West CP, Dyrbye LN, Satele DV et. al. Concurrent validity of single-item measures of emotional exhaustion and depersonalization in burnout assessment. J Gen Intern Med. (2012) 27:1445-52
20. West CP, Dyrbye LN, Erwin PJ, et al. Interventions to address and reduce physician burnout: a systematic review and meta-analysis. Lancet. (2016) 388:2272-81
21. Wuest TK, Goldberg MJ, Kelly JD Clinical faceoff: Physician burnout-Fact, fantasy, or the fourth component of the triple aim? Clin Orthop Relat Res. (2016) doi: 10.1007/5-11999-016-5193-5
Last month, we introduced the epidemic of burnout and the adverse consequences for both our vascular surgery patients and ourselves. Today we will outline a framework for addressing these issues. The foundation of this framework is informed by the social and neurosciences.
From the perspective of the social scientist: Christina Maslach, the originator of the widely used Maslach Burnout Inventory, theorized that burnout arises from a chronic mismatch between people and their work setting in some or all of the following domains: Workload (too much, wrong kind); control (lack of autonomy, or insufficient control over resources); reward (insufficient financial or social rewards commensurate with achievements); community (loss of positive connection with others); fairness (lack of perceived fairness, inequity of work, pay, or promotion); and values (conflict of personal and organizational values). The reality of practicing medicine in today’s business milieu – of achieving service efficiencies by meeting performance targets – brings many of these mismatches into sharp focus.
From the perspective of the neuroscientist: Recent advances, including functional MRI, have demonstrated that the human brain is hard wired for compassion. Compassion is the deep feeling that arises when confronted with another’s suffering, coupled with a strong desire to alleviate that suffering. There are at least two neural pathways: one activated during empathy, having us experience another’s pain; and the other activated during compassion, resulting in our sense of reward. Thus, burnout is thought to occur when you know what your patient needs but you are unable to deliver it. Compassionate medical care is purposeful work, which promotes a sense of reward and mitigates burnout.
Because burnout affects all caregivers (anyone who touches the patient), a successful program addressing workforce well-being must be comprehensive and organization wide, similar to successful patient safety, CPI [continuous process improvement] and LEAN [Six Sigma] initiatives.
There are no shortcuts. Creating a culture of compassionate, collaborative care requires an understanding of the interrelationships between the individual provider, the unit or team, and organizational leadership.
1) The individual provider: There is evidence to support the use of programs that build personal resilience. A recently published meta-analysis by West and colleagues concluded that while no specific physician burnout intervention has been shown to be better than other types of interventions, mindfulness, stress management, and small-group discussions can be effective approaches to reducing burnout scores. Strategies to build individual resilience, such as mindfulness and meditation, are easy to teach but place the burden for success on the individual. No amount of resilience can withstand an unsupportive or toxic workplace environment, so both individual and organizational strategies in combination are necessary.
2) The unit or team: Scheduling time for open and honest discussion of social and emotional issues that arise in caring for patients helps nourish caregiver to caregiver compassion. The Schwartz Center for Compassionate Healthcare is a national nonprofit leading the movement to bring compassion to every patient-caregiver interaction. More than 425 health care organization are Schwartz Center members and conduct Schwartz Rounds™ to bring doctors, nurses, and other caregivers together to discuss the human side of health care. (www.theschwartzcenter.org). Team member to team member support is essential for navigating the stressors of practice. With having lunch in front of your computer being the norm, and the disappearance of traditional spaces for colleagues to connect (for example, nurses’ lounge, physician dining rooms), the opportunity for caregivers to have a safe place to escape, a place to have their own humanity reaffirmed, a place to offer support to their peers, has been eliminated.
3) Organizational Leadership: Making compassion a core value, articulating it, and establishing metrics whereby it can be measured, is a good start. The barriers to a culture of compassion are related to our systems of care. There are burgeoning administrative and documentation tasks to be performed, and productivity expectations that turn our clinics and hospitals into assembly lines. No, we cannot expect the EMR [electronic medical records] to be eliminated, but workforce well-being cannot be sustainable in the context of inadequate resources. A culture of compassionate collaborative care requires programs and policies that are implemented by the organization itself. Examples of organization-wide initiatives that support workforce well-being and provider engagement include: screening for caregiver burnout, establishing policies for managing adverse events with an eye toward the second victim, and most importantly, supporting systems that preserve work control autonomy of physicians and nurses in clinical settings. The business sector has long recognized that workplace stress is a function of how demanding a person’s job is and how much control that person has over his or her responsibilities. The business community has also recognized that the experience of the worker (provider) drives the experience of the customer (patient). In a study of hospital compassionate practices and HCAHPS [the Hospital Consumer Assessment of Healthcare Providers and Systems], McClelland and Vogus reported that how well a hospital compassionately supports it employees and rewards compassionate acts is significantly and positively is associated with that hospital’s ratings and likelihood of patients recommending it.
How does the Society of Vascular Surgery, or any professional medical/nursing society for that matter, fit into this model?
We propose that the SVS find ways to empower their members to be agents for culture change within their own health care organizations. How might this be done:
- Teach organizational leadership skills, starting with the SVS Board of Directors, the presidential line, and the chairs of committees. Offer leadership courses at the Annual Meeting.
- Develop a community of caregivers committed to creating a compassionate collaborative culture. The SVS is a founding member of the Schwartz Center Healthcare Society Leadership Council, and you, as members of the SVS benefit from reduced registration at the Annual Compassion in Action Healthcare Conference, June 24-27, 2017 in Boston. (http://compassioninactionconference.org) This conference is designed to be highly experiential, using a hands-on “how to do it” model.
- The SVS should make improving the overall wellness of its members a specific goal and find specific metrics to monitor our progress towards this goal. Members can be provided with the tools to identify, monitor, and measure burnout and compassion. Each committee and council of the SVS can reexamine their objectives through the lens of reducing burnout and improving the wellness of vascular surgeons.
- Provide members with evidence-based programs that build personal resilience. This will not be a successful initiative unless our surgeons recognize and acknowledge the symptoms of burnout, and are willing to admit vulnerability. Without doing so, it is difficult to reach out for help.
- Redesign postgraduate resident and fellowship education. Standardizing clinical care may reduce variation and promote efficiency. However, when processes such as time-limited appointment scheduling, EMR templates, and protocols that drive physician-patient interactions are embedded in Resident and Fellowship education, the result may well be inflexibility in practice, reduced face time with patients, and interactions that lack compassion; all leading to burnout. Graduate Medical Education leaders must develop programs that support the learner’s ability to connect with patients and families, cultivate and role-model skills and behaviors that strengthen compassionate interactions, and strive to develop clinical practice models that increase Resident and Fellow work control autonomy.
The SVS should work proactively to optimize workload, fairness, and reward on a larger scale for its members as it relates to the EMR, reimbursement, and systems coverage. While we may be relatively small in size, as leaders, we are perfectly poised to address these larger, global issues. Perhaps working within the current system (i.e., PAC and APM task force) and considering innovative solutions at a national leadership scale, the SVS can direct real change!
Changing culture is not easy, nor quick, nor does it have an easy-to-follow blueprint. The first step is recognizing the need. The second is taking a leadership role. The third is thinking deeply about implementation.
*The authors extend their thanks and appreciation for the guidance, resources and support of Michael Goldberg, MD, scholar in residence, Schwartz Center for Compassionate Care, Boston and clinical professor of orthopedics at Seattle Children’s Hospital.
REFERENCES
1. J Managerial Psychol. (2007) 22:309-28
2. Annu Rev Neurosci. (2012) 35:1-23
3. Medicine. (2016) 44:583-5
4. J Health Organization Manag. (2015) 29:973-87
5. De Zulueta P Developing compassionate leadership in health care: an integrative review. J Healthcare Leadership. (2016) 8:1-10
6. Dolan ED, Morh D, Lempa M et al. Using a single item to measure burnout in primary care staff: A psychometry evaluation. J Gen Intern Med. (2015) 30:582-7
7. Karasek RA Job demands, job decision latitude, and mental strain: implications for job design. Administrative Sciences Quarterly (1979) 24: 285-308
8. Lee VS, Miller T, Daniels C, et al. Creating the exceptional patient experience in one academic health system. Acad Med. (2016) 91:338-44
9. Linzer M, Levine R, Meltzer D, et al. 10 bold steps to prevent burnout in general internal medicine. J Gen Intern Med. (2013) 29:18-20
10. Lown BA, Manning CF The Schwartz Center Rounds: Evaluation of an interdisciplinary approach to enhancing patient-centered communication, teamwork, and provider support. Acad Med. (2010) 85:1073-81
11. Lown BA, Muncer SJ, Chadwick R Can compassionate healthcare be measured? The Schwartz Center Compassionate Care Scale. Patient Education and Counseling (2015) 98:1005-10
12. Lown BA, McIntosh S, Gaines ME, et. al. Integrating compassionate collaborative care (“the Triple C”) into health professional education to advance the triple aim of health care. Acad Med (2016) 91:1-7
13. Lown BA A social neuroscience-informed model for teaching and practicing compassion in health care. Medical Education (2016) 50: 332-342
14. Maslach C, Schaufeli WG, Leiter MP Job burnout. Annu Rev Psychol (2001) 52:397-422
15. McClelland LE, Vogus TJ Compassion practices and HCAHPS: Does rewarding and supporting workplace compassion influence patient perceptions? HSR: Health Serv Res. (2014) 49:1670-83
16. Shanafelt TD, Noseworthy JH Executive leadership and physician well-being: Nine organizational strategies to promote engagement and reduce burnout. Mayo Clin Proc. (2016) 6:1-18
17. Shanafelt TD, Dyrbye LN, West CP Addressing physician burnout: the way forward. JAMA (2017) 317:901-2
18. Singer T, Klimecki OM Empathy and compassion Curr Biol. (2014) 24: R875-8
19. West CP, Dyrbye LN, Satele DV et. al. Concurrent validity of single-item measures of emotional exhaustion and depersonalization in burnout assessment. J Gen Intern Med. (2012) 27:1445-52
20. West CP, Dyrbye LN, Erwin PJ, et al. Interventions to address and reduce physician burnout: a systematic review and meta-analysis. Lancet. (2016) 388:2272-81
21. Wuest TK, Goldberg MJ, Kelly JD Clinical faceoff: Physician burnout-Fact, fantasy, or the fourth component of the triple aim? Clin Orthop Relat Res. (2016) doi: 10.1007/5-11999-016-5193-5
Last month, we introduced the epidemic of burnout and the adverse consequences for both our vascular surgery patients and ourselves. Today we will outline a framework for addressing these issues. The foundation of this framework is informed by the social and neurosciences.
From the perspective of the social scientist: Christina Maslach, the originator of the widely used Maslach Burnout Inventory, theorized that burnout arises from a chronic mismatch between people and their work setting in some or all of the following domains: Workload (too much, wrong kind); control (lack of autonomy, or insufficient control over resources); reward (insufficient financial or social rewards commensurate with achievements); community (loss of positive connection with others); fairness (lack of perceived fairness, inequity of work, pay, or promotion); and values (conflict of personal and organizational values). The reality of practicing medicine in today’s business milieu – of achieving service efficiencies by meeting performance targets – brings many of these mismatches into sharp focus.
From the perspective of the neuroscientist: Recent advances, including functional MRI, have demonstrated that the human brain is hard wired for compassion. Compassion is the deep feeling that arises when confronted with another’s suffering, coupled with a strong desire to alleviate that suffering. There are at least two neural pathways: one activated during empathy, having us experience another’s pain; and the other activated during compassion, resulting in our sense of reward. Thus, burnout is thought to occur when you know what your patient needs but you are unable to deliver it. Compassionate medical care is purposeful work, which promotes a sense of reward and mitigates burnout.
Because burnout affects all caregivers (anyone who touches the patient), a successful program addressing workforce well-being must be comprehensive and organization wide, similar to successful patient safety, CPI [continuous process improvement] and LEAN [Six Sigma] initiatives.
There are no shortcuts. Creating a culture of compassionate, collaborative care requires an understanding of the interrelationships between the individual provider, the unit or team, and organizational leadership.
1) The individual provider: There is evidence to support the use of programs that build personal resilience. A recently published meta-analysis by West and colleagues concluded that while no specific physician burnout intervention has been shown to be better than other types of interventions, mindfulness, stress management, and small-group discussions can be effective approaches to reducing burnout scores. Strategies to build individual resilience, such as mindfulness and meditation, are easy to teach but place the burden for success on the individual. No amount of resilience can withstand an unsupportive or toxic workplace environment, so both individual and organizational strategies in combination are necessary.
2) The unit or team: Scheduling time for open and honest discussion of social and emotional issues that arise in caring for patients helps nourish caregiver to caregiver compassion. The Schwartz Center for Compassionate Healthcare is a national nonprofit leading the movement to bring compassion to every patient-caregiver interaction. More than 425 health care organization are Schwartz Center members and conduct Schwartz Rounds™ to bring doctors, nurses, and other caregivers together to discuss the human side of health care. (www.theschwartzcenter.org). Team member to team member support is essential for navigating the stressors of practice. With having lunch in front of your computer being the norm, and the disappearance of traditional spaces for colleagues to connect (for example, nurses’ lounge, physician dining rooms), the opportunity for caregivers to have a safe place to escape, a place to have their own humanity reaffirmed, a place to offer support to their peers, has been eliminated.
3) Organizational Leadership: Making compassion a core value, articulating it, and establishing metrics whereby it can be measured, is a good start. The barriers to a culture of compassion are related to our systems of care. There are burgeoning administrative and documentation tasks to be performed, and productivity expectations that turn our clinics and hospitals into assembly lines. No, we cannot expect the EMR [electronic medical records] to be eliminated, but workforce well-being cannot be sustainable in the context of inadequate resources. A culture of compassionate collaborative care requires programs and policies that are implemented by the organization itself. Examples of organization-wide initiatives that support workforce well-being and provider engagement include: screening for caregiver burnout, establishing policies for managing adverse events with an eye toward the second victim, and most importantly, supporting systems that preserve work control autonomy of physicians and nurses in clinical settings. The business sector has long recognized that workplace stress is a function of how demanding a person’s job is and how much control that person has over his or her responsibilities. The business community has also recognized that the experience of the worker (provider) drives the experience of the customer (patient). In a study of hospital compassionate practices and HCAHPS [the Hospital Consumer Assessment of Healthcare Providers and Systems], McClelland and Vogus reported that how well a hospital compassionately supports it employees and rewards compassionate acts is significantly and positively is associated with that hospital’s ratings and likelihood of patients recommending it.
How does the Society of Vascular Surgery, or any professional medical/nursing society for that matter, fit into this model?
We propose that the SVS find ways to empower their members to be agents for culture change within their own health care organizations. How might this be done:
- Teach organizational leadership skills, starting with the SVS Board of Directors, the presidential line, and the chairs of committees. Offer leadership courses at the Annual Meeting.
- Develop a community of caregivers committed to creating a compassionate collaborative culture. The SVS is a founding member of the Schwartz Center Healthcare Society Leadership Council, and you, as members of the SVS benefit from reduced registration at the Annual Compassion in Action Healthcare Conference, June 24-27, 2017 in Boston. (http://compassioninactionconference.org) This conference is designed to be highly experiential, using a hands-on “how to do it” model.
- The SVS should make improving the overall wellness of its members a specific goal and find specific metrics to monitor our progress towards this goal. Members can be provided with the tools to identify, monitor, and measure burnout and compassion. Each committee and council of the SVS can reexamine their objectives through the lens of reducing burnout and improving the wellness of vascular surgeons.
- Provide members with evidence-based programs that build personal resilience. This will not be a successful initiative unless our surgeons recognize and acknowledge the symptoms of burnout, and are willing to admit vulnerability. Without doing so, it is difficult to reach out for help.
- Redesign postgraduate resident and fellowship education. Standardizing clinical care may reduce variation and promote efficiency. However, when processes such as time-limited appointment scheduling, EMR templates, and protocols that drive physician-patient interactions are embedded in Resident and Fellowship education, the result may well be inflexibility in practice, reduced face time with patients, and interactions that lack compassion; all leading to burnout. Graduate Medical Education leaders must develop programs that support the learner’s ability to connect with patients and families, cultivate and role-model skills and behaviors that strengthen compassionate interactions, and strive to develop clinical practice models that increase Resident and Fellow work control autonomy.
The SVS should work proactively to optimize workload, fairness, and reward on a larger scale for its members as it relates to the EMR, reimbursement, and systems coverage. While we may be relatively small in size, as leaders, we are perfectly poised to address these larger, global issues. Perhaps working within the current system (i.e., PAC and APM task force) and considering innovative solutions at a national leadership scale, the SVS can direct real change!
Changing culture is not easy, nor quick, nor does it have an easy-to-follow blueprint. The first step is recognizing the need. The second is taking a leadership role. The third is thinking deeply about implementation.
*The authors extend their thanks and appreciation for the guidance, resources and support of Michael Goldberg, MD, scholar in residence, Schwartz Center for Compassionate Care, Boston and clinical professor of orthopedics at Seattle Children’s Hospital.
REFERENCES
1. J Managerial Psychol. (2007) 22:309-28
2. Annu Rev Neurosci. (2012) 35:1-23
3. Medicine. (2016) 44:583-5
4. J Health Organization Manag. (2015) 29:973-87
5. De Zulueta P Developing compassionate leadership in health care: an integrative review. J Healthcare Leadership. (2016) 8:1-10
6. Dolan ED, Morh D, Lempa M et al. Using a single item to measure burnout in primary care staff: A psychometry evaluation. J Gen Intern Med. (2015) 30:582-7
7. Karasek RA Job demands, job decision latitude, and mental strain: implications for job design. Administrative Sciences Quarterly (1979) 24: 285-308
8. Lee VS, Miller T, Daniels C, et al. Creating the exceptional patient experience in one academic health system. Acad Med. (2016) 91:338-44
9. Linzer M, Levine R, Meltzer D, et al. 10 bold steps to prevent burnout in general internal medicine. J Gen Intern Med. (2013) 29:18-20
10. Lown BA, Manning CF The Schwartz Center Rounds: Evaluation of an interdisciplinary approach to enhancing patient-centered communication, teamwork, and provider support. Acad Med. (2010) 85:1073-81
11. Lown BA, Muncer SJ, Chadwick R Can compassionate healthcare be measured? The Schwartz Center Compassionate Care Scale. Patient Education and Counseling (2015) 98:1005-10
12. Lown BA, McIntosh S, Gaines ME, et. al. Integrating compassionate collaborative care (“the Triple C”) into health professional education to advance the triple aim of health care. Acad Med (2016) 91:1-7
13. Lown BA A social neuroscience-informed model for teaching and practicing compassion in health care. Medical Education (2016) 50: 332-342
14. Maslach C, Schaufeli WG, Leiter MP Job burnout. Annu Rev Psychol (2001) 52:397-422
15. McClelland LE, Vogus TJ Compassion practices and HCAHPS: Does rewarding and supporting workplace compassion influence patient perceptions? HSR: Health Serv Res. (2014) 49:1670-83
16. Shanafelt TD, Noseworthy JH Executive leadership and physician well-being: Nine organizational strategies to promote engagement and reduce burnout. Mayo Clin Proc. (2016) 6:1-18
17. Shanafelt TD, Dyrbye LN, West CP Addressing physician burnout: the way forward. JAMA (2017) 317:901-2
18. Singer T, Klimecki OM Empathy and compassion Curr Biol. (2014) 24: R875-8
19. West CP, Dyrbye LN, Satele DV et. al. Concurrent validity of single-item measures of emotional exhaustion and depersonalization in burnout assessment. J Gen Intern Med. (2012) 27:1445-52
20. West CP, Dyrbye LN, Erwin PJ, et al. Interventions to address and reduce physician burnout: a systematic review and meta-analysis. Lancet. (2016) 388:2272-81
21. Wuest TK, Goldberg MJ, Kelly JD Clinical faceoff: Physician burnout-Fact, fantasy, or the fourth component of the triple aim? Clin Orthop Relat Res. (2016) doi: 10.1007/5-11999-016-5193-5
Transplantation palliative care: The time is ripe
Over 10 years ago, a challenge was made in a surgical publication for increased collaboration between the fields of transplantation and palliative care.1
Since that time not much progress has been made bringing these fields together in a consistent way that would mutually benefit patients and the specialties. However, other progress has been made, particularly in the field of palliative care, which could brighten the prospects and broaden the opportunities to accomplish collaboration between palliative care and transplantation.
Growth of palliative services
During the past decade there has been a robust proliferation of hospital-based palliative care programs in the United States. In all, 67% of U.S. hospitals with 50 or more beds report palliative care teams, up from 63% in 2011 and 53% in 2008.
Only a decade ago, critical care and palliative care were generally considered mutually exclusive. Evidence is trickling in to suggest that this is no longer the case. Although palliative care was not an integral part of critical care at that time, patients, families, and even practitioners began to demand these services. Cook and Rocker have eloquently advocated the rightful place of palliative care in the ICU.2
Studies in recent years have shown that the integration of palliative care into critical care decreases in length of ICU and hospital stay, decreases costs, enhances patient/family satisfaction, and promotes a more rapid consensus about goals of care, without increasing mortality. The ICU experience to date could be considered a reassuring precedent for transplantation palliative care.
Integration of palliative care with transplantation
Early palliative care intervention has been shown to improve symptom burden and depression scores in end-stage liver disease patients awaiting transplant. In addition, early palliative care consultation in conjunction with cancer treatment has been associated with increased survival in non–small-cell lung cancer patients. It has been demonstrated that early integration of palliative care in the surgical ICU alongside disease-directed curative care can be accomplished without change in mortality, while improving end-of-life practice in liver transplant patients.3
What palliative care can do for transplant patients
What does palliative care mean for the person (and family) awaiting transplantation? For the cirrhotic patient with cachexia, ascites, and encephalopathy, it means access to the services of a team trained in the management of these symptoms. Palliative care teams can also provide psychosocial and spiritual support for patients and families who are intimidated by the complex navigation of the health care system and the existential threat that end-stage organ failure presents to them. Skilled palliative care and services can be the difference between failing and extended life with a higher quality of life for these very sick patients
Resuscitation of a patient, whether through restoration of organ function or interdicting the progression of disease, begins with resuscitation of hope. Nothing achieves this more quickly than amelioration of burdensome symptoms for the patient and family.
The barriers for transplant surgeons and teams referring and incorporating palliative care services in their practices are multiple and profound. The unique dilemma facing the transplant team is to balance the treatment of the failing organ, the treatment of the patient (and family and friends), and the best use of the graft, a precious gift of society.
Palliative surgery has been defined as any invasive procedure in which the main intention is to mitigate physical symptoms in patients with noncurable disease without causing premature death. The very success of transplantation over the past 3 decades has obscured our memory of transplantation as a type of palliative surgery. It is a well-known axiom of reconstructive surgery that the reconstructed site should be compared to what was there, not to “normal.” Even in the current era of improved immunosuppression and posttransplant support services, one could hardly describe even a successful transplant patient’s experience as “normal.” These patients’ lives may be extended and/or enhanced but they need palliative care before, during, and after transplantation. The growing availability of trained palliative care clinicians and teams, the increased familiarity of palliative and end-of-life care to surgical residents and fellows, and quality metrics measuring palliative care outcomes will provide reassurance and guidance to address reservations about the convergence of the two seemingly opposite realities.
A modest proposal
We propose that palliative care be presented to the entire spectrum of transplantation care: on the ward, in the ICU, and after transplantation. More specific “triggers” for palliative care for referral of transplant patients should be identified. Wentlandt et al.4 have described a promising model for an ambulatory clinic, which provides early, integrated palliative care to patients awaiting and receiving organ transplantation. In addition, we propose an application for grant funding for a conference and eventual formation of a work group of transplant surgeons and team members, palliative care clinicians, and patient/families who have experienced one of the aspects of the transplant spectrum. We await the subspecialty certification in hospice and palliative medicine of a transplant surgeon. Outside of transplantation, every other surgical specialty in the United States has diplomates certified in hospice and palliative medicine. We await the benefits that will accrue from research about the merging of these fields.
1. Molmenti EP, Dunn GP: Transplantation and palliative care: The convergence of two seemingly opposite realities. Surg Clin North Am. 2005;85:373-82.
2. Cook D, Rocker G. Dying with dignity in the intensive care unit. N Engl J Med. 2014;370:2506-14.
3. Lamba S, Murphy P, McVicker S, Smith JH, and Mosenthal AC. Changing end-of-life care practice for liver transplant patients: structured palliative care intervention in the surgical intensive care unit. J Pain Symptom Manage. 2012; 44(4):508-19.
4. Wentlandt, K., Dall’Osto, A., Freeman, N., Le, L. W., Kaya, E., Ross, H., Singer, L. G., Abbey, S., Clarke, H. and Zimmermann, C. (2016), The Transplant Palliative Care Clinic: An early palliative care model for patients in a transplant program. Clin Transplant. 2016 Nov 4; doi: 10.1111/ctr.12838.
Dr. Azoulay is a transplantation specialist of Assistance Publique – Hôpitaux de Paris, and the University of Paris. Dr. Dunn is medical director of the Palliative Care Consultation Service at the University of Pittsburgh Medical Center Hamot, and vice-chair of the ACS Committee on Surgical Palliative Care.
Over 10 years ago, a challenge was made in a surgical publication for increased collaboration between the fields of transplantation and palliative care.1
Since that time not much progress has been made bringing these fields together in a consistent way that would mutually benefit patients and the specialties. However, other progress has been made, particularly in the field of palliative care, which could brighten the prospects and broaden the opportunities to accomplish collaboration between palliative care and transplantation.
Growth of palliative services
During the past decade there has been a robust proliferation of hospital-based palliative care programs in the United States. In all, 67% of U.S. hospitals with 50 or more beds report palliative care teams, up from 63% in 2011 and 53% in 2008.
Only a decade ago, critical care and palliative care were generally considered mutually exclusive. Evidence is trickling in to suggest that this is no longer the case. Although palliative care was not an integral part of critical care at that time, patients, families, and even practitioners began to demand these services. Cook and Rocker have eloquently advocated the rightful place of palliative care in the ICU.2
Studies in recent years have shown that the integration of palliative care into critical care decreases in length of ICU and hospital stay, decreases costs, enhances patient/family satisfaction, and promotes a more rapid consensus about goals of care, without increasing mortality. The ICU experience to date could be considered a reassuring precedent for transplantation palliative care.
Integration of palliative care with transplantation
Early palliative care intervention has been shown to improve symptom burden and depression scores in end-stage liver disease patients awaiting transplant. In addition, early palliative care consultation in conjunction with cancer treatment has been associated with increased survival in non–small-cell lung cancer patients. It has been demonstrated that early integration of palliative care in the surgical ICU alongside disease-directed curative care can be accomplished without change in mortality, while improving end-of-life practice in liver transplant patients.3
What palliative care can do for transplant patients
What does palliative care mean for the person (and family) awaiting transplantation? For the cirrhotic patient with cachexia, ascites, and encephalopathy, it means access to the services of a team trained in the management of these symptoms. Palliative care teams can also provide psychosocial and spiritual support for patients and families who are intimidated by the complex navigation of the health care system and the existential threat that end-stage organ failure presents to them. Skilled palliative care and services can be the difference between failing and extended life with a higher quality of life for these very sick patients
Resuscitation of a patient, whether through restoration of organ function or interdicting the progression of disease, begins with resuscitation of hope. Nothing achieves this more quickly than amelioration of burdensome symptoms for the patient and family.
The barriers for transplant surgeons and teams referring and incorporating palliative care services in their practices are multiple and profound. The unique dilemma facing the transplant team is to balance the treatment of the failing organ, the treatment of the patient (and family and friends), and the best use of the graft, a precious gift of society.
Palliative surgery has been defined as any invasive procedure in which the main intention is to mitigate physical symptoms in patients with noncurable disease without causing premature death. The very success of transplantation over the past 3 decades has obscured our memory of transplantation as a type of palliative surgery. It is a well-known axiom of reconstructive surgery that the reconstructed site should be compared to what was there, not to “normal.” Even in the current era of improved immunosuppression and posttransplant support services, one could hardly describe even a successful transplant patient’s experience as “normal.” These patients’ lives may be extended and/or enhanced but they need palliative care before, during, and after transplantation. The growing availability of trained palliative care clinicians and teams, the increased familiarity of palliative and end-of-life care to surgical residents and fellows, and quality metrics measuring palliative care outcomes will provide reassurance and guidance to address reservations about the convergence of the two seemingly opposite realities.
A modest proposal
We propose that palliative care be presented to the entire spectrum of transplantation care: on the ward, in the ICU, and after transplantation. More specific “triggers” for palliative care for referral of transplant patients should be identified. Wentlandt et al.4 have described a promising model for an ambulatory clinic, which provides early, integrated palliative care to patients awaiting and receiving organ transplantation. In addition, we propose an application for grant funding for a conference and eventual formation of a work group of transplant surgeons and team members, palliative care clinicians, and patient/families who have experienced one of the aspects of the transplant spectrum. We await the subspecialty certification in hospice and palliative medicine of a transplant surgeon. Outside of transplantation, every other surgical specialty in the United States has diplomates certified in hospice and palliative medicine. We await the benefits that will accrue from research about the merging of these fields.
1. Molmenti EP, Dunn GP: Transplantation and palliative care: The convergence of two seemingly opposite realities. Surg Clin North Am. 2005;85:373-82.
2. Cook D, Rocker G. Dying with dignity in the intensive care unit. N Engl J Med. 2014;370:2506-14.
3. Lamba S, Murphy P, McVicker S, Smith JH, and Mosenthal AC. Changing end-of-life care practice for liver transplant patients: structured palliative care intervention in the surgical intensive care unit. J Pain Symptom Manage. 2012; 44(4):508-19.
4. Wentlandt, K., Dall’Osto, A., Freeman, N., Le, L. W., Kaya, E., Ross, H., Singer, L. G., Abbey, S., Clarke, H. and Zimmermann, C. (2016), The Transplant Palliative Care Clinic: An early palliative care model for patients in a transplant program. Clin Transplant. 2016 Nov 4; doi: 10.1111/ctr.12838.
Dr. Azoulay is a transplantation specialist of Assistance Publique – Hôpitaux de Paris, and the University of Paris. Dr. Dunn is medical director of the Palliative Care Consultation Service at the University of Pittsburgh Medical Center Hamot, and vice-chair of the ACS Committee on Surgical Palliative Care.
Over 10 years ago, a challenge was made in a surgical publication for increased collaboration between the fields of transplantation and palliative care.1
Since that time not much progress has been made bringing these fields together in a consistent way that would mutually benefit patients and the specialties. However, other progress has been made, particularly in the field of palliative care, which could brighten the prospects and broaden the opportunities to accomplish collaboration between palliative care and transplantation.
Growth of palliative services
During the past decade there has been a robust proliferation of hospital-based palliative care programs in the United States. In all, 67% of U.S. hospitals with 50 or more beds report palliative care teams, up from 63% in 2011 and 53% in 2008.
Only a decade ago, critical care and palliative care were generally considered mutually exclusive. Evidence is trickling in to suggest that this is no longer the case. Although palliative care was not an integral part of critical care at that time, patients, families, and even practitioners began to demand these services. Cook and Rocker have eloquently advocated the rightful place of palliative care in the ICU.2
Studies in recent years have shown that the integration of palliative care into critical care decreases in length of ICU and hospital stay, decreases costs, enhances patient/family satisfaction, and promotes a more rapid consensus about goals of care, without increasing mortality. The ICU experience to date could be considered a reassuring precedent for transplantation palliative care.
Integration of palliative care with transplantation
Early palliative care intervention has been shown to improve symptom burden and depression scores in end-stage liver disease patients awaiting transplant. In addition, early palliative care consultation in conjunction with cancer treatment has been associated with increased survival in non–small-cell lung cancer patients. It has been demonstrated that early integration of palliative care in the surgical ICU alongside disease-directed curative care can be accomplished without change in mortality, while improving end-of-life practice in liver transplant patients.3
What palliative care can do for transplant patients
What does palliative care mean for the person (and family) awaiting transplantation? For the cirrhotic patient with cachexia, ascites, and encephalopathy, it means access to the services of a team trained in the management of these symptoms. Palliative care teams can also provide psychosocial and spiritual support for patients and families who are intimidated by the complex navigation of the health care system and the existential threat that end-stage organ failure presents to them. Skilled palliative care and services can be the difference between failing and extended life with a higher quality of life for these very sick patients
Resuscitation of a patient, whether through restoration of organ function or interdicting the progression of disease, begins with resuscitation of hope. Nothing achieves this more quickly than amelioration of burdensome symptoms for the patient and family.
The barriers for transplant surgeons and teams referring and incorporating palliative care services in their practices are multiple and profound. The unique dilemma facing the transplant team is to balance the treatment of the failing organ, the treatment of the patient (and family and friends), and the best use of the graft, a precious gift of society.
Palliative surgery has been defined as any invasive procedure in which the main intention is to mitigate physical symptoms in patients with noncurable disease without causing premature death. The very success of transplantation over the past 3 decades has obscured our memory of transplantation as a type of palliative surgery. It is a well-known axiom of reconstructive surgery that the reconstructed site should be compared to what was there, not to “normal.” Even in the current era of improved immunosuppression and posttransplant support services, one could hardly describe even a successful transplant patient’s experience as “normal.” These patients’ lives may be extended and/or enhanced but they need palliative care before, during, and after transplantation. The growing availability of trained palliative care clinicians and teams, the increased familiarity of palliative and end-of-life care to surgical residents and fellows, and quality metrics measuring palliative care outcomes will provide reassurance and guidance to address reservations about the convergence of the two seemingly opposite realities.
A modest proposal
We propose that palliative care be presented to the entire spectrum of transplantation care: on the ward, in the ICU, and after transplantation. More specific “triggers” for palliative care for referral of transplant patients should be identified. Wentlandt et al.4 have described a promising model for an ambulatory clinic, which provides early, integrated palliative care to patients awaiting and receiving organ transplantation. In addition, we propose an application for grant funding for a conference and eventual formation of a work group of transplant surgeons and team members, palliative care clinicians, and patient/families who have experienced one of the aspects of the transplant spectrum. We await the subspecialty certification in hospice and palliative medicine of a transplant surgeon. Outside of transplantation, every other surgical specialty in the United States has diplomates certified in hospice and palliative medicine. We await the benefits that will accrue from research about the merging of these fields.
1. Molmenti EP, Dunn GP: Transplantation and palliative care: The convergence of two seemingly opposite realities. Surg Clin North Am. 2005;85:373-82.
2. Cook D, Rocker G. Dying with dignity in the intensive care unit. N Engl J Med. 2014;370:2506-14.
3. Lamba S, Murphy P, McVicker S, Smith JH, and Mosenthal AC. Changing end-of-life care practice for liver transplant patients: structured palliative care intervention in the surgical intensive care unit. J Pain Symptom Manage. 2012; 44(4):508-19.
4. Wentlandt, K., Dall’Osto, A., Freeman, N., Le, L. W., Kaya, E., Ross, H., Singer, L. G., Abbey, S., Clarke, H. and Zimmermann, C. (2016), The Transplant Palliative Care Clinic: An early palliative care model for patients in a transplant program. Clin Transplant. 2016 Nov 4; doi: 10.1111/ctr.12838.
Dr. Azoulay is a transplantation specialist of Assistance Publique – Hôpitaux de Paris, and the University of Paris. Dr. Dunn is medical director of the Palliative Care Consultation Service at the University of Pittsburgh Medical Center Hamot, and vice-chair of the ACS Committee on Surgical Palliative Care.
Meeting the Needs of Those Who Have Served: The Role of VHA Specialized Mental Health Centers of Excellence
Meeting the Needs of Those Who Have Served: The Role of VHA Specialized Mental Health Centers of Excellence
Accessible and effective mental health services are a vital component of the Veterans Health Administration (VHA) mission to provide exceptional care that improves veterans’ health and well-being. Veterans are seeking mental health care at the VHA at significantly higher rates than in previous years. From 2009 through 2024, the number of veterans who received direct mental health care from the VHA increased 78%.1 The proportion of veterans enrolled in the VHA who also received direct mental health care expanded from 23% of total enrollees in 2009 to 33% in 2024. The increase in VHA mental health care delivery is also reflected in the number of outpatient mental health care and treatment visits at the VHA, which increased from 12.7 million to 21.5 million over the same period.
The Sergeant First Class (SFC) Heath Robinson Honoring Our Promise to Address Comprehensive Toxics (PACT) Act of 2022 expanded eligibility for VHA services, including mental health care and counseling, to new cohorts of toxic-exposed veterans. From 2022 to 2024, > 680,000 veterans enrolled in VHA care, and 45% of those were PACT Act-eligible cohorts.2 Research highlighted the high prevalence of physical and psychiatric comorbidities in toxic-exposed veterans.3 As such, the VHA may face greater demand for direct mental health care with these newly eligible cohorts of veterans.
Veterans often have mental health conditions (eg, depression, posttraumatic stress disorder [PTSD]), substance use disorders (SUDs), unique military experiences (eg, deployments), and injuries and illnesses (eg, traumatic brain injury [TBI]) that increase the complexity of their clinical presentation. The varied nature of these mental health conditions, as well as veterans’ unique military-related experiences, necessitates specialized centers focused on distinct high-priority areas of mental health in the VHA.
A series of public laws charged the US Department of Veterans Affairs (VA) with developing specialized mental health Centers of Excellence (CoEs) focused on high-priority areas of veteran mental health. The first of these laws, Public Law 98- 528, established the National Center for PTSD (NCPTSD), which opened in 1989.4 In 1996, Congress established specialized mental health CoEs known as Mental Illness Research, Education, and Clinical Centers (MIRECCs) across the VHA.5 To address the unique needs of post-9/11 veterans, 3 additional specialized centers were established in 2005.6 Finally, under the authority of the Secretary of the VA, specialized mental health CoEs were established to focus on SUD and integrated health care.
There are 17 geographically diverse mental health CoEs and MIRECCs across the VA (Table). CoEs are embedded in VA medical centers (VAMCs) with strong medical school academic affiliations. Organizational oversight of the CoEs is provided by the VA Office of Mental Health and Office of Suicide Prevention, respectively. As part of the oversight process, CoEs submit annual reports detailing their advancements in research, education and training, and clinical activities, as well as participate in a peer-reviewed renewal process.
These specialized centers are united in a shared tripartite mission to generate new knowledge about the causes and treatments of mental health conditions, to educate and train VHA clinicians and personnel, and to develop and implement innovative clinical programs within the VHA. This combined focus on research, education, and improved clinical care reduces the time from discovery to implementation and improves the health and well-being of veterans.
Examples of this acceleration translation from scientific discovery to clinical practice are evident throughout mental health CoEs. The NCPTSD is a leader in research on PTSD and traumatic stress. Its work led to the development of national training programs for VHA clinicians, facilitating the broad dissemination of efficacious PTSD treatments across the enterprise.7 Researchers at the Veterans Integrated Service Network (VISN) 21 Sierra Pacific MIRECC identified depression as a significant risk factor for dementia and subsequently launched the first multisite trial of repetitive transcranial magnetic stimulation (rTMS) in the VHA in 2012 (CSP #556: the effectiveness of rTMS in depressed VA patients).8 This project laid the groundwork for the national clinical rTMS program launched in 2017, which is now clinically available at 60 VAMCs. In the largest pragmatic randomized clinical trial of its kind, the VISN 4 Philadelphia and Pittsburgh MIRECC found that pharmacogenomic testing significantly reduced the number of prescription medications with predicted drug-gene interactions and improved clinical outcomes among veterans with depression.9
Mental health CoEs are also leaders in suicide prevention, a top clinical priority for the VHA. The VISN 2 New York MIRECC developed Project Life Force, a safety planning skills group for veterans with suicidal ideation, now implemented across 10 VAMCs, including telehealth hubs, outpatient settings, and veteran peer programs.10 The VISN 2 CoE for Suicide Prevention and VISN 19 Rocky Mountain MIRECC coordinate key suicide prevention services for VA, including the analysis of suicide surveillance data; evaluation of national VA suicide prevention initiatives; the support of veterans, families, and clinicians; and enhanced access to evidence-based treatments for at-risk veterans.
Mental health CoEs are a key operational partner in VHA treatment of SUDs. The CoEs in Substance Addiction Treatment and Education (CESATEs) are national resources dedicated to improving the quality, clinical outcomes, and cost-effectiveness of VHA SUD treatment. CESATEs developed and implemented a national rollout of an effective treatment for stimulant use disorders, training staff at > 120 VA programs. The VISN 1 Mid-Atlantic MIRECC’s focus on SUD and comorbid/co-occurring mental health conditions has highlighted the significant prevalence of these conditions and the impact they have on treatment response.11
Serious mental illness (SMI) (eg, schizophrenia, schizoaffective disorder) impacts up to 5% of veterans.12 VISN 22 Desert Pacific MIRECC has developed interventions to improve the lives of veterans with SMI. Its research established supported employment as an effective intervention to improve outcomes in veterans with psychotic disorders and supported its implementation in the VHA.13 Peer specialists are a cornerstone in the VHA commitment to recovery-oriented services for veterans with SMI. VISN 5 Capitol MIRECC has long championed research, clinical training, and educational activities that contributed to the effective deployment of peer specialists across the VHA enterprise.
Veterans have unique military-related experiences (eg, deployment, traumatic stress, transition to civilian status) and injuries and illnesses (eg, TBI, posttraumatic headaches) that significantly impact their mental health and quality of life.
The period between active duty and transition to civilian status is a critical time in a veteran’s life. The VISN 17 CoE Veteran Sponsorship Initiative connects veterans with VA care within 30 days postdischarge, with the option of additional support in the community. The VISN 22 CoE for Stress and Mental Health (CESAMH) develops, evaluates, and disseminates diagnostics and treatments for veterans affected by traumatic events and brain injuries, with a unique focus on supporting their whole health needs. The VISN 6 Mid-Atlantic MIRECC leads the ongoing VISN-6 Post-Deployment Mental Health (PDMH) study, the largest biorepository of post-9/11 veterans. PDMH has greatly expanded the understanding of the unique needs of post-9/11 veterans, with > 100 peer-reviewed publications to date. Veterans with mild TBI frequently experience chronic posttraumatic headaches that can be disabling and nonresponsive to treatment. The VISN 20 Northwest MIRECC demonstrated that prazosin, a repurposed, low-cost, widely available, nonaddictive medication, can safely and effectively reduce the frequency of these headaches and improve functional impairment in veterans and active-duty service members.14
Increased and enhanced access to effective mental health treatment is a priority for VA. In 2007, the VA launched the National Primary Care Mental Health Integration program, which integrated mental health services into primary care settings. The Center for Integrated Healthcare (CIH) has supported the VA in these efforts. In 2024, CIH trained > 5000 health care staff on high-fidelity integration of behavioral health and medical care. VA has also focused on increasing access to mental health services via expanded telehealth offerings. The VISN 16 MIRECC, with its unique focus on increasing access to care for rural veterans via distance-based and digital health technology, supported the VA Offices of Mental Health and Connected Care to virtualize mental health care and promote adoption and sustained use of VA Video Connect across the enterprise.
Specialized MH CoEs are uniquely equipped to support the VHA in providing training and education to VA clinicians, veterans, care partners and family members, and the community on high-priority mental health topics. Education is a core component of the MH CoEs tripartite mission. As such, MH CoEs offer national trainings, conferences, consultation services, clinical demonstration projects, development of clinical dashboards and toolkits, and public awareness campaigns. Researchers, educators, and clinicians at the CoEs frequently serve as subject matter experts on topics aligned with their respective missions. Several national rollout programs that disseminated evidence-based treatments for mental health conditions to the field (eg, cognitive behavioral therapy for depression, cognitive behavioral therapy for insomnia, and prolonged exposure) were developed at specialized CoEs.
The VHA provides advanced training, residencies, and fellowships to > 120,000 trainees annually. Many of these trainees choose to remain at the VA. Seven of 10 VHA psychologists and 6 of 10 VHA physicians trained within the VHA prior to their employment.15 The MH CoEs and MIRECCs play an important role in preparing these trainees for VHA mental health careers. These centers are funded to provide advanced postdoctoral training to physicians as well as allied health professionals in clinical and counseling psychology, social work, pharmacy, and nursing. Training is not limited to postdoctoral fellows: graduate students, residents, and interns from affiliated accredited training programs may rotate through mental health CoEs each academic year.
Conclusions
For > 30 years, mental health CoEs have brought thousands of veterans advanced treatments for their mental health needs and helped reduce death by suicide. The centers have a bright future ahead, harnessing advances in artificial intelligence and genomics to permit the matching of the individual veterans to the treatment most likely to benefit them. Precision medicine, as espoused by the Hannon Act, will not only encourage the efficient use of health care resources but also rapidly reduce pain in veterans with mental health and SUDs.
- Congressionally Mandated Report: Report on Transparency in Mental Health Care Services. US Dept of Veterans Affairs; December 2022. Accessed December 5, 2025. https://www.govinfo.gov/content/pkg/CMR-VA1-00181657/pdf/CMR-VA1-00181657.pdf
- Beckman AL, Jacobs J, Elnahal SM. The PACT Act—expanding coverage and access for veterans. JAMA. 2024;332:1423-1424. doi:10.1001/jama.2024.16013
- Morse JL, Acheson DT, Almklov E, et al. Associations among environmental exposures and physical and psychiatric symptoms in a care-seeking sample of U.S. military veterans. Mil Med. 2024;189:e1397-e1402. doi:10.1093/milmed/usae035
- Veterans’ Health Care Act of 1984, 38 USC §98-528 (1984). Accessed March 27, 2026. https://www.congress.gov/bill/98th-congress/house-bill/5618/text
- Veterans’ Health Care Eligibility Reform Act of 1996, 38 USC §104-262 (1996). Accessed March 27, 2026. https://www.congress.gov/bill/104th-congress/house-bill/3118/text
- Military Quality of Life and Veterans Affairs Appropriations Act, 2006. Pub L No. 109-114, 119 Stat. 2372. Accessed March 27, 2026. https://www.congress.gov/bill/109th-congress/house-bill/2528/text
- Karlin BE, Ruzek JI, Chard KM, et al. Dissemination of evidence‐based psychological treatments for posttraumatic stress disorder in the Veterans Health Administration. J Trauma Stress. 2010;23:663-673. doi:10.1002/jts.20588
- Byers AL, Covinsky KE, Barnes DE, et al. Dysthymia and depression increase risk of dementia and mortality among older veterans. Am J Geriatr Psychiatry. 2012;20:664-672. doi:10.1097/JGP.0b013e31822001c1
- Oslin DW, Lynch KG, Shih MC, et al. Effect of pharmacogenomic testing for drug-gene interactions on medication selection and remission of symptoms in major depressive disorder: the PRIME Care randomized clinical trial. JAMA. 2022;328:151-161. doi:10.1001/jama.2022.9805
- Goodman M, Brown GK, Galfalvy HC, et al. Group (“Project Life Force”) versus individual suicide safety planning: a randomized clinical trial. Contemp Clin Trials Commun. 2020;17:100520. doi:10.1016/j.conctc.2020.100520
- Na PJ, Ralevski E, Jegede O, et al. Depression and/or PTSD comorbidity affects response to antidepressants in those with alcohol use disorder. Front Psychiatry. 2022;12:768318. doi:10.3389/fpsyt.2021.768318
- McCarthy JF, Blow FC, Valenstein M, et al. Veterans Affairs Health System and mental health treatment retention among patients with serious mental illness: evaluating accessibility and availability barriers. Health Serv Res. 2007;42:1042-1060. doi:10.1111/j.1475-6773.2006.00642.x
- Glynn SM, Marder SR, Noordsy DL, et al. An RCT evaluating the effects of skills training and medication type on work outcomes among patients with schizophrenia. Psychiatr Serv. 2016;67:500-506. doi:10.1176/appips201500171
- Mayer CL, Savage PJ, Engle CK, et al. Randomized controlled pilot trial of prazosin for prophylaxis of posttraumatic headaches in active-duty service members and veterans. Headache. 2023;63:751-762. doi:10.1111/head.14529
- Hill C. Medical education at VA: it’s all about the veterans. VA News. August 18, 2021. Accessed December 5, 2025. https://news.va.gov/93370/medical-education-at-va-its-all-about-the-veterans/
Accessible and effective mental health services are a vital component of the Veterans Health Administration (VHA) mission to provide exceptional care that improves veterans’ health and well-being. Veterans are seeking mental health care at the VHA at significantly higher rates than in previous years. From 2009 through 2024, the number of veterans who received direct mental health care from the VHA increased 78%.1 The proportion of veterans enrolled in the VHA who also received direct mental health care expanded from 23% of total enrollees in 2009 to 33% in 2024. The increase in VHA mental health care delivery is also reflected in the number of outpatient mental health care and treatment visits at the VHA, which increased from 12.7 million to 21.5 million over the same period.
The Sergeant First Class (SFC) Heath Robinson Honoring Our Promise to Address Comprehensive Toxics (PACT) Act of 2022 expanded eligibility for VHA services, including mental health care and counseling, to new cohorts of toxic-exposed veterans. From 2022 to 2024, > 680,000 veterans enrolled in VHA care, and 45% of those were PACT Act-eligible cohorts.2 Research highlighted the high prevalence of physical and psychiatric comorbidities in toxic-exposed veterans.3 As such, the VHA may face greater demand for direct mental health care with these newly eligible cohorts of veterans.
Veterans often have mental health conditions (eg, depression, posttraumatic stress disorder [PTSD]), substance use disorders (SUDs), unique military experiences (eg, deployments), and injuries and illnesses (eg, traumatic brain injury [TBI]) that increase the complexity of their clinical presentation. The varied nature of these mental health conditions, as well as veterans’ unique military-related experiences, necessitates specialized centers focused on distinct high-priority areas of mental health in the VHA.
A series of public laws charged the US Department of Veterans Affairs (VA) with developing specialized mental health Centers of Excellence (CoEs) focused on high-priority areas of veteran mental health. The first of these laws, Public Law 98- 528, established the National Center for PTSD (NCPTSD), which opened in 1989.4 In 1996, Congress established specialized mental health CoEs known as Mental Illness Research, Education, and Clinical Centers (MIRECCs) across the VHA.5 To address the unique needs of post-9/11 veterans, 3 additional specialized centers were established in 2005.6 Finally, under the authority of the Secretary of the VA, specialized mental health CoEs were established to focus on SUD and integrated health care.
There are 17 geographically diverse mental health CoEs and MIRECCs across the VA (Table). CoEs are embedded in VA medical centers (VAMCs) with strong medical school academic affiliations. Organizational oversight of the CoEs is provided by the VA Office of Mental Health and Office of Suicide Prevention, respectively. As part of the oversight process, CoEs submit annual reports detailing their advancements in research, education and training, and clinical activities, as well as participate in a peer-reviewed renewal process.

These specialized centers are united in a shared tripartite mission to generate new knowledge about the causes and treatments of mental health conditions, to educate and train VHA clinicians and personnel, and to develop and implement innovative clinical programs within the VHA. This combined focus on research, education, and improved clinical care reduces the time from discovery to implementation and improves the health and well-being of veterans.
Examples of this acceleration translation from scientific discovery to clinical practice are evident throughout mental health CoEs. The NCPTSD is a leader in research on PTSD and traumatic stress. Its work led to the development of national training programs for VHA clinicians, facilitating the broad dissemination of efficacious PTSD treatments across the enterprise.7 Researchers at the Veterans Integrated Service Network (VISN) 21 Sierra Pacific MIRECC identified depression as a significant risk factor for dementia and subsequently launched the first multisite trial of repetitive transcranial magnetic stimulation (rTMS) in the VHA in 2012 (CSP #556: the effectiveness of rTMS in depressed VA patients).8 This project laid the groundwork for the national clinical rTMS program launched in 2017, which is now clinically available at 60 VAMCs. In the largest pragmatic randomized clinical trial of its kind, the VISN 4 Philadelphia and Pittsburgh MIRECC found that pharmacogenomic testing significantly reduced the number of prescription medications with predicted drug-gene interactions and improved clinical outcomes among veterans with depression.9
Mental health CoEs are also leaders in suicide prevention, a top clinical priority for the VHA. The VISN 2 New York MIRECC developed Project Life Force, a safety planning skills group for veterans with suicidal ideation, now implemented across 10 VAMCs, including telehealth hubs, outpatient settings, and veteran peer programs.10 The VISN 2 CoE for Suicide Prevention and VISN 19 Rocky Mountain MIRECC coordinate key suicide prevention services for VA, including the analysis of suicide surveillance data; evaluation of national VA suicide prevention initiatives; the support of veterans, families, and clinicians; and enhanced access to evidence-based treatments for at-risk veterans.
Mental health CoEs are a key operational partner in VHA treatment of SUDs. The CoEs in Substance Addiction Treatment and Education (CESATEs) are national resources dedicated to improving the quality, clinical outcomes, and cost-effectiveness of VHA SUD treatment. CESATEs developed and implemented a national rollout of an effective treatment for stimulant use disorders, training staff at > 120 VA programs. The VISN 1 Mid-Atlantic MIRECC’s focus on SUD and comorbid/co-occurring mental health conditions has highlighted the significant prevalence of these conditions and the impact they have on treatment response.11
Serious mental illness (SMI) (eg, schizophrenia, schizoaffective disorder) impacts up to 5% of veterans.12 VISN 22 Desert Pacific MIRECC has developed interventions to improve the lives of veterans with SMI. Its research established supported employment as an effective intervention to improve outcomes in veterans with psychotic disorders and supported its implementation in the VHA.13 Peer specialists are a cornerstone in the VHA commitment to recovery-oriented services for veterans with SMI. VISN 5 Capitol MIRECC has long championed research, clinical training, and educational activities that contributed to the effective deployment of peer specialists across the VHA enterprise.
Veterans have unique military-related experiences (eg, deployment, traumatic stress, transition to civilian status) and injuries and illnesses (eg, TBI, posttraumatic headaches) that significantly impact their mental health and quality of life.
The period between active duty and transition to civilian status is a critical time in a veteran’s life. The VISN 17 CoE Veteran Sponsorship Initiative connects veterans with VA care within 30 days postdischarge, with the option of additional support in the community. The VISN 22 CoE for Stress and Mental Health (CESAMH) develops, evaluates, and disseminates diagnostics and treatments for veterans affected by traumatic events and brain injuries, with a unique focus on supporting their whole health needs. The VISN 6 Mid-Atlantic MIRECC leads the ongoing VISN-6 Post-Deployment Mental Health (PDMH) study, the largest biorepository of post-9/11 veterans. PDMH has greatly expanded the understanding of the unique needs of post-9/11 veterans, with > 100 peer-reviewed publications to date. Veterans with mild TBI frequently experience chronic posttraumatic headaches that can be disabling and nonresponsive to treatment. The VISN 20 Northwest MIRECC demonstrated that prazosin, a repurposed, low-cost, widely available, nonaddictive medication, can safely and effectively reduce the frequency of these headaches and improve functional impairment in veterans and active-duty service members.14
Increased and enhanced access to effective mental health treatment is a priority for VA. In 2007, the VA launched the National Primary Care Mental Health Integration program, which integrated mental health services into primary care settings. The Center for Integrated Healthcare (CIH) has supported the VA in these efforts. In 2024, CIH trained > 5000 health care staff on high-fidelity integration of behavioral health and medical care. VA has also focused on increasing access to mental health services via expanded telehealth offerings. The VISN 16 MIRECC, with its unique focus on increasing access to care for rural veterans via distance-based and digital health technology, supported the VA Offices of Mental Health and Connected Care to virtualize mental health care and promote adoption and sustained use of VA Video Connect across the enterprise.
Specialized MH CoEs are uniquely equipped to support the VHA in providing training and education to VA clinicians, veterans, care partners and family members, and the community on high-priority mental health topics. Education is a core component of the MH CoEs tripartite mission. As such, MH CoEs offer national trainings, conferences, consultation services, clinical demonstration projects, development of clinical dashboards and toolkits, and public awareness campaigns. Researchers, educators, and clinicians at the CoEs frequently serve as subject matter experts on topics aligned with their respective missions. Several national rollout programs that disseminated evidence-based treatments for mental health conditions to the field (eg, cognitive behavioral therapy for depression, cognitive behavioral therapy for insomnia, and prolonged exposure) were developed at specialized CoEs.
The VHA provides advanced training, residencies, and fellowships to > 120,000 trainees annually. Many of these trainees choose to remain at the VA. Seven of 10 VHA psychologists and 6 of 10 VHA physicians trained within the VHA prior to their employment.15 The MH CoEs and MIRECCs play an important role in preparing these trainees for VHA mental health careers. These centers are funded to provide advanced postdoctoral training to physicians as well as allied health professionals in clinical and counseling psychology, social work, pharmacy, and nursing. Training is not limited to postdoctoral fellows: graduate students, residents, and interns from affiliated accredited training programs may rotate through mental health CoEs each academic year.
Conclusions
For > 30 years, mental health CoEs have brought thousands of veterans advanced treatments for their mental health needs and helped reduce death by suicide. The centers have a bright future ahead, harnessing advances in artificial intelligence and genomics to permit the matching of the individual veterans to the treatment most likely to benefit them. Precision medicine, as espoused by the Hannon Act, will not only encourage the efficient use of health care resources but also rapidly reduce pain in veterans with mental health and SUDs.
Accessible and effective mental health services are a vital component of the Veterans Health Administration (VHA) mission to provide exceptional care that improves veterans’ health and well-being. Veterans are seeking mental health care at the VHA at significantly higher rates than in previous years. From 2009 through 2024, the number of veterans who received direct mental health care from the VHA increased 78%.1 The proportion of veterans enrolled in the VHA who also received direct mental health care expanded from 23% of total enrollees in 2009 to 33% in 2024. The increase in VHA mental health care delivery is also reflected in the number of outpatient mental health care and treatment visits at the VHA, which increased from 12.7 million to 21.5 million over the same period.
The Sergeant First Class (SFC) Heath Robinson Honoring Our Promise to Address Comprehensive Toxics (PACT) Act of 2022 expanded eligibility for VHA services, including mental health care and counseling, to new cohorts of toxic-exposed veterans. From 2022 to 2024, > 680,000 veterans enrolled in VHA care, and 45% of those were PACT Act-eligible cohorts.2 Research highlighted the high prevalence of physical and psychiatric comorbidities in toxic-exposed veterans.3 As such, the VHA may face greater demand for direct mental health care with these newly eligible cohorts of veterans.
Veterans often have mental health conditions (eg, depression, posttraumatic stress disorder [PTSD]), substance use disorders (SUDs), unique military experiences (eg, deployments), and injuries and illnesses (eg, traumatic brain injury [TBI]) that increase the complexity of their clinical presentation. The varied nature of these mental health conditions, as well as veterans’ unique military-related experiences, necessitates specialized centers focused on distinct high-priority areas of mental health in the VHA.
A series of public laws charged the US Department of Veterans Affairs (VA) with developing specialized mental health Centers of Excellence (CoEs) focused on high-priority areas of veteran mental health. The first of these laws, Public Law 98- 528, established the National Center for PTSD (NCPTSD), which opened in 1989.4 In 1996, Congress established specialized mental health CoEs known as Mental Illness Research, Education, and Clinical Centers (MIRECCs) across the VHA.5 To address the unique needs of post-9/11 veterans, 3 additional specialized centers were established in 2005.6 Finally, under the authority of the Secretary of the VA, specialized mental health CoEs were established to focus on SUD and integrated health care.
There are 17 geographically diverse mental health CoEs and MIRECCs across the VA (Table). CoEs are embedded in VA medical centers (VAMCs) with strong medical school academic affiliations. Organizational oversight of the CoEs is provided by the VA Office of Mental Health and Office of Suicide Prevention, respectively. As part of the oversight process, CoEs submit annual reports detailing their advancements in research, education and training, and clinical activities, as well as participate in a peer-reviewed renewal process.

These specialized centers are united in a shared tripartite mission to generate new knowledge about the causes and treatments of mental health conditions, to educate and train VHA clinicians and personnel, and to develop and implement innovative clinical programs within the VHA. This combined focus on research, education, and improved clinical care reduces the time from discovery to implementation and improves the health and well-being of veterans.
Examples of this acceleration translation from scientific discovery to clinical practice are evident throughout mental health CoEs. The NCPTSD is a leader in research on PTSD and traumatic stress. Its work led to the development of national training programs for VHA clinicians, facilitating the broad dissemination of efficacious PTSD treatments across the enterprise.7 Researchers at the Veterans Integrated Service Network (VISN) 21 Sierra Pacific MIRECC identified depression as a significant risk factor for dementia and subsequently launched the first multisite trial of repetitive transcranial magnetic stimulation (rTMS) in the VHA in 2012 (CSP #556: the effectiveness of rTMS in depressed VA patients).8 This project laid the groundwork for the national clinical rTMS program launched in 2017, which is now clinically available at 60 VAMCs. In the largest pragmatic randomized clinical trial of its kind, the VISN 4 Philadelphia and Pittsburgh MIRECC found that pharmacogenomic testing significantly reduced the number of prescription medications with predicted drug-gene interactions and improved clinical outcomes among veterans with depression.9
Mental health CoEs are also leaders in suicide prevention, a top clinical priority for the VHA. The VISN 2 New York MIRECC developed Project Life Force, a safety planning skills group for veterans with suicidal ideation, now implemented across 10 VAMCs, including telehealth hubs, outpatient settings, and veteran peer programs.10 The VISN 2 CoE for Suicide Prevention and VISN 19 Rocky Mountain MIRECC coordinate key suicide prevention services for VA, including the analysis of suicide surveillance data; evaluation of national VA suicide prevention initiatives; the support of veterans, families, and clinicians; and enhanced access to evidence-based treatments for at-risk veterans.
Mental health CoEs are a key operational partner in VHA treatment of SUDs. The CoEs in Substance Addiction Treatment and Education (CESATEs) are national resources dedicated to improving the quality, clinical outcomes, and cost-effectiveness of VHA SUD treatment. CESATEs developed and implemented a national rollout of an effective treatment for stimulant use disorders, training staff at > 120 VA programs. The VISN 1 Mid-Atlantic MIRECC’s focus on SUD and comorbid/co-occurring mental health conditions has highlighted the significant prevalence of these conditions and the impact they have on treatment response.11
Serious mental illness (SMI) (eg, schizophrenia, schizoaffective disorder) impacts up to 5% of veterans.12 VISN 22 Desert Pacific MIRECC has developed interventions to improve the lives of veterans with SMI. Its research established supported employment as an effective intervention to improve outcomes in veterans with psychotic disorders and supported its implementation in the VHA.13 Peer specialists are a cornerstone in the VHA commitment to recovery-oriented services for veterans with SMI. VISN 5 Capitol MIRECC has long championed research, clinical training, and educational activities that contributed to the effective deployment of peer specialists across the VHA enterprise.
Veterans have unique military-related experiences (eg, deployment, traumatic stress, transition to civilian status) and injuries and illnesses (eg, TBI, posttraumatic headaches) that significantly impact their mental health and quality of life.
The period between active duty and transition to civilian status is a critical time in a veteran’s life. The VISN 17 CoE Veteran Sponsorship Initiative connects veterans with VA care within 30 days postdischarge, with the option of additional support in the community. The VISN 22 CoE for Stress and Mental Health (CESAMH) develops, evaluates, and disseminates diagnostics and treatments for veterans affected by traumatic events and brain injuries, with a unique focus on supporting their whole health needs. The VISN 6 Mid-Atlantic MIRECC leads the ongoing VISN-6 Post-Deployment Mental Health (PDMH) study, the largest biorepository of post-9/11 veterans. PDMH has greatly expanded the understanding of the unique needs of post-9/11 veterans, with > 100 peer-reviewed publications to date. Veterans with mild TBI frequently experience chronic posttraumatic headaches that can be disabling and nonresponsive to treatment. The VISN 20 Northwest MIRECC demonstrated that prazosin, a repurposed, low-cost, widely available, nonaddictive medication, can safely and effectively reduce the frequency of these headaches and improve functional impairment in veterans and active-duty service members.14
Increased and enhanced access to effective mental health treatment is a priority for VA. In 2007, the VA launched the National Primary Care Mental Health Integration program, which integrated mental health services into primary care settings. The Center for Integrated Healthcare (CIH) has supported the VA in these efforts. In 2024, CIH trained > 5000 health care staff on high-fidelity integration of behavioral health and medical care. VA has also focused on increasing access to mental health services via expanded telehealth offerings. The VISN 16 MIRECC, with its unique focus on increasing access to care for rural veterans via distance-based and digital health technology, supported the VA Offices of Mental Health and Connected Care to virtualize mental health care and promote adoption and sustained use of VA Video Connect across the enterprise.
Specialized MH CoEs are uniquely equipped to support the VHA in providing training and education to VA clinicians, veterans, care partners and family members, and the community on high-priority mental health topics. Education is a core component of the MH CoEs tripartite mission. As such, MH CoEs offer national trainings, conferences, consultation services, clinical demonstration projects, development of clinical dashboards and toolkits, and public awareness campaigns. Researchers, educators, and clinicians at the CoEs frequently serve as subject matter experts on topics aligned with their respective missions. Several national rollout programs that disseminated evidence-based treatments for mental health conditions to the field (eg, cognitive behavioral therapy for depression, cognitive behavioral therapy for insomnia, and prolonged exposure) were developed at specialized CoEs.
The VHA provides advanced training, residencies, and fellowships to > 120,000 trainees annually. Many of these trainees choose to remain at the VA. Seven of 10 VHA psychologists and 6 of 10 VHA physicians trained within the VHA prior to their employment.15 The MH CoEs and MIRECCs play an important role in preparing these trainees for VHA mental health careers. These centers are funded to provide advanced postdoctoral training to physicians as well as allied health professionals in clinical and counseling psychology, social work, pharmacy, and nursing. Training is not limited to postdoctoral fellows: graduate students, residents, and interns from affiliated accredited training programs may rotate through mental health CoEs each academic year.
Conclusions
For > 30 years, mental health CoEs have brought thousands of veterans advanced treatments for their mental health needs and helped reduce death by suicide. The centers have a bright future ahead, harnessing advances in artificial intelligence and genomics to permit the matching of the individual veterans to the treatment most likely to benefit them. Precision medicine, as espoused by the Hannon Act, will not only encourage the efficient use of health care resources but also rapidly reduce pain in veterans with mental health and SUDs.
- Congressionally Mandated Report: Report on Transparency in Mental Health Care Services. US Dept of Veterans Affairs; December 2022. Accessed December 5, 2025. https://www.govinfo.gov/content/pkg/CMR-VA1-00181657/pdf/CMR-VA1-00181657.pdf
- Beckman AL, Jacobs J, Elnahal SM. The PACT Act—expanding coverage and access for veterans. JAMA. 2024;332:1423-1424. doi:10.1001/jama.2024.16013
- Morse JL, Acheson DT, Almklov E, et al. Associations among environmental exposures and physical and psychiatric symptoms in a care-seeking sample of U.S. military veterans. Mil Med. 2024;189:e1397-e1402. doi:10.1093/milmed/usae035
- Veterans’ Health Care Act of 1984, 38 USC §98-528 (1984). Accessed March 27, 2026. https://www.congress.gov/bill/98th-congress/house-bill/5618/text
- Veterans’ Health Care Eligibility Reform Act of 1996, 38 USC §104-262 (1996). Accessed March 27, 2026. https://www.congress.gov/bill/104th-congress/house-bill/3118/text
- Military Quality of Life and Veterans Affairs Appropriations Act, 2006. Pub L No. 109-114, 119 Stat. 2372. Accessed March 27, 2026. https://www.congress.gov/bill/109th-congress/house-bill/2528/text
- Karlin BE, Ruzek JI, Chard KM, et al. Dissemination of evidence‐based psychological treatments for posttraumatic stress disorder in the Veterans Health Administration. J Trauma Stress. 2010;23:663-673. doi:10.1002/jts.20588
- Byers AL, Covinsky KE, Barnes DE, et al. Dysthymia and depression increase risk of dementia and mortality among older veterans. Am J Geriatr Psychiatry. 2012;20:664-672. doi:10.1097/JGP.0b013e31822001c1
- Oslin DW, Lynch KG, Shih MC, et al. Effect of pharmacogenomic testing for drug-gene interactions on medication selection and remission of symptoms in major depressive disorder: the PRIME Care randomized clinical trial. JAMA. 2022;328:151-161. doi:10.1001/jama.2022.9805
- Goodman M, Brown GK, Galfalvy HC, et al. Group (“Project Life Force”) versus individual suicide safety planning: a randomized clinical trial. Contemp Clin Trials Commun. 2020;17:100520. doi:10.1016/j.conctc.2020.100520
- Na PJ, Ralevski E, Jegede O, et al. Depression and/or PTSD comorbidity affects response to antidepressants in those with alcohol use disorder. Front Psychiatry. 2022;12:768318. doi:10.3389/fpsyt.2021.768318
- McCarthy JF, Blow FC, Valenstein M, et al. Veterans Affairs Health System and mental health treatment retention among patients with serious mental illness: evaluating accessibility and availability barriers. Health Serv Res. 2007;42:1042-1060. doi:10.1111/j.1475-6773.2006.00642.x
- Glynn SM, Marder SR, Noordsy DL, et al. An RCT evaluating the effects of skills training and medication type on work outcomes among patients with schizophrenia. Psychiatr Serv. 2016;67:500-506. doi:10.1176/appips201500171
- Mayer CL, Savage PJ, Engle CK, et al. Randomized controlled pilot trial of prazosin for prophylaxis of posttraumatic headaches in active-duty service members and veterans. Headache. 2023;63:751-762. doi:10.1111/head.14529
- Hill C. Medical education at VA: it’s all about the veterans. VA News. August 18, 2021. Accessed December 5, 2025. https://news.va.gov/93370/medical-education-at-va-its-all-about-the-veterans/
- Congressionally Mandated Report: Report on Transparency in Mental Health Care Services. US Dept of Veterans Affairs; December 2022. Accessed December 5, 2025. https://www.govinfo.gov/content/pkg/CMR-VA1-00181657/pdf/CMR-VA1-00181657.pdf
- Beckman AL, Jacobs J, Elnahal SM. The PACT Act—expanding coverage and access for veterans. JAMA. 2024;332:1423-1424. doi:10.1001/jama.2024.16013
- Morse JL, Acheson DT, Almklov E, et al. Associations among environmental exposures and physical and psychiatric symptoms in a care-seeking sample of U.S. military veterans. Mil Med. 2024;189:e1397-e1402. doi:10.1093/milmed/usae035
- Veterans’ Health Care Act of 1984, 38 USC §98-528 (1984). Accessed March 27, 2026. https://www.congress.gov/bill/98th-congress/house-bill/5618/text
- Veterans’ Health Care Eligibility Reform Act of 1996, 38 USC §104-262 (1996). Accessed March 27, 2026. https://www.congress.gov/bill/104th-congress/house-bill/3118/text
- Military Quality of Life and Veterans Affairs Appropriations Act, 2006. Pub L No. 109-114, 119 Stat. 2372. Accessed March 27, 2026. https://www.congress.gov/bill/109th-congress/house-bill/2528/text
- Karlin BE, Ruzek JI, Chard KM, et al. Dissemination of evidence‐based psychological treatments for posttraumatic stress disorder in the Veterans Health Administration. J Trauma Stress. 2010;23:663-673. doi:10.1002/jts.20588
- Byers AL, Covinsky KE, Barnes DE, et al. Dysthymia and depression increase risk of dementia and mortality among older veterans. Am J Geriatr Psychiatry. 2012;20:664-672. doi:10.1097/JGP.0b013e31822001c1
- Oslin DW, Lynch KG, Shih MC, et al. Effect of pharmacogenomic testing for drug-gene interactions on medication selection and remission of symptoms in major depressive disorder: the PRIME Care randomized clinical trial. JAMA. 2022;328:151-161. doi:10.1001/jama.2022.9805
- Goodman M, Brown GK, Galfalvy HC, et al. Group (“Project Life Force”) versus individual suicide safety planning: a randomized clinical trial. Contemp Clin Trials Commun. 2020;17:100520. doi:10.1016/j.conctc.2020.100520
- Na PJ, Ralevski E, Jegede O, et al. Depression and/or PTSD comorbidity affects response to antidepressants in those with alcohol use disorder. Front Psychiatry. 2022;12:768318. doi:10.3389/fpsyt.2021.768318
- McCarthy JF, Blow FC, Valenstein M, et al. Veterans Affairs Health System and mental health treatment retention among patients with serious mental illness: evaluating accessibility and availability barriers. Health Serv Res. 2007;42:1042-1060. doi:10.1111/j.1475-6773.2006.00642.x
- Glynn SM, Marder SR, Noordsy DL, et al. An RCT evaluating the effects of skills training and medication type on work outcomes among patients with schizophrenia. Psychiatr Serv. 2016;67:500-506. doi:10.1176/appips201500171
- Mayer CL, Savage PJ, Engle CK, et al. Randomized controlled pilot trial of prazosin for prophylaxis of posttraumatic headaches in active-duty service members and veterans. Headache. 2023;63:751-762. doi:10.1111/head.14529
- Hill C. Medical education at VA: it’s all about the veterans. VA News. August 18, 2021. Accessed December 5, 2025. https://news.va.gov/93370/medical-education-at-va-its-all-about-the-veterans/
Meeting the Needs of Those Who Have Served: The Role of VHA Specialized Mental Health Centers of Excellence
Meeting the Needs of Those Who Have Served: The Role of VHA Specialized Mental Health Centers of Excellence
Optimizing Patch Testing in Clinical Practice: Insights From Amber Reck Atwater, MD
Optimizing Patch Testing in Clinical Practice: Insights From Amber Reck Atwater, MD
What evidence exists on the impact of systemic immunosuppressants, biologics, and small-molecule inhibitors on patch test sensitivity and specificity?
DR. ATWATER: Guidance on this topic recently was published by the North American Contact Dermatitis Group (NACDG) in the Journal of the American Academy of Dermatology in June 2025. The article outlined expert recommendations on whether systemic immunosuppressants, biologics, and small-molecule inhibitors should be held before patch testing, how long they should be withheld, and the maximum recommended doses that can be used during testing.
How can dermatologists perform patch testing or use alternative diagnostic strategies when systemic therapy cannot safely be withheld?
DR. ATWATER: When systemic therapy cannot safely be withheld and patch testing is needed for diagnostic purposes, I typically proceed with the understanding that there is risk of false-negative reactions. If the patient has dermatitis on systemic therapy, it suggests that an allergic response on patch testing is also possible. I generally follow the NACDG guidelines mentioned above, and I hold systemic medications during the week of testing, when possible. If the patient has diffuse dermatitis on systemic therapy and their skin is not clear enough to proceed, I prescribe a prednisone taper and patch test on 10 mg per day for the entire week of testing. In patients taking systemic medications, I typically consider doubtful (+/−) patch test reactions to be the equivalent of a positive (1+) reaction.
One alternative diagnostic strategy is to create a safe list that avoids common allergens and have the patient use only products on this list. If their skin clears with avoidance, it suggests that they may have a contact allergy, and you can proceed with patch testing.
In patients with a convincing history of contact dermatitis but a negative patch test, what are the most common causes of false negatives, and how do you distinguish those from true negatives?
DR. ATWATER: In this setting, the most common cause of a false-negative patch test is not testing the correct allergens. This may occur when too few allergens are tested or when relevant allergens are not tested. Other potential causes of false negatives are incorrect timing of allergen exposure and readings, inadequate allergen adherence, expired allergens, and testing with the incorrect vehicle or concentration of allergen. Some immunosuppressant medications also can cause a false-negative patch test. The only way to distinguish false negatives from true negatives is to be aware of these potential pitfalls and continuously work to avoid errors whenever possible.
What technical and practical factors most influence false negatives/positives, and what steps do you recommend to standardize and improve test yield?
DR. ATWATER: Not testing the correct allergens is a potential pitfall in patch testing. For example, when comparing the 35 allergens in the T.R.U.E. Test (thin layer rapid-use epicutaneous test) to the 80 allergens tested by the NACDG in 2021 to 2022, up to 48% of NACDG allergens are missed when testing with only the T.R.U.E. Test. This argues for comprehensive patch testing and testing of at least 80 to 90 allergens whenever possible. Another example is failure to test allergens relevant to occupational exposures, such as in the case of a hairdresser or nail technician. When patches are not applied for the correct period (48 hours) and the final reading isn’t completed in the recommended timeframe (96 to 168 hours), there is increased risk for false negatives and positives. Both false negatives and positives can occur if you complete your final reading too early, whereas false negatives may be more likely if you complete your final reading late. Poor allergen adherence, which can be caused by hair, sweat, poor tape application, water, and exercise, also can result in false negatives. Allergen concentration that is too low to elicit a reaction, as well as too little allergen placed into the chamber, also could increase the risk of false negatives. Too much allergen in the chamber or too high a concentration of the allergen can result in false positives.
Topical medications applied to the patch test site prior to or during patch testing, as well as phototherapy, sun exposure, intramuscular triamcinolone, immunosuppressants, biologics, and small-molecule inhibitors can suppress the immune response to allergen exposure.
To identify true positive patch test reactions, use side lighting and palpate the skin. Be aware of the appearance of irritant reactions, patch test reaction variants such as follicular reactions, and the poral reaction, which can be seen with cobalt. Strong knowledge of how to read patch test reactions will decrease your risk of false-positive and -negative reactions. Training and protocols are vital for standardization and accurate patch testing. We train our staff on the technicalities of patch testing and utilize patch test orders and checklists in our office. We take photos to confirm application sites and visually track reactions between visits. We also provide verbal and written patch test care instructions for our patients and reinforce instructions at each clinic visit.
What are your top practical tips for dermatologists to maximize diagnostic accuracy and patient safety?
DR. ATWATER: My first tip is to develop patch test protocols that are followed by staff and physicians—every time—for every patient. My second tip is to make sure you understand and are comfortable with the patch test process. There are several great patch test resources that can help, including Introduction to Patch Testing, a recently developed CME module in the AAD Learning Center (https://learning.aad.org/Listing/Introduction-to-Patch- Testing-20366). There also are patch test training courses and other resources offered by the American Contact Dermatitis Society (https://www.contactderm.org/).
What single guideline change would most improve patch testing?
DR. ATWATER: The single guideline change that would most improve the practice of patch testing is removal of payer limitations on the number of patches that can be applied per day. For many payers in the United States, this limit is 80 to 90 allergens, and it is sometimes lower. Limits on the number of allergens that can be applied per day may result in false-negative patch testing (when limits are applied), patient inconvenience (if testing is completed over 2 different application days), and insufficient reimbursement (if more allergens are tested than the limit allows).
What evidence exists on the impact of systemic immunosuppressants, biologics, and small-molecule inhibitors on patch test sensitivity and specificity?
DR. ATWATER: Guidance on this topic recently was published by the North American Contact Dermatitis Group (NACDG) in the Journal of the American Academy of Dermatology in June 2025. The article outlined expert recommendations on whether systemic immunosuppressants, biologics, and small-molecule inhibitors should be held before patch testing, how long they should be withheld, and the maximum recommended doses that can be used during testing.
How can dermatologists perform patch testing or use alternative diagnostic strategies when systemic therapy cannot safely be withheld?
DR. ATWATER: When systemic therapy cannot safely be withheld and patch testing is needed for diagnostic purposes, I typically proceed with the understanding that there is risk of false-negative reactions. If the patient has dermatitis on systemic therapy, it suggests that an allergic response on patch testing is also possible. I generally follow the NACDG guidelines mentioned above, and I hold systemic medications during the week of testing, when possible. If the patient has diffuse dermatitis on systemic therapy and their skin is not clear enough to proceed, I prescribe a prednisone taper and patch test on 10 mg per day for the entire week of testing. In patients taking systemic medications, I typically consider doubtful (+/−) patch test reactions to be the equivalent of a positive (1+) reaction.
One alternative diagnostic strategy is to create a safe list that avoids common allergens and have the patient use only products on this list. If their skin clears with avoidance, it suggests that they may have a contact allergy, and you can proceed with patch testing.
In patients with a convincing history of contact dermatitis but a negative patch test, what are the most common causes of false negatives, and how do you distinguish those from true negatives?
DR. ATWATER: In this setting, the most common cause of a false-negative patch test is not testing the correct allergens. This may occur when too few allergens are tested or when relevant allergens are not tested. Other potential causes of false negatives are incorrect timing of allergen exposure and readings, inadequate allergen adherence, expired allergens, and testing with the incorrect vehicle or concentration of allergen. Some immunosuppressant medications also can cause a false-negative patch test. The only way to distinguish false negatives from true negatives is to be aware of these potential pitfalls and continuously work to avoid errors whenever possible.
What technical and practical factors most influence false negatives/positives, and what steps do you recommend to standardize and improve test yield?
DR. ATWATER: Not testing the correct allergens is a potential pitfall in patch testing. For example, when comparing the 35 allergens in the T.R.U.E. Test (thin layer rapid-use epicutaneous test) to the 80 allergens tested by the NACDG in 2021 to 2022, up to 48% of NACDG allergens are missed when testing with only the T.R.U.E. Test. This argues for comprehensive patch testing and testing of at least 80 to 90 allergens whenever possible. Another example is failure to test allergens relevant to occupational exposures, such as in the case of a hairdresser or nail technician. When patches are not applied for the correct period (48 hours) and the final reading isn’t completed in the recommended timeframe (96 to 168 hours), there is increased risk for false negatives and positives. Both false negatives and positives can occur if you complete your final reading too early, whereas false negatives may be more likely if you complete your final reading late. Poor allergen adherence, which can be caused by hair, sweat, poor tape application, water, and exercise, also can result in false negatives. Allergen concentration that is too low to elicit a reaction, as well as too little allergen placed into the chamber, also could increase the risk of false negatives. Too much allergen in the chamber or too high a concentration of the allergen can result in false positives.
Topical medications applied to the patch test site prior to or during patch testing, as well as phototherapy, sun exposure, intramuscular triamcinolone, immunosuppressants, biologics, and small-molecule inhibitors can suppress the immune response to allergen exposure.
To identify true positive patch test reactions, use side lighting and palpate the skin. Be aware of the appearance of irritant reactions, patch test reaction variants such as follicular reactions, and the poral reaction, which can be seen with cobalt. Strong knowledge of how to read patch test reactions will decrease your risk of false-positive and -negative reactions. Training and protocols are vital for standardization and accurate patch testing. We train our staff on the technicalities of patch testing and utilize patch test orders and checklists in our office. We take photos to confirm application sites and visually track reactions between visits. We also provide verbal and written patch test care instructions for our patients and reinforce instructions at each clinic visit.
What are your top practical tips for dermatologists to maximize diagnostic accuracy and patient safety?
DR. ATWATER: My first tip is to develop patch test protocols that are followed by staff and physicians—every time—for every patient. My second tip is to make sure you understand and are comfortable with the patch test process. There are several great patch test resources that can help, including Introduction to Patch Testing, a recently developed CME module in the AAD Learning Center (https://learning.aad.org/Listing/Introduction-to-Patch- Testing-20366). There also are patch test training courses and other resources offered by the American Contact Dermatitis Society (https://www.contactderm.org/).
What single guideline change would most improve patch testing?
DR. ATWATER: The single guideline change that would most improve the practice of patch testing is removal of payer limitations on the number of patches that can be applied per day. For many payers in the United States, this limit is 80 to 90 allergens, and it is sometimes lower. Limits on the number of allergens that can be applied per day may result in false-negative patch testing (when limits are applied), patient inconvenience (if testing is completed over 2 different application days), and insufficient reimbursement (if more allergens are tested than the limit allows).
What evidence exists on the impact of systemic immunosuppressants, biologics, and small-molecule inhibitors on patch test sensitivity and specificity?
DR. ATWATER: Guidance on this topic recently was published by the North American Contact Dermatitis Group (NACDG) in the Journal of the American Academy of Dermatology in June 2025. The article outlined expert recommendations on whether systemic immunosuppressants, biologics, and small-molecule inhibitors should be held before patch testing, how long they should be withheld, and the maximum recommended doses that can be used during testing.
How can dermatologists perform patch testing or use alternative diagnostic strategies when systemic therapy cannot safely be withheld?
DR. ATWATER: When systemic therapy cannot safely be withheld and patch testing is needed for diagnostic purposes, I typically proceed with the understanding that there is risk of false-negative reactions. If the patient has dermatitis on systemic therapy, it suggests that an allergic response on patch testing is also possible. I generally follow the NACDG guidelines mentioned above, and I hold systemic medications during the week of testing, when possible. If the patient has diffuse dermatitis on systemic therapy and their skin is not clear enough to proceed, I prescribe a prednisone taper and patch test on 10 mg per day for the entire week of testing. In patients taking systemic medications, I typically consider doubtful (+/−) patch test reactions to be the equivalent of a positive (1+) reaction.
One alternative diagnostic strategy is to create a safe list that avoids common allergens and have the patient use only products on this list. If their skin clears with avoidance, it suggests that they may have a contact allergy, and you can proceed with patch testing.
In patients with a convincing history of contact dermatitis but a negative patch test, what are the most common causes of false negatives, and how do you distinguish those from true negatives?
DR. ATWATER: In this setting, the most common cause of a false-negative patch test is not testing the correct allergens. This may occur when too few allergens are tested or when relevant allergens are not tested. Other potential causes of false negatives are incorrect timing of allergen exposure and readings, inadequate allergen adherence, expired allergens, and testing with the incorrect vehicle or concentration of allergen. Some immunosuppressant medications also can cause a false-negative patch test. The only way to distinguish false negatives from true negatives is to be aware of these potential pitfalls and continuously work to avoid errors whenever possible.
What technical and practical factors most influence false negatives/positives, and what steps do you recommend to standardize and improve test yield?
DR. ATWATER: Not testing the correct allergens is a potential pitfall in patch testing. For example, when comparing the 35 allergens in the T.R.U.E. Test (thin layer rapid-use epicutaneous test) to the 80 allergens tested by the NACDG in 2021 to 2022, up to 48% of NACDG allergens are missed when testing with only the T.R.U.E. Test. This argues for comprehensive patch testing and testing of at least 80 to 90 allergens whenever possible. Another example is failure to test allergens relevant to occupational exposures, such as in the case of a hairdresser or nail technician. When patches are not applied for the correct period (48 hours) and the final reading isn’t completed in the recommended timeframe (96 to 168 hours), there is increased risk for false negatives and positives. Both false negatives and positives can occur if you complete your final reading too early, whereas false negatives may be more likely if you complete your final reading late. Poor allergen adherence, which can be caused by hair, sweat, poor tape application, water, and exercise, also can result in false negatives. Allergen concentration that is too low to elicit a reaction, as well as too little allergen placed into the chamber, also could increase the risk of false negatives. Too much allergen in the chamber or too high a concentration of the allergen can result in false positives.
Topical medications applied to the patch test site prior to or during patch testing, as well as phototherapy, sun exposure, intramuscular triamcinolone, immunosuppressants, biologics, and small-molecule inhibitors can suppress the immune response to allergen exposure.
To identify true positive patch test reactions, use side lighting and palpate the skin. Be aware of the appearance of irritant reactions, patch test reaction variants such as follicular reactions, and the poral reaction, which can be seen with cobalt. Strong knowledge of how to read patch test reactions will decrease your risk of false-positive and -negative reactions. Training and protocols are vital for standardization and accurate patch testing. We train our staff on the technicalities of patch testing and utilize patch test orders and checklists in our office. We take photos to confirm application sites and visually track reactions between visits. We also provide verbal and written patch test care instructions for our patients and reinforce instructions at each clinic visit.
What are your top practical tips for dermatologists to maximize diagnostic accuracy and patient safety?
DR. ATWATER: My first tip is to develop patch test protocols that are followed by staff and physicians—every time—for every patient. My second tip is to make sure you understand and are comfortable with the patch test process. There are several great patch test resources that can help, including Introduction to Patch Testing, a recently developed CME module in the AAD Learning Center (https://learning.aad.org/Listing/Introduction-to-Patch- Testing-20366). There also are patch test training courses and other resources offered by the American Contact Dermatitis Society (https://www.contactderm.org/).
What single guideline change would most improve patch testing?
DR. ATWATER: The single guideline change that would most improve the practice of patch testing is removal of payer limitations on the number of patches that can be applied per day. For many payers in the United States, this limit is 80 to 90 allergens, and it is sometimes lower. Limits on the number of allergens that can be applied per day may result in false-negative patch testing (when limits are applied), patient inconvenience (if testing is completed over 2 different application days), and insufficient reimbursement (if more allergens are tested than the limit allows).
Optimizing Patch Testing in Clinical Practice: Insights From Amber Reck Atwater, MD
Optimizing Patch Testing in Clinical Practice: Insights From Amber Reck Atwater, MD
Dupilumab for Bullous Pemphigoid: To Treat or Not to Treat?
Dupilumab for Bullous Pemphigoid: To Treat or Not to Treat?
Bullous pemphigoid (BP) is a chronic autoimmune skin condition characterized by large, fluid-filled blisters (bullae) on the skin. There usually is severe itching, which may antedate the bullae by several weeks. Bullous pemphigoid most commonly affects adults aged 60 years and older with comorbidities such as metabolic syndrome or other autoimmune diseases. The typical blistering is subepidermal, and its occurrence seems to be driven by autoantibodies targeting the epidermal basement membrane zone.
Conventional treatment for BP has long included systemic corticosteroids and immunosuppressants; this combination of drugs effectively controls inflammation but also is associated with considerable risks such as infection, hyperglycemia with worsening diabetes, hypertension, osteoporosis and fracture risk, and delirium, particularly in older patients. Recent developments in biologic therapy have supported the utilization of potentially safer alternatives. Specifically, dupilumab has demonstrated efficacy in the treatment of atopic dermatitis. Case reports and small research studies included in a recent review have suggested that dupilumab could be effective in managing BP, potentially offering a safer treatment option.1 Importantly, dupilumab recently was approved for BP by the US Food and Drug Administration, making it the first biologic approved for treating this condition.2 In this article, we discuss dupilumab as an option for clinicians to prescribe as an initial therapy for BP.
Pathophysiology of Bullous Pemphigoid and Dupilumab
Bullous pemphigoid pathophysiology is driven by 2 main components: immunologic and inflammatory processes. The immune-mediated attack consists of autoantibodies against the hemidesmosomal proteins BP180 and BP230 in the basement membrane zone, which anchor the epidermis to the dermis.1 When these proteins are targeted, a cascade of responses is triggered, including the release of inflammatory cells, particularly eosinophils and neutrophils, into the skin. These cells release inflammatory mediators that break down the basement membrane, resulting in the separation of the epidermis from the dermis and leading to subepidermal blister formation.
The inflammation associated with BP largely is driven by a T helper 2–dominant immune response, with increased levels of cytokines such as interleukin (IL) 4 and IL-13.1 The chronic inflammation contributes to the characteristic blisters and intense pruritus seen in patients with BP. Traditional treatments such as corticosteroids work broadly to suppress this inflammation by reducing the production of all proinflammatory cytokines; however, they lack specificity, which dampens the targeted immune response against BP and broadly suppresses the immune system, increasing the risk for infection and other adverse effects. For older patients with multiple health conditions, these adverse effects can impact quality of life and pose health risks.
Interleukin 4 and IL-13 contribute to eosinophilia by upregulating the expression of eosinophil chemoattractant cytokines that promote eosinophil migration and adhesion.1 This is where dupilumab presents a promising alternative. Dupilumab is a monoclonal antibody that selectively targets the key cytokines implicated in BP. It binds to the IL-4 receptor alpha subunit, which IL-4 and IL-13 share, thereby inhibiting the signaling of both cytokines.1
Dupilumab Reports and Case Series
Current evidence supporting the use of dupilumab for BP consists primarily of case series and uncontrolled clinical observations, both of which are useful to formulate but not to test hypotheses.3 One case series of 13 patients diagnosed with BP reported that 92.3% (12/13) of patients achieved disease clearance or satisfactory control, with 53.8% (7/13) achieving total lesion clearance and no adverse events observed.4 In a larger case series, 87.0% of 146 patients achieved disease control with an initial dose of dupilumab 600 mg followed by 300 mg every 2 weeks for 4 weeks, though the study also looked at secondary outcomes for up to 64 weeks.5 Both these findings are compatible with the idea that dupilumab has efficacy in BP, but they cannot be used to test hypotheses.3,4
In a single case report of refractory BP, the combination of dupilumab and omalizumab—a monoclonal antibody that blocks the binding of IgE to mast cells, basophils, and dendritic cells—has shown efficacy, particularly when BP is unresponsive to standard therapies.6 The case involved a 72-year-old woman with unresponsive BP who was treated with a combination of dupilumab and omalizumab. She experienced marked improvement in pruritus and bullae accompanied by normalization of anti-BP180 autoantibody levels. This result points to the possibility that IL-4 and IL-13 blockade could influence autoantibody production. The authors attributed the reduction in pruritus to dupilumab, although the drug was administered in combination with omalizumab.6
Small Randomized Trial of Dupilumab
In addition to these uncontrolled clinical observations, dupilumab recently demonstrated positive results in a small randomized trial.7 Patients who were assigned to receive dupilumab were 5 times more likely to achieve sustained remission compared to those assigned to a placebo. All randomized patients also were treated with a standard-of-care oral corticosteroid regimen, which complicated the ability to isolate the specific effects of dupilumab.7 Finally, a completed totality of evidence on the prescription of dupilumab for refractory BP should include more reliable results from larger randomized trials.8
Final Thoughts
Dupilumab is now approved by the US Food and Drug Administration for the treatment of BP, marking an important milestone as the first targeted biologic therapy for this condition. While the totality of evidence is still evolving, the available data indicate both efficacy and a favorable safety profile when compared to traditional corticosteroids and immunosuppressants. Based on the available data, we suggest that health care providers consider dupilumab as a first-line monotherapy in appropriate patients with BP without specific contraindications, particularly as a practical and safe alternative to corticosteroids and immunosuppressive therapies in older adults with comorbidities. If the patient demonstrates clinical improvement in symptom relief, the therapy can be continued. This strategy minimizes initial exposure to more potent biologics or therapies with higher toxicity profiles. For patients without adequate disease control or with severe initial presentations requiring rapid improvement, clinicians may consider escalation to adjunctive or alternative therapies, such as rituximab, omalizumab, or a short course of systemic corticosteroids for acute management.
Emerging data suggest that dupilumab is a promising therapy for BP. We suggest that it may be a safer targeted alternative to traditional corticosteroids and immunosuppressive therapies. The ability of dupilumab to mitigate inflammation without broadly suppressing the immune system suggests that it may turn out to be an especially valuable option for older patients and those with comorbid conditions.
Acknowledgement—The authors thank Jodi Fiedler, MD (Boca Raton, FL), for her expert advice and assistance.
- Zhang L, Chen Z, Wang L, et al. Bullous pemphigoid: the role of type 2 inflammation in its pathogenesis and the prospect of targeted therapy. Front Immunol. 2023;14:1115083. doi:10.3389/fimmu.2023.1115083
- Sanofi. Dupixent approved in the US as the only targeted medicine to treat patients with bullous pemphigoid. News release. June 20, 2025. Accessed September 8, 2025. https://www.sanofi.com/en/media-room/press-releases/2025/2025-06-20-05-00-00-3102518
- Hennekens CH, DeMets D. Statistical association and causation: contributions of different types of evidence. JAMA. 2011;306:1134-1136. doi:10.1001/jama.2011.322
- Abdat R, Waldman RA, de Bedout V, et al. Dupilumab as a novel therapy for bullous pemphigoid: a multicenter case series. J Am Acad Dermatol. 2020;83:46-52. doi:10.1016/j.jaad.2020.01.089
- Zhao L, Wang Q, Liang G, et al. Evaluation of dupilumab in patients with bullous pemphigoid. JAMA Dermatol. 2023;159:953-960. doi:10.1001/jamadermatol.2023.2428
- Seyed Jafari SM, Feldmeyer L, Bossart S, et al. Case report: combination of omalizumab and dupilumab for recalcitrant bullous pemphigoid. Front Immunol. 2021;11:611549. doi:10.3389/fimmu.2020.611549
- US National Library of Medicine. A multicenter, randomized, double-blind, placebo-controlled, parallel-group study to evaluate the efficacy and safety of dupilumab in adult patients with bullous pemphigoid. ClinicalTrials.gov. Published 2019. Accessed March 10, 2026. https://clinicaltrials.gov/ct2/show/NCT04206553
- Hennekens CH, DeMets D. The need for large scale randomized evidence without undue emphasis on small trials, meta-analyses or subgroup analyses. JAMA. 2009;302:2361-2362. doi:10.1001/jama.2009.1829
Bullous pemphigoid (BP) is a chronic autoimmune skin condition characterized by large, fluid-filled blisters (bullae) on the skin. There usually is severe itching, which may antedate the bullae by several weeks. Bullous pemphigoid most commonly affects adults aged 60 years and older with comorbidities such as metabolic syndrome or other autoimmune diseases. The typical blistering is subepidermal, and its occurrence seems to be driven by autoantibodies targeting the epidermal basement membrane zone.
Conventional treatment for BP has long included systemic corticosteroids and immunosuppressants; this combination of drugs effectively controls inflammation but also is associated with considerable risks such as infection, hyperglycemia with worsening diabetes, hypertension, osteoporosis and fracture risk, and delirium, particularly in older patients. Recent developments in biologic therapy have supported the utilization of potentially safer alternatives. Specifically, dupilumab has demonstrated efficacy in the treatment of atopic dermatitis. Case reports and small research studies included in a recent review have suggested that dupilumab could be effective in managing BP, potentially offering a safer treatment option.1 Importantly, dupilumab recently was approved for BP by the US Food and Drug Administration, making it the first biologic approved for treating this condition.2 In this article, we discuss dupilumab as an option for clinicians to prescribe as an initial therapy for BP.
Pathophysiology of Bullous Pemphigoid and Dupilumab
Bullous pemphigoid pathophysiology is driven by 2 main components: immunologic and inflammatory processes. The immune-mediated attack consists of autoantibodies against the hemidesmosomal proteins BP180 and BP230 in the basement membrane zone, which anchor the epidermis to the dermis.1 When these proteins are targeted, a cascade of responses is triggered, including the release of inflammatory cells, particularly eosinophils and neutrophils, into the skin. These cells release inflammatory mediators that break down the basement membrane, resulting in the separation of the epidermis from the dermis and leading to subepidermal blister formation.
The inflammation associated with BP largely is driven by a T helper 2–dominant immune response, with increased levels of cytokines such as interleukin (IL) 4 and IL-13.1 The chronic inflammation contributes to the characteristic blisters and intense pruritus seen in patients with BP. Traditional treatments such as corticosteroids work broadly to suppress this inflammation by reducing the production of all proinflammatory cytokines; however, they lack specificity, which dampens the targeted immune response against BP and broadly suppresses the immune system, increasing the risk for infection and other adverse effects. For older patients with multiple health conditions, these adverse effects can impact quality of life and pose health risks.
Interleukin 4 and IL-13 contribute to eosinophilia by upregulating the expression of eosinophil chemoattractant cytokines that promote eosinophil migration and adhesion.1 This is where dupilumab presents a promising alternative. Dupilumab is a monoclonal antibody that selectively targets the key cytokines implicated in BP. It binds to the IL-4 receptor alpha subunit, which IL-4 and IL-13 share, thereby inhibiting the signaling of both cytokines.1
Dupilumab Reports and Case Series
Current evidence supporting the use of dupilumab for BP consists primarily of case series and uncontrolled clinical observations, both of which are useful to formulate but not to test hypotheses.3 One case series of 13 patients diagnosed with BP reported that 92.3% (12/13) of patients achieved disease clearance or satisfactory control, with 53.8% (7/13) achieving total lesion clearance and no adverse events observed.4 In a larger case series, 87.0% of 146 patients achieved disease control with an initial dose of dupilumab 600 mg followed by 300 mg every 2 weeks for 4 weeks, though the study also looked at secondary outcomes for up to 64 weeks.5 Both these findings are compatible with the idea that dupilumab has efficacy in BP, but they cannot be used to test hypotheses.3,4
In a single case report of refractory BP, the combination of dupilumab and omalizumab—a monoclonal antibody that blocks the binding of IgE to mast cells, basophils, and dendritic cells—has shown efficacy, particularly when BP is unresponsive to standard therapies.6 The case involved a 72-year-old woman with unresponsive BP who was treated with a combination of dupilumab and omalizumab. She experienced marked improvement in pruritus and bullae accompanied by normalization of anti-BP180 autoantibody levels. This result points to the possibility that IL-4 and IL-13 blockade could influence autoantibody production. The authors attributed the reduction in pruritus to dupilumab, although the drug was administered in combination with omalizumab.6
Small Randomized Trial of Dupilumab
In addition to these uncontrolled clinical observations, dupilumab recently demonstrated positive results in a small randomized trial.7 Patients who were assigned to receive dupilumab were 5 times more likely to achieve sustained remission compared to those assigned to a placebo. All randomized patients also were treated with a standard-of-care oral corticosteroid regimen, which complicated the ability to isolate the specific effects of dupilumab.7 Finally, a completed totality of evidence on the prescription of dupilumab for refractory BP should include more reliable results from larger randomized trials.8
Final Thoughts
Dupilumab is now approved by the US Food and Drug Administration for the treatment of BP, marking an important milestone as the first targeted biologic therapy for this condition. While the totality of evidence is still evolving, the available data indicate both efficacy and a favorable safety profile when compared to traditional corticosteroids and immunosuppressants. Based on the available data, we suggest that health care providers consider dupilumab as a first-line monotherapy in appropriate patients with BP without specific contraindications, particularly as a practical and safe alternative to corticosteroids and immunosuppressive therapies in older adults with comorbidities. If the patient demonstrates clinical improvement in symptom relief, the therapy can be continued. This strategy minimizes initial exposure to more potent biologics or therapies with higher toxicity profiles. For patients without adequate disease control or with severe initial presentations requiring rapid improvement, clinicians may consider escalation to adjunctive or alternative therapies, such as rituximab, omalizumab, or a short course of systemic corticosteroids for acute management.
Emerging data suggest that dupilumab is a promising therapy for BP. We suggest that it may be a safer targeted alternative to traditional corticosteroids and immunosuppressive therapies. The ability of dupilumab to mitigate inflammation without broadly suppressing the immune system suggests that it may turn out to be an especially valuable option for older patients and those with comorbid conditions.
Acknowledgement—The authors thank Jodi Fiedler, MD (Boca Raton, FL), for her expert advice and assistance.
Bullous pemphigoid (BP) is a chronic autoimmune skin condition characterized by large, fluid-filled blisters (bullae) on the skin. There usually is severe itching, which may antedate the bullae by several weeks. Bullous pemphigoid most commonly affects adults aged 60 years and older with comorbidities such as metabolic syndrome or other autoimmune diseases. The typical blistering is subepidermal, and its occurrence seems to be driven by autoantibodies targeting the epidermal basement membrane zone.
Conventional treatment for BP has long included systemic corticosteroids and immunosuppressants; this combination of drugs effectively controls inflammation but also is associated with considerable risks such as infection, hyperglycemia with worsening diabetes, hypertension, osteoporosis and fracture risk, and delirium, particularly in older patients. Recent developments in biologic therapy have supported the utilization of potentially safer alternatives. Specifically, dupilumab has demonstrated efficacy in the treatment of atopic dermatitis. Case reports and small research studies included in a recent review have suggested that dupilumab could be effective in managing BP, potentially offering a safer treatment option.1 Importantly, dupilumab recently was approved for BP by the US Food and Drug Administration, making it the first biologic approved for treating this condition.2 In this article, we discuss dupilumab as an option for clinicians to prescribe as an initial therapy for BP.
Pathophysiology of Bullous Pemphigoid and Dupilumab
Bullous pemphigoid pathophysiology is driven by 2 main components: immunologic and inflammatory processes. The immune-mediated attack consists of autoantibodies against the hemidesmosomal proteins BP180 and BP230 in the basement membrane zone, which anchor the epidermis to the dermis.1 When these proteins are targeted, a cascade of responses is triggered, including the release of inflammatory cells, particularly eosinophils and neutrophils, into the skin. These cells release inflammatory mediators that break down the basement membrane, resulting in the separation of the epidermis from the dermis and leading to subepidermal blister formation.
The inflammation associated with BP largely is driven by a T helper 2–dominant immune response, with increased levels of cytokines such as interleukin (IL) 4 and IL-13.1 The chronic inflammation contributes to the characteristic blisters and intense pruritus seen in patients with BP. Traditional treatments such as corticosteroids work broadly to suppress this inflammation by reducing the production of all proinflammatory cytokines; however, they lack specificity, which dampens the targeted immune response against BP and broadly suppresses the immune system, increasing the risk for infection and other adverse effects. For older patients with multiple health conditions, these adverse effects can impact quality of life and pose health risks.
Interleukin 4 and IL-13 contribute to eosinophilia by upregulating the expression of eosinophil chemoattractant cytokines that promote eosinophil migration and adhesion.1 This is where dupilumab presents a promising alternative. Dupilumab is a monoclonal antibody that selectively targets the key cytokines implicated in BP. It binds to the IL-4 receptor alpha subunit, which IL-4 and IL-13 share, thereby inhibiting the signaling of both cytokines.1
Dupilumab Reports and Case Series
Current evidence supporting the use of dupilumab for BP consists primarily of case series and uncontrolled clinical observations, both of which are useful to formulate but not to test hypotheses.3 One case series of 13 patients diagnosed with BP reported that 92.3% (12/13) of patients achieved disease clearance or satisfactory control, with 53.8% (7/13) achieving total lesion clearance and no adverse events observed.4 In a larger case series, 87.0% of 146 patients achieved disease control with an initial dose of dupilumab 600 mg followed by 300 mg every 2 weeks for 4 weeks, though the study also looked at secondary outcomes for up to 64 weeks.5 Both these findings are compatible with the idea that dupilumab has efficacy in BP, but they cannot be used to test hypotheses.3,4
In a single case report of refractory BP, the combination of dupilumab and omalizumab—a monoclonal antibody that blocks the binding of IgE to mast cells, basophils, and dendritic cells—has shown efficacy, particularly when BP is unresponsive to standard therapies.6 The case involved a 72-year-old woman with unresponsive BP who was treated with a combination of dupilumab and omalizumab. She experienced marked improvement in pruritus and bullae accompanied by normalization of anti-BP180 autoantibody levels. This result points to the possibility that IL-4 and IL-13 blockade could influence autoantibody production. The authors attributed the reduction in pruritus to dupilumab, although the drug was administered in combination with omalizumab.6
Small Randomized Trial of Dupilumab
In addition to these uncontrolled clinical observations, dupilumab recently demonstrated positive results in a small randomized trial.7 Patients who were assigned to receive dupilumab were 5 times more likely to achieve sustained remission compared to those assigned to a placebo. All randomized patients also were treated with a standard-of-care oral corticosteroid regimen, which complicated the ability to isolate the specific effects of dupilumab.7 Finally, a completed totality of evidence on the prescription of dupilumab for refractory BP should include more reliable results from larger randomized trials.8
Final Thoughts
Dupilumab is now approved by the US Food and Drug Administration for the treatment of BP, marking an important milestone as the first targeted biologic therapy for this condition. While the totality of evidence is still evolving, the available data indicate both efficacy and a favorable safety profile when compared to traditional corticosteroids and immunosuppressants. Based on the available data, we suggest that health care providers consider dupilumab as a first-line monotherapy in appropriate patients with BP without specific contraindications, particularly as a practical and safe alternative to corticosteroids and immunosuppressive therapies in older adults with comorbidities. If the patient demonstrates clinical improvement in symptom relief, the therapy can be continued. This strategy minimizes initial exposure to more potent biologics or therapies with higher toxicity profiles. For patients without adequate disease control or with severe initial presentations requiring rapid improvement, clinicians may consider escalation to adjunctive or alternative therapies, such as rituximab, omalizumab, or a short course of systemic corticosteroids for acute management.
Emerging data suggest that dupilumab is a promising therapy for BP. We suggest that it may be a safer targeted alternative to traditional corticosteroids and immunosuppressive therapies. The ability of dupilumab to mitigate inflammation without broadly suppressing the immune system suggests that it may turn out to be an especially valuable option for older patients and those with comorbid conditions.
Acknowledgement—The authors thank Jodi Fiedler, MD (Boca Raton, FL), for her expert advice and assistance.
- Zhang L, Chen Z, Wang L, et al. Bullous pemphigoid: the role of type 2 inflammation in its pathogenesis and the prospect of targeted therapy. Front Immunol. 2023;14:1115083. doi:10.3389/fimmu.2023.1115083
- Sanofi. Dupixent approved in the US as the only targeted medicine to treat patients with bullous pemphigoid. News release. June 20, 2025. Accessed September 8, 2025. https://www.sanofi.com/en/media-room/press-releases/2025/2025-06-20-05-00-00-3102518
- Hennekens CH, DeMets D. Statistical association and causation: contributions of different types of evidence. JAMA. 2011;306:1134-1136. doi:10.1001/jama.2011.322
- Abdat R, Waldman RA, de Bedout V, et al. Dupilumab as a novel therapy for bullous pemphigoid: a multicenter case series. J Am Acad Dermatol. 2020;83:46-52. doi:10.1016/j.jaad.2020.01.089
- Zhao L, Wang Q, Liang G, et al. Evaluation of dupilumab in patients with bullous pemphigoid. JAMA Dermatol. 2023;159:953-960. doi:10.1001/jamadermatol.2023.2428
- Seyed Jafari SM, Feldmeyer L, Bossart S, et al. Case report: combination of omalizumab and dupilumab for recalcitrant bullous pemphigoid. Front Immunol. 2021;11:611549. doi:10.3389/fimmu.2020.611549
- US National Library of Medicine. A multicenter, randomized, double-blind, placebo-controlled, parallel-group study to evaluate the efficacy and safety of dupilumab in adult patients with bullous pemphigoid. ClinicalTrials.gov. Published 2019. Accessed March 10, 2026. https://clinicaltrials.gov/ct2/show/NCT04206553
- Hennekens CH, DeMets D. The need for large scale randomized evidence without undue emphasis on small trials, meta-analyses or subgroup analyses. JAMA. 2009;302:2361-2362. doi:10.1001/jama.2009.1829
- Zhang L, Chen Z, Wang L, et al. Bullous pemphigoid: the role of type 2 inflammation in its pathogenesis and the prospect of targeted therapy. Front Immunol. 2023;14:1115083. doi:10.3389/fimmu.2023.1115083
- Sanofi. Dupixent approved in the US as the only targeted medicine to treat patients with bullous pemphigoid. News release. June 20, 2025. Accessed September 8, 2025. https://www.sanofi.com/en/media-room/press-releases/2025/2025-06-20-05-00-00-3102518
- Hennekens CH, DeMets D. Statistical association and causation: contributions of different types of evidence. JAMA. 2011;306:1134-1136. doi:10.1001/jama.2011.322
- Abdat R, Waldman RA, de Bedout V, et al. Dupilumab as a novel therapy for bullous pemphigoid: a multicenter case series. J Am Acad Dermatol. 2020;83:46-52. doi:10.1016/j.jaad.2020.01.089
- Zhao L, Wang Q, Liang G, et al. Evaluation of dupilumab in patients with bullous pemphigoid. JAMA Dermatol. 2023;159:953-960. doi:10.1001/jamadermatol.2023.2428
- Seyed Jafari SM, Feldmeyer L, Bossart S, et al. Case report: combination of omalizumab and dupilumab for recalcitrant bullous pemphigoid. Front Immunol. 2021;11:611549. doi:10.3389/fimmu.2020.611549
- US National Library of Medicine. A multicenter, randomized, double-blind, placebo-controlled, parallel-group study to evaluate the efficacy and safety of dupilumab in adult patients with bullous pemphigoid. ClinicalTrials.gov. Published 2019. Accessed March 10, 2026. https://clinicaltrials.gov/ct2/show/NCT04206553
- Hennekens CH, DeMets D. The need for large scale randomized evidence without undue emphasis on small trials, meta-analyses or subgroup analyses. JAMA. 2009;302:2361-2362. doi:10.1001/jama.2009.1829
Dupilumab for Bullous Pemphigoid: To Treat or Not to Treat?
Dupilumab for Bullous Pemphigoid: To Treat or Not to Treat?
PRACTICE POINTS
- Bullous pemphigoid (BP) is a pruritic autoimmune blistering disease in older adults that often requires systemic therapy with associated risks.
- Dupilumab targets interleukin (IL) 4/IL-13–driven inflammation and shows promising efficacy in BP.
- As the first approved biologic for BP, dupilumab may offer a safer alternative to corticosteroids in select patients.
Choosing the Right Electronic Medical Record: Key Features and Considerations for Dermatology Practices
Choosing the Right Electronic Medical Record: Key Features and Considerations for Dermatology Practices
Choosing an electronic medical record (EMR) is one of the most important clinical and financial decisions a dermatology practice will make. An effective system can help streamline workflows, support high-quality patient care, and protect revenue, while the wrong choice can slow clinicians down and add to the administrative burden.
Dermatology workflows involve unique documentation, imaging, and billing needs that are not always well served by generic EMR platforms. To help guide the selection of an EMR, the following framework outlines key features and practice considerations specific to dermatology practices.
While many general EMRs offer customization, dermatology practices benefit greatly from ready-built, specialty-specific documentation tools. Key elements to evaluate include the following:
- Preconfigured dermatology templates for common conditions and procedures (eg, acne, psoriasis, melanoma, biopsies, cosmetic treatments)
- Smart-phrase libraries tailored to dermatologic language and examinations
- Ability to create, modify, and share custom templates across providers
Why It Matters—Efficient charting reduces documentation time, improves consistency, and supports accurate coding.
Dermatology is a highly visual specialty, making clinical photography and image management essential. Important capabilities of an EMR include the following:
- Easy capture, annotation, and longitudinal tracking of clinical images
- Seamless embedding of photographs directly into the patient chart
- Side-by-side comparison of current and prior images
- Secure image storage and camera integration
- Body-mapping tools to mark and track lesion locations visually
Why It Matters—A high-quality image workflow supports diagnosis, treatment planning, patient education, and medicolegal documentation.
For insurance-based practices, robust billing and revenue cycle management (RCM) tools are critical. For direct-care models, some of these items may be prioritized lower. Key features to compare include the following:
- Support for International Classification of Diseases, 10th Revision, Clinical Modification; Current Procedural Terminology; and dermatology-specific code sets
- Automated coding suggestions tied to clinical documentation
- Reviewing claims for errors and inconsistencies prior to submitting to payers’ insurance eligibility verification
- Electronic Remittance Advice/Explanation of Benefits posting and denial management workflows
- Support for cosmetic and self-pay billing
- Ability to generate superbills (itemized receipts for medical services that include International Classification of Diseases Tenth Revision and Current Procedural Terminology codes; patients can submit these directly to their insurance company for reimbursement) for direct-pay practices
Why It Matters—Strong RCM functionality protects revenue, reduces denials, and minimizes staff workload.
The most effective EMRs tightly integrate clinical charting with daily practice operations. Features to evaluate include the following:
- Integrated scheduling with color-coded calendars
- Appointment-type templates and block scheduling
- Automated patient reminders via text or email
- Support for multiprovider and multilocation practices
- Integration with outside pathology or lab services
Why It Matters—Clear and templated scheduling and practice integration help practices run more smoothly by reducing administrative workload and errors and coordinating communication between providers and even ancillary services.
Patient communication and virtual care are increasingly important in dermatology. When evaluating EMRs, compare the following:
- Built-in telehealth functionality vs third-party integrations
- Automated appointment reminders
- Patient portal features (forms, messaging, results)
- Online booking capabilities
Why It Matters—Integrated telehealth and patient communication tools improve access to care, enhance patient engagement, and streamline scheduling, messaging, and virtual visits within dermatology workflows.
Reporting capabilities support clinical quality, compliance, and business decision-making. Key reporting areas include the following:
- Clinical reports (outcomes, lesion tracking, disease management)
- Financial reports (revenue per provider, payer mix, visit types)
- Customizable or exportable reporting tools
Why It Matters—Robust reporting and analytics help dermatology practices track clinical outcomes, monitor financial performance, and make data-driven decisions to improve both patient care and practice management.
The user experience after implementation of the EMR is just as important as the software itself. Evaluate the following after the EMR is implemented:
- Initial training and onboarding resources
- Availability of dermatology-specific support teams
- Ongoing education, help centers, or user communities
- Access to dedicated implementation or success managers
Why It Matters—Strong training and support resources help ensure a smoother EMR implementation, faster staff adoption, and ongoing optimization of the system for dermatology workflows.
Finally, look beyond the sticker price. The total cost of ownership includes far more than monthly fees. Compare the following:
- Upfront costs (implementation, data migration, training)
- Subscription pricing (per provider or per user)
- Billing or RCM fees (including percentages of collections if applicable) and payment processing fees
- Costs for add-on modules (telehealth, imaging, analytics)
- Contract length and termination terms
Why It Matters—Understanding the full cost of ownership helps dermatology practices choose an EMR that fits their budget long-term while avoiding unexpected fees and contractual limitations.
There is no single “best” EMR for every dermatology practice. The right choice depends on your practice model, payer mix, clinical focus, and growth plans. By evaluating EMRs through a dermatology-specific lens and asking the right questions, you can choose a system that supports both excellent patient care and long-term practice success.
Choosing an electronic medical record (EMR) is one of the most important clinical and financial decisions a dermatology practice will make. An effective system can help streamline workflows, support high-quality patient care, and protect revenue, while the wrong choice can slow clinicians down and add to the administrative burden.
Dermatology workflows involve unique documentation, imaging, and billing needs that are not always well served by generic EMR platforms. To help guide the selection of an EMR, the following framework outlines key features and practice considerations specific to dermatology practices.
While many general EMRs offer customization, dermatology practices benefit greatly from ready-built, specialty-specific documentation tools. Key elements to evaluate include the following:
- Preconfigured dermatology templates for common conditions and procedures (eg, acne, psoriasis, melanoma, biopsies, cosmetic treatments)
- Smart-phrase libraries tailored to dermatologic language and examinations
- Ability to create, modify, and share custom templates across providers
Why It Matters—Efficient charting reduces documentation time, improves consistency, and supports accurate coding.
Dermatology is a highly visual specialty, making clinical photography and image management essential. Important capabilities of an EMR include the following:
- Easy capture, annotation, and longitudinal tracking of clinical images
- Seamless embedding of photographs directly into the patient chart
- Side-by-side comparison of current and prior images
- Secure image storage and camera integration
- Body-mapping tools to mark and track lesion locations visually
Why It Matters—A high-quality image workflow supports diagnosis, treatment planning, patient education, and medicolegal documentation.
For insurance-based practices, robust billing and revenue cycle management (RCM) tools are critical. For direct-care models, some of these items may be prioritized lower. Key features to compare include the following:
- Support for International Classification of Diseases, 10th Revision, Clinical Modification; Current Procedural Terminology; and dermatology-specific code sets
- Automated coding suggestions tied to clinical documentation
- Reviewing claims for errors and inconsistencies prior to submitting to payers’ insurance eligibility verification
- Electronic Remittance Advice/Explanation of Benefits posting and denial management workflows
- Support for cosmetic and self-pay billing
- Ability to generate superbills (itemized receipts for medical services that include International Classification of Diseases Tenth Revision and Current Procedural Terminology codes; patients can submit these directly to their insurance company for reimbursement) for direct-pay practices
Why It Matters—Strong RCM functionality protects revenue, reduces denials, and minimizes staff workload.
The most effective EMRs tightly integrate clinical charting with daily practice operations. Features to evaluate include the following:
- Integrated scheduling with color-coded calendars
- Appointment-type templates and block scheduling
- Automated patient reminders via text or email
- Support for multiprovider and multilocation practices
- Integration with outside pathology or lab services
Why It Matters—Clear and templated scheduling and practice integration help practices run more smoothly by reducing administrative workload and errors and coordinating communication between providers and even ancillary services.
Patient communication and virtual care are increasingly important in dermatology. When evaluating EMRs, compare the following:
- Built-in telehealth functionality vs third-party integrations
- Automated appointment reminders
- Patient portal features (forms, messaging, results)
- Online booking capabilities
Why It Matters—Integrated telehealth and patient communication tools improve access to care, enhance patient engagement, and streamline scheduling, messaging, and virtual visits within dermatology workflows.
Reporting capabilities support clinical quality, compliance, and business decision-making. Key reporting areas include the following:
- Clinical reports (outcomes, lesion tracking, disease management)
- Financial reports (revenue per provider, payer mix, visit types)
- Customizable or exportable reporting tools
Why It Matters—Robust reporting and analytics help dermatology practices track clinical outcomes, monitor financial performance, and make data-driven decisions to improve both patient care and practice management.
The user experience after implementation of the EMR is just as important as the software itself. Evaluate the following after the EMR is implemented:
- Initial training and onboarding resources
- Availability of dermatology-specific support teams
- Ongoing education, help centers, or user communities
- Access to dedicated implementation or success managers
Why It Matters—Strong training and support resources help ensure a smoother EMR implementation, faster staff adoption, and ongoing optimization of the system for dermatology workflows.
Finally, look beyond the sticker price. The total cost of ownership includes far more than monthly fees. Compare the following:
- Upfront costs (implementation, data migration, training)
- Subscription pricing (per provider or per user)
- Billing or RCM fees (including percentages of collections if applicable) and payment processing fees
- Costs for add-on modules (telehealth, imaging, analytics)
- Contract length and termination terms
Why It Matters—Understanding the full cost of ownership helps dermatology practices choose an EMR that fits their budget long-term while avoiding unexpected fees and contractual limitations.
There is no single “best” EMR for every dermatology practice. The right choice depends on your practice model, payer mix, clinical focus, and growth plans. By evaluating EMRs through a dermatology-specific lens and asking the right questions, you can choose a system that supports both excellent patient care and long-term practice success.
Choosing an electronic medical record (EMR) is one of the most important clinical and financial decisions a dermatology practice will make. An effective system can help streamline workflows, support high-quality patient care, and protect revenue, while the wrong choice can slow clinicians down and add to the administrative burden.
Dermatology workflows involve unique documentation, imaging, and billing needs that are not always well served by generic EMR platforms. To help guide the selection of an EMR, the following framework outlines key features and practice considerations specific to dermatology practices.
While many general EMRs offer customization, dermatology practices benefit greatly from ready-built, specialty-specific documentation tools. Key elements to evaluate include the following:
- Preconfigured dermatology templates for common conditions and procedures (eg, acne, psoriasis, melanoma, biopsies, cosmetic treatments)
- Smart-phrase libraries tailored to dermatologic language and examinations
- Ability to create, modify, and share custom templates across providers
Why It Matters—Efficient charting reduces documentation time, improves consistency, and supports accurate coding.
Dermatology is a highly visual specialty, making clinical photography and image management essential. Important capabilities of an EMR include the following:
- Easy capture, annotation, and longitudinal tracking of clinical images
- Seamless embedding of photographs directly into the patient chart
- Side-by-side comparison of current and prior images
- Secure image storage and camera integration
- Body-mapping tools to mark and track lesion locations visually
Why It Matters—A high-quality image workflow supports diagnosis, treatment planning, patient education, and medicolegal documentation.
For insurance-based practices, robust billing and revenue cycle management (RCM) tools are critical. For direct-care models, some of these items may be prioritized lower. Key features to compare include the following:
- Support for International Classification of Diseases, 10th Revision, Clinical Modification; Current Procedural Terminology; and dermatology-specific code sets
- Automated coding suggestions tied to clinical documentation
- Reviewing claims for errors and inconsistencies prior to submitting to payers’ insurance eligibility verification
- Electronic Remittance Advice/Explanation of Benefits posting and denial management workflows
- Support for cosmetic and self-pay billing
- Ability to generate superbills (itemized receipts for medical services that include International Classification of Diseases Tenth Revision and Current Procedural Terminology codes; patients can submit these directly to their insurance company for reimbursement) for direct-pay practices
Why It Matters—Strong RCM functionality protects revenue, reduces denials, and minimizes staff workload.
The most effective EMRs tightly integrate clinical charting with daily practice operations. Features to evaluate include the following:
- Integrated scheduling with color-coded calendars
- Appointment-type templates and block scheduling
- Automated patient reminders via text or email
- Support for multiprovider and multilocation practices
- Integration with outside pathology or lab services
Why It Matters—Clear and templated scheduling and practice integration help practices run more smoothly by reducing administrative workload and errors and coordinating communication between providers and even ancillary services.
Patient communication and virtual care are increasingly important in dermatology. When evaluating EMRs, compare the following:
- Built-in telehealth functionality vs third-party integrations
- Automated appointment reminders
- Patient portal features (forms, messaging, results)
- Online booking capabilities
Why It Matters—Integrated telehealth and patient communication tools improve access to care, enhance patient engagement, and streamline scheduling, messaging, and virtual visits within dermatology workflows.
Reporting capabilities support clinical quality, compliance, and business decision-making. Key reporting areas include the following:
- Clinical reports (outcomes, lesion tracking, disease management)
- Financial reports (revenue per provider, payer mix, visit types)
- Customizable or exportable reporting tools
Why It Matters—Robust reporting and analytics help dermatology practices track clinical outcomes, monitor financial performance, and make data-driven decisions to improve both patient care and practice management.
The user experience after implementation of the EMR is just as important as the software itself. Evaluate the following after the EMR is implemented:
- Initial training and onboarding resources
- Availability of dermatology-specific support teams
- Ongoing education, help centers, or user communities
- Access to dedicated implementation or success managers
Why It Matters—Strong training and support resources help ensure a smoother EMR implementation, faster staff adoption, and ongoing optimization of the system for dermatology workflows.
Finally, look beyond the sticker price. The total cost of ownership includes far more than monthly fees. Compare the following:
- Upfront costs (implementation, data migration, training)
- Subscription pricing (per provider or per user)
- Billing or RCM fees (including percentages of collections if applicable) and payment processing fees
- Costs for add-on modules (telehealth, imaging, analytics)
- Contract length and termination terms
Why It Matters—Understanding the full cost of ownership helps dermatology practices choose an EMR that fits their budget long-term while avoiding unexpected fees and contractual limitations.
There is no single “best” EMR for every dermatology practice. The right choice depends on your practice model, payer mix, clinical focus, and growth plans. By evaluating EMRs through a dermatology-specific lens and asking the right questions, you can choose a system that supports both excellent patient care and long-term practice success.
Choosing the Right Electronic Medical Record: Key Features and Considerations for Dermatology Practices
Choosing the Right Electronic Medical Record: Key Features and Considerations for Dermatology Practices
PRACTICE POINTS
- Choosing an electronic medical record (EMR) built for dermatology workflow is a critical part of practice management.
- Features of an EMR that should be evaluated include support for clinical documentation, scheduling and billing, and customer support.
- The proper EMR can reduce administrative tasks and protect practice revenue, but there is no one-size-fits-all option.
Beyond Conventional Paradigms: Rethinking the Evaluation and Management of Melasma
Beyond Conventional Paradigms: Rethinking the Evaluation and Management of Melasma
Melasma is a chronic hyperpigmentation disorder with a high recurrence rate. Advances in pathogenesis, diagnosis, and treatment have shifted dermatologic approaches for both initial management and long-term care. These advances highlight the need to move beyond conventional paradigms for melasma to adopt a more comprehensive and holistic approach to evaluation and management.
Diagnosis: Beyond the Wood Lamp
Diagnosis of melasma is primarily clinical, based on visual examination, with tools such as a Wood lamp (320-400 nm) aiding in classification as epidermal, dermal, or mixed types.1 Dermoscopy is another useful tool that commonly demonstrates a brown reticular pseudonetwork, vascular patterns, and brown clods.2 Reflectance confocal microscopy allows detailed assessment of pigment distribution across skin layers and may serve as a useful diagnostic adjunct.3 Two-photon excitation microscopy also has shown concordance with reflectance confocal microscopy in identifying key pathologic features of melasma and allows quantitative assessment of pigment burden.4,5 Biopsy remains warranted in cases when the diagnosis is unclear.6,7 These advanced tools provide additional options for noninvasive imaging of melasma, which may be useful during both diagnosis and treatment.
Pathogenesis: Beyond the Melanocyte
Recent insights into pathogenesis have shifted the view of melasma from a purely melanocyte-driven disorder to one involving complex epidermal-dermal interactions influenced by chronic UV and visible light exposure and sustained cutaneous inflammation. Evidence suggests involvement of additional cell types and structural components, including epidermal barrier dysfunction, basement membrane alterations, senescent fibroblasts, mast cell activity, vascular remodeling, and dermal solar elastosis. Senescent fibroblasts secrete melanogenic and angiogenic mediators (including stem cell factor, vascular endothelial growth factor, endothelin 1, and matrix metalloproteinases) while mast cell degranulation releases histamine, tryptase, and other proteases that drive melanocyte activation, extracellular matrix degradation, and persistent hyperpigmentation.8 New research highlights the importance of epidermal-dermal communication in melasma disease activity.8,9 Exposure to UV and visible light leads to keratinocyte injury and fibroblast dysfunction, promoting the release of inflammatory and melanogenic mediators that stimulate melanocytes and weaken the basement membrane.8 Mast cell activation and vascular signaling also have been implicated, reinforcing the contribution of inflammation and dermal remodeling to ongoing pigmentation.9 This broader understanding helps explain the difficulty of sustaining long-term remission.8,9 The multifaceted and evolving understanding of melasma’s complex pathogenesis highlights areas for future therapeutic targets beyond melanin, which may help lead to greater clearance and remission periods.
Treatment: Beyond Hydroquinone
Melasma treatment has shifted toward a longitudinal approach reflecting its chronic relapsing nature. Effective management should include combination therapy of broad-spectrum photoprotection, topical therapies, systemic therapies (when appropriate), and adjunctive procedural modalities, while emphasizing the importance of maintenance therapy and relapse prevention.10,11 Growing evidence demonstrates that UV and visible light contribute to melanogenesis and prolonged pigmentation, particularly in individuals with skin of color.12,13 Visible light, particularly within the high-energy blue wavelength spectrum (400-500 nm), induces reactive oxygen species and stimulates melanogenesis, resulting in persistent hyperpigmentation and exacerbation of melasma in darker skin types.13 Iron oxide, a mineral-based physical filter commonly used in tinted mineral sunscreens, enhances protection from visible light, thus improving melasma outcomes.12,13 In a randomized double-blind placebo-controlled trial, oral polypodium leucotomos extract, an antioxidant with photoprotective effects, improved Melasma Area and Severity Index (MASI)/modified MASI (mMASI) scores and melasma-rated quality of life measures when used as an adjunct to sunscreen with or without topical hydroquinone.14
Topical therapy serves as the mainstay of melasma treatment. Hydroquinone- and retinoid-based regimens including triple-combination therapy remain an effective gold standard for active disease; however, there still are concerns about irritant dermatitis with both as well as ochronosis with long-term use of hydroquinone.15 Recent studies highlight the efficacy of nonhydroquinone topical therapies such as azelaic acid, tranexamic acid (TXA), thiamidol, cysteamine, metformin, malassezin, 2-mercaptonicotinoyl glycine, niacinamide, kojic acid, ascorbic acid, and botanical-derived compounds, many of which demonstrate reductions in disease severity and more favorable tolerability profiles.16-19 Combinations of these ingredients can allow for addressing the various pathogenic factors of melasma beyond pigmentation, including vascularity, inflammation, and solar damage.
Systemic therapy, particularly oral TXA, has emerged as an option for patients with moderate to severe or refractory melasma. Clinical trials and observational studies demonstrate improvement with oral TXA; however, relapse after discontinuation is common, emphasizing the importance of careful patient selection, established treatment duration, and concurrent maintenance therapy.16,20 Although it is well tolerated, adverse effects include gastrointestinal discomfort, menstrual irregularities, and headache. Additionally, prescreening for contraindications and clotting risk factors is needed to avoid thromboembolic events.21
Procedural interventions are best used as adjunctive therapies rather than primary treatment. Superficial and medium peels can be used depending on baseline skin type.22 Platelet-rich plasma is a promising adjunctive therapy for melasma, demonstrating reductions in MASI scores following intradermal or microneedling-assisted delivery, likely mediated through growth factor–driven inhibition of melanogenesis and modulation of tyrosinase activity. Platelet-rich plasma promotes dermal remodeling through angiogenesis, collagen synthesis, and extracellular matrix production improving skin texture and pigmentation.23 Botulinum toxin A may improve pigmentation by modulating melanogenesis as well as inflammatory and vascular pathways.24
Radiofrequency microneedling provides a dermal-targeting therapeutic approach, with some evidence showing that monthly treatments can help maintain improvements achieved with conventional therapy by modulating photoaged dermal structures impacted by melanogenic signaling.25 Histopathologic correlation studies suggest that microneedling used alone or as a delivery platform alongside topical agents may be beneficial in dermal-predominant disease, while laser-based therapies should be reserved for recalcitrant cases due to the risk for postinflammatory hyperpigmentation and melasma recurrence, particularly in those with darker skin types.26 Lasers such as Q-switched 1064-nm Nd:YAG as well as nonablative lasers used in conservative settings are options for melasma treatment, including in patients with skin of color.26,27 Laser toning with serial low-fluence (<3 J/cm2), large-spot 1064-nm Nd:YAG has been used to reduce inflammation and risk for postinflammatory hyperpigmentation.28 Additionally, randomized split-face data demonstrate that picosecond 1064-nm devices achieve comparable reductions in mMASI scores to traditional nanosecond devices, and combination strategies incorporating intense pulsed light (IPL) have shown greater MASI improvement compared with IPL alone.28,29 However, IPL generally is limited to lighter skin phototypes due to the risk for postinflammatory hyperpigmentation in darker skin.30
Lastly, long-term management requires patient education on the importance of year-round maintenance therapy, consistency, and compliance. Reinforcing adherence to photoprotection, supporting barrier repair, and setting expectations regarding prognosis are essential components to optimizing melasma treatment outcomes.12
Future Insights: Beyond the Current State of Melasma Research and Treatment
Future advances in melasma management likely will focus on improving disease assessment and long-term treatment durability. Tools such as the MASI and mMASI are widely used but are limited by interobserver variability and reduced sensitivity in darker skin types, highlighting the need to incorporate objective measures such as colorimetry.10,26 There also is a need for studies that relate clinical improvement to underlying pathology. Histopathologic correlation remains limited across most therapeutic trials, and more studies should assess long-term outcomes and quality-of-life measures given the relapsing nature of melasma.26
Future interventions will continue to move past the treatment of pigment alone. Increasing recognition of vascular, inflammatory, and barrier-related contributions to melasma, as well as targeting of other cell types affected by melasma, will be important considerations in future treatment innovations.8,11 In summary, improved inclusion of patients with skin of color in clinical trials, expansion of clinical outcomes measures and objective parameters of disease evaluation, novel approaches to treatment and relapse prevention, and greater attention to patient adherence and education will be essential to achieving advancements in melasma care.12
- Sanchez NP, Pathak MA, Sato S, et al. Melasma: a clinical, light microscopic, ultrastructural, and immunofluorescence study. J Am Acad Dermatol. 1981;4:698-710.
- Bostan E, Cakir A. The dermoscopic characteristics of melasma in relation to different skin phototypes, distribution patterns and wood lamp findings: a cross-sectional study of 236 melasma lesions. Arch Dermatol Res. 2023;315:1927-1938.
- Guida S, Longo C, Ronga R, et al. Melasma and reflectance confocal microscopy: from baseline to treatment monitoring. Int J Dermatol. 2024;63:1007-1012.
- Ning X, Yang J, Ouyang H, et al. In vivo evaluation of melasma pathologic features and treatment response by 2-photon microscopy. JAMA Dermatol. 2025;161:1039-1047.
- Chen K-J, Han Y, Wang Z-Y, et al. Submicron resolution techniques: multiphoton microscopy in skin disease. Exp Dermatol. 2023;32:1613-1623.
- Philipp-Dormston WG. Melasma: a step-by-step approach towards a multimodal combination therapy. Clin Cosmet Investig Dermatol. 2024;17:1203-1216.
- Doolan B, Gupta M. Melasma. Aust J Gen Pract. 2021;50:880-885.
- Ali L, Al Niaimi F. Pathogenesis of melasma explained. Int J Dermatol. 2025;64:1201-1212.
- Artzi O, Horovitz T, Bar-Ilan E, et al. The pathogenesis of melasma and implications for treatment. J Cosmet Dermatol. 2021;20:3432-3445.
- Gan C, Rodrigues M. An update on new and existing treatments for the management of melasma. Am J Clin Dermatol. 2024;25:717-733.
- Sarkar R, Desai SR, Sinha S, et al. Delphi consensus on melasma management by international experts and pigmentary disorders society. J Eur Acad Dermatol Venereol. Published online September 25, 2025.
- Desai SR, Alexis AF, Elbuluk N, et al. Best practices in the treatment of melasma with a focus on patients with skin of color. J Am Acad Dermatol. 2024;90:269-279.
- Lim HW, Kohli I, Ruvolo E, et al. Impact of visible light on skin health: the role of antioxidants and free radical quenchers in skin protection. J Am Acad Deramtol. 2022;86(3 suppl):S27-S37.
- Goh CL, Chuah SY, Tien S, et al. Double-blind, placebo-controlled trial to evaluate the effectiveness of polypodium leucotomos extract in the treatment of melasma in Asian skin: a pilot study. J Clin Aesthet Dermatol. 2018;11:14-19.
- Mahajan VK, Patil A, Blicharz L, et al. Medical therapies for melasma. J Cosmet Dermatol. 2022;21:3707-3728.
- Suliman RS, Alhuwayshil J, Almuflehi AA, et al. Emerging topical therapies for melasma: a comparative analysis of efficacy and safety. J Dermatolog Treat. 2025;36:2591502.
- Austin E, Nguyen JK, Jagdeo J. Topical treatments for melasma: a systematic review of randomized controlled trials. J Drugs Dermatol. 2019;18:S1545961619P1156X.
- Grimes PE, Dias S, Oparaugo NC, et al. A randomized, controlled, split-face, double-blind study comparing topical malassezin to hydroquinone 4% for melasma. J Drugs Dermatol. 2026;25:25-29.
- Passeron T, Kerob D, Le Dantec G, et al. Efficacy and tolerability of a new facial 2-mercaptonicotinoyl glycine-containing depigmenting serum versus hydroquinone 4% over 3-month treatment of facial melasma. Dermatol Ther (Heidelb). 2025;15:2379-2390.
- McKesey J, Tovar-Garza A, Pandya AG. Melasma treatment: an evidence-based review. Am J Clin Dermatol. 2020;21:173-225.
- Bala HR, Lee S, Wong C, et al. Oral tranexamic acid for the treatment of melasma: a review. Dermatol Surg. 2018;44:814-825.
- Sarkar R, Katoch S. Chemical peels in treatment of melasma. Dermatol Clin. 2024;42:21-32.
- Sarkar R, Gupta M. Platelet-rich plasma in melasma—a systematic review. Dermatol Surg. 2022;48:131-134.
- Pongklaokam J, Manuskiatti W, Wanitphakdeedecha R, et al. Efficacy and safety of letibotulinum toxin A for the treatment of melasma in two different dilutions: a randomized double-blind split-face study. Toxins (Basel). 2025;17:349.
- Han HJ, Kim JC, Park YJ, et al. Targeting the dermis for melasma maintenance treatment. Sci Rep. 2024;14:949.
- Zhang AD, Lazar M, Akhundova E, et al. A scoping review on melasma treatments and their histopathologic correlates. Dermatopathology (Basel). 2025;12:13.
- Goldman MP, Wu DC, Khetarpal S, et al. Dual 1550-nm erbium glass fiber and 1927-nm thulium fiber non-ablative fractional laser system treatment in patients with skin of color: a review of clinical studies and unmet needs. Lasers Surg Med. Published online February 10, 2026.
- Chen J, Yu N, Peng L, et al. Efficacy of low-fluence 1064 nm Q-switched Nd: YAG laser for the treatment of melasma: a meta-analysis and systematic review. J Cosmet Dermatol. 2022;21.
- Feng J, Huang L. Comparison of picosecond and nanosecond Nd:YAG 1064-nm lasers in the treatment of melasma: a split-face randomized clinical trial. Plast Reconstr Surg. 2023;151:772-777.
- Passeron T, Genedy R, Salah L, et al., Laser treatment of hyperpigmented lesions: position statement of the European Society of Laser in Dermatology. J Eur Acad Dermatol Venereol. 2019;33:987-1005.
Melasma is a chronic hyperpigmentation disorder with a high recurrence rate. Advances in pathogenesis, diagnosis, and treatment have shifted dermatologic approaches for both initial management and long-term care. These advances highlight the need to move beyond conventional paradigms for melasma to adopt a more comprehensive and holistic approach to evaluation and management.
Diagnosis: Beyond the Wood Lamp
Diagnosis of melasma is primarily clinical, based on visual examination, with tools such as a Wood lamp (320-400 nm) aiding in classification as epidermal, dermal, or mixed types.1 Dermoscopy is another useful tool that commonly demonstrates a brown reticular pseudonetwork, vascular patterns, and brown clods.2 Reflectance confocal microscopy allows detailed assessment of pigment distribution across skin layers and may serve as a useful diagnostic adjunct.3 Two-photon excitation microscopy also has shown concordance with reflectance confocal microscopy in identifying key pathologic features of melasma and allows quantitative assessment of pigment burden.4,5 Biopsy remains warranted in cases when the diagnosis is unclear.6,7 These advanced tools provide additional options for noninvasive imaging of melasma, which may be useful during both diagnosis and treatment.
Pathogenesis: Beyond the Melanocyte
Recent insights into pathogenesis have shifted the view of melasma from a purely melanocyte-driven disorder to one involving complex epidermal-dermal interactions influenced by chronic UV and visible light exposure and sustained cutaneous inflammation. Evidence suggests involvement of additional cell types and structural components, including epidermal barrier dysfunction, basement membrane alterations, senescent fibroblasts, mast cell activity, vascular remodeling, and dermal solar elastosis. Senescent fibroblasts secrete melanogenic and angiogenic mediators (including stem cell factor, vascular endothelial growth factor, endothelin 1, and matrix metalloproteinases) while mast cell degranulation releases histamine, tryptase, and other proteases that drive melanocyte activation, extracellular matrix degradation, and persistent hyperpigmentation.8 New research highlights the importance of epidermal-dermal communication in melasma disease activity.8,9 Exposure to UV and visible light leads to keratinocyte injury and fibroblast dysfunction, promoting the release of inflammatory and melanogenic mediators that stimulate melanocytes and weaken the basement membrane.8 Mast cell activation and vascular signaling also have been implicated, reinforcing the contribution of inflammation and dermal remodeling to ongoing pigmentation.9 This broader understanding helps explain the difficulty of sustaining long-term remission.8,9 The multifaceted and evolving understanding of melasma’s complex pathogenesis highlights areas for future therapeutic targets beyond melanin, which may help lead to greater clearance and remission periods.
Treatment: Beyond Hydroquinone
Melasma treatment has shifted toward a longitudinal approach reflecting its chronic relapsing nature. Effective management should include combination therapy of broad-spectrum photoprotection, topical therapies, systemic therapies (when appropriate), and adjunctive procedural modalities, while emphasizing the importance of maintenance therapy and relapse prevention.10,11 Growing evidence demonstrates that UV and visible light contribute to melanogenesis and prolonged pigmentation, particularly in individuals with skin of color.12,13 Visible light, particularly within the high-energy blue wavelength spectrum (400-500 nm), induces reactive oxygen species and stimulates melanogenesis, resulting in persistent hyperpigmentation and exacerbation of melasma in darker skin types.13 Iron oxide, a mineral-based physical filter commonly used in tinted mineral sunscreens, enhances protection from visible light, thus improving melasma outcomes.12,13 In a randomized double-blind placebo-controlled trial, oral polypodium leucotomos extract, an antioxidant with photoprotective effects, improved Melasma Area and Severity Index (MASI)/modified MASI (mMASI) scores and melasma-rated quality of life measures when used as an adjunct to sunscreen with or without topical hydroquinone.14
Topical therapy serves as the mainstay of melasma treatment. Hydroquinone- and retinoid-based regimens including triple-combination therapy remain an effective gold standard for active disease; however, there still are concerns about irritant dermatitis with both as well as ochronosis with long-term use of hydroquinone.15 Recent studies highlight the efficacy of nonhydroquinone topical therapies such as azelaic acid, tranexamic acid (TXA), thiamidol, cysteamine, metformin, malassezin, 2-mercaptonicotinoyl glycine, niacinamide, kojic acid, ascorbic acid, and botanical-derived compounds, many of which demonstrate reductions in disease severity and more favorable tolerability profiles.16-19 Combinations of these ingredients can allow for addressing the various pathogenic factors of melasma beyond pigmentation, including vascularity, inflammation, and solar damage.
Systemic therapy, particularly oral TXA, has emerged as an option for patients with moderate to severe or refractory melasma. Clinical trials and observational studies demonstrate improvement with oral TXA; however, relapse after discontinuation is common, emphasizing the importance of careful patient selection, established treatment duration, and concurrent maintenance therapy.16,20 Although it is well tolerated, adverse effects include gastrointestinal discomfort, menstrual irregularities, and headache. Additionally, prescreening for contraindications and clotting risk factors is needed to avoid thromboembolic events.21
Procedural interventions are best used as adjunctive therapies rather than primary treatment. Superficial and medium peels can be used depending on baseline skin type.22 Platelet-rich plasma is a promising adjunctive therapy for melasma, demonstrating reductions in MASI scores following intradermal or microneedling-assisted delivery, likely mediated through growth factor–driven inhibition of melanogenesis and modulation of tyrosinase activity. Platelet-rich plasma promotes dermal remodeling through angiogenesis, collagen synthesis, and extracellular matrix production improving skin texture and pigmentation.23 Botulinum toxin A may improve pigmentation by modulating melanogenesis as well as inflammatory and vascular pathways.24
Radiofrequency microneedling provides a dermal-targeting therapeutic approach, with some evidence showing that monthly treatments can help maintain improvements achieved with conventional therapy by modulating photoaged dermal structures impacted by melanogenic signaling.25 Histopathologic correlation studies suggest that microneedling used alone or as a delivery platform alongside topical agents may be beneficial in dermal-predominant disease, while laser-based therapies should be reserved for recalcitrant cases due to the risk for postinflammatory hyperpigmentation and melasma recurrence, particularly in those with darker skin types.26 Lasers such as Q-switched 1064-nm Nd:YAG as well as nonablative lasers used in conservative settings are options for melasma treatment, including in patients with skin of color.26,27 Laser toning with serial low-fluence (<3 J/cm2), large-spot 1064-nm Nd:YAG has been used to reduce inflammation and risk for postinflammatory hyperpigmentation.28 Additionally, randomized split-face data demonstrate that picosecond 1064-nm devices achieve comparable reductions in mMASI scores to traditional nanosecond devices, and combination strategies incorporating intense pulsed light (IPL) have shown greater MASI improvement compared with IPL alone.28,29 However, IPL generally is limited to lighter skin phototypes due to the risk for postinflammatory hyperpigmentation in darker skin.30
Lastly, long-term management requires patient education on the importance of year-round maintenance therapy, consistency, and compliance. Reinforcing adherence to photoprotection, supporting barrier repair, and setting expectations regarding prognosis are essential components to optimizing melasma treatment outcomes.12
Future Insights: Beyond the Current State of Melasma Research and Treatment
Future advances in melasma management likely will focus on improving disease assessment and long-term treatment durability. Tools such as the MASI and mMASI are widely used but are limited by interobserver variability and reduced sensitivity in darker skin types, highlighting the need to incorporate objective measures such as colorimetry.10,26 There also is a need for studies that relate clinical improvement to underlying pathology. Histopathologic correlation remains limited across most therapeutic trials, and more studies should assess long-term outcomes and quality-of-life measures given the relapsing nature of melasma.26
Future interventions will continue to move past the treatment of pigment alone. Increasing recognition of vascular, inflammatory, and barrier-related contributions to melasma, as well as targeting of other cell types affected by melasma, will be important considerations in future treatment innovations.8,11 In summary, improved inclusion of patients with skin of color in clinical trials, expansion of clinical outcomes measures and objective parameters of disease evaluation, novel approaches to treatment and relapse prevention, and greater attention to patient adherence and education will be essential to achieving advancements in melasma care.12
Melasma is a chronic hyperpigmentation disorder with a high recurrence rate. Advances in pathogenesis, diagnosis, and treatment have shifted dermatologic approaches for both initial management and long-term care. These advances highlight the need to move beyond conventional paradigms for melasma to adopt a more comprehensive and holistic approach to evaluation and management.
Diagnosis: Beyond the Wood Lamp
Diagnosis of melasma is primarily clinical, based on visual examination, with tools such as a Wood lamp (320-400 nm) aiding in classification as epidermal, dermal, or mixed types.1 Dermoscopy is another useful tool that commonly demonstrates a brown reticular pseudonetwork, vascular patterns, and brown clods.2 Reflectance confocal microscopy allows detailed assessment of pigment distribution across skin layers and may serve as a useful diagnostic adjunct.3 Two-photon excitation microscopy also has shown concordance with reflectance confocal microscopy in identifying key pathologic features of melasma and allows quantitative assessment of pigment burden.4,5 Biopsy remains warranted in cases when the diagnosis is unclear.6,7 These advanced tools provide additional options for noninvasive imaging of melasma, which may be useful during both diagnosis and treatment.
Pathogenesis: Beyond the Melanocyte
Recent insights into pathogenesis have shifted the view of melasma from a purely melanocyte-driven disorder to one involving complex epidermal-dermal interactions influenced by chronic UV and visible light exposure and sustained cutaneous inflammation. Evidence suggests involvement of additional cell types and structural components, including epidermal barrier dysfunction, basement membrane alterations, senescent fibroblasts, mast cell activity, vascular remodeling, and dermal solar elastosis. Senescent fibroblasts secrete melanogenic and angiogenic mediators (including stem cell factor, vascular endothelial growth factor, endothelin 1, and matrix metalloproteinases) while mast cell degranulation releases histamine, tryptase, and other proteases that drive melanocyte activation, extracellular matrix degradation, and persistent hyperpigmentation.8 New research highlights the importance of epidermal-dermal communication in melasma disease activity.8,9 Exposure to UV and visible light leads to keratinocyte injury and fibroblast dysfunction, promoting the release of inflammatory and melanogenic mediators that stimulate melanocytes and weaken the basement membrane.8 Mast cell activation and vascular signaling also have been implicated, reinforcing the contribution of inflammation and dermal remodeling to ongoing pigmentation.9 This broader understanding helps explain the difficulty of sustaining long-term remission.8,9 The multifaceted and evolving understanding of melasma’s complex pathogenesis highlights areas for future therapeutic targets beyond melanin, which may help lead to greater clearance and remission periods.
Treatment: Beyond Hydroquinone
Melasma treatment has shifted toward a longitudinal approach reflecting its chronic relapsing nature. Effective management should include combination therapy of broad-spectrum photoprotection, topical therapies, systemic therapies (when appropriate), and adjunctive procedural modalities, while emphasizing the importance of maintenance therapy and relapse prevention.10,11 Growing evidence demonstrates that UV and visible light contribute to melanogenesis and prolonged pigmentation, particularly in individuals with skin of color.12,13 Visible light, particularly within the high-energy blue wavelength spectrum (400-500 nm), induces reactive oxygen species and stimulates melanogenesis, resulting in persistent hyperpigmentation and exacerbation of melasma in darker skin types.13 Iron oxide, a mineral-based physical filter commonly used in tinted mineral sunscreens, enhances protection from visible light, thus improving melasma outcomes.12,13 In a randomized double-blind placebo-controlled trial, oral polypodium leucotomos extract, an antioxidant with photoprotective effects, improved Melasma Area and Severity Index (MASI)/modified MASI (mMASI) scores and melasma-rated quality of life measures when used as an adjunct to sunscreen with or without topical hydroquinone.14
Topical therapy serves as the mainstay of melasma treatment. Hydroquinone- and retinoid-based regimens including triple-combination therapy remain an effective gold standard for active disease; however, there still are concerns about irritant dermatitis with both as well as ochronosis with long-term use of hydroquinone.15 Recent studies highlight the efficacy of nonhydroquinone topical therapies such as azelaic acid, tranexamic acid (TXA), thiamidol, cysteamine, metformin, malassezin, 2-mercaptonicotinoyl glycine, niacinamide, kojic acid, ascorbic acid, and botanical-derived compounds, many of which demonstrate reductions in disease severity and more favorable tolerability profiles.16-19 Combinations of these ingredients can allow for addressing the various pathogenic factors of melasma beyond pigmentation, including vascularity, inflammation, and solar damage.
Systemic therapy, particularly oral TXA, has emerged as an option for patients with moderate to severe or refractory melasma. Clinical trials and observational studies demonstrate improvement with oral TXA; however, relapse after discontinuation is common, emphasizing the importance of careful patient selection, established treatment duration, and concurrent maintenance therapy.16,20 Although it is well tolerated, adverse effects include gastrointestinal discomfort, menstrual irregularities, and headache. Additionally, prescreening for contraindications and clotting risk factors is needed to avoid thromboembolic events.21
Procedural interventions are best used as adjunctive therapies rather than primary treatment. Superficial and medium peels can be used depending on baseline skin type.22 Platelet-rich plasma is a promising adjunctive therapy for melasma, demonstrating reductions in MASI scores following intradermal or microneedling-assisted delivery, likely mediated through growth factor–driven inhibition of melanogenesis and modulation of tyrosinase activity. Platelet-rich plasma promotes dermal remodeling through angiogenesis, collagen synthesis, and extracellular matrix production improving skin texture and pigmentation.23 Botulinum toxin A may improve pigmentation by modulating melanogenesis as well as inflammatory and vascular pathways.24
Radiofrequency microneedling provides a dermal-targeting therapeutic approach, with some evidence showing that monthly treatments can help maintain improvements achieved with conventional therapy by modulating photoaged dermal structures impacted by melanogenic signaling.25 Histopathologic correlation studies suggest that microneedling used alone or as a delivery platform alongside topical agents may be beneficial in dermal-predominant disease, while laser-based therapies should be reserved for recalcitrant cases due to the risk for postinflammatory hyperpigmentation and melasma recurrence, particularly in those with darker skin types.26 Lasers such as Q-switched 1064-nm Nd:YAG as well as nonablative lasers used in conservative settings are options for melasma treatment, including in patients with skin of color.26,27 Laser toning with serial low-fluence (<3 J/cm2), large-spot 1064-nm Nd:YAG has been used to reduce inflammation and risk for postinflammatory hyperpigmentation.28 Additionally, randomized split-face data demonstrate that picosecond 1064-nm devices achieve comparable reductions in mMASI scores to traditional nanosecond devices, and combination strategies incorporating intense pulsed light (IPL) have shown greater MASI improvement compared with IPL alone.28,29 However, IPL generally is limited to lighter skin phototypes due to the risk for postinflammatory hyperpigmentation in darker skin.30
Lastly, long-term management requires patient education on the importance of year-round maintenance therapy, consistency, and compliance. Reinforcing adherence to photoprotection, supporting barrier repair, and setting expectations regarding prognosis are essential components to optimizing melasma treatment outcomes.12
Future Insights: Beyond the Current State of Melasma Research and Treatment
Future advances in melasma management likely will focus on improving disease assessment and long-term treatment durability. Tools such as the MASI and mMASI are widely used but are limited by interobserver variability and reduced sensitivity in darker skin types, highlighting the need to incorporate objective measures such as colorimetry.10,26 There also is a need for studies that relate clinical improvement to underlying pathology. Histopathologic correlation remains limited across most therapeutic trials, and more studies should assess long-term outcomes and quality-of-life measures given the relapsing nature of melasma.26
Future interventions will continue to move past the treatment of pigment alone. Increasing recognition of vascular, inflammatory, and barrier-related contributions to melasma, as well as targeting of other cell types affected by melasma, will be important considerations in future treatment innovations.8,11 In summary, improved inclusion of patients with skin of color in clinical trials, expansion of clinical outcomes measures and objective parameters of disease evaluation, novel approaches to treatment and relapse prevention, and greater attention to patient adherence and education will be essential to achieving advancements in melasma care.12
- Sanchez NP, Pathak MA, Sato S, et al. Melasma: a clinical, light microscopic, ultrastructural, and immunofluorescence study. J Am Acad Dermatol. 1981;4:698-710.
- Bostan E, Cakir A. The dermoscopic characteristics of melasma in relation to different skin phototypes, distribution patterns and wood lamp findings: a cross-sectional study of 236 melasma lesions. Arch Dermatol Res. 2023;315:1927-1938.
- Guida S, Longo C, Ronga R, et al. Melasma and reflectance confocal microscopy: from baseline to treatment monitoring. Int J Dermatol. 2024;63:1007-1012.
- Ning X, Yang J, Ouyang H, et al. In vivo evaluation of melasma pathologic features and treatment response by 2-photon microscopy. JAMA Dermatol. 2025;161:1039-1047.
- Chen K-J, Han Y, Wang Z-Y, et al. Submicron resolution techniques: multiphoton microscopy in skin disease. Exp Dermatol. 2023;32:1613-1623.
- Philipp-Dormston WG. Melasma: a step-by-step approach towards a multimodal combination therapy. Clin Cosmet Investig Dermatol. 2024;17:1203-1216.
- Doolan B, Gupta M. Melasma. Aust J Gen Pract. 2021;50:880-885.
- Ali L, Al Niaimi F. Pathogenesis of melasma explained. Int J Dermatol. 2025;64:1201-1212.
- Artzi O, Horovitz T, Bar-Ilan E, et al. The pathogenesis of melasma and implications for treatment. J Cosmet Dermatol. 2021;20:3432-3445.
- Gan C, Rodrigues M. An update on new and existing treatments for the management of melasma. Am J Clin Dermatol. 2024;25:717-733.
- Sarkar R, Desai SR, Sinha S, et al. Delphi consensus on melasma management by international experts and pigmentary disorders society. J Eur Acad Dermatol Venereol. Published online September 25, 2025.
- Desai SR, Alexis AF, Elbuluk N, et al. Best practices in the treatment of melasma with a focus on patients with skin of color. J Am Acad Dermatol. 2024;90:269-279.
- Lim HW, Kohli I, Ruvolo E, et al. Impact of visible light on skin health: the role of antioxidants and free radical quenchers in skin protection. J Am Acad Deramtol. 2022;86(3 suppl):S27-S37.
- Goh CL, Chuah SY, Tien S, et al. Double-blind, placebo-controlled trial to evaluate the effectiveness of polypodium leucotomos extract in the treatment of melasma in Asian skin: a pilot study. J Clin Aesthet Dermatol. 2018;11:14-19.
- Mahajan VK, Patil A, Blicharz L, et al. Medical therapies for melasma. J Cosmet Dermatol. 2022;21:3707-3728.
- Suliman RS, Alhuwayshil J, Almuflehi AA, et al. Emerging topical therapies for melasma: a comparative analysis of efficacy and safety. J Dermatolog Treat. 2025;36:2591502.
- Austin E, Nguyen JK, Jagdeo J. Topical treatments for melasma: a systematic review of randomized controlled trials. J Drugs Dermatol. 2019;18:S1545961619P1156X.
- Grimes PE, Dias S, Oparaugo NC, et al. A randomized, controlled, split-face, double-blind study comparing topical malassezin to hydroquinone 4% for melasma. J Drugs Dermatol. 2026;25:25-29.
- Passeron T, Kerob D, Le Dantec G, et al. Efficacy and tolerability of a new facial 2-mercaptonicotinoyl glycine-containing depigmenting serum versus hydroquinone 4% over 3-month treatment of facial melasma. Dermatol Ther (Heidelb). 2025;15:2379-2390.
- McKesey J, Tovar-Garza A, Pandya AG. Melasma treatment: an evidence-based review. Am J Clin Dermatol. 2020;21:173-225.
- Bala HR, Lee S, Wong C, et al. Oral tranexamic acid for the treatment of melasma: a review. Dermatol Surg. 2018;44:814-825.
- Sarkar R, Katoch S. Chemical peels in treatment of melasma. Dermatol Clin. 2024;42:21-32.
- Sarkar R, Gupta M. Platelet-rich plasma in melasma—a systematic review. Dermatol Surg. 2022;48:131-134.
- Pongklaokam J, Manuskiatti W, Wanitphakdeedecha R, et al. Efficacy and safety of letibotulinum toxin A for the treatment of melasma in two different dilutions: a randomized double-blind split-face study. Toxins (Basel). 2025;17:349.
- Han HJ, Kim JC, Park YJ, et al. Targeting the dermis for melasma maintenance treatment. Sci Rep. 2024;14:949.
- Zhang AD, Lazar M, Akhundova E, et al. A scoping review on melasma treatments and their histopathologic correlates. Dermatopathology (Basel). 2025;12:13.
- Goldman MP, Wu DC, Khetarpal S, et al. Dual 1550-nm erbium glass fiber and 1927-nm thulium fiber non-ablative fractional laser system treatment in patients with skin of color: a review of clinical studies and unmet needs. Lasers Surg Med. Published online February 10, 2026.
- Chen J, Yu N, Peng L, et al. Efficacy of low-fluence 1064 nm Q-switched Nd: YAG laser for the treatment of melasma: a meta-analysis and systematic review. J Cosmet Dermatol. 2022;21.
- Feng J, Huang L. Comparison of picosecond and nanosecond Nd:YAG 1064-nm lasers in the treatment of melasma: a split-face randomized clinical trial. Plast Reconstr Surg. 2023;151:772-777.
- Passeron T, Genedy R, Salah L, et al., Laser treatment of hyperpigmented lesions: position statement of the European Society of Laser in Dermatology. J Eur Acad Dermatol Venereol. 2019;33:987-1005.
- Sanchez NP, Pathak MA, Sato S, et al. Melasma: a clinical, light microscopic, ultrastructural, and immunofluorescence study. J Am Acad Dermatol. 1981;4:698-710.
- Bostan E, Cakir A. The dermoscopic characteristics of melasma in relation to different skin phototypes, distribution patterns and wood lamp findings: a cross-sectional study of 236 melasma lesions. Arch Dermatol Res. 2023;315:1927-1938.
- Guida S, Longo C, Ronga R, et al. Melasma and reflectance confocal microscopy: from baseline to treatment monitoring. Int J Dermatol. 2024;63:1007-1012.
- Ning X, Yang J, Ouyang H, et al. In vivo evaluation of melasma pathologic features and treatment response by 2-photon microscopy. JAMA Dermatol. 2025;161:1039-1047.
- Chen K-J, Han Y, Wang Z-Y, et al. Submicron resolution techniques: multiphoton microscopy in skin disease. Exp Dermatol. 2023;32:1613-1623.
- Philipp-Dormston WG. Melasma: a step-by-step approach towards a multimodal combination therapy. Clin Cosmet Investig Dermatol. 2024;17:1203-1216.
- Doolan B, Gupta M. Melasma. Aust J Gen Pract. 2021;50:880-885.
- Ali L, Al Niaimi F. Pathogenesis of melasma explained. Int J Dermatol. 2025;64:1201-1212.
- Artzi O, Horovitz T, Bar-Ilan E, et al. The pathogenesis of melasma and implications for treatment. J Cosmet Dermatol. 2021;20:3432-3445.
- Gan C, Rodrigues M. An update on new and existing treatments for the management of melasma. Am J Clin Dermatol. 2024;25:717-733.
- Sarkar R, Desai SR, Sinha S, et al. Delphi consensus on melasma management by international experts and pigmentary disorders society. J Eur Acad Dermatol Venereol. Published online September 25, 2025.
- Desai SR, Alexis AF, Elbuluk N, et al. Best practices in the treatment of melasma with a focus on patients with skin of color. J Am Acad Dermatol. 2024;90:269-279.
- Lim HW, Kohli I, Ruvolo E, et al. Impact of visible light on skin health: the role of antioxidants and free radical quenchers in skin protection. J Am Acad Deramtol. 2022;86(3 suppl):S27-S37.
- Goh CL, Chuah SY, Tien S, et al. Double-blind, placebo-controlled trial to evaluate the effectiveness of polypodium leucotomos extract in the treatment of melasma in Asian skin: a pilot study. J Clin Aesthet Dermatol. 2018;11:14-19.
- Mahajan VK, Patil A, Blicharz L, et al. Medical therapies for melasma. J Cosmet Dermatol. 2022;21:3707-3728.
- Suliman RS, Alhuwayshil J, Almuflehi AA, et al. Emerging topical therapies for melasma: a comparative analysis of efficacy and safety. J Dermatolog Treat. 2025;36:2591502.
- Austin E, Nguyen JK, Jagdeo J. Topical treatments for melasma: a systematic review of randomized controlled trials. J Drugs Dermatol. 2019;18:S1545961619P1156X.
- Grimes PE, Dias S, Oparaugo NC, et al. A randomized, controlled, split-face, double-blind study comparing topical malassezin to hydroquinone 4% for melasma. J Drugs Dermatol. 2026;25:25-29.
- Passeron T, Kerob D, Le Dantec G, et al. Efficacy and tolerability of a new facial 2-mercaptonicotinoyl glycine-containing depigmenting serum versus hydroquinone 4% over 3-month treatment of facial melasma. Dermatol Ther (Heidelb). 2025;15:2379-2390.
- McKesey J, Tovar-Garza A, Pandya AG. Melasma treatment: an evidence-based review. Am J Clin Dermatol. 2020;21:173-225.
- Bala HR, Lee S, Wong C, et al. Oral tranexamic acid for the treatment of melasma: a review. Dermatol Surg. 2018;44:814-825.
- Sarkar R, Katoch S. Chemical peels in treatment of melasma. Dermatol Clin. 2024;42:21-32.
- Sarkar R, Gupta M. Platelet-rich plasma in melasma—a systematic review. Dermatol Surg. 2022;48:131-134.
- Pongklaokam J, Manuskiatti W, Wanitphakdeedecha R, et al. Efficacy and safety of letibotulinum toxin A for the treatment of melasma in two different dilutions: a randomized double-blind split-face study. Toxins (Basel). 2025;17:349.
- Han HJ, Kim JC, Park YJ, et al. Targeting the dermis for melasma maintenance treatment. Sci Rep. 2024;14:949.
- Zhang AD, Lazar M, Akhundova E, et al. A scoping review on melasma treatments and their histopathologic correlates. Dermatopathology (Basel). 2025;12:13.
- Goldman MP, Wu DC, Khetarpal S, et al. Dual 1550-nm erbium glass fiber and 1927-nm thulium fiber non-ablative fractional laser system treatment in patients with skin of color: a review of clinical studies and unmet needs. Lasers Surg Med. Published online February 10, 2026.
- Chen J, Yu N, Peng L, et al. Efficacy of low-fluence 1064 nm Q-switched Nd: YAG laser for the treatment of melasma: a meta-analysis and systematic review. J Cosmet Dermatol. 2022;21.
- Feng J, Huang L. Comparison of picosecond and nanosecond Nd:YAG 1064-nm lasers in the treatment of melasma: a split-face randomized clinical trial. Plast Reconstr Surg. 2023;151:772-777.
- Passeron T, Genedy R, Salah L, et al., Laser treatment of hyperpigmented lesions: position statement of the European Society of Laser in Dermatology. J Eur Acad Dermatol Venereol. 2019;33:987-1005.
Beyond Conventional Paradigms: Rethinking the Evaluation and Management of Melasma
Beyond Conventional Paradigms: Rethinking the Evaluation and Management of Melasma