User login
Man with distal flexion deformities
On the basis of history and presentation, this patient's psoriatic disease has probably evolved to psoriatic arthritis mutilans (PAM). PAM is considered the most severe form of psoriatic arthritis (PsA), causing joint destruction and functional disability. It is estimated to affect about 5% of patients with PsA, with an equal sex distribution. Psoriatic nail dystrophy, a hallmark of PsA, appears to be a clinical biomarker of PAM development. Patients with PAM are generally younger at diagnosis than those with less severe forms of disease. Disease-modifying antirheumatic drugs and anti-TNF therapy do not appear to prevent the development of PAM, as evidenced by the present case.
In general, clinical presentation of PsA is heterogeneous and can be similar to that of other rheumatic diseases such as rheumatoid arthritis or osteoarthritis, complicating the differential diagnosis. The Classification Criteria for Psoriatic Arthritis (CASPAR) are considered the most sensitive diagnostic criteria, encompassing evidence of psoriasis; nail dystrophy; lab findings of typical autoantibodies (negative rheumatoid factor); and phenomena that are characteristic of PsA, like dactylitis.
Workup for PAM often includes radiography, ultrasound, and MRI or CT. With no established consensus, classification systems for the condition vary clinically and radiographically. Radiographic features suggestive of PAM include osteolysis or extended bone resorption; pencil-in-cup changes; joint subluxation; and, less often, ankylosis. Osteolysis has been defined as bone resorption with more than 50% loss of joint surface on both sides of the joint. Clinically, dissolution of the joint causes redundant, overlying skin with a telescoping motion of the digit. Other clinical features of PAM include digital shortening and flail joints. Of note, involvement of one small joint in the hands or feet is diagnostic of PAM.
In the setting of PsA, multiple genetic factors have been described, including presence of HLA-B27 and HLA-DRB1, but none are considered defining factors for the disease. A recent population-based study shows that presence of HLA-B27 was significantly increased among patients with PAM (45%) compared with patients with less severe PsA (13%) and healthy controls (13%).
According to the American College of Rheumatology guidelines, first-line therapy in adult patients who have active PsA and are treatment-naive is a TNFi biologic agent. For the patient in this case, who has active PsA despite treatment with TNFi biologic monotherapy, switching to a different TNFi biologic may be appropriate; however, switching to an interleukin-17 inhibitor may also be considered because this patient has severe disease. Data on the comparative efficacy of different biological agents for treatment of PAM are not yet available.
Herbert S. Diamond, MD, Professor of Medicine (retired), Temple University School of Medicine, University of Pittsburgh; Chairman, Department of Medicine Emeritus, Western Pennsylvania Hospital, Pittsburgh, PA.
Herbert S. Diamond, MD, has disclosed no relevant financial relationships.
Image Quizzes are fictional or fictionalized clinical scenarios intended to provide evidence-based educational takeaways.
On the basis of history and presentation, this patient's psoriatic disease has probably evolved to psoriatic arthritis mutilans (PAM). PAM is considered the most severe form of psoriatic arthritis (PsA), causing joint destruction and functional disability. It is estimated to affect about 5% of patients with PsA, with an equal sex distribution. Psoriatic nail dystrophy, a hallmark of PsA, appears to be a clinical biomarker of PAM development. Patients with PAM are generally younger at diagnosis than those with less severe forms of disease. Disease-modifying antirheumatic drugs and anti-TNF therapy do not appear to prevent the development of PAM, as evidenced by the present case.
In general, clinical presentation of PsA is heterogeneous and can be similar to that of other rheumatic diseases such as rheumatoid arthritis or osteoarthritis, complicating the differential diagnosis. The Classification Criteria for Psoriatic Arthritis (CASPAR) are considered the most sensitive diagnostic criteria, encompassing evidence of psoriasis; nail dystrophy; lab findings of typical autoantibodies (negative rheumatoid factor); and phenomena that are characteristic of PsA, like dactylitis.
Workup for PAM often includes radiography, ultrasound, and MRI or CT. With no established consensus, classification systems for the condition vary clinically and radiographically. Radiographic features suggestive of PAM include osteolysis or extended bone resorption; pencil-in-cup changes; joint subluxation; and, less often, ankylosis. Osteolysis has been defined as bone resorption with more than 50% loss of joint surface on both sides of the joint. Clinically, dissolution of the joint causes redundant, overlying skin with a telescoping motion of the digit. Other clinical features of PAM include digital shortening and flail joints. Of note, involvement of one small joint in the hands or feet is diagnostic of PAM.
In the setting of PsA, multiple genetic factors have been described, including presence of HLA-B27 and HLA-DRB1, but none are considered defining factors for the disease. A recent population-based study shows that presence of HLA-B27 was significantly increased among patients with PAM (45%) compared with patients with less severe PsA (13%) and healthy controls (13%).
According to the American College of Rheumatology guidelines, first-line therapy in adult patients who have active PsA and are treatment-naive is a TNFi biologic agent. For the patient in this case, who has active PsA despite treatment with TNFi biologic monotherapy, switching to a different TNFi biologic may be appropriate; however, switching to an interleukin-17 inhibitor may also be considered because this patient has severe disease. Data on the comparative efficacy of different biological agents for treatment of PAM are not yet available.
Herbert S. Diamond, MD, Professor of Medicine (retired), Temple University School of Medicine, University of Pittsburgh; Chairman, Department of Medicine Emeritus, Western Pennsylvania Hospital, Pittsburgh, PA.
Herbert S. Diamond, MD, has disclosed no relevant financial relationships.
Image Quizzes are fictional or fictionalized clinical scenarios intended to provide evidence-based educational takeaways.
On the basis of history and presentation, this patient's psoriatic disease has probably evolved to psoriatic arthritis mutilans (PAM). PAM is considered the most severe form of psoriatic arthritis (PsA), causing joint destruction and functional disability. It is estimated to affect about 5% of patients with PsA, with an equal sex distribution. Psoriatic nail dystrophy, a hallmark of PsA, appears to be a clinical biomarker of PAM development. Patients with PAM are generally younger at diagnosis than those with less severe forms of disease. Disease-modifying antirheumatic drugs and anti-TNF therapy do not appear to prevent the development of PAM, as evidenced by the present case.
In general, clinical presentation of PsA is heterogeneous and can be similar to that of other rheumatic diseases such as rheumatoid arthritis or osteoarthritis, complicating the differential diagnosis. The Classification Criteria for Psoriatic Arthritis (CASPAR) are considered the most sensitive diagnostic criteria, encompassing evidence of psoriasis; nail dystrophy; lab findings of typical autoantibodies (negative rheumatoid factor); and phenomena that are characteristic of PsA, like dactylitis.
Workup for PAM often includes radiography, ultrasound, and MRI or CT. With no established consensus, classification systems for the condition vary clinically and radiographically. Radiographic features suggestive of PAM include osteolysis or extended bone resorption; pencil-in-cup changes; joint subluxation; and, less often, ankylosis. Osteolysis has been defined as bone resorption with more than 50% loss of joint surface on both sides of the joint. Clinically, dissolution of the joint causes redundant, overlying skin with a telescoping motion of the digit. Other clinical features of PAM include digital shortening and flail joints. Of note, involvement of one small joint in the hands or feet is diagnostic of PAM.
In the setting of PsA, multiple genetic factors have been described, including presence of HLA-B27 and HLA-DRB1, but none are considered defining factors for the disease. A recent population-based study shows that presence of HLA-B27 was significantly increased among patients with PAM (45%) compared with patients with less severe PsA (13%) and healthy controls (13%).
According to the American College of Rheumatology guidelines, first-line therapy in adult patients who have active PsA and are treatment-naive is a TNFi biologic agent. For the patient in this case, who has active PsA despite treatment with TNFi biologic monotherapy, switching to a different TNFi biologic may be appropriate; however, switching to an interleukin-17 inhibitor may also be considered because this patient has severe disease. Data on the comparative efficacy of different biological agents for treatment of PAM are not yet available.
Herbert S. Diamond, MD, Professor of Medicine (retired), Temple University School of Medicine, University of Pittsburgh; Chairman, Department of Medicine Emeritus, Western Pennsylvania Hospital, Pittsburgh, PA.
Herbert S. Diamond, MD, has disclosed no relevant financial relationships.
Image Quizzes are fictional or fictionalized clinical scenarios intended to provide evidence-based educational takeaways.
A 43-year-old man presents with distal flexion deformities and telescoping of the digits. The patient was diagnosed with psoriasis at age 31 and he has several immediate family members who previously received the same diagnosis. He has been treated intermittently with tumor necrosis factor inhibitor (TNFi) biologic monotherapy but admits to nonadherence when disease activity seems to quiet down. Radiography shows osteolysis and dissolution of the joint.
Hormones account for 10% of lipid changes after menopause
The transition from perimenopause to menopause is accompanied by a proatherogenic shift in lipids and other circulating metabolites that potentially predispose women to cardiovascular disease (CVD). Now, for the first time, a new prospective cohort study quantifies the link between hormonal shifts and these lipid changes.
However, hormone therapy (HT) somewhat mitigates the shift and may help protect menopausal women from some elevated CVD risk, the same study suggests.
“Menopause is not avoidable, but perhaps the negative metabolite shift can be diminished by lifestyle choices such as eating healthily and being physically active,” senior author Eija Laakkonen, MD, University of Jyväskylä, Finland, told this news organization in an email.
“And women should especially pay attention to the quality of dietary fats and amount of exercise [they get] to maintain cardiorespiratory fitness,” she said, adding that women should discuss the option of HT with their health care providers.
Asked to comment, JoAnn Manson, MD, of Harvard Medical School, Boston, and past president of the North American Menopause Society, said there is strong evidence that women undergo negative cardiometabolic changes during the menopausal transition.
Changes include those in body composition (an increase in visceral fat and waist circumference), as well as unfavorable shifts in the lipid profile, as reflected by increases in low-density lipoprotein cholesterol (LDL-C) and triglycerides and a decrease in high-density lipoprotein cholesterol (HDL-C).
It’s also clear from a variety of cohort studies that HT blunts menopausal-related increases in body weight, percentage of body fat, as well as visceral fat, she said.
So the new findings do seem to “parallel” those of other perimenopausal to menopausal transition studies, which include HT having “favorable effects on lipids,” Dr. Manson said. HT “lowers LDL-C and increases HDL-C, and this is especially true when it is given orally,” but even transdermal delivery has shown some benefits, she observed.
Shift in hormones causes 10% of lipid changes after menopause
The new study, by Jari E. Karppinen, also of the University of Jyväskylä, and colleagues, was recently published in the European Journal of Preventive Cardiology. The data are from the Estrogenic Regulation of Muscle Apoptosis (ERMA) prospective cohort study.
In total, 218 women were tracked from perimenopause through to early postmenopause, 35 of whom started HT, mostly oral preparations. The women were followed for a median of 14 months. Their mean age was 51.7 years when their hormone and metabolite profiles were first measured.
Previous studies have shown that menopause is associated with levels of metabolites that promote CVD, but this study is the first to specifically link this shift with changes in female sex hormones, the researchers stress.
“Menopause was associated with a statistically significant change in 85 metabolite measures,” Mr. Karppinen and colleagues report.
Analyses showed that the menopausal hormonal shift directly explained the change in 64 of the 85 metabolites, with effect sizes ranging from 2.1% to 11.2%.
These included increases in LDL-C, triglycerides, and fatty acids. Analyses were adjusted for age at baseline, duration of follow-up, education level, smoking status, alcohol use, physical activity, and diet quality.
More specifically, investigators found that all apoB-containing particle counts as well as particle diameters increased over follow-up, although no change occurred in HDL particles.
They also found cholesterol concentrations in all apoB-containing lipoprotein classes to increase and triglyceride concentrations to increase in very low-density lipoprotein and HDL particles.
“These findings, including HDL triglycerides, can be interpreted as signs of poor metabolic health since, despite higher HDL-C being good for health, high HDL triglyceride levels are associated with a higher risk of coronary heart disease,” Dr. Laakkonen emphasized.
Among the 35 women who initiated HT on study enrollment, investigators did note, on exploratory analysis, increases in HDL-C and reductions in LDL-C.
“The number of women starting HT was small, and the type of HT was not controlled,” Dr. Laakkonen cautioned, however.
“Nevertheless, our observations support clinical guidelines to initiate HT early into menopause, as this timing offers the greatest cardioprotective effect,” she added.
The study was supported by the Academy of Finland. The authors and Dr. Manson have reported no relevant financial relationships. Dr. Manson is a contributor to Medscape.
This article was updated on 5/20/2022.
A version of this article first appeared on Medscape.com.
The transition from perimenopause to menopause is accompanied by a proatherogenic shift in lipids and other circulating metabolites that potentially predispose women to cardiovascular disease (CVD). Now, for the first time, a new prospective cohort study quantifies the link between hormonal shifts and these lipid changes.
However, hormone therapy (HT) somewhat mitigates the shift and may help protect menopausal women from some elevated CVD risk, the same study suggests.
“Menopause is not avoidable, but perhaps the negative metabolite shift can be diminished by lifestyle choices such as eating healthily and being physically active,” senior author Eija Laakkonen, MD, University of Jyväskylä, Finland, told this news organization in an email.
“And women should especially pay attention to the quality of dietary fats and amount of exercise [they get] to maintain cardiorespiratory fitness,” she said, adding that women should discuss the option of HT with their health care providers.
Asked to comment, JoAnn Manson, MD, of Harvard Medical School, Boston, and past president of the North American Menopause Society, said there is strong evidence that women undergo negative cardiometabolic changes during the menopausal transition.
Changes include those in body composition (an increase in visceral fat and waist circumference), as well as unfavorable shifts in the lipid profile, as reflected by increases in low-density lipoprotein cholesterol (LDL-C) and triglycerides and a decrease in high-density lipoprotein cholesterol (HDL-C).
It’s also clear from a variety of cohort studies that HT blunts menopausal-related increases in body weight, percentage of body fat, as well as visceral fat, she said.
So the new findings do seem to “parallel” those of other perimenopausal to menopausal transition studies, which include HT having “favorable effects on lipids,” Dr. Manson said. HT “lowers LDL-C and increases HDL-C, and this is especially true when it is given orally,” but even transdermal delivery has shown some benefits, she observed.
Shift in hormones causes 10% of lipid changes after menopause
The new study, by Jari E. Karppinen, also of the University of Jyväskylä, and colleagues, was recently published in the European Journal of Preventive Cardiology. The data are from the Estrogenic Regulation of Muscle Apoptosis (ERMA) prospective cohort study.
In total, 218 women were tracked from perimenopause through to early postmenopause, 35 of whom started HT, mostly oral preparations. The women were followed for a median of 14 months. Their mean age was 51.7 years when their hormone and metabolite profiles were first measured.
Previous studies have shown that menopause is associated with levels of metabolites that promote CVD, but this study is the first to specifically link this shift with changes in female sex hormones, the researchers stress.
“Menopause was associated with a statistically significant change in 85 metabolite measures,” Mr. Karppinen and colleagues report.
Analyses showed that the menopausal hormonal shift directly explained the change in 64 of the 85 metabolites, with effect sizes ranging from 2.1% to 11.2%.
These included increases in LDL-C, triglycerides, and fatty acids. Analyses were adjusted for age at baseline, duration of follow-up, education level, smoking status, alcohol use, physical activity, and diet quality.
More specifically, investigators found that all apoB-containing particle counts as well as particle diameters increased over follow-up, although no change occurred in HDL particles.
They also found cholesterol concentrations in all apoB-containing lipoprotein classes to increase and triglyceride concentrations to increase in very low-density lipoprotein and HDL particles.
“These findings, including HDL triglycerides, can be interpreted as signs of poor metabolic health since, despite higher HDL-C being good for health, high HDL triglyceride levels are associated with a higher risk of coronary heart disease,” Dr. Laakkonen emphasized.
Among the 35 women who initiated HT on study enrollment, investigators did note, on exploratory analysis, increases in HDL-C and reductions in LDL-C.
“The number of women starting HT was small, and the type of HT was not controlled,” Dr. Laakkonen cautioned, however.
“Nevertheless, our observations support clinical guidelines to initiate HT early into menopause, as this timing offers the greatest cardioprotective effect,” she added.
The study was supported by the Academy of Finland. The authors and Dr. Manson have reported no relevant financial relationships. Dr. Manson is a contributor to Medscape.
This article was updated on 5/20/2022.
A version of this article first appeared on Medscape.com.
The transition from perimenopause to menopause is accompanied by a proatherogenic shift in lipids and other circulating metabolites that potentially predispose women to cardiovascular disease (CVD). Now, for the first time, a new prospective cohort study quantifies the link between hormonal shifts and these lipid changes.
However, hormone therapy (HT) somewhat mitigates the shift and may help protect menopausal women from some elevated CVD risk, the same study suggests.
“Menopause is not avoidable, but perhaps the negative metabolite shift can be diminished by lifestyle choices such as eating healthily and being physically active,” senior author Eija Laakkonen, MD, University of Jyväskylä, Finland, told this news organization in an email.
“And women should especially pay attention to the quality of dietary fats and amount of exercise [they get] to maintain cardiorespiratory fitness,” she said, adding that women should discuss the option of HT with their health care providers.
Asked to comment, JoAnn Manson, MD, of Harvard Medical School, Boston, and past president of the North American Menopause Society, said there is strong evidence that women undergo negative cardiometabolic changes during the menopausal transition.
Changes include those in body composition (an increase in visceral fat and waist circumference), as well as unfavorable shifts in the lipid profile, as reflected by increases in low-density lipoprotein cholesterol (LDL-C) and triglycerides and a decrease in high-density lipoprotein cholesterol (HDL-C).
It’s also clear from a variety of cohort studies that HT blunts menopausal-related increases in body weight, percentage of body fat, as well as visceral fat, she said.
So the new findings do seem to “parallel” those of other perimenopausal to menopausal transition studies, which include HT having “favorable effects on lipids,” Dr. Manson said. HT “lowers LDL-C and increases HDL-C, and this is especially true when it is given orally,” but even transdermal delivery has shown some benefits, she observed.
Shift in hormones causes 10% of lipid changes after menopause
The new study, by Jari E. Karppinen, also of the University of Jyväskylä, and colleagues, was recently published in the European Journal of Preventive Cardiology. The data are from the Estrogenic Regulation of Muscle Apoptosis (ERMA) prospective cohort study.
In total, 218 women were tracked from perimenopause through to early postmenopause, 35 of whom started HT, mostly oral preparations. The women were followed for a median of 14 months. Their mean age was 51.7 years when their hormone and metabolite profiles were first measured.
Previous studies have shown that menopause is associated with levels of metabolites that promote CVD, but this study is the first to specifically link this shift with changes in female sex hormones, the researchers stress.
“Menopause was associated with a statistically significant change in 85 metabolite measures,” Mr. Karppinen and colleagues report.
Analyses showed that the menopausal hormonal shift directly explained the change in 64 of the 85 metabolites, with effect sizes ranging from 2.1% to 11.2%.
These included increases in LDL-C, triglycerides, and fatty acids. Analyses were adjusted for age at baseline, duration of follow-up, education level, smoking status, alcohol use, physical activity, and diet quality.
More specifically, investigators found that all apoB-containing particle counts as well as particle diameters increased over follow-up, although no change occurred in HDL particles.
They also found cholesterol concentrations in all apoB-containing lipoprotein classes to increase and triglyceride concentrations to increase in very low-density lipoprotein and HDL particles.
“These findings, including HDL triglycerides, can be interpreted as signs of poor metabolic health since, despite higher HDL-C being good for health, high HDL triglyceride levels are associated with a higher risk of coronary heart disease,” Dr. Laakkonen emphasized.
Among the 35 women who initiated HT on study enrollment, investigators did note, on exploratory analysis, increases in HDL-C and reductions in LDL-C.
“The number of women starting HT was small, and the type of HT was not controlled,” Dr. Laakkonen cautioned, however.
“Nevertheless, our observations support clinical guidelines to initiate HT early into menopause, as this timing offers the greatest cardioprotective effect,” she added.
The study was supported by the Academy of Finland. The authors and Dr. Manson have reported no relevant financial relationships. Dr. Manson is a contributor to Medscape.
This article was updated on 5/20/2022.
A version of this article first appeared on Medscape.com.
FROM THE EUROPEAN JOURNAL OF PREVENTIVE CARDIOLOGY
Improved cancer survival in states with ACA Medicaid expansion
compared with patients in states that did not adopt the expansion.
The finding comes from an American Cancer Society study of more than 2 million patients with newly diagnosed cancer, published online in the Journal of the National Cancer Institute.
The analysis also showed that the evidence was strongest for malignancies with poor prognosis such as lung, pancreatic, and liver cancer, and also for colorectal cancer.
Importantly, improvements in survival were larger in non-Hispanic Black patients and individuals residing in rural areas, suggesting there was a narrowing of disparities in cancer survival by race and rurality.
“Our findings provide further evidence of the importance of expanding Medicaid eligibility in all states, particularly considering the economic crisis and health care disruptions caused by the COVID-19 pandemic,” said lead author Xuesong Han, PhD, scientific director of health services research at the American Cancer Society, in a statement. “What’s encouraging is the American Rescue Plan Act of 2021 provides new incentives for Medicaid expansion in states that have yet to increase eligibility.”
The ACA provided states with incentives to expand Medicaid eligibility to all low-income adults under 138% federal poverty level, regardless of parental status.
As of last month, just 12 states have not yet opted for Medicaid expansion, even though the American Rescue Plan Act of 2021 provides new incentives for those remaining jurisdictions. But to date, none of the remaining states have taken advantage of these new incentives.
An interactive map showing the status of Medicare expansion by state is available here. The 12 states that have not adopted Medicare expansion (as of April) are Alabama, Florida, Georgia, Kansas, Mississippi, North Carolina, South Carolina, South Dakota, Tennessee, Texas, Wisconsin, and Wyoming.
The benefit of Medicaid expansion on cancer outcomes has already been observed in other studies. The first study to show a survival benefit was presented at the 2020 American Society of Clinical Oncology annual meeting. That analysis showed that cancer mortality declined by 29% in states that expanded Medicaid and by 25% in those that did not. The authors also noted that the greatest mortality benefit was observed in Hispanic patients.
Improved survival with expansion
In the current paper, Dr. Han and colleagues used population-based cancer registries from 42 states and compared data on patients aged 18-62 years who were diagnosed with cancer in a period of 2 years before (2010-2012) and after (2014-2016) ACA Medicaid expansion. They were followed through Sept. 30, 2013, and Dec. 31, 2017, respectively.
The analysis involved a total of 2.5 million patients, of whom 1.52 million lived in states that adopted Medicaid expansion and compared with 1 million patients were in states that did not.
Patients with grouped by sex, race and ethnicity, census tract-level poverty, and rurality. The authors note that non-Hispanic Black patients and those from high poverty areas and nonmetropolitan areas were disproportionately represented in nonexpansion states.
During the 2-year follow-up period, a total of 453,487 deaths occurred (257,950 in expansion states and 195,537 in nonexpansion states).
Overall, patients in expansion states generally had better survival versus those in nonexpansion states, the authors comment. However, for most cancer types, overall survival improved after the ACA for both groups of states.
The 2-year overall survival increased from 80.6% before the ACA to 82.2% post ACA in expansion states and from 78.7% to 80% in nonexpansion states.
This extrapolated to net increase of 0.44 percentage points in expansion states after adjusting for sociodemographic factors. By cancer site, the net increase was greater for colorectal cancer, lung cancer, non-Hodgkin’s lymphoma, pancreatic cancer, and liver cancer.
For Hispanic patients, 2-year survival also increased but was similar in expansion and nonexpansion states, and little net change was associated with Medicaid expansion.
“Our study shows that the increase was largely driven by improvements in survival for cancer types with poor prognosis, suggesting improved access to timely and effective treatments,” said Dr. Han. “It adds to accumulating evidence of the multiple benefits of Medicaid expansion.”
A version of this article first appeared on Medscape.com.
compared with patients in states that did not adopt the expansion.
The finding comes from an American Cancer Society study of more than 2 million patients with newly diagnosed cancer, published online in the Journal of the National Cancer Institute.
The analysis also showed that the evidence was strongest for malignancies with poor prognosis such as lung, pancreatic, and liver cancer, and also for colorectal cancer.
Importantly, improvements in survival were larger in non-Hispanic Black patients and individuals residing in rural areas, suggesting there was a narrowing of disparities in cancer survival by race and rurality.
“Our findings provide further evidence of the importance of expanding Medicaid eligibility in all states, particularly considering the economic crisis and health care disruptions caused by the COVID-19 pandemic,” said lead author Xuesong Han, PhD, scientific director of health services research at the American Cancer Society, in a statement. “What’s encouraging is the American Rescue Plan Act of 2021 provides new incentives for Medicaid expansion in states that have yet to increase eligibility.”
The ACA provided states with incentives to expand Medicaid eligibility to all low-income adults under 138% federal poverty level, regardless of parental status.
As of last month, just 12 states have not yet opted for Medicaid expansion, even though the American Rescue Plan Act of 2021 provides new incentives for those remaining jurisdictions. But to date, none of the remaining states have taken advantage of these new incentives.
An interactive map showing the status of Medicare expansion by state is available here. The 12 states that have not adopted Medicare expansion (as of April) are Alabama, Florida, Georgia, Kansas, Mississippi, North Carolina, South Carolina, South Dakota, Tennessee, Texas, Wisconsin, and Wyoming.
The benefit of Medicaid expansion on cancer outcomes has already been observed in other studies. The first study to show a survival benefit was presented at the 2020 American Society of Clinical Oncology annual meeting. That analysis showed that cancer mortality declined by 29% in states that expanded Medicaid and by 25% in those that did not. The authors also noted that the greatest mortality benefit was observed in Hispanic patients.
Improved survival with expansion
In the current paper, Dr. Han and colleagues used population-based cancer registries from 42 states and compared data on patients aged 18-62 years who were diagnosed with cancer in a period of 2 years before (2010-2012) and after (2014-2016) ACA Medicaid expansion. They were followed through Sept. 30, 2013, and Dec. 31, 2017, respectively.
The analysis involved a total of 2.5 million patients, of whom 1.52 million lived in states that adopted Medicaid expansion and compared with 1 million patients were in states that did not.
Patients with grouped by sex, race and ethnicity, census tract-level poverty, and rurality. The authors note that non-Hispanic Black patients and those from high poverty areas and nonmetropolitan areas were disproportionately represented in nonexpansion states.
During the 2-year follow-up period, a total of 453,487 deaths occurred (257,950 in expansion states and 195,537 in nonexpansion states).
Overall, patients in expansion states generally had better survival versus those in nonexpansion states, the authors comment. However, for most cancer types, overall survival improved after the ACA for both groups of states.
The 2-year overall survival increased from 80.6% before the ACA to 82.2% post ACA in expansion states and from 78.7% to 80% in nonexpansion states.
This extrapolated to net increase of 0.44 percentage points in expansion states after adjusting for sociodemographic factors. By cancer site, the net increase was greater for colorectal cancer, lung cancer, non-Hodgkin’s lymphoma, pancreatic cancer, and liver cancer.
For Hispanic patients, 2-year survival also increased but was similar in expansion and nonexpansion states, and little net change was associated with Medicaid expansion.
“Our study shows that the increase was largely driven by improvements in survival for cancer types with poor prognosis, suggesting improved access to timely and effective treatments,” said Dr. Han. “It adds to accumulating evidence of the multiple benefits of Medicaid expansion.”
A version of this article first appeared on Medscape.com.
compared with patients in states that did not adopt the expansion.
The finding comes from an American Cancer Society study of more than 2 million patients with newly diagnosed cancer, published online in the Journal of the National Cancer Institute.
The analysis also showed that the evidence was strongest for malignancies with poor prognosis such as lung, pancreatic, and liver cancer, and also for colorectal cancer.
Importantly, improvements in survival were larger in non-Hispanic Black patients and individuals residing in rural areas, suggesting there was a narrowing of disparities in cancer survival by race and rurality.
“Our findings provide further evidence of the importance of expanding Medicaid eligibility in all states, particularly considering the economic crisis and health care disruptions caused by the COVID-19 pandemic,” said lead author Xuesong Han, PhD, scientific director of health services research at the American Cancer Society, in a statement. “What’s encouraging is the American Rescue Plan Act of 2021 provides new incentives for Medicaid expansion in states that have yet to increase eligibility.”
The ACA provided states with incentives to expand Medicaid eligibility to all low-income adults under 138% federal poverty level, regardless of parental status.
As of last month, just 12 states have not yet opted for Medicaid expansion, even though the American Rescue Plan Act of 2021 provides new incentives for those remaining jurisdictions. But to date, none of the remaining states have taken advantage of these new incentives.
An interactive map showing the status of Medicare expansion by state is available here. The 12 states that have not adopted Medicare expansion (as of April) are Alabama, Florida, Georgia, Kansas, Mississippi, North Carolina, South Carolina, South Dakota, Tennessee, Texas, Wisconsin, and Wyoming.
The benefit of Medicaid expansion on cancer outcomes has already been observed in other studies. The first study to show a survival benefit was presented at the 2020 American Society of Clinical Oncology annual meeting. That analysis showed that cancer mortality declined by 29% in states that expanded Medicaid and by 25% in those that did not. The authors also noted that the greatest mortality benefit was observed in Hispanic patients.
Improved survival with expansion
In the current paper, Dr. Han and colleagues used population-based cancer registries from 42 states and compared data on patients aged 18-62 years who were diagnosed with cancer in a period of 2 years before (2010-2012) and after (2014-2016) ACA Medicaid expansion. They were followed through Sept. 30, 2013, and Dec. 31, 2017, respectively.
The analysis involved a total of 2.5 million patients, of whom 1.52 million lived in states that adopted Medicaid expansion and compared with 1 million patients were in states that did not.
Patients with grouped by sex, race and ethnicity, census tract-level poverty, and rurality. The authors note that non-Hispanic Black patients and those from high poverty areas and nonmetropolitan areas were disproportionately represented in nonexpansion states.
During the 2-year follow-up period, a total of 453,487 deaths occurred (257,950 in expansion states and 195,537 in nonexpansion states).
Overall, patients in expansion states generally had better survival versus those in nonexpansion states, the authors comment. However, for most cancer types, overall survival improved after the ACA for both groups of states.
The 2-year overall survival increased from 80.6% before the ACA to 82.2% post ACA in expansion states and from 78.7% to 80% in nonexpansion states.
This extrapolated to net increase of 0.44 percentage points in expansion states after adjusting for sociodemographic factors. By cancer site, the net increase was greater for colorectal cancer, lung cancer, non-Hodgkin’s lymphoma, pancreatic cancer, and liver cancer.
For Hispanic patients, 2-year survival also increased but was similar in expansion and nonexpansion states, and little net change was associated with Medicaid expansion.
“Our study shows that the increase was largely driven by improvements in survival for cancer types with poor prognosis, suggesting improved access to timely and effective treatments,” said Dr. Han. “It adds to accumulating evidence of the multiple benefits of Medicaid expansion.”
A version of this article first appeared on Medscape.com.
Steroid use associated with higher relapse risk in some UC
Patients who have histologically active ulcerative colitis (UC) with a Mayo endoscopic subscore (MES) of 1 and a history of steroid use may be at increased risk for clinical relapse, according to a new single-center, retrospective analysis.
In recent years, treat-to-target approaches in UC have incorporated clinician and patient-reported outcomes, along with endoscopic remission, defined as MES 1 or less. However, additional studies showed a higher risk of relapse with MES 1, and the STRIDE-II (Selecting Therapeutic Targets in Inflammatory Bowel Disease) guidelines released in 2021 suggest that only MES 0 indicates endoscopic healing.
Nevertheless, there is concern that striving to achieve MES 0 could lead to overtreatment, since MES values are somewhat subjective, and patients with MES 1 sometimes report few or no symptoms. This led to the current study by Gyeol Seong, MD, and colleagues, published in Inflammatory Bowel Diseases.
Commenting on the study, Miguel Regueiro, MD, chair for the Digestive Disease and Surgery Institute and a professor of medicine at the Cleveland Clinic said “the question has always been, if the patient is in endoscopic remission and clinically feels well, how important is it to have histologically normal mucosa?”
In their retrospective study, Dr. Seong and colleagues analyzed data from 492 patients. The median age was 48, and 51.6% were male. The median duration of disease was 78 months. During a median follow-up of 549 days, 18.7% experienced a relapse, defined as “change or escalation of medication, hospitalization due to the aggravation of UC, or total colectomy.” Of these patients, 39.4% had used steroids and 51.4% had a MES score of 0 at the index endoscopy, and 70.5% had histologic improvement, as defined by a Geboes score of less than 3.1.
A Geboes score of 3.1 or higher and a history of steroid use was associated with an increased cumulative incidence of clinical relapse, compared with a Geboes score lower than 3.1 and no steroid use (P = .001). In a multivariate analysis, histologic activity alone was not a predictor of relapse. Among patients with an MES score of 1, a history of steroid use predicted risk of relapse (adjusted hazard ratio, 2.102; P = .006).
Although Dr. Regueiro said the new findings would not change his current practice, he does incorporate histopathology in clinical decision-making. In cases where a patient is in endoscopic remission but has histologic activity, he will consider adjusting the current medication rather than changing to a different therapeutic. “I wouldn’t recommend that we switch patients off one therapy to another just because of histologic activity.”
That’s because patients often improve dramatically after effective therapy. Dr. Regueiro said: “The one concern is whether the next medicine works as well as what the patient is already on? I’m also worried that we burn through our biologics too quickly. We go from one to another and another, sometimes I think we just need to optimize the one they’re on.”
The study does have the potential to influence surveillance for dysplasia, according to Dr. Regueiro, noting that a recent retrospective analysis showed an increased risk of dysplasia in UC with persistent histologic activity. “This suggests the need for a colonoscopy in a patient who has had ulcerative colitis for a number of years, even if there’s improvement but, on biopsy, there’s evidence of inflammation.”
According to Dr. Regueiro, such patients might benefit from a fecal calprotectin measurement in 3-6 months to monitor for colonic inflammation. Increased levels might be a sign that the patient is skipping medication doses, although other factors, like respiratory infections, can also explain the results. If the patient’s medication adherence is good, another therapy can be added or the dose increased, and the next endoscopy can be pushed up.
A key limitation to the study by Dr. Seong and colleagues is that it was based in South Korea. “Would this be the same in different countries and different populations? That is still a question,” said Dr. Regueiro.
Dr. Regueiro has received unrestricted educational grants from Abbvie, Janssen, UCB, Pfizer, Takeda, Celgene, Genentech, Gilead, Bristol-Myers Squibb, and Lilly. He has been on advisory boards or consulted for numerous companies. He also has relationships with the following CME companies: CME Outfitters, Imedex, GI Health Foundation, Cornerstones, Remedy, MJH Life Sciences, Medscape, MDEducation, WebMD, and HMPGlobal.
Patients who have histologically active ulcerative colitis (UC) with a Mayo endoscopic subscore (MES) of 1 and a history of steroid use may be at increased risk for clinical relapse, according to a new single-center, retrospective analysis.
In recent years, treat-to-target approaches in UC have incorporated clinician and patient-reported outcomes, along with endoscopic remission, defined as MES 1 or less. However, additional studies showed a higher risk of relapse with MES 1, and the STRIDE-II (Selecting Therapeutic Targets in Inflammatory Bowel Disease) guidelines released in 2021 suggest that only MES 0 indicates endoscopic healing.
Nevertheless, there is concern that striving to achieve MES 0 could lead to overtreatment, since MES values are somewhat subjective, and patients with MES 1 sometimes report few or no symptoms. This led to the current study by Gyeol Seong, MD, and colleagues, published in Inflammatory Bowel Diseases.
Commenting on the study, Miguel Regueiro, MD, chair for the Digestive Disease and Surgery Institute and a professor of medicine at the Cleveland Clinic said “the question has always been, if the patient is in endoscopic remission and clinically feels well, how important is it to have histologically normal mucosa?”
In their retrospective study, Dr. Seong and colleagues analyzed data from 492 patients. The median age was 48, and 51.6% were male. The median duration of disease was 78 months. During a median follow-up of 549 days, 18.7% experienced a relapse, defined as “change or escalation of medication, hospitalization due to the aggravation of UC, or total colectomy.” Of these patients, 39.4% had used steroids and 51.4% had a MES score of 0 at the index endoscopy, and 70.5% had histologic improvement, as defined by a Geboes score of less than 3.1.
A Geboes score of 3.1 or higher and a history of steroid use was associated with an increased cumulative incidence of clinical relapse, compared with a Geboes score lower than 3.1 and no steroid use (P = .001). In a multivariate analysis, histologic activity alone was not a predictor of relapse. Among patients with an MES score of 1, a history of steroid use predicted risk of relapse (adjusted hazard ratio, 2.102; P = .006).
Although Dr. Regueiro said the new findings would not change his current practice, he does incorporate histopathology in clinical decision-making. In cases where a patient is in endoscopic remission but has histologic activity, he will consider adjusting the current medication rather than changing to a different therapeutic. “I wouldn’t recommend that we switch patients off one therapy to another just because of histologic activity.”
That’s because patients often improve dramatically after effective therapy. Dr. Regueiro said: “The one concern is whether the next medicine works as well as what the patient is already on? I’m also worried that we burn through our biologics too quickly. We go from one to another and another, sometimes I think we just need to optimize the one they’re on.”
The study does have the potential to influence surveillance for dysplasia, according to Dr. Regueiro, noting that a recent retrospective analysis showed an increased risk of dysplasia in UC with persistent histologic activity. “This suggests the need for a colonoscopy in a patient who has had ulcerative colitis for a number of years, even if there’s improvement but, on biopsy, there’s evidence of inflammation.”
According to Dr. Regueiro, such patients might benefit from a fecal calprotectin measurement in 3-6 months to monitor for colonic inflammation. Increased levels might be a sign that the patient is skipping medication doses, although other factors, like respiratory infections, can also explain the results. If the patient’s medication adherence is good, another therapy can be added or the dose increased, and the next endoscopy can be pushed up.
A key limitation to the study by Dr. Seong and colleagues is that it was based in South Korea. “Would this be the same in different countries and different populations? That is still a question,” said Dr. Regueiro.
Dr. Regueiro has received unrestricted educational grants from Abbvie, Janssen, UCB, Pfizer, Takeda, Celgene, Genentech, Gilead, Bristol-Myers Squibb, and Lilly. He has been on advisory boards or consulted for numerous companies. He also has relationships with the following CME companies: CME Outfitters, Imedex, GI Health Foundation, Cornerstones, Remedy, MJH Life Sciences, Medscape, MDEducation, WebMD, and HMPGlobal.
Patients who have histologically active ulcerative colitis (UC) with a Mayo endoscopic subscore (MES) of 1 and a history of steroid use may be at increased risk for clinical relapse, according to a new single-center, retrospective analysis.
In recent years, treat-to-target approaches in UC have incorporated clinician and patient-reported outcomes, along with endoscopic remission, defined as MES 1 or less. However, additional studies showed a higher risk of relapse with MES 1, and the STRIDE-II (Selecting Therapeutic Targets in Inflammatory Bowel Disease) guidelines released in 2021 suggest that only MES 0 indicates endoscopic healing.
Nevertheless, there is concern that striving to achieve MES 0 could lead to overtreatment, since MES values are somewhat subjective, and patients with MES 1 sometimes report few or no symptoms. This led to the current study by Gyeol Seong, MD, and colleagues, published in Inflammatory Bowel Diseases.
Commenting on the study, Miguel Regueiro, MD, chair for the Digestive Disease and Surgery Institute and a professor of medicine at the Cleveland Clinic said “the question has always been, if the patient is in endoscopic remission and clinically feels well, how important is it to have histologically normal mucosa?”
In their retrospective study, Dr. Seong and colleagues analyzed data from 492 patients. The median age was 48, and 51.6% were male. The median duration of disease was 78 months. During a median follow-up of 549 days, 18.7% experienced a relapse, defined as “change or escalation of medication, hospitalization due to the aggravation of UC, or total colectomy.” Of these patients, 39.4% had used steroids and 51.4% had a MES score of 0 at the index endoscopy, and 70.5% had histologic improvement, as defined by a Geboes score of less than 3.1.
A Geboes score of 3.1 or higher and a history of steroid use was associated with an increased cumulative incidence of clinical relapse, compared with a Geboes score lower than 3.1 and no steroid use (P = .001). In a multivariate analysis, histologic activity alone was not a predictor of relapse. Among patients with an MES score of 1, a history of steroid use predicted risk of relapse (adjusted hazard ratio, 2.102; P = .006).
Although Dr. Regueiro said the new findings would not change his current practice, he does incorporate histopathology in clinical decision-making. In cases where a patient is in endoscopic remission but has histologic activity, he will consider adjusting the current medication rather than changing to a different therapeutic. “I wouldn’t recommend that we switch patients off one therapy to another just because of histologic activity.”
That’s because patients often improve dramatically after effective therapy. Dr. Regueiro said: “The one concern is whether the next medicine works as well as what the patient is already on? I’m also worried that we burn through our biologics too quickly. We go from one to another and another, sometimes I think we just need to optimize the one they’re on.”
The study does have the potential to influence surveillance for dysplasia, according to Dr. Regueiro, noting that a recent retrospective analysis showed an increased risk of dysplasia in UC with persistent histologic activity. “This suggests the need for a colonoscopy in a patient who has had ulcerative colitis for a number of years, even if there’s improvement but, on biopsy, there’s evidence of inflammation.”
According to Dr. Regueiro, such patients might benefit from a fecal calprotectin measurement in 3-6 months to monitor for colonic inflammation. Increased levels might be a sign that the patient is skipping medication doses, although other factors, like respiratory infections, can also explain the results. If the patient’s medication adherence is good, another therapy can be added or the dose increased, and the next endoscopy can be pushed up.
A key limitation to the study by Dr. Seong and colleagues is that it was based in South Korea. “Would this be the same in different countries and different populations? That is still a question,” said Dr. Regueiro.
Dr. Regueiro has received unrestricted educational grants from Abbvie, Janssen, UCB, Pfizer, Takeda, Celgene, Genentech, Gilead, Bristol-Myers Squibb, and Lilly. He has been on advisory boards or consulted for numerous companies. He also has relationships with the following CME companies: CME Outfitters, Imedex, GI Health Foundation, Cornerstones, Remedy, MJH Life Sciences, Medscape, MDEducation, WebMD, and HMPGlobal.
FROM INFLAMMATORY BOWEL DISEASES
NAVIGATOR steers uncontrolled asthma toward calmer seas
SAN FRANCISCO – Nearly half of all patients with severe, uncontrolled asthma who received a full course of the biologic agent tezepelumab (Tezspire) in the NAVIGATOR trial had a complete response to treatment at 1 year, results of a prespecified exploratory analysis indicated.
Among 471 patients assigned to tezepelumab who completed the on-treatment period of the phase 3 randomized trial, 46% had a complete response at 52 weeks, compared with 24% of patients assigned to placebo.
Complete response was defined as reduction in exacerbations of at least 50% over the previous year, improvement from baseline in Asthma Control Questionnaire 6 (ACQ-6) total score of at least 0.5 points, improvement in prebronchodilator forced expiratory volume in 1 second (pre-BD FEV1), and physician-assessed Clinical Global Impression measure of clinical change (CGI-C) score.
“These data further support the efficacy of tezepelumab in a broad population of patients with severe, uncontrolled asthma,” said Njira Lugogo, MD, of the division of pulmonary and critical care medicine at the University of Michigan, Ann Arbor.
Dr. Lugogo presented results of the exploratory analysis at the American Thoracic Society’s international conference.
Exacerbations reduced, lung function improved
Primary results from NAVIGATOR, published in The New England Journal of Medicine, showed that patients with severe, uncontrolled asthma randomly assigned to tezepelumab had fewer exacerbations and better lung function, asthma control, and health-related quality of life compared with patients assigned to placebo.
The investigators noted that approximately 10% of patients with asthma have symptoms and exacerbations despite maximal standard-of-care controller therapy.
Tezepelumab is a human monoclonal antibody that inhibits action of thymic stromal lymphopoietin (TSLP), an epithelial cytokine that is released in response to airborne triggers of asthma. TSLP is a major contributor to initiation and persistence of airway inflammation, Dr. Lugogo said.
The on-treatment analysis looked at all patients in the trial who completed 52 weeks of treatment and had complete data for all criteria studied.
The odds ratios (OR) for patients on tezepelumab achieving each of the response criteria are shown in the table.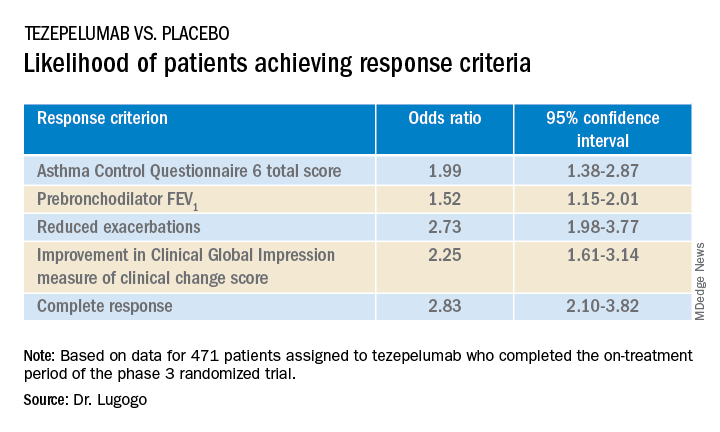
Exacerbations explored
In a separate presentation, Christopher S. Ambrose, MD, MBA, of AstraZeneca in Gaithersburg, Md., presented information from investigator-narrative descriptions of all hospitalization events related to asthma exacerbations (mild, moderate, or severe) that occurred while the investigator was blinded to each patient’s treatment assignment in NAVIGATOR.
In all, 39 of 531 patients (7.3%) assigned to placebo had a total of 78 exacerbations requiring hospitalization, compared with 13 of 528 patients (2.5%) assigned to tezepelumab. The latter group had a total of 14 exacerbations requiring hospitalization during the study.
Among hospitalized patients, 32 of the 39 assigned to placebo had severe, incapacitating exacerbations, compared with 5 of 13 assigned to tezepelumab.
Reported symptoms were generally similar between hospitalized patients in the two treatment groups, although there appeared to be trends toward lower incidence of dyspnea, fever, and tachycardia with tezepelumab.
Health care resource utilization, a surrogate marker for disease burden, was substantially lower for patients assigned to tezepelumab.
Infections were the most common triggers of exacerbations in both groups.
“These data provide further evidence that tezepelumab can reduce the burden of disease of severe uncontrolled asthma, both to patients and to health care systems,” Dr. Ambrose said.
Head-to-head studies needed
Although there have been no head-to-head comparisons of biologic agents for asthma to date, results of these studies suggest that tezepelumab has efficacy similar to that of other agents for reducing exacerbation, said Fernando Holguin, MD, MPH, from the University of Colorado at Denver, Aurora, who comoderated the oral session where the data were presented but was not involved in the study.
Biologic agents appear to be slightly more effective against type 2 inflammation in asthma, “but in general I think we give it to a broader severe population, so that’s exciting,” he told this news organization.
Comoderator Amisha Barochia, MBBS, MHS, of the National Institutes of Health, Bethesda, Md., told this news organization that head-to-head trials of biologic agents would provide important clinical information going forward.
“Should we switch to a different biologic or add a second biologic? Those are questions we need answers for,” she said.
The NAVIGATOR trial is funded by AstraZeneca and Amgen. Dr. Lugogo disclosed financial relationships with both companies. Dr. Holguin and Dr. Barochia have disclosed no financial relationships relevant to the studies presented.
A version of this article first appeared on Medscape.com.
SAN FRANCISCO – Nearly half of all patients with severe, uncontrolled asthma who received a full course of the biologic agent tezepelumab (Tezspire) in the NAVIGATOR trial had a complete response to treatment at 1 year, results of a prespecified exploratory analysis indicated.
Among 471 patients assigned to tezepelumab who completed the on-treatment period of the phase 3 randomized trial, 46% had a complete response at 52 weeks, compared with 24% of patients assigned to placebo.
Complete response was defined as reduction in exacerbations of at least 50% over the previous year, improvement from baseline in Asthma Control Questionnaire 6 (ACQ-6) total score of at least 0.5 points, improvement in prebronchodilator forced expiratory volume in 1 second (pre-BD FEV1), and physician-assessed Clinical Global Impression measure of clinical change (CGI-C) score.
“These data further support the efficacy of tezepelumab in a broad population of patients with severe, uncontrolled asthma,” said Njira Lugogo, MD, of the division of pulmonary and critical care medicine at the University of Michigan, Ann Arbor.
Dr. Lugogo presented results of the exploratory analysis at the American Thoracic Society’s international conference.
Exacerbations reduced, lung function improved
Primary results from NAVIGATOR, published in The New England Journal of Medicine, showed that patients with severe, uncontrolled asthma randomly assigned to tezepelumab had fewer exacerbations and better lung function, asthma control, and health-related quality of life compared with patients assigned to placebo.
The investigators noted that approximately 10% of patients with asthma have symptoms and exacerbations despite maximal standard-of-care controller therapy.
Tezepelumab is a human monoclonal antibody that inhibits action of thymic stromal lymphopoietin (TSLP), an epithelial cytokine that is released in response to airborne triggers of asthma. TSLP is a major contributor to initiation and persistence of airway inflammation, Dr. Lugogo said.
The on-treatment analysis looked at all patients in the trial who completed 52 weeks of treatment and had complete data for all criteria studied.
The odds ratios (OR) for patients on tezepelumab achieving each of the response criteria are shown in the table.
Exacerbations explored
In a separate presentation, Christopher S. Ambrose, MD, MBA, of AstraZeneca in Gaithersburg, Md., presented information from investigator-narrative descriptions of all hospitalization events related to asthma exacerbations (mild, moderate, or severe) that occurred while the investigator was blinded to each patient’s treatment assignment in NAVIGATOR.
In all, 39 of 531 patients (7.3%) assigned to placebo had a total of 78 exacerbations requiring hospitalization, compared with 13 of 528 patients (2.5%) assigned to tezepelumab. The latter group had a total of 14 exacerbations requiring hospitalization during the study.
Among hospitalized patients, 32 of the 39 assigned to placebo had severe, incapacitating exacerbations, compared with 5 of 13 assigned to tezepelumab.
Reported symptoms were generally similar between hospitalized patients in the two treatment groups, although there appeared to be trends toward lower incidence of dyspnea, fever, and tachycardia with tezepelumab.
Health care resource utilization, a surrogate marker for disease burden, was substantially lower for patients assigned to tezepelumab.
Infections were the most common triggers of exacerbations in both groups.
“These data provide further evidence that tezepelumab can reduce the burden of disease of severe uncontrolled asthma, both to patients and to health care systems,” Dr. Ambrose said.
Head-to-head studies needed
Although there have been no head-to-head comparisons of biologic agents for asthma to date, results of these studies suggest that tezepelumab has efficacy similar to that of other agents for reducing exacerbation, said Fernando Holguin, MD, MPH, from the University of Colorado at Denver, Aurora, who comoderated the oral session where the data were presented but was not involved in the study.
Biologic agents appear to be slightly more effective against type 2 inflammation in asthma, “but in general I think we give it to a broader severe population, so that’s exciting,” he told this news organization.
Comoderator Amisha Barochia, MBBS, MHS, of the National Institutes of Health, Bethesda, Md., told this news organization that head-to-head trials of biologic agents would provide important clinical information going forward.
“Should we switch to a different biologic or add a second biologic? Those are questions we need answers for,” she said.
The NAVIGATOR trial is funded by AstraZeneca and Amgen. Dr. Lugogo disclosed financial relationships with both companies. Dr. Holguin and Dr. Barochia have disclosed no financial relationships relevant to the studies presented.
A version of this article first appeared on Medscape.com.
SAN FRANCISCO – Nearly half of all patients with severe, uncontrolled asthma who received a full course of the biologic agent tezepelumab (Tezspire) in the NAVIGATOR trial had a complete response to treatment at 1 year, results of a prespecified exploratory analysis indicated.
Among 471 patients assigned to tezepelumab who completed the on-treatment period of the phase 3 randomized trial, 46% had a complete response at 52 weeks, compared with 24% of patients assigned to placebo.
Complete response was defined as reduction in exacerbations of at least 50% over the previous year, improvement from baseline in Asthma Control Questionnaire 6 (ACQ-6) total score of at least 0.5 points, improvement in prebronchodilator forced expiratory volume in 1 second (pre-BD FEV1), and physician-assessed Clinical Global Impression measure of clinical change (CGI-C) score.
“These data further support the efficacy of tezepelumab in a broad population of patients with severe, uncontrolled asthma,” said Njira Lugogo, MD, of the division of pulmonary and critical care medicine at the University of Michigan, Ann Arbor.
Dr. Lugogo presented results of the exploratory analysis at the American Thoracic Society’s international conference.
Exacerbations reduced, lung function improved
Primary results from NAVIGATOR, published in The New England Journal of Medicine, showed that patients with severe, uncontrolled asthma randomly assigned to tezepelumab had fewer exacerbations and better lung function, asthma control, and health-related quality of life compared with patients assigned to placebo.
The investigators noted that approximately 10% of patients with asthma have symptoms and exacerbations despite maximal standard-of-care controller therapy.
Tezepelumab is a human monoclonal antibody that inhibits action of thymic stromal lymphopoietin (TSLP), an epithelial cytokine that is released in response to airborne triggers of asthma. TSLP is a major contributor to initiation and persistence of airway inflammation, Dr. Lugogo said.
The on-treatment analysis looked at all patients in the trial who completed 52 weeks of treatment and had complete data for all criteria studied.
The odds ratios (OR) for patients on tezepelumab achieving each of the response criteria are shown in the table.
Exacerbations explored
In a separate presentation, Christopher S. Ambrose, MD, MBA, of AstraZeneca in Gaithersburg, Md., presented information from investigator-narrative descriptions of all hospitalization events related to asthma exacerbations (mild, moderate, or severe) that occurred while the investigator was blinded to each patient’s treatment assignment in NAVIGATOR.
In all, 39 of 531 patients (7.3%) assigned to placebo had a total of 78 exacerbations requiring hospitalization, compared with 13 of 528 patients (2.5%) assigned to tezepelumab. The latter group had a total of 14 exacerbations requiring hospitalization during the study.
Among hospitalized patients, 32 of the 39 assigned to placebo had severe, incapacitating exacerbations, compared with 5 of 13 assigned to tezepelumab.
Reported symptoms were generally similar between hospitalized patients in the two treatment groups, although there appeared to be trends toward lower incidence of dyspnea, fever, and tachycardia with tezepelumab.
Health care resource utilization, a surrogate marker for disease burden, was substantially lower for patients assigned to tezepelumab.
Infections were the most common triggers of exacerbations in both groups.
“These data provide further evidence that tezepelumab can reduce the burden of disease of severe uncontrolled asthma, both to patients and to health care systems,” Dr. Ambrose said.
Head-to-head studies needed
Although there have been no head-to-head comparisons of biologic agents for asthma to date, results of these studies suggest that tezepelumab has efficacy similar to that of other agents for reducing exacerbation, said Fernando Holguin, MD, MPH, from the University of Colorado at Denver, Aurora, who comoderated the oral session where the data were presented but was not involved in the study.
Biologic agents appear to be slightly more effective against type 2 inflammation in asthma, “but in general I think we give it to a broader severe population, so that’s exciting,” he told this news organization.
Comoderator Amisha Barochia, MBBS, MHS, of the National Institutes of Health, Bethesda, Md., told this news organization that head-to-head trials of biologic agents would provide important clinical information going forward.
“Should we switch to a different biologic or add a second biologic? Those are questions we need answers for,” she said.
The NAVIGATOR trial is funded by AstraZeneca and Amgen. Dr. Lugogo disclosed financial relationships with both companies. Dr. Holguin and Dr. Barochia have disclosed no financial relationships relevant to the studies presented.
A version of this article first appeared on Medscape.com.
AT ATS 2022
Jury is in? Survival benefit with lap surgery for rectal cancer
, according to findings from a large meta-analysis.
The estimated 5-year OS rate for patients who underwent laparoscopic surgery was 76.2%, vs. 72.7% for those who had open surgery.
“The survival benefit of laparoscopic surgery is encouraging and supports the routine use of laparoscopic surgery for adult patients with rectal cancer in the era of minimally invasive surgery,” wrote the authors, led by Leping Li, MD, of the department of gastrointestinal surgery, Shandong (China) Provincial Hospital.
The article was published online in JAMA Network Open.
Surgery is an essential component in treating rectal cancer, but the benefits of laparoscopic vs. open surgery are not clear. Over the past 15 years, randomized clinical trials (RCTs) have shown comparable long-term outcomes for laparoscopic and open surgery. However, in most meta-analyses that assessed the evidence more broadly, researchers used an “inappropriate” method for the pooled analysis. Dr. Li and colleagues wanted to perform their own meta-analysis to more definitively understand whether the evidence on long-term outcomes supports or opposes the use of laparoscopic surgery for rectal cancer.
In the current study, the authors conducted an individual participant data meta-analysis using time-to-event data and focused on the long-term survival outcomes after laparoscopic or open surgery for adult patients with rectal cancer.
Ten articles involving 12 RCTs and 3,709 participants were included. In these, 2,097 patients were randomly assigned to undergo laparoscopic surgery, and 1,612 were randomly assigned to undergo open surgery. The studies covered a global population, with participants from Europe, North America, and East Asia.
In a one-stage analysis, the authors found that disease-free survival was slightly better among patients who underwent laparoscopic surgery, but the results were statistically similar (hazard ratio [HR], 0.92; P = .26).
However, when it came to OS, those who had undergone laparoscopic surgery fared significantly better (HR, 0.85; P = .02).
These results held up in the two-stage analysis for both disease-free survival (HR, 0.92; P = .25) and OS (HR, 0.85; P = .02). A sensitivity analyses conducted with large RCTs yielded similar pooled effect sizes for disease-free survival (HR, 0.91; P = .20) and OS (HR, 0.84; P = .03).
The authors highlighted several reasons why laparoscopic surgery may be associated with better survival. First, the faster recovery from the minimally invasive procedure could allow patients to begin adjuvant therapy earlier. In addition, the reduced stress responses and higher levels of immune function among patients undergoing minimally invasive surgery may contribute to a long-term survival advantage.
“These findings address concerns regarding the effectiveness of laparoscopic surgery,” the authors wrote. However, “further studies are necessary to explore the specific mechanisms underlying the positive effect of laparoscopic surgery on OS.”
No outside funding source was noted. The authors have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
, according to findings from a large meta-analysis.
The estimated 5-year OS rate for patients who underwent laparoscopic surgery was 76.2%, vs. 72.7% for those who had open surgery.
“The survival benefit of laparoscopic surgery is encouraging and supports the routine use of laparoscopic surgery for adult patients with rectal cancer in the era of minimally invasive surgery,” wrote the authors, led by Leping Li, MD, of the department of gastrointestinal surgery, Shandong (China) Provincial Hospital.
The article was published online in JAMA Network Open.
Surgery is an essential component in treating rectal cancer, but the benefits of laparoscopic vs. open surgery are not clear. Over the past 15 years, randomized clinical trials (RCTs) have shown comparable long-term outcomes for laparoscopic and open surgery. However, in most meta-analyses that assessed the evidence more broadly, researchers used an “inappropriate” method for the pooled analysis. Dr. Li and colleagues wanted to perform their own meta-analysis to more definitively understand whether the evidence on long-term outcomes supports or opposes the use of laparoscopic surgery for rectal cancer.
In the current study, the authors conducted an individual participant data meta-analysis using time-to-event data and focused on the long-term survival outcomes after laparoscopic or open surgery for adult patients with rectal cancer.
Ten articles involving 12 RCTs and 3,709 participants were included. In these, 2,097 patients were randomly assigned to undergo laparoscopic surgery, and 1,612 were randomly assigned to undergo open surgery. The studies covered a global population, with participants from Europe, North America, and East Asia.
In a one-stage analysis, the authors found that disease-free survival was slightly better among patients who underwent laparoscopic surgery, but the results were statistically similar (hazard ratio [HR], 0.92; P = .26).
However, when it came to OS, those who had undergone laparoscopic surgery fared significantly better (HR, 0.85; P = .02).
These results held up in the two-stage analysis for both disease-free survival (HR, 0.92; P = .25) and OS (HR, 0.85; P = .02). A sensitivity analyses conducted with large RCTs yielded similar pooled effect sizes for disease-free survival (HR, 0.91; P = .20) and OS (HR, 0.84; P = .03).
The authors highlighted several reasons why laparoscopic surgery may be associated with better survival. First, the faster recovery from the minimally invasive procedure could allow patients to begin adjuvant therapy earlier. In addition, the reduced stress responses and higher levels of immune function among patients undergoing minimally invasive surgery may contribute to a long-term survival advantage.
“These findings address concerns regarding the effectiveness of laparoscopic surgery,” the authors wrote. However, “further studies are necessary to explore the specific mechanisms underlying the positive effect of laparoscopic surgery on OS.”
No outside funding source was noted. The authors have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
, according to findings from a large meta-analysis.
The estimated 5-year OS rate for patients who underwent laparoscopic surgery was 76.2%, vs. 72.7% for those who had open surgery.
“The survival benefit of laparoscopic surgery is encouraging and supports the routine use of laparoscopic surgery for adult patients with rectal cancer in the era of minimally invasive surgery,” wrote the authors, led by Leping Li, MD, of the department of gastrointestinal surgery, Shandong (China) Provincial Hospital.
The article was published online in JAMA Network Open.
Surgery is an essential component in treating rectal cancer, but the benefits of laparoscopic vs. open surgery are not clear. Over the past 15 years, randomized clinical trials (RCTs) have shown comparable long-term outcomes for laparoscopic and open surgery. However, in most meta-analyses that assessed the evidence more broadly, researchers used an “inappropriate” method for the pooled analysis. Dr. Li and colleagues wanted to perform their own meta-analysis to more definitively understand whether the evidence on long-term outcomes supports or opposes the use of laparoscopic surgery for rectal cancer.
In the current study, the authors conducted an individual participant data meta-analysis using time-to-event data and focused on the long-term survival outcomes after laparoscopic or open surgery for adult patients with rectal cancer.
Ten articles involving 12 RCTs and 3,709 participants were included. In these, 2,097 patients were randomly assigned to undergo laparoscopic surgery, and 1,612 were randomly assigned to undergo open surgery. The studies covered a global population, with participants from Europe, North America, and East Asia.
In a one-stage analysis, the authors found that disease-free survival was slightly better among patients who underwent laparoscopic surgery, but the results were statistically similar (hazard ratio [HR], 0.92; P = .26).
However, when it came to OS, those who had undergone laparoscopic surgery fared significantly better (HR, 0.85; P = .02).
These results held up in the two-stage analysis for both disease-free survival (HR, 0.92; P = .25) and OS (HR, 0.85; P = .02). A sensitivity analyses conducted with large RCTs yielded similar pooled effect sizes for disease-free survival (HR, 0.91; P = .20) and OS (HR, 0.84; P = .03).
The authors highlighted several reasons why laparoscopic surgery may be associated with better survival. First, the faster recovery from the minimally invasive procedure could allow patients to begin adjuvant therapy earlier. In addition, the reduced stress responses and higher levels of immune function among patients undergoing minimally invasive surgery may contribute to a long-term survival advantage.
“These findings address concerns regarding the effectiveness of laparoscopic surgery,” the authors wrote. However, “further studies are necessary to explore the specific mechanisms underlying the positive effect of laparoscopic surgery on OS.”
No outside funding source was noted. The authors have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FROM JAMA NETWORK OPEN
Male breast cancer risk linked with infertility
, according to new research funded by the charity Breast Cancer Now and published in Breast Cancer Research. The study is one of the largest ever into male breast cancer, enabling the team to show a highly statistically significant association.
A link with infertility had been suspected, since parity markedly reduces the risk of female breast cancer; there are known genetic links in both sexes, and a high risk of both breast cancer and infertility among men with Klinefelter syndrome, suggesting some sex hormone-related involvement. However, the rarity of breast cancer in men – with an annual incidence of about 370 cases and 80 deaths per year in the United Kingdom – meant that past studies were necessarily small and yielded mixed results.
“Compared with previous studies, our study of male breast cancer is large,” said study coauthor Michael Jones, PhD, of the division of genetics and epidemiology at the Institute of Cancer Research (ICR) in London. “It was carried out nationwide across England and Wales and was set in motion more than 15 years ago. Because of how rare male breast cancer is, it took us over 12 years to identify and interview the nearly 2,000 men with breast cancer who were part of this study.”
The latest research is part of the wider Breast Cancer Now Male Breast Cancer Study, launched by the charity in 2007. For the new study, the ICR team interviewed 1,998 males living in England and Wales who had been diagnosed with breast cancer between 2005 and 2017. All were aged under 80 but most 60 or older at diagnosis; 92% of their tumors were invasive, and almost all were estrogen receptor positive (98.5% of those with known status).
Their responses were compared with those of a control group of 1,597 men without breast cancer, matched by age at diagnosis and geographic region, recruited from male non-blood relatives of cases and from husbands of women participating in the Generations cohort study of breast cancer etiology.
Raised risk with history of male infertility
Overall, 112 cases (5.6%) and 80 controls (5.0%) reported that they had had infertility problems for which they or their partner had consulted a doctor or infertility clinic. This represented a raised odds ratio of 1.29 (95% confidence interval, 0.94-1.77), which was statistically not significant. However, when analyzed by outcome of the infertility consultation, there was a significant and more than doubled risk of breast cancer among men who were diagnosed as the source of the couple’s infertility (OR = 2.03 [1.18-3.49]), whereas this was not the case among men whose partner was the source (OR = 0.86 [0.51-1.45]) or for whom no source was identified (OR = 1.26 [0.71-2.24]).
In addition, proportionately fewer cases (1,615, or 80.8%) compared with controls (1,423, or 89.1%) had fathered any children, also giving a statistically significantly raised risk of breast cancer for men with no biological children (OR = 1.50 [1.21-1.86], P < .001), “congruent with infertility as a risk factor,” the authors said. The risk was statistically significant for invasive tumors but not for the much smaller number of in situ tumors.
Analysis by number of children showed a decreasing risk with increasing numbers of children, with a highly significant (P < .001) inverse trend where zero was included as a value, but a borderline significant trend (P = .04) if it was not. The team noted that number of children beyond one is difficult to interpret as an indicator of male fertility, since it may more reflect social and cultural factors than fertility per se.
Baseline demographic factors were adjusted for in the risk analyses, and results were not materially changed by sensitivity analyses adjusting additionally for alcohol consumption, smoking, liver disease, and family history of breast cancer. The association also largely remained after exclusion of patients with other preexisting potential confounders including severe obesity and testicular abnormalities, and was consistent irrespective of HER-2 status (there were too few ER-negative tumors to analyze results by ER status).
Potential underlying factors
“The causes of breast cancer in men are largely unknown, partly because it is rare and partly because previous studies have been small,” Dr. Jones said. “The evidence presented in our study suggests that the association of infertility and breast cancer should be confirmed with further research, and future investigations are needed into the potential underlying factors, such as hormone imbalances.”
Commenting on the study, Fiona Osgun, senior health information manager at Cancer Research UK, told this news organization: “Overall, there isn’t strong evidence that infertility is a risk factor for male breast cancer. This study helps to shed light onto a cancer type that is sadly still not very well understood, but much more research is needed to say that infertility is a risk factor for male breast cancer.”
She added that although male breast cancer is a rare condition, it’s still important for men to be aware of what looks and feels normal for them, and to be encouraged to seek medical advice if something is not quite right.
A spokesperson for Breast Cancer UK told this news organization: “[We] believe it’s important to understand what leads to breast cancer in men as well as women and that high quality, long-term studies such as this will help with this understanding.
The findings are consistent with an earlier study that found that U.S. men who have never fathered children are at higher risk of breast cancer. This new long-term U.K. study provides strong evidence, which supports this finding.
“As the authors note, the biological reasons are unclear, but may be associated with altered hormone levels. The ratio of circulating levels of estrogen and androgens (e.g. testosterone) is crucial in healthy functioning of breast tissue. Disruption to this, for example as a result of damage to testes, may affect both fertility and breast cancer risk.
“It is also possible that external factors, such as exposure to certain endocrine (hormone) disrupting chemicals (EDCs), which affect sex hormones, may also affect both fertility and breast cancer risk.
“More studies into breast cancer in men are needed to help us understand better all the risk factors associated with this disease including both hormonal factors and chemical exposures.”
Simon Vincent, PhD, director of research, support, and influencing at Breast Cancer Now, said: “Research has discovered different treatments directed at some features of breast cancer in women; however, breast cancer is not as well understood for men. This is why Breast Cancer Now funds the Male Breast Cancer Study, which looks at what might cause the disease in men. Discovering a link between infertility and male breast cancer is a step towards us understanding male breast cancer and how we could find more ways to diagnose and treat men – and possibly women – with this devastating disease.”
A version of this article first appeared on Medscape UK.
, according to new research funded by the charity Breast Cancer Now and published in Breast Cancer Research. The study is one of the largest ever into male breast cancer, enabling the team to show a highly statistically significant association.
A link with infertility had been suspected, since parity markedly reduces the risk of female breast cancer; there are known genetic links in both sexes, and a high risk of both breast cancer and infertility among men with Klinefelter syndrome, suggesting some sex hormone-related involvement. However, the rarity of breast cancer in men – with an annual incidence of about 370 cases and 80 deaths per year in the United Kingdom – meant that past studies were necessarily small and yielded mixed results.
“Compared with previous studies, our study of male breast cancer is large,” said study coauthor Michael Jones, PhD, of the division of genetics and epidemiology at the Institute of Cancer Research (ICR) in London. “It was carried out nationwide across England and Wales and was set in motion more than 15 years ago. Because of how rare male breast cancer is, it took us over 12 years to identify and interview the nearly 2,000 men with breast cancer who were part of this study.”
The latest research is part of the wider Breast Cancer Now Male Breast Cancer Study, launched by the charity in 2007. For the new study, the ICR team interviewed 1,998 males living in England and Wales who had been diagnosed with breast cancer between 2005 and 2017. All were aged under 80 but most 60 or older at diagnosis; 92% of their tumors were invasive, and almost all were estrogen receptor positive (98.5% of those with known status).
Their responses were compared with those of a control group of 1,597 men without breast cancer, matched by age at diagnosis and geographic region, recruited from male non-blood relatives of cases and from husbands of women participating in the Generations cohort study of breast cancer etiology.
Raised risk with history of male infertility
Overall, 112 cases (5.6%) and 80 controls (5.0%) reported that they had had infertility problems for which they or their partner had consulted a doctor or infertility clinic. This represented a raised odds ratio of 1.29 (95% confidence interval, 0.94-1.77), which was statistically not significant. However, when analyzed by outcome of the infertility consultation, there was a significant and more than doubled risk of breast cancer among men who were diagnosed as the source of the couple’s infertility (OR = 2.03 [1.18-3.49]), whereas this was not the case among men whose partner was the source (OR = 0.86 [0.51-1.45]) or for whom no source was identified (OR = 1.26 [0.71-2.24]).
In addition, proportionately fewer cases (1,615, or 80.8%) compared with controls (1,423, or 89.1%) had fathered any children, also giving a statistically significantly raised risk of breast cancer for men with no biological children (OR = 1.50 [1.21-1.86], P < .001), “congruent with infertility as a risk factor,” the authors said. The risk was statistically significant for invasive tumors but not for the much smaller number of in situ tumors.
Analysis by number of children showed a decreasing risk with increasing numbers of children, with a highly significant (P < .001) inverse trend where zero was included as a value, but a borderline significant trend (P = .04) if it was not. The team noted that number of children beyond one is difficult to interpret as an indicator of male fertility, since it may more reflect social and cultural factors than fertility per se.
Baseline demographic factors were adjusted for in the risk analyses, and results were not materially changed by sensitivity analyses adjusting additionally for alcohol consumption, smoking, liver disease, and family history of breast cancer. The association also largely remained after exclusion of patients with other preexisting potential confounders including severe obesity and testicular abnormalities, and was consistent irrespective of HER-2 status (there were too few ER-negative tumors to analyze results by ER status).
Potential underlying factors
“The causes of breast cancer in men are largely unknown, partly because it is rare and partly because previous studies have been small,” Dr. Jones said. “The evidence presented in our study suggests that the association of infertility and breast cancer should be confirmed with further research, and future investigations are needed into the potential underlying factors, such as hormone imbalances.”
Commenting on the study, Fiona Osgun, senior health information manager at Cancer Research UK, told this news organization: “Overall, there isn’t strong evidence that infertility is a risk factor for male breast cancer. This study helps to shed light onto a cancer type that is sadly still not very well understood, but much more research is needed to say that infertility is a risk factor for male breast cancer.”
She added that although male breast cancer is a rare condition, it’s still important for men to be aware of what looks and feels normal for them, and to be encouraged to seek medical advice if something is not quite right.
A spokesperson for Breast Cancer UK told this news organization: “[We] believe it’s important to understand what leads to breast cancer in men as well as women and that high quality, long-term studies such as this will help with this understanding.
The findings are consistent with an earlier study that found that U.S. men who have never fathered children are at higher risk of breast cancer. This new long-term U.K. study provides strong evidence, which supports this finding.
“As the authors note, the biological reasons are unclear, but may be associated with altered hormone levels. The ratio of circulating levels of estrogen and androgens (e.g. testosterone) is crucial in healthy functioning of breast tissue. Disruption to this, for example as a result of damage to testes, may affect both fertility and breast cancer risk.
“It is also possible that external factors, such as exposure to certain endocrine (hormone) disrupting chemicals (EDCs), which affect sex hormones, may also affect both fertility and breast cancer risk.
“More studies into breast cancer in men are needed to help us understand better all the risk factors associated with this disease including both hormonal factors and chemical exposures.”
Simon Vincent, PhD, director of research, support, and influencing at Breast Cancer Now, said: “Research has discovered different treatments directed at some features of breast cancer in women; however, breast cancer is not as well understood for men. This is why Breast Cancer Now funds the Male Breast Cancer Study, which looks at what might cause the disease in men. Discovering a link between infertility and male breast cancer is a step towards us understanding male breast cancer and how we could find more ways to diagnose and treat men – and possibly women – with this devastating disease.”
A version of this article first appeared on Medscape UK.
, according to new research funded by the charity Breast Cancer Now and published in Breast Cancer Research. The study is one of the largest ever into male breast cancer, enabling the team to show a highly statistically significant association.
A link with infertility had been suspected, since parity markedly reduces the risk of female breast cancer; there are known genetic links in both sexes, and a high risk of both breast cancer and infertility among men with Klinefelter syndrome, suggesting some sex hormone-related involvement. However, the rarity of breast cancer in men – with an annual incidence of about 370 cases and 80 deaths per year in the United Kingdom – meant that past studies were necessarily small and yielded mixed results.
“Compared with previous studies, our study of male breast cancer is large,” said study coauthor Michael Jones, PhD, of the division of genetics and epidemiology at the Institute of Cancer Research (ICR) in London. “It was carried out nationwide across England and Wales and was set in motion more than 15 years ago. Because of how rare male breast cancer is, it took us over 12 years to identify and interview the nearly 2,000 men with breast cancer who were part of this study.”
The latest research is part of the wider Breast Cancer Now Male Breast Cancer Study, launched by the charity in 2007. For the new study, the ICR team interviewed 1,998 males living in England and Wales who had been diagnosed with breast cancer between 2005 and 2017. All were aged under 80 but most 60 or older at diagnosis; 92% of their tumors were invasive, and almost all were estrogen receptor positive (98.5% of those with known status).
Their responses were compared with those of a control group of 1,597 men without breast cancer, matched by age at diagnosis and geographic region, recruited from male non-blood relatives of cases and from husbands of women participating in the Generations cohort study of breast cancer etiology.
Raised risk with history of male infertility
Overall, 112 cases (5.6%) and 80 controls (5.0%) reported that they had had infertility problems for which they or their partner had consulted a doctor or infertility clinic. This represented a raised odds ratio of 1.29 (95% confidence interval, 0.94-1.77), which was statistically not significant. However, when analyzed by outcome of the infertility consultation, there was a significant and more than doubled risk of breast cancer among men who were diagnosed as the source of the couple’s infertility (OR = 2.03 [1.18-3.49]), whereas this was not the case among men whose partner was the source (OR = 0.86 [0.51-1.45]) or for whom no source was identified (OR = 1.26 [0.71-2.24]).
In addition, proportionately fewer cases (1,615, or 80.8%) compared with controls (1,423, or 89.1%) had fathered any children, also giving a statistically significantly raised risk of breast cancer for men with no biological children (OR = 1.50 [1.21-1.86], P < .001), “congruent with infertility as a risk factor,” the authors said. The risk was statistically significant for invasive tumors but not for the much smaller number of in situ tumors.
Analysis by number of children showed a decreasing risk with increasing numbers of children, with a highly significant (P < .001) inverse trend where zero was included as a value, but a borderline significant trend (P = .04) if it was not. The team noted that number of children beyond one is difficult to interpret as an indicator of male fertility, since it may more reflect social and cultural factors than fertility per se.
Baseline demographic factors were adjusted for in the risk analyses, and results were not materially changed by sensitivity analyses adjusting additionally for alcohol consumption, smoking, liver disease, and family history of breast cancer. The association also largely remained after exclusion of patients with other preexisting potential confounders including severe obesity and testicular abnormalities, and was consistent irrespective of HER-2 status (there were too few ER-negative tumors to analyze results by ER status).
Potential underlying factors
“The causes of breast cancer in men are largely unknown, partly because it is rare and partly because previous studies have been small,” Dr. Jones said. “The evidence presented in our study suggests that the association of infertility and breast cancer should be confirmed with further research, and future investigations are needed into the potential underlying factors, such as hormone imbalances.”
Commenting on the study, Fiona Osgun, senior health information manager at Cancer Research UK, told this news organization: “Overall, there isn’t strong evidence that infertility is a risk factor for male breast cancer. This study helps to shed light onto a cancer type that is sadly still not very well understood, but much more research is needed to say that infertility is a risk factor for male breast cancer.”
She added that although male breast cancer is a rare condition, it’s still important for men to be aware of what looks and feels normal for them, and to be encouraged to seek medical advice if something is not quite right.
A spokesperson for Breast Cancer UK told this news organization: “[We] believe it’s important to understand what leads to breast cancer in men as well as women and that high quality, long-term studies such as this will help with this understanding.
The findings are consistent with an earlier study that found that U.S. men who have never fathered children are at higher risk of breast cancer. This new long-term U.K. study provides strong evidence, which supports this finding.
“As the authors note, the biological reasons are unclear, but may be associated with altered hormone levels. The ratio of circulating levels of estrogen and androgens (e.g. testosterone) is crucial in healthy functioning of breast tissue. Disruption to this, for example as a result of damage to testes, may affect both fertility and breast cancer risk.
“It is also possible that external factors, such as exposure to certain endocrine (hormone) disrupting chemicals (EDCs), which affect sex hormones, may also affect both fertility and breast cancer risk.
“More studies into breast cancer in men are needed to help us understand better all the risk factors associated with this disease including both hormonal factors and chemical exposures.”
Simon Vincent, PhD, director of research, support, and influencing at Breast Cancer Now, said: “Research has discovered different treatments directed at some features of breast cancer in women; however, breast cancer is not as well understood for men. This is why Breast Cancer Now funds the Male Breast Cancer Study, which looks at what might cause the disease in men. Discovering a link between infertility and male breast cancer is a step towards us understanding male breast cancer and how we could find more ways to diagnose and treat men – and possibly women – with this devastating disease.”
A version of this article first appeared on Medscape UK.
FROM BREAST CANCER RESEARCH
The psychopathic brain: New insight
Using MRI, researchers found that the striatum was about 10% larger on average in adults with psychopathic traits than in matched control persons and that this relationship was mediated by stimulation seeking and impulsivity.
The striatum is a subcortical region of the forebrain involved in the cognitive processing of reward-related information and motivational aspects of behavior.
“Our study’s results help advance our knowledge about what underlies antisocial behavior such as psychopathy,” co-author and neurocriminologist Olivia Choy, PhD, with Nanyang Technological University, Singapore, said in a news release.
“In addition to social environmental influences, it is important to consider that there can be differences in biology – in this case, the size of brain structures – between antisocial and non-antisocial individuals,” Dr. Choy added.
The study was published online in the Journal of Psychiatric Research.
Antisocial, egocentric
Individuals with psychopathic traits typically have an egocentric and antisocial personality. They generally lack remorse for their actions or empathy for others and often have criminal tendencies.
Some prior research suggests links between psychopathy and an overactive striatum, but it was unclear what role striatal volume plays in this behavior.
For the study, investigators assessed striatal volume using MRI in 120 adults living in the community, and they assessed psychopathy using the Psychopathy Checklist – Revised.
Correlational analyses showed that increased striatal volumes were associated with more psychopathic traits (P = .001) in both men and women.
Volumetric increases were found for all subregions of the striatum in psychopathic individuals, after controlling for age, substance dependence, substance abuse, antisocial personality disorder, attention-deficit/hyperactivity disorder, social adversity, and total brain volume.
An analysis of 18 psychopathic individuals showed that striatal volumes were increased 9.4%, compared with 18 propensity-matched control persons (P = .01).
Abnormal reward processing
Stimulation seeking and impulsivity partly mediated the striatal-psychopathy relationship, accounting for 49.4% of this association.
These findings “replicate and build on initial studies indicating striatal enlargement in adults with psychopathy, yielding an updated effect size of d = 0.48,” the researchers note.
The results are “consistent with the notion that striatal abnormalities in individuals with psychopathy partly reflect increased sensation-seeking and impulsivity and support the hypothesis of abnormal reward processing in psychopathy,” they add.
“We have always known that psychopaths go to extreme lengths to seek out rewards, including criminal activities that involve property, sex, and drugs,” co-author Adrian Raine, DPhil, department of criminology, psychiatry, and psychology, University of Pennsylvania, Philadelphia, said in a news release.
“We are now finding out a neurobiological underpinning of this impulsive and stimulating behavior in the form of enlargement to the striatum, a key brain area involved in rewards,” Dr. Raine added.
What causes striatal enlargement in individuals with psychopathy still needs to be determined.
In human development, the striatum typically becomes smaller as a child matures, suggesting that psychopathy is associated with differences in brain development, the researchers suggest.
“Because biological traits, such as the size of one’s striatum, can be inherited to child from parent, these findings give added support to neurodevelopmental perspectives of psychopathy – that the brains of these offenders do not develop normally throughout childhood and adolescence,” said Dr. Raine.
Larger studies needed
Commenting on the findings for this news organization, Terrie E. Moffitt, PhD, professor of psychology, Duke University, Durham, N.C., noted that there is “general consensus among brain-imaging researchers that testing brain-behavior relations requires very large samples in the thousands and also samples of research participants who represent the full extent of variation in the population as well as possible – from rich to poor, from well to unwell, from high IQ to low IQ, from strong mental health to mental illness, etc.
“It would be grand to see this study’s provocative finding replicated in a large, representative sampling design,” Dr. Moffitt said.
The study was supported in part by the National Institutes of Health. Dr. Choy, Dr. Raine, and Dr. Moffitt have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Using MRI, researchers found that the striatum was about 10% larger on average in adults with psychopathic traits than in matched control persons and that this relationship was mediated by stimulation seeking and impulsivity.
The striatum is a subcortical region of the forebrain involved in the cognitive processing of reward-related information and motivational aspects of behavior.
“Our study’s results help advance our knowledge about what underlies antisocial behavior such as psychopathy,” co-author and neurocriminologist Olivia Choy, PhD, with Nanyang Technological University, Singapore, said in a news release.
“In addition to social environmental influences, it is important to consider that there can be differences in biology – in this case, the size of brain structures – between antisocial and non-antisocial individuals,” Dr. Choy added.
The study was published online in the Journal of Psychiatric Research.
Antisocial, egocentric
Individuals with psychopathic traits typically have an egocentric and antisocial personality. They generally lack remorse for their actions or empathy for others and often have criminal tendencies.
Some prior research suggests links between psychopathy and an overactive striatum, but it was unclear what role striatal volume plays in this behavior.
For the study, investigators assessed striatal volume using MRI in 120 adults living in the community, and they assessed psychopathy using the Psychopathy Checklist – Revised.
Correlational analyses showed that increased striatal volumes were associated with more psychopathic traits (P = .001) in both men and women.
Volumetric increases were found for all subregions of the striatum in psychopathic individuals, after controlling for age, substance dependence, substance abuse, antisocial personality disorder, attention-deficit/hyperactivity disorder, social adversity, and total brain volume.
An analysis of 18 psychopathic individuals showed that striatal volumes were increased 9.4%, compared with 18 propensity-matched control persons (P = .01).
Abnormal reward processing
Stimulation seeking and impulsivity partly mediated the striatal-psychopathy relationship, accounting for 49.4% of this association.
These findings “replicate and build on initial studies indicating striatal enlargement in adults with psychopathy, yielding an updated effect size of d = 0.48,” the researchers note.
The results are “consistent with the notion that striatal abnormalities in individuals with psychopathy partly reflect increased sensation-seeking and impulsivity and support the hypothesis of abnormal reward processing in psychopathy,” they add.
“We have always known that psychopaths go to extreme lengths to seek out rewards, including criminal activities that involve property, sex, and drugs,” co-author Adrian Raine, DPhil, department of criminology, psychiatry, and psychology, University of Pennsylvania, Philadelphia, said in a news release.
“We are now finding out a neurobiological underpinning of this impulsive and stimulating behavior in the form of enlargement to the striatum, a key brain area involved in rewards,” Dr. Raine added.
What causes striatal enlargement in individuals with psychopathy still needs to be determined.
In human development, the striatum typically becomes smaller as a child matures, suggesting that psychopathy is associated with differences in brain development, the researchers suggest.
“Because biological traits, such as the size of one’s striatum, can be inherited to child from parent, these findings give added support to neurodevelopmental perspectives of psychopathy – that the brains of these offenders do not develop normally throughout childhood and adolescence,” said Dr. Raine.
Larger studies needed
Commenting on the findings for this news organization, Terrie E. Moffitt, PhD, professor of psychology, Duke University, Durham, N.C., noted that there is “general consensus among brain-imaging researchers that testing brain-behavior relations requires very large samples in the thousands and also samples of research participants who represent the full extent of variation in the population as well as possible – from rich to poor, from well to unwell, from high IQ to low IQ, from strong mental health to mental illness, etc.
“It would be grand to see this study’s provocative finding replicated in a large, representative sampling design,” Dr. Moffitt said.
The study was supported in part by the National Institutes of Health. Dr. Choy, Dr. Raine, and Dr. Moffitt have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Using MRI, researchers found that the striatum was about 10% larger on average in adults with psychopathic traits than in matched control persons and that this relationship was mediated by stimulation seeking and impulsivity.
The striatum is a subcortical region of the forebrain involved in the cognitive processing of reward-related information and motivational aspects of behavior.
“Our study’s results help advance our knowledge about what underlies antisocial behavior such as psychopathy,” co-author and neurocriminologist Olivia Choy, PhD, with Nanyang Technological University, Singapore, said in a news release.
“In addition to social environmental influences, it is important to consider that there can be differences in biology – in this case, the size of brain structures – between antisocial and non-antisocial individuals,” Dr. Choy added.
The study was published online in the Journal of Psychiatric Research.
Antisocial, egocentric
Individuals with psychopathic traits typically have an egocentric and antisocial personality. They generally lack remorse for their actions or empathy for others and often have criminal tendencies.
Some prior research suggests links between psychopathy and an overactive striatum, but it was unclear what role striatal volume plays in this behavior.
For the study, investigators assessed striatal volume using MRI in 120 adults living in the community, and they assessed psychopathy using the Psychopathy Checklist – Revised.
Correlational analyses showed that increased striatal volumes were associated with more psychopathic traits (P = .001) in both men and women.
Volumetric increases were found for all subregions of the striatum in psychopathic individuals, after controlling for age, substance dependence, substance abuse, antisocial personality disorder, attention-deficit/hyperactivity disorder, social adversity, and total brain volume.
An analysis of 18 psychopathic individuals showed that striatal volumes were increased 9.4%, compared with 18 propensity-matched control persons (P = .01).
Abnormal reward processing
Stimulation seeking and impulsivity partly mediated the striatal-psychopathy relationship, accounting for 49.4% of this association.
These findings “replicate and build on initial studies indicating striatal enlargement in adults with psychopathy, yielding an updated effect size of d = 0.48,” the researchers note.
The results are “consistent with the notion that striatal abnormalities in individuals with psychopathy partly reflect increased sensation-seeking and impulsivity and support the hypothesis of abnormal reward processing in psychopathy,” they add.
“We have always known that psychopaths go to extreme lengths to seek out rewards, including criminal activities that involve property, sex, and drugs,” co-author Adrian Raine, DPhil, department of criminology, psychiatry, and psychology, University of Pennsylvania, Philadelphia, said in a news release.
“We are now finding out a neurobiological underpinning of this impulsive and stimulating behavior in the form of enlargement to the striatum, a key brain area involved in rewards,” Dr. Raine added.
What causes striatal enlargement in individuals with psychopathy still needs to be determined.
In human development, the striatum typically becomes smaller as a child matures, suggesting that psychopathy is associated with differences in brain development, the researchers suggest.
“Because biological traits, such as the size of one’s striatum, can be inherited to child from parent, these findings give added support to neurodevelopmental perspectives of psychopathy – that the brains of these offenders do not develop normally throughout childhood and adolescence,” said Dr. Raine.
Larger studies needed
Commenting on the findings for this news organization, Terrie E. Moffitt, PhD, professor of psychology, Duke University, Durham, N.C., noted that there is “general consensus among brain-imaging researchers that testing brain-behavior relations requires very large samples in the thousands and also samples of research participants who represent the full extent of variation in the population as well as possible – from rich to poor, from well to unwell, from high IQ to low IQ, from strong mental health to mental illness, etc.
“It would be grand to see this study’s provocative finding replicated in a large, representative sampling design,” Dr. Moffitt said.
The study was supported in part by the National Institutes of Health. Dr. Choy, Dr. Raine, and Dr. Moffitt have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Cluttered consciousness: The mental effects of growing up with a hoarder
Many of us are reluctant to throw things out.
We buy. We accumulate. We collect. Eventually our attics are packed with dusty heirlooms that we rarely, if ever, look at. Eventually we’re forced to pare down and head to the Goodwill.
But not all of us.
Hoarding – or the prolonged difficulty of discarding unneeded possessions – is pervasive in our culture, affecting nearly 3% of the population. This compulsive collecting, and unwillingness to part with “stuff,” is even the subject of multiple popular television series.
How do you conceptualize hoarding behavior?
The core feature of hoarding is the inability to throw things away. This can be due to many different reasons, whether there’s a strong sentimental attachment or the belief that you will need these items one day. Compulsive buying is often involved, and inevitable clutter.
How was hoarding first conceptualized among psychiatrists and psychologists? And when did the term first enter the lexicon?
It was originally conceptualized as a difficult-to-treat subtype of obsessive-compulsive disorder (OCD). A lot of that work identifying this subgroup was going on in the late 1980s and early 1990s. There was a small but growing group of researchers demonstrating that this is fundamentally different from OCD in several ways.
In terms of the clinical presentation, the comorbidity patterns are different from those for OCD. And the course is a little bit different; we see a progressive development across the lifespan, as opposed to a clear-cut diagnosis earlier in life, as is typically seen with OCD. By the time a lot of people seek treatment, they’re often being brought in by, say, family members when they’re a little bit older. With hoarding, there is also this consistent pattern of poor treatment response across the board, whether to selective serotonin reuptake inhibitors or behavioral therapy.
A lot of this work together led to advocacy for recognizing hoarding as an independent diagnosis in the DSM-5. I think official recognition by our “big book” prompted more attention to this population. Previously these patients probably would have been diagnosed with OCD, and it really isn’t appropriate to think of hoarding as purely an anxiety disorder.
Hoarding exposure and future mental health
You have a new study, published in Annals of Clinical Psychiatry, looking at mental health among adult children of parents with hoarding problems. Can you tell us what inspired you to run this study, and what you found?
There were a couple of factors.
We’d seen a lot of folks with hoarding in OCD specialty clinics, so my clinical experiences with this population certainly drew me to this general area. But then, at the same time, I have this broad training in child mental health. And childhood trauma or adverse childhood experiences, which can include being around hoarding, can be a very difficult thing to live through and deal with. And here I have to give a lot of credit to Suzanne Chabaud, PhD, of the OCD Institute of Greater New Orleans, who’s one of the coauthors on the paper. She’s been beating the drum of thinking about the family and kids of people with hoarding disorders for years. My interests came from some of those experiences, but she had the good idea of really looking at this problem in a detailed way.
Prior to your paper, had there been research on the prevalence of mental illnesses such as anxiety and depression in the children of people with hoarding behaviors?
That particular question was new to our paper. It was the first time anyone, to my knowledge, had looked at a validated assessment of anxiety and depression in this population.
How did you assess their symptoms and what did you find?
We asked study participants to think back on how they felt throughout their teenage years and gauged their responses with the Patient Health Questionnaire (PHQ), a measure of mental health disorders. I should say up front that we didn’t have a control group. But we found that among our 414 study participants, somewhere between 30% and 50% reported clinically significant anxiety or depressive symptoms, far higher than you’d expect in the normal population. So when looking back on how they were feeling as teenagers in that environment, they were struggling, and they often felt rejected by their parents.
We also found that almost 10% of participants were threatened with eviction at some point in their childhood; 15% had to live outside of their home at some point, because of the clutter; and 2% had involvement from child protective services and were removed from the home.
I know you recruited patients from online forums established by the children of hoarding parents. Presumably, these are the people most affected by this phenomenon. How does this play out in people who simply like to, say, collect something? Is this a continuum of behavior, with a breaking point at which it becomes a pathology?
I think it’s safe to conceptualize collecting and hoarding as a continuum, and you’ve got to draw a line somewhere in terms of clinical significance.
Did you assess whether the children of hoarders were more likely to hoard themselves as adults?
This is our follow-up paper; we haven’t looked at it yet.
But in looking at preliminary data, the prevalence seems pretty low, actually, at least in our sample. And as you mentioned, in our study there were folks who were seeking support specifically because they grew up in a really cluttered home.
Management
How do mental health providers typically address and treat hoarding?
To my knowledge, there are no current Food and Drug Administration–approved medications for hoarding, though psychiatrists will prescribe SSRIs and try to treat co-occurring problems such as depression and anxiety symptoms.
I can speak to cognitive-behavioral therapy (CBT) in a bit more detail. A number of randomized controlled trials support CBT for hoarding. I mentioned before that when we as a field treated hoarding akin to OCD and did exposure and response prevention therapy, we didn’t really target the specific features of hoarding. People didn’t do that well.
But now researchers are focusing on CBT interventions focused on discarding tasks that really address hoarding. You can create different categories for different items: Patients can either keep them, throw them out, or donate them. You can explore what thoughts or expectations are associated with these items and try to address them. Clinicians can help patients look at, say, different areas of their house and discuss what they might be willing to part with or at least think about parting with. You find their internal motivations for keeping things.
This sort of therapy generally takes longer than it does for, say, OCD. It can be a little bit slower, particularly if someone has a lot of stuff. And often it can involve doing home visits. In the age of Zoom this is a little bit easier because home visits aren’t always feasible.
What role does family play in managing hoarding? I imagine that including loved ones and friends in the process could be quite helpful.
Yes, absolutely. And social support, more broadly.
A colleague I worked with did a really interesting study where she looked at psychologist-delivered versus peer-delivered CBT for hoarding. They found that the biggest predictor of improved outcomes was having what they called a “clutter buddy,” which follows the Alcoholics Anonymous sponsor model. This would be somebody else struggling with the same problem who’s an accountability partner helping a patient follow through with their goals related to discarding. I think that finding underscores how important that social support is.
Any final thoughts for our audience of clinicians and researchers on how to approach hoarding?
I think there’s been a stigma – at least in psychology circles – that it’s not really treatable because of that earlier work with OCD. But on the CBT side, there’s now good reason to believe that people can live much happier lives and overcome this problem. CBT does seem to work for a lot of people with hoarding. That’s what I’d like to emphasize.
Dr. Stetka is executive editor for Medscape. A version of this article first appeared on Medscape.com.
Many of us are reluctant to throw things out.
We buy. We accumulate. We collect. Eventually our attics are packed with dusty heirlooms that we rarely, if ever, look at. Eventually we’re forced to pare down and head to the Goodwill.
But not all of us.
Hoarding – or the prolonged difficulty of discarding unneeded possessions – is pervasive in our culture, affecting nearly 3% of the population. This compulsive collecting, and unwillingness to part with “stuff,” is even the subject of multiple popular television series.
How do you conceptualize hoarding behavior?
The core feature of hoarding is the inability to throw things away. This can be due to many different reasons, whether there’s a strong sentimental attachment or the belief that you will need these items one day. Compulsive buying is often involved, and inevitable clutter.
How was hoarding first conceptualized among psychiatrists and psychologists? And when did the term first enter the lexicon?
It was originally conceptualized as a difficult-to-treat subtype of obsessive-compulsive disorder (OCD). A lot of that work identifying this subgroup was going on in the late 1980s and early 1990s. There was a small but growing group of researchers demonstrating that this is fundamentally different from OCD in several ways.
In terms of the clinical presentation, the comorbidity patterns are different from those for OCD. And the course is a little bit different; we see a progressive development across the lifespan, as opposed to a clear-cut diagnosis earlier in life, as is typically seen with OCD. By the time a lot of people seek treatment, they’re often being brought in by, say, family members when they’re a little bit older. With hoarding, there is also this consistent pattern of poor treatment response across the board, whether to selective serotonin reuptake inhibitors or behavioral therapy.
A lot of this work together led to advocacy for recognizing hoarding as an independent diagnosis in the DSM-5. I think official recognition by our “big book” prompted more attention to this population. Previously these patients probably would have been diagnosed with OCD, and it really isn’t appropriate to think of hoarding as purely an anxiety disorder.
Hoarding exposure and future mental health
You have a new study, published in Annals of Clinical Psychiatry, looking at mental health among adult children of parents with hoarding problems. Can you tell us what inspired you to run this study, and what you found?
There were a couple of factors.
We’d seen a lot of folks with hoarding in OCD specialty clinics, so my clinical experiences with this population certainly drew me to this general area. But then, at the same time, I have this broad training in child mental health. And childhood trauma or adverse childhood experiences, which can include being around hoarding, can be a very difficult thing to live through and deal with. And here I have to give a lot of credit to Suzanne Chabaud, PhD, of the OCD Institute of Greater New Orleans, who’s one of the coauthors on the paper. She’s been beating the drum of thinking about the family and kids of people with hoarding disorders for years. My interests came from some of those experiences, but she had the good idea of really looking at this problem in a detailed way.
Prior to your paper, had there been research on the prevalence of mental illnesses such as anxiety and depression in the children of people with hoarding behaviors?
That particular question was new to our paper. It was the first time anyone, to my knowledge, had looked at a validated assessment of anxiety and depression in this population.
How did you assess their symptoms and what did you find?
We asked study participants to think back on how they felt throughout their teenage years and gauged their responses with the Patient Health Questionnaire (PHQ), a measure of mental health disorders. I should say up front that we didn’t have a control group. But we found that among our 414 study participants, somewhere between 30% and 50% reported clinically significant anxiety or depressive symptoms, far higher than you’d expect in the normal population. So when looking back on how they were feeling as teenagers in that environment, they were struggling, and they often felt rejected by their parents.
We also found that almost 10% of participants were threatened with eviction at some point in their childhood; 15% had to live outside of their home at some point, because of the clutter; and 2% had involvement from child protective services and were removed from the home.
I know you recruited patients from online forums established by the children of hoarding parents. Presumably, these are the people most affected by this phenomenon. How does this play out in people who simply like to, say, collect something? Is this a continuum of behavior, with a breaking point at which it becomes a pathology?
I think it’s safe to conceptualize collecting and hoarding as a continuum, and you’ve got to draw a line somewhere in terms of clinical significance.
Did you assess whether the children of hoarders were more likely to hoard themselves as adults?
This is our follow-up paper; we haven’t looked at it yet.
But in looking at preliminary data, the prevalence seems pretty low, actually, at least in our sample. And as you mentioned, in our study there were folks who were seeking support specifically because they grew up in a really cluttered home.
Management
How do mental health providers typically address and treat hoarding?
To my knowledge, there are no current Food and Drug Administration–approved medications for hoarding, though psychiatrists will prescribe SSRIs and try to treat co-occurring problems such as depression and anxiety symptoms.
I can speak to cognitive-behavioral therapy (CBT) in a bit more detail. A number of randomized controlled trials support CBT for hoarding. I mentioned before that when we as a field treated hoarding akin to OCD and did exposure and response prevention therapy, we didn’t really target the specific features of hoarding. People didn’t do that well.
But now researchers are focusing on CBT interventions focused on discarding tasks that really address hoarding. You can create different categories for different items: Patients can either keep them, throw them out, or donate them. You can explore what thoughts or expectations are associated with these items and try to address them. Clinicians can help patients look at, say, different areas of their house and discuss what they might be willing to part with or at least think about parting with. You find their internal motivations for keeping things.
This sort of therapy generally takes longer than it does for, say, OCD. It can be a little bit slower, particularly if someone has a lot of stuff. And often it can involve doing home visits. In the age of Zoom this is a little bit easier because home visits aren’t always feasible.
What role does family play in managing hoarding? I imagine that including loved ones and friends in the process could be quite helpful.
Yes, absolutely. And social support, more broadly.
A colleague I worked with did a really interesting study where she looked at psychologist-delivered versus peer-delivered CBT for hoarding. They found that the biggest predictor of improved outcomes was having what they called a “clutter buddy,” which follows the Alcoholics Anonymous sponsor model. This would be somebody else struggling with the same problem who’s an accountability partner helping a patient follow through with their goals related to discarding. I think that finding underscores how important that social support is.
Any final thoughts for our audience of clinicians and researchers on how to approach hoarding?
I think there’s been a stigma – at least in psychology circles – that it’s not really treatable because of that earlier work with OCD. But on the CBT side, there’s now good reason to believe that people can live much happier lives and overcome this problem. CBT does seem to work for a lot of people with hoarding. That’s what I’d like to emphasize.
Dr. Stetka is executive editor for Medscape. A version of this article first appeared on Medscape.com.
Many of us are reluctant to throw things out.
We buy. We accumulate. We collect. Eventually our attics are packed with dusty heirlooms that we rarely, if ever, look at. Eventually we’re forced to pare down and head to the Goodwill.
But not all of us.
Hoarding – or the prolonged difficulty of discarding unneeded possessions – is pervasive in our culture, affecting nearly 3% of the population. This compulsive collecting, and unwillingness to part with “stuff,” is even the subject of multiple popular television series.
How do you conceptualize hoarding behavior?
The core feature of hoarding is the inability to throw things away. This can be due to many different reasons, whether there’s a strong sentimental attachment or the belief that you will need these items one day. Compulsive buying is often involved, and inevitable clutter.
How was hoarding first conceptualized among psychiatrists and psychologists? And when did the term first enter the lexicon?
It was originally conceptualized as a difficult-to-treat subtype of obsessive-compulsive disorder (OCD). A lot of that work identifying this subgroup was going on in the late 1980s and early 1990s. There was a small but growing group of researchers demonstrating that this is fundamentally different from OCD in several ways.
In terms of the clinical presentation, the comorbidity patterns are different from those for OCD. And the course is a little bit different; we see a progressive development across the lifespan, as opposed to a clear-cut diagnosis earlier in life, as is typically seen with OCD. By the time a lot of people seek treatment, they’re often being brought in by, say, family members when they’re a little bit older. With hoarding, there is also this consistent pattern of poor treatment response across the board, whether to selective serotonin reuptake inhibitors or behavioral therapy.
A lot of this work together led to advocacy for recognizing hoarding as an independent diagnosis in the DSM-5. I think official recognition by our “big book” prompted more attention to this population. Previously these patients probably would have been diagnosed with OCD, and it really isn’t appropriate to think of hoarding as purely an anxiety disorder.
Hoarding exposure and future mental health
You have a new study, published in Annals of Clinical Psychiatry, looking at mental health among adult children of parents with hoarding problems. Can you tell us what inspired you to run this study, and what you found?
There were a couple of factors.
We’d seen a lot of folks with hoarding in OCD specialty clinics, so my clinical experiences with this population certainly drew me to this general area. But then, at the same time, I have this broad training in child mental health. And childhood trauma or adverse childhood experiences, which can include being around hoarding, can be a very difficult thing to live through and deal with. And here I have to give a lot of credit to Suzanne Chabaud, PhD, of the OCD Institute of Greater New Orleans, who’s one of the coauthors on the paper. She’s been beating the drum of thinking about the family and kids of people with hoarding disorders for years. My interests came from some of those experiences, but she had the good idea of really looking at this problem in a detailed way.
Prior to your paper, had there been research on the prevalence of mental illnesses such as anxiety and depression in the children of people with hoarding behaviors?
That particular question was new to our paper. It was the first time anyone, to my knowledge, had looked at a validated assessment of anxiety and depression in this population.
How did you assess their symptoms and what did you find?
We asked study participants to think back on how they felt throughout their teenage years and gauged their responses with the Patient Health Questionnaire (PHQ), a measure of mental health disorders. I should say up front that we didn’t have a control group. But we found that among our 414 study participants, somewhere between 30% and 50% reported clinically significant anxiety or depressive symptoms, far higher than you’d expect in the normal population. So when looking back on how they were feeling as teenagers in that environment, they were struggling, and they often felt rejected by their parents.
We also found that almost 10% of participants were threatened with eviction at some point in their childhood; 15% had to live outside of their home at some point, because of the clutter; and 2% had involvement from child protective services and were removed from the home.
I know you recruited patients from online forums established by the children of hoarding parents. Presumably, these are the people most affected by this phenomenon. How does this play out in people who simply like to, say, collect something? Is this a continuum of behavior, with a breaking point at which it becomes a pathology?
I think it’s safe to conceptualize collecting and hoarding as a continuum, and you’ve got to draw a line somewhere in terms of clinical significance.
Did you assess whether the children of hoarders were more likely to hoard themselves as adults?
This is our follow-up paper; we haven’t looked at it yet.
But in looking at preliminary data, the prevalence seems pretty low, actually, at least in our sample. And as you mentioned, in our study there were folks who were seeking support specifically because they grew up in a really cluttered home.
Management
How do mental health providers typically address and treat hoarding?
To my knowledge, there are no current Food and Drug Administration–approved medications for hoarding, though psychiatrists will prescribe SSRIs and try to treat co-occurring problems such as depression and anxiety symptoms.
I can speak to cognitive-behavioral therapy (CBT) in a bit more detail. A number of randomized controlled trials support CBT for hoarding. I mentioned before that when we as a field treated hoarding akin to OCD and did exposure and response prevention therapy, we didn’t really target the specific features of hoarding. People didn’t do that well.
But now researchers are focusing on CBT interventions focused on discarding tasks that really address hoarding. You can create different categories for different items: Patients can either keep them, throw them out, or donate them. You can explore what thoughts or expectations are associated with these items and try to address them. Clinicians can help patients look at, say, different areas of their house and discuss what they might be willing to part with or at least think about parting with. You find their internal motivations for keeping things.
This sort of therapy generally takes longer than it does for, say, OCD. It can be a little bit slower, particularly if someone has a lot of stuff. And often it can involve doing home visits. In the age of Zoom this is a little bit easier because home visits aren’t always feasible.
What role does family play in managing hoarding? I imagine that including loved ones and friends in the process could be quite helpful.
Yes, absolutely. And social support, more broadly.
A colleague I worked with did a really interesting study where she looked at psychologist-delivered versus peer-delivered CBT for hoarding. They found that the biggest predictor of improved outcomes was having what they called a “clutter buddy,” which follows the Alcoholics Anonymous sponsor model. This would be somebody else struggling with the same problem who’s an accountability partner helping a patient follow through with their goals related to discarding. I think that finding underscores how important that social support is.
Any final thoughts for our audience of clinicians and researchers on how to approach hoarding?
I think there’s been a stigma – at least in psychology circles – that it’s not really treatable because of that earlier work with OCD. But on the CBT side, there’s now good reason to believe that people can live much happier lives and overcome this problem. CBT does seem to work for a lot of people with hoarding. That’s what I’d like to emphasize.
Dr. Stetka is executive editor for Medscape. A version of this article first appeared on Medscape.com.
Cancer patients unaware of their increased thrombosis risk
It is up to their physician to discuss this with them.
This link is explained by the authors of an article in Cancer Treatment and Research Communications that reports results of a survey carried out by the European Cancer Patient Coalition (ECPC). “The aim of this pan-European patient survey was to assess patient awareness and knowledge about cancer-associated thrombosis (CAT), including risk factors, signs and symptoms, and interventions, to better prevent and treat CAT,” write the authors. “The idea was to create a sort of starting point for subsequent communication and information strategies and for comparing the results of any action taken in this area,” they add.
A roundtable discussion that included oncology healthcare professionals, policymakers, and patient advocates was convened to discuss and review the evidence regarding their ongoing concerns of excessive CAT-associated morbidity and mortality, as well as patients’ desire for greater CAT awareness.
“These discussions demonstrated that very little change had occurred over the years and that greater knowledge about CAT was still needed across the spectrum of healthcare practitioners and patients, particularly regarding primary and secondary prevention of thrombosis,” the authors write.
It was from this starting point that the idea for the pan-European survey was born. The ECPC, widely viewed as the “unified voice of cancer patients across Europe,” led the survey. This survey spanned six countries (France, Germany, Greece, Italy, United Kingdom, and Spain) and involved 1,365 patients and caregivers. The ECPC survey result was originally released at World Thrombosis Day in late 2018.
In an interview, Anna Falanga, MD, the main author of the article and professor of hematology at the University of Milan-Bicocca, Italy, reviewed the results and explained how to improve knowledge of CAT among patients with cancer.
“Data support that up to 20% of patients with cancer will experience venous thromboembolism (VTE), which is approximately 4–5 times higher than the general population,” said Dr. Falanga, who is also chief of the department of immunohematology and transfusion medicine and the Thrombosis and Hemostasis Center at the Hospital Papa Giovanni XXIII, in Bergamo, Italy.
“We have known about the link between thrombosis and cancer since the 19th century, but it has taken until midway through the last century for our level of understanding and awareness of the problem to reach its current level. Initially, this was limited to fundamental research, with large advances in our understanding of the mechanisms of the link between the two; it has only been more recently that we have had clinical studies that have piqued the interest of healthcare professionals, who were previously uninterested in the topic,” she said.
Poor understanding
One piece of data stands out from the European survey: Nearly three quarters of respondents (72%) said that before taking part in the survey, they were not aware that people with cancer have a higher-than-normal risk of developing thrombosis. “We asked participants to rate their overall understanding of CAT on a scale of 1 (low) to 10 (high), with the average (mean) score obtained being 4.1. Only 21% of patients gave a rating of 7 or above (high understanding). The average rating was very similar in the different countries surveyed,” write the authors. They note that the survey also assessed how much participants had learned about the topic from their physicians.
Approximately 35% of patients were made aware of CAT either immediately before or at the time of their cancer diagnosis. Of particular concern, one quarter (26%) of respondents (the largest proportion) noted that they first became aware of CAT when they suffered a blood clot. The average rating was very similar in the different countries surveyed. “Let us not forget that cancer and cancer treatments themselves cause a number of side effects, some of which can be very serious, so in some ways, a clot might be seen as a minor problem. Yet, in reality, it isn’t. It is a significant cause of death and disease in cancer patients,” said Dr. Falanga.
When discussing prevention, most respondents (87%) said they were aware that taking a walk could reduce their risk. Slightly fewer were aware that stopping smoking could reduce their risk (75%), and even fewer were aware that keeping hydrated (63%) and stretching their legs (55%) could reduce their risk.
Symptoms of CAT appeared to be relatively well known; 73% of survey participants indicated that they were aware that swelling in the foot, ankle, or leg could be a sign of DVT, and 71% indicated that shortness of breath could be a sign of pulmonary embolism (PE). “Other symptoms, however, were less well known, with just over half (57%) of participants being aware that pain, cramping, and tenderness could be a sign of DVT. About one third (33%) knew that irregular heartbeat could be a sign of PE. These results varied between countries,” according to the authors.
The survey highlighted that just over a third of respondents said that they were currently using anticoagulants, although almost all (96%) knew that anticoagulants could be used to effectively treat thrombosis. Only 41% of those using anticoagulants said they had been told about any possible side effects.
The Italian situation
The report containing the full results of the European survey goes even further, since, in addition to its overall results, it also gives information about individual countries.
The data from Italy, which are based on 246 persons, show that only 27% of patients and caregivers were aware of the increased risk of thrombosis after a cancer diagnosis. This figure is in line with the overall results of the survey, although the average score of the 10-point scale was lower for the Italy cohort (3.3/10 vs 4.1/10).
The results are more variable in terms of knowledge of risk factors. Most respondents (89%) said that they were aware of the risks related to inactivity. Just over half (52%), however, said that they were aware of the risks related to radiotherapy. Nevertheless, 75% of participants knew about the risks relating to cancer surgery and chemotherapy. “To all intents and purposes, all types of cancer drug can significantly affect the risk of developing a clot. And this is also the case for more modern types of treatment, such as immunotherapy,” said Dr. Falanga.
Most respondents reported that they got information about cancer-associated thrombosis verbally, usually from their hospital doctor (11%). Some respondents (6%) said that they found out about it from their own research, usually online. Almost 1 in 4 patients (24%) in Italy said that they first became aware of CAT when they suffered a blood clot. Answers to questions about knowledge of symptoms show that 58% of Italian patients and caregivers know that swelling of the lower limbs can be a symptom of DVT, and the same percentage knows that shortness of breath might indicate PE.
In terms of preventive action, the picture in Italy is somewhat variable: 74% of participants were aware of the importance of walking, but far fewer knew about the need to stop smoking (57%) and stretch the legs (35%). Of the 41% of Italians who were also taking an anticoagulant drug, 53% said that they knew about the possible side effects of such medication.
Which way forward?
“The high rate of CAT suggests that, despite the clinical evidence and clear guideline recommendations for patients with cancer, CAT prevention and recognition remain low among healthcare professionals,” the authors write.
The results of the ECPC survey further confirm those of previous studies, highlighting patients’ lack of knowledge about CAT and the need for more in-depth discussions between physician and patient.
So, what can be done? As highlighted by previous studies, “patients’ experiences are an education in themselves, particularly for the oncology care team,” the authors write. “Once the patient has a thrombosis, the opportunity for thrombosis prevention, which should be the most crucial focus of the care clinics (surgical, oncology, and palliative care), is gone,” they add.
“Oncology professionals, as well as other members of the patient’s care team (eg, internists, surgeons, nurses), need to perform better, at every stage of the patient’s cancer pathway, to ensure patients are aware of CAT and their individual risk to develop a blood clot,” said Dr. Falanga. She explained that in this group, it is the general practitioner who is the first contact. “These professionals are on the front line of the battle; they are among the first healthcare workers given the chance to suspect a clot and should, therefore, be fully aware of the increased risk in oncology patients,” she reiterated.
Experts agree on the fact that a multidisciplinary approach is of utmost importance in this context: the different roles in the team must be clear. “It is also fundamental to establish who does what in terms of educating and informing the patient,” said Dr. Falanga.
The researchers also put forward an example of a successful initiative: the Venous Thromboembolism Prevention in the Ambulatory Cancer Clinic (VTE-PACC) program. The initiative was developed by experts from the University of Vermont and was described in a recent article in JCO Oncology Practice.
Numerous resources are available online to help physicians talk to their patients and explain the risks linked to CAT along the continuum of cancer care. Among them is a resource titled, “Cancer Associated Thrombosis (CAT): Be Clot Conscious,” which can be found on the ECPC’s website.
“We have a collective responsibility using the ECPC patient survey as a baseline to inform patients with cancer on how to identify signs and symptoms of CAT to enable faster diagnosis and treatment,” the authors conclude.
This article was translated from Univadis Italy.
It is up to their physician to discuss this with them.
This link is explained by the authors of an article in Cancer Treatment and Research Communications that reports results of a survey carried out by the European Cancer Patient Coalition (ECPC). “The aim of this pan-European patient survey was to assess patient awareness and knowledge about cancer-associated thrombosis (CAT), including risk factors, signs and symptoms, and interventions, to better prevent and treat CAT,” write the authors. “The idea was to create a sort of starting point for subsequent communication and information strategies and for comparing the results of any action taken in this area,” they add.
A roundtable discussion that included oncology healthcare professionals, policymakers, and patient advocates was convened to discuss and review the evidence regarding their ongoing concerns of excessive CAT-associated morbidity and mortality, as well as patients’ desire for greater CAT awareness.
“These discussions demonstrated that very little change had occurred over the years and that greater knowledge about CAT was still needed across the spectrum of healthcare practitioners and patients, particularly regarding primary and secondary prevention of thrombosis,” the authors write.
It was from this starting point that the idea for the pan-European survey was born. The ECPC, widely viewed as the “unified voice of cancer patients across Europe,” led the survey. This survey spanned six countries (France, Germany, Greece, Italy, United Kingdom, and Spain) and involved 1,365 patients and caregivers. The ECPC survey result was originally released at World Thrombosis Day in late 2018.
In an interview, Anna Falanga, MD, the main author of the article and professor of hematology at the University of Milan-Bicocca, Italy, reviewed the results and explained how to improve knowledge of CAT among patients with cancer.
“Data support that up to 20% of patients with cancer will experience venous thromboembolism (VTE), which is approximately 4–5 times higher than the general population,” said Dr. Falanga, who is also chief of the department of immunohematology and transfusion medicine and the Thrombosis and Hemostasis Center at the Hospital Papa Giovanni XXIII, in Bergamo, Italy.
“We have known about the link between thrombosis and cancer since the 19th century, but it has taken until midway through the last century for our level of understanding and awareness of the problem to reach its current level. Initially, this was limited to fundamental research, with large advances in our understanding of the mechanisms of the link between the two; it has only been more recently that we have had clinical studies that have piqued the interest of healthcare professionals, who were previously uninterested in the topic,” she said.
Poor understanding
One piece of data stands out from the European survey: Nearly three quarters of respondents (72%) said that before taking part in the survey, they were not aware that people with cancer have a higher-than-normal risk of developing thrombosis. “We asked participants to rate their overall understanding of CAT on a scale of 1 (low) to 10 (high), with the average (mean) score obtained being 4.1. Only 21% of patients gave a rating of 7 or above (high understanding). The average rating was very similar in the different countries surveyed,” write the authors. They note that the survey also assessed how much participants had learned about the topic from their physicians.
Approximately 35% of patients were made aware of CAT either immediately before or at the time of their cancer diagnosis. Of particular concern, one quarter (26%) of respondents (the largest proportion) noted that they first became aware of CAT when they suffered a blood clot. The average rating was very similar in the different countries surveyed. “Let us not forget that cancer and cancer treatments themselves cause a number of side effects, some of which can be very serious, so in some ways, a clot might be seen as a minor problem. Yet, in reality, it isn’t. It is a significant cause of death and disease in cancer patients,” said Dr. Falanga.
When discussing prevention, most respondents (87%) said they were aware that taking a walk could reduce their risk. Slightly fewer were aware that stopping smoking could reduce their risk (75%), and even fewer were aware that keeping hydrated (63%) and stretching their legs (55%) could reduce their risk.
Symptoms of CAT appeared to be relatively well known; 73% of survey participants indicated that they were aware that swelling in the foot, ankle, or leg could be a sign of DVT, and 71% indicated that shortness of breath could be a sign of pulmonary embolism (PE). “Other symptoms, however, were less well known, with just over half (57%) of participants being aware that pain, cramping, and tenderness could be a sign of DVT. About one third (33%) knew that irregular heartbeat could be a sign of PE. These results varied between countries,” according to the authors.
The survey highlighted that just over a third of respondents said that they were currently using anticoagulants, although almost all (96%) knew that anticoagulants could be used to effectively treat thrombosis. Only 41% of those using anticoagulants said they had been told about any possible side effects.
The Italian situation
The report containing the full results of the European survey goes even further, since, in addition to its overall results, it also gives information about individual countries.
The data from Italy, which are based on 246 persons, show that only 27% of patients and caregivers were aware of the increased risk of thrombosis after a cancer diagnosis. This figure is in line with the overall results of the survey, although the average score of the 10-point scale was lower for the Italy cohort (3.3/10 vs 4.1/10).
The results are more variable in terms of knowledge of risk factors. Most respondents (89%) said that they were aware of the risks related to inactivity. Just over half (52%), however, said that they were aware of the risks related to radiotherapy. Nevertheless, 75% of participants knew about the risks relating to cancer surgery and chemotherapy. “To all intents and purposes, all types of cancer drug can significantly affect the risk of developing a clot. And this is also the case for more modern types of treatment, such as immunotherapy,” said Dr. Falanga.
Most respondents reported that they got information about cancer-associated thrombosis verbally, usually from their hospital doctor (11%). Some respondents (6%) said that they found out about it from their own research, usually online. Almost 1 in 4 patients (24%) in Italy said that they first became aware of CAT when they suffered a blood clot. Answers to questions about knowledge of symptoms show that 58% of Italian patients and caregivers know that swelling of the lower limbs can be a symptom of DVT, and the same percentage knows that shortness of breath might indicate PE.
In terms of preventive action, the picture in Italy is somewhat variable: 74% of participants were aware of the importance of walking, but far fewer knew about the need to stop smoking (57%) and stretch the legs (35%). Of the 41% of Italians who were also taking an anticoagulant drug, 53% said that they knew about the possible side effects of such medication.
Which way forward?
“The high rate of CAT suggests that, despite the clinical evidence and clear guideline recommendations for patients with cancer, CAT prevention and recognition remain low among healthcare professionals,” the authors write.
The results of the ECPC survey further confirm those of previous studies, highlighting patients’ lack of knowledge about CAT and the need for more in-depth discussions between physician and patient.
So, what can be done? As highlighted by previous studies, “patients’ experiences are an education in themselves, particularly for the oncology care team,” the authors write. “Once the patient has a thrombosis, the opportunity for thrombosis prevention, which should be the most crucial focus of the care clinics (surgical, oncology, and palliative care), is gone,” they add.
“Oncology professionals, as well as other members of the patient’s care team (eg, internists, surgeons, nurses), need to perform better, at every stage of the patient’s cancer pathway, to ensure patients are aware of CAT and their individual risk to develop a blood clot,” said Dr. Falanga. She explained that in this group, it is the general practitioner who is the first contact. “These professionals are on the front line of the battle; they are among the first healthcare workers given the chance to suspect a clot and should, therefore, be fully aware of the increased risk in oncology patients,” she reiterated.
Experts agree on the fact that a multidisciplinary approach is of utmost importance in this context: the different roles in the team must be clear. “It is also fundamental to establish who does what in terms of educating and informing the patient,” said Dr. Falanga.
The researchers also put forward an example of a successful initiative: the Venous Thromboembolism Prevention in the Ambulatory Cancer Clinic (VTE-PACC) program. The initiative was developed by experts from the University of Vermont and was described in a recent article in JCO Oncology Practice.
Numerous resources are available online to help physicians talk to their patients and explain the risks linked to CAT along the continuum of cancer care. Among them is a resource titled, “Cancer Associated Thrombosis (CAT): Be Clot Conscious,” which can be found on the ECPC’s website.
“We have a collective responsibility using the ECPC patient survey as a baseline to inform patients with cancer on how to identify signs and symptoms of CAT to enable faster diagnosis and treatment,” the authors conclude.
This article was translated from Univadis Italy.
It is up to their physician to discuss this with them.
This link is explained by the authors of an article in Cancer Treatment and Research Communications that reports results of a survey carried out by the European Cancer Patient Coalition (ECPC). “The aim of this pan-European patient survey was to assess patient awareness and knowledge about cancer-associated thrombosis (CAT), including risk factors, signs and symptoms, and interventions, to better prevent and treat CAT,” write the authors. “The idea was to create a sort of starting point for subsequent communication and information strategies and for comparing the results of any action taken in this area,” they add.
A roundtable discussion that included oncology healthcare professionals, policymakers, and patient advocates was convened to discuss and review the evidence regarding their ongoing concerns of excessive CAT-associated morbidity and mortality, as well as patients’ desire for greater CAT awareness.
“These discussions demonstrated that very little change had occurred over the years and that greater knowledge about CAT was still needed across the spectrum of healthcare practitioners and patients, particularly regarding primary and secondary prevention of thrombosis,” the authors write.
It was from this starting point that the idea for the pan-European survey was born. The ECPC, widely viewed as the “unified voice of cancer patients across Europe,” led the survey. This survey spanned six countries (France, Germany, Greece, Italy, United Kingdom, and Spain) and involved 1,365 patients and caregivers. The ECPC survey result was originally released at World Thrombosis Day in late 2018.
In an interview, Anna Falanga, MD, the main author of the article and professor of hematology at the University of Milan-Bicocca, Italy, reviewed the results and explained how to improve knowledge of CAT among patients with cancer.
“Data support that up to 20% of patients with cancer will experience venous thromboembolism (VTE), which is approximately 4–5 times higher than the general population,” said Dr. Falanga, who is also chief of the department of immunohematology and transfusion medicine and the Thrombosis and Hemostasis Center at the Hospital Papa Giovanni XXIII, in Bergamo, Italy.
“We have known about the link between thrombosis and cancer since the 19th century, but it has taken until midway through the last century for our level of understanding and awareness of the problem to reach its current level. Initially, this was limited to fundamental research, with large advances in our understanding of the mechanisms of the link between the two; it has only been more recently that we have had clinical studies that have piqued the interest of healthcare professionals, who were previously uninterested in the topic,” she said.
Poor understanding
One piece of data stands out from the European survey: Nearly three quarters of respondents (72%) said that before taking part in the survey, they were not aware that people with cancer have a higher-than-normal risk of developing thrombosis. “We asked participants to rate their overall understanding of CAT on a scale of 1 (low) to 10 (high), with the average (mean) score obtained being 4.1. Only 21% of patients gave a rating of 7 or above (high understanding). The average rating was very similar in the different countries surveyed,” write the authors. They note that the survey also assessed how much participants had learned about the topic from their physicians.
Approximately 35% of patients were made aware of CAT either immediately before or at the time of their cancer diagnosis. Of particular concern, one quarter (26%) of respondents (the largest proportion) noted that they first became aware of CAT when they suffered a blood clot. The average rating was very similar in the different countries surveyed. “Let us not forget that cancer and cancer treatments themselves cause a number of side effects, some of which can be very serious, so in some ways, a clot might be seen as a minor problem. Yet, in reality, it isn’t. It is a significant cause of death and disease in cancer patients,” said Dr. Falanga.
When discussing prevention, most respondents (87%) said they were aware that taking a walk could reduce their risk. Slightly fewer were aware that stopping smoking could reduce their risk (75%), and even fewer were aware that keeping hydrated (63%) and stretching their legs (55%) could reduce their risk.
Symptoms of CAT appeared to be relatively well known; 73% of survey participants indicated that they were aware that swelling in the foot, ankle, or leg could be a sign of DVT, and 71% indicated that shortness of breath could be a sign of pulmonary embolism (PE). “Other symptoms, however, were less well known, with just over half (57%) of participants being aware that pain, cramping, and tenderness could be a sign of DVT. About one third (33%) knew that irregular heartbeat could be a sign of PE. These results varied between countries,” according to the authors.
The survey highlighted that just over a third of respondents said that they were currently using anticoagulants, although almost all (96%) knew that anticoagulants could be used to effectively treat thrombosis. Only 41% of those using anticoagulants said they had been told about any possible side effects.
The Italian situation
The report containing the full results of the European survey goes even further, since, in addition to its overall results, it also gives information about individual countries.
The data from Italy, which are based on 246 persons, show that only 27% of patients and caregivers were aware of the increased risk of thrombosis after a cancer diagnosis. This figure is in line with the overall results of the survey, although the average score of the 10-point scale was lower for the Italy cohort (3.3/10 vs 4.1/10).
The results are more variable in terms of knowledge of risk factors. Most respondents (89%) said that they were aware of the risks related to inactivity. Just over half (52%), however, said that they were aware of the risks related to radiotherapy. Nevertheless, 75% of participants knew about the risks relating to cancer surgery and chemotherapy. “To all intents and purposes, all types of cancer drug can significantly affect the risk of developing a clot. And this is also the case for more modern types of treatment, such as immunotherapy,” said Dr. Falanga.
Most respondents reported that they got information about cancer-associated thrombosis verbally, usually from their hospital doctor (11%). Some respondents (6%) said that they found out about it from their own research, usually online. Almost 1 in 4 patients (24%) in Italy said that they first became aware of CAT when they suffered a blood clot. Answers to questions about knowledge of symptoms show that 58% of Italian patients and caregivers know that swelling of the lower limbs can be a symptom of DVT, and the same percentage knows that shortness of breath might indicate PE.
In terms of preventive action, the picture in Italy is somewhat variable: 74% of participants were aware of the importance of walking, but far fewer knew about the need to stop smoking (57%) and stretch the legs (35%). Of the 41% of Italians who were also taking an anticoagulant drug, 53% said that they knew about the possible side effects of such medication.
Which way forward?
“The high rate of CAT suggests that, despite the clinical evidence and clear guideline recommendations for patients with cancer, CAT prevention and recognition remain low among healthcare professionals,” the authors write.
The results of the ECPC survey further confirm those of previous studies, highlighting patients’ lack of knowledge about CAT and the need for more in-depth discussions between physician and patient.
So, what can be done? As highlighted by previous studies, “patients’ experiences are an education in themselves, particularly for the oncology care team,” the authors write. “Once the patient has a thrombosis, the opportunity for thrombosis prevention, which should be the most crucial focus of the care clinics (surgical, oncology, and palliative care), is gone,” they add.
“Oncology professionals, as well as other members of the patient’s care team (eg, internists, surgeons, nurses), need to perform better, at every stage of the patient’s cancer pathway, to ensure patients are aware of CAT and their individual risk to develop a blood clot,” said Dr. Falanga. She explained that in this group, it is the general practitioner who is the first contact. “These professionals are on the front line of the battle; they are among the first healthcare workers given the chance to suspect a clot and should, therefore, be fully aware of the increased risk in oncology patients,” she reiterated.
Experts agree on the fact that a multidisciplinary approach is of utmost importance in this context: the different roles in the team must be clear. “It is also fundamental to establish who does what in terms of educating and informing the patient,” said Dr. Falanga.
The researchers also put forward an example of a successful initiative: the Venous Thromboembolism Prevention in the Ambulatory Cancer Clinic (VTE-PACC) program. The initiative was developed by experts from the University of Vermont and was described in a recent article in JCO Oncology Practice.
Numerous resources are available online to help physicians talk to their patients and explain the risks linked to CAT along the continuum of cancer care. Among them is a resource titled, “Cancer Associated Thrombosis (CAT): Be Clot Conscious,” which can be found on the ECPC’s website.
“We have a collective responsibility using the ECPC patient survey as a baseline to inform patients with cancer on how to identify signs and symptoms of CAT to enable faster diagnosis and treatment,” the authors conclude.
This article was translated from Univadis Italy.
FROM CANCER TREATMENT AND RESEARCH COMMUNICATIONS







