User login
Bedside Arts
Hospital healthcare providers have supplemented clinical care with creative arts since the mid-20th century. For example, art and dance therapy have played a supporting role in hospital patient care since the 1930s. Using music to soothe cancer patients during treatment was pioneered at the University of Chicago Hospital as early as 1948. Music was also piped into some hospitals’ surgical suites to calm patients under various forms of spinal, local, or regional anesthesia.1
In recent years, there has been a groundswell of interest in both art therapy and the expressive arts in healthcare, resulting in the proliferation of bedside programs involving not only the visual arts and music, but also dance and creative writing.
According to the Art Therapy Credentials Board, “[A]rt therapy is a human service profession, which utilizes art media, images, the creative art process, and patient/client responses to the created art productions as reflections of an individual’s development, abilities, personality, concerns, and conflicts.”2 Anecdotal evidence has long supported the efficacy of art therapy in treating the chronically ill. But only recently have clinical studies proved that making art and the creative process it involves helps hospitalized patients heal in a quantifiable way.
One such study, conducted among adult cancer inpatients at Chicago’s Northwestern Memorial Hospital, was published in the February Journal of Pain and Symptom Management. That study determined that a series of one-hour art making sessions with a therapist yielded statistically significant decreases in a broad spectrum of symptoms, including pain, fatigue, depression, anxiety, lack of appetite, and shortness of breath. It also helped reduce apprehension, tension, nervousness, and worry. In addition to the quantifiable positive effects of art making, “subjects made numerous anecdotal comments that the art therapy had energized them.”3
But art therapy, administered by a credentialed practitioner with the specific goal of treating emotional and psychological issues associated with illness as a clinical practice, is not the only type of bedside artistic production happening in hospitals. Expressive art making, which falls under the umbrella of the arts in healthcare movement, has gained a significant foothold. In 2002 the National Endowment for the Arts (NEA), which funds arts in healthcare research, issued a call for creative artists not specifically trained as art therapists to play a larger role in patient care.
According to Elizabeth A. Curry, MA, coordinator of the Mayo Clinic Center for Humanities in Medicine, expressive bedside art-making has a different goal than therapy.
“The art therapist is part of the care plan team,” says Curry. “She writes in the charts.” As guided by a creative artist rather than by a therapist, the very experience of making art—rather than the information a finished work of art may furnish the care plan team—is central to the undertaking. It is the artist’s experience with the patient and the patient’s experience with the media that are important—not the end result. Of this model of bedside art making, Curry says, “it has no therapeutic goal other than to relieve stress.” The scope of expressive bedside art creation administered by an arts in healthcare program is potentially much broader than one that is therapy-based and has the potential to reach more patients.
Among studies of a number of arts-in-healthcare programs, the NEA cites the success of Healing Icons, an art-support program for young adult cancer patients age 16 and older.
According to the NEA, “In the program, patients create a three-dimensional mixed-media art piece to convey a unique personal perspective on receiving a diagnosis of cancer and then experiencing treatments.”
The mixed-media piece “provides a way for unstructured expression of feelings and thoughts.”4 The NEA also points to an article published in The Lancet (May 2001) that discussed the creative output of several expressive arts programs implemented in the United Kingdom. In one, comic artists held a series of workshops with young patients at the Great Ormond Street Children’s Hospital. Those workshops resulted in a comic book, HospiTales. Not only did this undertaking produce “interesting therapeutic and creative results” for the participants, the finished HospiTales promoted “a positive view of being in hospital, which makes it seem a less scary place for young patients.”5
Not surprisingly, the number of new arts in healthcare initiatives has continued to grow. For two months, beginning in August, for example, the Mayo Clinic Center for Humanities in Medicine began a pilot program in conjunction with the Rochester Arts Center in Minnesota to bring bedside art making to the Hematology Department. The Hematology Department, in particular, appears to be the ideal place to conduct such a pilot program. “People are stuck in the hospital with a lot of uncertainty, stress, and discomfort,” says Mayo’s Curry. “Making art can give people back a sense of control and relieve some anxiety.”
At the outset, Education Coordinator Michele Heidel of the Rochester Art Center will work with the nursing staff to identify 20 patients who might be interested in participating in what have been termed “art interventions.” Patients will paint and draw with professional artist-educators, who will offer participants a variety of media such as oil pastels, chalk, charcoal, and watercolors in which to work. These materials are chosen not only for their ease of use, but because they are safe, nontoxic, clean, and conform to ASTM Standard D-4236 Practice for Labeling Art Materials for Chronic Health Hazards. They also carry No-Odor labeling.
Prior to engaging with patients, artist-educators receive training in infection control, including OSHA Bloodborne Pathogen training and instruction in disinfecting equipment, art supplies, and work surfaces. They are also briefed on HIPAA compliance. Attendance at the Arts in Healthcare Summer Intensive Training at the University of Florida (Gainesville) and a site visit to the Mayo Clinic Jacksonville Arts at the Bedside program completes their orientation. Though they are not officially part of the care plan team, artist-educators also attend hematology inpatient rounds.
Each of the 20 patients chosen to participate in the pilot study will be assessed both before and after working with the educator-artist by means of questionnaires, as well as by Visual Analogue Scales to see how a single art intervention affects anxiety, discomfort, and stress. Ultimately, the purpose of this benefactor-funded pilot program is to provide quantifiable evidence for the efficacy of bedside art making.
According to Curry, the Center for Humanities in Medicine would like to grow the program significantly, eventually offering patients a menu of choices of creative arts in which to participate. This menu would also include music, dance, and creative writing. “It’s a big goal for the future,” says Curry.
For patients participating in Mayo’s pilot study, talent or artistic ability is not an issue. According to Curry, the program is process oriented rather than project oriented. Unlike the HospiTales project, the pilot study focuses on the relationship among the patient, the artist, and the media rather than on creating a finished piece. The Mayo Clinic’s Center for Humanities in Medicine has no specific plans either to exhibit or publish any of the artistic productions created by study participants.
“There will be a lot of amazing art and amazing writing,” says Curry. But the legal technicalities involved in publishing or mounting an exhibition of art work, including the necessity of having patients give permission and sign release forms, may simply be too daunting for those involved. Curry does not, however, rule out an exhibit or a book of patient work in the future.
In conjunction with Arizona State University, the Mayo Clinic’s Scottsdale center has also introduced several arts programs, including music at the bedside in Palliative Care, and a bedside creative writing program. During sessions that last about 45 minutes and center around the one-on-one interaction between the artist and the patient, patients narrate their personal stories, from which participating writers generate original works on hand-made paper. The finished pieces are then returned to the patient-narrators. These works have proved extremely meaningful not only to the people whose stories they tell, but to the storytellers’ families as well.
Based on its own successful programs, which include bedside art making, the Integrative Medicine department at Hartford Hospital in Connecticut has published an on-line Program Development Manual, “Building Bridges,” which provides “a blueprint for spanning the not-yet-connected terrain of Conventional Medicine and Complementary and Alternative Medicine,”6 Indeed, in addition to sections dealing such practices as massage therapy, acupuncture, Reiki, and Tai Chi, as well as guided imagery, “a mind-body intervention that focuses the imagination and the five senses to create soothing and relaxing images,”7 “Building Bridges” includes a chapter on “Creating an Art for Healing Program,” written by Diana S. Boehnert, artist-in-residence and coordinator of the Art for Healing Program.
According to Boehnert, art making as part of a larger Integrative Medicine program “creates a better quality of life for people with chronic illness.” Hartford Hospital’s program, which she administers, employs both clinically trained art therapy interns and volunteers, whose work follows the expressive bedside art making model. As such, the Art for Healing section of “Building Bridges” deals extensively with the training and preparation of artists. According to the manual, candidates without previous experience working in a hospital setting benefit from partnering with a clinical staff member as part of the training process. In addition to the requisite “orientation to patient care area with review of patient care environment, equipment, safety issues, and the needs of the specific patent population,” “Building Bridges” suggests that trainees also engage in “mock art sessions with a preceptor or mentor.”8 While it is also recommended that candidates have some background in the expressive arts, formal art training is not an absolute requirement. In reality, says Boehnert, “It doesn’t matter how much [formal art] training they have, the patient does the work.”
Unlike the Mayo Clinic’s pilot study, Hartford Hospital’s program is project oriented. “The project is the impetus that gets the patient going,” says Boehnert. “Adults aren’t willing to play without a purpose. They just want a little direction.”
For the most part, individual projects are small. They range from mandalas (circular designs generally associated with Buddhist and Hindu practice) to cards for family members. “Intuition,” explains Boehnert, “tells the volunteers what will work best with a patient.”
Hartford Hospital’s Arts for Healing is not limited to patients in a single department. Boehnert, whose previous experience with arts in healthcare included plaster cast mask-making with survivors of domestic violence, began working with rehab patients and extended the program to include dialysis patients. It’s now available in various departments throughout the hospital. Some of the work created by Arts for Healing participants in the Art for Healing program is on display in a small gallery in the hospital.
According to Boehnert, patient response to arts initiatives like the ones advocated in “Building Bridges” has been overwhelmingly positive. As an example, she cites a heart transplant patient who was introduced to the expressive arts during his six-week stay at Hartford Hospital. Before he was discharged, he created his own little gallery in his room. A patient being treated for leukemia also created an impressive body of work, giving pieces away to cheer up fellow patients who were not having good days. Staff, too, says Boehnert, benefit from Art for Healing: “My volunteers also go home better than when they came.”
Since 1991, the Society for the Arts in Healthcare (SAH) has provided support for programs such as the ones at Hartford Hospital and the Mayo Clinic, as well as others like the Artists in Residence program at Florida’s Shands HealthCare hospitals, which offers patients a variety of bedside art making activities. Examples include Art Infusion, a multi-media program for adults on chemotherapy, creative arts for pediatric inpatients, and (like Mayo Scottsdale) an oral history program which seeks to transcribe patients’ personal stories.
“In a lot of places, funding is a struggle,” explains Curry. To help secure funding for arts in healthcare programs, the SAH provides grant opportunities, like the SAH/Johnson & Johnson Partnership to Promote Arts and Healing and SAH Consulting Grants, as well as several awards.
In April, the SAH hosted its 15th international arts in healthcare conference in Chicago, the topic of which was “Vision + Voice—Charting the Course of Arts, Health and Medicine.” The conference urged attendees to “focus (their) vision for the future.”9 Given ever-increasing interest in integrating the arts into healthcare—especially inpatient care—be it by means of the clinical practice of art therapy or by expressive, creative arts programs, the future of such programs seems bright. As Dana Gioia, chair of the NEA, says: “The arts have an extraordinary ability to enhance our lives, to help us heal, and to bring us comfort in times of great stress. We must reconnect the arts with the actual human existence that Americans lead, the journeys we take in life, which lead us through hospitals, to hospices, to the end of life.”10 TH
Roberta Newman is based in Brooklyn, N.Y.
References
- NEA News Room: Arts in Healthcare Research. Available at: http://arts.endow.gov/news/news03/AIHResearch.html. Last accessed June 16, 2006.
- Art Therapy Credentials Board, “What is Art Therapy?” Available at: www.atcb.org. Last accessed June 16, 2006.
- Nainis N, Paice J, Ratner J. Relieving symptoms in cancer: innovative use of art therapy. J Pain Symptom Manage. 2006 Feb;31(2):162-169.
- Arts in Healthcare Research. National Endowment For the Arts New Room. Available at: http://arts.endow.gov/news/news03/AIHResearch.html. Last accessed June 12, 2006.
- Foster H. Medical settings foster the creation of art. Lancet. 2001;357(9268):1627.
- Foreword. “Building Bridges.” Available at: www.harthosp.org/IntMed/manual/foreword.asp. Last accessed June 18, 2006.
- Guided Imagery. “Building Bridges.” Available at www.harthosp.org/IntMed/manual/guidedimagery.asp. Last accessed June 18, 2006.
- Creating an Art for Healing Program: Training. “Building Bridges.” Available at: www.harthosp.org/IntMed/manual/art.asp. Last accessed June 18, 2006.
- Vision +Voice—Charting the Course of Arts, Health and Medicine Society for the Arts in Healthcare 15th Annual International Conference Program. Available at: www.thesah.org/doc/FINAL%20program.pdf. Last accessed June 18, 2006.
- Society for the Arts in Healthcare Fact Sheet. Available at: www.thesah.org. Last accessed June 19, 2006.
Hospital healthcare providers have supplemented clinical care with creative arts since the mid-20th century. For example, art and dance therapy have played a supporting role in hospital patient care since the 1930s. Using music to soothe cancer patients during treatment was pioneered at the University of Chicago Hospital as early as 1948. Music was also piped into some hospitals’ surgical suites to calm patients under various forms of spinal, local, or regional anesthesia.1
In recent years, there has been a groundswell of interest in both art therapy and the expressive arts in healthcare, resulting in the proliferation of bedside programs involving not only the visual arts and music, but also dance and creative writing.
According to the Art Therapy Credentials Board, “[A]rt therapy is a human service profession, which utilizes art media, images, the creative art process, and patient/client responses to the created art productions as reflections of an individual’s development, abilities, personality, concerns, and conflicts.”2 Anecdotal evidence has long supported the efficacy of art therapy in treating the chronically ill. But only recently have clinical studies proved that making art and the creative process it involves helps hospitalized patients heal in a quantifiable way.
One such study, conducted among adult cancer inpatients at Chicago’s Northwestern Memorial Hospital, was published in the February Journal of Pain and Symptom Management. That study determined that a series of one-hour art making sessions with a therapist yielded statistically significant decreases in a broad spectrum of symptoms, including pain, fatigue, depression, anxiety, lack of appetite, and shortness of breath. It also helped reduce apprehension, tension, nervousness, and worry. In addition to the quantifiable positive effects of art making, “subjects made numerous anecdotal comments that the art therapy had energized them.”3
But art therapy, administered by a credentialed practitioner with the specific goal of treating emotional and psychological issues associated with illness as a clinical practice, is not the only type of bedside artistic production happening in hospitals. Expressive art making, which falls under the umbrella of the arts in healthcare movement, has gained a significant foothold. In 2002 the National Endowment for the Arts (NEA), which funds arts in healthcare research, issued a call for creative artists not specifically trained as art therapists to play a larger role in patient care.
According to Elizabeth A. Curry, MA, coordinator of the Mayo Clinic Center for Humanities in Medicine, expressive bedside art-making has a different goal than therapy.
“The art therapist is part of the care plan team,” says Curry. “She writes in the charts.” As guided by a creative artist rather than by a therapist, the very experience of making art—rather than the information a finished work of art may furnish the care plan team—is central to the undertaking. It is the artist’s experience with the patient and the patient’s experience with the media that are important—not the end result. Of this model of bedside art making, Curry says, “it has no therapeutic goal other than to relieve stress.” The scope of expressive bedside art creation administered by an arts in healthcare program is potentially much broader than one that is therapy-based and has the potential to reach more patients.
Among studies of a number of arts-in-healthcare programs, the NEA cites the success of Healing Icons, an art-support program for young adult cancer patients age 16 and older.
According to the NEA, “In the program, patients create a three-dimensional mixed-media art piece to convey a unique personal perspective on receiving a diagnosis of cancer and then experiencing treatments.”
The mixed-media piece “provides a way for unstructured expression of feelings and thoughts.”4 The NEA also points to an article published in The Lancet (May 2001) that discussed the creative output of several expressive arts programs implemented in the United Kingdom. In one, comic artists held a series of workshops with young patients at the Great Ormond Street Children’s Hospital. Those workshops resulted in a comic book, HospiTales. Not only did this undertaking produce “interesting therapeutic and creative results” for the participants, the finished HospiTales promoted “a positive view of being in hospital, which makes it seem a less scary place for young patients.”5
Not surprisingly, the number of new arts in healthcare initiatives has continued to grow. For two months, beginning in August, for example, the Mayo Clinic Center for Humanities in Medicine began a pilot program in conjunction with the Rochester Arts Center in Minnesota to bring bedside art making to the Hematology Department. The Hematology Department, in particular, appears to be the ideal place to conduct such a pilot program. “People are stuck in the hospital with a lot of uncertainty, stress, and discomfort,” says Mayo’s Curry. “Making art can give people back a sense of control and relieve some anxiety.”
At the outset, Education Coordinator Michele Heidel of the Rochester Art Center will work with the nursing staff to identify 20 patients who might be interested in participating in what have been termed “art interventions.” Patients will paint and draw with professional artist-educators, who will offer participants a variety of media such as oil pastels, chalk, charcoal, and watercolors in which to work. These materials are chosen not only for their ease of use, but because they are safe, nontoxic, clean, and conform to ASTM Standard D-4236 Practice for Labeling Art Materials for Chronic Health Hazards. They also carry No-Odor labeling.
Prior to engaging with patients, artist-educators receive training in infection control, including OSHA Bloodborne Pathogen training and instruction in disinfecting equipment, art supplies, and work surfaces. They are also briefed on HIPAA compliance. Attendance at the Arts in Healthcare Summer Intensive Training at the University of Florida (Gainesville) and a site visit to the Mayo Clinic Jacksonville Arts at the Bedside program completes their orientation. Though they are not officially part of the care plan team, artist-educators also attend hematology inpatient rounds.
Each of the 20 patients chosen to participate in the pilot study will be assessed both before and after working with the educator-artist by means of questionnaires, as well as by Visual Analogue Scales to see how a single art intervention affects anxiety, discomfort, and stress. Ultimately, the purpose of this benefactor-funded pilot program is to provide quantifiable evidence for the efficacy of bedside art making.
According to Curry, the Center for Humanities in Medicine would like to grow the program significantly, eventually offering patients a menu of choices of creative arts in which to participate. This menu would also include music, dance, and creative writing. “It’s a big goal for the future,” says Curry.
For patients participating in Mayo’s pilot study, talent or artistic ability is not an issue. According to Curry, the program is process oriented rather than project oriented. Unlike the HospiTales project, the pilot study focuses on the relationship among the patient, the artist, and the media rather than on creating a finished piece. The Mayo Clinic’s Center for Humanities in Medicine has no specific plans either to exhibit or publish any of the artistic productions created by study participants.
“There will be a lot of amazing art and amazing writing,” says Curry. But the legal technicalities involved in publishing or mounting an exhibition of art work, including the necessity of having patients give permission and sign release forms, may simply be too daunting for those involved. Curry does not, however, rule out an exhibit or a book of patient work in the future.
In conjunction with Arizona State University, the Mayo Clinic’s Scottsdale center has also introduced several arts programs, including music at the bedside in Palliative Care, and a bedside creative writing program. During sessions that last about 45 minutes and center around the one-on-one interaction between the artist and the patient, patients narrate their personal stories, from which participating writers generate original works on hand-made paper. The finished pieces are then returned to the patient-narrators. These works have proved extremely meaningful not only to the people whose stories they tell, but to the storytellers’ families as well.
Based on its own successful programs, which include bedside art making, the Integrative Medicine department at Hartford Hospital in Connecticut has published an on-line Program Development Manual, “Building Bridges,” which provides “a blueprint for spanning the not-yet-connected terrain of Conventional Medicine and Complementary and Alternative Medicine,”6 Indeed, in addition to sections dealing such practices as massage therapy, acupuncture, Reiki, and Tai Chi, as well as guided imagery, “a mind-body intervention that focuses the imagination and the five senses to create soothing and relaxing images,”7 “Building Bridges” includes a chapter on “Creating an Art for Healing Program,” written by Diana S. Boehnert, artist-in-residence and coordinator of the Art for Healing Program.
According to Boehnert, art making as part of a larger Integrative Medicine program “creates a better quality of life for people with chronic illness.” Hartford Hospital’s program, which she administers, employs both clinically trained art therapy interns and volunteers, whose work follows the expressive bedside art making model. As such, the Art for Healing section of “Building Bridges” deals extensively with the training and preparation of artists. According to the manual, candidates without previous experience working in a hospital setting benefit from partnering with a clinical staff member as part of the training process. In addition to the requisite “orientation to patient care area with review of patient care environment, equipment, safety issues, and the needs of the specific patent population,” “Building Bridges” suggests that trainees also engage in “mock art sessions with a preceptor or mentor.”8 While it is also recommended that candidates have some background in the expressive arts, formal art training is not an absolute requirement. In reality, says Boehnert, “It doesn’t matter how much [formal art] training they have, the patient does the work.”
Unlike the Mayo Clinic’s pilot study, Hartford Hospital’s program is project oriented. “The project is the impetus that gets the patient going,” says Boehnert. “Adults aren’t willing to play without a purpose. They just want a little direction.”
For the most part, individual projects are small. They range from mandalas (circular designs generally associated with Buddhist and Hindu practice) to cards for family members. “Intuition,” explains Boehnert, “tells the volunteers what will work best with a patient.”
Hartford Hospital’s Arts for Healing is not limited to patients in a single department. Boehnert, whose previous experience with arts in healthcare included plaster cast mask-making with survivors of domestic violence, began working with rehab patients and extended the program to include dialysis patients. It’s now available in various departments throughout the hospital. Some of the work created by Arts for Healing participants in the Art for Healing program is on display in a small gallery in the hospital.
According to Boehnert, patient response to arts initiatives like the ones advocated in “Building Bridges” has been overwhelmingly positive. As an example, she cites a heart transplant patient who was introduced to the expressive arts during his six-week stay at Hartford Hospital. Before he was discharged, he created his own little gallery in his room. A patient being treated for leukemia also created an impressive body of work, giving pieces away to cheer up fellow patients who were not having good days. Staff, too, says Boehnert, benefit from Art for Healing: “My volunteers also go home better than when they came.”
Since 1991, the Society for the Arts in Healthcare (SAH) has provided support for programs such as the ones at Hartford Hospital and the Mayo Clinic, as well as others like the Artists in Residence program at Florida’s Shands HealthCare hospitals, which offers patients a variety of bedside art making activities. Examples include Art Infusion, a multi-media program for adults on chemotherapy, creative arts for pediatric inpatients, and (like Mayo Scottsdale) an oral history program which seeks to transcribe patients’ personal stories.
“In a lot of places, funding is a struggle,” explains Curry. To help secure funding for arts in healthcare programs, the SAH provides grant opportunities, like the SAH/Johnson & Johnson Partnership to Promote Arts and Healing and SAH Consulting Grants, as well as several awards.
In April, the SAH hosted its 15th international arts in healthcare conference in Chicago, the topic of which was “Vision + Voice—Charting the Course of Arts, Health and Medicine.” The conference urged attendees to “focus (their) vision for the future.”9 Given ever-increasing interest in integrating the arts into healthcare—especially inpatient care—be it by means of the clinical practice of art therapy or by expressive, creative arts programs, the future of such programs seems bright. As Dana Gioia, chair of the NEA, says: “The arts have an extraordinary ability to enhance our lives, to help us heal, and to bring us comfort in times of great stress. We must reconnect the arts with the actual human existence that Americans lead, the journeys we take in life, which lead us through hospitals, to hospices, to the end of life.”10 TH
Roberta Newman is based in Brooklyn, N.Y.
References
- NEA News Room: Arts in Healthcare Research. Available at: http://arts.endow.gov/news/news03/AIHResearch.html. Last accessed June 16, 2006.
- Art Therapy Credentials Board, “What is Art Therapy?” Available at: www.atcb.org. Last accessed June 16, 2006.
- Nainis N, Paice J, Ratner J. Relieving symptoms in cancer: innovative use of art therapy. J Pain Symptom Manage. 2006 Feb;31(2):162-169.
- Arts in Healthcare Research. National Endowment For the Arts New Room. Available at: http://arts.endow.gov/news/news03/AIHResearch.html. Last accessed June 12, 2006.
- Foster H. Medical settings foster the creation of art. Lancet. 2001;357(9268):1627.
- Foreword. “Building Bridges.” Available at: www.harthosp.org/IntMed/manual/foreword.asp. Last accessed June 18, 2006.
- Guided Imagery. “Building Bridges.” Available at www.harthosp.org/IntMed/manual/guidedimagery.asp. Last accessed June 18, 2006.
- Creating an Art for Healing Program: Training. “Building Bridges.” Available at: www.harthosp.org/IntMed/manual/art.asp. Last accessed June 18, 2006.
- Vision +Voice—Charting the Course of Arts, Health and Medicine Society for the Arts in Healthcare 15th Annual International Conference Program. Available at: www.thesah.org/doc/FINAL%20program.pdf. Last accessed June 18, 2006.
- Society for the Arts in Healthcare Fact Sheet. Available at: www.thesah.org. Last accessed June 19, 2006.
Hospital healthcare providers have supplemented clinical care with creative arts since the mid-20th century. For example, art and dance therapy have played a supporting role in hospital patient care since the 1930s. Using music to soothe cancer patients during treatment was pioneered at the University of Chicago Hospital as early as 1948. Music was also piped into some hospitals’ surgical suites to calm patients under various forms of spinal, local, or regional anesthesia.1
In recent years, there has been a groundswell of interest in both art therapy and the expressive arts in healthcare, resulting in the proliferation of bedside programs involving not only the visual arts and music, but also dance and creative writing.
According to the Art Therapy Credentials Board, “[A]rt therapy is a human service profession, which utilizes art media, images, the creative art process, and patient/client responses to the created art productions as reflections of an individual’s development, abilities, personality, concerns, and conflicts.”2 Anecdotal evidence has long supported the efficacy of art therapy in treating the chronically ill. But only recently have clinical studies proved that making art and the creative process it involves helps hospitalized patients heal in a quantifiable way.
One such study, conducted among adult cancer inpatients at Chicago’s Northwestern Memorial Hospital, was published in the February Journal of Pain and Symptom Management. That study determined that a series of one-hour art making sessions with a therapist yielded statistically significant decreases in a broad spectrum of symptoms, including pain, fatigue, depression, anxiety, lack of appetite, and shortness of breath. It also helped reduce apprehension, tension, nervousness, and worry. In addition to the quantifiable positive effects of art making, “subjects made numerous anecdotal comments that the art therapy had energized them.”3
But art therapy, administered by a credentialed practitioner with the specific goal of treating emotional and psychological issues associated with illness as a clinical practice, is not the only type of bedside artistic production happening in hospitals. Expressive art making, which falls under the umbrella of the arts in healthcare movement, has gained a significant foothold. In 2002 the National Endowment for the Arts (NEA), which funds arts in healthcare research, issued a call for creative artists not specifically trained as art therapists to play a larger role in patient care.
According to Elizabeth A. Curry, MA, coordinator of the Mayo Clinic Center for Humanities in Medicine, expressive bedside art-making has a different goal than therapy.
“The art therapist is part of the care plan team,” says Curry. “She writes in the charts.” As guided by a creative artist rather than by a therapist, the very experience of making art—rather than the information a finished work of art may furnish the care plan team—is central to the undertaking. It is the artist’s experience with the patient and the patient’s experience with the media that are important—not the end result. Of this model of bedside art making, Curry says, “it has no therapeutic goal other than to relieve stress.” The scope of expressive bedside art creation administered by an arts in healthcare program is potentially much broader than one that is therapy-based and has the potential to reach more patients.
Among studies of a number of arts-in-healthcare programs, the NEA cites the success of Healing Icons, an art-support program for young adult cancer patients age 16 and older.
According to the NEA, “In the program, patients create a three-dimensional mixed-media art piece to convey a unique personal perspective on receiving a diagnosis of cancer and then experiencing treatments.”
The mixed-media piece “provides a way for unstructured expression of feelings and thoughts.”4 The NEA also points to an article published in The Lancet (May 2001) that discussed the creative output of several expressive arts programs implemented in the United Kingdom. In one, comic artists held a series of workshops with young patients at the Great Ormond Street Children’s Hospital. Those workshops resulted in a comic book, HospiTales. Not only did this undertaking produce “interesting therapeutic and creative results” for the participants, the finished HospiTales promoted “a positive view of being in hospital, which makes it seem a less scary place for young patients.”5
Not surprisingly, the number of new arts in healthcare initiatives has continued to grow. For two months, beginning in August, for example, the Mayo Clinic Center for Humanities in Medicine began a pilot program in conjunction with the Rochester Arts Center in Minnesota to bring bedside art making to the Hematology Department. The Hematology Department, in particular, appears to be the ideal place to conduct such a pilot program. “People are stuck in the hospital with a lot of uncertainty, stress, and discomfort,” says Mayo’s Curry. “Making art can give people back a sense of control and relieve some anxiety.”
At the outset, Education Coordinator Michele Heidel of the Rochester Art Center will work with the nursing staff to identify 20 patients who might be interested in participating in what have been termed “art interventions.” Patients will paint and draw with professional artist-educators, who will offer participants a variety of media such as oil pastels, chalk, charcoal, and watercolors in which to work. These materials are chosen not only for their ease of use, but because they are safe, nontoxic, clean, and conform to ASTM Standard D-4236 Practice for Labeling Art Materials for Chronic Health Hazards. They also carry No-Odor labeling.
Prior to engaging with patients, artist-educators receive training in infection control, including OSHA Bloodborne Pathogen training and instruction in disinfecting equipment, art supplies, and work surfaces. They are also briefed on HIPAA compliance. Attendance at the Arts in Healthcare Summer Intensive Training at the University of Florida (Gainesville) and a site visit to the Mayo Clinic Jacksonville Arts at the Bedside program completes their orientation. Though they are not officially part of the care plan team, artist-educators also attend hematology inpatient rounds.
Each of the 20 patients chosen to participate in the pilot study will be assessed both before and after working with the educator-artist by means of questionnaires, as well as by Visual Analogue Scales to see how a single art intervention affects anxiety, discomfort, and stress. Ultimately, the purpose of this benefactor-funded pilot program is to provide quantifiable evidence for the efficacy of bedside art making.
According to Curry, the Center for Humanities in Medicine would like to grow the program significantly, eventually offering patients a menu of choices of creative arts in which to participate. This menu would also include music, dance, and creative writing. “It’s a big goal for the future,” says Curry.
For patients participating in Mayo’s pilot study, talent or artistic ability is not an issue. According to Curry, the program is process oriented rather than project oriented. Unlike the HospiTales project, the pilot study focuses on the relationship among the patient, the artist, and the media rather than on creating a finished piece. The Mayo Clinic’s Center for Humanities in Medicine has no specific plans either to exhibit or publish any of the artistic productions created by study participants.
“There will be a lot of amazing art and amazing writing,” says Curry. But the legal technicalities involved in publishing or mounting an exhibition of art work, including the necessity of having patients give permission and sign release forms, may simply be too daunting for those involved. Curry does not, however, rule out an exhibit or a book of patient work in the future.
In conjunction with Arizona State University, the Mayo Clinic’s Scottsdale center has also introduced several arts programs, including music at the bedside in Palliative Care, and a bedside creative writing program. During sessions that last about 45 minutes and center around the one-on-one interaction between the artist and the patient, patients narrate their personal stories, from which participating writers generate original works on hand-made paper. The finished pieces are then returned to the patient-narrators. These works have proved extremely meaningful not only to the people whose stories they tell, but to the storytellers’ families as well.
Based on its own successful programs, which include bedside art making, the Integrative Medicine department at Hartford Hospital in Connecticut has published an on-line Program Development Manual, “Building Bridges,” which provides “a blueprint for spanning the not-yet-connected terrain of Conventional Medicine and Complementary and Alternative Medicine,”6 Indeed, in addition to sections dealing such practices as massage therapy, acupuncture, Reiki, and Tai Chi, as well as guided imagery, “a mind-body intervention that focuses the imagination and the five senses to create soothing and relaxing images,”7 “Building Bridges” includes a chapter on “Creating an Art for Healing Program,” written by Diana S. Boehnert, artist-in-residence and coordinator of the Art for Healing Program.
According to Boehnert, art making as part of a larger Integrative Medicine program “creates a better quality of life for people with chronic illness.” Hartford Hospital’s program, which she administers, employs both clinically trained art therapy interns and volunteers, whose work follows the expressive bedside art making model. As such, the Art for Healing section of “Building Bridges” deals extensively with the training and preparation of artists. According to the manual, candidates without previous experience working in a hospital setting benefit from partnering with a clinical staff member as part of the training process. In addition to the requisite “orientation to patient care area with review of patient care environment, equipment, safety issues, and the needs of the specific patent population,” “Building Bridges” suggests that trainees also engage in “mock art sessions with a preceptor or mentor.”8 While it is also recommended that candidates have some background in the expressive arts, formal art training is not an absolute requirement. In reality, says Boehnert, “It doesn’t matter how much [formal art] training they have, the patient does the work.”
Unlike the Mayo Clinic’s pilot study, Hartford Hospital’s program is project oriented. “The project is the impetus that gets the patient going,” says Boehnert. “Adults aren’t willing to play without a purpose. They just want a little direction.”
For the most part, individual projects are small. They range from mandalas (circular designs generally associated with Buddhist and Hindu practice) to cards for family members. “Intuition,” explains Boehnert, “tells the volunteers what will work best with a patient.”
Hartford Hospital’s Arts for Healing is not limited to patients in a single department. Boehnert, whose previous experience with arts in healthcare included plaster cast mask-making with survivors of domestic violence, began working with rehab patients and extended the program to include dialysis patients. It’s now available in various departments throughout the hospital. Some of the work created by Arts for Healing participants in the Art for Healing program is on display in a small gallery in the hospital.
According to Boehnert, patient response to arts initiatives like the ones advocated in “Building Bridges” has been overwhelmingly positive. As an example, she cites a heart transplant patient who was introduced to the expressive arts during his six-week stay at Hartford Hospital. Before he was discharged, he created his own little gallery in his room. A patient being treated for leukemia also created an impressive body of work, giving pieces away to cheer up fellow patients who were not having good days. Staff, too, says Boehnert, benefit from Art for Healing: “My volunteers also go home better than when they came.”
Since 1991, the Society for the Arts in Healthcare (SAH) has provided support for programs such as the ones at Hartford Hospital and the Mayo Clinic, as well as others like the Artists in Residence program at Florida’s Shands HealthCare hospitals, which offers patients a variety of bedside art making activities. Examples include Art Infusion, a multi-media program for adults on chemotherapy, creative arts for pediatric inpatients, and (like Mayo Scottsdale) an oral history program which seeks to transcribe patients’ personal stories.
“In a lot of places, funding is a struggle,” explains Curry. To help secure funding for arts in healthcare programs, the SAH provides grant opportunities, like the SAH/Johnson & Johnson Partnership to Promote Arts and Healing and SAH Consulting Grants, as well as several awards.
In April, the SAH hosted its 15th international arts in healthcare conference in Chicago, the topic of which was “Vision + Voice—Charting the Course of Arts, Health and Medicine.” The conference urged attendees to “focus (their) vision for the future.”9 Given ever-increasing interest in integrating the arts into healthcare—especially inpatient care—be it by means of the clinical practice of art therapy or by expressive, creative arts programs, the future of such programs seems bright. As Dana Gioia, chair of the NEA, says: “The arts have an extraordinary ability to enhance our lives, to help us heal, and to bring us comfort in times of great stress. We must reconnect the arts with the actual human existence that Americans lead, the journeys we take in life, which lead us through hospitals, to hospices, to the end of life.”10 TH
Roberta Newman is based in Brooklyn, N.Y.
References
- NEA News Room: Arts in Healthcare Research. Available at: http://arts.endow.gov/news/news03/AIHResearch.html. Last accessed June 16, 2006.
- Art Therapy Credentials Board, “What is Art Therapy?” Available at: www.atcb.org. Last accessed June 16, 2006.
- Nainis N, Paice J, Ratner J. Relieving symptoms in cancer: innovative use of art therapy. J Pain Symptom Manage. 2006 Feb;31(2):162-169.
- Arts in Healthcare Research. National Endowment For the Arts New Room. Available at: http://arts.endow.gov/news/news03/AIHResearch.html. Last accessed June 12, 2006.
- Foster H. Medical settings foster the creation of art. Lancet. 2001;357(9268):1627.
- Foreword. “Building Bridges.” Available at: www.harthosp.org/IntMed/manual/foreword.asp. Last accessed June 18, 2006.
- Guided Imagery. “Building Bridges.” Available at www.harthosp.org/IntMed/manual/guidedimagery.asp. Last accessed June 18, 2006.
- Creating an Art for Healing Program: Training. “Building Bridges.” Available at: www.harthosp.org/IntMed/manual/art.asp. Last accessed June 18, 2006.
- Vision +Voice—Charting the Course of Arts, Health and Medicine Society for the Arts in Healthcare 15th Annual International Conference Program. Available at: www.thesah.org/doc/FINAL%20program.pdf. Last accessed June 18, 2006.
- Society for the Arts in Healthcare Fact Sheet. Available at: www.thesah.org. Last accessed June 19, 2006.
Things You Can Do To Save Lives
In April 2005, the American Hospital Association’s magazine, Hospital and Health Networks (H&HN), published the article “25 Things You Can Do to Save Lives Now.”1 In it, experts from the Joint Commission on Accreditation of Healthcare Organizations (JCAHO), the Institute for Healthcare Improvement (IHI), the National Quality Forum (NQF), and the Centers for Medicare and Medicaid Services (CMS), commented on an action plan to advance hospitals’ patient safety activities.
Now The Hospitalist has researched hospitalists’ views on these same 25 items. Those views are presented below.
A number of these items “are already highly ensconced in the JCAHO and CMS criteria,” says Dennis Manning, MD, FACP, FACC, director of quality in the Department of Medicine and an assistant professor at the Mayo Clinic College of Medicine, Rochester, Minn. “In terms of power of the things on the list for potentially saving lives, what we sometimes look at are the things that have the potential for the most prevention.”
Brian Alverson, MD, pediatric hospitalist at Hasbro Children’s Hospital in Providence, R.I., adds his thoughts on the 25 items: “We have to hold in our minds a healthy nervousness about patients being hospitalized, in that there is an inherent danger to that phenomenon. No matter how hard we strive for perfection in patient care, to err is human.”
Shortening hospital length of stay to within a safe range, he believes, is one of the best ways to reduce those daily dangers.
Some of the 25 items pose more challenges for hospitalists than others, and the contrary is true as well. Some were judged to be of lesser concern due to guidelines or imperatives imposed on hospitals by regulatory organizations. Other items fall outside hospitalists’ accountabilities, such as incorrect labeling on X-rays or CT scans, overly long working hours, medical mishaps (such as wrong-site, wrong-person, and wrong-implant surgeries), and ventilator-associated pneumonia. A few items were those that hospitalists found challenging, but for which they had few suggestions for solutions. In some, there were obstacles standing in the way of their making headway toward conquering the menace. These included:
1. Improper Patient Identification
“Until we set up a system that improves that, such as an automated system,” says one hospitalist, “I’ll be honest with you, I think we can remind ourselves ’till we’re blue in the face and we’re still going to make mistakes.”
2. Flu Shots
“Flu shots are probably more important in the pediatrics group than in any [other] except the geriatric group,” says Dr. Alverson, who strongly believes that pediatricians should be able to administer flu shots in the inpatient setting, “because we can catch these kids with chronic lung disease—many of [whom] are admitted multiple times.”
3. Fall Prevention
This item is one of the National Patient Safety goals, and one that every institution is trying to address. In pediatrics, says Dr. Alverson, the greater problem “is getting people to raise the rails of cribs. Kids often fall out of cribs because people forget to raise the rail afterwards, or don’t raise it high enough for a particularly athletic or acrobatic toddler.”
The other items on the list of 25 are below, including a section for medication-related items and the sidebar on a venous thromboembolism (VTE) prevention program.
4. Wash Hands
Provider hand-washing has been well studied, says one hospitalist, and “the data are so depressing that no one wants to deal with it.” Another says, “We just nag the hell out of people.”
One of the hospitalists interviewed for this story read the H&HN article and responds, “We do all these things.” But a lack of self-perception regarding this issue—as well as others—is also well-documented: Physicians who are queried will say they always wash their hands when, in fact, they do so less than 50% of the time.2-5
Despite the value of hand sanitizers—whether they are available at unit entrances, along the floors, at individual rooms, or carried in tiny dispensers that can be attached to a stethoscope—some pathogens, such as the now-epidemic Clostridium difficile, are not vulnerable to the antisepsis in those mechanisms.
“C. dif is a set of spores that are less effectively cleaned by the topical hand sanitizers,” says Dr. Alverson, who is also an assistant professor of pediatrics at Brown University in Providence, R.I. “In those cases, soap and water is what you need.”
Peter Angood, MD, FRCS(C), FACS, FCCM, vice president and chief patient safety officer of JCAHO, Oakbrook, Ill., says provider hand-washing is a huge patient safety issue and, in general, a multi-factorial problem that is more complicated than it would seem on the surface.
“We can rationalize and cut [providers] all kinds of slack, but at the bottom line is human behavior and their willingness to comply or not comply,” he says. “It’s like everything else: Why do some people speed when they know the speed limit is 55?”
Addressing the solution must be multi-factorial as well, but all hospitalists can serve as role models for their colleagues and students, including remaining open to reminders from patients and families.
5. Remain on Kidney Alert
Contrast media in radiologic procedures can cause allergic reactions that lead to kidney failure. This is a particularly vexing problem for elderly patients at the end stages of renal dysfunction and patients who have vascular disease, says Dr. Manning. Although the effects are not generally fatal, the medium can be organ-damaging. “This is a hazard that’s known, and it has some mitigating strategies,” he says, “but often it can’t be entirely eliminated.”
Measures that reduce the chance of injury, say Dr. Manning, include ensuring that the contrast medium is required; confirming that the procedure is correct for the patient, with the right diagnosis, with a regulated creatinine, and well coordinated with the radiology department; “and then getting true informed consent.” But at a minimum, he emphasizes, is the importance of hydration. “There is some evidence that hydration with particular types of intravenous fluids can help reduce the incidence of the kidney revolting.” And, he says, “there are a number of things that we have to do to make sure this is standardized.”
6. Use Rapid Response Teams
Use of “[r]apid response teams [RRTs] is one of the most powerful items on the list,” says Dr. Manning, who serves on SHM’s committee on Hospital Quality and Patient Safety as well as the committee helping to design the Ideal Discharge for the Elderly Patient checklist. “Whereas every hospital has a plan for response,” he says, RRTs are “really a backup plan.”
In 2003, Dr. Manning served as faculty for an IHI program in which a collaborative aimed at reducing overall hospital mortality. The formation and application of RRTs at six hospitals in the United States and two in the United Kingdom was the most promising of the several interventions, with impact on a variety of patients whose conditions were deteriorating in non-ICU care areas.
The advantage of RRTs with children, says Daniel Rauch, MD, FAAP, director of the Pediatric Hospitalist Program at NYU Medical Center, New York City, is that it is often difficult for providers to know what may be wrong with a child who is exhibiting symptoms. “Is the kid grunting because they’re constipated, because that’s the developmental stage they’re in, they’re in pain, or are they really cramping on you?” he asks.
7. Check for Pressure Ulcers
Checking for pressure ulcers is the task of nurses and physicians, say hospitalists, and they agree that it has to be done at admission. “The patient’s entire skin needs to be checked,” says Dr. Manning, “and often it takes both the nurse and doctor to roll the patient and get a good look at their bottom or their back … especially if the patient might have come from a nursing home and has a chronic serious illness.”
Also important, he says, is to fully assess the type of decubitus skin situation or any skin problem and then to monitor the patient to prevent advancement. “Multidisciplinary rounds can help,” he says, “and collaborative communication is key.”
8. Give the Patient and Family a Voice
“We fully embrace the involvement of the patient in the process of their care,” says Dr. Angood, who is also the co-director of the Joint Commission International Center for Patient Safety, for which patient and family involvement is a priority.
Giving patients and family members a voice is a fine idea, say our hospitalists, especially with children: No one knows a patient like their parents. As the H&HN article points out, anecdotal evidence is largely responsible for the belief that patient and family involvement helps reduce the likelihood for errors, and patient and family participation on safety committees can be a boon to advancing safety as well as satisfaction. But, says Dr. Alverson, “one has to keep in mind that parents have a perspective and not the only perspective on patient safety. I think a broad group of people has to sit down to address these issues.”
In the post-surgical setting, says Dr. Rauch, hospitalists make an invaluable contribution. “If surgeons don’t even come by to listen to the parents or see the child, it’s helpful to have that co-management of someone who’s used to listening to parents, who credits the parents for knowing their kids, and who will do the appropriate thing.” Dr. Angood, who is a past president of the Society of Critical Care Medicine, believes “that that patient-physician relationship is still going to be the driver for the majority of healthcare for some time yet.”
9. Reduce Catheter-Related Bloodstream Infections
“If it’s not required, we want every foreign body out,” says Dr. Manning. “We have to ask ourselves every day whether they are still required.”
The geriatric service at the Mayo Clinic (Rochester, Minn.) developed a daily mnemonic of A-B-C-D-E where B stands for binders. This, he says, “is a way for us to remind ourselves that any therapeutic foreign objects that are tethering the patient—and many of them are catheters—are of concern. We need to push the question [Is this still required?] to ourselves and then act on it.”
Dr. Alverson says, “There are certain infections [for which] we’re starting to move away from PICC [peripherally inserted central catheters] line management, and one way to mitigate that is to be on top of when you can actually discontinue the catheter.” For example, “in pediatrics, there are emerging data that with osteomyelitis you can have a shortened course of IV antibiotics and then switch to oral antibiotics. … That can reduce by half your PICC line duration. Being savvy about this is important.”
10. Reduce Heart Attack Death Rates
“There are about eight interventions for heart attacks that have increased survival,” says Dr. Manning. “So every hospital is working with these. We are using the all-or-none criteria, meaning that there are assurances [in place] that every patient will get all of them.”
Re-engineering systems has been particularly meaningful in preventing and treating heart attacks, says Dr. Manning, who represented SHM at a meeting of the Alliance for Cardiac Care Excellence (ACE), a CMS-based coalition that includes leaders from more than 30 healthcare organizations, and is working to ensure that all hospitalized cardiac patients regularly receive care consistent with nationally accepted standards.
11. Institute Multidisciplinary Rounds
Time constraints mean rounding with 10 people will necessarily be slower, says Dr. Alverson. In academic institutions where the hospitalist has the dual responsibility of teaching, this is especially time-consuming. Although there is an increasing emphasis that providers should participate at bedside rounds, and this is “clearly better from the patient’s perspective and, I would argue, better from the educational perspective,” says Dr. Alverson, it is “fairly bad from the getting-things-done-in-a-timely-fashion perspective. So it’s tough, and to a certain degree, in a practical world you have to pick and choose.”
When a nurse representative is there to respond to the question, “ ‘Why didn’t the kid get his formula? [and says] because he didn’t like the taste,’ that’s something that we might not pick up on,” says Dr. Alverson.
At NYU Medical Center, where Dr. Rauch works, formal rounds take place at least once a week (sometimes more), depending on volume, and they informally take place twice a day, every day.
“It works pretty well,” he says. “The nurses are a critically important part of teams; everybody recognizes that, and they are included in decisions.” Physicians put out the welcome mat for nurses even in casual circumstances. “Sometimes I am discussing things with the house staff [and] a nurse will pull up a chair and become part of the conversation. It’s a part of our culture.”
Although it is unusual to get a pharmacist to round with his team, says Dr. Alverson, a nearby pharmacy school sends students to join rounds, providing what might otherwise be a missing element of education.
12. Avoid Miscommunication
A number of the hospitalists interviewed were asked what they considered to be the top two or three communication points for hospitalists. Verbal orders, clarifying with read-backs, clear handwriting, and order sets were named frequently. In academic settings, says one hospitalist, instructors should be careful to make sure that residents, interns, and medical students understand what you’re saying and why you’re saying it. Good communication with the family was also cited as crucial.
“The most challenging issue is communicating at all,” says Dr. Rauch, who is also an associate professor of pediatrics at the NYU School of Medicine. Although he was the only one to phrase it this way, it is probably not a unique view. “In a large, old, academic medical institution, there are a lot of hierarchical issues that [impede] rapidly responding to [patients’] needs.” Unfortunately, it may mean communicating up one authorization pathway and down another. “And you can see the layers of time and the game of telephone as the concerns go around,” he says. “We’ve tried to break that down so the people who are on site can speak to someone who can make a decision.”
Along with that, he says, it is important from the outset to make it very clear who makes the decision. “For example, when the patient is a child getting neurosurgery because they have a seizure disorder and they also are developmentally delayed and they have medical issues, you now have at least three services involved with managing the child,” says Dr. Rauch. When three people are making decisions, he points out, no decision gets made. “You really have to decide when that child comes in who is going to call the shots for what issue. It’s usually the hospitalist who brings it up, and when it works, it works well.”
13. Empower Nurses and Other Clinicians
Nursing staff should have the power to halt unsafe practices. To Tahl Humes, MD, hospitalist at Exempla St. Joseph’s Hospital in Denver, halting unsafe practices depends, once again, on good lines of communication, and recognizing that patient care is a joint responsibility. For example, she says, “instead of just going to see the patient, writing the note, writing the order, and putting the chart away,” the hospitalists “talk with nurses daily and tell them what they’re planning to do,” so there is more opportunity to catch what might be unsafe practices.
14. Reduce Wound Infections
Although reducing wound infections is something in which their surgical colleagues take the lead, says Dr. Manning, “in our perioperative consultation care, we often work with surgery and anesthesiology in the pre-op evaluations of the patients. So in the surgical care improvement projects, we are often partners.”
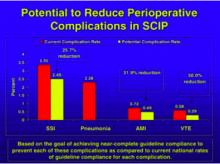
Hospitalists are also frequently members on quality committees that help to brainstorm solutions to serious problems. One such project is the Surgical Care Improvement Program (SCIP), spearheaded by David Hunt, MD, with the Office of Clinical Standards and Quality, CMS. SCIP is an effort to transform the prevention of postoperative complications. Its goal is to reduce surgical complications by 25% in the United States by the year 2010 in four target areas: surgical site infections, and cardiac, respiratory, and venous thromboembolic complications. (See Figures 1 and 2, p. 33.)
This includes those patients who are already on beta-blockers. “From the hospitalist’s standpoint,” says Dr. Manning, “we have a real role in … [ensuring] that their beta blockade is maintained.”
Dr. Humes says that at her institution, a wound care nurse can have that responsibility. If a provider is concerned about any patient in this regard, he or she can order that the patient be seen by a wound care nurse and, depending on what’s needed, by a physical therapist.
Now we move on to address those issues that are medication-related:
15. Know Risky Meds
Pediatric hospitalists are involved with postoperative patients at Dr. Rauch’s institution. All patients’ orders are double-checked, he says, and computer order entry also helps providers calculate pediatric dosage norms or dosages calculated by weight.
The hospitalist has the opportunity to be involved in the pharmacy’s selection of drugs for the formulary, says Erin Stucky, MD, pediatric hospitalist at University of California, San Diego, and to help decide the drug choices within a certain class and limit the numbers of things that are used most frequently that are visually different in appearance. “And although that’s the pharmacist’s purview,” she says, “the hospitalist has a vested interest in being on the Pharmacy and Therapeutics Committee to review and restate to pharmacists what they’re using based on clinical need and to find a way for that drug to be safely stored in pharmacy if, indeed, there are a couple of drugs from one class that are truly useful.”
A drug’s generic name, brand name, dose strength, frequency of administration, place of use, indications, and contraindications are all important factors to determine the potential risks of drugs. But “you can’t say a list of risky medications at one institution is the same as it should be elsewhere,” says Dr. Stucky. Risky medications will depend on the setting in which the physician works. Hospitalists need to think logically about the drugs that are the most used or are new, including any new drug that has a different method by which it is administered or a different interaction capability with standard drugs.
“If there’s a new antibiotic that’s known to be processed through the liver and you have multiple patients with heart failure medications who have a medication basis that could be at conflict with that new drug, that’s a potentially risky medication,” she points out. “It may be easier in some ways for the pharmacist to be the rate-limiting factor for how they’re dispensed and for which patients they recheck [against] that incompatibility list.” But in large part, the avoidance of those risky-medication errors must be a commitment of the pharmacist and a bedside nurse.
16. Beware of Sound-Alike and Look-Alike Drugs
Dr. Stucky believes a majority of physicians don’t know the color or size of the pills they’re prescribing. “I would challenge all hospitalists to take every opportunity at the bedside,” she says, “to watch the process happening and know what those drugs and pills need to look like.”
Another opportunity is to educate family members “to remind them that [the patient is] going to be getting these medicines, these are the names of the medicines, and please ask the nurse about these medicines when you get them,” she says. If the hospitalist gives a new drug to the patient, the family can be another safeguard.
Dr. Stucky points out that you can tell the patient and family, “I’m going to tell the nurse that you’re going to be asking about this because … you are the best guide to help us make sure that these medicines are administered safely.” She also emphasizes that assigning this responsibility to the patient is important “because when people leave the hospital, we suddenly expect them to know how to take 18 pills.”
If, on a given unit, you have to handle cases with multiple diagnoses, says Dr. Stucky, it may be difficult to physically isolate the look-alike drugs. “At our institution we found that we actually had to pull the machines out,” she says, referring to the PIXUS units. “You can’t have them on the same wall even in different locations. You have to choose one or the other [similar looking pills].”
The sound-alike drugs are most ripe for errors with verbal orders. “Hospitalists can set a precedent in their institutions that any verbal orders should have the reason for that order given,” she explains. If you order clonazepam, after you finish giving the order verbally to the nurse, you should state, “This is for seizures.”
“When the nurse is writing it down, she may or may not be the one to know that that drug name is indeed in that drug class, but the pharmacist will know,” explains Dr. Stucky.
17. Reconcile Medications
“It is important for people to do verbal sign-out, certainly among attendings,” says Dr. Alverson, “to explain [in better depth] what’s going on with the patient and to maintain those avenues of communication in case something goes wrong. Hospitals get in trouble when physicians aren’t able to communicate or speak with each other readily.”
The biggest challenge for the pediatric hospitalists at NYU Hospital, says Dr. Rauch, is assessing the most up-to-date list of medications. “For instance,” he says, “we had a child yesterday as part of post-op care. I hadn’t met them pre-op. The father said, ‘I think my daughter’s on an experimental protocol with this additional medication.’ It wasn’t something we were used to so we called Mom: Can you bring in the protocol? She said, ‘Oh, she hasn’t been on that drug in a long time.’”
In fact, whether the patient is a child or adult, the majority of cases assigned to hospitalists are unplanned admissions and this is something with which all hospitalists struggle. But regarding transferring patients from unit to unit, says Dr. Stucky, “this is a whole different ballgame. That’s where we have a huge opportunity to make an impact.”
She suggests that matching medications to patients can be ameliorated by computer-based systems in which at each new place the hospitalist can fill in a printout regarding whether they’re continuing a drug order, changing it, or discontinuing it, and this system also works effectively on discharge. “In a perfect world,” says Dr. Stucky, … “the hospitalist would be the implementer of this kind of medication reconciliation in their institution.”
18. Avoid Unacceptable Drug Abbreviations
Some medications have abbreviations that can be misinterpreted. The classic ones, say several hospitalists, are magnesium and morphine. Others pertain to miswritten units of administration.
Read-backs on verbal medication orders was one of the elements most cited by our hospitalists as priority communication practices. Eliminating confusing abbreviations is one of JCAHO’s National Patient Safety Goals and “hospitals are aggressively rolling out ways to remind physicians not to use them,” says Dr. Alverson.
At Dr. Manning’s institution they use the Safest in America criteria, a collaboration of 10 Twin Cities and the hospital systems in Rochester, Minn., as well as the Institute for Clinical Systems Improvement. At Mayo, they call it “Write It Right.” An accentuated campaign to reduce ambiguities in medication communications, he says, has resulted in “profound improvement” in standardizing medication prescribing and following the read-back rules.
Dr. Stucky suggests that hospitalists take on mini-projects where they review the past six months of order-writing errors in their institutions, noticing any trends and, particularly, any unit-specific trends (such as the misunderstanding of the abbreviation cc). If you notice the errors are unit-specific, you can also analyze whether they are treatment-specific. In that way, “order sets can be pre-typed and all the providers have to do is fill in the numbers,” she says, adding that hospitalists can perform these analyses outside their own patient area.
19. Improper Drug Labeling, Packaging, and Storage
Drug names, labels, and packaging contribute significantly to medication errors. The risk for errors is determined by both the product and the environment in which it is used. Most hospitalists say they are continually developing new protocols and checking information multiple times. Sometimes, small changes go a long way. “Our patient safety officer has a favorite phrase: ‘How can I facilitate you to do something different next Tuesday?’ Within your own hospital, that means look at your system and pick something you know you can change,” says Dr. Stucky. “You can’t buy IT tomorrow; you can’t do physician order entries [because] your computer system doesn’t allow it—but what can you do?”
Conclusion
Dr. Angood encourages hospitalists to continue learning how to interact with other disciplines that are also evolving into hospital-based practices and to learn how to manage the specific details-of-change topics such as this list of 25—not just to gloss over them, but to understand them, and to encourage patient involvement and nurture the physician-patient relationship to help change the culture within health care.
“We can pick these kinds of topics and can dissect them all down, but each time, in the end, it is a matter of people and their behaviors as a culture inside a system,” he says. “The system can be changed a little bit, but still it is ultimately about the culture of people.” TH
Andrea Sattinger writes regularly for The Hospitalist.
References
- Runy LA. 25 things you can do to save lives now. Hosp Health Netw. 2005 Apr;79(4):27-28.
- Meengs MR, Giles BK, Chisholm CD, et al. Hand washing frequency in an emergency department. Ann Emerg Med. 1994 Jun;23(6):1307-1312.
- McGuckina M, Watermana R, Storrb J, et al. Evaluation of a patient-empowering hand hygiene programme in the UK. J Hosp Infect. 2001 Jul;48(3):222-227.
- Whitby M, McLaws ML, Ross MW. Why healthcare workers don't wash their hands: a behavioral explanation. Infect Control Hosp Epidemiol. 2006 May;27(5):484-492.
- Lipsett PA, Swoboda SM. Handwashing compliance depends on professional status. Surg Infect. 2001 Fall;2(3):241-245.
In April 2005, the American Hospital Association’s magazine, Hospital and Health Networks (H&HN), published the article “25 Things You Can Do to Save Lives Now.”1 In it, experts from the Joint Commission on Accreditation of Healthcare Organizations (JCAHO), the Institute for Healthcare Improvement (IHI), the National Quality Forum (NQF), and the Centers for Medicare and Medicaid Services (CMS), commented on an action plan to advance hospitals’ patient safety activities.
Now The Hospitalist has researched hospitalists’ views on these same 25 items. Those views are presented below.
A number of these items “are already highly ensconced in the JCAHO and CMS criteria,” says Dennis Manning, MD, FACP, FACC, director of quality in the Department of Medicine and an assistant professor at the Mayo Clinic College of Medicine, Rochester, Minn. “In terms of power of the things on the list for potentially saving lives, what we sometimes look at are the things that have the potential for the most prevention.”
Brian Alverson, MD, pediatric hospitalist at Hasbro Children’s Hospital in Providence, R.I., adds his thoughts on the 25 items: “We have to hold in our minds a healthy nervousness about patients being hospitalized, in that there is an inherent danger to that phenomenon. No matter how hard we strive for perfection in patient care, to err is human.”
Shortening hospital length of stay to within a safe range, he believes, is one of the best ways to reduce those daily dangers.
Some of the 25 items pose more challenges for hospitalists than others, and the contrary is true as well. Some were judged to be of lesser concern due to guidelines or imperatives imposed on hospitals by regulatory organizations. Other items fall outside hospitalists’ accountabilities, such as incorrect labeling on X-rays or CT scans, overly long working hours, medical mishaps (such as wrong-site, wrong-person, and wrong-implant surgeries), and ventilator-associated pneumonia. A few items were those that hospitalists found challenging, but for which they had few suggestions for solutions. In some, there were obstacles standing in the way of their making headway toward conquering the menace. These included:
1. Improper Patient Identification
“Until we set up a system that improves that, such as an automated system,” says one hospitalist, “I’ll be honest with you, I think we can remind ourselves ’till we’re blue in the face and we’re still going to make mistakes.”
2. Flu Shots
“Flu shots are probably more important in the pediatrics group than in any [other] except the geriatric group,” says Dr. Alverson, who strongly believes that pediatricians should be able to administer flu shots in the inpatient setting, “because we can catch these kids with chronic lung disease—many of [whom] are admitted multiple times.”
3. Fall Prevention
This item is one of the National Patient Safety goals, and one that every institution is trying to address. In pediatrics, says Dr. Alverson, the greater problem “is getting people to raise the rails of cribs. Kids often fall out of cribs because people forget to raise the rail afterwards, or don’t raise it high enough for a particularly athletic or acrobatic toddler.”
The other items on the list of 25 are below, including a section for medication-related items and the sidebar on a venous thromboembolism (VTE) prevention program.
4. Wash Hands
Provider hand-washing has been well studied, says one hospitalist, and “the data are so depressing that no one wants to deal with it.” Another says, “We just nag the hell out of people.”
One of the hospitalists interviewed for this story read the H&HN article and responds, “We do all these things.” But a lack of self-perception regarding this issue—as well as others—is also well-documented: Physicians who are queried will say they always wash their hands when, in fact, they do so less than 50% of the time.2-5
Despite the value of hand sanitizers—whether they are available at unit entrances, along the floors, at individual rooms, or carried in tiny dispensers that can be attached to a stethoscope—some pathogens, such as the now-epidemic Clostridium difficile, are not vulnerable to the antisepsis in those mechanisms.
“C. dif is a set of spores that are less effectively cleaned by the topical hand sanitizers,” says Dr. Alverson, who is also an assistant professor of pediatrics at Brown University in Providence, R.I. “In those cases, soap and water is what you need.”
Peter Angood, MD, FRCS(C), FACS, FCCM, vice president and chief patient safety officer of JCAHO, Oakbrook, Ill., says provider hand-washing is a huge patient safety issue and, in general, a multi-factorial problem that is more complicated than it would seem on the surface.
“We can rationalize and cut [providers] all kinds of slack, but at the bottom line is human behavior and their willingness to comply or not comply,” he says. “It’s like everything else: Why do some people speed when they know the speed limit is 55?”
Addressing the solution must be multi-factorial as well, but all hospitalists can serve as role models for their colleagues and students, including remaining open to reminders from patients and families.
5. Remain on Kidney Alert
Contrast media in radiologic procedures can cause allergic reactions that lead to kidney failure. This is a particularly vexing problem for elderly patients at the end stages of renal dysfunction and patients who have vascular disease, says Dr. Manning. Although the effects are not generally fatal, the medium can be organ-damaging. “This is a hazard that’s known, and it has some mitigating strategies,” he says, “but often it can’t be entirely eliminated.”
Measures that reduce the chance of injury, say Dr. Manning, include ensuring that the contrast medium is required; confirming that the procedure is correct for the patient, with the right diagnosis, with a regulated creatinine, and well coordinated with the radiology department; “and then getting true informed consent.” But at a minimum, he emphasizes, is the importance of hydration. “There is some evidence that hydration with particular types of intravenous fluids can help reduce the incidence of the kidney revolting.” And, he says, “there are a number of things that we have to do to make sure this is standardized.”
6. Use Rapid Response Teams
Use of “[r]apid response teams [RRTs] is one of the most powerful items on the list,” says Dr. Manning, who serves on SHM’s committee on Hospital Quality and Patient Safety as well as the committee helping to design the Ideal Discharge for the Elderly Patient checklist. “Whereas every hospital has a plan for response,” he says, RRTs are “really a backup plan.”
In 2003, Dr. Manning served as faculty for an IHI program in which a collaborative aimed at reducing overall hospital mortality. The formation and application of RRTs at six hospitals in the United States and two in the United Kingdom was the most promising of the several interventions, with impact on a variety of patients whose conditions were deteriorating in non-ICU care areas.
The advantage of RRTs with children, says Daniel Rauch, MD, FAAP, director of the Pediatric Hospitalist Program at NYU Medical Center, New York City, is that it is often difficult for providers to know what may be wrong with a child who is exhibiting symptoms. “Is the kid grunting because they’re constipated, because that’s the developmental stage they’re in, they’re in pain, or are they really cramping on you?” he asks.
7. Check for Pressure Ulcers
Checking for pressure ulcers is the task of nurses and physicians, say hospitalists, and they agree that it has to be done at admission. “The patient’s entire skin needs to be checked,” says Dr. Manning, “and often it takes both the nurse and doctor to roll the patient and get a good look at their bottom or their back … especially if the patient might have come from a nursing home and has a chronic serious illness.”
Also important, he says, is to fully assess the type of decubitus skin situation or any skin problem and then to monitor the patient to prevent advancement. “Multidisciplinary rounds can help,” he says, “and collaborative communication is key.”
8. Give the Patient and Family a Voice
“We fully embrace the involvement of the patient in the process of their care,” says Dr. Angood, who is also the co-director of the Joint Commission International Center for Patient Safety, for which patient and family involvement is a priority.
Giving patients and family members a voice is a fine idea, say our hospitalists, especially with children: No one knows a patient like their parents. As the H&HN article points out, anecdotal evidence is largely responsible for the belief that patient and family involvement helps reduce the likelihood for errors, and patient and family participation on safety committees can be a boon to advancing safety as well as satisfaction. But, says Dr. Alverson, “one has to keep in mind that parents have a perspective and not the only perspective on patient safety. I think a broad group of people has to sit down to address these issues.”
In the post-surgical setting, says Dr. Rauch, hospitalists make an invaluable contribution. “If surgeons don’t even come by to listen to the parents or see the child, it’s helpful to have that co-management of someone who’s used to listening to parents, who credits the parents for knowing their kids, and who will do the appropriate thing.” Dr. Angood, who is a past president of the Society of Critical Care Medicine, believes “that that patient-physician relationship is still going to be the driver for the majority of healthcare for some time yet.”
9. Reduce Catheter-Related Bloodstream Infections
“If it’s not required, we want every foreign body out,” says Dr. Manning. “We have to ask ourselves every day whether they are still required.”
The geriatric service at the Mayo Clinic (Rochester, Minn.) developed a daily mnemonic of A-B-C-D-E where B stands for binders. This, he says, “is a way for us to remind ourselves that any therapeutic foreign objects that are tethering the patient—and many of them are catheters—are of concern. We need to push the question [Is this still required?] to ourselves and then act on it.”
Dr. Alverson says, “There are certain infections [for which] we’re starting to move away from PICC [peripherally inserted central catheters] line management, and one way to mitigate that is to be on top of when you can actually discontinue the catheter.” For example, “in pediatrics, there are emerging data that with osteomyelitis you can have a shortened course of IV antibiotics and then switch to oral antibiotics. … That can reduce by half your PICC line duration. Being savvy about this is important.”
10. Reduce Heart Attack Death Rates
“There are about eight interventions for heart attacks that have increased survival,” says Dr. Manning. “So every hospital is working with these. We are using the all-or-none criteria, meaning that there are assurances [in place] that every patient will get all of them.”
Re-engineering systems has been particularly meaningful in preventing and treating heart attacks, says Dr. Manning, who represented SHM at a meeting of the Alliance for Cardiac Care Excellence (ACE), a CMS-based coalition that includes leaders from more than 30 healthcare organizations, and is working to ensure that all hospitalized cardiac patients regularly receive care consistent with nationally accepted standards.
11. Institute Multidisciplinary Rounds
Time constraints mean rounding with 10 people will necessarily be slower, says Dr. Alverson. In academic institutions where the hospitalist has the dual responsibility of teaching, this is especially time-consuming. Although there is an increasing emphasis that providers should participate at bedside rounds, and this is “clearly better from the patient’s perspective and, I would argue, better from the educational perspective,” says Dr. Alverson, it is “fairly bad from the getting-things-done-in-a-timely-fashion perspective. So it’s tough, and to a certain degree, in a practical world you have to pick and choose.”
When a nurse representative is there to respond to the question, “ ‘Why didn’t the kid get his formula? [and says] because he didn’t like the taste,’ that’s something that we might not pick up on,” says Dr. Alverson.
At NYU Medical Center, where Dr. Rauch works, formal rounds take place at least once a week (sometimes more), depending on volume, and they informally take place twice a day, every day.
“It works pretty well,” he says. “The nurses are a critically important part of teams; everybody recognizes that, and they are included in decisions.” Physicians put out the welcome mat for nurses even in casual circumstances. “Sometimes I am discussing things with the house staff [and] a nurse will pull up a chair and become part of the conversation. It’s a part of our culture.”
Although it is unusual to get a pharmacist to round with his team, says Dr. Alverson, a nearby pharmacy school sends students to join rounds, providing what might otherwise be a missing element of education.
12. Avoid Miscommunication
A number of the hospitalists interviewed were asked what they considered to be the top two or three communication points for hospitalists. Verbal orders, clarifying with read-backs, clear handwriting, and order sets were named frequently. In academic settings, says one hospitalist, instructors should be careful to make sure that residents, interns, and medical students understand what you’re saying and why you’re saying it. Good communication with the family was also cited as crucial.
“The most challenging issue is communicating at all,” says Dr. Rauch, who is also an associate professor of pediatrics at the NYU School of Medicine. Although he was the only one to phrase it this way, it is probably not a unique view. “In a large, old, academic medical institution, there are a lot of hierarchical issues that [impede] rapidly responding to [patients’] needs.” Unfortunately, it may mean communicating up one authorization pathway and down another. “And you can see the layers of time and the game of telephone as the concerns go around,” he says. “We’ve tried to break that down so the people who are on site can speak to someone who can make a decision.”
Along with that, he says, it is important from the outset to make it very clear who makes the decision. “For example, when the patient is a child getting neurosurgery because they have a seizure disorder and they also are developmentally delayed and they have medical issues, you now have at least three services involved with managing the child,” says Dr. Rauch. When three people are making decisions, he points out, no decision gets made. “You really have to decide when that child comes in who is going to call the shots for what issue. It’s usually the hospitalist who brings it up, and when it works, it works well.”
13. Empower Nurses and Other Clinicians
Nursing staff should have the power to halt unsafe practices. To Tahl Humes, MD, hospitalist at Exempla St. Joseph’s Hospital in Denver, halting unsafe practices depends, once again, on good lines of communication, and recognizing that patient care is a joint responsibility. For example, she says, “instead of just going to see the patient, writing the note, writing the order, and putting the chart away,” the hospitalists “talk with nurses daily and tell them what they’re planning to do,” so there is more opportunity to catch what might be unsafe practices.
14. Reduce Wound Infections
Although reducing wound infections is something in which their surgical colleagues take the lead, says Dr. Manning, “in our perioperative consultation care, we often work with surgery and anesthesiology in the pre-op evaluations of the patients. So in the surgical care improvement projects, we are often partners.”
Hospitalists are also frequently members on quality committees that help to brainstorm solutions to serious problems. One such project is the Surgical Care Improvement Program (SCIP), spearheaded by David Hunt, MD, with the Office of Clinical Standards and Quality, CMS. SCIP is an effort to transform the prevention of postoperative complications. Its goal is to reduce surgical complications by 25% in the United States by the year 2010 in four target areas: surgical site infections, and cardiac, respiratory, and venous thromboembolic complications. (See Figures 1 and 2, p. 33.)
This includes those patients who are already on beta-blockers. “From the hospitalist’s standpoint,” says Dr. Manning, “we have a real role in … [ensuring] that their beta blockade is maintained.”
Dr. Humes says that at her institution, a wound care nurse can have that responsibility. If a provider is concerned about any patient in this regard, he or she can order that the patient be seen by a wound care nurse and, depending on what’s needed, by a physical therapist.
Now we move on to address those issues that are medication-related:
15. Know Risky Meds
Pediatric hospitalists are involved with postoperative patients at Dr. Rauch’s institution. All patients’ orders are double-checked, he says, and computer order entry also helps providers calculate pediatric dosage norms or dosages calculated by weight.
The hospitalist has the opportunity to be involved in the pharmacy’s selection of drugs for the formulary, says Erin Stucky, MD, pediatric hospitalist at University of California, San Diego, and to help decide the drug choices within a certain class and limit the numbers of things that are used most frequently that are visually different in appearance. “And although that’s the pharmacist’s purview,” she says, “the hospitalist has a vested interest in being on the Pharmacy and Therapeutics Committee to review and restate to pharmacists what they’re using based on clinical need and to find a way for that drug to be safely stored in pharmacy if, indeed, there are a couple of drugs from one class that are truly useful.”
A drug’s generic name, brand name, dose strength, frequency of administration, place of use, indications, and contraindications are all important factors to determine the potential risks of drugs. But “you can’t say a list of risky medications at one institution is the same as it should be elsewhere,” says Dr. Stucky. Risky medications will depend on the setting in which the physician works. Hospitalists need to think logically about the drugs that are the most used or are new, including any new drug that has a different method by which it is administered or a different interaction capability with standard drugs.
“If there’s a new antibiotic that’s known to be processed through the liver and you have multiple patients with heart failure medications who have a medication basis that could be at conflict with that new drug, that’s a potentially risky medication,” she points out. “It may be easier in some ways for the pharmacist to be the rate-limiting factor for how they’re dispensed and for which patients they recheck [against] that incompatibility list.” But in large part, the avoidance of those risky-medication errors must be a commitment of the pharmacist and a bedside nurse.
16. Beware of Sound-Alike and Look-Alike Drugs
Dr. Stucky believes a majority of physicians don’t know the color or size of the pills they’re prescribing. “I would challenge all hospitalists to take every opportunity at the bedside,” she says, “to watch the process happening and know what those drugs and pills need to look like.”
Another opportunity is to educate family members “to remind them that [the patient is] going to be getting these medicines, these are the names of the medicines, and please ask the nurse about these medicines when you get them,” she says. If the hospitalist gives a new drug to the patient, the family can be another safeguard.
Dr. Stucky points out that you can tell the patient and family, “I’m going to tell the nurse that you’re going to be asking about this because … you are the best guide to help us make sure that these medicines are administered safely.” She also emphasizes that assigning this responsibility to the patient is important “because when people leave the hospital, we suddenly expect them to know how to take 18 pills.”
If, on a given unit, you have to handle cases with multiple diagnoses, says Dr. Stucky, it may be difficult to physically isolate the look-alike drugs. “At our institution we found that we actually had to pull the machines out,” she says, referring to the PIXUS units. “You can’t have them on the same wall even in different locations. You have to choose one or the other [similar looking pills].”
The sound-alike drugs are most ripe for errors with verbal orders. “Hospitalists can set a precedent in their institutions that any verbal orders should have the reason for that order given,” she explains. If you order clonazepam, after you finish giving the order verbally to the nurse, you should state, “This is for seizures.”
“When the nurse is writing it down, she may or may not be the one to know that that drug name is indeed in that drug class, but the pharmacist will know,” explains Dr. Stucky.
17. Reconcile Medications
“It is important for people to do verbal sign-out, certainly among attendings,” says Dr. Alverson, “to explain [in better depth] what’s going on with the patient and to maintain those avenues of communication in case something goes wrong. Hospitals get in trouble when physicians aren’t able to communicate or speak with each other readily.”
The biggest challenge for the pediatric hospitalists at NYU Hospital, says Dr. Rauch, is assessing the most up-to-date list of medications. “For instance,” he says, “we had a child yesterday as part of post-op care. I hadn’t met them pre-op. The father said, ‘I think my daughter’s on an experimental protocol with this additional medication.’ It wasn’t something we were used to so we called Mom: Can you bring in the protocol? She said, ‘Oh, she hasn’t been on that drug in a long time.’”
In fact, whether the patient is a child or adult, the majority of cases assigned to hospitalists are unplanned admissions and this is something with which all hospitalists struggle. But regarding transferring patients from unit to unit, says Dr. Stucky, “this is a whole different ballgame. That’s where we have a huge opportunity to make an impact.”
She suggests that matching medications to patients can be ameliorated by computer-based systems in which at each new place the hospitalist can fill in a printout regarding whether they’re continuing a drug order, changing it, or discontinuing it, and this system also works effectively on discharge. “In a perfect world,” says Dr. Stucky, … “the hospitalist would be the implementer of this kind of medication reconciliation in their institution.”
18. Avoid Unacceptable Drug Abbreviations
Some medications have abbreviations that can be misinterpreted. The classic ones, say several hospitalists, are magnesium and morphine. Others pertain to miswritten units of administration.
Read-backs on verbal medication orders was one of the elements most cited by our hospitalists as priority communication practices. Eliminating confusing abbreviations is one of JCAHO’s National Patient Safety Goals and “hospitals are aggressively rolling out ways to remind physicians not to use them,” says Dr. Alverson.
At Dr. Manning’s institution they use the Safest in America criteria, a collaboration of 10 Twin Cities and the hospital systems in Rochester, Minn., as well as the Institute for Clinical Systems Improvement. At Mayo, they call it “Write It Right.” An accentuated campaign to reduce ambiguities in medication communications, he says, has resulted in “profound improvement” in standardizing medication prescribing and following the read-back rules.
Dr. Stucky suggests that hospitalists take on mini-projects where they review the past six months of order-writing errors in their institutions, noticing any trends and, particularly, any unit-specific trends (such as the misunderstanding of the abbreviation cc). If you notice the errors are unit-specific, you can also analyze whether they are treatment-specific. In that way, “order sets can be pre-typed and all the providers have to do is fill in the numbers,” she says, adding that hospitalists can perform these analyses outside their own patient area.
19. Improper Drug Labeling, Packaging, and Storage
Drug names, labels, and packaging contribute significantly to medication errors. The risk for errors is determined by both the product and the environment in which it is used. Most hospitalists say they are continually developing new protocols and checking information multiple times. Sometimes, small changes go a long way. “Our patient safety officer has a favorite phrase: ‘How can I facilitate you to do something different next Tuesday?’ Within your own hospital, that means look at your system and pick something you know you can change,” says Dr. Stucky. “You can’t buy IT tomorrow; you can’t do physician order entries [because] your computer system doesn’t allow it—but what can you do?”
Conclusion
Dr. Angood encourages hospitalists to continue learning how to interact with other disciplines that are also evolving into hospital-based practices and to learn how to manage the specific details-of-change topics such as this list of 25—not just to gloss over them, but to understand them, and to encourage patient involvement and nurture the physician-patient relationship to help change the culture within health care.
“We can pick these kinds of topics and can dissect them all down, but each time, in the end, it is a matter of people and their behaviors as a culture inside a system,” he says. “The system can be changed a little bit, but still it is ultimately about the culture of people.” TH
Andrea Sattinger writes regularly for The Hospitalist.
References
- Runy LA. 25 things you can do to save lives now. Hosp Health Netw. 2005 Apr;79(4):27-28.
- Meengs MR, Giles BK, Chisholm CD, et al. Hand washing frequency in an emergency department. Ann Emerg Med. 1994 Jun;23(6):1307-1312.
- McGuckina M, Watermana R, Storrb J, et al. Evaluation of a patient-empowering hand hygiene programme in the UK. J Hosp Infect. 2001 Jul;48(3):222-227.
- Whitby M, McLaws ML, Ross MW. Why healthcare workers don't wash their hands: a behavioral explanation. Infect Control Hosp Epidemiol. 2006 May;27(5):484-492.
- Lipsett PA, Swoboda SM. Handwashing compliance depends on professional status. Surg Infect. 2001 Fall;2(3):241-245.
In April 2005, the American Hospital Association’s magazine, Hospital and Health Networks (H&HN), published the article “25 Things You Can Do to Save Lives Now.”1 In it, experts from the Joint Commission on Accreditation of Healthcare Organizations (JCAHO), the Institute for Healthcare Improvement (IHI), the National Quality Forum (NQF), and the Centers for Medicare and Medicaid Services (CMS), commented on an action plan to advance hospitals’ patient safety activities.
Now The Hospitalist has researched hospitalists’ views on these same 25 items. Those views are presented below.
A number of these items “are already highly ensconced in the JCAHO and CMS criteria,” says Dennis Manning, MD, FACP, FACC, director of quality in the Department of Medicine and an assistant professor at the Mayo Clinic College of Medicine, Rochester, Minn. “In terms of power of the things on the list for potentially saving lives, what we sometimes look at are the things that have the potential for the most prevention.”
Brian Alverson, MD, pediatric hospitalist at Hasbro Children’s Hospital in Providence, R.I., adds his thoughts on the 25 items: “We have to hold in our minds a healthy nervousness about patients being hospitalized, in that there is an inherent danger to that phenomenon. No matter how hard we strive for perfection in patient care, to err is human.”
Shortening hospital length of stay to within a safe range, he believes, is one of the best ways to reduce those daily dangers.
Some of the 25 items pose more challenges for hospitalists than others, and the contrary is true as well. Some were judged to be of lesser concern due to guidelines or imperatives imposed on hospitals by regulatory organizations. Other items fall outside hospitalists’ accountabilities, such as incorrect labeling on X-rays or CT scans, overly long working hours, medical mishaps (such as wrong-site, wrong-person, and wrong-implant surgeries), and ventilator-associated pneumonia. A few items were those that hospitalists found challenging, but for which they had few suggestions for solutions. In some, there were obstacles standing in the way of their making headway toward conquering the menace. These included:
1. Improper Patient Identification
“Until we set up a system that improves that, such as an automated system,” says one hospitalist, “I’ll be honest with you, I think we can remind ourselves ’till we’re blue in the face and we’re still going to make mistakes.”
2. Flu Shots
“Flu shots are probably more important in the pediatrics group than in any [other] except the geriatric group,” says Dr. Alverson, who strongly believes that pediatricians should be able to administer flu shots in the inpatient setting, “because we can catch these kids with chronic lung disease—many of [whom] are admitted multiple times.”
3. Fall Prevention
This item is one of the National Patient Safety goals, and one that every institution is trying to address. In pediatrics, says Dr. Alverson, the greater problem “is getting people to raise the rails of cribs. Kids often fall out of cribs because people forget to raise the rail afterwards, or don’t raise it high enough for a particularly athletic or acrobatic toddler.”
The other items on the list of 25 are below, including a section for medication-related items and the sidebar on a venous thromboembolism (VTE) prevention program.
4. Wash Hands
Provider hand-washing has been well studied, says one hospitalist, and “the data are so depressing that no one wants to deal with it.” Another says, “We just nag the hell out of people.”
One of the hospitalists interviewed for this story read the H&HN article and responds, “We do all these things.” But a lack of self-perception regarding this issue—as well as others—is also well-documented: Physicians who are queried will say they always wash their hands when, in fact, they do so less than 50% of the time.2-5
Despite the value of hand sanitizers—whether they are available at unit entrances, along the floors, at individual rooms, or carried in tiny dispensers that can be attached to a stethoscope—some pathogens, such as the now-epidemic Clostridium difficile, are not vulnerable to the antisepsis in those mechanisms.
“C. dif is a set of spores that are less effectively cleaned by the topical hand sanitizers,” says Dr. Alverson, who is also an assistant professor of pediatrics at Brown University in Providence, R.I. “In those cases, soap and water is what you need.”
Peter Angood, MD, FRCS(C), FACS, FCCM, vice president and chief patient safety officer of JCAHO, Oakbrook, Ill., says provider hand-washing is a huge patient safety issue and, in general, a multi-factorial problem that is more complicated than it would seem on the surface.
“We can rationalize and cut [providers] all kinds of slack, but at the bottom line is human behavior and their willingness to comply or not comply,” he says. “It’s like everything else: Why do some people speed when they know the speed limit is 55?”
Addressing the solution must be multi-factorial as well, but all hospitalists can serve as role models for their colleagues and students, including remaining open to reminders from patients and families.
5. Remain on Kidney Alert
Contrast media in radiologic procedures can cause allergic reactions that lead to kidney failure. This is a particularly vexing problem for elderly patients at the end stages of renal dysfunction and patients who have vascular disease, says Dr. Manning. Although the effects are not generally fatal, the medium can be organ-damaging. “This is a hazard that’s known, and it has some mitigating strategies,” he says, “but often it can’t be entirely eliminated.”
Measures that reduce the chance of injury, say Dr. Manning, include ensuring that the contrast medium is required; confirming that the procedure is correct for the patient, with the right diagnosis, with a regulated creatinine, and well coordinated with the radiology department; “and then getting true informed consent.” But at a minimum, he emphasizes, is the importance of hydration. “There is some evidence that hydration with particular types of intravenous fluids can help reduce the incidence of the kidney revolting.” And, he says, “there are a number of things that we have to do to make sure this is standardized.”
6. Use Rapid Response Teams
Use of “[r]apid response teams [RRTs] is one of the most powerful items on the list,” says Dr. Manning, who serves on SHM’s committee on Hospital Quality and Patient Safety as well as the committee helping to design the Ideal Discharge for the Elderly Patient checklist. “Whereas every hospital has a plan for response,” he says, RRTs are “really a backup plan.”
In 2003, Dr. Manning served as faculty for an IHI program in which a collaborative aimed at reducing overall hospital mortality. The formation and application of RRTs at six hospitals in the United States and two in the United Kingdom was the most promising of the several interventions, with impact on a variety of patients whose conditions were deteriorating in non-ICU care areas.
The advantage of RRTs with children, says Daniel Rauch, MD, FAAP, director of the Pediatric Hospitalist Program at NYU Medical Center, New York City, is that it is often difficult for providers to know what may be wrong with a child who is exhibiting symptoms. “Is the kid grunting because they’re constipated, because that’s the developmental stage they’re in, they’re in pain, or are they really cramping on you?” he asks.
7. Check for Pressure Ulcers
Checking for pressure ulcers is the task of nurses and physicians, say hospitalists, and they agree that it has to be done at admission. “The patient’s entire skin needs to be checked,” says Dr. Manning, “and often it takes both the nurse and doctor to roll the patient and get a good look at their bottom or their back … especially if the patient might have come from a nursing home and has a chronic serious illness.”
Also important, he says, is to fully assess the type of decubitus skin situation or any skin problem and then to monitor the patient to prevent advancement. “Multidisciplinary rounds can help,” he says, “and collaborative communication is key.”
8. Give the Patient and Family a Voice
“We fully embrace the involvement of the patient in the process of their care,” says Dr. Angood, who is also the co-director of the Joint Commission International Center for Patient Safety, for which patient and family involvement is a priority.
Giving patients and family members a voice is a fine idea, say our hospitalists, especially with children: No one knows a patient like their parents. As the H&HN article points out, anecdotal evidence is largely responsible for the belief that patient and family involvement helps reduce the likelihood for errors, and patient and family participation on safety committees can be a boon to advancing safety as well as satisfaction. But, says Dr. Alverson, “one has to keep in mind that parents have a perspective and not the only perspective on patient safety. I think a broad group of people has to sit down to address these issues.”
In the post-surgical setting, says Dr. Rauch, hospitalists make an invaluable contribution. “If surgeons don’t even come by to listen to the parents or see the child, it’s helpful to have that co-management of someone who’s used to listening to parents, who credits the parents for knowing their kids, and who will do the appropriate thing.” Dr. Angood, who is a past president of the Society of Critical Care Medicine, believes “that that patient-physician relationship is still going to be the driver for the majority of healthcare for some time yet.”
9. Reduce Catheter-Related Bloodstream Infections
“If it’s not required, we want every foreign body out,” says Dr. Manning. “We have to ask ourselves every day whether they are still required.”
The geriatric service at the Mayo Clinic (Rochester, Minn.) developed a daily mnemonic of A-B-C-D-E where B stands for binders. This, he says, “is a way for us to remind ourselves that any therapeutic foreign objects that are tethering the patient—and many of them are catheters—are of concern. We need to push the question [Is this still required?] to ourselves and then act on it.”
Dr. Alverson says, “There are certain infections [for which] we’re starting to move away from PICC [peripherally inserted central catheters] line management, and one way to mitigate that is to be on top of when you can actually discontinue the catheter.” For example, “in pediatrics, there are emerging data that with osteomyelitis you can have a shortened course of IV antibiotics and then switch to oral antibiotics. … That can reduce by half your PICC line duration. Being savvy about this is important.”
10. Reduce Heart Attack Death Rates
“There are about eight interventions for heart attacks that have increased survival,” says Dr. Manning. “So every hospital is working with these. We are using the all-or-none criteria, meaning that there are assurances [in place] that every patient will get all of them.”
Re-engineering systems has been particularly meaningful in preventing and treating heart attacks, says Dr. Manning, who represented SHM at a meeting of the Alliance for Cardiac Care Excellence (ACE), a CMS-based coalition that includes leaders from more than 30 healthcare organizations, and is working to ensure that all hospitalized cardiac patients regularly receive care consistent with nationally accepted standards.
11. Institute Multidisciplinary Rounds
Time constraints mean rounding with 10 people will necessarily be slower, says Dr. Alverson. In academic institutions where the hospitalist has the dual responsibility of teaching, this is especially time-consuming. Although there is an increasing emphasis that providers should participate at bedside rounds, and this is “clearly better from the patient’s perspective and, I would argue, better from the educational perspective,” says Dr. Alverson, it is “fairly bad from the getting-things-done-in-a-timely-fashion perspective. So it’s tough, and to a certain degree, in a practical world you have to pick and choose.”
When a nurse representative is there to respond to the question, “ ‘Why didn’t the kid get his formula? [and says] because he didn’t like the taste,’ that’s something that we might not pick up on,” says Dr. Alverson.
At NYU Medical Center, where Dr. Rauch works, formal rounds take place at least once a week (sometimes more), depending on volume, and they informally take place twice a day, every day.
“It works pretty well,” he says. “The nurses are a critically important part of teams; everybody recognizes that, and they are included in decisions.” Physicians put out the welcome mat for nurses even in casual circumstances. “Sometimes I am discussing things with the house staff [and] a nurse will pull up a chair and become part of the conversation. It’s a part of our culture.”
Although it is unusual to get a pharmacist to round with his team, says Dr. Alverson, a nearby pharmacy school sends students to join rounds, providing what might otherwise be a missing element of education.
12. Avoid Miscommunication
A number of the hospitalists interviewed were asked what they considered to be the top two or three communication points for hospitalists. Verbal orders, clarifying with read-backs, clear handwriting, and order sets were named frequently. In academic settings, says one hospitalist, instructors should be careful to make sure that residents, interns, and medical students understand what you’re saying and why you’re saying it. Good communication with the family was also cited as crucial.
“The most challenging issue is communicating at all,” says Dr. Rauch, who is also an associate professor of pediatrics at the NYU School of Medicine. Although he was the only one to phrase it this way, it is probably not a unique view. “In a large, old, academic medical institution, there are a lot of hierarchical issues that [impede] rapidly responding to [patients’] needs.” Unfortunately, it may mean communicating up one authorization pathway and down another. “And you can see the layers of time and the game of telephone as the concerns go around,” he says. “We’ve tried to break that down so the people who are on site can speak to someone who can make a decision.”
Along with that, he says, it is important from the outset to make it very clear who makes the decision. “For example, when the patient is a child getting neurosurgery because they have a seizure disorder and they also are developmentally delayed and they have medical issues, you now have at least three services involved with managing the child,” says Dr. Rauch. When three people are making decisions, he points out, no decision gets made. “You really have to decide when that child comes in who is going to call the shots for what issue. It’s usually the hospitalist who brings it up, and when it works, it works well.”
13. Empower Nurses and Other Clinicians
Nursing staff should have the power to halt unsafe practices. To Tahl Humes, MD, hospitalist at Exempla St. Joseph’s Hospital in Denver, halting unsafe practices depends, once again, on good lines of communication, and recognizing that patient care is a joint responsibility. For example, she says, “instead of just going to see the patient, writing the note, writing the order, and putting the chart away,” the hospitalists “talk with nurses daily and tell them what they’re planning to do,” so there is more opportunity to catch what might be unsafe practices.
14. Reduce Wound Infections
Although reducing wound infections is something in which their surgical colleagues take the lead, says Dr. Manning, “in our perioperative consultation care, we often work with surgery and anesthesiology in the pre-op evaluations of the patients. So in the surgical care improvement projects, we are often partners.”
Hospitalists are also frequently members on quality committees that help to brainstorm solutions to serious problems. One such project is the Surgical Care Improvement Program (SCIP), spearheaded by David Hunt, MD, with the Office of Clinical Standards and Quality, CMS. SCIP is an effort to transform the prevention of postoperative complications. Its goal is to reduce surgical complications by 25% in the United States by the year 2010 in four target areas: surgical site infections, and cardiac, respiratory, and venous thromboembolic complications. (See Figures 1 and 2, p. 33.)
This includes those patients who are already on beta-blockers. “From the hospitalist’s standpoint,” says Dr. Manning, “we have a real role in … [ensuring] that their beta blockade is maintained.”
Dr. Humes says that at her institution, a wound care nurse can have that responsibility. If a provider is concerned about any patient in this regard, he or she can order that the patient be seen by a wound care nurse and, depending on what’s needed, by a physical therapist.
Now we move on to address those issues that are medication-related:
15. Know Risky Meds
Pediatric hospitalists are involved with postoperative patients at Dr. Rauch’s institution. All patients’ orders are double-checked, he says, and computer order entry also helps providers calculate pediatric dosage norms or dosages calculated by weight.
The hospitalist has the opportunity to be involved in the pharmacy’s selection of drugs for the formulary, says Erin Stucky, MD, pediatric hospitalist at University of California, San Diego, and to help decide the drug choices within a certain class and limit the numbers of things that are used most frequently that are visually different in appearance. “And although that’s the pharmacist’s purview,” she says, “the hospitalist has a vested interest in being on the Pharmacy and Therapeutics Committee to review and restate to pharmacists what they’re using based on clinical need and to find a way for that drug to be safely stored in pharmacy if, indeed, there are a couple of drugs from one class that are truly useful.”
A drug’s generic name, brand name, dose strength, frequency of administration, place of use, indications, and contraindications are all important factors to determine the potential risks of drugs. But “you can’t say a list of risky medications at one institution is the same as it should be elsewhere,” says Dr. Stucky. Risky medications will depend on the setting in which the physician works. Hospitalists need to think logically about the drugs that are the most used or are new, including any new drug that has a different method by which it is administered or a different interaction capability with standard drugs.
“If there’s a new antibiotic that’s known to be processed through the liver and you have multiple patients with heart failure medications who have a medication basis that could be at conflict with that new drug, that’s a potentially risky medication,” she points out. “It may be easier in some ways for the pharmacist to be the rate-limiting factor for how they’re dispensed and for which patients they recheck [against] that incompatibility list.” But in large part, the avoidance of those risky-medication errors must be a commitment of the pharmacist and a bedside nurse.
16. Beware of Sound-Alike and Look-Alike Drugs
Dr. Stucky believes a majority of physicians don’t know the color or size of the pills they’re prescribing. “I would challenge all hospitalists to take every opportunity at the bedside,” she says, “to watch the process happening and know what those drugs and pills need to look like.”
Another opportunity is to educate family members “to remind them that [the patient is] going to be getting these medicines, these are the names of the medicines, and please ask the nurse about these medicines when you get them,” she says. If the hospitalist gives a new drug to the patient, the family can be another safeguard.
Dr. Stucky points out that you can tell the patient and family, “I’m going to tell the nurse that you’re going to be asking about this because … you are the best guide to help us make sure that these medicines are administered safely.” She also emphasizes that assigning this responsibility to the patient is important “because when people leave the hospital, we suddenly expect them to know how to take 18 pills.”
If, on a given unit, you have to handle cases with multiple diagnoses, says Dr. Stucky, it may be difficult to physically isolate the look-alike drugs. “At our institution we found that we actually had to pull the machines out,” she says, referring to the PIXUS units. “You can’t have them on the same wall even in different locations. You have to choose one or the other [similar looking pills].”
The sound-alike drugs are most ripe for errors with verbal orders. “Hospitalists can set a precedent in their institutions that any verbal orders should have the reason for that order given,” she explains. If you order clonazepam, after you finish giving the order verbally to the nurse, you should state, “This is for seizures.”
“When the nurse is writing it down, she may or may not be the one to know that that drug name is indeed in that drug class, but the pharmacist will know,” explains Dr. Stucky.
17. Reconcile Medications
“It is important for people to do verbal sign-out, certainly among attendings,” says Dr. Alverson, “to explain [in better depth] what’s going on with the patient and to maintain those avenues of communication in case something goes wrong. Hospitals get in trouble when physicians aren’t able to communicate or speak with each other readily.”
The biggest challenge for the pediatric hospitalists at NYU Hospital, says Dr. Rauch, is assessing the most up-to-date list of medications. “For instance,” he says, “we had a child yesterday as part of post-op care. I hadn’t met them pre-op. The father said, ‘I think my daughter’s on an experimental protocol with this additional medication.’ It wasn’t something we were used to so we called Mom: Can you bring in the protocol? She said, ‘Oh, she hasn’t been on that drug in a long time.’”
In fact, whether the patient is a child or adult, the majority of cases assigned to hospitalists are unplanned admissions and this is something with which all hospitalists struggle. But regarding transferring patients from unit to unit, says Dr. Stucky, “this is a whole different ballgame. That’s where we have a huge opportunity to make an impact.”
She suggests that matching medications to patients can be ameliorated by computer-based systems in which at each new place the hospitalist can fill in a printout regarding whether they’re continuing a drug order, changing it, or discontinuing it, and this system also works effectively on discharge. “In a perfect world,” says Dr. Stucky, … “the hospitalist would be the implementer of this kind of medication reconciliation in their institution.”
18. Avoid Unacceptable Drug Abbreviations
Some medications have abbreviations that can be misinterpreted. The classic ones, say several hospitalists, are magnesium and morphine. Others pertain to miswritten units of administration.
Read-backs on verbal medication orders was one of the elements most cited by our hospitalists as priority communication practices. Eliminating confusing abbreviations is one of JCAHO’s National Patient Safety Goals and “hospitals are aggressively rolling out ways to remind physicians not to use them,” says Dr. Alverson.
At Dr. Manning’s institution they use the Safest in America criteria, a collaboration of 10 Twin Cities and the hospital systems in Rochester, Minn., as well as the Institute for Clinical Systems Improvement. At Mayo, they call it “Write It Right.” An accentuated campaign to reduce ambiguities in medication communications, he says, has resulted in “profound improvement” in standardizing medication prescribing and following the read-back rules.
Dr. Stucky suggests that hospitalists take on mini-projects where they review the past six months of order-writing errors in their institutions, noticing any trends and, particularly, any unit-specific trends (such as the misunderstanding of the abbreviation cc). If you notice the errors are unit-specific, you can also analyze whether they are treatment-specific. In that way, “order sets can be pre-typed and all the providers have to do is fill in the numbers,” she says, adding that hospitalists can perform these analyses outside their own patient area.
19. Improper Drug Labeling, Packaging, and Storage
Drug names, labels, and packaging contribute significantly to medication errors. The risk for errors is determined by both the product and the environment in which it is used. Most hospitalists say they are continually developing new protocols and checking information multiple times. Sometimes, small changes go a long way. “Our patient safety officer has a favorite phrase: ‘How can I facilitate you to do something different next Tuesday?’ Within your own hospital, that means look at your system and pick something you know you can change,” says Dr. Stucky. “You can’t buy IT tomorrow; you can’t do physician order entries [because] your computer system doesn’t allow it—but what can you do?”
Conclusion
Dr. Angood encourages hospitalists to continue learning how to interact with other disciplines that are also evolving into hospital-based practices and to learn how to manage the specific details-of-change topics such as this list of 25—not just to gloss over them, but to understand them, and to encourage patient involvement and nurture the physician-patient relationship to help change the culture within health care.
“We can pick these kinds of topics and can dissect them all down, but each time, in the end, it is a matter of people and their behaviors as a culture inside a system,” he says. “The system can be changed a little bit, but still it is ultimately about the culture of people.” TH
Andrea Sattinger writes regularly for The Hospitalist.
References
- Runy LA. 25 things you can do to save lives now. Hosp Health Netw. 2005 Apr;79(4):27-28.
- Meengs MR, Giles BK, Chisholm CD, et al. Hand washing frequency in an emergency department. Ann Emerg Med. 1994 Jun;23(6):1307-1312.
- McGuckina M, Watermana R, Storrb J, et al. Evaluation of a patient-empowering hand hygiene programme in the UK. J Hosp Infect. 2001 Jul;48(3):222-227.
- Whitby M, McLaws ML, Ross MW. Why healthcare workers don't wash their hands: a behavioral explanation. Infect Control Hosp Epidemiol. 2006 May;27(5):484-492.
- Lipsett PA, Swoboda SM. Handwashing compliance depends on professional status. Surg Infect. 2001 Fall;2(3):241-245.
A New Hospitalist in the House
Effective spiritual care interventions require the right person with the right training who understands, for example, that spiritual care is more than intercessory prayer alone.1 No one on the healthcare team knows this better than the clinically trained, board-certified professional chaplain.2 When it comes to understanding the unique role of the professional chaplain on the healthcare team, no metaphor communicates this better to physicians and other healthcare providers than that of “hospitalist.” The “chaplain as hospitalist” differentiates the in-house chaplain’s care from that of other spiritual care providers and thereby sheds light on the problematic nexus of managing the provision of spiritual care in healthcare today.3-6
“Hospitalist” as a metaphor for chaplains communicates well with other healthcare professionals whose preconceived notions of chaplains may be based on parish-based models for pastoral care, or on chaplains as harbingers of death, or on the notion that chaplains only hold hands and pray with patients. As a metaphor, the hospitalist compares with the primary care physician as the chaplain compares to local clergy. That is, among all spiritual care providers, including local parish-based clergy and other healthcare professionals (such as physicians, nurses, and social workers) who might consider spirituality as part of their care, the professional chaplain is the “hospitalist.”
Like the hospitalist, the clinically trained and board-certified [hospital-based] chaplain is thoroughly and specifically trained in hospital-based interventions. These interventions range from the more traditional listening, prayer, and religious rituals to those interventions that incorporate an awareness of multi-cultural concerns, including ethical decision-making on end-of-life issues, and those that utilize current psychosocial and behavioral models as they relate to illness responses.7
Unlike many local pastors who come from the surrounding communities to visit hospitalized parishioners, the in-house chaplain is available 24/7 for emergent care and for consultations and provides a continuum of spiritual care from admission through discharge. In-house chaplains also understand and can respond to the variety of spiritual stresses that patients and families experience as a result of being hospitalized.
Whereas local faith representatives from the surrounding communities have little to no input in the patient’s overall plan of care, chaplains—as clinical members of the healthcare team—can advocate effectively for the patients’ spiritual, pastoral, and religious needs through interdisciplinary rounds and charting. In addition, as a valued member of the healthcare team, the in-house chaplain provides spiritual support for staff who are affected by the stresses of working in a healthcare environment.8
Like hospitalists, chaplains provide teaching, research, and leadership within the hospital. Finally, many healthcare providers understand spiritual support as a part of their overall care, but do not see themselves as ultimately responsible for such care.9,10 The “chaplain as hospitalist” can communicate to healthcare providers that the chaplain has the skills, abilities, and responsibility to assess and manage this particular aspect of their clinical care.3-6 When it comes to providing overall spiritual care for inpatients within the hospital context, chaplains are exceptional.
Problems encountered by the hospitalist movement are informative for professional chaplaincy. For example, upon admission and discharge, medical care is transferred between the primary care physician and the hospitalist. Patients often dislike this transfer, and the continuum of care may be disrupted.11 Educating patients and families concerning the benefits of hospitalists helps facilitate this transfer of care, and good communication between hospitalists and primary care physicians ensures a continuum of professional care.12
Complete transfer of spiritual care from local pastor to in-house chaplain will most likely never become standard practice, though it already occurs in special circumstances, as when local clergy are unavailable or in an emerging crisis. Yet, the hospitalist metaphor invites us to examine intentionally how spiritual care is shared among hospital-based chaplains and community-based local clergy. Formalizing and communicating a confidential and effective transfer or sharing of spiritual care will be difficult, but the hospitalist movement provides models that will help.
In summary, using “hospitalist” as a metaphor to describe professional chaplains has advantages and disadvantages. Within the context and culture of healthcare it is advantageous in providing a clinically based metaphor readily accessible to healthcare providers that communicates well the chaplain’s unique role and work. Within the context and culture of faith communities, however, its disadvantage rests in suggesting a transfer of spiritual care between providers.
Regardless of this disadvantage, the “chaplain as hospitalist” metaphor highlights a problematic nexus where authorities, responsibilities, and accountabilities overlap in the provision of spiritual care. It also suggests a possible solution: the chaplain as manager, but not sole provider, of spiritual care. Naming and examining this nexus can only enhance the overall provision of spiritual care. TH
Rev. Dr. Mark LaRocca-Pitts is a board-certified staff chaplain at Athens (Ga.) Regional Medical Center and is endorsed by the United Methodist (UM) Church. He also teaches as an adjunct professor in the Religion Department at the University of Georgia and pastors three small rural UM churches. Rev. Dr. LaRocca-Pitts received his MDiv from Harvard Divinity School and his PhD from Harvard University in Near Eastern Languages and Civilizations. He completed his clinical training in chaplaincy at Emory University and Duke University.
References
- Benson H, Dusek JA, Sherwood JB, et al. Study of the therapeutic effects of intercessory prayer (STEP) in cardiac bypass patients: a multicenter randomized trial of uncertainty and certainty of receiving intercessory prayer. Am Heart J. 2006 Apr;151(4):934-942.
- VandeCreek L, Burton L, eds. Professional chaplaincy: its role and importance in healthcare. J Pastoral Care. 2001 Spring;55(1):81-97.
- Handzo G, Koenig HG. Spiritual care: whose job is it anyway? South Med J. 2004 Dec;97(12):1242-1244.
- Post SG, Puchalski CM, Larson DB. Physicians and patient spirituality: professional boundaries, competency, and ethics. Ann Intern Med. 2000 Apr 4;132(7):578-583.
- Bessinger D, Kuhne T. Medical spirituality: defining domains and boundaries. South Med J. 2002 Dec;95:1385-1388.
- Sloan RP, Bagiella E, VandeCreek L, et al. Should physicians prescribe religious activity? N Engl J Med. 2000 Jun;342(25):1913-1916.7.
- LaRocca-Pitts M. Walking the wards as a spiritual specialist. Harvard Divinity Bulletin. 2004;32,3:20,29.
- LaRocca-Pitts M. The chaplain’s response to moral distress. Chaplaincy Today. 2004;20,2:23-29
- Kristeller JL, Zumbrun CS, Schilling RF. “I would if I could”: how oncologists and oncology nurses address spiritual distress in cancer patients. Psychooncology. 1999 Sep-Oct;8(5):451-458.
- Meyer CL. How effective are nurse educators preparing students to provide spiritual care? Nurse Educ. 2003;28:185-190
- Pantilat SZ, Alpers A, Wachter RM. A new doctor in the house: ethical issues in hospitalist systems. JAMA. 1999 Jul 14; 282(2):171-174.
- Wachter RM, Goldman L. The hospitalist movement 5 years later. JAMA. 2002 Jan 23-30;287(4):487-494.
Resource
For further information on board-certified chaplains, see the Association of Professional Chaplains at www.professionalchaplains.org.
Effective spiritual care interventions require the right person with the right training who understands, for example, that spiritual care is more than intercessory prayer alone.1 No one on the healthcare team knows this better than the clinically trained, board-certified professional chaplain.2 When it comes to understanding the unique role of the professional chaplain on the healthcare team, no metaphor communicates this better to physicians and other healthcare providers than that of “hospitalist.” The “chaplain as hospitalist” differentiates the in-house chaplain’s care from that of other spiritual care providers and thereby sheds light on the problematic nexus of managing the provision of spiritual care in healthcare today.3-6
“Hospitalist” as a metaphor for chaplains communicates well with other healthcare professionals whose preconceived notions of chaplains may be based on parish-based models for pastoral care, or on chaplains as harbingers of death, or on the notion that chaplains only hold hands and pray with patients. As a metaphor, the hospitalist compares with the primary care physician as the chaplain compares to local clergy. That is, among all spiritual care providers, including local parish-based clergy and other healthcare professionals (such as physicians, nurses, and social workers) who might consider spirituality as part of their care, the professional chaplain is the “hospitalist.”
Like the hospitalist, the clinically trained and board-certified [hospital-based] chaplain is thoroughly and specifically trained in hospital-based interventions. These interventions range from the more traditional listening, prayer, and religious rituals to those interventions that incorporate an awareness of multi-cultural concerns, including ethical decision-making on end-of-life issues, and those that utilize current psychosocial and behavioral models as they relate to illness responses.7
Unlike many local pastors who come from the surrounding communities to visit hospitalized parishioners, the in-house chaplain is available 24/7 for emergent care and for consultations and provides a continuum of spiritual care from admission through discharge. In-house chaplains also understand and can respond to the variety of spiritual stresses that patients and families experience as a result of being hospitalized.
Whereas local faith representatives from the surrounding communities have little to no input in the patient’s overall plan of care, chaplains—as clinical members of the healthcare team—can advocate effectively for the patients’ spiritual, pastoral, and religious needs through interdisciplinary rounds and charting. In addition, as a valued member of the healthcare team, the in-house chaplain provides spiritual support for staff who are affected by the stresses of working in a healthcare environment.8
Like hospitalists, chaplains provide teaching, research, and leadership within the hospital. Finally, many healthcare providers understand spiritual support as a part of their overall care, but do not see themselves as ultimately responsible for such care.9,10 The “chaplain as hospitalist” can communicate to healthcare providers that the chaplain has the skills, abilities, and responsibility to assess and manage this particular aspect of their clinical care.3-6 When it comes to providing overall spiritual care for inpatients within the hospital context, chaplains are exceptional.
Problems encountered by the hospitalist movement are informative for professional chaplaincy. For example, upon admission and discharge, medical care is transferred between the primary care physician and the hospitalist. Patients often dislike this transfer, and the continuum of care may be disrupted.11 Educating patients and families concerning the benefits of hospitalists helps facilitate this transfer of care, and good communication between hospitalists and primary care physicians ensures a continuum of professional care.12
Complete transfer of spiritual care from local pastor to in-house chaplain will most likely never become standard practice, though it already occurs in special circumstances, as when local clergy are unavailable or in an emerging crisis. Yet, the hospitalist metaphor invites us to examine intentionally how spiritual care is shared among hospital-based chaplains and community-based local clergy. Formalizing and communicating a confidential and effective transfer or sharing of spiritual care will be difficult, but the hospitalist movement provides models that will help.
In summary, using “hospitalist” as a metaphor to describe professional chaplains has advantages and disadvantages. Within the context and culture of healthcare it is advantageous in providing a clinically based metaphor readily accessible to healthcare providers that communicates well the chaplain’s unique role and work. Within the context and culture of faith communities, however, its disadvantage rests in suggesting a transfer of spiritual care between providers.
Regardless of this disadvantage, the “chaplain as hospitalist” metaphor highlights a problematic nexus where authorities, responsibilities, and accountabilities overlap in the provision of spiritual care. It also suggests a possible solution: the chaplain as manager, but not sole provider, of spiritual care. Naming and examining this nexus can only enhance the overall provision of spiritual care. TH
Rev. Dr. Mark LaRocca-Pitts is a board-certified staff chaplain at Athens (Ga.) Regional Medical Center and is endorsed by the United Methodist (UM) Church. He also teaches as an adjunct professor in the Religion Department at the University of Georgia and pastors three small rural UM churches. Rev. Dr. LaRocca-Pitts received his MDiv from Harvard Divinity School and his PhD from Harvard University in Near Eastern Languages and Civilizations. He completed his clinical training in chaplaincy at Emory University and Duke University.
References
- Benson H, Dusek JA, Sherwood JB, et al. Study of the therapeutic effects of intercessory prayer (STEP) in cardiac bypass patients: a multicenter randomized trial of uncertainty and certainty of receiving intercessory prayer. Am Heart J. 2006 Apr;151(4):934-942.
- VandeCreek L, Burton L, eds. Professional chaplaincy: its role and importance in healthcare. J Pastoral Care. 2001 Spring;55(1):81-97.
- Handzo G, Koenig HG. Spiritual care: whose job is it anyway? South Med J. 2004 Dec;97(12):1242-1244.
- Post SG, Puchalski CM, Larson DB. Physicians and patient spirituality: professional boundaries, competency, and ethics. Ann Intern Med. 2000 Apr 4;132(7):578-583.
- Bessinger D, Kuhne T. Medical spirituality: defining domains and boundaries. South Med J. 2002 Dec;95:1385-1388.
- Sloan RP, Bagiella E, VandeCreek L, et al. Should physicians prescribe religious activity? N Engl J Med. 2000 Jun;342(25):1913-1916.7.
- LaRocca-Pitts M. Walking the wards as a spiritual specialist. Harvard Divinity Bulletin. 2004;32,3:20,29.
- LaRocca-Pitts M. The chaplain’s response to moral distress. Chaplaincy Today. 2004;20,2:23-29
- Kristeller JL, Zumbrun CS, Schilling RF. “I would if I could”: how oncologists and oncology nurses address spiritual distress in cancer patients. Psychooncology. 1999 Sep-Oct;8(5):451-458.
- Meyer CL. How effective are nurse educators preparing students to provide spiritual care? Nurse Educ. 2003;28:185-190
- Pantilat SZ, Alpers A, Wachter RM. A new doctor in the house: ethical issues in hospitalist systems. JAMA. 1999 Jul 14; 282(2):171-174.
- Wachter RM, Goldman L. The hospitalist movement 5 years later. JAMA. 2002 Jan 23-30;287(4):487-494.
Resource
For further information on board-certified chaplains, see the Association of Professional Chaplains at www.professionalchaplains.org.
Effective spiritual care interventions require the right person with the right training who understands, for example, that spiritual care is more than intercessory prayer alone.1 No one on the healthcare team knows this better than the clinically trained, board-certified professional chaplain.2 When it comes to understanding the unique role of the professional chaplain on the healthcare team, no metaphor communicates this better to physicians and other healthcare providers than that of “hospitalist.” The “chaplain as hospitalist” differentiates the in-house chaplain’s care from that of other spiritual care providers and thereby sheds light on the problematic nexus of managing the provision of spiritual care in healthcare today.3-6
“Hospitalist” as a metaphor for chaplains communicates well with other healthcare professionals whose preconceived notions of chaplains may be based on parish-based models for pastoral care, or on chaplains as harbingers of death, or on the notion that chaplains only hold hands and pray with patients. As a metaphor, the hospitalist compares with the primary care physician as the chaplain compares to local clergy. That is, among all spiritual care providers, including local parish-based clergy and other healthcare professionals (such as physicians, nurses, and social workers) who might consider spirituality as part of their care, the professional chaplain is the “hospitalist.”
Like the hospitalist, the clinically trained and board-certified [hospital-based] chaplain is thoroughly and specifically trained in hospital-based interventions. These interventions range from the more traditional listening, prayer, and religious rituals to those interventions that incorporate an awareness of multi-cultural concerns, including ethical decision-making on end-of-life issues, and those that utilize current psychosocial and behavioral models as they relate to illness responses.7
Unlike many local pastors who come from the surrounding communities to visit hospitalized parishioners, the in-house chaplain is available 24/7 for emergent care and for consultations and provides a continuum of spiritual care from admission through discharge. In-house chaplains also understand and can respond to the variety of spiritual stresses that patients and families experience as a result of being hospitalized.
Whereas local faith representatives from the surrounding communities have little to no input in the patient’s overall plan of care, chaplains—as clinical members of the healthcare team—can advocate effectively for the patients’ spiritual, pastoral, and religious needs through interdisciplinary rounds and charting. In addition, as a valued member of the healthcare team, the in-house chaplain provides spiritual support for staff who are affected by the stresses of working in a healthcare environment.8
Like hospitalists, chaplains provide teaching, research, and leadership within the hospital. Finally, many healthcare providers understand spiritual support as a part of their overall care, but do not see themselves as ultimately responsible for such care.9,10 The “chaplain as hospitalist” can communicate to healthcare providers that the chaplain has the skills, abilities, and responsibility to assess and manage this particular aspect of their clinical care.3-6 When it comes to providing overall spiritual care for inpatients within the hospital context, chaplains are exceptional.
Problems encountered by the hospitalist movement are informative for professional chaplaincy. For example, upon admission and discharge, medical care is transferred between the primary care physician and the hospitalist. Patients often dislike this transfer, and the continuum of care may be disrupted.11 Educating patients and families concerning the benefits of hospitalists helps facilitate this transfer of care, and good communication between hospitalists and primary care physicians ensures a continuum of professional care.12
Complete transfer of spiritual care from local pastor to in-house chaplain will most likely never become standard practice, though it already occurs in special circumstances, as when local clergy are unavailable or in an emerging crisis. Yet, the hospitalist metaphor invites us to examine intentionally how spiritual care is shared among hospital-based chaplains and community-based local clergy. Formalizing and communicating a confidential and effective transfer or sharing of spiritual care will be difficult, but the hospitalist movement provides models that will help.
In summary, using “hospitalist” as a metaphor to describe professional chaplains has advantages and disadvantages. Within the context and culture of healthcare it is advantageous in providing a clinically based metaphor readily accessible to healthcare providers that communicates well the chaplain’s unique role and work. Within the context and culture of faith communities, however, its disadvantage rests in suggesting a transfer of spiritual care between providers.
Regardless of this disadvantage, the “chaplain as hospitalist” metaphor highlights a problematic nexus where authorities, responsibilities, and accountabilities overlap in the provision of spiritual care. It also suggests a possible solution: the chaplain as manager, but not sole provider, of spiritual care. Naming and examining this nexus can only enhance the overall provision of spiritual care. TH
Rev. Dr. Mark LaRocca-Pitts is a board-certified staff chaplain at Athens (Ga.) Regional Medical Center and is endorsed by the United Methodist (UM) Church. He also teaches as an adjunct professor in the Religion Department at the University of Georgia and pastors three small rural UM churches. Rev. Dr. LaRocca-Pitts received his MDiv from Harvard Divinity School and his PhD from Harvard University in Near Eastern Languages and Civilizations. He completed his clinical training in chaplaincy at Emory University and Duke University.
References
- Benson H, Dusek JA, Sherwood JB, et al. Study of the therapeutic effects of intercessory prayer (STEP) in cardiac bypass patients: a multicenter randomized trial of uncertainty and certainty of receiving intercessory prayer. Am Heart J. 2006 Apr;151(4):934-942.
- VandeCreek L, Burton L, eds. Professional chaplaincy: its role and importance in healthcare. J Pastoral Care. 2001 Spring;55(1):81-97.
- Handzo G, Koenig HG. Spiritual care: whose job is it anyway? South Med J. 2004 Dec;97(12):1242-1244.
- Post SG, Puchalski CM, Larson DB. Physicians and patient spirituality: professional boundaries, competency, and ethics. Ann Intern Med. 2000 Apr 4;132(7):578-583.
- Bessinger D, Kuhne T. Medical spirituality: defining domains and boundaries. South Med J. 2002 Dec;95:1385-1388.
- Sloan RP, Bagiella E, VandeCreek L, et al. Should physicians prescribe religious activity? N Engl J Med. 2000 Jun;342(25):1913-1916.7.
- LaRocca-Pitts M. Walking the wards as a spiritual specialist. Harvard Divinity Bulletin. 2004;32,3:20,29.
- LaRocca-Pitts M. The chaplain’s response to moral distress. Chaplaincy Today. 2004;20,2:23-29
- Kristeller JL, Zumbrun CS, Schilling RF. “I would if I could”: how oncologists and oncology nurses address spiritual distress in cancer patients. Psychooncology. 1999 Sep-Oct;8(5):451-458.
- Meyer CL. How effective are nurse educators preparing students to provide spiritual care? Nurse Educ. 2003;28:185-190
- Pantilat SZ, Alpers A, Wachter RM. A new doctor in the house: ethical issues in hospitalist systems. JAMA. 1999 Jul 14; 282(2):171-174.
- Wachter RM, Goldman L. The hospitalist movement 5 years later. JAMA. 2002 Jan 23-30;287(4):487-494.
Resource
For further information on board-certified chaplains, see the Association of Professional Chaplains at www.professionalchaplains.org.
No Shortcuts
Despite repeated warnings for more than 25 years by the Institute for Safe Medication Practices (ISMP) and other organizations, one of the major causes of medication errors is the ongoing use of potentially dangerous abbreviations and dose expressions. Root cause analyses information contained in the Joint Commission Sentinel Event Database shows that the underlying factors contributing to many of these medication errors are illegible or confusing handwriting by clinicians and the failure of healthcare providers to communicate clearly with one another.
Symbols and abbreviations are frequently used to save time and effort when writing prescriptions and documenting in patient charts; however, some symbols and abbreviations have the potential for misinterpretation or confusion. Examples of especially problematic abbreviations include “U” for “units” and “µg” for “micrograms.” When “U” is handwritten, it can often look like a zero. There are numerous case reports where the root cause of sentinel events related to insulin dosage has been the interpretation of a “U” as a zero. Using the abbreviation “µg” instead of “mcg” has also been the source of errors because when handwritten, the symbol “µ” can look like an “m.” The use of trailing zeros (e.g., 2.0 versus 2) or use of a leading decimal point without a leading zero (e.g. .2 instead of 0.2) are other dangerous order-writing practices. The decimal point is sometimes not seen when orders are handwritten using trailing zeros or no leading zeros. Misinterpretation of such orders could lead to a 10-fold dosing error.
A New Approach
As part of efforts to improve patient safety, the Joint Commission has long worked with hospitals to develop practical, cost-effective strategies that can be implemented at organizations regardless of unique characteristics, such as ownership, size, or location. One such Joint Commission initiative is a National Patient Safety Goal to improve communication. This goal and one of its requirements specifically addresses the role that abbreviations, acronyms, symbols, and dose designations play in medication errors.
The Joint Commission began establishing National Patient Safety Goals in 2002 as a means to target critical areas where patient safety can be improved through specific action in healthcare organizations. The resulting National Patient Safety Goals are designed to give focus to evidence-based or expert consensus-based, well-defined, practical, and cost-effective actions that have potential for significant improvement in the safety of individuals receiving care. New Goals are recommended annually by the Sentinel Event Advisory Group, a Joint Commission-appointed, multidisciplinary group of patient safety experts.
JCAHO Expectations
In order to comply with the National Patient Safety Goal related to abbreviations, an organization must conduct a thorough review of its approved abbreviation list and develop a list of unacceptable abbreviations and symbols with the involvement of physicians. In addition, organizations must do the following to meet this goal:
- The list of prohibited abbreviations, acronyms, symbols, and dose designations must be implemented for all handwritten, patient-specific communications, not just medication orders;
- These requirements apply to printed or electronic communications;
- This goal requires organizations to achieve 100% compliance with a reasonably comprehensive list of prohibited dangerous abbreviations, acronyms, symbols, and dose designations. This list need not be as extensive as some published lists, but must, at a minimum, include a set of Joint Commission-specified dangerous abbreviations, acro-nyms, symbols, and dose designations (see “Minimum List of Abbreviations, Acronyms, and Symbols Not to Use,” top right) and
- An abbreviation on the “do not use” list should not be used in any of its forms—uppercase or lowercase, with or without periods.
In addition to this minimum list, each organization should consider which abbreviations, acronyms, symbols, and dose designations it commonly uses; examine the risks associated with usage; and develop strategies to reduce usage. Hospitals also may wish to look to expert resources such as the Institute of Safe Medication Practices (ISMP)—available at www.ismp.org/Tools/abbreviationslist.pdf—and the National Coordinating Council for Medication Error Reporting and Prevention (NCC MERP)—available at www.nccmerp.org/dangerousAbbrev.html—to develop a list of prohibited abbreviations. Finally, organizations may wish to consider the Joint Commission list (see table below) of abbreviations, symbols, and acronyms for future possible inclusion in the official “do not use” list.
Risk Reduction Strategies
To comply with this National Patient Safety Goal Requirement, hospitals may wish to consider implementing the following risk reduction strategies:
- Examine medication error data in the organization. By identifying and selecting an organization-specific set of prohibited abbreviations, it will be much easier to gain support to eliminate certain abbreviations that have been found to be problematic at that organization.
- Provide simplified alternative abbreviations. For example, some staff members may resist writing out “international units” in place of “IU.” A simpler alternative such as “Intl Units” may be a solution.
- Make the list visible. Print the list on brightly colored paper or stickers and place it in patient charts.
- Provide staff with pocket-sized cards with the “do not use” list.
- Print the list in the margin or bottom of the physician order sheets and/or progress notes.
- Attach laminated copies of the list to the back of the physician order divider in the patient chart.
- Send monthly reminders to staff.
- Delete prohibited abbreviations from preprinted order sheets and other forms.
- Work with software vendors to ensure changes are made to be consistent with the list.
- Take a digital picture or scan the document containing the prohibited abbreviation and send it via e-mail directly to the offending prescriber to call attention to the issue.
- Direct the pharmacy not to accept any of the prohibited abbreviations. Orders with dangerous abbreviations or illegible handwriting must be corrected before being dispensed.
- Conduct a mock survey to test staff knowledge.
- At every staff meeting give patient safety updates, including information about the prohibited abbreviations.
- Ask all staff to sign a statement that he or she has received the list and agrees not to use the abbreviations.
- Promote a “do-not-use abbreviation of the month” policy.
- Develop and implement a policy to ensure that staff refer to the list and take steps to ensure compliance. Consider including a policy that states if an unacceptable abbreviation is used, the prescriber verifies the prescription order before it is filled.
- Monitor staff compliance with the list and offer additional education and training, as appropriate.
Conclusion
During the past decade, healthcare providers have been searching for more effective ways to reduce the risk of systems breakdowns that result in serious harm to patients. The Joint Commission is committed to working with organizations through the accreditation process on ways to anticipate and prevent errors. National Patient Safety Goals, such as the one associated with prohibited abbreviations, are one such method to promote specific improvements in patient safety. By using the principals of sound system design, organizations and providers further strengthen foundations that support safe, high-quality care. TH
Dr. Jacott is special advisor for professional relations for the Joint Commission on Accreditation of Healthcare Organizations.
Despite repeated warnings for more than 25 years by the Institute for Safe Medication Practices (ISMP) and other organizations, one of the major causes of medication errors is the ongoing use of potentially dangerous abbreviations and dose expressions. Root cause analyses information contained in the Joint Commission Sentinel Event Database shows that the underlying factors contributing to many of these medication errors are illegible or confusing handwriting by clinicians and the failure of healthcare providers to communicate clearly with one another.
Symbols and abbreviations are frequently used to save time and effort when writing prescriptions and documenting in patient charts; however, some symbols and abbreviations have the potential for misinterpretation or confusion. Examples of especially problematic abbreviations include “U” for “units” and “µg” for “micrograms.” When “U” is handwritten, it can often look like a zero. There are numerous case reports where the root cause of sentinel events related to insulin dosage has been the interpretation of a “U” as a zero. Using the abbreviation “µg” instead of “mcg” has also been the source of errors because when handwritten, the symbol “µ” can look like an “m.” The use of trailing zeros (e.g., 2.0 versus 2) or use of a leading decimal point without a leading zero (e.g. .2 instead of 0.2) are other dangerous order-writing practices. The decimal point is sometimes not seen when orders are handwritten using trailing zeros or no leading zeros. Misinterpretation of such orders could lead to a 10-fold dosing error.
A New Approach
As part of efforts to improve patient safety, the Joint Commission has long worked with hospitals to develop practical, cost-effective strategies that can be implemented at organizations regardless of unique characteristics, such as ownership, size, or location. One such Joint Commission initiative is a National Patient Safety Goal to improve communication. This goal and one of its requirements specifically addresses the role that abbreviations, acronyms, symbols, and dose designations play in medication errors.
The Joint Commission began establishing National Patient Safety Goals in 2002 as a means to target critical areas where patient safety can be improved through specific action in healthcare organizations. The resulting National Patient Safety Goals are designed to give focus to evidence-based or expert consensus-based, well-defined, practical, and cost-effective actions that have potential for significant improvement in the safety of individuals receiving care. New Goals are recommended annually by the Sentinel Event Advisory Group, a Joint Commission-appointed, multidisciplinary group of patient safety experts.
JCAHO Expectations
In order to comply with the National Patient Safety Goal related to abbreviations, an organization must conduct a thorough review of its approved abbreviation list and develop a list of unacceptable abbreviations and symbols with the involvement of physicians. In addition, organizations must do the following to meet this goal:
- The list of prohibited abbreviations, acronyms, symbols, and dose designations must be implemented for all handwritten, patient-specific communications, not just medication orders;
- These requirements apply to printed or electronic communications;
- This goal requires organizations to achieve 100% compliance with a reasonably comprehensive list of prohibited dangerous abbreviations, acronyms, symbols, and dose designations. This list need not be as extensive as some published lists, but must, at a minimum, include a set of Joint Commission-specified dangerous abbreviations, acro-nyms, symbols, and dose designations (see “Minimum List of Abbreviations, Acronyms, and Symbols Not to Use,” top right) and
- An abbreviation on the “do not use” list should not be used in any of its forms—uppercase or lowercase, with or without periods.
In addition to this minimum list, each organization should consider which abbreviations, acronyms, symbols, and dose designations it commonly uses; examine the risks associated with usage; and develop strategies to reduce usage. Hospitals also may wish to look to expert resources such as the Institute of Safe Medication Practices (ISMP)—available at www.ismp.org/Tools/abbreviationslist.pdf—and the National Coordinating Council for Medication Error Reporting and Prevention (NCC MERP)—available at www.nccmerp.org/dangerousAbbrev.html—to develop a list of prohibited abbreviations. Finally, organizations may wish to consider the Joint Commission list (see table below) of abbreviations, symbols, and acronyms for future possible inclusion in the official “do not use” list.
Risk Reduction Strategies
To comply with this National Patient Safety Goal Requirement, hospitals may wish to consider implementing the following risk reduction strategies:
- Examine medication error data in the organization. By identifying and selecting an organization-specific set of prohibited abbreviations, it will be much easier to gain support to eliminate certain abbreviations that have been found to be problematic at that organization.
- Provide simplified alternative abbreviations. For example, some staff members may resist writing out “international units” in place of “IU.” A simpler alternative such as “Intl Units” may be a solution.
- Make the list visible. Print the list on brightly colored paper or stickers and place it in patient charts.
- Provide staff with pocket-sized cards with the “do not use” list.
- Print the list in the margin or bottom of the physician order sheets and/or progress notes.
- Attach laminated copies of the list to the back of the physician order divider in the patient chart.
- Send monthly reminders to staff.
- Delete prohibited abbreviations from preprinted order sheets and other forms.
- Work with software vendors to ensure changes are made to be consistent with the list.
- Take a digital picture or scan the document containing the prohibited abbreviation and send it via e-mail directly to the offending prescriber to call attention to the issue.
- Direct the pharmacy not to accept any of the prohibited abbreviations. Orders with dangerous abbreviations or illegible handwriting must be corrected before being dispensed.
- Conduct a mock survey to test staff knowledge.
- At every staff meeting give patient safety updates, including information about the prohibited abbreviations.
- Ask all staff to sign a statement that he or she has received the list and agrees not to use the abbreviations.
- Promote a “do-not-use abbreviation of the month” policy.
- Develop and implement a policy to ensure that staff refer to the list and take steps to ensure compliance. Consider including a policy that states if an unacceptable abbreviation is used, the prescriber verifies the prescription order before it is filled.
- Monitor staff compliance with the list and offer additional education and training, as appropriate.
Conclusion
During the past decade, healthcare providers have been searching for more effective ways to reduce the risk of systems breakdowns that result in serious harm to patients. The Joint Commission is committed to working with organizations through the accreditation process on ways to anticipate and prevent errors. National Patient Safety Goals, such as the one associated with prohibited abbreviations, are one such method to promote specific improvements in patient safety. By using the principals of sound system design, organizations and providers further strengthen foundations that support safe, high-quality care. TH
Dr. Jacott is special advisor for professional relations for the Joint Commission on Accreditation of Healthcare Organizations.
Despite repeated warnings for more than 25 years by the Institute for Safe Medication Practices (ISMP) and other organizations, one of the major causes of medication errors is the ongoing use of potentially dangerous abbreviations and dose expressions. Root cause analyses information contained in the Joint Commission Sentinel Event Database shows that the underlying factors contributing to many of these medication errors are illegible or confusing handwriting by clinicians and the failure of healthcare providers to communicate clearly with one another.
Symbols and abbreviations are frequently used to save time and effort when writing prescriptions and documenting in patient charts; however, some symbols and abbreviations have the potential for misinterpretation or confusion. Examples of especially problematic abbreviations include “U” for “units” and “µg” for “micrograms.” When “U” is handwritten, it can often look like a zero. There are numerous case reports where the root cause of sentinel events related to insulin dosage has been the interpretation of a “U” as a zero. Using the abbreviation “µg” instead of “mcg” has also been the source of errors because when handwritten, the symbol “µ” can look like an “m.” The use of trailing zeros (e.g., 2.0 versus 2) or use of a leading decimal point without a leading zero (e.g. .2 instead of 0.2) are other dangerous order-writing practices. The decimal point is sometimes not seen when orders are handwritten using trailing zeros or no leading zeros. Misinterpretation of such orders could lead to a 10-fold dosing error.
A New Approach
As part of efforts to improve patient safety, the Joint Commission has long worked with hospitals to develop practical, cost-effective strategies that can be implemented at organizations regardless of unique characteristics, such as ownership, size, or location. One such Joint Commission initiative is a National Patient Safety Goal to improve communication. This goal and one of its requirements specifically addresses the role that abbreviations, acronyms, symbols, and dose designations play in medication errors.
The Joint Commission began establishing National Patient Safety Goals in 2002 as a means to target critical areas where patient safety can be improved through specific action in healthcare organizations. The resulting National Patient Safety Goals are designed to give focus to evidence-based or expert consensus-based, well-defined, practical, and cost-effective actions that have potential for significant improvement in the safety of individuals receiving care. New Goals are recommended annually by the Sentinel Event Advisory Group, a Joint Commission-appointed, multidisciplinary group of patient safety experts.
JCAHO Expectations
In order to comply with the National Patient Safety Goal related to abbreviations, an organization must conduct a thorough review of its approved abbreviation list and develop a list of unacceptable abbreviations and symbols with the involvement of physicians. In addition, organizations must do the following to meet this goal:
- The list of prohibited abbreviations, acronyms, symbols, and dose designations must be implemented for all handwritten, patient-specific communications, not just medication orders;
- These requirements apply to printed or electronic communications;
- This goal requires organizations to achieve 100% compliance with a reasonably comprehensive list of prohibited dangerous abbreviations, acronyms, symbols, and dose designations. This list need not be as extensive as some published lists, but must, at a minimum, include a set of Joint Commission-specified dangerous abbreviations, acro-nyms, symbols, and dose designations (see “Minimum List of Abbreviations, Acronyms, and Symbols Not to Use,” top right) and
- An abbreviation on the “do not use” list should not be used in any of its forms—uppercase or lowercase, with or without periods.
In addition to this minimum list, each organization should consider which abbreviations, acronyms, symbols, and dose designations it commonly uses; examine the risks associated with usage; and develop strategies to reduce usage. Hospitals also may wish to look to expert resources such as the Institute of Safe Medication Practices (ISMP)—available at www.ismp.org/Tools/abbreviationslist.pdf—and the National Coordinating Council for Medication Error Reporting and Prevention (NCC MERP)—available at www.nccmerp.org/dangerousAbbrev.html—to develop a list of prohibited abbreviations. Finally, organizations may wish to consider the Joint Commission list (see table below) of abbreviations, symbols, and acronyms for future possible inclusion in the official “do not use” list.
Risk Reduction Strategies
To comply with this National Patient Safety Goal Requirement, hospitals may wish to consider implementing the following risk reduction strategies:
- Examine medication error data in the organization. By identifying and selecting an organization-specific set of prohibited abbreviations, it will be much easier to gain support to eliminate certain abbreviations that have been found to be problematic at that organization.
- Provide simplified alternative abbreviations. For example, some staff members may resist writing out “international units” in place of “IU.” A simpler alternative such as “Intl Units” may be a solution.
- Make the list visible. Print the list on brightly colored paper or stickers and place it in patient charts.
- Provide staff with pocket-sized cards with the “do not use” list.
- Print the list in the margin or bottom of the physician order sheets and/or progress notes.
- Attach laminated copies of the list to the back of the physician order divider in the patient chart.
- Send monthly reminders to staff.
- Delete prohibited abbreviations from preprinted order sheets and other forms.
- Work with software vendors to ensure changes are made to be consistent with the list.
- Take a digital picture or scan the document containing the prohibited abbreviation and send it via e-mail directly to the offending prescriber to call attention to the issue.
- Direct the pharmacy not to accept any of the prohibited abbreviations. Orders with dangerous abbreviations or illegible handwriting must be corrected before being dispensed.
- Conduct a mock survey to test staff knowledge.
- At every staff meeting give patient safety updates, including information about the prohibited abbreviations.
- Ask all staff to sign a statement that he or she has received the list and agrees not to use the abbreviations.
- Promote a “do-not-use abbreviation of the month” policy.
- Develop and implement a policy to ensure that staff refer to the list and take steps to ensure compliance. Consider including a policy that states if an unacceptable abbreviation is used, the prescriber verifies the prescription order before it is filled.
- Monitor staff compliance with the list and offer additional education and training, as appropriate.
Conclusion
During the past decade, healthcare providers have been searching for more effective ways to reduce the risk of systems breakdowns that result in serious harm to patients. The Joint Commission is committed to working with organizations through the accreditation process on ways to anticipate and prevent errors. National Patient Safety Goals, such as the one associated with prohibited abbreviations, are one such method to promote specific improvements in patient safety. By using the principals of sound system design, organizations and providers further strengthen foundations that support safe, high-quality care. TH
Dr. Jacott is special advisor for professional relations for the Joint Commission on Accreditation of Healthcare Organizations.
Pain Killers
A number of pharmaceutical agents are FDA-approved to treat different types of neuropathic pain. For example, the most recently approved agents include pregabalin (Lyrica, Pfizer) and duloxetine (Cymbalta, Eli Lilly & Company). Additionally Neurontin (gabapentin, Pfizer) and its generics have been available since the 1990s, and have also been used to treat neuropathic pain (it is specifically FDA-approved to treat post-herpetic neuralgia [PHN]).
Lyrica
In June 2005, Lyrica received FDA-approval for treatment of neuropathic pain associated with diabetic peripheral neuropathy (DPN) and PHN as well as for treatment of adults with partial onset seizures. The maximum recommended dose of Lyrica for DPN is 100mg three times daily (TID). When treating patients with a creatinine clearance (CrCL) of < 60 mL/min, initiate at a lower dose (50 mg TID) because it is primarily renally eliminated. When discontinuing Lyrica, it should be gradually tapered over a minimum of one week. The recommended dose for treatment of PHN is 75–150 mg twice daily (BID), or 50–100 mg TID for patients with a CrCL of at least 60mL/min. The dose should be reduced for patients with a CrCL < 60mL/min. If no pain relief is obtained within two to four weeks at this dose, it may be gradually increased to 600 mg/day (200 mg TID or 300 mg BID); however, side effects may be more severe because they are dose-dependent (e.g., ataxia, dizziness, dry mouth, peripheral edema, somnolence, and weight gain).
Other guidelines for dosing in renally impaired patients should be reviewed in the product labeling. Because Lyrica has been reported to cause euphoria, it has been designated as a controlled substance (C-V). There are no specific drug interactions except with the glitazones (weight gain, fluid retention, or congestive heart failure exacerbation may occur) and the possibility of additive central nervous system (CNS) effects if given with other agents that affect the CNS. Lyrica is available in the following strength capsules: 25, 50, 75, 100, 150, 200, 225, and 300mg. Head-to-head comparisons to similar agents are not available.
Cymbalta
In August 2004, the FDA approved Cymbalta for treatment of DPN and major depressive disorder (MDD). Dosing for treatment of DPN is 60 mg once daily. A lower starting dose may be used in patients with renal impairment. (It should not be used in patients with a CrCL <30 mL/min.) Common adverse effects include nausea, dizziness, somnolence, constipation, dry mouth, and increased sweating. Serum transaminase elevations have also been reported. Because Cymbalta is metabolized by CYP1A2 and CYP2D6, numerous drug interactions may occur. Drug discontinuation should be performed gradually to avoid withdrawal symptoms. Cymbalta capsules are delayed-release and are available in the following strengths: 20, 30, and 60 mg. (They should not be opened or crushed prior to administration).
Diabetic Peripheral Neuropathic Pain
Earlier this year, a Consensus Guideline on the management of diabetic peripheral neuropathic pain (DPNP) was published, the first of its kind. Treatment of DPNP may mirror other peripheral neuropathic pain syndromes, and, therefore, this guideline may assist in managing other similar patients.
A goal of 100% pain relief is ideal but often unrealistic. Many patients will only experience a 30%-50% reduction in pain relief; however, this may enable the patient to return to social activities or work and improve their quality of life. Hospitalists and other members of the healthcare team must keep in mind that the patients’ treatment goals may significantly differ from their own goals of therapy. In managing these complex patients we must bear in mind that complete pain relief may not be attainable. We must also continue to communicate with our patients and provide them with information on what is known and unknown about the mechanisms and treatment of neuropathic pain. By developing and maintaining these patient relationships, our patients will apt to be more satisfied with their treatment, even if they do not have 100% improvement.
In DPNP (there are many patients who may have this and not know that they are diabetic or may be in denial about the degree of their diabetes), it is important for the patient to play an active role in their care (e.g., glycemic control, foot care, analgesic treatment). If treatment plans are not for FDA-approved uses, obtain patient consent. Remember, patients now have access to approved labeling via the internet. If they feel that their healthcare providers are not being “above aboard,” lack of trust can significantly affect care.
Neurontin
One of the more commonly used agents to treat neuropathic syndromes is Neurontin (gabapentin). Disadvantages to the use of gabapentin include the need for dose titration and multiple daily doses. Gabapentin is a good alternative as a second-line agent for patients with DPNP who don’t respond well to or can’t tolerate first-line agents (approved agents or others with evidence: e.g., oxycodone controlled-release, tricyclic antidepressants, Lyrica, Cymbalta).
It is recommended that treatment is begun using a first-line agent. Then each time you evaluate the patient, ask them whether the pain is worse or whether the nature of the pain has changed. They should also be asked if they are experiencing any adverse effects. The agent should be titrated to the maximum tolerated dose with an expected goal of at least 50% pain reduction from baseline. Some pain improvement should be expected within three weeks of therapy initiation. Therefore ascertain that this is followed upon hospital discharge. If no improvement is noted within three weeks, modification of therapy may be warranted.
If the patient derives some (but not optimal) therapy benefit without adverse effects, consider adding a second agent. The agent can be another first-line agent or a second-line agent. Consider rational pharmacotherapy (e.g., avoid additive side effects, consider synergy of agents, avoid drug interactions), and use an agent with a different mechanism of action.
If the patient is receiving no benefit from the current therapy or they are experiencing intolerable adverse effects, consider changing to another agent with a different mechanism of action. If the current agent is Cymbalta, Lyrica, or Neurontin (and the patient has no risk of seizures), taper the drug off over at least one week. When starting a new treatment, always take into consideration the patient’s medical and psychiatric comorbidities, any potential contraindications, and other factors such as the potential for drug interactions, side effects (e.g., weight gain, edema), and/or cost.
Topical therapies may also provide some benefit to the patient with neuropathic pain syndromes (e.g., capsaicin, lidocaine 5% patch). Remember that a specialist can always be consulted for expert advice or for difficult-to-manage patients. TH
Michele B. Kaufman is a drug information specialist and a medical writer based in New York City.
References
- Neurontin (gabapentin) [package insert]. New York: Pfizer Inc;. December 2005.
- Lyrica (pregabalin) [package insert]. New York: Pfizer Inc; March 2006.
- Cymbalta (duloxetine) [package insert]. Indianapolis: Eli Lilly and Company; December 14, 2005.
- The Medical Letter on Drugs and Therapeutics. Duloxetine; Volume 47 (Issue 1215/1216), August 15/29, 2005; 67-68.
- Pregabalin. The Medical Letter on Drugs and Therapeutics. 47(1217) :75-77. Available online at www.medletter.com/restricted/articles/w1217b.pdf. Last accessed on July 28, 2006.
- Facts and Comparisons Updated Monthly. Wolters Kluwer Health: October 2005.
- Thomson Micromedex Healthcare Series: Document Comparison of Pregabalin and Gabapentin. Available at: www.thomsonhc.com/hcs/librarian/PFDefaultActionId/pf.PrintReady. Last accessed June 7, 2006.
- Argoff CE, Backonia M-M, Belgrade ML, et al. Consensus Guidelines: treatment planning and options. Mayo Clin Proc. 2006;81(4):S12-S25.
- Argoff CE, Cole BE, Fishbain DA, et al. Diabetic Peripheral neuropathic pain: clinical and quality-of-life issues. Mayo Clin Proc. 2006;81(4, suppl):S3-S11.
- Belgrade ML, Cole BE, McCarberg BH, et al. Diabetic peripheral neuropathic pain: case studies. Mayo Clin Proc. 2006 Apr;81(4 Suppl):S26-S32.
A number of pharmaceutical agents are FDA-approved to treat different types of neuropathic pain. For example, the most recently approved agents include pregabalin (Lyrica, Pfizer) and duloxetine (Cymbalta, Eli Lilly & Company). Additionally Neurontin (gabapentin, Pfizer) and its generics have been available since the 1990s, and have also been used to treat neuropathic pain (it is specifically FDA-approved to treat post-herpetic neuralgia [PHN]).
Lyrica
In June 2005, Lyrica received FDA-approval for treatment of neuropathic pain associated with diabetic peripheral neuropathy (DPN) and PHN as well as for treatment of adults with partial onset seizures. The maximum recommended dose of Lyrica for DPN is 100mg three times daily (TID). When treating patients with a creatinine clearance (CrCL) of < 60 mL/min, initiate at a lower dose (50 mg TID) because it is primarily renally eliminated. When discontinuing Lyrica, it should be gradually tapered over a minimum of one week. The recommended dose for treatment of PHN is 75–150 mg twice daily (BID), or 50–100 mg TID for patients with a CrCL of at least 60mL/min. The dose should be reduced for patients with a CrCL < 60mL/min. If no pain relief is obtained within two to four weeks at this dose, it may be gradually increased to 600 mg/day (200 mg TID or 300 mg BID); however, side effects may be more severe because they are dose-dependent (e.g., ataxia, dizziness, dry mouth, peripheral edema, somnolence, and weight gain).
Other guidelines for dosing in renally impaired patients should be reviewed in the product labeling. Because Lyrica has been reported to cause euphoria, it has been designated as a controlled substance (C-V). There are no specific drug interactions except with the glitazones (weight gain, fluid retention, or congestive heart failure exacerbation may occur) and the possibility of additive central nervous system (CNS) effects if given with other agents that affect the CNS. Lyrica is available in the following strength capsules: 25, 50, 75, 100, 150, 200, 225, and 300mg. Head-to-head comparisons to similar agents are not available.
Cymbalta
In August 2004, the FDA approved Cymbalta for treatment of DPN and major depressive disorder (MDD). Dosing for treatment of DPN is 60 mg once daily. A lower starting dose may be used in patients with renal impairment. (It should not be used in patients with a CrCL <30 mL/min.) Common adverse effects include nausea, dizziness, somnolence, constipation, dry mouth, and increased sweating. Serum transaminase elevations have also been reported. Because Cymbalta is metabolized by CYP1A2 and CYP2D6, numerous drug interactions may occur. Drug discontinuation should be performed gradually to avoid withdrawal symptoms. Cymbalta capsules are delayed-release and are available in the following strengths: 20, 30, and 60 mg. (They should not be opened or crushed prior to administration).
Diabetic Peripheral Neuropathic Pain
Earlier this year, a Consensus Guideline on the management of diabetic peripheral neuropathic pain (DPNP) was published, the first of its kind. Treatment of DPNP may mirror other peripheral neuropathic pain syndromes, and, therefore, this guideline may assist in managing other similar patients.
A goal of 100% pain relief is ideal but often unrealistic. Many patients will only experience a 30%-50% reduction in pain relief; however, this may enable the patient to return to social activities or work and improve their quality of life. Hospitalists and other members of the healthcare team must keep in mind that the patients’ treatment goals may significantly differ from their own goals of therapy. In managing these complex patients we must bear in mind that complete pain relief may not be attainable. We must also continue to communicate with our patients and provide them with information on what is known and unknown about the mechanisms and treatment of neuropathic pain. By developing and maintaining these patient relationships, our patients will apt to be more satisfied with their treatment, even if they do not have 100% improvement.
In DPNP (there are many patients who may have this and not know that they are diabetic or may be in denial about the degree of their diabetes), it is important for the patient to play an active role in their care (e.g., glycemic control, foot care, analgesic treatment). If treatment plans are not for FDA-approved uses, obtain patient consent. Remember, patients now have access to approved labeling via the internet. If they feel that their healthcare providers are not being “above aboard,” lack of trust can significantly affect care.
Neurontin
One of the more commonly used agents to treat neuropathic syndromes is Neurontin (gabapentin). Disadvantages to the use of gabapentin include the need for dose titration and multiple daily doses. Gabapentin is a good alternative as a second-line agent for patients with DPNP who don’t respond well to or can’t tolerate first-line agents (approved agents or others with evidence: e.g., oxycodone controlled-release, tricyclic antidepressants, Lyrica, Cymbalta).
It is recommended that treatment is begun using a first-line agent. Then each time you evaluate the patient, ask them whether the pain is worse or whether the nature of the pain has changed. They should also be asked if they are experiencing any adverse effects. The agent should be titrated to the maximum tolerated dose with an expected goal of at least 50% pain reduction from baseline. Some pain improvement should be expected within three weeks of therapy initiation. Therefore ascertain that this is followed upon hospital discharge. If no improvement is noted within three weeks, modification of therapy may be warranted.
If the patient derives some (but not optimal) therapy benefit without adverse effects, consider adding a second agent. The agent can be another first-line agent or a second-line agent. Consider rational pharmacotherapy (e.g., avoid additive side effects, consider synergy of agents, avoid drug interactions), and use an agent with a different mechanism of action.
If the patient is receiving no benefit from the current therapy or they are experiencing intolerable adverse effects, consider changing to another agent with a different mechanism of action. If the current agent is Cymbalta, Lyrica, or Neurontin (and the patient has no risk of seizures), taper the drug off over at least one week. When starting a new treatment, always take into consideration the patient’s medical and psychiatric comorbidities, any potential contraindications, and other factors such as the potential for drug interactions, side effects (e.g., weight gain, edema), and/or cost.
Topical therapies may also provide some benefit to the patient with neuropathic pain syndromes (e.g., capsaicin, lidocaine 5% patch). Remember that a specialist can always be consulted for expert advice or for difficult-to-manage patients. TH
Michele B. Kaufman is a drug information specialist and a medical writer based in New York City.
References
- Neurontin (gabapentin) [package insert]. New York: Pfizer Inc;. December 2005.
- Lyrica (pregabalin) [package insert]. New York: Pfizer Inc; March 2006.
- Cymbalta (duloxetine) [package insert]. Indianapolis: Eli Lilly and Company; December 14, 2005.
- The Medical Letter on Drugs and Therapeutics. Duloxetine; Volume 47 (Issue 1215/1216), August 15/29, 2005; 67-68.
- Pregabalin. The Medical Letter on Drugs and Therapeutics. 47(1217) :75-77. Available online at www.medletter.com/restricted/articles/w1217b.pdf. Last accessed on July 28, 2006.
- Facts and Comparisons Updated Monthly. Wolters Kluwer Health: October 2005.
- Thomson Micromedex Healthcare Series: Document Comparison of Pregabalin and Gabapentin. Available at: www.thomsonhc.com/hcs/librarian/PFDefaultActionId/pf.PrintReady. Last accessed June 7, 2006.
- Argoff CE, Backonia M-M, Belgrade ML, et al. Consensus Guidelines: treatment planning and options. Mayo Clin Proc. 2006;81(4):S12-S25.
- Argoff CE, Cole BE, Fishbain DA, et al. Diabetic Peripheral neuropathic pain: clinical and quality-of-life issues. Mayo Clin Proc. 2006;81(4, suppl):S3-S11.
- Belgrade ML, Cole BE, McCarberg BH, et al. Diabetic peripheral neuropathic pain: case studies. Mayo Clin Proc. 2006 Apr;81(4 Suppl):S26-S32.
A number of pharmaceutical agents are FDA-approved to treat different types of neuropathic pain. For example, the most recently approved agents include pregabalin (Lyrica, Pfizer) and duloxetine (Cymbalta, Eli Lilly & Company). Additionally Neurontin (gabapentin, Pfizer) and its generics have been available since the 1990s, and have also been used to treat neuropathic pain (it is specifically FDA-approved to treat post-herpetic neuralgia [PHN]).
Lyrica
In June 2005, Lyrica received FDA-approval for treatment of neuropathic pain associated with diabetic peripheral neuropathy (DPN) and PHN as well as for treatment of adults with partial onset seizures. The maximum recommended dose of Lyrica for DPN is 100mg three times daily (TID). When treating patients with a creatinine clearance (CrCL) of < 60 mL/min, initiate at a lower dose (50 mg TID) because it is primarily renally eliminated. When discontinuing Lyrica, it should be gradually tapered over a minimum of one week. The recommended dose for treatment of PHN is 75–150 mg twice daily (BID), or 50–100 mg TID for patients with a CrCL of at least 60mL/min. The dose should be reduced for patients with a CrCL < 60mL/min. If no pain relief is obtained within two to four weeks at this dose, it may be gradually increased to 600 mg/day (200 mg TID or 300 mg BID); however, side effects may be more severe because they are dose-dependent (e.g., ataxia, dizziness, dry mouth, peripheral edema, somnolence, and weight gain).
Other guidelines for dosing in renally impaired patients should be reviewed in the product labeling. Because Lyrica has been reported to cause euphoria, it has been designated as a controlled substance (C-V). There are no specific drug interactions except with the glitazones (weight gain, fluid retention, or congestive heart failure exacerbation may occur) and the possibility of additive central nervous system (CNS) effects if given with other agents that affect the CNS. Lyrica is available in the following strength capsules: 25, 50, 75, 100, 150, 200, 225, and 300mg. Head-to-head comparisons to similar agents are not available.
Cymbalta
In August 2004, the FDA approved Cymbalta for treatment of DPN and major depressive disorder (MDD). Dosing for treatment of DPN is 60 mg once daily. A lower starting dose may be used in patients with renal impairment. (It should not be used in patients with a CrCL <30 mL/min.) Common adverse effects include nausea, dizziness, somnolence, constipation, dry mouth, and increased sweating. Serum transaminase elevations have also been reported. Because Cymbalta is metabolized by CYP1A2 and CYP2D6, numerous drug interactions may occur. Drug discontinuation should be performed gradually to avoid withdrawal symptoms. Cymbalta capsules are delayed-release and are available in the following strengths: 20, 30, and 60 mg. (They should not be opened or crushed prior to administration).
Diabetic Peripheral Neuropathic Pain
Earlier this year, a Consensus Guideline on the management of diabetic peripheral neuropathic pain (DPNP) was published, the first of its kind. Treatment of DPNP may mirror other peripheral neuropathic pain syndromes, and, therefore, this guideline may assist in managing other similar patients.
A goal of 100% pain relief is ideal but often unrealistic. Many patients will only experience a 30%-50% reduction in pain relief; however, this may enable the patient to return to social activities or work and improve their quality of life. Hospitalists and other members of the healthcare team must keep in mind that the patients’ treatment goals may significantly differ from their own goals of therapy. In managing these complex patients we must bear in mind that complete pain relief may not be attainable. We must also continue to communicate with our patients and provide them with information on what is known and unknown about the mechanisms and treatment of neuropathic pain. By developing and maintaining these patient relationships, our patients will apt to be more satisfied with their treatment, even if they do not have 100% improvement.
In DPNP (there are many patients who may have this and not know that they are diabetic or may be in denial about the degree of their diabetes), it is important for the patient to play an active role in their care (e.g., glycemic control, foot care, analgesic treatment). If treatment plans are not for FDA-approved uses, obtain patient consent. Remember, patients now have access to approved labeling via the internet. If they feel that their healthcare providers are not being “above aboard,” lack of trust can significantly affect care.
Neurontin
One of the more commonly used agents to treat neuropathic syndromes is Neurontin (gabapentin). Disadvantages to the use of gabapentin include the need for dose titration and multiple daily doses. Gabapentin is a good alternative as a second-line agent for patients with DPNP who don’t respond well to or can’t tolerate first-line agents (approved agents or others with evidence: e.g., oxycodone controlled-release, tricyclic antidepressants, Lyrica, Cymbalta).
It is recommended that treatment is begun using a first-line agent. Then each time you evaluate the patient, ask them whether the pain is worse or whether the nature of the pain has changed. They should also be asked if they are experiencing any adverse effects. The agent should be titrated to the maximum tolerated dose with an expected goal of at least 50% pain reduction from baseline. Some pain improvement should be expected within three weeks of therapy initiation. Therefore ascertain that this is followed upon hospital discharge. If no improvement is noted within three weeks, modification of therapy may be warranted.
If the patient derives some (but not optimal) therapy benefit without adverse effects, consider adding a second agent. The agent can be another first-line agent or a second-line agent. Consider rational pharmacotherapy (e.g., avoid additive side effects, consider synergy of agents, avoid drug interactions), and use an agent with a different mechanism of action.
If the patient is receiving no benefit from the current therapy or they are experiencing intolerable adverse effects, consider changing to another agent with a different mechanism of action. If the current agent is Cymbalta, Lyrica, or Neurontin (and the patient has no risk of seizures), taper the drug off over at least one week. When starting a new treatment, always take into consideration the patient’s medical and psychiatric comorbidities, any potential contraindications, and other factors such as the potential for drug interactions, side effects (e.g., weight gain, edema), and/or cost.
Topical therapies may also provide some benefit to the patient with neuropathic pain syndromes (e.g., capsaicin, lidocaine 5% patch). Remember that a specialist can always be consulted for expert advice or for difficult-to-manage patients. TH
Michele B. Kaufman is a drug information specialist and a medical writer based in New York City.
References
- Neurontin (gabapentin) [package insert]. New York: Pfizer Inc;. December 2005.
- Lyrica (pregabalin) [package insert]. New York: Pfizer Inc; March 2006.
- Cymbalta (duloxetine) [package insert]. Indianapolis: Eli Lilly and Company; December 14, 2005.
- The Medical Letter on Drugs and Therapeutics. Duloxetine; Volume 47 (Issue 1215/1216), August 15/29, 2005; 67-68.
- Pregabalin. The Medical Letter on Drugs and Therapeutics. 47(1217) :75-77. Available online at www.medletter.com/restricted/articles/w1217b.pdf. Last accessed on July 28, 2006.
- Facts and Comparisons Updated Monthly. Wolters Kluwer Health: October 2005.
- Thomson Micromedex Healthcare Series: Document Comparison of Pregabalin and Gabapentin. Available at: www.thomsonhc.com/hcs/librarian/PFDefaultActionId/pf.PrintReady. Last accessed June 7, 2006.
- Argoff CE, Backonia M-M, Belgrade ML, et al. Consensus Guidelines: treatment planning and options. Mayo Clin Proc. 2006;81(4):S12-S25.
- Argoff CE, Cole BE, Fishbain DA, et al. Diabetic Peripheral neuropathic pain: clinical and quality-of-life issues. Mayo Clin Proc. 2006;81(4, suppl):S3-S11.
- Belgrade ML, Cole BE, McCarberg BH, et al. Diabetic peripheral neuropathic pain: case studies. Mayo Clin Proc. 2006 Apr;81(4 Suppl):S26-S32.
Caution! Job Opportunities Ahead
Recent studies, published in mass media and professional publications alike, warn of a physician shortage expected to worsen progressively during the next 15 years and to peak around 2020. The predicted scope of the national deficit ranges from 85,000 to 200,000 physicians. Analyses of the causes of the shortage conclude that the rate of population growth will exceed growth in the number of physicians, while demand for physician services continues to expand. The projected shortage of physicians is likely to have the greatest effect on underserved and poorer communities that have historically had the most difficulty recruiting and retaining physicians.
In response, hospitals are implementing, expanding, or refining recruitment and retention programs. Practices are increasingly offering part-time work, flex-time, and job-sharing arrangements. In addition to flexible schedules, physician recruitment packages may include sign-on bonuses, relocation allowances, student loan repayment, income guarantees, favorable loans, and other incentives. Additional incentives are available to physicians willing to practice in federally recognized Health Professional Shortage Areas (HPSA) and to those who practice in specialties governed by HPSA regulations, such as family practice, internal medicine, pediatrics, obstetrics and gynecology, oral surgery, mental health, vision care, and podiatry.
These recruiting arrangements are governed by a variety of laws and regulations. Because of the infinite number of factors affecting any given recruitment arrangement, a comprehensive discussion of all the legal implications in recruitment arrangements is beyond the scope of this article. We will, however, provide an overview of three key legal concepts that physicians should consider when evaluating a hospital’s recruiting package. We’ll address recruiting packages offered by medical practices in a later article.
Stark Law
Unless an express exception applies, federal legislation prohibits physicians from making referrals for “designated health services” to entities with which the physician (or an immediate family member) has a “financial relationship.” The law also prohibits entities from submitting claims for services provided in the course of making a prohibited referral.
The Stark legislation broadly defines “financial relationships” as including direct ownership, indirect ownership, investment interests, and compensation arrangements. Similarly, the law broadly defines “designated health services” to include clinical laboratory, physical therapy, occupational therapy, speech pathology, radiology, radiation therapy, home health, and inpatient and outpatient hospital services. “Designated health services” are not limited purely to services rendered and include the provision of radiation therapy supplies, durable medical equipment and supplies, certain nutrients, prosthetics, orthotics, and prescription drugs. A classic example of an arrangement that would violate the Stark legislation is an orthopedic surgeon who refers patients to a physical therapy facility in which he owns a controlling interest.
In general, physicians should be wary of any relationship that involves referring patients to entities in which they have any financial interest, but there are a number of exceptions within the Stark legislation. The Stark law is a strict liability statute; thus, unless an express exception applies, a violation of the statute subjects the provider and entity to liability. For this article, we are concerned with only one of the exceptions, which applies when hospitals and Federally Qualified Health Centers recruit physicians to their geographic service areas.
Specifically, these entities may offer remuneration to induce physicians to relocate and join the medical staff as long as the recruited physicians are not required to refer patients to the facility and provided that the amount of any physician remuneration does not take into account the volume or value of patient referrals. In other words, although the hospital may recruit a physician, it cannot use the recruiting contract to require the physician to make a certain number of referrals or generate a certain amount of revenue.
It is also important to note that the recruiting exception is designed to promote true recruiting, not simply to entice an established physician in the community to move her practice to a competing hospital. Consequently, the recruiting exception does not apply unless the recruited physician will either move her practice at least 25 miles or generate 75% of her revenues from new patients.
Anti-Kickback Statute
The anti-kickback statute prohibits healthcare providers or entities from knowingly offering or accepting remuneration to induce or reward referrals. Federal regulations create “safe harbors” outlining criteria that, if met, shield providers and entities from anti-kickback liability. The recruitment safe harbor requires a recruited physician leaving an existing medical practice to relocate at least 100 miles away and to generate 85% of new practice revenues from patients not seen at the former practice. Further, the recruited physician must agree to treat Medicare and Medicaid patients.
Internal Revenue Code
Any tax-exempt entity and its physician recruits must carefully structure recruiting arrangements to avoid jeopardizing the entity’s tax-exempt status. Moreover, certain recruitment incentives have tax consequences for an individual recruit.
Tax-exempt entities: Generally, a tax-exempt entity’s earnings may not benefit private individuals. If an improper benefit is found, both the entity and the individual are subject to penalties, including the potential loss of the entity’s tax-exempt status. Thus, physician-recruiting payments by tax-exempt hospitals must fit within IRS requirements.
Specifically, when a tax-exempt hospital recruits a physician to provide service on behalf of the organization, the arrangement must meet an “operational test.” The operational test requires the hospital to account for all of the physician’s services and demonstrate that it is paying reasonable compensation. Consequently, when a tax-exempt hospital recruits a physician to provide services not just for the hospital but also for the surrounding community, it must ensure that all conduct is consistent with the facility’s tax-exempt purpose. Thus, for example, a tax-exempt hospital that has a charitable purpose may be able to justify a recruiting arrangement that allows a physician to provide services that promote the health of the surrounding community.
Ultimately, the IRS is responsible for determining that a tax-exempt hospital is not using its funds solely to promote the physician’s personal interests. Consequently, a tax-exempt hospital should be prepared to demonstrate the reasons the physician was recruited, the need the recruited physician fills, ways in which the recruitment furthers the hospital’s purpose, evidence that the recruiting agreement was negotiated in good faith, and proof that none of the participants in the negotiation suffered from a conflict of interest.
Tax consequences to recruits: Notably, recruiting packages may offer incentives to the recruit that the IRS may consider taxable income. For example, many recruiting arrangements include loans to guarantee a certain level of income and cover the costs associated with starting up a new practice or adding a physician to an existing practice. These loans may be forgiven over time if the recruited physician continues practicing in the community. Generally, proceeds of a loan do not constitute taxable income because the benefit is offset by an obligation to repay. When a loan is subject to forgiveness, however, the forgiven portion may be taxable. Consequently, recruits should evaluate any recruitment incentives in the context of their long-term tax consequences.
Conclusion
Federal law recognizes that communities benefit when hospitals recruit physicians to meet a particular need, but the law does not allow hospitals or physicians to abuse the recruiting relationship.
In evaluating a hospital’s recruiting agreement, physicians should ensure that the agreement does not require them to refer patients to a particular facility, does not calculate their remuneration based upon the number or value of referrals, meets the requirements of relocation or establishment of a new patient base, is consistent with the hospital’s tax status, and does not expose the physician to unintended tax liabilities. TH
O’Rourke works in the Office of University Counsel, Department of Litigation, Denver.
Recent studies, published in mass media and professional publications alike, warn of a physician shortage expected to worsen progressively during the next 15 years and to peak around 2020. The predicted scope of the national deficit ranges from 85,000 to 200,000 physicians. Analyses of the causes of the shortage conclude that the rate of population growth will exceed growth in the number of physicians, while demand for physician services continues to expand. The projected shortage of physicians is likely to have the greatest effect on underserved and poorer communities that have historically had the most difficulty recruiting and retaining physicians.
In response, hospitals are implementing, expanding, or refining recruitment and retention programs. Practices are increasingly offering part-time work, flex-time, and job-sharing arrangements. In addition to flexible schedules, physician recruitment packages may include sign-on bonuses, relocation allowances, student loan repayment, income guarantees, favorable loans, and other incentives. Additional incentives are available to physicians willing to practice in federally recognized Health Professional Shortage Areas (HPSA) and to those who practice in specialties governed by HPSA regulations, such as family practice, internal medicine, pediatrics, obstetrics and gynecology, oral surgery, mental health, vision care, and podiatry.
These recruiting arrangements are governed by a variety of laws and regulations. Because of the infinite number of factors affecting any given recruitment arrangement, a comprehensive discussion of all the legal implications in recruitment arrangements is beyond the scope of this article. We will, however, provide an overview of three key legal concepts that physicians should consider when evaluating a hospital’s recruiting package. We’ll address recruiting packages offered by medical practices in a later article.
Stark Law
Unless an express exception applies, federal legislation prohibits physicians from making referrals for “designated health services” to entities with which the physician (or an immediate family member) has a “financial relationship.” The law also prohibits entities from submitting claims for services provided in the course of making a prohibited referral.
The Stark legislation broadly defines “financial relationships” as including direct ownership, indirect ownership, investment interests, and compensation arrangements. Similarly, the law broadly defines “designated health services” to include clinical laboratory, physical therapy, occupational therapy, speech pathology, radiology, radiation therapy, home health, and inpatient and outpatient hospital services. “Designated health services” are not limited purely to services rendered and include the provision of radiation therapy supplies, durable medical equipment and supplies, certain nutrients, prosthetics, orthotics, and prescription drugs. A classic example of an arrangement that would violate the Stark legislation is an orthopedic surgeon who refers patients to a physical therapy facility in which he owns a controlling interest.
In general, physicians should be wary of any relationship that involves referring patients to entities in which they have any financial interest, but there are a number of exceptions within the Stark legislation. The Stark law is a strict liability statute; thus, unless an express exception applies, a violation of the statute subjects the provider and entity to liability. For this article, we are concerned with only one of the exceptions, which applies when hospitals and Federally Qualified Health Centers recruit physicians to their geographic service areas.
Specifically, these entities may offer remuneration to induce physicians to relocate and join the medical staff as long as the recruited physicians are not required to refer patients to the facility and provided that the amount of any physician remuneration does not take into account the volume or value of patient referrals. In other words, although the hospital may recruit a physician, it cannot use the recruiting contract to require the physician to make a certain number of referrals or generate a certain amount of revenue.
It is also important to note that the recruiting exception is designed to promote true recruiting, not simply to entice an established physician in the community to move her practice to a competing hospital. Consequently, the recruiting exception does not apply unless the recruited physician will either move her practice at least 25 miles or generate 75% of her revenues from new patients.
Anti-Kickback Statute
The anti-kickback statute prohibits healthcare providers or entities from knowingly offering or accepting remuneration to induce or reward referrals. Federal regulations create “safe harbors” outlining criteria that, if met, shield providers and entities from anti-kickback liability. The recruitment safe harbor requires a recruited physician leaving an existing medical practice to relocate at least 100 miles away and to generate 85% of new practice revenues from patients not seen at the former practice. Further, the recruited physician must agree to treat Medicare and Medicaid patients.
Internal Revenue Code
Any tax-exempt entity and its physician recruits must carefully structure recruiting arrangements to avoid jeopardizing the entity’s tax-exempt status. Moreover, certain recruitment incentives have tax consequences for an individual recruit.
Tax-exempt entities: Generally, a tax-exempt entity’s earnings may not benefit private individuals. If an improper benefit is found, both the entity and the individual are subject to penalties, including the potential loss of the entity’s tax-exempt status. Thus, physician-recruiting payments by tax-exempt hospitals must fit within IRS requirements.
Specifically, when a tax-exempt hospital recruits a physician to provide service on behalf of the organization, the arrangement must meet an “operational test.” The operational test requires the hospital to account for all of the physician’s services and demonstrate that it is paying reasonable compensation. Consequently, when a tax-exempt hospital recruits a physician to provide services not just for the hospital but also for the surrounding community, it must ensure that all conduct is consistent with the facility’s tax-exempt purpose. Thus, for example, a tax-exempt hospital that has a charitable purpose may be able to justify a recruiting arrangement that allows a physician to provide services that promote the health of the surrounding community.
Ultimately, the IRS is responsible for determining that a tax-exempt hospital is not using its funds solely to promote the physician’s personal interests. Consequently, a tax-exempt hospital should be prepared to demonstrate the reasons the physician was recruited, the need the recruited physician fills, ways in which the recruitment furthers the hospital’s purpose, evidence that the recruiting agreement was negotiated in good faith, and proof that none of the participants in the negotiation suffered from a conflict of interest.
Tax consequences to recruits: Notably, recruiting packages may offer incentives to the recruit that the IRS may consider taxable income. For example, many recruiting arrangements include loans to guarantee a certain level of income and cover the costs associated with starting up a new practice or adding a physician to an existing practice. These loans may be forgiven over time if the recruited physician continues practicing in the community. Generally, proceeds of a loan do not constitute taxable income because the benefit is offset by an obligation to repay. When a loan is subject to forgiveness, however, the forgiven portion may be taxable. Consequently, recruits should evaluate any recruitment incentives in the context of their long-term tax consequences.
Conclusion
Federal law recognizes that communities benefit when hospitals recruit physicians to meet a particular need, but the law does not allow hospitals or physicians to abuse the recruiting relationship.
In evaluating a hospital’s recruiting agreement, physicians should ensure that the agreement does not require them to refer patients to a particular facility, does not calculate their remuneration based upon the number or value of referrals, meets the requirements of relocation or establishment of a new patient base, is consistent with the hospital’s tax status, and does not expose the physician to unintended tax liabilities. TH
O’Rourke works in the Office of University Counsel, Department of Litigation, Denver.
Recent studies, published in mass media and professional publications alike, warn of a physician shortage expected to worsen progressively during the next 15 years and to peak around 2020. The predicted scope of the national deficit ranges from 85,000 to 200,000 physicians. Analyses of the causes of the shortage conclude that the rate of population growth will exceed growth in the number of physicians, while demand for physician services continues to expand. The projected shortage of physicians is likely to have the greatest effect on underserved and poorer communities that have historically had the most difficulty recruiting and retaining physicians.
In response, hospitals are implementing, expanding, or refining recruitment and retention programs. Practices are increasingly offering part-time work, flex-time, and job-sharing arrangements. In addition to flexible schedules, physician recruitment packages may include sign-on bonuses, relocation allowances, student loan repayment, income guarantees, favorable loans, and other incentives. Additional incentives are available to physicians willing to practice in federally recognized Health Professional Shortage Areas (HPSA) and to those who practice in specialties governed by HPSA regulations, such as family practice, internal medicine, pediatrics, obstetrics and gynecology, oral surgery, mental health, vision care, and podiatry.
These recruiting arrangements are governed by a variety of laws and regulations. Because of the infinite number of factors affecting any given recruitment arrangement, a comprehensive discussion of all the legal implications in recruitment arrangements is beyond the scope of this article. We will, however, provide an overview of three key legal concepts that physicians should consider when evaluating a hospital’s recruiting package. We’ll address recruiting packages offered by medical practices in a later article.
Stark Law
Unless an express exception applies, federal legislation prohibits physicians from making referrals for “designated health services” to entities with which the physician (or an immediate family member) has a “financial relationship.” The law also prohibits entities from submitting claims for services provided in the course of making a prohibited referral.
The Stark legislation broadly defines “financial relationships” as including direct ownership, indirect ownership, investment interests, and compensation arrangements. Similarly, the law broadly defines “designated health services” to include clinical laboratory, physical therapy, occupational therapy, speech pathology, radiology, radiation therapy, home health, and inpatient and outpatient hospital services. “Designated health services” are not limited purely to services rendered and include the provision of radiation therapy supplies, durable medical equipment and supplies, certain nutrients, prosthetics, orthotics, and prescription drugs. A classic example of an arrangement that would violate the Stark legislation is an orthopedic surgeon who refers patients to a physical therapy facility in which he owns a controlling interest.
In general, physicians should be wary of any relationship that involves referring patients to entities in which they have any financial interest, but there are a number of exceptions within the Stark legislation. The Stark law is a strict liability statute; thus, unless an express exception applies, a violation of the statute subjects the provider and entity to liability. For this article, we are concerned with only one of the exceptions, which applies when hospitals and Federally Qualified Health Centers recruit physicians to their geographic service areas.
Specifically, these entities may offer remuneration to induce physicians to relocate and join the medical staff as long as the recruited physicians are not required to refer patients to the facility and provided that the amount of any physician remuneration does not take into account the volume or value of patient referrals. In other words, although the hospital may recruit a physician, it cannot use the recruiting contract to require the physician to make a certain number of referrals or generate a certain amount of revenue.
It is also important to note that the recruiting exception is designed to promote true recruiting, not simply to entice an established physician in the community to move her practice to a competing hospital. Consequently, the recruiting exception does not apply unless the recruited physician will either move her practice at least 25 miles or generate 75% of her revenues from new patients.
Anti-Kickback Statute
The anti-kickback statute prohibits healthcare providers or entities from knowingly offering or accepting remuneration to induce or reward referrals. Federal regulations create “safe harbors” outlining criteria that, if met, shield providers and entities from anti-kickback liability. The recruitment safe harbor requires a recruited physician leaving an existing medical practice to relocate at least 100 miles away and to generate 85% of new practice revenues from patients not seen at the former practice. Further, the recruited physician must agree to treat Medicare and Medicaid patients.
Internal Revenue Code
Any tax-exempt entity and its physician recruits must carefully structure recruiting arrangements to avoid jeopardizing the entity’s tax-exempt status. Moreover, certain recruitment incentives have tax consequences for an individual recruit.
Tax-exempt entities: Generally, a tax-exempt entity’s earnings may not benefit private individuals. If an improper benefit is found, both the entity and the individual are subject to penalties, including the potential loss of the entity’s tax-exempt status. Thus, physician-recruiting payments by tax-exempt hospitals must fit within IRS requirements.
Specifically, when a tax-exempt hospital recruits a physician to provide service on behalf of the organization, the arrangement must meet an “operational test.” The operational test requires the hospital to account for all of the physician’s services and demonstrate that it is paying reasonable compensation. Consequently, when a tax-exempt hospital recruits a physician to provide services not just for the hospital but also for the surrounding community, it must ensure that all conduct is consistent with the facility’s tax-exempt purpose. Thus, for example, a tax-exempt hospital that has a charitable purpose may be able to justify a recruiting arrangement that allows a physician to provide services that promote the health of the surrounding community.
Ultimately, the IRS is responsible for determining that a tax-exempt hospital is not using its funds solely to promote the physician’s personal interests. Consequently, a tax-exempt hospital should be prepared to demonstrate the reasons the physician was recruited, the need the recruited physician fills, ways in which the recruitment furthers the hospital’s purpose, evidence that the recruiting agreement was negotiated in good faith, and proof that none of the participants in the negotiation suffered from a conflict of interest.
Tax consequences to recruits: Notably, recruiting packages may offer incentives to the recruit that the IRS may consider taxable income. For example, many recruiting arrangements include loans to guarantee a certain level of income and cover the costs associated with starting up a new practice or adding a physician to an existing practice. These loans may be forgiven over time if the recruited physician continues practicing in the community. Generally, proceeds of a loan do not constitute taxable income because the benefit is offset by an obligation to repay. When a loan is subject to forgiveness, however, the forgiven portion may be taxable. Consequently, recruits should evaluate any recruitment incentives in the context of their long-term tax consequences.
Conclusion
Federal law recognizes that communities benefit when hospitals recruit physicians to meet a particular need, but the law does not allow hospitals or physicians to abuse the recruiting relationship.
In evaluating a hospital’s recruiting agreement, physicians should ensure that the agreement does not require them to refer patients to a particular facility, does not calculate their remuneration based upon the number or value of referrals, meets the requirements of relocation or establishment of a new patient base, is consistent with the hospital’s tax status, and does not expose the physician to unintended tax liabilities. TH
O’Rourke works in the Office of University Counsel, Department of Litigation, Denver.
Abdominal Pain and Weight Loss
A58-year-old white female presented with an eight-month history of progressive lower abdominal pain and bloating. She experienced intermittent constipation followed by a five-month period of persistent loose, watery diarrhea, a 35-pound weight loss, fatigue, anorexia, and avoidance of food.
Her past medical and surgical history were significant for hypertension, depression, appendectomy, laparoscopic ovarian cystectomy (of benign histology), and cholelithiasis. Her medication list consisted of pindolol and sertraline. Her physical exam was remarkable for abdominal distention, palpable mass, fluid wave, shifting dullness, and hypoactive bowel sounds. There was no tenderness or organomegaly. She had a mild microcytic anemia with no leukocytosis. Liver chemistries and electrolytes were normal. The erythrocyte sedimentation rate was 51. The initial CT scan of the abdomen is shown at right. TH
What is the most likely differential diagnosis?
- Pneumatosis intestinalis;
- Ovarian carcinoma or peritoneal carcinomatosis;
- Sclerosing mesenteritis;
- Spontaneous bacterial peritonitis; or
- Lymphoma.
Discussion
The answer is C: sclerosing mesenteritis (SM). The CT scan shows a bulky heterogeneous mesenteric mass measuring approximately 8.7 x 6 x 10 cm, with a focal, 2-cm calcification at the lateral margin. The mass began at the proximal superior mesenteric artery, extended inferiorly to the top of the pelvis, and encased the body of the pancreas, central mesenteric vessels and the confluence of the portal, splenic, and superior mesenteric veins (SMV). The SMV was poorly visualized and may have been compressed or occluded by the mass. Diffuse abdominal and pelvic ascites also were seen.
Results of a needle biopsy of the mesenteric mass showed fibrous tissue and a mixed population of B cells and T cells consistent with sclerosing mesenteritis, a fibroinflammatory reactive process.
Sclerosing or retractile mesenteritis is an uncommon, idiopathic, nonneoplastic, tumor-like lesion that thickens and shortens the mesentery.1 The condition consists of a pathophysiological spectrum of disease, the classification of which is based on the predominant histological finding on tissue biopsy. Cases in which the predominant findings are fatty degeneration and necrosis are known as the mesenteric lipodystrophy variant; those in which chronic inflammation predominates are known as the mesenteric panniculitis variant; and finally, the predominantly fibrotic form is known as the retractile mesenteritis or mesenteric fibrosis variant.
The presence of some degree of fibrosis, chronic inflammation, and fat necrosis in all three lesions, as well as their common demographic and clinical characteristics, suggest that the three diagnostic groups represent a single clinical entity.2 Sclerosing mesenteritis is used as an umbrella term that encompasses all three histologic variants.
Numerous theories exist to explain the pathogenesis of the condition, most commonly relating to a non-specific reaction to mesenteric injuries such as antecedent surgery or abdominal trauma, or possibly autoimmunity, ischemia/infection, and or paraneoplastic phenomena.
A study of 68 cases of mesenteric panniculitis undertaken by Durst, et al., showed that the age range is diverse, affecting patients from seven to 82 years.3 In another study, Emory, et al., reported an average age of 60.1 years at presentation.2 In both studies there was a slight male preponderance, with a male: female ratio of 1.9:1, and 1.8:1.2,3
In the largest reported experience with patients with sclerosing mesenteritis from a single institution, the majority were male (72%) with an age range of 34-87 years.10,11 Of these patients, the most common presenting complaints are abdominal pain (36%-67%), vomiting (18%-32%), palpable abdominal mass (16%), anorexia (7%-17%), weight loss (14%-45%), constipation (8%-15%), diarrhea (7%-26%), and rectal bleeding (5%). The duration of symptoms varies from 24 hours to two years.3-10 In most cases, the blood chemistry and urinalysis were reported to be normal, although an elevated sedimentation rate has been reported in a minority.3,11
Diagnosis usually depends on imaging studies (most often CT scan) and pathological review of biopsy specimens. Concurrent pathology is found in 18%-25% of cases, including lymphoma (7%), cholelithiasis (4%), cirrhosis of the liver (3%), and abdominal aortic aneurysm (3%).3,10 Other conditions associated with idiopathic fibrosis, such as retroperitoneal fibrosis and sclerosing pancreatitis, have been reported to occur together with SM.4,10
To date, treatment options are guided by anecdotal experience and reports of open label clinical results.10,11 Clinical and experimental studies have suggested a possible hormonal influence on fibrous proliferation in retroperitoneal fibrosis, which led to the discovery of the beneficial effect of anti-estrogenic treatment with tamoxifen.5,6 Clark, et al., had noticed that tamoxifen was successfully used in the treatment of desmoid tumors and then reported its use in two cases of retroperitoneal fibrosis.5 Others have since used tamoxifen in sclerosing mesenteritis, including a relatively large experience from our institution that suggests the combination of tamoxifen with a prednisone taper may be the most beneficial treatment for this condition.7,10,11
Other treatments that have been reported to be of some benefit include combination of corticosteroids and colchicine or azathioprine, although these reports include very small numbers of patients.8,10 Ginsburg, et al., reported their experience with thalidomide in a short term, open-label study in five symptomatic subjects.12 Four patients (80%) experienced an improvement in symptoms, and one achieved complete remission by week four, which was sustained. There was also a decrease in inflammatory markers, although no changes were noted on follow-up CT scans.
The natural history of sclerosing mesenteritis is quite variable, with a few patients undergoing spontaneous remission, many experiencing an indolent course, and others progressing to bowel obstruction or other complications, including chylous ascites, mesenteric venous or arterial occlusion, and malnutrition (often resulting in the need for parenteral nutrition).10,11 There have been fatal cases of this condition reported in the literature; however, Durst, et al., found that of 40 patients undergoing exploratory laparotomy and biopsy, only one patient died as a direct result of the disease after 12 years and several explorations.9-11 In the remaining 39 patients, their symptoms resolved without any further treatment, although in some the abdominal mass was persistently palpable.3. Similarly Emory, et al., found that after following 42 patients for an average of 9.5 years, only three patients had complications that resulted in death, all of which occurred in the postoperative period.2
In our experience, three of 92 patients (3%) died from causes that were thought to be attributable to sclerosing mesenteritis or its treatment.10,11
In summary, sclerosing mesenteritis is a rare disease entity that thickens and shortens the mesentery due to a non-specific fibroinflammatory reaction in the mesentery. Diagnosis relies on CT scanning and tissue biopsy, which shows variable degrees of fibrosis, chronic inflammation, and fat necrosis. Treatment options are based on anecdotal experience only. Tamoxifen, with or without combination therapy with prednisone, colchicine, azathioprine, and thalidomide, appears to be of some benefit. Prognosis is variable, with some patients achieving remission while others die from complications related to disease progression such as bowel obstruction, mesenteric vascular occlusion, and malnutrition; however, many have persistent symptoms that may improve but not resolve with medical therapy. TH
References
- Kelly JK, Hwang WS. Idiopathic retractile (sclerosing) mesenteritis and its differential diagnosis. Am J Surg Pathol. 1989;13(6):513-521.
- Emory TS, Monihan JM, Carr NJ, et al. Sclerosing mesenteritis, mesenteric panniculitis and mesenteric lipodystrophy: a single entity? Am J Surg Pathol. 1997 Apr;21(4):392.
- Durst AL, Freund H, Rosenmann E, et al. Mesenteric panniculitis: review of the literature and presentation of cases. Surgery. 1977;81:203.
- Chew CK, Jarzylo SV, Valberg LS. Idiopathic retroperitoneal fibrosis with protein-losing enteropathy and duodenal obstruction successfully treated with corticosteroids. Can Med Assoc J. 1966;95(23):1183-1188.
- Clark CP, Vanderpool D, Preskitt JT. The response of retroperitoneal fibrosis to tamoxifen. Surgery. 1991 Apr;109(4):502-506.
- Owens LV, Cance WG, Huth JF. Retroperitoneal fibrosis treated with tamoxifen. Am Surg. 1995;61:842-844.
- Venkataramani A, Behling CA, Lynche KD. Sclerosing mesenteritis: an unusual cause of abdominal pain in an HIV-positive patient. Am J Gastroenterol. 1997 Jun;92(6):1059-1060.
- Genereau T, Bellin MF, Wechsler B. Demonstration of efficacy of combining corticosteroids and colchicine in two patients with idiopathic sclerosing mesenteritis. Dig Dis Sci. 1996;41(4):684-688.
- Andersen JA, Rasmussen NR, Pedersen JK. Mesenteric panniculitis: a fatal case. Am J Gastroenterol. 1982 Jul;77(7):523-525.
- Akram S, Pardi DS, Smyrk TC. Sclerosing mesenteritis: The Mayo Clinic Experience. (Clinical features and tx response) Gastroenterology. 2003;A-190.
- Akram S, Pardi DS, Smyrk TC. Effect of tamoxifen on clinical course of sclerosing mesenteritis. Gastroenterology. 2006;130:A-322.
- Ginsburg PM, Ehrenpreis ED. A pilot study of thalidomide for patients with symptomatic mesenteric panniculitis. Aliment Pharmacol Ther. 2002 Dec;16(12):2115-2122.
A58-year-old white female presented with an eight-month history of progressive lower abdominal pain and bloating. She experienced intermittent constipation followed by a five-month period of persistent loose, watery diarrhea, a 35-pound weight loss, fatigue, anorexia, and avoidance of food.
Her past medical and surgical history were significant for hypertension, depression, appendectomy, laparoscopic ovarian cystectomy (of benign histology), and cholelithiasis. Her medication list consisted of pindolol and sertraline. Her physical exam was remarkable for abdominal distention, palpable mass, fluid wave, shifting dullness, and hypoactive bowel sounds. There was no tenderness or organomegaly. She had a mild microcytic anemia with no leukocytosis. Liver chemistries and electrolytes were normal. The erythrocyte sedimentation rate was 51. The initial CT scan of the abdomen is shown at right. TH
What is the most likely differential diagnosis?
- Pneumatosis intestinalis;
- Ovarian carcinoma or peritoneal carcinomatosis;
- Sclerosing mesenteritis;
- Spontaneous bacterial peritonitis; or
- Lymphoma.
Discussion
The answer is C: sclerosing mesenteritis (SM). The CT scan shows a bulky heterogeneous mesenteric mass measuring approximately 8.7 x 6 x 10 cm, with a focal, 2-cm calcification at the lateral margin. The mass began at the proximal superior mesenteric artery, extended inferiorly to the top of the pelvis, and encased the body of the pancreas, central mesenteric vessels and the confluence of the portal, splenic, and superior mesenteric veins (SMV). The SMV was poorly visualized and may have been compressed or occluded by the mass. Diffuse abdominal and pelvic ascites also were seen.
Results of a needle biopsy of the mesenteric mass showed fibrous tissue and a mixed population of B cells and T cells consistent with sclerosing mesenteritis, a fibroinflammatory reactive process.
Sclerosing or retractile mesenteritis is an uncommon, idiopathic, nonneoplastic, tumor-like lesion that thickens and shortens the mesentery.1 The condition consists of a pathophysiological spectrum of disease, the classification of which is based on the predominant histological finding on tissue biopsy. Cases in which the predominant findings are fatty degeneration and necrosis are known as the mesenteric lipodystrophy variant; those in which chronic inflammation predominates are known as the mesenteric panniculitis variant; and finally, the predominantly fibrotic form is known as the retractile mesenteritis or mesenteric fibrosis variant.
The presence of some degree of fibrosis, chronic inflammation, and fat necrosis in all three lesions, as well as their common demographic and clinical characteristics, suggest that the three diagnostic groups represent a single clinical entity.2 Sclerosing mesenteritis is used as an umbrella term that encompasses all three histologic variants.
Numerous theories exist to explain the pathogenesis of the condition, most commonly relating to a non-specific reaction to mesenteric injuries such as antecedent surgery or abdominal trauma, or possibly autoimmunity, ischemia/infection, and or paraneoplastic phenomena.
A study of 68 cases of mesenteric panniculitis undertaken by Durst, et al., showed that the age range is diverse, affecting patients from seven to 82 years.3 In another study, Emory, et al., reported an average age of 60.1 years at presentation.2 In both studies there was a slight male preponderance, with a male: female ratio of 1.9:1, and 1.8:1.2,3
In the largest reported experience with patients with sclerosing mesenteritis from a single institution, the majority were male (72%) with an age range of 34-87 years.10,11 Of these patients, the most common presenting complaints are abdominal pain (36%-67%), vomiting (18%-32%), palpable abdominal mass (16%), anorexia (7%-17%), weight loss (14%-45%), constipation (8%-15%), diarrhea (7%-26%), and rectal bleeding (5%). The duration of symptoms varies from 24 hours to two years.3-10 In most cases, the blood chemistry and urinalysis were reported to be normal, although an elevated sedimentation rate has been reported in a minority.3,11
Diagnosis usually depends on imaging studies (most often CT scan) and pathological review of biopsy specimens. Concurrent pathology is found in 18%-25% of cases, including lymphoma (7%), cholelithiasis (4%), cirrhosis of the liver (3%), and abdominal aortic aneurysm (3%).3,10 Other conditions associated with idiopathic fibrosis, such as retroperitoneal fibrosis and sclerosing pancreatitis, have been reported to occur together with SM.4,10
To date, treatment options are guided by anecdotal experience and reports of open label clinical results.10,11 Clinical and experimental studies have suggested a possible hormonal influence on fibrous proliferation in retroperitoneal fibrosis, which led to the discovery of the beneficial effect of anti-estrogenic treatment with tamoxifen.5,6 Clark, et al., had noticed that tamoxifen was successfully used in the treatment of desmoid tumors and then reported its use in two cases of retroperitoneal fibrosis.5 Others have since used tamoxifen in sclerosing mesenteritis, including a relatively large experience from our institution that suggests the combination of tamoxifen with a prednisone taper may be the most beneficial treatment for this condition.7,10,11
Other treatments that have been reported to be of some benefit include combination of corticosteroids and colchicine or azathioprine, although these reports include very small numbers of patients.8,10 Ginsburg, et al., reported their experience with thalidomide in a short term, open-label study in five symptomatic subjects.12 Four patients (80%) experienced an improvement in symptoms, and one achieved complete remission by week four, which was sustained. There was also a decrease in inflammatory markers, although no changes were noted on follow-up CT scans.
The natural history of sclerosing mesenteritis is quite variable, with a few patients undergoing spontaneous remission, many experiencing an indolent course, and others progressing to bowel obstruction or other complications, including chylous ascites, mesenteric venous or arterial occlusion, and malnutrition (often resulting in the need for parenteral nutrition).10,11 There have been fatal cases of this condition reported in the literature; however, Durst, et al., found that of 40 patients undergoing exploratory laparotomy and biopsy, only one patient died as a direct result of the disease after 12 years and several explorations.9-11 In the remaining 39 patients, their symptoms resolved without any further treatment, although in some the abdominal mass was persistently palpable.3. Similarly Emory, et al., found that after following 42 patients for an average of 9.5 years, only three patients had complications that resulted in death, all of which occurred in the postoperative period.2
In our experience, three of 92 patients (3%) died from causes that were thought to be attributable to sclerosing mesenteritis or its treatment.10,11
In summary, sclerosing mesenteritis is a rare disease entity that thickens and shortens the mesentery due to a non-specific fibroinflammatory reaction in the mesentery. Diagnosis relies on CT scanning and tissue biopsy, which shows variable degrees of fibrosis, chronic inflammation, and fat necrosis. Treatment options are based on anecdotal experience only. Tamoxifen, with or without combination therapy with prednisone, colchicine, azathioprine, and thalidomide, appears to be of some benefit. Prognosis is variable, with some patients achieving remission while others die from complications related to disease progression such as bowel obstruction, mesenteric vascular occlusion, and malnutrition; however, many have persistent symptoms that may improve but not resolve with medical therapy. TH
References
- Kelly JK, Hwang WS. Idiopathic retractile (sclerosing) mesenteritis and its differential diagnosis. Am J Surg Pathol. 1989;13(6):513-521.
- Emory TS, Monihan JM, Carr NJ, et al. Sclerosing mesenteritis, mesenteric panniculitis and mesenteric lipodystrophy: a single entity? Am J Surg Pathol. 1997 Apr;21(4):392.
- Durst AL, Freund H, Rosenmann E, et al. Mesenteric panniculitis: review of the literature and presentation of cases. Surgery. 1977;81:203.
- Chew CK, Jarzylo SV, Valberg LS. Idiopathic retroperitoneal fibrosis with protein-losing enteropathy and duodenal obstruction successfully treated with corticosteroids. Can Med Assoc J. 1966;95(23):1183-1188.
- Clark CP, Vanderpool D, Preskitt JT. The response of retroperitoneal fibrosis to tamoxifen. Surgery. 1991 Apr;109(4):502-506.
- Owens LV, Cance WG, Huth JF. Retroperitoneal fibrosis treated with tamoxifen. Am Surg. 1995;61:842-844.
- Venkataramani A, Behling CA, Lynche KD. Sclerosing mesenteritis: an unusual cause of abdominal pain in an HIV-positive patient. Am J Gastroenterol. 1997 Jun;92(6):1059-1060.
- Genereau T, Bellin MF, Wechsler B. Demonstration of efficacy of combining corticosteroids and colchicine in two patients with idiopathic sclerosing mesenteritis. Dig Dis Sci. 1996;41(4):684-688.
- Andersen JA, Rasmussen NR, Pedersen JK. Mesenteric panniculitis: a fatal case. Am J Gastroenterol. 1982 Jul;77(7):523-525.
- Akram S, Pardi DS, Smyrk TC. Sclerosing mesenteritis: The Mayo Clinic Experience. (Clinical features and tx response) Gastroenterology. 2003;A-190.
- Akram S, Pardi DS, Smyrk TC. Effect of tamoxifen on clinical course of sclerosing mesenteritis. Gastroenterology. 2006;130:A-322.
- Ginsburg PM, Ehrenpreis ED. A pilot study of thalidomide for patients with symptomatic mesenteric panniculitis. Aliment Pharmacol Ther. 2002 Dec;16(12):2115-2122.
A58-year-old white female presented with an eight-month history of progressive lower abdominal pain and bloating. She experienced intermittent constipation followed by a five-month period of persistent loose, watery diarrhea, a 35-pound weight loss, fatigue, anorexia, and avoidance of food.
Her past medical and surgical history were significant for hypertension, depression, appendectomy, laparoscopic ovarian cystectomy (of benign histology), and cholelithiasis. Her medication list consisted of pindolol and sertraline. Her physical exam was remarkable for abdominal distention, palpable mass, fluid wave, shifting dullness, and hypoactive bowel sounds. There was no tenderness or organomegaly. She had a mild microcytic anemia with no leukocytosis. Liver chemistries and electrolytes were normal. The erythrocyte sedimentation rate was 51. The initial CT scan of the abdomen is shown at right. TH
What is the most likely differential diagnosis?
- Pneumatosis intestinalis;
- Ovarian carcinoma or peritoneal carcinomatosis;
- Sclerosing mesenteritis;
- Spontaneous bacterial peritonitis; or
- Lymphoma.
Discussion
The answer is C: sclerosing mesenteritis (SM). The CT scan shows a bulky heterogeneous mesenteric mass measuring approximately 8.7 x 6 x 10 cm, with a focal, 2-cm calcification at the lateral margin. The mass began at the proximal superior mesenteric artery, extended inferiorly to the top of the pelvis, and encased the body of the pancreas, central mesenteric vessels and the confluence of the portal, splenic, and superior mesenteric veins (SMV). The SMV was poorly visualized and may have been compressed or occluded by the mass. Diffuse abdominal and pelvic ascites also were seen.
Results of a needle biopsy of the mesenteric mass showed fibrous tissue and a mixed population of B cells and T cells consistent with sclerosing mesenteritis, a fibroinflammatory reactive process.
Sclerosing or retractile mesenteritis is an uncommon, idiopathic, nonneoplastic, tumor-like lesion that thickens and shortens the mesentery.1 The condition consists of a pathophysiological spectrum of disease, the classification of which is based on the predominant histological finding on tissue biopsy. Cases in which the predominant findings are fatty degeneration and necrosis are known as the mesenteric lipodystrophy variant; those in which chronic inflammation predominates are known as the mesenteric panniculitis variant; and finally, the predominantly fibrotic form is known as the retractile mesenteritis or mesenteric fibrosis variant.
The presence of some degree of fibrosis, chronic inflammation, and fat necrosis in all three lesions, as well as their common demographic and clinical characteristics, suggest that the three diagnostic groups represent a single clinical entity.2 Sclerosing mesenteritis is used as an umbrella term that encompasses all three histologic variants.
Numerous theories exist to explain the pathogenesis of the condition, most commonly relating to a non-specific reaction to mesenteric injuries such as antecedent surgery or abdominal trauma, or possibly autoimmunity, ischemia/infection, and or paraneoplastic phenomena.
A study of 68 cases of mesenteric panniculitis undertaken by Durst, et al., showed that the age range is diverse, affecting patients from seven to 82 years.3 In another study, Emory, et al., reported an average age of 60.1 years at presentation.2 In both studies there was a slight male preponderance, with a male: female ratio of 1.9:1, and 1.8:1.2,3
In the largest reported experience with patients with sclerosing mesenteritis from a single institution, the majority were male (72%) with an age range of 34-87 years.10,11 Of these patients, the most common presenting complaints are abdominal pain (36%-67%), vomiting (18%-32%), palpable abdominal mass (16%), anorexia (7%-17%), weight loss (14%-45%), constipation (8%-15%), diarrhea (7%-26%), and rectal bleeding (5%). The duration of symptoms varies from 24 hours to two years.3-10 In most cases, the blood chemistry and urinalysis were reported to be normal, although an elevated sedimentation rate has been reported in a minority.3,11
Diagnosis usually depends on imaging studies (most often CT scan) and pathological review of biopsy specimens. Concurrent pathology is found in 18%-25% of cases, including lymphoma (7%), cholelithiasis (4%), cirrhosis of the liver (3%), and abdominal aortic aneurysm (3%).3,10 Other conditions associated with idiopathic fibrosis, such as retroperitoneal fibrosis and sclerosing pancreatitis, have been reported to occur together with SM.4,10
To date, treatment options are guided by anecdotal experience and reports of open label clinical results.10,11 Clinical and experimental studies have suggested a possible hormonal influence on fibrous proliferation in retroperitoneal fibrosis, which led to the discovery of the beneficial effect of anti-estrogenic treatment with tamoxifen.5,6 Clark, et al., had noticed that tamoxifen was successfully used in the treatment of desmoid tumors and then reported its use in two cases of retroperitoneal fibrosis.5 Others have since used tamoxifen in sclerosing mesenteritis, including a relatively large experience from our institution that suggests the combination of tamoxifen with a prednisone taper may be the most beneficial treatment for this condition.7,10,11
Other treatments that have been reported to be of some benefit include combination of corticosteroids and colchicine or azathioprine, although these reports include very small numbers of patients.8,10 Ginsburg, et al., reported their experience with thalidomide in a short term, open-label study in five symptomatic subjects.12 Four patients (80%) experienced an improvement in symptoms, and one achieved complete remission by week four, which was sustained. There was also a decrease in inflammatory markers, although no changes were noted on follow-up CT scans.
The natural history of sclerosing mesenteritis is quite variable, with a few patients undergoing spontaneous remission, many experiencing an indolent course, and others progressing to bowel obstruction or other complications, including chylous ascites, mesenteric venous or arterial occlusion, and malnutrition (often resulting in the need for parenteral nutrition).10,11 There have been fatal cases of this condition reported in the literature; however, Durst, et al., found that of 40 patients undergoing exploratory laparotomy and biopsy, only one patient died as a direct result of the disease after 12 years and several explorations.9-11 In the remaining 39 patients, their symptoms resolved without any further treatment, although in some the abdominal mass was persistently palpable.3. Similarly Emory, et al., found that after following 42 patients for an average of 9.5 years, only three patients had complications that resulted in death, all of which occurred in the postoperative period.2
In our experience, three of 92 patients (3%) died from causes that were thought to be attributable to sclerosing mesenteritis or its treatment.10,11
In summary, sclerosing mesenteritis is a rare disease entity that thickens and shortens the mesentery due to a non-specific fibroinflammatory reaction in the mesentery. Diagnosis relies on CT scanning and tissue biopsy, which shows variable degrees of fibrosis, chronic inflammation, and fat necrosis. Treatment options are based on anecdotal experience only. Tamoxifen, with or without combination therapy with prednisone, colchicine, azathioprine, and thalidomide, appears to be of some benefit. Prognosis is variable, with some patients achieving remission while others die from complications related to disease progression such as bowel obstruction, mesenteric vascular occlusion, and malnutrition; however, many have persistent symptoms that may improve but not resolve with medical therapy. TH
References
- Kelly JK, Hwang WS. Idiopathic retractile (sclerosing) mesenteritis and its differential diagnosis. Am J Surg Pathol. 1989;13(6):513-521.
- Emory TS, Monihan JM, Carr NJ, et al. Sclerosing mesenteritis, mesenteric panniculitis and mesenteric lipodystrophy: a single entity? Am J Surg Pathol. 1997 Apr;21(4):392.
- Durst AL, Freund H, Rosenmann E, et al. Mesenteric panniculitis: review of the literature and presentation of cases. Surgery. 1977;81:203.
- Chew CK, Jarzylo SV, Valberg LS. Idiopathic retroperitoneal fibrosis with protein-losing enteropathy and duodenal obstruction successfully treated with corticosteroids. Can Med Assoc J. 1966;95(23):1183-1188.
- Clark CP, Vanderpool D, Preskitt JT. The response of retroperitoneal fibrosis to tamoxifen. Surgery. 1991 Apr;109(4):502-506.
- Owens LV, Cance WG, Huth JF. Retroperitoneal fibrosis treated with tamoxifen. Am Surg. 1995;61:842-844.
- Venkataramani A, Behling CA, Lynche KD. Sclerosing mesenteritis: an unusual cause of abdominal pain in an HIV-positive patient. Am J Gastroenterol. 1997 Jun;92(6):1059-1060.
- Genereau T, Bellin MF, Wechsler B. Demonstration of efficacy of combining corticosteroids and colchicine in two patients with idiopathic sclerosing mesenteritis. Dig Dis Sci. 1996;41(4):684-688.
- Andersen JA, Rasmussen NR, Pedersen JK. Mesenteric panniculitis: a fatal case. Am J Gastroenterol. 1982 Jul;77(7):523-525.
- Akram S, Pardi DS, Smyrk TC. Sclerosing mesenteritis: The Mayo Clinic Experience. (Clinical features and tx response) Gastroenterology. 2003;A-190.
- Akram S, Pardi DS, Smyrk TC. Effect of tamoxifen on clinical course of sclerosing mesenteritis. Gastroenterology. 2006;130:A-322.
- Ginsburg PM, Ehrenpreis ED. A pilot study of thalidomide for patients with symptomatic mesenteric panniculitis. Aliment Pharmacol Ther. 2002 Dec;16(12):2115-2122.
The Hospitalist Generation
If hospital medicine had to be associated with a single generation, it would most likely be generation X. According to SHM’s recently released 2005-2006 “Survey of Hospitalist Productivity and Compensation,” the average age of hospitalists is 37—the current average age of generation X. But more than the same life stage, this generation shares common characteristics, perspectives, and habits that seem consistent with hospitalists of all ages.
Meet Generation X
Generation X is the term for the generation born between 1965 and 1976. Because they were influenced by the same world events and social trends, this generation (as all generations) brings its own traits and values to the workplace. “The career hallmarks of this generation include their independence and enterprising desire to make things happen,” says Devon Scheef, a partner in The Learning Café ([email protected]), a consulting firm that specializes in helping managers overcome generational differences.
According to The Learning Café, the 51 million members of generation X grew up in a much different world than previous generations. Divorce and two-income families created latchkey kids out of many in this generation, leading to traits of independence, resilience, and adaptability. Members of generation X feel strongly that they don't need someone looking over their shoulders.
This independence can make hospital medicine especially appealing to young physicians because they can often structure their daily work to suit themselves.
When Work Ethics Collide
Generation X and baby boomer workers most often butt heads over differing work ethics. This is true across all industries, including the medical field—in hospitals and other healthcare settings. Baby boomers tend to put in long hours and devote themselves to their work. “They [baby boomers] work hard—maybe too hard,” says Diane Thielfoldt, partner in The Learning Café. “This is the generation that increased our workweek from 40 hours to 70 or 80 hours.” And baby boomers often expect this level of dedication from their colleagues.
Generation Xers are not interested in working these hours; they do not equate long hours with job efficiency. One of the big draws of hospital medicine is the flexible schedule; a young physician can work a set schedule, such as seven-on, seven-off, and know that when she’s not working, she’s free to do what she wants.
Of generation X physicians, Lawrence G. Smith, FACP, chief academic officer, North Shore-Long Island Jewish Health System, says, “Led by women, this generation of [medical] students will work fewer hours and demand flexible employment opportunities.”1
Young physicians who choose private practice or other specialties may find it difficult to fight the work ethic expectations of older physicians and administrators. “Aging boomers—a terrible term for those in the 46- to 55-year-old bracket—have really run into a wall of work-life balance,” Scheef points out. “They are scaling back on work and looking ahead to retirement. This is hard for this go-go generation. Many are looking to the younger generation—we hear this in healthcare, particularly—to lighten their workload.”
How Generation X Works
Contrary to what many baby boomer bosses may think, members of generation X are terrific employees. They simply have their own way of getting things done.
“Gen Xers are in a new stage of life now, in their mid- to late 30s, and we’re seeing some interesting trends,” says Thielfoldt. “Gen Xers have an entrepreneurial spirit. This trend has become stronger as the generation has gotten older, which is surprising.” Some young hospitalists have taken this trend to the extreme, founding their own hospital medicine groups; others build their careers by creating and running new projects and committees. “They seem very oriented to ownership and accountability to define, create, and implement in their careers,” continues Thielfoldt. “Gen Xers are driven out of organizations when asked to focus on just one piece of the process.” Again, this fits in with the hospitalist personality.
At the same time, generation X values learning new skills or specialties. “As a generation, they tend to be very portfolio-oriented, in any industry,” says Thielfoldt. “They want to build a skills portfolio as they would a financial portfolio. They want expertise and new skills. This is more important to them than how much or how little time they work.”
Generation X may also find hospital medicine to be the perfect job because the management style suits them so well. “Gen Xers are very sensitive to micromanaging,” says Scheef. “And now that they have the confidence of experience, [the negative reaction to micromanaging by a supervisor] is worse. Instead of traditional managing, they are very receptive to expert coaching or formal mentoring. This can tie in really well with helping them develop that skills portfolio.”
Changes Are Coming
As more baby boomers retire and generation X becomes firmly established, healthcare and other industries may change the way they operate. “Gen Xers have growing families now. They have come to the work-life balance issue much earlier than any other generation,” says Thielfoldt. “And unlike previous generations, their top priority is spending time with their children. That sounds crazy, right? Boomers have doted on their children—but they spend their time at work, especially fathers. Gen X fathers spend more time on average with their kids than boomers did at the same age.” The eventual outcome: “Employers need to respect this as a top priority and provide flexible scheduling to allow it.”
Many hospital medicine programs are doing just that, searching for ways to adequately cover the patient census and workload and keep hospitalists happy.
Dr. Smith agrees that change must come to healthcare: “The future environment … must recognize that physician well-being and balance in life is a valid and important concern and does not negate the attainment of professionalism. It must reward excellence, not endurance.”1 TH
Jane Jerrard regularly writes “Career Development.”
References
- Smith, LG. Medical professionalism and the generation gap. Am J Med. 2005 Apr;118(4):439-442.
If hospital medicine had to be associated with a single generation, it would most likely be generation X. According to SHM’s recently released 2005-2006 “Survey of Hospitalist Productivity and Compensation,” the average age of hospitalists is 37—the current average age of generation X. But more than the same life stage, this generation shares common characteristics, perspectives, and habits that seem consistent with hospitalists of all ages.
Meet Generation X
Generation X is the term for the generation born between 1965 and 1976. Because they were influenced by the same world events and social trends, this generation (as all generations) brings its own traits and values to the workplace. “The career hallmarks of this generation include their independence and enterprising desire to make things happen,” says Devon Scheef, a partner in The Learning Café ([email protected]), a consulting firm that specializes in helping managers overcome generational differences.
According to The Learning Café, the 51 million members of generation X grew up in a much different world than previous generations. Divorce and two-income families created latchkey kids out of many in this generation, leading to traits of independence, resilience, and adaptability. Members of generation X feel strongly that they don't need someone looking over their shoulders.
This independence can make hospital medicine especially appealing to young physicians because they can often structure their daily work to suit themselves.
When Work Ethics Collide
Generation X and baby boomer workers most often butt heads over differing work ethics. This is true across all industries, including the medical field—in hospitals and other healthcare settings. Baby boomers tend to put in long hours and devote themselves to their work. “They [baby boomers] work hard—maybe too hard,” says Diane Thielfoldt, partner in The Learning Café. “This is the generation that increased our workweek from 40 hours to 70 or 80 hours.” And baby boomers often expect this level of dedication from their colleagues.
Generation Xers are not interested in working these hours; they do not equate long hours with job efficiency. One of the big draws of hospital medicine is the flexible schedule; a young physician can work a set schedule, such as seven-on, seven-off, and know that when she’s not working, she’s free to do what she wants.
Of generation X physicians, Lawrence G. Smith, FACP, chief academic officer, North Shore-Long Island Jewish Health System, says, “Led by women, this generation of [medical] students will work fewer hours and demand flexible employment opportunities.”1
Young physicians who choose private practice or other specialties may find it difficult to fight the work ethic expectations of older physicians and administrators. “Aging boomers—a terrible term for those in the 46- to 55-year-old bracket—have really run into a wall of work-life balance,” Scheef points out. “They are scaling back on work and looking ahead to retirement. This is hard for this go-go generation. Many are looking to the younger generation—we hear this in healthcare, particularly—to lighten their workload.”
How Generation X Works
Contrary to what many baby boomer bosses may think, members of generation X are terrific employees. They simply have their own way of getting things done.
“Gen Xers are in a new stage of life now, in their mid- to late 30s, and we’re seeing some interesting trends,” says Thielfoldt. “Gen Xers have an entrepreneurial spirit. This trend has become stronger as the generation has gotten older, which is surprising.” Some young hospitalists have taken this trend to the extreme, founding their own hospital medicine groups; others build their careers by creating and running new projects and committees. “They seem very oriented to ownership and accountability to define, create, and implement in their careers,” continues Thielfoldt. “Gen Xers are driven out of organizations when asked to focus on just one piece of the process.” Again, this fits in with the hospitalist personality.
At the same time, generation X values learning new skills or specialties. “As a generation, they tend to be very portfolio-oriented, in any industry,” says Thielfoldt. “They want to build a skills portfolio as they would a financial portfolio. They want expertise and new skills. This is more important to them than how much or how little time they work.”
Generation X may also find hospital medicine to be the perfect job because the management style suits them so well. “Gen Xers are very sensitive to micromanaging,” says Scheef. “And now that they have the confidence of experience, [the negative reaction to micromanaging by a supervisor] is worse. Instead of traditional managing, they are very receptive to expert coaching or formal mentoring. This can tie in really well with helping them develop that skills portfolio.”
Changes Are Coming
As more baby boomers retire and generation X becomes firmly established, healthcare and other industries may change the way they operate. “Gen Xers have growing families now. They have come to the work-life balance issue much earlier than any other generation,” says Thielfoldt. “And unlike previous generations, their top priority is spending time with their children. That sounds crazy, right? Boomers have doted on their children—but they spend their time at work, especially fathers. Gen X fathers spend more time on average with their kids than boomers did at the same age.” The eventual outcome: “Employers need to respect this as a top priority and provide flexible scheduling to allow it.”
Many hospital medicine programs are doing just that, searching for ways to adequately cover the patient census and workload and keep hospitalists happy.
Dr. Smith agrees that change must come to healthcare: “The future environment … must recognize that physician well-being and balance in life is a valid and important concern and does not negate the attainment of professionalism. It must reward excellence, not endurance.”1 TH
Jane Jerrard regularly writes “Career Development.”
References
- Smith, LG. Medical professionalism and the generation gap. Am J Med. 2005 Apr;118(4):439-442.
If hospital medicine had to be associated with a single generation, it would most likely be generation X. According to SHM’s recently released 2005-2006 “Survey of Hospitalist Productivity and Compensation,” the average age of hospitalists is 37—the current average age of generation X. But more than the same life stage, this generation shares common characteristics, perspectives, and habits that seem consistent with hospitalists of all ages.
Meet Generation X
Generation X is the term for the generation born between 1965 and 1976. Because they were influenced by the same world events and social trends, this generation (as all generations) brings its own traits and values to the workplace. “The career hallmarks of this generation include their independence and enterprising desire to make things happen,” says Devon Scheef, a partner in The Learning Café ([email protected]), a consulting firm that specializes in helping managers overcome generational differences.
According to The Learning Café, the 51 million members of generation X grew up in a much different world than previous generations. Divorce and two-income families created latchkey kids out of many in this generation, leading to traits of independence, resilience, and adaptability. Members of generation X feel strongly that they don't need someone looking over their shoulders.
This independence can make hospital medicine especially appealing to young physicians because they can often structure their daily work to suit themselves.
When Work Ethics Collide
Generation X and baby boomer workers most often butt heads over differing work ethics. This is true across all industries, including the medical field—in hospitals and other healthcare settings. Baby boomers tend to put in long hours and devote themselves to their work. “They [baby boomers] work hard—maybe too hard,” says Diane Thielfoldt, partner in The Learning Café. “This is the generation that increased our workweek from 40 hours to 70 or 80 hours.” And baby boomers often expect this level of dedication from their colleagues.
Generation Xers are not interested in working these hours; they do not equate long hours with job efficiency. One of the big draws of hospital medicine is the flexible schedule; a young physician can work a set schedule, such as seven-on, seven-off, and know that when she’s not working, she’s free to do what she wants.
Of generation X physicians, Lawrence G. Smith, FACP, chief academic officer, North Shore-Long Island Jewish Health System, says, “Led by women, this generation of [medical] students will work fewer hours and demand flexible employment opportunities.”1
Young physicians who choose private practice or other specialties may find it difficult to fight the work ethic expectations of older physicians and administrators. “Aging boomers—a terrible term for those in the 46- to 55-year-old bracket—have really run into a wall of work-life balance,” Scheef points out. “They are scaling back on work and looking ahead to retirement. This is hard for this go-go generation. Many are looking to the younger generation—we hear this in healthcare, particularly—to lighten their workload.”
How Generation X Works
Contrary to what many baby boomer bosses may think, members of generation X are terrific employees. They simply have their own way of getting things done.
“Gen Xers are in a new stage of life now, in their mid- to late 30s, and we’re seeing some interesting trends,” says Thielfoldt. “Gen Xers have an entrepreneurial spirit. This trend has become stronger as the generation has gotten older, which is surprising.” Some young hospitalists have taken this trend to the extreme, founding their own hospital medicine groups; others build their careers by creating and running new projects and committees. “They seem very oriented to ownership and accountability to define, create, and implement in their careers,” continues Thielfoldt. “Gen Xers are driven out of organizations when asked to focus on just one piece of the process.” Again, this fits in with the hospitalist personality.
At the same time, generation X values learning new skills or specialties. “As a generation, they tend to be very portfolio-oriented, in any industry,” says Thielfoldt. “They want to build a skills portfolio as they would a financial portfolio. They want expertise and new skills. This is more important to them than how much or how little time they work.”
Generation X may also find hospital medicine to be the perfect job because the management style suits them so well. “Gen Xers are very sensitive to micromanaging,” says Scheef. “And now that they have the confidence of experience, [the negative reaction to micromanaging by a supervisor] is worse. Instead of traditional managing, they are very receptive to expert coaching or formal mentoring. This can tie in really well with helping them develop that skills portfolio.”
Changes Are Coming
As more baby boomers retire and generation X becomes firmly established, healthcare and other industries may change the way they operate. “Gen Xers have growing families now. They have come to the work-life balance issue much earlier than any other generation,” says Thielfoldt. “And unlike previous generations, their top priority is spending time with their children. That sounds crazy, right? Boomers have doted on their children—but they spend their time at work, especially fathers. Gen X fathers spend more time on average with their kids than boomers did at the same age.” The eventual outcome: “Employers need to respect this as a top priority and provide flexible scheduling to allow it.”
Many hospital medicine programs are doing just that, searching for ways to adequately cover the patient census and workload and keep hospitalists happy.
Dr. Smith agrees that change must come to healthcare: “The future environment … must recognize that physician well-being and balance in life is a valid and important concern and does not negate the attainment of professionalism. It must reward excellence, not endurance.”1 TH
Jane Jerrard regularly writes “Career Development.”
References
- Smith, LG. Medical professionalism and the generation gap. Am J Med. 2005 Apr;118(4):439-442.
SHM Shapes Pay for Performance
Federal officials are increasingly embracing pay for performance (P4P) in an effort to promote high-quality, cost effective care in government health programs. As the Centers for Medicaid and Medicare Services (CMS) and Congress move forward to implement this concept, SHM is working to ensure that the views of hospitalists are represented in this important debate.
More than 100 P4P programs are already up and running in the private sector in an attempt to reward quality healthcare by setting different payment levels for providers based on how well they meet benchmarks of quality and efficiency. CMS is testing the feasibility of applying this concept to the Medicare program through a number of initiatives.
SHM’s Public Policy and Hospital Quality and Patient Safety committees have been involved in evaluating CMS’ Physician Voluntary Reporting Program (PVRP), launched earlier this year and widely believed to be the precursor to an eventual P4P program for physicians’ services. Under this initiative, physicians are encouraged to submit quality data on a “starter set” of 16 evidence-based measures for certain primary care, surgery, nephrology, and emergency medical services. Physicians who participate in the program receive confidential reports on their performance.
SHM has recommended that hospitalists register their intent to report and begin reporting on relevant performance measures to the extent practicable as a way of becoming more familiar with the program. Because these initial 16 measures have only limited relevance to services billed by hospitalists, SHM is engaged in discussions with lawmakers and their staffs, CMS officials, and consensus organizations involved in developing quality measures, to expand the program’s scope.
As part of Advocacy Day on May 3, some 70 SHM members met with legislators and their staffs and conveyed SHM’s support for initiatives like the PVRP that seek to measure resource use and improve quality, to attain better value for the Medicare program. (See coverage in The Hospitalist SHM Meeting Reporter, July 2006, p. 1.) Participants also educated lawmakers on the role of hospitalists in helping their institutions meet quality reporting requirements mandated under the Medicare Modernization Act, which ties annual hospital payment updates to the submission of performance data for 10 quality measures. The participants also expressed SHM’s interest in working with CMS on demonstration projects that assess the contributions of hospital medicine programs to improved patient care and more efficient management of hospital resources.
In addition to Congress and CMS, non-governmental groups such as the National Quality Forum (NQF), the American Medical Association Physician Consortium for Performance Improvement (PCPI), and the Ambulatory Care Quality Alliance are actively engaged in providing input to CMS on the PVRP and other P4P-related initiatives. SHM has joined the PCPI, which works with medical specialty organizations to develop physician-level performance measures.
As a new member of the PCPI, SHM submitted feedback during the public comment period on perioperative care measures, the development of which was led by the American College of Surgeons, along with input from other medical specialties. SHM will also participate in two upcoming workgroups—one on emergency medicine, which will focus on treating for MI and pneumonia, and another on geriatrics, which will look at falls, urinary incontinence, and end-of-life care. At least through 2006, the PCPI is focusing on measures that fall under the CMS contract and will be included in the PVRP.
SHM is also a member of the NQF, a nonprofit organization that Congress has charged with endorsing consensus-based national standards for measurement and public reporting of healthcare performance data. NQF is seeking nominations for members of the steering committee and technical advisory panels that will oversee the work on new consensus standards for hospital care. This project, sponsored by the AHRQ, will address issues of patient safety, pediatrics, and inpatient care. SHM will submit nominations during this process and plans to be fully engaged.
To further develop its agenda on performance and quality standards in inpatient hospital care, SHM recently established a new Performance and Standards Task Force. This task force is charged with developing a coordinated approach for SHM to work with external organizations in the performance and standards and quality arena and comprises the chairs of the PPC and HQPS committees and other SHM leaders active in organizations like the JCAHO and the IHI.
P4P is here to stay. SHM is well positioned to influence the development and implementation of inpatient quality measures that may eventually become part of a Medicare P4P program for physician services. TH
Allendorf is senior advisor for Advocacy and Government Affairs at SHM. Epstein is senior advisor for Quality Standards and Compliance at SHM.
SHM: BEHIND THE SCENES
How SHM Manages Your Money
By Steven Poitras
In the past three months you have read articles from my peers, including one by Todd Von Deak concerning the great strides he is making in the membership department by ensuring that we are generating the most benefits for our members; one from Scott Johnson, who is taking us into the future with regard to information services; and from Geri Barnes, our education and quality initiatives director, who is helping drive our organizational mission of promoting excellence in the practice of hospital medicine.
This month I want to tell you about our organization, its structure, and what we are collectively doing to ensure that the dues and grants we receive are put to good use to benefit our members and ensure our place in driving hospital medicine forward.
Studies show that 70% of small businesses and small non-profit organizations survive their first year, 30% survive their second year, and only 20% survive after five years. SHM will celebrate its 10th year in 2007. Congratulations and thank you. It’s you, our members, who have taken us this far. It’s you, our members, who will take us into the next 10, 20, 50 years, and beyond. We are not the same organization we were 10 years ago, and I suspect we will not be the same organization in 10 years that we are today.
Over the years I have worked and consulted in many organizations ranging from small mom-and-pops to large, multinational corporations. More often than not, despite a common desire to succeed, conflict exists within various constituencies (e.g., employees, executive management, board of directors) that leads to differing opinions on the best strategy for the company moving forward. I’m proud to say that this isn’t the case with SHM.
We have formulated a business model that uses tried-and-true corporate tools to ensure our members receive the maximum possible value in areas of the greatest impact. It encompasses both our paid and non-paid staff and volunteers to validate what matters most. Your dollars are put to good use. We concentrate on doing things that can be done and done well. If it’s a great idea but doesn’t get to the heart of what our members need or want, we won’t siphon off money and time. We receive money from our members and grantors with the intent of fulfilling a promise or mission. We try to understand what can be accomplished, and then we apply our resources to those goals, ensuring that a higher percentage of those resources are going to mission critical programs and services.
Both our members and grantors look for a strong return on their investment for the monies they send and set aside for our cause. Performance standards are necessary, not only to ensure high level delivery of services but also to ensure our organization’s fiscal responsibility. We employ a staff with many qualifications and backgrounds and coordinate our efforts further with diverse, educated, and dedicated volunteers who are experts in their areas.
Not only is our staff concerned with producing measurable results attributed to the dollars we receive, but we are also measuring mission success in numeric terms other than profit and loss, most specifically within our education and quality initiatives as well as our membership departments. Together these departments are spearheading metrics initiatives that are, for the most part, completely new to our organization but essential to our growth. We involve our staff at all levels of the organization in the pursuit of obtaining these metrics. We strive to collaboratively fulfill SHM’s objectives, and our adaptability allows us to scan the external environment and respond to the ever-changing needs of our members and grantors. Consistency of these values and the internal systems from our information services department that support problem solving, efficiency, and effectiveness at every level across our organizational boundaries help us obtain fulfilling our mission.
Communication about our direction is provided at every opportunity: staff meetings, board meetings, brown-bag lunches, and one-on-one employee discussions. Teamwork is emphasized as the primary means for accomplishing work. When decisions need to be made, all employees and volunteers are sought for counsel and advice from them, their peers, and others who might have insight about our programs. Armed with knowledge, strategic and tactical objectives that are clearly defined and pursued with greater focus, conviction, and diligence our employees embrace the change that is so evident in our fast moving organization.
Everyone in our organization understands where we are going, how we intend to get there, and how he or she fits into our organization’s strategy. The culture of our organization has shifted, but our focus on the benefits of hospital medicine has remained constant. By utilizing these strategic planning tools and techniques, we are redeveloping and revitalizing our corporate mission statement by understanding our organizations strengths, weaknesses, opportunities, threats, and actual and potential competitive advantages. This allows us to move beyond just mere expectations to actually setting the standard by which everyone will be judged.
Our employees, volunteers, members, and grantors remind us that while the business is managed on a daily basis, during our strategic planning and project implementation we focus our strategy on the vital few rather than the trivial many. This allows us to put our organization on the right track for survival and long-term growth.
As we grow, we will search harder and further for experts to help us fulfill our promise of setting SHM as the standard in hospital medicine. I encourage your feedback. Please reach me at [email protected]. You will find that I am always accessible and open to your thoughts and ideas.
Next month you will hear from Laura Allendorf, senior advisor for advocacy and government affairs.
Poitras is director of Business Operations at SHM.
Federal officials are increasingly embracing pay for performance (P4P) in an effort to promote high-quality, cost effective care in government health programs. As the Centers for Medicaid and Medicare Services (CMS) and Congress move forward to implement this concept, SHM is working to ensure that the views of hospitalists are represented in this important debate.
More than 100 P4P programs are already up and running in the private sector in an attempt to reward quality healthcare by setting different payment levels for providers based on how well they meet benchmarks of quality and efficiency. CMS is testing the feasibility of applying this concept to the Medicare program through a number of initiatives.
SHM’s Public Policy and Hospital Quality and Patient Safety committees have been involved in evaluating CMS’ Physician Voluntary Reporting Program (PVRP), launched earlier this year and widely believed to be the precursor to an eventual P4P program for physicians’ services. Under this initiative, physicians are encouraged to submit quality data on a “starter set” of 16 evidence-based measures for certain primary care, surgery, nephrology, and emergency medical services. Physicians who participate in the program receive confidential reports on their performance.
SHM has recommended that hospitalists register their intent to report and begin reporting on relevant performance measures to the extent practicable as a way of becoming more familiar with the program. Because these initial 16 measures have only limited relevance to services billed by hospitalists, SHM is engaged in discussions with lawmakers and their staffs, CMS officials, and consensus organizations involved in developing quality measures, to expand the program’s scope.
As part of Advocacy Day on May 3, some 70 SHM members met with legislators and their staffs and conveyed SHM’s support for initiatives like the PVRP that seek to measure resource use and improve quality, to attain better value for the Medicare program. (See coverage in The Hospitalist SHM Meeting Reporter, July 2006, p. 1.) Participants also educated lawmakers on the role of hospitalists in helping their institutions meet quality reporting requirements mandated under the Medicare Modernization Act, which ties annual hospital payment updates to the submission of performance data for 10 quality measures. The participants also expressed SHM’s interest in working with CMS on demonstration projects that assess the contributions of hospital medicine programs to improved patient care and more efficient management of hospital resources.
In addition to Congress and CMS, non-governmental groups such as the National Quality Forum (NQF), the American Medical Association Physician Consortium for Performance Improvement (PCPI), and the Ambulatory Care Quality Alliance are actively engaged in providing input to CMS on the PVRP and other P4P-related initiatives. SHM has joined the PCPI, which works with medical specialty organizations to develop physician-level performance measures.
As a new member of the PCPI, SHM submitted feedback during the public comment period on perioperative care measures, the development of which was led by the American College of Surgeons, along with input from other medical specialties. SHM will also participate in two upcoming workgroups—one on emergency medicine, which will focus on treating for MI and pneumonia, and another on geriatrics, which will look at falls, urinary incontinence, and end-of-life care. At least through 2006, the PCPI is focusing on measures that fall under the CMS contract and will be included in the PVRP.
SHM is also a member of the NQF, a nonprofit organization that Congress has charged with endorsing consensus-based national standards for measurement and public reporting of healthcare performance data. NQF is seeking nominations for members of the steering committee and technical advisory panels that will oversee the work on new consensus standards for hospital care. This project, sponsored by the AHRQ, will address issues of patient safety, pediatrics, and inpatient care. SHM will submit nominations during this process and plans to be fully engaged.
To further develop its agenda on performance and quality standards in inpatient hospital care, SHM recently established a new Performance and Standards Task Force. This task force is charged with developing a coordinated approach for SHM to work with external organizations in the performance and standards and quality arena and comprises the chairs of the PPC and HQPS committees and other SHM leaders active in organizations like the JCAHO and the IHI.
P4P is here to stay. SHM is well positioned to influence the development and implementation of inpatient quality measures that may eventually become part of a Medicare P4P program for physician services. TH
Allendorf is senior advisor for Advocacy and Government Affairs at SHM. Epstein is senior advisor for Quality Standards and Compliance at SHM.
SHM: BEHIND THE SCENES
How SHM Manages Your Money
By Steven Poitras
In the past three months you have read articles from my peers, including one by Todd Von Deak concerning the great strides he is making in the membership department by ensuring that we are generating the most benefits for our members; one from Scott Johnson, who is taking us into the future with regard to information services; and from Geri Barnes, our education and quality initiatives director, who is helping drive our organizational mission of promoting excellence in the practice of hospital medicine.
This month I want to tell you about our organization, its structure, and what we are collectively doing to ensure that the dues and grants we receive are put to good use to benefit our members and ensure our place in driving hospital medicine forward.
Studies show that 70% of small businesses and small non-profit organizations survive their first year, 30% survive their second year, and only 20% survive after five years. SHM will celebrate its 10th year in 2007. Congratulations and thank you. It’s you, our members, who have taken us this far. It’s you, our members, who will take us into the next 10, 20, 50 years, and beyond. We are not the same organization we were 10 years ago, and I suspect we will not be the same organization in 10 years that we are today.
Over the years I have worked and consulted in many organizations ranging from small mom-and-pops to large, multinational corporations. More often than not, despite a common desire to succeed, conflict exists within various constituencies (e.g., employees, executive management, board of directors) that leads to differing opinions on the best strategy for the company moving forward. I’m proud to say that this isn’t the case with SHM.
We have formulated a business model that uses tried-and-true corporate tools to ensure our members receive the maximum possible value in areas of the greatest impact. It encompasses both our paid and non-paid staff and volunteers to validate what matters most. Your dollars are put to good use. We concentrate on doing things that can be done and done well. If it’s a great idea but doesn’t get to the heart of what our members need or want, we won’t siphon off money and time. We receive money from our members and grantors with the intent of fulfilling a promise or mission. We try to understand what can be accomplished, and then we apply our resources to those goals, ensuring that a higher percentage of those resources are going to mission critical programs and services.
Both our members and grantors look for a strong return on their investment for the monies they send and set aside for our cause. Performance standards are necessary, not only to ensure high level delivery of services but also to ensure our organization’s fiscal responsibility. We employ a staff with many qualifications and backgrounds and coordinate our efforts further with diverse, educated, and dedicated volunteers who are experts in their areas.
Not only is our staff concerned with producing measurable results attributed to the dollars we receive, but we are also measuring mission success in numeric terms other than profit and loss, most specifically within our education and quality initiatives as well as our membership departments. Together these departments are spearheading metrics initiatives that are, for the most part, completely new to our organization but essential to our growth. We involve our staff at all levels of the organization in the pursuit of obtaining these metrics. We strive to collaboratively fulfill SHM’s objectives, and our adaptability allows us to scan the external environment and respond to the ever-changing needs of our members and grantors. Consistency of these values and the internal systems from our information services department that support problem solving, efficiency, and effectiveness at every level across our organizational boundaries help us obtain fulfilling our mission.
Communication about our direction is provided at every opportunity: staff meetings, board meetings, brown-bag lunches, and one-on-one employee discussions. Teamwork is emphasized as the primary means for accomplishing work. When decisions need to be made, all employees and volunteers are sought for counsel and advice from them, their peers, and others who might have insight about our programs. Armed with knowledge, strategic and tactical objectives that are clearly defined and pursued with greater focus, conviction, and diligence our employees embrace the change that is so evident in our fast moving organization.
Everyone in our organization understands where we are going, how we intend to get there, and how he or she fits into our organization’s strategy. The culture of our organization has shifted, but our focus on the benefits of hospital medicine has remained constant. By utilizing these strategic planning tools and techniques, we are redeveloping and revitalizing our corporate mission statement by understanding our organizations strengths, weaknesses, opportunities, threats, and actual and potential competitive advantages. This allows us to move beyond just mere expectations to actually setting the standard by which everyone will be judged.
Our employees, volunteers, members, and grantors remind us that while the business is managed on a daily basis, during our strategic planning and project implementation we focus our strategy on the vital few rather than the trivial many. This allows us to put our organization on the right track for survival and long-term growth.
As we grow, we will search harder and further for experts to help us fulfill our promise of setting SHM as the standard in hospital medicine. I encourage your feedback. Please reach me at [email protected]. You will find that I am always accessible and open to your thoughts and ideas.
Next month you will hear from Laura Allendorf, senior advisor for advocacy and government affairs.
Poitras is director of Business Operations at SHM.
Federal officials are increasingly embracing pay for performance (P4P) in an effort to promote high-quality, cost effective care in government health programs. As the Centers for Medicaid and Medicare Services (CMS) and Congress move forward to implement this concept, SHM is working to ensure that the views of hospitalists are represented in this important debate.
More than 100 P4P programs are already up and running in the private sector in an attempt to reward quality healthcare by setting different payment levels for providers based on how well they meet benchmarks of quality and efficiency. CMS is testing the feasibility of applying this concept to the Medicare program through a number of initiatives.
SHM’s Public Policy and Hospital Quality and Patient Safety committees have been involved in evaluating CMS’ Physician Voluntary Reporting Program (PVRP), launched earlier this year and widely believed to be the precursor to an eventual P4P program for physicians’ services. Under this initiative, physicians are encouraged to submit quality data on a “starter set” of 16 evidence-based measures for certain primary care, surgery, nephrology, and emergency medical services. Physicians who participate in the program receive confidential reports on their performance.
SHM has recommended that hospitalists register their intent to report and begin reporting on relevant performance measures to the extent practicable as a way of becoming more familiar with the program. Because these initial 16 measures have only limited relevance to services billed by hospitalists, SHM is engaged in discussions with lawmakers and their staffs, CMS officials, and consensus organizations involved in developing quality measures, to expand the program’s scope.
As part of Advocacy Day on May 3, some 70 SHM members met with legislators and their staffs and conveyed SHM’s support for initiatives like the PVRP that seek to measure resource use and improve quality, to attain better value for the Medicare program. (See coverage in The Hospitalist SHM Meeting Reporter, July 2006, p. 1.) Participants also educated lawmakers on the role of hospitalists in helping their institutions meet quality reporting requirements mandated under the Medicare Modernization Act, which ties annual hospital payment updates to the submission of performance data for 10 quality measures. The participants also expressed SHM’s interest in working with CMS on demonstration projects that assess the contributions of hospital medicine programs to improved patient care and more efficient management of hospital resources.
In addition to Congress and CMS, non-governmental groups such as the National Quality Forum (NQF), the American Medical Association Physician Consortium for Performance Improvement (PCPI), and the Ambulatory Care Quality Alliance are actively engaged in providing input to CMS on the PVRP and other P4P-related initiatives. SHM has joined the PCPI, which works with medical specialty organizations to develop physician-level performance measures.
As a new member of the PCPI, SHM submitted feedback during the public comment period on perioperative care measures, the development of which was led by the American College of Surgeons, along with input from other medical specialties. SHM will also participate in two upcoming workgroups—one on emergency medicine, which will focus on treating for MI and pneumonia, and another on geriatrics, which will look at falls, urinary incontinence, and end-of-life care. At least through 2006, the PCPI is focusing on measures that fall under the CMS contract and will be included in the PVRP.
SHM is also a member of the NQF, a nonprofit organization that Congress has charged with endorsing consensus-based national standards for measurement and public reporting of healthcare performance data. NQF is seeking nominations for members of the steering committee and technical advisory panels that will oversee the work on new consensus standards for hospital care. This project, sponsored by the AHRQ, will address issues of patient safety, pediatrics, and inpatient care. SHM will submit nominations during this process and plans to be fully engaged.
To further develop its agenda on performance and quality standards in inpatient hospital care, SHM recently established a new Performance and Standards Task Force. This task force is charged with developing a coordinated approach for SHM to work with external organizations in the performance and standards and quality arena and comprises the chairs of the PPC and HQPS committees and other SHM leaders active in organizations like the JCAHO and the IHI.
P4P is here to stay. SHM is well positioned to influence the development and implementation of inpatient quality measures that may eventually become part of a Medicare P4P program for physician services. TH
Allendorf is senior advisor for Advocacy and Government Affairs at SHM. Epstein is senior advisor for Quality Standards and Compliance at SHM.
SHM: BEHIND THE SCENES
How SHM Manages Your Money
By Steven Poitras
In the past three months you have read articles from my peers, including one by Todd Von Deak concerning the great strides he is making in the membership department by ensuring that we are generating the most benefits for our members; one from Scott Johnson, who is taking us into the future with regard to information services; and from Geri Barnes, our education and quality initiatives director, who is helping drive our organizational mission of promoting excellence in the practice of hospital medicine.
This month I want to tell you about our organization, its structure, and what we are collectively doing to ensure that the dues and grants we receive are put to good use to benefit our members and ensure our place in driving hospital medicine forward.
Studies show that 70% of small businesses and small non-profit organizations survive their first year, 30% survive their second year, and only 20% survive after five years. SHM will celebrate its 10th year in 2007. Congratulations and thank you. It’s you, our members, who have taken us this far. It’s you, our members, who will take us into the next 10, 20, 50 years, and beyond. We are not the same organization we were 10 years ago, and I suspect we will not be the same organization in 10 years that we are today.
Over the years I have worked and consulted in many organizations ranging from small mom-and-pops to large, multinational corporations. More often than not, despite a common desire to succeed, conflict exists within various constituencies (e.g., employees, executive management, board of directors) that leads to differing opinions on the best strategy for the company moving forward. I’m proud to say that this isn’t the case with SHM.
We have formulated a business model that uses tried-and-true corporate tools to ensure our members receive the maximum possible value in areas of the greatest impact. It encompasses both our paid and non-paid staff and volunteers to validate what matters most. Your dollars are put to good use. We concentrate on doing things that can be done and done well. If it’s a great idea but doesn’t get to the heart of what our members need or want, we won’t siphon off money and time. We receive money from our members and grantors with the intent of fulfilling a promise or mission. We try to understand what can be accomplished, and then we apply our resources to those goals, ensuring that a higher percentage of those resources are going to mission critical programs and services.
Both our members and grantors look for a strong return on their investment for the monies they send and set aside for our cause. Performance standards are necessary, not only to ensure high level delivery of services but also to ensure our organization’s fiscal responsibility. We employ a staff with many qualifications and backgrounds and coordinate our efforts further with diverse, educated, and dedicated volunteers who are experts in their areas.
Not only is our staff concerned with producing measurable results attributed to the dollars we receive, but we are also measuring mission success in numeric terms other than profit and loss, most specifically within our education and quality initiatives as well as our membership departments. Together these departments are spearheading metrics initiatives that are, for the most part, completely new to our organization but essential to our growth. We involve our staff at all levels of the organization in the pursuit of obtaining these metrics. We strive to collaboratively fulfill SHM’s objectives, and our adaptability allows us to scan the external environment and respond to the ever-changing needs of our members and grantors. Consistency of these values and the internal systems from our information services department that support problem solving, efficiency, and effectiveness at every level across our organizational boundaries help us obtain fulfilling our mission.
Communication about our direction is provided at every opportunity: staff meetings, board meetings, brown-bag lunches, and one-on-one employee discussions. Teamwork is emphasized as the primary means for accomplishing work. When decisions need to be made, all employees and volunteers are sought for counsel and advice from them, their peers, and others who might have insight about our programs. Armed with knowledge, strategic and tactical objectives that are clearly defined and pursued with greater focus, conviction, and diligence our employees embrace the change that is so evident in our fast moving organization.
Everyone in our organization understands where we are going, how we intend to get there, and how he or she fits into our organization’s strategy. The culture of our organization has shifted, but our focus on the benefits of hospital medicine has remained constant. By utilizing these strategic planning tools and techniques, we are redeveloping and revitalizing our corporate mission statement by understanding our organizations strengths, weaknesses, opportunities, threats, and actual and potential competitive advantages. This allows us to move beyond just mere expectations to actually setting the standard by which everyone will be judged.
Our employees, volunteers, members, and grantors remind us that while the business is managed on a daily basis, during our strategic planning and project implementation we focus our strategy on the vital few rather than the trivial many. This allows us to put our organization on the right track for survival and long-term growth.
As we grow, we will search harder and further for experts to help us fulfill our promise of setting SHM as the standard in hospital medicine. I encourage your feedback. Please reach me at [email protected]. You will find that I am always accessible and open to your thoughts and ideas.
Next month you will hear from Laura Allendorf, senior advisor for advocacy and government affairs.
Poitras is director of Business Operations at SHM.
SHM Varietals
Gardens are not made by sitting in the shade.—Rudyard Kipling
For those of us who live in a climate where the vegetation dies in the winter, this is a great time of year. In the spring our plants and trees revive themselves and begin to give us color and aromas. We tend to our plants and add to our gardens with new flowers. We plant our seeds for vegetables and anxiously await the first fruit.
As I watched my garden blossoming this year, I was reminded of our Society. Perhaps, this is too poetic a comparison, but one that I think fits. No garden is pretty if it comprises all the same plant or grass or color. One of the most appealing things about a garden is its great variety. There are many variations of plants: grasses, flowers, trees, and groundcover. In a great garden, there may be many types of grass in many colors and shapes. There are flowers of various kinds. They all bloom at different times from spring to fall. Their colors range from white to the darkest blues and purples. Their fragrances are apparent in the morning or evening or at night. Some of them are phosphorescent. There are rocks and stones, water features, birds, and insects. Each part of the garden has a different role and yet together they create a peaceful and pleasing environment—a place we want to be. Without the many kinds of sites, smells, and sounds, a garden would not be interesting or pleasing to us.
Our organization is like a garden: We have many types of groups that make up the whole. For instance, we have groups of specialties. We are pediatricians. We are family medicine physicians. We are internists.
We have different professions. Our nurse practitioner and physician assistant members have a unique role. Their support of the organization and insights into patient care and care delivery give us all another perspective. In this complex age of pharmacology and polypharmacy, our pharmacy members assist us all in compiling a treatment plan for patients that helps them and does as little harm as possible. They educate us all about the many interactions and side effects of our medications.
We practice in different settings. Some of us see patients at the bedside in a community hospital. There we attempt to bring the best knowledge and skill we can to care for the individual. In addition, we bring our ideas and innovation to all the patients in our hospital by establishing new treatment plans and processes. We educate our nursing staffs and our fellow physicians. We educate our administrators about healthcare changes that can improve our community and our patients. We create liaisons with other specialists in the emergency department, the operating room, and in the ICU. Some of us practice at the bedside in a teaching setting. We care for patients with residents and medical students. We hope to bring to them a good example of empathy and care at the bedside as well as medical knowledge consistent with the best medical practice. Here, we also create liaisons with other specialists in the emergency department, the operating room, and in the ICU. By working in a multidisciplinary fashion, we hope to advance the care of all hospitalized patients.
We contribute by leading research and studies that discover new methods or new types of care. These results guide others at the bedside and in training. We have different interests. Thanks to this diversity we have individuals working on many topics to advance our specialty. Some individuals pursue public policy. They want to advance our field through the actions of the government and the national payers. The members’ focus on education has led to the core competency product, many wonderful national meetings, and active committee work. Those interested in research strive to produce a world-class journal, research opportunities, and a place for all hospitalists’ researchers. Quality and safety are important to our national healthcare. Our members have created resource rooms to disseminate information on these topics. Others, still, want to advance our field through the study of lifestyle and practice setting issues. Finally, we move toward recognition as a board certified specialty.
Regardless of how one looks at our SHM garden, we would be less of an organization without our variety and diversity. We continue to strive to meet the needs of and provide opportunities for all our members, regardless of their interests, practice setting, or discipline. We do not have a finished garden because gardens are always a work in progress, but we hope to continue to be the best place for all hospitalists to grow. TH
Dr. Gorman is the president of SHM.
Gardens are not made by sitting in the shade.—Rudyard Kipling
For those of us who live in a climate where the vegetation dies in the winter, this is a great time of year. In the spring our plants and trees revive themselves and begin to give us color and aromas. We tend to our plants and add to our gardens with new flowers. We plant our seeds for vegetables and anxiously await the first fruit.
As I watched my garden blossoming this year, I was reminded of our Society. Perhaps, this is too poetic a comparison, but one that I think fits. No garden is pretty if it comprises all the same plant or grass or color. One of the most appealing things about a garden is its great variety. There are many variations of plants: grasses, flowers, trees, and groundcover. In a great garden, there may be many types of grass in many colors and shapes. There are flowers of various kinds. They all bloom at different times from spring to fall. Their colors range from white to the darkest blues and purples. Their fragrances are apparent in the morning or evening or at night. Some of them are phosphorescent. There are rocks and stones, water features, birds, and insects. Each part of the garden has a different role and yet together they create a peaceful and pleasing environment—a place we want to be. Without the many kinds of sites, smells, and sounds, a garden would not be interesting or pleasing to us.
Our organization is like a garden: We have many types of groups that make up the whole. For instance, we have groups of specialties. We are pediatricians. We are family medicine physicians. We are internists.
We have different professions. Our nurse practitioner and physician assistant members have a unique role. Their support of the organization and insights into patient care and care delivery give us all another perspective. In this complex age of pharmacology and polypharmacy, our pharmacy members assist us all in compiling a treatment plan for patients that helps them and does as little harm as possible. They educate us all about the many interactions and side effects of our medications.
We practice in different settings. Some of us see patients at the bedside in a community hospital. There we attempt to bring the best knowledge and skill we can to care for the individual. In addition, we bring our ideas and innovation to all the patients in our hospital by establishing new treatment plans and processes. We educate our nursing staffs and our fellow physicians. We educate our administrators about healthcare changes that can improve our community and our patients. We create liaisons with other specialists in the emergency department, the operating room, and in the ICU. Some of us practice at the bedside in a teaching setting. We care for patients with residents and medical students. We hope to bring to them a good example of empathy and care at the bedside as well as medical knowledge consistent with the best medical practice. Here, we also create liaisons with other specialists in the emergency department, the operating room, and in the ICU. By working in a multidisciplinary fashion, we hope to advance the care of all hospitalized patients.
We contribute by leading research and studies that discover new methods or new types of care. These results guide others at the bedside and in training. We have different interests. Thanks to this diversity we have individuals working on many topics to advance our specialty. Some individuals pursue public policy. They want to advance our field through the actions of the government and the national payers. The members’ focus on education has led to the core competency product, many wonderful national meetings, and active committee work. Those interested in research strive to produce a world-class journal, research opportunities, and a place for all hospitalists’ researchers. Quality and safety are important to our national healthcare. Our members have created resource rooms to disseminate information on these topics. Others, still, want to advance our field through the study of lifestyle and practice setting issues. Finally, we move toward recognition as a board certified specialty.
Regardless of how one looks at our SHM garden, we would be less of an organization without our variety and diversity. We continue to strive to meet the needs of and provide opportunities for all our members, regardless of their interests, practice setting, or discipline. We do not have a finished garden because gardens are always a work in progress, but we hope to continue to be the best place for all hospitalists to grow. TH
Dr. Gorman is the president of SHM.
Gardens are not made by sitting in the shade.—Rudyard Kipling
For those of us who live in a climate where the vegetation dies in the winter, this is a great time of year. In the spring our plants and trees revive themselves and begin to give us color and aromas. We tend to our plants and add to our gardens with new flowers. We plant our seeds for vegetables and anxiously await the first fruit.
As I watched my garden blossoming this year, I was reminded of our Society. Perhaps, this is too poetic a comparison, but one that I think fits. No garden is pretty if it comprises all the same plant or grass or color. One of the most appealing things about a garden is its great variety. There are many variations of plants: grasses, flowers, trees, and groundcover. In a great garden, there may be many types of grass in many colors and shapes. There are flowers of various kinds. They all bloom at different times from spring to fall. Their colors range from white to the darkest blues and purples. Their fragrances are apparent in the morning or evening or at night. Some of them are phosphorescent. There are rocks and stones, water features, birds, and insects. Each part of the garden has a different role and yet together they create a peaceful and pleasing environment—a place we want to be. Without the many kinds of sites, smells, and sounds, a garden would not be interesting or pleasing to us.
Our organization is like a garden: We have many types of groups that make up the whole. For instance, we have groups of specialties. We are pediatricians. We are family medicine physicians. We are internists.
We have different professions. Our nurse practitioner and physician assistant members have a unique role. Their support of the organization and insights into patient care and care delivery give us all another perspective. In this complex age of pharmacology and polypharmacy, our pharmacy members assist us all in compiling a treatment plan for patients that helps them and does as little harm as possible. They educate us all about the many interactions and side effects of our medications.
We practice in different settings. Some of us see patients at the bedside in a community hospital. There we attempt to bring the best knowledge and skill we can to care for the individual. In addition, we bring our ideas and innovation to all the patients in our hospital by establishing new treatment plans and processes. We educate our nursing staffs and our fellow physicians. We educate our administrators about healthcare changes that can improve our community and our patients. We create liaisons with other specialists in the emergency department, the operating room, and in the ICU. Some of us practice at the bedside in a teaching setting. We care for patients with residents and medical students. We hope to bring to them a good example of empathy and care at the bedside as well as medical knowledge consistent with the best medical practice. Here, we also create liaisons with other specialists in the emergency department, the operating room, and in the ICU. By working in a multidisciplinary fashion, we hope to advance the care of all hospitalized patients.
We contribute by leading research and studies that discover new methods or new types of care. These results guide others at the bedside and in training. We have different interests. Thanks to this diversity we have individuals working on many topics to advance our specialty. Some individuals pursue public policy. They want to advance our field through the actions of the government and the national payers. The members’ focus on education has led to the core competency product, many wonderful national meetings, and active committee work. Those interested in research strive to produce a world-class journal, research opportunities, and a place for all hospitalists’ researchers. Quality and safety are important to our national healthcare. Our members have created resource rooms to disseminate information on these topics. Others, still, want to advance our field through the study of lifestyle and practice setting issues. Finally, we move toward recognition as a board certified specialty.
Regardless of how one looks at our SHM garden, we would be less of an organization without our variety and diversity. We continue to strive to meet the needs of and provide opportunities for all our members, regardless of their interests, practice setting, or discipline. We do not have a finished garden because gardens are always a work in progress, but we hope to continue to be the best place for all hospitalists to grow. TH
Dr. Gorman is the president of SHM.