User login
Positive results reported for ixekizumab versus adalimumab in PsA
Eli Lilly and Co. has announced positive results from the phase 3b/4, multicenter, randomized, open-label, parallel-group SPIRIT-H2H trial, which compared ixekizumab (Taltz) with adalimumab (Humira) in patients with psoriatic arthritis who had previously not taken a biologic disease-modifying antirheumatic drug.
The 52-week study included 566 patients with psoriatic arthritis. Patients received either ixekizumab at 80 mg every 4 weeks after a 160-mg loading dose or adalimumab at 40 mg every 2 weeks. The primary endpoint was the proportion of patients achieving at least a 50% reduction in American College of Rheumatology (ACR50) criteria at 24 weeks.
After 24 weeks, patients in the ixekizumab group were more likely to achieve ACR50, compared with those in the adalimumab group. In addition, patients receiving ixekizumab were more likely to achieve 100% skin clearance according to the Psoriasis Area and Severity Index. Ixekizumab also met all secondary trial endpoints.
“The positive results from the SPIRIT-H2H trial reinforce that Taltz effectively treats the debilitating joint signs and symptoms of active psoriatic arthritis, while also providing skin clearance. These results provide evidence that Taltz can be used as a first-line biologic treatment for patients with active psoriatic arthritis,” Lotus Mallbris, MD, PhD, vice president of immunology development at Lilly, said in the press release.
More detailed results will be presented at meetings and published in peer-reviewed journals in 2019, the company said.
Eli Lilly and Co. has announced positive results from the phase 3b/4, multicenter, randomized, open-label, parallel-group SPIRIT-H2H trial, which compared ixekizumab (Taltz) with adalimumab (Humira) in patients with psoriatic arthritis who had previously not taken a biologic disease-modifying antirheumatic drug.
The 52-week study included 566 patients with psoriatic arthritis. Patients received either ixekizumab at 80 mg every 4 weeks after a 160-mg loading dose or adalimumab at 40 mg every 2 weeks. The primary endpoint was the proportion of patients achieving at least a 50% reduction in American College of Rheumatology (ACR50) criteria at 24 weeks.
After 24 weeks, patients in the ixekizumab group were more likely to achieve ACR50, compared with those in the adalimumab group. In addition, patients receiving ixekizumab were more likely to achieve 100% skin clearance according to the Psoriasis Area and Severity Index. Ixekizumab also met all secondary trial endpoints.
“The positive results from the SPIRIT-H2H trial reinforce that Taltz effectively treats the debilitating joint signs and symptoms of active psoriatic arthritis, while also providing skin clearance. These results provide evidence that Taltz can be used as a first-line biologic treatment for patients with active psoriatic arthritis,” Lotus Mallbris, MD, PhD, vice president of immunology development at Lilly, said in the press release.
More detailed results will be presented at meetings and published in peer-reviewed journals in 2019, the company said.
Eli Lilly and Co. has announced positive results from the phase 3b/4, multicenter, randomized, open-label, parallel-group SPIRIT-H2H trial, which compared ixekizumab (Taltz) with adalimumab (Humira) in patients with psoriatic arthritis who had previously not taken a biologic disease-modifying antirheumatic drug.
The 52-week study included 566 patients with psoriatic arthritis. Patients received either ixekizumab at 80 mg every 4 weeks after a 160-mg loading dose or adalimumab at 40 mg every 2 weeks. The primary endpoint was the proportion of patients achieving at least a 50% reduction in American College of Rheumatology (ACR50) criteria at 24 weeks.
After 24 weeks, patients in the ixekizumab group were more likely to achieve ACR50, compared with those in the adalimumab group. In addition, patients receiving ixekizumab were more likely to achieve 100% skin clearance according to the Psoriasis Area and Severity Index. Ixekizumab also met all secondary trial endpoints.
“The positive results from the SPIRIT-H2H trial reinforce that Taltz effectively treats the debilitating joint signs and symptoms of active psoriatic arthritis, while also providing skin clearance. These results provide evidence that Taltz can be used as a first-line biologic treatment for patients with active psoriatic arthritis,” Lotus Mallbris, MD, PhD, vice president of immunology development at Lilly, said in the press release.
More detailed results will be presented at meetings and published in peer-reviewed journals in 2019, the company said.
Natural killer cells implicated in psoriatic arthritis
CHICAGO –
This natural killer cell interacts with the CD94/NKG2A receptor, part of a system believed to have been in place in humans for more than 90 million years.
“We believe there is a possible role for the innate immune system in the development of psoriatic arthritis and its distinction from psoriasis,” Vinod Chandran, MD, PhD, declared at the annual meeting of the American College of Rheumatology.
Dr. Chandran, of the University of Toronto, presented an analysis of a discovery cohort comprising 1,155 patients with dermatologist-diagnosed psoriasis of greater than 10 years duration, 664 rheumatologist-diagnosed psoriatic arthritis patients, and 3,118 controls, all participants in the International Psoriasis and Arthritis Research Team program. These findings were then independently confirmed in a separate University of Toronto replication cohort of 659 psoriasis patients, 1,177 psoriatic arthritis patients of European ancestry, and 1,096 controls.
By way of background, the rheumatologist explained that psoriasis and psoriatic arthritis are known to differ in terms of their genetic architecture, the biggest difference being in the HLA class I region, where HLA-C predominates in psoriasis and HLA-B in psoriatic arthritis. These structurally unrelated forms of HLA class I are known to educate natural killer cells and shape their function. Dr. Chandran and his coinvestigators were eager to shed new light on the mechanisms by which this leads to rheumatic disease.
Humans can be divided into three groups based upon whether they are HLA-B21 methionine/methionine (M/M), HLA-B21 M/threonine (T), or HLA-B21 T/T. The B21 M types educate CD94/NKG2A-positive natural killer cells by delivering functional peptides to the CD94/NKG2A receptor, while the B21 T/T version does not.
In the discovery cohort, individuals with psoriatic arthritis turned out to be 36% more likely to be HLA-B21 M/M or HLA-B21 M/T than were the psoriasis patients, while the psoriasis patients were 22% less likely to be B21 M–positive than controls. These relationships were confirmed in the replication cohort, where psoriatic arthritis patients were 40% more likely to be B21 M–positive than psoriasis patients, and psoriasis patients were 18% less likely to be B21 M–positive than controls, with all of these differences being statistically significant.
While this is translational science, Dr. Chandran explained that it has important clinical implications. He and his coinvestigators are developing a genetic marker panel to differentiate psoriatic arthritis from psoriasis, as are other research groups. And the Toronto investigators are now convinced that including HLA-B21 M/M and HLA-B21 M/T in their evolving genetic test is worthwhile in terms of boosting the test’s predictive power. The 36%-40% increased risk of psoriatic arthritis associated with B21 M–positivity isn’t sufficiently large for it to serve as a standalone test, but when the genetic test panel is finalized and the investigators can evaluate its positive and negative predictive value, it will be clear that the B21 M component will provide added value, he predicted.
Because psoriatic arthritis can take on a variety of disparate forms clinically, Dr. Chandran and his coworkers believe their genetic test will prove most useful for nonrheumatologists, especially dermatologists and primary care physicians.
He reported having no relevant financial relationships regarding this study, funded by the Canadian Institutes of Health Research, the Krembil Foundation, and the Arthritis Foundation.
SOURCE: Chandran V et al. Arthritis Rheumatol. 2018;70(Suppl 10), Abstract 2787.
CHICAGO –
This natural killer cell interacts with the CD94/NKG2A receptor, part of a system believed to have been in place in humans for more than 90 million years.
“We believe there is a possible role for the innate immune system in the development of psoriatic arthritis and its distinction from psoriasis,” Vinod Chandran, MD, PhD, declared at the annual meeting of the American College of Rheumatology.
Dr. Chandran, of the University of Toronto, presented an analysis of a discovery cohort comprising 1,155 patients with dermatologist-diagnosed psoriasis of greater than 10 years duration, 664 rheumatologist-diagnosed psoriatic arthritis patients, and 3,118 controls, all participants in the International Psoriasis and Arthritis Research Team program. These findings were then independently confirmed in a separate University of Toronto replication cohort of 659 psoriasis patients, 1,177 psoriatic arthritis patients of European ancestry, and 1,096 controls.
By way of background, the rheumatologist explained that psoriasis and psoriatic arthritis are known to differ in terms of their genetic architecture, the biggest difference being in the HLA class I region, where HLA-C predominates in psoriasis and HLA-B in psoriatic arthritis. These structurally unrelated forms of HLA class I are known to educate natural killer cells and shape their function. Dr. Chandran and his coinvestigators were eager to shed new light on the mechanisms by which this leads to rheumatic disease.
Humans can be divided into three groups based upon whether they are HLA-B21 methionine/methionine (M/M), HLA-B21 M/threonine (T), or HLA-B21 T/T. The B21 M types educate CD94/NKG2A-positive natural killer cells by delivering functional peptides to the CD94/NKG2A receptor, while the B21 T/T version does not.
In the discovery cohort, individuals with psoriatic arthritis turned out to be 36% more likely to be HLA-B21 M/M or HLA-B21 M/T than were the psoriasis patients, while the psoriasis patients were 22% less likely to be B21 M–positive than controls. These relationships were confirmed in the replication cohort, where psoriatic arthritis patients were 40% more likely to be B21 M–positive than psoriasis patients, and psoriasis patients were 18% less likely to be B21 M–positive than controls, with all of these differences being statistically significant.
While this is translational science, Dr. Chandran explained that it has important clinical implications. He and his coinvestigators are developing a genetic marker panel to differentiate psoriatic arthritis from psoriasis, as are other research groups. And the Toronto investigators are now convinced that including HLA-B21 M/M and HLA-B21 M/T in their evolving genetic test is worthwhile in terms of boosting the test’s predictive power. The 36%-40% increased risk of psoriatic arthritis associated with B21 M–positivity isn’t sufficiently large for it to serve as a standalone test, but when the genetic test panel is finalized and the investigators can evaluate its positive and negative predictive value, it will be clear that the B21 M component will provide added value, he predicted.
Because psoriatic arthritis can take on a variety of disparate forms clinically, Dr. Chandran and his coworkers believe their genetic test will prove most useful for nonrheumatologists, especially dermatologists and primary care physicians.
He reported having no relevant financial relationships regarding this study, funded by the Canadian Institutes of Health Research, the Krembil Foundation, and the Arthritis Foundation.
SOURCE: Chandran V et al. Arthritis Rheumatol. 2018;70(Suppl 10), Abstract 2787.
CHICAGO –
This natural killer cell interacts with the CD94/NKG2A receptor, part of a system believed to have been in place in humans for more than 90 million years.
“We believe there is a possible role for the innate immune system in the development of psoriatic arthritis and its distinction from psoriasis,” Vinod Chandran, MD, PhD, declared at the annual meeting of the American College of Rheumatology.
Dr. Chandran, of the University of Toronto, presented an analysis of a discovery cohort comprising 1,155 patients with dermatologist-diagnosed psoriasis of greater than 10 years duration, 664 rheumatologist-diagnosed psoriatic arthritis patients, and 3,118 controls, all participants in the International Psoriasis and Arthritis Research Team program. These findings were then independently confirmed in a separate University of Toronto replication cohort of 659 psoriasis patients, 1,177 psoriatic arthritis patients of European ancestry, and 1,096 controls.
By way of background, the rheumatologist explained that psoriasis and psoriatic arthritis are known to differ in terms of their genetic architecture, the biggest difference being in the HLA class I region, where HLA-C predominates in psoriasis and HLA-B in psoriatic arthritis. These structurally unrelated forms of HLA class I are known to educate natural killer cells and shape their function. Dr. Chandran and his coinvestigators were eager to shed new light on the mechanisms by which this leads to rheumatic disease.
Humans can be divided into three groups based upon whether they are HLA-B21 methionine/methionine (M/M), HLA-B21 M/threonine (T), or HLA-B21 T/T. The B21 M types educate CD94/NKG2A-positive natural killer cells by delivering functional peptides to the CD94/NKG2A receptor, while the B21 T/T version does not.
In the discovery cohort, individuals with psoriatic arthritis turned out to be 36% more likely to be HLA-B21 M/M or HLA-B21 M/T than were the psoriasis patients, while the psoriasis patients were 22% less likely to be B21 M–positive than controls. These relationships were confirmed in the replication cohort, where psoriatic arthritis patients were 40% more likely to be B21 M–positive than psoriasis patients, and psoriasis patients were 18% less likely to be B21 M–positive than controls, with all of these differences being statistically significant.
While this is translational science, Dr. Chandran explained that it has important clinical implications. He and his coinvestigators are developing a genetic marker panel to differentiate psoriatic arthritis from psoriasis, as are other research groups. And the Toronto investigators are now convinced that including HLA-B21 M/M and HLA-B21 M/T in their evolving genetic test is worthwhile in terms of boosting the test’s predictive power. The 36%-40% increased risk of psoriatic arthritis associated with B21 M–positivity isn’t sufficiently large for it to serve as a standalone test, but when the genetic test panel is finalized and the investigators can evaluate its positive and negative predictive value, it will be clear that the B21 M component will provide added value, he predicted.
Because psoriatic arthritis can take on a variety of disparate forms clinically, Dr. Chandran and his coworkers believe their genetic test will prove most useful for nonrheumatologists, especially dermatologists and primary care physicians.
He reported having no relevant financial relationships regarding this study, funded by the Canadian Institutes of Health Research, the Krembil Foundation, and the Arthritis Foundation.
SOURCE: Chandran V et al. Arthritis Rheumatol. 2018;70(Suppl 10), Abstract 2787.
REPORTING FROM THE ACR ANNUAL MEETING
Key clinical point: A genetic panel designed to differentiate psoriatic arthritis from psoriasis is drawing closer to fruition.
Major finding: The prevalence of HLA-B21 methionine is increased by roughly 40% in patients with psoriatic arthritis, compared with psoriasis patients.
Study details: This translational study included two independent cohorts totaling 1,814 psoriasis patients, 1,841 with psoriatic arthritis, and 4,214 controls.
Disclosures: The presenter reported having no relevant financial relationships regarding this study, which was funded by the Canadian Institutes of Health Research, the Krembil Foundation, and the Arthritis Foundation.
Source: Chandran V et al. Arthritis Rheumatol. 2018;70(Suppl 10), Abstract 2787.
Algorithm proposes approach for managing TNF inhibitor–induced psoriasis
Patients with tumor necrosis factor inhibitor–induced psoriasis could potentially be switched to a different drug class if they have moderate to severe skin eruption or mild skin eruption with an uncontrolled underlying disease such as inflammatory bowel disease, psoriasis, psoriatic arthritis, or rheumatoid arthritis, according to a new treatment algorithm proposed by researchers from Brigham and Women’s Hospital and Harvard Medical School in Boston.
The researchers outlined the prevalence of tumor necrosis factor–alpha inhibitor (TNFi)-induced psoriasis in a literature review of inflammatory bowel disease (IBD), psoriasis, psoriatic arthritis (PsA), and rheumatoid arthritis (RA) and identified an estimated rate of between 2.3% and 5% in patients with RA and between 1.6% and 2.7% in patients with IBD. Although there have been reports of TNFi-induced psoriasis in patients with psoriasis and PsA, the prevalence is unclear, they wrote in the Journal of Psoriasis and Psoriatic Arthritis.
The authors then created an algorithm to manage and treat TNFi-induced psoriasiform skin eruptions with decisions to continue therapy and “treat through” symptoms, switch to a different anti-TNF therapy, or switch to a different drug class based on severity of symptoms, whether the underlying disease is well controlled, and how patients with those underlying diseases have fared with those specific therapies or agents.
“We’ve shifted gears over the past decade, and we’ve gone from having very few agents and trying to keep patients desperately on one or two agents because we didn’t want to have to give up on them for their other comorbid disease, whether it was Crohn’s, colitis, RA, or whatever it may be,” senior author Joseph Merola, MD, director of the Center for Skin and Related Musculoskeletal Diseases at Brigham and Women’s Hospital, Boston, said in an interview. “We’re now in an area where we can have an algorithm like this, and we have so many more mechanistic options to move to.”
Dr. Merola, who is board certified in dermatology and rheumatology, said the algorithm is meant to “open a dialogue” with other specialists in different areas and raise awareness of treatments in related but separate fields. For diseases not often seen by more than one specialty, with the exception of psoriasis and PsA, he said that “the idea is to start a dialogue and increase communication between specialists.”
Dr. Merola noted that while the algorithm in many respects is meant to guide a physician in a specialty in appropriate medication decisions, at the same time he hopes that “it opens a dialogue and communication with the other specialty who tends to oversee this particular disease state or class of medicine to really work together to try to find the right drug for the right person.”
For patients with a mild skin eruption and a controlled underlying disease, the algorithm recommends a “treat through” approach by continuing anti-TNF therapy and treating psoriasis symptoms with topical steroids, ultraviolet therapy, methotrexate, cyclosporine, or acitretin, and to consider dapsone in cases of pustular psoriasis. However, the researchers noted that “treat through” studies have reported complete symptom resolution in 26%-41% of patients.
For patients with recalcitrant or worsening TNFi-induced psoriasis or patients with mild skin eruptions with an uncontrolled underlying disease, the researchers proposed considering switching to a different anti-TNF therapy, although studies have shown complete resolution of symptoms in only 5%-37% of patients.
If patients worsen from there, or if they have moderate to-severe skin eruption with uncontrolled underlying disease, they could be considered for switching to a different drug class and treated based on their underlying disease, along with treatment for psoriasis symptoms. This approach has been shown to completely resolve lesions in up to 64% of cases, they said. IBD patients could benefit from ustekinumab, vedolizumab, 6-mercaptopurine, or azathioprine as an alternative to anti-TNF therapy. Those patients with psoriasis should be considered for guselkumab, while ustekinumab, ixekizumab, secukinumab, and apremilast are effective treatments for patients with psoriasis and PsA. Patients with RA could receive treatment with tocilizumab, rituximab, abatacept, and tofacitinib, the authors wrote.
Dr. Merola reported serving as a consultant and/or investigator for Merck Research Laboratories, AbbVie, Dermavant, Eli Lilly, Novartis, Janssen, UCB, Samumed, Celgene, Sanofi Regeneron, GlaxoSmithKline, Almirall, Sun Pharma, Biogen, Pfizer, Incyte, Aclaris, and Leo Pharma.
SOURCE: Li SJ et al. J Psoriasis Psoriatic Arthritis. 2018 Nov 21. doi: 10.1177/2475530318810851.
Patients with tumor necrosis factor inhibitor–induced psoriasis could potentially be switched to a different drug class if they have moderate to severe skin eruption or mild skin eruption with an uncontrolled underlying disease such as inflammatory bowel disease, psoriasis, psoriatic arthritis, or rheumatoid arthritis, according to a new treatment algorithm proposed by researchers from Brigham and Women’s Hospital and Harvard Medical School in Boston.
The researchers outlined the prevalence of tumor necrosis factor–alpha inhibitor (TNFi)-induced psoriasis in a literature review of inflammatory bowel disease (IBD), psoriasis, psoriatic arthritis (PsA), and rheumatoid arthritis (RA) and identified an estimated rate of between 2.3% and 5% in patients with RA and between 1.6% and 2.7% in patients with IBD. Although there have been reports of TNFi-induced psoriasis in patients with psoriasis and PsA, the prevalence is unclear, they wrote in the Journal of Psoriasis and Psoriatic Arthritis.
The authors then created an algorithm to manage and treat TNFi-induced psoriasiform skin eruptions with decisions to continue therapy and “treat through” symptoms, switch to a different anti-TNF therapy, or switch to a different drug class based on severity of symptoms, whether the underlying disease is well controlled, and how patients with those underlying diseases have fared with those specific therapies or agents.
“We’ve shifted gears over the past decade, and we’ve gone from having very few agents and trying to keep patients desperately on one or two agents because we didn’t want to have to give up on them for their other comorbid disease, whether it was Crohn’s, colitis, RA, or whatever it may be,” senior author Joseph Merola, MD, director of the Center for Skin and Related Musculoskeletal Diseases at Brigham and Women’s Hospital, Boston, said in an interview. “We’re now in an area where we can have an algorithm like this, and we have so many more mechanistic options to move to.”
Dr. Merola, who is board certified in dermatology and rheumatology, said the algorithm is meant to “open a dialogue” with other specialists in different areas and raise awareness of treatments in related but separate fields. For diseases not often seen by more than one specialty, with the exception of psoriasis and PsA, he said that “the idea is to start a dialogue and increase communication between specialists.”
Dr. Merola noted that while the algorithm in many respects is meant to guide a physician in a specialty in appropriate medication decisions, at the same time he hopes that “it opens a dialogue and communication with the other specialty who tends to oversee this particular disease state or class of medicine to really work together to try to find the right drug for the right person.”
For patients with a mild skin eruption and a controlled underlying disease, the algorithm recommends a “treat through” approach by continuing anti-TNF therapy and treating psoriasis symptoms with topical steroids, ultraviolet therapy, methotrexate, cyclosporine, or acitretin, and to consider dapsone in cases of pustular psoriasis. However, the researchers noted that “treat through” studies have reported complete symptom resolution in 26%-41% of patients.
For patients with recalcitrant or worsening TNFi-induced psoriasis or patients with mild skin eruptions with an uncontrolled underlying disease, the researchers proposed considering switching to a different anti-TNF therapy, although studies have shown complete resolution of symptoms in only 5%-37% of patients.
If patients worsen from there, or if they have moderate to-severe skin eruption with uncontrolled underlying disease, they could be considered for switching to a different drug class and treated based on their underlying disease, along with treatment for psoriasis symptoms. This approach has been shown to completely resolve lesions in up to 64% of cases, they said. IBD patients could benefit from ustekinumab, vedolizumab, 6-mercaptopurine, or azathioprine as an alternative to anti-TNF therapy. Those patients with psoriasis should be considered for guselkumab, while ustekinumab, ixekizumab, secukinumab, and apremilast are effective treatments for patients with psoriasis and PsA. Patients with RA could receive treatment with tocilizumab, rituximab, abatacept, and tofacitinib, the authors wrote.
Dr. Merola reported serving as a consultant and/or investigator for Merck Research Laboratories, AbbVie, Dermavant, Eli Lilly, Novartis, Janssen, UCB, Samumed, Celgene, Sanofi Regeneron, GlaxoSmithKline, Almirall, Sun Pharma, Biogen, Pfizer, Incyte, Aclaris, and Leo Pharma.
SOURCE: Li SJ et al. J Psoriasis Psoriatic Arthritis. 2018 Nov 21. doi: 10.1177/2475530318810851.
Patients with tumor necrosis factor inhibitor–induced psoriasis could potentially be switched to a different drug class if they have moderate to severe skin eruption or mild skin eruption with an uncontrolled underlying disease such as inflammatory bowel disease, psoriasis, psoriatic arthritis, or rheumatoid arthritis, according to a new treatment algorithm proposed by researchers from Brigham and Women’s Hospital and Harvard Medical School in Boston.
The researchers outlined the prevalence of tumor necrosis factor–alpha inhibitor (TNFi)-induced psoriasis in a literature review of inflammatory bowel disease (IBD), psoriasis, psoriatic arthritis (PsA), and rheumatoid arthritis (RA) and identified an estimated rate of between 2.3% and 5% in patients with RA and between 1.6% and 2.7% in patients with IBD. Although there have been reports of TNFi-induced psoriasis in patients with psoriasis and PsA, the prevalence is unclear, they wrote in the Journal of Psoriasis and Psoriatic Arthritis.
The authors then created an algorithm to manage and treat TNFi-induced psoriasiform skin eruptions with decisions to continue therapy and “treat through” symptoms, switch to a different anti-TNF therapy, or switch to a different drug class based on severity of symptoms, whether the underlying disease is well controlled, and how patients with those underlying diseases have fared with those specific therapies or agents.
“We’ve shifted gears over the past decade, and we’ve gone from having very few agents and trying to keep patients desperately on one or two agents because we didn’t want to have to give up on them for their other comorbid disease, whether it was Crohn’s, colitis, RA, or whatever it may be,” senior author Joseph Merola, MD, director of the Center for Skin and Related Musculoskeletal Diseases at Brigham and Women’s Hospital, Boston, said in an interview. “We’re now in an area where we can have an algorithm like this, and we have so many more mechanistic options to move to.”
Dr. Merola, who is board certified in dermatology and rheumatology, said the algorithm is meant to “open a dialogue” with other specialists in different areas and raise awareness of treatments in related but separate fields. For diseases not often seen by more than one specialty, with the exception of psoriasis and PsA, he said that “the idea is to start a dialogue and increase communication between specialists.”
Dr. Merola noted that while the algorithm in many respects is meant to guide a physician in a specialty in appropriate medication decisions, at the same time he hopes that “it opens a dialogue and communication with the other specialty who tends to oversee this particular disease state or class of medicine to really work together to try to find the right drug for the right person.”
For patients with a mild skin eruption and a controlled underlying disease, the algorithm recommends a “treat through” approach by continuing anti-TNF therapy and treating psoriasis symptoms with topical steroids, ultraviolet therapy, methotrexate, cyclosporine, or acitretin, and to consider dapsone in cases of pustular psoriasis. However, the researchers noted that “treat through” studies have reported complete symptom resolution in 26%-41% of patients.
For patients with recalcitrant or worsening TNFi-induced psoriasis or patients with mild skin eruptions with an uncontrolled underlying disease, the researchers proposed considering switching to a different anti-TNF therapy, although studies have shown complete resolution of symptoms in only 5%-37% of patients.
If patients worsen from there, or if they have moderate to-severe skin eruption with uncontrolled underlying disease, they could be considered for switching to a different drug class and treated based on their underlying disease, along with treatment for psoriasis symptoms. This approach has been shown to completely resolve lesions in up to 64% of cases, they said. IBD patients could benefit from ustekinumab, vedolizumab, 6-mercaptopurine, or azathioprine as an alternative to anti-TNF therapy. Those patients with psoriasis should be considered for guselkumab, while ustekinumab, ixekizumab, secukinumab, and apremilast are effective treatments for patients with psoriasis and PsA. Patients with RA could receive treatment with tocilizumab, rituximab, abatacept, and tofacitinib, the authors wrote.
Dr. Merola reported serving as a consultant and/or investigator for Merck Research Laboratories, AbbVie, Dermavant, Eli Lilly, Novartis, Janssen, UCB, Samumed, Celgene, Sanofi Regeneron, GlaxoSmithKline, Almirall, Sun Pharma, Biogen, Pfizer, Incyte, Aclaris, and Leo Pharma.
SOURCE: Li SJ et al. J Psoriasis Psoriatic Arthritis. 2018 Nov 21. doi: 10.1177/2475530318810851.
FROM JOURNAL OF PSORIASIS AND PSORIATIC ARTHRITIS
ACR, NPF unveil new psoriatic arthritis treatment guideline
The American College of Rheumatology and the National Psoriasis Foundation have released a joint treatment guideline for psoriatic arthritis that, for the first time, includes a conditional recommendation to use tumor necrosis factor–inhibitor(TNFi) biologics over methotrexate and other oral small molecules as a first-line therapy in patients with active disease.
“The available low-quality evidence is inconclusive regarding the efficacy of OSMs [oral small molecules] in management of PsA, whereas there is moderate-quality evidence of the benefits of TNFi biologics, in particular regarding their impact on the prevention of disease progression and joint damage,” wrote the panel of authors, who were led by Jasvinder A. Singh, MD, of the University of Alabama at Birmingham. “In making their recommendation, the panel recognized the cost implications, but put considerations of quality of evidence for benefit over other considerations. This guideline provides recommendations for early and aggressive therapy in patients with newly diagnosed PsA.”
The 28-page guideline, published online Nov. 30 in the Journal of Psoriasis and Psoriatic Arthritis and also in Arthritis Care & Research and Arthritis & Rheumatology, is the first set of PsA-specific recommendations to be assembled using the Grading of Recommendations Assessment, Development and Evaluation (GRADE) methodology that the ACR has used for RA and other conditions. GRADE uses systematic reviews of the scientific literature available to evaluate and grade the quality of evidence in a particular domain. The evidence reviews are then used to create guideline recommendations for or against particular therapy options that range from strong to conditional, depending on the quality of evidence available.
Based on the GRADE methodology and consensus building, the guideline authors crafted recommendations for eight different clinical scenarios, including the initial treatment of patients with active PsA who have not received either OSMs or other treatments; treatment of patients with active PsA despite treatment with an OSM; treatment of patients with active PsA despite treatment with a TNFi biologic either as monotherapy or in combination with methotrexate; treatment of patients with active PsA despite treatment with an interleukin (IL)-17 inhibitor or IL-12/23 inhibitor monotherapy; treatment of patients with active PsA including treat-to-target, active axial disease, enthesitis, or active inflammatory bowel disease; treatment of patients with active PsA and comorbidities, including concomitant diabetes and recurrent serious infections; vaccination in patients with active PsA; and treatment of patients with active PsA with nonpharmacologic interventions such as yoga and weight loss. Most of the treatment recommendations are conditional based on very low to moderate quality evidence. “Health care providers and patients must take into consideration all active disease domains, comorbidities, and the patient’s functional status in choosing the optimal therapy for an individual at a given point in time,” the authors emphasized.
Only five of the recommendations are listed as strong, including smoking cessation. Three of the strong recommendations concern adult patients with active PsA and concomitant active inflammatory bowel disease despite treatment with an OSM. They are “switch to a monoclonal antibody TNFi biologic over a TNFi biologic soluble receptor biologic,” “switch to a TNFi monoclonal antibody biologic over an IL-7i biologic,” and “switch to an IL-12/23i biologic over switching to an IL-17i biologic.”
The process of creating the guideline included input from a panel of nine adults who provided the authors with perspective on their values and preferences. “The concept of treat-to-target was challenging for patients,” the authors noted. “Although they saw value in improved outcomes, they also thought this strategy could increase costs to the patient (e.g., copayments, time traveling to more frequent appointments, etc.) and potentially increase adverse events. Therefore, a detailed conversation with the patient is needed to make decisions regarding treat-to-target.”
The authors concluded the guideline by calling for more comparative data to inform treatment selection in the future. “Several ongoing trials, including a trial to compare a TNFi biologic combination therapy with a TNFi biologic monotherapy and MTX monotherapy, will inform treatment decisions,” they wrote. “We anticipate future updates to the guideline when new evidence is available.”
Dr. Singh, who is also a staff rheumatologist at the Birmingham (Ala.) Veterans Affairs Medical Center, led development of the 2012 and 2015 ACR treatment guidelines for RA. He has received consulting fees from a variety of companies marketing rheumatologic drugs as well as research support from Takeda and Savient. The other guideline authors reported having numerous financial ties to industry.
SOURCE: Singh J et al. Arthritis Care Res. 2018 Nov 30. doi: 10.1002/acr.23789.
The American College of Rheumatology and the National Psoriasis Foundation have released a joint treatment guideline for psoriatic arthritis that, for the first time, includes a conditional recommendation to use tumor necrosis factor–inhibitor(TNFi) biologics over methotrexate and other oral small molecules as a first-line therapy in patients with active disease.
“The available low-quality evidence is inconclusive regarding the efficacy of OSMs [oral small molecules] in management of PsA, whereas there is moderate-quality evidence of the benefits of TNFi biologics, in particular regarding their impact on the prevention of disease progression and joint damage,” wrote the panel of authors, who were led by Jasvinder A. Singh, MD, of the University of Alabama at Birmingham. “In making their recommendation, the panel recognized the cost implications, but put considerations of quality of evidence for benefit over other considerations. This guideline provides recommendations for early and aggressive therapy in patients with newly diagnosed PsA.”
The 28-page guideline, published online Nov. 30 in the Journal of Psoriasis and Psoriatic Arthritis and also in Arthritis Care & Research and Arthritis & Rheumatology, is the first set of PsA-specific recommendations to be assembled using the Grading of Recommendations Assessment, Development and Evaluation (GRADE) methodology that the ACR has used for RA and other conditions. GRADE uses systematic reviews of the scientific literature available to evaluate and grade the quality of evidence in a particular domain. The evidence reviews are then used to create guideline recommendations for or against particular therapy options that range from strong to conditional, depending on the quality of evidence available.
Based on the GRADE methodology and consensus building, the guideline authors crafted recommendations for eight different clinical scenarios, including the initial treatment of patients with active PsA who have not received either OSMs or other treatments; treatment of patients with active PsA despite treatment with an OSM; treatment of patients with active PsA despite treatment with a TNFi biologic either as monotherapy or in combination with methotrexate; treatment of patients with active PsA despite treatment with an interleukin (IL)-17 inhibitor or IL-12/23 inhibitor monotherapy; treatment of patients with active PsA including treat-to-target, active axial disease, enthesitis, or active inflammatory bowel disease; treatment of patients with active PsA and comorbidities, including concomitant diabetes and recurrent serious infections; vaccination in patients with active PsA; and treatment of patients with active PsA with nonpharmacologic interventions such as yoga and weight loss. Most of the treatment recommendations are conditional based on very low to moderate quality evidence. “Health care providers and patients must take into consideration all active disease domains, comorbidities, and the patient’s functional status in choosing the optimal therapy for an individual at a given point in time,” the authors emphasized.
Only five of the recommendations are listed as strong, including smoking cessation. Three of the strong recommendations concern adult patients with active PsA and concomitant active inflammatory bowel disease despite treatment with an OSM. They are “switch to a monoclonal antibody TNFi biologic over a TNFi biologic soluble receptor biologic,” “switch to a TNFi monoclonal antibody biologic over an IL-7i biologic,” and “switch to an IL-12/23i biologic over switching to an IL-17i biologic.”
The process of creating the guideline included input from a panel of nine adults who provided the authors with perspective on their values and preferences. “The concept of treat-to-target was challenging for patients,” the authors noted. “Although they saw value in improved outcomes, they also thought this strategy could increase costs to the patient (e.g., copayments, time traveling to more frequent appointments, etc.) and potentially increase adverse events. Therefore, a detailed conversation with the patient is needed to make decisions regarding treat-to-target.”
The authors concluded the guideline by calling for more comparative data to inform treatment selection in the future. “Several ongoing trials, including a trial to compare a TNFi biologic combination therapy with a TNFi biologic monotherapy and MTX monotherapy, will inform treatment decisions,” they wrote. “We anticipate future updates to the guideline when new evidence is available.”
Dr. Singh, who is also a staff rheumatologist at the Birmingham (Ala.) Veterans Affairs Medical Center, led development of the 2012 and 2015 ACR treatment guidelines for RA. He has received consulting fees from a variety of companies marketing rheumatologic drugs as well as research support from Takeda and Savient. The other guideline authors reported having numerous financial ties to industry.
SOURCE: Singh J et al. Arthritis Care Res. 2018 Nov 30. doi: 10.1002/acr.23789.
The American College of Rheumatology and the National Psoriasis Foundation have released a joint treatment guideline for psoriatic arthritis that, for the first time, includes a conditional recommendation to use tumor necrosis factor–inhibitor(TNFi) biologics over methotrexate and other oral small molecules as a first-line therapy in patients with active disease.
“The available low-quality evidence is inconclusive regarding the efficacy of OSMs [oral small molecules] in management of PsA, whereas there is moderate-quality evidence of the benefits of TNFi biologics, in particular regarding their impact on the prevention of disease progression and joint damage,” wrote the panel of authors, who were led by Jasvinder A. Singh, MD, of the University of Alabama at Birmingham. “In making their recommendation, the panel recognized the cost implications, but put considerations of quality of evidence for benefit over other considerations. This guideline provides recommendations for early and aggressive therapy in patients with newly diagnosed PsA.”
The 28-page guideline, published online Nov. 30 in the Journal of Psoriasis and Psoriatic Arthritis and also in Arthritis Care & Research and Arthritis & Rheumatology, is the first set of PsA-specific recommendations to be assembled using the Grading of Recommendations Assessment, Development and Evaluation (GRADE) methodology that the ACR has used for RA and other conditions. GRADE uses systematic reviews of the scientific literature available to evaluate and grade the quality of evidence in a particular domain. The evidence reviews are then used to create guideline recommendations for or against particular therapy options that range from strong to conditional, depending on the quality of evidence available.
Based on the GRADE methodology and consensus building, the guideline authors crafted recommendations for eight different clinical scenarios, including the initial treatment of patients with active PsA who have not received either OSMs or other treatments; treatment of patients with active PsA despite treatment with an OSM; treatment of patients with active PsA despite treatment with a TNFi biologic either as monotherapy or in combination with methotrexate; treatment of patients with active PsA despite treatment with an interleukin (IL)-17 inhibitor or IL-12/23 inhibitor monotherapy; treatment of patients with active PsA including treat-to-target, active axial disease, enthesitis, or active inflammatory bowel disease; treatment of patients with active PsA and comorbidities, including concomitant diabetes and recurrent serious infections; vaccination in patients with active PsA; and treatment of patients with active PsA with nonpharmacologic interventions such as yoga and weight loss. Most of the treatment recommendations are conditional based on very low to moderate quality evidence. “Health care providers and patients must take into consideration all active disease domains, comorbidities, and the patient’s functional status in choosing the optimal therapy for an individual at a given point in time,” the authors emphasized.
Only five of the recommendations are listed as strong, including smoking cessation. Three of the strong recommendations concern adult patients with active PsA and concomitant active inflammatory bowel disease despite treatment with an OSM. They are “switch to a monoclonal antibody TNFi biologic over a TNFi biologic soluble receptor biologic,” “switch to a TNFi monoclonal antibody biologic over an IL-7i biologic,” and “switch to an IL-12/23i biologic over switching to an IL-17i biologic.”
The process of creating the guideline included input from a panel of nine adults who provided the authors with perspective on their values and preferences. “The concept of treat-to-target was challenging for patients,” the authors noted. “Although they saw value in improved outcomes, they also thought this strategy could increase costs to the patient (e.g., copayments, time traveling to more frequent appointments, etc.) and potentially increase adverse events. Therefore, a detailed conversation with the patient is needed to make decisions regarding treat-to-target.”
The authors concluded the guideline by calling for more comparative data to inform treatment selection in the future. “Several ongoing trials, including a trial to compare a TNFi biologic combination therapy with a TNFi biologic monotherapy and MTX monotherapy, will inform treatment decisions,” they wrote. “We anticipate future updates to the guideline when new evidence is available.”
Dr. Singh, who is also a staff rheumatologist at the Birmingham (Ala.) Veterans Affairs Medical Center, led development of the 2012 and 2015 ACR treatment guidelines for RA. He has received consulting fees from a variety of companies marketing rheumatologic drugs as well as research support from Takeda and Savient. The other guideline authors reported having numerous financial ties to industry.
SOURCE: Singh J et al. Arthritis Care Res. 2018 Nov 30. doi: 10.1002/acr.23789.
FROM ARTHRITIS CARE & RESEARCH
Filgotinib shows efficacy, safety in RA phase 3 and PsA phase 2 trials
CHICAGO – The selective Janus kinase 1 inhibitor filgotinib showed efficacy and safety for patients with rheumatoid arthritis in a phase 3 trial, and efficacy and safety for treating patients with psoriatic arthritis in results from a phase 2 study in two separate reports at the annual meeting of the American College of Rheumatology.
In the phase 3 study, treatment with filgotinib at an oral dosage of 200 mg once daily led to a 66% incidence of American College of Rheumatology 20 (ACR20) responses after 16 weeks of treatment in 147 patients with moderately to severely active rheumatoid arthritis (RA), compared with a 31% rate among 148 patients randomized to receive placebo, a statistically significant improvement for the study’s primary efficacy endpoint, Mark C. Genovese, MD, reported in a poster at the meeting. The rate of ACR20 responses among the 153 RA patients who received 100 mg/day filgotinib was 58%, reported Dr. Genovese, professor of medicine and director of the rheumatology clinic at Stanford (Calif.) University.
After 24 weeks of daily treatment, the longest duration studied in the trial, ACR20 rates were 69%, 55%, and 35% in the 200-mg, 100-mg, and placebo patients, respectively. Dr. Genovese also reported that after 24 weeks on treatment, the rates of patients achieving low disease activity measured by their disease activity score based on 28 joints and C-reactive protein level (DAS28-CRP) were 48%, 38%, and 21%, respectively, and the percentages of patients achieving complete remission at 24 weeks based on their DAS28-CRP scores were 31%, 26%, and 12%, respectively.
“We were incredibly fortunate to see such positive results. The drug worked very well in very-challenging-to-treat patients,” Dr. Genovese said in an interview. All of the RA patients enrolled in the study had not previously responded to or were intolerant of prior treatment with at least one biologic disease-modifying antirheumatic drug (DMARD), and almost a quarter of enrolled patients had failed prior treatment with at least three different biologic DMARDs. The number of biologic DMARDs a patient had previously received showed no relationship to how well patients responded to filgotinib, he noted.
Dr. Genovese also highlighted the relatively high percentage of patients who achieved low disease activity and remission. The 48% and 31% rates, respectively, of low disease activity and remission among patients treated with the higher filgotinib dosage for 24 weeks “is fairly impressive in patients who did not previously respond to a biologic DMARD,” the researcher said. These findings are similar to data previously reported for upadacitinib, another Janus kinase (JAK) inhibitor that, like filgotinib, is selective for the JAK1 receptor, noted Dr. Genovese, who also was the lead investigator for a phase 3 study of upadacitinib in RA patients (Lancet. 2018 June 23;391[10139]:2513-24).
The filgotinib data he presented came from the FINCH 2 (Filgotinib Versus Placebo in Adults With Active Rheumatoid Arthritis Who Have an Inadequate Response to Biologic Disease-Modifying Anti-Rheumatic Drug[s] Treatment) trial, which was run at 104 sites in 15 countries, including the United States. The results also showed a “favorable safety profile and stable laboratory parameters,” Dr. Genovese reported. Results from two additional phase 3 trials in RA patients are expected in 2019, he said.
Filgotinib studied in psoriatic arthritis
The separate, phase 2 study of filgotinib in patients with psoriatic arthritis (PsA) reported during the meeting showed safety “in line with previous reports without new safety signals” in a multicenter trial with 131 patients randomized to receive oral filgotinib 200 mg daily for 16 weeks or placebo, Philip J. Mease, MD, reported in a talk at the meeting. For the primary endpoint of achievement of ACR20 response after 16 weeks, the rate was 80% of the filgotinib-treated patients and 33% of patients in the placebo group, a statistically significant difference, said Dr. Mease, a rheumatologist at Swedish Medical Center in Seattle.
EQUATOR (A Study to Assess Efficacy and Safety of Filgotinib in Active Psoriatic Arthritis) enrolled patients at sites in seven European countries who had “very active” PsA and either a history of or current plaque psoriasis. All patients had to have a history of either insufficient response to or intolerance of at least one conventional synthetic DMARD. The enrollment criteria had no specifications for prior use of an anti–tumor necrosis factor drug, and about 15% of patients had used least one of these drugs. At entry, about three-quarters of patients were on treatment with a conventional synthetic DMARD and about a quarter received treatment with a glucocorticoid.
The results showed statistically significant benefits from filgotinib, compared with placebo, for several other measures of arthritis activity, as well as measures of psoriasis, enthesitis, and pain, Dr. Mease reported. He also highlighted a “lack of meaningful changes in hemoglobin” or other laboratory measures that, along with the efficacy findings, make filgotinib “a promising first step” for patients with PsA. Dr. Mease also noted that roughly concurrently with his report, a separate group of researchers published results from a phase 2 study of filgotinib in patients with ankylosing spondylitis that also found evidence for efficacy and safety during 12 weeks of treating 116 randomized patients (Lancet. 2018 Oct 22. doi: 10.1016/S0140-6736[18]32463-2).
FINCH 2 was sponsored by Galapagos and Gilead, the two companies developing filgotinib. Dr. Genovese has had financial relationships with Galapagos and Gilead and also with AbbVie, Lilly, and Pfizer. Dr. Mease has had financial relationships with Galapagos and Gilead and a dozen other companies.
SOURCES: Genovese M et al. Arthritis Rheumatol. 2018;70(Suppl 10), Abstract L06; Mease P et al. Arthritis Rheumatol. 2018;70(Suppl 10), Abstract 1821.
CHICAGO – The selective Janus kinase 1 inhibitor filgotinib showed efficacy and safety for patients with rheumatoid arthritis in a phase 3 trial, and efficacy and safety for treating patients with psoriatic arthritis in results from a phase 2 study in two separate reports at the annual meeting of the American College of Rheumatology.
In the phase 3 study, treatment with filgotinib at an oral dosage of 200 mg once daily led to a 66% incidence of American College of Rheumatology 20 (ACR20) responses after 16 weeks of treatment in 147 patients with moderately to severely active rheumatoid arthritis (RA), compared with a 31% rate among 148 patients randomized to receive placebo, a statistically significant improvement for the study’s primary efficacy endpoint, Mark C. Genovese, MD, reported in a poster at the meeting. The rate of ACR20 responses among the 153 RA patients who received 100 mg/day filgotinib was 58%, reported Dr. Genovese, professor of medicine and director of the rheumatology clinic at Stanford (Calif.) University.
After 24 weeks of daily treatment, the longest duration studied in the trial, ACR20 rates were 69%, 55%, and 35% in the 200-mg, 100-mg, and placebo patients, respectively. Dr. Genovese also reported that after 24 weeks on treatment, the rates of patients achieving low disease activity measured by their disease activity score based on 28 joints and C-reactive protein level (DAS28-CRP) were 48%, 38%, and 21%, respectively, and the percentages of patients achieving complete remission at 24 weeks based on their DAS28-CRP scores were 31%, 26%, and 12%, respectively.
“We were incredibly fortunate to see such positive results. The drug worked very well in very-challenging-to-treat patients,” Dr. Genovese said in an interview. All of the RA patients enrolled in the study had not previously responded to or were intolerant of prior treatment with at least one biologic disease-modifying antirheumatic drug (DMARD), and almost a quarter of enrolled patients had failed prior treatment with at least three different biologic DMARDs. The number of biologic DMARDs a patient had previously received showed no relationship to how well patients responded to filgotinib, he noted.
Dr. Genovese also highlighted the relatively high percentage of patients who achieved low disease activity and remission. The 48% and 31% rates, respectively, of low disease activity and remission among patients treated with the higher filgotinib dosage for 24 weeks “is fairly impressive in patients who did not previously respond to a biologic DMARD,” the researcher said. These findings are similar to data previously reported for upadacitinib, another Janus kinase (JAK) inhibitor that, like filgotinib, is selective for the JAK1 receptor, noted Dr. Genovese, who also was the lead investigator for a phase 3 study of upadacitinib in RA patients (Lancet. 2018 June 23;391[10139]:2513-24).
The filgotinib data he presented came from the FINCH 2 (Filgotinib Versus Placebo in Adults With Active Rheumatoid Arthritis Who Have an Inadequate Response to Biologic Disease-Modifying Anti-Rheumatic Drug[s] Treatment) trial, which was run at 104 sites in 15 countries, including the United States. The results also showed a “favorable safety profile and stable laboratory parameters,” Dr. Genovese reported. Results from two additional phase 3 trials in RA patients are expected in 2019, he said.
Filgotinib studied in psoriatic arthritis
The separate, phase 2 study of filgotinib in patients with psoriatic arthritis (PsA) reported during the meeting showed safety “in line with previous reports without new safety signals” in a multicenter trial with 131 patients randomized to receive oral filgotinib 200 mg daily for 16 weeks or placebo, Philip J. Mease, MD, reported in a talk at the meeting. For the primary endpoint of achievement of ACR20 response after 16 weeks, the rate was 80% of the filgotinib-treated patients and 33% of patients in the placebo group, a statistically significant difference, said Dr. Mease, a rheumatologist at Swedish Medical Center in Seattle.
EQUATOR (A Study to Assess Efficacy and Safety of Filgotinib in Active Psoriatic Arthritis) enrolled patients at sites in seven European countries who had “very active” PsA and either a history of or current plaque psoriasis. All patients had to have a history of either insufficient response to or intolerance of at least one conventional synthetic DMARD. The enrollment criteria had no specifications for prior use of an anti–tumor necrosis factor drug, and about 15% of patients had used least one of these drugs. At entry, about three-quarters of patients were on treatment with a conventional synthetic DMARD and about a quarter received treatment with a glucocorticoid.
The results showed statistically significant benefits from filgotinib, compared with placebo, for several other measures of arthritis activity, as well as measures of psoriasis, enthesitis, and pain, Dr. Mease reported. He also highlighted a “lack of meaningful changes in hemoglobin” or other laboratory measures that, along with the efficacy findings, make filgotinib “a promising first step” for patients with PsA. Dr. Mease also noted that roughly concurrently with his report, a separate group of researchers published results from a phase 2 study of filgotinib in patients with ankylosing spondylitis that also found evidence for efficacy and safety during 12 weeks of treating 116 randomized patients (Lancet. 2018 Oct 22. doi: 10.1016/S0140-6736[18]32463-2).
FINCH 2 was sponsored by Galapagos and Gilead, the two companies developing filgotinib. Dr. Genovese has had financial relationships with Galapagos and Gilead and also with AbbVie, Lilly, and Pfizer. Dr. Mease has had financial relationships with Galapagos and Gilead and a dozen other companies.
SOURCES: Genovese M et al. Arthritis Rheumatol. 2018;70(Suppl 10), Abstract L06; Mease P et al. Arthritis Rheumatol. 2018;70(Suppl 10), Abstract 1821.
CHICAGO – The selective Janus kinase 1 inhibitor filgotinib showed efficacy and safety for patients with rheumatoid arthritis in a phase 3 trial, and efficacy and safety for treating patients with psoriatic arthritis in results from a phase 2 study in two separate reports at the annual meeting of the American College of Rheumatology.
In the phase 3 study, treatment with filgotinib at an oral dosage of 200 mg once daily led to a 66% incidence of American College of Rheumatology 20 (ACR20) responses after 16 weeks of treatment in 147 patients with moderately to severely active rheumatoid arthritis (RA), compared with a 31% rate among 148 patients randomized to receive placebo, a statistically significant improvement for the study’s primary efficacy endpoint, Mark C. Genovese, MD, reported in a poster at the meeting. The rate of ACR20 responses among the 153 RA patients who received 100 mg/day filgotinib was 58%, reported Dr. Genovese, professor of medicine and director of the rheumatology clinic at Stanford (Calif.) University.
After 24 weeks of daily treatment, the longest duration studied in the trial, ACR20 rates were 69%, 55%, and 35% in the 200-mg, 100-mg, and placebo patients, respectively. Dr. Genovese also reported that after 24 weeks on treatment, the rates of patients achieving low disease activity measured by their disease activity score based on 28 joints and C-reactive protein level (DAS28-CRP) were 48%, 38%, and 21%, respectively, and the percentages of patients achieving complete remission at 24 weeks based on their DAS28-CRP scores were 31%, 26%, and 12%, respectively.
“We were incredibly fortunate to see such positive results. The drug worked very well in very-challenging-to-treat patients,” Dr. Genovese said in an interview. All of the RA patients enrolled in the study had not previously responded to or were intolerant of prior treatment with at least one biologic disease-modifying antirheumatic drug (DMARD), and almost a quarter of enrolled patients had failed prior treatment with at least three different biologic DMARDs. The number of biologic DMARDs a patient had previously received showed no relationship to how well patients responded to filgotinib, he noted.
Dr. Genovese also highlighted the relatively high percentage of patients who achieved low disease activity and remission. The 48% and 31% rates, respectively, of low disease activity and remission among patients treated with the higher filgotinib dosage for 24 weeks “is fairly impressive in patients who did not previously respond to a biologic DMARD,” the researcher said. These findings are similar to data previously reported for upadacitinib, another Janus kinase (JAK) inhibitor that, like filgotinib, is selective for the JAK1 receptor, noted Dr. Genovese, who also was the lead investigator for a phase 3 study of upadacitinib in RA patients (Lancet. 2018 June 23;391[10139]:2513-24).
The filgotinib data he presented came from the FINCH 2 (Filgotinib Versus Placebo in Adults With Active Rheumatoid Arthritis Who Have an Inadequate Response to Biologic Disease-Modifying Anti-Rheumatic Drug[s] Treatment) trial, which was run at 104 sites in 15 countries, including the United States. The results also showed a “favorable safety profile and stable laboratory parameters,” Dr. Genovese reported. Results from two additional phase 3 trials in RA patients are expected in 2019, he said.
Filgotinib studied in psoriatic arthritis
The separate, phase 2 study of filgotinib in patients with psoriatic arthritis (PsA) reported during the meeting showed safety “in line with previous reports without new safety signals” in a multicenter trial with 131 patients randomized to receive oral filgotinib 200 mg daily for 16 weeks or placebo, Philip J. Mease, MD, reported in a talk at the meeting. For the primary endpoint of achievement of ACR20 response after 16 weeks, the rate was 80% of the filgotinib-treated patients and 33% of patients in the placebo group, a statistically significant difference, said Dr. Mease, a rheumatologist at Swedish Medical Center in Seattle.
EQUATOR (A Study to Assess Efficacy and Safety of Filgotinib in Active Psoriatic Arthritis) enrolled patients at sites in seven European countries who had “very active” PsA and either a history of or current plaque psoriasis. All patients had to have a history of either insufficient response to or intolerance of at least one conventional synthetic DMARD. The enrollment criteria had no specifications for prior use of an anti–tumor necrosis factor drug, and about 15% of patients had used least one of these drugs. At entry, about three-quarters of patients were on treatment with a conventional synthetic DMARD and about a quarter received treatment with a glucocorticoid.
The results showed statistically significant benefits from filgotinib, compared with placebo, for several other measures of arthritis activity, as well as measures of psoriasis, enthesitis, and pain, Dr. Mease reported. He also highlighted a “lack of meaningful changes in hemoglobin” or other laboratory measures that, along with the efficacy findings, make filgotinib “a promising first step” for patients with PsA. Dr. Mease also noted that roughly concurrently with his report, a separate group of researchers published results from a phase 2 study of filgotinib in patients with ankylosing spondylitis that also found evidence for efficacy and safety during 12 weeks of treating 116 randomized patients (Lancet. 2018 Oct 22. doi: 10.1016/S0140-6736[18]32463-2).
FINCH 2 was sponsored by Galapagos and Gilead, the two companies developing filgotinib. Dr. Genovese has had financial relationships with Galapagos and Gilead and also with AbbVie, Lilly, and Pfizer. Dr. Mease has had financial relationships with Galapagos and Gilead and a dozen other companies.
SOURCES: Genovese M et al. Arthritis Rheumatol. 2018;70(Suppl 10), Abstract L06; Mease P et al. Arthritis Rheumatol. 2018;70(Suppl 10), Abstract 1821.
REPORTING FROM THE ACR ANNUAL MEETING
Weight loss cuts risk of psoriatic arthritis
CHICAGO – Overweight and obese psoriasis patients have it within their power to reduce their risk of developing psoriatic arthritis through weight loss, according to a large British longitudinal study.
Of the three modifiable lifestyle factors evaluated in the study as potential risk factors for the development of psoriatic arthritis in psoriasis patients – body mass index, smoking, and alcohol intake – reduction in BMI over time was clearly the winning strategy, Neil McHugh, MD, said at the annual meeting of the American College of Rheumatology.
The message from this study of 90,189 incident cases of psoriasis identified in the U.K. Clinical Practice Research Datalink was unequivocal: “If you’re overweight and have psoriasis and you lose weight, you reduce your chance of developing a nasty form of arthritis,” said Dr. McHugh, professor of pharmacoepidemiology and a rheumatologist at the University of Bath, England.
“As psoriatic arthritis affects around 20% of people with psoriasis, weight reduction amongst those who are obese may have the potential to greatly reduce their risk of psoriatic arthritis in addition to providing additional health benefits,” he added.
Among the more than 90,000 patients diagnosed with psoriasis, 1,409 subsequently developed psoriatic arthritis, with an overall incidence rate of 2.72 cases per 1,000 person-years. Baseline BMI was strongly associated in stepwise fashion with subsequent psoriatic arthritis. Psoriasis patients with a baseline BMI of 25-29.9 kg/m2 were at an adjusted 1.76-fold increased risk of later developing psoriatic arthritis, compared with psoriasis patients having a BMI of less than 25. For those with a BMI of 30-34.9 kg/m2, the risk of subsequent psoriatic arthritis was increased 2.04-fold. And for those with a baseline BMI of 35 kg/m2 or more, the risk was increased 2.42-fold in analyses adjusted for age, sex, psoriasis duration and severity, history of trauma, and diabetes.
In contrast, the risk of developing psoriatic arthritis wasn’t significantly different between psoriasis patients who were nonsmokers, ex-smokers, or current smokers. And while there was a significantly increased risk of developing psoriatic arthritis in psoriasis patients who were current drinkers, compared with nondrinkers, the risk in ex-drinkers and heavy drinkers was similar to that in nondrinkers, a counterintuitive finding Dr. McHugh suspects was a distortion due to small numbers.
While the observed relationship between baseline BMI and subsequent risk of psoriatic arthritis was informative, it only tells part of the story, since body weight so often changes over time. Dr. McHugh and his coinvestigators had data on change in BMI over the course of 10 years of follow-up in 15,627 psoriasis patients free of psoriatic arthritis at the time their psoriasis was diagnosed. The researchers developed a BMI risk calculator that expressed the effect of change in BMI over time on the cumulative risk of developing psoriatic arthritis.
“We were able to show that if, for instance, you started with a BMI of 25 at baseline and ended up with a BMI of 30, your risk of psoriatic arthritis goes up by 13%, whereas if you start at 30 and come down to 25, your risk decreases by 13%. And the more weight you lose, the greater you reduce your risk of developing psoriatic arthritis,” the rheumatologist explained in an interview.
Indeed, with more extreme changes in BMI over the course of a decade following diagnosis of psoriasis – for example, dropping from a baseline BMI of 36 kg/m2 to 23 kg/m2 – the risk of developing psoriatic arthritis fell by close to 30%.
Dr. McHugh reported having no financial conflicts regarding this study, funded by the U.K. National Institute for Health Research.
SOURCE: Green A et al. Arthritis Rheumatol. 2018;70(Suppl 10): Abstract 2134.
CHICAGO – Overweight and obese psoriasis patients have it within their power to reduce their risk of developing psoriatic arthritis through weight loss, according to a large British longitudinal study.
Of the three modifiable lifestyle factors evaluated in the study as potential risk factors for the development of psoriatic arthritis in psoriasis patients – body mass index, smoking, and alcohol intake – reduction in BMI over time was clearly the winning strategy, Neil McHugh, MD, said at the annual meeting of the American College of Rheumatology.
The message from this study of 90,189 incident cases of psoriasis identified in the U.K. Clinical Practice Research Datalink was unequivocal: “If you’re overweight and have psoriasis and you lose weight, you reduce your chance of developing a nasty form of arthritis,” said Dr. McHugh, professor of pharmacoepidemiology and a rheumatologist at the University of Bath, England.
“As psoriatic arthritis affects around 20% of people with psoriasis, weight reduction amongst those who are obese may have the potential to greatly reduce their risk of psoriatic arthritis in addition to providing additional health benefits,” he added.
Among the more than 90,000 patients diagnosed with psoriasis, 1,409 subsequently developed psoriatic arthritis, with an overall incidence rate of 2.72 cases per 1,000 person-years. Baseline BMI was strongly associated in stepwise fashion with subsequent psoriatic arthritis. Psoriasis patients with a baseline BMI of 25-29.9 kg/m2 were at an adjusted 1.76-fold increased risk of later developing psoriatic arthritis, compared with psoriasis patients having a BMI of less than 25. For those with a BMI of 30-34.9 kg/m2, the risk of subsequent psoriatic arthritis was increased 2.04-fold. And for those with a baseline BMI of 35 kg/m2 or more, the risk was increased 2.42-fold in analyses adjusted for age, sex, psoriasis duration and severity, history of trauma, and diabetes.
In contrast, the risk of developing psoriatic arthritis wasn’t significantly different between psoriasis patients who were nonsmokers, ex-smokers, or current smokers. And while there was a significantly increased risk of developing psoriatic arthritis in psoriasis patients who were current drinkers, compared with nondrinkers, the risk in ex-drinkers and heavy drinkers was similar to that in nondrinkers, a counterintuitive finding Dr. McHugh suspects was a distortion due to small numbers.
While the observed relationship between baseline BMI and subsequent risk of psoriatic arthritis was informative, it only tells part of the story, since body weight so often changes over time. Dr. McHugh and his coinvestigators had data on change in BMI over the course of 10 years of follow-up in 15,627 psoriasis patients free of psoriatic arthritis at the time their psoriasis was diagnosed. The researchers developed a BMI risk calculator that expressed the effect of change in BMI over time on the cumulative risk of developing psoriatic arthritis.
“We were able to show that if, for instance, you started with a BMI of 25 at baseline and ended up with a BMI of 30, your risk of psoriatic arthritis goes up by 13%, whereas if you start at 30 and come down to 25, your risk decreases by 13%. And the more weight you lose, the greater you reduce your risk of developing psoriatic arthritis,” the rheumatologist explained in an interview.
Indeed, with more extreme changes in BMI over the course of a decade following diagnosis of psoriasis – for example, dropping from a baseline BMI of 36 kg/m2 to 23 kg/m2 – the risk of developing psoriatic arthritis fell by close to 30%.
Dr. McHugh reported having no financial conflicts regarding this study, funded by the U.K. National Institute for Health Research.
SOURCE: Green A et al. Arthritis Rheumatol. 2018;70(Suppl 10): Abstract 2134.
CHICAGO – Overweight and obese psoriasis patients have it within their power to reduce their risk of developing psoriatic arthritis through weight loss, according to a large British longitudinal study.
Of the three modifiable lifestyle factors evaluated in the study as potential risk factors for the development of psoriatic arthritis in psoriasis patients – body mass index, smoking, and alcohol intake – reduction in BMI over time was clearly the winning strategy, Neil McHugh, MD, said at the annual meeting of the American College of Rheumatology.
The message from this study of 90,189 incident cases of psoriasis identified in the U.K. Clinical Practice Research Datalink was unequivocal: “If you’re overweight and have psoriasis and you lose weight, you reduce your chance of developing a nasty form of arthritis,” said Dr. McHugh, professor of pharmacoepidemiology and a rheumatologist at the University of Bath, England.
“As psoriatic arthritis affects around 20% of people with psoriasis, weight reduction amongst those who are obese may have the potential to greatly reduce their risk of psoriatic arthritis in addition to providing additional health benefits,” he added.
Among the more than 90,000 patients diagnosed with psoriasis, 1,409 subsequently developed psoriatic arthritis, with an overall incidence rate of 2.72 cases per 1,000 person-years. Baseline BMI was strongly associated in stepwise fashion with subsequent psoriatic arthritis. Psoriasis patients with a baseline BMI of 25-29.9 kg/m2 were at an adjusted 1.76-fold increased risk of later developing psoriatic arthritis, compared with psoriasis patients having a BMI of less than 25. For those with a BMI of 30-34.9 kg/m2, the risk of subsequent psoriatic arthritis was increased 2.04-fold. And for those with a baseline BMI of 35 kg/m2 or more, the risk was increased 2.42-fold in analyses adjusted for age, sex, psoriasis duration and severity, history of trauma, and diabetes.
In contrast, the risk of developing psoriatic arthritis wasn’t significantly different between psoriasis patients who were nonsmokers, ex-smokers, or current smokers. And while there was a significantly increased risk of developing psoriatic arthritis in psoriasis patients who were current drinkers, compared with nondrinkers, the risk in ex-drinkers and heavy drinkers was similar to that in nondrinkers, a counterintuitive finding Dr. McHugh suspects was a distortion due to small numbers.
While the observed relationship between baseline BMI and subsequent risk of psoriatic arthritis was informative, it only tells part of the story, since body weight so often changes over time. Dr. McHugh and his coinvestigators had data on change in BMI over the course of 10 years of follow-up in 15,627 psoriasis patients free of psoriatic arthritis at the time their psoriasis was diagnosed. The researchers developed a BMI risk calculator that expressed the effect of change in BMI over time on the cumulative risk of developing psoriatic arthritis.
“We were able to show that if, for instance, you started with a BMI of 25 at baseline and ended up with a BMI of 30, your risk of psoriatic arthritis goes up by 13%, whereas if you start at 30 and come down to 25, your risk decreases by 13%. And the more weight you lose, the greater you reduce your risk of developing psoriatic arthritis,” the rheumatologist explained in an interview.
Indeed, with more extreme changes in BMI over the course of a decade following diagnosis of psoriasis – for example, dropping from a baseline BMI of 36 kg/m2 to 23 kg/m2 – the risk of developing psoriatic arthritis fell by close to 30%.
Dr. McHugh reported having no financial conflicts regarding this study, funded by the U.K. National Institute for Health Research.
SOURCE: Green A et al. Arthritis Rheumatol. 2018;70(Suppl 10): Abstract 2134.
REPORTING FROM THE ACR ANNUAL MEETING
Key clinical point:
Major finding: A psoriasis patient’s risk of developing psoriatic arthritis increases stepwise with greater body mass index, and the converse is true as well.
Study details: This study included more than 90,000 patients with a diagnosis of psoriasis in the U.K. Clinical Practice Research Datalink.
Disclosures: The presenter reported having no financial conflicts regarding this study, funded by the U.K. National Institute for Health Research.
Source: Green A et al. Arthritis Rheumatol. 2018;70(Suppl 10): Abstract 2134.
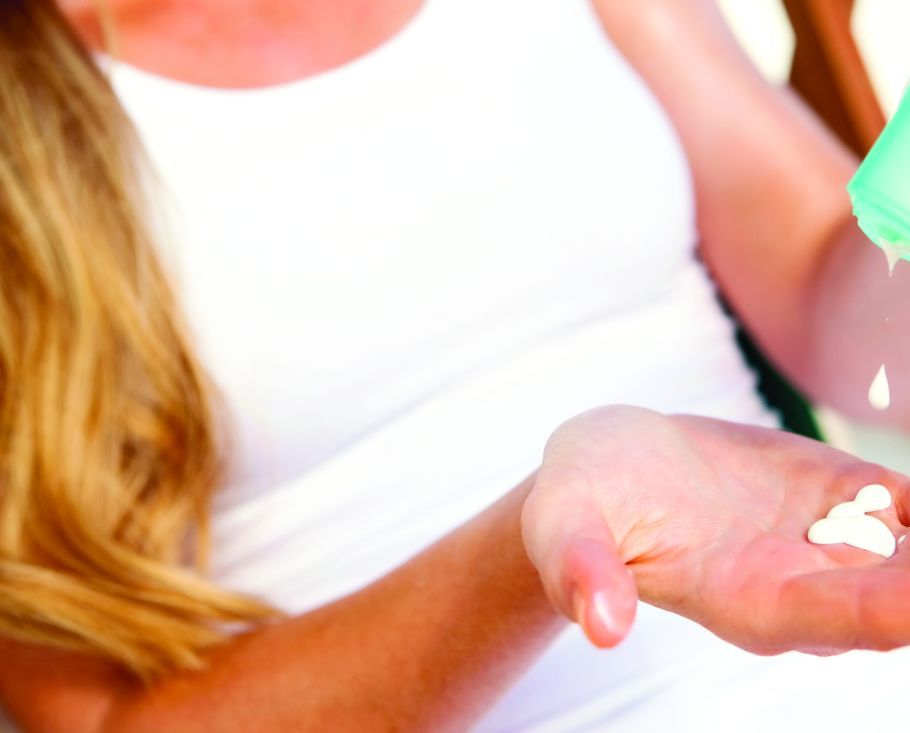
Topical treatments remain a good option for psoriasis
LAS VEGAS – in mild cases of psoriasis.
Topicals often are a worthwhile complement to even the most advanced systemic medications, according to Linda Stein Gold, MD, director of clinical research in the department of dermatology at the Henry Ford Health System, Detroit.
Speaking at the Skin Disease Education Foundation’s annual Las Vegas Dermatology Seminar, she pointed out that as the variety of vehicles for topical treatments has grown, so has the need to pay attention to the potency of these treatments. “Traditionally, we had thought we had to use a thick ointment to drive the drug in and get the best efficacy,” she said. “But we’ve changed our thought process.”
For example, betamethasone dipropionate 0.05%, now comes in multiple types of ointments and creams, with different potency classes, including Diprolene ointment, 0.05%, Diprosone cream, 0.05%, Diprolene cream AF, 0.05%, and Diprolene cream, 0.05%, as well as a lotion and an emollient spray.
“It’s the same active drug, but different vehicles absolutely change the potency of the drug,” Dr. Stein Gold said.
So which is the most potent? She said you can’t tell just by the vehicle. In this case, the most potent forms – in the “superpotent” class 1 – are Diprolene cream, 0.05%, and Diprolene ointment, 0.05%. (The National Psoriasis Foundation has a potency chart for topical psoriasis medications.)
She also recommended considering combination therapy with tazarotene. Tazarotene, a vitamin A derivative, is associated with a variety of side effects in 10%-30% of patients, including pruritus, erythema, irritation, skin pain, psoriasis worsening, and burning/stinging. But combination therapy with topical corticosteroids can reduce adverse effects, and it boosts efficacy as well, Dr. Stein Gold said.
She added that tazarotene can be a tool against acne. The 0.1% cream and gel formulations are approved by the Food and Drug Administration for treating acne; the 0.05% cream and gel forms are approved only for psoriasis. “Both concentrations work well and hit the different pillars of the pathogenesis of acne,” she said.
In addition, Dr. Stein Gold noted that she led two 2018 studies that found a fixed combination of halobetasol propionate 0.01% and tazarotene 0.045% lotion in moderate to severe plaque psoriasis was associated with significant reductions in the severity of the clinical signs of psoriasis, and minimal safety concerns (J Am Acad Dermatol. 2018 Aug;79[2]:287-93).
As for the future in topical treatment for psoriasis, she said researchers are exploring phosphodiesterase-4 inhibitors, Janus kinase inhibitors, and aryl hydrocarbon receptor agonists.
Dr. Stein Gold disclosed speaker bureau relationships with Galderma, Leo, Valeant, Novartis, Celgene and Allergan; consulting for Sol‐Gel, Galderma, Leo, Novan, Valeant, Dermira, Novartis, Celgene, Allergan, Foamix, Promius, Anacor and Medimetriks; receiving grant/research support from Galderma, Leo, Novan, Valeant, Dermira, Novartis, Celgene, Allergan and Foamix; and serving on scientific advisory boards for Galderma, Leo, Novan, Valeant, Dermira, Novartis, Celgene, Allergan, Foamix and Promius.
SDEF and this news organization are owned by the same parent company.
LAS VEGAS – in mild cases of psoriasis.
Topicals often are a worthwhile complement to even the most advanced systemic medications, according to Linda Stein Gold, MD, director of clinical research in the department of dermatology at the Henry Ford Health System, Detroit.
Speaking at the Skin Disease Education Foundation’s annual Las Vegas Dermatology Seminar, she pointed out that as the variety of vehicles for topical treatments has grown, so has the need to pay attention to the potency of these treatments. “Traditionally, we had thought we had to use a thick ointment to drive the drug in and get the best efficacy,” she said. “But we’ve changed our thought process.”
For example, betamethasone dipropionate 0.05%, now comes in multiple types of ointments and creams, with different potency classes, including Diprolene ointment, 0.05%, Diprosone cream, 0.05%, Diprolene cream AF, 0.05%, and Diprolene cream, 0.05%, as well as a lotion and an emollient spray.
“It’s the same active drug, but different vehicles absolutely change the potency of the drug,” Dr. Stein Gold said.
So which is the most potent? She said you can’t tell just by the vehicle. In this case, the most potent forms – in the “superpotent” class 1 – are Diprolene cream, 0.05%, and Diprolene ointment, 0.05%. (The National Psoriasis Foundation has a potency chart for topical psoriasis medications.)
She also recommended considering combination therapy with tazarotene. Tazarotene, a vitamin A derivative, is associated with a variety of side effects in 10%-30% of patients, including pruritus, erythema, irritation, skin pain, psoriasis worsening, and burning/stinging. But combination therapy with topical corticosteroids can reduce adverse effects, and it boosts efficacy as well, Dr. Stein Gold said.
She added that tazarotene can be a tool against acne. The 0.1% cream and gel formulations are approved by the Food and Drug Administration for treating acne; the 0.05% cream and gel forms are approved only for psoriasis. “Both concentrations work well and hit the different pillars of the pathogenesis of acne,” she said.
In addition, Dr. Stein Gold noted that she led two 2018 studies that found a fixed combination of halobetasol propionate 0.01% and tazarotene 0.045% lotion in moderate to severe plaque psoriasis was associated with significant reductions in the severity of the clinical signs of psoriasis, and minimal safety concerns (J Am Acad Dermatol. 2018 Aug;79[2]:287-93).
As for the future in topical treatment for psoriasis, she said researchers are exploring phosphodiesterase-4 inhibitors, Janus kinase inhibitors, and aryl hydrocarbon receptor agonists.
Dr. Stein Gold disclosed speaker bureau relationships with Galderma, Leo, Valeant, Novartis, Celgene and Allergan; consulting for Sol‐Gel, Galderma, Leo, Novan, Valeant, Dermira, Novartis, Celgene, Allergan, Foamix, Promius, Anacor and Medimetriks; receiving grant/research support from Galderma, Leo, Novan, Valeant, Dermira, Novartis, Celgene, Allergan and Foamix; and serving on scientific advisory boards for Galderma, Leo, Novan, Valeant, Dermira, Novartis, Celgene, Allergan, Foamix and Promius.
SDEF and this news organization are owned by the same parent company.
LAS VEGAS – in mild cases of psoriasis.
Topicals often are a worthwhile complement to even the most advanced systemic medications, according to Linda Stein Gold, MD, director of clinical research in the department of dermatology at the Henry Ford Health System, Detroit.
Speaking at the Skin Disease Education Foundation’s annual Las Vegas Dermatology Seminar, she pointed out that as the variety of vehicles for topical treatments has grown, so has the need to pay attention to the potency of these treatments. “Traditionally, we had thought we had to use a thick ointment to drive the drug in and get the best efficacy,” she said. “But we’ve changed our thought process.”
For example, betamethasone dipropionate 0.05%, now comes in multiple types of ointments and creams, with different potency classes, including Diprolene ointment, 0.05%, Diprosone cream, 0.05%, Diprolene cream AF, 0.05%, and Diprolene cream, 0.05%, as well as a lotion and an emollient spray.
“It’s the same active drug, but different vehicles absolutely change the potency of the drug,” Dr. Stein Gold said.
So which is the most potent? She said you can’t tell just by the vehicle. In this case, the most potent forms – in the “superpotent” class 1 – are Diprolene cream, 0.05%, and Diprolene ointment, 0.05%. (The National Psoriasis Foundation has a potency chart for topical psoriasis medications.)
She also recommended considering combination therapy with tazarotene. Tazarotene, a vitamin A derivative, is associated with a variety of side effects in 10%-30% of patients, including pruritus, erythema, irritation, skin pain, psoriasis worsening, and burning/stinging. But combination therapy with topical corticosteroids can reduce adverse effects, and it boosts efficacy as well, Dr. Stein Gold said.
She added that tazarotene can be a tool against acne. The 0.1% cream and gel formulations are approved by the Food and Drug Administration for treating acne; the 0.05% cream and gel forms are approved only for psoriasis. “Both concentrations work well and hit the different pillars of the pathogenesis of acne,” she said.
In addition, Dr. Stein Gold noted that she led two 2018 studies that found a fixed combination of halobetasol propionate 0.01% and tazarotene 0.045% lotion in moderate to severe plaque psoriasis was associated with significant reductions in the severity of the clinical signs of psoriasis, and minimal safety concerns (J Am Acad Dermatol. 2018 Aug;79[2]:287-93).
As for the future in topical treatment for psoriasis, she said researchers are exploring phosphodiesterase-4 inhibitors, Janus kinase inhibitors, and aryl hydrocarbon receptor agonists.
Dr. Stein Gold disclosed speaker bureau relationships with Galderma, Leo, Valeant, Novartis, Celgene and Allergan; consulting for Sol‐Gel, Galderma, Leo, Novan, Valeant, Dermira, Novartis, Celgene, Allergan, Foamix, Promius, Anacor and Medimetriks; receiving grant/research support from Galderma, Leo, Novan, Valeant, Dermira, Novartis, Celgene, Allergan and Foamix; and serving on scientific advisory boards for Galderma, Leo, Novan, Valeant, Dermira, Novartis, Celgene, Allergan, Foamix and Promius.
SDEF and this news organization are owned by the same parent company.
EXPERT ANALYSIS FROM SDEF LAS VEGAS DERMATOLOGY SEMINAR
Etanercept bests methotrexate for PsA; combo adds little benefit
CHICAGO – Etanercept monotherapy showed greater efficacy, compared with methotrexate monotherapy for the treatment of psoriatic arthritis, and combining the two agents provided no benefit over etanercept alone for most outcomes in the randomized, controlled, international, phase 3 SEAM-PsA study.
A 20% improvement in American College of Rheumatology criteria at week 24 – the primary endpoint of the study – was significantly greater in 284 patients treated with etanercept monotherapy and in 283 patients treated with combination etanercept and methotrexate than in 284 patients treated with methotrexate monotherapy (60.9% and 65.0% vs. 50.7%, respectively), Philip J. Mease, MD, of the Swedish Medical Center and the University of Washington, Seattle, and his colleagues reported in a late-breaking poster on the SEAM-PsA (Etanercept and Methotrexate in Subjects with Psoriatic Arthritis) study at the annual meeting of the American College of Rheumatology.
The key secondary endpoint of minimal disease activity response at week 24 also was significantly greater in the etanercept monotherapy and combination groups than in the methotrexate monotherapy group (35.9% and 35.7% vs. 22.9%, respectively), the investigators noted.
Additionally, at week 48, the etanercept monotherapy group and combination group both showed less radiographic progression than did the methotrexate monotherapy arm (mean change in modified total Sharp score from baseline, –0.04 and –0.01 vs. 0.08).
Overall, the etanercept monotherapy group and combination therapy group had similar results, with some differences in skin outcomes. Treatment was well tolerated, and except for more nausea occurring with methotrexate, adverse event rates were similar in the three study arms. No new safety signals were observed.
“The most common serious adverse events were infections and infestations, which occurred in 1.1% of patients in the methotrexate monotherapy arm, 2.8% of patients in the etanercept monotherapy arm, and 2.5% of patients in the combination therapy arm,” they wrote.
Study participants were biologic-naive adults with active PsA and no prior methotrexate treatment for their disease. They had a mean age of 48.4 years, most were white, and median disease duration was 0.6 years.
They were randomized to receive either 50 mg subcutaneous injections of etanercept plus oral placebo weekly, 50 mg subcutaneous etanercept plus 20 mg oral methotrexate weekly, or 20 mg oral methotrexate plus placebo injections weekly; the groups were well balanced with respect to baseline characteristics, the investigators said.
Rescue therapy of etanercept plus methotrexate was given after 24 weeks in patients with less than 20% improvement in tender joint counts and swollen joint counts from baseline.
“Agents used to treat PsA include disease-modifying antirheumatic drugs such as methotrexate and tumor necrosis factor inhibitors, but how to optimally use these agents to treat PsA is unknown,” they wrote, explaining that while methotrexate is widely used in this setting, little clinical evidence exists to guide its use, and that while tumor necrosis factor inhibitors have shown efficacy in PsA, the benefit of adding methotrexate remains unclear.
The current findings, however, demonstrate that adding methotrexate does not appear to increase the efficacy of etanercept monotherapy for most outcomes.
An exception was with combination therapy for some skin-related outcomes, including percent improvement in psoriasis-affected body surface area and percentage of patients with “status clear or almost clear,” they said.
Further, methotrexate monotherapy in this study appeared to have some “meaningful efficacy for both articular and nonarticular PsA symptoms,” the investigators noted.
“These results provide information of practical value for clinical practice when considering treatment option for PsA,” they concluded.
The study was supported by Amgen. Dr. Mease reported receiving research grants, speaker fees, and/or consulting fees from AbbVie, Amgen, Bristol-Myers Squibb, Celgene, Galapagos, Genentech, Janssen Pharmaceuticals, Eli Lilly, Novartis, Pfizer, Sun Pharmaceutical, and UCB.
SOURCE: Mease PJ et al. Arthritis Rheumatol. 2018;70(Suppl 10): Abstract L11.
CHICAGO – Etanercept monotherapy showed greater efficacy, compared with methotrexate monotherapy for the treatment of psoriatic arthritis, and combining the two agents provided no benefit over etanercept alone for most outcomes in the randomized, controlled, international, phase 3 SEAM-PsA study.
A 20% improvement in American College of Rheumatology criteria at week 24 – the primary endpoint of the study – was significantly greater in 284 patients treated with etanercept monotherapy and in 283 patients treated with combination etanercept and methotrexate than in 284 patients treated with methotrexate monotherapy (60.9% and 65.0% vs. 50.7%, respectively), Philip J. Mease, MD, of the Swedish Medical Center and the University of Washington, Seattle, and his colleagues reported in a late-breaking poster on the SEAM-PsA (Etanercept and Methotrexate in Subjects with Psoriatic Arthritis) study at the annual meeting of the American College of Rheumatology.
The key secondary endpoint of minimal disease activity response at week 24 also was significantly greater in the etanercept monotherapy and combination groups than in the methotrexate monotherapy group (35.9% and 35.7% vs. 22.9%, respectively), the investigators noted.
Additionally, at week 48, the etanercept monotherapy group and combination group both showed less radiographic progression than did the methotrexate monotherapy arm (mean change in modified total Sharp score from baseline, –0.04 and –0.01 vs. 0.08).
Overall, the etanercept monotherapy group and combination therapy group had similar results, with some differences in skin outcomes. Treatment was well tolerated, and except for more nausea occurring with methotrexate, adverse event rates were similar in the three study arms. No new safety signals were observed.
“The most common serious adverse events were infections and infestations, which occurred in 1.1% of patients in the methotrexate monotherapy arm, 2.8% of patients in the etanercept monotherapy arm, and 2.5% of patients in the combination therapy arm,” they wrote.
Study participants were biologic-naive adults with active PsA and no prior methotrexate treatment for their disease. They had a mean age of 48.4 years, most were white, and median disease duration was 0.6 years.
They were randomized to receive either 50 mg subcutaneous injections of etanercept plus oral placebo weekly, 50 mg subcutaneous etanercept plus 20 mg oral methotrexate weekly, or 20 mg oral methotrexate plus placebo injections weekly; the groups were well balanced with respect to baseline characteristics, the investigators said.
Rescue therapy of etanercept plus methotrexate was given after 24 weeks in patients with less than 20% improvement in tender joint counts and swollen joint counts from baseline.
“Agents used to treat PsA include disease-modifying antirheumatic drugs such as methotrexate and tumor necrosis factor inhibitors, but how to optimally use these agents to treat PsA is unknown,” they wrote, explaining that while methotrexate is widely used in this setting, little clinical evidence exists to guide its use, and that while tumor necrosis factor inhibitors have shown efficacy in PsA, the benefit of adding methotrexate remains unclear.
The current findings, however, demonstrate that adding methotrexate does not appear to increase the efficacy of etanercept monotherapy for most outcomes.
An exception was with combination therapy for some skin-related outcomes, including percent improvement in psoriasis-affected body surface area and percentage of patients with “status clear or almost clear,” they said.
Further, methotrexate monotherapy in this study appeared to have some “meaningful efficacy for both articular and nonarticular PsA symptoms,” the investigators noted.
“These results provide information of practical value for clinical practice when considering treatment option for PsA,” they concluded.
The study was supported by Amgen. Dr. Mease reported receiving research grants, speaker fees, and/or consulting fees from AbbVie, Amgen, Bristol-Myers Squibb, Celgene, Galapagos, Genentech, Janssen Pharmaceuticals, Eli Lilly, Novartis, Pfizer, Sun Pharmaceutical, and UCB.
SOURCE: Mease PJ et al. Arthritis Rheumatol. 2018;70(Suppl 10): Abstract L11.
CHICAGO – Etanercept monotherapy showed greater efficacy, compared with methotrexate monotherapy for the treatment of psoriatic arthritis, and combining the two agents provided no benefit over etanercept alone for most outcomes in the randomized, controlled, international, phase 3 SEAM-PsA study.
A 20% improvement in American College of Rheumatology criteria at week 24 – the primary endpoint of the study – was significantly greater in 284 patients treated with etanercept monotherapy and in 283 patients treated with combination etanercept and methotrexate than in 284 patients treated with methotrexate monotherapy (60.9% and 65.0% vs. 50.7%, respectively), Philip J. Mease, MD, of the Swedish Medical Center and the University of Washington, Seattle, and his colleagues reported in a late-breaking poster on the SEAM-PsA (Etanercept and Methotrexate in Subjects with Psoriatic Arthritis) study at the annual meeting of the American College of Rheumatology.
The key secondary endpoint of minimal disease activity response at week 24 also was significantly greater in the etanercept monotherapy and combination groups than in the methotrexate monotherapy group (35.9% and 35.7% vs. 22.9%, respectively), the investigators noted.
Additionally, at week 48, the etanercept monotherapy group and combination group both showed less radiographic progression than did the methotrexate monotherapy arm (mean change in modified total Sharp score from baseline, –0.04 and –0.01 vs. 0.08).
Overall, the etanercept monotherapy group and combination therapy group had similar results, with some differences in skin outcomes. Treatment was well tolerated, and except for more nausea occurring with methotrexate, adverse event rates were similar in the three study arms. No new safety signals were observed.
“The most common serious adverse events were infections and infestations, which occurred in 1.1% of patients in the methotrexate monotherapy arm, 2.8% of patients in the etanercept monotherapy arm, and 2.5% of patients in the combination therapy arm,” they wrote.
Study participants were biologic-naive adults with active PsA and no prior methotrexate treatment for their disease. They had a mean age of 48.4 years, most were white, and median disease duration was 0.6 years.
They were randomized to receive either 50 mg subcutaneous injections of etanercept plus oral placebo weekly, 50 mg subcutaneous etanercept plus 20 mg oral methotrexate weekly, or 20 mg oral methotrexate plus placebo injections weekly; the groups were well balanced with respect to baseline characteristics, the investigators said.
Rescue therapy of etanercept plus methotrexate was given after 24 weeks in patients with less than 20% improvement in tender joint counts and swollen joint counts from baseline.
“Agents used to treat PsA include disease-modifying antirheumatic drugs such as methotrexate and tumor necrosis factor inhibitors, but how to optimally use these agents to treat PsA is unknown,” they wrote, explaining that while methotrexate is widely used in this setting, little clinical evidence exists to guide its use, and that while tumor necrosis factor inhibitors have shown efficacy in PsA, the benefit of adding methotrexate remains unclear.
The current findings, however, demonstrate that adding methotrexate does not appear to increase the efficacy of etanercept monotherapy for most outcomes.
An exception was with combination therapy for some skin-related outcomes, including percent improvement in psoriasis-affected body surface area and percentage of patients with “status clear or almost clear,” they said.
Further, methotrexate monotherapy in this study appeared to have some “meaningful efficacy for both articular and nonarticular PsA symptoms,” the investigators noted.
“These results provide information of practical value for clinical practice when considering treatment option for PsA,” they concluded.
The study was supported by Amgen. Dr. Mease reported receiving research grants, speaker fees, and/or consulting fees from AbbVie, Amgen, Bristol-Myers Squibb, Celgene, Galapagos, Genentech, Janssen Pharmaceuticals, Eli Lilly, Novartis, Pfizer, Sun Pharmaceutical, and UCB.
SOURCE: Mease PJ et al. Arthritis Rheumatol. 2018;70(Suppl 10): Abstract L11.
REPORTING FROM THE ACR ANNUAL MEETING
Key clinical point: Etanercept monotherapy shows greater efficacy versus methotrexate monotherapy for psoriatic arthritis.
Major finding: A total of 60.9% achieved a 20% improvement in American College of Rheumatology criteria with etanercept monotherapy, compared with 65.0% on combination therapy and 50.7% on methotrexate monotherapy.
Study details: A randomized, controlled, phase 3 study of 851 patients.
Disclosures: The study was supported by Amgen. Dr. Mease reported receiving research grants, speaker fees, and/or consulting fees from AbbVie, Amgen, Bristol-Myers Squibb, Celgene, Galapagos, Genentech, Janssen Pharmaceuticals, Eli Lilly, Novartis, Pfizer, Sun Pharmaceutical, and UCB.
Source: Mease PJ et al. Arthritis Rheumatol. 2018;70(Suppl 10): Abstract L11.
Comorbidities are important in psoriasis care
LAS VEGAS – Pay attention to comorbidities in your psoriasis patients because there may not be anyone else doing so.
“Many of our patients don’t have primary care physicians; many are untreated for psoriasis. They come to a clinical trial to get treated – some of them may not have insurance – so it is important for us to watch for these comorbidities,” Kristina C. Duffin, MD, said at Skin Disease Education Foundation’s annual Las Vegas Dermatology Seminar.
Yet, that does not seem to be happening consistently, according to Dr. Duffin, of the department of dermatology at the University of Utah, Salt Lake City. One in five dermatologists admitted to never screening or referring their psoriasis patients for management of cardiovascular risks in a 2015 survey (J Am Acad Dermatol. 2015 doi: 10.1016/j.jaad.2015.07.029).
Often patients at the start of biologic therapy are counseled about the risk for developing tuberculosis, yet the lifetime risk for doing so in the United States is 0.3%. Similarly, patients are often counseled on the risk for developing lymphoma, even though the excess risk for developing lymphoma that can be attributed to psoriasis treatment is 7.9 per 100,000 psoriasis patients per year. That screening seems to be driven by warnings issued in direct-to-consumer advertising, Dr. Duffin suggested.
“Although psoriasis patients have an increased relative risk of lymphoma, the absolute risk attributable to psoriasis is low,” Dr. Duffin pointed out.
Some of the comorbidities she advised dermatologists to watch for are described below.
Psoriatic arthritis
Psoriatic arthritis is the most important psoriasis comorbidity, Dr. Duffin said. Between 20% and 30% of psoriasis patients will develop psoriatic arthritis.
In a study of 1,511 patients in 48 centers in Germany, 21% of psoriasis patients were diagnosed with psoriatic arthritis and of those, more than 95% had active arthritis and 53% had five or more affected joints (Br J Dermatol. 2009;160[5]:1040-7).
The GRAPPA app is an easy, free screening tool for psoriatic arthritis; patients who score 3 or more out of 5 items on the psoriasis epidemiology screening tool (PEST) are deemed positive for psoriatic arthritis, Dr. Duffin noted.
Cardiovascular disease
Psoriasis patients are at increased risk of myocardial infarction, stroke, cardiovascular death, diabetes, and chronic kidney disease, Dr. Duffin said. In fact, CV risk from severe psoriasis is similar to the risk conferred by diabetes.
She added that there is epidemiologic evidence for CV risk modification with several of the biologics approved for psoriasis.
Hypertension
Hypertension is prevalent and more severe in psoriasis patients, Dr. Duffin said, citing a 2011 case-control study of electronic medical records at the University of California, Davis. Psoriasis patients with hypertension were 5 times more likely than patients without psoriasis to be on one antihypertensive medication, 9.5 times more likely to be on two, and almost 20 times more likely to be on four antihypertensive medications (PLoS One. 2011 Mar 29;6[3]:e18227. doi: 10.1371/journal.pone.0018227).
Importantly, few primary care physicians and cardiologists are aware of the increased risk for hypertension in psoriasis patients.
Less than half (45%) of primary care physicians and 57% of cardiologists reported they were aware that psoriasis was associated with worse cardiovascular outcome, and only 43% of physicians reported screening psoriasis patients for hypertension starting at age 20 years, according to a 2012 survey of 251 physicians (J Am Acad Dermatol. 2012 Sep;67[3]:357-62).
Dr. Duffin called on dermatologists to ensure that the primary care physicians they work with understand these increased risks.
“Commit to including a comment in consultation letters or letters back to primary care physicians that talks about the cardiovascular risks of the disease,” she said.
Dr. Duffin reported that she is a consultant and has received grant or contracted research support for many companies that manufacture dermatologic therapies.
SDEF and this news organization are owned by the same parent company.
LAS VEGAS – Pay attention to comorbidities in your psoriasis patients because there may not be anyone else doing so.
“Many of our patients don’t have primary care physicians; many are untreated for psoriasis. They come to a clinical trial to get treated – some of them may not have insurance – so it is important for us to watch for these comorbidities,” Kristina C. Duffin, MD, said at Skin Disease Education Foundation’s annual Las Vegas Dermatology Seminar.
Yet, that does not seem to be happening consistently, according to Dr. Duffin, of the department of dermatology at the University of Utah, Salt Lake City. One in five dermatologists admitted to never screening or referring their psoriasis patients for management of cardiovascular risks in a 2015 survey (J Am Acad Dermatol. 2015 doi: 10.1016/j.jaad.2015.07.029).
Often patients at the start of biologic therapy are counseled about the risk for developing tuberculosis, yet the lifetime risk for doing so in the United States is 0.3%. Similarly, patients are often counseled on the risk for developing lymphoma, even though the excess risk for developing lymphoma that can be attributed to psoriasis treatment is 7.9 per 100,000 psoriasis patients per year. That screening seems to be driven by warnings issued in direct-to-consumer advertising, Dr. Duffin suggested.
“Although psoriasis patients have an increased relative risk of lymphoma, the absolute risk attributable to psoriasis is low,” Dr. Duffin pointed out.
Some of the comorbidities she advised dermatologists to watch for are described below.
Psoriatic arthritis
Psoriatic arthritis is the most important psoriasis comorbidity, Dr. Duffin said. Between 20% and 30% of psoriasis patients will develop psoriatic arthritis.
In a study of 1,511 patients in 48 centers in Germany, 21% of psoriasis patients were diagnosed with psoriatic arthritis and of those, more than 95% had active arthritis and 53% had five or more affected joints (Br J Dermatol. 2009;160[5]:1040-7).
The GRAPPA app is an easy, free screening tool for psoriatic arthritis; patients who score 3 or more out of 5 items on the psoriasis epidemiology screening tool (PEST) are deemed positive for psoriatic arthritis, Dr. Duffin noted.
Cardiovascular disease
Psoriasis patients are at increased risk of myocardial infarction, stroke, cardiovascular death, diabetes, and chronic kidney disease, Dr. Duffin said. In fact, CV risk from severe psoriasis is similar to the risk conferred by diabetes.
She added that there is epidemiologic evidence for CV risk modification with several of the biologics approved for psoriasis.
Hypertension
Hypertension is prevalent and more severe in psoriasis patients, Dr. Duffin said, citing a 2011 case-control study of electronic medical records at the University of California, Davis. Psoriasis patients with hypertension were 5 times more likely than patients without psoriasis to be on one antihypertensive medication, 9.5 times more likely to be on two, and almost 20 times more likely to be on four antihypertensive medications (PLoS One. 2011 Mar 29;6[3]:e18227. doi: 10.1371/journal.pone.0018227).
Importantly, few primary care physicians and cardiologists are aware of the increased risk for hypertension in psoriasis patients.
Less than half (45%) of primary care physicians and 57% of cardiologists reported they were aware that psoriasis was associated with worse cardiovascular outcome, and only 43% of physicians reported screening psoriasis patients for hypertension starting at age 20 years, according to a 2012 survey of 251 physicians (J Am Acad Dermatol. 2012 Sep;67[3]:357-62).
Dr. Duffin called on dermatologists to ensure that the primary care physicians they work with understand these increased risks.
“Commit to including a comment in consultation letters or letters back to primary care physicians that talks about the cardiovascular risks of the disease,” she said.
Dr. Duffin reported that she is a consultant and has received grant or contracted research support for many companies that manufacture dermatologic therapies.
SDEF and this news organization are owned by the same parent company.
LAS VEGAS – Pay attention to comorbidities in your psoriasis patients because there may not be anyone else doing so.
“Many of our patients don’t have primary care physicians; many are untreated for psoriasis. They come to a clinical trial to get treated – some of them may not have insurance – so it is important for us to watch for these comorbidities,” Kristina C. Duffin, MD, said at Skin Disease Education Foundation’s annual Las Vegas Dermatology Seminar.
Yet, that does not seem to be happening consistently, according to Dr. Duffin, of the department of dermatology at the University of Utah, Salt Lake City. One in five dermatologists admitted to never screening or referring their psoriasis patients for management of cardiovascular risks in a 2015 survey (J Am Acad Dermatol. 2015 doi: 10.1016/j.jaad.2015.07.029).
Often patients at the start of biologic therapy are counseled about the risk for developing tuberculosis, yet the lifetime risk for doing so in the United States is 0.3%. Similarly, patients are often counseled on the risk for developing lymphoma, even though the excess risk for developing lymphoma that can be attributed to psoriasis treatment is 7.9 per 100,000 psoriasis patients per year. That screening seems to be driven by warnings issued in direct-to-consumer advertising, Dr. Duffin suggested.
“Although psoriasis patients have an increased relative risk of lymphoma, the absolute risk attributable to psoriasis is low,” Dr. Duffin pointed out.
Some of the comorbidities she advised dermatologists to watch for are described below.
Psoriatic arthritis
Psoriatic arthritis is the most important psoriasis comorbidity, Dr. Duffin said. Between 20% and 30% of psoriasis patients will develop psoriatic arthritis.
In a study of 1,511 patients in 48 centers in Germany, 21% of psoriasis patients were diagnosed with psoriatic arthritis and of those, more than 95% had active arthritis and 53% had five or more affected joints (Br J Dermatol. 2009;160[5]:1040-7).
The GRAPPA app is an easy, free screening tool for psoriatic arthritis; patients who score 3 or more out of 5 items on the psoriasis epidemiology screening tool (PEST) are deemed positive for psoriatic arthritis, Dr. Duffin noted.
Cardiovascular disease
Psoriasis patients are at increased risk of myocardial infarction, stroke, cardiovascular death, diabetes, and chronic kidney disease, Dr. Duffin said. In fact, CV risk from severe psoriasis is similar to the risk conferred by diabetes.
She added that there is epidemiologic evidence for CV risk modification with several of the biologics approved for psoriasis.
Hypertension
Hypertension is prevalent and more severe in psoriasis patients, Dr. Duffin said, citing a 2011 case-control study of electronic medical records at the University of California, Davis. Psoriasis patients with hypertension were 5 times more likely than patients without psoriasis to be on one antihypertensive medication, 9.5 times more likely to be on two, and almost 20 times more likely to be on four antihypertensive medications (PLoS One. 2011 Mar 29;6[3]:e18227. doi: 10.1371/journal.pone.0018227).
Importantly, few primary care physicians and cardiologists are aware of the increased risk for hypertension in psoriasis patients.
Less than half (45%) of primary care physicians and 57% of cardiologists reported they were aware that psoriasis was associated with worse cardiovascular outcome, and only 43% of physicians reported screening psoriasis patients for hypertension starting at age 20 years, according to a 2012 survey of 251 physicians (J Am Acad Dermatol. 2012 Sep;67[3]:357-62).
Dr. Duffin called on dermatologists to ensure that the primary care physicians they work with understand these increased risks.
“Commit to including a comment in consultation letters or letters back to primary care physicians that talks about the cardiovascular risks of the disease,” she said.
Dr. Duffin reported that she is a consultant and has received grant or contracted research support for many companies that manufacture dermatologic therapies.
SDEF and this news organization are owned by the same parent company.
REPORTING FROM SDEF LAS VEGAS DERMATOLOGY SEMINAR
Over one-third of psoriasis patients have PsA
Over one-third of psoriasis patients have PsA
About two-thirds of patients with psoriasis in a national registry also had psoriatic arthritis (PsA) and/or psoriasis in at least one challenging-to-treat (CTT) area, and one-quarter had both, according to Kristina Callis Duffin, MD, of the University of Utah, Salt Lake City, and her associates.
Their analysis included 2,042 psoriasis patients who were enrolled in the Corrona Psoriasis Registry between April 2015 and May 2018 and initiated biologic treatment during that time. The mean age was 49.6 years, 80% of the patients were white, and 51% were obese. Mean disease duration was 19.9 years and 89.2% of the patients had moderate to severe disease. CTT areas include the scalp, nails, and palmoplantar areas.
A total of 784 people in the cohort (38.4%) had PsA, 778 (38.1%) had scalp psoriasis, 326 (16.0%) had nail psoriasis, 223 (10.9%) had palmoplantar psoriasis, and 535 (26.2%) had both PsA and psoriasis in at least two CTT areas. The most common combinations were PsA plus scalp psoriasis and PsA plus nail and scalp psoriasis.
“These results indicate a need to further characterize patients with psoriasis who have PsA and CTT areas and evaluate the impact of these factors to better understand their treatment needs,” the investigators noted.
The Corrona registry has been supported by numerous pharmaceutical companies, and the study authors reported numerous financial relationships with industry; two authors are Novartis employees.
Secukinumab effective for slowing radiographic progression in active PsA
Treatment with secukinumab significantly reduced radiographic progression in patients with active PsA, according to Désirée van der Heijde, MD, PhD, professor of rheumatology at Leiden University Medical Center, and her associates.
The results come from an analysis of the FUTURE 5 trial, a study of 996 patients with active PsA despite previous NSAID treatment, disease-modifying antirheumatic drug treatment, or anti–tumor necrosis factor (TNF) therapy. Patients were randomized to receive 300 mg subcutaneous secukinumab with loading dose, 150 mg secukinumab with loading dose, 150 mg secukinumab without loading dose, or placebo, at baseline; weeks 1, 2, 3, and 4; then every 4 weeks.
After 24 weeks, the mean change in van der Heijde–modified Total Sharp Score for PsA was 0.08 for the 300-mg secukinumab group (P less than .01), 0.17 for the 150-mg secukinumab with loading dose group (P less than .05), a reduction of 0.09 for the 150-mg secukinumab without loading dose group (P less than .01), and 0.50 for the placebo group. Lower radiographic progression was seen regardless of prior anti-TNF or concomitant methotrexate treatment.
The study was funded by Novartis. The study authors reported financial disclosures with numerous companies; five authors are Novartis employees.
Tildrakizumab sustains efficacy in plaque psoriasis treatment after 1 year
Nearly all patients receiving the interleukin-23 inhibitor tildrakizumab for the treatment of moderate to severe plaque psoriasis maintained or improved their Psoriasis Area and Severity Index (PASI) response rate after 52 weeks of treatment, compared with their response after 28 weeks.
The analysis, conducted by Boni E. Elewski, MD, of the University of Alabama at Birmingham, and her associates, included 352 patients who received 100 mg tildrakizumab and 313 who received 200 mg tildrakizumab. Treatment was received at baseline, at 4 weeks, and then every 12 weeks afterward.
At week 28, the proportions of patients achieving PASI 100, PASI 90-99, PASI 75-89, and PASI 50-74 at week 28 were 25.9%, 38.4%, 25.3%, and 10.5%, respectively, among those treated with the 100-mg dose. The proportions were 24.6%, 24.3%, 19.5%, and 31.6%, respectively, among those treated with the 200-mg dose.
In patients who achieved at least PASI 90 on either dose at week 28, 88.9%-89.4% maintained that response at week 52. For patients with PASI 75-89, 39.3%-40.4% maintained that response and 33.7%-41.0% achieved a PASI 90 response. At week 52, in patients with PASI 50-74, 20.2%-29.7% achieved at least a PASI 90, 52.5%-64.9% achieved PASI 75, and only 2.6% of patients on either dose had fallen below PASI 50.
Four study authors reported being clinical investigators on studies sponsored by Merck and Sun Pharmaceuticals; five authors are employees of Sun Pharmaceuticals.
Halobetasol/tazarotene combination most effective for plaque psoriasis treatment
A fixed combination of halobetasol propionate 0.01% and tazarotene 0.045% lotion provided a synergistic effect over either component on its own for the treatment of plaque psoriasis, according to Leon H. Kircik, MD, of Indiana University, Indianapolis, and his associates.
The investigators performed a post hoc analysis of 212 patients with moderate to severe plaque psoriasis randomized to receive either the halobetasol/tazarotene combination, halobetasol only, tazarotene only, or vehicle only for 8 weeks, with follow-up at 12 weeks. Treatment success was based on the proportion of patients who achieved at least a 2-grade improvement in the Investigator Global Assessment (IGA) score, IGA scores of “clear” or “almost clear,” and percent change from baseline in IGA multiplied by Body Surface Area (BSA) composite score (IGAxBSA). “Synergy was calculated by summing up the contribution of the individual active ingredients (HP and TAZ) to overall efficacy and comparing to the efficacy achieved with HP/TAZ lotion relative to vehicle,” the authors explained.
Relative to vehicle, treatment success for halobetasol/tazarotene after 8 weeks was 42.8%, 23.6% for halobetasol alone, and 9.0% for tazarotene alone. After 12 weeks, the difference was 31.3%, 14.1%, and 5.9%, respectively. The percent change in IGAxBSA scores from baseline after 8 weeks, relative to vehicle, were 51.6%, 37.3%, and 3.3%, respectively. After 12 weeks, the change was 47.3%, 25.7%, and 8.6%, respectively.
After 8 weeks, the synergy ratio for treatment success and IGAxBSA scores for the halobetasol/tazarotene combination was 1.3. After 12 weeks, the synergy ratio for treatment success was 1.6 and the ratio for IGAxBSA scores was 1.4.
“By combining two agents into one once-daily formulation, this novel formulation reduces the number of product applications and may help patient adherence,” the study authors noted.
Dr. Kircik reported serving as a consultant and investigator for Valeant Pharmaceuticals. One study author is an employee of Bausch Health and Ortho Dermatologics, and another is an employee of Dow Pharmaceutical Sciences (a division of Valeant).
Brodalumab demonstrates low immunogenicity in moderate to severe psoriasis
The immunogenicity of brodalumab in patients with moderate to severe plaque psoriasis was low and did not compromise the efficacy or safety profile of the drug, according to Kristian Reich, MD, of Dermatologikum Berlin and SCIderm Research Institute in Hamburg, Germany, and his associates.
Data from a 12-week, phase 2 trial with a 352-week, open-label extension and three 52-week phase 3 trials were included in the analysis. Antidrug antibodies (ADAs) were tested, and positive samples were further analyzed for neutralizing ADAs by a cell-based assay.
Out of the 4,461 patients who received brodalumab, 122 (2.7%) were positive for ADAs after starting brodalumab. The incidence rate ranged from 1.9% to 3.4% between all dosing groups (140 mg, 210 mg, variable dosing, and 210 mg of brodalumab after ustekinumab). In 58 (1.4%) of patients, ADAs were transient. No patients had neutralizing ADAs, and no evidence of altered pharmacokinetics, loss of efficacy, or changes in the safety profile of brodalumab in subjects positive for ADAs was seen.
No significant difference was seen in the incidence rate of hypersensitivity or injection site reactions in brodalumab, compared with placebo or ustekinumab. The most common injection site reactions were injection site pain, erythema, and bruising.
The study was supported by Amgen. The study authors reported numerous disclosures. Two authors are employees of Leo Pharma, one author is a former employee of the company.
Secukinumab improves patient-reported outcomes in CTT psoriasis
Treatment with secukinumab significantly improved patient-reported outcomes such as fatigue, itch, pain, and quality of life measures in patients with CTT psoriasis after 6 months, according to Jerry Bagel, MD, of the Psoriasis Treatment Center of Central New Jersey, East Windsor, and his associates.
A total of 68 patients with psoriasis localized to at least one CTT area who were enrolled in the Corrona Psoriasis Registry from April 15, 2015, through May 10, 2018, and were receiving secukinumab for the entirety of the 6-month study period were included in the analysis. Patient-reported outcomes included in the analysis were fatigue, itch, pain, Dermatology Quality of Life Index (DLQI) score, and Work Productivity and Activity Impairment (WPAI) scale.
The mean age at enrollment was 51.2 years and almost 80% of patients were white. Mean psoriasis duration was 21.8 years and nearly half had PsA.
Visual analog scale scores improved over baseline for fatigue (mean, 23.2 vs. 33.2; P = .01), itch (20.9 vs. 49.6; P less than .0001), and pain (12.1 vs. 33.8; P less than .0001). DLQI scores also improved (2.9 vs. 8.1; P less than .0001), and the proportion of patients who reported that psoriasis had at least a moderate effect on their life was reduced after 6 months (22.1% vs. 59.7%; P less than .0001).
Based on WPAI results, patients experienced significant improvements in the percentage of daily activities impaired (mean, 9.5% vs 17.5%; P = .0075); of the 42 patients who were employed, both impairment percentage (3.7% vs. 11.2%; P = .0148) and percentage of work hours affected (4.9% vs. 11.9%; P = .0486) were reduced from baseline.
“These results are consistent with previous reports from secukinumab clinical trials; however, additional real-world studies are needed to evaluate the long-term effectiveness of secukinumab for improving [patient-reported outcomes] in patients with psoriasis in CTT areas,” the authors noted.
The Corrona registry has been supported by numerous pharmaceutical companies, and several study authors reported various disclosures with industry. Two authors are Novartis employees. The study was supported by Novartis; the company participated in the interpretation of data and review and approval of the abstract.
These posters were presented at Skin Disease Education Foundation’s annual Las Vegas Dermatology Seminar. SDEF and this news organization are owned by the same parent company.
Over one-third of psoriasis patients have PsA
About two-thirds of patients with psoriasis in a national registry also had psoriatic arthritis (PsA) and/or psoriasis in at least one challenging-to-treat (CTT) area, and one-quarter had both, according to Kristina Callis Duffin, MD, of the University of Utah, Salt Lake City, and her associates.
Their analysis included 2,042 psoriasis patients who were enrolled in the Corrona Psoriasis Registry between April 2015 and May 2018 and initiated biologic treatment during that time. The mean age was 49.6 years, 80% of the patients were white, and 51% were obese. Mean disease duration was 19.9 years and 89.2% of the patients had moderate to severe disease. CTT areas include the scalp, nails, and palmoplantar areas.
A total of 784 people in the cohort (38.4%) had PsA, 778 (38.1%) had scalp psoriasis, 326 (16.0%) had nail psoriasis, 223 (10.9%) had palmoplantar psoriasis, and 535 (26.2%) had both PsA and psoriasis in at least two CTT areas. The most common combinations were PsA plus scalp psoriasis and PsA plus nail and scalp psoriasis.
“These results indicate a need to further characterize patients with psoriasis who have PsA and CTT areas and evaluate the impact of these factors to better understand their treatment needs,” the investigators noted.
The Corrona registry has been supported by numerous pharmaceutical companies, and the study authors reported numerous financial relationships with industry; two authors are Novartis employees.
Secukinumab effective for slowing radiographic progression in active PsA
Treatment with secukinumab significantly reduced radiographic progression in patients with active PsA, according to Désirée van der Heijde, MD, PhD, professor of rheumatology at Leiden University Medical Center, and her associates.
The results come from an analysis of the FUTURE 5 trial, a study of 996 patients with active PsA despite previous NSAID treatment, disease-modifying antirheumatic drug treatment, or anti–tumor necrosis factor (TNF) therapy. Patients were randomized to receive 300 mg subcutaneous secukinumab with loading dose, 150 mg secukinumab with loading dose, 150 mg secukinumab without loading dose, or placebo, at baseline; weeks 1, 2, 3, and 4; then every 4 weeks.
After 24 weeks, the mean change in van der Heijde–modified Total Sharp Score for PsA was 0.08 for the 300-mg secukinumab group (P less than .01), 0.17 for the 150-mg secukinumab with loading dose group (P less than .05), a reduction of 0.09 for the 150-mg secukinumab without loading dose group (P less than .01), and 0.50 for the placebo group. Lower radiographic progression was seen regardless of prior anti-TNF or concomitant methotrexate treatment.
The study was funded by Novartis. The study authors reported financial disclosures with numerous companies; five authors are Novartis employees.
Tildrakizumab sustains efficacy in plaque psoriasis treatment after 1 year
Nearly all patients receiving the interleukin-23 inhibitor tildrakizumab for the treatment of moderate to severe plaque psoriasis maintained or improved their Psoriasis Area and Severity Index (PASI) response rate after 52 weeks of treatment, compared with their response after 28 weeks.
The analysis, conducted by Boni E. Elewski, MD, of the University of Alabama at Birmingham, and her associates, included 352 patients who received 100 mg tildrakizumab and 313 who received 200 mg tildrakizumab. Treatment was received at baseline, at 4 weeks, and then every 12 weeks afterward.
At week 28, the proportions of patients achieving PASI 100, PASI 90-99, PASI 75-89, and PASI 50-74 at week 28 were 25.9%, 38.4%, 25.3%, and 10.5%, respectively, among those treated with the 100-mg dose. The proportions were 24.6%, 24.3%, 19.5%, and 31.6%, respectively, among those treated with the 200-mg dose.
In patients who achieved at least PASI 90 on either dose at week 28, 88.9%-89.4% maintained that response at week 52. For patients with PASI 75-89, 39.3%-40.4% maintained that response and 33.7%-41.0% achieved a PASI 90 response. At week 52, in patients with PASI 50-74, 20.2%-29.7% achieved at least a PASI 90, 52.5%-64.9% achieved PASI 75, and only 2.6% of patients on either dose had fallen below PASI 50.
Four study authors reported being clinical investigators on studies sponsored by Merck and Sun Pharmaceuticals; five authors are employees of Sun Pharmaceuticals.
Halobetasol/tazarotene combination most effective for plaque psoriasis treatment
A fixed combination of halobetasol propionate 0.01% and tazarotene 0.045% lotion provided a synergistic effect over either component on its own for the treatment of plaque psoriasis, according to Leon H. Kircik, MD, of Indiana University, Indianapolis, and his associates.
The investigators performed a post hoc analysis of 212 patients with moderate to severe plaque psoriasis randomized to receive either the halobetasol/tazarotene combination, halobetasol only, tazarotene only, or vehicle only for 8 weeks, with follow-up at 12 weeks. Treatment success was based on the proportion of patients who achieved at least a 2-grade improvement in the Investigator Global Assessment (IGA) score, IGA scores of “clear” or “almost clear,” and percent change from baseline in IGA multiplied by Body Surface Area (BSA) composite score (IGAxBSA). “Synergy was calculated by summing up the contribution of the individual active ingredients (HP and TAZ) to overall efficacy and comparing to the efficacy achieved with HP/TAZ lotion relative to vehicle,” the authors explained.
Relative to vehicle, treatment success for halobetasol/tazarotene after 8 weeks was 42.8%, 23.6% for halobetasol alone, and 9.0% for tazarotene alone. After 12 weeks, the difference was 31.3%, 14.1%, and 5.9%, respectively. The percent change in IGAxBSA scores from baseline after 8 weeks, relative to vehicle, were 51.6%, 37.3%, and 3.3%, respectively. After 12 weeks, the change was 47.3%, 25.7%, and 8.6%, respectively.
After 8 weeks, the synergy ratio for treatment success and IGAxBSA scores for the halobetasol/tazarotene combination was 1.3. After 12 weeks, the synergy ratio for treatment success was 1.6 and the ratio for IGAxBSA scores was 1.4.
“By combining two agents into one once-daily formulation, this novel formulation reduces the number of product applications and may help patient adherence,” the study authors noted.
Dr. Kircik reported serving as a consultant and investigator for Valeant Pharmaceuticals. One study author is an employee of Bausch Health and Ortho Dermatologics, and another is an employee of Dow Pharmaceutical Sciences (a division of Valeant).
Brodalumab demonstrates low immunogenicity in moderate to severe psoriasis
The immunogenicity of brodalumab in patients with moderate to severe plaque psoriasis was low and did not compromise the efficacy or safety profile of the drug, according to Kristian Reich, MD, of Dermatologikum Berlin and SCIderm Research Institute in Hamburg, Germany, and his associates.
Data from a 12-week, phase 2 trial with a 352-week, open-label extension and three 52-week phase 3 trials were included in the analysis. Antidrug antibodies (ADAs) were tested, and positive samples were further analyzed for neutralizing ADAs by a cell-based assay.
Out of the 4,461 patients who received brodalumab, 122 (2.7%) were positive for ADAs after starting brodalumab. The incidence rate ranged from 1.9% to 3.4% between all dosing groups (140 mg, 210 mg, variable dosing, and 210 mg of brodalumab after ustekinumab). In 58 (1.4%) of patients, ADAs were transient. No patients had neutralizing ADAs, and no evidence of altered pharmacokinetics, loss of efficacy, or changes in the safety profile of brodalumab in subjects positive for ADAs was seen.
No significant difference was seen in the incidence rate of hypersensitivity or injection site reactions in brodalumab, compared with placebo or ustekinumab. The most common injection site reactions were injection site pain, erythema, and bruising.
The study was supported by Amgen. The study authors reported numerous disclosures. Two authors are employees of Leo Pharma, one author is a former employee of the company.
Secukinumab improves patient-reported outcomes in CTT psoriasis
Treatment with secukinumab significantly improved patient-reported outcomes such as fatigue, itch, pain, and quality of life measures in patients with CTT psoriasis after 6 months, according to Jerry Bagel, MD, of the Psoriasis Treatment Center of Central New Jersey, East Windsor, and his associates.
A total of 68 patients with psoriasis localized to at least one CTT area who were enrolled in the Corrona Psoriasis Registry from April 15, 2015, through May 10, 2018, and were receiving secukinumab for the entirety of the 6-month study period were included in the analysis. Patient-reported outcomes included in the analysis were fatigue, itch, pain, Dermatology Quality of Life Index (DLQI) score, and Work Productivity and Activity Impairment (WPAI) scale.
The mean age at enrollment was 51.2 years and almost 80% of patients were white. Mean psoriasis duration was 21.8 years and nearly half had PsA.
Visual analog scale scores improved over baseline for fatigue (mean, 23.2 vs. 33.2; P = .01), itch (20.9 vs. 49.6; P less than .0001), and pain (12.1 vs. 33.8; P less than .0001). DLQI scores also improved (2.9 vs. 8.1; P less than .0001), and the proportion of patients who reported that psoriasis had at least a moderate effect on their life was reduced after 6 months (22.1% vs. 59.7%; P less than .0001).
Based on WPAI results, patients experienced significant improvements in the percentage of daily activities impaired (mean, 9.5% vs 17.5%; P = .0075); of the 42 patients who were employed, both impairment percentage (3.7% vs. 11.2%; P = .0148) and percentage of work hours affected (4.9% vs. 11.9%; P = .0486) were reduced from baseline.
“These results are consistent with previous reports from secukinumab clinical trials; however, additional real-world studies are needed to evaluate the long-term effectiveness of secukinumab for improving [patient-reported outcomes] in patients with psoriasis in CTT areas,” the authors noted.
The Corrona registry has been supported by numerous pharmaceutical companies, and several study authors reported various disclosures with industry. Two authors are Novartis employees. The study was supported by Novartis; the company participated in the interpretation of data and review and approval of the abstract.
These posters were presented at Skin Disease Education Foundation’s annual Las Vegas Dermatology Seminar. SDEF and this news organization are owned by the same parent company.
Over one-third of psoriasis patients have PsA
About two-thirds of patients with psoriasis in a national registry also had psoriatic arthritis (PsA) and/or psoriasis in at least one challenging-to-treat (CTT) area, and one-quarter had both, according to Kristina Callis Duffin, MD, of the University of Utah, Salt Lake City, and her associates.
Their analysis included 2,042 psoriasis patients who were enrolled in the Corrona Psoriasis Registry between April 2015 and May 2018 and initiated biologic treatment during that time. The mean age was 49.6 years, 80% of the patients were white, and 51% were obese. Mean disease duration was 19.9 years and 89.2% of the patients had moderate to severe disease. CTT areas include the scalp, nails, and palmoplantar areas.
A total of 784 people in the cohort (38.4%) had PsA, 778 (38.1%) had scalp psoriasis, 326 (16.0%) had nail psoriasis, 223 (10.9%) had palmoplantar psoriasis, and 535 (26.2%) had both PsA and psoriasis in at least two CTT areas. The most common combinations were PsA plus scalp psoriasis and PsA plus nail and scalp psoriasis.
“These results indicate a need to further characterize patients with psoriasis who have PsA and CTT areas and evaluate the impact of these factors to better understand their treatment needs,” the investigators noted.
The Corrona registry has been supported by numerous pharmaceutical companies, and the study authors reported numerous financial relationships with industry; two authors are Novartis employees.
Secukinumab effective for slowing radiographic progression in active PsA
Treatment with secukinumab significantly reduced radiographic progression in patients with active PsA, according to Désirée van der Heijde, MD, PhD, professor of rheumatology at Leiden University Medical Center, and her associates.
The results come from an analysis of the FUTURE 5 trial, a study of 996 patients with active PsA despite previous NSAID treatment, disease-modifying antirheumatic drug treatment, or anti–tumor necrosis factor (TNF) therapy. Patients were randomized to receive 300 mg subcutaneous secukinumab with loading dose, 150 mg secukinumab with loading dose, 150 mg secukinumab without loading dose, or placebo, at baseline; weeks 1, 2, 3, and 4; then every 4 weeks.
After 24 weeks, the mean change in van der Heijde–modified Total Sharp Score for PsA was 0.08 for the 300-mg secukinumab group (P less than .01), 0.17 for the 150-mg secukinumab with loading dose group (P less than .05), a reduction of 0.09 for the 150-mg secukinumab without loading dose group (P less than .01), and 0.50 for the placebo group. Lower radiographic progression was seen regardless of prior anti-TNF or concomitant methotrexate treatment.
The study was funded by Novartis. The study authors reported financial disclosures with numerous companies; five authors are Novartis employees.
Tildrakizumab sustains efficacy in plaque psoriasis treatment after 1 year
Nearly all patients receiving the interleukin-23 inhibitor tildrakizumab for the treatment of moderate to severe plaque psoriasis maintained or improved their Psoriasis Area and Severity Index (PASI) response rate after 52 weeks of treatment, compared with their response after 28 weeks.
The analysis, conducted by Boni E. Elewski, MD, of the University of Alabama at Birmingham, and her associates, included 352 patients who received 100 mg tildrakizumab and 313 who received 200 mg tildrakizumab. Treatment was received at baseline, at 4 weeks, and then every 12 weeks afterward.
At week 28, the proportions of patients achieving PASI 100, PASI 90-99, PASI 75-89, and PASI 50-74 at week 28 were 25.9%, 38.4%, 25.3%, and 10.5%, respectively, among those treated with the 100-mg dose. The proportions were 24.6%, 24.3%, 19.5%, and 31.6%, respectively, among those treated with the 200-mg dose.
In patients who achieved at least PASI 90 on either dose at week 28, 88.9%-89.4% maintained that response at week 52. For patients with PASI 75-89, 39.3%-40.4% maintained that response and 33.7%-41.0% achieved a PASI 90 response. At week 52, in patients with PASI 50-74, 20.2%-29.7% achieved at least a PASI 90, 52.5%-64.9% achieved PASI 75, and only 2.6% of patients on either dose had fallen below PASI 50.
Four study authors reported being clinical investigators on studies sponsored by Merck and Sun Pharmaceuticals; five authors are employees of Sun Pharmaceuticals.
Halobetasol/tazarotene combination most effective for plaque psoriasis treatment
A fixed combination of halobetasol propionate 0.01% and tazarotene 0.045% lotion provided a synergistic effect over either component on its own for the treatment of plaque psoriasis, according to Leon H. Kircik, MD, of Indiana University, Indianapolis, and his associates.
The investigators performed a post hoc analysis of 212 patients with moderate to severe plaque psoriasis randomized to receive either the halobetasol/tazarotene combination, halobetasol only, tazarotene only, or vehicle only for 8 weeks, with follow-up at 12 weeks. Treatment success was based on the proportion of patients who achieved at least a 2-grade improvement in the Investigator Global Assessment (IGA) score, IGA scores of “clear” or “almost clear,” and percent change from baseline in IGA multiplied by Body Surface Area (BSA) composite score (IGAxBSA). “Synergy was calculated by summing up the contribution of the individual active ingredients (HP and TAZ) to overall efficacy and comparing to the efficacy achieved with HP/TAZ lotion relative to vehicle,” the authors explained.
Relative to vehicle, treatment success for halobetasol/tazarotene after 8 weeks was 42.8%, 23.6% for halobetasol alone, and 9.0% for tazarotene alone. After 12 weeks, the difference was 31.3%, 14.1%, and 5.9%, respectively. The percent change in IGAxBSA scores from baseline after 8 weeks, relative to vehicle, were 51.6%, 37.3%, and 3.3%, respectively. After 12 weeks, the change was 47.3%, 25.7%, and 8.6%, respectively.
After 8 weeks, the synergy ratio for treatment success and IGAxBSA scores for the halobetasol/tazarotene combination was 1.3. After 12 weeks, the synergy ratio for treatment success was 1.6 and the ratio for IGAxBSA scores was 1.4.
“By combining two agents into one once-daily formulation, this novel formulation reduces the number of product applications and may help patient adherence,” the study authors noted.
Dr. Kircik reported serving as a consultant and investigator for Valeant Pharmaceuticals. One study author is an employee of Bausch Health and Ortho Dermatologics, and another is an employee of Dow Pharmaceutical Sciences (a division of Valeant).
Brodalumab demonstrates low immunogenicity in moderate to severe psoriasis
The immunogenicity of brodalumab in patients with moderate to severe plaque psoriasis was low and did not compromise the efficacy or safety profile of the drug, according to Kristian Reich, MD, of Dermatologikum Berlin and SCIderm Research Institute in Hamburg, Germany, and his associates.
Data from a 12-week, phase 2 trial with a 352-week, open-label extension and three 52-week phase 3 trials were included in the analysis. Antidrug antibodies (ADAs) were tested, and positive samples were further analyzed for neutralizing ADAs by a cell-based assay.
Out of the 4,461 patients who received brodalumab, 122 (2.7%) were positive for ADAs after starting brodalumab. The incidence rate ranged from 1.9% to 3.4% between all dosing groups (140 mg, 210 mg, variable dosing, and 210 mg of brodalumab after ustekinumab). In 58 (1.4%) of patients, ADAs were transient. No patients had neutralizing ADAs, and no evidence of altered pharmacokinetics, loss of efficacy, or changes in the safety profile of brodalumab in subjects positive for ADAs was seen.
No significant difference was seen in the incidence rate of hypersensitivity or injection site reactions in brodalumab, compared with placebo or ustekinumab. The most common injection site reactions were injection site pain, erythema, and bruising.
The study was supported by Amgen. The study authors reported numerous disclosures. Two authors are employees of Leo Pharma, one author is a former employee of the company.
Secukinumab improves patient-reported outcomes in CTT psoriasis
Treatment with secukinumab significantly improved patient-reported outcomes such as fatigue, itch, pain, and quality of life measures in patients with CTT psoriasis after 6 months, according to Jerry Bagel, MD, of the Psoriasis Treatment Center of Central New Jersey, East Windsor, and his associates.
A total of 68 patients with psoriasis localized to at least one CTT area who were enrolled in the Corrona Psoriasis Registry from April 15, 2015, through May 10, 2018, and were receiving secukinumab for the entirety of the 6-month study period were included in the analysis. Patient-reported outcomes included in the analysis were fatigue, itch, pain, Dermatology Quality of Life Index (DLQI) score, and Work Productivity and Activity Impairment (WPAI) scale.
The mean age at enrollment was 51.2 years and almost 80% of patients were white. Mean psoriasis duration was 21.8 years and nearly half had PsA.
Visual analog scale scores improved over baseline for fatigue (mean, 23.2 vs. 33.2; P = .01), itch (20.9 vs. 49.6; P less than .0001), and pain (12.1 vs. 33.8; P less than .0001). DLQI scores also improved (2.9 vs. 8.1; P less than .0001), and the proportion of patients who reported that psoriasis had at least a moderate effect on their life was reduced after 6 months (22.1% vs. 59.7%; P less than .0001).
Based on WPAI results, patients experienced significant improvements in the percentage of daily activities impaired (mean, 9.5% vs 17.5%; P = .0075); of the 42 patients who were employed, both impairment percentage (3.7% vs. 11.2%; P = .0148) and percentage of work hours affected (4.9% vs. 11.9%; P = .0486) were reduced from baseline.
“These results are consistent with previous reports from secukinumab clinical trials; however, additional real-world studies are needed to evaluate the long-term effectiveness of secukinumab for improving [patient-reported outcomes] in patients with psoriasis in CTT areas,” the authors noted.
The Corrona registry has been supported by numerous pharmaceutical companies, and several study authors reported various disclosures with industry. Two authors are Novartis employees. The study was supported by Novartis; the company participated in the interpretation of data and review and approval of the abstract.
These posters were presented at Skin Disease Education Foundation’s annual Las Vegas Dermatology Seminar. SDEF and this news organization are owned by the same parent company.
FROM SDEF LAS VEGAS DERMATOLOGY SEMINAR