User login
New Model May Predict Risk of Acute Kidney Injury in Orthopedic Patients
Clinical question: What is the risk of acute kidney injury after orthopedic surgery, and does it impact mortality?
Background: Current studies show that acute kidney injury is associated with increased long-term mortality, future development of chronic kidney disease, and increased healthcare costs. However, no externally validated models are available to predict patients undergoing non-cardiac surgery at risk of postoperative acute kidney injury.
Study design: Observational, cohort study.
Setting: Teaching and private hospitals in the National Health Service (NHS) in the Tayside region of Scotland.
Synopsis: Investigators enrolled 10,615 adults >18 years of age undergoing orthopedic surgery into two groups: development cohort (6,220 patients) and validation cohort (4,395 patients). Using the development cohort, seven predictors were identified in the risk model: age at operation, male sex, diabetes, lower estimated glomerular filtration rate (GFR), use of ACE inhibitor/ARB, number of prescribing drugs, and American Society of Anesthesiologists (ASA) grade.
The model’s predictive performance for discrimination was good in the development cohort (C statistic 0.74; 95% CI, 0.72–0.76) and validation cohort (C statistic 0.7). Calibration was good in the development cohort but overestimated the risk in the validation cohort. Postoperative acute kidney injury developed in 672 (10.8%) patients in the development cohort and 295 (6.7%) in the validation cohort. Thirty percent (3,166) of the 10,615 patients enrolled in this study died over the median follow-up of 4.58 years. Survival was worse in the patients with acute kidney injury (adjusted hazard ratio 1.53; 95% CI, 1.38–1.70), worse in the short term (90-day adjusted hazard ratio 2.36; 95% CI, 1.94–2.87), and diminished over time.
Bottom line: A predictive model using age, male sex, diabetes, lower GFR, use of ACE inhibitor/ARB, multiple medications, and ASA grades might predict risk of postoperative acute kidney injury in orthopedic patients.
Citation: Bell S, Dekker FW, Vadiveloo T, et al. Risk of postoperative acute kidney injury in patients undergoing orthopaedic surgery—development and validation of a risk score and effect of acute kidney injury on survival: observational cohort study. BMJ 2015; 351:h5639.
Clinical question: What is the risk of acute kidney injury after orthopedic surgery, and does it impact mortality?
Background: Current studies show that acute kidney injury is associated with increased long-term mortality, future development of chronic kidney disease, and increased healthcare costs. However, no externally validated models are available to predict patients undergoing non-cardiac surgery at risk of postoperative acute kidney injury.
Study design: Observational, cohort study.
Setting: Teaching and private hospitals in the National Health Service (NHS) in the Tayside region of Scotland.
Synopsis: Investigators enrolled 10,615 adults >18 years of age undergoing orthopedic surgery into two groups: development cohort (6,220 patients) and validation cohort (4,395 patients). Using the development cohort, seven predictors were identified in the risk model: age at operation, male sex, diabetes, lower estimated glomerular filtration rate (GFR), use of ACE inhibitor/ARB, number of prescribing drugs, and American Society of Anesthesiologists (ASA) grade.
The model’s predictive performance for discrimination was good in the development cohort (C statistic 0.74; 95% CI, 0.72–0.76) and validation cohort (C statistic 0.7). Calibration was good in the development cohort but overestimated the risk in the validation cohort. Postoperative acute kidney injury developed in 672 (10.8%) patients in the development cohort and 295 (6.7%) in the validation cohort. Thirty percent (3,166) of the 10,615 patients enrolled in this study died over the median follow-up of 4.58 years. Survival was worse in the patients with acute kidney injury (adjusted hazard ratio 1.53; 95% CI, 1.38–1.70), worse in the short term (90-day adjusted hazard ratio 2.36; 95% CI, 1.94–2.87), and diminished over time.
Bottom line: A predictive model using age, male sex, diabetes, lower GFR, use of ACE inhibitor/ARB, multiple medications, and ASA grades might predict risk of postoperative acute kidney injury in orthopedic patients.
Citation: Bell S, Dekker FW, Vadiveloo T, et al. Risk of postoperative acute kidney injury in patients undergoing orthopaedic surgery—development and validation of a risk score and effect of acute kidney injury on survival: observational cohort study. BMJ 2015; 351:h5639.
Clinical question: What is the risk of acute kidney injury after orthopedic surgery, and does it impact mortality?
Background: Current studies show that acute kidney injury is associated with increased long-term mortality, future development of chronic kidney disease, and increased healthcare costs. However, no externally validated models are available to predict patients undergoing non-cardiac surgery at risk of postoperative acute kidney injury.
Study design: Observational, cohort study.
Setting: Teaching and private hospitals in the National Health Service (NHS) in the Tayside region of Scotland.
Synopsis: Investigators enrolled 10,615 adults >18 years of age undergoing orthopedic surgery into two groups: development cohort (6,220 patients) and validation cohort (4,395 patients). Using the development cohort, seven predictors were identified in the risk model: age at operation, male sex, diabetes, lower estimated glomerular filtration rate (GFR), use of ACE inhibitor/ARB, number of prescribing drugs, and American Society of Anesthesiologists (ASA) grade.
The model’s predictive performance for discrimination was good in the development cohort (C statistic 0.74; 95% CI, 0.72–0.76) and validation cohort (C statistic 0.7). Calibration was good in the development cohort but overestimated the risk in the validation cohort. Postoperative acute kidney injury developed in 672 (10.8%) patients in the development cohort and 295 (6.7%) in the validation cohort. Thirty percent (3,166) of the 10,615 patients enrolled in this study died over the median follow-up of 4.58 years. Survival was worse in the patients with acute kidney injury (adjusted hazard ratio 1.53; 95% CI, 1.38–1.70), worse in the short term (90-day adjusted hazard ratio 2.36; 95% CI, 1.94–2.87), and diminished over time.
Bottom line: A predictive model using age, male sex, diabetes, lower GFR, use of ACE inhibitor/ARB, multiple medications, and ASA grades might predict risk of postoperative acute kidney injury in orthopedic patients.
Citation: Bell S, Dekker FW, Vadiveloo T, et al. Risk of postoperative acute kidney injury in patients undergoing orthopaedic surgery—development and validation of a risk score and effect of acute kidney injury on survival: observational cohort study. BMJ 2015; 351:h5639.
Treating Asymptomatic Bacteriuria Can Be Dangerous
Clinical question: Does treating asymptomatic bacteriuria (AB) cause harm in women?
Background: In women with recurrent UTIs, AB is often treated, increasing the risk of multi-drug-resistant bacteria. At the same time, little data exist on the relationship between AB treatment and risk of higher antibiotic resistance in women with recurrent UTIs.
Study design: Follow-up observational, analytical, longitudinal study on a previously randomized clinical trial (RCT).
Setting: Sexually transmitted disease (STD) center in Florence, Italy.
Synopsis: Using the patients from the authors’ previous RCT, the study followed 550 women with recurrent UTIs and AB for a mean of 38.8 months in parallel groups: One group had AB treated, and the other group did not. In the group of women treated with antibiotics, the recurrence rate was 69.6% versus 37.7% in the group not treated (P<0.001). In addition, E. coli isolates showed more resistance to amoxicillin/clavulanic acid (P=0.03), trimethoprim/sulfamethazole (P=0.01), and ciprofloxacin (P=0.03) in the group previously treated with antibiotics.
Given the observational design of the study, data must be interpreted with caution in determining a causal relationship. However, prior studies have shown this relationship, and current Infectious Diseases Society of America guidelines support neither screening nor treating AB.
Bottom line: In women with recurrent UTIs, previous treatment of AB is associated with higher rates of antibiotic-resistant bacteria, causing symptomatic UTIs.
Citation: Cai T, Nsei G, Mazzoli S, et al. Asymptomatic bacteriuria treatment is associated with a higher prevalence of antibiotic resistant strains in women with urinary tract infection. Clin Infect Dis. 2015;61(11):1655-1661.
Short Take
National Healthcare Spending Increased in 2014
Led by expansions under the Affordable Care Act, healthcare spending increased 5.3% from the previous year and now totals $3 trillion, which represents 17.5% of the gross domestic product.
Citation: Martin AB, Hartman M, Benson J, Catlin A. National health spending in 2014: faster growth drive by coverage expansion and prescription drug spending. Health Aff. 2016;35(1):150-160.
Clinical question: Does treating asymptomatic bacteriuria (AB) cause harm in women?
Background: In women with recurrent UTIs, AB is often treated, increasing the risk of multi-drug-resistant bacteria. At the same time, little data exist on the relationship between AB treatment and risk of higher antibiotic resistance in women with recurrent UTIs.
Study design: Follow-up observational, analytical, longitudinal study on a previously randomized clinical trial (RCT).
Setting: Sexually transmitted disease (STD) center in Florence, Italy.
Synopsis: Using the patients from the authors’ previous RCT, the study followed 550 women with recurrent UTIs and AB for a mean of 38.8 months in parallel groups: One group had AB treated, and the other group did not. In the group of women treated with antibiotics, the recurrence rate was 69.6% versus 37.7% in the group not treated (P<0.001). In addition, E. coli isolates showed more resistance to amoxicillin/clavulanic acid (P=0.03), trimethoprim/sulfamethazole (P=0.01), and ciprofloxacin (P=0.03) in the group previously treated with antibiotics.
Given the observational design of the study, data must be interpreted with caution in determining a causal relationship. However, prior studies have shown this relationship, and current Infectious Diseases Society of America guidelines support neither screening nor treating AB.
Bottom line: In women with recurrent UTIs, previous treatment of AB is associated with higher rates of antibiotic-resistant bacteria, causing symptomatic UTIs.
Citation: Cai T, Nsei G, Mazzoli S, et al. Asymptomatic bacteriuria treatment is associated with a higher prevalence of antibiotic resistant strains in women with urinary tract infection. Clin Infect Dis. 2015;61(11):1655-1661.
Short Take
National Healthcare Spending Increased in 2014
Led by expansions under the Affordable Care Act, healthcare spending increased 5.3% from the previous year and now totals $3 trillion, which represents 17.5% of the gross domestic product.
Citation: Martin AB, Hartman M, Benson J, Catlin A. National health spending in 2014: faster growth drive by coverage expansion and prescription drug spending. Health Aff. 2016;35(1):150-160.
Clinical question: Does treating asymptomatic bacteriuria (AB) cause harm in women?
Background: In women with recurrent UTIs, AB is often treated, increasing the risk of multi-drug-resistant bacteria. At the same time, little data exist on the relationship between AB treatment and risk of higher antibiotic resistance in women with recurrent UTIs.
Study design: Follow-up observational, analytical, longitudinal study on a previously randomized clinical trial (RCT).
Setting: Sexually transmitted disease (STD) center in Florence, Italy.
Synopsis: Using the patients from the authors’ previous RCT, the study followed 550 women with recurrent UTIs and AB for a mean of 38.8 months in parallel groups: One group had AB treated, and the other group did not. In the group of women treated with antibiotics, the recurrence rate was 69.6% versus 37.7% in the group not treated (P<0.001). In addition, E. coli isolates showed more resistance to amoxicillin/clavulanic acid (P=0.03), trimethoprim/sulfamethazole (P=0.01), and ciprofloxacin (P=0.03) in the group previously treated with antibiotics.
Given the observational design of the study, data must be interpreted with caution in determining a causal relationship. However, prior studies have shown this relationship, and current Infectious Diseases Society of America guidelines support neither screening nor treating AB.
Bottom line: In women with recurrent UTIs, previous treatment of AB is associated with higher rates of antibiotic-resistant bacteria, causing symptomatic UTIs.
Citation: Cai T, Nsei G, Mazzoli S, et al. Asymptomatic bacteriuria treatment is associated with a higher prevalence of antibiotic resistant strains in women with urinary tract infection. Clin Infect Dis. 2015;61(11):1655-1661.
Short Take
National Healthcare Spending Increased in 2014
Led by expansions under the Affordable Care Act, healthcare spending increased 5.3% from the previous year and now totals $3 trillion, which represents 17.5% of the gross domestic product.
Citation: Martin AB, Hartman M, Benson J, Catlin A. National health spending in 2014: faster growth drive by coverage expansion and prescription drug spending. Health Aff. 2016;35(1):150-160.
Edoxban has Advantages over Warfarin for Patients with Venous Thromboembolism
NEW YORK (Reuters Health) - Edoxaban (Savaysa, Daiichi-Sankyo) shows advantages over warfarin in long-term treatment of patients with venous thromboembolism (VTE), according to a post-hoc analysis of multinational trial data.
As Dr. Gary Raskob told Reuters Health by email, "Our results indicate that once-daily edoxaban provides an effective
and more convenient alternative to warfarin, with lower major bleeding risk, for patients who require extended treatment
beyond three months to prevent recurrent venous thromboembolism."
In a March 22 online paper in the Lancet Haematology, Dr.Raskob, of the University of Oklahoma, Oklahoma City, and colleagues note that guidelines recommend anticoagulant treatment for at least three months. However, "The risk of recurrence is substantial for patients with unprovoked venous thromboembolism or continuing risk factors and many of these
patients need extended anticoagulation therapy beyond three months."
To shed more light on longer term effects, the team examined outcome after three to 12 months in 3,633 patients treated with heparin and edoxaban and 3,594 treated with heparin and warfarin who took part in a randomized, double-blind trial. Median treatment duration was close to 9 months.
At three months, recurrent VTE was seen in 1.1% of the edoxaban group and 1.2% of the warfarin patients. At three to six months, the corresponding proportions were 0.7% and 0.5%. At more than six but less than 12 months, they were 0.2% and 0.8%.
Among other findings was that the cumulative incidence of major bleeding was 0.3% in the edoxaban-treated group and 0.7%
in the warfarin-treated patients. Intention-to-treat analysis gave similar results to these per-protocol findings.
Use of edoxaban, Dr. Raskob concluded, "may enable more patients to stay on extended anticoagulant treatment and help reduce the burden from recurrent venous thromboembolism."
Commenting on the findings by email, Dr. Jerrold H. Levy, coauthor of an accompanying editorial, told Reuters Health, "This post-hoc analysis reports that edoxaban is an alternative to warfarin for extended use in the secondary prevention of venous thromboembolism."
Dr. Levy, of Duke University Hospital, Durham, North Carolina, concluded, "The only other study where a direct oral anticoagulant was compared with warfarin for extended use in this setting was the RE-MEDY trial that compared dabigatran with warfarin in patients for six to 36 months and found dabigatran was similar to warfarin for efficacy with a lower incidence of clinically relevant major bleeding."
This study was funded by Daiichi-Sankyo. Dr. Raskob received fees from the company during the study. Other coauthors
also have ties to the company and a number are employees of Daiichi-Sankyo.
NEW YORK (Reuters Health) - Edoxaban (Savaysa, Daiichi-Sankyo) shows advantages over warfarin in long-term treatment of patients with venous thromboembolism (VTE), according to a post-hoc analysis of multinational trial data.
As Dr. Gary Raskob told Reuters Health by email, "Our results indicate that once-daily edoxaban provides an effective
and more convenient alternative to warfarin, with lower major bleeding risk, for patients who require extended treatment
beyond three months to prevent recurrent venous thromboembolism."
In a March 22 online paper in the Lancet Haematology, Dr.Raskob, of the University of Oklahoma, Oklahoma City, and colleagues note that guidelines recommend anticoagulant treatment for at least three months. However, "The risk of recurrence is substantial for patients with unprovoked venous thromboembolism or continuing risk factors and many of these
patients need extended anticoagulation therapy beyond three months."
To shed more light on longer term effects, the team examined outcome after three to 12 months in 3,633 patients treated with heparin and edoxaban and 3,594 treated with heparin and warfarin who took part in a randomized, double-blind trial. Median treatment duration was close to 9 months.
At three months, recurrent VTE was seen in 1.1% of the edoxaban group and 1.2% of the warfarin patients. At three to six months, the corresponding proportions were 0.7% and 0.5%. At more than six but less than 12 months, they were 0.2% and 0.8%.
Among other findings was that the cumulative incidence of major bleeding was 0.3% in the edoxaban-treated group and 0.7%
in the warfarin-treated patients. Intention-to-treat analysis gave similar results to these per-protocol findings.
Use of edoxaban, Dr. Raskob concluded, "may enable more patients to stay on extended anticoagulant treatment and help reduce the burden from recurrent venous thromboembolism."
Commenting on the findings by email, Dr. Jerrold H. Levy, coauthor of an accompanying editorial, told Reuters Health, "This post-hoc analysis reports that edoxaban is an alternative to warfarin for extended use in the secondary prevention of venous thromboembolism."
Dr. Levy, of Duke University Hospital, Durham, North Carolina, concluded, "The only other study where a direct oral anticoagulant was compared with warfarin for extended use in this setting was the RE-MEDY trial that compared dabigatran with warfarin in patients for six to 36 months and found dabigatran was similar to warfarin for efficacy with a lower incidence of clinically relevant major bleeding."
This study was funded by Daiichi-Sankyo. Dr. Raskob received fees from the company during the study. Other coauthors
also have ties to the company and a number are employees of Daiichi-Sankyo.
NEW YORK (Reuters Health) - Edoxaban (Savaysa, Daiichi-Sankyo) shows advantages over warfarin in long-term treatment of patients with venous thromboembolism (VTE), according to a post-hoc analysis of multinational trial data.
As Dr. Gary Raskob told Reuters Health by email, "Our results indicate that once-daily edoxaban provides an effective
and more convenient alternative to warfarin, with lower major bleeding risk, for patients who require extended treatment
beyond three months to prevent recurrent venous thromboembolism."
In a March 22 online paper in the Lancet Haematology, Dr.Raskob, of the University of Oklahoma, Oklahoma City, and colleagues note that guidelines recommend anticoagulant treatment for at least three months. However, "The risk of recurrence is substantial for patients with unprovoked venous thromboembolism or continuing risk factors and many of these
patients need extended anticoagulation therapy beyond three months."
To shed more light on longer term effects, the team examined outcome after three to 12 months in 3,633 patients treated with heparin and edoxaban and 3,594 treated with heparin and warfarin who took part in a randomized, double-blind trial. Median treatment duration was close to 9 months.
At three months, recurrent VTE was seen in 1.1% of the edoxaban group and 1.2% of the warfarin patients. At three to six months, the corresponding proportions were 0.7% and 0.5%. At more than six but less than 12 months, they were 0.2% and 0.8%.
Among other findings was that the cumulative incidence of major bleeding was 0.3% in the edoxaban-treated group and 0.7%
in the warfarin-treated patients. Intention-to-treat analysis gave similar results to these per-protocol findings.
Use of edoxaban, Dr. Raskob concluded, "may enable more patients to stay on extended anticoagulant treatment and help reduce the burden from recurrent venous thromboembolism."
Commenting on the findings by email, Dr. Jerrold H. Levy, coauthor of an accompanying editorial, told Reuters Health, "This post-hoc analysis reports that edoxaban is an alternative to warfarin for extended use in the secondary prevention of venous thromboembolism."
Dr. Levy, of Duke University Hospital, Durham, North Carolina, concluded, "The only other study where a direct oral anticoagulant was compared with warfarin for extended use in this setting was the RE-MEDY trial that compared dabigatran with warfarin in patients for six to 36 months and found dabigatran was similar to warfarin for efficacy with a lower incidence of clinically relevant major bleeding."
This study was funded by Daiichi-Sankyo. Dr. Raskob received fees from the company during the study. Other coauthors
also have ties to the company and a number are employees of Daiichi-Sankyo.
3 Hospitalists Honored on Modern Healthcare’s List of 50 Most Influential Physician Executives and Leaders
The Society of Hospital Medicine (SHM) congratulates three hospitalists who appear on Modern Healthcare’s list of the 50 Most Influential Physician Executives and Leaders of 2016. Pediatric hospitalist Patrick Conway, MD, MSc, MHM, Deputy Administrator for Innovation and Quality and Chief Medical Officer of the Centers for Medicare and Medicaid Services (CMS), takes the No. 3 spot; national leader in healthcare quality and an architect of the hospital medicine movement, Robert M. Wachter, MD, MHM, of the University of California, San Francisco (UCSF), comes in at No. 4; and US Surgeon General and hospitalist Vivek Murthy, MD, MBA, ranks at No. 22.
In this Year of the Hospitalist, SHM’s celebration of the 20th anniversary of the New England Journal of Medicine paper by Drs. Robert Wachter and Lee Goldman that first used the term “hospitalist” to describe physicians who care for hospitalized patients, this distinct honor demonstrates hospitalists’ integral role in the hospital and health system, particularly in this era of monumental change. By nature of their work, hospitalists possess a deep understanding of how hospitals operate and how to add value to their institutions, improve patient outcomes and lead teams to solve the most complex problems both inside hospitals’ walls and in the U.S. healthcare system.
“I’m always proud to be a hospitalist, but it is in moments like these when hospitalists are recognized on the national stage that my pride is confirmed and amplified, validating that we are the change agents for improved quality of care and teamwork across the care continuum,” SHM President Brian Harte, MD, SFHM, said. “Congratulations to our colleagues and respected leaders, Drs. Conway, Wachter, and Murthy!”
The Society of Hospital Medicine (SHM) congratulates three hospitalists who appear on Modern Healthcare’s list of the 50 Most Influential Physician Executives and Leaders of 2016. Pediatric hospitalist Patrick Conway, MD, MSc, MHM, Deputy Administrator for Innovation and Quality and Chief Medical Officer of the Centers for Medicare and Medicaid Services (CMS), takes the No. 3 spot; national leader in healthcare quality and an architect of the hospital medicine movement, Robert M. Wachter, MD, MHM, of the University of California, San Francisco (UCSF), comes in at No. 4; and US Surgeon General and hospitalist Vivek Murthy, MD, MBA, ranks at No. 22.
In this Year of the Hospitalist, SHM’s celebration of the 20th anniversary of the New England Journal of Medicine paper by Drs. Robert Wachter and Lee Goldman that first used the term “hospitalist” to describe physicians who care for hospitalized patients, this distinct honor demonstrates hospitalists’ integral role in the hospital and health system, particularly in this era of monumental change. By nature of their work, hospitalists possess a deep understanding of how hospitals operate and how to add value to their institutions, improve patient outcomes and lead teams to solve the most complex problems both inside hospitals’ walls and in the U.S. healthcare system.
“I’m always proud to be a hospitalist, but it is in moments like these when hospitalists are recognized on the national stage that my pride is confirmed and amplified, validating that we are the change agents for improved quality of care and teamwork across the care continuum,” SHM President Brian Harte, MD, SFHM, said. “Congratulations to our colleagues and respected leaders, Drs. Conway, Wachter, and Murthy!”
The Society of Hospital Medicine (SHM) congratulates three hospitalists who appear on Modern Healthcare’s list of the 50 Most Influential Physician Executives and Leaders of 2016. Pediatric hospitalist Patrick Conway, MD, MSc, MHM, Deputy Administrator for Innovation and Quality and Chief Medical Officer of the Centers for Medicare and Medicaid Services (CMS), takes the No. 3 spot; national leader in healthcare quality and an architect of the hospital medicine movement, Robert M. Wachter, MD, MHM, of the University of California, San Francisco (UCSF), comes in at No. 4; and US Surgeon General and hospitalist Vivek Murthy, MD, MBA, ranks at No. 22.
In this Year of the Hospitalist, SHM’s celebration of the 20th anniversary of the New England Journal of Medicine paper by Drs. Robert Wachter and Lee Goldman that first used the term “hospitalist” to describe physicians who care for hospitalized patients, this distinct honor demonstrates hospitalists’ integral role in the hospital and health system, particularly in this era of monumental change. By nature of their work, hospitalists possess a deep understanding of how hospitals operate and how to add value to their institutions, improve patient outcomes and lead teams to solve the most complex problems both inside hospitals’ walls and in the U.S. healthcare system.
“I’m always proud to be a hospitalist, but it is in moments like these when hospitalists are recognized on the national stage that my pride is confirmed and amplified, validating that we are the change agents for improved quality of care and teamwork across the care continuum,” SHM President Brian Harte, MD, SFHM, said. “Congratulations to our colleagues and respected leaders, Drs. Conway, Wachter, and Murthy!”
Higher Risk of Cataracts After Percutaneous Coronary Intervention
NEW YORK (Reuters Health) - The risk of cataracts increases after percutaneous coronary intervention (PCI), suggesting the need for eye protection in patients undergoing these procedures, researchers from Taiwan report.
Although previous studies have identified a link between occupational radiation exposure and excess risk of cataract formation, the research in patients has been more limited. Lead eyeglasses are currently recommended for interventionists, but there are no guidelines for patient eye protection.
Dr. Yu-Tung Huang, from Kaohsiung Medical University,Kaohsiung, Taiwan, and colleagues used data from Taiwan's National Health Insurance research database to evaluate the risk of cataract surgery in 13,807 patients exposed to PCI and 27,614 patients not exposed to PCI.
Patients who underwent PCI were 25% more likely than those not exposed to PCI to have cataract surgery, according to the April 4 JAMA Internal Medicine online report.
The risk of cataract surgery increased with increasing numbers of PCI procedures, from 23% increased risk with one procedure to 29% increased risk with two to four procedures to 43% increased risk with five or more procedures.
"Because this was an observational study," they note, "we cannot establish causation, and there may be unmeasured confounders."
Nevertheless, the researchers conclude, "providing lead eyeglasses to protect patients' eyes, as is already done during cosmetic laser procedures, during the PCI procedures is recommended."
Dr. Huang did not respond to a request for comments.The authors reported no funding or disclosures.
NEW YORK (Reuters Health) - The risk of cataracts increases after percutaneous coronary intervention (PCI), suggesting the need for eye protection in patients undergoing these procedures, researchers from Taiwan report.
Although previous studies have identified a link between occupational radiation exposure and excess risk of cataract formation, the research in patients has been more limited. Lead eyeglasses are currently recommended for interventionists, but there are no guidelines for patient eye protection.
Dr. Yu-Tung Huang, from Kaohsiung Medical University,Kaohsiung, Taiwan, and colleagues used data from Taiwan's National Health Insurance research database to evaluate the risk of cataract surgery in 13,807 patients exposed to PCI and 27,614 patients not exposed to PCI.
Patients who underwent PCI were 25% more likely than those not exposed to PCI to have cataract surgery, according to the April 4 JAMA Internal Medicine online report.
The risk of cataract surgery increased with increasing numbers of PCI procedures, from 23% increased risk with one procedure to 29% increased risk with two to four procedures to 43% increased risk with five or more procedures.
"Because this was an observational study," they note, "we cannot establish causation, and there may be unmeasured confounders."
Nevertheless, the researchers conclude, "providing lead eyeglasses to protect patients' eyes, as is already done during cosmetic laser procedures, during the PCI procedures is recommended."
Dr. Huang did not respond to a request for comments.The authors reported no funding or disclosures.
NEW YORK (Reuters Health) - The risk of cataracts increases after percutaneous coronary intervention (PCI), suggesting the need for eye protection in patients undergoing these procedures, researchers from Taiwan report.
Although previous studies have identified a link between occupational radiation exposure and excess risk of cataract formation, the research in patients has been more limited. Lead eyeglasses are currently recommended for interventionists, but there are no guidelines for patient eye protection.
Dr. Yu-Tung Huang, from Kaohsiung Medical University,Kaohsiung, Taiwan, and colleagues used data from Taiwan's National Health Insurance research database to evaluate the risk of cataract surgery in 13,807 patients exposed to PCI and 27,614 patients not exposed to PCI.
Patients who underwent PCI were 25% more likely than those not exposed to PCI to have cataract surgery, according to the April 4 JAMA Internal Medicine online report.
The risk of cataract surgery increased with increasing numbers of PCI procedures, from 23% increased risk with one procedure to 29% increased risk with two to four procedures to 43% increased risk with five or more procedures.
"Because this was an observational study," they note, "we cannot establish causation, and there may be unmeasured confounders."
Nevertheless, the researchers conclude, "providing lead eyeglasses to protect patients' eyes, as is already done during cosmetic laser procedures, during the PCI procedures is recommended."
Dr. Huang did not respond to a request for comments.The authors reported no funding or disclosures.
Ten-Year Results Find that Bypass Lowers the Odds of Cardiovascular Death
NEW YORK (Reuters Health) - Ten-year results from a comparison of coronary-artery bypass grafting (CABG) with medical therapy alone has found that bypass lowers the odds of cardiovascular death by about nine percentage points in people with ischemic cardiomyopathy.
The 16% reduction in the death rate meant volunteers who received bypass grafting typically lived nearly a year and a half longer than those who received optimal nonsurgical therapy.
The findings are based on a study population of 1,212 patients with an ejection fraction of 35% or less who received bypass grafts between 2002 and 2007.
Death from any cause, the primary endpoint, occurred in 66.1% of the control patients and 58.9% of the bypass recipients (p=0.02).
When the researchers looked exclusively at cardiovascular deaths, the rates were 49.3% in the medical-therapy group and 40.5% in the group receiving bypass in addition to standard medical care (p=0.006).
The combined odds of hospitalization or death from any cause were also lower with bypass.
For death or hospitalization for cardiovascular causes, there was a 10.4 percentage point difference (p<0.001). For death or hospitalization for heart failure, the difference was 8.6 points (p<0.002). It was for 6.4 percentage points for death or hospitalization for any cause (p=0.001).
Results from the study, known as STICHES, were released April 3 at the American College of Cardiology annual scientific session in Chicago and online by the New England Journal of Medicine.
Bypass grafting "was associated with more favorable results than medical therapy alone across all clinically relevant long-term outcomes we evaluated," said the team, led by Dr. Eric Velazquez of the Duke Clinical Research Institute at Duke University, Durham, North Carolina.
The trends "resulted from a persistent and perhaps increasing effect size over time," they said. "Thus, it appears that the operative risk associated with CABG is offset by a durable effect that translates into increasing clinical benefit to at least 10 years."
Coronary artery disease kills more than 538,000 people in the United States each year. Most of the studies establishing the benefits of bypass grafting were done more than 40 years ago.
The initial version of the new study was known as STICH and it followed patients at 99 sites in 22 countries for a median of just over four and a half years. It found no significant difference between the two treatments when it came to the rate of death from any cause. However, bypass recipients were less likely to die from a cardiovascular event or to die from any cause or be hospitalized for cardiovascular causes.
STICHES extends those results to 10 years and found that any-cause death became significant.
Median survival was 7.73 years with bypass and 6.29 years without. The researchers calculated that the number needed to treat to prevent one death was 14 patients. The number needed to prevent one death from a cardiovascular cause was 11.
Other secondary measures were the rates of death combined with specific cardiovascular events. Once again, they consistently found a benefit for bypass.
There was a 15.8 percentage point difference for any cause of death or revascularization (p<0.001). For death or nonfatal myocardial infarction the difference was 6.3 points and it was for 7.2 percentage points for death or nonfatal stroke (both p=0.03).
Among the 610 people originally assigned to the bypass group, 9% did not receive a graft before the end of the trial. In the 602 assigned to medical therapy alone, 19.8% underwent bypass surgery. Eleven percent had it within the first year.
In an accompanying editorial, Drs. Robert Guyton and Andrew Smith of Emory University in Atlanta, wrote, "The STICHES 10-year results firmly extend the survival benefit of CABG in patients with advanced coronary artery disease to patients with heart failure and severe ischemic cardiomyopathy. These findings should prompt strong consideration of coronary bypass as an addition to medical therapy in shared decision making with these patients."
The National Heart Lung and Blood Institute funded this research. Two coauthors reported disclosures.
NEW YORK (Reuters Health) - Ten-year results from a comparison of coronary-artery bypass grafting (CABG) with medical therapy alone has found that bypass lowers the odds of cardiovascular death by about nine percentage points in people with ischemic cardiomyopathy.
The 16% reduction in the death rate meant volunteers who received bypass grafting typically lived nearly a year and a half longer than those who received optimal nonsurgical therapy.
The findings are based on a study population of 1,212 patients with an ejection fraction of 35% or less who received bypass grafts between 2002 and 2007.
Death from any cause, the primary endpoint, occurred in 66.1% of the control patients and 58.9% of the bypass recipients (p=0.02).
When the researchers looked exclusively at cardiovascular deaths, the rates were 49.3% in the medical-therapy group and 40.5% in the group receiving bypass in addition to standard medical care (p=0.006).
The combined odds of hospitalization or death from any cause were also lower with bypass.
For death or hospitalization for cardiovascular causes, there was a 10.4 percentage point difference (p<0.001). For death or hospitalization for heart failure, the difference was 8.6 points (p<0.002). It was for 6.4 percentage points for death or hospitalization for any cause (p=0.001).
Results from the study, known as STICHES, were released April 3 at the American College of Cardiology annual scientific session in Chicago and online by the New England Journal of Medicine.
Bypass grafting "was associated with more favorable results than medical therapy alone across all clinically relevant long-term outcomes we evaluated," said the team, led by Dr. Eric Velazquez of the Duke Clinical Research Institute at Duke University, Durham, North Carolina.
The trends "resulted from a persistent and perhaps increasing effect size over time," they said. "Thus, it appears that the operative risk associated with CABG is offset by a durable effect that translates into increasing clinical benefit to at least 10 years."
Coronary artery disease kills more than 538,000 people in the United States each year. Most of the studies establishing the benefits of bypass grafting were done more than 40 years ago.
The initial version of the new study was known as STICH and it followed patients at 99 sites in 22 countries for a median of just over four and a half years. It found no significant difference between the two treatments when it came to the rate of death from any cause. However, bypass recipients were less likely to die from a cardiovascular event or to die from any cause or be hospitalized for cardiovascular causes.
STICHES extends those results to 10 years and found that any-cause death became significant.
Median survival was 7.73 years with bypass and 6.29 years without. The researchers calculated that the number needed to treat to prevent one death was 14 patients. The number needed to prevent one death from a cardiovascular cause was 11.
Other secondary measures were the rates of death combined with specific cardiovascular events. Once again, they consistently found a benefit for bypass.
There was a 15.8 percentage point difference for any cause of death or revascularization (p<0.001). For death or nonfatal myocardial infarction the difference was 6.3 points and it was for 7.2 percentage points for death or nonfatal stroke (both p=0.03).
Among the 610 people originally assigned to the bypass group, 9% did not receive a graft before the end of the trial. In the 602 assigned to medical therapy alone, 19.8% underwent bypass surgery. Eleven percent had it within the first year.
In an accompanying editorial, Drs. Robert Guyton and Andrew Smith of Emory University in Atlanta, wrote, "The STICHES 10-year results firmly extend the survival benefit of CABG in patients with advanced coronary artery disease to patients with heart failure and severe ischemic cardiomyopathy. These findings should prompt strong consideration of coronary bypass as an addition to medical therapy in shared decision making with these patients."
The National Heart Lung and Blood Institute funded this research. Two coauthors reported disclosures.
NEW YORK (Reuters Health) - Ten-year results from a comparison of coronary-artery bypass grafting (CABG) with medical therapy alone has found that bypass lowers the odds of cardiovascular death by about nine percentage points in people with ischemic cardiomyopathy.
The 16% reduction in the death rate meant volunteers who received bypass grafting typically lived nearly a year and a half longer than those who received optimal nonsurgical therapy.
The findings are based on a study population of 1,212 patients with an ejection fraction of 35% or less who received bypass grafts between 2002 and 2007.
Death from any cause, the primary endpoint, occurred in 66.1% of the control patients and 58.9% of the bypass recipients (p=0.02).
When the researchers looked exclusively at cardiovascular deaths, the rates were 49.3% in the medical-therapy group and 40.5% in the group receiving bypass in addition to standard medical care (p=0.006).
The combined odds of hospitalization or death from any cause were also lower with bypass.
For death or hospitalization for cardiovascular causes, there was a 10.4 percentage point difference (p<0.001). For death or hospitalization for heart failure, the difference was 8.6 points (p<0.002). It was for 6.4 percentage points for death or hospitalization for any cause (p=0.001).
Results from the study, known as STICHES, were released April 3 at the American College of Cardiology annual scientific session in Chicago and online by the New England Journal of Medicine.
Bypass grafting "was associated with more favorable results than medical therapy alone across all clinically relevant long-term outcomes we evaluated," said the team, led by Dr. Eric Velazquez of the Duke Clinical Research Institute at Duke University, Durham, North Carolina.
The trends "resulted from a persistent and perhaps increasing effect size over time," they said. "Thus, it appears that the operative risk associated with CABG is offset by a durable effect that translates into increasing clinical benefit to at least 10 years."
Coronary artery disease kills more than 538,000 people in the United States each year. Most of the studies establishing the benefits of bypass grafting were done more than 40 years ago.
The initial version of the new study was known as STICH and it followed patients at 99 sites in 22 countries for a median of just over four and a half years. It found no significant difference between the two treatments when it came to the rate of death from any cause. However, bypass recipients were less likely to die from a cardiovascular event or to die from any cause or be hospitalized for cardiovascular causes.
STICHES extends those results to 10 years and found that any-cause death became significant.
Median survival was 7.73 years with bypass and 6.29 years without. The researchers calculated that the number needed to treat to prevent one death was 14 patients. The number needed to prevent one death from a cardiovascular cause was 11.
Other secondary measures were the rates of death combined with specific cardiovascular events. Once again, they consistently found a benefit for bypass.
There was a 15.8 percentage point difference for any cause of death or revascularization (p<0.001). For death or nonfatal myocardial infarction the difference was 6.3 points and it was for 7.2 percentage points for death or nonfatal stroke (both p=0.03).
Among the 610 people originally assigned to the bypass group, 9% did not receive a graft before the end of the trial. In the 602 assigned to medical therapy alone, 19.8% underwent bypass surgery. Eleven percent had it within the first year.
In an accompanying editorial, Drs. Robert Guyton and Andrew Smith of Emory University in Atlanta, wrote, "The STICHES 10-year results firmly extend the survival benefit of CABG in patients with advanced coronary artery disease to patients with heart failure and severe ischemic cardiomyopathy. These findings should prompt strong consideration of coronary bypass as an addition to medical therapy in shared decision making with these patients."
The National Heart Lung and Blood Institute funded this research. Two coauthors reported disclosures.
Study Finds: Blood Pressure Medication and Stain Cholesterol Fighter Decreases the Risk of Heart Attack and Stroke in Patients with Hypertension and Moderate Risk of Heart Disease
(Reuters) - Patients with hypertension and moderate risk of heart disease slashed their long-term risk of heart attack and stroke 40 percent by taking a blood pressure medication as well as a statin cholesterol fighter, according to a large global study that could change medical practice.
Results from the trial, called HOPE-3, could prod far more doctors to add a statin to antihypertensive therapy for such patients who have no prior history of heart attack or stroke, researchers said.
The data was presented on Saturday at the annual scientific session of the American College of Cardiology in Chicago.
To enroll in the trial, patients had to have at least one risk factor for heart disease such as obesity or smoking, in addition to being over 60 for women and over 55 for men.
"Intermediate-risk people with hypertension had a clear benefit when taking both a statin and an agent that lowers blood pressure," Dr. Salim Yusuf, a professor of cardiology at McMaster University in Ontario, Canada who headed the 12,000-patient global study, said in an interview.
Patients with systolic blood pressure of 140 and higher were deemed in the study to have high blood pressure. They experienced a 40 percent reduced risk of heart attack and stroke over a six-year period when taking AstraZeneca Plc's statin Crestor (rosuvastatin) as well as a combination tablet containing blood pressure treatment candesartan and the diuretic hydrochlorothiazide.
Patients with normal or low systolic pressure had the same approximate 25 percent reduction in cardiovascular events as seen among patients in one arm of the study who took only statins.
Yusuf said the trial underscores that if a patient at moderate heart risk has high blood pressure, defined as 140 or higher, "give them both a statin and a blood pressure medication as a matter of course." He said statins are not automatically given now to patients with hypertension that are at only moderate risk of heart attack or stroke.
Yusuf's trial included research centers in China, India, Latin America, Africa and Canada, but not the United States because of far greater research costs there. The trial was funded by the Canadian Institutes of Health Research and AstraZeneca.
A separate study published in November found that lowering blood pressure to below 120 dramatically reduced heart failure and risk of death in adults aged 50 and older. But the five-year U.S. government-sponsored study of more than 9,300 patients showed a higher rate of adverse side effects, including kidney damage, in the aggressively treated patients.
(Reuters) - Patients with hypertension and moderate risk of heart disease slashed their long-term risk of heart attack and stroke 40 percent by taking a blood pressure medication as well as a statin cholesterol fighter, according to a large global study that could change medical practice.
Results from the trial, called HOPE-3, could prod far more doctors to add a statin to antihypertensive therapy for such patients who have no prior history of heart attack or stroke, researchers said.
The data was presented on Saturday at the annual scientific session of the American College of Cardiology in Chicago.
To enroll in the trial, patients had to have at least one risk factor for heart disease such as obesity or smoking, in addition to being over 60 for women and over 55 for men.
"Intermediate-risk people with hypertension had a clear benefit when taking both a statin and an agent that lowers blood pressure," Dr. Salim Yusuf, a professor of cardiology at McMaster University in Ontario, Canada who headed the 12,000-patient global study, said in an interview.
Patients with systolic blood pressure of 140 and higher were deemed in the study to have high blood pressure. They experienced a 40 percent reduced risk of heart attack and stroke over a six-year period when taking AstraZeneca Plc's statin Crestor (rosuvastatin) as well as a combination tablet containing blood pressure treatment candesartan and the diuretic hydrochlorothiazide.
Patients with normal or low systolic pressure had the same approximate 25 percent reduction in cardiovascular events as seen among patients in one arm of the study who took only statins.
Yusuf said the trial underscores that if a patient at moderate heart risk has high blood pressure, defined as 140 or higher, "give them both a statin and a blood pressure medication as a matter of course." He said statins are not automatically given now to patients with hypertension that are at only moderate risk of heart attack or stroke.
Yusuf's trial included research centers in China, India, Latin America, Africa and Canada, but not the United States because of far greater research costs there. The trial was funded by the Canadian Institutes of Health Research and AstraZeneca.
A separate study published in November found that lowering blood pressure to below 120 dramatically reduced heart failure and risk of death in adults aged 50 and older. But the five-year U.S. government-sponsored study of more than 9,300 patients showed a higher rate of adverse side effects, including kidney damage, in the aggressively treated patients.
(Reuters) - Patients with hypertension and moderate risk of heart disease slashed their long-term risk of heart attack and stroke 40 percent by taking a blood pressure medication as well as a statin cholesterol fighter, according to a large global study that could change medical practice.
Results from the trial, called HOPE-3, could prod far more doctors to add a statin to antihypertensive therapy for such patients who have no prior history of heart attack or stroke, researchers said.
The data was presented on Saturday at the annual scientific session of the American College of Cardiology in Chicago.
To enroll in the trial, patients had to have at least one risk factor for heart disease such as obesity or smoking, in addition to being over 60 for women and over 55 for men.
"Intermediate-risk people with hypertension had a clear benefit when taking both a statin and an agent that lowers blood pressure," Dr. Salim Yusuf, a professor of cardiology at McMaster University in Ontario, Canada who headed the 12,000-patient global study, said in an interview.
Patients with systolic blood pressure of 140 and higher were deemed in the study to have high blood pressure. They experienced a 40 percent reduced risk of heart attack and stroke over a six-year period when taking AstraZeneca Plc's statin Crestor (rosuvastatin) as well as a combination tablet containing blood pressure treatment candesartan and the diuretic hydrochlorothiazide.
Patients with normal or low systolic pressure had the same approximate 25 percent reduction in cardiovascular events as seen among patients in one arm of the study who took only statins.
Yusuf said the trial underscores that if a patient at moderate heart risk has high blood pressure, defined as 140 or higher, "give them both a statin and a blood pressure medication as a matter of course." He said statins are not automatically given now to patients with hypertension that are at only moderate risk of heart attack or stroke.
Yusuf's trial included research centers in China, India, Latin America, Africa and Canada, but not the United States because of far greater research costs there. The trial was funded by the Canadian Institutes of Health Research and AstraZeneca.
A separate study published in November found that lowering blood pressure to below 120 dramatically reduced heart failure and risk of death in adults aged 50 and older. But the five-year U.S. government-sponsored study of more than 9,300 patients showed a higher rate of adverse side effects, including kidney damage, in the aggressively treated patients.
Failure of Promising Treatments for Controlling Cholesterol Leads to More Studies
(Reuters) - New ways of controlling cholesterol, including possibly directly injecting "good" HDL cholesterol into patients, need to be studied following the failure of promising treatments from Eli Lilly, Pfizer Inc and Roche Holding AG, according to top heart researchers.
Lilly in October halted a 12,000-patient study of its experimental drug evacetrapib, an oral medication that in smaller earlier studies slashed "bad" LDL cholesterol and doubled levels of HDL.
But improved cholesterol levels did not prevent heart attacks and strokes, diminishing hopes for the approach to treating heart disease - by raising HDL through blockage of a protein called CETP.
Roche in 2012 scrapped its own CETP inhibitor after it also failed to help patients. Pfizer's similar drug was discontinued in 2006 after being linked to deaths in trials.
Although Merck & Co continues to develop its own CETP inhibitor in a 30,000-patient study expected to be completed next year, researchers on Sunday said the failures of the Lilly, Roche and Pfizer drugs bode poorly for it.
"Merck's drug is the fourth shot on goal for CETP inhibitors, but with disappointment or lack of success for the other agents you have to be increasingly pessimistic" about the class of drugs, said Dr. Stephen Nicholls, deputy director of the South Australian Health and Medical Research Institute in Adelaide, Australia. He was a lead investigator for the failed trial of Lilly's drug.
Nicholls and Dr. Steve Nissen, the head of cardiology for the Cleveland Clinic, who co-lead the evacetrapib study, on Sunday reviewed the baffling evacetrapib data in a presentation at the annual scientific sessions of the American College of Cardiology in Chicago.
"This drug lowered LDL by 37 percent and raised HDL by 130 percent and had absolutely no effect" on preventing deaths and heart attacks, Nissen said in an interview.
Although other ways of raising HDL cholesterol might eventually prove protective, Nissen said all attempts so far have been fruitless.
Nicholls said he remains hopeful of future HDL therapies and is testing whether artificial HDL can be made in the laboratory and injected directly into high-risk heart patients. "There is enthusiasm it may be able to shrink plaque" in heart arteries, he said.
He said he is studying variations of that approach with French drugmaker Cerenis Therapeutics and the Medicines Company. Nicholls said another possible approach would be to instruct the liver to make more HDL.
(Reuters) - New ways of controlling cholesterol, including possibly directly injecting "good" HDL cholesterol into patients, need to be studied following the failure of promising treatments from Eli Lilly, Pfizer Inc and Roche Holding AG, according to top heart researchers.
Lilly in October halted a 12,000-patient study of its experimental drug evacetrapib, an oral medication that in smaller earlier studies slashed "bad" LDL cholesterol and doubled levels of HDL.
But improved cholesterol levels did not prevent heart attacks and strokes, diminishing hopes for the approach to treating heart disease - by raising HDL through blockage of a protein called CETP.
Roche in 2012 scrapped its own CETP inhibitor after it also failed to help patients. Pfizer's similar drug was discontinued in 2006 after being linked to deaths in trials.
Although Merck & Co continues to develop its own CETP inhibitor in a 30,000-patient study expected to be completed next year, researchers on Sunday said the failures of the Lilly, Roche and Pfizer drugs bode poorly for it.
"Merck's drug is the fourth shot on goal for CETP inhibitors, but with disappointment or lack of success for the other agents you have to be increasingly pessimistic" about the class of drugs, said Dr. Stephen Nicholls, deputy director of the South Australian Health and Medical Research Institute in Adelaide, Australia. He was a lead investigator for the failed trial of Lilly's drug.
Nicholls and Dr. Steve Nissen, the head of cardiology for the Cleveland Clinic, who co-lead the evacetrapib study, on Sunday reviewed the baffling evacetrapib data in a presentation at the annual scientific sessions of the American College of Cardiology in Chicago.
"This drug lowered LDL by 37 percent and raised HDL by 130 percent and had absolutely no effect" on preventing deaths and heart attacks, Nissen said in an interview.
Although other ways of raising HDL cholesterol might eventually prove protective, Nissen said all attempts so far have been fruitless.
Nicholls said he remains hopeful of future HDL therapies and is testing whether artificial HDL can be made in the laboratory and injected directly into high-risk heart patients. "There is enthusiasm it may be able to shrink plaque" in heart arteries, he said.
He said he is studying variations of that approach with French drugmaker Cerenis Therapeutics and the Medicines Company. Nicholls said another possible approach would be to instruct the liver to make more HDL.
(Reuters) - New ways of controlling cholesterol, including possibly directly injecting "good" HDL cholesterol into patients, need to be studied following the failure of promising treatments from Eli Lilly, Pfizer Inc and Roche Holding AG, according to top heart researchers.
Lilly in October halted a 12,000-patient study of its experimental drug evacetrapib, an oral medication that in smaller earlier studies slashed "bad" LDL cholesterol and doubled levels of HDL.
But improved cholesterol levels did not prevent heart attacks and strokes, diminishing hopes for the approach to treating heart disease - by raising HDL through blockage of a protein called CETP.
Roche in 2012 scrapped its own CETP inhibitor after it also failed to help patients. Pfizer's similar drug was discontinued in 2006 after being linked to deaths in trials.
Although Merck & Co continues to develop its own CETP inhibitor in a 30,000-patient study expected to be completed next year, researchers on Sunday said the failures of the Lilly, Roche and Pfizer drugs bode poorly for it.
"Merck's drug is the fourth shot on goal for CETP inhibitors, but with disappointment or lack of success for the other agents you have to be increasingly pessimistic" about the class of drugs, said Dr. Stephen Nicholls, deputy director of the South Australian Health and Medical Research Institute in Adelaide, Australia. He was a lead investigator for the failed trial of Lilly's drug.
Nicholls and Dr. Steve Nissen, the head of cardiology for the Cleveland Clinic, who co-lead the evacetrapib study, on Sunday reviewed the baffling evacetrapib data in a presentation at the annual scientific sessions of the American College of Cardiology in Chicago.
"This drug lowered LDL by 37 percent and raised HDL by 130 percent and had absolutely no effect" on preventing deaths and heart attacks, Nissen said in an interview.
Although other ways of raising HDL cholesterol might eventually prove protective, Nissen said all attempts so far have been fruitless.
Nicholls said he remains hopeful of future HDL therapies and is testing whether artificial HDL can be made in the laboratory and injected directly into high-risk heart patients. "There is enthusiasm it may be able to shrink plaque" in heart arteries, he said.
He said he is studying variations of that approach with French drugmaker Cerenis Therapeutics and the Medicines Company. Nicholls said another possible approach would be to instruct the liver to make more HDL.
HM16 Q&A: How Do You try to Make a Positive Impact on Public Health in Your Daily Work?
U.S. Surgeon General Vivek Murthy, MD, MBA, formerly a hospitalist in Boston, said in a keynote speech that hospitalists have more power than they think to improve public health. The Hospitalist asked HM16 attendees: As a hospitalist, how empowered do you feel in having a positive impact on public health, and how do you try to make such an impact in your daily work?
L. Scott Sussman, MD, associate director of the hospitalist service, Yale-New Haven Hospital, Conn.
“We see patients in a time of crisis, so they’re coming in, they’re scared, they’re sick, and it’s a really teachable moment. Because once we start treating them, they start feeling better … [and] what happens is we start to gain their confidence. We use that as a point where we can say, ‘Let’s talk about your other health issues.’”
Nazima Allaudeen, MD, hospitalist, director of quality improvement, Veterans Affairs Palo Alto Healthcare System, Calif.
“I don’t feel like I do [have an impact] very much, but I think his talk kind of made me feel like we probably can and should do a lot more. … You know how sometimes you’ll get an email that has a standard thing that you can send to your congressperson about something like that? I feel like we were all like, ‘Yes, we should,’ and it would have been a great opportunity. … I feel like we kind of need it spelled out for us, you know? But it’s a good reminder.”
Gilbert Wergowske, MD, TeamHealth hospitalist, Vallejo, Calif.
“We try to arrange the best follow-up you can. That’s a difficult problem. The area that I work in is not an area that really has a lot of options … as far as giving care is concerned. So it’s really important to try to get in touch with the primary care provider directly. If that doesn’t happen, most of our patients will end up using the ED as their primary care provider. It’s important mainly for two reasons: Disjointed care like that is suboptimal; the patients do not get the best benefit from the care. And also it’s very wasteful because tests and procedures tend to be repeated over and over again.”
Cory Ritter, MD, hospitalist, soon to be working at the Veterans Affairs Medical Center, Houston
“After Dr. Murthy’s talk, I feel much more empowered to call up my local government representatives and express what I feel should be done for public health in our area. Before today, I didn’t think there was much we could do except for supporting the Society of Hospital Medicine because I know they’ve been very engaged in Obamacare and CMS. But otherwise, before today, it wasn’t much on my radar. … On a one-on-one level, you have some impact, but I’ve never really thought much about a larger public health–scale type of impact.”
U.S. Surgeon General Vivek Murthy, MD, MBA, formerly a hospitalist in Boston, said in a keynote speech that hospitalists have more power than they think to improve public health. The Hospitalist asked HM16 attendees: As a hospitalist, how empowered do you feel in having a positive impact on public health, and how do you try to make such an impact in your daily work?
L. Scott Sussman, MD, associate director of the hospitalist service, Yale-New Haven Hospital, Conn.
“We see patients in a time of crisis, so they’re coming in, they’re scared, they’re sick, and it’s a really teachable moment. Because once we start treating them, they start feeling better … [and] what happens is we start to gain their confidence. We use that as a point where we can say, ‘Let’s talk about your other health issues.’”
Nazima Allaudeen, MD, hospitalist, director of quality improvement, Veterans Affairs Palo Alto Healthcare System, Calif.
“I don’t feel like I do [have an impact] very much, but I think his talk kind of made me feel like we probably can and should do a lot more. … You know how sometimes you’ll get an email that has a standard thing that you can send to your congressperson about something like that? I feel like we were all like, ‘Yes, we should,’ and it would have been a great opportunity. … I feel like we kind of need it spelled out for us, you know? But it’s a good reminder.”
Gilbert Wergowske, MD, TeamHealth hospitalist, Vallejo, Calif.
“We try to arrange the best follow-up you can. That’s a difficult problem. The area that I work in is not an area that really has a lot of options … as far as giving care is concerned. So it’s really important to try to get in touch with the primary care provider directly. If that doesn’t happen, most of our patients will end up using the ED as their primary care provider. It’s important mainly for two reasons: Disjointed care like that is suboptimal; the patients do not get the best benefit from the care. And also it’s very wasteful because tests and procedures tend to be repeated over and over again.”
Cory Ritter, MD, hospitalist, soon to be working at the Veterans Affairs Medical Center, Houston
“After Dr. Murthy’s talk, I feel much more empowered to call up my local government representatives and express what I feel should be done for public health in our area. Before today, I didn’t think there was much we could do except for supporting the Society of Hospital Medicine because I know they’ve been very engaged in Obamacare and CMS. But otherwise, before today, it wasn’t much on my radar. … On a one-on-one level, you have some impact, but I’ve never really thought much about a larger public health–scale type of impact.”
U.S. Surgeon General Vivek Murthy, MD, MBA, formerly a hospitalist in Boston, said in a keynote speech that hospitalists have more power than they think to improve public health. The Hospitalist asked HM16 attendees: As a hospitalist, how empowered do you feel in having a positive impact on public health, and how do you try to make such an impact in your daily work?
L. Scott Sussman, MD, associate director of the hospitalist service, Yale-New Haven Hospital, Conn.
“We see patients in a time of crisis, so they’re coming in, they’re scared, they’re sick, and it’s a really teachable moment. Because once we start treating them, they start feeling better … [and] what happens is we start to gain their confidence. We use that as a point where we can say, ‘Let’s talk about your other health issues.’”
Nazima Allaudeen, MD, hospitalist, director of quality improvement, Veterans Affairs Palo Alto Healthcare System, Calif.
“I don’t feel like I do [have an impact] very much, but I think his talk kind of made me feel like we probably can and should do a lot more. … You know how sometimes you’ll get an email that has a standard thing that you can send to your congressperson about something like that? I feel like we were all like, ‘Yes, we should,’ and it would have been a great opportunity. … I feel like we kind of need it spelled out for us, you know? But it’s a good reminder.”
Gilbert Wergowske, MD, TeamHealth hospitalist, Vallejo, Calif.
“We try to arrange the best follow-up you can. That’s a difficult problem. The area that I work in is not an area that really has a lot of options … as far as giving care is concerned. So it’s really important to try to get in touch with the primary care provider directly. If that doesn’t happen, most of our patients will end up using the ED as their primary care provider. It’s important mainly for two reasons: Disjointed care like that is suboptimal; the patients do not get the best benefit from the care. And also it’s very wasteful because tests and procedures tend to be repeated over and over again.”
Cory Ritter, MD, hospitalist, soon to be working at the Veterans Affairs Medical Center, Houston
“After Dr. Murthy’s talk, I feel much more empowered to call up my local government representatives and express what I feel should be done for public health in our area. Before today, I didn’t think there was much we could do except for supporting the Society of Hospital Medicine because I know they’ve been very engaged in Obamacare and CMS. But otherwise, before today, it wasn’t much on my radar. … On a one-on-one level, you have some impact, but I’ve never really thought much about a larger public health–scale type of impact.”
Improve Your Treatment of VTE
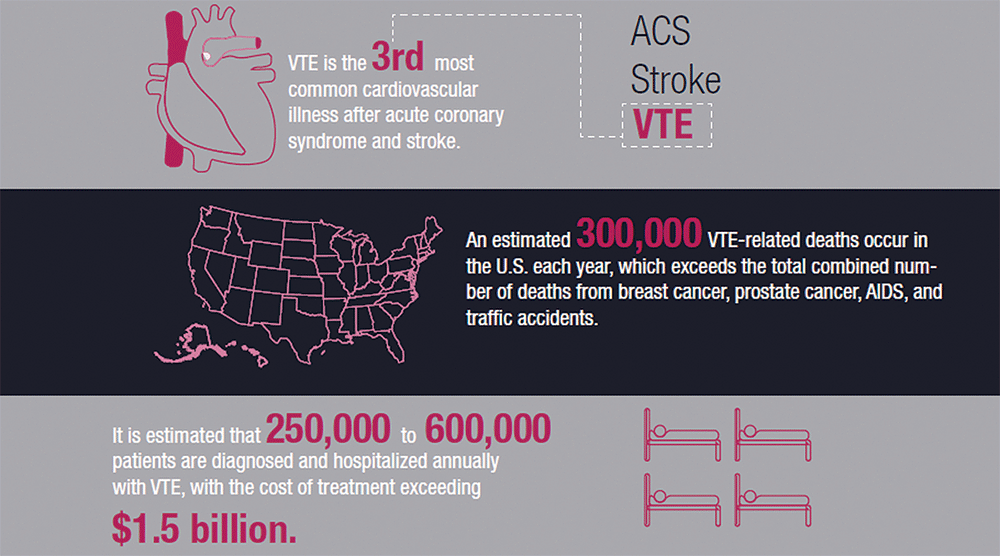
It is estimated that 250,000 to 600,000 patients are diagnosed and hospitalized annually with venous thromboembolism (VTE), the third most common cardiovascular illness after acute coronary syndrome and stroke. An estimated 300,000 VTE-related deaths occur in the U.S. each year, which exceeds the total number of deaths from breast cancer, prostate cancer, AIDS, and traffic accidents combined.
Communication among care providers is an essential part of medical care; it influences patients’ quality of life and effective disease treatment. Hospitalists educate both patients and providers regarding appropriate steps to take to improve care transitions and reduce any associated risks.
To ensure hospitalists have the latest information about diagnosis, treatment, and transition of inpatients with VTE, SHM is developing:
- An online tool kit, including a literature review; implementation guide; and other references, materials, and tools (e.g., discharge instructions and checklists)
- A webinar series with free CME
To be notified when SHM’s VTE resources become available, visit www.hospitalmedicine.org/VTEtreatment.
It is estimated that 250,000 to 600,000 patients are diagnosed and hospitalized annually with venous thromboembolism (VTE), the third most common cardiovascular illness after acute coronary syndrome and stroke. An estimated 300,000 VTE-related deaths occur in the U.S. each year, which exceeds the total number of deaths from breast cancer, prostate cancer, AIDS, and traffic accidents combined.
Communication among care providers is an essential part of medical care; it influences patients’ quality of life and effective disease treatment. Hospitalists educate both patients and providers regarding appropriate steps to take to improve care transitions and reduce any associated risks.
To ensure hospitalists have the latest information about diagnosis, treatment, and transition of inpatients with VTE, SHM is developing:
- An online tool kit, including a literature review; implementation guide; and other references, materials, and tools (e.g., discharge instructions and checklists)
- A webinar series with free CME
To be notified when SHM’s VTE resources become available, visit www.hospitalmedicine.org/VTEtreatment.
It is estimated that 250,000 to 600,000 patients are diagnosed and hospitalized annually with venous thromboembolism (VTE), the third most common cardiovascular illness after acute coronary syndrome and stroke. An estimated 300,000 VTE-related deaths occur in the U.S. each year, which exceeds the total number of deaths from breast cancer, prostate cancer, AIDS, and traffic accidents combined.
Communication among care providers is an essential part of medical care; it influences patients’ quality of life and effective disease treatment. Hospitalists educate both patients and providers regarding appropriate steps to take to improve care transitions and reduce any associated risks.
To ensure hospitalists have the latest information about diagnosis, treatment, and transition of inpatients with VTE, SHM is developing:
- An online tool kit, including a literature review; implementation guide; and other references, materials, and tools (e.g., discharge instructions and checklists)
- A webinar series with free CME
To be notified when SHM’s VTE resources become available, visit www.hospitalmedicine.org/VTEtreatment.