User login
The role of aspirin in primary prevention of cardiovascular disease
Background: Previous studies have shown that aspirin reduces the relative risk of cardiovascular disease (CVD) but also increases the relative risk of bleeding. It is unclear if there are patients without known CVD in whom the absolute risk reduction of CVD outweighs the absolute risk of bleeding. Prognostic CVD and bleeding risk models allow for an assessment of absolute risks and primary preventive interventions.
Study design: Individualized risk-benefit analysis based on sex-specific risk scores and estimates from PREDICT cohort data.
Setting: Primary care practices in New Zealand.
Synopsis: Using the New Zealand–based PREDICT online tool, 245,048 patients had their CVD risk assessed and did not meet exclusion criteria. The online tool predicts CVD events avoided and bleeding events caused by aspirin. When one CVD event was equivalent to one major bleeding event, 2.5% of women and 12.1% of men were classified as benefiting from aspirin (more CVD events avoided than bleeding events caused). When one CVD event was equivalent to two major bleeding events, 21.4% of women and 40.7% of men were classified as benefiting from aspirin. The net-benefit subgroups were older, and had higher baseline 5-year CVD risk, fewer risk factors for bleeding, higher systolic blood pressure, and a higher total cholesterol to HDL cholesterol ratio. Ethnicity and socioeconomic index also influenced benefit or harm.
With use of the upper and lower limits of 95% confidence intervals for models, there were considerable ranges of benefit versus harm. Sex-specific risk scores and meta-analysis have intrinsic uncertainties and results potentially not generalizable outside New Zealand population. Ultimate decision to use aspirin requires shared decision making.
Bottom line: Some patients are likely to derive a net benefit from aspirin for primary prevention of CVD. Risk-benefit models with online tools can help providers and patients estimate these factors to inform shared decision making.
Citation: Selak V et al. Personalized prediction of cardiovascular benefits and bleeding harms for aspirin for primary prevention, a benefit-harm analysis. Ann Intern Med. 2019;71(8):529-39.
Dr. Rupp is a hospitalist and clinical instructor of medicine at the University of Utah, Salt Lake City.
Background: Previous studies have shown that aspirin reduces the relative risk of cardiovascular disease (CVD) but also increases the relative risk of bleeding. It is unclear if there are patients without known CVD in whom the absolute risk reduction of CVD outweighs the absolute risk of bleeding. Prognostic CVD and bleeding risk models allow for an assessment of absolute risks and primary preventive interventions.
Study design: Individualized risk-benefit analysis based on sex-specific risk scores and estimates from PREDICT cohort data.
Setting: Primary care practices in New Zealand.
Synopsis: Using the New Zealand–based PREDICT online tool, 245,048 patients had their CVD risk assessed and did not meet exclusion criteria. The online tool predicts CVD events avoided and bleeding events caused by aspirin. When one CVD event was equivalent to one major bleeding event, 2.5% of women and 12.1% of men were classified as benefiting from aspirin (more CVD events avoided than bleeding events caused). When one CVD event was equivalent to two major bleeding events, 21.4% of women and 40.7% of men were classified as benefiting from aspirin. The net-benefit subgroups were older, and had higher baseline 5-year CVD risk, fewer risk factors for bleeding, higher systolic blood pressure, and a higher total cholesterol to HDL cholesterol ratio. Ethnicity and socioeconomic index also influenced benefit or harm.
With use of the upper and lower limits of 95% confidence intervals for models, there were considerable ranges of benefit versus harm. Sex-specific risk scores and meta-analysis have intrinsic uncertainties and results potentially not generalizable outside New Zealand population. Ultimate decision to use aspirin requires shared decision making.
Bottom line: Some patients are likely to derive a net benefit from aspirin for primary prevention of CVD. Risk-benefit models with online tools can help providers and patients estimate these factors to inform shared decision making.
Citation: Selak V et al. Personalized prediction of cardiovascular benefits and bleeding harms for aspirin for primary prevention, a benefit-harm analysis. Ann Intern Med. 2019;71(8):529-39.
Dr. Rupp is a hospitalist and clinical instructor of medicine at the University of Utah, Salt Lake City.
Background: Previous studies have shown that aspirin reduces the relative risk of cardiovascular disease (CVD) but also increases the relative risk of bleeding. It is unclear if there are patients without known CVD in whom the absolute risk reduction of CVD outweighs the absolute risk of bleeding. Prognostic CVD and bleeding risk models allow for an assessment of absolute risks and primary preventive interventions.
Study design: Individualized risk-benefit analysis based on sex-specific risk scores and estimates from PREDICT cohort data.
Setting: Primary care practices in New Zealand.
Synopsis: Using the New Zealand–based PREDICT online tool, 245,048 patients had their CVD risk assessed and did not meet exclusion criteria. The online tool predicts CVD events avoided and bleeding events caused by aspirin. When one CVD event was equivalent to one major bleeding event, 2.5% of women and 12.1% of men were classified as benefiting from aspirin (more CVD events avoided than bleeding events caused). When one CVD event was equivalent to two major bleeding events, 21.4% of women and 40.7% of men were classified as benefiting from aspirin. The net-benefit subgroups were older, and had higher baseline 5-year CVD risk, fewer risk factors for bleeding, higher systolic blood pressure, and a higher total cholesterol to HDL cholesterol ratio. Ethnicity and socioeconomic index also influenced benefit or harm.
With use of the upper and lower limits of 95% confidence intervals for models, there were considerable ranges of benefit versus harm. Sex-specific risk scores and meta-analysis have intrinsic uncertainties and results potentially not generalizable outside New Zealand population. Ultimate decision to use aspirin requires shared decision making.
Bottom line: Some patients are likely to derive a net benefit from aspirin for primary prevention of CVD. Risk-benefit models with online tools can help providers and patients estimate these factors to inform shared decision making.
Citation: Selak V et al. Personalized prediction of cardiovascular benefits and bleeding harms for aspirin for primary prevention, a benefit-harm analysis. Ann Intern Med. 2019;71(8):529-39.
Dr. Rupp is a hospitalist and clinical instructor of medicine at the University of Utah, Salt Lake City.
COVID-19 in 2020: Deaths and disparities
COVID-19 was the third-leading cause of death in the United States in 2020, but that mortality burden did not fall evenly along racial/ethnic lines, according to a provisional report from the Centers for Disease Control and Prevention.
Only heart disease and cancer caused more deaths than SARS-CoV-2, which took the lives of almost 378,000 Americans last year, Farida B. Ahmad, MPH, and associates at the National Center for Health Statistics noted March 31 in the Morbidity and Mortality Weekly Report.
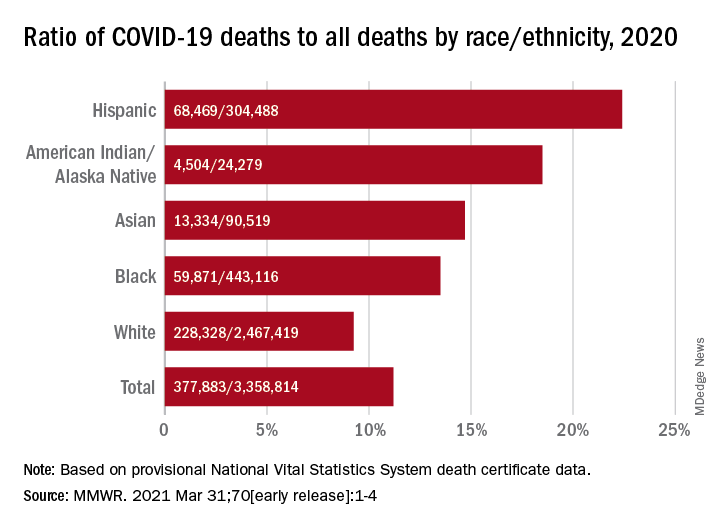
That represents 11.2% of the almost 3.36 million total deaths recorded in 2020. The racial/ethnics demographics, however, show that 22.4% of all deaths among Hispanic Americans were COVID-19–related, as were 18.6% of deaths in American Indians/Alaska Natives. Deaths among Asian persons, at 14.7%, and African Americans, at 13.5%, were closer but still above the national figure, while Whites (9.3%) were the only major subgroup below it, based on data from the National Vital Statistics System.
Age-adjusted death rates tell a somewhat different story: American Indian/Alaska native persons were highest with a rate of 187.8 COVID-19–associated deaths per 100,000 standard population, with Hispanic persons second at 164.3 per 100,000. Blacks were next at 151.1 deaths per 100,000, but Whites had a higher rate (72.5) than did Asian Americans (66.7), the CDC investigators reported.
“During January-December 2020, the estimated 2020 age-adjusted death rate increased for the first time since 2017, with an increase of 15.9% compared with 2019, from 715.2 to 828.7 deaths per 100,000 population,” they wrote, noting that “certain categories of race (i.e., AI/AN and Asian) and Hispanic ethnicity reported on death certificates might have been misclassified, possibly resulting in underestimates of death rates for some groups.”
COVID-19 was the third-leading cause of death in the United States in 2020, but that mortality burden did not fall evenly along racial/ethnic lines, according to a provisional report from the Centers for Disease Control and Prevention.

Only heart disease and cancer caused more deaths than SARS-CoV-2, which took the lives of almost 378,000 Americans last year, Farida B. Ahmad, MPH, and associates at the National Center for Health Statistics noted March 31 in the Morbidity and Mortality Weekly Report.
That represents 11.2% of the almost 3.36 million total deaths recorded in 2020. The racial/ethnics demographics, however, show that 22.4% of all deaths among Hispanic Americans were COVID-19–related, as were 18.6% of deaths in American Indians/Alaska Natives. Deaths among Asian persons, at 14.7%, and African Americans, at 13.5%, were closer but still above the national figure, while Whites (9.3%) were the only major subgroup below it, based on data from the National Vital Statistics System.
Age-adjusted death rates tell a somewhat different story: American Indian/Alaska native persons were highest with a rate of 187.8 COVID-19–associated deaths per 100,000 standard population, with Hispanic persons second at 164.3 per 100,000. Blacks were next at 151.1 deaths per 100,000, but Whites had a higher rate (72.5) than did Asian Americans (66.7), the CDC investigators reported.
“During January-December 2020, the estimated 2020 age-adjusted death rate increased for the first time since 2017, with an increase of 15.9% compared with 2019, from 715.2 to 828.7 deaths per 100,000 population,” they wrote, noting that “certain categories of race (i.e., AI/AN and Asian) and Hispanic ethnicity reported on death certificates might have been misclassified, possibly resulting in underestimates of death rates for some groups.”
COVID-19 was the third-leading cause of death in the United States in 2020, but that mortality burden did not fall evenly along racial/ethnic lines, according to a provisional report from the Centers for Disease Control and Prevention.

Only heart disease and cancer caused more deaths than SARS-CoV-2, which took the lives of almost 378,000 Americans last year, Farida B. Ahmad, MPH, and associates at the National Center for Health Statistics noted March 31 in the Morbidity and Mortality Weekly Report.
That represents 11.2% of the almost 3.36 million total deaths recorded in 2020. The racial/ethnics demographics, however, show that 22.4% of all deaths among Hispanic Americans were COVID-19–related, as were 18.6% of deaths in American Indians/Alaska Natives. Deaths among Asian persons, at 14.7%, and African Americans, at 13.5%, were closer but still above the national figure, while Whites (9.3%) were the only major subgroup below it, based on data from the National Vital Statistics System.
Age-adjusted death rates tell a somewhat different story: American Indian/Alaska native persons were highest with a rate of 187.8 COVID-19–associated deaths per 100,000 standard population, with Hispanic persons second at 164.3 per 100,000. Blacks were next at 151.1 deaths per 100,000, but Whites had a higher rate (72.5) than did Asian Americans (66.7), the CDC investigators reported.
“During January-December 2020, the estimated 2020 age-adjusted death rate increased for the first time since 2017, with an increase of 15.9% compared with 2019, from 715.2 to 828.7 deaths per 100,000 population,” they wrote, noting that “certain categories of race (i.e., AI/AN and Asian) and Hispanic ethnicity reported on death certificates might have been misclassified, possibly resulting in underestimates of death rates for some groups.”
FROM MMWR
Pfizer: Vaccine shown 100% effective in children aged 12-15
The study enrolled 2,260 adolescents aged 12-15. No infections were reported in the group given the vaccine produced by Pfizer and its European partner, BioNTech, the release said. The placebo group reported 18 cases of COVID-19.
The vaccinated children showed a strong antibody response with no serious side effects.
Albert Bourla, PhD, chairman and CEO of Pfizer, said the company plans to seek Food and Drug Administration emergency use authorization, which could allow this age group to be vaccinated before the start of the next school year. Pfizer will also seek authorization from the European Medicines Agency.
“We share the urgency to expand the authorization of our vaccine to use in younger populations and are encouraged by the clinical trial data from adolescents between the ages of 12 and 15,” Dr. Bourla said in the release.
The clinical trials showed a stronger response in children aged 12-15 than the 95% effectiveness reported in clinical trials in adults. The Pfizer vaccine is now authorized to be given to people aged 16 and up in the United States.
Health experts said the clinical trials – while not peer-reviewed – amounted to very good news.
“The sooner that we can get vaccines into as many people as possible, regardless of their age, the sooner we will be able to really feel like we’re ending this pandemic for good,” Angela Rasmussen, PhD, a virologist affiliated with Georgetown University in Washington, told The New York Times.
Anthony Fauci, MD, director of the National Institute of Allergy and Infectious Diseases, recently said that getting children vaccinated is an important step toward achieving herd immunity.
“We don’t really know what that magical point of herd immunity is, but we do know that if we get the overwhelming population vaccinated, we’re going to be in good shape,” he said earlier in March during a hearing of the Senate Health, Education, Labor, and Pensions Committee. “We ultimately would like to get and have to get children into that mix.”
Pfizer said it started clinical trials during the week of March 23 with children aged 5-11 and will next start trials with children aged 2-5, followed by children aged 6 months to 2 years. Vaccine makers Moderna and AstraZeneca also have started clinical trials in younger children.
A version of this article first appeared on WebMD.com.
The study enrolled 2,260 adolescents aged 12-15. No infections were reported in the group given the vaccine produced by Pfizer and its European partner, BioNTech, the release said. The placebo group reported 18 cases of COVID-19.
The vaccinated children showed a strong antibody response with no serious side effects.
Albert Bourla, PhD, chairman and CEO of Pfizer, said the company plans to seek Food and Drug Administration emergency use authorization, which could allow this age group to be vaccinated before the start of the next school year. Pfizer will also seek authorization from the European Medicines Agency.
“We share the urgency to expand the authorization of our vaccine to use in younger populations and are encouraged by the clinical trial data from adolescents between the ages of 12 and 15,” Dr. Bourla said in the release.
The clinical trials showed a stronger response in children aged 12-15 than the 95% effectiveness reported in clinical trials in adults. The Pfizer vaccine is now authorized to be given to people aged 16 and up in the United States.
Health experts said the clinical trials – while not peer-reviewed – amounted to very good news.
“The sooner that we can get vaccines into as many people as possible, regardless of their age, the sooner we will be able to really feel like we’re ending this pandemic for good,” Angela Rasmussen, PhD, a virologist affiliated with Georgetown University in Washington, told The New York Times.
Anthony Fauci, MD, director of the National Institute of Allergy and Infectious Diseases, recently said that getting children vaccinated is an important step toward achieving herd immunity.
“We don’t really know what that magical point of herd immunity is, but we do know that if we get the overwhelming population vaccinated, we’re going to be in good shape,” he said earlier in March during a hearing of the Senate Health, Education, Labor, and Pensions Committee. “We ultimately would like to get and have to get children into that mix.”
Pfizer said it started clinical trials during the week of March 23 with children aged 5-11 and will next start trials with children aged 2-5, followed by children aged 6 months to 2 years. Vaccine makers Moderna and AstraZeneca also have started clinical trials in younger children.
A version of this article first appeared on WebMD.com.
The study enrolled 2,260 adolescents aged 12-15. No infections were reported in the group given the vaccine produced by Pfizer and its European partner, BioNTech, the release said. The placebo group reported 18 cases of COVID-19.
The vaccinated children showed a strong antibody response with no serious side effects.
Albert Bourla, PhD, chairman and CEO of Pfizer, said the company plans to seek Food and Drug Administration emergency use authorization, which could allow this age group to be vaccinated before the start of the next school year. Pfizer will also seek authorization from the European Medicines Agency.
“We share the urgency to expand the authorization of our vaccine to use in younger populations and are encouraged by the clinical trial data from adolescents between the ages of 12 and 15,” Dr. Bourla said in the release.
The clinical trials showed a stronger response in children aged 12-15 than the 95% effectiveness reported in clinical trials in adults. The Pfizer vaccine is now authorized to be given to people aged 16 and up in the United States.
Health experts said the clinical trials – while not peer-reviewed – amounted to very good news.
“The sooner that we can get vaccines into as many people as possible, regardless of their age, the sooner we will be able to really feel like we’re ending this pandemic for good,” Angela Rasmussen, PhD, a virologist affiliated with Georgetown University in Washington, told The New York Times.
Anthony Fauci, MD, director of the National Institute of Allergy and Infectious Diseases, recently said that getting children vaccinated is an important step toward achieving herd immunity.
“We don’t really know what that magical point of herd immunity is, but we do know that if we get the overwhelming population vaccinated, we’re going to be in good shape,” he said earlier in March during a hearing of the Senate Health, Education, Labor, and Pensions Committee. “We ultimately would like to get and have to get children into that mix.”
Pfizer said it started clinical trials during the week of March 23 with children aged 5-11 and will next start trials with children aged 2-5, followed by children aged 6 months to 2 years. Vaccine makers Moderna and AstraZeneca also have started clinical trials in younger children.
A version of this article first appeared on WebMD.com.
CDC adds new medical conditions to COVID-19 high-risk list
The Centers for Disease Control and Prevention has added several new medical conditions to its list of those that predispose adults to more severe COVID-19 illness.
Conditions that had previously been categorized as “might be” placing individuals at increased risk – but now are listed as high risk – include type 1 diabetes (in addition to type 2), moderate-to-severe asthma, liver disease, dementia or other neurologic conditions, stroke/cerebrovascular disease, HIV infection, cystic fibrosis, and overweight (in addition to obesity).
Substance use disorders, which hadn’t been previously listed, are now also considered high risk.
The new list groups together certain categories, such as chronic lung diseases (chronic obstructive pulmonary disease, asthma, cystic fibrosis, etc) and heart conditions (heart failure, coronary artery disease, hypertension, etc).
Both diabetes types are now grouped under “diabetes.”
The added medical conditions were posted on the CDC website’s COVID-19 page on March 29.
Type 1 diabetes and other conditions now priority for vaccination
The CDC refers to the medical conditions list as phase 1c in regard to COVID-19 vaccine prioritization, which means that anyone with any of these conditions can now be prioritized for vaccination, following those in groups 1a (frontline essential workers and those in long-term care facilities) and 1b (people aged 65-74 years; other essential workers; and people aged 16-64 years with underlying conditions that increase the risk of serious, life-threatening complications from COVID-19).
But in many cases, multiple states have already either fully opened up vaccine eligibility to all adults or have created their own lists of underlying high-risk medical conditions, CDC spokeswoman Kristen Nordlund told this news organization.
No conditions have been removed from the list.
In January, the American Diabetes Association and 18 other organizations sent a letter to the CDC requesting that type 1 diabetes be prioritized along with type 2, based on data from studies showing people with both types to be at high risk for severe COVID-19 illness.
Now, ADA says, “this updated guidance will help to address the fact that in many states, millions of people with type 1 diabetes have not been prioritized equally, slowing their access to critical vaccines.”
While awaiting this latest CDC move, ADA had been urging state governors to prioritize type 1 and type 2 diabetes equally. As of now, 38 states and the District of Columbia had either done so or announced that they would.
A version of this article first appeared on Medscape.com.
The Centers for Disease Control and Prevention has added several new medical conditions to its list of those that predispose adults to more severe COVID-19 illness.
Conditions that had previously been categorized as “might be” placing individuals at increased risk – but now are listed as high risk – include type 1 diabetes (in addition to type 2), moderate-to-severe asthma, liver disease, dementia or other neurologic conditions, stroke/cerebrovascular disease, HIV infection, cystic fibrosis, and overweight (in addition to obesity).
Substance use disorders, which hadn’t been previously listed, are now also considered high risk.
The new list groups together certain categories, such as chronic lung diseases (chronic obstructive pulmonary disease, asthma, cystic fibrosis, etc) and heart conditions (heart failure, coronary artery disease, hypertension, etc).
Both diabetes types are now grouped under “diabetes.”
The added medical conditions were posted on the CDC website’s COVID-19 page on March 29.
Type 1 diabetes and other conditions now priority for vaccination
The CDC refers to the medical conditions list as phase 1c in regard to COVID-19 vaccine prioritization, which means that anyone with any of these conditions can now be prioritized for vaccination, following those in groups 1a (frontline essential workers and those in long-term care facilities) and 1b (people aged 65-74 years; other essential workers; and people aged 16-64 years with underlying conditions that increase the risk of serious, life-threatening complications from COVID-19).
But in many cases, multiple states have already either fully opened up vaccine eligibility to all adults or have created their own lists of underlying high-risk medical conditions, CDC spokeswoman Kristen Nordlund told this news organization.
No conditions have been removed from the list.
In January, the American Diabetes Association and 18 other organizations sent a letter to the CDC requesting that type 1 diabetes be prioritized along with type 2, based on data from studies showing people with both types to be at high risk for severe COVID-19 illness.
Now, ADA says, “this updated guidance will help to address the fact that in many states, millions of people with type 1 diabetes have not been prioritized equally, slowing their access to critical vaccines.”
While awaiting this latest CDC move, ADA had been urging state governors to prioritize type 1 and type 2 diabetes equally. As of now, 38 states and the District of Columbia had either done so or announced that they would.
A version of this article first appeared on Medscape.com.
The Centers for Disease Control and Prevention has added several new medical conditions to its list of those that predispose adults to more severe COVID-19 illness.
Conditions that had previously been categorized as “might be” placing individuals at increased risk – but now are listed as high risk – include type 1 diabetes (in addition to type 2), moderate-to-severe asthma, liver disease, dementia or other neurologic conditions, stroke/cerebrovascular disease, HIV infection, cystic fibrosis, and overweight (in addition to obesity).
Substance use disorders, which hadn’t been previously listed, are now also considered high risk.
The new list groups together certain categories, such as chronic lung diseases (chronic obstructive pulmonary disease, asthma, cystic fibrosis, etc) and heart conditions (heart failure, coronary artery disease, hypertension, etc).
Both diabetes types are now grouped under “diabetes.”
The added medical conditions were posted on the CDC website’s COVID-19 page on March 29.
Type 1 diabetes and other conditions now priority for vaccination
The CDC refers to the medical conditions list as phase 1c in regard to COVID-19 vaccine prioritization, which means that anyone with any of these conditions can now be prioritized for vaccination, following those in groups 1a (frontline essential workers and those in long-term care facilities) and 1b (people aged 65-74 years; other essential workers; and people aged 16-64 years with underlying conditions that increase the risk of serious, life-threatening complications from COVID-19).
But in many cases, multiple states have already either fully opened up vaccine eligibility to all adults or have created their own lists of underlying high-risk medical conditions, CDC spokeswoman Kristen Nordlund told this news organization.
No conditions have been removed from the list.
In January, the American Diabetes Association and 18 other organizations sent a letter to the CDC requesting that type 1 diabetes be prioritized along with type 2, based on data from studies showing people with both types to be at high risk for severe COVID-19 illness.
Now, ADA says, “this updated guidance will help to address the fact that in many states, millions of people with type 1 diabetes have not been prioritized equally, slowing their access to critical vaccines.”
While awaiting this latest CDC move, ADA had been urging state governors to prioritize type 1 and type 2 diabetes equally. As of now, 38 states and the District of Columbia had either done so or announced that they would.
A version of this article first appeared on Medscape.com.
Black inpatients at higher risk of poor safety outcomes
One expert says these findings should be a call to action for hospitals and physicians.
The Urban Institute, which is funded in part by the Robert Wood Johnson Foundation, looked at differences in Black and White patient safety measures among adults receiving inpatient care in 26 states.
Care quality was measured by the rate of preventable adverse hospital patient safety events per 1,000 at-risk discharges using data from the Agency for Healthcare Research and Quality (AHRQ).
Researchers compared experience by race on 11 patient safety indicators – four related to general patient safety, and seven linked to risk of adverse events with surgical procedures.
Surgical risk differences significant
The gaps were widest surrounding surgical care. Black patients were 7.9 percentage points more likely to be in a hospital considered low quality across all surgical safety measures. They were 4.9 percentage points more likely to be admitted to a hospital considered low quality across all general safety indicators.
“If you’re a Black patient getting surgery – relative to a White patient – in my study, you were 25% less likely to be in a hospital that prevented hemorrhage during surgery; you were 26% less likely to be in a hospital that prevented postoperative respiratory failure; and you were more than 30% less likely to be in a hospital that is effective in preventing postoperative sepsis,” Anuj Gangopadhyaya, PhD, senior research associate at the Urban Institute, said in an interview.
According to the report, Black patients were also 31.9% less likely than were White patients to be admitted into hospitals considered high quality in preventing pressure ulcers and 22.8% less likely to be in a hospital good at preventing iatrogenic pneumothorax.
Dr. Gangopadhyaya said this may be the first study to compare the numbers after the inception of the Affordable Care Act. These data were collected in 2017, 3 years after the core elements of the ACA kicked in.
He said that although the ACA has done much to narrow the racial gap in terms of insurance coverage, it has not been effective in reducing the heightened safety risk to Black patients in the hospital.
‘Shocking, though not surprising’
Uché Blackstock, MD, founder and CEO of Advancing Health Equity in New York City, called the findings “shocking, though not surprising.”
Though these data were collected before COVID-19, the pandemic has exposed profound racial inequities, she noted.
She cited the example of Susan Moore, MD, a Black physician in Carmel, Ind., who died from COVID-19 at age 52 in December after experiencing what she said was systemic racism in her care.
“We saw in the death of Dr. Susan Moore that even having a formal education and being a physician is not protective for Black patients. These findings only reaffirm what we already know – that Black patients receive worse and lower-quality care than White patients,” Dr. Blackstock said in an interview.
“These findings are not a result of Black patients’ individual choices as is often suggested, but rather the results of a health care system that has devalued the lives of Black patients and inherently provides poorer quality of care to them.”
Dr. Blackstock said this report represents a call to action.
Health care institutions must, she said, “look inward at the intentional and critical antiracism work that must be done on provider, organizational, and systems levels by allocating the necessary resources, continuing to track disaggregated health metrics, and committing to structural change within health care systems.”
Resources instead of penalties?
Dr. Gangopadhyaya said the second phase of the research will compare safety outcomes between Black and White patients in the same hospital. Those results will shed more light on what’s driving the differences in risk on safety measures.
He acknowledged that, particularly in an emergency, there is little choice involved with which hospital a patient enters. Patients typically go to a hospital in their neighborhood. And it’s well established that ZIP codes can determine health care outcomes.
But he suspects the differences cannot be explained simply by socioeconomic factors.
He pointed out that previous research has found disparities among Black and White patients in the same neighborhoods.
In one part of this study, researchers narrowed the comparison to Black and White adults with Medicare coverage, with similar provider networks and reimbursement structure, to test whether insurance was playing a significant role.
“Even among that group, you still see the persistent differences in the safety risks driven by the hospitals patients are admitted to,” Dr. Gangopadhyaya said.
He suggests two policy approaches to address the gaps: Either find ways for high-quality hospitals to reach more people of color, or find out what’s keeping the low-quality hospitals from implementing the practices that are effective in high-quality hospitals.
Currently, the ACA has penalties in place when hospitals score low for specific safety risks, he noted, saying that approach doesn’t appear to be working.
“Perhaps instead of penalizing hospitals, we might want to consider providing resources to hospitals that help them better adopt the successful protocols in their high-quality counterparts,” he said.
Dr. Gangopadhyaya has disclosed no relevant financial relationships. Dr. Blackstock has disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
This article was updated 4/2/21.
One expert says these findings should be a call to action for hospitals and physicians.
The Urban Institute, which is funded in part by the Robert Wood Johnson Foundation, looked at differences in Black and White patient safety measures among adults receiving inpatient care in 26 states.
Care quality was measured by the rate of preventable adverse hospital patient safety events per 1,000 at-risk discharges using data from the Agency for Healthcare Research and Quality (AHRQ).
Researchers compared experience by race on 11 patient safety indicators – four related to general patient safety, and seven linked to risk of adverse events with surgical procedures.
Surgical risk differences significant
The gaps were widest surrounding surgical care. Black patients were 7.9 percentage points more likely to be in a hospital considered low quality across all surgical safety measures. They were 4.9 percentage points more likely to be admitted to a hospital considered low quality across all general safety indicators.
“If you’re a Black patient getting surgery – relative to a White patient – in my study, you were 25% less likely to be in a hospital that prevented hemorrhage during surgery; you were 26% less likely to be in a hospital that prevented postoperative respiratory failure; and you were more than 30% less likely to be in a hospital that is effective in preventing postoperative sepsis,” Anuj Gangopadhyaya, PhD, senior research associate at the Urban Institute, said in an interview.
According to the report, Black patients were also 31.9% less likely than were White patients to be admitted into hospitals considered high quality in preventing pressure ulcers and 22.8% less likely to be in a hospital good at preventing iatrogenic pneumothorax.
Dr. Gangopadhyaya said this may be the first study to compare the numbers after the inception of the Affordable Care Act. These data were collected in 2017, 3 years after the core elements of the ACA kicked in.
He said that although the ACA has done much to narrow the racial gap in terms of insurance coverage, it has not been effective in reducing the heightened safety risk to Black patients in the hospital.
‘Shocking, though not surprising’
Uché Blackstock, MD, founder and CEO of Advancing Health Equity in New York City, called the findings “shocking, though not surprising.”
Though these data were collected before COVID-19, the pandemic has exposed profound racial inequities, she noted.
She cited the example of Susan Moore, MD, a Black physician in Carmel, Ind., who died from COVID-19 at age 52 in December after experiencing what she said was systemic racism in her care.
“We saw in the death of Dr. Susan Moore that even having a formal education and being a physician is not protective for Black patients. These findings only reaffirm what we already know – that Black patients receive worse and lower-quality care than White patients,” Dr. Blackstock said in an interview.
“These findings are not a result of Black patients’ individual choices as is often suggested, but rather the results of a health care system that has devalued the lives of Black patients and inherently provides poorer quality of care to them.”
Dr. Blackstock said this report represents a call to action.
Health care institutions must, she said, “look inward at the intentional and critical antiracism work that must be done on provider, organizational, and systems levels by allocating the necessary resources, continuing to track disaggregated health metrics, and committing to structural change within health care systems.”
Resources instead of penalties?
Dr. Gangopadhyaya said the second phase of the research will compare safety outcomes between Black and White patients in the same hospital. Those results will shed more light on what’s driving the differences in risk on safety measures.
He acknowledged that, particularly in an emergency, there is little choice involved with which hospital a patient enters. Patients typically go to a hospital in their neighborhood. And it’s well established that ZIP codes can determine health care outcomes.
But he suspects the differences cannot be explained simply by socioeconomic factors.
He pointed out that previous research has found disparities among Black and White patients in the same neighborhoods.
In one part of this study, researchers narrowed the comparison to Black and White adults with Medicare coverage, with similar provider networks and reimbursement structure, to test whether insurance was playing a significant role.
“Even among that group, you still see the persistent differences in the safety risks driven by the hospitals patients are admitted to,” Dr. Gangopadhyaya said.
He suggests two policy approaches to address the gaps: Either find ways for high-quality hospitals to reach more people of color, or find out what’s keeping the low-quality hospitals from implementing the practices that are effective in high-quality hospitals.
Currently, the ACA has penalties in place when hospitals score low for specific safety risks, he noted, saying that approach doesn’t appear to be working.
“Perhaps instead of penalizing hospitals, we might want to consider providing resources to hospitals that help them better adopt the successful protocols in their high-quality counterparts,” he said.
Dr. Gangopadhyaya has disclosed no relevant financial relationships. Dr. Blackstock has disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
This article was updated 4/2/21.
One expert says these findings should be a call to action for hospitals and physicians.
The Urban Institute, which is funded in part by the Robert Wood Johnson Foundation, looked at differences in Black and White patient safety measures among adults receiving inpatient care in 26 states.
Care quality was measured by the rate of preventable adverse hospital patient safety events per 1,000 at-risk discharges using data from the Agency for Healthcare Research and Quality (AHRQ).
Researchers compared experience by race on 11 patient safety indicators – four related to general patient safety, and seven linked to risk of adverse events with surgical procedures.
Surgical risk differences significant
The gaps were widest surrounding surgical care. Black patients were 7.9 percentage points more likely to be in a hospital considered low quality across all surgical safety measures. They were 4.9 percentage points more likely to be admitted to a hospital considered low quality across all general safety indicators.
“If you’re a Black patient getting surgery – relative to a White patient – in my study, you were 25% less likely to be in a hospital that prevented hemorrhage during surgery; you were 26% less likely to be in a hospital that prevented postoperative respiratory failure; and you were more than 30% less likely to be in a hospital that is effective in preventing postoperative sepsis,” Anuj Gangopadhyaya, PhD, senior research associate at the Urban Institute, said in an interview.
According to the report, Black patients were also 31.9% less likely than were White patients to be admitted into hospitals considered high quality in preventing pressure ulcers and 22.8% less likely to be in a hospital good at preventing iatrogenic pneumothorax.
Dr. Gangopadhyaya said this may be the first study to compare the numbers after the inception of the Affordable Care Act. These data were collected in 2017, 3 years after the core elements of the ACA kicked in.
He said that although the ACA has done much to narrow the racial gap in terms of insurance coverage, it has not been effective in reducing the heightened safety risk to Black patients in the hospital.
‘Shocking, though not surprising’
Uché Blackstock, MD, founder and CEO of Advancing Health Equity in New York City, called the findings “shocking, though not surprising.”
Though these data were collected before COVID-19, the pandemic has exposed profound racial inequities, she noted.
She cited the example of Susan Moore, MD, a Black physician in Carmel, Ind., who died from COVID-19 at age 52 in December after experiencing what she said was systemic racism in her care.
“We saw in the death of Dr. Susan Moore that even having a formal education and being a physician is not protective for Black patients. These findings only reaffirm what we already know – that Black patients receive worse and lower-quality care than White patients,” Dr. Blackstock said in an interview.
“These findings are not a result of Black patients’ individual choices as is often suggested, but rather the results of a health care system that has devalued the lives of Black patients and inherently provides poorer quality of care to them.”
Dr. Blackstock said this report represents a call to action.
Health care institutions must, she said, “look inward at the intentional and critical antiracism work that must be done on provider, organizational, and systems levels by allocating the necessary resources, continuing to track disaggregated health metrics, and committing to structural change within health care systems.”
Resources instead of penalties?
Dr. Gangopadhyaya said the second phase of the research will compare safety outcomes between Black and White patients in the same hospital. Those results will shed more light on what’s driving the differences in risk on safety measures.
He acknowledged that, particularly in an emergency, there is little choice involved with which hospital a patient enters. Patients typically go to a hospital in their neighborhood. And it’s well established that ZIP codes can determine health care outcomes.
But he suspects the differences cannot be explained simply by socioeconomic factors.
He pointed out that previous research has found disparities among Black and White patients in the same neighborhoods.
In one part of this study, researchers narrowed the comparison to Black and White adults with Medicare coverage, with similar provider networks and reimbursement structure, to test whether insurance was playing a significant role.
“Even among that group, you still see the persistent differences in the safety risks driven by the hospitals patients are admitted to,” Dr. Gangopadhyaya said.
He suggests two policy approaches to address the gaps: Either find ways for high-quality hospitals to reach more people of color, or find out what’s keeping the low-quality hospitals from implementing the practices that are effective in high-quality hospitals.
Currently, the ACA has penalties in place when hospitals score low for specific safety risks, he noted, saying that approach doesn’t appear to be working.
“Perhaps instead of penalizing hospitals, we might want to consider providing resources to hospitals that help them better adopt the successful protocols in their high-quality counterparts,” he said.
Dr. Gangopadhyaya has disclosed no relevant financial relationships. Dr. Blackstock has disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
This article was updated 4/2/21.
Delirium risk factors identified in ICU cancer patients
Hematology-oncology patients who receive treatment in the intensive care unit often develop delirium, and according to new findings, mechanical ventilation, high-dose corticosteroid use, and brain metastases were identified as independent risk factors.
Roughly half of all hematology-oncology patients who were admitted to the ICU experienced delirium, explained lead author Rachel Klosko, PharmD, PGY-2 cardiology pharmacy resident at the Ohio State University, Columbus.
“Delirium was associated with increased mortality, an increase in hospital length of stay, and increased length of stay in the ICU,” she said.
Dr. Klosko presented the study results at the at the Critical Care Congress sponsored by the Society of Critical Care Medicine (SCCM), which was held virtually this year.
Delirium is an acute and fluctuating disturbance of consciousness and cognition and fluctuates in severity. Critically ill patients are subject to numerous risk factors for delirium. “It can occur in independently of any known neurological disorder,” said Dr. Klosko, adding that its occurrence has been associated with poorer outcomes in ICU patients.
In this study, Dr. Klosko and colleagues sought to determine the incidence of delirium in cancer patients who were admitted to the ICU, as well as identify the associated risk factors and recognize potential consequences of the development of delirium in this patient population.
They conducted a single center, retrospective, cohort study that evaluated patients between the ages of 18 and 89 years who were admitted to the hematology-oncology medical or surgical ICU between July 1, 2018, and June 30, 2019.
The study’s primary endpoint was the incidence of delirium within 7 days of ICU admission, defined as two positive Confusion Assessment Method for the ICU (CAM-ICU) assessments within 24 hours. Patients identified with delirium were compared to those without it, for the evaluation of secondary endpoints that included hospital mortality and ICU and hospital length of stay. The researchers also sought to identify independent risk factors for delirium in this population.
A total of 244 patients were included in the final analysis. Of this group, 125 (51.2%) experienced delirium during their stay in the ICU, and 119 (48.8%) did not.
Mortality in the delirium group was significantly higher at 32.8% vs. 15.1% (P = .001). In addition, the delirium group was associated with significantly higher ICU length of stay (6 days vs. 3 days, P < .001) and hospital length of stay (21 days vs. 12 days, P < .001).
“When comparing the baseline characteristics between the two groups, the delirium group had a longer hospital length prior to ICU admission, a higher SOFA score, a higher rate of brain metastases, a higher rate of shock, and higher receipt of high-dose steroids, benzodiazepines, and immunotherapy,” said Dr. Klosko.
After multivariable regression, four variables were included in the final model. Among patients with delirium, the SOFA score increased by 25% (odds ratio[OR] 1.25, P < .001), while the odds of delirium were almost four times higher among those treated with high-dose corticosteroids (OR 3.79, P = .004). Delirium was also eight times higher (OR 8.48, P < .001) among those who received mechanical ventilation and five times higher in (OR 5.38, P = .015) in patients with brain metastases.
Dr. Klosko noted that the main limitations for this study were that it was a single center retrospective analysis, and that patients were reviewed within the first 7 days of ICU admission. “This potentially missed patients who developed delirium outside of this time frame,” she said. In addition, “too few patients received high-dose benzodiazepines,” and “none of the patients received continuous neuromuscular blockade.”
However, in “contrast to these limitations, this is the largest study to date that has analyzed delirium in this population,” Dr. Klosko said.
Commenting on the study, Brenda Pun, DNP, RN, director of data quality at the Vanderbilt Critical Illness, Brain Dysfunction, and Survivorship Center, Nashville, Tenn., pointed out that the goal of this study was to describe delirium in this specific population. “But I will take a step backward and say that they are just confirming that these patients look like other ICU patients in many regards,” she said.
She explained that the sicker patients are, the higher the rates of delirium. “We have implemented strategies to lower these rates, and they have improved,” Dr. Pun said. “Ten years ago, I would say that 80% of patients who were on a ventilator would have delirium but now the rates are around 50% and that’s what we are typically seeing now.”
Dr. Pun emphasized that this study shows that delirium is like the “canary in the coal mine” or a red flag. “It’s a sign that something is wrong and that we need to pay attention, because the patient’s outcome may be worse,” she said. “So this is saying that we need to see if there is something that can be changed or modified to decrease the incidence of delirium—these are important questions.”
There was no outside sponsor. The authors had no disclosures. Dr. Pun has no disclosures.
Hematology-oncology patients who receive treatment in the intensive care unit often develop delirium, and according to new findings, mechanical ventilation, high-dose corticosteroid use, and brain metastases were identified as independent risk factors.
Roughly half of all hematology-oncology patients who were admitted to the ICU experienced delirium, explained lead author Rachel Klosko, PharmD, PGY-2 cardiology pharmacy resident at the Ohio State University, Columbus.
“Delirium was associated with increased mortality, an increase in hospital length of stay, and increased length of stay in the ICU,” she said.
Dr. Klosko presented the study results at the at the Critical Care Congress sponsored by the Society of Critical Care Medicine (SCCM), which was held virtually this year.
Delirium is an acute and fluctuating disturbance of consciousness and cognition and fluctuates in severity. Critically ill patients are subject to numerous risk factors for delirium. “It can occur in independently of any known neurological disorder,” said Dr. Klosko, adding that its occurrence has been associated with poorer outcomes in ICU patients.
In this study, Dr. Klosko and colleagues sought to determine the incidence of delirium in cancer patients who were admitted to the ICU, as well as identify the associated risk factors and recognize potential consequences of the development of delirium in this patient population.
They conducted a single center, retrospective, cohort study that evaluated patients between the ages of 18 and 89 years who were admitted to the hematology-oncology medical or surgical ICU between July 1, 2018, and June 30, 2019.
The study’s primary endpoint was the incidence of delirium within 7 days of ICU admission, defined as two positive Confusion Assessment Method for the ICU (CAM-ICU) assessments within 24 hours. Patients identified with delirium were compared to those without it, for the evaluation of secondary endpoints that included hospital mortality and ICU and hospital length of stay. The researchers also sought to identify independent risk factors for delirium in this population.
A total of 244 patients were included in the final analysis. Of this group, 125 (51.2%) experienced delirium during their stay in the ICU, and 119 (48.8%) did not.
Mortality in the delirium group was significantly higher at 32.8% vs. 15.1% (P = .001). In addition, the delirium group was associated with significantly higher ICU length of stay (6 days vs. 3 days, P < .001) and hospital length of stay (21 days vs. 12 days, P < .001).
“When comparing the baseline characteristics between the two groups, the delirium group had a longer hospital length prior to ICU admission, a higher SOFA score, a higher rate of brain metastases, a higher rate of shock, and higher receipt of high-dose steroids, benzodiazepines, and immunotherapy,” said Dr. Klosko.
After multivariable regression, four variables were included in the final model. Among patients with delirium, the SOFA score increased by 25% (odds ratio[OR] 1.25, P < .001), while the odds of delirium were almost four times higher among those treated with high-dose corticosteroids (OR 3.79, P = .004). Delirium was also eight times higher (OR 8.48, P < .001) among those who received mechanical ventilation and five times higher in (OR 5.38, P = .015) in patients with brain metastases.
Dr. Klosko noted that the main limitations for this study were that it was a single center retrospective analysis, and that patients were reviewed within the first 7 days of ICU admission. “This potentially missed patients who developed delirium outside of this time frame,” she said. In addition, “too few patients received high-dose benzodiazepines,” and “none of the patients received continuous neuromuscular blockade.”
However, in “contrast to these limitations, this is the largest study to date that has analyzed delirium in this population,” Dr. Klosko said.
Commenting on the study, Brenda Pun, DNP, RN, director of data quality at the Vanderbilt Critical Illness, Brain Dysfunction, and Survivorship Center, Nashville, Tenn., pointed out that the goal of this study was to describe delirium in this specific population. “But I will take a step backward and say that they are just confirming that these patients look like other ICU patients in many regards,” she said.
She explained that the sicker patients are, the higher the rates of delirium. “We have implemented strategies to lower these rates, and they have improved,” Dr. Pun said. “Ten years ago, I would say that 80% of patients who were on a ventilator would have delirium but now the rates are around 50% and that’s what we are typically seeing now.”
Dr. Pun emphasized that this study shows that delirium is like the “canary in the coal mine” or a red flag. “It’s a sign that something is wrong and that we need to pay attention, because the patient’s outcome may be worse,” she said. “So this is saying that we need to see if there is something that can be changed or modified to decrease the incidence of delirium—these are important questions.”
There was no outside sponsor. The authors had no disclosures. Dr. Pun has no disclosures.
Hematology-oncology patients who receive treatment in the intensive care unit often develop delirium, and according to new findings, mechanical ventilation, high-dose corticosteroid use, and brain metastases were identified as independent risk factors.
Roughly half of all hematology-oncology patients who were admitted to the ICU experienced delirium, explained lead author Rachel Klosko, PharmD, PGY-2 cardiology pharmacy resident at the Ohio State University, Columbus.
“Delirium was associated with increased mortality, an increase in hospital length of stay, and increased length of stay in the ICU,” she said.
Dr. Klosko presented the study results at the at the Critical Care Congress sponsored by the Society of Critical Care Medicine (SCCM), which was held virtually this year.
Delirium is an acute and fluctuating disturbance of consciousness and cognition and fluctuates in severity. Critically ill patients are subject to numerous risk factors for delirium. “It can occur in independently of any known neurological disorder,” said Dr. Klosko, adding that its occurrence has been associated with poorer outcomes in ICU patients.
In this study, Dr. Klosko and colleagues sought to determine the incidence of delirium in cancer patients who were admitted to the ICU, as well as identify the associated risk factors and recognize potential consequences of the development of delirium in this patient population.
They conducted a single center, retrospective, cohort study that evaluated patients between the ages of 18 and 89 years who were admitted to the hematology-oncology medical or surgical ICU between July 1, 2018, and June 30, 2019.
The study’s primary endpoint was the incidence of delirium within 7 days of ICU admission, defined as two positive Confusion Assessment Method for the ICU (CAM-ICU) assessments within 24 hours. Patients identified with delirium were compared to those without it, for the evaluation of secondary endpoints that included hospital mortality and ICU and hospital length of stay. The researchers also sought to identify independent risk factors for delirium in this population.
A total of 244 patients were included in the final analysis. Of this group, 125 (51.2%) experienced delirium during their stay in the ICU, and 119 (48.8%) did not.
Mortality in the delirium group was significantly higher at 32.8% vs. 15.1% (P = .001). In addition, the delirium group was associated with significantly higher ICU length of stay (6 days vs. 3 days, P < .001) and hospital length of stay (21 days vs. 12 days, P < .001).
“When comparing the baseline characteristics between the two groups, the delirium group had a longer hospital length prior to ICU admission, a higher SOFA score, a higher rate of brain metastases, a higher rate of shock, and higher receipt of high-dose steroids, benzodiazepines, and immunotherapy,” said Dr. Klosko.
After multivariable regression, four variables were included in the final model. Among patients with delirium, the SOFA score increased by 25% (odds ratio[OR] 1.25, P < .001), while the odds of delirium were almost four times higher among those treated with high-dose corticosteroids (OR 3.79, P = .004). Delirium was also eight times higher (OR 8.48, P < .001) among those who received mechanical ventilation and five times higher in (OR 5.38, P = .015) in patients with brain metastases.
Dr. Klosko noted that the main limitations for this study were that it was a single center retrospective analysis, and that patients were reviewed within the first 7 days of ICU admission. “This potentially missed patients who developed delirium outside of this time frame,” she said. In addition, “too few patients received high-dose benzodiazepines,” and “none of the patients received continuous neuromuscular blockade.”
However, in “contrast to these limitations, this is the largest study to date that has analyzed delirium in this population,” Dr. Klosko said.
Commenting on the study, Brenda Pun, DNP, RN, director of data quality at the Vanderbilt Critical Illness, Brain Dysfunction, and Survivorship Center, Nashville, Tenn., pointed out that the goal of this study was to describe delirium in this specific population. “But I will take a step backward and say that they are just confirming that these patients look like other ICU patients in many regards,” she said.
She explained that the sicker patients are, the higher the rates of delirium. “We have implemented strategies to lower these rates, and they have improved,” Dr. Pun said. “Ten years ago, I would say that 80% of patients who were on a ventilator would have delirium but now the rates are around 50% and that’s what we are typically seeing now.”
Dr. Pun emphasized that this study shows that delirium is like the “canary in the coal mine” or a red flag. “It’s a sign that something is wrong and that we need to pay attention, because the patient’s outcome may be worse,” she said. “So this is saying that we need to see if there is something that can be changed or modified to decrease the incidence of delirium—these are important questions.”
There was no outside sponsor. The authors had no disclosures. Dr. Pun has no disclosures.
FROM CCC50
PCI and CABG for left main disease have equal outcomes at 5 years
Background: While PCI with drug-eluting stents has become more accepted as treatment for some patients with left main disease, long-term outcomes from randomized control trials comparing PCI with CABG have yet to be clearly established.
Study design: International, open-label, multicenter, randomized trial.
Setting: A total of 126 sites in 17 countries.
Synopsis: Patients with low or intermediate anatomical complexity with 70% visual stenosis of the left main coronary artery or 50%-70% stenosis by noninvasive testing were randomized to either PCI (948) or CABG (957). Dual-antiplatelet therapy was given to PCI patients and aspirin to CABG patients. At 5 years there was no significant difference in the rate of the composite of death, stroke, or myocardial infarction (22.0% with PCI vs. 19.2% with CABG; difference, 2.8 percentage points; 95% CI, –0.9 to 6.5; P = .13). This was consistent across subgroups.
There were numerical differences in nonpowered secondary outcomes that may represent effects but should be interpreted cautiously: ischemia-driven revascularization (16.9% with PCI vs. 10% with CABG), transient ischemic attack plus stroke (3.3% with PCI vs. 5.2% with CABG), and death from any cause (3% with PCI vs. 9.9% with CABG). There was no significant difference in cardiovascular events, MI, or stroke.
One interesting limitation was that patients who had PCI were more commonly on dual-antiplatelet therapy and angiotensin converting–enzyme inhibitors, whereas CABG patients were more often on beta-blockers, diuretics, anticoagulants, and antiarrhythmics.
Bottom line: PCI and CABG treatments for left main disease have no significant difference in the composite outcome of death, stroke, or MI at 5 years.
Citation: Stone GW et al. Five-year outcomes after PCI or CABG for left main coronary disease. N Engl J Med. 2019;381:1820-30.
Dr. Horton is a hospitalist and clinical instructor of medicine at the University of Utah, Salt Lake City.
Background: While PCI with drug-eluting stents has become more accepted as treatment for some patients with left main disease, long-term outcomes from randomized control trials comparing PCI with CABG have yet to be clearly established.
Study design: International, open-label, multicenter, randomized trial.
Setting: A total of 126 sites in 17 countries.
Synopsis: Patients with low or intermediate anatomical complexity with 70% visual stenosis of the left main coronary artery or 50%-70% stenosis by noninvasive testing were randomized to either PCI (948) or CABG (957). Dual-antiplatelet therapy was given to PCI patients and aspirin to CABG patients. At 5 years there was no significant difference in the rate of the composite of death, stroke, or myocardial infarction (22.0% with PCI vs. 19.2% with CABG; difference, 2.8 percentage points; 95% CI, –0.9 to 6.5; P = .13). This was consistent across subgroups.
There were numerical differences in nonpowered secondary outcomes that may represent effects but should be interpreted cautiously: ischemia-driven revascularization (16.9% with PCI vs. 10% with CABG), transient ischemic attack plus stroke (3.3% with PCI vs. 5.2% with CABG), and death from any cause (3% with PCI vs. 9.9% with CABG). There was no significant difference in cardiovascular events, MI, or stroke.
One interesting limitation was that patients who had PCI were more commonly on dual-antiplatelet therapy and angiotensin converting–enzyme inhibitors, whereas CABG patients were more often on beta-blockers, diuretics, anticoagulants, and antiarrhythmics.
Bottom line: PCI and CABG treatments for left main disease have no significant difference in the composite outcome of death, stroke, or MI at 5 years.
Citation: Stone GW et al. Five-year outcomes after PCI or CABG for left main coronary disease. N Engl J Med. 2019;381:1820-30.
Dr. Horton is a hospitalist and clinical instructor of medicine at the University of Utah, Salt Lake City.
Background: While PCI with drug-eluting stents has become more accepted as treatment for some patients with left main disease, long-term outcomes from randomized control trials comparing PCI with CABG have yet to be clearly established.
Study design: International, open-label, multicenter, randomized trial.
Setting: A total of 126 sites in 17 countries.
Synopsis: Patients with low or intermediate anatomical complexity with 70% visual stenosis of the left main coronary artery or 50%-70% stenosis by noninvasive testing were randomized to either PCI (948) or CABG (957). Dual-antiplatelet therapy was given to PCI patients and aspirin to CABG patients. At 5 years there was no significant difference in the rate of the composite of death, stroke, or myocardial infarction (22.0% with PCI vs. 19.2% with CABG; difference, 2.8 percentage points; 95% CI, –0.9 to 6.5; P = .13). This was consistent across subgroups.
There were numerical differences in nonpowered secondary outcomes that may represent effects but should be interpreted cautiously: ischemia-driven revascularization (16.9% with PCI vs. 10% with CABG), transient ischemic attack plus stroke (3.3% with PCI vs. 5.2% with CABG), and death from any cause (3% with PCI vs. 9.9% with CABG). There was no significant difference in cardiovascular events, MI, or stroke.
One interesting limitation was that patients who had PCI were more commonly on dual-antiplatelet therapy and angiotensin converting–enzyme inhibitors, whereas CABG patients were more often on beta-blockers, diuretics, anticoagulants, and antiarrhythmics.
Bottom line: PCI and CABG treatments for left main disease have no significant difference in the composite outcome of death, stroke, or MI at 5 years.
Citation: Stone GW et al. Five-year outcomes after PCI or CABG for left main coronary disease. N Engl J Med. 2019;381:1820-30.
Dr. Horton is a hospitalist and clinical instructor of medicine at the University of Utah, Salt Lake City.
Vaccine mismatch: What to do after dose 1 when plans change
Ideally, Americans receiving their Pfizer/BioNTech or Moderna COVID-19 vaccines will get both doses from the same manufacturer, said Gregory Poland, MD, a vaccinologist at the Mayo Clinic in Rochester, Minn.
After all, that’s how they were tested for efficacy and safety, and it was results from those studies that led to emergency use authorization (EUA) being granted by the Food and Drug Administration.
But states and countries have struggled to keep up with the demand for vaccine, and more flexible vaccination schedules could help.
So researchers are exploring whether it is safe and effective to get the first and second doses from different manufacturers. And they are even wondering whether mixing doses from different manufacturers could increase effectiveness, particularly in light of emerging variants.
It’s called the “interchangeability issue,” said Dr. Poland, who has gotten a steady stream of questions about it.
For example, a patient recently asked about options for his father, who had gotten his first dose of the AstraZeneca vaccine in Ecuador, but had since moved to the United States, where that product has not been approved for use.
Dr. Poland said in an interview that he prefaces each answer with: “I’ve got no science for what I’m about to tell you.”
In this particular case, he recommended that the man’s father talk with his doctor about his level of COVID-19 risk and consider whether he should gamble on the AstraZeneca vaccine getting approved in the United States soon, or whether he should ask for a second dose from one of the three vaccines currently approved.
On March 22, 2021, AstraZeneca released positive results from its phase 3 trial, which will likely speed its path toward use in the United States.
Although clinical trials have started to test combinations and boosters, there’s currently no definitive evidence from human trials on mixing COVID vaccines, Dr. Poland pointed out.
But a study of a mixed-vaccine regimen is currently underway in the United Kingdom.
Participants in that 13-month trial will be given the Oxford/AstraZeneca and Pfizer/BioNTech vaccines in different combinations and at different intervals. The first results from that trial are expected this summer.
And interim results from a trial combining Russia’s Sputnik V and the AstraZeneca vaccines are expected in 2 months, according to a Reuters report.
Mix only in ‘exceptional situations’
The Centers for Disease Control and Prevention has been hesitant to open the door to mixing Pfizer and Moderna vaccinations, noting that the two “are not interchangeable.” But CDC guidance has changed slightly. Now, instead of saying the two vaccines should not be mixed, CDC guidance says they can be mixed in “exceptional situations,” and that the second dose can be administered up to 6 weeks after the first dose.
It is reasonable to assume that mixing COVID-19 vaccines that use the same platform – such as the mRNA platform used by both the Pfizer and Moderna vaccines – will be acceptable, Dr. Poland said, although human trials have not proven that.
However, it is unclear whether vaccines that use different platforms can be mixed. Can the first dose of an mRNA vaccine be followed by an adenovirus-based vaccine, like the Johnson & Johnson product or Novavax, if that vaccine is granted an EUA?
Ross Kedl, PhD, a vaccine researcher and professor of immunology at the University of Colorado at Denver, Aurora, said matching vaccine platforms might not be the preferred vaccination strategy.
He disagreed that there’s a lack of science surrounding the issue, and said all signs point to mixing as not only a good option, but probably a better one.
Researcher says science backs mixing
A mix of two different vaccine platforms likely enhances immunity, Dr. Kedl said. The heterologous prime-boost strategy has been used in animal studies for decades, “and it is well known that this promotes a much better immune response than when immunizing with the same vaccine twice.
“If you think about it in a Venn diagram sort of way, it makes sense,” he said in an interview. “Each vaccine has a number of components in it that influence immunity in various ways, but between the two of them, they only have one component that is similar. In the case of the coronavirus vaccines, the one thing both have in common is the spike protein from SARS-CoV-2. In essence, this gives you two shots at generating immunity against the one thing in each vaccine you care most about, but only one shot for the other vaccine components in each platform, resulting in an amplified response against the common target.”
In fact, the heterologous prime-boost vaccination strategy has proven to be effective in humans in early studies.
For example, an Ebola regimen that consisted of an adenovirus vector, similar to the AstraZeneca COVID vaccine, and a modified vaccinia virus vector showed promise in a phase 1 study. And an HIV regimen that consisted of the combination of a DNA vaccine, similar to the Pfizer and Moderna mRNA vaccines, and another viral vector showed encouraging results in a proof-of-concept study.
In both these cases, the heterologous prime-boost strategy was far better than single-vaccine prime-boost regimens, Dr. Kedl pointed out. And neither study reported any safety issues with the combinations.
For now, it’s best to stick with the same manufacturer for both shots, as the CDC guidance suggests, he said, agreeing with Dr. Poland.
But “I would be very surprised if we didn’t move to a mixing of vaccine platforms for the population,” Dr. Kedl said.
A version of this article first appeared on Medscape.com.
Ideally, Americans receiving their Pfizer/BioNTech or Moderna COVID-19 vaccines will get both doses from the same manufacturer, said Gregory Poland, MD, a vaccinologist at the Mayo Clinic in Rochester, Minn.
After all, that’s how they were tested for efficacy and safety, and it was results from those studies that led to emergency use authorization (EUA) being granted by the Food and Drug Administration.
But states and countries have struggled to keep up with the demand for vaccine, and more flexible vaccination schedules could help.
So researchers are exploring whether it is safe and effective to get the first and second doses from different manufacturers. And they are even wondering whether mixing doses from different manufacturers could increase effectiveness, particularly in light of emerging variants.
It’s called the “interchangeability issue,” said Dr. Poland, who has gotten a steady stream of questions about it.
For example, a patient recently asked about options for his father, who had gotten his first dose of the AstraZeneca vaccine in Ecuador, but had since moved to the United States, where that product has not been approved for use.
Dr. Poland said in an interview that he prefaces each answer with: “I’ve got no science for what I’m about to tell you.”
In this particular case, he recommended that the man’s father talk with his doctor about his level of COVID-19 risk and consider whether he should gamble on the AstraZeneca vaccine getting approved in the United States soon, or whether he should ask for a second dose from one of the three vaccines currently approved.
On March 22, 2021, AstraZeneca released positive results from its phase 3 trial, which will likely speed its path toward use in the United States.
Although clinical trials have started to test combinations and boosters, there’s currently no definitive evidence from human trials on mixing COVID vaccines, Dr. Poland pointed out.
But a study of a mixed-vaccine regimen is currently underway in the United Kingdom.
Participants in that 13-month trial will be given the Oxford/AstraZeneca and Pfizer/BioNTech vaccines in different combinations and at different intervals. The first results from that trial are expected this summer.
And interim results from a trial combining Russia’s Sputnik V and the AstraZeneca vaccines are expected in 2 months, according to a Reuters report.
Mix only in ‘exceptional situations’
The Centers for Disease Control and Prevention has been hesitant to open the door to mixing Pfizer and Moderna vaccinations, noting that the two “are not interchangeable.” But CDC guidance has changed slightly. Now, instead of saying the two vaccines should not be mixed, CDC guidance says they can be mixed in “exceptional situations,” and that the second dose can be administered up to 6 weeks after the first dose.
It is reasonable to assume that mixing COVID-19 vaccines that use the same platform – such as the mRNA platform used by both the Pfizer and Moderna vaccines – will be acceptable, Dr. Poland said, although human trials have not proven that.
However, it is unclear whether vaccines that use different platforms can be mixed. Can the first dose of an mRNA vaccine be followed by an adenovirus-based vaccine, like the Johnson & Johnson product or Novavax, if that vaccine is granted an EUA?
Ross Kedl, PhD, a vaccine researcher and professor of immunology at the University of Colorado at Denver, Aurora, said matching vaccine platforms might not be the preferred vaccination strategy.
He disagreed that there’s a lack of science surrounding the issue, and said all signs point to mixing as not only a good option, but probably a better one.
Researcher says science backs mixing
A mix of two different vaccine platforms likely enhances immunity, Dr. Kedl said. The heterologous prime-boost strategy has been used in animal studies for decades, “and it is well known that this promotes a much better immune response than when immunizing with the same vaccine twice.
“If you think about it in a Venn diagram sort of way, it makes sense,” he said in an interview. “Each vaccine has a number of components in it that influence immunity in various ways, but between the two of them, they only have one component that is similar. In the case of the coronavirus vaccines, the one thing both have in common is the spike protein from SARS-CoV-2. In essence, this gives you two shots at generating immunity against the one thing in each vaccine you care most about, but only one shot for the other vaccine components in each platform, resulting in an amplified response against the common target.”
In fact, the heterologous prime-boost vaccination strategy has proven to be effective in humans in early studies.
For example, an Ebola regimen that consisted of an adenovirus vector, similar to the AstraZeneca COVID vaccine, and a modified vaccinia virus vector showed promise in a phase 1 study. And an HIV regimen that consisted of the combination of a DNA vaccine, similar to the Pfizer and Moderna mRNA vaccines, and another viral vector showed encouraging results in a proof-of-concept study.
In both these cases, the heterologous prime-boost strategy was far better than single-vaccine prime-boost regimens, Dr. Kedl pointed out. And neither study reported any safety issues with the combinations.
For now, it’s best to stick with the same manufacturer for both shots, as the CDC guidance suggests, he said, agreeing with Dr. Poland.
But “I would be very surprised if we didn’t move to a mixing of vaccine platforms for the population,” Dr. Kedl said.
A version of this article first appeared on Medscape.com.
Ideally, Americans receiving their Pfizer/BioNTech or Moderna COVID-19 vaccines will get both doses from the same manufacturer, said Gregory Poland, MD, a vaccinologist at the Mayo Clinic in Rochester, Minn.
After all, that’s how they were tested for efficacy and safety, and it was results from those studies that led to emergency use authorization (EUA) being granted by the Food and Drug Administration.
But states and countries have struggled to keep up with the demand for vaccine, and more flexible vaccination schedules could help.
So researchers are exploring whether it is safe and effective to get the first and second doses from different manufacturers. And they are even wondering whether mixing doses from different manufacturers could increase effectiveness, particularly in light of emerging variants.
It’s called the “interchangeability issue,” said Dr. Poland, who has gotten a steady stream of questions about it.
For example, a patient recently asked about options for his father, who had gotten his first dose of the AstraZeneca vaccine in Ecuador, but had since moved to the United States, where that product has not been approved for use.
Dr. Poland said in an interview that he prefaces each answer with: “I’ve got no science for what I’m about to tell you.”
In this particular case, he recommended that the man’s father talk with his doctor about his level of COVID-19 risk and consider whether he should gamble on the AstraZeneca vaccine getting approved in the United States soon, or whether he should ask for a second dose from one of the three vaccines currently approved.
On March 22, 2021, AstraZeneca released positive results from its phase 3 trial, which will likely speed its path toward use in the United States.
Although clinical trials have started to test combinations and boosters, there’s currently no definitive evidence from human trials on mixing COVID vaccines, Dr. Poland pointed out.
But a study of a mixed-vaccine regimen is currently underway in the United Kingdom.
Participants in that 13-month trial will be given the Oxford/AstraZeneca and Pfizer/BioNTech vaccines in different combinations and at different intervals. The first results from that trial are expected this summer.
And interim results from a trial combining Russia’s Sputnik V and the AstraZeneca vaccines are expected in 2 months, according to a Reuters report.
Mix only in ‘exceptional situations’
The Centers for Disease Control and Prevention has been hesitant to open the door to mixing Pfizer and Moderna vaccinations, noting that the two “are not interchangeable.” But CDC guidance has changed slightly. Now, instead of saying the two vaccines should not be mixed, CDC guidance says they can be mixed in “exceptional situations,” and that the second dose can be administered up to 6 weeks after the first dose.
It is reasonable to assume that mixing COVID-19 vaccines that use the same platform – such as the mRNA platform used by both the Pfizer and Moderna vaccines – will be acceptable, Dr. Poland said, although human trials have not proven that.
However, it is unclear whether vaccines that use different platforms can be mixed. Can the first dose of an mRNA vaccine be followed by an adenovirus-based vaccine, like the Johnson & Johnson product or Novavax, if that vaccine is granted an EUA?
Ross Kedl, PhD, a vaccine researcher and professor of immunology at the University of Colorado at Denver, Aurora, said matching vaccine platforms might not be the preferred vaccination strategy.
He disagreed that there’s a lack of science surrounding the issue, and said all signs point to mixing as not only a good option, but probably a better one.
Researcher says science backs mixing
A mix of two different vaccine platforms likely enhances immunity, Dr. Kedl said. The heterologous prime-boost strategy has been used in animal studies for decades, “and it is well known that this promotes a much better immune response than when immunizing with the same vaccine twice.
“If you think about it in a Venn diagram sort of way, it makes sense,” he said in an interview. “Each vaccine has a number of components in it that influence immunity in various ways, but between the two of them, they only have one component that is similar. In the case of the coronavirus vaccines, the one thing both have in common is the spike protein from SARS-CoV-2. In essence, this gives you two shots at generating immunity against the one thing in each vaccine you care most about, but only one shot for the other vaccine components in each platform, resulting in an amplified response against the common target.”
In fact, the heterologous prime-boost vaccination strategy has proven to be effective in humans in early studies.
For example, an Ebola regimen that consisted of an adenovirus vector, similar to the AstraZeneca COVID vaccine, and a modified vaccinia virus vector showed promise in a phase 1 study. And an HIV regimen that consisted of the combination of a DNA vaccine, similar to the Pfizer and Moderna mRNA vaccines, and another viral vector showed encouraging results in a proof-of-concept study.
In both these cases, the heterologous prime-boost strategy was far better than single-vaccine prime-boost regimens, Dr. Kedl pointed out. And neither study reported any safety issues with the combinations.
For now, it’s best to stick with the same manufacturer for both shots, as the CDC guidance suggests, he said, agreeing with Dr. Poland.
But “I would be very surprised if we didn’t move to a mixing of vaccine platforms for the population,” Dr. Kedl said.
A version of this article first appeared on Medscape.com.
COVID vaccines could lose their punch within a year, experts say
In a survey of 77 epidemiologists from 28 countries by the People’s Vaccine Alliance, 66.2% predicted that the world has a year or less before variants make current vaccines ineffective. The People’s Vaccine Alliance is a coalition of more than 50 organizations, including the African Alliance, Oxfam, Public Citizen, and UNAIDS (the Joint United Nations Programme on HIV/AIDS).
Almost a third (32.5%) of those surveyed said ineffectiveness would happen in 9 months or less; 18.2% said 6 months or less.
Paul A. Offit, MD, director of the Vaccine Education Center at the Children’s Hospital of Philadelphia, said in an interview that, while it’s hard to say whether vaccines could become ineffective in that time frame, “It’s perfectly reasonable to think it could happen.”
The good news, said Dr. Offit, who was not involved with the survey, is that SARS-CoV-2 mutates slowly, compared with other viruses such as influenza.
“To date,” he said, “the mutations that have occurred are not far enough away from the immunity induced by your natural infection or immunization such that one isn’t protected at least against severe and critical disease.”
That’s the goal of vaccines, he noted: “to keep people from suffering mightily.”
A line may be crossed
“And so far that’s happening, even with the variants,” Dr. Offit said. “That line has not been crossed. But I think we should assume that it might be.”
Dr. Offit said it will be critical to monitor anyone who gets hospitalized who is known to have been infected or fully vaccinated. Then countries need to get really good at sequencing those viruses.
The great majority of those surveyed (88%) said that persistently low vaccine coverage in many countries would make it more likely that vaccine-resistant mutations will appear.
Coverage comparisons between countries are stark.
Many countries haven’t given a single vaccine dose
While rich countries are giving COVID-19 vaccinations at the rate of a person a second, many of the poorest countries have given hardly any vaccines, the People’s Vaccine Alliance says.
Additionally, according to researchers at the Global Health Innovation Center at Duke University, Durham, N.C., high- and upper-middle–income countries, which represent one-fifth of the world’s population, have bought about 6 billion doses. But low- and lower-middle–income countries, which make up four-fifths of the population, have bought only about 2.6 billion, an article in Nature reports.
“You’re only as strong as your weakest country,” Dr. Offit said. “If we haven’t learned that what happens in other countries can [affect the global population], we haven’t been paying attention.”
Gregg Gonsalves, PhD, associate professor of epidemiology at Yale University, New Haven, Conn., one of the academic centers surveyed, didn’t specify a timeline for when vaccines would become ineffective, but said in a press release that the urgency for widespread global vaccination is real.
“Unless we vaccinate the world,” he said, “we leave the playing field open to more and more mutations, which could churn out variants that could evade our current vaccines and require booster shots to deal with them.”
“Dire, but not surprising”
Panagis Galiatsatos, MD, MHS, a pulmonologist at John Hopkins University, Baltimore, whose research focuses on health care disparities, said the survey findings were “dire, but not surprising.”
Johns Hopkins was another of the centers surveyed, but Dr. Galiatsatos wasn’t personally involved with the survey.
COVID-19, Dr. Galiatsatos pointed out, has laid bare disparities, both in who gets the vaccine and who’s involved in trials to develop the vaccines.
“It’s morally concerning and an ethical reckoning,” he said in an interview.
Recognition of the borderless swath of destruction the virus is exacting is critical, he said.
The United States “has to realize this can’t be a U.S.-centric issue,” he said. “We’re going to be back to the beginning if we don’t make sure that every country is doing well. We haven’t seen that level of uniform approach.”
He noted that scientists have always known that viruses mutate, but now the race is on to find the parts of SARS-CoV-2 that don’t mutate as much.
“My suspicion is we’ll probably need boosters instead of a whole different vaccine,” Dr. Galiatsatos said.
Among the strategies sought by the People’s Vaccine Alliance is for all pharmaceutical companies working on COVID-19 vaccines to openly share technology and intellectual property through the World Health Organization COVID-19 Technology Access Pool, to speed production and rollout of vaccines to all countries.
In the survey, 74% said that open sharing of technology and intellectual property could boost global vaccine coverage; 23% said maybe and 3% said it wouldn’t help.
The survey was carried out between Feb. 17 and March 25, 2021. Respondents included epidemiologists, virologists, and infection disease specialists from the following countries: Algeria, Argentina, Australia, Belgium, Bolivia, Canada, Denmark, Ethiopia, France, Guatemala, India, Italy, Kenya, Lebanon, Norway, Philippines, Senegal, Somalia, South Africa, South Sudan, Spain, United Arab Emirates, Uganda, United Kingdom, United States, Vietnam, Zambia, and Zimbabwe.
Dr. Offit and Dr. Galiatsatos reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
In a survey of 77 epidemiologists from 28 countries by the People’s Vaccine Alliance, 66.2% predicted that the world has a year or less before variants make current vaccines ineffective. The People’s Vaccine Alliance is a coalition of more than 50 organizations, including the African Alliance, Oxfam, Public Citizen, and UNAIDS (the Joint United Nations Programme on HIV/AIDS).
Almost a third (32.5%) of those surveyed said ineffectiveness would happen in 9 months or less; 18.2% said 6 months or less.
Paul A. Offit, MD, director of the Vaccine Education Center at the Children’s Hospital of Philadelphia, said in an interview that, while it’s hard to say whether vaccines could become ineffective in that time frame, “It’s perfectly reasonable to think it could happen.”
The good news, said Dr. Offit, who was not involved with the survey, is that SARS-CoV-2 mutates slowly, compared with other viruses such as influenza.
“To date,” he said, “the mutations that have occurred are not far enough away from the immunity induced by your natural infection or immunization such that one isn’t protected at least against severe and critical disease.”
That’s the goal of vaccines, he noted: “to keep people from suffering mightily.”
A line may be crossed
“And so far that’s happening, even with the variants,” Dr. Offit said. “That line has not been crossed. But I think we should assume that it might be.”
Dr. Offit said it will be critical to monitor anyone who gets hospitalized who is known to have been infected or fully vaccinated. Then countries need to get really good at sequencing those viruses.
The great majority of those surveyed (88%) said that persistently low vaccine coverage in many countries would make it more likely that vaccine-resistant mutations will appear.
Coverage comparisons between countries are stark.
Many countries haven’t given a single vaccine dose
While rich countries are giving COVID-19 vaccinations at the rate of a person a second, many of the poorest countries have given hardly any vaccines, the People’s Vaccine Alliance says.
Additionally, according to researchers at the Global Health Innovation Center at Duke University, Durham, N.C., high- and upper-middle–income countries, which represent one-fifth of the world’s population, have bought about 6 billion doses. But low- and lower-middle–income countries, which make up four-fifths of the population, have bought only about 2.6 billion, an article in Nature reports.
“You’re only as strong as your weakest country,” Dr. Offit said. “If we haven’t learned that what happens in other countries can [affect the global population], we haven’t been paying attention.”
Gregg Gonsalves, PhD, associate professor of epidemiology at Yale University, New Haven, Conn., one of the academic centers surveyed, didn’t specify a timeline for when vaccines would become ineffective, but said in a press release that the urgency for widespread global vaccination is real.
“Unless we vaccinate the world,” he said, “we leave the playing field open to more and more mutations, which could churn out variants that could evade our current vaccines and require booster shots to deal with them.”
“Dire, but not surprising”
Panagis Galiatsatos, MD, MHS, a pulmonologist at John Hopkins University, Baltimore, whose research focuses on health care disparities, said the survey findings were “dire, but not surprising.”
Johns Hopkins was another of the centers surveyed, but Dr. Galiatsatos wasn’t personally involved with the survey.
COVID-19, Dr. Galiatsatos pointed out, has laid bare disparities, both in who gets the vaccine and who’s involved in trials to develop the vaccines.
“It’s morally concerning and an ethical reckoning,” he said in an interview.
Recognition of the borderless swath of destruction the virus is exacting is critical, he said.
The United States “has to realize this can’t be a U.S.-centric issue,” he said. “We’re going to be back to the beginning if we don’t make sure that every country is doing well. We haven’t seen that level of uniform approach.”
He noted that scientists have always known that viruses mutate, but now the race is on to find the parts of SARS-CoV-2 that don’t mutate as much.
“My suspicion is we’ll probably need boosters instead of a whole different vaccine,” Dr. Galiatsatos said.
Among the strategies sought by the People’s Vaccine Alliance is for all pharmaceutical companies working on COVID-19 vaccines to openly share technology and intellectual property through the World Health Organization COVID-19 Technology Access Pool, to speed production and rollout of vaccines to all countries.
In the survey, 74% said that open sharing of technology and intellectual property could boost global vaccine coverage; 23% said maybe and 3% said it wouldn’t help.
The survey was carried out between Feb. 17 and March 25, 2021. Respondents included epidemiologists, virologists, and infection disease specialists from the following countries: Algeria, Argentina, Australia, Belgium, Bolivia, Canada, Denmark, Ethiopia, France, Guatemala, India, Italy, Kenya, Lebanon, Norway, Philippines, Senegal, Somalia, South Africa, South Sudan, Spain, United Arab Emirates, Uganda, United Kingdom, United States, Vietnam, Zambia, and Zimbabwe.
Dr. Offit and Dr. Galiatsatos reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
In a survey of 77 epidemiologists from 28 countries by the People’s Vaccine Alliance, 66.2% predicted that the world has a year or less before variants make current vaccines ineffective. The People’s Vaccine Alliance is a coalition of more than 50 organizations, including the African Alliance, Oxfam, Public Citizen, and UNAIDS (the Joint United Nations Programme on HIV/AIDS).
Almost a third (32.5%) of those surveyed said ineffectiveness would happen in 9 months or less; 18.2% said 6 months or less.
Paul A. Offit, MD, director of the Vaccine Education Center at the Children’s Hospital of Philadelphia, said in an interview that, while it’s hard to say whether vaccines could become ineffective in that time frame, “It’s perfectly reasonable to think it could happen.”
The good news, said Dr. Offit, who was not involved with the survey, is that SARS-CoV-2 mutates slowly, compared with other viruses such as influenza.
“To date,” he said, “the mutations that have occurred are not far enough away from the immunity induced by your natural infection or immunization such that one isn’t protected at least against severe and critical disease.”
That’s the goal of vaccines, he noted: “to keep people from suffering mightily.”
A line may be crossed
“And so far that’s happening, even with the variants,” Dr. Offit said. “That line has not been crossed. But I think we should assume that it might be.”
Dr. Offit said it will be critical to monitor anyone who gets hospitalized who is known to have been infected or fully vaccinated. Then countries need to get really good at sequencing those viruses.
The great majority of those surveyed (88%) said that persistently low vaccine coverage in many countries would make it more likely that vaccine-resistant mutations will appear.
Coverage comparisons between countries are stark.
Many countries haven’t given a single vaccine dose
While rich countries are giving COVID-19 vaccinations at the rate of a person a second, many of the poorest countries have given hardly any vaccines, the People’s Vaccine Alliance says.
Additionally, according to researchers at the Global Health Innovation Center at Duke University, Durham, N.C., high- and upper-middle–income countries, which represent one-fifth of the world’s population, have bought about 6 billion doses. But low- and lower-middle–income countries, which make up four-fifths of the population, have bought only about 2.6 billion, an article in Nature reports.
“You’re only as strong as your weakest country,” Dr. Offit said. “If we haven’t learned that what happens in other countries can [affect the global population], we haven’t been paying attention.”
Gregg Gonsalves, PhD, associate professor of epidemiology at Yale University, New Haven, Conn., one of the academic centers surveyed, didn’t specify a timeline for when vaccines would become ineffective, but said in a press release that the urgency for widespread global vaccination is real.
“Unless we vaccinate the world,” he said, “we leave the playing field open to more and more mutations, which could churn out variants that could evade our current vaccines and require booster shots to deal with them.”
“Dire, but not surprising”
Panagis Galiatsatos, MD, MHS, a pulmonologist at John Hopkins University, Baltimore, whose research focuses on health care disparities, said the survey findings were “dire, but not surprising.”
Johns Hopkins was another of the centers surveyed, but Dr. Galiatsatos wasn’t personally involved with the survey.
COVID-19, Dr. Galiatsatos pointed out, has laid bare disparities, both in who gets the vaccine and who’s involved in trials to develop the vaccines.
“It’s morally concerning and an ethical reckoning,” he said in an interview.
Recognition of the borderless swath of destruction the virus is exacting is critical, he said.
The United States “has to realize this can’t be a U.S.-centric issue,” he said. “We’re going to be back to the beginning if we don’t make sure that every country is doing well. We haven’t seen that level of uniform approach.”
He noted that scientists have always known that viruses mutate, but now the race is on to find the parts of SARS-CoV-2 that don’t mutate as much.
“My suspicion is we’ll probably need boosters instead of a whole different vaccine,” Dr. Galiatsatos said.
Among the strategies sought by the People’s Vaccine Alliance is for all pharmaceutical companies working on COVID-19 vaccines to openly share technology and intellectual property through the World Health Organization COVID-19 Technology Access Pool, to speed production and rollout of vaccines to all countries.
In the survey, 74% said that open sharing of technology and intellectual property could boost global vaccine coverage; 23% said maybe and 3% said it wouldn’t help.
The survey was carried out between Feb. 17 and March 25, 2021. Respondents included epidemiologists, virologists, and infection disease specialists from the following countries: Algeria, Argentina, Australia, Belgium, Bolivia, Canada, Denmark, Ethiopia, France, Guatemala, India, Italy, Kenya, Lebanon, Norway, Philippines, Senegal, Somalia, South Africa, South Sudan, Spain, United Arab Emirates, Uganda, United Kingdom, United States, Vietnam, Zambia, and Zimbabwe.
Dr. Offit and Dr. Galiatsatos reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
New guidelines on the diagnosis and treatment of adults with CAP
Background: More than a decade has passed since the last CAP guidelines. Since then there have been new trials and epidemiological studies. There have also been changes to the process for guideline development. This guideline has moved away from the narrative style of guidelines to the GRADE format and PICO framework with hopes of answering specific questions by looking at the quality of evidence.
Study design: Multidisciplinary panel conducted pragmatic systemic reviews of high-quality studies.
Setting: The panel revised and built upon the 2007 guidelines, addressing 16 clinical questions to be used in immunocompetent patients with radiographic evidence of CAP in the United States with no recent foreign travel.
Synopsis: Changes from the 2007 guidelines are as follows: Sputum and blood cultures, previously recommended only in patients with severe CAP, are now also recommended for inpatients being empirically treated for Pseudomonas or methicillin-resistant Staphylococcus aureus (MRSA) and for those who have received IV antibiotics in the previous 90 days; use of procalcitonin is not recommended to decide whether to withhold antibiotics; steroids are not recommended unless being used for shock; HCAP categorization should be abandoned and need for empiric coverage of MRSA and Pseudomonas should be based on local epidemiology and local validated risk factors; B-lactam/macrolide is favored over fluoroquinolone for severe CAP therapy; and routine follow-up chest x-ray is not recommended.
Other recommendations include not routinely testing for urine pneumococcal or legionella antigens in nonsevere CAP; using PSI over CURB-65, in addition to clinical judgment, to determine need for inpatient care; using severe CAP criteria and clinical judgment for determining ICU need; not adding anaerobic coverage for aspiration pneumonia; and treating most cases of CAP that are clinically stable and uncomplicated for 5-7 days.
Bottom line: Given new data, updated recommendations have been made to help optimize CAP therapy.
Citation: Metlay JP et al. Diagnosis and treatment of adults with community-acquired pneumonia: An official clinical practice guideline of the American Thoracic Society and Infectious Diseases Society of America. Am J Respir Crit Care Med. 2019 Oct 1;200(7):e45-67.
Dr. Horton is a hospitalist and clinical instructor of medicine at the University of Utah, Salt Lake City.
Background: More than a decade has passed since the last CAP guidelines. Since then there have been new trials and epidemiological studies. There have also been changes to the process for guideline development. This guideline has moved away from the narrative style of guidelines to the GRADE format and PICO framework with hopes of answering specific questions by looking at the quality of evidence.
Study design: Multidisciplinary panel conducted pragmatic systemic reviews of high-quality studies.
Setting: The panel revised and built upon the 2007 guidelines, addressing 16 clinical questions to be used in immunocompetent patients with radiographic evidence of CAP in the United States with no recent foreign travel.
Synopsis: Changes from the 2007 guidelines are as follows: Sputum and blood cultures, previously recommended only in patients with severe CAP, are now also recommended for inpatients being empirically treated for Pseudomonas or methicillin-resistant Staphylococcus aureus (MRSA) and for those who have received IV antibiotics in the previous 90 days; use of procalcitonin is not recommended to decide whether to withhold antibiotics; steroids are not recommended unless being used for shock; HCAP categorization should be abandoned and need for empiric coverage of MRSA and Pseudomonas should be based on local epidemiology and local validated risk factors; B-lactam/macrolide is favored over fluoroquinolone for severe CAP therapy; and routine follow-up chest x-ray is not recommended.
Other recommendations include not routinely testing for urine pneumococcal or legionella antigens in nonsevere CAP; using PSI over CURB-65, in addition to clinical judgment, to determine need for inpatient care; using severe CAP criteria and clinical judgment for determining ICU need; not adding anaerobic coverage for aspiration pneumonia; and treating most cases of CAP that are clinically stable and uncomplicated for 5-7 days.
Bottom line: Given new data, updated recommendations have been made to help optimize CAP therapy.
Citation: Metlay JP et al. Diagnosis and treatment of adults with community-acquired pneumonia: An official clinical practice guideline of the American Thoracic Society and Infectious Diseases Society of America. Am J Respir Crit Care Med. 2019 Oct 1;200(7):e45-67.
Dr. Horton is a hospitalist and clinical instructor of medicine at the University of Utah, Salt Lake City.
Background: More than a decade has passed since the last CAP guidelines. Since then there have been new trials and epidemiological studies. There have also been changes to the process for guideline development. This guideline has moved away from the narrative style of guidelines to the GRADE format and PICO framework with hopes of answering specific questions by looking at the quality of evidence.
Study design: Multidisciplinary panel conducted pragmatic systemic reviews of high-quality studies.
Setting: The panel revised and built upon the 2007 guidelines, addressing 16 clinical questions to be used in immunocompetent patients with radiographic evidence of CAP in the United States with no recent foreign travel.
Synopsis: Changes from the 2007 guidelines are as follows: Sputum and blood cultures, previously recommended only in patients with severe CAP, are now also recommended for inpatients being empirically treated for Pseudomonas or methicillin-resistant Staphylococcus aureus (MRSA) and for those who have received IV antibiotics in the previous 90 days; use of procalcitonin is not recommended to decide whether to withhold antibiotics; steroids are not recommended unless being used for shock; HCAP categorization should be abandoned and need for empiric coverage of MRSA and Pseudomonas should be based on local epidemiology and local validated risk factors; B-lactam/macrolide is favored over fluoroquinolone for severe CAP therapy; and routine follow-up chest x-ray is not recommended.
Other recommendations include not routinely testing for urine pneumococcal or legionella antigens in nonsevere CAP; using PSI over CURB-65, in addition to clinical judgment, to determine need for inpatient care; using severe CAP criteria and clinical judgment for determining ICU need; not adding anaerobic coverage for aspiration pneumonia; and treating most cases of CAP that are clinically stable and uncomplicated for 5-7 days.
Bottom line: Given new data, updated recommendations have been made to help optimize CAP therapy.
Citation: Metlay JP et al. Diagnosis and treatment of adults with community-acquired pneumonia: An official clinical practice guideline of the American Thoracic Society and Infectious Diseases Society of America. Am J Respir Crit Care Med. 2019 Oct 1;200(7):e45-67.
Dr. Horton is a hospitalist and clinical instructor of medicine at the University of Utah, Salt Lake City.