User login
New pediatric cases down as U.S. tops 2 million children with COVID-19
The United States exceeded 2 million reported cases of COVID-19 in children just 6 weeks after recording its 1 millionth case, according to a report from the American Academy of Pediatrics and the Children’s Hospital Association.
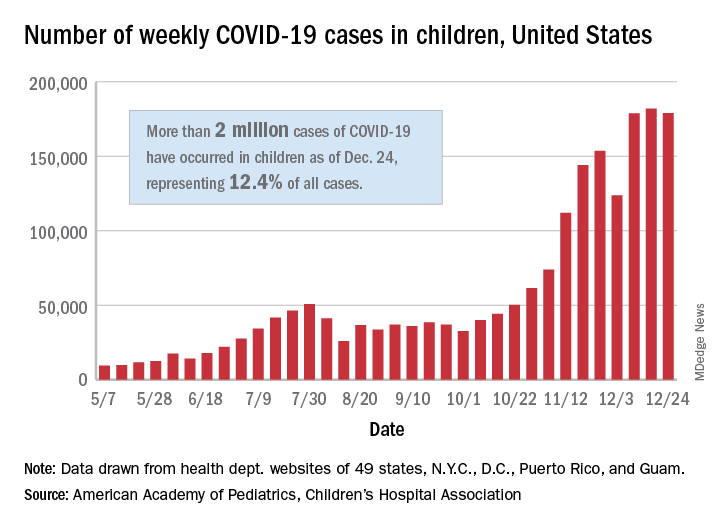
The total number of cases in children was 2,000,681 as of Dec. 24, which represents 12.4% of all cases reported by the health departments of 49 states (excluding New York), the District of Columbia, New York City, Puerto Rico, and Guam, the AAP and CHA stated Dec. 29.
The case count for just the latest week, 178,935, was actually down 1.7% from the 182,018 reported the week before, marking the second drop since the beginning of December. The first came during the week ending Dec. 3, when the number of cases dropped more than 19% from the previous week, based on data from the AAP/CHA report.
The cumulative national rate of coronavirus infection is now 2,658 cases per 100,000 children, and “13 states have reported more than 4,000 cases per 100,000,” the two groups said.
The highest rate for any state can be found in North Dakota, which has had 7,722 cases of COVID-19 per 100,000 children. Wyoming has the highest proportion of cases in children at 20.5%, and California has reported the most cases overall, 234,174, the report shows.
Data on testing, hospitalization, and mortality were not included in the Dec. 29 report because of the holiday but will be available in the next edition, scheduled for release on Jan. 5, 2021.
The United States exceeded 2 million reported cases of COVID-19 in children just 6 weeks after recording its 1 millionth case, according to a report from the American Academy of Pediatrics and the Children’s Hospital Association.

The total number of cases in children was 2,000,681 as of Dec. 24, which represents 12.4% of all cases reported by the health departments of 49 states (excluding New York), the District of Columbia, New York City, Puerto Rico, and Guam, the AAP and CHA stated Dec. 29.
The case count for just the latest week, 178,935, was actually down 1.7% from the 182,018 reported the week before, marking the second drop since the beginning of December. The first came during the week ending Dec. 3, when the number of cases dropped more than 19% from the previous week, based on data from the AAP/CHA report.
The cumulative national rate of coronavirus infection is now 2,658 cases per 100,000 children, and “13 states have reported more than 4,000 cases per 100,000,” the two groups said.
The highest rate for any state can be found in North Dakota, which has had 7,722 cases of COVID-19 per 100,000 children. Wyoming has the highest proportion of cases in children at 20.5%, and California has reported the most cases overall, 234,174, the report shows.
Data on testing, hospitalization, and mortality were not included in the Dec. 29 report because of the holiday but will be available in the next edition, scheduled for release on Jan. 5, 2021.
The United States exceeded 2 million reported cases of COVID-19 in children just 6 weeks after recording its 1 millionth case, according to a report from the American Academy of Pediatrics and the Children’s Hospital Association.

The total number of cases in children was 2,000,681 as of Dec. 24, which represents 12.4% of all cases reported by the health departments of 49 states (excluding New York), the District of Columbia, New York City, Puerto Rico, and Guam, the AAP and CHA stated Dec. 29.
The case count for just the latest week, 178,935, was actually down 1.7% from the 182,018 reported the week before, marking the second drop since the beginning of December. The first came during the week ending Dec. 3, when the number of cases dropped more than 19% from the previous week, based on data from the AAP/CHA report.
The cumulative national rate of coronavirus infection is now 2,658 cases per 100,000 children, and “13 states have reported more than 4,000 cases per 100,000,” the two groups said.
The highest rate for any state can be found in North Dakota, which has had 7,722 cases of COVID-19 per 100,000 children. Wyoming has the highest proportion of cases in children at 20.5%, and California has reported the most cases overall, 234,174, the report shows.
Data on testing, hospitalization, and mortality were not included in the Dec. 29 report because of the holiday but will be available in the next edition, scheduled for release on Jan. 5, 2021.
2.1 Million COVID Vaccine Doses Given in U.S.
The U.S. has distributed more than 11.4 million doses of the Pfizer and Moderna COVID-19 vaccines, and more than 2.1 million of those had been given to people as of December 28, according to the CDC.
The CDC’s COVID Data Tracker showed the updated numbers as of 9 a.m. on that day. The distribution total is based on the CDC’s Vaccine Tracking System, and the administered total is based on reports from state and local public health departments, as well as updates from five federal agencies: the Bureau of Prisons, Veterans Administration, Department of Defense, Department of State, and Indian Health Services.
Health care providers report to public health agencies up to 72 hours after the vaccine is given, and public health agencies report to the CDC after that, so there may be a lag in the data. The CDC’s numbers will be updated on Mondays, Wednesdays, and Fridays.
“A large difference between the number of doses distributed and the number of doses administered is expected at this point in the COVID vaccination program due to several factors,” the CDC says.
Delays could occur due to the reporting of doses given, how states and local vaccine sites are managing vaccines, and the pending launch of vaccination through the federal Pharmacy Partnership for Long-Term Care Program.
“Numbers reported on other websites may differ from what is posted on CDC’s website because CDC’s overall numbers are validated through a data submission process with each jurisdiction,” the CDC says.
On Dec. 26, the agency’s tally showed that 9.5 million doses had been distributed and 1.9 million had been given, according to Reuters.
Public health officials and health care workers have begun to voice their concerns about the delay in giving the vaccines.
“We certainly are not at the numbers that we wanted to be at the end of December,” Anthony Fauci, MD, director of the National Institute of Allergy and Infectious Diseases, told CNNDec. 29.
Operation Warp Speed had planned for 20 million people to be vaccinated by the end of the year. Fauci said he hopes that number will be achieved next month.
“I believe that as we get into January, we are going to see an increase in the momentum,” he said.
Shipment delays have affected other priority groups as well. The New York Police Department anticipated a rollout Dec. 29, but it’s now been delayed since the department hasn’t received enough Moderna doses to start giving the shots, according to the New York Daily News.
“We’ve made numerous attempts to get updated information, and when we get further word on its availability, we will immediately keep our members appraised of the new date and the method of distribution,” Paul DiGiacomo, president of the Detectives’ Endowment Association, wrote in a memo to members on Dec. 28.
“Every detective squad has been crushed with [COVID-19],” he told the newspaper. “Within the last couple of weeks, we’ve had at least two detectives hospitalized.”
President-elect Joe Biden will receive a briefing from his COVID-19 advisory team, provide a general update on the pandemic, and describe his own plan for vaccinating people quickly during an address Dec. 29, a transition official told Axios. Biden has pledged to administer 100 million vaccine doses in his first 100 days in office.
A version of this article originally appeared on WebMd.
The U.S. has distributed more than 11.4 million doses of the Pfizer and Moderna COVID-19 vaccines, and more than 2.1 million of those had been given to people as of December 28, according to the CDC.
The CDC’s COVID Data Tracker showed the updated numbers as of 9 a.m. on that day. The distribution total is based on the CDC’s Vaccine Tracking System, and the administered total is based on reports from state and local public health departments, as well as updates from five federal agencies: the Bureau of Prisons, Veterans Administration, Department of Defense, Department of State, and Indian Health Services.
Health care providers report to public health agencies up to 72 hours after the vaccine is given, and public health agencies report to the CDC after that, so there may be a lag in the data. The CDC’s numbers will be updated on Mondays, Wednesdays, and Fridays.
“A large difference between the number of doses distributed and the number of doses administered is expected at this point in the COVID vaccination program due to several factors,” the CDC says.
Delays could occur due to the reporting of doses given, how states and local vaccine sites are managing vaccines, and the pending launch of vaccination through the federal Pharmacy Partnership for Long-Term Care Program.
“Numbers reported on other websites may differ from what is posted on CDC’s website because CDC’s overall numbers are validated through a data submission process with each jurisdiction,” the CDC says.
On Dec. 26, the agency’s tally showed that 9.5 million doses had been distributed and 1.9 million had been given, according to Reuters.
Public health officials and health care workers have begun to voice their concerns about the delay in giving the vaccines.
“We certainly are not at the numbers that we wanted to be at the end of December,” Anthony Fauci, MD, director of the National Institute of Allergy and Infectious Diseases, told CNNDec. 29.
Operation Warp Speed had planned for 20 million people to be vaccinated by the end of the year. Fauci said he hopes that number will be achieved next month.
“I believe that as we get into January, we are going to see an increase in the momentum,” he said.
Shipment delays have affected other priority groups as well. The New York Police Department anticipated a rollout Dec. 29, but it’s now been delayed since the department hasn’t received enough Moderna doses to start giving the shots, according to the New York Daily News.
“We’ve made numerous attempts to get updated information, and when we get further word on its availability, we will immediately keep our members appraised of the new date and the method of distribution,” Paul DiGiacomo, president of the Detectives’ Endowment Association, wrote in a memo to members on Dec. 28.
“Every detective squad has been crushed with [COVID-19],” he told the newspaper. “Within the last couple of weeks, we’ve had at least two detectives hospitalized.”
President-elect Joe Biden will receive a briefing from his COVID-19 advisory team, provide a general update on the pandemic, and describe his own plan for vaccinating people quickly during an address Dec. 29, a transition official told Axios. Biden has pledged to administer 100 million vaccine doses in his first 100 days in office.
A version of this article originally appeared on WebMd.
The U.S. has distributed more than 11.4 million doses of the Pfizer and Moderna COVID-19 vaccines, and more than 2.1 million of those had been given to people as of December 28, according to the CDC.
The CDC’s COVID Data Tracker showed the updated numbers as of 9 a.m. on that day. The distribution total is based on the CDC’s Vaccine Tracking System, and the administered total is based on reports from state and local public health departments, as well as updates from five federal agencies: the Bureau of Prisons, Veterans Administration, Department of Defense, Department of State, and Indian Health Services.
Health care providers report to public health agencies up to 72 hours after the vaccine is given, and public health agencies report to the CDC after that, so there may be a lag in the data. The CDC’s numbers will be updated on Mondays, Wednesdays, and Fridays.
“A large difference between the number of doses distributed and the number of doses administered is expected at this point in the COVID vaccination program due to several factors,” the CDC says.
Delays could occur due to the reporting of doses given, how states and local vaccine sites are managing vaccines, and the pending launch of vaccination through the federal Pharmacy Partnership for Long-Term Care Program.
“Numbers reported on other websites may differ from what is posted on CDC’s website because CDC’s overall numbers are validated through a data submission process with each jurisdiction,” the CDC says.
On Dec. 26, the agency’s tally showed that 9.5 million doses had been distributed and 1.9 million had been given, according to Reuters.
Public health officials and health care workers have begun to voice their concerns about the delay in giving the vaccines.
“We certainly are not at the numbers that we wanted to be at the end of December,” Anthony Fauci, MD, director of the National Institute of Allergy and Infectious Diseases, told CNNDec. 29.
Operation Warp Speed had planned for 20 million people to be vaccinated by the end of the year. Fauci said he hopes that number will be achieved next month.
“I believe that as we get into January, we are going to see an increase in the momentum,” he said.
Shipment delays have affected other priority groups as well. The New York Police Department anticipated a rollout Dec. 29, but it’s now been delayed since the department hasn’t received enough Moderna doses to start giving the shots, according to the New York Daily News.
“We’ve made numerous attempts to get updated information, and when we get further word on its availability, we will immediately keep our members appraised of the new date and the method of distribution,” Paul DiGiacomo, president of the Detectives’ Endowment Association, wrote in a memo to members on Dec. 28.
“Every detective squad has been crushed with [COVID-19],” he told the newspaper. “Within the last couple of weeks, we’ve had at least two detectives hospitalized.”
President-elect Joe Biden will receive a briefing from his COVID-19 advisory team, provide a general update on the pandemic, and describe his own plan for vaccinating people quickly during an address Dec. 29, a transition official told Axios. Biden has pledged to administer 100 million vaccine doses in his first 100 days in office.
A version of this article originally appeared on WebMd.
CDC issues COVID-19 vaccine guidance for underlying conditions
The Centers for Disease Control and Prevention has issued updated guidance for people with underlying medical conditions who are considering getting the coronavirus vaccine.
“Adults of any age with certain underlying medical conditions are at increased risk for severe illness from the virus that causes COVID-19,” the CDC said in the guidance, posted on Dec. 26. “mRNA COVID-19 vaccines may be administered to people with underlying medical conditions provided they have not had a severe allergic reaction to any of the ingredients in the vaccine.”
Both the Pfizer and Moderna vaccines use mRNA, or messenger RNA.
The CDC guidance had specific information for people with HIV, weakened immune systems, and autoimmune conditions such as Guillain-Barré syndrome (GBS) and Bell’s palsy who are thinking of getting the vaccine.
People with HIV and weakened immune systems “may receive a COVID-19 vaccine. However, they should be aware of the limited safety data,” the CDC said.
There’s no information available yet about the safety of the vaccines for people with weakened immune systems. People with HIV were included in clinical trials, but “safety data specific to this group are not yet available at this time,” the CDC said.
Cases of Bell’s palsy, a temporary facial paralysis, were reported in people receiving the Pfizer and Moderna vaccines in clinical trials, the Food and Drug Administration said Dec. 17.
But the new CDC guidance said that the FDA “does not consider these to be above the rate expected in the general population. They have not concluded these cases were caused by vaccination. Therefore, persons who have previously had Bell’s palsy may receive an mRNA COVID-19 vaccine.”
Researchers have determined the vaccines are safe for people with GBS, a rare autoimmune disorder in which the body’s immune system attacks nerves just as they leave the spinal cord, the CDC said.
“To date, no cases of GBS have been reported following vaccination among participants in the mRNA COVID-19 vaccine clinical trials,” the CDC guidance said. “With few exceptions, the independent Advisory Committee on Immunization Practices general best practice guidelines for immunization do not include a history of GBS as a precaution to vaccination with other vaccines.”
For months, the CDC and other health authorities have said that people with certain medical conditions are at an increased risk of developing severe cases of COVID-19.
A version of this article first appeared on Medscape.com.
The Centers for Disease Control and Prevention has issued updated guidance for people with underlying medical conditions who are considering getting the coronavirus vaccine.
“Adults of any age with certain underlying medical conditions are at increased risk for severe illness from the virus that causes COVID-19,” the CDC said in the guidance, posted on Dec. 26. “mRNA COVID-19 vaccines may be administered to people with underlying medical conditions provided they have not had a severe allergic reaction to any of the ingredients in the vaccine.”
Both the Pfizer and Moderna vaccines use mRNA, or messenger RNA.
The CDC guidance had specific information for people with HIV, weakened immune systems, and autoimmune conditions such as Guillain-Barré syndrome (GBS) and Bell’s palsy who are thinking of getting the vaccine.
People with HIV and weakened immune systems “may receive a COVID-19 vaccine. However, they should be aware of the limited safety data,” the CDC said.
There’s no information available yet about the safety of the vaccines for people with weakened immune systems. People with HIV were included in clinical trials, but “safety data specific to this group are not yet available at this time,” the CDC said.
Cases of Bell’s palsy, a temporary facial paralysis, were reported in people receiving the Pfizer and Moderna vaccines in clinical trials, the Food and Drug Administration said Dec. 17.
But the new CDC guidance said that the FDA “does not consider these to be above the rate expected in the general population. They have not concluded these cases were caused by vaccination. Therefore, persons who have previously had Bell’s palsy may receive an mRNA COVID-19 vaccine.”
Researchers have determined the vaccines are safe for people with GBS, a rare autoimmune disorder in which the body’s immune system attacks nerves just as they leave the spinal cord, the CDC said.
“To date, no cases of GBS have been reported following vaccination among participants in the mRNA COVID-19 vaccine clinical trials,” the CDC guidance said. “With few exceptions, the independent Advisory Committee on Immunization Practices general best practice guidelines for immunization do not include a history of GBS as a precaution to vaccination with other vaccines.”
For months, the CDC and other health authorities have said that people with certain medical conditions are at an increased risk of developing severe cases of COVID-19.
A version of this article first appeared on Medscape.com.
The Centers for Disease Control and Prevention has issued updated guidance for people with underlying medical conditions who are considering getting the coronavirus vaccine.
“Adults of any age with certain underlying medical conditions are at increased risk for severe illness from the virus that causes COVID-19,” the CDC said in the guidance, posted on Dec. 26. “mRNA COVID-19 vaccines may be administered to people with underlying medical conditions provided they have not had a severe allergic reaction to any of the ingredients in the vaccine.”
Both the Pfizer and Moderna vaccines use mRNA, or messenger RNA.
The CDC guidance had specific information for people with HIV, weakened immune systems, and autoimmune conditions such as Guillain-Barré syndrome (GBS) and Bell’s palsy who are thinking of getting the vaccine.
People with HIV and weakened immune systems “may receive a COVID-19 vaccine. However, they should be aware of the limited safety data,” the CDC said.
There’s no information available yet about the safety of the vaccines for people with weakened immune systems. People with HIV were included in clinical trials, but “safety data specific to this group are not yet available at this time,” the CDC said.
Cases of Bell’s palsy, a temporary facial paralysis, were reported in people receiving the Pfizer and Moderna vaccines in clinical trials, the Food and Drug Administration said Dec. 17.
But the new CDC guidance said that the FDA “does not consider these to be above the rate expected in the general population. They have not concluded these cases were caused by vaccination. Therefore, persons who have previously had Bell’s palsy may receive an mRNA COVID-19 vaccine.”
Researchers have determined the vaccines are safe for people with GBS, a rare autoimmune disorder in which the body’s immune system attacks nerves just as they leave the spinal cord, the CDC said.
“To date, no cases of GBS have been reported following vaccination among participants in the mRNA COVID-19 vaccine clinical trials,” the CDC guidance said. “With few exceptions, the independent Advisory Committee on Immunization Practices general best practice guidelines for immunization do not include a history of GBS as a precaution to vaccination with other vaccines.”
For months, the CDC and other health authorities have said that people with certain medical conditions are at an increased risk of developing severe cases of COVID-19.
A version of this article first appeared on Medscape.com.
Scant risk for SARS-CoV-2 from hospital air
Everywhere they look within hospitals, researchers find RNA from SARS-CoV-2 in the air. But viable viruses typically are found only close to patients, according to a review of published studies.
The finding supports recommendations to use surgical masks in most parts of the hospital, reserving respirators (such as N95 or FFP2) for aerosol-generating procedures on patients’ respiratory tracts, said Gabriel Birgand, PhD, an infectious disease researcher at Imperial College London.
“When the virus is spreading a lot in the community, it’s probably more likely for you to be contaminated in your friends’ areas or in your building than in your work area, where you are well equipped and compliant with all the measures,” he said in an interview. “So it’s pretty good news.”
The systematic review by Dr. Birgand and colleagues was published in JAMA Network Open.
Recommended precautions to protect health care workers from SARS-CoV-2 infections remain controversial. Most authorities believe droplets are the primary route of transmission, which would mean surgical masks may be sufficient protection. But some research has suggested transmission by aerosols as well, making N95 respirators seem necessary. There is even disagreement about the definitions of the words “aerosol” and “droplet.”
To better understand where traces of the virus can be found in the air in hospitals, Dr. Birgand and colleagues analyzed all the studies they could find on the subject in English.
They identified 24 articles with original data. All of the studies used reverse transcription–polymerase chain reaction (PCR) tests to identify SARS-CoV-2 RNA. In five studies, attempts were also made to culture viable viruses. Three studies assessed the particle size relative to RNA concentration or viral titer.
Of 893 air samples across the 24 studies, 52.7% were taken from areas close to patients, 26.5% were taken in clinical areas, 13.7% in staff areas, 4.7% in public areas, and 2.4% in toilets or bathrooms.
Among those studies that quantified RNA, the median interquartile range of concentrations varied from 1.0 x 103 copies/m3 in clinical areas to 9.7 x 103 copies/m3 in toilets or bathrooms.
One study found an RNA concentration of 2.0 x 103 copies for particle sizes >4 mcm and 1.3 x 103 copies/m3 for particle sizes ≤4 mcm, both in patients’ rooms.
Three studies included viral cultures; of those, two resulted in positive cultures, both in a non-ICU setting. In one study, 3 of 39 samples were positive, and in the other, 4 of 4 were positive. Viral cultures in toilets, clinical areas, staff areas, and public areas were negative.
One of these studies assessed viral concentration and found that the median interquartile range was 4.8 tissue culture infectious dose (TCID50)/m3 for particles <1 mcm, 4.27 TCID50/m3 for particles 1-4 mcm, and 1.82 TCID50/m3 for particles >4 mcm.
Although viable viruses weren’t found in staff areas, the presence of viral RNA in places such as dining rooms and meeting rooms raises a concern, Dr. Birgand said.
“All of these staff areas are probably playing an important role in contamination,” he said. “It’s pretty easy to see when you are dining, you are not wearing a face mask, and it’s associated with a strong risk when there is a strong dissemination of the virus in the community.”
Studies on contact tracing among health care workers have also identified meeting rooms and dining rooms as the second most common source of infection after community contact, he said.
In general, the findings of the review correspond to epidemiologic studies, said Angela Rasmussen, PhD, a virologist with the Georgetown University Center for Global Health Science and Security, Washington, who was not involved in the review. “Absent aerosol-generating procedures, health care workers are largely not getting infected when they take droplet precautions.”
One reason may be that patients shed the most infectious viruses a couple of days before and after symptoms begin. By the time they’re hospitalized, they’re less likely to be contagious but may continue to shed viral RNA.
“We don’t really know the basis for the persistence of RNA being produced long after people have been infected and have recovered from the acute infection,” she said, “but it has been observed quite frequently.”
Although the virus cannot remain viable for very long in the air, remnants may still be detected in the form of RNA, Dr. Rasmussen said. In addition, hospitals often do a good job of ventilation.
She pointed out that it can be difficult to cultivate viruses in air samples because of contaminants such as bacteria and fungi. “That’s one of the limitations of a study like this. You’re not really sure if it’s because there’s no viable virus there or because you just aren’t able to collect samples that would allow you to determine that.”
Dr. Birgand and colleagues acknowledged other limitations. The studies they reviewed used different approaches to sampling. Different procedures may have been underway in the rooms being sampled, and factors such as temperature and humidity could have affected the results. In addition, the studies used different cycle thresholds for PCR positivity.
A version of this article first appeared on Medscape.com.
Everywhere they look within hospitals, researchers find RNA from SARS-CoV-2 in the air. But viable viruses typically are found only close to patients, according to a review of published studies.
The finding supports recommendations to use surgical masks in most parts of the hospital, reserving respirators (such as N95 or FFP2) for aerosol-generating procedures on patients’ respiratory tracts, said Gabriel Birgand, PhD, an infectious disease researcher at Imperial College London.
“When the virus is spreading a lot in the community, it’s probably more likely for you to be contaminated in your friends’ areas or in your building than in your work area, where you are well equipped and compliant with all the measures,” he said in an interview. “So it’s pretty good news.”
The systematic review by Dr. Birgand and colleagues was published in JAMA Network Open.
Recommended precautions to protect health care workers from SARS-CoV-2 infections remain controversial. Most authorities believe droplets are the primary route of transmission, which would mean surgical masks may be sufficient protection. But some research has suggested transmission by aerosols as well, making N95 respirators seem necessary. There is even disagreement about the definitions of the words “aerosol” and “droplet.”
To better understand where traces of the virus can be found in the air in hospitals, Dr. Birgand and colleagues analyzed all the studies they could find on the subject in English.
They identified 24 articles with original data. All of the studies used reverse transcription–polymerase chain reaction (PCR) tests to identify SARS-CoV-2 RNA. In five studies, attempts were also made to culture viable viruses. Three studies assessed the particle size relative to RNA concentration or viral titer.
Of 893 air samples across the 24 studies, 52.7% were taken from areas close to patients, 26.5% were taken in clinical areas, 13.7% in staff areas, 4.7% in public areas, and 2.4% in toilets or bathrooms.
Among those studies that quantified RNA, the median interquartile range of concentrations varied from 1.0 x 103 copies/m3 in clinical areas to 9.7 x 103 copies/m3 in toilets or bathrooms.
One study found an RNA concentration of 2.0 x 103 copies for particle sizes >4 mcm and 1.3 x 103 copies/m3 for particle sizes ≤4 mcm, both in patients’ rooms.
Three studies included viral cultures; of those, two resulted in positive cultures, both in a non-ICU setting. In one study, 3 of 39 samples were positive, and in the other, 4 of 4 were positive. Viral cultures in toilets, clinical areas, staff areas, and public areas were negative.
One of these studies assessed viral concentration and found that the median interquartile range was 4.8 tissue culture infectious dose (TCID50)/m3 for particles <1 mcm, 4.27 TCID50/m3 for particles 1-4 mcm, and 1.82 TCID50/m3 for particles >4 mcm.
Although viable viruses weren’t found in staff areas, the presence of viral RNA in places such as dining rooms and meeting rooms raises a concern, Dr. Birgand said.
“All of these staff areas are probably playing an important role in contamination,” he said. “It’s pretty easy to see when you are dining, you are not wearing a face mask, and it’s associated with a strong risk when there is a strong dissemination of the virus in the community.”
Studies on contact tracing among health care workers have also identified meeting rooms and dining rooms as the second most common source of infection after community contact, he said.
In general, the findings of the review correspond to epidemiologic studies, said Angela Rasmussen, PhD, a virologist with the Georgetown University Center for Global Health Science and Security, Washington, who was not involved in the review. “Absent aerosol-generating procedures, health care workers are largely not getting infected when they take droplet precautions.”
One reason may be that patients shed the most infectious viruses a couple of days before and after symptoms begin. By the time they’re hospitalized, they’re less likely to be contagious but may continue to shed viral RNA.
“We don’t really know the basis for the persistence of RNA being produced long after people have been infected and have recovered from the acute infection,” she said, “but it has been observed quite frequently.”
Although the virus cannot remain viable for very long in the air, remnants may still be detected in the form of RNA, Dr. Rasmussen said. In addition, hospitals often do a good job of ventilation.
She pointed out that it can be difficult to cultivate viruses in air samples because of contaminants such as bacteria and fungi. “That’s one of the limitations of a study like this. You’re not really sure if it’s because there’s no viable virus there or because you just aren’t able to collect samples that would allow you to determine that.”
Dr. Birgand and colleagues acknowledged other limitations. The studies they reviewed used different approaches to sampling. Different procedures may have been underway in the rooms being sampled, and factors such as temperature and humidity could have affected the results. In addition, the studies used different cycle thresholds for PCR positivity.
A version of this article first appeared on Medscape.com.
Everywhere they look within hospitals, researchers find RNA from SARS-CoV-2 in the air. But viable viruses typically are found only close to patients, according to a review of published studies.
The finding supports recommendations to use surgical masks in most parts of the hospital, reserving respirators (such as N95 or FFP2) for aerosol-generating procedures on patients’ respiratory tracts, said Gabriel Birgand, PhD, an infectious disease researcher at Imperial College London.
“When the virus is spreading a lot in the community, it’s probably more likely for you to be contaminated in your friends’ areas or in your building than in your work area, where you are well equipped and compliant with all the measures,” he said in an interview. “So it’s pretty good news.”
The systematic review by Dr. Birgand and colleagues was published in JAMA Network Open.
Recommended precautions to protect health care workers from SARS-CoV-2 infections remain controversial. Most authorities believe droplets are the primary route of transmission, which would mean surgical masks may be sufficient protection. But some research has suggested transmission by aerosols as well, making N95 respirators seem necessary. There is even disagreement about the definitions of the words “aerosol” and “droplet.”
To better understand where traces of the virus can be found in the air in hospitals, Dr. Birgand and colleagues analyzed all the studies they could find on the subject in English.
They identified 24 articles with original data. All of the studies used reverse transcription–polymerase chain reaction (PCR) tests to identify SARS-CoV-2 RNA. In five studies, attempts were also made to culture viable viruses. Three studies assessed the particle size relative to RNA concentration or viral titer.
Of 893 air samples across the 24 studies, 52.7% were taken from areas close to patients, 26.5% were taken in clinical areas, 13.7% in staff areas, 4.7% in public areas, and 2.4% in toilets or bathrooms.
Among those studies that quantified RNA, the median interquartile range of concentrations varied from 1.0 x 103 copies/m3 in clinical areas to 9.7 x 103 copies/m3 in toilets or bathrooms.
One study found an RNA concentration of 2.0 x 103 copies for particle sizes >4 mcm and 1.3 x 103 copies/m3 for particle sizes ≤4 mcm, both in patients’ rooms.
Three studies included viral cultures; of those, two resulted in positive cultures, both in a non-ICU setting. In one study, 3 of 39 samples were positive, and in the other, 4 of 4 were positive. Viral cultures in toilets, clinical areas, staff areas, and public areas were negative.
One of these studies assessed viral concentration and found that the median interquartile range was 4.8 tissue culture infectious dose (TCID50)/m3 for particles <1 mcm, 4.27 TCID50/m3 for particles 1-4 mcm, and 1.82 TCID50/m3 for particles >4 mcm.
Although viable viruses weren’t found in staff areas, the presence of viral RNA in places such as dining rooms and meeting rooms raises a concern, Dr. Birgand said.
“All of these staff areas are probably playing an important role in contamination,” he said. “It’s pretty easy to see when you are dining, you are not wearing a face mask, and it’s associated with a strong risk when there is a strong dissemination of the virus in the community.”
Studies on contact tracing among health care workers have also identified meeting rooms and dining rooms as the second most common source of infection after community contact, he said.
In general, the findings of the review correspond to epidemiologic studies, said Angela Rasmussen, PhD, a virologist with the Georgetown University Center for Global Health Science and Security, Washington, who was not involved in the review. “Absent aerosol-generating procedures, health care workers are largely not getting infected when they take droplet precautions.”
One reason may be that patients shed the most infectious viruses a couple of days before and after symptoms begin. By the time they’re hospitalized, they’re less likely to be contagious but may continue to shed viral RNA.
“We don’t really know the basis for the persistence of RNA being produced long after people have been infected and have recovered from the acute infection,” she said, “but it has been observed quite frequently.”
Although the virus cannot remain viable for very long in the air, remnants may still be detected in the form of RNA, Dr. Rasmussen said. In addition, hospitals often do a good job of ventilation.
She pointed out that it can be difficult to cultivate viruses in air samples because of contaminants such as bacteria and fungi. “That’s one of the limitations of a study like this. You’re not really sure if it’s because there’s no viable virus there or because you just aren’t able to collect samples that would allow you to determine that.”
Dr. Birgand and colleagues acknowledged other limitations. The studies they reviewed used different approaches to sampling. Different procedures may have been underway in the rooms being sampled, and factors such as temperature and humidity could have affected the results. In addition, the studies used different cycle thresholds for PCR positivity.
A version of this article first appeared on Medscape.com.
New resilience center targets traumatized health care workers
A physician assistant participating in a virtual workshop began to cry, confessing that she felt overwhelmed with guilt because New Yorkers were hailing her as a frontline hero in the pandemic. That was when Joe Ciavarro knew he was in the right place.
“She was saying all the things I could not verbalize because I, too, didn’t feel like I deserved all this praise and thousands of people cheering for us every evening when people were losing jobs, didn’t have money for food, and their loved ones were dying without family at their side,” says Mr. Ciavarro, a PA at Mount Sinai Medical Center in New York.
Mr. Ciavarro, who also manages 170 other PAs on two of Mount Sinai’s campuses in Manhattan, has been on the front lines since COVID-19 first hit; he lost a colleague and friend to suicide in September.
The mental anguish from his job prompted him to sign up for the resilience workshop offered by Mount Sinai’s Center for Stress, Resilience, and Personal Growth. The center – the first of its kind in North America – was launched in June to help health care workers like him cope with the intense psychological pressures they were facing. The weekly workshops became a safe place where Mr. Ciavarro and other staff members could share their darkest fears and learn ways to help them deal with their situation.
“It’s been grueling but we learned how to take care of ourselves so we can take care of our patients,” said Mr. Ciavarro. “This has become like a guided group therapy session on ways to manage and develop resilience. And I feel like my emotions are validated, knowing that others feel the same way.”
Caring for their own
Medical professionals treating patients with COVID-19 are in similar predicaments, and the psychological fallout is enormous: They’re exhausted by the seemingly never-ending patient load and staffing shortages, and haunted by fears for their own safety and that of their families. Studies in China, Canada, and Italy have revealed that a significant number of doctors and nurses in the early days of the pandemic experienced high levels of distress, depression, anxiety, nightmares, and insomnia.
after witnessing the deaths of so many patients who were alone, without family.
But the resilience workshop that Mr. Ciavarro attended offers some hope and is part of a multifaceted program that aims to be a model for other institutions and communities. The Mount Sinai health system already had some programs in place, including centers for 9/11 responders, for spirituality and health, and a wellness program to aid burned-out doctors. But the leadership at Mount Sinai, which includes psychiatrist Dennis Charney, MD, dean of the medical school and a leading expert on PTSD, knew early in the pandemic that emotional and psychological distress would plague health care workers, according to Deborah Marin, MD, director of the new center.
“We decided to quickly put in place a program that we could do virtually, with workshops and apps, that would give access to several services above and beyond what was already going on,” says Dr. Marin, a professor of psychiatry at the Icahn School of Medicine at Mount Sinai, New York, who also directs their center for spirituality and health.
The key components include a comprehensive screening tool that helps doctors at the center identify which potential participants are most at risk. Participants build personal inventories that detail the intensity of work-related exposures, personal or family stressors that have arisen because of the pandemic, or any mental health conditions or substance abuse problems that may make staff members more vulnerable.
The weekly workshops led by trained staff are designed to give participants the tools to foster resilience and process their experiences. Online apps provide feedback on their progress and engage them with video and other resources around meditation, relaxation, and resilience techniques.
In addition, all 40,000 members of the Mount Sinai staff are eligible for up to 14 one-on-one sessions with psychologists and psychiatrists who specialize in treating trauma.
“That’s highly unusual – to offer this at no cost to everyone,” said Dr. Marin. “We also have a treatment service that is specifically focused on behavioral health care, so people can learn better coping strategies, and we also have social workers to provide coaching.”
While the center doesn’t have specific numbers on how many nurses, physicians, and other staff have participated in treatment, they have trained over 70 peer leaders for their five workshops that home in on the most important factors of resilience.
“We’ve gotten enthusiastic responses from PAs and nurses,” said Craig Katz, MD, an expert in disaster psychiatry at Mount Sinai and a workshop moderator. Physicians have been slower to get on board. “Doctors are a tough nut to crack – it’s largely a culture where they may burn out but don’t want to talk about it. And asking for help is a hard transition for physicians to make.”
How to protect in midst of trauma
In formulating the program’s platform, Mount Sinai experts drew upon their extensive experience aiding 9/11 responders at the World Trade Center (WTC), as well as their system-wide wellness program that aids demoralized and burned-out physicians. While the reach of the pandemic is much broader than 9/11, experts see some commonalities in conditions that emerge after traumatic events, and they also discovered what can help.
“We learned from our WTC experience about what are protective factors – what are the social supports that buffer against depression, anxiety, and PTSD,” said Jonathan DePierro, PhD, clinical director of CSRPG and a psychologist at the Mount Sinai WTC Mental Health Program. “We also learned that people who have more prolonged exposures are at greater risk of developing mental health difficulties.”
The program itself reflects these lessons – and that’s why it’s open to all employees, not just medical professionals. Housekeepers, security staffers, even construction workers are also dealing with their lives being in danger. “That wasn’t in their job description,” said Dr. DePierro. “These people tend to have fewer social and economic resources, make less money and have fewer structural supports, which makes them even more vulnerable.”
Dr. Charney’s strategies on building resilience became a bible of sorts for the workshops, according to Dr. Katz, who authored the training curriculum. Sessions deal with how to build up reservoirs of realistic optimism, keep gratitude journals, find spiritual meaning in their lives, maintain physical wellness and create networks of social support. The workshops are meant to help participants create action plans, to reach out for support in their social networks, and keep the focus on the positives.
The goal is to give demoralized health care workers a renewed sense of competence. “The resilience workshop is a launching point to get people to show up and talk,” said Dr. Katz. “And if we do that, we’ve accomplished a lot just getting people in the door.”
The center will also have a research component to identify what works and what doesn’t so their platform can provide a template for other institutions; Dr. Marin said they’ve gotten inquiries about the program from major hospital systems in Michigan and California. They’ll also conduct longitudinal research to determine what lingering problems persist among healthcare workers over time.
Since the center opened its virtual doors, the curriculum has also been altered in response to feedback from the support staff, many of whom live in the community that surrounds Mount Sinai in northern Manhattan, which is largely lower-income Latinx and Black individuals. Workshop materials have been translated into Spanish and now feature people who reflect a more diverse set of experiences.
“Many of our employees and the population we serve identify as non-White so we’ve been doing outreach with a lot of the local unions,” said Dr. Marin. “Our next step is to take what we’re doing and work with local community organizations.”
A version of this article first appeared on Medscape.com.
A physician assistant participating in a virtual workshop began to cry, confessing that she felt overwhelmed with guilt because New Yorkers were hailing her as a frontline hero in the pandemic. That was when Joe Ciavarro knew he was in the right place.
“She was saying all the things I could not verbalize because I, too, didn’t feel like I deserved all this praise and thousands of people cheering for us every evening when people were losing jobs, didn’t have money for food, and their loved ones were dying without family at their side,” says Mr. Ciavarro, a PA at Mount Sinai Medical Center in New York.
Mr. Ciavarro, who also manages 170 other PAs on two of Mount Sinai’s campuses in Manhattan, has been on the front lines since COVID-19 first hit; he lost a colleague and friend to suicide in September.
The mental anguish from his job prompted him to sign up for the resilience workshop offered by Mount Sinai’s Center for Stress, Resilience, and Personal Growth. The center – the first of its kind in North America – was launched in June to help health care workers like him cope with the intense psychological pressures they were facing. The weekly workshops became a safe place where Mr. Ciavarro and other staff members could share their darkest fears and learn ways to help them deal with their situation.
“It’s been grueling but we learned how to take care of ourselves so we can take care of our patients,” said Mr. Ciavarro. “This has become like a guided group therapy session on ways to manage and develop resilience. And I feel like my emotions are validated, knowing that others feel the same way.”
Caring for their own
Medical professionals treating patients with COVID-19 are in similar predicaments, and the psychological fallout is enormous: They’re exhausted by the seemingly never-ending patient load and staffing shortages, and haunted by fears for their own safety and that of their families. Studies in China, Canada, and Italy have revealed that a significant number of doctors and nurses in the early days of the pandemic experienced high levels of distress, depression, anxiety, nightmares, and insomnia.
after witnessing the deaths of so many patients who were alone, without family.
But the resilience workshop that Mr. Ciavarro attended offers some hope and is part of a multifaceted program that aims to be a model for other institutions and communities. The Mount Sinai health system already had some programs in place, including centers for 9/11 responders, for spirituality and health, and a wellness program to aid burned-out doctors. But the leadership at Mount Sinai, which includes psychiatrist Dennis Charney, MD, dean of the medical school and a leading expert on PTSD, knew early in the pandemic that emotional and psychological distress would plague health care workers, according to Deborah Marin, MD, director of the new center.
“We decided to quickly put in place a program that we could do virtually, with workshops and apps, that would give access to several services above and beyond what was already going on,” says Dr. Marin, a professor of psychiatry at the Icahn School of Medicine at Mount Sinai, New York, who also directs their center for spirituality and health.
The key components include a comprehensive screening tool that helps doctors at the center identify which potential participants are most at risk. Participants build personal inventories that detail the intensity of work-related exposures, personal or family stressors that have arisen because of the pandemic, or any mental health conditions or substance abuse problems that may make staff members more vulnerable.
The weekly workshops led by trained staff are designed to give participants the tools to foster resilience and process their experiences. Online apps provide feedback on their progress and engage them with video and other resources around meditation, relaxation, and resilience techniques.
In addition, all 40,000 members of the Mount Sinai staff are eligible for up to 14 one-on-one sessions with psychologists and psychiatrists who specialize in treating trauma.
“That’s highly unusual – to offer this at no cost to everyone,” said Dr. Marin. “We also have a treatment service that is specifically focused on behavioral health care, so people can learn better coping strategies, and we also have social workers to provide coaching.”
While the center doesn’t have specific numbers on how many nurses, physicians, and other staff have participated in treatment, they have trained over 70 peer leaders for their five workshops that home in on the most important factors of resilience.
“We’ve gotten enthusiastic responses from PAs and nurses,” said Craig Katz, MD, an expert in disaster psychiatry at Mount Sinai and a workshop moderator. Physicians have been slower to get on board. “Doctors are a tough nut to crack – it’s largely a culture where they may burn out but don’t want to talk about it. And asking for help is a hard transition for physicians to make.”
How to protect in midst of trauma
In formulating the program’s platform, Mount Sinai experts drew upon their extensive experience aiding 9/11 responders at the World Trade Center (WTC), as well as their system-wide wellness program that aids demoralized and burned-out physicians. While the reach of the pandemic is much broader than 9/11, experts see some commonalities in conditions that emerge after traumatic events, and they also discovered what can help.
“We learned from our WTC experience about what are protective factors – what are the social supports that buffer against depression, anxiety, and PTSD,” said Jonathan DePierro, PhD, clinical director of CSRPG and a psychologist at the Mount Sinai WTC Mental Health Program. “We also learned that people who have more prolonged exposures are at greater risk of developing mental health difficulties.”
The program itself reflects these lessons – and that’s why it’s open to all employees, not just medical professionals. Housekeepers, security staffers, even construction workers are also dealing with their lives being in danger. “That wasn’t in their job description,” said Dr. DePierro. “These people tend to have fewer social and economic resources, make less money and have fewer structural supports, which makes them even more vulnerable.”
Dr. Charney’s strategies on building resilience became a bible of sorts for the workshops, according to Dr. Katz, who authored the training curriculum. Sessions deal with how to build up reservoirs of realistic optimism, keep gratitude journals, find spiritual meaning in their lives, maintain physical wellness and create networks of social support. The workshops are meant to help participants create action plans, to reach out for support in their social networks, and keep the focus on the positives.
The goal is to give demoralized health care workers a renewed sense of competence. “The resilience workshop is a launching point to get people to show up and talk,” said Dr. Katz. “And if we do that, we’ve accomplished a lot just getting people in the door.”
The center will also have a research component to identify what works and what doesn’t so their platform can provide a template for other institutions; Dr. Marin said they’ve gotten inquiries about the program from major hospital systems in Michigan and California. They’ll also conduct longitudinal research to determine what lingering problems persist among healthcare workers over time.
Since the center opened its virtual doors, the curriculum has also been altered in response to feedback from the support staff, many of whom live in the community that surrounds Mount Sinai in northern Manhattan, which is largely lower-income Latinx and Black individuals. Workshop materials have been translated into Spanish and now feature people who reflect a more diverse set of experiences.
“Many of our employees and the population we serve identify as non-White so we’ve been doing outreach with a lot of the local unions,” said Dr. Marin. “Our next step is to take what we’re doing and work with local community organizations.”
A version of this article first appeared on Medscape.com.
A physician assistant participating in a virtual workshop began to cry, confessing that she felt overwhelmed with guilt because New Yorkers were hailing her as a frontline hero in the pandemic. That was when Joe Ciavarro knew he was in the right place.
“She was saying all the things I could not verbalize because I, too, didn’t feel like I deserved all this praise and thousands of people cheering for us every evening when people were losing jobs, didn’t have money for food, and their loved ones were dying without family at their side,” says Mr. Ciavarro, a PA at Mount Sinai Medical Center in New York.
Mr. Ciavarro, who also manages 170 other PAs on two of Mount Sinai’s campuses in Manhattan, has been on the front lines since COVID-19 first hit; he lost a colleague and friend to suicide in September.
The mental anguish from his job prompted him to sign up for the resilience workshop offered by Mount Sinai’s Center for Stress, Resilience, and Personal Growth. The center – the first of its kind in North America – was launched in June to help health care workers like him cope with the intense psychological pressures they were facing. The weekly workshops became a safe place where Mr. Ciavarro and other staff members could share their darkest fears and learn ways to help them deal with their situation.
“It’s been grueling but we learned how to take care of ourselves so we can take care of our patients,” said Mr. Ciavarro. “This has become like a guided group therapy session on ways to manage and develop resilience. And I feel like my emotions are validated, knowing that others feel the same way.”
Caring for their own
Medical professionals treating patients with COVID-19 are in similar predicaments, and the psychological fallout is enormous: They’re exhausted by the seemingly never-ending patient load and staffing shortages, and haunted by fears for their own safety and that of their families. Studies in China, Canada, and Italy have revealed that a significant number of doctors and nurses in the early days of the pandemic experienced high levels of distress, depression, anxiety, nightmares, and insomnia.
after witnessing the deaths of so many patients who were alone, without family.
But the resilience workshop that Mr. Ciavarro attended offers some hope and is part of a multifaceted program that aims to be a model for other institutions and communities. The Mount Sinai health system already had some programs in place, including centers for 9/11 responders, for spirituality and health, and a wellness program to aid burned-out doctors. But the leadership at Mount Sinai, which includes psychiatrist Dennis Charney, MD, dean of the medical school and a leading expert on PTSD, knew early in the pandemic that emotional and psychological distress would plague health care workers, according to Deborah Marin, MD, director of the new center.
“We decided to quickly put in place a program that we could do virtually, with workshops and apps, that would give access to several services above and beyond what was already going on,” says Dr. Marin, a professor of psychiatry at the Icahn School of Medicine at Mount Sinai, New York, who also directs their center for spirituality and health.
The key components include a comprehensive screening tool that helps doctors at the center identify which potential participants are most at risk. Participants build personal inventories that detail the intensity of work-related exposures, personal or family stressors that have arisen because of the pandemic, or any mental health conditions or substance abuse problems that may make staff members more vulnerable.
The weekly workshops led by trained staff are designed to give participants the tools to foster resilience and process their experiences. Online apps provide feedback on their progress and engage them with video and other resources around meditation, relaxation, and resilience techniques.
In addition, all 40,000 members of the Mount Sinai staff are eligible for up to 14 one-on-one sessions with psychologists and psychiatrists who specialize in treating trauma.
“That’s highly unusual – to offer this at no cost to everyone,” said Dr. Marin. “We also have a treatment service that is specifically focused on behavioral health care, so people can learn better coping strategies, and we also have social workers to provide coaching.”
While the center doesn’t have specific numbers on how many nurses, physicians, and other staff have participated in treatment, they have trained over 70 peer leaders for their five workshops that home in on the most important factors of resilience.
“We’ve gotten enthusiastic responses from PAs and nurses,” said Craig Katz, MD, an expert in disaster psychiatry at Mount Sinai and a workshop moderator. Physicians have been slower to get on board. “Doctors are a tough nut to crack – it’s largely a culture where they may burn out but don’t want to talk about it. And asking for help is a hard transition for physicians to make.”
How to protect in midst of trauma
In formulating the program’s platform, Mount Sinai experts drew upon their extensive experience aiding 9/11 responders at the World Trade Center (WTC), as well as their system-wide wellness program that aids demoralized and burned-out physicians. While the reach of the pandemic is much broader than 9/11, experts see some commonalities in conditions that emerge after traumatic events, and they also discovered what can help.
“We learned from our WTC experience about what are protective factors – what are the social supports that buffer against depression, anxiety, and PTSD,” said Jonathan DePierro, PhD, clinical director of CSRPG and a psychologist at the Mount Sinai WTC Mental Health Program. “We also learned that people who have more prolonged exposures are at greater risk of developing mental health difficulties.”
The program itself reflects these lessons – and that’s why it’s open to all employees, not just medical professionals. Housekeepers, security staffers, even construction workers are also dealing with their lives being in danger. “That wasn’t in their job description,” said Dr. DePierro. “These people tend to have fewer social and economic resources, make less money and have fewer structural supports, which makes them even more vulnerable.”
Dr. Charney’s strategies on building resilience became a bible of sorts for the workshops, according to Dr. Katz, who authored the training curriculum. Sessions deal with how to build up reservoirs of realistic optimism, keep gratitude journals, find spiritual meaning in their lives, maintain physical wellness and create networks of social support. The workshops are meant to help participants create action plans, to reach out for support in their social networks, and keep the focus on the positives.
The goal is to give demoralized health care workers a renewed sense of competence. “The resilience workshop is a launching point to get people to show up and talk,” said Dr. Katz. “And if we do that, we’ve accomplished a lot just getting people in the door.”
The center will also have a research component to identify what works and what doesn’t so their platform can provide a template for other institutions; Dr. Marin said they’ve gotten inquiries about the program from major hospital systems in Michigan and California. They’ll also conduct longitudinal research to determine what lingering problems persist among healthcare workers over time.
Since the center opened its virtual doors, the curriculum has also been altered in response to feedback from the support staff, many of whom live in the community that surrounds Mount Sinai in northern Manhattan, which is largely lower-income Latinx and Black individuals. Workshop materials have been translated into Spanish and now feature people who reflect a more diverse set of experiences.
“Many of our employees and the population we serve identify as non-White so we’ve been doing outreach with a lot of the local unions,” said Dr. Marin. “Our next step is to take what we’re doing and work with local community organizations.”
A version of this article first appeared on Medscape.com.
COVID-19 mortality rates declined, but vary by hospital
Mortality rates for inpatients with COVID-19 dropped significantly during the first 6 months of the pandemic, but outcomes depend on the hospital where patients receive care, new data show.
“[T]he characteristic that is most associated with poor or worsening hospital outcomes is high or increasing community case rates,” write David A. Asch, MD, MBA, executive director of the Center for Health Care Innovation at the University of Pennsylvania in Philadelphia, and colleagues.
The relationship between COVID-19 mortality rates and local disease prevalence suggests that “hospitals do worse when they are burdened with cases and is consistent with imperatives to flatten the curve,” the authors continue. “As case rates of COVID-19 increase across the nation, hospital mortality outcomes may worsen.”
The researchers published their study online December 22 in JAMA Internal Medicine.
The quick and substantial improvement in survival “is a tribute in part to new science — for example, the science that revealed the benefits of dexamethasone,” Asch told Medscape Medical News. “But it’s also a tribute to the doctors and nurses in the hospitals who developed experience. It’s a cliché to refer to them as heroes, but that is what they are. The science and the heroic experience continues on, and so I’m optimistic that we’ll see even more improvement over time.”
However, the data also indicate that “with lots of disease in the community, hospitals may have a harder time keeping patients alive,” Asch said. “And of course the reason this is bad news is that community level case rates are rising all over, and in some cases at rapid rates. With that rise, we might be giving back some of our past gains in survival — just as the vaccine is beginning to be distributed.”
Examining mortality trends
The researchers analyzed administrative claims data from a large national health insurer. They included data from 38,517 adults who were admitted with COVID-19 to 955 US hospitals between January 1 and June 30 of this year. The investigators estimated hospitals’ risk-standardized rate of 30-day in-hospital mortality or referral to hospice, adjusted for patient-level characteristics.
Overall, 3179 patients (8.25%) died, and 1433 patients (3.7%) were referred to hospice. Risk-standardized mortality or hospice referral rates for individual hospitals ranged from 5.7% to 24.7%. The average rate was 9.1% in the best-performing quintile, compared with 15.7% in the worst-performing quintile.
In a subset of 398 hospitals that had at least 10 patients admitted for COVID-19 during early (January 1 through April 30) and later periods (between May 1 and June 30), rates in all but one hospital improved, and 94% improved by at least 25%. The average risk-standardized event rate declined from 16.6% to 9.3%.
“That rate of relative improvement is striking and encouraging, but perhaps not surprising,” Asch and coauthors write. “Early efforts at treating patients with COVID-19 were based on experience with previously known causes of severe respiratory illness. Later efforts could draw on experiences specific to SARS-CoV-2 infection.”
For instance, doctors tried different inpatient management approaches, such as early vs late assisted ventilation, differences in oxygen flow, prone or supine positioning, and anticoagulation. “Those efforts varied in how systematically they were evaluated, but our results suggest that valuable experience was gained,” the authors note.
In addition, variation between hospitals could reflect differences in quality or different admission thresholds, they continue.
The study provides “a reason for optimism that our healthcare system has improved in our ability to care for persons with COVID-19,” write Leon Boudourakis, MD, MHS, and Amit Uppal, MD, in a related commentary. Boudourakis and Uppal are both affiliated with NYC Health + Hospitals in New York City and with SUNY Downstate and New York University School of Medicine, respectively.
Similar improvements in mortality rates have been reported in the United Kingdom and in a New York City hospital system, the editorialists note. The lower mortality rates may represent clinical, healthcare system, and epidemiologic trends.
“Since the first wave of serious COVID-19 cases, physicians have learned a great deal about the best ways to treat this serious infection,” they say. “Steroids may decrease mortality in patients with respiratory failure. Remdesivir may shorten hospitalizations of patients with serious illness. Anticoagulation and prone positioning may help certain patients. Using noninvasive ventilation and high-flow oxygen therapy may spare subsets of patients from the harms of intubation, such as ventilator-induced lung injury.»
Overwhelmed hospitals
“Hospitals do not perform as well when they are overwhelmed,” which may be a reason for the correlation between community prevalence and mortality rates, Boudourakis and Uppal suggested. “In particular, patients with a precarious respiratory status require expert, meticulous therapy to avoid intubation; those who undergo intubation or have kidney failure require nuanced and timely expert care with ventilatory adjustments and kidney replacement therapy, which are difficult to perform optimally when hospital capacity is strained.”
Although the death rate has fallen to about 9% for hospitalized patients, “9% is still high,” Asch said.
“Our results show that hospitals can’t do it on their own,” Asch said. “They need all of us to keep the community spread of the disease down. The right answer now is the right answer since the beginning of the pandemic: Keep your distance, wash your hands, and wear a mask.”
Asch, Boudourakis, and Uppal have disclosed no relevant financial relationships. A study coauthor reported personal fees and grants from pharmaceutical companies outside the submitted work.
A version of this article first appeared on Medscape.com.
Mortality rates for inpatients with COVID-19 dropped significantly during the first 6 months of the pandemic, but outcomes depend on the hospital where patients receive care, new data show.
“[T]he characteristic that is most associated with poor or worsening hospital outcomes is high or increasing community case rates,” write David A. Asch, MD, MBA, executive director of the Center for Health Care Innovation at the University of Pennsylvania in Philadelphia, and colleagues.
The relationship between COVID-19 mortality rates and local disease prevalence suggests that “hospitals do worse when they are burdened with cases and is consistent with imperatives to flatten the curve,” the authors continue. “As case rates of COVID-19 increase across the nation, hospital mortality outcomes may worsen.”
The researchers published their study online December 22 in JAMA Internal Medicine.
The quick and substantial improvement in survival “is a tribute in part to new science — for example, the science that revealed the benefits of dexamethasone,” Asch told Medscape Medical News. “But it’s also a tribute to the doctors and nurses in the hospitals who developed experience. It’s a cliché to refer to them as heroes, but that is what they are. The science and the heroic experience continues on, and so I’m optimistic that we’ll see even more improvement over time.”
However, the data also indicate that “with lots of disease in the community, hospitals may have a harder time keeping patients alive,” Asch said. “And of course the reason this is bad news is that community level case rates are rising all over, and in some cases at rapid rates. With that rise, we might be giving back some of our past gains in survival — just as the vaccine is beginning to be distributed.”
Examining mortality trends
The researchers analyzed administrative claims data from a large national health insurer. They included data from 38,517 adults who were admitted with COVID-19 to 955 US hospitals between January 1 and June 30 of this year. The investigators estimated hospitals’ risk-standardized rate of 30-day in-hospital mortality or referral to hospice, adjusted for patient-level characteristics.
Overall, 3179 patients (8.25%) died, and 1433 patients (3.7%) were referred to hospice. Risk-standardized mortality or hospice referral rates for individual hospitals ranged from 5.7% to 24.7%. The average rate was 9.1% in the best-performing quintile, compared with 15.7% in the worst-performing quintile.
In a subset of 398 hospitals that had at least 10 patients admitted for COVID-19 during early (January 1 through April 30) and later periods (between May 1 and June 30), rates in all but one hospital improved, and 94% improved by at least 25%. The average risk-standardized event rate declined from 16.6% to 9.3%.
“That rate of relative improvement is striking and encouraging, but perhaps not surprising,” Asch and coauthors write. “Early efforts at treating patients with COVID-19 were based on experience with previously known causes of severe respiratory illness. Later efforts could draw on experiences specific to SARS-CoV-2 infection.”
For instance, doctors tried different inpatient management approaches, such as early vs late assisted ventilation, differences in oxygen flow, prone or supine positioning, and anticoagulation. “Those efforts varied in how systematically they were evaluated, but our results suggest that valuable experience was gained,” the authors note.
In addition, variation between hospitals could reflect differences in quality or different admission thresholds, they continue.
The study provides “a reason for optimism that our healthcare system has improved in our ability to care for persons with COVID-19,” write Leon Boudourakis, MD, MHS, and Amit Uppal, MD, in a related commentary. Boudourakis and Uppal are both affiliated with NYC Health + Hospitals in New York City and with SUNY Downstate and New York University School of Medicine, respectively.
Similar improvements in mortality rates have been reported in the United Kingdom and in a New York City hospital system, the editorialists note. The lower mortality rates may represent clinical, healthcare system, and epidemiologic trends.
“Since the first wave of serious COVID-19 cases, physicians have learned a great deal about the best ways to treat this serious infection,” they say. “Steroids may decrease mortality in patients with respiratory failure. Remdesivir may shorten hospitalizations of patients with serious illness. Anticoagulation and prone positioning may help certain patients. Using noninvasive ventilation and high-flow oxygen therapy may spare subsets of patients from the harms of intubation, such as ventilator-induced lung injury.»
Overwhelmed hospitals
“Hospitals do not perform as well when they are overwhelmed,” which may be a reason for the correlation between community prevalence and mortality rates, Boudourakis and Uppal suggested. “In particular, patients with a precarious respiratory status require expert, meticulous therapy to avoid intubation; those who undergo intubation or have kidney failure require nuanced and timely expert care with ventilatory adjustments and kidney replacement therapy, which are difficult to perform optimally when hospital capacity is strained.”
Although the death rate has fallen to about 9% for hospitalized patients, “9% is still high,” Asch said.
“Our results show that hospitals can’t do it on their own,” Asch said. “They need all of us to keep the community spread of the disease down. The right answer now is the right answer since the beginning of the pandemic: Keep your distance, wash your hands, and wear a mask.”
Asch, Boudourakis, and Uppal have disclosed no relevant financial relationships. A study coauthor reported personal fees and grants from pharmaceutical companies outside the submitted work.
A version of this article first appeared on Medscape.com.
Mortality rates for inpatients with COVID-19 dropped significantly during the first 6 months of the pandemic, but outcomes depend on the hospital where patients receive care, new data show.
“[T]he characteristic that is most associated with poor or worsening hospital outcomes is high or increasing community case rates,” write David A. Asch, MD, MBA, executive director of the Center for Health Care Innovation at the University of Pennsylvania in Philadelphia, and colleagues.
The relationship between COVID-19 mortality rates and local disease prevalence suggests that “hospitals do worse when they are burdened with cases and is consistent with imperatives to flatten the curve,” the authors continue. “As case rates of COVID-19 increase across the nation, hospital mortality outcomes may worsen.”
The researchers published their study online December 22 in JAMA Internal Medicine.
The quick and substantial improvement in survival “is a tribute in part to new science — for example, the science that revealed the benefits of dexamethasone,” Asch told Medscape Medical News. “But it’s also a tribute to the doctors and nurses in the hospitals who developed experience. It’s a cliché to refer to them as heroes, but that is what they are. The science and the heroic experience continues on, and so I’m optimistic that we’ll see even more improvement over time.”
However, the data also indicate that “with lots of disease in the community, hospitals may have a harder time keeping patients alive,” Asch said. “And of course the reason this is bad news is that community level case rates are rising all over, and in some cases at rapid rates. With that rise, we might be giving back some of our past gains in survival — just as the vaccine is beginning to be distributed.”
Examining mortality trends
The researchers analyzed administrative claims data from a large national health insurer. They included data from 38,517 adults who were admitted with COVID-19 to 955 US hospitals between January 1 and June 30 of this year. The investigators estimated hospitals’ risk-standardized rate of 30-day in-hospital mortality or referral to hospice, adjusted for patient-level characteristics.
Overall, 3179 patients (8.25%) died, and 1433 patients (3.7%) were referred to hospice. Risk-standardized mortality or hospice referral rates for individual hospitals ranged from 5.7% to 24.7%. The average rate was 9.1% in the best-performing quintile, compared with 15.7% in the worst-performing quintile.
In a subset of 398 hospitals that had at least 10 patients admitted for COVID-19 during early (January 1 through April 30) and later periods (between May 1 and June 30), rates in all but one hospital improved, and 94% improved by at least 25%. The average risk-standardized event rate declined from 16.6% to 9.3%.
“That rate of relative improvement is striking and encouraging, but perhaps not surprising,” Asch and coauthors write. “Early efforts at treating patients with COVID-19 were based on experience with previously known causes of severe respiratory illness. Later efforts could draw on experiences specific to SARS-CoV-2 infection.”
For instance, doctors tried different inpatient management approaches, such as early vs late assisted ventilation, differences in oxygen flow, prone or supine positioning, and anticoagulation. “Those efforts varied in how systematically they were evaluated, but our results suggest that valuable experience was gained,” the authors note.
In addition, variation between hospitals could reflect differences in quality or different admission thresholds, they continue.
The study provides “a reason for optimism that our healthcare system has improved in our ability to care for persons with COVID-19,” write Leon Boudourakis, MD, MHS, and Amit Uppal, MD, in a related commentary. Boudourakis and Uppal are both affiliated with NYC Health + Hospitals in New York City and with SUNY Downstate and New York University School of Medicine, respectively.
Similar improvements in mortality rates have been reported in the United Kingdom and in a New York City hospital system, the editorialists note. The lower mortality rates may represent clinical, healthcare system, and epidemiologic trends.
“Since the first wave of serious COVID-19 cases, physicians have learned a great deal about the best ways to treat this serious infection,” they say. “Steroids may decrease mortality in patients with respiratory failure. Remdesivir may shorten hospitalizations of patients with serious illness. Anticoagulation and prone positioning may help certain patients. Using noninvasive ventilation and high-flow oxygen therapy may spare subsets of patients from the harms of intubation, such as ventilator-induced lung injury.»
Overwhelmed hospitals
“Hospitals do not perform as well when they are overwhelmed,” which may be a reason for the correlation between community prevalence and mortality rates, Boudourakis and Uppal suggested. “In particular, patients with a precarious respiratory status require expert, meticulous therapy to avoid intubation; those who undergo intubation or have kidney failure require nuanced and timely expert care with ventilatory adjustments and kidney replacement therapy, which are difficult to perform optimally when hospital capacity is strained.”
Although the death rate has fallen to about 9% for hospitalized patients, “9% is still high,” Asch said.
“Our results show that hospitals can’t do it on their own,” Asch said. “They need all of us to keep the community spread of the disease down. The right answer now is the right answer since the beginning of the pandemic: Keep your distance, wash your hands, and wear a mask.”
Asch, Boudourakis, and Uppal have disclosed no relevant financial relationships. A study coauthor reported personal fees and grants from pharmaceutical companies outside the submitted work.
A version of this article first appeared on Medscape.com.
Moderna’s COVID-19 vaccine deemed ‘highly effective,’ but further studies needed
The Food and Drug Administration’s Vaccines and Related Biological Products Advisory Committee (VRBPAC) evaluated
The panel acknowledged that further studies will be required post issuance of an Emergency Use Authorization (EUA) to collect additional data on the safety and effectiveness of the vaccine. A briefing document released by the FDA on Dec. 17, 2020, summarized interim results and included recommendations from VRBPAC on use of Moderna’s mRNA-1273 COVID-19 vaccine.
“On November 30, 2020, ModernaTX (the Sponsor) submitted an EUA request to FDA for an investigational COVID-19 vaccine (mRNA-1273) intended to prevent COVID-19,” the committee wrote.
The mRNA-1273 vaccine trial
Among 30,351 individuals aged 18 years and older, the efficacy, safety, and immunogenicity of the mRNA-1273 vaccine candidate was evaluated in a randomized, stratified, observer-blind, placebo-controlled phase 3 study. Participants were randomly assigned (1:1) to receive two injections of either 100 mcg of mRNA-1273 (n = 15,181) or saline placebo (n = 15,170) administered intramuscularly on day 1 and day 29.
The primary efficacy endpoint was efficacy of mRNA-1273 against PCR-confirmed COVID-19 with onset at least 14 days following the second dose. The primary safety endpoint was to characterize the safety of the vaccine following one or two doses.
Efficacy
Among 27,817 subjects included in the first interim analysis (data cutoff: Nov. 7, 2020), 5 cases of COVID-19 with onset at least 14 days after the second dose occurred among vaccine recipients and 90 case occurred among placebo recipients, corresponding to 94.5% vaccine efficacy (95% confidence interval, 86.5%-97.8%).
“Subgroup analyses of the primary efficacy endpoint showed similar efficacy point estimates across age groups, genders, racial and ethnic groups, and participants with medical comorbidities associated with high risk of severe COVID-19,” they reported.
Data from the final scheduled analysis of the primary efficacy endpoint (data cutoff: Nov. 21, 2020; median follow-up of >2 months after dose 2), demonstrated 94.1% vaccine efficacy (95% confidence interval, 89.3%-96.8%), corresponding to 11 cases of COVID-19 in the vaccine group and 185 cases in the placebo group.
When stratified by age, the vaccine efficacy was 95.6% (95% CI, 90.6%-97.9%) for individuals 18-64 years of age and 86.4% (95% CI, 61.4%-95.5%) for those 65 years of age or older.
In addition, results from secondary analyses indicated benefit for mRNA-1273 in preventing severe COVID-19 cases, COVID-19 in those with prior SARS-CoV-2 infection, and infection after the first dose, but these data were not conclusive.
Safety
Among 30,350 subjects included in the first interim analysis (data cutoff: Nov. 11, 2020; median follow-up of 7 weeks post second dose), no specific safety concerns were observed that would prevent issuance of an EUA.
Additional safety data (data cutoff: Nov. 25, 2020; median follow-up of 9 weeks post second dose) were provided on Dec. 7, 2020, but did not change the conclusions from the first interim analysis.
The most common vaccine-related adverse reactions were injection site pain (91.6%), fatigue (68.5%), headache (63.0%), muscle pain (59.6%), joint pain (44.8%), and chills (43.4%).
“The frequency of serious adverse events (SAEs) was low (1.0% in the mRNA-1273 arm and 1.0% in the placebo arm), without meaningful imbalances between study arms,” they reported.
Myocardial infarction (0.03%), nephrolithiasis (0.02%), and cholecystitis (0.02%) were the most common SAEs that were numerically greater in the vaccine arm than the placebo arm; however, the small number of cases does not infer a casual relationship.
“The 2-dose vaccination regimen was highly effective in preventing PCR-confirmed COVID-19 occurring at least 14 days after receipt of the second dose,” the committee wrote. “[However], it is critical to continue to gather data about the vaccine even after it is made available under EUA.”
The associated phase 3 study was sponsored by ModernaTX.
SOURCE: FDA Briefing Document: Moderna COVID-19 Vaccine. FDA Vaccines and Related Biological Products Advisory Committee. Published Dec. 17, 2020.
The Food and Drug Administration’s Vaccines and Related Biological Products Advisory Committee (VRBPAC) evaluated
The panel acknowledged that further studies will be required post issuance of an Emergency Use Authorization (EUA) to collect additional data on the safety and effectiveness of the vaccine. A briefing document released by the FDA on Dec. 17, 2020, summarized interim results and included recommendations from VRBPAC on use of Moderna’s mRNA-1273 COVID-19 vaccine.
“On November 30, 2020, ModernaTX (the Sponsor) submitted an EUA request to FDA for an investigational COVID-19 vaccine (mRNA-1273) intended to prevent COVID-19,” the committee wrote.
The mRNA-1273 vaccine trial
Among 30,351 individuals aged 18 years and older, the efficacy, safety, and immunogenicity of the mRNA-1273 vaccine candidate was evaluated in a randomized, stratified, observer-blind, placebo-controlled phase 3 study. Participants were randomly assigned (1:1) to receive two injections of either 100 mcg of mRNA-1273 (n = 15,181) or saline placebo (n = 15,170) administered intramuscularly on day 1 and day 29.
The primary efficacy endpoint was efficacy of mRNA-1273 against PCR-confirmed COVID-19 with onset at least 14 days following the second dose. The primary safety endpoint was to characterize the safety of the vaccine following one or two doses.
Efficacy
Among 27,817 subjects included in the first interim analysis (data cutoff: Nov. 7, 2020), 5 cases of COVID-19 with onset at least 14 days after the second dose occurred among vaccine recipients and 90 case occurred among placebo recipients, corresponding to 94.5% vaccine efficacy (95% confidence interval, 86.5%-97.8%).
“Subgroup analyses of the primary efficacy endpoint showed similar efficacy point estimates across age groups, genders, racial and ethnic groups, and participants with medical comorbidities associated with high risk of severe COVID-19,” they reported.
Data from the final scheduled analysis of the primary efficacy endpoint (data cutoff: Nov. 21, 2020; median follow-up of >2 months after dose 2), demonstrated 94.1% vaccine efficacy (95% confidence interval, 89.3%-96.8%), corresponding to 11 cases of COVID-19 in the vaccine group and 185 cases in the placebo group.
When stratified by age, the vaccine efficacy was 95.6% (95% CI, 90.6%-97.9%) for individuals 18-64 years of age and 86.4% (95% CI, 61.4%-95.5%) for those 65 years of age or older.
In addition, results from secondary analyses indicated benefit for mRNA-1273 in preventing severe COVID-19 cases, COVID-19 in those with prior SARS-CoV-2 infection, and infection after the first dose, but these data were not conclusive.
Safety
Among 30,350 subjects included in the first interim analysis (data cutoff: Nov. 11, 2020; median follow-up of 7 weeks post second dose), no specific safety concerns were observed that would prevent issuance of an EUA.
Additional safety data (data cutoff: Nov. 25, 2020; median follow-up of 9 weeks post second dose) were provided on Dec. 7, 2020, but did not change the conclusions from the first interim analysis.
The most common vaccine-related adverse reactions were injection site pain (91.6%), fatigue (68.5%), headache (63.0%), muscle pain (59.6%), joint pain (44.8%), and chills (43.4%).
“The frequency of serious adverse events (SAEs) was low (1.0% in the mRNA-1273 arm and 1.0% in the placebo arm), without meaningful imbalances between study arms,” they reported.
Myocardial infarction (0.03%), nephrolithiasis (0.02%), and cholecystitis (0.02%) were the most common SAEs that were numerically greater in the vaccine arm than the placebo arm; however, the small number of cases does not infer a casual relationship.
“The 2-dose vaccination regimen was highly effective in preventing PCR-confirmed COVID-19 occurring at least 14 days after receipt of the second dose,” the committee wrote. “[However], it is critical to continue to gather data about the vaccine even after it is made available under EUA.”
The associated phase 3 study was sponsored by ModernaTX.
SOURCE: FDA Briefing Document: Moderna COVID-19 Vaccine. FDA Vaccines and Related Biological Products Advisory Committee. Published Dec. 17, 2020.
The Food and Drug Administration’s Vaccines and Related Biological Products Advisory Committee (VRBPAC) evaluated
The panel acknowledged that further studies will be required post issuance of an Emergency Use Authorization (EUA) to collect additional data on the safety and effectiveness of the vaccine. A briefing document released by the FDA on Dec. 17, 2020, summarized interim results and included recommendations from VRBPAC on use of Moderna’s mRNA-1273 COVID-19 vaccine.
“On November 30, 2020, ModernaTX (the Sponsor) submitted an EUA request to FDA for an investigational COVID-19 vaccine (mRNA-1273) intended to prevent COVID-19,” the committee wrote.
The mRNA-1273 vaccine trial
Among 30,351 individuals aged 18 years and older, the efficacy, safety, and immunogenicity of the mRNA-1273 vaccine candidate was evaluated in a randomized, stratified, observer-blind, placebo-controlled phase 3 study. Participants were randomly assigned (1:1) to receive two injections of either 100 mcg of mRNA-1273 (n = 15,181) or saline placebo (n = 15,170) administered intramuscularly on day 1 and day 29.
The primary efficacy endpoint was efficacy of mRNA-1273 against PCR-confirmed COVID-19 with onset at least 14 days following the second dose. The primary safety endpoint was to characterize the safety of the vaccine following one or two doses.
Efficacy
Among 27,817 subjects included in the first interim analysis (data cutoff: Nov. 7, 2020), 5 cases of COVID-19 with onset at least 14 days after the second dose occurred among vaccine recipients and 90 case occurred among placebo recipients, corresponding to 94.5% vaccine efficacy (95% confidence interval, 86.5%-97.8%).
“Subgroup analyses of the primary efficacy endpoint showed similar efficacy point estimates across age groups, genders, racial and ethnic groups, and participants with medical comorbidities associated with high risk of severe COVID-19,” they reported.
Data from the final scheduled analysis of the primary efficacy endpoint (data cutoff: Nov. 21, 2020; median follow-up of >2 months after dose 2), demonstrated 94.1% vaccine efficacy (95% confidence interval, 89.3%-96.8%), corresponding to 11 cases of COVID-19 in the vaccine group and 185 cases in the placebo group.
When stratified by age, the vaccine efficacy was 95.6% (95% CI, 90.6%-97.9%) for individuals 18-64 years of age and 86.4% (95% CI, 61.4%-95.5%) for those 65 years of age or older.
In addition, results from secondary analyses indicated benefit for mRNA-1273 in preventing severe COVID-19 cases, COVID-19 in those with prior SARS-CoV-2 infection, and infection after the first dose, but these data were not conclusive.
Safety
Among 30,350 subjects included in the first interim analysis (data cutoff: Nov. 11, 2020; median follow-up of 7 weeks post second dose), no specific safety concerns were observed that would prevent issuance of an EUA.
Additional safety data (data cutoff: Nov. 25, 2020; median follow-up of 9 weeks post second dose) were provided on Dec. 7, 2020, but did not change the conclusions from the first interim analysis.
The most common vaccine-related adverse reactions were injection site pain (91.6%), fatigue (68.5%), headache (63.0%), muscle pain (59.6%), joint pain (44.8%), and chills (43.4%).
“The frequency of serious adverse events (SAEs) was low (1.0% in the mRNA-1273 arm and 1.0% in the placebo arm), without meaningful imbalances between study arms,” they reported.
Myocardial infarction (0.03%), nephrolithiasis (0.02%), and cholecystitis (0.02%) were the most common SAEs that were numerically greater in the vaccine arm than the placebo arm; however, the small number of cases does not infer a casual relationship.
“The 2-dose vaccination regimen was highly effective in preventing PCR-confirmed COVID-19 occurring at least 14 days after receipt of the second dose,” the committee wrote. “[However], it is critical to continue to gather data about the vaccine even after it is made available under EUA.”
The associated phase 3 study was sponsored by ModernaTX.
SOURCE: FDA Briefing Document: Moderna COVID-19 Vaccine. FDA Vaccines and Related Biological Products Advisory Committee. Published Dec. 17, 2020.
Key clinical point: The FDA’s Vaccines and Related Biological Products Advisory Committee regarded Moderna’s COVID-19 vaccine as highly effective with a favorable safety profile, based on interim phase 3 results.
Major finding: The two-dose vaccine regimen had a low frequency of serious adverse events (1.0% each in the mRNA-1273 and placebo arms, respectively) and demonstrated 94.1% (95% CI, 89.3%-96.8%) vaccine efficacy.
Study details: A briefing document summarized interim data and recommendations from the FDA’s VRBPAC on Moderna’s mRNA-1273 COVID-19 vaccine.
Disclosures: The associated phase 3 study was sponsored by ModernaTX.
Source: FDA Briefing Document: Moderna COVID-19 Vaccine. FDA Vaccines and Related Biological Products Advisory Committee. Published Dec. 17, 2020.
Child abuse visits to EDs declined in 2020, but not admissions
but the visits in 2020 were significantly more likely to result in hospitalization, based on analysis of a national ED database.
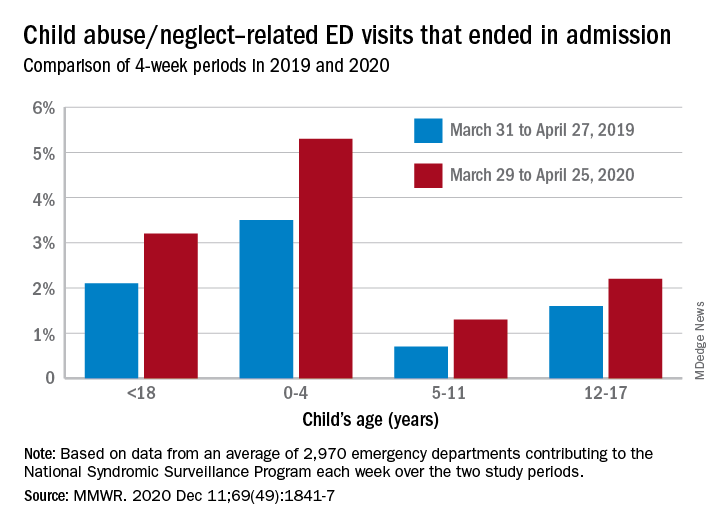
The number of ED visits involving child abuse and neglect was down by 53% during the 4-week period from March 29 to April 25, 2020, compared with the 4 weeks from March 31 to April 27, 2019. The proportion of those ED visits that ended in hospitalizations, however, increased from 2.1% in 2019 to 3.2% in 2020, Elizabeth Swedo, MD, and associates at the Centers for Disease Control and Prevention said in the Morbidity and Mortality Weekly Report.
“ED visits related to suspected or confirmed child abuse and neglect decreased beginning the week of March 15, 2020, coinciding with the declaration of a national emergency related to COVID-19 and implementation of community mitigation measures,” they wrote.
An earlier study involving the same database (the National Syndromic Surveillance Program) showed that, over the two same 4-week periods, the volume of all ED visits in 2020 was down 72% for children aged 10 years and younger and 71% for those aged 11-14 years.
In the current study, however, all age subgroups had significant increases in hospital admissions. The proportion of ED visits related to child abuse and neglect that resulted in hospitalization rose from 3.5% in 2019 to 5.3% in 2020 among ages 0-4 years, 0.7% to 1.3% for ages 5-11 years, and 1.6% to 2.2% for adolescents aged 12-17, Dr. Swedo and associates reported.
The absence of a corresponding drop in hospitalizations may be tied to risk factors related to the pandemic, “such as loss of income, increased stress related to parental child care and schooling responsibilities, and increased substance use and mental health conditions among adults,” the investigators added.
The National Syndromic Surveillance Program receives daily data from 3,310 EDs in 47 states, but the number of facilities meeting the investigators’ criteria averaged 2,970 a week for the 8 weeks of the study period.
SOURCE: Swedo E et al. MMWR. 2020 Dec. 11;69(49):1841-7.
but the visits in 2020 were significantly more likely to result in hospitalization, based on analysis of a national ED database.

The number of ED visits involving child abuse and neglect was down by 53% during the 4-week period from March 29 to April 25, 2020, compared with the 4 weeks from March 31 to April 27, 2019. The proportion of those ED visits that ended in hospitalizations, however, increased from 2.1% in 2019 to 3.2% in 2020, Elizabeth Swedo, MD, and associates at the Centers for Disease Control and Prevention said in the Morbidity and Mortality Weekly Report.
“ED visits related to suspected or confirmed child abuse and neglect decreased beginning the week of March 15, 2020, coinciding with the declaration of a national emergency related to COVID-19 and implementation of community mitigation measures,” they wrote.
An earlier study involving the same database (the National Syndromic Surveillance Program) showed that, over the two same 4-week periods, the volume of all ED visits in 2020 was down 72% for children aged 10 years and younger and 71% for those aged 11-14 years.
In the current study, however, all age subgroups had significant increases in hospital admissions. The proportion of ED visits related to child abuse and neglect that resulted in hospitalization rose from 3.5% in 2019 to 5.3% in 2020 among ages 0-4 years, 0.7% to 1.3% for ages 5-11 years, and 1.6% to 2.2% for adolescents aged 12-17, Dr. Swedo and associates reported.
The absence of a corresponding drop in hospitalizations may be tied to risk factors related to the pandemic, “such as loss of income, increased stress related to parental child care and schooling responsibilities, and increased substance use and mental health conditions among adults,” the investigators added.
The National Syndromic Surveillance Program receives daily data from 3,310 EDs in 47 states, but the number of facilities meeting the investigators’ criteria averaged 2,970 a week for the 8 weeks of the study period.
SOURCE: Swedo E et al. MMWR. 2020 Dec. 11;69(49):1841-7.
but the visits in 2020 were significantly more likely to result in hospitalization, based on analysis of a national ED database.

The number of ED visits involving child abuse and neglect was down by 53% during the 4-week period from March 29 to April 25, 2020, compared with the 4 weeks from March 31 to April 27, 2019. The proportion of those ED visits that ended in hospitalizations, however, increased from 2.1% in 2019 to 3.2% in 2020, Elizabeth Swedo, MD, and associates at the Centers for Disease Control and Prevention said in the Morbidity and Mortality Weekly Report.
“ED visits related to suspected or confirmed child abuse and neglect decreased beginning the week of March 15, 2020, coinciding with the declaration of a national emergency related to COVID-19 and implementation of community mitigation measures,” they wrote.
An earlier study involving the same database (the National Syndromic Surveillance Program) showed that, over the two same 4-week periods, the volume of all ED visits in 2020 was down 72% for children aged 10 years and younger and 71% for those aged 11-14 years.
In the current study, however, all age subgroups had significant increases in hospital admissions. The proportion of ED visits related to child abuse and neglect that resulted in hospitalization rose from 3.5% in 2019 to 5.3% in 2020 among ages 0-4 years, 0.7% to 1.3% for ages 5-11 years, and 1.6% to 2.2% for adolescents aged 12-17, Dr. Swedo and associates reported.
The absence of a corresponding drop in hospitalizations may be tied to risk factors related to the pandemic, “such as loss of income, increased stress related to parental child care and schooling responsibilities, and increased substance use and mental health conditions among adults,” the investigators added.
The National Syndromic Surveillance Program receives daily data from 3,310 EDs in 47 states, but the number of facilities meeting the investigators’ criteria averaged 2,970 a week for the 8 weeks of the study period.
SOURCE: Swedo E et al. MMWR. 2020 Dec. 11;69(49):1841-7.
FROM MMWR
COVID-19 anticoagulation trials ‘paused’ for futility, safety
Parts of three linked studies investigating increased levels of anticoagulation in hospitalized COVID-19 patients have been “paused” because of futility and safety concerns, a statement from the U.S. National Heart, Lung, and Blood Institute (NHLBI) confirms.
The trials involved are the REMAP-CAP, ACTIV-4, and ATTACC studies.
The statement also says that a potential for harm in this subgroup could not be excluded, noting that increased bleeding is a known complication of full-dose anticoagulation. The trials are working urgently to undertake additional analyses, which will be made available as soon as possible.
The three clinical trial platforms are working together to test the effects of full therapeutic doses of anticoagulants vs. lower prophylactic doses in COVID-19 patients.
Informed by the deliberations of the data safety monitoring boards of these trials, all of the trial sites have paused enrollment of the most critically ill hospitalized patients with COVID-19.
Enrollment continues in the trials for moderately ill hospitalized COVID-19 patients, the statement notes.
“Whether the use of full-dose compared to low-dose anticoagulants leads to better outcomes in hospitalized patients with less COVID-19 severe disease remains a very important question,” the NHLBI statement says.
Patients who require full dose anticoagulants for another medical indication are not included in these trials.
The statement explains that COVID-19 is associated with significant inflammation and clinical and pathologic evidence of widespread blood clots. These trials were launched because clinicians have observed that many patients ill with COVID-19, including those who have died from the disease, formed blood clots throughout their bodies, even in their smallest blood vessels. This unusual clotting can cause multiple health complications, including lung failure, myocardial infarction, and stroke.
The three trials are the result of a collaboration between major international partners. The trials include: the Randomized, Embedded, Multi-factorial Adaptive Platform Trial for Community-Acquired Pneumonia (REMAP-CAP) Therapeutic Anticoagulation; Accelerating COVID-19 Therapeutic Interventions and Vaccines-4 (ACTIV-4) Antithrombotics Inpatient; and Antithrombotic Therapy to Ameliorate Complications of COVID-19 (ATTACC).
The trials, which span four continents, have the common goal of assessing the benefit of full doses of anticoagulants to treat moderately ill or critically ill adults hospitalized for COVID-19, compared with a lower dose often used to prevent blood clots in hospitalized patients.
In the United States, the ACTIV-4 trial is being led by a collaborative effort involving a number of universities, including the University of Pittsburgh and New York University.
The trials are supported by multiple international funding organizations including the National Institutes of Health, Canadian Institutes of Health Research, the National Institute for Health Research (UK), the National Health and Medical Research Council (Australia), and the PREPARE and RECOVER consortia (European Union).
A version of this story first appeared on Medscape.com.
Parts of three linked studies investigating increased levels of anticoagulation in hospitalized COVID-19 patients have been “paused” because of futility and safety concerns, a statement from the U.S. National Heart, Lung, and Blood Institute (NHLBI) confirms.
The trials involved are the REMAP-CAP, ACTIV-4, and ATTACC studies.
The statement also says that a potential for harm in this subgroup could not be excluded, noting that increased bleeding is a known complication of full-dose anticoagulation. The trials are working urgently to undertake additional analyses, which will be made available as soon as possible.
The three clinical trial platforms are working together to test the effects of full therapeutic doses of anticoagulants vs. lower prophylactic doses in COVID-19 patients.
Informed by the deliberations of the data safety monitoring boards of these trials, all of the trial sites have paused enrollment of the most critically ill hospitalized patients with COVID-19.
Enrollment continues in the trials for moderately ill hospitalized COVID-19 patients, the statement notes.
“Whether the use of full-dose compared to low-dose anticoagulants leads to better outcomes in hospitalized patients with less COVID-19 severe disease remains a very important question,” the NHLBI statement says.
Patients who require full dose anticoagulants for another medical indication are not included in these trials.
The statement explains that COVID-19 is associated with significant inflammation and clinical and pathologic evidence of widespread blood clots. These trials were launched because clinicians have observed that many patients ill with COVID-19, including those who have died from the disease, formed blood clots throughout their bodies, even in their smallest blood vessels. This unusual clotting can cause multiple health complications, including lung failure, myocardial infarction, and stroke.
The three trials are the result of a collaboration between major international partners. The trials include: the Randomized, Embedded, Multi-factorial Adaptive Platform Trial for Community-Acquired Pneumonia (REMAP-CAP) Therapeutic Anticoagulation; Accelerating COVID-19 Therapeutic Interventions and Vaccines-4 (ACTIV-4) Antithrombotics Inpatient; and Antithrombotic Therapy to Ameliorate Complications of COVID-19 (ATTACC).
The trials, which span four continents, have the common goal of assessing the benefit of full doses of anticoagulants to treat moderately ill or critically ill adults hospitalized for COVID-19, compared with a lower dose often used to prevent blood clots in hospitalized patients.
In the United States, the ACTIV-4 trial is being led by a collaborative effort involving a number of universities, including the University of Pittsburgh and New York University.
The trials are supported by multiple international funding organizations including the National Institutes of Health, Canadian Institutes of Health Research, the National Institute for Health Research (UK), the National Health and Medical Research Council (Australia), and the PREPARE and RECOVER consortia (European Union).
A version of this story first appeared on Medscape.com.
Parts of three linked studies investigating increased levels of anticoagulation in hospitalized COVID-19 patients have been “paused” because of futility and safety concerns, a statement from the U.S. National Heart, Lung, and Blood Institute (NHLBI) confirms.
The trials involved are the REMAP-CAP, ACTIV-4, and ATTACC studies.
The statement also says that a potential for harm in this subgroup could not be excluded, noting that increased bleeding is a known complication of full-dose anticoagulation. The trials are working urgently to undertake additional analyses, which will be made available as soon as possible.
The three clinical trial platforms are working together to test the effects of full therapeutic doses of anticoagulants vs. lower prophylactic doses in COVID-19 patients.
Informed by the deliberations of the data safety monitoring boards of these trials, all of the trial sites have paused enrollment of the most critically ill hospitalized patients with COVID-19.
Enrollment continues in the trials for moderately ill hospitalized COVID-19 patients, the statement notes.
“Whether the use of full-dose compared to low-dose anticoagulants leads to better outcomes in hospitalized patients with less COVID-19 severe disease remains a very important question,” the NHLBI statement says.
Patients who require full dose anticoagulants for another medical indication are not included in these trials.
The statement explains that COVID-19 is associated with significant inflammation and clinical and pathologic evidence of widespread blood clots. These trials were launched because clinicians have observed that many patients ill with COVID-19, including those who have died from the disease, formed blood clots throughout their bodies, even in their smallest blood vessels. This unusual clotting can cause multiple health complications, including lung failure, myocardial infarction, and stroke.
The three trials are the result of a collaboration between major international partners. The trials include: the Randomized, Embedded, Multi-factorial Adaptive Platform Trial for Community-Acquired Pneumonia (REMAP-CAP) Therapeutic Anticoagulation; Accelerating COVID-19 Therapeutic Interventions and Vaccines-4 (ACTIV-4) Antithrombotics Inpatient; and Antithrombotic Therapy to Ameliorate Complications of COVID-19 (ATTACC).
The trials, which span four continents, have the common goal of assessing the benefit of full doses of anticoagulants to treat moderately ill or critically ill adults hospitalized for COVID-19, compared with a lower dose often used to prevent blood clots in hospitalized patients.
In the United States, the ACTIV-4 trial is being led by a collaborative effort involving a number of universities, including the University of Pittsburgh and New York University.
The trials are supported by multiple international funding organizations including the National Institutes of Health, Canadian Institutes of Health Research, the National Institute for Health Research (UK), the National Health and Medical Research Council (Australia), and the PREPARE and RECOVER consortia (European Union).
A version of this story first appeared on Medscape.com.
Latest rise in child COVID-19 cases is relatively small
For the seventh week out of the last eight, more new cases of COVID-19 in children were reported in the United States than any week before, according to a report from the American Academy of Pediatrics and the Children’s Hospital Association.
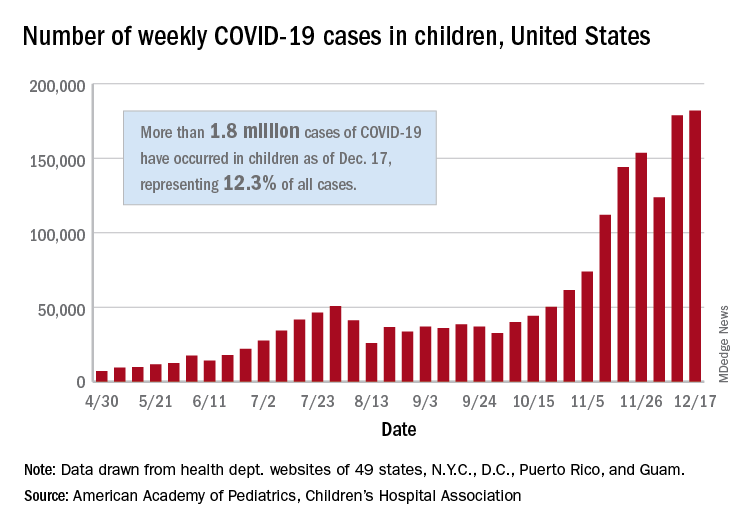
There were just over 182,000 new cases of COVID-19 in children during the week ending Dec. 17, topping the previous high of almost 179,000 set the previous week. – a stretch of 11 weeks that has produced only one decline, based on data from the latest AAP/CHA weekly report.
As of Dec. 17, there had been over 1.8 million cases of COVID-19 in children, which represents 12.3% of all U.S. cases. For the week, 14% of all cases occurred in children, which was up slightly from 13.8% the week before (Dec. 10). The overall rate of coronavirus infection is now 2,420 cases per 100,000 children in the population, the AAP and CHA said.
A total of 30 states are above that national rate, with North Dakota the highest at 7,515 cases per 100,000 children, followed by South Dakota (5,618), Wyoming (5,157), Wisconsin (5,106), and Tennessee (4,994). Wyoming has the highest proportion of cases occurring in children at 20.8%, but that is down from 23.4% in mid-November, based on data collected by the AAP and CHA from the health department websites of 49 states (New York does not provide age distributions), the District of Columbia, New York City, Puerto Rico, and Guam.
In the last 2 weeks, however, the largest percent increases in new cases came in states with low-to-average rates of cumulative child infection. California, Connecticut, Delaware, Maine, Maryland, New Hampshire, and Vermont all saw increases of over 35% from Dec. 3 to Dec. 17, while the smallest increases occurred in Hawaii, North Dakota, and Wyoming, the AAP and CHA reported.
For the seventh week out of the last eight, more new cases of COVID-19 in children were reported in the United States than any week before, according to a report from the American Academy of Pediatrics and the Children’s Hospital Association.

There were just over 182,000 new cases of COVID-19 in children during the week ending Dec. 17, topping the previous high of almost 179,000 set the previous week. – a stretch of 11 weeks that has produced only one decline, based on data from the latest AAP/CHA weekly report.
As of Dec. 17, there had been over 1.8 million cases of COVID-19 in children, which represents 12.3% of all U.S. cases. For the week, 14% of all cases occurred in children, which was up slightly from 13.8% the week before (Dec. 10). The overall rate of coronavirus infection is now 2,420 cases per 100,000 children in the population, the AAP and CHA said.
A total of 30 states are above that national rate, with North Dakota the highest at 7,515 cases per 100,000 children, followed by South Dakota (5,618), Wyoming (5,157), Wisconsin (5,106), and Tennessee (4,994). Wyoming has the highest proportion of cases occurring in children at 20.8%, but that is down from 23.4% in mid-November, based on data collected by the AAP and CHA from the health department websites of 49 states (New York does not provide age distributions), the District of Columbia, New York City, Puerto Rico, and Guam.
In the last 2 weeks, however, the largest percent increases in new cases came in states with low-to-average rates of cumulative child infection. California, Connecticut, Delaware, Maine, Maryland, New Hampshire, and Vermont all saw increases of over 35% from Dec. 3 to Dec. 17, while the smallest increases occurred in Hawaii, North Dakota, and Wyoming, the AAP and CHA reported.
For the seventh week out of the last eight, more new cases of COVID-19 in children were reported in the United States than any week before, according to a report from the American Academy of Pediatrics and the Children’s Hospital Association.

There were just over 182,000 new cases of COVID-19 in children during the week ending Dec. 17, topping the previous high of almost 179,000 set the previous week. – a stretch of 11 weeks that has produced only one decline, based on data from the latest AAP/CHA weekly report.
As of Dec. 17, there had been over 1.8 million cases of COVID-19 in children, which represents 12.3% of all U.S. cases. For the week, 14% of all cases occurred in children, which was up slightly from 13.8% the week before (Dec. 10). The overall rate of coronavirus infection is now 2,420 cases per 100,000 children in the population, the AAP and CHA said.
A total of 30 states are above that national rate, with North Dakota the highest at 7,515 cases per 100,000 children, followed by South Dakota (5,618), Wyoming (5,157), Wisconsin (5,106), and Tennessee (4,994). Wyoming has the highest proportion of cases occurring in children at 20.8%, but that is down from 23.4% in mid-November, based on data collected by the AAP and CHA from the health department websites of 49 states (New York does not provide age distributions), the District of Columbia, New York City, Puerto Rico, and Guam.
In the last 2 weeks, however, the largest percent increases in new cases came in states with low-to-average rates of cumulative child infection. California, Connecticut, Delaware, Maine, Maryland, New Hampshire, and Vermont all saw increases of over 35% from Dec. 3 to Dec. 17, while the smallest increases occurred in Hawaii, North Dakota, and Wyoming, the AAP and CHA reported.