User login
Drug receives orphan designation for MM
The US Food and Drug Administration (FDA) has granted orphan drug designation to SRF231 for the treatment of multiple myeloma (MM).
SRF231 is a fully human antibody that inhibits the activity of CD47, a protein that is overexpressed on many cancer cells and prevents them from being engulfed and eliminated by macrophages.
Surface Oncology, the company developing SRF231, is currently conducting a phase 1 trial (NCT03512340) of SRF231 in patients with solid tumors and hematologic malignancies.
Preclinical research on SRF231 was presented at the 2016 ASH Annual Meeting.
SRF231 demonstrated “potent” activity against hematologic malignancies, according to researchers.
The team said SRF231 promoted macrophage-mediated phagocytic clearance of several hematologic primary tumor samples and cell lines in vitro.
SRF231 also demonstrated activity in murine xenograft models of hematologic malignancies. Specifically, the researchers observed “profound tumor growth inhibition” in models of MM, diffuse large B-cell lymphoma, and Burkitt lymphoma.
The team said SRF231 demonstrated activity when given alone or in combination with opsonizing antibodies.
Results also showed that SRF231 did not induce hemagglutination or phagocytosis of red blood cells in vitro.
About orphan designation
The FDA grants orphan designation to products intended to treat, diagnose, or prevent diseases/disorders that affect fewer than 200,000 people in the US.
Orphan designation provides incentives for sponsors to develop products for rare diseases. This may include tax credits toward the cost of clinical trials, prescription drug user fee waivers, and 7 years of market exclusivity if the product is approved.
The US Food and Drug Administration (FDA) has granted orphan drug designation to SRF231 for the treatment of multiple myeloma (MM).
SRF231 is a fully human antibody that inhibits the activity of CD47, a protein that is overexpressed on many cancer cells and prevents them from being engulfed and eliminated by macrophages.
Surface Oncology, the company developing SRF231, is currently conducting a phase 1 trial (NCT03512340) of SRF231 in patients with solid tumors and hematologic malignancies.
Preclinical research on SRF231 was presented at the 2016 ASH Annual Meeting.
SRF231 demonstrated “potent” activity against hematologic malignancies, according to researchers.
The team said SRF231 promoted macrophage-mediated phagocytic clearance of several hematologic primary tumor samples and cell lines in vitro.
SRF231 also demonstrated activity in murine xenograft models of hematologic malignancies. Specifically, the researchers observed “profound tumor growth inhibition” in models of MM, diffuse large B-cell lymphoma, and Burkitt lymphoma.
The team said SRF231 demonstrated activity when given alone or in combination with opsonizing antibodies.
Results also showed that SRF231 did not induce hemagglutination or phagocytosis of red blood cells in vitro.
About orphan designation
The FDA grants orphan designation to products intended to treat, diagnose, or prevent diseases/disorders that affect fewer than 200,000 people in the US.
Orphan designation provides incentives for sponsors to develop products for rare diseases. This may include tax credits toward the cost of clinical trials, prescription drug user fee waivers, and 7 years of market exclusivity if the product is approved.
The US Food and Drug Administration (FDA) has granted orphan drug designation to SRF231 for the treatment of multiple myeloma (MM).
SRF231 is a fully human antibody that inhibits the activity of CD47, a protein that is overexpressed on many cancer cells and prevents them from being engulfed and eliminated by macrophages.
Surface Oncology, the company developing SRF231, is currently conducting a phase 1 trial (NCT03512340) of SRF231 in patients with solid tumors and hematologic malignancies.
Preclinical research on SRF231 was presented at the 2016 ASH Annual Meeting.
SRF231 demonstrated “potent” activity against hematologic malignancies, according to researchers.
The team said SRF231 promoted macrophage-mediated phagocytic clearance of several hematologic primary tumor samples and cell lines in vitro.
SRF231 also demonstrated activity in murine xenograft models of hematologic malignancies. Specifically, the researchers observed “profound tumor growth inhibition” in models of MM, diffuse large B-cell lymphoma, and Burkitt lymphoma.
The team said SRF231 demonstrated activity when given alone or in combination with opsonizing antibodies.
Results also showed that SRF231 did not induce hemagglutination or phagocytosis of red blood cells in vitro.
About orphan designation
The FDA grants orphan designation to products intended to treat, diagnose, or prevent diseases/disorders that affect fewer than 200,000 people in the US.
Orphan designation provides incentives for sponsors to develop products for rare diseases. This may include tax credits toward the cost of clinical trials, prescription drug user fee waivers, and 7 years of market exclusivity if the product is approved.
CHMP supports expanded approval for epoetin alfa products
The European Medicines Agency’s Committee for Medicinal Products for Human Use (CHMP) has recommended a change to the terms of marketing authorization for 3 versions of epoetin alfa—Abseamed, Binocrit, and Epoetin alfa Hexal.
The CHMP is recommending that all 3 products be approved to treat symptomatic anemia (hemoglobin concentration of ≤ 10 g/dL) in adults with low- or intermediate-1-risk primary myelodysplastic syndromes who have low serum erythropoietin (< 200 mU/mL).
The CHMP’s recommendation will be reviewed by the European Commission (EC), which has the authority to approve medicines for use in the European Union, Norway, Iceland, and Liechtenstein.
The EC usually makes a decision within 67 days of the CHMP’s recommendation.
Abseamed, Binocrit, and Epoetin alfa Hexal are already EC-approved for the following indications:
- To treat symptomatic anemia associated with chronic renal failure in adults and children ages 1 to 18 on hemodialysis and adults on peritoneal dialysis
- To treat symptomatic anemia associated with chronic renal failure in adults with renal insufficiency not yet undergoing dialysis for the treatment of severe anemia of renal origin accompanied by clinical symptoms in patients
- For adults receiving chemotherapy for solid tumors, malignant lymphoma, or multiple myeloma who are at risk of transfusion as assessed by the patient’s general status (eg, cardiovascular status, pre-existing anemia at the start of chemotherapy) for the treatment of anemia and reduction of transfusion requirements
- For adults in a predonation program to increase the yield of autologous blood; treatment should only be given to patients with moderate anemia (hemoglobin concentration range between 10 to 13 g/dL [6.2 to 8.1 mmol/L], no iron deficiency) if blood-saving procedures are not available or insufficient when the scheduled major elective surgery requires a large volume of blood (4 or more units for females or 5 or more units for males)
- For non-iron-deficient adults set to undergo major elective orthopedic surgery who have a high perceived risk for transfusion complications to reduce exposure to allogeneic blood transfusions; use should be restricted to patients with moderate anemia (eg, hemoglobin concentration range between 10 to 13 g/dL or 6.2 to 8.1 mmol/L) who do not have an autologous predonation program available and with expected moderate blood loss (900 to 1800 mL).
The marketing authorization holders are Medice Arzneimittel Pütter GmbH & Co. KG for Abseamed, Sandoz GmbH for Binocrit, and Hexal AG for Epoetin alfa Hexal.
The European Medicines Agency’s Committee for Medicinal Products for Human Use (CHMP) has recommended a change to the terms of marketing authorization for 3 versions of epoetin alfa—Abseamed, Binocrit, and Epoetin alfa Hexal.
The CHMP is recommending that all 3 products be approved to treat symptomatic anemia (hemoglobin concentration of ≤ 10 g/dL) in adults with low- or intermediate-1-risk primary myelodysplastic syndromes who have low serum erythropoietin (< 200 mU/mL).
The CHMP’s recommendation will be reviewed by the European Commission (EC), which has the authority to approve medicines for use in the European Union, Norway, Iceland, and Liechtenstein.
The EC usually makes a decision within 67 days of the CHMP’s recommendation.
Abseamed, Binocrit, and Epoetin alfa Hexal are already EC-approved for the following indications:
- To treat symptomatic anemia associated with chronic renal failure in adults and children ages 1 to 18 on hemodialysis and adults on peritoneal dialysis
- To treat symptomatic anemia associated with chronic renal failure in adults with renal insufficiency not yet undergoing dialysis for the treatment of severe anemia of renal origin accompanied by clinical symptoms in patients
- For adults receiving chemotherapy for solid tumors, malignant lymphoma, or multiple myeloma who are at risk of transfusion as assessed by the patient’s general status (eg, cardiovascular status, pre-existing anemia at the start of chemotherapy) for the treatment of anemia and reduction of transfusion requirements
- For adults in a predonation program to increase the yield of autologous blood; treatment should only be given to patients with moderate anemia (hemoglobin concentration range between 10 to 13 g/dL [6.2 to 8.1 mmol/L], no iron deficiency) if blood-saving procedures are not available or insufficient when the scheduled major elective surgery requires a large volume of blood (4 or more units for females or 5 or more units for males)
- For non-iron-deficient adults set to undergo major elective orthopedic surgery who have a high perceived risk for transfusion complications to reduce exposure to allogeneic blood transfusions; use should be restricted to patients with moderate anemia (eg, hemoglobin concentration range between 10 to 13 g/dL or 6.2 to 8.1 mmol/L) who do not have an autologous predonation program available and with expected moderate blood loss (900 to 1800 mL).
The marketing authorization holders are Medice Arzneimittel Pütter GmbH & Co. KG for Abseamed, Sandoz GmbH for Binocrit, and Hexal AG for Epoetin alfa Hexal.
The European Medicines Agency’s Committee for Medicinal Products for Human Use (CHMP) has recommended a change to the terms of marketing authorization for 3 versions of epoetin alfa—Abseamed, Binocrit, and Epoetin alfa Hexal.
The CHMP is recommending that all 3 products be approved to treat symptomatic anemia (hemoglobin concentration of ≤ 10 g/dL) in adults with low- or intermediate-1-risk primary myelodysplastic syndromes who have low serum erythropoietin (< 200 mU/mL).
The CHMP’s recommendation will be reviewed by the European Commission (EC), which has the authority to approve medicines for use in the European Union, Norway, Iceland, and Liechtenstein.
The EC usually makes a decision within 67 days of the CHMP’s recommendation.
Abseamed, Binocrit, and Epoetin alfa Hexal are already EC-approved for the following indications:
- To treat symptomatic anemia associated with chronic renal failure in adults and children ages 1 to 18 on hemodialysis and adults on peritoneal dialysis
- To treat symptomatic anemia associated with chronic renal failure in adults with renal insufficiency not yet undergoing dialysis for the treatment of severe anemia of renal origin accompanied by clinical symptoms in patients
- For adults receiving chemotherapy for solid tumors, malignant lymphoma, or multiple myeloma who are at risk of transfusion as assessed by the patient’s general status (eg, cardiovascular status, pre-existing anemia at the start of chemotherapy) for the treatment of anemia and reduction of transfusion requirements
- For adults in a predonation program to increase the yield of autologous blood; treatment should only be given to patients with moderate anemia (hemoglobin concentration range between 10 to 13 g/dL [6.2 to 8.1 mmol/L], no iron deficiency) if blood-saving procedures are not available or insufficient when the scheduled major elective surgery requires a large volume of blood (4 or more units for females or 5 or more units for males)
- For non-iron-deficient adults set to undergo major elective orthopedic surgery who have a high perceived risk for transfusion complications to reduce exposure to allogeneic blood transfusions; use should be restricted to patients with moderate anemia (eg, hemoglobin concentration range between 10 to 13 g/dL or 6.2 to 8.1 mmol/L) who do not have an autologous predonation program available and with expected moderate blood loss (900 to 1800 mL).
The marketing authorization holders are Medice Arzneimittel Pütter GmbH & Co. KG for Abseamed, Sandoz GmbH for Binocrit, and Hexal AG for Epoetin alfa Hexal.
Drug receives fast track designation for WM
The US Food and Drug Administration (FDA) has granted fast track designation to zanubrutinib for the treatment of Waldenström’s macroglobulinemia (WM).
Zanubrutinib (BGB-3111) is a BTK inhibitor being developed by BeiGene to treat various B-cell malignancies.
BeiGene is preparing to submit to the FDA, in the first half of 2019, a new drug application seeking accelerated approval of zanubrutinib for patients with WM.
The application will be supported by results from a phase 1 study. Results from this trial were presented at the 14th International Conference on Malignant Lymphoma (14-ICML) last year.
Researchers are also evaluating zanubrutinib in phase 2 (NCT03332173) and phase 3 (NCT03053440) trials of WM patients. In the phase 3 trial, researchers are comparing zanubrutinib to the BTK inhibitor ibrutinib.
Phase 1 results
As of March 31, 2017, 48 WM patients were enrolled in the phase 1 study. Thirty-eight patients had relapsed/refractory disease, and 10 patients were treatment-naïve.
There was a dose-escalation phase and a dose-expansion phase. The dose-expansion phase included doses of 160 mg twice a day or 320 mg once a day.
The most common (>10%) adverse events, (AEs) of any attribution were petechiae/purpura/contusion (35%), upper respiratory tract infection (31%), constipation (25%), diarrhea (19%), epistaxis (19%), nausea (17%), cough (15%), anemia (15%), headache (15%), neutropenia (13%), and rash (13%).
Most of these events were grade 1 or 2 in severity. The exceptions were grade 3/4 anemia and neutropenia (8% each) as well as grade 3/4 diarrhea and headache (2% each).
Five serious AEs were considered possibly related to zanubrutinib—1 case each of hemothorax, atrial fibrillation, colitis, febrile neutropenia, and headache. Three AEs led to treatment discontinuation—1 case each of bronchiectasis, prostate adenocarcinoma, and adenocarcinoma of pylorus.
At the time of the data cutoff, 42 patients were evaluable for response. At a median follow-up of 12.3 months (range, 4.4 to 30.5 months), the overall response rate was 90% (38/42).
The major response rate was 76% (32/42), with very good partial responses in 43% (18/42) of patients and partial responses in 33% (14/42) of patients. There were no complete responses and 2 cases of disease progression.
About fast track designation
The FDA’s fast track development program is designed to expedite clinical development and submission of applications for products with the potential to treat serious or life-threatening conditions and address unmet medical needs.
Fast track designation facilitates frequent interactions with the FDA review team, including meetings to discuss the product’s development plan and written communications about issues such as trial design and use of biomarkers.
Products that receive fast track designation may be eligible for accelerated approval and priority review if relevant criteria are met. Such products may also be eligible for rolling review, which allows a developer to submit individual sections of a product’s application for review as they are ready, rather than waiting until all sections are complete.
The US Food and Drug Administration (FDA) has granted fast track designation to zanubrutinib for the treatment of Waldenström’s macroglobulinemia (WM).
Zanubrutinib (BGB-3111) is a BTK inhibitor being developed by BeiGene to treat various B-cell malignancies.
BeiGene is preparing to submit to the FDA, in the first half of 2019, a new drug application seeking accelerated approval of zanubrutinib for patients with WM.
The application will be supported by results from a phase 1 study. Results from this trial were presented at the 14th International Conference on Malignant Lymphoma (14-ICML) last year.
Researchers are also evaluating zanubrutinib in phase 2 (NCT03332173) and phase 3 (NCT03053440) trials of WM patients. In the phase 3 trial, researchers are comparing zanubrutinib to the BTK inhibitor ibrutinib.
Phase 1 results
As of March 31, 2017, 48 WM patients were enrolled in the phase 1 study. Thirty-eight patients had relapsed/refractory disease, and 10 patients were treatment-naïve.
There was a dose-escalation phase and a dose-expansion phase. The dose-expansion phase included doses of 160 mg twice a day or 320 mg once a day.
The most common (>10%) adverse events, (AEs) of any attribution were petechiae/purpura/contusion (35%), upper respiratory tract infection (31%), constipation (25%), diarrhea (19%), epistaxis (19%), nausea (17%), cough (15%), anemia (15%), headache (15%), neutropenia (13%), and rash (13%).
Most of these events were grade 1 or 2 in severity. The exceptions were grade 3/4 anemia and neutropenia (8% each) as well as grade 3/4 diarrhea and headache (2% each).
Five serious AEs were considered possibly related to zanubrutinib—1 case each of hemothorax, atrial fibrillation, colitis, febrile neutropenia, and headache. Three AEs led to treatment discontinuation—1 case each of bronchiectasis, prostate adenocarcinoma, and adenocarcinoma of pylorus.
At the time of the data cutoff, 42 patients were evaluable for response. At a median follow-up of 12.3 months (range, 4.4 to 30.5 months), the overall response rate was 90% (38/42).
The major response rate was 76% (32/42), with very good partial responses in 43% (18/42) of patients and partial responses in 33% (14/42) of patients. There were no complete responses and 2 cases of disease progression.
About fast track designation
The FDA’s fast track development program is designed to expedite clinical development and submission of applications for products with the potential to treat serious or life-threatening conditions and address unmet medical needs.
Fast track designation facilitates frequent interactions with the FDA review team, including meetings to discuss the product’s development plan and written communications about issues such as trial design and use of biomarkers.
Products that receive fast track designation may be eligible for accelerated approval and priority review if relevant criteria are met. Such products may also be eligible for rolling review, which allows a developer to submit individual sections of a product’s application for review as they are ready, rather than waiting until all sections are complete.
The US Food and Drug Administration (FDA) has granted fast track designation to zanubrutinib for the treatment of Waldenström’s macroglobulinemia (WM).
Zanubrutinib (BGB-3111) is a BTK inhibitor being developed by BeiGene to treat various B-cell malignancies.
BeiGene is preparing to submit to the FDA, in the first half of 2019, a new drug application seeking accelerated approval of zanubrutinib for patients with WM.
The application will be supported by results from a phase 1 study. Results from this trial were presented at the 14th International Conference on Malignant Lymphoma (14-ICML) last year.
Researchers are also evaluating zanubrutinib in phase 2 (NCT03332173) and phase 3 (NCT03053440) trials of WM patients. In the phase 3 trial, researchers are comparing zanubrutinib to the BTK inhibitor ibrutinib.
Phase 1 results
As of March 31, 2017, 48 WM patients were enrolled in the phase 1 study. Thirty-eight patients had relapsed/refractory disease, and 10 patients were treatment-naïve.
There was a dose-escalation phase and a dose-expansion phase. The dose-expansion phase included doses of 160 mg twice a day or 320 mg once a day.
The most common (>10%) adverse events, (AEs) of any attribution were petechiae/purpura/contusion (35%), upper respiratory tract infection (31%), constipation (25%), diarrhea (19%), epistaxis (19%), nausea (17%), cough (15%), anemia (15%), headache (15%), neutropenia (13%), and rash (13%).
Most of these events were grade 1 or 2 in severity. The exceptions were grade 3/4 anemia and neutropenia (8% each) as well as grade 3/4 diarrhea and headache (2% each).
Five serious AEs were considered possibly related to zanubrutinib—1 case each of hemothorax, atrial fibrillation, colitis, febrile neutropenia, and headache. Three AEs led to treatment discontinuation—1 case each of bronchiectasis, prostate adenocarcinoma, and adenocarcinoma of pylorus.
At the time of the data cutoff, 42 patients were evaluable for response. At a median follow-up of 12.3 months (range, 4.4 to 30.5 months), the overall response rate was 90% (38/42).
The major response rate was 76% (32/42), with very good partial responses in 43% (18/42) of patients and partial responses in 33% (14/42) of patients. There were no complete responses and 2 cases of disease progression.
About fast track designation
The FDA’s fast track development program is designed to expedite clinical development and submission of applications for products with the potential to treat serious or life-threatening conditions and address unmet medical needs.
Fast track designation facilitates frequent interactions with the FDA review team, including meetings to discuss the product’s development plan and written communications about issues such as trial design and use of biomarkers.
Products that receive fast track designation may be eligible for accelerated approval and priority review if relevant criteria are met. Such products may also be eligible for rolling review, which allows a developer to submit individual sections of a product’s application for review as they are ready, rather than waiting until all sections are complete.
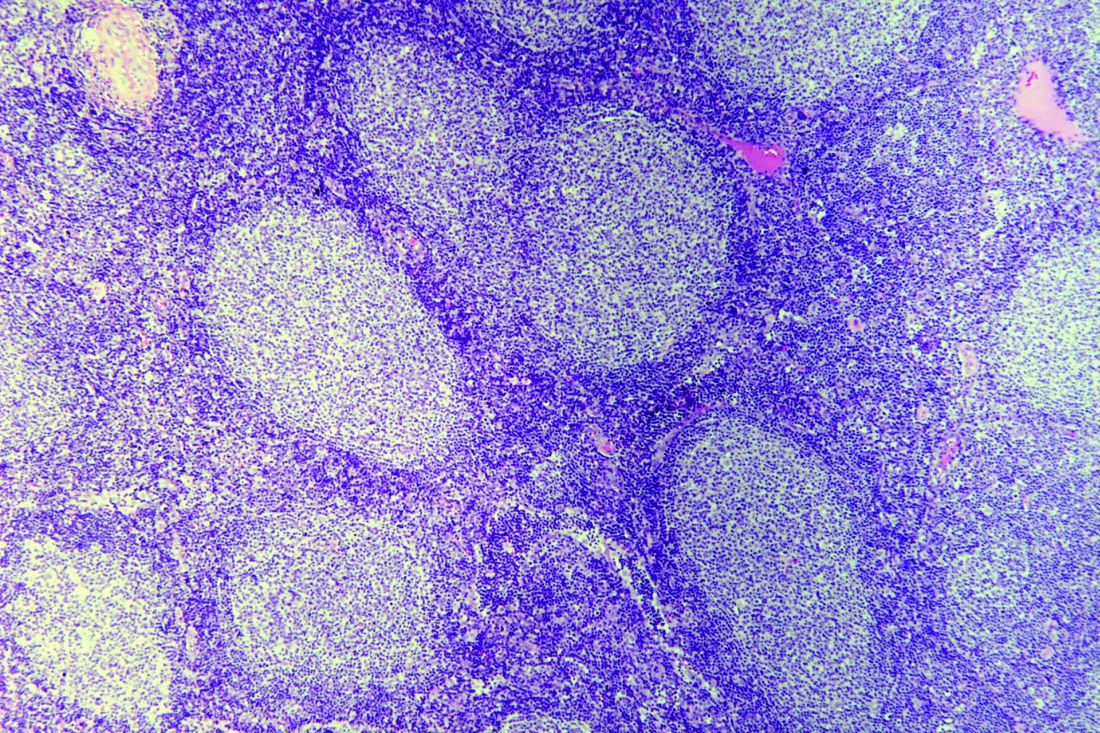
Rituximab reduces risk of follicular lymphoma transformation
Rituximab-based chemotherapy can significantly reduce the risk of transformation of follicular lymphoma (FL) from an indolent to an aggressive histology, such as diffuse large B-cell lymphoma, results of a retrospective pooled analysis have suggested.
“Despite the intrinsic limitations related to the retrospective nature of our study, we confirmed that the cumulative hazard of histological transformation as a first event in follicular lymphoma can be reduced significantly by introducing rituximab to a backbone therapy. Moreover, our data also confirm that histological transformation still has an adverse effect on patient outcome, although it is less catastrophic than the pre-rituximab regimens,” they wrote in the Lancet Haematology.
These investigators, from 11 cooperative groups or institutions across Europe, pooled data on patients aged 18 years and older who had a histologically confirmed diagnosis of grade 1, 2, or 3a FL between Jan. 2, 1997, and Dec. 20, 2013.
They defined histologic transformation as a biopsy-proven aggressive lymphoma that occurred as a first event after first-line therapy.
Data on a total of 8,116 patients were available for analysis; 509 of these patients had had histologic transformations. After a median follow-up of 87 months, the 10-year cumulative hazard for all patients was 7.7%. The 10-year cumulative hazard – one of two primary endpoints – was 5.2% for patients who had received any rituximab versus 8.7% for those who did not, which translated into a hazard ratio of 0.73 (P = .004).
Among patients who received rituximab during induction only, the 10-year cumulative hazard was 5.9%, and it was 3.6% among those who received rituximab during induction and maintenance phases of treatment. This difference translated into a HR of 0.55 (P = .003).
The benefit of rituximab induction and maintenance – compared with induction only – held up in a multivariate analysis controlling for age at diagnosis, sex, FLIPI (Follicular Lymphoma International Prognostic Index) score, active surveillance vs. treatment, and FL grade (HR, 0.55; P = .016).
There were 287 deaths among the 509 patients with transformation, resulting in a 10-year survival after transformation of 32%.
The 5-year survival after transformation was 38% for patients who were not exposed to rituximab, 42% for patients who received induction rituximab, and 43% for those who received both induction and maintenance rituximab, but the differences between the three groups were not statistically significant.
“More comprehensive knowledge of the biological risk factors for follicular lymphoma transformation and the molecular pathways involved is likely to help clinicians make more accurate prognostic assessments and also inform the potential usefulness of novel drugs for the treatment of follicular lymphoma,” the researchers wrote.
The study was funded by the European Lymphoma Institute and other research groups. The researchers reported having no financial disclosures.
SOURCE: Federico M et al. Lancet Haematol. 2018 Jul 4. doi: 10.1016/S2352-3026(18)30090-5.
Rituximab-based chemotherapy can significantly reduce the risk of transformation of follicular lymphoma (FL) from an indolent to an aggressive histology, such as diffuse large B-cell lymphoma, results of a retrospective pooled analysis have suggested.
“Despite the intrinsic limitations related to the retrospective nature of our study, we confirmed that the cumulative hazard of histological transformation as a first event in follicular lymphoma can be reduced significantly by introducing rituximab to a backbone therapy. Moreover, our data also confirm that histological transformation still has an adverse effect on patient outcome, although it is less catastrophic than the pre-rituximab regimens,” they wrote in the Lancet Haematology.
These investigators, from 11 cooperative groups or institutions across Europe, pooled data on patients aged 18 years and older who had a histologically confirmed diagnosis of grade 1, 2, or 3a FL between Jan. 2, 1997, and Dec. 20, 2013.
They defined histologic transformation as a biopsy-proven aggressive lymphoma that occurred as a first event after first-line therapy.
Data on a total of 8,116 patients were available for analysis; 509 of these patients had had histologic transformations. After a median follow-up of 87 months, the 10-year cumulative hazard for all patients was 7.7%. The 10-year cumulative hazard – one of two primary endpoints – was 5.2% for patients who had received any rituximab versus 8.7% for those who did not, which translated into a hazard ratio of 0.73 (P = .004).
Among patients who received rituximab during induction only, the 10-year cumulative hazard was 5.9%, and it was 3.6% among those who received rituximab during induction and maintenance phases of treatment. This difference translated into a HR of 0.55 (P = .003).
The benefit of rituximab induction and maintenance – compared with induction only – held up in a multivariate analysis controlling for age at diagnosis, sex, FLIPI (Follicular Lymphoma International Prognostic Index) score, active surveillance vs. treatment, and FL grade (HR, 0.55; P = .016).
There were 287 deaths among the 509 patients with transformation, resulting in a 10-year survival after transformation of 32%.
The 5-year survival after transformation was 38% for patients who were not exposed to rituximab, 42% for patients who received induction rituximab, and 43% for those who received both induction and maintenance rituximab, but the differences between the three groups were not statistically significant.
“More comprehensive knowledge of the biological risk factors for follicular lymphoma transformation and the molecular pathways involved is likely to help clinicians make more accurate prognostic assessments and also inform the potential usefulness of novel drugs for the treatment of follicular lymphoma,” the researchers wrote.
The study was funded by the European Lymphoma Institute and other research groups. The researchers reported having no financial disclosures.
SOURCE: Federico M et al. Lancet Haematol. 2018 Jul 4. doi: 10.1016/S2352-3026(18)30090-5.
Rituximab-based chemotherapy can significantly reduce the risk of transformation of follicular lymphoma (FL) from an indolent to an aggressive histology, such as diffuse large B-cell lymphoma, results of a retrospective pooled analysis have suggested.
“Despite the intrinsic limitations related to the retrospective nature of our study, we confirmed that the cumulative hazard of histological transformation as a first event in follicular lymphoma can be reduced significantly by introducing rituximab to a backbone therapy. Moreover, our data also confirm that histological transformation still has an adverse effect on patient outcome, although it is less catastrophic than the pre-rituximab regimens,” they wrote in the Lancet Haematology.
These investigators, from 11 cooperative groups or institutions across Europe, pooled data on patients aged 18 years and older who had a histologically confirmed diagnosis of grade 1, 2, or 3a FL between Jan. 2, 1997, and Dec. 20, 2013.
They defined histologic transformation as a biopsy-proven aggressive lymphoma that occurred as a first event after first-line therapy.
Data on a total of 8,116 patients were available for analysis; 509 of these patients had had histologic transformations. After a median follow-up of 87 months, the 10-year cumulative hazard for all patients was 7.7%. The 10-year cumulative hazard – one of two primary endpoints – was 5.2% for patients who had received any rituximab versus 8.7% for those who did not, which translated into a hazard ratio of 0.73 (P = .004).
Among patients who received rituximab during induction only, the 10-year cumulative hazard was 5.9%, and it was 3.6% among those who received rituximab during induction and maintenance phases of treatment. This difference translated into a HR of 0.55 (P = .003).
The benefit of rituximab induction and maintenance – compared with induction only – held up in a multivariate analysis controlling for age at diagnosis, sex, FLIPI (Follicular Lymphoma International Prognostic Index) score, active surveillance vs. treatment, and FL grade (HR, 0.55; P = .016).
There were 287 deaths among the 509 patients with transformation, resulting in a 10-year survival after transformation of 32%.
The 5-year survival after transformation was 38% for patients who were not exposed to rituximab, 42% for patients who received induction rituximab, and 43% for those who received both induction and maintenance rituximab, but the differences between the three groups were not statistically significant.
“More comprehensive knowledge of the biological risk factors for follicular lymphoma transformation and the molecular pathways involved is likely to help clinicians make more accurate prognostic assessments and also inform the potential usefulness of novel drugs for the treatment of follicular lymphoma,” the researchers wrote.
The study was funded by the European Lymphoma Institute and other research groups. The researchers reported having no financial disclosures.
SOURCE: Federico M et al. Lancet Haematol. 2018 Jul 4. doi: 10.1016/S2352-3026(18)30090-5.
FROM THE LANCET HAEMATOLOGY
Key clinical point:
Major finding: The 10-year cumulative hazard of histologic transformation was 5.2% for patients who had received rituximab and 8.7% for those who had not.
Study details: Retrospective pooled analysis of 8,116 patients with FL, 509 of whom had transformation over a 10-year period.
Disclosures: The study was funded by Associazione Angela Serra per la Ricerca sul Cancro, European Lymphoma Institute, European Hematology Association Lymphoma Group, Fondazione Italiana Linfomi, and the Spanish Group of Lymphoma and Bone Marrow Transplantation. The researchers reported having no financial disclosures.
Source: Federico M et al. Lancet Haematol. 2018 Jul 4. doi: 10.1016/S2352-3026(18)30090-5.
Ibrutinib stacks up well on safety in pooled analysis
in the treatment of chronic lymphocytic leukemia/small lymphocytic lymphoma (CLL/SLL) and mantle cell lymphoma (MCL), according to findings from a pooled analysis.
Susan M. O’Brien, MD, of the University of California, Irvine, and her colleagues reported pooled data from four randomized, controlled trials that included a 756 patients treated with ibrutinib and 749 patients who received a comparator drug. Patients were treated for either CLL/SLL or MCL, and safety was assessed by comparing crude and exposure-adjusted incidence rates of reported adverse events (AEs).
The comparator drugs included intravenous ofatumumab, oral chlorambucil, intravenous bendamustine plus rituximab, and intravenous temsirolimus.
While adverse event data have been published for each study analyzed, the researchers noted that the pooled analysis allows for an “in-depth assessment of the frequency and severity of both common AEs as well as additional AEs of clinical interest.”
Ibrutinib-treated patients had low rates of treatment discontinuation, compared with comparator-treatment patients (27% vs. 85%), the researchers reported in Clinical Lymphoma, Myeloma & Leukemia. Most discontinuations were caused by disease progression.
In terms of AEs, the types of events reported were similar among the drugs, with the three most common being infections, gastrointestinal disorders, and general disorders/administration-site conditions.
Diarrhea, muscle spasms, and arthralgia were reported more often among ibrutinib-treated patients. The prevalence of the most common all-grade AEs generally decreased over time with ibrutinib, peaking in the first 3 months of treatment. For serious AEs, only atrial fibrillation was higher with ibrutinib than comparator drugs when adjusted for exposure.
SOURCE: O’Brien SM et al. Clin Lymphoma Myeloma Leuk. 2018 Jun 27. doi: 10.1016/j.clml.2018.06.016.
in the treatment of chronic lymphocytic leukemia/small lymphocytic lymphoma (CLL/SLL) and mantle cell lymphoma (MCL), according to findings from a pooled analysis.
Susan M. O’Brien, MD, of the University of California, Irvine, and her colleagues reported pooled data from four randomized, controlled trials that included a 756 patients treated with ibrutinib and 749 patients who received a comparator drug. Patients were treated for either CLL/SLL or MCL, and safety was assessed by comparing crude and exposure-adjusted incidence rates of reported adverse events (AEs).
The comparator drugs included intravenous ofatumumab, oral chlorambucil, intravenous bendamustine plus rituximab, and intravenous temsirolimus.
While adverse event data have been published for each study analyzed, the researchers noted that the pooled analysis allows for an “in-depth assessment of the frequency and severity of both common AEs as well as additional AEs of clinical interest.”
Ibrutinib-treated patients had low rates of treatment discontinuation, compared with comparator-treatment patients (27% vs. 85%), the researchers reported in Clinical Lymphoma, Myeloma & Leukemia. Most discontinuations were caused by disease progression.
In terms of AEs, the types of events reported were similar among the drugs, with the three most common being infections, gastrointestinal disorders, and general disorders/administration-site conditions.
Diarrhea, muscle spasms, and arthralgia were reported more often among ibrutinib-treated patients. The prevalence of the most common all-grade AEs generally decreased over time with ibrutinib, peaking in the first 3 months of treatment. For serious AEs, only atrial fibrillation was higher with ibrutinib than comparator drugs when adjusted for exposure.
SOURCE: O’Brien SM et al. Clin Lymphoma Myeloma Leuk. 2018 Jun 27. doi: 10.1016/j.clml.2018.06.016.
in the treatment of chronic lymphocytic leukemia/small lymphocytic lymphoma (CLL/SLL) and mantle cell lymphoma (MCL), according to findings from a pooled analysis.
Susan M. O’Brien, MD, of the University of California, Irvine, and her colleagues reported pooled data from four randomized, controlled trials that included a 756 patients treated with ibrutinib and 749 patients who received a comparator drug. Patients were treated for either CLL/SLL or MCL, and safety was assessed by comparing crude and exposure-adjusted incidence rates of reported adverse events (AEs).
The comparator drugs included intravenous ofatumumab, oral chlorambucil, intravenous bendamustine plus rituximab, and intravenous temsirolimus.
While adverse event data have been published for each study analyzed, the researchers noted that the pooled analysis allows for an “in-depth assessment of the frequency and severity of both common AEs as well as additional AEs of clinical interest.”
Ibrutinib-treated patients had low rates of treatment discontinuation, compared with comparator-treatment patients (27% vs. 85%), the researchers reported in Clinical Lymphoma, Myeloma & Leukemia. Most discontinuations were caused by disease progression.
In terms of AEs, the types of events reported were similar among the drugs, with the three most common being infections, gastrointestinal disorders, and general disorders/administration-site conditions.
Diarrhea, muscle spasms, and arthralgia were reported more often among ibrutinib-treated patients. The prevalence of the most common all-grade AEs generally decreased over time with ibrutinib, peaking in the first 3 months of treatment. For serious AEs, only atrial fibrillation was higher with ibrutinib than comparator drugs when adjusted for exposure.
SOURCE: O’Brien SM et al. Clin Lymphoma Myeloma Leuk. 2018 Jun 27. doi: 10.1016/j.clml.2018.06.016.
FROM CLINICAL LYMPHOMA, MYELOMA & LEUKEMIA
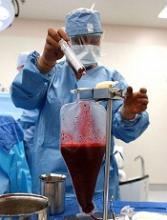
FDA grants UCB product orphan designation
The US Food and Drug Administration (FDA) has granted orphan drug designation to NiCord for hematopoietic stem cell transplant.
NiCord is created by expanding and enriching a unit of umbilical cord blood (UCB).
The product consists of a CD133-positive fraction—which is cultured for 21 days with nicotinamide, thrombopoietin, IL-6, FLT-3 ligand, and stem cell factor—and a CD133-negative fraction that is provided at the time of transplant.
NiCord already has orphan drug designation from the FDA as a treatment for acute lymphoblastic leukemia (ALL), acute myeloid leukemia (AML), Hodgkin lymphoma, and myelodysplastic syndromes (MDS).
The product also has breakthrough therapy designation from the FDA.
NiCord trials
Final results from a phase 1/2 study suggested that NiCord can be used as a stand-alone graft in patients with high-risk hematologic malignancies. The results were presented at the 2018 BMT Tandem Meetings in February.
The trial included 36 adolescents and adults with AML (n=17), ALL (n=9), MDS (n=7), chronic myeloid leukemia (CML, n=2), and Hodgkin lymphoma (n=1).
All patients received a single NiCord unit. Researchers compared engraftment results in the NiCord recipients to results in a cohort of 148 patients from the CIBMTR registry.
The registry patients underwent standard UCB transplants and had similar characteristics as the NiCord recipients. However, only 20% of the CIBMTR patients received a single UCB unit.
The median time to neutrophil engraftment was 11.5 days (range, 6-26) with NiCord and 21 days in the control cohort (P<0.001). The cumulative incidence of neutrophil engraftment was 94.4% and 89.7%, respectively.
The median time to platelet engraftment was 34 days (range, 25-96) with NiCord and 46 days in the controls (P<0.001). The cumulative incidence of platelet engraftment was 80.6% and 67.1%, respectively.
There was 1 case of primary graft failure among the NiCord recipients and 2 cases of secondary graft failure.
The estimated 2-year rate of non-relapse mortality in NiCord recipients was 23.8%, and the 2-year incidence of relapse was 33.2%.
The estimated disease-free survival was 49.1% at 1 year and 43.0% at 2 years. The overall survival was 51.2% at 1 year and 2 years.
At 100 days, the rate of grade 2-4 acute graft-vs-host disease (GVHD) was 44.0%, and the rate of grade 3-4 acute GVHD was 11.1%. The estimated 1-year rate of mild to severe chronic GVHD was 40.5%, and the 2-year rate of moderate to severe chronic GVHD was 9.8%.
These results prompted a phase 3 study of NiCord in patients with AML, ALL, CML, MDS, and lymphoma (NCT02730299). In this trial, researchers are comparing NiCord to standard single or double UCB transplant.
About orphan and breakthrough designation
The FDA grants orphan designation to products intended to treat, diagnose, or prevent diseases/disorders that affect fewer than 200,000 people in the US.
Orphan designation provides incentives for sponsors to develop products for rare diseases. This may include tax credits toward the cost of clinical trials, prescription drug user fee waivers, and 7 years of market exclusivity if the product is approved.
The FDA’s breakthrough designation is intended to expedite the development and review of new treatments for serious or life-threatening conditions.
Breakthrough designation entitles sponsors to more intensive FDA guidance on an efficient and accelerated development program, as well as eligibility for other actions to expedite FDA review, such as rolling submission and priority review.
To earn breakthrough designation, a treatment must show encouraging early clinical results demonstrating substantial improvement over available therapies with regard to a clinically significant endpoint, or it must fulfill an unmet need.
The US Food and Drug Administration (FDA) has granted orphan drug designation to NiCord for hematopoietic stem cell transplant.
NiCord is created by expanding and enriching a unit of umbilical cord blood (UCB).
The product consists of a CD133-positive fraction—which is cultured for 21 days with nicotinamide, thrombopoietin, IL-6, FLT-3 ligand, and stem cell factor—and a CD133-negative fraction that is provided at the time of transplant.
NiCord already has orphan drug designation from the FDA as a treatment for acute lymphoblastic leukemia (ALL), acute myeloid leukemia (AML), Hodgkin lymphoma, and myelodysplastic syndromes (MDS).
The product also has breakthrough therapy designation from the FDA.
NiCord trials
Final results from a phase 1/2 study suggested that NiCord can be used as a stand-alone graft in patients with high-risk hematologic malignancies. The results were presented at the 2018 BMT Tandem Meetings in February.
The trial included 36 adolescents and adults with AML (n=17), ALL (n=9), MDS (n=7), chronic myeloid leukemia (CML, n=2), and Hodgkin lymphoma (n=1).
All patients received a single NiCord unit. Researchers compared engraftment results in the NiCord recipients to results in a cohort of 148 patients from the CIBMTR registry.
The registry patients underwent standard UCB transplants and had similar characteristics as the NiCord recipients. However, only 20% of the CIBMTR patients received a single UCB unit.
The median time to neutrophil engraftment was 11.5 days (range, 6-26) with NiCord and 21 days in the control cohort (P<0.001). The cumulative incidence of neutrophil engraftment was 94.4% and 89.7%, respectively.
The median time to platelet engraftment was 34 days (range, 25-96) with NiCord and 46 days in the controls (P<0.001). The cumulative incidence of platelet engraftment was 80.6% and 67.1%, respectively.
There was 1 case of primary graft failure among the NiCord recipients and 2 cases of secondary graft failure.
The estimated 2-year rate of non-relapse mortality in NiCord recipients was 23.8%, and the 2-year incidence of relapse was 33.2%.
The estimated disease-free survival was 49.1% at 1 year and 43.0% at 2 years. The overall survival was 51.2% at 1 year and 2 years.
At 100 days, the rate of grade 2-4 acute graft-vs-host disease (GVHD) was 44.0%, and the rate of grade 3-4 acute GVHD was 11.1%. The estimated 1-year rate of mild to severe chronic GVHD was 40.5%, and the 2-year rate of moderate to severe chronic GVHD was 9.8%.
These results prompted a phase 3 study of NiCord in patients with AML, ALL, CML, MDS, and lymphoma (NCT02730299). In this trial, researchers are comparing NiCord to standard single or double UCB transplant.
About orphan and breakthrough designation
The FDA grants orphan designation to products intended to treat, diagnose, or prevent diseases/disorders that affect fewer than 200,000 people in the US.
Orphan designation provides incentives for sponsors to develop products for rare diseases. This may include tax credits toward the cost of clinical trials, prescription drug user fee waivers, and 7 years of market exclusivity if the product is approved.
The FDA’s breakthrough designation is intended to expedite the development and review of new treatments for serious or life-threatening conditions.
Breakthrough designation entitles sponsors to more intensive FDA guidance on an efficient and accelerated development program, as well as eligibility for other actions to expedite FDA review, such as rolling submission and priority review.
To earn breakthrough designation, a treatment must show encouraging early clinical results demonstrating substantial improvement over available therapies with regard to a clinically significant endpoint, or it must fulfill an unmet need.
The US Food and Drug Administration (FDA) has granted orphan drug designation to NiCord for hematopoietic stem cell transplant.
NiCord is created by expanding and enriching a unit of umbilical cord blood (UCB).
The product consists of a CD133-positive fraction—which is cultured for 21 days with nicotinamide, thrombopoietin, IL-6, FLT-3 ligand, and stem cell factor—and a CD133-negative fraction that is provided at the time of transplant.
NiCord already has orphan drug designation from the FDA as a treatment for acute lymphoblastic leukemia (ALL), acute myeloid leukemia (AML), Hodgkin lymphoma, and myelodysplastic syndromes (MDS).
The product also has breakthrough therapy designation from the FDA.
NiCord trials
Final results from a phase 1/2 study suggested that NiCord can be used as a stand-alone graft in patients with high-risk hematologic malignancies. The results were presented at the 2018 BMT Tandem Meetings in February.
The trial included 36 adolescents and adults with AML (n=17), ALL (n=9), MDS (n=7), chronic myeloid leukemia (CML, n=2), and Hodgkin lymphoma (n=1).
All patients received a single NiCord unit. Researchers compared engraftment results in the NiCord recipients to results in a cohort of 148 patients from the CIBMTR registry.
The registry patients underwent standard UCB transplants and had similar characteristics as the NiCord recipients. However, only 20% of the CIBMTR patients received a single UCB unit.
The median time to neutrophil engraftment was 11.5 days (range, 6-26) with NiCord and 21 days in the control cohort (P<0.001). The cumulative incidence of neutrophil engraftment was 94.4% and 89.7%, respectively.
The median time to platelet engraftment was 34 days (range, 25-96) with NiCord and 46 days in the controls (P<0.001). The cumulative incidence of platelet engraftment was 80.6% and 67.1%, respectively.
There was 1 case of primary graft failure among the NiCord recipients and 2 cases of secondary graft failure.
The estimated 2-year rate of non-relapse mortality in NiCord recipients was 23.8%, and the 2-year incidence of relapse was 33.2%.
The estimated disease-free survival was 49.1% at 1 year and 43.0% at 2 years. The overall survival was 51.2% at 1 year and 2 years.
At 100 days, the rate of grade 2-4 acute graft-vs-host disease (GVHD) was 44.0%, and the rate of grade 3-4 acute GVHD was 11.1%. The estimated 1-year rate of mild to severe chronic GVHD was 40.5%, and the 2-year rate of moderate to severe chronic GVHD was 9.8%.
These results prompted a phase 3 study of NiCord in patients with AML, ALL, CML, MDS, and lymphoma (NCT02730299). In this trial, researchers are comparing NiCord to standard single or double UCB transplant.
About orphan and breakthrough designation
The FDA grants orphan designation to products intended to treat, diagnose, or prevent diseases/disorders that affect fewer than 200,000 people in the US.
Orphan designation provides incentives for sponsors to develop products for rare diseases. This may include tax credits toward the cost of clinical trials, prescription drug user fee waivers, and 7 years of market exclusivity if the product is approved.
The FDA’s breakthrough designation is intended to expedite the development and review of new treatments for serious or life-threatening conditions.
Breakthrough designation entitles sponsors to more intensive FDA guidance on an efficient and accelerated development program, as well as eligibility for other actions to expedite FDA review, such as rolling submission and priority review.
To earn breakthrough designation, a treatment must show encouraging early clinical results demonstrating substantial improvement over available therapies with regard to a clinically significant endpoint, or it must fulfill an unmet need.
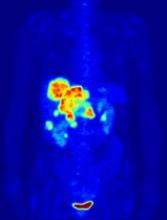
PET-guided treatment didn’t improve outcomes
In the PETAL trial, treatment intensification based on results of an interim positron emission tomography (PET) scan did not improve survival outcomes for patients with aggressive lymphomas.
PET-positive patients did not benefit by switching from R-CHOP to a more intensive chemotherapy regimen.
PET-negative patients did not benefit from 2 additional cycles of rituximab after R-CHOP.
These results were published in the Journal of Clinical Oncology.
PETAL was a randomized trial of patients with newly diagnosed T- or B-cell lymphomas.
Patients received 2 cycles of CHOP (cyclophosphamide, doxorubicin, vincristine, prednisone)—plus rituximab (R-CHOP) in CD20-positive lymphomas—followed by a PET scan.
PET-positive patients were randomized to receive 6 additional cycles of R-CHOP or 6 blocks of an intensive protocol used to treat Burkitt lymphoma. This protocol consisted of high-dose methotrexate, cytarabine, hyperfractionated cyclophosphamide and ifosfamide, split-dose doxorubicin and etoposide, vincristine, vindesine, and dexamethasone.
PET-negative patients with CD20-positive lymphomas were randomized to receive 4 additional cycles of R-CHOP or 4 additional cycles of R-CHOP followed by 2 more doses of rituximab.
Among patients with T-cell lymphomas, only PET-positive individuals underwent randomization. PET-negative patients received CHOP. Patients with CD20-positive T-cell lymphomas also received rituximab.
PET-positive results
Of the PET-positive patients (108/862), 52 were randomized to receive 6 additional cycles of R-CHOP, and 56 were randomized to 6 cycles of the Burkitt protocol.
In general, survival rates were similar regardless of treatment. The 2-year overall survival (OS) rate was 63.6% for patients who received R-CHOP and 55.4% for those who received the more intensive protocol.
Two-year progression-free survival (PFS) rates were 49.4% and 43.1%, respectively. Two-year event-free survival (EFS) rates were 42.0% and 31.6%, respectively.
Among patients with diffuse large B-cell lymphoma (DLBCL), the OS rate was 64.8% for patients who received R-CHOP and 47.1% for those on the Burkitt protocol. PFS rates were 55.5% and 41.4%, respectively.
There was a significant difference in EFS rates among the DLBCL patients—52.4% in the R-CHOP arm and 28.3% in the intensive arm (P=0.0186).
Among T-cell lymphoma patients, the OS rate was 22.2% in the R-CHOP arm and 30.0% in the intensive arm. The PFS rates were 12.7% and 30%, respectively. The EFS rates were the same as the PFS rates.
Overall, patients who received the Burkitt protocol had significantly higher rates of grade 3/4 hematologic toxicities, infection, and mucositis.
PET-negative results
Of 754 PET-negative patients, 697 had CD20-positive lymphomas, and 255 of those patients (all with B-cell lymphomas) underwent randomization.
There were 129 patients who were randomized to receive 6 cycles of R-CHOP (2 before and 4 after randomization) and 126 who were randomized to receive 6 cycles of R-CHOP plus 2 additional cycles of rituximab.
Again, survival rates were similar regardless of treatment.
The 2-year OS was 88.2% for patients who received only R-CHOP and 87.2% for those with additional rituximab exposure. PFS rates were 82.0% and 77.5%, respectively. EFS rates were 76.4% and 73.5%, respectively.
In the DLBCL patients, the OS rate was 88.5% in the R-CHOP arm and 85.8% in the intensive arm. PFS rates were 82.3% and 77.7%, respectively. EFS rates were 72.6% and 78.9%, respectively.
As increasing the dose of rituximab did not improve outcomes, the investigators concluded that 6 cycles of R-CHOP should be the standard of care for these patients.
The team also said interim PET scanning is “a powerful tool” for identifying chemotherapy-resistant lymphomas, and PET-positive patients may be candidates for immunologic treatment approaches.
In the PETAL trial, treatment intensification based on results of an interim positron emission tomography (PET) scan did not improve survival outcomes for patients with aggressive lymphomas.
PET-positive patients did not benefit by switching from R-CHOP to a more intensive chemotherapy regimen.
PET-negative patients did not benefit from 2 additional cycles of rituximab after R-CHOP.
These results were published in the Journal of Clinical Oncology.
PETAL was a randomized trial of patients with newly diagnosed T- or B-cell lymphomas.
Patients received 2 cycles of CHOP (cyclophosphamide, doxorubicin, vincristine, prednisone)—plus rituximab (R-CHOP) in CD20-positive lymphomas—followed by a PET scan.
PET-positive patients were randomized to receive 6 additional cycles of R-CHOP or 6 blocks of an intensive protocol used to treat Burkitt lymphoma. This protocol consisted of high-dose methotrexate, cytarabine, hyperfractionated cyclophosphamide and ifosfamide, split-dose doxorubicin and etoposide, vincristine, vindesine, and dexamethasone.
PET-negative patients with CD20-positive lymphomas were randomized to receive 4 additional cycles of R-CHOP or 4 additional cycles of R-CHOP followed by 2 more doses of rituximab.
Among patients with T-cell lymphomas, only PET-positive individuals underwent randomization. PET-negative patients received CHOP. Patients with CD20-positive T-cell lymphomas also received rituximab.
PET-positive results
Of the PET-positive patients (108/862), 52 were randomized to receive 6 additional cycles of R-CHOP, and 56 were randomized to 6 cycles of the Burkitt protocol.
In general, survival rates were similar regardless of treatment. The 2-year overall survival (OS) rate was 63.6% for patients who received R-CHOP and 55.4% for those who received the more intensive protocol.
Two-year progression-free survival (PFS) rates were 49.4% and 43.1%, respectively. Two-year event-free survival (EFS) rates were 42.0% and 31.6%, respectively.
Among patients with diffuse large B-cell lymphoma (DLBCL), the OS rate was 64.8% for patients who received R-CHOP and 47.1% for those on the Burkitt protocol. PFS rates were 55.5% and 41.4%, respectively.
There was a significant difference in EFS rates among the DLBCL patients—52.4% in the R-CHOP arm and 28.3% in the intensive arm (P=0.0186).
Among T-cell lymphoma patients, the OS rate was 22.2% in the R-CHOP arm and 30.0% in the intensive arm. The PFS rates were 12.7% and 30%, respectively. The EFS rates were the same as the PFS rates.
Overall, patients who received the Burkitt protocol had significantly higher rates of grade 3/4 hematologic toxicities, infection, and mucositis.
PET-negative results
Of 754 PET-negative patients, 697 had CD20-positive lymphomas, and 255 of those patients (all with B-cell lymphomas) underwent randomization.
There were 129 patients who were randomized to receive 6 cycles of R-CHOP (2 before and 4 after randomization) and 126 who were randomized to receive 6 cycles of R-CHOP plus 2 additional cycles of rituximab.
Again, survival rates were similar regardless of treatment.
The 2-year OS was 88.2% for patients who received only R-CHOP and 87.2% for those with additional rituximab exposure. PFS rates were 82.0% and 77.5%, respectively. EFS rates were 76.4% and 73.5%, respectively.
In the DLBCL patients, the OS rate was 88.5% in the R-CHOP arm and 85.8% in the intensive arm. PFS rates were 82.3% and 77.7%, respectively. EFS rates were 72.6% and 78.9%, respectively.
As increasing the dose of rituximab did not improve outcomes, the investigators concluded that 6 cycles of R-CHOP should be the standard of care for these patients.
The team also said interim PET scanning is “a powerful tool” for identifying chemotherapy-resistant lymphomas, and PET-positive patients may be candidates for immunologic treatment approaches.
In the PETAL trial, treatment intensification based on results of an interim positron emission tomography (PET) scan did not improve survival outcomes for patients with aggressive lymphomas.
PET-positive patients did not benefit by switching from R-CHOP to a more intensive chemotherapy regimen.
PET-negative patients did not benefit from 2 additional cycles of rituximab after R-CHOP.
These results were published in the Journal of Clinical Oncology.
PETAL was a randomized trial of patients with newly diagnosed T- or B-cell lymphomas.
Patients received 2 cycles of CHOP (cyclophosphamide, doxorubicin, vincristine, prednisone)—plus rituximab (R-CHOP) in CD20-positive lymphomas—followed by a PET scan.
PET-positive patients were randomized to receive 6 additional cycles of R-CHOP or 6 blocks of an intensive protocol used to treat Burkitt lymphoma. This protocol consisted of high-dose methotrexate, cytarabine, hyperfractionated cyclophosphamide and ifosfamide, split-dose doxorubicin and etoposide, vincristine, vindesine, and dexamethasone.
PET-negative patients with CD20-positive lymphomas were randomized to receive 4 additional cycles of R-CHOP or 4 additional cycles of R-CHOP followed by 2 more doses of rituximab.
Among patients with T-cell lymphomas, only PET-positive individuals underwent randomization. PET-negative patients received CHOP. Patients with CD20-positive T-cell lymphomas also received rituximab.
PET-positive results
Of the PET-positive patients (108/862), 52 were randomized to receive 6 additional cycles of R-CHOP, and 56 were randomized to 6 cycles of the Burkitt protocol.
In general, survival rates were similar regardless of treatment. The 2-year overall survival (OS) rate was 63.6% for patients who received R-CHOP and 55.4% for those who received the more intensive protocol.
Two-year progression-free survival (PFS) rates were 49.4% and 43.1%, respectively. Two-year event-free survival (EFS) rates were 42.0% and 31.6%, respectively.
Among patients with diffuse large B-cell lymphoma (DLBCL), the OS rate was 64.8% for patients who received R-CHOP and 47.1% for those on the Burkitt protocol. PFS rates were 55.5% and 41.4%, respectively.
There was a significant difference in EFS rates among the DLBCL patients—52.4% in the R-CHOP arm and 28.3% in the intensive arm (P=0.0186).
Among T-cell lymphoma patients, the OS rate was 22.2% in the R-CHOP arm and 30.0% in the intensive arm. The PFS rates were 12.7% and 30%, respectively. The EFS rates were the same as the PFS rates.
Overall, patients who received the Burkitt protocol had significantly higher rates of grade 3/4 hematologic toxicities, infection, and mucositis.
PET-negative results
Of 754 PET-negative patients, 697 had CD20-positive lymphomas, and 255 of those patients (all with B-cell lymphomas) underwent randomization.
There were 129 patients who were randomized to receive 6 cycles of R-CHOP (2 before and 4 after randomization) and 126 who were randomized to receive 6 cycles of R-CHOP plus 2 additional cycles of rituximab.
Again, survival rates were similar regardless of treatment.
The 2-year OS was 88.2% for patients who received only R-CHOP and 87.2% for those with additional rituximab exposure. PFS rates were 82.0% and 77.5%, respectively. EFS rates were 76.4% and 73.5%, respectively.
In the DLBCL patients, the OS rate was 88.5% in the R-CHOP arm and 85.8% in the intensive arm. PFS rates were 82.3% and 77.7%, respectively. EFS rates were 72.6% and 78.9%, respectively.
As increasing the dose of rituximab did not improve outcomes, the investigators concluded that 6 cycles of R-CHOP should be the standard of care for these patients.
The team also said interim PET scanning is “a powerful tool” for identifying chemotherapy-resistant lymphomas, and PET-positive patients may be candidates for immunologic treatment approaches.
New guideline for managing MCL
Rituximab should be included in first-line chemotherapy when treating mantle cell lymphoma, according to a new management guideline from the British Society for Haematology.
The best outcome data is for the R-CHOP regimen (rituximab plus cyclophosphamide, doxorubicin, vincristine, and prednisolone) followed by maintenance treatment with rituximab, wrote Pamela McKay, MD, of Beatson West of Scotland Cancer Centre in Glasgow, and her colleagues. The report was published in the British Journal of Haematology. But the combination of rituximab and bendamustine is also effective and a more favorable safety profile, according to the guideline. Single agent rituximab is not recommended.
At relapse, the guideline calls on physicians to take an individualized approach based on age, comorbidities, performance status, and response to prior therapy. Some options to consider include ibrutinib as a single agent or rituximab plus chemotherapy. The authors cautioned that there is little evidence to support maintenance rituximab after relapse treatment.
The guideline also explores the role of autologous stem cell transplantation (ASCT) and allogeneic SCT (alloSCT). The authors recommend that ASCT be considered as consolidation of first-line therapy for patients who are fit for intensive therapy. AlloSCT is a viable option in second remission among fit patients who have an appropriate donor and it may also be effective as a rescue therapy for patients who relapse after ASCT. But alloSCT is appropriate only as a first-line therapy for high-risk patients and is best used as part of a clinical trial, according to the recommendations.
The British Society of Haematology previously issued guidance on mantle cell lymphoma in 2012, but the updated document includes new drug therapeutic options and transplant data. The guideline includes a therapeutic algorithm to assist physicians in choosing first-line therapy, options after first relapse, and management in the case of higher relapse.
The guideline authors reported having no conflicts of interest.
SOURCE: McKay P et al. Br J Haematol. 2018 Jul;182(1):46-62.
Rituximab should be included in first-line chemotherapy when treating mantle cell lymphoma, according to a new management guideline from the British Society for Haematology.
The best outcome data is for the R-CHOP regimen (rituximab plus cyclophosphamide, doxorubicin, vincristine, and prednisolone) followed by maintenance treatment with rituximab, wrote Pamela McKay, MD, of Beatson West of Scotland Cancer Centre in Glasgow, and her colleagues. The report was published in the British Journal of Haematology. But the combination of rituximab and bendamustine is also effective and a more favorable safety profile, according to the guideline. Single agent rituximab is not recommended.
At relapse, the guideline calls on physicians to take an individualized approach based on age, comorbidities, performance status, and response to prior therapy. Some options to consider include ibrutinib as a single agent or rituximab plus chemotherapy. The authors cautioned that there is little evidence to support maintenance rituximab after relapse treatment.
The guideline also explores the role of autologous stem cell transplantation (ASCT) and allogeneic SCT (alloSCT). The authors recommend that ASCT be considered as consolidation of first-line therapy for patients who are fit for intensive therapy. AlloSCT is a viable option in second remission among fit patients who have an appropriate donor and it may also be effective as a rescue therapy for patients who relapse after ASCT. But alloSCT is appropriate only as a first-line therapy for high-risk patients and is best used as part of a clinical trial, according to the recommendations.
The British Society of Haematology previously issued guidance on mantle cell lymphoma in 2012, but the updated document includes new drug therapeutic options and transplant data. The guideline includes a therapeutic algorithm to assist physicians in choosing first-line therapy, options after first relapse, and management in the case of higher relapse.
The guideline authors reported having no conflicts of interest.
SOURCE: McKay P et al. Br J Haematol. 2018 Jul;182(1):46-62.
Rituximab should be included in first-line chemotherapy when treating mantle cell lymphoma, according to a new management guideline from the British Society for Haematology.
The best outcome data is for the R-CHOP regimen (rituximab plus cyclophosphamide, doxorubicin, vincristine, and prednisolone) followed by maintenance treatment with rituximab, wrote Pamela McKay, MD, of Beatson West of Scotland Cancer Centre in Glasgow, and her colleagues. The report was published in the British Journal of Haematology. But the combination of rituximab and bendamustine is also effective and a more favorable safety profile, according to the guideline. Single agent rituximab is not recommended.
At relapse, the guideline calls on physicians to take an individualized approach based on age, comorbidities, performance status, and response to prior therapy. Some options to consider include ibrutinib as a single agent or rituximab plus chemotherapy. The authors cautioned that there is little evidence to support maintenance rituximab after relapse treatment.
The guideline also explores the role of autologous stem cell transplantation (ASCT) and allogeneic SCT (alloSCT). The authors recommend that ASCT be considered as consolidation of first-line therapy for patients who are fit for intensive therapy. AlloSCT is a viable option in second remission among fit patients who have an appropriate donor and it may also be effective as a rescue therapy for patients who relapse after ASCT. But alloSCT is appropriate only as a first-line therapy for high-risk patients and is best used as part of a clinical trial, according to the recommendations.
The British Society of Haematology previously issued guidance on mantle cell lymphoma in 2012, but the updated document includes new drug therapeutic options and transplant data. The guideline includes a therapeutic algorithm to assist physicians in choosing first-line therapy, options after first relapse, and management in the case of higher relapse.
The guideline authors reported having no conflicts of interest.
SOURCE: McKay P et al. Br J Haematol. 2018 Jul;182(1):46-62.
FROM THE BRITISH JOURNAL OF HAEMATOLOGY
Genentech submits sNDA for venetoclax in untreated AML
A supplemental new drug application (sNDA) for venetoclax (Venclexta) used in combination with either a hypomethylating agent or low-dose cytarabine (LDAC) for previously untreated acute myeloid leukemia has been submitted to the Food and Drug Administration by Genentech, which developed it.
Specifically, the sNDA is for these drug combinations in the treatment of AML patients ineligible for intensive chemotherapy, according to the announcement from Genentech.
The sNDA is based on results of two trials that included patients in this population. In the phase 1b M14-358 (NCT02203773), venetoclax was combined with either azacitidine or decitabine; patients treated with 400 mg of venetoclax had a complete remission rate of 73%, and the median overall survival across all doses of venetoclax was 17.5 months. Low white blood cell count with fever, low white blood cell count, anemia, low platelet count, and decreased potassium levels were the most common grade 3/4 adverse events (occurring in 10% or more of patients). In the phase 1b/2 study M14-387 (NCT02287233), venetoclax was used in combination with LDAC; patients treated with a 600-mg dose of venetoclax showed a complete response rate of 62%, and a median overall survival of 11.4 months. Low white blood cell count with fever, decreased potassium levels, pneumonia, disease progression, decreased phosphate levels, high blood pressure, and sepsis were the most common grade 3/4 adverse events seen in this study.
This sNDA follows FDA breakthrough therapy designations, based on these same trials, for these uses of venetoclax with either hypomethylating agents or LDAC. The FDA also recently approved venetoclax in combination with rituximab (Rituxan) for treatment of patients who have chronic lymphocytic leukemia or small lymphocytic lymphoma, with or without 17p depletion, and have been treated with at least one prior therapy.
“AML is an aggressive disease with the lowest survival rate of all leukemias, and we look forward to working closely with the FDA to bring this potential option to patients with this very difficult-to-treat blood cancer as soon as possible,” said Sandra Horning, MD, chief medical officer at Genentech.
More information is included in the full release.
A supplemental new drug application (sNDA) for venetoclax (Venclexta) used in combination with either a hypomethylating agent or low-dose cytarabine (LDAC) for previously untreated acute myeloid leukemia has been submitted to the Food and Drug Administration by Genentech, which developed it.
Specifically, the sNDA is for these drug combinations in the treatment of AML patients ineligible for intensive chemotherapy, according to the announcement from Genentech.
The sNDA is based on results of two trials that included patients in this population. In the phase 1b M14-358 (NCT02203773), venetoclax was combined with either azacitidine or decitabine; patients treated with 400 mg of venetoclax had a complete remission rate of 73%, and the median overall survival across all doses of venetoclax was 17.5 months. Low white blood cell count with fever, low white blood cell count, anemia, low platelet count, and decreased potassium levels were the most common grade 3/4 adverse events (occurring in 10% or more of patients). In the phase 1b/2 study M14-387 (NCT02287233), venetoclax was used in combination with LDAC; patients treated with a 600-mg dose of venetoclax showed a complete response rate of 62%, and a median overall survival of 11.4 months. Low white blood cell count with fever, decreased potassium levels, pneumonia, disease progression, decreased phosphate levels, high blood pressure, and sepsis were the most common grade 3/4 adverse events seen in this study.
This sNDA follows FDA breakthrough therapy designations, based on these same trials, for these uses of venetoclax with either hypomethylating agents or LDAC. The FDA also recently approved venetoclax in combination with rituximab (Rituxan) for treatment of patients who have chronic lymphocytic leukemia or small lymphocytic lymphoma, with or without 17p depletion, and have been treated with at least one prior therapy.
“AML is an aggressive disease with the lowest survival rate of all leukemias, and we look forward to working closely with the FDA to bring this potential option to patients with this very difficult-to-treat blood cancer as soon as possible,” said Sandra Horning, MD, chief medical officer at Genentech.
More information is included in the full release.
A supplemental new drug application (sNDA) for venetoclax (Venclexta) used in combination with either a hypomethylating agent or low-dose cytarabine (LDAC) for previously untreated acute myeloid leukemia has been submitted to the Food and Drug Administration by Genentech, which developed it.
Specifically, the sNDA is for these drug combinations in the treatment of AML patients ineligible for intensive chemotherapy, according to the announcement from Genentech.
The sNDA is based on results of two trials that included patients in this population. In the phase 1b M14-358 (NCT02203773), venetoclax was combined with either azacitidine or decitabine; patients treated with 400 mg of venetoclax had a complete remission rate of 73%, and the median overall survival across all doses of venetoclax was 17.5 months. Low white blood cell count with fever, low white blood cell count, anemia, low platelet count, and decreased potassium levels were the most common grade 3/4 adverse events (occurring in 10% or more of patients). In the phase 1b/2 study M14-387 (NCT02287233), venetoclax was used in combination with LDAC; patients treated with a 600-mg dose of venetoclax showed a complete response rate of 62%, and a median overall survival of 11.4 months. Low white blood cell count with fever, decreased potassium levels, pneumonia, disease progression, decreased phosphate levels, high blood pressure, and sepsis were the most common grade 3/4 adverse events seen in this study.
This sNDA follows FDA breakthrough therapy designations, based on these same trials, for these uses of venetoclax with either hypomethylating agents or LDAC. The FDA also recently approved venetoclax in combination with rituximab (Rituxan) for treatment of patients who have chronic lymphocytic leukemia or small lymphocytic lymphoma, with or without 17p depletion, and have been treated with at least one prior therapy.
“AML is an aggressive disease with the lowest survival rate of all leukemias, and we look forward to working closely with the FDA to bring this potential option to patients with this very difficult-to-treat blood cancer as soon as possible,” said Sandra Horning, MD, chief medical officer at Genentech.
More information is included in the full release.
Transplant strategy not viable for aggressive B-NHL
Transplant with radioimmunotherapy (RIT)-based conditioning is a viable treatment option for patients with indolent—but not aggressive—B-cell non-Hodgkin lymphomas (NHLs), according to researchers.
Long-term follow-up data showed “excellent” outcomes in patients with indolent B-NHL who received conditioning with 90Y-ibritumomab tiuxetan plus fludarabine and low-dose total body irradiation (TBI) prior to HLA-matched hematopoietic stem cell transplant (HSCT).
However, long-term outcomes were inferior in patients with diffuse large B-cell lymphoma (DLBCL) and mantle cell lymphoma (MCL).
Camille E. Puronen, MD, of the University of Washington in Seattle, and her colleagues reported these results in Biology of Blood and Marrow Transplantation.
The study enrolled 40 patients with high-risk B-NHL. This included DLBCL (n=14), chronic lymphocytic leukemia (CLL; n=10), MCL (n=8), follicular lymphoma (FL; n=6); hairy cell leukemia (HCL; n=1), and marginal zone lymphoma (MZL; n=1).
Patients were treated with 0.4 mCi/kg 90Y-ibritumomab tiuxetan, given 2 weeks prior to HSCT, to a maximum dose of 32 mCi.
Patients also received fludarabine at 30 mg/m2 on day 5, 6, and 7 prior to HSCT and 2 Gy TBI given on the day of transplant.
In an earlier report, the objective response rate (ORR) was 60%, and 35% of patients had a complete response (CR) or unconfirmed CR.
The researchers said early responses were not associated with disease bulk or chemoresistance, as the ORR was 59% in patients with bulky or chemoresistant disease.
However, responses were associated with histology, as the ORR was 38% in patients with DLBCL, 50% in those with MCL, 83% in those with FL, and 90% in those with CLL.
Long-term survival
In the current report, 11 of 40 patients were still alive at a median follow up of 9 years (range, 5.3 to 10.2). Fourteen patients died of disease progression, and 14 died from complications of HSCT.
The 5-year overall survival (OS) was 40%, and the 5-year progression-free survival (PFS) was 28%.
The best survival rates were in patients with indolent histology. The 5-year PFS was 44% in these patients, and the 5-year OS was 67%.
The researchers said early CR was not associated with long-term survival. However, patients who had at least stable disease (SD) at earlier time points did have the opportunity to achieve long-term survival. All patients who progressed before day 84 were dead by the 1-year mark.
Of the 11 patients who were still alive at a median follow up of 9 years, 4 had a CR or unconfirmed CR at day 84 (FL: 1; CLL: 2; MCL: 1); 6 were in partial response (CLL: 3; FL: 1; MCL: 1; MZL: 1); and 1 patient with FL had SD.
Among the 18 patients with indolent NHL, long-term PFS was observed in 5 of the 7 patients who achieved early CR and 8 of the 11 patients who did not achieve early CR.
Two of the 4 MCL patients who achieved an early CR had long-term PFS, but none of the MCL patients without an early CR had long-term PFS.
Among DLBCL patients, 1 of the 4 who achieved early CR had long-term PFS, but none of the patients without an early CR had long-term PFS. Only 1 DLBCL patient survived beyond 5 years. None survived beyond 8 years.
The researchers said the favorable outcomes in patients with indolent B-NHL are consistent with the known efficacy of RIT and the graft-versus-leukemia effect in these patients.
The team also noted that, since this trial began, several novel agents have been approved for the treatment of indolent B-NHL, which means allogeneic HSCT is often moved to later in the disease course.
The researchers concluded that 90Y-ibritumomab tiuxetan-based conditioning could “continue to play an important role in these settings,” but “improved strategies are needed” for patients with MCL and DLBCL.
Transplant with radioimmunotherapy (RIT)-based conditioning is a viable treatment option for patients with indolent—but not aggressive—B-cell non-Hodgkin lymphomas (NHLs), according to researchers.
Long-term follow-up data showed “excellent” outcomes in patients with indolent B-NHL who received conditioning with 90Y-ibritumomab tiuxetan plus fludarabine and low-dose total body irradiation (TBI) prior to HLA-matched hematopoietic stem cell transplant (HSCT).
However, long-term outcomes were inferior in patients with diffuse large B-cell lymphoma (DLBCL) and mantle cell lymphoma (MCL).
Camille E. Puronen, MD, of the University of Washington in Seattle, and her colleagues reported these results in Biology of Blood and Marrow Transplantation.
The study enrolled 40 patients with high-risk B-NHL. This included DLBCL (n=14), chronic lymphocytic leukemia (CLL; n=10), MCL (n=8), follicular lymphoma (FL; n=6); hairy cell leukemia (HCL; n=1), and marginal zone lymphoma (MZL; n=1).
Patients were treated with 0.4 mCi/kg 90Y-ibritumomab tiuxetan, given 2 weeks prior to HSCT, to a maximum dose of 32 mCi.
Patients also received fludarabine at 30 mg/m2 on day 5, 6, and 7 prior to HSCT and 2 Gy TBI given on the day of transplant.
In an earlier report, the objective response rate (ORR) was 60%, and 35% of patients had a complete response (CR) or unconfirmed CR.
The researchers said early responses were not associated with disease bulk or chemoresistance, as the ORR was 59% in patients with bulky or chemoresistant disease.
However, responses were associated with histology, as the ORR was 38% in patients with DLBCL, 50% in those with MCL, 83% in those with FL, and 90% in those with CLL.
Long-term survival
In the current report, 11 of 40 patients were still alive at a median follow up of 9 years (range, 5.3 to 10.2). Fourteen patients died of disease progression, and 14 died from complications of HSCT.
The 5-year overall survival (OS) was 40%, and the 5-year progression-free survival (PFS) was 28%.
The best survival rates were in patients with indolent histology. The 5-year PFS was 44% in these patients, and the 5-year OS was 67%.
The researchers said early CR was not associated with long-term survival. However, patients who had at least stable disease (SD) at earlier time points did have the opportunity to achieve long-term survival. All patients who progressed before day 84 were dead by the 1-year mark.
Of the 11 patients who were still alive at a median follow up of 9 years, 4 had a CR or unconfirmed CR at day 84 (FL: 1; CLL: 2; MCL: 1); 6 were in partial response (CLL: 3; FL: 1; MCL: 1; MZL: 1); and 1 patient with FL had SD.
Among the 18 patients with indolent NHL, long-term PFS was observed in 5 of the 7 patients who achieved early CR and 8 of the 11 patients who did not achieve early CR.
Two of the 4 MCL patients who achieved an early CR had long-term PFS, but none of the MCL patients without an early CR had long-term PFS.
Among DLBCL patients, 1 of the 4 who achieved early CR had long-term PFS, but none of the patients without an early CR had long-term PFS. Only 1 DLBCL patient survived beyond 5 years. None survived beyond 8 years.
The researchers said the favorable outcomes in patients with indolent B-NHL are consistent with the known efficacy of RIT and the graft-versus-leukemia effect in these patients.
The team also noted that, since this trial began, several novel agents have been approved for the treatment of indolent B-NHL, which means allogeneic HSCT is often moved to later in the disease course.
The researchers concluded that 90Y-ibritumomab tiuxetan-based conditioning could “continue to play an important role in these settings,” but “improved strategies are needed” for patients with MCL and DLBCL.
Transplant with radioimmunotherapy (RIT)-based conditioning is a viable treatment option for patients with indolent—but not aggressive—B-cell non-Hodgkin lymphomas (NHLs), according to researchers.
Long-term follow-up data showed “excellent” outcomes in patients with indolent B-NHL who received conditioning with 90Y-ibritumomab tiuxetan plus fludarabine and low-dose total body irradiation (TBI) prior to HLA-matched hematopoietic stem cell transplant (HSCT).
However, long-term outcomes were inferior in patients with diffuse large B-cell lymphoma (DLBCL) and mantle cell lymphoma (MCL).
Camille E. Puronen, MD, of the University of Washington in Seattle, and her colleagues reported these results in Biology of Blood and Marrow Transplantation.
The study enrolled 40 patients with high-risk B-NHL. This included DLBCL (n=14), chronic lymphocytic leukemia (CLL; n=10), MCL (n=8), follicular lymphoma (FL; n=6); hairy cell leukemia (HCL; n=1), and marginal zone lymphoma (MZL; n=1).
Patients were treated with 0.4 mCi/kg 90Y-ibritumomab tiuxetan, given 2 weeks prior to HSCT, to a maximum dose of 32 mCi.
Patients also received fludarabine at 30 mg/m2 on day 5, 6, and 7 prior to HSCT and 2 Gy TBI given on the day of transplant.
In an earlier report, the objective response rate (ORR) was 60%, and 35% of patients had a complete response (CR) or unconfirmed CR.
The researchers said early responses were not associated with disease bulk or chemoresistance, as the ORR was 59% in patients with bulky or chemoresistant disease.
However, responses were associated with histology, as the ORR was 38% in patients with DLBCL, 50% in those with MCL, 83% in those with FL, and 90% in those with CLL.
Long-term survival
In the current report, 11 of 40 patients were still alive at a median follow up of 9 years (range, 5.3 to 10.2). Fourteen patients died of disease progression, and 14 died from complications of HSCT.
The 5-year overall survival (OS) was 40%, and the 5-year progression-free survival (PFS) was 28%.
The best survival rates were in patients with indolent histology. The 5-year PFS was 44% in these patients, and the 5-year OS was 67%.
The researchers said early CR was not associated with long-term survival. However, patients who had at least stable disease (SD) at earlier time points did have the opportunity to achieve long-term survival. All patients who progressed before day 84 were dead by the 1-year mark.
Of the 11 patients who were still alive at a median follow up of 9 years, 4 had a CR or unconfirmed CR at day 84 (FL: 1; CLL: 2; MCL: 1); 6 were in partial response (CLL: 3; FL: 1; MCL: 1; MZL: 1); and 1 patient with FL had SD.
Among the 18 patients with indolent NHL, long-term PFS was observed in 5 of the 7 patients who achieved early CR and 8 of the 11 patients who did not achieve early CR.
Two of the 4 MCL patients who achieved an early CR had long-term PFS, but none of the MCL patients without an early CR had long-term PFS.
Among DLBCL patients, 1 of the 4 who achieved early CR had long-term PFS, but none of the patients without an early CR had long-term PFS. Only 1 DLBCL patient survived beyond 5 years. None survived beyond 8 years.
The researchers said the favorable outcomes in patients with indolent B-NHL are consistent with the known efficacy of RIT and the graft-versus-leukemia effect in these patients.
The team also noted that, since this trial began, several novel agents have been approved for the treatment of indolent B-NHL, which means allogeneic HSCT is often moved to later in the disease course.
The researchers concluded that 90Y-ibritumomab tiuxetan-based conditioning could “continue to play an important role in these settings,” but “improved strategies are needed” for patients with MCL and DLBCL.