User login
NET can benefit breast cancer patients with delayed surgery
SEATTLE – A short course of neoadjuvant therapy could be considered in breast cancer patients with expected delays to resection, while they are awaiting surgery, according to study findings presented at the annual Society of Surgical Oncology Cancer Symposium.
More than half of breast cancer patients who undergo surgical resection as the initial modality will experience delays to surgery of more than 4 weeks. Of this group, more than half of patients receive shorter than standard courses of neoadjuvant therapy (NET), and the patients most likely to benefit were those older than 50 years, with ductal tumors, and the effect was seen in all T stages.
Recent reports show that NET is increasing. However, Dr. De Andrade pointed out, delays in receiving surgery remain a problem in breast cancer treatment and are associated with worse overall and cancer specific survival.
“Off-label use of NET is sometimes used in patients undergoing surgical delays,” he said.
NET use for 3 months has been associated with decreasing the size of tumors in patients with hormone receptor–positive (HR+) invasive breast cancer and allowing for breast conservation therapy. While short-term NET is sometimes used in women who are experiencing delay to surgery, the incidence and efficacy of this regimen remains undefined.
In the current study, Dr. De Andrade and his colleagues sought to answer three clinical questions:
• How long are patients with operable breast cancer waiting to undergo surgery?
• What is the pattern of use of short-course NET?
• What are the effects of short-course NET on outcomes?
The investigators used the National Cancer Database (NCDB) to identify women who had undergone surgery for stage 1-3 HR+ invasive breast cancer from 2004 to 2013. A total of 530,009 patients met inclusion criteria.
The primary outcomes of the study were time to surgery, the duration of NET, and if the pathologic stage at surgery was lower than clinical stage.
Among patients who did not receive NET, 49.3% underwent surgery within 30 days of diagnosis. More than a third (37.2%) underwent surgery within 60 days of diagnosis, and 13.5% did not have surgery until more than 60 days after their initial diagnosis. A total of 1.8% (9,664) patients underwent NET.
When looking at NET duration, 48% underwent NET for 12 or more weeks, while 52% received NET for less than 12 weeks; 27% received NET for less than 4 weeks, 17% for 4-8 weeks, and 9% for 8-12 weeks.
Downstaging from clinical stage to final pathology stage increased with longer duration of NET. It was 5.5% for less than 1 month on therapy, 9.7% for 1-2 months, and 17.2% for 2-3 months.
“For less than 4 weeks, there was no improvement in N or T downstaging,” said Dr. De Andrade. “As the amount of time on NET increased, it was associated with greater T downstaging. But for N downstaging, it was only at the standard of 12 or more weeks that a difference was seen in nodal downstaging.”
Standard NET of 12 or more weeks was associated with reduced mastectomy rates, but mastectomy rates were not lower in short-course NET.
Among patients undergoing breast conservation therapy, longer duration NET was also associated with a lower risk for re-excision (1-2 months: odds ratio, 0.82, P = .02; 2-3 months: OR, 0.40, P < .001). There was no reduction in re-excision for shorter courses of therapy.
Dr. De Andrade had no disclosures.
SEATTLE – A short course of neoadjuvant therapy could be considered in breast cancer patients with expected delays to resection, while they are awaiting surgery, according to study findings presented at the annual Society of Surgical Oncology Cancer Symposium.
More than half of breast cancer patients who undergo surgical resection as the initial modality will experience delays to surgery of more than 4 weeks. Of this group, more than half of patients receive shorter than standard courses of neoadjuvant therapy (NET), and the patients most likely to benefit were those older than 50 years, with ductal tumors, and the effect was seen in all T stages.
Recent reports show that NET is increasing. However, Dr. De Andrade pointed out, delays in receiving surgery remain a problem in breast cancer treatment and are associated with worse overall and cancer specific survival.
“Off-label use of NET is sometimes used in patients undergoing surgical delays,” he said.
NET use for 3 months has been associated with decreasing the size of tumors in patients with hormone receptor–positive (HR+) invasive breast cancer and allowing for breast conservation therapy. While short-term NET is sometimes used in women who are experiencing delay to surgery, the incidence and efficacy of this regimen remains undefined.
In the current study, Dr. De Andrade and his colleagues sought to answer three clinical questions:
• How long are patients with operable breast cancer waiting to undergo surgery?
• What is the pattern of use of short-course NET?
• What are the effects of short-course NET on outcomes?
The investigators used the National Cancer Database (NCDB) to identify women who had undergone surgery for stage 1-3 HR+ invasive breast cancer from 2004 to 2013. A total of 530,009 patients met inclusion criteria.
The primary outcomes of the study were time to surgery, the duration of NET, and if the pathologic stage at surgery was lower than clinical stage.
Among patients who did not receive NET, 49.3% underwent surgery within 30 days of diagnosis. More than a third (37.2%) underwent surgery within 60 days of diagnosis, and 13.5% did not have surgery until more than 60 days after their initial diagnosis. A total of 1.8% (9,664) patients underwent NET.
When looking at NET duration, 48% underwent NET for 12 or more weeks, while 52% received NET for less than 12 weeks; 27% received NET for less than 4 weeks, 17% for 4-8 weeks, and 9% for 8-12 weeks.
Downstaging from clinical stage to final pathology stage increased with longer duration of NET. It was 5.5% for less than 1 month on therapy, 9.7% for 1-2 months, and 17.2% for 2-3 months.
“For less than 4 weeks, there was no improvement in N or T downstaging,” said Dr. De Andrade. “As the amount of time on NET increased, it was associated with greater T downstaging. But for N downstaging, it was only at the standard of 12 or more weeks that a difference was seen in nodal downstaging.”
Standard NET of 12 or more weeks was associated with reduced mastectomy rates, but mastectomy rates were not lower in short-course NET.
Among patients undergoing breast conservation therapy, longer duration NET was also associated with a lower risk for re-excision (1-2 months: odds ratio, 0.82, P = .02; 2-3 months: OR, 0.40, P < .001). There was no reduction in re-excision for shorter courses of therapy.
Dr. De Andrade had no disclosures.
SEATTLE – A short course of neoadjuvant therapy could be considered in breast cancer patients with expected delays to resection, while they are awaiting surgery, according to study findings presented at the annual Society of Surgical Oncology Cancer Symposium.
More than half of breast cancer patients who undergo surgical resection as the initial modality will experience delays to surgery of more than 4 weeks. Of this group, more than half of patients receive shorter than standard courses of neoadjuvant therapy (NET), and the patients most likely to benefit were those older than 50 years, with ductal tumors, and the effect was seen in all T stages.
Recent reports show that NET is increasing. However, Dr. De Andrade pointed out, delays in receiving surgery remain a problem in breast cancer treatment and are associated with worse overall and cancer specific survival.
“Off-label use of NET is sometimes used in patients undergoing surgical delays,” he said.
NET use for 3 months has been associated with decreasing the size of tumors in patients with hormone receptor–positive (HR+) invasive breast cancer and allowing for breast conservation therapy. While short-term NET is sometimes used in women who are experiencing delay to surgery, the incidence and efficacy of this regimen remains undefined.
In the current study, Dr. De Andrade and his colleagues sought to answer three clinical questions:
• How long are patients with operable breast cancer waiting to undergo surgery?
• What is the pattern of use of short-course NET?
• What are the effects of short-course NET on outcomes?
The investigators used the National Cancer Database (NCDB) to identify women who had undergone surgery for stage 1-3 HR+ invasive breast cancer from 2004 to 2013. A total of 530,009 patients met inclusion criteria.
The primary outcomes of the study were time to surgery, the duration of NET, and if the pathologic stage at surgery was lower than clinical stage.
Among patients who did not receive NET, 49.3% underwent surgery within 30 days of diagnosis. More than a third (37.2%) underwent surgery within 60 days of diagnosis, and 13.5% did not have surgery until more than 60 days after their initial diagnosis. A total of 1.8% (9,664) patients underwent NET.
When looking at NET duration, 48% underwent NET for 12 or more weeks, while 52% received NET for less than 12 weeks; 27% received NET for less than 4 weeks, 17% for 4-8 weeks, and 9% for 8-12 weeks.
Downstaging from clinical stage to final pathology stage increased with longer duration of NET. It was 5.5% for less than 1 month on therapy, 9.7% for 1-2 months, and 17.2% for 2-3 months.
“For less than 4 weeks, there was no improvement in N or T downstaging,” said Dr. De Andrade. “As the amount of time on NET increased, it was associated with greater T downstaging. But for N downstaging, it was only at the standard of 12 or more weeks that a difference was seen in nodal downstaging.”
Standard NET of 12 or more weeks was associated with reduced mastectomy rates, but mastectomy rates were not lower in short-course NET.
Among patients undergoing breast conservation therapy, longer duration NET was also associated with a lower risk for re-excision (1-2 months: odds ratio, 0.82, P = .02; 2-3 months: OR, 0.40, P < .001). There was no reduction in re-excision for shorter courses of therapy.
Dr. De Andrade had no disclosures.
AT SSO 2017
Key clinical point: Short-course neoadjuvant therapy is an option for breast cancer patients with expected delays to surgery.
Major finding: Use of neoadjuvant therapy was associated with downstaging from clinical stage to final pathology stage and reducing re-excision in breast conservation surgery.
Data source: The National Cancer Database was used to identify 530,009 patients.
Disclosures: Dr. De Andrade had no disclosures.
Atezolizumab improved survival in triple-negative breast cancer
Treatment with the anti-PD-L1 cancer immunotherapy atezolizumab produced a durable clinical benefit in patients with metastatic triple-negative breast cancer who responded to treatment, according to results from a phase I study.
Overall survival (OS) rates were 41% at 1 year and 22% at both year 2 and year 3. Patients with PD-L1 on 5% or more of tumor-infiltrating immune cells (IC2/3) achieved even better clinical outcomes: Their OS rates at 1, 2, and 3 years were 45%, 28%, and 28%.
The findings from this early phase I trial, which were presented at the annual meeting of the American Association for Cancer Research, also demonstrated that response rates were higher in the first-line setting, and an exploratory biomarker analysis suggested that higher CD8 T cell and tumor-infiltrating lymphocyte counts also contributed to a better response.
“We have no targeted therapy at the moment for triple-negative breast cancer,” said study lead author Dr. Peter Schmid, director of the St. Bartholomew’s Breast Centre at St. Bartholomew’s Hospital and Barts Cancer Institute in London, during a media briefing. “The treatment we have is chemotherapy, and most patients develop resistance relatively quickly.”
Dr. Schmid noted that the median survival for these patients is still relatively short – about 9-12 months – so, the data from this trial need to be seen in that context.
“On the other hand, triple-negative breast cancer is probably the best subtype of breast cancer in terms of selecting patients for immune therapy,” said Dr. Schmid. “This is based on a high degree of genetic instability, a high rate of mutations, higher levels of PD-L1 expression, and tumor infiltrating lymphocytes inside the tumor.”
Atezolizumab is a humanized monoclonal antibody that disrupts the PD pathway, inhibits the binding of PD-L1 to PD-1 and B7.1, and, in doing so, restores tumor-specific T-cell immunity.
In this study, Dr Schmid and colleagues recruited patients with metastatic triple-negative breast cancer to one of the expansion cohorts of a phase I trial. A total of 112 patients were evaluable for response. Of this group, 19 received atezolizumab as first-line treatment, and 93 had received at least two lines of prior therapy.
Atezolizumab was administered every 3 weeks at 15 mg/kg or 20 mg/kg, and the level of PD-L1 expression on tumor-infiltrating immune cells was evaluated. The primary endpoint of the study was safety, with overall response rate, duration of response, and progression-free survival as key secondary endpoints.
The 1- and 2-year overall survival rates for responders were 100%, but that dropped to 33% and 11%, respectively, for nonresponders. Of the 11 responders, 5 received atezolizumab as first-line therapy, while 9 had high PD-L1 expression (IC2/3).
For patients who received atezolizumab in the first-line setting, 1-year overall survival was 63%, and 2-year overall survival was 47%. The rates were lower for second-line and beyond; 37% and 18%, respectively.
For IC2/3 patients, 1-year overall survival was 45%, compared with 37% for those with low to no PD-L1 expression (IC0/1).
Only 11% of patients experienced treatment-related grade 3 or greater adverse events, and side effects led to treatment discontinuation in 3% of patients.
A key message was that the duration of response had a median of 21 months, and that is significant in this disease setting, explained Dr. Schmid. Another important point was that “overall survival was significantly longer that what we see with chemotherapy.”
Genentech funded the study. Dr Schmid’s spouse is a consultant to Roche/Genentech.
Treatment with the anti-PD-L1 cancer immunotherapy atezolizumab produced a durable clinical benefit in patients with metastatic triple-negative breast cancer who responded to treatment, according to results from a phase I study.
Overall survival (OS) rates were 41% at 1 year and 22% at both year 2 and year 3. Patients with PD-L1 on 5% or more of tumor-infiltrating immune cells (IC2/3) achieved even better clinical outcomes: Their OS rates at 1, 2, and 3 years were 45%, 28%, and 28%.
The findings from this early phase I trial, which were presented at the annual meeting of the American Association for Cancer Research, also demonstrated that response rates were higher in the first-line setting, and an exploratory biomarker analysis suggested that higher CD8 T cell and tumor-infiltrating lymphocyte counts also contributed to a better response.
“We have no targeted therapy at the moment for triple-negative breast cancer,” said study lead author Dr. Peter Schmid, director of the St. Bartholomew’s Breast Centre at St. Bartholomew’s Hospital and Barts Cancer Institute in London, during a media briefing. “The treatment we have is chemotherapy, and most patients develop resistance relatively quickly.”
Dr. Schmid noted that the median survival for these patients is still relatively short – about 9-12 months – so, the data from this trial need to be seen in that context.
“On the other hand, triple-negative breast cancer is probably the best subtype of breast cancer in terms of selecting patients for immune therapy,” said Dr. Schmid. “This is based on a high degree of genetic instability, a high rate of mutations, higher levels of PD-L1 expression, and tumor infiltrating lymphocytes inside the tumor.”
Atezolizumab is a humanized monoclonal antibody that disrupts the PD pathway, inhibits the binding of PD-L1 to PD-1 and B7.1, and, in doing so, restores tumor-specific T-cell immunity.
In this study, Dr Schmid and colleagues recruited patients with metastatic triple-negative breast cancer to one of the expansion cohorts of a phase I trial. A total of 112 patients were evaluable for response. Of this group, 19 received atezolizumab as first-line treatment, and 93 had received at least two lines of prior therapy.
Atezolizumab was administered every 3 weeks at 15 mg/kg or 20 mg/kg, and the level of PD-L1 expression on tumor-infiltrating immune cells was evaluated. The primary endpoint of the study was safety, with overall response rate, duration of response, and progression-free survival as key secondary endpoints.
The 1- and 2-year overall survival rates for responders were 100%, but that dropped to 33% and 11%, respectively, for nonresponders. Of the 11 responders, 5 received atezolizumab as first-line therapy, while 9 had high PD-L1 expression (IC2/3).
For patients who received atezolizumab in the first-line setting, 1-year overall survival was 63%, and 2-year overall survival was 47%. The rates were lower for second-line and beyond; 37% and 18%, respectively.
For IC2/3 patients, 1-year overall survival was 45%, compared with 37% for those with low to no PD-L1 expression (IC0/1).
Only 11% of patients experienced treatment-related grade 3 or greater adverse events, and side effects led to treatment discontinuation in 3% of patients.
A key message was that the duration of response had a median of 21 months, and that is significant in this disease setting, explained Dr. Schmid. Another important point was that “overall survival was significantly longer that what we see with chemotherapy.”
Genentech funded the study. Dr Schmid’s spouse is a consultant to Roche/Genentech.
Treatment with the anti-PD-L1 cancer immunotherapy atezolizumab produced a durable clinical benefit in patients with metastatic triple-negative breast cancer who responded to treatment, according to results from a phase I study.
Overall survival (OS) rates were 41% at 1 year and 22% at both year 2 and year 3. Patients with PD-L1 on 5% or more of tumor-infiltrating immune cells (IC2/3) achieved even better clinical outcomes: Their OS rates at 1, 2, and 3 years were 45%, 28%, and 28%.
The findings from this early phase I trial, which were presented at the annual meeting of the American Association for Cancer Research, also demonstrated that response rates were higher in the first-line setting, and an exploratory biomarker analysis suggested that higher CD8 T cell and tumor-infiltrating lymphocyte counts also contributed to a better response.
“We have no targeted therapy at the moment for triple-negative breast cancer,” said study lead author Dr. Peter Schmid, director of the St. Bartholomew’s Breast Centre at St. Bartholomew’s Hospital and Barts Cancer Institute in London, during a media briefing. “The treatment we have is chemotherapy, and most patients develop resistance relatively quickly.”
Dr. Schmid noted that the median survival for these patients is still relatively short – about 9-12 months – so, the data from this trial need to be seen in that context.
“On the other hand, triple-negative breast cancer is probably the best subtype of breast cancer in terms of selecting patients for immune therapy,” said Dr. Schmid. “This is based on a high degree of genetic instability, a high rate of mutations, higher levels of PD-L1 expression, and tumor infiltrating lymphocytes inside the tumor.”
Atezolizumab is a humanized monoclonal antibody that disrupts the PD pathway, inhibits the binding of PD-L1 to PD-1 and B7.1, and, in doing so, restores tumor-specific T-cell immunity.
In this study, Dr Schmid and colleagues recruited patients with metastatic triple-negative breast cancer to one of the expansion cohorts of a phase I trial. A total of 112 patients were evaluable for response. Of this group, 19 received atezolizumab as first-line treatment, and 93 had received at least two lines of prior therapy.
Atezolizumab was administered every 3 weeks at 15 mg/kg or 20 mg/kg, and the level of PD-L1 expression on tumor-infiltrating immune cells was evaluated. The primary endpoint of the study was safety, with overall response rate, duration of response, and progression-free survival as key secondary endpoints.
The 1- and 2-year overall survival rates for responders were 100%, but that dropped to 33% and 11%, respectively, for nonresponders. Of the 11 responders, 5 received atezolizumab as first-line therapy, while 9 had high PD-L1 expression (IC2/3).
For patients who received atezolizumab in the first-line setting, 1-year overall survival was 63%, and 2-year overall survival was 47%. The rates were lower for second-line and beyond; 37% and 18%, respectively.
For IC2/3 patients, 1-year overall survival was 45%, compared with 37% for those with low to no PD-L1 expression (IC0/1).
Only 11% of patients experienced treatment-related grade 3 or greater adverse events, and side effects led to treatment discontinuation in 3% of patients.
A key message was that the duration of response had a median of 21 months, and that is significant in this disease setting, explained Dr. Schmid. Another important point was that “overall survival was significantly longer that what we see with chemotherapy.”
Genentech funded the study. Dr Schmid’s spouse is a consultant to Roche/Genentech.
FROM THE AACR ANNUAL MEETING
Key clinical point: In a phase I trial, the immunotherapy agent atezolizumab improved survival in triple-negative breast cancer.
Major finding: Overall survival rates were 41% at 1 year and 22% at both year 2 and year 3.
Data source: A phase I trial with a total of 112 patients who were evaluable for response.
Disclosures: Genentech funded the study. Dr Schmid’s spouse is a consultant to Roche/Genentech.
BRCA2 mutations linked to greater risk for pancreatic cancer
MIAMI BEACH – Although population-wide screening for pancreatic cancer is considered unfeasible and costly, new evidence suggests a benefit to screening a select population: people who test positive for BRCA2 genetic mutations.
Cross-sectional imaging of 117 people with BRCA2 mutations revealed pancreatic abnormalities in 10 patients, including a patient with pancreatic cancer whose only symptom was unexplained weight loss.
Pancreatic cancer is not as common as are some other malignancies, with an incidence estimated between 1% and 3%. However, it is a particularly deadly form of cancer, with only 7.7% of people living to 5 years after diagnosis, according to data from the National Cancer Institute.
A relatively low incidence is a good thing, but it also limits widespread screening. “There is a low predictive value of screening the population at large, and it is not considered cost effective,” said Eugene P. Ceppa, MD, a general surgeon at IU Health University Hospital, Indianapolis. However, patients at high risk for pancreatic adenocarcinoma might be worth targeting for screening, he added.
“This represents a 21% increase in the chance of pancreatic cancer in these patients,” Dr. Ceppa said.
Buoyed by these and other findings, Dr. Ceppa and his colleagues launched a study of their own. “Our hypothesis is that screening all BRCA2s would identify more patients with pancreatic cancer,” he said at the annual meeting of the Americas Hepato-Pancreato-Biliary Association.
Dr. Ceppa and coinvestigators reviewed electronic medical records at their institution from 2005 to 2015. They identified 204 BRCA mutation carriers, and after excluding 87 BRCA1 positive patients, further assessed the 117 with documented BRCA2 mutations. A total 47 people (40%) of this group had undergone cross-sectional imaging. The images were initially reviewed, and then re-reviewed for the study, by radiologists with specific expertise in pancreatology.
The cross-sectional imaging revealed pancreatic abnormalities in 10 people, including 1 patient with a pancreatic ductal adenocarcinoma located in the head of the pancreas. Another nine patients had intraductal papillary mucinous neoplasms (IPMNs). There were no significant demographic or clinical differences between the groups of patients with and without the imaging abnormalities, Dr. Ceppa said.
The investigators also compared the patients with BRCA2 mutations against a historical cohort representing the general population. They found 21% of patients with BRCA2 had a defined pancreatic abnormality, compared with 8% in the general population. The difference was statistically significant (P = .007).
Interestingly, the same comparison also revealed a rate of IPMN of 19%, versus 1%, respectively (P less than .001). “BRCA2 mutation carriers have significantly higher incidence of IPMN than the general population,” Dr. Ceppa said.
The study results support a high-risk screening protocol in asymptomatic BRCA patients regardless of family history, he said. In fact, a high-risk screening protocol implemented at his institution in 2013 led to a 14% detection rate of pancreatic cancer among BRCA2-positive patients, compared with a 3% rate in the general population.
“Your most significant finding might be the more IPMN patients – but how do we follow them, and will it be cost effective?” asked invited discussant Matthew J. Weiss, MD, of Johns Hopkins Medicine in Baltimore.
One of the most notable impacts of instituting the high-risk screening protocol has been an increase in patient referrals from other specialists at Dr. Ceppa’s institution. “I’ve looked at every single breast surgeon in our department, and I know how each of them are referring,” he explained.
Following initial screening of BRCA2 mutation patients, Dr. Ceppa repeats screening at 6 months, 1 year, and then annually. “However, some insurers may balk at our recommendations for frequency of screening,” he noted.
Dr. Ceppa and Dr. Weiss had no relevant financial disclosures.
MIAMI BEACH – Although population-wide screening for pancreatic cancer is considered unfeasible and costly, new evidence suggests a benefit to screening a select population: people who test positive for BRCA2 genetic mutations.
Cross-sectional imaging of 117 people with BRCA2 mutations revealed pancreatic abnormalities in 10 patients, including a patient with pancreatic cancer whose only symptom was unexplained weight loss.
Pancreatic cancer is not as common as are some other malignancies, with an incidence estimated between 1% and 3%. However, it is a particularly deadly form of cancer, with only 7.7% of people living to 5 years after diagnosis, according to data from the National Cancer Institute.
A relatively low incidence is a good thing, but it also limits widespread screening. “There is a low predictive value of screening the population at large, and it is not considered cost effective,” said Eugene P. Ceppa, MD, a general surgeon at IU Health University Hospital, Indianapolis. However, patients at high risk for pancreatic adenocarcinoma might be worth targeting for screening, he added.
“This represents a 21% increase in the chance of pancreatic cancer in these patients,” Dr. Ceppa said.
Buoyed by these and other findings, Dr. Ceppa and his colleagues launched a study of their own. “Our hypothesis is that screening all BRCA2s would identify more patients with pancreatic cancer,” he said at the annual meeting of the Americas Hepato-Pancreato-Biliary Association.
Dr. Ceppa and coinvestigators reviewed electronic medical records at their institution from 2005 to 2015. They identified 204 BRCA mutation carriers, and after excluding 87 BRCA1 positive patients, further assessed the 117 with documented BRCA2 mutations. A total 47 people (40%) of this group had undergone cross-sectional imaging. The images were initially reviewed, and then re-reviewed for the study, by radiologists with specific expertise in pancreatology.
The cross-sectional imaging revealed pancreatic abnormalities in 10 people, including 1 patient with a pancreatic ductal adenocarcinoma located in the head of the pancreas. Another nine patients had intraductal papillary mucinous neoplasms (IPMNs). There were no significant demographic or clinical differences between the groups of patients with and without the imaging abnormalities, Dr. Ceppa said.
The investigators also compared the patients with BRCA2 mutations against a historical cohort representing the general population. They found 21% of patients with BRCA2 had a defined pancreatic abnormality, compared with 8% in the general population. The difference was statistically significant (P = .007).
Interestingly, the same comparison also revealed a rate of IPMN of 19%, versus 1%, respectively (P less than .001). “BRCA2 mutation carriers have significantly higher incidence of IPMN than the general population,” Dr. Ceppa said.
The study results support a high-risk screening protocol in asymptomatic BRCA patients regardless of family history, he said. In fact, a high-risk screening protocol implemented at his institution in 2013 led to a 14% detection rate of pancreatic cancer among BRCA2-positive patients, compared with a 3% rate in the general population.
“Your most significant finding might be the more IPMN patients – but how do we follow them, and will it be cost effective?” asked invited discussant Matthew J. Weiss, MD, of Johns Hopkins Medicine in Baltimore.
One of the most notable impacts of instituting the high-risk screening protocol has been an increase in patient referrals from other specialists at Dr. Ceppa’s institution. “I’ve looked at every single breast surgeon in our department, and I know how each of them are referring,” he explained.
Following initial screening of BRCA2 mutation patients, Dr. Ceppa repeats screening at 6 months, 1 year, and then annually. “However, some insurers may balk at our recommendations for frequency of screening,” he noted.
Dr. Ceppa and Dr. Weiss had no relevant financial disclosures.
MIAMI BEACH – Although population-wide screening for pancreatic cancer is considered unfeasible and costly, new evidence suggests a benefit to screening a select population: people who test positive for BRCA2 genetic mutations.
Cross-sectional imaging of 117 people with BRCA2 mutations revealed pancreatic abnormalities in 10 patients, including a patient with pancreatic cancer whose only symptom was unexplained weight loss.
Pancreatic cancer is not as common as are some other malignancies, with an incidence estimated between 1% and 3%. However, it is a particularly deadly form of cancer, with only 7.7% of people living to 5 years after diagnosis, according to data from the National Cancer Institute.
A relatively low incidence is a good thing, but it also limits widespread screening. “There is a low predictive value of screening the population at large, and it is not considered cost effective,” said Eugene P. Ceppa, MD, a general surgeon at IU Health University Hospital, Indianapolis. However, patients at high risk for pancreatic adenocarcinoma might be worth targeting for screening, he added.
“This represents a 21% increase in the chance of pancreatic cancer in these patients,” Dr. Ceppa said.
Buoyed by these and other findings, Dr. Ceppa and his colleagues launched a study of their own. “Our hypothesis is that screening all BRCA2s would identify more patients with pancreatic cancer,” he said at the annual meeting of the Americas Hepato-Pancreato-Biliary Association.
Dr. Ceppa and coinvestigators reviewed electronic medical records at their institution from 2005 to 2015. They identified 204 BRCA mutation carriers, and after excluding 87 BRCA1 positive patients, further assessed the 117 with documented BRCA2 mutations. A total 47 people (40%) of this group had undergone cross-sectional imaging. The images were initially reviewed, and then re-reviewed for the study, by radiologists with specific expertise in pancreatology.
The cross-sectional imaging revealed pancreatic abnormalities in 10 people, including 1 patient with a pancreatic ductal adenocarcinoma located in the head of the pancreas. Another nine patients had intraductal papillary mucinous neoplasms (IPMNs). There were no significant demographic or clinical differences between the groups of patients with and without the imaging abnormalities, Dr. Ceppa said.
The investigators also compared the patients with BRCA2 mutations against a historical cohort representing the general population. They found 21% of patients with BRCA2 had a defined pancreatic abnormality, compared with 8% in the general population. The difference was statistically significant (P = .007).
Interestingly, the same comparison also revealed a rate of IPMN of 19%, versus 1%, respectively (P less than .001). “BRCA2 mutation carriers have significantly higher incidence of IPMN than the general population,” Dr. Ceppa said.
The study results support a high-risk screening protocol in asymptomatic BRCA patients regardless of family history, he said. In fact, a high-risk screening protocol implemented at his institution in 2013 led to a 14% detection rate of pancreatic cancer among BRCA2-positive patients, compared with a 3% rate in the general population.
“Your most significant finding might be the more IPMN patients – but how do we follow them, and will it be cost effective?” asked invited discussant Matthew J. Weiss, MD, of Johns Hopkins Medicine in Baltimore.
One of the most notable impacts of instituting the high-risk screening protocol has been an increase in patient referrals from other specialists at Dr. Ceppa’s institution. “I’ve looked at every single breast surgeon in our department, and I know how each of them are referring,” he explained.
Following initial screening of BRCA2 mutation patients, Dr. Ceppa repeats screening at 6 months, 1 year, and then annually. “However, some insurers may balk at our recommendations for frequency of screening,” he noted.
Dr. Ceppa and Dr. Weiss had no relevant financial disclosures.
Key clinical point: Although general population screening for pancreatic cancer is considered costly, with a low predictive value, targeting screening to patients with BRCA2 mutations could detect more cases of this deadly disease.
Major finding: People with BRCA2 mutations had a significantly greater incidence of intraductal papillary mucinous neoplasms, 19%, versus 1% in the general population (P less than .001).
Data source: Retrospective study of electronic medical records of 117 patients with BRCA2 mutations at a single academic institution.
Disclosures: Dr. Ceppa and Dr. Weiss had no relevant financial disclosures.
Liquid gold: blood-based biopsies make headway
Pathologic and, increasingly, molecular analysis of tumor tissue biopsies is the gold standard in initial diagnosis of cancer, but liquid biopsies, which analyze tumor-derived material circulating in the bloodstream are gaining traction. Here, we discuss the current state of development of this complementary and potentially alternative approach to tumor analysis.
Liquid biopsy gaining traction
Biopsies enable oncologists to gather information about a potential or established tumor, including confirmation of the presence of cancerous tissue and determination of its histological characteristics, such as tumor grade and stage, as well as its molecular features, such as the presence of certain gene mutations. Ultimately, this information can be put to use in determining the most appropriate course of treatment.
The current gold standard is a tissue biopsy that typically involves an invasive procedure to permit the collection of a piece of tumor tissue. Yet, tissue biopsies are not always feasible because of the location of the tumor or the poor performance status of many patients with advanced disease. They also provide only a snapshot of the disease at the time at which they were taken and don’t necessarily reflect the genetic heterogeneity or evolution of a tumor over time.
The detection of components that are derived from the tumor circulating in the blood of cancer patients had fueled the idea of blood-based diagnostics in oncology – so-called liquid biopsies. These have rapidly gained traction in the past several decades as a less expensive (the cost of performing genomic analyses on blood samples is at least an order of magnitude less than on tissue samples), less invasive (requiring only a simple blood draw) alternative source of information about tumors.1
As researchers have refined the ability to exploit liquid biopsies, commercial interest has been piqued. More than 35 companies within the United States alone are developing liquid biopsies, and it’s easy to see why with a market projected to be in the many billions of dollars.2
Seeking out tumor clues in the blood
Liquid biopsies consist of a 10-15 mL blood sample drawn into a tube that contains an anticoagulant and it can contain several different types of tumor-associated material. Thus far, two components – circulating tumor cells (CTCs) and circulating tumor DNA (ctDNA) – have formed the cornerstone of liquid biopsies. At present, it is not clear whether these components are released randomly, as a by-product of tumor cell death or if they are released as part of a specific biologic process, such as for the colonization of metastatic sites. It reality, it may be a little of both, and active dissemination may be particularly relevant for CTCs, among which are postulated to be a population of cancer stem cells that can initiate distant metastases.3,4
The discovery of CTCs dates back to the 1860s, when cells that were morphologically identical to the tumor were identified in the blood of a patient with metastatic cancer. Their potential significance was not fully realized until a few decades ago, when they were found to exist from early on in the course of disease.3,4
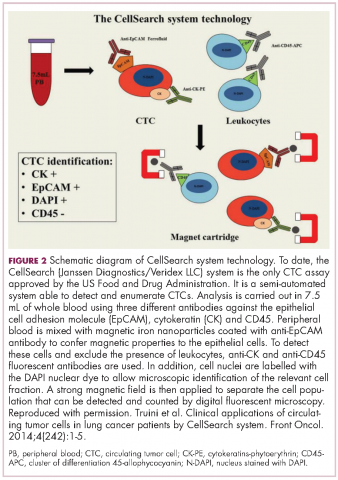
CTCs, which can be either single cells or clusters of cells known as microemboli, have a short half-life in the bloodstream – less than 2 ½ hours – and are also extremely rare (1 mL of blood contains 1-10 CTCs) against a background of many millions of normal cells. Thus the detection and isolation of CTCs presents a significant challenge. More than 40 different platforms are being developed for the isolation and enrichment of CTCs. For the most part, these use a method called positive selection to pick out CTCs.1,3,4
Positive selection exploits the biological or physical properties that are specific to CTCs and absent in normal cells, for example, the presence of a specific tumor-associated antigen on their surface or differences in size, density or electric charge. The limitations of this method are that, not only do you need to know something about CTCs to begin to understand what makes them truly unique and ensure only isolation of CTCs, but their phenotype is also thought to be continually changing.1,3,4
In recent years, the focus has shifted toward technologies that use negative depletion, meaning that they target the other types of cells in the blood sample and filter those away until only the CTCs are left behind. The most advanced are devices that use microfluidic technology to sort the cells, such as the CTC-iChip system being developed by researchers at Massachusetts General Hospital in Boston.5
ctDNA consists of small fragments of nucleic acids that are not contained within a cell or associated with cell fragments and is thought to be present in 50%-90% of patients, depending on the type of cancer they have. ctDNA has a similarly short half-life in the circulation to CTCs and, like CTCs, ctDNA is present at very low levels in the bloodstream. Although levels of ctDNA have been shown to increase with increasing tumor burden, it is still often obscured by the presence of other cell-free DNA derived from non-tumor cells.
ctDNA can be distinguished from other cell-free DNA by the presence of somatic mutations and a number of highly sensitive methods have been developed to detect them, including the amplification-refractory mutation system (ARMS); digital polymerase chain reaction; and the beads, emulsification, amplification, and magnetics (BEAMing) system. Next-generation sequencing technologies, including tagged-amplicon deep sequencing (TAm-Seq), the Safe-Sequencing System (Safe-SeqS), and cancer personalized profiling by deep sequencing (CAPP-seq), can also be used and the race for ever more sensitive analytical tools is ongoing.1,3,4,6
Applying liquid biopsies now and in the future
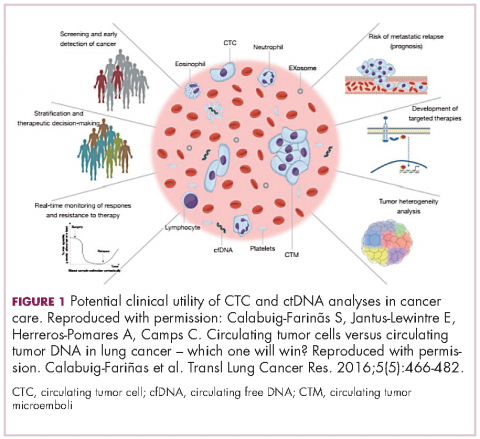
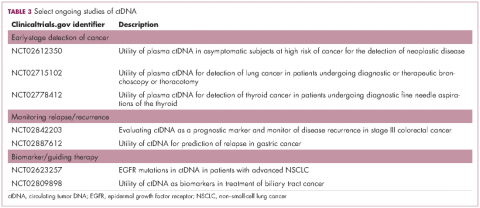
There are a plethora of potential applications for liquid biopsies3,7 (Figure 1), and probably the most exciting among them is the potential for screening for and early detection of cancer. The fact that ctDNA and CTCs have both been shown to be present from the earliest stages of disease has sparked interest in the possibility of developing simple blood tests to identify tumors before they become detectable by other methods and at a point at which they may be curable.
Given that both are present at such low levels within the circulation and are particularly sparse at earlier stages of disease, current technologies may lack the specificity and sensitivity for this application at present. However, numerous clinical trials are ongoing.
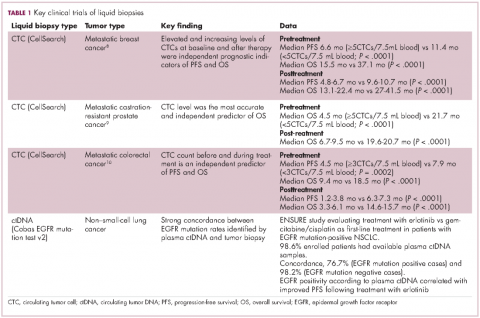
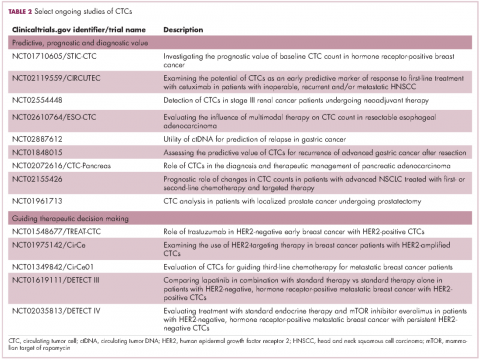
For CTCs, simple enumeration has been the most extensively investigated application to date. Numerous studies have shown that the number of CTCs in the bloodstream has prognostic significance in various different tumor types. Three such studies led to the first regulatory approval for a CTC detection system (Table 1 and Table 2).8-10
One area in which liquid biopsies could really come into their own is in providing more real-time analysis of tumors. This is something that has proven particularly challenging with tissue biopsies because repeating these invasive procedures is problematic. But the ease of repeat blood draws means that serial liquid biopsies could be performed and might offer the possibility of monitoring disease progression and evolution over the course of disease and particularly in response to treatment.
Indeed, studies have shown that in addition to baseline CTC counts, changes in CTC number during treatment are also prognostic. There was improved survival among patients whose CTC counts decreased below a threshold value during treatment and vice versa. This is also an approved use for CellSearch though at present it is not widely clinically implemented.12
Clinical utility remains elusive
The ultimate goal would be for liquid biopsies to have an impact on treatment decisions, allowing oncologists to change management strategy based on predicted sensitivity or resistance to therapy, so-called clinical utility. Thus far, clinical utility has proved elusive, though liquid biopsies using ctDNA to evaluate tumor genotype have come closest.
The Cobas EGFR Mutation Test v2 recently became the first ctDNA-based liquid biopsy to receive regulatory approval. It was approved as a companion diagnostic to identify patients with advanced non–small-cell lung cancer (NSCLC) who have specific mutations in the epidermal growth factor receptor (EGFR) gene and are therefore eligible for treatment with the EGFR inhibitor erlotinib.13
Approval was based on comparison of EGFR mutation identification rates using plasma ctDNA samples and tumor tissue samples from patients enrolled in the phase 3 ENSURE trial, which compared the efficacy of erlotinib with chemotherapy as first-line therapy in patients with advanced NSCLC. Of the 217 patients enrolled in the trial, 98.6% of patients had both tumor biopsy and plasma ctDNA samples available for testing. Concordance between the two types of biopsy in identifying patients with EGFR mutations was high and patients with EGFR positivity according to liquid biopsy results demonstrated improved progression-free survival when treated with erlotinib.14
The results of a large-scale genomic analysis of various different types of tumors using ctDNA were also recently presented at the 2016 American Society of Clinical Oncology meeting. Blood samples from more than 15,000 patients with 50 different tumor types, including advanced lung cancer (37%), breast cancer (14%), and CRC (10%), were collected and compared with either available tumor biopsy samples from the same cases (n = 398) or, in the majority of cases, with The Cancer Genome Atlas database, which uses tumor biopsies to perform genome-wide sequencing studies. Both types of biopsy revealed very similar mutation patterns when the Guardant360 next-generation sequencing test, which targets 70 genes, was applied. In particular, when EGFR, BRAF, KRAS, ALK, RET, and ROS1 mutations were identified by tumor tissue biopsy, the same mutations were reported in 94%-100% of plasma samples.15
Studies of the clinical utility of ctDNA and CTCs are among ongoing clinical trials of liquid biopsies (Tables 2 and 3). The potential for using CTCs to guide treatment decisions has become particularly relevant in breast cancer in light of results showing that patients with primary tumors that are negative for human epidermal growth factor receptor 2 (HER2) amplification, an important biomarker in breast cancer, may have CTCs that are HER2-positive, in up to 30% of cases. These patients may therefore still benefit from HER2-targeted therapy.16
The DETECT studies are the first phase 3 trials in which treatment decisions are being based on the phenotypic characteristics of CTCs. DETECT III (NCT01619111) is comparing lapatinib in combination with standard therapy with standard therapy alone in patients with HER2-negative metastatic breast cancer who have HER2-positive CTCs, whereas DETECT IV (NCT02035813) is enrolling patients with HER2-negative, hormone receptor-positive metastatic breast cancer and persistent HER2-negative CTCs to receive standard endocrine therapy and the mammalian target of rapamycin inhibitor everolimus.
Other targets and sources for liquid biopsy
Another approach to liquid biopsies that is also beginning to take off is to collect tumor-derived exosomes from the bloodstream. Exosomes are tiny, fluid-filled, membrane-bound sacks that bud off from the surface of a cell to expel waste or to transport cargo from one cell to another. DNA, RNA, and protein can be extracted from tumor-derived exosomes and could also serve as molecular biomarkers relating to the cancer cells from which they came.6,7
Exosome Diagnostics is bringing the first exosome-based diagnostic tests to the market and recently teamed up with Amgen for the development of these liquid biopsies.17 In January 2016, they launched ExoDx Lung (ALK), for detection of EML4-ALK gene fusions in patients with NSCLC, using a proprietary platform for the isolation of RNA from exosomes. Data that was presented at several different conferences in 2015 demonstrated a sensitivity of 88% and specificity of 100% for this diagnostic when compared with tissue ALK status in NSCLC patients receiving a second-generation ALK inhibitor following progression on prior ALK inhibitor therapy.18
In September, they subsequently announced the launch of a test that analyses genetic information from exosomes collected from a urine sample taken from prostate cancer patients. Using a 3-gene signature, in combination with a proprietary algorithm, this diagnostic generates a score assessing a prostate cancer patient’s risk for higher grade, more aggressive disease. It is designed to complement the prostate-specific antigen score and has demonstrated accuracy in ruling out the presence of high-grade cancer before an initial biopsy in more than 1,
1. Lennon NK, Adalsteinsson VA, Gabriel SB. Technological considerations for genome-guided diagnosis and management of cancer. Genome Med. 2016;8:112.
2. MIT Technology Review website. Liquid biopsy: fast DNA-sequencing machines are leading to simple blood tests for cancer. https://www.technologyreview.com/s/534991/liquid-biopsy/. Published 2015. Accessed December 19, 2016.
3. Alix-Panabières C and Pantel K. Clinical applications of circulating tumor cells and circulating tumor DNA as liquid biopsy. Cancer Discov. 2016;6(5):479-491.
4. Calabuig-Farinãs S, Jantus-Lewintre E, Herreros-Pomares A, Camps C. Circulating tumor cells versus circulating tumor DNA in lung cancer – which one will win? Transl Lung Cancer Res. 2016;5(5):466-482.
5. Karabacak, NM, Spuhler PS, Fachin F, et al. Microfluidic, marker-free isolation of circulating tumor cells from blood samples. Nat Protoc. 2014;9:694-710.
6. Buder A, Tomuta C, and Filipits M. The potential of liquid biopsies. Curr Opin Oncol. 2016;28:130-134.
7. Hofman P, Popper HH. Pathologists and liquid biopsies: to be or not to be? Virchows Arch. 2016;469:601-609.
8. Bidard FC, Peeters DJ, Fehm T, et al. Clinical validity of circulating tumor cells in patients with metastatic breast cancer: a pooled analysis of individual patient data. Lancet Oncol. 2014;15(4):406-414.
9. de Bono JS, Scher HI, Montgomery RB, et al. Circulating tumor cells predict survival benefit from treatment in metastatic castration-resistant prostate cancer. Clin Cancer Res. 2008;14(19):6302-6309.
10. Cohen SJ, Punt CJ, Iannotti N, et al. Relationship of circulating tumor cells to tumor response, progression-free survival, and overall survival in patients with metastatic colorectal cancer. J Clin Oncol. 2008;26(19):3213-3221.
11. CellSearch Web site. What is the CELLSEARCH® System? https://www.cellsearchctc.com/product-systems-overview/cellsearch-system-overview. Last updated December 5th, 2016. Accessed online December 19th, 2016.
12. CellSearch Web site [advertisement]. https://www.cellsearchctc.com/clinical-applications/clinical-applications-overview. Last updated December 5, 2016. Accessed December 19, 2016.
13. US Food and Drug Administration. cobas EGFR Mutation Test v2 – P150047. http://www.fda.gov/MedicalDevices/ProductsandMedicalProcedures/DeviceApprovalsandClearances/Recently-ApprovedDevices/ucm519922.htm. Last updated September 9, 2016. Accessed December 19, 2016.
14. Wu YL, Zhou C, Liam CK, et al. First-line erlotinib versus gemcitabine/cisplatin in patients with advanced EGFR mutation-positive non-small cell lung cancer: analyses from the phase III, randomized, open-label, ENSURE study. Ann Oncol. 2015;26(9):1883-1889.
15. Zill OA, Mortimer S, Banks KC, et al. Somatic genomic landscape of over 15,000 patients with advanced-stage cancer from clinical next-generation sequencing analysis of circulating tumor DNA. J Clin Oncol. 2016;34(suppl;abstr LBA11501).
16. Jordan NV, Bardia A, Wittner BS, et al. HER2 expression identifies dynamic functional states within circulating breast cancer cells. Nature. 2016;537:102-106.
17. Exosome Diagnostics. Exosome diagnostics enters agreement with Amgen. http://www.exosomedx.com/news-events/press-releases/exosome-diagnostics-enters-agreement-amgen. Published October 3, 2016. Accessed December 19, 2016.
18. Brinkman K, Emenegger J, Tannous B, et al. Exosomal RNA-based liquid biopsy detection of EML4-ALK in plasma from NSCLC patients [2015 World Conference on Lung Cancer, Denver, CO; abstract 2591]. http://library.iaslc.org/search-speaker?search_speaker=30493. Accessed January 6, 2017.
19. Exosome Diagnostics website. Prostate cancer. http://www.exosomedx.com/prostate-cancer-0. Last updated 2017. Accessed online December 19, 2016.
Pathologic and, increasingly, molecular analysis of tumor tissue biopsies is the gold standard in initial diagnosis of cancer, but liquid biopsies, which analyze tumor-derived material circulating in the bloodstream are gaining traction. Here, we discuss the current state of development of this complementary and potentially alternative approach to tumor analysis.
Liquid biopsy gaining traction
Biopsies enable oncologists to gather information about a potential or established tumor, including confirmation of the presence of cancerous tissue and determination of its histological characteristics, such as tumor grade and stage, as well as its molecular features, such as the presence of certain gene mutations. Ultimately, this information can be put to use in determining the most appropriate course of treatment.
The current gold standard is a tissue biopsy that typically involves an invasive procedure to permit the collection of a piece of tumor tissue. Yet, tissue biopsies are not always feasible because of the location of the tumor or the poor performance status of many patients with advanced disease. They also provide only a snapshot of the disease at the time at which they were taken and don’t necessarily reflect the genetic heterogeneity or evolution of a tumor over time.
The detection of components that are derived from the tumor circulating in the blood of cancer patients had fueled the idea of blood-based diagnostics in oncology – so-called liquid biopsies. These have rapidly gained traction in the past several decades as a less expensive (the cost of performing genomic analyses on blood samples is at least an order of magnitude less than on tissue samples), less invasive (requiring only a simple blood draw) alternative source of information about tumors.1
As researchers have refined the ability to exploit liquid biopsies, commercial interest has been piqued. More than 35 companies within the United States alone are developing liquid biopsies, and it’s easy to see why with a market projected to be in the many billions of dollars.2
Seeking out tumor clues in the blood
Liquid biopsies consist of a 10-15 mL blood sample drawn into a tube that contains an anticoagulant and it can contain several different types of tumor-associated material. Thus far, two components – circulating tumor cells (CTCs) and circulating tumor DNA (ctDNA) – have formed the cornerstone of liquid biopsies. At present, it is not clear whether these components are released randomly, as a by-product of tumor cell death or if they are released as part of a specific biologic process, such as for the colonization of metastatic sites. It reality, it may be a little of both, and active dissemination may be particularly relevant for CTCs, among which are postulated to be a population of cancer stem cells that can initiate distant metastases.3,4
The discovery of CTCs dates back to the 1860s, when cells that were morphologically identical to the tumor were identified in the blood of a patient with metastatic cancer. Their potential significance was not fully realized until a few decades ago, when they were found to exist from early on in the course of disease.3,4
CTCs, which can be either single cells or clusters of cells known as microemboli, have a short half-life in the bloodstream – less than 2 ½ hours – and are also extremely rare (1 mL of blood contains 1-10 CTCs) against a background of many millions of normal cells. Thus the detection and isolation of CTCs presents a significant challenge. More than 40 different platforms are being developed for the isolation and enrichment of CTCs. For the most part, these use a method called positive selection to pick out CTCs.1,3,4
Positive selection exploits the biological or physical properties that are specific to CTCs and absent in normal cells, for example, the presence of a specific tumor-associated antigen on their surface or differences in size, density or electric charge. The limitations of this method are that, not only do you need to know something about CTCs to begin to understand what makes them truly unique and ensure only isolation of CTCs, but their phenotype is also thought to be continually changing.1,3,4
In recent years, the focus has shifted toward technologies that use negative depletion, meaning that they target the other types of cells in the blood sample and filter those away until only the CTCs are left behind. The most advanced are devices that use microfluidic technology to sort the cells, such as the CTC-iChip system being developed by researchers at Massachusetts General Hospital in Boston.5
ctDNA consists of small fragments of nucleic acids that are not contained within a cell or associated with cell fragments and is thought to be present in 50%-90% of patients, depending on the type of cancer they have. ctDNA has a similarly short half-life in the circulation to CTCs and, like CTCs, ctDNA is present at very low levels in the bloodstream. Although levels of ctDNA have been shown to increase with increasing tumor burden, it is still often obscured by the presence of other cell-free DNA derived from non-tumor cells.
ctDNA can be distinguished from other cell-free DNA by the presence of somatic mutations and a number of highly sensitive methods have been developed to detect them, including the amplification-refractory mutation system (ARMS); digital polymerase chain reaction; and the beads, emulsification, amplification, and magnetics (BEAMing) system. Next-generation sequencing technologies, including tagged-amplicon deep sequencing (TAm-Seq), the Safe-Sequencing System (Safe-SeqS), and cancer personalized profiling by deep sequencing (CAPP-seq), can also be used and the race for ever more sensitive analytical tools is ongoing.1,3,4,6
Applying liquid biopsies now and in the future
There are a plethora of potential applications for liquid biopsies3,7 (Figure 1), and probably the most exciting among them is the potential for screening for and early detection of cancer. The fact that ctDNA and CTCs have both been shown to be present from the earliest stages of disease has sparked interest in the possibility of developing simple blood tests to identify tumors before they become detectable by other methods and at a point at which they may be curable.
Given that both are present at such low levels within the circulation and are particularly sparse at earlier stages of disease, current technologies may lack the specificity and sensitivity for this application at present. However, numerous clinical trials are ongoing.
For CTCs, simple enumeration has been the most extensively investigated application to date. Numerous studies have shown that the number of CTCs in the bloodstream has prognostic significance in various different tumor types. Three such studies led to the first regulatory approval for a CTC detection system (Table 1 and Table 2).8-10
One area in which liquid biopsies could really come into their own is in providing more real-time analysis of tumors. This is something that has proven particularly challenging with tissue biopsies because repeating these invasive procedures is problematic. But the ease of repeat blood draws means that serial liquid biopsies could be performed and might offer the possibility of monitoring disease progression and evolution over the course of disease and particularly in response to treatment.
Indeed, studies have shown that in addition to baseline CTC counts, changes in CTC number during treatment are also prognostic. There was improved survival among patients whose CTC counts decreased below a threshold value during treatment and vice versa. This is also an approved use for CellSearch though at present it is not widely clinically implemented.12
Clinical utility remains elusive
The ultimate goal would be for liquid biopsies to have an impact on treatment decisions, allowing oncologists to change management strategy based on predicted sensitivity or resistance to therapy, so-called clinical utility. Thus far, clinical utility has proved elusive, though liquid biopsies using ctDNA to evaluate tumor genotype have come closest.
The Cobas EGFR Mutation Test v2 recently became the first ctDNA-based liquid biopsy to receive regulatory approval. It was approved as a companion diagnostic to identify patients with advanced non–small-cell lung cancer (NSCLC) who have specific mutations in the epidermal growth factor receptor (EGFR) gene and are therefore eligible for treatment with the EGFR inhibitor erlotinib.13
Approval was based on comparison of EGFR mutation identification rates using plasma ctDNA samples and tumor tissue samples from patients enrolled in the phase 3 ENSURE trial, which compared the efficacy of erlotinib with chemotherapy as first-line therapy in patients with advanced NSCLC. Of the 217 patients enrolled in the trial, 98.6% of patients had both tumor biopsy and plasma ctDNA samples available for testing. Concordance between the two types of biopsy in identifying patients with EGFR mutations was high and patients with EGFR positivity according to liquid biopsy results demonstrated improved progression-free survival when treated with erlotinib.14
The results of a large-scale genomic analysis of various different types of tumors using ctDNA were also recently presented at the 2016 American Society of Clinical Oncology meeting. Blood samples from more than 15,000 patients with 50 different tumor types, including advanced lung cancer (37%), breast cancer (14%), and CRC (10%), were collected and compared with either available tumor biopsy samples from the same cases (n = 398) or, in the majority of cases, with The Cancer Genome Atlas database, which uses tumor biopsies to perform genome-wide sequencing studies. Both types of biopsy revealed very similar mutation patterns when the Guardant360 next-generation sequencing test, which targets 70 genes, was applied. In particular, when EGFR, BRAF, KRAS, ALK, RET, and ROS1 mutations were identified by tumor tissue biopsy, the same mutations were reported in 94%-100% of plasma samples.15
Studies of the clinical utility of ctDNA and CTCs are among ongoing clinical trials of liquid biopsies (Tables 2 and 3). The potential for using CTCs to guide treatment decisions has become particularly relevant in breast cancer in light of results showing that patients with primary tumors that are negative for human epidermal growth factor receptor 2 (HER2) amplification, an important biomarker in breast cancer, may have CTCs that are HER2-positive, in up to 30% of cases. These patients may therefore still benefit from HER2-targeted therapy.16
The DETECT studies are the first phase 3 trials in which treatment decisions are being based on the phenotypic characteristics of CTCs. DETECT III (NCT01619111) is comparing lapatinib in combination with standard therapy with standard therapy alone in patients with HER2-negative metastatic breast cancer who have HER2-positive CTCs, whereas DETECT IV (NCT02035813) is enrolling patients with HER2-negative, hormone receptor-positive metastatic breast cancer and persistent HER2-negative CTCs to receive standard endocrine therapy and the mammalian target of rapamycin inhibitor everolimus.
Other targets and sources for liquid biopsy
Another approach to liquid biopsies that is also beginning to take off is to collect tumor-derived exosomes from the bloodstream. Exosomes are tiny, fluid-filled, membrane-bound sacks that bud off from the surface of a cell to expel waste or to transport cargo from one cell to another. DNA, RNA, and protein can be extracted from tumor-derived exosomes and could also serve as molecular biomarkers relating to the cancer cells from which they came.6,7
Exosome Diagnostics is bringing the first exosome-based diagnostic tests to the market and recently teamed up with Amgen for the development of these liquid biopsies.17 In January 2016, they launched ExoDx Lung (ALK), for detection of EML4-ALK gene fusions in patients with NSCLC, using a proprietary platform for the isolation of RNA from exosomes. Data that was presented at several different conferences in 2015 demonstrated a sensitivity of 88% and specificity of 100% for this diagnostic when compared with tissue ALK status in NSCLC patients receiving a second-generation ALK inhibitor following progression on prior ALK inhibitor therapy.18
In September, they subsequently announced the launch of a test that analyses genetic information from exosomes collected from a urine sample taken from prostate cancer patients. Using a 3-gene signature, in combination with a proprietary algorithm, this diagnostic generates a score assessing a prostate cancer patient’s risk for higher grade, more aggressive disease. It is designed to complement the prostate-specific antigen score and has demonstrated accuracy in ruling out the presence of high-grade cancer before an initial biopsy in more than 1,
Pathologic and, increasingly, molecular analysis of tumor tissue biopsies is the gold standard in initial diagnosis of cancer, but liquid biopsies, which analyze tumor-derived material circulating in the bloodstream are gaining traction. Here, we discuss the current state of development of this complementary and potentially alternative approach to tumor analysis.
Liquid biopsy gaining traction
Biopsies enable oncologists to gather information about a potential or established tumor, including confirmation of the presence of cancerous tissue and determination of its histological characteristics, such as tumor grade and stage, as well as its molecular features, such as the presence of certain gene mutations. Ultimately, this information can be put to use in determining the most appropriate course of treatment.
The current gold standard is a tissue biopsy that typically involves an invasive procedure to permit the collection of a piece of tumor tissue. Yet, tissue biopsies are not always feasible because of the location of the tumor or the poor performance status of many patients with advanced disease. They also provide only a snapshot of the disease at the time at which they were taken and don’t necessarily reflect the genetic heterogeneity or evolution of a tumor over time.
The detection of components that are derived from the tumor circulating in the blood of cancer patients had fueled the idea of blood-based diagnostics in oncology – so-called liquid biopsies. These have rapidly gained traction in the past several decades as a less expensive (the cost of performing genomic analyses on blood samples is at least an order of magnitude less than on tissue samples), less invasive (requiring only a simple blood draw) alternative source of information about tumors.1
As researchers have refined the ability to exploit liquid biopsies, commercial interest has been piqued. More than 35 companies within the United States alone are developing liquid biopsies, and it’s easy to see why with a market projected to be in the many billions of dollars.2
Seeking out tumor clues in the blood
Liquid biopsies consist of a 10-15 mL blood sample drawn into a tube that contains an anticoagulant and it can contain several different types of tumor-associated material. Thus far, two components – circulating tumor cells (CTCs) and circulating tumor DNA (ctDNA) – have formed the cornerstone of liquid biopsies. At present, it is not clear whether these components are released randomly, as a by-product of tumor cell death or if they are released as part of a specific biologic process, such as for the colonization of metastatic sites. It reality, it may be a little of both, and active dissemination may be particularly relevant for CTCs, among which are postulated to be a population of cancer stem cells that can initiate distant metastases.3,4
The discovery of CTCs dates back to the 1860s, when cells that were morphologically identical to the tumor were identified in the blood of a patient with metastatic cancer. Their potential significance was not fully realized until a few decades ago, when they were found to exist from early on in the course of disease.3,4
CTCs, which can be either single cells or clusters of cells known as microemboli, have a short half-life in the bloodstream – less than 2 ½ hours – and are also extremely rare (1 mL of blood contains 1-10 CTCs) against a background of many millions of normal cells. Thus the detection and isolation of CTCs presents a significant challenge. More than 40 different platforms are being developed for the isolation and enrichment of CTCs. For the most part, these use a method called positive selection to pick out CTCs.1,3,4
Positive selection exploits the biological or physical properties that are specific to CTCs and absent in normal cells, for example, the presence of a specific tumor-associated antigen on their surface or differences in size, density or electric charge. The limitations of this method are that, not only do you need to know something about CTCs to begin to understand what makes them truly unique and ensure only isolation of CTCs, but their phenotype is also thought to be continually changing.1,3,4
In recent years, the focus has shifted toward technologies that use negative depletion, meaning that they target the other types of cells in the blood sample and filter those away until only the CTCs are left behind. The most advanced are devices that use microfluidic technology to sort the cells, such as the CTC-iChip system being developed by researchers at Massachusetts General Hospital in Boston.5
ctDNA consists of small fragments of nucleic acids that are not contained within a cell or associated with cell fragments and is thought to be present in 50%-90% of patients, depending on the type of cancer they have. ctDNA has a similarly short half-life in the circulation to CTCs and, like CTCs, ctDNA is present at very low levels in the bloodstream. Although levels of ctDNA have been shown to increase with increasing tumor burden, it is still often obscured by the presence of other cell-free DNA derived from non-tumor cells.
ctDNA can be distinguished from other cell-free DNA by the presence of somatic mutations and a number of highly sensitive methods have been developed to detect them, including the amplification-refractory mutation system (ARMS); digital polymerase chain reaction; and the beads, emulsification, amplification, and magnetics (BEAMing) system. Next-generation sequencing technologies, including tagged-amplicon deep sequencing (TAm-Seq), the Safe-Sequencing System (Safe-SeqS), and cancer personalized profiling by deep sequencing (CAPP-seq), can also be used and the race for ever more sensitive analytical tools is ongoing.1,3,4,6
Applying liquid biopsies now and in the future
There are a plethora of potential applications for liquid biopsies3,7 (Figure 1), and probably the most exciting among them is the potential for screening for and early detection of cancer. The fact that ctDNA and CTCs have both been shown to be present from the earliest stages of disease has sparked interest in the possibility of developing simple blood tests to identify tumors before they become detectable by other methods and at a point at which they may be curable.
Given that both are present at such low levels within the circulation and are particularly sparse at earlier stages of disease, current technologies may lack the specificity and sensitivity for this application at present. However, numerous clinical trials are ongoing.
For CTCs, simple enumeration has been the most extensively investigated application to date. Numerous studies have shown that the number of CTCs in the bloodstream has prognostic significance in various different tumor types. Three such studies led to the first regulatory approval for a CTC detection system (Table 1 and Table 2).8-10
One area in which liquid biopsies could really come into their own is in providing more real-time analysis of tumors. This is something that has proven particularly challenging with tissue biopsies because repeating these invasive procedures is problematic. But the ease of repeat blood draws means that serial liquid biopsies could be performed and might offer the possibility of monitoring disease progression and evolution over the course of disease and particularly in response to treatment.
Indeed, studies have shown that in addition to baseline CTC counts, changes in CTC number during treatment are also prognostic. There was improved survival among patients whose CTC counts decreased below a threshold value during treatment and vice versa. This is also an approved use for CellSearch though at present it is not widely clinically implemented.12
Clinical utility remains elusive
The ultimate goal would be for liquid biopsies to have an impact on treatment decisions, allowing oncologists to change management strategy based on predicted sensitivity or resistance to therapy, so-called clinical utility. Thus far, clinical utility has proved elusive, though liquid biopsies using ctDNA to evaluate tumor genotype have come closest.
The Cobas EGFR Mutation Test v2 recently became the first ctDNA-based liquid biopsy to receive regulatory approval. It was approved as a companion diagnostic to identify patients with advanced non–small-cell lung cancer (NSCLC) who have specific mutations in the epidermal growth factor receptor (EGFR) gene and are therefore eligible for treatment with the EGFR inhibitor erlotinib.13
Approval was based on comparison of EGFR mutation identification rates using plasma ctDNA samples and tumor tissue samples from patients enrolled in the phase 3 ENSURE trial, which compared the efficacy of erlotinib with chemotherapy as first-line therapy in patients with advanced NSCLC. Of the 217 patients enrolled in the trial, 98.6% of patients had both tumor biopsy and plasma ctDNA samples available for testing. Concordance between the two types of biopsy in identifying patients with EGFR mutations was high and patients with EGFR positivity according to liquid biopsy results demonstrated improved progression-free survival when treated with erlotinib.14
The results of a large-scale genomic analysis of various different types of tumors using ctDNA were also recently presented at the 2016 American Society of Clinical Oncology meeting. Blood samples from more than 15,000 patients with 50 different tumor types, including advanced lung cancer (37%), breast cancer (14%), and CRC (10%), were collected and compared with either available tumor biopsy samples from the same cases (n = 398) or, in the majority of cases, with The Cancer Genome Atlas database, which uses tumor biopsies to perform genome-wide sequencing studies. Both types of biopsy revealed very similar mutation patterns when the Guardant360 next-generation sequencing test, which targets 70 genes, was applied. In particular, when EGFR, BRAF, KRAS, ALK, RET, and ROS1 mutations were identified by tumor tissue biopsy, the same mutations were reported in 94%-100% of plasma samples.15
Studies of the clinical utility of ctDNA and CTCs are among ongoing clinical trials of liquid biopsies (Tables 2 and 3). The potential for using CTCs to guide treatment decisions has become particularly relevant in breast cancer in light of results showing that patients with primary tumors that are negative for human epidermal growth factor receptor 2 (HER2) amplification, an important biomarker in breast cancer, may have CTCs that are HER2-positive, in up to 30% of cases. These patients may therefore still benefit from HER2-targeted therapy.16
The DETECT studies are the first phase 3 trials in which treatment decisions are being based on the phenotypic characteristics of CTCs. DETECT III (NCT01619111) is comparing lapatinib in combination with standard therapy with standard therapy alone in patients with HER2-negative metastatic breast cancer who have HER2-positive CTCs, whereas DETECT IV (NCT02035813) is enrolling patients with HER2-negative, hormone receptor-positive metastatic breast cancer and persistent HER2-negative CTCs to receive standard endocrine therapy and the mammalian target of rapamycin inhibitor everolimus.
Other targets and sources for liquid biopsy
Another approach to liquid biopsies that is also beginning to take off is to collect tumor-derived exosomes from the bloodstream. Exosomes are tiny, fluid-filled, membrane-bound sacks that bud off from the surface of a cell to expel waste or to transport cargo from one cell to another. DNA, RNA, and protein can be extracted from tumor-derived exosomes and could also serve as molecular biomarkers relating to the cancer cells from which they came.6,7
Exosome Diagnostics is bringing the first exosome-based diagnostic tests to the market and recently teamed up with Amgen for the development of these liquid biopsies.17 In January 2016, they launched ExoDx Lung (ALK), for detection of EML4-ALK gene fusions in patients with NSCLC, using a proprietary platform for the isolation of RNA from exosomes. Data that was presented at several different conferences in 2015 demonstrated a sensitivity of 88% and specificity of 100% for this diagnostic when compared with tissue ALK status in NSCLC patients receiving a second-generation ALK inhibitor following progression on prior ALK inhibitor therapy.18
In September, they subsequently announced the launch of a test that analyses genetic information from exosomes collected from a urine sample taken from prostate cancer patients. Using a 3-gene signature, in combination with a proprietary algorithm, this diagnostic generates a score assessing a prostate cancer patient’s risk for higher grade, more aggressive disease. It is designed to complement the prostate-specific antigen score and has demonstrated accuracy in ruling out the presence of high-grade cancer before an initial biopsy in more than 1,
1. Lennon NK, Adalsteinsson VA, Gabriel SB. Technological considerations for genome-guided diagnosis and management of cancer. Genome Med. 2016;8:112.
2. MIT Technology Review website. Liquid biopsy: fast DNA-sequencing machines are leading to simple blood tests for cancer. https://www.technologyreview.com/s/534991/liquid-biopsy/. Published 2015. Accessed December 19, 2016.
3. Alix-Panabières C and Pantel K. Clinical applications of circulating tumor cells and circulating tumor DNA as liquid biopsy. Cancer Discov. 2016;6(5):479-491.
4. Calabuig-Farinãs S, Jantus-Lewintre E, Herreros-Pomares A, Camps C. Circulating tumor cells versus circulating tumor DNA in lung cancer – which one will win? Transl Lung Cancer Res. 2016;5(5):466-482.
5. Karabacak, NM, Spuhler PS, Fachin F, et al. Microfluidic, marker-free isolation of circulating tumor cells from blood samples. Nat Protoc. 2014;9:694-710.
6. Buder A, Tomuta C, and Filipits M. The potential of liquid biopsies. Curr Opin Oncol. 2016;28:130-134.
7. Hofman P, Popper HH. Pathologists and liquid biopsies: to be or not to be? Virchows Arch. 2016;469:601-609.
8. Bidard FC, Peeters DJ, Fehm T, et al. Clinical validity of circulating tumor cells in patients with metastatic breast cancer: a pooled analysis of individual patient data. Lancet Oncol. 2014;15(4):406-414.
9. de Bono JS, Scher HI, Montgomery RB, et al. Circulating tumor cells predict survival benefit from treatment in metastatic castration-resistant prostate cancer. Clin Cancer Res. 2008;14(19):6302-6309.
10. Cohen SJ, Punt CJ, Iannotti N, et al. Relationship of circulating tumor cells to tumor response, progression-free survival, and overall survival in patients with metastatic colorectal cancer. J Clin Oncol. 2008;26(19):3213-3221.
11. CellSearch Web site. What is the CELLSEARCH® System? https://www.cellsearchctc.com/product-systems-overview/cellsearch-system-overview. Last updated December 5th, 2016. Accessed online December 19th, 2016.
12. CellSearch Web site [advertisement]. https://www.cellsearchctc.com/clinical-applications/clinical-applications-overview. Last updated December 5, 2016. Accessed December 19, 2016.
13. US Food and Drug Administration. cobas EGFR Mutation Test v2 – P150047. http://www.fda.gov/MedicalDevices/ProductsandMedicalProcedures/DeviceApprovalsandClearances/Recently-ApprovedDevices/ucm519922.htm. Last updated September 9, 2016. Accessed December 19, 2016.
14. Wu YL, Zhou C, Liam CK, et al. First-line erlotinib versus gemcitabine/cisplatin in patients with advanced EGFR mutation-positive non-small cell lung cancer: analyses from the phase III, randomized, open-label, ENSURE study. Ann Oncol. 2015;26(9):1883-1889.
15. Zill OA, Mortimer S, Banks KC, et al. Somatic genomic landscape of over 15,000 patients with advanced-stage cancer from clinical next-generation sequencing analysis of circulating tumor DNA. J Clin Oncol. 2016;34(suppl;abstr LBA11501).
16. Jordan NV, Bardia A, Wittner BS, et al. HER2 expression identifies dynamic functional states within circulating breast cancer cells. Nature. 2016;537:102-106.
17. Exosome Diagnostics. Exosome diagnostics enters agreement with Amgen. http://www.exosomedx.com/news-events/press-releases/exosome-diagnostics-enters-agreement-amgen. Published October 3, 2016. Accessed December 19, 2016.
18. Brinkman K, Emenegger J, Tannous B, et al. Exosomal RNA-based liquid biopsy detection of EML4-ALK in plasma from NSCLC patients [2015 World Conference on Lung Cancer, Denver, CO; abstract 2591]. http://library.iaslc.org/search-speaker?search_speaker=30493. Accessed January 6, 2017.
19. Exosome Diagnostics website. Prostate cancer. http://www.exosomedx.com/prostate-cancer-0. Last updated 2017. Accessed online December 19, 2016.
1. Lennon NK, Adalsteinsson VA, Gabriel SB. Technological considerations for genome-guided diagnosis and management of cancer. Genome Med. 2016;8:112.
2. MIT Technology Review website. Liquid biopsy: fast DNA-sequencing machines are leading to simple blood tests for cancer. https://www.technologyreview.com/s/534991/liquid-biopsy/. Published 2015. Accessed December 19, 2016.
3. Alix-Panabières C and Pantel K. Clinical applications of circulating tumor cells and circulating tumor DNA as liquid biopsy. Cancer Discov. 2016;6(5):479-491.
4. Calabuig-Farinãs S, Jantus-Lewintre E, Herreros-Pomares A, Camps C. Circulating tumor cells versus circulating tumor DNA in lung cancer – which one will win? Transl Lung Cancer Res. 2016;5(5):466-482.
5. Karabacak, NM, Spuhler PS, Fachin F, et al. Microfluidic, marker-free isolation of circulating tumor cells from blood samples. Nat Protoc. 2014;9:694-710.
6. Buder A, Tomuta C, and Filipits M. The potential of liquid biopsies. Curr Opin Oncol. 2016;28:130-134.
7. Hofman P, Popper HH. Pathologists and liquid biopsies: to be or not to be? Virchows Arch. 2016;469:601-609.
8. Bidard FC, Peeters DJ, Fehm T, et al. Clinical validity of circulating tumor cells in patients with metastatic breast cancer: a pooled analysis of individual patient data. Lancet Oncol. 2014;15(4):406-414.
9. de Bono JS, Scher HI, Montgomery RB, et al. Circulating tumor cells predict survival benefit from treatment in metastatic castration-resistant prostate cancer. Clin Cancer Res. 2008;14(19):6302-6309.
10. Cohen SJ, Punt CJ, Iannotti N, et al. Relationship of circulating tumor cells to tumor response, progression-free survival, and overall survival in patients with metastatic colorectal cancer. J Clin Oncol. 2008;26(19):3213-3221.
11. CellSearch Web site. What is the CELLSEARCH® System? https://www.cellsearchctc.com/product-systems-overview/cellsearch-system-overview. Last updated December 5th, 2016. Accessed online December 19th, 2016.
12. CellSearch Web site [advertisement]. https://www.cellsearchctc.com/clinical-applications/clinical-applications-overview. Last updated December 5, 2016. Accessed December 19, 2016.
13. US Food and Drug Administration. cobas EGFR Mutation Test v2 – P150047. http://www.fda.gov/MedicalDevices/ProductsandMedicalProcedures/DeviceApprovalsandClearances/Recently-ApprovedDevices/ucm519922.htm. Last updated September 9, 2016. Accessed December 19, 2016.
14. Wu YL, Zhou C, Liam CK, et al. First-line erlotinib versus gemcitabine/cisplatin in patients with advanced EGFR mutation-positive non-small cell lung cancer: analyses from the phase III, randomized, open-label, ENSURE study. Ann Oncol. 2015;26(9):1883-1889.
15. Zill OA, Mortimer S, Banks KC, et al. Somatic genomic landscape of over 15,000 patients with advanced-stage cancer from clinical next-generation sequencing analysis of circulating tumor DNA. J Clin Oncol. 2016;34(suppl;abstr LBA11501).
16. Jordan NV, Bardia A, Wittner BS, et al. HER2 expression identifies dynamic functional states within circulating breast cancer cells. Nature. 2016;537:102-106.
17. Exosome Diagnostics. Exosome diagnostics enters agreement with Amgen. http://www.exosomedx.com/news-events/press-releases/exosome-diagnostics-enters-agreement-amgen. Published October 3, 2016. Accessed December 19, 2016.
18. Brinkman K, Emenegger J, Tannous B, et al. Exosomal RNA-based liquid biopsy detection of EML4-ALK in plasma from NSCLC patients [2015 World Conference on Lung Cancer, Denver, CO; abstract 2591]. http://library.iaslc.org/search-speaker?search_speaker=30493. Accessed January 6, 2017.
19. Exosome Diagnostics website. Prostate cancer. http://www.exosomedx.com/prostate-cancer-0. Last updated 2017. Accessed online December 19, 2016.
Advances in Targeted Therapy for Breast Cancer
It is estimated that there were more than 3.1 million women living in the U.S. with a history of invasive breast cancer as of January 1, 2014, and an additional 231,840 women will be newly diagnosed with invasive breast cancer in 2015.1,2 The median age at the time of breast cancer diagnosis is 61 years. About 20% of breast cancers occur among women aged < 50 years, and 43% occur in women aged > 65 years.
The treatment and prognosis for breast cancer depend on the stage at diagnosis, the biologic characteristics of the tumor, and the age and health of the patient. The overall 5-year relative survival rate for female patients with breast cancer has improved from 75% to 90% from 1975 to 1977 and from 2003 to 2009, respectively, largely due to improvements in treatment (ie, chemotherapy, hormone therapy, and targeted drugs) and because of earlier diagnosis resulting from the widespread use of mammography and other screening tools.2
Estrogen Receptor-Positive Therapies
Women with breast cancer who test positive for hormone receptors are candidates for treatment with hormone therapy to reduce the likelihood of recurrence or as a core component of treatment for advanced disease. Currently available endocrine strategies for the treatment of estrogen receptor- (ER) positive breast cancer include targeting the ER with the antiestrogen drug tamoxifen. Another option is suppressing the amount of available ligand (estrogen) for the receptor either with gonadal suppression in premenopausal oophorectomy, or luteinizing hormonereleasing hormone agonists, or with the aromatase inhibitors (AIs) anastrozole, exemestane, and letrozole in postmenopausal women and by downregulating the receptor with fulvestrant. Given their proven efficacy and generally favorable adverse effect (AE) profile, these endocrine therapies are widely used in the treatment of both early-stage and recurrent and/or metastatic breast cancer.
Recent studies have offered new treatments for patients with hormone receptor-positive, human epidermal growth factor receptor 2 (HER2)-negative breast cancer. Innovative hormonal and targeted therapies for advanced disease as well as new data on adjuvant hormonal therapy for young high-risk patients are changing the available therapeutic options.
Advanced Metastatic Treatments
Treatment for metastatic hormone receptor-positive breast cancer has shifted from traditional cytotoxic chemotherapies to targeted therapeutic options. Most treatment guidelines, including the National Comprehensive Cancer Network guidelines, recommend targeted therapy with AIs or selective ER modulators rather than chemotherapy, except in the case of visceral crisis.3
Until recently, there had been relatively little guidance to inform which hormonal therapy was most appropriate. Aromatase inhibitors were generally reserved for postmenopausal women, whereas tamoxifen was preferred in premenopausal women.
Fulvestrant
The FDA initially approved fulvestrant, a hormone receptor downregulator, in 2002 at a 250-mg dose, following progression on an anti-estrogen therapy, such as tamoxifen in postmenopausal women with stage IV breast cancer. The FDA approval was based on similar response rates for the already approved agent anastrozole.4 However, pharmacokinetic findings from the phase 3 EFECT trial in 2008 prompted researchers to explore a 500-mg dose of fulvestrant.5
The recently published FIRST study is a phase 2, randomized, open-label study comparing fulvestrant 500 mg with anastrozole 1 mg as first-line hormonal therapy for postmenopausal women with hormone receptorpositive advanced breast cancer. Fulvestrant was given 500 mg once monthly with an extra dose given on day 14 of month 1. The trial enrolled 233 patients. The median time to progression was 23.4 months for fulvestrant and 13.1 months for anastrozole. These results translate into a 34% reduction in the risk of progression.6
These outcomes suggest that fulvestrant is as viable and perhaps even preferred first-line therapy for postmenopausal women with hormone receptor-positive, HER2-negative advanced breast cancer. The impressive results from this trial are likely, because the study used the 500-mg dose of fulvestrant, which is twice the dose used in the original trials. However, the 500-mg dose has previously been studied, and long-term outcome data suggest both safety and efficiency. The large randomized, double-blinded phase 3 CONFIRM trial, published in 2013, compared the 250-mg dose with the 500-mg dose and found that the higher dose was associated with a 19% reduction in the risk of death and a 4.1 month increase in median overall survival (OS) without any new safety concerns.5
Palbociclib
The FDA recently granted accelerated approval to palbociclib in combination with letrozole for the first-line therapy of advanced hormone receptor-positive, HER2-negative breast cancer in postmenopausal women. Palbociclib is an oral small-molecular inhibitor of cyclindependent kinases 4 and 6. Preclinical data suggested synergy with anti-estrogen therapies and inhibition of breast cancer cell growth.7
A phase 2, open-label randomized trial (PALOMA-1/TRIO-18) enrolled 165 patients. Progression-free survival (PFS) was 20.2 months for the palbociclib plus letrozole arm and 10.2 months for the letrozole alone arm. Significant toxicities were noted in the palbociclib arm, including 54% of people experiencing grade 3 to 4 neutropenia (vs 1% in the letrozole arm), leukopenia in 19% (vs 0%) and fatigue in 4% (vs 1%). A phase 3 trial is currently enrolling patients.7 While we await the results of the phase 3 trial and long-term follow-up data, palbociclib plus letrozole is a new, viable option for metastatic hormone receptor-positive advanced breast cancer.
Although many practitioners will continue to reasonably use any AI or selective ER modulator when treating metastatic breast cancer, both fulvestrant and palbociclib in combination with letrozole are new evidence-based, first-line options worth considering.
Early-Stage Treatment Options
There are many acceptable therapeutic options for treating early stage breast cancer. Tamoxifen has traditionally been used in the adjuvant setting for premenopausal women, whereas AIs are often used in postmenopausal women. There has also been a long-standing debate about the role of ovarian suppression in premenopausal women.
The recently published phase 3 TEXT and SOFT trials attempted to provide answers to these long-standing therapeutic dilemmas. The SOFT trial randomly assigned 3,066 premenopausal women to 5 years of tamoxifen, 5 years of tamoxifen plus ovarian suppression, or exemestane plus ovarian suppression. The TEXT trial randomly assigned 2,672 women to receive either exemestane plus ovarian suppression or tamoxifen plus ovarian suppression. The studies showed that subjecting all women receiving tamoxifen to ovarian suppression did not provide any significant benefit.8,9
However, the subgroup of women with high-risk disease who required adjuvant chemotherapy and remained premenopausal experienced improved outcomes from ovarian suppression. This high-risk subgroup when given tamoxifen plus ovarian suppression had a 4.5% absolute reduction in breast cancer recurrence at 5 years compared with the group that received tamoxifen alone. When this high-risk subgroup was given exemestane plus ovarian suppression, the women had a 7.7% absolute reduction in breast cancer recurrence at 5 years compared with the group that received tamoxifen alone.8
Ovarian suppression resulted in significant additional AEs, including depression and menopausal symptoms. The authors of the study also pointed out the additional risk of hypertension, musculoskeletal AEs, and decreased bone density. Furthermore, the OS data from these studies are premature, because the patients had fewer AEs than initially anticipated; this resulted in an only 5% mortality at publication.
The study design also raised several interesting questions. The primary endpoint was disease-free survival. The authors defined this as the time from randomization to the first appearance of invasive recurrence of breast cancer (local, regional, or distant), invasive contralateral breast cancer, second (non-breast) invasive cancer, or death without breast cancer recurrence or second invasive cancer. When studying adjuvant therapy for diseases, such as breast cancer, which carry long-term survival, studies often use PFS with various modified definitions as a surrogate marker for OS. Clinicians are then left to decide whether this surrogate marker is an accurate predictor of OS or other important clinical outcomes.
In the combined analysis of the TEXT and SOFT trials, only 60% of the first recurrences, second invasive cancers, or deaths involved recurrence of breast cancer
at a distant site.9 Because locally recurrent breast cancer is highly treatable and often curable, clinicians must ask whether the increased toxicities of ovarian suppression are worth the large number of women who experienced local recurrence given the still relatively small absolute reduction in recurrence risk.
Last, the study authors retrospectively reviewed data from the International Breast Cancer Study Group and U.S. Intergroup trials and concluded that women aged < 35 years were most likely to be at high-risk for AEs.10,11 A subgroup analysis of women aged < 35 years in the SOFT trial noted that breast cancer recurred within 5 years in one-third of women receiving tamoxifen alone, whereas only in one-sixth of women receiving exemestane plus ovarian suppression.8 This is the basis for the conclusion that premenopausal women, particularly those aged < 35 years, with high-risk disease who receive chemotherapy and remain premenopausal after chemotherapy, benefit from ovarian suppression in combination with tamoxifen, and even more impressively from ovarian suppression combined with exemestane.
The problem is that the study did not risk-stratify patients based on those aged < 35 years, and the conclusion is based on a subgroup analysis using a primary endpoint that may not accurately predict OS. Nonetheless, although not definitive, the data from the TEXT and SOFT trials raise interesting therapeutic questions that require further study and certainly provide tempting therapeutic options in patients who are clinically at high risk for recurrence.
HER2-Positive Breast Cancer
Up to 20% of invasive breast cancers are a result of HER2 gene amplification or overexpression of the HER2 protein, a tyrosine kinase transmembrane receptor, resulting in a more aggressive phenotype and a poor prognosis. Anti-HER2 drugs have changed the landscape of the disease previously known as aggressive breast cancer with a poor survival rate.
Treatment with the anti-HER2 humanized monoclonal antibody trastuzumab in addition to chemotherapy, compared with chemotherapy alone, significantly improves PFS and OS among patients with HER2-positive metastatic as well as early breast cancer. However, in most patients with HER2-positive metastatic breast cancer, the disease progresses, highlighting the need for new, targeted therapies for advanced disease.
New Standard of Care
The original studies of trastuzumab showed improved OS in late-stage (metastatic) breast cancer from 20.3 to 25.1 months, and in early-stage breast cancer, it reduced the risk of cancer returning after surgery by an absolute risk of 9.5% and the risk of death by an absolute risk of 3%.
New therapies directed at HER2 are being developed, among them pertuzumab, a humanized monoclonal antibody that binds HER2 at a different epitope of the HER2 extracellular domain (subdomain 2) than that at which trastuzumab binds. Pertuzumab prevents HER2 from dimerizing with other ligand-activated HER receptors, most notably HER3. Like trastuzumab, pertuzumab stimulates antibody-dependent, cell-mediated cytotoxicity. Because pertuzumab and trastuzumab bind to different HER2 epitopes and have complementary mechanisms of action, these 2 agents, when given together, provide a more comprehensive blockade of HER2 signaling and result in greater antitumor activity than does either agent alone in HER2-positive tumor models.12 In phase 2 studies, a pertuzumab–trastuzumab regimen has shown activity in patients with HER2-positive metastatic breast cancer and in patients with early breast cancer.13
In the phase 3 CLEOPATRA study, the combination of pertuzumab plus trastuzumab plus docetaxel, used as first-line treatment for HER2-positive metastatic breast cancer compared with placebo plus trastuzumab plus docetaxel, significantly prolonged PFS (18.5 months vs 12.4 months), with no increase in cardiac toxic effects.12 In a recent updated follow-up of the CLEOPATRA study, the addition of pertuzumab to trastuzumab and docetaxel showed a significantly better median OS (56.5 months vs 40.8 months; hazard ratio, 0.68; P < .001).14 From these results, this combination regimen is now considered a first-line therapy for patients with HER2-positive metastatic breast cancer.
However, the cost of cancer treatment has become a mounting concern during the past decade, as new therapies come down the pipeline with ever-increasing price tags. Trastuzumab costs about $4,500 a month, and the newer pertuzumab runs about 30% higher, at $6,000 a month. For a full course of treatment, the cost of the pertuzumab and trastuzumab combination could go as high as $195,000, depending on the duration of therapy and the choice of taxanes.
Conclusions
The landscape of therapeutic options in high-risk, young patients with early-stage breast cancer as well as patients with advanced or metastatic disease is changing rapidly.
Clinicians now have 2 new first-line options for the treatment of advanced hormone receptor-positive, HER2-negative breast cancer. A phase 3 trial demonstrated that fulvestrant monotherapy offers improved PFS and some improvement in OS compared with anastrazole in postmenopausal women. A phase 2 trial showed that palbociclib plus letrozole offers improved PFS in postmenopausal women. Based on the SOFT and TEXT trials, clinicians treating high-risk premenopausal women now have some data to inform the debate about whether ovarian suppression should be added to hormone therapy.
Based on the CLEOPATRA trial, clinicians can now consider combination pertuzumab and trastuzumab and docetaxel as first-line therapy for patients with HER2-positive metastatic breast cancer.
Author disclosures
The authors report no actual or potential conflicts of interest with regard to this article.
Disclaimer
The opinions expressed herein are those of the authors and do not necessarily reflect those of Federal Practitioner, Frontline Medical Communications Inc., the U.S. Government, or any of its agencies. This article may discuss unlabeled or investigational use of certain drugs. Please review complete prescribing information for specific drugs or drug combinations—including indications, contraindications, warnings, and adverse effects—before administering pharmacologic therapy to patients.
Click here to read the digital edition.
1. American Cancer Society. Cancer facts & figures, 2015. Atlanta, GA: American Cancer Society; 2015.
2. American Cancer Society. Cancer treatment & survivorship facts & figures, 2014-2015. Atlanta, GA: American Cancer Society; 2014.
3. National Comprehensive Cancer Network. NCCN clinical Practice guidelines in oncology: breast Cancer. Version 1. 2015. Fort Washington, PA: National Comprehensive Cancer Network; 2015:BINV-19.
4. Howell A, Robertson JF, Quaresma Albano J. Fulvestrant, formerly ICI 182,780, is as effective as anastrozole in postmenopausal women with advanced breast cancer progressing after prior endocrine treatment. J Clin Oncol. 2002;20(16):3396-3403.
5. Di Leo A, Jerusalem G, Petruzelka L, et al. Final overall survival: fulvestrant 500 mg vs 250 mg in the randomized CONFIRM trial. J Natl Cancer Inst. 2014;106(1):djt337.
6. Robertson JF, Lindemann JB, Llombart-Cussac A, et al. Fulvestrant 500 mg versus anastrozole 1 mg for the first-line treatment of advanced breast cancer: follow-up analysis from the randomized ‘FIRST’ study. Breast Cancer Res Treat. 2012;136(2):503-511.
7. Finn RS, Crown JP, Lang I, et al. The cyclin-dependent kinase 4/6 inhibitor palbociclib in combination with letrozole versus letrozole alone as first-line treatment of oestrogen receptor-positive, HER2-negative, advanced breast cancer (PALOMA-1/TRIO-18): a randomised phase 2 study. Lancet Oncol. 2015;16(1):25-35.
8. Francis PA, Regan MM, Fleming GF, et al; SOFT Investigators; International Breast Cancer Study Group. Adjuvant ovarian suppression in premenopausal breast cancer. N Engl J Med. 2015;372(5):436-446.
9. Pagani O. Regan MM, Walley BA, et al. TEXT and SOFT Investigators; International Breast Cancer Study Group. Adjuvant exemestane with ovarian suppression in premenopausal breast cancer. N Engl J Med. 2014;371(2):107-118.
10. Aebi S, Gelber S, Castiglione-Gertsch M, et al. Is chemotherapy alone adequate for young women with oestrogen-receptor-positive breast cancer? Lancet. 2000;355:1869-1874.
11. Goldhirsch A, Gelber RD, Yothers G, et al. Adjuvant therapy for very young women with breast cancer: need for tailored treatments. J Natl Cancer Inst Monogr. 2001;(30):44-51
12. Hudis CA. Trastuzumab—mechanism of action and use in clinical practice. N Engl J Med. 2007;357(1):39-51.
13. Baselga J, Cortés J, Kim SB, et al; CLEOPATRA Study Group. Pertuzumab plus trastuzumab plus docetaxel for metastatic breast cancer. N Engl J Med. 2012;366(2):109-119.
14. Swain SM, Baselga J, Kim SB, et al; CLEOPATRA Study Group. Pertuzumab, trastuzumab, and docetaxel in HER2-positive metastatic breast cancer. N Engl J Med. 2015;372(8):724-734.
It is estimated that there were more than 3.1 million women living in the U.S. with a history of invasive breast cancer as of January 1, 2014, and an additional 231,840 women will be newly diagnosed with invasive breast cancer in 2015.1,2 The median age at the time of breast cancer diagnosis is 61 years. About 20% of breast cancers occur among women aged < 50 years, and 43% occur in women aged > 65 years.
The treatment and prognosis for breast cancer depend on the stage at diagnosis, the biologic characteristics of the tumor, and the age and health of the patient. The overall 5-year relative survival rate for female patients with breast cancer has improved from 75% to 90% from 1975 to 1977 and from 2003 to 2009, respectively, largely due to improvements in treatment (ie, chemotherapy, hormone therapy, and targeted drugs) and because of earlier diagnosis resulting from the widespread use of mammography and other screening tools.2
Estrogen Receptor-Positive Therapies
Women with breast cancer who test positive for hormone receptors are candidates for treatment with hormone therapy to reduce the likelihood of recurrence or as a core component of treatment for advanced disease. Currently available endocrine strategies for the treatment of estrogen receptor- (ER) positive breast cancer include targeting the ER with the antiestrogen drug tamoxifen. Another option is suppressing the amount of available ligand (estrogen) for the receptor either with gonadal suppression in premenopausal oophorectomy, or luteinizing hormonereleasing hormone agonists, or with the aromatase inhibitors (AIs) anastrozole, exemestane, and letrozole in postmenopausal women and by downregulating the receptor with fulvestrant. Given their proven efficacy and generally favorable adverse effect (AE) profile, these endocrine therapies are widely used in the treatment of both early-stage and recurrent and/or metastatic breast cancer.
Recent studies have offered new treatments for patients with hormone receptor-positive, human epidermal growth factor receptor 2 (HER2)-negative breast cancer. Innovative hormonal and targeted therapies for advanced disease as well as new data on adjuvant hormonal therapy for young high-risk patients are changing the available therapeutic options.
Advanced Metastatic Treatments
Treatment for metastatic hormone receptor-positive breast cancer has shifted from traditional cytotoxic chemotherapies to targeted therapeutic options. Most treatment guidelines, including the National Comprehensive Cancer Network guidelines, recommend targeted therapy with AIs or selective ER modulators rather than chemotherapy, except in the case of visceral crisis.3
Until recently, there had been relatively little guidance to inform which hormonal therapy was most appropriate. Aromatase inhibitors were generally reserved for postmenopausal women, whereas tamoxifen was preferred in premenopausal women.
Fulvestrant
The FDA initially approved fulvestrant, a hormone receptor downregulator, in 2002 at a 250-mg dose, following progression on an anti-estrogen therapy, such as tamoxifen in postmenopausal women with stage IV breast cancer. The FDA approval was based on similar response rates for the already approved agent anastrozole.4 However, pharmacokinetic findings from the phase 3 EFECT trial in 2008 prompted researchers to explore a 500-mg dose of fulvestrant.5
The recently published FIRST study is a phase 2, randomized, open-label study comparing fulvestrant 500 mg with anastrozole 1 mg as first-line hormonal therapy for postmenopausal women with hormone receptorpositive advanced breast cancer. Fulvestrant was given 500 mg once monthly with an extra dose given on day 14 of month 1. The trial enrolled 233 patients. The median time to progression was 23.4 months for fulvestrant and 13.1 months for anastrozole. These results translate into a 34% reduction in the risk of progression.6
These outcomes suggest that fulvestrant is as viable and perhaps even preferred first-line therapy for postmenopausal women with hormone receptor-positive, HER2-negative advanced breast cancer. The impressive results from this trial are likely, because the study used the 500-mg dose of fulvestrant, which is twice the dose used in the original trials. However, the 500-mg dose has previously been studied, and long-term outcome data suggest both safety and efficiency. The large randomized, double-blinded phase 3 CONFIRM trial, published in 2013, compared the 250-mg dose with the 500-mg dose and found that the higher dose was associated with a 19% reduction in the risk of death and a 4.1 month increase in median overall survival (OS) without any new safety concerns.5
Palbociclib
The FDA recently granted accelerated approval to palbociclib in combination with letrozole for the first-line therapy of advanced hormone receptor-positive, HER2-negative breast cancer in postmenopausal women. Palbociclib is an oral small-molecular inhibitor of cyclindependent kinases 4 and 6. Preclinical data suggested synergy with anti-estrogen therapies and inhibition of breast cancer cell growth.7
A phase 2, open-label randomized trial (PALOMA-1/TRIO-18) enrolled 165 patients. Progression-free survival (PFS) was 20.2 months for the palbociclib plus letrozole arm and 10.2 months for the letrozole alone arm. Significant toxicities were noted in the palbociclib arm, including 54% of people experiencing grade 3 to 4 neutropenia (vs 1% in the letrozole arm), leukopenia in 19% (vs 0%) and fatigue in 4% (vs 1%). A phase 3 trial is currently enrolling patients.7 While we await the results of the phase 3 trial and long-term follow-up data, palbociclib plus letrozole is a new, viable option for metastatic hormone receptor-positive advanced breast cancer.
Although many practitioners will continue to reasonably use any AI or selective ER modulator when treating metastatic breast cancer, both fulvestrant and palbociclib in combination with letrozole are new evidence-based, first-line options worth considering.
Early-Stage Treatment Options
There are many acceptable therapeutic options for treating early stage breast cancer. Tamoxifen has traditionally been used in the adjuvant setting for premenopausal women, whereas AIs are often used in postmenopausal women. There has also been a long-standing debate about the role of ovarian suppression in premenopausal women.
The recently published phase 3 TEXT and SOFT trials attempted to provide answers to these long-standing therapeutic dilemmas. The SOFT trial randomly assigned 3,066 premenopausal women to 5 years of tamoxifen, 5 years of tamoxifen plus ovarian suppression, or exemestane plus ovarian suppression. The TEXT trial randomly assigned 2,672 women to receive either exemestane plus ovarian suppression or tamoxifen plus ovarian suppression. The studies showed that subjecting all women receiving tamoxifen to ovarian suppression did not provide any significant benefit.8,9
However, the subgroup of women with high-risk disease who required adjuvant chemotherapy and remained premenopausal experienced improved outcomes from ovarian suppression. This high-risk subgroup when given tamoxifen plus ovarian suppression had a 4.5% absolute reduction in breast cancer recurrence at 5 years compared with the group that received tamoxifen alone. When this high-risk subgroup was given exemestane plus ovarian suppression, the women had a 7.7% absolute reduction in breast cancer recurrence at 5 years compared with the group that received tamoxifen alone.8
Ovarian suppression resulted in significant additional AEs, including depression and menopausal symptoms. The authors of the study also pointed out the additional risk of hypertension, musculoskeletal AEs, and decreased bone density. Furthermore, the OS data from these studies are premature, because the patients had fewer AEs than initially anticipated; this resulted in an only 5% mortality at publication.
The study design also raised several interesting questions. The primary endpoint was disease-free survival. The authors defined this as the time from randomization to the first appearance of invasive recurrence of breast cancer (local, regional, or distant), invasive contralateral breast cancer, second (non-breast) invasive cancer, or death without breast cancer recurrence or second invasive cancer. When studying adjuvant therapy for diseases, such as breast cancer, which carry long-term survival, studies often use PFS with various modified definitions as a surrogate marker for OS. Clinicians are then left to decide whether this surrogate marker is an accurate predictor of OS or other important clinical outcomes.
In the combined analysis of the TEXT and SOFT trials, only 60% of the first recurrences, second invasive cancers, or deaths involved recurrence of breast cancer
at a distant site.9 Because locally recurrent breast cancer is highly treatable and often curable, clinicians must ask whether the increased toxicities of ovarian suppression are worth the large number of women who experienced local recurrence given the still relatively small absolute reduction in recurrence risk.
Last, the study authors retrospectively reviewed data from the International Breast Cancer Study Group and U.S. Intergroup trials and concluded that women aged < 35 years were most likely to be at high-risk for AEs.10,11 A subgroup analysis of women aged < 35 years in the SOFT trial noted that breast cancer recurred within 5 years in one-third of women receiving tamoxifen alone, whereas only in one-sixth of women receiving exemestane plus ovarian suppression.8 This is the basis for the conclusion that premenopausal women, particularly those aged < 35 years, with high-risk disease who receive chemotherapy and remain premenopausal after chemotherapy, benefit from ovarian suppression in combination with tamoxifen, and even more impressively from ovarian suppression combined with exemestane.
The problem is that the study did not risk-stratify patients based on those aged < 35 years, and the conclusion is based on a subgroup analysis using a primary endpoint that may not accurately predict OS. Nonetheless, although not definitive, the data from the TEXT and SOFT trials raise interesting therapeutic questions that require further study and certainly provide tempting therapeutic options in patients who are clinically at high risk for recurrence.
HER2-Positive Breast Cancer
Up to 20% of invasive breast cancers are a result of HER2 gene amplification or overexpression of the HER2 protein, a tyrosine kinase transmembrane receptor, resulting in a more aggressive phenotype and a poor prognosis. Anti-HER2 drugs have changed the landscape of the disease previously known as aggressive breast cancer with a poor survival rate.
Treatment with the anti-HER2 humanized monoclonal antibody trastuzumab in addition to chemotherapy, compared with chemotherapy alone, significantly improves PFS and OS among patients with HER2-positive metastatic as well as early breast cancer. However, in most patients with HER2-positive metastatic breast cancer, the disease progresses, highlighting the need for new, targeted therapies for advanced disease.
New Standard of Care
The original studies of trastuzumab showed improved OS in late-stage (metastatic) breast cancer from 20.3 to 25.1 months, and in early-stage breast cancer, it reduced the risk of cancer returning after surgery by an absolute risk of 9.5% and the risk of death by an absolute risk of 3%.
New therapies directed at HER2 are being developed, among them pertuzumab, a humanized monoclonal antibody that binds HER2 at a different epitope of the HER2 extracellular domain (subdomain 2) than that at which trastuzumab binds. Pertuzumab prevents HER2 from dimerizing with other ligand-activated HER receptors, most notably HER3. Like trastuzumab, pertuzumab stimulates antibody-dependent, cell-mediated cytotoxicity. Because pertuzumab and trastuzumab bind to different HER2 epitopes and have complementary mechanisms of action, these 2 agents, when given together, provide a more comprehensive blockade of HER2 signaling and result in greater antitumor activity than does either agent alone in HER2-positive tumor models.12 In phase 2 studies, a pertuzumab–trastuzumab regimen has shown activity in patients with HER2-positive metastatic breast cancer and in patients with early breast cancer.13
In the phase 3 CLEOPATRA study, the combination of pertuzumab plus trastuzumab plus docetaxel, used as first-line treatment for HER2-positive metastatic breast cancer compared with placebo plus trastuzumab plus docetaxel, significantly prolonged PFS (18.5 months vs 12.4 months), with no increase in cardiac toxic effects.12 In a recent updated follow-up of the CLEOPATRA study, the addition of pertuzumab to trastuzumab and docetaxel showed a significantly better median OS (56.5 months vs 40.8 months; hazard ratio, 0.68; P < .001).14 From these results, this combination regimen is now considered a first-line therapy for patients with HER2-positive metastatic breast cancer.
However, the cost of cancer treatment has become a mounting concern during the past decade, as new therapies come down the pipeline with ever-increasing price tags. Trastuzumab costs about $4,500 a month, and the newer pertuzumab runs about 30% higher, at $6,000 a month. For a full course of treatment, the cost of the pertuzumab and trastuzumab combination could go as high as $195,000, depending on the duration of therapy and the choice of taxanes.
Conclusions
The landscape of therapeutic options in high-risk, young patients with early-stage breast cancer as well as patients with advanced or metastatic disease is changing rapidly.
Clinicians now have 2 new first-line options for the treatment of advanced hormone receptor-positive, HER2-negative breast cancer. A phase 3 trial demonstrated that fulvestrant monotherapy offers improved PFS and some improvement in OS compared with anastrazole in postmenopausal women. A phase 2 trial showed that palbociclib plus letrozole offers improved PFS in postmenopausal women. Based on the SOFT and TEXT trials, clinicians treating high-risk premenopausal women now have some data to inform the debate about whether ovarian suppression should be added to hormone therapy.
Based on the CLEOPATRA trial, clinicians can now consider combination pertuzumab and trastuzumab and docetaxel as first-line therapy for patients with HER2-positive metastatic breast cancer.
Author disclosures
The authors report no actual or potential conflicts of interest with regard to this article.
Disclaimer
The opinions expressed herein are those of the authors and do not necessarily reflect those of Federal Practitioner, Frontline Medical Communications Inc., the U.S. Government, or any of its agencies. This article may discuss unlabeled or investigational use of certain drugs. Please review complete prescribing information for specific drugs or drug combinations—including indications, contraindications, warnings, and adverse effects—before administering pharmacologic therapy to patients.
Click here to read the digital edition.
It is estimated that there were more than 3.1 million women living in the U.S. with a history of invasive breast cancer as of January 1, 2014, and an additional 231,840 women will be newly diagnosed with invasive breast cancer in 2015.1,2 The median age at the time of breast cancer diagnosis is 61 years. About 20% of breast cancers occur among women aged < 50 years, and 43% occur in women aged > 65 years.
The treatment and prognosis for breast cancer depend on the stage at diagnosis, the biologic characteristics of the tumor, and the age and health of the patient. The overall 5-year relative survival rate for female patients with breast cancer has improved from 75% to 90% from 1975 to 1977 and from 2003 to 2009, respectively, largely due to improvements in treatment (ie, chemotherapy, hormone therapy, and targeted drugs) and because of earlier diagnosis resulting from the widespread use of mammography and other screening tools.2
Estrogen Receptor-Positive Therapies
Women with breast cancer who test positive for hormone receptors are candidates for treatment with hormone therapy to reduce the likelihood of recurrence or as a core component of treatment for advanced disease. Currently available endocrine strategies for the treatment of estrogen receptor- (ER) positive breast cancer include targeting the ER with the antiestrogen drug tamoxifen. Another option is suppressing the amount of available ligand (estrogen) for the receptor either with gonadal suppression in premenopausal oophorectomy, or luteinizing hormonereleasing hormone agonists, or with the aromatase inhibitors (AIs) anastrozole, exemestane, and letrozole in postmenopausal women and by downregulating the receptor with fulvestrant. Given their proven efficacy and generally favorable adverse effect (AE) profile, these endocrine therapies are widely used in the treatment of both early-stage and recurrent and/or metastatic breast cancer.
Recent studies have offered new treatments for patients with hormone receptor-positive, human epidermal growth factor receptor 2 (HER2)-negative breast cancer. Innovative hormonal and targeted therapies for advanced disease as well as new data on adjuvant hormonal therapy for young high-risk patients are changing the available therapeutic options.
Advanced Metastatic Treatments
Treatment for metastatic hormone receptor-positive breast cancer has shifted from traditional cytotoxic chemotherapies to targeted therapeutic options. Most treatment guidelines, including the National Comprehensive Cancer Network guidelines, recommend targeted therapy with AIs or selective ER modulators rather than chemotherapy, except in the case of visceral crisis.3
Until recently, there had been relatively little guidance to inform which hormonal therapy was most appropriate. Aromatase inhibitors were generally reserved for postmenopausal women, whereas tamoxifen was preferred in premenopausal women.
Fulvestrant
The FDA initially approved fulvestrant, a hormone receptor downregulator, in 2002 at a 250-mg dose, following progression on an anti-estrogen therapy, such as tamoxifen in postmenopausal women with stage IV breast cancer. The FDA approval was based on similar response rates for the already approved agent anastrozole.4 However, pharmacokinetic findings from the phase 3 EFECT trial in 2008 prompted researchers to explore a 500-mg dose of fulvestrant.5
The recently published FIRST study is a phase 2, randomized, open-label study comparing fulvestrant 500 mg with anastrozole 1 mg as first-line hormonal therapy for postmenopausal women with hormone receptorpositive advanced breast cancer. Fulvestrant was given 500 mg once monthly with an extra dose given on day 14 of month 1. The trial enrolled 233 patients. The median time to progression was 23.4 months for fulvestrant and 13.1 months for anastrozole. These results translate into a 34% reduction in the risk of progression.6
These outcomes suggest that fulvestrant is as viable and perhaps even preferred first-line therapy for postmenopausal women with hormone receptor-positive, HER2-negative advanced breast cancer. The impressive results from this trial are likely, because the study used the 500-mg dose of fulvestrant, which is twice the dose used in the original trials. However, the 500-mg dose has previously been studied, and long-term outcome data suggest both safety and efficiency. The large randomized, double-blinded phase 3 CONFIRM trial, published in 2013, compared the 250-mg dose with the 500-mg dose and found that the higher dose was associated with a 19% reduction in the risk of death and a 4.1 month increase in median overall survival (OS) without any new safety concerns.5
Palbociclib
The FDA recently granted accelerated approval to palbociclib in combination with letrozole for the first-line therapy of advanced hormone receptor-positive, HER2-negative breast cancer in postmenopausal women. Palbociclib is an oral small-molecular inhibitor of cyclindependent kinases 4 and 6. Preclinical data suggested synergy with anti-estrogen therapies and inhibition of breast cancer cell growth.7
A phase 2, open-label randomized trial (PALOMA-1/TRIO-18) enrolled 165 patients. Progression-free survival (PFS) was 20.2 months for the palbociclib plus letrozole arm and 10.2 months for the letrozole alone arm. Significant toxicities were noted in the palbociclib arm, including 54% of people experiencing grade 3 to 4 neutropenia (vs 1% in the letrozole arm), leukopenia in 19% (vs 0%) and fatigue in 4% (vs 1%). A phase 3 trial is currently enrolling patients.7 While we await the results of the phase 3 trial and long-term follow-up data, palbociclib plus letrozole is a new, viable option for metastatic hormone receptor-positive advanced breast cancer.
Although many practitioners will continue to reasonably use any AI or selective ER modulator when treating metastatic breast cancer, both fulvestrant and palbociclib in combination with letrozole are new evidence-based, first-line options worth considering.
Early-Stage Treatment Options
There are many acceptable therapeutic options for treating early stage breast cancer. Tamoxifen has traditionally been used in the adjuvant setting for premenopausal women, whereas AIs are often used in postmenopausal women. There has also been a long-standing debate about the role of ovarian suppression in premenopausal women.
The recently published phase 3 TEXT and SOFT trials attempted to provide answers to these long-standing therapeutic dilemmas. The SOFT trial randomly assigned 3,066 premenopausal women to 5 years of tamoxifen, 5 years of tamoxifen plus ovarian suppression, or exemestane plus ovarian suppression. The TEXT trial randomly assigned 2,672 women to receive either exemestane plus ovarian suppression or tamoxifen plus ovarian suppression. The studies showed that subjecting all women receiving tamoxifen to ovarian suppression did not provide any significant benefit.8,9
However, the subgroup of women with high-risk disease who required adjuvant chemotherapy and remained premenopausal experienced improved outcomes from ovarian suppression. This high-risk subgroup when given tamoxifen plus ovarian suppression had a 4.5% absolute reduction in breast cancer recurrence at 5 years compared with the group that received tamoxifen alone. When this high-risk subgroup was given exemestane plus ovarian suppression, the women had a 7.7% absolute reduction in breast cancer recurrence at 5 years compared with the group that received tamoxifen alone.8
Ovarian suppression resulted in significant additional AEs, including depression and menopausal symptoms. The authors of the study also pointed out the additional risk of hypertension, musculoskeletal AEs, and decreased bone density. Furthermore, the OS data from these studies are premature, because the patients had fewer AEs than initially anticipated; this resulted in an only 5% mortality at publication.
The study design also raised several interesting questions. The primary endpoint was disease-free survival. The authors defined this as the time from randomization to the first appearance of invasive recurrence of breast cancer (local, regional, or distant), invasive contralateral breast cancer, second (non-breast) invasive cancer, or death without breast cancer recurrence or second invasive cancer. When studying adjuvant therapy for diseases, such as breast cancer, which carry long-term survival, studies often use PFS with various modified definitions as a surrogate marker for OS. Clinicians are then left to decide whether this surrogate marker is an accurate predictor of OS or other important clinical outcomes.
In the combined analysis of the TEXT and SOFT trials, only 60% of the first recurrences, second invasive cancers, or deaths involved recurrence of breast cancer
at a distant site.9 Because locally recurrent breast cancer is highly treatable and often curable, clinicians must ask whether the increased toxicities of ovarian suppression are worth the large number of women who experienced local recurrence given the still relatively small absolute reduction in recurrence risk.
Last, the study authors retrospectively reviewed data from the International Breast Cancer Study Group and U.S. Intergroup trials and concluded that women aged < 35 years were most likely to be at high-risk for AEs.10,11 A subgroup analysis of women aged < 35 years in the SOFT trial noted that breast cancer recurred within 5 years in one-third of women receiving tamoxifen alone, whereas only in one-sixth of women receiving exemestane plus ovarian suppression.8 This is the basis for the conclusion that premenopausal women, particularly those aged < 35 years, with high-risk disease who receive chemotherapy and remain premenopausal after chemotherapy, benefit from ovarian suppression in combination with tamoxifen, and even more impressively from ovarian suppression combined with exemestane.
The problem is that the study did not risk-stratify patients based on those aged < 35 years, and the conclusion is based on a subgroup analysis using a primary endpoint that may not accurately predict OS. Nonetheless, although not definitive, the data from the TEXT and SOFT trials raise interesting therapeutic questions that require further study and certainly provide tempting therapeutic options in patients who are clinically at high risk for recurrence.
HER2-Positive Breast Cancer
Up to 20% of invasive breast cancers are a result of HER2 gene amplification or overexpression of the HER2 protein, a tyrosine kinase transmembrane receptor, resulting in a more aggressive phenotype and a poor prognosis. Anti-HER2 drugs have changed the landscape of the disease previously known as aggressive breast cancer with a poor survival rate.
Treatment with the anti-HER2 humanized monoclonal antibody trastuzumab in addition to chemotherapy, compared with chemotherapy alone, significantly improves PFS and OS among patients with HER2-positive metastatic as well as early breast cancer. However, in most patients with HER2-positive metastatic breast cancer, the disease progresses, highlighting the need for new, targeted therapies for advanced disease.
New Standard of Care
The original studies of trastuzumab showed improved OS in late-stage (metastatic) breast cancer from 20.3 to 25.1 months, and in early-stage breast cancer, it reduced the risk of cancer returning after surgery by an absolute risk of 9.5% and the risk of death by an absolute risk of 3%.
New therapies directed at HER2 are being developed, among them pertuzumab, a humanized monoclonal antibody that binds HER2 at a different epitope of the HER2 extracellular domain (subdomain 2) than that at which trastuzumab binds. Pertuzumab prevents HER2 from dimerizing with other ligand-activated HER receptors, most notably HER3. Like trastuzumab, pertuzumab stimulates antibody-dependent, cell-mediated cytotoxicity. Because pertuzumab and trastuzumab bind to different HER2 epitopes and have complementary mechanisms of action, these 2 agents, when given together, provide a more comprehensive blockade of HER2 signaling and result in greater antitumor activity than does either agent alone in HER2-positive tumor models.12 In phase 2 studies, a pertuzumab–trastuzumab regimen has shown activity in patients with HER2-positive metastatic breast cancer and in patients with early breast cancer.13
In the phase 3 CLEOPATRA study, the combination of pertuzumab plus trastuzumab plus docetaxel, used as first-line treatment for HER2-positive metastatic breast cancer compared with placebo plus trastuzumab plus docetaxel, significantly prolonged PFS (18.5 months vs 12.4 months), with no increase in cardiac toxic effects.12 In a recent updated follow-up of the CLEOPATRA study, the addition of pertuzumab to trastuzumab and docetaxel showed a significantly better median OS (56.5 months vs 40.8 months; hazard ratio, 0.68; P < .001).14 From these results, this combination regimen is now considered a first-line therapy for patients with HER2-positive metastatic breast cancer.
However, the cost of cancer treatment has become a mounting concern during the past decade, as new therapies come down the pipeline with ever-increasing price tags. Trastuzumab costs about $4,500 a month, and the newer pertuzumab runs about 30% higher, at $6,000 a month. For a full course of treatment, the cost of the pertuzumab and trastuzumab combination could go as high as $195,000, depending on the duration of therapy and the choice of taxanes.
Conclusions
The landscape of therapeutic options in high-risk, young patients with early-stage breast cancer as well as patients with advanced or metastatic disease is changing rapidly.
Clinicians now have 2 new first-line options for the treatment of advanced hormone receptor-positive, HER2-negative breast cancer. A phase 3 trial demonstrated that fulvestrant monotherapy offers improved PFS and some improvement in OS compared with anastrazole in postmenopausal women. A phase 2 trial showed that palbociclib plus letrozole offers improved PFS in postmenopausal women. Based on the SOFT and TEXT trials, clinicians treating high-risk premenopausal women now have some data to inform the debate about whether ovarian suppression should be added to hormone therapy.
Based on the CLEOPATRA trial, clinicians can now consider combination pertuzumab and trastuzumab and docetaxel as first-line therapy for patients with HER2-positive metastatic breast cancer.
Author disclosures
The authors report no actual or potential conflicts of interest with regard to this article.
Disclaimer
The opinions expressed herein are those of the authors and do not necessarily reflect those of Federal Practitioner, Frontline Medical Communications Inc., the U.S. Government, or any of its agencies. This article may discuss unlabeled or investigational use of certain drugs. Please review complete prescribing information for specific drugs or drug combinations—including indications, contraindications, warnings, and adverse effects—before administering pharmacologic therapy to patients.
Click here to read the digital edition.
1. American Cancer Society. Cancer facts & figures, 2015. Atlanta, GA: American Cancer Society; 2015.
2. American Cancer Society. Cancer treatment & survivorship facts & figures, 2014-2015. Atlanta, GA: American Cancer Society; 2014.
3. National Comprehensive Cancer Network. NCCN clinical Practice guidelines in oncology: breast Cancer. Version 1. 2015. Fort Washington, PA: National Comprehensive Cancer Network; 2015:BINV-19.
4. Howell A, Robertson JF, Quaresma Albano J. Fulvestrant, formerly ICI 182,780, is as effective as anastrozole in postmenopausal women with advanced breast cancer progressing after prior endocrine treatment. J Clin Oncol. 2002;20(16):3396-3403.
5. Di Leo A, Jerusalem G, Petruzelka L, et al. Final overall survival: fulvestrant 500 mg vs 250 mg in the randomized CONFIRM trial. J Natl Cancer Inst. 2014;106(1):djt337.
6. Robertson JF, Lindemann JB, Llombart-Cussac A, et al. Fulvestrant 500 mg versus anastrozole 1 mg for the first-line treatment of advanced breast cancer: follow-up analysis from the randomized ‘FIRST’ study. Breast Cancer Res Treat. 2012;136(2):503-511.
7. Finn RS, Crown JP, Lang I, et al. The cyclin-dependent kinase 4/6 inhibitor palbociclib in combination with letrozole versus letrozole alone as first-line treatment of oestrogen receptor-positive, HER2-negative, advanced breast cancer (PALOMA-1/TRIO-18): a randomised phase 2 study. Lancet Oncol. 2015;16(1):25-35.
8. Francis PA, Regan MM, Fleming GF, et al; SOFT Investigators; International Breast Cancer Study Group. Adjuvant ovarian suppression in premenopausal breast cancer. N Engl J Med. 2015;372(5):436-446.
9. Pagani O. Regan MM, Walley BA, et al. TEXT and SOFT Investigators; International Breast Cancer Study Group. Adjuvant exemestane with ovarian suppression in premenopausal breast cancer. N Engl J Med. 2014;371(2):107-118.
10. Aebi S, Gelber S, Castiglione-Gertsch M, et al. Is chemotherapy alone adequate for young women with oestrogen-receptor-positive breast cancer? Lancet. 2000;355:1869-1874.
11. Goldhirsch A, Gelber RD, Yothers G, et al. Adjuvant therapy for very young women with breast cancer: need for tailored treatments. J Natl Cancer Inst Monogr. 2001;(30):44-51
12. Hudis CA. Trastuzumab—mechanism of action and use in clinical practice. N Engl J Med. 2007;357(1):39-51.
13. Baselga J, Cortés J, Kim SB, et al; CLEOPATRA Study Group. Pertuzumab plus trastuzumab plus docetaxel for metastatic breast cancer. N Engl J Med. 2012;366(2):109-119.
14. Swain SM, Baselga J, Kim SB, et al; CLEOPATRA Study Group. Pertuzumab, trastuzumab, and docetaxel in HER2-positive metastatic breast cancer. N Engl J Med. 2015;372(8):724-734.
1. American Cancer Society. Cancer facts & figures, 2015. Atlanta, GA: American Cancer Society; 2015.
2. American Cancer Society. Cancer treatment & survivorship facts & figures, 2014-2015. Atlanta, GA: American Cancer Society; 2014.
3. National Comprehensive Cancer Network. NCCN clinical Practice guidelines in oncology: breast Cancer. Version 1. 2015. Fort Washington, PA: National Comprehensive Cancer Network; 2015:BINV-19.
4. Howell A, Robertson JF, Quaresma Albano J. Fulvestrant, formerly ICI 182,780, is as effective as anastrozole in postmenopausal women with advanced breast cancer progressing after prior endocrine treatment. J Clin Oncol. 2002;20(16):3396-3403.
5. Di Leo A, Jerusalem G, Petruzelka L, et al. Final overall survival: fulvestrant 500 mg vs 250 mg in the randomized CONFIRM trial. J Natl Cancer Inst. 2014;106(1):djt337.
6. Robertson JF, Lindemann JB, Llombart-Cussac A, et al. Fulvestrant 500 mg versus anastrozole 1 mg for the first-line treatment of advanced breast cancer: follow-up analysis from the randomized ‘FIRST’ study. Breast Cancer Res Treat. 2012;136(2):503-511.
7. Finn RS, Crown JP, Lang I, et al. The cyclin-dependent kinase 4/6 inhibitor palbociclib in combination with letrozole versus letrozole alone as first-line treatment of oestrogen receptor-positive, HER2-negative, advanced breast cancer (PALOMA-1/TRIO-18): a randomised phase 2 study. Lancet Oncol. 2015;16(1):25-35.
8. Francis PA, Regan MM, Fleming GF, et al; SOFT Investigators; International Breast Cancer Study Group. Adjuvant ovarian suppression in premenopausal breast cancer. N Engl J Med. 2015;372(5):436-446.
9. Pagani O. Regan MM, Walley BA, et al. TEXT and SOFT Investigators; International Breast Cancer Study Group. Adjuvant exemestane with ovarian suppression in premenopausal breast cancer. N Engl J Med. 2014;371(2):107-118.
10. Aebi S, Gelber S, Castiglione-Gertsch M, et al. Is chemotherapy alone adequate for young women with oestrogen-receptor-positive breast cancer? Lancet. 2000;355:1869-1874.
11. Goldhirsch A, Gelber RD, Yothers G, et al. Adjuvant therapy for very young women with breast cancer: need for tailored treatments. J Natl Cancer Inst Monogr. 2001;(30):44-51
12. Hudis CA. Trastuzumab—mechanism of action and use in clinical practice. N Engl J Med. 2007;357(1):39-51.
13. Baselga J, Cortés J, Kim SB, et al; CLEOPATRA Study Group. Pertuzumab plus trastuzumab plus docetaxel for metastatic breast cancer. N Engl J Med. 2012;366(2):109-119.
14. Swain SM, Baselga J, Kim SB, et al; CLEOPATRA Study Group. Pertuzumab, trastuzumab, and docetaxel in HER2-positive metastatic breast cancer. N Engl J Med. 2015;372(8):724-734.
Safe to avoid sentinel node biopsy in some breast cancer patients
SEATTLE – Sentinel lymph node biopsy is widely used in patients with early-stage breast cancer for staging the axilla, but it can be safely omitted in some patients, according to new research presented at the annual Society of Surgical Oncology Cancer Symposium.
In women aged 70 years and older with hormone receptor (HR)–positive invasive breast cancer, the risk of nodal involvement is 14%-15%, which adds support to the premise that sentinel lymph node surgery could be avoided in many of the women deemed to be low risk.
The Choosing Wisely campaign was initiated to reduce excess cost and expenditures in health care. The Society of Surgical Oncology recently released five Choosing Wisely guidelines that included specific tests or procedures commonly ordered but not always necessary in surgical oncology, explained study author Jessemae Welsh, MD, of the Mayo Clinic, Rochester, Minn. One of the recommendations was to avoid routine sentinel node biopsy in clinically node-negative women over age 70 years with hormone receptor–positive invasive breast cancer.
“Their rationale is that hormone therapy is the standard of care in these women and sentinel node surgery has shown no impact on local regional recurrence or breast cancer mortality,” said Dr. Welsh. “Therefore it would be safe to treat this population without any axillary node staging.”
She noted that the average 70-year-old woman may live another 14-16 years. “So the question is, how should we be applying the Choosing Wisely guidelines?”
Dr. Welsh and her colleagues evaluated the factors that might be impacting nodal positivity in this population, and in particular, they looked at T stage and tumor grade.
They used two large databases to identify all women over the age of 70 years with HR+ cN0 invasive disease in the institutional breast surgery database (IBSD, 2008-2016) from the Mayo Clinic and the National Cancer Database (NCDB, 2004-2013).
The rates of patients who were node positive (pN+) were based on those who had undergone axillary surgery.
The researchers then stratified patients by clinical T stage and tumor grade to compare risk of pN+ across strata.
Of 705 selected patients in the IBSD, 191 or 14.3% were pN+ and a similar rate was observed in the NCDB; 15.2% (19,607/129,216). Tumor grade and clinical T stage were associated with pN+.
“The overall rates were about 14% for both databases, and when we stratified this by T stage, we could see increasing node positivity with increasing T stage,” said Dr. Welsh.
In similar fashion, the researchers observed comparable increases when they stratified it by grade. “Increasing grades were associated with increasing rates, especially for grade 2 and higher,” said Dr. Welsh.
When the two factors were combined, the researchers were able to define low-risk criteria as clinical T1a-b, grade 1-2 or clinical T1c, grade 1. The low-risk group accounted for 54.3% (IBSD) and 43.2% (NCDB) of patients, and pN+ rates within this group were 7.6% (IBSD) and 7.4% (NCDB).
Patients outside of this subcohort had pN+ rates of 22.4% (IBSD) and 23.0% (NCDB), which extrapolated to a relative risk of 2.95 (95% CI: 1.97-4.42) and 3.11 (95% CI: 2.99-3.23), respectively (each P less than .001).
“Women in the high-risk group had three times the risk of node positivity as the low-risk group,” she said. “Based on our data, we can say that for grade 1 T1a-c we can omit sentinel node surgery, and also for grade 2 T1 a-b.”
But for grade 3, T2 or higher, or any grade 2 Tc tumors, clinicians should continue to consider sentinel node surgery, taking into account individual patient factors.
The investigator had no disclosures.
Are there patients older than 70 years of age who have a low risk of nodal metastasis and/or even if they had nodal metastasis, could be adequately treated with anti-hormones?
If the answer is “yes,” then sentinel node sampling can be avoided in these patients. This study identified a group with a low risk for nodal metastasis. Even though it may be difficult to estimate tumor size preoperatively for lobular cancers, this is less of a problem for ductal cancers. Another approach is to use molecular profiling to determine which patients may “skip” sentinel node biopsy. Molecular profiling can identify patients who will have an excellent outcome with adjuvant anti-hormones even in the presence of nodal metastases.
Maureen Chung, MD, FACS, is medical director of the breast care program at Southcoast Health, North Dartmouth, Mass.
Are there patients older than 70 years of age who have a low risk of nodal metastasis and/or even if they had nodal metastasis, could be adequately treated with anti-hormones?
If the answer is “yes,” then sentinel node sampling can be avoided in these patients. This study identified a group with a low risk for nodal metastasis. Even though it may be difficult to estimate tumor size preoperatively for lobular cancers, this is less of a problem for ductal cancers. Another approach is to use molecular profiling to determine which patients may “skip” sentinel node biopsy. Molecular profiling can identify patients who will have an excellent outcome with adjuvant anti-hormones even in the presence of nodal metastases.
Maureen Chung, MD, FACS, is medical director of the breast care program at Southcoast Health, North Dartmouth, Mass.
Are there patients older than 70 years of age who have a low risk of nodal metastasis and/or even if they had nodal metastasis, could be adequately treated with anti-hormones?
If the answer is “yes,” then sentinel node sampling can be avoided in these patients. This study identified a group with a low risk for nodal metastasis. Even though it may be difficult to estimate tumor size preoperatively for lobular cancers, this is less of a problem for ductal cancers. Another approach is to use molecular profiling to determine which patients may “skip” sentinel node biopsy. Molecular profiling can identify patients who will have an excellent outcome with adjuvant anti-hormones even in the presence of nodal metastases.
Maureen Chung, MD, FACS, is medical director of the breast care program at Southcoast Health, North Dartmouth, Mass.
SEATTLE – Sentinel lymph node biopsy is widely used in patients with early-stage breast cancer for staging the axilla, but it can be safely omitted in some patients, according to new research presented at the annual Society of Surgical Oncology Cancer Symposium.
In women aged 70 years and older with hormone receptor (HR)–positive invasive breast cancer, the risk of nodal involvement is 14%-15%, which adds support to the premise that sentinel lymph node surgery could be avoided in many of the women deemed to be low risk.
The Choosing Wisely campaign was initiated to reduce excess cost and expenditures in health care. The Society of Surgical Oncology recently released five Choosing Wisely guidelines that included specific tests or procedures commonly ordered but not always necessary in surgical oncology, explained study author Jessemae Welsh, MD, of the Mayo Clinic, Rochester, Minn. One of the recommendations was to avoid routine sentinel node biopsy in clinically node-negative women over age 70 years with hormone receptor–positive invasive breast cancer.
“Their rationale is that hormone therapy is the standard of care in these women and sentinel node surgery has shown no impact on local regional recurrence or breast cancer mortality,” said Dr. Welsh. “Therefore it would be safe to treat this population without any axillary node staging.”
She noted that the average 70-year-old woman may live another 14-16 years. “So the question is, how should we be applying the Choosing Wisely guidelines?”
Dr. Welsh and her colleagues evaluated the factors that might be impacting nodal positivity in this population, and in particular, they looked at T stage and tumor grade.
They used two large databases to identify all women over the age of 70 years with HR+ cN0 invasive disease in the institutional breast surgery database (IBSD, 2008-2016) from the Mayo Clinic and the National Cancer Database (NCDB, 2004-2013).
The rates of patients who were node positive (pN+) were based on those who had undergone axillary surgery.
The researchers then stratified patients by clinical T stage and tumor grade to compare risk of pN+ across strata.
Of 705 selected patients in the IBSD, 191 or 14.3% were pN+ and a similar rate was observed in the NCDB; 15.2% (19,607/129,216). Tumor grade and clinical T stage were associated with pN+.
“The overall rates were about 14% for both databases, and when we stratified this by T stage, we could see increasing node positivity with increasing T stage,” said Dr. Welsh.
In similar fashion, the researchers observed comparable increases when they stratified it by grade. “Increasing grades were associated with increasing rates, especially for grade 2 and higher,” said Dr. Welsh.
When the two factors were combined, the researchers were able to define low-risk criteria as clinical T1a-b, grade 1-2 or clinical T1c, grade 1. The low-risk group accounted for 54.3% (IBSD) and 43.2% (NCDB) of patients, and pN+ rates within this group were 7.6% (IBSD) and 7.4% (NCDB).
Patients outside of this subcohort had pN+ rates of 22.4% (IBSD) and 23.0% (NCDB), which extrapolated to a relative risk of 2.95 (95% CI: 1.97-4.42) and 3.11 (95% CI: 2.99-3.23), respectively (each P less than .001).
“Women in the high-risk group had three times the risk of node positivity as the low-risk group,” she said. “Based on our data, we can say that for grade 1 T1a-c we can omit sentinel node surgery, and also for grade 2 T1 a-b.”
But for grade 3, T2 or higher, or any grade 2 Tc tumors, clinicians should continue to consider sentinel node surgery, taking into account individual patient factors.
The investigator had no disclosures.
SEATTLE – Sentinel lymph node biopsy is widely used in patients with early-stage breast cancer for staging the axilla, but it can be safely omitted in some patients, according to new research presented at the annual Society of Surgical Oncology Cancer Symposium.
In women aged 70 years and older with hormone receptor (HR)–positive invasive breast cancer, the risk of nodal involvement is 14%-15%, which adds support to the premise that sentinel lymph node surgery could be avoided in many of the women deemed to be low risk.
The Choosing Wisely campaign was initiated to reduce excess cost and expenditures in health care. The Society of Surgical Oncology recently released five Choosing Wisely guidelines that included specific tests or procedures commonly ordered but not always necessary in surgical oncology, explained study author Jessemae Welsh, MD, of the Mayo Clinic, Rochester, Minn. One of the recommendations was to avoid routine sentinel node biopsy in clinically node-negative women over age 70 years with hormone receptor–positive invasive breast cancer.
“Their rationale is that hormone therapy is the standard of care in these women and sentinel node surgery has shown no impact on local regional recurrence or breast cancer mortality,” said Dr. Welsh. “Therefore it would be safe to treat this population without any axillary node staging.”
She noted that the average 70-year-old woman may live another 14-16 years. “So the question is, how should we be applying the Choosing Wisely guidelines?”
Dr. Welsh and her colleagues evaluated the factors that might be impacting nodal positivity in this population, and in particular, they looked at T stage and tumor grade.
They used two large databases to identify all women over the age of 70 years with HR+ cN0 invasive disease in the institutional breast surgery database (IBSD, 2008-2016) from the Mayo Clinic and the National Cancer Database (NCDB, 2004-2013).
The rates of patients who were node positive (pN+) were based on those who had undergone axillary surgery.
The researchers then stratified patients by clinical T stage and tumor grade to compare risk of pN+ across strata.
Of 705 selected patients in the IBSD, 191 or 14.3% were pN+ and a similar rate was observed in the NCDB; 15.2% (19,607/129,216). Tumor grade and clinical T stage were associated with pN+.
“The overall rates were about 14% for both databases, and when we stratified this by T stage, we could see increasing node positivity with increasing T stage,” said Dr. Welsh.
In similar fashion, the researchers observed comparable increases when they stratified it by grade. “Increasing grades were associated with increasing rates, especially for grade 2 and higher,” said Dr. Welsh.
When the two factors were combined, the researchers were able to define low-risk criteria as clinical T1a-b, grade 1-2 or clinical T1c, grade 1. The low-risk group accounted for 54.3% (IBSD) and 43.2% (NCDB) of patients, and pN+ rates within this group were 7.6% (IBSD) and 7.4% (NCDB).
Patients outside of this subcohort had pN+ rates of 22.4% (IBSD) and 23.0% (NCDB), which extrapolated to a relative risk of 2.95 (95% CI: 1.97-4.42) and 3.11 (95% CI: 2.99-3.23), respectively (each P less than .001).
“Women in the high-risk group had three times the risk of node positivity as the low-risk group,” she said. “Based on our data, we can say that for grade 1 T1a-c we can omit sentinel node surgery, and also for grade 2 T1 a-b.”
But for grade 3, T2 or higher, or any grade 2 Tc tumors, clinicians should continue to consider sentinel node surgery, taking into account individual patient factors.
The investigator had no disclosures.
Key clinical point: Sentinel node biopsy can be safely avoided in certain populations of breast cancer patients.
Major finding: In women 70 years and older with hormone receptor (HR)–positive invasive breast cancer who are at low risk, sentinel node surgery can safely be avoided.
Data source: Two large databases of more than 150,000 women, from the Mayo Clinic and the National Cancer Database.
Disclosures: There was no funding source disclosed. The author had no disclosures.
Survivorship care models work, some better than others
ORLANDO – Accumulating experience is showing the benefits of various models of care for cancer survivors in terms of health care use and costs, while also suggesting that some provide higher-quality care than others, according to a pair of studies reported at a symposium on quality care sponsored by the American Society of Clinical Oncology.
Initiative for breast cancer survivors
“In 2011, Cancer Care Ontario did a quick environmental scan of our 14 regional cancer centers and found that the transition of breast cancer survivors from oncologists to primary care was very variable, and that centers often didn’t transition patients very frequently,” said Nicole Mittmann, PhD, first author on one of the studies, chief research officer for Cancer Care Ontario, and an investigator at Sunnybrook Research Institute, Toronto.
The advisory organization therefore implemented the Well Follow-Up Care Initiative to facilitate appropriate transition of breast cancer survivors. Each regional center was given a $100,000 incentive to roll out a model of the initiative.
Dr. Mittmann and her coinvestigators used provincial administrative databases to compare health care use and associated costs between 2,324 breast cancer survivors who were transitioned with the initiative and 2,324 propensity-matched control survivors who were not. The survivors were about 5 years out from their breast cancer diagnosis at baseline and had median follow-up of 2 years.
Study results reported at the symposium showed that the mean annual total cost of care per patient paid for by the provincial health ministry was $6,575 for the transitioned group and $10,832 for the nontransitioned group, a difference of $4,257 (39%). The main drivers were reduced costs of long-term care and cancer clinic visits.
Findings were similar for median annual costs, which amounted to $2,261 for the transitioned group and $2,903 for the control group, a difference of $638.
Compared with the nontransitioned group, the transitioned group had significantly fewer annual visits to medical oncologists (0.39 vs. 1.29) and radiation oncologists (0.16 vs. 0.36), while visits to general or family practitioners were statistically indistinguishable (7.35 and 7.91), Dr. Mittmann reported. There was also a trend toward fewer emergency department visits.
The transitioned group had fewer bone scans, CT and MRI scans, and radiographs annually, but differences were not significant.
Reassuringly, Dr. Mittmann said, survivors who were transitioned did not fare worse than their nontransitioned counterparts in overall survival; if anything, they tended to live longer. “We think that because the individual cancer centers enrolled patients that they thought were very well that this is a very well and highly selected and maybe a biased group,” Dr. Mittmann acknowledged. “But we certainly see that they are not doing worse than the control group.”
“About $1.4 million was distributed to the cancer centers” for the initiative, she noted. “That generated a savings for the health system of $1.5 million, if you are looking at median costs, to $9.9 million, if you are looking at mean costs.
“The transition of appropriate breast cancer survivors to the community appears to be safe and effective outside of a clinical trial, at least based on this particular retrospective analysis using databases,” she said. “The overall costs are not increased, and they may actually be decreased based on our data, and certainly these results will inform policy.”
The investigators plan several next steps, such as encouraging senior leadership at Cancer Care Ontario and the Ministry of Health to endorse the findings, according to Dr. Mittmann. In addition, “[we plan to] engage with both oncology and primary care leadership and think about how we can potentially roll out a program like this, and develop tools, whether those are letters or information packages, and education, to … appropriately transition individuals.”
Considerations in interpreting the study’s findings include the quality of the matching of survivors, according to invited discussant Monika K. Krzyzanowska, MD, a medical oncologist at Princess Margaret Cancer Centre, an associate professor at the University of Toronto, and a clinical lead of Quality Care and Access, Systemic Treatment Program, at Cancer Care Ontario. “The quality of that match depends on what’s in the model, so there could be potential for residual confounding, and administrative data may not have all of the elements that you would need to get a perfect match.”
Additional considerations include costs not covered by the payer, impact of the initiative on delivery of guideline-recommended care and patient and provider satisfaction, generalizability of the findings, and long-term outcomes.
“This is a proof of concept, certainly, that transition of low-risk cancer survivors to primary care is feasible and potentially economically attractive,” Dr. Krzyzanowska concluded. “It would be useful to have a formal evaluation of effectiveness that would inform a comprehensive value assessment. And we do have data from a randomized trial about the safety of this particular approach, but it would be nice to see that following implementation in real practices, those safety considerations played out the same way.”
Comparison of survivorship care models
Two-thirds of the large and growing population of cancer survivors are at least 5 years out from diagnosis, stimulating considerable discussion in the oncology community about how to best address their needs, according to Sarah Raskin, PhD, senior author on the second study and a research scientist at the Institute for Patient-Centered Initiatives and Health Equity at George Washington University Cancer Center, Washington.
“Yet, for a lack of cancer survivorship–specific guidelines from research or practice, cancer centers are increasingly developing survivorship care in a variety of ways, many of which are ad hoc or unproven as yet,” she said.
Dr. Raskin and her colleagues compared three emerging models of survivorship care: a specialized consultative model and a specialized longitudinal model – whereby patients have a single or multiple formalized survivorship visits, respectively, with care typically led by an oncology nurse-practitioner – and an oncology-embedded model – whereby survivorship is addressed as a part of ongoing oncology follow-up care, typically by the oncologist.
The investigators worked with survivors to develop the Patient-Prioritized Measure of High-Quality Survivorship Care, a 46-question scale assessing nine components of survivorship care that capture the health care priorities and needs that matter most to patients. Each component is rated on a scale from 0 (not at all met) to 1 (somewhat met) to 2 (definitely met).
Analyses were based on responses of 827 survivors of breast, colorectal, and prostate cancer who received care at 28 U.S. institutions using one of the above models and who were surveyed by telephone about the care received 1 week after their initial survivorship visit.
Results showed that survivors cared for under the three models differed significantly with respect to scores for seven of the nine components of quality of care, Dr. Raskin reported. The exceptions were practical life support, where the mean score was about 0.6-0.8 across the board, and having a medical home, where the mean score was about 1.8-1.9 across the board.
The specialized consult model of care had the highest scores for mental health and social support, information and resources, and supportive and prepared clinicians. The specialized longitudinal model of care had the highest scores for empowered and engaged patients, open patient-clinician communication, care coordination and transitions, and access to full spectrum of care. The oncology-embedded model had the lowest scores. Analysis of the tool’s 46 individual questions showed that patients cared for at institutions using the oncology-embedded model were significantly less likely than were counterparts cared for at institutions using the specialized models to report that the institution performed various activities such as offering a treatment summary, inquiring about the patient’s biggest worries or problems, and explaining the reasons why tests were needed (P less than .05 for each).
For some metrics, the overall proportion reporting that an activity was performed was low, regardless of the model being used. For example, only 48% of all patients reported being helped to set goals or make short-term plans to manage follow-up care and improve health, merely 24% reported being provided emotional and social support to deal with changes in relationships, and just 19% reported being referred to special providers for other medical problems.
“Overall, all three models are performing highly in terms of providing survivors with a medical home and communicating with patients. However, all three are performing quite low in terms of providing mental health and social support, as well as practical life support,” said Dr. Raskin.
“By model, we see that the embedded ongoing care model is significantly underperforming compared with both specialized models on seven of nine components, and we have some hypotheses from our early work with [Commission on Cancer]–accredited centers to explain this,” she added. “Embedded survivorship models have a lot of variability – many are high performers but others are low performers as compared with specialized programs. Embedded survivorship care models are typically led by the treating oncologist, who historically has focused on treating sick patients and less so on providing social supports for follow-up of well patients or ‘well-er’ patients. At the same time, specialized models focus predominantly on survivorship care and providing services and referrals for survivors, which may explain their high scores.
“We know that the higher quality of care measures presented here do not necessarily translate to better patient outcomes, and that’s actually going to be the next phase of our analysis,” she concluded.
The study sample may have had some selection bias, and it is unclear how well validated the tool was, according to Dr. Krzyzanowska, the discussant. Another issue was its assessment of quality of care at only a single time point.
Nonetheless, the findings show “that measuring quality of survivorship care from a patient perspective is feasible and valuable. We have already heard about [need for] survivorship plans in survivorship care, so certainly the work that was just presented is extremely important to help to fill some of these gaps,” she said.
“I’m not sure that we yet know what the optimal model of survivorship care is without the information of the other outcomes. Furthermore, there’s different survivor populations and different ways that health care is organized, so perhaps there isn’t really one optimal model, but the model has to fit with the context,” Dr. Krzyzanowska concluded. “That being said … the tool that they have created can be a great tool for existing survivorship care programs to assess and improve the quality of their care.”
Dr. Mittmann and Dr. Raskin had no disclosures to report.
ORLANDO – Accumulating experience is showing the benefits of various models of care for cancer survivors in terms of health care use and costs, while also suggesting that some provide higher-quality care than others, according to a pair of studies reported at a symposium on quality care sponsored by the American Society of Clinical Oncology.
Initiative for breast cancer survivors
“In 2011, Cancer Care Ontario did a quick environmental scan of our 14 regional cancer centers and found that the transition of breast cancer survivors from oncologists to primary care was very variable, and that centers often didn’t transition patients very frequently,” said Nicole Mittmann, PhD, first author on one of the studies, chief research officer for Cancer Care Ontario, and an investigator at Sunnybrook Research Institute, Toronto.
The advisory organization therefore implemented the Well Follow-Up Care Initiative to facilitate appropriate transition of breast cancer survivors. Each regional center was given a $100,000 incentive to roll out a model of the initiative.
Dr. Mittmann and her coinvestigators used provincial administrative databases to compare health care use and associated costs between 2,324 breast cancer survivors who were transitioned with the initiative and 2,324 propensity-matched control survivors who were not. The survivors were about 5 years out from their breast cancer diagnosis at baseline and had median follow-up of 2 years.
Study results reported at the symposium showed that the mean annual total cost of care per patient paid for by the provincial health ministry was $6,575 for the transitioned group and $10,832 for the nontransitioned group, a difference of $4,257 (39%). The main drivers were reduced costs of long-term care and cancer clinic visits.
Findings were similar for median annual costs, which amounted to $2,261 for the transitioned group and $2,903 for the control group, a difference of $638.
Compared with the nontransitioned group, the transitioned group had significantly fewer annual visits to medical oncologists (0.39 vs. 1.29) and radiation oncologists (0.16 vs. 0.36), while visits to general or family practitioners were statistically indistinguishable (7.35 and 7.91), Dr. Mittmann reported. There was also a trend toward fewer emergency department visits.
The transitioned group had fewer bone scans, CT and MRI scans, and radiographs annually, but differences were not significant.
Reassuringly, Dr. Mittmann said, survivors who were transitioned did not fare worse than their nontransitioned counterparts in overall survival; if anything, they tended to live longer. “We think that because the individual cancer centers enrolled patients that they thought were very well that this is a very well and highly selected and maybe a biased group,” Dr. Mittmann acknowledged. “But we certainly see that they are not doing worse than the control group.”
“About $1.4 million was distributed to the cancer centers” for the initiative, she noted. “That generated a savings for the health system of $1.5 million, if you are looking at median costs, to $9.9 million, if you are looking at mean costs.
“The transition of appropriate breast cancer survivors to the community appears to be safe and effective outside of a clinical trial, at least based on this particular retrospective analysis using databases,” she said. “The overall costs are not increased, and they may actually be decreased based on our data, and certainly these results will inform policy.”
The investigators plan several next steps, such as encouraging senior leadership at Cancer Care Ontario and the Ministry of Health to endorse the findings, according to Dr. Mittmann. In addition, “[we plan to] engage with both oncology and primary care leadership and think about how we can potentially roll out a program like this, and develop tools, whether those are letters or information packages, and education, to … appropriately transition individuals.”
Considerations in interpreting the study’s findings include the quality of the matching of survivors, according to invited discussant Monika K. Krzyzanowska, MD, a medical oncologist at Princess Margaret Cancer Centre, an associate professor at the University of Toronto, and a clinical lead of Quality Care and Access, Systemic Treatment Program, at Cancer Care Ontario. “The quality of that match depends on what’s in the model, so there could be potential for residual confounding, and administrative data may not have all of the elements that you would need to get a perfect match.”
Additional considerations include costs not covered by the payer, impact of the initiative on delivery of guideline-recommended care and patient and provider satisfaction, generalizability of the findings, and long-term outcomes.
“This is a proof of concept, certainly, that transition of low-risk cancer survivors to primary care is feasible and potentially economically attractive,” Dr. Krzyzanowska concluded. “It would be useful to have a formal evaluation of effectiveness that would inform a comprehensive value assessment. And we do have data from a randomized trial about the safety of this particular approach, but it would be nice to see that following implementation in real practices, those safety considerations played out the same way.”
Comparison of survivorship care models
Two-thirds of the large and growing population of cancer survivors are at least 5 years out from diagnosis, stimulating considerable discussion in the oncology community about how to best address their needs, according to Sarah Raskin, PhD, senior author on the second study and a research scientist at the Institute for Patient-Centered Initiatives and Health Equity at George Washington University Cancer Center, Washington.
“Yet, for a lack of cancer survivorship–specific guidelines from research or practice, cancer centers are increasingly developing survivorship care in a variety of ways, many of which are ad hoc or unproven as yet,” she said.
Dr. Raskin and her colleagues compared three emerging models of survivorship care: a specialized consultative model and a specialized longitudinal model – whereby patients have a single or multiple formalized survivorship visits, respectively, with care typically led by an oncology nurse-practitioner – and an oncology-embedded model – whereby survivorship is addressed as a part of ongoing oncology follow-up care, typically by the oncologist.
The investigators worked with survivors to develop the Patient-Prioritized Measure of High-Quality Survivorship Care, a 46-question scale assessing nine components of survivorship care that capture the health care priorities and needs that matter most to patients. Each component is rated on a scale from 0 (not at all met) to 1 (somewhat met) to 2 (definitely met).
Analyses were based on responses of 827 survivors of breast, colorectal, and prostate cancer who received care at 28 U.S. institutions using one of the above models and who were surveyed by telephone about the care received 1 week after their initial survivorship visit.
Results showed that survivors cared for under the three models differed significantly with respect to scores for seven of the nine components of quality of care, Dr. Raskin reported. The exceptions were practical life support, where the mean score was about 0.6-0.8 across the board, and having a medical home, where the mean score was about 1.8-1.9 across the board.
The specialized consult model of care had the highest scores for mental health and social support, information and resources, and supportive and prepared clinicians. The specialized longitudinal model of care had the highest scores for empowered and engaged patients, open patient-clinician communication, care coordination and transitions, and access to full spectrum of care. The oncology-embedded model had the lowest scores. Analysis of the tool’s 46 individual questions showed that patients cared for at institutions using the oncology-embedded model were significantly less likely than were counterparts cared for at institutions using the specialized models to report that the institution performed various activities such as offering a treatment summary, inquiring about the patient’s biggest worries or problems, and explaining the reasons why tests were needed (P less than .05 for each).
For some metrics, the overall proportion reporting that an activity was performed was low, regardless of the model being used. For example, only 48% of all patients reported being helped to set goals or make short-term plans to manage follow-up care and improve health, merely 24% reported being provided emotional and social support to deal with changes in relationships, and just 19% reported being referred to special providers for other medical problems.
“Overall, all three models are performing highly in terms of providing survivors with a medical home and communicating with patients. However, all three are performing quite low in terms of providing mental health and social support, as well as practical life support,” said Dr. Raskin.
“By model, we see that the embedded ongoing care model is significantly underperforming compared with both specialized models on seven of nine components, and we have some hypotheses from our early work with [Commission on Cancer]–accredited centers to explain this,” she added. “Embedded survivorship models have a lot of variability – many are high performers but others are low performers as compared with specialized programs. Embedded survivorship care models are typically led by the treating oncologist, who historically has focused on treating sick patients and less so on providing social supports for follow-up of well patients or ‘well-er’ patients. At the same time, specialized models focus predominantly on survivorship care and providing services and referrals for survivors, which may explain their high scores.
“We know that the higher quality of care measures presented here do not necessarily translate to better patient outcomes, and that’s actually going to be the next phase of our analysis,” she concluded.
The study sample may have had some selection bias, and it is unclear how well validated the tool was, according to Dr. Krzyzanowska, the discussant. Another issue was its assessment of quality of care at only a single time point.
Nonetheless, the findings show “that measuring quality of survivorship care from a patient perspective is feasible and valuable. We have already heard about [need for] survivorship plans in survivorship care, so certainly the work that was just presented is extremely important to help to fill some of these gaps,” she said.
“I’m not sure that we yet know what the optimal model of survivorship care is without the information of the other outcomes. Furthermore, there’s different survivor populations and different ways that health care is organized, so perhaps there isn’t really one optimal model, but the model has to fit with the context,” Dr. Krzyzanowska concluded. “That being said … the tool that they have created can be a great tool for existing survivorship care programs to assess and improve the quality of their care.”
Dr. Mittmann and Dr. Raskin had no disclosures to report.
ORLANDO – Accumulating experience is showing the benefits of various models of care for cancer survivors in terms of health care use and costs, while also suggesting that some provide higher-quality care than others, according to a pair of studies reported at a symposium on quality care sponsored by the American Society of Clinical Oncology.
Initiative for breast cancer survivors
“In 2011, Cancer Care Ontario did a quick environmental scan of our 14 regional cancer centers and found that the transition of breast cancer survivors from oncologists to primary care was very variable, and that centers often didn’t transition patients very frequently,” said Nicole Mittmann, PhD, first author on one of the studies, chief research officer for Cancer Care Ontario, and an investigator at Sunnybrook Research Institute, Toronto.
The advisory organization therefore implemented the Well Follow-Up Care Initiative to facilitate appropriate transition of breast cancer survivors. Each regional center was given a $100,000 incentive to roll out a model of the initiative.
Dr. Mittmann and her coinvestigators used provincial administrative databases to compare health care use and associated costs between 2,324 breast cancer survivors who were transitioned with the initiative and 2,324 propensity-matched control survivors who were not. The survivors were about 5 years out from their breast cancer diagnosis at baseline and had median follow-up of 2 years.
Study results reported at the symposium showed that the mean annual total cost of care per patient paid for by the provincial health ministry was $6,575 for the transitioned group and $10,832 for the nontransitioned group, a difference of $4,257 (39%). The main drivers were reduced costs of long-term care and cancer clinic visits.
Findings were similar for median annual costs, which amounted to $2,261 for the transitioned group and $2,903 for the control group, a difference of $638.
Compared with the nontransitioned group, the transitioned group had significantly fewer annual visits to medical oncologists (0.39 vs. 1.29) and radiation oncologists (0.16 vs. 0.36), while visits to general or family practitioners were statistically indistinguishable (7.35 and 7.91), Dr. Mittmann reported. There was also a trend toward fewer emergency department visits.
The transitioned group had fewer bone scans, CT and MRI scans, and radiographs annually, but differences were not significant.
Reassuringly, Dr. Mittmann said, survivors who were transitioned did not fare worse than their nontransitioned counterparts in overall survival; if anything, they tended to live longer. “We think that because the individual cancer centers enrolled patients that they thought were very well that this is a very well and highly selected and maybe a biased group,” Dr. Mittmann acknowledged. “But we certainly see that they are not doing worse than the control group.”
“About $1.4 million was distributed to the cancer centers” for the initiative, she noted. “That generated a savings for the health system of $1.5 million, if you are looking at median costs, to $9.9 million, if you are looking at mean costs.
“The transition of appropriate breast cancer survivors to the community appears to be safe and effective outside of a clinical trial, at least based on this particular retrospective analysis using databases,” she said. “The overall costs are not increased, and they may actually be decreased based on our data, and certainly these results will inform policy.”
The investigators plan several next steps, such as encouraging senior leadership at Cancer Care Ontario and the Ministry of Health to endorse the findings, according to Dr. Mittmann. In addition, “[we plan to] engage with both oncology and primary care leadership and think about how we can potentially roll out a program like this, and develop tools, whether those are letters or information packages, and education, to … appropriately transition individuals.”
Considerations in interpreting the study’s findings include the quality of the matching of survivors, according to invited discussant Monika K. Krzyzanowska, MD, a medical oncologist at Princess Margaret Cancer Centre, an associate professor at the University of Toronto, and a clinical lead of Quality Care and Access, Systemic Treatment Program, at Cancer Care Ontario. “The quality of that match depends on what’s in the model, so there could be potential for residual confounding, and administrative data may not have all of the elements that you would need to get a perfect match.”
Additional considerations include costs not covered by the payer, impact of the initiative on delivery of guideline-recommended care and patient and provider satisfaction, generalizability of the findings, and long-term outcomes.
“This is a proof of concept, certainly, that transition of low-risk cancer survivors to primary care is feasible and potentially economically attractive,” Dr. Krzyzanowska concluded. “It would be useful to have a formal evaluation of effectiveness that would inform a comprehensive value assessment. And we do have data from a randomized trial about the safety of this particular approach, but it would be nice to see that following implementation in real practices, those safety considerations played out the same way.”
Comparison of survivorship care models
Two-thirds of the large and growing population of cancer survivors are at least 5 years out from diagnosis, stimulating considerable discussion in the oncology community about how to best address their needs, according to Sarah Raskin, PhD, senior author on the second study and a research scientist at the Institute for Patient-Centered Initiatives and Health Equity at George Washington University Cancer Center, Washington.
“Yet, for a lack of cancer survivorship–specific guidelines from research or practice, cancer centers are increasingly developing survivorship care in a variety of ways, many of which are ad hoc or unproven as yet,” she said.
Dr. Raskin and her colleagues compared three emerging models of survivorship care: a specialized consultative model and a specialized longitudinal model – whereby patients have a single or multiple formalized survivorship visits, respectively, with care typically led by an oncology nurse-practitioner – and an oncology-embedded model – whereby survivorship is addressed as a part of ongoing oncology follow-up care, typically by the oncologist.
The investigators worked with survivors to develop the Patient-Prioritized Measure of High-Quality Survivorship Care, a 46-question scale assessing nine components of survivorship care that capture the health care priorities and needs that matter most to patients. Each component is rated on a scale from 0 (not at all met) to 1 (somewhat met) to 2 (definitely met).
Analyses were based on responses of 827 survivors of breast, colorectal, and prostate cancer who received care at 28 U.S. institutions using one of the above models and who were surveyed by telephone about the care received 1 week after their initial survivorship visit.
Results showed that survivors cared for under the three models differed significantly with respect to scores for seven of the nine components of quality of care, Dr. Raskin reported. The exceptions were practical life support, where the mean score was about 0.6-0.8 across the board, and having a medical home, where the mean score was about 1.8-1.9 across the board.
The specialized consult model of care had the highest scores for mental health and social support, information and resources, and supportive and prepared clinicians. The specialized longitudinal model of care had the highest scores for empowered and engaged patients, open patient-clinician communication, care coordination and transitions, and access to full spectrum of care. The oncology-embedded model had the lowest scores. Analysis of the tool’s 46 individual questions showed that patients cared for at institutions using the oncology-embedded model were significantly less likely than were counterparts cared for at institutions using the specialized models to report that the institution performed various activities such as offering a treatment summary, inquiring about the patient’s biggest worries or problems, and explaining the reasons why tests were needed (P less than .05 for each).
For some metrics, the overall proportion reporting that an activity was performed was low, regardless of the model being used. For example, only 48% of all patients reported being helped to set goals or make short-term plans to manage follow-up care and improve health, merely 24% reported being provided emotional and social support to deal with changes in relationships, and just 19% reported being referred to special providers for other medical problems.
“Overall, all three models are performing highly in terms of providing survivors with a medical home and communicating with patients. However, all three are performing quite low in terms of providing mental health and social support, as well as practical life support,” said Dr. Raskin.
“By model, we see that the embedded ongoing care model is significantly underperforming compared with both specialized models on seven of nine components, and we have some hypotheses from our early work with [Commission on Cancer]–accredited centers to explain this,” she added. “Embedded survivorship models have a lot of variability – many are high performers but others are low performers as compared with specialized programs. Embedded survivorship care models are typically led by the treating oncologist, who historically has focused on treating sick patients and less so on providing social supports for follow-up of well patients or ‘well-er’ patients. At the same time, specialized models focus predominantly on survivorship care and providing services and referrals for survivors, which may explain their high scores.
“We know that the higher quality of care measures presented here do not necessarily translate to better patient outcomes, and that’s actually going to be the next phase of our analysis,” she concluded.
The study sample may have had some selection bias, and it is unclear how well validated the tool was, according to Dr. Krzyzanowska, the discussant. Another issue was its assessment of quality of care at only a single time point.
Nonetheless, the findings show “that measuring quality of survivorship care from a patient perspective is feasible and valuable. We have already heard about [need for] survivorship plans in survivorship care, so certainly the work that was just presented is extremely important to help to fill some of these gaps,” she said.
“I’m not sure that we yet know what the optimal model of survivorship care is without the information of the other outcomes. Furthermore, there’s different survivor populations and different ways that health care is organized, so perhaps there isn’t really one optimal model, but the model has to fit with the context,” Dr. Krzyzanowska concluded. “That being said … the tool that they have created can be a great tool for existing survivorship care programs to assess and improve the quality of their care.”
Dr. Mittmann and Dr. Raskin had no disclosures to report.
AT THE QUALITY CARE SYMPOSIUM
Key clinical point:
Major finding: Mean annual health care costs were $4,257 (39%) lower for breast cancer survivors actively transitioned to primary care versus control peers. Specialized consult and specialized longitudinal models outperformed an oncology-embedded model on seven quality metrics.
Data source: A cohort study of 2,324 breast cancer survivors transitioned to primary care and 2,324 not transitioned. A cohort study of 827 survivors of breast, colorectal, and prostate cancer receiving care under three differing models.
Disclosures: Dr. Mittmann and Dr. Raskin had no disclosures to report.
Unavoidable, random DNA replication errors are the most common cancer drivers
Up to two-thirds of the mutations that drive human cancers may be due to DNA replication errors in normally dividing stem cells, not by inherited or environmentally induced mutations, according to a mathematical modeling study.
The proportion of replication error-driven mutations varied widely among 17 cancers analyzed, but the overall attributable risk of these errors was remarkably consistent among 69 countries included in the study, said Cristian Tomasetti, PhD, a coauthor of the paper and a biostatistician at Johns Hopkins University, Baltimore.
The findings should be a game-changer in the cancer field, Dr. Tomasetti said during a press briefing sponsored by the American Association for the Advancement of Science. Research dogma has long held that most cancers are related to lifestyle and environmental exposure, with a few primarily due to genetic factors.
“We have now determined that there is a third factor, and that it causes most of the mutations that drive cancer,” Dr. Tomasetti said. “We cannot ignore it and pretend it doesn’t exist. This is a complete paradigm shift in how we think of cancer and what causes it.”
The finding that 66% of cancer-driving mutations are based on unavoidable replication errors doesn’t challenge well-established epidemiology, said Dr. Tomasetti and his coauthor, Bert Vogelstein, MD. Rather, it fits perfectly with several key understandings of cancer: that about 40% of cases are preventable, that rapidly dividing tissues are more prone to develop cancers, and that cancer incidence rises exponentially as humans age.
“If we have as our starting point the assumption that 42% of cancers are preventable, we are completely consistent with that,” in finding that about 60% of cancers are unavoidable, Dr. Tomasetti said. “Those two numbers go perfectly together.”
The study also found that replication-error mutations (R) were most likely to drive cancers in tissues with rapid turnover, such as colorectal tissue. This makes intuitive sense, given that basal mutation rates hover at about three errors per cell replication cycle regardless of tissue type.
“The basal mutation rate in all cells is pretty even,” said Dr. Vogelstein, the Clayton Professor of Oncology and Pathology at John Hopkins University, Baltimore. “The difference is the number of stem cells. The more cells, the more divisions, and the more mistakes.”
R-mutations also contribute to age-related cancer incidence. As a person ages, more cell divisions accumulate, thus increasing the risk of a cancer-driving R-error. But these mutations also occur in children, who have rapid cell division in all their tissues. In fact, the colleagues suspect that R-errors are the main drivers of almost all pediatric cancers.
The new study bolsters the duo’s controversial 2015 work.
The theory sparked controversy among scholars and researchers. They challenged it on a number of technical fronts, from stem cell counts and division rates to charges that it didn’t adequately assess the interaction between R-mutations and environmental risks.
Some commentators, perceiving nihilism in the paper, expressed concern that clinicians and patients would get the idea that cancer prevention strategies were useless, since most cancers were simply a case of “bad luck.”
A pervading theme of these counter arguments was one familiar to any researcher: Correlation does not equal causation. The new study was an attempt to expand upon and strengthen the original findings, Dr. Tomasetti said.
“There are well-known environmental risk variations across the world, and there was a question of how our findings might change if we did this analysis in a different country. This paper is also the very first time that someone has ever looked at the proportions of mutations in each cancer type and assigned them to these factors.”
The new study employed a similar mathematical model, but comprised data from 423 cancer registries in 69 countries. The researchers examined the relationship between the lifetime risk of 17 cancers (including breast and prostate, which were not included in the 2015 study) and lifetime stem cell divisions for each tissue. The median correlation coefficient was 0.80; 89% of the countries examined had a correlation of greater than 0.70. This was “remarkably similar” to the correlation determined in the 2015 U.S.-only study.
The team’s next step was to determine what fraction of cancer-driving mutations arose from R-errors, from environmental factors (E), and from hereditary factors (H). They examined these proportions in 32 different cancers in which environmental, lifestyle, and genetic factors have been thoroughly studied. Overall, 29% of the driver mutations were due to environment, 5% to heredity, and 66% to R-errors.
The proportions of these drivers did vary widely between the cancer types, the team noted. For example, lung and esophageal cancers and melanoma were primarily driven by environmental factors (more than 60% each). However, they wrote, “even in lung adenocarcinomas, R contributes a third of the total mutations, with tobacco smoke [including secondhand smoke], diet, radiation, and occupational exposures contributing the remainder. In cancers that are less strongly associated with environmental factors, such as those of the pancreas, brain, bone, or prostate, the majority of the mutations are attributable to R.”
During the press briefing, Dr. Tomasetti and Dr. Vogelstein stressed that most of the inevitable R-errors don’t precipitate cancer – and that even if they do increase risk, that risk may not ever trip the disease process.
“Most of the time these replicative mutations do no harm,” Dr Vogelstein said. “They occur in junk DNA genes, or in areas that are unimportant with respect to cancer. That’s the good luck. Occasionally, they occur in a cancer driver gene, and that is bad luck.”
But even a dose of bad luck isn’t enough to cause cancer. Most cancers require multiple hits to develop – which makes primary prevention strategies more important than ever, Dr. Tomasetti said.
“In the case of lung cancer, for instance, three or more mutations are needed. We showed that these mutations are caused by a combination of environment and R-errors. In theory, then, all of these cancers are preventable because if we can prevent even one of the environmentally caused mutations, then that patient won’t develop cancer.”
However, he said, some cancers do appear to be entirely driven by E-errors and, thus, appear entirely unavoidable. This is an extremely difficult area for clinicians and patients to navigate, said Dr. Vogelstein, a former pediatrician.
“We hope that understanding this will offer some comfort to the literally millions of patients who develop cancer despite having lead a near-perfect life,” in terms of managing risk factors. “Cancer develops in people who haven’t smoked, who avoided the sun and wore sunscreen, who eat perfectly healthy diets and exercise regularly. This is a particularly important concept for parents of children who have cancer, who think ‘I either transmitted a bad gene or unknowingly exposed my child to an environmental agent that caused their cancer.’ They need to understand that these cancers would have occurred no matter what they did.”
Dr. Tomasetti had no disclosures. Dr. Vogelstein is on the scientific advisory boards of Morphotek, Exelixis GP, and Sysmex Inostics, and is a founder of PapGene and Personal Genome Diagnostics.
[email protected]
On Twitter @Alz_gal
Up to two-thirds of the mutations that drive human cancers may be due to DNA replication errors in normally dividing stem cells, not by inherited or environmentally induced mutations, according to a mathematical modeling study.
The proportion of replication error-driven mutations varied widely among 17 cancers analyzed, but the overall attributable risk of these errors was remarkably consistent among 69 countries included in the study, said Cristian Tomasetti, PhD, a coauthor of the paper and a biostatistician at Johns Hopkins University, Baltimore.
The findings should be a game-changer in the cancer field, Dr. Tomasetti said during a press briefing sponsored by the American Association for the Advancement of Science. Research dogma has long held that most cancers are related to lifestyle and environmental exposure, with a few primarily due to genetic factors.
“We have now determined that there is a third factor, and that it causes most of the mutations that drive cancer,” Dr. Tomasetti said. “We cannot ignore it and pretend it doesn’t exist. This is a complete paradigm shift in how we think of cancer and what causes it.”
The finding that 66% of cancer-driving mutations are based on unavoidable replication errors doesn’t challenge well-established epidemiology, said Dr. Tomasetti and his coauthor, Bert Vogelstein, MD. Rather, it fits perfectly with several key understandings of cancer: that about 40% of cases are preventable, that rapidly dividing tissues are more prone to develop cancers, and that cancer incidence rises exponentially as humans age.
“If we have as our starting point the assumption that 42% of cancers are preventable, we are completely consistent with that,” in finding that about 60% of cancers are unavoidable, Dr. Tomasetti said. “Those two numbers go perfectly together.”
The study also found that replication-error mutations (R) were most likely to drive cancers in tissues with rapid turnover, such as colorectal tissue. This makes intuitive sense, given that basal mutation rates hover at about three errors per cell replication cycle regardless of tissue type.
“The basal mutation rate in all cells is pretty even,” said Dr. Vogelstein, the Clayton Professor of Oncology and Pathology at John Hopkins University, Baltimore. “The difference is the number of stem cells. The more cells, the more divisions, and the more mistakes.”
R-mutations also contribute to age-related cancer incidence. As a person ages, more cell divisions accumulate, thus increasing the risk of a cancer-driving R-error. But these mutations also occur in children, who have rapid cell division in all their tissues. In fact, the colleagues suspect that R-errors are the main drivers of almost all pediatric cancers.
The new study bolsters the duo’s controversial 2015 work.
The theory sparked controversy among scholars and researchers. They challenged it on a number of technical fronts, from stem cell counts and division rates to charges that it didn’t adequately assess the interaction between R-mutations and environmental risks.
Some commentators, perceiving nihilism in the paper, expressed concern that clinicians and patients would get the idea that cancer prevention strategies were useless, since most cancers were simply a case of “bad luck.”
A pervading theme of these counter arguments was one familiar to any researcher: Correlation does not equal causation. The new study was an attempt to expand upon and strengthen the original findings, Dr. Tomasetti said.
“There are well-known environmental risk variations across the world, and there was a question of how our findings might change if we did this analysis in a different country. This paper is also the very first time that someone has ever looked at the proportions of mutations in each cancer type and assigned them to these factors.”
The new study employed a similar mathematical model, but comprised data from 423 cancer registries in 69 countries. The researchers examined the relationship between the lifetime risk of 17 cancers (including breast and prostate, which were not included in the 2015 study) and lifetime stem cell divisions for each tissue. The median correlation coefficient was 0.80; 89% of the countries examined had a correlation of greater than 0.70. This was “remarkably similar” to the correlation determined in the 2015 U.S.-only study.
The team’s next step was to determine what fraction of cancer-driving mutations arose from R-errors, from environmental factors (E), and from hereditary factors (H). They examined these proportions in 32 different cancers in which environmental, lifestyle, and genetic factors have been thoroughly studied. Overall, 29% of the driver mutations were due to environment, 5% to heredity, and 66% to R-errors.
The proportions of these drivers did vary widely between the cancer types, the team noted. For example, lung and esophageal cancers and melanoma were primarily driven by environmental factors (more than 60% each). However, they wrote, “even in lung adenocarcinomas, R contributes a third of the total mutations, with tobacco smoke [including secondhand smoke], diet, radiation, and occupational exposures contributing the remainder. In cancers that are less strongly associated with environmental factors, such as those of the pancreas, brain, bone, or prostate, the majority of the mutations are attributable to R.”
During the press briefing, Dr. Tomasetti and Dr. Vogelstein stressed that most of the inevitable R-errors don’t precipitate cancer – and that even if they do increase risk, that risk may not ever trip the disease process.
“Most of the time these replicative mutations do no harm,” Dr Vogelstein said. “They occur in junk DNA genes, or in areas that are unimportant with respect to cancer. That’s the good luck. Occasionally, they occur in a cancer driver gene, and that is bad luck.”
But even a dose of bad luck isn’t enough to cause cancer. Most cancers require multiple hits to develop – which makes primary prevention strategies more important than ever, Dr. Tomasetti said.
“In the case of lung cancer, for instance, three or more mutations are needed. We showed that these mutations are caused by a combination of environment and R-errors. In theory, then, all of these cancers are preventable because if we can prevent even one of the environmentally caused mutations, then that patient won’t develop cancer.”
However, he said, some cancers do appear to be entirely driven by E-errors and, thus, appear entirely unavoidable. This is an extremely difficult area for clinicians and patients to navigate, said Dr. Vogelstein, a former pediatrician.
“We hope that understanding this will offer some comfort to the literally millions of patients who develop cancer despite having lead a near-perfect life,” in terms of managing risk factors. “Cancer develops in people who haven’t smoked, who avoided the sun and wore sunscreen, who eat perfectly healthy diets and exercise regularly. This is a particularly important concept for parents of children who have cancer, who think ‘I either transmitted a bad gene or unknowingly exposed my child to an environmental agent that caused their cancer.’ They need to understand that these cancers would have occurred no matter what they did.”
Dr. Tomasetti had no disclosures. Dr. Vogelstein is on the scientific advisory boards of Morphotek, Exelixis GP, and Sysmex Inostics, and is a founder of PapGene and Personal Genome Diagnostics.
[email protected]
On Twitter @Alz_gal
Up to two-thirds of the mutations that drive human cancers may be due to DNA replication errors in normally dividing stem cells, not by inherited or environmentally induced mutations, according to a mathematical modeling study.
The proportion of replication error-driven mutations varied widely among 17 cancers analyzed, but the overall attributable risk of these errors was remarkably consistent among 69 countries included in the study, said Cristian Tomasetti, PhD, a coauthor of the paper and a biostatistician at Johns Hopkins University, Baltimore.
The findings should be a game-changer in the cancer field, Dr. Tomasetti said during a press briefing sponsored by the American Association for the Advancement of Science. Research dogma has long held that most cancers are related to lifestyle and environmental exposure, with a few primarily due to genetic factors.
“We have now determined that there is a third factor, and that it causes most of the mutations that drive cancer,” Dr. Tomasetti said. “We cannot ignore it and pretend it doesn’t exist. This is a complete paradigm shift in how we think of cancer and what causes it.”
The finding that 66% of cancer-driving mutations are based on unavoidable replication errors doesn’t challenge well-established epidemiology, said Dr. Tomasetti and his coauthor, Bert Vogelstein, MD. Rather, it fits perfectly with several key understandings of cancer: that about 40% of cases are preventable, that rapidly dividing tissues are more prone to develop cancers, and that cancer incidence rises exponentially as humans age.
“If we have as our starting point the assumption that 42% of cancers are preventable, we are completely consistent with that,” in finding that about 60% of cancers are unavoidable, Dr. Tomasetti said. “Those two numbers go perfectly together.”
The study also found that replication-error mutations (R) were most likely to drive cancers in tissues with rapid turnover, such as colorectal tissue. This makes intuitive sense, given that basal mutation rates hover at about three errors per cell replication cycle regardless of tissue type.
“The basal mutation rate in all cells is pretty even,” said Dr. Vogelstein, the Clayton Professor of Oncology and Pathology at John Hopkins University, Baltimore. “The difference is the number of stem cells. The more cells, the more divisions, and the more mistakes.”
R-mutations also contribute to age-related cancer incidence. As a person ages, more cell divisions accumulate, thus increasing the risk of a cancer-driving R-error. But these mutations also occur in children, who have rapid cell division in all their tissues. In fact, the colleagues suspect that R-errors are the main drivers of almost all pediatric cancers.
The new study bolsters the duo’s controversial 2015 work.
The theory sparked controversy among scholars and researchers. They challenged it on a number of technical fronts, from stem cell counts and division rates to charges that it didn’t adequately assess the interaction between R-mutations and environmental risks.
Some commentators, perceiving nihilism in the paper, expressed concern that clinicians and patients would get the idea that cancer prevention strategies were useless, since most cancers were simply a case of “bad luck.”
A pervading theme of these counter arguments was one familiar to any researcher: Correlation does not equal causation. The new study was an attempt to expand upon and strengthen the original findings, Dr. Tomasetti said.
“There are well-known environmental risk variations across the world, and there was a question of how our findings might change if we did this analysis in a different country. This paper is also the very first time that someone has ever looked at the proportions of mutations in each cancer type and assigned them to these factors.”
The new study employed a similar mathematical model, but comprised data from 423 cancer registries in 69 countries. The researchers examined the relationship between the lifetime risk of 17 cancers (including breast and prostate, which were not included in the 2015 study) and lifetime stem cell divisions for each tissue. The median correlation coefficient was 0.80; 89% of the countries examined had a correlation of greater than 0.70. This was “remarkably similar” to the correlation determined in the 2015 U.S.-only study.
The team’s next step was to determine what fraction of cancer-driving mutations arose from R-errors, from environmental factors (E), and from hereditary factors (H). They examined these proportions in 32 different cancers in which environmental, lifestyle, and genetic factors have been thoroughly studied. Overall, 29% of the driver mutations were due to environment, 5% to heredity, and 66% to R-errors.
The proportions of these drivers did vary widely between the cancer types, the team noted. For example, lung and esophageal cancers and melanoma were primarily driven by environmental factors (more than 60% each). However, they wrote, “even in lung adenocarcinomas, R contributes a third of the total mutations, with tobacco smoke [including secondhand smoke], diet, radiation, and occupational exposures contributing the remainder. In cancers that are less strongly associated with environmental factors, such as those of the pancreas, brain, bone, or prostate, the majority of the mutations are attributable to R.”
During the press briefing, Dr. Tomasetti and Dr. Vogelstein stressed that most of the inevitable R-errors don’t precipitate cancer – and that even if they do increase risk, that risk may not ever trip the disease process.
“Most of the time these replicative mutations do no harm,” Dr Vogelstein said. “They occur in junk DNA genes, or in areas that are unimportant with respect to cancer. That’s the good luck. Occasionally, they occur in a cancer driver gene, and that is bad luck.”
But even a dose of bad luck isn’t enough to cause cancer. Most cancers require multiple hits to develop – which makes primary prevention strategies more important than ever, Dr. Tomasetti said.
“In the case of lung cancer, for instance, three or more mutations are needed. We showed that these mutations are caused by a combination of environment and R-errors. In theory, then, all of these cancers are preventable because if we can prevent even one of the environmentally caused mutations, then that patient won’t develop cancer.”
However, he said, some cancers do appear to be entirely driven by E-errors and, thus, appear entirely unavoidable. This is an extremely difficult area for clinicians and patients to navigate, said Dr. Vogelstein, a former pediatrician.
“We hope that understanding this will offer some comfort to the literally millions of patients who develop cancer despite having lead a near-perfect life,” in terms of managing risk factors. “Cancer develops in people who haven’t smoked, who avoided the sun and wore sunscreen, who eat perfectly healthy diets and exercise regularly. This is a particularly important concept for parents of children who have cancer, who think ‘I either transmitted a bad gene or unknowingly exposed my child to an environmental agent that caused their cancer.’ They need to understand that these cancers would have occurred no matter what they did.”
Dr. Tomasetti had no disclosures. Dr. Vogelstein is on the scientific advisory boards of Morphotek, Exelixis GP, and Sysmex Inostics, and is a founder of PapGene and Personal Genome Diagnostics.
[email protected]
On Twitter @Alz_gal
Key clinical point:
Major finding: Two-thirds (66%) of cancer drivers are replication errors, 29% are environmentally induced, and 5% are hereditary.
Data source: The researchers examined cancer mutation drivers in two cohorts that spanned 69 countries.
Disclosures: Dr. Tomasetti had no disclosures. Dr. Vogelstein is on the scientific advisory boards of Morphotek, Exelixis GP, and Sysmex Inostics, and is a founder of PapGene and Personal Genome Diagnostics.
Breast cancer info on centers’ sites leaves room for improvement
ORLANDO – Women with breast cancer who look for well-rounded information about treatment on the websites of prominent U.S. cancer centers are likely to come up short, suggests a study presented at a symposium on quality care sponsored by the American Society of Clinical Oncology.
Research has shown that nearly all women with breast cancer search the Internet for information about its treatment, and two-thirds report that what they find has a strong influence on their decision-making process (J Cancer Educ. 2013;28[4]:662-8).
But the analysis of content on the websites of 63 National Cancer Institute–designated comprehensive cancer centers or clinical cancer centers found that, on average, they addressed only 21% of a set of key concepts that women need to understand to make informed decisions about surgery, radiation therapy, chemotherapy, hormone therapy, and breast reconstruction. This contrasted starkly with 85% for the National Cancer Institute’s own website (cancer.gov) and 88% for the Susan G. Komen Foundation’s website (komen.org).
“These are websites that we think are reliable, cancer center websites, and these are the most prominent cancer centers in the country,” first author Caleb Dulaney, MD, a resident in radiation oncology at the University of Alabama at Birmingham, said in an interview. “This is where a lot of people receive their care, so they should be very reliable as to the information they provide.
“A lot of websites just had information from the NCI basically integrated into their website or a link to the NCI website,” he acknowledged. “Is it really the goal of the cancer center’s website to provide information? It may not be. But you have to take responsibility for being a trusted source of information. So if you are not going to provide it, you should at least direct people to very accurate, reliable information, and it can also kind of inform what you talk about in clinic.”
All of the investigators evaluating sites in the study were medical professionals, so the team has initiated a new study in which patients will instead perform the evaluations.
“We found that for a few websites, one person found a lot of information and another found no information. So the information may technically be there, but is it transmitted to the patient? Can they find it, and do they understand it?” Dr. Dulaney said. “So it will be interesting when we use patients to evaluate these websites. I’ll be curious to see how many questions they are able to find answers to.”
Study details
For the study, the investigators developed a list of 33 decision-specific knowledge questions about breast cancer treatment by drawing on decision quality instruments that assess how informed a woman’s decision-making process is. The primary outcome was whether the website provided sufficient information to answer each question. The researchers assessed seven measures of accessibility as secondary outcomes.
Results showed that websites contained sufficient content to address only 21% of the decision-specific knowledge questions, Dr. Dulaney reported in a poster session. The value was 17% for questions pertaining to breast surgery and radiation therapy, 18% for those pertaining to chemotherapy and hormone therapy, and 21% for those pertaining to breast reconstruction.
In addition, “a lot of websites put the information in silos,” he noted. “You can read about mastectomy, you can read about lumpectomy, you can read about chemo. But you can’t really get the big picture, which is how do these compare to each other, and which treatment is best for me.”
Even the most commonly addressed single question – what type of reconstruction is most likely to require more than one surgery or procedure – was addressed by only 51% of sites. Proportions were similar for questions pertaining to the type of tumors against which hormone therapy works best (48%) and the schedule for radiation therapy after lumpectomy (47%).
At the other extreme, however, very small proportions of sites addressed questions pertaining to how many women with treated early breast cancer will die from the disease (7%), how many undergoing breast reconstruction will experience complications requiring hospitalization or an unplanned procedure (4%), how skipping chemotherapy and hormone therapy influences risk of death (2%), and whether waiting several weeks to decide about those therapies affects survival (2%). These topics are more negative, Dr. Dulaney observed, “but these are things women need to know.”
None of the websites provided sufficient information to answer all 33 knowledge questions. But perhaps more worrisome, 16% did not provide sufficient information to answer any of them, he said.
When it came to accessibility of information, 94% of sites clearly had a breast cancer–specific page, 87% had information about breast cancer–specific trials, and 86% showed members of the center’s breast cancer team. But only 59% were mobile device friendly as assessed with a Google tool, and merely 24% had obvious links to view information in Spanish.
“A lot of minorities and people of lower socioeconomic status exclusively access the Internet via mobile devices, so they may not have a computer or [other] access to the Internet. But they have a cell phone that is probably a smartphone, and they can get online and search for information that way,” Dr. Dulaney said.
Many oncologists may not have had any say regarding the content and accessibility features of their institution’s website, he acknowledged.
“So we should maybe, number one, try to have more involvement in what information goes on to the website, and two, take a look at our own websites to see what’s on there, because patients are going to look for you, and they are going to associate this information with you,” he said. “If you are at a big institution and you really can’t make a change on your website, you can use alternatives such as social media platforms, things like that, to try and get information out to people.”
From a larger perspective, oncologists have often simply counseled patients that they can’t rely on information they have found online, according to Dr. Dulaney.
“But in this day and age, that can’t really be an answer,” he concluded. “Information on the web is ubiquitous, and there is good information out there. We need to do a better job of speaking up in the conversation. We have the answers to a lot of these questions, we just need to make our voices heard and also direct patients to reliable sources of information.”
ORLANDO – Women with breast cancer who look for well-rounded information about treatment on the websites of prominent U.S. cancer centers are likely to come up short, suggests a study presented at a symposium on quality care sponsored by the American Society of Clinical Oncology.
Research has shown that nearly all women with breast cancer search the Internet for information about its treatment, and two-thirds report that what they find has a strong influence on their decision-making process (J Cancer Educ. 2013;28[4]:662-8).
But the analysis of content on the websites of 63 National Cancer Institute–designated comprehensive cancer centers or clinical cancer centers found that, on average, they addressed only 21% of a set of key concepts that women need to understand to make informed decisions about surgery, radiation therapy, chemotherapy, hormone therapy, and breast reconstruction. This contrasted starkly with 85% for the National Cancer Institute’s own website (cancer.gov) and 88% for the Susan G. Komen Foundation’s website (komen.org).
“These are websites that we think are reliable, cancer center websites, and these are the most prominent cancer centers in the country,” first author Caleb Dulaney, MD, a resident in radiation oncology at the University of Alabama at Birmingham, said in an interview. “This is where a lot of people receive their care, so they should be very reliable as to the information they provide.
“A lot of websites just had information from the NCI basically integrated into their website or a link to the NCI website,” he acknowledged. “Is it really the goal of the cancer center’s website to provide information? It may not be. But you have to take responsibility for being a trusted source of information. So if you are not going to provide it, you should at least direct people to very accurate, reliable information, and it can also kind of inform what you talk about in clinic.”
All of the investigators evaluating sites in the study were medical professionals, so the team has initiated a new study in which patients will instead perform the evaluations.
“We found that for a few websites, one person found a lot of information and another found no information. So the information may technically be there, but is it transmitted to the patient? Can they find it, and do they understand it?” Dr. Dulaney said. “So it will be interesting when we use patients to evaluate these websites. I’ll be curious to see how many questions they are able to find answers to.”
Study details
For the study, the investigators developed a list of 33 decision-specific knowledge questions about breast cancer treatment by drawing on decision quality instruments that assess how informed a woman’s decision-making process is. The primary outcome was whether the website provided sufficient information to answer each question. The researchers assessed seven measures of accessibility as secondary outcomes.
Results showed that websites contained sufficient content to address only 21% of the decision-specific knowledge questions, Dr. Dulaney reported in a poster session. The value was 17% for questions pertaining to breast surgery and radiation therapy, 18% for those pertaining to chemotherapy and hormone therapy, and 21% for those pertaining to breast reconstruction.
In addition, “a lot of websites put the information in silos,” he noted. “You can read about mastectomy, you can read about lumpectomy, you can read about chemo. But you can’t really get the big picture, which is how do these compare to each other, and which treatment is best for me.”
Even the most commonly addressed single question – what type of reconstruction is most likely to require more than one surgery or procedure – was addressed by only 51% of sites. Proportions were similar for questions pertaining to the type of tumors against which hormone therapy works best (48%) and the schedule for radiation therapy after lumpectomy (47%).
At the other extreme, however, very small proportions of sites addressed questions pertaining to how many women with treated early breast cancer will die from the disease (7%), how many undergoing breast reconstruction will experience complications requiring hospitalization or an unplanned procedure (4%), how skipping chemotherapy and hormone therapy influences risk of death (2%), and whether waiting several weeks to decide about those therapies affects survival (2%). These topics are more negative, Dr. Dulaney observed, “but these are things women need to know.”
None of the websites provided sufficient information to answer all 33 knowledge questions. But perhaps more worrisome, 16% did not provide sufficient information to answer any of them, he said.
When it came to accessibility of information, 94% of sites clearly had a breast cancer–specific page, 87% had information about breast cancer–specific trials, and 86% showed members of the center’s breast cancer team. But only 59% were mobile device friendly as assessed with a Google tool, and merely 24% had obvious links to view information in Spanish.
“A lot of minorities and people of lower socioeconomic status exclusively access the Internet via mobile devices, so they may not have a computer or [other] access to the Internet. But they have a cell phone that is probably a smartphone, and they can get online and search for information that way,” Dr. Dulaney said.
Many oncologists may not have had any say regarding the content and accessibility features of their institution’s website, he acknowledged.
“So we should maybe, number one, try to have more involvement in what information goes on to the website, and two, take a look at our own websites to see what’s on there, because patients are going to look for you, and they are going to associate this information with you,” he said. “If you are at a big institution and you really can’t make a change on your website, you can use alternatives such as social media platforms, things like that, to try and get information out to people.”
From a larger perspective, oncologists have often simply counseled patients that they can’t rely on information they have found online, according to Dr. Dulaney.
“But in this day and age, that can’t really be an answer,” he concluded. “Information on the web is ubiquitous, and there is good information out there. We need to do a better job of speaking up in the conversation. We have the answers to a lot of these questions, we just need to make our voices heard and also direct patients to reliable sources of information.”
ORLANDO – Women with breast cancer who look for well-rounded information about treatment on the websites of prominent U.S. cancer centers are likely to come up short, suggests a study presented at a symposium on quality care sponsored by the American Society of Clinical Oncology.
Research has shown that nearly all women with breast cancer search the Internet for information about its treatment, and two-thirds report that what they find has a strong influence on their decision-making process (J Cancer Educ. 2013;28[4]:662-8).
But the analysis of content on the websites of 63 National Cancer Institute–designated comprehensive cancer centers or clinical cancer centers found that, on average, they addressed only 21% of a set of key concepts that women need to understand to make informed decisions about surgery, radiation therapy, chemotherapy, hormone therapy, and breast reconstruction. This contrasted starkly with 85% for the National Cancer Institute’s own website (cancer.gov) and 88% for the Susan G. Komen Foundation’s website (komen.org).
“These are websites that we think are reliable, cancer center websites, and these are the most prominent cancer centers in the country,” first author Caleb Dulaney, MD, a resident in radiation oncology at the University of Alabama at Birmingham, said in an interview. “This is where a lot of people receive their care, so they should be very reliable as to the information they provide.
“A lot of websites just had information from the NCI basically integrated into their website or a link to the NCI website,” he acknowledged. “Is it really the goal of the cancer center’s website to provide information? It may not be. But you have to take responsibility for being a trusted source of information. So if you are not going to provide it, you should at least direct people to very accurate, reliable information, and it can also kind of inform what you talk about in clinic.”
All of the investigators evaluating sites in the study were medical professionals, so the team has initiated a new study in which patients will instead perform the evaluations.
“We found that for a few websites, one person found a lot of information and another found no information. So the information may technically be there, but is it transmitted to the patient? Can they find it, and do they understand it?” Dr. Dulaney said. “So it will be interesting when we use patients to evaluate these websites. I’ll be curious to see how many questions they are able to find answers to.”
Study details
For the study, the investigators developed a list of 33 decision-specific knowledge questions about breast cancer treatment by drawing on decision quality instruments that assess how informed a woman’s decision-making process is. The primary outcome was whether the website provided sufficient information to answer each question. The researchers assessed seven measures of accessibility as secondary outcomes.
Results showed that websites contained sufficient content to address only 21% of the decision-specific knowledge questions, Dr. Dulaney reported in a poster session. The value was 17% for questions pertaining to breast surgery and radiation therapy, 18% for those pertaining to chemotherapy and hormone therapy, and 21% for those pertaining to breast reconstruction.
In addition, “a lot of websites put the information in silos,” he noted. “You can read about mastectomy, you can read about lumpectomy, you can read about chemo. But you can’t really get the big picture, which is how do these compare to each other, and which treatment is best for me.”
Even the most commonly addressed single question – what type of reconstruction is most likely to require more than one surgery or procedure – was addressed by only 51% of sites. Proportions were similar for questions pertaining to the type of tumors against which hormone therapy works best (48%) and the schedule for radiation therapy after lumpectomy (47%).
At the other extreme, however, very small proportions of sites addressed questions pertaining to how many women with treated early breast cancer will die from the disease (7%), how many undergoing breast reconstruction will experience complications requiring hospitalization or an unplanned procedure (4%), how skipping chemotherapy and hormone therapy influences risk of death (2%), and whether waiting several weeks to decide about those therapies affects survival (2%). These topics are more negative, Dr. Dulaney observed, “but these are things women need to know.”
None of the websites provided sufficient information to answer all 33 knowledge questions. But perhaps more worrisome, 16% did not provide sufficient information to answer any of them, he said.
When it came to accessibility of information, 94% of sites clearly had a breast cancer–specific page, 87% had information about breast cancer–specific trials, and 86% showed members of the center’s breast cancer team. But only 59% were mobile device friendly as assessed with a Google tool, and merely 24% had obvious links to view information in Spanish.
“A lot of minorities and people of lower socioeconomic status exclusively access the Internet via mobile devices, so they may not have a computer or [other] access to the Internet. But they have a cell phone that is probably a smartphone, and they can get online and search for information that way,” Dr. Dulaney said.
Many oncologists may not have had any say regarding the content and accessibility features of their institution’s website, he acknowledged.
“So we should maybe, number one, try to have more involvement in what information goes on to the website, and two, take a look at our own websites to see what’s on there, because patients are going to look for you, and they are going to associate this information with you,” he said. “If you are at a big institution and you really can’t make a change on your website, you can use alternatives such as social media platforms, things like that, to try and get information out to people.”
From a larger perspective, oncologists have often simply counseled patients that they can’t rely on information they have found online, according to Dr. Dulaney.
“But in this day and age, that can’t really be an answer,” he concluded. “Information on the web is ubiquitous, and there is good information out there. We need to do a better job of speaking up in the conversation. We have the answers to a lot of these questions, we just need to make our voices heard and also direct patients to reliable sources of information.”
AT THE QUALITY CARE SYMPOSIUM
Key clinical point:
Major finding: On average, the sites addressed 21% of 33 key concepts needed to make informed decisions about treatment.
Data source: An analysis of breast cancer information on the websites of 63 NCI-designated comprehensive cancer centers or clinical cancer centers.
Disclosures: Dr. Dulaney disclosed that he had no relevant conflicts of interest.
FDA approves ribociclib for HR+, HER2– advanced breast cancer
The Food and Drug Administration has approved ribociclib, a cyclin-dependent kinase (CDK) 4/6 inhibitor, in combination with an aromatase inhibitor as initial endocrine-based therapy for the treatment of postmenopausal women with hormone receptor (HR)-positive, human epidermal growth factor receptor 2 (HER2)-negative advanced or metastatic breast cancer.
Overall survival data is immature but approval was based on improvement in progression-free survival (PFS) among 334 women randomized to receive ribociclib plus letrozole compared to 334 women randomized to receive placebo plus letrozole in phase III MONALEESA-2 (hazard ratio, 0.556; 95% CI: 0.429, 0.720; P less than .0001).
Ribociclib 600 mg or placebo was administered orally once daily for 21 consecutive days, followed by 7 days off, with letrozole 2.5 mg administered orally once daily for 28 days. All patients were postmenopausal women with HR-positive, HER2-negative advanced or metastatic breast cancer who received no prior therapy for advanced disease. Treatment continued until disease progression or unacceptable toxicity,
The most common adverse reactions in patients taking ribociclib were neutropenia, nausea, fatigue, diarrhea, leukopenia, alopecia, vomiting, constipation, headache, and back pain. The most common grade 3 or 4 adverse reactions were neutropenia, leukopenia, abnormal liver function tests, lymphopenia, and vomiting. Ribociclib has been shown to prolong the QT interval in a concentration-dependent manner, the FDA warns.
Ribociclib is the second CDK4/6 inhibitor to receive approval for advanced breast cancer, following the accelerated approval of palbociclib (Ibrance) plus letrozole (Femara) as a first-line treatment for postmenopausal women with ER-postive, HER2-negative metastatic breast cancer in 2015. The FDA expanded the indication for fulvestrant to include use in combination with palbociclib in 2016.
Phase II trial data indicating activity of a third CDK 4/6 inhibitor, abemaciclib, in this patient population was presented at the 2016 ASCO Annual Meeting.
The recommended starting dose of ribociclib is 600 mg orally (three 200-mg tablets) taken once daily with or without food for 21 consecutive days followed by 7 days off treatment.
Full prescribing information for ribociclib is available here.
Ribociclib is being marketed as Kisqali by Novartis Pharmaceuticals Corp.
[email protected]
On Twitter @NikolaidesLaura
The Food and Drug Administration has approved ribociclib, a cyclin-dependent kinase (CDK) 4/6 inhibitor, in combination with an aromatase inhibitor as initial endocrine-based therapy for the treatment of postmenopausal women with hormone receptor (HR)-positive, human epidermal growth factor receptor 2 (HER2)-negative advanced or metastatic breast cancer.
Overall survival data is immature but approval was based on improvement in progression-free survival (PFS) among 334 women randomized to receive ribociclib plus letrozole compared to 334 women randomized to receive placebo plus letrozole in phase III MONALEESA-2 (hazard ratio, 0.556; 95% CI: 0.429, 0.720; P less than .0001).
Ribociclib 600 mg or placebo was administered orally once daily for 21 consecutive days, followed by 7 days off, with letrozole 2.5 mg administered orally once daily for 28 days. All patients were postmenopausal women with HR-positive, HER2-negative advanced or metastatic breast cancer who received no prior therapy for advanced disease. Treatment continued until disease progression or unacceptable toxicity,
The most common adverse reactions in patients taking ribociclib were neutropenia, nausea, fatigue, diarrhea, leukopenia, alopecia, vomiting, constipation, headache, and back pain. The most common grade 3 or 4 adverse reactions were neutropenia, leukopenia, abnormal liver function tests, lymphopenia, and vomiting. Ribociclib has been shown to prolong the QT interval in a concentration-dependent manner, the FDA warns.
Ribociclib is the second CDK4/6 inhibitor to receive approval for advanced breast cancer, following the accelerated approval of palbociclib (Ibrance) plus letrozole (Femara) as a first-line treatment for postmenopausal women with ER-postive, HER2-negative metastatic breast cancer in 2015. The FDA expanded the indication for fulvestrant to include use in combination with palbociclib in 2016.
Phase II trial data indicating activity of a third CDK 4/6 inhibitor, abemaciclib, in this patient population was presented at the 2016 ASCO Annual Meeting.
The recommended starting dose of ribociclib is 600 mg orally (three 200-mg tablets) taken once daily with or without food for 21 consecutive days followed by 7 days off treatment.
Full prescribing information for ribociclib is available here.
Ribociclib is being marketed as Kisqali by Novartis Pharmaceuticals Corp.
[email protected]
On Twitter @NikolaidesLaura
The Food and Drug Administration has approved ribociclib, a cyclin-dependent kinase (CDK) 4/6 inhibitor, in combination with an aromatase inhibitor as initial endocrine-based therapy for the treatment of postmenopausal women with hormone receptor (HR)-positive, human epidermal growth factor receptor 2 (HER2)-negative advanced or metastatic breast cancer.
Overall survival data is immature but approval was based on improvement in progression-free survival (PFS) among 334 women randomized to receive ribociclib plus letrozole compared to 334 women randomized to receive placebo plus letrozole in phase III MONALEESA-2 (hazard ratio, 0.556; 95% CI: 0.429, 0.720; P less than .0001).
Ribociclib 600 mg or placebo was administered orally once daily for 21 consecutive days, followed by 7 days off, with letrozole 2.5 mg administered orally once daily for 28 days. All patients were postmenopausal women with HR-positive, HER2-negative advanced or metastatic breast cancer who received no prior therapy for advanced disease. Treatment continued until disease progression or unacceptable toxicity,
The most common adverse reactions in patients taking ribociclib were neutropenia, nausea, fatigue, diarrhea, leukopenia, alopecia, vomiting, constipation, headache, and back pain. The most common grade 3 or 4 adverse reactions were neutropenia, leukopenia, abnormal liver function tests, lymphopenia, and vomiting. Ribociclib has been shown to prolong the QT interval in a concentration-dependent manner, the FDA warns.
Ribociclib is the second CDK4/6 inhibitor to receive approval for advanced breast cancer, following the accelerated approval of palbociclib (Ibrance) plus letrozole (Femara) as a first-line treatment for postmenopausal women with ER-postive, HER2-negative metastatic breast cancer in 2015. The FDA expanded the indication for fulvestrant to include use in combination with palbociclib in 2016.
Phase II trial data indicating activity of a third CDK 4/6 inhibitor, abemaciclib, in this patient population was presented at the 2016 ASCO Annual Meeting.
The recommended starting dose of ribociclib is 600 mg orally (three 200-mg tablets) taken once daily with or without food for 21 consecutive days followed by 7 days off treatment.
Full prescribing information for ribociclib is available here.
Ribociclib is being marketed as Kisqali by Novartis Pharmaceuticals Corp.
[email protected]
On Twitter @NikolaidesLaura