User login
Report details progress, obstacles in cancer research and care
Deaths from cancer are on the decline in the US, but new cases of cancer are on the rise, according to the 7th annual American Association for Cancer Research (AACR) Cancer Progress Report.
The data suggest the cancer death rate declined by 35% from 1991 to 2014 for children and by 25% for adults, a reduction that translates to 2.1 million cancer deaths avoided.
However, 600,920 people in the US are projected to die from cancer in 2017.
And the number of new cancer cases is predicted to rise from 1.7 million in 2017 to 2.3 million in 2030.
The report also estimates there will be 62,130 new cases of leukemia in 2017 and 24,500 leukemia deaths this year.
This includes:
- 5970 cases of acute lymphocytic leukemia and 1440 deaths
- 20,110 cases of chronic lymphocytic leukemia and 4660 deaths
- 21,380 cases of acute myeloid leukemia (AML) and 10,590 deaths
- 8950 cases of chronic myeloid leukemia and 1080 deaths.
The estimate for lymphomas is 80,500 new cases and 21,210 deaths.
This includes:
- 8260 cases of Hodgkin lymphoma (HL) and 1070 deaths
- 72,240 cases of non-Hodgkin lymphoma and 20,140 deaths.
The estimate for myeloma is 30,280 new cases and 12,590 deaths.
The report says the estimated new cases of cancer are based on cancer incidence rates from 49 states and the District of Columbia from 1995 through 2013, as reported by the North American Association of Central Cancer Registries. This represents about 98% of the US population.
The estimated deaths are based on US mortality data from 1997 through 2013, taken from the National Center for Health Statistics of the Centers for Disease Control and Prevention.
Drug approvals
The AACR report notes that, between August 1, 2016, and July 31, 2017, the US Food and Drug Administration (FDA) approved new uses for 15 anticancer agents, 9 of which had no previous FDA approval.
Five of the agents are immunotherapies, which the report dubs “revolutionary treatments that are increasing survival and improving quality of life for patients.”
Among the recently approved therapies are 3 used for hematology indications:
- Ibrutinib (Imbruvica), approved to treat patients with relapsed/refractory marginal zone lymphoma who require systemic therapy and have received at least 1 prior anti-CD20-based therapy
- Midostaurin (Rydapt), approved as monotherapy for adults with advanced systemic mastocytosis and for use in combination with standard cytarabine and daunorubicin induction, followed by cytarabine consolidation, in adults with newly diagnosed AML who are FLT3 mutation-positive, as detected by an FDA-approved test.
- Pembrolizumab (Keytruda), approved to treat adult and pediatric patients with refractory classical HL or those with classical HL who have relapsed after 3 or more prior lines of therapy.
Disparities and costs
The AACR report points out that advances against cancer have not benefited everyone equally, and cancer health disparities are some of the most pressing challenges.
Among the disparities listed is the fact that adolescents and young adults (ages 15 to 39) with AML have a 5-year relative survival rate that is 22% lower than that of children (ages 1 to 14) with AML.
And Hispanic children are 24% more likely to develop leukemia than non-Hispanic children.
Another concern mentioned in the report is the cost of cancer care. The direct medical costs of cancer care in 2014 were estimated to be nearly $87.6 billion. This number does not include the indirect costs of lost productivity due to cancer-related morbidity and mortality.
With this in mind, the AACR is calling for a $2 billion increase in funding for the National Institutes of Health in fiscal year 2018, for a total funding level of $36.2 billion.
The AACR also recommends an $80 million increase in the FDA budget, bringing it to $2.8 billion for fiscal year 2018. ![]()
Deaths from cancer are on the decline in the US, but new cases of cancer are on the rise, according to the 7th annual American Association for Cancer Research (AACR) Cancer Progress Report.
The data suggest the cancer death rate declined by 35% from 1991 to 2014 for children and by 25% for adults, a reduction that translates to 2.1 million cancer deaths avoided.
However, 600,920 people in the US are projected to die from cancer in 2017.
And the number of new cancer cases is predicted to rise from 1.7 million in 2017 to 2.3 million in 2030.
The report also estimates there will be 62,130 new cases of leukemia in 2017 and 24,500 leukemia deaths this year.
This includes:
- 5970 cases of acute lymphocytic leukemia and 1440 deaths
- 20,110 cases of chronic lymphocytic leukemia and 4660 deaths
- 21,380 cases of acute myeloid leukemia (AML) and 10,590 deaths
- 8950 cases of chronic myeloid leukemia and 1080 deaths.
The estimate for lymphomas is 80,500 new cases and 21,210 deaths.
This includes:
- 8260 cases of Hodgkin lymphoma (HL) and 1070 deaths
- 72,240 cases of non-Hodgkin lymphoma and 20,140 deaths.
The estimate for myeloma is 30,280 new cases and 12,590 deaths.
The report says the estimated new cases of cancer are based on cancer incidence rates from 49 states and the District of Columbia from 1995 through 2013, as reported by the North American Association of Central Cancer Registries. This represents about 98% of the US population.
The estimated deaths are based on US mortality data from 1997 through 2013, taken from the National Center for Health Statistics of the Centers for Disease Control and Prevention.
Drug approvals
The AACR report notes that, between August 1, 2016, and July 31, 2017, the US Food and Drug Administration (FDA) approved new uses for 15 anticancer agents, 9 of which had no previous FDA approval.
Five of the agents are immunotherapies, which the report dubs “revolutionary treatments that are increasing survival and improving quality of life for patients.”
Among the recently approved therapies are 3 used for hematology indications:
- Ibrutinib (Imbruvica), approved to treat patients with relapsed/refractory marginal zone lymphoma who require systemic therapy and have received at least 1 prior anti-CD20-based therapy
- Midostaurin (Rydapt), approved as monotherapy for adults with advanced systemic mastocytosis and for use in combination with standard cytarabine and daunorubicin induction, followed by cytarabine consolidation, in adults with newly diagnosed AML who are FLT3 mutation-positive, as detected by an FDA-approved test.
- Pembrolizumab (Keytruda), approved to treat adult and pediatric patients with refractory classical HL or those with classical HL who have relapsed after 3 or more prior lines of therapy.
Disparities and costs
The AACR report points out that advances against cancer have not benefited everyone equally, and cancer health disparities are some of the most pressing challenges.
Among the disparities listed is the fact that adolescents and young adults (ages 15 to 39) with AML have a 5-year relative survival rate that is 22% lower than that of children (ages 1 to 14) with AML.
And Hispanic children are 24% more likely to develop leukemia than non-Hispanic children.
Another concern mentioned in the report is the cost of cancer care. The direct medical costs of cancer care in 2014 were estimated to be nearly $87.6 billion. This number does not include the indirect costs of lost productivity due to cancer-related morbidity and mortality.
With this in mind, the AACR is calling for a $2 billion increase in funding for the National Institutes of Health in fiscal year 2018, for a total funding level of $36.2 billion.
The AACR also recommends an $80 million increase in the FDA budget, bringing it to $2.8 billion for fiscal year 2018. ![]()
Deaths from cancer are on the decline in the US, but new cases of cancer are on the rise, according to the 7th annual American Association for Cancer Research (AACR) Cancer Progress Report.
The data suggest the cancer death rate declined by 35% from 1991 to 2014 for children and by 25% for adults, a reduction that translates to 2.1 million cancer deaths avoided.
However, 600,920 people in the US are projected to die from cancer in 2017.
And the number of new cancer cases is predicted to rise from 1.7 million in 2017 to 2.3 million in 2030.
The report also estimates there will be 62,130 new cases of leukemia in 2017 and 24,500 leukemia deaths this year.
This includes:
- 5970 cases of acute lymphocytic leukemia and 1440 deaths
- 20,110 cases of chronic lymphocytic leukemia and 4660 deaths
- 21,380 cases of acute myeloid leukemia (AML) and 10,590 deaths
- 8950 cases of chronic myeloid leukemia and 1080 deaths.
The estimate for lymphomas is 80,500 new cases and 21,210 deaths.
This includes:
- 8260 cases of Hodgkin lymphoma (HL) and 1070 deaths
- 72,240 cases of non-Hodgkin lymphoma and 20,140 deaths.
The estimate for myeloma is 30,280 new cases and 12,590 deaths.
The report says the estimated new cases of cancer are based on cancer incidence rates from 49 states and the District of Columbia from 1995 through 2013, as reported by the North American Association of Central Cancer Registries. This represents about 98% of the US population.
The estimated deaths are based on US mortality data from 1997 through 2013, taken from the National Center for Health Statistics of the Centers for Disease Control and Prevention.
Drug approvals
The AACR report notes that, between August 1, 2016, and July 31, 2017, the US Food and Drug Administration (FDA) approved new uses for 15 anticancer agents, 9 of which had no previous FDA approval.
Five of the agents are immunotherapies, which the report dubs “revolutionary treatments that are increasing survival and improving quality of life for patients.”
Among the recently approved therapies are 3 used for hematology indications:
- Ibrutinib (Imbruvica), approved to treat patients with relapsed/refractory marginal zone lymphoma who require systemic therapy and have received at least 1 prior anti-CD20-based therapy
- Midostaurin (Rydapt), approved as monotherapy for adults with advanced systemic mastocytosis and for use in combination with standard cytarabine and daunorubicin induction, followed by cytarabine consolidation, in adults with newly diagnosed AML who are FLT3 mutation-positive, as detected by an FDA-approved test.
- Pembrolizumab (Keytruda), approved to treat adult and pediatric patients with refractory classical HL or those with classical HL who have relapsed after 3 or more prior lines of therapy.
Disparities and costs
The AACR report points out that advances against cancer have not benefited everyone equally, and cancer health disparities are some of the most pressing challenges.
Among the disparities listed is the fact that adolescents and young adults (ages 15 to 39) with AML have a 5-year relative survival rate that is 22% lower than that of children (ages 1 to 14) with AML.
And Hispanic children are 24% more likely to develop leukemia than non-Hispanic children.
Another concern mentioned in the report is the cost of cancer care. The direct medical costs of cancer care in 2014 were estimated to be nearly $87.6 billion. This number does not include the indirect costs of lost productivity due to cancer-related morbidity and mortality.
With this in mind, the AACR is calling for a $2 billion increase in funding for the National Institutes of Health in fiscal year 2018, for a total funding level of $36.2 billion.
The AACR also recommends an $80 million increase in the FDA budget, bringing it to $2.8 billion for fiscal year 2018. ![]()
TKI granted priority review for newly diagnosed CML
The US Food and Drug Administration (FDA) has granted priority review to a supplemental new drug application (sNDA) for the tyrosine kinase inhibitor (TKI) bosutinib (Bosulif®).
If approved, the sNDA would expand the use of bosutinib to include patients with newly diagnosed, chronic phase, Philadelphia chromosome-positive (Ph+) chronic myeloid leukemia (CML).
Bosutinib is currently FDA-approved to treat adults with chronic, accelerated, or blast phase Ph+ CML with resistance or intolerance to prior therapy.
The FDA grants priority review to applications for products that may provide significant improvements in the treatment, diagnosis, or prevention of serious conditions.
The agency’s goal is to take action on a priority review application within 6 months of receiving it, rather than the standard 10 months.
The FDA plans to make a decision on the sNDA for bosutinib by the end of this year.
Meanwhile, the European Medicines Agency (EMA) has validated for review a type II variation application for bosutinib in patients with newly diagnosed, chronic phase Ph+ CML.
Bosutinib already has conditional marketing authorization in the European Economic Area for the treatment of adults with Ph+ CML who previously received at least 1 TKI and adults with Ph+ CML for whom imatinib, nilotinib, and dasatinib are not considered appropriate.
Phase 3 trial
The applications submitted to the EMA and FDA are both supported by early results from the phase 3 BFORE trial. Results from this trial were presented at the ASCO Annual Meeting in May.
In this ongoing study, researchers are comparing bosutinib and imatinib as first-line treatment of chronic phase CML.
As of the ASCO presentation, the trial had enrolled 536 patients who were randomized 1:1 to receive bosutinib (n=268) or imatinib (n=268).
The presentation included results in a modified intent-to-treat population of Ph+ patients with e13a2/e14a2 transcripts who had at least 12 months of follow-up. In this group, there were 246 patients in the bosutinib arm and 241 in the imatinib arm.
Most of the patients were still on therapy at the 12-month mark or beyond—78% in the bosutinib arm and 73.2% in the imatinib arm. The median treatment duration was 14.1 months and 13.8 months, respectively.
At 12 months, the rate of major molecular response was 47.2% in the bosutinib arm and 36.9% in the imatinib arm (P= 0.02). The rate of complete cytogenetic response was 77.2% and 66.4%, respectively (P<0.008).
One patient in the bosutinib arm and 4 in the imatinib arm discontinued treatment due to disease progression, while 12.7% and 8.7%, respectively, discontinued treatment due to drug-related toxicity.
Adverse events that were more common in the bosutinib arm than the imatinib arm included grade 3 or higher diarrhea (7.8% vs 0.8%), increased alanine levels (19% vs 1.5%), increased aspartate levels (9.7% vs 1.9%), cardiovascular events (3% vs 0.4%), and peripheral vascular events (1.5% vs 1.1%). Cerebrovascular events were more common with imatinib than bosutinib (0.4% and 0%, respectively). ![]()
The US Food and Drug Administration (FDA) has granted priority review to a supplemental new drug application (sNDA) for the tyrosine kinase inhibitor (TKI) bosutinib (Bosulif®).
If approved, the sNDA would expand the use of bosutinib to include patients with newly diagnosed, chronic phase, Philadelphia chromosome-positive (Ph+) chronic myeloid leukemia (CML).
Bosutinib is currently FDA-approved to treat adults with chronic, accelerated, or blast phase Ph+ CML with resistance or intolerance to prior therapy.
The FDA grants priority review to applications for products that may provide significant improvements in the treatment, diagnosis, or prevention of serious conditions.
The agency’s goal is to take action on a priority review application within 6 months of receiving it, rather than the standard 10 months.
The FDA plans to make a decision on the sNDA for bosutinib by the end of this year.
Meanwhile, the European Medicines Agency (EMA) has validated for review a type II variation application for bosutinib in patients with newly diagnosed, chronic phase Ph+ CML.
Bosutinib already has conditional marketing authorization in the European Economic Area for the treatment of adults with Ph+ CML who previously received at least 1 TKI and adults with Ph+ CML for whom imatinib, nilotinib, and dasatinib are not considered appropriate.
Phase 3 trial
The applications submitted to the EMA and FDA are both supported by early results from the phase 3 BFORE trial. Results from this trial were presented at the ASCO Annual Meeting in May.
In this ongoing study, researchers are comparing bosutinib and imatinib as first-line treatment of chronic phase CML.
As of the ASCO presentation, the trial had enrolled 536 patients who were randomized 1:1 to receive bosutinib (n=268) or imatinib (n=268).
The presentation included results in a modified intent-to-treat population of Ph+ patients with e13a2/e14a2 transcripts who had at least 12 months of follow-up. In this group, there were 246 patients in the bosutinib arm and 241 in the imatinib arm.
Most of the patients were still on therapy at the 12-month mark or beyond—78% in the bosutinib arm and 73.2% in the imatinib arm. The median treatment duration was 14.1 months and 13.8 months, respectively.
At 12 months, the rate of major molecular response was 47.2% in the bosutinib arm and 36.9% in the imatinib arm (P= 0.02). The rate of complete cytogenetic response was 77.2% and 66.4%, respectively (P<0.008).
One patient in the bosutinib arm and 4 in the imatinib arm discontinued treatment due to disease progression, while 12.7% and 8.7%, respectively, discontinued treatment due to drug-related toxicity.
Adverse events that were more common in the bosutinib arm than the imatinib arm included grade 3 or higher diarrhea (7.8% vs 0.8%), increased alanine levels (19% vs 1.5%), increased aspartate levels (9.7% vs 1.9%), cardiovascular events (3% vs 0.4%), and peripheral vascular events (1.5% vs 1.1%). Cerebrovascular events were more common with imatinib than bosutinib (0.4% and 0%, respectively). ![]()
The US Food and Drug Administration (FDA) has granted priority review to a supplemental new drug application (sNDA) for the tyrosine kinase inhibitor (TKI) bosutinib (Bosulif®).
If approved, the sNDA would expand the use of bosutinib to include patients with newly diagnosed, chronic phase, Philadelphia chromosome-positive (Ph+) chronic myeloid leukemia (CML).
Bosutinib is currently FDA-approved to treat adults with chronic, accelerated, or blast phase Ph+ CML with resistance or intolerance to prior therapy.
The FDA grants priority review to applications for products that may provide significant improvements in the treatment, diagnosis, or prevention of serious conditions.
The agency’s goal is to take action on a priority review application within 6 months of receiving it, rather than the standard 10 months.
The FDA plans to make a decision on the sNDA for bosutinib by the end of this year.
Meanwhile, the European Medicines Agency (EMA) has validated for review a type II variation application for bosutinib in patients with newly diagnosed, chronic phase Ph+ CML.
Bosutinib already has conditional marketing authorization in the European Economic Area for the treatment of adults with Ph+ CML who previously received at least 1 TKI and adults with Ph+ CML for whom imatinib, nilotinib, and dasatinib are not considered appropriate.
Phase 3 trial
The applications submitted to the EMA and FDA are both supported by early results from the phase 3 BFORE trial. Results from this trial were presented at the ASCO Annual Meeting in May.
In this ongoing study, researchers are comparing bosutinib and imatinib as first-line treatment of chronic phase CML.
As of the ASCO presentation, the trial had enrolled 536 patients who were randomized 1:1 to receive bosutinib (n=268) or imatinib (n=268).
The presentation included results in a modified intent-to-treat population of Ph+ patients with e13a2/e14a2 transcripts who had at least 12 months of follow-up. In this group, there were 246 patients in the bosutinib arm and 241 in the imatinib arm.
Most of the patients were still on therapy at the 12-month mark or beyond—78% in the bosutinib arm and 73.2% in the imatinib arm. The median treatment duration was 14.1 months and 13.8 months, respectively.
At 12 months, the rate of major molecular response was 47.2% in the bosutinib arm and 36.9% in the imatinib arm (P= 0.02). The rate of complete cytogenetic response was 77.2% and 66.4%, respectively (P<0.008).
One patient in the bosutinib arm and 4 in the imatinib arm discontinued treatment due to disease progression, while 12.7% and 8.7%, respectively, discontinued treatment due to drug-related toxicity.
Adverse events that were more common in the bosutinib arm than the imatinib arm included grade 3 or higher diarrhea (7.8% vs 0.8%), increased alanine levels (19% vs 1.5%), increased aspartate levels (9.7% vs 1.9%), cardiovascular events (3% vs 0.4%), and peripheral vascular events (1.5% vs 1.1%). Cerebrovascular events were more common with imatinib than bosutinib (0.4% and 0%, respectively). ![]()
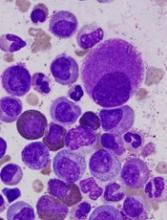
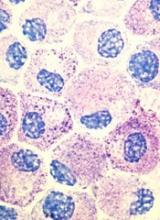
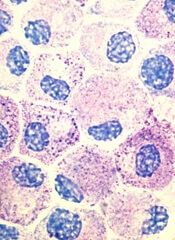
Popular theory of mast cell development is wrong, team says
Stem cell factor (SCF) and KIT signaling are not necessary for early mast cell development, according to research published in Blood.
It has been assumed that the differentiation of hematopoietic progenitors to mast cells requires SCF and KIT signaling.
However, researchers found that mast cell progenitors can survive, mature, and proliferate in the absence of SCF and KIT signaling.
The researchers began this work by analyzing mast cell progenitor populations in samples from healthy subjects, patients with chronic myeloid leukemia (CML) or gastrointestinal stromal tumors (GIST) who were treated with imatinib, and patients with systemic mastocytosis carrying the D816V KIT mutation.
Imatinib inhibits KIT signaling, and the D816V KIT mutation causes KIT signaling to be constitutively active.
The researchers found the imatinib-treated CML and GIST patients and the patients with systemic mastocytosis all had mast cell progenitor populations similar to those observed in healthy subjects.
The team therefore concluded that dysfunctional KIT signaling does not affect the frequency of circulating mast cell progenitors in vivo.
On the other hand, the researchers also found that circulating mast cells were sensitive to imatinib in patients with CML. The patients had higher numbers of peripheral blood mast cells at diagnosis than they did after treatment with imatinib.
“When the patients were treated with the drug imatinib, which blocks the effect of stem cell factor, the number of mature mast cells dropped, while the number of progenitor cells did not change,” said study author Gunnar Nilsson, PhD, of Karolinska Institutet in Stockholm, Sweden.
Subsequent experiments showed that mast cell progenitors can survive in vitro without KIT signaling and without SCF. In addition, mast cell progenitors were able to mature and proliferate in vitro without SCF.
In fact, the researchers said they found that interleukin 3 was sufficient to promote the survival of mast cell progenitors in vitro.
“The study increases our understanding of how mast cells are formed and could be important in the development of new therapies, for example, for mastocytosis . . . ,” said study author Joakim Dahlin, PhD, of the University of Cambridge in the UK.
“One hypothesis that we will now test is whether interleukin 3 can be a new target in the treatment of mast cell-driven diseases.” ![]()
Stem cell factor (SCF) and KIT signaling are not necessary for early mast cell development, according to research published in Blood.
It has been assumed that the differentiation of hematopoietic progenitors to mast cells requires SCF and KIT signaling.
However, researchers found that mast cell progenitors can survive, mature, and proliferate in the absence of SCF and KIT signaling.
The researchers began this work by analyzing mast cell progenitor populations in samples from healthy subjects, patients with chronic myeloid leukemia (CML) or gastrointestinal stromal tumors (GIST) who were treated with imatinib, and patients with systemic mastocytosis carrying the D816V KIT mutation.
Imatinib inhibits KIT signaling, and the D816V KIT mutation causes KIT signaling to be constitutively active.
The researchers found the imatinib-treated CML and GIST patients and the patients with systemic mastocytosis all had mast cell progenitor populations similar to those observed in healthy subjects.
The team therefore concluded that dysfunctional KIT signaling does not affect the frequency of circulating mast cell progenitors in vivo.
On the other hand, the researchers also found that circulating mast cells were sensitive to imatinib in patients with CML. The patients had higher numbers of peripheral blood mast cells at diagnosis than they did after treatment with imatinib.
“When the patients were treated with the drug imatinib, which blocks the effect of stem cell factor, the number of mature mast cells dropped, while the number of progenitor cells did not change,” said study author Gunnar Nilsson, PhD, of Karolinska Institutet in Stockholm, Sweden.
Subsequent experiments showed that mast cell progenitors can survive in vitro without KIT signaling and without SCF. In addition, mast cell progenitors were able to mature and proliferate in vitro without SCF.
In fact, the researchers said they found that interleukin 3 was sufficient to promote the survival of mast cell progenitors in vitro.
“The study increases our understanding of how mast cells are formed and could be important in the development of new therapies, for example, for mastocytosis . . . ,” said study author Joakim Dahlin, PhD, of the University of Cambridge in the UK.
“One hypothesis that we will now test is whether interleukin 3 can be a new target in the treatment of mast cell-driven diseases.” ![]()
Stem cell factor (SCF) and KIT signaling are not necessary for early mast cell development, according to research published in Blood.
It has been assumed that the differentiation of hematopoietic progenitors to mast cells requires SCF and KIT signaling.
However, researchers found that mast cell progenitors can survive, mature, and proliferate in the absence of SCF and KIT signaling.
The researchers began this work by analyzing mast cell progenitor populations in samples from healthy subjects, patients with chronic myeloid leukemia (CML) or gastrointestinal stromal tumors (GIST) who were treated with imatinib, and patients with systemic mastocytosis carrying the D816V KIT mutation.
Imatinib inhibits KIT signaling, and the D816V KIT mutation causes KIT signaling to be constitutively active.
The researchers found the imatinib-treated CML and GIST patients and the patients with systemic mastocytosis all had mast cell progenitor populations similar to those observed in healthy subjects.
The team therefore concluded that dysfunctional KIT signaling does not affect the frequency of circulating mast cell progenitors in vivo.
On the other hand, the researchers also found that circulating mast cells were sensitive to imatinib in patients with CML. The patients had higher numbers of peripheral blood mast cells at diagnosis than they did after treatment with imatinib.
“When the patients were treated with the drug imatinib, which blocks the effect of stem cell factor, the number of mature mast cells dropped, while the number of progenitor cells did not change,” said study author Gunnar Nilsson, PhD, of Karolinska Institutet in Stockholm, Sweden.
Subsequent experiments showed that mast cell progenitors can survive in vitro without KIT signaling and without SCF. In addition, mast cell progenitors were able to mature and proliferate in vitro without SCF.
In fact, the researchers said they found that interleukin 3 was sufficient to promote the survival of mast cell progenitors in vitro.
“The study increases our understanding of how mast cells are formed and could be important in the development of new therapies, for example, for mastocytosis . . . ,” said study author Joakim Dahlin, PhD, of the University of Cambridge in the UK.
“One hypothesis that we will now test is whether interleukin 3 can be a new target in the treatment of mast cell-driven diseases.” ![]()
TKI dose de-escalation feasible in CML with stable MMR
Tyrosine kinase inhibitor de-escalation is generally safe and may lead to symptom improvement in patients with chronic myeloid leukemia who respond well to the therapy, according to an interim analysis of the nonrandomized phase 2 DESTINY trial.
The findings imply that some patients are unnecessarily overtreated, as responses were sometimes maintained with lower tyrosine kinase inhibitor (TKI) doses, according to Richard E. Clark, MD, of the University of Liverpool, England, and his colleagues (Lancet Haematol. 2017 May 26;4:e310-16).
Of 174 patients with chronic myeloid leukemia (CML) in first chronic phase who had received TKIs for at least 3 years and who were either in stable molecular response 4 logs below the standard arbitrary baseline (MR4, 125 patients) or stable major molecular response (MMR) but not MR4 (49 patients) for at least 12 months at the time of enrollment, 12 (7%) had molecular recurrence during 12 months of half-dose TKI therapy, and all patients who experienced molecular recurrence regained MMR within 4 months of being back on full-dose TKIs, the researchers noted.
The median time to recovery was 77 days.
The recurrence rate was significantly lower in the MR4 cohort than in the MMR cohort (2% vs. 19%; hazard ratio, 0.12), and time to relapse was significantly longer in the MR4 cohort (median, 8.7 vs. 4.4 months), the investigators said.
Recurrence was not associated with age, sex, weight, performance status, BCR-ABL1 transcript type, or duration of TKI therapy, which was a median of 6.9 years overall, they noted.
During the first 3 months of de-escalation, adverse events associated with treatment, including lethargy, diarrhea, rash, and nausea, all improved.
Adverse events during de-escalation occurred in 16 patients, and included one fatality due to worsening of peripheral arterial occlusive disease, but all were deemed unrelated to the TKI or underlying chronic CML, the investigators noted.
Study subjects were adults with positive BCR-ABL1 transcripts, with either e13a2, e14a2, or e19a2 fusion transcript. They were enrolled between December 2013 and April 2015 from 20 hospitals in the United Kingdom; 148 were receiving imatinib, 16 were receiving nilotinib, and 10 were receiving dasatinib. They received half of their standard dose of imatinib (200 mg daily), dasatinib (50 mg daily), or nilotinib (200 mg twice daily) for 12 months. Recurrence during de-escalation was defined as BCR-ABL1:ABL1 ratio greater than 0.1% on two consecutive measurements.
“Although several studies of TKI cessation have been reported, little is known about the feasibility of treatment de-escalation in patients with stable molecular responses,” the investigators wrote, adding that the current study shows that de-escalation is “clearly safe for patients in stable MR4 or deeper remission” and could be a “reasonable option.”
This “practice-changing view” is reinforced by the finding of general improvement of adverse events in both cohorts, they said.
Also of note, de-escalation in this study was associated with savings of almost half the expected TKI costs, they added.
The findings imply that many patients with stable responses might be able to maintain responses on lower TKI doses, they concluded, adding that studies of more ambitious de-escalation are warranted.
Newcastle University and Bloodwise funded the study. Dr. Clark received other grants from Bloodwise during the study, as well as grants and personal fees from Novartis, Bristol-Myers Squibb, and Pfizer, and personal fees from Ariad/Incyte.
The interim DESTINY trial findings indicate the feasibility of a dose reduction approach to minimize adverse events associated with TKI treatment and to improve quality of life, Elisabetta Abruzzese, MD, wrote in an editorial.
De-escalation also appears to provide important cost savings, she noted (Lancet Haematol. 2017 Jul;4:e304-5).
“For patients who are unable to stop therapy, adjustment of the treatment doses without jeopardizing the clinical outcome has important clinical implications. Combination of reduced dosage TKIs with newer (ABL001) or older approaches (immunomodulation) might represent a future answer to that,” she said.
Dr. Abruzzese is with Tor Vergata University, Rome. She reported receiving personal fees from Ariad/Incyte, and serving on the advisory board or as a consultant for Novartis, Pfizer, Ariad/Incyte, and Bristol-Myers Squibb.
The interim DESTINY trial findings indicate the feasibility of a dose reduction approach to minimize adverse events associated with TKI treatment and to improve quality of life, Elisabetta Abruzzese, MD, wrote in an editorial.
De-escalation also appears to provide important cost savings, she noted (Lancet Haematol. 2017 Jul;4:e304-5).
“For patients who are unable to stop therapy, adjustment of the treatment doses without jeopardizing the clinical outcome has important clinical implications. Combination of reduced dosage TKIs with newer (ABL001) or older approaches (immunomodulation) might represent a future answer to that,” she said.
Dr. Abruzzese is with Tor Vergata University, Rome. She reported receiving personal fees from Ariad/Incyte, and serving on the advisory board or as a consultant for Novartis, Pfizer, Ariad/Incyte, and Bristol-Myers Squibb.
The interim DESTINY trial findings indicate the feasibility of a dose reduction approach to minimize adverse events associated with TKI treatment and to improve quality of life, Elisabetta Abruzzese, MD, wrote in an editorial.
De-escalation also appears to provide important cost savings, she noted (Lancet Haematol. 2017 Jul;4:e304-5).
“For patients who are unable to stop therapy, adjustment of the treatment doses without jeopardizing the clinical outcome has important clinical implications. Combination of reduced dosage TKIs with newer (ABL001) or older approaches (immunomodulation) might represent a future answer to that,” she said.
Dr. Abruzzese is with Tor Vergata University, Rome. She reported receiving personal fees from Ariad/Incyte, and serving on the advisory board or as a consultant for Novartis, Pfizer, Ariad/Incyte, and Bristol-Myers Squibb.
Tyrosine kinase inhibitor de-escalation is generally safe and may lead to symptom improvement in patients with chronic myeloid leukemia who respond well to the therapy, according to an interim analysis of the nonrandomized phase 2 DESTINY trial.
The findings imply that some patients are unnecessarily overtreated, as responses were sometimes maintained with lower tyrosine kinase inhibitor (TKI) doses, according to Richard E. Clark, MD, of the University of Liverpool, England, and his colleagues (Lancet Haematol. 2017 May 26;4:e310-16).
Of 174 patients with chronic myeloid leukemia (CML) in first chronic phase who had received TKIs for at least 3 years and who were either in stable molecular response 4 logs below the standard arbitrary baseline (MR4, 125 patients) or stable major molecular response (MMR) but not MR4 (49 patients) for at least 12 months at the time of enrollment, 12 (7%) had molecular recurrence during 12 months of half-dose TKI therapy, and all patients who experienced molecular recurrence regained MMR within 4 months of being back on full-dose TKIs, the researchers noted.
The median time to recovery was 77 days.
The recurrence rate was significantly lower in the MR4 cohort than in the MMR cohort (2% vs. 19%; hazard ratio, 0.12), and time to relapse was significantly longer in the MR4 cohort (median, 8.7 vs. 4.4 months), the investigators said.
Recurrence was not associated with age, sex, weight, performance status, BCR-ABL1 transcript type, or duration of TKI therapy, which was a median of 6.9 years overall, they noted.
During the first 3 months of de-escalation, adverse events associated with treatment, including lethargy, diarrhea, rash, and nausea, all improved.
Adverse events during de-escalation occurred in 16 patients, and included one fatality due to worsening of peripheral arterial occlusive disease, but all were deemed unrelated to the TKI or underlying chronic CML, the investigators noted.
Study subjects were adults with positive BCR-ABL1 transcripts, with either e13a2, e14a2, or e19a2 fusion transcript. They were enrolled between December 2013 and April 2015 from 20 hospitals in the United Kingdom; 148 were receiving imatinib, 16 were receiving nilotinib, and 10 were receiving dasatinib. They received half of their standard dose of imatinib (200 mg daily), dasatinib (50 mg daily), or nilotinib (200 mg twice daily) for 12 months. Recurrence during de-escalation was defined as BCR-ABL1:ABL1 ratio greater than 0.1% on two consecutive measurements.
“Although several studies of TKI cessation have been reported, little is known about the feasibility of treatment de-escalation in patients with stable molecular responses,” the investigators wrote, adding that the current study shows that de-escalation is “clearly safe for patients in stable MR4 or deeper remission” and could be a “reasonable option.”
This “practice-changing view” is reinforced by the finding of general improvement of adverse events in both cohorts, they said.
Also of note, de-escalation in this study was associated with savings of almost half the expected TKI costs, they added.
The findings imply that many patients with stable responses might be able to maintain responses on lower TKI doses, they concluded, adding that studies of more ambitious de-escalation are warranted.
Newcastle University and Bloodwise funded the study. Dr. Clark received other grants from Bloodwise during the study, as well as grants and personal fees from Novartis, Bristol-Myers Squibb, and Pfizer, and personal fees from Ariad/Incyte.
Tyrosine kinase inhibitor de-escalation is generally safe and may lead to symptom improvement in patients with chronic myeloid leukemia who respond well to the therapy, according to an interim analysis of the nonrandomized phase 2 DESTINY trial.
The findings imply that some patients are unnecessarily overtreated, as responses were sometimes maintained with lower tyrosine kinase inhibitor (TKI) doses, according to Richard E. Clark, MD, of the University of Liverpool, England, and his colleagues (Lancet Haematol. 2017 May 26;4:e310-16).
Of 174 patients with chronic myeloid leukemia (CML) in first chronic phase who had received TKIs for at least 3 years and who were either in stable molecular response 4 logs below the standard arbitrary baseline (MR4, 125 patients) or stable major molecular response (MMR) but not MR4 (49 patients) for at least 12 months at the time of enrollment, 12 (7%) had molecular recurrence during 12 months of half-dose TKI therapy, and all patients who experienced molecular recurrence regained MMR within 4 months of being back on full-dose TKIs, the researchers noted.
The median time to recovery was 77 days.
The recurrence rate was significantly lower in the MR4 cohort than in the MMR cohort (2% vs. 19%; hazard ratio, 0.12), and time to relapse was significantly longer in the MR4 cohort (median, 8.7 vs. 4.4 months), the investigators said.
Recurrence was not associated with age, sex, weight, performance status, BCR-ABL1 transcript type, or duration of TKI therapy, which was a median of 6.9 years overall, they noted.
During the first 3 months of de-escalation, adverse events associated with treatment, including lethargy, diarrhea, rash, and nausea, all improved.
Adverse events during de-escalation occurred in 16 patients, and included one fatality due to worsening of peripheral arterial occlusive disease, but all were deemed unrelated to the TKI or underlying chronic CML, the investigators noted.
Study subjects were adults with positive BCR-ABL1 transcripts, with either e13a2, e14a2, or e19a2 fusion transcript. They were enrolled between December 2013 and April 2015 from 20 hospitals in the United Kingdom; 148 were receiving imatinib, 16 were receiving nilotinib, and 10 were receiving dasatinib. They received half of their standard dose of imatinib (200 mg daily), dasatinib (50 mg daily), or nilotinib (200 mg twice daily) for 12 months. Recurrence during de-escalation was defined as BCR-ABL1:ABL1 ratio greater than 0.1% on two consecutive measurements.
“Although several studies of TKI cessation have been reported, little is known about the feasibility of treatment de-escalation in patients with stable molecular responses,” the investigators wrote, adding that the current study shows that de-escalation is “clearly safe for patients in stable MR4 or deeper remission” and could be a “reasonable option.”
This “practice-changing view” is reinforced by the finding of general improvement of adverse events in both cohorts, they said.
Also of note, de-escalation in this study was associated with savings of almost half the expected TKI costs, they added.
The findings imply that many patients with stable responses might be able to maintain responses on lower TKI doses, they concluded, adding that studies of more ambitious de-escalation are warranted.
Newcastle University and Bloodwise funded the study. Dr. Clark received other grants from Bloodwise during the study, as well as grants and personal fees from Novartis, Bristol-Myers Squibb, and Pfizer, and personal fees from Ariad/Incyte.
FROM LANCET HAEMATOLOGY
Key clinical point:
Major finding: Only 12 of 174 CML patients had molecular recurrence during 12 months of half-dose TKI therapy, and all regained MMR within 4 months of being back on full-dose TKIs.
Data source: The nonrandomized phase 2 DESTINY study of 174 adults with CML.
Disclosures: Newcastle University and Bloodwise funded the study. Dr. Clark received other grants from Bloodwise during the study, as well as grants and personal fees from Novartis, Bristol-Myers Squibb, and Pfizer, and personal fees from Ariad/Incyte.
De-escalation may improve success of TKI cessation
MADRID—Results of the DESTINY trial suggest that chronic myeloid leukemia (CML) patients may improve their chances of successfully stopping treatment with tyrosine kinase inhibitors (TKIs) by first reducing the dose they receive.
CML patients in deep molecular response (MR4) at study entry had a low rate of recurrence when they first de-escalated their TKI dose—receiving half the standard dose—for a year and then completely stopped receiving TKI treatment for a year.
The 2-year recurrence-free survival (RFS) rate was 77%, which is better than the RFS in any comparable study to date, according to Richard Clark, MD, of the University of Liverpool in Liverpool, UK, and his colleagues.
Dr Clark presented results from DESTINY at the 22nd Congress of the European Hematology Association (EHA) as abstract S423.
DESTINY included 174 CML patients (98 male, 76 female) in stable major molecular response (MMR).
At study entry, patients had received imatinib (n=148), nilotinib (n=16), or dasatinib (n=10) for a median duration of 6.8 years.
For the first 12 months of the study, patients had their TKI dose reduced to half the standard dose. So they received imatinib at 200 mg daily, dasatinib at 50 mg daily, or nilotinib at 200 mg twice daily. After that, patients stopped treatment completely.
After the first 12 months, molecular recurrence was lower in patients with stable MR4 at study entry than in patients who were not in MR4 (but still in MMR)—2.4% (3/125) and 18.4% (9/49), respectively (P<0.001).
During the following 12 months, in which patients had completely stopped TKI treatment, there were 26 recurrences and 4 withdrawals among the remaining 117 patients who were in MR4 at baseline, as well as 20 recurrences and 4 withdrawals among the 36 patients not in MR4.
So the RFS was 77% among patients in MR4 at baseline and 39% among the patients not in MR4 (P<0.001).
The researchers said the probability of RFS was unrelated to patients’ age, gender, performance status, or the prior TKI they received (imatinib vs second-generation TKI).
All patients with recurrence ultimately returned to deep remissions when they resumed their TKI treatment.
“TKI de-escalation is safe for most CML patients with stable and excellent responses to TKI therapy after some years of treatment and is associated with improvement in symptoms,” Dr Clark said.
“Overall, our findings are better than any other studies worldwide and imply that our unique, gradual withdrawal of treatment might be important. We don’t yet understand why our results are so good, but this is a happy problem to have.” ![]()
MADRID—Results of the DESTINY trial suggest that chronic myeloid leukemia (CML) patients may improve their chances of successfully stopping treatment with tyrosine kinase inhibitors (TKIs) by first reducing the dose they receive.
CML patients in deep molecular response (MR4) at study entry had a low rate of recurrence when they first de-escalated their TKI dose—receiving half the standard dose—for a year and then completely stopped receiving TKI treatment for a year.
The 2-year recurrence-free survival (RFS) rate was 77%, which is better than the RFS in any comparable study to date, according to Richard Clark, MD, of the University of Liverpool in Liverpool, UK, and his colleagues.
Dr Clark presented results from DESTINY at the 22nd Congress of the European Hematology Association (EHA) as abstract S423.
DESTINY included 174 CML patients (98 male, 76 female) in stable major molecular response (MMR).
At study entry, patients had received imatinib (n=148), nilotinib (n=16), or dasatinib (n=10) for a median duration of 6.8 years.
For the first 12 months of the study, patients had their TKI dose reduced to half the standard dose. So they received imatinib at 200 mg daily, dasatinib at 50 mg daily, or nilotinib at 200 mg twice daily. After that, patients stopped treatment completely.
After the first 12 months, molecular recurrence was lower in patients with stable MR4 at study entry than in patients who were not in MR4 (but still in MMR)—2.4% (3/125) and 18.4% (9/49), respectively (P<0.001).
During the following 12 months, in which patients had completely stopped TKI treatment, there were 26 recurrences and 4 withdrawals among the remaining 117 patients who were in MR4 at baseline, as well as 20 recurrences and 4 withdrawals among the 36 patients not in MR4.
So the RFS was 77% among patients in MR4 at baseline and 39% among the patients not in MR4 (P<0.001).
The researchers said the probability of RFS was unrelated to patients’ age, gender, performance status, or the prior TKI they received (imatinib vs second-generation TKI).
All patients with recurrence ultimately returned to deep remissions when they resumed their TKI treatment.
“TKI de-escalation is safe for most CML patients with stable and excellent responses to TKI therapy after some years of treatment and is associated with improvement in symptoms,” Dr Clark said.
“Overall, our findings are better than any other studies worldwide and imply that our unique, gradual withdrawal of treatment might be important. We don’t yet understand why our results are so good, but this is a happy problem to have.” ![]()
MADRID—Results of the DESTINY trial suggest that chronic myeloid leukemia (CML) patients may improve their chances of successfully stopping treatment with tyrosine kinase inhibitors (TKIs) by first reducing the dose they receive.
CML patients in deep molecular response (MR4) at study entry had a low rate of recurrence when they first de-escalated their TKI dose—receiving half the standard dose—for a year and then completely stopped receiving TKI treatment for a year.
The 2-year recurrence-free survival (RFS) rate was 77%, which is better than the RFS in any comparable study to date, according to Richard Clark, MD, of the University of Liverpool in Liverpool, UK, and his colleagues.
Dr Clark presented results from DESTINY at the 22nd Congress of the European Hematology Association (EHA) as abstract S423.
DESTINY included 174 CML patients (98 male, 76 female) in stable major molecular response (MMR).
At study entry, patients had received imatinib (n=148), nilotinib (n=16), or dasatinib (n=10) for a median duration of 6.8 years.
For the first 12 months of the study, patients had their TKI dose reduced to half the standard dose. So they received imatinib at 200 mg daily, dasatinib at 50 mg daily, or nilotinib at 200 mg twice daily. After that, patients stopped treatment completely.
After the first 12 months, molecular recurrence was lower in patients with stable MR4 at study entry than in patients who were not in MR4 (but still in MMR)—2.4% (3/125) and 18.4% (9/49), respectively (P<0.001).
During the following 12 months, in which patients had completely stopped TKI treatment, there were 26 recurrences and 4 withdrawals among the remaining 117 patients who were in MR4 at baseline, as well as 20 recurrences and 4 withdrawals among the 36 patients not in MR4.
So the RFS was 77% among patients in MR4 at baseline and 39% among the patients not in MR4 (P<0.001).
The researchers said the probability of RFS was unrelated to patients’ age, gender, performance status, or the prior TKI they received (imatinib vs second-generation TKI).
All patients with recurrence ultimately returned to deep remissions when they resumed their TKI treatment.
“TKI de-escalation is safe for most CML patients with stable and excellent responses to TKI therapy after some years of treatment and is associated with improvement in symptoms,” Dr Clark said.
“Overall, our findings are better than any other studies worldwide and imply that our unique, gradual withdrawal of treatment might be important. We don’t yet understand why our results are so good, but this is a happy problem to have.” ![]()
FDA grants priority review to sNDA for dasatinib
The US Food and Drug Administration (FDA) has accepted for priority review a supplemental new drug application (sNDA) for dasatinib (Sprycel).
Bristol Myers Squibb is seeking approval for dasatinib as a treatment for children with Philadelphia chromosome-positive (Ph+) chronic phase (CP) chronic myeloid leukemia (CML), as well as approval for a powder formulation of dasatinib for oral suspension.
The FDA grants priority review to applications for products that may provide significant improvements in the treatment, diagnosis, or prevention of serious conditions.
The agency’s goal is to take action on a priority review application within 6 months of receiving it, rather than the standard 10 months.
The FDA plans to make a decision on the dasatinib sNDA by November 9, 2017.
The sNDA includes data from CA180-226 (NCT00777036), an ongoing, phase 2 trial of dasatinib in pediatric patients with CP-CML who are resistant to or cannot tolerate imatinib and pediatric patients newly diagnosed with CP-CML.
The trial enrolled patients aged 18 and younger with newly diagnosed CML or Ph+ leukemias resistant to or intolerant of imatinib.
Cohort 1 included 29 CP-CML patients resistant to or intolerant of imatinib. Cohort 2 included patients with accelerated/blast phase CML or Ph+ acute lymphoblastic leukemia. Cohort 3 included 84 patients with newly diagnosed CP-CML.
Data from Cohorts 1 and 3 were recently presented at the 2017 ASCO Annual Meeting.
Three months into treatment with dasatinib, patients with CP-CML who were resistant to or intolerant of imatinib (Cohort 1) had a cumulative major cytogenetic response rate of 55.2%. This response rate increased over time to exceed 90% at 24 months.
Newly diagnosed patients with CP-CML (Cohort 3) received dasatinib orally or as powder for oral suspension once daily. They achieved a cumulative complete cytogenetic response rate of 64% as early as 6 months into treatment. This response rate increased to 94% at 24 months.
The median duration of response was not estimable or not yet reached in each cohort at the time of follow-up.
The estimated progression-free survival at 48 months was greater than 75% for patients in Cohort 1 and greater than 90% for patients in Cohort 3.
The safety profile of dasatinib in this study was deemed comparable to that reported in adults with CP-CML. In this study, there were no reported events of pleural/pericardial effusion, pulmonary edema/hypertension, or pulmonary arterial hypertension related to dasatinib.
Dasatinib first received FDA approval in 2006. The drug is currently approved to treat adults with:
- Newly diagnosed Ph+ CP-CML
- Chronic, accelerated, or blast phase Ph+ CML with resistance or intolerance to prior therapy including imatinib
- Ph+ acute lymphoblastic leukemia with resistance or intolerance to prior therapy.

The US Food and Drug Administration (FDA) has accepted for priority review a supplemental new drug application (sNDA) for dasatinib (Sprycel).
Bristol Myers Squibb is seeking approval for dasatinib as a treatment for children with Philadelphia chromosome-positive (Ph+) chronic phase (CP) chronic myeloid leukemia (CML), as well as approval for a powder formulation of dasatinib for oral suspension.
The FDA grants priority review to applications for products that may provide significant improvements in the treatment, diagnosis, or prevention of serious conditions.
The agency’s goal is to take action on a priority review application within 6 months of receiving it, rather than the standard 10 months.
The FDA plans to make a decision on the dasatinib sNDA by November 9, 2017.
The sNDA includes data from CA180-226 (NCT00777036), an ongoing, phase 2 trial of dasatinib in pediatric patients with CP-CML who are resistant to or cannot tolerate imatinib and pediatric patients newly diagnosed with CP-CML.
The trial enrolled patients aged 18 and younger with newly diagnosed CML or Ph+ leukemias resistant to or intolerant of imatinib.
Cohort 1 included 29 CP-CML patients resistant to or intolerant of imatinib. Cohort 2 included patients with accelerated/blast phase CML or Ph+ acute lymphoblastic leukemia. Cohort 3 included 84 patients with newly diagnosed CP-CML.
Data from Cohorts 1 and 3 were recently presented at the 2017 ASCO Annual Meeting.
Three months into treatment with dasatinib, patients with CP-CML who were resistant to or intolerant of imatinib (Cohort 1) had a cumulative major cytogenetic response rate of 55.2%. This response rate increased over time to exceed 90% at 24 months.
Newly diagnosed patients with CP-CML (Cohort 3) received dasatinib orally or as powder for oral suspension once daily. They achieved a cumulative complete cytogenetic response rate of 64% as early as 6 months into treatment. This response rate increased to 94% at 24 months.
The median duration of response was not estimable or not yet reached in each cohort at the time of follow-up.
The estimated progression-free survival at 48 months was greater than 75% for patients in Cohort 1 and greater than 90% for patients in Cohort 3.
The safety profile of dasatinib in this study was deemed comparable to that reported in adults with CP-CML. In this study, there were no reported events of pleural/pericardial effusion, pulmonary edema/hypertension, or pulmonary arterial hypertension related to dasatinib.
Dasatinib first received FDA approval in 2006. The drug is currently approved to treat adults with:
- Newly diagnosed Ph+ CP-CML
- Chronic, accelerated, or blast phase Ph+ CML with resistance or intolerance to prior therapy including imatinib
- Ph+ acute lymphoblastic leukemia with resistance or intolerance to prior therapy.

The US Food and Drug Administration (FDA) has accepted for priority review a supplemental new drug application (sNDA) for dasatinib (Sprycel).
Bristol Myers Squibb is seeking approval for dasatinib as a treatment for children with Philadelphia chromosome-positive (Ph+) chronic phase (CP) chronic myeloid leukemia (CML), as well as approval for a powder formulation of dasatinib for oral suspension.
The FDA grants priority review to applications for products that may provide significant improvements in the treatment, diagnosis, or prevention of serious conditions.
The agency’s goal is to take action on a priority review application within 6 months of receiving it, rather than the standard 10 months.
The FDA plans to make a decision on the dasatinib sNDA by November 9, 2017.
The sNDA includes data from CA180-226 (NCT00777036), an ongoing, phase 2 trial of dasatinib in pediatric patients with CP-CML who are resistant to or cannot tolerate imatinib and pediatric patients newly diagnosed with CP-CML.
The trial enrolled patients aged 18 and younger with newly diagnosed CML or Ph+ leukemias resistant to or intolerant of imatinib.
Cohort 1 included 29 CP-CML patients resistant to or intolerant of imatinib. Cohort 2 included patients with accelerated/blast phase CML or Ph+ acute lymphoblastic leukemia. Cohort 3 included 84 patients with newly diagnosed CP-CML.
Data from Cohorts 1 and 3 were recently presented at the 2017 ASCO Annual Meeting.
Three months into treatment with dasatinib, patients with CP-CML who were resistant to or intolerant of imatinib (Cohort 1) had a cumulative major cytogenetic response rate of 55.2%. This response rate increased over time to exceed 90% at 24 months.
Newly diagnosed patients with CP-CML (Cohort 3) received dasatinib orally or as powder for oral suspension once daily. They achieved a cumulative complete cytogenetic response rate of 64% as early as 6 months into treatment. This response rate increased to 94% at 24 months.
The median duration of response was not estimable or not yet reached in each cohort at the time of follow-up.
The estimated progression-free survival at 48 months was greater than 75% for patients in Cohort 1 and greater than 90% for patients in Cohort 3.
The safety profile of dasatinib in this study was deemed comparable to that reported in adults with CP-CML. In this study, there were no reported events of pleural/pericardial effusion, pulmonary edema/hypertension, or pulmonary arterial hypertension related to dasatinib.
Dasatinib first received FDA approval in 2006. The drug is currently approved to treat adults with:
- Newly diagnosed Ph+ CP-CML
- Chronic, accelerated, or blast phase Ph+ CML with resistance or intolerance to prior therapy including imatinib
- Ph+ acute lymphoblastic leukemia with resistance or intolerance to prior therapy.

EU label for nilotinib now includes info on stopping therapy
The European Commission has approved the inclusion of treatment-free remission (TFR) data in the European Union (EU) product information for nilotinib (Tasigna®).
TFR is the ability to maintain molecular response after stopping tyrosine kinase inhibitor (TKI) therapy in patients with Ph+ chronic myeloid leukemia in chronic phase (CML-CP).
Nilotinib is approved for the treatment of CML-CP in adults resistant or intolerant to at least one prior therapy, including imatinib, and for the treatment of adults with newly diagnosed Ph+ CML.
Nilotinib is the first and only TKI to include TFR data in its product information in the EU, according to Novartis, developer of the drug.
Two large international Ph+ CML trials assessing nilotinib discontinuation—ENESTfreedom and ENESTop—formed the basis of the decision.
The two open-label trials showed that half of Ph+ CML-CP patients who met rigorous predefined response criteria maintained TFR after stopping nilotinib, both in the frontline setting and after switching from imatinib.
Both trials included regular and frequent molecular monitoring of BCR-ABL levels with an assay that could measure transcript levels down to a molecular response (MR) of 4.5. Frequent monitoring after discontinuation helped determine loss of MR 4.0 and major molecular response, indicating the need to re-initiate treatment.
This phase 2 trial (Evaluating Nilotinib Efficacy and Safety in Clinical Trials - Following REsponsE in De nOvo CML-CP Patients) involved 215 patients with Ph+ CML-CP treated at 132 sites across 19 countries.
The trial evaluated whether treatment could be stopped in 190 of the patients after they had achieved a response of MR 4.5 and a sustained deep molecular response for 1 year as first-line treatment.
The results showed that 51.6% of the patients in the trial could discontinue therapy and remain in TFR at the 48-week analysis.
However, ENESTfreedom did not meet its primary endpoint of 50% or more of patients in MMR at 48 weeks in the TFR phase. The median treatment duration was 3.6 years.
Adverse events in the trial included musculoskeletal pain (24.7%) during the first years of the TFR phase compared with 16.3% in those still on nilotinib.
No patient progressed to advanced phase/blast crisis.
This phase 2 trial (Evaluating Nilotinib Efficacy and Safety Trial) involved 163 patients with Ph+ CML-CP treated at 63 sites across 18 countries. Investigators evaluated 126 patients who had been treated with imatinib and then switched to nilotinib and had achieved a sustained deep molecular response for 1 year while on nilotinib.
The trial demonstrated that 57.9% of patients maintained a molecular resonse at 48 weeks after stopping treatment. The trial met its primary endpoint—the proportion of patients who did not lose MR 4.0 or MMR within 48 weeks of discontinuing nilotinib in the TFR phase.
Again, the rates of musuloskeletal pain were higher in the first year of TFR than in patients still on nilotinib in the consolidation phase, 42.1% and 14.3%, respectively.
Results of the EURO-SKI trial also support the idea that certain CML patients can safely stop TKI therapy.
For full prescribing information, see the product insert. ![]()
The European Commission has approved the inclusion of treatment-free remission (TFR) data in the European Union (EU) product information for nilotinib (Tasigna®).
TFR is the ability to maintain molecular response after stopping tyrosine kinase inhibitor (TKI) therapy in patients with Ph+ chronic myeloid leukemia in chronic phase (CML-CP).
Nilotinib is approved for the treatment of CML-CP in adults resistant or intolerant to at least one prior therapy, including imatinib, and for the treatment of adults with newly diagnosed Ph+ CML.
Nilotinib is the first and only TKI to include TFR data in its product information in the EU, according to Novartis, developer of the drug.
Two large international Ph+ CML trials assessing nilotinib discontinuation—ENESTfreedom and ENESTop—formed the basis of the decision.
The two open-label trials showed that half of Ph+ CML-CP patients who met rigorous predefined response criteria maintained TFR after stopping nilotinib, both in the frontline setting and after switching from imatinib.
Both trials included regular and frequent molecular monitoring of BCR-ABL levels with an assay that could measure transcript levels down to a molecular response (MR) of 4.5. Frequent monitoring after discontinuation helped determine loss of MR 4.0 and major molecular response, indicating the need to re-initiate treatment.
This phase 2 trial (Evaluating Nilotinib Efficacy and Safety in Clinical Trials - Following REsponsE in De nOvo CML-CP Patients) involved 215 patients with Ph+ CML-CP treated at 132 sites across 19 countries.
The trial evaluated whether treatment could be stopped in 190 of the patients after they had achieved a response of MR 4.5 and a sustained deep molecular response for 1 year as first-line treatment.
The results showed that 51.6% of the patients in the trial could discontinue therapy and remain in TFR at the 48-week analysis.
However, ENESTfreedom did not meet its primary endpoint of 50% or more of patients in MMR at 48 weeks in the TFR phase. The median treatment duration was 3.6 years.
Adverse events in the trial included musculoskeletal pain (24.7%) during the first years of the TFR phase compared with 16.3% in those still on nilotinib.
No patient progressed to advanced phase/blast crisis.
This phase 2 trial (Evaluating Nilotinib Efficacy and Safety Trial) involved 163 patients with Ph+ CML-CP treated at 63 sites across 18 countries. Investigators evaluated 126 patients who had been treated with imatinib and then switched to nilotinib and had achieved a sustained deep molecular response for 1 year while on nilotinib.
The trial demonstrated that 57.9% of patients maintained a molecular resonse at 48 weeks after stopping treatment. The trial met its primary endpoint—the proportion of patients who did not lose MR 4.0 or MMR within 48 weeks of discontinuing nilotinib in the TFR phase.
Again, the rates of musuloskeletal pain were higher in the first year of TFR than in patients still on nilotinib in the consolidation phase, 42.1% and 14.3%, respectively.
Results of the EURO-SKI trial also support the idea that certain CML patients can safely stop TKI therapy.
For full prescribing information, see the product insert. ![]()
The European Commission has approved the inclusion of treatment-free remission (TFR) data in the European Union (EU) product information for nilotinib (Tasigna®).
TFR is the ability to maintain molecular response after stopping tyrosine kinase inhibitor (TKI) therapy in patients with Ph+ chronic myeloid leukemia in chronic phase (CML-CP).
Nilotinib is approved for the treatment of CML-CP in adults resistant or intolerant to at least one prior therapy, including imatinib, and for the treatment of adults with newly diagnosed Ph+ CML.
Nilotinib is the first and only TKI to include TFR data in its product information in the EU, according to Novartis, developer of the drug.
Two large international Ph+ CML trials assessing nilotinib discontinuation—ENESTfreedom and ENESTop—formed the basis of the decision.
The two open-label trials showed that half of Ph+ CML-CP patients who met rigorous predefined response criteria maintained TFR after stopping nilotinib, both in the frontline setting and after switching from imatinib.
Both trials included regular and frequent molecular monitoring of BCR-ABL levels with an assay that could measure transcript levels down to a molecular response (MR) of 4.5. Frequent monitoring after discontinuation helped determine loss of MR 4.0 and major molecular response, indicating the need to re-initiate treatment.
This phase 2 trial (Evaluating Nilotinib Efficacy and Safety in Clinical Trials - Following REsponsE in De nOvo CML-CP Patients) involved 215 patients with Ph+ CML-CP treated at 132 sites across 19 countries.
The trial evaluated whether treatment could be stopped in 190 of the patients after they had achieved a response of MR 4.5 and a sustained deep molecular response for 1 year as first-line treatment.
The results showed that 51.6% of the patients in the trial could discontinue therapy and remain in TFR at the 48-week analysis.
However, ENESTfreedom did not meet its primary endpoint of 50% or more of patients in MMR at 48 weeks in the TFR phase. The median treatment duration was 3.6 years.
Adverse events in the trial included musculoskeletal pain (24.7%) during the first years of the TFR phase compared with 16.3% in those still on nilotinib.
No patient progressed to advanced phase/blast crisis.
This phase 2 trial (Evaluating Nilotinib Efficacy and Safety Trial) involved 163 patients with Ph+ CML-CP treated at 63 sites across 18 countries. Investigators evaluated 126 patients who had been treated with imatinib and then switched to nilotinib and had achieved a sustained deep molecular response for 1 year while on nilotinib.
The trial demonstrated that 57.9% of patients maintained a molecular resonse at 48 weeks after stopping treatment. The trial met its primary endpoint—the proportion of patients who did not lose MR 4.0 or MMR within 48 weeks of discontinuing nilotinib in the TFR phase.
Again, the rates of musuloskeletal pain were higher in the first year of TFR than in patients still on nilotinib in the consolidation phase, 42.1% and 14.3%, respectively.
Results of the EURO-SKI trial also support the idea that certain CML patients can safely stop TKI therapy.
For full prescribing information, see the product insert. ![]()
Dasatinib potentially a new SOC for children with CML-CP
CHICAGO—The largest ongoing and prospective trial of pediatric patients with chronic myeloid leukemia in chronic phase (CML-CP), according to the best knowledge of the investigators, has found dasatinib to be safe and effective as first- or second-line therapy for these children.
Patients refractory to or intolerant of imatinib had a major cytogenetic response (MCyR) by 3 months and responses at 12 and 24 months exceeded 90%.
Newly diagnosed patients had a complete cytogenetic response (CCyR) by 6 months.
“We believe our data suggests that dasatinib could be considered as a new standard of care (SOC) for children with CML in chronic phase,” said study author Lia Gore, MD, of the University of Colorado School of Medicine/Children’s Hospital Colorado in Aurora.
She presented the findings of the study at the ASCO 2017 Annual Meeting (abstract 10511).
Study design
CA 180-226 is a phase 2, open-label, nonrandomized, prospective study conducted in 18 countries. Patients younger than 18 years with newly diagnosed CML-CP, or imatinib-resistant/intolerant (R/I) CML-CP, or CML in accelerated phase, or Ph+ acute lymphoblastic leukemia (ALL) were enrolled on the study between March 2009 and September 2014.
Dr Gore’s presentation focused on the CML-CP patients in the study, both the newly diagnosed and the imatinib-R/I patients.
The imatinib-R/I patients received dasatinib 60 mg/m2 tablets once daily, and the newly diagnosed patients received the same tablet dosage daily or a powdered formulation for oral suspension (PFOS) of dasatinib at 72 mg/m2 daily.
Dr Gore noted the different dosage in the oral suspension formulation is based on bioavailability studies performed in adults, which was determined to be equivalent to the 60 mg/m2 tablet formulation.
Once accrual was reached in the tablet cohort, newly diagnosed patients were accrued to the PFOS cohort. The patients on PFOS could switch to tablets after a year or more on the oral suspension.
Patients remained on treatment until disease progression, unacceptable toxicity occurred, or the patient/physician preference.
All patients had a minimum follow-up of 2 years. The longest follow-up was more than 90 months.
Primary objectives of the study were MCyR greater than 30% for imatinib-R/I patients and complete CCyR greater than 55% for newly diagnosed patients.
Secondary objectives included time to and duration of response, major molecular response (MMR), progression-free survival (PFS), overall survival (OS), and safety.
Baseline patient characteristics
One hundred thirteen patients were treated across the 3 cohorts—29 in the imatinib-R/I receiving tablets, 51 newly diagnosed patients receiving tablets, and 33 newly diagnosed patients in the PFOS arm. A total of 84 patients had newly diagnosed disease.
Of the 29 imatinib-R/I patients, 25 were resistant, 2 intolerant, and 2 undetermined. And 6 of the 25 resistant patients had defined imatinib-resistance mutations.
Median age was 13.8 years in the imatinib-R/I cohort, 12.9 years in the newly diagnosed on tablets, and 11.7 years in the PFOS group. Other baseline characteristics were similar among the cohorts.
“Importantly, there were 3 patients less than 2 years of age, and a substantial proportion of patients were actually less than 12 years of age,” Dr Gore pointed out.
Dasatinib exposure
The median duration of therapy was 50 months and 42 months in the R/I and newly diagnosed cohorts, respectively.
Forty-eight percent of the imatinib-R/I patients and 73% of the newly diagnosed patients are still on treatment. A relatively small number of patients discontinued therapy.
The median duration of therapy was shorter in the PFOS cohort because they were enrolled only after accrual to the tablet cohort. However, they were also followed up for more than 2 years.
Of the 33 patients on PFOS, 22 eventually switched to tablet formulation.
Results
The primary endpoint for imatinib-R/I patients—MCyR greater than 30%—was reached by 3 months, and MCyR at 12 and 24 months exceeded 90%. The median time to response was 3.1 months (range, 2.8 – 4.1), and median duration of response was not yet reached (range, 54.9 – not estimable).
For newly diagnosed patients, the preset defined rate of interest of 55% for CCyR was reached as early as 6 months, and exceeded 90% by 12 and 24 months.
Dr Gore pointed out that intolerant patients also reached CCyR relatively quickly, although it was not a specified endpoint.
Data indicate that responses occurred relatively quickly and continued to increase over time of follow-up.
MMR also continued to increase over time and showed no difference between formulation and response rate.
Median PFS has not been reached, as only 7 patients in each cohort had disease progression.
One imatinib-R/I patient died 1 year after stopping treatment. The patient, who had a GI bleed unrelated to dasatinib, had discontinued therapy for progressive disease with loss of MCyR.
Safety
Overall safety was very similar to the dasatinib exposure and experience in adults, Dr Gore said, and there were no differences in events between PFOS and tablets.
One patient in the PFOS cohort had a dasatinib-related grade 3 hypersensitivity reaction, which resolved after discontinuation of dasatinib.
“What’s important here,” she said, “is that there were almost no adverse events of severity in either cohort, only 1 in the imatinib refractory and intolerant and 1 in the newly diagnosed cohorts.”
“Most importantly for those of us with a lot of experience in this field,” she added, “there were no occurrences of pleural effusion, pericardial effusion, pulmonary edema, pulmonary hypertension, or any vascular occlusive events in patients noted on this trial.”
“Additionally, for pediatricians, we care a lot about what happens to growth in these patients and prospectively we collected a lot of data related to growth parameters in bone growth and development."
Of the dasatinib-related adverse events occurring in 10% or more of patients, there were only 5 growth and development events noted out of the 113 patients treated and all were grade 1 or 2 events, Dr Gore pointed out.
In the R/I cohort, one patient had osteopenia and gynecomastia. At the time of data analysis, this event had resolved even though the patient continued on dasatinib.
“We believe our data suggests that dasatinib could be considered as a new standard of care for children with CML in chronic phase,” she said.
“It includes the advantage of a liquid formulation as well as the advantages of once daily dosing and administration without regard to fed or fasting state,” she added, “which for all of us who treat children know could be quite important.”
The study was funded by Bristol-Myers Squibb. ![]()
CHICAGO—The largest ongoing and prospective trial of pediatric patients with chronic myeloid leukemia in chronic phase (CML-CP), according to the best knowledge of the investigators, has found dasatinib to be safe and effective as first- or second-line therapy for these children.
Patients refractory to or intolerant of imatinib had a major cytogenetic response (MCyR) by 3 months and responses at 12 and 24 months exceeded 90%.
Newly diagnosed patients had a complete cytogenetic response (CCyR) by 6 months.
“We believe our data suggests that dasatinib could be considered as a new standard of care (SOC) for children with CML in chronic phase,” said study author Lia Gore, MD, of the University of Colorado School of Medicine/Children’s Hospital Colorado in Aurora.
She presented the findings of the study at the ASCO 2017 Annual Meeting (abstract 10511).
Study design
CA 180-226 is a phase 2, open-label, nonrandomized, prospective study conducted in 18 countries. Patients younger than 18 years with newly diagnosed CML-CP, or imatinib-resistant/intolerant (R/I) CML-CP, or CML in accelerated phase, or Ph+ acute lymphoblastic leukemia (ALL) were enrolled on the study between March 2009 and September 2014.
Dr Gore’s presentation focused on the CML-CP patients in the study, both the newly diagnosed and the imatinib-R/I patients.
The imatinib-R/I patients received dasatinib 60 mg/m2 tablets once daily, and the newly diagnosed patients received the same tablet dosage daily or a powdered formulation for oral suspension (PFOS) of dasatinib at 72 mg/m2 daily.
Dr Gore noted the different dosage in the oral suspension formulation is based on bioavailability studies performed in adults, which was determined to be equivalent to the 60 mg/m2 tablet formulation.
Once accrual was reached in the tablet cohort, newly diagnosed patients were accrued to the PFOS cohort. The patients on PFOS could switch to tablets after a year or more on the oral suspension.
Patients remained on treatment until disease progression, unacceptable toxicity occurred, or the patient/physician preference.
All patients had a minimum follow-up of 2 years. The longest follow-up was more than 90 months.
Primary objectives of the study were MCyR greater than 30% for imatinib-R/I patients and complete CCyR greater than 55% for newly diagnosed patients.
Secondary objectives included time to and duration of response, major molecular response (MMR), progression-free survival (PFS), overall survival (OS), and safety.
Baseline patient characteristics
One hundred thirteen patients were treated across the 3 cohorts—29 in the imatinib-R/I receiving tablets, 51 newly diagnosed patients receiving tablets, and 33 newly diagnosed patients in the PFOS arm. A total of 84 patients had newly diagnosed disease.
Of the 29 imatinib-R/I patients, 25 were resistant, 2 intolerant, and 2 undetermined. And 6 of the 25 resistant patients had defined imatinib-resistance mutations.
Median age was 13.8 years in the imatinib-R/I cohort, 12.9 years in the newly diagnosed on tablets, and 11.7 years in the PFOS group. Other baseline characteristics were similar among the cohorts.
“Importantly, there were 3 patients less than 2 years of age, and a substantial proportion of patients were actually less than 12 years of age,” Dr Gore pointed out.
Dasatinib exposure
The median duration of therapy was 50 months and 42 months in the R/I and newly diagnosed cohorts, respectively.
Forty-eight percent of the imatinib-R/I patients and 73% of the newly diagnosed patients are still on treatment. A relatively small number of patients discontinued therapy.
The median duration of therapy was shorter in the PFOS cohort because they were enrolled only after accrual to the tablet cohort. However, they were also followed up for more than 2 years.
Of the 33 patients on PFOS, 22 eventually switched to tablet formulation.
Results
The primary endpoint for imatinib-R/I patients—MCyR greater than 30%—was reached by 3 months, and MCyR at 12 and 24 months exceeded 90%. The median time to response was 3.1 months (range, 2.8 – 4.1), and median duration of response was not yet reached (range, 54.9 – not estimable).
For newly diagnosed patients, the preset defined rate of interest of 55% for CCyR was reached as early as 6 months, and exceeded 90% by 12 and 24 months.
Dr Gore pointed out that intolerant patients also reached CCyR relatively quickly, although it was not a specified endpoint.
Data indicate that responses occurred relatively quickly and continued to increase over time of follow-up.
MMR also continued to increase over time and showed no difference between formulation and response rate.
Median PFS has not been reached, as only 7 patients in each cohort had disease progression.
One imatinib-R/I patient died 1 year after stopping treatment. The patient, who had a GI bleed unrelated to dasatinib, had discontinued therapy for progressive disease with loss of MCyR.
Safety
Overall safety was very similar to the dasatinib exposure and experience in adults, Dr Gore said, and there were no differences in events between PFOS and tablets.
One patient in the PFOS cohort had a dasatinib-related grade 3 hypersensitivity reaction, which resolved after discontinuation of dasatinib.
“What’s important here,” she said, “is that there were almost no adverse events of severity in either cohort, only 1 in the imatinib refractory and intolerant and 1 in the newly diagnosed cohorts.”
“Most importantly for those of us with a lot of experience in this field,” she added, “there were no occurrences of pleural effusion, pericardial effusion, pulmonary edema, pulmonary hypertension, or any vascular occlusive events in patients noted on this trial.”
“Additionally, for pediatricians, we care a lot about what happens to growth in these patients and prospectively we collected a lot of data related to growth parameters in bone growth and development."
Of the dasatinib-related adverse events occurring in 10% or more of patients, there were only 5 growth and development events noted out of the 113 patients treated and all were grade 1 or 2 events, Dr Gore pointed out.
In the R/I cohort, one patient had osteopenia and gynecomastia. At the time of data analysis, this event had resolved even though the patient continued on dasatinib.
“We believe our data suggests that dasatinib could be considered as a new standard of care for children with CML in chronic phase,” she said.
“It includes the advantage of a liquid formulation as well as the advantages of once daily dosing and administration without regard to fed or fasting state,” she added, “which for all of us who treat children know could be quite important.”
The study was funded by Bristol-Myers Squibb. ![]()
CHICAGO—The largest ongoing and prospective trial of pediatric patients with chronic myeloid leukemia in chronic phase (CML-CP), according to the best knowledge of the investigators, has found dasatinib to be safe and effective as first- or second-line therapy for these children.
Patients refractory to or intolerant of imatinib had a major cytogenetic response (MCyR) by 3 months and responses at 12 and 24 months exceeded 90%.
Newly diagnosed patients had a complete cytogenetic response (CCyR) by 6 months.
“We believe our data suggests that dasatinib could be considered as a new standard of care (SOC) for children with CML in chronic phase,” said study author Lia Gore, MD, of the University of Colorado School of Medicine/Children’s Hospital Colorado in Aurora.
She presented the findings of the study at the ASCO 2017 Annual Meeting (abstract 10511).
Study design
CA 180-226 is a phase 2, open-label, nonrandomized, prospective study conducted in 18 countries. Patients younger than 18 years with newly diagnosed CML-CP, or imatinib-resistant/intolerant (R/I) CML-CP, or CML in accelerated phase, or Ph+ acute lymphoblastic leukemia (ALL) were enrolled on the study between March 2009 and September 2014.
Dr Gore’s presentation focused on the CML-CP patients in the study, both the newly diagnosed and the imatinib-R/I patients.
The imatinib-R/I patients received dasatinib 60 mg/m2 tablets once daily, and the newly diagnosed patients received the same tablet dosage daily or a powdered formulation for oral suspension (PFOS) of dasatinib at 72 mg/m2 daily.
Dr Gore noted the different dosage in the oral suspension formulation is based on bioavailability studies performed in adults, which was determined to be equivalent to the 60 mg/m2 tablet formulation.
Once accrual was reached in the tablet cohort, newly diagnosed patients were accrued to the PFOS cohort. The patients on PFOS could switch to tablets after a year or more on the oral suspension.
Patients remained on treatment until disease progression, unacceptable toxicity occurred, or the patient/physician preference.
All patients had a minimum follow-up of 2 years. The longest follow-up was more than 90 months.
Primary objectives of the study were MCyR greater than 30% for imatinib-R/I patients and complete CCyR greater than 55% for newly diagnosed patients.
Secondary objectives included time to and duration of response, major molecular response (MMR), progression-free survival (PFS), overall survival (OS), and safety.
Baseline patient characteristics
One hundred thirteen patients were treated across the 3 cohorts—29 in the imatinib-R/I receiving tablets, 51 newly diagnosed patients receiving tablets, and 33 newly diagnosed patients in the PFOS arm. A total of 84 patients had newly diagnosed disease.
Of the 29 imatinib-R/I patients, 25 were resistant, 2 intolerant, and 2 undetermined. And 6 of the 25 resistant patients had defined imatinib-resistance mutations.
Median age was 13.8 years in the imatinib-R/I cohort, 12.9 years in the newly diagnosed on tablets, and 11.7 years in the PFOS group. Other baseline characteristics were similar among the cohorts.
“Importantly, there were 3 patients less than 2 years of age, and a substantial proportion of patients were actually less than 12 years of age,” Dr Gore pointed out.
Dasatinib exposure
The median duration of therapy was 50 months and 42 months in the R/I and newly diagnosed cohorts, respectively.
Forty-eight percent of the imatinib-R/I patients and 73% of the newly diagnosed patients are still on treatment. A relatively small number of patients discontinued therapy.
The median duration of therapy was shorter in the PFOS cohort because they were enrolled only after accrual to the tablet cohort. However, they were also followed up for more than 2 years.
Of the 33 patients on PFOS, 22 eventually switched to tablet formulation.
Results
The primary endpoint for imatinib-R/I patients—MCyR greater than 30%—was reached by 3 months, and MCyR at 12 and 24 months exceeded 90%. The median time to response was 3.1 months (range, 2.8 – 4.1), and median duration of response was not yet reached (range, 54.9 – not estimable).
For newly diagnosed patients, the preset defined rate of interest of 55% for CCyR was reached as early as 6 months, and exceeded 90% by 12 and 24 months.
Dr Gore pointed out that intolerant patients also reached CCyR relatively quickly, although it was not a specified endpoint.
Data indicate that responses occurred relatively quickly and continued to increase over time of follow-up.
MMR also continued to increase over time and showed no difference between formulation and response rate.
Median PFS has not been reached, as only 7 patients in each cohort had disease progression.
One imatinib-R/I patient died 1 year after stopping treatment. The patient, who had a GI bleed unrelated to dasatinib, had discontinued therapy for progressive disease with loss of MCyR.
Safety
Overall safety was very similar to the dasatinib exposure and experience in adults, Dr Gore said, and there were no differences in events between PFOS and tablets.
One patient in the PFOS cohort had a dasatinib-related grade 3 hypersensitivity reaction, which resolved after discontinuation of dasatinib.
“What’s important here,” she said, “is that there were almost no adverse events of severity in either cohort, only 1 in the imatinib refractory and intolerant and 1 in the newly diagnosed cohorts.”
“Most importantly for those of us with a lot of experience in this field,” she added, “there were no occurrences of pleural effusion, pericardial effusion, pulmonary edema, pulmonary hypertension, or any vascular occlusive events in patients noted on this trial.”
“Additionally, for pediatricians, we care a lot about what happens to growth in these patients and prospectively we collected a lot of data related to growth parameters in bone growth and development."
Of the dasatinib-related adverse events occurring in 10% or more of patients, there were only 5 growth and development events noted out of the 113 patients treated and all were grade 1 or 2 events, Dr Gore pointed out.
In the R/I cohort, one patient had osteopenia and gynecomastia. At the time of data analysis, this event had resolved even though the patient continued on dasatinib.
“We believe our data suggests that dasatinib could be considered as a new standard of care for children with CML in chronic phase,” she said.
“It includes the advantage of a liquid formulation as well as the advantages of once daily dosing and administration without regard to fed or fasting state,” she added, “which for all of us who treat children know could be quite important.”
The study was funded by Bristol-Myers Squibb.
Authority on hematologic malignancies dies
Physician, researcher, and educator H. Jean Khoury, MD, recently passed away.
He died on Monday, May 22, at the age of 50, after a year-long battle with esophageal cancer.
Dr Khoury led the division of hematology at Winship Cancer Institute of Emory University in Atlanta, Georgia.
He was considered an authority on hematologic malignancies, particularly chronic myeloid leukemia (CML), acute leukemia, and myelodysplastic syndromes (MDS).
Dr Khoury joined Winship Cancer Institute in 2004 as director of the Leukemia Service and associate professor in the Emory School of Medicine.
In 2009, he was promoted to professor and director of the Division of Hematology in the Department of Hematology and Medical Oncology, and he was later named to the R. Randall Rollins Chair in Oncology.
“We are all deeply grieving the loss of this remarkable man who gave so much to Winship,” said Walter J. Curran, Jr, MD, Winship Cancer Institute’s executive director.
“His enthusiasm and love for his patients and his commitment to lessening the burden of cancer for all has been unwavering throughout his life.”
A native of Beirut, Lebanon, Dr Khoury came to the Winship Cancer Institute from Washington University in St Louis, Missouri, where he served on the faculty after completing a fellowship in hematology-oncology.
He earned his medical degree from the Université Catholique de Louvain in Brussels, Belgium, and completed a residency in internal medicine at Memorial Medical Center in Savannah, Georgia.
Dr Khoury was recruited to Winship Cancer Institute by Fadlo R. Khuri, MD, former deputy director of the institute and now president of the American University of Beirut. What he first saw in Dr Khoury was someone who was “in the best sense, a disruptive presence.”
“What you always want in a leader is someone who’s not afraid to be wrong, to take risks,” Dr Khuri said. “Being wrong disrupts the pattern, and Jean was very brave. He didn’t like business as usual, and that showed in the way he took about redeveloping the hematology division, the leukemia program, and his interactions with the transplant division, with faculty, and all across Winship.”
According to his colleagues, Dr Khoury’s guiding principle was how to improve his patients’ lives, whether through research discoveries or through compassionate care.
Even after being diagnosed with cancer himself, Dr Khoury continued to see patients and carry on his work in the clinic and his research.
Dr Khoury pioneered the development of personalized treatment for CML patients and better approaches to improve quality of life for survivors. His research focused on drug development in leukemia and MDS, genomic abnormalities in leukemia, development of cost-effective practice models, and outcome analysis of bone marrow transplant.
He conducted several leukemia and transplant clinical trials, including trials that led to the approval of drugs such as imatinib, dasatinib, and nilotinib.
Dr Khoury received the Georgia Cancer Coalition Distinguished Cancer Scholarship, which allowed for establishment of the Hematological Disorders Tissue Bank at Emory, which now contains annotated germline and somatic samples from more than 800 patients with various hematologic disorders.
Dr Khoury died at home with his family by his side. He is survived by his wife, Angela, and 3 children, Mikhail, Iman, and Alya.
In lieu of flowers, the family requests that contributions be made to a new fund at Winship Cancer Institute that will memorialize the life and work of Dr Khoury by supporting a fellowship program that was so meaningful to him.
Contributions, marked in Memory of Dr H. Jean Khoury, can be sent to Winship Cancer Institute of Emory University, Office of Gift Records, Emory University, 1762 Clifton Rd. NE, Suite 1400, Atlanta, GA 30322. Gifts can also be made online.
There will be a memorial service for Dr Khoury on Wednesday, May 31, at 4:30 pm at Glenn Memorial Church, 1652 North Decatur Road in Atlanta, Georgia.
Physician, researcher, and educator H. Jean Khoury, MD, recently passed away.
He died on Monday, May 22, at the age of 50, after a year-long battle with esophageal cancer.
Dr Khoury led the division of hematology at Winship Cancer Institute of Emory University in Atlanta, Georgia.
He was considered an authority on hematologic malignancies, particularly chronic myeloid leukemia (CML), acute leukemia, and myelodysplastic syndromes (MDS).
Dr Khoury joined Winship Cancer Institute in 2004 as director of the Leukemia Service and associate professor in the Emory School of Medicine.
In 2009, he was promoted to professor and director of the Division of Hematology in the Department of Hematology and Medical Oncology, and he was later named to the R. Randall Rollins Chair in Oncology.
“We are all deeply grieving the loss of this remarkable man who gave so much to Winship,” said Walter J. Curran, Jr, MD, Winship Cancer Institute’s executive director.
“His enthusiasm and love for his patients and his commitment to lessening the burden of cancer for all has been unwavering throughout his life.”
A native of Beirut, Lebanon, Dr Khoury came to the Winship Cancer Institute from Washington University in St Louis, Missouri, where he served on the faculty after completing a fellowship in hematology-oncology.
He earned his medical degree from the Université Catholique de Louvain in Brussels, Belgium, and completed a residency in internal medicine at Memorial Medical Center in Savannah, Georgia.
Dr Khoury was recruited to Winship Cancer Institute by Fadlo R. Khuri, MD, former deputy director of the institute and now president of the American University of Beirut. What he first saw in Dr Khoury was someone who was “in the best sense, a disruptive presence.”
“What you always want in a leader is someone who’s not afraid to be wrong, to take risks,” Dr Khuri said. “Being wrong disrupts the pattern, and Jean was very brave. He didn’t like business as usual, and that showed in the way he took about redeveloping the hematology division, the leukemia program, and his interactions with the transplant division, with faculty, and all across Winship.”
According to his colleagues, Dr Khoury’s guiding principle was how to improve his patients’ lives, whether through research discoveries or through compassionate care.
Even after being diagnosed with cancer himself, Dr Khoury continued to see patients and carry on his work in the clinic and his research.
Dr Khoury pioneered the development of personalized treatment for CML patients and better approaches to improve quality of life for survivors. His research focused on drug development in leukemia and MDS, genomic abnormalities in leukemia, development of cost-effective practice models, and outcome analysis of bone marrow transplant.
He conducted several leukemia and transplant clinical trials, including trials that led to the approval of drugs such as imatinib, dasatinib, and nilotinib.
Dr Khoury received the Georgia Cancer Coalition Distinguished Cancer Scholarship, which allowed for establishment of the Hematological Disorders Tissue Bank at Emory, which now contains annotated germline and somatic samples from more than 800 patients with various hematologic disorders.
Dr Khoury died at home with his family by his side. He is survived by his wife, Angela, and 3 children, Mikhail, Iman, and Alya.
In lieu of flowers, the family requests that contributions be made to a new fund at Winship Cancer Institute that will memorialize the life and work of Dr Khoury by supporting a fellowship program that was so meaningful to him.
Contributions, marked in Memory of Dr H. Jean Khoury, can be sent to Winship Cancer Institute of Emory University, Office of Gift Records, Emory University, 1762 Clifton Rd. NE, Suite 1400, Atlanta, GA 30322. Gifts can also be made online.
There will be a memorial service for Dr Khoury on Wednesday, May 31, at 4:30 pm at Glenn Memorial Church, 1652 North Decatur Road in Atlanta, Georgia.
Physician, researcher, and educator H. Jean Khoury, MD, recently passed away.
He died on Monday, May 22, at the age of 50, after a year-long battle with esophageal cancer.
Dr Khoury led the division of hematology at Winship Cancer Institute of Emory University in Atlanta, Georgia.
He was considered an authority on hematologic malignancies, particularly chronic myeloid leukemia (CML), acute leukemia, and myelodysplastic syndromes (MDS).
Dr Khoury joined Winship Cancer Institute in 2004 as director of the Leukemia Service and associate professor in the Emory School of Medicine.
In 2009, he was promoted to professor and director of the Division of Hematology in the Department of Hematology and Medical Oncology, and he was later named to the R. Randall Rollins Chair in Oncology.
“We are all deeply grieving the loss of this remarkable man who gave so much to Winship,” said Walter J. Curran, Jr, MD, Winship Cancer Institute’s executive director.
“His enthusiasm and love for his patients and his commitment to lessening the burden of cancer for all has been unwavering throughout his life.”
A native of Beirut, Lebanon, Dr Khoury came to the Winship Cancer Institute from Washington University in St Louis, Missouri, where he served on the faculty after completing a fellowship in hematology-oncology.
He earned his medical degree from the Université Catholique de Louvain in Brussels, Belgium, and completed a residency in internal medicine at Memorial Medical Center in Savannah, Georgia.
Dr Khoury was recruited to Winship Cancer Institute by Fadlo R. Khuri, MD, former deputy director of the institute and now president of the American University of Beirut. What he first saw in Dr Khoury was someone who was “in the best sense, a disruptive presence.”
“What you always want in a leader is someone who’s not afraid to be wrong, to take risks,” Dr Khuri said. “Being wrong disrupts the pattern, and Jean was very brave. He didn’t like business as usual, and that showed in the way he took about redeveloping the hematology division, the leukemia program, and his interactions with the transplant division, with faculty, and all across Winship.”
According to his colleagues, Dr Khoury’s guiding principle was how to improve his patients’ lives, whether through research discoveries or through compassionate care.
Even after being diagnosed with cancer himself, Dr Khoury continued to see patients and carry on his work in the clinic and his research.
Dr Khoury pioneered the development of personalized treatment for CML patients and better approaches to improve quality of life for survivors. His research focused on drug development in leukemia and MDS, genomic abnormalities in leukemia, development of cost-effective practice models, and outcome analysis of bone marrow transplant.
He conducted several leukemia and transplant clinical trials, including trials that led to the approval of drugs such as imatinib, dasatinib, and nilotinib.
Dr Khoury received the Georgia Cancer Coalition Distinguished Cancer Scholarship, which allowed for establishment of the Hematological Disorders Tissue Bank at Emory, which now contains annotated germline and somatic samples from more than 800 patients with various hematologic disorders.
Dr Khoury died at home with his family by his side. He is survived by his wife, Angela, and 3 children, Mikhail, Iman, and Alya.
In lieu of flowers, the family requests that contributions be made to a new fund at Winship Cancer Institute that will memorialize the life and work of Dr Khoury by supporting a fellowship program that was so meaningful to him.
Contributions, marked in Memory of Dr H. Jean Khoury, can be sent to Winship Cancer Institute of Emory University, Office of Gift Records, Emory University, 1762 Clifton Rd. NE, Suite 1400, Atlanta, GA 30322. Gifts can also be made online.
There will be a memorial service for Dr Khoury on Wednesday, May 31, at 4:30 pm at Glenn Memorial Church, 1652 North Decatur Road in Atlanta, Georgia.
Single-cell analysis reveals TKI-resistant CML stem cells
Researchers say they’ve developed a technique for single-cell analysis that has revealed a population of treatment-resistant stem cells in patients with chronic myeloid leukemia (CML).
“It is increasingly recognized that tumors contain a variety of different cell types, including so-called cancer stem cells, that drive the growth and relapse of a patient’s cancer,” said study author Adam Mead, BM BCh, PhD, of University of Oxford in the UK.
“These cells can be very rare and extremely difficult to find after treatment as they become hidden within the normal tissue. We used a new genetic technique to identify and analyze single cancer stem cells in leukemia patients before and after treatment.”
Dr Mead and his colleagues detailed this research in Nature Medicine.
The team’s single-cell analysis technique combines high-sensitivity mutation detection with whole-transcriptome analysis.
The researchers used the method to analyze stem cells from patients with CML and found the cells to be heterogeneous.
In addition, the team was able to identify a subset of CML stem cells that proved resistant to treatment with tyrosine kinase inhibitors (TKIs).
“We found that, even in individual cases of leukemia, there are various types of cancer stem cell that respond differently to the treatment,” Dr Mead said.
“A small number of these cells are highly resistant to the treatment and are likely to be responsible for disease recurrence when the treatment is stopped. Our research allowed us uniquely to analyze these crucial cells that evade treatment so that we might learn how to more effectively eradicate them.”
The researchers said these TKI-resistant CML stem cells were “transcriptionally distinct” from normal hematopoietic stem cells.
The TKI-resistant cells were characterized by dysregulation of specific genes and pathways—TGF-β, TNF-α, JAK–STAT, CTNNB1, and NFKB1A—that could potentially be targeted to improve the treatment of CML.
The researchers also said their single-cell analysis technique can be used beyond CML.
“This technique could be adapted to analyze a range of different cancers to help predict both the likely response to treatment and the risk of the disease returning in the future,” Dr Mead said. “This should eventually enable treatment to be tailored to target each and every type of cancer stem cell that may be present.”
Researchers say they’ve developed a technique for single-cell analysis that has revealed a population of treatment-resistant stem cells in patients with chronic myeloid leukemia (CML).
“It is increasingly recognized that tumors contain a variety of different cell types, including so-called cancer stem cells, that drive the growth and relapse of a patient’s cancer,” said study author Adam Mead, BM BCh, PhD, of University of Oxford in the UK.
“These cells can be very rare and extremely difficult to find after treatment as they become hidden within the normal tissue. We used a new genetic technique to identify and analyze single cancer stem cells in leukemia patients before and after treatment.”
Dr Mead and his colleagues detailed this research in Nature Medicine.
The team’s single-cell analysis technique combines high-sensitivity mutation detection with whole-transcriptome analysis.
The researchers used the method to analyze stem cells from patients with CML and found the cells to be heterogeneous.
In addition, the team was able to identify a subset of CML stem cells that proved resistant to treatment with tyrosine kinase inhibitors (TKIs).
“We found that, even in individual cases of leukemia, there are various types of cancer stem cell that respond differently to the treatment,” Dr Mead said.
“A small number of these cells are highly resistant to the treatment and are likely to be responsible for disease recurrence when the treatment is stopped. Our research allowed us uniquely to analyze these crucial cells that evade treatment so that we might learn how to more effectively eradicate them.”
The researchers said these TKI-resistant CML stem cells were “transcriptionally distinct” from normal hematopoietic stem cells.
The TKI-resistant cells were characterized by dysregulation of specific genes and pathways—TGF-β, TNF-α, JAK–STAT, CTNNB1, and NFKB1A—that could potentially be targeted to improve the treatment of CML.
The researchers also said their single-cell analysis technique can be used beyond CML.
“This technique could be adapted to analyze a range of different cancers to help predict both the likely response to treatment and the risk of the disease returning in the future,” Dr Mead said. “This should eventually enable treatment to be tailored to target each and every type of cancer stem cell that may be present.”
Researchers say they’ve developed a technique for single-cell analysis that has revealed a population of treatment-resistant stem cells in patients with chronic myeloid leukemia (CML).
“It is increasingly recognized that tumors contain a variety of different cell types, including so-called cancer stem cells, that drive the growth and relapse of a patient’s cancer,” said study author Adam Mead, BM BCh, PhD, of University of Oxford in the UK.
“These cells can be very rare and extremely difficult to find after treatment as they become hidden within the normal tissue. We used a new genetic technique to identify and analyze single cancer stem cells in leukemia patients before and after treatment.”
Dr Mead and his colleagues detailed this research in Nature Medicine.
The team’s single-cell analysis technique combines high-sensitivity mutation detection with whole-transcriptome analysis.
The researchers used the method to analyze stem cells from patients with CML and found the cells to be heterogeneous.
In addition, the team was able to identify a subset of CML stem cells that proved resistant to treatment with tyrosine kinase inhibitors (TKIs).
“We found that, even in individual cases of leukemia, there are various types of cancer stem cell that respond differently to the treatment,” Dr Mead said.
“A small number of these cells are highly resistant to the treatment and are likely to be responsible for disease recurrence when the treatment is stopped. Our research allowed us uniquely to analyze these crucial cells that evade treatment so that we might learn how to more effectively eradicate them.”
The researchers said these TKI-resistant CML stem cells were “transcriptionally distinct” from normal hematopoietic stem cells.
The TKI-resistant cells were characterized by dysregulation of specific genes and pathways—TGF-β, TNF-α, JAK–STAT, CTNNB1, and NFKB1A—that could potentially be targeted to improve the treatment of CML.
The researchers also said their single-cell analysis technique can be used beyond CML.
“This technique could be adapted to analyze a range of different cancers to help predict both the likely response to treatment and the risk of the disease returning in the future,” Dr Mead said. “This should eventually enable treatment to be tailored to target each and every type of cancer stem cell that may be present.”