User login
Polatuzumab plus BR improves efficacy in DLBCL
CHICAGO—Polatuzumab vedotin, when added to bendamustine (B) and rituximab (R), significantly improved response and survival rates in a cohort of patients with relapsed/refractory diffuse large B-cell lymphoma (DLBCL), according to a phase 2 study.
By contrast, there were no such improvements in a cohort of follicular lymphoma (FL) patients, at least in short-term follow-up, investigator Laurie Helen Sehn, MD, of the BC Cancer Agency in Vancouver, Canada, said at the 2018 ASCO Annual Meeting.
However, the improvement in overall survival in DLBCL patients is “remarkable,” Dr Sehn affirmed in an oral presentation (abstract 7507).
“Based on these encouraging results, polatuzumab vedotin has received breakthrough therapy designation and priority medicines designation by the FDA and EMA for patients with relapsed or refractory DLBCL,” she said.
Polatuzumab-BR study (NCT02257567)
The study by Dr Sehn and colleagues included a cohort of 80 DLBCL patients randomized to BR or polatuzumab-BR for 6 planned 21-day cycles.
Investigators randomized another cohort of 80 FL patients to BR or polatuzumab-BR for 6 planned 28-day cycles.
The primary endpoint was complete response (CR) assessed by fluorodeoxyglucose positron emission tomography (FDG-PET) at 6 to 8 weeks after the end of treatment.
DLBCL patients
A total of 40% of polatuzumab-BR-treated DLBCL patients achieved CR at the end of treatment, versus 15% of BR-treated patients (P=0.012).
That CR improvement translated into a significantly higher progression-free survival (PFS) (6.7 months for polatuzumab-BR vs 2.0 months for BR, P<0.0001) and overall survival (11.8 months versus 4.7 months, P=0.0008), according to Dr Sehn.
The FDG-PET CR rates were higher in the polatuzumab-BR arm regardless of the number of prior lines of treatment for DLBCL, and regardless of relapsed versus refractory status, Dr. Sehn added.
FL patients
By contrast, in the FL cohort, the FDG-PET CR rate was high for both arms, at 69% for polatuzumab-BR and 63% for BR.
And there was no significant difference in progression-free survival (P=0.58) with “relatively short-term follow-up,” she said.
Adverse events
The most common grades 3 – 5 adverse events for both DLBCL and FL patients were higher in the polatuzumab-BR arm than the BR arm and included cytopenias, febrile neutropenia, and infections.
Serious AEs were also higher in the polatuzumab-BR arm and included febrile neutropenia for both FL and DLBCL patients and infection for FL patients.
Five percent of FL patients and 18% of DLBCL had a grade 5 event.
Commentary
Whether polatuzumab vedotin will change treatment paradigms for DLBCL patients may be answered by the ongoing POLARIX study, according to Alison Moskowitz, MD, of Memorial Sloan Kettering Cancer Center in New York, NY.
The randomized phase 3 POLARIX study (abstract TPS7589) is comparing polatuzumab plus R-CHP to R-CHOP in patients with previously untreated DLBCL.
“Certainly, there are patients who do very well with R-CHOP chemotherapy alone, and so we need to learn whether this is necessary for all patients, or only the high-risk patients,” Dr Moskowitz said in a talk at ASCO commenting on the results of the polatuzumab-BR study.
Hoffman-LaRoche is the sponsor of the study.
CHICAGO—Polatuzumab vedotin, when added to bendamustine (B) and rituximab (R), significantly improved response and survival rates in a cohort of patients with relapsed/refractory diffuse large B-cell lymphoma (DLBCL), according to a phase 2 study.
By contrast, there were no such improvements in a cohort of follicular lymphoma (FL) patients, at least in short-term follow-up, investigator Laurie Helen Sehn, MD, of the BC Cancer Agency in Vancouver, Canada, said at the 2018 ASCO Annual Meeting.
However, the improvement in overall survival in DLBCL patients is “remarkable,” Dr Sehn affirmed in an oral presentation (abstract 7507).
“Based on these encouraging results, polatuzumab vedotin has received breakthrough therapy designation and priority medicines designation by the FDA and EMA for patients with relapsed or refractory DLBCL,” she said.
Polatuzumab-BR study (NCT02257567)
The study by Dr Sehn and colleagues included a cohort of 80 DLBCL patients randomized to BR or polatuzumab-BR for 6 planned 21-day cycles.
Investigators randomized another cohort of 80 FL patients to BR or polatuzumab-BR for 6 planned 28-day cycles.
The primary endpoint was complete response (CR) assessed by fluorodeoxyglucose positron emission tomography (FDG-PET) at 6 to 8 weeks after the end of treatment.
DLBCL patients
A total of 40% of polatuzumab-BR-treated DLBCL patients achieved CR at the end of treatment, versus 15% of BR-treated patients (P=0.012).
That CR improvement translated into a significantly higher progression-free survival (PFS) (6.7 months for polatuzumab-BR vs 2.0 months for BR, P<0.0001) and overall survival (11.8 months versus 4.7 months, P=0.0008), according to Dr Sehn.
The FDG-PET CR rates were higher in the polatuzumab-BR arm regardless of the number of prior lines of treatment for DLBCL, and regardless of relapsed versus refractory status, Dr. Sehn added.
FL patients
By contrast, in the FL cohort, the FDG-PET CR rate was high for both arms, at 69% for polatuzumab-BR and 63% for BR.
And there was no significant difference in progression-free survival (P=0.58) with “relatively short-term follow-up,” she said.
Adverse events
The most common grades 3 – 5 adverse events for both DLBCL and FL patients were higher in the polatuzumab-BR arm than the BR arm and included cytopenias, febrile neutropenia, and infections.
Serious AEs were also higher in the polatuzumab-BR arm and included febrile neutropenia for both FL and DLBCL patients and infection for FL patients.
Five percent of FL patients and 18% of DLBCL had a grade 5 event.
Commentary
Whether polatuzumab vedotin will change treatment paradigms for DLBCL patients may be answered by the ongoing POLARIX study, according to Alison Moskowitz, MD, of Memorial Sloan Kettering Cancer Center in New York, NY.
The randomized phase 3 POLARIX study (abstract TPS7589) is comparing polatuzumab plus R-CHP to R-CHOP in patients with previously untreated DLBCL.
“Certainly, there are patients who do very well with R-CHOP chemotherapy alone, and so we need to learn whether this is necessary for all patients, or only the high-risk patients,” Dr Moskowitz said in a talk at ASCO commenting on the results of the polatuzumab-BR study.
Hoffman-LaRoche is the sponsor of the study.
CHICAGO—Polatuzumab vedotin, when added to bendamustine (B) and rituximab (R), significantly improved response and survival rates in a cohort of patients with relapsed/refractory diffuse large B-cell lymphoma (DLBCL), according to a phase 2 study.
By contrast, there were no such improvements in a cohort of follicular lymphoma (FL) patients, at least in short-term follow-up, investigator Laurie Helen Sehn, MD, of the BC Cancer Agency in Vancouver, Canada, said at the 2018 ASCO Annual Meeting.
However, the improvement in overall survival in DLBCL patients is “remarkable,” Dr Sehn affirmed in an oral presentation (abstract 7507).
“Based on these encouraging results, polatuzumab vedotin has received breakthrough therapy designation and priority medicines designation by the FDA and EMA for patients with relapsed or refractory DLBCL,” she said.
Polatuzumab-BR study (NCT02257567)
The study by Dr Sehn and colleagues included a cohort of 80 DLBCL patients randomized to BR or polatuzumab-BR for 6 planned 21-day cycles.
Investigators randomized another cohort of 80 FL patients to BR or polatuzumab-BR for 6 planned 28-day cycles.
The primary endpoint was complete response (CR) assessed by fluorodeoxyglucose positron emission tomography (FDG-PET) at 6 to 8 weeks after the end of treatment.
DLBCL patients
A total of 40% of polatuzumab-BR-treated DLBCL patients achieved CR at the end of treatment, versus 15% of BR-treated patients (P=0.012).
That CR improvement translated into a significantly higher progression-free survival (PFS) (6.7 months for polatuzumab-BR vs 2.0 months for BR, P<0.0001) and overall survival (11.8 months versus 4.7 months, P=0.0008), according to Dr Sehn.
The FDG-PET CR rates were higher in the polatuzumab-BR arm regardless of the number of prior lines of treatment for DLBCL, and regardless of relapsed versus refractory status, Dr. Sehn added.
FL patients
By contrast, in the FL cohort, the FDG-PET CR rate was high for both arms, at 69% for polatuzumab-BR and 63% for BR.
And there was no significant difference in progression-free survival (P=0.58) with “relatively short-term follow-up,” she said.
Adverse events
The most common grades 3 – 5 adverse events for both DLBCL and FL patients were higher in the polatuzumab-BR arm than the BR arm and included cytopenias, febrile neutropenia, and infections.
Serious AEs were also higher in the polatuzumab-BR arm and included febrile neutropenia for both FL and DLBCL patients and infection for FL patients.
Five percent of FL patients and 18% of DLBCL had a grade 5 event.
Commentary
Whether polatuzumab vedotin will change treatment paradigms for DLBCL patients may be answered by the ongoing POLARIX study, according to Alison Moskowitz, MD, of Memorial Sloan Kettering Cancer Center in New York, NY.
The randomized phase 3 POLARIX study (abstract TPS7589) is comparing polatuzumab plus R-CHP to R-CHOP in patients with previously untreated DLBCL.
“Certainly, there are patients who do very well with R-CHOP chemotherapy alone, and so we need to learn whether this is necessary for all patients, or only the high-risk patients,” Dr Moskowitz said in a talk at ASCO commenting on the results of the polatuzumab-BR study.
Hoffman-LaRoche is the sponsor of the study.
Chemo-free regimen appears viable in previously untreated FL
CHICAGO – Lenalidomide plus rituximab (R2) had comparable efficacy versus standard chemoimmunotherapy in patients with previously untreated follicular lymphoma, according to results from a phase 3 trial.
RELEVANCE is the first randomized, phase 3 trial to examine a chemotherapy-free regimen in this setting.
Response and progression-free survival (PFS) results were similar for patients who received R2 followed by rituximab maintenance and patients assigned to chemotherapy plus rituximab and rituximab maintenance, in study results presented at the annual meeting of the American Society of Clinical Oncology.
“These results show that lenalidomide plus rituximab, which is a novel immunomodulatory approach, is a potential first-line option for patients with follicular lymphoma that require treatment,” said investigator Nathan H. Fowler, MD, of the University of Texas MD Anderson Cancer Center, Houston.
But since the study was designed as a superiority trial, rather than a noninferiority trial, and it failed to meet its primary endpoint of superior complete remission (CR) or CR unconfirmed (CRu) at 120 weeks, said Bruce D. Cheson, MD, head of hematology at Georgetown University, Washington.
R2 had a similar PFS overall and in all major patient subgroups, similar overall survival, less nonhematologic toxicity aside from rash, less neutropenia, and fewer infections despite increased use of growth factors in the chemoimmunotherapy arm, Dr. Cheson said in a presentation commenting on the results. “Therefore, I agree with Dr. Fowler’s conclusion that R2 can be considered as an option for the front-line therapy of patients with follicular lymphoma,” Dr. Cheson said.
The RELEVANCE study included 1,030 patients (median age, 59 years) with previously untreated, advanced follicular lymphoma requiring treatment. They were randomized 1:1 to either lenalidomide plus rituximab followed by rituximab maintenance, or R-chemotherapy followed by rituximab maintenance.
For patients randomly assigned to R-chemotherapy, physicians could choose among three standard regimens: rituximab plus bendamustine (R-B), rituximab plus cyclophosphamide, doxorubicin, vincristine, and prednisone (R-CHOP), or rituximab plus cyclophosphamide, vincristine, and prednisone (R-CVP).
There was no statistical difference between treatment approaches in CR/CRu at 120 weeks, which was 48% in the R2 arm and 53% in the R-chemotherapy arm (P = 0.13). Best CR/CRu also was not statistically different between arms (59% and 67%, respectively), as was best overall response rate (84% and 89%). The 3-year duration of response was 77% in the R2 arm and 74% for R-chemotherapy.
With 37.9 months median follow-up, progression-free survival was “nearly identical” between the two groups, Dr. Fowler said, at 77% for R2 and 78% for R-chemotherapy (P = 0.48). The 3-year overall survival was 94% in both the R2 and R-chemotherapy arms, though survival data are still immature, Dr. Fowler noted.
Grade 3/4 neutropenia was more common in the R-chemotherapy arm, resulting in higher rates of febrile neutropenia, according to Dr. Fowler, who also noted that rash and cutaneous reactions were more common with R2. About 70% of patients in each arm were able to tolerate treatment, and reasons for discontinuation were “fairly similar” between arms, Dr. Fowler added.
Second primary malignancies occurred in 7% of patients in the R2 arm and 10% of the R-chemotherapy arm.
The study was sponsored was Celgene and the Lymphoma Academic Research Organisation. Dr. Fowler reported disclosures related to Abbvie, Celgene, Janssen, Merck, and Roche.
SOURCE: Fowler NH et al. ASCO 2018, Abstract 7500.
CHICAGO – Lenalidomide plus rituximab (R2) had comparable efficacy versus standard chemoimmunotherapy in patients with previously untreated follicular lymphoma, according to results from a phase 3 trial.
RELEVANCE is the first randomized, phase 3 trial to examine a chemotherapy-free regimen in this setting.
Response and progression-free survival (PFS) results were similar for patients who received R2 followed by rituximab maintenance and patients assigned to chemotherapy plus rituximab and rituximab maintenance, in study results presented at the annual meeting of the American Society of Clinical Oncology.
“These results show that lenalidomide plus rituximab, which is a novel immunomodulatory approach, is a potential first-line option for patients with follicular lymphoma that require treatment,” said investigator Nathan H. Fowler, MD, of the University of Texas MD Anderson Cancer Center, Houston.
But since the study was designed as a superiority trial, rather than a noninferiority trial, and it failed to meet its primary endpoint of superior complete remission (CR) or CR unconfirmed (CRu) at 120 weeks, said Bruce D. Cheson, MD, head of hematology at Georgetown University, Washington.
R2 had a similar PFS overall and in all major patient subgroups, similar overall survival, less nonhematologic toxicity aside from rash, less neutropenia, and fewer infections despite increased use of growth factors in the chemoimmunotherapy arm, Dr. Cheson said in a presentation commenting on the results. “Therefore, I agree with Dr. Fowler’s conclusion that R2 can be considered as an option for the front-line therapy of patients with follicular lymphoma,” Dr. Cheson said.
The RELEVANCE study included 1,030 patients (median age, 59 years) with previously untreated, advanced follicular lymphoma requiring treatment. They were randomized 1:1 to either lenalidomide plus rituximab followed by rituximab maintenance, or R-chemotherapy followed by rituximab maintenance.
For patients randomly assigned to R-chemotherapy, physicians could choose among three standard regimens: rituximab plus bendamustine (R-B), rituximab plus cyclophosphamide, doxorubicin, vincristine, and prednisone (R-CHOP), or rituximab plus cyclophosphamide, vincristine, and prednisone (R-CVP).
There was no statistical difference between treatment approaches in CR/CRu at 120 weeks, which was 48% in the R2 arm and 53% in the R-chemotherapy arm (P = 0.13). Best CR/CRu also was not statistically different between arms (59% and 67%, respectively), as was best overall response rate (84% and 89%). The 3-year duration of response was 77% in the R2 arm and 74% for R-chemotherapy.
With 37.9 months median follow-up, progression-free survival was “nearly identical” between the two groups, Dr. Fowler said, at 77% for R2 and 78% for R-chemotherapy (P = 0.48). The 3-year overall survival was 94% in both the R2 and R-chemotherapy arms, though survival data are still immature, Dr. Fowler noted.
Grade 3/4 neutropenia was more common in the R-chemotherapy arm, resulting in higher rates of febrile neutropenia, according to Dr. Fowler, who also noted that rash and cutaneous reactions were more common with R2. About 70% of patients in each arm were able to tolerate treatment, and reasons for discontinuation were “fairly similar” between arms, Dr. Fowler added.
Second primary malignancies occurred in 7% of patients in the R2 arm and 10% of the R-chemotherapy arm.
The study was sponsored was Celgene and the Lymphoma Academic Research Organisation. Dr. Fowler reported disclosures related to Abbvie, Celgene, Janssen, Merck, and Roche.
SOURCE: Fowler NH et al. ASCO 2018, Abstract 7500.
CHICAGO – Lenalidomide plus rituximab (R2) had comparable efficacy versus standard chemoimmunotherapy in patients with previously untreated follicular lymphoma, according to results from a phase 3 trial.
RELEVANCE is the first randomized, phase 3 trial to examine a chemotherapy-free regimen in this setting.
Response and progression-free survival (PFS) results were similar for patients who received R2 followed by rituximab maintenance and patients assigned to chemotherapy plus rituximab and rituximab maintenance, in study results presented at the annual meeting of the American Society of Clinical Oncology.
“These results show that lenalidomide plus rituximab, which is a novel immunomodulatory approach, is a potential first-line option for patients with follicular lymphoma that require treatment,” said investigator Nathan H. Fowler, MD, of the University of Texas MD Anderson Cancer Center, Houston.
But since the study was designed as a superiority trial, rather than a noninferiority trial, and it failed to meet its primary endpoint of superior complete remission (CR) or CR unconfirmed (CRu) at 120 weeks, said Bruce D. Cheson, MD, head of hematology at Georgetown University, Washington.
R2 had a similar PFS overall and in all major patient subgroups, similar overall survival, less nonhematologic toxicity aside from rash, less neutropenia, and fewer infections despite increased use of growth factors in the chemoimmunotherapy arm, Dr. Cheson said in a presentation commenting on the results. “Therefore, I agree with Dr. Fowler’s conclusion that R2 can be considered as an option for the front-line therapy of patients with follicular lymphoma,” Dr. Cheson said.
The RELEVANCE study included 1,030 patients (median age, 59 years) with previously untreated, advanced follicular lymphoma requiring treatment. They were randomized 1:1 to either lenalidomide plus rituximab followed by rituximab maintenance, or R-chemotherapy followed by rituximab maintenance.
For patients randomly assigned to R-chemotherapy, physicians could choose among three standard regimens: rituximab plus bendamustine (R-B), rituximab plus cyclophosphamide, doxorubicin, vincristine, and prednisone (R-CHOP), or rituximab plus cyclophosphamide, vincristine, and prednisone (R-CVP).
There was no statistical difference between treatment approaches in CR/CRu at 120 weeks, which was 48% in the R2 arm and 53% in the R-chemotherapy arm (P = 0.13). Best CR/CRu also was not statistically different between arms (59% and 67%, respectively), as was best overall response rate (84% and 89%). The 3-year duration of response was 77% in the R2 arm and 74% for R-chemotherapy.
With 37.9 months median follow-up, progression-free survival was “nearly identical” between the two groups, Dr. Fowler said, at 77% for R2 and 78% for R-chemotherapy (P = 0.48). The 3-year overall survival was 94% in both the R2 and R-chemotherapy arms, though survival data are still immature, Dr. Fowler noted.
Grade 3/4 neutropenia was more common in the R-chemotherapy arm, resulting in higher rates of febrile neutropenia, according to Dr. Fowler, who also noted that rash and cutaneous reactions were more common with R2. About 70% of patients in each arm were able to tolerate treatment, and reasons for discontinuation were “fairly similar” between arms, Dr. Fowler added.
Second primary malignancies occurred in 7% of patients in the R2 arm and 10% of the R-chemotherapy arm.
The study was sponsored was Celgene and the Lymphoma Academic Research Organisation. Dr. Fowler reported disclosures related to Abbvie, Celgene, Janssen, Merck, and Roche.
SOURCE: Fowler NH et al. ASCO 2018, Abstract 7500.
REPORTING FROM ASCO 2018
Key clinical point: in patients with previously untreated follicular lymphoma.
Major finding: With 37.9 months’ median follow-up, progression-free survival was “nearly identical” between the two groups, at 77% for R2 and 78% for rituximab chemotherapy (P = 0.48).
Study details: RELEVANCE, a phase 3, randomized clinical trial including 1,030 patients with previously untreated, advanced follicular lymphoma requiring treatment.
Disclosures: The study was sponsored was Celgene and the Lymphoma Academic Research Organisation. Dr. Fowler reported disclosures related to AbbVie, Celgene, Janssen, Merck, and Roche.
Source: Fowler NH et al. ASCO 2018, Abstract 7500.
Novel antibody shifts ‘eat me/don’t eat me’ balance in refractory NHL
CHICAGO – A first-in-class antibody targeting the macrophage checkpoint CD47 is a promising novel immunotherapy in non-Hodgkin lymphoma, according to Ranjana H. Advani, MD, of Stanford (Calif.) Cancer Institute.
Treatment with Hu5F9-G4 (5F9), an antibody designed to overcome the “don’t eat me” signal associated with CD47, produced “encouraging” antitumor activity in a phase 1b study of 22 patients, Dr. Advani said in an oral abstract presentation at the annual meeting of the American Society of Clinical Oncology.
“5F9 was well tolerated in combination with rituximab, with no maximum tolerated dose achieved,” said Dr. Advani, noting that there were complete remissions in 43% of the refractory follicular lymphoma patients and 33% of refractory diffuse large B-cell lymphoma patients in the phase 1b/2 study.
The antibody has an on-target anemia effect that occurs upon administration, but that was mitigated considerably by a priming and maintenance dosing approach, she added.
The study has demonstrated “excellent” response rates in a highly refractory patient population, said Caron A. Jacobson, MD, of Dana-Farber Cancer Institute and Harvard Medical School in Boston. “Targeting CD47 ... really helps to shift the balance from ‘don’t eat me’ to ‘eat me,’ ” Dr. Jacobson said at the meeting.
“Importantly, we saw very little toxicity in the study, with very few grade 4 adverse events and no immune-related adverse events,” she added.
Most adverse events were grade 1 or 2, with the most common being the expected on-target anemia associated with 5F9. Using an initial priming dose of 5F9 results in a “temporary and mild decline” in hemoglobin due to clearance of aged red blood cells, Dr. Advani said.
The objective response rate in the study was 50%, with efficacy observed in rituximab-refractory patients, Dr. Advani said. With a median follow-up of greater than 6 months, just 1 of 11 responders had progressed. The median duration of response was not reached, with the longest complete remission lasting more than 14 months.
5F9 is able to selectively eliminate cancer cells through blockade of CD47, while rituximab enhances 5F9’s activity via antibody-dependent cellular phagocytosis, according to Dr. Advani.
“CD47 blockade takes the foot off the brakes, while rituximab puts the foot on the accelerator, leading to maximal tumor phagocytosis,” she said.
The Food and Drug Administration recently granted 5F9 a fast track designation for both diffuse large B-cell lymphoma and follicular lymphoma. Phase 2 investigations of 5F9 in these lymphomas are ongoing, Dr. Advani said.
The trial is sponsored by Forty Seven. Dr. Advani reported research funding from Forty Seven, which is developing 5F9, as well as disclosures related to AstraZeneca, Bayer, Bristol-Myers Squibb, Cell Medica, Genentech/Roche, Gilead Sciences, Pharmacyclics, and Seattle Genetics, among others.
SOURCE: Advani RH et al. ASCO 2018, abstract 7504.
CHICAGO – A first-in-class antibody targeting the macrophage checkpoint CD47 is a promising novel immunotherapy in non-Hodgkin lymphoma, according to Ranjana H. Advani, MD, of Stanford (Calif.) Cancer Institute.
Treatment with Hu5F9-G4 (5F9), an antibody designed to overcome the “don’t eat me” signal associated with CD47, produced “encouraging” antitumor activity in a phase 1b study of 22 patients, Dr. Advani said in an oral abstract presentation at the annual meeting of the American Society of Clinical Oncology.
“5F9 was well tolerated in combination with rituximab, with no maximum tolerated dose achieved,” said Dr. Advani, noting that there were complete remissions in 43% of the refractory follicular lymphoma patients and 33% of refractory diffuse large B-cell lymphoma patients in the phase 1b/2 study.
The antibody has an on-target anemia effect that occurs upon administration, but that was mitigated considerably by a priming and maintenance dosing approach, she added.
The study has demonstrated “excellent” response rates in a highly refractory patient population, said Caron A. Jacobson, MD, of Dana-Farber Cancer Institute and Harvard Medical School in Boston. “Targeting CD47 ... really helps to shift the balance from ‘don’t eat me’ to ‘eat me,’ ” Dr. Jacobson said at the meeting.
“Importantly, we saw very little toxicity in the study, with very few grade 4 adverse events and no immune-related adverse events,” she added.
Most adverse events were grade 1 or 2, with the most common being the expected on-target anemia associated with 5F9. Using an initial priming dose of 5F9 results in a “temporary and mild decline” in hemoglobin due to clearance of aged red blood cells, Dr. Advani said.
The objective response rate in the study was 50%, with efficacy observed in rituximab-refractory patients, Dr. Advani said. With a median follow-up of greater than 6 months, just 1 of 11 responders had progressed. The median duration of response was not reached, with the longest complete remission lasting more than 14 months.
5F9 is able to selectively eliminate cancer cells through blockade of CD47, while rituximab enhances 5F9’s activity via antibody-dependent cellular phagocytosis, according to Dr. Advani.
“CD47 blockade takes the foot off the brakes, while rituximab puts the foot on the accelerator, leading to maximal tumor phagocytosis,” she said.
The Food and Drug Administration recently granted 5F9 a fast track designation for both diffuse large B-cell lymphoma and follicular lymphoma. Phase 2 investigations of 5F9 in these lymphomas are ongoing, Dr. Advani said.
The trial is sponsored by Forty Seven. Dr. Advani reported research funding from Forty Seven, which is developing 5F9, as well as disclosures related to AstraZeneca, Bayer, Bristol-Myers Squibb, Cell Medica, Genentech/Roche, Gilead Sciences, Pharmacyclics, and Seattle Genetics, among others.
SOURCE: Advani RH et al. ASCO 2018, abstract 7504.
CHICAGO – A first-in-class antibody targeting the macrophage checkpoint CD47 is a promising novel immunotherapy in non-Hodgkin lymphoma, according to Ranjana H. Advani, MD, of Stanford (Calif.) Cancer Institute.
Treatment with Hu5F9-G4 (5F9), an antibody designed to overcome the “don’t eat me” signal associated with CD47, produced “encouraging” antitumor activity in a phase 1b study of 22 patients, Dr. Advani said in an oral abstract presentation at the annual meeting of the American Society of Clinical Oncology.
“5F9 was well tolerated in combination with rituximab, with no maximum tolerated dose achieved,” said Dr. Advani, noting that there were complete remissions in 43% of the refractory follicular lymphoma patients and 33% of refractory diffuse large B-cell lymphoma patients in the phase 1b/2 study.
The antibody has an on-target anemia effect that occurs upon administration, but that was mitigated considerably by a priming and maintenance dosing approach, she added.
The study has demonstrated “excellent” response rates in a highly refractory patient population, said Caron A. Jacobson, MD, of Dana-Farber Cancer Institute and Harvard Medical School in Boston. “Targeting CD47 ... really helps to shift the balance from ‘don’t eat me’ to ‘eat me,’ ” Dr. Jacobson said at the meeting.
“Importantly, we saw very little toxicity in the study, with very few grade 4 adverse events and no immune-related adverse events,” she added.
Most adverse events were grade 1 or 2, with the most common being the expected on-target anemia associated with 5F9. Using an initial priming dose of 5F9 results in a “temporary and mild decline” in hemoglobin due to clearance of aged red blood cells, Dr. Advani said.
The objective response rate in the study was 50%, with efficacy observed in rituximab-refractory patients, Dr. Advani said. With a median follow-up of greater than 6 months, just 1 of 11 responders had progressed. The median duration of response was not reached, with the longest complete remission lasting more than 14 months.
5F9 is able to selectively eliminate cancer cells through blockade of CD47, while rituximab enhances 5F9’s activity via antibody-dependent cellular phagocytosis, according to Dr. Advani.
“CD47 blockade takes the foot off the brakes, while rituximab puts the foot on the accelerator, leading to maximal tumor phagocytosis,” she said.
The Food and Drug Administration recently granted 5F9 a fast track designation for both diffuse large B-cell lymphoma and follicular lymphoma. Phase 2 investigations of 5F9 in these lymphomas are ongoing, Dr. Advani said.
The trial is sponsored by Forty Seven. Dr. Advani reported research funding from Forty Seven, which is developing 5F9, as well as disclosures related to AstraZeneca, Bayer, Bristol-Myers Squibb, Cell Medica, Genentech/Roche, Gilead Sciences, Pharmacyclics, and Seattle Genetics, among others.
SOURCE: Advani RH et al. ASCO 2018, abstract 7504.
REPORTING FROM ASCO 2018
Key clinical point:
Major finding: Complete responses were seen in 43% of follicular lymphoma (FL) patients and 33% of diffuse large B-cell lymphoma (DLBCL) patients.
Study details: Initial reported results from a phase 1b/2 study of 7 patients with FL and 15 patients with DLBCL.
Disclosures: Forty Seven sponsored the trial. Dr. Advani reported research funding from Forty Seven, which is developing 5F9, as well as disclosures related to AstraZeneca, Bayer, Bristol-Myers Squibb, Cell Medica, Genentech/Roche, Gilead Sciences, Pharmacyclics, and Seattle Genetics, among others.
Source: Advani RH et al. ASCO 2018, abstract 7504.
Aggressive B-Cell Non-Hodgkin Lymphoma
Introduction
Non-Hodgkin lymphoma (NHL) comprises a wide variety of malignant hematologic disorders with varying clinical and biological features. The more than 60 separate NHL subtypes can be classified according to cell of origin (B cell versus T cell), anatomical location (eg, orbital, testicular, bone, central nervous system), clinical behavior (indolent versus aggressive), histological features, or cytogenetic abnormalities. Although various NHL classification schemes have been used over the years, the World Health Organization (WHO) classification is now widely accepted as the definitive pathologic classification system for lymphoproliferative disorders, incorporating morphologic, immunohistochemical, flow cytometric, cytogenetic, and molecular features.1 While the pathologic and molecular subclassification of NHL has become increasingly refined in recent years, from a management standpoint, classification based on clinical behavior remains very useful. This approach separates NHL subtypes into indolent versus aggressive categories. Whereas indolent NHLs may remain clinically insignificant for months to years, aggressive B-cell NHLs generally become life-threatening within weeks to months without treatment.
Epidemiology
Data from cancer registries show a steady, unexplainable increase in the incidence of NHL during the second half of the 20th century; the incidence has subsequently plateaued. There was a significant increase in NHL incidence between 1970 and 1995, which has been attributed in part to the HIV epidemic. More than 72,000 new cases of NHL were diagnosed in the United States in 2017, compared to just over 8000 cases of Hodgkin lymphoma, making NHL the sixth most common cancer in adult men and the fifth most common in adult women.2 NHL appears to occur more frequently in Western countries than in Asian populations.
Various factors associated with increased risk for B-cell NHL have been identified over the years, including occupational and environmental exposure to certain pesticides and herbicides,3 immunosuppression associated with HIV infection,4 autoimmune disorders,5 iatrogenically induced immune suppression in the post-transplant and other settings,6 family history of NHL,7 and a personal history of a prior cancer, including Hodgkin lymphoma and prior NHL.8 In terms of infectious agents associated with aggressive B-cell NHLs, Epstein-Barr virus (EBV) has a clear pathogenic role in Burkitt lymphoma, in many cases of post-transplant lymphoproliferative disorders, and in some cases of HIV-related aggressive B-cell lymphoma.9 Human herpesvirus-8 viral genomes have been found in virtually all cases of primary effusion lymphomas.10 Epidemiological studies also have linked hepatitis B and C to increased incidences of certain NHL subtypes,11–13 including primary hepatic diffuse large B-cell lymphoma (DLBCL). Similarly, Helicobacter pylori has been associated with gastric DLBCL.
Staging and Work-Up
A tissue biopsy is essential in the diagnosis and management of NHL. The most significant disadvantage of fine-needle aspiration cytology is the lack of histologic architecture. The optimal specimen is an excisional biopsy; when this cannot be performed, a core needle biopsy, ideally using a 16-gauge or larger caliber needle, is the next best choice.
The baseline tests appropriate for most cases of newly diagnosed aggressive B-cell NHL are listed in Table 1. Both hepatitis B and C have been associated with increased risk of NHL. In addition, there is a risk of hepatitis B reactivation following certain NHL therapies. A contrast-enhanced computed tomography (CT) scan in addition to positron emission tomography (PET) is useful to define the extent of disease in situations needing greater definition (eg, lymphadenopathy close to the bowel, cervical and supraclavicular nodal involvement, and lymphadenopathy causing thrombosis or compression of nearby structures).14 In cases where it is apparent that the patient has advanced stage disease (Ann Arbor stage III/IV) based on imaging, bone marrow biopsy is unlikely to alter the treatment plan. For such patients, if the complete blood count is unremarkable, deferral of bone marrow biopsy may be reasonable. For new cases of DLBCL, assessment for MYC translocation by fluorescence in situ hybridization (FISH) is recommended. If a MYC translocation is identified, then testing for BCL2 and BCL6 translocations by FISH should be performed.
Prior to the initiation of treatment, patients should always undergo a thorough cardiac and pulmonary evaluation, especially if the patient will be treated with an anthracycline or mediastinal irradiation. Central nervous system (CNS) evaluation with magnetic resonance imaging (MRI) and lumbar puncture is essential if there are neurological signs or symptoms. In addition, certain anatomical sites including the testicles, paranasal sinuses, kidney, adrenal glands, and epidural space have been associated with increased involvement of the CNS and may warrant MRI evaluation and lumbar puncture. Certain NHL subtypes like Burkitt lymphoma, high-grade NHL with translocations of MYC and BCL-2 or BCL-6 (double-hit lymphoma), blastoid mantle cell lymphoma, and lymphoblastic lymphoma have a high risk of CNS involvement, and patients with these subtypes need CNS evaluation.
The Lugano classification is used to stage patients with NHL.14 This classification is based on the Ann Arbor staging system and uses the distribution and number of tumor sites to stage disease. In general, this staging system in isolation is of limited value in predicting survival after treatment. However, the Ann Arbor stage does have prognostic impact when incorporated into risk scoring systems such as the International Prognostic Index (IPI). In clinical practice, the Ann Arbor stage is useful primarily to determine eligibility for localized therapy approaches. The absence or presence of systemic symptoms such as fevers, drenching night sweats, or weight loss (> 10% of baseline over 6 months or less) is designated by A or B, respectively.
Diffuse Large B-Cell Lymphoma
DLBCL is the most common lymphoid neoplasm in adults, accounting for about 25% of all NHL cases.2 It is increasingly clear that the diagnostic category of DLBCL is quite heterogeneous in terms of morphology, genetics, and biologic behavior. A number of clinicopathologic subtypes of DLBCL exist, such as T cell/histiocyte–rich large B-cell lymphoma, primary mediastinal large B-cell lymphoma, intravascular large B-cell lymphoma, DLBCL associated with chronic inflammation, lymphomatoid granulomatosis, and EBV-positive large B-cell lymphoma, among others. Gene expression profiling (GEP) can distinguish 2 cell of origin DLBCL subtypes: the germinal center B-cell (GCB) and activated B-cell (ABC) subtypes.15
DLBCL may be primary (de novo) or may arise through the transformation of many different types of low-grade B-cell lymphomas. This latter scenario is referred to as histologic transformation or transformed lymphoma. In some cases, patients may have a previously diagnosed low-grade B-cell NHL; in other cases, both low-grade and aggressive B-cell NHL may be diagnosed concurrently. The presence of elements of both low-grade and aggressive B-cell NHL in the same biopsy specimen is sometimes referred to as a composite lymphoma.
In the United States, incidence varies by ethnicity, with DLBCL being more common in Caucasians than other races.16 There is a slight male predominance (55%), median age at diagnosis is 65 years,16,17 and the incidence increases with age.
Presentation, Pathology, and Prognostic Factors
The most common presentation of patients with DLBCL is rapidly enlarging lymphadenopathy, usually in the neck or abdomen. Extranodal/extramedullary presentation is seen in approximately 40% of cases, with the gastrointestinal (GI) tract being the most common site. However, extranodal DLBCL can arise in virtually any tissue.18 Nodal DLBCL presents with symptoms related to the sites of involvement (eg, shortness of breath or chest pain with mediastinal lymphadenopathy), while extranodal DLBCL typically presents with symptoms secondary to dysfunction at the site of origin. Up to one third of patients present with constitutional symptoms (B symptoms) and more than 50% have elevated serum lactate dehydrogenase (LDH) at diagnosis.19
Approximately 40% of patients present with stage I/II disease. Of these, only a subset present with stage I, or truly localized disease (defined as that which can be contained within 1 irradiation field). About 60% of patients present with advanced (stage III–IV) disease.20 The bone marrow is involved in about 15% to 30% of cases. DLBCL involvement of the bone marrow is associated with a less favorable prognosis. Patients with DLBCL elsewhere may have low-grade NHL involvement of the bone marrow. Referred to as discordant bone marrow involvement,21 this feature does not carry the same poor prognosis associated with transformed disease22 or DLBCL involvement of the bone marrow.23
DLBCL is defined as a neoplasm of large B-lymphoid cells with a diffuse growth pattern. The proliferative fraction of cells, as determined by Ki-67 staining, is usually greater than 40%, and may even exceed 90%. Lymph nodes usually demonstrate complete effacement of the normal architecture by sheets of atypical lymphoid cells. Tumor cells in DLBCL generally express pan B-cell antigens (CD19, CD20, CD22, CD79a, Pax-5) as well as CD45 and surface immunoglobulin. Between 20% and 37% of DLBCL cases express the BCL-2 protein,24 and about 70% express the BCL-6 protein.25 C-MYC protein expression is seen in a higher percentage (~ 30%–50%) of cases of DLBCL.26
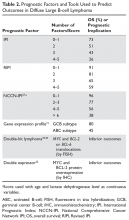
Many factors are associated with outcome in DLBCL. The IPI score was developed in the pre-rituximab era and is a robust prognostic tool. This simple tool uses 5 easily obtained clinical factors (age > 60 years, impaired performance status, elevated LDH, > 1 extranodal site of disease, and stage III/IV disease). By summing these factors, 4 groups with distinct 5-year overall survival (OS) rates ranging from 26% to 73% were identified (Table 2). Subsequently, modifications were made to adjust for age and stage, with the latest iteration being the NCCN (National Comprehensive Cancer Network) IPI.27 This tool uses age, performance status, LDH ratio (relative to the upper limit of normal), a more precise definition for presence of extranodal sites of disease (defined as lymphomatous involvement in the bone marrow, CNS, liver/GI tract, or lung), and Ann Arbor stage to stratify patients into 4 risk groups with significantly different 5-year OS, ranging from 38% to 96% based on the subgroup. Importantly, the NCCN-IPI was derived in a cohort of patients treated with rituximab-based therapy.
Cytogenetic and molecular factors also predict outcome in DLBCL. The ABC subtype distinguished by GEP has consistently been shown to have inferior outcomes with first-line therapy. As GEP is not routinely available in clinical practice, immunohistochemical (IHC) approaches (eg, the Hans algorithm) have been developed that can approximate the GEP subtypes. These IHC approaches have approximately 80% concordance with GEP.28 The 3 most common chromosomal translocations in DLBCL involve BCL-2, BCL-6 and MYC. MYC-rearranged DLBCLs have a less favorable prognosis.29,30 Cases in which a MYC translocation occurs in combination with a BCL-2 or BCL-6 translocation are commonly referred to as double-hit lymphoma (DHL); cases with all 3 translocations are referred to as triple-hit lymphoma (THL). Both DHL and THL have a worse prognosis with standard DLBCL therapy compared to non-DHL/THL cases. In the 2016 revised WHO classification, DHL and THL are an entity technically distinct from DLBCL, referred to as high-grade B-cell lymphoma.1 In some cases, MYC and BCL-2 protein overexpression occurs in the absence of chromosomal translocations. Cases in which MYC and BCL-2 are overexpressed (by IHC) are referred to as double expressor lymphoma (DEL), and also have inferior outcome compared with non-DEL DLBCL.31,32 Interestingly, MYC protein expression alone does not confer inferior outcomes, unlike isolated MYC translocation, which is associated with inferior outcomes.
Treatment
First-Line Therapy
DLBCL is an aggressive disease and, in most cases, survival without treatment can be measured in weeks to months. The advent of combination chemotherapy (CHOP [cyclophosphamide, doxorubicin, vincristine, and prednisone] or CHOP-like regimens) led to disease-free survival (DFS) rates of 35% to 40% at 3 to 5 years.33 The addition of rituximab to CHOP (R-CHOP) has improved both progression-free surivial (PFS) and OS.34,35
Treatment options vary for patients with localized (stage I/II) and advanced (stage III/IV) disease. Options for limited-stage DLBCL include an abbreviated course of R-CHOP (3 or 4 cycles) with involved-field radiation therapy (IFRT) versus a full course (6–8 cycles) of R-CHOP without radiation therapy (RT). Most studies comparing combined modality therapy (chemotherapy plus RT) versus chemotherapy alone were conducted in the pre-rituximab era. With the introduction of rituximab, Persky and colleagues36 studied the use of 3 cycles of R-CHOP followed by RT, demonstrating a slightly improved OS of 92% at 4 years as compared to 88% in a historical cohort. The French LYSA/GOELAMS group performed the only direct comparison in the rituximab era (4 cycles of R-CHOP followed by RT versus 4 cycles of R-CHOP followed by 2 additional cycles of R-CHOP) and reported similar outcomes between both arms,37 with OS of 92% in the R-CHOP alone arm and 96% in the R-CHOP + RT arm (nonsignificant difference statistically). IFRT alone is not recommended other than for palliation in patients who cannot tolerate chemotherapy or combined modality therapy. Stage I and II patients with bulky disease (> 10 cm) have a prognosis similar to patients with advanced DLBCL and should be treated aggressively with 6 to 8 cycles of R-CHOP with or without RT.36
For patients with advanced stage disease, a full course of R-CHOP-21 (6–8 cycles given on a 21-day cycle) is the standard of care. This approach results in OS rates of 70% and 60% at 2 and 5 years, respectively. For older adults unable to tolerate full-dose R-CHOP, attenuated versions of R-CHOP with decreased dose density or decreased dose intensity have been developed.38 Numerous randomized trials have attempted to improve upon the results of R-CHOP-21 using strategies such as infusional chemotherapy (DA-EPOCH-R [etoposide, prednisone, vincristine, cyclophosphamide, doxorubicin, rituximab]);39 dose-dense therapy (R-CHOP-14);replacement of rituximab with obinutuzuimab;40 addition of novel agents such as bortezomib,41 lenalidomide,42 or ibrutinib43,44 to R-CHOP; and various maintenance strategies such as rituximab, lenalidomide,45 enzastaurin,46 and everolimus.47 Unfortunately, none of these strategies has been shown to improve OS in DLBCL. In part this appears to be due to the fact that inclusion/exclusion criteria for DLBCL trials have been too strict, such that the most severely ill DLBCL patients are typically not included. As a result, the results in the control arms have ended up better than what was expected based on historical data. Efforts are underway to include all patients in future first-line DLBCL studies.
Currently, autologous hematopoietic cell transplantation (auto-HCT) is not routinely used in the initial treatment of DLBCL. In the pre-rituximab era, numerous trials were conducted in DLBCL patients with high and/or high-intermediate risk disease based on the IPI score to determine if outcomes could be improved with high-dose therapy and auto-HCT as consolidation after patients achieved complete remission with first-line therapy. The results of these trials were conflicting. A 2003 meta-analysis of 11 such trials concluded that the results were very heterogeneous and showed no OS benefit.48 More recently, the Southwestern Oncology Group published the results of a prospective trial testing the impact of auto-HCT for consolidation of aggressive NHL patients with an IPI score of 3 to 5 who achieved complete remission with first-line therapy with CHOP or R-CHOP. In this study, 75% of the patients had DLBCL and, of the B-cell NHL patients, 47% received R-CHOP. A survival benefit was seen only in the subgroup that had an IPI score of 4 or 5; a subgroup analysis restricted to those receiving R-CHOP as induction was not performed, however.49 As a result, this area remains controversial, with most institutions not routinely performing auto-HCT for any DLBCL patients in first complete remission and some institutions considering auto-HCT in first complete remission for patients with an IPI score of 4 or 5. These studies all used the IPI score to identify high-risk patients. It is possible that the use of newer biomarkers or minimal-residual disease analysis will lead to a more robust algorithm for identifying high-risk patients and selecting patients who might benefit from consolidation of first complete remission with auto-HCT.
For patients with DHL or THL, long-term PFS with standard R-CHOP therapy is poor (20% to 40%).50,51 Treatment with more intensive first-line regimens such as DA-EPOCH-R, R-hyperCVAD (rituximab plus hyperfractionated cyclophosphamide, vincristine, doxorubicin, dexamethasone), or CODOX-M/IVAC±R (cyclophosphamide, vincristine, doxorubicin, high‐dose methotrexate/ifosfamide, etoposide, high‐dose cytarabine ± rituximab), along with CNS prophylaxis, however, has been shown to produce superior outcomes,52 with 3-year relapse-free survival rates of 88% compared to 56% for R-CHOP. For patients who achieve a complete response by PET/CT scan after intensive induction, consolidation with auto-HCT has not been shown to improve outcomes based on retrospective analysis. However for DHL/THL patients who achieve complete response after R-CHOP, PFS was improved if auto-HCT was given as consolidation of first remission.53
Patients with DLBCL have an approximately 5% risk of subsequently developing CNS involvement. Historically (in the pre-rituximab era), patients who presented with multiple sites of extranodal disease and/or extensive bone marrow involvement and/or an elevated LDH had an increased risk (up to 20%–30%) of developing CNS involvement. In addition, patients with involvement of certain anatomical sites (testicular, paranasal sinuses, epidural space) had an increased risk of CNS disease. Several algorithms have been proposed to identify patients who should receive prophylactic CNS therapy. One of the most robust tools for this purpose is the CNS-IPI, which is a 6-point score consisting of the 5 IPI elements, plus 1 additional point if the adrenal glands or kidneys are involved. Importantly, the CNS-IPI was developed and validated in patients treated with R-CHOP-like therapy. Subsequent risk of CNS relapse was 0.6%, 3.4%, and 10.2% for those with low-, intermediate- and high-risk CNS-IPI scores, respectively.54 A reasonable strategy, therefore, is to perform CNS prophylaxis in those with a CNS-IPI score of 4 to 6. When CNS prophylaxis is used, intrathecal methotrexate or high-dose systemic methotrexate is most frequently given, with high-dose systemic methotrexate favored over intrathecal chemotherapy given that high-dose methotrexate penetrates the brain and spinal cord parenchyma, in addition to treating the cerebrospinal fluid (CSF).55 In contrast, intrathecal therapy only treats the CSF and requires repeated lumbar punctures or placement of an Ommaya reservoir. For DLBCL patients who present with active CSF involvement (known as lymphomatous meningitis), intrathecal chemotherapy treatments are typically given 2 or 3 times weekly until the CSF clears, followed by weekly intrathecal treatment for 4 weeks, and then monthly intrathecal treatment for 4 months.56 For those with concurrent systemic and brain parenchymal DLBCL, a strategy of alternating R-CHOP with mid-cycle high-dose methotrexate can be successful. In addition, consolidation with high-dose therapy and auto-HCT improved survival in such patients in 1 retrospective series.57
Relapsed/Refractory Disease
Between 30% and 40% of patients with advanced stage DLBCL will either fail to attain a remission with primary therapy (referred to as primary induction failure) or will relapse. In general, for those with progressive or relapsed disease, an updated tissue biopsy is recommended. This is especially true for patients who have had prior complete remission and have new lymph node enlargement, or those who have emergence of new sites of disease at the completion of first-line therapy.
Patients with relapsed disease are treated with systemic second-line platinum-based chemoimmunotherapy, with the usual goal of ultimately proceeding to auto-HCT. A number of platinum-based regimens have been used in this setting such as R-ICE, R-DHAP, R-GDP, R-Gem-Ox, and R-ESHAP. None of these regimens has been shown to be superior in terms of efficacy, and the choice of regimen is typically made based on the anticipated tolerance of the patient in light of comorbidities, laboratory studies, and physician preference. In the CORAL study, R-DHAP (rituximab, dexamethasone, high-dose cytarabine, cisplatin) seemed to show superior PFS in patients with the GCB subtype.58 However, this was an unplanned subgroup analysis and R-DHAP was associated with higher renal toxicity.
Several studies have demonstrated that long-term PFS can be observed for relapsed/refractory DLBCL patients who respond to second-line therapy and then undergo high-dose therapy with auto-HCT. The Parma trial remains the only published prospective randomized trial performed in relapsed DLBCL comparing a transplant strategy to a non-transplant strategy. This study, performed in the pre-rituximab era, clearly showed a benefit in terms of DFS and OS in favor of auto-HCT versus salvage therapy alone.59 The benefit of auto-HCT in patients treated in the rituximab era, even in patients who experience early failure (within 1 year of diagnosis), was confirmed in a retrospective analysis by the Center for International Blood and Marrow Transplant Research. In this study, a 44% 3-year PFS was seen in the early failure cohort versus 52% in the late failure cohort.60
Some DLBCL patients are very unlikely to benefit from auto-HCT. The REFINE study focused on patients with primary induction failure or early relapse within 6 months of completing first-line therapy. Among such patients, primary progressive disease (defined as progression while still receiving first-line therapy), a high NCCN-IPI score at relapse, and MYC rearrangement were risk factors for poor PFS following auto-HCT.61 Patients with 2 or 3 high-risk features had a 2-year OS of 10.7% compared to 74.3% for those without any high-risk features.
Allogeneic HCT (allo-HCT) is a treatment option for relapsed/refractory DLBCL. This option is more commonly considered for patients in whom an autotransplant has failed to achieve durable remission. For properly selected patients in this setting, a long-term PFS in the 30% to 40% range can be attained.62 However, in practice, only about 20% of patients who fail auto-HCT end up undergoing allo-HCT due to rapid progression of disease, age, poor performance status, or lack of suitable donor. It has been proposed that in the coming years, allo-HCT will be utilized less commonly in this setting due to the advent of chimeric antigen receptor T-cell (CAR T) therapy.
CAR T-cell therapy genetically modifies the patient’s own T lymphocytes with a gene that encodes an antigen receptor to direct the T cells against lymphoma cells. Typically, the T cells are genetically modified and expanded in a production facility and then infused back into the patient. Axicabtagene ciloleucel is directed against the CD-19 receptor and has been approved by the US Food and Drug Administration (FDA) for treatment of patients with DLBCL who have failed 2 or more lines of systemic therapy. Use of CAR-T therapy in such patients was examined in a multicenter trial (ZUMA-1), which reported a 54% complete response rate and 52% OS rate at 18 months.63 CAR-T therapy is associated with serious side effects such as cytokine release syndrome, neurological toxicities, and prolonged cytopenias. While there are now some patients with ongoing remission 2 or more years after undergoing CAR-T therapy, it remains uncertain what proportion of patients have been truly cured with this modality. Nevertheless, this new treatment option remains a source of optimism for relapsed and refractory DLBCL patients.
Primary Mediastinal Large B-Cell Lymphoma
Primary mediastinal large B-cell lymphoma (PMBCL) is a form of DLBCL arising in the mediastinum from the thymic B cell. It is an uncommon entity and has clinical and pathologic features distinct from systemic DLBCL.64 PMBCL accounts for 2% of all NHLs and about 7% of all DLBCL.20 It typically affects women in the third to fourth decade of life.
Presentation and Prognostic Features
PMBCL usually presents as a locally invasive anterior mediastinal mass, often with a superior vena cava syndrome which may or may not be clinically obvious.64 Other presentations include pericardial tamponade, thrombosis of neck veins, and acute airway obstruction. About 80% of patients present with bulky (> 10 cm) stage I or II disease,65 with distant spread uncommon on presentation. Morphologically and on GEP, PMBL has a profile more similar to classical Hodgkin lymphoma (cHL) than non-mediastinal DLBCL.66 PMBL is distinguished from cHL by immunophenotyping: unlike cHL, PMBCL has pan B cell markers, rarely expresses CD15, and has weak CD30.
Poor prognostic features in PMBCL are Eastern Cooperative Oncology Group (ECOG) performance status greater than 2, pericardial effusion, bulky disease, and elevated serum LDH. The diagnosis of PMBCL can be difficult because the tumor is often encased with extensive fibrosis and necrosis. As a result, a needle biopsy may not yield sufficient tissue, thus making a surgical biopsy often the only viable way to obtain sufficient tissue.
Treatment
Early series suggested that PMBCL is unusually aggressive, with a poor prognosis.67 This led to studies using more aggressive chemotherapy regimens (often in combination with mediastinal radiation) as well as upfront auto-HCT.68–70 The addition of rituximab to treatment regimens significantly improved outcomes in PMBCL. For example, a subgroup analysis of the PMBCL patients in the MinT trial revealed a 3-year event-free survival (EFS) of 78%71 when rituximab was combined with CHOP. Because of previous reports demonstrating radiosensitivity of PMBL, radiation was traditionally sequenced into treatment regimens for PMBL. However, this is associated with higher long-term toxicities, often a concern in PMBCL patients given that the disease frequently affects younger females, and given that breast tissue will be in the radiation field. For patients with a strong personal or family history of breast cancer or cardiovascular disease, these concerns are even more significant. More recently, the DA-EPOCH-R regimen has been shown to produce very high rates (80%–90%) of long-term DFS, without the need for mediastinal radiation in most cases.72,73 For patients receiving R-CHOP, consolidation with mediastinal radiation is still commonly given. This approach also leads to high rates of long-term remission and, although utilizing mediastinal radiation, allows for less intensive chemotherapy. Determining which approach is most appropriate for an individual patient requires an assessment of the risks of each treatment option for that patient. A randomized trial by the International Extranodal Lymphoma Study Group (IELSG37) is evaluating whether RT may be safely omitted in PMBCL patients who achieve a complete metabolic response after R-CHOP.
Most relapses of PMBCL occur within the first 1 to 2 years and often present with extranodal disease in various organs. For those with relapsed or refractory disease, high-dose chemotherapy followed by auto-HCT provides 5-year survival rates of 50% to 80%.74–76 In a phase 1b trial evaluating the role of pembrolizumab in relapsed/refractory patients (KEYNOTE-13), 7 of 17 PMBCL patients achieved responses, with an additional 6 demonstrating stable disease.77 This provides an additional option for patients who might be too weak to undergo auto-HCT or for those who relapse following auto-HCT.
Mantle Cell Lymphoma
The name mantle cell lymphoma (MCL) is based on the presumed normal cell counterpart to MCL, which is believed to be found in the mantle zone surrounding germinal center follicles. It represents approximately 6% of all NHL cases in the United States and Europe.78 MCL occurs at a median age of 63 to 68 years and has a male predominance.
Presentation and Prognostic Features
Patients can present with a broad spectrum of clinical features, and most patients (70%) present with advanced disease.79 Up to one third of patients have B symptoms, with most demonstrating lymphadenopathy and bone marrow involvement. Approximately 25% present with extranodal disease as the primary presentation (eg, GI tract, pleura, breast, or orbits). MCL can involve any part of the GI tract and often presents as polypoid lesions.
Histologically, the pattern of MCL may be diffuse, nodular, mantle zone, or a combination of the these; morphologically, MCL can range from small, more irregular lymphocytes to lymphoblast-like cells. Blastoid and pleomorphic variants of MCL have a higher proliferation index and a more aggressive clinical course than other variants. MCL is characterized by the expression of pan B cell antigens (CD19+, CD20+) with coexpression of the T-cell antigen CD5, lack of CD23 expression, and nuclear expression of cyclin D1. Nuclear staining for cyclin D1 is present in more than 98% of cases.80 In rare cases, CD5 or cyclin D1 may be negative.80 Most MCL cases have a unique translocation that fuses the immunoglobulin heavy chain gene promoter (14q32) to the promoter of the BCL-1 gene (11q13), which encodes the cyclin D1 protein. This translocation is not unique to MCL and can be present in multiple myeloma as well. Interestingly, cyclin D1 is overproduced in cases lacking t(11:14), likely from other point mutations resulting in its overexpression.81 Cyclin D1–negative tumors overexpress cyclin D2 or D3, with no apparent difference in clinical behavior or outcome.82 In cyclin D1–negative cases, SOX11 expression may help with diagnosis.83 A proliferation rate greater than 30% (as measured by Ki-67 staining), low SOX11 expression, and presence of p53 mutations have all been associated with adverse outcome.
In a minority of cases, MCL follows an indolent clinical course. For the remainder, however, MCL is an aggressive disease that generally requires treatment soon after diagnosis. When initially described in the 1980s and 1990s, treatment of MCL was characterized by low complete response rates, short durations of remission, repeated recurrences, and a median survival in the 2- to 5-year range.84 In recent years, intensive regimens incorporating rituximab and high-dose cytarabine with or without auto-HCT have been developed and are associated with high complete response rates and median duration of first remission in the 6- to 9-year range.85–87 Several prognostic indices have been applied to patients with MCL, including the IPI, the Follicular Lymphoma International Prognostic Index , and the Mantle Cell Lymphoma International Prognostic Index (MIPI). The MIPI was originally described based on a cohort from the period 1996 to 2004,88 and subsequently confirmed in a separate cohort of 958 patients with MCL treated on prospective trials between 2004 and 2010.89 The MIPI score can identify 3 risk groups with significant survival differences (83%, 63%, and 34% survival at 5 years). A refined version of the MIPI score, the combined MIPI or MIPI-c, incorporates proliferation rate and is better able to stratify patients.90 The blastoid variant of MCL follows a more aggressive clinical course and is associated with a high proliferation rate, shorter remissions, and a higher rate of CNS involvement.91
In most patients, MCL is an aggressive disease with a short OS without treatment. A subset of patients may have a more indolent course,92 but unfortunately reliable factors that identify this group at the time of diagnosis are not available. Pretreatment evaluation is as with other lymphomas, with lumbar puncture and MRI of the brain also recommended for patients with the blastoid variant. For those presenting with GI symptoms, endoscopy is recommended as part of the initial evaluation as well.
Treatment
First-line Therapy
For patients under age 65 to 70 years with a good performance status and few comorbidities, an intensive induction regimen (such as R-CHOP/R-DHAP, Maxi-R-CHOP/R-araC, or R-DHAP) followed by consolidation with auto-HCT is commonly given, with a goal of achieving a durable (6–9 year) first remission.87,93,94 Auto-HCT is now routinely followed by 3 years of maintenance rituximab based on the survival benefit seen in the recent LYSA trial.93 At many centers, auto-HCT in first remission is a standard of care, with the greatest benefit seen in patients who have achieved a complete remission with no more than 2 lines of chemotherapy.95 However, there remains some controversy about whether all patients truly benefit from auto-HCT in first remission, and current research efforts are focused on identifying patients most likely to benefit from auto-HCT and incorporation of new agents into first-line regimens. For patients who are not candidates for auto-HCT, bendamustine plus rituximab (BR) or R-CHOP alone or followed by maintenance rituximab is a reasonable approach.96 Based on the StiL and BRIGHT trials, BR seems to have less toxicity and higher rates of response with no difference in OS when compared to R-CHOP.97,98
In summary, dose-intense induction chemotherapy with consolidative auto-HCT results in high rates of long-term remission and can be considered in MCL patients who lack significant comorbidities and who understand the risks and benefits of this approach. For other patients, the less aggressive frontline approaches are more appropriate.
Relapsed/Refractory Disease
Despite initial high response rates, most patients with MCL will eventually relapse. For example, most patients given CHOP or R-CHOP alone as first-line therapy will relapse within 2 years.99 In recent years, a number of therapies have emerged for relapsed/refractory MCL; however, the optimal sequencing of these is unclear. FDA-approved options for relapsed/refractory MCL include the proteasome inhibitor bortezomib,100,101 the BTK inhibitors ibrutinib102,103 and acalabrutinib,104 and the immunomodulatory agent lenalidomide.105
Auto-HCT can be considered for patients who did not undergo auto-HCT as part of first-line therapy and who had a reasonably long first remission.95 Allo-HCT has curative potential in MCL with good evidence of a graft-versus-lymphoma effect. With a matched related or matched unrelated donor, the chance for treatment-related mortality is 15% to 25% at 1 to 2 years, with a 50% to 60% chance for long-term PFS. However, given the risk of treatment-related mortality and graft-versus-host disease, this option is typically reserved for patients with early relapse after auto-HCT, multiple relapses, or relatively chemotherapy-unresponsive disease.95,106 A number of clinical trials for relapsed/refractory MCL are ongoing, and participation in these is encouraged whenever possible.
Burkitt Lymphoma
Burkitt lymphoma is a rare, aggressive and highly curable subtype of NHL. It can occur at any age, although peak incidence is in the first decade of life. There are 3 distinct clinical forms of Burkitt lymphoma.107 The endemic form is common in African children and commonly involves the jaw and kidneys. The sporadic (nonendemic) form accounts for 1% to 2% of all lymphomas in the United States and Western Europe and usually has an abdominal presentation. The immunodeficiency-associated form is commonly seen in HIV patients with a relatively preserved CD4 cell count.
Patients typically present with rapidly growing masses and tumor lysis syndrome. CNS and bone marrow involvement are common. Burkitt lymphoma cells are high-grade, rapidly proliferating medium-sized cells with a monomorphic appearance. Biopsies show a classic histological appearance known as a “starry sky pattern” due to benign macrophages engulfing debris resulting from apoptosis. It is derived from a germinal center B cell and has distinct oncogenic pathways. Translocations such as t(8;14), t(2;8) or t(8;22) juxtapose the MYC locus with immunoglobulin heavy or light chain loci and result in MYC overexpression. Burkitt lymphoma is typically CD10-positive and BCL-2-negative, with a MYC translocation and a proliferation rate greater than 95%.
With conventional NHL regimens, Burkitt lymphoma had a poor prognosis, with complete remission in the 30% to 70% range and low rates of long-term remission. With the introduction of short-term, dose-intensive, multiagent chemotherapy regimens (adapted from pediatric acute lymphoblastic leukemia [ALL] regimens), the complete remission rate improved to 60% to 90%.107 Early stage disease (localized or completely resected intra-abdominal disease) can have complete remission rates of 100%, with 2- to 5-year freedom-from-progression rates of 95%. CNS prophylaxis, including high-dose methotrexate, high-dose cytarabine, and intrathecal chemotherapy, is a standard component of Burkitt lymphoma regimens (CNS relapse rates can reach 50% without prophylactic therapy). Crucially, relapse after 1 to 2 years is very rare following complete response to induction therapy. Classically, several intensive regimens have been used for Burkitt lymphoma. In recent years, the most commonly used regimens have been the modified Magrath regimen of R-CODOX-M/IVAC and R-hyperCVAD. DA-EPOCH-R has also been used, typically for older, more frail, or HIV-positive patients. However, at the American Society of Hematology 2017 annual meeting, results from the NCI 9177 trial were presented which validated, in a prospective multi-center fashion, the use of DA-EPOCH-R in all Burkitt lymphoma patients.108 In NCI 9177, low-risk patients (defined as normal LDH, ECOG performance score 0 or 1, ≤ stage II, and no tumor lesion > 7 cm) received 2 cycles of DA-EPOCH-R without intrathecal therapy followed by PET. If interim PET was negative, low-risk patients then received 1 more cycle of DA-EPOCH-R. High-risk patients with negative brain MRI and CSF cytology/flow cytometry received 2 cycles of DA-EPOCH-R with intrathecal therapy (2 doses per cycle) followed by PET. Unless interim PET showed progression, high-risk patients received 4 additional cycles of DA-EPOCH-R including methotrexate 12 mg intrathecally on days 1 and 5 (8 total doses). With a median follow-up of 36 months, this regimen resulted in an EFS of 85.7%. As expected, patients with CNS, marrow, or peripheral blood involvement fared worse. For those without CNS, marrow, or peripheral blood involvement, the results were excellent, with an EFS of 94.6% compared to 62.8% for those with CNS, bone marrow, or blood involvement at diagnosis.
Although no standard of care has been defined, patients with relapsed/refractory Burkitt lymphoma are often given standard second-line aggressive NHL regimens (eg, R-ICE); for those with chemosensitive disease, auto- or allo-HCT is often pursued, with long-term remissions possible following HCT.109
Lymphoblastic Lymphoma
Lymphoblastic lymphoma (LBL) is a rare disease postulated to arise from precursor B or T lymphoblasts at varying stages of differentiation. Accounting for approximately 2% of all NHLs, 85% to 90% of all cases have a T-cell phenotype, while B-cell LBL comprises approximately 10% to 15% of cases. LBL and ALL are thought to represent the same disease entity, but LBL has been arbitrarily defined as cases with lymph node or mediastinal disease. Those with significant (> 25%) bone marrow or peripheral blood involvement are classified as ALL.
Precursor T-cell LBL patients are usually adolescent and young males who commonly present with a mediastinal mass and peripheral lymphadenopathy. Precursor B-cell LBL patients are usually older (median age 39 years) with peripheral lymphadenopathy and extranodal involvement. Mediastinal involvement with B-cell LBL is uncommon, and there is no male predominance. LBL has a propensity for dissemination to the bone marrow and CNS.
Morphologically, the tumor cells are medium sized, with a scant cytoplasm and finely dispersed chromatin. Mitotic features and apoptotic bodies are present since it is a high-grade malignancy. The lymphoblasts are typically positive for CD7 and either surface or cytoplasmic CD3. Terminal deoxynucleotidyl transferase expression is a defining feature. Other markers such as CD19, CD22, CD20, CD79a, CD45, and CD10 are variably expressed. Poor prognostic factors in T-cell LBL are female gender, age greater than 35 years, complex cytogenetics, and lack of a matched sibling donor.
Regimens for LBL are based on dose-dense, multi-agent protocols used in ALL. Most of these regimens are characterized by intensive remission-induction chemotherapy, CNS prophylaxis, a phase of consolidation therapy, and a prolonged maintenance phase, often lasting for 12 to 18 months with long-term DFS rates of 40% to 70%.110,111 High-dose therapy with auto-HCT or allo-HCT in first complete response has been evaluated in an attempt to reduce the incidence of relapse.112 However, the intensity of primary chemotherapy appears to be a stronger determinant of long-term survival than the use of HCT as consolidation. As a result, HCT is not routinely applied to patients in first complete remission following modern induction regimens. After relapse, prognosis is poor, with median survival rates of 6 to 9 months with conventional chemotherapy, although long-term survival rates of 30% and 20%, respectively, are reported after HCT in relapsed and primary refractory disease.113
Treatment options in relapsed disease are limited. Nelarabine can produce responses in up to 40% of relapsed/refractory LBL/ALL patients.114
Summary
Aggressive NHLs are characterized by rapid clinical progression without therapy. However, a significant proportion of patients are cured with appropriate combination chemotherapy or combined modality (chemotherapy + RT) regimens. In contrast, the indolent lymphomas have a relatively good prognosis (median survival of 10 years or longer) but usually are not curable in advanced clinical stages. Overall 5-year survival for aggressive NHLs with current treatment is approximately 50% to 60%, with relapses typically occurring within the first 5 years. Treatment strategies for relapsed patients offer some potential for cure; however, clinical trial participation should be encouraged whenever possible to investigate new approaches for improving outcomes in this patient population.
1. Swerdlow, SH, Campo E, Harris NL, et al. WHO classification of tumours of haematopoietic and lymphoid tissues. Revised 4th edition. Lyon, France: World Health Organization; 2017.
2. Surveillance, Epidemiology, and End Results (SEER) Program. www.seer.cancer.gov. Research Data 2017.
3. Boffetta P, de Vocht F. Occupation and the risk of non-Hodgkin lymphoma. Cancer Epidemiol Biomarkers Prev 2007;16:369–72.
4. Bower M. Acquired immunodeficiency syndrome-related systemic non-Hodgkin’s lymphoma. Br J Haematol 2001;112:863–73.
5. Ekstrom Smedby K, Vajdic CM, Falster M, et al. Autoimmune disorders and risk of non-Hodgkin lymphoma subtypes: a pooled analysis within the InterLymph Consortium. Blood 2008;111:4029–38.
6. Clarke CA, Morton LM, Lynch C, et al. Risk of lymphoma subtypes after solid organ transplantation in the United States. Br J Cancer 2013;109:280–8.
7. Wang SS, Slager SL, Brennan P, et al. Family history of hematopoietic malignancies and risk of non-Hodgkin lymphoma (NHL): a pooled analysis of 10 211 cases and 11 905 controls from the International Lymphoma Epidemiology Consortium (InterLymph). Blood 2007;109:3479–88.
8. Dong C, Hemminki K. Second primary neoplasms among 53 159 haematolymphoproliferative malignancy patients in Sweden, 1958–1996: a search for common mechanisms. Br J Cancer 2001;85:997–1005.
9. Hummel M, Anagnostopoulos I, Korbjuhn P, Stein H. Epstein-Barr virus in B-cell non-Hodgkin’s lymphomas: unexpected infection patterns and different infection incidence in low- and high-grade types. J Pathol 1995;175:263–71.
10. Cesarman E, Chang Y, Moore PS, et al. Kaposi’s sarcoma-associated herpesvirus-like DNA sequences in AIDS-related body-cavity-based lymphomas. N Engl J Med 1995;332:1186–91.
11. Viswanatha DS, Dogan A. Hepatitis C virus and lymphoma. J Clin Pathol 2007;60:1378–83.
12. Engels EA, Cho ER, Jee SH. Hepatitis B virus infection and risk of non-Hodgkin lymphoma in South Korea: a cohort study. Lancet Oncol 2010;11:827–34.
13. Marcucci F, Mele A. Hepatitis viruses and non-Hodgkin lymphoma: epidemiology, mechanisms of tumorigenesis, and therapeutic opportunities. Blood 2011;117:1792–8.
14. Cheson BD, Fisher RI, Barrington SF, et al. Recommendations for initial evaluation, staging, and response assessment of Hodgkin and non-Hodgkin lymphoma: the Lugano classification. J Clin Oncol 2014;32:3059–68.
15. Rosenwald A, Wright G, Chan WC, et al. The use of molecular profiling to predict survival after chemotherapy for diffuse large-B-cell lymphoma. N Engl J Med 2002;346:1937–47.
16. Teras LR, DeSantis CE, Cerhan JR, et al. 2016 US lymphoid malignancy statistics by World Health Organization subtypes. CA Cancer J Clin 2016;66:443–59.
17. Morton LM, Wang SS, Devesa SS, et al. Lymphoma incidence patterns by WHO subtype in the United States, 1992-2001. Blood 2006;107:265–76.
18. Møller MB, Pedersen NT, Christensen BE. Diffuse large B-cell lymphoma: clinical implications of extranodal versus nodal presentation--a population-based study of 1575 cases. Br J Haematol 2004;124:151–9.
19. Armitage JO, Weisenburger DD. New approach to classifying non-Hodgkin’s lymphomas: clinical features of the major histologic subtypes. Non-Hodgkin’s Lymphoma Classification Project. J Clin Oncol 1998;16:2780–95.
20. A clinical evaluation of the International Lymphoma Study Group classification of non-Hodgkin’s lymphoma. The Non-Hodgkin’s Lymphoma Classification Project. Blood 1997;89:3909–18.
21. Sehn LH, Scott DW, Chhanabhai M, et al. Impact of concordant and discordant bone marrow involvement on outcome in diffuse large B-cell lymphoma treated with R-CHOP. J Clin Oncol 2011;29:1452–7.
22. Fisher DE, Jacobson JO, Ault KA, Harris NL. Diffuse large cell lymphoma with discordant bone marrow histology. Clinical features and biological implications. Cancer 1989;64:1879–87.
23. Yao Z, Deng L, Xu-Monette ZY, et al. Concordant bone marrow involvement of diffuse large B-cell lymphoma represents a distinct clinical and biological entity in the era of immunotherapy. Leukemia 2018;32:353–63.
24. Gascoyne RD, Adomat SA, Krajewski S, et al. Prognostic significance of Bcl-2 protein expression and Bcl-2 gene rearrangement in diffuse aggressive non-Hodgkin’s lymphoma. Blood 1997;90:244–51.
25. Skinnider BF, Horsman DE, Dupuis B, Gascoyne RD. Bcl-6 and Bcl-2 protein expression in diffuse large B-cell lymphoma and follicular lymphoma: correlation with 3q27 and 18q21 chromosomal abnormalities. Hum Pathol 1999;30:803–8.
26. Chisholm KM, Bangs CD, Bacchi CE, et al. Expression profiles of MYC protein and MYC gene rearrangement in lymphomas. Am J Surg Pathol 2015;39:294–303.
27. Zhou Z, Sehn LH, Rademaker AW, et al. An enhanced International Prognostic Index (NCCN-IPI) for patients with diffuse large B-cell lymphoma treated in the rituximab era. Blood 2014;123:837–42.
28. Hans CP, Weisenburger DD, Greiner TC, et al. Confirmation of the molecular classification of diffuse large B-cell lymphoma by immunohistochemistry using a tissue microarray. Blood 2004;103:275–82.
29. Horn H, Ziepert M, Becher C, et al. MYC status in concert with BCL2 and BCL6 expression predicts outcome in diffuse large B-cell lymphoma. Blood 2013;121:2253–63.
30. Barrans S, Crouch S, Smith A, et al. Rearrangement of MYC is associated with poor prognosis in patients with diffuse large B-cell lymphoma treated in the era of rituximab. J Clin Oncol 2010;28:3360–5.
31. Hu S, Xu-Monette ZY, Tzankov A, et al. MYC/BCL2 protein coexpression contributes to the inferior survival of activated B-cell subtype of diffuse large B-cell lymphoma and demonstrates high-risk gene expression signatures: a report from The International DLBCL Rituximab-CHOP Consortium Program. Blood 2013;121:4021–31.
32. Green TM, Young KH, Visco C, et al. Immunohistochemical double-hit score is a strong predictor of outcome in patients with diffuse large B-cell lymphoma treated with rituximab plus cyclophosphamide, doxorubicin, vincristine, and prednisone. J Clin Oncol 2012;30:3460–7.
33. Fisher RI, Gaynor ER, Dahlberg S, et al. Comparison of a standard regimen (CHOP) with three intensive chemotherapy regimens for advanced non-Hodgkin’s lymphoma. N Engl J Med 1993;328:1002–6.
34. Pfreundschuh M, Kuhnt E, Trümper L, et al. CHOP-like chemotherapy with or without rituximab in young patients with good-prognosis diffuse large-B-cell lymphoma: 6-year results of an open-label randomised study of the MabThera International Trial (MInT) Group. Lancet Oncol 2011;12:1013–22.
35. Coiffier B, Lepage E, Brière J, et al. CHOP chemotherapy plus rituximab compared with CHOP alone in elderly patients with diffuse large-B-cell lymphoma. N Engl J Med 2002;346:235–42.
36. Persky DO, Unger JM, Spier CM, et al. Phase II study of rituximab plus three cycles of CHOP and involved-field radiotherapy for patients with limited-stage aggressive B-cell lymphoma: Southwest Oncology Group study 0014. J Clin Oncol 2008;26:2258–63.
37. Lamy T, Damaj G, Soubeyran P, et al. R-CHOP 14 with or without radiotherapy in nonbulky limited-stage diffuse large B-cell lymphoma. Blood 2018;131:174–81.
38. Peyrade F, Jardin F, Thieblemont C, et al. Attenuated immunochemotherapy regimen (R-miniCHOP) in elderly patients older than 80 years with diffuse large B-cell lymphoma: a multicentre, single-arm, phase 2 trial. Lancet Oncol 2011;12:460–8.
39. Wilson WH, sin-Ho J, Pitcher BN, et al. Phase III randomized study of R-CHOP versus DA-EPOCH-R and molecular analysis of untreated diffuse large B-cell lymphoma: CALGB/Alliance 50303. Blood 2016;128:469 LP-469. 38.
40. Vitolo U, Trne˘ný M, Belada D, et al. Obinutuzumab or rituximab plus cyclophosphamide, doxorubicin, vincristine, and prednisone in previously untreated diffuse large B-cell lymphoma. J Clin Oncol 2017;35:3529–37.
41. Leonard JP, Kolibaba KS, Reeves JA, et al. Randomized phase II study of R-CHOP with or without bortezomib in previously untreated patients with non-germinal center B-cell-like diffuse large B-cell lymphoma. J Clin Oncol 2017;35:3538–46.
42. Nowakowski GS, LaPlant B, Macon WR, et al. Lenalidomide combined with R-CHOP overcomes negative prognostic impact of non-germinal center B-cell phenotype in newly diagnosed diffuse large B-Cell lymphoma: a phase II study. J Clin Oncol 2015;33:251–7.
43. Younes A, Thieblemont C, Morschhauser F, et al. Combination of ibrutinib with rituximab, cyclophosphamide, doxorubicin, vincristine, and prednisone (R-CHOP) for treatment-naive patients with CD20-positive B-cell non-Hodgkin lymphoma: a non-randomised, phase 1b study. Lancet Oncol 2014;15:1019–26.
44. Younes A, Zinzani PL, Sehn LH, et al. A randomized, double-blind, placebo-controlled phase 3 study of ibrutinib in combination with rituximab, cyclophosphamide, doxorubicin, vincristine, and prednisone (R-CHOP) in subjects with newly diagnosed nongerminal center B-cell subtype of diffuse large B-cell lymphoma (DLBCL). J Clin Oncol 2014;32(15_suppl):TPS8615.
45. Delarue R, Tilly H, Mounier N, et al. Dose-dense rituximab-CHOP compared with standard rituximab-CHOP in elderly patients with diffuse large B-cell lymphoma (the LNH03-6B study): a randomised phase 3 trial. Lancet Oncol 2013;14:525–33.
46. Leppä S, Fayad LE, Lee J-J, et al. A phase III study of enzastaurin in patients with high-risk diffuse large B cell lymphoma following response to primary treatment: the Prelude trial. Blood 2013;122:371 LP-371.
47. Witzig TE, Tobinai K, Rigacci L, et al. Adjuvant everolimus in high-risk diffuse large B-cell lymphoma: final results from the PILLAR-2 randomized phase III trial. Ann Oncol 2018;29:707–14.
48. Strehl J, Mey U, Glasmacher A, et al. High-dose chemotherapy followed by autologous stem cell transplantation as first-line therapy in aggressive non-Hodgkin’s lymphoma: a meta-analysis. Haematologica 2003;88:1304–15.
49. Stiff PJ, Unger JM, Cook JR, et al. Autologous transplantation as consolidation for aggressive non-Hodgkin’s lymphoma. N Engl J Med 2013;369:1681–90.
50. Oki Y, Noorani M, Lin P, et al. Double hit lymphoma: the MD Anderson Cancer Center clinical experience. Br J Haematol 2014;166:891–901.
51. Petrich AM, Gandhi M, Jovanovic B, et al. Impact of induction regimen and stem cell transplantation on outcomes in double-hit lymphoma: a multicenter retrospective analysis. Blood 2014;124:2354–61.
52. Howlett C, Snedecor SJ, Landsburg DJ, et al. Front-line, dose-escalated immunochemotherapy is associated with a significant progression-free survival advantage in patients with double-hit lymphomas: a systematic review and meta-analysis. Br J Haematol 2015;170:504–14.
53. Landsburg DJ, Falkiewicz MK, Maly J, et al. Outcomes of patients with double-hit lymphoma who achieve first complete remission. J Clin Oncol 2017;35:2260–7.
54. Schmitz N, Zeynalova S, Nickelsen M, et al. CNS International Prognostic Index: a risk model for CNS relapse in patients with diffuse large B-cell lymphoma treated with R-CHOP. J Clin Oncol 2016;34:3150–6.
55. Abramson JS, Hellmann M, Barnes JA, et al. Intravenous methotrexate as central nervous system (CNS) prophylaxis is associated with a low risk of CNS recurrence in high-risk patients with diffuse large B-cell lymphoma. Cancer 2010;116:4283–90.
56. Dunleavy K, Roschewski M, Abramson JS, et al. Risk-adapted therapy in adults with Burkitt lymphoma: updated results of a multicenter prospective phase II study of DA-EPOCH-R. Hematol Oncol 2017;35(S2):133–4.
57. Damaj G, Ivanoff S, Coso D, et al. Concomitant systemic and central nervous system non-Hodgkin lymphoma: the role of consolidation in terms of high dose therapy and autologous stem cell transplantation. A 60-case retrospective study from LYSA and the LOC network. Haematologica 2015;100:1199–206.
58. Thieblemont C, Briere J, Mounier N, et al. The germinal center/activated B-cell subclassification has a prognostic impact for response to salvage therapy in relapsed/refractory diffuse large B-cell lymphoma: a bio-CORAL study. J Clin Oncol 2011;29:4079–87.
59. Philip T, Guglielmi C, Hagenbeek A, et al. Autologous bone marrow transplantation as compared with dalvage vhemotherapy in relapses of chemotherapy-densitive non-Hodgkin’s lymphoma. N Engl J Med 1995;333:1540–5.
60. Hamadani M, Hari PN, Zhang Y, et al. Early failure of frontline rituximab-containing chemo-immunotherapy in diffuse large B cell lymphoma does not predict futility of autologous hematopoietic cell transplantation. Biol Blood Marrow Transplant 2014;20:1729–36.
61. Costa LJ, Maddocks K, Epperla N, et al. Diffuse large B-cell lymphoma with primary treatment failure: Ultra-high risk features and benchmarking for experimental therapies. Am J Hematol 2017;92:e24615.
62. Fenske TS, Ahn KW, Graff TM, et al. Allogeneic transplantation provides durable remission in a subset of DLBCL patients relapsing after autologous transplantation. Br J Haematol 2016;174:235–48.
63. Neelapu SS, Locke FL, Bartlett NL, et al. Axicabtagene ciloleucel CAR T-cell therapy in refractory large B-cell lymphoma. N Engl J Med 2017;377:2531–44.
64. van Besien K, Kelta M, Bahaguna P. Primary mediastinal B-cell lymphoma: a review of pathology and management. J Clin Oncol 2001;19:1855–64.
65. Savage KJ, Al-Rajhi N, Voss N, et al. Favorable outcome of primary mediastinal large B-cell lymphoma in a single institution: the British Columbia experience. Ann Oncol Off J Eur Soc Med Oncol 2006;17:123–30.
66. Rosenwald A, Wright G, Leroy K, et al. Molecular diagnosis of primary mediastinal B cell lymphoma identifies a clinically favorable subgroup of diffuse large B cell lymphoma related to Hodgkin lymphoma. J Exp Med 2003;198:851–62.
67. Lavabre-Bertrand T, Donadio D, Fegueux N, et al. A study of 15 cases of primary mediastinal lymphoma of B-cell type. Cancer 1992;69:2561–6.
68. Lazzarino M, Orlandi E, Paulli M, et al. Treatment outcome and prognostic factors for primary mediastinal (thymic) B-cell lymphoma: a multicenter study of 106 patients. J Clin Oncol 1997;15:1646–53.
69. Zinzani PL, Martelli M, Magagnoli M, et al. Treatment and clinical management of primary mediastinal large B-cell lymphoma with sclerosis: MACOP-B regimen and mediastinal radiotherapy monitored by (67)Gallium scan in 50 patients. Blood 1999;94:3289–93.
70. Todeschini G, Secchi S, Morra E, et al. Primary mediastinal large B-cell lymphoma (PMLBCL): long-term results from a retrospective multicentre Italian experience in 138 patients treated with CHOP or MACOP-B/VACOP-B. Br J Cancer 2004;90:372–6.
71. Rieger M, Osterborg A, Pettengell R, et al. Primary mediastinal B-cell lymphoma treated with CHOP-like chemotherapy with or without rituximab: results of the Mabthera International Trial Group study. Ann Oncol Off J Eur Soc Med Oncol 2011;22:664–70.
72. Shah NN, Szabo A, Huntington SF, et al. R-CHOP versus dose-adjusted R-EPOCH in frontline management of primary mediastinal B-cell lymphoma: a multi-centre analysis. Br J Haematol 2018;180:534–44.
73. Dunleavy K, Pittaluga S, Maeda LS, et al. Dose-adjusted EPOCH-rituximab therapy in primary mediastinal B-cell lymphoma. N Engl J Med 2013;368:1408–16.
74. Aoki T, Shimada K, Suzuki R, et al. High-dose chemotherapy followed by autologous stem cell transplantation for relapsed/refractory primary mediastinal large B-cell lymphoma. Blood Cancer J 2015;5:e372–e372.
75. Sehn LH, Antin JH, Shulman LN, et al. Primary diffuse large B-cell lymphoma of the mediastinum: outcome following high-dose chemotherapy and autologous hematopoietic cell transplantation. Blood 1998;91:717–23.
76. Kuruvilla J, Pintilie M, Tsang R, et al. Salvage chemotherapy and autologous stem cell transplantation are inferior for relapsed or refractory primary mediastinal large B-cell lymphoma compared with diffuse large B-cell lymphoma. Leuk Lymphoma 2008;49:1329–36.
77. Zinzani PL, Ribrag V, Moskowitz CH, et al. Safety and tolerability of pembrolizumab in patients with relapsed/refractory primary mediastinal large B-cell lymphoma. Blood 2017;130:267–70.
78. Smith A, Howell D, Patmore R, et al. Incidence of haematological malignancy by sub-type: a report from the Haematological Malignancy Research Network. Br J Cancer 2011;105:1684–92.
79. Argatoff LH, Connors JM, Klasa RJ, et al. Mantle cell lymphoma: a clinicopathologic study of 80 cases. Blood 1997;89:2067–78.
80. Zukerberg LR, Yang WI, Arnold A, Harris NL. Cyclin D1 expression in non-Hodgkin’s lymphomas. Detection by immunohistochemistry. Am J Clin Pathol 1995;103:756–60.
81. Wiestner A, Tehrani M, Chiorazzi M, et al. Point mutations and genomic deletions in CCND1 create stable truncated cyclin D1 mRNAs that are associated with increased proliferation rate and shorter survival. Blood 2007;109:4599–606.
82. Fu K, Weisenburger DD, Greiner TC, et al. Cyclin D1-negative mantle cell lymphoma: a clinicopathologic study based on gene expression profiling. Blood 2005;106:4315–21.
83. Mozos A, Royo C, Hartmann E, et al. SOX11 expression is highly specific for mantle cell lymphoma and identifies the cyclin D1-negative subtype. Haematologica 2009;94:1555–62.
84. Norton AJ, Matthews J, Pappa V, et al. Mantle cell lymphoma: Natural history defined in a serially biopsied population over a 20-year period. Ann Oncol 1995;6:249–56.
85. Chihara D, Cheah CY, Westin JR, et al. Rituximab plus hyper-CVAD alternating with MTX/Ara-C in patients with newly diagnosed mantle cell lymphoma: 15-year follow-up of a phase II study from the MD Anderson Cancer Center. Br J Haematol 2016;172:80–8.
86. Delarue R, Haioun C, Ribrag V, et al. CHOP and DHAP plus rituximab followed by autologous stem cell transplantation in mantle cell lymphoma: a phase 2 study from the Groupe d’Etude des Lymphomes de l’Adulte. Blood 2013;121:48–53.
87. Eskelund CW, Kolstad A, Jerkeman M, et al. 15-year follow-up of the Second Nordic Mantle Cell Lymphoma trial (MCL2): prolonged remissions without survival plateau. Br J Haematol 2016;175:410–8.
88. Hoster E, Dreyling M, Klapper W, et al. A new prognostic index (MIPI) for patients with advanced-stage mantle cell lymphoma. Blood 2008;111:558–65.
89. Hoster E, Klapper W, Hermine O, et al. Confirmation of the mantle-cell lymphoma International Prognostic Index in randomized trials of the European Mantle-Cell Lymphoma Network. J Clin Oncol 2014;32:1338–46.
90. Hoster E, Rosenwald A, Berger F, et al. Prognostic value of Ki-67 index, cytology, and growth pattern in mantle-cell lymphoma: Results from randomized trials of the European Mantle Cell Lymphoma Network. J Clin Oncol 2016;34:1386–94.
91. Bernard M, Gressin R, Lefrère F, et al. Blastic variant of mantle cell lymphoma: a rare but highly aggressive subtype. Leukemia 2001;15:1785–91.
92. Martin P, Chadburn A, Christos P, et al. Outcome of deferred initial therapy in mantle-cell lymphoma. J Clin Oncol 2009;27:1209–13.
93. Le Gouill S, Thieblemont C, Oberic L, et al. Rituximab after autologous stem-cell transplantation in mantle-cell lymphoma. N Engl J Med. 2017 Sep 28;377(13):1250–60.
94. Hermine O, Hoster E, Walewski J, et al. Addition of high-dose cytarabine to immunochemotherapy before autologous stem-cell transplantation in patients aged 65 years or younger with mantle cell lymphoma (MCL Younger): a randomised, open-label, phase 3 trial of the European Mantle Cell Lymphoma Network. Lancet 2016;388:565–75.
95. Fenske TS, Zhang M-J, Carreras J, et al. Autologous or reduced-intensity conditioning allogeneic hematopoietic cell transplantation for chemotherapy-sensitive mantle-cell lymphoma: analysis of transplantation timing and modality. J Clin Oncol 2014;32:273–81.
96. Kluin-Nelemans HC, Hoster E, Hermine O, et al. Treatment of older patients with mantle-cell lymphoma. N Engl J Med 2012;367:520–31.
97. Flinn IW, van der Jagt R, Kahl BS, et al. Randomized trial of bendamustine-rituximab or R-CHOP/R-CVP in first-line treatment of indolent NHL or MCL: the BRIGHT study. Blood 2014;123:2944–52.
98. Rummel MJ, Niederle N, Maschmeyer G, et al. Bendamustine plus rituximab versus CHOP plus rituximab as first-line treatment for patients with indolent and mantle-cell lymphomas: an open-label, multicentre, randomised, phase 3 non-inferiority trial. Lancet 2013;381:1203–10.
99. Lenz G, Dreyling M, Hoster E, et al. Immunochemotherapy with rituximab and cyclophosphamide, doxorubicin, vincristine, and prednisone significantly improves response and time to treatment failure, but not long-term outcome in patients with previously untreated mantle cell lymphoma: results of a prospective randomized trial of the German Low Grade Lymphoma Study Group (GLSG). J Clin Oncol 2005;23:1984–92.
100. Belch A, Kouroukis CT, Crump M, et al. A phase II study of bortezomib in mantle cell lymphoma: the National Cancer Institute of Canada Clinical Trials Group trial IND.150. Ann Oncol Off J Eur Soc Med Oncol 2007;18:116–21.
101. Fisher RI, Bernstein SH, Kahl BS, et al. Multicenter phase II study of bortezomib in patients with relapsed or refractory mantle cell lymphoma. J Clin Oncol 2006;24:4867–74.
102. Dreyling M, Jurczak W, Jerkeman M, et al. Ibrutinib versus temsirolimus in patients with relapsed or refractory mantle-cell lymphoma: an international, randomised, open-label, phase 3 study. Lancet 2016;387:770–8.
103. Wang ML, Rule S, Martin P, Goy A, et al. Targeting BTK with ibrutinib in relapsed or refractory mantle-cell lymphoma. N Engl J Med 2013;369:507–16.
104. Wang M, Rule S, Zinzani PL, et al. Acalabrutinib in relapsed or refractory mantle cell lymphoma (ACE-LY-004): a single-arm, multicentre, phase 2 trial. Lancet 2018;391:659–67.
105. Goy A, Sinha R, Williams ME, et al. Single-agent lenalidomide in patients with mantle-cell lymphoma who relapsed or progressed after or were refractory to bortezomib: phase II MCL-001 (EMERGE) study. J Clin Oncol 2013;31:3688–95.
106. Khouri IF, Lee M-S, Saliba RM, et al. Nonablative allogeneic stem-cell transplantation for advanced/recurrent mantle-cell lymphoma. J Clin Oncol 2003;21:4407–12.
107. Blum KA, Lozanski G, Byrd JC. Adult Burkitt leukemia and lymphoma. Blood 2004;104:3009–20.
108. Roschewski M, Dunleavy K, Abramson JS, et al. Risk-adapted therapy in adults with Burkitt lymphoma: results of NCI 9177, a multicenter prospective phase II study of DA-EPOCH-R. Blood American Society of Hematology;2017;130(Suppl 1):188.
109. Maramattom L V, Hari PN, Burns LJ, et al. Autologous and allogeneic transplantation for burkitt lymphoma outcomes and changes in utilization: a report from the center for international blood and marrow transplant research. Biol Blood Marrow Transplant 2013;19:173–9.
110. Zinzani PL, Bendandi M, Visani G, et al. Adult lymphoblastic lymphoma: clinical features and prognostic factors in 53 patients. Leuk Lymphoma 1996;23:577–82.
111. Thomas DA, O’Brien S, Cortes J, et al. Outcome with the hyper-CVAD regimens in lymphoblastic lymphoma. Blood 2004;104:1624–30.
112. Aljurf M, Zaidi SZA. Chemotherapy and hematopoietic stem cell transplantation for adult T-cell lymphoblastic lymphoma: current status and controversies. Biol Blood Marrow Transplant 2005;11:739–54.
113. Sweetenham JW, Santini G, Qian W, et al. High-dose therapy and autologous stem-cell transplantation versus conventional-dose consolidation/maintenance therapy as postremission therapy for adult patients with lymphoblastic lymphoma: results of a randomized trial of the European Group for Blood and Marrow Transplantation and the United Kingdom Lymphoma Group. J Clin Oncol 2001;19:2927–36.
114. Zwaan CM, Kowalczyk J, Schmitt C, et al. Safety and efficacy of nelarabine in children and young adults with relapsed or refractory T-lineage acute lymphoblastic leukaemia or T-lineage lymphoblastic lymphoma: results of a phase 4 study. Br J Haematol 2017;179:284–93.
Introduction
Non-Hodgkin lymphoma (NHL) comprises a wide variety of malignant hematologic disorders with varying clinical and biological features. The more than 60 separate NHL subtypes can be classified according to cell of origin (B cell versus T cell), anatomical location (eg, orbital, testicular, bone, central nervous system), clinical behavior (indolent versus aggressive), histological features, or cytogenetic abnormalities. Although various NHL classification schemes have been used over the years, the World Health Organization (WHO) classification is now widely accepted as the definitive pathologic classification system for lymphoproliferative disorders, incorporating morphologic, immunohistochemical, flow cytometric, cytogenetic, and molecular features.1 While the pathologic and molecular subclassification of NHL has become increasingly refined in recent years, from a management standpoint, classification based on clinical behavior remains very useful. This approach separates NHL subtypes into indolent versus aggressive categories. Whereas indolent NHLs may remain clinically insignificant for months to years, aggressive B-cell NHLs generally become life-threatening within weeks to months without treatment.
Epidemiology
Data from cancer registries show a steady, unexplainable increase in the incidence of NHL during the second half of the 20th century; the incidence has subsequently plateaued. There was a significant increase in NHL incidence between 1970 and 1995, which has been attributed in part to the HIV epidemic. More than 72,000 new cases of NHL were diagnosed in the United States in 2017, compared to just over 8000 cases of Hodgkin lymphoma, making NHL the sixth most common cancer in adult men and the fifth most common in adult women.2 NHL appears to occur more frequently in Western countries than in Asian populations.
Various factors associated with increased risk for B-cell NHL have been identified over the years, including occupational and environmental exposure to certain pesticides and herbicides,3 immunosuppression associated with HIV infection,4 autoimmune disorders,5 iatrogenically induced immune suppression in the post-transplant and other settings,6 family history of NHL,7 and a personal history of a prior cancer, including Hodgkin lymphoma and prior NHL.8 In terms of infectious agents associated with aggressive B-cell NHLs, Epstein-Barr virus (EBV) has a clear pathogenic role in Burkitt lymphoma, in many cases of post-transplant lymphoproliferative disorders, and in some cases of HIV-related aggressive B-cell lymphoma.9 Human herpesvirus-8 viral genomes have been found in virtually all cases of primary effusion lymphomas.10 Epidemiological studies also have linked hepatitis B and C to increased incidences of certain NHL subtypes,11–13 including primary hepatic diffuse large B-cell lymphoma (DLBCL). Similarly, Helicobacter pylori has been associated with gastric DLBCL.
Staging and Work-Up
A tissue biopsy is essential in the diagnosis and management of NHL. The most significant disadvantage of fine-needle aspiration cytology is the lack of histologic architecture. The optimal specimen is an excisional biopsy; when this cannot be performed, a core needle biopsy, ideally using a 16-gauge or larger caliber needle, is the next best choice.
The baseline tests appropriate for most cases of newly diagnosed aggressive B-cell NHL are listed in Table 1. Both hepatitis B and C have been associated with increased risk of NHL. In addition, there is a risk of hepatitis B reactivation following certain NHL therapies. A contrast-enhanced computed tomography (CT) scan in addition to positron emission tomography (PET) is useful to define the extent of disease in situations needing greater definition (eg, lymphadenopathy close to the bowel, cervical and supraclavicular nodal involvement, and lymphadenopathy causing thrombosis or compression of nearby structures).14 In cases where it is apparent that the patient has advanced stage disease (Ann Arbor stage III/IV) based on imaging, bone marrow biopsy is unlikely to alter the treatment plan. For such patients, if the complete blood count is unremarkable, deferral of bone marrow biopsy may be reasonable. For new cases of DLBCL, assessment for MYC translocation by fluorescence in situ hybridization (FISH) is recommended. If a MYC translocation is identified, then testing for BCL2 and BCL6 translocations by FISH should be performed.
Prior to the initiation of treatment, patients should always undergo a thorough cardiac and pulmonary evaluation, especially if the patient will be treated with an anthracycline or mediastinal irradiation. Central nervous system (CNS) evaluation with magnetic resonance imaging (MRI) and lumbar puncture is essential if there are neurological signs or symptoms. In addition, certain anatomical sites including the testicles, paranasal sinuses, kidney, adrenal glands, and epidural space have been associated with increased involvement of the CNS and may warrant MRI evaluation and lumbar puncture. Certain NHL subtypes like Burkitt lymphoma, high-grade NHL with translocations of MYC and BCL-2 or BCL-6 (double-hit lymphoma), blastoid mantle cell lymphoma, and lymphoblastic lymphoma have a high risk of CNS involvement, and patients with these subtypes need CNS evaluation.
The Lugano classification is used to stage patients with NHL.14 This classification is based on the Ann Arbor staging system and uses the distribution and number of tumor sites to stage disease. In general, this staging system in isolation is of limited value in predicting survival after treatment. However, the Ann Arbor stage does have prognostic impact when incorporated into risk scoring systems such as the International Prognostic Index (IPI). In clinical practice, the Ann Arbor stage is useful primarily to determine eligibility for localized therapy approaches. The absence or presence of systemic symptoms such as fevers, drenching night sweats, or weight loss (> 10% of baseline over 6 months or less) is designated by A or B, respectively.
Diffuse Large B-Cell Lymphoma
DLBCL is the most common lymphoid neoplasm in adults, accounting for about 25% of all NHL cases.2 It is increasingly clear that the diagnostic category of DLBCL is quite heterogeneous in terms of morphology, genetics, and biologic behavior. A number of clinicopathologic subtypes of DLBCL exist, such as T cell/histiocyte–rich large B-cell lymphoma, primary mediastinal large B-cell lymphoma, intravascular large B-cell lymphoma, DLBCL associated with chronic inflammation, lymphomatoid granulomatosis, and EBV-positive large B-cell lymphoma, among others. Gene expression profiling (GEP) can distinguish 2 cell of origin DLBCL subtypes: the germinal center B-cell (GCB) and activated B-cell (ABC) subtypes.15
DLBCL may be primary (de novo) or may arise through the transformation of many different types of low-grade B-cell lymphomas. This latter scenario is referred to as histologic transformation or transformed lymphoma. In some cases, patients may have a previously diagnosed low-grade B-cell NHL; in other cases, both low-grade and aggressive B-cell NHL may be diagnosed concurrently. The presence of elements of both low-grade and aggressive B-cell NHL in the same biopsy specimen is sometimes referred to as a composite lymphoma.
In the United States, incidence varies by ethnicity, with DLBCL being more common in Caucasians than other races.16 There is a slight male predominance (55%), median age at diagnosis is 65 years,16,17 and the incidence increases with age.
Presentation, Pathology, and Prognostic Factors
The most common presentation of patients with DLBCL is rapidly enlarging lymphadenopathy, usually in the neck or abdomen. Extranodal/extramedullary presentation is seen in approximately 40% of cases, with the gastrointestinal (GI) tract being the most common site. However, extranodal DLBCL can arise in virtually any tissue.18 Nodal DLBCL presents with symptoms related to the sites of involvement (eg, shortness of breath or chest pain with mediastinal lymphadenopathy), while extranodal DLBCL typically presents with symptoms secondary to dysfunction at the site of origin. Up to one third of patients present with constitutional symptoms (B symptoms) and more than 50% have elevated serum lactate dehydrogenase (LDH) at diagnosis.19
Approximately 40% of patients present with stage I/II disease. Of these, only a subset present with stage I, or truly localized disease (defined as that which can be contained within 1 irradiation field). About 60% of patients present with advanced (stage III–IV) disease.20 The bone marrow is involved in about 15% to 30% of cases. DLBCL involvement of the bone marrow is associated with a less favorable prognosis. Patients with DLBCL elsewhere may have low-grade NHL involvement of the bone marrow. Referred to as discordant bone marrow involvement,21 this feature does not carry the same poor prognosis associated with transformed disease22 or DLBCL involvement of the bone marrow.23
DLBCL is defined as a neoplasm of large B-lymphoid cells with a diffuse growth pattern. The proliferative fraction of cells, as determined by Ki-67 staining, is usually greater than 40%, and may even exceed 90%. Lymph nodes usually demonstrate complete effacement of the normal architecture by sheets of atypical lymphoid cells. Tumor cells in DLBCL generally express pan B-cell antigens (CD19, CD20, CD22, CD79a, Pax-5) as well as CD45 and surface immunoglobulin. Between 20% and 37% of DLBCL cases express the BCL-2 protein,24 and about 70% express the BCL-6 protein.25 C-MYC protein expression is seen in a higher percentage (~ 30%–50%) of cases of DLBCL.26
Many factors are associated with outcome in DLBCL. The IPI score was developed in the pre-rituximab era and is a robust prognostic tool. This simple tool uses 5 easily obtained clinical factors (age > 60 years, impaired performance status, elevated LDH, > 1 extranodal site of disease, and stage III/IV disease). By summing these factors, 4 groups with distinct 5-year overall survival (OS) rates ranging from 26% to 73% were identified (Table 2). Subsequently, modifications were made to adjust for age and stage, with the latest iteration being the NCCN (National Comprehensive Cancer Network) IPI.27 This tool uses age, performance status, LDH ratio (relative to the upper limit of normal), a more precise definition for presence of extranodal sites of disease (defined as lymphomatous involvement in the bone marrow, CNS, liver/GI tract, or lung), and Ann Arbor stage to stratify patients into 4 risk groups with significantly different 5-year OS, ranging from 38% to 96% based on the subgroup. Importantly, the NCCN-IPI was derived in a cohort of patients treated with rituximab-based therapy.
Cytogenetic and molecular factors also predict outcome in DLBCL. The ABC subtype distinguished by GEP has consistently been shown to have inferior outcomes with first-line therapy. As GEP is not routinely available in clinical practice, immunohistochemical (IHC) approaches (eg, the Hans algorithm) have been developed that can approximate the GEP subtypes. These IHC approaches have approximately 80% concordance with GEP.28 The 3 most common chromosomal translocations in DLBCL involve BCL-2, BCL-6 and MYC. MYC-rearranged DLBCLs have a less favorable prognosis.29,30 Cases in which a MYC translocation occurs in combination with a BCL-2 or BCL-6 translocation are commonly referred to as double-hit lymphoma (DHL); cases with all 3 translocations are referred to as triple-hit lymphoma (THL). Both DHL and THL have a worse prognosis with standard DLBCL therapy compared to non-DHL/THL cases. In the 2016 revised WHO classification, DHL and THL are an entity technically distinct from DLBCL, referred to as high-grade B-cell lymphoma.1 In some cases, MYC and BCL-2 protein overexpression occurs in the absence of chromosomal translocations. Cases in which MYC and BCL-2 are overexpressed (by IHC) are referred to as double expressor lymphoma (DEL), and also have inferior outcome compared with non-DEL DLBCL.31,32 Interestingly, MYC protein expression alone does not confer inferior outcomes, unlike isolated MYC translocation, which is associated with inferior outcomes.
Treatment
First-Line Therapy
DLBCL is an aggressive disease and, in most cases, survival without treatment can be measured in weeks to months. The advent of combination chemotherapy (CHOP [cyclophosphamide, doxorubicin, vincristine, and prednisone] or CHOP-like regimens) led to disease-free survival (DFS) rates of 35% to 40% at 3 to 5 years.33 The addition of rituximab to CHOP (R-CHOP) has improved both progression-free surivial (PFS) and OS.34,35
Treatment options vary for patients with localized (stage I/II) and advanced (stage III/IV) disease. Options for limited-stage DLBCL include an abbreviated course of R-CHOP (3 or 4 cycles) with involved-field radiation therapy (IFRT) versus a full course (6–8 cycles) of R-CHOP without radiation therapy (RT). Most studies comparing combined modality therapy (chemotherapy plus RT) versus chemotherapy alone were conducted in the pre-rituximab era. With the introduction of rituximab, Persky and colleagues36 studied the use of 3 cycles of R-CHOP followed by RT, demonstrating a slightly improved OS of 92% at 4 years as compared to 88% in a historical cohort. The French LYSA/GOELAMS group performed the only direct comparison in the rituximab era (4 cycles of R-CHOP followed by RT versus 4 cycles of R-CHOP followed by 2 additional cycles of R-CHOP) and reported similar outcomes between both arms,37 with OS of 92% in the R-CHOP alone arm and 96% in the R-CHOP + RT arm (nonsignificant difference statistically). IFRT alone is not recommended other than for palliation in patients who cannot tolerate chemotherapy or combined modality therapy. Stage I and II patients with bulky disease (> 10 cm) have a prognosis similar to patients with advanced DLBCL and should be treated aggressively with 6 to 8 cycles of R-CHOP with or without RT.36
For patients with advanced stage disease, a full course of R-CHOP-21 (6–8 cycles given on a 21-day cycle) is the standard of care. This approach results in OS rates of 70% and 60% at 2 and 5 years, respectively. For older adults unable to tolerate full-dose R-CHOP, attenuated versions of R-CHOP with decreased dose density or decreased dose intensity have been developed.38 Numerous randomized trials have attempted to improve upon the results of R-CHOP-21 using strategies such as infusional chemotherapy (DA-EPOCH-R [etoposide, prednisone, vincristine, cyclophosphamide, doxorubicin, rituximab]);39 dose-dense therapy (R-CHOP-14);replacement of rituximab with obinutuzuimab;40 addition of novel agents such as bortezomib,41 lenalidomide,42 or ibrutinib43,44 to R-CHOP; and various maintenance strategies such as rituximab, lenalidomide,45 enzastaurin,46 and everolimus.47 Unfortunately, none of these strategies has been shown to improve OS in DLBCL. In part this appears to be due to the fact that inclusion/exclusion criteria for DLBCL trials have been too strict, such that the most severely ill DLBCL patients are typically not included. As a result, the results in the control arms have ended up better than what was expected based on historical data. Efforts are underway to include all patients in future first-line DLBCL studies.
Currently, autologous hematopoietic cell transplantation (auto-HCT) is not routinely used in the initial treatment of DLBCL. In the pre-rituximab era, numerous trials were conducted in DLBCL patients with high and/or high-intermediate risk disease based on the IPI score to determine if outcomes could be improved with high-dose therapy and auto-HCT as consolidation after patients achieved complete remission with first-line therapy. The results of these trials were conflicting. A 2003 meta-analysis of 11 such trials concluded that the results were very heterogeneous and showed no OS benefit.48 More recently, the Southwestern Oncology Group published the results of a prospective trial testing the impact of auto-HCT for consolidation of aggressive NHL patients with an IPI score of 3 to 5 who achieved complete remission with first-line therapy with CHOP or R-CHOP. In this study, 75% of the patients had DLBCL and, of the B-cell NHL patients, 47% received R-CHOP. A survival benefit was seen only in the subgroup that had an IPI score of 4 or 5; a subgroup analysis restricted to those receiving R-CHOP as induction was not performed, however.49 As a result, this area remains controversial, with most institutions not routinely performing auto-HCT for any DLBCL patients in first complete remission and some institutions considering auto-HCT in first complete remission for patients with an IPI score of 4 or 5. These studies all used the IPI score to identify high-risk patients. It is possible that the use of newer biomarkers or minimal-residual disease analysis will lead to a more robust algorithm for identifying high-risk patients and selecting patients who might benefit from consolidation of first complete remission with auto-HCT.
For patients with DHL or THL, long-term PFS with standard R-CHOP therapy is poor (20% to 40%).50,51 Treatment with more intensive first-line regimens such as DA-EPOCH-R, R-hyperCVAD (rituximab plus hyperfractionated cyclophosphamide, vincristine, doxorubicin, dexamethasone), or CODOX-M/IVAC±R (cyclophosphamide, vincristine, doxorubicin, high‐dose methotrexate/ifosfamide, etoposide, high‐dose cytarabine ± rituximab), along with CNS prophylaxis, however, has been shown to produce superior outcomes,52 with 3-year relapse-free survival rates of 88% compared to 56% for R-CHOP. For patients who achieve a complete response by PET/CT scan after intensive induction, consolidation with auto-HCT has not been shown to improve outcomes based on retrospective analysis. However for DHL/THL patients who achieve complete response after R-CHOP, PFS was improved if auto-HCT was given as consolidation of first remission.53
Patients with DLBCL have an approximately 5% risk of subsequently developing CNS involvement. Historically (in the pre-rituximab era), patients who presented with multiple sites of extranodal disease and/or extensive bone marrow involvement and/or an elevated LDH had an increased risk (up to 20%–30%) of developing CNS involvement. In addition, patients with involvement of certain anatomical sites (testicular, paranasal sinuses, epidural space) had an increased risk of CNS disease. Several algorithms have been proposed to identify patients who should receive prophylactic CNS therapy. One of the most robust tools for this purpose is the CNS-IPI, which is a 6-point score consisting of the 5 IPI elements, plus 1 additional point if the adrenal glands or kidneys are involved. Importantly, the CNS-IPI was developed and validated in patients treated with R-CHOP-like therapy. Subsequent risk of CNS relapse was 0.6%, 3.4%, and 10.2% for those with low-, intermediate- and high-risk CNS-IPI scores, respectively.54 A reasonable strategy, therefore, is to perform CNS prophylaxis in those with a CNS-IPI score of 4 to 6. When CNS prophylaxis is used, intrathecal methotrexate or high-dose systemic methotrexate is most frequently given, with high-dose systemic methotrexate favored over intrathecal chemotherapy given that high-dose methotrexate penetrates the brain and spinal cord parenchyma, in addition to treating the cerebrospinal fluid (CSF).55 In contrast, intrathecal therapy only treats the CSF and requires repeated lumbar punctures or placement of an Ommaya reservoir. For DLBCL patients who present with active CSF involvement (known as lymphomatous meningitis), intrathecal chemotherapy treatments are typically given 2 or 3 times weekly until the CSF clears, followed by weekly intrathecal treatment for 4 weeks, and then monthly intrathecal treatment for 4 months.56 For those with concurrent systemic and brain parenchymal DLBCL, a strategy of alternating R-CHOP with mid-cycle high-dose methotrexate can be successful. In addition, consolidation with high-dose therapy and auto-HCT improved survival in such patients in 1 retrospective series.57
Relapsed/Refractory Disease
Between 30% and 40% of patients with advanced stage DLBCL will either fail to attain a remission with primary therapy (referred to as primary induction failure) or will relapse. In general, for those with progressive or relapsed disease, an updated tissue biopsy is recommended. This is especially true for patients who have had prior complete remission and have new lymph node enlargement, or those who have emergence of new sites of disease at the completion of first-line therapy.
Patients with relapsed disease are treated with systemic second-line platinum-based chemoimmunotherapy, with the usual goal of ultimately proceeding to auto-HCT. A number of platinum-based regimens have been used in this setting such as R-ICE, R-DHAP, R-GDP, R-Gem-Ox, and R-ESHAP. None of these regimens has been shown to be superior in terms of efficacy, and the choice of regimen is typically made based on the anticipated tolerance of the patient in light of comorbidities, laboratory studies, and physician preference. In the CORAL study, R-DHAP (rituximab, dexamethasone, high-dose cytarabine, cisplatin) seemed to show superior PFS in patients with the GCB subtype.58 However, this was an unplanned subgroup analysis and R-DHAP was associated with higher renal toxicity.
Several studies have demonstrated that long-term PFS can be observed for relapsed/refractory DLBCL patients who respond to second-line therapy and then undergo high-dose therapy with auto-HCT. The Parma trial remains the only published prospective randomized trial performed in relapsed DLBCL comparing a transplant strategy to a non-transplant strategy. This study, performed in the pre-rituximab era, clearly showed a benefit in terms of DFS and OS in favor of auto-HCT versus salvage therapy alone.59 The benefit of auto-HCT in patients treated in the rituximab era, even in patients who experience early failure (within 1 year of diagnosis), was confirmed in a retrospective analysis by the Center for International Blood and Marrow Transplant Research. In this study, a 44% 3-year PFS was seen in the early failure cohort versus 52% in the late failure cohort.60
Some DLBCL patients are very unlikely to benefit from auto-HCT. The REFINE study focused on patients with primary induction failure or early relapse within 6 months of completing first-line therapy. Among such patients, primary progressive disease (defined as progression while still receiving first-line therapy), a high NCCN-IPI score at relapse, and MYC rearrangement were risk factors for poor PFS following auto-HCT.61 Patients with 2 or 3 high-risk features had a 2-year OS of 10.7% compared to 74.3% for those without any high-risk features.
Allogeneic HCT (allo-HCT) is a treatment option for relapsed/refractory DLBCL. This option is more commonly considered for patients in whom an autotransplant has failed to achieve durable remission. For properly selected patients in this setting, a long-term PFS in the 30% to 40% range can be attained.62 However, in practice, only about 20% of patients who fail auto-HCT end up undergoing allo-HCT due to rapid progression of disease, age, poor performance status, or lack of suitable donor. It has been proposed that in the coming years, allo-HCT will be utilized less commonly in this setting due to the advent of chimeric antigen receptor T-cell (CAR T) therapy.
CAR T-cell therapy genetically modifies the patient’s own T lymphocytes with a gene that encodes an antigen receptor to direct the T cells against lymphoma cells. Typically, the T cells are genetically modified and expanded in a production facility and then infused back into the patient. Axicabtagene ciloleucel is directed against the CD-19 receptor and has been approved by the US Food and Drug Administration (FDA) for treatment of patients with DLBCL who have failed 2 or more lines of systemic therapy. Use of CAR-T therapy in such patients was examined in a multicenter trial (ZUMA-1), which reported a 54% complete response rate and 52% OS rate at 18 months.63 CAR-T therapy is associated with serious side effects such as cytokine release syndrome, neurological toxicities, and prolonged cytopenias. While there are now some patients with ongoing remission 2 or more years after undergoing CAR-T therapy, it remains uncertain what proportion of patients have been truly cured with this modality. Nevertheless, this new treatment option remains a source of optimism for relapsed and refractory DLBCL patients.
Primary Mediastinal Large B-Cell Lymphoma
Primary mediastinal large B-cell lymphoma (PMBCL) is a form of DLBCL arising in the mediastinum from the thymic B cell. It is an uncommon entity and has clinical and pathologic features distinct from systemic DLBCL.64 PMBCL accounts for 2% of all NHLs and about 7% of all DLBCL.20 It typically affects women in the third to fourth decade of life.
Presentation and Prognostic Features
PMBCL usually presents as a locally invasive anterior mediastinal mass, often with a superior vena cava syndrome which may or may not be clinically obvious.64 Other presentations include pericardial tamponade, thrombosis of neck veins, and acute airway obstruction. About 80% of patients present with bulky (> 10 cm) stage I or II disease,65 with distant spread uncommon on presentation. Morphologically and on GEP, PMBL has a profile more similar to classical Hodgkin lymphoma (cHL) than non-mediastinal DLBCL.66 PMBL is distinguished from cHL by immunophenotyping: unlike cHL, PMBCL has pan B cell markers, rarely expresses CD15, and has weak CD30.
Poor prognostic features in PMBCL are Eastern Cooperative Oncology Group (ECOG) performance status greater than 2, pericardial effusion, bulky disease, and elevated serum LDH. The diagnosis of PMBCL can be difficult because the tumor is often encased with extensive fibrosis and necrosis. As a result, a needle biopsy may not yield sufficient tissue, thus making a surgical biopsy often the only viable way to obtain sufficient tissue.
Treatment
Early series suggested that PMBCL is unusually aggressive, with a poor prognosis.67 This led to studies using more aggressive chemotherapy regimens (often in combination with mediastinal radiation) as well as upfront auto-HCT.68–70 The addition of rituximab to treatment regimens significantly improved outcomes in PMBCL. For example, a subgroup analysis of the PMBCL patients in the MinT trial revealed a 3-year event-free survival (EFS) of 78%71 when rituximab was combined with CHOP. Because of previous reports demonstrating radiosensitivity of PMBL, radiation was traditionally sequenced into treatment regimens for PMBL. However, this is associated with higher long-term toxicities, often a concern in PMBCL patients given that the disease frequently affects younger females, and given that breast tissue will be in the radiation field. For patients with a strong personal or family history of breast cancer or cardiovascular disease, these concerns are even more significant. More recently, the DA-EPOCH-R regimen has been shown to produce very high rates (80%–90%) of long-term DFS, without the need for mediastinal radiation in most cases.72,73 For patients receiving R-CHOP, consolidation with mediastinal radiation is still commonly given. This approach also leads to high rates of long-term remission and, although utilizing mediastinal radiation, allows for less intensive chemotherapy. Determining which approach is most appropriate for an individual patient requires an assessment of the risks of each treatment option for that patient. A randomized trial by the International Extranodal Lymphoma Study Group (IELSG37) is evaluating whether RT may be safely omitted in PMBCL patients who achieve a complete metabolic response after R-CHOP.
Most relapses of PMBCL occur within the first 1 to 2 years and often present with extranodal disease in various organs. For those with relapsed or refractory disease, high-dose chemotherapy followed by auto-HCT provides 5-year survival rates of 50% to 80%.74–76 In a phase 1b trial evaluating the role of pembrolizumab in relapsed/refractory patients (KEYNOTE-13), 7 of 17 PMBCL patients achieved responses, with an additional 6 demonstrating stable disease.77 This provides an additional option for patients who might be too weak to undergo auto-HCT or for those who relapse following auto-HCT.
Mantle Cell Lymphoma
The name mantle cell lymphoma (MCL) is based on the presumed normal cell counterpart to MCL, which is believed to be found in the mantle zone surrounding germinal center follicles. It represents approximately 6% of all NHL cases in the United States and Europe.78 MCL occurs at a median age of 63 to 68 years and has a male predominance.
Presentation and Prognostic Features
Patients can present with a broad spectrum of clinical features, and most patients (70%) present with advanced disease.79 Up to one third of patients have B symptoms, with most demonstrating lymphadenopathy and bone marrow involvement. Approximately 25% present with extranodal disease as the primary presentation (eg, GI tract, pleura, breast, or orbits). MCL can involve any part of the GI tract and often presents as polypoid lesions.
Histologically, the pattern of MCL may be diffuse, nodular, mantle zone, or a combination of the these; morphologically, MCL can range from small, more irregular lymphocytes to lymphoblast-like cells. Blastoid and pleomorphic variants of MCL have a higher proliferation index and a more aggressive clinical course than other variants. MCL is characterized by the expression of pan B cell antigens (CD19+, CD20+) with coexpression of the T-cell antigen CD5, lack of CD23 expression, and nuclear expression of cyclin D1. Nuclear staining for cyclin D1 is present in more than 98% of cases.80 In rare cases, CD5 or cyclin D1 may be negative.80 Most MCL cases have a unique translocation that fuses the immunoglobulin heavy chain gene promoter (14q32) to the promoter of the BCL-1 gene (11q13), which encodes the cyclin D1 protein. This translocation is not unique to MCL and can be present in multiple myeloma as well. Interestingly, cyclin D1 is overproduced in cases lacking t(11:14), likely from other point mutations resulting in its overexpression.81 Cyclin D1–negative tumors overexpress cyclin D2 or D3, with no apparent difference in clinical behavior or outcome.82 In cyclin D1–negative cases, SOX11 expression may help with diagnosis.83 A proliferation rate greater than 30% (as measured by Ki-67 staining), low SOX11 expression, and presence of p53 mutations have all been associated with adverse outcome.
In a minority of cases, MCL follows an indolent clinical course. For the remainder, however, MCL is an aggressive disease that generally requires treatment soon after diagnosis. When initially described in the 1980s and 1990s, treatment of MCL was characterized by low complete response rates, short durations of remission, repeated recurrences, and a median survival in the 2- to 5-year range.84 In recent years, intensive regimens incorporating rituximab and high-dose cytarabine with or without auto-HCT have been developed and are associated with high complete response rates and median duration of first remission in the 6- to 9-year range.85–87 Several prognostic indices have been applied to patients with MCL, including the IPI, the Follicular Lymphoma International Prognostic Index , and the Mantle Cell Lymphoma International Prognostic Index (MIPI). The MIPI was originally described based on a cohort from the period 1996 to 2004,88 and subsequently confirmed in a separate cohort of 958 patients with MCL treated on prospective trials between 2004 and 2010.89 The MIPI score can identify 3 risk groups with significant survival differences (83%, 63%, and 34% survival at 5 years). A refined version of the MIPI score, the combined MIPI or MIPI-c, incorporates proliferation rate and is better able to stratify patients.90 The blastoid variant of MCL follows a more aggressive clinical course and is associated with a high proliferation rate, shorter remissions, and a higher rate of CNS involvement.91
In most patients, MCL is an aggressive disease with a short OS without treatment. A subset of patients may have a more indolent course,92 but unfortunately reliable factors that identify this group at the time of diagnosis are not available. Pretreatment evaluation is as with other lymphomas, with lumbar puncture and MRI of the brain also recommended for patients with the blastoid variant. For those presenting with GI symptoms, endoscopy is recommended as part of the initial evaluation as well.
Treatment
First-line Therapy
For patients under age 65 to 70 years with a good performance status and few comorbidities, an intensive induction regimen (such as R-CHOP/R-DHAP, Maxi-R-CHOP/R-araC, or R-DHAP) followed by consolidation with auto-HCT is commonly given, with a goal of achieving a durable (6–9 year) first remission.87,93,94 Auto-HCT is now routinely followed by 3 years of maintenance rituximab based on the survival benefit seen in the recent LYSA trial.93 At many centers, auto-HCT in first remission is a standard of care, with the greatest benefit seen in patients who have achieved a complete remission with no more than 2 lines of chemotherapy.95 However, there remains some controversy about whether all patients truly benefit from auto-HCT in first remission, and current research efforts are focused on identifying patients most likely to benefit from auto-HCT and incorporation of new agents into first-line regimens. For patients who are not candidates for auto-HCT, bendamustine plus rituximab (BR) or R-CHOP alone or followed by maintenance rituximab is a reasonable approach.96 Based on the StiL and BRIGHT trials, BR seems to have less toxicity and higher rates of response with no difference in OS when compared to R-CHOP.97,98
In summary, dose-intense induction chemotherapy with consolidative auto-HCT results in high rates of long-term remission and can be considered in MCL patients who lack significant comorbidities and who understand the risks and benefits of this approach. For other patients, the less aggressive frontline approaches are more appropriate.
Relapsed/Refractory Disease
Despite initial high response rates, most patients with MCL will eventually relapse. For example, most patients given CHOP or R-CHOP alone as first-line therapy will relapse within 2 years.99 In recent years, a number of therapies have emerged for relapsed/refractory MCL; however, the optimal sequencing of these is unclear. FDA-approved options for relapsed/refractory MCL include the proteasome inhibitor bortezomib,100,101 the BTK inhibitors ibrutinib102,103 and acalabrutinib,104 and the immunomodulatory agent lenalidomide.105
Auto-HCT can be considered for patients who did not undergo auto-HCT as part of first-line therapy and who had a reasonably long first remission.95 Allo-HCT has curative potential in MCL with good evidence of a graft-versus-lymphoma effect. With a matched related or matched unrelated donor, the chance for treatment-related mortality is 15% to 25% at 1 to 2 years, with a 50% to 60% chance for long-term PFS. However, given the risk of treatment-related mortality and graft-versus-host disease, this option is typically reserved for patients with early relapse after auto-HCT, multiple relapses, or relatively chemotherapy-unresponsive disease.95,106 A number of clinical trials for relapsed/refractory MCL are ongoing, and participation in these is encouraged whenever possible.
Burkitt Lymphoma
Burkitt lymphoma is a rare, aggressive and highly curable subtype of NHL. It can occur at any age, although peak incidence is in the first decade of life. There are 3 distinct clinical forms of Burkitt lymphoma.107 The endemic form is common in African children and commonly involves the jaw and kidneys. The sporadic (nonendemic) form accounts for 1% to 2% of all lymphomas in the United States and Western Europe and usually has an abdominal presentation. The immunodeficiency-associated form is commonly seen in HIV patients with a relatively preserved CD4 cell count.
Patients typically present with rapidly growing masses and tumor lysis syndrome. CNS and bone marrow involvement are common. Burkitt lymphoma cells are high-grade, rapidly proliferating medium-sized cells with a monomorphic appearance. Biopsies show a classic histological appearance known as a “starry sky pattern” due to benign macrophages engulfing debris resulting from apoptosis. It is derived from a germinal center B cell and has distinct oncogenic pathways. Translocations such as t(8;14), t(2;8) or t(8;22) juxtapose the MYC locus with immunoglobulin heavy or light chain loci and result in MYC overexpression. Burkitt lymphoma is typically CD10-positive and BCL-2-negative, with a MYC translocation and a proliferation rate greater than 95%.
With conventional NHL regimens, Burkitt lymphoma had a poor prognosis, with complete remission in the 30% to 70% range and low rates of long-term remission. With the introduction of short-term, dose-intensive, multiagent chemotherapy regimens (adapted from pediatric acute lymphoblastic leukemia [ALL] regimens), the complete remission rate improved to 60% to 90%.107 Early stage disease (localized or completely resected intra-abdominal disease) can have complete remission rates of 100%, with 2- to 5-year freedom-from-progression rates of 95%. CNS prophylaxis, including high-dose methotrexate, high-dose cytarabine, and intrathecal chemotherapy, is a standard component of Burkitt lymphoma regimens (CNS relapse rates can reach 50% without prophylactic therapy). Crucially, relapse after 1 to 2 years is very rare following complete response to induction therapy. Classically, several intensive regimens have been used for Burkitt lymphoma. In recent years, the most commonly used regimens have been the modified Magrath regimen of R-CODOX-M/IVAC and R-hyperCVAD. DA-EPOCH-R has also been used, typically for older, more frail, or HIV-positive patients. However, at the American Society of Hematology 2017 annual meeting, results from the NCI 9177 trial were presented which validated, in a prospective multi-center fashion, the use of DA-EPOCH-R in all Burkitt lymphoma patients.108 In NCI 9177, low-risk patients (defined as normal LDH, ECOG performance score 0 or 1, ≤ stage II, and no tumor lesion > 7 cm) received 2 cycles of DA-EPOCH-R without intrathecal therapy followed by PET. If interim PET was negative, low-risk patients then received 1 more cycle of DA-EPOCH-R. High-risk patients with negative brain MRI and CSF cytology/flow cytometry received 2 cycles of DA-EPOCH-R with intrathecal therapy (2 doses per cycle) followed by PET. Unless interim PET showed progression, high-risk patients received 4 additional cycles of DA-EPOCH-R including methotrexate 12 mg intrathecally on days 1 and 5 (8 total doses). With a median follow-up of 36 months, this regimen resulted in an EFS of 85.7%. As expected, patients with CNS, marrow, or peripheral blood involvement fared worse. For those without CNS, marrow, or peripheral blood involvement, the results were excellent, with an EFS of 94.6% compared to 62.8% for those with CNS, bone marrow, or blood involvement at diagnosis.
Although no standard of care has been defined, patients with relapsed/refractory Burkitt lymphoma are often given standard second-line aggressive NHL regimens (eg, R-ICE); for those with chemosensitive disease, auto- or allo-HCT is often pursued, with long-term remissions possible following HCT.109
Lymphoblastic Lymphoma
Lymphoblastic lymphoma (LBL) is a rare disease postulated to arise from precursor B or T lymphoblasts at varying stages of differentiation. Accounting for approximately 2% of all NHLs, 85% to 90% of all cases have a T-cell phenotype, while B-cell LBL comprises approximately 10% to 15% of cases. LBL and ALL are thought to represent the same disease entity, but LBL has been arbitrarily defined as cases with lymph node or mediastinal disease. Those with significant (> 25%) bone marrow or peripheral blood involvement are classified as ALL.
Precursor T-cell LBL patients are usually adolescent and young males who commonly present with a mediastinal mass and peripheral lymphadenopathy. Precursor B-cell LBL patients are usually older (median age 39 years) with peripheral lymphadenopathy and extranodal involvement. Mediastinal involvement with B-cell LBL is uncommon, and there is no male predominance. LBL has a propensity for dissemination to the bone marrow and CNS.
Morphologically, the tumor cells are medium sized, with a scant cytoplasm and finely dispersed chromatin. Mitotic features and apoptotic bodies are present since it is a high-grade malignancy. The lymphoblasts are typically positive for CD7 and either surface or cytoplasmic CD3. Terminal deoxynucleotidyl transferase expression is a defining feature. Other markers such as CD19, CD22, CD20, CD79a, CD45, and CD10 are variably expressed. Poor prognostic factors in T-cell LBL are female gender, age greater than 35 years, complex cytogenetics, and lack of a matched sibling donor.
Regimens for LBL are based on dose-dense, multi-agent protocols used in ALL. Most of these regimens are characterized by intensive remission-induction chemotherapy, CNS prophylaxis, a phase of consolidation therapy, and a prolonged maintenance phase, often lasting for 12 to 18 months with long-term DFS rates of 40% to 70%.110,111 High-dose therapy with auto-HCT or allo-HCT in first complete response has been evaluated in an attempt to reduce the incidence of relapse.112 However, the intensity of primary chemotherapy appears to be a stronger determinant of long-term survival than the use of HCT as consolidation. As a result, HCT is not routinely applied to patients in first complete remission following modern induction regimens. After relapse, prognosis is poor, with median survival rates of 6 to 9 months with conventional chemotherapy, although long-term survival rates of 30% and 20%, respectively, are reported after HCT in relapsed and primary refractory disease.113
Treatment options in relapsed disease are limited. Nelarabine can produce responses in up to 40% of relapsed/refractory LBL/ALL patients.114
Summary
Aggressive NHLs are characterized by rapid clinical progression without therapy. However, a significant proportion of patients are cured with appropriate combination chemotherapy or combined modality (chemotherapy + RT) regimens. In contrast, the indolent lymphomas have a relatively good prognosis (median survival of 10 years or longer) but usually are not curable in advanced clinical stages. Overall 5-year survival for aggressive NHLs with current treatment is approximately 50% to 60%, with relapses typically occurring within the first 5 years. Treatment strategies for relapsed patients offer some potential for cure; however, clinical trial participation should be encouraged whenever possible to investigate new approaches for improving outcomes in this patient population.
Introduction
Non-Hodgkin lymphoma (NHL) comprises a wide variety of malignant hematologic disorders with varying clinical and biological features. The more than 60 separate NHL subtypes can be classified according to cell of origin (B cell versus T cell), anatomical location (eg, orbital, testicular, bone, central nervous system), clinical behavior (indolent versus aggressive), histological features, or cytogenetic abnormalities. Although various NHL classification schemes have been used over the years, the World Health Organization (WHO) classification is now widely accepted as the definitive pathologic classification system for lymphoproliferative disorders, incorporating morphologic, immunohistochemical, flow cytometric, cytogenetic, and molecular features.1 While the pathologic and molecular subclassification of NHL has become increasingly refined in recent years, from a management standpoint, classification based on clinical behavior remains very useful. This approach separates NHL subtypes into indolent versus aggressive categories. Whereas indolent NHLs may remain clinically insignificant for months to years, aggressive B-cell NHLs generally become life-threatening within weeks to months without treatment.
Epidemiology
Data from cancer registries show a steady, unexplainable increase in the incidence of NHL during the second half of the 20th century; the incidence has subsequently plateaued. There was a significant increase in NHL incidence between 1970 and 1995, which has been attributed in part to the HIV epidemic. More than 72,000 new cases of NHL were diagnosed in the United States in 2017, compared to just over 8000 cases of Hodgkin lymphoma, making NHL the sixth most common cancer in adult men and the fifth most common in adult women.2 NHL appears to occur more frequently in Western countries than in Asian populations.
Various factors associated with increased risk for B-cell NHL have been identified over the years, including occupational and environmental exposure to certain pesticides and herbicides,3 immunosuppression associated with HIV infection,4 autoimmune disorders,5 iatrogenically induced immune suppression in the post-transplant and other settings,6 family history of NHL,7 and a personal history of a prior cancer, including Hodgkin lymphoma and prior NHL.8 In terms of infectious agents associated with aggressive B-cell NHLs, Epstein-Barr virus (EBV) has a clear pathogenic role in Burkitt lymphoma, in many cases of post-transplant lymphoproliferative disorders, and in some cases of HIV-related aggressive B-cell lymphoma.9 Human herpesvirus-8 viral genomes have been found in virtually all cases of primary effusion lymphomas.10 Epidemiological studies also have linked hepatitis B and C to increased incidences of certain NHL subtypes,11–13 including primary hepatic diffuse large B-cell lymphoma (DLBCL). Similarly, Helicobacter pylori has been associated with gastric DLBCL.
Staging and Work-Up
A tissue biopsy is essential in the diagnosis and management of NHL. The most significant disadvantage of fine-needle aspiration cytology is the lack of histologic architecture. The optimal specimen is an excisional biopsy; when this cannot be performed, a core needle biopsy, ideally using a 16-gauge or larger caliber needle, is the next best choice.
The baseline tests appropriate for most cases of newly diagnosed aggressive B-cell NHL are listed in Table 1. Both hepatitis B and C have been associated with increased risk of NHL. In addition, there is a risk of hepatitis B reactivation following certain NHL therapies. A contrast-enhanced computed tomography (CT) scan in addition to positron emission tomography (PET) is useful to define the extent of disease in situations needing greater definition (eg, lymphadenopathy close to the bowel, cervical and supraclavicular nodal involvement, and lymphadenopathy causing thrombosis or compression of nearby structures).14 In cases where it is apparent that the patient has advanced stage disease (Ann Arbor stage III/IV) based on imaging, bone marrow biopsy is unlikely to alter the treatment plan. For such patients, if the complete blood count is unremarkable, deferral of bone marrow biopsy may be reasonable. For new cases of DLBCL, assessment for MYC translocation by fluorescence in situ hybridization (FISH) is recommended. If a MYC translocation is identified, then testing for BCL2 and BCL6 translocations by FISH should be performed.
Prior to the initiation of treatment, patients should always undergo a thorough cardiac and pulmonary evaluation, especially if the patient will be treated with an anthracycline or mediastinal irradiation. Central nervous system (CNS) evaluation with magnetic resonance imaging (MRI) and lumbar puncture is essential if there are neurological signs or symptoms. In addition, certain anatomical sites including the testicles, paranasal sinuses, kidney, adrenal glands, and epidural space have been associated with increased involvement of the CNS and may warrant MRI evaluation and lumbar puncture. Certain NHL subtypes like Burkitt lymphoma, high-grade NHL with translocations of MYC and BCL-2 or BCL-6 (double-hit lymphoma), blastoid mantle cell lymphoma, and lymphoblastic lymphoma have a high risk of CNS involvement, and patients with these subtypes need CNS evaluation.
The Lugano classification is used to stage patients with NHL.14 This classification is based on the Ann Arbor staging system and uses the distribution and number of tumor sites to stage disease. In general, this staging system in isolation is of limited value in predicting survival after treatment. However, the Ann Arbor stage does have prognostic impact when incorporated into risk scoring systems such as the International Prognostic Index (IPI). In clinical practice, the Ann Arbor stage is useful primarily to determine eligibility for localized therapy approaches. The absence or presence of systemic symptoms such as fevers, drenching night sweats, or weight loss (> 10% of baseline over 6 months or less) is designated by A or B, respectively.
Diffuse Large B-Cell Lymphoma
DLBCL is the most common lymphoid neoplasm in adults, accounting for about 25% of all NHL cases.2 It is increasingly clear that the diagnostic category of DLBCL is quite heterogeneous in terms of morphology, genetics, and biologic behavior. A number of clinicopathologic subtypes of DLBCL exist, such as T cell/histiocyte–rich large B-cell lymphoma, primary mediastinal large B-cell lymphoma, intravascular large B-cell lymphoma, DLBCL associated with chronic inflammation, lymphomatoid granulomatosis, and EBV-positive large B-cell lymphoma, among others. Gene expression profiling (GEP) can distinguish 2 cell of origin DLBCL subtypes: the germinal center B-cell (GCB) and activated B-cell (ABC) subtypes.15
DLBCL may be primary (de novo) or may arise through the transformation of many different types of low-grade B-cell lymphomas. This latter scenario is referred to as histologic transformation or transformed lymphoma. In some cases, patients may have a previously diagnosed low-grade B-cell NHL; in other cases, both low-grade and aggressive B-cell NHL may be diagnosed concurrently. The presence of elements of both low-grade and aggressive B-cell NHL in the same biopsy specimen is sometimes referred to as a composite lymphoma.
In the United States, incidence varies by ethnicity, with DLBCL being more common in Caucasians than other races.16 There is a slight male predominance (55%), median age at diagnosis is 65 years,16,17 and the incidence increases with age.
Presentation, Pathology, and Prognostic Factors
The most common presentation of patients with DLBCL is rapidly enlarging lymphadenopathy, usually in the neck or abdomen. Extranodal/extramedullary presentation is seen in approximately 40% of cases, with the gastrointestinal (GI) tract being the most common site. However, extranodal DLBCL can arise in virtually any tissue.18 Nodal DLBCL presents with symptoms related to the sites of involvement (eg, shortness of breath or chest pain with mediastinal lymphadenopathy), while extranodal DLBCL typically presents with symptoms secondary to dysfunction at the site of origin. Up to one third of patients present with constitutional symptoms (B symptoms) and more than 50% have elevated serum lactate dehydrogenase (LDH) at diagnosis.19
Approximately 40% of patients present with stage I/II disease. Of these, only a subset present with stage I, or truly localized disease (defined as that which can be contained within 1 irradiation field). About 60% of patients present with advanced (stage III–IV) disease.20 The bone marrow is involved in about 15% to 30% of cases. DLBCL involvement of the bone marrow is associated with a less favorable prognosis. Patients with DLBCL elsewhere may have low-grade NHL involvement of the bone marrow. Referred to as discordant bone marrow involvement,21 this feature does not carry the same poor prognosis associated with transformed disease22 or DLBCL involvement of the bone marrow.23
DLBCL is defined as a neoplasm of large B-lymphoid cells with a diffuse growth pattern. The proliferative fraction of cells, as determined by Ki-67 staining, is usually greater than 40%, and may even exceed 90%. Lymph nodes usually demonstrate complete effacement of the normal architecture by sheets of atypical lymphoid cells. Tumor cells in DLBCL generally express pan B-cell antigens (CD19, CD20, CD22, CD79a, Pax-5) as well as CD45 and surface immunoglobulin. Between 20% and 37% of DLBCL cases express the BCL-2 protein,24 and about 70% express the BCL-6 protein.25 C-MYC protein expression is seen in a higher percentage (~ 30%–50%) of cases of DLBCL.26
Many factors are associated with outcome in DLBCL. The IPI score was developed in the pre-rituximab era and is a robust prognostic tool. This simple tool uses 5 easily obtained clinical factors (age > 60 years, impaired performance status, elevated LDH, > 1 extranodal site of disease, and stage III/IV disease). By summing these factors, 4 groups with distinct 5-year overall survival (OS) rates ranging from 26% to 73% were identified (Table 2). Subsequently, modifications were made to adjust for age and stage, with the latest iteration being the NCCN (National Comprehensive Cancer Network) IPI.27 This tool uses age, performance status, LDH ratio (relative to the upper limit of normal), a more precise definition for presence of extranodal sites of disease (defined as lymphomatous involvement in the bone marrow, CNS, liver/GI tract, or lung), and Ann Arbor stage to stratify patients into 4 risk groups with significantly different 5-year OS, ranging from 38% to 96% based on the subgroup. Importantly, the NCCN-IPI was derived in a cohort of patients treated with rituximab-based therapy.
Cytogenetic and molecular factors also predict outcome in DLBCL. The ABC subtype distinguished by GEP has consistently been shown to have inferior outcomes with first-line therapy. As GEP is not routinely available in clinical practice, immunohistochemical (IHC) approaches (eg, the Hans algorithm) have been developed that can approximate the GEP subtypes. These IHC approaches have approximately 80% concordance with GEP.28 The 3 most common chromosomal translocations in DLBCL involve BCL-2, BCL-6 and MYC. MYC-rearranged DLBCLs have a less favorable prognosis.29,30 Cases in which a MYC translocation occurs in combination with a BCL-2 or BCL-6 translocation are commonly referred to as double-hit lymphoma (DHL); cases with all 3 translocations are referred to as triple-hit lymphoma (THL). Both DHL and THL have a worse prognosis with standard DLBCL therapy compared to non-DHL/THL cases. In the 2016 revised WHO classification, DHL and THL are an entity technically distinct from DLBCL, referred to as high-grade B-cell lymphoma.1 In some cases, MYC and BCL-2 protein overexpression occurs in the absence of chromosomal translocations. Cases in which MYC and BCL-2 are overexpressed (by IHC) are referred to as double expressor lymphoma (DEL), and also have inferior outcome compared with non-DEL DLBCL.31,32 Interestingly, MYC protein expression alone does not confer inferior outcomes, unlike isolated MYC translocation, which is associated with inferior outcomes.
Treatment
First-Line Therapy
DLBCL is an aggressive disease and, in most cases, survival without treatment can be measured in weeks to months. The advent of combination chemotherapy (CHOP [cyclophosphamide, doxorubicin, vincristine, and prednisone] or CHOP-like regimens) led to disease-free survival (DFS) rates of 35% to 40% at 3 to 5 years.33 The addition of rituximab to CHOP (R-CHOP) has improved both progression-free surivial (PFS) and OS.34,35
Treatment options vary for patients with localized (stage I/II) and advanced (stage III/IV) disease. Options for limited-stage DLBCL include an abbreviated course of R-CHOP (3 or 4 cycles) with involved-field radiation therapy (IFRT) versus a full course (6–8 cycles) of R-CHOP without radiation therapy (RT). Most studies comparing combined modality therapy (chemotherapy plus RT) versus chemotherapy alone were conducted in the pre-rituximab era. With the introduction of rituximab, Persky and colleagues36 studied the use of 3 cycles of R-CHOP followed by RT, demonstrating a slightly improved OS of 92% at 4 years as compared to 88% in a historical cohort. The French LYSA/GOELAMS group performed the only direct comparison in the rituximab era (4 cycles of R-CHOP followed by RT versus 4 cycles of R-CHOP followed by 2 additional cycles of R-CHOP) and reported similar outcomes between both arms,37 with OS of 92% in the R-CHOP alone arm and 96% in the R-CHOP + RT arm (nonsignificant difference statistically). IFRT alone is not recommended other than for palliation in patients who cannot tolerate chemotherapy or combined modality therapy. Stage I and II patients with bulky disease (> 10 cm) have a prognosis similar to patients with advanced DLBCL and should be treated aggressively with 6 to 8 cycles of R-CHOP with or without RT.36
For patients with advanced stage disease, a full course of R-CHOP-21 (6–8 cycles given on a 21-day cycle) is the standard of care. This approach results in OS rates of 70% and 60% at 2 and 5 years, respectively. For older adults unable to tolerate full-dose R-CHOP, attenuated versions of R-CHOP with decreased dose density or decreased dose intensity have been developed.38 Numerous randomized trials have attempted to improve upon the results of R-CHOP-21 using strategies such as infusional chemotherapy (DA-EPOCH-R [etoposide, prednisone, vincristine, cyclophosphamide, doxorubicin, rituximab]);39 dose-dense therapy (R-CHOP-14);replacement of rituximab with obinutuzuimab;40 addition of novel agents such as bortezomib,41 lenalidomide,42 or ibrutinib43,44 to R-CHOP; and various maintenance strategies such as rituximab, lenalidomide,45 enzastaurin,46 and everolimus.47 Unfortunately, none of these strategies has been shown to improve OS in DLBCL. In part this appears to be due to the fact that inclusion/exclusion criteria for DLBCL trials have been too strict, such that the most severely ill DLBCL patients are typically not included. As a result, the results in the control arms have ended up better than what was expected based on historical data. Efforts are underway to include all patients in future first-line DLBCL studies.
Currently, autologous hematopoietic cell transplantation (auto-HCT) is not routinely used in the initial treatment of DLBCL. In the pre-rituximab era, numerous trials were conducted in DLBCL patients with high and/or high-intermediate risk disease based on the IPI score to determine if outcomes could be improved with high-dose therapy and auto-HCT as consolidation after patients achieved complete remission with first-line therapy. The results of these trials were conflicting. A 2003 meta-analysis of 11 such trials concluded that the results were very heterogeneous and showed no OS benefit.48 More recently, the Southwestern Oncology Group published the results of a prospective trial testing the impact of auto-HCT for consolidation of aggressive NHL patients with an IPI score of 3 to 5 who achieved complete remission with first-line therapy with CHOP or R-CHOP. In this study, 75% of the patients had DLBCL and, of the B-cell NHL patients, 47% received R-CHOP. A survival benefit was seen only in the subgroup that had an IPI score of 4 or 5; a subgroup analysis restricted to those receiving R-CHOP as induction was not performed, however.49 As a result, this area remains controversial, with most institutions not routinely performing auto-HCT for any DLBCL patients in first complete remission and some institutions considering auto-HCT in first complete remission for patients with an IPI score of 4 or 5. These studies all used the IPI score to identify high-risk patients. It is possible that the use of newer biomarkers or minimal-residual disease analysis will lead to a more robust algorithm for identifying high-risk patients and selecting patients who might benefit from consolidation of first complete remission with auto-HCT.
For patients with DHL or THL, long-term PFS with standard R-CHOP therapy is poor (20% to 40%).50,51 Treatment with more intensive first-line regimens such as DA-EPOCH-R, R-hyperCVAD (rituximab plus hyperfractionated cyclophosphamide, vincristine, doxorubicin, dexamethasone), or CODOX-M/IVAC±R (cyclophosphamide, vincristine, doxorubicin, high‐dose methotrexate/ifosfamide, etoposide, high‐dose cytarabine ± rituximab), along with CNS prophylaxis, however, has been shown to produce superior outcomes,52 with 3-year relapse-free survival rates of 88% compared to 56% for R-CHOP. For patients who achieve a complete response by PET/CT scan after intensive induction, consolidation with auto-HCT has not been shown to improve outcomes based on retrospective analysis. However for DHL/THL patients who achieve complete response after R-CHOP, PFS was improved if auto-HCT was given as consolidation of first remission.53
Patients with DLBCL have an approximately 5% risk of subsequently developing CNS involvement. Historically (in the pre-rituximab era), patients who presented with multiple sites of extranodal disease and/or extensive bone marrow involvement and/or an elevated LDH had an increased risk (up to 20%–30%) of developing CNS involvement. In addition, patients with involvement of certain anatomical sites (testicular, paranasal sinuses, epidural space) had an increased risk of CNS disease. Several algorithms have been proposed to identify patients who should receive prophylactic CNS therapy. One of the most robust tools for this purpose is the CNS-IPI, which is a 6-point score consisting of the 5 IPI elements, plus 1 additional point if the adrenal glands or kidneys are involved. Importantly, the CNS-IPI was developed and validated in patients treated with R-CHOP-like therapy. Subsequent risk of CNS relapse was 0.6%, 3.4%, and 10.2% for those with low-, intermediate- and high-risk CNS-IPI scores, respectively.54 A reasonable strategy, therefore, is to perform CNS prophylaxis in those with a CNS-IPI score of 4 to 6. When CNS prophylaxis is used, intrathecal methotrexate or high-dose systemic methotrexate is most frequently given, with high-dose systemic methotrexate favored over intrathecal chemotherapy given that high-dose methotrexate penetrates the brain and spinal cord parenchyma, in addition to treating the cerebrospinal fluid (CSF).55 In contrast, intrathecal therapy only treats the CSF and requires repeated lumbar punctures or placement of an Ommaya reservoir. For DLBCL patients who present with active CSF involvement (known as lymphomatous meningitis), intrathecal chemotherapy treatments are typically given 2 or 3 times weekly until the CSF clears, followed by weekly intrathecal treatment for 4 weeks, and then monthly intrathecal treatment for 4 months.56 For those with concurrent systemic and brain parenchymal DLBCL, a strategy of alternating R-CHOP with mid-cycle high-dose methotrexate can be successful. In addition, consolidation with high-dose therapy and auto-HCT improved survival in such patients in 1 retrospective series.57
Relapsed/Refractory Disease
Between 30% and 40% of patients with advanced stage DLBCL will either fail to attain a remission with primary therapy (referred to as primary induction failure) or will relapse. In general, for those with progressive or relapsed disease, an updated tissue biopsy is recommended. This is especially true for patients who have had prior complete remission and have new lymph node enlargement, or those who have emergence of new sites of disease at the completion of first-line therapy.
Patients with relapsed disease are treated with systemic second-line platinum-based chemoimmunotherapy, with the usual goal of ultimately proceeding to auto-HCT. A number of platinum-based regimens have been used in this setting such as R-ICE, R-DHAP, R-GDP, R-Gem-Ox, and R-ESHAP. None of these regimens has been shown to be superior in terms of efficacy, and the choice of regimen is typically made based on the anticipated tolerance of the patient in light of comorbidities, laboratory studies, and physician preference. In the CORAL study, R-DHAP (rituximab, dexamethasone, high-dose cytarabine, cisplatin) seemed to show superior PFS in patients with the GCB subtype.58 However, this was an unplanned subgroup analysis and R-DHAP was associated with higher renal toxicity.
Several studies have demonstrated that long-term PFS can be observed for relapsed/refractory DLBCL patients who respond to second-line therapy and then undergo high-dose therapy with auto-HCT. The Parma trial remains the only published prospective randomized trial performed in relapsed DLBCL comparing a transplant strategy to a non-transplant strategy. This study, performed in the pre-rituximab era, clearly showed a benefit in terms of DFS and OS in favor of auto-HCT versus salvage therapy alone.59 The benefit of auto-HCT in patients treated in the rituximab era, even in patients who experience early failure (within 1 year of diagnosis), was confirmed in a retrospective analysis by the Center for International Blood and Marrow Transplant Research. In this study, a 44% 3-year PFS was seen in the early failure cohort versus 52% in the late failure cohort.60
Some DLBCL patients are very unlikely to benefit from auto-HCT. The REFINE study focused on patients with primary induction failure or early relapse within 6 months of completing first-line therapy. Among such patients, primary progressive disease (defined as progression while still receiving first-line therapy), a high NCCN-IPI score at relapse, and MYC rearrangement were risk factors for poor PFS following auto-HCT.61 Patients with 2 or 3 high-risk features had a 2-year OS of 10.7% compared to 74.3% for those without any high-risk features.
Allogeneic HCT (allo-HCT) is a treatment option for relapsed/refractory DLBCL. This option is more commonly considered for patients in whom an autotransplant has failed to achieve durable remission. For properly selected patients in this setting, a long-term PFS in the 30% to 40% range can be attained.62 However, in practice, only about 20% of patients who fail auto-HCT end up undergoing allo-HCT due to rapid progression of disease, age, poor performance status, or lack of suitable donor. It has been proposed that in the coming years, allo-HCT will be utilized less commonly in this setting due to the advent of chimeric antigen receptor T-cell (CAR T) therapy.
CAR T-cell therapy genetically modifies the patient’s own T lymphocytes with a gene that encodes an antigen receptor to direct the T cells against lymphoma cells. Typically, the T cells are genetically modified and expanded in a production facility and then infused back into the patient. Axicabtagene ciloleucel is directed against the CD-19 receptor and has been approved by the US Food and Drug Administration (FDA) for treatment of patients with DLBCL who have failed 2 or more lines of systemic therapy. Use of CAR-T therapy in such patients was examined in a multicenter trial (ZUMA-1), which reported a 54% complete response rate and 52% OS rate at 18 months.63 CAR-T therapy is associated with serious side effects such as cytokine release syndrome, neurological toxicities, and prolonged cytopenias. While there are now some patients with ongoing remission 2 or more years after undergoing CAR-T therapy, it remains uncertain what proportion of patients have been truly cured with this modality. Nevertheless, this new treatment option remains a source of optimism for relapsed and refractory DLBCL patients.
Primary Mediastinal Large B-Cell Lymphoma
Primary mediastinal large B-cell lymphoma (PMBCL) is a form of DLBCL arising in the mediastinum from the thymic B cell. It is an uncommon entity and has clinical and pathologic features distinct from systemic DLBCL.64 PMBCL accounts for 2% of all NHLs and about 7% of all DLBCL.20 It typically affects women in the third to fourth decade of life.
Presentation and Prognostic Features
PMBCL usually presents as a locally invasive anterior mediastinal mass, often with a superior vena cava syndrome which may or may not be clinically obvious.64 Other presentations include pericardial tamponade, thrombosis of neck veins, and acute airway obstruction. About 80% of patients present with bulky (> 10 cm) stage I or II disease,65 with distant spread uncommon on presentation. Morphologically and on GEP, PMBL has a profile more similar to classical Hodgkin lymphoma (cHL) than non-mediastinal DLBCL.66 PMBL is distinguished from cHL by immunophenotyping: unlike cHL, PMBCL has pan B cell markers, rarely expresses CD15, and has weak CD30.
Poor prognostic features in PMBCL are Eastern Cooperative Oncology Group (ECOG) performance status greater than 2, pericardial effusion, bulky disease, and elevated serum LDH. The diagnosis of PMBCL can be difficult because the tumor is often encased with extensive fibrosis and necrosis. As a result, a needle biopsy may not yield sufficient tissue, thus making a surgical biopsy often the only viable way to obtain sufficient tissue.
Treatment
Early series suggested that PMBCL is unusually aggressive, with a poor prognosis.67 This led to studies using more aggressive chemotherapy regimens (often in combination with mediastinal radiation) as well as upfront auto-HCT.68–70 The addition of rituximab to treatment regimens significantly improved outcomes in PMBCL. For example, a subgroup analysis of the PMBCL patients in the MinT trial revealed a 3-year event-free survival (EFS) of 78%71 when rituximab was combined with CHOP. Because of previous reports demonstrating radiosensitivity of PMBL, radiation was traditionally sequenced into treatment regimens for PMBL. However, this is associated with higher long-term toxicities, often a concern in PMBCL patients given that the disease frequently affects younger females, and given that breast tissue will be in the radiation field. For patients with a strong personal or family history of breast cancer or cardiovascular disease, these concerns are even more significant. More recently, the DA-EPOCH-R regimen has been shown to produce very high rates (80%–90%) of long-term DFS, without the need for mediastinal radiation in most cases.72,73 For patients receiving R-CHOP, consolidation with mediastinal radiation is still commonly given. This approach also leads to high rates of long-term remission and, although utilizing mediastinal radiation, allows for less intensive chemotherapy. Determining which approach is most appropriate for an individual patient requires an assessment of the risks of each treatment option for that patient. A randomized trial by the International Extranodal Lymphoma Study Group (IELSG37) is evaluating whether RT may be safely omitted in PMBCL patients who achieve a complete metabolic response after R-CHOP.
Most relapses of PMBCL occur within the first 1 to 2 years and often present with extranodal disease in various organs. For those with relapsed or refractory disease, high-dose chemotherapy followed by auto-HCT provides 5-year survival rates of 50% to 80%.74–76 In a phase 1b trial evaluating the role of pembrolizumab in relapsed/refractory patients (KEYNOTE-13), 7 of 17 PMBCL patients achieved responses, with an additional 6 demonstrating stable disease.77 This provides an additional option for patients who might be too weak to undergo auto-HCT or for those who relapse following auto-HCT.
Mantle Cell Lymphoma
The name mantle cell lymphoma (MCL) is based on the presumed normal cell counterpart to MCL, which is believed to be found in the mantle zone surrounding germinal center follicles. It represents approximately 6% of all NHL cases in the United States and Europe.78 MCL occurs at a median age of 63 to 68 years and has a male predominance.
Presentation and Prognostic Features
Patients can present with a broad spectrum of clinical features, and most patients (70%) present with advanced disease.79 Up to one third of patients have B symptoms, with most demonstrating lymphadenopathy and bone marrow involvement. Approximately 25% present with extranodal disease as the primary presentation (eg, GI tract, pleura, breast, or orbits). MCL can involve any part of the GI tract and often presents as polypoid lesions.
Histologically, the pattern of MCL may be diffuse, nodular, mantle zone, or a combination of the these; morphologically, MCL can range from small, more irregular lymphocytes to lymphoblast-like cells. Blastoid and pleomorphic variants of MCL have a higher proliferation index and a more aggressive clinical course than other variants. MCL is characterized by the expression of pan B cell antigens (CD19+, CD20+) with coexpression of the T-cell antigen CD5, lack of CD23 expression, and nuclear expression of cyclin D1. Nuclear staining for cyclin D1 is present in more than 98% of cases.80 In rare cases, CD5 or cyclin D1 may be negative.80 Most MCL cases have a unique translocation that fuses the immunoglobulin heavy chain gene promoter (14q32) to the promoter of the BCL-1 gene (11q13), which encodes the cyclin D1 protein. This translocation is not unique to MCL and can be present in multiple myeloma as well. Interestingly, cyclin D1 is overproduced in cases lacking t(11:14), likely from other point mutations resulting in its overexpression.81 Cyclin D1–negative tumors overexpress cyclin D2 or D3, with no apparent difference in clinical behavior or outcome.82 In cyclin D1–negative cases, SOX11 expression may help with diagnosis.83 A proliferation rate greater than 30% (as measured by Ki-67 staining), low SOX11 expression, and presence of p53 mutations have all been associated with adverse outcome.
In a minority of cases, MCL follows an indolent clinical course. For the remainder, however, MCL is an aggressive disease that generally requires treatment soon after diagnosis. When initially described in the 1980s and 1990s, treatment of MCL was characterized by low complete response rates, short durations of remission, repeated recurrences, and a median survival in the 2- to 5-year range.84 In recent years, intensive regimens incorporating rituximab and high-dose cytarabine with or without auto-HCT have been developed and are associated with high complete response rates and median duration of first remission in the 6- to 9-year range.85–87 Several prognostic indices have been applied to patients with MCL, including the IPI, the Follicular Lymphoma International Prognostic Index , and the Mantle Cell Lymphoma International Prognostic Index (MIPI). The MIPI was originally described based on a cohort from the period 1996 to 2004,88 and subsequently confirmed in a separate cohort of 958 patients with MCL treated on prospective trials between 2004 and 2010.89 The MIPI score can identify 3 risk groups with significant survival differences (83%, 63%, and 34% survival at 5 years). A refined version of the MIPI score, the combined MIPI or MIPI-c, incorporates proliferation rate and is better able to stratify patients.90 The blastoid variant of MCL follows a more aggressive clinical course and is associated with a high proliferation rate, shorter remissions, and a higher rate of CNS involvement.91
In most patients, MCL is an aggressive disease with a short OS without treatment. A subset of patients may have a more indolent course,92 but unfortunately reliable factors that identify this group at the time of diagnosis are not available. Pretreatment evaluation is as with other lymphomas, with lumbar puncture and MRI of the brain also recommended for patients with the blastoid variant. For those presenting with GI symptoms, endoscopy is recommended as part of the initial evaluation as well.
Treatment
First-line Therapy
For patients under age 65 to 70 years with a good performance status and few comorbidities, an intensive induction regimen (such as R-CHOP/R-DHAP, Maxi-R-CHOP/R-araC, or R-DHAP) followed by consolidation with auto-HCT is commonly given, with a goal of achieving a durable (6–9 year) first remission.87,93,94 Auto-HCT is now routinely followed by 3 years of maintenance rituximab based on the survival benefit seen in the recent LYSA trial.93 At many centers, auto-HCT in first remission is a standard of care, with the greatest benefit seen in patients who have achieved a complete remission with no more than 2 lines of chemotherapy.95 However, there remains some controversy about whether all patients truly benefit from auto-HCT in first remission, and current research efforts are focused on identifying patients most likely to benefit from auto-HCT and incorporation of new agents into first-line regimens. For patients who are not candidates for auto-HCT, bendamustine plus rituximab (BR) or R-CHOP alone or followed by maintenance rituximab is a reasonable approach.96 Based on the StiL and BRIGHT trials, BR seems to have less toxicity and higher rates of response with no difference in OS when compared to R-CHOP.97,98
In summary, dose-intense induction chemotherapy with consolidative auto-HCT results in high rates of long-term remission and can be considered in MCL patients who lack significant comorbidities and who understand the risks and benefits of this approach. For other patients, the less aggressive frontline approaches are more appropriate.
Relapsed/Refractory Disease
Despite initial high response rates, most patients with MCL will eventually relapse. For example, most patients given CHOP or R-CHOP alone as first-line therapy will relapse within 2 years.99 In recent years, a number of therapies have emerged for relapsed/refractory MCL; however, the optimal sequencing of these is unclear. FDA-approved options for relapsed/refractory MCL include the proteasome inhibitor bortezomib,100,101 the BTK inhibitors ibrutinib102,103 and acalabrutinib,104 and the immunomodulatory agent lenalidomide.105
Auto-HCT can be considered for patients who did not undergo auto-HCT as part of first-line therapy and who had a reasonably long first remission.95 Allo-HCT has curative potential in MCL with good evidence of a graft-versus-lymphoma effect. With a matched related or matched unrelated donor, the chance for treatment-related mortality is 15% to 25% at 1 to 2 years, with a 50% to 60% chance for long-term PFS. However, given the risk of treatment-related mortality and graft-versus-host disease, this option is typically reserved for patients with early relapse after auto-HCT, multiple relapses, or relatively chemotherapy-unresponsive disease.95,106 A number of clinical trials for relapsed/refractory MCL are ongoing, and participation in these is encouraged whenever possible.
Burkitt Lymphoma
Burkitt lymphoma is a rare, aggressive and highly curable subtype of NHL. It can occur at any age, although peak incidence is in the first decade of life. There are 3 distinct clinical forms of Burkitt lymphoma.107 The endemic form is common in African children and commonly involves the jaw and kidneys. The sporadic (nonendemic) form accounts for 1% to 2% of all lymphomas in the United States and Western Europe and usually has an abdominal presentation. The immunodeficiency-associated form is commonly seen in HIV patients with a relatively preserved CD4 cell count.
Patients typically present with rapidly growing masses and tumor lysis syndrome. CNS and bone marrow involvement are common. Burkitt lymphoma cells are high-grade, rapidly proliferating medium-sized cells with a monomorphic appearance. Biopsies show a classic histological appearance known as a “starry sky pattern” due to benign macrophages engulfing debris resulting from apoptosis. It is derived from a germinal center B cell and has distinct oncogenic pathways. Translocations such as t(8;14), t(2;8) or t(8;22) juxtapose the MYC locus with immunoglobulin heavy or light chain loci and result in MYC overexpression. Burkitt lymphoma is typically CD10-positive and BCL-2-negative, with a MYC translocation and a proliferation rate greater than 95%.
With conventional NHL regimens, Burkitt lymphoma had a poor prognosis, with complete remission in the 30% to 70% range and low rates of long-term remission. With the introduction of short-term, dose-intensive, multiagent chemotherapy regimens (adapted from pediatric acute lymphoblastic leukemia [ALL] regimens), the complete remission rate improved to 60% to 90%.107 Early stage disease (localized or completely resected intra-abdominal disease) can have complete remission rates of 100%, with 2- to 5-year freedom-from-progression rates of 95%. CNS prophylaxis, including high-dose methotrexate, high-dose cytarabine, and intrathecal chemotherapy, is a standard component of Burkitt lymphoma regimens (CNS relapse rates can reach 50% without prophylactic therapy). Crucially, relapse after 1 to 2 years is very rare following complete response to induction therapy. Classically, several intensive regimens have been used for Burkitt lymphoma. In recent years, the most commonly used regimens have been the modified Magrath regimen of R-CODOX-M/IVAC and R-hyperCVAD. DA-EPOCH-R has also been used, typically for older, more frail, or HIV-positive patients. However, at the American Society of Hematology 2017 annual meeting, results from the NCI 9177 trial were presented which validated, in a prospective multi-center fashion, the use of DA-EPOCH-R in all Burkitt lymphoma patients.108 In NCI 9177, low-risk patients (defined as normal LDH, ECOG performance score 0 or 1, ≤ stage II, and no tumor lesion > 7 cm) received 2 cycles of DA-EPOCH-R without intrathecal therapy followed by PET. If interim PET was negative, low-risk patients then received 1 more cycle of DA-EPOCH-R. High-risk patients with negative brain MRI and CSF cytology/flow cytometry received 2 cycles of DA-EPOCH-R with intrathecal therapy (2 doses per cycle) followed by PET. Unless interim PET showed progression, high-risk patients received 4 additional cycles of DA-EPOCH-R including methotrexate 12 mg intrathecally on days 1 and 5 (8 total doses). With a median follow-up of 36 months, this regimen resulted in an EFS of 85.7%. As expected, patients with CNS, marrow, or peripheral blood involvement fared worse. For those without CNS, marrow, or peripheral blood involvement, the results were excellent, with an EFS of 94.6% compared to 62.8% for those with CNS, bone marrow, or blood involvement at diagnosis.
Although no standard of care has been defined, patients with relapsed/refractory Burkitt lymphoma are often given standard second-line aggressive NHL regimens (eg, R-ICE); for those with chemosensitive disease, auto- or allo-HCT is often pursued, with long-term remissions possible following HCT.109
Lymphoblastic Lymphoma
Lymphoblastic lymphoma (LBL) is a rare disease postulated to arise from precursor B or T lymphoblasts at varying stages of differentiation. Accounting for approximately 2% of all NHLs, 85% to 90% of all cases have a T-cell phenotype, while B-cell LBL comprises approximately 10% to 15% of cases. LBL and ALL are thought to represent the same disease entity, but LBL has been arbitrarily defined as cases with lymph node or mediastinal disease. Those with significant (> 25%) bone marrow or peripheral blood involvement are classified as ALL.
Precursor T-cell LBL patients are usually adolescent and young males who commonly present with a mediastinal mass and peripheral lymphadenopathy. Precursor B-cell LBL patients are usually older (median age 39 years) with peripheral lymphadenopathy and extranodal involvement. Mediastinal involvement with B-cell LBL is uncommon, and there is no male predominance. LBL has a propensity for dissemination to the bone marrow and CNS.
Morphologically, the tumor cells are medium sized, with a scant cytoplasm and finely dispersed chromatin. Mitotic features and apoptotic bodies are present since it is a high-grade malignancy. The lymphoblasts are typically positive for CD7 and either surface or cytoplasmic CD3. Terminal deoxynucleotidyl transferase expression is a defining feature. Other markers such as CD19, CD22, CD20, CD79a, CD45, and CD10 are variably expressed. Poor prognostic factors in T-cell LBL are female gender, age greater than 35 years, complex cytogenetics, and lack of a matched sibling donor.
Regimens for LBL are based on dose-dense, multi-agent protocols used in ALL. Most of these regimens are characterized by intensive remission-induction chemotherapy, CNS prophylaxis, a phase of consolidation therapy, and a prolonged maintenance phase, often lasting for 12 to 18 months with long-term DFS rates of 40% to 70%.110,111 High-dose therapy with auto-HCT or allo-HCT in first complete response has been evaluated in an attempt to reduce the incidence of relapse.112 However, the intensity of primary chemotherapy appears to be a stronger determinant of long-term survival than the use of HCT as consolidation. As a result, HCT is not routinely applied to patients in first complete remission following modern induction regimens. After relapse, prognosis is poor, with median survival rates of 6 to 9 months with conventional chemotherapy, although long-term survival rates of 30% and 20%, respectively, are reported after HCT in relapsed and primary refractory disease.113
Treatment options in relapsed disease are limited. Nelarabine can produce responses in up to 40% of relapsed/refractory LBL/ALL patients.114
Summary
Aggressive NHLs are characterized by rapid clinical progression without therapy. However, a significant proportion of patients are cured with appropriate combination chemotherapy or combined modality (chemotherapy + RT) regimens. In contrast, the indolent lymphomas have a relatively good prognosis (median survival of 10 years or longer) but usually are not curable in advanced clinical stages. Overall 5-year survival for aggressive NHLs with current treatment is approximately 50% to 60%, with relapses typically occurring within the first 5 years. Treatment strategies for relapsed patients offer some potential for cure; however, clinical trial participation should be encouraged whenever possible to investigate new approaches for improving outcomes in this patient population.
1. Swerdlow, SH, Campo E, Harris NL, et al. WHO classification of tumours of haematopoietic and lymphoid tissues. Revised 4th edition. Lyon, France: World Health Organization; 2017.
2. Surveillance, Epidemiology, and End Results (SEER) Program. www.seer.cancer.gov. Research Data 2017.
3. Boffetta P, de Vocht F. Occupation and the risk of non-Hodgkin lymphoma. Cancer Epidemiol Biomarkers Prev 2007;16:369–72.
4. Bower M. Acquired immunodeficiency syndrome-related systemic non-Hodgkin’s lymphoma. Br J Haematol 2001;112:863–73.
5. Ekstrom Smedby K, Vajdic CM, Falster M, et al. Autoimmune disorders and risk of non-Hodgkin lymphoma subtypes: a pooled analysis within the InterLymph Consortium. Blood 2008;111:4029–38.
6. Clarke CA, Morton LM, Lynch C, et al. Risk of lymphoma subtypes after solid organ transplantation in the United States. Br J Cancer 2013;109:280–8.
7. Wang SS, Slager SL, Brennan P, et al. Family history of hematopoietic malignancies and risk of non-Hodgkin lymphoma (NHL): a pooled analysis of 10 211 cases and 11 905 controls from the International Lymphoma Epidemiology Consortium (InterLymph). Blood 2007;109:3479–88.
8. Dong C, Hemminki K. Second primary neoplasms among 53 159 haematolymphoproliferative malignancy patients in Sweden, 1958–1996: a search for common mechanisms. Br J Cancer 2001;85:997–1005.
9. Hummel M, Anagnostopoulos I, Korbjuhn P, Stein H. Epstein-Barr virus in B-cell non-Hodgkin’s lymphomas: unexpected infection patterns and different infection incidence in low- and high-grade types. J Pathol 1995;175:263–71.
10. Cesarman E, Chang Y, Moore PS, et al. Kaposi’s sarcoma-associated herpesvirus-like DNA sequences in AIDS-related body-cavity-based lymphomas. N Engl J Med 1995;332:1186–91.
11. Viswanatha DS, Dogan A. Hepatitis C virus and lymphoma. J Clin Pathol 2007;60:1378–83.
12. Engels EA, Cho ER, Jee SH. Hepatitis B virus infection and risk of non-Hodgkin lymphoma in South Korea: a cohort study. Lancet Oncol 2010;11:827–34.
13. Marcucci F, Mele A. Hepatitis viruses and non-Hodgkin lymphoma: epidemiology, mechanisms of tumorigenesis, and therapeutic opportunities. Blood 2011;117:1792–8.
14. Cheson BD, Fisher RI, Barrington SF, et al. Recommendations for initial evaluation, staging, and response assessment of Hodgkin and non-Hodgkin lymphoma: the Lugano classification. J Clin Oncol 2014;32:3059–68.
15. Rosenwald A, Wright G, Chan WC, et al. The use of molecular profiling to predict survival after chemotherapy for diffuse large-B-cell lymphoma. N Engl J Med 2002;346:1937–47.
16. Teras LR, DeSantis CE, Cerhan JR, et al. 2016 US lymphoid malignancy statistics by World Health Organization subtypes. CA Cancer J Clin 2016;66:443–59.
17. Morton LM, Wang SS, Devesa SS, et al. Lymphoma incidence patterns by WHO subtype in the United States, 1992-2001. Blood 2006;107:265–76.
18. Møller MB, Pedersen NT, Christensen BE. Diffuse large B-cell lymphoma: clinical implications of extranodal versus nodal presentation--a population-based study of 1575 cases. Br J Haematol 2004;124:151–9.
19. Armitage JO, Weisenburger DD. New approach to classifying non-Hodgkin’s lymphomas: clinical features of the major histologic subtypes. Non-Hodgkin’s Lymphoma Classification Project. J Clin Oncol 1998;16:2780–95.
20. A clinical evaluation of the International Lymphoma Study Group classification of non-Hodgkin’s lymphoma. The Non-Hodgkin’s Lymphoma Classification Project. Blood 1997;89:3909–18.
21. Sehn LH, Scott DW, Chhanabhai M, et al. Impact of concordant and discordant bone marrow involvement on outcome in diffuse large B-cell lymphoma treated with R-CHOP. J Clin Oncol 2011;29:1452–7.
22. Fisher DE, Jacobson JO, Ault KA, Harris NL. Diffuse large cell lymphoma with discordant bone marrow histology. Clinical features and biological implications. Cancer 1989;64:1879–87.
23. Yao Z, Deng L, Xu-Monette ZY, et al. Concordant bone marrow involvement of diffuse large B-cell lymphoma represents a distinct clinical and biological entity in the era of immunotherapy. Leukemia 2018;32:353–63.
24. Gascoyne RD, Adomat SA, Krajewski S, et al. Prognostic significance of Bcl-2 protein expression and Bcl-2 gene rearrangement in diffuse aggressive non-Hodgkin’s lymphoma. Blood 1997;90:244–51.
25. Skinnider BF, Horsman DE, Dupuis B, Gascoyne RD. Bcl-6 and Bcl-2 protein expression in diffuse large B-cell lymphoma and follicular lymphoma: correlation with 3q27 and 18q21 chromosomal abnormalities. Hum Pathol 1999;30:803–8.
26. Chisholm KM, Bangs CD, Bacchi CE, et al. Expression profiles of MYC protein and MYC gene rearrangement in lymphomas. Am J Surg Pathol 2015;39:294–303.
27. Zhou Z, Sehn LH, Rademaker AW, et al. An enhanced International Prognostic Index (NCCN-IPI) for patients with diffuse large B-cell lymphoma treated in the rituximab era. Blood 2014;123:837–42.
28. Hans CP, Weisenburger DD, Greiner TC, et al. Confirmation of the molecular classification of diffuse large B-cell lymphoma by immunohistochemistry using a tissue microarray. Blood 2004;103:275–82.
29. Horn H, Ziepert M, Becher C, et al. MYC status in concert with BCL2 and BCL6 expression predicts outcome in diffuse large B-cell lymphoma. Blood 2013;121:2253–63.
30. Barrans S, Crouch S, Smith A, et al. Rearrangement of MYC is associated with poor prognosis in patients with diffuse large B-cell lymphoma treated in the era of rituximab. J Clin Oncol 2010;28:3360–5.
31. Hu S, Xu-Monette ZY, Tzankov A, et al. MYC/BCL2 protein coexpression contributes to the inferior survival of activated B-cell subtype of diffuse large B-cell lymphoma and demonstrates high-risk gene expression signatures: a report from The International DLBCL Rituximab-CHOP Consortium Program. Blood 2013;121:4021–31.
32. Green TM, Young KH, Visco C, et al. Immunohistochemical double-hit score is a strong predictor of outcome in patients with diffuse large B-cell lymphoma treated with rituximab plus cyclophosphamide, doxorubicin, vincristine, and prednisone. J Clin Oncol 2012;30:3460–7.
33. Fisher RI, Gaynor ER, Dahlberg S, et al. Comparison of a standard regimen (CHOP) with three intensive chemotherapy regimens for advanced non-Hodgkin’s lymphoma. N Engl J Med 1993;328:1002–6.
34. Pfreundschuh M, Kuhnt E, Trümper L, et al. CHOP-like chemotherapy with or without rituximab in young patients with good-prognosis diffuse large-B-cell lymphoma: 6-year results of an open-label randomised study of the MabThera International Trial (MInT) Group. Lancet Oncol 2011;12:1013–22.
35. Coiffier B, Lepage E, Brière J, et al. CHOP chemotherapy plus rituximab compared with CHOP alone in elderly patients with diffuse large-B-cell lymphoma. N Engl J Med 2002;346:235–42.
36. Persky DO, Unger JM, Spier CM, et al. Phase II study of rituximab plus three cycles of CHOP and involved-field radiotherapy for patients with limited-stage aggressive B-cell lymphoma: Southwest Oncology Group study 0014. J Clin Oncol 2008;26:2258–63.
37. Lamy T, Damaj G, Soubeyran P, et al. R-CHOP 14 with or without radiotherapy in nonbulky limited-stage diffuse large B-cell lymphoma. Blood 2018;131:174–81.
38. Peyrade F, Jardin F, Thieblemont C, et al. Attenuated immunochemotherapy regimen (R-miniCHOP) in elderly patients older than 80 years with diffuse large B-cell lymphoma: a multicentre, single-arm, phase 2 trial. Lancet Oncol 2011;12:460–8.
39. Wilson WH, sin-Ho J, Pitcher BN, et al. Phase III randomized study of R-CHOP versus DA-EPOCH-R and molecular analysis of untreated diffuse large B-cell lymphoma: CALGB/Alliance 50303. Blood 2016;128:469 LP-469. 38.
40. Vitolo U, Trne˘ný M, Belada D, et al. Obinutuzumab or rituximab plus cyclophosphamide, doxorubicin, vincristine, and prednisone in previously untreated diffuse large B-cell lymphoma. J Clin Oncol 2017;35:3529–37.
41. Leonard JP, Kolibaba KS, Reeves JA, et al. Randomized phase II study of R-CHOP with or without bortezomib in previously untreated patients with non-germinal center B-cell-like diffuse large B-cell lymphoma. J Clin Oncol 2017;35:3538–46.
42. Nowakowski GS, LaPlant B, Macon WR, et al. Lenalidomide combined with R-CHOP overcomes negative prognostic impact of non-germinal center B-cell phenotype in newly diagnosed diffuse large B-Cell lymphoma: a phase II study. J Clin Oncol 2015;33:251–7.
43. Younes A, Thieblemont C, Morschhauser F, et al. Combination of ibrutinib with rituximab, cyclophosphamide, doxorubicin, vincristine, and prednisone (R-CHOP) for treatment-naive patients with CD20-positive B-cell non-Hodgkin lymphoma: a non-randomised, phase 1b study. Lancet Oncol 2014;15:1019–26.
44. Younes A, Zinzani PL, Sehn LH, et al. A randomized, double-blind, placebo-controlled phase 3 study of ibrutinib in combination with rituximab, cyclophosphamide, doxorubicin, vincristine, and prednisone (R-CHOP) in subjects with newly diagnosed nongerminal center B-cell subtype of diffuse large B-cell lymphoma (DLBCL). J Clin Oncol 2014;32(15_suppl):TPS8615.
45. Delarue R, Tilly H, Mounier N, et al. Dose-dense rituximab-CHOP compared with standard rituximab-CHOP in elderly patients with diffuse large B-cell lymphoma (the LNH03-6B study): a randomised phase 3 trial. Lancet Oncol 2013;14:525–33.
46. Leppä S, Fayad LE, Lee J-J, et al. A phase III study of enzastaurin in patients with high-risk diffuse large B cell lymphoma following response to primary treatment: the Prelude trial. Blood 2013;122:371 LP-371.
47. Witzig TE, Tobinai K, Rigacci L, et al. Adjuvant everolimus in high-risk diffuse large B-cell lymphoma: final results from the PILLAR-2 randomized phase III trial. Ann Oncol 2018;29:707–14.
48. Strehl J, Mey U, Glasmacher A, et al. High-dose chemotherapy followed by autologous stem cell transplantation as first-line therapy in aggressive non-Hodgkin’s lymphoma: a meta-analysis. Haematologica 2003;88:1304–15.
49. Stiff PJ, Unger JM, Cook JR, et al. Autologous transplantation as consolidation for aggressive non-Hodgkin’s lymphoma. N Engl J Med 2013;369:1681–90.
50. Oki Y, Noorani M, Lin P, et al. Double hit lymphoma: the MD Anderson Cancer Center clinical experience. Br J Haematol 2014;166:891–901.
51. Petrich AM, Gandhi M, Jovanovic B, et al. Impact of induction regimen and stem cell transplantation on outcomes in double-hit lymphoma: a multicenter retrospective analysis. Blood 2014;124:2354–61.
52. Howlett C, Snedecor SJ, Landsburg DJ, et al. Front-line, dose-escalated immunochemotherapy is associated with a significant progression-free survival advantage in patients with double-hit lymphomas: a systematic review and meta-analysis. Br J Haematol 2015;170:504–14.
53. Landsburg DJ, Falkiewicz MK, Maly J, et al. Outcomes of patients with double-hit lymphoma who achieve first complete remission. J Clin Oncol 2017;35:2260–7.
54. Schmitz N, Zeynalova S, Nickelsen M, et al. CNS International Prognostic Index: a risk model for CNS relapse in patients with diffuse large B-cell lymphoma treated with R-CHOP. J Clin Oncol 2016;34:3150–6.
55. Abramson JS, Hellmann M, Barnes JA, et al. Intravenous methotrexate as central nervous system (CNS) prophylaxis is associated with a low risk of CNS recurrence in high-risk patients with diffuse large B-cell lymphoma. Cancer 2010;116:4283–90.
56. Dunleavy K, Roschewski M, Abramson JS, et al. Risk-adapted therapy in adults with Burkitt lymphoma: updated results of a multicenter prospective phase II study of DA-EPOCH-R. Hematol Oncol 2017;35(S2):133–4.
57. Damaj G, Ivanoff S, Coso D, et al. Concomitant systemic and central nervous system non-Hodgkin lymphoma: the role of consolidation in terms of high dose therapy and autologous stem cell transplantation. A 60-case retrospective study from LYSA and the LOC network. Haematologica 2015;100:1199–206.
58. Thieblemont C, Briere J, Mounier N, et al. The germinal center/activated B-cell subclassification has a prognostic impact for response to salvage therapy in relapsed/refractory diffuse large B-cell lymphoma: a bio-CORAL study. J Clin Oncol 2011;29:4079–87.
59. Philip T, Guglielmi C, Hagenbeek A, et al. Autologous bone marrow transplantation as compared with dalvage vhemotherapy in relapses of chemotherapy-densitive non-Hodgkin’s lymphoma. N Engl J Med 1995;333:1540–5.
60. Hamadani M, Hari PN, Zhang Y, et al. Early failure of frontline rituximab-containing chemo-immunotherapy in diffuse large B cell lymphoma does not predict futility of autologous hematopoietic cell transplantation. Biol Blood Marrow Transplant 2014;20:1729–36.
61. Costa LJ, Maddocks K, Epperla N, et al. Diffuse large B-cell lymphoma with primary treatment failure: Ultra-high risk features and benchmarking for experimental therapies. Am J Hematol 2017;92:e24615.
62. Fenske TS, Ahn KW, Graff TM, et al. Allogeneic transplantation provides durable remission in a subset of DLBCL patients relapsing after autologous transplantation. Br J Haematol 2016;174:235–48.
63. Neelapu SS, Locke FL, Bartlett NL, et al. Axicabtagene ciloleucel CAR T-cell therapy in refractory large B-cell lymphoma. N Engl J Med 2017;377:2531–44.
64. van Besien K, Kelta M, Bahaguna P. Primary mediastinal B-cell lymphoma: a review of pathology and management. J Clin Oncol 2001;19:1855–64.
65. Savage KJ, Al-Rajhi N, Voss N, et al. Favorable outcome of primary mediastinal large B-cell lymphoma in a single institution: the British Columbia experience. Ann Oncol Off J Eur Soc Med Oncol 2006;17:123–30.
66. Rosenwald A, Wright G, Leroy K, et al. Molecular diagnosis of primary mediastinal B cell lymphoma identifies a clinically favorable subgroup of diffuse large B cell lymphoma related to Hodgkin lymphoma. J Exp Med 2003;198:851–62.
67. Lavabre-Bertrand T, Donadio D, Fegueux N, et al. A study of 15 cases of primary mediastinal lymphoma of B-cell type. Cancer 1992;69:2561–6.
68. Lazzarino M, Orlandi E, Paulli M, et al. Treatment outcome and prognostic factors for primary mediastinal (thymic) B-cell lymphoma: a multicenter study of 106 patients. J Clin Oncol 1997;15:1646–53.
69. Zinzani PL, Martelli M, Magagnoli M, et al. Treatment and clinical management of primary mediastinal large B-cell lymphoma with sclerosis: MACOP-B regimen and mediastinal radiotherapy monitored by (67)Gallium scan in 50 patients. Blood 1999;94:3289–93.
70. Todeschini G, Secchi S, Morra E, et al. Primary mediastinal large B-cell lymphoma (PMLBCL): long-term results from a retrospective multicentre Italian experience in 138 patients treated with CHOP or MACOP-B/VACOP-B. Br J Cancer 2004;90:372–6.
71. Rieger M, Osterborg A, Pettengell R, et al. Primary mediastinal B-cell lymphoma treated with CHOP-like chemotherapy with or without rituximab: results of the Mabthera International Trial Group study. Ann Oncol Off J Eur Soc Med Oncol 2011;22:664–70.
72. Shah NN, Szabo A, Huntington SF, et al. R-CHOP versus dose-adjusted R-EPOCH in frontline management of primary mediastinal B-cell lymphoma: a multi-centre analysis. Br J Haematol 2018;180:534–44.
73. Dunleavy K, Pittaluga S, Maeda LS, et al. Dose-adjusted EPOCH-rituximab therapy in primary mediastinal B-cell lymphoma. N Engl J Med 2013;368:1408–16.
74. Aoki T, Shimada K, Suzuki R, et al. High-dose chemotherapy followed by autologous stem cell transplantation for relapsed/refractory primary mediastinal large B-cell lymphoma. Blood Cancer J 2015;5:e372–e372.
75. Sehn LH, Antin JH, Shulman LN, et al. Primary diffuse large B-cell lymphoma of the mediastinum: outcome following high-dose chemotherapy and autologous hematopoietic cell transplantation. Blood 1998;91:717–23.
76. Kuruvilla J, Pintilie M, Tsang R, et al. Salvage chemotherapy and autologous stem cell transplantation are inferior for relapsed or refractory primary mediastinal large B-cell lymphoma compared with diffuse large B-cell lymphoma. Leuk Lymphoma 2008;49:1329–36.
77. Zinzani PL, Ribrag V, Moskowitz CH, et al. Safety and tolerability of pembrolizumab in patients with relapsed/refractory primary mediastinal large B-cell lymphoma. Blood 2017;130:267–70.
78. Smith A, Howell D, Patmore R, et al. Incidence of haematological malignancy by sub-type: a report from the Haematological Malignancy Research Network. Br J Cancer 2011;105:1684–92.
79. Argatoff LH, Connors JM, Klasa RJ, et al. Mantle cell lymphoma: a clinicopathologic study of 80 cases. Blood 1997;89:2067–78.
80. Zukerberg LR, Yang WI, Arnold A, Harris NL. Cyclin D1 expression in non-Hodgkin’s lymphomas. Detection by immunohistochemistry. Am J Clin Pathol 1995;103:756–60.
81. Wiestner A, Tehrani M, Chiorazzi M, et al. Point mutations and genomic deletions in CCND1 create stable truncated cyclin D1 mRNAs that are associated with increased proliferation rate and shorter survival. Blood 2007;109:4599–606.
82. Fu K, Weisenburger DD, Greiner TC, et al. Cyclin D1-negative mantle cell lymphoma: a clinicopathologic study based on gene expression profiling. Blood 2005;106:4315–21.
83. Mozos A, Royo C, Hartmann E, et al. SOX11 expression is highly specific for mantle cell lymphoma and identifies the cyclin D1-negative subtype. Haematologica 2009;94:1555–62.
84. Norton AJ, Matthews J, Pappa V, et al. Mantle cell lymphoma: Natural history defined in a serially biopsied population over a 20-year period. Ann Oncol 1995;6:249–56.
85. Chihara D, Cheah CY, Westin JR, et al. Rituximab plus hyper-CVAD alternating with MTX/Ara-C in patients with newly diagnosed mantle cell lymphoma: 15-year follow-up of a phase II study from the MD Anderson Cancer Center. Br J Haematol 2016;172:80–8.
86. Delarue R, Haioun C, Ribrag V, et al. CHOP and DHAP plus rituximab followed by autologous stem cell transplantation in mantle cell lymphoma: a phase 2 study from the Groupe d’Etude des Lymphomes de l’Adulte. Blood 2013;121:48–53.
87. Eskelund CW, Kolstad A, Jerkeman M, et al. 15-year follow-up of the Second Nordic Mantle Cell Lymphoma trial (MCL2): prolonged remissions without survival plateau. Br J Haematol 2016;175:410–8.
88. Hoster E, Dreyling M, Klapper W, et al. A new prognostic index (MIPI) for patients with advanced-stage mantle cell lymphoma. Blood 2008;111:558–65.
89. Hoster E, Klapper W, Hermine O, et al. Confirmation of the mantle-cell lymphoma International Prognostic Index in randomized trials of the European Mantle-Cell Lymphoma Network. J Clin Oncol 2014;32:1338–46.
90. Hoster E, Rosenwald A, Berger F, et al. Prognostic value of Ki-67 index, cytology, and growth pattern in mantle-cell lymphoma: Results from randomized trials of the European Mantle Cell Lymphoma Network. J Clin Oncol 2016;34:1386–94.
91. Bernard M, Gressin R, Lefrère F, et al. Blastic variant of mantle cell lymphoma: a rare but highly aggressive subtype. Leukemia 2001;15:1785–91.
92. Martin P, Chadburn A, Christos P, et al. Outcome of deferred initial therapy in mantle-cell lymphoma. J Clin Oncol 2009;27:1209–13.
93. Le Gouill S, Thieblemont C, Oberic L, et al. Rituximab after autologous stem-cell transplantation in mantle-cell lymphoma. N Engl J Med. 2017 Sep 28;377(13):1250–60.
94. Hermine O, Hoster E, Walewski J, et al. Addition of high-dose cytarabine to immunochemotherapy before autologous stem-cell transplantation in patients aged 65 years or younger with mantle cell lymphoma (MCL Younger): a randomised, open-label, phase 3 trial of the European Mantle Cell Lymphoma Network. Lancet 2016;388:565–75.
95. Fenske TS, Zhang M-J, Carreras J, et al. Autologous or reduced-intensity conditioning allogeneic hematopoietic cell transplantation for chemotherapy-sensitive mantle-cell lymphoma: analysis of transplantation timing and modality. J Clin Oncol 2014;32:273–81.
96. Kluin-Nelemans HC, Hoster E, Hermine O, et al. Treatment of older patients with mantle-cell lymphoma. N Engl J Med 2012;367:520–31.
97. Flinn IW, van der Jagt R, Kahl BS, et al. Randomized trial of bendamustine-rituximab or R-CHOP/R-CVP in first-line treatment of indolent NHL or MCL: the BRIGHT study. Blood 2014;123:2944–52.
98. Rummel MJ, Niederle N, Maschmeyer G, et al. Bendamustine plus rituximab versus CHOP plus rituximab as first-line treatment for patients with indolent and mantle-cell lymphomas: an open-label, multicentre, randomised, phase 3 non-inferiority trial. Lancet 2013;381:1203–10.
99. Lenz G, Dreyling M, Hoster E, et al. Immunochemotherapy with rituximab and cyclophosphamide, doxorubicin, vincristine, and prednisone significantly improves response and time to treatment failure, but not long-term outcome in patients with previously untreated mantle cell lymphoma: results of a prospective randomized trial of the German Low Grade Lymphoma Study Group (GLSG). J Clin Oncol 2005;23:1984–92.
100. Belch A, Kouroukis CT, Crump M, et al. A phase II study of bortezomib in mantle cell lymphoma: the National Cancer Institute of Canada Clinical Trials Group trial IND.150. Ann Oncol Off J Eur Soc Med Oncol 2007;18:116–21.
101. Fisher RI, Bernstein SH, Kahl BS, et al. Multicenter phase II study of bortezomib in patients with relapsed or refractory mantle cell lymphoma. J Clin Oncol 2006;24:4867–74.
102. Dreyling M, Jurczak W, Jerkeman M, et al. Ibrutinib versus temsirolimus in patients with relapsed or refractory mantle-cell lymphoma: an international, randomised, open-label, phase 3 study. Lancet 2016;387:770–8.
103. Wang ML, Rule S, Martin P, Goy A, et al. Targeting BTK with ibrutinib in relapsed or refractory mantle-cell lymphoma. N Engl J Med 2013;369:507–16.
104. Wang M, Rule S, Zinzani PL, et al. Acalabrutinib in relapsed or refractory mantle cell lymphoma (ACE-LY-004): a single-arm, multicentre, phase 2 trial. Lancet 2018;391:659–67.
105. Goy A, Sinha R, Williams ME, et al. Single-agent lenalidomide in patients with mantle-cell lymphoma who relapsed or progressed after or were refractory to bortezomib: phase II MCL-001 (EMERGE) study. J Clin Oncol 2013;31:3688–95.
106. Khouri IF, Lee M-S, Saliba RM, et al. Nonablative allogeneic stem-cell transplantation for advanced/recurrent mantle-cell lymphoma. J Clin Oncol 2003;21:4407–12.
107. Blum KA, Lozanski G, Byrd JC. Adult Burkitt leukemia and lymphoma. Blood 2004;104:3009–20.
108. Roschewski M, Dunleavy K, Abramson JS, et al. Risk-adapted therapy in adults with Burkitt lymphoma: results of NCI 9177, a multicenter prospective phase II study of DA-EPOCH-R. Blood American Society of Hematology;2017;130(Suppl 1):188.
109. Maramattom L V, Hari PN, Burns LJ, et al. Autologous and allogeneic transplantation for burkitt lymphoma outcomes and changes in utilization: a report from the center for international blood and marrow transplant research. Biol Blood Marrow Transplant 2013;19:173–9.
110. Zinzani PL, Bendandi M, Visani G, et al. Adult lymphoblastic lymphoma: clinical features and prognostic factors in 53 patients. Leuk Lymphoma 1996;23:577–82.
111. Thomas DA, O’Brien S, Cortes J, et al. Outcome with the hyper-CVAD regimens in lymphoblastic lymphoma. Blood 2004;104:1624–30.
112. Aljurf M, Zaidi SZA. Chemotherapy and hematopoietic stem cell transplantation for adult T-cell lymphoblastic lymphoma: current status and controversies. Biol Blood Marrow Transplant 2005;11:739–54.
113. Sweetenham JW, Santini G, Qian W, et al. High-dose therapy and autologous stem-cell transplantation versus conventional-dose consolidation/maintenance therapy as postremission therapy for adult patients with lymphoblastic lymphoma: results of a randomized trial of the European Group for Blood and Marrow Transplantation and the United Kingdom Lymphoma Group. J Clin Oncol 2001;19:2927–36.
114. Zwaan CM, Kowalczyk J, Schmitt C, et al. Safety and efficacy of nelarabine in children and young adults with relapsed or refractory T-lineage acute lymphoblastic leukaemia or T-lineage lymphoblastic lymphoma: results of a phase 4 study. Br J Haematol 2017;179:284–93.
1. Swerdlow, SH, Campo E, Harris NL, et al. WHO classification of tumours of haematopoietic and lymphoid tissues. Revised 4th edition. Lyon, France: World Health Organization; 2017.
2. Surveillance, Epidemiology, and End Results (SEER) Program. www.seer.cancer.gov. Research Data 2017.
3. Boffetta P, de Vocht F. Occupation and the risk of non-Hodgkin lymphoma. Cancer Epidemiol Biomarkers Prev 2007;16:369–72.
4. Bower M. Acquired immunodeficiency syndrome-related systemic non-Hodgkin’s lymphoma. Br J Haematol 2001;112:863–73.
5. Ekstrom Smedby K, Vajdic CM, Falster M, et al. Autoimmune disorders and risk of non-Hodgkin lymphoma subtypes: a pooled analysis within the InterLymph Consortium. Blood 2008;111:4029–38.
6. Clarke CA, Morton LM, Lynch C, et al. Risk of lymphoma subtypes after solid organ transplantation in the United States. Br J Cancer 2013;109:280–8.
7. Wang SS, Slager SL, Brennan P, et al. Family history of hematopoietic malignancies and risk of non-Hodgkin lymphoma (NHL): a pooled analysis of 10 211 cases and 11 905 controls from the International Lymphoma Epidemiology Consortium (InterLymph). Blood 2007;109:3479–88.
8. Dong C, Hemminki K. Second primary neoplasms among 53 159 haematolymphoproliferative malignancy patients in Sweden, 1958–1996: a search for common mechanisms. Br J Cancer 2001;85:997–1005.
9. Hummel M, Anagnostopoulos I, Korbjuhn P, Stein H. Epstein-Barr virus in B-cell non-Hodgkin’s lymphomas: unexpected infection patterns and different infection incidence in low- and high-grade types. J Pathol 1995;175:263–71.
10. Cesarman E, Chang Y, Moore PS, et al. Kaposi’s sarcoma-associated herpesvirus-like DNA sequences in AIDS-related body-cavity-based lymphomas. N Engl J Med 1995;332:1186–91.
11. Viswanatha DS, Dogan A. Hepatitis C virus and lymphoma. J Clin Pathol 2007;60:1378–83.
12. Engels EA, Cho ER, Jee SH. Hepatitis B virus infection and risk of non-Hodgkin lymphoma in South Korea: a cohort study. Lancet Oncol 2010;11:827–34.
13. Marcucci F, Mele A. Hepatitis viruses and non-Hodgkin lymphoma: epidemiology, mechanisms of tumorigenesis, and therapeutic opportunities. Blood 2011;117:1792–8.
14. Cheson BD, Fisher RI, Barrington SF, et al. Recommendations for initial evaluation, staging, and response assessment of Hodgkin and non-Hodgkin lymphoma: the Lugano classification. J Clin Oncol 2014;32:3059–68.
15. Rosenwald A, Wright G, Chan WC, et al. The use of molecular profiling to predict survival after chemotherapy for diffuse large-B-cell lymphoma. N Engl J Med 2002;346:1937–47.
16. Teras LR, DeSantis CE, Cerhan JR, et al. 2016 US lymphoid malignancy statistics by World Health Organization subtypes. CA Cancer J Clin 2016;66:443–59.
17. Morton LM, Wang SS, Devesa SS, et al. Lymphoma incidence patterns by WHO subtype in the United States, 1992-2001. Blood 2006;107:265–76.
18. Møller MB, Pedersen NT, Christensen BE. Diffuse large B-cell lymphoma: clinical implications of extranodal versus nodal presentation--a population-based study of 1575 cases. Br J Haematol 2004;124:151–9.
19. Armitage JO, Weisenburger DD. New approach to classifying non-Hodgkin’s lymphomas: clinical features of the major histologic subtypes. Non-Hodgkin’s Lymphoma Classification Project. J Clin Oncol 1998;16:2780–95.
20. A clinical evaluation of the International Lymphoma Study Group classification of non-Hodgkin’s lymphoma. The Non-Hodgkin’s Lymphoma Classification Project. Blood 1997;89:3909–18.
21. Sehn LH, Scott DW, Chhanabhai M, et al. Impact of concordant and discordant bone marrow involvement on outcome in diffuse large B-cell lymphoma treated with R-CHOP. J Clin Oncol 2011;29:1452–7.
22. Fisher DE, Jacobson JO, Ault KA, Harris NL. Diffuse large cell lymphoma with discordant bone marrow histology. Clinical features and biological implications. Cancer 1989;64:1879–87.
23. Yao Z, Deng L, Xu-Monette ZY, et al. Concordant bone marrow involvement of diffuse large B-cell lymphoma represents a distinct clinical and biological entity in the era of immunotherapy. Leukemia 2018;32:353–63.
24. Gascoyne RD, Adomat SA, Krajewski S, et al. Prognostic significance of Bcl-2 protein expression and Bcl-2 gene rearrangement in diffuse aggressive non-Hodgkin’s lymphoma. Blood 1997;90:244–51.
25. Skinnider BF, Horsman DE, Dupuis B, Gascoyne RD. Bcl-6 and Bcl-2 protein expression in diffuse large B-cell lymphoma and follicular lymphoma: correlation with 3q27 and 18q21 chromosomal abnormalities. Hum Pathol 1999;30:803–8.
26. Chisholm KM, Bangs CD, Bacchi CE, et al. Expression profiles of MYC protein and MYC gene rearrangement in lymphomas. Am J Surg Pathol 2015;39:294–303.
27. Zhou Z, Sehn LH, Rademaker AW, et al. An enhanced International Prognostic Index (NCCN-IPI) for patients with diffuse large B-cell lymphoma treated in the rituximab era. Blood 2014;123:837–42.
28. Hans CP, Weisenburger DD, Greiner TC, et al. Confirmation of the molecular classification of diffuse large B-cell lymphoma by immunohistochemistry using a tissue microarray. Blood 2004;103:275–82.
29. Horn H, Ziepert M, Becher C, et al. MYC status in concert with BCL2 and BCL6 expression predicts outcome in diffuse large B-cell lymphoma. Blood 2013;121:2253–63.
30. Barrans S, Crouch S, Smith A, et al. Rearrangement of MYC is associated with poor prognosis in patients with diffuse large B-cell lymphoma treated in the era of rituximab. J Clin Oncol 2010;28:3360–5.
31. Hu S, Xu-Monette ZY, Tzankov A, et al. MYC/BCL2 protein coexpression contributes to the inferior survival of activated B-cell subtype of diffuse large B-cell lymphoma and demonstrates high-risk gene expression signatures: a report from The International DLBCL Rituximab-CHOP Consortium Program. Blood 2013;121:4021–31.
32. Green TM, Young KH, Visco C, et al. Immunohistochemical double-hit score is a strong predictor of outcome in patients with diffuse large B-cell lymphoma treated with rituximab plus cyclophosphamide, doxorubicin, vincristine, and prednisone. J Clin Oncol 2012;30:3460–7.
33. Fisher RI, Gaynor ER, Dahlberg S, et al. Comparison of a standard regimen (CHOP) with three intensive chemotherapy regimens for advanced non-Hodgkin’s lymphoma. N Engl J Med 1993;328:1002–6.
34. Pfreundschuh M, Kuhnt E, Trümper L, et al. CHOP-like chemotherapy with or without rituximab in young patients with good-prognosis diffuse large-B-cell lymphoma: 6-year results of an open-label randomised study of the MabThera International Trial (MInT) Group. Lancet Oncol 2011;12:1013–22.
35. Coiffier B, Lepage E, Brière J, et al. CHOP chemotherapy plus rituximab compared with CHOP alone in elderly patients with diffuse large-B-cell lymphoma. N Engl J Med 2002;346:235–42.
36. Persky DO, Unger JM, Spier CM, et al. Phase II study of rituximab plus three cycles of CHOP and involved-field radiotherapy for patients with limited-stage aggressive B-cell lymphoma: Southwest Oncology Group study 0014. J Clin Oncol 2008;26:2258–63.
37. Lamy T, Damaj G, Soubeyran P, et al. R-CHOP 14 with or without radiotherapy in nonbulky limited-stage diffuse large B-cell lymphoma. Blood 2018;131:174–81.
38. Peyrade F, Jardin F, Thieblemont C, et al. Attenuated immunochemotherapy regimen (R-miniCHOP) in elderly patients older than 80 years with diffuse large B-cell lymphoma: a multicentre, single-arm, phase 2 trial. Lancet Oncol 2011;12:460–8.
39. Wilson WH, sin-Ho J, Pitcher BN, et al. Phase III randomized study of R-CHOP versus DA-EPOCH-R and molecular analysis of untreated diffuse large B-cell lymphoma: CALGB/Alliance 50303. Blood 2016;128:469 LP-469. 38.
40. Vitolo U, Trne˘ný M, Belada D, et al. Obinutuzumab or rituximab plus cyclophosphamide, doxorubicin, vincristine, and prednisone in previously untreated diffuse large B-cell lymphoma. J Clin Oncol 2017;35:3529–37.
41. Leonard JP, Kolibaba KS, Reeves JA, et al. Randomized phase II study of R-CHOP with or without bortezomib in previously untreated patients with non-germinal center B-cell-like diffuse large B-cell lymphoma. J Clin Oncol 2017;35:3538–46.
42. Nowakowski GS, LaPlant B, Macon WR, et al. Lenalidomide combined with R-CHOP overcomes negative prognostic impact of non-germinal center B-cell phenotype in newly diagnosed diffuse large B-Cell lymphoma: a phase II study. J Clin Oncol 2015;33:251–7.
43. Younes A, Thieblemont C, Morschhauser F, et al. Combination of ibrutinib with rituximab, cyclophosphamide, doxorubicin, vincristine, and prednisone (R-CHOP) for treatment-naive patients with CD20-positive B-cell non-Hodgkin lymphoma: a non-randomised, phase 1b study. Lancet Oncol 2014;15:1019–26.
44. Younes A, Zinzani PL, Sehn LH, et al. A randomized, double-blind, placebo-controlled phase 3 study of ibrutinib in combination with rituximab, cyclophosphamide, doxorubicin, vincristine, and prednisone (R-CHOP) in subjects with newly diagnosed nongerminal center B-cell subtype of diffuse large B-cell lymphoma (DLBCL). J Clin Oncol 2014;32(15_suppl):TPS8615.
45. Delarue R, Tilly H, Mounier N, et al. Dose-dense rituximab-CHOP compared with standard rituximab-CHOP in elderly patients with diffuse large B-cell lymphoma (the LNH03-6B study): a randomised phase 3 trial. Lancet Oncol 2013;14:525–33.
46. Leppä S, Fayad LE, Lee J-J, et al. A phase III study of enzastaurin in patients with high-risk diffuse large B cell lymphoma following response to primary treatment: the Prelude trial. Blood 2013;122:371 LP-371.
47. Witzig TE, Tobinai K, Rigacci L, et al. Adjuvant everolimus in high-risk diffuse large B-cell lymphoma: final results from the PILLAR-2 randomized phase III trial. Ann Oncol 2018;29:707–14.
48. Strehl J, Mey U, Glasmacher A, et al. High-dose chemotherapy followed by autologous stem cell transplantation as first-line therapy in aggressive non-Hodgkin’s lymphoma: a meta-analysis. Haematologica 2003;88:1304–15.
49. Stiff PJ, Unger JM, Cook JR, et al. Autologous transplantation as consolidation for aggressive non-Hodgkin’s lymphoma. N Engl J Med 2013;369:1681–90.
50. Oki Y, Noorani M, Lin P, et al. Double hit lymphoma: the MD Anderson Cancer Center clinical experience. Br J Haematol 2014;166:891–901.
51. Petrich AM, Gandhi M, Jovanovic B, et al. Impact of induction regimen and stem cell transplantation on outcomes in double-hit lymphoma: a multicenter retrospective analysis. Blood 2014;124:2354–61.
52. Howlett C, Snedecor SJ, Landsburg DJ, et al. Front-line, dose-escalated immunochemotherapy is associated with a significant progression-free survival advantage in patients with double-hit lymphomas: a systematic review and meta-analysis. Br J Haematol 2015;170:504–14.
53. Landsburg DJ, Falkiewicz MK, Maly J, et al. Outcomes of patients with double-hit lymphoma who achieve first complete remission. J Clin Oncol 2017;35:2260–7.
54. Schmitz N, Zeynalova S, Nickelsen M, et al. CNS International Prognostic Index: a risk model for CNS relapse in patients with diffuse large B-cell lymphoma treated with R-CHOP. J Clin Oncol 2016;34:3150–6.
55. Abramson JS, Hellmann M, Barnes JA, et al. Intravenous methotrexate as central nervous system (CNS) prophylaxis is associated with a low risk of CNS recurrence in high-risk patients with diffuse large B-cell lymphoma. Cancer 2010;116:4283–90.
56. Dunleavy K, Roschewski M, Abramson JS, et al. Risk-adapted therapy in adults with Burkitt lymphoma: updated results of a multicenter prospective phase II study of DA-EPOCH-R. Hematol Oncol 2017;35(S2):133–4.
57. Damaj G, Ivanoff S, Coso D, et al. Concomitant systemic and central nervous system non-Hodgkin lymphoma: the role of consolidation in terms of high dose therapy and autologous stem cell transplantation. A 60-case retrospective study from LYSA and the LOC network. Haematologica 2015;100:1199–206.
58. Thieblemont C, Briere J, Mounier N, et al. The germinal center/activated B-cell subclassification has a prognostic impact for response to salvage therapy in relapsed/refractory diffuse large B-cell lymphoma: a bio-CORAL study. J Clin Oncol 2011;29:4079–87.
59. Philip T, Guglielmi C, Hagenbeek A, et al. Autologous bone marrow transplantation as compared with dalvage vhemotherapy in relapses of chemotherapy-densitive non-Hodgkin’s lymphoma. N Engl J Med 1995;333:1540–5.
60. Hamadani M, Hari PN, Zhang Y, et al. Early failure of frontline rituximab-containing chemo-immunotherapy in diffuse large B cell lymphoma does not predict futility of autologous hematopoietic cell transplantation. Biol Blood Marrow Transplant 2014;20:1729–36.
61. Costa LJ, Maddocks K, Epperla N, et al. Diffuse large B-cell lymphoma with primary treatment failure: Ultra-high risk features and benchmarking for experimental therapies. Am J Hematol 2017;92:e24615.
62. Fenske TS, Ahn KW, Graff TM, et al. Allogeneic transplantation provides durable remission in a subset of DLBCL patients relapsing after autologous transplantation. Br J Haematol 2016;174:235–48.
63. Neelapu SS, Locke FL, Bartlett NL, et al. Axicabtagene ciloleucel CAR T-cell therapy in refractory large B-cell lymphoma. N Engl J Med 2017;377:2531–44.
64. van Besien K, Kelta M, Bahaguna P. Primary mediastinal B-cell lymphoma: a review of pathology and management. J Clin Oncol 2001;19:1855–64.
65. Savage KJ, Al-Rajhi N, Voss N, et al. Favorable outcome of primary mediastinal large B-cell lymphoma in a single institution: the British Columbia experience. Ann Oncol Off J Eur Soc Med Oncol 2006;17:123–30.
66. Rosenwald A, Wright G, Leroy K, et al. Molecular diagnosis of primary mediastinal B cell lymphoma identifies a clinically favorable subgroup of diffuse large B cell lymphoma related to Hodgkin lymphoma. J Exp Med 2003;198:851–62.
67. Lavabre-Bertrand T, Donadio D, Fegueux N, et al. A study of 15 cases of primary mediastinal lymphoma of B-cell type. Cancer 1992;69:2561–6.
68. Lazzarino M, Orlandi E, Paulli M, et al. Treatment outcome and prognostic factors for primary mediastinal (thymic) B-cell lymphoma: a multicenter study of 106 patients. J Clin Oncol 1997;15:1646–53.
69. Zinzani PL, Martelli M, Magagnoli M, et al. Treatment and clinical management of primary mediastinal large B-cell lymphoma with sclerosis: MACOP-B regimen and mediastinal radiotherapy monitored by (67)Gallium scan in 50 patients. Blood 1999;94:3289–93.
70. Todeschini G, Secchi S, Morra E, et al. Primary mediastinal large B-cell lymphoma (PMLBCL): long-term results from a retrospective multicentre Italian experience in 138 patients treated with CHOP or MACOP-B/VACOP-B. Br J Cancer 2004;90:372–6.
71. Rieger M, Osterborg A, Pettengell R, et al. Primary mediastinal B-cell lymphoma treated with CHOP-like chemotherapy with or without rituximab: results of the Mabthera International Trial Group study. Ann Oncol Off J Eur Soc Med Oncol 2011;22:664–70.
72. Shah NN, Szabo A, Huntington SF, et al. R-CHOP versus dose-adjusted R-EPOCH in frontline management of primary mediastinal B-cell lymphoma: a multi-centre analysis. Br J Haematol 2018;180:534–44.
73. Dunleavy K, Pittaluga S, Maeda LS, et al. Dose-adjusted EPOCH-rituximab therapy in primary mediastinal B-cell lymphoma. N Engl J Med 2013;368:1408–16.
74. Aoki T, Shimada K, Suzuki R, et al. High-dose chemotherapy followed by autologous stem cell transplantation for relapsed/refractory primary mediastinal large B-cell lymphoma. Blood Cancer J 2015;5:e372–e372.
75. Sehn LH, Antin JH, Shulman LN, et al. Primary diffuse large B-cell lymphoma of the mediastinum: outcome following high-dose chemotherapy and autologous hematopoietic cell transplantation. Blood 1998;91:717–23.
76. Kuruvilla J, Pintilie M, Tsang R, et al. Salvage chemotherapy and autologous stem cell transplantation are inferior for relapsed or refractory primary mediastinal large B-cell lymphoma compared with diffuse large B-cell lymphoma. Leuk Lymphoma 2008;49:1329–36.
77. Zinzani PL, Ribrag V, Moskowitz CH, et al. Safety and tolerability of pembrolizumab in patients with relapsed/refractory primary mediastinal large B-cell lymphoma. Blood 2017;130:267–70.
78. Smith A, Howell D, Patmore R, et al. Incidence of haematological malignancy by sub-type: a report from the Haematological Malignancy Research Network. Br J Cancer 2011;105:1684–92.
79. Argatoff LH, Connors JM, Klasa RJ, et al. Mantle cell lymphoma: a clinicopathologic study of 80 cases. Blood 1997;89:2067–78.
80. Zukerberg LR, Yang WI, Arnold A, Harris NL. Cyclin D1 expression in non-Hodgkin’s lymphomas. Detection by immunohistochemistry. Am J Clin Pathol 1995;103:756–60.
81. Wiestner A, Tehrani M, Chiorazzi M, et al. Point mutations and genomic deletions in CCND1 create stable truncated cyclin D1 mRNAs that are associated with increased proliferation rate and shorter survival. Blood 2007;109:4599–606.
82. Fu K, Weisenburger DD, Greiner TC, et al. Cyclin D1-negative mantle cell lymphoma: a clinicopathologic study based on gene expression profiling. Blood 2005;106:4315–21.
83. Mozos A, Royo C, Hartmann E, et al. SOX11 expression is highly specific for mantle cell lymphoma and identifies the cyclin D1-negative subtype. Haematologica 2009;94:1555–62.
84. Norton AJ, Matthews J, Pappa V, et al. Mantle cell lymphoma: Natural history defined in a serially biopsied population over a 20-year period. Ann Oncol 1995;6:249–56.
85. Chihara D, Cheah CY, Westin JR, et al. Rituximab plus hyper-CVAD alternating with MTX/Ara-C in patients with newly diagnosed mantle cell lymphoma: 15-year follow-up of a phase II study from the MD Anderson Cancer Center. Br J Haematol 2016;172:80–8.
86. Delarue R, Haioun C, Ribrag V, et al. CHOP and DHAP plus rituximab followed by autologous stem cell transplantation in mantle cell lymphoma: a phase 2 study from the Groupe d’Etude des Lymphomes de l’Adulte. Blood 2013;121:48–53.
87. Eskelund CW, Kolstad A, Jerkeman M, et al. 15-year follow-up of the Second Nordic Mantle Cell Lymphoma trial (MCL2): prolonged remissions without survival plateau. Br J Haematol 2016;175:410–8.
88. Hoster E, Dreyling M, Klapper W, et al. A new prognostic index (MIPI) for patients with advanced-stage mantle cell lymphoma. Blood 2008;111:558–65.
89. Hoster E, Klapper W, Hermine O, et al. Confirmation of the mantle-cell lymphoma International Prognostic Index in randomized trials of the European Mantle-Cell Lymphoma Network. J Clin Oncol 2014;32:1338–46.
90. Hoster E, Rosenwald A, Berger F, et al. Prognostic value of Ki-67 index, cytology, and growth pattern in mantle-cell lymphoma: Results from randomized trials of the European Mantle Cell Lymphoma Network. J Clin Oncol 2016;34:1386–94.
91. Bernard M, Gressin R, Lefrère F, et al. Blastic variant of mantle cell lymphoma: a rare but highly aggressive subtype. Leukemia 2001;15:1785–91.
92. Martin P, Chadburn A, Christos P, et al. Outcome of deferred initial therapy in mantle-cell lymphoma. J Clin Oncol 2009;27:1209–13.
93. Le Gouill S, Thieblemont C, Oberic L, et al. Rituximab after autologous stem-cell transplantation in mantle-cell lymphoma. N Engl J Med. 2017 Sep 28;377(13):1250–60.
94. Hermine O, Hoster E, Walewski J, et al. Addition of high-dose cytarabine to immunochemotherapy before autologous stem-cell transplantation in patients aged 65 years or younger with mantle cell lymphoma (MCL Younger): a randomised, open-label, phase 3 trial of the European Mantle Cell Lymphoma Network. Lancet 2016;388:565–75.
95. Fenske TS, Zhang M-J, Carreras J, et al. Autologous or reduced-intensity conditioning allogeneic hematopoietic cell transplantation for chemotherapy-sensitive mantle-cell lymphoma: analysis of transplantation timing and modality. J Clin Oncol 2014;32:273–81.
96. Kluin-Nelemans HC, Hoster E, Hermine O, et al. Treatment of older patients with mantle-cell lymphoma. N Engl J Med 2012;367:520–31.
97. Flinn IW, van der Jagt R, Kahl BS, et al. Randomized trial of bendamustine-rituximab or R-CHOP/R-CVP in first-line treatment of indolent NHL or MCL: the BRIGHT study. Blood 2014;123:2944–52.
98. Rummel MJ, Niederle N, Maschmeyer G, et al. Bendamustine plus rituximab versus CHOP plus rituximab as first-line treatment for patients with indolent and mantle-cell lymphomas: an open-label, multicentre, randomised, phase 3 non-inferiority trial. Lancet 2013;381:1203–10.
99. Lenz G, Dreyling M, Hoster E, et al. Immunochemotherapy with rituximab and cyclophosphamide, doxorubicin, vincristine, and prednisone significantly improves response and time to treatment failure, but not long-term outcome in patients with previously untreated mantle cell lymphoma: results of a prospective randomized trial of the German Low Grade Lymphoma Study Group (GLSG). J Clin Oncol 2005;23:1984–92.
100. Belch A, Kouroukis CT, Crump M, et al. A phase II study of bortezomib in mantle cell lymphoma: the National Cancer Institute of Canada Clinical Trials Group trial IND.150. Ann Oncol Off J Eur Soc Med Oncol 2007;18:116–21.
101. Fisher RI, Bernstein SH, Kahl BS, et al. Multicenter phase II study of bortezomib in patients with relapsed or refractory mantle cell lymphoma. J Clin Oncol 2006;24:4867–74.
102. Dreyling M, Jurczak W, Jerkeman M, et al. Ibrutinib versus temsirolimus in patients with relapsed or refractory mantle-cell lymphoma: an international, randomised, open-label, phase 3 study. Lancet 2016;387:770–8.
103. Wang ML, Rule S, Martin P, Goy A, et al. Targeting BTK with ibrutinib in relapsed or refractory mantle-cell lymphoma. N Engl J Med 2013;369:507–16.
104. Wang M, Rule S, Zinzani PL, et al. Acalabrutinib in relapsed or refractory mantle cell lymphoma (ACE-LY-004): a single-arm, multicentre, phase 2 trial. Lancet 2018;391:659–67.
105. Goy A, Sinha R, Williams ME, et al. Single-agent lenalidomide in patients with mantle-cell lymphoma who relapsed or progressed after or were refractory to bortezomib: phase II MCL-001 (EMERGE) study. J Clin Oncol 2013;31:3688–95.
106. Khouri IF, Lee M-S, Saliba RM, et al. Nonablative allogeneic stem-cell transplantation for advanced/recurrent mantle-cell lymphoma. J Clin Oncol 2003;21:4407–12.
107. Blum KA, Lozanski G, Byrd JC. Adult Burkitt leukemia and lymphoma. Blood 2004;104:3009–20.
108. Roschewski M, Dunleavy K, Abramson JS, et al. Risk-adapted therapy in adults with Burkitt lymphoma: results of NCI 9177, a multicenter prospective phase II study of DA-EPOCH-R. Blood American Society of Hematology;2017;130(Suppl 1):188.
109. Maramattom L V, Hari PN, Burns LJ, et al. Autologous and allogeneic transplantation for burkitt lymphoma outcomes and changes in utilization: a report from the center for international blood and marrow transplant research. Biol Blood Marrow Transplant 2013;19:173–9.
110. Zinzani PL, Bendandi M, Visani G, et al. Adult lymphoblastic lymphoma: clinical features and prognostic factors in 53 patients. Leuk Lymphoma 1996;23:577–82.
111. Thomas DA, O’Brien S, Cortes J, et al. Outcome with the hyper-CVAD regimens in lymphoblastic lymphoma. Blood 2004;104:1624–30.
112. Aljurf M, Zaidi SZA. Chemotherapy and hematopoietic stem cell transplantation for adult T-cell lymphoblastic lymphoma: current status and controversies. Biol Blood Marrow Transplant 2005;11:739–54.
113. Sweetenham JW, Santini G, Qian W, et al. High-dose therapy and autologous stem-cell transplantation versus conventional-dose consolidation/maintenance therapy as postremission therapy for adult patients with lymphoblastic lymphoma: results of a randomized trial of the European Group for Blood and Marrow Transplantation and the United Kingdom Lymphoma Group. J Clin Oncol 2001;19:2927–36.
114. Zwaan CM, Kowalczyk J, Schmitt C, et al. Safety and efficacy of nelarabine in children and young adults with relapsed or refractory T-lineage acute lymphoblastic leukaemia or T-lineage lymphoblastic lymphoma: results of a phase 4 study. Br J Haematol 2017;179:284–93.
Umbralisib has ‘distinct’ safety profile
Phase 1 trial results suggest umbralisib, a PI3Kδ/CK1ε inhibitor, can be safe and active in patients with relapsed or refractory B-cell malignancies.
Researchers said the safety profile of umbralisib “was distinct from that of other PI3Kδ inhibitors,” as it produced few immune-mediated adverse events (AEs).
Umbralisib also produced an objective response rate of 37% in the entire study cohort, 80% in patients with chronic lymphocytic leukemia (CLL), 53% in patients with follicular lymphoma (FL), and 31% in patients with diffuse large B-cell lymphoma (DLBCL).
These results were published in The Lancet Oncology. The study was sponsored by TG Therapeutics, Inc.
The trial enrolled 90 patients between January 17, 2013, and January 14, 2016.
There were 24 patients with CLL, 22 with FL, 16 with DLBCL, 11 with Hodgkin lymphoma, 6 with mantle cell lymphoma, 5 with marginal zone lymphoma, 3 with Waldenstrom’s macroglobulinemia, 2 with T-cell lymphoma, and 1 with hairy cell leukemia.
The median number of prior therapies was 3 (range, 2-5), and 49% of patients were refractory to previous therapy.
Treatment
Patients took umbralisib once daily in 28-day cycles until disease progression, unacceptable toxicity, or withdrawal of consent.
Initially, patients took the drug in a fasting state at doses of 50 mg, 100 mg, 200 mg, 400 mg, 800 mg, 1200 mg, or 1800 mg.
In April 2014, the researchers did a second dose-escalation with a micronized formulation of umbralisib, taken with food, at doses of 200 mg, 400 mg, 800 mg, 1200 mg, or 1800 mg.
In August, 2014, all patients who were still on the study transitioned to the 800 mg dose of the micronized formulation. This was the recommended phase 2 dose.
At the data cutoff in November 2016, 44 patients (49%) had received umbralisib for more than 6 cycles, and 23 (26%) had received the drug for more than 12 cycles. Thirteen patients (14%) were still taking umbralisib at the end of the study.
Most patients who stopped treatment did so because of disease progression (n=50, 56%) or AEs (n=9, 10%).
“We are pleased to have treated the first patient ever with umbralisib over 5 years ago and believe it has an important place in the treatment landscape for patients with hematologic malignancies,” said study author Howard A. Burris, MD, of the Sarah Cannon Research Institute in Nashville, Tennessee.
“Several patients from this phase 1 study are still on study today, approaching 5 years of continuous daily therapy, speaking to both the safety and efficacy profile of this unique agent.”
Safety
Dose-limiting toxicities (DLTs) occurred in 4 patients. One DLT was grade 3 maculopapular rash in a patient receiving the 800 mg dose of the initial formulation.
Another DLT was grade 3 hypokalemia in a patient receiving 1800 mg of the initial formulation. A third DLT was grade 3 fatigue, which occurred in 2 patients receiving 1800 mg of the micronized formulation.
Because of these toxicities, the maximum tolerated dose was 1200 mg of the micronized formulation.
The most common treatment-emergent AEs were diarrhea (43%), nausea (42%), and fatigue (31%). The most common grade 3/4 AEs were neutropenia (13%), anemia (9%), and thrombocytopenia (7%).
Serious AEs considered at least possibly related to umbralisib were pneumonia (3%), lung infection (1%), febrile neutropenia (1%), and colitis (2%).
Treatment discontinuation due to AEs considered at least possibly related to umbralisib occurred in 6 patients (7%). Two patients had grade 3 colitis, 2 had increased ALT/AST (grade 1 and grade 4), 1 had grade 2 diarrhea, and 1 had grade 3 fatigue.
There were no treatment-related deaths.
The researchers said the safety profile of umbralisib was distinct from that of other PI3Kδ inhibitors, as patients in this trial had fewer occurrences of autoimmune-like toxicities, such as colitis.
“Preclinically, umbralisib has a very unique profile, selectively inhibiting both PI3Kδ and CK1ε,” said study author Owen O’Connor, MD, PhD, of Columbia Presbyterian Medical Center in New York, New York.
“The clinical results in this paper support our thesis that the differentiated preclinical profile explains the differences seen in the clinic between umbralisib and the other PI3Kδ inhibitors.”
Response
The objective response rate was 37%, with 33 patients achieving a response and 3 patients having a complete response (CR).
Sixteen CLL patients responded (80%), all with partial responses (PRs). Four DLBCL patients responded (31%), all with PRs. And 9 FL patients responded (53%), 2 with CRs.
The remaining CR occurred in a Hodgkin lymphoma patient, and this was the only response in this patient group.
One patient with marginal zone lymphoma had a PR, as did 1 patient with mantle cell lymphoma. All other patients had stable disease or progressed.
The mean duration of response was 13.4 months in the CLL patients, 6.4 months in the DLBCL patients, and 9.3 months in the FL patients.
Phase 1 trial results suggest umbralisib, a PI3Kδ/CK1ε inhibitor, can be safe and active in patients with relapsed or refractory B-cell malignancies.
Researchers said the safety profile of umbralisib “was distinct from that of other PI3Kδ inhibitors,” as it produced few immune-mediated adverse events (AEs).
Umbralisib also produced an objective response rate of 37% in the entire study cohort, 80% in patients with chronic lymphocytic leukemia (CLL), 53% in patients with follicular lymphoma (FL), and 31% in patients with diffuse large B-cell lymphoma (DLBCL).
These results were published in The Lancet Oncology. The study was sponsored by TG Therapeutics, Inc.
The trial enrolled 90 patients between January 17, 2013, and January 14, 2016.
There were 24 patients with CLL, 22 with FL, 16 with DLBCL, 11 with Hodgkin lymphoma, 6 with mantle cell lymphoma, 5 with marginal zone lymphoma, 3 with Waldenstrom’s macroglobulinemia, 2 with T-cell lymphoma, and 1 with hairy cell leukemia.
The median number of prior therapies was 3 (range, 2-5), and 49% of patients were refractory to previous therapy.
Treatment
Patients took umbralisib once daily in 28-day cycles until disease progression, unacceptable toxicity, or withdrawal of consent.
Initially, patients took the drug in a fasting state at doses of 50 mg, 100 mg, 200 mg, 400 mg, 800 mg, 1200 mg, or 1800 mg.
In April 2014, the researchers did a second dose-escalation with a micronized formulation of umbralisib, taken with food, at doses of 200 mg, 400 mg, 800 mg, 1200 mg, or 1800 mg.
In August, 2014, all patients who were still on the study transitioned to the 800 mg dose of the micronized formulation. This was the recommended phase 2 dose.
At the data cutoff in November 2016, 44 patients (49%) had received umbralisib for more than 6 cycles, and 23 (26%) had received the drug for more than 12 cycles. Thirteen patients (14%) were still taking umbralisib at the end of the study.
Most patients who stopped treatment did so because of disease progression (n=50, 56%) or AEs (n=9, 10%).
“We are pleased to have treated the first patient ever with umbralisib over 5 years ago and believe it has an important place in the treatment landscape for patients with hematologic malignancies,” said study author Howard A. Burris, MD, of the Sarah Cannon Research Institute in Nashville, Tennessee.
“Several patients from this phase 1 study are still on study today, approaching 5 years of continuous daily therapy, speaking to both the safety and efficacy profile of this unique agent.”
Safety
Dose-limiting toxicities (DLTs) occurred in 4 patients. One DLT was grade 3 maculopapular rash in a patient receiving the 800 mg dose of the initial formulation.
Another DLT was grade 3 hypokalemia in a patient receiving 1800 mg of the initial formulation. A third DLT was grade 3 fatigue, which occurred in 2 patients receiving 1800 mg of the micronized formulation.
Because of these toxicities, the maximum tolerated dose was 1200 mg of the micronized formulation.
The most common treatment-emergent AEs were diarrhea (43%), nausea (42%), and fatigue (31%). The most common grade 3/4 AEs were neutropenia (13%), anemia (9%), and thrombocytopenia (7%).
Serious AEs considered at least possibly related to umbralisib were pneumonia (3%), lung infection (1%), febrile neutropenia (1%), and colitis (2%).
Treatment discontinuation due to AEs considered at least possibly related to umbralisib occurred in 6 patients (7%). Two patients had grade 3 colitis, 2 had increased ALT/AST (grade 1 and grade 4), 1 had grade 2 diarrhea, and 1 had grade 3 fatigue.
There were no treatment-related deaths.
The researchers said the safety profile of umbralisib was distinct from that of other PI3Kδ inhibitors, as patients in this trial had fewer occurrences of autoimmune-like toxicities, such as colitis.
“Preclinically, umbralisib has a very unique profile, selectively inhibiting both PI3Kδ and CK1ε,” said study author Owen O’Connor, MD, PhD, of Columbia Presbyterian Medical Center in New York, New York.
“The clinical results in this paper support our thesis that the differentiated preclinical profile explains the differences seen in the clinic between umbralisib and the other PI3Kδ inhibitors.”
Response
The objective response rate was 37%, with 33 patients achieving a response and 3 patients having a complete response (CR).
Sixteen CLL patients responded (80%), all with partial responses (PRs). Four DLBCL patients responded (31%), all with PRs. And 9 FL patients responded (53%), 2 with CRs.
The remaining CR occurred in a Hodgkin lymphoma patient, and this was the only response in this patient group.
One patient with marginal zone lymphoma had a PR, as did 1 patient with mantle cell lymphoma. All other patients had stable disease or progressed.
The mean duration of response was 13.4 months in the CLL patients, 6.4 months in the DLBCL patients, and 9.3 months in the FL patients.
Phase 1 trial results suggest umbralisib, a PI3Kδ/CK1ε inhibitor, can be safe and active in patients with relapsed or refractory B-cell malignancies.
Researchers said the safety profile of umbralisib “was distinct from that of other PI3Kδ inhibitors,” as it produced few immune-mediated adverse events (AEs).
Umbralisib also produced an objective response rate of 37% in the entire study cohort, 80% in patients with chronic lymphocytic leukemia (CLL), 53% in patients with follicular lymphoma (FL), and 31% in patients with diffuse large B-cell lymphoma (DLBCL).
These results were published in The Lancet Oncology. The study was sponsored by TG Therapeutics, Inc.
The trial enrolled 90 patients between January 17, 2013, and January 14, 2016.
There were 24 patients with CLL, 22 with FL, 16 with DLBCL, 11 with Hodgkin lymphoma, 6 with mantle cell lymphoma, 5 with marginal zone lymphoma, 3 with Waldenstrom’s macroglobulinemia, 2 with T-cell lymphoma, and 1 with hairy cell leukemia.
The median number of prior therapies was 3 (range, 2-5), and 49% of patients were refractory to previous therapy.
Treatment
Patients took umbralisib once daily in 28-day cycles until disease progression, unacceptable toxicity, or withdrawal of consent.
Initially, patients took the drug in a fasting state at doses of 50 mg, 100 mg, 200 mg, 400 mg, 800 mg, 1200 mg, or 1800 mg.
In April 2014, the researchers did a second dose-escalation with a micronized formulation of umbralisib, taken with food, at doses of 200 mg, 400 mg, 800 mg, 1200 mg, or 1800 mg.
In August, 2014, all patients who were still on the study transitioned to the 800 mg dose of the micronized formulation. This was the recommended phase 2 dose.
At the data cutoff in November 2016, 44 patients (49%) had received umbralisib for more than 6 cycles, and 23 (26%) had received the drug for more than 12 cycles. Thirteen patients (14%) were still taking umbralisib at the end of the study.
Most patients who stopped treatment did so because of disease progression (n=50, 56%) or AEs (n=9, 10%).
“We are pleased to have treated the first patient ever with umbralisib over 5 years ago and believe it has an important place in the treatment landscape for patients with hematologic malignancies,” said study author Howard A. Burris, MD, of the Sarah Cannon Research Institute in Nashville, Tennessee.
“Several patients from this phase 1 study are still on study today, approaching 5 years of continuous daily therapy, speaking to both the safety and efficacy profile of this unique agent.”
Safety
Dose-limiting toxicities (DLTs) occurred in 4 patients. One DLT was grade 3 maculopapular rash in a patient receiving the 800 mg dose of the initial formulation.
Another DLT was grade 3 hypokalemia in a patient receiving 1800 mg of the initial formulation. A third DLT was grade 3 fatigue, which occurred in 2 patients receiving 1800 mg of the micronized formulation.
Because of these toxicities, the maximum tolerated dose was 1200 mg of the micronized formulation.
The most common treatment-emergent AEs were diarrhea (43%), nausea (42%), and fatigue (31%). The most common grade 3/4 AEs were neutropenia (13%), anemia (9%), and thrombocytopenia (7%).
Serious AEs considered at least possibly related to umbralisib were pneumonia (3%), lung infection (1%), febrile neutropenia (1%), and colitis (2%).
Treatment discontinuation due to AEs considered at least possibly related to umbralisib occurred in 6 patients (7%). Two patients had grade 3 colitis, 2 had increased ALT/AST (grade 1 and grade 4), 1 had grade 2 diarrhea, and 1 had grade 3 fatigue.
There were no treatment-related deaths.
The researchers said the safety profile of umbralisib was distinct from that of other PI3Kδ inhibitors, as patients in this trial had fewer occurrences of autoimmune-like toxicities, such as colitis.
“Preclinically, umbralisib has a very unique profile, selectively inhibiting both PI3Kδ and CK1ε,” said study author Owen O’Connor, MD, PhD, of Columbia Presbyterian Medical Center in New York, New York.
“The clinical results in this paper support our thesis that the differentiated preclinical profile explains the differences seen in the clinic between umbralisib and the other PI3Kδ inhibitors.”
Response
The objective response rate was 37%, with 33 patients achieving a response and 3 patients having a complete response (CR).
Sixteen CLL patients responded (80%), all with partial responses (PRs). Four DLBCL patients responded (31%), all with PRs. And 9 FL patients responded (53%), 2 with CRs.
The remaining CR occurred in a Hodgkin lymphoma patient, and this was the only response in this patient group.
One patient with marginal zone lymphoma had a PR, as did 1 patient with mantle cell lymphoma. All other patients had stable disease or progressed.
The mean duration of response was 13.4 months in the CLL patients, 6.4 months in the DLBCL patients, and 9.3 months in the FL patients.
2017 notches up some landmark approvals
With advances in the understanding of cellular pathways, molecular genetics, and immunology, new drugs for cancer are being released at an increasing rate. A variety of novel agents have recently become available for use, generating excitement for patients and oncologists. Keeping track of all of these new agents is increasingly challenging. This brief review will summarize some of the newest drugs, their indications, and benefits (see related article).
Therapies by tumor
Breast cancer
CDK4/6 inhibitors. The CDK4/6 inhibitor palbociclib was approved in 2015 for the treatment of estrogen-positive, HER2-negative advanced breast cancer, and this year, two more drugs in this class – ribociclib and abemaciclib – were approved for the treatment of hormone receptor–positive breast cancer.
Ribociclib (Kisqali) 600 mg daily (3 weeks on, 1 week off) is approved for use in combination with an aromatase inhibitor. In the study on which the approval was based, there was a response rate of 53% for patients in the study group, compared with 37% for those who received aromatase inhibitor alone (progression-free survival (PFS), not reached vs 14.7 months for single-agent aromatase inhibitor).1 The occurrence of neutropenia seemed to be similar to that in patients receiving palbociclib. However, unlike with palbociclib, ribociclib requires ECG monitoring for QTc prolongation as well as monitoring of liver function tests.
Abemaciclib (Verzenio) has been approved in combination with fulvestrant as well as a monotherapy.2 PFS was 16.4 months for abemaciclib (150 mg bid in combination with fulvestrant), compared with 9.3 months for fulvestrant alone, with corresponding response rates of 48% and 21%. As monotherapy, abemaciclib 200 mg bid had a response rate of 20% with a duration of response of 8.6 months.
Tyrosine kinase inhibitors. The tyrosine kinase inhibitor neratinib (Nerlynx) was approved for extended adjuvant treatment of HER2-positive breast cancer after 1 year of adjuvant trastuzumab.3 Given at 240 mg (6 tablets) daily for a year, compared with a no-treatment control arm, it demonstrated an improvement in invasive disease-free survival (DFS) at 2 years from 91.9% to 94.2%, with no difference in overall survival yet noted. It is associated with diarrhea and also requires hepatic function monitoring.
Acute myelogenous leukemia
Multiple new agents were recently approved for use in acute myelogenous leukemia (AML), after decades of slow advance in new drug development.
Midostaurin (Rydapt) is an FLT3 inhibitor approved for use in combination with daunorubicin and Ara-C (cytosine arabinoside) for newly diagnosed AML with FLT3 mutations, which occur in about 30% of AML patients.4 It is given orally on days 8-21 at 50 mg bid with induction and consolidation.
In the study on which the approval was based, there was a 10% improvement in overall survival for this subset of AML patients who have a typically a worse prognosis. Event-free survival in patients in the study group was 8.2 months, compared with 3 months in the control arm patients, who did not receive the agent. The drug was also approved for aggressive systemic mastocytosis.
Enasidenib (Idhifa) has been approved for AML with an IDH2 mutation in the refractory/relapsed settings.5IDH2 mutations are present in about 20% of patients with AML. Given orally at 100 mg daily as a single agent, enasidenib was associated with a 19% complete remission rate. Patients need to be monitored for differentiation syndrome, somewhat similar to what is seen with ATRA with acute promyelocytic leukemia.
Liposomal daunorubicin and cytarabine (Vyxeos) was approved for newly diagnosed therapy- or myelodysplasia-related AML.6 This novel liposomal formulation combines two standard agents and is given intravenously on days 1, 3 and 5 over 90 minutes as daunorubicin 44 mg/m2 and cytarabine 100 mg/ m2. (For a second induction and in lower dose on consolidation cycles, it is given only on days 1 and 3). The liposomal formulation achieved a superior complete response rate compared with the standard 7+3 daunorubicin plus cytarabine regimen (38% vs 26%, respectively) and longer overall survival (9.6 versus 5.9 months) in these generally poor prognosis subsets.
Gemtuzumab ozogamicin (Mylotarg) was initially approved in 2000 but withdrawn from use in 2010 after trials failed to confirm benefit and demonstrated safety concerns. It has now been re-released in a lower dose and schedule from its original label.7 This immunoconjugate of an anti-CD33 bound to calicheamicin is approved for CD33-positive AML. Given at 3 mg/m2 on days 1, 4, and 7 in combination with standard daunorubicin–cytarabine induction chemotherapy, it improved event-free survival from 9.5 to 17.3 months. When administered as a single agent (6 mg/m2 on day 1 and 3 mg/m2 on day 8) in patients who were unable or unwilling to tolerate standard chemotherapy, it improved overall survival (4.9 months versus 3.6 months for best supportive care). As a single agent in relapsed AML, given at 3 mg/m2 days 1, 4, and 7 and followed by cytarabine consolidation, it was associated with a 26% complete response rate, with a median relapse-free survival of 11.6 months.
Ovarian/fallopian tube cancers
PARP inhibitors. For patients with ovarian/fallopian tube cancer, there are new indications and agents for PARP inhibition, including for patients with BRCA mutations (both somatic and germline) and those without BRCA mutations.
Olaparib (Lynparza) was previously approved only in a fourth-line setting for germline BRCA-mutated patients with advanced ovarian cancer, with a response rate of 34% with a median duration of 7.9 months. Given at 300 mg orally bid, it is now approved for use in maintenance in recurrence after response to platinum-based chemotherapy after 2 or more lines of therapy regardless of BRCA status. In this setting, progression-free survival increased to 8.4 months, compared with 4.8 months for placebo.8
Rubicarib (Rubraca) is approved for BRCA-mutated patients (either germline or somatic) with advanced ovarian cancer after two or more lines of chemotherapy.9 At 600 mg orally bid, results from phase 2 trials noted a 54% response rate, with a median duration of 9.2 months.
Niraparib (Zejula) is approved for use in maintenance in recurrent epithelial ovarian, fallopian tube, and primary peritoneal cancers after platinum-based chemotherapy.10 In patients with germline BRCA mutations, niraparib at 300 mg orally daily resulted in a PFS of 21 months, compared with 5.5 months with placebo; PFS in patients with nongermline BRCA mutations was 9.3 versus 3.9 months, respectively.
Non-small cell lung cancer with EML-4 alk translocation
Crizotinib (Xalkori) has been the mainstay for treatment of for EML4-alk translocated non-small cell lung cancer. However, alectinib (Alcensa), previously for predominantly second-line use, seems more active than crizotinib in the first-line setting, particularly in the treatment and prevention of CNS metastases.
In addition, brigantinib (Alunbrig) has been approved for patients who are intolerant/refractory to crizotinib.11 At 90 mg once daily for 7 days, then escalating to 180 mg daily, it was noted to have a 50% response rate in crizotinib failures, including in the CNS.
Ceritinib (Zykadia) was approved at 750 mg once daily for EML4 alk positive NSCLC.12 In first line it had a response rate of 73% (versus 27% for chemotherapy) with a remission duration of 23.9 months (versus 11.1 months for chemotherapy).
Therapies by drug class
PD-1/PD-L1 antibodies
Anti-PD-1 antibodies nivolumab (Opdivo) and pembrolizumab (Keytruda) are widely used for a range of tumor types. Newer approvals for pembrolizumab are for adenocarcinoma of the stomach/gastro-esophageal junction with at least 1% PD-L1 expression, and in any tumor demonstrated to be MSI-high. Newer indications for nivolumab are for bladder cancer, MSI-high colon cancer, and for hepatoma previously treated with sorafenib. The anti-PD-L1 antibody atezolizumab (Tencentriq) is now approved for platinum-resistant metastatic lung cancer, in addition to platinum-ineligible and platinum-resistant urothelial cancer.
Avelumab (Bavencio) is an anti-PD-L1 approved for both Merkel cell and previously treated urothelial cancers at a dose of 10 mg/kg every 2 weeks.13 It demonstrated a 33% response rate for Merkel cell and a 16% response rate for urothelial cancer.
Durvalumab (Imfinzi) is another anti PD-L1 antibody approved at 10 mg/kg every 2 weeks for previously treated urothelial cancer with a 17% response rate (RR: PD-L1 high, 26%; low, 4%).14
PI3K kinase inhibitors
Copanlisib (Aliqopa) is a PI3K inhibitor approved for relapsed follicular lymphoma in patients who have progressed after two previous lines of therapy.15 It is a 60-mg, 1-hour infusion given on days 1, 8, and 15 every 28 days. In a phase 2 tria
BTK inhibitors
Acalabruitnib (Calquence) is approved for adults with previously treated mantle cell lymphoma. In a phase 2 trial at 100 mg orally bid, it achieved an 80% overall and 40% complete response rate.16 These response rates are higher than were seen for ibrutinib in its original phase 2 trial. The spectrum of toxicities seems similar to ibruitinib and includes bleeding, cytopenias, infection, and atrial fibrillation.
CD19 CAR-T cells
Perhaps the most exciting and novel new agents are genetically engineered autologous T cells. Tisagenlecleucel (Kymriah), a chimeric antigen receptor T cell (CART) that targets CD19 is approved for refractory B cell precursor acute lymphoblastic leukemia (in patients under 25 years) where the complete response rate was 83% (including patients with incomplete blood count recovery).17
Axicabtagene ciloleucel (aci-cel; Yescarta), also CD19-directed CART, is approved for adults with relapsed or refractory non-Hodgkin lymphoma after two lines of previous therapy (specifically large-cell lymphoma, primary mediastinal large B-cell lymphoma, and transformed follicular lymphoma). Response rate was 72% (complete, 51%; partial, 21%), with a median duration of response of 9.2 months.18
1. Hortobagyi GN, Stemmer SM, Burris HA, et al. Ribociclib as first-line therapy for HR-positive, advanced breast cancer. N Engl J Med. 2016;375(18):1738-1748.
2. Goetz MP, Toi M, Campone M, et al. MONARCH 3: Abemaciclib as initial therapy for advanced breast cancer. J Clin Oncol. 2017;35(32):3638-3646.
3. Chan A, Delaloge S, Holmes FA, et al. Neratinib after trastuzumab-based adjuvant therapy in patients with HER2-positive breast cancer (ExteNET): a multicentre, randomised, double-blind, placebo-controlled, phase 3 trial. Lancet Oncol. 2016;17(3):367-377.
4. Stone RM, Mandrekar SJ, Sanford BL, et al. Midostaurin plus chemotherapy for acute myeloid leukemia with a FLT3 mutation. N Engl J Med. 2017;377(5):454-464.
5. Stein EM, DiNardo CD, Pollyea DA, et al. Enasidenib in mutant IDH2 relapsed or refractory acute myeloid leukemia. Blood. 2017 Aug 10;130(6):722-731.
6. Lancet JE, Rizzieri D, Schiller GJ, et al. Overall survival (OS) with CPX-351 versus 7+3 in older adults with newly diagnosed, therapy-related acute myeloid leukemia (tAML): subgroup analysis of a phase III study. http://ascopubs.org/doi/abs/10.1200/JCO.2017.35.15_suppl.7035. Published May 2017. Accessed November 20, 2017.
7. Appelbaum FR, Bernstein ID. Gemtuzumab ozogamicin for acute myeloid leukemia. http://www.bloodjournal.org/content/early/2017/10/11/blood-2017-09-797712?sso-checked=true. September 2017. Accessed November 20, 2017.
8. Kim G, Ison G, McKee AE, et al. FDA approval summary: olaparib monotherapy in patients with deleterious germline BRCA-mutated advanced ovarian cancer treated with three or more lines of chemotherapy. Clin Cancer Res. 2015;21:4257-4261.
9. Swisher EM, Lin KK, Oza AM, et al. Rucaparib in relapsed, platinum-sensitive high-grade ovarian carcinoma (ARIEL2 part 1): an international, multicentre, open-label, phase 2 trial. Lancet Oncol. 2017;18:75-87.
10. Mirza MR, Monk BJ, Herrstedt J, et al. Niraparib maintenance therapy in platinum-sensitive, recurrent ovarian cancer. N Engl J Med. 2016;375:2154-2164.
11. Kim DW, Tiseo M, Ahn MJ, Reckamp KL, et al. Brigatinib in patients with crizotinib-refractory anaplastic lymphoma kinase-positive non-small-cell lung cancer: a randomized, multicenter phase II trial. J Clin Oncol. 2017;35(22):2490-2498.
12. Soria J-C, Tan DSW, MD, Chiari R, et al. First-line ceritinib versus platinum-based chemotherapy in advanced ALK-rearranged non-small-cell lung cancer (ASCEND-4): a randomised, open-label, phase 3 study. Lancet. 2017;389(10072):917-929.
13. Apolo AB, Infante JR, Balmanoukian A et al. Avelumab, an anti–programmed death-ligand 1 antibody, in patients with refractory metastatic urothelial carcinoma: results from a multicenter, phase Ib study. J Clin Oncol. 2017;35(19):2117-2124.
14. Massard C, Gordon MS, Sharma S, et al. Safety and efficacy of durvalumab (MEDI4736), an anti–programmed cell death ligand-1 immune checkpoint inhibitor, in patients with advanced urothelial bladder cancer. J Clin Oncol. 2016;34(26):3119-3125.
15. Dreyling M, Morschhauser F, Bouabdallah K, et al. Phase II study of copanlisib, a PI3K inhibitor, in relapsed or refractory, indolent or aggressive lymphoma. Ann Oncol. 2017;28(9):2169-2178.
16. Wu J, Zhang M, Liu D. Acalabrutinib (ACP-196): a selective second-generation BTK inhibitor. https://jhoonline.biomedcentral.com/articles/10.1186/s13045-016-0250-9. Published March 9, 2016. Accessed November 20, 2017.
17. Maude SL, Frey N, Shaw PA, et al. Chimeric antigen receptor T cells for sustained remissions in leukemia. N Engl J Med. 2014;371(16):1507-1517.
18. Locke FL, Neelapu SS, Bartlett NL, et al. Phase 1 results of ZUMA-1: a multicenter study of KTE-C19 anti-CD19 CAR T cell therapy in refractory aggressive lymphoma. Mol Ther. 2017;25(1):285-295.
With advances in the understanding of cellular pathways, molecular genetics, and immunology, new drugs for cancer are being released at an increasing rate. A variety of novel agents have recently become available for use, generating excitement for patients and oncologists. Keeping track of all of these new agents is increasingly challenging. This brief review will summarize some of the newest drugs, their indications, and benefits (see related article).
Therapies by tumor
Breast cancer
CDK4/6 inhibitors. The CDK4/6 inhibitor palbociclib was approved in 2015 for the treatment of estrogen-positive, HER2-negative advanced breast cancer, and this year, two more drugs in this class – ribociclib and abemaciclib – were approved for the treatment of hormone receptor–positive breast cancer.
Ribociclib (Kisqali) 600 mg daily (3 weeks on, 1 week off) is approved for use in combination with an aromatase inhibitor. In the study on which the approval was based, there was a response rate of 53% for patients in the study group, compared with 37% for those who received aromatase inhibitor alone (progression-free survival (PFS), not reached vs 14.7 months for single-agent aromatase inhibitor).1 The occurrence of neutropenia seemed to be similar to that in patients receiving palbociclib. However, unlike with palbociclib, ribociclib requires ECG monitoring for QTc prolongation as well as monitoring of liver function tests.
Abemaciclib (Verzenio) has been approved in combination with fulvestrant as well as a monotherapy.2 PFS was 16.4 months for abemaciclib (150 mg bid in combination with fulvestrant), compared with 9.3 months for fulvestrant alone, with corresponding response rates of 48% and 21%. As monotherapy, abemaciclib 200 mg bid had a response rate of 20% with a duration of response of 8.6 months.
Tyrosine kinase inhibitors. The tyrosine kinase inhibitor neratinib (Nerlynx) was approved for extended adjuvant treatment of HER2-positive breast cancer after 1 year of adjuvant trastuzumab.3 Given at 240 mg (6 tablets) daily for a year, compared with a no-treatment control arm, it demonstrated an improvement in invasive disease-free survival (DFS) at 2 years from 91.9% to 94.2%, with no difference in overall survival yet noted. It is associated with diarrhea and also requires hepatic function monitoring.
Acute myelogenous leukemia
Multiple new agents were recently approved for use in acute myelogenous leukemia (AML), after decades of slow advance in new drug development.
Midostaurin (Rydapt) is an FLT3 inhibitor approved for use in combination with daunorubicin and Ara-C (cytosine arabinoside) for newly diagnosed AML with FLT3 mutations, which occur in about 30% of AML patients.4 It is given orally on days 8-21 at 50 mg bid with induction and consolidation.
In the study on which the approval was based, there was a 10% improvement in overall survival for this subset of AML patients who have a typically a worse prognosis. Event-free survival in patients in the study group was 8.2 months, compared with 3 months in the control arm patients, who did not receive the agent. The drug was also approved for aggressive systemic mastocytosis.
Enasidenib (Idhifa) has been approved for AML with an IDH2 mutation in the refractory/relapsed settings.5IDH2 mutations are present in about 20% of patients with AML. Given orally at 100 mg daily as a single agent, enasidenib was associated with a 19% complete remission rate. Patients need to be monitored for differentiation syndrome, somewhat similar to what is seen with ATRA with acute promyelocytic leukemia.
Liposomal daunorubicin and cytarabine (Vyxeos) was approved for newly diagnosed therapy- or myelodysplasia-related AML.6 This novel liposomal formulation combines two standard agents and is given intravenously on days 1, 3 and 5 over 90 minutes as daunorubicin 44 mg/m2 and cytarabine 100 mg/ m2. (For a second induction and in lower dose on consolidation cycles, it is given only on days 1 and 3). The liposomal formulation achieved a superior complete response rate compared with the standard 7+3 daunorubicin plus cytarabine regimen (38% vs 26%, respectively) and longer overall survival (9.6 versus 5.9 months) in these generally poor prognosis subsets.
Gemtuzumab ozogamicin (Mylotarg) was initially approved in 2000 but withdrawn from use in 2010 after trials failed to confirm benefit and demonstrated safety concerns. It has now been re-released in a lower dose and schedule from its original label.7 This immunoconjugate of an anti-CD33 bound to calicheamicin is approved for CD33-positive AML. Given at 3 mg/m2 on days 1, 4, and 7 in combination with standard daunorubicin–cytarabine induction chemotherapy, it improved event-free survival from 9.5 to 17.3 months. When administered as a single agent (6 mg/m2 on day 1 and 3 mg/m2 on day 8) in patients who were unable or unwilling to tolerate standard chemotherapy, it improved overall survival (4.9 months versus 3.6 months for best supportive care). As a single agent in relapsed AML, given at 3 mg/m2 days 1, 4, and 7 and followed by cytarabine consolidation, it was associated with a 26% complete response rate, with a median relapse-free survival of 11.6 months.
Ovarian/fallopian tube cancers
PARP inhibitors. For patients with ovarian/fallopian tube cancer, there are new indications and agents for PARP inhibition, including for patients with BRCA mutations (both somatic and germline) and those without BRCA mutations.
Olaparib (Lynparza) was previously approved only in a fourth-line setting for germline BRCA-mutated patients with advanced ovarian cancer, with a response rate of 34% with a median duration of 7.9 months. Given at 300 mg orally bid, it is now approved for use in maintenance in recurrence after response to platinum-based chemotherapy after 2 or more lines of therapy regardless of BRCA status. In this setting, progression-free survival increased to 8.4 months, compared with 4.8 months for placebo.8
Rubicarib (Rubraca) is approved for BRCA-mutated patients (either germline or somatic) with advanced ovarian cancer after two or more lines of chemotherapy.9 At 600 mg orally bid, results from phase 2 trials noted a 54% response rate, with a median duration of 9.2 months.
Niraparib (Zejula) is approved for use in maintenance in recurrent epithelial ovarian, fallopian tube, and primary peritoneal cancers after platinum-based chemotherapy.10 In patients with germline BRCA mutations, niraparib at 300 mg orally daily resulted in a PFS of 21 months, compared with 5.5 months with placebo; PFS in patients with nongermline BRCA mutations was 9.3 versus 3.9 months, respectively.
Non-small cell lung cancer with EML-4 alk translocation
Crizotinib (Xalkori) has been the mainstay for treatment of for EML4-alk translocated non-small cell lung cancer. However, alectinib (Alcensa), previously for predominantly second-line use, seems more active than crizotinib in the first-line setting, particularly in the treatment and prevention of CNS metastases.
In addition, brigantinib (Alunbrig) has been approved for patients who are intolerant/refractory to crizotinib.11 At 90 mg once daily for 7 days, then escalating to 180 mg daily, it was noted to have a 50% response rate in crizotinib failures, including in the CNS.
Ceritinib (Zykadia) was approved at 750 mg once daily for EML4 alk positive NSCLC.12 In first line it had a response rate of 73% (versus 27% for chemotherapy) with a remission duration of 23.9 months (versus 11.1 months for chemotherapy).
Therapies by drug class
PD-1/PD-L1 antibodies
Anti-PD-1 antibodies nivolumab (Opdivo) and pembrolizumab (Keytruda) are widely used for a range of tumor types. Newer approvals for pembrolizumab are for adenocarcinoma of the stomach/gastro-esophageal junction with at least 1% PD-L1 expression, and in any tumor demonstrated to be MSI-high. Newer indications for nivolumab are for bladder cancer, MSI-high colon cancer, and for hepatoma previously treated with sorafenib. The anti-PD-L1 antibody atezolizumab (Tencentriq) is now approved for platinum-resistant metastatic lung cancer, in addition to platinum-ineligible and platinum-resistant urothelial cancer.
Avelumab (Bavencio) is an anti-PD-L1 approved for both Merkel cell and previously treated urothelial cancers at a dose of 10 mg/kg every 2 weeks.13 It demonstrated a 33% response rate for Merkel cell and a 16% response rate for urothelial cancer.
Durvalumab (Imfinzi) is another anti PD-L1 antibody approved at 10 mg/kg every 2 weeks for previously treated urothelial cancer with a 17% response rate (RR: PD-L1 high, 26%; low, 4%).14
PI3K kinase inhibitors
Copanlisib (Aliqopa) is a PI3K inhibitor approved for relapsed follicular lymphoma in patients who have progressed after two previous lines of therapy.15 It is a 60-mg, 1-hour infusion given on days 1, 8, and 15 every 28 days. In a phase 2 tria
BTK inhibitors
Acalabruitnib (Calquence) is approved for adults with previously treated mantle cell lymphoma. In a phase 2 trial at 100 mg orally bid, it achieved an 80% overall and 40% complete response rate.16 These response rates are higher than were seen for ibrutinib in its original phase 2 trial. The spectrum of toxicities seems similar to ibruitinib and includes bleeding, cytopenias, infection, and atrial fibrillation.
CD19 CAR-T cells
Perhaps the most exciting and novel new agents are genetically engineered autologous T cells. Tisagenlecleucel (Kymriah), a chimeric antigen receptor T cell (CART) that targets CD19 is approved for refractory B cell precursor acute lymphoblastic leukemia (in patients under 25 years) where the complete response rate was 83% (including patients with incomplete blood count recovery).17
Axicabtagene ciloleucel (aci-cel; Yescarta), also CD19-directed CART, is approved for adults with relapsed or refractory non-Hodgkin lymphoma after two lines of previous therapy (specifically large-cell lymphoma, primary mediastinal large B-cell lymphoma, and transformed follicular lymphoma). Response rate was 72% (complete, 51%; partial, 21%), with a median duration of response of 9.2 months.18
With advances in the understanding of cellular pathways, molecular genetics, and immunology, new drugs for cancer are being released at an increasing rate. A variety of novel agents have recently become available for use, generating excitement for patients and oncologists. Keeping track of all of these new agents is increasingly challenging. This brief review will summarize some of the newest drugs, their indications, and benefits (see related article).
Therapies by tumor
Breast cancer
CDK4/6 inhibitors. The CDK4/6 inhibitor palbociclib was approved in 2015 for the treatment of estrogen-positive, HER2-negative advanced breast cancer, and this year, two more drugs in this class – ribociclib and abemaciclib – were approved for the treatment of hormone receptor–positive breast cancer.
Ribociclib (Kisqali) 600 mg daily (3 weeks on, 1 week off) is approved for use in combination with an aromatase inhibitor. In the study on which the approval was based, there was a response rate of 53% for patients in the study group, compared with 37% for those who received aromatase inhibitor alone (progression-free survival (PFS), not reached vs 14.7 months for single-agent aromatase inhibitor).1 The occurrence of neutropenia seemed to be similar to that in patients receiving palbociclib. However, unlike with palbociclib, ribociclib requires ECG monitoring for QTc prolongation as well as monitoring of liver function tests.
Abemaciclib (Verzenio) has been approved in combination with fulvestrant as well as a monotherapy.2 PFS was 16.4 months for abemaciclib (150 mg bid in combination with fulvestrant), compared with 9.3 months for fulvestrant alone, with corresponding response rates of 48% and 21%. As monotherapy, abemaciclib 200 mg bid had a response rate of 20% with a duration of response of 8.6 months.
Tyrosine kinase inhibitors. The tyrosine kinase inhibitor neratinib (Nerlynx) was approved for extended adjuvant treatment of HER2-positive breast cancer after 1 year of adjuvant trastuzumab.3 Given at 240 mg (6 tablets) daily for a year, compared with a no-treatment control arm, it demonstrated an improvement in invasive disease-free survival (DFS) at 2 years from 91.9% to 94.2%, with no difference in overall survival yet noted. It is associated with diarrhea and also requires hepatic function monitoring.
Acute myelogenous leukemia
Multiple new agents were recently approved for use in acute myelogenous leukemia (AML), after decades of slow advance in new drug development.
Midostaurin (Rydapt) is an FLT3 inhibitor approved for use in combination with daunorubicin and Ara-C (cytosine arabinoside) for newly diagnosed AML with FLT3 mutations, which occur in about 30% of AML patients.4 It is given orally on days 8-21 at 50 mg bid with induction and consolidation.
In the study on which the approval was based, there was a 10% improvement in overall survival for this subset of AML patients who have a typically a worse prognosis. Event-free survival in patients in the study group was 8.2 months, compared with 3 months in the control arm patients, who did not receive the agent. The drug was also approved for aggressive systemic mastocytosis.
Enasidenib (Idhifa) has been approved for AML with an IDH2 mutation in the refractory/relapsed settings.5IDH2 mutations are present in about 20% of patients with AML. Given orally at 100 mg daily as a single agent, enasidenib was associated with a 19% complete remission rate. Patients need to be monitored for differentiation syndrome, somewhat similar to what is seen with ATRA with acute promyelocytic leukemia.
Liposomal daunorubicin and cytarabine (Vyxeos) was approved for newly diagnosed therapy- or myelodysplasia-related AML.6 This novel liposomal formulation combines two standard agents and is given intravenously on days 1, 3 and 5 over 90 minutes as daunorubicin 44 mg/m2 and cytarabine 100 mg/ m2. (For a second induction and in lower dose on consolidation cycles, it is given only on days 1 and 3). The liposomal formulation achieved a superior complete response rate compared with the standard 7+3 daunorubicin plus cytarabine regimen (38% vs 26%, respectively) and longer overall survival (9.6 versus 5.9 months) in these generally poor prognosis subsets.
Gemtuzumab ozogamicin (Mylotarg) was initially approved in 2000 but withdrawn from use in 2010 after trials failed to confirm benefit and demonstrated safety concerns. It has now been re-released in a lower dose and schedule from its original label.7 This immunoconjugate of an anti-CD33 bound to calicheamicin is approved for CD33-positive AML. Given at 3 mg/m2 on days 1, 4, and 7 in combination with standard daunorubicin–cytarabine induction chemotherapy, it improved event-free survival from 9.5 to 17.3 months. When administered as a single agent (6 mg/m2 on day 1 and 3 mg/m2 on day 8) in patients who were unable or unwilling to tolerate standard chemotherapy, it improved overall survival (4.9 months versus 3.6 months for best supportive care). As a single agent in relapsed AML, given at 3 mg/m2 days 1, 4, and 7 and followed by cytarabine consolidation, it was associated with a 26% complete response rate, with a median relapse-free survival of 11.6 months.
Ovarian/fallopian tube cancers
PARP inhibitors. For patients with ovarian/fallopian tube cancer, there are new indications and agents for PARP inhibition, including for patients with BRCA mutations (both somatic and germline) and those without BRCA mutations.
Olaparib (Lynparza) was previously approved only in a fourth-line setting for germline BRCA-mutated patients with advanced ovarian cancer, with a response rate of 34% with a median duration of 7.9 months. Given at 300 mg orally bid, it is now approved for use in maintenance in recurrence after response to platinum-based chemotherapy after 2 or more lines of therapy regardless of BRCA status. In this setting, progression-free survival increased to 8.4 months, compared with 4.8 months for placebo.8
Rubicarib (Rubraca) is approved for BRCA-mutated patients (either germline or somatic) with advanced ovarian cancer after two or more lines of chemotherapy.9 At 600 mg orally bid, results from phase 2 trials noted a 54% response rate, with a median duration of 9.2 months.
Niraparib (Zejula) is approved for use in maintenance in recurrent epithelial ovarian, fallopian tube, and primary peritoneal cancers after platinum-based chemotherapy.10 In patients with germline BRCA mutations, niraparib at 300 mg orally daily resulted in a PFS of 21 months, compared with 5.5 months with placebo; PFS in patients with nongermline BRCA mutations was 9.3 versus 3.9 months, respectively.
Non-small cell lung cancer with EML-4 alk translocation
Crizotinib (Xalkori) has been the mainstay for treatment of for EML4-alk translocated non-small cell lung cancer. However, alectinib (Alcensa), previously for predominantly second-line use, seems more active than crizotinib in the first-line setting, particularly in the treatment and prevention of CNS metastases.
In addition, brigantinib (Alunbrig) has been approved for patients who are intolerant/refractory to crizotinib.11 At 90 mg once daily for 7 days, then escalating to 180 mg daily, it was noted to have a 50% response rate in crizotinib failures, including in the CNS.
Ceritinib (Zykadia) was approved at 750 mg once daily for EML4 alk positive NSCLC.12 In first line it had a response rate of 73% (versus 27% for chemotherapy) with a remission duration of 23.9 months (versus 11.1 months for chemotherapy).
Therapies by drug class
PD-1/PD-L1 antibodies
Anti-PD-1 antibodies nivolumab (Opdivo) and pembrolizumab (Keytruda) are widely used for a range of tumor types. Newer approvals for pembrolizumab are for adenocarcinoma of the stomach/gastro-esophageal junction with at least 1% PD-L1 expression, and in any tumor demonstrated to be MSI-high. Newer indications for nivolumab are for bladder cancer, MSI-high colon cancer, and for hepatoma previously treated with sorafenib. The anti-PD-L1 antibody atezolizumab (Tencentriq) is now approved for platinum-resistant metastatic lung cancer, in addition to platinum-ineligible and platinum-resistant urothelial cancer.
Avelumab (Bavencio) is an anti-PD-L1 approved for both Merkel cell and previously treated urothelial cancers at a dose of 10 mg/kg every 2 weeks.13 It demonstrated a 33% response rate for Merkel cell and a 16% response rate for urothelial cancer.
Durvalumab (Imfinzi) is another anti PD-L1 antibody approved at 10 mg/kg every 2 weeks for previously treated urothelial cancer with a 17% response rate (RR: PD-L1 high, 26%; low, 4%).14
PI3K kinase inhibitors
Copanlisib (Aliqopa) is a PI3K inhibitor approved for relapsed follicular lymphoma in patients who have progressed after two previous lines of therapy.15 It is a 60-mg, 1-hour infusion given on days 1, 8, and 15 every 28 days. In a phase 2 tria
BTK inhibitors
Acalabruitnib (Calquence) is approved for adults with previously treated mantle cell lymphoma. In a phase 2 trial at 100 mg orally bid, it achieved an 80% overall and 40% complete response rate.16 These response rates are higher than were seen for ibrutinib in its original phase 2 trial. The spectrum of toxicities seems similar to ibruitinib and includes bleeding, cytopenias, infection, and atrial fibrillation.
CD19 CAR-T cells
Perhaps the most exciting and novel new agents are genetically engineered autologous T cells. Tisagenlecleucel (Kymriah), a chimeric antigen receptor T cell (CART) that targets CD19 is approved for refractory B cell precursor acute lymphoblastic leukemia (in patients under 25 years) where the complete response rate was 83% (including patients with incomplete blood count recovery).17
Axicabtagene ciloleucel (aci-cel; Yescarta), also CD19-directed CART, is approved for adults with relapsed or refractory non-Hodgkin lymphoma after two lines of previous therapy (specifically large-cell lymphoma, primary mediastinal large B-cell lymphoma, and transformed follicular lymphoma). Response rate was 72% (complete, 51%; partial, 21%), with a median duration of response of 9.2 months.18
1. Hortobagyi GN, Stemmer SM, Burris HA, et al. Ribociclib as first-line therapy for HR-positive, advanced breast cancer. N Engl J Med. 2016;375(18):1738-1748.
2. Goetz MP, Toi M, Campone M, et al. MONARCH 3: Abemaciclib as initial therapy for advanced breast cancer. J Clin Oncol. 2017;35(32):3638-3646.
3. Chan A, Delaloge S, Holmes FA, et al. Neratinib after trastuzumab-based adjuvant therapy in patients with HER2-positive breast cancer (ExteNET): a multicentre, randomised, double-blind, placebo-controlled, phase 3 trial. Lancet Oncol. 2016;17(3):367-377.
4. Stone RM, Mandrekar SJ, Sanford BL, et al. Midostaurin plus chemotherapy for acute myeloid leukemia with a FLT3 mutation. N Engl J Med. 2017;377(5):454-464.
5. Stein EM, DiNardo CD, Pollyea DA, et al. Enasidenib in mutant IDH2 relapsed or refractory acute myeloid leukemia. Blood. 2017 Aug 10;130(6):722-731.
6. Lancet JE, Rizzieri D, Schiller GJ, et al. Overall survival (OS) with CPX-351 versus 7+3 in older adults with newly diagnosed, therapy-related acute myeloid leukemia (tAML): subgroup analysis of a phase III study. http://ascopubs.org/doi/abs/10.1200/JCO.2017.35.15_suppl.7035. Published May 2017. Accessed November 20, 2017.
7. Appelbaum FR, Bernstein ID. Gemtuzumab ozogamicin for acute myeloid leukemia. http://www.bloodjournal.org/content/early/2017/10/11/blood-2017-09-797712?sso-checked=true. September 2017. Accessed November 20, 2017.
8. Kim G, Ison G, McKee AE, et al. FDA approval summary: olaparib monotherapy in patients with deleterious germline BRCA-mutated advanced ovarian cancer treated with three or more lines of chemotherapy. Clin Cancer Res. 2015;21:4257-4261.
9. Swisher EM, Lin KK, Oza AM, et al. Rucaparib in relapsed, platinum-sensitive high-grade ovarian carcinoma (ARIEL2 part 1): an international, multicentre, open-label, phase 2 trial. Lancet Oncol. 2017;18:75-87.
10. Mirza MR, Monk BJ, Herrstedt J, et al. Niraparib maintenance therapy in platinum-sensitive, recurrent ovarian cancer. N Engl J Med. 2016;375:2154-2164.
11. Kim DW, Tiseo M, Ahn MJ, Reckamp KL, et al. Brigatinib in patients with crizotinib-refractory anaplastic lymphoma kinase-positive non-small-cell lung cancer: a randomized, multicenter phase II trial. J Clin Oncol. 2017;35(22):2490-2498.
12. Soria J-C, Tan DSW, MD, Chiari R, et al. First-line ceritinib versus platinum-based chemotherapy in advanced ALK-rearranged non-small-cell lung cancer (ASCEND-4): a randomised, open-label, phase 3 study. Lancet. 2017;389(10072):917-929.
13. Apolo AB, Infante JR, Balmanoukian A et al. Avelumab, an anti–programmed death-ligand 1 antibody, in patients with refractory metastatic urothelial carcinoma: results from a multicenter, phase Ib study. J Clin Oncol. 2017;35(19):2117-2124.
14. Massard C, Gordon MS, Sharma S, et al. Safety and efficacy of durvalumab (MEDI4736), an anti–programmed cell death ligand-1 immune checkpoint inhibitor, in patients with advanced urothelial bladder cancer. J Clin Oncol. 2016;34(26):3119-3125.
15. Dreyling M, Morschhauser F, Bouabdallah K, et al. Phase II study of copanlisib, a PI3K inhibitor, in relapsed or refractory, indolent or aggressive lymphoma. Ann Oncol. 2017;28(9):2169-2178.
16. Wu J, Zhang M, Liu D. Acalabrutinib (ACP-196): a selective second-generation BTK inhibitor. https://jhoonline.biomedcentral.com/articles/10.1186/s13045-016-0250-9. Published March 9, 2016. Accessed November 20, 2017.
17. Maude SL, Frey N, Shaw PA, et al. Chimeric antigen receptor T cells for sustained remissions in leukemia. N Engl J Med. 2014;371(16):1507-1517.
18. Locke FL, Neelapu SS, Bartlett NL, et al. Phase 1 results of ZUMA-1: a multicenter study of KTE-C19 anti-CD19 CAR T cell therapy in refractory aggressive lymphoma. Mol Ther. 2017;25(1):285-295.
1. Hortobagyi GN, Stemmer SM, Burris HA, et al. Ribociclib as first-line therapy for HR-positive, advanced breast cancer. N Engl J Med. 2016;375(18):1738-1748.
2. Goetz MP, Toi M, Campone M, et al. MONARCH 3: Abemaciclib as initial therapy for advanced breast cancer. J Clin Oncol. 2017;35(32):3638-3646.
3. Chan A, Delaloge S, Holmes FA, et al. Neratinib after trastuzumab-based adjuvant therapy in patients with HER2-positive breast cancer (ExteNET): a multicentre, randomised, double-blind, placebo-controlled, phase 3 trial. Lancet Oncol. 2016;17(3):367-377.
4. Stone RM, Mandrekar SJ, Sanford BL, et al. Midostaurin plus chemotherapy for acute myeloid leukemia with a FLT3 mutation. N Engl J Med. 2017;377(5):454-464.
5. Stein EM, DiNardo CD, Pollyea DA, et al. Enasidenib in mutant IDH2 relapsed or refractory acute myeloid leukemia. Blood. 2017 Aug 10;130(6):722-731.
6. Lancet JE, Rizzieri D, Schiller GJ, et al. Overall survival (OS) with CPX-351 versus 7+3 in older adults with newly diagnosed, therapy-related acute myeloid leukemia (tAML): subgroup analysis of a phase III study. http://ascopubs.org/doi/abs/10.1200/JCO.2017.35.15_suppl.7035. Published May 2017. Accessed November 20, 2017.
7. Appelbaum FR, Bernstein ID. Gemtuzumab ozogamicin for acute myeloid leukemia. http://www.bloodjournal.org/content/early/2017/10/11/blood-2017-09-797712?sso-checked=true. September 2017. Accessed November 20, 2017.
8. Kim G, Ison G, McKee AE, et al. FDA approval summary: olaparib monotherapy in patients with deleterious germline BRCA-mutated advanced ovarian cancer treated with three or more lines of chemotherapy. Clin Cancer Res. 2015;21:4257-4261.
9. Swisher EM, Lin KK, Oza AM, et al. Rucaparib in relapsed, platinum-sensitive high-grade ovarian carcinoma (ARIEL2 part 1): an international, multicentre, open-label, phase 2 trial. Lancet Oncol. 2017;18:75-87.
10. Mirza MR, Monk BJ, Herrstedt J, et al. Niraparib maintenance therapy in platinum-sensitive, recurrent ovarian cancer. N Engl J Med. 2016;375:2154-2164.
11. Kim DW, Tiseo M, Ahn MJ, Reckamp KL, et al. Brigatinib in patients with crizotinib-refractory anaplastic lymphoma kinase-positive non-small-cell lung cancer: a randomized, multicenter phase II trial. J Clin Oncol. 2017;35(22):2490-2498.
12. Soria J-C, Tan DSW, MD, Chiari R, et al. First-line ceritinib versus platinum-based chemotherapy in advanced ALK-rearranged non-small-cell lung cancer (ASCEND-4): a randomised, open-label, phase 3 study. Lancet. 2017;389(10072):917-929.
13. Apolo AB, Infante JR, Balmanoukian A et al. Avelumab, an anti–programmed death-ligand 1 antibody, in patients with refractory metastatic urothelial carcinoma: results from a multicenter, phase Ib study. J Clin Oncol. 2017;35(19):2117-2124.
14. Massard C, Gordon MS, Sharma S, et al. Safety and efficacy of durvalumab (MEDI4736), an anti–programmed cell death ligand-1 immune checkpoint inhibitor, in patients with advanced urothelial bladder cancer. J Clin Oncol. 2016;34(26):3119-3125.
15. Dreyling M, Morschhauser F, Bouabdallah K, et al. Phase II study of copanlisib, a PI3K inhibitor, in relapsed or refractory, indolent or aggressive lymphoma. Ann Oncol. 2017;28(9):2169-2178.
16. Wu J, Zhang M, Liu D. Acalabrutinib (ACP-196): a selective second-generation BTK inhibitor. https://jhoonline.biomedcentral.com/articles/10.1186/s13045-016-0250-9. Published March 9, 2016. Accessed November 20, 2017.
17. Maude SL, Frey N, Shaw PA, et al. Chimeric antigen receptor T cells for sustained remissions in leukemia. N Engl J Med. 2014;371(16):1507-1517.
18. Locke FL, Neelapu SS, Bartlett NL, et al. Phase 1 results of ZUMA-1: a multicenter study of KTE-C19 anti-CD19 CAR T cell therapy in refractory aggressive lymphoma. Mol Ther. 2017;25(1):285-295.
Drug granted fast track designations for FL, DLBCL
The US Food and Drug Administration (FDA) has granted 2 fast track designations to 5F9, an anti-CD47 antibody.
The designations are for 5F9 as a treatment for relapsed or refractory diffuse large B-cell lymphoma (DLBCL) and follicular lymphoma (FL).
Data supporting the fast track designations were derived from a phase 1b/2 trial of 5F9 in combination with rituximab in patients with relapsed or refractory B-cell non-Hodgkin lymphoma, including DLBCL and FL.
Forty Seven, Inc., the company developing 5F9, expects to announce initial safety and efficacy data from the phase 1b portion of the trial in the second quarter of 2018.
About fast track designation
The FDA’s fast track drug development program is designed to expedite clinical development and submission of applications for drugs with the potential to treat serious or life-threatening conditions and address unmet medical needs.
Fast track designation facilitates frequent interactions with the FDA review team, including meetings to discuss the drug’s development plan and written communications about issues such as trial design and use of biomarkers.
Drugs that receive fast track designation may be eligible for accelerated approval and priority review if relevant criteria are met.
Fast track drugs may also be eligible for rolling review, which allows a developer to submit individual sections of a drug’s application for review as they are ready, rather than waiting until all sections are complete.
The US Food and Drug Administration (FDA) has granted 2 fast track designations to 5F9, an anti-CD47 antibody.
The designations are for 5F9 as a treatment for relapsed or refractory diffuse large B-cell lymphoma (DLBCL) and follicular lymphoma (FL).
Data supporting the fast track designations were derived from a phase 1b/2 trial of 5F9 in combination with rituximab in patients with relapsed or refractory B-cell non-Hodgkin lymphoma, including DLBCL and FL.
Forty Seven, Inc., the company developing 5F9, expects to announce initial safety and efficacy data from the phase 1b portion of the trial in the second quarter of 2018.
About fast track designation
The FDA’s fast track drug development program is designed to expedite clinical development and submission of applications for drugs with the potential to treat serious or life-threatening conditions and address unmet medical needs.
Fast track designation facilitates frequent interactions with the FDA review team, including meetings to discuss the drug’s development plan and written communications about issues such as trial design and use of biomarkers.
Drugs that receive fast track designation may be eligible for accelerated approval and priority review if relevant criteria are met.
Fast track drugs may also be eligible for rolling review, which allows a developer to submit individual sections of a drug’s application for review as they are ready, rather than waiting until all sections are complete.
The US Food and Drug Administration (FDA) has granted 2 fast track designations to 5F9, an anti-CD47 antibody.
The designations are for 5F9 as a treatment for relapsed or refractory diffuse large B-cell lymphoma (DLBCL) and follicular lymphoma (FL).
Data supporting the fast track designations were derived from a phase 1b/2 trial of 5F9 in combination with rituximab in patients with relapsed or refractory B-cell non-Hodgkin lymphoma, including DLBCL and FL.
Forty Seven, Inc., the company developing 5F9, expects to announce initial safety and efficacy data from the phase 1b portion of the trial in the second quarter of 2018.
About fast track designation
The FDA’s fast track drug development program is designed to expedite clinical development and submission of applications for drugs with the potential to treat serious or life-threatening conditions and address unmet medical needs.
Fast track designation facilitates frequent interactions with the FDA review team, including meetings to discuss the drug’s development plan and written communications about issues such as trial design and use of biomarkers.
Drugs that receive fast track designation may be eligible for accelerated approval and priority review if relevant criteria are met.
Fast track drugs may also be eligible for rolling review, which allows a developer to submit individual sections of a drug’s application for review as they are ready, rather than waiting until all sections are complete.
Early results favor combo IL-15/anti-CD20 in indolent NHL
CHICAGO – A combination of an immunostimulatory IL-15-based agent, ALT-803, with a therapeutic monoclonal antibody (mAb) against CD20, was well tolerated and had clinical activity in patients with indolent non-Hodgkin lymphoma (iNHL), according to preliminary findings from a phase 1 study.
“The cancer immunotherapy breakthrough that happened several years ago continues year after year, with a plethora of different modalities of immunotherapy at our disposal,” Todd A. Fehniger, MD, PhD, said at the annual meeting of the American Association for Cancer Research.
Immunotherapy with anti-CD20 mAbs, alone or in combination with chemotherapy, is a standard therapy for iNHL patients. Since iNHL cells express CD20, targeting it with mAbs triggers antitumor responses via cell surface receptors resulting in a potent antibody-dependent cellular toxicity. However, response in patients is highly heterogeneous, with relapse within a few months in a subset of patients. In addition, chemotherapeutic combinations can be toxic and result in serious and long-term complications.
“Relapsed or refractory iNHL is not curable and treatment strategies without long-term complications are needed,” said Dr. Fehniger, associate professor of medicine at Washington University, St. Louis.
In an attempt to address this, Dr. Fehniger and his colleagues combined rituximab, an anti-CD20 antibody, with a relatively new IL-15 agonist immunostimulatory agent called ALT-803.
In the phase 1 trial, the researchers enrolled patients with indolent non-Hodgkin lymphoma who had relapsed after at least 1 prior to CD20 antibody containing therapy. The study was a standard 3+3 dose escalation design with rituximab administered by intravenous infusion, 375 mg/m2 in four weekly doses, followed by a rest and four consolidation doses every 8 weeks for four cycles.
ALT-803 was administered concurrently at dose levels of 1 mcg/kg, 3 mcg/kg, and 6 mcg/kg IV followed by 6 mcg/kg, 10 mcg/kg, 15 mcg/kg, and 20 mcg/kg subcutaneously.
In total, 21 patients were treated: 16 patients had follicular lymphoma, four patients had marginal zone lymphoma, and one patient had small lymphocytic lymphoma. The median prior therapies received was two (range: 1-18) and five patients were treated who were refractory to prior anti-CD20 MAb therapy.
ALT-803 was well tolerated with no dose limiting toxicities or grade 4 or 5 adverse events. No patients discontinued ALT-803 and the recommended phase 2 dose was 20 mcg/kg subcutaneously. Grade 3 adverse events, regardless of attribution to ALT-803, included transient hypertension (14%), anemia (5%), nausea (5%), chills (5%), fever (5%), neutropenia (5%), and hyperglycemia (5%).
“Patients who received [subcutaneous] ALT-803 developed a unique injection site rash reaction that peaked 7-10 days later but resolved typically within 14 days. It was self-limited and resolved on its own,” Dr. Fehniger said.
At the time of the presentation, the best overall response rate was achieved in 11 of 21 patients (52%), with 9 complete responders (43%), and 2 partial responders (10%).
Of the 12 patients treated with ALT-803 subcutaneously, 11 patients had either stable disease, or partial or complete responses. All 11 patients remained on study and were in consolidation or follow-up and have not relapsed, Dr. Fehniger reported.
Among the five rituximab-refractory patients, the researchers observed one complete response, two patients with stable disease (45% and 36% tumor volume decrease), and two patients with partial disease. The durability of the responses can only be understood with longer follow-up, Dr. Fehniger said.
The peripheral blood of the patients was analyzed via flow cytometry and mass cytometry. Over the duration of four weekly doses, there was an increase in percentage (sixfold, P less than .001) and absolute number (10-fold, P less than .001) of natural killer cells at the 15-mcg/kg and 20-mcg/kg subcutaneous dose levels of ALT-803.
These results suggest that further studies of ALT-803 with other therapeutic targeting mAbs, or other immunotherapy modalities, are warranted, the researchers concluded.
Dr. Fehniger reported research funding from Altor BioScience.
SOURCE: Fehniger TA et al. AACR Annual Meeting, Abstract CT146.
CHICAGO – A combination of an immunostimulatory IL-15-based agent, ALT-803, with a therapeutic monoclonal antibody (mAb) against CD20, was well tolerated and had clinical activity in patients with indolent non-Hodgkin lymphoma (iNHL), according to preliminary findings from a phase 1 study.
“The cancer immunotherapy breakthrough that happened several years ago continues year after year, with a plethora of different modalities of immunotherapy at our disposal,” Todd A. Fehniger, MD, PhD, said at the annual meeting of the American Association for Cancer Research.
Immunotherapy with anti-CD20 mAbs, alone or in combination with chemotherapy, is a standard therapy for iNHL patients. Since iNHL cells express CD20, targeting it with mAbs triggers antitumor responses via cell surface receptors resulting in a potent antibody-dependent cellular toxicity. However, response in patients is highly heterogeneous, with relapse within a few months in a subset of patients. In addition, chemotherapeutic combinations can be toxic and result in serious and long-term complications.
“Relapsed or refractory iNHL is not curable and treatment strategies without long-term complications are needed,” said Dr. Fehniger, associate professor of medicine at Washington University, St. Louis.
In an attempt to address this, Dr. Fehniger and his colleagues combined rituximab, an anti-CD20 antibody, with a relatively new IL-15 agonist immunostimulatory agent called ALT-803.
In the phase 1 trial, the researchers enrolled patients with indolent non-Hodgkin lymphoma who had relapsed after at least 1 prior to CD20 antibody containing therapy. The study was a standard 3+3 dose escalation design with rituximab administered by intravenous infusion, 375 mg/m2 in four weekly doses, followed by a rest and four consolidation doses every 8 weeks for four cycles.
ALT-803 was administered concurrently at dose levels of 1 mcg/kg, 3 mcg/kg, and 6 mcg/kg IV followed by 6 mcg/kg, 10 mcg/kg, 15 mcg/kg, and 20 mcg/kg subcutaneously.
In total, 21 patients were treated: 16 patients had follicular lymphoma, four patients had marginal zone lymphoma, and one patient had small lymphocytic lymphoma. The median prior therapies received was two (range: 1-18) and five patients were treated who were refractory to prior anti-CD20 MAb therapy.
ALT-803 was well tolerated with no dose limiting toxicities or grade 4 or 5 adverse events. No patients discontinued ALT-803 and the recommended phase 2 dose was 20 mcg/kg subcutaneously. Grade 3 adverse events, regardless of attribution to ALT-803, included transient hypertension (14%), anemia (5%), nausea (5%), chills (5%), fever (5%), neutropenia (5%), and hyperglycemia (5%).
“Patients who received [subcutaneous] ALT-803 developed a unique injection site rash reaction that peaked 7-10 days later but resolved typically within 14 days. It was self-limited and resolved on its own,” Dr. Fehniger said.
At the time of the presentation, the best overall response rate was achieved in 11 of 21 patients (52%), with 9 complete responders (43%), and 2 partial responders (10%).
Of the 12 patients treated with ALT-803 subcutaneously, 11 patients had either stable disease, or partial or complete responses. All 11 patients remained on study and were in consolidation or follow-up and have not relapsed, Dr. Fehniger reported.
Among the five rituximab-refractory patients, the researchers observed one complete response, two patients with stable disease (45% and 36% tumor volume decrease), and two patients with partial disease. The durability of the responses can only be understood with longer follow-up, Dr. Fehniger said.
The peripheral blood of the patients was analyzed via flow cytometry and mass cytometry. Over the duration of four weekly doses, there was an increase in percentage (sixfold, P less than .001) and absolute number (10-fold, P less than .001) of natural killer cells at the 15-mcg/kg and 20-mcg/kg subcutaneous dose levels of ALT-803.
These results suggest that further studies of ALT-803 with other therapeutic targeting mAbs, or other immunotherapy modalities, are warranted, the researchers concluded.
Dr. Fehniger reported research funding from Altor BioScience.
SOURCE: Fehniger TA et al. AACR Annual Meeting, Abstract CT146.
CHICAGO – A combination of an immunostimulatory IL-15-based agent, ALT-803, with a therapeutic monoclonal antibody (mAb) against CD20, was well tolerated and had clinical activity in patients with indolent non-Hodgkin lymphoma (iNHL), according to preliminary findings from a phase 1 study.
“The cancer immunotherapy breakthrough that happened several years ago continues year after year, with a plethora of different modalities of immunotherapy at our disposal,” Todd A. Fehniger, MD, PhD, said at the annual meeting of the American Association for Cancer Research.
Immunotherapy with anti-CD20 mAbs, alone or in combination with chemotherapy, is a standard therapy for iNHL patients. Since iNHL cells express CD20, targeting it with mAbs triggers antitumor responses via cell surface receptors resulting in a potent antibody-dependent cellular toxicity. However, response in patients is highly heterogeneous, with relapse within a few months in a subset of patients. In addition, chemotherapeutic combinations can be toxic and result in serious and long-term complications.
“Relapsed or refractory iNHL is not curable and treatment strategies without long-term complications are needed,” said Dr. Fehniger, associate professor of medicine at Washington University, St. Louis.
In an attempt to address this, Dr. Fehniger and his colleagues combined rituximab, an anti-CD20 antibody, with a relatively new IL-15 agonist immunostimulatory agent called ALT-803.
In the phase 1 trial, the researchers enrolled patients with indolent non-Hodgkin lymphoma who had relapsed after at least 1 prior to CD20 antibody containing therapy. The study was a standard 3+3 dose escalation design with rituximab administered by intravenous infusion, 375 mg/m2 in four weekly doses, followed by a rest and four consolidation doses every 8 weeks for four cycles.
ALT-803 was administered concurrently at dose levels of 1 mcg/kg, 3 mcg/kg, and 6 mcg/kg IV followed by 6 mcg/kg, 10 mcg/kg, 15 mcg/kg, and 20 mcg/kg subcutaneously.
In total, 21 patients were treated: 16 patients had follicular lymphoma, four patients had marginal zone lymphoma, and one patient had small lymphocytic lymphoma. The median prior therapies received was two (range: 1-18) and five patients were treated who were refractory to prior anti-CD20 MAb therapy.
ALT-803 was well tolerated with no dose limiting toxicities or grade 4 or 5 adverse events. No patients discontinued ALT-803 and the recommended phase 2 dose was 20 mcg/kg subcutaneously. Grade 3 adverse events, regardless of attribution to ALT-803, included transient hypertension (14%), anemia (5%), nausea (5%), chills (5%), fever (5%), neutropenia (5%), and hyperglycemia (5%).
“Patients who received [subcutaneous] ALT-803 developed a unique injection site rash reaction that peaked 7-10 days later but resolved typically within 14 days. It was self-limited and resolved on its own,” Dr. Fehniger said.
At the time of the presentation, the best overall response rate was achieved in 11 of 21 patients (52%), with 9 complete responders (43%), and 2 partial responders (10%).
Of the 12 patients treated with ALT-803 subcutaneously, 11 patients had either stable disease, or partial or complete responses. All 11 patients remained on study and were in consolidation or follow-up and have not relapsed, Dr. Fehniger reported.
Among the five rituximab-refractory patients, the researchers observed one complete response, two patients with stable disease (45% and 36% tumor volume decrease), and two patients with partial disease. The durability of the responses can only be understood with longer follow-up, Dr. Fehniger said.
The peripheral blood of the patients was analyzed via flow cytometry and mass cytometry. Over the duration of four weekly doses, there was an increase in percentage (sixfold, P less than .001) and absolute number (10-fold, P less than .001) of natural killer cells at the 15-mcg/kg and 20-mcg/kg subcutaneous dose levels of ALT-803.
These results suggest that further studies of ALT-803 with other therapeutic targeting mAbs, or other immunotherapy modalities, are warranted, the researchers concluded.
Dr. Fehniger reported research funding from Altor BioScience.
SOURCE: Fehniger TA et al. AACR Annual Meeting, Abstract CT146.
REPORTING FROM THE AACR ANNUAL MEETING
Key clinical point:
Major finding: The ALT-803 plus rituximab combination achieved an overall response rate in 52% of patients, a complete response in 43%, and partial response in 10%.
Study details: A phase 1 study of 21 patients with indolent non-Hodgkin lymphoma.
Disclosures: Dr. Fehniger reported research funding from Altor BioScience LLC.
Source: Fehniger TA et al. AACR Annual Meeting, Abstract CT146.
Novartis CAR T-cell therapy adds a lymphoma indication
Novartis’s after failure of two or more lines of systemic therapy.
The Food and Drug Administration approved the expanded indication on May 1. The chimeric antigen receptor (CAR) T-cell therapy was initially approved in Aug. 2017 for refractory or relapsed B-cell precursor acute lymphoblastic leukemia (ALL) in patients up to 25 years old. The new approval brings tisagenlecleucel into direct competition with Gilead Science’s CAR T-cell therapy axicabtagene ciloleucel (Yescarta), which was approved in Oct. 2017 for B-cell lymphoma.
Besides matching the competition, she said the lower price is because tisagenlecleucel takes longer to work for lymphoma, and the response isn’t as potent as for childhood ALL. Novartis is looking into chronic lymphocytic leukemia, multiple myeloma, and solid tumor indications for tisagenlecleucel and other CAR T-cell agents, she added.
The Centers for Medicare & Medicaid Services recently committed to covering outpatient administration of both agents for their initial indications; Novartis is working with CMS for coverage of the new lymphoma indication.
With both products, T cells are collected then shipped off to a company facility where a CAR gene is spliced into their DNA, essentially programming the T cells to attack the targeted cancer. The cells are then infused back into the patient.
In the phase 2 JULIET trial, tisagenlecleucel showed an overall response rate of 50% among 68 B-cell lymphoma patients, with 32% achieving complete response (CR) and 18% achieving partial response (PR). The median duration of response was not reached.
Axicabtagene ciloleucel’s label reports an objective response rate of 72% among 101 patients, with CR in 51% and PR in 21%. Median duration of response was 9.2 months but was also not reached among complete responders.
“Different trials. Different CARTs. Different levels of disease. Our drug is cryopreserved and theirs is not. No way to compare them,” the Novartis spokeswoman said when asked about the response differences.
T-cell reprogramming isn’t clean at this point in medical history; both agents carry black box warnings of potentially fatal cytokine release syndrome and neurologic toxicity, and both are subject to Risk Evaluation and Mitigation Strategy programs.
The B-cell lymphoma indication for both therapies includes diffuse large B-cell lymphoma (DLBCL), high grade B-cell lymphoma, and DLBCL arising from follicular lymphoma. The Gilead product carries an additional indication for primary mediastinal large B-cell lymphoma. Neither agent is indicated for primary central nervous system lymphoma. Both labels say that patients should not donate blood, organs, or tissues after treatment. Tisagenlecleucel labeling also notes that some commercial HIV nucleic acid tests may yield false positives after treatment.
Novartis said in a press release that T cells are treated at the company’s Morris Plains, N.J., facility with a turnaround time of about 22 days. Cryopreservation of the harvested cells gives providers some flexibility in treatment timing.
Novartis’s after failure of two or more lines of systemic therapy.
The Food and Drug Administration approved the expanded indication on May 1. The chimeric antigen receptor (CAR) T-cell therapy was initially approved in Aug. 2017 for refractory or relapsed B-cell precursor acute lymphoblastic leukemia (ALL) in patients up to 25 years old. The new approval brings tisagenlecleucel into direct competition with Gilead Science’s CAR T-cell therapy axicabtagene ciloleucel (Yescarta), which was approved in Oct. 2017 for B-cell lymphoma.
Besides matching the competition, she said the lower price is because tisagenlecleucel takes longer to work for lymphoma, and the response isn’t as potent as for childhood ALL. Novartis is looking into chronic lymphocytic leukemia, multiple myeloma, and solid tumor indications for tisagenlecleucel and other CAR T-cell agents, she added.
The Centers for Medicare & Medicaid Services recently committed to covering outpatient administration of both agents for their initial indications; Novartis is working with CMS for coverage of the new lymphoma indication.
With both products, T cells are collected then shipped off to a company facility where a CAR gene is spliced into their DNA, essentially programming the T cells to attack the targeted cancer. The cells are then infused back into the patient.
In the phase 2 JULIET trial, tisagenlecleucel showed an overall response rate of 50% among 68 B-cell lymphoma patients, with 32% achieving complete response (CR) and 18% achieving partial response (PR). The median duration of response was not reached.
Axicabtagene ciloleucel’s label reports an objective response rate of 72% among 101 patients, with CR in 51% and PR in 21%. Median duration of response was 9.2 months but was also not reached among complete responders.
“Different trials. Different CARTs. Different levels of disease. Our drug is cryopreserved and theirs is not. No way to compare them,” the Novartis spokeswoman said when asked about the response differences.
T-cell reprogramming isn’t clean at this point in medical history; both agents carry black box warnings of potentially fatal cytokine release syndrome and neurologic toxicity, and both are subject to Risk Evaluation and Mitigation Strategy programs.
The B-cell lymphoma indication for both therapies includes diffuse large B-cell lymphoma (DLBCL), high grade B-cell lymphoma, and DLBCL arising from follicular lymphoma. The Gilead product carries an additional indication for primary mediastinal large B-cell lymphoma. Neither agent is indicated for primary central nervous system lymphoma. Both labels say that patients should not donate blood, organs, or tissues after treatment. Tisagenlecleucel labeling also notes that some commercial HIV nucleic acid tests may yield false positives after treatment.
Novartis said in a press release that T cells are treated at the company’s Morris Plains, N.J., facility with a turnaround time of about 22 days. Cryopreservation of the harvested cells gives providers some flexibility in treatment timing.
Novartis’s after failure of two or more lines of systemic therapy.
The Food and Drug Administration approved the expanded indication on May 1. The chimeric antigen receptor (CAR) T-cell therapy was initially approved in Aug. 2017 for refractory or relapsed B-cell precursor acute lymphoblastic leukemia (ALL) in patients up to 25 years old. The new approval brings tisagenlecleucel into direct competition with Gilead Science’s CAR T-cell therapy axicabtagene ciloleucel (Yescarta), which was approved in Oct. 2017 for B-cell lymphoma.
Besides matching the competition, she said the lower price is because tisagenlecleucel takes longer to work for lymphoma, and the response isn’t as potent as for childhood ALL. Novartis is looking into chronic lymphocytic leukemia, multiple myeloma, and solid tumor indications for tisagenlecleucel and other CAR T-cell agents, she added.
The Centers for Medicare & Medicaid Services recently committed to covering outpatient administration of both agents for their initial indications; Novartis is working with CMS for coverage of the new lymphoma indication.
With both products, T cells are collected then shipped off to a company facility where a CAR gene is spliced into their DNA, essentially programming the T cells to attack the targeted cancer. The cells are then infused back into the patient.
In the phase 2 JULIET trial, tisagenlecleucel showed an overall response rate of 50% among 68 B-cell lymphoma patients, with 32% achieving complete response (CR) and 18% achieving partial response (PR). The median duration of response was not reached.
Axicabtagene ciloleucel’s label reports an objective response rate of 72% among 101 patients, with CR in 51% and PR in 21%. Median duration of response was 9.2 months but was also not reached among complete responders.
“Different trials. Different CARTs. Different levels of disease. Our drug is cryopreserved and theirs is not. No way to compare them,” the Novartis spokeswoman said when asked about the response differences.
T-cell reprogramming isn’t clean at this point in medical history; both agents carry black box warnings of potentially fatal cytokine release syndrome and neurologic toxicity, and both are subject to Risk Evaluation and Mitigation Strategy programs.
The B-cell lymphoma indication for both therapies includes diffuse large B-cell lymphoma (DLBCL), high grade B-cell lymphoma, and DLBCL arising from follicular lymphoma. The Gilead product carries an additional indication for primary mediastinal large B-cell lymphoma. Neither agent is indicated for primary central nervous system lymphoma. Both labels say that patients should not donate blood, organs, or tissues after treatment. Tisagenlecleucel labeling also notes that some commercial HIV nucleic acid tests may yield false positives after treatment.
Novartis said in a press release that T cells are treated at the company’s Morris Plains, N.J., facility with a turnaround time of about 22 days. Cryopreservation of the harvested cells gives providers some flexibility in treatment timing.
FDA issues CRL for proposed biosimilar rituximab
The US Food and Drug Administration (FDA) has issued a complete response letter (CRL) saying the agency cannot approve Sandoz’s proposed biosimilar rituximab.
Sandoz submitted the biologics licensing application for the product, known as GP2013, in September 2017.
The company was seeking approval for GP2013 to treat follicular lymphoma (FL), diffuse large B-cell lymphoma, chronic lymphocytic leukemia, rheumatoid arthritis, granulomatosis with polyangiitis, and microscopic polyangiitis.
The drug already has approval for these indications in Europe. The European Commission approved GP2013 (Rixathon) in June 2017.
As for US approval, Sandoz said it is evaluating the content of the FDA’s CRL and “remains committed to further discussions with FDA in order to bring this important medicine to US patients as soon as possible.”
The company said it “stands behind the robust body of evidence included in the regulatory submission” for GP2013.
Part of this evidence is the ASSIST-FL trial, in which researchers compared GP2013 to the reference product, Roche’s MabThera. Results from this trial were published in The Lancet Haematology and presented at ESMO 2017 Congress.
This phase 3 trial included adults with previously untreated, advanced stage FL. Patients received 8 cycles of cyclophosphamide, vincristine, and prednisone with either GP2013 or reference rituximab. Responders then received GP2013 or rituximab monotherapy as maintenance for up to 2 years.
At a median follow-up of 11.6 months, the overall response rate was 87% (271/311) in the GP2013 arm and 88% in the rituximab arm (274/313). Complete response rates were 15% (n=46) and 13% (n=42), respectively.
Rates of adverse events (AEs) were 93% in the GP2013 arm and 91% in the rituximab arm. Rates of serious AEs were 23% and 20%, respectively. The rate of discontinuation due to AEs was 7% in both arms.
The most common AE was neutropenia, which occurred in 26% of patients in the GP2013 arm and 30% of those in the rituximab arm in the combination phase. Rates of neutropenia in the maintenance phase were 10% and 6%, respectively.
The US Food and Drug Administration (FDA) has issued a complete response letter (CRL) saying the agency cannot approve Sandoz’s proposed biosimilar rituximab.
Sandoz submitted the biologics licensing application for the product, known as GP2013, in September 2017.
The company was seeking approval for GP2013 to treat follicular lymphoma (FL), diffuse large B-cell lymphoma, chronic lymphocytic leukemia, rheumatoid arthritis, granulomatosis with polyangiitis, and microscopic polyangiitis.
The drug already has approval for these indications in Europe. The European Commission approved GP2013 (Rixathon) in June 2017.
As for US approval, Sandoz said it is evaluating the content of the FDA’s CRL and “remains committed to further discussions with FDA in order to bring this important medicine to US patients as soon as possible.”
The company said it “stands behind the robust body of evidence included in the regulatory submission” for GP2013.
Part of this evidence is the ASSIST-FL trial, in which researchers compared GP2013 to the reference product, Roche’s MabThera. Results from this trial were published in The Lancet Haematology and presented at ESMO 2017 Congress.
This phase 3 trial included adults with previously untreated, advanced stage FL. Patients received 8 cycles of cyclophosphamide, vincristine, and prednisone with either GP2013 or reference rituximab. Responders then received GP2013 or rituximab monotherapy as maintenance for up to 2 years.
At a median follow-up of 11.6 months, the overall response rate was 87% (271/311) in the GP2013 arm and 88% in the rituximab arm (274/313). Complete response rates were 15% (n=46) and 13% (n=42), respectively.
Rates of adverse events (AEs) were 93% in the GP2013 arm and 91% in the rituximab arm. Rates of serious AEs were 23% and 20%, respectively. The rate of discontinuation due to AEs was 7% in both arms.
The most common AE was neutropenia, which occurred in 26% of patients in the GP2013 arm and 30% of those in the rituximab arm in the combination phase. Rates of neutropenia in the maintenance phase were 10% and 6%, respectively.
The US Food and Drug Administration (FDA) has issued a complete response letter (CRL) saying the agency cannot approve Sandoz’s proposed biosimilar rituximab.
Sandoz submitted the biologics licensing application for the product, known as GP2013, in September 2017.
The company was seeking approval for GP2013 to treat follicular lymphoma (FL), diffuse large B-cell lymphoma, chronic lymphocytic leukemia, rheumatoid arthritis, granulomatosis with polyangiitis, and microscopic polyangiitis.
The drug already has approval for these indications in Europe. The European Commission approved GP2013 (Rixathon) in June 2017.
As for US approval, Sandoz said it is evaluating the content of the FDA’s CRL and “remains committed to further discussions with FDA in order to bring this important medicine to US patients as soon as possible.”
The company said it “stands behind the robust body of evidence included in the regulatory submission” for GP2013.
Part of this evidence is the ASSIST-FL trial, in which researchers compared GP2013 to the reference product, Roche’s MabThera. Results from this trial were published in The Lancet Haematology and presented at ESMO 2017 Congress.
This phase 3 trial included adults with previously untreated, advanced stage FL. Patients received 8 cycles of cyclophosphamide, vincristine, and prednisone with either GP2013 or reference rituximab. Responders then received GP2013 or rituximab monotherapy as maintenance for up to 2 years.
At a median follow-up of 11.6 months, the overall response rate was 87% (271/311) in the GP2013 arm and 88% in the rituximab arm (274/313). Complete response rates were 15% (n=46) and 13% (n=42), respectively.
Rates of adverse events (AEs) were 93% in the GP2013 arm and 91% in the rituximab arm. Rates of serious AEs were 23% and 20%, respectively. The rate of discontinuation due to AEs was 7% in both arms.
The most common AE was neutropenia, which occurred in 26% of patients in the GP2013 arm and 30% of those in the rituximab arm in the combination phase. Rates of neutropenia in the maintenance phase were 10% and 6%, respectively.