User login
Immunotherapy with less intense chemotherapy safe and effective for older patients with ALL
according to the results of a single-arm phase 2 study.
After a median follow-up of 29 months, 2-year progression-free survival was 59% with a median of 25 months for 52 patients with newly diagnosed disease who were aged 60 years or older, Hagop Kantarjian, MD, and his associates reported online in the Lancet Oncology.
Nearly every patient experienced an overall response (98%). The treatment also appeared to be safe, Dr. Kantarjian and associates wrote.
They reported that no patient died within 4 weeks of treatment and that four patients experienced veno-occlusive disease. Common grade 3 or 4 side effects included thrombocytopenia (81%), consolidation chemotherapy (69%), hyperglycemia (54%), infections during induction (52%), hypokalemia (31%), increased aminotransferases (19%), hyperbilirubinemia (17%), and hemorrhage (15%). Six patients died from treatment-related side effects, five of whom died from sepsis and one of whom died as a result of veno-occlusive disease.
Inotuzumab ozogamicin is an anti-CD22 monoclonal antibody that is bound to the toxin calicheamicin. It has shown “substantial improvements” for some patient subgroups when added to hyper-CVAD chemotherapy (hyperfractionated cyclophosphamide, vincristine, doxorubicin, and dexamethasone alternating with methotrexate and high-dose cytarabine); however, older patients have yet to experience a similar benefit, Dr. Kantarjian and his associates said.
Both CVAD and inotuzumab ozogamicin have shown to be quite toxic for older patients. So – in order limit increased toxicity – Dr. Kantarjian and his associates lowered the intensity of hyper-CVAD. They accomplished this by reducing some aspects of the regimen by up to 50% and eliminating anthracyclines altogether. The researchers referred to this regimen as “mini–hyper-CVD.” The mini–hyper-CVD did not appear to impact activity.
“In our study, activity did not appear to be compromised by the use of lower-intensity chemotherapy in combination with a novel monoclonal antibody,” they wrote.
Dr. Kantarjian and his associates noted that, while they observed fewer deaths compared with previously published data, the number of deaths they did observe in patients who achieved a complete response was high.
They suggested that modifications to the regimen could further improve safety and that such modifications required further research.
For the time being, however, the researchers noted that this low-intensity regimen, in combination in inotuzumab ozogamicin, is highly effective and that these data now require prospective confirmation in a randomized, phase 3 setting.
The study was funded by the MD Anderson Cancer Center. Researchers reported that Pfizer provided inotuzumab ozogamicin free of charge and that Dr. Kantarjian and two other researchers received grants from Pfizer.
SOURCE: Kantarjian H et al. Lancet Oncol. 2018 Jan 15. doi: 10.1016/S1470-2945(18)30011-1.
The efficacy and safety of inotuzumab ozogamicin reported by Hagop Kantarjian, MD, and his colleagues are an encouraging building block for older patents with acute lymphoblastic leukemia, according to Carmelo Rizzari, MD.
“The authors have upgraded the valuable premises available from early inotuzumab ozogamicin studies into more concrete promises by rationally integrating the drug in a reduced-intensity chemotherapy regimen,” wrote Dr. Rizzari in an accompanying editorial published Lancet Oncology.
Adding inotuzumab ozogamicin to chemotherapy has shown efficacy and safety for both pediatric and adult patients with relapsed or refractory acute lymphoblastic leukemia and therefore deserves more investigation in the “neglected” subgroup of older patients, according to Dr. Rizzari.
“Monoclonal antibodies represent, in the wider settings of immunotherapy, viable options to improve the results obtained in different subsets of patients with acute lymphoblastic leukemia,” wrote Dr. Rizzari.
Echoing the researchers, Dr. Rizzari mentioned the importance of the next step – a phase 3 trial comparing inotuzumab ozogamicin with the current standard of care – but also mentioned that a specific, widely recognized standard of care for this patient subgroup is difficult to pin down.
Dr. Rizzari is with the Pediatric Hematology-Oncology Unit of the University of Milano-Bicocca in Milan. These comments are based on an accompanying editorial published online in Lancet Oncology (2018 Jan 15. doi: 10.1016/S1470-2045[18]30013-5 ). Dr. Rizzari declared no competing interests.
The efficacy and safety of inotuzumab ozogamicin reported by Hagop Kantarjian, MD, and his colleagues are an encouraging building block for older patents with acute lymphoblastic leukemia, according to Carmelo Rizzari, MD.
“The authors have upgraded the valuable premises available from early inotuzumab ozogamicin studies into more concrete promises by rationally integrating the drug in a reduced-intensity chemotherapy regimen,” wrote Dr. Rizzari in an accompanying editorial published Lancet Oncology.
Adding inotuzumab ozogamicin to chemotherapy has shown efficacy and safety for both pediatric and adult patients with relapsed or refractory acute lymphoblastic leukemia and therefore deserves more investigation in the “neglected” subgroup of older patients, according to Dr. Rizzari.
“Monoclonal antibodies represent, in the wider settings of immunotherapy, viable options to improve the results obtained in different subsets of patients with acute lymphoblastic leukemia,” wrote Dr. Rizzari.
Echoing the researchers, Dr. Rizzari mentioned the importance of the next step – a phase 3 trial comparing inotuzumab ozogamicin with the current standard of care – but also mentioned that a specific, widely recognized standard of care for this patient subgroup is difficult to pin down.
Dr. Rizzari is with the Pediatric Hematology-Oncology Unit of the University of Milano-Bicocca in Milan. These comments are based on an accompanying editorial published online in Lancet Oncology (2018 Jan 15. doi: 10.1016/S1470-2045[18]30013-5 ). Dr. Rizzari declared no competing interests.
The efficacy and safety of inotuzumab ozogamicin reported by Hagop Kantarjian, MD, and his colleagues are an encouraging building block for older patents with acute lymphoblastic leukemia, according to Carmelo Rizzari, MD.
“The authors have upgraded the valuable premises available from early inotuzumab ozogamicin studies into more concrete promises by rationally integrating the drug in a reduced-intensity chemotherapy regimen,” wrote Dr. Rizzari in an accompanying editorial published Lancet Oncology.
Adding inotuzumab ozogamicin to chemotherapy has shown efficacy and safety for both pediatric and adult patients with relapsed or refractory acute lymphoblastic leukemia and therefore deserves more investigation in the “neglected” subgroup of older patients, according to Dr. Rizzari.
“Monoclonal antibodies represent, in the wider settings of immunotherapy, viable options to improve the results obtained in different subsets of patients with acute lymphoblastic leukemia,” wrote Dr. Rizzari.
Echoing the researchers, Dr. Rizzari mentioned the importance of the next step – a phase 3 trial comparing inotuzumab ozogamicin with the current standard of care – but also mentioned that a specific, widely recognized standard of care for this patient subgroup is difficult to pin down.
Dr. Rizzari is with the Pediatric Hematology-Oncology Unit of the University of Milano-Bicocca in Milan. These comments are based on an accompanying editorial published online in Lancet Oncology (2018 Jan 15. doi: 10.1016/S1470-2045[18]30013-5 ). Dr. Rizzari declared no competing interests.
according to the results of a single-arm phase 2 study.
After a median follow-up of 29 months, 2-year progression-free survival was 59% with a median of 25 months for 52 patients with newly diagnosed disease who were aged 60 years or older, Hagop Kantarjian, MD, and his associates reported online in the Lancet Oncology.
Nearly every patient experienced an overall response (98%). The treatment also appeared to be safe, Dr. Kantarjian and associates wrote.
They reported that no patient died within 4 weeks of treatment and that four patients experienced veno-occlusive disease. Common grade 3 or 4 side effects included thrombocytopenia (81%), consolidation chemotherapy (69%), hyperglycemia (54%), infections during induction (52%), hypokalemia (31%), increased aminotransferases (19%), hyperbilirubinemia (17%), and hemorrhage (15%). Six patients died from treatment-related side effects, five of whom died from sepsis and one of whom died as a result of veno-occlusive disease.
Inotuzumab ozogamicin is an anti-CD22 monoclonal antibody that is bound to the toxin calicheamicin. It has shown “substantial improvements” for some patient subgroups when added to hyper-CVAD chemotherapy (hyperfractionated cyclophosphamide, vincristine, doxorubicin, and dexamethasone alternating with methotrexate and high-dose cytarabine); however, older patients have yet to experience a similar benefit, Dr. Kantarjian and his associates said.
Both CVAD and inotuzumab ozogamicin have shown to be quite toxic for older patients. So – in order limit increased toxicity – Dr. Kantarjian and his associates lowered the intensity of hyper-CVAD. They accomplished this by reducing some aspects of the regimen by up to 50% and eliminating anthracyclines altogether. The researchers referred to this regimen as “mini–hyper-CVD.” The mini–hyper-CVD did not appear to impact activity.
“In our study, activity did not appear to be compromised by the use of lower-intensity chemotherapy in combination with a novel monoclonal antibody,” they wrote.
Dr. Kantarjian and his associates noted that, while they observed fewer deaths compared with previously published data, the number of deaths they did observe in patients who achieved a complete response was high.
They suggested that modifications to the regimen could further improve safety and that such modifications required further research.
For the time being, however, the researchers noted that this low-intensity regimen, in combination in inotuzumab ozogamicin, is highly effective and that these data now require prospective confirmation in a randomized, phase 3 setting.
The study was funded by the MD Anderson Cancer Center. Researchers reported that Pfizer provided inotuzumab ozogamicin free of charge and that Dr. Kantarjian and two other researchers received grants from Pfizer.
SOURCE: Kantarjian H et al. Lancet Oncol. 2018 Jan 15. doi: 10.1016/S1470-2945(18)30011-1.
according to the results of a single-arm phase 2 study.
After a median follow-up of 29 months, 2-year progression-free survival was 59% with a median of 25 months for 52 patients with newly diagnosed disease who were aged 60 years or older, Hagop Kantarjian, MD, and his associates reported online in the Lancet Oncology.
Nearly every patient experienced an overall response (98%). The treatment also appeared to be safe, Dr. Kantarjian and associates wrote.
They reported that no patient died within 4 weeks of treatment and that four patients experienced veno-occlusive disease. Common grade 3 or 4 side effects included thrombocytopenia (81%), consolidation chemotherapy (69%), hyperglycemia (54%), infections during induction (52%), hypokalemia (31%), increased aminotransferases (19%), hyperbilirubinemia (17%), and hemorrhage (15%). Six patients died from treatment-related side effects, five of whom died from sepsis and one of whom died as a result of veno-occlusive disease.
Inotuzumab ozogamicin is an anti-CD22 monoclonal antibody that is bound to the toxin calicheamicin. It has shown “substantial improvements” for some patient subgroups when added to hyper-CVAD chemotherapy (hyperfractionated cyclophosphamide, vincristine, doxorubicin, and dexamethasone alternating with methotrexate and high-dose cytarabine); however, older patients have yet to experience a similar benefit, Dr. Kantarjian and his associates said.
Both CVAD and inotuzumab ozogamicin have shown to be quite toxic for older patients. So – in order limit increased toxicity – Dr. Kantarjian and his associates lowered the intensity of hyper-CVAD. They accomplished this by reducing some aspects of the regimen by up to 50% and eliminating anthracyclines altogether. The researchers referred to this regimen as “mini–hyper-CVD.” The mini–hyper-CVD did not appear to impact activity.
“In our study, activity did not appear to be compromised by the use of lower-intensity chemotherapy in combination with a novel monoclonal antibody,” they wrote.
Dr. Kantarjian and his associates noted that, while they observed fewer deaths compared with previously published data, the number of deaths they did observe in patients who achieved a complete response was high.
They suggested that modifications to the regimen could further improve safety and that such modifications required further research.
For the time being, however, the researchers noted that this low-intensity regimen, in combination in inotuzumab ozogamicin, is highly effective and that these data now require prospective confirmation in a randomized, phase 3 setting.
The study was funded by the MD Anderson Cancer Center. Researchers reported that Pfizer provided inotuzumab ozogamicin free of charge and that Dr. Kantarjian and two other researchers received grants from Pfizer.
SOURCE: Kantarjian H et al. Lancet Oncol. 2018 Jan 15. doi: 10.1016/S1470-2945(18)30011-1.
FROM LANCET ONCOLOGY
Key clinical point: Inotuzumab ozogamicin with less intense chemotherapy is safe and effective for older patients with ALL.
Major finding: After a median follow-up of 29 months, 59% of patients experienced 2-year recurrence-free survival, 56% experienced 3-year overall survival and only four patients died within four weeks of treatment.
Data source: Single-arm phase 2 study of 52 patients aged 60 years or older with newly diagnosed Philadelphia chromosome–negative ALL.
Disclosures: The study was funded by a grant from the MD Anderson Cancer Center. Pfizer provided inotuzumab ozogamicin free of charge. Dr. Kantarjian and two other researchers reported receiving grants from Pfizer.
Source: Kantarjian H et al. Lancet Oncol. 2018 Jan 15. doi: 10.1016/S1470-2945(18)30011-1.
RELAZA2: MRD-guided azacitidine reduces relapse risk in MDS and AML
ATLANTA – in patients with myelodysplastic syndrome (MDS) or acute myeloid leukemia (AML) who are at high risk for relapse, according to findings from the open-label, interventional RELAZA2 trial.
Of 205 patients screened between 2011 and 2015 at 11 centers in Germany, 53 became minimal residual disease (MRD) positive while remaining in hematological remission. All 53 started azacitidine-based preemptive treatment, and 6 months after the initiation of the MRD-guided therapy, 31 (58%) were still in complete remission, while 22 (42%) relapsed after a median of three treatment cycles, Uwe Platzbecker, MD, reported at the annual meeting of the American Society of Hematology.
Of those still in complete remission, 21 patients responded with a decline of MRD below a predefined threshold, and 10 achieved stabilization in the absence of relapse, said Dr. Platzbecker of the University Hospital Carl Gustav Carus Dresden, Germany.
The overall response rate was greater in those who underwent allogeneic hematopoietic stem cell transplantation (71% vs. 48%), he noted.
“After 6 months, 24 patients continued to receive a median of nine subsequent azacitidine cycles. Seven patients completed 24 months of treatment according to protocol. Eventually, hematologic relapse occurred in eight of those patients (33%) but was delayed until a median of 397 days after initial MRD detection,” he said in an interview, adding that, overall, 26 of the 53 patients in the study (49%) experienced hematologic relapse, which was delayed until a median of 422 days after initial MRD detection.
Study subjects were adults with a median age of 59 years with measurable MRD suggestive of imminent relapse but who were still in CR. Most (48) had AML, and 5 had MDS. They were treated preemptively with six cycles of 75 mg/m2 of azacitidine given subcutaneously on days 1-7 of each 1-month cycle. Those who continued treatment beyond the initial 6 months were treated with risk-adapted azacitidine-based therapy for up to 18 additional months.
Treatment was well tolerated. Grade 3 or 4 thrombocytopenia occurred in three patients, and grade 3 or 4 neutropenia occurred in 45 patients. Infections and pneumonia, which occurred in four and three patients, respectively, were the main serious side effects during the first 6 cycles.
“With a median follow-up of 13 months after the start of MRD-guided preemptive treatment, the actual overall and progression free survival rate was 76% and 42%, respectively,” Dr. Platzbecker said.
Chemotherapy frequently results in complete remission in patients with MDS or AML, but a substantial proportion of patients relapse even after allogeneic stem cell transplantation, he said, noting that treatment options in these patients are limited.
In the prospective RELAZA 1 trial, short-term preemptive azacitidine therapy was associated with sustained responses. RELAZA2 was designed to assess the ability of early nonintensive azacitidine treatment, directed by MRD monitoring after allogeneic stem cell transplantation and chemotherapy, prior to avert relapse.
The findings suggest that this approach is effective in patients at higher risk of relapse, but the success of treatment seems to be context dependent, Dr. Platzbecker said, explaining that this finding emphasizes the potential immunomodulatory role of hypomethylating agents.
“The study supports the prognostic importance of MRD in AML and may serve as a platform for future studies in combining hypomethylating agents and novel targeted therapies,” he concluded.
The RELAZA2 trial is sponsored by Technische Universität Dresden. Dr. Platzbecker reported serving as a consultant for, and receiving honoraria and research funding from Celgene, Janssen, Novartis, and Acceleron.
SOURCE: Platzbecker U et al. ASH 2017 Abstract #565.
ATLANTA – in patients with myelodysplastic syndrome (MDS) or acute myeloid leukemia (AML) who are at high risk for relapse, according to findings from the open-label, interventional RELAZA2 trial.
Of 205 patients screened between 2011 and 2015 at 11 centers in Germany, 53 became minimal residual disease (MRD) positive while remaining in hematological remission. All 53 started azacitidine-based preemptive treatment, and 6 months after the initiation of the MRD-guided therapy, 31 (58%) were still in complete remission, while 22 (42%) relapsed after a median of three treatment cycles, Uwe Platzbecker, MD, reported at the annual meeting of the American Society of Hematology.
Of those still in complete remission, 21 patients responded with a decline of MRD below a predefined threshold, and 10 achieved stabilization in the absence of relapse, said Dr. Platzbecker of the University Hospital Carl Gustav Carus Dresden, Germany.
The overall response rate was greater in those who underwent allogeneic hematopoietic stem cell transplantation (71% vs. 48%), he noted.
“After 6 months, 24 patients continued to receive a median of nine subsequent azacitidine cycles. Seven patients completed 24 months of treatment according to protocol. Eventually, hematologic relapse occurred in eight of those patients (33%) but was delayed until a median of 397 days after initial MRD detection,” he said in an interview, adding that, overall, 26 of the 53 patients in the study (49%) experienced hematologic relapse, which was delayed until a median of 422 days after initial MRD detection.
Study subjects were adults with a median age of 59 years with measurable MRD suggestive of imminent relapse but who were still in CR. Most (48) had AML, and 5 had MDS. They were treated preemptively with six cycles of 75 mg/m2 of azacitidine given subcutaneously on days 1-7 of each 1-month cycle. Those who continued treatment beyond the initial 6 months were treated with risk-adapted azacitidine-based therapy for up to 18 additional months.
Treatment was well tolerated. Grade 3 or 4 thrombocytopenia occurred in three patients, and grade 3 or 4 neutropenia occurred in 45 patients. Infections and pneumonia, which occurred in four and three patients, respectively, were the main serious side effects during the first 6 cycles.
“With a median follow-up of 13 months after the start of MRD-guided preemptive treatment, the actual overall and progression free survival rate was 76% and 42%, respectively,” Dr. Platzbecker said.
Chemotherapy frequently results in complete remission in patients with MDS or AML, but a substantial proportion of patients relapse even after allogeneic stem cell transplantation, he said, noting that treatment options in these patients are limited.
In the prospective RELAZA 1 trial, short-term preemptive azacitidine therapy was associated with sustained responses. RELAZA2 was designed to assess the ability of early nonintensive azacitidine treatment, directed by MRD monitoring after allogeneic stem cell transplantation and chemotherapy, prior to avert relapse.
The findings suggest that this approach is effective in patients at higher risk of relapse, but the success of treatment seems to be context dependent, Dr. Platzbecker said, explaining that this finding emphasizes the potential immunomodulatory role of hypomethylating agents.
“The study supports the prognostic importance of MRD in AML and may serve as a platform for future studies in combining hypomethylating agents and novel targeted therapies,” he concluded.
The RELAZA2 trial is sponsored by Technische Universität Dresden. Dr. Platzbecker reported serving as a consultant for, and receiving honoraria and research funding from Celgene, Janssen, Novartis, and Acceleron.
SOURCE: Platzbecker U et al. ASH 2017 Abstract #565.
ATLANTA – in patients with myelodysplastic syndrome (MDS) or acute myeloid leukemia (AML) who are at high risk for relapse, according to findings from the open-label, interventional RELAZA2 trial.
Of 205 patients screened between 2011 and 2015 at 11 centers in Germany, 53 became minimal residual disease (MRD) positive while remaining in hematological remission. All 53 started azacitidine-based preemptive treatment, and 6 months after the initiation of the MRD-guided therapy, 31 (58%) were still in complete remission, while 22 (42%) relapsed after a median of three treatment cycles, Uwe Platzbecker, MD, reported at the annual meeting of the American Society of Hematology.
Of those still in complete remission, 21 patients responded with a decline of MRD below a predefined threshold, and 10 achieved stabilization in the absence of relapse, said Dr. Platzbecker of the University Hospital Carl Gustav Carus Dresden, Germany.
The overall response rate was greater in those who underwent allogeneic hematopoietic stem cell transplantation (71% vs. 48%), he noted.
“After 6 months, 24 patients continued to receive a median of nine subsequent azacitidine cycles. Seven patients completed 24 months of treatment according to protocol. Eventually, hematologic relapse occurred in eight of those patients (33%) but was delayed until a median of 397 days after initial MRD detection,” he said in an interview, adding that, overall, 26 of the 53 patients in the study (49%) experienced hematologic relapse, which was delayed until a median of 422 days after initial MRD detection.
Study subjects were adults with a median age of 59 years with measurable MRD suggestive of imminent relapse but who were still in CR. Most (48) had AML, and 5 had MDS. They were treated preemptively with six cycles of 75 mg/m2 of azacitidine given subcutaneously on days 1-7 of each 1-month cycle. Those who continued treatment beyond the initial 6 months were treated with risk-adapted azacitidine-based therapy for up to 18 additional months.
Treatment was well tolerated. Grade 3 or 4 thrombocytopenia occurred in three patients, and grade 3 or 4 neutropenia occurred in 45 patients. Infections and pneumonia, which occurred in four and three patients, respectively, were the main serious side effects during the first 6 cycles.
“With a median follow-up of 13 months after the start of MRD-guided preemptive treatment, the actual overall and progression free survival rate was 76% and 42%, respectively,” Dr. Platzbecker said.
Chemotherapy frequently results in complete remission in patients with MDS or AML, but a substantial proportion of patients relapse even after allogeneic stem cell transplantation, he said, noting that treatment options in these patients are limited.
In the prospective RELAZA 1 trial, short-term preemptive azacitidine therapy was associated with sustained responses. RELAZA2 was designed to assess the ability of early nonintensive azacitidine treatment, directed by MRD monitoring after allogeneic stem cell transplantation and chemotherapy, prior to avert relapse.
The findings suggest that this approach is effective in patients at higher risk of relapse, but the success of treatment seems to be context dependent, Dr. Platzbecker said, explaining that this finding emphasizes the potential immunomodulatory role of hypomethylating agents.
“The study supports the prognostic importance of MRD in AML and may serve as a platform for future studies in combining hypomethylating agents and novel targeted therapies,” he concluded.
The RELAZA2 trial is sponsored by Technische Universität Dresden. Dr. Platzbecker reported serving as a consultant for, and receiving honoraria and research funding from Celgene, Janssen, Novartis, and Acceleron.
SOURCE: Platzbecker U et al. ASH 2017 Abstract #565.
REPORTING FROM ASH 2017
Key clinical point: MRD-guided azacitidine therapy reduces hematological relapse in high-risk MDS/AML.
Major finding: The relapse-free survival rate at 6 months was 58%.
Study details: An analysis of 53 patients from the open-label RELAZA2 trial.
Disclosures: The RELAZA2 trial is sponsored by Technische Universität Dresden, Germany. Dr. Platzbecker reported serving as a consultant for and receiving honoraria and research funding from Celgene, Janssen, Novartis, and Acceleron Pharma.
Source: Platzbecker U et al. ASH 2017 Abstract #565.
Predicting response to AZA in MDS, CMML
Researchers have developed a technique that may help predict whether patients with myelodysplastic syndromes (MDS) or chronic myelomonocytic leukemia (CMML) will respond to treatment with azacytidine (AZA).
“The new method, called AZA-MS, utilizes a cutting-edge technique known as mass spectrometry to measure the different forms of AZA inside blood cells of patients—such as the AZA molecules that are incorporated into the DNA or RNA,” said Ashwin Unnikrishnan, PhD, of the University of New South Wales in Sydney, Australia.
With this method, Dr Unnikrishnan and his colleagues found that patients who do not respond to AZA may incorporate fewer AZA molecules in their DNA and have lower DNA demethylation than responders. However, this is not always the case.
The researchers reported these findings in Leukemia.
The team initially tested AZA-MS in AZA-treated RKO cells and found that AZA-MS could quantify the ribonucleoside (5-AZA-cR) and deoxyribonucleoside (5-AZA-CdR) forms of AZA in RNA, DNA, and the cytoplasm—all in the same sample.
The researchers also found that AZA induced dose-dependent DNA demethylation but did not have an effect on RNA methylation.
The team then used AZA-MS to analyze bone marrow samples from patients with MDS (n=4) or CMML (n=4) who were undergoing treatment with AZA. All of the patients had received at least 6 cycles of the drug.
Each patient had 3 bone marrow samples collected—one immediately before starting treatment; one on day 8 of cycle 1 (C1d8); and one on day 28 of cycle 1 (C1d28), when they had spent 20 days off the drug.
Four of the patients were complete responders, and 4 were nonresponders. In each group, 2 patients had MDS, and 2 had CMML.
At C1d8, DNA-5-AZA-CdR was significantly greater in responders than nonresponders. And, overall, responders had increased DNA demethylation compared to nonresponders.
However, the researchers also observed differences among the nonresponders. Two nonresponders had very low levels of DNA-5-AZA-CdR at C1d8 and no demethylation. The other 2 nonresponders had much higher DNA-5-AZA-CdR and DNA demethylation levels, which were comparable to levels in responders.
The researchers said they could detect AZA and DNA-5-AZA-CdR intracellularly, as well as RNA-AZA, in the nonresponders with minimal DNA-5-AZA-CdR and DNA demethylation.
The team said this suggests that neither cellular uptake nor intracellular metabolism explain the low DNA-5-AZA-CdR in these patients. Instead, the researchers believe these patients may have a greater proportion of bone marrow cells that are quiescent and not undergoing DNA replication.
The researchers also believe the nonresponders with higher DNA-5-AZA-CdR may be explained by a failure to induce an interferon response, which is necessary for a clinical response.
On the other hand, these nonresponders could have defective immune cell-mediated clearance of dysplastic cells or increased tolerance to this clearance, the researchers said.
The team also noted that, at C1d28, DNA-5-AZA-CdR levels dropped (but were still detectable) in all 8 patients, and DNA methylation had nearly returned to pretreatment levels in all patients. ![]()
Researchers have developed a technique that may help predict whether patients with myelodysplastic syndromes (MDS) or chronic myelomonocytic leukemia (CMML) will respond to treatment with azacytidine (AZA).
“The new method, called AZA-MS, utilizes a cutting-edge technique known as mass spectrometry to measure the different forms of AZA inside blood cells of patients—such as the AZA molecules that are incorporated into the DNA or RNA,” said Ashwin Unnikrishnan, PhD, of the University of New South Wales in Sydney, Australia.
With this method, Dr Unnikrishnan and his colleagues found that patients who do not respond to AZA may incorporate fewer AZA molecules in their DNA and have lower DNA demethylation than responders. However, this is not always the case.
The researchers reported these findings in Leukemia.
The team initially tested AZA-MS in AZA-treated RKO cells and found that AZA-MS could quantify the ribonucleoside (5-AZA-cR) and deoxyribonucleoside (5-AZA-CdR) forms of AZA in RNA, DNA, and the cytoplasm—all in the same sample.
The researchers also found that AZA induced dose-dependent DNA demethylation but did not have an effect on RNA methylation.
The team then used AZA-MS to analyze bone marrow samples from patients with MDS (n=4) or CMML (n=4) who were undergoing treatment with AZA. All of the patients had received at least 6 cycles of the drug.
Each patient had 3 bone marrow samples collected—one immediately before starting treatment; one on day 8 of cycle 1 (C1d8); and one on day 28 of cycle 1 (C1d28), when they had spent 20 days off the drug.
Four of the patients were complete responders, and 4 were nonresponders. In each group, 2 patients had MDS, and 2 had CMML.
At C1d8, DNA-5-AZA-CdR was significantly greater in responders than nonresponders. And, overall, responders had increased DNA demethylation compared to nonresponders.
However, the researchers also observed differences among the nonresponders. Two nonresponders had very low levels of DNA-5-AZA-CdR at C1d8 and no demethylation. The other 2 nonresponders had much higher DNA-5-AZA-CdR and DNA demethylation levels, which were comparable to levels in responders.
The researchers said they could detect AZA and DNA-5-AZA-CdR intracellularly, as well as RNA-AZA, in the nonresponders with minimal DNA-5-AZA-CdR and DNA demethylation.
The team said this suggests that neither cellular uptake nor intracellular metabolism explain the low DNA-5-AZA-CdR in these patients. Instead, the researchers believe these patients may have a greater proportion of bone marrow cells that are quiescent and not undergoing DNA replication.
The researchers also believe the nonresponders with higher DNA-5-AZA-CdR may be explained by a failure to induce an interferon response, which is necessary for a clinical response.
On the other hand, these nonresponders could have defective immune cell-mediated clearance of dysplastic cells or increased tolerance to this clearance, the researchers said.
The team also noted that, at C1d28, DNA-5-AZA-CdR levels dropped (but were still detectable) in all 8 patients, and DNA methylation had nearly returned to pretreatment levels in all patients. ![]()
Researchers have developed a technique that may help predict whether patients with myelodysplastic syndromes (MDS) or chronic myelomonocytic leukemia (CMML) will respond to treatment with azacytidine (AZA).
“The new method, called AZA-MS, utilizes a cutting-edge technique known as mass spectrometry to measure the different forms of AZA inside blood cells of patients—such as the AZA molecules that are incorporated into the DNA or RNA,” said Ashwin Unnikrishnan, PhD, of the University of New South Wales in Sydney, Australia.
With this method, Dr Unnikrishnan and his colleagues found that patients who do not respond to AZA may incorporate fewer AZA molecules in their DNA and have lower DNA demethylation than responders. However, this is not always the case.
The researchers reported these findings in Leukemia.
The team initially tested AZA-MS in AZA-treated RKO cells and found that AZA-MS could quantify the ribonucleoside (5-AZA-cR) and deoxyribonucleoside (5-AZA-CdR) forms of AZA in RNA, DNA, and the cytoplasm—all in the same sample.
The researchers also found that AZA induced dose-dependent DNA demethylation but did not have an effect on RNA methylation.
The team then used AZA-MS to analyze bone marrow samples from patients with MDS (n=4) or CMML (n=4) who were undergoing treatment with AZA. All of the patients had received at least 6 cycles of the drug.
Each patient had 3 bone marrow samples collected—one immediately before starting treatment; one on day 8 of cycle 1 (C1d8); and one on day 28 of cycle 1 (C1d28), when they had spent 20 days off the drug.
Four of the patients were complete responders, and 4 were nonresponders. In each group, 2 patients had MDS, and 2 had CMML.
At C1d8, DNA-5-AZA-CdR was significantly greater in responders than nonresponders. And, overall, responders had increased DNA demethylation compared to nonresponders.
However, the researchers also observed differences among the nonresponders. Two nonresponders had very low levels of DNA-5-AZA-CdR at C1d8 and no demethylation. The other 2 nonresponders had much higher DNA-5-AZA-CdR and DNA demethylation levels, which were comparable to levels in responders.
The researchers said they could detect AZA and DNA-5-AZA-CdR intracellularly, as well as RNA-AZA, in the nonresponders with minimal DNA-5-AZA-CdR and DNA demethylation.
The team said this suggests that neither cellular uptake nor intracellular metabolism explain the low DNA-5-AZA-CdR in these patients. Instead, the researchers believe these patients may have a greater proportion of bone marrow cells that are quiescent and not undergoing DNA replication.
The researchers also believe the nonresponders with higher DNA-5-AZA-CdR may be explained by a failure to induce an interferon response, which is necessary for a clinical response.
On the other hand, these nonresponders could have defective immune cell-mediated clearance of dysplastic cells or increased tolerance to this clearance, the researchers said.
The team also noted that, at C1d28, DNA-5-AZA-CdR levels dropped (but were still detectable) in all 8 patients, and DNA methylation had nearly returned to pretreatment levels in all patients. ![]()
Bright light therapy improves sleep in cancer survivors
Results of a pilot study suggest that systematic bright light exposure can improve sleep in fatigued cancer survivors.
Subjects who were exposed to bright light every morning for 4 weeks had a significantly greater improvement in sleep efficiency than those who were exposed to dim light over the same period.
In fact, subjects in the bright light group were able to achieve clinically normal levels of sleep efficiency, and subjects in the dim light group were not.
Sleep efficiency is the percentage of time in bed that subjects spent sleeping.
Lisa M. Wu, PhD, of Northwestern University in Chicago, Illinois, and her colleagues reported these results in the Journal of Clinical Sleep Medicine.
The team noted that cancer patients report sleep disturbances at a significantly higher rate than the general population. Between 23% and 44% of cancer patients experience insomnia symptoms even years after treatment.
With this in mind, the researchers studied 44 individuals who had completed cancer treatment and met criteria for clinically significant fatigue at screening.
The subjects had an average age of 53.6, and 75% percent were female. Roughly 55% (n=24) had been diagnosed with a hematologic malignancy.
The subjects were randomized to a bright white light intervention or a dim red light intervention. Subjects in both treatment arms were provided with a light box and instructed to use it every morning for 30 minutes for 4 weeks. Sleep was evaluated using wrist actigraphy and the Pittsburgh Sleep Quality Index.
At baseline, 52.6% of subjects in the dim light group and 60% in the bright light group exceeded the clinical cutoff for poor sleep efficiency (≤ 85%). The mean sleep efficiency was 81.8% and 82.8%, respectively.
During the study period, sleep efficiency improved significantly more among subjects exposed to the bright light than those exposed to the dim light (P=0.003).
The mean sleep efficiency was in the clinically normal range for subjects in the bright light group at the end of the intervention (86.06%) and 3 weeks after (85.77%).
However, the cutoff for poor sleep efficiency was not reached in the dim light group, either at the end of the intervention (mean=79.35%) or 3 weeks after (mean=80.88%).
Total sleep time tended to increase over the study period for subjects in the bright light group, but there was no significant difference in total sleep time between the bright light and dim light groups.
Likewise, there was no significant between-group difference in waking after sleep onset, although this outcome tended to decrease over the study period for subjects in the bright light group.
“Systematic light exposure using bright white light is a low-cost and easily disseminated intervention that offers a feasible and potentially effective alternative to improve sleep in cancer survivors,” Dr Wu said.
However, she and her colleagues noted that larger-scale studies are needed. ![]()
Results of a pilot study suggest that systematic bright light exposure can improve sleep in fatigued cancer survivors.
Subjects who were exposed to bright light every morning for 4 weeks had a significantly greater improvement in sleep efficiency than those who were exposed to dim light over the same period.
In fact, subjects in the bright light group were able to achieve clinically normal levels of sleep efficiency, and subjects in the dim light group were not.
Sleep efficiency is the percentage of time in bed that subjects spent sleeping.
Lisa M. Wu, PhD, of Northwestern University in Chicago, Illinois, and her colleagues reported these results in the Journal of Clinical Sleep Medicine.
The team noted that cancer patients report sleep disturbances at a significantly higher rate than the general population. Between 23% and 44% of cancer patients experience insomnia symptoms even years after treatment.
With this in mind, the researchers studied 44 individuals who had completed cancer treatment and met criteria for clinically significant fatigue at screening.
The subjects had an average age of 53.6, and 75% percent were female. Roughly 55% (n=24) had been diagnosed with a hematologic malignancy.
The subjects were randomized to a bright white light intervention or a dim red light intervention. Subjects in both treatment arms were provided with a light box and instructed to use it every morning for 30 minutes for 4 weeks. Sleep was evaluated using wrist actigraphy and the Pittsburgh Sleep Quality Index.
At baseline, 52.6% of subjects in the dim light group and 60% in the bright light group exceeded the clinical cutoff for poor sleep efficiency (≤ 85%). The mean sleep efficiency was 81.8% and 82.8%, respectively.
During the study period, sleep efficiency improved significantly more among subjects exposed to the bright light than those exposed to the dim light (P=0.003).
The mean sleep efficiency was in the clinically normal range for subjects in the bright light group at the end of the intervention (86.06%) and 3 weeks after (85.77%).
However, the cutoff for poor sleep efficiency was not reached in the dim light group, either at the end of the intervention (mean=79.35%) or 3 weeks after (mean=80.88%).
Total sleep time tended to increase over the study period for subjects in the bright light group, but there was no significant difference in total sleep time between the bright light and dim light groups.
Likewise, there was no significant between-group difference in waking after sleep onset, although this outcome tended to decrease over the study period for subjects in the bright light group.
“Systematic light exposure using bright white light is a low-cost and easily disseminated intervention that offers a feasible and potentially effective alternative to improve sleep in cancer survivors,” Dr Wu said.
However, she and her colleagues noted that larger-scale studies are needed. ![]()
Results of a pilot study suggest that systematic bright light exposure can improve sleep in fatigued cancer survivors.
Subjects who were exposed to bright light every morning for 4 weeks had a significantly greater improvement in sleep efficiency than those who were exposed to dim light over the same period.
In fact, subjects in the bright light group were able to achieve clinically normal levels of sleep efficiency, and subjects in the dim light group were not.
Sleep efficiency is the percentage of time in bed that subjects spent sleeping.
Lisa M. Wu, PhD, of Northwestern University in Chicago, Illinois, and her colleagues reported these results in the Journal of Clinical Sleep Medicine.
The team noted that cancer patients report sleep disturbances at a significantly higher rate than the general population. Between 23% and 44% of cancer patients experience insomnia symptoms even years after treatment.
With this in mind, the researchers studied 44 individuals who had completed cancer treatment and met criteria for clinically significant fatigue at screening.
The subjects had an average age of 53.6, and 75% percent were female. Roughly 55% (n=24) had been diagnosed with a hematologic malignancy.
The subjects were randomized to a bright white light intervention or a dim red light intervention. Subjects in both treatment arms were provided with a light box and instructed to use it every morning for 30 minutes for 4 weeks. Sleep was evaluated using wrist actigraphy and the Pittsburgh Sleep Quality Index.
At baseline, 52.6% of subjects in the dim light group and 60% in the bright light group exceeded the clinical cutoff for poor sleep efficiency (≤ 85%). The mean sleep efficiency was 81.8% and 82.8%, respectively.
During the study period, sleep efficiency improved significantly more among subjects exposed to the bright light than those exposed to the dim light (P=0.003).
The mean sleep efficiency was in the clinically normal range for subjects in the bright light group at the end of the intervention (86.06%) and 3 weeks after (85.77%).
However, the cutoff for poor sleep efficiency was not reached in the dim light group, either at the end of the intervention (mean=79.35%) or 3 weeks after (mean=80.88%).
Total sleep time tended to increase over the study period for subjects in the bright light group, but there was no significant difference in total sleep time between the bright light and dim light groups.
Likewise, there was no significant between-group difference in waking after sleep onset, although this outcome tended to decrease over the study period for subjects in the bright light group.
“Systematic light exposure using bright white light is a low-cost and easily disseminated intervention that offers a feasible and potentially effective alternative to improve sleep in cancer survivors,” Dr Wu said.
However, she and her colleagues noted that larger-scale studies are needed. ![]()
CAR T-cell therapy on fast track in US, EU
The chimeric antigen receptor (CAR) T-cell therapy tisagenlecleucel (Kymriah, formerly CTL019) is getting fast-tracked in the United States (US) and European Union (EU).
The US Food and Drug Administration (FDA) has accepted for priority review the supplemental biologics license application (sBLA) for tisagenlecleucel for the treatment of adults with relapsed or refractory (R/R) diffuse large B-cell lymphoma (DLBCL) who are ineligible for, or relapse after, autologous hematopoietic stem cell transplant (auto-HSCT).
Meanwhile, the European Medicines Agency (EMA) has granted accelerated assessment to the marketing authorization application (MAA) for tisagenlecleucel for the treatment of children and young adults with R/R B-cell acute lymphoblastic leukemia (ALL) and for adults with R/R DLBCL who are ineligible for auto-HSCT.
If the sBLA and MAA are approved, tisagenlecleucel will be the first CAR T-cell therapy available for 2 distinct indications in non-Hodgkin lymphoma and B-cell ALL.
Tisagenlecleucel became the first CAR T-cell therapy to receive regulatory approval when it was approved by the FDA in August 2017 for use in patients up to 25 years of age who have B-cell precursor ALL that is refractory or in second or later relapse.
Supporting data
The regulatory applications for tisagenlecleucel in the US and EU are supported by data from the Novartis-sponsored global clinical trial program in children and young adults with R/R B-cell ALL and adults with R/R DLBCL.
Results from the phase 2 JULIET trial served as the basis of the sBLA and MAA for tisagenlecleucel in adults with R/R DLCBL. Data from this trial were presented at the 2017 ASH Annual Meeting in December.
Results from the phase 2 ELIANA study were submitted as part of the MAA for tisagenlecleucel in children and young adults with R/R B-cell ALL. Data from this trial were presented at the 2017 EHA Congress last June.
About priority review, accelerated assessment
The FDA grants priority review to applications for products that may provide significant improvements in the treatment, diagnosis, or prevention of serious conditions.
The FDA’s goal is to take action on a priority review application within 6 months of receiving it, rather than the standard 10 months.
The EMA grants accelerated assessment when a product is expected to be of major public health interest, particularly from the point of view of therapeutic innovation.
Accelerated assessment shortens the review period from 210 days to 150 days. ![]()
The chimeric antigen receptor (CAR) T-cell therapy tisagenlecleucel (Kymriah, formerly CTL019) is getting fast-tracked in the United States (US) and European Union (EU).
The US Food and Drug Administration (FDA) has accepted for priority review the supplemental biologics license application (sBLA) for tisagenlecleucel for the treatment of adults with relapsed or refractory (R/R) diffuse large B-cell lymphoma (DLBCL) who are ineligible for, or relapse after, autologous hematopoietic stem cell transplant (auto-HSCT).
Meanwhile, the European Medicines Agency (EMA) has granted accelerated assessment to the marketing authorization application (MAA) for tisagenlecleucel for the treatment of children and young adults with R/R B-cell acute lymphoblastic leukemia (ALL) and for adults with R/R DLBCL who are ineligible for auto-HSCT.
If the sBLA and MAA are approved, tisagenlecleucel will be the first CAR T-cell therapy available for 2 distinct indications in non-Hodgkin lymphoma and B-cell ALL.
Tisagenlecleucel became the first CAR T-cell therapy to receive regulatory approval when it was approved by the FDA in August 2017 for use in patients up to 25 years of age who have B-cell precursor ALL that is refractory or in second or later relapse.
Supporting data
The regulatory applications for tisagenlecleucel in the US and EU are supported by data from the Novartis-sponsored global clinical trial program in children and young adults with R/R B-cell ALL and adults with R/R DLBCL.
Results from the phase 2 JULIET trial served as the basis of the sBLA and MAA for tisagenlecleucel in adults with R/R DLCBL. Data from this trial were presented at the 2017 ASH Annual Meeting in December.
Results from the phase 2 ELIANA study were submitted as part of the MAA for tisagenlecleucel in children and young adults with R/R B-cell ALL. Data from this trial were presented at the 2017 EHA Congress last June.
About priority review, accelerated assessment
The FDA grants priority review to applications for products that may provide significant improvements in the treatment, diagnosis, or prevention of serious conditions.
The FDA’s goal is to take action on a priority review application within 6 months of receiving it, rather than the standard 10 months.
The EMA grants accelerated assessment when a product is expected to be of major public health interest, particularly from the point of view of therapeutic innovation.
Accelerated assessment shortens the review period from 210 days to 150 days. ![]()
The chimeric antigen receptor (CAR) T-cell therapy tisagenlecleucel (Kymriah, formerly CTL019) is getting fast-tracked in the United States (US) and European Union (EU).
The US Food and Drug Administration (FDA) has accepted for priority review the supplemental biologics license application (sBLA) for tisagenlecleucel for the treatment of adults with relapsed or refractory (R/R) diffuse large B-cell lymphoma (DLBCL) who are ineligible for, or relapse after, autologous hematopoietic stem cell transplant (auto-HSCT).
Meanwhile, the European Medicines Agency (EMA) has granted accelerated assessment to the marketing authorization application (MAA) for tisagenlecleucel for the treatment of children and young adults with R/R B-cell acute lymphoblastic leukemia (ALL) and for adults with R/R DLBCL who are ineligible for auto-HSCT.
If the sBLA and MAA are approved, tisagenlecleucel will be the first CAR T-cell therapy available for 2 distinct indications in non-Hodgkin lymphoma and B-cell ALL.
Tisagenlecleucel became the first CAR T-cell therapy to receive regulatory approval when it was approved by the FDA in August 2017 for use in patients up to 25 years of age who have B-cell precursor ALL that is refractory or in second or later relapse.
Supporting data
The regulatory applications for tisagenlecleucel in the US and EU are supported by data from the Novartis-sponsored global clinical trial program in children and young adults with R/R B-cell ALL and adults with R/R DLBCL.
Results from the phase 2 JULIET trial served as the basis of the sBLA and MAA for tisagenlecleucel in adults with R/R DLCBL. Data from this trial were presented at the 2017 ASH Annual Meeting in December.
Results from the phase 2 ELIANA study were submitted as part of the MAA for tisagenlecleucel in children and young adults with R/R B-cell ALL. Data from this trial were presented at the 2017 EHA Congress last June.
About priority review, accelerated assessment
The FDA grants priority review to applications for products that may provide significant improvements in the treatment, diagnosis, or prevention of serious conditions.
The FDA’s goal is to take action on a priority review application within 6 months of receiving it, rather than the standard 10 months.
The EMA grants accelerated assessment when a product is expected to be of major public health interest, particularly from the point of view of therapeutic innovation.
Accelerated assessment shortens the review period from 210 days to 150 days. ![]()
FDA approves injection treatment for low-risk APL
The Food and Drug Administration announced the approval of arsenic trioxide injection (Trisenox) in combination with tretinoin for the treatment of adults with newly diagnosed, low-risk acute promyelocytic leukemia (APL) characterized by t(15;17) translocation or PML/RAR-alpha gene expression.
The expanded indication was granted by the FDA on Jan. 12 after priority review. It is based on published studies and a review of Teva’s global safety database for arsenic trioxide.
A recent randomized, phase 3 trial compared tretinoin plus arsenic trioxide with tretinoin plus chemotherapy as first-line treatment for APL (J Clin Oncol. 2017 Feb 20;35[6]:605-12). It found that 100% of 127 patients in the tretinoin plus arsenic trioxide arm achieved complete remission, compared with 97% of 136 patients in the tretinoin plus chemotherapy arm. After a median follow-up of 40.6 months, the event-free survival at 50 months for patients in the tretinoin/arsenic trioxide arm was 97.3% vs. 80% for tretinoin/chemotherapy (P = .001).
The arsenic trioxide injection carries a boxed warning for differentiation syndrome and cardiac conduction abnormalities.
The Food and Drug Administration announced the approval of arsenic trioxide injection (Trisenox) in combination with tretinoin for the treatment of adults with newly diagnosed, low-risk acute promyelocytic leukemia (APL) characterized by t(15;17) translocation or PML/RAR-alpha gene expression.
The expanded indication was granted by the FDA on Jan. 12 after priority review. It is based on published studies and a review of Teva’s global safety database for arsenic trioxide.
A recent randomized, phase 3 trial compared tretinoin plus arsenic trioxide with tretinoin plus chemotherapy as first-line treatment for APL (J Clin Oncol. 2017 Feb 20;35[6]:605-12). It found that 100% of 127 patients in the tretinoin plus arsenic trioxide arm achieved complete remission, compared with 97% of 136 patients in the tretinoin plus chemotherapy arm. After a median follow-up of 40.6 months, the event-free survival at 50 months for patients in the tretinoin/arsenic trioxide arm was 97.3% vs. 80% for tretinoin/chemotherapy (P = .001).
The arsenic trioxide injection carries a boxed warning for differentiation syndrome and cardiac conduction abnormalities.
The Food and Drug Administration announced the approval of arsenic trioxide injection (Trisenox) in combination with tretinoin for the treatment of adults with newly diagnosed, low-risk acute promyelocytic leukemia (APL) characterized by t(15;17) translocation or PML/RAR-alpha gene expression.
The expanded indication was granted by the FDA on Jan. 12 after priority review. It is based on published studies and a review of Teva’s global safety database for arsenic trioxide.
A recent randomized, phase 3 trial compared tretinoin plus arsenic trioxide with tretinoin plus chemotherapy as first-line treatment for APL (J Clin Oncol. 2017 Feb 20;35[6]:605-12). It found that 100% of 127 patients in the tretinoin plus arsenic trioxide arm achieved complete remission, compared with 97% of 136 patients in the tretinoin plus chemotherapy arm. After a median follow-up of 40.6 months, the event-free survival at 50 months for patients in the tretinoin/arsenic trioxide arm was 97.3% vs. 80% for tretinoin/chemotherapy (P = .001).
The arsenic trioxide injection carries a boxed warning for differentiation syndrome and cardiac conduction abnormalities.
Drug’s label updated to include risk of allergic reactions
The US Food and Drug Administration (FDA) and Tesaro, Inc., have updated the prescribing information for Varubi® (rolapitant) injectable emulsion to include a new warning about the risk of allergic reactions.
Varubi injectable emulsion is a substance P/neurokinin receptor antagonist approved to prevent delayed nausea and vomiting associated with chemotherapy in adults.
Since Varubi injectable emulsion gained FDA approval, there have been reports of anaphylaxis, anaphylactic shock, and other serious hypersensitivity reactions to the drug, some of which required hospitalization.
Now, the labeling for Varubi injectable emulsion has been changed to include information about these events. The changes include modifications to the CONTRAINDICATIONS, WARNINGS and PRECAUTIONS, and ADVERSE REACTIONS sections of the label.
Since Varubi injectable emulsion was introduced to the US market in late November 2017, at least 7000 doses of the drug have been administered to patients receiving emetogenic chemotherapy in the US, according to Tesaro.
Anaphylaxis, anaphylactic shock, and other serious hypersensitivity reactions have occurred during or soon after the infusion of Varubi. Most reactions have occurred within the first few minutes of administration.
The FDA has advised that patients who are hypersensitive to any component of Varubi injectable emulsion (including soybean oil) do not receive the drug. And patients with known allergies to legumes or other related allergens should be monitored closely.
The FDA said healthcare professionals should be vigilant for signs of hypersensitivity or anaphylaxis in all patients receiving Varubi injectable emulsion, both during administration and afterward.
Symptoms of anaphylaxis can include wheezing, difficulty breathing, swelling of the face or throat, hives, flushing, itching, abdominal cramping, abdominal pain, vomiting, back pain, chest pain, hypotension, and shock.
If anaphylaxis or any other serious hypersensitivity/infusion reaction occurs, Varubi injectable emulsion should be stopped immediately and permanently. The patient should receive appropriate medical management, including epinephrine and/or antihistamines.
To ensure patients and healthcare professionals are aware of the label update to Varubi injectable emulsion, Tesaro has issued a Dear Healthcare Professional letter. In addition, the updated prescribing information has been posted on the Varubi website.
For any questions about the use of Varubi injectable emulsion or to report adverse events related to the drug, contact Tesaro’s medical information department at 1-844-4-TESARO (1-844-483-7276).
Adverse events related to Varubi should also be reported to the FDA’s MedWatch Safety Information and Adverse Event Reporting Program. ![]()
The US Food and Drug Administration (FDA) and Tesaro, Inc., have updated the prescribing information for Varubi® (rolapitant) injectable emulsion to include a new warning about the risk of allergic reactions.
Varubi injectable emulsion is a substance P/neurokinin receptor antagonist approved to prevent delayed nausea and vomiting associated with chemotherapy in adults.
Since Varubi injectable emulsion gained FDA approval, there have been reports of anaphylaxis, anaphylactic shock, and other serious hypersensitivity reactions to the drug, some of which required hospitalization.
Now, the labeling for Varubi injectable emulsion has been changed to include information about these events. The changes include modifications to the CONTRAINDICATIONS, WARNINGS and PRECAUTIONS, and ADVERSE REACTIONS sections of the label.
Since Varubi injectable emulsion was introduced to the US market in late November 2017, at least 7000 doses of the drug have been administered to patients receiving emetogenic chemotherapy in the US, according to Tesaro.
Anaphylaxis, anaphylactic shock, and other serious hypersensitivity reactions have occurred during or soon after the infusion of Varubi. Most reactions have occurred within the first few minutes of administration.
The FDA has advised that patients who are hypersensitive to any component of Varubi injectable emulsion (including soybean oil) do not receive the drug. And patients with known allergies to legumes or other related allergens should be monitored closely.
The FDA said healthcare professionals should be vigilant for signs of hypersensitivity or anaphylaxis in all patients receiving Varubi injectable emulsion, both during administration and afterward.
Symptoms of anaphylaxis can include wheezing, difficulty breathing, swelling of the face or throat, hives, flushing, itching, abdominal cramping, abdominal pain, vomiting, back pain, chest pain, hypotension, and shock.
If anaphylaxis or any other serious hypersensitivity/infusion reaction occurs, Varubi injectable emulsion should be stopped immediately and permanently. The patient should receive appropriate medical management, including epinephrine and/or antihistamines.
To ensure patients and healthcare professionals are aware of the label update to Varubi injectable emulsion, Tesaro has issued a Dear Healthcare Professional letter. In addition, the updated prescribing information has been posted on the Varubi website.
For any questions about the use of Varubi injectable emulsion or to report adverse events related to the drug, contact Tesaro’s medical information department at 1-844-4-TESARO (1-844-483-7276).
Adverse events related to Varubi should also be reported to the FDA’s MedWatch Safety Information and Adverse Event Reporting Program. ![]()
The US Food and Drug Administration (FDA) and Tesaro, Inc., have updated the prescribing information for Varubi® (rolapitant) injectable emulsion to include a new warning about the risk of allergic reactions.
Varubi injectable emulsion is a substance P/neurokinin receptor antagonist approved to prevent delayed nausea and vomiting associated with chemotherapy in adults.
Since Varubi injectable emulsion gained FDA approval, there have been reports of anaphylaxis, anaphylactic shock, and other serious hypersensitivity reactions to the drug, some of which required hospitalization.
Now, the labeling for Varubi injectable emulsion has been changed to include information about these events. The changes include modifications to the CONTRAINDICATIONS, WARNINGS and PRECAUTIONS, and ADVERSE REACTIONS sections of the label.
Since Varubi injectable emulsion was introduced to the US market in late November 2017, at least 7000 doses of the drug have been administered to patients receiving emetogenic chemotherapy in the US, according to Tesaro.
Anaphylaxis, anaphylactic shock, and other serious hypersensitivity reactions have occurred during or soon after the infusion of Varubi. Most reactions have occurred within the first few minutes of administration.
The FDA has advised that patients who are hypersensitive to any component of Varubi injectable emulsion (including soybean oil) do not receive the drug. And patients with known allergies to legumes or other related allergens should be monitored closely.
The FDA said healthcare professionals should be vigilant for signs of hypersensitivity or anaphylaxis in all patients receiving Varubi injectable emulsion, both during administration and afterward.
Symptoms of anaphylaxis can include wheezing, difficulty breathing, swelling of the face or throat, hives, flushing, itching, abdominal cramping, abdominal pain, vomiting, back pain, chest pain, hypotension, and shock.
If anaphylaxis or any other serious hypersensitivity/infusion reaction occurs, Varubi injectable emulsion should be stopped immediately and permanently. The patient should receive appropriate medical management, including epinephrine and/or antihistamines.
To ensure patients and healthcare professionals are aware of the label update to Varubi injectable emulsion, Tesaro has issued a Dear Healthcare Professional letter. In addition, the updated prescribing information has been posted on the Varubi website.
For any questions about the use of Varubi injectable emulsion or to report adverse events related to the drug, contact Tesaro’s medical information department at 1-844-4-TESARO (1-844-483-7276).
Adverse events related to Varubi should also be reported to the FDA’s MedWatch Safety Information and Adverse Event Reporting Program. ![]()
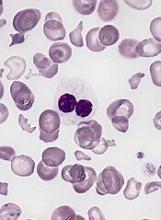
Children with pathogenic TP53 variants face increased risk of hard-to-treat ALL
Pathogenic loss-of-function germline variants in the TP53 gene predispose children to acute lymphoblastic leukemia (ALL), and, later, to solid tumors that may be related to cancer therapy, according to results from a genetic sequencing study.
Researchers at St. Jude Children’s Hospital in Memphis have identified 49 unique variants of the gene among 3,801 children with newly diagnosed B-cell ALL; 22 variants were deemed pathogenic. Children with these variants were at a “dramatically higher risk” of secondary cancers, which occurred in 25% within 5 years of ALL treatment, compared with 0.7% among children without the pathogenic genetic signal, according to Maoxiang Qian, PhD, and colleagues. The report was published in the Journal of Clinical Oncology.
The increased risk of secondary cancers is probably related to a common characteristic of the pathogenic variants: the ablation of the p53-mediated DNA damage response. This increases the risk of the genotoxic therapy given during ALL treatment, according to the researchers.
“In fact, of the five patients with TP53 pathogenic variants who also had second cancers, two received irradiation therapy, including total body irradiation, and both subsequently developed solid tumors,” the researchers wrote. “The exact lifelong risk of second cancer in these patients is difficult to ascertain as many patients might have succumbed to relapsed ALL before they had the chance to develop second cancers.”
The research team conducted targeted sequencing of TP53 coding regions in 3,801 children who were enrolled in two trials sponsored by the Children’s Oncology Group (AALL0232 and P9900). They compared the results to TP53 mutations seen in almost 61,000 children enrolled in the Exome Aggregation Consortium (ExAC) cohort.
The researchers identified nine exonic nonsilent TP53 variants in the ALL cohort, all of which had an allele frequency of less than 0.5%. Of the variants, 22 were deemed pathogenic: Twelve showed a complete loss of transcriptional activity, three showed a partial loss of p53 function, and seven showed loss of the critical core DNA-binding domain in p53. The rest of the variants were deemed of unknown significance (VUS).
Pathogenic variants occurred in 26 children in the ALL cohort – significantly more often than in the control cohort (0.7% vs. 0.1%; odds ratio, 5.2). The VUS risk was not significantly elevated compared to controls, however.
Children with the pathogenic variants were significantly older at ALL diagnosis (15.5 vs. 6.6 years) and had significantly lower leukocyte count. Of the 26 with a pathogenic variant, 17 (65.4%) showed hypodiploidy in ALL blasts.
Pathogenic variants negatively affected ALL treatment outcomes, quadrupling the risk of lower event-free survival and lower overall survival (hazard ratio, 4.2 and 3.9, respectively) in both ALL cohorts.
Of the children with pathogenic variants, 14 experienced a pathological clinical event, including five ALL relapses and five second cancers, each accounting for 36% of all events.
“This pattern of events was dramatically different from that in patients with wild-type TP53 or VUS, for whom ALL relapse accounted for 75% of all events, with only 4% as second cancers,” the researchers wrote. “In fact, within hypodiploid ALL patients who experienced an event, the frequency of second cancer was significantly higher in those with TP53 pathogenic variants than in those without [50% vs. 5%], which additionally suggests that germline TP53 variation, instead of hypodiploid ALL, was the underlying cause of second cancers in these patients.”
The study was supported by grants from the National Institutes of Health and the American Lebanese Syrian Associated Charities. Dr. Qian reported having no financial disclosures. Other researchers reported funding from various pharmaceutical companies.
SOURCE: Qian et al. JCO 2018 Jan 4. doi: 10.1200/JCO.2017.75.5215
Pathogenic loss-of-function germline variants in the TP53 gene predispose children to acute lymphoblastic leukemia (ALL), and, later, to solid tumors that may be related to cancer therapy, according to results from a genetic sequencing study.
Researchers at St. Jude Children’s Hospital in Memphis have identified 49 unique variants of the gene among 3,801 children with newly diagnosed B-cell ALL; 22 variants were deemed pathogenic. Children with these variants were at a “dramatically higher risk” of secondary cancers, which occurred in 25% within 5 years of ALL treatment, compared with 0.7% among children without the pathogenic genetic signal, according to Maoxiang Qian, PhD, and colleagues. The report was published in the Journal of Clinical Oncology.
The increased risk of secondary cancers is probably related to a common characteristic of the pathogenic variants: the ablation of the p53-mediated DNA damage response. This increases the risk of the genotoxic therapy given during ALL treatment, according to the researchers.
“In fact, of the five patients with TP53 pathogenic variants who also had second cancers, two received irradiation therapy, including total body irradiation, and both subsequently developed solid tumors,” the researchers wrote. “The exact lifelong risk of second cancer in these patients is difficult to ascertain as many patients might have succumbed to relapsed ALL before they had the chance to develop second cancers.”
The research team conducted targeted sequencing of TP53 coding regions in 3,801 children who were enrolled in two trials sponsored by the Children’s Oncology Group (AALL0232 and P9900). They compared the results to TP53 mutations seen in almost 61,000 children enrolled in the Exome Aggregation Consortium (ExAC) cohort.
The researchers identified nine exonic nonsilent TP53 variants in the ALL cohort, all of which had an allele frequency of less than 0.5%. Of the variants, 22 were deemed pathogenic: Twelve showed a complete loss of transcriptional activity, three showed a partial loss of p53 function, and seven showed loss of the critical core DNA-binding domain in p53. The rest of the variants were deemed of unknown significance (VUS).
Pathogenic variants occurred in 26 children in the ALL cohort – significantly more often than in the control cohort (0.7% vs. 0.1%; odds ratio, 5.2). The VUS risk was not significantly elevated compared to controls, however.
Children with the pathogenic variants were significantly older at ALL diagnosis (15.5 vs. 6.6 years) and had significantly lower leukocyte count. Of the 26 with a pathogenic variant, 17 (65.4%) showed hypodiploidy in ALL blasts.
Pathogenic variants negatively affected ALL treatment outcomes, quadrupling the risk of lower event-free survival and lower overall survival (hazard ratio, 4.2 and 3.9, respectively) in both ALL cohorts.
Of the children with pathogenic variants, 14 experienced a pathological clinical event, including five ALL relapses and five second cancers, each accounting for 36% of all events.
“This pattern of events was dramatically different from that in patients with wild-type TP53 or VUS, for whom ALL relapse accounted for 75% of all events, with only 4% as second cancers,” the researchers wrote. “In fact, within hypodiploid ALL patients who experienced an event, the frequency of second cancer was significantly higher in those with TP53 pathogenic variants than in those without [50% vs. 5%], which additionally suggests that germline TP53 variation, instead of hypodiploid ALL, was the underlying cause of second cancers in these patients.”
The study was supported by grants from the National Institutes of Health and the American Lebanese Syrian Associated Charities. Dr. Qian reported having no financial disclosures. Other researchers reported funding from various pharmaceutical companies.
SOURCE: Qian et al. JCO 2018 Jan 4. doi: 10.1200/JCO.2017.75.5215
Pathogenic loss-of-function germline variants in the TP53 gene predispose children to acute lymphoblastic leukemia (ALL), and, later, to solid tumors that may be related to cancer therapy, according to results from a genetic sequencing study.
Researchers at St. Jude Children’s Hospital in Memphis have identified 49 unique variants of the gene among 3,801 children with newly diagnosed B-cell ALL; 22 variants were deemed pathogenic. Children with these variants were at a “dramatically higher risk” of secondary cancers, which occurred in 25% within 5 years of ALL treatment, compared with 0.7% among children without the pathogenic genetic signal, according to Maoxiang Qian, PhD, and colleagues. The report was published in the Journal of Clinical Oncology.
The increased risk of secondary cancers is probably related to a common characteristic of the pathogenic variants: the ablation of the p53-mediated DNA damage response. This increases the risk of the genotoxic therapy given during ALL treatment, according to the researchers.
“In fact, of the five patients with TP53 pathogenic variants who also had second cancers, two received irradiation therapy, including total body irradiation, and both subsequently developed solid tumors,” the researchers wrote. “The exact lifelong risk of second cancer in these patients is difficult to ascertain as many patients might have succumbed to relapsed ALL before they had the chance to develop second cancers.”
The research team conducted targeted sequencing of TP53 coding regions in 3,801 children who were enrolled in two trials sponsored by the Children’s Oncology Group (AALL0232 and P9900). They compared the results to TP53 mutations seen in almost 61,000 children enrolled in the Exome Aggregation Consortium (ExAC) cohort.
The researchers identified nine exonic nonsilent TP53 variants in the ALL cohort, all of which had an allele frequency of less than 0.5%. Of the variants, 22 were deemed pathogenic: Twelve showed a complete loss of transcriptional activity, three showed a partial loss of p53 function, and seven showed loss of the critical core DNA-binding domain in p53. The rest of the variants were deemed of unknown significance (VUS).
Pathogenic variants occurred in 26 children in the ALL cohort – significantly more often than in the control cohort (0.7% vs. 0.1%; odds ratio, 5.2). The VUS risk was not significantly elevated compared to controls, however.
Children with the pathogenic variants were significantly older at ALL diagnosis (15.5 vs. 6.6 years) and had significantly lower leukocyte count. Of the 26 with a pathogenic variant, 17 (65.4%) showed hypodiploidy in ALL blasts.
Pathogenic variants negatively affected ALL treatment outcomes, quadrupling the risk of lower event-free survival and lower overall survival (hazard ratio, 4.2 and 3.9, respectively) in both ALL cohorts.
Of the children with pathogenic variants, 14 experienced a pathological clinical event, including five ALL relapses and five second cancers, each accounting for 36% of all events.
“This pattern of events was dramatically different from that in patients with wild-type TP53 or VUS, for whom ALL relapse accounted for 75% of all events, with only 4% as second cancers,” the researchers wrote. “In fact, within hypodiploid ALL patients who experienced an event, the frequency of second cancer was significantly higher in those with TP53 pathogenic variants than in those without [50% vs. 5%], which additionally suggests that germline TP53 variation, instead of hypodiploid ALL, was the underlying cause of second cancers in these patients.”
The study was supported by grants from the National Institutes of Health and the American Lebanese Syrian Associated Charities. Dr. Qian reported having no financial disclosures. Other researchers reported funding from various pharmaceutical companies.
SOURCE: Qian et al. JCO 2018 Jan 4. doi: 10.1200/JCO.2017.75.5215
FROM THE JOURNAL OF CLINICAL ONCOLOGY
Key clinical point:
Major finding: TP53 variants quadrupled the risk of lower event-free survival and lower overall survival (HR 4.2 and 3.9, respectively).
Study details: The genetic sequencing study comprised 3,801 children with newly diagnosed B-cell ALL.
Disclosures: The study was supported by grants from the National Institutes of Health and the American Lebanese Syrian Associated Charities. Dr. Qian reported having no financial disclosures. Other researchers reported funding from various pharmaceutical companies.
Source: Qian et al. JCO 2018 Jan 4. doi: 10.1200/JCO.2017.75.5215.
FDA expands approval for arsenic trioxide
The US Food and Drug Administration (FDA) has expanded the approved use of arsenic trioxide (TRISENOX®) injection.
The drug is now approved for use in combination with all-trans retinoic acid (ATRA) for the treatment of adults with newly diagnosed, low-risk acute promyelocytic leukemia (APL) whose disease is characterized by the presence of the t(15;17) translocation or PML/RAR-alpha gene expression.
Arsenic trioxide is also FDA-approved for induction of remission and consolidation in patients with APL who are refractory to, or have relapsed after, retinoid and anthracycline chemotherapy and whose APL is characterized by the presence of the t(15;17) translocation or PML/RAR-alpha gene expression.
“This label expansion represents an important benefit, as TRISENOX is now an FDA-approved, first-line treatment option for patients with acute promyelocytic leukemia,” said Paul Rittman, senior vice-president and general manager of Teva Oncology.
The expanded approval for arsenic trioxide was based on a priority review by the FDA of data from the scientific literature and a review of Teva’s global safety database for arsenic trioxide.
Data from this database were presented at the 2016 ASH Annual Meeting.
According to the presentation, the most common adverse events observed in patients receiving arsenic trioxide were QT prolongation, decrease in white blood cells, APL differentiation syndrome, febrile neutropenia, neutropenia, pyrexia, alanine aminotransferase increase, neutrophil decrease, platelet count decrease, aspartate aminotransferase increase, leukocytosis, and pancytopenia.
The combination of arsenic trioxide and ATRA was evaluated in a phase 3 trial of patients with APL. Results from this trial were published in the Journal of Clinical Oncology in February 2017.
The study included 276 adults (ages 18 to 71) with newly diagnosed, low- or intermediate-risk APL. Patients were randomized to receive ATRA plus arsenic trioxide or ATRA plus chemotherapy.
A total of 263 patients were evaluable for response to induction. One hundred percent of patients in the arsenic trioxide arm (127/127) achieved a complete response (CR), as did 97% (132/136) of patients in the chemotherapy arm (P=0.12).
After a median follow-up of 40.6 months, the event-free survival was 97.3% in the arsenic trioxide arm and 80% in the chemotherapy arm (P<0.001). The cumulative incidence of relapse was 1.9% and 13.9%, respectively (P=0.0013).
At 50 months, the overall survival was 99.2% in the arsenic trioxide arm and 92.6% in the chemotherapy arm (P=0.0073).
After induction, there were 2 relapses and 1 death in CR in the arsenic trioxide arm.
In the chemotherapy arm, there were 2 instances of molecular resistance after third consolidation, 15 relapses, 5 deaths in CR, and 2 patients who developed a therapy-related myeloid neoplasm. ![]()
The US Food and Drug Administration (FDA) has expanded the approved use of arsenic trioxide (TRISENOX®) injection.
The drug is now approved for use in combination with all-trans retinoic acid (ATRA) for the treatment of adults with newly diagnosed, low-risk acute promyelocytic leukemia (APL) whose disease is characterized by the presence of the t(15;17) translocation or PML/RAR-alpha gene expression.
Arsenic trioxide is also FDA-approved for induction of remission and consolidation in patients with APL who are refractory to, or have relapsed after, retinoid and anthracycline chemotherapy and whose APL is characterized by the presence of the t(15;17) translocation or PML/RAR-alpha gene expression.
“This label expansion represents an important benefit, as TRISENOX is now an FDA-approved, first-line treatment option for patients with acute promyelocytic leukemia,” said Paul Rittman, senior vice-president and general manager of Teva Oncology.
The expanded approval for arsenic trioxide was based on a priority review by the FDA of data from the scientific literature and a review of Teva’s global safety database for arsenic trioxide.
Data from this database were presented at the 2016 ASH Annual Meeting.
According to the presentation, the most common adverse events observed in patients receiving arsenic trioxide were QT prolongation, decrease in white blood cells, APL differentiation syndrome, febrile neutropenia, neutropenia, pyrexia, alanine aminotransferase increase, neutrophil decrease, platelet count decrease, aspartate aminotransferase increase, leukocytosis, and pancytopenia.
The combination of arsenic trioxide and ATRA was evaluated in a phase 3 trial of patients with APL. Results from this trial were published in the Journal of Clinical Oncology in February 2017.
The study included 276 adults (ages 18 to 71) with newly diagnosed, low- or intermediate-risk APL. Patients were randomized to receive ATRA plus arsenic trioxide or ATRA plus chemotherapy.
A total of 263 patients were evaluable for response to induction. One hundred percent of patients in the arsenic trioxide arm (127/127) achieved a complete response (CR), as did 97% (132/136) of patients in the chemotherapy arm (P=0.12).
After a median follow-up of 40.6 months, the event-free survival was 97.3% in the arsenic trioxide arm and 80% in the chemotherapy arm (P<0.001). The cumulative incidence of relapse was 1.9% and 13.9%, respectively (P=0.0013).
At 50 months, the overall survival was 99.2% in the arsenic trioxide arm and 92.6% in the chemotherapy arm (P=0.0073).
After induction, there were 2 relapses and 1 death in CR in the arsenic trioxide arm.
In the chemotherapy arm, there were 2 instances of molecular resistance after third consolidation, 15 relapses, 5 deaths in CR, and 2 patients who developed a therapy-related myeloid neoplasm. ![]()
The US Food and Drug Administration (FDA) has expanded the approved use of arsenic trioxide (TRISENOX®) injection.
The drug is now approved for use in combination with all-trans retinoic acid (ATRA) for the treatment of adults with newly diagnosed, low-risk acute promyelocytic leukemia (APL) whose disease is characterized by the presence of the t(15;17) translocation or PML/RAR-alpha gene expression.
Arsenic trioxide is also FDA-approved for induction of remission and consolidation in patients with APL who are refractory to, or have relapsed after, retinoid and anthracycline chemotherapy and whose APL is characterized by the presence of the t(15;17) translocation or PML/RAR-alpha gene expression.
“This label expansion represents an important benefit, as TRISENOX is now an FDA-approved, first-line treatment option for patients with acute promyelocytic leukemia,” said Paul Rittman, senior vice-president and general manager of Teva Oncology.
The expanded approval for arsenic trioxide was based on a priority review by the FDA of data from the scientific literature and a review of Teva’s global safety database for arsenic trioxide.
Data from this database were presented at the 2016 ASH Annual Meeting.
According to the presentation, the most common adverse events observed in patients receiving arsenic trioxide were QT prolongation, decrease in white blood cells, APL differentiation syndrome, febrile neutropenia, neutropenia, pyrexia, alanine aminotransferase increase, neutrophil decrease, platelet count decrease, aspartate aminotransferase increase, leukocytosis, and pancytopenia.
The combination of arsenic trioxide and ATRA was evaluated in a phase 3 trial of patients with APL. Results from this trial were published in the Journal of Clinical Oncology in February 2017.
The study included 276 adults (ages 18 to 71) with newly diagnosed, low- or intermediate-risk APL. Patients were randomized to receive ATRA plus arsenic trioxide or ATRA plus chemotherapy.
A total of 263 patients were evaluable for response to induction. One hundred percent of patients in the arsenic trioxide arm (127/127) achieved a complete response (CR), as did 97% (132/136) of patients in the chemotherapy arm (P=0.12).
After a median follow-up of 40.6 months, the event-free survival was 97.3% in the arsenic trioxide arm and 80% in the chemotherapy arm (P<0.001). The cumulative incidence of relapse was 1.9% and 13.9%, respectively (P=0.0013).
At 50 months, the overall survival was 99.2% in the arsenic trioxide arm and 92.6% in the chemotherapy arm (P=0.0073).
After induction, there were 2 relapses and 1 death in CR in the arsenic trioxide arm.
In the chemotherapy arm, there were 2 instances of molecular resistance after third consolidation, 15 relapses, 5 deaths in CR, and 2 patients who developed a therapy-related myeloid neoplasm. ![]()
EMA recommends orphan designation for pracinostat
The European Medicines Agency (EMA) has recommended that pracinostat receive orphan drug designation.
Pracinostat is an oral histone deacetylase inhibitor currently under investigation in a phase 3 study in combination with azacitidine for the treatment of acute myeloid leukemia (AML) in adult patients unfit to receive induction chemotherapy.
Orphan designation provides regulatory and financial incentives for companies to develop and market therapies that treat life-threatening or chronically debilitating conditions affecting no more than 5 in 10,000 people in the European Union, and where no satisfactory treatment is available.
Orphan designation provides incentives for companies seeking protocol assistance from the EMA during the product development phase and direct access to the centralized authorization procedure. The designation also provides a 10-year period of marketing exclusivity if a therapy receives regulatory approval.
The EMA’s Committee for Orphan Medicinal Products adopts an opinion on the granting of orphan drug designation, and that opinion is submitted to the European Commission for a final decision.
Phase 2 study
The EMA’s recommendation that pracinostat receive orphan drug designation is based on results of a phase 2 study, which were presented at the 2016 ASH Annual Meeting.
The study included 50 patients who had a median age of 75 (range, 66-84). Sixty-six percent of patients had de novo AML, and 34% had secondary AML.
The patients received pracinostat at 60 mg orally on days 1, 3, and 5 of each week for 21 days of each 28-day cycle. They received azacitidine at 75 mg/m2 subcutaneously or intravenously on days 1-7 or days 1-5 and 8-9 (per site preference) of each 28-day cycle.
As of October 15, 2016, 90% of patients had discontinued treatment, 42% due to progressive disease, 28% due to adverse events (AEs), 14% due to patient decision, and 6% due to investigator decision.
Fifty-two percent of patients (n=26) achieved the primary endpoint of complete response (CR) plus CR with incomplete count recovery (CRi) plus morphologic leukemia-free state (MLFS).
Forty-two percent of patients had a CR, 4% had a CRi, and 6% achieved MLFS. The median duration of CR/CRi/MLFS was 13.2 months. The median duration of CR/CRi was 17.2 months.
The median overall survival was 19.1 months. The 1-year survival rate was 62%, and the 2-year survival rate was 41%.
The most common treatment-emergent AEs were nausea (78%), constipation (70%), fatigue (62%), decreased appetite (56%), diarrhea (50%), vomiting (40%), cough (36%), dyspnea (34%), hypokalemia (34%), peripheral edema (34%), pyrexia (34%), dizziness (32%), back pain (28%), insomnia (28%), febrile neutropenia (48%), thrombocytopenia (46%), anemia (38%), and neutropenia (38%).
Treatment-emergent AEs led to discontinuation in 14 patients. Three of these patients developed sepsis that proved fatal.
The other AEs leading to discontinuation included grade 3 acute axonal neuropathy, grade 3 parainfluenza, grade 3 prolonged QTc/atrial fibrillation, grade 1 acute kidney injury, grade 3 diverticulitis, grade 3 supraglottic ulcer, grade 2 upper respiratory infection, grade 3 fatigue (n=2), and grades 1 and 3 intermittent fatigue (n=2). ![]()
The European Medicines Agency (EMA) has recommended that pracinostat receive orphan drug designation.
Pracinostat is an oral histone deacetylase inhibitor currently under investigation in a phase 3 study in combination with azacitidine for the treatment of acute myeloid leukemia (AML) in adult patients unfit to receive induction chemotherapy.
Orphan designation provides regulatory and financial incentives for companies to develop and market therapies that treat life-threatening or chronically debilitating conditions affecting no more than 5 in 10,000 people in the European Union, and where no satisfactory treatment is available.
Orphan designation provides incentives for companies seeking protocol assistance from the EMA during the product development phase and direct access to the centralized authorization procedure. The designation also provides a 10-year period of marketing exclusivity if a therapy receives regulatory approval.
The EMA’s Committee for Orphan Medicinal Products adopts an opinion on the granting of orphan drug designation, and that opinion is submitted to the European Commission for a final decision.
Phase 2 study
The EMA’s recommendation that pracinostat receive orphan drug designation is based on results of a phase 2 study, which were presented at the 2016 ASH Annual Meeting.
The study included 50 patients who had a median age of 75 (range, 66-84). Sixty-six percent of patients had de novo AML, and 34% had secondary AML.
The patients received pracinostat at 60 mg orally on days 1, 3, and 5 of each week for 21 days of each 28-day cycle. They received azacitidine at 75 mg/m2 subcutaneously or intravenously on days 1-7 or days 1-5 and 8-9 (per site preference) of each 28-day cycle.
As of October 15, 2016, 90% of patients had discontinued treatment, 42% due to progressive disease, 28% due to adverse events (AEs), 14% due to patient decision, and 6% due to investigator decision.
Fifty-two percent of patients (n=26) achieved the primary endpoint of complete response (CR) plus CR with incomplete count recovery (CRi) plus morphologic leukemia-free state (MLFS).
Forty-two percent of patients had a CR, 4% had a CRi, and 6% achieved MLFS. The median duration of CR/CRi/MLFS was 13.2 months. The median duration of CR/CRi was 17.2 months.
The median overall survival was 19.1 months. The 1-year survival rate was 62%, and the 2-year survival rate was 41%.
The most common treatment-emergent AEs were nausea (78%), constipation (70%), fatigue (62%), decreased appetite (56%), diarrhea (50%), vomiting (40%), cough (36%), dyspnea (34%), hypokalemia (34%), peripheral edema (34%), pyrexia (34%), dizziness (32%), back pain (28%), insomnia (28%), febrile neutropenia (48%), thrombocytopenia (46%), anemia (38%), and neutropenia (38%).
Treatment-emergent AEs led to discontinuation in 14 patients. Three of these patients developed sepsis that proved fatal.
The other AEs leading to discontinuation included grade 3 acute axonal neuropathy, grade 3 parainfluenza, grade 3 prolonged QTc/atrial fibrillation, grade 1 acute kidney injury, grade 3 diverticulitis, grade 3 supraglottic ulcer, grade 2 upper respiratory infection, grade 3 fatigue (n=2), and grades 1 and 3 intermittent fatigue (n=2). ![]()
The European Medicines Agency (EMA) has recommended that pracinostat receive orphan drug designation.
Pracinostat is an oral histone deacetylase inhibitor currently under investigation in a phase 3 study in combination with azacitidine for the treatment of acute myeloid leukemia (AML) in adult patients unfit to receive induction chemotherapy.
Orphan designation provides regulatory and financial incentives for companies to develop and market therapies that treat life-threatening or chronically debilitating conditions affecting no more than 5 in 10,000 people in the European Union, and where no satisfactory treatment is available.
Orphan designation provides incentives for companies seeking protocol assistance from the EMA during the product development phase and direct access to the centralized authorization procedure. The designation also provides a 10-year period of marketing exclusivity if a therapy receives regulatory approval.
The EMA’s Committee for Orphan Medicinal Products adopts an opinion on the granting of orphan drug designation, and that opinion is submitted to the European Commission for a final decision.
Phase 2 study
The EMA’s recommendation that pracinostat receive orphan drug designation is based on results of a phase 2 study, which were presented at the 2016 ASH Annual Meeting.
The study included 50 patients who had a median age of 75 (range, 66-84). Sixty-six percent of patients had de novo AML, and 34% had secondary AML.
The patients received pracinostat at 60 mg orally on days 1, 3, and 5 of each week for 21 days of each 28-day cycle. They received azacitidine at 75 mg/m2 subcutaneously or intravenously on days 1-7 or days 1-5 and 8-9 (per site preference) of each 28-day cycle.
As of October 15, 2016, 90% of patients had discontinued treatment, 42% due to progressive disease, 28% due to adverse events (AEs), 14% due to patient decision, and 6% due to investigator decision.
Fifty-two percent of patients (n=26) achieved the primary endpoint of complete response (CR) plus CR with incomplete count recovery (CRi) plus morphologic leukemia-free state (MLFS).
Forty-two percent of patients had a CR, 4% had a CRi, and 6% achieved MLFS. The median duration of CR/CRi/MLFS was 13.2 months. The median duration of CR/CRi was 17.2 months.
The median overall survival was 19.1 months. The 1-year survival rate was 62%, and the 2-year survival rate was 41%.
The most common treatment-emergent AEs were nausea (78%), constipation (70%), fatigue (62%), decreased appetite (56%), diarrhea (50%), vomiting (40%), cough (36%), dyspnea (34%), hypokalemia (34%), peripheral edema (34%), pyrexia (34%), dizziness (32%), back pain (28%), insomnia (28%), febrile neutropenia (48%), thrombocytopenia (46%), anemia (38%), and neutropenia (38%).
Treatment-emergent AEs led to discontinuation in 14 patients. Three of these patients developed sepsis that proved fatal.
The other AEs leading to discontinuation included grade 3 acute axonal neuropathy, grade 3 parainfluenza, grade 3 prolonged QTc/atrial fibrillation, grade 1 acute kidney injury, grade 3 diverticulitis, grade 3 supraglottic ulcer, grade 2 upper respiratory infection, grade 3 fatigue (n=2), and grades 1 and 3 intermittent fatigue (n=2). ![]()