User login
Psychiatric issues common among hepatitis C inpatients
Adult inpatients with hepatitis C are much more likely to have mental health comorbidities, compared with those who do not have hepatitis C, according to the Agency for Healthcare Research and Quality.
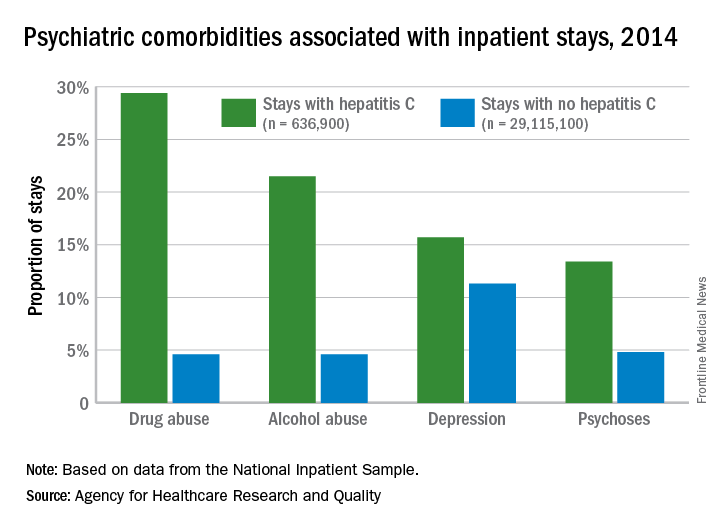
All four comorbidities skewed younger, and the oldest patients (73 years and older) with hepatitis C presented with each condition at about the same rate as the non–hepatitis C population. The proportions of hepatitis C–related inpatient stays with alcohol abuse by age, for example, were 20.5% for 18-51 years, 23.3% for those aged 52-72, and 5.8% for the 73-and-older group, according to data from the National Inpatient Sample, which includes more than 95% of all discharges from community (short-term, nonfederal, nonrehabilitation) hospitals in the United States.
Adult inpatients with hepatitis C are much more likely to have mental health comorbidities, compared with those who do not have hepatitis C, according to the Agency for Healthcare Research and Quality.

All four comorbidities skewed younger, and the oldest patients (73 years and older) with hepatitis C presented with each condition at about the same rate as the non–hepatitis C population. The proportions of hepatitis C–related inpatient stays with alcohol abuse by age, for example, were 20.5% for 18-51 years, 23.3% for those aged 52-72, and 5.8% for the 73-and-older group, according to data from the National Inpatient Sample, which includes more than 95% of all discharges from community (short-term, nonfederal, nonrehabilitation) hospitals in the United States.
Adult inpatients with hepatitis C are much more likely to have mental health comorbidities, compared with those who do not have hepatitis C, according to the Agency for Healthcare Research and Quality.

All four comorbidities skewed younger, and the oldest patients (73 years and older) with hepatitis C presented with each condition at about the same rate as the non–hepatitis C population. The proportions of hepatitis C–related inpatient stays with alcohol abuse by age, for example, were 20.5% for 18-51 years, 23.3% for those aged 52-72, and 5.8% for the 73-and-older group, according to data from the National Inpatient Sample, which includes more than 95% of all discharges from community (short-term, nonfederal, nonrehabilitation) hospitals in the United States.
Baby boomers are the hepatitis C generation
Increases in hepatitis C–related inpatient stays for baby boomers from 2005 to 2014 far outpaced those of older adults, while younger adults saw their admissions drop over that period, according to the Agency for Healthcare Research and Quality.
For the baby boomers (adults aged 52-72 years), the rate of inpatient stays involving hepatitis C with or without hepatitis B, HIV, or alcoholic liver disease rose from 300.7 per 100,000 population in 2005 to 503.1 per 100,000 in 2014 – an increase of over 67%. For patients aged 73 years and older, that rate went from 104.4 in 2005 to 117.1 in 2014, which translates to a 12% increase, and for patients aged 18-51 years, it dropped 15%, from 182.5 to 155.4, the AHRQ said in a statistical brief.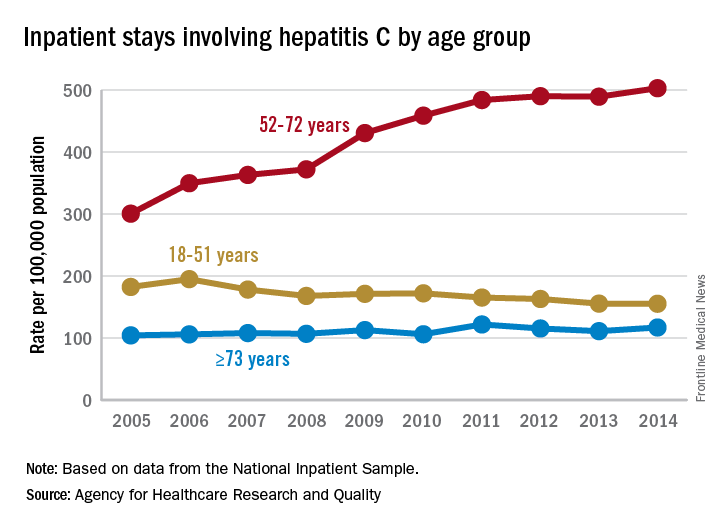
Along with the increased hospitalizations, “acute hepatitis C cases nearly tripled from 2010 through 2015,” the report noted, which was “likely the result of increasing injection drug use due to the growing opioid epidemic.”
AGA offers tools to help you become more efficient, understand quality standards, and improve the process of care for your hepatitis C patients.
Increases in hepatitis C–related inpatient stays for baby boomers from 2005 to 2014 far outpaced those of older adults, while younger adults saw their admissions drop over that period, according to the Agency for Healthcare Research and Quality.
For the baby boomers (adults aged 52-72 years), the rate of inpatient stays involving hepatitis C with or without hepatitis B, HIV, or alcoholic liver disease rose from 300.7 per 100,000 population in 2005 to 503.1 per 100,000 in 2014 – an increase of over 67%. For patients aged 73 years and older, that rate went from 104.4 in 2005 to 117.1 in 2014, which translates to a 12% increase, and for patients aged 18-51 years, it dropped 15%, from 182.5 to 155.4, the AHRQ said in a statistical brief.
Along with the increased hospitalizations, “acute hepatitis C cases nearly tripled from 2010 through 2015,” the report noted, which was “likely the result of increasing injection drug use due to the growing opioid epidemic.”
AGA offers tools to help you become more efficient, understand quality standards, and improve the process of care for your hepatitis C patients.
Increases in hepatitis C–related inpatient stays for baby boomers from 2005 to 2014 far outpaced those of older adults, while younger adults saw their admissions drop over that period, according to the Agency for Healthcare Research and Quality.
For the baby boomers (adults aged 52-72 years), the rate of inpatient stays involving hepatitis C with or without hepatitis B, HIV, or alcoholic liver disease rose from 300.7 per 100,000 population in 2005 to 503.1 per 100,000 in 2014 – an increase of over 67%. For patients aged 73 years and older, that rate went from 104.4 in 2005 to 117.1 in 2014, which translates to a 12% increase, and for patients aged 18-51 years, it dropped 15%, from 182.5 to 155.4, the AHRQ said in a statistical brief.
Along with the increased hospitalizations, “acute hepatitis C cases nearly tripled from 2010 through 2015,” the report noted, which was “likely the result of increasing injection drug use due to the growing opioid epidemic.”
AGA offers tools to help you become more efficient, understand quality standards, and improve the process of care for your hepatitis C patients.
FDA adds boxed warning to obeticholic acid label
The Food and Drug Administration is requiring a boxed warning on the label for obeticholic acid (Ocaliva) to highlight the correct weekly dosing regimen after incorrect daily dosing caused severe liver injury in patients with moderate to severe primary biliary cholangitis (PBC).
“FDA is adding a new Boxed Warning, FDA’s most prominent warning, to highlight this information in the prescribing information of the drug label,” FDA officials said in a statement Feb. 1. “To ensure correct dosing and reduce the risk of liver problems, FDA is clarifying the current recommendations for screening, dosing, monitoring, and managing PBC patients with moderate to severe liver disease taking Ocaliva.”
FDA recommends that “health care professionals should follow the Ocaliva dosing regimen in the drug label. … Dosing higher than recommended in the drug label can increase the risk for liver decompensation, liver failure, and sometimes death. Routinely monitor all patients for biochemical response, tolerability, and PBC progression, and reevaluate Child-Pugh classification to determine if dosage adjustment is needed.”
Manufacturer Intercept Pharmaceuticals was required to continue studying obeticholic acid in patients with advanced PBC as a condition of its FDA approval. Results from these studies are expected in 2023, FDA noted.
To report adverse medication events and side effects to the FDA, access the MedWatch program.
The Food and Drug Administration is requiring a boxed warning on the label for obeticholic acid (Ocaliva) to highlight the correct weekly dosing regimen after incorrect daily dosing caused severe liver injury in patients with moderate to severe primary biliary cholangitis (PBC).
“FDA is adding a new Boxed Warning, FDA’s most prominent warning, to highlight this information in the prescribing information of the drug label,” FDA officials said in a statement Feb. 1. “To ensure correct dosing and reduce the risk of liver problems, FDA is clarifying the current recommendations for screening, dosing, monitoring, and managing PBC patients with moderate to severe liver disease taking Ocaliva.”
FDA recommends that “health care professionals should follow the Ocaliva dosing regimen in the drug label. … Dosing higher than recommended in the drug label can increase the risk for liver decompensation, liver failure, and sometimes death. Routinely monitor all patients for biochemical response, tolerability, and PBC progression, and reevaluate Child-Pugh classification to determine if dosage adjustment is needed.”
Manufacturer Intercept Pharmaceuticals was required to continue studying obeticholic acid in patients with advanced PBC as a condition of its FDA approval. Results from these studies are expected in 2023, FDA noted.
To report adverse medication events and side effects to the FDA, access the MedWatch program.
The Food and Drug Administration is requiring a boxed warning on the label for obeticholic acid (Ocaliva) to highlight the correct weekly dosing regimen after incorrect daily dosing caused severe liver injury in patients with moderate to severe primary biliary cholangitis (PBC).
“FDA is adding a new Boxed Warning, FDA’s most prominent warning, to highlight this information in the prescribing information of the drug label,” FDA officials said in a statement Feb. 1. “To ensure correct dosing and reduce the risk of liver problems, FDA is clarifying the current recommendations for screening, dosing, monitoring, and managing PBC patients with moderate to severe liver disease taking Ocaliva.”
FDA recommends that “health care professionals should follow the Ocaliva dosing regimen in the drug label. … Dosing higher than recommended in the drug label can increase the risk for liver decompensation, liver failure, and sometimes death. Routinely monitor all patients for biochemical response, tolerability, and PBC progression, and reevaluate Child-Pugh classification to determine if dosage adjustment is needed.”
Manufacturer Intercept Pharmaceuticals was required to continue studying obeticholic acid in patients with advanced PBC as a condition of its FDA approval. Results from these studies are expected in 2023, FDA noted.
To report adverse medication events and side effects to the FDA, access the MedWatch program.
Three in 10 diabetic patients may have liver fibrosis
LOS ANGELES – For every 10 adult patients with type 2 diabetes, three are likely to have moderate to severe liver fibrosis, according to Kenneth Cusi, MD, FACP, FACE.
“The question is, How are we going to tackle this problem? My academic goal is that we incorporate screening for NASH [nonalcoholic steatohepatitis], or for fibrosis more specifically, in the same way we do for retinopathy or nephropathy [in diabetes], because we do have a way to treat it,” he said at the World Congress on Insulin Resistance, Diabetes & Cardiovascular Disease.
Dr. Cusi, chief of the division of endocrinology, diabetes, and metabolism at the University of Florida, Gainesville, predicted that obesity will become the No. 1 cause of liver transplantation. “It’s a real epidemic; you’re not seeing it because the inflexion of obesity happened just 2 decades ago,” he said. “Patients with diabetes face the greatest risk of fatty liver and of fibrosis. Untreated, it’s the equivalent of having macroalbuminuria. If you do nothing and they don’t die of cardiovascular disease, they’re going to have a good chance of getting fibrosis.”
As part of the large population-based Rotterdam study of individuals aged 45 years and older, researchers found that liver stiffness of 8 kPa or more by transient elastography was present in 5.6% of the study participants and was strongly associated with steatosis and diabetes (Hepatology. 2016;63:138-47). According to Dr. Cusi, individuals who have steatosis without diabetes face a 5%-10% risk of fibrosis, while those with steatosis and diabetes face a 15%-20% risk. “It’s well established in a number of studies that if you have fibrosis, you’re at high risk not only of cirrhosis, but also of hepatocellular carcinoma,” he said. “The key thing is not detecting fat, which is not really the target. The target is if there’s fibrosis or not.” Three ways to assess for fibrosis include MR elastography, transient elastography (which is the most commonly used), and fibrosis marker panels.
Liver fibrosis likely starts with adipose tissue dysfunction, said Dr. Cusi, who authored a review on the pathophysiology of interactions between adipose tissue and target organs in obesity and the resulting clinical implications for the management of nonalcoholic steatohepatitis (Gastroenterology. 2012;142[4]:711-25.e6). “When you have insulin-resistant, sick adipose tissue, that leads to the accumulation of fat in the liver,” he said. “. Even if you get people who are matched for BMIs [body mass indexes] between 30 and 35 kg/m2, there is a spectrum in which some individuals have very insulin-resistant adipose tissue and others less so. I would say that 1 out of 10 are metabolically healthy, and we don’t understand exactly why.”
In a recent cross-sectional analysis of 352 healthy individuals, Dr. Cusi and his associates found that intrahepatic triglyceride (IHTG) accumulation is strongly associated with adipose tissue insulin resistance, supporting the current theory of lipotoxicity as a driver of IHTG accumulation (Hepatology. 2017;65[4]:1132-44). The researchers observed that once IHTG accumulation reaches about 6%, skeletal muscle insulin resistance, hypertriglyceridemia, and low HDL cholesterol become fully established. “The next question is, How does this correlate with NASH?” Dr. Cusi said. “Our take is that there is a threshold effect. Once you have a critical amount of triglycerides in your liver, some individuals are going to activate pathways that are harmful. NASH is not something exclusive to individuals who are obese. Lean people can also develop NASH. The key feature is insulin resistance, not metabolic syndrome. Once you develop a fatty liver, your chances of NASH are comparable to that of an obese individual. The paradox is that lean individuals get a fatty liver, but when they get a fatty liver, they are at risk for NASH and for fibrosis.”
Why lean individuals develop NASH is not fully understood, but Dr. Cusi said he suspects that the problem develops at the mitochondrial level. Results from an unpublished animal model in which mice were fed a high–trans-fat diet for 24 weeks showed that the mice developed steatosis by week 8 and NASH by week 24. The mice had an increase in the tricarboxylic acid (TCA) cycle, which is typical of the NASH period, as well as an increase in ceramides. “Perhaps a unifying hypothesis would be that the development of NASH is linked to inflammation and to insulin signaling,” Dr. Cusi said. “Not surprisingly, it had a number of effects on the mitochondria, and in this animal model it decreases the TCA.” He noted that the biology of fibrosis remains unknown in humans. “What we have been familiar with is the high-triglyceride, low-HDL pattern,” he said. “If you look at how that correlates with the amount of liver fat, it is basically a threshold effect. Once you have steatosis, you don’t see much worse dyslipidemia, which is typical of these patients.”
Recently published guidance from the American Association for the Study of Liver Diseases on the diagnosis and management of nonalcoholic fatty liver disease (NAFLD) suggests that patients require a weight loss of 3%-5% to improve steatosis, but a loss of 7%-10% to improve most histologic features of NASH, including fibrosis (Hepatology. 2018;67[1]:328-57). Exercise alone may prevent or reduce steatosis, but its ability to improve other aspects of liver histology remains unknown. Bariatric surgery can be considered in otherwise eligible obese individuals with NAFLD or NASH. The procedure’s impact on fibrosis is unknown.
The AASLD practice guideline notes that metformin is not recommended for treating NASH in adult patients, but pioglitazone improves liver histology in patients with and without type 2 diabetes with biopsy-proven NASH. “Pioglitazone has had the greatest benefit in terms of treatment effect, compared to placebo,” Dr. Cusi said. “It’s a generic drug; at the VA [Veterans Affairs], it costs 8 cents per tablet. I think that pioglitazone will be to NASH what metformin has been to type 2 diabetes. The most common side effect is weight gain, typically between 4 and 9 lb. Risks and benefits should be discussed with each patient. It should not be used for NAFLD without biopsy-proven NASH.” The guideline goes on to say that it’s currently premature to consider GLP-1 (glucagonlike peptide–1) agonists for treating liver disease in patients with NAFLD or NASH. Meanwhile, vitamin E at 800 IU has been shown to improve liver histology in nondiabetic adults with NASH, but the risks and benefits should be discussed with each patient. Vitamin E is not recommended for NASH in diabetic patients, NAFLD without a liver biopsy, NASH cirrhosis, or cryptogenic cirrhosis.
The AASLD practice guideline also states that the best evidence for using SGLT2 (sodium-glucose cotransporter–2) inhibitors in NAFLD comes from animal studies, which report a reduction in steatosis with and without weight loss. Clinical studies reporting a reduction in steatosis are limited. There are positive observational studies with a reduction in alanine aminotransferase and some studies that have shown a reduction in liver fat. “For me, the best option is to tailor treatment to the pathophysiology of the disease,” Dr. Cusi said. “You reduce fat by weight loss in some way, or you change the biology of fat with a thiazolidinedione.”
Dr. Cusi reported that he has received grant support from the Burroughs Wellcome Fund, the American Diabetes Association, and the National Institutes of Health.
LOS ANGELES – For every 10 adult patients with type 2 diabetes, three are likely to have moderate to severe liver fibrosis, according to Kenneth Cusi, MD, FACP, FACE.
“The question is, How are we going to tackle this problem? My academic goal is that we incorporate screening for NASH [nonalcoholic steatohepatitis], or for fibrosis more specifically, in the same way we do for retinopathy or nephropathy [in diabetes], because we do have a way to treat it,” he said at the World Congress on Insulin Resistance, Diabetes & Cardiovascular Disease.
Dr. Cusi, chief of the division of endocrinology, diabetes, and metabolism at the University of Florida, Gainesville, predicted that obesity will become the No. 1 cause of liver transplantation. “It’s a real epidemic; you’re not seeing it because the inflexion of obesity happened just 2 decades ago,” he said. “Patients with diabetes face the greatest risk of fatty liver and of fibrosis. Untreated, it’s the equivalent of having macroalbuminuria. If you do nothing and they don’t die of cardiovascular disease, they’re going to have a good chance of getting fibrosis.”
As part of the large population-based Rotterdam study of individuals aged 45 years and older, researchers found that liver stiffness of 8 kPa or more by transient elastography was present in 5.6% of the study participants and was strongly associated with steatosis and diabetes (Hepatology. 2016;63:138-47). According to Dr. Cusi, individuals who have steatosis without diabetes face a 5%-10% risk of fibrosis, while those with steatosis and diabetes face a 15%-20% risk. “It’s well established in a number of studies that if you have fibrosis, you’re at high risk not only of cirrhosis, but also of hepatocellular carcinoma,” he said. “The key thing is not detecting fat, which is not really the target. The target is if there’s fibrosis or not.” Three ways to assess for fibrosis include MR elastography, transient elastography (which is the most commonly used), and fibrosis marker panels.
Liver fibrosis likely starts with adipose tissue dysfunction, said Dr. Cusi, who authored a review on the pathophysiology of interactions between adipose tissue and target organs in obesity and the resulting clinical implications for the management of nonalcoholic steatohepatitis (Gastroenterology. 2012;142[4]:711-25.e6). “When you have insulin-resistant, sick adipose tissue, that leads to the accumulation of fat in the liver,” he said. “. Even if you get people who are matched for BMIs [body mass indexes] between 30 and 35 kg/m2, there is a spectrum in which some individuals have very insulin-resistant adipose tissue and others less so. I would say that 1 out of 10 are metabolically healthy, and we don’t understand exactly why.”
In a recent cross-sectional analysis of 352 healthy individuals, Dr. Cusi and his associates found that intrahepatic triglyceride (IHTG) accumulation is strongly associated with adipose tissue insulin resistance, supporting the current theory of lipotoxicity as a driver of IHTG accumulation (Hepatology. 2017;65[4]:1132-44). The researchers observed that once IHTG accumulation reaches about 6%, skeletal muscle insulin resistance, hypertriglyceridemia, and low HDL cholesterol become fully established. “The next question is, How does this correlate with NASH?” Dr. Cusi said. “Our take is that there is a threshold effect. Once you have a critical amount of triglycerides in your liver, some individuals are going to activate pathways that are harmful. NASH is not something exclusive to individuals who are obese. Lean people can also develop NASH. The key feature is insulin resistance, not metabolic syndrome. Once you develop a fatty liver, your chances of NASH are comparable to that of an obese individual. The paradox is that lean individuals get a fatty liver, but when they get a fatty liver, they are at risk for NASH and for fibrosis.”
Why lean individuals develop NASH is not fully understood, but Dr. Cusi said he suspects that the problem develops at the mitochondrial level. Results from an unpublished animal model in which mice were fed a high–trans-fat diet for 24 weeks showed that the mice developed steatosis by week 8 and NASH by week 24. The mice had an increase in the tricarboxylic acid (TCA) cycle, which is typical of the NASH period, as well as an increase in ceramides. “Perhaps a unifying hypothesis would be that the development of NASH is linked to inflammation and to insulin signaling,” Dr. Cusi said. “Not surprisingly, it had a number of effects on the mitochondria, and in this animal model it decreases the TCA.” He noted that the biology of fibrosis remains unknown in humans. “What we have been familiar with is the high-triglyceride, low-HDL pattern,” he said. “If you look at how that correlates with the amount of liver fat, it is basically a threshold effect. Once you have steatosis, you don’t see much worse dyslipidemia, which is typical of these patients.”
Recently published guidance from the American Association for the Study of Liver Diseases on the diagnosis and management of nonalcoholic fatty liver disease (NAFLD) suggests that patients require a weight loss of 3%-5% to improve steatosis, but a loss of 7%-10% to improve most histologic features of NASH, including fibrosis (Hepatology. 2018;67[1]:328-57). Exercise alone may prevent or reduce steatosis, but its ability to improve other aspects of liver histology remains unknown. Bariatric surgery can be considered in otherwise eligible obese individuals with NAFLD or NASH. The procedure’s impact on fibrosis is unknown.
The AASLD practice guideline notes that metformin is not recommended for treating NASH in adult patients, but pioglitazone improves liver histology in patients with and without type 2 diabetes with biopsy-proven NASH. “Pioglitazone has had the greatest benefit in terms of treatment effect, compared to placebo,” Dr. Cusi said. “It’s a generic drug; at the VA [Veterans Affairs], it costs 8 cents per tablet. I think that pioglitazone will be to NASH what metformin has been to type 2 diabetes. The most common side effect is weight gain, typically between 4 and 9 lb. Risks and benefits should be discussed with each patient. It should not be used for NAFLD without biopsy-proven NASH.” The guideline goes on to say that it’s currently premature to consider GLP-1 (glucagonlike peptide–1) agonists for treating liver disease in patients with NAFLD or NASH. Meanwhile, vitamin E at 800 IU has been shown to improve liver histology in nondiabetic adults with NASH, but the risks and benefits should be discussed with each patient. Vitamin E is not recommended for NASH in diabetic patients, NAFLD without a liver biopsy, NASH cirrhosis, or cryptogenic cirrhosis.
The AASLD practice guideline also states that the best evidence for using SGLT2 (sodium-glucose cotransporter–2) inhibitors in NAFLD comes from animal studies, which report a reduction in steatosis with and without weight loss. Clinical studies reporting a reduction in steatosis are limited. There are positive observational studies with a reduction in alanine aminotransferase and some studies that have shown a reduction in liver fat. “For me, the best option is to tailor treatment to the pathophysiology of the disease,” Dr. Cusi said. “You reduce fat by weight loss in some way, or you change the biology of fat with a thiazolidinedione.”
Dr. Cusi reported that he has received grant support from the Burroughs Wellcome Fund, the American Diabetes Association, and the National Institutes of Health.
LOS ANGELES – For every 10 adult patients with type 2 diabetes, three are likely to have moderate to severe liver fibrosis, according to Kenneth Cusi, MD, FACP, FACE.
“The question is, How are we going to tackle this problem? My academic goal is that we incorporate screening for NASH [nonalcoholic steatohepatitis], or for fibrosis more specifically, in the same way we do for retinopathy or nephropathy [in diabetes], because we do have a way to treat it,” he said at the World Congress on Insulin Resistance, Diabetes & Cardiovascular Disease.
Dr. Cusi, chief of the division of endocrinology, diabetes, and metabolism at the University of Florida, Gainesville, predicted that obesity will become the No. 1 cause of liver transplantation. “It’s a real epidemic; you’re not seeing it because the inflexion of obesity happened just 2 decades ago,” he said. “Patients with diabetes face the greatest risk of fatty liver and of fibrosis. Untreated, it’s the equivalent of having macroalbuminuria. If you do nothing and they don’t die of cardiovascular disease, they’re going to have a good chance of getting fibrosis.”
As part of the large population-based Rotterdam study of individuals aged 45 years and older, researchers found that liver stiffness of 8 kPa or more by transient elastography was present in 5.6% of the study participants and was strongly associated with steatosis and diabetes (Hepatology. 2016;63:138-47). According to Dr. Cusi, individuals who have steatosis without diabetes face a 5%-10% risk of fibrosis, while those with steatosis and diabetes face a 15%-20% risk. “It’s well established in a number of studies that if you have fibrosis, you’re at high risk not only of cirrhosis, but also of hepatocellular carcinoma,” he said. “The key thing is not detecting fat, which is not really the target. The target is if there’s fibrosis or not.” Three ways to assess for fibrosis include MR elastography, transient elastography (which is the most commonly used), and fibrosis marker panels.
Liver fibrosis likely starts with adipose tissue dysfunction, said Dr. Cusi, who authored a review on the pathophysiology of interactions between adipose tissue and target organs in obesity and the resulting clinical implications for the management of nonalcoholic steatohepatitis (Gastroenterology. 2012;142[4]:711-25.e6). “When you have insulin-resistant, sick adipose tissue, that leads to the accumulation of fat in the liver,” he said. “. Even if you get people who are matched for BMIs [body mass indexes] between 30 and 35 kg/m2, there is a spectrum in which some individuals have very insulin-resistant adipose tissue and others less so. I would say that 1 out of 10 are metabolically healthy, and we don’t understand exactly why.”
In a recent cross-sectional analysis of 352 healthy individuals, Dr. Cusi and his associates found that intrahepatic triglyceride (IHTG) accumulation is strongly associated with adipose tissue insulin resistance, supporting the current theory of lipotoxicity as a driver of IHTG accumulation (Hepatology. 2017;65[4]:1132-44). The researchers observed that once IHTG accumulation reaches about 6%, skeletal muscle insulin resistance, hypertriglyceridemia, and low HDL cholesterol become fully established. “The next question is, How does this correlate with NASH?” Dr. Cusi said. “Our take is that there is a threshold effect. Once you have a critical amount of triglycerides in your liver, some individuals are going to activate pathways that are harmful. NASH is not something exclusive to individuals who are obese. Lean people can also develop NASH. The key feature is insulin resistance, not metabolic syndrome. Once you develop a fatty liver, your chances of NASH are comparable to that of an obese individual. The paradox is that lean individuals get a fatty liver, but when they get a fatty liver, they are at risk for NASH and for fibrosis.”
Why lean individuals develop NASH is not fully understood, but Dr. Cusi said he suspects that the problem develops at the mitochondrial level. Results from an unpublished animal model in which mice were fed a high–trans-fat diet for 24 weeks showed that the mice developed steatosis by week 8 and NASH by week 24. The mice had an increase in the tricarboxylic acid (TCA) cycle, which is typical of the NASH period, as well as an increase in ceramides. “Perhaps a unifying hypothesis would be that the development of NASH is linked to inflammation and to insulin signaling,” Dr. Cusi said. “Not surprisingly, it had a number of effects on the mitochondria, and in this animal model it decreases the TCA.” He noted that the biology of fibrosis remains unknown in humans. “What we have been familiar with is the high-triglyceride, low-HDL pattern,” he said. “If you look at how that correlates with the amount of liver fat, it is basically a threshold effect. Once you have steatosis, you don’t see much worse dyslipidemia, which is typical of these patients.”
Recently published guidance from the American Association for the Study of Liver Diseases on the diagnosis and management of nonalcoholic fatty liver disease (NAFLD) suggests that patients require a weight loss of 3%-5% to improve steatosis, but a loss of 7%-10% to improve most histologic features of NASH, including fibrosis (Hepatology. 2018;67[1]:328-57). Exercise alone may prevent or reduce steatosis, but its ability to improve other aspects of liver histology remains unknown. Bariatric surgery can be considered in otherwise eligible obese individuals with NAFLD or NASH. The procedure’s impact on fibrosis is unknown.
The AASLD practice guideline notes that metformin is not recommended for treating NASH in adult patients, but pioglitazone improves liver histology in patients with and without type 2 diabetes with biopsy-proven NASH. “Pioglitazone has had the greatest benefit in terms of treatment effect, compared to placebo,” Dr. Cusi said. “It’s a generic drug; at the VA [Veterans Affairs], it costs 8 cents per tablet. I think that pioglitazone will be to NASH what metformin has been to type 2 diabetes. The most common side effect is weight gain, typically between 4 and 9 lb. Risks and benefits should be discussed with each patient. It should not be used for NAFLD without biopsy-proven NASH.” The guideline goes on to say that it’s currently premature to consider GLP-1 (glucagonlike peptide–1) agonists for treating liver disease in patients with NAFLD or NASH. Meanwhile, vitamin E at 800 IU has been shown to improve liver histology in nondiabetic adults with NASH, but the risks and benefits should be discussed with each patient. Vitamin E is not recommended for NASH in diabetic patients, NAFLD without a liver biopsy, NASH cirrhosis, or cryptogenic cirrhosis.
The AASLD practice guideline also states that the best evidence for using SGLT2 (sodium-glucose cotransporter–2) inhibitors in NAFLD comes from animal studies, which report a reduction in steatosis with and without weight loss. Clinical studies reporting a reduction in steatosis are limited. There are positive observational studies with a reduction in alanine aminotransferase and some studies that have shown a reduction in liver fat. “For me, the best option is to tailor treatment to the pathophysiology of the disease,” Dr. Cusi said. “You reduce fat by weight loss in some way, or you change the biology of fat with a thiazolidinedione.”
Dr. Cusi reported that he has received grant support from the Burroughs Wellcome Fund, the American Diabetes Association, and the National Institutes of Health.
EXPERT ANALYSIS FROM WCIRDC 2017
Isolated severe tricuspid regurgitation: An emerging disease
SNOWMASS, COLO. – that’s treatable, provided affected patients are referred for surgery before the clinical course progresses to intractable right heart disease with cirrhosis and liver failure, Rick A. Nishimura, MD, said at the Annual Cardiovascular Conference at Snowmass.
“This is an emerging disease that you’re all going to see in your practices this year. You’ve got to know what to do with these patients. Get to them early,” urged Dr. Nishimura, professor of cardiovascular sciences and hypertension at the Mayo Clinic in Rochester, Minn.
The medical textbooks don’t discuss isolated severe tricuspid regurgitation (ISTR) or its etiology. ISTR is a disorder of progressive right ventricular dilation and dysfunction whose etiology involves either longstanding atrial fibrillation or valvular disruption due to interference from a crossing lead of a permanent pacemaker or implantable cardioverter defibrillator.
“This is something different. These patients have a normal left heart and left heart valves and normal pressures, with no pulmonary hypertension. So it doesn’t fit into any of the textbook categories of tricuspid regurgitation,” the cardiologist said.
Moreover, the current American College of Cardiology/American Heart Association guidelines on valvular heart disease don’t address ISTR, either. Physicians who attempt to apply the guidelines in deciding when to refer a patient with ISTR for surgery will oftentimes find they’ve waited too long and the patient has started to develop end-stage disease, according to Dr. Nishimura.
And he should know: He was lead author of the current ACC/AHA guidelines (J Am Coll Cardiol. 2014 Jun 10;63[22]:2438-88).
Decades ago, Eugene Braunwald, MD, of Harvard Medical School, Boston, famously called the tricuspid valve “the forgotten valve.” The history of the Snowmass winter cardiology conference bears that out. During 2007-2017, the conference featured an average of 5.4 sessions per year on aortic valve disease, 4.5 sessions per year on mitral valve disease, and not a single session on tricuspid valve disease. But the tricuspid valve is forgotten no longer, Dr. Nishimura emphasized.
How ISTR presents
The affected patient has a history of either longstanding atrial fibrillation or a permanent pacemaker or ICD.
“This is something that 4 or 5 years ago people said didn’t exist. Our pacemaker people told me, ‘Nah, you can never get tricuspid regurgitation from our leads.’ Now it’s one of the leading causes of tricuspid regurgitation going to operation,” said Dr. Nishimura.
The presenting symptoms of ISTR are typically ascites, edema, and shortness of breath.
“Why should patients with a right heart problem get dyspnea? It turns out that when the right ventricle dilates it pushes the septum in, so the effective operative compliance of the left ventricle decreases and you actually see the pulmonary artery wedge pressure go up,” he explained.
On physical examination, the patient will have elevated jugular venous pressure with large V waves.
“This is a clue that something is going on. The patient will have neck veins jumping up to her ear lobes. The ear lobes are going to wiggle with every heart beat – boom, boom, boom. If you see that, you start to figure out what’s going on. You need nothing else,” Dr. Nishimura said.
The patient will likely also have a pulsatile enlarged liver and, even though this is valve disease, a murmur that’s either soft or inaudible.
Echocardiographic diagnosis
Echocardiography will show a dilated right ventricle and right atrium, a dilated inferior vena cava, and a normal left ventricle with no pulmonary hypertension. The classic sign of ISTR on continuous wave Doppler echocardiography is a dagger-shaped tricuspid regurgitation peak velocity signal of less than 2.5 meters/sec, which indicates the absence of pulmonary hypertension. This dagger shape occurs because the right atrial pressure equalizes the right ventricular pressure.
It’s also important to point the echo probe at the hepatic veins to spot another echocardiographic hallmark of ISTR: systolic reversal.
A thorough echo exam makes hemodynamic catheterization unnecessary in these patients, Dr. Nishimura added.
When to refer for tricuspid valve repair or replacement
The clinical course of ISTR is progressive, often rapidly so. It starts with elevated jugular venous pressure, then comes fatigue and shortness of breath, moving on to ascites and edema, then finally cirrhosis and renal failure. It’s a vicious cycle in which tricuspid regurgitation begets annular dilation, which causes chordal stretching and worsening tricuspid regurgitation, leading to further annular dilation.
Patients typically aren’t referred for surgery – and may not even present to a physician – until they’ve already developed end-stage disease. That’s probably why the outcomes of surgery for ISTR are so poor. Dr. Nishimura was senior investigator of a recent retrospective study of national trends and outcomes for ISTR surgery based on the National Inpatient Sample. The number of operations increased by 250% during a recent 10-year period, but the surgery is still rare: 290 operations in 2004, climbing to 780 nationwide in 2013.
In-hospital mortality remained steady over time at 8.8%, far higher than rates of in-hospital mortality for surgery for aortic and mitral valve disease, which today stand at 1%-2% or less. The adjusted risk of in-hospital mortality for tricuspid valve replacement in patients with ISTR was 1.9-fold greater than for valve repair (J Am Coll Cardiol. 2017 Dec 19;70[24]:2953-60).
“I think the reason the operative risk of valve surgery for ISTR is so high is that we’re waiting until patients have end-stage disease,” Dr. Nishimura said.
Indeed, he recommends referral for surgery as soon as the echocardiographic diagnosis of ISTR is made in a patient with huge neck veins.
“This will probably take the operative risk down by going to a time when the right ventricle can still recover,” he added.
In a patient with ISTR and pacemaker or defibrillator leads crossing the valve, tricuspid valve repair or replacement should be accompanied by exteriorization of the leads.
Dr. Nishimura reported having no financial conflicts of interest regarding his presentation.
SNOWMASS, COLO. – that’s treatable, provided affected patients are referred for surgery before the clinical course progresses to intractable right heart disease with cirrhosis and liver failure, Rick A. Nishimura, MD, said at the Annual Cardiovascular Conference at Snowmass.
“This is an emerging disease that you’re all going to see in your practices this year. You’ve got to know what to do with these patients. Get to them early,” urged Dr. Nishimura, professor of cardiovascular sciences and hypertension at the Mayo Clinic in Rochester, Minn.
The medical textbooks don’t discuss isolated severe tricuspid regurgitation (ISTR) or its etiology. ISTR is a disorder of progressive right ventricular dilation and dysfunction whose etiology involves either longstanding atrial fibrillation or valvular disruption due to interference from a crossing lead of a permanent pacemaker or implantable cardioverter defibrillator.
“This is something different. These patients have a normal left heart and left heart valves and normal pressures, with no pulmonary hypertension. So it doesn’t fit into any of the textbook categories of tricuspid regurgitation,” the cardiologist said.
Moreover, the current American College of Cardiology/American Heart Association guidelines on valvular heart disease don’t address ISTR, either. Physicians who attempt to apply the guidelines in deciding when to refer a patient with ISTR for surgery will oftentimes find they’ve waited too long and the patient has started to develop end-stage disease, according to Dr. Nishimura.
And he should know: He was lead author of the current ACC/AHA guidelines (J Am Coll Cardiol. 2014 Jun 10;63[22]:2438-88).
Decades ago, Eugene Braunwald, MD, of Harvard Medical School, Boston, famously called the tricuspid valve “the forgotten valve.” The history of the Snowmass winter cardiology conference bears that out. During 2007-2017, the conference featured an average of 5.4 sessions per year on aortic valve disease, 4.5 sessions per year on mitral valve disease, and not a single session on tricuspid valve disease. But the tricuspid valve is forgotten no longer, Dr. Nishimura emphasized.
How ISTR presents
The affected patient has a history of either longstanding atrial fibrillation or a permanent pacemaker or ICD.
“This is something that 4 or 5 years ago people said didn’t exist. Our pacemaker people told me, ‘Nah, you can never get tricuspid regurgitation from our leads.’ Now it’s one of the leading causes of tricuspid regurgitation going to operation,” said Dr. Nishimura.
The presenting symptoms of ISTR are typically ascites, edema, and shortness of breath.
“Why should patients with a right heart problem get dyspnea? It turns out that when the right ventricle dilates it pushes the septum in, so the effective operative compliance of the left ventricle decreases and you actually see the pulmonary artery wedge pressure go up,” he explained.
On physical examination, the patient will have elevated jugular venous pressure with large V waves.
“This is a clue that something is going on. The patient will have neck veins jumping up to her ear lobes. The ear lobes are going to wiggle with every heart beat – boom, boom, boom. If you see that, you start to figure out what’s going on. You need nothing else,” Dr. Nishimura said.
The patient will likely also have a pulsatile enlarged liver and, even though this is valve disease, a murmur that’s either soft or inaudible.
Echocardiographic diagnosis
Echocardiography will show a dilated right ventricle and right atrium, a dilated inferior vena cava, and a normal left ventricle with no pulmonary hypertension. The classic sign of ISTR on continuous wave Doppler echocardiography is a dagger-shaped tricuspid regurgitation peak velocity signal of less than 2.5 meters/sec, which indicates the absence of pulmonary hypertension. This dagger shape occurs because the right atrial pressure equalizes the right ventricular pressure.
It’s also important to point the echo probe at the hepatic veins to spot another echocardiographic hallmark of ISTR: systolic reversal.
A thorough echo exam makes hemodynamic catheterization unnecessary in these patients, Dr. Nishimura added.
When to refer for tricuspid valve repair or replacement
The clinical course of ISTR is progressive, often rapidly so. It starts with elevated jugular venous pressure, then comes fatigue and shortness of breath, moving on to ascites and edema, then finally cirrhosis and renal failure. It’s a vicious cycle in which tricuspid regurgitation begets annular dilation, which causes chordal stretching and worsening tricuspid regurgitation, leading to further annular dilation.
Patients typically aren’t referred for surgery – and may not even present to a physician – until they’ve already developed end-stage disease. That’s probably why the outcomes of surgery for ISTR are so poor. Dr. Nishimura was senior investigator of a recent retrospective study of national trends and outcomes for ISTR surgery based on the National Inpatient Sample. The number of operations increased by 250% during a recent 10-year period, but the surgery is still rare: 290 operations in 2004, climbing to 780 nationwide in 2013.
In-hospital mortality remained steady over time at 8.8%, far higher than rates of in-hospital mortality for surgery for aortic and mitral valve disease, which today stand at 1%-2% or less. The adjusted risk of in-hospital mortality for tricuspid valve replacement in patients with ISTR was 1.9-fold greater than for valve repair (J Am Coll Cardiol. 2017 Dec 19;70[24]:2953-60).
“I think the reason the operative risk of valve surgery for ISTR is so high is that we’re waiting until patients have end-stage disease,” Dr. Nishimura said.
Indeed, he recommends referral for surgery as soon as the echocardiographic diagnosis of ISTR is made in a patient with huge neck veins.
“This will probably take the operative risk down by going to a time when the right ventricle can still recover,” he added.
In a patient with ISTR and pacemaker or defibrillator leads crossing the valve, tricuspid valve repair or replacement should be accompanied by exteriorization of the leads.
Dr. Nishimura reported having no financial conflicts of interest regarding his presentation.
SNOWMASS, COLO. – that’s treatable, provided affected patients are referred for surgery before the clinical course progresses to intractable right heart disease with cirrhosis and liver failure, Rick A. Nishimura, MD, said at the Annual Cardiovascular Conference at Snowmass.
“This is an emerging disease that you’re all going to see in your practices this year. You’ve got to know what to do with these patients. Get to them early,” urged Dr. Nishimura, professor of cardiovascular sciences and hypertension at the Mayo Clinic in Rochester, Minn.
The medical textbooks don’t discuss isolated severe tricuspid regurgitation (ISTR) or its etiology. ISTR is a disorder of progressive right ventricular dilation and dysfunction whose etiology involves either longstanding atrial fibrillation or valvular disruption due to interference from a crossing lead of a permanent pacemaker or implantable cardioverter defibrillator.
“This is something different. These patients have a normal left heart and left heart valves and normal pressures, with no pulmonary hypertension. So it doesn’t fit into any of the textbook categories of tricuspid regurgitation,” the cardiologist said.
Moreover, the current American College of Cardiology/American Heart Association guidelines on valvular heart disease don’t address ISTR, either. Physicians who attempt to apply the guidelines in deciding when to refer a patient with ISTR for surgery will oftentimes find they’ve waited too long and the patient has started to develop end-stage disease, according to Dr. Nishimura.
And he should know: He was lead author of the current ACC/AHA guidelines (J Am Coll Cardiol. 2014 Jun 10;63[22]:2438-88).
Decades ago, Eugene Braunwald, MD, of Harvard Medical School, Boston, famously called the tricuspid valve “the forgotten valve.” The history of the Snowmass winter cardiology conference bears that out. During 2007-2017, the conference featured an average of 5.4 sessions per year on aortic valve disease, 4.5 sessions per year on mitral valve disease, and not a single session on tricuspid valve disease. But the tricuspid valve is forgotten no longer, Dr. Nishimura emphasized.
How ISTR presents
The affected patient has a history of either longstanding atrial fibrillation or a permanent pacemaker or ICD.
“This is something that 4 or 5 years ago people said didn’t exist. Our pacemaker people told me, ‘Nah, you can never get tricuspid regurgitation from our leads.’ Now it’s one of the leading causes of tricuspid regurgitation going to operation,” said Dr. Nishimura.
The presenting symptoms of ISTR are typically ascites, edema, and shortness of breath.
“Why should patients with a right heart problem get dyspnea? It turns out that when the right ventricle dilates it pushes the septum in, so the effective operative compliance of the left ventricle decreases and you actually see the pulmonary artery wedge pressure go up,” he explained.
On physical examination, the patient will have elevated jugular venous pressure with large V waves.
“This is a clue that something is going on. The patient will have neck veins jumping up to her ear lobes. The ear lobes are going to wiggle with every heart beat – boom, boom, boom. If you see that, you start to figure out what’s going on. You need nothing else,” Dr. Nishimura said.
The patient will likely also have a pulsatile enlarged liver and, even though this is valve disease, a murmur that’s either soft or inaudible.
Echocardiographic diagnosis
Echocardiography will show a dilated right ventricle and right atrium, a dilated inferior vena cava, and a normal left ventricle with no pulmonary hypertension. The classic sign of ISTR on continuous wave Doppler echocardiography is a dagger-shaped tricuspid regurgitation peak velocity signal of less than 2.5 meters/sec, which indicates the absence of pulmonary hypertension. This dagger shape occurs because the right atrial pressure equalizes the right ventricular pressure.
It’s also important to point the echo probe at the hepatic veins to spot another echocardiographic hallmark of ISTR: systolic reversal.
A thorough echo exam makes hemodynamic catheterization unnecessary in these patients, Dr. Nishimura added.
When to refer for tricuspid valve repair or replacement
The clinical course of ISTR is progressive, often rapidly so. It starts with elevated jugular venous pressure, then comes fatigue and shortness of breath, moving on to ascites and edema, then finally cirrhosis and renal failure. It’s a vicious cycle in which tricuspid regurgitation begets annular dilation, which causes chordal stretching and worsening tricuspid regurgitation, leading to further annular dilation.
Patients typically aren’t referred for surgery – and may not even present to a physician – until they’ve already developed end-stage disease. That’s probably why the outcomes of surgery for ISTR are so poor. Dr. Nishimura was senior investigator of a recent retrospective study of national trends and outcomes for ISTR surgery based on the National Inpatient Sample. The number of operations increased by 250% during a recent 10-year period, but the surgery is still rare: 290 operations in 2004, climbing to 780 nationwide in 2013.
In-hospital mortality remained steady over time at 8.8%, far higher than rates of in-hospital mortality for surgery for aortic and mitral valve disease, which today stand at 1%-2% or less. The adjusted risk of in-hospital mortality for tricuspid valve replacement in patients with ISTR was 1.9-fold greater than for valve repair (J Am Coll Cardiol. 2017 Dec 19;70[24]:2953-60).
“I think the reason the operative risk of valve surgery for ISTR is so high is that we’re waiting until patients have end-stage disease,” Dr. Nishimura said.
Indeed, he recommends referral for surgery as soon as the echocardiographic diagnosis of ISTR is made in a patient with huge neck veins.
“This will probably take the operative risk down by going to a time when the right ventricle can still recover,” he added.
In a patient with ISTR and pacemaker or defibrillator leads crossing the valve, tricuspid valve repair or replacement should be accompanied by exteriorization of the leads.
Dr. Nishimura reported having no financial conflicts of interest regarding his presentation.
EXPERT ANALYSIS FROM THE CARDIOVASCULAR CONFERENCE AT SNOWMASS
Eradicating HCV significantly improved liver stiffness in meta-analysis
Eradicating chronic hepatitis C virus (HCV) infection led to significant decreases in liver stiffness in a systematic review and meta-analysis of nearly 3,000 patients.
Mean liver stiffness fell by 4.1 kPa (kilopascals) (95% confidence interval, 3.3-4.9 kPa) 12 or more months after patients achieved sustained virologic response to treatment, but did not significantly change in patients who did not achieve SVR, reported Siddharth Singh, MD, of the University of San Diego, La Jolla, Calif., and his associates in the January issue of Clinical Gastroenterology and Hepatology (doi: 10.1016/j.cgh.2017.04.038). The results were especially striking among patients who received direct-acting antiviral agents (DAAs) or who had high baseline levels of inflammation, the investigators added.
SOURCE: AMERICAN GASTROENTEROLOGICAL ASSOCIATION
Based on these findings, about 47% of patients with advanced fibrosis or cirrhosis at baseline will drop below 9.5 kPa after achieving SVR, they reported. “With this decline in liver stiffness, it is conceivable that risk of liver-related complications would decrease, particularly in patients without cirrhosis,” they added. “Future research is warranted on the impact of magnitude and kinetics of decline in liver stiffness on improvement in liver-related outcomes.”
Eradicating HCV infection was known to decrease liver stiffness, but the magnitude of decline was not well understood. Therefore, the reviewers searched the literature through October 2016 for studies of HCV-infected adults who underwent liver stiffness measurement by vibration-controlled transient elastography before and at least once after completing HCV treatment. All studies also included data on median liver stiffness among patients who did and did not achieve SVR. The search identified 23 observational studies and one post hoc analysis of a randomized controlled trial, for a total of 2,934 patients, of whom 2,214 achieved SVR.
Among patients who achieved SVR, mean liver stiffness dropped by 2.4 kPa at the end of treatment (95% CI, 1.7-3.0 kPa), by 3.1 kPa 1-6 months later (95% CI, 1.6-4.7 kPa), and by 3.2 kPa 6-12 months after completing treatment (90% CI, 2.6-3.9 kPa). A year or more after finishing treatment, patients who achieved SVR had a 28% median decrease in liver stiffness (interquartile range, 22%-35%). However, liver stiffness did not significantly change among patients who did not achieve SVR, the reviewers reported.
Mean liver stiffness declined significantly more among patients who received DAAs (4.5 kPa) than among recipients of interferon-based regimens (2.6 kPa; P = .03). However, studies of DAAs included patients with greater liver stiffness at baseline, which could at least partially explain this discrepancy, the investigators said. Baseline cirrhosis also was associated with a greater decline in liver stiffness (mean, 5.1 kPa, vs. 2.8 kPa in patients without cirrhosis; P = .02), as was high baseline alanine aminotransferase level (P less than .01). Among patients whose baseline liver stiffness measurement exceeded 9.5 kPa, 47% had their liver stiffness drop to less than 9.5 kPa after achieving SVR.
Coinfection with HIV did not significantly alter the magnitude of decline in liver stiffness 6-12 months after treatment in patients who achieved SVR, the reviewers noted. “[Follow-up] assessment after SVR was relatively short; hence, long-term evolution of liver stiffness after antiviral therapy and impact of decline in liver stiffness on patient clinical outcomes could not be ascertained,” they wrote. The studies also did not consistently assess potential confounders such as nonalcoholic fatty liver disease, diabetes, and alcohol consumption.
One reviewer disclosed funding from the National Institutes of Health/National Library of Medicine. None had conflicts of interest.
The current era of new-generation direct-acting antiviral agents have revolutionized the treatment landscape of chronic hepatitis C virus infection, providing short-duration, safe, and consistently effective regimens that achieve SVR or cure in nearly 100% of patients. While achieving SVR is important, even more important is the long-term impact of SVR and whether cure translates into outcomes such as improved mortality or a reduced risk of disease progression. Although improved mortality after SVR has been demonstrated, one of the main drivers of risk of disease progression is the severity of hepatic fibrosis.
Robert J. Wong, MD, MS, is with the department of medicine and is director of research and education, division of gastroenterology and hepatology, Alameda Health System – Highland Hospital, Oakland, Calif. He has received a 2017-2019 Clinical Translational Research Award from AASLD, has received research funding from Gilead and AbbVie, and is on the speakers bureau of Gilead, Salix, and Bayer. He has also done consulting for and been an advisory board member for Gilead.
The current era of new-generation direct-acting antiviral agents have revolutionized the treatment landscape of chronic hepatitis C virus infection, providing short-duration, safe, and consistently effective regimens that achieve SVR or cure in nearly 100% of patients. While achieving SVR is important, even more important is the long-term impact of SVR and whether cure translates into outcomes such as improved mortality or a reduced risk of disease progression. Although improved mortality after SVR has been demonstrated, one of the main drivers of risk of disease progression is the severity of hepatic fibrosis.
Robert J. Wong, MD, MS, is with the department of medicine and is director of research and education, division of gastroenterology and hepatology, Alameda Health System – Highland Hospital, Oakland, Calif. He has received a 2017-2019 Clinical Translational Research Award from AASLD, has received research funding from Gilead and AbbVie, and is on the speakers bureau of Gilead, Salix, and Bayer. He has also done consulting for and been an advisory board member for Gilead.
The current era of new-generation direct-acting antiviral agents have revolutionized the treatment landscape of chronic hepatitis C virus infection, providing short-duration, safe, and consistently effective regimens that achieve SVR or cure in nearly 100% of patients. While achieving SVR is important, even more important is the long-term impact of SVR and whether cure translates into outcomes such as improved mortality or a reduced risk of disease progression. Although improved mortality after SVR has been demonstrated, one of the main drivers of risk of disease progression is the severity of hepatic fibrosis.
Robert J. Wong, MD, MS, is with the department of medicine and is director of research and education, division of gastroenterology and hepatology, Alameda Health System – Highland Hospital, Oakland, Calif. He has received a 2017-2019 Clinical Translational Research Award from AASLD, has received research funding from Gilead and AbbVie, and is on the speakers bureau of Gilead, Salix, and Bayer. He has also done consulting for and been an advisory board member for Gilead.
Eradicating chronic hepatitis C virus (HCV) infection led to significant decreases in liver stiffness in a systematic review and meta-analysis of nearly 3,000 patients.
Mean liver stiffness fell by 4.1 kPa (kilopascals) (95% confidence interval, 3.3-4.9 kPa) 12 or more months after patients achieved sustained virologic response to treatment, but did not significantly change in patients who did not achieve SVR, reported Siddharth Singh, MD, of the University of San Diego, La Jolla, Calif., and his associates in the January issue of Clinical Gastroenterology and Hepatology (doi: 10.1016/j.cgh.2017.04.038). The results were especially striking among patients who received direct-acting antiviral agents (DAAs) or who had high baseline levels of inflammation, the investigators added.
SOURCE: AMERICAN GASTROENTEROLOGICAL ASSOCIATION
Based on these findings, about 47% of patients with advanced fibrosis or cirrhosis at baseline will drop below 9.5 kPa after achieving SVR, they reported. “With this decline in liver stiffness, it is conceivable that risk of liver-related complications would decrease, particularly in patients without cirrhosis,” they added. “Future research is warranted on the impact of magnitude and kinetics of decline in liver stiffness on improvement in liver-related outcomes.”
Eradicating HCV infection was known to decrease liver stiffness, but the magnitude of decline was not well understood. Therefore, the reviewers searched the literature through October 2016 for studies of HCV-infected adults who underwent liver stiffness measurement by vibration-controlled transient elastography before and at least once after completing HCV treatment. All studies also included data on median liver stiffness among patients who did and did not achieve SVR. The search identified 23 observational studies and one post hoc analysis of a randomized controlled trial, for a total of 2,934 patients, of whom 2,214 achieved SVR.
Among patients who achieved SVR, mean liver stiffness dropped by 2.4 kPa at the end of treatment (95% CI, 1.7-3.0 kPa), by 3.1 kPa 1-6 months later (95% CI, 1.6-4.7 kPa), and by 3.2 kPa 6-12 months after completing treatment (90% CI, 2.6-3.9 kPa). A year or more after finishing treatment, patients who achieved SVR had a 28% median decrease in liver stiffness (interquartile range, 22%-35%). However, liver stiffness did not significantly change among patients who did not achieve SVR, the reviewers reported.
Mean liver stiffness declined significantly more among patients who received DAAs (4.5 kPa) than among recipients of interferon-based regimens (2.6 kPa; P = .03). However, studies of DAAs included patients with greater liver stiffness at baseline, which could at least partially explain this discrepancy, the investigators said. Baseline cirrhosis also was associated with a greater decline in liver stiffness (mean, 5.1 kPa, vs. 2.8 kPa in patients without cirrhosis; P = .02), as was high baseline alanine aminotransferase level (P less than .01). Among patients whose baseline liver stiffness measurement exceeded 9.5 kPa, 47% had their liver stiffness drop to less than 9.5 kPa after achieving SVR.
Coinfection with HIV did not significantly alter the magnitude of decline in liver stiffness 6-12 months after treatment in patients who achieved SVR, the reviewers noted. “[Follow-up] assessment after SVR was relatively short; hence, long-term evolution of liver stiffness after antiviral therapy and impact of decline in liver stiffness on patient clinical outcomes could not be ascertained,” they wrote. The studies also did not consistently assess potential confounders such as nonalcoholic fatty liver disease, diabetes, and alcohol consumption.
One reviewer disclosed funding from the National Institutes of Health/National Library of Medicine. None had conflicts of interest.
Eradicating chronic hepatitis C virus (HCV) infection led to significant decreases in liver stiffness in a systematic review and meta-analysis of nearly 3,000 patients.
Mean liver stiffness fell by 4.1 kPa (kilopascals) (95% confidence interval, 3.3-4.9 kPa) 12 or more months after patients achieved sustained virologic response to treatment, but did not significantly change in patients who did not achieve SVR, reported Siddharth Singh, MD, of the University of San Diego, La Jolla, Calif., and his associates in the January issue of Clinical Gastroenterology and Hepatology (doi: 10.1016/j.cgh.2017.04.038). The results were especially striking among patients who received direct-acting antiviral agents (DAAs) or who had high baseline levels of inflammation, the investigators added.
SOURCE: AMERICAN GASTROENTEROLOGICAL ASSOCIATION
Based on these findings, about 47% of patients with advanced fibrosis or cirrhosis at baseline will drop below 9.5 kPa after achieving SVR, they reported. “With this decline in liver stiffness, it is conceivable that risk of liver-related complications would decrease, particularly in patients without cirrhosis,” they added. “Future research is warranted on the impact of magnitude and kinetics of decline in liver stiffness on improvement in liver-related outcomes.”
Eradicating HCV infection was known to decrease liver stiffness, but the magnitude of decline was not well understood. Therefore, the reviewers searched the literature through October 2016 for studies of HCV-infected adults who underwent liver stiffness measurement by vibration-controlled transient elastography before and at least once after completing HCV treatment. All studies also included data on median liver stiffness among patients who did and did not achieve SVR. The search identified 23 observational studies and one post hoc analysis of a randomized controlled trial, for a total of 2,934 patients, of whom 2,214 achieved SVR.
Among patients who achieved SVR, mean liver stiffness dropped by 2.4 kPa at the end of treatment (95% CI, 1.7-3.0 kPa), by 3.1 kPa 1-6 months later (95% CI, 1.6-4.7 kPa), and by 3.2 kPa 6-12 months after completing treatment (90% CI, 2.6-3.9 kPa). A year or more after finishing treatment, patients who achieved SVR had a 28% median decrease in liver stiffness (interquartile range, 22%-35%). However, liver stiffness did not significantly change among patients who did not achieve SVR, the reviewers reported.
Mean liver stiffness declined significantly more among patients who received DAAs (4.5 kPa) than among recipients of interferon-based regimens (2.6 kPa; P = .03). However, studies of DAAs included patients with greater liver stiffness at baseline, which could at least partially explain this discrepancy, the investigators said. Baseline cirrhosis also was associated with a greater decline in liver stiffness (mean, 5.1 kPa, vs. 2.8 kPa in patients without cirrhosis; P = .02), as was high baseline alanine aminotransferase level (P less than .01). Among patients whose baseline liver stiffness measurement exceeded 9.5 kPa, 47% had their liver stiffness drop to less than 9.5 kPa after achieving SVR.
Coinfection with HIV did not significantly alter the magnitude of decline in liver stiffness 6-12 months after treatment in patients who achieved SVR, the reviewers noted. “[Follow-up] assessment after SVR was relatively short; hence, long-term evolution of liver stiffness after antiviral therapy and impact of decline in liver stiffness on patient clinical outcomes could not be ascertained,” they wrote. The studies also did not consistently assess potential confounders such as nonalcoholic fatty liver disease, diabetes, and alcohol consumption.
One reviewer disclosed funding from the National Institutes of Health/National Library of Medicine. None had conflicts of interest.
FROM CLINICAL GASTROENTEROLOGY AND HEPATOLOGY
Key clinical point: Eradicating chronic hepatitis C virus infection led to significant decreases in liver stiffness.
Major finding: Mean liver stiffness decreased by 4.1 kPa 12 or more months after patients achieved sustained virologic response to treatment, but did not significantly improve in patients who lacked SVR.
Data source: A systematic review and meta-analysis of 2,934 patients from 23 observational studies and one post hoc analysis of a randomized controlled trial.
Disclosures: One reviewer disclosed funding from the National Institutes of Health/National Library of Medicine. The reviewers reported having no conflicts of interest.
Model validates use of HCV+ livers for transplant
As the evidence supporting the idea of transplanting livers infected with hepatitis C into patients who do not have the disease continues to mount, a multi-institutional team of researchers has developed a mathematical model that shows when hepatitis C–positive-to-negative transplant may improve survival for patients who might otherwise die awaiting a disease-free liver.
In a report published in the journal Hepatology (doi: 10.1002/hep.29723), the researchers noted how direct-acting antivirals (DAAs) have changed the calculus of hepatitis C (HCV) status in liver transplant by reducing the number of HCV-positive patients on the wait list and providing treatment for HCV-negative patients who receive HCV-positive livers. “It is important that further research in this area continues, as we expect that the supply of HCV-positive organs may continue to increase in light of the growing opioid epidemic,” said lead author Jagpreet Chhatwal, PhD, of Massachusetts General Hospital Institute for Technology Assessment in Boston.
Dr. Chhatwal and coauthors claimed their study provides some of the first empirical data for transplanting livers from patients with HCV into patients who do not have the disease.
The researchers performed their analysis using a Markov-based mathematical model known as Simulation of Liver Transplant Candidates (SIM-LT). The model had been validated in previous studies that Dr. Chhatwal and some coauthors had published (Hepatology. 2017;65:777-88; Clin Gastroenterol Hepatol 2018;16:115-22). Dr. Chhatwal and coauthors revised the SIM-LT model to simulate a virtual trial of HCV-negative patients on the liver transplant waiting list to compare outcomes in patients willing to accept any liver to those willing to accept only HCV-negative livers.
The patients willing to receive HCV-positive livers were given 12 weeks of DAA therapy preemptively and had a higher risk of graft failure. The model incorporated data from published studies using the United Network for Organ Sharing (UNOS) and used reported outcomes of the Organ Procurement and Transplantation Network to validate the findings.
The study showed that the clinical benefits of an HCV-negative patient receiving an HCV-positive liver depend on the patient’s Model for End-Stage Liver Disease (MELD) score. Using the measured change in life-years, the researchers found that patients with a MELD score below 20 actually witnessed reduction in life-years when accepting any liver, but that the benefits of accepting any liver started to accrue at MELD score 20. The benefit topped out at MELD 28, with 0.172 life years gained, but even sustained at 0.06 life years gained at MELD 40.
The effectiveness of using HCV-positive livers may also depend on region. UNOS Region 1 – essentially New England minus western Vermont – has the highest rate of HCV-positive organs, and a patient there with MELD 28 would gain 0.36 life-years by accepting any liver regardless of HCV status. However, Region 7 – the Dakotas and upper Midwest plus Illinois – has the lowest HCV-positive organ rate, and a MELD 28 patient there would gain only 0.1 life-year accepting any liver.
“Transplanting HCV-positive livers into HCV-negative patients receiving preemptive DAA therapy could be a viable option for improving patient survival on the LT waiting list, especially in UNOS regions with high HCV-positive donor organ rates,” said Dr. Chhatwal and coauthors. They concluded that their analysis could help direct future clinical trials evaluating the effectiveness of DAA therapy in liver transplant by recognizing patients who could benefit most from accepting HCV-positive donor organs.
The study authors reported having no financial disclosures. The study was supported by grants from the American Cancer Society, Health Resources and Services Administration, National Institutes of Health, National Science Foundation, and Massachusetts General Hospital Research Scholars Program. Coauthor Fasiha Kanwal, MD, received support from the Veterans Administration Health Services, Research & Development Center for Innovations in Quality, Effectiveness and Safety and Public Health Service.
SOURCE: Chhatwal J et al. Hepatology. doi:10.1002/hep.29723.
As the evidence supporting the idea of transplanting livers infected with hepatitis C into patients who do not have the disease continues to mount, a multi-institutional team of researchers has developed a mathematical model that shows when hepatitis C–positive-to-negative transplant may improve survival for patients who might otherwise die awaiting a disease-free liver.
In a report published in the journal Hepatology (doi: 10.1002/hep.29723), the researchers noted how direct-acting antivirals (DAAs) have changed the calculus of hepatitis C (HCV) status in liver transplant by reducing the number of HCV-positive patients on the wait list and providing treatment for HCV-negative patients who receive HCV-positive livers. “It is important that further research in this area continues, as we expect that the supply of HCV-positive organs may continue to increase in light of the growing opioid epidemic,” said lead author Jagpreet Chhatwal, PhD, of Massachusetts General Hospital Institute for Technology Assessment in Boston.
Dr. Chhatwal and coauthors claimed their study provides some of the first empirical data for transplanting livers from patients with HCV into patients who do not have the disease.
The researchers performed their analysis using a Markov-based mathematical model known as Simulation of Liver Transplant Candidates (SIM-LT). The model had been validated in previous studies that Dr. Chhatwal and some coauthors had published (Hepatology. 2017;65:777-88; Clin Gastroenterol Hepatol 2018;16:115-22). Dr. Chhatwal and coauthors revised the SIM-LT model to simulate a virtual trial of HCV-negative patients on the liver transplant waiting list to compare outcomes in patients willing to accept any liver to those willing to accept only HCV-negative livers.
The patients willing to receive HCV-positive livers were given 12 weeks of DAA therapy preemptively and had a higher risk of graft failure. The model incorporated data from published studies using the United Network for Organ Sharing (UNOS) and used reported outcomes of the Organ Procurement and Transplantation Network to validate the findings.
The study showed that the clinical benefits of an HCV-negative patient receiving an HCV-positive liver depend on the patient’s Model for End-Stage Liver Disease (MELD) score. Using the measured change in life-years, the researchers found that patients with a MELD score below 20 actually witnessed reduction in life-years when accepting any liver, but that the benefits of accepting any liver started to accrue at MELD score 20. The benefit topped out at MELD 28, with 0.172 life years gained, but even sustained at 0.06 life years gained at MELD 40.
The effectiveness of using HCV-positive livers may also depend on region. UNOS Region 1 – essentially New England minus western Vermont – has the highest rate of HCV-positive organs, and a patient there with MELD 28 would gain 0.36 life-years by accepting any liver regardless of HCV status. However, Region 7 – the Dakotas and upper Midwest plus Illinois – has the lowest HCV-positive organ rate, and a MELD 28 patient there would gain only 0.1 life-year accepting any liver.
“Transplanting HCV-positive livers into HCV-negative patients receiving preemptive DAA therapy could be a viable option for improving patient survival on the LT waiting list, especially in UNOS regions with high HCV-positive donor organ rates,” said Dr. Chhatwal and coauthors. They concluded that their analysis could help direct future clinical trials evaluating the effectiveness of DAA therapy in liver transplant by recognizing patients who could benefit most from accepting HCV-positive donor organs.
The study authors reported having no financial disclosures. The study was supported by grants from the American Cancer Society, Health Resources and Services Administration, National Institutes of Health, National Science Foundation, and Massachusetts General Hospital Research Scholars Program. Coauthor Fasiha Kanwal, MD, received support from the Veterans Administration Health Services, Research & Development Center for Innovations in Quality, Effectiveness and Safety and Public Health Service.
SOURCE: Chhatwal J et al. Hepatology. doi:10.1002/hep.29723.
As the evidence supporting the idea of transplanting livers infected with hepatitis C into patients who do not have the disease continues to mount, a multi-institutional team of researchers has developed a mathematical model that shows when hepatitis C–positive-to-negative transplant may improve survival for patients who might otherwise die awaiting a disease-free liver.
In a report published in the journal Hepatology (doi: 10.1002/hep.29723), the researchers noted how direct-acting antivirals (DAAs) have changed the calculus of hepatitis C (HCV) status in liver transplant by reducing the number of HCV-positive patients on the wait list and providing treatment for HCV-negative patients who receive HCV-positive livers. “It is important that further research in this area continues, as we expect that the supply of HCV-positive organs may continue to increase in light of the growing opioid epidemic,” said lead author Jagpreet Chhatwal, PhD, of Massachusetts General Hospital Institute for Technology Assessment in Boston.
Dr. Chhatwal and coauthors claimed their study provides some of the first empirical data for transplanting livers from patients with HCV into patients who do not have the disease.
The researchers performed their analysis using a Markov-based mathematical model known as Simulation of Liver Transplant Candidates (SIM-LT). The model had been validated in previous studies that Dr. Chhatwal and some coauthors had published (Hepatology. 2017;65:777-88; Clin Gastroenterol Hepatol 2018;16:115-22). Dr. Chhatwal and coauthors revised the SIM-LT model to simulate a virtual trial of HCV-negative patients on the liver transplant waiting list to compare outcomes in patients willing to accept any liver to those willing to accept only HCV-negative livers.
The patients willing to receive HCV-positive livers were given 12 weeks of DAA therapy preemptively and had a higher risk of graft failure. The model incorporated data from published studies using the United Network for Organ Sharing (UNOS) and used reported outcomes of the Organ Procurement and Transplantation Network to validate the findings.
The study showed that the clinical benefits of an HCV-negative patient receiving an HCV-positive liver depend on the patient’s Model for End-Stage Liver Disease (MELD) score. Using the measured change in life-years, the researchers found that patients with a MELD score below 20 actually witnessed reduction in life-years when accepting any liver, but that the benefits of accepting any liver started to accrue at MELD score 20. The benefit topped out at MELD 28, with 0.172 life years gained, but even sustained at 0.06 life years gained at MELD 40.
The effectiveness of using HCV-positive livers may also depend on region. UNOS Region 1 – essentially New England minus western Vermont – has the highest rate of HCV-positive organs, and a patient there with MELD 28 would gain 0.36 life-years by accepting any liver regardless of HCV status. However, Region 7 – the Dakotas and upper Midwest plus Illinois – has the lowest HCV-positive organ rate, and a MELD 28 patient there would gain only 0.1 life-year accepting any liver.
“Transplanting HCV-positive livers into HCV-negative patients receiving preemptive DAA therapy could be a viable option for improving patient survival on the LT waiting list, especially in UNOS regions with high HCV-positive donor organ rates,” said Dr. Chhatwal and coauthors. They concluded that their analysis could help direct future clinical trials evaluating the effectiveness of DAA therapy in liver transplant by recognizing patients who could benefit most from accepting HCV-positive donor organs.
The study authors reported having no financial disclosures. The study was supported by grants from the American Cancer Society, Health Resources and Services Administration, National Institutes of Health, National Science Foundation, and Massachusetts General Hospital Research Scholars Program. Coauthor Fasiha Kanwal, MD, received support from the Veterans Administration Health Services, Research & Development Center for Innovations in Quality, Effectiveness and Safety and Public Health Service.
SOURCE: Chhatwal J et al. Hepatology. doi:10.1002/hep.29723.
FROM HEPATOLOGY
Key clinical point: Making hepatitis C virus–positive livers available to HCV-negative patients awaiting liver transplant could improve survival of patients on the liver transplant waiting list.
Major finding: Patients with a Model for End-Stage Liver Disease score of 28 willing to receive any liver gained 0.172 life-years.
Data source: Simulated trial using Markov-based mathematical model and data from published studies and the United Network for Organ Sharing.
Disclosures: Dr. Chhatwal and coauthors reported having no financial disclosures. The study was supported by grants from the American Cancer Society, Health Resources and Services Administration, National Institutes of Health, National Science Foundation, and Massachusetts General Hospital Research Scholars Program. Coauthor Fasiha Kanwal, MD, received support from the Veterans Administration Health Services, Research & Development Center for Innovations in Quality, Effectiveness and Safety and Public Health Service.
Source: Chhatwal J et al. Hepatology. doi:10.1002/hep.29723.
Risks identified for drug-resistant bacteremia in cirrhosis
In patients hospitalized with cirrhosis, biliary cirrhosis, recent health care exposure, nonwhite race, and cultures taken more than 48 hours after admission all independently predicted that bacteremia would be caused by multidrug-resistant organisms (MDROs), according to a medical record review at CHI St. Luke’s Medical Center, an 850-bed tertiary care center in Houston.
“These variables along with severity of infection and liver disease may help clinicians identify patients who will benefit most from broader-spectrum empiric antimicrobial therapy,” wrote the investigators, led by Jennifer Addo Smith, PharmD, of St. Luke’s, in the Journal of Clinical Gastroenterology.
But local epidemiology remains important. “Although a gram-positive agent (e.g., vancomycin) and a carbapenem-sparing gram-negative agent (e.g., ceftriaxone, cefepime) are reasonable empiric agents at our center, other centers with different resistance patterns may warrant different empiric therapy. Given the low prevalence of VRE [vancomycin-resistant Enterococcus] in this study ... and E. faecium in other studies (4%-7%), an empiric agent active against VRE does not seem to be routinely required,” they said.
The team looked into the issue because there hasn’t been much investigation in the United States of the role of multidrug resistant organisms in bacteremia among patients hospitalized with cirrhosis.
Thirty patients in the study had bacteremia caused by MDROs while 60 had bacteremia from non-MDROs, giving a 33% prevalence of MDRO bacteremia, which was consistent with previous, mostly European studies.
Enterobacteriaceae (43%), Staphylococcus aureus (18%), Streptococcus spp. (11%), Enterococcus spp. (10%), and nonfermenting gram-negative bacilli (6%) were the main causes of bacteremia overall.
Among the 30 MDRO cases, methicillin-resistant S. aureus was isolated in seven (23%); methicillin-resistant coagulase-negative Staphylococci in four (13%); fluoroquinolone-resistant Enterobacteriaceae in nine (30%); extended spectrum beta-lactamase–producing Enterobacteriaceae in three (10%), and VRE in two (7%). No carbapenemase-producing gram-negative bacteria were identified.
The predictors of MDRO bacteremia emerged on multivariate analysis and included biliary cirrhosis (adjusted odds ratio, 11.75; 95% confidence interval, 2.08-66.32); recent health care exposure (aOR, 9.81; 95% CI, 2.15-44.88); blood cultures obtained 48 hours after hospital admission (aOR, 6.02; 95% CI, 1.70-21.40) and nonwhite race (aOR , 3.35; 95% CI, 1.19-9.38).
Blood cultures past 48 hours and recent health care exposure – generally hospitalization within the past 90 days – were likely surrogates for nosocomial infection.
The link with biliary cirrhosis is unclear. “Compared with other cirrhotic patients, perhaps patients with PBC [primary biliary cholangitis] have had more cumulative antimicrobial exposure because of [their] higher risk for UTIs [urinary tract infections] and therefore are at increased risk for MDROs,” they wrote.
The median age in the study was 59 years. Half of the patients were white; 46% were women. Hepatitis C was the most common cause of cirrhosis, followed by alcohol.
MDRO was defined in the study as bacteria not susceptible to at least one antibiotic in at least three antimicrobial categories; 90 cirrhosis patients without bacteremia served as controls.
The funding source was not reported. Dr. Addo Smith had no disclosures.
SOURCE: Smith JA et al. J Clin Gastroenterol. 2017 Nov 23. doi: 10.1097/MCG.0000000000000964.
*This story was updated on 1/10/2018.
In patients hospitalized with cirrhosis, biliary cirrhosis, recent health care exposure, nonwhite race, and cultures taken more than 48 hours after admission all independently predicted that bacteremia would be caused by multidrug-resistant organisms (MDROs), according to a medical record review at CHI St. Luke’s Medical Center, an 850-bed tertiary care center in Houston.
“These variables along with severity of infection and liver disease may help clinicians identify patients who will benefit most from broader-spectrum empiric antimicrobial therapy,” wrote the investigators, led by Jennifer Addo Smith, PharmD, of St. Luke’s, in the Journal of Clinical Gastroenterology.
But local epidemiology remains important. “Although a gram-positive agent (e.g., vancomycin) and a carbapenem-sparing gram-negative agent (e.g., ceftriaxone, cefepime) are reasonable empiric agents at our center, other centers with different resistance patterns may warrant different empiric therapy. Given the low prevalence of VRE [vancomycin-resistant Enterococcus] in this study ... and E. faecium in other studies (4%-7%), an empiric agent active against VRE does not seem to be routinely required,” they said.
The team looked into the issue because there hasn’t been much investigation in the United States of the role of multidrug resistant organisms in bacteremia among patients hospitalized with cirrhosis.
Thirty patients in the study had bacteremia caused by MDROs while 60 had bacteremia from non-MDROs, giving a 33% prevalence of MDRO bacteremia, which was consistent with previous, mostly European studies.
Enterobacteriaceae (43%), Staphylococcus aureus (18%), Streptococcus spp. (11%), Enterococcus spp. (10%), and nonfermenting gram-negative bacilli (6%) were the main causes of bacteremia overall.
Among the 30 MDRO cases, methicillin-resistant S. aureus was isolated in seven (23%); methicillin-resistant coagulase-negative Staphylococci in four (13%); fluoroquinolone-resistant Enterobacteriaceae in nine (30%); extended spectrum beta-lactamase–producing Enterobacteriaceae in three (10%), and VRE in two (7%). No carbapenemase-producing gram-negative bacteria were identified.
The predictors of MDRO bacteremia emerged on multivariate analysis and included biliary cirrhosis (adjusted odds ratio, 11.75; 95% confidence interval, 2.08-66.32); recent health care exposure (aOR, 9.81; 95% CI, 2.15-44.88); blood cultures obtained 48 hours after hospital admission (aOR, 6.02; 95% CI, 1.70-21.40) and nonwhite race (aOR , 3.35; 95% CI, 1.19-9.38).
Blood cultures past 48 hours and recent health care exposure – generally hospitalization within the past 90 days – were likely surrogates for nosocomial infection.
The link with biliary cirrhosis is unclear. “Compared with other cirrhotic patients, perhaps patients with PBC [primary biliary cholangitis] have had more cumulative antimicrobial exposure because of [their] higher risk for UTIs [urinary tract infections] and therefore are at increased risk for MDROs,” they wrote.
The median age in the study was 59 years. Half of the patients were white; 46% were women. Hepatitis C was the most common cause of cirrhosis, followed by alcohol.
MDRO was defined in the study as bacteria not susceptible to at least one antibiotic in at least three antimicrobial categories; 90 cirrhosis patients without bacteremia served as controls.
The funding source was not reported. Dr. Addo Smith had no disclosures.
SOURCE: Smith JA et al. J Clin Gastroenterol. 2017 Nov 23. doi: 10.1097/MCG.0000000000000964.
*This story was updated on 1/10/2018.
In patients hospitalized with cirrhosis, biliary cirrhosis, recent health care exposure, nonwhite race, and cultures taken more than 48 hours after admission all independently predicted that bacteremia would be caused by multidrug-resistant organisms (MDROs), according to a medical record review at CHI St. Luke’s Medical Center, an 850-bed tertiary care center in Houston.
“These variables along with severity of infection and liver disease may help clinicians identify patients who will benefit most from broader-spectrum empiric antimicrobial therapy,” wrote the investigators, led by Jennifer Addo Smith, PharmD, of St. Luke’s, in the Journal of Clinical Gastroenterology.
But local epidemiology remains important. “Although a gram-positive agent (e.g., vancomycin) and a carbapenem-sparing gram-negative agent (e.g., ceftriaxone, cefepime) are reasonable empiric agents at our center, other centers with different resistance patterns may warrant different empiric therapy. Given the low prevalence of VRE [vancomycin-resistant Enterococcus] in this study ... and E. faecium in other studies (4%-7%), an empiric agent active against VRE does not seem to be routinely required,” they said.
The team looked into the issue because there hasn’t been much investigation in the United States of the role of multidrug resistant organisms in bacteremia among patients hospitalized with cirrhosis.
Thirty patients in the study had bacteremia caused by MDROs while 60 had bacteremia from non-MDROs, giving a 33% prevalence of MDRO bacteremia, which was consistent with previous, mostly European studies.
Enterobacteriaceae (43%), Staphylococcus aureus (18%), Streptococcus spp. (11%), Enterococcus spp. (10%), and nonfermenting gram-negative bacilli (6%) were the main causes of bacteremia overall.
Among the 30 MDRO cases, methicillin-resistant S. aureus was isolated in seven (23%); methicillin-resistant coagulase-negative Staphylococci in four (13%); fluoroquinolone-resistant Enterobacteriaceae in nine (30%); extended spectrum beta-lactamase–producing Enterobacteriaceae in three (10%), and VRE in two (7%). No carbapenemase-producing gram-negative bacteria were identified.
The predictors of MDRO bacteremia emerged on multivariate analysis and included biliary cirrhosis (adjusted odds ratio, 11.75; 95% confidence interval, 2.08-66.32); recent health care exposure (aOR, 9.81; 95% CI, 2.15-44.88); blood cultures obtained 48 hours after hospital admission (aOR, 6.02; 95% CI, 1.70-21.40) and nonwhite race (aOR , 3.35; 95% CI, 1.19-9.38).
Blood cultures past 48 hours and recent health care exposure – generally hospitalization within the past 90 days – were likely surrogates for nosocomial infection.
The link with biliary cirrhosis is unclear. “Compared with other cirrhotic patients, perhaps patients with PBC [primary biliary cholangitis] have had more cumulative antimicrobial exposure because of [their] higher risk for UTIs [urinary tract infections] and therefore are at increased risk for MDROs,” they wrote.
The median age in the study was 59 years. Half of the patients were white; 46% were women. Hepatitis C was the most common cause of cirrhosis, followed by alcohol.
MDRO was defined in the study as bacteria not susceptible to at least one antibiotic in at least three antimicrobial categories; 90 cirrhosis patients without bacteremia served as controls.
The funding source was not reported. Dr. Addo Smith had no disclosures.
SOURCE: Smith JA et al. J Clin Gastroenterol. 2017 Nov 23. doi: 10.1097/MCG.0000000000000964.
*This story was updated on 1/10/2018.
FROM THE JOURNAL OF CLINICAL GASTROENTEROLOGY
Key clinical point: In patients hospitalized with cirrhosis, nonwhite race, biliary involvement, recent health care exposure, and cultures taken more than 48 hours after hospital admission all independently predicted that bacteremia would be caused by multidrug-resistant organisms.
Major finding: The predictors of multidrug-resistant organism bacteremia emerged on multivariate analysis and included biliary cirrhosis (aOR 11.75; 95% CI, 2.08-66.32); recent health care exposure (aOR 9.81; 95% CI, 2.15-44.88); and blood cultures obtained 48 hours after hospital admission (aOR 6.02; 95% CI, 1.70-21.40).
Study details: Review of 90 cirrhotic patients with bacteremia, plus 90 controls.
Disclosures: The lead investigator had no disclosures.
Source: Smith JA et al. J Clin Gastroenterol. 2017 Nov 23. doi: 10.1097/MCG.0000000000000964.
Amyloid depleter/antibody duo reduces systemic amyloidosis severity
A one-two combination consisting of a drug to deplete amyloid fibrils from circulation and a monoclonal antibody to mop up residual amyloid protein appears to be safe and shows promising activity against systemic amyloidosis in an early clinical trial.
Pairing the investigational small molecule agent miridesap and the experimental monoclonal antibody dezamizumab resulted in clearance or reductions in amyloid deposits in the livers, spleens, and kidneys of adults with systemic amyloidosis, reported Prof. Sir Mark B. Pepys of University College, London, and his colleagues.
Systemic amyloidosis is caused by extracellular deposition of amyloid fibrils coated with serum amyloid P component (SAP), a normal plasma protein. Miridesap has been shown to deplete levels of circulating SAP, but fails to clear SAP lingering in amyloid deposits. Dezamizumab is a fully humanized immunoglobulin G1 antibody that is directed against SAP and provokes immune system clearance of residual protein.
The investigators enrolled 23 adults with systemic amyloidosis into an open-label, nonrandomized trial of safety, pharmacokinetics, and dose responses to up to three cycles of miridesap followed by dezamizumab. They used scintigraphy with radiolabeling to measure amyloid burden, equilibrium MRI to measure organ extracellular volume, and transient elastography to evaluate liver stiffness.
They found that a 2,000-mg dose of dezamizumab reduced amyloid load in the liver and/or spleen in five of seven patients, and that liver stiffness, a sensitive marker for hepatic amyloid load, initially increased in some patients, but then fell “substantially,” suggesting amyloid clearance and resolution of cellular infiltrates. Progressive dose-related clearance of amyloid in the liver was also associated with improvements in liver function tests.
In addition, 7 of 11 patients had reductions in renal amyloid burden, with the greatest benefit seen in patients treated with higher doses of the antibody.
The trial cohort also included six patients with clinical cardiac amyloidosis: three with monoclonal immunoglobulin light-chain amyloidosis (AL), and three with ATTR (transthyretin) amyloidosis. Although the investigators had excluded patients with cardiac amyloidosis in the first part of the study out of safety concerns, there were no new arrhythmias and no increases in cardiac troponin T or creatine kinase concentrations. One patient had a 17% reduction in left ventricular mass, suggesting a reduction in cardiac amyloid burden.
Patients tolerated the treatment, with self-limiting, early-onset rashes after higher antibody doses being the most frequent adverse event.
“However, the identified and potential risks of the intervention are considered manageable, and thus acceptable, in relation to the demonstrable clinical benefits of removing amyloid from vital organs,” the investigators wrote.
GlaxoSmithKline funded the study. Three of the coauthors are GSK employees and stockholders. Prof. Pepys is the inventor on patents for miridesap and miridesap plus anti-SAP antibody.
Source: Richards D et al. Sci. Transl. Med. 2018. doi: 10.1126/scitranslmed.aan3128.
A one-two combination consisting of a drug to deplete amyloid fibrils from circulation and a monoclonal antibody to mop up residual amyloid protein appears to be safe and shows promising activity against systemic amyloidosis in an early clinical trial.
Pairing the investigational small molecule agent miridesap and the experimental monoclonal antibody dezamizumab resulted in clearance or reductions in amyloid deposits in the livers, spleens, and kidneys of adults with systemic amyloidosis, reported Prof. Sir Mark B. Pepys of University College, London, and his colleagues.
Systemic amyloidosis is caused by extracellular deposition of amyloid fibrils coated with serum amyloid P component (SAP), a normal plasma protein. Miridesap has been shown to deplete levels of circulating SAP, but fails to clear SAP lingering in amyloid deposits. Dezamizumab is a fully humanized immunoglobulin G1 antibody that is directed against SAP and provokes immune system clearance of residual protein.
The investigators enrolled 23 adults with systemic amyloidosis into an open-label, nonrandomized trial of safety, pharmacokinetics, and dose responses to up to three cycles of miridesap followed by dezamizumab. They used scintigraphy with radiolabeling to measure amyloid burden, equilibrium MRI to measure organ extracellular volume, and transient elastography to evaluate liver stiffness.
They found that a 2,000-mg dose of dezamizumab reduced amyloid load in the liver and/or spleen in five of seven patients, and that liver stiffness, a sensitive marker for hepatic amyloid load, initially increased in some patients, but then fell “substantially,” suggesting amyloid clearance and resolution of cellular infiltrates. Progressive dose-related clearance of amyloid in the liver was also associated with improvements in liver function tests.
In addition, 7 of 11 patients had reductions in renal amyloid burden, with the greatest benefit seen in patients treated with higher doses of the antibody.
The trial cohort also included six patients with clinical cardiac amyloidosis: three with monoclonal immunoglobulin light-chain amyloidosis (AL), and three with ATTR (transthyretin) amyloidosis. Although the investigators had excluded patients with cardiac amyloidosis in the first part of the study out of safety concerns, there were no new arrhythmias and no increases in cardiac troponin T or creatine kinase concentrations. One patient had a 17% reduction in left ventricular mass, suggesting a reduction in cardiac amyloid burden.
Patients tolerated the treatment, with self-limiting, early-onset rashes after higher antibody doses being the most frequent adverse event.
“However, the identified and potential risks of the intervention are considered manageable, and thus acceptable, in relation to the demonstrable clinical benefits of removing amyloid from vital organs,” the investigators wrote.
GlaxoSmithKline funded the study. Three of the coauthors are GSK employees and stockholders. Prof. Pepys is the inventor on patents for miridesap and miridesap plus anti-SAP antibody.
Source: Richards D et al. Sci. Transl. Med. 2018. doi: 10.1126/scitranslmed.aan3128.
A one-two combination consisting of a drug to deplete amyloid fibrils from circulation and a monoclonal antibody to mop up residual amyloid protein appears to be safe and shows promising activity against systemic amyloidosis in an early clinical trial.
Pairing the investigational small molecule agent miridesap and the experimental monoclonal antibody dezamizumab resulted in clearance or reductions in amyloid deposits in the livers, spleens, and kidneys of adults with systemic amyloidosis, reported Prof. Sir Mark B. Pepys of University College, London, and his colleagues.
Systemic amyloidosis is caused by extracellular deposition of amyloid fibrils coated with serum amyloid P component (SAP), a normal plasma protein. Miridesap has been shown to deplete levels of circulating SAP, but fails to clear SAP lingering in amyloid deposits. Dezamizumab is a fully humanized immunoglobulin G1 antibody that is directed against SAP and provokes immune system clearance of residual protein.
The investigators enrolled 23 adults with systemic amyloidosis into an open-label, nonrandomized trial of safety, pharmacokinetics, and dose responses to up to three cycles of miridesap followed by dezamizumab. They used scintigraphy with radiolabeling to measure amyloid burden, equilibrium MRI to measure organ extracellular volume, and transient elastography to evaluate liver stiffness.
They found that a 2,000-mg dose of dezamizumab reduced amyloid load in the liver and/or spleen in five of seven patients, and that liver stiffness, a sensitive marker for hepatic amyloid load, initially increased in some patients, but then fell “substantially,” suggesting amyloid clearance and resolution of cellular infiltrates. Progressive dose-related clearance of amyloid in the liver was also associated with improvements in liver function tests.
In addition, 7 of 11 patients had reductions in renal amyloid burden, with the greatest benefit seen in patients treated with higher doses of the antibody.
The trial cohort also included six patients with clinical cardiac amyloidosis: three with monoclonal immunoglobulin light-chain amyloidosis (AL), and three with ATTR (transthyretin) amyloidosis. Although the investigators had excluded patients with cardiac amyloidosis in the first part of the study out of safety concerns, there were no new arrhythmias and no increases in cardiac troponin T or creatine kinase concentrations. One patient had a 17% reduction in left ventricular mass, suggesting a reduction in cardiac amyloid burden.
Patients tolerated the treatment, with self-limiting, early-onset rashes after higher antibody doses being the most frequent adverse event.
“However, the identified and potential risks of the intervention are considered manageable, and thus acceptable, in relation to the demonstrable clinical benefits of removing amyloid from vital organs,” the investigators wrote.
GlaxoSmithKline funded the study. Three of the coauthors are GSK employees and stockholders. Prof. Pepys is the inventor on patents for miridesap and miridesap plus anti-SAP antibody.
Source: Richards D et al. Sci. Transl. Med. 2018. doi: 10.1126/scitranslmed.aan3128.
FROM SCIENCE TRANSLATIONAL MEDICINE
Key clinical point: A combination of an amyloid-depleting agent and immunotherapeutic antibody appears effective and safe in patients with systemic amyloidosis.
Major finding: A 2,000-mg dose of dezamizumab reduced amyloid load in the liver and/or spleen in five of seven patients.
Data source: Open-label, nonrandomized clinical trial of 23 adults with various subtypes of systemic amyloidosis.
Disclosures: GlaxoSmithKline funded the study. Three of the coauthors are GSK employees and stockholders. Prof. Pepys is the inventor on patents for miridesap and miridesap plus anti-SAP antibody.
Source: Richards D et al. Sci. Transl. Med. 2018;10:eaan3128.
PBC disease progression no worse for patients with concomitant NAFLD
, according to Gerald Yosel Minuk, MD, and his associates.
At baseline, the 168 patients in the PBC-only group had higher serum alkaline phosphatase and gamma-glutamyl transferase values than the 68 patients in the NAFLD/PBC group, as well as having higher FIB-4 scores. The percentage of patients with aspartate aminotransferase/platelet ratio indexes (APRI) greater than 1.5 was slightly higher in the PBC-only group, but the difference was not significant.
After follow-up periods averaging 6.7 years in the PBC-only group and 6.4 years in the NAFLD/PBC group, yearly increases in FIB-4 and prevalence of APRI greater than 1.5 were greater in the PBC-only group, though the difference did not reach significance. PBC-only patients were more likely to have developed radiologic evidence of cirrhosis during the follow-up period (42% vs. 19%, P less than .001).
“Were the results of the present study to be confirmed by others, the question arises as to why NAFLD does not adversely and may favorably impact on PBC. Here, it is tempting to speculate that because PBC livers are associated with a paucity of immunosuppressive regulator T cells (Tregs) whereas in NAFLD, Tregs are recruited to the liver in increased numbers, a restoration of the immune balance in PBC livers might explain these findings,” the investigators noted.
Find the full study in Liver International (doi: 10.1111/liv.13644).
, according to Gerald Yosel Minuk, MD, and his associates.
At baseline, the 168 patients in the PBC-only group had higher serum alkaline phosphatase and gamma-glutamyl transferase values than the 68 patients in the NAFLD/PBC group, as well as having higher FIB-4 scores. The percentage of patients with aspartate aminotransferase/platelet ratio indexes (APRI) greater than 1.5 was slightly higher in the PBC-only group, but the difference was not significant.
After follow-up periods averaging 6.7 years in the PBC-only group and 6.4 years in the NAFLD/PBC group, yearly increases in FIB-4 and prevalence of APRI greater than 1.5 were greater in the PBC-only group, though the difference did not reach significance. PBC-only patients were more likely to have developed radiologic evidence of cirrhosis during the follow-up period (42% vs. 19%, P less than .001).
“Were the results of the present study to be confirmed by others, the question arises as to why NAFLD does not adversely and may favorably impact on PBC. Here, it is tempting to speculate that because PBC livers are associated with a paucity of immunosuppressive regulator T cells (Tregs) whereas in NAFLD, Tregs are recruited to the liver in increased numbers, a restoration of the immune balance in PBC livers might explain these findings,” the investigators noted.
Find the full study in Liver International (doi: 10.1111/liv.13644).
, according to Gerald Yosel Minuk, MD, and his associates.
At baseline, the 168 patients in the PBC-only group had higher serum alkaline phosphatase and gamma-glutamyl transferase values than the 68 patients in the NAFLD/PBC group, as well as having higher FIB-4 scores. The percentage of patients with aspartate aminotransferase/platelet ratio indexes (APRI) greater than 1.5 was slightly higher in the PBC-only group, but the difference was not significant.
After follow-up periods averaging 6.7 years in the PBC-only group and 6.4 years in the NAFLD/PBC group, yearly increases in FIB-4 and prevalence of APRI greater than 1.5 were greater in the PBC-only group, though the difference did not reach significance. PBC-only patients were more likely to have developed radiologic evidence of cirrhosis during the follow-up period (42% vs. 19%, P less than .001).
“Were the results of the present study to be confirmed by others, the question arises as to why NAFLD does not adversely and may favorably impact on PBC. Here, it is tempting to speculate that because PBC livers are associated with a paucity of immunosuppressive regulator T cells (Tregs) whereas in NAFLD, Tregs are recruited to the liver in increased numbers, a restoration of the immune balance in PBC livers might explain these findings,” the investigators noted.
Find the full study in Liver International (doi: 10.1111/liv.13644).
FROM LIVER INTERNATIONAL