User login
Model reveals how to target cancer’s weaknesses

Credit: PNAS
A new model suggests we should be targeting cancers’ weaknesses instead of their strengths.
An article in BioEssays proposes that cancers form when recently evolved genes are damaged, and cancer cells have to revert to using older, inappropriate genetic pathways.
So we should create treatments that take advantage of capabilities humans have developed more recently—such as the adaptive immune system—instead of trying to target older capabilities—such as the innate immune system and cell proliferation.
“The rapid proliferation of cancer cells is an ancient, default capability that became regulated during the evolution of multicellularity about a billion years ago,” said study author Charley Lineweaver, PhD, of The Australian National University in Canberra.
“Our model suggests that cancer progression is the accumulation of damage to the more recently acquired genes. Without the regulation of these recent genes, cell physiology reverts to earlier programs, such as unregulated cell proliferation.”
To develop their model, Dr Lineweaver and his colleagues turned to knowledge uncovered by genome sequencing in a range of our distant relatives, including fish, coral, and sponges.
This knowledge has allowed scientists to establish the order in which genes evolved and is the basis of the new therapeutic implications of the model, Dr Lineweaver said.
He noted that the standard model of cancer development suggests that selection produces the acquired capabilities of cancer—such as sustained proliferative signaling and evading apoptosis—and they evolve during the lifetime of the patient.
But Dr Lineweaver’s model suggests the capabilities of cancer are acquired atavisms. They are activated during early embryogenesis and wound healing and reactivated inappropriately during carcinogenesis.
The most recent capabilities—mammalian and vertebrate capabilities—are the least entrenched in cancer. So they should be targeted with therapy.
The older capabilities—last eukaryotic common ancestor (LECA) capabilities, stem eukaryote capabilities, and the earliest evolved capabilities—are maintained in cancer and are therefore difficult to target.
For example, some human ATP binding cassette (ABC) transporters are ancient, and some are quite recent. Dr Lineweaver and his colleagues found that older ABC proteins were more likely to be active in cancer.
So the researchers believe we should create treatments that can be expelled by the newer ABC transporters. That way, normal cells will expel the treatment, but cancer cells will not.
Another potential treatment avenue, according to Dr Lineweaver, is targeting the adaptive immune system.
“The adaptive immune system that humans have has evolved relatively recently, and it seems cancer cells do not have the ability to talk to and be protected by it,” he noted.
“The new therapeutic strategies we are proposing target these weaknesses. These strategies are very different from current therapies, which attack cancer’s strength—its ability to proliferate rapidly.” ![]()

Credit: PNAS
A new model suggests we should be targeting cancers’ weaknesses instead of their strengths.
An article in BioEssays proposes that cancers form when recently evolved genes are damaged, and cancer cells have to revert to using older, inappropriate genetic pathways.
So we should create treatments that take advantage of capabilities humans have developed more recently—such as the adaptive immune system—instead of trying to target older capabilities—such as the innate immune system and cell proliferation.
“The rapid proliferation of cancer cells is an ancient, default capability that became regulated during the evolution of multicellularity about a billion years ago,” said study author Charley Lineweaver, PhD, of The Australian National University in Canberra.
“Our model suggests that cancer progression is the accumulation of damage to the more recently acquired genes. Without the regulation of these recent genes, cell physiology reverts to earlier programs, such as unregulated cell proliferation.”
To develop their model, Dr Lineweaver and his colleagues turned to knowledge uncovered by genome sequencing in a range of our distant relatives, including fish, coral, and sponges.
This knowledge has allowed scientists to establish the order in which genes evolved and is the basis of the new therapeutic implications of the model, Dr Lineweaver said.
He noted that the standard model of cancer development suggests that selection produces the acquired capabilities of cancer—such as sustained proliferative signaling and evading apoptosis—and they evolve during the lifetime of the patient.
But Dr Lineweaver’s model suggests the capabilities of cancer are acquired atavisms. They are activated during early embryogenesis and wound healing and reactivated inappropriately during carcinogenesis.
The most recent capabilities—mammalian and vertebrate capabilities—are the least entrenched in cancer. So they should be targeted with therapy.
The older capabilities—last eukaryotic common ancestor (LECA) capabilities, stem eukaryote capabilities, and the earliest evolved capabilities—are maintained in cancer and are therefore difficult to target.
For example, some human ATP binding cassette (ABC) transporters are ancient, and some are quite recent. Dr Lineweaver and his colleagues found that older ABC proteins were more likely to be active in cancer.
So the researchers believe we should create treatments that can be expelled by the newer ABC transporters. That way, normal cells will expel the treatment, but cancer cells will not.
Another potential treatment avenue, according to Dr Lineweaver, is targeting the adaptive immune system.
“The adaptive immune system that humans have has evolved relatively recently, and it seems cancer cells do not have the ability to talk to and be protected by it,” he noted.
“The new therapeutic strategies we are proposing target these weaknesses. These strategies are very different from current therapies, which attack cancer’s strength—its ability to proliferate rapidly.” ![]()

Credit: PNAS
A new model suggests we should be targeting cancers’ weaknesses instead of their strengths.
An article in BioEssays proposes that cancers form when recently evolved genes are damaged, and cancer cells have to revert to using older, inappropriate genetic pathways.
So we should create treatments that take advantage of capabilities humans have developed more recently—such as the adaptive immune system—instead of trying to target older capabilities—such as the innate immune system and cell proliferation.
“The rapid proliferation of cancer cells is an ancient, default capability that became regulated during the evolution of multicellularity about a billion years ago,” said study author Charley Lineweaver, PhD, of The Australian National University in Canberra.
“Our model suggests that cancer progression is the accumulation of damage to the more recently acquired genes. Without the regulation of these recent genes, cell physiology reverts to earlier programs, such as unregulated cell proliferation.”
To develop their model, Dr Lineweaver and his colleagues turned to knowledge uncovered by genome sequencing in a range of our distant relatives, including fish, coral, and sponges.
This knowledge has allowed scientists to establish the order in which genes evolved and is the basis of the new therapeutic implications of the model, Dr Lineweaver said.
He noted that the standard model of cancer development suggests that selection produces the acquired capabilities of cancer—such as sustained proliferative signaling and evading apoptosis—and they evolve during the lifetime of the patient.
But Dr Lineweaver’s model suggests the capabilities of cancer are acquired atavisms. They are activated during early embryogenesis and wound healing and reactivated inappropriately during carcinogenesis.
The most recent capabilities—mammalian and vertebrate capabilities—are the least entrenched in cancer. So they should be targeted with therapy.
The older capabilities—last eukaryotic common ancestor (LECA) capabilities, stem eukaryote capabilities, and the earliest evolved capabilities—are maintained in cancer and are therefore difficult to target.
For example, some human ATP binding cassette (ABC) transporters are ancient, and some are quite recent. Dr Lineweaver and his colleagues found that older ABC proteins were more likely to be active in cancer.
So the researchers believe we should create treatments that can be expelled by the newer ABC transporters. That way, normal cells will expel the treatment, but cancer cells will not.
Another potential treatment avenue, according to Dr Lineweaver, is targeting the adaptive immune system.
“The adaptive immune system that humans have has evolved relatively recently, and it seems cancer cells do not have the ability to talk to and be protected by it,” he noted.
“The new therapeutic strategies we are proposing target these weaknesses. These strategies are very different from current therapies, which attack cancer’s strength—its ability to proliferate rapidly.” ![]()
New insight into MYC-induced lymphoma

Credit: Juha Klefstrom
Investigators have identified biological signatures in lymphoma cells that can be traced back to the original oncogene.
The team analyzed mouse models and patient samples of MYC-induced lymphoma. And they discovered lipid signatures that corresponded with the level of MYC expression.
The investigators believe this discovery could be the first step toward developing a technique to identify the origin of lymphomas and other malignancies.
They described their discovery in PNAS.
“The same cancer can occur because of different genes, but, in certain cases, the aggressiveness and the type of treatment actually depend a lot on what oncogene caused that cancer,” said study author Livia Eberlin, PhD, of Stanford University in California.
With that in mind, she and her colleagues looked at MYC, an oncogene that’s responsible for approximately half of all human cancers. They wanted to find a biological signature that would trace the mutating cancer cells back to the original oncogene.
“When cancer takes place, the cell loves to gobble up glucose—that’s a sugar—and glutamine,” said Richard Zare, PhD, also of Stanford. “It takes those and makes different lipids—different fatty molecules than what it normally makes.”
So the investigators set out to evaluate changes in lipid profiles in MYC-induced lymphoma. They compared lipid signatures in MYC-induced transgenic mouse models to those in normal control mice.
The team identified 104 molecular ions that were either increased or decreased in the MYC lymphoma models compared to controls. And 86 of these ions were complex phospholipids.
Most of the lipids that were increased in lymphoma were glycerophosphoglycerols and cardiolipins, with a higher content of monounsaturated fatty acids when compared with controls.
To determine if these findings might also apply to humans, the investigators examined 15 samples from lymphoma patients.
The samples had varying expression levels of MYC oncoprotein, and the team observed distinct lipid profiles in lymphomas with high and low MYC expression. This included many of the lipid species they had identified in the animal models of MYC-induced lymphoma.
The investigators said their results suggest a relationship between specific lipid species and the overexpression of MYC. And this information could have both diagnostic and prognostic applications. ![]()

Credit: Juha Klefstrom
Investigators have identified biological signatures in lymphoma cells that can be traced back to the original oncogene.
The team analyzed mouse models and patient samples of MYC-induced lymphoma. And they discovered lipid signatures that corresponded with the level of MYC expression.
The investigators believe this discovery could be the first step toward developing a technique to identify the origin of lymphomas and other malignancies.
They described their discovery in PNAS.
“The same cancer can occur because of different genes, but, in certain cases, the aggressiveness and the type of treatment actually depend a lot on what oncogene caused that cancer,” said study author Livia Eberlin, PhD, of Stanford University in California.
With that in mind, she and her colleagues looked at MYC, an oncogene that’s responsible for approximately half of all human cancers. They wanted to find a biological signature that would trace the mutating cancer cells back to the original oncogene.
“When cancer takes place, the cell loves to gobble up glucose—that’s a sugar—and glutamine,” said Richard Zare, PhD, also of Stanford. “It takes those and makes different lipids—different fatty molecules than what it normally makes.”
So the investigators set out to evaluate changes in lipid profiles in MYC-induced lymphoma. They compared lipid signatures in MYC-induced transgenic mouse models to those in normal control mice.
The team identified 104 molecular ions that were either increased or decreased in the MYC lymphoma models compared to controls. And 86 of these ions were complex phospholipids.
Most of the lipids that were increased in lymphoma were glycerophosphoglycerols and cardiolipins, with a higher content of monounsaturated fatty acids when compared with controls.
To determine if these findings might also apply to humans, the investigators examined 15 samples from lymphoma patients.
The samples had varying expression levels of MYC oncoprotein, and the team observed distinct lipid profiles in lymphomas with high and low MYC expression. This included many of the lipid species they had identified in the animal models of MYC-induced lymphoma.
The investigators said their results suggest a relationship between specific lipid species and the overexpression of MYC. And this information could have both diagnostic and prognostic applications. ![]()

Credit: Juha Klefstrom
Investigators have identified biological signatures in lymphoma cells that can be traced back to the original oncogene.
The team analyzed mouse models and patient samples of MYC-induced lymphoma. And they discovered lipid signatures that corresponded with the level of MYC expression.
The investigators believe this discovery could be the first step toward developing a technique to identify the origin of lymphomas and other malignancies.
They described their discovery in PNAS.
“The same cancer can occur because of different genes, but, in certain cases, the aggressiveness and the type of treatment actually depend a lot on what oncogene caused that cancer,” said study author Livia Eberlin, PhD, of Stanford University in California.
With that in mind, she and her colleagues looked at MYC, an oncogene that’s responsible for approximately half of all human cancers. They wanted to find a biological signature that would trace the mutating cancer cells back to the original oncogene.
“When cancer takes place, the cell loves to gobble up glucose—that’s a sugar—and glutamine,” said Richard Zare, PhD, also of Stanford. “It takes those and makes different lipids—different fatty molecules than what it normally makes.”
So the investigators set out to evaluate changes in lipid profiles in MYC-induced lymphoma. They compared lipid signatures in MYC-induced transgenic mouse models to those in normal control mice.
The team identified 104 molecular ions that were either increased or decreased in the MYC lymphoma models compared to controls. And 86 of these ions were complex phospholipids.
Most of the lipids that were increased in lymphoma were glycerophosphoglycerols and cardiolipins, with a higher content of monounsaturated fatty acids when compared with controls.
To determine if these findings might also apply to humans, the investigators examined 15 samples from lymphoma patients.
The samples had varying expression levels of MYC oncoprotein, and the team observed distinct lipid profiles in lymphomas with high and low MYC expression. This included many of the lipid species they had identified in the animal models of MYC-induced lymphoma.
The investigators said their results suggest a relationship between specific lipid species and the overexpression of MYC. And this information could have both diagnostic and prognostic applications. ![]()
Antibody may treat cancer cachexia

Preclinical research raises the prospect of more effective treatments for cachexia, a profound wasting of fat and muscle that can increase the risk of death in cancer patients.
In mouse models, an antibody effectively improved or prevented symptoms of cachexia.
The antibody inhibited the effects of parathyroid hormone-related protein (PTHrP), which is released from many types of cancer cells.
The researchers said their findings, published in Nature, are the first to explain in detail how PTHrP from tumors switches on a thermogenic process in fatty tissues, resulting in unhealthy weight loss.
The team carried out 2 experiments using mice that developed lung tumors and cachexia. In the first, a polyclonal antibody that specifically neutralizes PTHrP prevented cachexia almost completely, while untreated animals became mildly cachexic.
Anti-PTHrP treatment prevented the shrinkage of fat droplets. It blocked thermogenic gene expression in epididymal white adipose tissue, interscapular brown adipose tissue, and inguinal white adipose tissue, which suggests thermogenesis has a causal role in fat wasting.
Treatment with the anti-PTHrP antibody also lowered oxygen consumption in the mice, increased their physical activity, and reduced their heat production.
In the second experiment, the researchers treated mice with the anti-PTHrP antibody until they observed severe cachexia in control animals. The antibody significantly preserved muscle mass, which was evident by improved grip strength and in situ muscle contraction.
“You would have expected, based on our first experiments in cell culture, that blocking PTHrP in the mice would reduce browning of the fat,” said study author Bruce Spiegelman, PhD, of the Dana-Farber Cancer Institute in Boston.
“But we were surprised that it also affected the loss of muscle mass and improved health.”
Additional experiments, in which the researchers injected PTHrP into healthy and tumor-bearing mice, suggested that PTHrP alone doesn’t directly cause muscle wasting. But blocking the protein’s activity still prevents cachexia.
Thus, the role of PTHrP “is definitely not the whole answer” to the riddle of cachexia, Dr Spiegelman noted. Furthermore, it may turn out that the PTHrP mechanism is responsible for cachexia in only a subset of cancer patients.
The researchers analyzed blood samples from 47 cachexic patients with lung or colon cancer. And they found increased levels of PTHrP in 17 of the patients. Those patients had significantly lower lean body mass and were producing more heat energy at rest than the other patients in the group.
Dr Spiegelman noted that, before they test the anti-PTHrP antibody in clinical trials, clinicians would likely want to determine if the protein is elevated in certain cancers and determine which patients would be good candidates for the treatment. ![]()

Preclinical research raises the prospect of more effective treatments for cachexia, a profound wasting of fat and muscle that can increase the risk of death in cancer patients.
In mouse models, an antibody effectively improved or prevented symptoms of cachexia.
The antibody inhibited the effects of parathyroid hormone-related protein (PTHrP), which is released from many types of cancer cells.
The researchers said their findings, published in Nature, are the first to explain in detail how PTHrP from tumors switches on a thermogenic process in fatty tissues, resulting in unhealthy weight loss.
The team carried out 2 experiments using mice that developed lung tumors and cachexia. In the first, a polyclonal antibody that specifically neutralizes PTHrP prevented cachexia almost completely, while untreated animals became mildly cachexic.
Anti-PTHrP treatment prevented the shrinkage of fat droplets. It blocked thermogenic gene expression in epididymal white adipose tissue, interscapular brown adipose tissue, and inguinal white adipose tissue, which suggests thermogenesis has a causal role in fat wasting.
Treatment with the anti-PTHrP antibody also lowered oxygen consumption in the mice, increased their physical activity, and reduced their heat production.
In the second experiment, the researchers treated mice with the anti-PTHrP antibody until they observed severe cachexia in control animals. The antibody significantly preserved muscle mass, which was evident by improved grip strength and in situ muscle contraction.
“You would have expected, based on our first experiments in cell culture, that blocking PTHrP in the mice would reduce browning of the fat,” said study author Bruce Spiegelman, PhD, of the Dana-Farber Cancer Institute in Boston.
“But we were surprised that it also affected the loss of muscle mass and improved health.”
Additional experiments, in which the researchers injected PTHrP into healthy and tumor-bearing mice, suggested that PTHrP alone doesn’t directly cause muscle wasting. But blocking the protein’s activity still prevents cachexia.
Thus, the role of PTHrP “is definitely not the whole answer” to the riddle of cachexia, Dr Spiegelman noted. Furthermore, it may turn out that the PTHrP mechanism is responsible for cachexia in only a subset of cancer patients.
The researchers analyzed blood samples from 47 cachexic patients with lung or colon cancer. And they found increased levels of PTHrP in 17 of the patients. Those patients had significantly lower lean body mass and were producing more heat energy at rest than the other patients in the group.
Dr Spiegelman noted that, before they test the anti-PTHrP antibody in clinical trials, clinicians would likely want to determine if the protein is elevated in certain cancers and determine which patients would be good candidates for the treatment. ![]()

Preclinical research raises the prospect of more effective treatments for cachexia, a profound wasting of fat and muscle that can increase the risk of death in cancer patients.
In mouse models, an antibody effectively improved or prevented symptoms of cachexia.
The antibody inhibited the effects of parathyroid hormone-related protein (PTHrP), which is released from many types of cancer cells.
The researchers said their findings, published in Nature, are the first to explain in detail how PTHrP from tumors switches on a thermogenic process in fatty tissues, resulting in unhealthy weight loss.
The team carried out 2 experiments using mice that developed lung tumors and cachexia. In the first, a polyclonal antibody that specifically neutralizes PTHrP prevented cachexia almost completely, while untreated animals became mildly cachexic.
Anti-PTHrP treatment prevented the shrinkage of fat droplets. It blocked thermogenic gene expression in epididymal white adipose tissue, interscapular brown adipose tissue, and inguinal white adipose tissue, which suggests thermogenesis has a causal role in fat wasting.
Treatment with the anti-PTHrP antibody also lowered oxygen consumption in the mice, increased their physical activity, and reduced their heat production.
In the second experiment, the researchers treated mice with the anti-PTHrP antibody until they observed severe cachexia in control animals. The antibody significantly preserved muscle mass, which was evident by improved grip strength and in situ muscle contraction.
“You would have expected, based on our first experiments in cell culture, that blocking PTHrP in the mice would reduce browning of the fat,” said study author Bruce Spiegelman, PhD, of the Dana-Farber Cancer Institute in Boston.
“But we were surprised that it also affected the loss of muscle mass and improved health.”
Additional experiments, in which the researchers injected PTHrP into healthy and tumor-bearing mice, suggested that PTHrP alone doesn’t directly cause muscle wasting. But blocking the protein’s activity still prevents cachexia.
Thus, the role of PTHrP “is definitely not the whole answer” to the riddle of cachexia, Dr Spiegelman noted. Furthermore, it may turn out that the PTHrP mechanism is responsible for cachexia in only a subset of cancer patients.
The researchers analyzed blood samples from 47 cachexic patients with lung or colon cancer. And they found increased levels of PTHrP in 17 of the patients. Those patients had significantly lower lean body mass and were producing more heat energy at rest than the other patients in the group.
Dr Spiegelman noted that, before they test the anti-PTHrP antibody in clinical trials, clinicians would likely want to determine if the protein is elevated in certain cancers and determine which patients would be good candidates for the treatment. ![]()
Survival differences in blood cancers across Europe

Credit: Rhoda Baer
Differences in treatment access and quality may explain why survival rates vary widely for European patients with hematologic malignancies, researchers have reported in The Lancet Oncology.
“The good news is that 5-year survival for most cancers of the blood has increased over the past 11 years, most likely reflecting the approval of new targeted drugs in the early 2000s . . . ,” said Milena Sant, MD, of the Fondazione IRCCS Istituto Nazionale dei Tumori in Milan, Italy.
“But there continue to be persistent differences between regions. For example, the uptake and use of new technologies and effective treatments has been far slower in eastern Europe than other regions. This might have contributed to the large differences in the management and outcomes of patients.”
Dr Sant and her colleagues uncovered these differences by analyzing data from 30 cancer registries covering all patients diagnosed in 20 European countries.*
The researchers compared changes in 5-year survival for 560,444 adults (aged 15 years and older) who were diagnosed with 11 lymphoid and myeloid cancers between 1997 and 2008, and followed up to the end of 2008.
Some cancers have shown particularly large increases in survival between 1997-1999 and 2006-2008, such as follicular lymphoma (59% to 74%), diffuse large B-cell lymphoma (42% to 55%), chronic myeloid leukemia (32% to 54%), and acute promyelocytic leukemia (50% to 62%).
The greatest improvements in survival have been in northern, central, and eastern Europe, even though adults in eastern Europe (where survival in 1997 was the lowest) continue to have lower survival for most hematologic malignancies than elsewhere.
Survival gains have been lower in southern Europe and the UK. For example, improvements in 5-year chronic myeloid leukemia survival in northern Europe (29% to 60%) and central Europe (34% to 65%) have been persistently higher than in the UK (35% to 56%) and southern Europe (37% to 55%).
Overall, the risk of death within 5 years from diagnosis fell significantly for all malignancies except myelodysplastic syndromes. But not all regions have seen such improvements.
For example, compared with the UK, the excess risk of death was significantly higher in eastern Europe than in other regions for most of the cancers investigated, but significantly lower in northern Europe.
The researchers said the most likely reasons for continuing geographical differences in survival are inequalities in the provision of care and in the availability and use of new treatments.
“We know that rituximab, imatinib, thalidomide, and bortezomib were first made available for general use in Europe in 1997, 2001, 1998, and 2003, respectively,” the researchers wrote.
“The years following general release of these drugs coincided with large increases in survival for chronic myeloid leukemia, diffuse large B-cell lymphoma, and follicular lymphoma, with a smaller but still significant survival increase for multiple myeloma plasmacytoma.”
However, they pointed out that the uptake and use of these drugs has not been uniform across Europe. For example, market uptake of rituximab, imatinib, and bortezomib was lower in eastern Europe than elsewhere and might explain the consistently lower survival in this region.
Writing in a linked comment article, Alastair Munro, MD, of the University of Dundee Medical School in Scotland, questioned whether improvements in survival can be attributed to drugs alone.
He said that better understanding of the conclusions from this study (called EUROCARE-5) requires additional information about changes affecting survival according to disease categories, the distribution of histological subtypes and their relation with the age distribution of the population, the distribution of stages at diagnosis, and the timing of active intervention for indolent tumors. ![]()
*The areas included in the study were northern Europe (Denmark, Iceland, and Norway), the UK (England, Northern Ireland, Scotland, and Wales), central Europe (Austria, France, Germany, Switzerland, and The Netherlands), eastern Europe (Bulgaria, Estonia, Lithuania, Poland, and Slovakia), and southern Europe (Italy, Malta, and Slovenia).

Credit: Rhoda Baer
Differences in treatment access and quality may explain why survival rates vary widely for European patients with hematologic malignancies, researchers have reported in The Lancet Oncology.
“The good news is that 5-year survival for most cancers of the blood has increased over the past 11 years, most likely reflecting the approval of new targeted drugs in the early 2000s . . . ,” said Milena Sant, MD, of the Fondazione IRCCS Istituto Nazionale dei Tumori in Milan, Italy.
“But there continue to be persistent differences between regions. For example, the uptake and use of new technologies and effective treatments has been far slower in eastern Europe than other regions. This might have contributed to the large differences in the management and outcomes of patients.”
Dr Sant and her colleagues uncovered these differences by analyzing data from 30 cancer registries covering all patients diagnosed in 20 European countries.*
The researchers compared changes in 5-year survival for 560,444 adults (aged 15 years and older) who were diagnosed with 11 lymphoid and myeloid cancers between 1997 and 2008, and followed up to the end of 2008.
Some cancers have shown particularly large increases in survival between 1997-1999 and 2006-2008, such as follicular lymphoma (59% to 74%), diffuse large B-cell lymphoma (42% to 55%), chronic myeloid leukemia (32% to 54%), and acute promyelocytic leukemia (50% to 62%).
The greatest improvements in survival have been in northern, central, and eastern Europe, even though adults in eastern Europe (where survival in 1997 was the lowest) continue to have lower survival for most hematologic malignancies than elsewhere.
Survival gains have been lower in southern Europe and the UK. For example, improvements in 5-year chronic myeloid leukemia survival in northern Europe (29% to 60%) and central Europe (34% to 65%) have been persistently higher than in the UK (35% to 56%) and southern Europe (37% to 55%).
Overall, the risk of death within 5 years from diagnosis fell significantly for all malignancies except myelodysplastic syndromes. But not all regions have seen such improvements.
For example, compared with the UK, the excess risk of death was significantly higher in eastern Europe than in other regions for most of the cancers investigated, but significantly lower in northern Europe.
The researchers said the most likely reasons for continuing geographical differences in survival are inequalities in the provision of care and in the availability and use of new treatments.
“We know that rituximab, imatinib, thalidomide, and bortezomib were first made available for general use in Europe in 1997, 2001, 1998, and 2003, respectively,” the researchers wrote.
“The years following general release of these drugs coincided with large increases in survival for chronic myeloid leukemia, diffuse large B-cell lymphoma, and follicular lymphoma, with a smaller but still significant survival increase for multiple myeloma plasmacytoma.”
However, they pointed out that the uptake and use of these drugs has not been uniform across Europe. For example, market uptake of rituximab, imatinib, and bortezomib was lower in eastern Europe than elsewhere and might explain the consistently lower survival in this region.
Writing in a linked comment article, Alastair Munro, MD, of the University of Dundee Medical School in Scotland, questioned whether improvements in survival can be attributed to drugs alone.
He said that better understanding of the conclusions from this study (called EUROCARE-5) requires additional information about changes affecting survival according to disease categories, the distribution of histological subtypes and their relation with the age distribution of the population, the distribution of stages at diagnosis, and the timing of active intervention for indolent tumors. ![]()
*The areas included in the study were northern Europe (Denmark, Iceland, and Norway), the UK (England, Northern Ireland, Scotland, and Wales), central Europe (Austria, France, Germany, Switzerland, and The Netherlands), eastern Europe (Bulgaria, Estonia, Lithuania, Poland, and Slovakia), and southern Europe (Italy, Malta, and Slovenia).

Credit: Rhoda Baer
Differences in treatment access and quality may explain why survival rates vary widely for European patients with hematologic malignancies, researchers have reported in The Lancet Oncology.
“The good news is that 5-year survival for most cancers of the blood has increased over the past 11 years, most likely reflecting the approval of new targeted drugs in the early 2000s . . . ,” said Milena Sant, MD, of the Fondazione IRCCS Istituto Nazionale dei Tumori in Milan, Italy.
“But there continue to be persistent differences between regions. For example, the uptake and use of new technologies and effective treatments has been far slower in eastern Europe than other regions. This might have contributed to the large differences in the management and outcomes of patients.”
Dr Sant and her colleagues uncovered these differences by analyzing data from 30 cancer registries covering all patients diagnosed in 20 European countries.*
The researchers compared changes in 5-year survival for 560,444 adults (aged 15 years and older) who were diagnosed with 11 lymphoid and myeloid cancers between 1997 and 2008, and followed up to the end of 2008.
Some cancers have shown particularly large increases in survival between 1997-1999 and 2006-2008, such as follicular lymphoma (59% to 74%), diffuse large B-cell lymphoma (42% to 55%), chronic myeloid leukemia (32% to 54%), and acute promyelocytic leukemia (50% to 62%).
The greatest improvements in survival have been in northern, central, and eastern Europe, even though adults in eastern Europe (where survival in 1997 was the lowest) continue to have lower survival for most hematologic malignancies than elsewhere.
Survival gains have been lower in southern Europe and the UK. For example, improvements in 5-year chronic myeloid leukemia survival in northern Europe (29% to 60%) and central Europe (34% to 65%) have been persistently higher than in the UK (35% to 56%) and southern Europe (37% to 55%).
Overall, the risk of death within 5 years from diagnosis fell significantly for all malignancies except myelodysplastic syndromes. But not all regions have seen such improvements.
For example, compared with the UK, the excess risk of death was significantly higher in eastern Europe than in other regions for most of the cancers investigated, but significantly lower in northern Europe.
The researchers said the most likely reasons for continuing geographical differences in survival are inequalities in the provision of care and in the availability and use of new treatments.
“We know that rituximab, imatinib, thalidomide, and bortezomib were first made available for general use in Europe in 1997, 2001, 1998, and 2003, respectively,” the researchers wrote.
“The years following general release of these drugs coincided with large increases in survival for chronic myeloid leukemia, diffuse large B-cell lymphoma, and follicular lymphoma, with a smaller but still significant survival increase for multiple myeloma plasmacytoma.”
However, they pointed out that the uptake and use of these drugs has not been uniform across Europe. For example, market uptake of rituximab, imatinib, and bortezomib was lower in eastern Europe than elsewhere and might explain the consistently lower survival in this region.
Writing in a linked comment article, Alastair Munro, MD, of the University of Dundee Medical School in Scotland, questioned whether improvements in survival can be attributed to drugs alone.
He said that better understanding of the conclusions from this study (called EUROCARE-5) requires additional information about changes affecting survival according to disease categories, the distribution of histological subtypes and their relation with the age distribution of the population, the distribution of stages at diagnosis, and the timing of active intervention for indolent tumors. ![]()
*The areas included in the study were northern Europe (Denmark, Iceland, and Norway), the UK (England, Northern Ireland, Scotland, and Wales), central Europe (Austria, France, Germany, Switzerland, and The Netherlands), eastern Europe (Bulgaria, Estonia, Lithuania, Poland, and Slovakia), and southern Europe (Italy, Malta, and Slovenia).
Pair details ‘promise and perils’ of antioxidants

Two researchers have offered an explanation as to why antioxidants are not effective in fighting cancers and suggested a way to change that.
The duo proposed that antioxidants from supplements or dietary sources are proving ineffective because they are not acting where reactive oxygen species (ROS) are produced.
So therapies that directly inhibit the production of mitochondrial- and NADPH oxidase-derived ROS, or that scavenge ROS at these sites, may be more effective.
David Tuveson, MD, PhD, of the Cold Spring Harbor Laboratory in New York, and Navdeep S. Chandel, PhD, of the Feinberg School of Medicine at Northwestern University in Chicago, detailed these theories in a report published in The New England Journal of Medicine.
The pair’s insights are based on recent advances in understanding the cell system that establishes a natural balance between oxidizing and antioxidizing compounds.
Oxidants like hydrogen peroxide are manufactured within cells and are essential in small quantities. But oxidants are toxic in large amounts, and cells naturally generate their own antioxidants to neutralize oxidants.
It has seemed logical, therefore, to boost a person’s intake of antioxidants to counter the effects of hydrogen peroxide and other similarly toxic ROS. All the more because cancer cells are known to generate higher levels of ROS to help feed their abnormal growth.
However, Drs Tuveson and Chandel proposed that taking antioxidant pills or eating foods rich in antioxidants may be failing to show a beneficial effect against cancer because antioxidants do not act where tumor-promoting ROS are produced—at mitochondria.
Rather, supplements and dietary antioxidants tend to accumulate at scattered distant sites in the cell, “leaving tumor-promoting ROS relatively unperturbed.”
Therefore, the authors suggested therapies that directly inhibit the production of mitochondrial- and NADPH oxidase-derived ROS, or that scavenge ROS at these sites, will be more effective than dietary antioxidants.
An alternative approach
Drs Tuveson and Chandel also proposed an alternative approach: disabling antioxidants in cancer cells. They noted that quantities of both ROS and natural antioxidants are higher in cancer cells. The higher levels of antioxidants are a natural defense by cancer cells to keep their higher levels of oxidants in check so that growth can continue.
In fact, therapies that raise the levels of oxidants in cells can be beneficial, whereas those that act as antioxidants may further stimulate the cancer cells.
So the authors suggested that genetic or pharmacologic inhibition of antioxidant proteins—a concept tested successfully in rodent models of lung and pancreatic cancers—may be a useful therapeutic approach in humans.
The key challenge is to identify antioxidant proteins and pathways in cells that are used only by cancer cells and not by healthy cells. Impeding antioxidant production in healthy cells will upset the delicate redox balance upon which normal cellular function depends.
So it seems research is needed to profile antioxidant pathways in tumor and adjacent normal cells, to identify possible therapeutic targets. ![]()

Two researchers have offered an explanation as to why antioxidants are not effective in fighting cancers and suggested a way to change that.
The duo proposed that antioxidants from supplements or dietary sources are proving ineffective because they are not acting where reactive oxygen species (ROS) are produced.
So therapies that directly inhibit the production of mitochondrial- and NADPH oxidase-derived ROS, or that scavenge ROS at these sites, may be more effective.
David Tuveson, MD, PhD, of the Cold Spring Harbor Laboratory in New York, and Navdeep S. Chandel, PhD, of the Feinberg School of Medicine at Northwestern University in Chicago, detailed these theories in a report published in The New England Journal of Medicine.
The pair’s insights are based on recent advances in understanding the cell system that establishes a natural balance between oxidizing and antioxidizing compounds.
Oxidants like hydrogen peroxide are manufactured within cells and are essential in small quantities. But oxidants are toxic in large amounts, and cells naturally generate their own antioxidants to neutralize oxidants.
It has seemed logical, therefore, to boost a person’s intake of antioxidants to counter the effects of hydrogen peroxide and other similarly toxic ROS. All the more because cancer cells are known to generate higher levels of ROS to help feed their abnormal growth.
However, Drs Tuveson and Chandel proposed that taking antioxidant pills or eating foods rich in antioxidants may be failing to show a beneficial effect against cancer because antioxidants do not act where tumor-promoting ROS are produced—at mitochondria.
Rather, supplements and dietary antioxidants tend to accumulate at scattered distant sites in the cell, “leaving tumor-promoting ROS relatively unperturbed.”
Therefore, the authors suggested therapies that directly inhibit the production of mitochondrial- and NADPH oxidase-derived ROS, or that scavenge ROS at these sites, will be more effective than dietary antioxidants.
An alternative approach
Drs Tuveson and Chandel also proposed an alternative approach: disabling antioxidants in cancer cells. They noted that quantities of both ROS and natural antioxidants are higher in cancer cells. The higher levels of antioxidants are a natural defense by cancer cells to keep their higher levels of oxidants in check so that growth can continue.
In fact, therapies that raise the levels of oxidants in cells can be beneficial, whereas those that act as antioxidants may further stimulate the cancer cells.
So the authors suggested that genetic or pharmacologic inhibition of antioxidant proteins—a concept tested successfully in rodent models of lung and pancreatic cancers—may be a useful therapeutic approach in humans.
The key challenge is to identify antioxidant proteins and pathways in cells that are used only by cancer cells and not by healthy cells. Impeding antioxidant production in healthy cells will upset the delicate redox balance upon which normal cellular function depends.
So it seems research is needed to profile antioxidant pathways in tumor and adjacent normal cells, to identify possible therapeutic targets. ![]()

Two researchers have offered an explanation as to why antioxidants are not effective in fighting cancers and suggested a way to change that.
The duo proposed that antioxidants from supplements or dietary sources are proving ineffective because they are not acting where reactive oxygen species (ROS) are produced.
So therapies that directly inhibit the production of mitochondrial- and NADPH oxidase-derived ROS, or that scavenge ROS at these sites, may be more effective.
David Tuveson, MD, PhD, of the Cold Spring Harbor Laboratory in New York, and Navdeep S. Chandel, PhD, of the Feinberg School of Medicine at Northwestern University in Chicago, detailed these theories in a report published in The New England Journal of Medicine.
The pair’s insights are based on recent advances in understanding the cell system that establishes a natural balance between oxidizing and antioxidizing compounds.
Oxidants like hydrogen peroxide are manufactured within cells and are essential in small quantities. But oxidants are toxic in large amounts, and cells naturally generate their own antioxidants to neutralize oxidants.
It has seemed logical, therefore, to boost a person’s intake of antioxidants to counter the effects of hydrogen peroxide and other similarly toxic ROS. All the more because cancer cells are known to generate higher levels of ROS to help feed their abnormal growth.
However, Drs Tuveson and Chandel proposed that taking antioxidant pills or eating foods rich in antioxidants may be failing to show a beneficial effect against cancer because antioxidants do not act where tumor-promoting ROS are produced—at mitochondria.
Rather, supplements and dietary antioxidants tend to accumulate at scattered distant sites in the cell, “leaving tumor-promoting ROS relatively unperturbed.”
Therefore, the authors suggested therapies that directly inhibit the production of mitochondrial- and NADPH oxidase-derived ROS, or that scavenge ROS at these sites, will be more effective than dietary antioxidants.
An alternative approach
Drs Tuveson and Chandel also proposed an alternative approach: disabling antioxidants in cancer cells. They noted that quantities of both ROS and natural antioxidants are higher in cancer cells. The higher levels of antioxidants are a natural defense by cancer cells to keep their higher levels of oxidants in check so that growth can continue.
In fact, therapies that raise the levels of oxidants in cells can be beneficial, whereas those that act as antioxidants may further stimulate the cancer cells.
So the authors suggested that genetic or pharmacologic inhibition of antioxidant proteins—a concept tested successfully in rodent models of lung and pancreatic cancers—may be a useful therapeutic approach in humans.
The key challenge is to identify antioxidant proteins and pathways in cells that are used only by cancer cells and not by healthy cells. Impeding antioxidant production in healthy cells will upset the delicate redox balance upon which normal cellular function depends.
So it seems research is needed to profile antioxidant pathways in tumor and adjacent normal cells, to identify possible therapeutic targets. ![]()
Telomeres can help predict prognosis in CLL

with telomeres in green
Credit: Claus Azzalin
Measuring the length and function of telomeres can help us predict prognosis in patients with chronic lymphocytic leukemia (CLL), according to research published in the British Journal of Haematology.
Investigators found that CLL patients with short, dysfunctional telomeres had a considerably poorer clinical outcome than those with long, functional telomeres.
“For the first time, confident predictions of clinical outcome can be made for individual CLL patients at diagnosis based on accurate analysis of the length of telomeres in cancer cells,” said Chris Pepper, PhD, who led the research at Cardiff University’s School of Medicine in the UK.
“This should prove enormously valuable to doctors, patients, and their families, and there is no reason why there should not be widespread implementation of this powerful prognostic tool in the near future.”
CLL progression is known to be sped up by the loss of telomeres, which cap the ends of chromosomes and protect them from damage when a cell divides. Every time a cell divides, telomeres get shorter.
When they become too short in a healthy cell, signals are sent to instruct the cell to stop dividing and die. But this “safety check” does not occur in CLL cells. Telomeres become so short that chromosomes are left exposed and are prone to fusing together during cell division, causing even larger DNA faults and even greater instability.
So Dr Pepper and his colleagues set out to identify the telomere length at which fusions start to occur in CLL patients.
The team measured telomeres in patient samples using single telomere length analysis (STELA) along with an experimentally derived definition of telomere dysfunction. They defined the upper telomere length threshold at which telomere fusions occur and used the mean of the telomere “fusogenic” range as a prognostic tool.
The researchers first analyzed samples from 200 CLL patients and found that patients with telomeres below the fusogenic mean had significantly shorter overall survival than patients with telomeres above the fusogenic mean (hazard ratio [HR]=13.2, P<0.0001). This was also true for patients with early stage disease (HR=19.3, P<0.0001).
The investigators confirmed this association by analyzing samples from an additional 121 CLL patients. The prognostic impact of telomere dysfunction was evident in the entire cohort (HR=7.4, P<0.0001) and among patients classified as Binet stage A (HR=8.9, P<0.0001).
The researchers also found they could use telomere dysfunction to accurately classify Binet stage A patients into an indolent disease group and a poor prognostic group. At 10 years, the survival rate was 91% in the favorable prognostic group and 13% in the poor prognostic group.
Of note, patients with telomeres above the fusogenic mean had superior prognosis regardless of their IGHV mutation status or cytogenetic risk group. And in a multivariate analysis, the telomere fusogenic mean was associated with the highest hazard of progression and death, independent of all other biomarkers.
“The accuracy of this test in predicting how a person’s disease will develop is unprecedented and, if confirmed in clinical trials, would help doctors decide on the best treatment courses for individual CLL patients,” said Matt Kaiser, PhD, Head of Research at Leukaemia & Lymphoma Research, which funded this study.
“Telomeres are known to play a part in the progress of other forms of cancer, so this type of testing could have far-reaching benefits.” ![]()

with telomeres in green
Credit: Claus Azzalin
Measuring the length and function of telomeres can help us predict prognosis in patients with chronic lymphocytic leukemia (CLL), according to research published in the British Journal of Haematology.
Investigators found that CLL patients with short, dysfunctional telomeres had a considerably poorer clinical outcome than those with long, functional telomeres.
“For the first time, confident predictions of clinical outcome can be made for individual CLL patients at diagnosis based on accurate analysis of the length of telomeres in cancer cells,” said Chris Pepper, PhD, who led the research at Cardiff University’s School of Medicine in the UK.
“This should prove enormously valuable to doctors, patients, and their families, and there is no reason why there should not be widespread implementation of this powerful prognostic tool in the near future.”
CLL progression is known to be sped up by the loss of telomeres, which cap the ends of chromosomes and protect them from damage when a cell divides. Every time a cell divides, telomeres get shorter.
When they become too short in a healthy cell, signals are sent to instruct the cell to stop dividing and die. But this “safety check” does not occur in CLL cells. Telomeres become so short that chromosomes are left exposed and are prone to fusing together during cell division, causing even larger DNA faults and even greater instability.
So Dr Pepper and his colleagues set out to identify the telomere length at which fusions start to occur in CLL patients.
The team measured telomeres in patient samples using single telomere length analysis (STELA) along with an experimentally derived definition of telomere dysfunction. They defined the upper telomere length threshold at which telomere fusions occur and used the mean of the telomere “fusogenic” range as a prognostic tool.
The researchers first analyzed samples from 200 CLL patients and found that patients with telomeres below the fusogenic mean had significantly shorter overall survival than patients with telomeres above the fusogenic mean (hazard ratio [HR]=13.2, P<0.0001). This was also true for patients with early stage disease (HR=19.3, P<0.0001).
The investigators confirmed this association by analyzing samples from an additional 121 CLL patients. The prognostic impact of telomere dysfunction was evident in the entire cohort (HR=7.4, P<0.0001) and among patients classified as Binet stage A (HR=8.9, P<0.0001).
The researchers also found they could use telomere dysfunction to accurately classify Binet stage A patients into an indolent disease group and a poor prognostic group. At 10 years, the survival rate was 91% in the favorable prognostic group and 13% in the poor prognostic group.
Of note, patients with telomeres above the fusogenic mean had superior prognosis regardless of their IGHV mutation status or cytogenetic risk group. And in a multivariate analysis, the telomere fusogenic mean was associated with the highest hazard of progression and death, independent of all other biomarkers.
“The accuracy of this test in predicting how a person’s disease will develop is unprecedented and, if confirmed in clinical trials, would help doctors decide on the best treatment courses for individual CLL patients,” said Matt Kaiser, PhD, Head of Research at Leukaemia & Lymphoma Research, which funded this study.
“Telomeres are known to play a part in the progress of other forms of cancer, so this type of testing could have far-reaching benefits.” ![]()

with telomeres in green
Credit: Claus Azzalin
Measuring the length and function of telomeres can help us predict prognosis in patients with chronic lymphocytic leukemia (CLL), according to research published in the British Journal of Haematology.
Investigators found that CLL patients with short, dysfunctional telomeres had a considerably poorer clinical outcome than those with long, functional telomeres.
“For the first time, confident predictions of clinical outcome can be made for individual CLL patients at diagnosis based on accurate analysis of the length of telomeres in cancer cells,” said Chris Pepper, PhD, who led the research at Cardiff University’s School of Medicine in the UK.
“This should prove enormously valuable to doctors, patients, and their families, and there is no reason why there should not be widespread implementation of this powerful prognostic tool in the near future.”
CLL progression is known to be sped up by the loss of telomeres, which cap the ends of chromosomes and protect them from damage when a cell divides. Every time a cell divides, telomeres get shorter.
When they become too short in a healthy cell, signals are sent to instruct the cell to stop dividing and die. But this “safety check” does not occur in CLL cells. Telomeres become so short that chromosomes are left exposed and are prone to fusing together during cell division, causing even larger DNA faults and even greater instability.
So Dr Pepper and his colleagues set out to identify the telomere length at which fusions start to occur in CLL patients.
The team measured telomeres in patient samples using single telomere length analysis (STELA) along with an experimentally derived definition of telomere dysfunction. They defined the upper telomere length threshold at which telomere fusions occur and used the mean of the telomere “fusogenic” range as a prognostic tool.
The researchers first analyzed samples from 200 CLL patients and found that patients with telomeres below the fusogenic mean had significantly shorter overall survival than patients with telomeres above the fusogenic mean (hazard ratio [HR]=13.2, P<0.0001). This was also true for patients with early stage disease (HR=19.3, P<0.0001).
The investigators confirmed this association by analyzing samples from an additional 121 CLL patients. The prognostic impact of telomere dysfunction was evident in the entire cohort (HR=7.4, P<0.0001) and among patients classified as Binet stage A (HR=8.9, P<0.0001).
The researchers also found they could use telomere dysfunction to accurately classify Binet stage A patients into an indolent disease group and a poor prognostic group. At 10 years, the survival rate was 91% in the favorable prognostic group and 13% in the poor prognostic group.
Of note, patients with telomeres above the fusogenic mean had superior prognosis regardless of their IGHV mutation status or cytogenetic risk group. And in a multivariate analysis, the telomere fusogenic mean was associated with the highest hazard of progression and death, independent of all other biomarkers.
“The accuracy of this test in predicting how a person’s disease will develop is unprecedented and, if confirmed in clinical trials, would help doctors decide on the best treatment courses for individual CLL patients,” said Matt Kaiser, PhD, Head of Research at Leukaemia & Lymphoma Research, which funded this study.
“Telomeres are known to play a part in the progress of other forms of cancer, so this type of testing could have far-reaching benefits.” ![]()
New formulation improves chemo drug

Credit: Larry Ostby
A new formulation of the chemotherapy drug cisplatin can significantly increase the drug’s ability to target and destroy cancer cells, a new study suggests.
Scientists constructed a modified version of cisplatin called Platin-M, which is designed to overcome treatment resistance by attacking mitochondria within cancer cells.
“You can think of mitochondria as a kind of powerhouse for the cell, generating the energy it needs to grow and reproduce,” said Shanta Dhar, PhD, of the University of Georgia in Athens, Georgia.
“This prodrug delivers cisplatin directly to the mitochondria in cancerous cells. Without that essential powerhouse, the cell cannot survive.”
Dr Dhar and her colleagues described the creation of this prodrug in the Proceedings of the National Academy of Sciences.
Sean Marrache, a graduate student in Dr Dhar’s lab, entrapped Platin-M in a specially designed nanoparticle that seeks out the mitochondria and releases the drug. Once inside, Platin-M interferes with the mitochondria’s DNA, triggering cell death.
The researchers tested Platin-M on neuroblastoma cells. In experiments using a cisplatin-resistant cell culture, Platin-M nanoparticles were roughly 17 times more active than cisplatin alone.
“This technique could become a treatment for a number of cancers, but it may prove most useful for more aggressive forms of cancer that are resistant to current therapies,” said Rakesh Pathak, PhD, a postdoctoral researcher in Dr Dhar’s lab.
However, the researchers cautioned that these results are preliminary, and more work is necessary before Platin-M enters clinical trials. Still, their early results in mouse models are encouraging, and they are currently developing safety trials in larger animals.
“Cisplatin is a well-studied chemotherapy, so we hope our unique formulation will enhance its efficacy,” Dr Dhar said. “We are excited about these early results, which look very promising.” ![]()

Credit: Larry Ostby
A new formulation of the chemotherapy drug cisplatin can significantly increase the drug’s ability to target and destroy cancer cells, a new study suggests.
Scientists constructed a modified version of cisplatin called Platin-M, which is designed to overcome treatment resistance by attacking mitochondria within cancer cells.
“You can think of mitochondria as a kind of powerhouse for the cell, generating the energy it needs to grow and reproduce,” said Shanta Dhar, PhD, of the University of Georgia in Athens, Georgia.
“This prodrug delivers cisplatin directly to the mitochondria in cancerous cells. Without that essential powerhouse, the cell cannot survive.”
Dr Dhar and her colleagues described the creation of this prodrug in the Proceedings of the National Academy of Sciences.
Sean Marrache, a graduate student in Dr Dhar’s lab, entrapped Platin-M in a specially designed nanoparticle that seeks out the mitochondria and releases the drug. Once inside, Platin-M interferes with the mitochondria’s DNA, triggering cell death.
The researchers tested Platin-M on neuroblastoma cells. In experiments using a cisplatin-resistant cell culture, Platin-M nanoparticles were roughly 17 times more active than cisplatin alone.
“This technique could become a treatment for a number of cancers, but it may prove most useful for more aggressive forms of cancer that are resistant to current therapies,” said Rakesh Pathak, PhD, a postdoctoral researcher in Dr Dhar’s lab.
However, the researchers cautioned that these results are preliminary, and more work is necessary before Platin-M enters clinical trials. Still, their early results in mouse models are encouraging, and they are currently developing safety trials in larger animals.
“Cisplatin is a well-studied chemotherapy, so we hope our unique formulation will enhance its efficacy,” Dr Dhar said. “We are excited about these early results, which look very promising.” ![]()

Credit: Larry Ostby
A new formulation of the chemotherapy drug cisplatin can significantly increase the drug’s ability to target and destroy cancer cells, a new study suggests.
Scientists constructed a modified version of cisplatin called Platin-M, which is designed to overcome treatment resistance by attacking mitochondria within cancer cells.
“You can think of mitochondria as a kind of powerhouse for the cell, generating the energy it needs to grow and reproduce,” said Shanta Dhar, PhD, of the University of Georgia in Athens, Georgia.
“This prodrug delivers cisplatin directly to the mitochondria in cancerous cells. Without that essential powerhouse, the cell cannot survive.”
Dr Dhar and her colleagues described the creation of this prodrug in the Proceedings of the National Academy of Sciences.
Sean Marrache, a graduate student in Dr Dhar’s lab, entrapped Platin-M in a specially designed nanoparticle that seeks out the mitochondria and releases the drug. Once inside, Platin-M interferes with the mitochondria’s DNA, triggering cell death.
The researchers tested Platin-M on neuroblastoma cells. In experiments using a cisplatin-resistant cell culture, Platin-M nanoparticles were roughly 17 times more active than cisplatin alone.
“This technique could become a treatment for a number of cancers, but it may prove most useful for more aggressive forms of cancer that are resistant to current therapies,” said Rakesh Pathak, PhD, a postdoctoral researcher in Dr Dhar’s lab.
However, the researchers cautioned that these results are preliminary, and more work is necessary before Platin-M enters clinical trials. Still, their early results in mouse models are encouraging, and they are currently developing safety trials in larger animals.
“Cisplatin is a well-studied chemotherapy, so we hope our unique formulation will enhance its efficacy,” Dr Dhar said. “We are excited about these early results, which look very promising.”
CAR T cells may fight fungal infections
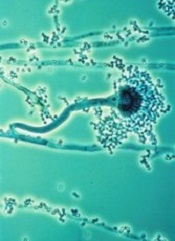
T cells modified using the Sleeping Beauty gene transfer system may help fight infections caused by invasive Aspergillus fungus.
Sleeping Beauty is already being used to create chimeric antigen receptor (CAR) T cells to treat leukemias and lymphomas.
And now, researchers have found the system may also be effective for combatting fungal infections that can be deadly for immunosuppressed patients, such as those receiving transplants to treat hematologic cancers.
“We demonstrated a new approach for Aspergillus immunotherapy based on redirecting T-cell specificity through a CAR that recognizes carbohydrate antigen on the fungal cell wall,” said study author Laurence Cooper, MD, PhD, of MD Anderson Cancer Center in Houston, Texas.
He and his colleagues described this approach in the Proceedings of the National Academy of Sciences.
Dr Cooper originally learned about Sleeping Beauty gene transfer from a study published by Perry Hackett, PhD, a professor at the University of Minnesota who created the process.
The system is named Sleeping Beauty because Dr Hackett was able to “awaken” an extinct transposon—DNA that can replicate itself and insert the copy back into the genome—and package it with a gene he wants to transfer into a plasmid. An associated transposase enzyme binds to the plasmid, cuts the transposon and gene out of the plasmid, and pastes it into the target DNA sequence.
Dr Cooper and his colleagues have found they can use this process to engineer T cells that target sugar molecules in the Aspergillus cell walls, thereby killing the fungus.
Specifically, the team adapted the pattern-recognition receptor Dectin-1 to activate T cells via chimeric CD28 and CD3-ζ (D-CAR) upon binding with carbohydrate in the cell wall of Aspergillus germlings. They used Sleeping Beauty to modify the T cells to express D-CAR.
These D-CAR+ T cells exhibited specificity for β-glucan, and this inhibited Aspergillus growth both in vitro and in vivo. Furthermore, the researchers found that treating D-CAR+ T cells with steroids did not significantly compromise antifungal activity.
“The [D-CAR+ T cells] can be manipulated in a manner suitable for human application,” Dr Cooper said, “enabling this immunology to be translated into immunotherapy.”

T cells modified using the Sleeping Beauty gene transfer system may help fight infections caused by invasive Aspergillus fungus.
Sleeping Beauty is already being used to create chimeric antigen receptor (CAR) T cells to treat leukemias and lymphomas.
And now, researchers have found the system may also be effective for combatting fungal infections that can be deadly for immunosuppressed patients, such as those receiving transplants to treat hematologic cancers.
“We demonstrated a new approach for Aspergillus immunotherapy based on redirecting T-cell specificity through a CAR that recognizes carbohydrate antigen on the fungal cell wall,” said study author Laurence Cooper, MD, PhD, of MD Anderson Cancer Center in Houston, Texas.
He and his colleagues described this approach in the Proceedings of the National Academy of Sciences.
Dr Cooper originally learned about Sleeping Beauty gene transfer from a study published by Perry Hackett, PhD, a professor at the University of Minnesota who created the process.
The system is named Sleeping Beauty because Dr Hackett was able to “awaken” an extinct transposon—DNA that can replicate itself and insert the copy back into the genome—and package it with a gene he wants to transfer into a plasmid. An associated transposase enzyme binds to the plasmid, cuts the transposon and gene out of the plasmid, and pastes it into the target DNA sequence.
Dr Cooper and his colleagues have found they can use this process to engineer T cells that target sugar molecules in the Aspergillus cell walls, thereby killing the fungus.
Specifically, the team adapted the pattern-recognition receptor Dectin-1 to activate T cells via chimeric CD28 and CD3-ζ (D-CAR) upon binding with carbohydrate in the cell wall of Aspergillus germlings. They used Sleeping Beauty to modify the T cells to express D-CAR.
These D-CAR+ T cells exhibited specificity for β-glucan, and this inhibited Aspergillus growth both in vitro and in vivo. Furthermore, the researchers found that treating D-CAR+ T cells with steroids did not significantly compromise antifungal activity.
“The [D-CAR+ T cells] can be manipulated in a manner suitable for human application,” Dr Cooper said, “enabling this immunology to be translated into immunotherapy.”

T cells modified using the Sleeping Beauty gene transfer system may help fight infections caused by invasive Aspergillus fungus.
Sleeping Beauty is already being used to create chimeric antigen receptor (CAR) T cells to treat leukemias and lymphomas.
And now, researchers have found the system may also be effective for combatting fungal infections that can be deadly for immunosuppressed patients, such as those receiving transplants to treat hematologic cancers.
“We demonstrated a new approach for Aspergillus immunotherapy based on redirecting T-cell specificity through a CAR that recognizes carbohydrate antigen on the fungal cell wall,” said study author Laurence Cooper, MD, PhD, of MD Anderson Cancer Center in Houston, Texas.
He and his colleagues described this approach in the Proceedings of the National Academy of Sciences.
Dr Cooper originally learned about Sleeping Beauty gene transfer from a study published by Perry Hackett, PhD, a professor at the University of Minnesota who created the process.
The system is named Sleeping Beauty because Dr Hackett was able to “awaken” an extinct transposon—DNA that can replicate itself and insert the copy back into the genome—and package it with a gene he wants to transfer into a plasmid. An associated transposase enzyme binds to the plasmid, cuts the transposon and gene out of the plasmid, and pastes it into the target DNA sequence.
Dr Cooper and his colleagues have found they can use this process to engineer T cells that target sugar molecules in the Aspergillus cell walls, thereby killing the fungus.
Specifically, the team adapted the pattern-recognition receptor Dectin-1 to activate T cells via chimeric CD28 and CD3-ζ (D-CAR) upon binding with carbohydrate in the cell wall of Aspergillus germlings. They used Sleeping Beauty to modify the T cells to express D-CAR.
These D-CAR+ T cells exhibited specificity for β-glucan, and this inhibited Aspergillus growth both in vitro and in vivo. Furthermore, the researchers found that treating D-CAR+ T cells with steroids did not significantly compromise antifungal activity.
“The [D-CAR+ T cells] can be manipulated in a manner suitable for human application,” Dr Cooper said, “enabling this immunology to be translated into immunotherapy.”
Increasing AYA enrollment in cancer trials

patient and her father
Credit: Rhoda Baer
Age limits on clinical trials must be more flexible to allow more adolescent and young adult (AYA) cancer patients the opportunity to access new treatments, according to a report published in The Lancet Oncology.
The report’s authors discovered that expanding age eligibility criteria for cancer trials increased the enrollment of AYA patients and patients belonging to other age groups. But there is still room for improvement, according to the authors.
“[R]ight now, too many of our young patients are needlessly falling through the gap between pediatric and adult cancer trials,” said Lorna Fern, PhD, of University College London Hospitals in the UK.
“By encouraging doctors to take into account the full age range of patients affected by individual types of cancer, we’ve shown that it’s possible to design trials that include teenage cancer patients and, importantly, that better match the underlying biology of the disease and the people affected.”
To assess AYA enrollment in cancer trials, Dr Fern and her colleagues looked at 68,275 cancer patients aged 0 to 59 years. They were diagnosed with leukemias, lymphomas, or solid tumor malignancies between April 1, 2005, and March 31, 2010.
During this 6-year period, trial participation increased among all age groups. There was a 13% increase in participation among 15- to 19-year-olds (from 24% to 37%), a 5% increase among 20- to 24-year-olds (from 13% to 18%), and a 6% increase among 0- to 14-year-olds (from 52% to 58%).
Dr Fern and her colleagues said the rise in enrollment, particularly among AYAs, was due to increased availability and access to trials; increased awareness from healthcare professionals, patients, and the public about research; and the opening of trials with broader age limits that allow AYAs to enter trials.
In light of this study, Cancer Research UK has started asking researchers to justify age restrictions on new studies, in an effort to recruit more AYA cancer patients onto its trials.
“[I]t’s vital that effective treatments are being developed to tackle cancer across all age brackets,” said Kate Law, Cancer Research UK’s director of clinical trials.
“We now only accept age limits on our clinical trials if they are backed up by hard evidence, which will hopefully mean more young cancer patients get the chance to contribute to research and have the latest experimental treatments.”

patient and her father
Credit: Rhoda Baer
Age limits on clinical trials must be more flexible to allow more adolescent and young adult (AYA) cancer patients the opportunity to access new treatments, according to a report published in The Lancet Oncology.
The report’s authors discovered that expanding age eligibility criteria for cancer trials increased the enrollment of AYA patients and patients belonging to other age groups. But there is still room for improvement, according to the authors.
“[R]ight now, too many of our young patients are needlessly falling through the gap between pediatric and adult cancer trials,” said Lorna Fern, PhD, of University College London Hospitals in the UK.
“By encouraging doctors to take into account the full age range of patients affected by individual types of cancer, we’ve shown that it’s possible to design trials that include teenage cancer patients and, importantly, that better match the underlying biology of the disease and the people affected.”
To assess AYA enrollment in cancer trials, Dr Fern and her colleagues looked at 68,275 cancer patients aged 0 to 59 years. They were diagnosed with leukemias, lymphomas, or solid tumor malignancies between April 1, 2005, and March 31, 2010.
During this 6-year period, trial participation increased among all age groups. There was a 13% increase in participation among 15- to 19-year-olds (from 24% to 37%), a 5% increase among 20- to 24-year-olds (from 13% to 18%), and a 6% increase among 0- to 14-year-olds (from 52% to 58%).
Dr Fern and her colleagues said the rise in enrollment, particularly among AYAs, was due to increased availability and access to trials; increased awareness from healthcare professionals, patients, and the public about research; and the opening of trials with broader age limits that allow AYAs to enter trials.
In light of this study, Cancer Research UK has started asking researchers to justify age restrictions on new studies, in an effort to recruit more AYA cancer patients onto its trials.
“[I]t’s vital that effective treatments are being developed to tackle cancer across all age brackets,” said Kate Law, Cancer Research UK’s director of clinical trials.
“We now only accept age limits on our clinical trials if they are backed up by hard evidence, which will hopefully mean more young cancer patients get the chance to contribute to research and have the latest experimental treatments.”

patient and her father
Credit: Rhoda Baer
Age limits on clinical trials must be more flexible to allow more adolescent and young adult (AYA) cancer patients the opportunity to access new treatments, according to a report published in The Lancet Oncology.
The report’s authors discovered that expanding age eligibility criteria for cancer trials increased the enrollment of AYA patients and patients belonging to other age groups. But there is still room for improvement, according to the authors.
“[R]ight now, too many of our young patients are needlessly falling through the gap between pediatric and adult cancer trials,” said Lorna Fern, PhD, of University College London Hospitals in the UK.
“By encouraging doctors to take into account the full age range of patients affected by individual types of cancer, we’ve shown that it’s possible to design trials that include teenage cancer patients and, importantly, that better match the underlying biology of the disease and the people affected.”
To assess AYA enrollment in cancer trials, Dr Fern and her colleagues looked at 68,275 cancer patients aged 0 to 59 years. They were diagnosed with leukemias, lymphomas, or solid tumor malignancies between April 1, 2005, and March 31, 2010.
During this 6-year period, trial participation increased among all age groups. There was a 13% increase in participation among 15- to 19-year-olds (from 24% to 37%), a 5% increase among 20- to 24-year-olds (from 13% to 18%), and a 6% increase among 0- to 14-year-olds (from 52% to 58%).
Dr Fern and her colleagues said the rise in enrollment, particularly among AYAs, was due to increased availability and access to trials; increased awareness from healthcare professionals, patients, and the public about research; and the opening of trials with broader age limits that allow AYAs to enter trials.
In light of this study, Cancer Research UK has started asking researchers to justify age restrictions on new studies, in an effort to recruit more AYA cancer patients onto its trials.
“[I]t’s vital that effective treatments are being developed to tackle cancer across all age brackets,” said Kate Law, Cancer Research UK’s director of clinical trials.
“We now only accept age limits on our clinical trials if they are backed up by hard evidence, which will hopefully mean more young cancer patients get the chance to contribute to research and have the latest experimental treatments.”
EC expands indication for ofatumumab

Credit: Linda Bartlett
The European Commission (EC) has expanded the indication for the anti-CD20 monoclonal antibody ofatumumab (Arzerra).
The EC granted conditional approval for ofatumumab in combination with chlorambucil or bendamustine to treat patients with chronic lymphocytic leukemia (CLL) who have not received prior therapy and are not eligible for fludarabine-based therapy.
In 2010, the EC granted ofatumumab conditional approval to treat CLL patients who are refractory to fludarabine and alemtuzumab.
Ofatumumab received conditional approval because the drug’s benefits appear to outweigh the risks it poses. Ofatumumab will not receive full approval until the drug’s developers, GlaxoSmithKline and GenMab, submit results of additional research to the EC.
Trial results
The EC’s new approval of ofatumumab, for previously untreated CLL patients, is based on results from 2 trials, COMPLEMENT 1 and OMB115991.
The phase 3 COMPLEMENT 1 trial was a comparison of ofatumumab plus chlorambucil (n=221) with chlorambucil alone (n=226) in CLL patients for whom fludarabine-based treatment was considered inappropriate.
In this study, ofatumumab plus chlorambucil improved progression-free survival compared to chlorambucil alone. The median times were 22.4 months and 13.1 months, respectively, and the hazard ratio was 0.57 (P<0.001).
In the phase 2 trial known as OMB115991, researchers evaluated ofatumumab in combination with bendamustine in 44 patients with previously untreated CLL for whom fludarabine-based treatment was considered inappropriate.
The combination elicited an overall response rate of 95% and a complete response rate of 43%.
The overall safety profile of ofatumumab in CLL (previously untreated and relapsed/refractory) is based on data from 511 patients in clinical trials.
This includes 250 patients with relapsed/refractory CLL who were treated with ofatumumab alone and 261 patients with previously untreated CLL who were treated in combination with an alkylating agent.
The most common adverse effects associated with ofatumumab were infusion reactions, neutropenia, anemia, febrile neutropenia, thrombocytopenia, leukopenia, lower respiratory tract infection (including pneumonia), upper respiratory tract infection, sepsis (including neutropenic sepsis and septic shock), herpes virus infection, and urinary tract infection.

Credit: Linda Bartlett
The European Commission (EC) has expanded the indication for the anti-CD20 monoclonal antibody ofatumumab (Arzerra).
The EC granted conditional approval for ofatumumab in combination with chlorambucil or bendamustine to treat patients with chronic lymphocytic leukemia (CLL) who have not received prior therapy and are not eligible for fludarabine-based therapy.
In 2010, the EC granted ofatumumab conditional approval to treat CLL patients who are refractory to fludarabine and alemtuzumab.
Ofatumumab received conditional approval because the drug’s benefits appear to outweigh the risks it poses. Ofatumumab will not receive full approval until the drug’s developers, GlaxoSmithKline and GenMab, submit results of additional research to the EC.
Trial results
The EC’s new approval of ofatumumab, for previously untreated CLL patients, is based on results from 2 trials, COMPLEMENT 1 and OMB115991.
The phase 3 COMPLEMENT 1 trial was a comparison of ofatumumab plus chlorambucil (n=221) with chlorambucil alone (n=226) in CLL patients for whom fludarabine-based treatment was considered inappropriate.
In this study, ofatumumab plus chlorambucil improved progression-free survival compared to chlorambucil alone. The median times were 22.4 months and 13.1 months, respectively, and the hazard ratio was 0.57 (P<0.001).
In the phase 2 trial known as OMB115991, researchers evaluated ofatumumab in combination with bendamustine in 44 patients with previously untreated CLL for whom fludarabine-based treatment was considered inappropriate.
The combination elicited an overall response rate of 95% and a complete response rate of 43%.
The overall safety profile of ofatumumab in CLL (previously untreated and relapsed/refractory) is based on data from 511 patients in clinical trials.
This includes 250 patients with relapsed/refractory CLL who were treated with ofatumumab alone and 261 patients with previously untreated CLL who were treated in combination with an alkylating agent.
The most common adverse effects associated with ofatumumab were infusion reactions, neutropenia, anemia, febrile neutropenia, thrombocytopenia, leukopenia, lower respiratory tract infection (including pneumonia), upper respiratory tract infection, sepsis (including neutropenic sepsis and septic shock), herpes virus infection, and urinary tract infection.

Credit: Linda Bartlett
The European Commission (EC) has expanded the indication for the anti-CD20 monoclonal antibody ofatumumab (Arzerra).
The EC granted conditional approval for ofatumumab in combination with chlorambucil or bendamustine to treat patients with chronic lymphocytic leukemia (CLL) who have not received prior therapy and are not eligible for fludarabine-based therapy.
In 2010, the EC granted ofatumumab conditional approval to treat CLL patients who are refractory to fludarabine and alemtuzumab.
Ofatumumab received conditional approval because the drug’s benefits appear to outweigh the risks it poses. Ofatumumab will not receive full approval until the drug’s developers, GlaxoSmithKline and GenMab, submit results of additional research to the EC.
Trial results
The EC’s new approval of ofatumumab, for previously untreated CLL patients, is based on results from 2 trials, COMPLEMENT 1 and OMB115991.
The phase 3 COMPLEMENT 1 trial was a comparison of ofatumumab plus chlorambucil (n=221) with chlorambucil alone (n=226) in CLL patients for whom fludarabine-based treatment was considered inappropriate.
In this study, ofatumumab plus chlorambucil improved progression-free survival compared to chlorambucil alone. The median times were 22.4 months and 13.1 months, respectively, and the hazard ratio was 0.57 (P<0.001).
In the phase 2 trial known as OMB115991, researchers evaluated ofatumumab in combination with bendamustine in 44 patients with previously untreated CLL for whom fludarabine-based treatment was considered inappropriate.
The combination elicited an overall response rate of 95% and a complete response rate of 43%.
The overall safety profile of ofatumumab in CLL (previously untreated and relapsed/refractory) is based on data from 511 patients in clinical trials.
This includes 250 patients with relapsed/refractory CLL who were treated with ofatumumab alone and 261 patients with previously untreated CLL who were treated in combination with an alkylating agent.
The most common adverse effects associated with ofatumumab were infusion reactions, neutropenia, anemia, febrile neutropenia, thrombocytopenia, leukopenia, lower respiratory tract infection (including pneumonia), upper respiratory tract infection, sepsis (including neutropenic sepsis and septic shock), herpes virus infection, and urinary tract infection.