User login
Isavuconazole resolved invasive fungal disease in patients on ibrutinib
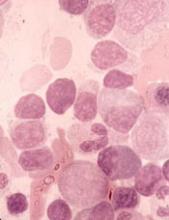
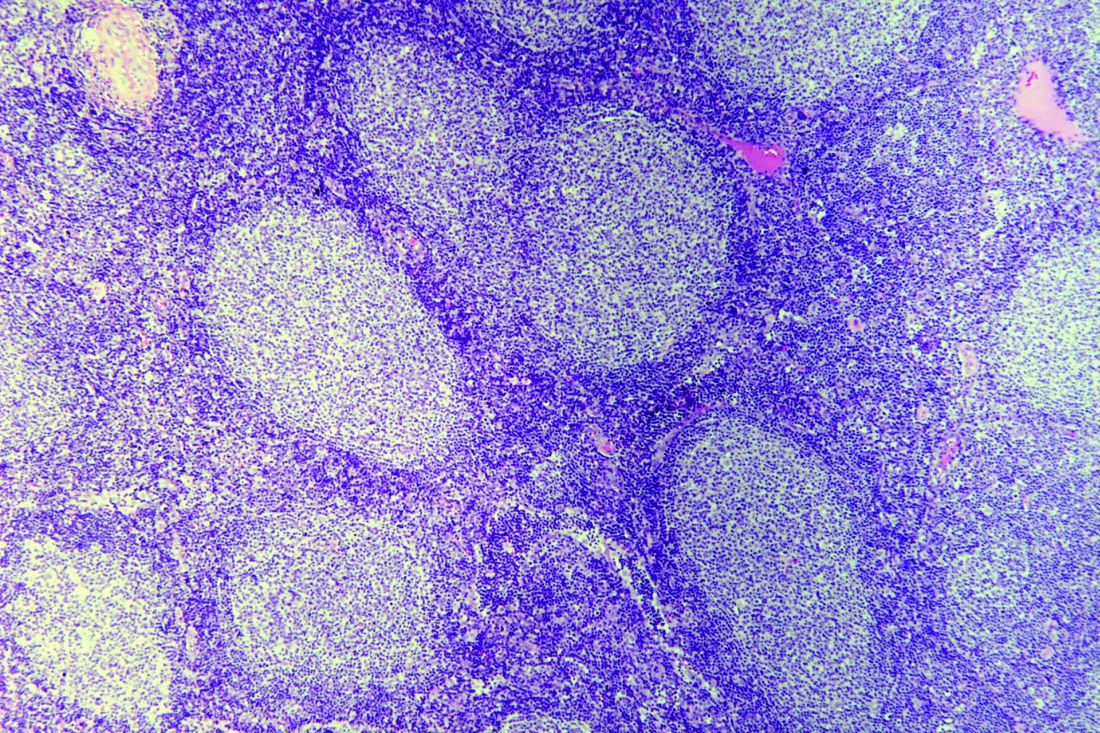
Treatment with isavuconazole resolved or substantially improved invasive fungal disease among seven of eight patients receiving concomitant ibrutinib, according to the results of a small two-center study.
The combination “was well-tolerated overall,” wrote Kaelyn C. Cummins of Brigham and Women’s Hospital, together with her associates there and at the Dana-Farber Cancer Institute, Boston. Their letter to the editor was published in Leukemia & Lymphoma.
Although ibrutinib is considered less immunosuppressive than conventional chemotherapy, it has been tied to invasive fungal infections, even in seemingly low-risk patients. The preferred treatment, voriconazole, is a very strong inhibitor of cytochrome P450 systems, of which ibrutinib is a substrate. For this study, the researchers queried the pharmacy databases of their institutions to identify adults who received concomitant isavuconazole (200 mg per day) and ibrutinib between 2015 and 2018. Drug exposures were confirmed by medical record review.
Four patients experienced clinical and radiologic resolution of invasive aspergillosis, fusariosis, mucormycosis, or phaeohyphomycosis. Another three had clinical and radiologic improvement of confirmed or probable aspergillosis or histoplasmosis. One of these patients underwent five debridements for central nervous system invasive aspergillosis but had 8 months of clinical improvement between debridements. This patient’s fungal isolate remained susceptible to isavuconazole throughout treatment. The patient who did not respond at all to isavuconazole had invasive aspergillosis with recurrent brain abscesses. The fungal isolate remained susceptible to isavuconazole, and the patient switched to long-term voriconazole therapy after stopping ibrutinib.
Several adverse events occurred while patients were on concomitant therapy. One patient developed paroxysmal atrial fibrillation that persisted after stopping ibrutinib. Another had worsening of preexisting thrombocytopenia. Among four patients with electrocardiogram data, two had transient QTc prolongation. No patient died within 12 weeks of starting concomitant therapy. Two patients eventually died after their cancer progressed.
The median age of the patients was 60 years (range, 38-76 years). Five were men. Six had chronic lymphocytic leukemia (CLL) and two had marginal zone lymphoma. Two CLL patients were on ibrutinib monotherapy, two also received rituximab, one also received umbralisib, and one also received obinutuzumab. One patient with marginal zone lymphoma was on ibrutinib monotherapy, and the other received concomitant rituximab, gemcitabine, dexamethasone, and cisplatin.
Researchers should study the mechanisms by which [Bruton’s tyrosine kinase] inhibitors might increase susceptibility to fungal infections among patients with lymphoma or CLL, said Ms. Cummins and her associates. Because the CYP3A enzyme system also metabolizes PI3K and BCL-2 inhibitors, their results “could be more broadly applicable.”
Ms. Cummins had no disclosures.
SOURCE: Cummins KC et al. Leuk. Lymphoma 2018 Jul 24. doi: 10.1080/10428194.2018.1485913.
Treatment with isavuconazole resolved or substantially improved invasive fungal disease among seven of eight patients receiving concomitant ibrutinib, according to the results of a small two-center study.
The combination “was well-tolerated overall,” wrote Kaelyn C. Cummins of Brigham and Women’s Hospital, together with her associates there and at the Dana-Farber Cancer Institute, Boston. Their letter to the editor was published in Leukemia & Lymphoma.
Although ibrutinib is considered less immunosuppressive than conventional chemotherapy, it has been tied to invasive fungal infections, even in seemingly low-risk patients. The preferred treatment, voriconazole, is a very strong inhibitor of cytochrome P450 systems, of which ibrutinib is a substrate. For this study, the researchers queried the pharmacy databases of their institutions to identify adults who received concomitant isavuconazole (200 mg per day) and ibrutinib between 2015 and 2018. Drug exposures were confirmed by medical record review.
Four patients experienced clinical and radiologic resolution of invasive aspergillosis, fusariosis, mucormycosis, or phaeohyphomycosis. Another three had clinical and radiologic improvement of confirmed or probable aspergillosis or histoplasmosis. One of these patients underwent five debridements for central nervous system invasive aspergillosis but had 8 months of clinical improvement between debridements. This patient’s fungal isolate remained susceptible to isavuconazole throughout treatment. The patient who did not respond at all to isavuconazole had invasive aspergillosis with recurrent brain abscesses. The fungal isolate remained susceptible to isavuconazole, and the patient switched to long-term voriconazole therapy after stopping ibrutinib.
Several adverse events occurred while patients were on concomitant therapy. One patient developed paroxysmal atrial fibrillation that persisted after stopping ibrutinib. Another had worsening of preexisting thrombocytopenia. Among four patients with electrocardiogram data, two had transient QTc prolongation. No patient died within 12 weeks of starting concomitant therapy. Two patients eventually died after their cancer progressed.
The median age of the patients was 60 years (range, 38-76 years). Five were men. Six had chronic lymphocytic leukemia (CLL) and two had marginal zone lymphoma. Two CLL patients were on ibrutinib monotherapy, two also received rituximab, one also received umbralisib, and one also received obinutuzumab. One patient with marginal zone lymphoma was on ibrutinib monotherapy, and the other received concomitant rituximab, gemcitabine, dexamethasone, and cisplatin.
Researchers should study the mechanisms by which [Bruton’s tyrosine kinase] inhibitors might increase susceptibility to fungal infections among patients with lymphoma or CLL, said Ms. Cummins and her associates. Because the CYP3A enzyme system also metabolizes PI3K and BCL-2 inhibitors, their results “could be more broadly applicable.”
Ms. Cummins had no disclosures.
SOURCE: Cummins KC et al. Leuk. Lymphoma 2018 Jul 24. doi: 10.1080/10428194.2018.1485913.
Treatment with isavuconazole resolved or substantially improved invasive fungal disease among seven of eight patients receiving concomitant ibrutinib, according to the results of a small two-center study.
The combination “was well-tolerated overall,” wrote Kaelyn C. Cummins of Brigham and Women’s Hospital, together with her associates there and at the Dana-Farber Cancer Institute, Boston. Their letter to the editor was published in Leukemia & Lymphoma.
Although ibrutinib is considered less immunosuppressive than conventional chemotherapy, it has been tied to invasive fungal infections, even in seemingly low-risk patients. The preferred treatment, voriconazole, is a very strong inhibitor of cytochrome P450 systems, of which ibrutinib is a substrate. For this study, the researchers queried the pharmacy databases of their institutions to identify adults who received concomitant isavuconazole (200 mg per day) and ibrutinib between 2015 and 2018. Drug exposures were confirmed by medical record review.
Four patients experienced clinical and radiologic resolution of invasive aspergillosis, fusariosis, mucormycosis, or phaeohyphomycosis. Another three had clinical and radiologic improvement of confirmed or probable aspergillosis or histoplasmosis. One of these patients underwent five debridements for central nervous system invasive aspergillosis but had 8 months of clinical improvement between debridements. This patient’s fungal isolate remained susceptible to isavuconazole throughout treatment. The patient who did not respond at all to isavuconazole had invasive aspergillosis with recurrent brain abscesses. The fungal isolate remained susceptible to isavuconazole, and the patient switched to long-term voriconazole therapy after stopping ibrutinib.
Several adverse events occurred while patients were on concomitant therapy. One patient developed paroxysmal atrial fibrillation that persisted after stopping ibrutinib. Another had worsening of preexisting thrombocytopenia. Among four patients with electrocardiogram data, two had transient QTc prolongation. No patient died within 12 weeks of starting concomitant therapy. Two patients eventually died after their cancer progressed.
The median age of the patients was 60 years (range, 38-76 years). Five were men. Six had chronic lymphocytic leukemia (CLL) and two had marginal zone lymphoma. Two CLL patients were on ibrutinib monotherapy, two also received rituximab, one also received umbralisib, and one also received obinutuzumab. One patient with marginal zone lymphoma was on ibrutinib monotherapy, and the other received concomitant rituximab, gemcitabine, dexamethasone, and cisplatin.
Researchers should study the mechanisms by which [Bruton’s tyrosine kinase] inhibitors might increase susceptibility to fungal infections among patients with lymphoma or CLL, said Ms. Cummins and her associates. Because the CYP3A enzyme system also metabolizes PI3K and BCL-2 inhibitors, their results “could be more broadly applicable.”
Ms. Cummins had no disclosures.
SOURCE: Cummins KC et al. Leuk. Lymphoma 2018 Jul 24. doi: 10.1080/10428194.2018.1485913.
FROM LEUKEMIA & LYMPHOMA
Key clinical point: Treatment with isavuconazole resolved or substantially improved invasive fungal disease in patients receiving concomitant ibrutinib.
Major finding: Seven of eight patients experienced clinical and radiographic resolution or improvement. Adverse events of concomitant treatment included paroxysmal atrial fibrillation, worsening of baseline thrombocytopenia, and QTc interval prolongation.
Study details: Retrospective study at two centers.
Disclosures: The article did not include information on funding sources or conflicts of interests.
Source: Cummins KC. et al. Leuk. Lymphoma 2018 Jul 24. doi: 10.1080/10428194.2018.1485913.
Global burden of hematologic malignancies
Research has shown an increase in the global incidence of leukemia and non-Hodgkin lymphoma (NHL) in recent years.
The Global Burden of Disease (GBD) study showed that, from 2006 to 2016, the incidence of NHL increased 45%, and the incidence of leukemia increased 26%.
These increases were largely due to population growth and aging.
Results from the GDB study were published in JAMA Oncology.
The study indicated that, in 2016, there were 17.2 million cases of cancer worldwide and 8.9 million cancer deaths.
One in 3 men were likely to get cancer during their lifetime, as were 1 in 5 women. Cancer was associated with 213.2 million disability-adjusted life years (DALYs).
The following table lists the 2016 global incidence and mortality figures for all cancers combined and for individual hematologic malignancies.
| Cancer type | Cases, thousands | Deaths, thousands |
| All cancers | 17,228 | 8927 |
| Leukemias | 467 | 310 |
| Acute lymphoid leukemia | 76 | 51 |
| Chronic lymphoid leukemia | 105 | 35 |
| Acute myeloid leukemia | 103 | 85 |
| Chronic myeloid leukemia | 32 | 22 |
| Other leukemias | 150 | 117 |
| Hodgkin lymphoma | 73 | 29 |
| NHL | 461 | 240 |
| Multiple myeloma | 139 | 98 |
Leukemia
In 2016, there were 467,000 new cases of leukemia and 310,000 leukemia deaths. Leukemia was responsible for 10.2 million DALYs. Leukemia developed in 1 in 118 men and 1 in 194 women worldwide.
Between 2006 and 2016, the global leukemia incidence increased by 26%—from 370,482 to 466,802 cases.
The researchers said the factors contributing to this increase were population growth (12%), population aging (10%), and an increase in age-specific incidence rates (3%).
NHL
In 2016, there were 461,000 new cases of NHL and 240,000 NHL deaths. NHL was responsible for 6.8 million DALYs. NHL developed in 1 in 110 men and 1 in 161 women worldwide.
Between 2006 and 2016, NHL increased by 45%, from 319,078 to 461,164 cases.
The factors contributing to this increase were increasing age-specific incidence rates (17%), changing population age structure (15%), and population growth (12%).
“A large proportion of the increase in cancer incidence can be explained by improving life expectancy and population growth—a development that can at least partially be attributed to a reduced burden from other common diseases,” the study authors wrote.
The authors also pointed out that prevention efforts are less effective for hematologic malignancies than for other cancers.
Research has shown an increase in the global incidence of leukemia and non-Hodgkin lymphoma (NHL) in recent years.
The Global Burden of Disease (GBD) study showed that, from 2006 to 2016, the incidence of NHL increased 45%, and the incidence of leukemia increased 26%.
These increases were largely due to population growth and aging.
Results from the GDB study were published in JAMA Oncology.
The study indicated that, in 2016, there were 17.2 million cases of cancer worldwide and 8.9 million cancer deaths.
One in 3 men were likely to get cancer during their lifetime, as were 1 in 5 women. Cancer was associated with 213.2 million disability-adjusted life years (DALYs).
The following table lists the 2016 global incidence and mortality figures for all cancers combined and for individual hematologic malignancies.
| Cancer type | Cases, thousands | Deaths, thousands |
| All cancers | 17,228 | 8927 |
| Leukemias | 467 | 310 |
| Acute lymphoid leukemia | 76 | 51 |
| Chronic lymphoid leukemia | 105 | 35 |
| Acute myeloid leukemia | 103 | 85 |
| Chronic myeloid leukemia | 32 | 22 |
| Other leukemias | 150 | 117 |
| Hodgkin lymphoma | 73 | 29 |
| NHL | 461 | 240 |
| Multiple myeloma | 139 | 98 |
Leukemia
In 2016, there were 467,000 new cases of leukemia and 310,000 leukemia deaths. Leukemia was responsible for 10.2 million DALYs. Leukemia developed in 1 in 118 men and 1 in 194 women worldwide.
Between 2006 and 2016, the global leukemia incidence increased by 26%—from 370,482 to 466,802 cases.
The researchers said the factors contributing to this increase were population growth (12%), population aging (10%), and an increase in age-specific incidence rates (3%).
NHL
In 2016, there were 461,000 new cases of NHL and 240,000 NHL deaths. NHL was responsible for 6.8 million DALYs. NHL developed in 1 in 110 men and 1 in 161 women worldwide.
Between 2006 and 2016, NHL increased by 45%, from 319,078 to 461,164 cases.
The factors contributing to this increase were increasing age-specific incidence rates (17%), changing population age structure (15%), and population growth (12%).
“A large proportion of the increase in cancer incidence can be explained by improving life expectancy and population growth—a development that can at least partially be attributed to a reduced burden from other common diseases,” the study authors wrote.
The authors also pointed out that prevention efforts are less effective for hematologic malignancies than for other cancers.
Research has shown an increase in the global incidence of leukemia and non-Hodgkin lymphoma (NHL) in recent years.
The Global Burden of Disease (GBD) study showed that, from 2006 to 2016, the incidence of NHL increased 45%, and the incidence of leukemia increased 26%.
These increases were largely due to population growth and aging.
Results from the GDB study were published in JAMA Oncology.
The study indicated that, in 2016, there were 17.2 million cases of cancer worldwide and 8.9 million cancer deaths.
One in 3 men were likely to get cancer during their lifetime, as were 1 in 5 women. Cancer was associated with 213.2 million disability-adjusted life years (DALYs).
The following table lists the 2016 global incidence and mortality figures for all cancers combined and for individual hematologic malignancies.
| Cancer type | Cases, thousands | Deaths, thousands |
| All cancers | 17,228 | 8927 |
| Leukemias | 467 | 310 |
| Acute lymphoid leukemia | 76 | 51 |
| Chronic lymphoid leukemia | 105 | 35 |
| Acute myeloid leukemia | 103 | 85 |
| Chronic myeloid leukemia | 32 | 22 |
| Other leukemias | 150 | 117 |
| Hodgkin lymphoma | 73 | 29 |
| NHL | 461 | 240 |
| Multiple myeloma | 139 | 98 |
Leukemia
In 2016, there were 467,000 new cases of leukemia and 310,000 leukemia deaths. Leukemia was responsible for 10.2 million DALYs. Leukemia developed in 1 in 118 men and 1 in 194 women worldwide.
Between 2006 and 2016, the global leukemia incidence increased by 26%—from 370,482 to 466,802 cases.
The researchers said the factors contributing to this increase were population growth (12%), population aging (10%), and an increase in age-specific incidence rates (3%).
NHL
In 2016, there were 461,000 new cases of NHL and 240,000 NHL deaths. NHL was responsible for 6.8 million DALYs. NHL developed in 1 in 110 men and 1 in 161 women worldwide.
Between 2006 and 2016, NHL increased by 45%, from 319,078 to 461,164 cases.
The factors contributing to this increase were increasing age-specific incidence rates (17%), changing population age structure (15%), and population growth (12%).
“A large proportion of the increase in cancer incidence can be explained by improving life expectancy and population growth—a development that can at least partially be attributed to a reduced burden from other common diseases,” the study authors wrote.
The authors also pointed out that prevention efforts are less effective for hematologic malignancies than for other cancers.
Late mortality risk after childhood BMT is substantial, persistent
Children who undergo allogeneic blood or marrow transplantation (BMT) remain at an elevated risk of premature death even 25 years after the procedure, results of large, retrospective cohort study suggest.
Despite a significant decrease over several decades, the risk of all-cause mortality remained elevated, compared with the general population, according to this study of individuals who had BMT performed in childhood between 1974 and 2010.
“These findings emphasize the need for lifelong follow-up care after allogeneic BMT performed in childhood,” reported Anna Sällfors Holmqvist, MD, PhD, of the department of clinical sciences at Skåne University Hospital, Lund University, Sweden, and her associates.
, Dr. Holmqvist and her colleagues reported in JAMA Oncology.
Their retrospective analysis included 1,388 individuals who lived at least 2 years after allogeneic BMT performed in childhood at one of three centers: the University of Alabama at Birmingham; the University of Minnesota, Minneapolis; and City of Hope, Duarte, Calif.
There were 295 deaths over a median of 14.9 years of follow-up, for an overall survival rate of 79.3% at 20 years after BMT, reported Dr. Holmqvist and her associates. The three leading causes of death were infection or chronic graft-versus-host disease in 49.6% of cases, primary disease in 24.6%, and later malignancies in 18.4%.
Relative to the general population, the cohort had a 14.4-fold increased risk of premature death (95% confidence interval, 12.8-16.1), compared with the general population. Relative mortality was highest 2-5 years after BMT and dropped substantially after that but remained elevated – even 25 years or more after the procedure, the investigators noted.
Mortality decreased significantly over the 3 decades evaluated in this study. The rate of all-cause, 10-year cumulative mortality was 18.9% before 1990, 12.9% from 1990 to 1999, and 11.0% from 2000 to 2010 (P = .002).
That decrease in cumulative mortality over time could not be explained by changes in transplant practice over those three time periods, according to results of a mediation analysis performed by Dr. Holmqvist and her associates.
That finding suggests that unmeasured variables might underlie the decrease in late mortality, the investigators said.
Those unmeasured variables might include supportive care strategies, management of chronic graft-versus-host disease, or improved patient selection, they noted.
Dr. Holmqvist and her associates cited as one limitation their reliance on death certificates for causes of death. In addition, the causes of death for 51 of the 295 deceased patients were lacking.
The study was supported in part by grants from the National Cancer Institute, the Leukemia Lymphoma Society, and the Swedish Childhood Cancer Foundation. Dr. Holmqvist and her associates reported no conflicts of interest.
SOURCE: Holmqvist AS et al. JAMA Oncol. 2018 Jul 26. doi: 10.1001/jamaoncol.2018.2453.
Children who undergo allogeneic blood or marrow transplantation (BMT) remain at an elevated risk of premature death even 25 years after the procedure, results of large, retrospective cohort study suggest.
Despite a significant decrease over several decades, the risk of all-cause mortality remained elevated, compared with the general population, according to this study of individuals who had BMT performed in childhood between 1974 and 2010.
“These findings emphasize the need for lifelong follow-up care after allogeneic BMT performed in childhood,” reported Anna Sällfors Holmqvist, MD, PhD, of the department of clinical sciences at Skåne University Hospital, Lund University, Sweden, and her associates.
, Dr. Holmqvist and her colleagues reported in JAMA Oncology.
Their retrospective analysis included 1,388 individuals who lived at least 2 years after allogeneic BMT performed in childhood at one of three centers: the University of Alabama at Birmingham; the University of Minnesota, Minneapolis; and City of Hope, Duarte, Calif.
There were 295 deaths over a median of 14.9 years of follow-up, for an overall survival rate of 79.3% at 20 years after BMT, reported Dr. Holmqvist and her associates. The three leading causes of death were infection or chronic graft-versus-host disease in 49.6% of cases, primary disease in 24.6%, and later malignancies in 18.4%.
Relative to the general population, the cohort had a 14.4-fold increased risk of premature death (95% confidence interval, 12.8-16.1), compared with the general population. Relative mortality was highest 2-5 years after BMT and dropped substantially after that but remained elevated – even 25 years or more after the procedure, the investigators noted.
Mortality decreased significantly over the 3 decades evaluated in this study. The rate of all-cause, 10-year cumulative mortality was 18.9% before 1990, 12.9% from 1990 to 1999, and 11.0% from 2000 to 2010 (P = .002).
That decrease in cumulative mortality over time could not be explained by changes in transplant practice over those three time periods, according to results of a mediation analysis performed by Dr. Holmqvist and her associates.
That finding suggests that unmeasured variables might underlie the decrease in late mortality, the investigators said.
Those unmeasured variables might include supportive care strategies, management of chronic graft-versus-host disease, or improved patient selection, they noted.
Dr. Holmqvist and her associates cited as one limitation their reliance on death certificates for causes of death. In addition, the causes of death for 51 of the 295 deceased patients were lacking.
The study was supported in part by grants from the National Cancer Institute, the Leukemia Lymphoma Society, and the Swedish Childhood Cancer Foundation. Dr. Holmqvist and her associates reported no conflicts of interest.
SOURCE: Holmqvist AS et al. JAMA Oncol. 2018 Jul 26. doi: 10.1001/jamaoncol.2018.2453.
Children who undergo allogeneic blood or marrow transplantation (BMT) remain at an elevated risk of premature death even 25 years after the procedure, results of large, retrospective cohort study suggest.
Despite a significant decrease over several decades, the risk of all-cause mortality remained elevated, compared with the general population, according to this study of individuals who had BMT performed in childhood between 1974 and 2010.
“These findings emphasize the need for lifelong follow-up care after allogeneic BMT performed in childhood,” reported Anna Sällfors Holmqvist, MD, PhD, of the department of clinical sciences at Skåne University Hospital, Lund University, Sweden, and her associates.
, Dr. Holmqvist and her colleagues reported in JAMA Oncology.
Their retrospective analysis included 1,388 individuals who lived at least 2 years after allogeneic BMT performed in childhood at one of three centers: the University of Alabama at Birmingham; the University of Minnesota, Minneapolis; and City of Hope, Duarte, Calif.
There were 295 deaths over a median of 14.9 years of follow-up, for an overall survival rate of 79.3% at 20 years after BMT, reported Dr. Holmqvist and her associates. The three leading causes of death were infection or chronic graft-versus-host disease in 49.6% of cases, primary disease in 24.6%, and later malignancies in 18.4%.
Relative to the general population, the cohort had a 14.4-fold increased risk of premature death (95% confidence interval, 12.8-16.1), compared with the general population. Relative mortality was highest 2-5 years after BMT and dropped substantially after that but remained elevated – even 25 years or more after the procedure, the investigators noted.
Mortality decreased significantly over the 3 decades evaluated in this study. The rate of all-cause, 10-year cumulative mortality was 18.9% before 1990, 12.9% from 1990 to 1999, and 11.0% from 2000 to 2010 (P = .002).
That decrease in cumulative mortality over time could not be explained by changes in transplant practice over those three time periods, according to results of a mediation analysis performed by Dr. Holmqvist and her associates.
That finding suggests that unmeasured variables might underlie the decrease in late mortality, the investigators said.
Those unmeasured variables might include supportive care strategies, management of chronic graft-versus-host disease, or improved patient selection, they noted.
Dr. Holmqvist and her associates cited as one limitation their reliance on death certificates for causes of death. In addition, the causes of death for 51 of the 295 deceased patients were lacking.
The study was supported in part by grants from the National Cancer Institute, the Leukemia Lymphoma Society, and the Swedish Childhood Cancer Foundation. Dr. Holmqvist and her associates reported no conflicts of interest.
SOURCE: Holmqvist AS et al. JAMA Oncol. 2018 Jul 26. doi: 10.1001/jamaoncol.2018.2453.
FROM JAMA ONCOLOGY
Key clinical point: Individuals undergoing allogeneic blood or marrow transplantation (BMT) in childhood require careful follow-up for many years because of a persistent elevated risk of premature death.
Major finding: Risk of premature death was increased 14.4-fold, compared with the general population (95% confidence interval, 12.8-16.1).
Study details: A retrospective cohort study including 1,388 individuals living 2 years or more after allogeneic BMT performed in childhood.
Disclosures: The study was supported in part by grants from the National Cancer Institute, the Leukemia Lymphoma Society, and the Swedish Childhood Cancer Foundation. Dr. Holmqvist and her coauthors reported no conflicts of interest.
Source: Holmqvist AS et al. JAMA Oncol. 2018 Jul 26. doi: 10.1001/jamaoncol.2018.2453.
Drug receives fast track designation for WM
The US Food and Drug Administration (FDA) has granted fast track designation to zanubrutinib for the treatment of Waldenström’s macroglobulinemia (WM).
Zanubrutinib (BGB-3111) is a BTK inhibitor being developed by BeiGene to treat various B-cell malignancies.
BeiGene is preparing to submit to the FDA, in the first half of 2019, a new drug application seeking accelerated approval of zanubrutinib for patients with WM.
The application will be supported by results from a phase 1 study. Results from this trial were presented at the 14th International Conference on Malignant Lymphoma (14-ICML) last year.
Researchers are also evaluating zanubrutinib in phase 2 (NCT03332173) and phase 3 (NCT03053440) trials of WM patients. In the phase 3 trial, researchers are comparing zanubrutinib to the BTK inhibitor ibrutinib.
Phase 1 results
As of March 31, 2017, 48 WM patients were enrolled in the phase 1 study. Thirty-eight patients had relapsed/refractory disease, and 10 patients were treatment-naïve.
There was a dose-escalation phase and a dose-expansion phase. The dose-expansion phase included doses of 160 mg twice a day or 320 mg once a day.
The most common (>10%) adverse events, (AEs) of any attribution were petechiae/purpura/contusion (35%), upper respiratory tract infection (31%), constipation (25%), diarrhea (19%), epistaxis (19%), nausea (17%), cough (15%), anemia (15%), headache (15%), neutropenia (13%), and rash (13%).
Most of these events were grade 1 or 2 in severity. The exceptions were grade 3/4 anemia and neutropenia (8% each) as well as grade 3/4 diarrhea and headache (2% each).
Five serious AEs were considered possibly related to zanubrutinib—1 case each of hemothorax, atrial fibrillation, colitis, febrile neutropenia, and headache. Three AEs led to treatment discontinuation—1 case each of bronchiectasis, prostate adenocarcinoma, and adenocarcinoma of pylorus.
At the time of the data cutoff, 42 patients were evaluable for response. At a median follow-up of 12.3 months (range, 4.4 to 30.5 months), the overall response rate was 90% (38/42).
The major response rate was 76% (32/42), with very good partial responses in 43% (18/42) of patients and partial responses in 33% (14/42) of patients. There were no complete responses and 2 cases of disease progression.
About fast track designation
The FDA’s fast track development program is designed to expedite clinical development and submission of applications for products with the potential to treat serious or life-threatening conditions and address unmet medical needs.
Fast track designation facilitates frequent interactions with the FDA review team, including meetings to discuss the product’s development plan and written communications about issues such as trial design and use of biomarkers.
Products that receive fast track designation may be eligible for accelerated approval and priority review if relevant criteria are met. Such products may also be eligible for rolling review, which allows a developer to submit individual sections of a product’s application for review as they are ready, rather than waiting until all sections are complete.
The US Food and Drug Administration (FDA) has granted fast track designation to zanubrutinib for the treatment of Waldenström’s macroglobulinemia (WM).
Zanubrutinib (BGB-3111) is a BTK inhibitor being developed by BeiGene to treat various B-cell malignancies.
BeiGene is preparing to submit to the FDA, in the first half of 2019, a new drug application seeking accelerated approval of zanubrutinib for patients with WM.
The application will be supported by results from a phase 1 study. Results from this trial were presented at the 14th International Conference on Malignant Lymphoma (14-ICML) last year.
Researchers are also evaluating zanubrutinib in phase 2 (NCT03332173) and phase 3 (NCT03053440) trials of WM patients. In the phase 3 trial, researchers are comparing zanubrutinib to the BTK inhibitor ibrutinib.
Phase 1 results
As of March 31, 2017, 48 WM patients were enrolled in the phase 1 study. Thirty-eight patients had relapsed/refractory disease, and 10 patients were treatment-naïve.
There was a dose-escalation phase and a dose-expansion phase. The dose-expansion phase included doses of 160 mg twice a day or 320 mg once a day.
The most common (>10%) adverse events, (AEs) of any attribution were petechiae/purpura/contusion (35%), upper respiratory tract infection (31%), constipation (25%), diarrhea (19%), epistaxis (19%), nausea (17%), cough (15%), anemia (15%), headache (15%), neutropenia (13%), and rash (13%).
Most of these events were grade 1 or 2 in severity. The exceptions were grade 3/4 anemia and neutropenia (8% each) as well as grade 3/4 diarrhea and headache (2% each).
Five serious AEs were considered possibly related to zanubrutinib—1 case each of hemothorax, atrial fibrillation, colitis, febrile neutropenia, and headache. Three AEs led to treatment discontinuation—1 case each of bronchiectasis, prostate adenocarcinoma, and adenocarcinoma of pylorus.
At the time of the data cutoff, 42 patients were evaluable for response. At a median follow-up of 12.3 months (range, 4.4 to 30.5 months), the overall response rate was 90% (38/42).
The major response rate was 76% (32/42), with very good partial responses in 43% (18/42) of patients and partial responses in 33% (14/42) of patients. There were no complete responses and 2 cases of disease progression.
About fast track designation
The FDA’s fast track development program is designed to expedite clinical development and submission of applications for products with the potential to treat serious or life-threatening conditions and address unmet medical needs.
Fast track designation facilitates frequent interactions with the FDA review team, including meetings to discuss the product’s development plan and written communications about issues such as trial design and use of biomarkers.
Products that receive fast track designation may be eligible for accelerated approval and priority review if relevant criteria are met. Such products may also be eligible for rolling review, which allows a developer to submit individual sections of a product’s application for review as they are ready, rather than waiting until all sections are complete.
The US Food and Drug Administration (FDA) has granted fast track designation to zanubrutinib for the treatment of Waldenström’s macroglobulinemia (WM).
Zanubrutinib (BGB-3111) is a BTK inhibitor being developed by BeiGene to treat various B-cell malignancies.
BeiGene is preparing to submit to the FDA, in the first half of 2019, a new drug application seeking accelerated approval of zanubrutinib for patients with WM.
The application will be supported by results from a phase 1 study. Results from this trial were presented at the 14th International Conference on Malignant Lymphoma (14-ICML) last year.
Researchers are also evaluating zanubrutinib in phase 2 (NCT03332173) and phase 3 (NCT03053440) trials of WM patients. In the phase 3 trial, researchers are comparing zanubrutinib to the BTK inhibitor ibrutinib.
Phase 1 results
As of March 31, 2017, 48 WM patients were enrolled in the phase 1 study. Thirty-eight patients had relapsed/refractory disease, and 10 patients were treatment-naïve.
There was a dose-escalation phase and a dose-expansion phase. The dose-expansion phase included doses of 160 mg twice a day or 320 mg once a day.
The most common (>10%) adverse events, (AEs) of any attribution were petechiae/purpura/contusion (35%), upper respiratory tract infection (31%), constipation (25%), diarrhea (19%), epistaxis (19%), nausea (17%), cough (15%), anemia (15%), headache (15%), neutropenia (13%), and rash (13%).
Most of these events were grade 1 or 2 in severity. The exceptions were grade 3/4 anemia and neutropenia (8% each) as well as grade 3/4 diarrhea and headache (2% each).
Five serious AEs were considered possibly related to zanubrutinib—1 case each of hemothorax, atrial fibrillation, colitis, febrile neutropenia, and headache. Three AEs led to treatment discontinuation—1 case each of bronchiectasis, prostate adenocarcinoma, and adenocarcinoma of pylorus.
At the time of the data cutoff, 42 patients were evaluable for response. At a median follow-up of 12.3 months (range, 4.4 to 30.5 months), the overall response rate was 90% (38/42).
The major response rate was 76% (32/42), with very good partial responses in 43% (18/42) of patients and partial responses in 33% (14/42) of patients. There were no complete responses and 2 cases of disease progression.
About fast track designation
The FDA’s fast track development program is designed to expedite clinical development and submission of applications for products with the potential to treat serious or life-threatening conditions and address unmet medical needs.
Fast track designation facilitates frequent interactions with the FDA review team, including meetings to discuss the product’s development plan and written communications about issues such as trial design and use of biomarkers.
Products that receive fast track designation may be eligible for accelerated approval and priority review if relevant criteria are met. Such products may also be eligible for rolling review, which allows a developer to submit individual sections of a product’s application for review as they are ready, rather than waiting until all sections are complete.
Rituximab reduces risk of follicular lymphoma transformation
Rituximab-based chemotherapy can significantly reduce the risk of transformation of follicular lymphoma (FL) from an indolent to an aggressive histology, such as diffuse large B-cell lymphoma, results of a retrospective pooled analysis have suggested.
“Despite the intrinsic limitations related to the retrospective nature of our study, we confirmed that the cumulative hazard of histological transformation as a first event in follicular lymphoma can be reduced significantly by introducing rituximab to a backbone therapy. Moreover, our data also confirm that histological transformation still has an adverse effect on patient outcome, although it is less catastrophic than the pre-rituximab regimens,” they wrote in the Lancet Haematology.
These investigators, from 11 cooperative groups or institutions across Europe, pooled data on patients aged 18 years and older who had a histologically confirmed diagnosis of grade 1, 2, or 3a FL between Jan. 2, 1997, and Dec. 20, 2013.
They defined histologic transformation as a biopsy-proven aggressive lymphoma that occurred as a first event after first-line therapy.
Data on a total of 8,116 patients were available for analysis; 509 of these patients had had histologic transformations. After a median follow-up of 87 months, the 10-year cumulative hazard for all patients was 7.7%. The 10-year cumulative hazard – one of two primary endpoints – was 5.2% for patients who had received any rituximab versus 8.7% for those who did not, which translated into a hazard ratio of 0.73 (P = .004).
Among patients who received rituximab during induction only, the 10-year cumulative hazard was 5.9%, and it was 3.6% among those who received rituximab during induction and maintenance phases of treatment. This difference translated into a HR of 0.55 (P = .003).
The benefit of rituximab induction and maintenance – compared with induction only – held up in a multivariate analysis controlling for age at diagnosis, sex, FLIPI (Follicular Lymphoma International Prognostic Index) score, active surveillance vs. treatment, and FL grade (HR, 0.55; P = .016).
There were 287 deaths among the 509 patients with transformation, resulting in a 10-year survival after transformation of 32%.
The 5-year survival after transformation was 38% for patients who were not exposed to rituximab, 42% for patients who received induction rituximab, and 43% for those who received both induction and maintenance rituximab, but the differences between the three groups were not statistically significant.
“More comprehensive knowledge of the biological risk factors for follicular lymphoma transformation and the molecular pathways involved is likely to help clinicians make more accurate prognostic assessments and also inform the potential usefulness of novel drugs for the treatment of follicular lymphoma,” the researchers wrote.
The study was funded by the European Lymphoma Institute and other research groups. The researchers reported having no financial disclosures.
SOURCE: Federico M et al. Lancet Haematol. 2018 Jul 4. doi: 10.1016/S2352-3026(18)30090-5.
Rituximab-based chemotherapy can significantly reduce the risk of transformation of follicular lymphoma (FL) from an indolent to an aggressive histology, such as diffuse large B-cell lymphoma, results of a retrospective pooled analysis have suggested.
“Despite the intrinsic limitations related to the retrospective nature of our study, we confirmed that the cumulative hazard of histological transformation as a first event in follicular lymphoma can be reduced significantly by introducing rituximab to a backbone therapy. Moreover, our data also confirm that histological transformation still has an adverse effect on patient outcome, although it is less catastrophic than the pre-rituximab regimens,” they wrote in the Lancet Haematology.
These investigators, from 11 cooperative groups or institutions across Europe, pooled data on patients aged 18 years and older who had a histologically confirmed diagnosis of grade 1, 2, or 3a FL between Jan. 2, 1997, and Dec. 20, 2013.
They defined histologic transformation as a biopsy-proven aggressive lymphoma that occurred as a first event after first-line therapy.
Data on a total of 8,116 patients were available for analysis; 509 of these patients had had histologic transformations. After a median follow-up of 87 months, the 10-year cumulative hazard for all patients was 7.7%. The 10-year cumulative hazard – one of two primary endpoints – was 5.2% for patients who had received any rituximab versus 8.7% for those who did not, which translated into a hazard ratio of 0.73 (P = .004).
Among patients who received rituximab during induction only, the 10-year cumulative hazard was 5.9%, and it was 3.6% among those who received rituximab during induction and maintenance phases of treatment. This difference translated into a HR of 0.55 (P = .003).
The benefit of rituximab induction and maintenance – compared with induction only – held up in a multivariate analysis controlling for age at diagnosis, sex, FLIPI (Follicular Lymphoma International Prognostic Index) score, active surveillance vs. treatment, and FL grade (HR, 0.55; P = .016).
There were 287 deaths among the 509 patients with transformation, resulting in a 10-year survival after transformation of 32%.
The 5-year survival after transformation was 38% for patients who were not exposed to rituximab, 42% for patients who received induction rituximab, and 43% for those who received both induction and maintenance rituximab, but the differences between the three groups were not statistically significant.
“More comprehensive knowledge of the biological risk factors for follicular lymphoma transformation and the molecular pathways involved is likely to help clinicians make more accurate prognostic assessments and also inform the potential usefulness of novel drugs for the treatment of follicular lymphoma,” the researchers wrote.
The study was funded by the European Lymphoma Institute and other research groups. The researchers reported having no financial disclosures.
SOURCE: Federico M et al. Lancet Haematol. 2018 Jul 4. doi: 10.1016/S2352-3026(18)30090-5.
Rituximab-based chemotherapy can significantly reduce the risk of transformation of follicular lymphoma (FL) from an indolent to an aggressive histology, such as diffuse large B-cell lymphoma, results of a retrospective pooled analysis have suggested.
“Despite the intrinsic limitations related to the retrospective nature of our study, we confirmed that the cumulative hazard of histological transformation as a first event in follicular lymphoma can be reduced significantly by introducing rituximab to a backbone therapy. Moreover, our data also confirm that histological transformation still has an adverse effect on patient outcome, although it is less catastrophic than the pre-rituximab regimens,” they wrote in the Lancet Haematology.
These investigators, from 11 cooperative groups or institutions across Europe, pooled data on patients aged 18 years and older who had a histologically confirmed diagnosis of grade 1, 2, or 3a FL between Jan. 2, 1997, and Dec. 20, 2013.
They defined histologic transformation as a biopsy-proven aggressive lymphoma that occurred as a first event after first-line therapy.
Data on a total of 8,116 patients were available for analysis; 509 of these patients had had histologic transformations. After a median follow-up of 87 months, the 10-year cumulative hazard for all patients was 7.7%. The 10-year cumulative hazard – one of two primary endpoints – was 5.2% for patients who had received any rituximab versus 8.7% for those who did not, which translated into a hazard ratio of 0.73 (P = .004).
Among patients who received rituximab during induction only, the 10-year cumulative hazard was 5.9%, and it was 3.6% among those who received rituximab during induction and maintenance phases of treatment. This difference translated into a HR of 0.55 (P = .003).
The benefit of rituximab induction and maintenance – compared with induction only – held up in a multivariate analysis controlling for age at diagnosis, sex, FLIPI (Follicular Lymphoma International Prognostic Index) score, active surveillance vs. treatment, and FL grade (HR, 0.55; P = .016).
There were 287 deaths among the 509 patients with transformation, resulting in a 10-year survival after transformation of 32%.
The 5-year survival after transformation was 38% for patients who were not exposed to rituximab, 42% for patients who received induction rituximab, and 43% for those who received both induction and maintenance rituximab, but the differences between the three groups were not statistically significant.
“More comprehensive knowledge of the biological risk factors for follicular lymphoma transformation and the molecular pathways involved is likely to help clinicians make more accurate prognostic assessments and also inform the potential usefulness of novel drugs for the treatment of follicular lymphoma,” the researchers wrote.
The study was funded by the European Lymphoma Institute and other research groups. The researchers reported having no financial disclosures.
SOURCE: Federico M et al. Lancet Haematol. 2018 Jul 4. doi: 10.1016/S2352-3026(18)30090-5.
FROM THE LANCET HAEMATOLOGY
Key clinical point:
Major finding: The 10-year cumulative hazard of histologic transformation was 5.2% for patients who had received rituximab and 8.7% for those who had not.
Study details: Retrospective pooled analysis of 8,116 patients with FL, 509 of whom had transformation over a 10-year period.
Disclosures: The study was funded by Associazione Angela Serra per la Ricerca sul Cancro, European Lymphoma Institute, European Hematology Association Lymphoma Group, Fondazione Italiana Linfomi, and the Spanish Group of Lymphoma and Bone Marrow Transplantation. The researchers reported having no financial disclosures.
Source: Federico M et al. Lancet Haematol. 2018 Jul 4. doi: 10.1016/S2352-3026(18)30090-5.
FDA approves biosimilar filgrastim
The US Food and Drug Administration (FDA) has approved the leukocyte growth factor Nivestym™ (filgrastim-aafi), a biosimilar to Neupogen (filgrastim).
Nivestym is approved to treat patients with nonmyeloid malignancies who are receiving myelosuppressive chemotherapy or undergoing bone marrow transplant, acute myeloid leukemia patients receiving induction or consolidation chemotherapy, patients undergoing autologous peripheral blood progenitor cell collection, and patients with severe chronic neutropenia.
The FDA’s approval of Nivestym was based on a review of evidence suggesting the drug is highly similar to Neupogen, according to Pfizer, the company developing Nivestym.
The full approved indication for Nivestym is as follows:
- To decrease the incidence of infection, as manifested by febrile neutropenia, in patients with nonmyeloid malignancies receiving myelosuppressive anticancer drugs associated with a significant incidence of severe neutropenia with fever
- To reduce the time to neutrophil recovery and the duration of fever following induction or consolidation chemotherapy in patients with acute myeloid leukemia
- To reduce the duration of neutropenia and neutropenia-related clinical sequelae (eg, febrile neutropenia) in patients with nonmyeloid malignancies undergoing myeloablative chemotherapy followed by bone marrow transplant
- For the mobilization of autologous hematopoietic progenitor cells into the peripheral blood for collection by leukapheresis
- For chronic administration to reduce the incidence and duration of sequelae of severe neutropenia (eg, fever, infections, oropharyngeal ulcers) in symptomatic patients with congenital neutropenia, cyclic neutropenia, or idiopathic neutropenia.
For more details on Nivestym, see the full prescribing information.
The US Food and Drug Administration (FDA) has approved the leukocyte growth factor Nivestym™ (filgrastim-aafi), a biosimilar to Neupogen (filgrastim).
Nivestym is approved to treat patients with nonmyeloid malignancies who are receiving myelosuppressive chemotherapy or undergoing bone marrow transplant, acute myeloid leukemia patients receiving induction or consolidation chemotherapy, patients undergoing autologous peripheral blood progenitor cell collection, and patients with severe chronic neutropenia.
The FDA’s approval of Nivestym was based on a review of evidence suggesting the drug is highly similar to Neupogen, according to Pfizer, the company developing Nivestym.
The full approved indication for Nivestym is as follows:
- To decrease the incidence of infection, as manifested by febrile neutropenia, in patients with nonmyeloid malignancies receiving myelosuppressive anticancer drugs associated with a significant incidence of severe neutropenia with fever
- To reduce the time to neutrophil recovery and the duration of fever following induction or consolidation chemotherapy in patients with acute myeloid leukemia
- To reduce the duration of neutropenia and neutropenia-related clinical sequelae (eg, febrile neutropenia) in patients with nonmyeloid malignancies undergoing myeloablative chemotherapy followed by bone marrow transplant
- For the mobilization of autologous hematopoietic progenitor cells into the peripheral blood for collection by leukapheresis
- For chronic administration to reduce the incidence and duration of sequelae of severe neutropenia (eg, fever, infections, oropharyngeal ulcers) in symptomatic patients with congenital neutropenia, cyclic neutropenia, or idiopathic neutropenia.
For more details on Nivestym, see the full prescribing information.
The US Food and Drug Administration (FDA) has approved the leukocyte growth factor Nivestym™ (filgrastim-aafi), a biosimilar to Neupogen (filgrastim).
Nivestym is approved to treat patients with nonmyeloid malignancies who are receiving myelosuppressive chemotherapy or undergoing bone marrow transplant, acute myeloid leukemia patients receiving induction or consolidation chemotherapy, patients undergoing autologous peripheral blood progenitor cell collection, and patients with severe chronic neutropenia.
The FDA’s approval of Nivestym was based on a review of evidence suggesting the drug is highly similar to Neupogen, according to Pfizer, the company developing Nivestym.
The full approved indication for Nivestym is as follows:
- To decrease the incidence of infection, as manifested by febrile neutropenia, in patients with nonmyeloid malignancies receiving myelosuppressive anticancer drugs associated with a significant incidence of severe neutropenia with fever
- To reduce the time to neutrophil recovery and the duration of fever following induction or consolidation chemotherapy in patients with acute myeloid leukemia
- To reduce the duration of neutropenia and neutropenia-related clinical sequelae (eg, febrile neutropenia) in patients with nonmyeloid malignancies undergoing myeloablative chemotherapy followed by bone marrow transplant
- For the mobilization of autologous hematopoietic progenitor cells into the peripheral blood for collection by leukapheresis
- For chronic administration to reduce the incidence and duration of sequelae of severe neutropenia (eg, fever, infections, oropharyngeal ulcers) in symptomatic patients with congenital neutropenia, cyclic neutropenia, or idiopathic neutropenia.
For more details on Nivestym, see the full prescribing information.
Diabetics have higher risk of hematologic, other cancers
A review of data from more than 19 million people indicates that diabetes significantly raises a person’s risk of developing cancer.
When researchers compared patients with diabetes and without, both male and female diabetics had an increased risk of leukemias and lymphomas as well as certain solid tumors.
Researchers also found that diabetes conferred a higher cancer risk for women than men, both for all cancers combined and for some specific cancers, including leukemia.
“The link between diabetes and the risk of developing cancer is now firmly established,” said Toshiaki Ohkuma, PhD, of The George Institute for Global Health at the University of New South Wales in Australia.
“We have also demonstrated, for the first time, that women with diabetes are more likely to develop any form of cancer and have a significantly higher chance of developing kidney, oral, and stomach cancers and leukemia.”
Dr Ohkuma and his colleagues reported these findings in Diabetologia.
The researchers conducted a systematic search in PubMed MEDLINE to identify reports on the links between diabetes and cancer. Additional reports were identified from the reference lists of the relevant studies.
Only those cohort studies providing relative risks (RRs) for the association between diabetes and cancer for both women and men were included. In total, 107 relevant articles were identified, along with 36 cohorts of individual participant data.
RRs for cancer were obtained for patients with diabetes (types 1 and 2 combined) versus those without diabetes, for both men and women. The women-to-men ratios of these relative risk ratios (RRRs) were then calculated to determine the excess risk in women if present.
Data on all-site cancer was available from 47 studies, involving 121 cohorts and 19,239,302 individuals.
Diabetics vs non-diabetics
Women with diabetes had a 27% higher risk of all-site cancer compared to women without diabetes (RR=1.27; 95% CI 1.21, 1.32; P<0.001).
For men, the risk of all-site cancer was 19% higher among those with diabetes than those without (RR=1.19; 95% CI 1.13, 1.25; P<0.001).
There were several hematologic malignancies for which diabetics had an increased risk, as shown in the following table.
| Cancer type | RR for women (99% CI) | RR for men (99% CI) |
| Lymphatic and hematopoietic tissue | 1.24 (1.05, 1.46)* | 1.21 (0.98, 1.48) |
| Leukemia | 1.53 (1.00, 2.33) | 1.22 (0.80, 1.85) |
| Myeloid leukemia | 0.83 (0.39, 1.76) | 1.12 (0.77, 1.62) |
| Acute myeloid leukemia | 1.33 (1.12, 1.57)* | 1.14 (0.56, 2.33) |
| Chronic myeloid leukemia | 1.67 (1.27, 2.20)* | 1.62 (1.32, 1.98)* |
| Lymphoid leukemia | 1.74 (0.31, 9.79) | 1.20 (0.86, 1.68) |
| Lymphoma | 2.31 (0.57, 9.30) | 1.80 (0.68, 4.75) |
| Non-Hodgkin lymphoma | 1.16 (1.02, 1.32)* | 1.20 (1.08, 1.34)* |
| Hodgkin lymphoma | 1.20 (0.61, 2.38) | 1.36 (1.05, 1.77)* |
| Multiple myeloma | 1.19 (0.97, 1.47) | 1.12 (0.90, 1.41) |
| *denotes statistical significance with a P value < 0.01 | ||
Sex comparison
Calculation of the women-to-men ratio revealed that women with diabetes had a 6% greater excess risk of all-site cancer compared to men with diabetes (RRR=1.06; 95% CI 1.03, 1.09; P<0.001).
The women-to-men ratios also showed significantly higher risks for female diabetics for:
- Kidney cancer—RRR=1.11 (99% CI 1.04, 1.18; P<0.001)
- Oral cancer—RRR=1.13 (99% CI 1.00, 1.28; P=0.009)
- Stomach cancer—RRR=1.14 (99% CI 1.07, 1.22; P<0.001)
- Leukemia—RRR=1.15 (99% CI 1.02, 1.28; P=0.002).
However, women had a significantly lower risk of liver cancer (RRR=0.88; 99% CI 0.79, 0.99; P=0.005).
There are several possible reasons for the excess cancer risk observed in women, according to study author Sanne Peters, PhD, of The George Institute for Global Health at the University of Oxford in the UK.
For example, on average, women are in the pre-diabetic state of impaired glucose tolerance 2 years longer than men.
“Historically, we know that women are often under-treated when they first present with symptoms of diabetes, are less likely to receive intensive care, and are not taking the same levels of medications as men,” Dr Peters said. “All of these could go some way into explaining why women are at greater risk of developing cancer, but, without more research, we can’t be certain.”
A review of data from more than 19 million people indicates that diabetes significantly raises a person’s risk of developing cancer.
When researchers compared patients with diabetes and without, both male and female diabetics had an increased risk of leukemias and lymphomas as well as certain solid tumors.
Researchers also found that diabetes conferred a higher cancer risk for women than men, both for all cancers combined and for some specific cancers, including leukemia.
“The link between diabetes and the risk of developing cancer is now firmly established,” said Toshiaki Ohkuma, PhD, of The George Institute for Global Health at the University of New South Wales in Australia.
“We have also demonstrated, for the first time, that women with diabetes are more likely to develop any form of cancer and have a significantly higher chance of developing kidney, oral, and stomach cancers and leukemia.”
Dr Ohkuma and his colleagues reported these findings in Diabetologia.
The researchers conducted a systematic search in PubMed MEDLINE to identify reports on the links between diabetes and cancer. Additional reports were identified from the reference lists of the relevant studies.
Only those cohort studies providing relative risks (RRs) for the association between diabetes and cancer for both women and men were included. In total, 107 relevant articles were identified, along with 36 cohorts of individual participant data.
RRs for cancer were obtained for patients with diabetes (types 1 and 2 combined) versus those without diabetes, for both men and women. The women-to-men ratios of these relative risk ratios (RRRs) were then calculated to determine the excess risk in women if present.
Data on all-site cancer was available from 47 studies, involving 121 cohorts and 19,239,302 individuals.
Diabetics vs non-diabetics
Women with diabetes had a 27% higher risk of all-site cancer compared to women without diabetes (RR=1.27; 95% CI 1.21, 1.32; P<0.001).
For men, the risk of all-site cancer was 19% higher among those with diabetes than those without (RR=1.19; 95% CI 1.13, 1.25; P<0.001).
There were several hematologic malignancies for which diabetics had an increased risk, as shown in the following table.
| Cancer type | RR for women (99% CI) | RR for men (99% CI) |
| Lymphatic and hematopoietic tissue | 1.24 (1.05, 1.46)* | 1.21 (0.98, 1.48) |
| Leukemia | 1.53 (1.00, 2.33) | 1.22 (0.80, 1.85) |
| Myeloid leukemia | 0.83 (0.39, 1.76) | 1.12 (0.77, 1.62) |
| Acute myeloid leukemia | 1.33 (1.12, 1.57)* | 1.14 (0.56, 2.33) |
| Chronic myeloid leukemia | 1.67 (1.27, 2.20)* | 1.62 (1.32, 1.98)* |
| Lymphoid leukemia | 1.74 (0.31, 9.79) | 1.20 (0.86, 1.68) |
| Lymphoma | 2.31 (0.57, 9.30) | 1.80 (0.68, 4.75) |
| Non-Hodgkin lymphoma | 1.16 (1.02, 1.32)* | 1.20 (1.08, 1.34)* |
| Hodgkin lymphoma | 1.20 (0.61, 2.38) | 1.36 (1.05, 1.77)* |
| Multiple myeloma | 1.19 (0.97, 1.47) | 1.12 (0.90, 1.41) |
| *denotes statistical significance with a P value < 0.01 | ||
Sex comparison
Calculation of the women-to-men ratio revealed that women with diabetes had a 6% greater excess risk of all-site cancer compared to men with diabetes (RRR=1.06; 95% CI 1.03, 1.09; P<0.001).
The women-to-men ratios also showed significantly higher risks for female diabetics for:
- Kidney cancer—RRR=1.11 (99% CI 1.04, 1.18; P<0.001)
- Oral cancer—RRR=1.13 (99% CI 1.00, 1.28; P=0.009)
- Stomach cancer—RRR=1.14 (99% CI 1.07, 1.22; P<0.001)
- Leukemia—RRR=1.15 (99% CI 1.02, 1.28; P=0.002).
However, women had a significantly lower risk of liver cancer (RRR=0.88; 99% CI 0.79, 0.99; P=0.005).
There are several possible reasons for the excess cancer risk observed in women, according to study author Sanne Peters, PhD, of The George Institute for Global Health at the University of Oxford in the UK.
For example, on average, women are in the pre-diabetic state of impaired glucose tolerance 2 years longer than men.
“Historically, we know that women are often under-treated when they first present with symptoms of diabetes, are less likely to receive intensive care, and are not taking the same levels of medications as men,” Dr Peters said. “All of these could go some way into explaining why women are at greater risk of developing cancer, but, without more research, we can’t be certain.”
A review of data from more than 19 million people indicates that diabetes significantly raises a person’s risk of developing cancer.
When researchers compared patients with diabetes and without, both male and female diabetics had an increased risk of leukemias and lymphomas as well as certain solid tumors.
Researchers also found that diabetes conferred a higher cancer risk for women than men, both for all cancers combined and for some specific cancers, including leukemia.
“The link between diabetes and the risk of developing cancer is now firmly established,” said Toshiaki Ohkuma, PhD, of The George Institute for Global Health at the University of New South Wales in Australia.
“We have also demonstrated, for the first time, that women with diabetes are more likely to develop any form of cancer and have a significantly higher chance of developing kidney, oral, and stomach cancers and leukemia.”
Dr Ohkuma and his colleagues reported these findings in Diabetologia.
The researchers conducted a systematic search in PubMed MEDLINE to identify reports on the links between diabetes and cancer. Additional reports were identified from the reference lists of the relevant studies.
Only those cohort studies providing relative risks (RRs) for the association between diabetes and cancer for both women and men were included. In total, 107 relevant articles were identified, along with 36 cohorts of individual participant data.
RRs for cancer were obtained for patients with diabetes (types 1 and 2 combined) versus those without diabetes, for both men and women. The women-to-men ratios of these relative risk ratios (RRRs) were then calculated to determine the excess risk in women if present.
Data on all-site cancer was available from 47 studies, involving 121 cohorts and 19,239,302 individuals.
Diabetics vs non-diabetics
Women with diabetes had a 27% higher risk of all-site cancer compared to women without diabetes (RR=1.27; 95% CI 1.21, 1.32; P<0.001).
For men, the risk of all-site cancer was 19% higher among those with diabetes than those without (RR=1.19; 95% CI 1.13, 1.25; P<0.001).
There were several hematologic malignancies for which diabetics had an increased risk, as shown in the following table.
| Cancer type | RR for women (99% CI) | RR for men (99% CI) |
| Lymphatic and hematopoietic tissue | 1.24 (1.05, 1.46)* | 1.21 (0.98, 1.48) |
| Leukemia | 1.53 (1.00, 2.33) | 1.22 (0.80, 1.85) |
| Myeloid leukemia | 0.83 (0.39, 1.76) | 1.12 (0.77, 1.62) |
| Acute myeloid leukemia | 1.33 (1.12, 1.57)* | 1.14 (0.56, 2.33) |
| Chronic myeloid leukemia | 1.67 (1.27, 2.20)* | 1.62 (1.32, 1.98)* |
| Lymphoid leukemia | 1.74 (0.31, 9.79) | 1.20 (0.86, 1.68) |
| Lymphoma | 2.31 (0.57, 9.30) | 1.80 (0.68, 4.75) |
| Non-Hodgkin lymphoma | 1.16 (1.02, 1.32)* | 1.20 (1.08, 1.34)* |
| Hodgkin lymphoma | 1.20 (0.61, 2.38) | 1.36 (1.05, 1.77)* |
| Multiple myeloma | 1.19 (0.97, 1.47) | 1.12 (0.90, 1.41) |
| *denotes statistical significance with a P value < 0.01 | ||
Sex comparison
Calculation of the women-to-men ratio revealed that women with diabetes had a 6% greater excess risk of all-site cancer compared to men with diabetes (RRR=1.06; 95% CI 1.03, 1.09; P<0.001).
The women-to-men ratios also showed significantly higher risks for female diabetics for:
- Kidney cancer—RRR=1.11 (99% CI 1.04, 1.18; P<0.001)
- Oral cancer—RRR=1.13 (99% CI 1.00, 1.28; P=0.009)
- Stomach cancer—RRR=1.14 (99% CI 1.07, 1.22; P<0.001)
- Leukemia—RRR=1.15 (99% CI 1.02, 1.28; P=0.002).
However, women had a significantly lower risk of liver cancer (RRR=0.88; 99% CI 0.79, 0.99; P=0.005).
There are several possible reasons for the excess cancer risk observed in women, according to study author Sanne Peters, PhD, of The George Institute for Global Health at the University of Oxford in the UK.
For example, on average, women are in the pre-diabetic state of impaired glucose tolerance 2 years longer than men.
“Historically, we know that women are often under-treated when they first present with symptoms of diabetes, are less likely to receive intensive care, and are not taking the same levels of medications as men,” Dr Peters said. “All of these could go some way into explaining why women are at greater risk of developing cancer, but, without more research, we can’t be certain.”
Adding elotuzumab improves myeloma PFS over pom/dex alone
STOCKHOLM –Adding the monoclonal antibody elotuzumab to pomalidomide and dexamethasone nearly doubled the overall response rate and median progression-free survival in patients with relapsed/refractory multiple myeloma compared with pomalidomide and dexamethasone alone, results of the phase 2 ELOQUENT-3 trial showed.
After a minimum follow-up of 9.1 months, median progression-free survival (PFS) for 60 patients assigned to receive elotuzumab (Empliciti), pomalidomide (Pomalyst), and dexamethasone (EPd) was 10.3 months, compared with 4.7 months for 60 patients assigned to pomalidomide and dexamethasone (Pd). This difference translated into a hazard ratio (HR) of 0.54 (P = .0078) favoring EPd, reported Meletios A. Dimopoulos, MD, of the National and Kapodistrian University of Athens (Greece).
“The study met its primary endpoint, which was specifically designed to detect a large treatment effect in a relatively small sample of patients. Elotuzumab with pomalidomide and dexamethasone showed a significant and clinically meaningful 46% reduction in the risk of progression or death,” he said at the annual congress of the European Hematology Association.
Elotuzumab is an immunoglobulin G (IgG) monoclonal antibody that targets signaling lymphocytic activation molecule F7 (SLAMF7) expressed on multiple myeloma cells. Pomalidomide, an immunomodulator, may act synergistically with elotuzumab through several different mechanisms to increase killing of multiple myeloma cells, Dr. Dimopoulos said.
In ELOQUENT-3, patients with relapsed or refractory multiple myeloma after 2 or more prior lines of therapy, including lenalidomide (Revlimid) and a proteasome inhibitor and no prior pomalidomide were enrolled and randomly assigned to receive either pomalidomide 4 mg orally on days 1-21 of each 28-day cycle plus oral dexamethasone 40 mg equivalent weekly, or to the same regimen plus intravenous elotuzumab 10 mg/kg weekly for cycles 1 and 2, and 20 mg/kg every 4 weeks for cycle 3 and subsequent cycles.
The trial met its primary endpoint of investigator-assessed PFS, with a 46% reduction in the risk of progression or death with EPd compared with Pd.
An analysis of PFS by subgroups showed that EPd was significantly superior to Pd for patients younger than 65 years, those with International Staging System stage I-II at study entry, patients with lactate dehydrogenase levels below 300 IU/L at baseline, patients who had two or three prior lines of therapy vs. four or more, and those who had disease that was refractory to both lenalidomide and a proteasome inhibitor.
EPd was also associated with a trend toward better PFS in an analysis combining patients with high-risk cytogenetics (deletion 17p or translocation 14;16) or high LDH levels, with a median of 7.7 months compared with 3.6 months for Pd. However, the HR, 0.55, was not statistically significant, likely because of the small sample size.
Similarly, the elotuzumab-containing combination showed a nonsignificant trend toward better PFS among patients without high risk disease, with a median PFS not reached, vs. not reached, vs. 4.7 months for patients treated with Pd.
The overall response rate with EPd was 53%, compared with 26% for Pd (odds ratio 3.5, P = .0029). The responses in the elotuzumab arm consisted of 8% complete response, 12% very good partial responses, and 33% partial responses. The respective rates in the Pd group were 2%, 7%, and 18%.
The median duration of response with EPd was not reached at the time of the database lock, compared with 8.3 months with Pd.
A preliminary analysis of overall survival showed a trend favoring EPd (13 deaths out to 22 months of follow-up, compared with 18 deaths out to 20 months in the Pd arm; HR 0.62, nonsignificant).
There were five treatment-related deaths in the EPd arm, and eight in the Pd arm. Grade 1 or 2 infusion reactions occurred in three patients in the EPd arm.
Other adverse events were comparable between the arms, with 57% of patients in the EPd arm and 60% in the Pd arm having at least one grade 3 or 4 adverse event.
“The hematologic toxicity was driven by pomalidomide and low-dose dexamethasone. For unclear reasons, there was less grade 3 or 4 neutropenia with the addition of elotuzumab to pomalidomide/dexamethasone, and also the infection rate was lower in the EPd arm,” Dr. Dimopoulos said.
SOURCE: Dimopoulos MA et al. EHA Congress, Abstract LB2606.
STOCKHOLM –Adding the monoclonal antibody elotuzumab to pomalidomide and dexamethasone nearly doubled the overall response rate and median progression-free survival in patients with relapsed/refractory multiple myeloma compared with pomalidomide and dexamethasone alone, results of the phase 2 ELOQUENT-3 trial showed.
After a minimum follow-up of 9.1 months, median progression-free survival (PFS) for 60 patients assigned to receive elotuzumab (Empliciti), pomalidomide (Pomalyst), and dexamethasone (EPd) was 10.3 months, compared with 4.7 months for 60 patients assigned to pomalidomide and dexamethasone (Pd). This difference translated into a hazard ratio (HR) of 0.54 (P = .0078) favoring EPd, reported Meletios A. Dimopoulos, MD, of the National and Kapodistrian University of Athens (Greece).
“The study met its primary endpoint, which was specifically designed to detect a large treatment effect in a relatively small sample of patients. Elotuzumab with pomalidomide and dexamethasone showed a significant and clinically meaningful 46% reduction in the risk of progression or death,” he said at the annual congress of the European Hematology Association.
Elotuzumab is an immunoglobulin G (IgG) monoclonal antibody that targets signaling lymphocytic activation molecule F7 (SLAMF7) expressed on multiple myeloma cells. Pomalidomide, an immunomodulator, may act synergistically with elotuzumab through several different mechanisms to increase killing of multiple myeloma cells, Dr. Dimopoulos said.
In ELOQUENT-3, patients with relapsed or refractory multiple myeloma after 2 or more prior lines of therapy, including lenalidomide (Revlimid) and a proteasome inhibitor and no prior pomalidomide were enrolled and randomly assigned to receive either pomalidomide 4 mg orally on days 1-21 of each 28-day cycle plus oral dexamethasone 40 mg equivalent weekly, or to the same regimen plus intravenous elotuzumab 10 mg/kg weekly for cycles 1 and 2, and 20 mg/kg every 4 weeks for cycle 3 and subsequent cycles.
The trial met its primary endpoint of investigator-assessed PFS, with a 46% reduction in the risk of progression or death with EPd compared with Pd.
An analysis of PFS by subgroups showed that EPd was significantly superior to Pd for patients younger than 65 years, those with International Staging System stage I-II at study entry, patients with lactate dehydrogenase levels below 300 IU/L at baseline, patients who had two or three prior lines of therapy vs. four or more, and those who had disease that was refractory to both lenalidomide and a proteasome inhibitor.
EPd was also associated with a trend toward better PFS in an analysis combining patients with high-risk cytogenetics (deletion 17p or translocation 14;16) or high LDH levels, with a median of 7.7 months compared with 3.6 months for Pd. However, the HR, 0.55, was not statistically significant, likely because of the small sample size.
Similarly, the elotuzumab-containing combination showed a nonsignificant trend toward better PFS among patients without high risk disease, with a median PFS not reached, vs. not reached, vs. 4.7 months for patients treated with Pd.
The overall response rate with EPd was 53%, compared with 26% for Pd (odds ratio 3.5, P = .0029). The responses in the elotuzumab arm consisted of 8% complete response, 12% very good partial responses, and 33% partial responses. The respective rates in the Pd group were 2%, 7%, and 18%.
The median duration of response with EPd was not reached at the time of the database lock, compared with 8.3 months with Pd.
A preliminary analysis of overall survival showed a trend favoring EPd (13 deaths out to 22 months of follow-up, compared with 18 deaths out to 20 months in the Pd arm; HR 0.62, nonsignificant).
There were five treatment-related deaths in the EPd arm, and eight in the Pd arm. Grade 1 or 2 infusion reactions occurred in three patients in the EPd arm.
Other adverse events were comparable between the arms, with 57% of patients in the EPd arm and 60% in the Pd arm having at least one grade 3 or 4 adverse event.
“The hematologic toxicity was driven by pomalidomide and low-dose dexamethasone. For unclear reasons, there was less grade 3 or 4 neutropenia with the addition of elotuzumab to pomalidomide/dexamethasone, and also the infection rate was lower in the EPd arm,” Dr. Dimopoulos said.
SOURCE: Dimopoulos MA et al. EHA Congress, Abstract LB2606.
STOCKHOLM –Adding the monoclonal antibody elotuzumab to pomalidomide and dexamethasone nearly doubled the overall response rate and median progression-free survival in patients with relapsed/refractory multiple myeloma compared with pomalidomide and dexamethasone alone, results of the phase 2 ELOQUENT-3 trial showed.
After a minimum follow-up of 9.1 months, median progression-free survival (PFS) for 60 patients assigned to receive elotuzumab (Empliciti), pomalidomide (Pomalyst), and dexamethasone (EPd) was 10.3 months, compared with 4.7 months for 60 patients assigned to pomalidomide and dexamethasone (Pd). This difference translated into a hazard ratio (HR) of 0.54 (P = .0078) favoring EPd, reported Meletios A. Dimopoulos, MD, of the National and Kapodistrian University of Athens (Greece).
“The study met its primary endpoint, which was specifically designed to detect a large treatment effect in a relatively small sample of patients. Elotuzumab with pomalidomide and dexamethasone showed a significant and clinically meaningful 46% reduction in the risk of progression or death,” he said at the annual congress of the European Hematology Association.
Elotuzumab is an immunoglobulin G (IgG) monoclonal antibody that targets signaling lymphocytic activation molecule F7 (SLAMF7) expressed on multiple myeloma cells. Pomalidomide, an immunomodulator, may act synergistically with elotuzumab through several different mechanisms to increase killing of multiple myeloma cells, Dr. Dimopoulos said.
In ELOQUENT-3, patients with relapsed or refractory multiple myeloma after 2 or more prior lines of therapy, including lenalidomide (Revlimid) and a proteasome inhibitor and no prior pomalidomide were enrolled and randomly assigned to receive either pomalidomide 4 mg orally on days 1-21 of each 28-day cycle plus oral dexamethasone 40 mg equivalent weekly, or to the same regimen plus intravenous elotuzumab 10 mg/kg weekly for cycles 1 and 2, and 20 mg/kg every 4 weeks for cycle 3 and subsequent cycles.
The trial met its primary endpoint of investigator-assessed PFS, with a 46% reduction in the risk of progression or death with EPd compared with Pd.
An analysis of PFS by subgroups showed that EPd was significantly superior to Pd for patients younger than 65 years, those with International Staging System stage I-II at study entry, patients with lactate dehydrogenase levels below 300 IU/L at baseline, patients who had two or three prior lines of therapy vs. four or more, and those who had disease that was refractory to both lenalidomide and a proteasome inhibitor.
EPd was also associated with a trend toward better PFS in an analysis combining patients with high-risk cytogenetics (deletion 17p or translocation 14;16) or high LDH levels, with a median of 7.7 months compared with 3.6 months for Pd. However, the HR, 0.55, was not statistically significant, likely because of the small sample size.
Similarly, the elotuzumab-containing combination showed a nonsignificant trend toward better PFS among patients without high risk disease, with a median PFS not reached, vs. not reached, vs. 4.7 months for patients treated with Pd.
The overall response rate with EPd was 53%, compared with 26% for Pd (odds ratio 3.5, P = .0029). The responses in the elotuzumab arm consisted of 8% complete response, 12% very good partial responses, and 33% partial responses. The respective rates in the Pd group were 2%, 7%, and 18%.
The median duration of response with EPd was not reached at the time of the database lock, compared with 8.3 months with Pd.
A preliminary analysis of overall survival showed a trend favoring EPd (13 deaths out to 22 months of follow-up, compared with 18 deaths out to 20 months in the Pd arm; HR 0.62, nonsignificant).
There were five treatment-related deaths in the EPd arm, and eight in the Pd arm. Grade 1 or 2 infusion reactions occurred in three patients in the EPd arm.
Other adverse events were comparable between the arms, with 57% of patients in the EPd arm and 60% in the Pd arm having at least one grade 3 or 4 adverse event.
“The hematologic toxicity was driven by pomalidomide and low-dose dexamethasone. For unclear reasons, there was less grade 3 or 4 neutropenia with the addition of elotuzumab to pomalidomide/dexamethasone, and also the infection rate was lower in the EPd arm,” Dr. Dimopoulos said.
SOURCE: Dimopoulos MA et al. EHA Congress, Abstract LB2606.
REPORTING FROM THE EHA CONGRESS
Key clinical point: Elotuzumab may have synergistic clinical activity with pomalidomide against multiple myeloma.
Major finding: Median PFS was 10.3 months with elotuzumab, pomalidomide, and dexamethasone vs. 4.7 months with pomalidomide and dexamethasone.
Study details: Randomized open-label phase 2 trial of 120 patients with multiple myeloma relapsed or refractory after 2 or more prior lines of therapy.
Disclosures: Bristol-Myers Squibb and AbbVie Biotherapeutics funded the study. Dr. Dimopoulos disclosed honoraria and/or consulting fees from Amgen, BMS, Celgene, Janssen and Takeda.
Source: Dimopoulos MA et al. EHA Congress, Abstract LB2606.
PET-guided treatment didn’t improve outcomes
In the PETAL trial, treatment intensification based on results of an interim positron emission tomography (PET) scan did not improve survival outcomes for patients with aggressive lymphomas.
PET-positive patients did not benefit by switching from R-CHOP to a more intensive chemotherapy regimen.
PET-negative patients did not benefit from 2 additional cycles of rituximab after R-CHOP.
These results were published in the Journal of Clinical Oncology.
PETAL was a randomized trial of patients with newly diagnosed T- or B-cell lymphomas.
Patients received 2 cycles of CHOP (cyclophosphamide, doxorubicin, vincristine, prednisone)—plus rituximab (R-CHOP) in CD20-positive lymphomas—followed by a PET scan.
PET-positive patients were randomized to receive 6 additional cycles of R-CHOP or 6 blocks of an intensive protocol used to treat Burkitt lymphoma. This protocol consisted of high-dose methotrexate, cytarabine, hyperfractionated cyclophosphamide and ifosfamide, split-dose doxorubicin and etoposide, vincristine, vindesine, and dexamethasone.
PET-negative patients with CD20-positive lymphomas were randomized to receive 4 additional cycles of R-CHOP or 4 additional cycles of R-CHOP followed by 2 more doses of rituximab.
Among patients with T-cell lymphomas, only PET-positive individuals underwent randomization. PET-negative patients received CHOP. Patients with CD20-positive T-cell lymphomas also received rituximab.
PET-positive results
Of the PET-positive patients (108/862), 52 were randomized to receive 6 additional cycles of R-CHOP, and 56 were randomized to 6 cycles of the Burkitt protocol.
In general, survival rates were similar regardless of treatment. The 2-year overall survival (OS) rate was 63.6% for patients who received R-CHOP and 55.4% for those who received the more intensive protocol.
Two-year progression-free survival (PFS) rates were 49.4% and 43.1%, respectively. Two-year event-free survival (EFS) rates were 42.0% and 31.6%, respectively.
Among patients with diffuse large B-cell lymphoma (DLBCL), the OS rate was 64.8% for patients who received R-CHOP and 47.1% for those on the Burkitt protocol. PFS rates were 55.5% and 41.4%, respectively.
There was a significant difference in EFS rates among the DLBCL patients—52.4% in the R-CHOP arm and 28.3% in the intensive arm (P=0.0186).
Among T-cell lymphoma patients, the OS rate was 22.2% in the R-CHOP arm and 30.0% in the intensive arm. The PFS rates were 12.7% and 30%, respectively. The EFS rates were the same as the PFS rates.
Overall, patients who received the Burkitt protocol had significantly higher rates of grade 3/4 hematologic toxicities, infection, and mucositis.
PET-negative results
Of 754 PET-negative patients, 697 had CD20-positive lymphomas, and 255 of those patients (all with B-cell lymphomas) underwent randomization.
There were 129 patients who were randomized to receive 6 cycles of R-CHOP (2 before and 4 after randomization) and 126 who were randomized to receive 6 cycles of R-CHOP plus 2 additional cycles of rituximab.
Again, survival rates were similar regardless of treatment.
The 2-year OS was 88.2% for patients who received only R-CHOP and 87.2% for those with additional rituximab exposure. PFS rates were 82.0% and 77.5%, respectively. EFS rates were 76.4% and 73.5%, respectively.
In the DLBCL patients, the OS rate was 88.5% in the R-CHOP arm and 85.8% in the intensive arm. PFS rates were 82.3% and 77.7%, respectively. EFS rates were 72.6% and 78.9%, respectively.
As increasing the dose of rituximab did not improve outcomes, the investigators concluded that 6 cycles of R-CHOP should be the standard of care for these patients.
The team also said interim PET scanning is “a powerful tool” for identifying chemotherapy-resistant lymphomas, and PET-positive patients may be candidates for immunologic treatment approaches.
In the PETAL trial, treatment intensification based on results of an interim positron emission tomography (PET) scan did not improve survival outcomes for patients with aggressive lymphomas.
PET-positive patients did not benefit by switching from R-CHOP to a more intensive chemotherapy regimen.
PET-negative patients did not benefit from 2 additional cycles of rituximab after R-CHOP.
These results were published in the Journal of Clinical Oncology.
PETAL was a randomized trial of patients with newly diagnosed T- or B-cell lymphomas.
Patients received 2 cycles of CHOP (cyclophosphamide, doxorubicin, vincristine, prednisone)—plus rituximab (R-CHOP) in CD20-positive lymphomas—followed by a PET scan.
PET-positive patients were randomized to receive 6 additional cycles of R-CHOP or 6 blocks of an intensive protocol used to treat Burkitt lymphoma. This protocol consisted of high-dose methotrexate, cytarabine, hyperfractionated cyclophosphamide and ifosfamide, split-dose doxorubicin and etoposide, vincristine, vindesine, and dexamethasone.
PET-negative patients with CD20-positive lymphomas were randomized to receive 4 additional cycles of R-CHOP or 4 additional cycles of R-CHOP followed by 2 more doses of rituximab.
Among patients with T-cell lymphomas, only PET-positive individuals underwent randomization. PET-negative patients received CHOP. Patients with CD20-positive T-cell lymphomas also received rituximab.
PET-positive results
Of the PET-positive patients (108/862), 52 were randomized to receive 6 additional cycles of R-CHOP, and 56 were randomized to 6 cycles of the Burkitt protocol.
In general, survival rates were similar regardless of treatment. The 2-year overall survival (OS) rate was 63.6% for patients who received R-CHOP and 55.4% for those who received the more intensive protocol.
Two-year progression-free survival (PFS) rates were 49.4% and 43.1%, respectively. Two-year event-free survival (EFS) rates were 42.0% and 31.6%, respectively.
Among patients with diffuse large B-cell lymphoma (DLBCL), the OS rate was 64.8% for patients who received R-CHOP and 47.1% for those on the Burkitt protocol. PFS rates were 55.5% and 41.4%, respectively.
There was a significant difference in EFS rates among the DLBCL patients—52.4% in the R-CHOP arm and 28.3% in the intensive arm (P=0.0186).
Among T-cell lymphoma patients, the OS rate was 22.2% in the R-CHOP arm and 30.0% in the intensive arm. The PFS rates were 12.7% and 30%, respectively. The EFS rates were the same as the PFS rates.
Overall, patients who received the Burkitt protocol had significantly higher rates of grade 3/4 hematologic toxicities, infection, and mucositis.
PET-negative results
Of 754 PET-negative patients, 697 had CD20-positive lymphomas, and 255 of those patients (all with B-cell lymphomas) underwent randomization.
There were 129 patients who were randomized to receive 6 cycles of R-CHOP (2 before and 4 after randomization) and 126 who were randomized to receive 6 cycles of R-CHOP plus 2 additional cycles of rituximab.
Again, survival rates were similar regardless of treatment.
The 2-year OS was 88.2% for patients who received only R-CHOP and 87.2% for those with additional rituximab exposure. PFS rates were 82.0% and 77.5%, respectively. EFS rates were 76.4% and 73.5%, respectively.
In the DLBCL patients, the OS rate was 88.5% in the R-CHOP arm and 85.8% in the intensive arm. PFS rates were 82.3% and 77.7%, respectively. EFS rates were 72.6% and 78.9%, respectively.
As increasing the dose of rituximab did not improve outcomes, the investigators concluded that 6 cycles of R-CHOP should be the standard of care for these patients.
The team also said interim PET scanning is “a powerful tool” for identifying chemotherapy-resistant lymphomas, and PET-positive patients may be candidates for immunologic treatment approaches.
In the PETAL trial, treatment intensification based on results of an interim positron emission tomography (PET) scan did not improve survival outcomes for patients with aggressive lymphomas.
PET-positive patients did not benefit by switching from R-CHOP to a more intensive chemotherapy regimen.
PET-negative patients did not benefit from 2 additional cycles of rituximab after R-CHOP.
These results were published in the Journal of Clinical Oncology.
PETAL was a randomized trial of patients with newly diagnosed T- or B-cell lymphomas.
Patients received 2 cycles of CHOP (cyclophosphamide, doxorubicin, vincristine, prednisone)—plus rituximab (R-CHOP) in CD20-positive lymphomas—followed by a PET scan.
PET-positive patients were randomized to receive 6 additional cycles of R-CHOP or 6 blocks of an intensive protocol used to treat Burkitt lymphoma. This protocol consisted of high-dose methotrexate, cytarabine, hyperfractionated cyclophosphamide and ifosfamide, split-dose doxorubicin and etoposide, vincristine, vindesine, and dexamethasone.
PET-negative patients with CD20-positive lymphomas were randomized to receive 4 additional cycles of R-CHOP or 4 additional cycles of R-CHOP followed by 2 more doses of rituximab.
Among patients with T-cell lymphomas, only PET-positive individuals underwent randomization. PET-negative patients received CHOP. Patients with CD20-positive T-cell lymphomas also received rituximab.
PET-positive results
Of the PET-positive patients (108/862), 52 were randomized to receive 6 additional cycles of R-CHOP, and 56 were randomized to 6 cycles of the Burkitt protocol.
In general, survival rates were similar regardless of treatment. The 2-year overall survival (OS) rate was 63.6% for patients who received R-CHOP and 55.4% for those who received the more intensive protocol.
Two-year progression-free survival (PFS) rates were 49.4% and 43.1%, respectively. Two-year event-free survival (EFS) rates were 42.0% and 31.6%, respectively.
Among patients with diffuse large B-cell lymphoma (DLBCL), the OS rate was 64.8% for patients who received R-CHOP and 47.1% for those on the Burkitt protocol. PFS rates were 55.5% and 41.4%, respectively.
There was a significant difference in EFS rates among the DLBCL patients—52.4% in the R-CHOP arm and 28.3% in the intensive arm (P=0.0186).
Among T-cell lymphoma patients, the OS rate was 22.2% in the R-CHOP arm and 30.0% in the intensive arm. The PFS rates were 12.7% and 30%, respectively. The EFS rates were the same as the PFS rates.
Overall, patients who received the Burkitt protocol had significantly higher rates of grade 3/4 hematologic toxicities, infection, and mucositis.
PET-negative results
Of 754 PET-negative patients, 697 had CD20-positive lymphomas, and 255 of those patients (all with B-cell lymphomas) underwent randomization.
There were 129 patients who were randomized to receive 6 cycles of R-CHOP (2 before and 4 after randomization) and 126 who were randomized to receive 6 cycles of R-CHOP plus 2 additional cycles of rituximab.
Again, survival rates were similar regardless of treatment.
The 2-year OS was 88.2% for patients who received only R-CHOP and 87.2% for those with additional rituximab exposure. PFS rates were 82.0% and 77.5%, respectively. EFS rates were 76.4% and 73.5%, respectively.
In the DLBCL patients, the OS rate was 88.5% in the R-CHOP arm and 85.8% in the intensive arm. PFS rates were 82.3% and 77.7%, respectively. EFS rates were 72.6% and 78.9%, respectively.
As increasing the dose of rituximab did not improve outcomes, the investigators concluded that 6 cycles of R-CHOP should be the standard of care for these patients.
The team also said interim PET scanning is “a powerful tool” for identifying chemotherapy-resistant lymphomas, and PET-positive patients may be candidates for immunologic treatment approaches.
Mutations linked to higher risk of SNs in CCSs
New research has shown that childhood cancer survivors (CCSs) with certain germline mutations have higher relative rates (RRs) of secondary neoplasms (SNs) later in life.
Mutation carriers had significantly higher rates of breast cancer and sarcoma if they had received radiation to treat their initial cancer.
Among CCSs who did not receive radiation, the mutations were associated with increased rates of any SN, breast cancer, nonmelanoma skin cancer, and 2 or more histologically distinct SNs.
These findings were reported in the Journal of Clinical Oncology.
Researchers sequenced samples from 3006 CCSs who were at least 5 years from their initial cancer diagnosis as well as 341 samples from cancer-free control subjects.
All subjects were participants in the St. Jude Lifetime Cohort Study, a retrospective study with prospective clinical follow-up.
Thirty-five percent of the CCSs had survived leukemia, and 19% had survived lymphoma.
The CCS’s median age at childhood cancer diagnosis was 7.1 years, and the median follow-up was 28 years. The controls had a median age of 36.4 at follow-up.
Results
There were 1120 SNs diagnosed in 439 CCSs (14.6%). Ninety-one CCSs developed 2 or more histologically distinct SNs. The median time to SN diagnosis was 25.6 years
Non-melanoma skin cancer (580 in 159 CCSs), meningiomas (233 in 102 CCSs), thyroid cancer (67 in 67 CCSs), and breast cancer (60 in 53 CCSs) were among the SNs reported.
There were 15 neoplasms recorded in the control group—14 cases of non-melanoma skin cancer and 1 meningioma.
Pathogenic or likely pathogenic (P/LP) mutations in 32 genes were reported in 175 CCSs. The prevalence in CCSs (5.8%) was nearly 10-fold higher than in controls (0.6%).
The most commonly mutated genes in CCSs were RB1 (n=43), NF1 (n=22), BRCA2 (n=14), BRCA1 (n=12), and TP53 (n=10).
In a multivariable analysis adjusted for sex, age at primary cancer diagnosis, and treatment, P/LP mutation carriers had a significantly higher rate of any SN (RR=1.8).
The rate of subsequent breast cancer was significantly increased among females with a P/LP mutation (RR= 9.4), recipients of chest radiation (RR=7.9), and those with higher anthracycline exposure (RR=2.4).
The rate of subsequent sarcoma was significantly increased for mutation carriers (RR=10.9) and CCSs with greater exposure to alkylating agents (RR=3.8).
Among irradiated CCSs, P/LP mutations were associated with significantly increased rates of breast cancer (RR=13.9) and sarcoma (RR=10.6)
Among non-irradiated CCSs, P/LP mutations were associated with significantly increased rates of any SN (RR=4.7), breast cancer (RR=7.7), nonmelanoma skin cancer (RR=11.0), and 2 or more histologically distinct SNs (RR=18.6).
The researchers said the higher risk of SNs in CCSs with P/LP mutations suggests all CCSs should be referred for genetic counseling.
New research has shown that childhood cancer survivors (CCSs) with certain germline mutations have higher relative rates (RRs) of secondary neoplasms (SNs) later in life.
Mutation carriers had significantly higher rates of breast cancer and sarcoma if they had received radiation to treat their initial cancer.
Among CCSs who did not receive radiation, the mutations were associated with increased rates of any SN, breast cancer, nonmelanoma skin cancer, and 2 or more histologically distinct SNs.
These findings were reported in the Journal of Clinical Oncology.
Researchers sequenced samples from 3006 CCSs who were at least 5 years from their initial cancer diagnosis as well as 341 samples from cancer-free control subjects.
All subjects were participants in the St. Jude Lifetime Cohort Study, a retrospective study with prospective clinical follow-up.
Thirty-five percent of the CCSs had survived leukemia, and 19% had survived lymphoma.
The CCS’s median age at childhood cancer diagnosis was 7.1 years, and the median follow-up was 28 years. The controls had a median age of 36.4 at follow-up.
Results
There were 1120 SNs diagnosed in 439 CCSs (14.6%). Ninety-one CCSs developed 2 or more histologically distinct SNs. The median time to SN diagnosis was 25.6 years
Non-melanoma skin cancer (580 in 159 CCSs), meningiomas (233 in 102 CCSs), thyroid cancer (67 in 67 CCSs), and breast cancer (60 in 53 CCSs) were among the SNs reported.
There were 15 neoplasms recorded in the control group—14 cases of non-melanoma skin cancer and 1 meningioma.
Pathogenic or likely pathogenic (P/LP) mutations in 32 genes were reported in 175 CCSs. The prevalence in CCSs (5.8%) was nearly 10-fold higher than in controls (0.6%).
The most commonly mutated genes in CCSs were RB1 (n=43), NF1 (n=22), BRCA2 (n=14), BRCA1 (n=12), and TP53 (n=10).
In a multivariable analysis adjusted for sex, age at primary cancer diagnosis, and treatment, P/LP mutation carriers had a significantly higher rate of any SN (RR=1.8).
The rate of subsequent breast cancer was significantly increased among females with a P/LP mutation (RR= 9.4), recipients of chest radiation (RR=7.9), and those with higher anthracycline exposure (RR=2.4).
The rate of subsequent sarcoma was significantly increased for mutation carriers (RR=10.9) and CCSs with greater exposure to alkylating agents (RR=3.8).
Among irradiated CCSs, P/LP mutations were associated with significantly increased rates of breast cancer (RR=13.9) and sarcoma (RR=10.6)
Among non-irradiated CCSs, P/LP mutations were associated with significantly increased rates of any SN (RR=4.7), breast cancer (RR=7.7), nonmelanoma skin cancer (RR=11.0), and 2 or more histologically distinct SNs (RR=18.6).
The researchers said the higher risk of SNs in CCSs with P/LP mutations suggests all CCSs should be referred for genetic counseling.
New research has shown that childhood cancer survivors (CCSs) with certain germline mutations have higher relative rates (RRs) of secondary neoplasms (SNs) later in life.
Mutation carriers had significantly higher rates of breast cancer and sarcoma if they had received radiation to treat their initial cancer.
Among CCSs who did not receive radiation, the mutations were associated with increased rates of any SN, breast cancer, nonmelanoma skin cancer, and 2 or more histologically distinct SNs.
These findings were reported in the Journal of Clinical Oncology.
Researchers sequenced samples from 3006 CCSs who were at least 5 years from their initial cancer diagnosis as well as 341 samples from cancer-free control subjects.
All subjects were participants in the St. Jude Lifetime Cohort Study, a retrospective study with prospective clinical follow-up.
Thirty-five percent of the CCSs had survived leukemia, and 19% had survived lymphoma.
The CCS’s median age at childhood cancer diagnosis was 7.1 years, and the median follow-up was 28 years. The controls had a median age of 36.4 at follow-up.
Results
There were 1120 SNs diagnosed in 439 CCSs (14.6%). Ninety-one CCSs developed 2 or more histologically distinct SNs. The median time to SN diagnosis was 25.6 years
Non-melanoma skin cancer (580 in 159 CCSs), meningiomas (233 in 102 CCSs), thyroid cancer (67 in 67 CCSs), and breast cancer (60 in 53 CCSs) were among the SNs reported.
There were 15 neoplasms recorded in the control group—14 cases of non-melanoma skin cancer and 1 meningioma.
Pathogenic or likely pathogenic (P/LP) mutations in 32 genes were reported in 175 CCSs. The prevalence in CCSs (5.8%) was nearly 10-fold higher than in controls (0.6%).
The most commonly mutated genes in CCSs were RB1 (n=43), NF1 (n=22), BRCA2 (n=14), BRCA1 (n=12), and TP53 (n=10).
In a multivariable analysis adjusted for sex, age at primary cancer diagnosis, and treatment, P/LP mutation carriers had a significantly higher rate of any SN (RR=1.8).
The rate of subsequent breast cancer was significantly increased among females with a P/LP mutation (RR= 9.4), recipients of chest radiation (RR=7.9), and those with higher anthracycline exposure (RR=2.4).
The rate of subsequent sarcoma was significantly increased for mutation carriers (RR=10.9) and CCSs with greater exposure to alkylating agents (RR=3.8).
Among irradiated CCSs, P/LP mutations were associated with significantly increased rates of breast cancer (RR=13.9) and sarcoma (RR=10.6)
Among non-irradiated CCSs, P/LP mutations were associated with significantly increased rates of any SN (RR=4.7), breast cancer (RR=7.7), nonmelanoma skin cancer (RR=11.0), and 2 or more histologically distinct SNs (RR=18.6).
The researchers said the higher risk of SNs in CCSs with P/LP mutations suggests all CCSs should be referred for genetic counseling.