User login
IRB members and industry relationships

Institutional Review Boards (IRBs) at academic health centers have gotten better at managing members’ industry relationships in the last several years, but issues persist, according to a new study.
The research is a follow-up to a study published in 2006.
The new study showed improvements in the management of industry relationships, with increased levels of disclosure and fewer “problematic relationships” than were reported in the previous study.
However, nearly a third of IRB members in the new study said they were unaware of their board’s conflict of interest policies. And a quarter of IRB members reported voting on protocols with which they have conflicts of interest, which is a violation of federal regulations.
“IRBs are the primary mechanisms by which institutions oversee research that involves human participants, and industry relationships of IRB members have the potential to impact their IRB-related activities in both positive and negative ways,” said Eric G. Campbell, PhD, of the Mongan Institute for Health Policy at Massachusetts General Hospital in Boston.
“While our principal findings are that, compared to a decade ago, IRBs appear to be doing a much better job at managing their members’ industry relationships, there still are findings that are concerning.”
Dr Campbell and his colleagues described these findings in JAMA Internal Medicine.
Every US institution that conducts research involving human participants must have an IRB, which reviews proposed studies to ensure their design is scientifically valid and does not violate ethical and regulatory guidelines. IRBs also monitor ongoing studies to make sure they maintain appropriate practices.
The first study to examine industry relationships of IRB members, conducted by Dr Campbell and his colleagues, was based on a survey of IRB members at 100 institutions that was published in NEJM in November 2006.
That study showed that more than a third of IRB members had some sort of financial relationship with commercial firms. And while most respondents did not believe that such relationships had an inappropriate impact on IRB decision making, significant percentages were not aware of institutional guidelines for disclosing industry relationships or defining conflicts of interest.
The current study involved an identical survey administered in 2014 to nearly 500 IRB members at 115 medical schools and teaching hospitals around the country.
As in the 2006 study, respondents were asked about specific types of relationships with commercial companies, whether they received any industry funding and for what purposes, and how many protocols had come before their IRB that involved companies with which they had a relationship.
For protocols with which IRB members had a conflict—relating either to companies they had relationships with or to competitors—they were asked whether they had disclosed their relationships to the IRB, whether they participated in discussion of those protocols, and whether they voted on the protocols.
Results
There was no significant difference between the current study and the previous study in the percentage of IRB members who reported having industry relationships overall—37% and 32%, respectively.
However, the percentage of respondents who reported receiving payments for attending meetings and conferences or for serving on speakers’ bureaus—relationships that are considered problematic—dropped significantly. The percentages fell from 16% to 9% for meeting/conference participation and from 14% to 4% for speakers’ bureaus.
“We were encouraged to see that the prevalence of potentially beneficial relationships—such as industry funding to support research studies—was essentially unchanged, indicating IRBs have not tried to eliminate members’ industry relationships across the board,” said Christine Vogeli, PhD, also of the Mongan Institute.
Survey respondents were more likely in the current study than in the previous one to say that their IRB had a written definition of conflicts of interest—63% compared with 46%. In addition, the percentage of respondents who reported their IRB had no policy related to conflicts dropped from 14% to 5%.
However, the researchers were concerned by the fact that 32% of respondents still did not know whether or not their IRB had a policy on conflicts, even though that number had dropped from 41% in the previous study.
The percentage of respondents who handled their conflicts of interest in an appropriate manner increased. Eighty percent of respondents with conflicts reported them to the IRB, up from 55% in the previous study.
And 68% of respondents said they always left the room when a protocol with which they had a conflict was being discussed, up from 38% in the previous study.
However, a quarter of respondents with conflicts indicated they had voted on protocols with which they had a conflict. While that was a drop from what was reported in the previous study, it was not statistically significant.
The percentage of respondents who felt that at least one protocol had been presented to their IRB in a biased fashion because of another member’s industry relationships dropped from 14% in the previous study to 8% in this study.
And when asked about the types of bias they perceived in the presentation—questions not included in the previous survey—8% of respondents reported a pro-industry bias, and 14% reported an anti-industry bias.
“The fact that we found any bias—either pro- or anti-industry—is an issue, since bias is antithetical to research and should be eliminated,” Dr Campbell said. “IRBs should address that issue, along with increasing efforts to educate their members about what constitutes a conflict of interest and the inappropriateness of voting on protocols with which they have a conflict.” ![]()

Institutional Review Boards (IRBs) at academic health centers have gotten better at managing members’ industry relationships in the last several years, but issues persist, according to a new study.
The research is a follow-up to a study published in 2006.
The new study showed improvements in the management of industry relationships, with increased levels of disclosure and fewer “problematic relationships” than were reported in the previous study.
However, nearly a third of IRB members in the new study said they were unaware of their board’s conflict of interest policies. And a quarter of IRB members reported voting on protocols with which they have conflicts of interest, which is a violation of federal regulations.
“IRBs are the primary mechanisms by which institutions oversee research that involves human participants, and industry relationships of IRB members have the potential to impact their IRB-related activities in both positive and negative ways,” said Eric G. Campbell, PhD, of the Mongan Institute for Health Policy at Massachusetts General Hospital in Boston.
“While our principal findings are that, compared to a decade ago, IRBs appear to be doing a much better job at managing their members’ industry relationships, there still are findings that are concerning.”
Dr Campbell and his colleagues described these findings in JAMA Internal Medicine.
Every US institution that conducts research involving human participants must have an IRB, which reviews proposed studies to ensure their design is scientifically valid and does not violate ethical and regulatory guidelines. IRBs also monitor ongoing studies to make sure they maintain appropriate practices.
The first study to examine industry relationships of IRB members, conducted by Dr Campbell and his colleagues, was based on a survey of IRB members at 100 institutions that was published in NEJM in November 2006.
That study showed that more than a third of IRB members had some sort of financial relationship with commercial firms. And while most respondents did not believe that such relationships had an inappropriate impact on IRB decision making, significant percentages were not aware of institutional guidelines for disclosing industry relationships or defining conflicts of interest.
The current study involved an identical survey administered in 2014 to nearly 500 IRB members at 115 medical schools and teaching hospitals around the country.
As in the 2006 study, respondents were asked about specific types of relationships with commercial companies, whether they received any industry funding and for what purposes, and how many protocols had come before their IRB that involved companies with which they had a relationship.
For protocols with which IRB members had a conflict—relating either to companies they had relationships with or to competitors—they were asked whether they had disclosed their relationships to the IRB, whether they participated in discussion of those protocols, and whether they voted on the protocols.
Results
There was no significant difference between the current study and the previous study in the percentage of IRB members who reported having industry relationships overall—37% and 32%, respectively.
However, the percentage of respondents who reported receiving payments for attending meetings and conferences or for serving on speakers’ bureaus—relationships that are considered problematic—dropped significantly. The percentages fell from 16% to 9% for meeting/conference participation and from 14% to 4% for speakers’ bureaus.
“We were encouraged to see that the prevalence of potentially beneficial relationships—such as industry funding to support research studies—was essentially unchanged, indicating IRBs have not tried to eliminate members’ industry relationships across the board,” said Christine Vogeli, PhD, also of the Mongan Institute.
Survey respondents were more likely in the current study than in the previous one to say that their IRB had a written definition of conflicts of interest—63% compared with 46%. In addition, the percentage of respondents who reported their IRB had no policy related to conflicts dropped from 14% to 5%.
However, the researchers were concerned by the fact that 32% of respondents still did not know whether or not their IRB had a policy on conflicts, even though that number had dropped from 41% in the previous study.
The percentage of respondents who handled their conflicts of interest in an appropriate manner increased. Eighty percent of respondents with conflicts reported them to the IRB, up from 55% in the previous study.
And 68% of respondents said they always left the room when a protocol with which they had a conflict was being discussed, up from 38% in the previous study.
However, a quarter of respondents with conflicts indicated they had voted on protocols with which they had a conflict. While that was a drop from what was reported in the previous study, it was not statistically significant.
The percentage of respondents who felt that at least one protocol had been presented to their IRB in a biased fashion because of another member’s industry relationships dropped from 14% in the previous study to 8% in this study.
And when asked about the types of bias they perceived in the presentation—questions not included in the previous survey—8% of respondents reported a pro-industry bias, and 14% reported an anti-industry bias.
“The fact that we found any bias—either pro- or anti-industry—is an issue, since bias is antithetical to research and should be eliminated,” Dr Campbell said. “IRBs should address that issue, along with increasing efforts to educate their members about what constitutes a conflict of interest and the inappropriateness of voting on protocols with which they have a conflict.” ![]()

Institutional Review Boards (IRBs) at academic health centers have gotten better at managing members’ industry relationships in the last several years, but issues persist, according to a new study.
The research is a follow-up to a study published in 2006.
The new study showed improvements in the management of industry relationships, with increased levels of disclosure and fewer “problematic relationships” than were reported in the previous study.
However, nearly a third of IRB members in the new study said they were unaware of their board’s conflict of interest policies. And a quarter of IRB members reported voting on protocols with which they have conflicts of interest, which is a violation of federal regulations.
“IRBs are the primary mechanisms by which institutions oversee research that involves human participants, and industry relationships of IRB members have the potential to impact their IRB-related activities in both positive and negative ways,” said Eric G. Campbell, PhD, of the Mongan Institute for Health Policy at Massachusetts General Hospital in Boston.
“While our principal findings are that, compared to a decade ago, IRBs appear to be doing a much better job at managing their members’ industry relationships, there still are findings that are concerning.”
Dr Campbell and his colleagues described these findings in JAMA Internal Medicine.
Every US institution that conducts research involving human participants must have an IRB, which reviews proposed studies to ensure their design is scientifically valid and does not violate ethical and regulatory guidelines. IRBs also monitor ongoing studies to make sure they maintain appropriate practices.
The first study to examine industry relationships of IRB members, conducted by Dr Campbell and his colleagues, was based on a survey of IRB members at 100 institutions that was published in NEJM in November 2006.
That study showed that more than a third of IRB members had some sort of financial relationship with commercial firms. And while most respondents did not believe that such relationships had an inappropriate impact on IRB decision making, significant percentages were not aware of institutional guidelines for disclosing industry relationships or defining conflicts of interest.
The current study involved an identical survey administered in 2014 to nearly 500 IRB members at 115 medical schools and teaching hospitals around the country.
As in the 2006 study, respondents were asked about specific types of relationships with commercial companies, whether they received any industry funding and for what purposes, and how many protocols had come before their IRB that involved companies with which they had a relationship.
For protocols with which IRB members had a conflict—relating either to companies they had relationships with or to competitors—they were asked whether they had disclosed their relationships to the IRB, whether they participated in discussion of those protocols, and whether they voted on the protocols.
Results
There was no significant difference between the current study and the previous study in the percentage of IRB members who reported having industry relationships overall—37% and 32%, respectively.
However, the percentage of respondents who reported receiving payments for attending meetings and conferences or for serving on speakers’ bureaus—relationships that are considered problematic—dropped significantly. The percentages fell from 16% to 9% for meeting/conference participation and from 14% to 4% for speakers’ bureaus.
“We were encouraged to see that the prevalence of potentially beneficial relationships—such as industry funding to support research studies—was essentially unchanged, indicating IRBs have not tried to eliminate members’ industry relationships across the board,” said Christine Vogeli, PhD, also of the Mongan Institute.
Survey respondents were more likely in the current study than in the previous one to say that their IRB had a written definition of conflicts of interest—63% compared with 46%. In addition, the percentage of respondents who reported their IRB had no policy related to conflicts dropped from 14% to 5%.
However, the researchers were concerned by the fact that 32% of respondents still did not know whether or not their IRB had a policy on conflicts, even though that number had dropped from 41% in the previous study.
The percentage of respondents who handled their conflicts of interest in an appropriate manner increased. Eighty percent of respondents with conflicts reported them to the IRB, up from 55% in the previous study.
And 68% of respondents said they always left the room when a protocol with which they had a conflict was being discussed, up from 38% in the previous study.
However, a quarter of respondents with conflicts indicated they had voted on protocols with which they had a conflict. While that was a drop from what was reported in the previous study, it was not statistically significant.
The percentage of respondents who felt that at least one protocol had been presented to their IRB in a biased fashion because of another member’s industry relationships dropped from 14% in the previous study to 8% in this study.
And when asked about the types of bias they perceived in the presentation—questions not included in the previous survey—8% of respondents reported a pro-industry bias, and 14% reported an anti-industry bias.
“The fact that we found any bias—either pro- or anti-industry—is an issue, since bias is antithetical to research and should be eliminated,” Dr Campbell said. “IRBs should address that issue, along with increasing efforts to educate their members about what constitutes a conflict of interest and the inappropriateness of voting on protocols with which they have a conflict.” ![]()
Length of cell-cycle phase affects HSC function

in the bone marrow
Shortening the G1 phase of the cell cycle can improve the production and function of hematopoietic stem cells (HSCs), according to research published in the Journal of Experimental Medicine.
When investigators shortened the G1 phase in human HSCs, they found the cells were better able to resist differentiation in vitro and exhibited enhanced engraftment in vivo.
However, these benefits only occurred when the team shortened the early phase of G1, not the late phase.
Claudia Waskow, PhD, of Technische Universitaet Dresden in Germany, and her colleagues conducted this research to determine whether the function of human HSCs is controlled by the kinetics of cell-cycle progression.
The investigators knew that the body’s pool of HSCs is maintained through self-renewing divisions tightly regulated by enzymatically active cyclin (CCN)/cyclin-dependent kinase (CDK) complexes.
So they enforced expression of functional CCND1–CDK4 complexes, which are important for progression through the early G1 phase of the cell cycle, and CCNE1–CDK2 complexes, which are key in the transition from the G1 phase to the S phase.
Overexpression of CCND1–CDK4 complexes (also referred to as elevated 4D) promoted the transit from G0 to G1 and successfully shortened the G1 phase. However, the total length of the cell cycle did not change much, as the G2 or M phase was prolonged slightly.
The investigators also found that elevated 4D levels protected HSCs from differentiation-inducing signals in vitro and provided a “competitive advantage” in vivo.
When they transplanted HSCs with elevated 4D into mice, the team observed improved donor-leukocyte engraftment but no increase in the HSC pool. They said the improvement in engraftment was based on an elevated output of myeloid cells.
In contrast to elevated 4D, overexpression of CCNE1–CDK2 (also referred to as elevated 2E) conferred detrimental effects. Elevated 2E did accelerate cell-cycle progression, but it led to the loss of functional HSCs and poor engraftment.
The investigators said a large proportion of cells with elevated 2E contained fragmented DNA and underwent apoptosis after transduction.
In addition, many HSCs with elevated 2E exited G0 and shifted to the S–G2–M phases of the cell cycle. The G1 phase was significantly shortened, and the time HSCs spent in each cycle was reduced.
Dr Waskow and her colleagues said these results suggest transit velocity through the early and late G1 phase is an important regulator of HSC function and therefore makes an essential contribution to the maintenance of hematopoiesis.
Furthermore, alterations of G1 transition kinetics may be the basis for functional defects observed in HSCs from old mice or elderly humans. ![]()

in the bone marrow
Shortening the G1 phase of the cell cycle can improve the production and function of hematopoietic stem cells (HSCs), according to research published in the Journal of Experimental Medicine.
When investigators shortened the G1 phase in human HSCs, they found the cells were better able to resist differentiation in vitro and exhibited enhanced engraftment in vivo.
However, these benefits only occurred when the team shortened the early phase of G1, not the late phase.
Claudia Waskow, PhD, of Technische Universitaet Dresden in Germany, and her colleagues conducted this research to determine whether the function of human HSCs is controlled by the kinetics of cell-cycle progression.
The investigators knew that the body’s pool of HSCs is maintained through self-renewing divisions tightly regulated by enzymatically active cyclin (CCN)/cyclin-dependent kinase (CDK) complexes.
So they enforced expression of functional CCND1–CDK4 complexes, which are important for progression through the early G1 phase of the cell cycle, and CCNE1–CDK2 complexes, which are key in the transition from the G1 phase to the S phase.
Overexpression of CCND1–CDK4 complexes (also referred to as elevated 4D) promoted the transit from G0 to G1 and successfully shortened the G1 phase. However, the total length of the cell cycle did not change much, as the G2 or M phase was prolonged slightly.
The investigators also found that elevated 4D levels protected HSCs from differentiation-inducing signals in vitro and provided a “competitive advantage” in vivo.
When they transplanted HSCs with elevated 4D into mice, the team observed improved donor-leukocyte engraftment but no increase in the HSC pool. They said the improvement in engraftment was based on an elevated output of myeloid cells.
In contrast to elevated 4D, overexpression of CCNE1–CDK2 (also referred to as elevated 2E) conferred detrimental effects. Elevated 2E did accelerate cell-cycle progression, but it led to the loss of functional HSCs and poor engraftment.
The investigators said a large proportion of cells with elevated 2E contained fragmented DNA and underwent apoptosis after transduction.
In addition, many HSCs with elevated 2E exited G0 and shifted to the S–G2–M phases of the cell cycle. The G1 phase was significantly shortened, and the time HSCs spent in each cycle was reduced.
Dr Waskow and her colleagues said these results suggest transit velocity through the early and late G1 phase is an important regulator of HSC function and therefore makes an essential contribution to the maintenance of hematopoiesis.
Furthermore, alterations of G1 transition kinetics may be the basis for functional defects observed in HSCs from old mice or elderly humans. ![]()

in the bone marrow
Shortening the G1 phase of the cell cycle can improve the production and function of hematopoietic stem cells (HSCs), according to research published in the Journal of Experimental Medicine.
When investigators shortened the G1 phase in human HSCs, they found the cells were better able to resist differentiation in vitro and exhibited enhanced engraftment in vivo.
However, these benefits only occurred when the team shortened the early phase of G1, not the late phase.
Claudia Waskow, PhD, of Technische Universitaet Dresden in Germany, and her colleagues conducted this research to determine whether the function of human HSCs is controlled by the kinetics of cell-cycle progression.
The investigators knew that the body’s pool of HSCs is maintained through self-renewing divisions tightly regulated by enzymatically active cyclin (CCN)/cyclin-dependent kinase (CDK) complexes.
So they enforced expression of functional CCND1–CDK4 complexes, which are important for progression through the early G1 phase of the cell cycle, and CCNE1–CDK2 complexes, which are key in the transition from the G1 phase to the S phase.
Overexpression of CCND1–CDK4 complexes (also referred to as elevated 4D) promoted the transit from G0 to G1 and successfully shortened the G1 phase. However, the total length of the cell cycle did not change much, as the G2 or M phase was prolonged slightly.
The investigators also found that elevated 4D levels protected HSCs from differentiation-inducing signals in vitro and provided a “competitive advantage” in vivo.
When they transplanted HSCs with elevated 4D into mice, the team observed improved donor-leukocyte engraftment but no increase in the HSC pool. They said the improvement in engraftment was based on an elevated output of myeloid cells.
In contrast to elevated 4D, overexpression of CCNE1–CDK2 (also referred to as elevated 2E) conferred detrimental effects. Elevated 2E did accelerate cell-cycle progression, but it led to the loss of functional HSCs and poor engraftment.
The investigators said a large proportion of cells with elevated 2E contained fragmented DNA and underwent apoptosis after transduction.
In addition, many HSCs with elevated 2E exited G0 and shifted to the S–G2–M phases of the cell cycle. The G1 phase was significantly shortened, and the time HSCs spent in each cycle was reduced.
Dr Waskow and her colleagues said these results suggest transit velocity through the early and late G1 phase is an important regulator of HSC function and therefore makes an essential contribution to the maintenance of hematopoiesis.
Furthermore, alterations of G1 transition kinetics may be the basis for functional defects observed in HSCs from old mice or elderly humans. ![]()
Gene regulates HSPC frequency, function

Photo by Aaron Logan
A genome-wide association study conducted in mice has provided new insights regarding hematopoietic stem/progenitor cells (HSPCs).
Researchers screened more than 100 mouse strains and found great variation in the frequency of 3 HSPC subpopulations.
The team also showed that Hopx, a gene that was not known to influence HSPC biology, regulates the frequency and function of HSPCs.
The researchers recounted these findings in Stem Cell Reports.
Hooman Allayee, PhD, of the University of Southern California in Los Angeles, and his colleagues screened 108 strains of mice known as the hybrid mouse diversity panel.
The screen revealed 3 HSPC subpopulations whose frequency varied greatly among the different mouse strains. The frequency of these HSPCs—Lin-Sca-1+c-Kit+ (LSK) cells, LSKCD150-CD48- cells, and LSKCD150+CD48- cells—varied roughly 120-fold to 300-fold.
The researchers then found that these 3 cell populations were significantly correlated with each other and with certain hematologic parameters. There was a significant positive association between LSK cells and total white blood cell (P=0.005), monocyte (P<0.0001), and lymphocyte counts (P=0.04).
LSKCD150-CD48- cells had a significant negative correlation with lymphocyte (P=0.006) and monocyte counts (P=0.002) as well as a significant positive association with granulocyte counts (P=0.0002).
LSKCD150+CD48- cells had a significant positive correlation with total white blood cell count (P=0.02) and a significant negative association with mean corpuscular hemoglobin (P<0.001).
Additional experiments showed that the frequency and function of LSKCD150-CD48- cells were regulated by Hopx, but the gene did not appear to impact LSK or LSKCD150+CD48- cells.
Mice lacking the Hopx gene had significantly lower numbers of LSKCD150-CD48- cells than wild-type mice, but LSK and LSKCD150+CD48- counts were similar between Hopx-/- and wild-type mice.
The researchers also conducted competitive repopulation assays with HSCs from Hopx-/- and wild-type mice. HSCs from Hopx-/- mice had significantly impaired engraftment at 16 weeks after transplant, which extended to 24 weeks.
Dr Allayee and his colleagues said that identifying this new role for Hopx could have clinical implications, and this research suggests the hybrid mouse diversity panel can be used to find genes that would otherwise go unnoticed.
“This powerful genetics platform has the potential to reveal the genes underlying other stem cell populations or a wide range of diseases that would be difficult to study in humans,” Dr Allayee said. ![]()

Photo by Aaron Logan
A genome-wide association study conducted in mice has provided new insights regarding hematopoietic stem/progenitor cells (HSPCs).
Researchers screened more than 100 mouse strains and found great variation in the frequency of 3 HSPC subpopulations.
The team also showed that Hopx, a gene that was not known to influence HSPC biology, regulates the frequency and function of HSPCs.
The researchers recounted these findings in Stem Cell Reports.
Hooman Allayee, PhD, of the University of Southern California in Los Angeles, and his colleagues screened 108 strains of mice known as the hybrid mouse diversity panel.
The screen revealed 3 HSPC subpopulations whose frequency varied greatly among the different mouse strains. The frequency of these HSPCs—Lin-Sca-1+c-Kit+ (LSK) cells, LSKCD150-CD48- cells, and LSKCD150+CD48- cells—varied roughly 120-fold to 300-fold.
The researchers then found that these 3 cell populations were significantly correlated with each other and with certain hematologic parameters. There was a significant positive association between LSK cells and total white blood cell (P=0.005), monocyte (P<0.0001), and lymphocyte counts (P=0.04).
LSKCD150-CD48- cells had a significant negative correlation with lymphocyte (P=0.006) and monocyte counts (P=0.002) as well as a significant positive association with granulocyte counts (P=0.0002).
LSKCD150+CD48- cells had a significant positive correlation with total white blood cell count (P=0.02) and a significant negative association with mean corpuscular hemoglobin (P<0.001).
Additional experiments showed that the frequency and function of LSKCD150-CD48- cells were regulated by Hopx, but the gene did not appear to impact LSK or LSKCD150+CD48- cells.
Mice lacking the Hopx gene had significantly lower numbers of LSKCD150-CD48- cells than wild-type mice, but LSK and LSKCD150+CD48- counts were similar between Hopx-/- and wild-type mice.
The researchers also conducted competitive repopulation assays with HSCs from Hopx-/- and wild-type mice. HSCs from Hopx-/- mice had significantly impaired engraftment at 16 weeks after transplant, which extended to 24 weeks.
Dr Allayee and his colleagues said that identifying this new role for Hopx could have clinical implications, and this research suggests the hybrid mouse diversity panel can be used to find genes that would otherwise go unnoticed.
“This powerful genetics platform has the potential to reveal the genes underlying other stem cell populations or a wide range of diseases that would be difficult to study in humans,” Dr Allayee said. ![]()

Photo by Aaron Logan
A genome-wide association study conducted in mice has provided new insights regarding hematopoietic stem/progenitor cells (HSPCs).
Researchers screened more than 100 mouse strains and found great variation in the frequency of 3 HSPC subpopulations.
The team also showed that Hopx, a gene that was not known to influence HSPC biology, regulates the frequency and function of HSPCs.
The researchers recounted these findings in Stem Cell Reports.
Hooman Allayee, PhD, of the University of Southern California in Los Angeles, and his colleagues screened 108 strains of mice known as the hybrid mouse diversity panel.
The screen revealed 3 HSPC subpopulations whose frequency varied greatly among the different mouse strains. The frequency of these HSPCs—Lin-Sca-1+c-Kit+ (LSK) cells, LSKCD150-CD48- cells, and LSKCD150+CD48- cells—varied roughly 120-fold to 300-fold.
The researchers then found that these 3 cell populations were significantly correlated with each other and with certain hematologic parameters. There was a significant positive association between LSK cells and total white blood cell (P=0.005), monocyte (P<0.0001), and lymphocyte counts (P=0.04).
LSKCD150-CD48- cells had a significant negative correlation with lymphocyte (P=0.006) and monocyte counts (P=0.002) as well as a significant positive association with granulocyte counts (P=0.0002).
LSKCD150+CD48- cells had a significant positive correlation with total white blood cell count (P=0.02) and a significant negative association with mean corpuscular hemoglobin (P<0.001).
Additional experiments showed that the frequency and function of LSKCD150-CD48- cells were regulated by Hopx, but the gene did not appear to impact LSK or LSKCD150+CD48- cells.
Mice lacking the Hopx gene had significantly lower numbers of LSKCD150-CD48- cells than wild-type mice, but LSK and LSKCD150+CD48- counts were similar between Hopx-/- and wild-type mice.
The researchers also conducted competitive repopulation assays with HSCs from Hopx-/- and wild-type mice. HSCs from Hopx-/- mice had significantly impaired engraftment at 16 weeks after transplant, which extended to 24 weeks.
Dr Allayee and his colleagues said that identifying this new role for Hopx could have clinical implications, and this research suggests the hybrid mouse diversity panel can be used to find genes that would otherwise go unnoticed.
“This powerful genetics platform has the potential to reveal the genes underlying other stem cell populations or a wide range of diseases that would be difficult to study in humans,” Dr Allayee said. ![]()
AYAs with cancer receive aggressive EOL care

patient and her father
Photo by Rhoda Baer
In a retrospective study, a majority of adolescents and young adults (AYAs) with terminal cancer received aggressive end-of-life (EOL) care.
Investigators looked at the use of intensive care, emergency room visits, chemotherapy use, and hospitalization among more than 600 AYAs with cancer who were treated at Kaiser Permanente in California.
Nearly 70% of patients made use of at least one of these measures in the last month of their life.
The investigators noted that their findings, which were published in JAMA Oncology, may not reflect care for the wider US population. But the study does suggest a need for more research into whether this
pattern reflects AYA cancer patients’ preferences for EOL care.
“A young person facing the end of life is a particularly difficult issue,” said study author Jennifer Mack, MD, MPH, of the Dana-Farber Cancer Institute in Boston, Massachusetts.
“While use of aggressive measures might be an informed decision by young people who would do anything they could to live longer, some interventions come with a cost, which is a poorer quality of life. This study raises questions about what kind of care they’re getting and how we can get them to the best quality of life at the end of their lives.”
The study included 633 patients, ages 15 to 39, who died of cancer between 2001 and 2010. The patients, who received care at Kaiser Permanente Southern California, had either been diagnosed with stage IV cancer or had a recurrence of stage I-III cancer. An initial review of a subset of 111 patients showed that death had been anticipated in 98% of cases.
The most common cancer diagnosis was gastrointestinal cancer (17%), while other common diagnoses were breast cancer (15%), genitourinary cancers (11%), leukemia (14%), and lymphoma (10%).
The investigators measured the use of 4 aggressive treatment measures—intensive care, emergency room visits, chemotherapy, and hospitalization—in patients’ last month of life.
Overall, 68% of patients (449/663) received at least one of these medically intensive EOL care measures. Eleven percent of patients (72/663) received chemotherapy, 22% (144/663) were admitted to the intensive care unit, 22% (147/663) had more than one emergency department visit, and 62% (413/663) were hospitalized.
Rates of hospitalization were higher among patients diagnosed with stage IV disease (66%) than among patients with stage I to III disease—66% and 58%, respectively (P=0.04).
The percentage of patients who received at least one medically intensive EOL care measure was higher in the stage IV cohort as well—71% and 63%, respectively (P=0.03). But there were no significant differences between the cohorts with regard to the other measures.
The investigators said these findings suggest the need to better understand EOL care preferences and decision-making in AYAs with cancer.
“We should think about talking with younger patients earlier about their prognoses, identifying their preferences, and working with them to deliver care that reflects those preferences,” Dr Mack said. “It may be that aggressive care is what they want, but they may end up on this pathway without thoughtful conversation and may be without recognition that they are dying.” ![]()

patient and her father
Photo by Rhoda Baer
In a retrospective study, a majority of adolescents and young adults (AYAs) with terminal cancer received aggressive end-of-life (EOL) care.
Investigators looked at the use of intensive care, emergency room visits, chemotherapy use, and hospitalization among more than 600 AYAs with cancer who were treated at Kaiser Permanente in California.
Nearly 70% of patients made use of at least one of these measures in the last month of their life.
The investigators noted that their findings, which were published in JAMA Oncology, may not reflect care for the wider US population. But the study does suggest a need for more research into whether this
pattern reflects AYA cancer patients’ preferences for EOL care.
“A young person facing the end of life is a particularly difficult issue,” said study author Jennifer Mack, MD, MPH, of the Dana-Farber Cancer Institute in Boston, Massachusetts.
“While use of aggressive measures might be an informed decision by young people who would do anything they could to live longer, some interventions come with a cost, which is a poorer quality of life. This study raises questions about what kind of care they’re getting and how we can get them to the best quality of life at the end of their lives.”
The study included 633 patients, ages 15 to 39, who died of cancer between 2001 and 2010. The patients, who received care at Kaiser Permanente Southern California, had either been diagnosed with stage IV cancer or had a recurrence of stage I-III cancer. An initial review of a subset of 111 patients showed that death had been anticipated in 98% of cases.
The most common cancer diagnosis was gastrointestinal cancer (17%), while other common diagnoses were breast cancer (15%), genitourinary cancers (11%), leukemia (14%), and lymphoma (10%).
The investigators measured the use of 4 aggressive treatment measures—intensive care, emergency room visits, chemotherapy, and hospitalization—in patients’ last month of life.
Overall, 68% of patients (449/663) received at least one of these medically intensive EOL care measures. Eleven percent of patients (72/663) received chemotherapy, 22% (144/663) were admitted to the intensive care unit, 22% (147/663) had more than one emergency department visit, and 62% (413/663) were hospitalized.
Rates of hospitalization were higher among patients diagnosed with stage IV disease (66%) than among patients with stage I to III disease—66% and 58%, respectively (P=0.04).
The percentage of patients who received at least one medically intensive EOL care measure was higher in the stage IV cohort as well—71% and 63%, respectively (P=0.03). But there were no significant differences between the cohorts with regard to the other measures.
The investigators said these findings suggest the need to better understand EOL care preferences and decision-making in AYAs with cancer.
“We should think about talking with younger patients earlier about their prognoses, identifying their preferences, and working with them to deliver care that reflects those preferences,” Dr Mack said. “It may be that aggressive care is what they want, but they may end up on this pathway without thoughtful conversation and may be without recognition that they are dying.” ![]()

patient and her father
Photo by Rhoda Baer
In a retrospective study, a majority of adolescents and young adults (AYAs) with terminal cancer received aggressive end-of-life (EOL) care.
Investigators looked at the use of intensive care, emergency room visits, chemotherapy use, and hospitalization among more than 600 AYAs with cancer who were treated at Kaiser Permanente in California.
Nearly 70% of patients made use of at least one of these measures in the last month of their life.
The investigators noted that their findings, which were published in JAMA Oncology, may not reflect care for the wider US population. But the study does suggest a need for more research into whether this
pattern reflects AYA cancer patients’ preferences for EOL care.
“A young person facing the end of life is a particularly difficult issue,” said study author Jennifer Mack, MD, MPH, of the Dana-Farber Cancer Institute in Boston, Massachusetts.
“While use of aggressive measures might be an informed decision by young people who would do anything they could to live longer, some interventions come with a cost, which is a poorer quality of life. This study raises questions about what kind of care they’re getting and how we can get them to the best quality of life at the end of their lives.”
The study included 633 patients, ages 15 to 39, who died of cancer between 2001 and 2010. The patients, who received care at Kaiser Permanente Southern California, had either been diagnosed with stage IV cancer or had a recurrence of stage I-III cancer. An initial review of a subset of 111 patients showed that death had been anticipated in 98% of cases.
The most common cancer diagnosis was gastrointestinal cancer (17%), while other common diagnoses were breast cancer (15%), genitourinary cancers (11%), leukemia (14%), and lymphoma (10%).
The investigators measured the use of 4 aggressive treatment measures—intensive care, emergency room visits, chemotherapy, and hospitalization—in patients’ last month of life.
Overall, 68% of patients (449/663) received at least one of these medically intensive EOL care measures. Eleven percent of patients (72/663) received chemotherapy, 22% (144/663) were admitted to the intensive care unit, 22% (147/663) had more than one emergency department visit, and 62% (413/663) were hospitalized.
Rates of hospitalization were higher among patients diagnosed with stage IV disease (66%) than among patients with stage I to III disease—66% and 58%, respectively (P=0.04).
The percentage of patients who received at least one medically intensive EOL care measure was higher in the stage IV cohort as well—71% and 63%, respectively (P=0.03). But there were no significant differences between the cohorts with regard to the other measures.
The investigators said these findings suggest the need to better understand EOL care preferences and decision-making in AYAs with cancer.
“We should think about talking with younger patients earlier about their prognoses, identifying their preferences, and working with them to deliver care that reflects those preferences,” Dr Mack said. “It may be that aggressive care is what they want, but they may end up on this pathway without thoughtful conversation and may be without recognition that they are dying.” ![]()
Model helps explain chaotic angiogenesis
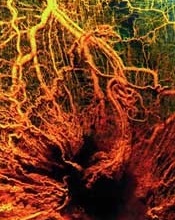
Image by Louis Heiser
and Robert Ackland
Scientists say they have developed a model that provides new insight into chaotic angiogenesis, a hallmark of cancer.
The model shows how proteins involved in angiogenesis communicate with each other and how tumors take charge of the protein signaling chain that controls blood vessel growth.
The work, published in PNAS, suggests that Jagged ligands play a major role in the chaotic vessel growth observed around tumors.
In normal growth, an endothelial cell sprouts from an existing blood vessel as a tip. Others that follow the tip cell become the stalk cells that ultimately form vessel walls. The Notch signaling pathway directs the endothelial cell’s decision to become a tip or stalk.
Notch receptors bind with Delta ligand or Jagged ligand molecules produced by cells. How they interact determines the cell’s fate.
When Notch and Delta bind, they prompt a few cells to be tips and adjacent ones to be stalks. The Notch-Delta pathway has been studied extensively and is the target of many angiogenesis inhibitors now in use, according to the scientists.
“We wondered exactly what Notch-Jagged signaling does that is not done in Notch-Delta signaling,” said study author Marcelo Boareto, PhD, of the Swiss Federal Institute of Technology in Zurich.
“We find that when the cells communicate mostly via Jagged, we see a new kind of cell that is not exactly tip and not exactly stalk, but somewhere in between. This compromised cell is the major difference between normal and tumor angiogenesis and suggests that if Notch-Jagged signaling can be somehow suppressed without affecting Notch-Delta, we can probably disrupt tumor angiogenesis.”
The scientists found the tip/stalk hybrid cells do form new blood vessels, but these vessels rarely mature.
“High levels of Jagged in the environment can trigger the formation of blood vessels that are useful to the tumor: fast-developing, leaky, and spread chaotically all over the tumor mass,” Dr Boareto said.
“Tumors don’t have to wait for the vessels to develop,” added study author José Onuchic, PhD, of Rice University in Houston, Texas. “They take advantage of the leakiness of the structure.”
The scientists’ model also takes into account the effect of vascular endothelial growth factor, which triggers angiogenesis and is overexpressed by tumor cells.
“It is very interesting how the tumor hijacks this important mechanism, which is needed for the development of a functional vessel, and amplifies it to generate pathological angiogenesis that leads to uncontrolled growth,” Dr Onuchic said. ![]()

Image by Louis Heiser
and Robert Ackland
Scientists say they have developed a model that provides new insight into chaotic angiogenesis, a hallmark of cancer.
The model shows how proteins involved in angiogenesis communicate with each other and how tumors take charge of the protein signaling chain that controls blood vessel growth.
The work, published in PNAS, suggests that Jagged ligands play a major role in the chaotic vessel growth observed around tumors.
In normal growth, an endothelial cell sprouts from an existing blood vessel as a tip. Others that follow the tip cell become the stalk cells that ultimately form vessel walls. The Notch signaling pathway directs the endothelial cell’s decision to become a tip or stalk.
Notch receptors bind with Delta ligand or Jagged ligand molecules produced by cells. How they interact determines the cell’s fate.
When Notch and Delta bind, they prompt a few cells to be tips and adjacent ones to be stalks. The Notch-Delta pathway has been studied extensively and is the target of many angiogenesis inhibitors now in use, according to the scientists.
“We wondered exactly what Notch-Jagged signaling does that is not done in Notch-Delta signaling,” said study author Marcelo Boareto, PhD, of the Swiss Federal Institute of Technology in Zurich.
“We find that when the cells communicate mostly via Jagged, we see a new kind of cell that is not exactly tip and not exactly stalk, but somewhere in between. This compromised cell is the major difference between normal and tumor angiogenesis and suggests that if Notch-Jagged signaling can be somehow suppressed without affecting Notch-Delta, we can probably disrupt tumor angiogenesis.”
The scientists found the tip/stalk hybrid cells do form new blood vessels, but these vessels rarely mature.
“High levels of Jagged in the environment can trigger the formation of blood vessels that are useful to the tumor: fast-developing, leaky, and spread chaotically all over the tumor mass,” Dr Boareto said.
“Tumors don’t have to wait for the vessels to develop,” added study author José Onuchic, PhD, of Rice University in Houston, Texas. “They take advantage of the leakiness of the structure.”
The scientists’ model also takes into account the effect of vascular endothelial growth factor, which triggers angiogenesis and is overexpressed by tumor cells.
“It is very interesting how the tumor hijacks this important mechanism, which is needed for the development of a functional vessel, and amplifies it to generate pathological angiogenesis that leads to uncontrolled growth,” Dr Onuchic said. ![]()

Image by Louis Heiser
and Robert Ackland
Scientists say they have developed a model that provides new insight into chaotic angiogenesis, a hallmark of cancer.
The model shows how proteins involved in angiogenesis communicate with each other and how tumors take charge of the protein signaling chain that controls blood vessel growth.
The work, published in PNAS, suggests that Jagged ligands play a major role in the chaotic vessel growth observed around tumors.
In normal growth, an endothelial cell sprouts from an existing blood vessel as a tip. Others that follow the tip cell become the stalk cells that ultimately form vessel walls. The Notch signaling pathway directs the endothelial cell’s decision to become a tip or stalk.
Notch receptors bind with Delta ligand or Jagged ligand molecules produced by cells. How they interact determines the cell’s fate.
When Notch and Delta bind, they prompt a few cells to be tips and adjacent ones to be stalks. The Notch-Delta pathway has been studied extensively and is the target of many angiogenesis inhibitors now in use, according to the scientists.
“We wondered exactly what Notch-Jagged signaling does that is not done in Notch-Delta signaling,” said study author Marcelo Boareto, PhD, of the Swiss Federal Institute of Technology in Zurich.
“We find that when the cells communicate mostly via Jagged, we see a new kind of cell that is not exactly tip and not exactly stalk, but somewhere in between. This compromised cell is the major difference between normal and tumor angiogenesis and suggests that if Notch-Jagged signaling can be somehow suppressed without affecting Notch-Delta, we can probably disrupt tumor angiogenesis.”
The scientists found the tip/stalk hybrid cells do form new blood vessels, but these vessels rarely mature.
“High levels of Jagged in the environment can trigger the formation of blood vessels that are useful to the tumor: fast-developing, leaky, and spread chaotically all over the tumor mass,” Dr Boareto said.
“Tumors don’t have to wait for the vessels to develop,” added study author José Onuchic, PhD, of Rice University in Houston, Texas. “They take advantage of the leakiness of the structure.”
The scientists’ model also takes into account the effect of vascular endothelial growth factor, which triggers angiogenesis and is overexpressed by tumor cells.
“It is very interesting how the tumor hijacks this important mechanism, which is needed for the development of a functional vessel, and amplifies it to generate pathological angiogenesis that leads to uncontrolled growth,” Dr Onuchic said. ![]()
Sepsis readmissions common and costly, group says

Photo courtesy of the CDC
Hospital readmissions for sepsis are under-recognized, and additional measures are needed to prevent these readmissions, researchers say.
The group conducted a study of California hospitals that showed sepsis accounts for roughly the same percentage of readmissions as heart attacks and congestive heart failure.
However, sepsis readmissions cost the healthcare system more than heart attack and congestive heart failure readmissions combined.
The research was published in Critical Care Medicine.
“Our study shows how common sepsis readmissions are and [elucidates] some of the factors that are associated with higher risk of readmission after these severe infections,” said study author Dong Chang, MD, of Harbor-UCLA Medical Center in Los Angeles, California.
“In addition, we show that sepsis readmissions have a significant impact on healthcare expenditures relative to other high-risk conditions that are receiving active attention and interventions. Based on these results, we believe that sepsis readmissions are under-recognized and should be among the conditions that are targeted for intervention by policymakers.”
The researchers analyzed data on sepsis admissions for adults 18 years of age and older at all California hospitals from 2009 through 2011. The team also analyzed data for congestive heart failure and heart attack admissions during the same period.
There were 240,198 admissions for sepsis, 193,153 for congestive heart failure, and 105,684 for heart attacks. The all-cause, 30-day readmission rate was 20.4% for sepsis, 23.6% for congestive heart failure, and 17.7% for heart attacks.
Patients with sepsis were readmitted because of respiratory failure; pneumonia; urinary tract infections; renal infections; renal failure; intestinal infections; complications with devices, implants, or grafts; and other causes.
Sepsis readmission rates were higher among young adults than older adults, among men than women, among black and Native American patients than other racial groups, and among lower-income patients than those with higher incomes. In addition, patients with other concurrent health problems were more likely to be readmitted than those with sepsis alone.
The estimated annual cost of sepsis-related readmissions in California during the study period was $500 million, compared with $229 million for congestive heart failure and $142 million for heart attacks.
“These findings suggest that efforts to reduce hospital readmissions need to include sepsis prominently, at least on par with heart failure and myocardial infarction,” said study author Martin Shapiro, MD, PhD, of the David Geffen School of Medicine at UCLA.
Next, the researchers plan to examine why patients are readmitted after sepsis and the percentage of those readmissions that are due to processes that can be improved upon, such as discharge practices, follow-up care, and teaching patients how to take their medications. ![]()

Photo courtesy of the CDC
Hospital readmissions for sepsis are under-recognized, and additional measures are needed to prevent these readmissions, researchers say.
The group conducted a study of California hospitals that showed sepsis accounts for roughly the same percentage of readmissions as heart attacks and congestive heart failure.
However, sepsis readmissions cost the healthcare system more than heart attack and congestive heart failure readmissions combined.
The research was published in Critical Care Medicine.
“Our study shows how common sepsis readmissions are and [elucidates] some of the factors that are associated with higher risk of readmission after these severe infections,” said study author Dong Chang, MD, of Harbor-UCLA Medical Center in Los Angeles, California.
“In addition, we show that sepsis readmissions have a significant impact on healthcare expenditures relative to other high-risk conditions that are receiving active attention and interventions. Based on these results, we believe that sepsis readmissions are under-recognized and should be among the conditions that are targeted for intervention by policymakers.”
The researchers analyzed data on sepsis admissions for adults 18 years of age and older at all California hospitals from 2009 through 2011. The team also analyzed data for congestive heart failure and heart attack admissions during the same period.
There were 240,198 admissions for sepsis, 193,153 for congestive heart failure, and 105,684 for heart attacks. The all-cause, 30-day readmission rate was 20.4% for sepsis, 23.6% for congestive heart failure, and 17.7% for heart attacks.
Patients with sepsis were readmitted because of respiratory failure; pneumonia; urinary tract infections; renal infections; renal failure; intestinal infections; complications with devices, implants, or grafts; and other causes.
Sepsis readmission rates were higher among young adults than older adults, among men than women, among black and Native American patients than other racial groups, and among lower-income patients than those with higher incomes. In addition, patients with other concurrent health problems were more likely to be readmitted than those with sepsis alone.
The estimated annual cost of sepsis-related readmissions in California during the study period was $500 million, compared with $229 million for congestive heart failure and $142 million for heart attacks.
“These findings suggest that efforts to reduce hospital readmissions need to include sepsis prominently, at least on par with heart failure and myocardial infarction,” said study author Martin Shapiro, MD, PhD, of the David Geffen School of Medicine at UCLA.
Next, the researchers plan to examine why patients are readmitted after sepsis and the percentage of those readmissions that are due to processes that can be improved upon, such as discharge practices, follow-up care, and teaching patients how to take their medications. ![]()

Photo courtesy of the CDC
Hospital readmissions for sepsis are under-recognized, and additional measures are needed to prevent these readmissions, researchers say.
The group conducted a study of California hospitals that showed sepsis accounts for roughly the same percentage of readmissions as heart attacks and congestive heart failure.
However, sepsis readmissions cost the healthcare system more than heart attack and congestive heart failure readmissions combined.
The research was published in Critical Care Medicine.
“Our study shows how common sepsis readmissions are and [elucidates] some of the factors that are associated with higher risk of readmission after these severe infections,” said study author Dong Chang, MD, of Harbor-UCLA Medical Center in Los Angeles, California.
“In addition, we show that sepsis readmissions have a significant impact on healthcare expenditures relative to other high-risk conditions that are receiving active attention and interventions. Based on these results, we believe that sepsis readmissions are under-recognized and should be among the conditions that are targeted for intervention by policymakers.”
The researchers analyzed data on sepsis admissions for adults 18 years of age and older at all California hospitals from 2009 through 2011. The team also analyzed data for congestive heart failure and heart attack admissions during the same period.
There were 240,198 admissions for sepsis, 193,153 for congestive heart failure, and 105,684 for heart attacks. The all-cause, 30-day readmission rate was 20.4% for sepsis, 23.6% for congestive heart failure, and 17.7% for heart attacks.
Patients with sepsis were readmitted because of respiratory failure; pneumonia; urinary tract infections; renal infections; renal failure; intestinal infections; complications with devices, implants, or grafts; and other causes.
Sepsis readmission rates were higher among young adults than older adults, among men than women, among black and Native American patients than other racial groups, and among lower-income patients than those with higher incomes. In addition, patients with other concurrent health problems were more likely to be readmitted than those with sepsis alone.
The estimated annual cost of sepsis-related readmissions in California during the study period was $500 million, compared with $229 million for congestive heart failure and $142 million for heart attacks.
“These findings suggest that efforts to reduce hospital readmissions need to include sepsis prominently, at least on par with heart failure and myocardial infarction,” said study author Martin Shapiro, MD, PhD, of the David Geffen School of Medicine at UCLA.
Next, the researchers plan to examine why patients are readmitted after sepsis and the percentage of those readmissions that are due to processes that can be improved upon, such as discharge practices, follow-up care, and teaching patients how to take their medications. ![]()
Drug may fight cancer by suppressing inflammation

Intermittent dosing with the immunosuppressant rapamycin can break an inflammatory loop that fuels cancer relapse, according to researchers.
The team found that rapamycin selectively breaks the cascade of inflammatory events that follow cellular senescence.
Once disrupted, it takes time for the inflammatory loop to re-establish itself. This suggests intermittent dosing could provide a way to reap the benefits of rapamycin while reducing side effects.
The researchers described these findings in Nature Cell Biology.
“We think this could provide a paradigm shift in the treatment of age-related disease, including cancer,” said study author Judith Campisi, PhD, of the Buck Institute for Research on Aging in Novato, California.
“Imagine the possibility of taking a pill for a few days or weeks every few years, as opposed to taking something with side effects every day for the rest of your life. It’s a new way of looking at how we could deal with age-related maladies.”
For this study, Dr Campisi and her colleagues administered rapamycin to mice with prostate cancer after they had received treatment with DNA-damaging chemotherapy.
The rapamycin reduced the secretion of inflammatory cytokines from senescent cells by suppressing the mTOR pathway. This suppressed the cells’ ability to stimulate tumor growth.
“DNA-damaging chemotherapy causes senescence, both to the tumor and its microenvironment,” Dr Campisi explained. “The tumor shrinks, but the immediate tissue environment is inflamed. We think signals from those inflamed cells trigger residual cancer cells to grow again. In the mice, rapamycin suppressed the ability of the tumor cells to relapse.”
The researchers said the paradigm-shifting potential of intermittent rapamycin dosing is based on the fact that it takes time for the inflammatory loop—fueled by the senescence-associated secretory phenotype (SASP)—to form and time for it to re-establish itself after a brief treatment period with rapamycin.
“Rapamycin blocks the production of a protein called IL-1α,” explained study author Remi-Martin Laberge, PhD, also of the Buck Institute.
“This in turn, suppresses IL6, a well-known inflammatory cytokine, at the level of transcription, which prevents the production of the IL6 protein. Because it acts at a deeper level within the cellular process, it takes longer for it to get started again.”
Dr Laberge also pointed out that treatment with rapamycin selectivity impacts the SASP, preserving the function of factors essential for wound healing.
“It’s an elegant solution,” Dr Laberge said. “Imagine using a small hammer to delicately knock out one thing that is causing problems. We knocked it out, and it stayed out long enough to benefit the health of the animal.”
“We have yet to fully understand why suppressing the mTOR pathway via rapamycin increases lifespan and health span in mice,” Dr Campisi added. “This work helps illuminate the puzzle. Perhaps the mice are living longer because they have less overall inflammation, and maybe intermittent dosing will make it possible for us to use [rapamycin] more widely in humans.” ![]()

Intermittent dosing with the immunosuppressant rapamycin can break an inflammatory loop that fuels cancer relapse, according to researchers.
The team found that rapamycin selectively breaks the cascade of inflammatory events that follow cellular senescence.
Once disrupted, it takes time for the inflammatory loop to re-establish itself. This suggests intermittent dosing could provide a way to reap the benefits of rapamycin while reducing side effects.
The researchers described these findings in Nature Cell Biology.
“We think this could provide a paradigm shift in the treatment of age-related disease, including cancer,” said study author Judith Campisi, PhD, of the Buck Institute for Research on Aging in Novato, California.
“Imagine the possibility of taking a pill for a few days or weeks every few years, as opposed to taking something with side effects every day for the rest of your life. It’s a new way of looking at how we could deal with age-related maladies.”
For this study, Dr Campisi and her colleagues administered rapamycin to mice with prostate cancer after they had received treatment with DNA-damaging chemotherapy.
The rapamycin reduced the secretion of inflammatory cytokines from senescent cells by suppressing the mTOR pathway. This suppressed the cells’ ability to stimulate tumor growth.
“DNA-damaging chemotherapy causes senescence, both to the tumor and its microenvironment,” Dr Campisi explained. “The tumor shrinks, but the immediate tissue environment is inflamed. We think signals from those inflamed cells trigger residual cancer cells to grow again. In the mice, rapamycin suppressed the ability of the tumor cells to relapse.”
The researchers said the paradigm-shifting potential of intermittent rapamycin dosing is based on the fact that it takes time for the inflammatory loop—fueled by the senescence-associated secretory phenotype (SASP)—to form and time for it to re-establish itself after a brief treatment period with rapamycin.
“Rapamycin blocks the production of a protein called IL-1α,” explained study author Remi-Martin Laberge, PhD, also of the Buck Institute.
“This in turn, suppresses IL6, a well-known inflammatory cytokine, at the level of transcription, which prevents the production of the IL6 protein. Because it acts at a deeper level within the cellular process, it takes longer for it to get started again.”
Dr Laberge also pointed out that treatment with rapamycin selectivity impacts the SASP, preserving the function of factors essential for wound healing.
“It’s an elegant solution,” Dr Laberge said. “Imagine using a small hammer to delicately knock out one thing that is causing problems. We knocked it out, and it stayed out long enough to benefit the health of the animal.”
“We have yet to fully understand why suppressing the mTOR pathway via rapamycin increases lifespan and health span in mice,” Dr Campisi added. “This work helps illuminate the puzzle. Perhaps the mice are living longer because they have less overall inflammation, and maybe intermittent dosing will make it possible for us to use [rapamycin] more widely in humans.” ![]()

Intermittent dosing with the immunosuppressant rapamycin can break an inflammatory loop that fuels cancer relapse, according to researchers.
The team found that rapamycin selectively breaks the cascade of inflammatory events that follow cellular senescence.
Once disrupted, it takes time for the inflammatory loop to re-establish itself. This suggests intermittent dosing could provide a way to reap the benefits of rapamycin while reducing side effects.
The researchers described these findings in Nature Cell Biology.
“We think this could provide a paradigm shift in the treatment of age-related disease, including cancer,” said study author Judith Campisi, PhD, of the Buck Institute for Research on Aging in Novato, California.
“Imagine the possibility of taking a pill for a few days or weeks every few years, as opposed to taking something with side effects every day for the rest of your life. It’s a new way of looking at how we could deal with age-related maladies.”
For this study, Dr Campisi and her colleagues administered rapamycin to mice with prostate cancer after they had received treatment with DNA-damaging chemotherapy.
The rapamycin reduced the secretion of inflammatory cytokines from senescent cells by suppressing the mTOR pathway. This suppressed the cells’ ability to stimulate tumor growth.
“DNA-damaging chemotherapy causes senescence, both to the tumor and its microenvironment,” Dr Campisi explained. “The tumor shrinks, but the immediate tissue environment is inflamed. We think signals from those inflamed cells trigger residual cancer cells to grow again. In the mice, rapamycin suppressed the ability of the tumor cells to relapse.”
The researchers said the paradigm-shifting potential of intermittent rapamycin dosing is based on the fact that it takes time for the inflammatory loop—fueled by the senescence-associated secretory phenotype (SASP)—to form and time for it to re-establish itself after a brief treatment period with rapamycin.
“Rapamycin blocks the production of a protein called IL-1α,” explained study author Remi-Martin Laberge, PhD, also of the Buck Institute.
“This in turn, suppresses IL6, a well-known inflammatory cytokine, at the level of transcription, which prevents the production of the IL6 protein. Because it acts at a deeper level within the cellular process, it takes longer for it to get started again.”
Dr Laberge also pointed out that treatment with rapamycin selectivity impacts the SASP, preserving the function of factors essential for wound healing.
“It’s an elegant solution,” Dr Laberge said. “Imagine using a small hammer to delicately knock out one thing that is causing problems. We knocked it out, and it stayed out long enough to benefit the health of the animal.”
“We have yet to fully understand why suppressing the mTOR pathway via rapamycin increases lifespan and health span in mice,” Dr Campisi added. “This work helps illuminate the puzzle. Perhaps the mice are living longer because they have less overall inflammation, and maybe intermittent dosing will make it possible for us to use [rapamycin] more widely in humans.”
Healthcare professionals work while sick despite risk to patients

Photo by Logan Tuttle
Results of a small survey showed that many healthcare professionals reported to work while sick, despite recognizing that this could put their patients at risk.
About 95% of survey respondents acknowledged that working while sick puts patients at risk, but 83% of respondents said they had worked while sick at least once in the past year.
About 9% of respondents reported working while sick at least 5 times.
Julia E. Szymczak, PhD, of the Children’s Hospital of Philadelphia in Pennsylvania, and her colleagues reported these results in JAMA Pediatrics alongside a related editorial.
The researchers administered an anonymous survey to attending physicians and advanced practice clinicians (APCs) at the Children’s Hospital of Philadelphia. APCs included certified registered nurse practitioners, physician assistants, clinical nurse specialists, certified registered nurse anesthetists, and certified nurse midwives.
Overall, 280 attending physicians (61%) and 256 APCs (54.5%) responded. Most respondents (504, 95.3%) said working while sick put patients at risk.
However, 446 respondents (83.1%) reported working while sick at least once in the past year, and 50 respondents (9.3%) reported working while sick at least 5 times.
Respondents reported working with symptoms that included diarrhea, fever, and the onset of significant respiratory symptoms.
The reasons physicians and APCs worked while sick included not wanting to let colleagues down (98.7%), staffing concerns (94.9%), not wanting to let patients down (92.5%), fear of being ostracized by colleagues (64%), and concerns about the continuity of care (63.8%).
An analysis of written comments about why respondents work while sick highlighted 3 areas: challenges in identifying and arranging someone to cover their work and a lack of resources to accommodate sick leave, a strong cultural norm in the hospital to report for work unless one is extremely ill, and ambiguity about what symptoms constitute being too sick to work.
Dr Szymczak and her colleagues said this study suggests complex social and logistical factors cause healthcare workers to report to work sick. But these results could inform efforts to help workers make the right choice to keep their patients and colleagues safe while caring for themselves.

Photo by Logan Tuttle
Results of a small survey showed that many healthcare professionals reported to work while sick, despite recognizing that this could put their patients at risk.
About 95% of survey respondents acknowledged that working while sick puts patients at risk, but 83% of respondents said they had worked while sick at least once in the past year.
About 9% of respondents reported working while sick at least 5 times.
Julia E. Szymczak, PhD, of the Children’s Hospital of Philadelphia in Pennsylvania, and her colleagues reported these results in JAMA Pediatrics alongside a related editorial.
The researchers administered an anonymous survey to attending physicians and advanced practice clinicians (APCs) at the Children’s Hospital of Philadelphia. APCs included certified registered nurse practitioners, physician assistants, clinical nurse specialists, certified registered nurse anesthetists, and certified nurse midwives.
Overall, 280 attending physicians (61%) and 256 APCs (54.5%) responded. Most respondents (504, 95.3%) said working while sick put patients at risk.
However, 446 respondents (83.1%) reported working while sick at least once in the past year, and 50 respondents (9.3%) reported working while sick at least 5 times.
Respondents reported working with symptoms that included diarrhea, fever, and the onset of significant respiratory symptoms.
The reasons physicians and APCs worked while sick included not wanting to let colleagues down (98.7%), staffing concerns (94.9%), not wanting to let patients down (92.5%), fear of being ostracized by colleagues (64%), and concerns about the continuity of care (63.8%).
An analysis of written comments about why respondents work while sick highlighted 3 areas: challenges in identifying and arranging someone to cover their work and a lack of resources to accommodate sick leave, a strong cultural norm in the hospital to report for work unless one is extremely ill, and ambiguity about what symptoms constitute being too sick to work.
Dr Szymczak and her colleagues said this study suggests complex social and logistical factors cause healthcare workers to report to work sick. But these results could inform efforts to help workers make the right choice to keep their patients and colleagues safe while caring for themselves.

Photo by Logan Tuttle
Results of a small survey showed that many healthcare professionals reported to work while sick, despite recognizing that this could put their patients at risk.
About 95% of survey respondents acknowledged that working while sick puts patients at risk, but 83% of respondents said they had worked while sick at least once in the past year.
About 9% of respondents reported working while sick at least 5 times.
Julia E. Szymczak, PhD, of the Children’s Hospital of Philadelphia in Pennsylvania, and her colleagues reported these results in JAMA Pediatrics alongside a related editorial.
The researchers administered an anonymous survey to attending physicians and advanced practice clinicians (APCs) at the Children’s Hospital of Philadelphia. APCs included certified registered nurse practitioners, physician assistants, clinical nurse specialists, certified registered nurse anesthetists, and certified nurse midwives.
Overall, 280 attending physicians (61%) and 256 APCs (54.5%) responded. Most respondents (504, 95.3%) said working while sick put patients at risk.
However, 446 respondents (83.1%) reported working while sick at least once in the past year, and 50 respondents (9.3%) reported working while sick at least 5 times.
Respondents reported working with symptoms that included diarrhea, fever, and the onset of significant respiratory symptoms.
The reasons physicians and APCs worked while sick included not wanting to let colleagues down (98.7%), staffing concerns (94.9%), not wanting to let patients down (92.5%), fear of being ostracized by colleagues (64%), and concerns about the continuity of care (63.8%).
An analysis of written comments about why respondents work while sick highlighted 3 areas: challenges in identifying and arranging someone to cover their work and a lack of resources to accommodate sick leave, a strong cultural norm in the hospital to report for work unless one is extremely ill, and ambiguity about what symptoms constitute being too sick to work.
Dr Szymczak and her colleagues said this study suggests complex social and logistical factors cause healthcare workers to report to work sick. But these results could inform efforts to help workers make the right choice to keep their patients and colleagues safe while caring for themselves.
PI3K inhibitors may promote cancer spread

Photo courtesy of
The Wistar Institute
Although PI3K inhibitors have been designed to treat cancer, new research indicates these drugs may actually exacerbate the disease.
Researchers found evidence to suggest that treatment with PI3K inhibitors alone can promote more aggressive tumor cell behavior and increase the likelihood that cancer will spread.
PI3K inhibitors appeared to reprogram the mitochondria of tumor cells and move them to “strategic” positions for invasion.
However, the researchers believe that targeting mitochondrial function along with PI3K could prevent this effect.
Dario C. Altieri, MD, of The Wistar Institute in Philadelphia, Pennsylvania, and his colleagues described these findings in PNAS.
The researchers decided to investigate how mitochondria are reprogrammed when exposed to PI3K inhibition and how mitochondria might prevent targeted agents from being as effective as expected.
“Our prior studies have confirmed that tumor cells rely on energy produced by mitochondria more significantly than previously thought,” Dr Altieri said.
“What we have shown in this study is that, in somewhat of a paradox, treatment with a PI3K inhibitor causes a tumor cell’s mitochondria to produce energy in a localized manner, promoting a far more aggressive and invasive phenotype. The treatment appears to be doing the opposite of its intended effect.”
The study showed that treatment with a PI3K inhibitor causes the mitochondria to migrate to the peripheral cytoskeleton of the tumor cells.
While the mitochondria in untreated cells cluster around the cell’s nucleus, exposure of tumor cells to PI3K therapy causes the mitochondria to move to specialized regions of the cell’s membrane implicated in cell motility and invasion.
In this “strategic” position, tumor mitochondria are ideally positioned to provide a concentrated source of energy to support an increase in cell migration and invasion.
However, the researchers said the dependence of this response on mitochondrial function may offer a new therapeutic angle.
Dr Altieri and his team have shown that targeting mitochondrial functions for tumor therapy is feasible and dramatically enhances the anticancer activity of PI3K inhibitors when used in combination.
“These findings continue to support the idea that the mitochondria of tumor cells are crucial to tumor survival and proliferation,” Dr Altieri said. “It’s certainly counterintuitive that a drug designed to fight cancer may in actuality help it spread, but by identifying why this is happening, we can develop better strategies that allow these drugs to treat tumors the way they should.”

Photo courtesy of
The Wistar Institute
Although PI3K inhibitors have been designed to treat cancer, new research indicates these drugs may actually exacerbate the disease.
Researchers found evidence to suggest that treatment with PI3K inhibitors alone can promote more aggressive tumor cell behavior and increase the likelihood that cancer will spread.
PI3K inhibitors appeared to reprogram the mitochondria of tumor cells and move them to “strategic” positions for invasion.
However, the researchers believe that targeting mitochondrial function along with PI3K could prevent this effect.
Dario C. Altieri, MD, of The Wistar Institute in Philadelphia, Pennsylvania, and his colleagues described these findings in PNAS.
The researchers decided to investigate how mitochondria are reprogrammed when exposed to PI3K inhibition and how mitochondria might prevent targeted agents from being as effective as expected.
“Our prior studies have confirmed that tumor cells rely on energy produced by mitochondria more significantly than previously thought,” Dr Altieri said.
“What we have shown in this study is that, in somewhat of a paradox, treatment with a PI3K inhibitor causes a tumor cell’s mitochondria to produce energy in a localized manner, promoting a far more aggressive and invasive phenotype. The treatment appears to be doing the opposite of its intended effect.”
The study showed that treatment with a PI3K inhibitor causes the mitochondria to migrate to the peripheral cytoskeleton of the tumor cells.
While the mitochondria in untreated cells cluster around the cell’s nucleus, exposure of tumor cells to PI3K therapy causes the mitochondria to move to specialized regions of the cell’s membrane implicated in cell motility and invasion.
In this “strategic” position, tumor mitochondria are ideally positioned to provide a concentrated source of energy to support an increase in cell migration and invasion.
However, the researchers said the dependence of this response on mitochondrial function may offer a new therapeutic angle.
Dr Altieri and his team have shown that targeting mitochondrial functions for tumor therapy is feasible and dramatically enhances the anticancer activity of PI3K inhibitors when used in combination.
“These findings continue to support the idea that the mitochondria of tumor cells are crucial to tumor survival and proliferation,” Dr Altieri said. “It’s certainly counterintuitive that a drug designed to fight cancer may in actuality help it spread, but by identifying why this is happening, we can develop better strategies that allow these drugs to treat tumors the way they should.”

Photo courtesy of
The Wistar Institute
Although PI3K inhibitors have been designed to treat cancer, new research indicates these drugs may actually exacerbate the disease.
Researchers found evidence to suggest that treatment with PI3K inhibitors alone can promote more aggressive tumor cell behavior and increase the likelihood that cancer will spread.
PI3K inhibitors appeared to reprogram the mitochondria of tumor cells and move them to “strategic” positions for invasion.
However, the researchers believe that targeting mitochondrial function along with PI3K could prevent this effect.
Dario C. Altieri, MD, of The Wistar Institute in Philadelphia, Pennsylvania, and his colleagues described these findings in PNAS.
The researchers decided to investigate how mitochondria are reprogrammed when exposed to PI3K inhibition and how mitochondria might prevent targeted agents from being as effective as expected.
“Our prior studies have confirmed that tumor cells rely on energy produced by mitochondria more significantly than previously thought,” Dr Altieri said.
“What we have shown in this study is that, in somewhat of a paradox, treatment with a PI3K inhibitor causes a tumor cell’s mitochondria to produce energy in a localized manner, promoting a far more aggressive and invasive phenotype. The treatment appears to be doing the opposite of its intended effect.”
The study showed that treatment with a PI3K inhibitor causes the mitochondria to migrate to the peripheral cytoskeleton of the tumor cells.
While the mitochondria in untreated cells cluster around the cell’s nucleus, exposure of tumor cells to PI3K therapy causes the mitochondria to move to specialized regions of the cell’s membrane implicated in cell motility and invasion.
In this “strategic” position, tumor mitochondria are ideally positioned to provide a concentrated source of energy to support an increase in cell migration and invasion.
However, the researchers said the dependence of this response on mitochondrial function may offer a new therapeutic angle.
Dr Altieri and his team have shown that targeting mitochondrial functions for tumor therapy is feasible and dramatically enhances the anticancer activity of PI3K inhibitors when used in combination.
“These findings continue to support the idea that the mitochondria of tumor cells are crucial to tumor survival and proliferation,” Dr Altieri said. “It’s certainly counterintuitive that a drug designed to fight cancer may in actuality help it spread, but by identifying why this is happening, we can develop better strategies that allow these drugs to treat tumors the way they should.”
New insight into blood production

in the bone marrow
Scientists say they have identified a family of multipotent progenitor (MPP) cells that are the first to arise from hematopoietic stem cells (HSCs).
The team said their discovery, published in Cell Stem Cell, raises the possibility that, by manipulating the fates of MPPs or HSCs, researchers could one day help overcome imbalances and deficiencies that can arise in the blood system due to aging or leukemia.
Similar imbalances can render patients vulnerable immediately following HSC transplants, especially following umbilical cord transplants, said study author Emmanuelle Passegué, PhD, of the University of California San Francisco.
Via experiments in mice, Dr Passegué and her colleagues found that HSCs “make educated decisions” when it comes time to differentiate.
“Previously, researchers thought that the developmental paths of daughter cells were randomly specified by the HSC, but we conclude that the HSC normally responds appropriately to signals in the environment, making the different MPPs in parallel, but at different speeds and in different amounts to meet the body’s needs,” Dr Passegué said.
To uncover these findings, she and her colleagues investigated patterns of gene expression and cell signaling that determine which developmental paths are favored when relatively rare HSCs spin off daughter cells. The team also explored HSCs’ responses during transplant.
The researchers found that the first daughter cells that arise from HSCs are already distinct, favoring the development of different specialized cell lineages.
The team identified two types of daughter cells, MPP2 and MPP3, which, under normal conditions, are rare. These cells work together with more common daughter cells, MPP4 cells, to control blood production.
MPP2 cells favor the production of platelets and red blood cells, while MPP3 cells favor production of inflammatory cells.
MPP4 cells are the main producers of lymphocytes that fight specific disease pathogens, but the researchers showed that MPP4 cells can easily be re-educated to make many inflammatory cells when regenerative needs are high, as they are following a transplant.
The team found that, during transplant, regenerating HSCs limit their own self-renewal and instead go to work overproducing MPP2 and MPP3 cells that quickly produce needed red blood cells, platelets, and inflammatory cells. Only later does MPP4 production return to normal, enabling the immune system to replenish lymphocytes.
“It will be compelling to test whether the developmental pathways leading to cell specialization can be manipulated to favor production of specific lineage-biased MPPs in order to optimize blood recovery following hematopoietic injury or to rebalance the output of various cell lineages in an aging or deregulated blood system,” Dr Passegué said.

in the bone marrow
Scientists say they have identified a family of multipotent progenitor (MPP) cells that are the first to arise from hematopoietic stem cells (HSCs).
The team said their discovery, published in Cell Stem Cell, raises the possibility that, by manipulating the fates of MPPs or HSCs, researchers could one day help overcome imbalances and deficiencies that can arise in the blood system due to aging or leukemia.
Similar imbalances can render patients vulnerable immediately following HSC transplants, especially following umbilical cord transplants, said study author Emmanuelle Passegué, PhD, of the University of California San Francisco.
Via experiments in mice, Dr Passegué and her colleagues found that HSCs “make educated decisions” when it comes time to differentiate.
“Previously, researchers thought that the developmental paths of daughter cells were randomly specified by the HSC, but we conclude that the HSC normally responds appropriately to signals in the environment, making the different MPPs in parallel, but at different speeds and in different amounts to meet the body’s needs,” Dr Passegué said.
To uncover these findings, she and her colleagues investigated patterns of gene expression and cell signaling that determine which developmental paths are favored when relatively rare HSCs spin off daughter cells. The team also explored HSCs’ responses during transplant.
The researchers found that the first daughter cells that arise from HSCs are already distinct, favoring the development of different specialized cell lineages.
The team identified two types of daughter cells, MPP2 and MPP3, which, under normal conditions, are rare. These cells work together with more common daughter cells, MPP4 cells, to control blood production.
MPP2 cells favor the production of platelets and red blood cells, while MPP3 cells favor production of inflammatory cells.
MPP4 cells are the main producers of lymphocytes that fight specific disease pathogens, but the researchers showed that MPP4 cells can easily be re-educated to make many inflammatory cells when regenerative needs are high, as they are following a transplant.
The team found that, during transplant, regenerating HSCs limit their own self-renewal and instead go to work overproducing MPP2 and MPP3 cells that quickly produce needed red blood cells, platelets, and inflammatory cells. Only later does MPP4 production return to normal, enabling the immune system to replenish lymphocytes.
“It will be compelling to test whether the developmental pathways leading to cell specialization can be manipulated to favor production of specific lineage-biased MPPs in order to optimize blood recovery following hematopoietic injury or to rebalance the output of various cell lineages in an aging or deregulated blood system,” Dr Passegué said.

in the bone marrow
Scientists say they have identified a family of multipotent progenitor (MPP) cells that are the first to arise from hematopoietic stem cells (HSCs).
The team said their discovery, published in Cell Stem Cell, raises the possibility that, by manipulating the fates of MPPs or HSCs, researchers could one day help overcome imbalances and deficiencies that can arise in the blood system due to aging or leukemia.
Similar imbalances can render patients vulnerable immediately following HSC transplants, especially following umbilical cord transplants, said study author Emmanuelle Passegué, PhD, of the University of California San Francisco.
Via experiments in mice, Dr Passegué and her colleagues found that HSCs “make educated decisions” when it comes time to differentiate.
“Previously, researchers thought that the developmental paths of daughter cells were randomly specified by the HSC, but we conclude that the HSC normally responds appropriately to signals in the environment, making the different MPPs in parallel, but at different speeds and in different amounts to meet the body’s needs,” Dr Passegué said.
To uncover these findings, she and her colleagues investigated patterns of gene expression and cell signaling that determine which developmental paths are favored when relatively rare HSCs spin off daughter cells. The team also explored HSCs’ responses during transplant.
The researchers found that the first daughter cells that arise from HSCs are already distinct, favoring the development of different specialized cell lineages.
The team identified two types of daughter cells, MPP2 and MPP3, which, under normal conditions, are rare. These cells work together with more common daughter cells, MPP4 cells, to control blood production.
MPP2 cells favor the production of platelets and red blood cells, while MPP3 cells favor production of inflammatory cells.
MPP4 cells are the main producers of lymphocytes that fight specific disease pathogens, but the researchers showed that MPP4 cells can easily be re-educated to make many inflammatory cells when regenerative needs are high, as they are following a transplant.
The team found that, during transplant, regenerating HSCs limit their own self-renewal and instead go to work overproducing MPP2 and MPP3 cells that quickly produce needed red blood cells, platelets, and inflammatory cells. Only later does MPP4 production return to normal, enabling the immune system to replenish lymphocytes.
“It will be compelling to test whether the developmental pathways leading to cell specialization can be manipulated to favor production of specific lineage-biased MPPs in order to optimize blood recovery following hematopoietic injury or to rebalance the output of various cell lineages in an aging or deregulated blood system,” Dr Passegué said.