User login
Team discovers how cerebral malaria kills children
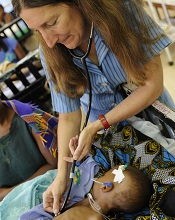
at Queen Elizabeth Hospital
Photo by Jim Peck
After grant money brought magnetic resonance imaging (MRI) to a hospital in Africa, researchers were able to uncover the cause of death in children with cerebral malaria.
MRI scans revealed that, in some children, the brain can become so swollen that it is forced out through the bottom of the skull and compresses the brain stem. This pressure causes the children to stop breathing and die.
The researchers reported these findings in NEJM.
“Because we know now that the brain swelling is what causes death, we can work to find new treatments,” said study author Terrie Taylor, DO, of Michigan State University in East Lansing.
“The next step is to identify what’s causing the swelling and then develop treatments targeting those causes. It’s also possible that using ventilators to keep the children breathing until the swelling subsides might save lives, but ventilators are few and far between in Africa at the moment.”
Scans reveal brain swelling
In 2008, GE Healthcare provided a $1 million MRI to the Queen Elizabeth Hospital in Blantyre, Malawi, where Dr Taylor spends 6 months of every year treating and studying children with malaria.
Dr Taylor and her colleagues used the MRI to view brain images from hundreds of children with cerebral malaria, comparing findings in those who died to those who survived.
The team imaged 168 children with cerebral malaria (as defined by the World Health Organization). Fifteen percent (25/168) of the children died. And 84% of these children (21/25) had evidence of severe brain swelling at admission.
In contrast, the researchers found evidence of severe brain swelling in 27% (39/143) of children who survived. And serial MRI scans revealed decreasing brain volume in the survivors who initially had brain swelling.
“We found that survivors’ brains were either never swollen or decreased in size after 2 to 3 days,” Dr Taylor said. “This was a triumphant moment. I wanted to say to the parasite, ‘Ha! You never thought we’d get an MRI, did you?’” ![]()

at Queen Elizabeth Hospital
Photo by Jim Peck
After grant money brought magnetic resonance imaging (MRI) to a hospital in Africa, researchers were able to uncover the cause of death in children with cerebral malaria.
MRI scans revealed that, in some children, the brain can become so swollen that it is forced out through the bottom of the skull and compresses the brain stem. This pressure causes the children to stop breathing and die.
The researchers reported these findings in NEJM.
“Because we know now that the brain swelling is what causes death, we can work to find new treatments,” said study author Terrie Taylor, DO, of Michigan State University in East Lansing.
“The next step is to identify what’s causing the swelling and then develop treatments targeting those causes. It’s also possible that using ventilators to keep the children breathing until the swelling subsides might save lives, but ventilators are few and far between in Africa at the moment.”
Scans reveal brain swelling
In 2008, GE Healthcare provided a $1 million MRI to the Queen Elizabeth Hospital in Blantyre, Malawi, where Dr Taylor spends 6 months of every year treating and studying children with malaria.
Dr Taylor and her colleagues used the MRI to view brain images from hundreds of children with cerebral malaria, comparing findings in those who died to those who survived.
The team imaged 168 children with cerebral malaria (as defined by the World Health Organization). Fifteen percent (25/168) of the children died. And 84% of these children (21/25) had evidence of severe brain swelling at admission.
In contrast, the researchers found evidence of severe brain swelling in 27% (39/143) of children who survived. And serial MRI scans revealed decreasing brain volume in the survivors who initially had brain swelling.
“We found that survivors’ brains were either never swollen or decreased in size after 2 to 3 days,” Dr Taylor said. “This was a triumphant moment. I wanted to say to the parasite, ‘Ha! You never thought we’d get an MRI, did you?’” ![]()

at Queen Elizabeth Hospital
Photo by Jim Peck
After grant money brought magnetic resonance imaging (MRI) to a hospital in Africa, researchers were able to uncover the cause of death in children with cerebral malaria.
MRI scans revealed that, in some children, the brain can become so swollen that it is forced out through the bottom of the skull and compresses the brain stem. This pressure causes the children to stop breathing and die.
The researchers reported these findings in NEJM.
“Because we know now that the brain swelling is what causes death, we can work to find new treatments,” said study author Terrie Taylor, DO, of Michigan State University in East Lansing.
“The next step is to identify what’s causing the swelling and then develop treatments targeting those causes. It’s also possible that using ventilators to keep the children breathing until the swelling subsides might save lives, but ventilators are few and far between in Africa at the moment.”
Scans reveal brain swelling
In 2008, GE Healthcare provided a $1 million MRI to the Queen Elizabeth Hospital in Blantyre, Malawi, where Dr Taylor spends 6 months of every year treating and studying children with malaria.
Dr Taylor and her colleagues used the MRI to view brain images from hundreds of children with cerebral malaria, comparing findings in those who died to those who survived.
The team imaged 168 children with cerebral malaria (as defined by the World Health Organization). Fifteen percent (25/168) of the children died. And 84% of these children (21/25) had evidence of severe brain swelling at admission.
In contrast, the researchers found evidence of severe brain swelling in 27% (39/143) of children who survived. And serial MRI scans revealed decreasing brain volume in the survivors who initially had brain swelling.
“We found that survivors’ brains were either never swollen or decreased in size after 2 to 3 days,” Dr Taylor said. “This was a triumphant moment. I wanted to say to the parasite, ‘Ha! You never thought we’d get an MRI, did you?’” ![]()
Malaria interventions prove insufficient

Photo by James Gathany
Current malaria interventions are failing to control the disease in high-transmission areas of sub-Saharan Africa, according to research published in The American Journal of Tropical Medicine & Hygiene.
A 2-year surveillance study revealed that the incidence of malaria in rural Uganda is high and continues to rise.
Researchers said this study offers the most accurate, comprehensive, and up-to-date measurement of the malaria disease burden in Uganda.
“Our findings suggest that current efforts at controlling malaria may not be as effective as previously believed in high-transmission areas, where the disease is the biggest threat,” said Grant Dorsey, MD, PhD, of the University of California, San Francisco.
“It’s important to tell the less happy story that we have not yet seen advances in more rural areas, including at least 2 sites in Uganda, where transmission has been historically high.”
To reach an accurate assessment of the malaria incidence in Uganda, Dr Dorsey and his colleagues gathered comprehensive surveillance data over 24 months, from August 2011 to September 2013.
Ultimately, the team evaluated 703 children between the ages of 6 months and 10 years. The children were randomly selected from 3 areas of Uganda with differing malaria characteristics.
The researchers found the incidence of malaria infection decreased in the relatively low-transmission, peri-urban Walukuba area during the study period—from an average of 0.51 to 0.31 episodes of malaria per person per year (P=0.001).
However, the incidence increased in the 2 rural areas. Episodes of malaria per person per year rose from an average of 0.97 to 1.93 (P<0.001) in the moderate-transmission area of Kihihi and rose from an average of 2.33 to 3.30 (P<0.001) in Nagongera, a high-transmission rural area near the southeastern border with Kenya.
Throughout the study period, families were provided with bednets and had access to 24-hour medical care free of charge at a designated study clinic for episodes of fever. The children were also routinely tested for malaria every 3 months, whether they had symptoms or not.
In addition, the researchers collected mosquito specimens monthly from light traps that were strategically placed in each house to estimate the percentages of malaria-carrying mosquitoes in the study areas.
Healthcare workers provided over 2500 treatments for malaria over the course of the study.
“Children in our study experienced a significantly high rate of infection, and that rate increased in the 2 rural areas,” Dr Dorsey said. “Based on prior data, our higher transmission sites are very likely to be representative of most of Uganda and perhaps of most other rural areas in sub-Saharan Africa as well.”
The researchers said these results suggest a need to further scale up campaigns to distribute insecticide-treated bednets and spray homes with insecticides. And high-transmission countries like Uganda may also require new interventions, such as using malaria drugs for prevention and controlling mosquito larvae, in order to match the malaria reduction successes seen elsewhere in the world.
In a related editorial, Steven Meshnick, MD, PhD, of the University of North Carolina, Chapel Hill, wrote, “The real take-home message of this study may be that malaria control in Africa requires sustained and consistent efforts over much more than 2 years.” ![]()

Photo by James Gathany
Current malaria interventions are failing to control the disease in high-transmission areas of sub-Saharan Africa, according to research published in The American Journal of Tropical Medicine & Hygiene.
A 2-year surveillance study revealed that the incidence of malaria in rural Uganda is high and continues to rise.
Researchers said this study offers the most accurate, comprehensive, and up-to-date measurement of the malaria disease burden in Uganda.
“Our findings suggest that current efforts at controlling malaria may not be as effective as previously believed in high-transmission areas, where the disease is the biggest threat,” said Grant Dorsey, MD, PhD, of the University of California, San Francisco.
“It’s important to tell the less happy story that we have not yet seen advances in more rural areas, including at least 2 sites in Uganda, where transmission has been historically high.”
To reach an accurate assessment of the malaria incidence in Uganda, Dr Dorsey and his colleagues gathered comprehensive surveillance data over 24 months, from August 2011 to September 2013.
Ultimately, the team evaluated 703 children between the ages of 6 months and 10 years. The children were randomly selected from 3 areas of Uganda with differing malaria characteristics.
The researchers found the incidence of malaria infection decreased in the relatively low-transmission, peri-urban Walukuba area during the study period—from an average of 0.51 to 0.31 episodes of malaria per person per year (P=0.001).
However, the incidence increased in the 2 rural areas. Episodes of malaria per person per year rose from an average of 0.97 to 1.93 (P<0.001) in the moderate-transmission area of Kihihi and rose from an average of 2.33 to 3.30 (P<0.001) in Nagongera, a high-transmission rural area near the southeastern border with Kenya.
Throughout the study period, families were provided with bednets and had access to 24-hour medical care free of charge at a designated study clinic for episodes of fever. The children were also routinely tested for malaria every 3 months, whether they had symptoms or not.
In addition, the researchers collected mosquito specimens monthly from light traps that were strategically placed in each house to estimate the percentages of malaria-carrying mosquitoes in the study areas.
Healthcare workers provided over 2500 treatments for malaria over the course of the study.
“Children in our study experienced a significantly high rate of infection, and that rate increased in the 2 rural areas,” Dr Dorsey said. “Based on prior data, our higher transmission sites are very likely to be representative of most of Uganda and perhaps of most other rural areas in sub-Saharan Africa as well.”
The researchers said these results suggest a need to further scale up campaigns to distribute insecticide-treated bednets and spray homes with insecticides. And high-transmission countries like Uganda may also require new interventions, such as using malaria drugs for prevention and controlling mosquito larvae, in order to match the malaria reduction successes seen elsewhere in the world.
In a related editorial, Steven Meshnick, MD, PhD, of the University of North Carolina, Chapel Hill, wrote, “The real take-home message of this study may be that malaria control in Africa requires sustained and consistent efforts over much more than 2 years.” ![]()

Photo by James Gathany
Current malaria interventions are failing to control the disease in high-transmission areas of sub-Saharan Africa, according to research published in The American Journal of Tropical Medicine & Hygiene.
A 2-year surveillance study revealed that the incidence of malaria in rural Uganda is high and continues to rise.
Researchers said this study offers the most accurate, comprehensive, and up-to-date measurement of the malaria disease burden in Uganda.
“Our findings suggest that current efforts at controlling malaria may not be as effective as previously believed in high-transmission areas, where the disease is the biggest threat,” said Grant Dorsey, MD, PhD, of the University of California, San Francisco.
“It’s important to tell the less happy story that we have not yet seen advances in more rural areas, including at least 2 sites in Uganda, where transmission has been historically high.”
To reach an accurate assessment of the malaria incidence in Uganda, Dr Dorsey and his colleagues gathered comprehensive surveillance data over 24 months, from August 2011 to September 2013.
Ultimately, the team evaluated 703 children between the ages of 6 months and 10 years. The children were randomly selected from 3 areas of Uganda with differing malaria characteristics.
The researchers found the incidence of malaria infection decreased in the relatively low-transmission, peri-urban Walukuba area during the study period—from an average of 0.51 to 0.31 episodes of malaria per person per year (P=0.001).
However, the incidence increased in the 2 rural areas. Episodes of malaria per person per year rose from an average of 0.97 to 1.93 (P<0.001) in the moderate-transmission area of Kihihi and rose from an average of 2.33 to 3.30 (P<0.001) in Nagongera, a high-transmission rural area near the southeastern border with Kenya.
Throughout the study period, families were provided with bednets and had access to 24-hour medical care free of charge at a designated study clinic for episodes of fever. The children were also routinely tested for malaria every 3 months, whether they had symptoms or not.
In addition, the researchers collected mosquito specimens monthly from light traps that were strategically placed in each house to estimate the percentages of malaria-carrying mosquitoes in the study areas.
Healthcare workers provided over 2500 treatments for malaria over the course of the study.
“Children in our study experienced a significantly high rate of infection, and that rate increased in the 2 rural areas,” Dr Dorsey said. “Based on prior data, our higher transmission sites are very likely to be representative of most of Uganda and perhaps of most other rural areas in sub-Saharan Africa as well.”
The researchers said these results suggest a need to further scale up campaigns to distribute insecticide-treated bednets and spray homes with insecticides. And high-transmission countries like Uganda may also require new interventions, such as using malaria drugs for prevention and controlling mosquito larvae, in order to match the malaria reduction successes seen elsewhere in the world.
In a related editorial, Steven Meshnick, MD, PhD, of the University of North Carolina, Chapel Hill, wrote, “The real take-home message of this study may be that malaria control in Africa requires sustained and consistent efforts over much more than 2 years.” ![]()
Experts urge review of global sepsis guidelines

Investigators are calling for a global review of guidelines used to diagnose sepsis, after a study showed that 1 in 8 patients with infections severe enough to necessitate admission to an intensive care unit did not meet current diagnostic criteria.
The researchers identified 109,663 patients with possible sepsis who had infection and organ failure. However, more than 13,000 patients from this group did not meet the classic criteria used to diagnose sepsis.
“To be diagnosed with sepsis, a patient must be thought to have an infection and exhibit at least 2 of the following criteria: abnormal body temperature or white blood cell count, high heart rate, high respiratory rate, or low carbon dioxide level in the blood,” said Maija Kaukonen, MD, PhD, of Monash University in Melbourne, Victoria, Australia.
“But our study found that many patients—for example, the elderly or those on medications that affect heart rate or the immune system—may not meet the classic criteria to diagnose sepsis, despite having severe infections and organ failure. If we continue to use these criteria, we may miss the opportunity to identify many critically ill patients with sepsis.”
The study was published in NEJM.
The investigators studied 1,171,797 patients from 172 intensive care units in New Zealand and Australia.
The team identified patients with infection and organ failure and categorized them according to whether they had signs meeting 2 or more systemic inflammatory response syndrome (SIRS) criteria (SIRS-positive severe sepsis) or less than 2 SIRS criteria (SIRS-negative severe sepsis).
Of the 109,663 patients who had infection and organ failure, 96,385 (87.9%) had SIRS-positive severe sepsis and 13,278 (12.1%) had SIRS-negative severe sepsis.
Over 14 years, the 2 patient groups had similar characteristics and changes in mortality. Mortality decreased from 36.1% (829/2296) to 18.3% (2037/11,119) in the SIRS-positive group (P<0.001) and from 27.7% (100/361) to 9.3% (122/1315) in the SIRS-negative group (P<0.001).
This similarity between the groups remained after the researchers adjusted their analysis for baseline characteristics. In both groups, the odds ratio was 0.96 (P=0.12).
The investigators also noted that, in the adjusted analysis, mortality increased linearly with each additional SIRS criterion (P<0.001), without any transitional increase in risk at a threshold of 2 SIRS criteria.
Rinaldo Bellomo, MD, PhD, also of Monash University, conceived this study. He said that although the classic definition of sepsis has been widely used throughout the world, he believed that, after 20 years, it was time for it to be reviewed.
“There are clear signs from this study that if we continue to use these criteria, we may fail to identify septic patients and, therefore, potentially delay their treatment,” he said. ![]()

Investigators are calling for a global review of guidelines used to diagnose sepsis, after a study showed that 1 in 8 patients with infections severe enough to necessitate admission to an intensive care unit did not meet current diagnostic criteria.
The researchers identified 109,663 patients with possible sepsis who had infection and organ failure. However, more than 13,000 patients from this group did not meet the classic criteria used to diagnose sepsis.
“To be diagnosed with sepsis, a patient must be thought to have an infection and exhibit at least 2 of the following criteria: abnormal body temperature or white blood cell count, high heart rate, high respiratory rate, or low carbon dioxide level in the blood,” said Maija Kaukonen, MD, PhD, of Monash University in Melbourne, Victoria, Australia.
“But our study found that many patients—for example, the elderly or those on medications that affect heart rate or the immune system—may not meet the classic criteria to diagnose sepsis, despite having severe infections and organ failure. If we continue to use these criteria, we may miss the opportunity to identify many critically ill patients with sepsis.”
The study was published in NEJM.
The investigators studied 1,171,797 patients from 172 intensive care units in New Zealand and Australia.
The team identified patients with infection and organ failure and categorized them according to whether they had signs meeting 2 or more systemic inflammatory response syndrome (SIRS) criteria (SIRS-positive severe sepsis) or less than 2 SIRS criteria (SIRS-negative severe sepsis).
Of the 109,663 patients who had infection and organ failure, 96,385 (87.9%) had SIRS-positive severe sepsis and 13,278 (12.1%) had SIRS-negative severe sepsis.
Over 14 years, the 2 patient groups had similar characteristics and changes in mortality. Mortality decreased from 36.1% (829/2296) to 18.3% (2037/11,119) in the SIRS-positive group (P<0.001) and from 27.7% (100/361) to 9.3% (122/1315) in the SIRS-negative group (P<0.001).
This similarity between the groups remained after the researchers adjusted their analysis for baseline characteristics. In both groups, the odds ratio was 0.96 (P=0.12).
The investigators also noted that, in the adjusted analysis, mortality increased linearly with each additional SIRS criterion (P<0.001), without any transitional increase in risk at a threshold of 2 SIRS criteria.
Rinaldo Bellomo, MD, PhD, also of Monash University, conceived this study. He said that although the classic definition of sepsis has been widely used throughout the world, he believed that, after 20 years, it was time for it to be reviewed.
“There are clear signs from this study that if we continue to use these criteria, we may fail to identify septic patients and, therefore, potentially delay their treatment,” he said. ![]()

Investigators are calling for a global review of guidelines used to diagnose sepsis, after a study showed that 1 in 8 patients with infections severe enough to necessitate admission to an intensive care unit did not meet current diagnostic criteria.
The researchers identified 109,663 patients with possible sepsis who had infection and organ failure. However, more than 13,000 patients from this group did not meet the classic criteria used to diagnose sepsis.
“To be diagnosed with sepsis, a patient must be thought to have an infection and exhibit at least 2 of the following criteria: abnormal body temperature or white blood cell count, high heart rate, high respiratory rate, or low carbon dioxide level in the blood,” said Maija Kaukonen, MD, PhD, of Monash University in Melbourne, Victoria, Australia.
“But our study found that many patients—for example, the elderly or those on medications that affect heart rate or the immune system—may not meet the classic criteria to diagnose sepsis, despite having severe infections and organ failure. If we continue to use these criteria, we may miss the opportunity to identify many critically ill patients with sepsis.”
The study was published in NEJM.
The investigators studied 1,171,797 patients from 172 intensive care units in New Zealand and Australia.
The team identified patients with infection and organ failure and categorized them according to whether they had signs meeting 2 or more systemic inflammatory response syndrome (SIRS) criteria (SIRS-positive severe sepsis) or less than 2 SIRS criteria (SIRS-negative severe sepsis).
Of the 109,663 patients who had infection and organ failure, 96,385 (87.9%) had SIRS-positive severe sepsis and 13,278 (12.1%) had SIRS-negative severe sepsis.
Over 14 years, the 2 patient groups had similar characteristics and changes in mortality. Mortality decreased from 36.1% (829/2296) to 18.3% (2037/11,119) in the SIRS-positive group (P<0.001) and from 27.7% (100/361) to 9.3% (122/1315) in the SIRS-negative group (P<0.001).
This similarity between the groups remained after the researchers adjusted their analysis for baseline characteristics. In both groups, the odds ratio was 0.96 (P=0.12).
The investigators also noted that, in the adjusted analysis, mortality increased linearly with each additional SIRS criterion (P<0.001), without any transitional increase in risk at a threshold of 2 SIRS criteria.
Rinaldo Bellomo, MD, PhD, also of Monash University, conceived this study. He said that although the classic definition of sepsis has been widely used throughout the world, he believed that, after 20 years, it was time for it to be reviewed.
“There are clear signs from this study that if we continue to use these criteria, we may fail to identify septic patients and, therefore, potentially delay their treatment,” he said. ![]()
Lowering the cost of cancer drugs in the US

Photo by Petr Kratochvil
Increasingly high prices for cancer drugs are affecting patient care and the overall healthcare system in the US, according to authors of an article in Mayo Clinic Proceedings.
The authors noted that the average price of cancer drugs for about a year of therapy increased from $5000 to $10,000 before 2000, and to more than $100,000 by 2012.
Over nearly the same period, the average household income in the US decreased by about 8%.
“Americans with cancer pay 50% to 100% more for the same patented drug than patients in other countries,” said author S. Vincent Rajkumar, MD, of the Mayo Clinic in Rochester, Minnesota.
“As oncologists, we have a moral obligation to advocate for affordable cancer drugs for our patients.”
Dr Rajkumar and co-author Hagop Kantarjian, MD, of MD Anderson Cancer Center in Houston, Texas, rebutted the major arguments the pharmaceutical industry uses to justify the high price of cancer drugs; namely, the expense of conducting research and drug development, the comparative benefits to patients, that market forces will settle prices to reasonable levels, and that price controls on cancer drugs will stifle innovation.
“One of the facts that people do not realize is that cancer drugs, for the most part, are not operating under a free market economy,” Dr Rajkumar said. “The fact that there are 5 approved drugs to treat an incurable cancer does not mean there is competition.”
“Typically, the standard of care is that each drug is used sequentially or in combination, so that each new drug represents a monopoly with exclusivity granted by patent protection for many years.”
Drs Rajkumar and Kantarjian said other reasons for the high cost of cancer drugs include legislation that prevents Medicare from being able to negotiate drug prices and a lack of value-based pricing, which ties the cost of a drug to its relative effectiveness compared to other drugs.
The authors recommended a set of potential solutions to help control and reduce the high cost of cancer drugs in the US. Some of their recommendations are already in practice in other developed countries. Their recommendations include:
- Allow Medicare to negotiate drug prices
- Develop cancer treatment pathways/guidelines that incorporate the cost and benefit of cancer drugs
- Allow the Food and Drug Administration or physician panels to recommend target prices based on a drug’s magnitude of benefit (value-based pricing)
- Eliminate “pay-for-delay” strategies in which a pharmaceutical company with a brand name drug shares profits on that drug with a generic drug manufacturer for the remainder of a patent period, effectively eliminating a patent challenge and competition
- Allow the importation of drugs from abroad for personal use
- Allow the Patient-Centered Outcomes Research Institute and other cancer advocacy groups to consider cost in their recommendations
- Create patient-driven grassroots movements and organizations to advocate effectively for the interests of patients with cancer to balance advocacy efforts of pharmaceutical companies, insurance companies, pharmacy outlets, and hospitals.
Dr Kantarjian has organized a petition, which is available on change.org, asking the federal government to implement these changes. ![]()

Photo by Petr Kratochvil
Increasingly high prices for cancer drugs are affecting patient care and the overall healthcare system in the US, according to authors of an article in Mayo Clinic Proceedings.
The authors noted that the average price of cancer drugs for about a year of therapy increased from $5000 to $10,000 before 2000, and to more than $100,000 by 2012.
Over nearly the same period, the average household income in the US decreased by about 8%.
“Americans with cancer pay 50% to 100% more for the same patented drug than patients in other countries,” said author S. Vincent Rajkumar, MD, of the Mayo Clinic in Rochester, Minnesota.
“As oncologists, we have a moral obligation to advocate for affordable cancer drugs for our patients.”
Dr Rajkumar and co-author Hagop Kantarjian, MD, of MD Anderson Cancer Center in Houston, Texas, rebutted the major arguments the pharmaceutical industry uses to justify the high price of cancer drugs; namely, the expense of conducting research and drug development, the comparative benefits to patients, that market forces will settle prices to reasonable levels, and that price controls on cancer drugs will stifle innovation.
“One of the facts that people do not realize is that cancer drugs, for the most part, are not operating under a free market economy,” Dr Rajkumar said. “The fact that there are 5 approved drugs to treat an incurable cancer does not mean there is competition.”
“Typically, the standard of care is that each drug is used sequentially or in combination, so that each new drug represents a monopoly with exclusivity granted by patent protection for many years.”
Drs Rajkumar and Kantarjian said other reasons for the high cost of cancer drugs include legislation that prevents Medicare from being able to negotiate drug prices and a lack of value-based pricing, which ties the cost of a drug to its relative effectiveness compared to other drugs.
The authors recommended a set of potential solutions to help control and reduce the high cost of cancer drugs in the US. Some of their recommendations are already in practice in other developed countries. Their recommendations include:
- Allow Medicare to negotiate drug prices
- Develop cancer treatment pathways/guidelines that incorporate the cost and benefit of cancer drugs
- Allow the Food and Drug Administration or physician panels to recommend target prices based on a drug’s magnitude of benefit (value-based pricing)
- Eliminate “pay-for-delay” strategies in which a pharmaceutical company with a brand name drug shares profits on that drug with a generic drug manufacturer for the remainder of a patent period, effectively eliminating a patent challenge and competition
- Allow the importation of drugs from abroad for personal use
- Allow the Patient-Centered Outcomes Research Institute and other cancer advocacy groups to consider cost in their recommendations
- Create patient-driven grassroots movements and organizations to advocate effectively for the interests of patients with cancer to balance advocacy efforts of pharmaceutical companies, insurance companies, pharmacy outlets, and hospitals.
Dr Kantarjian has organized a petition, which is available on change.org, asking the federal government to implement these changes. ![]()

Photo by Petr Kratochvil
Increasingly high prices for cancer drugs are affecting patient care and the overall healthcare system in the US, according to authors of an article in Mayo Clinic Proceedings.
The authors noted that the average price of cancer drugs for about a year of therapy increased from $5000 to $10,000 before 2000, and to more than $100,000 by 2012.
Over nearly the same period, the average household income in the US decreased by about 8%.
“Americans with cancer pay 50% to 100% more for the same patented drug than patients in other countries,” said author S. Vincent Rajkumar, MD, of the Mayo Clinic in Rochester, Minnesota.
“As oncologists, we have a moral obligation to advocate for affordable cancer drugs for our patients.”
Dr Rajkumar and co-author Hagop Kantarjian, MD, of MD Anderson Cancer Center in Houston, Texas, rebutted the major arguments the pharmaceutical industry uses to justify the high price of cancer drugs; namely, the expense of conducting research and drug development, the comparative benefits to patients, that market forces will settle prices to reasonable levels, and that price controls on cancer drugs will stifle innovation.
“One of the facts that people do not realize is that cancer drugs, for the most part, are not operating under a free market economy,” Dr Rajkumar said. “The fact that there are 5 approved drugs to treat an incurable cancer does not mean there is competition.”
“Typically, the standard of care is that each drug is used sequentially or in combination, so that each new drug represents a monopoly with exclusivity granted by patent protection for many years.”
Drs Rajkumar and Kantarjian said other reasons for the high cost of cancer drugs include legislation that prevents Medicare from being able to negotiate drug prices and a lack of value-based pricing, which ties the cost of a drug to its relative effectiveness compared to other drugs.
The authors recommended a set of potential solutions to help control and reduce the high cost of cancer drugs in the US. Some of their recommendations are already in practice in other developed countries. Their recommendations include:
- Allow Medicare to negotiate drug prices
- Develop cancer treatment pathways/guidelines that incorporate the cost and benefit of cancer drugs
- Allow the Food and Drug Administration or physician panels to recommend target prices based on a drug’s magnitude of benefit (value-based pricing)
- Eliminate “pay-for-delay” strategies in which a pharmaceutical company with a brand name drug shares profits on that drug with a generic drug manufacturer for the remainder of a patent period, effectively eliminating a patent challenge and competition
- Allow the importation of drugs from abroad for personal use
- Allow the Patient-Centered Outcomes Research Institute and other cancer advocacy groups to consider cost in their recommendations
- Create patient-driven grassroots movements and organizations to advocate effectively for the interests of patients with cancer to balance advocacy efforts of pharmaceutical companies, insurance companies, pharmacy outlets, and hospitals.
Dr Kantarjian has organized a petition, which is available on change.org, asking the federal government to implement these changes. ![]()
Clinical trial mandate not being followed

Photo by Esther Dyson
A study published in NEJM suggests that results from most trials on ClinicalTrials.gov are not posted on the site within a year of trial completion or termination, even though this violates the Food and Drug Administration Amendments Act (FDAAA) mandate.
Among all the trials analyzed, those funded by industry were the most likely to be publicly disclosed in a timely fashion, researchers found.
Even so, industry compliance with the FDAAA mandate was poor.
And compliance was worse for research funded by the National Institutes of Health (NIH) and other government and academic institutions.
Study authors said this lack of transparency has created a critical information gap about investigational drugs, devices, and biologic therapies that hampers progress and violates obligations to patients.
“Patients who participate in clinical research have the expectation that the risk of participation will be offset by the creation of generalizable knowledge and the advancement of science, and that is achieved through the availability of clinical trial results,” said study author Monique Anderson, MD, of the Duke Clinical Research Institute in Durham, North Carolina.
“Sponsors who lead clinical trials have an ethical and legal obligation to publically report their findings, whether the results are positive or negative.”
In 2000, the US Congress authorized the creation of the ClinicalTrials.gov registry to provide information about clinical trials. Seven years later, the FDAAA mandated that sponsors of most trials begin registering and reporting basic summary results on the registry so the American public could have access to the resulting data.
The requirement covers non-phase-1 trials of drugs, medical devices, or biologics that had at least 1 US research site. Trial results are to be reported by the sponsor within a year of completing data collection.
To gauge compliance with this mandate, Dr Anderson and her colleagues searched trials posted on ClinicalTrials.gov. They identified 13,327 trials that were likely to be subject to FDAAA provisions and were terminated or completed from January 1, 2008, through August 31, 2012.
Most trials were industry-funded (65.6%), 14.2% were funded by the NIH, and the remaining 20.2% were funded by other government or academic institutions.
In all, 13.4% of trials had results posted on ClinicalTrials.gov within 12 months of trial completion/termination, and 6.1% of trials did not have results reported but had a legally acceptable delay because of a certification or an exemption request.
A higher percentage of trials—38.3%—had results posted on ClinicalTrials.gov at any time during the 5-year study period.
By September 27, 2013, sponsors for 15.8% of all the trials analyzed had submitted a certification or extension request to delay reporting to ClinicalTrials.gov. Of these, 23.0% had results reported during the study period.
At 12 months, 17% of industry-funded trials had results posted on ClinicalTrials.gov, as did 8.1% of trials funded by the NIH, and 5.7% of trials funded by other government or academic institutions. At 5 years, those percentages had increased to 41.5%, 38.9%, and 27.7%, respectively.
“The law requiring public disclosure was enacted amid concerns that sponsors and investigators were selectively publishing trials that favored sponsors’ interests,” Dr Anderson said. “Industry sponsors in particular were criticized for selective reporting.”
“Since the law’s enactment, many companies have developed disclosure policies and have actively pursued expanded public disclosure of data, but there may be a lack of knowledge about the law in academia, or a lack of resources to ensure timely reporting.”
Dr Anderson said penalties for failing to submit data within the 1-year reporting period could be as high as $10,000 a day and/or the loss of NIH funding, but enforcement has not occurred, pending a rule approval. ![]()

Photo by Esther Dyson
A study published in NEJM suggests that results from most trials on ClinicalTrials.gov are not posted on the site within a year of trial completion or termination, even though this violates the Food and Drug Administration Amendments Act (FDAAA) mandate.
Among all the trials analyzed, those funded by industry were the most likely to be publicly disclosed in a timely fashion, researchers found.
Even so, industry compliance with the FDAAA mandate was poor.
And compliance was worse for research funded by the National Institutes of Health (NIH) and other government and academic institutions.
Study authors said this lack of transparency has created a critical information gap about investigational drugs, devices, and biologic therapies that hampers progress and violates obligations to patients.
“Patients who participate in clinical research have the expectation that the risk of participation will be offset by the creation of generalizable knowledge and the advancement of science, and that is achieved through the availability of clinical trial results,” said study author Monique Anderson, MD, of the Duke Clinical Research Institute in Durham, North Carolina.
“Sponsors who lead clinical trials have an ethical and legal obligation to publically report their findings, whether the results are positive or negative.”
In 2000, the US Congress authorized the creation of the ClinicalTrials.gov registry to provide information about clinical trials. Seven years later, the FDAAA mandated that sponsors of most trials begin registering and reporting basic summary results on the registry so the American public could have access to the resulting data.
The requirement covers non-phase-1 trials of drugs, medical devices, or biologics that had at least 1 US research site. Trial results are to be reported by the sponsor within a year of completing data collection.
To gauge compliance with this mandate, Dr Anderson and her colleagues searched trials posted on ClinicalTrials.gov. They identified 13,327 trials that were likely to be subject to FDAAA provisions and were terminated or completed from January 1, 2008, through August 31, 2012.
Most trials were industry-funded (65.6%), 14.2% were funded by the NIH, and the remaining 20.2% were funded by other government or academic institutions.
In all, 13.4% of trials had results posted on ClinicalTrials.gov within 12 months of trial completion/termination, and 6.1% of trials did not have results reported but had a legally acceptable delay because of a certification or an exemption request.
A higher percentage of trials—38.3%—had results posted on ClinicalTrials.gov at any time during the 5-year study period.
By September 27, 2013, sponsors for 15.8% of all the trials analyzed had submitted a certification or extension request to delay reporting to ClinicalTrials.gov. Of these, 23.0% had results reported during the study period.
At 12 months, 17% of industry-funded trials had results posted on ClinicalTrials.gov, as did 8.1% of trials funded by the NIH, and 5.7% of trials funded by other government or academic institutions. At 5 years, those percentages had increased to 41.5%, 38.9%, and 27.7%, respectively.
“The law requiring public disclosure was enacted amid concerns that sponsors and investigators were selectively publishing trials that favored sponsors’ interests,” Dr Anderson said. “Industry sponsors in particular were criticized for selective reporting.”
“Since the law’s enactment, many companies have developed disclosure policies and have actively pursued expanded public disclosure of data, but there may be a lack of knowledge about the law in academia, or a lack of resources to ensure timely reporting.”
Dr Anderson said penalties for failing to submit data within the 1-year reporting period could be as high as $10,000 a day and/or the loss of NIH funding, but enforcement has not occurred, pending a rule approval. ![]()

Photo by Esther Dyson
A study published in NEJM suggests that results from most trials on ClinicalTrials.gov are not posted on the site within a year of trial completion or termination, even though this violates the Food and Drug Administration Amendments Act (FDAAA) mandate.
Among all the trials analyzed, those funded by industry were the most likely to be publicly disclosed in a timely fashion, researchers found.
Even so, industry compliance with the FDAAA mandate was poor.
And compliance was worse for research funded by the National Institutes of Health (NIH) and other government and academic institutions.
Study authors said this lack of transparency has created a critical information gap about investigational drugs, devices, and biologic therapies that hampers progress and violates obligations to patients.
“Patients who participate in clinical research have the expectation that the risk of participation will be offset by the creation of generalizable knowledge and the advancement of science, and that is achieved through the availability of clinical trial results,” said study author Monique Anderson, MD, of the Duke Clinical Research Institute in Durham, North Carolina.
“Sponsors who lead clinical trials have an ethical and legal obligation to publically report their findings, whether the results are positive or negative.”
In 2000, the US Congress authorized the creation of the ClinicalTrials.gov registry to provide information about clinical trials. Seven years later, the FDAAA mandated that sponsors of most trials begin registering and reporting basic summary results on the registry so the American public could have access to the resulting data.
The requirement covers non-phase-1 trials of drugs, medical devices, or biologics that had at least 1 US research site. Trial results are to be reported by the sponsor within a year of completing data collection.
To gauge compliance with this mandate, Dr Anderson and her colleagues searched trials posted on ClinicalTrials.gov. They identified 13,327 trials that were likely to be subject to FDAAA provisions and were terminated or completed from January 1, 2008, through August 31, 2012.
Most trials were industry-funded (65.6%), 14.2% were funded by the NIH, and the remaining 20.2% were funded by other government or academic institutions.
In all, 13.4% of trials had results posted on ClinicalTrials.gov within 12 months of trial completion/termination, and 6.1% of trials did not have results reported but had a legally acceptable delay because of a certification or an exemption request.
A higher percentage of trials—38.3%—had results posted on ClinicalTrials.gov at any time during the 5-year study period.
By September 27, 2013, sponsors for 15.8% of all the trials analyzed had submitted a certification or extension request to delay reporting to ClinicalTrials.gov. Of these, 23.0% had results reported during the study period.
At 12 months, 17% of industry-funded trials had results posted on ClinicalTrials.gov, as did 8.1% of trials funded by the NIH, and 5.7% of trials funded by other government or academic institutions. At 5 years, those percentages had increased to 41.5%, 38.9%, and 27.7%, respectively.
“The law requiring public disclosure was enacted amid concerns that sponsors and investigators were selectively publishing trials that favored sponsors’ interests,” Dr Anderson said. “Industry sponsors in particular were criticized for selective reporting.”
“Since the law’s enactment, many companies have developed disclosure policies and have actively pursued expanded public disclosure of data, but there may be a lack of knowledge about the law in academia, or a lack of resources to ensure timely reporting.”
Dr Anderson said penalties for failing to submit data within the 1-year reporting period could be as high as $10,000 a day and/or the loss of NIH funding, but enforcement has not occurred, pending a rule approval. ![]()
Rehospitalization after severe sepsis often avoidable

A new analysis indicates that patients hospitalized for severe sepsis are often readmitted within 90 days, and many of these readmissions may be preventable.
About 43% of the patients studied were readmitted to the hospital within 90 days of their sepsis hospitalization.
And 42% of these hospitalizations were due to conditions that could potentially be prevented or treated early to avoid hospitalization, according to researchers.
Hallie C. Prescott, MD, of the University of Michigan, Ann Arbor, and her colleagues reported these findings in JAMA.
The researchers analyzed participants in the nationally representative US Health and Retirement Study, a sample of households with adults 50 years of age or older that is linked to Medicare claims (1998-2010).
The team examined the most common readmission diagnoses among patients who were hospitalized for severe sepsis, the extent to which readmissions might have been preventable, and whether the pattern of readmission diagnoses differed compared with that of other acute medical conditions.
To gauge what proportion of rehospitalizations might have been preventable, the researchers looked at ambulatory-care-sensitive conditions (ACSCs) identified by the Agency for Healthcare Research and Quality. They also expanded the definition of ACSCs to include conditions that aren’t common among the general population but arise more often in sepsis survivors.
So their potentially preventable readmission diagnoses included pneumonia, hypertension, dehydration, asthma, urinary tract infection, chronic obstructive pulmonary disease exacerbation, perforated appendix, diabetes, angina, congestive heart failure, sepsis, acute renal failure, skin or soft tissue infection, and aspiration pneumonitis.
Dr Prescott and her colleagues identified 2617 hospitalizations for severe sepsis that could be matched to hospitalizations for other acute medical conditions. And they found that 1115 of the severe sepsis survivors (42.6%) were rehospitalized within 90 days.
The 10 most common readmission diagnoses following severe sepsis were sepsis (6.4%), congestive heart failure (5.5%), pneumonia (3.5%), acute renal failure (3.3%), rehabilitation (2.8%), respiratory failure (2.5%), complication related to a device, implant, or graft (2%), exacerbation of chronic obstructive pulmonary disorder (1.9%), aspiration pneumonitis (1.8%), and urinary tract infection (1.7%).
Readmissions for a primary diagnosis of infection (sepsis, pneumonia, urinary tract, and skin or soft tissue infection) occurred in 11.9% of severe sepsis survivors and 8.0% of patients with acute medical conditions (P<0.001).
Likewise, readmissions for ACSCs were more common after severe sepsis than for patients with acute conditions—21.6% and 19.1%, respectively (P=0.02)—and accounted for a greater proportion of all 90-day readmissions—41.6% and 37%.1, respectively (P=0.009).
“Many of these conditions can be managed if the patient can get in to see a doctor at the start of the illness, meaning that we potentially avoid hospitalization,” Dr Prescott said. “We need to assess their vulnerability and design a better landing pad for patients when they leave the hospital, and avoid the second hit that derails recovery.” ![]()

A new analysis indicates that patients hospitalized for severe sepsis are often readmitted within 90 days, and many of these readmissions may be preventable.
About 43% of the patients studied were readmitted to the hospital within 90 days of their sepsis hospitalization.
And 42% of these hospitalizations were due to conditions that could potentially be prevented or treated early to avoid hospitalization, according to researchers.
Hallie C. Prescott, MD, of the University of Michigan, Ann Arbor, and her colleagues reported these findings in JAMA.
The researchers analyzed participants in the nationally representative US Health and Retirement Study, a sample of households with adults 50 years of age or older that is linked to Medicare claims (1998-2010).
The team examined the most common readmission diagnoses among patients who were hospitalized for severe sepsis, the extent to which readmissions might have been preventable, and whether the pattern of readmission diagnoses differed compared with that of other acute medical conditions.
To gauge what proportion of rehospitalizations might have been preventable, the researchers looked at ambulatory-care-sensitive conditions (ACSCs) identified by the Agency for Healthcare Research and Quality. They also expanded the definition of ACSCs to include conditions that aren’t common among the general population but arise more often in sepsis survivors.
So their potentially preventable readmission diagnoses included pneumonia, hypertension, dehydration, asthma, urinary tract infection, chronic obstructive pulmonary disease exacerbation, perforated appendix, diabetes, angina, congestive heart failure, sepsis, acute renal failure, skin or soft tissue infection, and aspiration pneumonitis.
Dr Prescott and her colleagues identified 2617 hospitalizations for severe sepsis that could be matched to hospitalizations for other acute medical conditions. And they found that 1115 of the severe sepsis survivors (42.6%) were rehospitalized within 90 days.
The 10 most common readmission diagnoses following severe sepsis were sepsis (6.4%), congestive heart failure (5.5%), pneumonia (3.5%), acute renal failure (3.3%), rehabilitation (2.8%), respiratory failure (2.5%), complication related to a device, implant, or graft (2%), exacerbation of chronic obstructive pulmonary disorder (1.9%), aspiration pneumonitis (1.8%), and urinary tract infection (1.7%).
Readmissions for a primary diagnosis of infection (sepsis, pneumonia, urinary tract, and skin or soft tissue infection) occurred in 11.9% of severe sepsis survivors and 8.0% of patients with acute medical conditions (P<0.001).
Likewise, readmissions for ACSCs were more common after severe sepsis than for patients with acute conditions—21.6% and 19.1%, respectively (P=0.02)—and accounted for a greater proportion of all 90-day readmissions—41.6% and 37%.1, respectively (P=0.009).
“Many of these conditions can be managed if the patient can get in to see a doctor at the start of the illness, meaning that we potentially avoid hospitalization,” Dr Prescott said. “We need to assess their vulnerability and design a better landing pad for patients when they leave the hospital, and avoid the second hit that derails recovery.” ![]()

A new analysis indicates that patients hospitalized for severe sepsis are often readmitted within 90 days, and many of these readmissions may be preventable.
About 43% of the patients studied were readmitted to the hospital within 90 days of their sepsis hospitalization.
And 42% of these hospitalizations were due to conditions that could potentially be prevented or treated early to avoid hospitalization, according to researchers.
Hallie C. Prescott, MD, of the University of Michigan, Ann Arbor, and her colleagues reported these findings in JAMA.
The researchers analyzed participants in the nationally representative US Health and Retirement Study, a sample of households with adults 50 years of age or older that is linked to Medicare claims (1998-2010).
The team examined the most common readmission diagnoses among patients who were hospitalized for severe sepsis, the extent to which readmissions might have been preventable, and whether the pattern of readmission diagnoses differed compared with that of other acute medical conditions.
To gauge what proportion of rehospitalizations might have been preventable, the researchers looked at ambulatory-care-sensitive conditions (ACSCs) identified by the Agency for Healthcare Research and Quality. They also expanded the definition of ACSCs to include conditions that aren’t common among the general population but arise more often in sepsis survivors.
So their potentially preventable readmission diagnoses included pneumonia, hypertension, dehydration, asthma, urinary tract infection, chronic obstructive pulmonary disease exacerbation, perforated appendix, diabetes, angina, congestive heart failure, sepsis, acute renal failure, skin or soft tissue infection, and aspiration pneumonitis.
Dr Prescott and her colleagues identified 2617 hospitalizations for severe sepsis that could be matched to hospitalizations for other acute medical conditions. And they found that 1115 of the severe sepsis survivors (42.6%) were rehospitalized within 90 days.
The 10 most common readmission diagnoses following severe sepsis were sepsis (6.4%), congestive heart failure (5.5%), pneumonia (3.5%), acute renal failure (3.3%), rehabilitation (2.8%), respiratory failure (2.5%), complication related to a device, implant, or graft (2%), exacerbation of chronic obstructive pulmonary disorder (1.9%), aspiration pneumonitis (1.8%), and urinary tract infection (1.7%).
Readmissions for a primary diagnosis of infection (sepsis, pneumonia, urinary tract, and skin or soft tissue infection) occurred in 11.9% of severe sepsis survivors and 8.0% of patients with acute medical conditions (P<0.001).
Likewise, readmissions for ACSCs were more common after severe sepsis than for patients with acute conditions—21.6% and 19.1%, respectively (P=0.02)—and accounted for a greater proportion of all 90-day readmissions—41.6% and 37%.1, respectively (P=0.009).
“Many of these conditions can be managed if the patient can get in to see a doctor at the start of the illness, meaning that we potentially avoid hospitalization,” Dr Prescott said. “We need to assess their vulnerability and design a better landing pad for patients when they leave the hospital, and avoid the second hit that derails recovery.” ![]()
ASH advocates use of systems-based hematologists

Photo courtesy of CDC
The American Society of Hematology (ASH) has released a report proposing a new role for hematologists specializing in non-malignant blood disorders.
ASH partnered with the healthcare consulting firm The Lewin Group to identify emerging career opportunities for health system- and hospital-based hematologists and to provide guidance on pursuing those opportunities.
The resulting report, published in Blood, outlines a few models for a systems-based clinical hematologist.
The report’s authors noted that demand for hematology expertise remains high nationwide. However, ASH and its members are concerned that changes to academic training will hinder both the recruitment of new talent to the field and the retention of seasoned experts.
The authors said that today’s hematology trainees are unlikely to receive the same non-malignant training as many “classic” hematologists trained in prior decades. And training shortfalls are further compounded by the fact that primary care physicians do not have the expertise to manage common blood disorders, which increases referrals to hematologists.
This results in higher demand for a smaller pool of hematologists entering the field with adequate training to effectively and efficiently manage non-malignant disorders.
“Given the rapid evolution and complexity of the field, the time is appropriate to identify career pathways that attract and enable physicians to practice non-malignant hematology in a sustainable manner,” said author Janis L. Abkowitz, MD, of the University of Washington in Seattle.
She and her colleagues noted that, in response to these challenges, US hematologists are defining new paths and assuming more centralized positions in large and small healthcare systems.
These systems-based hematologists are specialty-trained physicians—employed by a hospital, medical center, or health system—who optimize individual patient care as well as the overall system of healthcare delivery for patients with blood disorders.
For example, a systems-based hematologist could work closely with surgeons to minimize perioperative bleeding and could manage care pathways for patients with chronic blood diseases.
The report offered 4 examples where the involvement of a systems-based hematologist would lead to cost-effective decision-making. These were based upon interviews with 14 early adoptors of the systems-based approach to hematology.
The first example was heparin-induced thrombocytopenia (HIT). A systems-based hematologist could implement care pathways that focus on HIT by working to reduce unnecessary heparin exposure, optimizing laboratory testing for suspected HIT, and reducing unnecessary procedures in patients.
The second example was thrombotic thrombocytopenic purpura (TTP). A systems-based hematologist could optimize testing for TTP, which may reduce system-wide plasma use.
The third example was a medical director for hemostasis and thrombosis. A systems-based hematologist could foster appropriate and safe practices, including the implementation of and adherence to preventive care for thrombotic events and the optimal use of anticoagulant medications.
The fourth example was non-malignant hematology consultation in an accountable care organization (ACO) environment. The authors noted that ACOs have enabled more patients to be served by a health system, but there are fewer incentives for physicians to manage common hematology-related issues. A funded systems-based hematologist could ensure that patients have more timely access to hematology consultations.
“A systems-based hematologist position presents a unique opportunity for hematologists to design new models for care delivery and demonstrate their ability to improve clinical outcomes while maintaining or reducing costs,” Dr Abkowitz said. “Just as blood must flow throughout the body, the expertise of hematology must flow throughout the healthcare system.”
As a next step, ASH has invited its members to share practice models they have developed and examples of how they have collaborated with others to improve healthcare outcomes, reduce complications, and eliminate unnecessary spending. ![]()

Photo courtesy of CDC
The American Society of Hematology (ASH) has released a report proposing a new role for hematologists specializing in non-malignant blood disorders.
ASH partnered with the healthcare consulting firm The Lewin Group to identify emerging career opportunities for health system- and hospital-based hematologists and to provide guidance on pursuing those opportunities.
The resulting report, published in Blood, outlines a few models for a systems-based clinical hematologist.
The report’s authors noted that demand for hematology expertise remains high nationwide. However, ASH and its members are concerned that changes to academic training will hinder both the recruitment of new talent to the field and the retention of seasoned experts.
The authors said that today’s hematology trainees are unlikely to receive the same non-malignant training as many “classic” hematologists trained in prior decades. And training shortfalls are further compounded by the fact that primary care physicians do not have the expertise to manage common blood disorders, which increases referrals to hematologists.
This results in higher demand for a smaller pool of hematologists entering the field with adequate training to effectively and efficiently manage non-malignant disorders.
“Given the rapid evolution and complexity of the field, the time is appropriate to identify career pathways that attract and enable physicians to practice non-malignant hematology in a sustainable manner,” said author Janis L. Abkowitz, MD, of the University of Washington in Seattle.
She and her colleagues noted that, in response to these challenges, US hematologists are defining new paths and assuming more centralized positions in large and small healthcare systems.
These systems-based hematologists are specialty-trained physicians—employed by a hospital, medical center, or health system—who optimize individual patient care as well as the overall system of healthcare delivery for patients with blood disorders.
For example, a systems-based hematologist could work closely with surgeons to minimize perioperative bleeding and could manage care pathways for patients with chronic blood diseases.
The report offered 4 examples where the involvement of a systems-based hematologist would lead to cost-effective decision-making. These were based upon interviews with 14 early adoptors of the systems-based approach to hematology.
The first example was heparin-induced thrombocytopenia (HIT). A systems-based hematologist could implement care pathways that focus on HIT by working to reduce unnecessary heparin exposure, optimizing laboratory testing for suspected HIT, and reducing unnecessary procedures in patients.
The second example was thrombotic thrombocytopenic purpura (TTP). A systems-based hematologist could optimize testing for TTP, which may reduce system-wide plasma use.
The third example was a medical director for hemostasis and thrombosis. A systems-based hematologist could foster appropriate and safe practices, including the implementation of and adherence to preventive care for thrombotic events and the optimal use of anticoagulant medications.
The fourth example was non-malignant hematology consultation in an accountable care organization (ACO) environment. The authors noted that ACOs have enabled more patients to be served by a health system, but there are fewer incentives for physicians to manage common hematology-related issues. A funded systems-based hematologist could ensure that patients have more timely access to hematology consultations.
“A systems-based hematologist position presents a unique opportunity for hematologists to design new models for care delivery and demonstrate their ability to improve clinical outcomes while maintaining or reducing costs,” Dr Abkowitz said. “Just as blood must flow throughout the body, the expertise of hematology must flow throughout the healthcare system.”
As a next step, ASH has invited its members to share practice models they have developed and examples of how they have collaborated with others to improve healthcare outcomes, reduce complications, and eliminate unnecessary spending. ![]()

Photo courtesy of CDC
The American Society of Hematology (ASH) has released a report proposing a new role for hematologists specializing in non-malignant blood disorders.
ASH partnered with the healthcare consulting firm The Lewin Group to identify emerging career opportunities for health system- and hospital-based hematologists and to provide guidance on pursuing those opportunities.
The resulting report, published in Blood, outlines a few models for a systems-based clinical hematologist.
The report’s authors noted that demand for hematology expertise remains high nationwide. However, ASH and its members are concerned that changes to academic training will hinder both the recruitment of new talent to the field and the retention of seasoned experts.
The authors said that today’s hematology trainees are unlikely to receive the same non-malignant training as many “classic” hematologists trained in prior decades. And training shortfalls are further compounded by the fact that primary care physicians do not have the expertise to manage common blood disorders, which increases referrals to hematologists.
This results in higher demand for a smaller pool of hematologists entering the field with adequate training to effectively and efficiently manage non-malignant disorders.
“Given the rapid evolution and complexity of the field, the time is appropriate to identify career pathways that attract and enable physicians to practice non-malignant hematology in a sustainable manner,” said author Janis L. Abkowitz, MD, of the University of Washington in Seattle.
She and her colleagues noted that, in response to these challenges, US hematologists are defining new paths and assuming more centralized positions in large and small healthcare systems.
These systems-based hematologists are specialty-trained physicians—employed by a hospital, medical center, or health system—who optimize individual patient care as well as the overall system of healthcare delivery for patients with blood disorders.
For example, a systems-based hematologist could work closely with surgeons to minimize perioperative bleeding and could manage care pathways for patients with chronic blood diseases.
The report offered 4 examples where the involvement of a systems-based hematologist would lead to cost-effective decision-making. These were based upon interviews with 14 early adoptors of the systems-based approach to hematology.
The first example was heparin-induced thrombocytopenia (HIT). A systems-based hematologist could implement care pathways that focus on HIT by working to reduce unnecessary heparin exposure, optimizing laboratory testing for suspected HIT, and reducing unnecessary procedures in patients.
The second example was thrombotic thrombocytopenic purpura (TTP). A systems-based hematologist could optimize testing for TTP, which may reduce system-wide plasma use.
The third example was a medical director for hemostasis and thrombosis. A systems-based hematologist could foster appropriate and safe practices, including the implementation of and adherence to preventive care for thrombotic events and the optimal use of anticoagulant medications.
The fourth example was non-malignant hematology consultation in an accountable care organization (ACO) environment. The authors noted that ACOs have enabled more patients to be served by a health system, but there are fewer incentives for physicians to manage common hematology-related issues. A funded systems-based hematologist could ensure that patients have more timely access to hematology consultations.
“A systems-based hematologist position presents a unique opportunity for hematologists to design new models for care delivery and demonstrate their ability to improve clinical outcomes while maintaining or reducing costs,” Dr Abkowitz said. “Just as blood must flow throughout the body, the expertise of hematology must flow throughout the healthcare system.”
As a next step, ASH has invited its members to share practice models they have developed and examples of how they have collaborated with others to improve healthcare outcomes, reduce complications, and eliminate unnecessary spending.
New test can better predict cytokine storm, team says

Photo by Juan D. Alfonso
Scientists have developed a test that uses cells from a single donor’s blood to predict whether a new drug will cause a cytokine storm.
The group says this is an improvement over current tests, which use endothelial cells and peripheral blood mononuclear cells (PBMCs) from two separate donors and can therefore produce inaccurate results.
Furthermore, current tests cannot differentiate drugs that induce a mild cytokine storm from those that induce a severe one, but the new test can.
Jane Mitchell, PhD, of the National Heart and Lung Institute at Imperial College London in the UK, and her colleagues described the new test in The FASEB Journal.
Current tests for cytokine storm reactions use endothelial cells taken from the vessels of one donor and PBMCs from a different donor because endothelial cells are normally only grown from tissue removed in surgery or post-mortem, or from umbilical vessels after birth.
When cells from two different donors are used, one may have an immune reaction to the other. And this can result in the test falsely showing a severe immune reaction to a drug that is safe.
Dr Mitchell and her colleagues say they have solved this problem by isolating stem cells from the blood of a volunteer and using them to grow endothelial cells in a dish. The team then added PBMCs to the donor’s own endothelial cells to recreate the unique conditions found in their blood vessels.
When the scientists added the immunomodulatory drug TGN1412, the mixture of cells released a cytokine storm, as would happen inside the human body.
Responses to other drugs were consistent with those observed in humans as well. There was a modest response to alemtuzumab (Campath) and no response to the control antibodies trastuzumab (Herceptin), bevacizumab (Avastin), and ofatumumab (Arzerra).
“As biological therapies become more mainstream, it’s more likely that drugs being tested on humans for the first time will have unexpected and potentially catastrophic effects,” Dr Mitchell said.
“We’ve used adult stem cell technology to develop a laboratory test that could prevent another disaster like the TGN1412 trial [in which 6 healthy young men developed multi-organ failure]. Drug companies have the technical capacity to start using this test now, but we’re working on developing an off-the-shelf kit, which will make it easy to use on a large scale.”
The team has collaborated with the National Institute for Biological Standards and Control to validate the test and are now working with the clinical trials company Quintiles to develop the technology further.

Photo by Juan D. Alfonso
Scientists have developed a test that uses cells from a single donor’s blood to predict whether a new drug will cause a cytokine storm.
The group says this is an improvement over current tests, which use endothelial cells and peripheral blood mononuclear cells (PBMCs) from two separate donors and can therefore produce inaccurate results.
Furthermore, current tests cannot differentiate drugs that induce a mild cytokine storm from those that induce a severe one, but the new test can.
Jane Mitchell, PhD, of the National Heart and Lung Institute at Imperial College London in the UK, and her colleagues described the new test in The FASEB Journal.
Current tests for cytokine storm reactions use endothelial cells taken from the vessels of one donor and PBMCs from a different donor because endothelial cells are normally only grown from tissue removed in surgery or post-mortem, or from umbilical vessels after birth.
When cells from two different donors are used, one may have an immune reaction to the other. And this can result in the test falsely showing a severe immune reaction to a drug that is safe.
Dr Mitchell and her colleagues say they have solved this problem by isolating stem cells from the blood of a volunteer and using them to grow endothelial cells in a dish. The team then added PBMCs to the donor’s own endothelial cells to recreate the unique conditions found in their blood vessels.
When the scientists added the immunomodulatory drug TGN1412, the mixture of cells released a cytokine storm, as would happen inside the human body.
Responses to other drugs were consistent with those observed in humans as well. There was a modest response to alemtuzumab (Campath) and no response to the control antibodies trastuzumab (Herceptin), bevacizumab (Avastin), and ofatumumab (Arzerra).
“As biological therapies become more mainstream, it’s more likely that drugs being tested on humans for the first time will have unexpected and potentially catastrophic effects,” Dr Mitchell said.
“We’ve used adult stem cell technology to develop a laboratory test that could prevent another disaster like the TGN1412 trial [in which 6 healthy young men developed multi-organ failure]. Drug companies have the technical capacity to start using this test now, but we’re working on developing an off-the-shelf kit, which will make it easy to use on a large scale.”
The team has collaborated with the National Institute for Biological Standards and Control to validate the test and are now working with the clinical trials company Quintiles to develop the technology further.

Photo by Juan D. Alfonso
Scientists have developed a test that uses cells from a single donor’s blood to predict whether a new drug will cause a cytokine storm.
The group says this is an improvement over current tests, which use endothelial cells and peripheral blood mononuclear cells (PBMCs) from two separate donors and can therefore produce inaccurate results.
Furthermore, current tests cannot differentiate drugs that induce a mild cytokine storm from those that induce a severe one, but the new test can.
Jane Mitchell, PhD, of the National Heart and Lung Institute at Imperial College London in the UK, and her colleagues described the new test in The FASEB Journal.
Current tests for cytokine storm reactions use endothelial cells taken from the vessels of one donor and PBMCs from a different donor because endothelial cells are normally only grown from tissue removed in surgery or post-mortem, or from umbilical vessels after birth.
When cells from two different donors are used, one may have an immune reaction to the other. And this can result in the test falsely showing a severe immune reaction to a drug that is safe.
Dr Mitchell and her colleagues say they have solved this problem by isolating stem cells from the blood of a volunteer and using them to grow endothelial cells in a dish. The team then added PBMCs to the donor’s own endothelial cells to recreate the unique conditions found in their blood vessels.
When the scientists added the immunomodulatory drug TGN1412, the mixture of cells released a cytokine storm, as would happen inside the human body.
Responses to other drugs were consistent with those observed in humans as well. There was a modest response to alemtuzumab (Campath) and no response to the control antibodies trastuzumab (Herceptin), bevacizumab (Avastin), and ofatumumab (Arzerra).
“As biological therapies become more mainstream, it’s more likely that drugs being tested on humans for the first time will have unexpected and potentially catastrophic effects,” Dr Mitchell said.
“We’ve used adult stem cell technology to develop a laboratory test that could prevent another disaster like the TGN1412 trial [in which 6 healthy young men developed multi-organ failure]. Drug companies have the technical capacity to start using this test now, but we’re working on developing an off-the-shelf kit, which will make it easy to use on a large scale.”
The team has collaborated with the National Institute for Biological Standards and Control to validate the test and are now working with the clinical trials company Quintiles to develop the technology further.
FDA approves new antifungal drug

The US Food and Drug Administration (FDA) has approved isavuconazonium sulfate (Cresemba) to treat adults with invasive aspergillosis and invasive mucormycosis, life-threatening fungal infections that predominantly occur in immunocompromised patients.
Isavuconazonium sulfate is an azole antifungal agent that works by targeting the cell wall of a fungus. The drug is available in oral and intravenous formulations.
“[The] approval provides a new treatment option for patients with serious fungal infections and underscores the importance of having available safe and effective antifungal drugs,” said Edward Cox, MD, director of the Office of Antimicrobial Products in the FDA’s Center for Drug Evaluation and Research.
Clinical trials
The FDA approved isavuconazonium sulfate to treat invasive aspergillosis based on results of the phase 3 SECURE trial. The study included 516 adults with invasive aspergillosis who were randomized to receive isavuconazonium sulfate or voriconazole.
Isavuconazonium sulfate demonstrated non-inferiority to voriconazole on the primary endpoint of all-cause mortality. All-cause mortality through day 42 was 18.6% in the isavuconazonium sulfate arm and 20.2% in the voriconazole arm.
In addition, isavuconazonium sulfate demonstrated similar rates of mortality and non-fatal adverse events as voriconazole
The FDA approved isavuconazonium sulfate to treat invasive mucormycosis based on results of the phase 3 VITAL trial. This single-arm study included 37 patients with invasive mucormycosis who received isavuconazonium sulfate.
All-cause mortality in these patients was 38%. The efficacy of isavuconazonium sulfate as a treatment for invasive mucormycosis has not been evaluated in concurrent, controlled clinical trials.
The most frequent adverse events for patients treated with isavuconazonium sulfate in clinical trials were nausea (26%), vomiting (25%), diarrhea (22%), headache (17%), elevated liver chemistry tests (17%), hypokalemia (14%), constipation (13%), dyspnea (12%), cough (12%), peripheral edema (11%), and back pain (10%).
QIDP status
Isavuconazonium sulfate is the sixth approved antifungal/antibacterial drug product designated as a qualified infectious disease product (QIDP). This designation is given to antibacterial or antifungal products that treat serious or life-threatening infections.
As part of its QIDP designation, isavuconazonium sulfate was given priority review. The QIDP designation also qualifies the drug for an additional 5 years of marketing exclusivity to be added to certain exclusivity periods already provided by the Food, Drug, and Cosmetic Act.
As invasive aspergillosis and mucormycosis are rare, the FDA also granted isavuconazonium sulfate orphan drug designations to treat these infections.
Isavuconazonium sulfate is marketed as Cresemba by Astellas Pharma US, Inc., which is based in Northbrook, Illinois. For more information on the drug, see the full prescribing information.

The US Food and Drug Administration (FDA) has approved isavuconazonium sulfate (Cresemba) to treat adults with invasive aspergillosis and invasive mucormycosis, life-threatening fungal infections that predominantly occur in immunocompromised patients.
Isavuconazonium sulfate is an azole antifungal agent that works by targeting the cell wall of a fungus. The drug is available in oral and intravenous formulations.
“[The] approval provides a new treatment option for patients with serious fungal infections and underscores the importance of having available safe and effective antifungal drugs,” said Edward Cox, MD, director of the Office of Antimicrobial Products in the FDA’s Center for Drug Evaluation and Research.
Clinical trials
The FDA approved isavuconazonium sulfate to treat invasive aspergillosis based on results of the phase 3 SECURE trial. The study included 516 adults with invasive aspergillosis who were randomized to receive isavuconazonium sulfate or voriconazole.
Isavuconazonium sulfate demonstrated non-inferiority to voriconazole on the primary endpoint of all-cause mortality. All-cause mortality through day 42 was 18.6% in the isavuconazonium sulfate arm and 20.2% in the voriconazole arm.
In addition, isavuconazonium sulfate demonstrated similar rates of mortality and non-fatal adverse events as voriconazole
The FDA approved isavuconazonium sulfate to treat invasive mucormycosis based on results of the phase 3 VITAL trial. This single-arm study included 37 patients with invasive mucormycosis who received isavuconazonium sulfate.
All-cause mortality in these patients was 38%. The efficacy of isavuconazonium sulfate as a treatment for invasive mucormycosis has not been evaluated in concurrent, controlled clinical trials.
The most frequent adverse events for patients treated with isavuconazonium sulfate in clinical trials were nausea (26%), vomiting (25%), diarrhea (22%), headache (17%), elevated liver chemistry tests (17%), hypokalemia (14%), constipation (13%), dyspnea (12%), cough (12%), peripheral edema (11%), and back pain (10%).
QIDP status
Isavuconazonium sulfate is the sixth approved antifungal/antibacterial drug product designated as a qualified infectious disease product (QIDP). This designation is given to antibacterial or antifungal products that treat serious or life-threatening infections.
As part of its QIDP designation, isavuconazonium sulfate was given priority review. The QIDP designation also qualifies the drug for an additional 5 years of marketing exclusivity to be added to certain exclusivity periods already provided by the Food, Drug, and Cosmetic Act.
As invasive aspergillosis and mucormycosis are rare, the FDA also granted isavuconazonium sulfate orphan drug designations to treat these infections.
Isavuconazonium sulfate is marketed as Cresemba by Astellas Pharma US, Inc., which is based in Northbrook, Illinois. For more information on the drug, see the full prescribing information.

The US Food and Drug Administration (FDA) has approved isavuconazonium sulfate (Cresemba) to treat adults with invasive aspergillosis and invasive mucormycosis, life-threatening fungal infections that predominantly occur in immunocompromised patients.
Isavuconazonium sulfate is an azole antifungal agent that works by targeting the cell wall of a fungus. The drug is available in oral and intravenous formulations.
“[The] approval provides a new treatment option for patients with serious fungal infections and underscores the importance of having available safe and effective antifungal drugs,” said Edward Cox, MD, director of the Office of Antimicrobial Products in the FDA’s Center for Drug Evaluation and Research.
Clinical trials
The FDA approved isavuconazonium sulfate to treat invasive aspergillosis based on results of the phase 3 SECURE trial. The study included 516 adults with invasive aspergillosis who were randomized to receive isavuconazonium sulfate or voriconazole.
Isavuconazonium sulfate demonstrated non-inferiority to voriconazole on the primary endpoint of all-cause mortality. All-cause mortality through day 42 was 18.6% in the isavuconazonium sulfate arm and 20.2% in the voriconazole arm.
In addition, isavuconazonium sulfate demonstrated similar rates of mortality and non-fatal adverse events as voriconazole
The FDA approved isavuconazonium sulfate to treat invasive mucormycosis based on results of the phase 3 VITAL trial. This single-arm study included 37 patients with invasive mucormycosis who received isavuconazonium sulfate.
All-cause mortality in these patients was 38%. The efficacy of isavuconazonium sulfate as a treatment for invasive mucormycosis has not been evaluated in concurrent, controlled clinical trials.
The most frequent adverse events for patients treated with isavuconazonium sulfate in clinical trials were nausea (26%), vomiting (25%), diarrhea (22%), headache (17%), elevated liver chemistry tests (17%), hypokalemia (14%), constipation (13%), dyspnea (12%), cough (12%), peripheral edema (11%), and back pain (10%).
QIDP status
Isavuconazonium sulfate is the sixth approved antifungal/antibacterial drug product designated as a qualified infectious disease product (QIDP). This designation is given to antibacterial or antifungal products that treat serious or life-threatening infections.
As part of its QIDP designation, isavuconazonium sulfate was given priority review. The QIDP designation also qualifies the drug for an additional 5 years of marketing exclusivity to be added to certain exclusivity periods already provided by the Food, Drug, and Cosmetic Act.
As invasive aspergillosis and mucormycosis are rare, the FDA also granted isavuconazonium sulfate orphan drug designations to treat these infections.
Isavuconazonium sulfate is marketed as Cresemba by Astellas Pharma US, Inc., which is based in Northbrook, Illinois. For more information on the drug, see the full prescribing information.
Studies help explain multidrug resistance in cancer

(right) and Sung Chang Lee
Photo courtesy of
The Scripps Research Institute
Scientists have discovered how the primary protein responsible for multidrug chemotherapy resistance changes shape and reacts to drugs.
They believe this information will aid the design of better molecules to inhibit or evade multidrug resistance.
The researchers noted that the proteins at work in multidrug resistance are ABC transporters. An important ABC transporter, P-glycoprotein (P-gp), catches harmful toxins in a “binding pocket” and expels them from cells.
The problem is that, in cancer patients, P-gp sometimes begins recognizing chemotherapy drugs and expelling them too. Over time, more and more cancer cells can develop multidrug resistance, eliminating all possible treatments.
“Virtually all cancer deaths can be attributed to the failure of chemotherapy,” said study author Qinghai Zhang, PhD, of The Scripps Research Institute in La Jolla, California.
He and his colleagues theorized that scientists might be able to design more effective cancer drugs if they had a better understanding of P-gp and how it binds to molecules.
A better look at transporters
For their first study, published in Structure, the researchers looked at P-gp and MsbA, a similar transporter protein found in bacteria, under an electron microscope. This helped them solve a major problem in transporter research.
Until recently, scientists could only compare images of crystal structures made from transporter proteins. These crystallography images showed single snapshots of the transporter but didn’t show how the shape of the transporters could change.
Using electron microscopy, however, a whole range of different conformations of the structures could be visualized, essentially capturing P-gp and MsbA in action.
The research was also aided by the development of new chemical tools. The team used a solution of lipids and peptides to mimic natural conditions in the cell membrane. They used a novel chemical called beta-sheet peptide to stabilize the protein and provide enough stability for a new perspective.
Together with electron microscopy, this technique enabled the researchers to capture a series of images showing how transporter proteins change shape in response to drug and nucleotide binding. They found that transporter proteins have an open binding pocket that constantly switches to face different sides of membranes.
“The transporter goes through many steps,” Dr Zhang said. “It’s like a machine.”
A closer look at binding
In a second study, published in Acta Crystallographica Section D, the scientists investigated the drug binding sites of P-gp using higher-resolution X-ray crystallography. And they discovered how P-gp interacts with ligands.
The researchers studied crystals of the transporter bound to 4 different ligands to see how the transporters reacted. They found that when certain ligands bind to P-gp, they trigger local conformational changes in the transporter.
Binding also increased the rate of ATP hydrolysis, which provides mechanical energy and may be the first step in the process by which the binding pocket closes.
The team also discovered that ligands could bind to different areas of the transporter, leaving nearby slots open for other molecules. This suggests it may be difficult to completely halt the drug expulsion process.
Dr Zhang said the next step for this research is to develop molecules to evade P-gp binding.

(right) and Sung Chang Lee
Photo courtesy of
The Scripps Research Institute
Scientists have discovered how the primary protein responsible for multidrug chemotherapy resistance changes shape and reacts to drugs.
They believe this information will aid the design of better molecules to inhibit or evade multidrug resistance.
The researchers noted that the proteins at work in multidrug resistance are ABC transporters. An important ABC transporter, P-glycoprotein (P-gp), catches harmful toxins in a “binding pocket” and expels them from cells.
The problem is that, in cancer patients, P-gp sometimes begins recognizing chemotherapy drugs and expelling them too. Over time, more and more cancer cells can develop multidrug resistance, eliminating all possible treatments.
“Virtually all cancer deaths can be attributed to the failure of chemotherapy,” said study author Qinghai Zhang, PhD, of The Scripps Research Institute in La Jolla, California.
He and his colleagues theorized that scientists might be able to design more effective cancer drugs if they had a better understanding of P-gp and how it binds to molecules.
A better look at transporters
For their first study, published in Structure, the researchers looked at P-gp and MsbA, a similar transporter protein found in bacteria, under an electron microscope. This helped them solve a major problem in transporter research.
Until recently, scientists could only compare images of crystal structures made from transporter proteins. These crystallography images showed single snapshots of the transporter but didn’t show how the shape of the transporters could change.
Using electron microscopy, however, a whole range of different conformations of the structures could be visualized, essentially capturing P-gp and MsbA in action.
The research was also aided by the development of new chemical tools. The team used a solution of lipids and peptides to mimic natural conditions in the cell membrane. They used a novel chemical called beta-sheet peptide to stabilize the protein and provide enough stability for a new perspective.
Together with electron microscopy, this technique enabled the researchers to capture a series of images showing how transporter proteins change shape in response to drug and nucleotide binding. They found that transporter proteins have an open binding pocket that constantly switches to face different sides of membranes.
“The transporter goes through many steps,” Dr Zhang said. “It’s like a machine.”
A closer look at binding
In a second study, published in Acta Crystallographica Section D, the scientists investigated the drug binding sites of P-gp using higher-resolution X-ray crystallography. And they discovered how P-gp interacts with ligands.
The researchers studied crystals of the transporter bound to 4 different ligands to see how the transporters reacted. They found that when certain ligands bind to P-gp, they trigger local conformational changes in the transporter.
Binding also increased the rate of ATP hydrolysis, which provides mechanical energy and may be the first step in the process by which the binding pocket closes.
The team also discovered that ligands could bind to different areas of the transporter, leaving nearby slots open for other molecules. This suggests it may be difficult to completely halt the drug expulsion process.
Dr Zhang said the next step for this research is to develop molecules to evade P-gp binding.

(right) and Sung Chang Lee
Photo courtesy of
The Scripps Research Institute
Scientists have discovered how the primary protein responsible for multidrug chemotherapy resistance changes shape and reacts to drugs.
They believe this information will aid the design of better molecules to inhibit or evade multidrug resistance.
The researchers noted that the proteins at work in multidrug resistance are ABC transporters. An important ABC transporter, P-glycoprotein (P-gp), catches harmful toxins in a “binding pocket” and expels them from cells.
The problem is that, in cancer patients, P-gp sometimes begins recognizing chemotherapy drugs and expelling them too. Over time, more and more cancer cells can develop multidrug resistance, eliminating all possible treatments.
“Virtually all cancer deaths can be attributed to the failure of chemotherapy,” said study author Qinghai Zhang, PhD, of The Scripps Research Institute in La Jolla, California.
He and his colleagues theorized that scientists might be able to design more effective cancer drugs if they had a better understanding of P-gp and how it binds to molecules.
A better look at transporters
For their first study, published in Structure, the researchers looked at P-gp and MsbA, a similar transporter protein found in bacteria, under an electron microscope. This helped them solve a major problem in transporter research.
Until recently, scientists could only compare images of crystal structures made from transporter proteins. These crystallography images showed single snapshots of the transporter but didn’t show how the shape of the transporters could change.
Using electron microscopy, however, a whole range of different conformations of the structures could be visualized, essentially capturing P-gp and MsbA in action.
The research was also aided by the development of new chemical tools. The team used a solution of lipids and peptides to mimic natural conditions in the cell membrane. They used a novel chemical called beta-sheet peptide to stabilize the protein and provide enough stability for a new perspective.
Together with electron microscopy, this technique enabled the researchers to capture a series of images showing how transporter proteins change shape in response to drug and nucleotide binding. They found that transporter proteins have an open binding pocket that constantly switches to face different sides of membranes.
“The transporter goes through many steps,” Dr Zhang said. “It’s like a machine.”
A closer look at binding
In a second study, published in Acta Crystallographica Section D, the scientists investigated the drug binding sites of P-gp using higher-resolution X-ray crystallography. And they discovered how P-gp interacts with ligands.
The researchers studied crystals of the transporter bound to 4 different ligands to see how the transporters reacted. They found that when certain ligands bind to P-gp, they trigger local conformational changes in the transporter.
Binding also increased the rate of ATP hydrolysis, which provides mechanical energy and may be the first step in the process by which the binding pocket closes.
The team also discovered that ligands could bind to different areas of the transporter, leaving nearby slots open for other molecules. This suggests it may be difficult to completely halt the drug expulsion process.
Dr Zhang said the next step for this research is to develop molecules to evade P-gp binding.