User login
Elevated risk for cancer in RA patients treated with DMARD
Key clinical point: Compared with the general population, the risk for cancer was significantly higher in patients with rheumatoid arthritis (RA) treated with disease-modifying antirheumatic drugs (DMARD).
Major finding: The all-cancer risk was 20% higher in patients with RA than in the general population (standardized incidence ratio [SIR] 1.20; 95% CI 1.17-1.23). The risk was particularly higher for solid cancers like bladder cancer (SIR 2.38; 95% CI 2.25-2.51), cervical cancer (SIR 1.80; 95% CI 1.62-2.01), and lung cancer (SIR 1.41; 95% CI 1.36-1.46) and for hematological malignancies like Hodgkin’s lymphoma (SIR 2.73; 95% CI 2.31-3.23).
Study details: This population-based observational study included 257,074 patients with RA and without a history of cancer, who received DMARD and were compared with the general population.
Disclosures: This study was supported by unrestricted grants from the French National Cancer Institute and the Assistance Publique des Hôpitaux de Paris. Four authors declared receiving research grants, consulting fees, or support for meetings or travel from various sources.
Source: Beydon M et al. Risk of cancer for patients with rheumatoid arthritis versus general population: A national claims database cohort study. Lancet Reg Health Eur. 2023;35:100768 (Oct 29). doi: 10.1016/j.lanepe.2023.100768
Key clinical point: Compared with the general population, the risk for cancer was significantly higher in patients with rheumatoid arthritis (RA) treated with disease-modifying antirheumatic drugs (DMARD).
Major finding: The all-cancer risk was 20% higher in patients with RA than in the general population (standardized incidence ratio [SIR] 1.20; 95% CI 1.17-1.23). The risk was particularly higher for solid cancers like bladder cancer (SIR 2.38; 95% CI 2.25-2.51), cervical cancer (SIR 1.80; 95% CI 1.62-2.01), and lung cancer (SIR 1.41; 95% CI 1.36-1.46) and for hematological malignancies like Hodgkin’s lymphoma (SIR 2.73; 95% CI 2.31-3.23).
Study details: This population-based observational study included 257,074 patients with RA and without a history of cancer, who received DMARD and were compared with the general population.
Disclosures: This study was supported by unrestricted grants from the French National Cancer Institute and the Assistance Publique des Hôpitaux de Paris. Four authors declared receiving research grants, consulting fees, or support for meetings or travel from various sources.
Source: Beydon M et al. Risk of cancer for patients with rheumatoid arthritis versus general population: A national claims database cohort study. Lancet Reg Health Eur. 2023;35:100768 (Oct 29). doi: 10.1016/j.lanepe.2023.100768
Key clinical point: Compared with the general population, the risk for cancer was significantly higher in patients with rheumatoid arthritis (RA) treated with disease-modifying antirheumatic drugs (DMARD).
Major finding: The all-cancer risk was 20% higher in patients with RA than in the general population (standardized incidence ratio [SIR] 1.20; 95% CI 1.17-1.23). The risk was particularly higher for solid cancers like bladder cancer (SIR 2.38; 95% CI 2.25-2.51), cervical cancer (SIR 1.80; 95% CI 1.62-2.01), and lung cancer (SIR 1.41; 95% CI 1.36-1.46) and for hematological malignancies like Hodgkin’s lymphoma (SIR 2.73; 95% CI 2.31-3.23).
Study details: This population-based observational study included 257,074 patients with RA and without a history of cancer, who received DMARD and were compared with the general population.
Disclosures: This study was supported by unrestricted grants from the French National Cancer Institute and the Assistance Publique des Hôpitaux de Paris. Four authors declared receiving research grants, consulting fees, or support for meetings or travel from various sources.
Source: Beydon M et al. Risk of cancer for patients with rheumatoid arthritis versus general population: A national claims database cohort study. Lancet Reg Health Eur. 2023;35:100768 (Oct 29). doi: 10.1016/j.lanepe.2023.100768
Real-world study confirms efficacy of all JAK inhibitors in RA
Key clinical point: Janus kinase inhibitors (JAKi) like tofacitinib, baricitinib, peficitinib, and upadacitinib showed comparable efficacy, and all of these drugs had impressive remission rates in patients with rheumatoid arthritis (RA).
Major finding: At 6 months of treatment with JAKi, the average Clinical Disease Activity Index (CDAI) scores improved significantly (P < .001), with 1 out of 3 patients achieving CDAI-remission and ≥82% of patients achieving CDAI-low disease activity (LDA). The achievement rates of CDAI-remission and CDAI-LDA were comparable across the four treatment groups.
Study details: Findings are from analysis of a retrospective study including 361 patients with RA from the ANSWER cohort who received tofacitinib (n = 127), baricitinib (n = 153), peficitinib (n = 29), or upadacitinib (n = 52).
Disclosures: This study did not receive any specific funding. Some authors declared receiving research grants, payments for lectures, or speaking or consulting fees from various sources.
Source: Hayashi S et al. Real-world comparative study of the efficacy of Janus kinase inhibitors in patients with rheumatoid arthritis: The ANSWER cohort study. Rheumatology (Oxford). 2023 (Nov 1). doi: 10.1093/rheumatology/kead543
Key clinical point: Janus kinase inhibitors (JAKi) like tofacitinib, baricitinib, peficitinib, and upadacitinib showed comparable efficacy, and all of these drugs had impressive remission rates in patients with rheumatoid arthritis (RA).
Major finding: At 6 months of treatment with JAKi, the average Clinical Disease Activity Index (CDAI) scores improved significantly (P < .001), with 1 out of 3 patients achieving CDAI-remission and ≥82% of patients achieving CDAI-low disease activity (LDA). The achievement rates of CDAI-remission and CDAI-LDA were comparable across the four treatment groups.
Study details: Findings are from analysis of a retrospective study including 361 patients with RA from the ANSWER cohort who received tofacitinib (n = 127), baricitinib (n = 153), peficitinib (n = 29), or upadacitinib (n = 52).
Disclosures: This study did not receive any specific funding. Some authors declared receiving research grants, payments for lectures, or speaking or consulting fees from various sources.
Source: Hayashi S et al. Real-world comparative study of the efficacy of Janus kinase inhibitors in patients with rheumatoid arthritis: The ANSWER cohort study. Rheumatology (Oxford). 2023 (Nov 1). doi: 10.1093/rheumatology/kead543
Key clinical point: Janus kinase inhibitors (JAKi) like tofacitinib, baricitinib, peficitinib, and upadacitinib showed comparable efficacy, and all of these drugs had impressive remission rates in patients with rheumatoid arthritis (RA).
Major finding: At 6 months of treatment with JAKi, the average Clinical Disease Activity Index (CDAI) scores improved significantly (P < .001), with 1 out of 3 patients achieving CDAI-remission and ≥82% of patients achieving CDAI-low disease activity (LDA). The achievement rates of CDAI-remission and CDAI-LDA were comparable across the four treatment groups.
Study details: Findings are from analysis of a retrospective study including 361 patients with RA from the ANSWER cohort who received tofacitinib (n = 127), baricitinib (n = 153), peficitinib (n = 29), or upadacitinib (n = 52).
Disclosures: This study did not receive any specific funding. Some authors declared receiving research grants, payments for lectures, or speaking or consulting fees from various sources.
Source: Hayashi S et al. Real-world comparative study of the efficacy of Janus kinase inhibitors in patients with rheumatoid arthritis: The ANSWER cohort study. Rheumatology (Oxford). 2023 (Nov 1). doi: 10.1093/rheumatology/kead543
Commentary: Examining DMARD Use in PsA, December 2023
Limiting radiographic progression is an important long-term goal of treatment of PsA. In a post hoc analysis that included 449 biologic-naive patients with PsA from the DISCOVER-2 trial who received 100 mg guselkumab every 4 or 8 weeks, Mease and colleagues demonstrated that a greater improvement in the Disease Activity Index for PsA (DAPSA) scores as early as week 8 and the achievement of DAPSA low disease activity at week 8 were associated with a significantly lower progression of radiographic joint damage (total PsA-modified van der Heijde-Sharp score) through week 100. Thus, patients who respond well early have better long-term outcomes.
The safety of targeted therapies is always of concern and is inadequately addressed by individual clinical trials. Meta-analyses may provide further insights. In a network meta-analysis of 94 randomized controlled trials that included a total of 54,369 patients with PsA or psoriasis who were treated with 14 biologics, five small molecules, or placebo, Chiu and colleagues found that for patients with psoriasis, infliximab, deucravacitinib, and bimekizumab had the highest risks for infection. In patients with PsA, bimekizumab, apremilast, and 30 mg upadacitinib led to a significantly higher risk for infection compared with placebo, and 30 mg upadacitinib also increasing the risk for serious infection compared with placebo. The risk for infection in patients with PsA did not increase with most bDMARD and targeted synthetic DMARD (tsDMARD), except bimekizumab, apremilast, and 30 mg upadacitinib.
There is increasing recognition of the difficulty in managing patients with refractory PsA. One approach to such difficult-to-treat disease is dual targeted therapy (DTT). However, the safety of these combinations is of major concern. There is currently an ongoing clinical trial comparing a combination of guselkumab and golimumab vs guselkumab alone for treatment-resistant PsA. In the meantime, Valero-Martinez and colleagues have reported results from an observational, retrospective, cross-sectional study that included patients with refractory PsA (n = 14) or spondyloarthritis (n = 22) who simultaneously received two bDMARD or tsDMARD with different therapeutic targets. The most commonly used combinations were a tumor necrosis factor (TNF) inhibitor plus an interleukin (IL)-12/23 pathway inhibitor, followed by a TNF inhibitor plus an IL-17 inhibitor. They found that at a median exposure of 14.86 months, the DTT retention rate in patients with PsA was 42.8%, with 40.0% and 53.3% of patients achieving remission or low activity and major clinical improvements, respectively. Treatment discontinuation due to adverse events was reported in one patient with PsA and multiple comorbidities. Thus, DTT led to satisfactory clinical improvements and no serious adverse events in patients with refractory PsA. The results of larger observational and randomized trials are awaited.
Limiting radiographic progression is an important long-term goal of treatment of PsA. In a post hoc analysis that included 449 biologic-naive patients with PsA from the DISCOVER-2 trial who received 100 mg guselkumab every 4 or 8 weeks, Mease and colleagues demonstrated that a greater improvement in the Disease Activity Index for PsA (DAPSA) scores as early as week 8 and the achievement of DAPSA low disease activity at week 8 were associated with a significantly lower progression of radiographic joint damage (total PsA-modified van der Heijde-Sharp score) through week 100. Thus, patients who respond well early have better long-term outcomes.
The safety of targeted therapies is always of concern and is inadequately addressed by individual clinical trials. Meta-analyses may provide further insights. In a network meta-analysis of 94 randomized controlled trials that included a total of 54,369 patients with PsA or psoriasis who were treated with 14 biologics, five small molecules, or placebo, Chiu and colleagues found that for patients with psoriasis, infliximab, deucravacitinib, and bimekizumab had the highest risks for infection. In patients with PsA, bimekizumab, apremilast, and 30 mg upadacitinib led to a significantly higher risk for infection compared with placebo, and 30 mg upadacitinib also increasing the risk for serious infection compared with placebo. The risk for infection in patients with PsA did not increase with most bDMARD and targeted synthetic DMARD (tsDMARD), except bimekizumab, apremilast, and 30 mg upadacitinib.
There is increasing recognition of the difficulty in managing patients with refractory PsA. One approach to such difficult-to-treat disease is dual targeted therapy (DTT). However, the safety of these combinations is of major concern. There is currently an ongoing clinical trial comparing a combination of guselkumab and golimumab vs guselkumab alone for treatment-resistant PsA. In the meantime, Valero-Martinez and colleagues have reported results from an observational, retrospective, cross-sectional study that included patients with refractory PsA (n = 14) or spondyloarthritis (n = 22) who simultaneously received two bDMARD or tsDMARD with different therapeutic targets. The most commonly used combinations were a tumor necrosis factor (TNF) inhibitor plus an interleukin (IL)-12/23 pathway inhibitor, followed by a TNF inhibitor plus an IL-17 inhibitor. They found that at a median exposure of 14.86 months, the DTT retention rate in patients with PsA was 42.8%, with 40.0% and 53.3% of patients achieving remission or low activity and major clinical improvements, respectively. Treatment discontinuation due to adverse events was reported in one patient with PsA and multiple comorbidities. Thus, DTT led to satisfactory clinical improvements and no serious adverse events in patients with refractory PsA. The results of larger observational and randomized trials are awaited.
Limiting radiographic progression is an important long-term goal of treatment of PsA. In a post hoc analysis that included 449 biologic-naive patients with PsA from the DISCOVER-2 trial who received 100 mg guselkumab every 4 or 8 weeks, Mease and colleagues demonstrated that a greater improvement in the Disease Activity Index for PsA (DAPSA) scores as early as week 8 and the achievement of DAPSA low disease activity at week 8 were associated with a significantly lower progression of radiographic joint damage (total PsA-modified van der Heijde-Sharp score) through week 100. Thus, patients who respond well early have better long-term outcomes.
The safety of targeted therapies is always of concern and is inadequately addressed by individual clinical trials. Meta-analyses may provide further insights. In a network meta-analysis of 94 randomized controlled trials that included a total of 54,369 patients with PsA or psoriasis who were treated with 14 biologics, five small molecules, or placebo, Chiu and colleagues found that for patients with psoriasis, infliximab, deucravacitinib, and bimekizumab had the highest risks for infection. In patients with PsA, bimekizumab, apremilast, and 30 mg upadacitinib led to a significantly higher risk for infection compared with placebo, and 30 mg upadacitinib also increasing the risk for serious infection compared with placebo. The risk for infection in patients with PsA did not increase with most bDMARD and targeted synthetic DMARD (tsDMARD), except bimekizumab, apremilast, and 30 mg upadacitinib.
There is increasing recognition of the difficulty in managing patients with refractory PsA. One approach to such difficult-to-treat disease is dual targeted therapy (DTT). However, the safety of these combinations is of major concern. There is currently an ongoing clinical trial comparing a combination of guselkumab and golimumab vs guselkumab alone for treatment-resistant PsA. In the meantime, Valero-Martinez and colleagues have reported results from an observational, retrospective, cross-sectional study that included patients with refractory PsA (n = 14) or spondyloarthritis (n = 22) who simultaneously received two bDMARD or tsDMARD with different therapeutic targets. The most commonly used combinations were a tumor necrosis factor (TNF) inhibitor plus an interleukin (IL)-12/23 pathway inhibitor, followed by a TNF inhibitor plus an IL-17 inhibitor. They found that at a median exposure of 14.86 months, the DTT retention rate in patients with PsA was 42.8%, with 40.0% and 53.3% of patients achieving remission or low activity and major clinical improvements, respectively. Treatment discontinuation due to adverse events was reported in one patient with PsA and multiple comorbidities. Thus, DTT led to satisfactory clinical improvements and no serious adverse events in patients with refractory PsA. The results of larger observational and randomized trials are awaited.
2023 USPSTF mammography age to start screening in average-risk patients: What’s new is old again
The US Preventive Services Task Force (USPSTF)1 is comprised of an independent panel of preventive services clinician experts who make evidence-based recommendations, with the letter grade assigned based on the strength of the evidence, from A through D (TABLE 1), on preventive services such as health screenings, shared decision making patient counseling, and preventive medications. Both A and B recommendations are generally accepted by both government and most private health insurance companies as a covered preventive benefit with no or minimal co-pays.

In 2002, the USPSTF released a Grade B recommendation that screening mammography for average-risk patients (with patients referring to persons assigned female at birth who have not undergone bilateral mastectomy) should take place starting at age 40 and be repeated every 1 to 2 years.2 This was consistent with or endorsed by most other national breast cancer screening guidelines, including the American College of Obstetricians and Gynecologists (ACOG), National Comprehensive Cancer Network (NCCN), the American Cancer Society (ACS), and the American College of Radiology.
In 2009, the USPSTF changed this Grade B recommendation, instead recommending biennial screening mammography for women aged 50 to 74.3 The most significant change in the revised guideline was for patients aged 40 to 49, where the recommendation was “against routine screening mammography.” They went on to say that the decision to start “biennial screening mammography before the age of 50 years should be an individual one and take patient context into account, including the patient’s values regarding specific benefits and harms.” Other prominent national guideline groups (ACOG, NCCN, ACS) did not agree with this recommendation and maintained that patients aged 40 to 49 should continue to be offered routine screening mammography either annually (NCCN, ACS) or at 1-to-2-year intervals (ACOG).4-6 The American College of Physicians and the American Academy of Family Practice endorsed the 2016 USPSTF guidelines, creating a disparity in breast cancer mammography counseling for averagerisk patients in their 40s.7
In 2016, the USPSTF revisited their breast cancer screening recommendation and renewed their 2009 recommendation against routine screening in patients aged 40 to 49, with the American College of Physicians and the American Academy of Family Practice again endorsing these guidelines.8 ACOG, ACS, NCCN, and ACR continued to recommend age 40 as a starting age for routine mammography screening (TABLE 2). As a result, over the past 14 years, patients aged 40 to 49 were placed in an awkward position of potentially hearing different recommendations from their health care providers, those differences often depending on the specialty of the provider they were seeing.
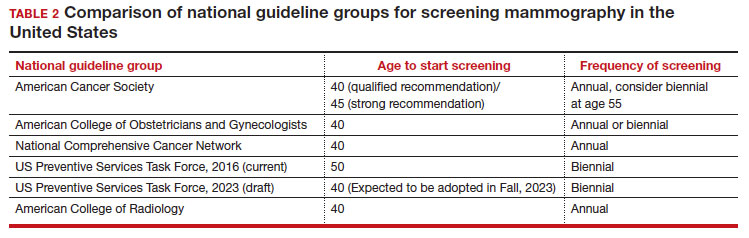
In 2023. On May 9, the USPSTF released a draft of their latest recommendation statement stating that all patients at average risk for breast cancer should get screened every other year beginning at age 40, bringing most of the national guideline groups into alignment with regard to age to start mammographic screening.9
- With an estimated more than 300,000 new cases in 2023, breast cancer has the highest incidence rate of any cancer in the United States
- The median age of patients with breast cancer in the United States is 58.0 years
- 1 in 5 new breast cancer diagnoses occur in patients between the ages of 40 and 49
- Despite lower incidence rates among Black vs White patients, Black patients have higher death rates from breast cancer
Why the change?
To answer this question, we need to examine the relevant epidemiology of breast cancer.
Continue to: Incidence...
Incidence
It is estimated that, in the United States in 2023, there will be 300,590 new cases of breast cancer, resulting in 43,700 deaths.10 From 2015–2019, there were 128.1 new breast cancer cases/100,000 population, which is the highest rate of cancer in the United States, regardless of sex.11 Diagnoses among patients aged 40 to 49 are rising at a faster rate than previously, about 2% per year between 2015 and 2019.
Racial and ethnic differences
In addition to the racial and ethnic epidemiologic differences in breast cancer, there are also disparities in breast cancer care and outcomes that need to be considered when making national guidelines/policy recommendations.
Black women have high mortality rates from breast cancer. While non-Hispanic White patients have the highest rates of breast cancer (TABLE 3), non-Hispanic Black patients have the highest rates of death due to breast cancer.10 There appear to be several reasons for the estimated 40%-higher rate of mortality among Black women, including:
- systemic racism in primary research, guidelines, and policy
- inequities in diagnostic follow-up and access to evidence-based cancer treatments
- biologic differences in breast cancer (ie, the incidence of triple-negative breast cancer (TNBC) is 2-fold higher in Black women compared with the other racial and ethnic groups in the United States).12-14
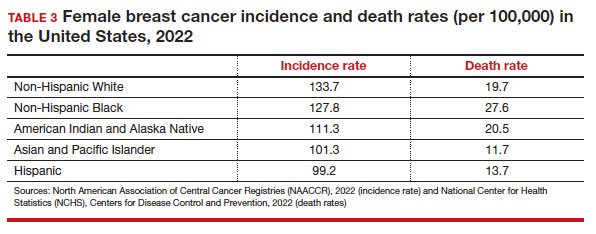
While prior studies have suggested that screening mammography might be less effective for patients with TNBC, a recent study demonstrated that patients who had mammography–screened-detected TNBC tumors were smaller and more likely to be node- negative compared with non-screened patients with TNBC.(14) Patients with screened-detected TNBCs were also more likely to undergo a lumpectomy instead of a mastectomy compared with non–screened detected TNBC (68.3% vs 46.1%; P = .002) (TABLE 4). These data strongly suggest that screening mammography is indeed effective in detecting TNBC at earlier stages, one of the best proxies for breast cancer mortality.
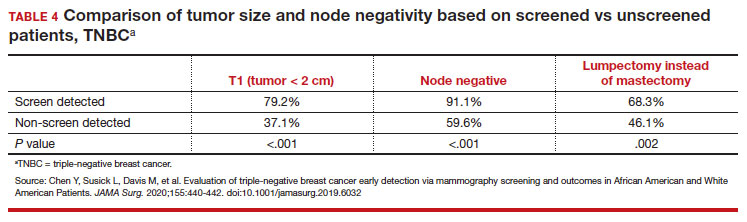
Non-White patients have higher incidence rates of breast cancer in their 40s. A second factor to consider in racial differences is the relatively higher incidence of breast cancer in Hispanic, Black, and Asian patients in their 40s compared with non-Hispanic White patients. In a recent analysis of data from 1973 to 2010 from the Surveillance, Epidemiology, and End Results (SEER) Program, the median age of patients with breast cancer in the United States was 58.0 years (interquartile range [IQR], 50.0–67.0 years).16 Across all US demographic populations by age at diagnosis, more than 20% of patients will have their initial diagnosis of breast cancer under the age of 50, and 1.55% (1 in 65) patients between ages 40 and 49 years will be diagnosed with breast cancer.4 However, among patients aged 50 and younger diagnosed with breast cancer, a significantly higher proportion are Black (31%), Hispanic (34.9%), or Asian (32.8%) versus White (23.1%) (P < .001 for all).16 So, for there to be similar racial and ethnic mammography capture rates with White patients, starting mammography screening ages would need to be lower for Black (age 47 years), Hispanic (and 46 years), and Asian (age 47 years) patients. Data from this study of the SEER database16 also demonstrated that more Black and Hispanic patients at age of diagnosis were diagnosed with advanced (regional or distant) breast cancer (46.6% and 42.9%, respectively) versus White or Asian patients (37.1% and 35.6%, respectively; P < .001 for all).
These findings led the authors of the study to conclude that the “Current [2016] USPSTF breast cancer screening recommendations do not reflect age-specific patterns based on race.” The USPSTF stated that this is one of the reasons why they reconsidered their stance on screening , and now recommend screening for all patients starting at age 40.
My current counseling approach
I encourage all racial and ethnic patients between the ages of 40 and 49 to undergo screening mammography because of the associated relative risk mortality reduction rates, which range from 15% to 50%. I also share that with my patients that, because of the younger average age of onset of breast cancer in Black, Hispanic, and Asian patients, they may derive additional benefit from screening starting at age 40.4
Impact of draft guidelines on breast cancer screening and mortality in younger patients
There is clear, unequivocal, and repeatable Level 1 evidence that screening mammography in the general population of patients aged 40 to 49 reduces breast cancer mortality. Breast cancer is the leading cause of cancer in the United States, the second leading cause of cancer mortality in patients, and 1 in 5 new breast cancer diagnoses occur in patients between the ages of 40 and 49. While recent efforts have been made to come to consensus on a screening starting age of 40 for patients at average risk for breast cancer, the USPSTF appeared to be an outlier with their 2016 recommendation to routinely start mammography screening at age 50 instead of 40.17
The USPSTF is a very important national voice in cancer prevention, and their 2023 (draft) revised guidelines to age 40 as the recommended starting screening age now agrees with the leading US guideline groups listed in Table 2. These guideline groups have gone through varying processes, and now have finally arrived at the same conclusion for age to start screening mammography in women of average risk. This agreement should come as a significant comfort to health care providers and patients alike. Changing the starting age to 40 years will result in thousands of lives and hundreds of thousands of life-years saved for patients aged 40 to 49. ●
- US Preventive Services Task Force website. Task Force at a glance. Accessed October 25, 2023. https://www.uspreventiveservicestaskforce.org /uspstf/about-uspstf/task-force-at-a-glance
- Humphrey LL, Helfand M, Chan BK, et al. Breast cancer screening: a summary of the evidence for the US Preventive Services Task Force. Ann Intern Med. 2002;137(5_Part_1):347-360.
- US Preventive Services Task Force. Screening for breast cancer: US Preventive Services Task Force recommendation statement. Ann Intern Med. 2009;151:716-726.
- Oeffinger KC, Fontham ET, Etzioni R, et al. Breast cancer screening for women at average risk: 2015 guideline update from the American Cancer Society. JAMA. 2015;314:1599-1614.
- American College of Obstetricans and Gynecologists. ACOG Practice Bulletin number 179: Breast cancer risk assessment and screening in average-risk women. Obstet Gynecol. 2017;130:e1e16. doi: 10.1097/AOG. 0000000000002158.
- Bevers TB, Helvie M, Bonaccio E, et al. Breast cancer screening and diagnosis, Version 3.2018, NCCN Clinical Practice Guidelines in Oncology. J Natl Compr Canc Netw. 2018;16:1362-1389.
- Qaseem A, Lin JS, Mustafa RA, et al. Screening for breast cancer in average-risk women: a guidance statement from the American College of Physicians. Ann Intern Med. 2019;170: 547-560.
- Siu AL, US Preventive Services Task Force. Screening for breast cancer: US Preventive Services Task Force recommendation statement. Ann Intern Med. 2016;164:279-296.
- US Preventive Services Task Force. Draft Recommendation Statement Breast Cancer: Screening. May 9, 2023. Accessed October 25, 2023. https://www.uspreventiveservicestaskforce .org/uspstf/draft-recommendation/breast -cancer-screening-adults#bcei-recommendation -title-area
- Siegel RL, Miller KD, Wagle NS, et al. Cancer statistics, 2023. CA: Cancer J Clin. 2023;73:17-48.
- American Cancer Society. Cancer Statistics Center: Breast. 2023. Accessed October 25, 2023. https ://cancerstatisticscenter.cancer.org/#!/cancer-site /Breast
- Bailey ZD, Krieger N, Agénor M, et al. Structural racism and health inequities in the USA: evidence and interventions. Lancet. 2017;389:1453-1463.
- Collin LJ, Gaglioti AH, Beyer KM, et al. Neighborhood-level redlining and lending bias are associated with breast cancer mortality in a large and diverse metropolitan area. Cancer Epidemiol, Biomarkers Prev. 2021;30:53-60.
- Goel N, Westrick AC, Bailey ZD, et al. Structural racism and breast cancer-specific survival: impact of economic and racial residential segregation. Ann Surg. 2022;275:776-783.
- Chen Y, Susick L, Davis M, et al. Evaluation of triple-negative breast cancer early detection via mammography screening and outcomes in African American and White American patients. JAMA Surg. 2020;155:440-442.
- Stapleton SM, Oseni TO, Bababekov YJ, et al. Race/ethnicity and age distribution of breast cancer diagnosis in the United States. JAMA Surg. 2018;153:594-595.
- Chelmow D, Pearlman MD, Young A, et al. Executive Summary of the Early-Onset Breast Cancer Evidence Review Conference. Obstet Gynecol. 2020;135:1457-1478.
The US Preventive Services Task Force (USPSTF)1 is comprised of an independent panel of preventive services clinician experts who make evidence-based recommendations, with the letter grade assigned based on the strength of the evidence, from A through D (TABLE 1), on preventive services such as health screenings, shared decision making patient counseling, and preventive medications. Both A and B recommendations are generally accepted by both government and most private health insurance companies as a covered preventive benefit with no or minimal co-pays.

In 2002, the USPSTF released a Grade B recommendation that screening mammography for average-risk patients (with patients referring to persons assigned female at birth who have not undergone bilateral mastectomy) should take place starting at age 40 and be repeated every 1 to 2 years.2 This was consistent with or endorsed by most other national breast cancer screening guidelines, including the American College of Obstetricians and Gynecologists (ACOG), National Comprehensive Cancer Network (NCCN), the American Cancer Society (ACS), and the American College of Radiology.
In 2009, the USPSTF changed this Grade B recommendation, instead recommending biennial screening mammography for women aged 50 to 74.3 The most significant change in the revised guideline was for patients aged 40 to 49, where the recommendation was “against routine screening mammography.” They went on to say that the decision to start “biennial screening mammography before the age of 50 years should be an individual one and take patient context into account, including the patient’s values regarding specific benefits and harms.” Other prominent national guideline groups (ACOG, NCCN, ACS) did not agree with this recommendation and maintained that patients aged 40 to 49 should continue to be offered routine screening mammography either annually (NCCN, ACS) or at 1-to-2-year intervals (ACOG).4-6 The American College of Physicians and the American Academy of Family Practice endorsed the 2016 USPSTF guidelines, creating a disparity in breast cancer mammography counseling for averagerisk patients in their 40s.7
In 2016, the USPSTF revisited their breast cancer screening recommendation and renewed their 2009 recommendation against routine screening in patients aged 40 to 49, with the American College of Physicians and the American Academy of Family Practice again endorsing these guidelines.8 ACOG, ACS, NCCN, and ACR continued to recommend age 40 as a starting age for routine mammography screening (TABLE 2). As a result, over the past 14 years, patients aged 40 to 49 were placed in an awkward position of potentially hearing different recommendations from their health care providers, those differences often depending on the specialty of the provider they were seeing.

In 2023. On May 9, the USPSTF released a draft of their latest recommendation statement stating that all patients at average risk for breast cancer should get screened every other year beginning at age 40, bringing most of the national guideline groups into alignment with regard to age to start mammographic screening.9
- With an estimated more than 300,000 new cases in 2023, breast cancer has the highest incidence rate of any cancer in the United States
- The median age of patients with breast cancer in the United States is 58.0 years
- 1 in 5 new breast cancer diagnoses occur in patients between the ages of 40 and 49
- Despite lower incidence rates among Black vs White patients, Black patients have higher death rates from breast cancer
Why the change?
To answer this question, we need to examine the relevant epidemiology of breast cancer.
Continue to: Incidence...
Incidence
It is estimated that, in the United States in 2023, there will be 300,590 new cases of breast cancer, resulting in 43,700 deaths.10 From 2015–2019, there were 128.1 new breast cancer cases/100,000 population, which is the highest rate of cancer in the United States, regardless of sex.11 Diagnoses among patients aged 40 to 49 are rising at a faster rate than previously, about 2% per year between 2015 and 2019.
Racial and ethnic differences
In addition to the racial and ethnic epidemiologic differences in breast cancer, there are also disparities in breast cancer care and outcomes that need to be considered when making national guidelines/policy recommendations.
Black women have high mortality rates from breast cancer. While non-Hispanic White patients have the highest rates of breast cancer (TABLE 3), non-Hispanic Black patients have the highest rates of death due to breast cancer.10 There appear to be several reasons for the estimated 40%-higher rate of mortality among Black women, including:
- systemic racism in primary research, guidelines, and policy
- inequities in diagnostic follow-up and access to evidence-based cancer treatments
- biologic differences in breast cancer (ie, the incidence of triple-negative breast cancer (TNBC) is 2-fold higher in Black women compared with the other racial and ethnic groups in the United States).12-14

While prior studies have suggested that screening mammography might be less effective for patients with TNBC, a recent study demonstrated that patients who had mammography–screened-detected TNBC tumors were smaller and more likely to be node- negative compared with non-screened patients with TNBC.(14) Patients with screened-detected TNBCs were also more likely to undergo a lumpectomy instead of a mastectomy compared with non–screened detected TNBC (68.3% vs 46.1%; P = .002) (TABLE 4). These data strongly suggest that screening mammography is indeed effective in detecting TNBC at earlier stages, one of the best proxies for breast cancer mortality.

Non-White patients have higher incidence rates of breast cancer in their 40s. A second factor to consider in racial differences is the relatively higher incidence of breast cancer in Hispanic, Black, and Asian patients in their 40s compared with non-Hispanic White patients. In a recent analysis of data from 1973 to 2010 from the Surveillance, Epidemiology, and End Results (SEER) Program, the median age of patients with breast cancer in the United States was 58.0 years (interquartile range [IQR], 50.0–67.0 years).16 Across all US demographic populations by age at diagnosis, more than 20% of patients will have their initial diagnosis of breast cancer under the age of 50, and 1.55% (1 in 65) patients between ages 40 and 49 years will be diagnosed with breast cancer.4 However, among patients aged 50 and younger diagnosed with breast cancer, a significantly higher proportion are Black (31%), Hispanic (34.9%), or Asian (32.8%) versus White (23.1%) (P < .001 for all).16 So, for there to be similar racial and ethnic mammography capture rates with White patients, starting mammography screening ages would need to be lower for Black (age 47 years), Hispanic (and 46 years), and Asian (age 47 years) patients. Data from this study of the SEER database16 also demonstrated that more Black and Hispanic patients at age of diagnosis were diagnosed with advanced (regional or distant) breast cancer (46.6% and 42.9%, respectively) versus White or Asian patients (37.1% and 35.6%, respectively; P < .001 for all).
These findings led the authors of the study to conclude that the “Current [2016] USPSTF breast cancer screening recommendations do not reflect age-specific patterns based on race.” The USPSTF stated that this is one of the reasons why they reconsidered their stance on screening , and now recommend screening for all patients starting at age 40.
My current counseling approach
I encourage all racial and ethnic patients between the ages of 40 and 49 to undergo screening mammography because of the associated relative risk mortality reduction rates, which range from 15% to 50%. I also share that with my patients that, because of the younger average age of onset of breast cancer in Black, Hispanic, and Asian patients, they may derive additional benefit from screening starting at age 40.4
Impact of draft guidelines on breast cancer screening and mortality in younger patients
There is clear, unequivocal, and repeatable Level 1 evidence that screening mammography in the general population of patients aged 40 to 49 reduces breast cancer mortality. Breast cancer is the leading cause of cancer in the United States, the second leading cause of cancer mortality in patients, and 1 in 5 new breast cancer diagnoses occur in patients between the ages of 40 and 49. While recent efforts have been made to come to consensus on a screening starting age of 40 for patients at average risk for breast cancer, the USPSTF appeared to be an outlier with their 2016 recommendation to routinely start mammography screening at age 50 instead of 40.17
The USPSTF is a very important national voice in cancer prevention, and their 2023 (draft) revised guidelines to age 40 as the recommended starting screening age now agrees with the leading US guideline groups listed in Table 2. These guideline groups have gone through varying processes, and now have finally arrived at the same conclusion for age to start screening mammography in women of average risk. This agreement should come as a significant comfort to health care providers and patients alike. Changing the starting age to 40 years will result in thousands of lives and hundreds of thousands of life-years saved for patients aged 40 to 49. ●
The US Preventive Services Task Force (USPSTF)1 is comprised of an independent panel of preventive services clinician experts who make evidence-based recommendations, with the letter grade assigned based on the strength of the evidence, from A through D (TABLE 1), on preventive services such as health screenings, shared decision making patient counseling, and preventive medications. Both A and B recommendations are generally accepted by both government and most private health insurance companies as a covered preventive benefit with no or minimal co-pays.

In 2002, the USPSTF released a Grade B recommendation that screening mammography for average-risk patients (with patients referring to persons assigned female at birth who have not undergone bilateral mastectomy) should take place starting at age 40 and be repeated every 1 to 2 years.2 This was consistent with or endorsed by most other national breast cancer screening guidelines, including the American College of Obstetricians and Gynecologists (ACOG), National Comprehensive Cancer Network (NCCN), the American Cancer Society (ACS), and the American College of Radiology.
In 2009, the USPSTF changed this Grade B recommendation, instead recommending biennial screening mammography for women aged 50 to 74.3 The most significant change in the revised guideline was for patients aged 40 to 49, where the recommendation was “against routine screening mammography.” They went on to say that the decision to start “biennial screening mammography before the age of 50 years should be an individual one and take patient context into account, including the patient’s values regarding specific benefits and harms.” Other prominent national guideline groups (ACOG, NCCN, ACS) did not agree with this recommendation and maintained that patients aged 40 to 49 should continue to be offered routine screening mammography either annually (NCCN, ACS) or at 1-to-2-year intervals (ACOG).4-6 The American College of Physicians and the American Academy of Family Practice endorsed the 2016 USPSTF guidelines, creating a disparity in breast cancer mammography counseling for averagerisk patients in their 40s.7
In 2016, the USPSTF revisited their breast cancer screening recommendation and renewed their 2009 recommendation against routine screening in patients aged 40 to 49, with the American College of Physicians and the American Academy of Family Practice again endorsing these guidelines.8 ACOG, ACS, NCCN, and ACR continued to recommend age 40 as a starting age for routine mammography screening (TABLE 2). As a result, over the past 14 years, patients aged 40 to 49 were placed in an awkward position of potentially hearing different recommendations from their health care providers, those differences often depending on the specialty of the provider they were seeing.

In 2023. On May 9, the USPSTF released a draft of their latest recommendation statement stating that all patients at average risk for breast cancer should get screened every other year beginning at age 40, bringing most of the national guideline groups into alignment with regard to age to start mammographic screening.9
- With an estimated more than 300,000 new cases in 2023, breast cancer has the highest incidence rate of any cancer in the United States
- The median age of patients with breast cancer in the United States is 58.0 years
- 1 in 5 new breast cancer diagnoses occur in patients between the ages of 40 and 49
- Despite lower incidence rates among Black vs White patients, Black patients have higher death rates from breast cancer
Why the change?
To answer this question, we need to examine the relevant epidemiology of breast cancer.
Continue to: Incidence...
Incidence
It is estimated that, in the United States in 2023, there will be 300,590 new cases of breast cancer, resulting in 43,700 deaths.10 From 2015–2019, there were 128.1 new breast cancer cases/100,000 population, which is the highest rate of cancer in the United States, regardless of sex.11 Diagnoses among patients aged 40 to 49 are rising at a faster rate than previously, about 2% per year between 2015 and 2019.
Racial and ethnic differences
In addition to the racial and ethnic epidemiologic differences in breast cancer, there are also disparities in breast cancer care and outcomes that need to be considered when making national guidelines/policy recommendations.
Black women have high mortality rates from breast cancer. While non-Hispanic White patients have the highest rates of breast cancer (TABLE 3), non-Hispanic Black patients have the highest rates of death due to breast cancer.10 There appear to be several reasons for the estimated 40%-higher rate of mortality among Black women, including:
- systemic racism in primary research, guidelines, and policy
- inequities in diagnostic follow-up and access to evidence-based cancer treatments
- biologic differences in breast cancer (ie, the incidence of triple-negative breast cancer (TNBC) is 2-fold higher in Black women compared with the other racial and ethnic groups in the United States).12-14

While prior studies have suggested that screening mammography might be less effective for patients with TNBC, a recent study demonstrated that patients who had mammography–screened-detected TNBC tumors were smaller and more likely to be node- negative compared with non-screened patients with TNBC.(14) Patients with screened-detected TNBCs were also more likely to undergo a lumpectomy instead of a mastectomy compared with non–screened detected TNBC (68.3% vs 46.1%; P = .002) (TABLE 4). These data strongly suggest that screening mammography is indeed effective in detecting TNBC at earlier stages, one of the best proxies for breast cancer mortality.

Non-White patients have higher incidence rates of breast cancer in their 40s. A second factor to consider in racial differences is the relatively higher incidence of breast cancer in Hispanic, Black, and Asian patients in their 40s compared with non-Hispanic White patients. In a recent analysis of data from 1973 to 2010 from the Surveillance, Epidemiology, and End Results (SEER) Program, the median age of patients with breast cancer in the United States was 58.0 years (interquartile range [IQR], 50.0–67.0 years).16 Across all US demographic populations by age at diagnosis, more than 20% of patients will have their initial diagnosis of breast cancer under the age of 50, and 1.55% (1 in 65) patients between ages 40 and 49 years will be diagnosed with breast cancer.4 However, among patients aged 50 and younger diagnosed with breast cancer, a significantly higher proportion are Black (31%), Hispanic (34.9%), or Asian (32.8%) versus White (23.1%) (P < .001 for all).16 So, for there to be similar racial and ethnic mammography capture rates with White patients, starting mammography screening ages would need to be lower for Black (age 47 years), Hispanic (and 46 years), and Asian (age 47 years) patients. Data from this study of the SEER database16 also demonstrated that more Black and Hispanic patients at age of diagnosis were diagnosed with advanced (regional or distant) breast cancer (46.6% and 42.9%, respectively) versus White or Asian patients (37.1% and 35.6%, respectively; P < .001 for all).
These findings led the authors of the study to conclude that the “Current [2016] USPSTF breast cancer screening recommendations do not reflect age-specific patterns based on race.” The USPSTF stated that this is one of the reasons why they reconsidered their stance on screening , and now recommend screening for all patients starting at age 40.
My current counseling approach
I encourage all racial and ethnic patients between the ages of 40 and 49 to undergo screening mammography because of the associated relative risk mortality reduction rates, which range from 15% to 50%. I also share that with my patients that, because of the younger average age of onset of breast cancer in Black, Hispanic, and Asian patients, they may derive additional benefit from screening starting at age 40.4
Impact of draft guidelines on breast cancer screening and mortality in younger patients
There is clear, unequivocal, and repeatable Level 1 evidence that screening mammography in the general population of patients aged 40 to 49 reduces breast cancer mortality. Breast cancer is the leading cause of cancer in the United States, the second leading cause of cancer mortality in patients, and 1 in 5 new breast cancer diagnoses occur in patients between the ages of 40 and 49. While recent efforts have been made to come to consensus on a screening starting age of 40 for patients at average risk for breast cancer, the USPSTF appeared to be an outlier with their 2016 recommendation to routinely start mammography screening at age 50 instead of 40.17
The USPSTF is a very important national voice in cancer prevention, and their 2023 (draft) revised guidelines to age 40 as the recommended starting screening age now agrees with the leading US guideline groups listed in Table 2. These guideline groups have gone through varying processes, and now have finally arrived at the same conclusion for age to start screening mammography in women of average risk. This agreement should come as a significant comfort to health care providers and patients alike. Changing the starting age to 40 years will result in thousands of lives and hundreds of thousands of life-years saved for patients aged 40 to 49. ●
- US Preventive Services Task Force website. Task Force at a glance. Accessed October 25, 2023. https://www.uspreventiveservicestaskforce.org /uspstf/about-uspstf/task-force-at-a-glance
- Humphrey LL, Helfand M, Chan BK, et al. Breast cancer screening: a summary of the evidence for the US Preventive Services Task Force. Ann Intern Med. 2002;137(5_Part_1):347-360.
- US Preventive Services Task Force. Screening for breast cancer: US Preventive Services Task Force recommendation statement. Ann Intern Med. 2009;151:716-726.
- Oeffinger KC, Fontham ET, Etzioni R, et al. Breast cancer screening for women at average risk: 2015 guideline update from the American Cancer Society. JAMA. 2015;314:1599-1614.
- American College of Obstetricans and Gynecologists. ACOG Practice Bulletin number 179: Breast cancer risk assessment and screening in average-risk women. Obstet Gynecol. 2017;130:e1e16. doi: 10.1097/AOG. 0000000000002158.
- Bevers TB, Helvie M, Bonaccio E, et al. Breast cancer screening and diagnosis, Version 3.2018, NCCN Clinical Practice Guidelines in Oncology. J Natl Compr Canc Netw. 2018;16:1362-1389.
- Qaseem A, Lin JS, Mustafa RA, et al. Screening for breast cancer in average-risk women: a guidance statement from the American College of Physicians. Ann Intern Med. 2019;170: 547-560.
- Siu AL, US Preventive Services Task Force. Screening for breast cancer: US Preventive Services Task Force recommendation statement. Ann Intern Med. 2016;164:279-296.
- US Preventive Services Task Force. Draft Recommendation Statement Breast Cancer: Screening. May 9, 2023. Accessed October 25, 2023. https://www.uspreventiveservicestaskforce .org/uspstf/draft-recommendation/breast -cancer-screening-adults#bcei-recommendation -title-area
- Siegel RL, Miller KD, Wagle NS, et al. Cancer statistics, 2023. CA: Cancer J Clin. 2023;73:17-48.
- American Cancer Society. Cancer Statistics Center: Breast. 2023. Accessed October 25, 2023. https ://cancerstatisticscenter.cancer.org/#!/cancer-site /Breast
- Bailey ZD, Krieger N, Agénor M, et al. Structural racism and health inequities in the USA: evidence and interventions. Lancet. 2017;389:1453-1463.
- Collin LJ, Gaglioti AH, Beyer KM, et al. Neighborhood-level redlining and lending bias are associated with breast cancer mortality in a large and diverse metropolitan area. Cancer Epidemiol, Biomarkers Prev. 2021;30:53-60.
- Goel N, Westrick AC, Bailey ZD, et al. Structural racism and breast cancer-specific survival: impact of economic and racial residential segregation. Ann Surg. 2022;275:776-783.
- Chen Y, Susick L, Davis M, et al. Evaluation of triple-negative breast cancer early detection via mammography screening and outcomes in African American and White American patients. JAMA Surg. 2020;155:440-442.
- Stapleton SM, Oseni TO, Bababekov YJ, et al. Race/ethnicity and age distribution of breast cancer diagnosis in the United States. JAMA Surg. 2018;153:594-595.
- Chelmow D, Pearlman MD, Young A, et al. Executive Summary of the Early-Onset Breast Cancer Evidence Review Conference. Obstet Gynecol. 2020;135:1457-1478.
- US Preventive Services Task Force website. Task Force at a glance. Accessed October 25, 2023. https://www.uspreventiveservicestaskforce.org /uspstf/about-uspstf/task-force-at-a-glance
- Humphrey LL, Helfand M, Chan BK, et al. Breast cancer screening: a summary of the evidence for the US Preventive Services Task Force. Ann Intern Med. 2002;137(5_Part_1):347-360.
- US Preventive Services Task Force. Screening for breast cancer: US Preventive Services Task Force recommendation statement. Ann Intern Med. 2009;151:716-726.
- Oeffinger KC, Fontham ET, Etzioni R, et al. Breast cancer screening for women at average risk: 2015 guideline update from the American Cancer Society. JAMA. 2015;314:1599-1614.
- American College of Obstetricans and Gynecologists. ACOG Practice Bulletin number 179: Breast cancer risk assessment and screening in average-risk women. Obstet Gynecol. 2017;130:e1e16. doi: 10.1097/AOG. 0000000000002158.
- Bevers TB, Helvie M, Bonaccio E, et al. Breast cancer screening and diagnosis, Version 3.2018, NCCN Clinical Practice Guidelines in Oncology. J Natl Compr Canc Netw. 2018;16:1362-1389.
- Qaseem A, Lin JS, Mustafa RA, et al. Screening for breast cancer in average-risk women: a guidance statement from the American College of Physicians. Ann Intern Med. 2019;170: 547-560.
- Siu AL, US Preventive Services Task Force. Screening for breast cancer: US Preventive Services Task Force recommendation statement. Ann Intern Med. 2016;164:279-296.
- US Preventive Services Task Force. Draft Recommendation Statement Breast Cancer: Screening. May 9, 2023. Accessed October 25, 2023. https://www.uspreventiveservicestaskforce .org/uspstf/draft-recommendation/breast -cancer-screening-adults#bcei-recommendation -title-area
- Siegel RL, Miller KD, Wagle NS, et al. Cancer statistics, 2023. CA: Cancer J Clin. 2023;73:17-48.
- American Cancer Society. Cancer Statistics Center: Breast. 2023. Accessed October 25, 2023. https ://cancerstatisticscenter.cancer.org/#!/cancer-site /Breast
- Bailey ZD, Krieger N, Agénor M, et al. Structural racism and health inequities in the USA: evidence and interventions. Lancet. 2017;389:1453-1463.
- Collin LJ, Gaglioti AH, Beyer KM, et al. Neighborhood-level redlining and lending bias are associated with breast cancer mortality in a large and diverse metropolitan area. Cancer Epidemiol, Biomarkers Prev. 2021;30:53-60.
- Goel N, Westrick AC, Bailey ZD, et al. Structural racism and breast cancer-specific survival: impact of economic and racial residential segregation. Ann Surg. 2022;275:776-783.
- Chen Y, Susick L, Davis M, et al. Evaluation of triple-negative breast cancer early detection via mammography screening and outcomes in African American and White American patients. JAMA Surg. 2020;155:440-442.
- Stapleton SM, Oseni TO, Bababekov YJ, et al. Race/ethnicity and age distribution of breast cancer diagnosis in the United States. JAMA Surg. 2018;153:594-595.
- Chelmow D, Pearlman MD, Young A, et al. Executive Summary of the Early-Onset Breast Cancer Evidence Review Conference. Obstet Gynecol. 2020;135:1457-1478.
Real-world study confirms benefits of erenumab for migraine prevention
Key clinical point: This real-world study confirms the efficacy and safety of erenumab in patients with migraine associated with extreme unmet needs.
Major finding: Overall, 52.9%, 58.5%, 57.0%, and 58.8% of patients receiving erenumab achieved ≥50% reduction in monthly migraine days at 3, 6, 9, and 12 months, respectively, with significant reductions in the proportion of patients with chronic migraine at all time points compared with baseline (P < .001). At month 3, 57.3% of patients changed from chronic migraine to episodic migraine. No treatment-related serious adverse events were reported.
Study details: Findings are from a 1-year prospective, real-word study including 140 patients with migraine and previous migraine preventive treatment failures who received ≥1 dose of erenumab.
Disclosures: This study did not receive funding from any source. Several authors declared receiving personal fees for consultancy activities or research support from various sources, and some authors declared no conflicts of interest.
Source: Lanteri-Minet M et al. One-year prospective real-world assessment of effectiveness and safety of erenumab in migraine prevention: Results of the French FHU INOVPAIN registry study. J Headache Pain. 2023;24:152 (Nov 8). doi: 10.1186/s10194-023-01680-4
Key clinical point: This real-world study confirms the efficacy and safety of erenumab in patients with migraine associated with extreme unmet needs.
Major finding: Overall, 52.9%, 58.5%, 57.0%, and 58.8% of patients receiving erenumab achieved ≥50% reduction in monthly migraine days at 3, 6, 9, and 12 months, respectively, with significant reductions in the proportion of patients with chronic migraine at all time points compared with baseline (P < .001). At month 3, 57.3% of patients changed from chronic migraine to episodic migraine. No treatment-related serious adverse events were reported.
Study details: Findings are from a 1-year prospective, real-word study including 140 patients with migraine and previous migraine preventive treatment failures who received ≥1 dose of erenumab.
Disclosures: This study did not receive funding from any source. Several authors declared receiving personal fees for consultancy activities or research support from various sources, and some authors declared no conflicts of interest.
Source: Lanteri-Minet M et al. One-year prospective real-world assessment of effectiveness and safety of erenumab in migraine prevention: Results of the French FHU INOVPAIN registry study. J Headache Pain. 2023;24:152 (Nov 8). doi: 10.1186/s10194-023-01680-4
Key clinical point: This real-world study confirms the efficacy and safety of erenumab in patients with migraine associated with extreme unmet needs.
Major finding: Overall, 52.9%, 58.5%, 57.0%, and 58.8% of patients receiving erenumab achieved ≥50% reduction in monthly migraine days at 3, 6, 9, and 12 months, respectively, with significant reductions in the proportion of patients with chronic migraine at all time points compared with baseline (P < .001). At month 3, 57.3% of patients changed from chronic migraine to episodic migraine. No treatment-related serious adverse events were reported.
Study details: Findings are from a 1-year prospective, real-word study including 140 patients with migraine and previous migraine preventive treatment failures who received ≥1 dose of erenumab.
Disclosures: This study did not receive funding from any source. Several authors declared receiving personal fees for consultancy activities or research support from various sources, and some authors declared no conflicts of interest.
Source: Lanteri-Minet M et al. One-year prospective real-world assessment of effectiveness and safety of erenumab in migraine prevention: Results of the French FHU INOVPAIN registry study. J Headache Pain. 2023;24:152 (Nov 8). doi: 10.1186/s10194-023-01680-4
Heavy secondhand smoke exposure tied to higher risk for severe headaches or migraine
Key clinical point: Heavy secondhand smoke (SHS) exposure was positively associated with a higher risk for severe headaches or migraine in adults who never smoked.
Major finding: Heavy SHS exposure (serum cotinine level 1-10 ng/mL) was positively associated with severe headaches or migraine (adjusted odds ratio 2.02; P = .011). No significant association was observed between low SHS exposure (serum cotinine level 0.05-0.99 ng/mL) and headaches or migraine (P = .226).
Study details: This nationwide cross-sectional study included 4560 adults who had never smoked, of which 20% had severe headaches or migraine.
Disclosures: This study did not receive any funding from external sources. The authors declared no conflicts of interest.
Source: Wu J, Yang P, et al. Association between secondhand smoke exposure and severe headaches or migraine in never-smoking adults. Headache. 2023 (Nov 8). doi: 10.1111/head.14640
Key clinical point: Heavy secondhand smoke (SHS) exposure was positively associated with a higher risk for severe headaches or migraine in adults who never smoked.
Major finding: Heavy SHS exposure (serum cotinine level 1-10 ng/mL) was positively associated with severe headaches or migraine (adjusted odds ratio 2.02; P = .011). No significant association was observed between low SHS exposure (serum cotinine level 0.05-0.99 ng/mL) and headaches or migraine (P = .226).
Study details: This nationwide cross-sectional study included 4560 adults who had never smoked, of which 20% had severe headaches or migraine.
Disclosures: This study did not receive any funding from external sources. The authors declared no conflicts of interest.
Source: Wu J, Yang P, et al. Association between secondhand smoke exposure and severe headaches or migraine in never-smoking adults. Headache. 2023 (Nov 8). doi: 10.1111/head.14640
Key clinical point: Heavy secondhand smoke (SHS) exposure was positively associated with a higher risk for severe headaches or migraine in adults who never smoked.
Major finding: Heavy SHS exposure (serum cotinine level 1-10 ng/mL) was positively associated with severe headaches or migraine (adjusted odds ratio 2.02; P = .011). No significant association was observed between low SHS exposure (serum cotinine level 0.05-0.99 ng/mL) and headaches or migraine (P = .226).
Study details: This nationwide cross-sectional study included 4560 adults who had never smoked, of which 20% had severe headaches or migraine.
Disclosures: This study did not receive any funding from external sources. The authors declared no conflicts of interest.
Source: Wu J, Yang P, et al. Association between secondhand smoke exposure and severe headaches or migraine in never-smoking adults. Headache. 2023 (Nov 8). doi: 10.1111/head.14640
Heart rate variability may help predict treatment response in chronic migraine
Key clinical point: Patients with chronic migraine have autonomic dysfunction, the extent of which is evaluated using heart rate variability (HRV) analysis, and a preserved function is associated with a superior response to flunarizine preventive treatment.
Major finding: Most heart-rate variability (HRV) parameters, except the ratios of low-frequency (LF) band and LF/high-frequency band, were significantly lower in patients with migraine vs control individuals (P < .001). The response to flunarizine treatment was superior in patients with normal HRV, as exemplified by higher reductions in monthly headache days after 3 months in those with normal vs lower HRV (─9.7 days vs ─6.2 days; P = .026).
Study details: This cross-sectional, prospective study included 81 prophylaxis-naive patients with chronic migraine and 58 age- and gender-matched control individuals. Patients with migraine initiated flunarizine as a preventive treatment for 12 weeks.
Disclosures: This study was supported by grants from the National Science and Technology Council, Taiwan, and others. SJ Wang declared being a principal investigator and receiving personal fees as an advisor or speaker from various sources.
Source: Chuang CH et al. Abnormal heart rate variability and its application in predicting treatment efficacy in patients with chronic migraine: An exploratory study. Cephalalgia. 2023 (Oct 18). doi: 10.1177/03331024231206781
Key clinical point: Patients with chronic migraine have autonomic dysfunction, the extent of which is evaluated using heart rate variability (HRV) analysis, and a preserved function is associated with a superior response to flunarizine preventive treatment.
Major finding: Most heart-rate variability (HRV) parameters, except the ratios of low-frequency (LF) band and LF/high-frequency band, were significantly lower in patients with migraine vs control individuals (P < .001). The response to flunarizine treatment was superior in patients with normal HRV, as exemplified by higher reductions in monthly headache days after 3 months in those with normal vs lower HRV (─9.7 days vs ─6.2 days; P = .026).
Study details: This cross-sectional, prospective study included 81 prophylaxis-naive patients with chronic migraine and 58 age- and gender-matched control individuals. Patients with migraine initiated flunarizine as a preventive treatment for 12 weeks.
Disclosures: This study was supported by grants from the National Science and Technology Council, Taiwan, and others. SJ Wang declared being a principal investigator and receiving personal fees as an advisor or speaker from various sources.
Source: Chuang CH et al. Abnormal heart rate variability and its application in predicting treatment efficacy in patients with chronic migraine: An exploratory study. Cephalalgia. 2023 (Oct 18). doi: 10.1177/03331024231206781
Key clinical point: Patients with chronic migraine have autonomic dysfunction, the extent of which is evaluated using heart rate variability (HRV) analysis, and a preserved function is associated with a superior response to flunarizine preventive treatment.
Major finding: Most heart-rate variability (HRV) parameters, except the ratios of low-frequency (LF) band and LF/high-frequency band, were significantly lower in patients with migraine vs control individuals (P < .001). The response to flunarizine treatment was superior in patients with normal HRV, as exemplified by higher reductions in monthly headache days after 3 months in those with normal vs lower HRV (─9.7 days vs ─6.2 days; P = .026).
Study details: This cross-sectional, prospective study included 81 prophylaxis-naive patients with chronic migraine and 58 age- and gender-matched control individuals. Patients with migraine initiated flunarizine as a preventive treatment for 12 weeks.
Disclosures: This study was supported by grants from the National Science and Technology Council, Taiwan, and others. SJ Wang declared being a principal investigator and receiving personal fees as an advisor or speaker from various sources.
Source: Chuang CH et al. Abnormal heart rate variability and its application in predicting treatment efficacy in patients with chronic migraine: An exploratory study. Cephalalgia. 2023 (Oct 18). doi: 10.1177/03331024231206781
Remote electrical neuromodulation: A pill-free, needle-free option for long-term migraine management
Key clinical point: This real-world study confirms the safety, efficacy, and tolerability of remote electrical neuromodulation (REN) for long-term management of acute migraine, thus establishing REN as a valuable comprehensive treatment for this chronic disease.
Major finding: Overall, 74.1% and 26.0% of patients achieved consistent pain relief and pain freedom with REN, respectively, and 70.2% and 33.7% achieved functional disability relief and functional disability freedom, respectively. The incidence of device-related adverse events (dAE) was low, ie, 1.96%, which included 0.49% negligible, 1.22% moderate, and 0.24% mild AE. No severe AE were reported, and all patients continued treatment despite dAE.
Study details: This real-world evidence study included 409 patients with migraine treated for 12 consecutive months with REN, a self-administered device used at the onset of migraine headache or aura for acute treatment.
Disclosures: This study was funded by Theranica Bio-Electronics Ltd. M Weinstein and A Synowiec declared serving as consultants for Theranica. A Stark-Inbar and A Ironi declared being employees of and hold stock options in Theranica. A Mauskop had no conflicts of interest to disclose.
Source: Synowiec A et al. One-year consistent safety, utilization, and efficacy assessment of remote electrical neuromodulation (REN) for migraine treatment. Adv Ther. 2023 (Oct 19). doi: 10.1007/s12325-023-02697-6
Key clinical point: This real-world study confirms the safety, efficacy, and tolerability of remote electrical neuromodulation (REN) for long-term management of acute migraine, thus establishing REN as a valuable comprehensive treatment for this chronic disease.
Major finding: Overall, 74.1% and 26.0% of patients achieved consistent pain relief and pain freedom with REN, respectively, and 70.2% and 33.7% achieved functional disability relief and functional disability freedom, respectively. The incidence of device-related adverse events (dAE) was low, ie, 1.96%, which included 0.49% negligible, 1.22% moderate, and 0.24% mild AE. No severe AE were reported, and all patients continued treatment despite dAE.
Study details: This real-world evidence study included 409 patients with migraine treated for 12 consecutive months with REN, a self-administered device used at the onset of migraine headache or aura for acute treatment.
Disclosures: This study was funded by Theranica Bio-Electronics Ltd. M Weinstein and A Synowiec declared serving as consultants for Theranica. A Stark-Inbar and A Ironi declared being employees of and hold stock options in Theranica. A Mauskop had no conflicts of interest to disclose.
Source: Synowiec A et al. One-year consistent safety, utilization, and efficacy assessment of remote electrical neuromodulation (REN) for migraine treatment. Adv Ther. 2023 (Oct 19). doi: 10.1007/s12325-023-02697-6
Key clinical point: This real-world study confirms the safety, efficacy, and tolerability of remote electrical neuromodulation (REN) for long-term management of acute migraine, thus establishing REN as a valuable comprehensive treatment for this chronic disease.
Major finding: Overall, 74.1% and 26.0% of patients achieved consistent pain relief and pain freedom with REN, respectively, and 70.2% and 33.7% achieved functional disability relief and functional disability freedom, respectively. The incidence of device-related adverse events (dAE) was low, ie, 1.96%, which included 0.49% negligible, 1.22% moderate, and 0.24% mild AE. No severe AE were reported, and all patients continued treatment despite dAE.
Study details: This real-world evidence study included 409 patients with migraine treated for 12 consecutive months with REN, a self-administered device used at the onset of migraine headache or aura for acute treatment.
Disclosures: This study was funded by Theranica Bio-Electronics Ltd. M Weinstein and A Synowiec declared serving as consultants for Theranica. A Stark-Inbar and A Ironi declared being employees of and hold stock options in Theranica. A Mauskop had no conflicts of interest to disclose.
Source: Synowiec A et al. One-year consistent safety, utilization, and efficacy assessment of remote electrical neuromodulation (REN) for migraine treatment. Adv Ther. 2023 (Oct 19). doi: 10.1007/s12325-023-02697-6
Ubrogepant and anti-CGRP mAb combo is effective for acute treatment of migraine
Key clinical point: The use of ubrogepant in combination with anti-calcitonin gene-related peptide (CGRP) monoclonal antibody (mAb) leads to meaningful pain relief (MPR), return to normal function (RNF), treatment satisfaction, and acute treatment optimization in patients with migraine.
Major finding: Following the first treated attack, 61.6% and 80.4% of patients achieved MPR and 34.7% and 55.5% of patients achieved RNF at 2 hours and 4 hours post-dose, respectively, in the ubrogepant plus anti-CGRP mAb arm. Moreover, 72.7% of patients reported being satisfied with ubrogepant when used in combination with anti-CGRP mAb, and 79.7% of patients achieved acute treatment optimization at 30 days.
Study details: Findings are from a prospective, observational study that included 245 patients with migraine who were treated with ubrogepant combined with anti-CGRP mAb, onabotulinumtoxinA, or both, for migraine prevention.
Disclosures: This study was funded by Allergan (prior to its acquisition by AbbVie). RB Lipton declared receiving research support, honoraria, and royalties from, and serving as a consultant and advisory board member for various sources, including AbbVie or Allergan.
Source: Lipton RB et al. Real-world use of ubrogepant as acute treatment for migraine with an anti-calcitonin gene-related peptide monoclonal antibody: Results from COURAGE. Neurol Ther. 2023 (Nov 1). doi: 10.1007/s40120-023-00556-8
Key clinical point: The use of ubrogepant in combination with anti-calcitonin gene-related peptide (CGRP) monoclonal antibody (mAb) leads to meaningful pain relief (MPR), return to normal function (RNF), treatment satisfaction, and acute treatment optimization in patients with migraine.
Major finding: Following the first treated attack, 61.6% and 80.4% of patients achieved MPR and 34.7% and 55.5% of patients achieved RNF at 2 hours and 4 hours post-dose, respectively, in the ubrogepant plus anti-CGRP mAb arm. Moreover, 72.7% of patients reported being satisfied with ubrogepant when used in combination with anti-CGRP mAb, and 79.7% of patients achieved acute treatment optimization at 30 days.
Study details: Findings are from a prospective, observational study that included 245 patients with migraine who were treated with ubrogepant combined with anti-CGRP mAb, onabotulinumtoxinA, or both, for migraine prevention.
Disclosures: This study was funded by Allergan (prior to its acquisition by AbbVie). RB Lipton declared receiving research support, honoraria, and royalties from, and serving as a consultant and advisory board member for various sources, including AbbVie or Allergan.
Source: Lipton RB et al. Real-world use of ubrogepant as acute treatment for migraine with an anti-calcitonin gene-related peptide monoclonal antibody: Results from COURAGE. Neurol Ther. 2023 (Nov 1). doi: 10.1007/s40120-023-00556-8
Key clinical point: The use of ubrogepant in combination with anti-calcitonin gene-related peptide (CGRP) monoclonal antibody (mAb) leads to meaningful pain relief (MPR), return to normal function (RNF), treatment satisfaction, and acute treatment optimization in patients with migraine.
Major finding: Following the first treated attack, 61.6% and 80.4% of patients achieved MPR and 34.7% and 55.5% of patients achieved RNF at 2 hours and 4 hours post-dose, respectively, in the ubrogepant plus anti-CGRP mAb arm. Moreover, 72.7% of patients reported being satisfied with ubrogepant when used in combination with anti-CGRP mAb, and 79.7% of patients achieved acute treatment optimization at 30 days.
Study details: Findings are from a prospective, observational study that included 245 patients with migraine who were treated with ubrogepant combined with anti-CGRP mAb, onabotulinumtoxinA, or both, for migraine prevention.
Disclosures: This study was funded by Allergan (prior to its acquisition by AbbVie). RB Lipton declared receiving research support, honoraria, and royalties from, and serving as a consultant and advisory board member for various sources, including AbbVie or Allergan.
Source: Lipton RB et al. Real-world use of ubrogepant as acute treatment for migraine with an anti-calcitonin gene-related peptide monoclonal antibody: Results from COURAGE. Neurol Ther. 2023 (Nov 1). doi: 10.1007/s40120-023-00556-8
Effect of CGRP mAb rollout on prescription patterns of other migraine preventive therapies
Key clinical point: The introduction of calcitonin gene-related peptide (CGRP) monoclonal antibodies (mAb) has led to a reduction in the prescription of other oral preventive therapies for chronic migraine, likely due to the similar efficacy and better safety profile of CGRP mAb.
Major finding: Overall, the percentage of commonly prescribed preventive medications reduced significantly from 46.3% before the introduction of CGRP mAb to 43.1% post introduction (P = .001), including a large decrease in the prescription of verapamil, tricyclic antidepressants, topiramate, onabotulinumtoxinA, valproate, duloxetine, memantine, and propranolol (all P < .05).
Study details: This retrospective cohort study compared the percentage of patients with chronic migraine who were prescribed oral preventive medications or onabotulinumtoxinA during the CGRP mAb pre-approval period (2015-2017; n = 3144) and post-approval period (2019-2021; n = 4629).
Disclosures: This study did not receive any funding. The authors declared no conflicts of interest.
Source: Moskatel LS et al. The introduction of the CGRP monoclonal antibodies and their effect on the prescription patterns of chronic migraine preventive medications in a tertiary headache center: A retrospective, observational analysis. Headache. 2023 (Oct 26). doi: 10.1111/head.14642
Key clinical point: The introduction of calcitonin gene-related peptide (CGRP) monoclonal antibodies (mAb) has led to a reduction in the prescription of other oral preventive therapies for chronic migraine, likely due to the similar efficacy and better safety profile of CGRP mAb.
Major finding: Overall, the percentage of commonly prescribed preventive medications reduced significantly from 46.3% before the introduction of CGRP mAb to 43.1% post introduction (P = .001), including a large decrease in the prescription of verapamil, tricyclic antidepressants, topiramate, onabotulinumtoxinA, valproate, duloxetine, memantine, and propranolol (all P < .05).
Study details: This retrospective cohort study compared the percentage of patients with chronic migraine who were prescribed oral preventive medications or onabotulinumtoxinA during the CGRP mAb pre-approval period (2015-2017; n = 3144) and post-approval period (2019-2021; n = 4629).
Disclosures: This study did not receive any funding. The authors declared no conflicts of interest.
Source: Moskatel LS et al. The introduction of the CGRP monoclonal antibodies and their effect on the prescription patterns of chronic migraine preventive medications in a tertiary headache center: A retrospective, observational analysis. Headache. 2023 (Oct 26). doi: 10.1111/head.14642
Key clinical point: The introduction of calcitonin gene-related peptide (CGRP) monoclonal antibodies (mAb) has led to a reduction in the prescription of other oral preventive therapies for chronic migraine, likely due to the similar efficacy and better safety profile of CGRP mAb.
Major finding: Overall, the percentage of commonly prescribed preventive medications reduced significantly from 46.3% before the introduction of CGRP mAb to 43.1% post introduction (P = .001), including a large decrease in the prescription of verapamil, tricyclic antidepressants, topiramate, onabotulinumtoxinA, valproate, duloxetine, memantine, and propranolol (all P < .05).
Study details: This retrospective cohort study compared the percentage of patients with chronic migraine who were prescribed oral preventive medications or onabotulinumtoxinA during the CGRP mAb pre-approval period (2015-2017; n = 3144) and post-approval period (2019-2021; n = 4629).
Disclosures: This study did not receive any funding. The authors declared no conflicts of interest.
Source: Moskatel LS et al. The introduction of the CGRP monoclonal antibodies and their effect on the prescription patterns of chronic migraine preventive medications in a tertiary headache center: A retrospective, observational analysis. Headache. 2023 (Oct 26). doi: 10.1111/head.14642

