User login
Physician Pay Rule Contains Good and Bad News
Medicare’s physician fee schedule for 2013 contains both a 26.5% pay cut based on the Sustainable Growth Rate formula and pay increases for care coordination and for primary care provided under Medicaid.
Under current law, the Sustainable Growth Rate (SGR) formula will kick in Jan. 1 and lop one-fourth off doctors’ pay under Medicare, unless Congress steps in to halt the cut.
In issuing the fee schedule final regulation on Nov. 1, the Obama administration noted that Congress has reversed the mandated cut every year since 2003.
The administration "is committed to fixing the SGR update methodology and ensuring these payment cuts do not take effect," according to a statement. "Predictable, fiscally responsible physician payments are essential for Medicare to sustain quality and lower health care costs over the long term."
The American Medical Association decried the SGR cut.
"Eliminating this failed formula will allow us to enter a period when physicians can begin transitioning to new payment and delivery models to help meet the overall goal of improving patient care and moving to a higher performing Medicare program," Dr. Ardis D. Hoven, AMA president-elect, said in a statement.*
The fee schedule final rule also includes changes to the value-based modifier program, designed to pay physicians based on the quality of care they deliver.
In a proposed rule issued earlier this year, physicians in groups of 25 or larger would have been subject to the new pay plan in 2015. The final rule increases the size of the group to 100 initially.
In addition, the final rule creates a new set of codes to pay physicians for care coordination in the 30 days after a patient is discharged from a hospital or nursing home. Those codes were initially proposed as G codes, but now will be full-fledged codes in the AMA Current Procedural Terminology (CPT). Physicians will be rewarded for patient interactions that are not face to face, such as phone consults, chart reviews, and email communications.
In another final rule issued Nov. 1, the CMS announced that, as expected, it will pay certain providers the Medicare pay rate for certain primary care services provided under Medicaid in 2013 and 2014.
As called for under the Affordable Care Act, the CMS will pay Medicare rates for evaluation and management codes between 99201 and 99499 when they are used by physicians who are board certified by the American Board of Medical Specialties, the American Osteopathic Association, and the American Board of Physician Specialties.
The codes cover not just primary care, but also hospital observation and consultation for inpatient services provided by nonadmitting physicians, emergency department services, and critical care services.
The final rule does not, however, allow emergency physicians or obstetricians to be compensated at the Medicare level for services under Medicaid, according to the consulting firm Washington Analysis.
The CMS estimated that increasing the Medicaid pay will cost $5.6 billion in 2013 and $5.7 billion in 2014.
Dr. Thomas K. McInerny, president of the American Academy of Pediatrics, said that pay parity between Medicaid and Medicare was "a monumental victory for children’s access to quality health care." He noted that children make up more than half of all recipients of Medicaid services, and that pediatricians provide almost two-thirds of the office visits for those children.
The American Academy of Family Physicians also praised the rule. Dr. Jeffrey Cain, AAFP president, agreed that pay parity would increase access to care.
"More than 6 in 10 (64%) family physicians accept new Medicaid patients, and these beneficiaries comprise 15% of the average family physician’s patient panel," he said in a statement. "However, increasingly inadequate Medicaid payment has forced nearly 2 in 10 family physicians to stop accepting new Medicaid patients."
The rules will be published in the Federal Register on Nov. 16 and comments will close on Dec. 31. Both rules take effect Jan. 1, 2013.
*Correction, 11/6/2012: An earlier version of this story mischaracterized Dr. Hoven's position within the AMA.
Medicare’s physician fee schedule for 2013 contains both a 26.5% pay cut based on the Sustainable Growth Rate formula and pay increases for care coordination and for primary care provided under Medicaid.
Under current law, the Sustainable Growth Rate (SGR) formula will kick in Jan. 1 and lop one-fourth off doctors’ pay under Medicare, unless Congress steps in to halt the cut.
In issuing the fee schedule final regulation on Nov. 1, the Obama administration noted that Congress has reversed the mandated cut every year since 2003.
The administration "is committed to fixing the SGR update methodology and ensuring these payment cuts do not take effect," according to a statement. "Predictable, fiscally responsible physician payments are essential for Medicare to sustain quality and lower health care costs over the long term."
The American Medical Association decried the SGR cut.
"Eliminating this failed formula will allow us to enter a period when physicians can begin transitioning to new payment and delivery models to help meet the overall goal of improving patient care and moving to a higher performing Medicare program," Dr. Ardis D. Hoven, AMA president-elect, said in a statement.*
The fee schedule final rule also includes changes to the value-based modifier program, designed to pay physicians based on the quality of care they deliver.
In a proposed rule issued earlier this year, physicians in groups of 25 or larger would have been subject to the new pay plan in 2015. The final rule increases the size of the group to 100 initially.
In addition, the final rule creates a new set of codes to pay physicians for care coordination in the 30 days after a patient is discharged from a hospital or nursing home. Those codes were initially proposed as G codes, but now will be full-fledged codes in the AMA Current Procedural Terminology (CPT). Physicians will be rewarded for patient interactions that are not face to face, such as phone consults, chart reviews, and email communications.
In another final rule issued Nov. 1, the CMS announced that, as expected, it will pay certain providers the Medicare pay rate for certain primary care services provided under Medicaid in 2013 and 2014.
As called for under the Affordable Care Act, the CMS will pay Medicare rates for evaluation and management codes between 99201 and 99499 when they are used by physicians who are board certified by the American Board of Medical Specialties, the American Osteopathic Association, and the American Board of Physician Specialties.
The codes cover not just primary care, but also hospital observation and consultation for inpatient services provided by nonadmitting physicians, emergency department services, and critical care services.
The final rule does not, however, allow emergency physicians or obstetricians to be compensated at the Medicare level for services under Medicaid, according to the consulting firm Washington Analysis.
The CMS estimated that increasing the Medicaid pay will cost $5.6 billion in 2013 and $5.7 billion in 2014.
Dr. Thomas K. McInerny, president of the American Academy of Pediatrics, said that pay parity between Medicaid and Medicare was "a monumental victory for children’s access to quality health care." He noted that children make up more than half of all recipients of Medicaid services, and that pediatricians provide almost two-thirds of the office visits for those children.
The American Academy of Family Physicians also praised the rule. Dr. Jeffrey Cain, AAFP president, agreed that pay parity would increase access to care.
"More than 6 in 10 (64%) family physicians accept new Medicaid patients, and these beneficiaries comprise 15% of the average family physician’s patient panel," he said in a statement. "However, increasingly inadequate Medicaid payment has forced nearly 2 in 10 family physicians to stop accepting new Medicaid patients."
The rules will be published in the Federal Register on Nov. 16 and comments will close on Dec. 31. Both rules take effect Jan. 1, 2013.
*Correction, 11/6/2012: An earlier version of this story mischaracterized Dr. Hoven's position within the AMA.
Medicare’s physician fee schedule for 2013 contains both a 26.5% pay cut based on the Sustainable Growth Rate formula and pay increases for care coordination and for primary care provided under Medicaid.
Under current law, the Sustainable Growth Rate (SGR) formula will kick in Jan. 1 and lop one-fourth off doctors’ pay under Medicare, unless Congress steps in to halt the cut.
In issuing the fee schedule final regulation on Nov. 1, the Obama administration noted that Congress has reversed the mandated cut every year since 2003.
The administration "is committed to fixing the SGR update methodology and ensuring these payment cuts do not take effect," according to a statement. "Predictable, fiscally responsible physician payments are essential for Medicare to sustain quality and lower health care costs over the long term."
The American Medical Association decried the SGR cut.
"Eliminating this failed formula will allow us to enter a period when physicians can begin transitioning to new payment and delivery models to help meet the overall goal of improving patient care and moving to a higher performing Medicare program," Dr. Ardis D. Hoven, AMA president-elect, said in a statement.*
The fee schedule final rule also includes changes to the value-based modifier program, designed to pay physicians based on the quality of care they deliver.
In a proposed rule issued earlier this year, physicians in groups of 25 or larger would have been subject to the new pay plan in 2015. The final rule increases the size of the group to 100 initially.
In addition, the final rule creates a new set of codes to pay physicians for care coordination in the 30 days after a patient is discharged from a hospital or nursing home. Those codes were initially proposed as G codes, but now will be full-fledged codes in the AMA Current Procedural Terminology (CPT). Physicians will be rewarded for patient interactions that are not face to face, such as phone consults, chart reviews, and email communications.
In another final rule issued Nov. 1, the CMS announced that, as expected, it will pay certain providers the Medicare pay rate for certain primary care services provided under Medicaid in 2013 and 2014.
As called for under the Affordable Care Act, the CMS will pay Medicare rates for evaluation and management codes between 99201 and 99499 when they are used by physicians who are board certified by the American Board of Medical Specialties, the American Osteopathic Association, and the American Board of Physician Specialties.
The codes cover not just primary care, but also hospital observation and consultation for inpatient services provided by nonadmitting physicians, emergency department services, and critical care services.
The final rule does not, however, allow emergency physicians or obstetricians to be compensated at the Medicare level for services under Medicaid, according to the consulting firm Washington Analysis.
The CMS estimated that increasing the Medicaid pay will cost $5.6 billion in 2013 and $5.7 billion in 2014.
Dr. Thomas K. McInerny, president of the American Academy of Pediatrics, said that pay parity between Medicaid and Medicare was "a monumental victory for children’s access to quality health care." He noted that children make up more than half of all recipients of Medicaid services, and that pediatricians provide almost two-thirds of the office visits for those children.
The American Academy of Family Physicians also praised the rule. Dr. Jeffrey Cain, AAFP president, agreed that pay parity would increase access to care.
"More than 6 in 10 (64%) family physicians accept new Medicaid patients, and these beneficiaries comprise 15% of the average family physician’s patient panel," he said in a statement. "However, increasingly inadequate Medicaid payment has forced nearly 2 in 10 family physicians to stop accepting new Medicaid patients."
The rules will be published in the Federal Register on Nov. 16 and comments will close on Dec. 31. Both rules take effect Jan. 1, 2013.
*Correction, 11/6/2012: An earlier version of this story mischaracterized Dr. Hoven's position within the AMA.
AAFP Congress Endorses Same-Sex Civil Marriage
PHILADELPHIA – The American Academy of Family Physicians concluded its 3-day Congress of Delegates meeting with a bold move forward – but it was probably not in an area that most members were expecting.
Debate among the 223 delegates and alternates regarding the academy’s participation in the American Medical Association’s Relative Value Scale Update Committee (RUC) ended up taking a backseat to controversy over resolutions urging the organization to endorse same-sex civil marriage*. In the end, delegates decided to maintain the status quo on the AAFP’s RUC efforts, but ended a 3-year gridlock over same-sex civil marriage by endorsing it by a vote of 75 to 44.
Debate over civil marriage for same-sex couples was intense and heated, with at least an hour of impassioned discussion at the Reference Committee on Advocacy, and close to that on the floor before the delegates voted to close debate.
Many supporters testified that their own same-sex partnerships had suffered as a result of their being denied the ability to marry.
Dr. Mark Dressner, president-elect of the California chapter, said that he felt that he and his partner had been treated as second-class citizens. Others spoke of the emotional and physical damage they had witnessed in patients and family members as a result of discrimination against homosexuals.
Dr. Lloyd Van Winkle of Castroville, Tex., who was just elected to the AAFP board, said that his cousin's son Christopher committed suicide at age 19 because he felt persecuted about his homosexuality.**
Dr. Paul W. Davis, a delegate from Alaska, spoke about his long journey from "the extreme conservative religious right wing on this debate to a committed solidarity with the opposite pole."
Dr. Davis said that he had watched patients struggle with major depression over their sexual and gender identity. "I am not proud of the fact that I am a late adopter on this important issue," he said, urging his fellow delegates that "we need to boldly make a decision and move forward."
But opponents said that the AAFP should not be wading so deeply into what they considered largely treacherous political waters.
Dr. Justin V. Bartos, a delegate from Texas, said that his state’s chapter opposed the resolution because it was too much of a political statement and diverged from the AAFP’s mission.
Members of the Texas Academy of Family Physicians "do not want us to endorse such a politically polarizing issue," Dr. Bartos said. He noted that family doctors in Texas are struggling to get their state legislature to restore funding for community-based residencies, and that lawmakers in the largely conservative state would note the AAFP’s support of gay marriage.
The Tennessee delegation also opposed the policy change for the same reasons – that it would politicize the AAFP.
Another Texas delegate, Dr. Erica Swegler, said that current AAFP policy was enough to indicate the organization’s support for equal access to health care for all Americans*.
During testimony before the reference committee charged with weighing the policy change, an Arkansas delegate said that more than 300 members might leave the AAFP if it gave its approval to gay marriage.
The AAFP policy will now state that the organization supports "civil marriage for same-gender couples to contribute to overall health and longevity, improved family stability and to benefit children of gay, lesbian, bisexual, transgender (GLBT) families."
That language was crafted in two resolutions brought forward by the resident and student sections. The AAFP joins the American Psychiatric Association and the American Psychological Association in supporting same-sex marriage. The psychiatrists approved a resolution of support in 2005, and the psychology association made it official policy in 2011.
The American Medical Association has not backed gay marriage specifically, but has a host of policies supporting equality for same-sex households.
The American College of Obstetricians and Gynecologists in 2009 issued a policy statement that same-sex couples should get the same legal rights as married heterosexuals.
Staying the Course on RUC
Many AAFP members have been dissatisfied with the RUC’s handling of primary care valuations and family medicine valuations in particular. Several resolutions were put forward at the congress to create a family medicine–specific set of codes and to make the AAFP the exclusive body for establishing billing codes for preventive and primary care services for Medicare.
The resolutions fostered some discussion – but most of it was fairly muted. The delegates, in the end, decided to instruct the organization to continue its efforts to seek higher valuations for primary care.
"This congress is very familiar with family medicine’s frustration with the RUC," said Dr. Glen Stream, whose term as AAFP president ended at the meeting. But he said that for the moment, "remaining in the RUC allows the opportunity to advocate and press for [the] changes" that the AAFP has requested.
Among those: creating primary care–specific E&M codes, valuing the codes to reflect the intensity and complexity of primary care, and paying for certain non-face-to-face services.
The AAFP has also hired Avalere Health to come up with data to support higher valuations, which the academy will take to the Centers for Medicare and Medicaid Services to use as a basis for the 2014 Medicare Physician Fee Schedule, said Dr. Stream, who now serves as AAFP board chair.
In Other Issues
The Congress of Delegates also approved a controversial resolution to end age restrictions on over-the-counter emergency contraception (EC). Under current law, women under age 16 must get a prescription to get EC. Supporters said that there is no scientific basis to prohibit younger women from getting the drug over the counter, and that it could help avert many abortions. Opponents, however, said that OTC availability would lead to fewer counseling opportunities with those young patients, and might lead to even greater sexual activity.
Among other resolutions, a proposal to oppose telemedicine when it is provided in the absence of a preexisting physician-patient relationship, was referred to the board of directors for study.
The board will also report back to the congress next year on developing model legislation that would exempt primary physicians from antitrust laws so they can collectively negotiate with health insurers. The goal is to help independent practices survive in the changing health delivery system.
Much discussion also centered on how to advance the patient-centered medical home model of care, including which, or how many organizations should certify the entities.
Dr. Douglas Henley, AAFP executive vice president, said that the medical home is the perfect environment to help control health costs and improve quality of care. The family physician can help prevent patients from becoming victims of unnecessary care, he said. Dr. Henley coined a new acronym to describe those patients: VOMIT, or "victims of multiple interventional technologies."
Finally, the AAFP leadership said that the board had given approval to generate a new report on what family medicine should look like over the next decade. The AAFP developed just such a blueprint 8 years ago; however, the research supporting that paper has become dated, said Dr. Henley.
"Now is a critical time to begin a discussion and perhaps revisit research or even initiate new research about what comprehensive primary care means in 2012 and into the future," said Dr. Henley.
That process has begun, he said, adding that AAFP members should expect to hear more in January.
*Correction, 11/06/12: A previous version of this story misstated the number of delegates and alternates at the meeting and misstated Dr. Erica Swegler's last name. The photo caption also misstated the name of the governing body.
**Correction, 12/10/12: A previous version of this story misstated Dr. Van Winkle's relation to Christopher.
PHILADELPHIA – The American Academy of Family Physicians concluded its 3-day Congress of Delegates meeting with a bold move forward – but it was probably not in an area that most members were expecting.
Debate among the 223 delegates and alternates regarding the academy’s participation in the American Medical Association’s Relative Value Scale Update Committee (RUC) ended up taking a backseat to controversy over resolutions urging the organization to endorse same-sex civil marriage*. In the end, delegates decided to maintain the status quo on the AAFP’s RUC efforts, but ended a 3-year gridlock over same-sex civil marriage by endorsing it by a vote of 75 to 44.
Debate over civil marriage for same-sex couples was intense and heated, with at least an hour of impassioned discussion at the Reference Committee on Advocacy, and close to that on the floor before the delegates voted to close debate.
Many supporters testified that their own same-sex partnerships had suffered as a result of their being denied the ability to marry.
Dr. Mark Dressner, president-elect of the California chapter, said that he felt that he and his partner had been treated as second-class citizens. Others spoke of the emotional and physical damage they had witnessed in patients and family members as a result of discrimination against homosexuals.
Dr. Lloyd Van Winkle of Castroville, Tex., who was just elected to the AAFP board, said that his cousin's son Christopher committed suicide at age 19 because he felt persecuted about his homosexuality.**
Dr. Paul W. Davis, a delegate from Alaska, spoke about his long journey from "the extreme conservative religious right wing on this debate to a committed solidarity with the opposite pole."
Dr. Davis said that he had watched patients struggle with major depression over their sexual and gender identity. "I am not proud of the fact that I am a late adopter on this important issue," he said, urging his fellow delegates that "we need to boldly make a decision and move forward."
But opponents said that the AAFP should not be wading so deeply into what they considered largely treacherous political waters.
Dr. Justin V. Bartos, a delegate from Texas, said that his state’s chapter opposed the resolution because it was too much of a political statement and diverged from the AAFP’s mission.
Members of the Texas Academy of Family Physicians "do not want us to endorse such a politically polarizing issue," Dr. Bartos said. He noted that family doctors in Texas are struggling to get their state legislature to restore funding for community-based residencies, and that lawmakers in the largely conservative state would note the AAFP’s support of gay marriage.
The Tennessee delegation also opposed the policy change for the same reasons – that it would politicize the AAFP.
Another Texas delegate, Dr. Erica Swegler, said that current AAFP policy was enough to indicate the organization’s support for equal access to health care for all Americans*.
During testimony before the reference committee charged with weighing the policy change, an Arkansas delegate said that more than 300 members might leave the AAFP if it gave its approval to gay marriage.
The AAFP policy will now state that the organization supports "civil marriage for same-gender couples to contribute to overall health and longevity, improved family stability and to benefit children of gay, lesbian, bisexual, transgender (GLBT) families."
That language was crafted in two resolutions brought forward by the resident and student sections. The AAFP joins the American Psychiatric Association and the American Psychological Association in supporting same-sex marriage. The psychiatrists approved a resolution of support in 2005, and the psychology association made it official policy in 2011.
The American Medical Association has not backed gay marriage specifically, but has a host of policies supporting equality for same-sex households.
The American College of Obstetricians and Gynecologists in 2009 issued a policy statement that same-sex couples should get the same legal rights as married heterosexuals.
Staying the Course on RUC
Many AAFP members have been dissatisfied with the RUC’s handling of primary care valuations and family medicine valuations in particular. Several resolutions were put forward at the congress to create a family medicine–specific set of codes and to make the AAFP the exclusive body for establishing billing codes for preventive and primary care services for Medicare.
The resolutions fostered some discussion – but most of it was fairly muted. The delegates, in the end, decided to instruct the organization to continue its efforts to seek higher valuations for primary care.
"This congress is very familiar with family medicine’s frustration with the RUC," said Dr. Glen Stream, whose term as AAFP president ended at the meeting. But he said that for the moment, "remaining in the RUC allows the opportunity to advocate and press for [the] changes" that the AAFP has requested.
Among those: creating primary care–specific E&M codes, valuing the codes to reflect the intensity and complexity of primary care, and paying for certain non-face-to-face services.
The AAFP has also hired Avalere Health to come up with data to support higher valuations, which the academy will take to the Centers for Medicare and Medicaid Services to use as a basis for the 2014 Medicare Physician Fee Schedule, said Dr. Stream, who now serves as AAFP board chair.
In Other Issues
The Congress of Delegates also approved a controversial resolution to end age restrictions on over-the-counter emergency contraception (EC). Under current law, women under age 16 must get a prescription to get EC. Supporters said that there is no scientific basis to prohibit younger women from getting the drug over the counter, and that it could help avert many abortions. Opponents, however, said that OTC availability would lead to fewer counseling opportunities with those young patients, and might lead to even greater sexual activity.
Among other resolutions, a proposal to oppose telemedicine when it is provided in the absence of a preexisting physician-patient relationship, was referred to the board of directors for study.
The board will also report back to the congress next year on developing model legislation that would exempt primary physicians from antitrust laws so they can collectively negotiate with health insurers. The goal is to help independent practices survive in the changing health delivery system.
Much discussion also centered on how to advance the patient-centered medical home model of care, including which, or how many organizations should certify the entities.
Dr. Douglas Henley, AAFP executive vice president, said that the medical home is the perfect environment to help control health costs and improve quality of care. The family physician can help prevent patients from becoming victims of unnecessary care, he said. Dr. Henley coined a new acronym to describe those patients: VOMIT, or "victims of multiple interventional technologies."
Finally, the AAFP leadership said that the board had given approval to generate a new report on what family medicine should look like over the next decade. The AAFP developed just such a blueprint 8 years ago; however, the research supporting that paper has become dated, said Dr. Henley.
"Now is a critical time to begin a discussion and perhaps revisit research or even initiate new research about what comprehensive primary care means in 2012 and into the future," said Dr. Henley.
That process has begun, he said, adding that AAFP members should expect to hear more in January.
*Correction, 11/06/12: A previous version of this story misstated the number of delegates and alternates at the meeting and misstated Dr. Erica Swegler's last name. The photo caption also misstated the name of the governing body.
**Correction, 12/10/12: A previous version of this story misstated Dr. Van Winkle's relation to Christopher.
PHILADELPHIA – The American Academy of Family Physicians concluded its 3-day Congress of Delegates meeting with a bold move forward – but it was probably not in an area that most members were expecting.
Debate among the 223 delegates and alternates regarding the academy’s participation in the American Medical Association’s Relative Value Scale Update Committee (RUC) ended up taking a backseat to controversy over resolutions urging the organization to endorse same-sex civil marriage*. In the end, delegates decided to maintain the status quo on the AAFP’s RUC efforts, but ended a 3-year gridlock over same-sex civil marriage by endorsing it by a vote of 75 to 44.
Debate over civil marriage for same-sex couples was intense and heated, with at least an hour of impassioned discussion at the Reference Committee on Advocacy, and close to that on the floor before the delegates voted to close debate.
Many supporters testified that their own same-sex partnerships had suffered as a result of their being denied the ability to marry.
Dr. Mark Dressner, president-elect of the California chapter, said that he felt that he and his partner had been treated as second-class citizens. Others spoke of the emotional and physical damage they had witnessed in patients and family members as a result of discrimination against homosexuals.
Dr. Lloyd Van Winkle of Castroville, Tex., who was just elected to the AAFP board, said that his cousin's son Christopher committed suicide at age 19 because he felt persecuted about his homosexuality.**
Dr. Paul W. Davis, a delegate from Alaska, spoke about his long journey from "the extreme conservative religious right wing on this debate to a committed solidarity with the opposite pole."
Dr. Davis said that he had watched patients struggle with major depression over their sexual and gender identity. "I am not proud of the fact that I am a late adopter on this important issue," he said, urging his fellow delegates that "we need to boldly make a decision and move forward."
But opponents said that the AAFP should not be wading so deeply into what they considered largely treacherous political waters.
Dr. Justin V. Bartos, a delegate from Texas, said that his state’s chapter opposed the resolution because it was too much of a political statement and diverged from the AAFP’s mission.
Members of the Texas Academy of Family Physicians "do not want us to endorse such a politically polarizing issue," Dr. Bartos said. He noted that family doctors in Texas are struggling to get their state legislature to restore funding for community-based residencies, and that lawmakers in the largely conservative state would note the AAFP’s support of gay marriage.
The Tennessee delegation also opposed the policy change for the same reasons – that it would politicize the AAFP.
Another Texas delegate, Dr. Erica Swegler, said that current AAFP policy was enough to indicate the organization’s support for equal access to health care for all Americans*.
During testimony before the reference committee charged with weighing the policy change, an Arkansas delegate said that more than 300 members might leave the AAFP if it gave its approval to gay marriage.
The AAFP policy will now state that the organization supports "civil marriage for same-gender couples to contribute to overall health and longevity, improved family stability and to benefit children of gay, lesbian, bisexual, transgender (GLBT) families."
That language was crafted in two resolutions brought forward by the resident and student sections. The AAFP joins the American Psychiatric Association and the American Psychological Association in supporting same-sex marriage. The psychiatrists approved a resolution of support in 2005, and the psychology association made it official policy in 2011.
The American Medical Association has not backed gay marriage specifically, but has a host of policies supporting equality for same-sex households.
The American College of Obstetricians and Gynecologists in 2009 issued a policy statement that same-sex couples should get the same legal rights as married heterosexuals.
Staying the Course on RUC
Many AAFP members have been dissatisfied with the RUC’s handling of primary care valuations and family medicine valuations in particular. Several resolutions were put forward at the congress to create a family medicine–specific set of codes and to make the AAFP the exclusive body for establishing billing codes for preventive and primary care services for Medicare.
The resolutions fostered some discussion – but most of it was fairly muted. The delegates, in the end, decided to instruct the organization to continue its efforts to seek higher valuations for primary care.
"This congress is very familiar with family medicine’s frustration with the RUC," said Dr. Glen Stream, whose term as AAFP president ended at the meeting. But he said that for the moment, "remaining in the RUC allows the opportunity to advocate and press for [the] changes" that the AAFP has requested.
Among those: creating primary care–specific E&M codes, valuing the codes to reflect the intensity and complexity of primary care, and paying for certain non-face-to-face services.
The AAFP has also hired Avalere Health to come up with data to support higher valuations, which the academy will take to the Centers for Medicare and Medicaid Services to use as a basis for the 2014 Medicare Physician Fee Schedule, said Dr. Stream, who now serves as AAFP board chair.
In Other Issues
The Congress of Delegates also approved a controversial resolution to end age restrictions on over-the-counter emergency contraception (EC). Under current law, women under age 16 must get a prescription to get EC. Supporters said that there is no scientific basis to prohibit younger women from getting the drug over the counter, and that it could help avert many abortions. Opponents, however, said that OTC availability would lead to fewer counseling opportunities with those young patients, and might lead to even greater sexual activity.
Among other resolutions, a proposal to oppose telemedicine when it is provided in the absence of a preexisting physician-patient relationship, was referred to the board of directors for study.
The board will also report back to the congress next year on developing model legislation that would exempt primary physicians from antitrust laws so they can collectively negotiate with health insurers. The goal is to help independent practices survive in the changing health delivery system.
Much discussion also centered on how to advance the patient-centered medical home model of care, including which, or how many organizations should certify the entities.
Dr. Douglas Henley, AAFP executive vice president, said that the medical home is the perfect environment to help control health costs and improve quality of care. The family physician can help prevent patients from becoming victims of unnecessary care, he said. Dr. Henley coined a new acronym to describe those patients: VOMIT, or "victims of multiple interventional technologies."
Finally, the AAFP leadership said that the board had given approval to generate a new report on what family medicine should look like over the next decade. The AAFP developed just such a blueprint 8 years ago; however, the research supporting that paper has become dated, said Dr. Henley.
"Now is a critical time to begin a discussion and perhaps revisit research or even initiate new research about what comprehensive primary care means in 2012 and into the future," said Dr. Henley.
That process has begun, he said, adding that AAFP members should expect to hear more in January.
*Correction, 11/06/12: A previous version of this story misstated the number of delegates and alternates at the meeting and misstated Dr. Erica Swegler's last name. The photo caption also misstated the name of the governing body.
**Correction, 12/10/12: A previous version of this story misstated Dr. Van Winkle's relation to Christopher.
AT THE AAFP CONGRESS OF DELEGATES
Sequestration Will Hit Pay but Spare Incentives
Medicare physician pay would be cut by at least 2% in 2013 while some Affordable Care Act programs would be spared the automatic cuts called for under the Budget Control Act of 2011, according to a report from the federal Office of Management and Budget.
The report provides details on how to cut $1.2 trillion from the federal budget by 2021 and was mandated after the Joint Select Committee on Deficit Reduction failed to do the same. The process – known as sequestration – would cut funding for most federal agencies across the board.
The Office of Management and Budget (OMB) report noted that the estimates are preliminary; the true magnitude of the cuts won’t be known until sequestration actually occurs.
The report also notes that the White House firmly opposes the automatic cuts.
"Sequestration is a blunt and indiscriminate instrument. It is not the responsible way for our nation to achieve deficit reduction," according to the report. OMB called on Congress to avoid sequestration either by passing the President’s budget proposals or otherwise approving "a comprehensive and balanced deficit reduction package."
Under sequestration, there would be a 9.4% cut to defense discretionary funding, 10% to mandatory defense programs, 8.2% reduction to nondefense discretionary funding, and 7.6% to nondefense mandatory programs.
Medicare – including physician and hospital payment – is slated for a 2%, or $11 billion, cut in 2013 alone. The 2% reduction will continue each year through 2021.
The American Hospital Association, American Medical Association, and American Nurses Association in a joint statement said that as many as 766,000 jobs in health care and related fields could be lost by 2021 if the Medicare cuts are allowed to stand, in part because of the reduction in payments to physicians and hospitals. They also predicted a ripple effect, including reduced spending on goods and services by health care providers and organizations, as well as reduced household purchases by health care workers who lose their jobs.
"Hospitals’ ability to maintain the kind of access to services that their communities need is being threatened," AHA President and CEO Rich Umbdenstock said in a statement.
Dr. Jeremy A. Lazarus, president of the AMA, said, "Coupled with the looming 27% Medicare physician payment cut, this 2% sequester will hurt patient access to care and will inject more uncertainty into our Medicare system. We need stability in Medicare physician payment as we work to improve our nation’s Medicare payment and delivery system to promote high-quality, high-value, better-coordinated care to our patients."
Among other programs at the Center for Medicare and Medicaid Services that would take a hit under sequestration: health insurance exchange grants ($66 million); state demonstration grants ($40 million); fraud and abuse ($78 million); the prevention and public health fund ($76 million); the Office of the National Coordinator for Health Information Technology ($1 million); the Office of Inspector General ($5 million); and the Hospital Insurance Trust Fund ($5.6 billion).
Many programs would be exempt from sequestration, however, including the Children’s Health Insurance Program, Medicare and Medicaid incentive payments for participating in health IT programs, and the consumer operated and oriented plan and the pre-existing condition insurance plan, both of which were created by the ACA.
Food and Drug Administration funding would be cut $319 million, and the Centers for Disease Control and Prevention would see at least a $490 million budget reduction, with $14 million of that coming out of the World Trade Center Health Program Fund. Funding for the National Institutes of Health would be cut $2.6 billion.
Medicare physician pay would be cut by at least 2% in 2013 while some Affordable Care Act programs would be spared the automatic cuts called for under the Budget Control Act of 2011, according to a report from the federal Office of Management and Budget.
The report provides details on how to cut $1.2 trillion from the federal budget by 2021 and was mandated after the Joint Select Committee on Deficit Reduction failed to do the same. The process – known as sequestration – would cut funding for most federal agencies across the board.
The Office of Management and Budget (OMB) report noted that the estimates are preliminary; the true magnitude of the cuts won’t be known until sequestration actually occurs.
The report also notes that the White House firmly opposes the automatic cuts.
"Sequestration is a blunt and indiscriminate instrument. It is not the responsible way for our nation to achieve deficit reduction," according to the report. OMB called on Congress to avoid sequestration either by passing the President’s budget proposals or otherwise approving "a comprehensive and balanced deficit reduction package."
Under sequestration, there would be a 9.4% cut to defense discretionary funding, 10% to mandatory defense programs, 8.2% reduction to nondefense discretionary funding, and 7.6% to nondefense mandatory programs.
Medicare – including physician and hospital payment – is slated for a 2%, or $11 billion, cut in 2013 alone. The 2% reduction will continue each year through 2021.
The American Hospital Association, American Medical Association, and American Nurses Association in a joint statement said that as many as 766,000 jobs in health care and related fields could be lost by 2021 if the Medicare cuts are allowed to stand, in part because of the reduction in payments to physicians and hospitals. They also predicted a ripple effect, including reduced spending on goods and services by health care providers and organizations, as well as reduced household purchases by health care workers who lose their jobs.
"Hospitals’ ability to maintain the kind of access to services that their communities need is being threatened," AHA President and CEO Rich Umbdenstock said in a statement.
Dr. Jeremy A. Lazarus, president of the AMA, said, "Coupled with the looming 27% Medicare physician payment cut, this 2% sequester will hurt patient access to care and will inject more uncertainty into our Medicare system. We need stability in Medicare physician payment as we work to improve our nation’s Medicare payment and delivery system to promote high-quality, high-value, better-coordinated care to our patients."
Among other programs at the Center for Medicare and Medicaid Services that would take a hit under sequestration: health insurance exchange grants ($66 million); state demonstration grants ($40 million); fraud and abuse ($78 million); the prevention and public health fund ($76 million); the Office of the National Coordinator for Health Information Technology ($1 million); the Office of Inspector General ($5 million); and the Hospital Insurance Trust Fund ($5.6 billion).
Many programs would be exempt from sequestration, however, including the Children’s Health Insurance Program, Medicare and Medicaid incentive payments for participating in health IT programs, and the consumer operated and oriented plan and the pre-existing condition insurance plan, both of which were created by the ACA.
Food and Drug Administration funding would be cut $319 million, and the Centers for Disease Control and Prevention would see at least a $490 million budget reduction, with $14 million of that coming out of the World Trade Center Health Program Fund. Funding for the National Institutes of Health would be cut $2.6 billion.
Medicare physician pay would be cut by at least 2% in 2013 while some Affordable Care Act programs would be spared the automatic cuts called for under the Budget Control Act of 2011, according to a report from the federal Office of Management and Budget.
The report provides details on how to cut $1.2 trillion from the federal budget by 2021 and was mandated after the Joint Select Committee on Deficit Reduction failed to do the same. The process – known as sequestration – would cut funding for most federal agencies across the board.
The Office of Management and Budget (OMB) report noted that the estimates are preliminary; the true magnitude of the cuts won’t be known until sequestration actually occurs.
The report also notes that the White House firmly opposes the automatic cuts.
"Sequestration is a blunt and indiscriminate instrument. It is not the responsible way for our nation to achieve deficit reduction," according to the report. OMB called on Congress to avoid sequestration either by passing the President’s budget proposals or otherwise approving "a comprehensive and balanced deficit reduction package."
Under sequestration, there would be a 9.4% cut to defense discretionary funding, 10% to mandatory defense programs, 8.2% reduction to nondefense discretionary funding, and 7.6% to nondefense mandatory programs.
Medicare – including physician and hospital payment – is slated for a 2%, or $11 billion, cut in 2013 alone. The 2% reduction will continue each year through 2021.
The American Hospital Association, American Medical Association, and American Nurses Association in a joint statement said that as many as 766,000 jobs in health care and related fields could be lost by 2021 if the Medicare cuts are allowed to stand, in part because of the reduction in payments to physicians and hospitals. They also predicted a ripple effect, including reduced spending on goods and services by health care providers and organizations, as well as reduced household purchases by health care workers who lose their jobs.
"Hospitals’ ability to maintain the kind of access to services that their communities need is being threatened," AHA President and CEO Rich Umbdenstock said in a statement.
Dr. Jeremy A. Lazarus, president of the AMA, said, "Coupled with the looming 27% Medicare physician payment cut, this 2% sequester will hurt patient access to care and will inject more uncertainty into our Medicare system. We need stability in Medicare physician payment as we work to improve our nation’s Medicare payment and delivery system to promote high-quality, high-value, better-coordinated care to our patients."
Among other programs at the Center for Medicare and Medicaid Services that would take a hit under sequestration: health insurance exchange grants ($66 million); state demonstration grants ($40 million); fraud and abuse ($78 million); the prevention and public health fund ($76 million); the Office of the National Coordinator for Health Information Technology ($1 million); the Office of Inspector General ($5 million); and the Hospital Insurance Trust Fund ($5.6 billion).
Many programs would be exempt from sequestration, however, including the Children’s Health Insurance Program, Medicare and Medicaid incentive payments for participating in health IT programs, and the consumer operated and oriented plan and the pre-existing condition insurance plan, both of which were created by the ACA.
Food and Drug Administration funding would be cut $319 million, and the Centers for Disease Control and Prevention would see at least a $490 million budget reduction, with $14 million of that coming out of the World Trade Center Health Program Fund. Funding for the National Institutes of Health would be cut $2.6 billion.
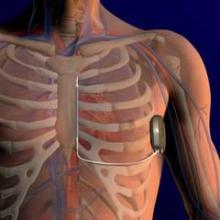
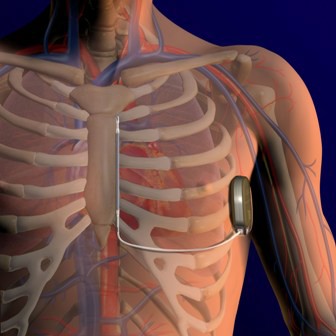
FDA Expands Sapien Approval
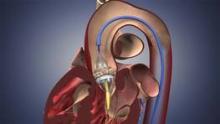
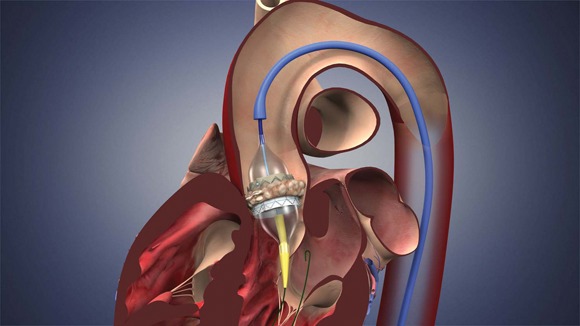
The Food and Drug Administration has expanded approval for the Sapien transcatheter heart valve for patients with aortic valve stenosis who are at high risk for serious surgical complications or death.
It is the second approval for the device, which is made by Edwards Lifesciences Corp. of Irvine, Calif. In November 2011, the FDA approved it for severe aortic valve stenosis in inoperable patients.
The indication for high-risk patients was reviewed by an FDA advisory panel in July. The majority of the panel indicated that the device was safe and effective in that group.
Both approvals were based on results from the PARTNER trial (N. Engl. J. Med. 2010;363:1597-607).
"Any procedure to replace the aortic valve carries the risk for serious complications, but for some patients with coexisting conditions or diseases, that risk may be especially high," Christy Foreman, director of the office of device evaluation at FDA’s Center for Devices and Radiological Health, said in a statement.
"The THV serves as an alternative for some very high-risk patients."
Larry L. Wood, Edwards’ corporate vice president, transcatheter heart valves, said in a statement: "It is extremely rewarding for us and our clinical partners to know that high-risk patients suffering from this often-debilitating disease will now have access to this life-saving therapy."
The Sapien THV is implanted at the site of the diseased valve using a catheter that is delivered either through a transfemoral or transapical approach. According to the Edwards statement, the transapical approach, which requires insertion through the ribs and myocardium, was not approved outside of clinical trials until now.
Transcatheter aortic valve replacement has become one of the driving forces in the heart valve market, which may hit $1.5 billion by 2016.
The new indication for the Sapien THV is expected to expand that market further. According to the FDA, the approved labeling indicates that a surgeon must determine whether a patient is eligible for the Sapien. But in fact, Edwards, the Centers for Medicare and Medicaid Services, and professional societies such as the American College of Cardiology and the Society of Thoracic Surgeons, have worked together to establish standards and requirements for "Heart Teams" that must be present during the procedure and evaluation of the patient by two cardiac surgeons.
There is an increased risk for major vascular complications and for stroke during the first month post implant. Thus, the device is contraindicated in patients who cannot tolerate anticoagulation/antiplatelet therapy.
Edwards will monitor safety and complication rates through the national Transcatheter Valve Therapy Registry.
The Food and Drug Administration has expanded approval for the Sapien transcatheter heart valve for patients with aortic valve stenosis who are at high risk for serious surgical complications or death.
It is the second approval for the device, which is made by Edwards Lifesciences Corp. of Irvine, Calif. In November 2011, the FDA approved it for severe aortic valve stenosis in inoperable patients.
The indication for high-risk patients was reviewed by an FDA advisory panel in July. The majority of the panel indicated that the device was safe and effective in that group.
Both approvals were based on results from the PARTNER trial (N. Engl. J. Med. 2010;363:1597-607).
"Any procedure to replace the aortic valve carries the risk for serious complications, but for some patients with coexisting conditions or diseases, that risk may be especially high," Christy Foreman, director of the office of device evaluation at FDA’s Center for Devices and Radiological Health, said in a statement.
"The THV serves as an alternative for some very high-risk patients."
Larry L. Wood, Edwards’ corporate vice president, transcatheter heart valves, said in a statement: "It is extremely rewarding for us and our clinical partners to know that high-risk patients suffering from this often-debilitating disease will now have access to this life-saving therapy."
The Sapien THV is implanted at the site of the diseased valve using a catheter that is delivered either through a transfemoral or transapical approach. According to the Edwards statement, the transapical approach, which requires insertion through the ribs and myocardium, was not approved outside of clinical trials until now.
Transcatheter aortic valve replacement has become one of the driving forces in the heart valve market, which may hit $1.5 billion by 2016.
The new indication for the Sapien THV is expected to expand that market further. According to the FDA, the approved labeling indicates that a surgeon must determine whether a patient is eligible for the Sapien. But in fact, Edwards, the Centers for Medicare and Medicaid Services, and professional societies such as the American College of Cardiology and the Society of Thoracic Surgeons, have worked together to establish standards and requirements for "Heart Teams" that must be present during the procedure and evaluation of the patient by two cardiac surgeons.
There is an increased risk for major vascular complications and for stroke during the first month post implant. Thus, the device is contraindicated in patients who cannot tolerate anticoagulation/antiplatelet therapy.
Edwards will monitor safety and complication rates through the national Transcatheter Valve Therapy Registry.
The Food and Drug Administration has expanded approval for the Sapien transcatheter heart valve for patients with aortic valve stenosis who are at high risk for serious surgical complications or death.
It is the second approval for the device, which is made by Edwards Lifesciences Corp. of Irvine, Calif. In November 2011, the FDA approved it for severe aortic valve stenosis in inoperable patients.
The indication for high-risk patients was reviewed by an FDA advisory panel in July. The majority of the panel indicated that the device was safe and effective in that group.
Both approvals were based on results from the PARTNER trial (N. Engl. J. Med. 2010;363:1597-607).
"Any procedure to replace the aortic valve carries the risk for serious complications, but for some patients with coexisting conditions or diseases, that risk may be especially high," Christy Foreman, director of the office of device evaluation at FDA’s Center for Devices and Radiological Health, said in a statement.
"The THV serves as an alternative for some very high-risk patients."
Larry L. Wood, Edwards’ corporate vice president, transcatheter heart valves, said in a statement: "It is extremely rewarding for us and our clinical partners to know that high-risk patients suffering from this often-debilitating disease will now have access to this life-saving therapy."
The Sapien THV is implanted at the site of the diseased valve using a catheter that is delivered either through a transfemoral or transapical approach. According to the Edwards statement, the transapical approach, which requires insertion through the ribs and myocardium, was not approved outside of clinical trials until now.
Transcatheter aortic valve replacement has become one of the driving forces in the heart valve market, which may hit $1.5 billion by 2016.
The new indication for the Sapien THV is expected to expand that market further. According to the FDA, the approved labeling indicates that a surgeon must determine whether a patient is eligible for the Sapien. But in fact, Edwards, the Centers for Medicare and Medicaid Services, and professional societies such as the American College of Cardiology and the Society of Thoracic Surgeons, have worked together to establish standards and requirements for "Heart Teams" that must be present during the procedure and evaluation of the patient by two cardiac surgeons.
There is an increased risk for major vascular complications and for stroke during the first month post implant. Thus, the device is contraindicated in patients who cannot tolerate anticoagulation/antiplatelet therapy.
Edwards will monitor safety and complication rates through the national Transcatheter Valve Therapy Registry.
ED to Wards: Beating the Boarding Problem
Boarding has been a growing problem over the last decade and may worsen – at least in the short term – with the fuller implementation of the Affordable Care Act, according to experts who are calling for hospitals to implement proven remedies.
Hospital efficiency has suffered as institutions seek to cut costs while coping with a rising influx of uninsured and Medicaid patients. That financial pressure will not improve under health care reform, at least not initially, as millions more Medicaid patients are likely to seek care in the ED when they cannot access primary care, Dr. Jody Crane, emergency department faculty for the Institute for Healthcare Improvement, Cambridge, Mass., said in an interview.
Medicaid pay is already low, and an estimated 8 million more Medicaid patients will visit EDs annually after reform. In addition, as Medicare dials back its reimbursement to hospitals and physicians to help fund the Affordable Care Act, Va. That will put a squeeze on already tight hospital margins, he said.
"Every hospital is going to have to do the same or more with less," said Dr. Crane. Boarding – holding patients in the ED while they wait for admission to inpatient services – can be costly, reduces patient satisfaction, and has been shown to ultimately increase length of stay and mortality.
"Boarding in some hospitals is a capacity issue, but in others it’s also a management issue," Dr. Crane pointed out. Technical issues, including matching resources to the demand coming through the door, are also factors, he said.
"Ultimately, a coordinated effort is needed between the ED and the hospitalist to ensure quality transitions of care that maximize both health and financial outcomes," said Dr. Franklin A. Michota, director of academic affairs in the department of hospital medicine at the Cleveland Clinic. "Each hospital does need to analyze where their bottlenecks exist and whether a staffing solution is part of the answer. But the ED also needs to look at the accuracy and quality of their triage and their ability to adequately prepare the patient for the trip upstairs."
"For example," Dr. Michota continued, "sending a patient upstairs with sickle cell crisis and inadequate pain control just ties up the hospitalist unnecessarily and will delay the hospitalist in getting to the next new admission. Or failing to order a CT scan that would have sent a patient to the operating room and bypass the medical hospitalist altogether will again just delay getting to the next admission.
"Everything relies on everything else....the chain is only as strong as its weakest link."
Indeed, "the primary reason it’s a hard problem to fix is because it’s so multifactorial," said Dr. Jesse Pines, associate professor of emergency medicine and health policy at George Washington University in Washington, D.C.
What Works?
A recent study in Health Affairs showed that many hospitals still are not taking advantage of proven strategies to reduce boarding – among them, so-called smoothing of elective surgical schedules, active bed management by hospitalists, and the "full capacity protocol" that was developed by Dr. Peter Viccellio and his colleagues at the State University of New York at Stony Brook.
"This paper is another in a long series of wake-up calls about the dangerous practice of ‘boarding’ admitted patients in [emergency departments]," ACEP President Dr. David Seaberg said in a statement. "I just hope that hospital administrators and legislators smell the coffee. Their leadership is critical to changing the flow of patients from the front door of the emergency department to the inpatient floors of the hospital."
Emergency departments and hospitals can work together to find ways to improve efficiency, but several important ingredients are needed, Dr. Pines said. The Health Affairs article (2012;31:1757-66 [doi: 10.1377/hlthaff.2011.0786]) cites certain protocols that work, "but you need people to make them work," he said. "You need a strong leader in the ED who can get buy-in from the emergency group. ... You also need a supportive administration and medical staff to make it happen."
It is important to have a local quality improvement group whose members can look at the overall process by which patients get moved from the ED to inpatient services, and examine how patients are managed in the ED after they are admitted, Dr. Pines said. After the existing process is analyzed, hospital staff can identify and address the inefficiencies causing the delays.
The inefficiencies are highly particular to each facility, and often to each day and each shift in the ED and on inpatient wards, he said.
Staffing shortfalls – especially inpatient nursing staff – can be a major contributor to bottlenecks, Dr. Crane said. In response to financial pressures, many hospitals are reducing nursing staff, resulting in increased nurse-to-bed ratios. "That prolongs the inpatient length of stay," he said, because it leads to more work per nurse and more delays in admitting and discharging patients.
There has been a recent administrative trend toward real-time capacity management,which can be an effective way to accommodate the variation in admissions over time.. With this approach, if there is not an identified demand for nurses, they can be sent home or told not to come to work.This ability to change staffing levels according to real-time needs can be an effective way to accommodate the variation in admissions over time
Handoffs and Beds
Dr. Crane contends there are three specific elements to improving patient flow: the physician handoff, the nursing handoff, and bed efficiency.
The physician handoff can be made more efficient by optimizing the coordination between hospitalists and emergency physicians. Due to the high variation in arrival of admissions from the ED and the low numbers of admitting hospitalists at certain times, patients can queue in the ED awaiting admission orders, Dr. Crane noted.
So now, many emergency physicians write holding orders – which have an expiration time – until the patient can be admitted. Holding orders allow the patient to be moved out of the ED, Dr. Crane said. Until recently, emergency physicians have been reluctant to write holding orders, but this trend has reversed because it can be dramatically effective.ACEP now endorses the practice and notes that any associated risk is limited.
The unpredictability of admissions "creates all kinds of logistical nightmares handing off from the emergency department to the floor nurse," Dr. Crane said. Just adding more nurses is not necessarily the answer, and, in general, hospitals don’t have the money to add staff. Instead, hospitals need to better align existing resources and staff. For instance, nurses should probably not be transporting patients or cleaning rooms. That can be done with lower-priced and less-resource-intensive staff, Dr. Crane said.
Bed capacity is the third major element that hospitals should address to reduce boarding, according to Dr. Crane. Demand from the ED cannot be controlled, but on the inpatient side, hospitals can "smooth" the elective surgery schedule by moving more procedures to the middle and end of the week so that all the beds aren’t occupied early in the week. And that is not necessarily easy: "When you change block schedules, you get into cultural issues" with surgeons who do not want to have to see patients on the weekends, Dr. Crane said.
There are also practical issues limiting weekend care. Namely, the typical services such as physical therapy and case management are not routinely present on the weekends. As such, patients who could otherwise be discharged stay in their beds until Monday, when bed demands from the ED and OR increase dramatically. Bed capacity could be improved if hospitals can find ways to make services like testing and therapy available on weekends, and find surgeons and other consultants who will round on Saturdays and Sundays. That might mean hiring a physician’s assistant or other midlevel provider to cover.
Observation Units
Hospitals have also started making use of "observation" status for patients or adding observation units.
At Mary Washington Hospital, where Dr. Crane practices, a team of physicians, nurses, and administrators is designing a streamlined protocol for observation patients. Instead of the standard admission history and physical, an abbreviated assessment has been designed, incorporating use of the ED record and a brief assessment format. Consultations are reduced, and evidence-based pathways are being created for a variety of admitting diagnoses. Patients are treated as outpatients unless test results indicate they need to be moved to the inpatient side, and due to the reduced workload, nurses may be able to work comfortably with higher bed-to-nurse ratios.
The target is to hold patients for observation for 24 hours or less, Dr. Crane said.
Because observation patients are considered outpatients, insurers will pay the hospital less for the visit than if the patient is admitted, Dr. Pines noted, and it leaves the patient vulnerable to higher expenses than just an ED copay.
Another recent study in Health Affairs found that observation stays have been rapidly increasing – from 86.9 observation stays per 1,000 inpatient admissions in 2007 for Medicare beneficiaries to 116.6 in 2009 (Health Affairs 2012;31:1251-9). Of every 1,000 Medicare beneficiaries, 2 were under observation each month in 2007 and almost 3 were under observation each month in 2009. Meanwhile, inpatient admissions per 1,000 declined.
The Sharp Stick of Regulation
Voluntary efforts by hospitals may not be enough, said Dr. Sandra Schneider, former president of the American College of Emergency Physicians. Because hospitals are still paid for the patients who are boarded, they might not see the hidden costs of boarding, she said.
"We have to change that playing field if we want to get serious about [boarding]," Dr. Schneider said.
Among the game changers she cited are the increasing requirements for public reporting on quality measures, including those related to boarding.
Currently, hospitals get paid more by Medicare if they report certain inpatient quality measures. On the outpatient side, emergency departments report on the median time from ED arrival to ED departure for discharged patients and on patients who leave without being seen. In addition, it is now voluntary for hospitals to report on the average time patients spend in the ED before being admitted as an inpatient, and the average time patients spend in the ED after admission but before leaving the ED for their inpatient room.
Data from hospitals that have reported on these measures can be found on the Hospital Compare website.
The Joint Commission is also revisiting its standard on patient flow in the ED, which will go into effect in January 2013. Among other things, the commission is considering defining an episode of boarding as any case in which a patient remains in the ED 4 hours or more after a decision has been made to admit or transfer that patient.
Dr. Pines said that hospital administrators will be motivated both by public reporting imperatives and by Joint Commission standards. Dr. Crane, however, said that "until there are some significant incentives to moving patients upstairs in a timely manner, what’s going to drive senior executives is finance." He thinks that these new standards may help, but more incentives may be needed, such as penalties imposed if boarding limits are exceeded.
Australia, Canada, the United Kingdom, and other developed countries impose such limits and levy penalties.
Hospital Compare data – which will be more fully reported at the end of the first quarter of 2013 – “is going to show us once and for all where the median is and who are the outliers,” Dr. Schneider said. It’s hoped that the outliers will "realize they don’t just have a boarding problem – they have a serious boarding problem," she said.
"If not, then we may have to do what the rest of the world is doing."
For hospitals that are full (above 95% occupied) and have high surgical and critical care volume, boarding in the ED does not lose money – in fact, it does the opposite. Surgery and critical care are well reimbursed and most hospitals would be well served financially to eliminate medical admissions altogether and simply take care of surgery and critical care patients. Of course, not- for-profit hospitals (like the Cleveland Clinic) have a duty and obligation to serve the community at large, and we must maintain an open ED and provide inpatient medical care as part of our mission (and tax status).
The vast majority of ED admissions today are medical patients who are Medicare or Medicare plus Medicaid, Even if the hospital takes care of these patients efficiently and matches the DRG reimbursement with the length of stay, the opportunity cost of having a nursing home patient with the flu tie up a bed that could have been used for an open heart surgery or lap chole, etc., is huge.
It is often said that from a financial perspective, a medical patient is only slightly better than an empty bed. One of the major operational drivers for hospitalist groups is to maximize efficiency on the medical patient so that there are always beds available for an elective surgical patient. Of course, if a hospital is not full there would be a financial loss for putting aa medical patient in an empty bed that would otherwise go unfilled altogether.
Emergency departments everywhere need to recognize and understand that no one “wins” by replacing an ED problem with an inpatient ward problem. Boarding in either location is dangerous and leads to poor patient satisfaction. The notion that the patient is better off upstairs in the hallway is misguided and not well proven.
Geographical units (i.e., similar patients with specific nurse competencies or service assignments in close proximity) have been shown in the hospital medicine literature to improve efficiency and outcomes. As such, we do not want patients strewn about the hospital for the sake of eliminating an ED boarder if it will lead to a longer length of stay and further inefficiency upstairs. Then, a hospital that should be at 85% capacity will find itself at 90% and once again, and the ED will suffer soon enough too.
Franklin A. Michota, M.D. is medical editor of Hospitalist News and director of academic affairs in the department of hospital medicine at the Cleveland Clinic.
For hospitals that are full (above 95% occupied) and have high surgical and critical care volume, boarding in the ED does not lose money – in fact, it does the opposite. Surgery and critical care are well reimbursed and most hospitals would be well served financially to eliminate medical admissions altogether and simply take care of surgery and critical care patients. Of course, not- for-profit hospitals (like the Cleveland Clinic) have a duty and obligation to serve the community at large, and we must maintain an open ED and provide inpatient medical care as part of our mission (and tax status).
The vast majority of ED admissions today are medical patients who are Medicare or Medicare plus Medicaid, Even if the hospital takes care of these patients efficiently and matches the DRG reimbursement with the length of stay, the opportunity cost of having a nursing home patient with the flu tie up a bed that could have been used for an open heart surgery or lap chole, etc., is huge.
It is often said that from a financial perspective, a medical patient is only slightly better than an empty bed. One of the major operational drivers for hospitalist groups is to maximize efficiency on the medical patient so that there are always beds available for an elective surgical patient. Of course, if a hospital is not full there would be a financial loss for putting aa medical patient in an empty bed that would otherwise go unfilled altogether.
Emergency departments everywhere need to recognize and understand that no one “wins” by replacing an ED problem with an inpatient ward problem. Boarding in either location is dangerous and leads to poor patient satisfaction. The notion that the patient is better off upstairs in the hallway is misguided and not well proven.
Geographical units (i.e., similar patients with specific nurse competencies or service assignments in close proximity) have been shown in the hospital medicine literature to improve efficiency and outcomes. As such, we do not want patients strewn about the hospital for the sake of eliminating an ED boarder if it will lead to a longer length of stay and further inefficiency upstairs. Then, a hospital that should be at 85% capacity will find itself at 90% and once again, and the ED will suffer soon enough too.
Franklin A. Michota, M.D. is medical editor of Hospitalist News and director of academic affairs in the department of hospital medicine at the Cleveland Clinic.
For hospitals that are full (above 95% occupied) and have high surgical and critical care volume, boarding in the ED does not lose money – in fact, it does the opposite. Surgery and critical care are well reimbursed and most hospitals would be well served financially to eliminate medical admissions altogether and simply take care of surgery and critical care patients. Of course, not- for-profit hospitals (like the Cleveland Clinic) have a duty and obligation to serve the community at large, and we must maintain an open ED and provide inpatient medical care as part of our mission (and tax status).
The vast majority of ED admissions today are medical patients who are Medicare or Medicare plus Medicaid, Even if the hospital takes care of these patients efficiently and matches the DRG reimbursement with the length of stay, the opportunity cost of having a nursing home patient with the flu tie up a bed that could have been used for an open heart surgery or lap chole, etc., is huge.
It is often said that from a financial perspective, a medical patient is only slightly better than an empty bed. One of the major operational drivers for hospitalist groups is to maximize efficiency on the medical patient so that there are always beds available for an elective surgical patient. Of course, if a hospital is not full there would be a financial loss for putting aa medical patient in an empty bed that would otherwise go unfilled altogether.
Emergency departments everywhere need to recognize and understand that no one “wins” by replacing an ED problem with an inpatient ward problem. Boarding in either location is dangerous and leads to poor patient satisfaction. The notion that the patient is better off upstairs in the hallway is misguided and not well proven.
Geographical units (i.e., similar patients with specific nurse competencies or service assignments in close proximity) have been shown in the hospital medicine literature to improve efficiency and outcomes. As such, we do not want patients strewn about the hospital for the sake of eliminating an ED boarder if it will lead to a longer length of stay and further inefficiency upstairs. Then, a hospital that should be at 85% capacity will find itself at 90% and once again, and the ED will suffer soon enough too.
Franklin A. Michota, M.D. is medical editor of Hospitalist News and director of academic affairs in the department of hospital medicine at the Cleveland Clinic.
Boarding has been a growing problem over the last decade and may worsen – at least in the short term – with the fuller implementation of the Affordable Care Act, according to experts who are calling for hospitals to implement proven remedies.
Hospital efficiency has suffered as institutions seek to cut costs while coping with a rising influx of uninsured and Medicaid patients. That financial pressure will not improve under health care reform, at least not initially, as millions more Medicaid patients are likely to seek care in the ED when they cannot access primary care, Dr. Jody Crane, emergency department faculty for the Institute for Healthcare Improvement, Cambridge, Mass., said in an interview.
Medicaid pay is already low, and an estimated 8 million more Medicaid patients will visit EDs annually after reform. In addition, as Medicare dials back its reimbursement to hospitals and physicians to help fund the Affordable Care Act, Va. That will put a squeeze on already tight hospital margins, he said.
"Every hospital is going to have to do the same or more with less," said Dr. Crane. Boarding – holding patients in the ED while they wait for admission to inpatient services – can be costly, reduces patient satisfaction, and has been shown to ultimately increase length of stay and mortality.
"Boarding in some hospitals is a capacity issue, but in others it’s also a management issue," Dr. Crane pointed out. Technical issues, including matching resources to the demand coming through the door, are also factors, he said.
"Ultimately, a coordinated effort is needed between the ED and the hospitalist to ensure quality transitions of care that maximize both health and financial outcomes," said Dr. Franklin A. Michota, director of academic affairs in the department of hospital medicine at the Cleveland Clinic. "Each hospital does need to analyze where their bottlenecks exist and whether a staffing solution is part of the answer. But the ED also needs to look at the accuracy and quality of their triage and their ability to adequately prepare the patient for the trip upstairs."
"For example," Dr. Michota continued, "sending a patient upstairs with sickle cell crisis and inadequate pain control just ties up the hospitalist unnecessarily and will delay the hospitalist in getting to the next new admission. Or failing to order a CT scan that would have sent a patient to the operating room and bypass the medical hospitalist altogether will again just delay getting to the next admission.
"Everything relies on everything else....the chain is only as strong as its weakest link."
Indeed, "the primary reason it’s a hard problem to fix is because it’s so multifactorial," said Dr. Jesse Pines, associate professor of emergency medicine and health policy at George Washington University in Washington, D.C.
What Works?
A recent study in Health Affairs showed that many hospitals still are not taking advantage of proven strategies to reduce boarding – among them, so-called smoothing of elective surgical schedules, active bed management by hospitalists, and the "full capacity protocol" that was developed by Dr. Peter Viccellio and his colleagues at the State University of New York at Stony Brook.
"This paper is another in a long series of wake-up calls about the dangerous practice of ‘boarding’ admitted patients in [emergency departments]," ACEP President Dr. David Seaberg said in a statement. "I just hope that hospital administrators and legislators smell the coffee. Their leadership is critical to changing the flow of patients from the front door of the emergency department to the inpatient floors of the hospital."
Emergency departments and hospitals can work together to find ways to improve efficiency, but several important ingredients are needed, Dr. Pines said. The Health Affairs article (2012;31:1757-66 [doi: 10.1377/hlthaff.2011.0786]) cites certain protocols that work, "but you need people to make them work," he said. "You need a strong leader in the ED who can get buy-in from the emergency group. ... You also need a supportive administration and medical staff to make it happen."
It is important to have a local quality improvement group whose members can look at the overall process by which patients get moved from the ED to inpatient services, and examine how patients are managed in the ED after they are admitted, Dr. Pines said. After the existing process is analyzed, hospital staff can identify and address the inefficiencies causing the delays.
The inefficiencies are highly particular to each facility, and often to each day and each shift in the ED and on inpatient wards, he said.
Staffing shortfalls – especially inpatient nursing staff – can be a major contributor to bottlenecks, Dr. Crane said. In response to financial pressures, many hospitals are reducing nursing staff, resulting in increased nurse-to-bed ratios. "That prolongs the inpatient length of stay," he said, because it leads to more work per nurse and more delays in admitting and discharging patients.
There has been a recent administrative trend toward real-time capacity management,which can be an effective way to accommodate the variation in admissions over time.. With this approach, if there is not an identified demand for nurses, they can be sent home or told not to come to work.This ability to change staffing levels according to real-time needs can be an effective way to accommodate the variation in admissions over time
Handoffs and Beds
Dr. Crane contends there are three specific elements to improving patient flow: the physician handoff, the nursing handoff, and bed efficiency.
The physician handoff can be made more efficient by optimizing the coordination between hospitalists and emergency physicians. Due to the high variation in arrival of admissions from the ED and the low numbers of admitting hospitalists at certain times, patients can queue in the ED awaiting admission orders, Dr. Crane noted.
So now, many emergency physicians write holding orders – which have an expiration time – until the patient can be admitted. Holding orders allow the patient to be moved out of the ED, Dr. Crane said. Until recently, emergency physicians have been reluctant to write holding orders, but this trend has reversed because it can be dramatically effective.ACEP now endorses the practice and notes that any associated risk is limited.
The unpredictability of admissions "creates all kinds of logistical nightmares handing off from the emergency department to the floor nurse," Dr. Crane said. Just adding more nurses is not necessarily the answer, and, in general, hospitals don’t have the money to add staff. Instead, hospitals need to better align existing resources and staff. For instance, nurses should probably not be transporting patients or cleaning rooms. That can be done with lower-priced and less-resource-intensive staff, Dr. Crane said.
Bed capacity is the third major element that hospitals should address to reduce boarding, according to Dr. Crane. Demand from the ED cannot be controlled, but on the inpatient side, hospitals can "smooth" the elective surgery schedule by moving more procedures to the middle and end of the week so that all the beds aren’t occupied early in the week. And that is not necessarily easy: "When you change block schedules, you get into cultural issues" with surgeons who do not want to have to see patients on the weekends, Dr. Crane said.
There are also practical issues limiting weekend care. Namely, the typical services such as physical therapy and case management are not routinely present on the weekends. As such, patients who could otherwise be discharged stay in their beds until Monday, when bed demands from the ED and OR increase dramatically. Bed capacity could be improved if hospitals can find ways to make services like testing and therapy available on weekends, and find surgeons and other consultants who will round on Saturdays and Sundays. That might mean hiring a physician’s assistant or other midlevel provider to cover.
Observation Units
Hospitals have also started making use of "observation" status for patients or adding observation units.
At Mary Washington Hospital, where Dr. Crane practices, a team of physicians, nurses, and administrators is designing a streamlined protocol for observation patients. Instead of the standard admission history and physical, an abbreviated assessment has been designed, incorporating use of the ED record and a brief assessment format. Consultations are reduced, and evidence-based pathways are being created for a variety of admitting diagnoses. Patients are treated as outpatients unless test results indicate they need to be moved to the inpatient side, and due to the reduced workload, nurses may be able to work comfortably with higher bed-to-nurse ratios.
The target is to hold patients for observation for 24 hours or less, Dr. Crane said.
Because observation patients are considered outpatients, insurers will pay the hospital less for the visit than if the patient is admitted, Dr. Pines noted, and it leaves the patient vulnerable to higher expenses than just an ED copay.
Another recent study in Health Affairs found that observation stays have been rapidly increasing – from 86.9 observation stays per 1,000 inpatient admissions in 2007 for Medicare beneficiaries to 116.6 in 2009 (Health Affairs 2012;31:1251-9). Of every 1,000 Medicare beneficiaries, 2 were under observation each month in 2007 and almost 3 were under observation each month in 2009. Meanwhile, inpatient admissions per 1,000 declined.
The Sharp Stick of Regulation
Voluntary efforts by hospitals may not be enough, said Dr. Sandra Schneider, former president of the American College of Emergency Physicians. Because hospitals are still paid for the patients who are boarded, they might not see the hidden costs of boarding, she said.
"We have to change that playing field if we want to get serious about [boarding]," Dr. Schneider said.
Among the game changers she cited are the increasing requirements for public reporting on quality measures, including those related to boarding.
Currently, hospitals get paid more by Medicare if they report certain inpatient quality measures. On the outpatient side, emergency departments report on the median time from ED arrival to ED departure for discharged patients and on patients who leave without being seen. In addition, it is now voluntary for hospitals to report on the average time patients spend in the ED before being admitted as an inpatient, and the average time patients spend in the ED after admission but before leaving the ED for their inpatient room.
Data from hospitals that have reported on these measures can be found on the Hospital Compare website.
The Joint Commission is also revisiting its standard on patient flow in the ED, which will go into effect in January 2013. Among other things, the commission is considering defining an episode of boarding as any case in which a patient remains in the ED 4 hours or more after a decision has been made to admit or transfer that patient.
Dr. Pines said that hospital administrators will be motivated both by public reporting imperatives and by Joint Commission standards. Dr. Crane, however, said that "until there are some significant incentives to moving patients upstairs in a timely manner, what’s going to drive senior executives is finance." He thinks that these new standards may help, but more incentives may be needed, such as penalties imposed if boarding limits are exceeded.
Australia, Canada, the United Kingdom, and other developed countries impose such limits and levy penalties.
Hospital Compare data – which will be more fully reported at the end of the first quarter of 2013 – “is going to show us once and for all where the median is and who are the outliers,” Dr. Schneider said. It’s hoped that the outliers will "realize they don’t just have a boarding problem – they have a serious boarding problem," she said.
"If not, then we may have to do what the rest of the world is doing."
Boarding has been a growing problem over the last decade and may worsen – at least in the short term – with the fuller implementation of the Affordable Care Act, according to experts who are calling for hospitals to implement proven remedies.
Hospital efficiency has suffered as institutions seek to cut costs while coping with a rising influx of uninsured and Medicaid patients. That financial pressure will not improve under health care reform, at least not initially, as millions more Medicaid patients are likely to seek care in the ED when they cannot access primary care, Dr. Jody Crane, emergency department faculty for the Institute for Healthcare Improvement, Cambridge, Mass., said in an interview.
Medicaid pay is already low, and an estimated 8 million more Medicaid patients will visit EDs annually after reform. In addition, as Medicare dials back its reimbursement to hospitals and physicians to help fund the Affordable Care Act, Va. That will put a squeeze on already tight hospital margins, he said.
"Every hospital is going to have to do the same or more with less," said Dr. Crane. Boarding – holding patients in the ED while they wait for admission to inpatient services – can be costly, reduces patient satisfaction, and has been shown to ultimately increase length of stay and mortality.
"Boarding in some hospitals is a capacity issue, but in others it’s also a management issue," Dr. Crane pointed out. Technical issues, including matching resources to the demand coming through the door, are also factors, he said.
"Ultimately, a coordinated effort is needed between the ED and the hospitalist to ensure quality transitions of care that maximize both health and financial outcomes," said Dr. Franklin A. Michota, director of academic affairs in the department of hospital medicine at the Cleveland Clinic. "Each hospital does need to analyze where their bottlenecks exist and whether a staffing solution is part of the answer. But the ED also needs to look at the accuracy and quality of their triage and their ability to adequately prepare the patient for the trip upstairs."
"For example," Dr. Michota continued, "sending a patient upstairs with sickle cell crisis and inadequate pain control just ties up the hospitalist unnecessarily and will delay the hospitalist in getting to the next new admission. Or failing to order a CT scan that would have sent a patient to the operating room and bypass the medical hospitalist altogether will again just delay getting to the next admission.
"Everything relies on everything else....the chain is only as strong as its weakest link."
Indeed, "the primary reason it’s a hard problem to fix is because it’s so multifactorial," said Dr. Jesse Pines, associate professor of emergency medicine and health policy at George Washington University in Washington, D.C.
What Works?
A recent study in Health Affairs showed that many hospitals still are not taking advantage of proven strategies to reduce boarding – among them, so-called smoothing of elective surgical schedules, active bed management by hospitalists, and the "full capacity protocol" that was developed by Dr. Peter Viccellio and his colleagues at the State University of New York at Stony Brook.
"This paper is another in a long series of wake-up calls about the dangerous practice of ‘boarding’ admitted patients in [emergency departments]," ACEP President Dr. David Seaberg said in a statement. "I just hope that hospital administrators and legislators smell the coffee. Their leadership is critical to changing the flow of patients from the front door of the emergency department to the inpatient floors of the hospital."
Emergency departments and hospitals can work together to find ways to improve efficiency, but several important ingredients are needed, Dr. Pines said. The Health Affairs article (2012;31:1757-66 [doi: 10.1377/hlthaff.2011.0786]) cites certain protocols that work, "but you need people to make them work," he said. "You need a strong leader in the ED who can get buy-in from the emergency group. ... You also need a supportive administration and medical staff to make it happen."
It is important to have a local quality improvement group whose members can look at the overall process by which patients get moved from the ED to inpatient services, and examine how patients are managed in the ED after they are admitted, Dr. Pines said. After the existing process is analyzed, hospital staff can identify and address the inefficiencies causing the delays.
The inefficiencies are highly particular to each facility, and often to each day and each shift in the ED and on inpatient wards, he said.
Staffing shortfalls – especially inpatient nursing staff – can be a major contributor to bottlenecks, Dr. Crane said. In response to financial pressures, many hospitals are reducing nursing staff, resulting in increased nurse-to-bed ratios. "That prolongs the inpatient length of stay," he said, because it leads to more work per nurse and more delays in admitting and discharging patients.
There has been a recent administrative trend toward real-time capacity management,which can be an effective way to accommodate the variation in admissions over time.. With this approach, if there is not an identified demand for nurses, they can be sent home or told not to come to work.This ability to change staffing levels according to real-time needs can be an effective way to accommodate the variation in admissions over time
Handoffs and Beds
Dr. Crane contends there are three specific elements to improving patient flow: the physician handoff, the nursing handoff, and bed efficiency.
The physician handoff can be made more efficient by optimizing the coordination between hospitalists and emergency physicians. Due to the high variation in arrival of admissions from the ED and the low numbers of admitting hospitalists at certain times, patients can queue in the ED awaiting admission orders, Dr. Crane noted.
So now, many emergency physicians write holding orders – which have an expiration time – until the patient can be admitted. Holding orders allow the patient to be moved out of the ED, Dr. Crane said. Until recently, emergency physicians have been reluctant to write holding orders, but this trend has reversed because it can be dramatically effective.ACEP now endorses the practice and notes that any associated risk is limited.
The unpredictability of admissions "creates all kinds of logistical nightmares handing off from the emergency department to the floor nurse," Dr. Crane said. Just adding more nurses is not necessarily the answer, and, in general, hospitals don’t have the money to add staff. Instead, hospitals need to better align existing resources and staff. For instance, nurses should probably not be transporting patients or cleaning rooms. That can be done with lower-priced and less-resource-intensive staff, Dr. Crane said.
Bed capacity is the third major element that hospitals should address to reduce boarding, according to Dr. Crane. Demand from the ED cannot be controlled, but on the inpatient side, hospitals can "smooth" the elective surgery schedule by moving more procedures to the middle and end of the week so that all the beds aren’t occupied early in the week. And that is not necessarily easy: "When you change block schedules, you get into cultural issues" with surgeons who do not want to have to see patients on the weekends, Dr. Crane said.
There are also practical issues limiting weekend care. Namely, the typical services such as physical therapy and case management are not routinely present on the weekends. As such, patients who could otherwise be discharged stay in their beds until Monday, when bed demands from the ED and OR increase dramatically. Bed capacity could be improved if hospitals can find ways to make services like testing and therapy available on weekends, and find surgeons and other consultants who will round on Saturdays and Sundays. That might mean hiring a physician’s assistant or other midlevel provider to cover.
Observation Units
Hospitals have also started making use of "observation" status for patients or adding observation units.
At Mary Washington Hospital, where Dr. Crane practices, a team of physicians, nurses, and administrators is designing a streamlined protocol for observation patients. Instead of the standard admission history and physical, an abbreviated assessment has been designed, incorporating use of the ED record and a brief assessment format. Consultations are reduced, and evidence-based pathways are being created for a variety of admitting diagnoses. Patients are treated as outpatients unless test results indicate they need to be moved to the inpatient side, and due to the reduced workload, nurses may be able to work comfortably with higher bed-to-nurse ratios.
The target is to hold patients for observation for 24 hours or less, Dr. Crane said.
Because observation patients are considered outpatients, insurers will pay the hospital less for the visit than if the patient is admitted, Dr. Pines noted, and it leaves the patient vulnerable to higher expenses than just an ED copay.
Another recent study in Health Affairs found that observation stays have been rapidly increasing – from 86.9 observation stays per 1,000 inpatient admissions in 2007 for Medicare beneficiaries to 116.6 in 2009 (Health Affairs 2012;31:1251-9). Of every 1,000 Medicare beneficiaries, 2 were under observation each month in 2007 and almost 3 were under observation each month in 2009. Meanwhile, inpatient admissions per 1,000 declined.
The Sharp Stick of Regulation
Voluntary efforts by hospitals may not be enough, said Dr. Sandra Schneider, former president of the American College of Emergency Physicians. Because hospitals are still paid for the patients who are boarded, they might not see the hidden costs of boarding, she said.
"We have to change that playing field if we want to get serious about [boarding]," Dr. Schneider said.
Among the game changers she cited are the increasing requirements for public reporting on quality measures, including those related to boarding.
Currently, hospitals get paid more by Medicare if they report certain inpatient quality measures. On the outpatient side, emergency departments report on the median time from ED arrival to ED departure for discharged patients and on patients who leave without being seen. In addition, it is now voluntary for hospitals to report on the average time patients spend in the ED before being admitted as an inpatient, and the average time patients spend in the ED after admission but before leaving the ED for their inpatient room.
Data from hospitals that have reported on these measures can be found on the Hospital Compare website.
The Joint Commission is also revisiting its standard on patient flow in the ED, which will go into effect in January 2013. Among other things, the commission is considering defining an episode of boarding as any case in which a patient remains in the ED 4 hours or more after a decision has been made to admit or transfer that patient.
Dr. Pines said that hospital administrators will be motivated both by public reporting imperatives and by Joint Commission standards. Dr. Crane, however, said that "until there are some significant incentives to moving patients upstairs in a timely manner, what’s going to drive senior executives is finance." He thinks that these new standards may help, but more incentives may be needed, such as penalties imposed if boarding limits are exceeded.
Australia, Canada, the United Kingdom, and other developed countries impose such limits and levy penalties.
Hospital Compare data – which will be more fully reported at the end of the first quarter of 2013 – “is going to show us once and for all where the median is and who are the outliers,” Dr. Schneider said. It’s hoped that the outliers will "realize they don’t just have a boarding problem – they have a serious boarding problem," she said.
"If not, then we may have to do what the rest of the world is doing."
Family Physicians Endorse Same-Sex Civil Marriage
PHILADELPHIA – The American Academy of Family Physicians Congress of Delegates has approved a new policy to support civil marriage for same-sex couples, making it one of the few medical societies to explicitly endorse gay marriage.
AAFP policy now states that the organization supports "civil marriage for same-gender couples to contribute to overall health and longevity, improved family stability, and to benefit children of gay, lesbian, bisexual, transgender (GLBT) families."
The congress voted 75-44 to approve that language, which was crafted from two resolutions brought forward by the resident and student sections. This was the third year in a row that the congress had weighed whether the organization should give its backing to same-sex marriage. And, as in past years, debate was lengthy and impassioned.
Many supporters testified that their own same-sex partnerships had suffered as a result of being denied the ability to marry.
Dr. Mark Dressner, president-elect of the California chapter, said that he felt that he and his partner had been treated as second-class citizens. Others spoke of the emotional and physical damage they had witnessed in patients and family members as a result of discrimination against homosexuals.
Dr. Lloyd Van Winkle of Castroville, Tex., who was just elected to the AAFP board, said that a young relation* committed suicide at age 19 because he felt persecuted about his homosexuality.
Dr. Paul W. Davis, a delegate from Alaska, spoke about his long journey from "the extreme conservative religious right wing on this debate to a committed solidarity with the opposite pole."
Dr. Davis said that he had watched patients struggle with major depression over their sexual and gender identity. "I am not proud of the fact that I am a late adopter on this important issue," he said, urging his fellow delegates that "we need to boldly make a decision and move forward."
But opponents said that AAFP should not be wading so deeply into what they considered largely treacherous political waters.
Dr. Justin V. Bartos, a delegate from Texas, said that his state’s chapter opposed the resolution because it was too much of a political statement and diverged from the AAFP’s mission.
Members of the Texas Academy of Family Physicians "do not want us to endorse such a politically polarizing issue," Dr. Bartos said. He noted that family doctors in Texas are struggling to get their state legislature to restore funding for community-based residencies, and that lawmakers in the largely conservative state would note the AAFP’s support of gay marriage.
The Tennessee delegation also opposed the policy change for the same reasons – that it would politicize the AAFP.
Another Texas delegate, Dr. Erica Swegler*, said that current AAFP policy was enough to indicate the organization’s support for equal access to health care for all Americans.
During testimony before the reference committee charged with weighing the policy change, an Arkansas delegate said that more than 300 members might leave the AAFP if it gave its approval to gay marriage.
The AAFP joins the American Psychiatric Association and the American Psychological Association in supporting same sex marriage. The psychiatrists approved a resolution of support in 2005, and the psychology association made it official policy in 2011.
The American Medical Association has not backed gay marriage specifically, but has a host of policies supporting equality for same-sex households.
The American College of Obstetricians and Gynecologists in 2009 issued a policy statement that same-sex couples should get same legal rights as married heterosexuals.
*Correction, 11/8/12: An earlier version of this story misidentified Dr. Van Winkle's relation and misspelled Dr. Swegler's name.
PHILADELPHIA – The American Academy of Family Physicians Congress of Delegates has approved a new policy to support civil marriage for same-sex couples, making it one of the few medical societies to explicitly endorse gay marriage.
AAFP policy now states that the organization supports "civil marriage for same-gender couples to contribute to overall health and longevity, improved family stability, and to benefit children of gay, lesbian, bisexual, transgender (GLBT) families."
The congress voted 75-44 to approve that language, which was crafted from two resolutions brought forward by the resident and student sections. This was the third year in a row that the congress had weighed whether the organization should give its backing to same-sex marriage. And, as in past years, debate was lengthy and impassioned.
Many supporters testified that their own same-sex partnerships had suffered as a result of being denied the ability to marry.
Dr. Mark Dressner, president-elect of the California chapter, said that he felt that he and his partner had been treated as second-class citizens. Others spoke of the emotional and physical damage they had witnessed in patients and family members as a result of discrimination against homosexuals.
Dr. Lloyd Van Winkle of Castroville, Tex., who was just elected to the AAFP board, said that a young relation* committed suicide at age 19 because he felt persecuted about his homosexuality.
Dr. Paul W. Davis, a delegate from Alaska, spoke about his long journey from "the extreme conservative religious right wing on this debate to a committed solidarity with the opposite pole."
Dr. Davis said that he had watched patients struggle with major depression over their sexual and gender identity. "I am not proud of the fact that I am a late adopter on this important issue," he said, urging his fellow delegates that "we need to boldly make a decision and move forward."
But opponents said that AAFP should not be wading so deeply into what they considered largely treacherous political waters.
Dr. Justin V. Bartos, a delegate from Texas, said that his state’s chapter opposed the resolution because it was too much of a political statement and diverged from the AAFP’s mission.
Members of the Texas Academy of Family Physicians "do not want us to endorse such a politically polarizing issue," Dr. Bartos said. He noted that family doctors in Texas are struggling to get their state legislature to restore funding for community-based residencies, and that lawmakers in the largely conservative state would note the AAFP’s support of gay marriage.
The Tennessee delegation also opposed the policy change for the same reasons – that it would politicize the AAFP.
Another Texas delegate, Dr. Erica Swegler*, said that current AAFP policy was enough to indicate the organization’s support for equal access to health care for all Americans.
During testimony before the reference committee charged with weighing the policy change, an Arkansas delegate said that more than 300 members might leave the AAFP if it gave its approval to gay marriage.
The AAFP joins the American Psychiatric Association and the American Psychological Association in supporting same sex marriage. The psychiatrists approved a resolution of support in 2005, and the psychology association made it official policy in 2011.
The American Medical Association has not backed gay marriage specifically, but has a host of policies supporting equality for same-sex households.
The American College of Obstetricians and Gynecologists in 2009 issued a policy statement that same-sex couples should get same legal rights as married heterosexuals.
*Correction, 11/8/12: An earlier version of this story misidentified Dr. Van Winkle's relation and misspelled Dr. Swegler's name.
PHILADELPHIA – The American Academy of Family Physicians Congress of Delegates has approved a new policy to support civil marriage for same-sex couples, making it one of the few medical societies to explicitly endorse gay marriage.
AAFP policy now states that the organization supports "civil marriage for same-gender couples to contribute to overall health and longevity, improved family stability, and to benefit children of gay, lesbian, bisexual, transgender (GLBT) families."
The congress voted 75-44 to approve that language, which was crafted from two resolutions brought forward by the resident and student sections. This was the third year in a row that the congress had weighed whether the organization should give its backing to same-sex marriage. And, as in past years, debate was lengthy and impassioned.
Many supporters testified that their own same-sex partnerships had suffered as a result of being denied the ability to marry.
Dr. Mark Dressner, president-elect of the California chapter, said that he felt that he and his partner had been treated as second-class citizens. Others spoke of the emotional and physical damage they had witnessed in patients and family members as a result of discrimination against homosexuals.
Dr. Lloyd Van Winkle of Castroville, Tex., who was just elected to the AAFP board, said that a young relation* committed suicide at age 19 because he felt persecuted about his homosexuality.
Dr. Paul W. Davis, a delegate from Alaska, spoke about his long journey from "the extreme conservative religious right wing on this debate to a committed solidarity with the opposite pole."
Dr. Davis said that he had watched patients struggle with major depression over their sexual and gender identity. "I am not proud of the fact that I am a late adopter on this important issue," he said, urging his fellow delegates that "we need to boldly make a decision and move forward."
But opponents said that AAFP should not be wading so deeply into what they considered largely treacherous political waters.
Dr. Justin V. Bartos, a delegate from Texas, said that his state’s chapter opposed the resolution because it was too much of a political statement and diverged from the AAFP’s mission.
Members of the Texas Academy of Family Physicians "do not want us to endorse such a politically polarizing issue," Dr. Bartos said. He noted that family doctors in Texas are struggling to get their state legislature to restore funding for community-based residencies, and that lawmakers in the largely conservative state would note the AAFP’s support of gay marriage.
The Tennessee delegation also opposed the policy change for the same reasons – that it would politicize the AAFP.
Another Texas delegate, Dr. Erica Swegler*, said that current AAFP policy was enough to indicate the organization’s support for equal access to health care for all Americans.
During testimony before the reference committee charged with weighing the policy change, an Arkansas delegate said that more than 300 members might leave the AAFP if it gave its approval to gay marriage.
The AAFP joins the American Psychiatric Association and the American Psychological Association in supporting same sex marriage. The psychiatrists approved a resolution of support in 2005, and the psychology association made it official policy in 2011.
The American Medical Association has not backed gay marriage specifically, but has a host of policies supporting equality for same-sex households.
The American College of Obstetricians and Gynecologists in 2009 issued a policy statement that same-sex couples should get same legal rights as married heterosexuals.
*Correction, 11/8/12: An earlier version of this story misidentified Dr. Van Winkle's relation and misspelled Dr. Swegler's name.
AT THE AAFP CONGRESS OF DELEGATES
AAFP Vows to Continue Fight for Better Pay
PHILADELPHIA – The American Academy of Family Physicians opened its Congress of Delegates meeting here with a vow to press forward with efforts to have government and private payers reimburse the specialty at a higher rate.
At a Town Hall forum on the evening of Oct. 14 and the first general session on Oct. 15, the academy’s leadership said that it would continue to fight for a higher valuation for family medicine through its representation on the American Medical Association’s Relative Value Scale Update Committee (RUC). In the fall of 2011, the AAFP threatened to pull out of the RUC if the payment concerns of family physicians were not addressed.
In March, however, the academy said it would consider alternate ways to support a revaluation of family medicine.
Many AAFP members have not been happy with that approach and have floated several resolutions to keep hammering at the issue. Among them: a resolution to create a set of family medicine–specific codes. Testimony at the AAFP’s Reference Committee on Practice Enhancement seemed to lean largely against that proposal, but there was consensus that family medicine is consistently undervalued.
"This congress is very familiar with family medicine’s frustration with the RUC," AAFP President Glen Stream said at the meeting. He noted, however, that for the moment, "remaining in the RUC allows the opportunity to advocate and press for [the] changes."
The full congress will probably take up the RUC-related resolutions and put them to a vote on Oct. 16 or Oct. 17.
The same committee also heard cautionary testimony on a resolution that would make it AAFP policy to oppose telemedicine that is provided in the absence of a pre-existing physician-patient relationship. The resolution emerged from concerns about the rise of for-profit entities – such as pharmacy chains – that bypass physicians in the community.
Impassioned and lengthy testimony at the Reference Committee on Advocacy addressed two resolutions seeking the AAFP’s backing of same-sex civil marriage. An overwhelming number of speakers said that such marriages would help improve public and individual health. More than a handful gave personal anecdotes about their own same-sex relationships, but several of the opposing delegates said that they would leave AAFP if gave its support to the resolution.
The advocacy committee also heard pro and con on a resolution to back over-the-counter emergency contraception for all women of childbearing age.
Finally, the AAFP leadership said that the board had given approval to generate a new report on what family medicine should look like going forward. The AAFP developed just such a blueprint 8 years ago, but the research that supported that paper has become dated, said Dr. Douglas Henley, AAFP executive vice president.
"Now is a critical time to begin a discussion and perhaps revisit research or even initiate new research about what comprehensive primary care means in 2012 and into the future," Dr. Henley said.
The process has begun, he noted, adding that AAFP members should expect to hear more in January.
PHILADELPHIA – The American Academy of Family Physicians opened its Congress of Delegates meeting here with a vow to press forward with efforts to have government and private payers reimburse the specialty at a higher rate.
At a Town Hall forum on the evening of Oct. 14 and the first general session on Oct. 15, the academy’s leadership said that it would continue to fight for a higher valuation for family medicine through its representation on the American Medical Association’s Relative Value Scale Update Committee (RUC). In the fall of 2011, the AAFP threatened to pull out of the RUC if the payment concerns of family physicians were not addressed.
In March, however, the academy said it would consider alternate ways to support a revaluation of family medicine.
Many AAFP members have not been happy with that approach and have floated several resolutions to keep hammering at the issue. Among them: a resolution to create a set of family medicine–specific codes. Testimony at the AAFP’s Reference Committee on Practice Enhancement seemed to lean largely against that proposal, but there was consensus that family medicine is consistently undervalued.
"This congress is very familiar with family medicine’s frustration with the RUC," AAFP President Glen Stream said at the meeting. He noted, however, that for the moment, "remaining in the RUC allows the opportunity to advocate and press for [the] changes."
The full congress will probably take up the RUC-related resolutions and put them to a vote on Oct. 16 or Oct. 17.
The same committee also heard cautionary testimony on a resolution that would make it AAFP policy to oppose telemedicine that is provided in the absence of a pre-existing physician-patient relationship. The resolution emerged from concerns about the rise of for-profit entities – such as pharmacy chains – that bypass physicians in the community.
Impassioned and lengthy testimony at the Reference Committee on Advocacy addressed two resolutions seeking the AAFP’s backing of same-sex civil marriage. An overwhelming number of speakers said that such marriages would help improve public and individual health. More than a handful gave personal anecdotes about their own same-sex relationships, but several of the opposing delegates said that they would leave AAFP if gave its support to the resolution.
The advocacy committee also heard pro and con on a resolution to back over-the-counter emergency contraception for all women of childbearing age.
Finally, the AAFP leadership said that the board had given approval to generate a new report on what family medicine should look like going forward. The AAFP developed just such a blueprint 8 years ago, but the research that supported that paper has become dated, said Dr. Douglas Henley, AAFP executive vice president.
"Now is a critical time to begin a discussion and perhaps revisit research or even initiate new research about what comprehensive primary care means in 2012 and into the future," Dr. Henley said.
The process has begun, he noted, adding that AAFP members should expect to hear more in January.
PHILADELPHIA – The American Academy of Family Physicians opened its Congress of Delegates meeting here with a vow to press forward with efforts to have government and private payers reimburse the specialty at a higher rate.
At a Town Hall forum on the evening of Oct. 14 and the first general session on Oct. 15, the academy’s leadership said that it would continue to fight for a higher valuation for family medicine through its representation on the American Medical Association’s Relative Value Scale Update Committee (RUC). In the fall of 2011, the AAFP threatened to pull out of the RUC if the payment concerns of family physicians were not addressed.
In March, however, the academy said it would consider alternate ways to support a revaluation of family medicine.
Many AAFP members have not been happy with that approach and have floated several resolutions to keep hammering at the issue. Among them: a resolution to create a set of family medicine–specific codes. Testimony at the AAFP’s Reference Committee on Practice Enhancement seemed to lean largely against that proposal, but there was consensus that family medicine is consistently undervalued.
"This congress is very familiar with family medicine’s frustration with the RUC," AAFP President Glen Stream said at the meeting. He noted, however, that for the moment, "remaining in the RUC allows the opportunity to advocate and press for [the] changes."
The full congress will probably take up the RUC-related resolutions and put them to a vote on Oct. 16 or Oct. 17.
The same committee also heard cautionary testimony on a resolution that would make it AAFP policy to oppose telemedicine that is provided in the absence of a pre-existing physician-patient relationship. The resolution emerged from concerns about the rise of for-profit entities – such as pharmacy chains – that bypass physicians in the community.
Impassioned and lengthy testimony at the Reference Committee on Advocacy addressed two resolutions seeking the AAFP’s backing of same-sex civil marriage. An overwhelming number of speakers said that such marriages would help improve public and individual health. More than a handful gave personal anecdotes about their own same-sex relationships, but several of the opposing delegates said that they would leave AAFP if gave its support to the resolution.
The advocacy committee also heard pro and con on a resolution to back over-the-counter emergency contraception for all women of childbearing age.
Finally, the AAFP leadership said that the board had given approval to generate a new report on what family medicine should look like going forward. The AAFP developed just such a blueprint 8 years ago, but the research that supported that paper has become dated, said Dr. Douglas Henley, AAFP executive vice president.
"Now is a critical time to begin a discussion and perhaps revisit research or even initiate new research about what comprehensive primary care means in 2012 and into the future," Dr. Henley said.
The process has begun, he noted, adding that AAFP members should expect to hear more in January.
AT THE AAFP CONGRESS OF DELEGATES
Alzheimer's Imaging Agent Considered for Medicare Coverage
The imaging agent florbetapir F 18 injection (Amyvid), used to exclude a diagnosis of Alzheimer’s disease, is being evaluated for Medicare reimbursement, the Centers for Medicare and Medicaid Services has announced.
The agency stated on Oct. 9 that it expected a preliminary decision by July 2013, and a final decision by October 2013.
The CMS is accepting public comments on its proposed coverage of florbetapir until Nov. 8.
Florbetapir, approved by the Food and Drug Administration in April, is used in conjunction with positron emission tomography (PET). Eli Lilly, which owns florbetapir maker Avid Radiopharmaceuticals, requested the coverage determination. The company laid out its case for reimbursement in a 47-page letter to the CMS in June.
In the request, Lilly noted that the CMS approved coverage of FDG-PET for patients who had a recent dementia diagnosis and cognitive decline over 6 months and who met diagnostic criteria for Alzheimer’s disease and frontotemporal dementia.
According to Avid, florbetapir binds to beta-amyloid plaques in the brain, which are considered markers for Alzheimer’s. Without the ability to see those plaques in real time, diagnosis has been based on symptoms and cognitive testing. But that is imperfect, missing perhaps a fifth of cases.
At the time of florbetapir’s approval, the Alzheimer's Association said the imaging agent would be useful in diagnosis and in tracking of treatment response, but also expressed concern about potentially unscrupulous uses of the technology.
The organization has convened a task force with the Society of Nuclear Medicine to develop guidelines on florbetapir use.
An estimated 5 million Americans have Alzheimer’s, and the incidence is rising with the aging of the U.S. population. Only 4% of those with the disease are under the Medicare age of 65, according to Lilly; 40%-45% of Americans over age 75 are estimated to have the disease.
The imaging agent florbetapir F 18 injection (Amyvid), used to exclude a diagnosis of Alzheimer’s disease, is being evaluated for Medicare reimbursement, the Centers for Medicare and Medicaid Services has announced.
The agency stated on Oct. 9 that it expected a preliminary decision by July 2013, and a final decision by October 2013.
The CMS is accepting public comments on its proposed coverage of florbetapir until Nov. 8.
Florbetapir, approved by the Food and Drug Administration in April, is used in conjunction with positron emission tomography (PET). Eli Lilly, which owns florbetapir maker Avid Radiopharmaceuticals, requested the coverage determination. The company laid out its case for reimbursement in a 47-page letter to the CMS in June.
In the request, Lilly noted that the CMS approved coverage of FDG-PET for patients who had a recent dementia diagnosis and cognitive decline over 6 months and who met diagnostic criteria for Alzheimer’s disease and frontotemporal dementia.
According to Avid, florbetapir binds to beta-amyloid plaques in the brain, which are considered markers for Alzheimer’s. Without the ability to see those plaques in real time, diagnosis has been based on symptoms and cognitive testing. But that is imperfect, missing perhaps a fifth of cases.
At the time of florbetapir’s approval, the Alzheimer's Association said the imaging agent would be useful in diagnosis and in tracking of treatment response, but also expressed concern about potentially unscrupulous uses of the technology.
The organization has convened a task force with the Society of Nuclear Medicine to develop guidelines on florbetapir use.
An estimated 5 million Americans have Alzheimer’s, and the incidence is rising with the aging of the U.S. population. Only 4% of those with the disease are under the Medicare age of 65, according to Lilly; 40%-45% of Americans over age 75 are estimated to have the disease.
The imaging agent florbetapir F 18 injection (Amyvid), used to exclude a diagnosis of Alzheimer’s disease, is being evaluated for Medicare reimbursement, the Centers for Medicare and Medicaid Services has announced.
The agency stated on Oct. 9 that it expected a preliminary decision by July 2013, and a final decision by October 2013.
The CMS is accepting public comments on its proposed coverage of florbetapir until Nov. 8.
Florbetapir, approved by the Food and Drug Administration in April, is used in conjunction with positron emission tomography (PET). Eli Lilly, which owns florbetapir maker Avid Radiopharmaceuticals, requested the coverage determination. The company laid out its case for reimbursement in a 47-page letter to the CMS in June.
In the request, Lilly noted that the CMS approved coverage of FDG-PET for patients who had a recent dementia diagnosis and cognitive decline over 6 months and who met diagnostic criteria for Alzheimer’s disease and frontotemporal dementia.
According to Avid, florbetapir binds to beta-amyloid plaques in the brain, which are considered markers for Alzheimer’s. Without the ability to see those plaques in real time, diagnosis has been based on symptoms and cognitive testing. But that is imperfect, missing perhaps a fifth of cases.
At the time of florbetapir’s approval, the Alzheimer's Association said the imaging agent would be useful in diagnosis and in tracking of treatment response, but also expressed concern about potentially unscrupulous uses of the technology.
The organization has convened a task force with the Society of Nuclear Medicine to develop guidelines on florbetapir use.
An estimated 5 million Americans have Alzheimer’s, and the incidence is rising with the aging of the U.S. population. Only 4% of those with the disease are under the Medicare age of 65, according to Lilly; 40%-45% of Americans over age 75 are estimated to have the disease.
FDA Targets Thousands of Illegal Online Pharmacies
The Food and Drug Administration took action this week against 4,100 Internet pharmacies selling counterfeit and illegal medicines, part of a coordinated international crackdown on the $75 billion counterfeit drug market.
The larger worldwide initiative, known as Operation Pangea V, shut down more than 18,000 illegal pharmacy websites and seized $10.5 million in pharmaceuticals worldwide during the week of Sept. 25-Oct. 2.
"Because these criminals do not respect international borders, the international coordinated law enforcement response represented by Operation Pangea demonstrates that international cooperation is the best way to protect the American public from the risk of unsafe drugs," said John Roth, director of the FDA’s Office of Criminal Investigations, in a statement.
Counterfeit drug sales hit $75 billion worldwide in 2010, a 90% increase from the previous year, according to the Center for Safe Internet Pharmacies (CSIP).
INTERPOL, the World Customs Organization, the Permanent Forum of International Pharmaceutical Crime, the Heads of Medicines Agencies Working Group of Enforcement Officers, Pharmaceutical Security Institute, and Europol directed the Pangea V, with participation from more than 100 countries.
The operation also received support from CSIP, as well as a group of Internet and e-commerce companies that includes Visa, American Express, MasterCard, Yahoo, Facebook, and PayPal.
The FDA said it targeted websites selling "unapproved and potentially dangerous medicines." The agency sent warning letters to the operators of more than 4,100 identified websites and then notified registries, Internet service providers, and domain name registrars that these websites were selling products in violation of U.S. law.
During the week-long crackdown, the FDA screened all drug products received through the international mail facilities. The screenings found antibiotics, antidepressants, and other drugs for high cholesterol, diabetes, and high blood pressure headed for American consumers. Many of those drugs could be risky to take without the supervision of a physician, the agency warned.
Some of the drugs intercepted also had been removed from the U.S. market, including domperidone, which was taken off the U.S. market in 1998 because of its potential to cause severe cardiac effects, including sudden death.
Also found in the mail were the acne drug isotretinoin, which is available in the United States only through a risk management distribution program; sildenafil citrate (Viagra); and the antiviral oseltamivir (Tamiflu), which is often sold as a "generic," though there is no such approved U.S. generic. Tests by the FDA have shown that fraudulent generic versions of Tamiflu contained the wrong active ingredient, which would not be effective in treating flu.
Seventy-nine people have been arrested or are under investigation, according to INTERPOL.
The FDA launched a consumer-oriented campaign in late September, called BeSafeRx–Know Your Online Pharmacy. The website gives consumers tips on how to understand who they are buying from and when it might not be a good idea to purchase through a website.
The agency said one-quarter of consumers buy prescription drugs online, according to a survey it conducted, but that one-third of respondents said they did not have confidence in how to make safe online purchases.
"Fraudulent and illegal online pharmacies often offer deeply discounted products," said FDA Commissioner Margaret Hamburg in a statement. "If the low prices seem too good to be true, they probably are," she said.
The FDA urged patients to only buy online through pharmacies that require a valid prescription from a doctor or other health care professional; are located in the United States; have a licensed pharmacist available for consultation; and are licensed by the patient’s state board of pharmacy.
The Food and Drug Administration took action this week against 4,100 Internet pharmacies selling counterfeit and illegal medicines, part of a coordinated international crackdown on the $75 billion counterfeit drug market.
The larger worldwide initiative, known as Operation Pangea V, shut down more than 18,000 illegal pharmacy websites and seized $10.5 million in pharmaceuticals worldwide during the week of Sept. 25-Oct. 2.
"Because these criminals do not respect international borders, the international coordinated law enforcement response represented by Operation Pangea demonstrates that international cooperation is the best way to protect the American public from the risk of unsafe drugs," said John Roth, director of the FDA’s Office of Criminal Investigations, in a statement.
Counterfeit drug sales hit $75 billion worldwide in 2010, a 90% increase from the previous year, according to the Center for Safe Internet Pharmacies (CSIP).
INTERPOL, the World Customs Organization, the Permanent Forum of International Pharmaceutical Crime, the Heads of Medicines Agencies Working Group of Enforcement Officers, Pharmaceutical Security Institute, and Europol directed the Pangea V, with participation from more than 100 countries.
The operation also received support from CSIP, as well as a group of Internet and e-commerce companies that includes Visa, American Express, MasterCard, Yahoo, Facebook, and PayPal.
The FDA said it targeted websites selling "unapproved and potentially dangerous medicines." The agency sent warning letters to the operators of more than 4,100 identified websites and then notified registries, Internet service providers, and domain name registrars that these websites were selling products in violation of U.S. law.
During the week-long crackdown, the FDA screened all drug products received through the international mail facilities. The screenings found antibiotics, antidepressants, and other drugs for high cholesterol, diabetes, and high blood pressure headed for American consumers. Many of those drugs could be risky to take without the supervision of a physician, the agency warned.
Some of the drugs intercepted also had been removed from the U.S. market, including domperidone, which was taken off the U.S. market in 1998 because of its potential to cause severe cardiac effects, including sudden death.
Also found in the mail were the acne drug isotretinoin, which is available in the United States only through a risk management distribution program; sildenafil citrate (Viagra); and the antiviral oseltamivir (Tamiflu), which is often sold as a "generic," though there is no such approved U.S. generic. Tests by the FDA have shown that fraudulent generic versions of Tamiflu contained the wrong active ingredient, which would not be effective in treating flu.
Seventy-nine people have been arrested or are under investigation, according to INTERPOL.
The FDA launched a consumer-oriented campaign in late September, called BeSafeRx–Know Your Online Pharmacy. The website gives consumers tips on how to understand who they are buying from and when it might not be a good idea to purchase through a website.
The agency said one-quarter of consumers buy prescription drugs online, according to a survey it conducted, but that one-third of respondents said they did not have confidence in how to make safe online purchases.
"Fraudulent and illegal online pharmacies often offer deeply discounted products," said FDA Commissioner Margaret Hamburg in a statement. "If the low prices seem too good to be true, they probably are," she said.
The FDA urged patients to only buy online through pharmacies that require a valid prescription from a doctor or other health care professional; are located in the United States; have a licensed pharmacist available for consultation; and are licensed by the patient’s state board of pharmacy.
The Food and Drug Administration took action this week against 4,100 Internet pharmacies selling counterfeit and illegal medicines, part of a coordinated international crackdown on the $75 billion counterfeit drug market.
The larger worldwide initiative, known as Operation Pangea V, shut down more than 18,000 illegal pharmacy websites and seized $10.5 million in pharmaceuticals worldwide during the week of Sept. 25-Oct. 2.
"Because these criminals do not respect international borders, the international coordinated law enforcement response represented by Operation Pangea demonstrates that international cooperation is the best way to protect the American public from the risk of unsafe drugs," said John Roth, director of the FDA’s Office of Criminal Investigations, in a statement.
Counterfeit drug sales hit $75 billion worldwide in 2010, a 90% increase from the previous year, according to the Center for Safe Internet Pharmacies (CSIP).
INTERPOL, the World Customs Organization, the Permanent Forum of International Pharmaceutical Crime, the Heads of Medicines Agencies Working Group of Enforcement Officers, Pharmaceutical Security Institute, and Europol directed the Pangea V, with participation from more than 100 countries.
The operation also received support from CSIP, as well as a group of Internet and e-commerce companies that includes Visa, American Express, MasterCard, Yahoo, Facebook, and PayPal.
The FDA said it targeted websites selling "unapproved and potentially dangerous medicines." The agency sent warning letters to the operators of more than 4,100 identified websites and then notified registries, Internet service providers, and domain name registrars that these websites were selling products in violation of U.S. law.
During the week-long crackdown, the FDA screened all drug products received through the international mail facilities. The screenings found antibiotics, antidepressants, and other drugs for high cholesterol, diabetes, and high blood pressure headed for American consumers. Many of those drugs could be risky to take without the supervision of a physician, the agency warned.
Some of the drugs intercepted also had been removed from the U.S. market, including domperidone, which was taken off the U.S. market in 1998 because of its potential to cause severe cardiac effects, including sudden death.
Also found in the mail were the acne drug isotretinoin, which is available in the United States only through a risk management distribution program; sildenafil citrate (Viagra); and the antiviral oseltamivir (Tamiflu), which is often sold as a "generic," though there is no such approved U.S. generic. Tests by the FDA have shown that fraudulent generic versions of Tamiflu contained the wrong active ingredient, which would not be effective in treating flu.
Seventy-nine people have been arrested or are under investigation, according to INTERPOL.
The FDA launched a consumer-oriented campaign in late September, called BeSafeRx–Know Your Online Pharmacy. The website gives consumers tips on how to understand who they are buying from and when it might not be a good idea to purchase through a website.
The agency said one-quarter of consumers buy prescription drugs online, according to a survey it conducted, but that one-third of respondents said they did not have confidence in how to make safe online purchases.
"Fraudulent and illegal online pharmacies often offer deeply discounted products," said FDA Commissioner Margaret Hamburg in a statement. "If the low prices seem too good to be true, they probably are," she said.
The FDA urged patients to only buy online through pharmacies that require a valid prescription from a doctor or other health care professional; are located in the United States; have a licensed pharmacist available for consultation; and are licensed by the patient’s state board of pharmacy.
FDA Approves First Subcutaneous ICD
The Food and Drug Administration has approved a new implantable cardioverter defibrillator that is implanted subcutaneously as a treatment for ventricular tachyarrhythmias.
The agency approved the Subcutaneous Implantable Defibrillator (S-ICD) System only for patients who do not require a pacemaker or pacing therapy.
The S-ICD System, made by Cameron Health of San Clemente, Calif., is the first such device that can be placed without transvenous leads. Instead, the leads are placed under the skin and then connected to the heart.
"The S-ICD System provides an alternative for treating patients with life-threatening heart arrhythmias for whom the routine ICD placement procedure is not ideal," said Christy Foreman, director of the Office of Device Evaluation at FDA’s Center for Devices and Radiological Health, in a statement. "Some patients with anatomy that makes it challenging to place one of the implantable defibrillators currently on the market may especially benefit from this device."
The device won near-unanimous backing of the FDA’s Circulatory System Devices Panel in April. The panel voted 7-1 that the benefits of the S-ICD system outweighed its risks in patients who meet the criteria in the indication proposed by Cameron Health: to provide "defibrillation therapy for the treatment of life-threatening ventricular tachyarrhythmias in patients who do not have symptomatic bradycardia; incessant ventricular tachycardia; or spontaneous, frequently recurring ventricular tachycardia that is reliably terminated with antitachycardia pacing."
Safety and efficacy data for the approval came from a 321-patient study, which was reported at the Heart Rhythm Society in May. In that trial, 304 patients were successfully implanted and the device converted all abnormal heart rhythms that it detected back to normal rhythms, according to the FDA. In the 6 months postimplantation, the S-ICD detected and recorded 78 spontaneous arrhythmias in 21 patients that were either all converted or resolved on their own.
The FDA said that the most common complications included inappropriate shocks, discomfort, system infection, and electrode movement. Eight patients died during the study, but the S-ICD was not definitively seen as the cause. There were 11 explants. Six months after implantation, 90% of the device recipients had no complications.
Cameron Health reported in May that the S-ICD is distributed in 10 other countries and that more than 1,200 patients have been implanted worldwide.
The company proposed a postmarketing study when it submitted its application for approval. The FDA will require that study, seeking assessment of how it performs over the long term, and across genders. The study will follow 1,616 patients for 5 years, according to the agency.
The Food and Drug Administration has approved a new implantable cardioverter defibrillator that is implanted subcutaneously as a treatment for ventricular tachyarrhythmias.
The agency approved the Subcutaneous Implantable Defibrillator (S-ICD) System only for patients who do not require a pacemaker or pacing therapy.
The S-ICD System, made by Cameron Health of San Clemente, Calif., is the first such device that can be placed without transvenous leads. Instead, the leads are placed under the skin and then connected to the heart.
"The S-ICD System provides an alternative for treating patients with life-threatening heart arrhythmias for whom the routine ICD placement procedure is not ideal," said Christy Foreman, director of the Office of Device Evaluation at FDA’s Center for Devices and Radiological Health, in a statement. "Some patients with anatomy that makes it challenging to place one of the implantable defibrillators currently on the market may especially benefit from this device."
The device won near-unanimous backing of the FDA’s Circulatory System Devices Panel in April. The panel voted 7-1 that the benefits of the S-ICD system outweighed its risks in patients who meet the criteria in the indication proposed by Cameron Health: to provide "defibrillation therapy for the treatment of life-threatening ventricular tachyarrhythmias in patients who do not have symptomatic bradycardia; incessant ventricular tachycardia; or spontaneous, frequently recurring ventricular tachycardia that is reliably terminated with antitachycardia pacing."
Safety and efficacy data for the approval came from a 321-patient study, which was reported at the Heart Rhythm Society in May. In that trial, 304 patients were successfully implanted and the device converted all abnormal heart rhythms that it detected back to normal rhythms, according to the FDA. In the 6 months postimplantation, the S-ICD detected and recorded 78 spontaneous arrhythmias in 21 patients that were either all converted or resolved on their own.
The FDA said that the most common complications included inappropriate shocks, discomfort, system infection, and electrode movement. Eight patients died during the study, but the S-ICD was not definitively seen as the cause. There were 11 explants. Six months after implantation, 90% of the device recipients had no complications.
Cameron Health reported in May that the S-ICD is distributed in 10 other countries and that more than 1,200 patients have been implanted worldwide.
The company proposed a postmarketing study when it submitted its application for approval. The FDA will require that study, seeking assessment of how it performs over the long term, and across genders. The study will follow 1,616 patients for 5 years, according to the agency.
The Food and Drug Administration has approved a new implantable cardioverter defibrillator that is implanted subcutaneously as a treatment for ventricular tachyarrhythmias.
The agency approved the Subcutaneous Implantable Defibrillator (S-ICD) System only for patients who do not require a pacemaker or pacing therapy.
The S-ICD System, made by Cameron Health of San Clemente, Calif., is the first such device that can be placed without transvenous leads. Instead, the leads are placed under the skin and then connected to the heart.
"The S-ICD System provides an alternative for treating patients with life-threatening heart arrhythmias for whom the routine ICD placement procedure is not ideal," said Christy Foreman, director of the Office of Device Evaluation at FDA’s Center for Devices and Radiological Health, in a statement. "Some patients with anatomy that makes it challenging to place one of the implantable defibrillators currently on the market may especially benefit from this device."
The device won near-unanimous backing of the FDA’s Circulatory System Devices Panel in April. The panel voted 7-1 that the benefits of the S-ICD system outweighed its risks in patients who meet the criteria in the indication proposed by Cameron Health: to provide "defibrillation therapy for the treatment of life-threatening ventricular tachyarrhythmias in patients who do not have symptomatic bradycardia; incessant ventricular tachycardia; or spontaneous, frequently recurring ventricular tachycardia that is reliably terminated with antitachycardia pacing."
Safety and efficacy data for the approval came from a 321-patient study, which was reported at the Heart Rhythm Society in May. In that trial, 304 patients were successfully implanted and the device converted all abnormal heart rhythms that it detected back to normal rhythms, according to the FDA. In the 6 months postimplantation, the S-ICD detected and recorded 78 spontaneous arrhythmias in 21 patients that were either all converted or resolved on their own.
The FDA said that the most common complications included inappropriate shocks, discomfort, system infection, and electrode movement. Eight patients died during the study, but the S-ICD was not definitively seen as the cause. There were 11 explants. Six months after implantation, 90% of the device recipients had no complications.
Cameron Health reported in May that the S-ICD is distributed in 10 other countries and that more than 1,200 patients have been implanted worldwide.
The company proposed a postmarketing study when it submitted its application for approval. The FDA will require that study, seeking assessment of how it performs over the long term, and across genders. The study will follow 1,616 patients for 5 years, according to the agency.