User login
Transfusions Worsen Outcomes After Colorectal Cancer Surgery
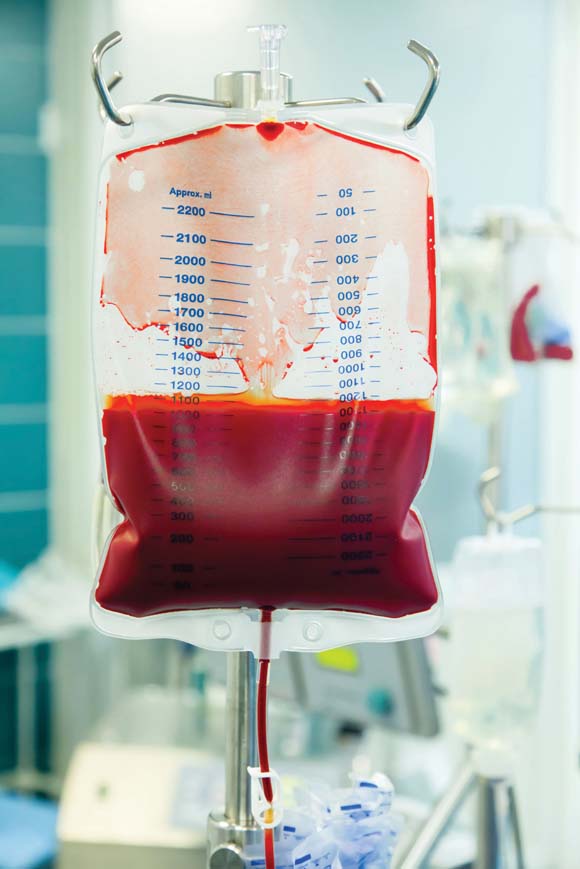
The risks of death, postoperative infection, and other adverse clinical outcomes were significantly increased among patients undergoing colorectal cancer surgery who received perioperative allogeneic blood transfusions, according to a meta-analysis of 55 studies published in the August issue of Annals of Surgery.
Dr. Austin G. Acheson of the Nottingham (England) Digestive Disease Center at Queen’s Medical Centre and his associates reviewed 12 prospective studies and 43 retrospective cohort studies published between December 2004 and October 2010. The studies included 20,795 patients who were followed for a mean of about 5 years after undergoing surgery for colorectal cancer. Almost 60% (12,242) of these patients received a mean of three units of allogeneic red blood cells (Ann. Surg. 2012;256:235-44).
Patients who were transfused tended to be older, and the transfusion rate was significantly higher in women, those undergoing rectal surgery (compared with those undergoing right or left colon surgery), those with greater surgical blood loss, and those with worsening Duke's stage.
All associations between ABT and the adverse clinical outcomes described below were statistically significant.
The rates of all-cause mortality in the 29 studies that looked at this outcome were 45% among the patients who received blood transfusions vs. 35% of those who did not, a significant difference that represented a 72% increased risk. After adjusting for the duration of the observation period, the investigators found that the annual incidence of all-cause mortality was almost 9% among the transfused patients vs. 6.5% among those who were not transfused.
The rate of cancer-related mortality was 31% vs. 24% in the 17 studies that measured this outcome, a 71% increased risk. After adjusting for the length of observation, the investigators said the annual incidence of cancer-related mortality was 5.4% of those transfused and 4% of those who were not.
The rate of the combined end point of death resulting from recurrence/metastasis was 43% of those who were transfused vs. 33% of those who were not transfused – a 66% increase in risk – in the 19 studies that measured this outcome.
The risk of postoperative infections in the 12 studies that measured this outcome was 29% among those who were transfused vs. 11% of those who were not transfused, which was more than a threefold increased risk. In the two studies that measured the need for surgical reintervention, the risk was increased fourfold among those who were transfused.
In the four studies that measured the length of hospitalization, the hospital stay was a mean of almost 18 days among those who received transfusions, compared with 14 days among those who did not.
Based on evidence in this meta-analysis and other studies showing that preoperative anemia is an independent risk factor for a worse prognosis after colon surgery, and based on the association between ABTs and poorer clinical outcomes in this meta-analysis, the authors wrote that "appropriate blood management measures should, therefore, be given an important place in the care of patients with CRC [colorectal cancer] undergoing elective surgery."
The use of ABT has dropped over the past 25 years because of improvements in patient care, and the authors stated that they believe efforts should be made to further minimize ABT use. Well-designed studies are needed "to determine whether and in which patients preoperative corrective measures of anemia, other than ABTs, will contribute not only in the field of colorectal surgery but also to improve clinical outcomes," they added.
The study was supported by an unrestricted research grant from Vifor Pharma AG, a specialty pharmaceuticals company focused on the treatment of iron deficiency, according to its website. The authors disclosed receiving honoraria and/or travel support for consulting or lecturing for companies that include Vifor, Ethicon Endosurgery, Johnson & Johnson, and AstraZeneca AG; their research departments received grant support from companies that included Vifor.
The meta-analysis by Dr. Acheson and his colleagues and another recent publication using the American College of Surgeons National Surgical Quality Improvement Program (Surgery 2012;152:344-54) highlight concerns about the increased mortality associated with perioperative blood transfusions. Intraoperative transfusion is associated with higher rates of 30-day operative mortality, major complications, total number of complications, and prolonged length of stay.
In general, patients requiring transfusions tend to be older, more
anemic and debilitated, have more extensive disease, and often need
larger surgical procedures with attendant greater blood loss. Patients
with these characteristics should be relatively evenly distributed among hospitals performing colorectal cancer surgery, and one would expect the frequency of transfusion to be similar across institutions.
However, Dr. Acheson and his associates found that the transfusion rate varied from 17.4% to 82.2%, suggesting that the indications for transfusion were quite
variable. Furthermore, the National Surgical Quality Improvement Program noted that more than 60% of patients received only one to two units of blood. This
raises concerns about whether blood transfusions are being given appropriately.
Some of the steps can be taken to decrease the use of perioperative blood transfusions and prevent transfusion-related morbidity are:
• Correcting anemia prior to
surgery by giving iron with or with
out erythropoietin;
• Using blood conservation methods when extensive surgery is
planned;
• Educating anesthesiologists and
surgeons about the indications for
transfusion (there is no rigid “trans
fusion trigger”; each patient must be
assessed for the need to augment the
oxygen-carrying capacity of the
blood);
• Avoiding prospective donors alloimmunized to white cell antigens
and leuko-reduce blood products
(Blood 2009;113:3406-17);
• Avoiding the use of blood that has
undergone prolonged storage (new
blood is better than old) (N. Engl. J.
Med. 2008;20:358:1229-39; Blood
2011;118:6675-82); and
• Recognizing that acute illness after perioperative transfusions might be due to transfusion-transmitted infections or transfusion-associated lung injury (TRALI) (Blood 2012;119:1757-67).
Perioperative allogeneic blood transfusions can be life-saving, but must be used judiciously and with the recognition that they occasionally increase morbidity and mortality.
DAVID GREEN, M.D., PH.D., is in the
division of hematology/oncology in
the department of medicine at
Northwestern University Feinberg
School of Medicine, Chicago. He has
no relevant disclosures.
The meta-analysis by Dr. Acheson and his colleagues and another recent publication using the American College of Surgeons National Surgical Quality Improvement Program (Surgery 2012;152:344-54) highlight concerns about the increased mortality associated with perioperative blood transfusions. Intraoperative transfusion is associated with higher rates of 30-day operative mortality, major complications, total number of complications, and prolonged length of stay.
In general, patients requiring transfusions tend to be older, more
anemic and debilitated, have more extensive disease, and often need
larger surgical procedures with attendant greater blood loss. Patients
with these characteristics should be relatively evenly distributed among hospitals performing colorectal cancer surgery, and one would expect the frequency of transfusion to be similar across institutions.
However, Dr. Acheson and his associates found that the transfusion rate varied from 17.4% to 82.2%, suggesting that the indications for transfusion were quite
variable. Furthermore, the National Surgical Quality Improvement Program noted that more than 60% of patients received only one to two units of blood. This
raises concerns about whether blood transfusions are being given appropriately.
Some of the steps can be taken to decrease the use of perioperative blood transfusions and prevent transfusion-related morbidity are:
• Correcting anemia prior to
surgery by giving iron with or with
out erythropoietin;
• Using blood conservation methods when extensive surgery is
planned;
• Educating anesthesiologists and
surgeons about the indications for
transfusion (there is no rigid “trans
fusion trigger”; each patient must be
assessed for the need to augment the
oxygen-carrying capacity of the
blood);
• Avoiding prospective donors alloimmunized to white cell antigens
and leuko-reduce blood products
(Blood 2009;113:3406-17);
• Avoiding the use of blood that has
undergone prolonged storage (new
blood is better than old) (N. Engl. J.
Med. 2008;20:358:1229-39; Blood
2011;118:6675-82); and
• Recognizing that acute illness after perioperative transfusions might be due to transfusion-transmitted infections or transfusion-associated lung injury (TRALI) (Blood 2012;119:1757-67).
Perioperative allogeneic blood transfusions can be life-saving, but must be used judiciously and with the recognition that they occasionally increase morbidity and mortality.
DAVID GREEN, M.D., PH.D., is in the
division of hematology/oncology in
the department of medicine at
Northwestern University Feinberg
School of Medicine, Chicago. He has
no relevant disclosures.
The meta-analysis by Dr. Acheson and his colleagues and another recent publication using the American College of Surgeons National Surgical Quality Improvement Program (Surgery 2012;152:344-54) highlight concerns about the increased mortality associated with perioperative blood transfusions. Intraoperative transfusion is associated with higher rates of 30-day operative mortality, major complications, total number of complications, and prolonged length of stay.
In general, patients requiring transfusions tend to be older, more
anemic and debilitated, have more extensive disease, and often need
larger surgical procedures with attendant greater blood loss. Patients
with these characteristics should be relatively evenly distributed among hospitals performing colorectal cancer surgery, and one would expect the frequency of transfusion to be similar across institutions.
However, Dr. Acheson and his associates found that the transfusion rate varied from 17.4% to 82.2%, suggesting that the indications for transfusion were quite
variable. Furthermore, the National Surgical Quality Improvement Program noted that more than 60% of patients received only one to two units of blood. This
raises concerns about whether blood transfusions are being given appropriately.
Some of the steps can be taken to decrease the use of perioperative blood transfusions and prevent transfusion-related morbidity are:
• Correcting anemia prior to
surgery by giving iron with or with
out erythropoietin;
• Using blood conservation methods when extensive surgery is
planned;
• Educating anesthesiologists and
surgeons about the indications for
transfusion (there is no rigid “trans
fusion trigger”; each patient must be
assessed for the need to augment the
oxygen-carrying capacity of the
blood);
• Avoiding prospective donors alloimmunized to white cell antigens
and leuko-reduce blood products
(Blood 2009;113:3406-17);
• Avoiding the use of blood that has
undergone prolonged storage (new
blood is better than old) (N. Engl. J.
Med. 2008;20:358:1229-39; Blood
2011;118:6675-82); and
• Recognizing that acute illness after perioperative transfusions might be due to transfusion-transmitted infections or transfusion-associated lung injury (TRALI) (Blood 2012;119:1757-67).
Perioperative allogeneic blood transfusions can be life-saving, but must be used judiciously and with the recognition that they occasionally increase morbidity and mortality.
DAVID GREEN, M.D., PH.D., is in the
division of hematology/oncology in
the department of medicine at
Northwestern University Feinberg
School of Medicine, Chicago. He has
no relevant disclosures.
The risks of death, postoperative infection, and other adverse clinical outcomes were significantly increased among patients undergoing colorectal cancer surgery who received perioperative allogeneic blood transfusions, according to a meta-analysis of 55 studies published in the August issue of Annals of Surgery.
Dr. Austin G. Acheson of the Nottingham (England) Digestive Disease Center at Queen’s Medical Centre and his associates reviewed 12 prospective studies and 43 retrospective cohort studies published between December 2004 and October 2010. The studies included 20,795 patients who were followed for a mean of about 5 years after undergoing surgery for colorectal cancer. Almost 60% (12,242) of these patients received a mean of three units of allogeneic red blood cells (Ann. Surg. 2012;256:235-44).
Patients who were transfused tended to be older, and the transfusion rate was significantly higher in women, those undergoing rectal surgery (compared with those undergoing right or left colon surgery), those with greater surgical blood loss, and those with worsening Duke's stage.
All associations between ABT and the adverse clinical outcomes described below were statistically significant.
The rates of all-cause mortality in the 29 studies that looked at this outcome were 45% among the patients who received blood transfusions vs. 35% of those who did not, a significant difference that represented a 72% increased risk. After adjusting for the duration of the observation period, the investigators found that the annual incidence of all-cause mortality was almost 9% among the transfused patients vs. 6.5% among those who were not transfused.
The rate of cancer-related mortality was 31% vs. 24% in the 17 studies that measured this outcome, a 71% increased risk. After adjusting for the length of observation, the investigators said the annual incidence of cancer-related mortality was 5.4% of those transfused and 4% of those who were not.
The rate of the combined end point of death resulting from recurrence/metastasis was 43% of those who were transfused vs. 33% of those who were not transfused – a 66% increase in risk – in the 19 studies that measured this outcome.
The risk of postoperative infections in the 12 studies that measured this outcome was 29% among those who were transfused vs. 11% of those who were not transfused, which was more than a threefold increased risk. In the two studies that measured the need for surgical reintervention, the risk was increased fourfold among those who were transfused.
In the four studies that measured the length of hospitalization, the hospital stay was a mean of almost 18 days among those who received transfusions, compared with 14 days among those who did not.
Based on evidence in this meta-analysis and other studies showing that preoperative anemia is an independent risk factor for a worse prognosis after colon surgery, and based on the association between ABTs and poorer clinical outcomes in this meta-analysis, the authors wrote that "appropriate blood management measures should, therefore, be given an important place in the care of patients with CRC [colorectal cancer] undergoing elective surgery."
The use of ABT has dropped over the past 25 years because of improvements in patient care, and the authors stated that they believe efforts should be made to further minimize ABT use. Well-designed studies are needed "to determine whether and in which patients preoperative corrective measures of anemia, other than ABTs, will contribute not only in the field of colorectal surgery but also to improve clinical outcomes," they added.
The study was supported by an unrestricted research grant from Vifor Pharma AG, a specialty pharmaceuticals company focused on the treatment of iron deficiency, according to its website. The authors disclosed receiving honoraria and/or travel support for consulting or lecturing for companies that include Vifor, Ethicon Endosurgery, Johnson & Johnson, and AstraZeneca AG; their research departments received grant support from companies that included Vifor.
The risks of death, postoperative infection, and other adverse clinical outcomes were significantly increased among patients undergoing colorectal cancer surgery who received perioperative allogeneic blood transfusions, according to a meta-analysis of 55 studies published in the August issue of Annals of Surgery.
Dr. Austin G. Acheson of the Nottingham (England) Digestive Disease Center at Queen’s Medical Centre and his associates reviewed 12 prospective studies and 43 retrospective cohort studies published between December 2004 and October 2010. The studies included 20,795 patients who were followed for a mean of about 5 years after undergoing surgery for colorectal cancer. Almost 60% (12,242) of these patients received a mean of three units of allogeneic red blood cells (Ann. Surg. 2012;256:235-44).
Patients who were transfused tended to be older, and the transfusion rate was significantly higher in women, those undergoing rectal surgery (compared with those undergoing right or left colon surgery), those with greater surgical blood loss, and those with worsening Duke's stage.
All associations between ABT and the adverse clinical outcomes described below were statistically significant.
The rates of all-cause mortality in the 29 studies that looked at this outcome were 45% among the patients who received blood transfusions vs. 35% of those who did not, a significant difference that represented a 72% increased risk. After adjusting for the duration of the observation period, the investigators found that the annual incidence of all-cause mortality was almost 9% among the transfused patients vs. 6.5% among those who were not transfused.
The rate of cancer-related mortality was 31% vs. 24% in the 17 studies that measured this outcome, a 71% increased risk. After adjusting for the length of observation, the investigators said the annual incidence of cancer-related mortality was 5.4% of those transfused and 4% of those who were not.
The rate of the combined end point of death resulting from recurrence/metastasis was 43% of those who were transfused vs. 33% of those who were not transfused – a 66% increase in risk – in the 19 studies that measured this outcome.
The risk of postoperative infections in the 12 studies that measured this outcome was 29% among those who were transfused vs. 11% of those who were not transfused, which was more than a threefold increased risk. In the two studies that measured the need for surgical reintervention, the risk was increased fourfold among those who were transfused.
In the four studies that measured the length of hospitalization, the hospital stay was a mean of almost 18 days among those who received transfusions, compared with 14 days among those who did not.
Based on evidence in this meta-analysis and other studies showing that preoperative anemia is an independent risk factor for a worse prognosis after colon surgery, and based on the association between ABTs and poorer clinical outcomes in this meta-analysis, the authors wrote that "appropriate blood management measures should, therefore, be given an important place in the care of patients with CRC [colorectal cancer] undergoing elective surgery."
The use of ABT has dropped over the past 25 years because of improvements in patient care, and the authors stated that they believe efforts should be made to further minimize ABT use. Well-designed studies are needed "to determine whether and in which patients preoperative corrective measures of anemia, other than ABTs, will contribute not only in the field of colorectal surgery but also to improve clinical outcomes," they added.
The study was supported by an unrestricted research grant from Vifor Pharma AG, a specialty pharmaceuticals company focused on the treatment of iron deficiency, according to its website. The authors disclosed receiving honoraria and/or travel support for consulting or lecturing for companies that include Vifor, Ethicon Endosurgery, Johnson & Johnson, and AstraZeneca AG; their research departments received grant support from companies that included Vifor.
FROM THE ANNALS OF SURGERY
Major Finding: Adverse clinical outcomes including mortality were significantly increased among patients who had an allogeneic blood transfusion around the time of undergoing colorectal cancer surgery, compared with those who were not transfused.
Data Source: A meta-analysis of 55 mostly retrospective cohort studies evaluated mortality and other clinical outcomes associated with the use of allogeneic red blood cell transfusions in almost 21,000 patients undergoing colorectal cancer surgery, 60% of whom were transfused.
Disclosures: The study was supported by an unrestricted research grant from Vifor Pharma AG. The authors disclosed receiving honoraria and/or travel support for consulting or lecturing from companies that include Vifor, Ethicon Endosurgery, Johnson & Johnson, and AstraZeneca AG; their research departments received grant support from companies including Vifor.
Rivaroxaban Not Approved for ACS Indication
The Food and Drug Administration has declined to approve the oral anticoagulant rivaroxaban as a treatment for patients with acute coronary syndrome, according to a statement issued by Johnson & Johnson.
The agency has issued a complete response letter regarding the supplemental indication for rivaroxaban for use in reducing the risk of secondary cardiovascular events in patients with acute coronary syndrome (ACS) that has been under review at the agency, the statement said.
The FDA issues complete response letters for a drug when there are outstanding issues that need to be resolved before approval; the FDA does not make these letters public, and the company statement did not provide any details about the issues that were raised in the FDA letter.
Rivaroxaban, an oral factor Xa inhibitor marketed as Xarelto by Janssen Pharmaceuticals, a Johnson & Johnson subsidiary, was initially approved in July 2011 for the prophylaxis of deep vein thrombosis in patients undergoing knee or hip replacement surgery; and in November 2011 for reducing the risk of stroke and systemic embolism in patients with nonvalvular atrial fibrillation.
In December of last year, Janssen submitted the ACS application for rivaroxaban at a dose of 2.5 mg twice a day, to "reduce the risk of thrombotic cardiovascular events in patients with ACS [ST-elevation myocardial infarction (STEMI), non-ST-elevation myocardial infarction (NSTEMI), or unstable angina (UA)] in combination with aspirin alone or with aspirin plus clopidogrel or ticlopidine."
But at a meeting in May, the majority of the FDA’s Cardiovascular and Renal Drugs Advisory Committee recommended against approval for this indication, with those voting no citing a large amount of missing data in ATLAS ACS, the pivotal study, as well as safety concerns, among the reasons for their votes (6 to 4 with one abstention).
In the study of 15,526 people with recent ACS, the risk of the combined end point of a composite of cardiovascular death, MI, or stroke, the primary efficacy end point, was reduced by 15% among those on the 2.5-mg twice-daily dosage who were also on aspirin plus a thienopyridine over those on placebo plus dual therapy (primarily driven by a reduction in CV deaths).
This was a statistically significant difference but with a marginal P value of .039. Among those on 2.5 mg twice a day, major bleeding was significantly higher (1.3%) than in those on placebo (0.4%). Intracranial hemorrhage and hemorrhagic stroke rates were also higher in those on rivaroxaban than in those on placebo (N. Engl. J. Med. 2012;366:9-19).
In an interview, Dr. Sanjay Kaul, one of the panel members who voted against approval, referred to the degree of missing data and the lack of robust data in favor of rivaroxaban in this study, pointing out that fewer than 10 excess events in the treatment arm would have negated the statistically significant treatment advantage.
He also referred to the lack of a dose response (there was a greater benefit with the lower dose), a differential impact of the two doses on cardiovascular mortality and MI that was difficult to explain, and the lack of external evidence supporting an incremental advantage of anticoagulant therapy over antiplatelet therapy.
Although the contents of the FDA’s letter are not made public, "I suspect some, if not all, of these issues likely contributed to the unfavorable verdict," said Dr. Kaul, who is the director of the vascular physiology and thrombosis research laboratory at the Burns and Allen Research Institute, Cedars-Sinai Medical Center, Los Angeles.
"I hope that with proper due diligence, which might involve additional trials, the sponsor will be able to respond satisfactorily to the FDA’s concerns," he added.
In the Johnson & Johnson statement, Dr. Paul Burton, vice president and cardiovascular franchise medical leader at Janssen R&D, said that the company "will continue to work with the FDA to fully address their questions as quickly as possible."
Warfarin and three P2Y12 inhibitors – ticagrelor (Brilinta), prasugrel (Effient), and ticlopidine (Ticlid) – are approved for reducing the risk of thrombotic CV events in patients with ACS.
Dr. Kaul holds stock in Johnson & Johnson, but less than the amount for which a waiver to be on an FDA panel is required.
The Food and Drug Administration has declined to approve the oral anticoagulant rivaroxaban as a treatment for patients with acute coronary syndrome, according to a statement issued by Johnson & Johnson.
The agency has issued a complete response letter regarding the supplemental indication for rivaroxaban for use in reducing the risk of secondary cardiovascular events in patients with acute coronary syndrome (ACS) that has been under review at the agency, the statement said.
The FDA issues complete response letters for a drug when there are outstanding issues that need to be resolved before approval; the FDA does not make these letters public, and the company statement did not provide any details about the issues that were raised in the FDA letter.
Rivaroxaban, an oral factor Xa inhibitor marketed as Xarelto by Janssen Pharmaceuticals, a Johnson & Johnson subsidiary, was initially approved in July 2011 for the prophylaxis of deep vein thrombosis in patients undergoing knee or hip replacement surgery; and in November 2011 for reducing the risk of stroke and systemic embolism in patients with nonvalvular atrial fibrillation.
In December of last year, Janssen submitted the ACS application for rivaroxaban at a dose of 2.5 mg twice a day, to "reduce the risk of thrombotic cardiovascular events in patients with ACS [ST-elevation myocardial infarction (STEMI), non-ST-elevation myocardial infarction (NSTEMI), or unstable angina (UA)] in combination with aspirin alone or with aspirin plus clopidogrel or ticlopidine."
But at a meeting in May, the majority of the FDA’s Cardiovascular and Renal Drugs Advisory Committee recommended against approval for this indication, with those voting no citing a large amount of missing data in ATLAS ACS, the pivotal study, as well as safety concerns, among the reasons for their votes (6 to 4 with one abstention).
In the study of 15,526 people with recent ACS, the risk of the combined end point of a composite of cardiovascular death, MI, or stroke, the primary efficacy end point, was reduced by 15% among those on the 2.5-mg twice-daily dosage who were also on aspirin plus a thienopyridine over those on placebo plus dual therapy (primarily driven by a reduction in CV deaths).
This was a statistically significant difference but with a marginal P value of .039. Among those on 2.5 mg twice a day, major bleeding was significantly higher (1.3%) than in those on placebo (0.4%). Intracranial hemorrhage and hemorrhagic stroke rates were also higher in those on rivaroxaban than in those on placebo (N. Engl. J. Med. 2012;366:9-19).
In an interview, Dr. Sanjay Kaul, one of the panel members who voted against approval, referred to the degree of missing data and the lack of robust data in favor of rivaroxaban in this study, pointing out that fewer than 10 excess events in the treatment arm would have negated the statistically significant treatment advantage.
He also referred to the lack of a dose response (there was a greater benefit with the lower dose), a differential impact of the two doses on cardiovascular mortality and MI that was difficult to explain, and the lack of external evidence supporting an incremental advantage of anticoagulant therapy over antiplatelet therapy.
Although the contents of the FDA’s letter are not made public, "I suspect some, if not all, of these issues likely contributed to the unfavorable verdict," said Dr. Kaul, who is the director of the vascular physiology and thrombosis research laboratory at the Burns and Allen Research Institute, Cedars-Sinai Medical Center, Los Angeles.
"I hope that with proper due diligence, which might involve additional trials, the sponsor will be able to respond satisfactorily to the FDA’s concerns," he added.
In the Johnson & Johnson statement, Dr. Paul Burton, vice president and cardiovascular franchise medical leader at Janssen R&D, said that the company "will continue to work with the FDA to fully address their questions as quickly as possible."
Warfarin and three P2Y12 inhibitors – ticagrelor (Brilinta), prasugrel (Effient), and ticlopidine (Ticlid) – are approved for reducing the risk of thrombotic CV events in patients with ACS.
Dr. Kaul holds stock in Johnson & Johnson, but less than the amount for which a waiver to be on an FDA panel is required.
The Food and Drug Administration has declined to approve the oral anticoagulant rivaroxaban as a treatment for patients with acute coronary syndrome, according to a statement issued by Johnson & Johnson.
The agency has issued a complete response letter regarding the supplemental indication for rivaroxaban for use in reducing the risk of secondary cardiovascular events in patients with acute coronary syndrome (ACS) that has been under review at the agency, the statement said.
The FDA issues complete response letters for a drug when there are outstanding issues that need to be resolved before approval; the FDA does not make these letters public, and the company statement did not provide any details about the issues that were raised in the FDA letter.
Rivaroxaban, an oral factor Xa inhibitor marketed as Xarelto by Janssen Pharmaceuticals, a Johnson & Johnson subsidiary, was initially approved in July 2011 for the prophylaxis of deep vein thrombosis in patients undergoing knee or hip replacement surgery; and in November 2011 for reducing the risk of stroke and systemic embolism in patients with nonvalvular atrial fibrillation.
In December of last year, Janssen submitted the ACS application for rivaroxaban at a dose of 2.5 mg twice a day, to "reduce the risk of thrombotic cardiovascular events in patients with ACS [ST-elevation myocardial infarction (STEMI), non-ST-elevation myocardial infarction (NSTEMI), or unstable angina (UA)] in combination with aspirin alone or with aspirin plus clopidogrel or ticlopidine."
But at a meeting in May, the majority of the FDA’s Cardiovascular and Renal Drugs Advisory Committee recommended against approval for this indication, with those voting no citing a large amount of missing data in ATLAS ACS, the pivotal study, as well as safety concerns, among the reasons for their votes (6 to 4 with one abstention).
In the study of 15,526 people with recent ACS, the risk of the combined end point of a composite of cardiovascular death, MI, or stroke, the primary efficacy end point, was reduced by 15% among those on the 2.5-mg twice-daily dosage who were also on aspirin plus a thienopyridine over those on placebo plus dual therapy (primarily driven by a reduction in CV deaths).
This was a statistically significant difference but with a marginal P value of .039. Among those on 2.5 mg twice a day, major bleeding was significantly higher (1.3%) than in those on placebo (0.4%). Intracranial hemorrhage and hemorrhagic stroke rates were also higher in those on rivaroxaban than in those on placebo (N. Engl. J. Med. 2012;366:9-19).
In an interview, Dr. Sanjay Kaul, one of the panel members who voted against approval, referred to the degree of missing data and the lack of robust data in favor of rivaroxaban in this study, pointing out that fewer than 10 excess events in the treatment arm would have negated the statistically significant treatment advantage.
He also referred to the lack of a dose response (there was a greater benefit with the lower dose), a differential impact of the two doses on cardiovascular mortality and MI that was difficult to explain, and the lack of external evidence supporting an incremental advantage of anticoagulant therapy over antiplatelet therapy.
Although the contents of the FDA’s letter are not made public, "I suspect some, if not all, of these issues likely contributed to the unfavorable verdict," said Dr. Kaul, who is the director of the vascular physiology and thrombosis research laboratory at the Burns and Allen Research Institute, Cedars-Sinai Medical Center, Los Angeles.
"I hope that with proper due diligence, which might involve additional trials, the sponsor will be able to respond satisfactorily to the FDA’s concerns," he added.
In the Johnson & Johnson statement, Dr. Paul Burton, vice president and cardiovascular franchise medical leader at Janssen R&D, said that the company "will continue to work with the FDA to fully address their questions as quickly as possible."
Warfarin and three P2Y12 inhibitors – ticagrelor (Brilinta), prasugrel (Effient), and ticlopidine (Ticlid) – are approved for reducing the risk of thrombotic CV events in patients with ACS.
Dr. Kaul holds stock in Johnson & Johnson, but less than the amount for which a waiver to be on an FDA panel is required.
Drug-Drug Interactions Added to Hepatitis C Drug Label
New information about interactions between boceprevir and several other drugs has been added to the prescribing information for the antiviral drug, the Food and Drug Administration announced Aug. 1.
Boceprevir (Victrelis), a protease inhibitor approved for treating hepatitis C in 2011, interacts with cyclosporine, tacrolimus (Prograf), escitalopram (Lexapro), atorvastatin (Lipitor), and pravastatin (Pravachol), according to the FDA statement.
The new information states that, when administered with boceprevir, exposure to atorvastatin increases. When the two drugs are used together, the lowest effective dose of atorvastatin should be used, not to exceed a daily dose of 40 mg, according to the FDA.
Dose adjustments of cyclosporine should be anticipated when it is given with boceprevir, and "should be guided by close monitoring of cyclosporine blood concentrations, and frequent assessments of renal function and cyclosporine-related side effects."
When administered with boceprevir, exposure of escitalopram "was slightly decreased," the statement said. Although selective serotonin reuptake inhibitors (SSRIs) such as escitalopram have a wide therapeutic index, it may be necessary to adjust the dosage when it is administered with boceprevir.
Coadministration of boceprevir with pravastatin increases exposure to pravastatin, but pravastatin can be started at the recommended dosage when coadministered with boceprevir. "Close clinical monitoring is warranted," the statement said.
Giving tacrolimus and boceprevir together "requires significant dose reduction and prolongation of the dosing interval for tacrolimus, with close monitoring of tacrolimus blood concentrations and frequent assessments of renal function and tacrolimus-related side effects," the statement said.
Boceprevir is manufactured in a capsule formulation by Merck Sharp & Dohme Corp., a subsidiary of Merck & Co., and is taken by mouth three times a day.
The drug-drug interaction data are from in vivo drug interaction trials, which the company conducted as part of its postmarketing commitments.
At a meeting in April 2011, an FDA advisory panel enthusiastically supported the approval of boceprevir for treating hepatitis C infection because of the antiviral’s efficacy but emphasized that postmarketing studies on interactions with other drugs, including antidepressants, were needed.
Serious adverse events associated with boceprevir should be reported to MedWatch or by phone at 800-332-1088.
New information about interactions between boceprevir and several other drugs has been added to the prescribing information for the antiviral drug, the Food and Drug Administration announced Aug. 1.
Boceprevir (Victrelis), a protease inhibitor approved for treating hepatitis C in 2011, interacts with cyclosporine, tacrolimus (Prograf), escitalopram (Lexapro), atorvastatin (Lipitor), and pravastatin (Pravachol), according to the FDA statement.
The new information states that, when administered with boceprevir, exposure to atorvastatin increases. When the two drugs are used together, the lowest effective dose of atorvastatin should be used, not to exceed a daily dose of 40 mg, according to the FDA.
Dose adjustments of cyclosporine should be anticipated when it is given with boceprevir, and "should be guided by close monitoring of cyclosporine blood concentrations, and frequent assessments of renal function and cyclosporine-related side effects."
When administered with boceprevir, exposure of escitalopram "was slightly decreased," the statement said. Although selective serotonin reuptake inhibitors (SSRIs) such as escitalopram have a wide therapeutic index, it may be necessary to adjust the dosage when it is administered with boceprevir.
Coadministration of boceprevir with pravastatin increases exposure to pravastatin, but pravastatin can be started at the recommended dosage when coadministered with boceprevir. "Close clinical monitoring is warranted," the statement said.
Giving tacrolimus and boceprevir together "requires significant dose reduction and prolongation of the dosing interval for tacrolimus, with close monitoring of tacrolimus blood concentrations and frequent assessments of renal function and tacrolimus-related side effects," the statement said.
Boceprevir is manufactured in a capsule formulation by Merck Sharp & Dohme Corp., a subsidiary of Merck & Co., and is taken by mouth three times a day.
The drug-drug interaction data are from in vivo drug interaction trials, which the company conducted as part of its postmarketing commitments.
At a meeting in April 2011, an FDA advisory panel enthusiastically supported the approval of boceprevir for treating hepatitis C infection because of the antiviral’s efficacy but emphasized that postmarketing studies on interactions with other drugs, including antidepressants, were needed.
Serious adverse events associated with boceprevir should be reported to MedWatch or by phone at 800-332-1088.
New information about interactions between boceprevir and several other drugs has been added to the prescribing information for the antiviral drug, the Food and Drug Administration announced Aug. 1.
Boceprevir (Victrelis), a protease inhibitor approved for treating hepatitis C in 2011, interacts with cyclosporine, tacrolimus (Prograf), escitalopram (Lexapro), atorvastatin (Lipitor), and pravastatin (Pravachol), according to the FDA statement.
The new information states that, when administered with boceprevir, exposure to atorvastatin increases. When the two drugs are used together, the lowest effective dose of atorvastatin should be used, not to exceed a daily dose of 40 mg, according to the FDA.
Dose adjustments of cyclosporine should be anticipated when it is given with boceprevir, and "should be guided by close monitoring of cyclosporine blood concentrations, and frequent assessments of renal function and cyclosporine-related side effects."
When administered with boceprevir, exposure of escitalopram "was slightly decreased," the statement said. Although selective serotonin reuptake inhibitors (SSRIs) such as escitalopram have a wide therapeutic index, it may be necessary to adjust the dosage when it is administered with boceprevir.
Coadministration of boceprevir with pravastatin increases exposure to pravastatin, but pravastatin can be started at the recommended dosage when coadministered with boceprevir. "Close clinical monitoring is warranted," the statement said.
Giving tacrolimus and boceprevir together "requires significant dose reduction and prolongation of the dosing interval for tacrolimus, with close monitoring of tacrolimus blood concentrations and frequent assessments of renal function and tacrolimus-related side effects," the statement said.
Boceprevir is manufactured in a capsule formulation by Merck Sharp & Dohme Corp., a subsidiary of Merck & Co., and is taken by mouth three times a day.
The drug-drug interaction data are from in vivo drug interaction trials, which the company conducted as part of its postmarketing commitments.
At a meeting in April 2011, an FDA advisory panel enthusiastically supported the approval of boceprevir for treating hepatitis C infection because of the antiviral’s efficacy but emphasized that postmarketing studies on interactions with other drugs, including antidepressants, were needed.
Serious adverse events associated with boceprevir should be reported to MedWatch or by phone at 800-332-1088.
Transfusion Rates Vary Widely at Academic Hospitals
Wide variations in perioperative blood transfusion rates among patients undergoing major noncardiac procedures across U.S. hospitals highlight the need to further investigate evidence-based "transfusion triggers" in this population of surgical patients, according to a study published ahead of print in Annals of Surgery.
"In light of the increased risk of mortality and major complications associated with blood transfusion, the extensive variability in hospital transfusion practice in noncardiac surgery may represent an important opportunity to improve surgical outcomes," wrote Feng Qian, Ph.D., of the University of Rochester (N.Y.), and associates.
The researchers used the University HealthSystem Consortium hospital database to compare transfusion rates of allogeneic red blood cells, fresh frozen plasma, and platelets in patients undergoing elective primary total hip replacement (54,405 patients), colectomy (21,334), or pancreaticoduodenectomy (7,929) at 77 hospitals between June 2006 and September 2010. Most of the hospitals were teaching hospitals with at least 500 beds.
Transfusion rates varied widely before and after adjustment for comorbidities and other patient risk factors. Patients who were treated in hospitals with high rates of transfusions were about twice as likely to receive a blood transfusion as were patients at hospitals with average transfusion rates (Ann. Surg. 2012 July 13[doi:10.1097/SLA.0b013e31825ffc37]).
In hospitals where the transfusion rate for one procedure was high, transfusion rates also tended to be high for the other two procedures. There was some evidence indicating that a higher volume of surgical cases was associated with lower transfusion rates.
After adjusting for patient risk factors, the authors determined that transfusion rates for the different blood components among those undergoing a total hip replacement ranged from 1.3% to almost 75% (red blood cells), from 0.1% to 7.7% (fresh frozen plasma), and from 0.1% to 2% (platelets). Among colectomy patients, transfusion rates ranged from 1.9% to 47.8% (RBCs), from 1.4% to 17.7% (fresh frozen plasma), and from 1.3% to 6.2% (platelets). Among those undergoing a pancreaticoduodenectomy, the rates ranged from 3% to 78.6% (RBCs), from 1% to 47% (fresh frozen plasma), and from 1.4% to 12.6% (platelets).
The variability, the authors said, "reflects, in part, the complexity of the medical decision-making process underlying transfusion therapy." Because the data included patients from 90% of academic medical centers in the United States, the results provide "a broad and contemporary picture of transfusion practices in academic surgical centers" and "reflect transfusion practices that are being taught to the next generation of academic and private-practice clinicians during residency training," they noted.
To the best of their knowledge, the authors said, there are no large randomized studies that have compared liberal and restrictive transfusion strategies in noncardiac surgery patients, and they believe that such trials are "urgently needed to better define evidence-based transfusion triggers for patients undergoing noncardiac surgery."
The study was supported by a grant from the Agency for Healthcare and Quality Research and funding from the department of anesthesiology at the University of Rochester. No disclosures were reported by the authors.
Wide variations in perioperative blood transfusion rates among patients undergoing major noncardiac procedures across U.S. hospitals highlight the need to further investigate evidence-based "transfusion triggers" in this population of surgical patients, according to a study published ahead of print in Annals of Surgery.
"In light of the increased risk of mortality and major complications associated with blood transfusion, the extensive variability in hospital transfusion practice in noncardiac surgery may represent an important opportunity to improve surgical outcomes," wrote Feng Qian, Ph.D., of the University of Rochester (N.Y.), and associates.
The researchers used the University HealthSystem Consortium hospital database to compare transfusion rates of allogeneic red blood cells, fresh frozen plasma, and platelets in patients undergoing elective primary total hip replacement (54,405 patients), colectomy (21,334), or pancreaticoduodenectomy (7,929) at 77 hospitals between June 2006 and September 2010. Most of the hospitals were teaching hospitals with at least 500 beds.
Transfusion rates varied widely before and after adjustment for comorbidities and other patient risk factors. Patients who were treated in hospitals with high rates of transfusions were about twice as likely to receive a blood transfusion as were patients at hospitals with average transfusion rates (Ann. Surg. 2012 July 13[doi:10.1097/SLA.0b013e31825ffc37]).
In hospitals where the transfusion rate for one procedure was high, transfusion rates also tended to be high for the other two procedures. There was some evidence indicating that a higher volume of surgical cases was associated with lower transfusion rates.
After adjusting for patient risk factors, the authors determined that transfusion rates for the different blood components among those undergoing a total hip replacement ranged from 1.3% to almost 75% (red blood cells), from 0.1% to 7.7% (fresh frozen plasma), and from 0.1% to 2% (platelets). Among colectomy patients, transfusion rates ranged from 1.9% to 47.8% (RBCs), from 1.4% to 17.7% (fresh frozen plasma), and from 1.3% to 6.2% (platelets). Among those undergoing a pancreaticoduodenectomy, the rates ranged from 3% to 78.6% (RBCs), from 1% to 47% (fresh frozen plasma), and from 1.4% to 12.6% (platelets).
The variability, the authors said, "reflects, in part, the complexity of the medical decision-making process underlying transfusion therapy." Because the data included patients from 90% of academic medical centers in the United States, the results provide "a broad and contemporary picture of transfusion practices in academic surgical centers" and "reflect transfusion practices that are being taught to the next generation of academic and private-practice clinicians during residency training," they noted.
To the best of their knowledge, the authors said, there are no large randomized studies that have compared liberal and restrictive transfusion strategies in noncardiac surgery patients, and they believe that such trials are "urgently needed to better define evidence-based transfusion triggers for patients undergoing noncardiac surgery."
The study was supported by a grant from the Agency for Healthcare and Quality Research and funding from the department of anesthesiology at the University of Rochester. No disclosures were reported by the authors.
Wide variations in perioperative blood transfusion rates among patients undergoing major noncardiac procedures across U.S. hospitals highlight the need to further investigate evidence-based "transfusion triggers" in this population of surgical patients, according to a study published ahead of print in Annals of Surgery.
"In light of the increased risk of mortality and major complications associated with blood transfusion, the extensive variability in hospital transfusion practice in noncardiac surgery may represent an important opportunity to improve surgical outcomes," wrote Feng Qian, Ph.D., of the University of Rochester (N.Y.), and associates.
The researchers used the University HealthSystem Consortium hospital database to compare transfusion rates of allogeneic red blood cells, fresh frozen plasma, and platelets in patients undergoing elective primary total hip replacement (54,405 patients), colectomy (21,334), or pancreaticoduodenectomy (7,929) at 77 hospitals between June 2006 and September 2010. Most of the hospitals were teaching hospitals with at least 500 beds.
Transfusion rates varied widely before and after adjustment for comorbidities and other patient risk factors. Patients who were treated in hospitals with high rates of transfusions were about twice as likely to receive a blood transfusion as were patients at hospitals with average transfusion rates (Ann. Surg. 2012 July 13[doi:10.1097/SLA.0b013e31825ffc37]).
In hospitals where the transfusion rate for one procedure was high, transfusion rates also tended to be high for the other two procedures. There was some evidence indicating that a higher volume of surgical cases was associated with lower transfusion rates.
After adjusting for patient risk factors, the authors determined that transfusion rates for the different blood components among those undergoing a total hip replacement ranged from 1.3% to almost 75% (red blood cells), from 0.1% to 7.7% (fresh frozen plasma), and from 0.1% to 2% (platelets). Among colectomy patients, transfusion rates ranged from 1.9% to 47.8% (RBCs), from 1.4% to 17.7% (fresh frozen plasma), and from 1.3% to 6.2% (platelets). Among those undergoing a pancreaticoduodenectomy, the rates ranged from 3% to 78.6% (RBCs), from 1% to 47% (fresh frozen plasma), and from 1.4% to 12.6% (platelets).
The variability, the authors said, "reflects, in part, the complexity of the medical decision-making process underlying transfusion therapy." Because the data included patients from 90% of academic medical centers in the United States, the results provide "a broad and contemporary picture of transfusion practices in academic surgical centers" and "reflect transfusion practices that are being taught to the next generation of academic and private-practice clinicians during residency training," they noted.
To the best of their knowledge, the authors said, there are no large randomized studies that have compared liberal and restrictive transfusion strategies in noncardiac surgery patients, and they believe that such trials are "urgently needed to better define evidence-based transfusion triggers for patients undergoing noncardiac surgery."
The study was supported by a grant from the Agency for Healthcare and Quality Research and funding from the department of anesthesiology at the University of Rochester. No disclosures were reported by the authors.
FROM THE ANNALS OF SURGERY
Major Finding: Transfusion rates of red blood cells, fresh frozen plasma, and platelets among patients undergoing noncardiac procedures varied widely across different U.S. academic-affiliated hospitals.
Data Source: Data from a national database of academic medical centers were used to compare transfusions in patients undergoing one of three elective noncardiac surgical procedures at 77 academic hospitals between June 2006 and September 2010.
Disclosures: The study was supported by a grant from the Agency for Healthcare and Quality Research and funding from the department of anesthesiology at the University of Rochester (N.Y.). The authors reported no disclosures.
Twice-Daily Inhaled Anticholinergic Approved for COPD
Aclidinium bromide, a long-acting anticholinergic bronchodilator, has been approved as a treatment for chronic obstructive pulmonary disease, for the long-term maintenance treatment of bronchospasm associated with COPD, including chronic bronchitis and emphysema, the Food and Drug Administration announced on July 23.
The product, a dry powder inhaler used twice a day, will be marketed as Tudorza Pressair by Forest Pharmaceuticals, a subsidiary of Forest Laboratories. A statement released by Forest Laboratories said that the product was expected to become available to wholesalers in the fourth quarter of 2012. The inhaler delivers 60 doses of aclidinium bromide powder for inhalation, the statement said.
Approval was based on three randomized, placebo-controlled, confirmatory clinical trials, which included 1,276 patients aged 40 years and older diagnosed with COPD. At a meeting in February 2012, the majority of the FDA’s Pulmonary-Allergy Drugs Advisory Committee agreed that the data in clinical trials provided evidence that this dose had a clinically meaningful benefit in patients, citing the significant increases in trough forced expiratory volume in 1 second (FEV1) from baseline (the primary efficacy end point) among those treated with 400 mcg twice a day, compared with those on placebo after 12 weeks of treatment in clinical studies.
The most common side effects reported by patients treated with aclidinium bromide included headache, nasopharyngitis, and cough. Serious adverse effects associated with treatment include paradoxical bronchospasm, new or worsened acute narrow-angle glaucoma, or new or worsened urinary retention, according to the FDA statement.
Forest Laboratories licensed the U.S. rights for aclidinium from Almirall, a pharmaceutical company based in Spain. In May, the Committee for Medicinal Products for Human Use of the European Medicines Agency has issued a positive opinion for approval of aclidinium for treating COPD in the E.U., where it will be marketed as Eklira Genuair, according to Almirall.
Aclidinium bromide, a long-acting anticholinergic bronchodilator, has been approved as a treatment for chronic obstructive pulmonary disease, for the long-term maintenance treatment of bronchospasm associated with COPD, including chronic bronchitis and emphysema, the Food and Drug Administration announced on July 23.
The product, a dry powder inhaler used twice a day, will be marketed as Tudorza Pressair by Forest Pharmaceuticals, a subsidiary of Forest Laboratories. A statement released by Forest Laboratories said that the product was expected to become available to wholesalers in the fourth quarter of 2012. The inhaler delivers 60 doses of aclidinium bromide powder for inhalation, the statement said.
Approval was based on three randomized, placebo-controlled, confirmatory clinical trials, which included 1,276 patients aged 40 years and older diagnosed with COPD. At a meeting in February 2012, the majority of the FDA’s Pulmonary-Allergy Drugs Advisory Committee agreed that the data in clinical trials provided evidence that this dose had a clinically meaningful benefit in patients, citing the significant increases in trough forced expiratory volume in 1 second (FEV1) from baseline (the primary efficacy end point) among those treated with 400 mcg twice a day, compared with those on placebo after 12 weeks of treatment in clinical studies.
The most common side effects reported by patients treated with aclidinium bromide included headache, nasopharyngitis, and cough. Serious adverse effects associated with treatment include paradoxical bronchospasm, new or worsened acute narrow-angle glaucoma, or new or worsened urinary retention, according to the FDA statement.
Forest Laboratories licensed the U.S. rights for aclidinium from Almirall, a pharmaceutical company based in Spain. In May, the Committee for Medicinal Products for Human Use of the European Medicines Agency has issued a positive opinion for approval of aclidinium for treating COPD in the E.U., where it will be marketed as Eklira Genuair, according to Almirall.
Aclidinium bromide, a long-acting anticholinergic bronchodilator, has been approved as a treatment for chronic obstructive pulmonary disease, for the long-term maintenance treatment of bronchospasm associated with COPD, including chronic bronchitis and emphysema, the Food and Drug Administration announced on July 23.
The product, a dry powder inhaler used twice a day, will be marketed as Tudorza Pressair by Forest Pharmaceuticals, a subsidiary of Forest Laboratories. A statement released by Forest Laboratories said that the product was expected to become available to wholesalers in the fourth quarter of 2012. The inhaler delivers 60 doses of aclidinium bromide powder for inhalation, the statement said.
Approval was based on three randomized, placebo-controlled, confirmatory clinical trials, which included 1,276 patients aged 40 years and older diagnosed with COPD. At a meeting in February 2012, the majority of the FDA’s Pulmonary-Allergy Drugs Advisory Committee agreed that the data in clinical trials provided evidence that this dose had a clinically meaningful benefit in patients, citing the significant increases in trough forced expiratory volume in 1 second (FEV1) from baseline (the primary efficacy end point) among those treated with 400 mcg twice a day, compared with those on placebo after 12 weeks of treatment in clinical studies.
The most common side effects reported by patients treated with aclidinium bromide included headache, nasopharyngitis, and cough. Serious adverse effects associated with treatment include paradoxical bronchospasm, new or worsened acute narrow-angle glaucoma, or new or worsened urinary retention, according to the FDA statement.
Forest Laboratories licensed the U.S. rights for aclidinium from Almirall, a pharmaceutical company based in Spain. In May, the Committee for Medicinal Products for Human Use of the European Medicines Agency has issued a positive opinion for approval of aclidinium for treating COPD in the E.U., where it will be marketed as Eklira Genuair, according to Almirall.
FDA Reports Seizure Risk With MS Drug
The Food and Drug Administration’s evaluation of postmarketing adverse event reports for the multiple sclerosis drug dalfampridine indicate that the risk of seizures is highest soon after starting treatment with the recommended dose.
Seizures are a known risk of the drug, and the majority of them occurred within days to weeks of starting treatment. Seizures also occurred in patients without any history of them, according to the FDA statement issued on July 23.
The risk of seizures increases with higher blood levels of the drug, and because it is eliminated through the kidneys, the risk of seizures is higher in patients with kidney impairment, the agency said.
The sustained-release formulation of the potassium channel blocker dalfampridine, marketed as Ampyra by Acorda Therapeutics, was approved in January 2010 to improve walking in people with multiple sclerosis, at a dose of 10 mg twice a day. For more than 20 years before approval, a compounded formulation of the drug, called fampridine, was used off label to improve walking ability in people with different neurologic conditions. However, it was difficult to regulate blood levels of fampridine, which has a narrow therapeutic range, and the sustained formulation, dalfampridine, was developed to overcome this limitation.
The FDA statement provided the following recommendations aimed at clinicians, which includes information in the original label (now updated):
• Dalfampridine is contraindicated in patients with a history of seizures or who have moderate to severe renal impairment (a creatine clearance below 50 mL/min).
• The potential benefits of treatment with dalfampridine "should be carefully considered against the risk of seizures" before prescribing the drug to patients with mild renal impairment (creatine clearance 51 mL/min to 80 mL/min).
• Before starting treatment, a patient’s creatinine clearance level should be known (using the Cockroft-Gault equation), and should be checked annually during treatment, "even when serum creatinine levels appear to be normal."
• Patients should be advised not to take double or extra doses of dalfampridine if they miss a dose; and they should take the tablets whole, and not divide, crush, chew, or dissolve the tablets.
• Dalfampridine should be discontinued permanently if a patient has a seizure.
Seizures and other adverse events associated with dalfampridine should be reported to the FDA’s MedWatch program or at 800-332-1088.
The Food and Drug Administration’s evaluation of postmarketing adverse event reports for the multiple sclerosis drug dalfampridine indicate that the risk of seizures is highest soon after starting treatment with the recommended dose.
Seizures are a known risk of the drug, and the majority of them occurred within days to weeks of starting treatment. Seizures also occurred in patients without any history of them, according to the FDA statement issued on July 23.
The risk of seizures increases with higher blood levels of the drug, and because it is eliminated through the kidneys, the risk of seizures is higher in patients with kidney impairment, the agency said.
The sustained-release formulation of the potassium channel blocker dalfampridine, marketed as Ampyra by Acorda Therapeutics, was approved in January 2010 to improve walking in people with multiple sclerosis, at a dose of 10 mg twice a day. For more than 20 years before approval, a compounded formulation of the drug, called fampridine, was used off label to improve walking ability in people with different neurologic conditions. However, it was difficult to regulate blood levels of fampridine, which has a narrow therapeutic range, and the sustained formulation, dalfampridine, was developed to overcome this limitation.
The FDA statement provided the following recommendations aimed at clinicians, which includes information in the original label (now updated):
• Dalfampridine is contraindicated in patients with a history of seizures or who have moderate to severe renal impairment (a creatine clearance below 50 mL/min).
• The potential benefits of treatment with dalfampridine "should be carefully considered against the risk of seizures" before prescribing the drug to patients with mild renal impairment (creatine clearance 51 mL/min to 80 mL/min).
• Before starting treatment, a patient’s creatinine clearance level should be known (using the Cockroft-Gault equation), and should be checked annually during treatment, "even when serum creatinine levels appear to be normal."
• Patients should be advised not to take double or extra doses of dalfampridine if they miss a dose; and they should take the tablets whole, and not divide, crush, chew, or dissolve the tablets.
• Dalfampridine should be discontinued permanently if a patient has a seizure.
Seizures and other adverse events associated with dalfampridine should be reported to the FDA’s MedWatch program or at 800-332-1088.
The Food and Drug Administration’s evaluation of postmarketing adverse event reports for the multiple sclerosis drug dalfampridine indicate that the risk of seizures is highest soon after starting treatment with the recommended dose.
Seizures are a known risk of the drug, and the majority of them occurred within days to weeks of starting treatment. Seizures also occurred in patients without any history of them, according to the FDA statement issued on July 23.
The risk of seizures increases with higher blood levels of the drug, and because it is eliminated through the kidneys, the risk of seizures is higher in patients with kidney impairment, the agency said.
The sustained-release formulation of the potassium channel blocker dalfampridine, marketed as Ampyra by Acorda Therapeutics, was approved in January 2010 to improve walking in people with multiple sclerosis, at a dose of 10 mg twice a day. For more than 20 years before approval, a compounded formulation of the drug, called fampridine, was used off label to improve walking ability in people with different neurologic conditions. However, it was difficult to regulate blood levels of fampridine, which has a narrow therapeutic range, and the sustained formulation, dalfampridine, was developed to overcome this limitation.
The FDA statement provided the following recommendations aimed at clinicians, which includes information in the original label (now updated):
• Dalfampridine is contraindicated in patients with a history of seizures or who have moderate to severe renal impairment (a creatine clearance below 50 mL/min).
• The potential benefits of treatment with dalfampridine "should be carefully considered against the risk of seizures" before prescribing the drug to patients with mild renal impairment (creatine clearance 51 mL/min to 80 mL/min).
• Before starting treatment, a patient’s creatinine clearance level should be known (using the Cockroft-Gault equation), and should be checked annually during treatment, "even when serum creatinine levels appear to be normal."
• Patients should be advised not to take double or extra doses of dalfampridine if they miss a dose; and they should take the tablets whole, and not divide, crush, chew, or dissolve the tablets.
• Dalfampridine should be discontinued permanently if a patient has a seizure.
Seizures and other adverse events associated with dalfampridine should be reported to the FDA’s MedWatch program or at 800-332-1088.
FDA Approves mTOR Inhibitor Everolimus for Breast Cancer
The approval of everolimus has been expanded to include a group of postmenopausal women with advanced hormone receptor–positive, HER2-negative breast cancer, the Food and Drug Administration announced on July 20.
A mammalian target of rapamycin (mTOR) inhibitor marketed as Afinitor, everolimus is indicated in combination with exemestane (Aromasin) for this group of women when they have had recurrence or progression of disease after treatment with letrozole (Femara) or anastrozole (Arimidex).
The drug becomes the first mTOR inhibitor approved for advanced hormone receptor–positive breast cancer. Exemestane, letrozole, and anastrozole are aromatase inhibitors, a class of drugs that has become an alternative to tamoxifen for adjuvant treatment of women with estrogen receptor– and/or progesterone receptor–positive breast cancer.
The new approval is based on the phase III BOLERO-2 trial, a randomized, double-blind, placebo-controlled study of 724 postmenopausal women who matched the population in the new indication.
Median progression-free survival, the primary end point, more than doubled among those treated with everolimus (the approved tablet dose of 10 mg per day) plus exemestane (25 mg per day). The median reached 7.8 months in this group, vs. 3.2 months among those treated with exemestane (25 mg per day) and placebo, a difference that was highly significant statistically.
The objective response rate was 12.6% among those on the combination, compared with 1.7% in the control group. There were 3 complete responses (0.6%) and 58 partial responses (12%) in women who received everolimus, compared with no complete responses and 4 partial responses (1.7%) among those given placebo.
Overall survival results "were not mature at the time of the interim analysis, and no statistically significant treatment-related difference in OS was noted," according to the prescribing information.
The most common adverse effects reported in patients treated with everolimus included stomatitis, infections, rash, fatigue, diarrhea, and decreased appetite. Adverse events are more common among patients aged 65 years and older, who should be monitored closely, according to the FDA statement announcing the approval.
Everolimus was first approved in 2009 as a treatment for advanced renal cell carcinoma that has progressed after treatment with other cancer therapies. This was followed by approvals for adults with progressive advanced neuroendocrine tumors of pancreatic origin, patients with renal angiomyolipoma and tuberous sclerosis complex (TSC) not requiring immediate surgery, and adults and children with subependymal giant cell astrocytoma associated with TSC who require treatment but are not candidates for curative surgery.
"This approval redefines the treatment and management of advanced hormone receptor–positive breast cancer, offering a critical new option for physicians and patients," Dr. Gabriel Hortobagyi, chair of Breast Medical Oncology, at the University of Texas M.D. Anderson Cancer Center, said in a statement released by Novartis Pharmaceuticals Corp., which markets everolimus. The company noted that two ongoing phase III trials are studying everolimus in HER2-positive breast cancer.
The approval of everolimus has been expanded to include a group of postmenopausal women with advanced hormone receptor–positive, HER2-negative breast cancer, the Food and Drug Administration announced on July 20.
A mammalian target of rapamycin (mTOR) inhibitor marketed as Afinitor, everolimus is indicated in combination with exemestane (Aromasin) for this group of women when they have had recurrence or progression of disease after treatment with letrozole (Femara) or anastrozole (Arimidex).
The drug becomes the first mTOR inhibitor approved for advanced hormone receptor–positive breast cancer. Exemestane, letrozole, and anastrozole are aromatase inhibitors, a class of drugs that has become an alternative to tamoxifen for adjuvant treatment of women with estrogen receptor– and/or progesterone receptor–positive breast cancer.
The new approval is based on the phase III BOLERO-2 trial, a randomized, double-blind, placebo-controlled study of 724 postmenopausal women who matched the population in the new indication.
Median progression-free survival, the primary end point, more than doubled among those treated with everolimus (the approved tablet dose of 10 mg per day) plus exemestane (25 mg per day). The median reached 7.8 months in this group, vs. 3.2 months among those treated with exemestane (25 mg per day) and placebo, a difference that was highly significant statistically.
The objective response rate was 12.6% among those on the combination, compared with 1.7% in the control group. There were 3 complete responses (0.6%) and 58 partial responses (12%) in women who received everolimus, compared with no complete responses and 4 partial responses (1.7%) among those given placebo.
Overall survival results "were not mature at the time of the interim analysis, and no statistically significant treatment-related difference in OS was noted," according to the prescribing information.
The most common adverse effects reported in patients treated with everolimus included stomatitis, infections, rash, fatigue, diarrhea, and decreased appetite. Adverse events are more common among patients aged 65 years and older, who should be monitored closely, according to the FDA statement announcing the approval.
Everolimus was first approved in 2009 as a treatment for advanced renal cell carcinoma that has progressed after treatment with other cancer therapies. This was followed by approvals for adults with progressive advanced neuroendocrine tumors of pancreatic origin, patients with renal angiomyolipoma and tuberous sclerosis complex (TSC) not requiring immediate surgery, and adults and children with subependymal giant cell astrocytoma associated with TSC who require treatment but are not candidates for curative surgery.
"This approval redefines the treatment and management of advanced hormone receptor–positive breast cancer, offering a critical new option for physicians and patients," Dr. Gabriel Hortobagyi, chair of Breast Medical Oncology, at the University of Texas M.D. Anderson Cancer Center, said in a statement released by Novartis Pharmaceuticals Corp., which markets everolimus. The company noted that two ongoing phase III trials are studying everolimus in HER2-positive breast cancer.
The approval of everolimus has been expanded to include a group of postmenopausal women with advanced hormone receptor–positive, HER2-negative breast cancer, the Food and Drug Administration announced on July 20.
A mammalian target of rapamycin (mTOR) inhibitor marketed as Afinitor, everolimus is indicated in combination with exemestane (Aromasin) for this group of women when they have had recurrence or progression of disease after treatment with letrozole (Femara) or anastrozole (Arimidex).
The drug becomes the first mTOR inhibitor approved for advanced hormone receptor–positive breast cancer. Exemestane, letrozole, and anastrozole are aromatase inhibitors, a class of drugs that has become an alternative to tamoxifen for adjuvant treatment of women with estrogen receptor– and/or progesterone receptor–positive breast cancer.
The new approval is based on the phase III BOLERO-2 trial, a randomized, double-blind, placebo-controlled study of 724 postmenopausal women who matched the population in the new indication.
Median progression-free survival, the primary end point, more than doubled among those treated with everolimus (the approved tablet dose of 10 mg per day) plus exemestane (25 mg per day). The median reached 7.8 months in this group, vs. 3.2 months among those treated with exemestane (25 mg per day) and placebo, a difference that was highly significant statistically.
The objective response rate was 12.6% among those on the combination, compared with 1.7% in the control group. There were 3 complete responses (0.6%) and 58 partial responses (12%) in women who received everolimus, compared with no complete responses and 4 partial responses (1.7%) among those given placebo.
Overall survival results "were not mature at the time of the interim analysis, and no statistically significant treatment-related difference in OS was noted," according to the prescribing information.
The most common adverse effects reported in patients treated with everolimus included stomatitis, infections, rash, fatigue, diarrhea, and decreased appetite. Adverse events are more common among patients aged 65 years and older, who should be monitored closely, according to the FDA statement announcing the approval.
Everolimus was first approved in 2009 as a treatment for advanced renal cell carcinoma that has progressed after treatment with other cancer therapies. This was followed by approvals for adults with progressive advanced neuroendocrine tumors of pancreatic origin, patients with renal angiomyolipoma and tuberous sclerosis complex (TSC) not requiring immediate surgery, and adults and children with subependymal giant cell astrocytoma associated with TSC who require treatment but are not candidates for curative surgery.
"This approval redefines the treatment and management of advanced hormone receptor–positive breast cancer, offering a critical new option for physicians and patients," Dr. Gabriel Hortobagyi, chair of Breast Medical Oncology, at the University of Texas M.D. Anderson Cancer Center, said in a statement released by Novartis Pharmaceuticals Corp., which markets everolimus. The company noted that two ongoing phase III trials are studying everolimus in HER2-positive breast cancer.
Carfilzomib Given the Go-Ahead for Multiple Myeloma
Carfilzomib, a second-generation proteasome inhibitor administered intravenously, has been approved to treat patients with refractory multiple myeloma, the Food and Drug Administration announced on July 20.
The indication specifies use in patients who have received at least two prior lines of therapy that included the first-generation proteasome inhibitor bortezomib (Velcade) and an immunomodulatory drug (IMiD), and who have evidence of disease progression on or within 60 days of completing the last therapy,
Bortezomib was approved in 2003, and is among the new agents credited with prolonging the lives of patients with multiple myeloma. Carfilzomib will be marketed as Kyprolis by Onyx Pharmaceuticals. In clinical trials, it was effective in patients who stopped responding to bortezomib and to IMiDs, and it also appeared to cause less peripheral neuropathy.
The prescribing information for carfilzomib states that approval was based on response rate, and that clinical benefit, such as improvement in survival or symptoms, "has not been verified."
Carfilzomib received an accelerated approval, based on clinical evidence that the treatment has an effect on a surrogate end point that is "reasonably likely to predict a clinical benefit to patients," according to the Food and Drug Administration. As a condition of accelerated approval, manufacturers are required to provide more clinical data confirming benefit, and if the follow-up studies fail to confirm benefit, the FDA can withdraw approval.
Onyx announced that it has completed enrollment in a phase III confirmatory study.
The accelerated approval was based on a phase IIb study of 266 patients with relapsed or refractory multiple myeloma, previously treated with at least two therapies, including bortezomib and an IMiD – thalidomide or lenalidomide (Revlimid). The patients started treatment with carfilzomib (administered twice weekly for 3 weeks, followed by a rest period, in a 28-day cycle) a median of 5.4 years after the initial diagnosis.
The overall response rate (complete responses, very good partial responses, and partial responses combined) was 23%, and the median duration of response was almost 8 months. Most responses were partial (18%), but one patient had a complete response.
The most commonly reported adverse effects associated with treatment were anemia, nausea, thrombocytopenia, dyspnea, diarrhea, and pyrexia, which were reported in 30% or more of patients in the study, according to the FDA. Serious adverse events were heart failure and shortness of breath.
At a meeting in June, the FDA’s Oncologic Drugs Advisory Committee supported accelerated approval of the drug for this population, noting that there are few if any treatment options available for patients with end-stage multiple myeloma, and that the response rate and safety profile observed in the phase II study were acceptable and justified an accelerated approval.
The confirmatory study, an international phase III study known as the ASPIRE study, is evaluating the combination of lenalidomide and low-dose dexamethasone with or without carfilzomib in patients with relapsed multiple myeloma who have received one to three previous therapies, according to the statement from Onyx announcing the approval.
Other studies that are underway include a phase III study evaluating carfilzomib as a single treatment for patients with relapsed and refractory myeloma who have received three or more prior therapies (FOCUS trial), which the company said is designed to "facilitate" approvals worldwide.
Another study, the ENDEAVOR trial, is comparing the combination of carfilzomib and low-dose dexamethasone to the combination of bortezomib and low-dose dexamethasone, according to Onyx.
The FDA statement cited American Cancer Society estimates that 21,700 people will be diagnosed with multiple myeloma and 10,710 will die from the disease in 2012.
"We are encouraged by the continued progress in the development of drugs for multiple myeloma over the past decade, offering improved treatment of this disease," Dr. Richard Pazdur, director of the Office of Hematology and Oncology Products in the FDA’s Center for Drug Evaluation and Research, said in a news release.
The Multiple Myeloma Research Foundation issued a statement commending the FDA and Onyx for making carfilzomib available to patients with multiple myeloma. The foundation and its affiliated clinical trials network, the Multiple Myeloma Research Consortium, had helped Onyx with the pivotal clinical trial.
Kathy Giusti, founder and chief executive officer of the groups, called the approval "an immensely important milestone for the multiple myeloma patient community, which continues to face significant unmet need in terms of safe and effective treatments for advanced disease. While we have seen tremendous progress in the past decade, multiple myeloma remains incurable," she said.
The carfilzomib prescribing information is available here.
Carfilzomib, a second-generation proteasome inhibitor administered intravenously, has been approved to treat patients with refractory multiple myeloma, the Food and Drug Administration announced on July 20.
The indication specifies use in patients who have received at least two prior lines of therapy that included the first-generation proteasome inhibitor bortezomib (Velcade) and an immunomodulatory drug (IMiD), and who have evidence of disease progression on or within 60 days of completing the last therapy,
Bortezomib was approved in 2003, and is among the new agents credited with prolonging the lives of patients with multiple myeloma. Carfilzomib will be marketed as Kyprolis by Onyx Pharmaceuticals. In clinical trials, it was effective in patients who stopped responding to bortezomib and to IMiDs, and it also appeared to cause less peripheral neuropathy.
The prescribing information for carfilzomib states that approval was based on response rate, and that clinical benefit, such as improvement in survival or symptoms, "has not been verified."
Carfilzomib received an accelerated approval, based on clinical evidence that the treatment has an effect on a surrogate end point that is "reasonably likely to predict a clinical benefit to patients," according to the Food and Drug Administration. As a condition of accelerated approval, manufacturers are required to provide more clinical data confirming benefit, and if the follow-up studies fail to confirm benefit, the FDA can withdraw approval.
Onyx announced that it has completed enrollment in a phase III confirmatory study.
The accelerated approval was based on a phase IIb study of 266 patients with relapsed or refractory multiple myeloma, previously treated with at least two therapies, including bortezomib and an IMiD – thalidomide or lenalidomide (Revlimid). The patients started treatment with carfilzomib (administered twice weekly for 3 weeks, followed by a rest period, in a 28-day cycle) a median of 5.4 years after the initial diagnosis.
The overall response rate (complete responses, very good partial responses, and partial responses combined) was 23%, and the median duration of response was almost 8 months. Most responses were partial (18%), but one patient had a complete response.
The most commonly reported adverse effects associated with treatment were anemia, nausea, thrombocytopenia, dyspnea, diarrhea, and pyrexia, which were reported in 30% or more of patients in the study, according to the FDA. Serious adverse events were heart failure and shortness of breath.
At a meeting in June, the FDA’s Oncologic Drugs Advisory Committee supported accelerated approval of the drug for this population, noting that there are few if any treatment options available for patients with end-stage multiple myeloma, and that the response rate and safety profile observed in the phase II study were acceptable and justified an accelerated approval.
The confirmatory study, an international phase III study known as the ASPIRE study, is evaluating the combination of lenalidomide and low-dose dexamethasone with or without carfilzomib in patients with relapsed multiple myeloma who have received one to three previous therapies, according to the statement from Onyx announcing the approval.
Other studies that are underway include a phase III study evaluating carfilzomib as a single treatment for patients with relapsed and refractory myeloma who have received three or more prior therapies (FOCUS trial), which the company said is designed to "facilitate" approvals worldwide.
Another study, the ENDEAVOR trial, is comparing the combination of carfilzomib and low-dose dexamethasone to the combination of bortezomib and low-dose dexamethasone, according to Onyx.
The FDA statement cited American Cancer Society estimates that 21,700 people will be diagnosed with multiple myeloma and 10,710 will die from the disease in 2012.
"We are encouraged by the continued progress in the development of drugs for multiple myeloma over the past decade, offering improved treatment of this disease," Dr. Richard Pazdur, director of the Office of Hematology and Oncology Products in the FDA’s Center for Drug Evaluation and Research, said in a news release.
The Multiple Myeloma Research Foundation issued a statement commending the FDA and Onyx for making carfilzomib available to patients with multiple myeloma. The foundation and its affiliated clinical trials network, the Multiple Myeloma Research Consortium, had helped Onyx with the pivotal clinical trial.
Kathy Giusti, founder and chief executive officer of the groups, called the approval "an immensely important milestone for the multiple myeloma patient community, which continues to face significant unmet need in terms of safe and effective treatments for advanced disease. While we have seen tremendous progress in the past decade, multiple myeloma remains incurable," she said.
The carfilzomib prescribing information is available here.
Carfilzomib, a second-generation proteasome inhibitor administered intravenously, has been approved to treat patients with refractory multiple myeloma, the Food and Drug Administration announced on July 20.
The indication specifies use in patients who have received at least two prior lines of therapy that included the first-generation proteasome inhibitor bortezomib (Velcade) and an immunomodulatory drug (IMiD), and who have evidence of disease progression on or within 60 days of completing the last therapy,
Bortezomib was approved in 2003, and is among the new agents credited with prolonging the lives of patients with multiple myeloma. Carfilzomib will be marketed as Kyprolis by Onyx Pharmaceuticals. In clinical trials, it was effective in patients who stopped responding to bortezomib and to IMiDs, and it also appeared to cause less peripheral neuropathy.
The prescribing information for carfilzomib states that approval was based on response rate, and that clinical benefit, such as improvement in survival or symptoms, "has not been verified."
Carfilzomib received an accelerated approval, based on clinical evidence that the treatment has an effect on a surrogate end point that is "reasonably likely to predict a clinical benefit to patients," according to the Food and Drug Administration. As a condition of accelerated approval, manufacturers are required to provide more clinical data confirming benefit, and if the follow-up studies fail to confirm benefit, the FDA can withdraw approval.
Onyx announced that it has completed enrollment in a phase III confirmatory study.
The accelerated approval was based on a phase IIb study of 266 patients with relapsed or refractory multiple myeloma, previously treated with at least two therapies, including bortezomib and an IMiD – thalidomide or lenalidomide (Revlimid). The patients started treatment with carfilzomib (administered twice weekly for 3 weeks, followed by a rest period, in a 28-day cycle) a median of 5.4 years after the initial diagnosis.
The overall response rate (complete responses, very good partial responses, and partial responses combined) was 23%, and the median duration of response was almost 8 months. Most responses were partial (18%), but one patient had a complete response.
The most commonly reported adverse effects associated with treatment were anemia, nausea, thrombocytopenia, dyspnea, diarrhea, and pyrexia, which were reported in 30% or more of patients in the study, according to the FDA. Serious adverse events were heart failure and shortness of breath.
At a meeting in June, the FDA’s Oncologic Drugs Advisory Committee supported accelerated approval of the drug for this population, noting that there are few if any treatment options available for patients with end-stage multiple myeloma, and that the response rate and safety profile observed in the phase II study were acceptable and justified an accelerated approval.
The confirmatory study, an international phase III study known as the ASPIRE study, is evaluating the combination of lenalidomide and low-dose dexamethasone with or without carfilzomib in patients with relapsed multiple myeloma who have received one to three previous therapies, according to the statement from Onyx announcing the approval.
Other studies that are underway include a phase III study evaluating carfilzomib as a single treatment for patients with relapsed and refractory myeloma who have received three or more prior therapies (FOCUS trial), which the company said is designed to "facilitate" approvals worldwide.
Another study, the ENDEAVOR trial, is comparing the combination of carfilzomib and low-dose dexamethasone to the combination of bortezomib and low-dose dexamethasone, according to Onyx.
The FDA statement cited American Cancer Society estimates that 21,700 people will be diagnosed with multiple myeloma and 10,710 will die from the disease in 2012.
"We are encouraged by the continued progress in the development of drugs for multiple myeloma over the past decade, offering improved treatment of this disease," Dr. Richard Pazdur, director of the Office of Hematology and Oncology Products in the FDA’s Center for Drug Evaluation and Research, said in a news release.
The Multiple Myeloma Research Foundation issued a statement commending the FDA and Onyx for making carfilzomib available to patients with multiple myeloma. The foundation and its affiliated clinical trials network, the Multiple Myeloma Research Consortium, had helped Onyx with the pivotal clinical trial.
Kathy Giusti, founder and chief executive officer of the groups, called the approval "an immensely important milestone for the multiple myeloma patient community, which continues to face significant unmet need in terms of safe and effective treatments for advanced disease. While we have seen tremendous progress in the past decade, multiple myeloma remains incurable," she said.
The carfilzomib prescribing information is available here.
Pertussis Epidemic in Washington: Teen Booster, Adult Vaccination Key
The current pertussis epidemic in Washington state and increased rates nationwide underline the critical importance of vaccinating pregnant women and other adults who are in contact with babies against pertussis, Dr. Anne Schuchat said during a Centers for Disease Control and Prevention telebriefing.
Reports of pertussis have been increasing in infants, as well as among children aged 10, 13, and 14 years in Washington and nationwide, making the recommended booster for children with the Tdap vaccine essential, said Dr. Schuchat, director of the National Center for Immunization and Respiratory Diseases at the CDC in Atlanta.
Pertussis vaccination "remains the single most effective approach to preventing infection," she said, noting that unvaccinated children are at an eightfold greater risk of getting pertussis than those who have been fully vaccinated with DTaP. And vaccinated children who do contract pertussis typically have milder cases.
In 2010, only 8% of adults had any history of having received a Tdap booster, she said.
In addition to encouraging patients to get the vaccine, clinicians also are being urged to consider pertussis as a possible diagnosis in their patients who have a persistent cough, she said.
Since the middle of 2011, cases of pertussis have been increasing in Washington state, and in April 2012, a pertussis epidemic was declared when 640 cases had been reported. As of June 16, 2,520 cases had been reported in 2012, a 1,300% increase from the same time period the year before, and the highest number of cases reported since the early 1940s (MMWR 2012;61:517-22).
To date, nine babies in the United States have died of whooping cough (one more than reported in the MMWR), Dr. Schuchat reported.
A higher-than-expected number of cases also have been reported in other states, with similar trends in the age groups affected, "and there may be many more coming," Dr. Schuchat said. In 2010, more than 27,000 cases were reported nationally, with 27 deaths (25 in infants).
Pertussis outbreaks occur in waves, with peaks every 3-5 years. During this current wave, the highest rates have been in infants under 1 year of age, with over half in babies under 3 months. These children are too young to be protected with the vaccine that is first administered at age 2 months, so they are dependent on the immunity of people around them, which is why pregnant women and adults who are around babies are being urged to get vaccinated, she said.
The rates of pertussis have started to increase after late childhood, with a higher rate in 10-year-olds, but then the rate decreases in 11- to 12-year-olds, and then increases among 13-year-olds, Dr. Schuchat said. These trends also point to the importance of the recommended Tdap vaccine in those aged 11 and 12 years .
The increase in pertussis cases reported in adolescents aged 13-14 in Washington state and nationally is a concern, and possible causes are being studied further. A contributing factor could be the switch from the whole cell to the acellular vaccine, which may have an effect on how long immunity persists. "Waning of protection over time may be part of the story," she said, pointing out that unvaccinated people are not thought to be driving this current wave of infection.
Dr. Schuchat said that the increase in pertussis cases goes beyond Washington State. "We really think the disease is spreading around the country. ...We’re in for a tough year and a tough couple of years."
The current pertussis epidemic in Washington state and increased rates nationwide underline the critical importance of vaccinating pregnant women and other adults who are in contact with babies against pertussis, Dr. Anne Schuchat said during a Centers for Disease Control and Prevention telebriefing.
Reports of pertussis have been increasing in infants, as well as among children aged 10, 13, and 14 years in Washington and nationwide, making the recommended booster for children with the Tdap vaccine essential, said Dr. Schuchat, director of the National Center for Immunization and Respiratory Diseases at the CDC in Atlanta.
Pertussis vaccination "remains the single most effective approach to preventing infection," she said, noting that unvaccinated children are at an eightfold greater risk of getting pertussis than those who have been fully vaccinated with DTaP. And vaccinated children who do contract pertussis typically have milder cases.
In 2010, only 8% of adults had any history of having received a Tdap booster, she said.
In addition to encouraging patients to get the vaccine, clinicians also are being urged to consider pertussis as a possible diagnosis in their patients who have a persistent cough, she said.
Since the middle of 2011, cases of pertussis have been increasing in Washington state, and in April 2012, a pertussis epidemic was declared when 640 cases had been reported. As of June 16, 2,520 cases had been reported in 2012, a 1,300% increase from the same time period the year before, and the highest number of cases reported since the early 1940s (MMWR 2012;61:517-22).
To date, nine babies in the United States have died of whooping cough (one more than reported in the MMWR), Dr. Schuchat reported.
A higher-than-expected number of cases also have been reported in other states, with similar trends in the age groups affected, "and there may be many more coming," Dr. Schuchat said. In 2010, more than 27,000 cases were reported nationally, with 27 deaths (25 in infants).
Pertussis outbreaks occur in waves, with peaks every 3-5 years. During this current wave, the highest rates have been in infants under 1 year of age, with over half in babies under 3 months. These children are too young to be protected with the vaccine that is first administered at age 2 months, so they are dependent on the immunity of people around them, which is why pregnant women and adults who are around babies are being urged to get vaccinated, she said.
The rates of pertussis have started to increase after late childhood, with a higher rate in 10-year-olds, but then the rate decreases in 11- to 12-year-olds, and then increases among 13-year-olds, Dr. Schuchat said. These trends also point to the importance of the recommended Tdap vaccine in those aged 11 and 12 years .
The increase in pertussis cases reported in adolescents aged 13-14 in Washington state and nationally is a concern, and possible causes are being studied further. A contributing factor could be the switch from the whole cell to the acellular vaccine, which may have an effect on how long immunity persists. "Waning of protection over time may be part of the story," she said, pointing out that unvaccinated people are not thought to be driving this current wave of infection.
Dr. Schuchat said that the increase in pertussis cases goes beyond Washington State. "We really think the disease is spreading around the country. ...We’re in for a tough year and a tough couple of years."
The current pertussis epidemic in Washington state and increased rates nationwide underline the critical importance of vaccinating pregnant women and other adults who are in contact with babies against pertussis, Dr. Anne Schuchat said during a Centers for Disease Control and Prevention telebriefing.
Reports of pertussis have been increasing in infants, as well as among children aged 10, 13, and 14 years in Washington and nationwide, making the recommended booster for children with the Tdap vaccine essential, said Dr. Schuchat, director of the National Center for Immunization and Respiratory Diseases at the CDC in Atlanta.
Pertussis vaccination "remains the single most effective approach to preventing infection," she said, noting that unvaccinated children are at an eightfold greater risk of getting pertussis than those who have been fully vaccinated with DTaP. And vaccinated children who do contract pertussis typically have milder cases.
In 2010, only 8% of adults had any history of having received a Tdap booster, she said.
In addition to encouraging patients to get the vaccine, clinicians also are being urged to consider pertussis as a possible diagnosis in their patients who have a persistent cough, she said.
Since the middle of 2011, cases of pertussis have been increasing in Washington state, and in April 2012, a pertussis epidemic was declared when 640 cases had been reported. As of June 16, 2,520 cases had been reported in 2012, a 1,300% increase from the same time period the year before, and the highest number of cases reported since the early 1940s (MMWR 2012;61:517-22).
To date, nine babies in the United States have died of whooping cough (one more than reported in the MMWR), Dr. Schuchat reported.
A higher-than-expected number of cases also have been reported in other states, with similar trends in the age groups affected, "and there may be many more coming," Dr. Schuchat said. In 2010, more than 27,000 cases were reported nationally, with 27 deaths (25 in infants).
Pertussis outbreaks occur in waves, with peaks every 3-5 years. During this current wave, the highest rates have been in infants under 1 year of age, with over half in babies under 3 months. These children are too young to be protected with the vaccine that is first administered at age 2 months, so they are dependent on the immunity of people around them, which is why pregnant women and adults who are around babies are being urged to get vaccinated, she said.
The rates of pertussis have started to increase after late childhood, with a higher rate in 10-year-olds, but then the rate decreases in 11- to 12-year-olds, and then increases among 13-year-olds, Dr. Schuchat said. These trends also point to the importance of the recommended Tdap vaccine in those aged 11 and 12 years .
The increase in pertussis cases reported in adolescents aged 13-14 in Washington state and nationally is a concern, and possible causes are being studied further. A contributing factor could be the switch from the whole cell to the acellular vaccine, which may have an effect on how long immunity persists. "Waning of protection over time may be part of the story," she said, pointing out that unvaccinated people are not thought to be driving this current wave of infection.
Dr. Schuchat said that the increase in pertussis cases goes beyond Washington State. "We really think the disease is spreading around the country. ...We’re in for a tough year and a tough couple of years."
Drug Twosome Protects Against Nausea and Vomiting
BALTIMORE – Starting treatment with a combination of doxylamine and pyridoxine before the onset of nausea and vomiting significantly reduced the incidence of severe nausea and vomiting of pregnancy, compared with starting treatment at the first sign of symptoms, in a study of 59 women with a history of severe nausea and vomiting of pregnancy, Caroline Maltepe reported.
In the prospective, randomized, open-label study of women with a history of nausea and vomiting of pregnancy (NVP), 30 women began taking the delayed-release combination of 10 mg of doxylamine and 10 mg of pyridoxine (vitamin B6) as soon as they learned they were pregnant, before they started to experience nausea and vomiting (at a mean of 4 weeks’ gestation). Another 29 pregnant women started taking the combination when they started to experience symptoms, Ms. Maltepe said at the annual meeting of the Teratology Society.
Women in both groups started to experience NVP symptoms at a mean of about 5 to almost 6 weeks, said Ms. Maltepe, the lead author of the study, who is with the Motherisk Program at the Hospital for Sick Children in Toronto. The program conducts research and provides information about the safety of maternal exposure to drugs, chemicals, diseases, radiation, and environmental agents to the developing fetus or infant.
The women (mean age, 31-32 years) had experienced severe NVP or hyperemesis gravidarum (HG) in their previous pregnancy, and enrolled when they were planning a pregnancy or in early pregnancy at a point when they had not yet experienced any symptoms. Women in both groups took 2-9 tablets of the doxylamine-pyridoxine combination a day, and received counseling and follow-up calls.
In Canada, the combination is approved and marketed as Diclectin, by Duchesnay, and is the only drug labeled for treating NVP in Canada. There it is considered a first-line treatment and is taken at a standard dose of four tablets a day, said Ms. Maltepe, the coordinator of the NVP helpline at Motherisk.
Diclectin is a generic form of the drug Bendectin, which was marketed in the United States until 1983, when it was voluntarily withdrawn by its manufacturer, Merrell Dow Pharmaceuticals, after a series of lawsuits claiming it caused birth defects. For several years, Duchesnay has been working toward Food and Drug Administration clearance to market Diclectin in the United States, but it would be premature to predict when it would become available in the United States, a spokesman for the company said.
In the study, symptoms were evaluated using the PUQE (Pregnancy–Unique Quantification of Emesis and Nausea) score, a validated scoring system that takes into account the frequency of vomiting; the frequency of retching, gagging, or dry heaves; and the number of hours spent feeling nauseous over a 24-hour period. The score was developed by Dr. Gideon Koren, director of Motherisk and coauthor of this study, and his associates. A score of 7-12 is considered moderate; a score of 13 or higher is considered severe.
In the study, preemptive treatment was associated with 70% fewer cases of moderate to severe NVP during the first 3 weeks women experienced symptoms: 4 of the 26 (15%) evaluable women had a PUQE score of 11 or more during the first 3 weeks of NVP symptoms, compared with 9 of the 23 (39%) evaluable women in the control group, a significant difference. Of the 19 women in the preemptively treated group who had HG during the previous pregnancy, 6 (32%) experienced HG, compared with 6 of the 11 (55%) in the control group, also a significant difference. Some women who enrolled were not evaluable for reasons that included having a miscarriage.
NVP resolved before labor in significantly more of the women in the preemptively treated group than in the control group (almost 80% vs. about 50%), and there was a negative correlation between PUQE scores and a well-being score, indicating that treatment is associated with improved quality of life for these patients, Ms. Maltepe said.
In women who have experienced severe NVP or HG in a previous pregnancy, preemptive treatment with doxylamine-pyridoxine events can reduce the recurrence of NVP in subsequent pregnancies, and improve their quality of life during pregnancy, as well as reduce time off from work and the need for enteral and parenteral therapy, hospitalization, and other costs associated with severe NVP, she concluded.
The NVP helpline at Motherisk is the only dedicated NVP helpline in the world, according to Ms. Maltepe, who provides evidence-based recommendations on pharmaceutical and nonpharmaceutical approaches to treating NVP for pregnant women, their family members, and clinicians via the helpline (800-436-8477 in North America).
The study was funded by Duchesnay. Ms. Maltepe and her coauthors said they had no relevant conflict of interest to disclose.
The Motherisk site is available at www.motherisk.org/women/index.jsp.
BALTIMORE – Starting treatment with a combination of doxylamine and pyridoxine before the onset of nausea and vomiting significantly reduced the incidence of severe nausea and vomiting of pregnancy, compared with starting treatment at the first sign of symptoms, in a study of 59 women with a history of severe nausea and vomiting of pregnancy, Caroline Maltepe reported.
In the prospective, randomized, open-label study of women with a history of nausea and vomiting of pregnancy (NVP), 30 women began taking the delayed-release combination of 10 mg of doxylamine and 10 mg of pyridoxine (vitamin B6) as soon as they learned they were pregnant, before they started to experience nausea and vomiting (at a mean of 4 weeks’ gestation). Another 29 pregnant women started taking the combination when they started to experience symptoms, Ms. Maltepe said at the annual meeting of the Teratology Society.
Women in both groups started to experience NVP symptoms at a mean of about 5 to almost 6 weeks, said Ms. Maltepe, the lead author of the study, who is with the Motherisk Program at the Hospital for Sick Children in Toronto. The program conducts research and provides information about the safety of maternal exposure to drugs, chemicals, diseases, radiation, and environmental agents to the developing fetus or infant.
The women (mean age, 31-32 years) had experienced severe NVP or hyperemesis gravidarum (HG) in their previous pregnancy, and enrolled when they were planning a pregnancy or in early pregnancy at a point when they had not yet experienced any symptoms. Women in both groups took 2-9 tablets of the doxylamine-pyridoxine combination a day, and received counseling and follow-up calls.
In Canada, the combination is approved and marketed as Diclectin, by Duchesnay, and is the only drug labeled for treating NVP in Canada. There it is considered a first-line treatment and is taken at a standard dose of four tablets a day, said Ms. Maltepe, the coordinator of the NVP helpline at Motherisk.
Diclectin is a generic form of the drug Bendectin, which was marketed in the United States until 1983, when it was voluntarily withdrawn by its manufacturer, Merrell Dow Pharmaceuticals, after a series of lawsuits claiming it caused birth defects. For several years, Duchesnay has been working toward Food and Drug Administration clearance to market Diclectin in the United States, but it would be premature to predict when it would become available in the United States, a spokesman for the company said.
In the study, symptoms were evaluated using the PUQE (Pregnancy–Unique Quantification of Emesis and Nausea) score, a validated scoring system that takes into account the frequency of vomiting; the frequency of retching, gagging, or dry heaves; and the number of hours spent feeling nauseous over a 24-hour period. The score was developed by Dr. Gideon Koren, director of Motherisk and coauthor of this study, and his associates. A score of 7-12 is considered moderate; a score of 13 or higher is considered severe.
In the study, preemptive treatment was associated with 70% fewer cases of moderate to severe NVP during the first 3 weeks women experienced symptoms: 4 of the 26 (15%) evaluable women had a PUQE score of 11 or more during the first 3 weeks of NVP symptoms, compared with 9 of the 23 (39%) evaluable women in the control group, a significant difference. Of the 19 women in the preemptively treated group who had HG during the previous pregnancy, 6 (32%) experienced HG, compared with 6 of the 11 (55%) in the control group, also a significant difference. Some women who enrolled were not evaluable for reasons that included having a miscarriage.
NVP resolved before labor in significantly more of the women in the preemptively treated group than in the control group (almost 80% vs. about 50%), and there was a negative correlation between PUQE scores and a well-being score, indicating that treatment is associated with improved quality of life for these patients, Ms. Maltepe said.
In women who have experienced severe NVP or HG in a previous pregnancy, preemptive treatment with doxylamine-pyridoxine events can reduce the recurrence of NVP in subsequent pregnancies, and improve their quality of life during pregnancy, as well as reduce time off from work and the need for enteral and parenteral therapy, hospitalization, and other costs associated with severe NVP, she concluded.
The NVP helpline at Motherisk is the only dedicated NVP helpline in the world, according to Ms. Maltepe, who provides evidence-based recommendations on pharmaceutical and nonpharmaceutical approaches to treating NVP for pregnant women, their family members, and clinicians via the helpline (800-436-8477 in North America).
The study was funded by Duchesnay. Ms. Maltepe and her coauthors said they had no relevant conflict of interest to disclose.
The Motherisk site is available at www.motherisk.org/women/index.jsp.
BALTIMORE – Starting treatment with a combination of doxylamine and pyridoxine before the onset of nausea and vomiting significantly reduced the incidence of severe nausea and vomiting of pregnancy, compared with starting treatment at the first sign of symptoms, in a study of 59 women with a history of severe nausea and vomiting of pregnancy, Caroline Maltepe reported.
In the prospective, randomized, open-label study of women with a history of nausea and vomiting of pregnancy (NVP), 30 women began taking the delayed-release combination of 10 mg of doxylamine and 10 mg of pyridoxine (vitamin B6) as soon as they learned they were pregnant, before they started to experience nausea and vomiting (at a mean of 4 weeks’ gestation). Another 29 pregnant women started taking the combination when they started to experience symptoms, Ms. Maltepe said at the annual meeting of the Teratology Society.
Women in both groups started to experience NVP symptoms at a mean of about 5 to almost 6 weeks, said Ms. Maltepe, the lead author of the study, who is with the Motherisk Program at the Hospital for Sick Children in Toronto. The program conducts research and provides information about the safety of maternal exposure to drugs, chemicals, diseases, radiation, and environmental agents to the developing fetus or infant.
The women (mean age, 31-32 years) had experienced severe NVP or hyperemesis gravidarum (HG) in their previous pregnancy, and enrolled when they were planning a pregnancy or in early pregnancy at a point when they had not yet experienced any symptoms. Women in both groups took 2-9 tablets of the doxylamine-pyridoxine combination a day, and received counseling and follow-up calls.
In Canada, the combination is approved and marketed as Diclectin, by Duchesnay, and is the only drug labeled for treating NVP in Canada. There it is considered a first-line treatment and is taken at a standard dose of four tablets a day, said Ms. Maltepe, the coordinator of the NVP helpline at Motherisk.
Diclectin is a generic form of the drug Bendectin, which was marketed in the United States until 1983, when it was voluntarily withdrawn by its manufacturer, Merrell Dow Pharmaceuticals, after a series of lawsuits claiming it caused birth defects. For several years, Duchesnay has been working toward Food and Drug Administration clearance to market Diclectin in the United States, but it would be premature to predict when it would become available in the United States, a spokesman for the company said.
In the study, symptoms were evaluated using the PUQE (Pregnancy–Unique Quantification of Emesis and Nausea) score, a validated scoring system that takes into account the frequency of vomiting; the frequency of retching, gagging, or dry heaves; and the number of hours spent feeling nauseous over a 24-hour period. The score was developed by Dr. Gideon Koren, director of Motherisk and coauthor of this study, and his associates. A score of 7-12 is considered moderate; a score of 13 or higher is considered severe.
In the study, preemptive treatment was associated with 70% fewer cases of moderate to severe NVP during the first 3 weeks women experienced symptoms: 4 of the 26 (15%) evaluable women had a PUQE score of 11 or more during the first 3 weeks of NVP symptoms, compared with 9 of the 23 (39%) evaluable women in the control group, a significant difference. Of the 19 women in the preemptively treated group who had HG during the previous pregnancy, 6 (32%) experienced HG, compared with 6 of the 11 (55%) in the control group, also a significant difference. Some women who enrolled were not evaluable for reasons that included having a miscarriage.
NVP resolved before labor in significantly more of the women in the preemptively treated group than in the control group (almost 80% vs. about 50%), and there was a negative correlation between PUQE scores and a well-being score, indicating that treatment is associated with improved quality of life for these patients, Ms. Maltepe said.
In women who have experienced severe NVP or HG in a previous pregnancy, preemptive treatment with doxylamine-pyridoxine events can reduce the recurrence of NVP in subsequent pregnancies, and improve their quality of life during pregnancy, as well as reduce time off from work and the need for enteral and parenteral therapy, hospitalization, and other costs associated with severe NVP, she concluded.
The NVP helpline at Motherisk is the only dedicated NVP helpline in the world, according to Ms. Maltepe, who provides evidence-based recommendations on pharmaceutical and nonpharmaceutical approaches to treating NVP for pregnant women, their family members, and clinicians via the helpline (800-436-8477 in North America).
The study was funded by Duchesnay. Ms. Maltepe and her coauthors said they had no relevant conflict of interest to disclose.
The Motherisk site is available at www.motherisk.org/women/index.jsp.
AT THE ANNUAL MEETING OF THE TERATOLOGY SOCIETY