User login
USPSTF: Visual skin cancer screening lacks supporting evidence
The benefits and harms of visual screen cancer screening exams for asymptomatic adults can’t be adequately assessed with current evidence, according to a new recommendation from the U.S. Preventive Services Task Force.
“Evidence is inadequate to reliably conclude that early detection of skin cancer through visual skin examination by a clinician reduces morbidity or mortality,” according to the statement published online July 26 in JAMA (2016;316[4]:429-435. doi:10.1001/jama.2016.8465).
Approximately 76,400 adults in the United States will develop melanoma, and more than 10,000 will die from it, according to the USPSTF. However, more than 98% of skin cancer cases in the United States are basal and squamous cell carcinoma, which have much lower morbidity and mortality rates, noted the USPSTF researchers, led by Kirsten Bibbins-Domingo, MD, PhD, of the University of California, San Francisco.
The current statement updates the USPSTF’s 2009 recommendation, which also found insufficient evidence to assess the harms and benefits of visual skin cancer screening in asymptomatic adults with no history of premalignant or malignant skin lesions. However, the current recommendation eliminates a statement about patients’ skin self-exams.
According to the USPSTF, evidence is “adequate” that a clinician’s visual skin exam has “modest sensitivity and specificity for detecting melanoma,” but evidence is inconsistent to support the ability of a visual skin exam to detect nonmelanoma skin cancer.
The USPSTF commissioned an evidence review that included 11 studies previously reviewed and 2 additional studies conducted since 2009. The two new studies included one that evaluated skin cancer screening performed by dermatologists or plastic surgeons and one that evaluated skin cancer screening performed by primary care physicians. Sensitivity and specificity in the two studies ranged from 40% to 70% and from 86% to 98%, respectively.
“None of the studies could draw reliable conclusions as to whether screening performed by any of the clinical specialties differed in diagnostic accuracy,” the researchers noted. In addition, “no [randomized controlled trial] has directly evaluated the effectiveness of the clinical visual skin examination for reducing skin cancer morbidity and mortality,” they wrote.
The recommendation was accompanied by several editorials published online July 26 in JAMA journals.
In JAMA, Hensin Tsao, MD, PhD, of Massachusetts General Hospital, Boston, and Martin Weinstock, MD, PhD, of Brown University, Providence, R.I., noted that the USPSTF considered the possibility of including information from high-quality case-control studies in lieu of randomized controlled trials, which have been difficult to conduct in skin cancer screening. “The evidentiary standard needs to be further refined to be appropriate to the modest magnitude of potential harms of a properly performed skin cancer screening,” they wrote (JAMA. 2016;316:398-400). Dr. Tsao disclosed an honorarium from Lubax.
In JAMA Dermatology, Susan Swetter, MD, of the Veterans Affairs Palo Alto (Calif.) Health Care System; Alan C. Geller, MPH, of Harvard School of Public Health, Boston; and Allan C. Halpern, MD, of Memorial Sloan Kettering Cancer Center, New York, wrote about ways to promote broader uptake of skin cancer screening. “Alternative models should be explored to bundle skin screening with other preventive services (e.g., blood pressure measurements or flu shots) and to engage advanced practice providers (e.g., nurse practitioners and physician assistants) to promote screening among individuals with less access to dermatologists,” they wrote (JAMA Dermatol. 2016. doi: 10.1001/jamadermatol.2016.2606).
In JAMA Oncology, Vinayak K. Nahar, MD, of the University of Mississippi Medical Center, Jackson; Jonathan E. Mayer, MD, of Johns Hopkins University, Baltimore; and Jane M. Grant-Kels, MD, of the University of Connecticut, Farmington, addressed concerns over performing more biopsies. “The USPSTF also raises concern over the number needed to biopsy to detect 1 case of melanoma. In weighing these data, one must also consider that many of the nonmelanomas biopsied were likely severely atypical nevi that have their own risk of malignant transformation. Although difficult to quantify, there is some benefit to removing a severely atypical nevus, both for risk of transformation and for a patient’s peace of mind,” they wrote (JAMA Oncol. 2016. doi: 10.1001/jamaoncol.2016.2440).
In JAMA Internal Medicine, Eleni Linos, MD, of the University of California, San Francisco; Kenneth A. Katz, MD, of Kaiser Permanente, San Francisco; and Graham A. Colditz, MD, of Washington University, St. Louis, cautioned that the USPSTF recommendations shouldn’t be interpreted as minimizing the importance of skin cancer. “Instead, the report should motivate us to improve the evidence base for identifying groups of people in whom the benefits of screening might outweigh risks,” they wrote. “Meanwhile, we should also fully implement skin cancer primary prevention by eliminating indoor tanning exposure, especially among youths, and increasing the use of sun-protection strategies that work” (JAMA Intern. Med. 2016. doi: 10.1001/jamaintermed.2016.5008).
The recommendations are not an official position of the U.S. Department of Health and Human Services or the Agency for Healthcare Research and Quality.
“The American Academy of Dermatology is disappointed with this recommendation, as dermatologists know that skin cancer screenings can save lives, yet we acknowledge the need for additional research on the benefits and harms of skin cancer screening in the primary care setting,” Dr. Abel Torres, president of the American Academy of Dermatology, said in a statement responding to the USPSTF skin cancer screening recommendations.
“It is important for the public to understand that the USPSTF is not recommending against skin cancer screenings; it means the group did not find conclusive evidence to make a recommendation one way or another,” Dr. Torres said. “The public should know that this recommendation does not apply to individuals with suspicious skin lesions and those with an increased skin cancer risk, and it does not address the practice of skin self-exams.”
“The AAD encourages everyone to serve as their own health advocate by regularly conducting skin self-exams. Individuals who notice any unusual spots on their skin, including those that are changing, itching, or bleeding, should make an appointment with a board-certified dermatologist. In addition, individuals with an increased risk of melanoma – including men older than 50; people with more than 50 moles, or large or unusual moles; individuals with fair skin; and those with a history of skin cancer – should talk to a dermatologist about how often they should receive a skin exam from a doctor.”
Dr. Abel Torres is president of the American Academy of Dermatology. The comments are taken from his AAD statement on USPSTF Recommendation on Skin Cancer Screening issued on July 26, 2016.
“The American Academy of Dermatology is disappointed with this recommendation, as dermatologists know that skin cancer screenings can save lives, yet we acknowledge the need for additional research on the benefits and harms of skin cancer screening in the primary care setting,” Dr. Abel Torres, president of the American Academy of Dermatology, said in a statement responding to the USPSTF skin cancer screening recommendations.
“It is important for the public to understand that the USPSTF is not recommending against skin cancer screenings; it means the group did not find conclusive evidence to make a recommendation one way or another,” Dr. Torres said. “The public should know that this recommendation does not apply to individuals with suspicious skin lesions and those with an increased skin cancer risk, and it does not address the practice of skin self-exams.”
“The AAD encourages everyone to serve as their own health advocate by regularly conducting skin self-exams. Individuals who notice any unusual spots on their skin, including those that are changing, itching, or bleeding, should make an appointment with a board-certified dermatologist. In addition, individuals with an increased risk of melanoma – including men older than 50; people with more than 50 moles, or large or unusual moles; individuals with fair skin; and those with a history of skin cancer – should talk to a dermatologist about how often they should receive a skin exam from a doctor.”
Dr. Abel Torres is president of the American Academy of Dermatology. The comments are taken from his AAD statement on USPSTF Recommendation on Skin Cancer Screening issued on July 26, 2016.
“The American Academy of Dermatology is disappointed with this recommendation, as dermatologists know that skin cancer screenings can save lives, yet we acknowledge the need for additional research on the benefits and harms of skin cancer screening in the primary care setting,” Dr. Abel Torres, president of the American Academy of Dermatology, said in a statement responding to the USPSTF skin cancer screening recommendations.
“It is important for the public to understand that the USPSTF is not recommending against skin cancer screenings; it means the group did not find conclusive evidence to make a recommendation one way or another,” Dr. Torres said. “The public should know that this recommendation does not apply to individuals with suspicious skin lesions and those with an increased skin cancer risk, and it does not address the practice of skin self-exams.”
“The AAD encourages everyone to serve as their own health advocate by regularly conducting skin self-exams. Individuals who notice any unusual spots on their skin, including those that are changing, itching, or bleeding, should make an appointment with a board-certified dermatologist. In addition, individuals with an increased risk of melanoma – including men older than 50; people with more than 50 moles, or large or unusual moles; individuals with fair skin; and those with a history of skin cancer – should talk to a dermatologist about how often they should receive a skin exam from a doctor.”
Dr. Abel Torres is president of the American Academy of Dermatology. The comments are taken from his AAD statement on USPSTF Recommendation on Skin Cancer Screening issued on July 26, 2016.
The benefits and harms of visual screen cancer screening exams for asymptomatic adults can’t be adequately assessed with current evidence, according to a new recommendation from the U.S. Preventive Services Task Force.
“Evidence is inadequate to reliably conclude that early detection of skin cancer through visual skin examination by a clinician reduces morbidity or mortality,” according to the statement published online July 26 in JAMA (2016;316[4]:429-435. doi:10.1001/jama.2016.8465).
Approximately 76,400 adults in the United States will develop melanoma, and more than 10,000 will die from it, according to the USPSTF. However, more than 98% of skin cancer cases in the United States are basal and squamous cell carcinoma, which have much lower morbidity and mortality rates, noted the USPSTF researchers, led by Kirsten Bibbins-Domingo, MD, PhD, of the University of California, San Francisco.
The current statement updates the USPSTF’s 2009 recommendation, which also found insufficient evidence to assess the harms and benefits of visual skin cancer screening in asymptomatic adults with no history of premalignant or malignant skin lesions. However, the current recommendation eliminates a statement about patients’ skin self-exams.
According to the USPSTF, evidence is “adequate” that a clinician’s visual skin exam has “modest sensitivity and specificity for detecting melanoma,” but evidence is inconsistent to support the ability of a visual skin exam to detect nonmelanoma skin cancer.
The USPSTF commissioned an evidence review that included 11 studies previously reviewed and 2 additional studies conducted since 2009. The two new studies included one that evaluated skin cancer screening performed by dermatologists or plastic surgeons and one that evaluated skin cancer screening performed by primary care physicians. Sensitivity and specificity in the two studies ranged from 40% to 70% and from 86% to 98%, respectively.
“None of the studies could draw reliable conclusions as to whether screening performed by any of the clinical specialties differed in diagnostic accuracy,” the researchers noted. In addition, “no [randomized controlled trial] has directly evaluated the effectiveness of the clinical visual skin examination for reducing skin cancer morbidity and mortality,” they wrote.
The recommendation was accompanied by several editorials published online July 26 in JAMA journals.
In JAMA, Hensin Tsao, MD, PhD, of Massachusetts General Hospital, Boston, and Martin Weinstock, MD, PhD, of Brown University, Providence, R.I., noted that the USPSTF considered the possibility of including information from high-quality case-control studies in lieu of randomized controlled trials, which have been difficult to conduct in skin cancer screening. “The evidentiary standard needs to be further refined to be appropriate to the modest magnitude of potential harms of a properly performed skin cancer screening,” they wrote (JAMA. 2016;316:398-400). Dr. Tsao disclosed an honorarium from Lubax.
In JAMA Dermatology, Susan Swetter, MD, of the Veterans Affairs Palo Alto (Calif.) Health Care System; Alan C. Geller, MPH, of Harvard School of Public Health, Boston; and Allan C. Halpern, MD, of Memorial Sloan Kettering Cancer Center, New York, wrote about ways to promote broader uptake of skin cancer screening. “Alternative models should be explored to bundle skin screening with other preventive services (e.g., blood pressure measurements or flu shots) and to engage advanced practice providers (e.g., nurse practitioners and physician assistants) to promote screening among individuals with less access to dermatologists,” they wrote (JAMA Dermatol. 2016. doi: 10.1001/jamadermatol.2016.2606).
In JAMA Oncology, Vinayak K. Nahar, MD, of the University of Mississippi Medical Center, Jackson; Jonathan E. Mayer, MD, of Johns Hopkins University, Baltimore; and Jane M. Grant-Kels, MD, of the University of Connecticut, Farmington, addressed concerns over performing more biopsies. “The USPSTF also raises concern over the number needed to biopsy to detect 1 case of melanoma. In weighing these data, one must also consider that many of the nonmelanomas biopsied were likely severely atypical nevi that have their own risk of malignant transformation. Although difficult to quantify, there is some benefit to removing a severely atypical nevus, both for risk of transformation and for a patient’s peace of mind,” they wrote (JAMA Oncol. 2016. doi: 10.1001/jamaoncol.2016.2440).
In JAMA Internal Medicine, Eleni Linos, MD, of the University of California, San Francisco; Kenneth A. Katz, MD, of Kaiser Permanente, San Francisco; and Graham A. Colditz, MD, of Washington University, St. Louis, cautioned that the USPSTF recommendations shouldn’t be interpreted as minimizing the importance of skin cancer. “Instead, the report should motivate us to improve the evidence base for identifying groups of people in whom the benefits of screening might outweigh risks,” they wrote. “Meanwhile, we should also fully implement skin cancer primary prevention by eliminating indoor tanning exposure, especially among youths, and increasing the use of sun-protection strategies that work” (JAMA Intern. Med. 2016. doi: 10.1001/jamaintermed.2016.5008).
The recommendations are not an official position of the U.S. Department of Health and Human Services or the Agency for Healthcare Research and Quality.
The benefits and harms of visual screen cancer screening exams for asymptomatic adults can’t be adequately assessed with current evidence, according to a new recommendation from the U.S. Preventive Services Task Force.
“Evidence is inadequate to reliably conclude that early detection of skin cancer through visual skin examination by a clinician reduces morbidity or mortality,” according to the statement published online July 26 in JAMA (2016;316[4]:429-435. doi:10.1001/jama.2016.8465).
Approximately 76,400 adults in the United States will develop melanoma, and more than 10,000 will die from it, according to the USPSTF. However, more than 98% of skin cancer cases in the United States are basal and squamous cell carcinoma, which have much lower morbidity and mortality rates, noted the USPSTF researchers, led by Kirsten Bibbins-Domingo, MD, PhD, of the University of California, San Francisco.
The current statement updates the USPSTF’s 2009 recommendation, which also found insufficient evidence to assess the harms and benefits of visual skin cancer screening in asymptomatic adults with no history of premalignant or malignant skin lesions. However, the current recommendation eliminates a statement about patients’ skin self-exams.
According to the USPSTF, evidence is “adequate” that a clinician’s visual skin exam has “modest sensitivity and specificity for detecting melanoma,” but evidence is inconsistent to support the ability of a visual skin exam to detect nonmelanoma skin cancer.
The USPSTF commissioned an evidence review that included 11 studies previously reviewed and 2 additional studies conducted since 2009. The two new studies included one that evaluated skin cancer screening performed by dermatologists or plastic surgeons and one that evaluated skin cancer screening performed by primary care physicians. Sensitivity and specificity in the two studies ranged from 40% to 70% and from 86% to 98%, respectively.
“None of the studies could draw reliable conclusions as to whether screening performed by any of the clinical specialties differed in diagnostic accuracy,” the researchers noted. In addition, “no [randomized controlled trial] has directly evaluated the effectiveness of the clinical visual skin examination for reducing skin cancer morbidity and mortality,” they wrote.
The recommendation was accompanied by several editorials published online July 26 in JAMA journals.
In JAMA, Hensin Tsao, MD, PhD, of Massachusetts General Hospital, Boston, and Martin Weinstock, MD, PhD, of Brown University, Providence, R.I., noted that the USPSTF considered the possibility of including information from high-quality case-control studies in lieu of randomized controlled trials, which have been difficult to conduct in skin cancer screening. “The evidentiary standard needs to be further refined to be appropriate to the modest magnitude of potential harms of a properly performed skin cancer screening,” they wrote (JAMA. 2016;316:398-400). Dr. Tsao disclosed an honorarium from Lubax.
In JAMA Dermatology, Susan Swetter, MD, of the Veterans Affairs Palo Alto (Calif.) Health Care System; Alan C. Geller, MPH, of Harvard School of Public Health, Boston; and Allan C. Halpern, MD, of Memorial Sloan Kettering Cancer Center, New York, wrote about ways to promote broader uptake of skin cancer screening. “Alternative models should be explored to bundle skin screening with other preventive services (e.g., blood pressure measurements or flu shots) and to engage advanced practice providers (e.g., nurse practitioners and physician assistants) to promote screening among individuals with less access to dermatologists,” they wrote (JAMA Dermatol. 2016. doi: 10.1001/jamadermatol.2016.2606).
In JAMA Oncology, Vinayak K. Nahar, MD, of the University of Mississippi Medical Center, Jackson; Jonathan E. Mayer, MD, of Johns Hopkins University, Baltimore; and Jane M. Grant-Kels, MD, of the University of Connecticut, Farmington, addressed concerns over performing more biopsies. “The USPSTF also raises concern over the number needed to biopsy to detect 1 case of melanoma. In weighing these data, one must also consider that many of the nonmelanomas biopsied were likely severely atypical nevi that have their own risk of malignant transformation. Although difficult to quantify, there is some benefit to removing a severely atypical nevus, both for risk of transformation and for a patient’s peace of mind,” they wrote (JAMA Oncol. 2016. doi: 10.1001/jamaoncol.2016.2440).
In JAMA Internal Medicine, Eleni Linos, MD, of the University of California, San Francisco; Kenneth A. Katz, MD, of Kaiser Permanente, San Francisco; and Graham A. Colditz, MD, of Washington University, St. Louis, cautioned that the USPSTF recommendations shouldn’t be interpreted as minimizing the importance of skin cancer. “Instead, the report should motivate us to improve the evidence base for identifying groups of people in whom the benefits of screening might outweigh risks,” they wrote. “Meanwhile, we should also fully implement skin cancer primary prevention by eliminating indoor tanning exposure, especially among youths, and increasing the use of sun-protection strategies that work” (JAMA Intern. Med. 2016. doi: 10.1001/jamaintermed.2016.5008).
The recommendations are not an official position of the U.S. Department of Health and Human Services or the Agency for Healthcare Research and Quality.
FROM JAMA
ACOG: Offer immediate postpartum LARC as option
Use of long-acting reversible contraception immediately post partum can help reduce the risk of unintended and short-interval pregnancy, according to a new policy statement from the American College of Obstetricians and Gynecologists.
“Immediate postpartum LARC should be offered as an effective option for postpartum contraception; there are few contraindications to postpartum IUDs and implants,” according to the statement issued by ACOG’s Committee on Obstetric Practice (Obstet Gynecol. 2016;128:e32-37).
Unintended pregnancies account for approximately 45% of pregnancies in the United States overall, and at least 70% of pregnancies in the first year post partum, the committee noted.
The statement recommends prenatal counseling about the risks and benefits of LARCs, along with alternatives, to help patients make informed decisions. The committee emphasized that health care providers “should counsel women about the convenience and effectiveness of immediate postpartum LARC, as well as the benefits of reducing unintended pregnancy and lengthening pregnancy intervals.”
However, immediate postpartum use of IUDs is contraindicated in women with intrauterine infection at delivery, postpartum hemorrhage, and puerperal sepsis.
The committee called for systems to provide LARC at the comprehensive postpartum visit if necessary, and recommended stocking LARC devices in labor and delivery units for immediate postpartum placement. In addition, coding and reimbursement strategies are needed to support immediate postpartum LARC, according to the statement.
The policy statement was endorsed by the American College of Nurse-Midwives and the Society for Maternal-Fetal Medicine.
Use of long-acting reversible contraception immediately post partum can help reduce the risk of unintended and short-interval pregnancy, according to a new policy statement from the American College of Obstetricians and Gynecologists.
“Immediate postpartum LARC should be offered as an effective option for postpartum contraception; there are few contraindications to postpartum IUDs and implants,” according to the statement issued by ACOG’s Committee on Obstetric Practice (Obstet Gynecol. 2016;128:e32-37).
Unintended pregnancies account for approximately 45% of pregnancies in the United States overall, and at least 70% of pregnancies in the first year post partum, the committee noted.
The statement recommends prenatal counseling about the risks and benefits of LARCs, along with alternatives, to help patients make informed decisions. The committee emphasized that health care providers “should counsel women about the convenience and effectiveness of immediate postpartum LARC, as well as the benefits of reducing unintended pregnancy and lengthening pregnancy intervals.”
However, immediate postpartum use of IUDs is contraindicated in women with intrauterine infection at delivery, postpartum hemorrhage, and puerperal sepsis.
The committee called for systems to provide LARC at the comprehensive postpartum visit if necessary, and recommended stocking LARC devices in labor and delivery units for immediate postpartum placement. In addition, coding and reimbursement strategies are needed to support immediate postpartum LARC, according to the statement.
The policy statement was endorsed by the American College of Nurse-Midwives and the Society for Maternal-Fetal Medicine.
Use of long-acting reversible contraception immediately post partum can help reduce the risk of unintended and short-interval pregnancy, according to a new policy statement from the American College of Obstetricians and Gynecologists.
“Immediate postpartum LARC should be offered as an effective option for postpartum contraception; there are few contraindications to postpartum IUDs and implants,” according to the statement issued by ACOG’s Committee on Obstetric Practice (Obstet Gynecol. 2016;128:e32-37).
Unintended pregnancies account for approximately 45% of pregnancies in the United States overall, and at least 70% of pregnancies in the first year post partum, the committee noted.
The statement recommends prenatal counseling about the risks and benefits of LARCs, along with alternatives, to help patients make informed decisions. The committee emphasized that health care providers “should counsel women about the convenience and effectiveness of immediate postpartum LARC, as well as the benefits of reducing unintended pregnancy and lengthening pregnancy intervals.”
However, immediate postpartum use of IUDs is contraindicated in women with intrauterine infection at delivery, postpartum hemorrhage, and puerperal sepsis.
The committee called for systems to provide LARC at the comprehensive postpartum visit if necessary, and recommended stocking LARC devices in labor and delivery units for immediate postpartum placement. In addition, coding and reimbursement strategies are needed to support immediate postpartum LARC, according to the statement.
The policy statement was endorsed by the American College of Nurse-Midwives and the Society for Maternal-Fetal Medicine.
FROM OBSTETRICS & GYNECOLOGY
ACOG supports evidence-based decisions on planned home birth
Women who are interested in a planned home birth are entitled to make medically informed decisions about where to deliver their babies, according to an updated policy statement from the American College of Obstetricians and Gynecologists.
But women should know the risks and benefits of home birth based on the latest evidence. Although planned home birth is associated with fewer maternal interventions than planned hospital birth, there is also a twofold increased risk of perinatal death (1-2 per 1,000 births) and a threefold increased risk of neonatal seizures or other neurologic problems (0.4-0.6 per 1,000 births), according to the statement from ACOG’s Committee on Obstetric Practice, which updates its 2011 policy (Obstet Gynecol. 2016;128:e26-31).
ACOG maintains that hospitals and accredited birth centers are the safest settings for birth, and that unplanned home births are associated with increased rates of perinatal and neonatal mortality. “The relative risk versus benefit of a planned home birth, however, remains the subject of debate,” the committee wrote.
Women considering home birth should attend to several factors to help promote a safe delivery, including being a suitable candidate for home birth; identifying a qualified nurse-midwife, a qualified midwife, or a physician practicing obstetrics who is available for consultation; and preparing for safe, timely transport to a hospital if necessary.
An arrangement with a hospital for transfer is a requirement for women considering a planned home birth, according to the statement. However, the statement advised the hospital and health care team to refrain from judgment should their help be needed.
“When antepartum, intrapartum, or postpartum transfer of a woman from home to a hospital occurs, the receiving health care provider should maintain a nonjudgmental demeanor with regard to the woman and those individuals accompanying her to the hospital,” the committee wrote.
Although the current statement supports a woman’s right to make a medically informed decision, it emphasizes that any of several factors including fetal malpresentation, multiple gestation, and prior cesarean delivery are “an absolute contraindication to planned home birth.”
Women who are interested in a planned home birth are entitled to make medically informed decisions about where to deliver their babies, according to an updated policy statement from the American College of Obstetricians and Gynecologists.
But women should know the risks and benefits of home birth based on the latest evidence. Although planned home birth is associated with fewer maternal interventions than planned hospital birth, there is also a twofold increased risk of perinatal death (1-2 per 1,000 births) and a threefold increased risk of neonatal seizures or other neurologic problems (0.4-0.6 per 1,000 births), according to the statement from ACOG’s Committee on Obstetric Practice, which updates its 2011 policy (Obstet Gynecol. 2016;128:e26-31).
ACOG maintains that hospitals and accredited birth centers are the safest settings for birth, and that unplanned home births are associated with increased rates of perinatal and neonatal mortality. “The relative risk versus benefit of a planned home birth, however, remains the subject of debate,” the committee wrote.
Women considering home birth should attend to several factors to help promote a safe delivery, including being a suitable candidate for home birth; identifying a qualified nurse-midwife, a qualified midwife, or a physician practicing obstetrics who is available for consultation; and preparing for safe, timely transport to a hospital if necessary.
An arrangement with a hospital for transfer is a requirement for women considering a planned home birth, according to the statement. However, the statement advised the hospital and health care team to refrain from judgment should their help be needed.
“When antepartum, intrapartum, or postpartum transfer of a woman from home to a hospital occurs, the receiving health care provider should maintain a nonjudgmental demeanor with regard to the woman and those individuals accompanying her to the hospital,” the committee wrote.
Although the current statement supports a woman’s right to make a medically informed decision, it emphasizes that any of several factors including fetal malpresentation, multiple gestation, and prior cesarean delivery are “an absolute contraindication to planned home birth.”
Women who are interested in a planned home birth are entitled to make medically informed decisions about where to deliver their babies, according to an updated policy statement from the American College of Obstetricians and Gynecologists.
But women should know the risks and benefits of home birth based on the latest evidence. Although planned home birth is associated with fewer maternal interventions than planned hospital birth, there is also a twofold increased risk of perinatal death (1-2 per 1,000 births) and a threefold increased risk of neonatal seizures or other neurologic problems (0.4-0.6 per 1,000 births), according to the statement from ACOG’s Committee on Obstetric Practice, which updates its 2011 policy (Obstet Gynecol. 2016;128:e26-31).
ACOG maintains that hospitals and accredited birth centers are the safest settings for birth, and that unplanned home births are associated with increased rates of perinatal and neonatal mortality. “The relative risk versus benefit of a planned home birth, however, remains the subject of debate,” the committee wrote.
Women considering home birth should attend to several factors to help promote a safe delivery, including being a suitable candidate for home birth; identifying a qualified nurse-midwife, a qualified midwife, or a physician practicing obstetrics who is available for consultation; and preparing for safe, timely transport to a hospital if necessary.
An arrangement with a hospital for transfer is a requirement for women considering a planned home birth, according to the statement. However, the statement advised the hospital and health care team to refrain from judgment should their help be needed.
“When antepartum, intrapartum, or postpartum transfer of a woman from home to a hospital occurs, the receiving health care provider should maintain a nonjudgmental demeanor with regard to the woman and those individuals accompanying her to the hospital,” the committee wrote.
Although the current statement supports a woman’s right to make a medically informed decision, it emphasizes that any of several factors including fetal malpresentation, multiple gestation, and prior cesarean delivery are “an absolute contraindication to planned home birth.”
FROM OBSTETRICS & GYNECOLOGY
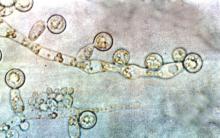
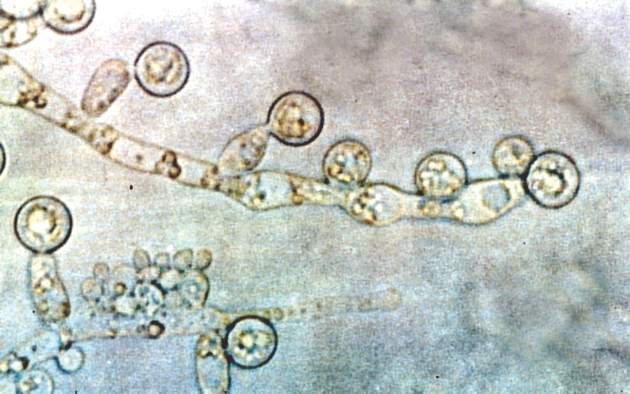
Candida colonization raises risk of Acinetobacter-based VAP
Acinetobacter baumanii was the most common cause of ventilator-associated pneumonia in ICU patients, and the risk of A. baumannii infection was significantly higher when airways were colonized with Candida species, based on data from 618 adults.
A. baumannii is a frequent cause of ventilator-associated pneumonia (VAP) in ICU patients, but its potential interactions with Candida species have not been well studied, wrote Dr. Xiaojiang Tan of Southern Medical University, Guangzhou, China, and colleagues (Med Mycol. 2016 Aug 1;54[6]:557-66. doi: 10.1093/mmy/myw009). The researchers reviewed data from 264 ICU patients on mechanical ventilation who had Candida species airway colonization and 354 who did not.
Overall, Candida was an independent risk factor for A. baumannii VAP; patients with Candida were significantly more likely than those without Candida to develop A. baumannii (23% vs. 15%). Other independent risk factors for A. baumannii VAP included the use of a central venous catheter and the use of mechanical ventilation for at least 7 days. Among patients on mechanical ventilation for at least 48 hours, Candida airway colonization occurred in 43%, and A. baumannii VAP occurred in 18%.
Candida albicans showed an especially strong association with A. baumannii VAP; it was identified in 38% of cases, compared with 21% caused by non-albicans species.
No significant differences in hospital stay or in-hospital mortality were noted between Candida-colonized and noncolonized patients, and antifungal treatment had no apparent impact on the development of A. baumannii VAP, but antifungals were associated with higher in-hospital mortality (53% vs. 39%, P = .037).
The results were limited by the retrospective nature of the study and the use of data from a single center; thus, the relationship between Candida and A. baumannii VAP may not be generalizable, the researchers noted. However, “the strong independent association between the two suggests that Candida spp. growth from the lower respiratory tract in intubated patients could be an important indicator of the risk for VAP, and even that C. albicans airway colonization may play a role in subsequent development of A. baumannii VAP,” they wrote.
The researchers reported having no relevant financial conflicts.
Acinetobacter baumanii was the most common cause of ventilator-associated pneumonia in ICU patients, and the risk of A. baumannii infection was significantly higher when airways were colonized with Candida species, based on data from 618 adults.
A. baumannii is a frequent cause of ventilator-associated pneumonia (VAP) in ICU patients, but its potential interactions with Candida species have not been well studied, wrote Dr. Xiaojiang Tan of Southern Medical University, Guangzhou, China, and colleagues (Med Mycol. 2016 Aug 1;54[6]:557-66. doi: 10.1093/mmy/myw009). The researchers reviewed data from 264 ICU patients on mechanical ventilation who had Candida species airway colonization and 354 who did not.
Overall, Candida was an independent risk factor for A. baumannii VAP; patients with Candida were significantly more likely than those without Candida to develop A. baumannii (23% vs. 15%). Other independent risk factors for A. baumannii VAP included the use of a central venous catheter and the use of mechanical ventilation for at least 7 days. Among patients on mechanical ventilation for at least 48 hours, Candida airway colonization occurred in 43%, and A. baumannii VAP occurred in 18%.
Candida albicans showed an especially strong association with A. baumannii VAP; it was identified in 38% of cases, compared with 21% caused by non-albicans species.
No significant differences in hospital stay or in-hospital mortality were noted between Candida-colonized and noncolonized patients, and antifungal treatment had no apparent impact on the development of A. baumannii VAP, but antifungals were associated with higher in-hospital mortality (53% vs. 39%, P = .037).
The results were limited by the retrospective nature of the study and the use of data from a single center; thus, the relationship between Candida and A. baumannii VAP may not be generalizable, the researchers noted. However, “the strong independent association between the two suggests that Candida spp. growth from the lower respiratory tract in intubated patients could be an important indicator of the risk for VAP, and even that C. albicans airway colonization may play a role in subsequent development of A. baumannii VAP,” they wrote.
The researchers reported having no relevant financial conflicts.
Acinetobacter baumanii was the most common cause of ventilator-associated pneumonia in ICU patients, and the risk of A. baumannii infection was significantly higher when airways were colonized with Candida species, based on data from 618 adults.
A. baumannii is a frequent cause of ventilator-associated pneumonia (VAP) in ICU patients, but its potential interactions with Candida species have not been well studied, wrote Dr. Xiaojiang Tan of Southern Medical University, Guangzhou, China, and colleagues (Med Mycol. 2016 Aug 1;54[6]:557-66. doi: 10.1093/mmy/myw009). The researchers reviewed data from 264 ICU patients on mechanical ventilation who had Candida species airway colonization and 354 who did not.
Overall, Candida was an independent risk factor for A. baumannii VAP; patients with Candida were significantly more likely than those without Candida to develop A. baumannii (23% vs. 15%). Other independent risk factors for A. baumannii VAP included the use of a central venous catheter and the use of mechanical ventilation for at least 7 days. Among patients on mechanical ventilation for at least 48 hours, Candida airway colonization occurred in 43%, and A. baumannii VAP occurred in 18%.
Candida albicans showed an especially strong association with A. baumannii VAP; it was identified in 38% of cases, compared with 21% caused by non-albicans species.
No significant differences in hospital stay or in-hospital mortality were noted between Candida-colonized and noncolonized patients, and antifungal treatment had no apparent impact on the development of A. baumannii VAP, but antifungals were associated with higher in-hospital mortality (53% vs. 39%, P = .037).
The results were limited by the retrospective nature of the study and the use of data from a single center; thus, the relationship between Candida and A. baumannii VAP may not be generalizable, the researchers noted. However, “the strong independent association between the two suggests that Candida spp. growth from the lower respiratory tract in intubated patients could be an important indicator of the risk for VAP, and even that C. albicans airway colonization may play a role in subsequent development of A. baumannii VAP,” they wrote.
The researchers reported having no relevant financial conflicts.
FROM MEDICAL MYCOLOGY
Key clinical point: Candida species colonization was an independent risk factor for ventilator-associated pneumonia caused by Acinetobacter baumannii among ICU patients.
Major finding: Hospitalized patients with Candida species airway colonization were significantly more likely to develop A. baumannii infection than were those without Candida (23% vs. 15%).
Data source: A retrospective case-control study of 618 ICU patients.
Disclosures: The researchers reported having no relevant financial conflicts.
Start time for estrogen conveys no cognitive impact
The cognitive effects of estrogen were no different in women who started taking the hormone within 6 years of menopause, compared with those who began it 10 or more years after menopause, based on data from the double-blind randomized ELITE-Cog trial.
“Some hormone effects on cognition are postulated to vary by age or by timing in relation to the menopause,” wrote Victor W. Henderson, M.D., of Stanford (Calif.) University and his colleagues (Neurology. 2016 Jul 20. doi: 10.1212/WNL.0000000000002980).
To test whether the timing of estrogen therapy affected the primary cognitive endpoint of verbal episodic memory, the researchers conducted the ELITE (Early vs. Late Intervention Trial With Estradiol) trial in which they randomized 567 healthy women within 6 years of menopause or 10 or more years post menopause to 1 mg/day of oral 17-beta-estradiol or a placebo. Women with a uterus also received either 45 mg progesterone as a 4% vaginal gel or a matched placebo gel.
The main ELITE trial tested the effect of the timing of estradiol, compared with placebo, on the progression of subclinical atherosclerosis, whereas the ELITE-Cog trial assessed the effects of the timing of estradiol on a composite test of verbal episodic memory.
Overall, composite scores of verbal memory were not significantly different in women randomized to estrogen vs. placebo based on a mean standardized difference of –0.06 (95% confidence interval, –0.22 to 0.09) after an average of 57 months of treatment. The mean standardized difference was similar in the early and late treatment groups. In addition, the mean standardized differences in measures of executive functions (–0.04; 95% CI, –0.21 to 0.14) and global cognition (–0.025; 95% CI, –0.18 to 0.13) were not significantly different between women who started estrogen within 6 years of menopause and those who started estrogen 10 years or more after menopause. Safety profiles were similar between the groups.
“Results of this randomized, double-blind placebo-controlled trial fail to confirm the timing hypothesis for cognitive outcomes in healthy postmenopausal women,” the researchers wrote.
The results don’t generalize to other subgroups including women of reproductive age, those in transition to menopause, or those with premature menopause caused by surgery or chemotherapy, and the study “was not designed to assess short-term cognitive effects of estradiol or effects on risks of mild cognitive impairment or Alzheimer disease,” the researchers noted. However, the findings should be reassuring to healthy, younger postmenopausal women considering estrogen therapy, they said.
The study was supported by a grant from the National Institutes of Health, and the study drugs and placebo were supplied by Teva, Watson, and Abbott Laboratories. Dr. Henderson disclosed research support from the NIH, as well as travel expenses from the NIH, American Academy of Neurology, and International Menopause Society.
The cognitive effects of estrogen were no different in women who started taking the hormone within 6 years of menopause, compared with those who began it 10 or more years after menopause, based on data from the double-blind randomized ELITE-Cog trial.
“Some hormone effects on cognition are postulated to vary by age or by timing in relation to the menopause,” wrote Victor W. Henderson, M.D., of Stanford (Calif.) University and his colleagues (Neurology. 2016 Jul 20. doi: 10.1212/WNL.0000000000002980).
To test whether the timing of estrogen therapy affected the primary cognitive endpoint of verbal episodic memory, the researchers conducted the ELITE (Early vs. Late Intervention Trial With Estradiol) trial in which they randomized 567 healthy women within 6 years of menopause or 10 or more years post menopause to 1 mg/day of oral 17-beta-estradiol or a placebo. Women with a uterus also received either 45 mg progesterone as a 4% vaginal gel or a matched placebo gel.
The main ELITE trial tested the effect of the timing of estradiol, compared with placebo, on the progression of subclinical atherosclerosis, whereas the ELITE-Cog trial assessed the effects of the timing of estradiol on a composite test of verbal episodic memory.
Overall, composite scores of verbal memory were not significantly different in women randomized to estrogen vs. placebo based on a mean standardized difference of –0.06 (95% confidence interval, –0.22 to 0.09) after an average of 57 months of treatment. The mean standardized difference was similar in the early and late treatment groups. In addition, the mean standardized differences in measures of executive functions (–0.04; 95% CI, –0.21 to 0.14) and global cognition (–0.025; 95% CI, –0.18 to 0.13) were not significantly different between women who started estrogen within 6 years of menopause and those who started estrogen 10 years or more after menopause. Safety profiles were similar between the groups.
“Results of this randomized, double-blind placebo-controlled trial fail to confirm the timing hypothesis for cognitive outcomes in healthy postmenopausal women,” the researchers wrote.
The results don’t generalize to other subgroups including women of reproductive age, those in transition to menopause, or those with premature menopause caused by surgery or chemotherapy, and the study “was not designed to assess short-term cognitive effects of estradiol or effects on risks of mild cognitive impairment or Alzheimer disease,” the researchers noted. However, the findings should be reassuring to healthy, younger postmenopausal women considering estrogen therapy, they said.
The study was supported by a grant from the National Institutes of Health, and the study drugs and placebo were supplied by Teva, Watson, and Abbott Laboratories. Dr. Henderson disclosed research support from the NIH, as well as travel expenses from the NIH, American Academy of Neurology, and International Menopause Society.
The cognitive effects of estrogen were no different in women who started taking the hormone within 6 years of menopause, compared with those who began it 10 or more years after menopause, based on data from the double-blind randomized ELITE-Cog trial.
“Some hormone effects on cognition are postulated to vary by age or by timing in relation to the menopause,” wrote Victor W. Henderson, M.D., of Stanford (Calif.) University and his colleagues (Neurology. 2016 Jul 20. doi: 10.1212/WNL.0000000000002980).
To test whether the timing of estrogen therapy affected the primary cognitive endpoint of verbal episodic memory, the researchers conducted the ELITE (Early vs. Late Intervention Trial With Estradiol) trial in which they randomized 567 healthy women within 6 years of menopause or 10 or more years post menopause to 1 mg/day of oral 17-beta-estradiol or a placebo. Women with a uterus also received either 45 mg progesterone as a 4% vaginal gel or a matched placebo gel.
The main ELITE trial tested the effect of the timing of estradiol, compared with placebo, on the progression of subclinical atherosclerosis, whereas the ELITE-Cog trial assessed the effects of the timing of estradiol on a composite test of verbal episodic memory.
Overall, composite scores of verbal memory were not significantly different in women randomized to estrogen vs. placebo based on a mean standardized difference of –0.06 (95% confidence interval, –0.22 to 0.09) after an average of 57 months of treatment. The mean standardized difference was similar in the early and late treatment groups. In addition, the mean standardized differences in measures of executive functions (–0.04; 95% CI, –0.21 to 0.14) and global cognition (–0.025; 95% CI, –0.18 to 0.13) were not significantly different between women who started estrogen within 6 years of menopause and those who started estrogen 10 years or more after menopause. Safety profiles were similar between the groups.
“Results of this randomized, double-blind placebo-controlled trial fail to confirm the timing hypothesis for cognitive outcomes in healthy postmenopausal women,” the researchers wrote.
The results don’t generalize to other subgroups including women of reproductive age, those in transition to menopause, or those with premature menopause caused by surgery or chemotherapy, and the study “was not designed to assess short-term cognitive effects of estradiol or effects on risks of mild cognitive impairment or Alzheimer disease,” the researchers noted. However, the findings should be reassuring to healthy, younger postmenopausal women considering estrogen therapy, they said.
The study was supported by a grant from the National Institutes of Health, and the study drugs and placebo were supplied by Teva, Watson, and Abbott Laboratories. Dr. Henderson disclosed research support from the NIH, as well as travel expenses from the NIH, American Academy of Neurology, and International Menopause Society.
FROM NEUROLOGY
Key clinical point: Estradiol had no significant impact on cognitive function when taken within 6 years of menopause or more than 10 years after menopause.
Major finding: Composite scores of verbal memory were not significantly different in women randomized to estrogen vs. placebo, based on a mean standardized difference of –0.06 (95% confidence interval, –0.22 to 0.09) after an average of 57 months of treatment. The mean standardized difference was similar in the early and late treatment groups.
Data source: A double-blind, randomized ELITE-Cog trial including 567 healthy women within 6 years of menopause or 10 or more years after menopause.
Disclosures: The study was supported by a grant from the National Institutes of Health, and the study drugs and placebo were supplied by Teva, Watson, and Abbott Laboratories. Dr. Henderson disclosed research support from the NIH, as well as travel expenses from the NIH, American Academy of Neurology, and International Menopause Society.
Task force affirms value of colorectal cancer screening
Colorectal cancer screening by a variety of methods is worthwhile and recommended for all adults aged 50-75 years, according to the latest recommendations from the U.S. Preventive Services Task Force. The USPSTF statement and summary of evidence were published in JAMA on June 15.
In addition, the USPSTF recommended selective screening for older adults aged 76-85 years, depending on their health status and screening history.
A team of researchers led by Dr. Jennifer S. Lin of Kaiser Permanente in Portland, Ore., reviewed studies on colorectal cancer screening published between Jan.1, 2008, and Dec. 31, 2014, with surveillance continuing through Feb. 23, 2016 (JAMA. 2016 Jun;315:2576-94 [doi: 10.1001/jama.2016.3332]). The USPSTF’s last recommendations on colorectal cancer screening were released in 2008.
“Although CRC screening has a large body of supporting evidence, additional research is still needed to weigh the relative benefits and harms of each test within a program of screening” for average-risk adults, the researchers noted.
The final recommendation statement includes three screening options that carry over from the 2008 guidelines: colonoscopy, sigmoidoscopy, and fecal immunochemical testing (FIT) with occult blood.
Other options now recommended include computed tomographic colonography (CTC), fecal immunochemical tests with DNA (FIT-DNA), guaiac-based fecal occult blood testing (gFOBT), and sigmoidoscopy plus FIT.
Some highlights from the analysis: Four randomized trials including 458,002 patients showed that one-time or two-time screening with flexible sigmoidoscopy was associated with decreased mortality from colorectal cancer, compared with no screening, for an incidence rate ratio of 0.73, the researchers wrote.
In addition, the researchers found that CTC had 73%-98% sensitivity and 89%-91% specificity to detect adenomas 6 mm and larger, compared with colonoscopy in seven studies. However, the risk of harm from low-dose ionizing radiation remains a consideration.
For diagnostic accuracy, colonoscopy showed per-person sensitivity of 89%-98% for adenomas 10 mm or larger, and 75%-93% for adenomas 6 mm or larger, in studies comparing it with CTC or as an adjunct to CTC. However, studies showing applicability to community practices were limited.
Fecal immunochemical tests (FITs) showed sensitivity ranging from 73% to 88% and specificity from 90% to 96%.
Data from five randomized, controlled trials evaluating multiple rounds of biennial screening using gFOBT showed a significant reduction in colorectal cancer mortality, from a relative risk of 0.91 at 19.5 years to a relative risk of 0.78 at 30 years.
Colonoscopy remains the standard by which other tests are assessed, although it has the highest risk of procedural complications, the researchers said. Three new randomized, controlled trials involving screening colonoscopy in average-risk adults scheduled for completion in 2021, 2026, and 2027, may yield more information on incidence and mortality, they added.
The evidence report and review was limited by its focus on average-risk adults; it did not address factors including screening for high-risk adults, availability and access to tests, potential risks of overdiagnosis, and overuse of screening after adenoma detection, the researchers said. In addition, “data are still needed on the differential uptake of and adherence to screening modalities and on continued adherence to repeated rounds of screening and diagnostic follow-up to screening over longer periods,” they said. However, they concluded, “colonoscopy, flexible sigmoidoscopy, CTC, and various stool tests have differing levels of evidence to support their use in CRC screening, ability to detect CRC and precursor lesions, and risk of serious adverse events in average-risk adults.”
The researchers had no relevant financial conflicts to disclose. The research was supported by the Agency for Healthcare Research and Quality under a contract with the U.S. Preventive Services Task Force.
The final recommendation statement is available online at USPSTF.
The recommendation puts emphasis on shared decision making between doctors and patients but does not provide much guidance on how to do it. Few, if any, practices would offer all of the screening tests and let patients choose. More commonly, a practice may offer one or two of the recommended options, and if patients do not accept those recommendations, would move on to other options.

|
Dr. David Lieberman |
Also missing from this document is a discussion of colon cancer prevention and screening. Data are provided in the supplements with regard to reduction in colon cancer incidence but not discussed in the text of the guideline. This could be an important element of shared decision making. Some patients may be willing to accept an option that is more likely to prevent cancer and avert the cost and morbidity of cancer care, even if it means more lifetime colonoscopies.
There is little discussion in the document about screening program quality. Programs that involve several steps may have issues with adherence, which may reduce effectiveness. Therefore, quality metrics ideally should be measured for all screening programs to ensure high-quality delivery of the program and adherence to the programmatic steps.
Dr. David Lieberman is vice president of the AGA Institute and chief, division of gastroenterology and hepatology, Oregon Health and Science University, Portland.
The recommendation puts emphasis on shared decision making between doctors and patients but does not provide much guidance on how to do it. Few, if any, practices would offer all of the screening tests and let patients choose. More commonly, a practice may offer one or two of the recommended options, and if patients do not accept those recommendations, would move on to other options.

|
Dr. David Lieberman |
Also missing from this document is a discussion of colon cancer prevention and screening. Data are provided in the supplements with regard to reduction in colon cancer incidence but not discussed in the text of the guideline. This could be an important element of shared decision making. Some patients may be willing to accept an option that is more likely to prevent cancer and avert the cost and morbidity of cancer care, even if it means more lifetime colonoscopies.
There is little discussion in the document about screening program quality. Programs that involve several steps may have issues with adherence, which may reduce effectiveness. Therefore, quality metrics ideally should be measured for all screening programs to ensure high-quality delivery of the program and adherence to the programmatic steps.
Dr. David Lieberman is vice president of the AGA Institute and chief, division of gastroenterology and hepatology, Oregon Health and Science University, Portland.
The recommendation puts emphasis on shared decision making between doctors and patients but does not provide much guidance on how to do it. Few, if any, practices would offer all of the screening tests and let patients choose. More commonly, a practice may offer one or two of the recommended options, and if patients do not accept those recommendations, would move on to other options.

|
Dr. David Lieberman |
Also missing from this document is a discussion of colon cancer prevention and screening. Data are provided in the supplements with regard to reduction in colon cancer incidence but not discussed in the text of the guideline. This could be an important element of shared decision making. Some patients may be willing to accept an option that is more likely to prevent cancer and avert the cost and morbidity of cancer care, even if it means more lifetime colonoscopies.
There is little discussion in the document about screening program quality. Programs that involve several steps may have issues with adherence, which may reduce effectiveness. Therefore, quality metrics ideally should be measured for all screening programs to ensure high-quality delivery of the program and adherence to the programmatic steps.
Dr. David Lieberman is vice president of the AGA Institute and chief, division of gastroenterology and hepatology, Oregon Health and Science University, Portland.
Colorectal cancer screening by a variety of methods is worthwhile and recommended for all adults aged 50-75 years, according to the latest recommendations from the U.S. Preventive Services Task Force. The USPSTF statement and summary of evidence were published in JAMA on June 15.
In addition, the USPSTF recommended selective screening for older adults aged 76-85 years, depending on their health status and screening history.
A team of researchers led by Dr. Jennifer S. Lin of Kaiser Permanente in Portland, Ore., reviewed studies on colorectal cancer screening published between Jan.1, 2008, and Dec. 31, 2014, with surveillance continuing through Feb. 23, 2016 (JAMA. 2016 Jun;315:2576-94 [doi: 10.1001/jama.2016.3332]). The USPSTF’s last recommendations on colorectal cancer screening were released in 2008.
“Although CRC screening has a large body of supporting evidence, additional research is still needed to weigh the relative benefits and harms of each test within a program of screening” for average-risk adults, the researchers noted.
The final recommendation statement includes three screening options that carry over from the 2008 guidelines: colonoscopy, sigmoidoscopy, and fecal immunochemical testing (FIT) with occult blood.
Other options now recommended include computed tomographic colonography (CTC), fecal immunochemical tests with DNA (FIT-DNA), guaiac-based fecal occult blood testing (gFOBT), and sigmoidoscopy plus FIT.
Some highlights from the analysis: Four randomized trials including 458,002 patients showed that one-time or two-time screening with flexible sigmoidoscopy was associated with decreased mortality from colorectal cancer, compared with no screening, for an incidence rate ratio of 0.73, the researchers wrote.
In addition, the researchers found that CTC had 73%-98% sensitivity and 89%-91% specificity to detect adenomas 6 mm and larger, compared with colonoscopy in seven studies. However, the risk of harm from low-dose ionizing radiation remains a consideration.
For diagnostic accuracy, colonoscopy showed per-person sensitivity of 89%-98% for adenomas 10 mm or larger, and 75%-93% for adenomas 6 mm or larger, in studies comparing it with CTC or as an adjunct to CTC. However, studies showing applicability to community practices were limited.
Fecal immunochemical tests (FITs) showed sensitivity ranging from 73% to 88% and specificity from 90% to 96%.
Data from five randomized, controlled trials evaluating multiple rounds of biennial screening using gFOBT showed a significant reduction in colorectal cancer mortality, from a relative risk of 0.91 at 19.5 years to a relative risk of 0.78 at 30 years.
Colonoscopy remains the standard by which other tests are assessed, although it has the highest risk of procedural complications, the researchers said. Three new randomized, controlled trials involving screening colonoscopy in average-risk adults scheduled for completion in 2021, 2026, and 2027, may yield more information on incidence and mortality, they added.
The evidence report and review was limited by its focus on average-risk adults; it did not address factors including screening for high-risk adults, availability and access to tests, potential risks of overdiagnosis, and overuse of screening after adenoma detection, the researchers said. In addition, “data are still needed on the differential uptake of and adherence to screening modalities and on continued adherence to repeated rounds of screening and diagnostic follow-up to screening over longer periods,” they said. However, they concluded, “colonoscopy, flexible sigmoidoscopy, CTC, and various stool tests have differing levels of evidence to support their use in CRC screening, ability to detect CRC and precursor lesions, and risk of serious adverse events in average-risk adults.”
The researchers had no relevant financial conflicts to disclose. The research was supported by the Agency for Healthcare Research and Quality under a contract with the U.S. Preventive Services Task Force.
The final recommendation statement is available online at USPSTF.
Colorectal cancer screening by a variety of methods is worthwhile and recommended for all adults aged 50-75 years, according to the latest recommendations from the U.S. Preventive Services Task Force. The USPSTF statement and summary of evidence were published in JAMA on June 15.
In addition, the USPSTF recommended selective screening for older adults aged 76-85 years, depending on their health status and screening history.
A team of researchers led by Dr. Jennifer S. Lin of Kaiser Permanente in Portland, Ore., reviewed studies on colorectal cancer screening published between Jan.1, 2008, and Dec. 31, 2014, with surveillance continuing through Feb. 23, 2016 (JAMA. 2016 Jun;315:2576-94 [doi: 10.1001/jama.2016.3332]). The USPSTF’s last recommendations on colorectal cancer screening were released in 2008.
“Although CRC screening has a large body of supporting evidence, additional research is still needed to weigh the relative benefits and harms of each test within a program of screening” for average-risk adults, the researchers noted.
The final recommendation statement includes three screening options that carry over from the 2008 guidelines: colonoscopy, sigmoidoscopy, and fecal immunochemical testing (FIT) with occult blood.
Other options now recommended include computed tomographic colonography (CTC), fecal immunochemical tests with DNA (FIT-DNA), guaiac-based fecal occult blood testing (gFOBT), and sigmoidoscopy plus FIT.
Some highlights from the analysis: Four randomized trials including 458,002 patients showed that one-time or two-time screening with flexible sigmoidoscopy was associated with decreased mortality from colorectal cancer, compared with no screening, for an incidence rate ratio of 0.73, the researchers wrote.
In addition, the researchers found that CTC had 73%-98% sensitivity and 89%-91% specificity to detect adenomas 6 mm and larger, compared with colonoscopy in seven studies. However, the risk of harm from low-dose ionizing radiation remains a consideration.
For diagnostic accuracy, colonoscopy showed per-person sensitivity of 89%-98% for adenomas 10 mm or larger, and 75%-93% for adenomas 6 mm or larger, in studies comparing it with CTC or as an adjunct to CTC. However, studies showing applicability to community practices were limited.
Fecal immunochemical tests (FITs) showed sensitivity ranging from 73% to 88% and specificity from 90% to 96%.
Data from five randomized, controlled trials evaluating multiple rounds of biennial screening using gFOBT showed a significant reduction in colorectal cancer mortality, from a relative risk of 0.91 at 19.5 years to a relative risk of 0.78 at 30 years.
Colonoscopy remains the standard by which other tests are assessed, although it has the highest risk of procedural complications, the researchers said. Three new randomized, controlled trials involving screening colonoscopy in average-risk adults scheduled for completion in 2021, 2026, and 2027, may yield more information on incidence and mortality, they added.
The evidence report and review was limited by its focus on average-risk adults; it did not address factors including screening for high-risk adults, availability and access to tests, potential risks of overdiagnosis, and overuse of screening after adenoma detection, the researchers said. In addition, “data are still needed on the differential uptake of and adherence to screening modalities and on continued adherence to repeated rounds of screening and diagnostic follow-up to screening over longer periods,” they said. However, they concluded, “colonoscopy, flexible sigmoidoscopy, CTC, and various stool tests have differing levels of evidence to support their use in CRC screening, ability to detect CRC and precursor lesions, and risk of serious adverse events in average-risk adults.”
The researchers had no relevant financial conflicts to disclose. The research was supported by the Agency for Healthcare Research and Quality under a contract with the U.S. Preventive Services Task Force.
The final recommendation statement is available online at USPSTF.
FROM JAMA
Key clinical point: Colorectal cancer screening is recommended for all adults aged 50-75 years, and several screening methods are supported by evidence-based research.
Major finding: In four randomized trials including 458,002 patients, one- or two-time screening via flexible sigmoidoscopy was associated with decreased mortality from colorectal cancer, compared with no screening, for an incidence rate ratio of 0.73.
Data source: Studies were selected based on searches of MEDLINE, PubMed, and the Cochrane Central Register of Controlled Trials.
Disclosures: The researchers had no relevant financial conflicts to disclose. The research was supported by AHRQ under a contract with the USPSTF.
Lasers and topicals both lighten solar lentigines
Quality-switched (QS) laser therapy with a ruby laser and topical triple-combination therapy (TCT) were similarly effective in lightening solar lentigines, based on data from a prospective, open label study of 15 adults. The findings were published in Dermatologic Surgery.
The QS ruby laser (QSRL) delivered longer-lasting skin-lightening results in less time, but the topical therapy is less expensive and has a lower risk of adverse events, wrote Dr. Laurence Imhof of the department of dermatology, University Hospital Zürich, and colleagues (Dermatol Surg. 2016;42:853-57. doi: 10.1097/DSS.0000000000000793). “Although therapy [for solar lentigines] is not medically indicated, there is a rising treatment demand for aesthetic reasons. Therefore, the treatment should be very safe and affordable,” they wrote.
The researchers compared the safety and efficacy of treating solar lentigines with a 694-nm QSRL and TCT (hydroquinone 5%, tretinoin 0.03%, and dexamethasone 0.03%). The patients included 14 women and 1 man with evenly distributed solar lentigines on the backs of both hands. Each patient was treated with 1-2 QSRL sessions on the back of the right hand and 7 weeks of once-daily application of TCT on the back of the left hand. The average age of the patients was 61 years; 13 were Fitzpatrick Skin Types II and III. Adverse events were mild and transient with both treatments, although the QSRL caused significantly more crusting and hyperpigmentation than the TCT.
Both treatments significantly reduced pigment at the end of treatment (day 56) and 12 weeks’ post treatment (140 days), compared with baseline. Treatment areas were evaluated at days 28, 56, and 140 by the treating physician, the patient, and a blinded physician (based on photos).The 6-point grading scale rated the degree of lightening/percentage of clearing, ranging from worse (less than 0%) to excellent (76%-100%).
At 56 days, the mean macroscopic improvement with QSRL laser was rated 3.5 by the treating physician and patient and 3 by a blinded physician; the mean macroscopic improvement with TCT was rated 2.4 by the treating physician, 2.2 by the patient, and 1.7 by the blinded physician. At 140 days, the mean macroscopic improvements with the laser were rated 2.9 by the treating physician, 2.7 by the patient, and 2.8 by the blinded physician, compared with 1.2, 1.1, and 0.7, respectively, with TCT.
The results were limited by the small size, short follow-up period, and open-label design, the researchers noted. However, the study is the first known to compare a laser and a topical treatment for the management of solar lentigines, and the findings suggest that both therapies can be recommended for the aesthetic treatment of solar lentigines, they said.
The TCT cream used in the study was provided by Louis Widmer SA, which provided a grant to the University of Zürich. The researchers had no financial conflicts to disclose.

|
Dr. Nazanin Saedi |
“We commend Imhof and colleagues for providing a long-awaited direct comparison of the Q-switched laser and topical triple-combination therapy,” wrote Dr. Laura M. Schilling and Dr. Nazanin Saedi, in an accompanying editorial (Dermatol Surg. 2016;42:8580859. doi: 10.1097/DSS.0000000000000792). Both treatments are viable options for patients, they emphasized. “It is fundamental, especially in this era of patient-centered care, to recognize that treatment options must be individualized. There are numerous variables to be considered when choosing the appropriate treatment for a patient including efficacy, skin type, cost, availability, and patient preference, among others.”
Dr. Schilling and Dr. Saedi are with the department of dermatology and cutaneous biology, Thomas Jefferson University, Philadelphia. They had no financial conflicts to disclose.

|
Dr. Nazanin Saedi |
“We commend Imhof and colleagues for providing a long-awaited direct comparison of the Q-switched laser and topical triple-combination therapy,” wrote Dr. Laura M. Schilling and Dr. Nazanin Saedi, in an accompanying editorial (Dermatol Surg. 2016;42:8580859. doi: 10.1097/DSS.0000000000000792). Both treatments are viable options for patients, they emphasized. “It is fundamental, especially in this era of patient-centered care, to recognize that treatment options must be individualized. There are numerous variables to be considered when choosing the appropriate treatment for a patient including efficacy, skin type, cost, availability, and patient preference, among others.”
Dr. Schilling and Dr. Saedi are with the department of dermatology and cutaneous biology, Thomas Jefferson University, Philadelphia. They had no financial conflicts to disclose.

|
Dr. Nazanin Saedi |
“We commend Imhof and colleagues for providing a long-awaited direct comparison of the Q-switched laser and topical triple-combination therapy,” wrote Dr. Laura M. Schilling and Dr. Nazanin Saedi, in an accompanying editorial (Dermatol Surg. 2016;42:8580859. doi: 10.1097/DSS.0000000000000792). Both treatments are viable options for patients, they emphasized. “It is fundamental, especially in this era of patient-centered care, to recognize that treatment options must be individualized. There are numerous variables to be considered when choosing the appropriate treatment for a patient including efficacy, skin type, cost, availability, and patient preference, among others.”
Dr. Schilling and Dr. Saedi are with the department of dermatology and cutaneous biology, Thomas Jefferson University, Philadelphia. They had no financial conflicts to disclose.
Quality-switched (QS) laser therapy with a ruby laser and topical triple-combination therapy (TCT) were similarly effective in lightening solar lentigines, based on data from a prospective, open label study of 15 adults. The findings were published in Dermatologic Surgery.
The QS ruby laser (QSRL) delivered longer-lasting skin-lightening results in less time, but the topical therapy is less expensive and has a lower risk of adverse events, wrote Dr. Laurence Imhof of the department of dermatology, University Hospital Zürich, and colleagues (Dermatol Surg. 2016;42:853-57. doi: 10.1097/DSS.0000000000000793). “Although therapy [for solar lentigines] is not medically indicated, there is a rising treatment demand for aesthetic reasons. Therefore, the treatment should be very safe and affordable,” they wrote.
The researchers compared the safety and efficacy of treating solar lentigines with a 694-nm QSRL and TCT (hydroquinone 5%, tretinoin 0.03%, and dexamethasone 0.03%). The patients included 14 women and 1 man with evenly distributed solar lentigines on the backs of both hands. Each patient was treated with 1-2 QSRL sessions on the back of the right hand and 7 weeks of once-daily application of TCT on the back of the left hand. The average age of the patients was 61 years; 13 were Fitzpatrick Skin Types II and III. Adverse events were mild and transient with both treatments, although the QSRL caused significantly more crusting and hyperpigmentation than the TCT.
Both treatments significantly reduced pigment at the end of treatment (day 56) and 12 weeks’ post treatment (140 days), compared with baseline. Treatment areas were evaluated at days 28, 56, and 140 by the treating physician, the patient, and a blinded physician (based on photos).The 6-point grading scale rated the degree of lightening/percentage of clearing, ranging from worse (less than 0%) to excellent (76%-100%).
At 56 days, the mean macroscopic improvement with QSRL laser was rated 3.5 by the treating physician and patient and 3 by a blinded physician; the mean macroscopic improvement with TCT was rated 2.4 by the treating physician, 2.2 by the patient, and 1.7 by the blinded physician. At 140 days, the mean macroscopic improvements with the laser were rated 2.9 by the treating physician, 2.7 by the patient, and 2.8 by the blinded physician, compared with 1.2, 1.1, and 0.7, respectively, with TCT.
The results were limited by the small size, short follow-up period, and open-label design, the researchers noted. However, the study is the first known to compare a laser and a topical treatment for the management of solar lentigines, and the findings suggest that both therapies can be recommended for the aesthetic treatment of solar lentigines, they said.
The TCT cream used in the study was provided by Louis Widmer SA, which provided a grant to the University of Zürich. The researchers had no financial conflicts to disclose.
Quality-switched (QS) laser therapy with a ruby laser and topical triple-combination therapy (TCT) were similarly effective in lightening solar lentigines, based on data from a prospective, open label study of 15 adults. The findings were published in Dermatologic Surgery.
The QS ruby laser (QSRL) delivered longer-lasting skin-lightening results in less time, but the topical therapy is less expensive and has a lower risk of adverse events, wrote Dr. Laurence Imhof of the department of dermatology, University Hospital Zürich, and colleagues (Dermatol Surg. 2016;42:853-57. doi: 10.1097/DSS.0000000000000793). “Although therapy [for solar lentigines] is not medically indicated, there is a rising treatment demand for aesthetic reasons. Therefore, the treatment should be very safe and affordable,” they wrote.
The researchers compared the safety and efficacy of treating solar lentigines with a 694-nm QSRL and TCT (hydroquinone 5%, tretinoin 0.03%, and dexamethasone 0.03%). The patients included 14 women and 1 man with evenly distributed solar lentigines on the backs of both hands. Each patient was treated with 1-2 QSRL sessions on the back of the right hand and 7 weeks of once-daily application of TCT on the back of the left hand. The average age of the patients was 61 years; 13 were Fitzpatrick Skin Types II and III. Adverse events were mild and transient with both treatments, although the QSRL caused significantly more crusting and hyperpigmentation than the TCT.
Both treatments significantly reduced pigment at the end of treatment (day 56) and 12 weeks’ post treatment (140 days), compared with baseline. Treatment areas were evaluated at days 28, 56, and 140 by the treating physician, the patient, and a blinded physician (based on photos).The 6-point grading scale rated the degree of lightening/percentage of clearing, ranging from worse (less than 0%) to excellent (76%-100%).
At 56 days, the mean macroscopic improvement with QSRL laser was rated 3.5 by the treating physician and patient and 3 by a blinded physician; the mean macroscopic improvement with TCT was rated 2.4 by the treating physician, 2.2 by the patient, and 1.7 by the blinded physician. At 140 days, the mean macroscopic improvements with the laser were rated 2.9 by the treating physician, 2.7 by the patient, and 2.8 by the blinded physician, compared with 1.2, 1.1, and 0.7, respectively, with TCT.
The results were limited by the small size, short follow-up period, and open-label design, the researchers noted. However, the study is the first known to compare a laser and a topical treatment for the management of solar lentigines, and the findings suggest that both therapies can be recommended for the aesthetic treatment of solar lentigines, they said.
The TCT cream used in the study was provided by Louis Widmer SA, which provided a grant to the University of Zürich. The researchers had no financial conflicts to disclose.
FROM DERMATOLOGIC SURGERY
Key clinical point: Both treatments significantly reduced pigment at the study conclusion (day 56) and at 12 weeks post treatment, compared with baseline.
Major finding: The mean macroscopic improvement with QSRL laser at 56 days was rated 3.5 by the treating physician and patient and 3 by a blinded physician; the mean macroscopic improvement with TCT was rated 2.4 by the treating physician, 2.2 by the patient, and 1.7 by the blinded physician.
Data source: A prospective, open-label 20-week study of 15 patients with solar lentigines.
Disclosures: The TCT cream used in the study was provided by Louis Widmer SA, which provided a grant to the University of Zürich. The researchers had no financial conflicts to disclose.
Don’t underestimate opioid use in HIV-positive adults
Prescription opioid use in HIV-positive individuals is underestimated, based on data from a 12-week study of medical and nonmedical prescription drug use in HIV patients attending a medical center and community clinic.
Of the 254 participants in the study, 43% reported medical use of prescription opioids, and 11% of the opioid use was reported as nonmedical.
Previous studies of prescription drug use in the HIV-positive population have mainly focused on nonmedical use, so “the existing research likely underestimates overall exposure of HIV-positive individuals to prescription medications,” wrote Abigail Norris Turner, PhD, of Ohio State University in Columbus, and her colleagues. Nonmedical use was defined as “using more medication than prescribed, using medication prescribed to someone else, or using medication for a purpose other than its prescribed use,” the researchers wrote.
The cross-sectional, self-administered survey involved data collection during July and August 2015 at a large, urban medical center (149 patients) and a community-based AIDS service organization (105 patients). Most of the study participants were male (91%), were identified as gay or bisexual (79%), and were at least 40 years old (61%).
Overall, 27% of the participants reported nonmedical use of any prescription drug during the previous year. Of these, 17% reported using one medication, 8% reported using two medications, and 2% reported using three or more medications. During the past month, 14% reported nonmedical use of a prescription drug, with 17% reporting one medication, 8% reporting two medications, and 2% reporting three or more medications. Reports of drug use were similar between the hospital group and clinic group.
The findings were limited by the cross-sectional nature of the study and the reliance on self-reports, the researchers noted; therefore, “we cannot make inferences about the causes and consequences of prescription medication use,” they said.
However, the results suggest that “it would be prudent for HIV providers to regularly review their opioid prescription practices to make sure they are appropriate; this review could also help identify patients at risk for opioid dependence and those who may benefit from referral to a pain medicine specialist or addiction medicine specialist,” they wrote.
The study was supported in part by the National Institutes of Health and the SOLAR Foundation Research Fund at the Ohio State University.
Read the full study here: AIDS Care 2016 Jun 20. doi: 10.1080/09540121.2016.1198746.
Prescription opioid use in HIV-positive individuals is underestimated, based on data from a 12-week study of medical and nonmedical prescription drug use in HIV patients attending a medical center and community clinic.
Of the 254 participants in the study, 43% reported medical use of prescription opioids, and 11% of the opioid use was reported as nonmedical.
Previous studies of prescription drug use in the HIV-positive population have mainly focused on nonmedical use, so “the existing research likely underestimates overall exposure of HIV-positive individuals to prescription medications,” wrote Abigail Norris Turner, PhD, of Ohio State University in Columbus, and her colleagues. Nonmedical use was defined as “using more medication than prescribed, using medication prescribed to someone else, or using medication for a purpose other than its prescribed use,” the researchers wrote.
The cross-sectional, self-administered survey involved data collection during July and August 2015 at a large, urban medical center (149 patients) and a community-based AIDS service organization (105 patients). Most of the study participants were male (91%), were identified as gay or bisexual (79%), and were at least 40 years old (61%).
Overall, 27% of the participants reported nonmedical use of any prescription drug during the previous year. Of these, 17% reported using one medication, 8% reported using two medications, and 2% reported using three or more medications. During the past month, 14% reported nonmedical use of a prescription drug, with 17% reporting one medication, 8% reporting two medications, and 2% reporting three or more medications. Reports of drug use were similar between the hospital group and clinic group.
The findings were limited by the cross-sectional nature of the study and the reliance on self-reports, the researchers noted; therefore, “we cannot make inferences about the causes and consequences of prescription medication use,” they said.
However, the results suggest that “it would be prudent for HIV providers to regularly review their opioid prescription practices to make sure they are appropriate; this review could also help identify patients at risk for opioid dependence and those who may benefit from referral to a pain medicine specialist or addiction medicine specialist,” they wrote.
The study was supported in part by the National Institutes of Health and the SOLAR Foundation Research Fund at the Ohio State University.
Read the full study here: AIDS Care 2016 Jun 20. doi: 10.1080/09540121.2016.1198746.
Prescription opioid use in HIV-positive individuals is underestimated, based on data from a 12-week study of medical and nonmedical prescription drug use in HIV patients attending a medical center and community clinic.
Of the 254 participants in the study, 43% reported medical use of prescription opioids, and 11% of the opioid use was reported as nonmedical.
Previous studies of prescription drug use in the HIV-positive population have mainly focused on nonmedical use, so “the existing research likely underestimates overall exposure of HIV-positive individuals to prescription medications,” wrote Abigail Norris Turner, PhD, of Ohio State University in Columbus, and her colleagues. Nonmedical use was defined as “using more medication than prescribed, using medication prescribed to someone else, or using medication for a purpose other than its prescribed use,” the researchers wrote.
The cross-sectional, self-administered survey involved data collection during July and August 2015 at a large, urban medical center (149 patients) and a community-based AIDS service organization (105 patients). Most of the study participants were male (91%), were identified as gay or bisexual (79%), and were at least 40 years old (61%).
Overall, 27% of the participants reported nonmedical use of any prescription drug during the previous year. Of these, 17% reported using one medication, 8% reported using two medications, and 2% reported using three or more medications. During the past month, 14% reported nonmedical use of a prescription drug, with 17% reporting one medication, 8% reporting two medications, and 2% reporting three or more medications. Reports of drug use were similar between the hospital group and clinic group.
The findings were limited by the cross-sectional nature of the study and the reliance on self-reports, the researchers noted; therefore, “we cannot make inferences about the causes and consequences of prescription medication use,” they said.
However, the results suggest that “it would be prudent for HIV providers to regularly review their opioid prescription practices to make sure they are appropriate; this review could also help identify patients at risk for opioid dependence and those who may benefit from referral to a pain medicine specialist or addiction medicine specialist,” they wrote.
The study was supported in part by the National Institutes of Health and the SOLAR Foundation Research Fund at the Ohio State University.
Read the full study here: AIDS Care 2016 Jun 20. doi: 10.1080/09540121.2016.1198746.
FROM AIDS CARE
Key clinical point: Opioid exposure in HIV-positive adults is more prevalent than previous research suggests.
Major finding: Based on data from 254 individuals, 43% reported medical opioid use, and 11% reported nonmedical opioid use.
Data source: A cross-sectional survey involving 254 HIV-positive adults at two study sites.
Disclosures: The study was supported in part by the National Institutes of Health and the SOLAR Foundation Research Fund at the Ohio State University.
Vasodilator, biomarkers present potential for preeclampsia care
Treatment with oral sildenafil citrate significantly extended gestation by 4 days, compared with placebo in women with preeclampsia in a randomized trial.
The findings come from one of a trio of studies looking at efforts to improve outcomes for women with preeclampsia, which were published online July 11 in Obstetrics & Gynecology.
“We hypothesized that the potential increase in in utero placental and fetoplacental blood flow with the use of sildenafil citrate may be associated with pregnancy prolongation (the primary study outcome) and improved maternal and perinatal outcomes,” wrote Alberto Trapani Jr., MD, PhD, of Hospital of Federal University of Santa Catarina, Florianopolis, Brazil, and his colleagues (Obstet Gynecol. 2016;128:253-259. doi: 10.1097/AOG.0000000000001518).
The researchers randomized 93 singleton pregnancies with preeclampsia at 24 to 33 weeks’ gestation to receive 50 mg oral sildenafil citrate (47 patients) or a placebo (46 patients) every 8 hours. Baseline characteristics were similar between the two groups.
Overall, pregnancies lasted an average of another 14.4 days in the sildenafil group, compared with 10.4 days in the placebo group. Additionally, the percent reduction in the pulsality indices of both uterine arteries and umbilical arteries were significantly higher in the sildenafil group, compared with the placebo group (22.5% vs. 2.1% and 18.5% vs. 2.5%, respectively). Maternal blood pressure before and 24 hours after randomization was lower with sildenafil. Incidence of adverse effects, perinatal morbidity, and mortality were similar between the two groups.
“These results, combined with the results of other studies, are promising,” although larger studies with earlier treatment initiation are needed, the researchers wrote. Evaluation of other phosphodiesterase type 5 inhibitors also may be useful in treating preeclampsia, they noted. Dr. Trapani and his colleagues reported having no financial conflicts.
Two other studies published in the same issue of Obstetrics & Gynecology focused on using biomarkers to help predict delivery times in women with suspected preeclampsia.
Any one of several single angiogenesis-related biomarkers can be a useful diagnostic test for preeclampsia, wrote Suzy Duckworth, MBBS, of King’s College London and her colleagues. In a prospective, observational study of 423 women with suspected preeclampsia, the researchers found that a combination of biomarkers including placental growth factor (PlGF) was not significantly better than PlGF alone (receiver operating curve area of 0.90 and 0.87, respectively) for predicting preeclampsia requiring delivery within 14 days. Two other single markers, soluble fms-like tyrosine kinase-1 (sFlt-1) and endoglin, yielded similar results; each demonstrated a receiver operating curve area of 0.83.
“It is currently difficult to distinguish preeclampsia of a severity that requires early delivery from other less serious phenotypes,” the researchers wrote. “An accurate biomarker (or panel of biomarkers) to enable prognosis of perinatal complications could have a substantial effect on management strategies, with the aim of minimizing adverse maternal and fetal outcomes,” they wrote (Obstet Gynecol. 2016;128:245-252. doi: 10.1097/AOG.0000000000001508).
Dr. Duckworth had no financial conflicts to disclose, but several of her coauthors disclosed relationships with pharmaceutical companies and one coauthor is a minority shareholder in Metabolomic Diagnostics, a company with an interest in preeclampsia biomarkers.
In another study including 1,041 women with suspected preeclampsia, those with a sFlt-1:PlGF ratio greater than 38 were nearly three times as likely to deliver on the day of the test than women with an sFlt-1:PlGF ratio of 38 or less, wrote Harald Zeisler, MD, of Medical University Vienna and his colleagues (Obstet Gynecol. 2016;128:261-269. doi: 10.1097/AOG.0000000000001525).
Women with an sFlt-1:PlGF ratio greater than 38 also had significantly shorter remaining times to delivery than women with an sFlt-1:PlGF ratio of 38 or less (median 17 days vs. 51 days). The study was a secondary analysis of an observational cohort study of women aged 18 years and older from 24 to 36 6/7 weeks’ gestation.
In a preterm birth analysis of 848 women, 71% of the 184 women with an sFlt-1:PlGF ratio greater than 38 had a preterm delivery, compared with 18% of the 664 women with an sFlt-1:PlGF ratio of 38 or less.
The future clinical use of the sFlt-1:PlGF ratio “may potentially assist in informing the health care team of an impending risk to the mother, fetus, or both that may require further assessment and medical intervention,” the researchers wrote.
Dr. Zeisler and several coauthors disclosed relationships with multiple companies, including Roche Diagnostics, which sponsored the study.
Treatment with oral sildenafil citrate significantly extended gestation by 4 days, compared with placebo in women with preeclampsia in a randomized trial.
The findings come from one of a trio of studies looking at efforts to improve outcomes for women with preeclampsia, which were published online July 11 in Obstetrics & Gynecology.
“We hypothesized that the potential increase in in utero placental and fetoplacental blood flow with the use of sildenafil citrate may be associated with pregnancy prolongation (the primary study outcome) and improved maternal and perinatal outcomes,” wrote Alberto Trapani Jr., MD, PhD, of Hospital of Federal University of Santa Catarina, Florianopolis, Brazil, and his colleagues (Obstet Gynecol. 2016;128:253-259. doi: 10.1097/AOG.0000000000001518).
The researchers randomized 93 singleton pregnancies with preeclampsia at 24 to 33 weeks’ gestation to receive 50 mg oral sildenafil citrate (47 patients) or a placebo (46 patients) every 8 hours. Baseline characteristics were similar between the two groups.
Overall, pregnancies lasted an average of another 14.4 days in the sildenafil group, compared with 10.4 days in the placebo group. Additionally, the percent reduction in the pulsality indices of both uterine arteries and umbilical arteries were significantly higher in the sildenafil group, compared with the placebo group (22.5% vs. 2.1% and 18.5% vs. 2.5%, respectively). Maternal blood pressure before and 24 hours after randomization was lower with sildenafil. Incidence of adverse effects, perinatal morbidity, and mortality were similar between the two groups.
“These results, combined with the results of other studies, are promising,” although larger studies with earlier treatment initiation are needed, the researchers wrote. Evaluation of other phosphodiesterase type 5 inhibitors also may be useful in treating preeclampsia, they noted. Dr. Trapani and his colleagues reported having no financial conflicts.
Two other studies published in the same issue of Obstetrics & Gynecology focused on using biomarkers to help predict delivery times in women with suspected preeclampsia.
Any one of several single angiogenesis-related biomarkers can be a useful diagnostic test for preeclampsia, wrote Suzy Duckworth, MBBS, of King’s College London and her colleagues. In a prospective, observational study of 423 women with suspected preeclampsia, the researchers found that a combination of biomarkers including placental growth factor (PlGF) was not significantly better than PlGF alone (receiver operating curve area of 0.90 and 0.87, respectively) for predicting preeclampsia requiring delivery within 14 days. Two other single markers, soluble fms-like tyrosine kinase-1 (sFlt-1) and endoglin, yielded similar results; each demonstrated a receiver operating curve area of 0.83.
“It is currently difficult to distinguish preeclampsia of a severity that requires early delivery from other less serious phenotypes,” the researchers wrote. “An accurate biomarker (or panel of biomarkers) to enable prognosis of perinatal complications could have a substantial effect on management strategies, with the aim of minimizing adverse maternal and fetal outcomes,” they wrote (Obstet Gynecol. 2016;128:245-252. doi: 10.1097/AOG.0000000000001508).
Dr. Duckworth had no financial conflicts to disclose, but several of her coauthors disclosed relationships with pharmaceutical companies and one coauthor is a minority shareholder in Metabolomic Diagnostics, a company with an interest in preeclampsia biomarkers.
In another study including 1,041 women with suspected preeclampsia, those with a sFlt-1:PlGF ratio greater than 38 were nearly three times as likely to deliver on the day of the test than women with an sFlt-1:PlGF ratio of 38 or less, wrote Harald Zeisler, MD, of Medical University Vienna and his colleagues (Obstet Gynecol. 2016;128:261-269. doi: 10.1097/AOG.0000000000001525).
Women with an sFlt-1:PlGF ratio greater than 38 also had significantly shorter remaining times to delivery than women with an sFlt-1:PlGF ratio of 38 or less (median 17 days vs. 51 days). The study was a secondary analysis of an observational cohort study of women aged 18 years and older from 24 to 36 6/7 weeks’ gestation.
In a preterm birth analysis of 848 women, 71% of the 184 women with an sFlt-1:PlGF ratio greater than 38 had a preterm delivery, compared with 18% of the 664 women with an sFlt-1:PlGF ratio of 38 or less.
The future clinical use of the sFlt-1:PlGF ratio “may potentially assist in informing the health care team of an impending risk to the mother, fetus, or both that may require further assessment and medical intervention,” the researchers wrote.
Dr. Zeisler and several coauthors disclosed relationships with multiple companies, including Roche Diagnostics, which sponsored the study.
Treatment with oral sildenafil citrate significantly extended gestation by 4 days, compared with placebo in women with preeclampsia in a randomized trial.
The findings come from one of a trio of studies looking at efforts to improve outcomes for women with preeclampsia, which were published online July 11 in Obstetrics & Gynecology.
“We hypothesized that the potential increase in in utero placental and fetoplacental blood flow with the use of sildenafil citrate may be associated with pregnancy prolongation (the primary study outcome) and improved maternal and perinatal outcomes,” wrote Alberto Trapani Jr., MD, PhD, of Hospital of Federal University of Santa Catarina, Florianopolis, Brazil, and his colleagues (Obstet Gynecol. 2016;128:253-259. doi: 10.1097/AOG.0000000000001518).
The researchers randomized 93 singleton pregnancies with preeclampsia at 24 to 33 weeks’ gestation to receive 50 mg oral sildenafil citrate (47 patients) or a placebo (46 patients) every 8 hours. Baseline characteristics were similar between the two groups.
Overall, pregnancies lasted an average of another 14.4 days in the sildenafil group, compared with 10.4 days in the placebo group. Additionally, the percent reduction in the pulsality indices of both uterine arteries and umbilical arteries were significantly higher in the sildenafil group, compared with the placebo group (22.5% vs. 2.1% and 18.5% vs. 2.5%, respectively). Maternal blood pressure before and 24 hours after randomization was lower with sildenafil. Incidence of adverse effects, perinatal morbidity, and mortality were similar between the two groups.
“These results, combined with the results of other studies, are promising,” although larger studies with earlier treatment initiation are needed, the researchers wrote. Evaluation of other phosphodiesterase type 5 inhibitors also may be useful in treating preeclampsia, they noted. Dr. Trapani and his colleagues reported having no financial conflicts.
Two other studies published in the same issue of Obstetrics & Gynecology focused on using biomarkers to help predict delivery times in women with suspected preeclampsia.
Any one of several single angiogenesis-related biomarkers can be a useful diagnostic test for preeclampsia, wrote Suzy Duckworth, MBBS, of King’s College London and her colleagues. In a prospective, observational study of 423 women with suspected preeclampsia, the researchers found that a combination of biomarkers including placental growth factor (PlGF) was not significantly better than PlGF alone (receiver operating curve area of 0.90 and 0.87, respectively) for predicting preeclampsia requiring delivery within 14 days. Two other single markers, soluble fms-like tyrosine kinase-1 (sFlt-1) and endoglin, yielded similar results; each demonstrated a receiver operating curve area of 0.83.
“It is currently difficult to distinguish preeclampsia of a severity that requires early delivery from other less serious phenotypes,” the researchers wrote. “An accurate biomarker (or panel of biomarkers) to enable prognosis of perinatal complications could have a substantial effect on management strategies, with the aim of minimizing adverse maternal and fetal outcomes,” they wrote (Obstet Gynecol. 2016;128:245-252. doi: 10.1097/AOG.0000000000001508).
Dr. Duckworth had no financial conflicts to disclose, but several of her coauthors disclosed relationships with pharmaceutical companies and one coauthor is a minority shareholder in Metabolomic Diagnostics, a company with an interest in preeclampsia biomarkers.
In another study including 1,041 women with suspected preeclampsia, those with a sFlt-1:PlGF ratio greater than 38 were nearly three times as likely to deliver on the day of the test than women with an sFlt-1:PlGF ratio of 38 or less, wrote Harald Zeisler, MD, of Medical University Vienna and his colleagues (Obstet Gynecol. 2016;128:261-269. doi: 10.1097/AOG.0000000000001525).
Women with an sFlt-1:PlGF ratio greater than 38 also had significantly shorter remaining times to delivery than women with an sFlt-1:PlGF ratio of 38 or less (median 17 days vs. 51 days). The study was a secondary analysis of an observational cohort study of women aged 18 years and older from 24 to 36 6/7 weeks’ gestation.
In a preterm birth analysis of 848 women, 71% of the 184 women with an sFlt-1:PlGF ratio greater than 38 had a preterm delivery, compared with 18% of the 664 women with an sFlt-1:PlGF ratio of 38 or less.
The future clinical use of the sFlt-1:PlGF ratio “may potentially assist in informing the health care team of an impending risk to the mother, fetus, or both that may require further assessment and medical intervention,” the researchers wrote.
Dr. Zeisler and several coauthors disclosed relationships with multiple companies, including Roche Diagnostics, which sponsored the study.
FROM OBSTETRICS & GYNECOLOGY
USPSTF leans away from pelvic screening
Current evidence is insufficient to support screening pelvic exams for the early detection of certain gynecologic conditions in asymptomatic, nonpregnant women, according to a draft recommendation statement issued by the U.S. Preventive Services Task Force.
The draft recommendations and draft evidence review are available online for public comment at www.uspreventiveservicestaskforce.org through July 25, 2016.
The USPSTF defined the lack of evidence to assess the balance of benefits vs. harms of screening pelvic exams as an “I” recommendation, meaning that the balance of benefits and harms cannot be determined. The draft recommendations note that many malignant conditions including gynecologic cancers; infectious diseases such as genital herpes: and benign conditions including polyps, cysts, and fibroids, may be found during a screening pelvic exam. However, potential harms include false-positive and false-negative results, and the potential for invasive treatment and unnecessary diagnostic work-ups.
This is the first statement by the USPSTF specifically addressing pelvic exams to screen for gynecologic conditions in asymptomatic, nonpregnant women. The statement does not change any existing recommendations, such as those for screening tests for early detection of cervical cancer, chlamydia, and gonorrhea.
Pelvic exams are currently covered as part of the well-woman visit under the Affordable Care Act. “Since the coding structure doesn’t parse out individual elements of the visit, this should not result in any coverage changes,” according to the American College of Obstetricians and Gynecologists communications office.
“ACOG is reviewing the USPSTF’s draft recommendation statement and the evidence upon which it is based to assess whether there is a need to update its guidance on the routine pelvic examination,” Dr. Thomas Gellhaus, president of ACOG, said in a statement. “ACOG recommends annual pelvic examinations for patients 21 years of age or older. However, the College recognizes that this recommendation is based on expert opinion, and limitations of the internal pelvic examination for screening should be recognized.”
“The annual well-woman visit is an opportunity for the patient and her ob.gyn. to discuss whether a pelvic examination is appropriate for her. ACOG promotes shared communication and decision making between the patient and the physician regarding pelvic examinations,” said Dr. Gellhaus of the University of Iowa Hospitals and Clinics in Iowa City.
According to the USPSTF draft statement, no studies “were identified that evaluated the benefit of screening with pelvic examination on all-cause mortality, disease-specific morbidity or mortality, or quality of life.” In addition, the USPSTF “found inadequate evidence on the harms of screening for a range of gynecologic conditions with pelvic examination” and “no studies quantified the amount of anxiety associated with screening pelvic examination.”
The draft recommendations are based on a review of the evidence on the benefits, potential harms, and accuracy of screening pelvic examinations for asymptomatic, nonpregnant women aged 18 years and older. The USPSTF concluded that research is lacking in several key areas including the accuracy and efficacy of screening pelvic exams to detect conditions other than trichomoniasis, genital herpes, bacterial vaginosis, and ovarian cancer; and the potential harms (including psychological) of screening pelvic examinations for asymptomatic women.
Current evidence is insufficient to support screening pelvic exams for the early detection of certain gynecologic conditions in asymptomatic, nonpregnant women, according to a draft recommendation statement issued by the U.S. Preventive Services Task Force.
The draft recommendations and draft evidence review are available online for public comment at www.uspreventiveservicestaskforce.org through July 25, 2016.
The USPSTF defined the lack of evidence to assess the balance of benefits vs. harms of screening pelvic exams as an “I” recommendation, meaning that the balance of benefits and harms cannot be determined. The draft recommendations note that many malignant conditions including gynecologic cancers; infectious diseases such as genital herpes: and benign conditions including polyps, cysts, and fibroids, may be found during a screening pelvic exam. However, potential harms include false-positive and false-negative results, and the potential for invasive treatment and unnecessary diagnostic work-ups.
This is the first statement by the USPSTF specifically addressing pelvic exams to screen for gynecologic conditions in asymptomatic, nonpregnant women. The statement does not change any existing recommendations, such as those for screening tests for early detection of cervical cancer, chlamydia, and gonorrhea.
Pelvic exams are currently covered as part of the well-woman visit under the Affordable Care Act. “Since the coding structure doesn’t parse out individual elements of the visit, this should not result in any coverage changes,” according to the American College of Obstetricians and Gynecologists communications office.
“ACOG is reviewing the USPSTF’s draft recommendation statement and the evidence upon which it is based to assess whether there is a need to update its guidance on the routine pelvic examination,” Dr. Thomas Gellhaus, president of ACOG, said in a statement. “ACOG recommends annual pelvic examinations for patients 21 years of age or older. However, the College recognizes that this recommendation is based on expert opinion, and limitations of the internal pelvic examination for screening should be recognized.”
“The annual well-woman visit is an opportunity for the patient and her ob.gyn. to discuss whether a pelvic examination is appropriate for her. ACOG promotes shared communication and decision making between the patient and the physician regarding pelvic examinations,” said Dr. Gellhaus of the University of Iowa Hospitals and Clinics in Iowa City.
According to the USPSTF draft statement, no studies “were identified that evaluated the benefit of screening with pelvic examination on all-cause mortality, disease-specific morbidity or mortality, or quality of life.” In addition, the USPSTF “found inadequate evidence on the harms of screening for a range of gynecologic conditions with pelvic examination” and “no studies quantified the amount of anxiety associated with screening pelvic examination.”
The draft recommendations are based on a review of the evidence on the benefits, potential harms, and accuracy of screening pelvic examinations for asymptomatic, nonpregnant women aged 18 years and older. The USPSTF concluded that research is lacking in several key areas including the accuracy and efficacy of screening pelvic exams to detect conditions other than trichomoniasis, genital herpes, bacterial vaginosis, and ovarian cancer; and the potential harms (including psychological) of screening pelvic examinations for asymptomatic women.
Current evidence is insufficient to support screening pelvic exams for the early detection of certain gynecologic conditions in asymptomatic, nonpregnant women, according to a draft recommendation statement issued by the U.S. Preventive Services Task Force.
The draft recommendations and draft evidence review are available online for public comment at www.uspreventiveservicestaskforce.org through July 25, 2016.
The USPSTF defined the lack of evidence to assess the balance of benefits vs. harms of screening pelvic exams as an “I” recommendation, meaning that the balance of benefits and harms cannot be determined. The draft recommendations note that many malignant conditions including gynecologic cancers; infectious diseases such as genital herpes: and benign conditions including polyps, cysts, and fibroids, may be found during a screening pelvic exam. However, potential harms include false-positive and false-negative results, and the potential for invasive treatment and unnecessary diagnostic work-ups.
This is the first statement by the USPSTF specifically addressing pelvic exams to screen for gynecologic conditions in asymptomatic, nonpregnant women. The statement does not change any existing recommendations, such as those for screening tests for early detection of cervical cancer, chlamydia, and gonorrhea.
Pelvic exams are currently covered as part of the well-woman visit under the Affordable Care Act. “Since the coding structure doesn’t parse out individual elements of the visit, this should not result in any coverage changes,” according to the American College of Obstetricians and Gynecologists communications office.
“ACOG is reviewing the USPSTF’s draft recommendation statement and the evidence upon which it is based to assess whether there is a need to update its guidance on the routine pelvic examination,” Dr. Thomas Gellhaus, president of ACOG, said in a statement. “ACOG recommends annual pelvic examinations for patients 21 years of age or older. However, the College recognizes that this recommendation is based on expert opinion, and limitations of the internal pelvic examination for screening should be recognized.”
“The annual well-woman visit is an opportunity for the patient and her ob.gyn. to discuss whether a pelvic examination is appropriate for her. ACOG promotes shared communication and decision making between the patient and the physician regarding pelvic examinations,” said Dr. Gellhaus of the University of Iowa Hospitals and Clinics in Iowa City.
According to the USPSTF draft statement, no studies “were identified that evaluated the benefit of screening with pelvic examination on all-cause mortality, disease-specific morbidity or mortality, or quality of life.” In addition, the USPSTF “found inadequate evidence on the harms of screening for a range of gynecologic conditions with pelvic examination” and “no studies quantified the amount of anxiety associated with screening pelvic examination.”
The draft recommendations are based on a review of the evidence on the benefits, potential harms, and accuracy of screening pelvic examinations for asymptomatic, nonpregnant women aged 18 years and older. The USPSTF concluded that research is lacking in several key areas including the accuracy and efficacy of screening pelvic exams to detect conditions other than trichomoniasis, genital herpes, bacterial vaginosis, and ovarian cancer; and the potential harms (including psychological) of screening pelvic examinations for asymptomatic women.