User login
Team creates bone marrow on a chip
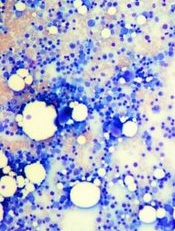
Image by Daniel E. Sabath
Engineered bone marrow grown in a microfluidic chip device mimics living bone marrow, according to research published in Tissue Engineering.
Experiments showed the engineered bone marrow responded in a way similar to living bone marrow when exposed to damaging radiation followed by treatment with compounds that aid in blood cell recovery.
The researchers said this new bone marrow-on-a-chip device holds promise for testing and developing improved radiation countermeasures.
Yu-suke Torisawa, PhD, of Kyoto University in Japan, and his colleagues conducted this research.
The team used a tissue engineering approach to induce formation of new marrow-containing bone in mice. They then surgically removed the bone, placed it in a microfluidic device, and continuously perfused it with medium in vitro.
Next, the researchers set out to determine if the device would keep the engineered bone marrow alive so they could perform tests on it.
To test the system, the team analyzed the dynamics of blood cell production and evaluated the radiation-protecting effects of granulocyte-colony stimulating factor (G-CSF) and bactericidal/permeability-increasing protein (BPI).
Experiments showed the microfluidic device could maintain hematopoietic stem and progenitor cells in normal proportions for at least 2 weeks in culture.
Over time, the researchers observed increases in the number of leukocytes and red blood cells in the microfluidic circulation. And they found that adding erythropoietin induced a significant increase in erythrocyte production.
When the researchers exposed the engineered bone marrow to gamma radiation, they saw reduced leukocyte production.
And when they treated the engineered bone marrow with G-CSF or BPI, the team saw significant increases in the number of hematopoietic stem cells and myeloid cells in the fluidic outflow.
On the other hand, BPI did not have such an effect on static bone marrow cultures. But the researchers pointed out that previous work has shown BPI can accelerate recovery from radiation-induced toxicity in vivo.
The team therefore concluded that, unlike static bone marrow cultures, engineered bone marrow grown in a microfluidic device effectively mimics the recovery response of bone marrow in the body. ![]()

Image by Daniel E. Sabath
Engineered bone marrow grown in a microfluidic chip device mimics living bone marrow, according to research published in Tissue Engineering.
Experiments showed the engineered bone marrow responded in a way similar to living bone marrow when exposed to damaging radiation followed by treatment with compounds that aid in blood cell recovery.
The researchers said this new bone marrow-on-a-chip device holds promise for testing and developing improved radiation countermeasures.
Yu-suke Torisawa, PhD, of Kyoto University in Japan, and his colleagues conducted this research.
The team used a tissue engineering approach to induce formation of new marrow-containing bone in mice. They then surgically removed the bone, placed it in a microfluidic device, and continuously perfused it with medium in vitro.
Next, the researchers set out to determine if the device would keep the engineered bone marrow alive so they could perform tests on it.
To test the system, the team analyzed the dynamics of blood cell production and evaluated the radiation-protecting effects of granulocyte-colony stimulating factor (G-CSF) and bactericidal/permeability-increasing protein (BPI).
Experiments showed the microfluidic device could maintain hematopoietic stem and progenitor cells in normal proportions for at least 2 weeks in culture.
Over time, the researchers observed increases in the number of leukocytes and red blood cells in the microfluidic circulation. And they found that adding erythropoietin induced a significant increase in erythrocyte production.
When the researchers exposed the engineered bone marrow to gamma radiation, they saw reduced leukocyte production.
And when they treated the engineered bone marrow with G-CSF or BPI, the team saw significant increases in the number of hematopoietic stem cells and myeloid cells in the fluidic outflow.
On the other hand, BPI did not have such an effect on static bone marrow cultures. But the researchers pointed out that previous work has shown BPI can accelerate recovery from radiation-induced toxicity in vivo.
The team therefore concluded that, unlike static bone marrow cultures, engineered bone marrow grown in a microfluidic device effectively mimics the recovery response of bone marrow in the body. ![]()

Image by Daniel E. Sabath
Engineered bone marrow grown in a microfluidic chip device mimics living bone marrow, according to research published in Tissue Engineering.
Experiments showed the engineered bone marrow responded in a way similar to living bone marrow when exposed to damaging radiation followed by treatment with compounds that aid in blood cell recovery.
The researchers said this new bone marrow-on-a-chip device holds promise for testing and developing improved radiation countermeasures.
Yu-suke Torisawa, PhD, of Kyoto University in Japan, and his colleagues conducted this research.
The team used a tissue engineering approach to induce formation of new marrow-containing bone in mice. They then surgically removed the bone, placed it in a microfluidic device, and continuously perfused it with medium in vitro.
Next, the researchers set out to determine if the device would keep the engineered bone marrow alive so they could perform tests on it.
To test the system, the team analyzed the dynamics of blood cell production and evaluated the radiation-protecting effects of granulocyte-colony stimulating factor (G-CSF) and bactericidal/permeability-increasing protein (BPI).
Experiments showed the microfluidic device could maintain hematopoietic stem and progenitor cells in normal proportions for at least 2 weeks in culture.
Over time, the researchers observed increases in the number of leukocytes and red blood cells in the microfluidic circulation. And they found that adding erythropoietin induced a significant increase in erythrocyte production.
When the researchers exposed the engineered bone marrow to gamma radiation, they saw reduced leukocyte production.
And when they treated the engineered bone marrow with G-CSF or BPI, the team saw significant increases in the number of hematopoietic stem cells and myeloid cells in the fluidic outflow.
On the other hand, BPI did not have such an effect on static bone marrow cultures. But the researchers pointed out that previous work has shown BPI can accelerate recovery from radiation-induced toxicity in vivo.
The team therefore concluded that, unlike static bone marrow cultures, engineered bone marrow grown in a microfluidic device effectively mimics the recovery response of bone marrow in the body. ![]()
Transparency doesn’t lower healthcare spending

Photo by Petr Kratochvil
Providing patients with a tool that enabled them to search for healthcare prices did not decrease their spending, according to a study published in JAMA.
Researchers studied the Truven Health Analytics Treatment Cost Calculator, an online price transparency tool that tells users how much they would pay out of pocket for services such as X-rays, lab tests, outpatient surgeries, or physician office visits at different sites.
The out-of-pocket cost estimates are based on the users’ health plan benefits and on how much they have already spent on healthcare during the year.
Two large national companies offered this tool to their employees in 2011 and 2012.
The researchers compared the healthcare spending patterns of employees (n=148,655) at these companies in the year before and after the tool was introduced with patterns among employees (n=295,983) of other companies that did not offer the tool.
Overall, having access to the tool was not associated with a reduction in outpatient spending, and subjects did not switch from more expensive outpatient hospital-based care to lower-cost settings.
The average outpatient spending among employees offered the tool was $2021 in the year before the tool was introduced and $2233 in the year after. Among control subjects, average outpatient spending increased from $1985 to $2138.
The average outpatient out-of-pocket spending among employees offered the tool was $507 in the year before it was introduced and $555 in the year after. In the control group, the average outpatient out-of-pocket spending increased from $490 to $520.
After the researchers adjusted for demographic and health characteristics, being offered the tool was associated with an average $59 increase in outpatient spending and an average $18 increase in out-of-pocket spending.
When the researchers looked only at patients with higher deductibles—who would be expected to have greater price-shopping incentives—they also found no evidence of reduction in spending.
“Despite large variation in healthcare prices, prevalence of high-deductible health plans, and widespread interest in price transparency, we did not find evidence that offering price transparency to employees generated savings,” said study author Sunita Desai, PhD, of Harvard Medical School in Boston, Massachusetts.
A possible explanation for this finding is that most patients did not actually use the tool. Only 10% of the employees who were offered the tool used it at least once in the first 12 months.
When patients did use the tool, more than half the searches were for relatively expensive services of over $1000.
“For expensive care that exceeds their deductible, patients may not see any reason to switch,” said study author Ateev Mehrotra, MD, also of Harvard Medical School. “They do not save by choosing a lower-cost provider, even if the health plan does.”
Still, the researchers said the tool does provide patients with valuable information, including their expected out-of-pocket costs, their deductible, and their health plan’s provider network.
“People might use the tools more—and focus more on choosing lower-priced care options—if they are combined with additional health plan benefit features that give greater incentive to price shop,” Dr Desai said. ![]()

Photo by Petr Kratochvil
Providing patients with a tool that enabled them to search for healthcare prices did not decrease their spending, according to a study published in JAMA.
Researchers studied the Truven Health Analytics Treatment Cost Calculator, an online price transparency tool that tells users how much they would pay out of pocket for services such as X-rays, lab tests, outpatient surgeries, or physician office visits at different sites.
The out-of-pocket cost estimates are based on the users’ health plan benefits and on how much they have already spent on healthcare during the year.
Two large national companies offered this tool to their employees in 2011 and 2012.
The researchers compared the healthcare spending patterns of employees (n=148,655) at these companies in the year before and after the tool was introduced with patterns among employees (n=295,983) of other companies that did not offer the tool.
Overall, having access to the tool was not associated with a reduction in outpatient spending, and subjects did not switch from more expensive outpatient hospital-based care to lower-cost settings.
The average outpatient spending among employees offered the tool was $2021 in the year before the tool was introduced and $2233 in the year after. Among control subjects, average outpatient spending increased from $1985 to $2138.
The average outpatient out-of-pocket spending among employees offered the tool was $507 in the year before it was introduced and $555 in the year after. In the control group, the average outpatient out-of-pocket spending increased from $490 to $520.
After the researchers adjusted for demographic and health characteristics, being offered the tool was associated with an average $59 increase in outpatient spending and an average $18 increase in out-of-pocket spending.
When the researchers looked only at patients with higher deductibles—who would be expected to have greater price-shopping incentives—they also found no evidence of reduction in spending.
“Despite large variation in healthcare prices, prevalence of high-deductible health plans, and widespread interest in price transparency, we did not find evidence that offering price transparency to employees generated savings,” said study author Sunita Desai, PhD, of Harvard Medical School in Boston, Massachusetts.
A possible explanation for this finding is that most patients did not actually use the tool. Only 10% of the employees who were offered the tool used it at least once in the first 12 months.
When patients did use the tool, more than half the searches were for relatively expensive services of over $1000.
“For expensive care that exceeds their deductible, patients may not see any reason to switch,” said study author Ateev Mehrotra, MD, also of Harvard Medical School. “They do not save by choosing a lower-cost provider, even if the health plan does.”
Still, the researchers said the tool does provide patients with valuable information, including their expected out-of-pocket costs, their deductible, and their health plan’s provider network.
“People might use the tools more—and focus more on choosing lower-priced care options—if they are combined with additional health plan benefit features that give greater incentive to price shop,” Dr Desai said. ![]()

Photo by Petr Kratochvil
Providing patients with a tool that enabled them to search for healthcare prices did not decrease their spending, according to a study published in JAMA.
Researchers studied the Truven Health Analytics Treatment Cost Calculator, an online price transparency tool that tells users how much they would pay out of pocket for services such as X-rays, lab tests, outpatient surgeries, or physician office visits at different sites.
The out-of-pocket cost estimates are based on the users’ health plan benefits and on how much they have already spent on healthcare during the year.
Two large national companies offered this tool to their employees in 2011 and 2012.
The researchers compared the healthcare spending patterns of employees (n=148,655) at these companies in the year before and after the tool was introduced with patterns among employees (n=295,983) of other companies that did not offer the tool.
Overall, having access to the tool was not associated with a reduction in outpatient spending, and subjects did not switch from more expensive outpatient hospital-based care to lower-cost settings.
The average outpatient spending among employees offered the tool was $2021 in the year before the tool was introduced and $2233 in the year after. Among control subjects, average outpatient spending increased from $1985 to $2138.
The average outpatient out-of-pocket spending among employees offered the tool was $507 in the year before it was introduced and $555 in the year after. In the control group, the average outpatient out-of-pocket spending increased from $490 to $520.
After the researchers adjusted for demographic and health characteristics, being offered the tool was associated with an average $59 increase in outpatient spending and an average $18 increase in out-of-pocket spending.
When the researchers looked only at patients with higher deductibles—who would be expected to have greater price-shopping incentives—they also found no evidence of reduction in spending.
“Despite large variation in healthcare prices, prevalence of high-deductible health plans, and widespread interest in price transparency, we did not find evidence that offering price transparency to employees generated savings,” said study author Sunita Desai, PhD, of Harvard Medical School in Boston, Massachusetts.
A possible explanation for this finding is that most patients did not actually use the tool. Only 10% of the employees who were offered the tool used it at least once in the first 12 months.
When patients did use the tool, more than half the searches were for relatively expensive services of over $1000.
“For expensive care that exceeds their deductible, patients may not see any reason to switch,” said study author Ateev Mehrotra, MD, also of Harvard Medical School. “They do not save by choosing a lower-cost provider, even if the health plan does.”
Still, the researchers said the tool does provide patients with valuable information, including their expected out-of-pocket costs, their deductible, and their health plan’s provider network.
“People might use the tools more—and focus more on choosing lower-priced care options—if they are combined with additional health plan benefit features that give greater incentive to price shop,” Dr Desai said. ![]()
FDA grants priority review for blinatumomab

and solution for infusion
Photo courtesy of Amgen
The US Food and Drug Administration (FDA) has accepted for priority review the supplemental biologics license application for blinatumomab (Blincyto) as a treatment for pediatric and adolescent patients with Philadelphia chromosome negative (Ph-) relapsed or refractory B-cell precursor acute lymphoblastic leukemia (ALL).
To grant an application priority review, the FDA must believe the drug would provide a significant improvement in the treatment, diagnosis, or prevention of a serious condition.
The priority review designation means the FDA’s goal is to take action on an application within 6 months, rather than the 10 months typically taken for a standard review.
The Prescription Drug User Fee Act target action date for the supplemental biologics license application for blinatumomab is September 1, 2016.
About blinatumomab
Blinatumomab is a bispecific, CD19-directed, CD3 T-cell engager (BiTE®) antibody construct that binds specifically to CD19 expressed on the surface of cells of B-lineage origin and CD3 expressed on the surface of T cells.
Blinatumomab was previously granted breakthrough therapy and priority review designations by the FDA and is now approved in the US for the treatment of adults with Ph- relapsed or refractory B-cell precursor ALL.
This indication is approved under accelerated approval. Continued approval for this indication may be contingent upon verification of clinical benefit in subsequent trials.
Blinatumomab is marketed by Amgen as Blincyto. The full US prescribing information is available at www.BLINCYTO.com.
‘205 trial
The supplemental biologics license application for blinatumomab in pediatric and adolescent patients is based on data from the phase 1/2 '205 trial.
In this study, researchers evaluated blinatumomab in patients younger than 18 years of age. The patients had B-cell precursor ALL that was refractory, had relapsed at least twice, or relapsed after an allogeneic hematopoietic stem cell transplant.
Treatment in this study has been completed, and subjects are being monitored for long-term efficacy. The data is being submitted for publication.
Preliminary data were presented at the 2014 ASH Annual Meeting (abstract 3703). ![]()

and solution for infusion
Photo courtesy of Amgen
The US Food and Drug Administration (FDA) has accepted for priority review the supplemental biologics license application for blinatumomab (Blincyto) as a treatment for pediatric and adolescent patients with Philadelphia chromosome negative (Ph-) relapsed or refractory B-cell precursor acute lymphoblastic leukemia (ALL).
To grant an application priority review, the FDA must believe the drug would provide a significant improvement in the treatment, diagnosis, or prevention of a serious condition.
The priority review designation means the FDA’s goal is to take action on an application within 6 months, rather than the 10 months typically taken for a standard review.
The Prescription Drug User Fee Act target action date for the supplemental biologics license application for blinatumomab is September 1, 2016.
About blinatumomab
Blinatumomab is a bispecific, CD19-directed, CD3 T-cell engager (BiTE®) antibody construct that binds specifically to CD19 expressed on the surface of cells of B-lineage origin and CD3 expressed on the surface of T cells.
Blinatumomab was previously granted breakthrough therapy and priority review designations by the FDA and is now approved in the US for the treatment of adults with Ph- relapsed or refractory B-cell precursor ALL.
This indication is approved under accelerated approval. Continued approval for this indication may be contingent upon verification of clinical benefit in subsequent trials.
Blinatumomab is marketed by Amgen as Blincyto. The full US prescribing information is available at www.BLINCYTO.com.
‘205 trial
The supplemental biologics license application for blinatumomab in pediatric and adolescent patients is based on data from the phase 1/2 '205 trial.
In this study, researchers evaluated blinatumomab in patients younger than 18 years of age. The patients had B-cell precursor ALL that was refractory, had relapsed at least twice, or relapsed after an allogeneic hematopoietic stem cell transplant.
Treatment in this study has been completed, and subjects are being monitored for long-term efficacy. The data is being submitted for publication.
Preliminary data were presented at the 2014 ASH Annual Meeting (abstract 3703). ![]()

and solution for infusion
Photo courtesy of Amgen
The US Food and Drug Administration (FDA) has accepted for priority review the supplemental biologics license application for blinatumomab (Blincyto) as a treatment for pediatric and adolescent patients with Philadelphia chromosome negative (Ph-) relapsed or refractory B-cell precursor acute lymphoblastic leukemia (ALL).
To grant an application priority review, the FDA must believe the drug would provide a significant improvement in the treatment, diagnosis, or prevention of a serious condition.
The priority review designation means the FDA’s goal is to take action on an application within 6 months, rather than the 10 months typically taken for a standard review.
The Prescription Drug User Fee Act target action date for the supplemental biologics license application for blinatumomab is September 1, 2016.
About blinatumomab
Blinatumomab is a bispecific, CD19-directed, CD3 T-cell engager (BiTE®) antibody construct that binds specifically to CD19 expressed on the surface of cells of B-lineage origin and CD3 expressed on the surface of T cells.
Blinatumomab was previously granted breakthrough therapy and priority review designations by the FDA and is now approved in the US for the treatment of adults with Ph- relapsed or refractory B-cell precursor ALL.
This indication is approved under accelerated approval. Continued approval for this indication may be contingent upon verification of clinical benefit in subsequent trials.
Blinatumomab is marketed by Amgen as Blincyto. The full US prescribing information is available at www.BLINCYTO.com.
‘205 trial
The supplemental biologics license application for blinatumomab in pediatric and adolescent patients is based on data from the phase 1/2 '205 trial.
In this study, researchers evaluated blinatumomab in patients younger than 18 years of age. The patients had B-cell precursor ALL that was refractory, had relapsed at least twice, or relapsed after an allogeneic hematopoietic stem cell transplant.
Treatment in this study has been completed, and subjects are being monitored for long-term efficacy. The data is being submitted for publication.
Preliminary data were presented at the 2014 ASH Annual Meeting (abstract 3703). ![]()
Low fitness levels linked to platelet activation in women

Women with poor physical fitness may have an increased risk of thrombotic events due to increased platelet activation, according to research published in Medicine & Science in Sports & Exercise.
In a study of 62 young women, those with poor cardiorespiratory fitness (CRF) had significantly higher platelet activation than women with average to very good CRF.
However, the study also showed that women with poor CRF were able to achieve normal platelet function by increasing their physical fitness.
The women achieved platelet normalization via endurance training—running for a maximum of 40 minutes—3 times a week over a 2-month period.
“Latently activated platelets release a number of mediators that can encourage the development of atherosclerotic vascular changes,” said study author Stefan Heber, of the Medical University of Vienna in Austria.
“If poor physical fitness is accompanied by a higher level of platelet activation, one can conclude that it also has an influence upon the early stages of this pathogenesis. The training effects we’ve found here are consistent with epidemiological data, according to which fit people have an approximately 40% lower risk of cardiovascular events than those who were physically inactive.”
To uncover their findings, Heber and his colleagues analyzed different aspects of platelet function in healthy, non-smoking women with low CRF, medium CRF, and high CRF.
The team assessed CRF using maximal oxygen consumption (VO2max), which was determined by an incremental treadmill exercise test. VO2max criteria were:
- less than 45 ml/min/kg bodyweight for the low CRF group
- 45 to 55 ml/min/kg bodyweight for the medium CRF group
- more than 55 ml/min/kg bodyweight for the high CRF group.
The researchers assessed platelet activation state and platelet reactivity in these groups of women by evaluating basal and agonist-induced surface expression of CD62P and CD40L, as well as the intraplatelet amount of reactive oxygen species.
The team observed higher basal platelet activation and agonist-induced platelet reactivity in the low CRF group, when compared to the medium and high CRF groups. However, platelet activation and reactivity were roughly the same in the medium and high CRF groups.
For the low CRF group, the researchers assessed platelet function again after the women completed a supervised endurance training program that lasted 2 menstrual cycles. The team found this program was able to normalize platelet function. ![]()

Women with poor physical fitness may have an increased risk of thrombotic events due to increased platelet activation, according to research published in Medicine & Science in Sports & Exercise.
In a study of 62 young women, those with poor cardiorespiratory fitness (CRF) had significantly higher platelet activation than women with average to very good CRF.
However, the study also showed that women with poor CRF were able to achieve normal platelet function by increasing their physical fitness.
The women achieved platelet normalization via endurance training—running for a maximum of 40 minutes—3 times a week over a 2-month period.
“Latently activated platelets release a number of mediators that can encourage the development of atherosclerotic vascular changes,” said study author Stefan Heber, of the Medical University of Vienna in Austria.
“If poor physical fitness is accompanied by a higher level of platelet activation, one can conclude that it also has an influence upon the early stages of this pathogenesis. The training effects we’ve found here are consistent with epidemiological data, according to which fit people have an approximately 40% lower risk of cardiovascular events than those who were physically inactive.”
To uncover their findings, Heber and his colleagues analyzed different aspects of platelet function in healthy, non-smoking women with low CRF, medium CRF, and high CRF.
The team assessed CRF using maximal oxygen consumption (VO2max), which was determined by an incremental treadmill exercise test. VO2max criteria were:
- less than 45 ml/min/kg bodyweight for the low CRF group
- 45 to 55 ml/min/kg bodyweight for the medium CRF group
- more than 55 ml/min/kg bodyweight for the high CRF group.
The researchers assessed platelet activation state and platelet reactivity in these groups of women by evaluating basal and agonist-induced surface expression of CD62P and CD40L, as well as the intraplatelet amount of reactive oxygen species.
The team observed higher basal platelet activation and agonist-induced platelet reactivity in the low CRF group, when compared to the medium and high CRF groups. However, platelet activation and reactivity were roughly the same in the medium and high CRF groups.
For the low CRF group, the researchers assessed platelet function again after the women completed a supervised endurance training program that lasted 2 menstrual cycles. The team found this program was able to normalize platelet function. ![]()

Women with poor physical fitness may have an increased risk of thrombotic events due to increased platelet activation, according to research published in Medicine & Science in Sports & Exercise.
In a study of 62 young women, those with poor cardiorespiratory fitness (CRF) had significantly higher platelet activation than women with average to very good CRF.
However, the study also showed that women with poor CRF were able to achieve normal platelet function by increasing their physical fitness.
The women achieved platelet normalization via endurance training—running for a maximum of 40 minutes—3 times a week over a 2-month period.
“Latently activated platelets release a number of mediators that can encourage the development of atherosclerotic vascular changes,” said study author Stefan Heber, of the Medical University of Vienna in Austria.
“If poor physical fitness is accompanied by a higher level of platelet activation, one can conclude that it also has an influence upon the early stages of this pathogenesis. The training effects we’ve found here are consistent with epidemiological data, according to which fit people have an approximately 40% lower risk of cardiovascular events than those who were physically inactive.”
To uncover their findings, Heber and his colleagues analyzed different aspects of platelet function in healthy, non-smoking women with low CRF, medium CRF, and high CRF.
The team assessed CRF using maximal oxygen consumption (VO2max), which was determined by an incremental treadmill exercise test. VO2max criteria were:
- less than 45 ml/min/kg bodyweight for the low CRF group
- 45 to 55 ml/min/kg bodyweight for the medium CRF group
- more than 55 ml/min/kg bodyweight for the high CRF group.
The researchers assessed platelet activation state and platelet reactivity in these groups of women by evaluating basal and agonist-induced surface expression of CD62P and CD40L, as well as the intraplatelet amount of reactive oxygen species.
The team observed higher basal platelet activation and agonist-induced platelet reactivity in the low CRF group, when compared to the medium and high CRF groups. However, platelet activation and reactivity were roughly the same in the medium and high CRF groups.
For the low CRF group, the researchers assessed platelet function again after the women completed a supervised endurance training program that lasted 2 menstrual cycles. The team found this program was able to normalize platelet function. ![]()
Drug produces similar results in older and younger ALL patients

Photo from MD Anderson
Data from two phase 2 studies suggests single-agent blinatumomab produces similar outcomes in adults with relapsed/refractory acute lymphoblastic leukemia (ALL), regardless of age.
Patients age 65 and older had similar hematologic response rates and relapse-free survival rates as patients younger than 65.
The incidence of grade 3 or higher adverse events (AEs) was similar between the age groups as well.
Older patients did have more serious AEs, however. And they had more neurologic events, but these were reversible.
Hagop M. Kantarjian, MD, of the University of Texas MD Anderson Cancer Center in Houston, and his colleagues reported these results in Cancer. The research was funded by Amgen Inc., makers of blinatumomab.
Patients
The researchers examined 261 adults with relapsed/refractory ALL who were enrolled in 2 different studies. There were 36 patients who were 65 or older and 225 patients who were younger than 65. The median ages were 70 (range, 65-79) and 34 (range, 18-64), respectively.
Among the older patients, 14% had primary refractory disease, 67% had 1 prior relapse, 14% had 2 prior relapses, and 6% had 3 or more. Among the younger patients, 9% had primary refractory disease, 55% had 1 prior relapse, 26% had 2 prior relapses, and 10% had 3 or more.
The younger patients were more likely to have received an allogeneic hematopoietic stem cell transplant (allo-HSCT) than the older patients—37% and 11%, respectively.
But older patients were more likely to have mild renal impairment (42% vs 13%) or moderate renal impairment (22% vs 1%).
Treatment
All patients received blinatumomab, and stepwise dosing was used to reduce the risk of cytokine release syndrome. A treatment cycle consisted of 4 weeks of continuous intravenous infusions, followed by a 2-week treatment-free interval.
The patients received 2 initial cycles. If they achieved a complete remission (CR) or CR with partial hematologic recovery (CRh) at this point, they could receive an additional 3 cycles as consolidation, unless they were scheduled to receive an allo-HSCT.
Patients also received intrathecal prophylaxis with dexamethasone and/or steroids, cytarabine, and methotrexate. And patients with a high blast percentage at baseline received a pre-phase treatment with dexamethasone and/or cyclophosphamide.
Older patients received a median of 2 cycles of blinatumomab (range, 1-6), as did the younger patients (range, 1-7).
Response and survival
Fifty-six percent of the older patients (20/36) achieved a CR/CRh during the first 2 cycles of blinatumomab, as did 46% of the younger patients (46/225). There were 14 CRs among the older patients (39%) and 78 CRs among the younger patients (35%).
There were 12 complete minimal residual disease responses among older patients (60% of responders) and 73 among the younger patients (70% of responders).
Of the responders, 3 older patients (15%) and 61 younger patients (59%) went on to allo-HSCT. Most of the patients received a transplant while in remission. However, 1 of the older patients and 8 of the younger patients went to transplant after an initial response to blinatumomab that was followed by a relapse.
The median relapse-free survival was 7.4 months for both age groups. The median overall survival was 5.5 months for older patients and 7.6 months for younger patients.
Safety
All of the older patients had at least 1 AE, and all but 1 of the younger patients had at least 1 AE. Older patients had higher rates of peripheral edema (42% vs 24%), fatigue (28% vs 18%), and dizziness (25% vs 11%) of any grade.
The incidence of grade 3 or higher AEs was similar between the groups—86% in the older group and 80% in the younger group. The same was true for AEs leading to treatment discontinuation—22% and 19%, respectively.
However, there was a higher incidence of serious AEs in the older patients (72% vs 64%). Device-related infection and encephalopathy were more common among older patients than younger patients (both 11% vs 3%).
The incidence of cytokine release syndrome was higher in the older group than the younger group—19% and 10%, respectively.
Older patients also had more neurologic events of any grade (72% vs 48%) and more grade 3 or higher neurologic events (28% vs 13%). However, all neurologic events were reversed by temporarily or permanently discontinuing blinatumomab.
There were 7 fatal treatment-emergent AEs in the older adults, including pneumonia (n=3), B-cell lymphoma (n=1), and disease progression (n=3). None of the fatal AEs were considered treatment-related. And none of the patients who were in remission died during treatment with blinatumomab. ![]()

Photo from MD Anderson
Data from two phase 2 studies suggests single-agent blinatumomab produces similar outcomes in adults with relapsed/refractory acute lymphoblastic leukemia (ALL), regardless of age.
Patients age 65 and older had similar hematologic response rates and relapse-free survival rates as patients younger than 65.
The incidence of grade 3 or higher adverse events (AEs) was similar between the age groups as well.
Older patients did have more serious AEs, however. And they had more neurologic events, but these were reversible.
Hagop M. Kantarjian, MD, of the University of Texas MD Anderson Cancer Center in Houston, and his colleagues reported these results in Cancer. The research was funded by Amgen Inc., makers of blinatumomab.
Patients
The researchers examined 261 adults with relapsed/refractory ALL who were enrolled in 2 different studies. There were 36 patients who were 65 or older and 225 patients who were younger than 65. The median ages were 70 (range, 65-79) and 34 (range, 18-64), respectively.
Among the older patients, 14% had primary refractory disease, 67% had 1 prior relapse, 14% had 2 prior relapses, and 6% had 3 or more. Among the younger patients, 9% had primary refractory disease, 55% had 1 prior relapse, 26% had 2 prior relapses, and 10% had 3 or more.
The younger patients were more likely to have received an allogeneic hematopoietic stem cell transplant (allo-HSCT) than the older patients—37% and 11%, respectively.
But older patients were more likely to have mild renal impairment (42% vs 13%) or moderate renal impairment (22% vs 1%).
Treatment
All patients received blinatumomab, and stepwise dosing was used to reduce the risk of cytokine release syndrome. A treatment cycle consisted of 4 weeks of continuous intravenous infusions, followed by a 2-week treatment-free interval.
The patients received 2 initial cycles. If they achieved a complete remission (CR) or CR with partial hematologic recovery (CRh) at this point, they could receive an additional 3 cycles as consolidation, unless they were scheduled to receive an allo-HSCT.
Patients also received intrathecal prophylaxis with dexamethasone and/or steroids, cytarabine, and methotrexate. And patients with a high blast percentage at baseline received a pre-phase treatment with dexamethasone and/or cyclophosphamide.
Older patients received a median of 2 cycles of blinatumomab (range, 1-6), as did the younger patients (range, 1-7).
Response and survival
Fifty-six percent of the older patients (20/36) achieved a CR/CRh during the first 2 cycles of blinatumomab, as did 46% of the younger patients (46/225). There were 14 CRs among the older patients (39%) and 78 CRs among the younger patients (35%).
There were 12 complete minimal residual disease responses among older patients (60% of responders) and 73 among the younger patients (70% of responders).
Of the responders, 3 older patients (15%) and 61 younger patients (59%) went on to allo-HSCT. Most of the patients received a transplant while in remission. However, 1 of the older patients and 8 of the younger patients went to transplant after an initial response to blinatumomab that was followed by a relapse.
The median relapse-free survival was 7.4 months for both age groups. The median overall survival was 5.5 months for older patients and 7.6 months for younger patients.
Safety
All of the older patients had at least 1 AE, and all but 1 of the younger patients had at least 1 AE. Older patients had higher rates of peripheral edema (42% vs 24%), fatigue (28% vs 18%), and dizziness (25% vs 11%) of any grade.
The incidence of grade 3 or higher AEs was similar between the groups—86% in the older group and 80% in the younger group. The same was true for AEs leading to treatment discontinuation—22% and 19%, respectively.
However, there was a higher incidence of serious AEs in the older patients (72% vs 64%). Device-related infection and encephalopathy were more common among older patients than younger patients (both 11% vs 3%).
The incidence of cytokine release syndrome was higher in the older group than the younger group—19% and 10%, respectively.
Older patients also had more neurologic events of any grade (72% vs 48%) and more grade 3 or higher neurologic events (28% vs 13%). However, all neurologic events were reversed by temporarily or permanently discontinuing blinatumomab.
There were 7 fatal treatment-emergent AEs in the older adults, including pneumonia (n=3), B-cell lymphoma (n=1), and disease progression (n=3). None of the fatal AEs were considered treatment-related. And none of the patients who were in remission died during treatment with blinatumomab. ![]()

Photo from MD Anderson
Data from two phase 2 studies suggests single-agent blinatumomab produces similar outcomes in adults with relapsed/refractory acute lymphoblastic leukemia (ALL), regardless of age.
Patients age 65 and older had similar hematologic response rates and relapse-free survival rates as patients younger than 65.
The incidence of grade 3 or higher adverse events (AEs) was similar between the age groups as well.
Older patients did have more serious AEs, however. And they had more neurologic events, but these were reversible.
Hagop M. Kantarjian, MD, of the University of Texas MD Anderson Cancer Center in Houston, and his colleagues reported these results in Cancer. The research was funded by Amgen Inc., makers of blinatumomab.
Patients
The researchers examined 261 adults with relapsed/refractory ALL who were enrolled in 2 different studies. There were 36 patients who were 65 or older and 225 patients who were younger than 65. The median ages were 70 (range, 65-79) and 34 (range, 18-64), respectively.
Among the older patients, 14% had primary refractory disease, 67% had 1 prior relapse, 14% had 2 prior relapses, and 6% had 3 or more. Among the younger patients, 9% had primary refractory disease, 55% had 1 prior relapse, 26% had 2 prior relapses, and 10% had 3 or more.
The younger patients were more likely to have received an allogeneic hematopoietic stem cell transplant (allo-HSCT) than the older patients—37% and 11%, respectively.
But older patients were more likely to have mild renal impairment (42% vs 13%) or moderate renal impairment (22% vs 1%).
Treatment
All patients received blinatumomab, and stepwise dosing was used to reduce the risk of cytokine release syndrome. A treatment cycle consisted of 4 weeks of continuous intravenous infusions, followed by a 2-week treatment-free interval.
The patients received 2 initial cycles. If they achieved a complete remission (CR) or CR with partial hematologic recovery (CRh) at this point, they could receive an additional 3 cycles as consolidation, unless they were scheduled to receive an allo-HSCT.
Patients also received intrathecal prophylaxis with dexamethasone and/or steroids, cytarabine, and methotrexate. And patients with a high blast percentage at baseline received a pre-phase treatment with dexamethasone and/or cyclophosphamide.
Older patients received a median of 2 cycles of blinatumomab (range, 1-6), as did the younger patients (range, 1-7).
Response and survival
Fifty-six percent of the older patients (20/36) achieved a CR/CRh during the first 2 cycles of blinatumomab, as did 46% of the younger patients (46/225). There were 14 CRs among the older patients (39%) and 78 CRs among the younger patients (35%).
There were 12 complete minimal residual disease responses among older patients (60% of responders) and 73 among the younger patients (70% of responders).
Of the responders, 3 older patients (15%) and 61 younger patients (59%) went on to allo-HSCT. Most of the patients received a transplant while in remission. However, 1 of the older patients and 8 of the younger patients went to transplant after an initial response to blinatumomab that was followed by a relapse.
The median relapse-free survival was 7.4 months for both age groups. The median overall survival was 5.5 months for older patients and 7.6 months for younger patients.
Safety
All of the older patients had at least 1 AE, and all but 1 of the younger patients had at least 1 AE. Older patients had higher rates of peripheral edema (42% vs 24%), fatigue (28% vs 18%), and dizziness (25% vs 11%) of any grade.
The incidence of grade 3 or higher AEs was similar between the groups—86% in the older group and 80% in the younger group. The same was true for AEs leading to treatment discontinuation—22% and 19%, respectively.
However, there was a higher incidence of serious AEs in the older patients (72% vs 64%). Device-related infection and encephalopathy were more common among older patients than younger patients (both 11% vs 3%).
The incidence of cytokine release syndrome was higher in the older group than the younger group—19% and 10%, respectively.
Older patients also had more neurologic events of any grade (72% vs 48%) and more grade 3 or higher neurologic events (28% vs 13%). However, all neurologic events were reversed by temporarily or permanently discontinuing blinatumomab.
There were 7 fatal treatment-emergent AEs in the older adults, including pneumonia (n=3), B-cell lymphoma (n=1), and disease progression (n=3). None of the fatal AEs were considered treatment-related. And none of the patients who were in remission died during treatment with blinatumomab. ![]()
Team identifies new function of Fanconi anemia genes

Image by Sarah Pfau
Research published in Cell has revealed a new function of genes in the Fanconi anemia (FA) pathway, and investigators believe this finding could have
implications for the treatment of FA and related disorders.
The team found that FA genes are required for selective autophagy.
In particular, the FANCC gene plays a key role in 2 types of selective autophagy: virophagy (the removal of viruses inside the cell) and mitophagy (the removal of mitochondria).
Experiments in mice showed that genetic deletion of FANCC blocks virophagy and increases the animals’ susceptibility to lethal viral encephalitis.
The investigators also found that FANCC protein is required for the clearance of damaged mitochondria and decreases the production of mitochondrial reactive oxygen species and inflammasome activation.
And other genes in the FA pathway are required for mitophagy as well—FANCA, FANCF, FANCL, FANCD2, BRCA1, and BRCA2.
“There’s increasing evidence that the failure of cells to appropriately clear damaged mitochondria leads to abnormal activation of the inflammasome—a process that is emerging as an important contributor to many different diseases,” said study author Beth Levine, MD, of UT Southwestern Medical Center in Dallas, Texas.
“The finding that FA genes function in clearing mitochondria and decreasing inflammasome activation provides a potential new inflammasome-targeted avenue of therapy for patients with diseases related to mutations in the FA genes.”
FA pathway genes were already known to play a role in DNA repair. The investigators said this new link to autophagy opens up unexplored horizons for understanding the function of these genes in human health and disease.
“Our findings suggest a novel mechanism by which mutations in FA genes may lead to the clinical manifestations in patients with FA and to cancers in patients with mutations in FA genes,” said study author Rhea Sumpter, MD, PhD, of UT Southwestern Medical Center.
“We’ve shown that this new function of the FA genes in the selective autophagy pathways does not depend on their role in DNA repair.”
In addition, the autophagy function may partly explain why patients with FA are highly susceptible to infection and cancer, Dr Levine said.
While further research is needed to understand how these findings may be used to treat disease, the investigators said they have identified a novel avenue for developing potential therapies for FA and cancer patients.
“I believe the clearest therapeutic possibilities to come from our study results are the development of new FA agents that target the inflammasome and production of interleukin 1 beta (IL-1β), a pro-inflammatory cytokine,” Dr Sumpter said.
“Clinically, IL-1β signaling has been targeted with FDA-approved drugs very successfully in several auto-inflammatory diseases that involve excessive inflammasome activation. Our results suggest that FA patients may also benefit from these therapies.” ![]()

Image by Sarah Pfau
Research published in Cell has revealed a new function of genes in the Fanconi anemia (FA) pathway, and investigators believe this finding could have
implications for the treatment of FA and related disorders.
The team found that FA genes are required for selective autophagy.
In particular, the FANCC gene plays a key role in 2 types of selective autophagy: virophagy (the removal of viruses inside the cell) and mitophagy (the removal of mitochondria).
Experiments in mice showed that genetic deletion of FANCC blocks virophagy and increases the animals’ susceptibility to lethal viral encephalitis.
The investigators also found that FANCC protein is required for the clearance of damaged mitochondria and decreases the production of mitochondrial reactive oxygen species and inflammasome activation.
And other genes in the FA pathway are required for mitophagy as well—FANCA, FANCF, FANCL, FANCD2, BRCA1, and BRCA2.
“There’s increasing evidence that the failure of cells to appropriately clear damaged mitochondria leads to abnormal activation of the inflammasome—a process that is emerging as an important contributor to many different diseases,” said study author Beth Levine, MD, of UT Southwestern Medical Center in Dallas, Texas.
“The finding that FA genes function in clearing mitochondria and decreasing inflammasome activation provides a potential new inflammasome-targeted avenue of therapy for patients with diseases related to mutations in the FA genes.”
FA pathway genes were already known to play a role in DNA repair. The investigators said this new link to autophagy opens up unexplored horizons for understanding the function of these genes in human health and disease.
“Our findings suggest a novel mechanism by which mutations in FA genes may lead to the clinical manifestations in patients with FA and to cancers in patients with mutations in FA genes,” said study author Rhea Sumpter, MD, PhD, of UT Southwestern Medical Center.
“We’ve shown that this new function of the FA genes in the selective autophagy pathways does not depend on their role in DNA repair.”
In addition, the autophagy function may partly explain why patients with FA are highly susceptible to infection and cancer, Dr Levine said.
While further research is needed to understand how these findings may be used to treat disease, the investigators said they have identified a novel avenue for developing potential therapies for FA and cancer patients.
“I believe the clearest therapeutic possibilities to come from our study results are the development of new FA agents that target the inflammasome and production of interleukin 1 beta (IL-1β), a pro-inflammatory cytokine,” Dr Sumpter said.
“Clinically, IL-1β signaling has been targeted with FDA-approved drugs very successfully in several auto-inflammatory diseases that involve excessive inflammasome activation. Our results suggest that FA patients may also benefit from these therapies.” ![]()

Image by Sarah Pfau
Research published in Cell has revealed a new function of genes in the Fanconi anemia (FA) pathway, and investigators believe this finding could have
implications for the treatment of FA and related disorders.
The team found that FA genes are required for selective autophagy.
In particular, the FANCC gene plays a key role in 2 types of selective autophagy: virophagy (the removal of viruses inside the cell) and mitophagy (the removal of mitochondria).
Experiments in mice showed that genetic deletion of FANCC blocks virophagy and increases the animals’ susceptibility to lethal viral encephalitis.
The investigators also found that FANCC protein is required for the clearance of damaged mitochondria and decreases the production of mitochondrial reactive oxygen species and inflammasome activation.
And other genes in the FA pathway are required for mitophagy as well—FANCA, FANCF, FANCL, FANCD2, BRCA1, and BRCA2.
“There’s increasing evidence that the failure of cells to appropriately clear damaged mitochondria leads to abnormal activation of the inflammasome—a process that is emerging as an important contributor to many different diseases,” said study author Beth Levine, MD, of UT Southwestern Medical Center in Dallas, Texas.
“The finding that FA genes function in clearing mitochondria and decreasing inflammasome activation provides a potential new inflammasome-targeted avenue of therapy for patients with diseases related to mutations in the FA genes.”
FA pathway genes were already known to play a role in DNA repair. The investigators said this new link to autophagy opens up unexplored horizons for understanding the function of these genes in human health and disease.
“Our findings suggest a novel mechanism by which mutations in FA genes may lead to the clinical manifestations in patients with FA and to cancers in patients with mutations in FA genes,” said study author Rhea Sumpter, MD, PhD, of UT Southwestern Medical Center.
“We’ve shown that this new function of the FA genes in the selective autophagy pathways does not depend on their role in DNA repair.”
In addition, the autophagy function may partly explain why patients with FA are highly susceptible to infection and cancer, Dr Levine said.
While further research is needed to understand how these findings may be used to treat disease, the investigators said they have identified a novel avenue for developing potential therapies for FA and cancer patients.
“I believe the clearest therapeutic possibilities to come from our study results are the development of new FA agents that target the inflammasome and production of interleukin 1 beta (IL-1β), a pro-inflammatory cytokine,” Dr Sumpter said.
“Clinically, IL-1β signaling has been targeted with FDA-approved drugs very successfully in several auto-inflammatory diseases that involve excessive inflammasome activation. Our results suggest that FA patients may also benefit from these therapies.” ![]()
Medical errors among leading causes of death in US

while another looks on
Photo courtesy of NCI
In recent years, medical errors may have become one of the top causes of death in the US, according to a study published in The BMJ.
Investigators analyzed medical death rate data over an 8-year period and calculated that more than 250,000 deaths per year may be due to medical error.
That figure surpasses the US Centers for Disease Control and Prevention’s (CDC) third leading cause of death—respiratory disease, which kills close to 150,000 people per year.
The investigators said the CDC’s way of collecting national health statistics fails to classify medical errors separately on the death certificate. So the team is advocating for updated criteria for classifying deaths.
“Incidence rates for deaths directly attributable to medical care gone awry haven’t been recognized in any standardized method for collecting national statistics,” said study author Martin Makary, MD, of the Johns Hopkins University School of Medicine in Baltimore, Maryland.
“The medical coding system was designed to maximize billing for physician services, not to collect national health statistics, as it is currently being used.”
Dr Makary noted that, in 1949, the US adopted an international form that used International Classification of Diseases (ICD) billing codes to tally causes of death.
“At that time, it was under-recognized that diagnostic errors, medical mistakes, and the absence of safety nets could result in someone’s death, and because of that, medical errors were unintentionally excluded from national health statistics,” Dr Makary said.
He pointed out that, since that time, national mortality statistics have been tabulated using billing codes, which don’t have a built-in way to recognize incidence rates of mortality due to medical care gone wrong.
For the current study, Dr Makary and Michael Daniel, also of Johns Hopkins, examined 4 separate studies that analyzed medical death rate data from 2000 to 2008.
Then, using hospital admission rates from 2013, the investigators extrapolated that, based on a total of 35,416,020 hospitalizations, 251,454 deaths stemmed from medical error. This translates to 9.5% of all deaths each year in the US.
According to the CDC, in 2013, 611,105 people died of heart disease, 584,881 died of cancer, and 149,205 died of chronic respiratory disease.
These were the top 3 causes of death in the US. The newly calculated figure for medical errors puts this cause of death behind cancer but ahead of respiratory disease.
“Top-ranked causes of death as reported by the CDC inform our country’s research funding and public health priorities,” Dr Makary said. “Right now, cancer and heart disease get a ton of attention, but since medical errors don’t appear on the list, the problem doesn’t get the funding and attention it deserves.”
The investigators said most medical errors aren’t due to inherently bad doctors, and reporting these errors shouldn’t be addressed by punishment or legal action.
Rather, the pair believes that most errors represent systemic problems, including poorly coordinated care, fragmented insurance networks, the absence or underuse of safety nets, and other protocols, in addition to unwarranted variation in physician practice patterns that lack accountability.
“Unwarranted variation is endemic in healthcare,” Dr Makary said. “Developing consensus protocols that streamline the delivery of medicine and reduce variability can improve quality and lower costs in healthcare. More research on preventing medical errors from occurring is needed to address the problem.” ![]()

while another looks on
Photo courtesy of NCI
In recent years, medical errors may have become one of the top causes of death in the US, according to a study published in The BMJ.
Investigators analyzed medical death rate data over an 8-year period and calculated that more than 250,000 deaths per year may be due to medical error.
That figure surpasses the US Centers for Disease Control and Prevention’s (CDC) third leading cause of death—respiratory disease, which kills close to 150,000 people per year.
The investigators said the CDC’s way of collecting national health statistics fails to classify medical errors separately on the death certificate. So the team is advocating for updated criteria for classifying deaths.
“Incidence rates for deaths directly attributable to medical care gone awry haven’t been recognized in any standardized method for collecting national statistics,” said study author Martin Makary, MD, of the Johns Hopkins University School of Medicine in Baltimore, Maryland.
“The medical coding system was designed to maximize billing for physician services, not to collect national health statistics, as it is currently being used.”
Dr Makary noted that, in 1949, the US adopted an international form that used International Classification of Diseases (ICD) billing codes to tally causes of death.
“At that time, it was under-recognized that diagnostic errors, medical mistakes, and the absence of safety nets could result in someone’s death, and because of that, medical errors were unintentionally excluded from national health statistics,” Dr Makary said.
He pointed out that, since that time, national mortality statistics have been tabulated using billing codes, which don’t have a built-in way to recognize incidence rates of mortality due to medical care gone wrong.
For the current study, Dr Makary and Michael Daniel, also of Johns Hopkins, examined 4 separate studies that analyzed medical death rate data from 2000 to 2008.
Then, using hospital admission rates from 2013, the investigators extrapolated that, based on a total of 35,416,020 hospitalizations, 251,454 deaths stemmed from medical error. This translates to 9.5% of all deaths each year in the US.
According to the CDC, in 2013, 611,105 people died of heart disease, 584,881 died of cancer, and 149,205 died of chronic respiratory disease.
These were the top 3 causes of death in the US. The newly calculated figure for medical errors puts this cause of death behind cancer but ahead of respiratory disease.
“Top-ranked causes of death as reported by the CDC inform our country’s research funding and public health priorities,” Dr Makary said. “Right now, cancer and heart disease get a ton of attention, but since medical errors don’t appear on the list, the problem doesn’t get the funding and attention it deserves.”
The investigators said most medical errors aren’t due to inherently bad doctors, and reporting these errors shouldn’t be addressed by punishment or legal action.
Rather, the pair believes that most errors represent systemic problems, including poorly coordinated care, fragmented insurance networks, the absence or underuse of safety nets, and other protocols, in addition to unwarranted variation in physician practice patterns that lack accountability.
“Unwarranted variation is endemic in healthcare,” Dr Makary said. “Developing consensus protocols that streamline the delivery of medicine and reduce variability can improve quality and lower costs in healthcare. More research on preventing medical errors from occurring is needed to address the problem.” ![]()

while another looks on
Photo courtesy of NCI
In recent years, medical errors may have become one of the top causes of death in the US, according to a study published in The BMJ.
Investigators analyzed medical death rate data over an 8-year period and calculated that more than 250,000 deaths per year may be due to medical error.
That figure surpasses the US Centers for Disease Control and Prevention’s (CDC) third leading cause of death—respiratory disease, which kills close to 150,000 people per year.
The investigators said the CDC’s way of collecting national health statistics fails to classify medical errors separately on the death certificate. So the team is advocating for updated criteria for classifying deaths.
“Incidence rates for deaths directly attributable to medical care gone awry haven’t been recognized in any standardized method for collecting national statistics,” said study author Martin Makary, MD, of the Johns Hopkins University School of Medicine in Baltimore, Maryland.
“The medical coding system was designed to maximize billing for physician services, not to collect national health statistics, as it is currently being used.”
Dr Makary noted that, in 1949, the US adopted an international form that used International Classification of Diseases (ICD) billing codes to tally causes of death.
“At that time, it was under-recognized that diagnostic errors, medical mistakes, and the absence of safety nets could result in someone’s death, and because of that, medical errors were unintentionally excluded from national health statistics,” Dr Makary said.
He pointed out that, since that time, national mortality statistics have been tabulated using billing codes, which don’t have a built-in way to recognize incidence rates of mortality due to medical care gone wrong.
For the current study, Dr Makary and Michael Daniel, also of Johns Hopkins, examined 4 separate studies that analyzed medical death rate data from 2000 to 2008.
Then, using hospital admission rates from 2013, the investigators extrapolated that, based on a total of 35,416,020 hospitalizations, 251,454 deaths stemmed from medical error. This translates to 9.5% of all deaths each year in the US.
According to the CDC, in 2013, 611,105 people died of heart disease, 584,881 died of cancer, and 149,205 died of chronic respiratory disease.
These were the top 3 causes of death in the US. The newly calculated figure for medical errors puts this cause of death behind cancer but ahead of respiratory disease.
“Top-ranked causes of death as reported by the CDC inform our country’s research funding and public health priorities,” Dr Makary said. “Right now, cancer and heart disease get a ton of attention, but since medical errors don’t appear on the list, the problem doesn’t get the funding and attention it deserves.”
The investigators said most medical errors aren’t due to inherently bad doctors, and reporting these errors shouldn’t be addressed by punishment or legal action.
Rather, the pair believes that most errors represent systemic problems, including poorly coordinated care, fragmented insurance networks, the absence or underuse of safety nets, and other protocols, in addition to unwarranted variation in physician practice patterns that lack accountability.
“Unwarranted variation is endemic in healthcare,” Dr Makary said. “Developing consensus protocols that streamline the delivery of medicine and reduce variability can improve quality and lower costs in healthcare. More research on preventing medical errors from occurring is needed to address the problem.”
CAR T-cell therapy granted orphan designation

The US Food and Drug Administration (FDA) has granted orphan drug designation for the chimeric antigen receptor (CAR) T-cell therapy KTE-C19 as a treatment for several hematologic malignancies.
This includes primary mediastinal B-cell lymphoma (PMBCL), mantle cell lymphoma (MCL), follicular lymphoma (FL), acute lymphoblastic leukemia (ALL), and chronic lymphocytic leukemia (CLL).
KTE-C19 previously received orphan designation from the FDA for the treatment of diffuse large B-cell lymphoma (DLBCL).
The FDA grants orphan designation to drugs and biologics intended to treat, diagnose, or prevent diseases/disorders that affect fewer than 200,000 people in the US.
The designation provides incentives for sponsors to develop products for rare diseases. This may include tax credits toward the cost of clinical trials, prescription drug user fee waivers, and 7 years of market exclusivity.
KTE-C19 also has breakthrough therapy designation from the FDA as a treatment for DLBCL, PMBCL, and transformed FL.
About KTE-C19
KTE-C19 is an investigational therapy in which a patient’s T cells are genetically modified to express a CAR designed to target CD19. The product is being developed by Kite Pharma, Inc.
In a study published in the Journal of Clinical Oncology, researchers evaluated KTE-C19 in 15 patients with advanced B-cell malignancies.
The patients received a conditioning regimen of cyclophosphamide and fludarabine, followed 1 day later by a single infusion of KTE-C19. The researchers noted that the conditioning regimen is known to be active against B-cell malignancies and could have made a direct contribution to patient responses.
Thirteen patients were evaluable for response. The overall response rate was 92%. Eight patients achieved a complete response (CR), and 4 had a partial response (PR).
Of the 7 patients with DLBCL, 4 achieved a CR, 2 achieved a PR, and 1 had stable disease. Three of the CRs were ongoing at the time of publication, with the duration ranging from 9 months to 22 months.
Of the 4 patients with CLL, 3 had a CR, and 1 had a PR. All 3 CRs were ongoing at the time of publication, with the duration ranging from 14 months to 23 months.
Among the 2 patients with indolent lymphomas, 1 achieved a CR, and 1 had a PR. The duration of the CR was 11 months at the time of publication.
KTE-C19 elicited a number of adverse events, including fever, hypotension, delirium, and other neurologic toxicities. All but 2 patients experienced grade 3/4 adverse events.
Three patients developed unexpected neurologic abnormalities. One patient experienced aphasia and right-sided facial paresis. One patient developed aphasia, confusion, and severe, generalized myoclonus. And 1 patient had aphasia, confusion, hemifacial spasms, apraxia, and gait disturbances.
KTE-C19 is currently under investigation in a phase 2 trial of refractory DLBCL, PMBCL, and transformed FL (ZUMA-1), a phase 2 trial of relapsed/refractory MCL (ZUMA-2), a phase 1/2 trial of relapsed/refractory adult ALL (ZUMA-3), and a phase 1/2 trial of relapsed/refractory pediatric ALL (ZUMA-4).
Results from ZUMA-1 were recently presented at the 2016 AACR Annual Meeting (abstract CT135).

The US Food and Drug Administration (FDA) has granted orphan drug designation for the chimeric antigen receptor (CAR) T-cell therapy KTE-C19 as a treatment for several hematologic malignancies.
This includes primary mediastinal B-cell lymphoma (PMBCL), mantle cell lymphoma (MCL), follicular lymphoma (FL), acute lymphoblastic leukemia (ALL), and chronic lymphocytic leukemia (CLL).
KTE-C19 previously received orphan designation from the FDA for the treatment of diffuse large B-cell lymphoma (DLBCL).
The FDA grants orphan designation to drugs and biologics intended to treat, diagnose, or prevent diseases/disorders that affect fewer than 200,000 people in the US.
The designation provides incentives for sponsors to develop products for rare diseases. This may include tax credits toward the cost of clinical trials, prescription drug user fee waivers, and 7 years of market exclusivity.
KTE-C19 also has breakthrough therapy designation from the FDA as a treatment for DLBCL, PMBCL, and transformed FL.
About KTE-C19
KTE-C19 is an investigational therapy in which a patient’s T cells are genetically modified to express a CAR designed to target CD19. The product is being developed by Kite Pharma, Inc.
In a study published in the Journal of Clinical Oncology, researchers evaluated KTE-C19 in 15 patients with advanced B-cell malignancies.
The patients received a conditioning regimen of cyclophosphamide and fludarabine, followed 1 day later by a single infusion of KTE-C19. The researchers noted that the conditioning regimen is known to be active against B-cell malignancies and could have made a direct contribution to patient responses.
Thirteen patients were evaluable for response. The overall response rate was 92%. Eight patients achieved a complete response (CR), and 4 had a partial response (PR).
Of the 7 patients with DLBCL, 4 achieved a CR, 2 achieved a PR, and 1 had stable disease. Three of the CRs were ongoing at the time of publication, with the duration ranging from 9 months to 22 months.
Of the 4 patients with CLL, 3 had a CR, and 1 had a PR. All 3 CRs were ongoing at the time of publication, with the duration ranging from 14 months to 23 months.
Among the 2 patients with indolent lymphomas, 1 achieved a CR, and 1 had a PR. The duration of the CR was 11 months at the time of publication.
KTE-C19 elicited a number of adverse events, including fever, hypotension, delirium, and other neurologic toxicities. All but 2 patients experienced grade 3/4 adverse events.
Three patients developed unexpected neurologic abnormalities. One patient experienced aphasia and right-sided facial paresis. One patient developed aphasia, confusion, and severe, generalized myoclonus. And 1 patient had aphasia, confusion, hemifacial spasms, apraxia, and gait disturbances.
KTE-C19 is currently under investigation in a phase 2 trial of refractory DLBCL, PMBCL, and transformed FL (ZUMA-1), a phase 2 trial of relapsed/refractory MCL (ZUMA-2), a phase 1/2 trial of relapsed/refractory adult ALL (ZUMA-3), and a phase 1/2 trial of relapsed/refractory pediatric ALL (ZUMA-4).
Results from ZUMA-1 were recently presented at the 2016 AACR Annual Meeting (abstract CT135).

The US Food and Drug Administration (FDA) has granted orphan drug designation for the chimeric antigen receptor (CAR) T-cell therapy KTE-C19 as a treatment for several hematologic malignancies.
This includes primary mediastinal B-cell lymphoma (PMBCL), mantle cell lymphoma (MCL), follicular lymphoma (FL), acute lymphoblastic leukemia (ALL), and chronic lymphocytic leukemia (CLL).
KTE-C19 previously received orphan designation from the FDA for the treatment of diffuse large B-cell lymphoma (DLBCL).
The FDA grants orphan designation to drugs and biologics intended to treat, diagnose, or prevent diseases/disorders that affect fewer than 200,000 people in the US.
The designation provides incentives for sponsors to develop products for rare diseases. This may include tax credits toward the cost of clinical trials, prescription drug user fee waivers, and 7 years of market exclusivity.
KTE-C19 also has breakthrough therapy designation from the FDA as a treatment for DLBCL, PMBCL, and transformed FL.
About KTE-C19
KTE-C19 is an investigational therapy in which a patient’s T cells are genetically modified to express a CAR designed to target CD19. The product is being developed by Kite Pharma, Inc.
In a study published in the Journal of Clinical Oncology, researchers evaluated KTE-C19 in 15 patients with advanced B-cell malignancies.
The patients received a conditioning regimen of cyclophosphamide and fludarabine, followed 1 day later by a single infusion of KTE-C19. The researchers noted that the conditioning regimen is known to be active against B-cell malignancies and could have made a direct contribution to patient responses.
Thirteen patients were evaluable for response. The overall response rate was 92%. Eight patients achieved a complete response (CR), and 4 had a partial response (PR).
Of the 7 patients with DLBCL, 4 achieved a CR, 2 achieved a PR, and 1 had stable disease. Three of the CRs were ongoing at the time of publication, with the duration ranging from 9 months to 22 months.
Of the 4 patients with CLL, 3 had a CR, and 1 had a PR. All 3 CRs were ongoing at the time of publication, with the duration ranging from 14 months to 23 months.
Among the 2 patients with indolent lymphomas, 1 achieved a CR, and 1 had a PR. The duration of the CR was 11 months at the time of publication.
KTE-C19 elicited a number of adverse events, including fever, hypotension, delirium, and other neurologic toxicities. All but 2 patients experienced grade 3/4 adverse events.
Three patients developed unexpected neurologic abnormalities. One patient experienced aphasia and right-sided facial paresis. One patient developed aphasia, confusion, and severe, generalized myoclonus. And 1 patient had aphasia, confusion, hemifacial spasms, apraxia, and gait disturbances.
KTE-C19 is currently under investigation in a phase 2 trial of refractory DLBCL, PMBCL, and transformed FL (ZUMA-1), a phase 2 trial of relapsed/refractory MCL (ZUMA-2), a phase 1/2 trial of relapsed/refractory adult ALL (ZUMA-3), and a phase 1/2 trial of relapsed/refractory pediatric ALL (ZUMA-4).
Results from ZUMA-1 were recently presented at the 2016 AACR Annual Meeting (abstract CT135).
Therapy may reduce memory problems related to chemo

patient and her father
Photo by Rhoda Baer
A type of cognitive behavioral therapy may help prevent some of the long-term memory issues caused by chemotherapy, according to research published in Cancer.
The therapy is called “Memory and Attention Adaptation Training” (MAAT).
It’s designed to help cancer survivors increase awareness of situations where memory problems can arise and develop skills to either prevent memory failure or compensate for memory dysfunction.
MAAT was developed by Robert Ferguson, PhD, of the University of Pittsburgh Cancer Institute in Pennsylvania, and his colleagues.
The researchers tested MAAT in a small, randomized study of 47 Caucasian breast cancer survivors who were an average of 4 years post-chemotherapy.
The patients were assigned to 8 visits of MAAT (30 to 45 minutes each visit) or supportive talk therapy for an identical time span. The intent of the supportive therapy was to control for the simple effects of interacting with a supportive clinician, or “behavioral placebo.”
Both treatments were delivered over a videoconference network between health centers to minimize patient travel.
All participants completed questionnaires assessing perceived memory difficulty and anxiety about memory problems. They were also tested over the phone with neuropsychological tests of verbal memory and processing speed, or the ability to automatically and fluently perform relatively easy cognitive tasks.
Participants were evaluated again after the 8 MAAT and supportive therapy videoconference visits, as well as 2 months after the conclusion of these therapies.
Compared with participants who received supportive therapy, MAAT participants reported significantly fewer memory problems (P=0.02) and improved processing speed post-treatment (P=0.03).
MAAT participants also reported less anxiety about cognitive problems compared with supportive therapy participants 2 months after MAAT concluded, but this was not a statistically significant finding.
“This is what we believe is the first randomized study with an active control condition that demonstrates improvement in cognitive symptoms in breast cancer survivors with long-term memory complaints,” Dr Ferguson said. “MAAT participants reported reduced anxiety and high satisfaction with this cognitive behavioral, non-drug approach.”
“Because treatment was delivered over videoconference device, this study demonstrates MAAT can be delivered electronically and survivors can reduce or eliminate travel to a cancer center. This can improve access to survivorship care.”
Dr Ferguson also noted that more research on MAAT is needed using a larger number of individuals with varied ethnic and cultural backgrounds and multiple clinicians delivering treatment.

patient and her father
Photo by Rhoda Baer
A type of cognitive behavioral therapy may help prevent some of the long-term memory issues caused by chemotherapy, according to research published in Cancer.
The therapy is called “Memory and Attention Adaptation Training” (MAAT).
It’s designed to help cancer survivors increase awareness of situations where memory problems can arise and develop skills to either prevent memory failure or compensate for memory dysfunction.
MAAT was developed by Robert Ferguson, PhD, of the University of Pittsburgh Cancer Institute in Pennsylvania, and his colleagues.
The researchers tested MAAT in a small, randomized study of 47 Caucasian breast cancer survivors who were an average of 4 years post-chemotherapy.
The patients were assigned to 8 visits of MAAT (30 to 45 minutes each visit) or supportive talk therapy for an identical time span. The intent of the supportive therapy was to control for the simple effects of interacting with a supportive clinician, or “behavioral placebo.”
Both treatments were delivered over a videoconference network between health centers to minimize patient travel.
All participants completed questionnaires assessing perceived memory difficulty and anxiety about memory problems. They were also tested over the phone with neuropsychological tests of verbal memory and processing speed, or the ability to automatically and fluently perform relatively easy cognitive tasks.
Participants were evaluated again after the 8 MAAT and supportive therapy videoconference visits, as well as 2 months after the conclusion of these therapies.
Compared with participants who received supportive therapy, MAAT participants reported significantly fewer memory problems (P=0.02) and improved processing speed post-treatment (P=0.03).
MAAT participants also reported less anxiety about cognitive problems compared with supportive therapy participants 2 months after MAAT concluded, but this was not a statistically significant finding.
“This is what we believe is the first randomized study with an active control condition that demonstrates improvement in cognitive symptoms in breast cancer survivors with long-term memory complaints,” Dr Ferguson said. “MAAT participants reported reduced anxiety and high satisfaction with this cognitive behavioral, non-drug approach.”
“Because treatment was delivered over videoconference device, this study demonstrates MAAT can be delivered electronically and survivors can reduce or eliminate travel to a cancer center. This can improve access to survivorship care.”
Dr Ferguson also noted that more research on MAAT is needed using a larger number of individuals with varied ethnic and cultural backgrounds and multiple clinicians delivering treatment.

patient and her father
Photo by Rhoda Baer
A type of cognitive behavioral therapy may help prevent some of the long-term memory issues caused by chemotherapy, according to research published in Cancer.
The therapy is called “Memory and Attention Adaptation Training” (MAAT).
It’s designed to help cancer survivors increase awareness of situations where memory problems can arise and develop skills to either prevent memory failure or compensate for memory dysfunction.
MAAT was developed by Robert Ferguson, PhD, of the University of Pittsburgh Cancer Institute in Pennsylvania, and his colleagues.
The researchers tested MAAT in a small, randomized study of 47 Caucasian breast cancer survivors who were an average of 4 years post-chemotherapy.
The patients were assigned to 8 visits of MAAT (30 to 45 minutes each visit) or supportive talk therapy for an identical time span. The intent of the supportive therapy was to control for the simple effects of interacting with a supportive clinician, or “behavioral placebo.”
Both treatments were delivered over a videoconference network between health centers to minimize patient travel.
All participants completed questionnaires assessing perceived memory difficulty and anxiety about memory problems. They were also tested over the phone with neuropsychological tests of verbal memory and processing speed, or the ability to automatically and fluently perform relatively easy cognitive tasks.
Participants were evaluated again after the 8 MAAT and supportive therapy videoconference visits, as well as 2 months after the conclusion of these therapies.
Compared with participants who received supportive therapy, MAAT participants reported significantly fewer memory problems (P=0.02) and improved processing speed post-treatment (P=0.03).
MAAT participants also reported less anxiety about cognitive problems compared with supportive therapy participants 2 months after MAAT concluded, but this was not a statistically significant finding.
“This is what we believe is the first randomized study with an active control condition that demonstrates improvement in cognitive symptoms in breast cancer survivors with long-term memory complaints,” Dr Ferguson said. “MAAT participants reported reduced anxiety and high satisfaction with this cognitive behavioral, non-drug approach.”
“Because treatment was delivered over videoconference device, this study demonstrates MAAT can be delivered electronically and survivors can reduce or eliminate travel to a cancer center. This can improve access to survivorship care.”
Dr Ferguson also noted that more research on MAAT is needed using a larger number of individuals with varied ethnic and cultural backgrounds and multiple clinicians delivering treatment.
Current cancer drug discovery method flawed, team says

Image courtesy of PNAS
The primary method used to test compounds for anticancer activity in vitro may produce inaccurate results, according to researchers.
Therefore, they have developed a new metric to evaluate a compound’s effect on cell proliferation—the drug-induced proliferation (DIP) rate.
They believe this metric, described in Nature Methods, overcomes the time-dependent bias of traditional proliferation assays.
“More than 90% of candidate cancer drugs fail in late-stage clinical trials, costing hundreds of millions of dollars,” said study author Vito Quaranta, MD, of Vanderbilt University School of Medicine in Nashville, Tennessee.
“The flawed in vitro drug discovery metric may not be the only responsible factor, but it may be worth pursuing an estimate of its impact.”
For more than 30 years, scientists have evaluated the ability of a compound to kill cells by adding the compound and counting how many cells are alive after 72 hours.
However, these proliferation assays, which measure cell number at a single time point, don’t take into account the bias introduced by exponential cell proliferation, even in the presence of the drug, said study author Darren Tyson, PhD, of Vanderbilt University School of Medicine.
“Cells are not uniform,” added Dr Quaranta. “They all proliferate exponentially but at different rates. At 72 hours, some cells will have doubled 3 times, and others will not have doubled at all.”
In addition, he noted, drugs don’t all behave the same way on every cell line. For example, a drug might have an immediate effect on one cell line and a delayed effect on another.
Therefore, he and his colleagues used a systems biology approach to demonstrate the time-dependent bias in static proliferation assays and to develop the time-independent DIP rate metric.
The researchers evaluated the responses of 4 different melanoma cell lines to the drug vemurafenib, currently used to treat melanoma, with the standard metric and with the DIP rate.
In one cell line, the team found a stark disagreement between the two metrics.
“The static metric says that the cell line is very sensitive to vemurafenib,” said Leonard Harris, PhD, of Vanderbilt University School of Medicine.
“However, our analysis shows this is not the case. A brief period of drug sensitivity, quickly followed by rebound, fools the static metric but not the DIP rate.”
The findings “suggest we should expect melanoma tumors treated with this drug to come back, and that’s what has happened, puzzling investigators,” Dr Quaranta said. “DIP rate analyses may help solve this conundrum, leading to better treatment strategies.”
These findings have particular importance in light of recent international efforts to generate data sets that include the responses of “thousands of cell lines to hundreds of compounds,” Dr Quaranta said.
The Cancer Cell Line Encyclopedia (CCLE) and Genomics of Drug Sensitivity in Cancer (GDSC) databases include drug response data along with genomic and proteomic data that detail each cell line’s molecular makeup.
“The idea is to look for statistical correlations—these particular cell lines with this particular makeup are sensitive to these types of compounds—to use these large databases as discovery tools for new therapeutic targets in cancer,” Dr Quaranta said. “If the metric by which you’ve evaluated the drug sensitivity of the cells is wrong, your statistical correlations are basically no good.”

Image courtesy of PNAS
The primary method used to test compounds for anticancer activity in vitro may produce inaccurate results, according to researchers.
Therefore, they have developed a new metric to evaluate a compound’s effect on cell proliferation—the drug-induced proliferation (DIP) rate.
They believe this metric, described in Nature Methods, overcomes the time-dependent bias of traditional proliferation assays.
“More than 90% of candidate cancer drugs fail in late-stage clinical trials, costing hundreds of millions of dollars,” said study author Vito Quaranta, MD, of Vanderbilt University School of Medicine in Nashville, Tennessee.
“The flawed in vitro drug discovery metric may not be the only responsible factor, but it may be worth pursuing an estimate of its impact.”
For more than 30 years, scientists have evaluated the ability of a compound to kill cells by adding the compound and counting how many cells are alive after 72 hours.
However, these proliferation assays, which measure cell number at a single time point, don’t take into account the bias introduced by exponential cell proliferation, even in the presence of the drug, said study author Darren Tyson, PhD, of Vanderbilt University School of Medicine.
“Cells are not uniform,” added Dr Quaranta. “They all proliferate exponentially but at different rates. At 72 hours, some cells will have doubled 3 times, and others will not have doubled at all.”
In addition, he noted, drugs don’t all behave the same way on every cell line. For example, a drug might have an immediate effect on one cell line and a delayed effect on another.
Therefore, he and his colleagues used a systems biology approach to demonstrate the time-dependent bias in static proliferation assays and to develop the time-independent DIP rate metric.
The researchers evaluated the responses of 4 different melanoma cell lines to the drug vemurafenib, currently used to treat melanoma, with the standard metric and with the DIP rate.
In one cell line, the team found a stark disagreement between the two metrics.
“The static metric says that the cell line is very sensitive to vemurafenib,” said Leonard Harris, PhD, of Vanderbilt University School of Medicine.
“However, our analysis shows this is not the case. A brief period of drug sensitivity, quickly followed by rebound, fools the static metric but not the DIP rate.”
The findings “suggest we should expect melanoma tumors treated with this drug to come back, and that’s what has happened, puzzling investigators,” Dr Quaranta said. “DIP rate analyses may help solve this conundrum, leading to better treatment strategies.”
These findings have particular importance in light of recent international efforts to generate data sets that include the responses of “thousands of cell lines to hundreds of compounds,” Dr Quaranta said.
The Cancer Cell Line Encyclopedia (CCLE) and Genomics of Drug Sensitivity in Cancer (GDSC) databases include drug response data along with genomic and proteomic data that detail each cell line’s molecular makeup.
“The idea is to look for statistical correlations—these particular cell lines with this particular makeup are sensitive to these types of compounds—to use these large databases as discovery tools for new therapeutic targets in cancer,” Dr Quaranta said. “If the metric by which you’ve evaluated the drug sensitivity of the cells is wrong, your statistical correlations are basically no good.”

Image courtesy of PNAS
The primary method used to test compounds for anticancer activity in vitro may produce inaccurate results, according to researchers.
Therefore, they have developed a new metric to evaluate a compound’s effect on cell proliferation—the drug-induced proliferation (DIP) rate.
They believe this metric, described in Nature Methods, overcomes the time-dependent bias of traditional proliferation assays.
“More than 90% of candidate cancer drugs fail in late-stage clinical trials, costing hundreds of millions of dollars,” said study author Vito Quaranta, MD, of Vanderbilt University School of Medicine in Nashville, Tennessee.
“The flawed in vitro drug discovery metric may not be the only responsible factor, but it may be worth pursuing an estimate of its impact.”
For more than 30 years, scientists have evaluated the ability of a compound to kill cells by adding the compound and counting how many cells are alive after 72 hours.
However, these proliferation assays, which measure cell number at a single time point, don’t take into account the bias introduced by exponential cell proliferation, even in the presence of the drug, said study author Darren Tyson, PhD, of Vanderbilt University School of Medicine.
“Cells are not uniform,” added Dr Quaranta. “They all proliferate exponentially but at different rates. At 72 hours, some cells will have doubled 3 times, and others will not have doubled at all.”
In addition, he noted, drugs don’t all behave the same way on every cell line. For example, a drug might have an immediate effect on one cell line and a delayed effect on another.
Therefore, he and his colleagues used a systems biology approach to demonstrate the time-dependent bias in static proliferation assays and to develop the time-independent DIP rate metric.
The researchers evaluated the responses of 4 different melanoma cell lines to the drug vemurafenib, currently used to treat melanoma, with the standard metric and with the DIP rate.
In one cell line, the team found a stark disagreement between the two metrics.
“The static metric says that the cell line is very sensitive to vemurafenib,” said Leonard Harris, PhD, of Vanderbilt University School of Medicine.
“However, our analysis shows this is not the case. A brief period of drug sensitivity, quickly followed by rebound, fools the static metric but not the DIP rate.”
The findings “suggest we should expect melanoma tumors treated with this drug to come back, and that’s what has happened, puzzling investigators,” Dr Quaranta said. “DIP rate analyses may help solve this conundrum, leading to better treatment strategies.”
These findings have particular importance in light of recent international efforts to generate data sets that include the responses of “thousands of cell lines to hundreds of compounds,” Dr Quaranta said.
The Cancer Cell Line Encyclopedia (CCLE) and Genomics of Drug Sensitivity in Cancer (GDSC) databases include drug response data along with genomic and proteomic data that detail each cell line’s molecular makeup.
“The idea is to look for statistical correlations—these particular cell lines with this particular makeup are sensitive to these types of compounds—to use these large databases as discovery tools for new therapeutic targets in cancer,” Dr Quaranta said. “If the metric by which you’ve evaluated the drug sensitivity of the cells is wrong, your statistical correlations are basically no good.”