User login
Targeting a protein to prevent malignancy

Photo by Aaron Logan
New research suggests hematologic malignancies driven by MYC might be prevented by lowering levels of another protein, MCL-1.
“Our colleagues had previously discovered that reducing the activity of MCL-1 is a promising strategy to treat malignant MYC-driven cancers,” said Stephanie Grabow, PhD, of the Walter and Eliza Hall Institute of Medical Research in Parkville, Victoria, Australia.
“We have now shown that the same approach might be able to prevent those cancers from forming in the first place.”
Dr Grabow and her colleagues described this work in Cell Reports.
Previous research indicated that expression from both MCL-1 alleles is essential for the survival of hematopoietic stem and progenitor cells during stress-induced repopulation of the hematopoietic system.
So, with this study, Dr Grabow and her colleagues set out to determine whether reducing MCL-1 protein levels might hinder the development of hematologic malignancies.
In experiments with mice, the investigators found that loss of one MCL-1 allele significantly delayed the development of MYC-driven lymphoma and reduced MYC-driven accumulation of pre-leukemic cancer-initiating cells.
However, loss of one p53 allele accelerated MYC-driven lymphomagenesis even when one MCL-1 allele was deleted. Loss of PUMA accelerated lymphoma development as well, though to a much lesser extent.
Loss of BIM substantially accelerated lymphomagenesis when one MCL-1 allele was deleted, restoring lymphoma-initiating cells and the rate of tumor development.
And loss of one BIM allele overrode the survival defect observed in pre-leukemic Eμ-Myc B-cell progenitors when one MCL-1 allele was deleted.
The investigators noted that loss of one MCL-1 allele did not noticeably impair the survival of normal B lymphoid cells even though it greatly diminished the survival of MYC-overexpressing B-cell progenitors.
“No one had realized just how vulnerable cells undergoing cancerous changes are to a relatively minor reduction in the levels of MCL-1,” Dr Grabow said.
“We found that MCL-1 is critical for keeping developing cancer cells alive through the stressful events that cause the transformation of a healthy cell into a cancerous cell. This result is particularly exciting because MCL-1 inhibitors are already in development as anticancer drugs.”
Study investigator Brandon Aubrey, MBBS, also of the Walter and Eliza Hall Institute, said this research could inform future strategies to prevent cancer.
“Early treatment or even cancer prevention are likely to be a more effective way to fight cancer than treating an established cancer after it has already formed and made a person sick,” he said. ”Our research has suggested that dependency on MCL-1 could be a key vulnerability of many developing cancers.”
“In the future, MCL-1 inhibitors might have potential benefit for treating the very early stages of MYC-driven cancers, or we may even be able use these agents to prevent people from getting cancer in the first place.” ![]()

Photo by Aaron Logan
New research suggests hematologic malignancies driven by MYC might be prevented by lowering levels of another protein, MCL-1.
“Our colleagues had previously discovered that reducing the activity of MCL-1 is a promising strategy to treat malignant MYC-driven cancers,” said Stephanie Grabow, PhD, of the Walter and Eliza Hall Institute of Medical Research in Parkville, Victoria, Australia.
“We have now shown that the same approach might be able to prevent those cancers from forming in the first place.”
Dr Grabow and her colleagues described this work in Cell Reports.
Previous research indicated that expression from both MCL-1 alleles is essential for the survival of hematopoietic stem and progenitor cells during stress-induced repopulation of the hematopoietic system.
So, with this study, Dr Grabow and her colleagues set out to determine whether reducing MCL-1 protein levels might hinder the development of hematologic malignancies.
In experiments with mice, the investigators found that loss of one MCL-1 allele significantly delayed the development of MYC-driven lymphoma and reduced MYC-driven accumulation of pre-leukemic cancer-initiating cells.
However, loss of one p53 allele accelerated MYC-driven lymphomagenesis even when one MCL-1 allele was deleted. Loss of PUMA accelerated lymphoma development as well, though to a much lesser extent.
Loss of BIM substantially accelerated lymphomagenesis when one MCL-1 allele was deleted, restoring lymphoma-initiating cells and the rate of tumor development.
And loss of one BIM allele overrode the survival defect observed in pre-leukemic Eμ-Myc B-cell progenitors when one MCL-1 allele was deleted.
The investigators noted that loss of one MCL-1 allele did not noticeably impair the survival of normal B lymphoid cells even though it greatly diminished the survival of MYC-overexpressing B-cell progenitors.
“No one had realized just how vulnerable cells undergoing cancerous changes are to a relatively minor reduction in the levels of MCL-1,” Dr Grabow said.
“We found that MCL-1 is critical for keeping developing cancer cells alive through the stressful events that cause the transformation of a healthy cell into a cancerous cell. This result is particularly exciting because MCL-1 inhibitors are already in development as anticancer drugs.”
Study investigator Brandon Aubrey, MBBS, also of the Walter and Eliza Hall Institute, said this research could inform future strategies to prevent cancer.
“Early treatment or even cancer prevention are likely to be a more effective way to fight cancer than treating an established cancer after it has already formed and made a person sick,” he said. ”Our research has suggested that dependency on MCL-1 could be a key vulnerability of many developing cancers.”
“In the future, MCL-1 inhibitors might have potential benefit for treating the very early stages of MYC-driven cancers, or we may even be able use these agents to prevent people from getting cancer in the first place.” ![]()

Photo by Aaron Logan
New research suggests hematologic malignancies driven by MYC might be prevented by lowering levels of another protein, MCL-1.
“Our colleagues had previously discovered that reducing the activity of MCL-1 is a promising strategy to treat malignant MYC-driven cancers,” said Stephanie Grabow, PhD, of the Walter and Eliza Hall Institute of Medical Research in Parkville, Victoria, Australia.
“We have now shown that the same approach might be able to prevent those cancers from forming in the first place.”
Dr Grabow and her colleagues described this work in Cell Reports.
Previous research indicated that expression from both MCL-1 alleles is essential for the survival of hematopoietic stem and progenitor cells during stress-induced repopulation of the hematopoietic system.
So, with this study, Dr Grabow and her colleagues set out to determine whether reducing MCL-1 protein levels might hinder the development of hematologic malignancies.
In experiments with mice, the investigators found that loss of one MCL-1 allele significantly delayed the development of MYC-driven lymphoma and reduced MYC-driven accumulation of pre-leukemic cancer-initiating cells.
However, loss of one p53 allele accelerated MYC-driven lymphomagenesis even when one MCL-1 allele was deleted. Loss of PUMA accelerated lymphoma development as well, though to a much lesser extent.
Loss of BIM substantially accelerated lymphomagenesis when one MCL-1 allele was deleted, restoring lymphoma-initiating cells and the rate of tumor development.
And loss of one BIM allele overrode the survival defect observed in pre-leukemic Eμ-Myc B-cell progenitors when one MCL-1 allele was deleted.
The investigators noted that loss of one MCL-1 allele did not noticeably impair the survival of normal B lymphoid cells even though it greatly diminished the survival of MYC-overexpressing B-cell progenitors.
“No one had realized just how vulnerable cells undergoing cancerous changes are to a relatively minor reduction in the levels of MCL-1,” Dr Grabow said.
“We found that MCL-1 is critical for keeping developing cancer cells alive through the stressful events that cause the transformation of a healthy cell into a cancerous cell. This result is particularly exciting because MCL-1 inhibitors are already in development as anticancer drugs.”
Study investigator Brandon Aubrey, MBBS, also of the Walter and Eliza Hall Institute, said this research could inform future strategies to prevent cancer.
“Early treatment or even cancer prevention are likely to be a more effective way to fight cancer than treating an established cancer after it has already formed and made a person sick,” he said. ”Our research has suggested that dependency on MCL-1 could be a key vulnerability of many developing cancers.”
“In the future, MCL-1 inhibitors might have potential benefit for treating the very early stages of MYC-driven cancers, or we may even be able use these agents to prevent people from getting cancer in the first place.” ![]()
Study sheds new light on blood clot structure

Photo by Vera Kratochvil
Researchers say they have discovered significant differences between blood clot structure in adults and newborns, a finding that could help us better understand the challenges in addressing post-operative bleeding in neonatal patients.
The researchers also found evidence to suggest the current standard of care for treating post-operative bleeding may pose an increased risk of thrombosis in newborns as compared to adults.
The team reported these findings in Anesthesiology.
“We knew that neonates—infants less than 1 month old—are more likely than adults to suffer from severe bleeding after heart surgery, which poses a variety of health risks,” said study author Ashley Brown, PhD, of the University of North Carolina at Chapel Hill.
“The current standard of care is to give neonatal patients blood products . . . derived from adult blood, but neonatal blood and adult blood aren’t the same. Many of the components involved in clotting in newborns have differing levels of activity, or effectiveness, compared to the same components in adults. Our goal was to better understand how clotting in neonates differs from that in adults so that we can move closer to developing more effective treatment strategies for these infants.”
The researchers’ hypothesis was that fibrinogen from neonates would form clots that are different from those formed by adult fibrinogen, and this proved correct. However, the team was surprised to find that fibrinogen from adults did not integrate well with fibrinogen from neonates.
To test their hypothesis, the researchers took samples of neonate fibrinogen and adult fibrinogen and compared clot formation. The team looked at clots formed solely of adult fibrinogen, clots formed solely of neonate fibrinogen, and clots made from a mixture of the two.
Neonate fibrinogen formed less dense, more fragile clots than adult fibrinogen. Likewise, a mixture of adult and neonate fibrinogen formed clots that were fragile and less dense, even if there was relatively little neonate fibrinogen in the mixture.
The researchers also evaluated how long it took these clots to dissolve. Clots of neonate fibrinogen dissolved about twice as quickly as clots formed from adult fibrinogen.
Clots formed from an adult and neonate fibrinogen mixture dissolved at approximately the same rate as adult-only clots, regardless of the percentage of neonate fibrinogen in the mixture.
“This suggests that using adult fibrinogen in neonatal patients may pose an increased risk of embolism or other adverse thrombotic events,” said study author Nina Guzzetta, MD, of the Emory University School of Medicine in Atlanta, Georgia.
“This work drives home that newborns are not just small adults, and we still have much to learn about clotting in neonates. It also tells us that there is a great deal of room for improvement in the current standard of care for post-operative bleeding in neonates.”
“We are investigating several approaches that may help address this problem, evaluating various modes of action,” Dr Brown added. “It is possible that we can use various external factors that promote clotting to stimulate the fibrinogen in neonates to form a denser clot.”
“We are investigating possible alternatives to help neonates form a better clot after major surgery without having to use adult fibrinogen. For example, we are investigating the use of synthetic platelet-like particles developed by our team to augment hemostasis . . . in blood samples collected from these patients.” ![]()

Photo by Vera Kratochvil
Researchers say they have discovered significant differences between blood clot structure in adults and newborns, a finding that could help us better understand the challenges in addressing post-operative bleeding in neonatal patients.
The researchers also found evidence to suggest the current standard of care for treating post-operative bleeding may pose an increased risk of thrombosis in newborns as compared to adults.
The team reported these findings in Anesthesiology.
“We knew that neonates—infants less than 1 month old—are more likely than adults to suffer from severe bleeding after heart surgery, which poses a variety of health risks,” said study author Ashley Brown, PhD, of the University of North Carolina at Chapel Hill.
“The current standard of care is to give neonatal patients blood products . . . derived from adult blood, but neonatal blood and adult blood aren’t the same. Many of the components involved in clotting in newborns have differing levels of activity, or effectiveness, compared to the same components in adults. Our goal was to better understand how clotting in neonates differs from that in adults so that we can move closer to developing more effective treatment strategies for these infants.”
The researchers’ hypothesis was that fibrinogen from neonates would form clots that are different from those formed by adult fibrinogen, and this proved correct. However, the team was surprised to find that fibrinogen from adults did not integrate well with fibrinogen from neonates.
To test their hypothesis, the researchers took samples of neonate fibrinogen and adult fibrinogen and compared clot formation. The team looked at clots formed solely of adult fibrinogen, clots formed solely of neonate fibrinogen, and clots made from a mixture of the two.
Neonate fibrinogen formed less dense, more fragile clots than adult fibrinogen. Likewise, a mixture of adult and neonate fibrinogen formed clots that were fragile and less dense, even if there was relatively little neonate fibrinogen in the mixture.
The researchers also evaluated how long it took these clots to dissolve. Clots of neonate fibrinogen dissolved about twice as quickly as clots formed from adult fibrinogen.
Clots formed from an adult and neonate fibrinogen mixture dissolved at approximately the same rate as adult-only clots, regardless of the percentage of neonate fibrinogen in the mixture.
“This suggests that using adult fibrinogen in neonatal patients may pose an increased risk of embolism or other adverse thrombotic events,” said study author Nina Guzzetta, MD, of the Emory University School of Medicine in Atlanta, Georgia.
“This work drives home that newborns are not just small adults, and we still have much to learn about clotting in neonates. It also tells us that there is a great deal of room for improvement in the current standard of care for post-operative bleeding in neonates.”
“We are investigating several approaches that may help address this problem, evaluating various modes of action,” Dr Brown added. “It is possible that we can use various external factors that promote clotting to stimulate the fibrinogen in neonates to form a denser clot.”
“We are investigating possible alternatives to help neonates form a better clot after major surgery without having to use adult fibrinogen. For example, we are investigating the use of synthetic platelet-like particles developed by our team to augment hemostasis . . . in blood samples collected from these patients.” ![]()

Photo by Vera Kratochvil
Researchers say they have discovered significant differences between blood clot structure in adults and newborns, a finding that could help us better understand the challenges in addressing post-operative bleeding in neonatal patients.
The researchers also found evidence to suggest the current standard of care for treating post-operative bleeding may pose an increased risk of thrombosis in newborns as compared to adults.
The team reported these findings in Anesthesiology.
“We knew that neonates—infants less than 1 month old—are more likely than adults to suffer from severe bleeding after heart surgery, which poses a variety of health risks,” said study author Ashley Brown, PhD, of the University of North Carolina at Chapel Hill.
“The current standard of care is to give neonatal patients blood products . . . derived from adult blood, but neonatal blood and adult blood aren’t the same. Many of the components involved in clotting in newborns have differing levels of activity, or effectiveness, compared to the same components in adults. Our goal was to better understand how clotting in neonates differs from that in adults so that we can move closer to developing more effective treatment strategies for these infants.”
The researchers’ hypothesis was that fibrinogen from neonates would form clots that are different from those formed by adult fibrinogen, and this proved correct. However, the team was surprised to find that fibrinogen from adults did not integrate well with fibrinogen from neonates.
To test their hypothesis, the researchers took samples of neonate fibrinogen and adult fibrinogen and compared clot formation. The team looked at clots formed solely of adult fibrinogen, clots formed solely of neonate fibrinogen, and clots made from a mixture of the two.
Neonate fibrinogen formed less dense, more fragile clots than adult fibrinogen. Likewise, a mixture of adult and neonate fibrinogen formed clots that were fragile and less dense, even if there was relatively little neonate fibrinogen in the mixture.
The researchers also evaluated how long it took these clots to dissolve. Clots of neonate fibrinogen dissolved about twice as quickly as clots formed from adult fibrinogen.
Clots formed from an adult and neonate fibrinogen mixture dissolved at approximately the same rate as adult-only clots, regardless of the percentage of neonate fibrinogen in the mixture.
“This suggests that using adult fibrinogen in neonatal patients may pose an increased risk of embolism or other adverse thrombotic events,” said study author Nina Guzzetta, MD, of the Emory University School of Medicine in Atlanta, Georgia.
“This work drives home that newborns are not just small adults, and we still have much to learn about clotting in neonates. It also tells us that there is a great deal of room for improvement in the current standard of care for post-operative bleeding in neonates.”
“We are investigating several approaches that may help address this problem, evaluating various modes of action,” Dr Brown added. “It is possible that we can use various external factors that promote clotting to stimulate the fibrinogen in neonates to form a denser clot.”
“We are investigating possible alternatives to help neonates form a better clot after major surgery without having to use adult fibrinogen. For example, we are investigating the use of synthetic platelet-like particles developed by our team to augment hemostasis . . . in blood samples collected from these patients.” ![]()
FDA approves long-acting hemophilia B therapy
The US Food and Drug Administration (FDA) has approved Idelvion (Coagulation Factor IX [Recombinant], Albumin Fusion Protein) for the treatment of hemophilia B.
The product—a fusion protein linking recombinant coagulation factor IX with recombinant albumin—is intended for use in children and adults for routine prophylaxis to prevent or reduce the frequency of bleeding episodes, for on-demand control and prevention of bleeding episodes, and for the perioperative management of bleeding.
Appropriate patients age 12 and older can go up to 14 days between Idelvion infusions. This dosing interval has been achieved while maintaining high levels of factor IX activity—above 5% over 14 days at 75 IU/kg.
Idelvion is the first FDA-approved recombinant factor IX therapy that can extend the dosing interval up to 14 days.
Idelvion is expected to be available in the US later this month. The product is being developed by CSL Behring. For more details on the drug, see the full prescribing information.
Phase 3 trial
The FDA approved Idelvion based on results of the PROLONG-9FP clinical development program. PROLONG-9FP includes phase 1, 2, and 3 studies evaluating the safety and efficacy of Idelvion in adults and children (ages 1 to 61) with hemophilia B.
Data from the phase 3 study were recently published in Blood. This study included 63 previously treated male patients with severe hemophilia B. They had a mean age of 33 (range, 12 to 61).
The patients were divided into 2 groups. Group 1 (n=40) received routine prophylaxis with Idelvion once every 7 days for 26 weeks, followed by a 7-, 10- or 14-day prophylaxis regimen for a mean of 50, 38, or 51 weeks, respectively.
Group 2 received on-demand treatment with Idelvion for bleeding episodes for 26 weeks (n=23) and then switched to a 7-day prophylaxis regimen for a mean of 45 weeks (n=19).
The median annualized bleeding rate (ABR) was 2.0 in the prophylaxis arm (group 1) and 23.5 in the on-demand treatment arm (group 2). The median spontaneous ABRs were 0.0 and 17.0, respectively.
For patients in group 2, there was a significant reduction in median ABRs when patients switched from on-demand treatment to prophylaxis—19.22 and 1.58, respectively (P<0.0001). And there was a significant reduction in median spontaneous ABRs—15.43 and 0.00, respectively (P<0.0001).
Overall, 98.6% of bleeding episodes were treated successfully, including 93.6% that were treated with a single injection of Idelvion.
None of the patients developed inhibitors or experienced thromboembolic events, anaphylaxis, or life-threatening adverse events (AEs).
There were 347 treatment-emergent AEs reported in 54 (85.7%) patients. The most common were nasopharyngitis (25.4%), headache (23.8%), arthralgia (4.3%), and influenza (11.1%).
Eleven mild/moderate AEs in 5 patients (7.9%) were considered possibly related to Idelvion. Two patients discontinued treatment due to AEs—1 with hypersensitivity and 1 with headache. ![]()
The US Food and Drug Administration (FDA) has approved Idelvion (Coagulation Factor IX [Recombinant], Albumin Fusion Protein) for the treatment of hemophilia B.
The product—a fusion protein linking recombinant coagulation factor IX with recombinant albumin—is intended for use in children and adults for routine prophylaxis to prevent or reduce the frequency of bleeding episodes, for on-demand control and prevention of bleeding episodes, and for the perioperative management of bleeding.
Appropriate patients age 12 and older can go up to 14 days between Idelvion infusions. This dosing interval has been achieved while maintaining high levels of factor IX activity—above 5% over 14 days at 75 IU/kg.
Idelvion is the first FDA-approved recombinant factor IX therapy that can extend the dosing interval up to 14 days.
Idelvion is expected to be available in the US later this month. The product is being developed by CSL Behring. For more details on the drug, see the full prescribing information.
Phase 3 trial
The FDA approved Idelvion based on results of the PROLONG-9FP clinical development program. PROLONG-9FP includes phase 1, 2, and 3 studies evaluating the safety and efficacy of Idelvion in adults and children (ages 1 to 61) with hemophilia B.
Data from the phase 3 study were recently published in Blood. This study included 63 previously treated male patients with severe hemophilia B. They had a mean age of 33 (range, 12 to 61).
The patients were divided into 2 groups. Group 1 (n=40) received routine prophylaxis with Idelvion once every 7 days for 26 weeks, followed by a 7-, 10- or 14-day prophylaxis regimen for a mean of 50, 38, or 51 weeks, respectively.
Group 2 received on-demand treatment with Idelvion for bleeding episodes for 26 weeks (n=23) and then switched to a 7-day prophylaxis regimen for a mean of 45 weeks (n=19).
The median annualized bleeding rate (ABR) was 2.0 in the prophylaxis arm (group 1) and 23.5 in the on-demand treatment arm (group 2). The median spontaneous ABRs were 0.0 and 17.0, respectively.
For patients in group 2, there was a significant reduction in median ABRs when patients switched from on-demand treatment to prophylaxis—19.22 and 1.58, respectively (P<0.0001). And there was a significant reduction in median spontaneous ABRs—15.43 and 0.00, respectively (P<0.0001).
Overall, 98.6% of bleeding episodes were treated successfully, including 93.6% that were treated with a single injection of Idelvion.
None of the patients developed inhibitors or experienced thromboembolic events, anaphylaxis, or life-threatening adverse events (AEs).
There were 347 treatment-emergent AEs reported in 54 (85.7%) patients. The most common were nasopharyngitis (25.4%), headache (23.8%), arthralgia (4.3%), and influenza (11.1%).
Eleven mild/moderate AEs in 5 patients (7.9%) were considered possibly related to Idelvion. Two patients discontinued treatment due to AEs—1 with hypersensitivity and 1 with headache. ![]()
The US Food and Drug Administration (FDA) has approved Idelvion (Coagulation Factor IX [Recombinant], Albumin Fusion Protein) for the treatment of hemophilia B.
The product—a fusion protein linking recombinant coagulation factor IX with recombinant albumin—is intended for use in children and adults for routine prophylaxis to prevent or reduce the frequency of bleeding episodes, for on-demand control and prevention of bleeding episodes, and for the perioperative management of bleeding.
Appropriate patients age 12 and older can go up to 14 days between Idelvion infusions. This dosing interval has been achieved while maintaining high levels of factor IX activity—above 5% over 14 days at 75 IU/kg.
Idelvion is the first FDA-approved recombinant factor IX therapy that can extend the dosing interval up to 14 days.
Idelvion is expected to be available in the US later this month. The product is being developed by CSL Behring. For more details on the drug, see the full prescribing information.
Phase 3 trial
The FDA approved Idelvion based on results of the PROLONG-9FP clinical development program. PROLONG-9FP includes phase 1, 2, and 3 studies evaluating the safety and efficacy of Idelvion in adults and children (ages 1 to 61) with hemophilia B.
Data from the phase 3 study were recently published in Blood. This study included 63 previously treated male patients with severe hemophilia B. They had a mean age of 33 (range, 12 to 61).
The patients were divided into 2 groups. Group 1 (n=40) received routine prophylaxis with Idelvion once every 7 days for 26 weeks, followed by a 7-, 10- or 14-day prophylaxis regimen for a mean of 50, 38, or 51 weeks, respectively.
Group 2 received on-demand treatment with Idelvion for bleeding episodes for 26 weeks (n=23) and then switched to a 7-day prophylaxis regimen for a mean of 45 weeks (n=19).
The median annualized bleeding rate (ABR) was 2.0 in the prophylaxis arm (group 1) and 23.5 in the on-demand treatment arm (group 2). The median spontaneous ABRs were 0.0 and 17.0, respectively.
For patients in group 2, there was a significant reduction in median ABRs when patients switched from on-demand treatment to prophylaxis—19.22 and 1.58, respectively (P<0.0001). And there was a significant reduction in median spontaneous ABRs—15.43 and 0.00, respectively (P<0.0001).
Overall, 98.6% of bleeding episodes were treated successfully, including 93.6% that were treated with a single injection of Idelvion.
None of the patients developed inhibitors or experienced thromboembolic events, anaphylaxis, or life-threatening adverse events (AEs).
There were 347 treatment-emergent AEs reported in 54 (85.7%) patients. The most common were nasopharyngitis (25.4%), headache (23.8%), arthralgia (4.3%), and influenza (11.1%).
Eleven mild/moderate AEs in 5 patients (7.9%) were considered possibly related to Idelvion. Two patients discontinued treatment due to AEs—1 with hypersensitivity and 1 with headache. ![]()
FDA approves ibrutinib as first-line CLL therapy

Photo courtesy of Janssen
The US Food and Drug Administration (FDA) has approved the BTK inhibitor ibrutinib (Imbruvica) as a first-line treatment for patients with chronic lymphocytic leukemia (CLL).
This means ibrutinib is now FDA-approved to treat CLL patients regardless of their treatment history, including patients with 17p deletion.
Ibrutinib is also FDA-approved to treat Waldenström’s macroglobulinemia, and the drug was granted accelerated approval to treat patients with mantle cell lymphoma who have received at least 1 prior therapy.
Ibrutinib is jointly developed and commercialized by Pharmacyclics LLC, an AbbVie company, and Janssen Biotech, Inc. For more details on the drug, see the full prescribing information, available at imbruvica.com.
RESONATE-2 trial
The latest FDA approval for ibrutinib is based on results from the phase 3 RESONATE-2 trial (PCYC-1115), which were presented at the 2015 ASH Annual Meeting and simultaneously published in NEJM.
RESONATE-2 enrolled 269 treatment-naïve patients with CLL or small lymphocytic lymphoma who were 65 or older.
Patients were randomized to receive ibrutinib (n=136) at 420 mg once a day until progression or unacceptable toxicity, or chlorambucil (n=133) on days 1 and 15 of each 28-day cycle for up to 12 cycles. The starting dose for chlorambucil in cycle 1 was 0.5 mg/kg and was increased based on tolerability in cycle 2 by increments of 0.1 mg/kg to a maximum of 0.8 mg/kg.
The primary endpoint of the study was progression-free survival (PFS), as assessed by an independent review committee (IRC) according to the International Workshop on Chronic Lymphocytic Leukemia (iWCLL) 2008 criteria, with modification for treatment-related lymphocytosis.
Key secondary endpoints included overall response rate (based on the same iWCLL criteria), overall survival (OS), and safety.
Ibrutinib significantly prolonged PFS, as determined by the IRC, reducing the risk of progression or death by 84% compared to chlorambucil. The hazard ratio was 0.16 (P<0.001). The median PFS was not reached in the ibrutinib arm but was 18.9 months for the chlorambucil arm.
Ibrutinib significantly prolonged OS as well, although the median OS was not reached in either treatment arm. The OS rate at 24 months was 98% with ibrutinib and 85% with chlorambucil. The relative risk of death with ibrutinib was 84% lower than that with chlorambucil. The hazard ratio was 0.16 (P=0.001).
Ibrutinib was associated with a significantly higher IRC-assessed overall response rate compared to chlorambucil—82% and 35%, respectively (P<0.0001). Five patients (4%) in the ibrutinib arm achieved a complete response, as did 2 patients (2%) in the chlorambucil arm.
The median duration of treatment was 17.4 months in the ibrutinib arm and 7.1 months in the chlorambucil arm.
The most common adverse events of any grade—in the ibrutinib and chlorambucil arms, respectively—were diarrhea (42% and 17%), fatigue (30% and 38%), cough (22% and 15%), nausea (22% and 39%), peripheral edema (19% and 9%), dry eye (17% and 5%), arthralgia (16% and 7%), neutropenia (16% and 23%), and vomiting (13% and 20%).
Adverse events of grade 3 or higher—in the ibrutinib and chlorambucil arms, respectively—were neutropenia (10% and 18%), anemia (6% and 8%), hypertension (4% and 0%), pneumonia (4% and 2%), diarrhea (4% and 0%), maculopapular rash (3% and 2%), decreased platelet count (3% and 1%), abdominal pain (3% and 1%), hyponatremia (3% and 0%), thrombocytopenia (2% and 6%), febrile neutropenia (2% and 2%), upper respiratory tract infection (2% and 2%), pleural effusion (2% and 1%), cellulitis (2% and 0%), fatigue (1% and 5%), syncope (1% and 2%), and hemolytic anemia (0% and 2%). ![]()

Photo courtesy of Janssen
The US Food and Drug Administration (FDA) has approved the BTK inhibitor ibrutinib (Imbruvica) as a first-line treatment for patients with chronic lymphocytic leukemia (CLL).
This means ibrutinib is now FDA-approved to treat CLL patients regardless of their treatment history, including patients with 17p deletion.
Ibrutinib is also FDA-approved to treat Waldenström’s macroglobulinemia, and the drug was granted accelerated approval to treat patients with mantle cell lymphoma who have received at least 1 prior therapy.
Ibrutinib is jointly developed and commercialized by Pharmacyclics LLC, an AbbVie company, and Janssen Biotech, Inc. For more details on the drug, see the full prescribing information, available at imbruvica.com.
RESONATE-2 trial
The latest FDA approval for ibrutinib is based on results from the phase 3 RESONATE-2 trial (PCYC-1115), which were presented at the 2015 ASH Annual Meeting and simultaneously published in NEJM.
RESONATE-2 enrolled 269 treatment-naïve patients with CLL or small lymphocytic lymphoma who were 65 or older.
Patients were randomized to receive ibrutinib (n=136) at 420 mg once a day until progression or unacceptable toxicity, or chlorambucil (n=133) on days 1 and 15 of each 28-day cycle for up to 12 cycles. The starting dose for chlorambucil in cycle 1 was 0.5 mg/kg and was increased based on tolerability in cycle 2 by increments of 0.1 mg/kg to a maximum of 0.8 mg/kg.
The primary endpoint of the study was progression-free survival (PFS), as assessed by an independent review committee (IRC) according to the International Workshop on Chronic Lymphocytic Leukemia (iWCLL) 2008 criteria, with modification for treatment-related lymphocytosis.
Key secondary endpoints included overall response rate (based on the same iWCLL criteria), overall survival (OS), and safety.
Ibrutinib significantly prolonged PFS, as determined by the IRC, reducing the risk of progression or death by 84% compared to chlorambucil. The hazard ratio was 0.16 (P<0.001). The median PFS was not reached in the ibrutinib arm but was 18.9 months for the chlorambucil arm.
Ibrutinib significantly prolonged OS as well, although the median OS was not reached in either treatment arm. The OS rate at 24 months was 98% with ibrutinib and 85% with chlorambucil. The relative risk of death with ibrutinib was 84% lower than that with chlorambucil. The hazard ratio was 0.16 (P=0.001).
Ibrutinib was associated with a significantly higher IRC-assessed overall response rate compared to chlorambucil—82% and 35%, respectively (P<0.0001). Five patients (4%) in the ibrutinib arm achieved a complete response, as did 2 patients (2%) in the chlorambucil arm.
The median duration of treatment was 17.4 months in the ibrutinib arm and 7.1 months in the chlorambucil arm.
The most common adverse events of any grade—in the ibrutinib and chlorambucil arms, respectively—were diarrhea (42% and 17%), fatigue (30% and 38%), cough (22% and 15%), nausea (22% and 39%), peripheral edema (19% and 9%), dry eye (17% and 5%), arthralgia (16% and 7%), neutropenia (16% and 23%), and vomiting (13% and 20%).
Adverse events of grade 3 or higher—in the ibrutinib and chlorambucil arms, respectively—were neutropenia (10% and 18%), anemia (6% and 8%), hypertension (4% and 0%), pneumonia (4% and 2%), diarrhea (4% and 0%), maculopapular rash (3% and 2%), decreased platelet count (3% and 1%), abdominal pain (3% and 1%), hyponatremia (3% and 0%), thrombocytopenia (2% and 6%), febrile neutropenia (2% and 2%), upper respiratory tract infection (2% and 2%), pleural effusion (2% and 1%), cellulitis (2% and 0%), fatigue (1% and 5%), syncope (1% and 2%), and hemolytic anemia (0% and 2%). ![]()

Photo courtesy of Janssen
The US Food and Drug Administration (FDA) has approved the BTK inhibitor ibrutinib (Imbruvica) as a first-line treatment for patients with chronic lymphocytic leukemia (CLL).
This means ibrutinib is now FDA-approved to treat CLL patients regardless of their treatment history, including patients with 17p deletion.
Ibrutinib is also FDA-approved to treat Waldenström’s macroglobulinemia, and the drug was granted accelerated approval to treat patients with mantle cell lymphoma who have received at least 1 prior therapy.
Ibrutinib is jointly developed and commercialized by Pharmacyclics LLC, an AbbVie company, and Janssen Biotech, Inc. For more details on the drug, see the full prescribing information, available at imbruvica.com.
RESONATE-2 trial
The latest FDA approval for ibrutinib is based on results from the phase 3 RESONATE-2 trial (PCYC-1115), which were presented at the 2015 ASH Annual Meeting and simultaneously published in NEJM.
RESONATE-2 enrolled 269 treatment-naïve patients with CLL or small lymphocytic lymphoma who were 65 or older.
Patients were randomized to receive ibrutinib (n=136) at 420 mg once a day until progression or unacceptable toxicity, or chlorambucil (n=133) on days 1 and 15 of each 28-day cycle for up to 12 cycles. The starting dose for chlorambucil in cycle 1 was 0.5 mg/kg and was increased based on tolerability in cycle 2 by increments of 0.1 mg/kg to a maximum of 0.8 mg/kg.
The primary endpoint of the study was progression-free survival (PFS), as assessed by an independent review committee (IRC) according to the International Workshop on Chronic Lymphocytic Leukemia (iWCLL) 2008 criteria, with modification for treatment-related lymphocytosis.
Key secondary endpoints included overall response rate (based on the same iWCLL criteria), overall survival (OS), and safety.
Ibrutinib significantly prolonged PFS, as determined by the IRC, reducing the risk of progression or death by 84% compared to chlorambucil. The hazard ratio was 0.16 (P<0.001). The median PFS was not reached in the ibrutinib arm but was 18.9 months for the chlorambucil arm.
Ibrutinib significantly prolonged OS as well, although the median OS was not reached in either treatment arm. The OS rate at 24 months was 98% with ibrutinib and 85% with chlorambucil. The relative risk of death with ibrutinib was 84% lower than that with chlorambucil. The hazard ratio was 0.16 (P=0.001).
Ibrutinib was associated with a significantly higher IRC-assessed overall response rate compared to chlorambucil—82% and 35%, respectively (P<0.0001). Five patients (4%) in the ibrutinib arm achieved a complete response, as did 2 patients (2%) in the chlorambucil arm.
The median duration of treatment was 17.4 months in the ibrutinib arm and 7.1 months in the chlorambucil arm.
The most common adverse events of any grade—in the ibrutinib and chlorambucil arms, respectively—were diarrhea (42% and 17%), fatigue (30% and 38%), cough (22% and 15%), nausea (22% and 39%), peripheral edema (19% and 9%), dry eye (17% and 5%), arthralgia (16% and 7%), neutropenia (16% and 23%), and vomiting (13% and 20%).
Adverse events of grade 3 or higher—in the ibrutinib and chlorambucil arms, respectively—were neutropenia (10% and 18%), anemia (6% and 8%), hypertension (4% and 0%), pneumonia (4% and 2%), diarrhea (4% and 0%), maculopapular rash (3% and 2%), decreased platelet count (3% and 1%), abdominal pain (3% and 1%), hyponatremia (3% and 0%), thrombocytopenia (2% and 6%), febrile neutropenia (2% and 2%), upper respiratory tract infection (2% and 2%), pleural effusion (2% and 1%), cellulitis (2% and 0%), fatigue (1% and 5%), syncope (1% and 2%), and hemolytic anemia (0% and 2%). ![]()
TXA may increase risk of DVT but not PE

Photo courtesy of NIH
ORLANDO, FL—Results of a large, retrospective study suggest tranexamic acid (TXA) can reduce the need for transfusion in patients undergoing hip and knee replacement surgery without increasing the overall risk of venous thromboembolism (VTE).
However, patients treated with TXA were significantly more likely to develop deep vein thrombosis (DVT) than patients who did not receive the drug.
There was no significant difference between the treatment groups with regard to pulmonary embolism (PE).
These results were presented at the American Academy of Orthopaedic Surgeons (AAOS) 2016 Annual Meeting (abstract P101).
“[C]onflicting results have been published regarding the use of TXA in patients undergoing hip and knee replacement,” said study investigator Geoffrey Westrich, MD, of the Hospital for Special Surgery in New York, New York.
To assess the safety and efficacy of TXA in this patient population, Dr Westrich and his colleagues retrospectively reviewed the records of 4449 patients who had hip or knee replacement over a 6-month period.
There were 720 patients who received TXA topically, 636 who received TXA intravenously, and 3093 patients who did not receive the drug.
The investigators found that 9.7% of patients treated with either type of TXA received a blood transfusion, as did 22.1% of patients who were not treated with TXA.
TXA-treated patients received an average of 0.13 units of blood, compared to 0.37 units for patients in the non-TXA group.
The investigators said there was no significant difference in efficacy between topical and intravenous TXA.
“At our institution, TXA in either intravenous or topical form was effective in decreasing the amount of blood transfusions, as well as the number of units of blood transfused in primary and revision hip and knee replacement,” Dr Westrich said.
“Furthermore, when safety was evaluated, there was no statistically significant difference in blood clots in patients who received IV or topical TXA, reconfirming its safety.”
The odds of developing a hospital-acquired VTE was 1.63 among patients treated with TXA, which was not significantly higher than the odds for patients who did not receive the drug (P=0.24).
When the investigators evaluated DVT and PE separately, they found the TXA group had a significant increase in DVT (P=0.03) but not PE (P=0.94). ![]()

Photo courtesy of NIH
ORLANDO, FL—Results of a large, retrospective study suggest tranexamic acid (TXA) can reduce the need for transfusion in patients undergoing hip and knee replacement surgery without increasing the overall risk of venous thromboembolism (VTE).
However, patients treated with TXA were significantly more likely to develop deep vein thrombosis (DVT) than patients who did not receive the drug.
There was no significant difference between the treatment groups with regard to pulmonary embolism (PE).
These results were presented at the American Academy of Orthopaedic Surgeons (AAOS) 2016 Annual Meeting (abstract P101).
“[C]onflicting results have been published regarding the use of TXA in patients undergoing hip and knee replacement,” said study investigator Geoffrey Westrich, MD, of the Hospital for Special Surgery in New York, New York.
To assess the safety and efficacy of TXA in this patient population, Dr Westrich and his colleagues retrospectively reviewed the records of 4449 patients who had hip or knee replacement over a 6-month period.
There were 720 patients who received TXA topically, 636 who received TXA intravenously, and 3093 patients who did not receive the drug.
The investigators found that 9.7% of patients treated with either type of TXA received a blood transfusion, as did 22.1% of patients who were not treated with TXA.
TXA-treated patients received an average of 0.13 units of blood, compared to 0.37 units for patients in the non-TXA group.
The investigators said there was no significant difference in efficacy between topical and intravenous TXA.
“At our institution, TXA in either intravenous or topical form was effective in decreasing the amount of blood transfusions, as well as the number of units of blood transfused in primary and revision hip and knee replacement,” Dr Westrich said.
“Furthermore, when safety was evaluated, there was no statistically significant difference in blood clots in patients who received IV or topical TXA, reconfirming its safety.”
The odds of developing a hospital-acquired VTE was 1.63 among patients treated with TXA, which was not significantly higher than the odds for patients who did not receive the drug (P=0.24).
When the investigators evaluated DVT and PE separately, they found the TXA group had a significant increase in DVT (P=0.03) but not PE (P=0.94). ![]()

Photo courtesy of NIH
ORLANDO, FL—Results of a large, retrospective study suggest tranexamic acid (TXA) can reduce the need for transfusion in patients undergoing hip and knee replacement surgery without increasing the overall risk of venous thromboembolism (VTE).
However, patients treated with TXA were significantly more likely to develop deep vein thrombosis (DVT) than patients who did not receive the drug.
There was no significant difference between the treatment groups with regard to pulmonary embolism (PE).
These results were presented at the American Academy of Orthopaedic Surgeons (AAOS) 2016 Annual Meeting (abstract P101).
“[C]onflicting results have been published regarding the use of TXA in patients undergoing hip and knee replacement,” said study investigator Geoffrey Westrich, MD, of the Hospital for Special Surgery in New York, New York.
To assess the safety and efficacy of TXA in this patient population, Dr Westrich and his colleagues retrospectively reviewed the records of 4449 patients who had hip or knee replacement over a 6-month period.
There were 720 patients who received TXA topically, 636 who received TXA intravenously, and 3093 patients who did not receive the drug.
The investigators found that 9.7% of patients treated with either type of TXA received a blood transfusion, as did 22.1% of patients who were not treated with TXA.
TXA-treated patients received an average of 0.13 units of blood, compared to 0.37 units for patients in the non-TXA group.
The investigators said there was no significant difference in efficacy between topical and intravenous TXA.
“At our institution, TXA in either intravenous or topical form was effective in decreasing the amount of blood transfusions, as well as the number of units of blood transfused in primary and revision hip and knee replacement,” Dr Westrich said.
“Furthermore, when safety was evaluated, there was no statistically significant difference in blood clots in patients who received IV or topical TXA, reconfirming its safety.”
The odds of developing a hospital-acquired VTE was 1.63 among patients treated with TXA, which was not significantly higher than the odds for patients who did not receive the drug (P=0.24).
When the investigators evaluated DVT and PE separately, they found the TXA group had a significant increase in DVT (P=0.03) but not PE (P=0.94). ![]()
Changes in chromosome structure contribute to T-ALL, other cancers
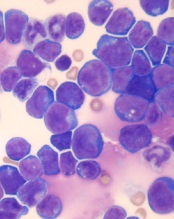
acute lymphoblastic leukemia
Image by Hind Medyouf
Breaches in looping chromosomal structures known as insulated neighborhoods can activate oncogenes capable of fueling aggressive tumor growth, according to research published in Science.
These neighborhood breaches were particularly frequent in T-cell acute lymphoblastic leukemia (T-ALL) and esophageal and liver carcinoma.
In some cases, the breaches allowed enhancer elements to activate previously silent oncogenes.
“This new understanding of the role of chromosome structure in cancer gene misregulation reveals the powerful influence of the genome’s structure in human health and disease,” said study author Richard Young, PhD, of the Whitehead Institute for Biomedical Research in Cambridge, Massachusetts.
These findings build on previous work in which Dr Young and his colleagues charted human genome structure and described its influence on gene control in healthy cells.
By mapping the genome’s 3-dimensional conformation, the researchers found that key genes controlling cell identity are found in insulated neighborhoods, whose loops are maintained through anchor sites bound by the protein CTCF.
All essential gene regulation, including the control of proper activation and repression, takes place within these enclosed neighborhoods.
The researchers also found these CTCF loop anchor sites are maintained across various cell types in the human body and are highly conserved in primate genomes. Such widespread structural conservation led the team to hypothesize that disruptions in genome conformation might be associated with disease, including cancers.
Sure enough, subsequent systematic genomic analysis of more than 50 cancer cell types revealed mutations affecting CTCF anchor sites, which led to the loss of insulated neighborhood boundaries.
By mapping insulated neighborhoods in T-ALL, the researchers found that tumor cell genomes contain recurrent microdeletions that eliminate the boundary sites of insulated neighborhoods containing prominent T-ALL proto-oncogenes.
The team also found the genomes of esophageal and liver carcinoma samples were enriched for boundary CTCF site mutations. The genes located in the most frequently mutated neighborhoods included known proto-oncogenes and genes not previously associated with these malignancies.
“We hadn’t known if these types of mutations contributed to cancer,” Dr Young said. “Now, we have multiple examples where these disruptions activate oncogenes that play major roles in tumorigenesis.”
The researchers noted that this oncogenic mechanism may be valuable for identifying genes that drive poorly understood cancers.
“In some cancers, such as esophageal carcinoma, the most frequent genetic mutation occurs at the CTCF sites, which is quite striking,” said Denes Hnisz, PhD, a researcher in the Young lab.
“In addition, there are still many cancers whose driver mutations and oncogenes are not known, and mapping altered structures may reveal the key oncogenes in these cancers.”
In an attempt to confirm the relationship between structural disruption and oncogenesis, the researchers used genome editing techniques to introduce CTCF anchor site deletions in non-malignant cells. They found these mutations were sufficient to activate oncogenes that are silent in normal cells.
The researchers said these findings suggest future mapping of genome structure in individual cancer patients might improve diagnosis and help guide treatment protocols.
“Now that we understand how perturbations in the genome’s structure can contribute to oncogenesis, we’re developing strategies to efficiently diagnose and potentially fix these faulty neighborhoods,” said Abe Weintraub, a graduate student in the Young lab. ![]()

acute lymphoblastic leukemia
Image by Hind Medyouf
Breaches in looping chromosomal structures known as insulated neighborhoods can activate oncogenes capable of fueling aggressive tumor growth, according to research published in Science.
These neighborhood breaches were particularly frequent in T-cell acute lymphoblastic leukemia (T-ALL) and esophageal and liver carcinoma.
In some cases, the breaches allowed enhancer elements to activate previously silent oncogenes.
“This new understanding of the role of chromosome structure in cancer gene misregulation reveals the powerful influence of the genome’s structure in human health and disease,” said study author Richard Young, PhD, of the Whitehead Institute for Biomedical Research in Cambridge, Massachusetts.
These findings build on previous work in which Dr Young and his colleagues charted human genome structure and described its influence on gene control in healthy cells.
By mapping the genome’s 3-dimensional conformation, the researchers found that key genes controlling cell identity are found in insulated neighborhoods, whose loops are maintained through anchor sites bound by the protein CTCF.
All essential gene regulation, including the control of proper activation and repression, takes place within these enclosed neighborhoods.
The researchers also found these CTCF loop anchor sites are maintained across various cell types in the human body and are highly conserved in primate genomes. Such widespread structural conservation led the team to hypothesize that disruptions in genome conformation might be associated with disease, including cancers.
Sure enough, subsequent systematic genomic analysis of more than 50 cancer cell types revealed mutations affecting CTCF anchor sites, which led to the loss of insulated neighborhood boundaries.
By mapping insulated neighborhoods in T-ALL, the researchers found that tumor cell genomes contain recurrent microdeletions that eliminate the boundary sites of insulated neighborhoods containing prominent T-ALL proto-oncogenes.
The team also found the genomes of esophageal and liver carcinoma samples were enriched for boundary CTCF site mutations. The genes located in the most frequently mutated neighborhoods included known proto-oncogenes and genes not previously associated with these malignancies.
“We hadn’t known if these types of mutations contributed to cancer,” Dr Young said. “Now, we have multiple examples where these disruptions activate oncogenes that play major roles in tumorigenesis.”
The researchers noted that this oncogenic mechanism may be valuable for identifying genes that drive poorly understood cancers.
“In some cancers, such as esophageal carcinoma, the most frequent genetic mutation occurs at the CTCF sites, which is quite striking,” said Denes Hnisz, PhD, a researcher in the Young lab.
“In addition, there are still many cancers whose driver mutations and oncogenes are not known, and mapping altered structures may reveal the key oncogenes in these cancers.”
In an attempt to confirm the relationship between structural disruption and oncogenesis, the researchers used genome editing techniques to introduce CTCF anchor site deletions in non-malignant cells. They found these mutations were sufficient to activate oncogenes that are silent in normal cells.
The researchers said these findings suggest future mapping of genome structure in individual cancer patients might improve diagnosis and help guide treatment protocols.
“Now that we understand how perturbations in the genome’s structure can contribute to oncogenesis, we’re developing strategies to efficiently diagnose and potentially fix these faulty neighborhoods,” said Abe Weintraub, a graduate student in the Young lab. ![]()

acute lymphoblastic leukemia
Image by Hind Medyouf
Breaches in looping chromosomal structures known as insulated neighborhoods can activate oncogenes capable of fueling aggressive tumor growth, according to research published in Science.
These neighborhood breaches were particularly frequent in T-cell acute lymphoblastic leukemia (T-ALL) and esophageal and liver carcinoma.
In some cases, the breaches allowed enhancer elements to activate previously silent oncogenes.
“This new understanding of the role of chromosome structure in cancer gene misregulation reveals the powerful influence of the genome’s structure in human health and disease,” said study author Richard Young, PhD, of the Whitehead Institute for Biomedical Research in Cambridge, Massachusetts.
These findings build on previous work in which Dr Young and his colleagues charted human genome structure and described its influence on gene control in healthy cells.
By mapping the genome’s 3-dimensional conformation, the researchers found that key genes controlling cell identity are found in insulated neighborhoods, whose loops are maintained through anchor sites bound by the protein CTCF.
All essential gene regulation, including the control of proper activation and repression, takes place within these enclosed neighborhoods.
The researchers also found these CTCF loop anchor sites are maintained across various cell types in the human body and are highly conserved in primate genomes. Such widespread structural conservation led the team to hypothesize that disruptions in genome conformation might be associated with disease, including cancers.
Sure enough, subsequent systematic genomic analysis of more than 50 cancer cell types revealed mutations affecting CTCF anchor sites, which led to the loss of insulated neighborhood boundaries.
By mapping insulated neighborhoods in T-ALL, the researchers found that tumor cell genomes contain recurrent microdeletions that eliminate the boundary sites of insulated neighborhoods containing prominent T-ALL proto-oncogenes.
The team also found the genomes of esophageal and liver carcinoma samples were enriched for boundary CTCF site mutations. The genes located in the most frequently mutated neighborhoods included known proto-oncogenes and genes not previously associated with these malignancies.
“We hadn’t known if these types of mutations contributed to cancer,” Dr Young said. “Now, we have multiple examples where these disruptions activate oncogenes that play major roles in tumorigenesis.”
The researchers noted that this oncogenic mechanism may be valuable for identifying genes that drive poorly understood cancers.
“In some cancers, such as esophageal carcinoma, the most frequent genetic mutation occurs at the CTCF sites, which is quite striking,” said Denes Hnisz, PhD, a researcher in the Young lab.
“In addition, there are still many cancers whose driver mutations and oncogenes are not known, and mapping altered structures may reveal the key oncogenes in these cancers.”
In an attempt to confirm the relationship between structural disruption and oncogenesis, the researchers used genome editing techniques to introduce CTCF anchor site deletions in non-malignant cells. They found these mutations were sufficient to activate oncogenes that are silent in normal cells.
The researchers said these findings suggest future mapping of genome structure in individual cancer patients might improve diagnosis and help guide treatment protocols.
“Now that we understand how perturbations in the genome’s structure can contribute to oncogenesis, we’re developing strategies to efficiently diagnose and potentially fix these faulty neighborhoods,” said Abe Weintraub, a graduate student in the Young lab. ![]()
Gene therapy granted orphan designation for hemophilia A

Image courtesy of NHLBI
The US Food and Drug Administration (FDA) has granted orphan designation to BMN 270, an investigational gene therapy, for the treatment of patients with hemophilia A.
BMN 270 is an adeno-associated virus-factor VIII (FVIII) vector designed to restore FVIII plasma concentrations in these patients.
The FDA grants orphan designation to drugs that are intended to treat diseases or conditions affecting fewer than 200,000 patients in the US.
The designation provides the drug’s sponsor with various development incentives, including opportunities to apply for research-related tax credits and grant funding, assistance in designing clinical trials, and 7 years of US market exclusivity if the drug is approved.
BMN 270 is under development by BioMarin Pharmaceutical Inc.
BioMarin is conducting a phase 1/2 study to evaluate the safety and efficacy of BMN 270 in up to 12 patients with severe hemophilia A.
Researchers are assessing the safety of a single infusion of BMN 270 and the change in FVIII expression level from baseline to 16 weeks after infusion.
The group is also assessing the impact of BMN 270 on the frequency of FVIII replacement therapy, the number of bleeding episodes requiring treatment, and any potential immune responses.
Patients will be monitored for safety and durability of effect for 5 years. BioMarin plans to provide an update on this trial in April. ![]()

Image courtesy of NHLBI
The US Food and Drug Administration (FDA) has granted orphan designation to BMN 270, an investigational gene therapy, for the treatment of patients with hemophilia A.
BMN 270 is an adeno-associated virus-factor VIII (FVIII) vector designed to restore FVIII plasma concentrations in these patients.
The FDA grants orphan designation to drugs that are intended to treat diseases or conditions affecting fewer than 200,000 patients in the US.
The designation provides the drug’s sponsor with various development incentives, including opportunities to apply for research-related tax credits and grant funding, assistance in designing clinical trials, and 7 years of US market exclusivity if the drug is approved.
BMN 270 is under development by BioMarin Pharmaceutical Inc.
BioMarin is conducting a phase 1/2 study to evaluate the safety and efficacy of BMN 270 in up to 12 patients with severe hemophilia A.
Researchers are assessing the safety of a single infusion of BMN 270 and the change in FVIII expression level from baseline to 16 weeks after infusion.
The group is also assessing the impact of BMN 270 on the frequency of FVIII replacement therapy, the number of bleeding episodes requiring treatment, and any potential immune responses.
Patients will be monitored for safety and durability of effect for 5 years. BioMarin plans to provide an update on this trial in April. ![]()

Image courtesy of NHLBI
The US Food and Drug Administration (FDA) has granted orphan designation to BMN 270, an investigational gene therapy, for the treatment of patients with hemophilia A.
BMN 270 is an adeno-associated virus-factor VIII (FVIII) vector designed to restore FVIII plasma concentrations in these patients.
The FDA grants orphan designation to drugs that are intended to treat diseases or conditions affecting fewer than 200,000 patients in the US.
The designation provides the drug’s sponsor with various development incentives, including opportunities to apply for research-related tax credits and grant funding, assistance in designing clinical trials, and 7 years of US market exclusivity if the drug is approved.
BMN 270 is under development by BioMarin Pharmaceutical Inc.
BioMarin is conducting a phase 1/2 study to evaluate the safety and efficacy of BMN 270 in up to 12 patients with severe hemophilia A.
Researchers are assessing the safety of a single infusion of BMN 270 and the change in FVIII expression level from baseline to 16 weeks after infusion.
The group is also assessing the impact of BMN 270 on the frequency of FVIII replacement therapy, the number of bleeding episodes requiring treatment, and any potential immune responses.
Patients will be monitored for safety and durability of effect for 5 years. BioMarin plans to provide an update on this trial in April.
Bundle can decrease CLABSI incidence

Staphylococcus infection
Photo by Bill Branson
A central catheter maintenance bundle can decrease the incidence of central line-associated bloodstream infections (CLABSIs), according to a study published in the American Journal of Critical Care.
A team from the healthcare company Select Medical developed and implemented the bundle at 30 long-term acute care hospitals.
The team used infection prevention guidelines from the Centers for Disease Control and Prevention as the core of the bundle, with mandatory use of alcohol-based central catheter caps and chlorhexidine gluconate dressings.
Ongoing education of clinical staff about the protocol and a checklist to track compliance were also key elements of the initiative.
At each hospital, staff nurses who demonstrated competency in the care of central catheters monitored implementation of the bundle for the initial 6 months of the study.
Researchers reviewed the medical records of 6660 patients discharged during the 14 months prior to the study and 6559 patients discharged after implementation of the bundle. Patient days and central catheter days before and after the bundle was implemented were comparable.
Six months after the bundle was implemented, the CLABSI standardized infection rate had dropped 29%. The rate was 1.28 in the 6 months before the bundle was implemented and 0.96 six months after implementation.
There was a mean reduction of 4.5 CLABSIs per hospital for 14 months after the bundle was implemented.
“Our results encourage the development and implementation of similar bundles as effective infection reduction strategies in [long-term acute care hospitals],” said study author Antony Grigonis, PhD, vice-president of quality and healthcare analytics at Select Medical.
“Preventing these infections can help reduce complications and the length of stay for other patients. This infection reduction could also translate to a savings of approximately $3.7 million annually for the 30 long-term acute care hospitals studied.”
Select Medical, which is based in Mechanicsburg, Pennsylvania, owns long-term acute care and inpatient rehabilitation hospitals, as well as occupational health and physical therapy clinics.

Staphylococcus infection
Photo by Bill Branson
A central catheter maintenance bundle can decrease the incidence of central line-associated bloodstream infections (CLABSIs), according to a study published in the American Journal of Critical Care.
A team from the healthcare company Select Medical developed and implemented the bundle at 30 long-term acute care hospitals.
The team used infection prevention guidelines from the Centers for Disease Control and Prevention as the core of the bundle, with mandatory use of alcohol-based central catheter caps and chlorhexidine gluconate dressings.
Ongoing education of clinical staff about the protocol and a checklist to track compliance were also key elements of the initiative.
At each hospital, staff nurses who demonstrated competency in the care of central catheters monitored implementation of the bundle for the initial 6 months of the study.
Researchers reviewed the medical records of 6660 patients discharged during the 14 months prior to the study and 6559 patients discharged after implementation of the bundle. Patient days and central catheter days before and after the bundle was implemented were comparable.
Six months after the bundle was implemented, the CLABSI standardized infection rate had dropped 29%. The rate was 1.28 in the 6 months before the bundle was implemented and 0.96 six months after implementation.
There was a mean reduction of 4.5 CLABSIs per hospital for 14 months after the bundle was implemented.
“Our results encourage the development and implementation of similar bundles as effective infection reduction strategies in [long-term acute care hospitals],” said study author Antony Grigonis, PhD, vice-president of quality and healthcare analytics at Select Medical.
“Preventing these infections can help reduce complications and the length of stay for other patients. This infection reduction could also translate to a savings of approximately $3.7 million annually for the 30 long-term acute care hospitals studied.”
Select Medical, which is based in Mechanicsburg, Pennsylvania, owns long-term acute care and inpatient rehabilitation hospitals, as well as occupational health and physical therapy clinics.

Staphylococcus infection
Photo by Bill Branson
A central catheter maintenance bundle can decrease the incidence of central line-associated bloodstream infections (CLABSIs), according to a study published in the American Journal of Critical Care.
A team from the healthcare company Select Medical developed and implemented the bundle at 30 long-term acute care hospitals.
The team used infection prevention guidelines from the Centers for Disease Control and Prevention as the core of the bundle, with mandatory use of alcohol-based central catheter caps and chlorhexidine gluconate dressings.
Ongoing education of clinical staff about the protocol and a checklist to track compliance were also key elements of the initiative.
At each hospital, staff nurses who demonstrated competency in the care of central catheters monitored implementation of the bundle for the initial 6 months of the study.
Researchers reviewed the medical records of 6660 patients discharged during the 14 months prior to the study and 6559 patients discharged after implementation of the bundle. Patient days and central catheter days before and after the bundle was implemented were comparable.
Six months after the bundle was implemented, the CLABSI standardized infection rate had dropped 29%. The rate was 1.28 in the 6 months before the bundle was implemented and 0.96 six months after implementation.
There was a mean reduction of 4.5 CLABSIs per hospital for 14 months after the bundle was implemented.
“Our results encourage the development and implementation of similar bundles as effective infection reduction strategies in [long-term acute care hospitals],” said study author Antony Grigonis, PhD, vice-president of quality and healthcare analytics at Select Medical.
“Preventing these infections can help reduce complications and the length of stay for other patients. This infection reduction could also translate to a savings of approximately $3.7 million annually for the 30 long-term acute care hospitals studied.”
Select Medical, which is based in Mechanicsburg, Pennsylvania, owns long-term acute care and inpatient rehabilitation hospitals, as well as occupational health and physical therapy clinics.
FDA issues new guidance on Zika virus transmission

Photo courtesy of NHS
The US Food and Drug Administration (FDA) has issued a guidance document intended to help reduce the risk of Zika virus transmission via human cells, tissues, and cellular and tissue-based products (HCT/Ps).
The guidance addresses donation of HCT/Ps from both living and deceased donors, including donors of umbilical cord blood and hematopoietic stem/progenitor cells.
In this document, the FDA recommends a 6-month deferral period for most HCT/P donors who may have been exposed to the Zika virus.
The exception is donors of gestational tissues, who should be considered ineligible if they were at risk of contracting the virus at any time during their pregnancy.
The new guidance is a part of the FDA’s ongoing efforts to protect HCT/Ps and blood products from Zika virus transmission. Last month, the agency issued recommendations for reducing the risk of Zika virus transmission via blood transfusion.
There is a potential risk that Zika virus can be transmitted by HCT/Ps used as part of a medical, surgical, or reproductive procedure. HCT/Ps include products such as corneas, bone, skin, heart valves, hematopoietic stem/progenitor cells, gestational tissues such as amniotic membrane, and reproductive tissues such as semen and oocytes.
According to the Centers for Disease Control and Prevention, Zika virus can be spread by a man to his sexual partners. To date, there have been several cases of sexual transmission in the US.
Current information about Zika virus detection in semen suggests the period of deferral for HCT/P donors should be longer than the period recommended for donors of whole blood and blood components.
Therefore, the FDA has recommended that living donors of HCT/Ps be considered ineligible if they were diagnosed with Zika virus infection, were in an area with active Zika virus transmission, or had sex with a male with either of those risk factors within the past 6 months.
Donors of umbilical cord blood, placenta, or other gestational tissues should be considered ineligible if they have had any of the above risk factors at any point during their pregnancy.
Deceased donors should be considered ineligible if they were diagnosed with Zika virus infection in the past 6 months.
A deferral period of 6 months was chosen because of the limited data available on the length of time the virus can persist in all tissues. Zika virus has been detected in tissues and body fluids after the virus is no longer detectable in the blood stream and has been detected in semen possibly up to 10 weeks after the onset of symptoms.
Less evidence exists regarding the potential for transmission of Zika virus by HCT/Ps typically recovered from deceased donors. As more information becomes available, the understanding of the risks to recipients of HCT/Ps, including HCT/Ps recovered from deceased donors, may evolve.
The FDA said it will continue to monitor the situation and evaluate new information regarding the associated risks as it becomes available.
In addition, the FDA said it is prioritizing the development of blood donor screening and diagnostic tests that may be useful for identifying the presence of or recent infection with the Zika virus. The agency is prepared to evaluate investigational vaccines and therapeutics that might be developed and review technology that may help suppress populations of the mosquitoes that can spread the virus.

Photo courtesy of NHS
The US Food and Drug Administration (FDA) has issued a guidance document intended to help reduce the risk of Zika virus transmission via human cells, tissues, and cellular and tissue-based products (HCT/Ps).
The guidance addresses donation of HCT/Ps from both living and deceased donors, including donors of umbilical cord blood and hematopoietic stem/progenitor cells.
In this document, the FDA recommends a 6-month deferral period for most HCT/P donors who may have been exposed to the Zika virus.
The exception is donors of gestational tissues, who should be considered ineligible if they were at risk of contracting the virus at any time during their pregnancy.
The new guidance is a part of the FDA’s ongoing efforts to protect HCT/Ps and blood products from Zika virus transmission. Last month, the agency issued recommendations for reducing the risk of Zika virus transmission via blood transfusion.
There is a potential risk that Zika virus can be transmitted by HCT/Ps used as part of a medical, surgical, or reproductive procedure. HCT/Ps include products such as corneas, bone, skin, heart valves, hematopoietic stem/progenitor cells, gestational tissues such as amniotic membrane, and reproductive tissues such as semen and oocytes.
According to the Centers for Disease Control and Prevention, Zika virus can be spread by a man to his sexual partners. To date, there have been several cases of sexual transmission in the US.
Current information about Zika virus detection in semen suggests the period of deferral for HCT/P donors should be longer than the period recommended for donors of whole blood and blood components.
Therefore, the FDA has recommended that living donors of HCT/Ps be considered ineligible if they were diagnosed with Zika virus infection, were in an area with active Zika virus transmission, or had sex with a male with either of those risk factors within the past 6 months.
Donors of umbilical cord blood, placenta, or other gestational tissues should be considered ineligible if they have had any of the above risk factors at any point during their pregnancy.
Deceased donors should be considered ineligible if they were diagnosed with Zika virus infection in the past 6 months.
A deferral period of 6 months was chosen because of the limited data available on the length of time the virus can persist in all tissues. Zika virus has been detected in tissues and body fluids after the virus is no longer detectable in the blood stream and has been detected in semen possibly up to 10 weeks after the onset of symptoms.
Less evidence exists regarding the potential for transmission of Zika virus by HCT/Ps typically recovered from deceased donors. As more information becomes available, the understanding of the risks to recipients of HCT/Ps, including HCT/Ps recovered from deceased donors, may evolve.
The FDA said it will continue to monitor the situation and evaluate new information regarding the associated risks as it becomes available.
In addition, the FDA said it is prioritizing the development of blood donor screening and diagnostic tests that may be useful for identifying the presence of or recent infection with the Zika virus. The agency is prepared to evaluate investigational vaccines and therapeutics that might be developed and review technology that may help suppress populations of the mosquitoes that can spread the virus.

Photo courtesy of NHS
The US Food and Drug Administration (FDA) has issued a guidance document intended to help reduce the risk of Zika virus transmission via human cells, tissues, and cellular and tissue-based products (HCT/Ps).
The guidance addresses donation of HCT/Ps from both living and deceased donors, including donors of umbilical cord blood and hematopoietic stem/progenitor cells.
In this document, the FDA recommends a 6-month deferral period for most HCT/P donors who may have been exposed to the Zika virus.
The exception is donors of gestational tissues, who should be considered ineligible if they were at risk of contracting the virus at any time during their pregnancy.
The new guidance is a part of the FDA’s ongoing efforts to protect HCT/Ps and blood products from Zika virus transmission. Last month, the agency issued recommendations for reducing the risk of Zika virus transmission via blood transfusion.
There is a potential risk that Zika virus can be transmitted by HCT/Ps used as part of a medical, surgical, or reproductive procedure. HCT/Ps include products such as corneas, bone, skin, heart valves, hematopoietic stem/progenitor cells, gestational tissues such as amniotic membrane, and reproductive tissues such as semen and oocytes.
According to the Centers for Disease Control and Prevention, Zika virus can be spread by a man to his sexual partners. To date, there have been several cases of sexual transmission in the US.
Current information about Zika virus detection in semen suggests the period of deferral for HCT/P donors should be longer than the period recommended for donors of whole blood and blood components.
Therefore, the FDA has recommended that living donors of HCT/Ps be considered ineligible if they were diagnosed with Zika virus infection, were in an area with active Zika virus transmission, or had sex with a male with either of those risk factors within the past 6 months.
Donors of umbilical cord blood, placenta, or other gestational tissues should be considered ineligible if they have had any of the above risk factors at any point during their pregnancy.
Deceased donors should be considered ineligible if they were diagnosed with Zika virus infection in the past 6 months.
A deferral period of 6 months was chosen because of the limited data available on the length of time the virus can persist in all tissues. Zika virus has been detected in tissues and body fluids after the virus is no longer detectable in the blood stream and has been detected in semen possibly up to 10 weeks after the onset of symptoms.
Less evidence exists regarding the potential for transmission of Zika virus by HCT/Ps typically recovered from deceased donors. As more information becomes available, the understanding of the risks to recipients of HCT/Ps, including HCT/Ps recovered from deceased donors, may evolve.
The FDA said it will continue to monitor the situation and evaluate new information regarding the associated risks as it becomes available.
In addition, the FDA said it is prioritizing the development of blood donor screening and diagnostic tests that may be useful for identifying the presence of or recent infection with the Zika virus. The agency is prepared to evaluate investigational vaccines and therapeutics that might be developed and review technology that may help suppress populations of the mosquitoes that can spread the virus.
Factors driving blood cell development
Researchers believe they have identified key factors that drive hematopoietic specification and differentiation.
The team performed detailed analyses on cells at 6 consecutive stages of development—starting with embryonic stem cells and ending with macrophages.
They said this revealed the complete set of regulatory elements driving differential gene expression during these stages of development.
The researchers described this work in Developmental Cell.
The team studied hematopoietic specification and differentiation by looking at 6 stages of cell development—embryonic stem cells, mesoderm cells, hemangioblasts, hemogenic endothelium, hematopoietic precursors, and macrophages.
“We examined how embryonic cells develop towards blood cells by collecting multi-omics data from measuring gene activity, changes in chromosome structure, and the interaction of regulatory factors with the genes themselves,” explained study author Constanze Bonifer, PhD, of the University of Birmingham in the UK.
“Our research shows, in unprecedented detail, how a vast network of interacting genes control blood cell development. It also shows how we can use such data to enhance our knowledge of this process.”
The researchers said their findings help explain how regulatory elements in the DNA work together, driving gene expression and the switch from one developmental stage to another.
The team believes the work also revealed the minimum requirements for generating blood cells from an unrelated, cultured cell type.
The researchers have made their findings available to the public on the following website: http://www.haemopoiesis.leeds.ac.uk/data_analysis/.

Researchers believe they have identified key factors that drive hematopoietic specification and differentiation.
The team performed detailed analyses on cells at 6 consecutive stages of development—starting with embryonic stem cells and ending with macrophages.
They said this revealed the complete set of regulatory elements driving differential gene expression during these stages of development.
The researchers described this work in Developmental Cell.
The team studied hematopoietic specification and differentiation by looking at 6 stages of cell development—embryonic stem cells, mesoderm cells, hemangioblasts, hemogenic endothelium, hematopoietic precursors, and macrophages.
“We examined how embryonic cells develop towards blood cells by collecting multi-omics data from measuring gene activity, changes in chromosome structure, and the interaction of regulatory factors with the genes themselves,” explained study author Constanze Bonifer, PhD, of the University of Birmingham in the UK.
“Our research shows, in unprecedented detail, how a vast network of interacting genes control blood cell development. It also shows how we can use such data to enhance our knowledge of this process.”
The researchers said their findings help explain how regulatory elements in the DNA work together, driving gene expression and the switch from one developmental stage to another.
The team believes the work also revealed the minimum requirements for generating blood cells from an unrelated, cultured cell type.
The researchers have made their findings available to the public on the following website: http://www.haemopoiesis.leeds.ac.uk/data_analysis/.

Researchers believe they have identified key factors that drive hematopoietic specification and differentiation.
The team performed detailed analyses on cells at 6 consecutive stages of development—starting with embryonic stem cells and ending with macrophages.
They said this revealed the complete set of regulatory elements driving differential gene expression during these stages of development.
The researchers described this work in Developmental Cell.
The team studied hematopoietic specification and differentiation by looking at 6 stages of cell development—embryonic stem cells, mesoderm cells, hemangioblasts, hemogenic endothelium, hematopoietic precursors, and macrophages.
“We examined how embryonic cells develop towards blood cells by collecting multi-omics data from measuring gene activity, changes in chromosome structure, and the interaction of regulatory factors with the genes themselves,” explained study author Constanze Bonifer, PhD, of the University of Birmingham in the UK.
“Our research shows, in unprecedented detail, how a vast network of interacting genes control blood cell development. It also shows how we can use such data to enhance our knowledge of this process.”
The researchers said their findings help explain how regulatory elements in the DNA work together, driving gene expression and the switch from one developmental stage to another.
The team believes the work also revealed the minimum requirements for generating blood cells from an unrelated, cultured cell type.
The researchers have made their findings available to the public on the following website: http://www.haemopoiesis.leeds.ac.uk/data_analysis/.