User login
Mississippi Law Threatens Abortion Access
Only obstetrician-gynecologists with privileges at local hospitals will be able to perform abortions under a new law in Mississippi.
The law, signed April 16 by Gov. Phil Bryant (R), requires that all physicians who perform abortions at an abortion facility must be board-certified or board-eligible ob.gyns. and must have staff and admitting privileges at a local hospital. The law also requires that a person trained in CPR must be present at the abortion clinic whenever it is open. The law is scheduled to go into effect in July.
The requirements are necessary, Gov. Bryant said, to protect patient safety in the event of a complication during an abortion procedure.
"I believe that all human life is precious, and as governor, I will work to ensure that the lives of the born and unborn are protected in Mississippi," he said in a statement.
Abortion rights advocates say the restrictions are an attempt to eliminate abortions in the state.
Currently, only one abortion clinic, Jackson Womens Health Organization, operates in the state. Shelley Abrams, the clinic’s executive director, said that only one of their physicians currently has admitting privileges at a local hospital.
Going forward, they will attempt to gain privileges for the other physicians who work at the clinic, but Ms. Abrams said that could be difficult. For instance, hospitals generally require that physicians with staff and admitting privileges take call and admit patients. That is a major hurdle for most the clinic’s doctors who live outside of Mississippi, she said.
Ms. Abrams said that she anticipates that hospitals will face pressure from antiabortion activists to deny privileges to the clinic’s physicians. "The antiabortionists in Jackson, Miss., have made the entire state a very inhospitable place for medical practitioners," she said.
If they are unable to gain privileges for their physicians, Ms. Abrams said that they will be forced to challenge the law in court to keep the clinic running.
The state has long been an abortion battleground. Last November, Mississippi voters rejected a "personhood amendment" that would have changed the state’s constitution to grant legal rights to embryos, starting at the time of fertilization.
The Mississippi law was signed just days after Arizona enacted controversial new abortion restrictions that will ban abortions at 20 weeks’ gestation and require that women receive an ultrasound before an abortion can be performed.
Arizona Gov. Jan Brewer (R) praised the law as "common sense" and added that "knowing that abortions become riskier the later they are performed in pregnancy, it only makes sense to prohibit these procedures past 20 weeks."
The Arizona law is one of the most extreme in the country, according to reproductive rights advocates. The Center for Reproductive Rights, which is challenging the law in federal court, noted that 18-20 weeks’ gestation is a time when many women undergo a comprehensive scan to uncover major fetal abnormalities and health risks to the mother.
"Some women at risk of grave complications will be forced to decide whether to proceed with their pregnancies in the dark, before they have all the information they need to arrive at their choices," Nancy Northup, the group’s president and CEO, said in a statement.
Seven other states have similar restrictions on abortions after 20 weeks, according to Gov. Brewer.
Abortion legislation is being considered in many statehouses this year: In the first three months of 2012, 75 abortion restrictions were passed by at least one state legislative chamber, according to an analysis from the Guttmacher Institute. That pace isn’t record setting, but it’s unusually high for an election year, the institute noted. So far, most of the pending legislation has focused on requirements that women receive an ultrasound before an abortion, limiting access to medical abortions, and bans on abortion after a certain gestational age.
Only obstetrician-gynecologists with privileges at local hospitals will be able to perform abortions under a new law in Mississippi.
The law, signed April 16 by Gov. Phil Bryant (R), requires that all physicians who perform abortions at an abortion facility must be board-certified or board-eligible ob.gyns. and must have staff and admitting privileges at a local hospital. The law also requires that a person trained in CPR must be present at the abortion clinic whenever it is open. The law is scheduled to go into effect in July.
The requirements are necessary, Gov. Bryant said, to protect patient safety in the event of a complication during an abortion procedure.
"I believe that all human life is precious, and as governor, I will work to ensure that the lives of the born and unborn are protected in Mississippi," he said in a statement.
Abortion rights advocates say the restrictions are an attempt to eliminate abortions in the state.
Currently, only one abortion clinic, Jackson Womens Health Organization, operates in the state. Shelley Abrams, the clinic’s executive director, said that only one of their physicians currently has admitting privileges at a local hospital.
Going forward, they will attempt to gain privileges for the other physicians who work at the clinic, but Ms. Abrams said that could be difficult. For instance, hospitals generally require that physicians with staff and admitting privileges take call and admit patients. That is a major hurdle for most the clinic’s doctors who live outside of Mississippi, she said.
Ms. Abrams said that she anticipates that hospitals will face pressure from antiabortion activists to deny privileges to the clinic’s physicians. "The antiabortionists in Jackson, Miss., have made the entire state a very inhospitable place for medical practitioners," she said.
If they are unable to gain privileges for their physicians, Ms. Abrams said that they will be forced to challenge the law in court to keep the clinic running.
The state has long been an abortion battleground. Last November, Mississippi voters rejected a "personhood amendment" that would have changed the state’s constitution to grant legal rights to embryos, starting at the time of fertilization.
The Mississippi law was signed just days after Arizona enacted controversial new abortion restrictions that will ban abortions at 20 weeks’ gestation and require that women receive an ultrasound before an abortion can be performed.
Arizona Gov. Jan Brewer (R) praised the law as "common sense" and added that "knowing that abortions become riskier the later they are performed in pregnancy, it only makes sense to prohibit these procedures past 20 weeks."
The Arizona law is one of the most extreme in the country, according to reproductive rights advocates. The Center for Reproductive Rights, which is challenging the law in federal court, noted that 18-20 weeks’ gestation is a time when many women undergo a comprehensive scan to uncover major fetal abnormalities and health risks to the mother.
"Some women at risk of grave complications will be forced to decide whether to proceed with their pregnancies in the dark, before they have all the information they need to arrive at their choices," Nancy Northup, the group’s president and CEO, said in a statement.
Seven other states have similar restrictions on abortions after 20 weeks, according to Gov. Brewer.
Abortion legislation is being considered in many statehouses this year: In the first three months of 2012, 75 abortion restrictions were passed by at least one state legislative chamber, according to an analysis from the Guttmacher Institute. That pace isn’t record setting, but it’s unusually high for an election year, the institute noted. So far, most of the pending legislation has focused on requirements that women receive an ultrasound before an abortion, limiting access to medical abortions, and bans on abortion after a certain gestational age.
Only obstetrician-gynecologists with privileges at local hospitals will be able to perform abortions under a new law in Mississippi.
The law, signed April 16 by Gov. Phil Bryant (R), requires that all physicians who perform abortions at an abortion facility must be board-certified or board-eligible ob.gyns. and must have staff and admitting privileges at a local hospital. The law also requires that a person trained in CPR must be present at the abortion clinic whenever it is open. The law is scheduled to go into effect in July.
The requirements are necessary, Gov. Bryant said, to protect patient safety in the event of a complication during an abortion procedure.
"I believe that all human life is precious, and as governor, I will work to ensure that the lives of the born and unborn are protected in Mississippi," he said in a statement.
Abortion rights advocates say the restrictions are an attempt to eliminate abortions in the state.
Currently, only one abortion clinic, Jackson Womens Health Organization, operates in the state. Shelley Abrams, the clinic’s executive director, said that only one of their physicians currently has admitting privileges at a local hospital.
Going forward, they will attempt to gain privileges for the other physicians who work at the clinic, but Ms. Abrams said that could be difficult. For instance, hospitals generally require that physicians with staff and admitting privileges take call and admit patients. That is a major hurdle for most the clinic’s doctors who live outside of Mississippi, she said.
Ms. Abrams said that she anticipates that hospitals will face pressure from antiabortion activists to deny privileges to the clinic’s physicians. "The antiabortionists in Jackson, Miss., have made the entire state a very inhospitable place for medical practitioners," she said.
If they are unable to gain privileges for their physicians, Ms. Abrams said that they will be forced to challenge the law in court to keep the clinic running.
The state has long been an abortion battleground. Last November, Mississippi voters rejected a "personhood amendment" that would have changed the state’s constitution to grant legal rights to embryos, starting at the time of fertilization.
The Mississippi law was signed just days after Arizona enacted controversial new abortion restrictions that will ban abortions at 20 weeks’ gestation and require that women receive an ultrasound before an abortion can be performed.
Arizona Gov. Jan Brewer (R) praised the law as "common sense" and added that "knowing that abortions become riskier the later they are performed in pregnancy, it only makes sense to prohibit these procedures past 20 weeks."
The Arizona law is one of the most extreme in the country, according to reproductive rights advocates. The Center for Reproductive Rights, which is challenging the law in federal court, noted that 18-20 weeks’ gestation is a time when many women undergo a comprehensive scan to uncover major fetal abnormalities and health risks to the mother.
"Some women at risk of grave complications will be forced to decide whether to proceed with their pregnancies in the dark, before they have all the information they need to arrive at their choices," Nancy Northup, the group’s president and CEO, said in a statement.
Seven other states have similar restrictions on abortions after 20 weeks, according to Gov. Brewer.
Abortion legislation is being considered in many statehouses this year: In the first three months of 2012, 75 abortion restrictions were passed by at least one state legislative chamber, according to an analysis from the Guttmacher Institute. That pace isn’t record setting, but it’s unusually high for an election year, the institute noted. So far, most of the pending legislation has focused on requirements that women receive an ultrasound before an abortion, limiting access to medical abortions, and bans on abortion after a certain gestational age.
Implementing Health Reform: Physician Compare
Starting next year, Medicare will begin publicly displaying group-level physician performance data. The data will come from the voluntary Physician Quality Reporting System (PQRS) and will be posted on the new Physician Compare website, as required by the Affordable Care Act.
The Centers for Medicare and Medicaid Services launched the Physician Compare website in January 2011; it currently displays mostly biographical information on physicians who accept Medicare patients. It also lists physicians who have successfully reported quality data to the agency through the PQRS. Beginning next January, CMS will add quality measure performance rates for group practices that voluntarily submitted data this year under the PQRS. The agency will only post data on measures that have at least a 35-patient sample size.
Dr. Glen R. Stream, president of the American Academy of Family Physicians, shared his views on how this information might be used by physicians and patients.
Question: Doctors have raised concerns about errors and inaccurate information on Physician Compare since it launched in 2011. Does the site provide a valuable service for patients?
Dr. Stream: It’s not surprising that in a database this large there would be some inaccurate or incomplete information. That was certainly one of the concerns for the AAFP. The challenge is to make sure this information is presented in a way that it can be understood, interpreted, and potentially acted on by the public. A lot of these quality measures are data that people in the health care industry understand but the general public may not. And that means that the expectations for performance may be different. A good performance on a given quality metric might be 50%, but a patient is going to look at that and say, "When I went to school 50% was an F." So the inclusion of some comparative data may be necessary. For instance, many reporting tools compare an individual physician or a medical group’s performance to the average performance of the entire data set. Or they clearly show what the target was and whether that group or person met it.
Question: CMS officials have said that they will begin the public reporting of quality data with group-level data rather than physician-level data. What’s the downside to using information about individuals?
Dr. Stream: One of the challenges when you get down to individual physician reporting is, do you have a sufficient number of patients in that category to have meaningful information? If I have 15 diabetic patients and my endocrinology colleague has 1,000, it’s hard to compare those two data sets. For example, I get a quality report card from one of the regional insurers in Washington state, where I practice, and when I get that report card it will say that I have X percent achievement of a certain quality metric. But it will also have an asterisk indicating whether there is actually a statistically valid number of patients in that metric. It may be that I got a great score, but if I have just four patients and they all met the metric, that’s not a valid number.
Question: Is performance in the PQRS the right data to use for public reporting?
Dr. Stream: Overall, I would say that the quality metrics that are in the PQRS quality data set are good ones. There are certainly some that could be improved. But the quality improvement on the measures themselves is ongoing, just like the quality improvement in the health care delivery to achieve them.
Question: Do you think that this type of public reporting will be an effective lever for changing physician behavior and improving care?
Dr. Stream: I think that’s a question to be answered over time. My personal opinion is that comparative information between physicians and medical groups has much more of an influence on the physicians and the medical groups than it does on the patients. Physicians are by nature competitive perfectionists. I want my quality metrics to be at the 99th percentile because I believe I’m a really good doctor and I want my metrics to reflect that. The transparency of this information from one physician and one medical group to another is perhaps more important than the public availability of it. My personal experience so far is that there are already a number of sites out there – some that are consumer sponsored and some that are health plan sponsored – that rate physicians. But most often patients choose specialty physicians based on the recommendation of their primary care physician. And otherwise they choose doctors based on the recommendations of friends, neighbors, and family.
Dr. Stream is the president of the American Academy of Family Physicians. He practices in a large multispecialty practice in Spokane, Wash.
Starting next year, Medicare will begin publicly displaying group-level physician performance data. The data will come from the voluntary Physician Quality Reporting System (PQRS) and will be posted on the new Physician Compare website, as required by the Affordable Care Act.
The Centers for Medicare and Medicaid Services launched the Physician Compare website in January 2011; it currently displays mostly biographical information on physicians who accept Medicare patients. It also lists physicians who have successfully reported quality data to the agency through the PQRS. Beginning next January, CMS will add quality measure performance rates for group practices that voluntarily submitted data this year under the PQRS. The agency will only post data on measures that have at least a 35-patient sample size.
Dr. Glen R. Stream, president of the American Academy of Family Physicians, shared his views on how this information might be used by physicians and patients.
Question: Doctors have raised concerns about errors and inaccurate information on Physician Compare since it launched in 2011. Does the site provide a valuable service for patients?
Dr. Stream: It’s not surprising that in a database this large there would be some inaccurate or incomplete information. That was certainly one of the concerns for the AAFP. The challenge is to make sure this information is presented in a way that it can be understood, interpreted, and potentially acted on by the public. A lot of these quality measures are data that people in the health care industry understand but the general public may not. And that means that the expectations for performance may be different. A good performance on a given quality metric might be 50%, but a patient is going to look at that and say, "When I went to school 50% was an F." So the inclusion of some comparative data may be necessary. For instance, many reporting tools compare an individual physician or a medical group’s performance to the average performance of the entire data set. Or they clearly show what the target was and whether that group or person met it.
Question: CMS officials have said that they will begin the public reporting of quality data with group-level data rather than physician-level data. What’s the downside to using information about individuals?
Dr. Stream: One of the challenges when you get down to individual physician reporting is, do you have a sufficient number of patients in that category to have meaningful information? If I have 15 diabetic patients and my endocrinology colleague has 1,000, it’s hard to compare those two data sets. For example, I get a quality report card from one of the regional insurers in Washington state, where I practice, and when I get that report card it will say that I have X percent achievement of a certain quality metric. But it will also have an asterisk indicating whether there is actually a statistically valid number of patients in that metric. It may be that I got a great score, but if I have just four patients and they all met the metric, that’s not a valid number.
Question: Is performance in the PQRS the right data to use for public reporting?
Dr. Stream: Overall, I would say that the quality metrics that are in the PQRS quality data set are good ones. There are certainly some that could be improved. But the quality improvement on the measures themselves is ongoing, just like the quality improvement in the health care delivery to achieve them.
Question: Do you think that this type of public reporting will be an effective lever for changing physician behavior and improving care?
Dr. Stream: I think that’s a question to be answered over time. My personal opinion is that comparative information between physicians and medical groups has much more of an influence on the physicians and the medical groups than it does on the patients. Physicians are by nature competitive perfectionists. I want my quality metrics to be at the 99th percentile because I believe I’m a really good doctor and I want my metrics to reflect that. The transparency of this information from one physician and one medical group to another is perhaps more important than the public availability of it. My personal experience so far is that there are already a number of sites out there – some that are consumer sponsored and some that are health plan sponsored – that rate physicians. But most often patients choose specialty physicians based on the recommendation of their primary care physician. And otherwise they choose doctors based on the recommendations of friends, neighbors, and family.
Dr. Stream is the president of the American Academy of Family Physicians. He practices in a large multispecialty practice in Spokane, Wash.
Starting next year, Medicare will begin publicly displaying group-level physician performance data. The data will come from the voluntary Physician Quality Reporting System (PQRS) and will be posted on the new Physician Compare website, as required by the Affordable Care Act.
The Centers for Medicare and Medicaid Services launched the Physician Compare website in January 2011; it currently displays mostly biographical information on physicians who accept Medicare patients. It also lists physicians who have successfully reported quality data to the agency through the PQRS. Beginning next January, CMS will add quality measure performance rates for group practices that voluntarily submitted data this year under the PQRS. The agency will only post data on measures that have at least a 35-patient sample size.
Dr. Glen R. Stream, president of the American Academy of Family Physicians, shared his views on how this information might be used by physicians and patients.
Question: Doctors have raised concerns about errors and inaccurate information on Physician Compare since it launched in 2011. Does the site provide a valuable service for patients?
Dr. Stream: It’s not surprising that in a database this large there would be some inaccurate or incomplete information. That was certainly one of the concerns for the AAFP. The challenge is to make sure this information is presented in a way that it can be understood, interpreted, and potentially acted on by the public. A lot of these quality measures are data that people in the health care industry understand but the general public may not. And that means that the expectations for performance may be different. A good performance on a given quality metric might be 50%, but a patient is going to look at that and say, "When I went to school 50% was an F." So the inclusion of some comparative data may be necessary. For instance, many reporting tools compare an individual physician or a medical group’s performance to the average performance of the entire data set. Or they clearly show what the target was and whether that group or person met it.
Question: CMS officials have said that they will begin the public reporting of quality data with group-level data rather than physician-level data. What’s the downside to using information about individuals?
Dr. Stream: One of the challenges when you get down to individual physician reporting is, do you have a sufficient number of patients in that category to have meaningful information? If I have 15 diabetic patients and my endocrinology colleague has 1,000, it’s hard to compare those two data sets. For example, I get a quality report card from one of the regional insurers in Washington state, where I practice, and when I get that report card it will say that I have X percent achievement of a certain quality metric. But it will also have an asterisk indicating whether there is actually a statistically valid number of patients in that metric. It may be that I got a great score, but if I have just four patients and they all met the metric, that’s not a valid number.
Question: Is performance in the PQRS the right data to use for public reporting?
Dr. Stream: Overall, I would say that the quality metrics that are in the PQRS quality data set are good ones. There are certainly some that could be improved. But the quality improvement on the measures themselves is ongoing, just like the quality improvement in the health care delivery to achieve them.
Question: Do you think that this type of public reporting will be an effective lever for changing physician behavior and improving care?
Dr. Stream: I think that’s a question to be answered over time. My personal opinion is that comparative information between physicians and medical groups has much more of an influence on the physicians and the medical groups than it does on the patients. Physicians are by nature competitive perfectionists. I want my quality metrics to be at the 99th percentile because I believe I’m a really good doctor and I want my metrics to reflect that. The transparency of this information from one physician and one medical group to another is perhaps more important than the public availability of it. My personal experience so far is that there are already a number of sites out there – some that are consumer sponsored and some that are health plan sponsored – that rate physicians. But most often patients choose specialty physicians based on the recommendation of their primary care physician. And otherwise they choose doctors based on the recommendations of friends, neighbors, and family.
Dr. Stream is the president of the American Academy of Family Physicians. He practices in a large multispecialty practice in Spokane, Wash.
Teamwork Mandate Headed for MOC
SAN DIEGO – Hospitalists undergoing maintenance of certification may soon be required to complete a module focused on team-based care, but be allowed drop one requirement related to practice improvement.
Currently, hospitalists who choose to recertify using the focused practice in hospital medicine maintenance of certification (MOC) program through the American Board of Internal Medicine have the option of substituting a teamwork module for 1 of the 2 required practice improvement modules, according to Dr. Jeffrey G. Wiese, past president of the Society of Hospital Medicine and associate dean for graduate medical education at Tulane University in New Orleans.
"The eventual goal will be to have the teamwork module a required part of the hospital medicine MOC process," he said. "We are hoping this will occur in 2014."
Hospitalists are dedicated to team-based care and the change reflects that focus, Dr. Wiese said.
Dr. Wiese, who spoke about the MOC process during the SHM annual meeting, addressed some of the confusion around the differences between MOC for hospital medicine vs. standard internal medicine. "This is not as confusing as it may first look," Dr. Wiese said.
Requirements for the two MOC pathways are very similar, he said. For instance, both pathways have the same four elements, and beginning this summer, both pathways will have the same renewal frequency for the various required elements.
Both pathways share the requirement that physicians be board certified and have a valid license to practice medicine. Physicians in the hospital medicine pathway are also required to attest to being a hospitalist by meeting a threshold of inpatient encounters and to be Advanced Cardiac Life Support (ACLS) certified.
The second element is the secure examination. Physicians in both pathways must take a secure exam every 10 years, but the content on the two exams is different. Dr. Wiese said hospitalists will find that the hospital medicine exam is "focused on content that you actually use as a hospitalist."
The third element is the self-evaluation requirement. The standard internal medicine MOC requires physicians to complete 1 practice improvement module every 5 years. The hospital medicine MOC currently requires completion of 2 practice improvement modules every 5 years, with the option of using the team-based module as a substitute for one of the practice improvement modules. The eventual goal, anticipated for 2014, is that the hospital medicine MOC pathway will require completion of the team-based module, and the practice improvement requirement will drop from two to one, making the requirements for practice improvement the same in both the hospitalist and internal medicine MOC pathways.
Currently, physicians in the hospital medicine focused practice MOC pathway are also required to complete one less medical knowledge module than their counterparts in the standard internal medicine MOC pathway.
Hospitalists have many good reasons to pay attention to the MOC process, said Dr. Patrick J. Torcson, chair of SHM’s Performance Measurement and Reporting Committee. In addition to the professional satisfaction that comes with being up-to-date on medical knowledge, payers are beginning to recognize the process. Medicare is offering a 0.5% bonus payment on allowable Medicare charges for physicians who participate in the MOC process. "For the typical hospitalist, that’s about $700 over the 10-year cycle, allowing them to defray part of the cost of the MOC or continuing medical education," said Dr. Torcson, who is the director of hospital medicine at St. Tammany Parish Hospital in Covington, La.
"I think it’s really the right thing to do for our developing specialty," he said. "We really have to distinguish ourselves and there’s really no better brand distinction than a professional designation based on board certification and maintenance of certification."
recertify, MOC, American Board of Internal Medicine, Dr. Jeffrey G. Wiese, the Society of Hospital Medicine,
SAN DIEGO – Hospitalists undergoing maintenance of certification may soon be required to complete a module focused on team-based care, but be allowed drop one requirement related to practice improvement.
Currently, hospitalists who choose to recertify using the focused practice in hospital medicine maintenance of certification (MOC) program through the American Board of Internal Medicine have the option of substituting a teamwork module for 1 of the 2 required practice improvement modules, according to Dr. Jeffrey G. Wiese, past president of the Society of Hospital Medicine and associate dean for graduate medical education at Tulane University in New Orleans.
"The eventual goal will be to have the teamwork module a required part of the hospital medicine MOC process," he said. "We are hoping this will occur in 2014."
Hospitalists are dedicated to team-based care and the change reflects that focus, Dr. Wiese said.
Dr. Wiese, who spoke about the MOC process during the SHM annual meeting, addressed some of the confusion around the differences between MOC for hospital medicine vs. standard internal medicine. "This is not as confusing as it may first look," Dr. Wiese said.
Requirements for the two MOC pathways are very similar, he said. For instance, both pathways have the same four elements, and beginning this summer, both pathways will have the same renewal frequency for the various required elements.
Both pathways share the requirement that physicians be board certified and have a valid license to practice medicine. Physicians in the hospital medicine pathway are also required to attest to being a hospitalist by meeting a threshold of inpatient encounters and to be Advanced Cardiac Life Support (ACLS) certified.
The second element is the secure examination. Physicians in both pathways must take a secure exam every 10 years, but the content on the two exams is different. Dr. Wiese said hospitalists will find that the hospital medicine exam is "focused on content that you actually use as a hospitalist."
The third element is the self-evaluation requirement. The standard internal medicine MOC requires physicians to complete 1 practice improvement module every 5 years. The hospital medicine MOC currently requires completion of 2 practice improvement modules every 5 years, with the option of using the team-based module as a substitute for one of the practice improvement modules. The eventual goal, anticipated for 2014, is that the hospital medicine MOC pathway will require completion of the team-based module, and the practice improvement requirement will drop from two to one, making the requirements for practice improvement the same in both the hospitalist and internal medicine MOC pathways.
Currently, physicians in the hospital medicine focused practice MOC pathway are also required to complete one less medical knowledge module than their counterparts in the standard internal medicine MOC pathway.
Hospitalists have many good reasons to pay attention to the MOC process, said Dr. Patrick J. Torcson, chair of SHM’s Performance Measurement and Reporting Committee. In addition to the professional satisfaction that comes with being up-to-date on medical knowledge, payers are beginning to recognize the process. Medicare is offering a 0.5% bonus payment on allowable Medicare charges for physicians who participate in the MOC process. "For the typical hospitalist, that’s about $700 over the 10-year cycle, allowing them to defray part of the cost of the MOC or continuing medical education," said Dr. Torcson, who is the director of hospital medicine at St. Tammany Parish Hospital in Covington, La.
"I think it’s really the right thing to do for our developing specialty," he said. "We really have to distinguish ourselves and there’s really no better brand distinction than a professional designation based on board certification and maintenance of certification."
SAN DIEGO – Hospitalists undergoing maintenance of certification may soon be required to complete a module focused on team-based care, but be allowed drop one requirement related to practice improvement.
Currently, hospitalists who choose to recertify using the focused practice in hospital medicine maintenance of certification (MOC) program through the American Board of Internal Medicine have the option of substituting a teamwork module for 1 of the 2 required practice improvement modules, according to Dr. Jeffrey G. Wiese, past president of the Society of Hospital Medicine and associate dean for graduate medical education at Tulane University in New Orleans.
"The eventual goal will be to have the teamwork module a required part of the hospital medicine MOC process," he said. "We are hoping this will occur in 2014."
Hospitalists are dedicated to team-based care and the change reflects that focus, Dr. Wiese said.
Dr. Wiese, who spoke about the MOC process during the SHM annual meeting, addressed some of the confusion around the differences between MOC for hospital medicine vs. standard internal medicine. "This is not as confusing as it may first look," Dr. Wiese said.
Requirements for the two MOC pathways are very similar, he said. For instance, both pathways have the same four elements, and beginning this summer, both pathways will have the same renewal frequency for the various required elements.
Both pathways share the requirement that physicians be board certified and have a valid license to practice medicine. Physicians in the hospital medicine pathway are also required to attest to being a hospitalist by meeting a threshold of inpatient encounters and to be Advanced Cardiac Life Support (ACLS) certified.
The second element is the secure examination. Physicians in both pathways must take a secure exam every 10 years, but the content on the two exams is different. Dr. Wiese said hospitalists will find that the hospital medicine exam is "focused on content that you actually use as a hospitalist."
The third element is the self-evaluation requirement. The standard internal medicine MOC requires physicians to complete 1 practice improvement module every 5 years. The hospital medicine MOC currently requires completion of 2 practice improvement modules every 5 years, with the option of using the team-based module as a substitute for one of the practice improvement modules. The eventual goal, anticipated for 2014, is that the hospital medicine MOC pathway will require completion of the team-based module, and the practice improvement requirement will drop from two to one, making the requirements for practice improvement the same in both the hospitalist and internal medicine MOC pathways.
Currently, physicians in the hospital medicine focused practice MOC pathway are also required to complete one less medical knowledge module than their counterparts in the standard internal medicine MOC pathway.
Hospitalists have many good reasons to pay attention to the MOC process, said Dr. Patrick J. Torcson, chair of SHM’s Performance Measurement and Reporting Committee. In addition to the professional satisfaction that comes with being up-to-date on medical knowledge, payers are beginning to recognize the process. Medicare is offering a 0.5% bonus payment on allowable Medicare charges for physicians who participate in the MOC process. "For the typical hospitalist, that’s about $700 over the 10-year cycle, allowing them to defray part of the cost of the MOC or continuing medical education," said Dr. Torcson, who is the director of hospital medicine at St. Tammany Parish Hospital in Covington, La.
"I think it’s really the right thing to do for our developing specialty," he said. "We really have to distinguish ourselves and there’s really no better brand distinction than a professional designation based on board certification and maintenance of certification."
recertify, MOC, American Board of Internal Medicine, Dr. Jeffrey G. Wiese, the Society of Hospital Medicine,
recertify, MOC, American Board of Internal Medicine, Dr. Jeffrey G. Wiese, the Society of Hospital Medicine,
FROM THE ANNUAL MEETING OF THE SOCIETY OF HOSPITAL MEDICINE
Experts Weigh In on Decoding Care Transitions
SAN DIEGO – Thinking through how to better prepare patients to go from the hospital to home occupies an increasing amount of time for the typical hospitalist. Aside from the fact that transitions of care are a core competency in the specialty, Medicare is also turning up the heat with its plan to impose financial penalties on hospitals with excessive readmission rates. Those penalties will take effect this October.
At the annual meeting of the Society of Hospital Medicine, Dr. Mark V. Williams, chief of hospital medicine at Northwestern University in Chicago and principal investigator for Project BOOST (Better Outcomes for Older Adults Through Safe Transitions) joined Dr. Eric A. Coleman, professor of medicine and director of the Care Transitions Program at the University of Colorado in Denver, for a freewheeling question-and-answer session.
The hourlong exchange covered everything from the state of research in the field to how to avoid Medicare penalties.
One take-home message from the discussion is that reducing preventable readmissions will be a focus for virtually every hospitalist, even though some of the key research questions that could help improve care transitions are still unanswered. For instance, there have been many prediction scales that have looked at risk for readmission. But the scales don’t work very well in practice.
"[The scales] come out to be okay, but not great," said Dr. Coleman, who has tried to develop a more reliable risk prediction tool as part of his own research efforts.
But the real lesson from the lukewarm success of existing risk prediction tools is that researchers and clinicians may be focusing on the wrong variables. "I’m not exactly sure that what we think we’re after is what we’re after," Dr. Coleman said.
Dr. Coleman said when he speaks to patients at the bedside about why they think they have been readmitted to the hospital multiple times, it’s not necessarily about organ systems function. And that supports the findings of some research he was involved in that found that factors such as cognition and literacy were the biggest predictors in whether patients would actually follow through on their discharge instructions. "These are not necessarily things that we routinely collect and screen for," he said.
Dr. Williams agreed that there’s a need to routinely collect more data, beyond whether or not patients have specific comorbidities. Quantitative data on the patient’s socioeconomic status, health literacy level, occupation status, and social support could shed a lot of light on whether they will be capable of following through on their care when they return home. Dr. Williams said one of the hospitalists who has been involved in Project BOOST once told him that when he sees a patient he always asks, "Do you have a daughter?" And if the patient says yes, he can relax because now he knows that there’s great social support at home.
While improving care transitions is good for patient care, it will soon also be good business for hospitals. Under a new readmission program from the Centers for Medicare and Medicaid Services, Medicare officials will cut payments to hospitals whose readmission rates for acute myocardial infarction, congestive heart failure, and pneumonia are considered too high. But what many hospitalists may not appreciate, Dr. Williams said, is that CMS officials will be basing that determination on the hospital’s 3-year average performance from fiscal year 2009 through 2011. The next year, the CMS will use data collected from fiscal years 2010-2012.
While reducing preventable readmissions is now a focus for most hospitals, they really should have started that work years ago, Dr. Williams said.
"We need to have been working on this for awhile because the clock is ticking; data are being collected," he said. "And very importantly, it will take awhile to improve those numbers unless you’re able to make staggering changes right off the bat because it’s a 3-year rolling average."
Hospitals that have been doing well for the past 3 years are probably in pretty good shape, Dr. Williams said. Those that have been doing poorly will have to play catch up. "I think right now every site ought to be looking at their care transitions process," he said.
Hospitalists can try to partner with physicians and other health care providers in the community, said Dr. Coleman.
"When you look at some of the more successful collaborations going on across the country that have been able to significantly reduce their readmission rates and subsequent admission rate as well, it is because of these developing partnerships and realizing that hospitals can’t do it all on their own," he said.
Dr. Coleman encouraged hospitalists to learn more about the Community-Based Care Transition Program, an initiative created under the Affordable Care Act, which provides grants for hospitals to collaborate with community partners. "I think that this is an opportunity for not only hospitals, but hospitalists, to begin taking an even greater leadership role" in what the hospitals can do and what hospitals can do very well with their partners, he said.
Dr. Williams agreed that partnerships with community physicians are a big part of the future of hospital medicine. But payers will have to step up and begin to reimburse physicians for doing that work, he said. For instance, insurers could pay hospitalists to call primary care physicians. "It would be fascinating to have some experiments with this to see how they impact continuity of care and utilization overall," Dr. Williams said.
SAN DIEGO – Thinking through how to better prepare patients to go from the hospital to home occupies an increasing amount of time for the typical hospitalist. Aside from the fact that transitions of care are a core competency in the specialty, Medicare is also turning up the heat with its plan to impose financial penalties on hospitals with excessive readmission rates. Those penalties will take effect this October.
At the annual meeting of the Society of Hospital Medicine, Dr. Mark V. Williams, chief of hospital medicine at Northwestern University in Chicago and principal investigator for Project BOOST (Better Outcomes for Older Adults Through Safe Transitions) joined Dr. Eric A. Coleman, professor of medicine and director of the Care Transitions Program at the University of Colorado in Denver, for a freewheeling question-and-answer session.
The hourlong exchange covered everything from the state of research in the field to how to avoid Medicare penalties.
One take-home message from the discussion is that reducing preventable readmissions will be a focus for virtually every hospitalist, even though some of the key research questions that could help improve care transitions are still unanswered. For instance, there have been many prediction scales that have looked at risk for readmission. But the scales don’t work very well in practice.
"[The scales] come out to be okay, but not great," said Dr. Coleman, who has tried to develop a more reliable risk prediction tool as part of his own research efforts.
But the real lesson from the lukewarm success of existing risk prediction tools is that researchers and clinicians may be focusing on the wrong variables. "I’m not exactly sure that what we think we’re after is what we’re after," Dr. Coleman said.
Dr. Coleman said when he speaks to patients at the bedside about why they think they have been readmitted to the hospital multiple times, it’s not necessarily about organ systems function. And that supports the findings of some research he was involved in that found that factors such as cognition and literacy were the biggest predictors in whether patients would actually follow through on their discharge instructions. "These are not necessarily things that we routinely collect and screen for," he said.
Dr. Williams agreed that there’s a need to routinely collect more data, beyond whether or not patients have specific comorbidities. Quantitative data on the patient’s socioeconomic status, health literacy level, occupation status, and social support could shed a lot of light on whether they will be capable of following through on their care when they return home. Dr. Williams said one of the hospitalists who has been involved in Project BOOST once told him that when he sees a patient he always asks, "Do you have a daughter?" And if the patient says yes, he can relax because now he knows that there’s great social support at home.
While improving care transitions is good for patient care, it will soon also be good business for hospitals. Under a new readmission program from the Centers for Medicare and Medicaid Services, Medicare officials will cut payments to hospitals whose readmission rates for acute myocardial infarction, congestive heart failure, and pneumonia are considered too high. But what many hospitalists may not appreciate, Dr. Williams said, is that CMS officials will be basing that determination on the hospital’s 3-year average performance from fiscal year 2009 through 2011. The next year, the CMS will use data collected from fiscal years 2010-2012.
While reducing preventable readmissions is now a focus for most hospitals, they really should have started that work years ago, Dr. Williams said.
"We need to have been working on this for awhile because the clock is ticking; data are being collected," he said. "And very importantly, it will take awhile to improve those numbers unless you’re able to make staggering changes right off the bat because it’s a 3-year rolling average."
Hospitals that have been doing well for the past 3 years are probably in pretty good shape, Dr. Williams said. Those that have been doing poorly will have to play catch up. "I think right now every site ought to be looking at their care transitions process," he said.
Hospitalists can try to partner with physicians and other health care providers in the community, said Dr. Coleman.
"When you look at some of the more successful collaborations going on across the country that have been able to significantly reduce their readmission rates and subsequent admission rate as well, it is because of these developing partnerships and realizing that hospitals can’t do it all on their own," he said.
Dr. Coleman encouraged hospitalists to learn more about the Community-Based Care Transition Program, an initiative created under the Affordable Care Act, which provides grants for hospitals to collaborate with community partners. "I think that this is an opportunity for not only hospitals, but hospitalists, to begin taking an even greater leadership role" in what the hospitals can do and what hospitals can do very well with their partners, he said.
Dr. Williams agreed that partnerships with community physicians are a big part of the future of hospital medicine. But payers will have to step up and begin to reimburse physicians for doing that work, he said. For instance, insurers could pay hospitalists to call primary care physicians. "It would be fascinating to have some experiments with this to see how they impact continuity of care and utilization overall," Dr. Williams said.
SAN DIEGO – Thinking through how to better prepare patients to go from the hospital to home occupies an increasing amount of time for the typical hospitalist. Aside from the fact that transitions of care are a core competency in the specialty, Medicare is also turning up the heat with its plan to impose financial penalties on hospitals with excessive readmission rates. Those penalties will take effect this October.
At the annual meeting of the Society of Hospital Medicine, Dr. Mark V. Williams, chief of hospital medicine at Northwestern University in Chicago and principal investigator for Project BOOST (Better Outcomes for Older Adults Through Safe Transitions) joined Dr. Eric A. Coleman, professor of medicine and director of the Care Transitions Program at the University of Colorado in Denver, for a freewheeling question-and-answer session.
The hourlong exchange covered everything from the state of research in the field to how to avoid Medicare penalties.
One take-home message from the discussion is that reducing preventable readmissions will be a focus for virtually every hospitalist, even though some of the key research questions that could help improve care transitions are still unanswered. For instance, there have been many prediction scales that have looked at risk for readmission. But the scales don’t work very well in practice.
"[The scales] come out to be okay, but not great," said Dr. Coleman, who has tried to develop a more reliable risk prediction tool as part of his own research efforts.
But the real lesson from the lukewarm success of existing risk prediction tools is that researchers and clinicians may be focusing on the wrong variables. "I’m not exactly sure that what we think we’re after is what we’re after," Dr. Coleman said.
Dr. Coleman said when he speaks to patients at the bedside about why they think they have been readmitted to the hospital multiple times, it’s not necessarily about organ systems function. And that supports the findings of some research he was involved in that found that factors such as cognition and literacy were the biggest predictors in whether patients would actually follow through on their discharge instructions. "These are not necessarily things that we routinely collect and screen for," he said.
Dr. Williams agreed that there’s a need to routinely collect more data, beyond whether or not patients have specific comorbidities. Quantitative data on the patient’s socioeconomic status, health literacy level, occupation status, and social support could shed a lot of light on whether they will be capable of following through on their care when they return home. Dr. Williams said one of the hospitalists who has been involved in Project BOOST once told him that when he sees a patient he always asks, "Do you have a daughter?" And if the patient says yes, he can relax because now he knows that there’s great social support at home.
While improving care transitions is good for patient care, it will soon also be good business for hospitals. Under a new readmission program from the Centers for Medicare and Medicaid Services, Medicare officials will cut payments to hospitals whose readmission rates for acute myocardial infarction, congestive heart failure, and pneumonia are considered too high. But what many hospitalists may not appreciate, Dr. Williams said, is that CMS officials will be basing that determination on the hospital’s 3-year average performance from fiscal year 2009 through 2011. The next year, the CMS will use data collected from fiscal years 2010-2012.
While reducing preventable readmissions is now a focus for most hospitals, they really should have started that work years ago, Dr. Williams said.
"We need to have been working on this for awhile because the clock is ticking; data are being collected," he said. "And very importantly, it will take awhile to improve those numbers unless you’re able to make staggering changes right off the bat because it’s a 3-year rolling average."
Hospitals that have been doing well for the past 3 years are probably in pretty good shape, Dr. Williams said. Those that have been doing poorly will have to play catch up. "I think right now every site ought to be looking at their care transitions process," he said.
Hospitalists can try to partner with physicians and other health care providers in the community, said Dr. Coleman.
"When you look at some of the more successful collaborations going on across the country that have been able to significantly reduce their readmission rates and subsequent admission rate as well, it is because of these developing partnerships and realizing that hospitals can’t do it all on their own," he said.
Dr. Coleman encouraged hospitalists to learn more about the Community-Based Care Transition Program, an initiative created under the Affordable Care Act, which provides grants for hospitals to collaborate with community partners. "I think that this is an opportunity for not only hospitals, but hospitalists, to begin taking an even greater leadership role" in what the hospitals can do and what hospitals can do very well with their partners, he said.
Dr. Williams agreed that partnerships with community physicians are a big part of the future of hospital medicine. But payers will have to step up and begin to reimburse physicians for doing that work, he said. For instance, insurers could pay hospitalists to call primary care physicians. "It would be fascinating to have some experiments with this to see how they impact continuity of care and utilization overall," Dr. Williams said.
FROM THE ANNUAL MEETING OF THE SOCIETY OF HOSPITAL MEDICINE
HHS Calls for 1-Year Delay for ICD-10
The federal government plans to delay for 1 year a requirement that doctors and other health care providers begin using the ICD-10 standard for diagnosis and procedure codes. Physicians will now have until Oct. 1, 2014, to come into compliance with the standard.
Federal officials are also hoping to ease administrative burdens by requiring health plans to use a single, uniform identifier for all their transactions. Both changes were addressed in a proposed rule released on April 9.
Health and Human Services Secretary Kathleen Sebelius announced earlier this year that the health care community would get more time to get up to speed on ICD-10 (formally known as the International Classification of Diseases, 10th Revision), but the proposed rule firms up that commitment.
Many physician organizations, including the American Medical Association, complained to the HHS and a member of Congress, arguing that the implementation of the ICD-10 codes will create a significant burden. And it would carry a hefty price tag – close to $100,000 for smaller practices and more than $2 million for large ones, according to the AMA.
Part of the problem, according to the HHS, is that medical practices are having trouble meeting the compliance deadline for a necessary prerequisite for ICD-10, the Associated Standard Committee’s X12 Version 5010 standards (Version 5010) for electronic health care transactions. Moving the compliance deadline for the so-called 5010 standard from October 2013 to October 2014 will give physicians more time to prepare and test their systems, the HHS wrote in its proposed rule.
The proposed rule also outlines a related measure: a plan to require health insurers to adopt a standard national unique health plan identifier, or HPID. Currently, health plans use multiple identifiers of differing lengths and formats. The vagaries of these identifiers can cause improper routing of transactions, difficulty in determining patient eligibility, and other claims processing errors. Health insurers must begin to use the standardized HPID by Oct. 1, 2012.
The use of a unique identifier will allow medical practices to make greater use of automation in claims processing, according to the proposed rule, in turn saving time and money. And, cleaner claims with fewer errors should compound the savings. HHS officials estimate that over 10 years, the return on investment for the entire health care industry will be between $700 million and $4.6 billion. The adoption of the unique health plan identifier is one of a series of regulations mandated by the Affordable Care Act and aimed at simplifying health care administrative transactions.
The federal government plans to delay for 1 year a requirement that doctors and other health care providers begin using the ICD-10 standard for diagnosis and procedure codes. Physicians will now have until Oct. 1, 2014, to come into compliance with the standard.
Federal officials are also hoping to ease administrative burdens by requiring health plans to use a single, uniform identifier for all their transactions. Both changes were addressed in a proposed rule released on April 9.
Health and Human Services Secretary Kathleen Sebelius announced earlier this year that the health care community would get more time to get up to speed on ICD-10 (formally known as the International Classification of Diseases, 10th Revision), but the proposed rule firms up that commitment.
Many physician organizations, including the American Medical Association, complained to the HHS and a member of Congress, arguing that the implementation of the ICD-10 codes will create a significant burden. And it would carry a hefty price tag – close to $100,000 for smaller practices and more than $2 million for large ones, according to the AMA.
Part of the problem, according to the HHS, is that medical practices are having trouble meeting the compliance deadline for a necessary prerequisite for ICD-10, the Associated Standard Committee’s X12 Version 5010 standards (Version 5010) for electronic health care transactions. Moving the compliance deadline for the so-called 5010 standard from October 2013 to October 2014 will give physicians more time to prepare and test their systems, the HHS wrote in its proposed rule.
The proposed rule also outlines a related measure: a plan to require health insurers to adopt a standard national unique health plan identifier, or HPID. Currently, health plans use multiple identifiers of differing lengths and formats. The vagaries of these identifiers can cause improper routing of transactions, difficulty in determining patient eligibility, and other claims processing errors. Health insurers must begin to use the standardized HPID by Oct. 1, 2012.
The use of a unique identifier will allow medical practices to make greater use of automation in claims processing, according to the proposed rule, in turn saving time and money. And, cleaner claims with fewer errors should compound the savings. HHS officials estimate that over 10 years, the return on investment for the entire health care industry will be between $700 million and $4.6 billion. The adoption of the unique health plan identifier is one of a series of regulations mandated by the Affordable Care Act and aimed at simplifying health care administrative transactions.
The federal government plans to delay for 1 year a requirement that doctors and other health care providers begin using the ICD-10 standard for diagnosis and procedure codes. Physicians will now have until Oct. 1, 2014, to come into compliance with the standard.
Federal officials are also hoping to ease administrative burdens by requiring health plans to use a single, uniform identifier for all their transactions. Both changes were addressed in a proposed rule released on April 9.
Health and Human Services Secretary Kathleen Sebelius announced earlier this year that the health care community would get more time to get up to speed on ICD-10 (formally known as the International Classification of Diseases, 10th Revision), but the proposed rule firms up that commitment.
Many physician organizations, including the American Medical Association, complained to the HHS and a member of Congress, arguing that the implementation of the ICD-10 codes will create a significant burden. And it would carry a hefty price tag – close to $100,000 for smaller practices and more than $2 million for large ones, according to the AMA.
Part of the problem, according to the HHS, is that medical practices are having trouble meeting the compliance deadline for a necessary prerequisite for ICD-10, the Associated Standard Committee’s X12 Version 5010 standards (Version 5010) for electronic health care transactions. Moving the compliance deadline for the so-called 5010 standard from October 2013 to October 2014 will give physicians more time to prepare and test their systems, the HHS wrote in its proposed rule.
The proposed rule also outlines a related measure: a plan to require health insurers to adopt a standard national unique health plan identifier, or HPID. Currently, health plans use multiple identifiers of differing lengths and formats. The vagaries of these identifiers can cause improper routing of transactions, difficulty in determining patient eligibility, and other claims processing errors. Health insurers must begin to use the standardized HPID by Oct. 1, 2012.
The use of a unique identifier will allow medical practices to make greater use of automation in claims processing, according to the proposed rule, in turn saving time and money. And, cleaner claims with fewer errors should compound the savings. HHS officials estimate that over 10 years, the return on investment for the entire health care industry will be between $700 million and $4.6 billion. The adoption of the unique health plan identifier is one of a series of regulations mandated by the Affordable Care Act and aimed at simplifying health care administrative transactions.
Leaders: Nudging Medicine's Glass Ceiling
When Dr. Patience Reich, an assistant professor of medicine at Wake Forest School of Medicine in Winston-Salem, N.C., came out of residency training, she knew she still had a lot to learn about the finances of a hospital medicine group, navigating hospital politics, and becoming a leader. So she asked for help, finding mentors in and out of hospital medicine and becoming one of the first classes of physicians to go through the Society of Hospital Medicine’s Leadership Academy.
Dr. Reich was able to achieve many of her career goals, serving as medical director of the Wake Forest School of Medicine’s hospital medicine program for several years and national chair of SHM’s Leadership Academy from 2009 to 2011.
Now she’s turning her passion for training hospital leaders toward the specialty’s gender gap. Following the release of survey data showing that women in hospital medicine make less than their male counterparts, Dr. Reich cochaired a session at the SHM annual meeting to discuss some of the reasons behind the pay inequality.
In an interview with Hospitalist News, Dr. Reich explained some of the challenges faced by women in the specialty and offered some advice from her own experience.
HN: Is the pay gap between men and women in hospital medicine something unique to the specialty or is it something that we hear about in other industries?
DR. REICH: It’s not unique to the specialty. I think the assumption was that hospital medicine was different. When we got some of the data back and found that it was not, that was rather disappointing.
HN: Why are so few women rising to leadership positions in hospital medicine?
DR. REICH: I think it’s a combination of factors. We are a young field and many people are in their child-bearing, child-raising years. It’s just very difficult to balance everything. And as long as women continue to do the majority of the work at home, that problem remains.
The lack of mentors is another big one. Since many of us didn’t have any training when we started out, the early medical directors and leaders were just struggling to keep their own heads above water. We had so many unique challenges, there was no time to really consider or set up mentoring programs.
HN: Do you feel like you’ve experienced gender bias in your career as a hospitalist?
DR. REICH: There was a time when I took some time off and did some locum tenens work to keep my skills from getting rusty. I worked as a hospitalist for a large internal medicine group in a medium-size community hospital, along with another woman and a man. The man would sometimes not show up for his assigned shifts. One weekend, I was forced to stay there because he was a no-show. It turned out he didn’t even have an emergency, he was just at the beach! Later, we discovered that he was getting paid a lot more than we were. By all metrics, we were doing a lot better job. I went to the locum company and asked them what this was about. They had no response. That was the last time I worked there.
HN: What advice would you give women who are aspiring to leadership positions in hospital medicine?
DR. REICH: You have to indicate that you are interested in leadership by taking on some task, even if it’s something small. One of the things about gender bias is that men tend to be judged on their potential, while women tend to be judged on their accomplishments. So, if you aspire to leadership, make sure you have some leadership accomplishments.
Women definitely need to seek out mentors. These mentors don’t have to be other women. The majority of my mentors have been men. I have had a specific kind of mentor known as a "sponsor." Sponsors are high ranking and influential in an organization, and the relationship is predicated on power and very focused on career advancement. Sponsors provide access to informal networks and help you get high visibility leadership assignments.
Self-promotion is often necessary in leadership, but frequently is seen as inappropriate when done by women. Sponsors can help you get around that by tooting your horn for you.
Take us to your leader. Nominate a hospitalist whose work inspires you. E-mail suggestions to [email protected]. Read previous columns at ehospitalistnews.com.
When Dr. Patience Reich, an assistant professor of medicine at Wake Forest School of Medicine in Winston-Salem, N.C., came out of residency training, she knew she still had a lot to learn about the finances of a hospital medicine group, navigating hospital politics, and becoming a leader. So she asked for help, finding mentors in and out of hospital medicine and becoming one of the first classes of physicians to go through the Society of Hospital Medicine’s Leadership Academy.
Dr. Reich was able to achieve many of her career goals, serving as medical director of the Wake Forest School of Medicine’s hospital medicine program for several years and national chair of SHM’s Leadership Academy from 2009 to 2011.
Now she’s turning her passion for training hospital leaders toward the specialty’s gender gap. Following the release of survey data showing that women in hospital medicine make less than their male counterparts, Dr. Reich cochaired a session at the SHM annual meeting to discuss some of the reasons behind the pay inequality.
In an interview with Hospitalist News, Dr. Reich explained some of the challenges faced by women in the specialty and offered some advice from her own experience.
HN: Is the pay gap between men and women in hospital medicine something unique to the specialty or is it something that we hear about in other industries?
DR. REICH: It’s not unique to the specialty. I think the assumption was that hospital medicine was different. When we got some of the data back and found that it was not, that was rather disappointing.
HN: Why are so few women rising to leadership positions in hospital medicine?
DR. REICH: I think it’s a combination of factors. We are a young field and many people are in their child-bearing, child-raising years. It’s just very difficult to balance everything. And as long as women continue to do the majority of the work at home, that problem remains.
The lack of mentors is another big one. Since many of us didn’t have any training when we started out, the early medical directors and leaders were just struggling to keep their own heads above water. We had so many unique challenges, there was no time to really consider or set up mentoring programs.
HN: Do you feel like you’ve experienced gender bias in your career as a hospitalist?
DR. REICH: There was a time when I took some time off and did some locum tenens work to keep my skills from getting rusty. I worked as a hospitalist for a large internal medicine group in a medium-size community hospital, along with another woman and a man. The man would sometimes not show up for his assigned shifts. One weekend, I was forced to stay there because he was a no-show. It turned out he didn’t even have an emergency, he was just at the beach! Later, we discovered that he was getting paid a lot more than we were. By all metrics, we were doing a lot better job. I went to the locum company and asked them what this was about. They had no response. That was the last time I worked there.
HN: What advice would you give women who are aspiring to leadership positions in hospital medicine?
DR. REICH: You have to indicate that you are interested in leadership by taking on some task, even if it’s something small. One of the things about gender bias is that men tend to be judged on their potential, while women tend to be judged on their accomplishments. So, if you aspire to leadership, make sure you have some leadership accomplishments.
Women definitely need to seek out mentors. These mentors don’t have to be other women. The majority of my mentors have been men. I have had a specific kind of mentor known as a "sponsor." Sponsors are high ranking and influential in an organization, and the relationship is predicated on power and very focused on career advancement. Sponsors provide access to informal networks and help you get high visibility leadership assignments.
Self-promotion is often necessary in leadership, but frequently is seen as inappropriate when done by women. Sponsors can help you get around that by tooting your horn for you.
Take us to your leader. Nominate a hospitalist whose work inspires you. E-mail suggestions to [email protected]. Read previous columns at ehospitalistnews.com.
When Dr. Patience Reich, an assistant professor of medicine at Wake Forest School of Medicine in Winston-Salem, N.C., came out of residency training, she knew she still had a lot to learn about the finances of a hospital medicine group, navigating hospital politics, and becoming a leader. So she asked for help, finding mentors in and out of hospital medicine and becoming one of the first classes of physicians to go through the Society of Hospital Medicine’s Leadership Academy.
Dr. Reich was able to achieve many of her career goals, serving as medical director of the Wake Forest School of Medicine’s hospital medicine program for several years and national chair of SHM’s Leadership Academy from 2009 to 2011.
Now she’s turning her passion for training hospital leaders toward the specialty’s gender gap. Following the release of survey data showing that women in hospital medicine make less than their male counterparts, Dr. Reich cochaired a session at the SHM annual meeting to discuss some of the reasons behind the pay inequality.
In an interview with Hospitalist News, Dr. Reich explained some of the challenges faced by women in the specialty and offered some advice from her own experience.
HN: Is the pay gap between men and women in hospital medicine something unique to the specialty or is it something that we hear about in other industries?
DR. REICH: It’s not unique to the specialty. I think the assumption was that hospital medicine was different. When we got some of the data back and found that it was not, that was rather disappointing.
HN: Why are so few women rising to leadership positions in hospital medicine?
DR. REICH: I think it’s a combination of factors. We are a young field and many people are in their child-bearing, child-raising years. It’s just very difficult to balance everything. And as long as women continue to do the majority of the work at home, that problem remains.
The lack of mentors is another big one. Since many of us didn’t have any training when we started out, the early medical directors and leaders were just struggling to keep their own heads above water. We had so many unique challenges, there was no time to really consider or set up mentoring programs.
HN: Do you feel like you’ve experienced gender bias in your career as a hospitalist?
DR. REICH: There was a time when I took some time off and did some locum tenens work to keep my skills from getting rusty. I worked as a hospitalist for a large internal medicine group in a medium-size community hospital, along with another woman and a man. The man would sometimes not show up for his assigned shifts. One weekend, I was forced to stay there because he was a no-show. It turned out he didn’t even have an emergency, he was just at the beach! Later, we discovered that he was getting paid a lot more than we were. By all metrics, we were doing a lot better job. I went to the locum company and asked them what this was about. They had no response. That was the last time I worked there.
HN: What advice would you give women who are aspiring to leadership positions in hospital medicine?
DR. REICH: You have to indicate that you are interested in leadership by taking on some task, even if it’s something small. One of the things about gender bias is that men tend to be judged on their potential, while women tend to be judged on their accomplishments. So, if you aspire to leadership, make sure you have some leadership accomplishments.
Women definitely need to seek out mentors. These mentors don’t have to be other women. The majority of my mentors have been men. I have had a specific kind of mentor known as a "sponsor." Sponsors are high ranking and influential in an organization, and the relationship is predicated on power and very focused on career advancement. Sponsors provide access to informal networks and help you get high visibility leadership assignments.
Self-promotion is often necessary in leadership, but frequently is seen as inappropriate when done by women. Sponsors can help you get around that by tooting your horn for you.
Take us to your leader. Nominate a hospitalist whose work inspires you. E-mail suggestions to [email protected]. Read previous columns at ehospitalistnews.com.
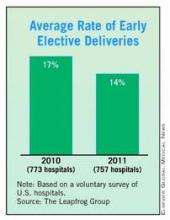
Baby Can Wait: Hospitals Curb Early Elective Deliveries
At North Shore Medical Center in Salem, Mass., interest in reducing elective deliveries before 39 weeks’ gestation started with a newborn with respiratory problems.
A few years ago, when a patient was induced at about 38 weeks’ gestation, the newborn ended up having significant respiratory complications and had to be transferred to a tertiary care center, said Dr. Allyson L. Preston, chair of obstetrics and gynecology there.
Although the baby recovered and did fine, a review of the case led to a deeper examination of the data on elective deliveries at 37-39 weeks. The result was a growing realization that early elective deliveries weren’t as benign as previously thought, Dr. Preston said in an interview.
After hearing about the success that Intermountain Healthcare in Utah had in reducing early elective deliveries, Dr. Preston worked with her physician and nursing colleagues in the department of ob.gyn. to develop a policy not to schedule either elective inductions or cesarean sections if the patient was at less than 39 weeks’ gestation, unless there was a medical or obstetrical indication that early delivery was necessary.
In early 2010, the hospital staff sent out new booking forms to area obstetrical offices. The new forms required physicians to note the indications for early delivery and the gestational age. The charge nurses at North Shore Medical Center were given the authority not to schedule procedures if a patient was at less than 39 weeks’ gestation and the physician did not include an accepted medical or obstetrical indication for delivery. Physicians who took exception to the policy were referred to Dr. Preston. For the most part, physicians have eagerly adopted the policy, she said.
"I think people realized we were serious about it," Dr. Preston said. "They realized it was the right thing to do."
The results since the policy was implemented have been fairly dramatic. Data released earlier this year by the Leapfrog Group show that in 2010, the early elective newborn delivery rate was 31.6% at North Shore, compared with just 1.6% in 2011. That means that among deliveries done at 37-39 weeks, more than 98% are now medically indicated, according to Dr. Preston.
Other hospitals around the country are experiencing similar success, according to data from the Leapfrog Group’s annual hospital survey. The survey of 757 hospitals found that 39% of reporting hospitals had early elective delivery rates of 5% or less in 2011, compared with 30% the previous year. The rate of elective deliveries represents the percentage of nonmedically indicated births at 37-39 weeks’ gestation delivered by caesarean section or induction. The average rate for early elective deliveries among these hospitals dropped from 17% to 14% between 2010 and 2011.
Despite the progress already being made to curb early deliveries in some hospitals, there was still a wide variation among the hospitals in the Leapfrog survey, with early elective delivery rates ranging from a low of less than 5% to more than 40%, according to the 2011 data.
The Leapfrog Group (a coalition of public and private purchasers of employee health benefits that works on health care quality improvement) began collecting data on early elective deliveries in 2009 and first publicly reported hospital data in 2010. The group has identified the trend toward early deliveries as both a safety and an economic issue. For instance, women who are induced in weeks 37 and 38 have a higher risk of needing a cesarean section than do women who go into labor on their own. Studies also have linked early elective deliveries to postpartum complications such as hematoma, wound dehiscence, anemia, endometriosis, urinary tract infections, and sepsis, according to the Leapfrog Group.
The Leapfrog Group is not the only organization that has made the reduction of early elective deliveries a priority. For many years, the American College of Obstetricians and Gynecologists has recommended against elective deliveries before 39 weeks. In February, the federal government added its weight to the push to reduce these types of elective deliveries with the launch of its Strong Start initiative. Under the new effort, officials at the Department of Health and Human Services will work with outside partners to test a nationwide awareness campaign on best practices to reduce the rate of early elective deliveries before 39 weeks among all pregnant women. They will also test "enhanced prenatal care" approaches for Medicaid beneficiaries who are at risk for preterm birth.
The latest Leapfrog data are consistent with what other efforts that are aimed at reducing early elective deliveries have been able to show, said Dr. Donald Dudley, professor and vice chair for research in the department of ob.gyn. at the University of Texas Health Sciences Center in San Antonio.
Dr. Dudley also has been active in the March of Dimes Big 5 Prematurity Collaborative, which has been working to reduce preterm births through evidence-based quality improvement projects.
The Big 5 Prematurity Collaborative worked with 25 hospitals in California, Florida, Illinois, New York, and Texas to implement a policy of not performing elective deliveries before 39 weeks unless they are medically indicated. They found that hospitals – regardless of their size or location – were able to rapidly reduce early elective deliveries. The key, Dr. Dudley said, was making physicians and nurses aware of the data on complications from early deliveries.
Dr. Dudley said some physicians are skeptical that those last few weeks make that big a difference. He had been one of them. But once he read through the studies, it became clear that deliveries before 39 weeks were not the safest path for mothers and babies. "You’ll come away saying it is really not a good idea to do elective deliveries at less than 39 weeks," he said.
Once the buy-in from providers is there, making the change is fairly simple and relatively cheap, Dr. Dudley said. Hospitals can take advantage of free toolkits like the one from the California Maternal Quality Care Collaborative and the March of Dimes.
The most costly and time-intensive part of any effort to reduce early elective deliveries is the data collection, he said. Hospitals need to know how they doing at the start, but many community hospitals don’t routinely collect those data.
Dr. Dudley cautioned hospitals to be careful not to push the policy so hard that they discourage physicians from exercising their clinical judgment. "One of the things we have to be really careful about is not to swing the pendulum so far the other way that we disengage our brains."
If there’s a reason for a patient to be delivered at less than 39 weeks, then the delivery is indicated, not elective, he said. And there is a long list of appropriate indications for an early delivery.
For example, Dr. Dudley said he recently had a patient whom he had been monitoring and hoping to deliver after 39 weeks, but at 38 and a half weeks her blood pressure began creeping up enough that it was necessary to do the delivery early. The patient had a history of hypertension, and the risk of waiting outweighed the risks of early induction, he said. "You don’t want to delay acting when there’s an acceptable indication."
Policies aimed at reducing early elective deliveries can introduce a reluctance to induce (either by the physician or the patient) that may result in delaying an induction that is actually indicated, said Dr. William H. Barth Jr., chief of the division of maternal-fetal medicine at Massachusetts General Hospital, Boston, where the early elective newborn delivery rate has declined from 8% to 4% between 2010 and 2011. "Recommending an induction is a complex decision," he said. "The recent push to decrease elective inductions is sort of one more factor in the mix of that complex decision."
But the other issue is simple math, Dr. Barth said. If more inductions are delayed – indicated or not – there will be more stillbirths. He pointed to a retrospective cohort study published last year in Obstetrics & Gynecology that looked at the effect of a policy limiting elective delivery before 39 weeks’ gestation. The researchers found a significant decrease in admissions to the neonatal ICU, but they also detected an increase in stillbirths at 37 and 38 weeks (Obstet. Gynecol. 2011;118:1047-55).
Dr. Barth advised hospitals considering formal policies to reduce early elective deliveries to track not only the number of elective deliveries, but also stillbirths. "Shared decision making between patients and physicians is best conducted with all of the information – good and bad," he said.
At North Shore Medical Center in Salem, Mass., interest in reducing elective deliveries before 39 weeks’ gestation started with a newborn with respiratory problems.
A few years ago, when a patient was induced at about 38 weeks’ gestation, the newborn ended up having significant respiratory complications and had to be transferred to a tertiary care center, said Dr. Allyson L. Preston, chair of obstetrics and gynecology there.
Although the baby recovered and did fine, a review of the case led to a deeper examination of the data on elective deliveries at 37-39 weeks. The result was a growing realization that early elective deliveries weren’t as benign as previously thought, Dr. Preston said in an interview.
After hearing about the success that Intermountain Healthcare in Utah had in reducing early elective deliveries, Dr. Preston worked with her physician and nursing colleagues in the department of ob.gyn. to develop a policy not to schedule either elective inductions or cesarean sections if the patient was at less than 39 weeks’ gestation, unless there was a medical or obstetrical indication that early delivery was necessary.
In early 2010, the hospital staff sent out new booking forms to area obstetrical offices. The new forms required physicians to note the indications for early delivery and the gestational age. The charge nurses at North Shore Medical Center were given the authority not to schedule procedures if a patient was at less than 39 weeks’ gestation and the physician did not include an accepted medical or obstetrical indication for delivery. Physicians who took exception to the policy were referred to Dr. Preston. For the most part, physicians have eagerly adopted the policy, she said.
"I think people realized we were serious about it," Dr. Preston said. "They realized it was the right thing to do."
The results since the policy was implemented have been fairly dramatic. Data released earlier this year by the Leapfrog Group show that in 2010, the early elective newborn delivery rate was 31.6% at North Shore, compared with just 1.6% in 2011. That means that among deliveries done at 37-39 weeks, more than 98% are now medically indicated, according to Dr. Preston.
Other hospitals around the country are experiencing similar success, according to data from the Leapfrog Group’s annual hospital survey. The survey of 757 hospitals found that 39% of reporting hospitals had early elective delivery rates of 5% or less in 2011, compared with 30% the previous year. The rate of elective deliveries represents the percentage of nonmedically indicated births at 37-39 weeks’ gestation delivered by caesarean section or induction. The average rate for early elective deliveries among these hospitals dropped from 17% to 14% between 2010 and 2011.
Despite the progress already being made to curb early deliveries in some hospitals, there was still a wide variation among the hospitals in the Leapfrog survey, with early elective delivery rates ranging from a low of less than 5% to more than 40%, according to the 2011 data.
The Leapfrog Group (a coalition of public and private purchasers of employee health benefits that works on health care quality improvement) began collecting data on early elective deliveries in 2009 and first publicly reported hospital data in 2010. The group has identified the trend toward early deliveries as both a safety and an economic issue. For instance, women who are induced in weeks 37 and 38 have a higher risk of needing a cesarean section than do women who go into labor on their own. Studies also have linked early elective deliveries to postpartum complications such as hematoma, wound dehiscence, anemia, endometriosis, urinary tract infections, and sepsis, according to the Leapfrog Group.
The Leapfrog Group is not the only organization that has made the reduction of early elective deliveries a priority. For many years, the American College of Obstetricians and Gynecologists has recommended against elective deliveries before 39 weeks. In February, the federal government added its weight to the push to reduce these types of elective deliveries with the launch of its Strong Start initiative. Under the new effort, officials at the Department of Health and Human Services will work with outside partners to test a nationwide awareness campaign on best practices to reduce the rate of early elective deliveries before 39 weeks among all pregnant women. They will also test "enhanced prenatal care" approaches for Medicaid beneficiaries who are at risk for preterm birth.
The latest Leapfrog data are consistent with what other efforts that are aimed at reducing early elective deliveries have been able to show, said Dr. Donald Dudley, professor and vice chair for research in the department of ob.gyn. at the University of Texas Health Sciences Center in San Antonio.
Dr. Dudley also has been active in the March of Dimes Big 5 Prematurity Collaborative, which has been working to reduce preterm births through evidence-based quality improvement projects.
The Big 5 Prematurity Collaborative worked with 25 hospitals in California, Florida, Illinois, New York, and Texas to implement a policy of not performing elective deliveries before 39 weeks unless they are medically indicated. They found that hospitals – regardless of their size or location – were able to rapidly reduce early elective deliveries. The key, Dr. Dudley said, was making physicians and nurses aware of the data on complications from early deliveries.
Dr. Dudley said some physicians are skeptical that those last few weeks make that big a difference. He had been one of them. But once he read through the studies, it became clear that deliveries before 39 weeks were not the safest path for mothers and babies. "You’ll come away saying it is really not a good idea to do elective deliveries at less than 39 weeks," he said.
Once the buy-in from providers is there, making the change is fairly simple and relatively cheap, Dr. Dudley said. Hospitals can take advantage of free toolkits like the one from the California Maternal Quality Care Collaborative and the March of Dimes.
The most costly and time-intensive part of any effort to reduce early elective deliveries is the data collection, he said. Hospitals need to know how they doing at the start, but many community hospitals don’t routinely collect those data.
Dr. Dudley cautioned hospitals to be careful not to push the policy so hard that they discourage physicians from exercising their clinical judgment. "One of the things we have to be really careful about is not to swing the pendulum so far the other way that we disengage our brains."
If there’s a reason for a patient to be delivered at less than 39 weeks, then the delivery is indicated, not elective, he said. And there is a long list of appropriate indications for an early delivery.
For example, Dr. Dudley said he recently had a patient whom he had been monitoring and hoping to deliver after 39 weeks, but at 38 and a half weeks her blood pressure began creeping up enough that it was necessary to do the delivery early. The patient had a history of hypertension, and the risk of waiting outweighed the risks of early induction, he said. "You don’t want to delay acting when there’s an acceptable indication."
Policies aimed at reducing early elective deliveries can introduce a reluctance to induce (either by the physician or the patient) that may result in delaying an induction that is actually indicated, said Dr. William H. Barth Jr., chief of the division of maternal-fetal medicine at Massachusetts General Hospital, Boston, where the early elective newborn delivery rate has declined from 8% to 4% between 2010 and 2011. "Recommending an induction is a complex decision," he said. "The recent push to decrease elective inductions is sort of one more factor in the mix of that complex decision."
But the other issue is simple math, Dr. Barth said. If more inductions are delayed – indicated or not – there will be more stillbirths. He pointed to a retrospective cohort study published last year in Obstetrics & Gynecology that looked at the effect of a policy limiting elective delivery before 39 weeks’ gestation. The researchers found a significant decrease in admissions to the neonatal ICU, but they also detected an increase in stillbirths at 37 and 38 weeks (Obstet. Gynecol. 2011;118:1047-55).
Dr. Barth advised hospitals considering formal policies to reduce early elective deliveries to track not only the number of elective deliveries, but also stillbirths. "Shared decision making between patients and physicians is best conducted with all of the information – good and bad," he said.
At North Shore Medical Center in Salem, Mass., interest in reducing elective deliveries before 39 weeks’ gestation started with a newborn with respiratory problems.
A few years ago, when a patient was induced at about 38 weeks’ gestation, the newborn ended up having significant respiratory complications and had to be transferred to a tertiary care center, said Dr. Allyson L. Preston, chair of obstetrics and gynecology there.
Although the baby recovered and did fine, a review of the case led to a deeper examination of the data on elective deliveries at 37-39 weeks. The result was a growing realization that early elective deliveries weren’t as benign as previously thought, Dr. Preston said in an interview.
After hearing about the success that Intermountain Healthcare in Utah had in reducing early elective deliveries, Dr. Preston worked with her physician and nursing colleagues in the department of ob.gyn. to develop a policy not to schedule either elective inductions or cesarean sections if the patient was at less than 39 weeks’ gestation, unless there was a medical or obstetrical indication that early delivery was necessary.
In early 2010, the hospital staff sent out new booking forms to area obstetrical offices. The new forms required physicians to note the indications for early delivery and the gestational age. The charge nurses at North Shore Medical Center were given the authority not to schedule procedures if a patient was at less than 39 weeks’ gestation and the physician did not include an accepted medical or obstetrical indication for delivery. Physicians who took exception to the policy were referred to Dr. Preston. For the most part, physicians have eagerly adopted the policy, she said.
"I think people realized we were serious about it," Dr. Preston said. "They realized it was the right thing to do."
The results since the policy was implemented have been fairly dramatic. Data released earlier this year by the Leapfrog Group show that in 2010, the early elective newborn delivery rate was 31.6% at North Shore, compared with just 1.6% in 2011. That means that among deliveries done at 37-39 weeks, more than 98% are now medically indicated, according to Dr. Preston.
Other hospitals around the country are experiencing similar success, according to data from the Leapfrog Group’s annual hospital survey. The survey of 757 hospitals found that 39% of reporting hospitals had early elective delivery rates of 5% or less in 2011, compared with 30% the previous year. The rate of elective deliveries represents the percentage of nonmedically indicated births at 37-39 weeks’ gestation delivered by caesarean section or induction. The average rate for early elective deliveries among these hospitals dropped from 17% to 14% between 2010 and 2011.
Despite the progress already being made to curb early deliveries in some hospitals, there was still a wide variation among the hospitals in the Leapfrog survey, with early elective delivery rates ranging from a low of less than 5% to more than 40%, according to the 2011 data.
The Leapfrog Group (a coalition of public and private purchasers of employee health benefits that works on health care quality improvement) began collecting data on early elective deliveries in 2009 and first publicly reported hospital data in 2010. The group has identified the trend toward early deliveries as both a safety and an economic issue. For instance, women who are induced in weeks 37 and 38 have a higher risk of needing a cesarean section than do women who go into labor on their own. Studies also have linked early elective deliveries to postpartum complications such as hematoma, wound dehiscence, anemia, endometriosis, urinary tract infections, and sepsis, according to the Leapfrog Group.
The Leapfrog Group is not the only organization that has made the reduction of early elective deliveries a priority. For many years, the American College of Obstetricians and Gynecologists has recommended against elective deliveries before 39 weeks. In February, the federal government added its weight to the push to reduce these types of elective deliveries with the launch of its Strong Start initiative. Under the new effort, officials at the Department of Health and Human Services will work with outside partners to test a nationwide awareness campaign on best practices to reduce the rate of early elective deliveries before 39 weeks among all pregnant women. They will also test "enhanced prenatal care" approaches for Medicaid beneficiaries who are at risk for preterm birth.
The latest Leapfrog data are consistent with what other efforts that are aimed at reducing early elective deliveries have been able to show, said Dr. Donald Dudley, professor and vice chair for research in the department of ob.gyn. at the University of Texas Health Sciences Center in San Antonio.
Dr. Dudley also has been active in the March of Dimes Big 5 Prematurity Collaborative, which has been working to reduce preterm births through evidence-based quality improvement projects.
The Big 5 Prematurity Collaborative worked with 25 hospitals in California, Florida, Illinois, New York, and Texas to implement a policy of not performing elective deliveries before 39 weeks unless they are medically indicated. They found that hospitals – regardless of their size or location – were able to rapidly reduce early elective deliveries. The key, Dr. Dudley said, was making physicians and nurses aware of the data on complications from early deliveries.
Dr. Dudley said some physicians are skeptical that those last few weeks make that big a difference. He had been one of them. But once he read through the studies, it became clear that deliveries before 39 weeks were not the safest path for mothers and babies. "You’ll come away saying it is really not a good idea to do elective deliveries at less than 39 weeks," he said.
Once the buy-in from providers is there, making the change is fairly simple and relatively cheap, Dr. Dudley said. Hospitals can take advantage of free toolkits like the one from the California Maternal Quality Care Collaborative and the March of Dimes.
The most costly and time-intensive part of any effort to reduce early elective deliveries is the data collection, he said. Hospitals need to know how they doing at the start, but many community hospitals don’t routinely collect those data.
Dr. Dudley cautioned hospitals to be careful not to push the policy so hard that they discourage physicians from exercising their clinical judgment. "One of the things we have to be really careful about is not to swing the pendulum so far the other way that we disengage our brains."
If there’s a reason for a patient to be delivered at less than 39 weeks, then the delivery is indicated, not elective, he said. And there is a long list of appropriate indications for an early delivery.
For example, Dr. Dudley said he recently had a patient whom he had been monitoring and hoping to deliver after 39 weeks, but at 38 and a half weeks her blood pressure began creeping up enough that it was necessary to do the delivery early. The patient had a history of hypertension, and the risk of waiting outweighed the risks of early induction, he said. "You don’t want to delay acting when there’s an acceptable indication."
Policies aimed at reducing early elective deliveries can introduce a reluctance to induce (either by the physician or the patient) that may result in delaying an induction that is actually indicated, said Dr. William H. Barth Jr., chief of the division of maternal-fetal medicine at Massachusetts General Hospital, Boston, where the early elective newborn delivery rate has declined from 8% to 4% between 2010 and 2011. "Recommending an induction is a complex decision," he said. "The recent push to decrease elective inductions is sort of one more factor in the mix of that complex decision."
But the other issue is simple math, Dr. Barth said. If more inductions are delayed – indicated or not – there will be more stillbirths. He pointed to a retrospective cohort study published last year in Obstetrics & Gynecology that looked at the effect of a policy limiting elective delivery before 39 weeks’ gestation. The researchers found a significant decrease in admissions to the neonatal ICU, but they also detected an increase in stillbirths at 37 and 38 weeks (Obstet. Gynecol. 2011;118:1047-55).
Dr. Barth advised hospitals considering formal policies to reduce early elective deliveries to track not only the number of elective deliveries, but also stillbirths. "Shared decision making between patients and physicians is best conducted with all of the information – good and bad," he said.
ACA Could Surface as Issue in Wisconsin Primary
The Republican presidential primary calendar is hitting the midway point, and health care continues to be an on-again, off-again issue for the candidates.
Throughout the contentious primary season, all of the candidates have pummeled President Obama for his signature domestic policy achievement – the Affordable Care Act (ACA). And in turn, many of the GOP candidates have tried to damage front-runner Mitt Romney by attacking the health care insurance mandate he supported as governor of Massachusetts. Experts say voters may be in for more of the same in the upcoming contests in Wisconsin, Maryland, and the District of Columbia on April 3.
Health care will likely be one of the "subplots" of the April 3 primary, said Christopher C. Hull, Ph.D., a Republican strategist and a former adjunct professor of government at Georgetown University.
The remaining Republican candidates will continue to assert that Mr. Romney’s support of the individual insurance mandate in Massachusetts will undermine his ability to attack President Obama and the federal law in the general election. And Mr. Romney’s public comments on the federal law have given his opponents more ammunition, Dr. Hull said. Mr. Romney has used careful language in talking about what he would do about the ACA if elected, saying he would direct his Health and Human Services secretary to give all 50 states a waiver from the law’s requirements. That’s short of an outright repeal of the law, Dr. Hull said.
These charges are most likely to surface in Wisconsin, he said. "Wisconsin is kind of ground zero in the debate that is playing out in the U.S. over the role of government."
The ACA has been a polarizing issue in the state, said Larry Pheifer, executive director of the Wisconsin Academy of Family Physicians, which has members on both sides of the law.
The Wisconsin Academy of Family Physicians has tried to focus on advancing primary care and getting policy makers to address the workforce shortages in the state, Mr. Pheifer said, through better reimbursement for the patient-centered medical home, loan forgiveness, and payment reform.
If health care is able to break through some of the other issues, Mr. Pheifer said he hopes that workforce issues will be part of the discussion.
Another hot-button health care issue that GOP candidates are likely to hear about when they arrive in Wisconsin is the future of Badgercare Plus, a state program that offers health insurance options for uninsured individuals and families who earn too much to qualify for Medicaid. The program, which was originally created as Badgercare in the late 1990s, was expanded in 2008.
Republican Gov. Scott Walker is seeking to raise premiums and tighten eligibility rules for Badgercare Plus to help close a budget shortfall in the state’s Medicaid program.
Mike Herl, chairman of the Republican party in Dane County, Wis., said the discussion surrounding Badgercare Plus is likely to feed into the larger debate over the ACA.
"We don’t want some behemoth program coming down our throats from the federal level," Mr. Herl said.
Gov. Walker opposes the ACA. When he took office in 2011, he immediately authorized the state’s attorney general to join the multistate lawsuit against the health law that was just heard by the Supreme Court. And in January, Gov. Walker refused to accept federal grant money to help establish a state-based health insurance exchange under the ACA.
The April 3 primary also includes GOP presidential contests in Maryland and the District of Columbia, two strongholds of support for Democrats.
Health care is likely to be dwarfed by the economy in these areas, said Elizabeth Rigby, Ph.D., an assistant professor of public policy at George Washington University, Washington. Although health care is a major issue in policy circles, it is not rising to the top of the list for voters.
"The economy is more what is driving people at this point," she said.
The other reason health care is unlikely to emerge as a key issue, Dr. Rigby said, is that D.C. and Maryland are moving forward with their implementation of the ACA with little or no resistance.
The rhetoric around repealing the ACA is unlikely to garner the same reaction in Maryland as in other places, said Gene M. Ransom III, CEO of the Maryland State Medical Society.
"I don’t see it as a big issue," he said.
The Republican presidential primary calendar is hitting the midway point, and health care continues to be an on-again, off-again issue for the candidates.
Throughout the contentious primary season, all of the candidates have pummeled President Obama for his signature domestic policy achievement – the Affordable Care Act (ACA). And in turn, many of the GOP candidates have tried to damage front-runner Mitt Romney by attacking the health care insurance mandate he supported as governor of Massachusetts. Experts say voters may be in for more of the same in the upcoming contests in Wisconsin, Maryland, and the District of Columbia on April 3.
Health care will likely be one of the "subplots" of the April 3 primary, said Christopher C. Hull, Ph.D., a Republican strategist and a former adjunct professor of government at Georgetown University.
The remaining Republican candidates will continue to assert that Mr. Romney’s support of the individual insurance mandate in Massachusetts will undermine his ability to attack President Obama and the federal law in the general election. And Mr. Romney’s public comments on the federal law have given his opponents more ammunition, Dr. Hull said. Mr. Romney has used careful language in talking about what he would do about the ACA if elected, saying he would direct his Health and Human Services secretary to give all 50 states a waiver from the law’s requirements. That’s short of an outright repeal of the law, Dr. Hull said.
These charges are most likely to surface in Wisconsin, he said. "Wisconsin is kind of ground zero in the debate that is playing out in the U.S. over the role of government."
The ACA has been a polarizing issue in the state, said Larry Pheifer, executive director of the Wisconsin Academy of Family Physicians, which has members on both sides of the law.
The Wisconsin Academy of Family Physicians has tried to focus on advancing primary care and getting policy makers to address the workforce shortages in the state, Mr. Pheifer said, through better reimbursement for the patient-centered medical home, loan forgiveness, and payment reform.
If health care is able to break through some of the other issues, Mr. Pheifer said he hopes that workforce issues will be part of the discussion.
Another hot-button health care issue that GOP candidates are likely to hear about when they arrive in Wisconsin is the future of Badgercare Plus, a state program that offers health insurance options for uninsured individuals and families who earn too much to qualify for Medicaid. The program, which was originally created as Badgercare in the late 1990s, was expanded in 2008.
Republican Gov. Scott Walker is seeking to raise premiums and tighten eligibility rules for Badgercare Plus to help close a budget shortfall in the state’s Medicaid program.
Mike Herl, chairman of the Republican party in Dane County, Wis., said the discussion surrounding Badgercare Plus is likely to feed into the larger debate over the ACA.
"We don’t want some behemoth program coming down our throats from the federal level," Mr. Herl said.
Gov. Walker opposes the ACA. When he took office in 2011, he immediately authorized the state’s attorney general to join the multistate lawsuit against the health law that was just heard by the Supreme Court. And in January, Gov. Walker refused to accept federal grant money to help establish a state-based health insurance exchange under the ACA.
The April 3 primary also includes GOP presidential contests in Maryland and the District of Columbia, two strongholds of support for Democrats.
Health care is likely to be dwarfed by the economy in these areas, said Elizabeth Rigby, Ph.D., an assistant professor of public policy at George Washington University, Washington. Although health care is a major issue in policy circles, it is not rising to the top of the list for voters.
"The economy is more what is driving people at this point," she said.
The other reason health care is unlikely to emerge as a key issue, Dr. Rigby said, is that D.C. and Maryland are moving forward with their implementation of the ACA with little or no resistance.
The rhetoric around repealing the ACA is unlikely to garner the same reaction in Maryland as in other places, said Gene M. Ransom III, CEO of the Maryland State Medical Society.
"I don’t see it as a big issue," he said.
The Republican presidential primary calendar is hitting the midway point, and health care continues to be an on-again, off-again issue for the candidates.
Throughout the contentious primary season, all of the candidates have pummeled President Obama for his signature domestic policy achievement – the Affordable Care Act (ACA). And in turn, many of the GOP candidates have tried to damage front-runner Mitt Romney by attacking the health care insurance mandate he supported as governor of Massachusetts. Experts say voters may be in for more of the same in the upcoming contests in Wisconsin, Maryland, and the District of Columbia on April 3.
Health care will likely be one of the "subplots" of the April 3 primary, said Christopher C. Hull, Ph.D., a Republican strategist and a former adjunct professor of government at Georgetown University.
The remaining Republican candidates will continue to assert that Mr. Romney’s support of the individual insurance mandate in Massachusetts will undermine his ability to attack President Obama and the federal law in the general election. And Mr. Romney’s public comments on the federal law have given his opponents more ammunition, Dr. Hull said. Mr. Romney has used careful language in talking about what he would do about the ACA if elected, saying he would direct his Health and Human Services secretary to give all 50 states a waiver from the law’s requirements. That’s short of an outright repeal of the law, Dr. Hull said.
These charges are most likely to surface in Wisconsin, he said. "Wisconsin is kind of ground zero in the debate that is playing out in the U.S. over the role of government."
The ACA has been a polarizing issue in the state, said Larry Pheifer, executive director of the Wisconsin Academy of Family Physicians, which has members on both sides of the law.
The Wisconsin Academy of Family Physicians has tried to focus on advancing primary care and getting policy makers to address the workforce shortages in the state, Mr. Pheifer said, through better reimbursement for the patient-centered medical home, loan forgiveness, and payment reform.
If health care is able to break through some of the other issues, Mr. Pheifer said he hopes that workforce issues will be part of the discussion.
Another hot-button health care issue that GOP candidates are likely to hear about when they arrive in Wisconsin is the future of Badgercare Plus, a state program that offers health insurance options for uninsured individuals and families who earn too much to qualify for Medicaid. The program, which was originally created as Badgercare in the late 1990s, was expanded in 2008.
Republican Gov. Scott Walker is seeking to raise premiums and tighten eligibility rules for Badgercare Plus to help close a budget shortfall in the state’s Medicaid program.
Mike Herl, chairman of the Republican party in Dane County, Wis., said the discussion surrounding Badgercare Plus is likely to feed into the larger debate over the ACA.
"We don’t want some behemoth program coming down our throats from the federal level," Mr. Herl said.
Gov. Walker opposes the ACA. When he took office in 2011, he immediately authorized the state’s attorney general to join the multistate lawsuit against the health law that was just heard by the Supreme Court. And in January, Gov. Walker refused to accept federal grant money to help establish a state-based health insurance exchange under the ACA.
The April 3 primary also includes GOP presidential contests in Maryland and the District of Columbia, two strongholds of support for Democrats.
Health care is likely to be dwarfed by the economy in these areas, said Elizabeth Rigby, Ph.D., an assistant professor of public policy at George Washington University, Washington. Although health care is a major issue in policy circles, it is not rising to the top of the list for voters.
"The economy is more what is driving people at this point," she said.
The other reason health care is unlikely to emerge as a key issue, Dr. Rigby said, is that D.C. and Maryland are moving forward with their implementation of the ACA with little or no resistance.
The rhetoric around repealing the ACA is unlikely to garner the same reaction in Maryland as in other places, said Gene M. Ransom III, CEO of the Maryland State Medical Society.
"I don’t see it as a big issue," he said.
Caution Urged in Moving More Drugs OTC
Although expanding nonprescription access to some medications could be beneficial, patients with a wide array of chronic conditions need regular medical attention.
That was the message physicians and nurses gave to the Food and Drug Administration at a hearing March 22-23 to explore moving more medications to nonprescription status.
In an early March notice in the Federal Register, the FDA outlined possible scenarios for moving drugs to nonprescription status with added requirements for safe use, including the following:
• Lengthen the time between follow-up visits. Under this scenario, patients would see their physician or other health care provider for an initial prescription, and the prescriber would authorize refills beyond what normally are currently allowed before a follow-up visit is required. In addition, conditions of safe use would be set for the refills. In its proposal, FDA officials noted that this approach could be used for rescue inhalers to treat asthma, or epinephrine for allergic reactions.
• Make use of new technology. A patient could use a pharmacy kiosk or website to self-screen for a particular condition; an associated algorithm would help the patient determine whether a particular medication is appropriate or contraindicated.
• Increase the role of the pharmacist. Certain drugs would be switched to OTC status, but in order to purchase the drug, the patient would need to visit with the pharmacist. Pharmacists could potentially be involved in conducting and interpreting diagnostics, including blood tests or liver function tests. Based on the results of those tests, pharmacists could help determine if a particular drug is appropriate or contraindicated.
In its notice, the FDA did not list any dermatology products for specific consideration for an OTC switch. But there are some topical medications that could fit into that framework, such as topical antibiotics for acne or antiviral medicines for cold sores, said Dr. Barbara R. Reed, clinical professor of dermatology at the University of Colorado in Denver.
Dr. Reed said the proposal could be a way to get patients more involved in their own health. "I love seeing people take more responsibility for their care," she said.
But she also said she has concerns about how the idea might be implemented. For instance, if the policy results in more patients mixing and matching drugs with limited or no monitoring, it could lead to an increase in adverse drug reactions. Another concern is that requiring patients to interact with the pharmacist as a condition of OTC purchase could quickly overburden pharmacists. There's also a need to ensure that patients get correct, standardized information about these medications, she said. "It's extremely complex."
Dr. Sandra Adamson Fryhofer, an internist and chair-elect of the American Medical Association’s Council on Science and Public Health, said that the FDA proposals for increasing OTC access to medications such as epinephrine autoinjectors for allergic reactions likely would be beneficial. But, she said, she is skeptical that chronic conditions could be managed that way. Since the disease progression changes over time and new comorbidities may appear, a lack of interaction with a physician is problematic, she said.
Pharmacists have done well in administering immunizations over the past several years, she said. But bypassing the physicians in treating chronic disease would be a "mistake."
Dr. Fryhofer also raised concerns about the FDA’s plans to use kiosks or computer programs to allow patients to self-diagnose and to determine if certain medications are appropriate. For instance, in the case of treatments for hyperlipidemia, patients may be able get access to an appropriate drug through a kiosk. However, without regular care by a physician, such patients could progress to heart disease or experience a cardiac event, yet not have a connection to the health care system in the event of an emergency, she said.
"The patient is sort of left out in the cold," Dr. Fryhofer said.
The proposed OTC option could also undermine efforts to create accountable care organizations (ACOs), in which a team of health care providers in the inpatient and outpatient settings work together on the care of patients.
Greater usage of OTC medications that isn’t captured, monitored, and shared with the members of the ACO would make it harder for the team to manage a patient’s health care, she said.
FDA officials acknowledged the concerns. But Dr. Janet Woodcock, director of the FDA’s Center for Drug Evaluation and Research, said that the agency is looking toward the future, when pharmacists, physicians, and patients will all be better connected through the use of electronic health records and patient portals. Greater pharmacist involvement also has the potential to bring nonadherent patients back into the health care system, she said.
"I strongly believe that medicine needs to take medical care to where the patients are," Dr. Woodcock said. "This is one way that we will really improve both access and adherence."
The FDA is not considering any fundamental changes to the scientific process for reclassifying products from prescription to OTC, Dr. Woodcock said. "This is more about how the information would be made available to the consumer, not what quantity of evidence would be used to decide that the product could be nonprescription."
Making the change will require rule making from the FDA, Dr. Woodcock said, so the agency is gathering public input. In addition to the testimony at the 2-day hearing, the agency is accepting public comments until May 7.
Jan Towers, Ph.D., the director of health policy and federal government and professional affairs for the American Academy of Nurse Practitioners, also urged the FDA to proceed with caution. Although moving certain drugs to OTC status is in many cases beneficial, the model that the agency is proposing would work best in a closed system like an HMO, or in retail clinics where pharmacists and physicians interact directly, she said.
She also urged FDA officials to consider whether a given disease or condition requires clinician oversight and how much, what kind, and how often. The major concern, Dr. Towers said, is that patients could fall through the cracks because too many different people are doing too many different things.
But not every health care provider at the hearing threw up the yellow caution flag. Some physician-researchers in ob.gyn. urged the FDA to consider moving oral contraceptives over the counter.
Dr. Daniel Grossman of the University of California San Francisco and a senior associate at Ibis Reproductive Health, said that the current data strongly support the safety of progestin-only oral contraception over the counter. These drugs have no significant toxicity, they are not addictive, users can self-diagnose, and users can safely take the drug without a doctor’s screening, he said.
Not surprisingly, pharmacists are eager to move forward with the FDA’s proposed options. Tom Menighan, executive vice president and CEO of the American Pharmacists Association, said that increasing the role of pharmacists will actually get patients back into the health care system by getting patients who have dropped their therapies back on medication. Pharmacists also would refer patients back to their physicians when appropriate, he said.
Mr. Menighan said that pharmacists’ role in administering immunizations can be a model for expanding their role in medications. Over the last decade, states have enacted laws allowing pharmacists to immunize, and now more than 175,000 pharmacists have completed certificate training programs to administer vaccines, he said, adding that in the 2010-2011 influenza season, pharmacists administered approximately 20 million influenza vaccines.
To move forward, pharmacists would need appropriate algorithms, standards of care, and documentation for specific products, but those are achievable, he said.
Beverly Schaefer, a pharmacist and pharmacy owner in Seattle, agreed that pharmacists are well positioned to take on an expanded role if the FDA decides to make more drugs available OTC with conditions of safe use. "Pharmacists are the most accessible health care provider," she said.
Patients who might most benefit from the FDA proposal would be those seeking symptomatic relief from acute conditions, such as antivirals for shingles or a cold sore outbreak. "When this happens on Friday night, what’s going to happen if you have to wait until Monday? Trouble," she said.
Ms. Schaefer highlighted other medicines that would be a good fit for the proposed new paradigm such as rescue inhalers for asthma attacks, burn ointment for minor burns, steroid nasal sprays to reduce sinus inflammations, epinephrine autoinjectors for allergic reactions, and antihistamine eye drops for allergy relief.
Dr. Bobby Q. Lanier, executive medical director for the American College of Allergy, Asthma, and Immunology, disagreed.
He called the FDA proposals "chilling and a little scary." Drugs such as albuterol inhalers require physician oversight because some patients are skilled at gaming the system to simply obtain their usual prescription and move on, he said. And he added that he didn’t know of any evidence that moving these types of drugs over the counter would improve compliance, adherence, or patient care.
Ms. Schaefer pointed out that pharmacists are already helping patients to be more adherent to their medicines, she said. Many community pharmacists offer medication reconciliation and medication therapy management programs, and pharmacists are assisting patients to correctly use, adhere to, and gain control of their medication regimen.
"We help patients understand why they are taking these medications," she said.
Although expanding nonprescription access to some medications could be beneficial, patients with a wide array of chronic conditions need regular medical attention.
That was the message physicians and nurses gave to the Food and Drug Administration at a hearing March 22-23 to explore moving more medications to nonprescription status.
In an early March notice in the Federal Register, the FDA outlined possible scenarios for moving drugs to nonprescription status with added requirements for safe use, including the following:
• Lengthen the time between follow-up visits. Under this scenario, patients would see their physician or other health care provider for an initial prescription, and the prescriber would authorize refills beyond what normally are currently allowed before a follow-up visit is required. In addition, conditions of safe use would be set for the refills. In its proposal, FDA officials noted that this approach could be used for rescue inhalers to treat asthma, or epinephrine for allergic reactions.
• Make use of new technology. A patient could use a pharmacy kiosk or website to self-screen for a particular condition; an associated algorithm would help the patient determine whether a particular medication is appropriate or contraindicated.
• Increase the role of the pharmacist. Certain drugs would be switched to OTC status, but in order to purchase the drug, the patient would need to visit with the pharmacist. Pharmacists could potentially be involved in conducting and interpreting diagnostics, including blood tests or liver function tests. Based on the results of those tests, pharmacists could help determine if a particular drug is appropriate or contraindicated.
In its notice, the FDA did not list any dermatology products for specific consideration for an OTC switch. But there are some topical medications that could fit into that framework, such as topical antibiotics for acne or antiviral medicines for cold sores, said Dr. Barbara R. Reed, clinical professor of dermatology at the University of Colorado in Denver.
Dr. Reed said the proposal could be a way to get patients more involved in their own health. "I love seeing people take more responsibility for their care," she said.
But she also said she has concerns about how the idea might be implemented. For instance, if the policy results in more patients mixing and matching drugs with limited or no monitoring, it could lead to an increase in adverse drug reactions. Another concern is that requiring patients to interact with the pharmacist as a condition of OTC purchase could quickly overburden pharmacists. There's also a need to ensure that patients get correct, standardized information about these medications, she said. "It's extremely complex."
Dr. Sandra Adamson Fryhofer, an internist and chair-elect of the American Medical Association’s Council on Science and Public Health, said that the FDA proposals for increasing OTC access to medications such as epinephrine autoinjectors for allergic reactions likely would be beneficial. But, she said, she is skeptical that chronic conditions could be managed that way. Since the disease progression changes over time and new comorbidities may appear, a lack of interaction with a physician is problematic, she said.
Pharmacists have done well in administering immunizations over the past several years, she said. But bypassing the physicians in treating chronic disease would be a "mistake."
Dr. Fryhofer also raised concerns about the FDA’s plans to use kiosks or computer programs to allow patients to self-diagnose and to determine if certain medications are appropriate. For instance, in the case of treatments for hyperlipidemia, patients may be able get access to an appropriate drug through a kiosk. However, without regular care by a physician, such patients could progress to heart disease or experience a cardiac event, yet not have a connection to the health care system in the event of an emergency, she said.
"The patient is sort of left out in the cold," Dr. Fryhofer said.
The proposed OTC option could also undermine efforts to create accountable care organizations (ACOs), in which a team of health care providers in the inpatient and outpatient settings work together on the care of patients.
Greater usage of OTC medications that isn’t captured, monitored, and shared with the members of the ACO would make it harder for the team to manage a patient’s health care, she said.
FDA officials acknowledged the concerns. But Dr. Janet Woodcock, director of the FDA’s Center for Drug Evaluation and Research, said that the agency is looking toward the future, when pharmacists, physicians, and patients will all be better connected through the use of electronic health records and patient portals. Greater pharmacist involvement also has the potential to bring nonadherent patients back into the health care system, she said.
"I strongly believe that medicine needs to take medical care to where the patients are," Dr. Woodcock said. "This is one way that we will really improve both access and adherence."
The FDA is not considering any fundamental changes to the scientific process for reclassifying products from prescription to OTC, Dr. Woodcock said. "This is more about how the information would be made available to the consumer, not what quantity of evidence would be used to decide that the product could be nonprescription."
Making the change will require rule making from the FDA, Dr. Woodcock said, so the agency is gathering public input. In addition to the testimony at the 2-day hearing, the agency is accepting public comments until May 7.
Jan Towers, Ph.D., the director of health policy and federal government and professional affairs for the American Academy of Nurse Practitioners, also urged the FDA to proceed with caution. Although moving certain drugs to OTC status is in many cases beneficial, the model that the agency is proposing would work best in a closed system like an HMO, or in retail clinics where pharmacists and physicians interact directly, she said.
She also urged FDA officials to consider whether a given disease or condition requires clinician oversight and how much, what kind, and how often. The major concern, Dr. Towers said, is that patients could fall through the cracks because too many different people are doing too many different things.
But not every health care provider at the hearing threw up the yellow caution flag. Some physician-researchers in ob.gyn. urged the FDA to consider moving oral contraceptives over the counter.
Dr. Daniel Grossman of the University of California San Francisco and a senior associate at Ibis Reproductive Health, said that the current data strongly support the safety of progestin-only oral contraception over the counter. These drugs have no significant toxicity, they are not addictive, users can self-diagnose, and users can safely take the drug without a doctor’s screening, he said.
Not surprisingly, pharmacists are eager to move forward with the FDA’s proposed options. Tom Menighan, executive vice president and CEO of the American Pharmacists Association, said that increasing the role of pharmacists will actually get patients back into the health care system by getting patients who have dropped their therapies back on medication. Pharmacists also would refer patients back to their physicians when appropriate, he said.
Mr. Menighan said that pharmacists’ role in administering immunizations can be a model for expanding their role in medications. Over the last decade, states have enacted laws allowing pharmacists to immunize, and now more than 175,000 pharmacists have completed certificate training programs to administer vaccines, he said, adding that in the 2010-2011 influenza season, pharmacists administered approximately 20 million influenza vaccines.
To move forward, pharmacists would need appropriate algorithms, standards of care, and documentation for specific products, but those are achievable, he said.
Beverly Schaefer, a pharmacist and pharmacy owner in Seattle, agreed that pharmacists are well positioned to take on an expanded role if the FDA decides to make more drugs available OTC with conditions of safe use. "Pharmacists are the most accessible health care provider," she said.
Patients who might most benefit from the FDA proposal would be those seeking symptomatic relief from acute conditions, such as antivirals for shingles or a cold sore outbreak. "When this happens on Friday night, what’s going to happen if you have to wait until Monday? Trouble," she said.
Ms. Schaefer highlighted other medicines that would be a good fit for the proposed new paradigm such as rescue inhalers for asthma attacks, burn ointment for minor burns, steroid nasal sprays to reduce sinus inflammations, epinephrine autoinjectors for allergic reactions, and antihistamine eye drops for allergy relief.
Dr. Bobby Q. Lanier, executive medical director for the American College of Allergy, Asthma, and Immunology, disagreed.
He called the FDA proposals "chilling and a little scary." Drugs such as albuterol inhalers require physician oversight because some patients are skilled at gaming the system to simply obtain their usual prescription and move on, he said. And he added that he didn’t know of any evidence that moving these types of drugs over the counter would improve compliance, adherence, or patient care.
Ms. Schaefer pointed out that pharmacists are already helping patients to be more adherent to their medicines, she said. Many community pharmacists offer medication reconciliation and medication therapy management programs, and pharmacists are assisting patients to correctly use, adhere to, and gain control of their medication regimen.
"We help patients understand why they are taking these medications," she said.
Although expanding nonprescription access to some medications could be beneficial, patients with a wide array of chronic conditions need regular medical attention.
That was the message physicians and nurses gave to the Food and Drug Administration at a hearing March 22-23 to explore moving more medications to nonprescription status.
In an early March notice in the Federal Register, the FDA outlined possible scenarios for moving drugs to nonprescription status with added requirements for safe use, including the following:
• Lengthen the time between follow-up visits. Under this scenario, patients would see their physician or other health care provider for an initial prescription, and the prescriber would authorize refills beyond what normally are currently allowed before a follow-up visit is required. In addition, conditions of safe use would be set for the refills. In its proposal, FDA officials noted that this approach could be used for rescue inhalers to treat asthma, or epinephrine for allergic reactions.
• Make use of new technology. A patient could use a pharmacy kiosk or website to self-screen for a particular condition; an associated algorithm would help the patient determine whether a particular medication is appropriate or contraindicated.
• Increase the role of the pharmacist. Certain drugs would be switched to OTC status, but in order to purchase the drug, the patient would need to visit with the pharmacist. Pharmacists could potentially be involved in conducting and interpreting diagnostics, including blood tests or liver function tests. Based on the results of those tests, pharmacists could help determine if a particular drug is appropriate or contraindicated.
In its notice, the FDA did not list any dermatology products for specific consideration for an OTC switch. But there are some topical medications that could fit into that framework, such as topical antibiotics for acne or antiviral medicines for cold sores, said Dr. Barbara R. Reed, clinical professor of dermatology at the University of Colorado in Denver.
Dr. Reed said the proposal could be a way to get patients more involved in their own health. "I love seeing people take more responsibility for their care," she said.
But she also said she has concerns about how the idea might be implemented. For instance, if the policy results in more patients mixing and matching drugs with limited or no monitoring, it could lead to an increase in adverse drug reactions. Another concern is that requiring patients to interact with the pharmacist as a condition of OTC purchase could quickly overburden pharmacists. There's also a need to ensure that patients get correct, standardized information about these medications, she said. "It's extremely complex."
Dr. Sandra Adamson Fryhofer, an internist and chair-elect of the American Medical Association’s Council on Science and Public Health, said that the FDA proposals for increasing OTC access to medications such as epinephrine autoinjectors for allergic reactions likely would be beneficial. But, she said, she is skeptical that chronic conditions could be managed that way. Since the disease progression changes over time and new comorbidities may appear, a lack of interaction with a physician is problematic, she said.
Pharmacists have done well in administering immunizations over the past several years, she said. But bypassing the physicians in treating chronic disease would be a "mistake."
Dr. Fryhofer also raised concerns about the FDA’s plans to use kiosks or computer programs to allow patients to self-diagnose and to determine if certain medications are appropriate. For instance, in the case of treatments for hyperlipidemia, patients may be able get access to an appropriate drug through a kiosk. However, without regular care by a physician, such patients could progress to heart disease or experience a cardiac event, yet not have a connection to the health care system in the event of an emergency, she said.
"The patient is sort of left out in the cold," Dr. Fryhofer said.
The proposed OTC option could also undermine efforts to create accountable care organizations (ACOs), in which a team of health care providers in the inpatient and outpatient settings work together on the care of patients.
Greater usage of OTC medications that isn’t captured, monitored, and shared with the members of the ACO would make it harder for the team to manage a patient’s health care, she said.
FDA officials acknowledged the concerns. But Dr. Janet Woodcock, director of the FDA’s Center for Drug Evaluation and Research, said that the agency is looking toward the future, when pharmacists, physicians, and patients will all be better connected through the use of electronic health records and patient portals. Greater pharmacist involvement also has the potential to bring nonadherent patients back into the health care system, she said.
"I strongly believe that medicine needs to take medical care to where the patients are," Dr. Woodcock said. "This is one way that we will really improve both access and adherence."
The FDA is not considering any fundamental changes to the scientific process for reclassifying products from prescription to OTC, Dr. Woodcock said. "This is more about how the information would be made available to the consumer, not what quantity of evidence would be used to decide that the product could be nonprescription."
Making the change will require rule making from the FDA, Dr. Woodcock said, so the agency is gathering public input. In addition to the testimony at the 2-day hearing, the agency is accepting public comments until May 7.
Jan Towers, Ph.D., the director of health policy and federal government and professional affairs for the American Academy of Nurse Practitioners, also urged the FDA to proceed with caution. Although moving certain drugs to OTC status is in many cases beneficial, the model that the agency is proposing would work best in a closed system like an HMO, or in retail clinics where pharmacists and physicians interact directly, she said.
She also urged FDA officials to consider whether a given disease or condition requires clinician oversight and how much, what kind, and how often. The major concern, Dr. Towers said, is that patients could fall through the cracks because too many different people are doing too many different things.
But not every health care provider at the hearing threw up the yellow caution flag. Some physician-researchers in ob.gyn. urged the FDA to consider moving oral contraceptives over the counter.
Dr. Daniel Grossman of the University of California San Francisco and a senior associate at Ibis Reproductive Health, said that the current data strongly support the safety of progestin-only oral contraception over the counter. These drugs have no significant toxicity, they are not addictive, users can self-diagnose, and users can safely take the drug without a doctor’s screening, he said.
Not surprisingly, pharmacists are eager to move forward with the FDA’s proposed options. Tom Menighan, executive vice president and CEO of the American Pharmacists Association, said that increasing the role of pharmacists will actually get patients back into the health care system by getting patients who have dropped their therapies back on medication. Pharmacists also would refer patients back to their physicians when appropriate, he said.
Mr. Menighan said that pharmacists’ role in administering immunizations can be a model for expanding their role in medications. Over the last decade, states have enacted laws allowing pharmacists to immunize, and now more than 175,000 pharmacists have completed certificate training programs to administer vaccines, he said, adding that in the 2010-2011 influenza season, pharmacists administered approximately 20 million influenza vaccines.
To move forward, pharmacists would need appropriate algorithms, standards of care, and documentation for specific products, but those are achievable, he said.
Beverly Schaefer, a pharmacist and pharmacy owner in Seattle, agreed that pharmacists are well positioned to take on an expanded role if the FDA decides to make more drugs available OTC with conditions of safe use. "Pharmacists are the most accessible health care provider," she said.
Patients who might most benefit from the FDA proposal would be those seeking symptomatic relief from acute conditions, such as antivirals for shingles or a cold sore outbreak. "When this happens on Friday night, what’s going to happen if you have to wait until Monday? Trouble," she said.
Ms. Schaefer highlighted other medicines that would be a good fit for the proposed new paradigm such as rescue inhalers for asthma attacks, burn ointment for minor burns, steroid nasal sprays to reduce sinus inflammations, epinephrine autoinjectors for allergic reactions, and antihistamine eye drops for allergy relief.
Dr. Bobby Q. Lanier, executive medical director for the American College of Allergy, Asthma, and Immunology, disagreed.
He called the FDA proposals "chilling and a little scary." Drugs such as albuterol inhalers require physician oversight because some patients are skilled at gaming the system to simply obtain their usual prescription and move on, he said. And he added that he didn’t know of any evidence that moving these types of drugs over the counter would improve compliance, adherence, or patient care.
Ms. Schaefer pointed out that pharmacists are already helping patients to be more adherent to their medicines, she said. Many community pharmacists offer medication reconciliation and medication therapy management programs, and pharmacists are assisting patients to correctly use, adhere to, and gain control of their medication regimen.
"We help patients understand why they are taking these medications," she said.
FROM A HEARING HELD BY THE FOOD AND DRUG ADMINISTRATION
Caution Urged in Moving More Drugs OTC
Although expanding nonprescription access to some medications could be beneficial, patients with a wide array of chronic conditions need regular medical attention.
That was the message physicians and nurses gave to the Food and Drug Administration at a hearing March 22-23 to explore moving more medications to nonprescription status.
In an early March notice in the Federal Register, the FDA outlined possible scenarios for moving drugs to nonprescription status with added requirements for safe use, including the following:
• Lengthen the time between follow-up visits. Under this scenario, patients would see their physician or other health care provider for an initial prescription, and the prescriber would authorize refills beyond what normally are currently allowed before a follow-up visit is required. In addition, conditions of safe use would be set for the refills. In its proposal, FDA officials noted that this approach could be used for rescue inhalers to treat asthma, or epinephrine for allergic reactions.
• Make use of new technology. A patient could use a pharmacy kiosk or website to self-screen for a particular condition; an associated algorithm would help the patient determine whether a particular medication is appropriate or contraindicated.
• Increase the role of the pharmacist. Certain drugs would be switched to OTC status, but in order to purchase the drug, the patient would need to visit with the pharmacist. Pharmacists could potentially be involved in conducting and interpreting diagnostics, including blood tests or liver function tests. Based on the results of those tests, pharmacists could help determine if a particular drug is appropriate or contraindicated.
Dr. Sandra Adamson Fryhofer, an internist and chair-elect of the American Medical Association’s Council on Science and Public Health, said that the FDA proposals for increasing OTC access to medications such as epinephrine autoinjectors for allergic reactions likely would be beneficial. But, she said, she is skeptical that chronic conditions could be managed that way. Since the disease progression changes over time and new comorbidities may appear, a lack of interaction with a physician is problematic, she said.
Pharmacists have done well in administering immunizations over the past several years, she said. But bypassing the physicians in treating chronic disease would be a "mistake."
Dr. Fryhofer also raised concerns about the FDA’s plans to use kiosks or computer programs to allow patients to self-diagnose and to determine if certain medications are appropriate. For instance, in the case of treatments for hyperlipidemia, patients may be able get access to an appropriate drug through a kiosk. However, without regular care by a physician, such patients could progress to heart disease or experience a cardiac event, yet not have a connection to the health care system in the event of an emergency, she said.
"The patient is sort of left out in the cold," Dr. Fryhofer said.
The proposed OTC option could also undermine efforts to create accountable care organizations (ACOs), in which a team of health care providers in the inpatient and outpatient settings work together on the care of patients.
Greater usage of OTC medications that isn’t captured, monitored, and shared with the members of the ACO would make it harder for the team to manage a patient’s health care, she said.
FDA officials acknowledged the concerns. But Dr. Janet Woodcock, director of the FDA’s Center for Drug Evaluation and Research, said that the agency is looking toward the future, when pharmacists, physicians, and patients will all be better connected through the use of electronic health records and patient portals. Greater pharmacist involvement also has the potential to bring nonadherent patients back into the health care system, she said.
"I strongly believe that medicine needs to take medical care to where the patients are," Dr. Woodcock said. "This is one way that we will really improve both access and adherence."
The FDA is not considering any fundamental changes to the scientific process for reclassifying products from prescription to OTC, Dr. Woodcock said. "This is more about how the information would be made available to the consumer, not what quantity of evidence would be used to decide that the product could be nonprescription."
Making the change will require rule making from the FDA, Dr. Woodcock said, so the agency is gathering public input. In addition to the testimony at the 2-day hearing, the agency is accepting public comments until May 7.
Jan Towers, Ph.D., the director of health policy and federal government and professional affairs for the American Academy of Nurse Practitioners, also urged the FDA to proceed with caution. Although moving certain drugs to OTC status is in many cases beneficial, the model that the agency is proposing would work best in a closed system like an HMO, or in retail clinics where pharmacists and physicians interact directly, she said.
She also urged FDA officials to consider whether a given disease or condition requires clinician oversight and how much, what kind, and how often. The major concern, Dr. Towers said, is that patients could fall through the cracks because too many different people are doing too many different things.
But not every health care provider at the hearing threw up the yellow caution flag. Some physician-researchers in ob.gyn. urged the FDA to consider moving oral contraceptives over the counter.
Dr. Daniel Grossman of the University of California San Francisco and a senior associate at Ibis Reproductive Health, said that the current data strongly support the safety of progestin-only oral contraception over the counter. These drugs have no significant toxicity, they are not addictive, users can self-diagnose, and users can safely take the drug without a doctor’s screening, he said.
Not surprisingly, pharmacists are eager to move forward with the FDA’s proposed options. Tom Menighan, executive vice president and CEO of the American Pharmacists Association, said that increasing the role of pharmacists will actually get patients back into the health care system by getting patients who have dropped their therapies back on medication. Pharmacists also would refer patients back to their physicians when appropriate, he said.
Mr. Menighan said that pharmacists’ role in administering immunizations can be a model for expanding their role in medications. Over the last decade, states have enacted laws allowing pharmacists to immunize, and now more than 175,000 pharmacists have completed certificate training programs to administer vaccines, he said, adding that in the 2010-2011 influenza season, pharmacists administered approximately 20 million influenza vaccines.
To move forward, pharmacists would need appropriate algorithms, standards of care, and documentation for specific products, but those are achievable, he said.
Beverly Schaefer, a pharmacist and pharmacy owner in Seattle, agreed that pharmacists are well positioned to take on an expanded role if the FDA decides to make more drugs available OTC with conditions of safe use. "Pharmacists are the most accessible health care provider," she said.
Patients who might most benefit from the FDA proposal would be those seeking symptomatic relief from acute conditions, such as antivirals for shingles or a cold sore outbreak. "When this happens on Friday night, what’s going to happen if you have to wait until Monday? Trouble," she said.
Ms. Schaefer highlighted other medicines that would be a good fit for the proposed new paradigm such as rescue inhalers for asthma attacks, burn ointment for minor burns, steroid nasal sprays to reduce sinus inflammations, epinephrine autoinjectors for allergic reactions, and antihistamine eye drops for allergy relief.
Dr. Bobby Q. Lanier, executive medical director for the American College of Allergy, Asthma, and Immunology, disagreed.
He called the FDA proposals "chilling and a little scary." Drugs such as albuterol inhalers require physician oversight because some patients are skilled at gaming the system to simply obtain their usual prescription and move on, he said. And he added that he didn’t know of any evidence that moving these types of drugs over the counter would improve compliance, adherence, or patient care.
Ms. Schaefer pointed out that pharmacists are already helping patients to be more adherent to their medicines, she said. Many community pharmacists offer medication reconciliation and medication therapy management programs, and pharmacists are assisting patients to correctly use, adhere to, and gain control of their medication regimen.
"We help patients understand why they are taking these medications," she said.
Although expanding nonprescription access to some medications could be beneficial, patients with a wide array of chronic conditions need regular medical attention.
That was the message physicians and nurses gave to the Food and Drug Administration at a hearing March 22-23 to explore moving more medications to nonprescription status.
In an early March notice in the Federal Register, the FDA outlined possible scenarios for moving drugs to nonprescription status with added requirements for safe use, including the following:
• Lengthen the time between follow-up visits. Under this scenario, patients would see their physician or other health care provider for an initial prescription, and the prescriber would authorize refills beyond what normally are currently allowed before a follow-up visit is required. In addition, conditions of safe use would be set for the refills. In its proposal, FDA officials noted that this approach could be used for rescue inhalers to treat asthma, or epinephrine for allergic reactions.
• Make use of new technology. A patient could use a pharmacy kiosk or website to self-screen for a particular condition; an associated algorithm would help the patient determine whether a particular medication is appropriate or contraindicated.
• Increase the role of the pharmacist. Certain drugs would be switched to OTC status, but in order to purchase the drug, the patient would need to visit with the pharmacist. Pharmacists could potentially be involved in conducting and interpreting diagnostics, including blood tests or liver function tests. Based on the results of those tests, pharmacists could help determine if a particular drug is appropriate or contraindicated.
Dr. Sandra Adamson Fryhofer, an internist and chair-elect of the American Medical Association’s Council on Science and Public Health, said that the FDA proposals for increasing OTC access to medications such as epinephrine autoinjectors for allergic reactions likely would be beneficial. But, she said, she is skeptical that chronic conditions could be managed that way. Since the disease progression changes over time and new comorbidities may appear, a lack of interaction with a physician is problematic, she said.
Pharmacists have done well in administering immunizations over the past several years, she said. But bypassing the physicians in treating chronic disease would be a "mistake."
Dr. Fryhofer also raised concerns about the FDA’s plans to use kiosks or computer programs to allow patients to self-diagnose and to determine if certain medications are appropriate. For instance, in the case of treatments for hyperlipidemia, patients may be able get access to an appropriate drug through a kiosk. However, without regular care by a physician, such patients could progress to heart disease or experience a cardiac event, yet not have a connection to the health care system in the event of an emergency, she said.
"The patient is sort of left out in the cold," Dr. Fryhofer said.
The proposed OTC option could also undermine efforts to create accountable care organizations (ACOs), in which a team of health care providers in the inpatient and outpatient settings work together on the care of patients.
Greater usage of OTC medications that isn’t captured, monitored, and shared with the members of the ACO would make it harder for the team to manage a patient’s health care, she said.
FDA officials acknowledged the concerns. But Dr. Janet Woodcock, director of the FDA’s Center for Drug Evaluation and Research, said that the agency is looking toward the future, when pharmacists, physicians, and patients will all be better connected through the use of electronic health records and patient portals. Greater pharmacist involvement also has the potential to bring nonadherent patients back into the health care system, she said.
"I strongly believe that medicine needs to take medical care to where the patients are," Dr. Woodcock said. "This is one way that we will really improve both access and adherence."
The FDA is not considering any fundamental changes to the scientific process for reclassifying products from prescription to OTC, Dr. Woodcock said. "This is more about how the information would be made available to the consumer, not what quantity of evidence would be used to decide that the product could be nonprescription."
Making the change will require rule making from the FDA, Dr. Woodcock said, so the agency is gathering public input. In addition to the testimony at the 2-day hearing, the agency is accepting public comments until May 7.
Jan Towers, Ph.D., the director of health policy and federal government and professional affairs for the American Academy of Nurse Practitioners, also urged the FDA to proceed with caution. Although moving certain drugs to OTC status is in many cases beneficial, the model that the agency is proposing would work best in a closed system like an HMO, or in retail clinics where pharmacists and physicians interact directly, she said.
She also urged FDA officials to consider whether a given disease or condition requires clinician oversight and how much, what kind, and how often. The major concern, Dr. Towers said, is that patients could fall through the cracks because too many different people are doing too many different things.
But not every health care provider at the hearing threw up the yellow caution flag. Some physician-researchers in ob.gyn. urged the FDA to consider moving oral contraceptives over the counter.
Dr. Daniel Grossman of the University of California San Francisco and a senior associate at Ibis Reproductive Health, said that the current data strongly support the safety of progestin-only oral contraception over the counter. These drugs have no significant toxicity, they are not addictive, users can self-diagnose, and users can safely take the drug without a doctor’s screening, he said.
Not surprisingly, pharmacists are eager to move forward with the FDA’s proposed options. Tom Menighan, executive vice president and CEO of the American Pharmacists Association, said that increasing the role of pharmacists will actually get patients back into the health care system by getting patients who have dropped their therapies back on medication. Pharmacists also would refer patients back to their physicians when appropriate, he said.
Mr. Menighan said that pharmacists’ role in administering immunizations can be a model for expanding their role in medications. Over the last decade, states have enacted laws allowing pharmacists to immunize, and now more than 175,000 pharmacists have completed certificate training programs to administer vaccines, he said, adding that in the 2010-2011 influenza season, pharmacists administered approximately 20 million influenza vaccines.
To move forward, pharmacists would need appropriate algorithms, standards of care, and documentation for specific products, but those are achievable, he said.
Beverly Schaefer, a pharmacist and pharmacy owner in Seattle, agreed that pharmacists are well positioned to take on an expanded role if the FDA decides to make more drugs available OTC with conditions of safe use. "Pharmacists are the most accessible health care provider," she said.
Patients who might most benefit from the FDA proposal would be those seeking symptomatic relief from acute conditions, such as antivirals for shingles or a cold sore outbreak. "When this happens on Friday night, what’s going to happen if you have to wait until Monday? Trouble," she said.
Ms. Schaefer highlighted other medicines that would be a good fit for the proposed new paradigm such as rescue inhalers for asthma attacks, burn ointment for minor burns, steroid nasal sprays to reduce sinus inflammations, epinephrine autoinjectors for allergic reactions, and antihistamine eye drops for allergy relief.
Dr. Bobby Q. Lanier, executive medical director for the American College of Allergy, Asthma, and Immunology, disagreed.
He called the FDA proposals "chilling and a little scary." Drugs such as albuterol inhalers require physician oversight because some patients are skilled at gaming the system to simply obtain their usual prescription and move on, he said. And he added that he didn’t know of any evidence that moving these types of drugs over the counter would improve compliance, adherence, or patient care.
Ms. Schaefer pointed out that pharmacists are already helping patients to be more adherent to their medicines, she said. Many community pharmacists offer medication reconciliation and medication therapy management programs, and pharmacists are assisting patients to correctly use, adhere to, and gain control of their medication regimen.
"We help patients understand why they are taking these medications," she said.
Although expanding nonprescription access to some medications could be beneficial, patients with a wide array of chronic conditions need regular medical attention.
That was the message physicians and nurses gave to the Food and Drug Administration at a hearing March 22-23 to explore moving more medications to nonprescription status.
In an early March notice in the Federal Register, the FDA outlined possible scenarios for moving drugs to nonprescription status with added requirements for safe use, including the following:
• Lengthen the time between follow-up visits. Under this scenario, patients would see their physician or other health care provider for an initial prescription, and the prescriber would authorize refills beyond what normally are currently allowed before a follow-up visit is required. In addition, conditions of safe use would be set for the refills. In its proposal, FDA officials noted that this approach could be used for rescue inhalers to treat asthma, or epinephrine for allergic reactions.
• Make use of new technology. A patient could use a pharmacy kiosk or website to self-screen for a particular condition; an associated algorithm would help the patient determine whether a particular medication is appropriate or contraindicated.
• Increase the role of the pharmacist. Certain drugs would be switched to OTC status, but in order to purchase the drug, the patient would need to visit with the pharmacist. Pharmacists could potentially be involved in conducting and interpreting diagnostics, including blood tests or liver function tests. Based on the results of those tests, pharmacists could help determine if a particular drug is appropriate or contraindicated.
Dr. Sandra Adamson Fryhofer, an internist and chair-elect of the American Medical Association’s Council on Science and Public Health, said that the FDA proposals for increasing OTC access to medications such as epinephrine autoinjectors for allergic reactions likely would be beneficial. But, she said, she is skeptical that chronic conditions could be managed that way. Since the disease progression changes over time and new comorbidities may appear, a lack of interaction with a physician is problematic, she said.
Pharmacists have done well in administering immunizations over the past several years, she said. But bypassing the physicians in treating chronic disease would be a "mistake."
Dr. Fryhofer also raised concerns about the FDA’s plans to use kiosks or computer programs to allow patients to self-diagnose and to determine if certain medications are appropriate. For instance, in the case of treatments for hyperlipidemia, patients may be able get access to an appropriate drug through a kiosk. However, without regular care by a physician, such patients could progress to heart disease or experience a cardiac event, yet not have a connection to the health care system in the event of an emergency, she said.
"The patient is sort of left out in the cold," Dr. Fryhofer said.
The proposed OTC option could also undermine efforts to create accountable care organizations (ACOs), in which a team of health care providers in the inpatient and outpatient settings work together on the care of patients.
Greater usage of OTC medications that isn’t captured, monitored, and shared with the members of the ACO would make it harder for the team to manage a patient’s health care, she said.
FDA officials acknowledged the concerns. But Dr. Janet Woodcock, director of the FDA’s Center for Drug Evaluation and Research, said that the agency is looking toward the future, when pharmacists, physicians, and patients will all be better connected through the use of electronic health records and patient portals. Greater pharmacist involvement also has the potential to bring nonadherent patients back into the health care system, she said.
"I strongly believe that medicine needs to take medical care to where the patients are," Dr. Woodcock said. "This is one way that we will really improve both access and adherence."
The FDA is not considering any fundamental changes to the scientific process for reclassifying products from prescription to OTC, Dr. Woodcock said. "This is more about how the information would be made available to the consumer, not what quantity of evidence would be used to decide that the product could be nonprescription."
Making the change will require rule making from the FDA, Dr. Woodcock said, so the agency is gathering public input. In addition to the testimony at the 2-day hearing, the agency is accepting public comments until May 7.
Jan Towers, Ph.D., the director of health policy and federal government and professional affairs for the American Academy of Nurse Practitioners, also urged the FDA to proceed with caution. Although moving certain drugs to OTC status is in many cases beneficial, the model that the agency is proposing would work best in a closed system like an HMO, or in retail clinics where pharmacists and physicians interact directly, she said.
She also urged FDA officials to consider whether a given disease or condition requires clinician oversight and how much, what kind, and how often. The major concern, Dr. Towers said, is that patients could fall through the cracks because too many different people are doing too many different things.
But not every health care provider at the hearing threw up the yellow caution flag. Some physician-researchers in ob.gyn. urged the FDA to consider moving oral contraceptives over the counter.
Dr. Daniel Grossman of the University of California San Francisco and a senior associate at Ibis Reproductive Health, said that the current data strongly support the safety of progestin-only oral contraception over the counter. These drugs have no significant toxicity, they are not addictive, users can self-diagnose, and users can safely take the drug without a doctor’s screening, he said.
Not surprisingly, pharmacists are eager to move forward with the FDA’s proposed options. Tom Menighan, executive vice president and CEO of the American Pharmacists Association, said that increasing the role of pharmacists will actually get patients back into the health care system by getting patients who have dropped their therapies back on medication. Pharmacists also would refer patients back to their physicians when appropriate, he said.
Mr. Menighan said that pharmacists’ role in administering immunizations can be a model for expanding their role in medications. Over the last decade, states have enacted laws allowing pharmacists to immunize, and now more than 175,000 pharmacists have completed certificate training programs to administer vaccines, he said, adding that in the 2010-2011 influenza season, pharmacists administered approximately 20 million influenza vaccines.
To move forward, pharmacists would need appropriate algorithms, standards of care, and documentation for specific products, but those are achievable, he said.
Beverly Schaefer, a pharmacist and pharmacy owner in Seattle, agreed that pharmacists are well positioned to take on an expanded role if the FDA decides to make more drugs available OTC with conditions of safe use. "Pharmacists are the most accessible health care provider," she said.
Patients who might most benefit from the FDA proposal would be those seeking symptomatic relief from acute conditions, such as antivirals for shingles or a cold sore outbreak. "When this happens on Friday night, what’s going to happen if you have to wait until Monday? Trouble," she said.
Ms. Schaefer highlighted other medicines that would be a good fit for the proposed new paradigm such as rescue inhalers for asthma attacks, burn ointment for minor burns, steroid nasal sprays to reduce sinus inflammations, epinephrine autoinjectors for allergic reactions, and antihistamine eye drops for allergy relief.
Dr. Bobby Q. Lanier, executive medical director for the American College of Allergy, Asthma, and Immunology, disagreed.
He called the FDA proposals "chilling and a little scary." Drugs such as albuterol inhalers require physician oversight because some patients are skilled at gaming the system to simply obtain their usual prescription and move on, he said. And he added that he didn’t know of any evidence that moving these types of drugs over the counter would improve compliance, adherence, or patient care.
Ms. Schaefer pointed out that pharmacists are already helping patients to be more adherent to their medicines, she said. Many community pharmacists offer medication reconciliation and medication therapy management programs, and pharmacists are assisting patients to correctly use, adhere to, and gain control of their medication regimen.
"We help patients understand why they are taking these medications," she said.
FROM A HEARING HELD BY THE FOOD AND DRUG ADMINISTRATION