User login
Telemedicine: Past, present, and future
Telemedicine has been used successfully to improve patient access to medical care while reducing healthcare costs. In 2016, an estimated 61% of US healthcare institutions and 40% to 50% of US hospitals used telemedicine.1 From 2012 to 2013, the telemedicine market grew by 60%. However, its widespread use has been limited by low reimbursement rates and interstate licensing and practice issues.
In this commentary, we discuss the history of telemedicine, current uses and challenges, and areas of future growth.
DEFINITION AND HISTORY
The World Health Organization defines telemedicine as “the delivery of health care services, where distance is a critical factor, by all health care professionals using information and communication technologies for the exchange of valid information for diagnosis, treatment and prevention of disease and injuries, research and evaluation, and for the continuing education of healthcare providers, all in the interests of advancing the health of individuals and their communities.”2
Modern telemedicine began in the early 1900s in the Netherlands with the transmission of heart rhythms over the telephone,3 which was followed by transmissions to radio consultation centers in Europe in the 1920s. In the 1940s, radiographic images were transmitted by telephone between cities in Pennsylvania.4
Today, telemedicine is used in a variety of specialties including radiology, neurology, and pathology5 and by organizations in the United States ranging from the National Aeronautics and Space Administration and Kaiser Permanente to the US Department of Veterans Affairs (VA). The VA, in particular, is a leader in telemedicine. In 2012, it reduced mental health hospitalizations by over 40%, heart failure hospitalizations by 25%, and diabetes and chronic obstructive pulmonary disease hospitalizations by about 20% using telemedicine programs.6 In 2015, it provided about 2.1 million telemedicine consultations to 677,000 veterans.7
TYPES OF TELEMEDICINE PROGRAMS
There are 2 types of telemedicine programs.
Synchronous programs take place in real time and are a live 2-way interaction between the patient and healthcare provider. This includes virtual appointments that are conducted using the patient’s smartphone, tablet, or computer with a camera. When using a smartphone or tablet, patients must first download an app that connects them with a provider.
Asynchronous programs, also known as “store and forward” applications, are not live and involve the transfer of images, videos, and other clinical information that a healthcare provider views and responds to at a later time. In this case, patients may wear medical devices to monitor and track health information (eg, blood pressure) in a personal health application that they forward to their healthcare provider.
IMPROVING PATIENT ACCESS TO CARE WHILE REDUCING COSTS
Telemedicine allows patients living in both rural and urban areas to access healthcare when they need it. Currently, about 59 million Americans reside in health professional-shortage areas, which are rural and urban areas with shortages of primary care providers.1 These patients often experience long delays when attempting to schedule a healthcare visit7 and may experience issues with continuity of care if they are unable to see the same care provider at every visit.
It also provides access to care to patients without reliable transportation or those who may be too sick to travel long distances. For some patients, such as those with cystic fibrosis who do not want to come to the hospital for fear of contracting multiple antibiotic-resistant bacteria, a virtual office visit may be safer.
At the same time, telemedicine helps reduce healthcare costs. For example, it:
- Optimizes staff distribution and healthcare resources within a healthcare facility and across an entire system
- Enables primary care providers to conduct appointments without additional office staff at any time, thereby extending office hours and availability
- Reduces the financial impact of patient no-shows
- Improves patient engagement and outcomes
- Reduces unnecessary office and emergency room visits and hospital admissions.
The last point is especially important for senior living and skilled nursing centers whose residents are known to have high rates of hospital admissions.8,9 In these facilities, 24-hour medical assistance may not be available, and telemedicine can help troubleshoot common problems.
LOW REIMBURSEMENT RATES CURTAIL USE
Limited reimbursement has curtailed the widespread use of telemedicine. Although rules for reimbursement are evolving, telemedicine still represents a small amount of total healthcare expenditures. In 2015, Medicare spent approximately $14.4 million on services delivered via telemedicine—less than 0.01% of total spending on healthcare services.1
Currently, 31 states and the District of Columbia have telemedicine parity laws that mandate private commercial insurers to pay for telemedicine services.10 Unfortunately, there is a lack of uniformity in the specifics of these laws, resulting in variations in reimbursement rates. Furthermore, a large number of larger insurers such as Medicare and Medicaid and many self-insured plans do not fall under these mandates.
Another factor that affects reimbursement for telemedicine services is the setting of the medical encounter. Medicare reimburses providers for telemedicine services only when they are conducted at specific sites such as physician’s offices, hospitals, rural health centers, and skilled nursing facilities. Additionally, Medicare only reimburses for services in areas with a shortage of healthcare professionals and in non-metropolitan areas, which excludes many urban patients.11
In contrast, more commercial reimbursement is occurring for online urgent care, and options for commercial reimbursement of online behavioral services are being explored.
INTERSTATE LICENSURE ISSUES
Current licensure laws also limit the ability of many healthcare providers to offer telemedicine services. Federal law requires providers to be fully licensed to practice medicine in the state where the patient is physically located. In cases of health systems that have locations in more than one state, providers may need to apply for and pay to maintain multiple licenses (current interstate licensing laws vary across states).
Interstate licensure is one way to solve this problem. Thus far, a number of states have joined the Interstate Medical Licensure Compact that intends to allow physicians to obtain expedited licenses to practice in multiple states.12
The federal TELE-MED Act was introduced in 2015 but not passed. It proposed to “allow a Medicare provider to provide telemedicine services to a Medicare beneficiary who is in a different state from the one in which the provider is licensed or authorized to provide healthcare services.”
CAN TELEMEDICINE FOSTER A GOOD PROVIDER-PATIENT RELATIONSHIP?
In-person encounters provide healthcare providers with the opportunity to build a therapeutic relationship with their patients. Face-to-face encounters also increase patient satisfaction scores and outcomes. As such, critics fear that patient relationships may suffer with the use of telemedicine. However, using video technology for new patient encounters may help overcome this challenge. During a video encounter, the provider can see the patient’s facial expressions and take cues from nonverbal behaviors.
At times, the element of distance may enhance the encounter. For example, in behavioral health, patients often feel more comfortable in their home environment than in a sterile office environment.
Telemedicine patients often have positive experiences, given the speed of access, precision, time savings, and the ability to stay in contact with healthcare providers from the comfort of their homes. Ultimately, these virtual visits may help improve compliance with follow-up consultations since the barriers of distance and transportation are circumvented.
WHO CAN CONDUCT TELEMEDICINE VISITS?
Although a patient’s healthcare team is likely to consist of members who are not physicians, including nurse practitioners, physician assistants, social workers, and psychologists, not everyone can, by law, conduct telemedicine visits. Currently, the rules and regulations addressing ancillary team members’ participation in telemedicine vary from state to state.
TELEMEDICINE VISITS AT CLEVELAND CLINIC
Our health system has several telemedicine programs, including our eHospital program. Launched in 2014, this program provides patients at 4 hospitals with input from staff intensivists and experienced critical care nurses during the night (7 pm to 7 am) via remote monitoring. These remote caregivers have full access to patient charts and, when signalled, can activate an in-room camera to initiate 2-way audio communication with patients, their families, and bedside caregivers.
In addition, new patient consults are being offered via telemedicine for several services including dermatology, where pictures of skin lesions are reviewed and triaged, and management recommendations are provided accordingly.
In 2016, Cleveland Clinic launched its Remote Hypertension Improvement Program—an enterprise-wide initiative to minimize hypertension-associated mortality and morbidity with the assistance of telehealth services. The program was first piloted in a group of 80 high-risk hypertensive patients who were monitored and followed through a Bluetooth-enabled remote monitoring tool, which exported blood pressure readings to a central dashboard. A multidisciplinary team of doctors, nurses, and pharmacists used this dashboard to adjust medication when needed and provide virtual lifestyle coaching. Over a 24-week period, the patients’ systolic blood pressure decreased by an average of 7.5 mm Hg and diastolic blood pressure by 3.1 mm Hg (unpublished data).
Beginning this year, blood pressure readings will be directly exported from the remote monitoring tool into the patient’s electronic medical record, providing the healthcare team with the information needed to make informed decisions to remotely manage patients with hypertension.
Remote monitoring of patients with hypertension is also being used at other institutions such as the VA. In 2016, almost 19,000 veterans were using the remote monitoring system, and this number is expected to increase with the enhanced adaptation of telemedicine services.13
FUTURE DIRECTIONS
About 50% of all adults in the United States have at least 1 chronic disease. In all, chronic disease accounts for roughly 75% of the total healthcare expenditure and 70% of all deaths.7,14 Recent data suggest that virtual chronic disease management represents an untapped market for telemedicine, given its relative underutilization compared to other services such as telebehavorial health and specialty telemedicine. These patients require frequent visits to the doctor, and targeting this patient population with telemedicine may decrease the number of emergency room visits and hospital admissions.
Another growing area in the field of telemedicine is the “hospital at home” model in which patients who meet the criteria for hospitalization but are otherwise stable are treated at home for diseases such as chronic obstructive pulmonary disease, pneumonia, and heart failure. Studies have shown that the hospital-at-home model, when used appropriately, is not only more cost-effective than hospitalization but results in a shorter treatment duration and lower rates of delirium.15–17
Finally, in the acute setting, we have seen wide success with telemedicine programs in stroke care, radiology, intensive care, and psychiatry, and several studies have shown mortality rates comparable to those with the traditional model.18,19 These encounters often require specialized skills and are the focus of multiple ongoing studies.
Acknowledgment: The authors would like to acknowledge and thank Matthew Faiman, MD, for providing information regarding the Remote Hypertension Program.
- US Department of Health and Human Services. Report to Congress: e-health and telemedicine. aspe.hhs.gov/system/files/pdf/206751/TelemedicineE-HealthReport.pdf. Accessed September 1, 2018.
- World Health Organization (WHO). A Health Telematics Policy in Support of WHO’s Health-For-All Strategy for Global Health Development: Report of the WHO Group Consultation on Health Telematics, 11–16 December, Geneva 1997. World Health Organization, Geneva, 1998.
- Bashshur RL, Shannon GW. History of telemedicine: evolution, context, and transformation. Mary Ann Liebert, Inc.: New Rochelle (NY), 2009.
- Bashshur RL, Goldberg MA. The origins of telemedicine and e-Health. Telemed J E Health 2014; 20(3):190–191. doi:10.1089/tmj.2014.9996
- Bashshur RL, Shannon G, Krupinski EA, Grigsby J. Sustaining and realizing the promise of telemedicine. Telemed J E Health 2013; 19(5):339–345. doi:10.1089/tmj.2012.0282
- American Hospital Association (AHA). Issue Brief. Telehealth: helping hospitals deliver cost-effective care. www.aha.org/system/files/content/16/16telehealthissuebrief.pdf. Accessed September 10, 2018.
- Congressional Research Service. Telehealth and Telemedicine: description and issues. March 29, 2016. www.senate.gov/CRSpubs/757e3b90-ff10-497c-8e8c-ac1bdbdb3aaf.pdf. Accessed August 8, 2018.
- Grabowski DC, Stewart KA, Broderick SM, Coots LA. Predictors of nursing home hospitalization: a review of the literature. Med Care Res Rev 2008; 65(1):3–39. doi:10.1177/1077558707308754
- Grabowski DC, O’Malley AJ. Use of telemedicine can reduce hospitalizations of nursing home residents and generate savings for Medicare. Health Aff (Millwood) 2014; 33(2):244–250. doi:10.1377/hlthaff.2013.0922
- Jones K. If not parity, clarity—getting doctors paid for telehealth. www.forbes.com/sites/realspin/2016/09/15/if-not-parity-clarity-getting-doctors-paid-for-telehealth/#43928587777f. Accessed September 1, 2018.
- Neufeld JD, Doarn CR. Telemedicine spending by Medicare: a snapshot from 2012. Telemed J E Health 2015; 21(8):686–693. doi:10.1089/tmj.2014.0185
- Chaudhry HJ, Robin LA, Fish EM, Polk DH, Gifford JD. Improving access and mobility—the Interstate Medical Licensure Compact. N Engl J Med 2015; 372(17):1581–1583. doi:10.1056/NEJMp1502639
- United States Government Accountability Office. Report to Congressional Committees. Healthcare: telehealth and remote patient monitoring use in Medicare and selected federal programs. www.gao.gov/assets/690/684115.pdf. Accessed September 1, 2018.
- Bashshur RL, Shannon GW, Smith BR, et al. The empirical foundations of telemedicine interventions for chronic disease management. Telemed J E Health 2014; 20(9):769–800. doi:10.1089/tmj.2014.9981
- Cryer L, Shannon SB, Van Amsterdam M, Leff B. Costs for ‘hospital at home’ patients were 19 percent lower, with equal or better outcomes compared to similar inpatients. Health Aff (Millwood) 2012; 31:1237–1243. doi:10.1377/hlthaff.2011.1132
- Leff B, Burton L, Mader SL, et al. Hospital at home: feasibility and outcomes of a program to provide hospital-level care at home for acutely ill older patients. Ann Intern Med 2005; 143(11):798–808. pmid:16330791
- Leff B, Soones T, DeCherrie L. The hospital at home program for older adults. JAMA Intern Med 2016; 176(11):1724–1725. doi:10.1001/jamainternmed.2016.6307
- Wechsler LR, Demaerschalk BM, Schwamm LH, et al; American Heart Association Stroke Council; Council on Epidemiology and Prevention; Council on Quality of Care and Outcomes Research. Telemedicine quality and outcomes in stroke: a scientific statement for healthcare professionals from the American Heart Association/American Stroke Association. Stroke 2017; 48(1):e3–e25. doi:10.1161/STR.0000000000000114
- Wilcox ME, Wiener-Kronish JP. Telemedicine in the intensive care unit: effect of a remote intensivist on outcomes. JAMA Intern Med 2014; 174(7):1167–1169. doi:10.1001/jamainternmed.2014.289
Telemedicine has been used successfully to improve patient access to medical care while reducing healthcare costs. In 2016, an estimated 61% of US healthcare institutions and 40% to 50% of US hospitals used telemedicine.1 From 2012 to 2013, the telemedicine market grew by 60%. However, its widespread use has been limited by low reimbursement rates and interstate licensing and practice issues.
In this commentary, we discuss the history of telemedicine, current uses and challenges, and areas of future growth.
DEFINITION AND HISTORY
The World Health Organization defines telemedicine as “the delivery of health care services, where distance is a critical factor, by all health care professionals using information and communication technologies for the exchange of valid information for diagnosis, treatment and prevention of disease and injuries, research and evaluation, and for the continuing education of healthcare providers, all in the interests of advancing the health of individuals and their communities.”2
Modern telemedicine began in the early 1900s in the Netherlands with the transmission of heart rhythms over the telephone,3 which was followed by transmissions to radio consultation centers in Europe in the 1920s. In the 1940s, radiographic images were transmitted by telephone between cities in Pennsylvania.4
Today, telemedicine is used in a variety of specialties including radiology, neurology, and pathology5 and by organizations in the United States ranging from the National Aeronautics and Space Administration and Kaiser Permanente to the US Department of Veterans Affairs (VA). The VA, in particular, is a leader in telemedicine. In 2012, it reduced mental health hospitalizations by over 40%, heart failure hospitalizations by 25%, and diabetes and chronic obstructive pulmonary disease hospitalizations by about 20% using telemedicine programs.6 In 2015, it provided about 2.1 million telemedicine consultations to 677,000 veterans.7
TYPES OF TELEMEDICINE PROGRAMS
There are 2 types of telemedicine programs.
Synchronous programs take place in real time and are a live 2-way interaction between the patient and healthcare provider. This includes virtual appointments that are conducted using the patient’s smartphone, tablet, or computer with a camera. When using a smartphone or tablet, patients must first download an app that connects them with a provider.
Asynchronous programs, also known as “store and forward” applications, are not live and involve the transfer of images, videos, and other clinical information that a healthcare provider views and responds to at a later time. In this case, patients may wear medical devices to monitor and track health information (eg, blood pressure) in a personal health application that they forward to their healthcare provider.
IMPROVING PATIENT ACCESS TO CARE WHILE REDUCING COSTS
Telemedicine allows patients living in both rural and urban areas to access healthcare when they need it. Currently, about 59 million Americans reside in health professional-shortage areas, which are rural and urban areas with shortages of primary care providers.1 These patients often experience long delays when attempting to schedule a healthcare visit7 and may experience issues with continuity of care if they are unable to see the same care provider at every visit.
It also provides access to care to patients without reliable transportation or those who may be too sick to travel long distances. For some patients, such as those with cystic fibrosis who do not want to come to the hospital for fear of contracting multiple antibiotic-resistant bacteria, a virtual office visit may be safer.
At the same time, telemedicine helps reduce healthcare costs. For example, it:
- Optimizes staff distribution and healthcare resources within a healthcare facility and across an entire system
- Enables primary care providers to conduct appointments without additional office staff at any time, thereby extending office hours and availability
- Reduces the financial impact of patient no-shows
- Improves patient engagement and outcomes
- Reduces unnecessary office and emergency room visits and hospital admissions.
The last point is especially important for senior living and skilled nursing centers whose residents are known to have high rates of hospital admissions.8,9 In these facilities, 24-hour medical assistance may not be available, and telemedicine can help troubleshoot common problems.
LOW REIMBURSEMENT RATES CURTAIL USE
Limited reimbursement has curtailed the widespread use of telemedicine. Although rules for reimbursement are evolving, telemedicine still represents a small amount of total healthcare expenditures. In 2015, Medicare spent approximately $14.4 million on services delivered via telemedicine—less than 0.01% of total spending on healthcare services.1
Currently, 31 states and the District of Columbia have telemedicine parity laws that mandate private commercial insurers to pay for telemedicine services.10 Unfortunately, there is a lack of uniformity in the specifics of these laws, resulting in variations in reimbursement rates. Furthermore, a large number of larger insurers such as Medicare and Medicaid and many self-insured plans do not fall under these mandates.
Another factor that affects reimbursement for telemedicine services is the setting of the medical encounter. Medicare reimburses providers for telemedicine services only when they are conducted at specific sites such as physician’s offices, hospitals, rural health centers, and skilled nursing facilities. Additionally, Medicare only reimburses for services in areas with a shortage of healthcare professionals and in non-metropolitan areas, which excludes many urban patients.11
In contrast, more commercial reimbursement is occurring for online urgent care, and options for commercial reimbursement of online behavioral services are being explored.
INTERSTATE LICENSURE ISSUES
Current licensure laws also limit the ability of many healthcare providers to offer telemedicine services. Federal law requires providers to be fully licensed to practice medicine in the state where the patient is physically located. In cases of health systems that have locations in more than one state, providers may need to apply for and pay to maintain multiple licenses (current interstate licensing laws vary across states).
Interstate licensure is one way to solve this problem. Thus far, a number of states have joined the Interstate Medical Licensure Compact that intends to allow physicians to obtain expedited licenses to practice in multiple states.12
The federal TELE-MED Act was introduced in 2015 but not passed. It proposed to “allow a Medicare provider to provide telemedicine services to a Medicare beneficiary who is in a different state from the one in which the provider is licensed or authorized to provide healthcare services.”
CAN TELEMEDICINE FOSTER A GOOD PROVIDER-PATIENT RELATIONSHIP?
In-person encounters provide healthcare providers with the opportunity to build a therapeutic relationship with their patients. Face-to-face encounters also increase patient satisfaction scores and outcomes. As such, critics fear that patient relationships may suffer with the use of telemedicine. However, using video technology for new patient encounters may help overcome this challenge. During a video encounter, the provider can see the patient’s facial expressions and take cues from nonverbal behaviors.
At times, the element of distance may enhance the encounter. For example, in behavioral health, patients often feel more comfortable in their home environment than in a sterile office environment.
Telemedicine patients often have positive experiences, given the speed of access, precision, time savings, and the ability to stay in contact with healthcare providers from the comfort of their homes. Ultimately, these virtual visits may help improve compliance with follow-up consultations since the barriers of distance and transportation are circumvented.
WHO CAN CONDUCT TELEMEDICINE VISITS?
Although a patient’s healthcare team is likely to consist of members who are not physicians, including nurse practitioners, physician assistants, social workers, and psychologists, not everyone can, by law, conduct telemedicine visits. Currently, the rules and regulations addressing ancillary team members’ participation in telemedicine vary from state to state.
TELEMEDICINE VISITS AT CLEVELAND CLINIC
Our health system has several telemedicine programs, including our eHospital program. Launched in 2014, this program provides patients at 4 hospitals with input from staff intensivists and experienced critical care nurses during the night (7 pm to 7 am) via remote monitoring. These remote caregivers have full access to patient charts and, when signalled, can activate an in-room camera to initiate 2-way audio communication with patients, their families, and bedside caregivers.
In addition, new patient consults are being offered via telemedicine for several services including dermatology, where pictures of skin lesions are reviewed and triaged, and management recommendations are provided accordingly.
In 2016, Cleveland Clinic launched its Remote Hypertension Improvement Program—an enterprise-wide initiative to minimize hypertension-associated mortality and morbidity with the assistance of telehealth services. The program was first piloted in a group of 80 high-risk hypertensive patients who were monitored and followed through a Bluetooth-enabled remote monitoring tool, which exported blood pressure readings to a central dashboard. A multidisciplinary team of doctors, nurses, and pharmacists used this dashboard to adjust medication when needed and provide virtual lifestyle coaching. Over a 24-week period, the patients’ systolic blood pressure decreased by an average of 7.5 mm Hg and diastolic blood pressure by 3.1 mm Hg (unpublished data).
Beginning this year, blood pressure readings will be directly exported from the remote monitoring tool into the patient’s electronic medical record, providing the healthcare team with the information needed to make informed decisions to remotely manage patients with hypertension.
Remote monitoring of patients with hypertension is also being used at other institutions such as the VA. In 2016, almost 19,000 veterans were using the remote monitoring system, and this number is expected to increase with the enhanced adaptation of telemedicine services.13
FUTURE DIRECTIONS
About 50% of all adults in the United States have at least 1 chronic disease. In all, chronic disease accounts for roughly 75% of the total healthcare expenditure and 70% of all deaths.7,14 Recent data suggest that virtual chronic disease management represents an untapped market for telemedicine, given its relative underutilization compared to other services such as telebehavorial health and specialty telemedicine. These patients require frequent visits to the doctor, and targeting this patient population with telemedicine may decrease the number of emergency room visits and hospital admissions.
Another growing area in the field of telemedicine is the “hospital at home” model in which patients who meet the criteria for hospitalization but are otherwise stable are treated at home for diseases such as chronic obstructive pulmonary disease, pneumonia, and heart failure. Studies have shown that the hospital-at-home model, when used appropriately, is not only more cost-effective than hospitalization but results in a shorter treatment duration and lower rates of delirium.15–17
Finally, in the acute setting, we have seen wide success with telemedicine programs in stroke care, radiology, intensive care, and psychiatry, and several studies have shown mortality rates comparable to those with the traditional model.18,19 These encounters often require specialized skills and are the focus of multiple ongoing studies.
Acknowledgment: The authors would like to acknowledge and thank Matthew Faiman, MD, for providing information regarding the Remote Hypertension Program.
Telemedicine has been used successfully to improve patient access to medical care while reducing healthcare costs. In 2016, an estimated 61% of US healthcare institutions and 40% to 50% of US hospitals used telemedicine.1 From 2012 to 2013, the telemedicine market grew by 60%. However, its widespread use has been limited by low reimbursement rates and interstate licensing and practice issues.
In this commentary, we discuss the history of telemedicine, current uses and challenges, and areas of future growth.
DEFINITION AND HISTORY
The World Health Organization defines telemedicine as “the delivery of health care services, where distance is a critical factor, by all health care professionals using information and communication technologies for the exchange of valid information for diagnosis, treatment and prevention of disease and injuries, research and evaluation, and for the continuing education of healthcare providers, all in the interests of advancing the health of individuals and their communities.”2
Modern telemedicine began in the early 1900s in the Netherlands with the transmission of heart rhythms over the telephone,3 which was followed by transmissions to radio consultation centers in Europe in the 1920s. In the 1940s, radiographic images were transmitted by telephone between cities in Pennsylvania.4
Today, telemedicine is used in a variety of specialties including radiology, neurology, and pathology5 and by organizations in the United States ranging from the National Aeronautics and Space Administration and Kaiser Permanente to the US Department of Veterans Affairs (VA). The VA, in particular, is a leader in telemedicine. In 2012, it reduced mental health hospitalizations by over 40%, heart failure hospitalizations by 25%, and diabetes and chronic obstructive pulmonary disease hospitalizations by about 20% using telemedicine programs.6 In 2015, it provided about 2.1 million telemedicine consultations to 677,000 veterans.7
TYPES OF TELEMEDICINE PROGRAMS
There are 2 types of telemedicine programs.
Synchronous programs take place in real time and are a live 2-way interaction between the patient and healthcare provider. This includes virtual appointments that are conducted using the patient’s smartphone, tablet, or computer with a camera. When using a smartphone or tablet, patients must first download an app that connects them with a provider.
Asynchronous programs, also known as “store and forward” applications, are not live and involve the transfer of images, videos, and other clinical information that a healthcare provider views and responds to at a later time. In this case, patients may wear medical devices to monitor and track health information (eg, blood pressure) in a personal health application that they forward to their healthcare provider.
IMPROVING PATIENT ACCESS TO CARE WHILE REDUCING COSTS
Telemedicine allows patients living in both rural and urban areas to access healthcare when they need it. Currently, about 59 million Americans reside in health professional-shortage areas, which are rural and urban areas with shortages of primary care providers.1 These patients often experience long delays when attempting to schedule a healthcare visit7 and may experience issues with continuity of care if they are unable to see the same care provider at every visit.
It also provides access to care to patients without reliable transportation or those who may be too sick to travel long distances. For some patients, such as those with cystic fibrosis who do not want to come to the hospital for fear of contracting multiple antibiotic-resistant bacteria, a virtual office visit may be safer.
At the same time, telemedicine helps reduce healthcare costs. For example, it:
- Optimizes staff distribution and healthcare resources within a healthcare facility and across an entire system
- Enables primary care providers to conduct appointments without additional office staff at any time, thereby extending office hours and availability
- Reduces the financial impact of patient no-shows
- Improves patient engagement and outcomes
- Reduces unnecessary office and emergency room visits and hospital admissions.
The last point is especially important for senior living and skilled nursing centers whose residents are known to have high rates of hospital admissions.8,9 In these facilities, 24-hour medical assistance may not be available, and telemedicine can help troubleshoot common problems.
LOW REIMBURSEMENT RATES CURTAIL USE
Limited reimbursement has curtailed the widespread use of telemedicine. Although rules for reimbursement are evolving, telemedicine still represents a small amount of total healthcare expenditures. In 2015, Medicare spent approximately $14.4 million on services delivered via telemedicine—less than 0.01% of total spending on healthcare services.1
Currently, 31 states and the District of Columbia have telemedicine parity laws that mandate private commercial insurers to pay for telemedicine services.10 Unfortunately, there is a lack of uniformity in the specifics of these laws, resulting in variations in reimbursement rates. Furthermore, a large number of larger insurers such as Medicare and Medicaid and many self-insured plans do not fall under these mandates.
Another factor that affects reimbursement for telemedicine services is the setting of the medical encounter. Medicare reimburses providers for telemedicine services only when they are conducted at specific sites such as physician’s offices, hospitals, rural health centers, and skilled nursing facilities. Additionally, Medicare only reimburses for services in areas with a shortage of healthcare professionals and in non-metropolitan areas, which excludes many urban patients.11
In contrast, more commercial reimbursement is occurring for online urgent care, and options for commercial reimbursement of online behavioral services are being explored.
INTERSTATE LICENSURE ISSUES
Current licensure laws also limit the ability of many healthcare providers to offer telemedicine services. Federal law requires providers to be fully licensed to practice medicine in the state where the patient is physically located. In cases of health systems that have locations in more than one state, providers may need to apply for and pay to maintain multiple licenses (current interstate licensing laws vary across states).
Interstate licensure is one way to solve this problem. Thus far, a number of states have joined the Interstate Medical Licensure Compact that intends to allow physicians to obtain expedited licenses to practice in multiple states.12
The federal TELE-MED Act was introduced in 2015 but not passed. It proposed to “allow a Medicare provider to provide telemedicine services to a Medicare beneficiary who is in a different state from the one in which the provider is licensed or authorized to provide healthcare services.”
CAN TELEMEDICINE FOSTER A GOOD PROVIDER-PATIENT RELATIONSHIP?
In-person encounters provide healthcare providers with the opportunity to build a therapeutic relationship with their patients. Face-to-face encounters also increase patient satisfaction scores and outcomes. As such, critics fear that patient relationships may suffer with the use of telemedicine. However, using video technology for new patient encounters may help overcome this challenge. During a video encounter, the provider can see the patient’s facial expressions and take cues from nonverbal behaviors.
At times, the element of distance may enhance the encounter. For example, in behavioral health, patients often feel more comfortable in their home environment than in a sterile office environment.
Telemedicine patients often have positive experiences, given the speed of access, precision, time savings, and the ability to stay in contact with healthcare providers from the comfort of their homes. Ultimately, these virtual visits may help improve compliance with follow-up consultations since the barriers of distance and transportation are circumvented.
WHO CAN CONDUCT TELEMEDICINE VISITS?
Although a patient’s healthcare team is likely to consist of members who are not physicians, including nurse practitioners, physician assistants, social workers, and psychologists, not everyone can, by law, conduct telemedicine visits. Currently, the rules and regulations addressing ancillary team members’ participation in telemedicine vary from state to state.
TELEMEDICINE VISITS AT CLEVELAND CLINIC
Our health system has several telemedicine programs, including our eHospital program. Launched in 2014, this program provides patients at 4 hospitals with input from staff intensivists and experienced critical care nurses during the night (7 pm to 7 am) via remote monitoring. These remote caregivers have full access to patient charts and, when signalled, can activate an in-room camera to initiate 2-way audio communication with patients, their families, and bedside caregivers.
In addition, new patient consults are being offered via telemedicine for several services including dermatology, where pictures of skin lesions are reviewed and triaged, and management recommendations are provided accordingly.
In 2016, Cleveland Clinic launched its Remote Hypertension Improvement Program—an enterprise-wide initiative to minimize hypertension-associated mortality and morbidity with the assistance of telehealth services. The program was first piloted in a group of 80 high-risk hypertensive patients who were monitored and followed through a Bluetooth-enabled remote monitoring tool, which exported blood pressure readings to a central dashboard. A multidisciplinary team of doctors, nurses, and pharmacists used this dashboard to adjust medication when needed and provide virtual lifestyle coaching. Over a 24-week period, the patients’ systolic blood pressure decreased by an average of 7.5 mm Hg and diastolic blood pressure by 3.1 mm Hg (unpublished data).
Beginning this year, blood pressure readings will be directly exported from the remote monitoring tool into the patient’s electronic medical record, providing the healthcare team with the information needed to make informed decisions to remotely manage patients with hypertension.
Remote monitoring of patients with hypertension is also being used at other institutions such as the VA. In 2016, almost 19,000 veterans were using the remote monitoring system, and this number is expected to increase with the enhanced adaptation of telemedicine services.13
FUTURE DIRECTIONS
About 50% of all adults in the United States have at least 1 chronic disease. In all, chronic disease accounts for roughly 75% of the total healthcare expenditure and 70% of all deaths.7,14 Recent data suggest that virtual chronic disease management represents an untapped market for telemedicine, given its relative underutilization compared to other services such as telebehavorial health and specialty telemedicine. These patients require frequent visits to the doctor, and targeting this patient population with telemedicine may decrease the number of emergency room visits and hospital admissions.
Another growing area in the field of telemedicine is the “hospital at home” model in which patients who meet the criteria for hospitalization but are otherwise stable are treated at home for diseases such as chronic obstructive pulmonary disease, pneumonia, and heart failure. Studies have shown that the hospital-at-home model, when used appropriately, is not only more cost-effective than hospitalization but results in a shorter treatment duration and lower rates of delirium.15–17
Finally, in the acute setting, we have seen wide success with telemedicine programs in stroke care, radiology, intensive care, and psychiatry, and several studies have shown mortality rates comparable to those with the traditional model.18,19 These encounters often require specialized skills and are the focus of multiple ongoing studies.
Acknowledgment: The authors would like to acknowledge and thank Matthew Faiman, MD, for providing information regarding the Remote Hypertension Program.
- US Department of Health and Human Services. Report to Congress: e-health and telemedicine. aspe.hhs.gov/system/files/pdf/206751/TelemedicineE-HealthReport.pdf. Accessed September 1, 2018.
- World Health Organization (WHO). A Health Telematics Policy in Support of WHO’s Health-For-All Strategy for Global Health Development: Report of the WHO Group Consultation on Health Telematics, 11–16 December, Geneva 1997. World Health Organization, Geneva, 1998.
- Bashshur RL, Shannon GW. History of telemedicine: evolution, context, and transformation. Mary Ann Liebert, Inc.: New Rochelle (NY), 2009.
- Bashshur RL, Goldberg MA. The origins of telemedicine and e-Health. Telemed J E Health 2014; 20(3):190–191. doi:10.1089/tmj.2014.9996
- Bashshur RL, Shannon G, Krupinski EA, Grigsby J. Sustaining and realizing the promise of telemedicine. Telemed J E Health 2013; 19(5):339–345. doi:10.1089/tmj.2012.0282
- American Hospital Association (AHA). Issue Brief. Telehealth: helping hospitals deliver cost-effective care. www.aha.org/system/files/content/16/16telehealthissuebrief.pdf. Accessed September 10, 2018.
- Congressional Research Service. Telehealth and Telemedicine: description and issues. March 29, 2016. www.senate.gov/CRSpubs/757e3b90-ff10-497c-8e8c-ac1bdbdb3aaf.pdf. Accessed August 8, 2018.
- Grabowski DC, Stewart KA, Broderick SM, Coots LA. Predictors of nursing home hospitalization: a review of the literature. Med Care Res Rev 2008; 65(1):3–39. doi:10.1177/1077558707308754
- Grabowski DC, O’Malley AJ. Use of telemedicine can reduce hospitalizations of nursing home residents and generate savings for Medicare. Health Aff (Millwood) 2014; 33(2):244–250. doi:10.1377/hlthaff.2013.0922
- Jones K. If not parity, clarity—getting doctors paid for telehealth. www.forbes.com/sites/realspin/2016/09/15/if-not-parity-clarity-getting-doctors-paid-for-telehealth/#43928587777f. Accessed September 1, 2018.
- Neufeld JD, Doarn CR. Telemedicine spending by Medicare: a snapshot from 2012. Telemed J E Health 2015; 21(8):686–693. doi:10.1089/tmj.2014.0185
- Chaudhry HJ, Robin LA, Fish EM, Polk DH, Gifford JD. Improving access and mobility—the Interstate Medical Licensure Compact. N Engl J Med 2015; 372(17):1581–1583. doi:10.1056/NEJMp1502639
- United States Government Accountability Office. Report to Congressional Committees. Healthcare: telehealth and remote patient monitoring use in Medicare and selected federal programs. www.gao.gov/assets/690/684115.pdf. Accessed September 1, 2018.
- Bashshur RL, Shannon GW, Smith BR, et al. The empirical foundations of telemedicine interventions for chronic disease management. Telemed J E Health 2014; 20(9):769–800. doi:10.1089/tmj.2014.9981
- Cryer L, Shannon SB, Van Amsterdam M, Leff B. Costs for ‘hospital at home’ patients were 19 percent lower, with equal or better outcomes compared to similar inpatients. Health Aff (Millwood) 2012; 31:1237–1243. doi:10.1377/hlthaff.2011.1132
- Leff B, Burton L, Mader SL, et al. Hospital at home: feasibility and outcomes of a program to provide hospital-level care at home for acutely ill older patients. Ann Intern Med 2005; 143(11):798–808. pmid:16330791
- Leff B, Soones T, DeCherrie L. The hospital at home program for older adults. JAMA Intern Med 2016; 176(11):1724–1725. doi:10.1001/jamainternmed.2016.6307
- Wechsler LR, Demaerschalk BM, Schwamm LH, et al; American Heart Association Stroke Council; Council on Epidemiology and Prevention; Council on Quality of Care and Outcomes Research. Telemedicine quality and outcomes in stroke: a scientific statement for healthcare professionals from the American Heart Association/American Stroke Association. Stroke 2017; 48(1):e3–e25. doi:10.1161/STR.0000000000000114
- Wilcox ME, Wiener-Kronish JP. Telemedicine in the intensive care unit: effect of a remote intensivist on outcomes. JAMA Intern Med 2014; 174(7):1167–1169. doi:10.1001/jamainternmed.2014.289
- US Department of Health and Human Services. Report to Congress: e-health and telemedicine. aspe.hhs.gov/system/files/pdf/206751/TelemedicineE-HealthReport.pdf. Accessed September 1, 2018.
- World Health Organization (WHO). A Health Telematics Policy in Support of WHO’s Health-For-All Strategy for Global Health Development: Report of the WHO Group Consultation on Health Telematics, 11–16 December, Geneva 1997. World Health Organization, Geneva, 1998.
- Bashshur RL, Shannon GW. History of telemedicine: evolution, context, and transformation. Mary Ann Liebert, Inc.: New Rochelle (NY), 2009.
- Bashshur RL, Goldberg MA. The origins of telemedicine and e-Health. Telemed J E Health 2014; 20(3):190–191. doi:10.1089/tmj.2014.9996
- Bashshur RL, Shannon G, Krupinski EA, Grigsby J. Sustaining and realizing the promise of telemedicine. Telemed J E Health 2013; 19(5):339–345. doi:10.1089/tmj.2012.0282
- American Hospital Association (AHA). Issue Brief. Telehealth: helping hospitals deliver cost-effective care. www.aha.org/system/files/content/16/16telehealthissuebrief.pdf. Accessed September 10, 2018.
- Congressional Research Service. Telehealth and Telemedicine: description and issues. March 29, 2016. www.senate.gov/CRSpubs/757e3b90-ff10-497c-8e8c-ac1bdbdb3aaf.pdf. Accessed August 8, 2018.
- Grabowski DC, Stewart KA, Broderick SM, Coots LA. Predictors of nursing home hospitalization: a review of the literature. Med Care Res Rev 2008; 65(1):3–39. doi:10.1177/1077558707308754
- Grabowski DC, O’Malley AJ. Use of telemedicine can reduce hospitalizations of nursing home residents and generate savings for Medicare. Health Aff (Millwood) 2014; 33(2):244–250. doi:10.1377/hlthaff.2013.0922
- Jones K. If not parity, clarity—getting doctors paid for telehealth. www.forbes.com/sites/realspin/2016/09/15/if-not-parity-clarity-getting-doctors-paid-for-telehealth/#43928587777f. Accessed September 1, 2018.
- Neufeld JD, Doarn CR. Telemedicine spending by Medicare: a snapshot from 2012. Telemed J E Health 2015; 21(8):686–693. doi:10.1089/tmj.2014.0185
- Chaudhry HJ, Robin LA, Fish EM, Polk DH, Gifford JD. Improving access and mobility—the Interstate Medical Licensure Compact. N Engl J Med 2015; 372(17):1581–1583. doi:10.1056/NEJMp1502639
- United States Government Accountability Office. Report to Congressional Committees. Healthcare: telehealth and remote patient monitoring use in Medicare and selected federal programs. www.gao.gov/assets/690/684115.pdf. Accessed September 1, 2018.
- Bashshur RL, Shannon GW, Smith BR, et al. The empirical foundations of telemedicine interventions for chronic disease management. Telemed J E Health 2014; 20(9):769–800. doi:10.1089/tmj.2014.9981
- Cryer L, Shannon SB, Van Amsterdam M, Leff B. Costs for ‘hospital at home’ patients were 19 percent lower, with equal or better outcomes compared to similar inpatients. Health Aff (Millwood) 2012; 31:1237–1243. doi:10.1377/hlthaff.2011.1132
- Leff B, Burton L, Mader SL, et al. Hospital at home: feasibility and outcomes of a program to provide hospital-level care at home for acutely ill older patients. Ann Intern Med 2005; 143(11):798–808. pmid:16330791
- Leff B, Soones T, DeCherrie L. The hospital at home program for older adults. JAMA Intern Med 2016; 176(11):1724–1725. doi:10.1001/jamainternmed.2016.6307
- Wechsler LR, Demaerschalk BM, Schwamm LH, et al; American Heart Association Stroke Council; Council on Epidemiology and Prevention; Council on Quality of Care and Outcomes Research. Telemedicine quality and outcomes in stroke: a scientific statement for healthcare professionals from the American Heart Association/American Stroke Association. Stroke 2017; 48(1):e3–e25. doi:10.1161/STR.0000000000000114
- Wilcox ME, Wiener-Kronish JP. Telemedicine in the intensive care unit: effect of a remote intensivist on outcomes. JAMA Intern Med 2014; 174(7):1167–1169. doi:10.1001/jamainternmed.2014.289
KEY POINTS
- An estimated 7 million patients in the United States will use telemedicine services this year alone; demand will continue to rise.
- Low reimbursement rates and current lack of interstate licensure laws limit the ability of many health care providers to offer telemedicine services.
- The rules and regulations addressing ancillary team members’ participation in telemedicine vary from state to state.
- Areas of future growth include chronic disease management and “hospital at home” care.
Stroke management and the impact of mobile stroke treatment units
Stroke is the fifth leading cause of death in the United States. Approximately 795,000 strokes occur every year and about 130,000 patients die.1 The impact of stroke-related medical costs and disability is significant, making it a key target for treatment and prevention strategies.
Stroke is defined as an acute loss of neurologic function caused by damaged brain tissue. There are two primary types: ischemic and hemorrhagic. Ischemic strokes are by far the most common, accounting for 87% of all strokes.2 An ischemic stroke is caused by an arterial occlusion that restricts cerebral blood flow; a hemorrhagic stroke is caused by a rupture or leak in the cerebrovasculature. Treatment of an ischemic stroke focuses on thrombolysis and revascularization strategies to restore blood flow, whereas with hemorrhagic stroke, treatment focuses on controlling intracerebral bleeding, elevated intracranial pressure, and secondary brain injury. This article addresses a key factor in improved stroke outcomes—time to treatment—and the impact that a mobile stroke treatment unit (MSTU) can have on this factor.
DECLINING MORBIDITY AND MORTALITY RATES
Although the morbidity and mortality associated with stroke are high, the rates have been declining. From 2001 o 2011, the stroke mortality rate declined by 35%.2 The American Heart Association attributes the reduction to improvements in both prevention and treatment.
A significant portion of the decline has come from population-wide stroke prevention efforts. These include community efforts to control the major cardiovascular risk factors for stroke, including hypertension and hypercholesterolemia. Treating hypertension can reduce the incidence of stroke by up to 40%.3 In addition, community education efforts aimed at improving awareness of stroke symptoms and early detection have contributed to the declining rates, although, by some estimates, only about one-third of the population knows the major signs and symptoms of stroke.
Improved stroke treatments have also contributed to better outcomes, primarily through the more widespread use of thrombolytics. When first approved by the US Food and Drug Administration (FDA), thrombolytics were primarily the purview of cardiologists. However, as outcomes data accumulated, neurologists recognized the utility of thrombolytics in treating ischemic cerebrovascular disease and began investigating their use in clinical trials. Positive outcomes from those trials led to their FDA approval for stroke treatment and universal recognition as the primary therapy for acute stroke. More recent efforts have concentrated on early treatment by bringing the therapy to the patient as opposed to the traditional treatment algorithm of providing care in the emergency department. If therapy is instituted quickly enough, ischemic stroke symptoms can be reversed.
TIME TO TREATMENT
Therapeutic use of tissue plasminogen activators (tPA) has had a major impact on morbidity and mortality in patients with acute ischemic strokes. The efficacy of tPA as thrombolytic therapy in this patient population is well documented.4
Also well documented is the significant impact of time-to-tPA treatment on outcomes. If therapy is started within 3 to 4.5 hours of ischemic stroke onset, patients have improved functional outcomes 3 to 6 months after the incident (Figure 1). Between 31% and 50% of patients treated with tPA within 3 hours experienced improved recovery at 3 months compared with 20% to 38% of patients treated with placebo.5–9 Faster onset to treatment, measured in 15-minute increments, has been shown to significantly reduce in-hospital mortality, reduce intracranial hemorrhage, increase ability to walk at discharge, and increase number discharged to home.6 Even a 1-minute delay in time-to-tPA treatment has a substantial impact on rates of morbidity and mortality (Table 1).10 National and international guidelines recommend starting intravenous tPA within 1 hour of patient arrival in the emergency department and not longer than 4.5 hours since symptom onset, although some evidence indicates a 3-hour window.5,11,12
Although the evidence supports the benefit of rapid therapy for acute ischemic stroke, the national percentage of patients who actually receive tPA within the therapeutic window is small, by some estimates as low as 3% to 5%.13 For optimal stroke care, the rate should be 30% to 50%.
IMPROVING TREATMENT TIMES
Studies have found that the major reason patients do not receive tPA is that they do not reach the hospital quickly enough to be assessed and treated within the treatment window.14,15 In essence, neurologists have the technology to treat most patients, but are waiting for the patients to arrive. Many factors contribute to this delayed arrival time. On the patient level, the primary factors are related to failure to recognize stroke symptoms as well as failure to understand their seriousness.
From the healthcare provider’s perspective, a major barrier to reducing the time-to-treatment window is the need to accurately assess patients with acute ischemic stroke who are eligible for thrombolytic therapy. This is difficult to achieve in clinical practice because it requires neurologic imaging primarily with computed tomography (CT) or magnetic resonance imaging (MRI) and laboratory analyses so that hemorrhagic stroke and other contraindications to thrombolysis can be excluded. Traditionally, this type of analytic equipment had been available only in emergency departments, requiring patients to be brought to those facilities.
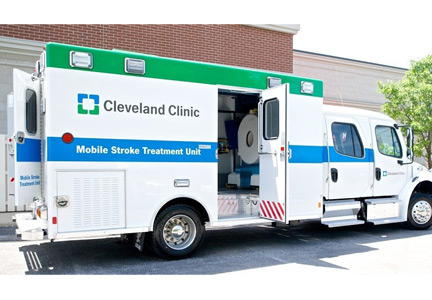
Recent innovation in this area led to the development of specialized ambulances equipped with a CT scanner, point-of-care laboratory equipment, and telemedicine connection along with the appropriate treatment options/medications and trained healthcare personnel to provide prehospital stroke treatment. These specially equipped ambulances are known as MSTUs or stroke emergency mobile (STEMO) units. Their development has dramatically altered the strategy from one of taking the patient to the treatment to taking the treatment to the patient.
MOBILE STROKE TREATMENT UNITS
Two technological innovations have been fundamental to the creation and success of MSTU: portable CT scanners and high-speed wireless data transmission.
CT scanners. A key element was the development of a portable diagnostic-quality head and neck CT scanner that can be fit inside a typical-sized ambulance. This 8-slice CT scanner is capable of creating the same scan types and quality found in radiology department CT scanners, including axial imaging, helical angiography, and perfusion imaging. The resolution and slice thickness (1.25 mm) of the images are of suitable quality to enable neurologists and neuroradiologists to exclude hemorrhage in acute stroke, to assess the degree of brain injury, and to identify the vascular lesion responsible for the ischemic deficit. These technologies also enable diagnostic differentiation between brain tissue that is irreversibly infarcted from that which is potentially salvageable, thereby allowing more accurate patient assessment. The imaging data currently obtainable by CT scanners fitted on ambulances is only likely to improve with future technological advances.
Wireless data transmission. Cellular wireless providers have developed the technology and equipment to provide high-speed wireless broadband capable of transmitting high-quality CT and MRI images. It also enables encrypted feed of video telemedicine, data transmission, and download of patient data. This allows the MSTU to electronically sit inside the firewalls of healthcare facilities, providing access to the patients’ electronic health records and to on-site stroke experts.
The successes have been impressive. Studies have found that the deployment of an MSTU significantly reduces the median time from 9-1-1 alarm to intravenous thrombolysis without increasing adverse events.16–19 These data are primarily from the PHANTOM-S study, a pilot program conducted in Germany.18,19 Results showed a significant reduction in alarm-to-treatment times, from 76 minutes in the hospital control group to 52 minutes in the MSTU group (Table 2).17,19 Further, among patients who suffered an ischemic stroke, the proportion who received tPA within 1 hour of symptom onset was sixfold higher after MSTU deployment (Table 3).18 In a separate European study, prehospital stroke assessment using an MSTU significantly reduced the median time from alarm-to-therapy decision: 35 minutes versus 76 in the hospital group.16
The prehospital cerebrovascular diagnostic workup provided by an MSTU also can improve the emergency management of other stroke types. By providing more diagnostic data and higher quality imaging, the units improve the accuracy of the diagnosis. In turn, this enables emergency personnel to provide accurate therapy and to transfer patients to hospitals with the appropriate level of stroke care, decreasing the need for additional intrahospital transfers.20
Overall, it has been shown that an MSTU equipped with the necessary imaging and laboratory testing equipment can provide appropriate, accurate, and safe ambulance-based prehospital tPA administration, reduce the time to tPA administration, and increase the number of patients who receive tPA administration. All of these factors combine to improve outcomes in patients with acute ischemic stroke.
CLEVELAND CLINIC EXPERIENCE
Cleveland Clinic has a tradition of providing high-quality and innovative stroke care. Recognizing the importance of an appropriately equipped MSTU in reducing the time to stroke treatment, especially tPA administration, Cleveland Clinic instituted a plan to develop an MSTU for the care of patients in the Cleveland area. The development required several planning, funding, and development phases.
Planning. Establishing relations with both city planners and area hospitals was central to planning the MSTU startup. An agreement with the city of Cleveland included creating an emergency medical system (EMS) triage algorithm for the 9-1-1 dispatch center. When a call is received, the dispatcher uses a stroke checklist to perform an initial screening. If a stroke is suspected, the MSTU is dispatched along with a Cleveland EMS or other first-responder unit.
As part of the agreement, Cleveland officials required that the MSTU treat all patients, regardless of their ability to pay. This requirement has been beneficial to the MSTU mission as it allows for treating more patients with tPA as quickly as possible without concern for health insurance, which maximizes the potential for neurologic recovery.
Staffing and procedures. The MSTU staff is composed of a paramedic, a critical care nurse, a CT technologist, and an emergency medicine technician/EMS driver. They perform CT scans and point-of-care laboratory tests on patients who have stroke symptoms. The CT scans and laboratory results are wirelessly transmitted to Cleveland Clinic. A neurologist assesses the data, consults with the MSTU staff on history and neurologic examination, and diagnoses the patient remotely. Patients are then transported to the closest hospital with the resources to meet their clinical needs. If thrombolytic therapy is indicated, intravenous tPA is initiated immediately at the scene. If the patient has sustained a hemorrhagic stroke, reversal of anticoagulation therapy is initiated, if indicated.
Outcomes. The success rates also have been impressive, with dramatic reductions in time to treatment. On average, patients received tPA 40 minutes faster in the MSTU model than in the standard model of ambulance transport and in-hospital evaluation and treatment: 64 minutes versus 104 minutes. Further, more patients in the MSTU group received tPA: 26% versus 14%. Results also showed a 21-minute reduction in time-to-CT completion, an important aspect of providing more timely care.21–23 This CT scanner is also capable of CT angiography. This enables large-vessel occlusion strokes to be identified in the field. When these types of strokes are identified in the field, the patients are transported directly to a stroke center capable of endovascular therapy, even bypassing some primary stroke centers.
Using the MSTU to bring diagnostic and stroke care to the patient has shown that the time between the onset of stroke-like symptoms and the delivery of treatment can be reduced. Thus, an MSTU has the potential to minimize the mortality and long-term morbidity associated with strokes.
- Centers for Disease Control and Prevention (CDC). Prevalence of stroke— United States, 2006–2010. MMWR Morb Mortal Wkly Rep 2012; 61:379–382.
- Mozaffarian D, Benjamin EJ, Go AS, et al. Heart disease and stroke statistics—2015 update: a report from the American Heart Association. Circulation 2015; 131:e29–e322.
- Howard G, Banach M, Cushman M, et al. Is blood pressure control for stroke prevention the correct goal? The lost opportunity of preventing hypertension. Stroke 2015; 46:1595–1600.
- Tissue plasminogen activator for acute ischemic stroke. The National Institute of Neurological Disorders and Stroke rt-PA Stroke Study Group. N Engl J Med 1995; 333:1581–1587.
- Lees KR, Bluhmki E, von Kummer R, et al; ECASS, ATLANTIS, NINDS, and EPITHET rt-PA Study Group. Time to treatment with intravenous alteplase and outcome in stroke: an updated pooled analysis of ECASS, ATLANTIS, NINDS, and EPITHET trials. Lancet 2010; 375:1695–1703.
- Saver JL, Fonarow GC, Smith EE, et al. Time to treatment with intravenous tissue plasminogen activator and outcome from acute ischemic stroke. JAMA 2013; 309:2480–2488.
- Hacke W, Donnan G, Fieschi C, et al; ATLANTIS Trials Investigators; ECASS Trials Investigators; NINDS rt-PA Study Group Investigators. Association of outcome with early stroke treatment: pooled analysis of ATLANTIS, ECASS, and NINDS rt-PA stroke trials. Lancet 2004; 363:768–774.
- Wardlaw JM, Murray V, Berge E, et al. Recombinant tissue plasminogen activator for acute ischaemic stroke: an updated systematic review and meta-analysis. Lancet 2012; 379:2364–2372.
- Prabhakaran S, Ruff I, Bernstein RA. Acute stroke intervention: a systematic review. JAMA 2015; 313:1451–1462.
- Saver JL. Time is brain—quantified. Stroke 2006; 37:263–266.
- Jauch EC, Saver JL, Adams HP Jr, et al; American Heart Association Stroke Council; Council on Cardiovascular Nursing; Council on Peripheral Vascular Disease; Council on Clinical Cardiology. Guidelines for the early management of patients with acute ischemic stroke: a guideline for healthcare professionals from the American Heart Association/American Stroke Association. Stroke 2013; 44:870–947.
- Hacke W, Kaste M, Bluhmki E, et al; ECASS Investigators. Thrombolysis with alteplase 3 to 4.5 hours after acute ischemic stroke. N Engl J Med 2008; 359:1317– 1329.
- Adeoye O, Hornung R, Khatri P, Kleindorfer D. Recombinant tissue-type plasminogen activator use for ischemic stroke in the United States: a doubling of treatment rates over the course of 5 years. Stroke 2011; 42:1952–1955.
- Barber PA, Zhang J, Demchuk AM, Hill MD, Buchan AM. Why are stroke patients excluded from TPA therapy? An analysis of patient eligibility. Neurology 2001; 56:1015–1020.
- Lichtman JH, Watanabe E, Allen NB, Jones SB, Dostal J, Goldstein LB. Hospital arrival time and intravenous t-PA use in US Academic Medical Centers, 2001– 2004. Stroke 2009; 40:3845–3850.
- Walter S, Kostopoulos P, Haass A, et al. Diagnosis and treatment of patients with stroke in a mobile stroke unit versus in hospital: a randomised controlled trial. Lancet Neurol 2012; 11:397–404.
- Ebinger M, Winter B, Wendt M, et al; STEMO Consortium. Effect of the use of ambulance-based thrombolysis on time to thrombolysis in acute ischemic stroke: a randomized clinical trial. JAMA 2014; 311:1622–1631.
- Ebinger M, Kunz A, Wendt M, et al. Effects of golden hour thrombolysis: a prehospital acute neurological treatment and optimization of medical care in stroke (PHANTOM-S) substudy. JAMA Neurol 2015; 72:25–30.
- Weber JE, Ebinger M, Rozansk M, et al; STEMO-Consortium. Prehospital thrombolysis in acute stroke: results of the PHANTOM-S pilot study. Neurology 2013; 80:163–168.
- Wendt M, Ebinger M, Kunz A, et al; STEMO Consortium. Improved prehospital triage of patients with stroke in a specialized stroke ambulance: results of the prehospital acute neurological therapy and optimization of medical care in stroke study. Stroke 2015; 46: 740–745.
- Cho S, Cerejo R, Taqui A, et al. Feasibility of telemedicine on a mobile stroke treatment unit. Stroke 2015; 46:ATP200.
- Sheikhi L, Itrat A, Cerejo R, et al. Does portable CT imaging in a mobile stroke treatment unit (MSTU) provide adequate quality for early critical decision making? Stroke 2015; 46:AWP31.
- Taqui A, Cerejo R, Itrat A, et al. Reduction in time to imaging and intravenous thrombolysis by in-field evaluation and treatment in a mobile stroke treatment unit. Stroke 2015; 46:A54.
Stroke is the fifth leading cause of death in the United States. Approximately 795,000 strokes occur every year and about 130,000 patients die.1 The impact of stroke-related medical costs and disability is significant, making it a key target for treatment and prevention strategies.
Stroke is defined as an acute loss of neurologic function caused by damaged brain tissue. There are two primary types: ischemic and hemorrhagic. Ischemic strokes are by far the most common, accounting for 87% of all strokes.2 An ischemic stroke is caused by an arterial occlusion that restricts cerebral blood flow; a hemorrhagic stroke is caused by a rupture or leak in the cerebrovasculature. Treatment of an ischemic stroke focuses on thrombolysis and revascularization strategies to restore blood flow, whereas with hemorrhagic stroke, treatment focuses on controlling intracerebral bleeding, elevated intracranial pressure, and secondary brain injury. This article addresses a key factor in improved stroke outcomes—time to treatment—and the impact that a mobile stroke treatment unit (MSTU) can have on this factor.
DECLINING MORBIDITY AND MORTALITY RATES
Although the morbidity and mortality associated with stroke are high, the rates have been declining. From 2001 o 2011, the stroke mortality rate declined by 35%.2 The American Heart Association attributes the reduction to improvements in both prevention and treatment.
A significant portion of the decline has come from population-wide stroke prevention efforts. These include community efforts to control the major cardiovascular risk factors for stroke, including hypertension and hypercholesterolemia. Treating hypertension can reduce the incidence of stroke by up to 40%.3 In addition, community education efforts aimed at improving awareness of stroke symptoms and early detection have contributed to the declining rates, although, by some estimates, only about one-third of the population knows the major signs and symptoms of stroke.
Improved stroke treatments have also contributed to better outcomes, primarily through the more widespread use of thrombolytics. When first approved by the US Food and Drug Administration (FDA), thrombolytics were primarily the purview of cardiologists. However, as outcomes data accumulated, neurologists recognized the utility of thrombolytics in treating ischemic cerebrovascular disease and began investigating their use in clinical trials. Positive outcomes from those trials led to their FDA approval for stroke treatment and universal recognition as the primary therapy for acute stroke. More recent efforts have concentrated on early treatment by bringing the therapy to the patient as opposed to the traditional treatment algorithm of providing care in the emergency department. If therapy is instituted quickly enough, ischemic stroke symptoms can be reversed.
TIME TO TREATMENT
Therapeutic use of tissue plasminogen activators (tPA) has had a major impact on morbidity and mortality in patients with acute ischemic strokes. The efficacy of tPA as thrombolytic therapy in this patient population is well documented.4
Also well documented is the significant impact of time-to-tPA treatment on outcomes. If therapy is started within 3 to 4.5 hours of ischemic stroke onset, patients have improved functional outcomes 3 to 6 months after the incident (Figure 1). Between 31% and 50% of patients treated with tPA within 3 hours experienced improved recovery at 3 months compared with 20% to 38% of patients treated with placebo.5–9 Faster onset to treatment, measured in 15-minute increments, has been shown to significantly reduce in-hospital mortality, reduce intracranial hemorrhage, increase ability to walk at discharge, and increase number discharged to home.6 Even a 1-minute delay in time-to-tPA treatment has a substantial impact on rates of morbidity and mortality (Table 1).10 National and international guidelines recommend starting intravenous tPA within 1 hour of patient arrival in the emergency department and not longer than 4.5 hours since symptom onset, although some evidence indicates a 3-hour window.5,11,12
Although the evidence supports the benefit of rapid therapy for acute ischemic stroke, the national percentage of patients who actually receive tPA within the therapeutic window is small, by some estimates as low as 3% to 5%.13 For optimal stroke care, the rate should be 30% to 50%.
IMPROVING TREATMENT TIMES
Studies have found that the major reason patients do not receive tPA is that they do not reach the hospital quickly enough to be assessed and treated within the treatment window.14,15 In essence, neurologists have the technology to treat most patients, but are waiting for the patients to arrive. Many factors contribute to this delayed arrival time. On the patient level, the primary factors are related to failure to recognize stroke symptoms as well as failure to understand their seriousness.
From the healthcare provider’s perspective, a major barrier to reducing the time-to-treatment window is the need to accurately assess patients with acute ischemic stroke who are eligible for thrombolytic therapy. This is difficult to achieve in clinical practice because it requires neurologic imaging primarily with computed tomography (CT) or magnetic resonance imaging (MRI) and laboratory analyses so that hemorrhagic stroke and other contraindications to thrombolysis can be excluded. Traditionally, this type of analytic equipment had been available only in emergency departments, requiring patients to be brought to those facilities.
Recent innovation in this area led to the development of specialized ambulances equipped with a CT scanner, point-of-care laboratory equipment, and telemedicine connection along with the appropriate treatment options/medications and trained healthcare personnel to provide prehospital stroke treatment. These specially equipped ambulances are known as MSTUs or stroke emergency mobile (STEMO) units. Their development has dramatically altered the strategy from one of taking the patient to the treatment to taking the treatment to the patient.
MOBILE STROKE TREATMENT UNITS
Two technological innovations have been fundamental to the creation and success of MSTU: portable CT scanners and high-speed wireless data transmission.
CT scanners. A key element was the development of a portable diagnostic-quality head and neck CT scanner that can be fit inside a typical-sized ambulance. This 8-slice CT scanner is capable of creating the same scan types and quality found in radiology department CT scanners, including axial imaging, helical angiography, and perfusion imaging. The resolution and slice thickness (1.25 mm) of the images are of suitable quality to enable neurologists and neuroradiologists to exclude hemorrhage in acute stroke, to assess the degree of brain injury, and to identify the vascular lesion responsible for the ischemic deficit. These technologies also enable diagnostic differentiation between brain tissue that is irreversibly infarcted from that which is potentially salvageable, thereby allowing more accurate patient assessment. The imaging data currently obtainable by CT scanners fitted on ambulances is only likely to improve with future technological advances.
Wireless data transmission. Cellular wireless providers have developed the technology and equipment to provide high-speed wireless broadband capable of transmitting high-quality CT and MRI images. It also enables encrypted feed of video telemedicine, data transmission, and download of patient data. This allows the MSTU to electronically sit inside the firewalls of healthcare facilities, providing access to the patients’ electronic health records and to on-site stroke experts.
The successes have been impressive. Studies have found that the deployment of an MSTU significantly reduces the median time from 9-1-1 alarm to intravenous thrombolysis without increasing adverse events.16–19 These data are primarily from the PHANTOM-S study, a pilot program conducted in Germany.18,19 Results showed a significant reduction in alarm-to-treatment times, from 76 minutes in the hospital control group to 52 minutes in the MSTU group (Table 2).17,19 Further, among patients who suffered an ischemic stroke, the proportion who received tPA within 1 hour of symptom onset was sixfold higher after MSTU deployment (Table 3).18 In a separate European study, prehospital stroke assessment using an MSTU significantly reduced the median time from alarm-to-therapy decision: 35 minutes versus 76 in the hospital group.16
The prehospital cerebrovascular diagnostic workup provided by an MSTU also can improve the emergency management of other stroke types. By providing more diagnostic data and higher quality imaging, the units improve the accuracy of the diagnosis. In turn, this enables emergency personnel to provide accurate therapy and to transfer patients to hospitals with the appropriate level of stroke care, decreasing the need for additional intrahospital transfers.20
Overall, it has been shown that an MSTU equipped with the necessary imaging and laboratory testing equipment can provide appropriate, accurate, and safe ambulance-based prehospital tPA administration, reduce the time to tPA administration, and increase the number of patients who receive tPA administration. All of these factors combine to improve outcomes in patients with acute ischemic stroke.
CLEVELAND CLINIC EXPERIENCE
Cleveland Clinic has a tradition of providing high-quality and innovative stroke care. Recognizing the importance of an appropriately equipped MSTU in reducing the time to stroke treatment, especially tPA administration, Cleveland Clinic instituted a plan to develop an MSTU for the care of patients in the Cleveland area. The development required several planning, funding, and development phases.
Planning. Establishing relations with both city planners and area hospitals was central to planning the MSTU startup. An agreement with the city of Cleveland included creating an emergency medical system (EMS) triage algorithm for the 9-1-1 dispatch center. When a call is received, the dispatcher uses a stroke checklist to perform an initial screening. If a stroke is suspected, the MSTU is dispatched along with a Cleveland EMS or other first-responder unit.
As part of the agreement, Cleveland officials required that the MSTU treat all patients, regardless of their ability to pay. This requirement has been beneficial to the MSTU mission as it allows for treating more patients with tPA as quickly as possible without concern for health insurance, which maximizes the potential for neurologic recovery.
Staffing and procedures. The MSTU staff is composed of a paramedic, a critical care nurse, a CT technologist, and an emergency medicine technician/EMS driver. They perform CT scans and point-of-care laboratory tests on patients who have stroke symptoms. The CT scans and laboratory results are wirelessly transmitted to Cleveland Clinic. A neurologist assesses the data, consults with the MSTU staff on history and neurologic examination, and diagnoses the patient remotely. Patients are then transported to the closest hospital with the resources to meet their clinical needs. If thrombolytic therapy is indicated, intravenous tPA is initiated immediately at the scene. If the patient has sustained a hemorrhagic stroke, reversal of anticoagulation therapy is initiated, if indicated.
Outcomes. The success rates also have been impressive, with dramatic reductions in time to treatment. On average, patients received tPA 40 minutes faster in the MSTU model than in the standard model of ambulance transport and in-hospital evaluation and treatment: 64 minutes versus 104 minutes. Further, more patients in the MSTU group received tPA: 26% versus 14%. Results also showed a 21-minute reduction in time-to-CT completion, an important aspect of providing more timely care.21–23 This CT scanner is also capable of CT angiography. This enables large-vessel occlusion strokes to be identified in the field. When these types of strokes are identified in the field, the patients are transported directly to a stroke center capable of endovascular therapy, even bypassing some primary stroke centers.
Using the MSTU to bring diagnostic and stroke care to the patient has shown that the time between the onset of stroke-like symptoms and the delivery of treatment can be reduced. Thus, an MSTU has the potential to minimize the mortality and long-term morbidity associated with strokes.
Stroke is the fifth leading cause of death in the United States. Approximately 795,000 strokes occur every year and about 130,000 patients die.1 The impact of stroke-related medical costs and disability is significant, making it a key target for treatment and prevention strategies.
Stroke is defined as an acute loss of neurologic function caused by damaged brain tissue. There are two primary types: ischemic and hemorrhagic. Ischemic strokes are by far the most common, accounting for 87% of all strokes.2 An ischemic stroke is caused by an arterial occlusion that restricts cerebral blood flow; a hemorrhagic stroke is caused by a rupture or leak in the cerebrovasculature. Treatment of an ischemic stroke focuses on thrombolysis and revascularization strategies to restore blood flow, whereas with hemorrhagic stroke, treatment focuses on controlling intracerebral bleeding, elevated intracranial pressure, and secondary brain injury. This article addresses a key factor in improved stroke outcomes—time to treatment—and the impact that a mobile stroke treatment unit (MSTU) can have on this factor.
DECLINING MORBIDITY AND MORTALITY RATES
Although the morbidity and mortality associated with stroke are high, the rates have been declining. From 2001 o 2011, the stroke mortality rate declined by 35%.2 The American Heart Association attributes the reduction to improvements in both prevention and treatment.
A significant portion of the decline has come from population-wide stroke prevention efforts. These include community efforts to control the major cardiovascular risk factors for stroke, including hypertension and hypercholesterolemia. Treating hypertension can reduce the incidence of stroke by up to 40%.3 In addition, community education efforts aimed at improving awareness of stroke symptoms and early detection have contributed to the declining rates, although, by some estimates, only about one-third of the population knows the major signs and symptoms of stroke.
Improved stroke treatments have also contributed to better outcomes, primarily through the more widespread use of thrombolytics. When first approved by the US Food and Drug Administration (FDA), thrombolytics were primarily the purview of cardiologists. However, as outcomes data accumulated, neurologists recognized the utility of thrombolytics in treating ischemic cerebrovascular disease and began investigating their use in clinical trials. Positive outcomes from those trials led to their FDA approval for stroke treatment and universal recognition as the primary therapy for acute stroke. More recent efforts have concentrated on early treatment by bringing the therapy to the patient as opposed to the traditional treatment algorithm of providing care in the emergency department. If therapy is instituted quickly enough, ischemic stroke symptoms can be reversed.
TIME TO TREATMENT
Therapeutic use of tissue plasminogen activators (tPA) has had a major impact on morbidity and mortality in patients with acute ischemic strokes. The efficacy of tPA as thrombolytic therapy in this patient population is well documented.4
Also well documented is the significant impact of time-to-tPA treatment on outcomes. If therapy is started within 3 to 4.5 hours of ischemic stroke onset, patients have improved functional outcomes 3 to 6 months after the incident (Figure 1). Between 31% and 50% of patients treated with tPA within 3 hours experienced improved recovery at 3 months compared with 20% to 38% of patients treated with placebo.5–9 Faster onset to treatment, measured in 15-minute increments, has been shown to significantly reduce in-hospital mortality, reduce intracranial hemorrhage, increase ability to walk at discharge, and increase number discharged to home.6 Even a 1-minute delay in time-to-tPA treatment has a substantial impact on rates of morbidity and mortality (Table 1).10 National and international guidelines recommend starting intravenous tPA within 1 hour of patient arrival in the emergency department and not longer than 4.5 hours since symptom onset, although some evidence indicates a 3-hour window.5,11,12
Although the evidence supports the benefit of rapid therapy for acute ischemic stroke, the national percentage of patients who actually receive tPA within the therapeutic window is small, by some estimates as low as 3% to 5%.13 For optimal stroke care, the rate should be 30% to 50%.
IMPROVING TREATMENT TIMES
Studies have found that the major reason patients do not receive tPA is that they do not reach the hospital quickly enough to be assessed and treated within the treatment window.14,15 In essence, neurologists have the technology to treat most patients, but are waiting for the patients to arrive. Many factors contribute to this delayed arrival time. On the patient level, the primary factors are related to failure to recognize stroke symptoms as well as failure to understand their seriousness.
From the healthcare provider’s perspective, a major barrier to reducing the time-to-treatment window is the need to accurately assess patients with acute ischemic stroke who are eligible for thrombolytic therapy. This is difficult to achieve in clinical practice because it requires neurologic imaging primarily with computed tomography (CT) or magnetic resonance imaging (MRI) and laboratory analyses so that hemorrhagic stroke and other contraindications to thrombolysis can be excluded. Traditionally, this type of analytic equipment had been available only in emergency departments, requiring patients to be brought to those facilities.
Recent innovation in this area led to the development of specialized ambulances equipped with a CT scanner, point-of-care laboratory equipment, and telemedicine connection along with the appropriate treatment options/medications and trained healthcare personnel to provide prehospital stroke treatment. These specially equipped ambulances are known as MSTUs or stroke emergency mobile (STEMO) units. Their development has dramatically altered the strategy from one of taking the patient to the treatment to taking the treatment to the patient.
MOBILE STROKE TREATMENT UNITS
Two technological innovations have been fundamental to the creation and success of MSTU: portable CT scanners and high-speed wireless data transmission.
CT scanners. A key element was the development of a portable diagnostic-quality head and neck CT scanner that can be fit inside a typical-sized ambulance. This 8-slice CT scanner is capable of creating the same scan types and quality found in radiology department CT scanners, including axial imaging, helical angiography, and perfusion imaging. The resolution and slice thickness (1.25 mm) of the images are of suitable quality to enable neurologists and neuroradiologists to exclude hemorrhage in acute stroke, to assess the degree of brain injury, and to identify the vascular lesion responsible for the ischemic deficit. These technologies also enable diagnostic differentiation between brain tissue that is irreversibly infarcted from that which is potentially salvageable, thereby allowing more accurate patient assessment. The imaging data currently obtainable by CT scanners fitted on ambulances is only likely to improve with future technological advances.
Wireless data transmission. Cellular wireless providers have developed the technology and equipment to provide high-speed wireless broadband capable of transmitting high-quality CT and MRI images. It also enables encrypted feed of video telemedicine, data transmission, and download of patient data. This allows the MSTU to electronically sit inside the firewalls of healthcare facilities, providing access to the patients’ electronic health records and to on-site stroke experts.
The successes have been impressive. Studies have found that the deployment of an MSTU significantly reduces the median time from 9-1-1 alarm to intravenous thrombolysis without increasing adverse events.16–19 These data are primarily from the PHANTOM-S study, a pilot program conducted in Germany.18,19 Results showed a significant reduction in alarm-to-treatment times, from 76 minutes in the hospital control group to 52 minutes in the MSTU group (Table 2).17,19 Further, among patients who suffered an ischemic stroke, the proportion who received tPA within 1 hour of symptom onset was sixfold higher after MSTU deployment (Table 3).18 In a separate European study, prehospital stroke assessment using an MSTU significantly reduced the median time from alarm-to-therapy decision: 35 minutes versus 76 in the hospital group.16
The prehospital cerebrovascular diagnostic workup provided by an MSTU also can improve the emergency management of other stroke types. By providing more diagnostic data and higher quality imaging, the units improve the accuracy of the diagnosis. In turn, this enables emergency personnel to provide accurate therapy and to transfer patients to hospitals with the appropriate level of stroke care, decreasing the need for additional intrahospital transfers.20
Overall, it has been shown that an MSTU equipped with the necessary imaging and laboratory testing equipment can provide appropriate, accurate, and safe ambulance-based prehospital tPA administration, reduce the time to tPA administration, and increase the number of patients who receive tPA administration. All of these factors combine to improve outcomes in patients with acute ischemic stroke.
CLEVELAND CLINIC EXPERIENCE
Cleveland Clinic has a tradition of providing high-quality and innovative stroke care. Recognizing the importance of an appropriately equipped MSTU in reducing the time to stroke treatment, especially tPA administration, Cleveland Clinic instituted a plan to develop an MSTU for the care of patients in the Cleveland area. The development required several planning, funding, and development phases.
Planning. Establishing relations with both city planners and area hospitals was central to planning the MSTU startup. An agreement with the city of Cleveland included creating an emergency medical system (EMS) triage algorithm for the 9-1-1 dispatch center. When a call is received, the dispatcher uses a stroke checklist to perform an initial screening. If a stroke is suspected, the MSTU is dispatched along with a Cleveland EMS or other first-responder unit.
As part of the agreement, Cleveland officials required that the MSTU treat all patients, regardless of their ability to pay. This requirement has been beneficial to the MSTU mission as it allows for treating more patients with tPA as quickly as possible without concern for health insurance, which maximizes the potential for neurologic recovery.
Staffing and procedures. The MSTU staff is composed of a paramedic, a critical care nurse, a CT technologist, and an emergency medicine technician/EMS driver. They perform CT scans and point-of-care laboratory tests on patients who have stroke symptoms. The CT scans and laboratory results are wirelessly transmitted to Cleveland Clinic. A neurologist assesses the data, consults with the MSTU staff on history and neurologic examination, and diagnoses the patient remotely. Patients are then transported to the closest hospital with the resources to meet their clinical needs. If thrombolytic therapy is indicated, intravenous tPA is initiated immediately at the scene. If the patient has sustained a hemorrhagic stroke, reversal of anticoagulation therapy is initiated, if indicated.
Outcomes. The success rates also have been impressive, with dramatic reductions in time to treatment. On average, patients received tPA 40 minutes faster in the MSTU model than in the standard model of ambulance transport and in-hospital evaluation and treatment: 64 minutes versus 104 minutes. Further, more patients in the MSTU group received tPA: 26% versus 14%. Results also showed a 21-minute reduction in time-to-CT completion, an important aspect of providing more timely care.21–23 This CT scanner is also capable of CT angiography. This enables large-vessel occlusion strokes to be identified in the field. When these types of strokes are identified in the field, the patients are transported directly to a stroke center capable of endovascular therapy, even bypassing some primary stroke centers.
Using the MSTU to bring diagnostic and stroke care to the patient has shown that the time between the onset of stroke-like symptoms and the delivery of treatment can be reduced. Thus, an MSTU has the potential to minimize the mortality and long-term morbidity associated with strokes.
- Centers for Disease Control and Prevention (CDC). Prevalence of stroke— United States, 2006–2010. MMWR Morb Mortal Wkly Rep 2012; 61:379–382.
- Mozaffarian D, Benjamin EJ, Go AS, et al. Heart disease and stroke statistics—2015 update: a report from the American Heart Association. Circulation 2015; 131:e29–e322.
- Howard G, Banach M, Cushman M, et al. Is blood pressure control for stroke prevention the correct goal? The lost opportunity of preventing hypertension. Stroke 2015; 46:1595–1600.
- Tissue plasminogen activator for acute ischemic stroke. The National Institute of Neurological Disorders and Stroke rt-PA Stroke Study Group. N Engl J Med 1995; 333:1581–1587.
- Lees KR, Bluhmki E, von Kummer R, et al; ECASS, ATLANTIS, NINDS, and EPITHET rt-PA Study Group. Time to treatment with intravenous alteplase and outcome in stroke: an updated pooled analysis of ECASS, ATLANTIS, NINDS, and EPITHET trials. Lancet 2010; 375:1695–1703.
- Saver JL, Fonarow GC, Smith EE, et al. Time to treatment with intravenous tissue plasminogen activator and outcome from acute ischemic stroke. JAMA 2013; 309:2480–2488.
- Hacke W, Donnan G, Fieschi C, et al; ATLANTIS Trials Investigators; ECASS Trials Investigators; NINDS rt-PA Study Group Investigators. Association of outcome with early stroke treatment: pooled analysis of ATLANTIS, ECASS, and NINDS rt-PA stroke trials. Lancet 2004; 363:768–774.
- Wardlaw JM, Murray V, Berge E, et al. Recombinant tissue plasminogen activator for acute ischaemic stroke: an updated systematic review and meta-analysis. Lancet 2012; 379:2364–2372.
- Prabhakaran S, Ruff I, Bernstein RA. Acute stroke intervention: a systematic review. JAMA 2015; 313:1451–1462.
- Saver JL. Time is brain—quantified. Stroke 2006; 37:263–266.
- Jauch EC, Saver JL, Adams HP Jr, et al; American Heart Association Stroke Council; Council on Cardiovascular Nursing; Council on Peripheral Vascular Disease; Council on Clinical Cardiology. Guidelines for the early management of patients with acute ischemic stroke: a guideline for healthcare professionals from the American Heart Association/American Stroke Association. Stroke 2013; 44:870–947.
- Hacke W, Kaste M, Bluhmki E, et al; ECASS Investigators. Thrombolysis with alteplase 3 to 4.5 hours after acute ischemic stroke. N Engl J Med 2008; 359:1317– 1329.
- Adeoye O, Hornung R, Khatri P, Kleindorfer D. Recombinant tissue-type plasminogen activator use for ischemic stroke in the United States: a doubling of treatment rates over the course of 5 years. Stroke 2011; 42:1952–1955.
- Barber PA, Zhang J, Demchuk AM, Hill MD, Buchan AM. Why are stroke patients excluded from TPA therapy? An analysis of patient eligibility. Neurology 2001; 56:1015–1020.
- Lichtman JH, Watanabe E, Allen NB, Jones SB, Dostal J, Goldstein LB. Hospital arrival time and intravenous t-PA use in US Academic Medical Centers, 2001– 2004. Stroke 2009; 40:3845–3850.
- Walter S, Kostopoulos P, Haass A, et al. Diagnosis and treatment of patients with stroke in a mobile stroke unit versus in hospital: a randomised controlled trial. Lancet Neurol 2012; 11:397–404.
- Ebinger M, Winter B, Wendt M, et al; STEMO Consortium. Effect of the use of ambulance-based thrombolysis on time to thrombolysis in acute ischemic stroke: a randomized clinical trial. JAMA 2014; 311:1622–1631.
- Ebinger M, Kunz A, Wendt M, et al. Effects of golden hour thrombolysis: a prehospital acute neurological treatment and optimization of medical care in stroke (PHANTOM-S) substudy. JAMA Neurol 2015; 72:25–30.
- Weber JE, Ebinger M, Rozansk M, et al; STEMO-Consortium. Prehospital thrombolysis in acute stroke: results of the PHANTOM-S pilot study. Neurology 2013; 80:163–168.
- Wendt M, Ebinger M, Kunz A, et al; STEMO Consortium. Improved prehospital triage of patients with stroke in a specialized stroke ambulance: results of the prehospital acute neurological therapy and optimization of medical care in stroke study. Stroke 2015; 46: 740–745.
- Cho S, Cerejo R, Taqui A, et al. Feasibility of telemedicine on a mobile stroke treatment unit. Stroke 2015; 46:ATP200.
- Sheikhi L, Itrat A, Cerejo R, et al. Does portable CT imaging in a mobile stroke treatment unit (MSTU) provide adequate quality for early critical decision making? Stroke 2015; 46:AWP31.
- Taqui A, Cerejo R, Itrat A, et al. Reduction in time to imaging and intravenous thrombolysis by in-field evaluation and treatment in a mobile stroke treatment unit. Stroke 2015; 46:A54.
- Centers for Disease Control and Prevention (CDC). Prevalence of stroke— United States, 2006–2010. MMWR Morb Mortal Wkly Rep 2012; 61:379–382.
- Mozaffarian D, Benjamin EJ, Go AS, et al. Heart disease and stroke statistics—2015 update: a report from the American Heart Association. Circulation 2015; 131:e29–e322.
- Howard G, Banach M, Cushman M, et al. Is blood pressure control for stroke prevention the correct goal? The lost opportunity of preventing hypertension. Stroke 2015; 46:1595–1600.
- Tissue plasminogen activator for acute ischemic stroke. The National Institute of Neurological Disorders and Stroke rt-PA Stroke Study Group. N Engl J Med 1995; 333:1581–1587.
- Lees KR, Bluhmki E, von Kummer R, et al; ECASS, ATLANTIS, NINDS, and EPITHET rt-PA Study Group. Time to treatment with intravenous alteplase and outcome in stroke: an updated pooled analysis of ECASS, ATLANTIS, NINDS, and EPITHET trials. Lancet 2010; 375:1695–1703.
- Saver JL, Fonarow GC, Smith EE, et al. Time to treatment with intravenous tissue plasminogen activator and outcome from acute ischemic stroke. JAMA 2013; 309:2480–2488.
- Hacke W, Donnan G, Fieschi C, et al; ATLANTIS Trials Investigators; ECASS Trials Investigators; NINDS rt-PA Study Group Investigators. Association of outcome with early stroke treatment: pooled analysis of ATLANTIS, ECASS, and NINDS rt-PA stroke trials. Lancet 2004; 363:768–774.
- Wardlaw JM, Murray V, Berge E, et al. Recombinant tissue plasminogen activator for acute ischaemic stroke: an updated systematic review and meta-analysis. Lancet 2012; 379:2364–2372.
- Prabhakaran S, Ruff I, Bernstein RA. Acute stroke intervention: a systematic review. JAMA 2015; 313:1451–1462.
- Saver JL. Time is brain—quantified. Stroke 2006; 37:263–266.
- Jauch EC, Saver JL, Adams HP Jr, et al; American Heart Association Stroke Council; Council on Cardiovascular Nursing; Council on Peripheral Vascular Disease; Council on Clinical Cardiology. Guidelines for the early management of patients with acute ischemic stroke: a guideline for healthcare professionals from the American Heart Association/American Stroke Association. Stroke 2013; 44:870–947.
- Hacke W, Kaste M, Bluhmki E, et al; ECASS Investigators. Thrombolysis with alteplase 3 to 4.5 hours after acute ischemic stroke. N Engl J Med 2008; 359:1317– 1329.
- Adeoye O, Hornung R, Khatri P, Kleindorfer D. Recombinant tissue-type plasminogen activator use for ischemic stroke in the United States: a doubling of treatment rates over the course of 5 years. Stroke 2011; 42:1952–1955.
- Barber PA, Zhang J, Demchuk AM, Hill MD, Buchan AM. Why are stroke patients excluded from TPA therapy? An analysis of patient eligibility. Neurology 2001; 56:1015–1020.
- Lichtman JH, Watanabe E, Allen NB, Jones SB, Dostal J, Goldstein LB. Hospital arrival time and intravenous t-PA use in US Academic Medical Centers, 2001– 2004. Stroke 2009; 40:3845–3850.
- Walter S, Kostopoulos P, Haass A, et al. Diagnosis and treatment of patients with stroke in a mobile stroke unit versus in hospital: a randomised controlled trial. Lancet Neurol 2012; 11:397–404.
- Ebinger M, Winter B, Wendt M, et al; STEMO Consortium. Effect of the use of ambulance-based thrombolysis on time to thrombolysis in acute ischemic stroke: a randomized clinical trial. JAMA 2014; 311:1622–1631.
- Ebinger M, Kunz A, Wendt M, et al. Effects of golden hour thrombolysis: a prehospital acute neurological treatment and optimization of medical care in stroke (PHANTOM-S) substudy. JAMA Neurol 2015; 72:25–30.
- Weber JE, Ebinger M, Rozansk M, et al; STEMO-Consortium. Prehospital thrombolysis in acute stroke: results of the PHANTOM-S pilot study. Neurology 2013; 80:163–168.
- Wendt M, Ebinger M, Kunz A, et al; STEMO Consortium. Improved prehospital triage of patients with stroke in a specialized stroke ambulance: results of the prehospital acute neurological therapy and optimization of medical care in stroke study. Stroke 2015; 46: 740–745.
- Cho S, Cerejo R, Taqui A, et al. Feasibility of telemedicine on a mobile stroke treatment unit. Stroke 2015; 46:ATP200.
- Sheikhi L, Itrat A, Cerejo R, et al. Does portable CT imaging in a mobile stroke treatment unit (MSTU) provide adequate quality for early critical decision making? Stroke 2015; 46:AWP31.
- Taqui A, Cerejo R, Itrat A, et al. Reduction in time to imaging and intravenous thrombolysis by in-field evaluation and treatment in a mobile stroke treatment unit. Stroke 2015; 46:A54.
KEY POINTS
- Therapeutic use of tissue plasminogen activators (tPA) has had a major impact on morbidity and mortality rates in patients with acute ischemic strokes.
- Even a 1-minute delay in time-to-tPA treatment affects morbidity and mortality rates.
- The major reason patients do not receive tPA is that they do not reach the hospital quickly enough to be assessed and treated within the treatment window.
- Portable computed tomography and high-speed wireless data transmission are fundamental to the success of mobile stroke treatment units.