User login
Bringing you the latest news, research and reviews, exclusive interviews, podcasts, quizzes, and more.
div[contains(@class, 'read-next-article')]
div[contains(@class, 'nav-primary')]
nav[contains(@class, 'nav-primary')]
section[contains(@class, 'footer-nav-section-wrapper')]
nav[contains(@class, 'nav-ce-stack nav-ce-stack__large-screen')]
header[@id='header']
div[contains(@class, 'header__large-screen')]
div[contains(@class, 'read-next-article')]
div[contains(@class, 'main-prefix')]
div[contains(@class, 'nav-primary')]
nav[contains(@class, 'nav-primary')]
section[contains(@class, 'footer-nav-section-wrapper')]
footer[@id='footer']
section[contains(@class, 'nav-hidden')]
div[contains(@class, 'ce-card-content')]
nav[contains(@class, 'nav-ce-stack')]
div[contains(@class, 'view-medstat-quiz-listing-panes')]
div[contains(@class, 'pane-article-sidebar-latest-news')]
Low HIV levels linked to ‘almost zero’ risk of sexual transmission
BRISBANE, AUSTRALIA – from the World Health Organization.
The announcement was made concurrently with the publication of definitive new research in The Lancet. The findings were presented virtually at the International AIDS Society conference on HIV Science.
The WHO estimates that 76% of the 39 million people worldwide living with HIV take antiretroviral therapy (ART).
“Antiretroviral therapy continues to transform the lives of people living with HIV,” a WHO news release stated. “People living with HIV who are diagnosed and treated early, and take their medication as prescribed, can expect to have the same health and life expectancy as their HIV-negative counterparts.”
The Lancet study showed that people who have a viral load of less than 1,000 copies per milliliter of blood have a tiny chance of transmitting the virus to sexual partners. Of 320 cases of transmission reviewed during the study, only 2 transmissions involved a partner with a load below that threshold. Those cases may have been affected by viral loads rising between the time of testing and transmission. The previous guideline for zero risk of transmission was 200 copies per milliliter.
People living with HIV who do not take ART can have viral loads ranging from 30,000 to more than 500,000 copies per milliliter, according a summary of the study distributed by The Lancet to the media.
The new findings do not apply to the transmission of HIV from mother to child, including during pregnancy, childbirth, and breastfeeding.
“The ultimate goal of antiretroviral therapy for people living with HIV is to maintain undetectable viral loads, which will improve their own health and prevent transmission to their sexual partners and children,” said researcher Lara Vojnov, PhD, diagnostics advisor to the WHO Department of Global HIV, Hepatitis and STI Programmes, in a statement. “But these new findings are also significant as they indicate that the risk of sexual transmission of HIV at low viral loads is almost zero. This provides a powerful opportunity to help destigmatize HIV, promote the benefits of adhering to antiretroviral therapy, and support people living with HIV.”
A version of this article first appeared on WebMD.com.
BRISBANE, AUSTRALIA – from the World Health Organization.
The announcement was made concurrently with the publication of definitive new research in The Lancet. The findings were presented virtually at the International AIDS Society conference on HIV Science.
The WHO estimates that 76% of the 39 million people worldwide living with HIV take antiretroviral therapy (ART).
“Antiretroviral therapy continues to transform the lives of people living with HIV,” a WHO news release stated. “People living with HIV who are diagnosed and treated early, and take their medication as prescribed, can expect to have the same health and life expectancy as their HIV-negative counterparts.”
The Lancet study showed that people who have a viral load of less than 1,000 copies per milliliter of blood have a tiny chance of transmitting the virus to sexual partners. Of 320 cases of transmission reviewed during the study, only 2 transmissions involved a partner with a load below that threshold. Those cases may have been affected by viral loads rising between the time of testing and transmission. The previous guideline for zero risk of transmission was 200 copies per milliliter.
People living with HIV who do not take ART can have viral loads ranging from 30,000 to more than 500,000 copies per milliliter, according a summary of the study distributed by The Lancet to the media.
The new findings do not apply to the transmission of HIV from mother to child, including during pregnancy, childbirth, and breastfeeding.
“The ultimate goal of antiretroviral therapy for people living with HIV is to maintain undetectable viral loads, which will improve their own health and prevent transmission to their sexual partners and children,” said researcher Lara Vojnov, PhD, diagnostics advisor to the WHO Department of Global HIV, Hepatitis and STI Programmes, in a statement. “But these new findings are also significant as they indicate that the risk of sexual transmission of HIV at low viral loads is almost zero. This provides a powerful opportunity to help destigmatize HIV, promote the benefits of adhering to antiretroviral therapy, and support people living with HIV.”
A version of this article first appeared on WebMD.com.
BRISBANE, AUSTRALIA – from the World Health Organization.
The announcement was made concurrently with the publication of definitive new research in The Lancet. The findings were presented virtually at the International AIDS Society conference on HIV Science.
The WHO estimates that 76% of the 39 million people worldwide living with HIV take antiretroviral therapy (ART).
“Antiretroviral therapy continues to transform the lives of people living with HIV,” a WHO news release stated. “People living with HIV who are diagnosed and treated early, and take their medication as prescribed, can expect to have the same health and life expectancy as their HIV-negative counterparts.”
The Lancet study showed that people who have a viral load of less than 1,000 copies per milliliter of blood have a tiny chance of transmitting the virus to sexual partners. Of 320 cases of transmission reviewed during the study, only 2 transmissions involved a partner with a load below that threshold. Those cases may have been affected by viral loads rising between the time of testing and transmission. The previous guideline for zero risk of transmission was 200 copies per milliliter.
People living with HIV who do not take ART can have viral loads ranging from 30,000 to more than 500,000 copies per milliliter, according a summary of the study distributed by The Lancet to the media.
The new findings do not apply to the transmission of HIV from mother to child, including during pregnancy, childbirth, and breastfeeding.
“The ultimate goal of antiretroviral therapy for people living with HIV is to maintain undetectable viral loads, which will improve their own health and prevent transmission to their sexual partners and children,” said researcher Lara Vojnov, PhD, diagnostics advisor to the WHO Department of Global HIV, Hepatitis and STI Programmes, in a statement. “But these new findings are also significant as they indicate that the risk of sexual transmission of HIV at low viral loads is almost zero. This provides a powerful opportunity to help destigmatize HIV, promote the benefits of adhering to antiretroviral therapy, and support people living with HIV.”
A version of this article first appeared on WebMD.com.
AT IAS 2023
SGLT2 inhibitors linked with fewer gout flares in diabetes
TOPLINE:
compared with matched patients treated with a dipeptidyl peptidase–4 (DPP-4) inhibitor.
METHODOLOGY:
- The study used observational data collected from the entire population of British Columbia that included 15,067 adults with both gout and type 2 diabetes in 2014-2020.
- The group included 8,318 patients who initiated an SGLT2 inhibitor and 6,749 patients who initiated a DPP-4 inhibitor during the study period after at least 1 year of continuous enrollment.
- Using propensity-score matching, 4,075 matched pairs were identified, where one person initiated an SGLT2 inhibitor and the other started a DPP-4 inhibitor.
- Primary outcome was recurrent gout flare counts during follow-up that required an ED visit, hospital admission, or an outpatient visit for a gout flare coupled with appropriate treatment, tallied from the first day of drug receipt until June 30, 2022, with an average follow-up of 1.6 years.
- Secondary endpoints included the incidence of myocardial infarction and stroke.
TAKEAWAY:
- Total gout-flare rates after SGLT2 inhibitor initiation were 52.4/1000 person-years and after DPP-4 inhibitor initiation were 79.7/1,000 person-years, an adjusted rate ratio of 0.66, a reduction significantly linked with SGLT2 inhibitor use.
- For flares that required an ED visit or hospitalization, initiation of an SGLT2 inhibitor was linked with a significant, reduced aRR of 0.52, compared with DPP-4 inhibitor initiation.
- The flare-rate reduction linked with SGLT2 inhibitor use was consistent regardless of sex, age, baseline diuretic use, prior treatment with a urate-lowering agent, and baseline gout intensity.
- SGLT2 inhibitor initiation was also significantly linked with an adjusted reduced hazard ratio of 0.69 in the incidence of myocardial infarction, compared with DPP-4 inhibitor initiation, but stroke incidence was not significantly different between the groups.
IN PRACTICE:
These findings suggest that SGLT2 inhibitors could have a much-needed ability to simultaneously reduce the burden of recurrent gout flares and coronary sequelae in patients with gout and type 2 diabetes, indicating that “SGLT2 inhibitors may offer distinct benefits,” making the drug class “a particularly attractive addition to current urate-lowering therapies,” the researchers write.
SOURCE:
The study was primarily conducted by researchers at Massachusetts General Hospital in Boston. The study was published online July 24 in Annals of Internal Medicine.
LIMITATIONS:
The data used in the study did not include gout flares that did not require medical attention and did not include laboratory findings for study participants. Because the data were observational the findings may be susceptible to unmeasured confounding.
DISCLOSURES:
The study received no commercial funding. One author has reported receiving consulting fees from ANI and LG Chem.
A version of this article first appeared on Medscape.com.
TOPLINE:
compared with matched patients treated with a dipeptidyl peptidase–4 (DPP-4) inhibitor.
METHODOLOGY:
- The study used observational data collected from the entire population of British Columbia that included 15,067 adults with both gout and type 2 diabetes in 2014-2020.
- The group included 8,318 patients who initiated an SGLT2 inhibitor and 6,749 patients who initiated a DPP-4 inhibitor during the study period after at least 1 year of continuous enrollment.
- Using propensity-score matching, 4,075 matched pairs were identified, where one person initiated an SGLT2 inhibitor and the other started a DPP-4 inhibitor.
- Primary outcome was recurrent gout flare counts during follow-up that required an ED visit, hospital admission, or an outpatient visit for a gout flare coupled with appropriate treatment, tallied from the first day of drug receipt until June 30, 2022, with an average follow-up of 1.6 years.
- Secondary endpoints included the incidence of myocardial infarction and stroke.
TAKEAWAY:
- Total gout-flare rates after SGLT2 inhibitor initiation were 52.4/1000 person-years and after DPP-4 inhibitor initiation were 79.7/1,000 person-years, an adjusted rate ratio of 0.66, a reduction significantly linked with SGLT2 inhibitor use.
- For flares that required an ED visit or hospitalization, initiation of an SGLT2 inhibitor was linked with a significant, reduced aRR of 0.52, compared with DPP-4 inhibitor initiation.
- The flare-rate reduction linked with SGLT2 inhibitor use was consistent regardless of sex, age, baseline diuretic use, prior treatment with a urate-lowering agent, and baseline gout intensity.
- SGLT2 inhibitor initiation was also significantly linked with an adjusted reduced hazard ratio of 0.69 in the incidence of myocardial infarction, compared with DPP-4 inhibitor initiation, but stroke incidence was not significantly different between the groups.
IN PRACTICE:
These findings suggest that SGLT2 inhibitors could have a much-needed ability to simultaneously reduce the burden of recurrent gout flares and coronary sequelae in patients with gout and type 2 diabetes, indicating that “SGLT2 inhibitors may offer distinct benefits,” making the drug class “a particularly attractive addition to current urate-lowering therapies,” the researchers write.
SOURCE:
The study was primarily conducted by researchers at Massachusetts General Hospital in Boston. The study was published online July 24 in Annals of Internal Medicine.
LIMITATIONS:
The data used in the study did not include gout flares that did not require medical attention and did not include laboratory findings for study participants. Because the data were observational the findings may be susceptible to unmeasured confounding.
DISCLOSURES:
The study received no commercial funding. One author has reported receiving consulting fees from ANI and LG Chem.
A version of this article first appeared on Medscape.com.
TOPLINE:
compared with matched patients treated with a dipeptidyl peptidase–4 (DPP-4) inhibitor.
METHODOLOGY:
- The study used observational data collected from the entire population of British Columbia that included 15,067 adults with both gout and type 2 diabetes in 2014-2020.
- The group included 8,318 patients who initiated an SGLT2 inhibitor and 6,749 patients who initiated a DPP-4 inhibitor during the study period after at least 1 year of continuous enrollment.
- Using propensity-score matching, 4,075 matched pairs were identified, where one person initiated an SGLT2 inhibitor and the other started a DPP-4 inhibitor.
- Primary outcome was recurrent gout flare counts during follow-up that required an ED visit, hospital admission, or an outpatient visit for a gout flare coupled with appropriate treatment, tallied from the first day of drug receipt until June 30, 2022, with an average follow-up of 1.6 years.
- Secondary endpoints included the incidence of myocardial infarction and stroke.
TAKEAWAY:
- Total gout-flare rates after SGLT2 inhibitor initiation were 52.4/1000 person-years and after DPP-4 inhibitor initiation were 79.7/1,000 person-years, an adjusted rate ratio of 0.66, a reduction significantly linked with SGLT2 inhibitor use.
- For flares that required an ED visit or hospitalization, initiation of an SGLT2 inhibitor was linked with a significant, reduced aRR of 0.52, compared with DPP-4 inhibitor initiation.
- The flare-rate reduction linked with SGLT2 inhibitor use was consistent regardless of sex, age, baseline diuretic use, prior treatment with a urate-lowering agent, and baseline gout intensity.
- SGLT2 inhibitor initiation was also significantly linked with an adjusted reduced hazard ratio of 0.69 in the incidence of myocardial infarction, compared with DPP-4 inhibitor initiation, but stroke incidence was not significantly different between the groups.
IN PRACTICE:
These findings suggest that SGLT2 inhibitors could have a much-needed ability to simultaneously reduce the burden of recurrent gout flares and coronary sequelae in patients with gout and type 2 diabetes, indicating that “SGLT2 inhibitors may offer distinct benefits,” making the drug class “a particularly attractive addition to current urate-lowering therapies,” the researchers write.
SOURCE:
The study was primarily conducted by researchers at Massachusetts General Hospital in Boston. The study was published online July 24 in Annals of Internal Medicine.
LIMITATIONS:
The data used in the study did not include gout flares that did not require medical attention and did not include laboratory findings for study participants. Because the data were observational the findings may be susceptible to unmeasured confounding.
DISCLOSURES:
The study received no commercial funding. One author has reported receiving consulting fees from ANI and LG Chem.
A version of this article first appeared on Medscape.com.
FROM ANNALS OF INTERNAL MEDICINE
Study examines burden of vitiligo in the U.S.
To investigate the incidence and prevalence of diagnosed vitiligo in the United States, researchers used a 15% random sample of electronic medical records from the IBM Explorys database. Two cohorts were included: 2,980,778 patients diagnosed with vitiligo between Jan. 1, 2015, and Dec. 31, 2019 (incidence analysis), and 1,057,534 patients diagnosed with vitiligo between Jan. 1 and Dec. 31, 2019 (prevalence analysis).The main outcomes were incidence (per 100,000 person-years) and prevalence of diagnosed vitiligo overall and by age, race/ethnicity, and sex. Amit Garg, MD, a dermatologist with Northwell Health, New Hyde Park, N.Y., led the study, which was published in JAMA Dermatology.
The age- and sex-adjusted overall incidence rate of diagnosed vitiligo was 22.6 per 100,000 person-years, and the prevalence was 0.16%, the authors reported. The sex-adjusted IR was highest among patients aged 60-69 years (25.3 per 100,000 person-years); prevalence was highest among patients aged 70 years or older (0.21%).
The highest age-adjusted IR was among Asian American patients (41.2 per 100,000 person-years), followed by Hispanic/Latino patients (37.3 per 100,000 PY), those reporting other or multiple races (31.1 per 100,000), Black patients (29.6 per 100,000 person-years), and White patients (18.7 per 100,000 person-years). The highest age-adjusted prevalence was among Hispanic/Latino patients (0.29%), followed by Asian American patients (0.27%), those reporting other or multiple races (0.24%), Black patients (0.22%), and White patients (0.13%).
The burden of vitiligo in the United States is poorly understood, and the findings “may support improving awareness of vitiligo disease burden in medical and public sectors, informing research agendas, improving enrollment of racial and ethnic minority populations in trials, and developing health policies,” the authors wrote.
Limitations of the study included that the analysis only captured patients who sought care in health systems included in the database, and there was the potential for underreporting, “since not all patients with vitiligo seek care,” the authors noted.
Dr. Garg reported being an adviser for and receiving honoraria from many pharmaceutical companies. He has also received research grants from AbbVie, UCB, the National Psoriasis Foundation, and the CHORD COUSIN Collaboration. No other disclosures were reported.
A version of this article first appeared on Medscape.com .
To investigate the incidence and prevalence of diagnosed vitiligo in the United States, researchers used a 15% random sample of electronic medical records from the IBM Explorys database. Two cohorts were included: 2,980,778 patients diagnosed with vitiligo between Jan. 1, 2015, and Dec. 31, 2019 (incidence analysis), and 1,057,534 patients diagnosed with vitiligo between Jan. 1 and Dec. 31, 2019 (prevalence analysis).The main outcomes were incidence (per 100,000 person-years) and prevalence of diagnosed vitiligo overall and by age, race/ethnicity, and sex. Amit Garg, MD, a dermatologist with Northwell Health, New Hyde Park, N.Y., led the study, which was published in JAMA Dermatology.
The age- and sex-adjusted overall incidence rate of diagnosed vitiligo was 22.6 per 100,000 person-years, and the prevalence was 0.16%, the authors reported. The sex-adjusted IR was highest among patients aged 60-69 years (25.3 per 100,000 person-years); prevalence was highest among patients aged 70 years or older (0.21%).
The highest age-adjusted IR was among Asian American patients (41.2 per 100,000 person-years), followed by Hispanic/Latino patients (37.3 per 100,000 PY), those reporting other or multiple races (31.1 per 100,000), Black patients (29.6 per 100,000 person-years), and White patients (18.7 per 100,000 person-years). The highest age-adjusted prevalence was among Hispanic/Latino patients (0.29%), followed by Asian American patients (0.27%), those reporting other or multiple races (0.24%), Black patients (0.22%), and White patients (0.13%).
The burden of vitiligo in the United States is poorly understood, and the findings “may support improving awareness of vitiligo disease burden in medical and public sectors, informing research agendas, improving enrollment of racial and ethnic minority populations in trials, and developing health policies,” the authors wrote.
Limitations of the study included that the analysis only captured patients who sought care in health systems included in the database, and there was the potential for underreporting, “since not all patients with vitiligo seek care,” the authors noted.
Dr. Garg reported being an adviser for and receiving honoraria from many pharmaceutical companies. He has also received research grants from AbbVie, UCB, the National Psoriasis Foundation, and the CHORD COUSIN Collaboration. No other disclosures were reported.
A version of this article first appeared on Medscape.com .
To investigate the incidence and prevalence of diagnosed vitiligo in the United States, researchers used a 15% random sample of electronic medical records from the IBM Explorys database. Two cohorts were included: 2,980,778 patients diagnosed with vitiligo between Jan. 1, 2015, and Dec. 31, 2019 (incidence analysis), and 1,057,534 patients diagnosed with vitiligo between Jan. 1 and Dec. 31, 2019 (prevalence analysis).The main outcomes were incidence (per 100,000 person-years) and prevalence of diagnosed vitiligo overall and by age, race/ethnicity, and sex. Amit Garg, MD, a dermatologist with Northwell Health, New Hyde Park, N.Y., led the study, which was published in JAMA Dermatology.
The age- and sex-adjusted overall incidence rate of diagnosed vitiligo was 22.6 per 100,000 person-years, and the prevalence was 0.16%, the authors reported. The sex-adjusted IR was highest among patients aged 60-69 years (25.3 per 100,000 person-years); prevalence was highest among patients aged 70 years or older (0.21%).
The highest age-adjusted IR was among Asian American patients (41.2 per 100,000 person-years), followed by Hispanic/Latino patients (37.3 per 100,000 PY), those reporting other or multiple races (31.1 per 100,000), Black patients (29.6 per 100,000 person-years), and White patients (18.7 per 100,000 person-years). The highest age-adjusted prevalence was among Hispanic/Latino patients (0.29%), followed by Asian American patients (0.27%), those reporting other or multiple races (0.24%), Black patients (0.22%), and White patients (0.13%).
The burden of vitiligo in the United States is poorly understood, and the findings “may support improving awareness of vitiligo disease burden in medical and public sectors, informing research agendas, improving enrollment of racial and ethnic minority populations in trials, and developing health policies,” the authors wrote.
Limitations of the study included that the analysis only captured patients who sought care in health systems included in the database, and there was the potential for underreporting, “since not all patients with vitiligo seek care,” the authors noted.
Dr. Garg reported being an adviser for and receiving honoraria from many pharmaceutical companies. He has also received research grants from AbbVie, UCB, the National Psoriasis Foundation, and the CHORD COUSIN Collaboration. No other disclosures were reported.
A version of this article first appeared on Medscape.com .
FROM JAMA DERMATOLOGY
Over-the-counter switches improve access but come with risks
On July 13, the Food and Drug Administration approved the first over-the-counter (OTC) norgestrel birth control pill (Opill). The daily oral contraceptive was approved for prescription use 5 decades ago, providing regulators with a half-century of data to show that the progestin-only drug can be used safely without a prescription.
The drug is the latest in a series of medications that have made the switch from behind the pharmacy counter to retail shelves.
Why switch?
When a drug manufacturer submits a proposal for a switch to OTC, the key question that the FDA considers is patient safety. Some risks can be mitigated by approving OTC drugs at lower doses than what is available as the prescription version.
“There is no drug that doesn’t have risks,” said Almut G. Winterstein, RPh, PhD, a distinguished professor in pharmaceutical outcomes and policy and director of the Center for Drug Evaluation and Safety at the University of Florida, Gainesville. “Risks are mitigated by putting specific constraints around access to those medications.”
Dr. Winterstein, a former chair of the FDA’s Drug Safety and Risk Management Advisory Committee, said that nonprescription drugs are unnecessary in a functional health care system.
Many patients may struggle with accessing health clinicians, so making medications available OTC fills gaps left by not being able to get a prescription, according to Dr. Winterstein.
A 2012 paper funded by the Consumer Healthcare Products Association (CHPA), the organization representing manufacturers and distributors of OTC medications, estimated that one quarter of people who bought OTC drugs would not otherwise seek treatment if these treatments were available only via prescription. The CHPA notes that the number of those who experience allergies who use nonprescription antihistamines and allergy-relief drugs increased by about 10% between 2009 and 2015.
Cholesterol drugs
Approximately 80 million U.S. adults are eligible for cholesterol-lowering medications, particularly statins, but nearly half don’t take them, according to the Centers for Disease Control and Prevention.
Fear of side effects is the most common reason people might avoid taking these drugs. But eliminating the need for a refill may encourage uptake of the statins.
“It’s refill, refill, refill,” said Allen J. Taylor, MD, chairman of cardiology at MedStar Heart and Vascular Institute, in Washington. “We spend a ton of time refilling statins and it’s a headache for patients, too.”
The need to secure regular prescriptions for the drug, “doesn’t put enough trust and faith in pharmacists and doesn’t put enough trust and faith in patients,” Dr. Taylor said.
Moving statins to the front end of a pharmacy might not be the best move given the potential for drug interactions, but a nonprescription behind-the-counter approach could work, according to Dr. Taylor.
“The concerns are modest at most, to where they can be monitored by a pharmacist,” he said. “There’s probably more people that would take a statin if they had that kind of access.”
Many statin manufacturers have attempted to make the prescription-to-OTC switch. In 2005, an FDA advisory panel rejected Merck’s proposal for OTC sales of lovastatin after reviewing a study that found only 55% of OTC purchases would have been medically appropriate.
In 2015, Pfizer pulled its application to make the cholesterol drug atorvastatin available to patients OTC because patients were not using the drug correctly. AstraZeneca is investigating an online platform that would allow patients to self-assess their eligibility for rosuvastatin.
Asthma inhalers
Inhalers are the main rescue therapy for asthma aside from a visit to the ED.
The only inhaler available OTC is epinephrine sold under the brand name Primatene Mist, but this type of medicine device is not recommended as a first-line therapy for acute asthma symptoms, according to the American Medical Association.
“It’s been around for a long time and has stayed over the counter even though newer, safer agents have come onto the market which aren’t available over the counter,” said William B. Feldman, MD, DPhil, MPH, a pulmonologist at Brigham and Women’s Hospital, Boston.
Patients who have a hard time getting to a doctor or patients who lack insurance often face barriers accessing albuterol inhalers and beta agonist–corticosteroid combinations, according to Dr. Feldman. A switch to OTC distribution would widen access.
“What we’re advocating is, if they’re going to have access to Primatene Mist, wouldn’t it be sensible to have access to a safer and more effective therapy?” Dr. Feldman said.
Triptans
Migraines affect an estimated 39 million people in the United States, according to the American Migraine Foundation. Several drugs to treat migraine are available OTC, including nonsteroidal anti-inflammatory drugs, aspirin, and acetaminophen. Triptans, drugs used for the short-term treatment of acute symptoms, are prescription-only in the United States.
But in the United Kingdom, triptans first became available in retail stores in 2006, leading to reduced costs for patients, employers, and the government. One study found that government health expenditures would be reduced by $84 million annually if the OTC switch were made in six European countries.
However, overuse of the drug and potential contraindications have been cited as concerns with OTC access.
For Dr. Winterstein, the decision to switch isn’t just about the freedom to buy a drug; it comes down to weighing potential risks and benefits.
“Drugs are only as good as if they’re used in the context of how they should be used,” Dr. Winterstein said. “It’s not candy.”
A version of this article first appeared on Medscape.com.
On July 13, the Food and Drug Administration approved the first over-the-counter (OTC) norgestrel birth control pill (Opill). The daily oral contraceptive was approved for prescription use 5 decades ago, providing regulators with a half-century of data to show that the progestin-only drug can be used safely without a prescription.
The drug is the latest in a series of medications that have made the switch from behind the pharmacy counter to retail shelves.
Why switch?
When a drug manufacturer submits a proposal for a switch to OTC, the key question that the FDA considers is patient safety. Some risks can be mitigated by approving OTC drugs at lower doses than what is available as the prescription version.
“There is no drug that doesn’t have risks,” said Almut G. Winterstein, RPh, PhD, a distinguished professor in pharmaceutical outcomes and policy and director of the Center for Drug Evaluation and Safety at the University of Florida, Gainesville. “Risks are mitigated by putting specific constraints around access to those medications.”
Dr. Winterstein, a former chair of the FDA’s Drug Safety and Risk Management Advisory Committee, said that nonprescription drugs are unnecessary in a functional health care system.
Many patients may struggle with accessing health clinicians, so making medications available OTC fills gaps left by not being able to get a prescription, according to Dr. Winterstein.
A 2012 paper funded by the Consumer Healthcare Products Association (CHPA), the organization representing manufacturers and distributors of OTC medications, estimated that one quarter of people who bought OTC drugs would not otherwise seek treatment if these treatments were available only via prescription. The CHPA notes that the number of those who experience allergies who use nonprescription antihistamines and allergy-relief drugs increased by about 10% between 2009 and 2015.
Cholesterol drugs
Approximately 80 million U.S. adults are eligible for cholesterol-lowering medications, particularly statins, but nearly half don’t take them, according to the Centers for Disease Control and Prevention.
Fear of side effects is the most common reason people might avoid taking these drugs. But eliminating the need for a refill may encourage uptake of the statins.
“It’s refill, refill, refill,” said Allen J. Taylor, MD, chairman of cardiology at MedStar Heart and Vascular Institute, in Washington. “We spend a ton of time refilling statins and it’s a headache for patients, too.”
The need to secure regular prescriptions for the drug, “doesn’t put enough trust and faith in pharmacists and doesn’t put enough trust and faith in patients,” Dr. Taylor said.
Moving statins to the front end of a pharmacy might not be the best move given the potential for drug interactions, but a nonprescription behind-the-counter approach could work, according to Dr. Taylor.
“The concerns are modest at most, to where they can be monitored by a pharmacist,” he said. “There’s probably more people that would take a statin if they had that kind of access.”
Many statin manufacturers have attempted to make the prescription-to-OTC switch. In 2005, an FDA advisory panel rejected Merck’s proposal for OTC sales of lovastatin after reviewing a study that found only 55% of OTC purchases would have been medically appropriate.
In 2015, Pfizer pulled its application to make the cholesterol drug atorvastatin available to patients OTC because patients were not using the drug correctly. AstraZeneca is investigating an online platform that would allow patients to self-assess their eligibility for rosuvastatin.
Asthma inhalers
Inhalers are the main rescue therapy for asthma aside from a visit to the ED.
The only inhaler available OTC is epinephrine sold under the brand name Primatene Mist, but this type of medicine device is not recommended as a first-line therapy for acute asthma symptoms, according to the American Medical Association.
“It’s been around for a long time and has stayed over the counter even though newer, safer agents have come onto the market which aren’t available over the counter,” said William B. Feldman, MD, DPhil, MPH, a pulmonologist at Brigham and Women’s Hospital, Boston.
Patients who have a hard time getting to a doctor or patients who lack insurance often face barriers accessing albuterol inhalers and beta agonist–corticosteroid combinations, according to Dr. Feldman. A switch to OTC distribution would widen access.
“What we’re advocating is, if they’re going to have access to Primatene Mist, wouldn’t it be sensible to have access to a safer and more effective therapy?” Dr. Feldman said.
Triptans
Migraines affect an estimated 39 million people in the United States, according to the American Migraine Foundation. Several drugs to treat migraine are available OTC, including nonsteroidal anti-inflammatory drugs, aspirin, and acetaminophen. Triptans, drugs used for the short-term treatment of acute symptoms, are prescription-only in the United States.
But in the United Kingdom, triptans first became available in retail stores in 2006, leading to reduced costs for patients, employers, and the government. One study found that government health expenditures would be reduced by $84 million annually if the OTC switch were made in six European countries.
However, overuse of the drug and potential contraindications have been cited as concerns with OTC access.
For Dr. Winterstein, the decision to switch isn’t just about the freedom to buy a drug; it comes down to weighing potential risks and benefits.
“Drugs are only as good as if they’re used in the context of how they should be used,” Dr. Winterstein said. “It’s not candy.”
A version of this article first appeared on Medscape.com.
On July 13, the Food and Drug Administration approved the first over-the-counter (OTC) norgestrel birth control pill (Opill). The daily oral contraceptive was approved for prescription use 5 decades ago, providing regulators with a half-century of data to show that the progestin-only drug can be used safely without a prescription.
The drug is the latest in a series of medications that have made the switch from behind the pharmacy counter to retail shelves.
Why switch?
When a drug manufacturer submits a proposal for a switch to OTC, the key question that the FDA considers is patient safety. Some risks can be mitigated by approving OTC drugs at lower doses than what is available as the prescription version.
“There is no drug that doesn’t have risks,” said Almut G. Winterstein, RPh, PhD, a distinguished professor in pharmaceutical outcomes and policy and director of the Center for Drug Evaluation and Safety at the University of Florida, Gainesville. “Risks are mitigated by putting specific constraints around access to those medications.”
Dr. Winterstein, a former chair of the FDA’s Drug Safety and Risk Management Advisory Committee, said that nonprescription drugs are unnecessary in a functional health care system.
Many patients may struggle with accessing health clinicians, so making medications available OTC fills gaps left by not being able to get a prescription, according to Dr. Winterstein.
A 2012 paper funded by the Consumer Healthcare Products Association (CHPA), the organization representing manufacturers and distributors of OTC medications, estimated that one quarter of people who bought OTC drugs would not otherwise seek treatment if these treatments were available only via prescription. The CHPA notes that the number of those who experience allergies who use nonprescription antihistamines and allergy-relief drugs increased by about 10% between 2009 and 2015.
Cholesterol drugs
Approximately 80 million U.S. adults are eligible for cholesterol-lowering medications, particularly statins, but nearly half don’t take them, according to the Centers for Disease Control and Prevention.
Fear of side effects is the most common reason people might avoid taking these drugs. But eliminating the need for a refill may encourage uptake of the statins.
“It’s refill, refill, refill,” said Allen J. Taylor, MD, chairman of cardiology at MedStar Heart and Vascular Institute, in Washington. “We spend a ton of time refilling statins and it’s a headache for patients, too.”
The need to secure regular prescriptions for the drug, “doesn’t put enough trust and faith in pharmacists and doesn’t put enough trust and faith in patients,” Dr. Taylor said.
Moving statins to the front end of a pharmacy might not be the best move given the potential for drug interactions, but a nonprescription behind-the-counter approach could work, according to Dr. Taylor.
“The concerns are modest at most, to where they can be monitored by a pharmacist,” he said. “There’s probably more people that would take a statin if they had that kind of access.”
Many statin manufacturers have attempted to make the prescription-to-OTC switch. In 2005, an FDA advisory panel rejected Merck’s proposal for OTC sales of lovastatin after reviewing a study that found only 55% of OTC purchases would have been medically appropriate.
In 2015, Pfizer pulled its application to make the cholesterol drug atorvastatin available to patients OTC because patients were not using the drug correctly. AstraZeneca is investigating an online platform that would allow patients to self-assess their eligibility for rosuvastatin.
Asthma inhalers
Inhalers are the main rescue therapy for asthma aside from a visit to the ED.
The only inhaler available OTC is epinephrine sold under the brand name Primatene Mist, but this type of medicine device is not recommended as a first-line therapy for acute asthma symptoms, according to the American Medical Association.
“It’s been around for a long time and has stayed over the counter even though newer, safer agents have come onto the market which aren’t available over the counter,” said William B. Feldman, MD, DPhil, MPH, a pulmonologist at Brigham and Women’s Hospital, Boston.
Patients who have a hard time getting to a doctor or patients who lack insurance often face barriers accessing albuterol inhalers and beta agonist–corticosteroid combinations, according to Dr. Feldman. A switch to OTC distribution would widen access.
“What we’re advocating is, if they’re going to have access to Primatene Mist, wouldn’t it be sensible to have access to a safer and more effective therapy?” Dr. Feldman said.
Triptans
Migraines affect an estimated 39 million people in the United States, according to the American Migraine Foundation. Several drugs to treat migraine are available OTC, including nonsteroidal anti-inflammatory drugs, aspirin, and acetaminophen. Triptans, drugs used for the short-term treatment of acute symptoms, are prescription-only in the United States.
But in the United Kingdom, triptans first became available in retail stores in 2006, leading to reduced costs for patients, employers, and the government. One study found that government health expenditures would be reduced by $84 million annually if the OTC switch were made in six European countries.
However, overuse of the drug and potential contraindications have been cited as concerns with OTC access.
For Dr. Winterstein, the decision to switch isn’t just about the freedom to buy a drug; it comes down to weighing potential risks and benefits.
“Drugs are only as good as if they’re used in the context of how they should be used,” Dr. Winterstein said. “It’s not candy.”
A version of this article first appeared on Medscape.com.
Intermittent fasting vs. calorie counting for weight loss
BOSTON –
For the study, 57 overweight and obese participants with type 2 diabetes were randomly assigned to three different groups: The first group ate between noon and 8 p.m., the second was asked to reduce caloric intake by 25% of maintenance calories, and the third, a control group, continued eating normally.
The calorie-restriction group tracked intake on MyFitnessPal, an app that logs the calorie content of different foods. Both the intermittent-fasting and calorie-restriction groups were assigned a dietitian to help with adherence.
After 6 months, participants in the intermittent-fasting group lost about 4.3% of body weight – the equivalent of 10 pounds of weight loss for a person weighing 230 pounds – whereas participants in the calorie-restriction group lost about 2.5% of body weight.
The difference between the two groups was not significant, so one approach isn’t necessarily better than the other for weight loss.
“Let’s not think of this as an approach that’s better than calorie restriction,” William Yancy, MD, MHS, an internist and weight management specialist at Duke Lifestyle and Weight Management Center, Durham, N.C., said in an interview. “It’s an alternative approach to calorie restriction.”
Participants’ willingness to adhere to the diet likely accounted for the percentage difference between the groups, study author Vasiliki Pavlou, RDN, told this news organization. Ms. Pavlou presented the findings at the Nutrition 2023 conference.
“People that have type 2 diabetes, they’ve already been to the doctor, they’ve already been told to count calories,” said Ms. Pavlou, a doctoral student at the University of Illinois at Chicago. “There were many weeks where they came to us with nothing on MyFitnessPal and we’d have to encourage them to start tracking again.”
The intermittent-fasting group adhered to the eating time window 6 out of 7 days of the week, with a 1-hour grace period for the noon-to-8-p.m. window. In comparison, one-third of the calorie-restriction group didn’t stay within 200 calories of the goal, according to Ms. Pavlou.
That meant the fasting group cut about 100 calories more per day than the calorie-restriction group, which was reflected in their weight loss, Ms. Pavlou said.
A1c levels dropped by about 1% in both the intermittent-fasting and calorie-restriction groups – a meaningful decrease, said Dr. Yancy. “I think a 0.5% difference would have some clinical significance in terms of complications from diabetes,” he said. “So 1% would be even more clinically meaningful.”
However, fewer participants taking insulin in the calorie-restriction group could explain the difference, according to Ms. Pavlou. “Usually, when someone goes on insulin, their pancreas is already not functioning as well,” she said. “And it’s way harder to see improvements in their A1c and glycemic control.”
Up to 90% of people with type 2 diabetes are overweight or obese. Weight loss is one of the major components of type 2 diabetes care, according to the American Diabetes Association, and studies have shown that even a 5% reduction in body weight can reduce blood glucose concentration and A1c. Some studies suggest diabetes remission can occur after a 10% loss in body weight, but Dr. Yancy said it depends on the person.
“It depends on the individual, their metabolic situation, how long they’ve had diabetes, what kind of approach they’re following, maybe what medicines they’re taking,” Dr. Yancy said. “There’s a lot of different factors involved in remission.”
The study cohort generally had advanced diabetes and was taking a mix of medications, so the results might not be applicable to people with a more recent diabetes diagnosis, according to Ms. Pavlou.
Dr. Yancy said intermittent fasting could work well for the right person. The success of the approach could depend on a person’s eating habits and whether their meals usually fall outside the time-restricted window, or it could depend on how well a person follows rules, according to Dr. Yancy.
“Some people might not eat much after 8 o’clock, and some people might skip breakfast,” Dr. Yancy said. “And if that’s the case, then it’s not going to make a big impact on their weight probably.”
Medication is also an important consideration. Not eating can be dangerous for patients taking short-acting insulin or sulfonylureas, according to Dr. Yancy.
Ms. Pavlou said these findings show intermittent fasting is another option for patients with type 2 diabetes trying to lose weight. “If you’ve tried calorie counting, that’s not working for you or if you’re kind of burnt out, this is something else that you could try,” she said.
“We have a lot of patients that need to lose weight, and we have patients who respond differently to different approaches,” said Dr. Yancy. “So having various approaches is really valuable.”
The manuscript is currently under review at JAMA Internal Medicine, said Ms. Pavlou.
A version of this article appeared on Medscape.com.
BOSTON –
For the study, 57 overweight and obese participants with type 2 diabetes were randomly assigned to three different groups: The first group ate between noon and 8 p.m., the second was asked to reduce caloric intake by 25% of maintenance calories, and the third, a control group, continued eating normally.
The calorie-restriction group tracked intake on MyFitnessPal, an app that logs the calorie content of different foods. Both the intermittent-fasting and calorie-restriction groups were assigned a dietitian to help with adherence.
After 6 months, participants in the intermittent-fasting group lost about 4.3% of body weight – the equivalent of 10 pounds of weight loss for a person weighing 230 pounds – whereas participants in the calorie-restriction group lost about 2.5% of body weight.
The difference between the two groups was not significant, so one approach isn’t necessarily better than the other for weight loss.
“Let’s not think of this as an approach that’s better than calorie restriction,” William Yancy, MD, MHS, an internist and weight management specialist at Duke Lifestyle and Weight Management Center, Durham, N.C., said in an interview. “It’s an alternative approach to calorie restriction.”
Participants’ willingness to adhere to the diet likely accounted for the percentage difference between the groups, study author Vasiliki Pavlou, RDN, told this news organization. Ms. Pavlou presented the findings at the Nutrition 2023 conference.
“People that have type 2 diabetes, they’ve already been to the doctor, they’ve already been told to count calories,” said Ms. Pavlou, a doctoral student at the University of Illinois at Chicago. “There were many weeks where they came to us with nothing on MyFitnessPal and we’d have to encourage them to start tracking again.”
The intermittent-fasting group adhered to the eating time window 6 out of 7 days of the week, with a 1-hour grace period for the noon-to-8-p.m. window. In comparison, one-third of the calorie-restriction group didn’t stay within 200 calories of the goal, according to Ms. Pavlou.
That meant the fasting group cut about 100 calories more per day than the calorie-restriction group, which was reflected in their weight loss, Ms. Pavlou said.
A1c levels dropped by about 1% in both the intermittent-fasting and calorie-restriction groups – a meaningful decrease, said Dr. Yancy. “I think a 0.5% difference would have some clinical significance in terms of complications from diabetes,” he said. “So 1% would be even more clinically meaningful.”
However, fewer participants taking insulin in the calorie-restriction group could explain the difference, according to Ms. Pavlou. “Usually, when someone goes on insulin, their pancreas is already not functioning as well,” she said. “And it’s way harder to see improvements in their A1c and glycemic control.”
Up to 90% of people with type 2 diabetes are overweight or obese. Weight loss is one of the major components of type 2 diabetes care, according to the American Diabetes Association, and studies have shown that even a 5% reduction in body weight can reduce blood glucose concentration and A1c. Some studies suggest diabetes remission can occur after a 10% loss in body weight, but Dr. Yancy said it depends on the person.
“It depends on the individual, their metabolic situation, how long they’ve had diabetes, what kind of approach they’re following, maybe what medicines they’re taking,” Dr. Yancy said. “There’s a lot of different factors involved in remission.”
The study cohort generally had advanced diabetes and was taking a mix of medications, so the results might not be applicable to people with a more recent diabetes diagnosis, according to Ms. Pavlou.
Dr. Yancy said intermittent fasting could work well for the right person. The success of the approach could depend on a person’s eating habits and whether their meals usually fall outside the time-restricted window, or it could depend on how well a person follows rules, according to Dr. Yancy.
“Some people might not eat much after 8 o’clock, and some people might skip breakfast,” Dr. Yancy said. “And if that’s the case, then it’s not going to make a big impact on their weight probably.”
Medication is also an important consideration. Not eating can be dangerous for patients taking short-acting insulin or sulfonylureas, according to Dr. Yancy.
Ms. Pavlou said these findings show intermittent fasting is another option for patients with type 2 diabetes trying to lose weight. “If you’ve tried calorie counting, that’s not working for you or if you’re kind of burnt out, this is something else that you could try,” she said.
“We have a lot of patients that need to lose weight, and we have patients who respond differently to different approaches,” said Dr. Yancy. “So having various approaches is really valuable.”
The manuscript is currently under review at JAMA Internal Medicine, said Ms. Pavlou.
A version of this article appeared on Medscape.com.
BOSTON –
For the study, 57 overweight and obese participants with type 2 diabetes were randomly assigned to three different groups: The first group ate between noon and 8 p.m., the second was asked to reduce caloric intake by 25% of maintenance calories, and the third, a control group, continued eating normally.
The calorie-restriction group tracked intake on MyFitnessPal, an app that logs the calorie content of different foods. Both the intermittent-fasting and calorie-restriction groups were assigned a dietitian to help with adherence.
After 6 months, participants in the intermittent-fasting group lost about 4.3% of body weight – the equivalent of 10 pounds of weight loss for a person weighing 230 pounds – whereas participants in the calorie-restriction group lost about 2.5% of body weight.
The difference between the two groups was not significant, so one approach isn’t necessarily better than the other for weight loss.
“Let’s not think of this as an approach that’s better than calorie restriction,” William Yancy, MD, MHS, an internist and weight management specialist at Duke Lifestyle and Weight Management Center, Durham, N.C., said in an interview. “It’s an alternative approach to calorie restriction.”
Participants’ willingness to adhere to the diet likely accounted for the percentage difference between the groups, study author Vasiliki Pavlou, RDN, told this news organization. Ms. Pavlou presented the findings at the Nutrition 2023 conference.
“People that have type 2 diabetes, they’ve already been to the doctor, they’ve already been told to count calories,” said Ms. Pavlou, a doctoral student at the University of Illinois at Chicago. “There were many weeks where they came to us with nothing on MyFitnessPal and we’d have to encourage them to start tracking again.”
The intermittent-fasting group adhered to the eating time window 6 out of 7 days of the week, with a 1-hour grace period for the noon-to-8-p.m. window. In comparison, one-third of the calorie-restriction group didn’t stay within 200 calories of the goal, according to Ms. Pavlou.
That meant the fasting group cut about 100 calories more per day than the calorie-restriction group, which was reflected in their weight loss, Ms. Pavlou said.
A1c levels dropped by about 1% in both the intermittent-fasting and calorie-restriction groups – a meaningful decrease, said Dr. Yancy. “I think a 0.5% difference would have some clinical significance in terms of complications from diabetes,” he said. “So 1% would be even more clinically meaningful.”
However, fewer participants taking insulin in the calorie-restriction group could explain the difference, according to Ms. Pavlou. “Usually, when someone goes on insulin, their pancreas is already not functioning as well,” she said. “And it’s way harder to see improvements in their A1c and glycemic control.”
Up to 90% of people with type 2 diabetes are overweight or obese. Weight loss is one of the major components of type 2 diabetes care, according to the American Diabetes Association, and studies have shown that even a 5% reduction in body weight can reduce blood glucose concentration and A1c. Some studies suggest diabetes remission can occur after a 10% loss in body weight, but Dr. Yancy said it depends on the person.
“It depends on the individual, their metabolic situation, how long they’ve had diabetes, what kind of approach they’re following, maybe what medicines they’re taking,” Dr. Yancy said. “There’s a lot of different factors involved in remission.”
The study cohort generally had advanced diabetes and was taking a mix of medications, so the results might not be applicable to people with a more recent diabetes diagnosis, according to Ms. Pavlou.
Dr. Yancy said intermittent fasting could work well for the right person. The success of the approach could depend on a person’s eating habits and whether their meals usually fall outside the time-restricted window, or it could depend on how well a person follows rules, according to Dr. Yancy.
“Some people might not eat much after 8 o’clock, and some people might skip breakfast,” Dr. Yancy said. “And if that’s the case, then it’s not going to make a big impact on their weight probably.”
Medication is also an important consideration. Not eating can be dangerous for patients taking short-acting insulin or sulfonylureas, according to Dr. Yancy.
Ms. Pavlou said these findings show intermittent fasting is another option for patients with type 2 diabetes trying to lose weight. “If you’ve tried calorie counting, that’s not working for you or if you’re kind of burnt out, this is something else that you could try,” she said.
“We have a lot of patients that need to lose weight, and we have patients who respond differently to different approaches,” said Dr. Yancy. “So having various approaches is really valuable.”
The manuscript is currently under review at JAMA Internal Medicine, said Ms. Pavlou.
A version of this article appeared on Medscape.com.
AT NUTRITION 2023
CDC offers guidance on RSV vaccines for adults
Two newly approved respiratory syncytial virus (RSV) vaccines for adults aged 60 years and older may be able to prevent illness in those at risk for severe RSV disease.
Most adult RSV illness occurs among the older age group and results in an estimated 60,000-160,000 hospitalizations and 6,000-10,000 deaths per year among people aged at least 65 years.
Older adults deciding whether to get the vaccines should weigh risks and their own preferences and make the decision in consultation with their clinicians, said authors of a Centers for Disease Control and Prevention report.
Michael Melgar, MD, with the Coronavirus and Other Respiratory Viruses Division at the CDC, was lead author on the report, published in the Morbidity and Mortality Weekly Report.
Two new vaccines
In May, the Food and Drug Administration approved the first of two vaccines for preventing RSV lower respiratory tract disease for adults aged at least 60 years.
On June 21, the Advisory Committee on Immunization Practices (ACIP) recommended that people in that age group receive a single dose of RSV vaccine using shared decision-making.
The recommendation for shared decision-making makes the ACIP decision different from routine and risk-based vaccine recommendations. Rather than targeting all in a particular age group or risk group, the decision calls for consideration of a patients’ risk for disease and their characteristics, preferences, and values; the health care professional’s clinical discretion; and performance of the vaccine.
Dr. Melgar and colleagues reported that vaccination with one dose of the GSK or Pfizer RSV vaccines has proved moderately to highly effective in preventing symptomatic RSV-associated lower respiratory tract disease over two consecutive RSV seasons among people aged 60 and older.
The trials that led to approval weren’t powered to gauge efficacy against RSV-associated hospitalization and death. However, the authors wrote, the prevention of lower respiratory tract disease, including medically attended illness, suggests that the shots might prevent considerable morbidity from RSV disease among those aged 60 and older.
Both vaccines were generally well tolerated with a good safety profile. However, six cases of inflammatory neurologic events (including Guillain-Barré Syndrome, acute disseminated encephalomyelitis, and others) were reported in clinical trials after RSV vaccination.
“Whether these events occurred due to chance, or whether RSV vaccination increases the risk for inflammatory neurologic events, is currently unknown,” the authors wrote.
Postmarketing surveillance may help clarify the existence of any potential risk, but until those results are clearer, the CDC researchers said, RSV vaccinations should be targeted to older adults at highest risk for severe RSV and those most likely to benefit from the shots.
At higher risk
Some adults with certain medical conditions have a higher risk for RSV-associated hospitalization, according to the report.
Those conditions include chronic obstructive pulmonary disease, asthma, heart failure, coronary artery disease, cerebrovascular disease, diabetes mellitus, and chronic kidney disease.
People who are frail and of advanced age also are at higher risk for RSV hospitalization. That risk increases with age and the highest risk is for people aged at least 75 years.
The researchers added that RSV can cause severe disease in those with compromised immunity, including people who have received hematopoietic stem cell transplants and patients taking immunosuppressive drugs such as those used with solid organ transplants, cancer treatment, or other conditions.
As for when physicians should offer the vaccinations, shots are optimally given before the start of the RSV season.
However, the COVID-19 pandemic interrupted the seasonality and the timing has not yet returned to prepandemic patterns.
For the 2023-24 season, this report states, clinicians should offer RSV vaccination to adults aged at least 60 years using shared clinical decision-making as early as vaccine supply is available and should continue to offer vaccination to eligible adults who remain unvaccinated.
RSV vaccines can be administered with other adult vaccines during the same visit, the authors confirmed.
A version of this article first appeared on Medscape.com.
Two newly approved respiratory syncytial virus (RSV) vaccines for adults aged 60 years and older may be able to prevent illness in those at risk for severe RSV disease.
Most adult RSV illness occurs among the older age group and results in an estimated 60,000-160,000 hospitalizations and 6,000-10,000 deaths per year among people aged at least 65 years.
Older adults deciding whether to get the vaccines should weigh risks and their own preferences and make the decision in consultation with their clinicians, said authors of a Centers for Disease Control and Prevention report.
Michael Melgar, MD, with the Coronavirus and Other Respiratory Viruses Division at the CDC, was lead author on the report, published in the Morbidity and Mortality Weekly Report.
Two new vaccines
In May, the Food and Drug Administration approved the first of two vaccines for preventing RSV lower respiratory tract disease for adults aged at least 60 years.
On June 21, the Advisory Committee on Immunization Practices (ACIP) recommended that people in that age group receive a single dose of RSV vaccine using shared decision-making.
The recommendation for shared decision-making makes the ACIP decision different from routine and risk-based vaccine recommendations. Rather than targeting all in a particular age group or risk group, the decision calls for consideration of a patients’ risk for disease and their characteristics, preferences, and values; the health care professional’s clinical discretion; and performance of the vaccine.
Dr. Melgar and colleagues reported that vaccination with one dose of the GSK or Pfizer RSV vaccines has proved moderately to highly effective in preventing symptomatic RSV-associated lower respiratory tract disease over two consecutive RSV seasons among people aged 60 and older.
The trials that led to approval weren’t powered to gauge efficacy against RSV-associated hospitalization and death. However, the authors wrote, the prevention of lower respiratory tract disease, including medically attended illness, suggests that the shots might prevent considerable morbidity from RSV disease among those aged 60 and older.
Both vaccines were generally well tolerated with a good safety profile. However, six cases of inflammatory neurologic events (including Guillain-Barré Syndrome, acute disseminated encephalomyelitis, and others) were reported in clinical trials after RSV vaccination.
“Whether these events occurred due to chance, or whether RSV vaccination increases the risk for inflammatory neurologic events, is currently unknown,” the authors wrote.
Postmarketing surveillance may help clarify the existence of any potential risk, but until those results are clearer, the CDC researchers said, RSV vaccinations should be targeted to older adults at highest risk for severe RSV and those most likely to benefit from the shots.
At higher risk
Some adults with certain medical conditions have a higher risk for RSV-associated hospitalization, according to the report.
Those conditions include chronic obstructive pulmonary disease, asthma, heart failure, coronary artery disease, cerebrovascular disease, diabetes mellitus, and chronic kidney disease.
People who are frail and of advanced age also are at higher risk for RSV hospitalization. That risk increases with age and the highest risk is for people aged at least 75 years.
The researchers added that RSV can cause severe disease in those with compromised immunity, including people who have received hematopoietic stem cell transplants and patients taking immunosuppressive drugs such as those used with solid organ transplants, cancer treatment, or other conditions.
As for when physicians should offer the vaccinations, shots are optimally given before the start of the RSV season.
However, the COVID-19 pandemic interrupted the seasonality and the timing has not yet returned to prepandemic patterns.
For the 2023-24 season, this report states, clinicians should offer RSV vaccination to adults aged at least 60 years using shared clinical decision-making as early as vaccine supply is available and should continue to offer vaccination to eligible adults who remain unvaccinated.
RSV vaccines can be administered with other adult vaccines during the same visit, the authors confirmed.
A version of this article first appeared on Medscape.com.
Two newly approved respiratory syncytial virus (RSV) vaccines for adults aged 60 years and older may be able to prevent illness in those at risk for severe RSV disease.
Most adult RSV illness occurs among the older age group and results in an estimated 60,000-160,000 hospitalizations and 6,000-10,000 deaths per year among people aged at least 65 years.
Older adults deciding whether to get the vaccines should weigh risks and their own preferences and make the decision in consultation with their clinicians, said authors of a Centers for Disease Control and Prevention report.
Michael Melgar, MD, with the Coronavirus and Other Respiratory Viruses Division at the CDC, was lead author on the report, published in the Morbidity and Mortality Weekly Report.
Two new vaccines
In May, the Food and Drug Administration approved the first of two vaccines for preventing RSV lower respiratory tract disease for adults aged at least 60 years.
On June 21, the Advisory Committee on Immunization Practices (ACIP) recommended that people in that age group receive a single dose of RSV vaccine using shared decision-making.
The recommendation for shared decision-making makes the ACIP decision different from routine and risk-based vaccine recommendations. Rather than targeting all in a particular age group or risk group, the decision calls for consideration of a patients’ risk for disease and their characteristics, preferences, and values; the health care professional’s clinical discretion; and performance of the vaccine.
Dr. Melgar and colleagues reported that vaccination with one dose of the GSK or Pfizer RSV vaccines has proved moderately to highly effective in preventing symptomatic RSV-associated lower respiratory tract disease over two consecutive RSV seasons among people aged 60 and older.
The trials that led to approval weren’t powered to gauge efficacy against RSV-associated hospitalization and death. However, the authors wrote, the prevention of lower respiratory tract disease, including medically attended illness, suggests that the shots might prevent considerable morbidity from RSV disease among those aged 60 and older.
Both vaccines were generally well tolerated with a good safety profile. However, six cases of inflammatory neurologic events (including Guillain-Barré Syndrome, acute disseminated encephalomyelitis, and others) were reported in clinical trials after RSV vaccination.
“Whether these events occurred due to chance, or whether RSV vaccination increases the risk for inflammatory neurologic events, is currently unknown,” the authors wrote.
Postmarketing surveillance may help clarify the existence of any potential risk, but until those results are clearer, the CDC researchers said, RSV vaccinations should be targeted to older adults at highest risk for severe RSV and those most likely to benefit from the shots.
At higher risk
Some adults with certain medical conditions have a higher risk for RSV-associated hospitalization, according to the report.
Those conditions include chronic obstructive pulmonary disease, asthma, heart failure, coronary artery disease, cerebrovascular disease, diabetes mellitus, and chronic kidney disease.
People who are frail and of advanced age also are at higher risk for RSV hospitalization. That risk increases with age and the highest risk is for people aged at least 75 years.
The researchers added that RSV can cause severe disease in those with compromised immunity, including people who have received hematopoietic stem cell transplants and patients taking immunosuppressive drugs such as those used with solid organ transplants, cancer treatment, or other conditions.
As for when physicians should offer the vaccinations, shots are optimally given before the start of the RSV season.
However, the COVID-19 pandemic interrupted the seasonality and the timing has not yet returned to prepandemic patterns.
For the 2023-24 season, this report states, clinicians should offer RSV vaccination to adults aged at least 60 years using shared clinical decision-making as early as vaccine supply is available and should continue to offer vaccination to eligible adults who remain unvaccinated.
RSV vaccines can be administered with other adult vaccines during the same visit, the authors confirmed.
A version of this article first appeared on Medscape.com.
FROM THE MMWR
Daily statin cuts cardiovascular risk in HIV
BRISBANE, AUSTRALIA – that show pitavastatin therapy is associated with a significantly lower risk of cardiovascular events than placebo.
“There was a significant 35% lower risk of major adverse cardiovascular events after a median follow-up of 5.1 years “ said Steven Grinspoon, MD, from Massachusetts General Hospital and Harvard Medical School in Boston, who presented the final analysis of data from the REPRIEVE trial at the International AIDS Society Conference on HIV Science.
The results were simultaneously published in the New England Journal of Medicine. The primary endpoint of major adverse cardiovascular events included a composite of outcomes that included cardiovascular death, stroke, myocardial infarction, hospitalization for unstable angina, and transient ischemic attack among those treated with pitavastatin, compared with placebo (95% confidence interval, 0.48-0.90; P = .002).
The REPRIEVE trial was halted earlier this year for efficacy after an interim analysis pointed to a significantly lower rate of cardiovascular events in the treatment group.
The international double-blind, placebo-controlled trial randomly assigned 7,769 people with HIV infection, who were at low to moderate risk of cardiovascular disease, to either 4 mg daily of pitavastatin calcium or placebo.
The secondary outcome – a composite of major cardiovascular events and all-cause mortality – also showed a significant 21% reduction in risk with pitavastatin treatment, compared with placebo (95% CI, 0.65-0.96).
Cardiovascular events in HIV
HIV infection is an independent risk factor for cardiovascular disease, Dr. Grinspoon pointed out, and those living with HIV have about double the risk of myocardial infarction and stroke, compared with the general population.
“There’s an unmet need for people living with HIV who have low to moderate traditional risk, for whom HIV is even considered a risk equivalent but for whom no primary prevention strategy has been tested in a large trial,” Dr. Grinspoon said during an interview.
Those enrolled in the study had a 10-year Atherosclerotic Cardiovascular Disease risk score ranging from 2.1% to 7%, with a median of 4.5%. While LDL cholesterol levels at baseline ranged from 87 to 128 mg/dL, the study showed a similar reduction in cardiovascular risk regardless of LDL.
“These are types of people who, if they came to the doctor’s office right now before REPRIEVE, they would largely be told your risk score is not really making you eligible for a statin,” Dr. Grinspoon said.
He explained that what is most interesting about the reduction in risk is that it was nearly twice what would be expected with LDL lowering, based on what has previously been seen in statin trials in non–HIV-positive populations.
“I think the data are suggesting that it’s certainly in part due to the reduction in LDL – that is very important – but it’s also due to other factors beyond changes in LDL,” Dr. Grinspoon said. He speculated that the statin could be affecting anti-inflammatory and immune pathways, and that this could account for some of the reduction in cardiovascular risk, but “those data are cooking, and they’re being analyzed as we speak.”
In a substudy analysis of REPRIEVE, Markella Zanni, MD, associate professor of medicine at Harvard Medical School and Massachusetts General Hospital, focused on the women in the clinical trial.
Women’s risk
In REPRIEVE, 31.1% of the study population were women. Dr. Zanni and her team investigated whether there are differences in the way HIV affects the risk of developing atherosclerotic cardiovascular disease in women, compared with men.
They found that women have both higher levels of inflammatory markers, such as interleukin-6, C-reactive protein, and D-dimer, but a lower prevalence of coronary artery plaques than men.
“This finding represents an interesting paradox given that high levels of select inflammatory markers have been associated with coronary artery plaque, both among women living with HIV and among men living with HIV,” Dr. Zanni explained.
She says the researchers were hoping to further explore whether inflammation is fueling the increased risk for atherosclerotic disease, and particularly the higher risk evident in women living with HIV, compared with men.
“Women living with HIV should discuss with their treating clinicians heart risks and possible prevention strategies, including statin therapy coupled with healthy lifestyle changes addressing modifiable, traditional metabolic risk factors” she said.
Time for primary prevention?
All patients in the study were on antiretroviral therapy and investigators report that pitavastatin does not interact with these medications. The median CD4 cell count was 621 cells/mm3, and 87.5% of participants had an HIV viral load below the lower limit of quantification.
Participants were enrolled from 12 countries including the United States, Spain, Brazil, South Africa, and Thailand, and around two-thirds were non-White. Individuals of South Asian ethnicity showed the biggest reduction in cardiovascular risk with pitavastatin treatment.
There was a 74% higher rate of muscle pain and weakness in the pitavastatin group – affecting 91 people in the treatment arm and 53 in the placebo arm – but the majority were low grade. The rate of rhabdomyolysis of grade 3 or above was lower in the statin group, with three cases, compared with four cases in the placebo group.
Commenting on the findings, Laura Waters, MD, a genitourinary and HIV medicine consultant at Central and North West London NHS Foundation Trust’s Mortimer Market Centre, said that, while HIV infection was considered a risk factor for cardiovascular disease, risk calculators don’t specifically adjust for HIV infection.
“Now that we’ve got effective HIV drugs and people can enjoy normal life expectancy, cardiovascular disease is a particular issue for people with HIV,” she said.
Dr. Waters, who was not involved with the study, suggested that people living with HIV should discuss the use of statins with their doctor, but she acknowledged there are some barriers to treatment in people living with HIV. “It’s another pill, and when it’s a borderline [decision] it is easy to say, ‘I have to think about it,’ ” she said, with the result that statin treatment is often deferred.
The REPRIEVE study was supported by grants from the National Institutes of Health, Kowa Pharmaceuticals America, Gilead Sciences, and ViiV Healthcare. Dr. Grinspoon declared institutional grants from National Institutes of Health, Kowa Pharmaceuticals America, Gilead Sciences, and ViiV Healthcare and consultancies unrelated to the study. Dr. Zanni reports no relevant financial relationships.
A version of this article first appeared on Medscape.com.
BRISBANE, AUSTRALIA – that show pitavastatin therapy is associated with a significantly lower risk of cardiovascular events than placebo.
“There was a significant 35% lower risk of major adverse cardiovascular events after a median follow-up of 5.1 years “ said Steven Grinspoon, MD, from Massachusetts General Hospital and Harvard Medical School in Boston, who presented the final analysis of data from the REPRIEVE trial at the International AIDS Society Conference on HIV Science.
The results were simultaneously published in the New England Journal of Medicine. The primary endpoint of major adverse cardiovascular events included a composite of outcomes that included cardiovascular death, stroke, myocardial infarction, hospitalization for unstable angina, and transient ischemic attack among those treated with pitavastatin, compared with placebo (95% confidence interval, 0.48-0.90; P = .002).
The REPRIEVE trial was halted earlier this year for efficacy after an interim analysis pointed to a significantly lower rate of cardiovascular events in the treatment group.
The international double-blind, placebo-controlled trial randomly assigned 7,769 people with HIV infection, who were at low to moderate risk of cardiovascular disease, to either 4 mg daily of pitavastatin calcium or placebo.
The secondary outcome – a composite of major cardiovascular events and all-cause mortality – also showed a significant 21% reduction in risk with pitavastatin treatment, compared with placebo (95% CI, 0.65-0.96).
Cardiovascular events in HIV
HIV infection is an independent risk factor for cardiovascular disease, Dr. Grinspoon pointed out, and those living with HIV have about double the risk of myocardial infarction and stroke, compared with the general population.
“There’s an unmet need for people living with HIV who have low to moderate traditional risk, for whom HIV is even considered a risk equivalent but for whom no primary prevention strategy has been tested in a large trial,” Dr. Grinspoon said during an interview.
Those enrolled in the study had a 10-year Atherosclerotic Cardiovascular Disease risk score ranging from 2.1% to 7%, with a median of 4.5%. While LDL cholesterol levels at baseline ranged from 87 to 128 mg/dL, the study showed a similar reduction in cardiovascular risk regardless of LDL.
“These are types of people who, if they came to the doctor’s office right now before REPRIEVE, they would largely be told your risk score is not really making you eligible for a statin,” Dr. Grinspoon said.
He explained that what is most interesting about the reduction in risk is that it was nearly twice what would be expected with LDL lowering, based on what has previously been seen in statin trials in non–HIV-positive populations.
“I think the data are suggesting that it’s certainly in part due to the reduction in LDL – that is very important – but it’s also due to other factors beyond changes in LDL,” Dr. Grinspoon said. He speculated that the statin could be affecting anti-inflammatory and immune pathways, and that this could account for some of the reduction in cardiovascular risk, but “those data are cooking, and they’re being analyzed as we speak.”
In a substudy analysis of REPRIEVE, Markella Zanni, MD, associate professor of medicine at Harvard Medical School and Massachusetts General Hospital, focused on the women in the clinical trial.
Women’s risk
In REPRIEVE, 31.1% of the study population were women. Dr. Zanni and her team investigated whether there are differences in the way HIV affects the risk of developing atherosclerotic cardiovascular disease in women, compared with men.
They found that women have both higher levels of inflammatory markers, such as interleukin-6, C-reactive protein, and D-dimer, but a lower prevalence of coronary artery plaques than men.
“This finding represents an interesting paradox given that high levels of select inflammatory markers have been associated with coronary artery plaque, both among women living with HIV and among men living with HIV,” Dr. Zanni explained.
She says the researchers were hoping to further explore whether inflammation is fueling the increased risk for atherosclerotic disease, and particularly the higher risk evident in women living with HIV, compared with men.
“Women living with HIV should discuss with their treating clinicians heart risks and possible prevention strategies, including statin therapy coupled with healthy lifestyle changes addressing modifiable, traditional metabolic risk factors” she said.
Time for primary prevention?
All patients in the study were on antiretroviral therapy and investigators report that pitavastatin does not interact with these medications. The median CD4 cell count was 621 cells/mm3, and 87.5% of participants had an HIV viral load below the lower limit of quantification.
Participants were enrolled from 12 countries including the United States, Spain, Brazil, South Africa, and Thailand, and around two-thirds were non-White. Individuals of South Asian ethnicity showed the biggest reduction in cardiovascular risk with pitavastatin treatment.
There was a 74% higher rate of muscle pain and weakness in the pitavastatin group – affecting 91 people in the treatment arm and 53 in the placebo arm – but the majority were low grade. The rate of rhabdomyolysis of grade 3 or above was lower in the statin group, with three cases, compared with four cases in the placebo group.
Commenting on the findings, Laura Waters, MD, a genitourinary and HIV medicine consultant at Central and North West London NHS Foundation Trust’s Mortimer Market Centre, said that, while HIV infection was considered a risk factor for cardiovascular disease, risk calculators don’t specifically adjust for HIV infection.
“Now that we’ve got effective HIV drugs and people can enjoy normal life expectancy, cardiovascular disease is a particular issue for people with HIV,” she said.
Dr. Waters, who was not involved with the study, suggested that people living with HIV should discuss the use of statins with their doctor, but she acknowledged there are some barriers to treatment in people living with HIV. “It’s another pill, and when it’s a borderline [decision] it is easy to say, ‘I have to think about it,’ ” she said, with the result that statin treatment is often deferred.
The REPRIEVE study was supported by grants from the National Institutes of Health, Kowa Pharmaceuticals America, Gilead Sciences, and ViiV Healthcare. Dr. Grinspoon declared institutional grants from National Institutes of Health, Kowa Pharmaceuticals America, Gilead Sciences, and ViiV Healthcare and consultancies unrelated to the study. Dr. Zanni reports no relevant financial relationships.
A version of this article first appeared on Medscape.com.
BRISBANE, AUSTRALIA – that show pitavastatin therapy is associated with a significantly lower risk of cardiovascular events than placebo.
“There was a significant 35% lower risk of major adverse cardiovascular events after a median follow-up of 5.1 years “ said Steven Grinspoon, MD, from Massachusetts General Hospital and Harvard Medical School in Boston, who presented the final analysis of data from the REPRIEVE trial at the International AIDS Society Conference on HIV Science.
The results were simultaneously published in the New England Journal of Medicine. The primary endpoint of major adverse cardiovascular events included a composite of outcomes that included cardiovascular death, stroke, myocardial infarction, hospitalization for unstable angina, and transient ischemic attack among those treated with pitavastatin, compared with placebo (95% confidence interval, 0.48-0.90; P = .002).
The REPRIEVE trial was halted earlier this year for efficacy after an interim analysis pointed to a significantly lower rate of cardiovascular events in the treatment group.
The international double-blind, placebo-controlled trial randomly assigned 7,769 people with HIV infection, who were at low to moderate risk of cardiovascular disease, to either 4 mg daily of pitavastatin calcium or placebo.
The secondary outcome – a composite of major cardiovascular events and all-cause mortality – also showed a significant 21% reduction in risk with pitavastatin treatment, compared with placebo (95% CI, 0.65-0.96).
Cardiovascular events in HIV
HIV infection is an independent risk factor for cardiovascular disease, Dr. Grinspoon pointed out, and those living with HIV have about double the risk of myocardial infarction and stroke, compared with the general population.
“There’s an unmet need for people living with HIV who have low to moderate traditional risk, for whom HIV is even considered a risk equivalent but for whom no primary prevention strategy has been tested in a large trial,” Dr. Grinspoon said during an interview.
Those enrolled in the study had a 10-year Atherosclerotic Cardiovascular Disease risk score ranging from 2.1% to 7%, with a median of 4.5%. While LDL cholesterol levels at baseline ranged from 87 to 128 mg/dL, the study showed a similar reduction in cardiovascular risk regardless of LDL.
“These are types of people who, if they came to the doctor’s office right now before REPRIEVE, they would largely be told your risk score is not really making you eligible for a statin,” Dr. Grinspoon said.
He explained that what is most interesting about the reduction in risk is that it was nearly twice what would be expected with LDL lowering, based on what has previously been seen in statin trials in non–HIV-positive populations.
“I think the data are suggesting that it’s certainly in part due to the reduction in LDL – that is very important – but it’s also due to other factors beyond changes in LDL,” Dr. Grinspoon said. He speculated that the statin could be affecting anti-inflammatory and immune pathways, and that this could account for some of the reduction in cardiovascular risk, but “those data are cooking, and they’re being analyzed as we speak.”
In a substudy analysis of REPRIEVE, Markella Zanni, MD, associate professor of medicine at Harvard Medical School and Massachusetts General Hospital, focused on the women in the clinical trial.
Women’s risk
In REPRIEVE, 31.1% of the study population were women. Dr. Zanni and her team investigated whether there are differences in the way HIV affects the risk of developing atherosclerotic cardiovascular disease in women, compared with men.
They found that women have both higher levels of inflammatory markers, such as interleukin-6, C-reactive protein, and D-dimer, but a lower prevalence of coronary artery plaques than men.
“This finding represents an interesting paradox given that high levels of select inflammatory markers have been associated with coronary artery plaque, both among women living with HIV and among men living with HIV,” Dr. Zanni explained.
She says the researchers were hoping to further explore whether inflammation is fueling the increased risk for atherosclerotic disease, and particularly the higher risk evident in women living with HIV, compared with men.
“Women living with HIV should discuss with their treating clinicians heart risks and possible prevention strategies, including statin therapy coupled with healthy lifestyle changes addressing modifiable, traditional metabolic risk factors” she said.
Time for primary prevention?
All patients in the study were on antiretroviral therapy and investigators report that pitavastatin does not interact with these medications. The median CD4 cell count was 621 cells/mm3, and 87.5% of participants had an HIV viral load below the lower limit of quantification.
Participants were enrolled from 12 countries including the United States, Spain, Brazil, South Africa, and Thailand, and around two-thirds were non-White. Individuals of South Asian ethnicity showed the biggest reduction in cardiovascular risk with pitavastatin treatment.
There was a 74% higher rate of muscle pain and weakness in the pitavastatin group – affecting 91 people in the treatment arm and 53 in the placebo arm – but the majority were low grade. The rate of rhabdomyolysis of grade 3 or above was lower in the statin group, with three cases, compared with four cases in the placebo group.
Commenting on the findings, Laura Waters, MD, a genitourinary and HIV medicine consultant at Central and North West London NHS Foundation Trust’s Mortimer Market Centre, said that, while HIV infection was considered a risk factor for cardiovascular disease, risk calculators don’t specifically adjust for HIV infection.
“Now that we’ve got effective HIV drugs and people can enjoy normal life expectancy, cardiovascular disease is a particular issue for people with HIV,” she said.
Dr. Waters, who was not involved with the study, suggested that people living with HIV should discuss the use of statins with their doctor, but she acknowledged there are some barriers to treatment in people living with HIV. “It’s another pill, and when it’s a borderline [decision] it is easy to say, ‘I have to think about it,’ ” she said, with the result that statin treatment is often deferred.
The REPRIEVE study was supported by grants from the National Institutes of Health, Kowa Pharmaceuticals America, Gilead Sciences, and ViiV Healthcare. Dr. Grinspoon declared institutional grants from National Institutes of Health, Kowa Pharmaceuticals America, Gilead Sciences, and ViiV Healthcare and consultancies unrelated to the study. Dr. Zanni reports no relevant financial relationships.
A version of this article first appeared on Medscape.com.
AT IAS 2023
Even exercise by ‘weekend warriors’ can cut CV risk
Moderate to vigorous physical activity (MVPA) is a familiar and established approach to reducing cardiovascular (CV) risk, but it’s often believed that the exercise should be spread out across the week rather than concentrated within a couple of days.
A challenge to that view comes from an observational study of accelerometer-confirmed exercise in almost 90,000 people in their 60s. It suggests,
Researchers compared three patterns of MVPA in their subjects who wore accelerometers on their wrists for 1 week. Active WW subjects obtained at least 2.5 hours of exercise weekly, with at least half the amount completed over 1-2 days; “active regular” subjects achieved that exercise level but not mostly during 1 or 2 days; and those who were “inactive” fell short of 2.5 hours of exercise during the week. The group used a median exercise threshold of 3 hours, 50 minutes in a separate analysis.
The “active” groups, compared with inactive subjects, achieved similar and significant reductions in risk for incident atrial fibrillation (AF), myocardial infarction (MI), stroke, and heart failure (HF) over a median follow-up of 6.3 years at both weekly exercise thresholds, the group reported.
“The take-home [message] is that efforts to optimize activity, even if concentrated within just a day or 2 each week, should be expected to result in improved cardiovascular risk profiles,” lead author Shaan Khurshid, MD, MPH, Massachusetts General Hospital, Boston, said in an interview.
The study was published online in JAMA.
The research “provides novel data on patterns of physical activity accumulation and the risk of developing cardiovascular diseases,” observed Peter Katzmarzyk, PhD, Pennington Biomedical Research Center, Baton Rouge, La., in an interview. He was not involved with the research. Its “marked strengths,” he noted, include a large sample population and “use of accelerometers to measure physical activity levels and patterns.”
Moreover, Dr. Katzmarzyk said, its findings are “important” for showing that physical activity “can be accumulated throughout the week in different ways, which opens up more options for busy people to get their physical activity in.”
Current guidelines from the World Health Organization and the American Heart Association recommend at least 150 minutes of MVPA weekly to lower risk for cardiovascular disease and death, but do not specify an optimal exercise time frame. The U.K. National Health Service recommends MVPA daily or spread evenly over perhaps 4-5 days.
“The weekend warrior pattern has been studied previously, but typically relying on self-reported data, which may be biased, or [in studies] too small to look at specific cardiovascular outcomes,” Dr. Khurshid explained.
In the UK Biobank database, he said, “We saw the opportunity to leverage the largest sample of measured activity to date” to address the question of whether exercise time pattern “affects specific major cardiovascular diseases differently,” Dr. Khurshid said
The primary analysis assessed exercise amount in a week based on the guideline-recommended threshold of at least 2.5 hours; a 3-hour, 50-minutes threshold was used in a secondary analysis. The group assessed multiple thresholds because optimal MVPS levels derived from wrist-based accelerometers are “unclear,” he said.
The sample consisted of 89,573 participants with a mean age 62; slightly more than half (56%) were women. Based on the weekly MVPA threshold of 2.5 hours , the WW, active regular, and inactive groups made up 42.2%, 24%, and 33.7% of the population, respectively.
Compared with the inactive group, the two active groups both showed significant risk reductions for the four clinical outcomes, to similar degrees, in multivariate analysis. The results were similar at the 230-minute weekly exercise threshold for incident AF, MI, and HF but not for stroke.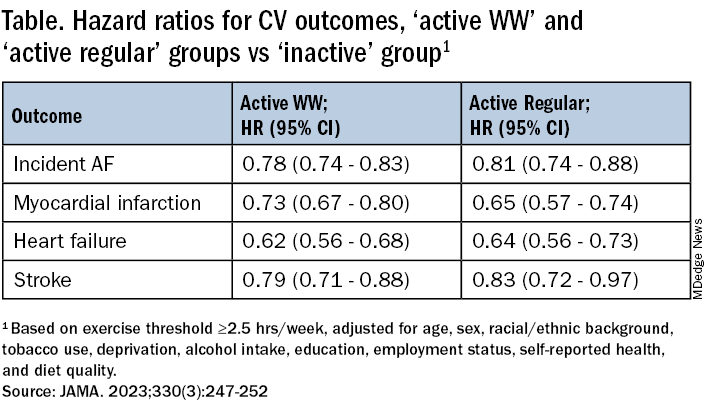
The findings were similarly consistent at the 3-hour, 50-minutes median threshold, although stroke differences were no longer significant.
Patients should be encouraged to exercise at recommended levels, “and should not be discouraged if, for whatever reasons, they are able to focus exercise within only 1 or a few days of the week,” said Dr. Khurshid. “Our findings suggest that it is the volume of activity, rather than the pattern, that matters most.”
The report notes several limitations of the study, including the exercise observation period limited to 1 week and that participants could have modified their behavior during the observation period. Also, the participants were almost all White, so the results may not be generalizable to other populations.
Clinicians should familiarize themselves with the “full range of recommendations” presented in the “Physical Activity Guidelines for Americans, 2nd Edition” “and personalize prescriptions by setting achievable physical activity goals” based on age, physical abilities, and activity levels, states an accompanying editorial from Dr. Katzmarzyk and John M. Jakicic, PhD, University of Kansas Medical Center, Kansas City.
Although MVPA at the recommended level of at least 2.5 hours per week will certainly be beneficial, they write, “the public health message should also clearly convey that every minute counts, especially among the three-quarters of U.S. adults who do not achieve that goal.”
Dr. Khurshid reported no relevant financial relationships; disclosures for the other authors are in the original article. Dr. Katzmarzyk reports no relevant financial relationships. Dr. Jakicic discloses receiving personal fees from Wondr Health, WW International (formerly Weight Watchers), and Educational Initiatives and grants from Epitomee Medical.
A version of this article appeared on Medscape.com.
Moderate to vigorous physical activity (MVPA) is a familiar and established approach to reducing cardiovascular (CV) risk, but it’s often believed that the exercise should be spread out across the week rather than concentrated within a couple of days.
A challenge to that view comes from an observational study of accelerometer-confirmed exercise in almost 90,000 people in their 60s. It suggests,
Researchers compared three patterns of MVPA in their subjects who wore accelerometers on their wrists for 1 week. Active WW subjects obtained at least 2.5 hours of exercise weekly, with at least half the amount completed over 1-2 days; “active regular” subjects achieved that exercise level but not mostly during 1 or 2 days; and those who were “inactive” fell short of 2.5 hours of exercise during the week. The group used a median exercise threshold of 3 hours, 50 minutes in a separate analysis.
The “active” groups, compared with inactive subjects, achieved similar and significant reductions in risk for incident atrial fibrillation (AF), myocardial infarction (MI), stroke, and heart failure (HF) over a median follow-up of 6.3 years at both weekly exercise thresholds, the group reported.
“The take-home [message] is that efforts to optimize activity, even if concentrated within just a day or 2 each week, should be expected to result in improved cardiovascular risk profiles,” lead author Shaan Khurshid, MD, MPH, Massachusetts General Hospital, Boston, said in an interview.
The study was published online in JAMA.
The research “provides novel data on patterns of physical activity accumulation and the risk of developing cardiovascular diseases,” observed Peter Katzmarzyk, PhD, Pennington Biomedical Research Center, Baton Rouge, La., in an interview. He was not involved with the research. Its “marked strengths,” he noted, include a large sample population and “use of accelerometers to measure physical activity levels and patterns.”
Moreover, Dr. Katzmarzyk said, its findings are “important” for showing that physical activity “can be accumulated throughout the week in different ways, which opens up more options for busy people to get their physical activity in.”
Current guidelines from the World Health Organization and the American Heart Association recommend at least 150 minutes of MVPA weekly to lower risk for cardiovascular disease and death, but do not specify an optimal exercise time frame. The U.K. National Health Service recommends MVPA daily or spread evenly over perhaps 4-5 days.
“The weekend warrior pattern has been studied previously, but typically relying on self-reported data, which may be biased, or [in studies] too small to look at specific cardiovascular outcomes,” Dr. Khurshid explained.
In the UK Biobank database, he said, “We saw the opportunity to leverage the largest sample of measured activity to date” to address the question of whether exercise time pattern “affects specific major cardiovascular diseases differently,” Dr. Khurshid said
The primary analysis assessed exercise amount in a week based on the guideline-recommended threshold of at least 2.5 hours; a 3-hour, 50-minutes threshold was used in a secondary analysis. The group assessed multiple thresholds because optimal MVPS levels derived from wrist-based accelerometers are “unclear,” he said.
The sample consisted of 89,573 participants with a mean age 62; slightly more than half (56%) were women. Based on the weekly MVPA threshold of 2.5 hours , the WW, active regular, and inactive groups made up 42.2%, 24%, and 33.7% of the population, respectively.
Compared with the inactive group, the two active groups both showed significant risk reductions for the four clinical outcomes, to similar degrees, in multivariate analysis. The results were similar at the 230-minute weekly exercise threshold for incident AF, MI, and HF but not for stroke.
The findings were similarly consistent at the 3-hour, 50-minutes median threshold, although stroke differences were no longer significant.
Patients should be encouraged to exercise at recommended levels, “and should not be discouraged if, for whatever reasons, they are able to focus exercise within only 1 or a few days of the week,” said Dr. Khurshid. “Our findings suggest that it is the volume of activity, rather than the pattern, that matters most.”
The report notes several limitations of the study, including the exercise observation period limited to 1 week and that participants could have modified their behavior during the observation period. Also, the participants were almost all White, so the results may not be generalizable to other populations.
Clinicians should familiarize themselves with the “full range of recommendations” presented in the “Physical Activity Guidelines for Americans, 2nd Edition” “and personalize prescriptions by setting achievable physical activity goals” based on age, physical abilities, and activity levels, states an accompanying editorial from Dr. Katzmarzyk and John M. Jakicic, PhD, University of Kansas Medical Center, Kansas City.
Although MVPA at the recommended level of at least 2.5 hours per week will certainly be beneficial, they write, “the public health message should also clearly convey that every minute counts, especially among the three-quarters of U.S. adults who do not achieve that goal.”
Dr. Khurshid reported no relevant financial relationships; disclosures for the other authors are in the original article. Dr. Katzmarzyk reports no relevant financial relationships. Dr. Jakicic discloses receiving personal fees from Wondr Health, WW International (formerly Weight Watchers), and Educational Initiatives and grants from Epitomee Medical.
A version of this article appeared on Medscape.com.
Moderate to vigorous physical activity (MVPA) is a familiar and established approach to reducing cardiovascular (CV) risk, but it’s often believed that the exercise should be spread out across the week rather than concentrated within a couple of days.
A challenge to that view comes from an observational study of accelerometer-confirmed exercise in almost 90,000 people in their 60s. It suggests,
Researchers compared three patterns of MVPA in their subjects who wore accelerometers on their wrists for 1 week. Active WW subjects obtained at least 2.5 hours of exercise weekly, with at least half the amount completed over 1-2 days; “active regular” subjects achieved that exercise level but not mostly during 1 or 2 days; and those who were “inactive” fell short of 2.5 hours of exercise during the week. The group used a median exercise threshold of 3 hours, 50 minutes in a separate analysis.
The “active” groups, compared with inactive subjects, achieved similar and significant reductions in risk for incident atrial fibrillation (AF), myocardial infarction (MI), stroke, and heart failure (HF) over a median follow-up of 6.3 years at both weekly exercise thresholds, the group reported.
“The take-home [message] is that efforts to optimize activity, even if concentrated within just a day or 2 each week, should be expected to result in improved cardiovascular risk profiles,” lead author Shaan Khurshid, MD, MPH, Massachusetts General Hospital, Boston, said in an interview.
The study was published online in JAMA.
The research “provides novel data on patterns of physical activity accumulation and the risk of developing cardiovascular diseases,” observed Peter Katzmarzyk, PhD, Pennington Biomedical Research Center, Baton Rouge, La., in an interview. He was not involved with the research. Its “marked strengths,” he noted, include a large sample population and “use of accelerometers to measure physical activity levels and patterns.”
Moreover, Dr. Katzmarzyk said, its findings are “important” for showing that physical activity “can be accumulated throughout the week in different ways, which opens up more options for busy people to get their physical activity in.”
Current guidelines from the World Health Organization and the American Heart Association recommend at least 150 minutes of MVPA weekly to lower risk for cardiovascular disease and death, but do not specify an optimal exercise time frame. The U.K. National Health Service recommends MVPA daily or spread evenly over perhaps 4-5 days.
“The weekend warrior pattern has been studied previously, but typically relying on self-reported data, which may be biased, or [in studies] too small to look at specific cardiovascular outcomes,” Dr. Khurshid explained.
In the UK Biobank database, he said, “We saw the opportunity to leverage the largest sample of measured activity to date” to address the question of whether exercise time pattern “affects specific major cardiovascular diseases differently,” Dr. Khurshid said
The primary analysis assessed exercise amount in a week based on the guideline-recommended threshold of at least 2.5 hours; a 3-hour, 50-minutes threshold was used in a secondary analysis. The group assessed multiple thresholds because optimal MVPS levels derived from wrist-based accelerometers are “unclear,” he said.
The sample consisted of 89,573 participants with a mean age 62; slightly more than half (56%) were women. Based on the weekly MVPA threshold of 2.5 hours , the WW, active regular, and inactive groups made up 42.2%, 24%, and 33.7% of the population, respectively.
Compared with the inactive group, the two active groups both showed significant risk reductions for the four clinical outcomes, to similar degrees, in multivariate analysis. The results were similar at the 230-minute weekly exercise threshold for incident AF, MI, and HF but not for stroke.
The findings were similarly consistent at the 3-hour, 50-minutes median threshold, although stroke differences were no longer significant.
Patients should be encouraged to exercise at recommended levels, “and should not be discouraged if, for whatever reasons, they are able to focus exercise within only 1 or a few days of the week,” said Dr. Khurshid. “Our findings suggest that it is the volume of activity, rather than the pattern, that matters most.”
The report notes several limitations of the study, including the exercise observation period limited to 1 week and that participants could have modified their behavior during the observation period. Also, the participants were almost all White, so the results may not be generalizable to other populations.
Clinicians should familiarize themselves with the “full range of recommendations” presented in the “Physical Activity Guidelines for Americans, 2nd Edition” “and personalize prescriptions by setting achievable physical activity goals” based on age, physical abilities, and activity levels, states an accompanying editorial from Dr. Katzmarzyk and John M. Jakicic, PhD, University of Kansas Medical Center, Kansas City.
Although MVPA at the recommended level of at least 2.5 hours per week will certainly be beneficial, they write, “the public health message should also clearly convey that every minute counts, especially among the three-quarters of U.S. adults who do not achieve that goal.”
Dr. Khurshid reported no relevant financial relationships; disclosures for the other authors are in the original article. Dr. Katzmarzyk reports no relevant financial relationships. Dr. Jakicic discloses receiving personal fees from Wondr Health, WW International (formerly Weight Watchers), and Educational Initiatives and grants from Epitomee Medical.
A version of this article appeared on Medscape.com.
FROM JAMA
EU agency issues positive opinion on ritlecitinib
, paving the way for possible marketing authorization of the drug in the European Union for individuals 12 years of age and older. A final decision is expected in the coming months.
The development, which was announced by the manufacturer, Pfizer, on July 21, 2023, follows approval of ritlecitinib (Litfulo) for the treatment of severe alopecia areata in adults and adolescents 12 years and older by the Food and Drug Administration and the Japanese Ministry of Health, Labour, and Welfare in June 2023. According to a press release from Pfizer, submissions to other regulatory agencies for the use of ritlecitinib in alopecia areata are ongoing.
The Marketing Authorization Application for ritlecitinib was based on results from a randomized, placebo-controlled, double-blind ALLEGRO Phase 2b/3 study.
, paving the way for possible marketing authorization of the drug in the European Union for individuals 12 years of age and older. A final decision is expected in the coming months.
The development, which was announced by the manufacturer, Pfizer, on July 21, 2023, follows approval of ritlecitinib (Litfulo) for the treatment of severe alopecia areata in adults and adolescents 12 years and older by the Food and Drug Administration and the Japanese Ministry of Health, Labour, and Welfare in June 2023. According to a press release from Pfizer, submissions to other regulatory agencies for the use of ritlecitinib in alopecia areata are ongoing.
The Marketing Authorization Application for ritlecitinib was based on results from a randomized, placebo-controlled, double-blind ALLEGRO Phase 2b/3 study.
, paving the way for possible marketing authorization of the drug in the European Union for individuals 12 years of age and older. A final decision is expected in the coming months.
The development, which was announced by the manufacturer, Pfizer, on July 21, 2023, follows approval of ritlecitinib (Litfulo) for the treatment of severe alopecia areata in adults and adolescents 12 years and older by the Food and Drug Administration and the Japanese Ministry of Health, Labour, and Welfare in June 2023. According to a press release from Pfizer, submissions to other regulatory agencies for the use of ritlecitinib in alopecia areata are ongoing.
The Marketing Authorization Application for ritlecitinib was based on results from a randomized, placebo-controlled, double-blind ALLEGRO Phase 2b/3 study.
Affording the cost of new obesity drugs? We can’t afford not to
SAN DIEGO – Although the glucagonlike peptide–1 (GLP-1) receptor agonists, such as liraglutide and semaglutide, have been revolutionary advances for the treatment of obesity, the cost-effectiveness of these agents for treating both obesity and type 2 diabetes remains uncertain based on published analyses.
But potential future changes in the cost-effectiveness dynamics of GLP-1 agonists could tip the balance in their favor. These include
Costs to people with obesity that are generally not part of cost-effectiveness calculations include pain, disability, depression, and bias that affect employment, Carol H. Wysham, MD, said at the recent scientific sessions of the American Diabetes Association.
Other costs to society left out of conventional calculations are items such as the incremental cost for fuel to transport a heavier population and the carbon-footprint costs for the production and transportation of the excess food produced to feed an over-fed population, added Dr. Wysham, an endocrinologist with MultiCare and the Rockwood Clinic in Spokane, Wash.
Analyses should include ‘things we don’t often think about’
“The impact of living with obesity is much greater than what we traditionally calculate in health economics,” commented Naveed Sattar, PhD, speaking from the floor during the session.
“Patient happiness and self-esteem are hard to measure and capture as cost impacts. We need to also add carbon dioxide effects and transportation costs, and governments are starting to get wise to this. How to run proper health economics analyses is the key question; we need to do better than what we currently do,” said Dr. Sattar, a professor of metabolic medicine at the University of Glasgow.
Dr. Sattar is lead author of a recent analysis that highlights the overwhelming importance of improved weight management in adults as they age to reduce their risk of developing a broad range of chronic disorders.
“Most chronic conditions are, to differing extents, caused or exacerbated by excess adiposity,” was a conclusion of his report.
“It’s important to include the costs to society, including things we don’t often think about. No one has ever done a cost analysis that includes all the factors” cited by Dr. Wysham, said Irl B. Hirsch, MD, another speaker at the session. “No one includes obstructive sleep apnea, degenerative arthritis, and the downstream effects of a high body mass index.”
The GLP-1 agonists “are great” for both weight loss and glycemic control, said Dr. Hirsch, an endocrinologist and professor at the University of Washington, Seattle. “We can’t afford not to use them. These agents have been transformational.”
U.S. has the highest drug costs
Another key factor driving cost-effectiveness is, of course, the relatively high cost of the agents in the class, especially in the United States. Dr. Hirsch cited a recently published report in Obesity that quoted monthly U.S. costs of $804 for weekly 2.4-mg injections of semaglutide (Wegovy) and $1418 for daily 3.0-mg injections of liraglutide (Saxenda). Highlighting the relatively high cost of medications in the United States, the report cited a monthly price tag of $95 for the same semaglutide regimen in Turkey and a monthly cost of $252 for the same liraglutide regimen in Norway.
U.S. prices for agents in this class may start to deflate as soon as 2024, when one or more generic versions of liraglutide are expected, following expiration of the U.S. patent later in 2023, Dr. Wysham said.
Another pending trigger for lower costs may be the possible decision by the World Health Organization to designate liraglutide an “essential medicine” later in 2023, she noted. The WHO received an application for this designation from four U.S. clinicians and is considering it as part of its planned 2023 update to the WHO’s Essential Medicines List. Dr. Wysham predicted this designation would “press international pharmaceutical companies to produce [liraglutide] at a much lower cost.”
“I’m not saying that drug companies should not profit, but they should not do it on the backs of patients,” Dr. Wysham declared. “What do we measure by ‘cost-effectiveness?’ There are so many complications of obesity. For patients with diabetes and obesity we need to look for a little different economic policy.”
Dr. Wysham has reported being an adviser to Abbott and CeQur and receiving research funding from Eli Lilly and Novo Nordisk. Dr. Hirsch has reported being a consultant for Abbott, Embecta, and Hagar, and receiving research funding from Dexcom and Insulet. Dr. Sattar has reported receiving consulting fees or speaker honoraria from Abbott Laboratories, Afimmune, Amgen, AstraZeneca, Boehringer Ingelheim, Eli Lilly, Hanmi Pharmaceuticals, Janssen, MSD, Novartis, Novo Nordisk, Pfizer, Roche Diagnostics, and Sanofi.
A version of this article appeared on Medscape.com.
SAN DIEGO – Although the glucagonlike peptide–1 (GLP-1) receptor agonists, such as liraglutide and semaglutide, have been revolutionary advances for the treatment of obesity, the cost-effectiveness of these agents for treating both obesity and type 2 diabetes remains uncertain based on published analyses.
But potential future changes in the cost-effectiveness dynamics of GLP-1 agonists could tip the balance in their favor. These include
Costs to people with obesity that are generally not part of cost-effectiveness calculations include pain, disability, depression, and bias that affect employment, Carol H. Wysham, MD, said at the recent scientific sessions of the American Diabetes Association.
Other costs to society left out of conventional calculations are items such as the incremental cost for fuel to transport a heavier population and the carbon-footprint costs for the production and transportation of the excess food produced to feed an over-fed population, added Dr. Wysham, an endocrinologist with MultiCare and the Rockwood Clinic in Spokane, Wash.
Analyses should include ‘things we don’t often think about’
“The impact of living with obesity is much greater than what we traditionally calculate in health economics,” commented Naveed Sattar, PhD, speaking from the floor during the session.
“Patient happiness and self-esteem are hard to measure and capture as cost impacts. We need to also add carbon dioxide effects and transportation costs, and governments are starting to get wise to this. How to run proper health economics analyses is the key question; we need to do better than what we currently do,” said Dr. Sattar, a professor of metabolic medicine at the University of Glasgow.
Dr. Sattar is lead author of a recent analysis that highlights the overwhelming importance of improved weight management in adults as they age to reduce their risk of developing a broad range of chronic disorders.
“Most chronic conditions are, to differing extents, caused or exacerbated by excess adiposity,” was a conclusion of his report.
“It’s important to include the costs to society, including things we don’t often think about. No one has ever done a cost analysis that includes all the factors” cited by Dr. Wysham, said Irl B. Hirsch, MD, another speaker at the session. “No one includes obstructive sleep apnea, degenerative arthritis, and the downstream effects of a high body mass index.”
The GLP-1 agonists “are great” for both weight loss and glycemic control, said Dr. Hirsch, an endocrinologist and professor at the University of Washington, Seattle. “We can’t afford not to use them. These agents have been transformational.”
U.S. has the highest drug costs
Another key factor driving cost-effectiveness is, of course, the relatively high cost of the agents in the class, especially in the United States. Dr. Hirsch cited a recently published report in Obesity that quoted monthly U.S. costs of $804 for weekly 2.4-mg injections of semaglutide (Wegovy) and $1418 for daily 3.0-mg injections of liraglutide (Saxenda). Highlighting the relatively high cost of medications in the United States, the report cited a monthly price tag of $95 for the same semaglutide regimen in Turkey and a monthly cost of $252 for the same liraglutide regimen in Norway.
U.S. prices for agents in this class may start to deflate as soon as 2024, when one or more generic versions of liraglutide are expected, following expiration of the U.S. patent later in 2023, Dr. Wysham said.
Another pending trigger for lower costs may be the possible decision by the World Health Organization to designate liraglutide an “essential medicine” later in 2023, she noted. The WHO received an application for this designation from four U.S. clinicians and is considering it as part of its planned 2023 update to the WHO’s Essential Medicines List. Dr. Wysham predicted this designation would “press international pharmaceutical companies to produce [liraglutide] at a much lower cost.”
“I’m not saying that drug companies should not profit, but they should not do it on the backs of patients,” Dr. Wysham declared. “What do we measure by ‘cost-effectiveness?’ There are so many complications of obesity. For patients with diabetes and obesity we need to look for a little different economic policy.”
Dr. Wysham has reported being an adviser to Abbott and CeQur and receiving research funding from Eli Lilly and Novo Nordisk. Dr. Hirsch has reported being a consultant for Abbott, Embecta, and Hagar, and receiving research funding from Dexcom and Insulet. Dr. Sattar has reported receiving consulting fees or speaker honoraria from Abbott Laboratories, Afimmune, Amgen, AstraZeneca, Boehringer Ingelheim, Eli Lilly, Hanmi Pharmaceuticals, Janssen, MSD, Novartis, Novo Nordisk, Pfizer, Roche Diagnostics, and Sanofi.
A version of this article appeared on Medscape.com.
SAN DIEGO – Although the glucagonlike peptide–1 (GLP-1) receptor agonists, such as liraglutide and semaglutide, have been revolutionary advances for the treatment of obesity, the cost-effectiveness of these agents for treating both obesity and type 2 diabetes remains uncertain based on published analyses.
But potential future changes in the cost-effectiveness dynamics of GLP-1 agonists could tip the balance in their favor. These include
Costs to people with obesity that are generally not part of cost-effectiveness calculations include pain, disability, depression, and bias that affect employment, Carol H. Wysham, MD, said at the recent scientific sessions of the American Diabetes Association.
Other costs to society left out of conventional calculations are items such as the incremental cost for fuel to transport a heavier population and the carbon-footprint costs for the production and transportation of the excess food produced to feed an over-fed population, added Dr. Wysham, an endocrinologist with MultiCare and the Rockwood Clinic in Spokane, Wash.
Analyses should include ‘things we don’t often think about’
“The impact of living with obesity is much greater than what we traditionally calculate in health economics,” commented Naveed Sattar, PhD, speaking from the floor during the session.
“Patient happiness and self-esteem are hard to measure and capture as cost impacts. We need to also add carbon dioxide effects and transportation costs, and governments are starting to get wise to this. How to run proper health economics analyses is the key question; we need to do better than what we currently do,” said Dr. Sattar, a professor of metabolic medicine at the University of Glasgow.
Dr. Sattar is lead author of a recent analysis that highlights the overwhelming importance of improved weight management in adults as they age to reduce their risk of developing a broad range of chronic disorders.
“Most chronic conditions are, to differing extents, caused or exacerbated by excess adiposity,” was a conclusion of his report.
“It’s important to include the costs to society, including things we don’t often think about. No one has ever done a cost analysis that includes all the factors” cited by Dr. Wysham, said Irl B. Hirsch, MD, another speaker at the session. “No one includes obstructive sleep apnea, degenerative arthritis, and the downstream effects of a high body mass index.”
The GLP-1 agonists “are great” for both weight loss and glycemic control, said Dr. Hirsch, an endocrinologist and professor at the University of Washington, Seattle. “We can’t afford not to use them. These agents have been transformational.”
U.S. has the highest drug costs
Another key factor driving cost-effectiveness is, of course, the relatively high cost of the agents in the class, especially in the United States. Dr. Hirsch cited a recently published report in Obesity that quoted monthly U.S. costs of $804 for weekly 2.4-mg injections of semaglutide (Wegovy) and $1418 for daily 3.0-mg injections of liraglutide (Saxenda). Highlighting the relatively high cost of medications in the United States, the report cited a monthly price tag of $95 for the same semaglutide regimen in Turkey and a monthly cost of $252 for the same liraglutide regimen in Norway.
U.S. prices for agents in this class may start to deflate as soon as 2024, when one or more generic versions of liraglutide are expected, following expiration of the U.S. patent later in 2023, Dr. Wysham said.
Another pending trigger for lower costs may be the possible decision by the World Health Organization to designate liraglutide an “essential medicine” later in 2023, she noted. The WHO received an application for this designation from four U.S. clinicians and is considering it as part of its planned 2023 update to the WHO’s Essential Medicines List. Dr. Wysham predicted this designation would “press international pharmaceutical companies to produce [liraglutide] at a much lower cost.”
“I’m not saying that drug companies should not profit, but they should not do it on the backs of patients,” Dr. Wysham declared. “What do we measure by ‘cost-effectiveness?’ There are so many complications of obesity. For patients with diabetes and obesity we need to look for a little different economic policy.”
Dr. Wysham has reported being an adviser to Abbott and CeQur and receiving research funding from Eli Lilly and Novo Nordisk. Dr. Hirsch has reported being a consultant for Abbott, Embecta, and Hagar, and receiving research funding from Dexcom and Insulet. Dr. Sattar has reported receiving consulting fees or speaker honoraria from Abbott Laboratories, Afimmune, Amgen, AstraZeneca, Boehringer Ingelheim, Eli Lilly, Hanmi Pharmaceuticals, Janssen, MSD, Novartis, Novo Nordisk, Pfizer, Roche Diagnostics, and Sanofi.
A version of this article appeared on Medscape.com.
AT ADA 2023