User login
FUO, pneumonia often distinguishes influenza from RSV in hospitalized young children
LJUBLJANA, SLOVENIA – as the cause of hospitalization in infants and young children, Cihan Papan, MD, reported at the annual meeting of the European Society for Paediatric Infectious Diseases.
Dr. Papan, a pediatrician at University Children’s Hospital Mannheim (Germany) and Heidelberg (Germany) University, presented a retrospective single-center study of all 573 children aged under 2 years hospitalized over the course of several seasons for respiratory syncytial virus (RSV) or influenza as confirmed by rapid antigen testing. Even though these are two of the leading causes of hospitalization among young children, there is surprisingly sparse data comparing the two in terms of disease severity and hospital resource utilization, including antibiotic consumption. That information gap provided the basis for this study.
There were 476 children with confirmed RSV, 96 with influenza, and 1 RSV/influenza coinfection. Notably, even though the RSV group had lower temperatures and C-reactive protein levels, they were nevertheless more likely to be treated with antibiotics, by a margin of 29% to 23%.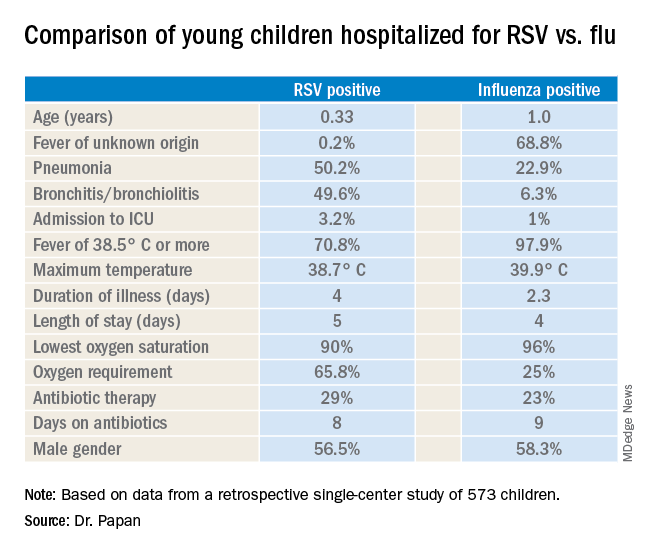
“These findings open new possibilities for antimicrobial stewardship in these groups of virally infected children,” observed Dr. Papan.
Fever of unknown origin was present in 68.8% of the influenza-positive patients, compared with just 0.2% of the RSV-positive children. In contrast, 50.2% of the RSV group had pneumonia and 49.6% had bronchitis or bronchiolitis, versus just 22.9% and 6.3% of the influenza patients, respectively. A larger proportion of the young children with RSV infection presented in a severely ill–looking condition. Children with RSV infection also were significantly younger.
Dr. Papan reported having no financial conflicts regarding his study.
LJUBLJANA, SLOVENIA – as the cause of hospitalization in infants and young children, Cihan Papan, MD, reported at the annual meeting of the European Society for Paediatric Infectious Diseases.
Dr. Papan, a pediatrician at University Children’s Hospital Mannheim (Germany) and Heidelberg (Germany) University, presented a retrospective single-center study of all 573 children aged under 2 years hospitalized over the course of several seasons for respiratory syncytial virus (RSV) or influenza as confirmed by rapid antigen testing. Even though these are two of the leading causes of hospitalization among young children, there is surprisingly sparse data comparing the two in terms of disease severity and hospital resource utilization, including antibiotic consumption. That information gap provided the basis for this study.
There were 476 children with confirmed RSV, 96 with influenza, and 1 RSV/influenza coinfection. Notably, even though the RSV group had lower temperatures and C-reactive protein levels, they were nevertheless more likely to be treated with antibiotics, by a margin of 29% to 23%.
“These findings open new possibilities for antimicrobial stewardship in these groups of virally infected children,” observed Dr. Papan.
Fever of unknown origin was present in 68.8% of the influenza-positive patients, compared with just 0.2% of the RSV-positive children. In contrast, 50.2% of the RSV group had pneumonia and 49.6% had bronchitis or bronchiolitis, versus just 22.9% and 6.3% of the influenza patients, respectively. A larger proportion of the young children with RSV infection presented in a severely ill–looking condition. Children with RSV infection also were significantly younger.
Dr. Papan reported having no financial conflicts regarding his study.
LJUBLJANA, SLOVENIA – as the cause of hospitalization in infants and young children, Cihan Papan, MD, reported at the annual meeting of the European Society for Paediatric Infectious Diseases.
Dr. Papan, a pediatrician at University Children’s Hospital Mannheim (Germany) and Heidelberg (Germany) University, presented a retrospective single-center study of all 573 children aged under 2 years hospitalized over the course of several seasons for respiratory syncytial virus (RSV) or influenza as confirmed by rapid antigen testing. Even though these are two of the leading causes of hospitalization among young children, there is surprisingly sparse data comparing the two in terms of disease severity and hospital resource utilization, including antibiotic consumption. That information gap provided the basis for this study.
There were 476 children with confirmed RSV, 96 with influenza, and 1 RSV/influenza coinfection. Notably, even though the RSV group had lower temperatures and C-reactive protein levels, they were nevertheless more likely to be treated with antibiotics, by a margin of 29% to 23%.
“These findings open new possibilities for antimicrobial stewardship in these groups of virally infected children,” observed Dr. Papan.
Fever of unknown origin was present in 68.8% of the influenza-positive patients, compared with just 0.2% of the RSV-positive children. In contrast, 50.2% of the RSV group had pneumonia and 49.6% had bronchitis or bronchiolitis, versus just 22.9% and 6.3% of the influenza patients, respectively. A larger proportion of the young children with RSV infection presented in a severely ill–looking condition. Children with RSV infection also were significantly younger.
Dr. Papan reported having no financial conflicts regarding his study.
REPORTING FROM ESPID 2019
No teen herd immunity for 4CMenB in landmark trial
LJUBLJANA, SLOVENIA – The 4CMenB vaccine didn’t affect carriage of disease-causing genogroups of Neisseria meningitidis in adolescents in the landmark Australian cluster-randomized trial of herd immunity known as the “B Part of It” study, Helen S. Marshall, MD, reported at the annual meeting of the European Society for Paediatric Infectious Diseases.
This was the largest-ever randomized trial of adolescents vaccinated against meningococcal disease, and the message, albeit somewhat disappointing, is clear: “MenB [Meningococcal serogroup B] vaccine programs should be designed to provide direct protection for those at highest risk of disease,” declared Dr. Marshall, professor of vaccinology and deputy director of the Robinson Research Institute at the University of Adelaide.
In other words, Youths in the age groups at highest risk of disease – infants and adolescents – need to routinely receive the vaccine.
The B Part of It study, whose sheer scope and rigor drew the attention of infectious disease clinical trialists the world over, randomized nearly 35,000 students at all high schools in the state of South Australia – whether urban, rural, or remote – to two doses of the 4CMenB vaccine known as Bexsero or to a nonvaccinated control group. This massive trial entailed training more than 250 nurses in the study procedures and involved 3,100 miles of travel to transport oropharyngeal swab samples obtained from students in outlying areas for centralized laboratory analysis using real-time polymerase chain reaction with meningococcal genotyping, culture for N. meningitidis, and whole-genome sequencing. Samples were obtained on day 1 of the study and 12 months later.
The investigators created widespread regional enthusiasm for this project through adept use of social media and other methods. As a result, 99.5% of students randomized to the intervention arm received one dose, while 97% got two doses. A gratifying unintended consequence of the study was that parents who’d never previously vaccinated their children enrolled them in B Part of It, Dr. Marshall noted.
The impetus for B Part of It was that, while the Australian national health insurance program covers a single dose of meningococcal conjugate MenACWY vaccine given at age 12 months and 14-19 years, MenB vaccine isn’t covered because of uncertainties about cost effectiveness and the vaccine’s impact on meningococcal carriage and herd immunity. B Part of It was designed to resolve those uncertainties.
South Australia has the highest rate of invasive meningococcal disease in the country, and more than 80% of cases there are caused by meningococcal serogroup B. Moreover, 75% of group B cases in South Australia involve the nasty hypervirulent New Zealand strain known as CC 41/44.
The primary outcome in B Part of It was the difference in carriage of the major disease-causing serotypes – groups A, B, C, W, X, and Y – between vaccinated and unvaccinated students at the 1-year follow-up mark. The carriage prevalence of all N. meningitidis in the vaccinated students went from 2.8% at baseline to 4.0% at 12 months, and similarly from 2.6% to 4.7% in unvaccinated controls. More importantly, the prevalence of disease-causing genotypes rose from 1.3% at baseline to 2.4% at follow-up in the vaccinated subjects, with a near-identical pattern seen in controls, where the prevalence rose from 1.4% to 2.4%. In an as-treated analysis, the rate of acquisition of carriage of disease-causing genotypes was identical at 2.0% in both study arms.
The 4CMenB vaccine proved reassuringly safe and effective in preventing meningococcal disease in vaccinated teens. With more than 58,000 doses of the vaccine given in the study, no new safety concerns or signals emerged. And the observed number of cases of invasive meningococcal disease in South Australian adolescent vaccine recipients to date has been significantly lower than expected.
Secondary and exploratory outcomes
Independent risk factors associated with N. meningitidis carriage in the study participants at the 1-year mark included smoking cigarettes or hookah, intimate kissing within the last week, and being in grades 11-12, as opposed to grade 10.
The vaccine had no significant impact on the carriage rate of the hypervirulent New Zealand serogroup B strain. Nor was there a vaccine impact on carriage density, as Mark McMillan, MD, reported elsewhere at ESPID 2019. But while the 4CMenB vaccine had minimal impact upon N. meningitidis carriage density, it was associated with a significant 41% increase in the likelihood of cleared carriage of disease-causing strains at 12 months, added Dr. McMillan, Dr. Marshall’s coinvestigator at University of Adelaide.
What’s next
The ongoing B Part of It School Leaver study is assessing carriage prevalence in vaccinated versus unvaccinated high schoolers in their first year after graduating.
In addition, the B Part of It investigators plan to prospectively study the impact of the 4CMen B vaccine on N. gonorrhoeae disease in an effort to confirm the intriguing findings of an earlier large, retrospective New Zealand case-control study. The Kiwis found that recipients of an outer membrane vesicle MenB vaccine had an adjusted 31% reduction in the risk of gonorrhea. This was the first-ever report of any vaccine effectiveness against this major global public health problem, in which antibiotic resistance is a growing concern (Lancet. 2017 Sep 30;390[10102]:1603-10). Dr. Marshall reported receiving research funding from GlaxoSmithKline, which markets Bexsero and was the major financial supporter of the B Part of It study.
But wait a minute...
Following Dr. Marshall’s report on the B Part of It study, outgoing ESPID president Adam Finn, MD, PhD, presented longitudinal data that he believes raise the possibility that protein-antigen vaccines such as Bexsero, which promote naturally acquired mucosal immunity, may impact on transmission population wide without reliably preventing acquisition. This would stand in stark contrast to conjugate meningococcus vaccines, which have a well-established massive impact on carriage and acquisition of N. meningitidis.
It may be that in studying throat carriage rates once in individuals immunized 12 months earlier, as in the B Part of It study, investigators are not asking the right question, proposed Dr. Finn, professor of pediatrics at the University of Bristol (England).
His research team has been obtaining throat swabs at monthly intervals in a population of 917 high schoolers aged 16-17 years. In 416 of the students, they also have collected saliva samples weekly both before and after immunization with 4CMenB vaccine, analyzing the samples for N. meningitidis by polymerase chain reaction. This is a novel method of studying meningococcal carriage they have found to be both reliable and far more acceptable to patients than oropharyngeal swabbing, which adolescents balk at if asked to do with any frequency (PLoS One. 2019 Feb 11;14[2]:e0209905).
Dr. Finn said that their findings, which need confirmation, suggest that N. meningitidis carriage is usually brief and dynamic. They also have found that carriage density varies markedly from month to month.
“We see much higher-density carriage in the adolescent population in the early months of the year in conjunction, we think, with viral infection with influenza and so forth,” he said, adding that this could have clinical implications. “It feels sort of intuitive that someone walking around with 1,000 or 10,000 times as many meningococci in their throat is more likely to be more infectious to people around them with a very small number, although this hasn’t been formally proven.”
He hopes that the Be on the TEAM (Teenagers Against Meningitis) study will help provide answers. The study is randomizing 24,000 U.K. high school students to vaccination with the meningococcal B protein–antigen vaccines Bexsero or Trumenba or to no vaccine in order to learn if there are significant herd immunity effects.
Dr. Finn’s meningococcal carriage research is funded by the Meningitis Research Foundation and the National Institute for Health Research. Dr. Marshall reported receiving research funding from GlaxoSmithKline, the major sponsor of the B Part of It study.
LJUBLJANA, SLOVENIA – The 4CMenB vaccine didn’t affect carriage of disease-causing genogroups of Neisseria meningitidis in adolescents in the landmark Australian cluster-randomized trial of herd immunity known as the “B Part of It” study, Helen S. Marshall, MD, reported at the annual meeting of the European Society for Paediatric Infectious Diseases.
This was the largest-ever randomized trial of adolescents vaccinated against meningococcal disease, and the message, albeit somewhat disappointing, is clear: “MenB [Meningococcal serogroup B] vaccine programs should be designed to provide direct protection for those at highest risk of disease,” declared Dr. Marshall, professor of vaccinology and deputy director of the Robinson Research Institute at the University of Adelaide.
In other words, Youths in the age groups at highest risk of disease – infants and adolescents – need to routinely receive the vaccine.
The B Part of It study, whose sheer scope and rigor drew the attention of infectious disease clinical trialists the world over, randomized nearly 35,000 students at all high schools in the state of South Australia – whether urban, rural, or remote – to two doses of the 4CMenB vaccine known as Bexsero or to a nonvaccinated control group. This massive trial entailed training more than 250 nurses in the study procedures and involved 3,100 miles of travel to transport oropharyngeal swab samples obtained from students in outlying areas for centralized laboratory analysis using real-time polymerase chain reaction with meningococcal genotyping, culture for N. meningitidis, and whole-genome sequencing. Samples were obtained on day 1 of the study and 12 months later.
The investigators created widespread regional enthusiasm for this project through adept use of social media and other methods. As a result, 99.5% of students randomized to the intervention arm received one dose, while 97% got two doses. A gratifying unintended consequence of the study was that parents who’d never previously vaccinated their children enrolled them in B Part of It, Dr. Marshall noted.
The impetus for B Part of It was that, while the Australian national health insurance program covers a single dose of meningococcal conjugate MenACWY vaccine given at age 12 months and 14-19 years, MenB vaccine isn’t covered because of uncertainties about cost effectiveness and the vaccine’s impact on meningococcal carriage and herd immunity. B Part of It was designed to resolve those uncertainties.
South Australia has the highest rate of invasive meningococcal disease in the country, and more than 80% of cases there are caused by meningococcal serogroup B. Moreover, 75% of group B cases in South Australia involve the nasty hypervirulent New Zealand strain known as CC 41/44.
The primary outcome in B Part of It was the difference in carriage of the major disease-causing serotypes – groups A, B, C, W, X, and Y – between vaccinated and unvaccinated students at the 1-year follow-up mark. The carriage prevalence of all N. meningitidis in the vaccinated students went from 2.8% at baseline to 4.0% at 12 months, and similarly from 2.6% to 4.7% in unvaccinated controls. More importantly, the prevalence of disease-causing genotypes rose from 1.3% at baseline to 2.4% at follow-up in the vaccinated subjects, with a near-identical pattern seen in controls, where the prevalence rose from 1.4% to 2.4%. In an as-treated analysis, the rate of acquisition of carriage of disease-causing genotypes was identical at 2.0% in both study arms.
The 4CMenB vaccine proved reassuringly safe and effective in preventing meningococcal disease in vaccinated teens. With more than 58,000 doses of the vaccine given in the study, no new safety concerns or signals emerged. And the observed number of cases of invasive meningococcal disease in South Australian adolescent vaccine recipients to date has been significantly lower than expected.
Secondary and exploratory outcomes
Independent risk factors associated with N. meningitidis carriage in the study participants at the 1-year mark included smoking cigarettes or hookah, intimate kissing within the last week, and being in grades 11-12, as opposed to grade 10.
The vaccine had no significant impact on the carriage rate of the hypervirulent New Zealand serogroup B strain. Nor was there a vaccine impact on carriage density, as Mark McMillan, MD, reported elsewhere at ESPID 2019. But while the 4CMenB vaccine had minimal impact upon N. meningitidis carriage density, it was associated with a significant 41% increase in the likelihood of cleared carriage of disease-causing strains at 12 months, added Dr. McMillan, Dr. Marshall’s coinvestigator at University of Adelaide.
What’s next
The ongoing B Part of It School Leaver study is assessing carriage prevalence in vaccinated versus unvaccinated high schoolers in their first year after graduating.
In addition, the B Part of It investigators plan to prospectively study the impact of the 4CMen B vaccine on N. gonorrhoeae disease in an effort to confirm the intriguing findings of an earlier large, retrospective New Zealand case-control study. The Kiwis found that recipients of an outer membrane vesicle MenB vaccine had an adjusted 31% reduction in the risk of gonorrhea. This was the first-ever report of any vaccine effectiveness against this major global public health problem, in which antibiotic resistance is a growing concern (Lancet. 2017 Sep 30;390[10102]:1603-10). Dr. Marshall reported receiving research funding from GlaxoSmithKline, which markets Bexsero and was the major financial supporter of the B Part of It study.
But wait a minute...
Following Dr. Marshall’s report on the B Part of It study, outgoing ESPID president Adam Finn, MD, PhD, presented longitudinal data that he believes raise the possibility that protein-antigen vaccines such as Bexsero, which promote naturally acquired mucosal immunity, may impact on transmission population wide without reliably preventing acquisition. This would stand in stark contrast to conjugate meningococcus vaccines, which have a well-established massive impact on carriage and acquisition of N. meningitidis.
It may be that in studying throat carriage rates once in individuals immunized 12 months earlier, as in the B Part of It study, investigators are not asking the right question, proposed Dr. Finn, professor of pediatrics at the University of Bristol (England).
His research team has been obtaining throat swabs at monthly intervals in a population of 917 high schoolers aged 16-17 years. In 416 of the students, they also have collected saliva samples weekly both before and after immunization with 4CMenB vaccine, analyzing the samples for N. meningitidis by polymerase chain reaction. This is a novel method of studying meningococcal carriage they have found to be both reliable and far more acceptable to patients than oropharyngeal swabbing, which adolescents balk at if asked to do with any frequency (PLoS One. 2019 Feb 11;14[2]:e0209905).
Dr. Finn said that their findings, which need confirmation, suggest that N. meningitidis carriage is usually brief and dynamic. They also have found that carriage density varies markedly from month to month.
“We see much higher-density carriage in the adolescent population in the early months of the year in conjunction, we think, with viral infection with influenza and so forth,” he said, adding that this could have clinical implications. “It feels sort of intuitive that someone walking around with 1,000 or 10,000 times as many meningococci in their throat is more likely to be more infectious to people around them with a very small number, although this hasn’t been formally proven.”
He hopes that the Be on the TEAM (Teenagers Against Meningitis) study will help provide answers. The study is randomizing 24,000 U.K. high school students to vaccination with the meningococcal B protein–antigen vaccines Bexsero or Trumenba or to no vaccine in order to learn if there are significant herd immunity effects.
Dr. Finn’s meningococcal carriage research is funded by the Meningitis Research Foundation and the National Institute for Health Research. Dr. Marshall reported receiving research funding from GlaxoSmithKline, the major sponsor of the B Part of It study.
LJUBLJANA, SLOVENIA – The 4CMenB vaccine didn’t affect carriage of disease-causing genogroups of Neisseria meningitidis in adolescents in the landmark Australian cluster-randomized trial of herd immunity known as the “B Part of It” study, Helen S. Marshall, MD, reported at the annual meeting of the European Society for Paediatric Infectious Diseases.
This was the largest-ever randomized trial of adolescents vaccinated against meningococcal disease, and the message, albeit somewhat disappointing, is clear: “MenB [Meningococcal serogroup B] vaccine programs should be designed to provide direct protection for those at highest risk of disease,” declared Dr. Marshall, professor of vaccinology and deputy director of the Robinson Research Institute at the University of Adelaide.
In other words, Youths in the age groups at highest risk of disease – infants and adolescents – need to routinely receive the vaccine.
The B Part of It study, whose sheer scope and rigor drew the attention of infectious disease clinical trialists the world over, randomized nearly 35,000 students at all high schools in the state of South Australia – whether urban, rural, or remote – to two doses of the 4CMenB vaccine known as Bexsero or to a nonvaccinated control group. This massive trial entailed training more than 250 nurses in the study procedures and involved 3,100 miles of travel to transport oropharyngeal swab samples obtained from students in outlying areas for centralized laboratory analysis using real-time polymerase chain reaction with meningococcal genotyping, culture for N. meningitidis, and whole-genome sequencing. Samples were obtained on day 1 of the study and 12 months later.
The investigators created widespread regional enthusiasm for this project through adept use of social media and other methods. As a result, 99.5% of students randomized to the intervention arm received one dose, while 97% got two doses. A gratifying unintended consequence of the study was that parents who’d never previously vaccinated their children enrolled them in B Part of It, Dr. Marshall noted.
The impetus for B Part of It was that, while the Australian national health insurance program covers a single dose of meningococcal conjugate MenACWY vaccine given at age 12 months and 14-19 years, MenB vaccine isn’t covered because of uncertainties about cost effectiveness and the vaccine’s impact on meningococcal carriage and herd immunity. B Part of It was designed to resolve those uncertainties.
South Australia has the highest rate of invasive meningococcal disease in the country, and more than 80% of cases there are caused by meningococcal serogroup B. Moreover, 75% of group B cases in South Australia involve the nasty hypervirulent New Zealand strain known as CC 41/44.
The primary outcome in B Part of It was the difference in carriage of the major disease-causing serotypes – groups A, B, C, W, X, and Y – between vaccinated and unvaccinated students at the 1-year follow-up mark. The carriage prevalence of all N. meningitidis in the vaccinated students went from 2.8% at baseline to 4.0% at 12 months, and similarly from 2.6% to 4.7% in unvaccinated controls. More importantly, the prevalence of disease-causing genotypes rose from 1.3% at baseline to 2.4% at follow-up in the vaccinated subjects, with a near-identical pattern seen in controls, where the prevalence rose from 1.4% to 2.4%. In an as-treated analysis, the rate of acquisition of carriage of disease-causing genotypes was identical at 2.0% in both study arms.
The 4CMenB vaccine proved reassuringly safe and effective in preventing meningococcal disease in vaccinated teens. With more than 58,000 doses of the vaccine given in the study, no new safety concerns or signals emerged. And the observed number of cases of invasive meningococcal disease in South Australian adolescent vaccine recipients to date has been significantly lower than expected.
Secondary and exploratory outcomes
Independent risk factors associated with N. meningitidis carriage in the study participants at the 1-year mark included smoking cigarettes or hookah, intimate kissing within the last week, and being in grades 11-12, as opposed to grade 10.
The vaccine had no significant impact on the carriage rate of the hypervirulent New Zealand serogroup B strain. Nor was there a vaccine impact on carriage density, as Mark McMillan, MD, reported elsewhere at ESPID 2019. But while the 4CMenB vaccine had minimal impact upon N. meningitidis carriage density, it was associated with a significant 41% increase in the likelihood of cleared carriage of disease-causing strains at 12 months, added Dr. McMillan, Dr. Marshall’s coinvestigator at University of Adelaide.
What’s next
The ongoing B Part of It School Leaver study is assessing carriage prevalence in vaccinated versus unvaccinated high schoolers in their first year after graduating.
In addition, the B Part of It investigators plan to prospectively study the impact of the 4CMen B vaccine on N. gonorrhoeae disease in an effort to confirm the intriguing findings of an earlier large, retrospective New Zealand case-control study. The Kiwis found that recipients of an outer membrane vesicle MenB vaccine had an adjusted 31% reduction in the risk of gonorrhea. This was the first-ever report of any vaccine effectiveness against this major global public health problem, in which antibiotic resistance is a growing concern (Lancet. 2017 Sep 30;390[10102]:1603-10). Dr. Marshall reported receiving research funding from GlaxoSmithKline, which markets Bexsero and was the major financial supporter of the B Part of It study.
But wait a minute...
Following Dr. Marshall’s report on the B Part of It study, outgoing ESPID president Adam Finn, MD, PhD, presented longitudinal data that he believes raise the possibility that protein-antigen vaccines such as Bexsero, which promote naturally acquired mucosal immunity, may impact on transmission population wide without reliably preventing acquisition. This would stand in stark contrast to conjugate meningococcus vaccines, which have a well-established massive impact on carriage and acquisition of N. meningitidis.
It may be that in studying throat carriage rates once in individuals immunized 12 months earlier, as in the B Part of It study, investigators are not asking the right question, proposed Dr. Finn, professor of pediatrics at the University of Bristol (England).
His research team has been obtaining throat swabs at monthly intervals in a population of 917 high schoolers aged 16-17 years. In 416 of the students, they also have collected saliva samples weekly both before and after immunization with 4CMenB vaccine, analyzing the samples for N. meningitidis by polymerase chain reaction. This is a novel method of studying meningococcal carriage they have found to be both reliable and far more acceptable to patients than oropharyngeal swabbing, which adolescents balk at if asked to do with any frequency (PLoS One. 2019 Feb 11;14[2]:e0209905).
Dr. Finn said that their findings, which need confirmation, suggest that N. meningitidis carriage is usually brief and dynamic. They also have found that carriage density varies markedly from month to month.
“We see much higher-density carriage in the adolescent population in the early months of the year in conjunction, we think, with viral infection with influenza and so forth,” he said, adding that this could have clinical implications. “It feels sort of intuitive that someone walking around with 1,000 or 10,000 times as many meningococci in their throat is more likely to be more infectious to people around them with a very small number, although this hasn’t been formally proven.”
He hopes that the Be on the TEAM (Teenagers Against Meningitis) study will help provide answers. The study is randomizing 24,000 U.K. high school students to vaccination with the meningococcal B protein–antigen vaccines Bexsero or Trumenba or to no vaccine in order to learn if there are significant herd immunity effects.
Dr. Finn’s meningococcal carriage research is funded by the Meningitis Research Foundation and the National Institute for Health Research. Dr. Marshall reported receiving research funding from GlaxoSmithKline, the major sponsor of the B Part of It study.
REPORTING FROM ESPID 2019
Differential monocytic HLA-DR expression prognostically useful in PICU
LJUBLJANA, SLOVENIA – During their first 4 days in the pediatric ICU, critically ill children have significantly reduced human leukocyte antigen (HLA)–DR expression within all three major subsets of monocytes. The reductions are seen regardless of whether the children were admitted for sepsis, trauma, or after surgery, Navin Boeddha, MD, PhD, reported in his PIDJ Award Lecture at the annual meeting of the European Society for Paediatric Infectious Diseases.
The PIDJ Award is given annually by the editors of the Pediatric Infectious Disease Journal in recognition of what they deem the most important study published in the journal during the prior year. This one stood out because it identified promising potential laboratory markers that have been sought as a prerequisite to developing immunostimulatory therapies aimed at improving outcomes in severely immunosuppressed children.
Researchers are particularly eager to explore this investigative treatment strategy because the mortality and long-term morbidity of pediatric sepsis, in particular, remain unacceptably high. The hope now is that HLA-DR expression on monocyte subsets will be helpful in directing granulocyte-macrophage colony-stimulating factor, interferon-gamma, and other immunostimulatory therapies to the pediatric ICU patients with the most favorable benefit/risk ratio, according to Dr. Boeddha of Sophia Children’s Hospital and Erasmus University, Rotterdam, the Netherlands.
He reported on 37 critically ill children admitted to a pediatric ICU – 12 for sepsis, 11 post surgery, 10 for trauma, and 4 for other reasons – as well as 37 healthy controls. HLA-DR expression on monocyte subsets was measured by flow cytometry upon admission and again on each of the following 3 days.
The impetus for this study is that severe infection, major surgery, and severe trauma are often associated with immunosuppression. And while prior work in septic adults has concluded that decreased monocytic HLA-DR expression is a marker for immunosuppression – and that the lower the level of such expression, the greater the risk of nosocomial infection and death – this phenomenon hasn’t been well studied in critically ill children, he explained.
Dr. Boeddha and coinvestigators found that monocytic HLA-DR expression, which plays a major role in presenting antigens to T cells, decreased over time during the critically ill children’s first 4 days in the pediatric ICU. Moreover, it was lower than in controls at all four time points. This was true both for the percentage of HLA-DR–expressing monocytes of all subsets, as well as for HLA-DR mean fluorescence intensity.
In the critically ill study population as a whole, the percentage of classical monocytes – that is, CD14++ CD16– monocytes – was significantly greater at admission than in healthy controls by margins of 95% and 87%, while the percentage of nonclassical CD14+/-CD16++ monocytes was markedly lower at 2% than the 9% figure in controls.
The biggest discrepancy in monocyte subset distribution was seen in patients admitted for sepsis. Their percentage of classical monocytes was lower than in controls by a margin of 82% versus 87%; however, their proportion of intermediate monocytes (CD14++ CD16+) upon admission was twice that of controls, and it climbed further to 14% on day 2.
Among the key findings in the Rotterdam study: 13 of 37 critically ill patients experienced at least one nosocomial infection while in the pediatric ICU. Their day 2 percentage of HLA-DR–expressing classical monocytes was 42%, strikingly lower than the 78% figure in patients who didn’t develop an infection. Also, the 6 patients who died had only a 33% rate of HLA-DR–expressing classical monocytes on day 3 after pediatric ICU admission versus a 63% rate in survivors of their critical illness.
Thus, low HLA-DR expression on classical monocytes early during the course of a pediatric ICU stay may be the sought-after biomarker that identifies a particularly high-risk subgroup of critically ill children in whom immunostimulatory therapies should be studied. However, future confirmatory studies should monitor monocytic HLA-DR expression in a larger critically ill patient population for a longer period in order to establish the time to recovery of low expression and its impact on long-term complications, the physician said.
Dr. Boeddha reported having no financial conflicts regarding the award-winning study, supported by the European Union and Erasmus University.
SOURCE: Boeddha NP et al. Pediatr Infect Dis J. 2018 Oct;37(10):1034-40.
LJUBLJANA, SLOVENIA – During their first 4 days in the pediatric ICU, critically ill children have significantly reduced human leukocyte antigen (HLA)–DR expression within all three major subsets of monocytes. The reductions are seen regardless of whether the children were admitted for sepsis, trauma, or after surgery, Navin Boeddha, MD, PhD, reported in his PIDJ Award Lecture at the annual meeting of the European Society for Paediatric Infectious Diseases.
The PIDJ Award is given annually by the editors of the Pediatric Infectious Disease Journal in recognition of what they deem the most important study published in the journal during the prior year. This one stood out because it identified promising potential laboratory markers that have been sought as a prerequisite to developing immunostimulatory therapies aimed at improving outcomes in severely immunosuppressed children.
Researchers are particularly eager to explore this investigative treatment strategy because the mortality and long-term morbidity of pediatric sepsis, in particular, remain unacceptably high. The hope now is that HLA-DR expression on monocyte subsets will be helpful in directing granulocyte-macrophage colony-stimulating factor, interferon-gamma, and other immunostimulatory therapies to the pediatric ICU patients with the most favorable benefit/risk ratio, according to Dr. Boeddha of Sophia Children’s Hospital and Erasmus University, Rotterdam, the Netherlands.
He reported on 37 critically ill children admitted to a pediatric ICU – 12 for sepsis, 11 post surgery, 10 for trauma, and 4 for other reasons – as well as 37 healthy controls. HLA-DR expression on monocyte subsets was measured by flow cytometry upon admission and again on each of the following 3 days.
The impetus for this study is that severe infection, major surgery, and severe trauma are often associated with immunosuppression. And while prior work in septic adults has concluded that decreased monocytic HLA-DR expression is a marker for immunosuppression – and that the lower the level of such expression, the greater the risk of nosocomial infection and death – this phenomenon hasn’t been well studied in critically ill children, he explained.
Dr. Boeddha and coinvestigators found that monocytic HLA-DR expression, which plays a major role in presenting antigens to T cells, decreased over time during the critically ill children’s first 4 days in the pediatric ICU. Moreover, it was lower than in controls at all four time points. This was true both for the percentage of HLA-DR–expressing monocytes of all subsets, as well as for HLA-DR mean fluorescence intensity.
In the critically ill study population as a whole, the percentage of classical monocytes – that is, CD14++ CD16– monocytes – was significantly greater at admission than in healthy controls by margins of 95% and 87%, while the percentage of nonclassical CD14+/-CD16++ monocytes was markedly lower at 2% than the 9% figure in controls.
The biggest discrepancy in monocyte subset distribution was seen in patients admitted for sepsis. Their percentage of classical monocytes was lower than in controls by a margin of 82% versus 87%; however, their proportion of intermediate monocytes (CD14++ CD16+) upon admission was twice that of controls, and it climbed further to 14% on day 2.
Among the key findings in the Rotterdam study: 13 of 37 critically ill patients experienced at least one nosocomial infection while in the pediatric ICU. Their day 2 percentage of HLA-DR–expressing classical monocytes was 42%, strikingly lower than the 78% figure in patients who didn’t develop an infection. Also, the 6 patients who died had only a 33% rate of HLA-DR–expressing classical monocytes on day 3 after pediatric ICU admission versus a 63% rate in survivors of their critical illness.
Thus, low HLA-DR expression on classical monocytes early during the course of a pediatric ICU stay may be the sought-after biomarker that identifies a particularly high-risk subgroup of critically ill children in whom immunostimulatory therapies should be studied. However, future confirmatory studies should monitor monocytic HLA-DR expression in a larger critically ill patient population for a longer period in order to establish the time to recovery of low expression and its impact on long-term complications, the physician said.
Dr. Boeddha reported having no financial conflicts regarding the award-winning study, supported by the European Union and Erasmus University.
SOURCE: Boeddha NP et al. Pediatr Infect Dis J. 2018 Oct;37(10):1034-40.
LJUBLJANA, SLOVENIA – During their first 4 days in the pediatric ICU, critically ill children have significantly reduced human leukocyte antigen (HLA)–DR expression within all three major subsets of monocytes. The reductions are seen regardless of whether the children were admitted for sepsis, trauma, or after surgery, Navin Boeddha, MD, PhD, reported in his PIDJ Award Lecture at the annual meeting of the European Society for Paediatric Infectious Diseases.
The PIDJ Award is given annually by the editors of the Pediatric Infectious Disease Journal in recognition of what they deem the most important study published in the journal during the prior year. This one stood out because it identified promising potential laboratory markers that have been sought as a prerequisite to developing immunostimulatory therapies aimed at improving outcomes in severely immunosuppressed children.
Researchers are particularly eager to explore this investigative treatment strategy because the mortality and long-term morbidity of pediatric sepsis, in particular, remain unacceptably high. The hope now is that HLA-DR expression on monocyte subsets will be helpful in directing granulocyte-macrophage colony-stimulating factor, interferon-gamma, and other immunostimulatory therapies to the pediatric ICU patients with the most favorable benefit/risk ratio, according to Dr. Boeddha of Sophia Children’s Hospital and Erasmus University, Rotterdam, the Netherlands.
He reported on 37 critically ill children admitted to a pediatric ICU – 12 for sepsis, 11 post surgery, 10 for trauma, and 4 for other reasons – as well as 37 healthy controls. HLA-DR expression on monocyte subsets was measured by flow cytometry upon admission and again on each of the following 3 days.
The impetus for this study is that severe infection, major surgery, and severe trauma are often associated with immunosuppression. And while prior work in septic adults has concluded that decreased monocytic HLA-DR expression is a marker for immunosuppression – and that the lower the level of such expression, the greater the risk of nosocomial infection and death – this phenomenon hasn’t been well studied in critically ill children, he explained.
Dr. Boeddha and coinvestigators found that monocytic HLA-DR expression, which plays a major role in presenting antigens to T cells, decreased over time during the critically ill children’s first 4 days in the pediatric ICU. Moreover, it was lower than in controls at all four time points. This was true both for the percentage of HLA-DR–expressing monocytes of all subsets, as well as for HLA-DR mean fluorescence intensity.
In the critically ill study population as a whole, the percentage of classical monocytes – that is, CD14++ CD16– monocytes – was significantly greater at admission than in healthy controls by margins of 95% and 87%, while the percentage of nonclassical CD14+/-CD16++ monocytes was markedly lower at 2% than the 9% figure in controls.
The biggest discrepancy in monocyte subset distribution was seen in patients admitted for sepsis. Their percentage of classical monocytes was lower than in controls by a margin of 82% versus 87%; however, their proportion of intermediate monocytes (CD14++ CD16+) upon admission was twice that of controls, and it climbed further to 14% on day 2.
Among the key findings in the Rotterdam study: 13 of 37 critically ill patients experienced at least one nosocomial infection while in the pediatric ICU. Their day 2 percentage of HLA-DR–expressing classical monocytes was 42%, strikingly lower than the 78% figure in patients who didn’t develop an infection. Also, the 6 patients who died had only a 33% rate of HLA-DR–expressing classical monocytes on day 3 after pediatric ICU admission versus a 63% rate in survivors of their critical illness.
Thus, low HLA-DR expression on classical monocytes early during the course of a pediatric ICU stay may be the sought-after biomarker that identifies a particularly high-risk subgroup of critically ill children in whom immunostimulatory therapies should be studied. However, future confirmatory studies should monitor monocytic HLA-DR expression in a larger critically ill patient population for a longer period in order to establish the time to recovery of low expression and its impact on long-term complications, the physician said.
Dr. Boeddha reported having no financial conflicts regarding the award-winning study, supported by the European Union and Erasmus University.
SOURCE: Boeddha NP et al. Pediatr Infect Dis J. 2018 Oct;37(10):1034-40.
REPORTING FROM ESPID 2019
Presepsin can rule out invasive bacterial infection in infants
LJUBLJANA, SLOVENIA – A point-of-care presepsin measurement in the emergency department displayed powerful accuracy for early rule-out of invasive bacterial infection in infants less than 3 months old presenting with fever without a source, based on results of a phase 3 multicenter Italian study.
“P-SEP [presepsin] is a promising new biomarker. P-SEP accuracy for invasive bacterial infection is comparable to procalcitonin, even though P-SEP, like procalcitonin, is probably not accurate enough to be used as a stand-alone marker to rule-in an invasive bacterial infection,” Luca Pierantoni, MD, said in presenting the preliminary study results at the annual meeting of the European Society for Paediatric Infectious Diseases.
The presepsin test is a rapid point-of-care test well-suited for the ED setting, with a cost equal to that of point-of-care procalcitonin.
Presepsin is a form of soluble CD14 that is released from the surface of macrophages, monocytes, and neutrophils when these immune cells are stimulated by pathogens. “We think it may be a reliable diagnostic and prognostic marker of sepsis in adults and neonates,” explained Dr. Pierantoni of the University of Bologna, Italy.
Indeed, studies in adults suggest presepsin has better sensitivity and specificity than other biomarkers for early diagnosis of sepsis, and that it provides useful information on severity and prognosis as well. But, Dr. Pierantoni and his coworkers wondered, how does it perform in febrile young infants?
The Italian study was designed to address an unmet need: Fever accounts for about one-third of ED visits in infants up to age 3 months, 20% of whom are initially categorized as having fever without source. Yet ultimately 10%-20% of those youngsters having fever without source are found to have an invasive bacterial infection – that is, sepsis or meningitis – or a severe bacterial infection such as pneumonia, a urinary tract infection, or an infected umbilical cord. The sooner these infants can be identified and appropriately treated, the better.
The study enrolled 284 children less than 3 months old who had fever without cause of a mean 10.5 hours duration and presented to the emergency departments of six Italian medical centers. Children were eligible for the study regardless of whether they appeared toxic or well. Presepsin, procalcitonin, and C-reactive protein levels were immediately measured in all participants. Ultimately, 5.6% of subjects were diagnosed with an invasive bacterial infection, and another 21.2% had a severe bacterial infection.
Using a cutoff value of 449 pg/mL, P-SEP had good diagnostic accuracy for invasive bacterial infection, with an area under the receiver operating characteristics curve of 0.81, essentially the same as the 0.82 value for procalcitonin. P-SEP had a sensitivity and specificity of 87% and 75%, respectively, placing it in the same ballpark as the 82% and 86% values for procalcitonin. The strong point for P-SEP was its 99% negative predictive value, as compared to 91% for procalcitonin. The positive predictive values were 17% for P-SEP and 20% for procalcitonin.
In response to an audience question, Dr. Pierantoni speculated that the best use for P-SEP in the setting of fever of unknown origin may be in combination with procalcitonin rather than as a replacement for it. The research team is now in the process of analyzing their study data to see if that is indeed the case.
He reported having no financial conflicts regarding his study, conducted free of commercial support.
LJUBLJANA, SLOVENIA – A point-of-care presepsin measurement in the emergency department displayed powerful accuracy for early rule-out of invasive bacterial infection in infants less than 3 months old presenting with fever without a source, based on results of a phase 3 multicenter Italian study.
“P-SEP [presepsin] is a promising new biomarker. P-SEP accuracy for invasive bacterial infection is comparable to procalcitonin, even though P-SEP, like procalcitonin, is probably not accurate enough to be used as a stand-alone marker to rule-in an invasive bacterial infection,” Luca Pierantoni, MD, said in presenting the preliminary study results at the annual meeting of the European Society for Paediatric Infectious Diseases.
The presepsin test is a rapid point-of-care test well-suited for the ED setting, with a cost equal to that of point-of-care procalcitonin.
Presepsin is a form of soluble CD14 that is released from the surface of macrophages, monocytes, and neutrophils when these immune cells are stimulated by pathogens. “We think it may be a reliable diagnostic and prognostic marker of sepsis in adults and neonates,” explained Dr. Pierantoni of the University of Bologna, Italy.
Indeed, studies in adults suggest presepsin has better sensitivity and specificity than other biomarkers for early diagnosis of sepsis, and that it provides useful information on severity and prognosis as well. But, Dr. Pierantoni and his coworkers wondered, how does it perform in febrile young infants?
The Italian study was designed to address an unmet need: Fever accounts for about one-third of ED visits in infants up to age 3 months, 20% of whom are initially categorized as having fever without source. Yet ultimately 10%-20% of those youngsters having fever without source are found to have an invasive bacterial infection – that is, sepsis or meningitis – or a severe bacterial infection such as pneumonia, a urinary tract infection, or an infected umbilical cord. The sooner these infants can be identified and appropriately treated, the better.
The study enrolled 284 children less than 3 months old who had fever without cause of a mean 10.5 hours duration and presented to the emergency departments of six Italian medical centers. Children were eligible for the study regardless of whether they appeared toxic or well. Presepsin, procalcitonin, and C-reactive protein levels were immediately measured in all participants. Ultimately, 5.6% of subjects were diagnosed with an invasive bacterial infection, and another 21.2% had a severe bacterial infection.
Using a cutoff value of 449 pg/mL, P-SEP had good diagnostic accuracy for invasive bacterial infection, with an area under the receiver operating characteristics curve of 0.81, essentially the same as the 0.82 value for procalcitonin. P-SEP had a sensitivity and specificity of 87% and 75%, respectively, placing it in the same ballpark as the 82% and 86% values for procalcitonin. The strong point for P-SEP was its 99% negative predictive value, as compared to 91% for procalcitonin. The positive predictive values were 17% for P-SEP and 20% for procalcitonin.
In response to an audience question, Dr. Pierantoni speculated that the best use for P-SEP in the setting of fever of unknown origin may be in combination with procalcitonin rather than as a replacement for it. The research team is now in the process of analyzing their study data to see if that is indeed the case.
He reported having no financial conflicts regarding his study, conducted free of commercial support.
LJUBLJANA, SLOVENIA – A point-of-care presepsin measurement in the emergency department displayed powerful accuracy for early rule-out of invasive bacterial infection in infants less than 3 months old presenting with fever without a source, based on results of a phase 3 multicenter Italian study.
“P-SEP [presepsin] is a promising new biomarker. P-SEP accuracy for invasive bacterial infection is comparable to procalcitonin, even though P-SEP, like procalcitonin, is probably not accurate enough to be used as a stand-alone marker to rule-in an invasive bacterial infection,” Luca Pierantoni, MD, said in presenting the preliminary study results at the annual meeting of the European Society for Paediatric Infectious Diseases.
The presepsin test is a rapid point-of-care test well-suited for the ED setting, with a cost equal to that of point-of-care procalcitonin.
Presepsin is a form of soluble CD14 that is released from the surface of macrophages, monocytes, and neutrophils when these immune cells are stimulated by pathogens. “We think it may be a reliable diagnostic and prognostic marker of sepsis in adults and neonates,” explained Dr. Pierantoni of the University of Bologna, Italy.
Indeed, studies in adults suggest presepsin has better sensitivity and specificity than other biomarkers for early diagnosis of sepsis, and that it provides useful information on severity and prognosis as well. But, Dr. Pierantoni and his coworkers wondered, how does it perform in febrile young infants?
The Italian study was designed to address an unmet need: Fever accounts for about one-third of ED visits in infants up to age 3 months, 20% of whom are initially categorized as having fever without source. Yet ultimately 10%-20% of those youngsters having fever without source are found to have an invasive bacterial infection – that is, sepsis or meningitis – or a severe bacterial infection such as pneumonia, a urinary tract infection, or an infected umbilical cord. The sooner these infants can be identified and appropriately treated, the better.
The study enrolled 284 children less than 3 months old who had fever without cause of a mean 10.5 hours duration and presented to the emergency departments of six Italian medical centers. Children were eligible for the study regardless of whether they appeared toxic or well. Presepsin, procalcitonin, and C-reactive protein levels were immediately measured in all participants. Ultimately, 5.6% of subjects were diagnosed with an invasive bacterial infection, and another 21.2% had a severe bacterial infection.
Using a cutoff value of 449 pg/mL, P-SEP had good diagnostic accuracy for invasive bacterial infection, with an area under the receiver operating characteristics curve of 0.81, essentially the same as the 0.82 value for procalcitonin. P-SEP had a sensitivity and specificity of 87% and 75%, respectively, placing it in the same ballpark as the 82% and 86% values for procalcitonin. The strong point for P-SEP was its 99% negative predictive value, as compared to 91% for procalcitonin. The positive predictive values were 17% for P-SEP and 20% for procalcitonin.
In response to an audience question, Dr. Pierantoni speculated that the best use for P-SEP in the setting of fever of unknown origin may be in combination with procalcitonin rather than as a replacement for it. The research team is now in the process of analyzing their study data to see if that is indeed the case.
He reported having no financial conflicts regarding his study, conducted free of commercial support.
REPORTING FROM ESPID 2019
Key clinical point: A rapid point-of-care measurement of presepsin in the ED can rule out invasive bacterial infection with 99% accuracy in young infants with fever of unknown source.
Major finding: The negative predictive value of a presepsin level below the cutoff value of 449 pg/mL was 99%.
Study details: This was a multicenter Italian observational study of 284 infants less than 3 months old who presented to emergency departments with fever without source.
Disclosures: The presenter reported having no financial conflicts regarding his study, conducted free of commercial support.
‘Substantial burden’ of enterovirus meningitis in young infants
LJUBLJANA, SLOVENIA – A prospective international surveillance study has provided new insights into the surprisingly substantial clinical burden of viral meningitis caused by enteroviruses and human parechoviruses in young infants, Seilesh Kadambari, MBBS, PhD, said in his ESPID Young Investigator Award Lecture at the annual meeting of the European Society for Paediatric Infectious Diseases.
This comprehensive study captured all cases of laboratory-confirmed enterovirus (EV) and human parechovirus (HPeV) meningitis in infants less than 90 days old seen by pediatricians in the United Kingdom and Ireland during a 13-month period starting in July 2014, a time free of outbreaks. Dr. Kadambari, a pediatrician at the University of Oxford (England), was first author of the study. It was for this project, as well as his earlier studies shedding light on congenital viral infections, that he received the Young Investigator honor.
Among the key findings of the U.K./Ireland surveillance study: The incidence of EV/HPeV meningitis was more than twice that of bacterial meningitis in the same age group and more than fivefold higher than that of group B streptococcal meningitis, the No. 1 cause of bacterial meningitis in early infancy. Moreover, more than one-half of infants with EV/HPeV meningitis had low levels of inflammatory markers and no cerebrospinal fluid pleocytosis, which underscores the importance of routinely testing the cerebrospinal fluid for viral causes of meningitis in such patients using modern molecular tools such as multiplex polymerase chain reaction, according to Dr. Kadambari.
“Also, not a single one of the patients with EV/HPeV meningitis had a secondary bacterial infection – and that has important implications for management of our antibiotic stewardship programs,” he observed.
The study (Arch Dis Child. 2019 Jun;104(6):552-7) identified 668 cases of EV meningitis and 35 of HPeV meningitis, for an incidence of 0.79 and 0.04 per 1,000 live births, respectively. The most common clinical presentations were those generally seen in meningitis: fever, irritability, and reduced feeding. Circulatory shock was present in 43% of the infants with HPeV and 27% of the infants with EV infections.
Of infants with EV meningitis, 11% required admission to an intensive care unit, as did 23% of those with HPeV meningitis. Two babies with EV meningitis died and four others had continued neurologic complications at 12 months of follow-up. In contrast, all infants with HPeV survived without long-term sequelae.
Reassuringly, none of the 189 infants who underwent formal hearing testing had sensorineural hearing loss.
The surveillance study data have played an influential role in evidence-based guidelines for EV diagnosis and characterization published by the European Society of Clinical Virology (J Clin Virol. 2018 Apr;101:11-7).
An earlier study led by Dr. Kadambari documented a hefty sevenfold increase in the rate of laboratory-confirmed viral meningo-encephalitis in England and Wales during 2004-2013 across all age groups (J Infect. 2014 Oct;69[4]:326-32).
He attributed this increase to improved diagnosis of viral forms of meningitis through greater use of polymerase chain reaction. The study, based upon National Health Service hospital records, showed that more than 90% of all cases of viral meningo-encephalitis in infants less than 90 days old were caused by EV, a finding that prompted the subsequent prospective U.K./Ireland surveillance study.
Dr. Kadambari closed by noting the past decade had seen a greatly improved ability to diagnose congenital viral infections, but those improvements are not good enough.
“In the decade ahead, we hope to improve the management of this poorly understood group of infections,” the pediatrician promised.
Planned efforts include a cost-effectiveness analysis of a cytomegalovirus vaccine, an ESPID-funded research project aimed at identifying which EV/HPeV strains are most responsible for outbreaks and isolated severe disease, and gaining insight into the host-immunity factors associated with a proclivity to develop EV/HPeV meningitis in early infancy.
Dr. Kadambari reported having no financial conflicts regarding his studies, which was funded largely by Public Health England and university grants.
SOURCE: Kadambari S et al. Arch Dis Child. 2019;104:552-7.
LJUBLJANA, SLOVENIA – A prospective international surveillance study has provided new insights into the surprisingly substantial clinical burden of viral meningitis caused by enteroviruses and human parechoviruses in young infants, Seilesh Kadambari, MBBS, PhD, said in his ESPID Young Investigator Award Lecture at the annual meeting of the European Society for Paediatric Infectious Diseases.
This comprehensive study captured all cases of laboratory-confirmed enterovirus (EV) and human parechovirus (HPeV) meningitis in infants less than 90 days old seen by pediatricians in the United Kingdom and Ireland during a 13-month period starting in July 2014, a time free of outbreaks. Dr. Kadambari, a pediatrician at the University of Oxford (England), was first author of the study. It was for this project, as well as his earlier studies shedding light on congenital viral infections, that he received the Young Investigator honor.
Among the key findings of the U.K./Ireland surveillance study: The incidence of EV/HPeV meningitis was more than twice that of bacterial meningitis in the same age group and more than fivefold higher than that of group B streptococcal meningitis, the No. 1 cause of bacterial meningitis in early infancy. Moreover, more than one-half of infants with EV/HPeV meningitis had low levels of inflammatory markers and no cerebrospinal fluid pleocytosis, which underscores the importance of routinely testing the cerebrospinal fluid for viral causes of meningitis in such patients using modern molecular tools such as multiplex polymerase chain reaction, according to Dr. Kadambari.
“Also, not a single one of the patients with EV/HPeV meningitis had a secondary bacterial infection – and that has important implications for management of our antibiotic stewardship programs,” he observed.
The study (Arch Dis Child. 2019 Jun;104(6):552-7) identified 668 cases of EV meningitis and 35 of HPeV meningitis, for an incidence of 0.79 and 0.04 per 1,000 live births, respectively. The most common clinical presentations were those generally seen in meningitis: fever, irritability, and reduced feeding. Circulatory shock was present in 43% of the infants with HPeV and 27% of the infants with EV infections.
Of infants with EV meningitis, 11% required admission to an intensive care unit, as did 23% of those with HPeV meningitis. Two babies with EV meningitis died and four others had continued neurologic complications at 12 months of follow-up. In contrast, all infants with HPeV survived without long-term sequelae.
Reassuringly, none of the 189 infants who underwent formal hearing testing had sensorineural hearing loss.
The surveillance study data have played an influential role in evidence-based guidelines for EV diagnosis and characterization published by the European Society of Clinical Virology (J Clin Virol. 2018 Apr;101:11-7).
An earlier study led by Dr. Kadambari documented a hefty sevenfold increase in the rate of laboratory-confirmed viral meningo-encephalitis in England and Wales during 2004-2013 across all age groups (J Infect. 2014 Oct;69[4]:326-32).
He attributed this increase to improved diagnosis of viral forms of meningitis through greater use of polymerase chain reaction. The study, based upon National Health Service hospital records, showed that more than 90% of all cases of viral meningo-encephalitis in infants less than 90 days old were caused by EV, a finding that prompted the subsequent prospective U.K./Ireland surveillance study.
Dr. Kadambari closed by noting the past decade had seen a greatly improved ability to diagnose congenital viral infections, but those improvements are not good enough.
“In the decade ahead, we hope to improve the management of this poorly understood group of infections,” the pediatrician promised.
Planned efforts include a cost-effectiveness analysis of a cytomegalovirus vaccine, an ESPID-funded research project aimed at identifying which EV/HPeV strains are most responsible for outbreaks and isolated severe disease, and gaining insight into the host-immunity factors associated with a proclivity to develop EV/HPeV meningitis in early infancy.
Dr. Kadambari reported having no financial conflicts regarding his studies, which was funded largely by Public Health England and university grants.
SOURCE: Kadambari S et al. Arch Dis Child. 2019;104:552-7.
LJUBLJANA, SLOVENIA – A prospective international surveillance study has provided new insights into the surprisingly substantial clinical burden of viral meningitis caused by enteroviruses and human parechoviruses in young infants, Seilesh Kadambari, MBBS, PhD, said in his ESPID Young Investigator Award Lecture at the annual meeting of the European Society for Paediatric Infectious Diseases.
This comprehensive study captured all cases of laboratory-confirmed enterovirus (EV) and human parechovirus (HPeV) meningitis in infants less than 90 days old seen by pediatricians in the United Kingdom and Ireland during a 13-month period starting in July 2014, a time free of outbreaks. Dr. Kadambari, a pediatrician at the University of Oxford (England), was first author of the study. It was for this project, as well as his earlier studies shedding light on congenital viral infections, that he received the Young Investigator honor.
Among the key findings of the U.K./Ireland surveillance study: The incidence of EV/HPeV meningitis was more than twice that of bacterial meningitis in the same age group and more than fivefold higher than that of group B streptococcal meningitis, the No. 1 cause of bacterial meningitis in early infancy. Moreover, more than one-half of infants with EV/HPeV meningitis had low levels of inflammatory markers and no cerebrospinal fluid pleocytosis, which underscores the importance of routinely testing the cerebrospinal fluid for viral causes of meningitis in such patients using modern molecular tools such as multiplex polymerase chain reaction, according to Dr. Kadambari.
“Also, not a single one of the patients with EV/HPeV meningitis had a secondary bacterial infection – and that has important implications for management of our antibiotic stewardship programs,” he observed.
The study (Arch Dis Child. 2019 Jun;104(6):552-7) identified 668 cases of EV meningitis and 35 of HPeV meningitis, for an incidence of 0.79 and 0.04 per 1,000 live births, respectively. The most common clinical presentations were those generally seen in meningitis: fever, irritability, and reduced feeding. Circulatory shock was present in 43% of the infants with HPeV and 27% of the infants with EV infections.
Of infants with EV meningitis, 11% required admission to an intensive care unit, as did 23% of those with HPeV meningitis. Two babies with EV meningitis died and four others had continued neurologic complications at 12 months of follow-up. In contrast, all infants with HPeV survived without long-term sequelae.
Reassuringly, none of the 189 infants who underwent formal hearing testing had sensorineural hearing loss.
The surveillance study data have played an influential role in evidence-based guidelines for EV diagnosis and characterization published by the European Society of Clinical Virology (J Clin Virol. 2018 Apr;101:11-7).
An earlier study led by Dr. Kadambari documented a hefty sevenfold increase in the rate of laboratory-confirmed viral meningo-encephalitis in England and Wales during 2004-2013 across all age groups (J Infect. 2014 Oct;69[4]:326-32).
He attributed this increase to improved diagnosis of viral forms of meningitis through greater use of polymerase chain reaction. The study, based upon National Health Service hospital records, showed that more than 90% of all cases of viral meningo-encephalitis in infants less than 90 days old were caused by EV, a finding that prompted the subsequent prospective U.K./Ireland surveillance study.
Dr. Kadambari closed by noting the past decade had seen a greatly improved ability to diagnose congenital viral infections, but those improvements are not good enough.
“In the decade ahead, we hope to improve the management of this poorly understood group of infections,” the pediatrician promised.
Planned efforts include a cost-effectiveness analysis of a cytomegalovirus vaccine, an ESPID-funded research project aimed at identifying which EV/HPeV strains are most responsible for outbreaks and isolated severe disease, and gaining insight into the host-immunity factors associated with a proclivity to develop EV/HPeV meningitis in early infancy.
Dr. Kadambari reported having no financial conflicts regarding his studies, which was funded largely by Public Health England and university grants.
SOURCE: Kadambari S et al. Arch Dis Child. 2019;104:552-7.
REPORTING FROM ESPID 2019
Are rigid HPV vaccination schedules really necessary?
LJUBLJANA, SLOVENIA – Vladimir Gilca, MD, PhD, reported at the annual meeting of the European Society for Paediatric Infectious Diseases.
This novel observation from a post hoc analysis of two clinical trials conducted by the same research team has important potential implications for both clinical practice and public health, according to Dr. Gilca of the Quebec National Institute of Public Health and Laval University, Quebec City.
“A less rigid immunization schedule might facilitate the coadministration of HPV vaccine with other vaccines, such as meningococcal or Tdap, and reduce the number of vaccination visits. Also, our data support the decision to offer only one dose in cases of vaccine shortage, like we have presently in many countries around the world, with the possibility of giving the second dose several years later when the shortage is resolved,” he said.
He presented a comparison of anti-HPV geometric mean IgG antibody titers and their distribution in two clinical trials with serologic assays performed in the same lab using the same enzyme-linked immunosorbent assay procedures. In the first study, 173 boys and girls aged 9-10 years received two doses of a 9-valent HPV vaccine 6 months apart. In the second trial, 31 girls were vaccinated with one dose of a quadrivalent HPV vaccine at age 9-14 years and then received a dose of the 9-valent vaccine at a mean of 5.4 years and maximum of 8 years later. Blood samples were obtained before and 1 month after the second dose in both trials.
Despite the enormous differences in the time between the first and second doses in the two studies, 100% of subjects in both trials were seropositive to HPV 6, 11, 16, and 18, with similar geometric mean titers and titer distributions before dose number two. Moreover, 1 month after the second dose, the geometric mean titers jumped 40-91 times in study participants with a 6-month dosing interval, and similarly by 60-82 times in those with the far lengthier interval. Titer distributions after the second dose were equivalent in the two studies.
Dr. Gilca and coinvestigators looked at subgroups who received their second dose 3-4 years, 6, or 7-8 years after the first. The time difference didn’t affect the distribution of antibodies.
“We conclude that delayed administration of the second dose has no negative impact on the magnitude of the immune response,” he declared.
There are abundant precedents for this phenomenon of high immunogenicity of delayed doses of vaccine. Rabies, anthrax, hepatitis A and B, and tick-borne encephalitis vaccines have all been shown to elicit at least a similar magnitude of immune response after delayed administration of a second or third dose, compared with dosing at the guideline-recommended intervals, he noted.
Asked about the possible approach of giving just one dose of HPV vaccine, as was supported based upon retrospective data in a high-profile presentation earlier at ESPID 2019, Dr. Gilca replied, “The data we’ve seen so far show clinical noninferiority between one, two, and three doses. An approach that might be used by at least some countries is to give, for example, one dose of HPV vaccine in grade 4 and to then wait for confirmatory data about the efficacy of one dose, which we expect in the next 4-5 years. At least five or six clinical trials are ongoing on one dose versus two or three doses.”
He reported having no financial conflicts of interest regarding his presentation.
SOURCE: Gilca V et al. ESPID 2019, Abstract.
LJUBLJANA, SLOVENIA – Vladimir Gilca, MD, PhD, reported at the annual meeting of the European Society for Paediatric Infectious Diseases.
This novel observation from a post hoc analysis of two clinical trials conducted by the same research team has important potential implications for both clinical practice and public health, according to Dr. Gilca of the Quebec National Institute of Public Health and Laval University, Quebec City.
“A less rigid immunization schedule might facilitate the coadministration of HPV vaccine with other vaccines, such as meningococcal or Tdap, and reduce the number of vaccination visits. Also, our data support the decision to offer only one dose in cases of vaccine shortage, like we have presently in many countries around the world, with the possibility of giving the second dose several years later when the shortage is resolved,” he said.
He presented a comparison of anti-HPV geometric mean IgG antibody titers and their distribution in two clinical trials with serologic assays performed in the same lab using the same enzyme-linked immunosorbent assay procedures. In the first study, 173 boys and girls aged 9-10 years received two doses of a 9-valent HPV vaccine 6 months apart. In the second trial, 31 girls were vaccinated with one dose of a quadrivalent HPV vaccine at age 9-14 years and then received a dose of the 9-valent vaccine at a mean of 5.4 years and maximum of 8 years later. Blood samples were obtained before and 1 month after the second dose in both trials.
Despite the enormous differences in the time between the first and second doses in the two studies, 100% of subjects in both trials were seropositive to HPV 6, 11, 16, and 18, with similar geometric mean titers and titer distributions before dose number two. Moreover, 1 month after the second dose, the geometric mean titers jumped 40-91 times in study participants with a 6-month dosing interval, and similarly by 60-82 times in those with the far lengthier interval. Titer distributions after the second dose were equivalent in the two studies.
Dr. Gilca and coinvestigators looked at subgroups who received their second dose 3-4 years, 6, or 7-8 years after the first. The time difference didn’t affect the distribution of antibodies.
“We conclude that delayed administration of the second dose has no negative impact on the magnitude of the immune response,” he declared.
There are abundant precedents for this phenomenon of high immunogenicity of delayed doses of vaccine. Rabies, anthrax, hepatitis A and B, and tick-borne encephalitis vaccines have all been shown to elicit at least a similar magnitude of immune response after delayed administration of a second or third dose, compared with dosing at the guideline-recommended intervals, he noted.
Asked about the possible approach of giving just one dose of HPV vaccine, as was supported based upon retrospective data in a high-profile presentation earlier at ESPID 2019, Dr. Gilca replied, “The data we’ve seen so far show clinical noninferiority between one, two, and three doses. An approach that might be used by at least some countries is to give, for example, one dose of HPV vaccine in grade 4 and to then wait for confirmatory data about the efficacy of one dose, which we expect in the next 4-5 years. At least five or six clinical trials are ongoing on one dose versus two or three doses.”
He reported having no financial conflicts of interest regarding his presentation.
SOURCE: Gilca V et al. ESPID 2019, Abstract.
LJUBLJANA, SLOVENIA – Vladimir Gilca, MD, PhD, reported at the annual meeting of the European Society for Paediatric Infectious Diseases.
This novel observation from a post hoc analysis of two clinical trials conducted by the same research team has important potential implications for both clinical practice and public health, according to Dr. Gilca of the Quebec National Institute of Public Health and Laval University, Quebec City.
“A less rigid immunization schedule might facilitate the coadministration of HPV vaccine with other vaccines, such as meningococcal or Tdap, and reduce the number of vaccination visits. Also, our data support the decision to offer only one dose in cases of vaccine shortage, like we have presently in many countries around the world, with the possibility of giving the second dose several years later when the shortage is resolved,” he said.
He presented a comparison of anti-HPV geometric mean IgG antibody titers and their distribution in two clinical trials with serologic assays performed in the same lab using the same enzyme-linked immunosorbent assay procedures. In the first study, 173 boys and girls aged 9-10 years received two doses of a 9-valent HPV vaccine 6 months apart. In the second trial, 31 girls were vaccinated with one dose of a quadrivalent HPV vaccine at age 9-14 years and then received a dose of the 9-valent vaccine at a mean of 5.4 years and maximum of 8 years later. Blood samples were obtained before and 1 month after the second dose in both trials.
Despite the enormous differences in the time between the first and second doses in the two studies, 100% of subjects in both trials were seropositive to HPV 6, 11, 16, and 18, with similar geometric mean titers and titer distributions before dose number two. Moreover, 1 month after the second dose, the geometric mean titers jumped 40-91 times in study participants with a 6-month dosing interval, and similarly by 60-82 times in those with the far lengthier interval. Titer distributions after the second dose were equivalent in the two studies.
Dr. Gilca and coinvestigators looked at subgroups who received their second dose 3-4 years, 6, or 7-8 years after the first. The time difference didn’t affect the distribution of antibodies.
“We conclude that delayed administration of the second dose has no negative impact on the magnitude of the immune response,” he declared.
There are abundant precedents for this phenomenon of high immunogenicity of delayed doses of vaccine. Rabies, anthrax, hepatitis A and B, and tick-borne encephalitis vaccines have all been shown to elicit at least a similar magnitude of immune response after delayed administration of a second or third dose, compared with dosing at the guideline-recommended intervals, he noted.
Asked about the possible approach of giving just one dose of HPV vaccine, as was supported based upon retrospective data in a high-profile presentation earlier at ESPID 2019, Dr. Gilca replied, “The data we’ve seen so far show clinical noninferiority between one, two, and three doses. An approach that might be used by at least some countries is to give, for example, one dose of HPV vaccine in grade 4 and to then wait for confirmatory data about the efficacy of one dose, which we expect in the next 4-5 years. At least five or six clinical trials are ongoing on one dose versus two or three doses.”
He reported having no financial conflicts of interest regarding his presentation.
SOURCE: Gilca V et al. ESPID 2019, Abstract.
REPORTING FROM ESPID 2019
Novel score spots high-risk febrile children in ED
LJUBLJANA, SLOVENIA – A new age-adjusted quick Sequential Organ Failure Assessment (qSOFA) score designed for use in children presenting to the ED with fever showed good predictive value for admission to critical care within the next 48 hours, Aakash Khanijau, MD, reported at the annual meeting of the European Society for Paediatric Infectious Diseases.
“In the needle-in-a-haystack scenario that’s seen in pediatric emergency departments, our novel, age-adjusted qSOFA score could potentially improve the rapid identification and treatment of children with suspected sepsis presenting to the ED,” said Dr. Khanijau of the University of Liverpool (England).
He presented an exceptionally large retrospective validation study of the score’s performance in 12,393 children (median age, 2.5 years) who presented to EDs with fever, of whom 1,521 were admitted for suspected sepsis. Of the hospitalized children, 145 were admitted to critical care within the first 48 hours.
The pediatric qSOFA score had 72% sensitivity and 85% specificity for critical care admission within 48 hours, with a positive predictive value of 5.4% and, more importantly, a whopping negative predictive value of 99.6%.
“That very high negative predictive value underlines the powerful discriminatory nature of our tool in the emergency department setting,” Dr. Khanijau observed, adding that the score’s area under the receiver operating characteristic curve was 0.81, which is considered a good predictive value.
The impetus for developing an age-adjusted pediatric qSOFA score stems from the fact that the original qSOFA score was designed for rapid assessment of adults with suspected sepsis and isn’t applicable in children. Other existing scores, including SIRS (the Systemic Inflammatory Response Syndrome criteria), the full SOFA, and PELOD-2 (the Pediatric Logistic Organ Dysfunction score), take longer to determine than the adapted qSOFA in a setting where speed is of the essence, he explained.
The original qSOFA components are altered mentation, systolic blood pressure, and respiratory rate. The novel score developed by Dr. Khanijau and coworkers swaps out systolic BP in favor of capillary refill time and age-adjusted heart rate using the thresholds previously established in a landmark study from the Children’s Hospital of Philadelphia (Pediatrics. 2013 Apr;131[4]:e1150-7.)
“Our reasoning here is that arterial hypertension is known to be a much later sign of circulatory compromise in children and may provide less discriminatory value than signs such as delayed capillary refill time and tachycardia early in presentation in the emergency department,” according to Dr. Khanijau.
The novel scoring system features four criteria. One point each is given for a capillary refill time of 3 seconds or longer; anything less than “Alert” on the Alert, Responds to Voice, Respond to Pain, and Unresponsive scale; a heart rate above the 99th percentile on the age-adjusted curves; and a respiratory rate above the age-adjusted 99th percentile. Thus, scores can range from 0 to 4. In the validation study, a score of 2 or more spelled a 890% increased likelihood of being admitted to a critical care setting within 48 hours. It was also associated with a 100-fold increased likelihood of death during the hospitalization, which occurred in 10 children.
Asked how the new predictive score could change clinical management, Dr. Khanijau replied, “I think the key thing it does here is it identifies the children at risk of requiring critical care and should therefore motivate us in the children achieving that threshold to promptly investigate thoroughly for suspected sepsis using the more comprehensive tools, like the full SOFA.”
He reported having no financial conflicts of interest regarding his study.
SOURCE: Khanijau A et al. ESPID 2019, Abstract.
LJUBLJANA, SLOVENIA – A new age-adjusted quick Sequential Organ Failure Assessment (qSOFA) score designed for use in children presenting to the ED with fever showed good predictive value for admission to critical care within the next 48 hours, Aakash Khanijau, MD, reported at the annual meeting of the European Society for Paediatric Infectious Diseases.
“In the needle-in-a-haystack scenario that’s seen in pediatric emergency departments, our novel, age-adjusted qSOFA score could potentially improve the rapid identification and treatment of children with suspected sepsis presenting to the ED,” said Dr. Khanijau of the University of Liverpool (England).
He presented an exceptionally large retrospective validation study of the score’s performance in 12,393 children (median age, 2.5 years) who presented to EDs with fever, of whom 1,521 were admitted for suspected sepsis. Of the hospitalized children, 145 were admitted to critical care within the first 48 hours.
The pediatric qSOFA score had 72% sensitivity and 85% specificity for critical care admission within 48 hours, with a positive predictive value of 5.4% and, more importantly, a whopping negative predictive value of 99.6%.
“That very high negative predictive value underlines the powerful discriminatory nature of our tool in the emergency department setting,” Dr. Khanijau observed, adding that the score’s area under the receiver operating characteristic curve was 0.81, which is considered a good predictive value.
The impetus for developing an age-adjusted pediatric qSOFA score stems from the fact that the original qSOFA score was designed for rapid assessment of adults with suspected sepsis and isn’t applicable in children. Other existing scores, including SIRS (the Systemic Inflammatory Response Syndrome criteria), the full SOFA, and PELOD-2 (the Pediatric Logistic Organ Dysfunction score), take longer to determine than the adapted qSOFA in a setting where speed is of the essence, he explained.
The original qSOFA components are altered mentation, systolic blood pressure, and respiratory rate. The novel score developed by Dr. Khanijau and coworkers swaps out systolic BP in favor of capillary refill time and age-adjusted heart rate using the thresholds previously established in a landmark study from the Children’s Hospital of Philadelphia (Pediatrics. 2013 Apr;131[4]:e1150-7.)
“Our reasoning here is that arterial hypertension is known to be a much later sign of circulatory compromise in children and may provide less discriminatory value than signs such as delayed capillary refill time and tachycardia early in presentation in the emergency department,” according to Dr. Khanijau.
The novel scoring system features four criteria. One point each is given for a capillary refill time of 3 seconds or longer; anything less than “Alert” on the Alert, Responds to Voice, Respond to Pain, and Unresponsive scale; a heart rate above the 99th percentile on the age-adjusted curves; and a respiratory rate above the age-adjusted 99th percentile. Thus, scores can range from 0 to 4. In the validation study, a score of 2 or more spelled a 890% increased likelihood of being admitted to a critical care setting within 48 hours. It was also associated with a 100-fold increased likelihood of death during the hospitalization, which occurred in 10 children.
Asked how the new predictive score could change clinical management, Dr. Khanijau replied, “I think the key thing it does here is it identifies the children at risk of requiring critical care and should therefore motivate us in the children achieving that threshold to promptly investigate thoroughly for suspected sepsis using the more comprehensive tools, like the full SOFA.”
He reported having no financial conflicts of interest regarding his study.
SOURCE: Khanijau A et al. ESPID 2019, Abstract.
LJUBLJANA, SLOVENIA – A new age-adjusted quick Sequential Organ Failure Assessment (qSOFA) score designed for use in children presenting to the ED with fever showed good predictive value for admission to critical care within the next 48 hours, Aakash Khanijau, MD, reported at the annual meeting of the European Society for Paediatric Infectious Diseases.
“In the needle-in-a-haystack scenario that’s seen in pediatric emergency departments, our novel, age-adjusted qSOFA score could potentially improve the rapid identification and treatment of children with suspected sepsis presenting to the ED,” said Dr. Khanijau of the University of Liverpool (England).
He presented an exceptionally large retrospective validation study of the score’s performance in 12,393 children (median age, 2.5 years) who presented to EDs with fever, of whom 1,521 were admitted for suspected sepsis. Of the hospitalized children, 145 were admitted to critical care within the first 48 hours.
The pediatric qSOFA score had 72% sensitivity and 85% specificity for critical care admission within 48 hours, with a positive predictive value of 5.4% and, more importantly, a whopping negative predictive value of 99.6%.
“That very high negative predictive value underlines the powerful discriminatory nature of our tool in the emergency department setting,” Dr. Khanijau observed, adding that the score’s area under the receiver operating characteristic curve was 0.81, which is considered a good predictive value.
The impetus for developing an age-adjusted pediatric qSOFA score stems from the fact that the original qSOFA score was designed for rapid assessment of adults with suspected sepsis and isn’t applicable in children. Other existing scores, including SIRS (the Systemic Inflammatory Response Syndrome criteria), the full SOFA, and PELOD-2 (the Pediatric Logistic Organ Dysfunction score), take longer to determine than the adapted qSOFA in a setting where speed is of the essence, he explained.
The original qSOFA components are altered mentation, systolic blood pressure, and respiratory rate. The novel score developed by Dr. Khanijau and coworkers swaps out systolic BP in favor of capillary refill time and age-adjusted heart rate using the thresholds previously established in a landmark study from the Children’s Hospital of Philadelphia (Pediatrics. 2013 Apr;131[4]:e1150-7.)
“Our reasoning here is that arterial hypertension is known to be a much later sign of circulatory compromise in children and may provide less discriminatory value than signs such as delayed capillary refill time and tachycardia early in presentation in the emergency department,” according to Dr. Khanijau.
The novel scoring system features four criteria. One point each is given for a capillary refill time of 3 seconds or longer; anything less than “Alert” on the Alert, Responds to Voice, Respond to Pain, and Unresponsive scale; a heart rate above the 99th percentile on the age-adjusted curves; and a respiratory rate above the age-adjusted 99th percentile. Thus, scores can range from 0 to 4. In the validation study, a score of 2 or more spelled a 890% increased likelihood of being admitted to a critical care setting within 48 hours. It was also associated with a 100-fold increased likelihood of death during the hospitalization, which occurred in 10 children.
Asked how the new predictive score could change clinical management, Dr. Khanijau replied, “I think the key thing it does here is it identifies the children at risk of requiring critical care and should therefore motivate us in the children achieving that threshold to promptly investigate thoroughly for suspected sepsis using the more comprehensive tools, like the full SOFA.”
He reported having no financial conflicts of interest regarding his study.
SOURCE: Khanijau A et al. ESPID 2019, Abstract.
REPORTING FROM ESPID 2019
Hospital slashes S. aureus vancomycin resistance
LJUBLJANA, SLOVENIA – , Johannes Huebner, MD, reported at the annual meeting of the European Society for Paediatric Infectious Diseases.
He presented a retrospective analysis of S. aureus isolates obtained from 540 patients at the Dr. von Hauner Children’s Hospital, Munich, from 2002 to 2017. All were either newly identified methicillin-resistant S. aureus (MRSA) or specimens from bacteremic children with invasive MRSA or methicillin-sensitive S. aureus (MSSA). The strains were tested for vancomycin resistance and minimum inhibitory concentration (MIC). The results from the 200 isolates obtained from 2002 to 2009 were then compared to the 340 specimens from 2010 to 2017, when antibiotic stewardship programs rose to the fore at the pediatric hospital.
All samples proved to be vancomycin sensitive. The further good news was there was absolutely no evidence of the worrisome vancomycin MIC creep that has been described at some centers. On the contrary, the MIC was significantly lower in the later samples, at 0.99 mcg/mL, compared with 1.11 mcg/mL in the earlier period. Moreover, the prevalence of heterogeneous glycopeptide-intermediate S. aureus (hGISA) – a phenotype that has been associated with increased rates of treatment failure – improved from 25% in the earlier period to 6% during the later period, reported Dr. Huebner, head of the division of pediatric infectious diseases at the children’s hospital, part of the University of Munich.
Vancomycin MICs weren’t significantly different between the MRSA and MSSA samples.
Based upon this favorable institutional experience, vancomycin remains the first-line treatment for suspected severe gram-positive cocci infections as well as proven infections involving MRSA at Dr. von Hauner Children’s Hospital.
These vancomycin MIC and hGISA data underscore the importance of periodically monitoring local S. aureus antimicrobial susceptibilities, which, as in this case, can differ from the broader global trends. The vancomycin MIC creep issue hadn’t been studied previously in German hospitals, according to Dr. Huebner.
He and his coworkers have published details of the elements of pediatric antibiotic stewardship programs they have found to be most effective (Infection. 2017 Aug;45[4]:493-504) as well as a systematic review of studies on the favorable economic impact of such programs (J Hosp Infect. 2019 Aug;102[4]:369-376).
Dr. Huebner reported having no financial conflicts regarding his study, which was conducted free of commercial support.
LJUBLJANA, SLOVENIA – , Johannes Huebner, MD, reported at the annual meeting of the European Society for Paediatric Infectious Diseases.
He presented a retrospective analysis of S. aureus isolates obtained from 540 patients at the Dr. von Hauner Children’s Hospital, Munich, from 2002 to 2017. All were either newly identified methicillin-resistant S. aureus (MRSA) or specimens from bacteremic children with invasive MRSA or methicillin-sensitive S. aureus (MSSA). The strains were tested for vancomycin resistance and minimum inhibitory concentration (MIC). The results from the 200 isolates obtained from 2002 to 2009 were then compared to the 340 specimens from 2010 to 2017, when antibiotic stewardship programs rose to the fore at the pediatric hospital.
All samples proved to be vancomycin sensitive. The further good news was there was absolutely no evidence of the worrisome vancomycin MIC creep that has been described at some centers. On the contrary, the MIC was significantly lower in the later samples, at 0.99 mcg/mL, compared with 1.11 mcg/mL in the earlier period. Moreover, the prevalence of heterogeneous glycopeptide-intermediate S. aureus (hGISA) – a phenotype that has been associated with increased rates of treatment failure – improved from 25% in the earlier period to 6% during the later period, reported Dr. Huebner, head of the division of pediatric infectious diseases at the children’s hospital, part of the University of Munich.
Vancomycin MICs weren’t significantly different between the MRSA and MSSA samples.
Based upon this favorable institutional experience, vancomycin remains the first-line treatment for suspected severe gram-positive cocci infections as well as proven infections involving MRSA at Dr. von Hauner Children’s Hospital.
These vancomycin MIC and hGISA data underscore the importance of periodically monitoring local S. aureus antimicrobial susceptibilities, which, as in this case, can differ from the broader global trends. The vancomycin MIC creep issue hadn’t been studied previously in German hospitals, according to Dr. Huebner.
He and his coworkers have published details of the elements of pediatric antibiotic stewardship programs they have found to be most effective (Infection. 2017 Aug;45[4]:493-504) as well as a systematic review of studies on the favorable economic impact of such programs (J Hosp Infect. 2019 Aug;102[4]:369-376).
Dr. Huebner reported having no financial conflicts regarding his study, which was conducted free of commercial support.
LJUBLJANA, SLOVENIA – , Johannes Huebner, MD, reported at the annual meeting of the European Society for Paediatric Infectious Diseases.
He presented a retrospective analysis of S. aureus isolates obtained from 540 patients at the Dr. von Hauner Children’s Hospital, Munich, from 2002 to 2017. All were either newly identified methicillin-resistant S. aureus (MRSA) or specimens from bacteremic children with invasive MRSA or methicillin-sensitive S. aureus (MSSA). The strains were tested for vancomycin resistance and minimum inhibitory concentration (MIC). The results from the 200 isolates obtained from 2002 to 2009 were then compared to the 340 specimens from 2010 to 2017, when antibiotic stewardship programs rose to the fore at the pediatric hospital.
All samples proved to be vancomycin sensitive. The further good news was there was absolutely no evidence of the worrisome vancomycin MIC creep that has been described at some centers. On the contrary, the MIC was significantly lower in the later samples, at 0.99 mcg/mL, compared with 1.11 mcg/mL in the earlier period. Moreover, the prevalence of heterogeneous glycopeptide-intermediate S. aureus (hGISA) – a phenotype that has been associated with increased rates of treatment failure – improved from 25% in the earlier period to 6% during the later period, reported Dr. Huebner, head of the division of pediatric infectious diseases at the children’s hospital, part of the University of Munich.
Vancomycin MICs weren’t significantly different between the MRSA and MSSA samples.
Based upon this favorable institutional experience, vancomycin remains the first-line treatment for suspected severe gram-positive cocci infections as well as proven infections involving MRSA at Dr. von Hauner Children’s Hospital.
These vancomycin MIC and hGISA data underscore the importance of periodically monitoring local S. aureus antimicrobial susceptibilities, which, as in this case, can differ from the broader global trends. The vancomycin MIC creep issue hadn’t been studied previously in German hospitals, according to Dr. Huebner.
He and his coworkers have published details of the elements of pediatric antibiotic stewardship programs they have found to be most effective (Infection. 2017 Aug;45[4]:493-504) as well as a systematic review of studies on the favorable economic impact of such programs (J Hosp Infect. 2019 Aug;102[4]:369-376).
Dr. Huebner reported having no financial conflicts regarding his study, which was conducted free of commercial support.
REPORTING FROM ESPID 2019
Key clinical point: Staphylococcus aureus vancomycin MIC creep is reversible through dedicated antimicrobial stewardship.
Major finding: The prevalence of hGISA in MRSA and MSSA specimens improved from 25% during 2002-2009 to 6% during 2010-2017 at one German tertiary children’s hospital.
Study details: This was a retrospective single-center analysis of vancomycin resistance trends over time in 540 S. aureus specimens gathered in 2002-2017.
Disclosures: The presenter reported having no financial conflicts regarding this study, which was conducted free of commercial support.
mRNA technology for respiratory vaccines impresses
LJUBLJANA, SLOVENIA – Encouraging safety and immunogenicity results reported from phase 1 studies of the first mRNA vaccines against the potentially pandemic H10N8 avian influenza and H7N9 influenza viruses suggest a bright future for what appears to be a breakthrough technology in vaccine development.
“We have developed an mRNA platform that has the potential to be quite applicable to the vaccine space. It’s an agile platform with the potential for relatively rapid development of vaccine antigen without the use of dedicated facilities, or growth in eggs, or insects, or mammalian cells,” Lori Panther, MD, said at the annual meeting of the European Society for Paediatric Infectious Diseases.
“We now have a platform that is relatively plug and play. If one has the mRNA sequence that you’re after to produce the protein that you’re after, it is a relatively repetitive process somewhat irrespective of the goal of the protein that you’re going to manufacture. We’re introducing an mRNA into our cellular machinery – the destination is the cellular ribosome – where it hopefully is able to be translated with fidelity into the target protein. Essentially it’s like the biological equivalent of a software hack for our own cells,” explained Dr. Panther, who is director of clinical development for infectious diseases at Moderna, in Cambridge, Mass.
Indeed, Moderna has numerous ongoing or recently completed phase 1 clinical trials of mRNA vaccines developed to protect against a raft of viral infections: respiratory syncytial virus, cytomegalovirus (NCT03382405), zika, chikungunya (NCT03829384), human metapneumovirus, and parainfluenza virus 3, as well as the aforementioned H10N8 and H7N9 influenza viruses. And an mRNA varicella zoster virus vaccine is in preclinical studies.
The mRNA vaccines closely mimic native viral infections, eliciting both B- and T-cell responses.
Moreover, the company also has ongoing phase 1 studies of mRNA-based cancer vaccines – therapies targeting solid tumors and lymphomas – as well as mRNA-directed increased production of relaxin as a treatment for heart failure and of vascular endothelial growth factor to treat myocardial ischemia.
“For the purposes of my company, the desired protein at this juncture could be an antibody, it could be a tumor antigen, it could be an enzyme that will replace an enzyme that’s lacking in somebody with an inborn error of metabolism. Or it could be a vaccine antigen target,” Dr. Panther said.
In addition to highlighting the results of the two phase 1 proof-of-concept studies of mRNA vaccines targeting the feared H10N8 and H7N9 influenza viruses, she presented interim results of an ongoing 1-year study of an mRNA vaccine that contains two antigens simultaneously targeting human metapneumovirus (hMPV) and parainfluenza virus 3 (PIV3).
“The rationale behind this study is that, taken together, these are two viruses that are responsible for a fair bit of disease burden in terms of lower respiratory tract infections and hospitalizations in children [younger] than 12 months of age, which will be the target population,” the infectious disease specialist noted.
The early positive results of the mRNA influenza vaccine studies were of particular interest to her audience of pediatric infectious disease specialists. Since the first human H7N9 infections were reported in China in 2013, five outbreaks have occurred involving more than 1,500 documented infections, resulting in more than 600 deaths. And ever since the virulent H10N8 avian influenza virus popped up on the radar in 2013, infectious disease physicians the world over have been waiting for the other shoe to drop.
There is obvious appeal to a novel, precise, and rapidly scalable technology such as that promised by intracellular delivery of mRNA in order to ramp up high-volume production of effective vaccines in the face of a looming pandemic threat. Elsewhere at the meeting, it was noted that, during the H1N1 pandemic of 2009, it took 6 months for the first vaccine doses to become available using current antiquated egg-based production methods. Another 2 months elapsed before the necessary millions of doses were produced.
The details of the two phase 1 studies of the mRNA vaccines against H7N9 and H10N8 influenza have recently been published (Vaccine. 2019 May 31;37[25]:3326-34). The vaccines, delivered in the conventional manner via injection into the deltoid muscle, were well tolerated, with the most common adverse events being the familiar ones: injection site pain, erythema, headache, fatigue, and myalgia. The immune response was robust and durable.
In response to an audience question, Dr. Panther said the mRNA vaccines are amenable to development as intranasal formulations.
The ongoing 12-month, phase 1, dose-ranging study of the mRNA hMPV/PIV3 virus vaccine includes 124 healthy adults at three U.S. sites who received two vaccinations on days 1 and 28. One month after a single vaccination, hMPV neutralizing antibody titers were 6.2-6.4 times those in the placebo arm; PIV3 neutralization titers were increased 3.3-fold. The second injection didn’t further boost antibody titers, suggesting that, at least in this study population of preexposed adults, a single vaccination is sufficient.
The use of mRNA technology has been a long time in coming. Dr. Panther explained why: “It’s a big trick to take an mRNA that by its own nature is a pretty fragile molecule and to get it past the degrading enzymes, like RNAses, that are out to chew it up immediately, and then to sneak it across the cellular membrane and into the cytoplasm, all the while avoiding the innate immune responses that exist solely to recognize RNA that looks foreign and chew it up.”
Moderna has accomplished this using a proprietary lipid nanoparticle delivery system.
“Essentially it’s a lipid shield that surrounds the mRNAs and ushers them past those enzymes and past the innate immune response that would otherwise destroy them,” according to Dr. Panther.
She and her colleagues believe they may eventually be able to change the nucleotide sequence of their manufactured mRNAs in order to expand the immunogenicity epitope and achieve a stronger immune response than would result from natural infection.
LJUBLJANA, SLOVENIA – Encouraging safety and immunogenicity results reported from phase 1 studies of the first mRNA vaccines against the potentially pandemic H10N8 avian influenza and H7N9 influenza viruses suggest a bright future for what appears to be a breakthrough technology in vaccine development.
“We have developed an mRNA platform that has the potential to be quite applicable to the vaccine space. It’s an agile platform with the potential for relatively rapid development of vaccine antigen without the use of dedicated facilities, or growth in eggs, or insects, or mammalian cells,” Lori Panther, MD, said at the annual meeting of the European Society for Paediatric Infectious Diseases.
“We now have a platform that is relatively plug and play. If one has the mRNA sequence that you’re after to produce the protein that you’re after, it is a relatively repetitive process somewhat irrespective of the goal of the protein that you’re going to manufacture. We’re introducing an mRNA into our cellular machinery – the destination is the cellular ribosome – where it hopefully is able to be translated with fidelity into the target protein. Essentially it’s like the biological equivalent of a software hack for our own cells,” explained Dr. Panther, who is director of clinical development for infectious diseases at Moderna, in Cambridge, Mass.
Indeed, Moderna has numerous ongoing or recently completed phase 1 clinical trials of mRNA vaccines developed to protect against a raft of viral infections: respiratory syncytial virus, cytomegalovirus (NCT03382405), zika, chikungunya (NCT03829384), human metapneumovirus, and parainfluenza virus 3, as well as the aforementioned H10N8 and H7N9 influenza viruses. And an mRNA varicella zoster virus vaccine is in preclinical studies.
The mRNA vaccines closely mimic native viral infections, eliciting both B- and T-cell responses.
Moreover, the company also has ongoing phase 1 studies of mRNA-based cancer vaccines – therapies targeting solid tumors and lymphomas – as well as mRNA-directed increased production of relaxin as a treatment for heart failure and of vascular endothelial growth factor to treat myocardial ischemia.
“For the purposes of my company, the desired protein at this juncture could be an antibody, it could be a tumor antigen, it could be an enzyme that will replace an enzyme that’s lacking in somebody with an inborn error of metabolism. Or it could be a vaccine antigen target,” Dr. Panther said.
In addition to highlighting the results of the two phase 1 proof-of-concept studies of mRNA vaccines targeting the feared H10N8 and H7N9 influenza viruses, she presented interim results of an ongoing 1-year study of an mRNA vaccine that contains two antigens simultaneously targeting human metapneumovirus (hMPV) and parainfluenza virus 3 (PIV3).
“The rationale behind this study is that, taken together, these are two viruses that are responsible for a fair bit of disease burden in terms of lower respiratory tract infections and hospitalizations in children [younger] than 12 months of age, which will be the target population,” the infectious disease specialist noted.
The early positive results of the mRNA influenza vaccine studies were of particular interest to her audience of pediatric infectious disease specialists. Since the first human H7N9 infections were reported in China in 2013, five outbreaks have occurred involving more than 1,500 documented infections, resulting in more than 600 deaths. And ever since the virulent H10N8 avian influenza virus popped up on the radar in 2013, infectious disease physicians the world over have been waiting for the other shoe to drop.
There is obvious appeal to a novel, precise, and rapidly scalable technology such as that promised by intracellular delivery of mRNA in order to ramp up high-volume production of effective vaccines in the face of a looming pandemic threat. Elsewhere at the meeting, it was noted that, during the H1N1 pandemic of 2009, it took 6 months for the first vaccine doses to become available using current antiquated egg-based production methods. Another 2 months elapsed before the necessary millions of doses were produced.
The details of the two phase 1 studies of the mRNA vaccines against H7N9 and H10N8 influenza have recently been published (Vaccine. 2019 May 31;37[25]:3326-34). The vaccines, delivered in the conventional manner via injection into the deltoid muscle, were well tolerated, with the most common adverse events being the familiar ones: injection site pain, erythema, headache, fatigue, and myalgia. The immune response was robust and durable.
In response to an audience question, Dr. Panther said the mRNA vaccines are amenable to development as intranasal formulations.
The ongoing 12-month, phase 1, dose-ranging study of the mRNA hMPV/PIV3 virus vaccine includes 124 healthy adults at three U.S. sites who received two vaccinations on days 1 and 28. One month after a single vaccination, hMPV neutralizing antibody titers were 6.2-6.4 times those in the placebo arm; PIV3 neutralization titers were increased 3.3-fold. The second injection didn’t further boost antibody titers, suggesting that, at least in this study population of preexposed adults, a single vaccination is sufficient.
The use of mRNA technology has been a long time in coming. Dr. Panther explained why: “It’s a big trick to take an mRNA that by its own nature is a pretty fragile molecule and to get it past the degrading enzymes, like RNAses, that are out to chew it up immediately, and then to sneak it across the cellular membrane and into the cytoplasm, all the while avoiding the innate immune responses that exist solely to recognize RNA that looks foreign and chew it up.”
Moderna has accomplished this using a proprietary lipid nanoparticle delivery system.
“Essentially it’s a lipid shield that surrounds the mRNAs and ushers them past those enzymes and past the innate immune response that would otherwise destroy them,” according to Dr. Panther.
She and her colleagues believe they may eventually be able to change the nucleotide sequence of their manufactured mRNAs in order to expand the immunogenicity epitope and achieve a stronger immune response than would result from natural infection.
LJUBLJANA, SLOVENIA – Encouraging safety and immunogenicity results reported from phase 1 studies of the first mRNA vaccines against the potentially pandemic H10N8 avian influenza and H7N9 influenza viruses suggest a bright future for what appears to be a breakthrough technology in vaccine development.
“We have developed an mRNA platform that has the potential to be quite applicable to the vaccine space. It’s an agile platform with the potential for relatively rapid development of vaccine antigen without the use of dedicated facilities, or growth in eggs, or insects, or mammalian cells,” Lori Panther, MD, said at the annual meeting of the European Society for Paediatric Infectious Diseases.
“We now have a platform that is relatively plug and play. If one has the mRNA sequence that you’re after to produce the protein that you’re after, it is a relatively repetitive process somewhat irrespective of the goal of the protein that you’re going to manufacture. We’re introducing an mRNA into our cellular machinery – the destination is the cellular ribosome – where it hopefully is able to be translated with fidelity into the target protein. Essentially it’s like the biological equivalent of a software hack for our own cells,” explained Dr. Panther, who is director of clinical development for infectious diseases at Moderna, in Cambridge, Mass.
Indeed, Moderna has numerous ongoing or recently completed phase 1 clinical trials of mRNA vaccines developed to protect against a raft of viral infections: respiratory syncytial virus, cytomegalovirus (NCT03382405), zika, chikungunya (NCT03829384), human metapneumovirus, and parainfluenza virus 3, as well as the aforementioned H10N8 and H7N9 influenza viruses. And an mRNA varicella zoster virus vaccine is in preclinical studies.
The mRNA vaccines closely mimic native viral infections, eliciting both B- and T-cell responses.
Moreover, the company also has ongoing phase 1 studies of mRNA-based cancer vaccines – therapies targeting solid tumors and lymphomas – as well as mRNA-directed increased production of relaxin as a treatment for heart failure and of vascular endothelial growth factor to treat myocardial ischemia.
“For the purposes of my company, the desired protein at this juncture could be an antibody, it could be a tumor antigen, it could be an enzyme that will replace an enzyme that’s lacking in somebody with an inborn error of metabolism. Or it could be a vaccine antigen target,” Dr. Panther said.
In addition to highlighting the results of the two phase 1 proof-of-concept studies of mRNA vaccines targeting the feared H10N8 and H7N9 influenza viruses, she presented interim results of an ongoing 1-year study of an mRNA vaccine that contains two antigens simultaneously targeting human metapneumovirus (hMPV) and parainfluenza virus 3 (PIV3).
“The rationale behind this study is that, taken together, these are two viruses that are responsible for a fair bit of disease burden in terms of lower respiratory tract infections and hospitalizations in children [younger] than 12 months of age, which will be the target population,” the infectious disease specialist noted.
The early positive results of the mRNA influenza vaccine studies were of particular interest to her audience of pediatric infectious disease specialists. Since the first human H7N9 infections were reported in China in 2013, five outbreaks have occurred involving more than 1,500 documented infections, resulting in more than 600 deaths. And ever since the virulent H10N8 avian influenza virus popped up on the radar in 2013, infectious disease physicians the world over have been waiting for the other shoe to drop.
There is obvious appeal to a novel, precise, and rapidly scalable technology such as that promised by intracellular delivery of mRNA in order to ramp up high-volume production of effective vaccines in the face of a looming pandemic threat. Elsewhere at the meeting, it was noted that, during the H1N1 pandemic of 2009, it took 6 months for the first vaccine doses to become available using current antiquated egg-based production methods. Another 2 months elapsed before the necessary millions of doses were produced.
The details of the two phase 1 studies of the mRNA vaccines against H7N9 and H10N8 influenza have recently been published (Vaccine. 2019 May 31;37[25]:3326-34). The vaccines, delivered in the conventional manner via injection into the deltoid muscle, were well tolerated, with the most common adverse events being the familiar ones: injection site pain, erythema, headache, fatigue, and myalgia. The immune response was robust and durable.
In response to an audience question, Dr. Panther said the mRNA vaccines are amenable to development as intranasal formulations.
The ongoing 12-month, phase 1, dose-ranging study of the mRNA hMPV/PIV3 virus vaccine includes 124 healthy adults at three U.S. sites who received two vaccinations on days 1 and 28. One month after a single vaccination, hMPV neutralizing antibody titers were 6.2-6.4 times those in the placebo arm; PIV3 neutralization titers were increased 3.3-fold. The second injection didn’t further boost antibody titers, suggesting that, at least in this study population of preexposed adults, a single vaccination is sufficient.
The use of mRNA technology has been a long time in coming. Dr. Panther explained why: “It’s a big trick to take an mRNA that by its own nature is a pretty fragile molecule and to get it past the degrading enzymes, like RNAses, that are out to chew it up immediately, and then to sneak it across the cellular membrane and into the cytoplasm, all the while avoiding the innate immune responses that exist solely to recognize RNA that looks foreign and chew it up.”
Moderna has accomplished this using a proprietary lipid nanoparticle delivery system.
“Essentially it’s a lipid shield that surrounds the mRNAs and ushers them past those enzymes and past the innate immune response that would otherwise destroy them,” according to Dr. Panther.
She and her colleagues believe they may eventually be able to change the nucleotide sequence of their manufactured mRNAs in order to expand the immunogenicity epitope and achieve a stronger immune response than would result from natural infection.
REPORTING FROM ESPID 2019
What’s new in pediatric sepsis
LJUBLJANA, SLOVENIA – The dogma of the “Golden Hour” for the immediate management of pediatric sepsis has been oversold and actually is based upon weak evidence, Luregn J. Schlapbach, MD, asserted at the annual meeting of the European Society for Paediatric Infectious Diseases.
The true Golden Hour – that is, the time frame within which it’s imperative to administer the sepsis bundle comprised of appropriate antibiotics, fluids, and inotropes – is probably more like 3 hours.
“The evidence suggests that up to 3 hours you don’t really have a big difference in outcomes for sepsis. If you recognize shock there’s no question: You should not even wait 1 hour. But if you’re not certain, it may be better to give up to 3 hours to work up the child and get the senior clinician involved before you make decisions about treatment. So I’m not advocating to delay anything, said Dr. Schlapbach, a pediatric intensivist at the Child Health Research Center at the University of Queensland in South Brisbane, Australia.
The problem with a 1-hour mandate for delivery of the sepsis bundle, as recommended in guidelines by the Surviving Sepsis Campaign and the American College of Critical Care Medicine, and endorsed in quality improvement initiatives, is that the time pressure pushes physicians to overprescribe antibiotics to children who don’t actually have a serious bacterial infection. And that, he noted, contributes to the growing problem of antimicrobial resistance.
“You may have a child where you’re not too sure. Usually you would have done a urine culture because UTI [urinary tract infection] is quite a common cause of these infections, and many of these kids aren’t necessarily septic. But if people tell you that within 1 hour you need to treat, are you going to take the time to do the urine culture, or are you just going to decide to treat?” he asked rhetorically.
Dr. Schlapbach is a world-renowned pediatric sepsis researcher. He is far from alone in his reservations about the Golden Hour mandate.
“This is one of the reasons why IDSA [the Infectious Diseases Society of America] has not endorsed the Surviving Sepsis Campaign,” according to the physician, who noted that, in a position statement, IDSA officials have declared that discrimination of sepsis from noninfectious conditions remains a challenge, and that a 60-minute time to antibiotics may jeopardize patient reassessment (Clin Infect Dis. 2018 May 15;66[10]:1631-5).
Dr. Schlapbach highlighted other recent developments in pediatric sepsis.
The definition of adult sepsis has changed, and the pediatric version needs to as well
The revised definition of sepsis, known as Sepsis-3, issued by the International Sepsis Definition Task Force in 2016 notably dropped systemic inflammatory response syndrome (SIRS), as a requirement for sepsis (JAMA. 2016;315[8]:801-10). The revised definition characterizes sepsis as a dysregulated host response to infection resulting in life-threatening organ dysfunction. But Sepsis-3 is based entirely on adult data and is not considered applicable to children.
The current Pediatric Sepsis Consensus Conference definition dates back to 2005. A comprehensive revision is getting underway. It, too, is likely to drop SIRS into the wastebasket, Dr. Schlapbach said.
“It is probably time to abandon the old view of sepsis disease progression, which proposes a progression from infection to SIRS to severe sepsis with organ dysfunction to septic shock, because most children with infection do manifest signs of SIRS, such as tachycardia, tachypnea, and fever, and these probably should be considered as more of an adaptive rather than a maladaptive response,” he explained.
The goal of the pediatric sepsis redefinition project is to come up with something more useful for clinicians than the Sepsis-3 definition. While the Sepsis-3 concept of a dysregulated host response to infection sounds nice, he explained, “we don’t actually know what it is.
“One of the challenges that you all know as pediatricians is that children who develop sepsis get sick very, very quickly. We all have memories of children who we saw and may have discharged, and they were dead 12 hours later,” he noted.
Indeed, he and others have shown in multiple studies that up to 50% of pediatric deaths caused by sepsis happen within 24 hours of presentation.
“So whatever happens, it happens very quickly. The true question for us is actually how and why do children progress from no organ dysfunction, where the mortality is close to zero, to organ dysfunction, where all of a sudden mortality jumps up dramatically. It’s this progression that we don’t understand at all,” according to Dr. Schlapbach.
The genetic contribution to fulminant sepsis in children may be substantial
One-third of pediatric sepsis deaths in high-income countries happen in previously healthy children. In a proof-of-concept study, Dr. Schlapbach and coinvestigators in the Swiss Pediatric Sepsis Study Group conducted exome-sequencing genetic studies in eight previously healthy children with no family history of immunodeficiency who died of severe sepsis because of community-acquired Pseudomonas aeruginosa infection. Two of the eight had rare loss-of-function mutations in genes known to cause primary immunodeficiencies. The investigators proposed that unusually severe sepsis in previously healthy children warrants exome sequencing to look for underlying previously undetected primary immunodeficiencies. That’s important information for survivors and/or affected families to have, they argued (Front Immunol. 2016 Sep 20;7:357. eCollection 2016).
“There are some indications that the genetic contribution in children with sepsis may be larger than previously assumed,” he said.
The longstanding practice of fluid bolus therapy for resuscitation in pediatric sepsis is being reexamined
The FEAST (Fluid Expansion As Supportive Therapy) study, a randomized trial of more than 3,000 children with severe febrile illness and impaired perfusion in sub-Saharan Africa, turned heads with its finding that fluid boluses significantly increased 48-hour mortality (BMC Med. 2013 Mar 14;11:67).
Indeed, the FEAST findings, supported by mechanistic animal studies, were sufficiently compelling that the use of fluid boluses in both pediatric and adult septic shock is now under scrutiny in two major randomized trials: RIFTS (the Restrictive IV Fluid Trial in Severe Sepsis and Septic Shock), and CLOVERS (Crystalloid Liberal or Vasopressors Early Resuscitation in Sepsis). Stay tuned.
Dr. Schlapbach reported having no financial conflicts regarding his presentation.
LJUBLJANA, SLOVENIA – The dogma of the “Golden Hour” for the immediate management of pediatric sepsis has been oversold and actually is based upon weak evidence, Luregn J. Schlapbach, MD, asserted at the annual meeting of the European Society for Paediatric Infectious Diseases.
The true Golden Hour – that is, the time frame within which it’s imperative to administer the sepsis bundle comprised of appropriate antibiotics, fluids, and inotropes – is probably more like 3 hours.
“The evidence suggests that up to 3 hours you don’t really have a big difference in outcomes for sepsis. If you recognize shock there’s no question: You should not even wait 1 hour. But if you’re not certain, it may be better to give up to 3 hours to work up the child and get the senior clinician involved before you make decisions about treatment. So I’m not advocating to delay anything, said Dr. Schlapbach, a pediatric intensivist at the Child Health Research Center at the University of Queensland in South Brisbane, Australia.
The problem with a 1-hour mandate for delivery of the sepsis bundle, as recommended in guidelines by the Surviving Sepsis Campaign and the American College of Critical Care Medicine, and endorsed in quality improvement initiatives, is that the time pressure pushes physicians to overprescribe antibiotics to children who don’t actually have a serious bacterial infection. And that, he noted, contributes to the growing problem of antimicrobial resistance.
“You may have a child where you’re not too sure. Usually you would have done a urine culture because UTI [urinary tract infection] is quite a common cause of these infections, and many of these kids aren’t necessarily septic. But if people tell you that within 1 hour you need to treat, are you going to take the time to do the urine culture, or are you just going to decide to treat?” he asked rhetorically.
Dr. Schlapbach is a world-renowned pediatric sepsis researcher. He is far from alone in his reservations about the Golden Hour mandate.
“This is one of the reasons why IDSA [the Infectious Diseases Society of America] has not endorsed the Surviving Sepsis Campaign,” according to the physician, who noted that, in a position statement, IDSA officials have declared that discrimination of sepsis from noninfectious conditions remains a challenge, and that a 60-minute time to antibiotics may jeopardize patient reassessment (Clin Infect Dis. 2018 May 15;66[10]:1631-5).
Dr. Schlapbach highlighted other recent developments in pediatric sepsis.
The definition of adult sepsis has changed, and the pediatric version needs to as well
The revised definition of sepsis, known as Sepsis-3, issued by the International Sepsis Definition Task Force in 2016 notably dropped systemic inflammatory response syndrome (SIRS), as a requirement for sepsis (JAMA. 2016;315[8]:801-10). The revised definition characterizes sepsis as a dysregulated host response to infection resulting in life-threatening organ dysfunction. But Sepsis-3 is based entirely on adult data and is not considered applicable to children.
The current Pediatric Sepsis Consensus Conference definition dates back to 2005. A comprehensive revision is getting underway. It, too, is likely to drop SIRS into the wastebasket, Dr. Schlapbach said.
“It is probably time to abandon the old view of sepsis disease progression, which proposes a progression from infection to SIRS to severe sepsis with organ dysfunction to septic shock, because most children with infection do manifest signs of SIRS, such as tachycardia, tachypnea, and fever, and these probably should be considered as more of an adaptive rather than a maladaptive response,” he explained.
The goal of the pediatric sepsis redefinition project is to come up with something more useful for clinicians than the Sepsis-3 definition. While the Sepsis-3 concept of a dysregulated host response to infection sounds nice, he explained, “we don’t actually know what it is.
“One of the challenges that you all know as pediatricians is that children who develop sepsis get sick very, very quickly. We all have memories of children who we saw and may have discharged, and they were dead 12 hours later,” he noted.
Indeed, he and others have shown in multiple studies that up to 50% of pediatric deaths caused by sepsis happen within 24 hours of presentation.
“So whatever happens, it happens very quickly. The true question for us is actually how and why do children progress from no organ dysfunction, where the mortality is close to zero, to organ dysfunction, where all of a sudden mortality jumps up dramatically. It’s this progression that we don’t understand at all,” according to Dr. Schlapbach.
The genetic contribution to fulminant sepsis in children may be substantial
One-third of pediatric sepsis deaths in high-income countries happen in previously healthy children. In a proof-of-concept study, Dr. Schlapbach and coinvestigators in the Swiss Pediatric Sepsis Study Group conducted exome-sequencing genetic studies in eight previously healthy children with no family history of immunodeficiency who died of severe sepsis because of community-acquired Pseudomonas aeruginosa infection. Two of the eight had rare loss-of-function mutations in genes known to cause primary immunodeficiencies. The investigators proposed that unusually severe sepsis in previously healthy children warrants exome sequencing to look for underlying previously undetected primary immunodeficiencies. That’s important information for survivors and/or affected families to have, they argued (Front Immunol. 2016 Sep 20;7:357. eCollection 2016).
“There are some indications that the genetic contribution in children with sepsis may be larger than previously assumed,” he said.
The longstanding practice of fluid bolus therapy for resuscitation in pediatric sepsis is being reexamined
The FEAST (Fluid Expansion As Supportive Therapy) study, a randomized trial of more than 3,000 children with severe febrile illness and impaired perfusion in sub-Saharan Africa, turned heads with its finding that fluid boluses significantly increased 48-hour mortality (BMC Med. 2013 Mar 14;11:67).
Indeed, the FEAST findings, supported by mechanistic animal studies, were sufficiently compelling that the use of fluid boluses in both pediatric and adult septic shock is now under scrutiny in two major randomized trials: RIFTS (the Restrictive IV Fluid Trial in Severe Sepsis and Septic Shock), and CLOVERS (Crystalloid Liberal or Vasopressors Early Resuscitation in Sepsis). Stay tuned.
Dr. Schlapbach reported having no financial conflicts regarding his presentation.
LJUBLJANA, SLOVENIA – The dogma of the “Golden Hour” for the immediate management of pediatric sepsis has been oversold and actually is based upon weak evidence, Luregn J. Schlapbach, MD, asserted at the annual meeting of the European Society for Paediatric Infectious Diseases.
The true Golden Hour – that is, the time frame within which it’s imperative to administer the sepsis bundle comprised of appropriate antibiotics, fluids, and inotropes – is probably more like 3 hours.
“The evidence suggests that up to 3 hours you don’t really have a big difference in outcomes for sepsis. If you recognize shock there’s no question: You should not even wait 1 hour. But if you’re not certain, it may be better to give up to 3 hours to work up the child and get the senior clinician involved before you make decisions about treatment. So I’m not advocating to delay anything, said Dr. Schlapbach, a pediatric intensivist at the Child Health Research Center at the University of Queensland in South Brisbane, Australia.
The problem with a 1-hour mandate for delivery of the sepsis bundle, as recommended in guidelines by the Surviving Sepsis Campaign and the American College of Critical Care Medicine, and endorsed in quality improvement initiatives, is that the time pressure pushes physicians to overprescribe antibiotics to children who don’t actually have a serious bacterial infection. And that, he noted, contributes to the growing problem of antimicrobial resistance.
“You may have a child where you’re not too sure. Usually you would have done a urine culture because UTI [urinary tract infection] is quite a common cause of these infections, and many of these kids aren’t necessarily septic. But if people tell you that within 1 hour you need to treat, are you going to take the time to do the urine culture, or are you just going to decide to treat?” he asked rhetorically.
Dr. Schlapbach is a world-renowned pediatric sepsis researcher. He is far from alone in his reservations about the Golden Hour mandate.
“This is one of the reasons why IDSA [the Infectious Diseases Society of America] has not endorsed the Surviving Sepsis Campaign,” according to the physician, who noted that, in a position statement, IDSA officials have declared that discrimination of sepsis from noninfectious conditions remains a challenge, and that a 60-minute time to antibiotics may jeopardize patient reassessment (Clin Infect Dis. 2018 May 15;66[10]:1631-5).
Dr. Schlapbach highlighted other recent developments in pediatric sepsis.
The definition of adult sepsis has changed, and the pediatric version needs to as well
The revised definition of sepsis, known as Sepsis-3, issued by the International Sepsis Definition Task Force in 2016 notably dropped systemic inflammatory response syndrome (SIRS), as a requirement for sepsis (JAMA. 2016;315[8]:801-10). The revised definition characterizes sepsis as a dysregulated host response to infection resulting in life-threatening organ dysfunction. But Sepsis-3 is based entirely on adult data and is not considered applicable to children.
The current Pediatric Sepsis Consensus Conference definition dates back to 2005. A comprehensive revision is getting underway. It, too, is likely to drop SIRS into the wastebasket, Dr. Schlapbach said.
“It is probably time to abandon the old view of sepsis disease progression, which proposes a progression from infection to SIRS to severe sepsis with organ dysfunction to septic shock, because most children with infection do manifest signs of SIRS, such as tachycardia, tachypnea, and fever, and these probably should be considered as more of an adaptive rather than a maladaptive response,” he explained.
The goal of the pediatric sepsis redefinition project is to come up with something more useful for clinicians than the Sepsis-3 definition. While the Sepsis-3 concept of a dysregulated host response to infection sounds nice, he explained, “we don’t actually know what it is.
“One of the challenges that you all know as pediatricians is that children who develop sepsis get sick very, very quickly. We all have memories of children who we saw and may have discharged, and they were dead 12 hours later,” he noted.
Indeed, he and others have shown in multiple studies that up to 50% of pediatric deaths caused by sepsis happen within 24 hours of presentation.
“So whatever happens, it happens very quickly. The true question for us is actually how and why do children progress from no organ dysfunction, where the mortality is close to zero, to organ dysfunction, where all of a sudden mortality jumps up dramatically. It’s this progression that we don’t understand at all,” according to Dr. Schlapbach.
The genetic contribution to fulminant sepsis in children may be substantial
One-third of pediatric sepsis deaths in high-income countries happen in previously healthy children. In a proof-of-concept study, Dr. Schlapbach and coinvestigators in the Swiss Pediatric Sepsis Study Group conducted exome-sequencing genetic studies in eight previously healthy children with no family history of immunodeficiency who died of severe sepsis because of community-acquired Pseudomonas aeruginosa infection. Two of the eight had rare loss-of-function mutations in genes known to cause primary immunodeficiencies. The investigators proposed that unusually severe sepsis in previously healthy children warrants exome sequencing to look for underlying previously undetected primary immunodeficiencies. That’s important information for survivors and/or affected families to have, they argued (Front Immunol. 2016 Sep 20;7:357. eCollection 2016).
“There are some indications that the genetic contribution in children with sepsis may be larger than previously assumed,” he said.
The longstanding practice of fluid bolus therapy for resuscitation in pediatric sepsis is being reexamined
The FEAST (Fluid Expansion As Supportive Therapy) study, a randomized trial of more than 3,000 children with severe febrile illness and impaired perfusion in sub-Saharan Africa, turned heads with its finding that fluid boluses significantly increased 48-hour mortality (BMC Med. 2013 Mar 14;11:67).
Indeed, the FEAST findings, supported by mechanistic animal studies, were sufficiently compelling that the use of fluid boluses in both pediatric and adult septic shock is now under scrutiny in two major randomized trials: RIFTS (the Restrictive IV Fluid Trial in Severe Sepsis and Septic Shock), and CLOVERS (Crystalloid Liberal or Vasopressors Early Resuscitation in Sepsis). Stay tuned.
Dr. Schlapbach reported having no financial conflicts regarding his presentation.
EXPERT ANALYSIS FROM ESPID 2019















