User login
COVID-19 drives nursing homes to overhaul infection control efforts
The toll that COVID-19 has taken on nursing homes and their postacute and long-term care residents has a multilayered backstory involving underresourced organizational structures, inherent susceptibilities, minimally trained infection prevention staff, variable abilities to isolate and quarantine large numbers of patients and residents, and a lack of governmental support.
“Nursing homes have been trying their best to combat this pandemic using the best infection control procedures they have, but blindfolded and with their hands tied behind their backs,” said Joseph G. Ouslander, MD, professor of geriatric medicine at Florida Atlantic University, Boca Raton, which has teaching affiliations with three senior communities.
Nursing home leaders are debating how to best use testing to guide transmission-based precautions and isolation strategies and how to keep residents safe while allowing some socialization after months of conflicting guidance from public health officials (on testing and on sites of care for patients discharged from the hospital, for instance), with a lack of adequate personal protective equipment (PPE) and testing supplies, and with nursing home resident deaths estimated to account for at least one-quarter of the total COVID-19–related mortality in the United States.
“COVID is not going away [over the next couple of years],” said Michael Wasserman, MD, medical director of the Eisenberg Village at the Los Angeles Jewish Home and president of the California Association of Long-Term Care Medicine.
Dr. Wasserman and other experts in both long-term care and infectious disease said in interviews that, through the rest of the pandemic and beyond, nursing homes need the following:
- Full-time, well-trained “infection preventionists” – infection prevention managers, in essence – who can lead improvements in emergency preparedness and infection prevention and control (IPC)
- Medical directors who are well qualified and engaged
- A survey/inspection process that is educational and not solely punitive
- More resources and attention to structural reform
“If this pandemic doesn’t create significant change in the nursing home industry, nothing ever will,” Dr. Wasserman said.
Prepandemic experience
When Ghinwa Dumyati, MD, began working with nursing homes in early March to prevent and contain COVID-19 outbreaks, her focus was on PPE.
Nursing home staff were intimately familiar with standard precautions, and many had used contact precautions to prevent transmission of infections like Clostridioides difficile and Candida auris, as well as droplet precautions for influenza. With the threat of COVID-19, nursing homes “had a brand-new requirement to do both contact and droplet precautions – with a new need for eye protection – and in some situations, respiratory precautions with N95 masks,” said Dr. Dumyati, professor of medicine and director of communicable disease surveillance and prevention at the University of Rochester (N.Y.) Medical Center. “And on top of that, [staff] had to learn to conserve and reuse PPE.”
Staff had not been fit-tested for use of N95 respirators, she noted. “The only time an N95 was used in the nursing home prior to COVID-19,” she said, “was for a suspected tuberculosis patient [before hospital admission].”
Similarly, nursing homes had experience in quarantining units to prevent transmission of illnesses like influenza or norovirus – keeping residents in their rooms with no visitations or social activity, for instance – but never did they have to arrange “massive movements of residents to completely new units or parts of a unit,” said Dr. Dumyati, who also has led hospital and nursing home collaborative programs in Rochester to beat back C. difficile, and is now helping to formulate COVID-19 recommendations and guidance for members of AMDA – The Society for Post-Acute and Long-Term Care.
As the SARS-CoV-2 virus began its spread through the United States, efforts to strengthen IPC programs in nursing homes in Rochester and elsewhere had been focused largely on multidrug resistant organisms (MDROs) and antibiotic stewardship – not on pandemic preparedness.
Reducing antibiotic use had become a national priority, and a 2016 rule by the Centers for Medicare & Medicaid Services required nursing homes to develop, over a 3-year period, an IPC program that included an antibiotic stewardship component and employment of a trained infection preventionist on at least a half-time basis. Emergency preparedness (e.g., having alternate energy sources for a facility) was also included in the rule, but it was only in 2019 when CMS updated its “Requirements for Participation” rule to stipulate that emergency preparedness include planning for “emerging infectious diseases.”
“The 2016 regulations came about because infections were so problematic in nursing homes,” especially urinary tract infections, C. difficile, and drug-resistant infections, said Patricia Stone, PhD, RN, of the Center for Health Policy at the Columbia University School of Nursing, New York, who has published widely on infection prevention and control in nursing homes.
An analysis of IPC practices in 2014 and in 2018 suggests that the IPC-focused rules were helping, mainly with antibiotic stewardship programs but also with respect to some of the practices aimed at outbreak control, such as having policies in place for grouping infected residents together, instructing infected staff to stay home, and quarantining units on which outbreaks occur, Dr. Stone said. Policies for confining residents to rooms were reported by approximately 74% of nursing homes in 2014, and by approximately 87% in 2018, for instance. Overall, nursing homes were “getting better policies in place,” she said. The analysis compared data from two cross-sectional surveys of nursing homes conducted in 2014 and 2018 (945 and 888 facilities, respectively).
Nursing homes “have a long way to go,” however, with respect to the training of infection preventionists, Dr. Stone said. In 2014, her analysis shows, almost 65% of infection preventionists had no specific infection-control training and less than 3% were Certified in Infection Control (CIC) – a credential awarded by the Certification Board of Infection Control & Epidemiology. Of the 35% who had some form of official training, most completed state or local training courses.
The numbers improved slightly in 2018, with 7% of nursing homes reporting their infection preventionists had the highest-level certification, and 44% reporting that their infection preventionists had no specific infection-control training. Research has shown that infection-control training of any kind has a “strong effect” on IPC-related outcomes. While not demonstrated in research thus far, it seems plausible that “facilities with certified [infection preventionists] will have better processes in place,” said Dr. Stone, whose research has documented the need for more monitoring of staff compliance with hand-washing and other IPC procedures.
Infection preventionists in nursing homes typically have been directors of nursing or assistant directors of nursing who fold IPC responsibilities into a multitude of other responsibilities. Before the 2016 rules, some smaller facilities hired off-site consultants to do the job.
CMS upped the ante after several months of COVID-19, recommending in mid-May that nursing homes assign at least one individual with training in infection control “to provide on-site management of the IPC program.” The infection preventionists should be a “full-time role” in facilities that have more than 100 residents, the CMS guidance said. (Prior to the pandemic, CMS issued proposed regulations in 2019 that would modify the time an infection preventionist must devote to a facility from “part time” to “sufficient time.”)
However, neither the 2016 rule nor the most recent guidance on infection preventionists define the length or content of training.
Swati Gaur, MD, chair of the Infection Advisory Committee of AMDA and a certified medical director of two skilled nursing facilities in Gainesville, Ga., said that the pandemic “has really started to crystallize some of the limitations of having a very vague role, not just in terms of what an [infection preventionists] does [in the nursing home] but also the training,”
Fortunately, Dr. Gaur said, when SARS-CoV-2 struck, she had just transitioned her facilities’ designated infection preventionist to work full-time on the role. She had worked closely with her infection preventionist on IPC issues but wishes she had arranged for more rigorous independent training. “The role of the [infection preventionist] is huge and complicated,” now involving employee health, contract tracing, cohorting, isolation, and compliance with precautions and use of PPE, in addition to surveillance, data reporting, and communication with public health officials, she said.
“Facilities are finding out now that [the infection preventionist] cannot be an afterthought. And it won’t end with COVID. We have other respiratory illnesses like flu and other viruses that we struggle with all the time,” said Dr. Gaur, who is working alongside Dr. Dumyati and two other long-term care experts on AMDA’s COVID-19 guidance. The nursing homes that Dr. Gaur directs are part of the Northeast Georgia Health Care System and together include 271 beds.
Moving forward
IPC practices often collide with facilities’ role as a home, especially to those receiving long-term care. “We always have to measure what we do [to prevent and control infections] against patient autonomy and residents’ rights,” said Dr. Gaur. “We have struggled with these issues, prior to the pandemic. If patients are positive for multidrug resistant organisms [for instance], how long can they be isolated in their own rooms? You can’t for days and months put someone in a single room and create isolation. That’s where the science of infection prevention can collide with residents’ rights.”
Over the years, the Centers for Disease Control and Prevention has acknowledged this discordance, leaving it to facilities to decide, for instance, whether to actively screen for colonization with MDROs. In 2019, to help nursing homes prevent the transmission of MDROs from residents who are colonized but not actively infected, the CDC introduced new “enhanced barrier precautions” that require the use of gowns and gloves for specific resident activities identified as having a high risk of MDRO transmission. The new category of precautions is less restrictive than traditional contact precautions, which keep residents in their rooms.
Infection control in nursing homes “isn’t where it needs to be ... but we’re always going to have in nursing homes a situation where there’s a high potential for rapid transmission of infectious disease,” said Christopher Crnich, MD, PhD, an infectious disease specialist at the University of Wisconsin–Madison who chairs the long-term care special interest group of the Society of Healthcare Epidemiology of America and has offered COVID-19 advice to his state’s department of public health.
“Anytime you have a congregative community, particularly one that involves susceptible hosts, there will be an intrinsically susceptible environment ... I’m a bit disturbed by the emphasis on saying, ‘This nursing home had a COVID-19 outbreak, therefore this nursing home did something wrong,’ ” Dr. Crnich said.
“How we mitigate the size of the outbreaks is where we need to focus our attention,” he said. The goal with SARS-CoV-2, he said, is to recognize its introduction “as rapidly as possible” and stop its spread through empiric symptom- and exposure-based isolation, multiple waves of targeted testing, widespread use of contact and droplet precautions, and isolating staff as necessary.
As awareness grew this year among long-term care leaders that relying too heavily on symptom-based strategies may not be effective to prevent introduction and transmission of SARS-CoV-2, a study published in April in the New England Journal of Medicine cemented the need for a testing strategy not limited to symptomatic individuals.
The study documented that more than half of residents in a nursing home who had positive polymerase chain reaction (PCR) test results were asymptomatic at the time of testing, and that most went on to develop symptoms. The study was conducted after one case of COVID-19 had been identified.
Some states issued calls this spring for “universal testing” of all nursing home patients and staff, and the CMS recommendations issued to state and local officials in mid-May for phased nursing home “reopening” call for baseline testing of all residents and staff, followed by retesting all residents weekly until all residents test negative and by retesting all staff continuing every week.
However, the experts contacted for this story said that, without a highly accurate and accessible point-of-care test (and even with one, considering the virus’ incubation period), a universal approach that includes all nursing home residents may have more limited value than is being touted. In many scenarios, they said, it is most meaningful to focus still-limited testing supplies on the staff, many of whom work at more than one facility and are believed to be primary vectors of SARS-CoV-2.
Dr. Ouslander, Dr. Wasserman and other long-term care leaders have been discussing testing at length, trying to reach consensus on best policies. “I don’t think there’s any uniform approach or uniform agreement,” said Dr. Ouslander. “For me, under ideal circumstances what needs to be done to protect older people in nursing homes is to get access to as many accurate viral tests as possible and test staff at least once a week or every 10 days.”
In some facilities, there may be an unspoken barrier to the frequent testing of staff: Fear that staff who test positive will need to be quarantined, with no one to take their place on the front line. Dr. Ouslander said he knows of one county health department that has discouraged nursing homes from testing asymptomatic staff. “It’s insane and truly shocking,” he said.
At the University of Rochester Medical Center, Dr. Dumyati said, staffing agencies are running short of nurse aide substitutes, and staffing issues have become the “biggest challenge” facing a regional multidisciplinary group of medical directors, hospital leaders, and health department officials who are working to troubleshoot COVID-19 issues. “Some of our nursing homes have ended up sending some of their residents to other nursing homes or to the hospital [because of the loss of staff],” she said.
Currently in the state of New York, she noted, COVID-19 patients may not be discharged to nursing homes until they test negative for the virus through PCR testing. “And some people don’t clear by PCR for 4-6 weeks.”
The barriers
Staffing shortages – real in some locales, and anticipated in others as economic reopening grows – are reflective of underlying structural and financial factors that work against optimal IPC, experts said. It’s not uncommon for certified nurse assistants (CNAs) to be assigned to 10-15 residents. And according to AMDA, 30%-46% of CNAs are reported to receive some form of public assistance. Low wages force many CNAs to work other jobs, including shifts at other nursing homes.
Turnover of nursing home leadership also creates problems. Dr. Crnich calls it “one of the biggest barriers” to effective IPC in nursing homes. “Facilities can tolerate some turnover in their front line staff,” he said, “as long as their leadership structure remains relatively stable.” Dr. Stone and her coinvestigators have documented at least yearly turnover in top positions: They found that, in 2018, approximately one-quarter of facilities reported employing three or more infection preventionists, three or more administrators, and three or more directors of nursing during the prior 3 years.
Medical directors, moreover, are not uniformly qualified, engaged with their facilities, or supported by nursing home administrators. “It’s an open secret, I think, that a lot of facilities want a medical director who is a good referral source,” said Dr. Gaur. “A medical director needs to be completely engaged in [quality improvement and] infection control practices.”
Some nursing home chains, she noted, “have realized the value of the medical director, and have changed the way they’re paying them. They’re actually holding them accountable [for quality and outcomes].”
Medical directors such as Dr. Wasserman, who previously oversaw a 74-facility nursing home chain in California as chief medical officer and then chief executive officer and has worked on nursing home quality improvement processes for his state, said there is much that can be done clinically to prevent the spread of infections, such as more frequent use of telemedicine, more attention to “deprescribing” unnecessary medications (which reduces the number of medication passes and, thus, the number of “transmission opportunities”), and the use of continuous remote monitoring. He has been trying to secure Bluetooth-enabled pulse oximetry and temperature monitoring for the Los Angeles Jewish Home and other facilities.
Dr. Wasserman and other long-term care leaders believe that a more educational inspection process would also lead to improvements in IPC. “The punitive nature of the survey process is morally deflating to frontline staff [and] penalties take money away from operations,” Dr. Wasserman said. “It’s not a productive approach to quality improvement.”
Dr. Stone agreed. Infection control is now the primary focus of CMS’s inspection process, and she said that increased regulatory scrutiny of IPC beyond COVID-19 is a “good thing.” Her research has shown that most deficiencies identified by inspectors are infection control deficiencies, and that in 2014 and 2018, approximately one-third of nursing homes had infection control citations. (CMS recently increased penalties and fines for identified deficiencies.)
“But my hope would be that the survey process would be more educational [as it is for hospitals],” she said. “We need to be supporting nursing homes to do a better job.”
A silver lining of the COVID-19 pandemic, as Dr. Stone sees it, is that nursing homes may be more engaged with data reporting and infection surveillance going forward. Nursing homes are now required to report their COVID-19 cases to the CDC through its hospital-dominant National Healthcare Safety Network, and the CDC has made technical changes that now make it “easier [than it was in the past] for nursing homes to join and participate,” she said. “Now that all nursing homes are engaged, will they be engaged post-COVID, too? I hope so. Surveillance [of infections] is a first step toward better outcomes.”
For now, said Dr. Crnich, the intensive prevention and mitigation efforts that are being required of nursing homes to minimize COVID-19’s impact is “a big deal and will tax the resources of most nursing homes and exceed the resources of many” without outside support, Dr. Crnich said. “This has been the most illuminating part of all this, and will probably require us to reconsider how we’re resourcing our nursing homes moving forward into the future.”
The toll that COVID-19 has taken on nursing homes and their postacute and long-term care residents has a multilayered backstory involving underresourced organizational structures, inherent susceptibilities, minimally trained infection prevention staff, variable abilities to isolate and quarantine large numbers of patients and residents, and a lack of governmental support.
“Nursing homes have been trying their best to combat this pandemic using the best infection control procedures they have, but blindfolded and with their hands tied behind their backs,” said Joseph G. Ouslander, MD, professor of geriatric medicine at Florida Atlantic University, Boca Raton, which has teaching affiliations with three senior communities.
Nursing home leaders are debating how to best use testing to guide transmission-based precautions and isolation strategies and how to keep residents safe while allowing some socialization after months of conflicting guidance from public health officials (on testing and on sites of care for patients discharged from the hospital, for instance), with a lack of adequate personal protective equipment (PPE) and testing supplies, and with nursing home resident deaths estimated to account for at least one-quarter of the total COVID-19–related mortality in the United States.
“COVID is not going away [over the next couple of years],” said Michael Wasserman, MD, medical director of the Eisenberg Village at the Los Angeles Jewish Home and president of the California Association of Long-Term Care Medicine.
Dr. Wasserman and other experts in both long-term care and infectious disease said in interviews that, through the rest of the pandemic and beyond, nursing homes need the following:
- Full-time, well-trained “infection preventionists” – infection prevention managers, in essence – who can lead improvements in emergency preparedness and infection prevention and control (IPC)
- Medical directors who are well qualified and engaged
- A survey/inspection process that is educational and not solely punitive
- More resources and attention to structural reform
“If this pandemic doesn’t create significant change in the nursing home industry, nothing ever will,” Dr. Wasserman said.
Prepandemic experience
When Ghinwa Dumyati, MD, began working with nursing homes in early March to prevent and contain COVID-19 outbreaks, her focus was on PPE.
Nursing home staff were intimately familiar with standard precautions, and many had used contact precautions to prevent transmission of infections like Clostridioides difficile and Candida auris, as well as droplet precautions for influenza. With the threat of COVID-19, nursing homes “had a brand-new requirement to do both contact and droplet precautions – with a new need for eye protection – and in some situations, respiratory precautions with N95 masks,” said Dr. Dumyati, professor of medicine and director of communicable disease surveillance and prevention at the University of Rochester (N.Y.) Medical Center. “And on top of that, [staff] had to learn to conserve and reuse PPE.”
Staff had not been fit-tested for use of N95 respirators, she noted. “The only time an N95 was used in the nursing home prior to COVID-19,” she said, “was for a suspected tuberculosis patient [before hospital admission].”
Similarly, nursing homes had experience in quarantining units to prevent transmission of illnesses like influenza or norovirus – keeping residents in their rooms with no visitations or social activity, for instance – but never did they have to arrange “massive movements of residents to completely new units or parts of a unit,” said Dr. Dumyati, who also has led hospital and nursing home collaborative programs in Rochester to beat back C. difficile, and is now helping to formulate COVID-19 recommendations and guidance for members of AMDA – The Society for Post-Acute and Long-Term Care.
As the SARS-CoV-2 virus began its spread through the United States, efforts to strengthen IPC programs in nursing homes in Rochester and elsewhere had been focused largely on multidrug resistant organisms (MDROs) and antibiotic stewardship – not on pandemic preparedness.
Reducing antibiotic use had become a national priority, and a 2016 rule by the Centers for Medicare & Medicaid Services required nursing homes to develop, over a 3-year period, an IPC program that included an antibiotic stewardship component and employment of a trained infection preventionist on at least a half-time basis. Emergency preparedness (e.g., having alternate energy sources for a facility) was also included in the rule, but it was only in 2019 when CMS updated its “Requirements for Participation” rule to stipulate that emergency preparedness include planning for “emerging infectious diseases.”
“The 2016 regulations came about because infections were so problematic in nursing homes,” especially urinary tract infections, C. difficile, and drug-resistant infections, said Patricia Stone, PhD, RN, of the Center for Health Policy at the Columbia University School of Nursing, New York, who has published widely on infection prevention and control in nursing homes.
An analysis of IPC practices in 2014 and in 2018 suggests that the IPC-focused rules were helping, mainly with antibiotic stewardship programs but also with respect to some of the practices aimed at outbreak control, such as having policies in place for grouping infected residents together, instructing infected staff to stay home, and quarantining units on which outbreaks occur, Dr. Stone said. Policies for confining residents to rooms were reported by approximately 74% of nursing homes in 2014, and by approximately 87% in 2018, for instance. Overall, nursing homes were “getting better policies in place,” she said. The analysis compared data from two cross-sectional surveys of nursing homes conducted in 2014 and 2018 (945 and 888 facilities, respectively).
Nursing homes “have a long way to go,” however, with respect to the training of infection preventionists, Dr. Stone said. In 2014, her analysis shows, almost 65% of infection preventionists had no specific infection-control training and less than 3% were Certified in Infection Control (CIC) – a credential awarded by the Certification Board of Infection Control & Epidemiology. Of the 35% who had some form of official training, most completed state or local training courses.
The numbers improved slightly in 2018, with 7% of nursing homes reporting their infection preventionists had the highest-level certification, and 44% reporting that their infection preventionists had no specific infection-control training. Research has shown that infection-control training of any kind has a “strong effect” on IPC-related outcomes. While not demonstrated in research thus far, it seems plausible that “facilities with certified [infection preventionists] will have better processes in place,” said Dr. Stone, whose research has documented the need for more monitoring of staff compliance with hand-washing and other IPC procedures.
Infection preventionists in nursing homes typically have been directors of nursing or assistant directors of nursing who fold IPC responsibilities into a multitude of other responsibilities. Before the 2016 rules, some smaller facilities hired off-site consultants to do the job.
CMS upped the ante after several months of COVID-19, recommending in mid-May that nursing homes assign at least one individual with training in infection control “to provide on-site management of the IPC program.” The infection preventionists should be a “full-time role” in facilities that have more than 100 residents, the CMS guidance said. (Prior to the pandemic, CMS issued proposed regulations in 2019 that would modify the time an infection preventionist must devote to a facility from “part time” to “sufficient time.”)
However, neither the 2016 rule nor the most recent guidance on infection preventionists define the length or content of training.
Swati Gaur, MD, chair of the Infection Advisory Committee of AMDA and a certified medical director of two skilled nursing facilities in Gainesville, Ga., said that the pandemic “has really started to crystallize some of the limitations of having a very vague role, not just in terms of what an [infection preventionists] does [in the nursing home] but also the training,”
Fortunately, Dr. Gaur said, when SARS-CoV-2 struck, she had just transitioned her facilities’ designated infection preventionist to work full-time on the role. She had worked closely with her infection preventionist on IPC issues but wishes she had arranged for more rigorous independent training. “The role of the [infection preventionist] is huge and complicated,” now involving employee health, contract tracing, cohorting, isolation, and compliance with precautions and use of PPE, in addition to surveillance, data reporting, and communication with public health officials, she said.
“Facilities are finding out now that [the infection preventionist] cannot be an afterthought. And it won’t end with COVID. We have other respiratory illnesses like flu and other viruses that we struggle with all the time,” said Dr. Gaur, who is working alongside Dr. Dumyati and two other long-term care experts on AMDA’s COVID-19 guidance. The nursing homes that Dr. Gaur directs are part of the Northeast Georgia Health Care System and together include 271 beds.
Moving forward
IPC practices often collide with facilities’ role as a home, especially to those receiving long-term care. “We always have to measure what we do [to prevent and control infections] against patient autonomy and residents’ rights,” said Dr. Gaur. “We have struggled with these issues, prior to the pandemic. If patients are positive for multidrug resistant organisms [for instance], how long can they be isolated in their own rooms? You can’t for days and months put someone in a single room and create isolation. That’s where the science of infection prevention can collide with residents’ rights.”
Over the years, the Centers for Disease Control and Prevention has acknowledged this discordance, leaving it to facilities to decide, for instance, whether to actively screen for colonization with MDROs. In 2019, to help nursing homes prevent the transmission of MDROs from residents who are colonized but not actively infected, the CDC introduced new “enhanced barrier precautions” that require the use of gowns and gloves for specific resident activities identified as having a high risk of MDRO transmission. The new category of precautions is less restrictive than traditional contact precautions, which keep residents in their rooms.
Infection control in nursing homes “isn’t where it needs to be ... but we’re always going to have in nursing homes a situation where there’s a high potential for rapid transmission of infectious disease,” said Christopher Crnich, MD, PhD, an infectious disease specialist at the University of Wisconsin–Madison who chairs the long-term care special interest group of the Society of Healthcare Epidemiology of America and has offered COVID-19 advice to his state’s department of public health.
“Anytime you have a congregative community, particularly one that involves susceptible hosts, there will be an intrinsically susceptible environment ... I’m a bit disturbed by the emphasis on saying, ‘This nursing home had a COVID-19 outbreak, therefore this nursing home did something wrong,’ ” Dr. Crnich said.
“How we mitigate the size of the outbreaks is where we need to focus our attention,” he said. The goal with SARS-CoV-2, he said, is to recognize its introduction “as rapidly as possible” and stop its spread through empiric symptom- and exposure-based isolation, multiple waves of targeted testing, widespread use of contact and droplet precautions, and isolating staff as necessary.
As awareness grew this year among long-term care leaders that relying too heavily on symptom-based strategies may not be effective to prevent introduction and transmission of SARS-CoV-2, a study published in April in the New England Journal of Medicine cemented the need for a testing strategy not limited to symptomatic individuals.
The study documented that more than half of residents in a nursing home who had positive polymerase chain reaction (PCR) test results were asymptomatic at the time of testing, and that most went on to develop symptoms. The study was conducted after one case of COVID-19 had been identified.
Some states issued calls this spring for “universal testing” of all nursing home patients and staff, and the CMS recommendations issued to state and local officials in mid-May for phased nursing home “reopening” call for baseline testing of all residents and staff, followed by retesting all residents weekly until all residents test negative and by retesting all staff continuing every week.
However, the experts contacted for this story said that, without a highly accurate and accessible point-of-care test (and even with one, considering the virus’ incubation period), a universal approach that includes all nursing home residents may have more limited value than is being touted. In many scenarios, they said, it is most meaningful to focus still-limited testing supplies on the staff, many of whom work at more than one facility and are believed to be primary vectors of SARS-CoV-2.
Dr. Ouslander, Dr. Wasserman and other long-term care leaders have been discussing testing at length, trying to reach consensus on best policies. “I don’t think there’s any uniform approach or uniform agreement,” said Dr. Ouslander. “For me, under ideal circumstances what needs to be done to protect older people in nursing homes is to get access to as many accurate viral tests as possible and test staff at least once a week or every 10 days.”
In some facilities, there may be an unspoken barrier to the frequent testing of staff: Fear that staff who test positive will need to be quarantined, with no one to take their place on the front line. Dr. Ouslander said he knows of one county health department that has discouraged nursing homes from testing asymptomatic staff. “It’s insane and truly shocking,” he said.
At the University of Rochester Medical Center, Dr. Dumyati said, staffing agencies are running short of nurse aide substitutes, and staffing issues have become the “biggest challenge” facing a regional multidisciplinary group of medical directors, hospital leaders, and health department officials who are working to troubleshoot COVID-19 issues. “Some of our nursing homes have ended up sending some of their residents to other nursing homes or to the hospital [because of the loss of staff],” she said.
Currently in the state of New York, she noted, COVID-19 patients may not be discharged to nursing homes until they test negative for the virus through PCR testing. “And some people don’t clear by PCR for 4-6 weeks.”
The barriers
Staffing shortages – real in some locales, and anticipated in others as economic reopening grows – are reflective of underlying structural and financial factors that work against optimal IPC, experts said. It’s not uncommon for certified nurse assistants (CNAs) to be assigned to 10-15 residents. And according to AMDA, 30%-46% of CNAs are reported to receive some form of public assistance. Low wages force many CNAs to work other jobs, including shifts at other nursing homes.
Turnover of nursing home leadership also creates problems. Dr. Crnich calls it “one of the biggest barriers” to effective IPC in nursing homes. “Facilities can tolerate some turnover in their front line staff,” he said, “as long as their leadership structure remains relatively stable.” Dr. Stone and her coinvestigators have documented at least yearly turnover in top positions: They found that, in 2018, approximately one-quarter of facilities reported employing three or more infection preventionists, three or more administrators, and three or more directors of nursing during the prior 3 years.
Medical directors, moreover, are not uniformly qualified, engaged with their facilities, or supported by nursing home administrators. “It’s an open secret, I think, that a lot of facilities want a medical director who is a good referral source,” said Dr. Gaur. “A medical director needs to be completely engaged in [quality improvement and] infection control practices.”
Some nursing home chains, she noted, “have realized the value of the medical director, and have changed the way they’re paying them. They’re actually holding them accountable [for quality and outcomes].”
Medical directors such as Dr. Wasserman, who previously oversaw a 74-facility nursing home chain in California as chief medical officer and then chief executive officer and has worked on nursing home quality improvement processes for his state, said there is much that can be done clinically to prevent the spread of infections, such as more frequent use of telemedicine, more attention to “deprescribing” unnecessary medications (which reduces the number of medication passes and, thus, the number of “transmission opportunities”), and the use of continuous remote monitoring. He has been trying to secure Bluetooth-enabled pulse oximetry and temperature monitoring for the Los Angeles Jewish Home and other facilities.
Dr. Wasserman and other long-term care leaders believe that a more educational inspection process would also lead to improvements in IPC. “The punitive nature of the survey process is morally deflating to frontline staff [and] penalties take money away from operations,” Dr. Wasserman said. “It’s not a productive approach to quality improvement.”
Dr. Stone agreed. Infection control is now the primary focus of CMS’s inspection process, and she said that increased regulatory scrutiny of IPC beyond COVID-19 is a “good thing.” Her research has shown that most deficiencies identified by inspectors are infection control deficiencies, and that in 2014 and 2018, approximately one-third of nursing homes had infection control citations. (CMS recently increased penalties and fines for identified deficiencies.)
“But my hope would be that the survey process would be more educational [as it is for hospitals],” she said. “We need to be supporting nursing homes to do a better job.”
A silver lining of the COVID-19 pandemic, as Dr. Stone sees it, is that nursing homes may be more engaged with data reporting and infection surveillance going forward. Nursing homes are now required to report their COVID-19 cases to the CDC through its hospital-dominant National Healthcare Safety Network, and the CDC has made technical changes that now make it “easier [than it was in the past] for nursing homes to join and participate,” she said. “Now that all nursing homes are engaged, will they be engaged post-COVID, too? I hope so. Surveillance [of infections] is a first step toward better outcomes.”
For now, said Dr. Crnich, the intensive prevention and mitigation efforts that are being required of nursing homes to minimize COVID-19’s impact is “a big deal and will tax the resources of most nursing homes and exceed the resources of many” without outside support, Dr. Crnich said. “This has been the most illuminating part of all this, and will probably require us to reconsider how we’re resourcing our nursing homes moving forward into the future.”
The toll that COVID-19 has taken on nursing homes and their postacute and long-term care residents has a multilayered backstory involving underresourced organizational structures, inherent susceptibilities, minimally trained infection prevention staff, variable abilities to isolate and quarantine large numbers of patients and residents, and a lack of governmental support.
“Nursing homes have been trying their best to combat this pandemic using the best infection control procedures they have, but blindfolded and with their hands tied behind their backs,” said Joseph G. Ouslander, MD, professor of geriatric medicine at Florida Atlantic University, Boca Raton, which has teaching affiliations with three senior communities.
Nursing home leaders are debating how to best use testing to guide transmission-based precautions and isolation strategies and how to keep residents safe while allowing some socialization after months of conflicting guidance from public health officials (on testing and on sites of care for patients discharged from the hospital, for instance), with a lack of adequate personal protective equipment (PPE) and testing supplies, and with nursing home resident deaths estimated to account for at least one-quarter of the total COVID-19–related mortality in the United States.
“COVID is not going away [over the next couple of years],” said Michael Wasserman, MD, medical director of the Eisenberg Village at the Los Angeles Jewish Home and president of the California Association of Long-Term Care Medicine.
Dr. Wasserman and other experts in both long-term care and infectious disease said in interviews that, through the rest of the pandemic and beyond, nursing homes need the following:
- Full-time, well-trained “infection preventionists” – infection prevention managers, in essence – who can lead improvements in emergency preparedness and infection prevention and control (IPC)
- Medical directors who are well qualified and engaged
- A survey/inspection process that is educational and not solely punitive
- More resources and attention to structural reform
“If this pandemic doesn’t create significant change in the nursing home industry, nothing ever will,” Dr. Wasserman said.
Prepandemic experience
When Ghinwa Dumyati, MD, began working with nursing homes in early March to prevent and contain COVID-19 outbreaks, her focus was on PPE.
Nursing home staff were intimately familiar with standard precautions, and many had used contact precautions to prevent transmission of infections like Clostridioides difficile and Candida auris, as well as droplet precautions for influenza. With the threat of COVID-19, nursing homes “had a brand-new requirement to do both contact and droplet precautions – with a new need for eye protection – and in some situations, respiratory precautions with N95 masks,” said Dr. Dumyati, professor of medicine and director of communicable disease surveillance and prevention at the University of Rochester (N.Y.) Medical Center. “And on top of that, [staff] had to learn to conserve and reuse PPE.”
Staff had not been fit-tested for use of N95 respirators, she noted. “The only time an N95 was used in the nursing home prior to COVID-19,” she said, “was for a suspected tuberculosis patient [before hospital admission].”
Similarly, nursing homes had experience in quarantining units to prevent transmission of illnesses like influenza or norovirus – keeping residents in their rooms with no visitations or social activity, for instance – but never did they have to arrange “massive movements of residents to completely new units or parts of a unit,” said Dr. Dumyati, who also has led hospital and nursing home collaborative programs in Rochester to beat back C. difficile, and is now helping to formulate COVID-19 recommendations and guidance for members of AMDA – The Society for Post-Acute and Long-Term Care.
As the SARS-CoV-2 virus began its spread through the United States, efforts to strengthen IPC programs in nursing homes in Rochester and elsewhere had been focused largely on multidrug resistant organisms (MDROs) and antibiotic stewardship – not on pandemic preparedness.
Reducing antibiotic use had become a national priority, and a 2016 rule by the Centers for Medicare & Medicaid Services required nursing homes to develop, over a 3-year period, an IPC program that included an antibiotic stewardship component and employment of a trained infection preventionist on at least a half-time basis. Emergency preparedness (e.g., having alternate energy sources for a facility) was also included in the rule, but it was only in 2019 when CMS updated its “Requirements for Participation” rule to stipulate that emergency preparedness include planning for “emerging infectious diseases.”
“The 2016 regulations came about because infections were so problematic in nursing homes,” especially urinary tract infections, C. difficile, and drug-resistant infections, said Patricia Stone, PhD, RN, of the Center for Health Policy at the Columbia University School of Nursing, New York, who has published widely on infection prevention and control in nursing homes.
An analysis of IPC practices in 2014 and in 2018 suggests that the IPC-focused rules were helping, mainly with antibiotic stewardship programs but also with respect to some of the practices aimed at outbreak control, such as having policies in place for grouping infected residents together, instructing infected staff to stay home, and quarantining units on which outbreaks occur, Dr. Stone said. Policies for confining residents to rooms were reported by approximately 74% of nursing homes in 2014, and by approximately 87% in 2018, for instance. Overall, nursing homes were “getting better policies in place,” she said. The analysis compared data from two cross-sectional surveys of nursing homes conducted in 2014 and 2018 (945 and 888 facilities, respectively).
Nursing homes “have a long way to go,” however, with respect to the training of infection preventionists, Dr. Stone said. In 2014, her analysis shows, almost 65% of infection preventionists had no specific infection-control training and less than 3% were Certified in Infection Control (CIC) – a credential awarded by the Certification Board of Infection Control & Epidemiology. Of the 35% who had some form of official training, most completed state or local training courses.
The numbers improved slightly in 2018, with 7% of nursing homes reporting their infection preventionists had the highest-level certification, and 44% reporting that their infection preventionists had no specific infection-control training. Research has shown that infection-control training of any kind has a “strong effect” on IPC-related outcomes. While not demonstrated in research thus far, it seems plausible that “facilities with certified [infection preventionists] will have better processes in place,” said Dr. Stone, whose research has documented the need for more monitoring of staff compliance with hand-washing and other IPC procedures.
Infection preventionists in nursing homes typically have been directors of nursing or assistant directors of nursing who fold IPC responsibilities into a multitude of other responsibilities. Before the 2016 rules, some smaller facilities hired off-site consultants to do the job.
CMS upped the ante after several months of COVID-19, recommending in mid-May that nursing homes assign at least one individual with training in infection control “to provide on-site management of the IPC program.” The infection preventionists should be a “full-time role” in facilities that have more than 100 residents, the CMS guidance said. (Prior to the pandemic, CMS issued proposed regulations in 2019 that would modify the time an infection preventionist must devote to a facility from “part time” to “sufficient time.”)
However, neither the 2016 rule nor the most recent guidance on infection preventionists define the length or content of training.
Swati Gaur, MD, chair of the Infection Advisory Committee of AMDA and a certified medical director of two skilled nursing facilities in Gainesville, Ga., said that the pandemic “has really started to crystallize some of the limitations of having a very vague role, not just in terms of what an [infection preventionists] does [in the nursing home] but also the training,”
Fortunately, Dr. Gaur said, when SARS-CoV-2 struck, she had just transitioned her facilities’ designated infection preventionist to work full-time on the role. She had worked closely with her infection preventionist on IPC issues but wishes she had arranged for more rigorous independent training. “The role of the [infection preventionist] is huge and complicated,” now involving employee health, contract tracing, cohorting, isolation, and compliance with precautions and use of PPE, in addition to surveillance, data reporting, and communication with public health officials, she said.
“Facilities are finding out now that [the infection preventionist] cannot be an afterthought. And it won’t end with COVID. We have other respiratory illnesses like flu and other viruses that we struggle with all the time,” said Dr. Gaur, who is working alongside Dr. Dumyati and two other long-term care experts on AMDA’s COVID-19 guidance. The nursing homes that Dr. Gaur directs are part of the Northeast Georgia Health Care System and together include 271 beds.
Moving forward
IPC practices often collide with facilities’ role as a home, especially to those receiving long-term care. “We always have to measure what we do [to prevent and control infections] against patient autonomy and residents’ rights,” said Dr. Gaur. “We have struggled with these issues, prior to the pandemic. If patients are positive for multidrug resistant organisms [for instance], how long can they be isolated in their own rooms? You can’t for days and months put someone in a single room and create isolation. That’s where the science of infection prevention can collide with residents’ rights.”
Over the years, the Centers for Disease Control and Prevention has acknowledged this discordance, leaving it to facilities to decide, for instance, whether to actively screen for colonization with MDROs. In 2019, to help nursing homes prevent the transmission of MDROs from residents who are colonized but not actively infected, the CDC introduced new “enhanced barrier precautions” that require the use of gowns and gloves for specific resident activities identified as having a high risk of MDRO transmission. The new category of precautions is less restrictive than traditional contact precautions, which keep residents in their rooms.
Infection control in nursing homes “isn’t where it needs to be ... but we’re always going to have in nursing homes a situation where there’s a high potential for rapid transmission of infectious disease,” said Christopher Crnich, MD, PhD, an infectious disease specialist at the University of Wisconsin–Madison who chairs the long-term care special interest group of the Society of Healthcare Epidemiology of America and has offered COVID-19 advice to his state’s department of public health.
“Anytime you have a congregative community, particularly one that involves susceptible hosts, there will be an intrinsically susceptible environment ... I’m a bit disturbed by the emphasis on saying, ‘This nursing home had a COVID-19 outbreak, therefore this nursing home did something wrong,’ ” Dr. Crnich said.
“How we mitigate the size of the outbreaks is where we need to focus our attention,” he said. The goal with SARS-CoV-2, he said, is to recognize its introduction “as rapidly as possible” and stop its spread through empiric symptom- and exposure-based isolation, multiple waves of targeted testing, widespread use of contact and droplet precautions, and isolating staff as necessary.
As awareness grew this year among long-term care leaders that relying too heavily on symptom-based strategies may not be effective to prevent introduction and transmission of SARS-CoV-2, a study published in April in the New England Journal of Medicine cemented the need for a testing strategy not limited to symptomatic individuals.
The study documented that more than half of residents in a nursing home who had positive polymerase chain reaction (PCR) test results were asymptomatic at the time of testing, and that most went on to develop symptoms. The study was conducted after one case of COVID-19 had been identified.
Some states issued calls this spring for “universal testing” of all nursing home patients and staff, and the CMS recommendations issued to state and local officials in mid-May for phased nursing home “reopening” call for baseline testing of all residents and staff, followed by retesting all residents weekly until all residents test negative and by retesting all staff continuing every week.
However, the experts contacted for this story said that, without a highly accurate and accessible point-of-care test (and even with one, considering the virus’ incubation period), a universal approach that includes all nursing home residents may have more limited value than is being touted. In many scenarios, they said, it is most meaningful to focus still-limited testing supplies on the staff, many of whom work at more than one facility and are believed to be primary vectors of SARS-CoV-2.
Dr. Ouslander, Dr. Wasserman and other long-term care leaders have been discussing testing at length, trying to reach consensus on best policies. “I don’t think there’s any uniform approach or uniform agreement,” said Dr. Ouslander. “For me, under ideal circumstances what needs to be done to protect older people in nursing homes is to get access to as many accurate viral tests as possible and test staff at least once a week or every 10 days.”
In some facilities, there may be an unspoken barrier to the frequent testing of staff: Fear that staff who test positive will need to be quarantined, with no one to take their place on the front line. Dr. Ouslander said he knows of one county health department that has discouraged nursing homes from testing asymptomatic staff. “It’s insane and truly shocking,” he said.
At the University of Rochester Medical Center, Dr. Dumyati said, staffing agencies are running short of nurse aide substitutes, and staffing issues have become the “biggest challenge” facing a regional multidisciplinary group of medical directors, hospital leaders, and health department officials who are working to troubleshoot COVID-19 issues. “Some of our nursing homes have ended up sending some of their residents to other nursing homes or to the hospital [because of the loss of staff],” she said.
Currently in the state of New York, she noted, COVID-19 patients may not be discharged to nursing homes until they test negative for the virus through PCR testing. “And some people don’t clear by PCR for 4-6 weeks.”
The barriers
Staffing shortages – real in some locales, and anticipated in others as economic reopening grows – are reflective of underlying structural and financial factors that work against optimal IPC, experts said. It’s not uncommon for certified nurse assistants (CNAs) to be assigned to 10-15 residents. And according to AMDA, 30%-46% of CNAs are reported to receive some form of public assistance. Low wages force many CNAs to work other jobs, including shifts at other nursing homes.
Turnover of nursing home leadership also creates problems. Dr. Crnich calls it “one of the biggest barriers” to effective IPC in nursing homes. “Facilities can tolerate some turnover in their front line staff,” he said, “as long as their leadership structure remains relatively stable.” Dr. Stone and her coinvestigators have documented at least yearly turnover in top positions: They found that, in 2018, approximately one-quarter of facilities reported employing three or more infection preventionists, three or more administrators, and three or more directors of nursing during the prior 3 years.
Medical directors, moreover, are not uniformly qualified, engaged with their facilities, or supported by nursing home administrators. “It’s an open secret, I think, that a lot of facilities want a medical director who is a good referral source,” said Dr. Gaur. “A medical director needs to be completely engaged in [quality improvement and] infection control practices.”
Some nursing home chains, she noted, “have realized the value of the medical director, and have changed the way they’re paying them. They’re actually holding them accountable [for quality and outcomes].”
Medical directors such as Dr. Wasserman, who previously oversaw a 74-facility nursing home chain in California as chief medical officer and then chief executive officer and has worked on nursing home quality improvement processes for his state, said there is much that can be done clinically to prevent the spread of infections, such as more frequent use of telemedicine, more attention to “deprescribing” unnecessary medications (which reduces the number of medication passes and, thus, the number of “transmission opportunities”), and the use of continuous remote monitoring. He has been trying to secure Bluetooth-enabled pulse oximetry and temperature monitoring for the Los Angeles Jewish Home and other facilities.
Dr. Wasserman and other long-term care leaders believe that a more educational inspection process would also lead to improvements in IPC. “The punitive nature of the survey process is morally deflating to frontline staff [and] penalties take money away from operations,” Dr. Wasserman said. “It’s not a productive approach to quality improvement.”
Dr. Stone agreed. Infection control is now the primary focus of CMS’s inspection process, and she said that increased regulatory scrutiny of IPC beyond COVID-19 is a “good thing.” Her research has shown that most deficiencies identified by inspectors are infection control deficiencies, and that in 2014 and 2018, approximately one-third of nursing homes had infection control citations. (CMS recently increased penalties and fines for identified deficiencies.)
“But my hope would be that the survey process would be more educational [as it is for hospitals],” she said. “We need to be supporting nursing homes to do a better job.”
A silver lining of the COVID-19 pandemic, as Dr. Stone sees it, is that nursing homes may be more engaged with data reporting and infection surveillance going forward. Nursing homes are now required to report their COVID-19 cases to the CDC through its hospital-dominant National Healthcare Safety Network, and the CDC has made technical changes that now make it “easier [than it was in the past] for nursing homes to join and participate,” she said. “Now that all nursing homes are engaged, will they be engaged post-COVID, too? I hope so. Surveillance [of infections] is a first step toward better outcomes.”
For now, said Dr. Crnich, the intensive prevention and mitigation efforts that are being required of nursing homes to minimize COVID-19’s impact is “a big deal and will tax the resources of most nursing homes and exceed the resources of many” without outside support, Dr. Crnich said. “This has been the most illuminating part of all this, and will probably require us to reconsider how we’re resourcing our nursing homes moving forward into the future.”
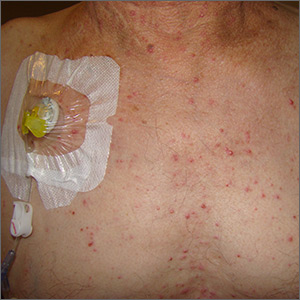
Diffuse pustules
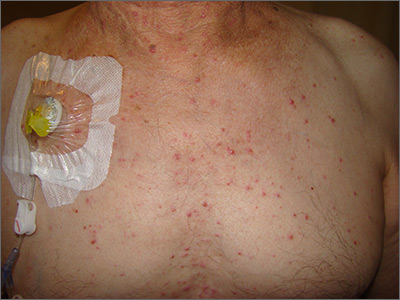
The presence of these sterile pustules with an erythematous base led to a diagnosis of acute generalized exanthematous pustulosis (AGEP), also known as a pustular drug eruption. Although pustules are present, AGEP is an allergic response to medications and not an infection.
AGEP can be associated with fever and leukocytosis. Interestingly, antibiotics are a frequent cause—not a treatment—since the pustules are sterile. It also is worth noting, in light of the COVID-19 pandemic, that hydroxychloroquine use has been linked to AGEP, although the number of cases cited in the literature is small.
Treatment is avoidance of the offending medication and symptomatic care. AGEP typically will resolve approximately 2 weeks after discontinuing the medication causing the reaction. Systemic steroids also may be used for treatment in severe cases.
This patient had a history of repeated episodes with his chemotherapy regimen, so he was treated symptomatically with diphenhydramine for the itching. Since chemotherapy was a priority to treat his colon cancer, avoidance of the offending agent was not an option. The Family Medicine Service recommended pretreatment with diphenhydramine 25 to 50 mg orally or intravenously for future rounds of chemotherapy to blunt future responses.
Photo and text courtesy of Daniel Stulberg, MD, FAAFP, Department of Family and Community Medicine, University of New Mexico School of Medicine, Albuquerque.
Mercogliano C, Khan M, Lin C, et al. AGEP overlap induced by hydroxychloroquine: a case report and literature review. J Community Hosp Intern Med Perspect. 2018;8:360-362.

The presence of these sterile pustules with an erythematous base led to a diagnosis of acute generalized exanthematous pustulosis (AGEP), also known as a pustular drug eruption. Although pustules are present, AGEP is an allergic response to medications and not an infection.
AGEP can be associated with fever and leukocytosis. Interestingly, antibiotics are a frequent cause—not a treatment—since the pustules are sterile. It also is worth noting, in light of the COVID-19 pandemic, that hydroxychloroquine use has been linked to AGEP, although the number of cases cited in the literature is small.
Treatment is avoidance of the offending medication and symptomatic care. AGEP typically will resolve approximately 2 weeks after discontinuing the medication causing the reaction. Systemic steroids also may be used for treatment in severe cases.
This patient had a history of repeated episodes with his chemotherapy regimen, so he was treated symptomatically with diphenhydramine for the itching. Since chemotherapy was a priority to treat his colon cancer, avoidance of the offending agent was not an option. The Family Medicine Service recommended pretreatment with diphenhydramine 25 to 50 mg orally or intravenously for future rounds of chemotherapy to blunt future responses.
Photo and text courtesy of Daniel Stulberg, MD, FAAFP, Department of Family and Community Medicine, University of New Mexico School of Medicine, Albuquerque.

The presence of these sterile pustules with an erythematous base led to a diagnosis of acute generalized exanthematous pustulosis (AGEP), also known as a pustular drug eruption. Although pustules are present, AGEP is an allergic response to medications and not an infection.
AGEP can be associated with fever and leukocytosis. Interestingly, antibiotics are a frequent cause—not a treatment—since the pustules are sterile. It also is worth noting, in light of the COVID-19 pandemic, that hydroxychloroquine use has been linked to AGEP, although the number of cases cited in the literature is small.
Treatment is avoidance of the offending medication and symptomatic care. AGEP typically will resolve approximately 2 weeks after discontinuing the medication causing the reaction. Systemic steroids also may be used for treatment in severe cases.
This patient had a history of repeated episodes with his chemotherapy regimen, so he was treated symptomatically with diphenhydramine for the itching. Since chemotherapy was a priority to treat his colon cancer, avoidance of the offending agent was not an option. The Family Medicine Service recommended pretreatment with diphenhydramine 25 to 50 mg orally or intravenously for future rounds of chemotherapy to blunt future responses.
Photo and text courtesy of Daniel Stulberg, MD, FAAFP, Department of Family and Community Medicine, University of New Mexico School of Medicine, Albuquerque.
Mercogliano C, Khan M, Lin C, et al. AGEP overlap induced by hydroxychloroquine: a case report and literature review. J Community Hosp Intern Med Perspect. 2018;8:360-362.
Mercogliano C, Khan M, Lin C, et al. AGEP overlap induced by hydroxychloroquine: a case report and literature review. J Community Hosp Intern Med Perspect. 2018;8:360-362.
CMS issues interim final rule
On Thursday, April 30, 2020, CMS released a new interim final rule. During the COVID-19 Public Health Emergency, the Interim Final Rule makes several new, important temporary changes to Medicare regulations and payments. One important change retroactively (to March 1, 2020) increased payments for telephone-only visits to established patients:
- CPT 99441: a 5- to 10-minute telephone visit, in lieu of a face-to-face office visit, will be reimbursed at a similar rate to a 99212, about $46 (99441 is usually reimbursed at about $14).
- CPT 99442: an 11- to 20-minute telephone visit, in lieu of a face-to-face office visit, will be reimbursed at a similar rate to a 99213, about $76 (99442 is usually reimbursed at about $28).
- CPT 99443: a 21- to 30-minute telephone visit, in lieu of a face-to-face office visit, will be reimbursed at a similar rate to a 99212, about $110 (99443 is usually reimbursed at about $41).
These telephone codes may be used when addressing a new or old problem for established patients. Choose the code to reflect only the billing provider time communicating with the patient. There should not be another patient encounter for 7 calendar days before or after the telephone visit.
In addition, the new Interim Final Rule now allows attending physicians at teaching institutions providing supervision under the Primary Care Exception to report for telephone (using 99441-99443) or video (using 99212-99215) telemedicine encounters by residents, when the supervision is provided immediately after the resident encounter, rather than during the telephone or video visit. However, most chest physicians at teaching institutions do not supervise residents or fellows under the Primary Care Exception.
A CMS press release about the rule is available at cms.gov.
On Thursday, April 30, 2020, CMS released a new interim final rule. During the COVID-19 Public Health Emergency, the Interim Final Rule makes several new, important temporary changes to Medicare regulations and payments. One important change retroactively (to March 1, 2020) increased payments for telephone-only visits to established patients:
- CPT 99441: a 5- to 10-minute telephone visit, in lieu of a face-to-face office visit, will be reimbursed at a similar rate to a 99212, about $46 (99441 is usually reimbursed at about $14).
- CPT 99442: an 11- to 20-minute telephone visit, in lieu of a face-to-face office visit, will be reimbursed at a similar rate to a 99213, about $76 (99442 is usually reimbursed at about $28).
- CPT 99443: a 21- to 30-minute telephone visit, in lieu of a face-to-face office visit, will be reimbursed at a similar rate to a 99212, about $110 (99443 is usually reimbursed at about $41).
These telephone codes may be used when addressing a new or old problem for established patients. Choose the code to reflect only the billing provider time communicating with the patient. There should not be another patient encounter for 7 calendar days before or after the telephone visit.
In addition, the new Interim Final Rule now allows attending physicians at teaching institutions providing supervision under the Primary Care Exception to report for telephone (using 99441-99443) or video (using 99212-99215) telemedicine encounters by residents, when the supervision is provided immediately after the resident encounter, rather than during the telephone or video visit. However, most chest physicians at teaching institutions do not supervise residents or fellows under the Primary Care Exception.
A CMS press release about the rule is available at cms.gov.
On Thursday, April 30, 2020, CMS released a new interim final rule. During the COVID-19 Public Health Emergency, the Interim Final Rule makes several new, important temporary changes to Medicare regulations and payments. One important change retroactively (to March 1, 2020) increased payments for telephone-only visits to established patients:
- CPT 99441: a 5- to 10-minute telephone visit, in lieu of a face-to-face office visit, will be reimbursed at a similar rate to a 99212, about $46 (99441 is usually reimbursed at about $14).
- CPT 99442: an 11- to 20-minute telephone visit, in lieu of a face-to-face office visit, will be reimbursed at a similar rate to a 99213, about $76 (99442 is usually reimbursed at about $28).
- CPT 99443: a 21- to 30-minute telephone visit, in lieu of a face-to-face office visit, will be reimbursed at a similar rate to a 99212, about $110 (99443 is usually reimbursed at about $41).
These telephone codes may be used when addressing a new or old problem for established patients. Choose the code to reflect only the billing provider time communicating with the patient. There should not be another patient encounter for 7 calendar days before or after the telephone visit.
In addition, the new Interim Final Rule now allows attending physicians at teaching institutions providing supervision under the Primary Care Exception to report for telephone (using 99441-99443) or video (using 99212-99215) telemedicine encounters by residents, when the supervision is provided immediately after the resident encounter, rather than during the telephone or video visit. However, most chest physicians at teaching institutions do not supervise residents or fellows under the Primary Care Exception.
A CMS press release about the rule is available at cms.gov.
Your CHEST Foundation: Supporting communities during COVID-2019
The entire world has been affected by the COVID-19 crisis, yet many of our most vulnerable continue to suffer in silence. The CHEST Foundation is diligently working to help give voice to these all-too-often isolated and forgotten patients. Make a donation today, and help those who need it most: our family, friends, neighbors, and those most vulnerable to this devastating disease.
In addition to providing reliable and educational resources that address COVID-19 for both clinicians and patients, the CHEST Foundation is:
- Launching a series of public service announcement videos to empower patients and caregivers living with COPD and interstitial lung disease by providing information on necessary skills, such as cleaning medical equipment, and helping them stay safe and healthy while coping with isolation;
- Partnering with AMITA Health in Chicago to bring telehealth opportunities to patients and support groups; and
- Providing grant funding, in partnership with the Feldman Family Foundation, that supports projects such as providing supplies and groceries to patients and caregivers, expediting training and the means to get caregivers to NYC, and providing needed technology to continue hosting support group meetings in local communities.
The CHEST Foundation has rebranded and relaunched its website in an effort to make it more user-friendly, patient-focused, and clinician-centered. We’ve upgraded our current content, written new pieces, and carefully curated a complete collection of tools that will help patients, caregivers, and clinicians better navigate the complexities of lung disease. Information on all of the content previously listed will be available on the CHEST Foundation’s website at chestfoundation.org.
Thank you for helping as we fulfill the urgent needs of our community during this crisis. Help support your community by making a donation today.
The entire world has been affected by the COVID-19 crisis, yet many of our most vulnerable continue to suffer in silence. The CHEST Foundation is diligently working to help give voice to these all-too-often isolated and forgotten patients. Make a donation today, and help those who need it most: our family, friends, neighbors, and those most vulnerable to this devastating disease.
In addition to providing reliable and educational resources that address COVID-19 for both clinicians and patients, the CHEST Foundation is:
- Launching a series of public service announcement videos to empower patients and caregivers living with COPD and interstitial lung disease by providing information on necessary skills, such as cleaning medical equipment, and helping them stay safe and healthy while coping with isolation;
- Partnering with AMITA Health in Chicago to bring telehealth opportunities to patients and support groups; and
- Providing grant funding, in partnership with the Feldman Family Foundation, that supports projects such as providing supplies and groceries to patients and caregivers, expediting training and the means to get caregivers to NYC, and providing needed technology to continue hosting support group meetings in local communities.
The CHEST Foundation has rebranded and relaunched its website in an effort to make it more user-friendly, patient-focused, and clinician-centered. We’ve upgraded our current content, written new pieces, and carefully curated a complete collection of tools that will help patients, caregivers, and clinicians better navigate the complexities of lung disease. Information on all of the content previously listed will be available on the CHEST Foundation’s website at chestfoundation.org.
Thank you for helping as we fulfill the urgent needs of our community during this crisis. Help support your community by making a donation today.
The entire world has been affected by the COVID-19 crisis, yet many of our most vulnerable continue to suffer in silence. The CHEST Foundation is diligently working to help give voice to these all-too-often isolated and forgotten patients. Make a donation today, and help those who need it most: our family, friends, neighbors, and those most vulnerable to this devastating disease.
In addition to providing reliable and educational resources that address COVID-19 for both clinicians and patients, the CHEST Foundation is:
- Launching a series of public service announcement videos to empower patients and caregivers living with COPD and interstitial lung disease by providing information on necessary skills, such as cleaning medical equipment, and helping them stay safe and healthy while coping with isolation;
- Partnering with AMITA Health in Chicago to bring telehealth opportunities to patients and support groups; and
- Providing grant funding, in partnership with the Feldman Family Foundation, that supports projects such as providing supplies and groceries to patients and caregivers, expediting training and the means to get caregivers to NYC, and providing needed technology to continue hosting support group meetings in local communities.
The CHEST Foundation has rebranded and relaunched its website in an effort to make it more user-friendly, patient-focused, and clinician-centered. We’ve upgraded our current content, written new pieces, and carefully curated a complete collection of tools that will help patients, caregivers, and clinicians better navigate the complexities of lung disease. Information on all of the content previously listed will be available on the CHEST Foundation’s website at chestfoundation.org.
Thank you for helping as we fulfill the urgent needs of our community during this crisis. Help support your community by making a donation today.
Today’s best bet – Get involved with CHEST!
I am often overheard encouraging colleagues to become involved with CHEST. I am a strong believer that you get far more out of participation than you will ever put into it. I have now been fortunate to have many leadership roles within CHEST and currently serve on the Board of Regents and as Chair of the Council of NetWorks. I have been able to work with a growing number of people, including faculty and CHEST staff. The more invested I have become, the more CHEST truly feels like family.
I understand that while it may be easy for me to tell members to get involved, it often feels much more difficult to actually get appointed to a leadership position. Early in my career, I was given the advice, “When you are given a task, make sure you blow it out of the water. That will only open more doors for you.” Making the most of a position on a NetWork or committee can create future opportunities. We recently had self-nominations for leadership positions within the NetWork steering committees and committees at large. Some positions have one to two openings for 20 applications. It can be frustrating not to get a position the first time around. However, it is common for members to have to apply numerous times prior to being appointed. When applying to these positions, be sure to highlight any prior CHEST involvement, as this may weigh in on an appointment to specific positions. Some of the decisions to appoint a nominee are based on prior engagement with CHEST.
So how can one get involved without holding a leadership position? My first piece of advice is to ensure you are getting CHEST emails. Check them regularly to so that you do not miss any opportunities. Next, be a member of at least one NetWork that is of interest to you. The NetWorks provide a smaller community within CHEST for special interests within our field. You will get emailed updates throughout the year that include any projects in which input is needed. At the CHEST annual meeting, each NetWork holds an Open Forum that functions as their annual face-to-face business meeting. These meetings are open to everyone. This is an excellent way to meet the current steering committee members and become involved in plans for the upcoming year. This year, we have made the dates and times of the NetWork steering committee calls public on the CHEST website. Any NetWork member can join these calls, even if they are not officially on the steering committee. All ongoing projects are discussed on these calls, so participation on the call offers an excellent opportunity to volunteer. You can also get involved with the NetWorks on social media by using the appropriate NetWork hashtags, along with tagging @accpchest to communicate with your NetWork colleagues.

Finally, the easiest way to embrace CHEST, and possibly the most obvious, is to get involved with the CHEST annual meeting. The meeting is at its best when planned and orchestrated by a diverse group of people. Annual meeting planning usually starts in November or December of the prior year. Submitting a proposal for a session at the annual meeting is strongly encouraged. Tips for how to submit a strong, well-rounded session are offered on the submission website. Reviewing these tips first can help strengthen your proposal. An easy way to become involved, even as a student or as a trainee, is to submit an abstract to the annual meeting
Summing up, I would encourage everyone to simply be an active participant: raise your hand to ask questions, introduce yourself to those around you, and attend the social events at CHEST annual meeting. Before you know it, new friends will become old friends, and attending the CHEST annual meeting will start to feel like going to a family reunion.
I am often overheard encouraging colleagues to become involved with CHEST. I am a strong believer that you get far more out of participation than you will ever put into it. I have now been fortunate to have many leadership roles within CHEST and currently serve on the Board of Regents and as Chair of the Council of NetWorks. I have been able to work with a growing number of people, including faculty and CHEST staff. The more invested I have become, the more CHEST truly feels like family.
I understand that while it may be easy for me to tell members to get involved, it often feels much more difficult to actually get appointed to a leadership position. Early in my career, I was given the advice, “When you are given a task, make sure you blow it out of the water. That will only open more doors for you.” Making the most of a position on a NetWork or committee can create future opportunities. We recently had self-nominations for leadership positions within the NetWork steering committees and committees at large. Some positions have one to two openings for 20 applications. It can be frustrating not to get a position the first time around. However, it is common for members to have to apply numerous times prior to being appointed. When applying to these positions, be sure to highlight any prior CHEST involvement, as this may weigh in on an appointment to specific positions. Some of the decisions to appoint a nominee are based on prior engagement with CHEST.
So how can one get involved without holding a leadership position? My first piece of advice is to ensure you are getting CHEST emails. Check them regularly to so that you do not miss any opportunities. Next, be a member of at least one NetWork that is of interest to you. The NetWorks provide a smaller community within CHEST for special interests within our field. You will get emailed updates throughout the year that include any projects in which input is needed. At the CHEST annual meeting, each NetWork holds an Open Forum that functions as their annual face-to-face business meeting. These meetings are open to everyone. This is an excellent way to meet the current steering committee members and become involved in plans for the upcoming year. This year, we have made the dates and times of the NetWork steering committee calls public on the CHEST website. Any NetWork member can join these calls, even if they are not officially on the steering committee. All ongoing projects are discussed on these calls, so participation on the call offers an excellent opportunity to volunteer. You can also get involved with the NetWorks on social media by using the appropriate NetWork hashtags, along with tagging @accpchest to communicate with your NetWork colleagues.

Finally, the easiest way to embrace CHEST, and possibly the most obvious, is to get involved with the CHEST annual meeting. The meeting is at its best when planned and orchestrated by a diverse group of people. Annual meeting planning usually starts in November or December of the prior year. Submitting a proposal for a session at the annual meeting is strongly encouraged. Tips for how to submit a strong, well-rounded session are offered on the submission website. Reviewing these tips first can help strengthen your proposal. An easy way to become involved, even as a student or as a trainee, is to submit an abstract to the annual meeting
Summing up, I would encourage everyone to simply be an active participant: raise your hand to ask questions, introduce yourself to those around you, and attend the social events at CHEST annual meeting. Before you know it, new friends will become old friends, and attending the CHEST annual meeting will start to feel like going to a family reunion.
I am often overheard encouraging colleagues to become involved with CHEST. I am a strong believer that you get far more out of participation than you will ever put into it. I have now been fortunate to have many leadership roles within CHEST and currently serve on the Board of Regents and as Chair of the Council of NetWorks. I have been able to work with a growing number of people, including faculty and CHEST staff. The more invested I have become, the more CHEST truly feels like family.
I understand that while it may be easy for me to tell members to get involved, it often feels much more difficult to actually get appointed to a leadership position. Early in my career, I was given the advice, “When you are given a task, make sure you blow it out of the water. That will only open more doors for you.” Making the most of a position on a NetWork or committee can create future opportunities. We recently had self-nominations for leadership positions within the NetWork steering committees and committees at large. Some positions have one to two openings for 20 applications. It can be frustrating not to get a position the first time around. However, it is common for members to have to apply numerous times prior to being appointed. When applying to these positions, be sure to highlight any prior CHEST involvement, as this may weigh in on an appointment to specific positions. Some of the decisions to appoint a nominee are based on prior engagement with CHEST.
So how can one get involved without holding a leadership position? My first piece of advice is to ensure you are getting CHEST emails. Check them regularly to so that you do not miss any opportunities. Next, be a member of at least one NetWork that is of interest to you. The NetWorks provide a smaller community within CHEST for special interests within our field. You will get emailed updates throughout the year that include any projects in which input is needed. At the CHEST annual meeting, each NetWork holds an Open Forum that functions as their annual face-to-face business meeting. These meetings are open to everyone. This is an excellent way to meet the current steering committee members and become involved in plans for the upcoming year. This year, we have made the dates and times of the NetWork steering committee calls public on the CHEST website. Any NetWork member can join these calls, even if they are not officially on the steering committee. All ongoing projects are discussed on these calls, so participation on the call offers an excellent opportunity to volunteer. You can also get involved with the NetWorks on social media by using the appropriate NetWork hashtags, along with tagging @accpchest to communicate with your NetWork colleagues.

Finally, the easiest way to embrace CHEST, and possibly the most obvious, is to get involved with the CHEST annual meeting. The meeting is at its best when planned and orchestrated by a diverse group of people. Annual meeting planning usually starts in November or December of the prior year. Submitting a proposal for a session at the annual meeting is strongly encouraged. Tips for how to submit a strong, well-rounded session are offered on the submission website. Reviewing these tips first can help strengthen your proposal. An easy way to become involved, even as a student or as a trainee, is to submit an abstract to the annual meeting
Summing up, I would encourage everyone to simply be an active participant: raise your hand to ask questions, introduce yourself to those around you, and attend the social events at CHEST annual meeting. Before you know it, new friends will become old friends, and attending the CHEST annual meeting will start to feel like going to a family reunion.
Meet the FISH Bowl finalists
CHEST 2019 marked the inaugural FISH Bowl competition for attendees. Inspired by Shark Tank, our kinder, gentler, yet still competitive and cutting-edge FISH Bowl (Furthering Innovation and Science for Health) featured CHEST members disrupting our beliefs about how clinical care and education are performed. As health-care providers, they presented innovative ideas pertaining to education and clinical disease for pulmonary, critical care, and sleep medicine. Six finalists were chosen from dozens of submissions, and three emerged winners! In this new Meet the FISH Bowl Finalists series, CHEST introduces you to many of them – including winner Dr. Rachel Quaney.
Name: Rachel Quaney, MD
Institutional Affiliation: The Ohio State University
Position: Pulmonary and Critical Care Medicine Fellow
Title: Teaching Assessment Committee (TAC)
Brief Summary of Submission: Teaching Assessment Committee (TAC) is a novel approach to faculty feedback. We are modeling it after the success of the Clinical Competency Committees, but, in reverse, as fellows will give group-consensus-based feedback to faculty members.
Fellows will meet twice yearly with trained facilitators who help elicit constructive, nuanced feedback. The group setting ensures personal anonymity, which will serve to encourage more honest feedback. Then delivering this consensus-based information to program leadership and faculty members will hopefully provide helpful feedback regarding what is going well and what could be improved.
This pilot feasibility project is being employed at three fellowship programs this academic year. The goal will be to improve the feedback that faculty receive, while simultaneously increasing both faculty and fellow satisfaction with the process and the learning environment.
1. What inspired your innovation? More like who – and that would be the esteemed Dr. Gabe Bosslet of Indiana University. He brought the faculty perspective that attendings want better feedback. And, I supplied the fellow perspective—that even those of us who prioritize all things medical education often do a subpar job at providing effective feedback.
2. Who do you think can benefit most from it, and why? With some variation, almost all graduate medical education programs could benefit from the TAC method of faculty feedback. However, the most benefit would likely be seen in small programs or those that struggle with anonymity using current feedback methods.
3. What do you see as challenges to your innovation gaining widespread acceptance? How can they be overcome? I foresee two main challenges to implementation: time and buy-in. Fellows and residents are busy individuals with plenty on their plates, and this would require asking them for more time. This barrier could be solved by program and leadership buy-in or be exacerbated if it is lacking. If the process is endorsed by departmental and program leadership, this will provide credibility and ensure the necessary time is allotted.
4. What impact has winning FISH Bowl 2019 had on your vision for the innovation? The big picture vision I have for my innovation has not changed, but I am more acutely aware of the challenges and opportunities I will have to navigate, thanks to Drs. Morris, Niven, and Schulman. I am simultaneously more excited about this project but also feel the pressure to not disappoint!
5. How do you think your success at FISH Bowl 2019 will continue to impact your career overall in the months and years to come? It’s hard to imagine in what exact ways my career will be impacted, but I feel strongly that it will be positively influenced by this experience. I had the privilege of meeting a lot of individuals who feel passionate about medical education, both those established in our field and those at the beginning of their careers. These connections will likely lead to future collaborations and innovations.
CHEST 2019 marked the inaugural FISH Bowl competition for attendees. Inspired by Shark Tank, our kinder, gentler, yet still competitive and cutting-edge FISH Bowl (Furthering Innovation and Science for Health) featured CHEST members disrupting our beliefs about how clinical care and education are performed. As health-care providers, they presented innovative ideas pertaining to education and clinical disease for pulmonary, critical care, and sleep medicine. Six finalists were chosen from dozens of submissions, and three emerged winners! In this new Meet the FISH Bowl Finalists series, CHEST introduces you to many of them – including winner Dr. Rachel Quaney.
Name: Rachel Quaney, MD
Institutional Affiliation: The Ohio State University
Position: Pulmonary and Critical Care Medicine Fellow
Title: Teaching Assessment Committee (TAC)
Brief Summary of Submission: Teaching Assessment Committee (TAC) is a novel approach to faculty feedback. We are modeling it after the success of the Clinical Competency Committees, but, in reverse, as fellows will give group-consensus-based feedback to faculty members.
Fellows will meet twice yearly with trained facilitators who help elicit constructive, nuanced feedback. The group setting ensures personal anonymity, which will serve to encourage more honest feedback. Then delivering this consensus-based information to program leadership and faculty members will hopefully provide helpful feedback regarding what is going well and what could be improved.
This pilot feasibility project is being employed at three fellowship programs this academic year. The goal will be to improve the feedback that faculty receive, while simultaneously increasing both faculty and fellow satisfaction with the process and the learning environment.
1. What inspired your innovation? More like who – and that would be the esteemed Dr. Gabe Bosslet of Indiana University. He brought the faculty perspective that attendings want better feedback. And, I supplied the fellow perspective—that even those of us who prioritize all things medical education often do a subpar job at providing effective feedback.
2. Who do you think can benefit most from it, and why? With some variation, almost all graduate medical education programs could benefit from the TAC method of faculty feedback. However, the most benefit would likely be seen in small programs or those that struggle with anonymity using current feedback methods.
3. What do you see as challenges to your innovation gaining widespread acceptance? How can they be overcome? I foresee two main challenges to implementation: time and buy-in. Fellows and residents are busy individuals with plenty on their plates, and this would require asking them for more time. This barrier could be solved by program and leadership buy-in or be exacerbated if it is lacking. If the process is endorsed by departmental and program leadership, this will provide credibility and ensure the necessary time is allotted.
4. What impact has winning FISH Bowl 2019 had on your vision for the innovation? The big picture vision I have for my innovation has not changed, but I am more acutely aware of the challenges and opportunities I will have to navigate, thanks to Drs. Morris, Niven, and Schulman. I am simultaneously more excited about this project but also feel the pressure to not disappoint!
5. How do you think your success at FISH Bowl 2019 will continue to impact your career overall in the months and years to come? It’s hard to imagine in what exact ways my career will be impacted, but I feel strongly that it will be positively influenced by this experience. I had the privilege of meeting a lot of individuals who feel passionate about medical education, both those established in our field and those at the beginning of their careers. These connections will likely lead to future collaborations and innovations.
CHEST 2019 marked the inaugural FISH Bowl competition for attendees. Inspired by Shark Tank, our kinder, gentler, yet still competitive and cutting-edge FISH Bowl (Furthering Innovation and Science for Health) featured CHEST members disrupting our beliefs about how clinical care and education are performed. As health-care providers, they presented innovative ideas pertaining to education and clinical disease for pulmonary, critical care, and sleep medicine. Six finalists were chosen from dozens of submissions, and three emerged winners! In this new Meet the FISH Bowl Finalists series, CHEST introduces you to many of them – including winner Dr. Rachel Quaney.
Name: Rachel Quaney, MD
Institutional Affiliation: The Ohio State University
Position: Pulmonary and Critical Care Medicine Fellow
Title: Teaching Assessment Committee (TAC)
Brief Summary of Submission: Teaching Assessment Committee (TAC) is a novel approach to faculty feedback. We are modeling it after the success of the Clinical Competency Committees, but, in reverse, as fellows will give group-consensus-based feedback to faculty members.
Fellows will meet twice yearly with trained facilitators who help elicit constructive, nuanced feedback. The group setting ensures personal anonymity, which will serve to encourage more honest feedback. Then delivering this consensus-based information to program leadership and faculty members will hopefully provide helpful feedback regarding what is going well and what could be improved.
This pilot feasibility project is being employed at three fellowship programs this academic year. The goal will be to improve the feedback that faculty receive, while simultaneously increasing both faculty and fellow satisfaction with the process and the learning environment.
1. What inspired your innovation? More like who – and that would be the esteemed Dr. Gabe Bosslet of Indiana University. He brought the faculty perspective that attendings want better feedback. And, I supplied the fellow perspective—that even those of us who prioritize all things medical education often do a subpar job at providing effective feedback.
2. Who do you think can benefit most from it, and why? With some variation, almost all graduate medical education programs could benefit from the TAC method of faculty feedback. However, the most benefit would likely be seen in small programs or those that struggle with anonymity using current feedback methods.
3. What do you see as challenges to your innovation gaining widespread acceptance? How can they be overcome? I foresee two main challenges to implementation: time and buy-in. Fellows and residents are busy individuals with plenty on their plates, and this would require asking them for more time. This barrier could be solved by program and leadership buy-in or be exacerbated if it is lacking. If the process is endorsed by departmental and program leadership, this will provide credibility and ensure the necessary time is allotted.
4. What impact has winning FISH Bowl 2019 had on your vision for the innovation? The big picture vision I have for my innovation has not changed, but I am more acutely aware of the challenges and opportunities I will have to navigate, thanks to Drs. Morris, Niven, and Schulman. I am simultaneously more excited about this project but also feel the pressure to not disappoint!
5. How do you think your success at FISH Bowl 2019 will continue to impact your career overall in the months and years to come? It’s hard to imagine in what exact ways my career will be impacted, but I feel strongly that it will be positively influenced by this experience. I had the privilege of meeting a lot of individuals who feel passionate about medical education, both those established in our field and those at the beginning of their careers. These connections will likely lead to future collaborations and innovations.
COVID-19: Just a virus, right?
My first exposure to the notion of scarce resources was in medical school. I had to discuss the ethical principles behind the allocation of organs for transplantation, specifically livers and the required abstinence from alcohol ... but this was just an exercise, right?
A few years later, during residency, I heard the anecdotes from one of my internal medicine attendings about the time he spent in Europe as a visiting geriatrics fellow in the 1970s. The health-care districts in the region would be allotted an annual budget, and it was up to those districts how to best allocate those resources to meet, to the best of their abilities, the health-care needs of their population. He vividly recalled that a patient he cared for, an individual over 65 in need of renal replacement therapy for a reversible condition, who was not offered such therapy despite the clear benefit. There was a finite amount of resources, and those resources were thought to be better spent on public health measures like vaccination ... but that was on another continent and in another era, right?
I remember when I first heard of an outbreak of viral pneumonia in China in January of this year. As someone prone to anxiety, my first strategy was to put my head in the sand and wait it out. This strategy didn’t last very long – within a couple of weeks, there were confirmed cases in the United States. It was now apparent that this virus was not going to be contained. In an impressively short amount of time, SARS-CoV 2 has infected over 3.5 million individuals and killed almost a quarter million people worldwide. In the United States, we have seen almost 1.2 million cases and lost over 68 thousand lives. This pandemic has managed to devastate multiple countries, health care systems, and economies. It has also challenged every physician’s ideas of beneficence and justice ... but it’s just a virus, right?
Beneficence, the principle of medical ethics regarding acting in the patient’s best interest, had always seemed to me to be a no-brainer. Not like autonomy, which can get sticky, or justice, which I really had not had to consider much prior to 2020. Of course, I would always do what was best for my patient, I thought, why wouldn’t I?
Justice, the principle that deals with the distribution of scarce health-care resources, is the wrench that has been thrown into the beneficence works in the age of COVID-19. In a country and an era in which I had not dreamed we would ever have to think about how to support multiple people with one ventilator, we have had to do just that (“Joint Statement on Multiple Patients per Ventilator,” CHEST News, Mar 27, 2020). Things that I have taken for granted through all of my training are now worth their weight in gold—from sedative drips and inhalers down to videolaryngoscopy blades and face masks. I can’t just do what is best for my patient because sometimes what is best for my patient is not what is best for my next patient, what is best for my team, or even what is best for me and for my family. COVID-19 has reminded us of the uncomfortable truth that when contemplating justice, the patient in front of us is not the only person we have to consider.
Early on, before things in the United States had surged, I asked the twitter community what I thought would be a hypothetical question: “An employee needs to urgently help a COVID-19 patient. There is no appropriate PPE available due to shortage. What should happen?”
Like the idea of splitting ventilators, it was a thought I had never considered pre-COVID-19. Our instinct as physicians, especially as critical care physicians, is to intervene in emergency situations as quickly as possible. The extensive PPE required to manage COVID-19 patients has slowed that process, but, as many institutions are reaching the ends of their PPE stores, our safety is now placed at odds with that of our patient’s. To stay back violates what we feel is our duty to our patients, to go in violates our duty to ourselves, to our families, and to the rest of our patients. To care adequately for your patient is to put yourself at risk (and vice-versa), and this is a problem that I don’t think we have an answer for.
COVID-19 threatens many good and noble things, and what is worse, it directly puts them at odds with one another. They are paired sliding scales, where more of one means less of the other. If I have enough masks, it means my colleague probably doesn’t. If we have enough ventilators, it means another city doesn’t. If I get a break to be with my family, it means someone else is having to leave theirs to tend to patients who are sicker, lonelier, and more numerous than in any other time in recent memory.
And if these situations and resource limitations don’t provide enough moral injury for health-care workers, there are some specifics of humanity’s response to the pandemic that are exceptionally hurtful.
We as a country had notice, which was squandered. Instead of caution and preparation, we saw the powers that be make light of the serious situation most scientists and clinicians warned was coming. Instead of efforts to find or create PPE, we saw accusations against us of misuse and waste (“Trump comments about hospital mask thefts spark backlash from doctors,” Newsweek, March 30, 2020). Instead of support, we saw our altruism taken advantage of and used against us in unsafe and unfair situations. We have seen physicians in training and full-fledged attendings alike treated unfairly by their supervisors, instead of protected. Every instance of anti-science opinion or action from our friends and families that we once tolerated now feels like a personal affront, as these directly increase our risk and our immediate family’s risk of contracting the illness. We are being touted as heroes and angels, but really, we’re afraid—afraid of our patients, afraid of illness, afraid for our families, and afraid of jobs that we used to love. We don’t want to be praised; we just want to work our regular jobs safely and with adequate support.
I don’t know what health care looks like at the end of all of this. Relationships between physicians and health-care administrations were strained before the pandemic, to say the least. How can health-care workers just go back to business as usual, working for entities that were so ill-prepared, and, in many cases, calloused toward the concerns of their employees?
COVID-19 has revealed the fragility of our health-care system, our public health capabilities, and our economy. The pandemic has forced us to finally acknowledge something that has been true all along—our resources are finite, and tension exists between what is right and what is profitable, and between what is just and what is easy.
But it’s just a virus, right?
Dr. Fridenmaker is a Pulmonary and Critical Care Fellow at the University of Kentucky, Lexington.
My first exposure to the notion of scarce resources was in medical school. I had to discuss the ethical principles behind the allocation of organs for transplantation, specifically livers and the required abstinence from alcohol ... but this was just an exercise, right?
A few years later, during residency, I heard the anecdotes from one of my internal medicine attendings about the time he spent in Europe as a visiting geriatrics fellow in the 1970s. The health-care districts in the region would be allotted an annual budget, and it was up to those districts how to best allocate those resources to meet, to the best of their abilities, the health-care needs of their population. He vividly recalled that a patient he cared for, an individual over 65 in need of renal replacement therapy for a reversible condition, who was not offered such therapy despite the clear benefit. There was a finite amount of resources, and those resources were thought to be better spent on public health measures like vaccination ... but that was on another continent and in another era, right?
I remember when I first heard of an outbreak of viral pneumonia in China in January of this year. As someone prone to anxiety, my first strategy was to put my head in the sand and wait it out. This strategy didn’t last very long – within a couple of weeks, there were confirmed cases in the United States. It was now apparent that this virus was not going to be contained. In an impressively short amount of time, SARS-CoV 2 has infected over 3.5 million individuals and killed almost a quarter million people worldwide. In the United States, we have seen almost 1.2 million cases and lost over 68 thousand lives. This pandemic has managed to devastate multiple countries, health care systems, and economies. It has also challenged every physician’s ideas of beneficence and justice ... but it’s just a virus, right?
Beneficence, the principle of medical ethics regarding acting in the patient’s best interest, had always seemed to me to be a no-brainer. Not like autonomy, which can get sticky, or justice, which I really had not had to consider much prior to 2020. Of course, I would always do what was best for my patient, I thought, why wouldn’t I?
Justice, the principle that deals with the distribution of scarce health-care resources, is the wrench that has been thrown into the beneficence works in the age of COVID-19. In a country and an era in which I had not dreamed we would ever have to think about how to support multiple people with one ventilator, we have had to do just that (“Joint Statement on Multiple Patients per Ventilator,” CHEST News, Mar 27, 2020). Things that I have taken for granted through all of my training are now worth their weight in gold—from sedative drips and inhalers down to videolaryngoscopy blades and face masks. I can’t just do what is best for my patient because sometimes what is best for my patient is not what is best for my next patient, what is best for my team, or even what is best for me and for my family. COVID-19 has reminded us of the uncomfortable truth that when contemplating justice, the patient in front of us is not the only person we have to consider.
Early on, before things in the United States had surged, I asked the twitter community what I thought would be a hypothetical question: “An employee needs to urgently help a COVID-19 patient. There is no appropriate PPE available due to shortage. What should happen?”
Like the idea of splitting ventilators, it was a thought I had never considered pre-COVID-19. Our instinct as physicians, especially as critical care physicians, is to intervene in emergency situations as quickly as possible. The extensive PPE required to manage COVID-19 patients has slowed that process, but, as many institutions are reaching the ends of their PPE stores, our safety is now placed at odds with that of our patient’s. To stay back violates what we feel is our duty to our patients, to go in violates our duty to ourselves, to our families, and to the rest of our patients. To care adequately for your patient is to put yourself at risk (and vice-versa), and this is a problem that I don’t think we have an answer for.
COVID-19 threatens many good and noble things, and what is worse, it directly puts them at odds with one another. They are paired sliding scales, where more of one means less of the other. If I have enough masks, it means my colleague probably doesn’t. If we have enough ventilators, it means another city doesn’t. If I get a break to be with my family, it means someone else is having to leave theirs to tend to patients who are sicker, lonelier, and more numerous than in any other time in recent memory.
And if these situations and resource limitations don’t provide enough moral injury for health-care workers, there are some specifics of humanity’s response to the pandemic that are exceptionally hurtful.
We as a country had notice, which was squandered. Instead of caution and preparation, we saw the powers that be make light of the serious situation most scientists and clinicians warned was coming. Instead of efforts to find or create PPE, we saw accusations against us of misuse and waste (“Trump comments about hospital mask thefts spark backlash from doctors,” Newsweek, March 30, 2020). Instead of support, we saw our altruism taken advantage of and used against us in unsafe and unfair situations. We have seen physicians in training and full-fledged attendings alike treated unfairly by their supervisors, instead of protected. Every instance of anti-science opinion or action from our friends and families that we once tolerated now feels like a personal affront, as these directly increase our risk and our immediate family’s risk of contracting the illness. We are being touted as heroes and angels, but really, we’re afraid—afraid of our patients, afraid of illness, afraid for our families, and afraid of jobs that we used to love. We don’t want to be praised; we just want to work our regular jobs safely and with adequate support.
I don’t know what health care looks like at the end of all of this. Relationships between physicians and health-care administrations were strained before the pandemic, to say the least. How can health-care workers just go back to business as usual, working for entities that were so ill-prepared, and, in many cases, calloused toward the concerns of their employees?
COVID-19 has revealed the fragility of our health-care system, our public health capabilities, and our economy. The pandemic has forced us to finally acknowledge something that has been true all along—our resources are finite, and tension exists between what is right and what is profitable, and between what is just and what is easy.
But it’s just a virus, right?
Dr. Fridenmaker is a Pulmonary and Critical Care Fellow at the University of Kentucky, Lexington.
My first exposure to the notion of scarce resources was in medical school. I had to discuss the ethical principles behind the allocation of organs for transplantation, specifically livers and the required abstinence from alcohol ... but this was just an exercise, right?
A few years later, during residency, I heard the anecdotes from one of my internal medicine attendings about the time he spent in Europe as a visiting geriatrics fellow in the 1970s. The health-care districts in the region would be allotted an annual budget, and it was up to those districts how to best allocate those resources to meet, to the best of their abilities, the health-care needs of their population. He vividly recalled that a patient he cared for, an individual over 65 in need of renal replacement therapy for a reversible condition, who was not offered such therapy despite the clear benefit. There was a finite amount of resources, and those resources were thought to be better spent on public health measures like vaccination ... but that was on another continent and in another era, right?
I remember when I first heard of an outbreak of viral pneumonia in China in January of this year. As someone prone to anxiety, my first strategy was to put my head in the sand and wait it out. This strategy didn’t last very long – within a couple of weeks, there were confirmed cases in the United States. It was now apparent that this virus was not going to be contained. In an impressively short amount of time, SARS-CoV 2 has infected over 3.5 million individuals and killed almost a quarter million people worldwide. In the United States, we have seen almost 1.2 million cases and lost over 68 thousand lives. This pandemic has managed to devastate multiple countries, health care systems, and economies. It has also challenged every physician’s ideas of beneficence and justice ... but it’s just a virus, right?
Beneficence, the principle of medical ethics regarding acting in the patient’s best interest, had always seemed to me to be a no-brainer. Not like autonomy, which can get sticky, or justice, which I really had not had to consider much prior to 2020. Of course, I would always do what was best for my patient, I thought, why wouldn’t I?
Justice, the principle that deals with the distribution of scarce health-care resources, is the wrench that has been thrown into the beneficence works in the age of COVID-19. In a country and an era in which I had not dreamed we would ever have to think about how to support multiple people with one ventilator, we have had to do just that (“Joint Statement on Multiple Patients per Ventilator,” CHEST News, Mar 27, 2020). Things that I have taken for granted through all of my training are now worth their weight in gold—from sedative drips and inhalers down to videolaryngoscopy blades and face masks. I can’t just do what is best for my patient because sometimes what is best for my patient is not what is best for my next patient, what is best for my team, or even what is best for me and for my family. COVID-19 has reminded us of the uncomfortable truth that when contemplating justice, the patient in front of us is not the only person we have to consider.
Early on, before things in the United States had surged, I asked the twitter community what I thought would be a hypothetical question: “An employee needs to urgently help a COVID-19 patient. There is no appropriate PPE available due to shortage. What should happen?”
Like the idea of splitting ventilators, it was a thought I had never considered pre-COVID-19. Our instinct as physicians, especially as critical care physicians, is to intervene in emergency situations as quickly as possible. The extensive PPE required to manage COVID-19 patients has slowed that process, but, as many institutions are reaching the ends of their PPE stores, our safety is now placed at odds with that of our patient’s. To stay back violates what we feel is our duty to our patients, to go in violates our duty to ourselves, to our families, and to the rest of our patients. To care adequately for your patient is to put yourself at risk (and vice-versa), and this is a problem that I don’t think we have an answer for.
COVID-19 threatens many good and noble things, and what is worse, it directly puts them at odds with one another. They are paired sliding scales, where more of one means less of the other. If I have enough masks, it means my colleague probably doesn’t. If we have enough ventilators, it means another city doesn’t. If I get a break to be with my family, it means someone else is having to leave theirs to tend to patients who are sicker, lonelier, and more numerous than in any other time in recent memory.
And if these situations and resource limitations don’t provide enough moral injury for health-care workers, there are some specifics of humanity’s response to the pandemic that are exceptionally hurtful.
We as a country had notice, which was squandered. Instead of caution and preparation, we saw the powers that be make light of the serious situation most scientists and clinicians warned was coming. Instead of efforts to find or create PPE, we saw accusations against us of misuse and waste (“Trump comments about hospital mask thefts spark backlash from doctors,” Newsweek, March 30, 2020). Instead of support, we saw our altruism taken advantage of and used against us in unsafe and unfair situations. We have seen physicians in training and full-fledged attendings alike treated unfairly by their supervisors, instead of protected. Every instance of anti-science opinion or action from our friends and families that we once tolerated now feels like a personal affront, as these directly increase our risk and our immediate family’s risk of contracting the illness. We are being touted as heroes and angels, but really, we’re afraid—afraid of our patients, afraid of illness, afraid for our families, and afraid of jobs that we used to love. We don’t want to be praised; we just want to work our regular jobs safely and with adequate support.
I don’t know what health care looks like at the end of all of this. Relationships between physicians and health-care administrations were strained before the pandemic, to say the least. How can health-care workers just go back to business as usual, working for entities that were so ill-prepared, and, in many cases, calloused toward the concerns of their employees?
COVID-19 has revealed the fragility of our health-care system, our public health capabilities, and our economy. The pandemic has forced us to finally acknowledge something that has been true all along—our resources are finite, and tension exists between what is right and what is profitable, and between what is just and what is easy.
But it’s just a virus, right?
Dr. Fridenmaker is a Pulmonary and Critical Care Fellow at the University of Kentucky, Lexington.
COVID-19 and the cardiovascular system. Thrombotic events in COVID-19. Interprofessional collaboration.
Cardiovascular medicine and surgery
COVID-19 and the cardiovascular system
With the global outbreak of severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) ongoing, there is increased awareness of the cardiovascular manifestations and implications of COVID-19. Approximately 20% of inpatients with COVID-19 have acute cardiac injury (defined as cardiac troponin elevation) (Shi S, et al. JAMA Cardiol. 2020 Mar 25. doi: 10.1001/jamacardio.2020.0950). Moreover, in one cohort, both acute cardiac injury and preexisting cardiovascular disease (CVD) were associated with COVID-19 hospital mortality: 69% with elevated troponin levels and underlying CVD vs 7.6% with neither (Guo T, et al. JAMA Cardiol. 2020 Mar 27. doi: 10.1001/jamacardio.2020.1017). Moreover, case reports suggest COVID-19 may present as myopericarditis, cardiomyopathy, acute on chronic decompensated heart failure, and acute coronary syndrome (Fried JA, et al. Circulation. 2020 Apr 3. doi: 10.1161/circulationaha.120.047164). Adding to this clinical variability, one case series suggests that electrocardiographic ST-segment elevation may not reliably identify obstructive coronary disease (Bangalore S, et al. N Engl J Med. 2020 Apr 17. doi: 10.1056/NEJMc2009020). Intriguingly, the angiotensin-converting enzyme 2 (ACE2) protein is the functional receptor for SARS-CoV-2 cell entry, and ACE2 is highly expressed in pulmonary and cardiac cells (Driggin E, et al. J Am Coll Cardiol. 2020;75[18]:2352). Given the central role of ACE2 and the renin-angiotensin-aldosterone (RAAS) system in cardiovascular pathophysiology and pharmacotherapy, RAAS modulation could have beneficial and/or detrimental effects with COVID-19 (Vaduganathan M, et al. N Engl J Med. 2020;382:1653). Available evidence and societal guidelines support continuing RAAS antagonists in patients per established clinical practice (Mancia G, et al. N Engl J Med. 2020 May 1. doi: 10.1056/NEJMoa2006923); (Mehra MR, et al. N Engl J Med. 2020 May 1. doi: 10.1056/NEJMoa2007621). A better understanding of the direct and indirect effect of SARS-CoV-2 on the cardiovascular system will require additional evidence.
Benjamin B. Kenigsberg, MD
Fellow-in-Training Steering Committee Member
Thrombotic events in COVID-19: Implications and evolving practice recommendations
A startling potential complication of infection with SARS-CoV2 has been the reported predisposition to thrombotic events. Mortality in COVID-19 patients is associated with notable increases in hemostatic parameters such as levels of
Vascular societies led by International Society on Thrombosis and Haemostasis (ISTH) have published consensus recommendations for guidance. If no contraindications exist, pharmacologic venous thromboembolism (VTE) prophylaxis with unfractionated heparin (UFH) or low-molecular-weight heparin (LMWH) is recommended for hospitalized patients with moderate or severe COVID-19 without disseminated intravascular coagulation (DIC). VTE prophylaxis should also be considered for patients with moderate or severe COVID-19 and in DIC but without overt bleeding. There is insufficient evidence to consider routine therapeutic or intermediate-dose parenteral anticoagulation with UFH or LMWH. Many institutions have developed protocols advising therapeutic-intensity anticoagulation when certain thresholds of
Saiprakash B. Venkateshiah, MD, FCCP,
Chair
Gabriela Magda, MD
Fellow-in-Training Steering Committee Member
Interprofessional Team
Quality of interprofessional collaboration in the medical intensive care unit: perceptions by caregivers
A recent study examining caregivers’ perceptions of team interactions and interprofessional collaborative practice (IPCP) behaviors offers new, exciting insights on the importance of interprofessional team functioning in the medical intensive care unit (MICU) (Chen DW, et al. J Gen Intern Med. 2018;33[10]:1708).
The Support Person Jefferson Teamwork Observation Guide (JTOG)TM survey was administered to 161 random caregivers of patients hospitalized in a single large urban academic medical center MICU between May 2016 and December 2016. The survey tool was designed to elicit the perceptions of caregivers regarding team functioning. Survey questions were directly mapped to the 2011 Interprofessional Education Collaborative (IPEC) Expert Panel core competencies for IPCP and divided into four domains (values/ethics; interprofessional communication; roles/responsibilities; teams/teamwork).
Also appended to the surveys were additional follow-up questions that addressed the overall satisfaction with the team and general attitudes regarding the importance of interprofessional team-based care. Caregivers agreed on the importance of health-care professionals working together as a team to provide patient care (3.97/4.00 Likert scale 4 = extremely important). Caregivers expressed satisfaction with the MICU team (3.74/4.00). Furthermore, caregivers agreed that the MICU team demonstrated competencies in all four domains of IPCP: values/ethics (3.55/4.00), interprofessional communication (3.58/4.00), roles/responsibilities (3.61/4.00), and teams/teamwork (3.64/4.00). Caregivers felt the MICU team provided patient/family-centered care (sub-competency 3.58/4.00). Notably, the overall caregiver survey scores detailing how well each MICU team functioned were positively correlated to overall satisfaction with the MICU team (r = 0.596 P < .01).
Limitations of the study included:
1. The sample is from a single institution, and perceptions of caregivers cannot be applied to all populations.
2. No information regarding patient, such as diagnosis, was obtained.
3. Caregivers satisfied with care might be more likely to participate.
4. No distinction was made between data collected from caregivers surveyed in the resident-fellow staffed MICU vs NP-staffed MICU.
It has been described that ineffective teamwork and team communication in health care settings are associated with increased patient harm and sentinel events (Kohn LT et al. Washington (DC): National Academies Press(US); 2000); (Page A, Washington (DC): National Academies Press (US); (The Joint Commission. Sentinel Event Alert 2008, 40); (Brennan TA, et al. N Engl J Med. 1991;324:370). Cultural differences between members of the health-care team and established hierarchies of control have been identified as barriers to communication and teamwork in ICUs (Alexanian JA, et al. J Crit Care Med. 2015;43[9]:1880); (Manias E, Street A. Int J Nurs Stud. 2001;38[2]:129).
Overall, the findings from this study emphasize the importance of interprofessional communication and teamwork in the MICU and delivery of patient/family-centered care from the caregivers’ perspective. The unique insight into caregivers’ perspectives on specific team behaviors may be the basis for future quality improvement initiatives.
Justin K. Lui, MD,
Mary Jo Farmer, MD, PhD, FCCP
Kristina E. Ramirez, RRT, MPH, FCCP
Cardiovascular medicine and surgery
COVID-19 and the cardiovascular system
With the global outbreak of severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) ongoing, there is increased awareness of the cardiovascular manifestations and implications of COVID-19. Approximately 20% of inpatients with COVID-19 have acute cardiac injury (defined as cardiac troponin elevation) (Shi S, et al. JAMA Cardiol. 2020 Mar 25. doi: 10.1001/jamacardio.2020.0950). Moreover, in one cohort, both acute cardiac injury and preexisting cardiovascular disease (CVD) were associated with COVID-19 hospital mortality: 69% with elevated troponin levels and underlying CVD vs 7.6% with neither (Guo T, et al. JAMA Cardiol. 2020 Mar 27. doi: 10.1001/jamacardio.2020.1017). Moreover, case reports suggest COVID-19 may present as myopericarditis, cardiomyopathy, acute on chronic decompensated heart failure, and acute coronary syndrome (Fried JA, et al. Circulation. 2020 Apr 3. doi: 10.1161/circulationaha.120.047164). Adding to this clinical variability, one case series suggests that electrocardiographic ST-segment elevation may not reliably identify obstructive coronary disease (Bangalore S, et al. N Engl J Med. 2020 Apr 17. doi: 10.1056/NEJMc2009020). Intriguingly, the angiotensin-converting enzyme 2 (ACE2) protein is the functional receptor for SARS-CoV-2 cell entry, and ACE2 is highly expressed in pulmonary and cardiac cells (Driggin E, et al. J Am Coll Cardiol. 2020;75[18]:2352). Given the central role of ACE2 and the renin-angiotensin-aldosterone (RAAS) system in cardiovascular pathophysiology and pharmacotherapy, RAAS modulation could have beneficial and/or detrimental effects with COVID-19 (Vaduganathan M, et al. N Engl J Med. 2020;382:1653). Available evidence and societal guidelines support continuing RAAS antagonists in patients per established clinical practice (Mancia G, et al. N Engl J Med. 2020 May 1. doi: 10.1056/NEJMoa2006923); (Mehra MR, et al. N Engl J Med. 2020 May 1. doi: 10.1056/NEJMoa2007621). A better understanding of the direct and indirect effect of SARS-CoV-2 on the cardiovascular system will require additional evidence.
Benjamin B. Kenigsberg, MD
Fellow-in-Training Steering Committee Member
Thrombotic events in COVID-19: Implications and evolving practice recommendations
A startling potential complication of infection with SARS-CoV2 has been the reported predisposition to thrombotic events. Mortality in COVID-19 patients is associated with notable increases in hemostatic parameters such as levels of
Vascular societies led by International Society on Thrombosis and Haemostasis (ISTH) have published consensus recommendations for guidance. If no contraindications exist, pharmacologic venous thromboembolism (VTE) prophylaxis with unfractionated heparin (UFH) or low-molecular-weight heparin (LMWH) is recommended for hospitalized patients with moderate or severe COVID-19 without disseminated intravascular coagulation (DIC). VTE prophylaxis should also be considered for patients with moderate or severe COVID-19 and in DIC but without overt bleeding. There is insufficient evidence to consider routine therapeutic or intermediate-dose parenteral anticoagulation with UFH or LMWH. Many institutions have developed protocols advising therapeutic-intensity anticoagulation when certain thresholds of
Saiprakash B. Venkateshiah, MD, FCCP,
Chair
Gabriela Magda, MD
Fellow-in-Training Steering Committee Member
Interprofessional Team
Quality of interprofessional collaboration in the medical intensive care unit: perceptions by caregivers
A recent study examining caregivers’ perceptions of team interactions and interprofessional collaborative practice (IPCP) behaviors offers new, exciting insights on the importance of interprofessional team functioning in the medical intensive care unit (MICU) (Chen DW, et al. J Gen Intern Med. 2018;33[10]:1708).
The Support Person Jefferson Teamwork Observation Guide (JTOG)TM survey was administered to 161 random caregivers of patients hospitalized in a single large urban academic medical center MICU between May 2016 and December 2016. The survey tool was designed to elicit the perceptions of caregivers regarding team functioning. Survey questions were directly mapped to the 2011 Interprofessional Education Collaborative (IPEC) Expert Panel core competencies for IPCP and divided into four domains (values/ethics; interprofessional communication; roles/responsibilities; teams/teamwork).
Also appended to the surveys were additional follow-up questions that addressed the overall satisfaction with the team and general attitudes regarding the importance of interprofessional team-based care. Caregivers agreed on the importance of health-care professionals working together as a team to provide patient care (3.97/4.00 Likert scale 4 = extremely important). Caregivers expressed satisfaction with the MICU team (3.74/4.00). Furthermore, caregivers agreed that the MICU team demonstrated competencies in all four domains of IPCP: values/ethics (3.55/4.00), interprofessional communication (3.58/4.00), roles/responsibilities (3.61/4.00), and teams/teamwork (3.64/4.00). Caregivers felt the MICU team provided patient/family-centered care (sub-competency 3.58/4.00). Notably, the overall caregiver survey scores detailing how well each MICU team functioned were positively correlated to overall satisfaction with the MICU team (r = 0.596 P < .01).
Limitations of the study included:
1. The sample is from a single institution, and perceptions of caregivers cannot be applied to all populations.
2. No information regarding patient, such as diagnosis, was obtained.
3. Caregivers satisfied with care might be more likely to participate.
4. No distinction was made between data collected from caregivers surveyed in the resident-fellow staffed MICU vs NP-staffed MICU.
It has been described that ineffective teamwork and team communication in health care settings are associated with increased patient harm and sentinel events (Kohn LT et al. Washington (DC): National Academies Press(US); 2000); (Page A, Washington (DC): National Academies Press (US); (The Joint Commission. Sentinel Event Alert 2008, 40); (Brennan TA, et al. N Engl J Med. 1991;324:370). Cultural differences between members of the health-care team and established hierarchies of control have been identified as barriers to communication and teamwork in ICUs (Alexanian JA, et al. J Crit Care Med. 2015;43[9]:1880); (Manias E, Street A. Int J Nurs Stud. 2001;38[2]:129).
Overall, the findings from this study emphasize the importance of interprofessional communication and teamwork in the MICU and delivery of patient/family-centered care from the caregivers’ perspective. The unique insight into caregivers’ perspectives on specific team behaviors may be the basis for future quality improvement initiatives.
Justin K. Lui, MD,
Mary Jo Farmer, MD, PhD, FCCP
Kristina E. Ramirez, RRT, MPH, FCCP
Cardiovascular medicine and surgery
COVID-19 and the cardiovascular system
With the global outbreak of severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) ongoing, there is increased awareness of the cardiovascular manifestations and implications of COVID-19. Approximately 20% of inpatients with COVID-19 have acute cardiac injury (defined as cardiac troponin elevation) (Shi S, et al. JAMA Cardiol. 2020 Mar 25. doi: 10.1001/jamacardio.2020.0950). Moreover, in one cohort, both acute cardiac injury and preexisting cardiovascular disease (CVD) were associated with COVID-19 hospital mortality: 69% with elevated troponin levels and underlying CVD vs 7.6% with neither (Guo T, et al. JAMA Cardiol. 2020 Mar 27. doi: 10.1001/jamacardio.2020.1017). Moreover, case reports suggest COVID-19 may present as myopericarditis, cardiomyopathy, acute on chronic decompensated heart failure, and acute coronary syndrome (Fried JA, et al. Circulation. 2020 Apr 3. doi: 10.1161/circulationaha.120.047164). Adding to this clinical variability, one case series suggests that electrocardiographic ST-segment elevation may not reliably identify obstructive coronary disease (Bangalore S, et al. N Engl J Med. 2020 Apr 17. doi: 10.1056/NEJMc2009020). Intriguingly, the angiotensin-converting enzyme 2 (ACE2) protein is the functional receptor for SARS-CoV-2 cell entry, and ACE2 is highly expressed in pulmonary and cardiac cells (Driggin E, et al. J Am Coll Cardiol. 2020;75[18]:2352). Given the central role of ACE2 and the renin-angiotensin-aldosterone (RAAS) system in cardiovascular pathophysiology and pharmacotherapy, RAAS modulation could have beneficial and/or detrimental effects with COVID-19 (Vaduganathan M, et al. N Engl J Med. 2020;382:1653). Available evidence and societal guidelines support continuing RAAS antagonists in patients per established clinical practice (Mancia G, et al. N Engl J Med. 2020 May 1. doi: 10.1056/NEJMoa2006923); (Mehra MR, et al. N Engl J Med. 2020 May 1. doi: 10.1056/NEJMoa2007621). A better understanding of the direct and indirect effect of SARS-CoV-2 on the cardiovascular system will require additional evidence.
Benjamin B. Kenigsberg, MD
Fellow-in-Training Steering Committee Member
Thrombotic events in COVID-19: Implications and evolving practice recommendations
A startling potential complication of infection with SARS-CoV2 has been the reported predisposition to thrombotic events. Mortality in COVID-19 patients is associated with notable increases in hemostatic parameters such as levels of
Vascular societies led by International Society on Thrombosis and Haemostasis (ISTH) have published consensus recommendations for guidance. If no contraindications exist, pharmacologic venous thromboembolism (VTE) prophylaxis with unfractionated heparin (UFH) or low-molecular-weight heparin (LMWH) is recommended for hospitalized patients with moderate or severe COVID-19 without disseminated intravascular coagulation (DIC). VTE prophylaxis should also be considered for patients with moderate or severe COVID-19 and in DIC but without overt bleeding. There is insufficient evidence to consider routine therapeutic or intermediate-dose parenteral anticoagulation with UFH or LMWH. Many institutions have developed protocols advising therapeutic-intensity anticoagulation when certain thresholds of
Saiprakash B. Venkateshiah, MD, FCCP,
Chair
Gabriela Magda, MD
Fellow-in-Training Steering Committee Member
Interprofessional Team
Quality of interprofessional collaboration in the medical intensive care unit: perceptions by caregivers
A recent study examining caregivers’ perceptions of team interactions and interprofessional collaborative practice (IPCP) behaviors offers new, exciting insights on the importance of interprofessional team functioning in the medical intensive care unit (MICU) (Chen DW, et al. J Gen Intern Med. 2018;33[10]:1708).
The Support Person Jefferson Teamwork Observation Guide (JTOG)TM survey was administered to 161 random caregivers of patients hospitalized in a single large urban academic medical center MICU between May 2016 and December 2016. The survey tool was designed to elicit the perceptions of caregivers regarding team functioning. Survey questions were directly mapped to the 2011 Interprofessional Education Collaborative (IPEC) Expert Panel core competencies for IPCP and divided into four domains (values/ethics; interprofessional communication; roles/responsibilities; teams/teamwork).
Also appended to the surveys were additional follow-up questions that addressed the overall satisfaction with the team and general attitudes regarding the importance of interprofessional team-based care. Caregivers agreed on the importance of health-care professionals working together as a team to provide patient care (3.97/4.00 Likert scale 4 = extremely important). Caregivers expressed satisfaction with the MICU team (3.74/4.00). Furthermore, caregivers agreed that the MICU team demonstrated competencies in all four domains of IPCP: values/ethics (3.55/4.00), interprofessional communication (3.58/4.00), roles/responsibilities (3.61/4.00), and teams/teamwork (3.64/4.00). Caregivers felt the MICU team provided patient/family-centered care (sub-competency 3.58/4.00). Notably, the overall caregiver survey scores detailing how well each MICU team functioned were positively correlated to overall satisfaction with the MICU team (r = 0.596 P < .01).
Limitations of the study included:
1. The sample is from a single institution, and perceptions of caregivers cannot be applied to all populations.
2. No information regarding patient, such as diagnosis, was obtained.
3. Caregivers satisfied with care might be more likely to participate.
4. No distinction was made between data collected from caregivers surveyed in the resident-fellow staffed MICU vs NP-staffed MICU.
It has been described that ineffective teamwork and team communication in health care settings are associated with increased patient harm and sentinel events (Kohn LT et al. Washington (DC): National Academies Press(US); 2000); (Page A, Washington (DC): National Academies Press (US); (The Joint Commission. Sentinel Event Alert 2008, 40); (Brennan TA, et al. N Engl J Med. 1991;324:370). Cultural differences between members of the health-care team and established hierarchies of control have been identified as barriers to communication and teamwork in ICUs (Alexanian JA, et al. J Crit Care Med. 2015;43[9]:1880); (Manias E, Street A. Int J Nurs Stud. 2001;38[2]:129).
Overall, the findings from this study emphasize the importance of interprofessional communication and teamwork in the MICU and delivery of patient/family-centered care from the caregivers’ perspective. The unique insight into caregivers’ perspectives on specific team behaviors may be the basis for future quality improvement initiatives.
Justin K. Lui, MD,
Mary Jo Farmer, MD, PhD, FCCP
Kristina E. Ramirez, RRT, MPH, FCCP
This month in the journal CHEST®
Editor’s picks
Preparing for the COVID-19 Pandemic: Our Experience in New York.By Dr. H. Zubair, et al.
The Utility of Electronic Inhaler Monitoring in COPD Management: Promises and Challenges.By Dr. A. H. Attaway, et al.
Patterns of Use of Adjunctive Therapies in Patients With Early Moderate-Severe Acute Respiratory Distress Syndrome: Insights From the LUNG SAFE Study.By Dr. A. Duggal, et al.
Clinical Evaluation of Deployed Military Personnel with Chronic Respiratory Symptoms: STAMPEDE III (Study of Active Duty Military for Pulmonary Disease Related to Environmental Deployment Exposures). By Dr. M. J. Morris, et al.
Editor’s picks
Editor’s picks
Preparing for the COVID-19 Pandemic: Our Experience in New York.By Dr. H. Zubair, et al.
The Utility of Electronic Inhaler Monitoring in COPD Management: Promises and Challenges.By Dr. A. H. Attaway, et al.
Patterns of Use of Adjunctive Therapies in Patients With Early Moderate-Severe Acute Respiratory Distress Syndrome: Insights From the LUNG SAFE Study.By Dr. A. Duggal, et al.
Clinical Evaluation of Deployed Military Personnel with Chronic Respiratory Symptoms: STAMPEDE III (Study of Active Duty Military for Pulmonary Disease Related to Environmental Deployment Exposures). By Dr. M. J. Morris, et al.
Preparing for the COVID-19 Pandemic: Our Experience in New York.By Dr. H. Zubair, et al.
The Utility of Electronic Inhaler Monitoring in COPD Management: Promises and Challenges.By Dr. A. H. Attaway, et al.
Patterns of Use of Adjunctive Therapies in Patients With Early Moderate-Severe Acute Respiratory Distress Syndrome: Insights From the LUNG SAFE Study.By Dr. A. Duggal, et al.
Clinical Evaluation of Deployed Military Personnel with Chronic Respiratory Symptoms: STAMPEDE III (Study of Active Duty Military for Pulmonary Disease Related to Environmental Deployment Exposures). By Dr. M. J. Morris, et al.
Evolving treatment of cystic fibrosis: Path toward a normal lifespan
Cystic fibrosis (CF) is an autosomal recessive disorder affecting thousands of people worldwide. When this genetic disease was first discovered in the first half of the 20th century, the median survival was approximately 5 years of age. Since then, median survival for patients with CF has steadily improved. Today, it is 47.4 years based on Cystic Fibrosis Foundation registry data from 2018. Patients with CF are living longer and staying healthier; the discussion to follow is how patients, researchers, and the CF Foundation reached this point.
In 1938, pediatrician and pathologist Dorothy Anderson observed on the autopsies of children thought to have celiac disease pancreatic lesions she termed “cystic fibrosis of the pancreas.” In addition to the abnormal pancreas, she noted abnormal lungs filled with mucus that obstructed the airways.
Paul Di Sant’Agnese recognized during a heatwave in late 1948 that children with CF were routinely being diagnosed with heatstroke and dehydration. This helped lead to the discovery that these children had elevated salt content in their sweat, paving the way for the development of the sweat chloride test in 1959 by Gibson and Cooke. Not only did Dr. Di Sant’Agnese recognize excess salt in the sweat of patients with CF, but with the help of several concerned parents of children with CF, he established the Cystic Fibrosis Foundation in 1955. The Foundation helped organize the care center model over the next decades, increasing from 30 care centers in 1962 to over 100 in 1978. The care center model also developed a patient registry to track patient care longitudinally.
In June 1989, Francis Collins and Lap-Chee Tsui discovered the location of the CF transmembrane conductance regulator (CFTR) protein using a novel technique called chromosome jumping (Rommens JM, et al. Science. 1989;245[4922]1059). The discovery was a breakthrough in basic science research, but it would take 3 more decades before this discovery could be translated into a medication that could be used by most patients for everyday care.
In the early 1990s, when median survival for patients with CF was 29 years of age, the CF Foundation and Genentech, Inc., coordinated a 24-week multicenter double-blind randomized control trial (RCT) for a new inhaled medication that digested the extracellular DNA from the neutrophils that accumulated in the airways of patients with CF. Inhaled recombinant human DNase in these patients reduced the risk of pulmonary exacerbations and also had a small improvement in pulmonary function in the group compared with the placebo group (Fuchs H, et al. N Engl J Med. 1994;331:637). Five years later, another double-blind RCT demonstrated that inhaled tobramycin in patients with CF whose disease was colonized with Pseudomonas aeruginosa improved pulmonary function and reduced the risk of hospitalizations (Ramsey B, et al. N Engl J Med. 1999;340:23). In 2006, the use of hypertonic saline solution in patients with CF decreased the overall pulmonary exacerbation rate (Elkins MR, et al. N Engl J Med. 2006;354:229). The combination of these inhaled medications, along with inhaled aztreonam, formed the backbone of inhalation therapy for CF care today.
In 1998, even with the ongoing development and approval of new CF medications by the pharmaceutical industry, Robert Beall, CEO of the CF Foundation, realized that he needed to challenge the current drug development paradigm. Instead of trying to convince companies to develop CF medications, he started a concept called venture philanthropy. This concept entailed the CF Foundation financially investing in pharmaceutical companies’ development of new medications. The Foundation first invested in a small company named Aurora Biosciences (known today as Vertex Pharmaceuticals) in 2000. Aurora Biosciences specialized in high throughput screening. This process uses a unique technology allowing one to test the therapeutic reaction of airway cells to thousands of chemical compounds in a single day, instead of using the traditional process of tediously pipetting compounds one by one. Today, the CF Foundation has invested millions of dollars into bioscience research to advance CF care.
In 2011, the results of a study were published in which a small molecule altered defective CFTR protein in patients with CF with the CFTR mutation G551D, thus improving chloride transport at the airway surface. In the original study, after 24 weeks of therapy receiving the medication known as ivacaftor, predicted FEV1 in patients with CF improved 10.6%, and the patients were 55% less likely to have a pulmonary exacerbation compared with those receiving a placebo. This breakthrough provided patients with CF the first medication that could correct the CFTR at the source of the problem (Ramsey BW, et al. N Engl J Med. 2011;365:1663). Ivacaftor was approved by the US FDA in 2012.
Ivacaftor provided proof of concept that using small molecules could improve CFTR function. Ivacaftor was only beneficial to a small percentage of patients and was not effective in patients with CF who had either 1 or 2 F508del CFTR mutations. In 2015, patients with CF with F508del homozygous treated with a combination therapy of lumacaftor/ivacaftor had predicted FEV1% improved 2.6% to 4.0%. More importantly, there was a significant reduction in the number of pulmonary exacerbations per year compared with placebo. Unexpectedly, some of the patients experienced bronchoconstriction while receiving lumacaftor/ivacaftor (Wainwright CE, et al. N Engl J Med. 2015; 373:220). The problem was recognized, and a new small molecule to improve the processing and trafficking of CFTR called tezacaftor was developed. The combination of tezacaftor/ivacaftor in patients with CF who were F508del homozygous demonstrated a similar reduction in pulmonary exacerbations, an absolute improvement of predicted FEV1 of 4%, and no increased respiratory symptoms compared with the placebo arm (Taylor-Cousar JL, et al. N Engl J Med. 2017;377[21]2013).
CFTR modulators were a major breakthrough for patients with CF, but the efficacy of these therapies was dependent on the patients’ genotype and ranged from mildly to moderately effective. Unfortunately, these therapies were ineffective for the patients who were delta 508 heterozygotes. Starting in the summer of 2018, VX 445-tezacaftor-ivacaftor (ETI) was compared with placebo in patients with CF who were 1 copy of F508del and a second CFTR mutation that has minimal function. The study found an absolute improvement in predicted FEV1 of 14.3% and a 63% reduction in exacerbations at 24 weeks compared with placebo (Middleton PG, et al. N Engl J Med. 2019;381:1809). In late 2019, based on these data, ETI was approved by the FDA for all patients with CF who were F508del heterozygous. This innovation provided effective therapy to 90% of the CF population.
With the discovery of many highly effective therapies beneficial in most patients, the CF Foundation started a program called Path to a Cure to find therapies for the 10% of patients with CF who were not candidates for ETI or other CFTR modulators. This program looks to develop novel methods to restore CFTR protein function and repair or replace the CFTR protein via gene editing or gene transfer. This process creates many challenges that are quite complex, but patients, researchers, physicians, and CF Foundation will not stop working until CF stands for CURE FOUND.
Today, patients with CF are living longer, and many are eligible or have already started ETI therapy. This medication and the many others being developed will hopefully lead to patients with CF living a normal lifespan in the near future.
Dr. Finklea is Assistant Professor of Medicine, Division of Pulmonary and Critical Care, University of Texas Southwestern, Dallas, Texas. Dr. Finklea receives grant support from the Cystic Fibrosis Foundation.
Cystic fibrosis (CF) is an autosomal recessive disorder affecting thousands of people worldwide. When this genetic disease was first discovered in the first half of the 20th century, the median survival was approximately 5 years of age. Since then, median survival for patients with CF has steadily improved. Today, it is 47.4 years based on Cystic Fibrosis Foundation registry data from 2018. Patients with CF are living longer and staying healthier; the discussion to follow is how patients, researchers, and the CF Foundation reached this point.
In 1938, pediatrician and pathologist Dorothy Anderson observed on the autopsies of children thought to have celiac disease pancreatic lesions she termed “cystic fibrosis of the pancreas.” In addition to the abnormal pancreas, she noted abnormal lungs filled with mucus that obstructed the airways.
Paul Di Sant’Agnese recognized during a heatwave in late 1948 that children with CF were routinely being diagnosed with heatstroke and dehydration. This helped lead to the discovery that these children had elevated salt content in their sweat, paving the way for the development of the sweat chloride test in 1959 by Gibson and Cooke. Not only did Dr. Di Sant’Agnese recognize excess salt in the sweat of patients with CF, but with the help of several concerned parents of children with CF, he established the Cystic Fibrosis Foundation in 1955. The Foundation helped organize the care center model over the next decades, increasing from 30 care centers in 1962 to over 100 in 1978. The care center model also developed a patient registry to track patient care longitudinally.
In June 1989, Francis Collins and Lap-Chee Tsui discovered the location of the CF transmembrane conductance regulator (CFTR) protein using a novel technique called chromosome jumping (Rommens JM, et al. Science. 1989;245[4922]1059). The discovery was a breakthrough in basic science research, but it would take 3 more decades before this discovery could be translated into a medication that could be used by most patients for everyday care.
In the early 1990s, when median survival for patients with CF was 29 years of age, the CF Foundation and Genentech, Inc., coordinated a 24-week multicenter double-blind randomized control trial (RCT) for a new inhaled medication that digested the extracellular DNA from the neutrophils that accumulated in the airways of patients with CF. Inhaled recombinant human DNase in these patients reduced the risk of pulmonary exacerbations and also had a small improvement in pulmonary function in the group compared with the placebo group (Fuchs H, et al. N Engl J Med. 1994;331:637). Five years later, another double-blind RCT demonstrated that inhaled tobramycin in patients with CF whose disease was colonized with Pseudomonas aeruginosa improved pulmonary function and reduced the risk of hospitalizations (Ramsey B, et al. N Engl J Med. 1999;340:23). In 2006, the use of hypertonic saline solution in patients with CF decreased the overall pulmonary exacerbation rate (Elkins MR, et al. N Engl J Med. 2006;354:229). The combination of these inhaled medications, along with inhaled aztreonam, formed the backbone of inhalation therapy for CF care today.
In 1998, even with the ongoing development and approval of new CF medications by the pharmaceutical industry, Robert Beall, CEO of the CF Foundation, realized that he needed to challenge the current drug development paradigm. Instead of trying to convince companies to develop CF medications, he started a concept called venture philanthropy. This concept entailed the CF Foundation financially investing in pharmaceutical companies’ development of new medications. The Foundation first invested in a small company named Aurora Biosciences (known today as Vertex Pharmaceuticals) in 2000. Aurora Biosciences specialized in high throughput screening. This process uses a unique technology allowing one to test the therapeutic reaction of airway cells to thousands of chemical compounds in a single day, instead of using the traditional process of tediously pipetting compounds one by one. Today, the CF Foundation has invested millions of dollars into bioscience research to advance CF care.
In 2011, the results of a study were published in which a small molecule altered defective CFTR protein in patients with CF with the CFTR mutation G551D, thus improving chloride transport at the airway surface. In the original study, after 24 weeks of therapy receiving the medication known as ivacaftor, predicted FEV1 in patients with CF improved 10.6%, and the patients were 55% less likely to have a pulmonary exacerbation compared with those receiving a placebo. This breakthrough provided patients with CF the first medication that could correct the CFTR at the source of the problem (Ramsey BW, et al. N Engl J Med. 2011;365:1663). Ivacaftor was approved by the US FDA in 2012.
Ivacaftor provided proof of concept that using small molecules could improve CFTR function. Ivacaftor was only beneficial to a small percentage of patients and was not effective in patients with CF who had either 1 or 2 F508del CFTR mutations. In 2015, patients with CF with F508del homozygous treated with a combination therapy of lumacaftor/ivacaftor had predicted FEV1% improved 2.6% to 4.0%. More importantly, there was a significant reduction in the number of pulmonary exacerbations per year compared with placebo. Unexpectedly, some of the patients experienced bronchoconstriction while receiving lumacaftor/ivacaftor (Wainwright CE, et al. N Engl J Med. 2015; 373:220). The problem was recognized, and a new small molecule to improve the processing and trafficking of CFTR called tezacaftor was developed. The combination of tezacaftor/ivacaftor in patients with CF who were F508del homozygous demonstrated a similar reduction in pulmonary exacerbations, an absolute improvement of predicted FEV1 of 4%, and no increased respiratory symptoms compared with the placebo arm (Taylor-Cousar JL, et al. N Engl J Med. 2017;377[21]2013).
CFTR modulators were a major breakthrough for patients with CF, but the efficacy of these therapies was dependent on the patients’ genotype and ranged from mildly to moderately effective. Unfortunately, these therapies were ineffective for the patients who were delta 508 heterozygotes. Starting in the summer of 2018, VX 445-tezacaftor-ivacaftor (ETI) was compared with placebo in patients with CF who were 1 copy of F508del and a second CFTR mutation that has minimal function. The study found an absolute improvement in predicted FEV1 of 14.3% and a 63% reduction in exacerbations at 24 weeks compared with placebo (Middleton PG, et al. N Engl J Med. 2019;381:1809). In late 2019, based on these data, ETI was approved by the FDA for all patients with CF who were F508del heterozygous. This innovation provided effective therapy to 90% of the CF population.
With the discovery of many highly effective therapies beneficial in most patients, the CF Foundation started a program called Path to a Cure to find therapies for the 10% of patients with CF who were not candidates for ETI or other CFTR modulators. This program looks to develop novel methods to restore CFTR protein function and repair or replace the CFTR protein via gene editing or gene transfer. This process creates many challenges that are quite complex, but patients, researchers, physicians, and CF Foundation will not stop working until CF stands for CURE FOUND.
Today, patients with CF are living longer, and many are eligible or have already started ETI therapy. This medication and the many others being developed will hopefully lead to patients with CF living a normal lifespan in the near future.
Dr. Finklea is Assistant Professor of Medicine, Division of Pulmonary and Critical Care, University of Texas Southwestern, Dallas, Texas. Dr. Finklea receives grant support from the Cystic Fibrosis Foundation.
Cystic fibrosis (CF) is an autosomal recessive disorder affecting thousands of people worldwide. When this genetic disease was first discovered in the first half of the 20th century, the median survival was approximately 5 years of age. Since then, median survival for patients with CF has steadily improved. Today, it is 47.4 years based on Cystic Fibrosis Foundation registry data from 2018. Patients with CF are living longer and staying healthier; the discussion to follow is how patients, researchers, and the CF Foundation reached this point.
In 1938, pediatrician and pathologist Dorothy Anderson observed on the autopsies of children thought to have celiac disease pancreatic lesions she termed “cystic fibrosis of the pancreas.” In addition to the abnormal pancreas, she noted abnormal lungs filled with mucus that obstructed the airways.
Paul Di Sant’Agnese recognized during a heatwave in late 1948 that children with CF were routinely being diagnosed with heatstroke and dehydration. This helped lead to the discovery that these children had elevated salt content in their sweat, paving the way for the development of the sweat chloride test in 1959 by Gibson and Cooke. Not only did Dr. Di Sant’Agnese recognize excess salt in the sweat of patients with CF, but with the help of several concerned parents of children with CF, he established the Cystic Fibrosis Foundation in 1955. The Foundation helped organize the care center model over the next decades, increasing from 30 care centers in 1962 to over 100 in 1978. The care center model also developed a patient registry to track patient care longitudinally.
In June 1989, Francis Collins and Lap-Chee Tsui discovered the location of the CF transmembrane conductance regulator (CFTR) protein using a novel technique called chromosome jumping (Rommens JM, et al. Science. 1989;245[4922]1059). The discovery was a breakthrough in basic science research, but it would take 3 more decades before this discovery could be translated into a medication that could be used by most patients for everyday care.
In the early 1990s, when median survival for patients with CF was 29 years of age, the CF Foundation and Genentech, Inc., coordinated a 24-week multicenter double-blind randomized control trial (RCT) for a new inhaled medication that digested the extracellular DNA from the neutrophils that accumulated in the airways of patients with CF. Inhaled recombinant human DNase in these patients reduced the risk of pulmonary exacerbations and also had a small improvement in pulmonary function in the group compared with the placebo group (Fuchs H, et al. N Engl J Med. 1994;331:637). Five years later, another double-blind RCT demonstrated that inhaled tobramycin in patients with CF whose disease was colonized with Pseudomonas aeruginosa improved pulmonary function and reduced the risk of hospitalizations (Ramsey B, et al. N Engl J Med. 1999;340:23). In 2006, the use of hypertonic saline solution in patients with CF decreased the overall pulmonary exacerbation rate (Elkins MR, et al. N Engl J Med. 2006;354:229). The combination of these inhaled medications, along with inhaled aztreonam, formed the backbone of inhalation therapy for CF care today.
In 1998, even with the ongoing development and approval of new CF medications by the pharmaceutical industry, Robert Beall, CEO of the CF Foundation, realized that he needed to challenge the current drug development paradigm. Instead of trying to convince companies to develop CF medications, he started a concept called venture philanthropy. This concept entailed the CF Foundation financially investing in pharmaceutical companies’ development of new medications. The Foundation first invested in a small company named Aurora Biosciences (known today as Vertex Pharmaceuticals) in 2000. Aurora Biosciences specialized in high throughput screening. This process uses a unique technology allowing one to test the therapeutic reaction of airway cells to thousands of chemical compounds in a single day, instead of using the traditional process of tediously pipetting compounds one by one. Today, the CF Foundation has invested millions of dollars into bioscience research to advance CF care.
In 2011, the results of a study were published in which a small molecule altered defective CFTR protein in patients with CF with the CFTR mutation G551D, thus improving chloride transport at the airway surface. In the original study, after 24 weeks of therapy receiving the medication known as ivacaftor, predicted FEV1 in patients with CF improved 10.6%, and the patients were 55% less likely to have a pulmonary exacerbation compared with those receiving a placebo. This breakthrough provided patients with CF the first medication that could correct the CFTR at the source of the problem (Ramsey BW, et al. N Engl J Med. 2011;365:1663). Ivacaftor was approved by the US FDA in 2012.
Ivacaftor provided proof of concept that using small molecules could improve CFTR function. Ivacaftor was only beneficial to a small percentage of patients and was not effective in patients with CF who had either 1 or 2 F508del CFTR mutations. In 2015, patients with CF with F508del homozygous treated with a combination therapy of lumacaftor/ivacaftor had predicted FEV1% improved 2.6% to 4.0%. More importantly, there was a significant reduction in the number of pulmonary exacerbations per year compared with placebo. Unexpectedly, some of the patients experienced bronchoconstriction while receiving lumacaftor/ivacaftor (Wainwright CE, et al. N Engl J Med. 2015; 373:220). The problem was recognized, and a new small molecule to improve the processing and trafficking of CFTR called tezacaftor was developed. The combination of tezacaftor/ivacaftor in patients with CF who were F508del homozygous demonstrated a similar reduction in pulmonary exacerbations, an absolute improvement of predicted FEV1 of 4%, and no increased respiratory symptoms compared with the placebo arm (Taylor-Cousar JL, et al. N Engl J Med. 2017;377[21]2013).
CFTR modulators were a major breakthrough for patients with CF, but the efficacy of these therapies was dependent on the patients’ genotype and ranged from mildly to moderately effective. Unfortunately, these therapies were ineffective for the patients who were delta 508 heterozygotes. Starting in the summer of 2018, VX 445-tezacaftor-ivacaftor (ETI) was compared with placebo in patients with CF who were 1 copy of F508del and a second CFTR mutation that has minimal function. The study found an absolute improvement in predicted FEV1 of 14.3% and a 63% reduction in exacerbations at 24 weeks compared with placebo (Middleton PG, et al. N Engl J Med. 2019;381:1809). In late 2019, based on these data, ETI was approved by the FDA for all patients with CF who were F508del heterozygous. This innovation provided effective therapy to 90% of the CF population.
With the discovery of many highly effective therapies beneficial in most patients, the CF Foundation started a program called Path to a Cure to find therapies for the 10% of patients with CF who were not candidates for ETI or other CFTR modulators. This program looks to develop novel methods to restore CFTR protein function and repair or replace the CFTR protein via gene editing or gene transfer. This process creates many challenges that are quite complex, but patients, researchers, physicians, and CF Foundation will not stop working until CF stands for CURE FOUND.
Today, patients with CF are living longer, and many are eligible or have already started ETI therapy. This medication and the many others being developed will hopefully lead to patients with CF living a normal lifespan in the near future.
Dr. Finklea is Assistant Professor of Medicine, Division of Pulmonary and Critical Care, University of Texas Southwestern, Dallas, Texas. Dr. Finklea receives grant support from the Cystic Fibrosis Foundation.